WO2012133533A1 - Qol改善又は持続剤 - Google Patents
Qol改善又は持続剤 Download PDFInfo
- Publication number
- WO2012133533A1 WO2012133533A1 PCT/JP2012/058145 JP2012058145W WO2012133533A1 WO 2012133533 A1 WO2012133533 A1 WO 2012133533A1 JP 2012058145 W JP2012058145 W JP 2012058145W WO 2012133533 A1 WO2012133533 A1 WO 2012133533A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- agent
- improving
- qol
- fatigue
- maintaining
- Prior art date
Links
- 241000186660 Lactobacillus Species 0.000 claims abstract description 79
- 229940039696 lactobacillus Drugs 0.000 claims abstract description 79
- 230000036541 health Effects 0.000 claims abstract description 76
- 230000002929 anti-fatigue Effects 0.000 claims abstract description 21
- 239000003795 chemical substances by application Substances 0.000 description 107
- 206010016256 fatigue Diseases 0.000 description 41
- 238000012360 testing method Methods 0.000 description 41
- 230000000694 effects Effects 0.000 description 34
- 230000006872 improvement Effects 0.000 description 32
- 241000894006 Bacteria Species 0.000 description 30
- 235000013305 food Nutrition 0.000 description 27
- 241001465754 Metazoa Species 0.000 description 25
- 238000012423 maintenance Methods 0.000 description 24
- 238000000034 method Methods 0.000 description 21
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 20
- 239000003814 drug Substances 0.000 description 15
- 238000011084 recovery Methods 0.000 description 15
- 239000000203 mixture Substances 0.000 description 13
- 235000014655 lactic acid Nutrition 0.000 description 10
- 239000004310 lactic acid Substances 0.000 description 10
- 239000000843 powder Substances 0.000 description 10
- 239000000047 product Substances 0.000 description 10
- 230000009471 action Effects 0.000 description 7
- 235000013361 beverage Nutrition 0.000 description 7
- 230000007423 decrease Effects 0.000 description 7
- 239000000654 additive Substances 0.000 description 6
- 230000003247 decreasing effect Effects 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- 230000006866 deterioration Effects 0.000 description 5
- 238000011156 evaluation Methods 0.000 description 5
- 230000000813 microbial effect Effects 0.000 description 5
- 229940068196 placebo Drugs 0.000 description 5
- 239000000902 placebo Substances 0.000 description 5
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 4
- 229920002472 Starch Polymers 0.000 description 4
- 238000012258 culturing Methods 0.000 description 4
- 239000003937 drug carrier Substances 0.000 description 4
- 230000002708 enhancing effect Effects 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 239000002609 medium Substances 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 239000008107 starch Substances 0.000 description 4
- 235000019698 starch Nutrition 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- 239000004375 Dextrin Substances 0.000 description 3
- 229920001353 Dextrin Polymers 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 230000000996 additive effect Effects 0.000 description 3
- 239000012736 aqueous medium Substances 0.000 description 3
- 230000001580 bacterial effect Effects 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 235000009508 confectionery Nutrition 0.000 description 3
- 235000019425 dextrin Nutrition 0.000 description 3
- 235000003599 food sweetener Nutrition 0.000 description 3
- 230000001965 increasing effect Effects 0.000 description 3
- 230000001788 irregular Effects 0.000 description 3
- 239000000314 lubricant Substances 0.000 description 3
- 230000003340 mental effect Effects 0.000 description 3
- 230000004630 mental health Effects 0.000 description 3
- 229920001542 oligosaccharide Polymers 0.000 description 3
- 150000002482 oligosaccharides Chemical class 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 230000035882 stress Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- 239000003765 sweetening agent Substances 0.000 description 3
- 239000002562 thickening agent Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 241000195940 Bryophyta Species 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 229910000019 calcium carbonate Inorganic materials 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 235000008504 concentrate Nutrition 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 235000013365 dairy product Nutrition 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 235000013399 edible fruits Nutrition 0.000 description 2
- 238000000855 fermentation Methods 0.000 description 2
- 230000004151 fermentation Effects 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- 238000004108 freeze drying Methods 0.000 description 2
- 235000015203 fruit juice Nutrition 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 238000000265 homogenisation Methods 0.000 description 2
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 2
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 2
- 230000001771 impaired effect Effects 0.000 description 2
- 235000015110 jellies Nutrition 0.000 description 2
- 239000008274 jelly Substances 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 235000012054 meals Nutrition 0.000 description 2
- -1 methylthicellulose Polymers 0.000 description 2
- 235000013336 milk Nutrition 0.000 description 2
- 239000008267 milk Substances 0.000 description 2
- 210000004080 milk Anatomy 0.000 description 2
- 230000000116 mitigating effect Effects 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 235000011929 mousse Nutrition 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- 230000008520 organization Effects 0.000 description 2
- 230000003204 osmotic effect Effects 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 239000012264 purified product Substances 0.000 description 2
- 235000013322 soy milk Nutrition 0.000 description 2
- 238000001694 spray drying Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 229910021654 trace metal Inorganic materials 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- 235000013311 vegetables Nutrition 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 235000013618 yogurt Nutrition 0.000 description 2
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- VZSRBBMJRBPUNF-UHFFFAOYSA-N 2-(2,3-dihydro-1H-inden-2-ylamino)-N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]pyrimidine-5-carboxamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C(=O)NCCC(N1CC2=C(CC1)NN=N2)=O VZSRBBMJRBPUNF-UHFFFAOYSA-N 0.000 description 1
- MIDXCONKKJTLDX-UHFFFAOYSA-N 3,5-dimethylcyclopentane-1,2-dione Chemical compound CC1CC(C)C(=O)C1=O MIDXCONKKJTLDX-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- WBZFUFAFFUEMEI-UHFFFAOYSA-M Acesulfame k Chemical compound [K+].CC1=CC(=O)[N-]S(=O)(=O)O1 WBZFUFAFFUEMEI-UHFFFAOYSA-M 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 241000193830 Bacillus <bacterium> Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 238000001061 Dunnett's test Methods 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 208000002979 Influenza in Birds Diseases 0.000 description 1
- 238000001265 Jonckheere trend test Methods 0.000 description 1
- 241000186684 Lactobacillus pentosus Species 0.000 description 1
- 240000006024 Lactobacillus plantarum Species 0.000 description 1
- 235000013965 Lactobacillus plantarum Nutrition 0.000 description 1
- 229920001543 Laminarin Polymers 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000001888 Peptone Substances 0.000 description 1
- 108010080698 Peptones Proteins 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 244000228451 Stevia rebaudiana Species 0.000 description 1
- 239000004376 Sucralose Substances 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 229930003270 Vitamin B Natural products 0.000 description 1
- 229930003268 Vitamin C Natural products 0.000 description 1
- 229930003316 Vitamin D Natural products 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- 229930003448 Vitamin K Natural products 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 239000000619 acesulfame-K Substances 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 235000010419 agar Nutrition 0.000 description 1
- 235000013334 alcoholic beverage Nutrition 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 239000000305 astragalus gummifer gum Substances 0.000 description 1
- 206010064097 avian influenza Diseases 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- VBICKXHEKHSIBG-UHFFFAOYSA-N beta-monoglyceryl stearate Natural products CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 1
- 235000015895 biscuits Nutrition 0.000 description 1
- 235000008429 bread Nutrition 0.000 description 1
- 229940041514 candida albicans extract Drugs 0.000 description 1
