WO2009153170A2 - Procede de preparation d'un polyamideimide, polyamideimide et composition comprenant ce polyamideimide - Google Patents
Procede de preparation d'un polyamideimide, polyamideimide et composition comprenant ce polyamideimide Download PDFInfo
- Publication number
- WO2009153170A2 WO2009153170A2 PCT/EP2009/056915 EP2009056915W WO2009153170A2 WO 2009153170 A2 WO2009153170 A2 WO 2009153170A2 EP 2009056915 W EP2009056915 W EP 2009056915W WO 2009153170 A2 WO2009153170 A2 WO 2009153170A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- polyamideimide
- aromatic
- aliphatic
- acid
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/14—Polyamide-imides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1075—Partially aromatic polyimides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1075—Partially aromatic polyimides
- C08G73/1082—Partially aromatic polyimides wholly aromatic in the tetracarboxylic moiety
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L77/00—Compositions of polyamides obtained by reactions forming a carboxylic amide link in the main chain; Compositions of derivatives of such polymers
- C08L77/06—Polyamides derived from polyamines and polycarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08L79/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
Definitions
- the present invention relates to a process for preparing a semi-aromatic polyamideimide, and a polyamideimide and a composition based on a thermoplastic matrix, comprising a polyamideimide.
- the invention relates more particularly to a process for the preparation of polyamideimide by melt polymerization of at least one organic compound having carboxyl groups, at least one diamine compound and optionally at least one diacid compound.
- Polyamidesimides are polymers of significant industrial and commercial interest. They are used in particular in the field of fire resistant textiles, or injected parts with high thermal resistance. Among them, semi-aromatic polyamidesimides are particularly interesting in terms of properties. Indeed they have intermediate properties between aromatic polyamidesimides on the one hand, and for example aliphatic polyamides on the other hand. Aromatic polyamidesimides are high performance polymers with very good mechanical properties. But they are difficult to synthesize and transformable in the molten route, unlike aliphatic polyamides. Semi-aromatic polyamidesimides are more easily converted than aromatic polyamidesimides (they have a glass transition temperature and a melting temperature lower than the melt forming temperatures), and they have better mechanical and thermomechanical properties than polyamides. aliphatic.
- Processes for the preparation of aromatic or semi-aromatic polyamidesimides are known. These processes are generally processes for the preparation of polyamidesimide in solution in organic solvents.
- organic solvents has major disadvantages.
- the recovery of the polymer after synthesis requires steps such as precipitation of the polymer in a non-solvent, washing and drying of the polymer.
- some solvents are toxic and therefore dangerous for man and the environment.
- the invention therefore provides a process for the preparation of semi-aromatic polyamideimides, which does not have these disadvantages.
- the invention provides, in a first object, a process for the preparation of semi-aromatic polyamideimide by melt polymerization of at least the following monomers:
- the molar proportion of compound c) relative to the sum of the compounds a) and c) is greater than or equal to 0.5% , advantageously greater than or equal to 25%, preferably greater than or equal to 50%.
- the invention provides a polyamideimide comprising