- 235000013736 caramel Nutrition 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 235000014171 carbonated beverage Nutrition 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 229920003123 carboxymethyl cellulose sodium Polymers 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 229940105329 carboxymethylcellulose Drugs 0.000 description 1
- 229940084030 carboxymethylcellulose calcium Drugs 0.000 description 1
- 229940063834 carboxymethylcellulose sodium Drugs 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 235000013339 cereals Nutrition 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 235000013351 cheese Nutrition 0.000 description 1
- 235000014510 cooky Nutrition 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 238000012136 culture method Methods 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 235000015872 dietary supplement Nutrition 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 238000005187 foaming Methods 0.000 description 1
- 235000012041 food component Nutrition 0.000 description 1
- FTSSQIKWUOOEGC-RULYVFMPSA-N fructooligosaccharide Chemical compound OC[C@H]1O[C@@](CO)(OC[C@@]2(OC[C@@]3(OC[C@@]4(OC[C@@]5(OC[C@@]6(OC[C@@]7(OC[C@@]8(OC[C@@]9(OC[C@@]%10(OC[C@@]%11(O[C@H]%12O[C@H](CO)[C@@H](O)[C@H](O)[C@H]%12O)O[C@H](CO)[C@@H](O)[C@@H]%11O)O[C@H](CO)[C@@H](O)[C@@H]%10O)O[C@H](CO)[C@@H](O)[C@@H]9O)O[C@H](CO)[C@@H](O)[C@@H]8O)O[C@H](CO)[C@@H](O)[C@@H]7O)O[C@H](CO)[C@@H](O)[C@@H]6O)O[C@H](CO)[C@@H](O)[C@@H]5O)O[C@H](CO)[C@@H](O)[C@@H]4O)O[C@H](CO)[C@@H](O)[C@@H]3O)O[C@H](CO)[C@@H](O)[C@@H]2O)[C@@H](O)[C@@H]1O FTSSQIKWUOOEGC-RULYVFMPSA-N 0.000 description 1
- 229940107187 fructooligosaccharide Drugs 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 235000021255 galacto-oligosaccharides Nutrition 0.000 description 1
- 150000003271 galactooligosaccharides Chemical class 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 230000003862 health status Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229940071676 hydroxypropylcellulose Drugs 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 239000000832 lactitol Substances 0.000 description 1
- VQHSOMBJVWLPSR-JVCRWLNRSA-N lactitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-JVCRWLNRSA-N 0.000 description 1
- 235000010448 lactitol Nutrition 0.000 description 1
- 229960003451 lactitol Drugs 0.000 description 1
- 229940072205 lactobacillus plantarum Drugs 0.000 description 1
- JCQLYHFGKNRPGE-FCVZTGTOSA-N lactulose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 JCQLYHFGKNRPGE-FCVZTGTOSA-N 0.000 description 1
- 229960000511 lactulose Drugs 0.000 description 1
- PFCRQPBOOFTZGQ-UHFFFAOYSA-N lactulose keto form Natural products OCC(=O)C(O)C(C(O)CO)OC1OC(CO)C(O)C(O)C1O PFCRQPBOOFTZGQ-UHFFFAOYSA-N 0.000 description 1
- DBTMGCOVALSLOR-VPNXCSTESA-N laminarin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)OC1O[C@@H]1[C@@H](O)C(O[C@H]2[C@@H]([C@@H](CO)OC(O)[C@@H]2O)O)O[C@H](CO)[C@H]1O DBTMGCOVALSLOR-VPNXCSTESA-N 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 229940031703 low substituted hydroxypropyl cellulose Drugs 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 239000003094 microcapsule Substances 0.000 description 1
- 235000020124 milk-based beverage Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 239000006872 mrs medium Substances 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 235000012149 noodles Nutrition 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 229960000292 pectin Drugs 0.000 description 1
- 235000019319 peptone Nutrition 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- SHUZOJHMOBOZST-UHFFFAOYSA-N phylloquinone Natural products CC(C)CCCCC(C)CCC(C)CCCC(=CCC1=C(C)C(=O)c2ccccc2C1=O)C SHUZOJHMOBOZST-UHFFFAOYSA-N 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 229940068984 polyvinyl alcohol Drugs 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- 235000011962 puddings Nutrition 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- HELXLJCILKEWJH-NCGAPWICSA-N rebaudioside A Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HELXLJCILKEWJH-NCGAPWICSA-N 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 235000019685 rice crackers Nutrition 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 235000020374 simple syrup Nutrition 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 235000014214 soft drink Nutrition 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000014347 soups Nutrition 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 235000019408 sucralose Nutrition 0.000 description 1
- BAQAVOSOZGMPRM-QBMZZYIRSA-N sucralose Chemical compound O[C@@H]1[C@@H](O)[C@@H](Cl)[C@@H](CO)O[C@@H]1O[C@@]1(CCl)[C@@H](O)[C@H](O)[C@@H](CCl)O1 BAQAVOSOZGMPRM-QBMZZYIRSA-N 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000013616 tea Nutrition 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 238000009210 therapy by ultrasound Methods 0.000 description 1
- 235000019156 vitamin B Nutrition 0.000 description 1
- 239000011720 vitamin B Substances 0.000 description 1
- 235000019154 vitamin C Nutrition 0.000 description 1
- 239000011718 vitamin C Substances 0.000 description 1
- 235000019166 vitamin D Nutrition 0.000 description 1
- 239000011710 vitamin D Substances 0.000 description 1
- 150000003710 vitamin D derivatives Chemical class 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 235000019168 vitamin K Nutrition 0.000 description 1
- 239000011712 vitamin K Substances 0.000 description 1
- 150000003721 vitamin K derivatives Chemical class 0.000 description 1
- 229940046008 vitamin d Drugs 0.000 description 1
- 229940046010 vitamin k Drugs 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
- 239000012138 yeast extract Substances 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/66—Microorganisms or materials therefrom
- A61K35/74—Bacteria
- A61K35/741—Probiotics
- A61K35/744—Lactic acid bacteria, e.g. enterococci, pediococci, lactococci, streptococci or leuconostocs
- A61K35/747—Lactobacilli, e.g. L. acidophilus or L. brevis
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/135—Bacteria or derivatives thereof, e.g. probiotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/66—Microorganisms or materials therefrom
- A61K35/74—Bacteria
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
Definitions
- the present invention relates to an improvement or sustaining agent of quality of life (QOL).
- QOL Quality of life
- Lactobacillus ONRICb0240 (FERM BP-10065) strain which is a kind of lactic acid bacteria, has a mucosal immunity stimulating action, particularly an IgA production promoting action (Patent Documents 1 and 2) and an anti-avian influenza antibody production promoting action (Patent Document 3). It is known to have However, regarding the lactic acid bacteria, there is no report on improvement of physical health and improvement of QOL based thereon.
- the present invention provides a QOL improving or maintaining agent using Lactobacillus ONRICb0240 (FERM BP-10065) strain (hereinafter sometimes referred to as Lactobacillus b0240 strain), particularly a physical health improving or maintaining agent.
- Lactobacillus b0240 strain particularly a physical health improving or maintaining agent.
- Another object of the present invention is to provide a vitality improving or maintaining agent, a fatigue recovery or relaxation agent, and an anti-fatigue agent using Lactobacillus ONRICb0240 strain.
- the Lactobacillus ONRICb0240 strain has an effect of enhancing physical health, particularly an activity of increasing vitality and reducing fatigue, and an effect of reducing and alleviating fatigue. I found out. Further, the present inventors have found that the physical health can be improved by the action, and therefore QOL can be improved from the physical aspect. The present invention has been completed by further studies based on these findings. That is, the present invention provides the following inventions. (1) Improvement or maintenance of QOL Item 1-1. A QOL improving or maintaining agent comprising Lactobacillus ONRICb0240 (FERM BP-10065). Item 1-2. Item 10.
- the QOL improving or maintaining agent according to Item 1-1 which contains 104 cells / mg or more of Lactobacillus ONRICb0240 (FERM BP-10065).
- the QOL improving or maintaining agent according to Item 1-1 which comprises 104 cells or more of Lactobacillus ONRICb0240 (FERM BP-10065).
- Item 1-4. A food or drink or pharmaceutical comprising the QOL improving or maintaining agent according to any one of Items 1-1 to 1-3.
- Item 1-5 A method for improving or maintaining QOL of an animal, comprising a step of allowing an animal that is required to improve or maintain QOL to ingest the QOL improving or maintaining agent according to any one of Items 1-1 to 1-3.
- a method for improving or maintaining QOL of an animal comprising a step of allowing an animal that is required to improve or maintain QOL to take the food or drink or medicine according to Item 1-4.
- (2) Improving or maintaining physical health Item 2-1.
- An agent for improving or maintaining physical health comprising Lactobacillus ONRICb0240 (FERM BP-10065).
- the physical health improving or maintaining agent according to Item 2-1 which contains 104 cells or more of Lactobacillus ONRICb0240 (FERM BP-10065).
- Item 2-4 A food or drink or a pharmaceutical comprising the physical health improving or maintaining agent according to any one of Items 2-1 to 2-3.
- Item 2-5 Improving or maintaining the physical health of the animal, comprising a step of ingesting the physical health improving or maintaining agent according to any one of Items 2-1 to 2-3 to an animal that is required to improve or maintain the physical health.
- Item 2-6 A method for improving or maintaining the physical health of an animal, comprising a step of allowing an animal that is required to improve or maintain physical health to ingest the food or drink or medicine according to Item 2-4.
- Item 2-7 A method for improving or maintaining the physical health of an animal, comprising a step of allowing an animal that is required to improve or maintain physical health to ingest the food or drink or medicine according to Item 2-4.
- Lactobacillus ONRICb0240 (FERM BP-10065) to produce a physical health improving or maintaining agent.
- (3) Improving or maintaining vitality Item 3-1.
- a vitality improving or maintaining agent comprising Lactobacillus ONRICb0240 (FERM BP-10065).
- the vitality improving or maintaining agent according to item 3-1 comprising Lactobacillus ONRICb0240 (FERM BP-10065) at 104 cells / mg or more.
- Item 3-3 Item 3.
- the vitality improving or maintaining agent according to item 3-1 comprising Lactobacillus ONRICb0240 (FERM BP-10065) in an amount of 104 cells or more.
- a food or drink or a pharmaceutical comprising the vitality improving or maintaining agent according to any one of Items 3-1 to 3-3.
- Item 3-5 A method for improving or maintaining the vitality of an animal, comprising a step of causing an animal that is required to improve or maintain the vitality to ingest the vitality improving or maintaining agent according to any one of Items 3-1 to 3-3.
- Item 3-6 A method for improving or maintaining the vitality of an animal, comprising a step of allowing an animal that is required to improve or maintain the vitality to ingest the food or drink or the medicine according to Item 3-4.
- Item 3-7 Use of Lactobacillus ONRICb0240 (FERM BP-10065) to produce a vitality improving or maintaining agent.
- a method for recovering or relieving fatigue of an animal comprising a step of allowing an animal that is required to recover or relieve fatigue to take the fatigue recovery or relieving agent according to any one of Items 4-1 to 4-3.
- Item 5. A method for recovering or relieving fatigue of an animal, comprising a step of allowing an animal that is required to recover or relieve fatigue to take the food or drink or medicine according to Item 4-4.
- Use of Lactobacillus ONRICb0240 (FERM BP-10065) to produce fatigue recovery or relaxation agents.
- Anti-fatigue Item 5-1 An anti-fatigue agent comprising Lactobacillus ONRICb0240 (FERM BP-10065).
- the anti-fatigue agent according to Item 5-1 comprising 104 cells / mg or more of Lactobacillus ONRICb0240 (FERM BP-10065).
- Item 5-3 Item 5.
- the anti-fatigue agent according to Item 5-1 comprising 104 cells or more of Lactobacillus ONRICb0240 (FERM BP-10065).
- Item 5-4 Item 5.
- a food or drink or pharmaceutical comprising the anti-fatigue agent according to any one of Items 5-1 to 5-3.
- Item 5-5 An anti-fatigue method for an animal, comprising a step of ingesting an anti-fatigue agent according to any one of Items 5-1 to 5-3 to an animal that requires anti-fatigue.
- Item 5-6 An anti-fatigue method for an animal, comprising a step of ingesting an anti-fatigue agent according to any one of Items 5-1 to 5-3 to an animal that requires anti-fatigue.
- a method for anti-fatigue of an animal comprising a step of causing an animal that requires anti-fatigue to ingest the food or drink or the medicine according to Item 5-4.
- the QOL improvement or maintenance agent of the present invention can improve or maintain the QOL that is important today.
- Lactobacillus ONRICb0240 strain has an effect of enhancing physical health.
- the QOL improving or maintaining agent of the present invention can improve or maintain QOL particularly from the physical aspect.
- the QOL improving or maintaining agent of the present invention is particularly useful as a physical health improving or maintaining agent.
- the Lactobacillus ONRICb0240 strain has an action of enhancing vitality, an action of making fatigue less likely to occur, and an action of reducing and mitigating fatigue.
- the QOL improvement or maintenance agent of the present invention is further useful as a vitality improvement or maintenance agent, fatigue recovery agent, fatigue relaxation agent, particularly physical fatigue recovery agent, physical fatigue relaxation agent, and anti-fatigue agent. is there.
- the present invention as described above, it is possible to prevent or reduce the deterioration of physical health caused by lack of rest and sleep, irregular meals, lack of exercise, intense exercise, aging, disturbance of life factors such as stress, etc. .
- the present invention can improve or maintain physical health from the above viewpoint regardless of age or gender.
- the QOL improving or maintaining agent of the present invention is characterized by containing a Lactobacillus ONRICb0240 (FERM BP-10065) strain as an active ingredient.
- the QOL improving or maintaining agent of the present invention can be used particularly as a physical health improving or maintaining agent, vitality improving or maintaining agent, fatigue recovery agent, fatigue relieving agent, anti-fatigue agent and the like. For this reason, these are similarly characterized by containing Lactobacillus ONRICb0240 strain as an active ingredient.
- the Lactobacillus ONRICb0240 strain used in the present invention is a lactic acid bacterium isolated from a natural product, and has an address at 1st Central, 1st Street, 1st Street, Tsukuba, Ibaraki, Japan on August 6, 2003. Deposited at the Patent Organism Depositary (IPOD), National Institute of Advanced Industrial Science and Technology as deposit number FERM P-19470, and currently transferred to international deposit, the international deposit number is FERM BP-10065.
- the mycological properties of Lactobacillus ONRICb0240 strain are known.
- Lactobacillus ONRICb0240 strain used in the present invention was classified as belonging to Lactobacillus plantarum at the time of international deposit, but the subsequent change in classification criteria (Francois Bringle et al. , International Journal of Systematic and Evolutionary Microbiology, Vol. 55, 2005, p.1629-1634), this strain was classified as Lactobacillus pentosus.
- QOL improvement or maintenance agent of the present invention Physical health improvement or maintenance agent, vitality improvement or maintenance agent, fatigue recovery agent, fatigue relieving agent, anti-fatigue agent, etc. (hereinafter sometimes referred to as QOL improvement or maintenance agent, etc.) )
- Contained in Lactobacillus ONRICb0240 strain may be in the state of live bacteria, may be in the state of dead bacteria, or may be a treated product of the cells, or in a state where these are mixed There may be.
- a living bacterium is a lactic acid bacterium in a living state, and a lactic acid bacterium culture solution, a suspension of the culture solution, a crudely purified product, a purified product, or a living lactic acid bacterium is freeze-dried or spray-dried. Also included are cell powders and the like dried by the like, and are not limited as long as they are alive.
- a dead bacteria is a lactic acid bacterium that has been sterilized by subjecting the lactic acid bacterium in a living state to physical or chemical treatment such as heat treatment or radiation treatment, and the sterilized lactic acid bacterium is freeze-dried or sprayed.
- the microbial cell processed product is a microbial cell disruption product obtained by destroying lactic acid bacteria by homogenization treatment, enzyme treatment, ultrasonic treatment, etc., and the microbial cell disruption product obtained by drying the microbial cell disruption product by freeze drying, spray drying, or the like.
- product powders are also included.
- the Lactobacillus ONRICb0240 strain contained in the present invention is preferably in a state of viable bacteria, dead bacteria, treated cells or a mixture thereof, and more preferably in the state of dead bacteria or a mixture of live bacteria and dead bacteria. More preferably, there is a state of dead bacteria.
- the Lactobacillus ONRICb0240 strain used for the QOL improvement or maintenance agent of the present invention can be grown by culturing in a medium suitable for the growth of the strain.
- the culture method is not limited, for example, the Lactobacillus ONRICb0240 strain can be grown by culturing at 30 ° C. for about 16 hours in a medium such as MRS medium, LBS medium, or Rogosa medium.
- the cells can be collected after culturing, for example, by centrifuging the culture (culture solution) (for example, 3,000 rpm, 4 ° C., 10 minutes).
- the Lactobacillus ONRICb0240 strain used in the present invention may be cultured (fermented) in the presence of materials such as milk, vegetables, fruits, soy milk and the like.
- the cells can be collected by centrifuging after culturing.
- the culture (fermented product) thus obtained, the collected bacterial cells, the culture or the suspension or concentrate of the bacterial cells, and the culture thus obtained.
- microbial cells, suspensions, and concentrates which are dried by freeze drying, spray drying, or the like and powdered can also be used. These preparations can be performed according to methods known in the art.
- cultivation (fermentation) is performed more efficiently, it is preferable that raw materials, such as milk, vegetables, fruits, and soy milk, before fermentation have fixed fluidity more than fixed.
- the QOL improving or maintaining agent of the present invention may contain the Lactobacillus ONRICb0240 strain as an active ingredient. Therefore, for example, the culture may be used as it is or after being subjected to a treatment such as homogenization and used as a QOL improving or maintaining agent, or the prepared product may be used as a QOL improving or maintaining agent or the like. .
- the growth of the Lactobacillus ONRICb0240 strain is further performed as necessary from the viewpoint of maintaining the living state better.
- a nutritional component suitable for the QOL improvement or maintenance agent include carbon sources such as glucose, starch, sucrose, lactose, dextrin, sorbitol, and fructose; nitrogen sources such as yeast extract and peptone; vitamins; minerals; trace metal elements; These components can be mentioned.
- vitamins include vitamin B, vitamin D, vitamin C, vitamin E, vitamin K and the like.
- Specific examples of the trace metal element include zinc and selenium.
- oligosaccharides such as dairy oligosaccharide, soybean oligosaccharide, lactulose, lactitol, fructooligosaccharide, and galactooligosaccharide.
- the QOL improving or maintaining agent of the present invention can further contain optional components as necessary.
- the optional components include edible or pharmaceutically acceptable carriers and additives.
- Edible or pharmaceutically acceptable carriers and additives include aqueous media, excipients, binders, disintegrants, lubricants, thickeners, surfactants, osmotic pressure regulators, wetting agents, Examples thereof include pH adjusters, sweeteners, fragrances, and pigments. These are known by those skilled in the art, and are appropriately selected and used.
- aqueous media such as water, physiological saline, fruit juice; lactose, sucrose, sodium chloride, glucose, urea, starch, calcium carbonate, kaolin, crystalline cellulose, silicic acid, potassium phosphate, Excipients such as corn starch, dextrin; binders such as water, ethanol, propanol, simple syrup, dextrose solution, starch solution, gelatin solution, carboxymethylcellulose, hydroxypropylcellulose, methylcellulose, polyvinylpyrrolidone; carboxymethylcellulose sodium, carboxymethylcellulose calcium , Low-substituted hydroxypropylcellulose, dry starch, sodium alginate, agar powder, laminaran powder, sodium bicarbonate, calcium carbonate, etc .; refined talc, stearate, phosphate Lubricants such as acid powder and polyethylene glycol; thickeners such as gelatin, gum arabic, dextrin, methylthicellulose, polyviny
- the components used as necessary can be appropriately selected by those skilled in the art, and the blending amount of the components is within a range that does not impede the effects of the present invention so as to conform to the target form, taste, and the like. Any adjustment may be made as appropriate.
- the form of the QOL improving or maintaining agent or the like of the present invention is not particularly limited.
- solid forms such as powders, granules, tablets, pills, and troches; semi-solid forms such as jelly, mousse, yogurt, pudding, and cream , Liquid forms such as solution, suspension, emulsion, syrup and the like.
- these forms can be filled into microcapsules, soft capsules, hard capsules and the like to form capsules.
- the QOL improving or maintaining agent of the present invention can be in the form of a foamed preparation.
- the content of the Lactobacillus ONRICb0240 strain in the QOL improving or maintaining agent or the like of the present invention is appropriately set according to the daily dose, dosage form, frequency of administration, purpose of use and the like. Although it is not limited as long as the effect of the present invention is exhibited, for example, the content of the Lactobacillus ONRICb0240 strain in the QOL improving or maintaining agent of the present invention is such that the Lactobacillus ONRICb0240 strain has ,
- the total number of dead bacteria and treated cells) is 10 4 cells / mg or more, preferably 10 5 to 10 12 cells / mg, more preferably 10 6 to 10 11 cells / mg.
- the content of the Lactobacillus ONRICb0240 strain in the QOL improvement or maintenance agent of the present invention is not limited as long as the effect of the present invention is exerted.
- the Lactobacillus ONRICb0240 strain is 10 4 cells or more, preferably 10 6 to 10 12 cells, more preferably 10 7 to 10 12 cells, and still more preferably 10 8 to 10 12 cells, particularly preferably from the viewpoint that a desired effect can be obtained efficiently.
- the total number of bacteria is 10 8 to 10 11 cells, and more preferably the total number of bacteria is 10 9 to 10 10 cells.
- the dose of the QOL improving or maintaining agent of the present invention is appropriately adjusted depending on age, sex, symptoms, etc.
- 10 4 cells or more of Lactobacillus ONRICb0240 strain as a total cell count Preferably 10 6 to 10 12 cells, more preferably 10 7 to 10 12 cells, still more preferably 10 8 to 10 12 cells, and particularly preferably 10 8 to 10 11 cells from the viewpoint of efficiently obtaining a desired effect, More preferably, 10 9 to 10 10 cells are exemplified.
- This can be administered once a day or divided into several times.
- the administration method is not limited as long as the effects of the present invention are exhibited, but oral administration is preferred.
- the application target in the present invention is not limited as long as it is an animal whose purpose is to improve or maintain QOL by improving or maintaining physical health.
- mammals such as humans can be cited as the animals, and besides these, various animals including pets and livestock other than mammals can be cited.
- the present invention may be applied to any age or sex.
- the QOL improvement or maintenance agent of the present invention can be applied to food and drink. That is, the QOL improvement or maintenance agent of the present invention can be used as an additive to food and drink. According to the food / beverage products containing the QOL improving or maintaining agent or the like of the present invention, the effect resulting from the QOL improving or maintaining agent or the like of the present invention, that is, the effect resulting from the Lactobacillus ONRICb0240 strain is obtained. Moreover, the QOL improvement or maintenance agent of this invention can be used as a pharmaceutical as it is. Further, the QOL improving or maintaining agent of the present invention can be used as an additive to pharmaceuticals. According to such a pharmaceutical containing the QOL improving or maintaining agent of the present invention, the effect resulting from the QOL improving or maintaining agent of the present invention, that is, the effect resulting from the Lactobacillus ONRICb0240 strain is obtained.
- the type of the food or drinks or pharmaceuticals is not limited, and the Lactobacillus ONRICb0240 strain can be incorporated into foods and drinks and pharmaceuticals as one component.
- any component such as an edible or pharmaceutically acceptable carrier or additive may be further contained as necessary.
- Edible or pharmaceutically acceptable carriers and additives include aqueous media, excipients, binders, disintegrants, lubricants, thickeners, surfactants, osmotic pressure regulators, moisturizers. Examples thereof include, but are not limited to, pH adjusters, sweeteners, fragrances, and pigments. These components can be appropriately selected by those skilled in the art, and the blending amount of the components is appropriately adjusted within a range that does not impede the effects of the present invention so as to conform to the intended form and taste. do it.
- food and drink or the medicine containing the QOL improving or maintaining agent of the present invention is not limited as long as the effects of the present invention are exhibited.
- food and drinks are not limited, but confectionery (gum, candy, cookies, gummi, rice crackers, biscuits, jelly, mousse, cream caramel, ramune confectionery, edible sheet, edible film, troche, etc.), mouth freshener (Gum, rice cake, gummi, edible film, troche, etc.), beverages (carbonated beverages, soft drinks, milk beverages, alcoholic beverages, fruit juice beverages, teas, nutritional beverages, etc.), powdered beverages (powdered juice, powdered soups, etc.) Dairy products (cheese, yogurt, etc.), bread, noodles, cereals and the like.
- examples of food and drink include food for specified health use, nutritional supplements, food for sick people, and the like.
- examples of food and drink include food for specified health use, nutritional supplements, food for sick people, and the like.
- solid form, semi-solid form, liquid form preparation, capsule, foaming preparation, etc. are mentioned. These production methods can be performed according to methods known in the art.
- Lactobacillus ONRICb0240 strain in foods and beverages or pharmaceuticals containing the QOL improving or maintaining agent of the present invention is appropriately set according to the daily dose, dosage form, number of administrations, purpose of use and the like. Although it is not limited as long as the effect of the present invention is exerted, for example, Lactobacillus ONRICb0240 strain is 10 4 cells / mg or more, preferably 10 5 to 10 12 cells / mg, more preferably 10 6 to 10 11 cells / mg.
- the content of the Lactobacillus ONRICb0240 strain in the food or drink or medicine is not limited as long as the effect of the present invention is exerted, but for example, the total number of Lactobacillus ONRICb0240 strain per food or drink or medicine is 10 4 cells or more, preferably 10 6 to 10 12 cells, more preferably 10 7 to 10 12 cells, still more preferably 10 8 to 10 12 cells, and particularly preferably total bacteria from the viewpoint that a desired effect can be obtained efficiently.
- the number is 10 8 to 10 11 cells, and more preferably the total number of bacteria is 10 9 to 10 10 cells.
- the dose of the food / beverage product or pharmaceutical is appropriately adjusted depending on age, sex, symptom, etc., but as a daily dose for adults, the total number of Lactobacillus ONRICb0240 strains is 10 4 cells or more, preferably 10 6 to 10 12 cells, more preferably 10 7 to 10 12 cells, still more preferably 10 8 to 10 12 cells, particularly preferably 10 8 to 10 11 cells, more preferably 10 from the viewpoint of obtaining a desired effect efficiently.
- 9 to 10 10 cells are exemplified. This can be administered once a day or divided into several times.
- the Lactobacillus ONRICb0240 strain can effectively promote improvement or maintenance of QOL. Therefore, the present invention further provides a method for improving or maintaining QOL using Lactobacillus ONRICb0240 strain.
- the method for improving or maintaining QOL of the present invention can be carried out by administering Lactobacillus ONRICb0240 strain to an animal seeking improvement or maintenance of QOL. That is, the method for improving or maintaining the QOL of the present invention of the present invention comprises an animal that is required to improve or maintain the QOL, the QOL is improved or maintained, or the QOL is improved or maintained. Including a step of ingesting a food or drink or a medicine.
- Lactobacillus ONRICb0240 strain in the method of the present invention the QOL improving or maintaining agent, the dose, the number of times of administration, the administration method, the administration site, etc. of the food or drink or drug containing the QOL improving or maintaining agent are as described above.
- QOL can be improved or maintained.
- the QOL improving or maintaining agent of the present invention has an effect of enhancing physical health.
- the QOL improving or maintaining agent can improve or maintain QOL particularly from the physical aspect, and can therefore be used as a physical health improving or maintaining agent.
- the QOL improving or maintaining agent of the present invention has an action of increasing vitality, an action of reducing or mitigating fatigue, and an action of making it difficult to cause fatigue.
- the QOL improving or maintaining agent can be further used as a vitality improving or maintaining agent, a fatigue recovery agent, a fatigue relaxation agent, particularly a physical fatigue recovery agent, a physical fatigue relaxation agent, an anti-fatigue agent and the like.
- the present invention as described above, it is possible to prevent or reduce the deterioration of physical health caused by lack of rest and sleep, irregular meals, lack of exercise, intense exercise, aging, disturbance of life factors such as stress, etc. .
- the present invention even when it is easy to feel tired from a constitution such as weak constitution, it is possible to prevent or improve the constitution. That is, the present invention can be applied when seeking improvement or maintenance of physical health, and therefore, a healthy person who does not need treatment from a medical point of view, or a person who lives the same life as a healthy person even if he / she goes to the hospital.
- SF-36v2 (registered trademark) is an improved version of SF-36 (registered trademark).
- the details of SF-36v2 (registered trademark) are described in the SF-36v2 TM Japanese version manual issued in October 2009 by the NPO Healthcare Evaluation and Research Organization.
- Example 1 QOL improvement or maintenance agent Two types of QOL improvement or maintenance agents having different contents of Lactobacillus ONRICb0240 strain were produced by the following procedure.
- the cultured Lactobacillus ONRICb0240 strain was recovered by centrifugation, suspended in distilled water, and freeze-dried to obtain a lyophilized bulk powder of Lactobacillus ONRICb0240 strain.
- the number of bacteria in the bulk powder was counted according to the manual using a flow cytometer (EPICS (registered trademark) XL-MCL, manufactured by Beckman Coulter, Inc.).
- Lactobacillus ONRICb0240 strain 4 ⁇ 10 9 cells / tablet was added to the excipient, and tablets were produced using a high-speed rotating small research tablet machine (VIRG 0512SS2AZ, manufactured by Kikusui Seisakusho Co., Ltd.).
- VIRG 0512SS2AZ high-speed rotating small research tablet machine
- another tablet was produced in the same manner except that the Lactobacillus ONRICb0240 strain was changed to 4 ⁇ 10 10 cells / tablet.
- the QOL improvement or maintenance agent thus obtained can be used as a physical health improvement or maintenance agent, vitality improvement or maintenance agent, fatigue recovery agent, fatigue relaxation agent, anti-fatigue agent, and the like.
- Test example 1 Test method Elderly people aged 65 years or older (65 to 84 years old) who obtained a consent form by free will before the start of the test were selected as test subjects. The subjects were appropriately adjusted according to age and gender ratio, and 300 people were randomly assigned to 3 groups, 100 people each.
- the placebo composition As a test substance, the placebo composition, the composition 1 produced in Example 1 (containing 4 ⁇ 10 9 cells of Lactobacillus ONRICb0240 strain), and the composition 2 produced in Example 1 (4 ⁇ Lactobacillus ONRICb0240 strain) 3 patterns of 10 10 cells) were prepared.
- the placebo composition was prepared in the same manner as Compositions 1 and 2 except that Lactobacillus ONRICb0240 strain was not used.
- the group administered with the placebo composition was “(I) group”
- the group administered with the composition containing Lactobacillus ONRICb0240 strain 4 ⁇ 10 9 cells was “(II) group”
- the Lactobacillus ONRICb0240 strain 4 ⁇ 10 10 cells The group to which the composition containing ⁇ was administered was designated as “(III) group”.
- test substance administration of the test substance was one tablet per day for 20 weeks. The same color was used for the taste and color of the test object, and the same packaging container was used. The study was conducted in a double-blind, randomized placebo control and parallel group comparison.
- the obtained answer was processed according to the SF-36v2 (registered trademark) manual. Specifically, processing was performed according to the SF-36v2 TM Japanese version manual issued in October 2009 by the NPO Health Care Evaluation Research Organization. Briefly, according to the manual, the raw score was calculated from the answers obtained in each health concept (subscale) and converted to the subscale score in the range of 0-100 points. In each health concept, a value obtained by subtracting the average value of the lower scale score obtained at the start of the test from the average value of the lower scale score obtained at the end of the test is shown as a ⁇ value and compared between the groups. Comparison between groups was performed using Dunnett's test, and dose dependence was evaluated using Jonckheere trend test (two-sided test).
- Test results Table 1 shows the comparison between the average value of the subscale scores (physical health score) of the subjects and the average value of the national survey sample, which was performed before the start of the test .
- SF-36v2 has eight health concepts (subscales), namely, body function PF, daily role function (body) RP, body pain BP, overall health GH, vitality VT, social function SF, daily role function ( Mental) RE and mental health MH can be evaluated.
- physical health includes physical function PF, daily role function (body) RP, body pain BP, overall health. It is judged that 5 items of GH and vitality VT are involved. Therefore, in this test example, evaluation was mainly performed on these five health concepts.
- the average value of the national survey sample is the SF-36v2 TM Japanese version manual p. 101 is a value described as the national average value of Japanese.
- the subject when comparing the score of the subject with the average value of the national survey sample, the subject showed a remarkably high value in any of the evaluation items.
- the subjects in this study example were 65 years old or older (65 to 84 years old), but the value is based on the average value of national survey samples of 60 years old and older (60 to 69 years old and 70 to 79 years old). It was much better.
- the value is superior to the standard value (overall) of the national survey sample with an average age of 50.5 years, and is particularly highly evaluated in the overall health feeling GH and vitality VT. there were. From this, it was judged that the subjects in this test example were significantly healthier than the national average.
- Table 2 shows the score at the start of the test, the score at the end of the test, and the difference between the score before the start of the test and the end of the test ( ⁇ value: a value obtained by subtracting the score before the start of the test from the score at the end of the test). showed that.
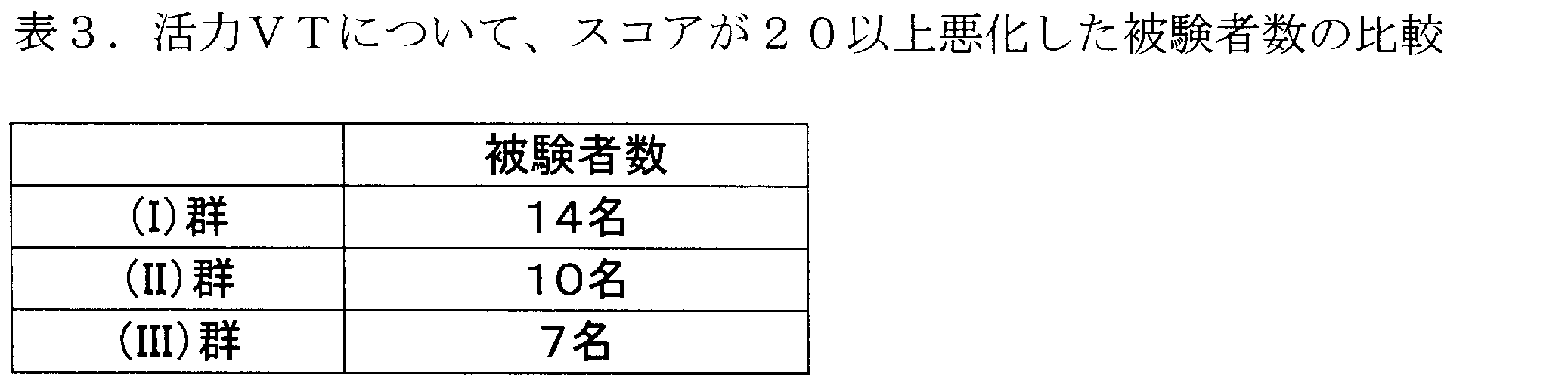
- Table 3 shows the number of subjects whose vitality VT had a score worse by 20 or more ( ⁇ 20) at the end of the test compared to the score before the start of the test.
- the vitality VT score (score category), the percentage of respondents who were “always” and “almost always” full of vitality, and that they felt tired.
- a table summarizing the relationship of the proportion of respondents is given (p.132, Table 11.2). A part of the table is extracted as shown in Table 4 below.
- the average value of the category according to score regarding the vitality VT before the start of the test of the subject was 70. From this, it can be said that the subjects were a group in which 84% felt full of vitality and 2.3% felt tired before the start of the test. In this group, if the score deteriorates by about 20 after the end of the test, the score category will be lowered to 50-59. As is clear from Table 4, in the score categories 50 to 59, 12.2% felt full of vitality, and 17.1% became a group that felt tired. In this way, the score worsening by 20 in this test means that the ratio of vitality has decreased significantly from 84% to 12.2%, and the ratio of feeling fatigued from 2.3%. It will increase significantly to 17.1%. Control of such deterioration is very important.
- Table 5 shows the number of subjects whose daily role function (body) RP deteriorated by 20 or more ( ⁇ 20) at the end of the test compared to the score before the start of the test.
- the daily role function (body) RP is a scale for evaluating the increase / decrease of the normal activity time and the availability of the activity. In this way, since the deterioration of the score was suppressed even in the daily role function (body) RP, the intake of Lactobacillus ONRICb0240 strain decreased the establishment of physical problems that could interfere with work and normal activities It was shown that it can be done.
- Lactobacillus ONRICb0240 strain is useful for improving or maintaining physical health, particularly improving or maintaining vitality, recovering physical fatigue, reducing physical fatigue, anti-fatigue, etc. Became clear.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Mycology (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Microbiology (AREA)
- Engineering & Computer Science (AREA)
- Nutrition Science (AREA)
- Epidemiology (AREA)
- Molecular Biology (AREA)
- Polymers & Plastics (AREA)
- Food Science & Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Diabetes (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
(1)QOL改善又は維持
項1-1.ラクトバチルスONRICb0240(FERM BP-10065)を含有する、QOL改善又は維持剤。
項1-2.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells/mg以上含有する、項1-1に記載のQOL改善又は維持剤。
項1-3.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells以上含有する、項1-1に記載のQOL改善又は維持剤。
項1-4.項1-1~1-3のいずれかに記載のQOL改善又は維持剤を含有する飲食品又は医薬品。
項1-5.QOL改善又は維持が要求される動物に、項1-1~1-3のいずれかに記載のQOL改善又は維持剤を摂取させる工程を含む、当該動物のQOL改善又は維持方法。
項1-6.QOL改善又は維持が要求される動物に、項1-4に記載の飲食品又は医薬品を摂取させる工程を含む、当該動物のQOL改善又は維持方法。
項1-7.QOL改善又は維持剤を製造するための、ラクトバチルスONRICb0240(FERM BP-10065)の使用。
(2)身体的健康改善又は維持
項2-1.ラクトバチルスONRICb0240(FERM BP-10065)を含有する、身体的健康改善又は維持剤。
項2-2.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells/mg以上含有する、項2-1に記載の身体的健康改善又は維持剤。
項2-3.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells以上含有する、項2-1に記載の身体的健康改善又は維持剤。
項2-4.項2-1~2-3のいずれかに記載の身体的健康改善又は維持剤を含有する飲食品又は医薬品。
項2-5.身体的健康改善又は維持が要求される動物に、項2-1~2-3のいずれかに記載の身体的健康改善又は維持剤を摂取させる工程を含む、当該動物の身体的健康改善又は維持方法。
項2-6.身体的健康改善又は維持が要求される動物に、項2-4に記載の飲食品又は医薬品を摂取させる工程を含む、当該動物の身体的健康改善又は維持方法。
項2-7.身体的健康改善又は維持剤を製造するための、ラクトバチルスONRICb0240(FERM BP-10065)の使用。
(3)活力改善又は維持
項3-1.ラクトバチルスONRICb0240(FERM BP-10065)を含有する、活力改善又は維持剤。
項3-2.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells/mg以上含有する、項3-1に記載の活力改善又は維持剤。
項3-3.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells以上含有する、項3-1に記載の活力改善又は維持剤。
項3-4.項3-1~3-3のいずれかに記載の活力改善又は維持剤を含有する飲食品又は医薬品。
項3-5.活力改善又は維持が要求される動物に、項3-1~3-3のいずれかに記載の活力改善又は維持剤を摂取させる工程を含む、当該動物の活力改善又は維持方法。
項3-6.活力改善又は維持が要求される動物に、項3-4に記載の飲食品又は医薬品を摂取させる工程を含む、当該動物の活力改善又は維持方法。
項3-7.活力改善又は維持剤を製造するための、ラクトバチルスONRICb0240(FERM BP-10065)の使用。
(4)疲労回復又は緩和
項4-1.ラクトバチルスONRICb0240(FERM BP-10065)を含有する、疲労回復又は緩和剤。
項4-2.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells/mg以上含有する、項4-1に記載の疲労回復又は緩和剤。
項4-3.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells以上含有する、項4-1に記載の疲労回復又は緩和剤。
項4-4.項4-1~4-3のいずれかに記載の疲労回復又は緩和剤を含有する飲食品又は医薬品。
項4-5.疲労回復又は緩和が要求される動物に、項4-1~4-3のいずれかに記載の疲労回復又は緩和剤を摂取させる工程を含む、当該動物の疲労回復又は緩和方法。
項4-6.疲労回復又は緩和が要求される動物に、項4-4に記載の飲食品又は医薬品を摂取させる工程を含む、当該動物の疲労回復又は緩和方法。
項4-7.疲労回復又は緩和剤を製造するための、ラクトバチルスONRICb0240(FERM BP-10065)の使用。
(5)抗疲労
項5-1.ラクトバチルスONRICb0240(FERM BP-10065)を含有する、抗疲労剤。
項5-2.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells/mg以上含有する、項5-1に記載の抗疲労剤。
項5-3.ラクトバチルスONRICb0240(FERM BP-10065)を、104cells以上含有する、項5-1に記載の抗疲労剤。
項5-4.項5-1~5-3のいずれかに記載の抗疲労剤を含有する飲食品又は医薬品。
項5-5.抗疲労が要求される動物に、項5-1~5-3のいずれかに記載の抗疲労剤を摂取させる工程を含む、当該動物の抗疲労方法。
項5-6.抗疲労が要求される動物に、項5-4に記載の飲食品又は医薬品を摂取させる工程を含む、当該動物の抗疲労方法。
項5-7.抗疲労剤を製造するための、ラクトバチルスONRICb0240(FERM BP-10065)の使用。
本発明のQOL改善又は維持剤は、ラクトバチルスONRICb0240(FERM BP-10065)株を有効成分として含有することを特徴とする。本発明のQOL改善又は維持剤は、前述のように、特に、身体的健康改善又は維持剤、活力改善又は維持剤、疲労回復剤、疲労緩和剤、抗疲労剤等として使用できる。このため、これらも同様に、ラクトバチルスONRICb0240株を有効成分として含有することを特徴とする。
実施例1:QOL改善又は維持剤
ラクトバチルスONRICb0240株の含有量が異なる2種類のQOL改善又は維持剤を以下の手順で製造した。
1.試験方法
試験開始前に自由意志による同意書を取得した65歳以上(65~84歳)の高齢者を試験対象者として選定した。対象者を年齢及び男女比で適宜調整し、300名を3群に100名ずつ無作為に割付した。
試験開始前に実施した、被験者の下位尺度得点の平均値(身体的健康度スコア)と全国調査サンプルの平均値との比較を表1に示す。なお、SF-36v2は8つの健康概念(下位尺度)、すなわち身体機能PF、日常役割機能(身体)RP、体の痛みBP、全体健康感GH、活力VT、社会的機能SF、日常役割機能(精神)RE、心の健康MHについて評価可能であるが、このうち、身体的な健康(身体的健康度)は、身体機能PF、日常役割機能(身体)RP、体の痛みBP、全体健康感GH、活力VTの5項目が関与していると判断されている。このことから、本試験例では、主にこれら5つの健康概念における評価を行った。ここで、全国調査サンプルの平均値とは、NPO健康医療評価研究機構より2009年10月に発行されているSF-36v2TM日本語版マニュアルp.101に日本人の国民平均値として記載された値である。
Claims (4)
- ラクトバチルスONRICb0240(FERM BP-10065)を含有する、QOL(Quality of Life)改善又は維持剤。
- ラクトバチルスONRICb0240(FERM BP-10065)を含有する、身体的健康改善又は維持剤。
- ラクトバチルスONRICb0240(FERM BP-10065)を含有する、活力改善又は維持剤。
- ラクトバチルスONRICb0240(FERM BP-10065)を含有する、抗疲労剤。
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SG2013069984A SG193508A1 (en) | 2011-03-31 | 2012-03-28 | Agent for improving or maintaining qol |
| CN2012800144844A CN103458905A (zh) | 2011-03-31 | 2012-03-28 | Qol改善或持续剂 |
| ES12762904.6T ES2617989T3 (es) | 2011-03-31 | 2012-03-28 | Agente para mejorar o mantener la QOL |
| JP2013507674A JP6061842B2 (ja) | 2011-03-31 | 2012-03-28 | Qol改善又は持続剤 |
| US14/008,325 US9623054B2 (en) | 2011-03-31 | 2012-03-28 | Agent for improving or maintaining QOL |
| AU2012233590A AU2012233590B2 (en) | 2011-03-31 | 2012-03-28 | Agent for improving or maintaining QOL |
| KR1020137028373A KR101878525B1 (ko) | 2011-03-31 | 2012-03-28 | Qol 개선 또는 지속제 |
| EP12762904.6A EP2692349B1 (en) | 2011-03-31 | 2012-03-28 | Agent for improving or maintaining qol |
| CA2831554A CA2831554C (en) | 2011-03-31 | 2012-03-28 | Lactobacillus strain onricb0240 and use of strain in methods and preparations to improve or maintain quality of life |
| US15/488,623 US10369177B2 (en) | 2011-03-31 | 2017-04-17 | Agent for improving or maintaining QOL |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011-080814 | 2011-03-31 | ||
| JP2011080814 | 2011-03-31 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/008,325 A-371-Of-International US9623054B2 (en) | 2011-03-31 | 2012-03-28 | Agent for improving or maintaining QOL |
| US15/488,623 Division US10369177B2 (en) | 2011-03-31 | 2017-04-17 | Agent for improving or maintaining QOL |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012133533A1 true WO2012133533A1 (ja) | 2012-10-04 |
Family
ID=46931242
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/058145 WO2012133533A1 (ja) | 2011-03-31 | 2012-03-28 | Qol改善又は持続剤 |
Country Status (11)
| Country | Link |
|---|---|
| US (2) | US9623054B2 (ja) |
| EP (1) | EP2692349B1 (ja) |
| JP (2) | JP6061842B2 (ja) |
| KR (1) | KR101878525B1 (ja) |
| CN (1) | CN103458905A (ja) |
| AU (1) | AU2012233590B2 (ja) |
| CA (1) | CA2831554C (ja) |
| ES (1) | ES2617989T3 (ja) |
| SG (3) | SG193508A1 (ja) |
| TW (1) | TWI642363B (ja) |
| WO (1) | WO2012133533A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017183595A1 (ja) * | 2016-04-20 | 2017-10-26 | 株式会社 明治 | 疲労感の改善用の発酵乳 |
| WO2020246430A1 (ja) * | 2019-06-03 | 2020-12-10 | アサヒグループホールディングス株式会社 | 歩行能力改善剤 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI583388B (zh) | 2016-08-24 | 2017-05-21 | 國立體育大學 | 胚芽乳酸桿菌用於製備改善運動表現及降低肌肉疲勞之益生菌組合物之用途 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005019438A1 (ja) * | 2003-08-21 | 2005-03-03 | Otsuka Pharmaceutical Co., Ltd. | 粘膜免疫賦活作用を有する乳酸菌 |
| WO2006090729A1 (ja) * | 2005-02-23 | 2006-08-31 | Otsuka Pharmaceutical Co., Ltd. | 茶-発酵飲料および茶飲料 |
| JP2010222329A (ja) * | 2009-03-25 | 2010-10-07 | Otsuka Pharmaceut Co Ltd | 抗鳥インフルエンザウイルス抗体の産生促進剤 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004112809A1 (ja) * | 2003-06-24 | 2004-12-29 | Otsuka Pharmaceutical Co., Ltd. | 血清尿酸低減組成物 |
| US8182849B2 (en) | 2005-02-23 | 2012-05-22 | Otsuka Pharmaceutical Co., Ltd. | Fermented tea beverage and tea beverage |
-
2012
- 2012-03-28 CA CA2831554A patent/CA2831554C/en active Active
- 2012-03-28 WO PCT/JP2012/058145 patent/WO2012133533A1/ja active Application Filing
- 2012-03-28 SG SG2013069984A patent/SG193508A1/en unknown
- 2012-03-28 AU AU2012233590A patent/AU2012233590B2/en active Active
- 2012-03-28 CN CN2012800144844A patent/CN103458905A/zh active Pending
- 2012-03-28 ES ES12762904.6T patent/ES2617989T3/es active Active
- 2012-03-28 SG SG10201602487SA patent/SG10201602487SA/en unknown
- 2012-03-28 KR KR1020137028373A patent/KR101878525B1/ko active IP Right Grant
- 2012-03-28 JP JP2013507674A patent/JP6061842B2/ja active Active
- 2012-03-28 SG SG10201912606QA patent/SG10201912606QA/en unknown
- 2012-03-28 US US14/008,325 patent/US9623054B2/en active Active
- 2012-03-28 EP EP12762904.6A patent/EP2692349B1/en active Active
- 2012-03-29 TW TW101111098A patent/TWI642363B/zh active
-
2016
- 2016-09-07 JP JP2016174646A patent/JP6261688B2/ja active Active
-
2017
- 2017-04-17 US US15/488,623 patent/US10369177B2/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005019438A1 (ja) * | 2003-08-21 | 2005-03-03 | Otsuka Pharmaceutical Co., Ltd. | 粘膜免疫賦活作用を有する乳酸菌 |
| WO2006090729A1 (ja) * | 2005-02-23 | 2006-08-31 | Otsuka Pharmaceutical Co., Ltd. | 茶-発酵飲料および茶飲料 |
| JP2010222329A (ja) * | 2009-03-25 | 2010-10-07 | Otsuka Pharmaceut Co Ltd | 抗鳥インフルエンザウイルス抗体の産生促進剤 |
Non-Patent Citations (7)
| Title |
|---|
| "SF-36v2Tm Japanese version manual", October 2009, INSTITUTE FOR HEALTH OUTCOMES & PROCESS EVALUATION RESEARCH |
| "SF-36v2Tm Japanese version manual", October 2009, INSTITUTE FOR HEALTH OUTCOMES & PROCESS EVALUATION RESEARCH, pages: 101 |
| FRANCOIS BRINGLE ET AL., INTERNATIONAL JOURNAL OF SYSTEMATIC AND EVOLUTIONARY MICROBIOLOGY, vol. 55, 2005, pages 1629 - 1634 |
| FUKUSHIMA, Y.: "Probiotics and Natural Defense Function of the Host", BIOSCIENCE AND MICROFLORA, vol. 26, no. 1, 2007, pages 1 - 10, XP008143493 * |
| MAKINO, S ET AL.: "Reducing the risk of infection in the elderly by dietary intake of yoghurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1", BR. J. NUTR., vol. 104, no. 7, 2010, pages 998 - 1006, XP055095009 * |
| See also references of EP2692349A1 * |
| TOSHITAKA ODAMAKI ET AL.: "Probiotics no Kino Kenkyu I BB536 Haigo Yogurt no Sugi Kafunsho Shojo Kaizen Koka Oyobi sono Sayo Kijo no Kento", SHOKUHIN KAKO, vol. 48, no. 18, 2005, pages 33 - 39, XP008171501 * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017183595A1 (ja) * | 2016-04-20 | 2017-10-26 | 株式会社 明治 | 疲労感の改善用の発酵乳 |
| JPWO2017183595A1 (ja) * | 2016-04-20 | 2019-02-21 | 株式会社明治 | 疲労感の改善用の発酵乳 |
| JP7248426B2 (ja) | 2016-04-20 | 2023-03-29 | 株式会社明治 | 夏バテの自覚症状改善用の発酵乳 |
| WO2020246430A1 (ja) * | 2019-06-03 | 2020-12-10 | アサヒグループホールディングス株式会社 | 歩行能力改善剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101878525B1 (ko) | 2018-07-13 |
| US20170216379A1 (en) | 2017-08-03 |
| US10369177B2 (en) | 2019-08-06 |
| AU2012233590A2 (en) | 2014-05-08 |
| US20140017217A1 (en) | 2014-01-16 |
| KR20140026433A (ko) | 2014-03-05 |
| SG10201912606QA (en) | 2020-02-27 |
| US9623054B2 (en) | 2017-04-18 |
| JP2017019841A (ja) | 2017-01-26 |
| TW201244647A (en) | 2012-11-16 |
| EP2692349B1 (en) | 2016-12-28 |
| JPWO2012133533A1 (ja) | 2014-07-28 |
| AU2012233590A1 (en) | 2013-10-10 |
| SG10201602487SA (en) | 2016-05-30 |
| EP2692349A1 (en) | 2014-02-05 |
| ES2617989T3 (es) | 2017-06-20 |
| TWI642363B (zh) | 2018-12-01 |
| CA2831554A1 (en) | 2012-10-04 |
| JP6061842B2 (ja) | 2017-01-18 |
| SG193508A1 (en) | 2013-10-30 |
| AU2012233590B2 (en) | 2017-06-08 |
| CN103458905A (zh) | 2013-12-18 |
| CA2831554C (en) | 2021-08-03 |
| EP2692349A4 (en) | 2014-11-05 |
| JP6261688B2 (ja) | 2018-01-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5791009B2 (ja) | 乳酸菌およびそれらを用いた飲食物又は化粧品 | |
| KR20060119869A (ko) | 점막 면역 부활 작용을 갖는 유산균 | |
| US11825856B2 (en) | Composition for preventing or improving functional gastrointestinal disorders, and, pharmaceutical composition, food/beverage composition, and method of preventing or improving functional gastrointestinal disorders using the composition for preventing or improving functional gastrointestinal disorders | |
| US11324784B2 (en) | Sleep-promoting composition, and medical composition and food beverage composition using said sleep-promoting composition | |
| CN111212575A (zh) | 肌肉增量用组合物 | |
| JP6261688B2 (ja) | Qol改善又は持続剤 | |
| WO2022196488A1 (ja) | Qolを改善するための組成物 | |
| EP3782632A1 (en) | Anti-stress composition | |
| US20220022516A1 (en) | Composition for Suppressing Norovirus Infection | |
| US20220233613A1 (en) | Walking ability improver | |
| JP2023126304A (ja) | 腎機能障害予防又は改善用組成物、並びに、該腎機能障害予防又は改善用組成物を用いた医薬品組成物及び飲食品組成物 | |
| JP2023106626A (ja) | 乳酸菌、及びセロトニン分泌促進剤 | |
| KR20070030961A (ko) | 항알레르기용 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12762904 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013507674 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2831554 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14008325 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1301005464 Country of ref document: TH |
|
| ENP | Entry into the national phase |
Ref document number: 2012233590 Country of ref document: AU Date of ref document: 20120328 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20137028373 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012762904 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012762904 Country of ref document: EP |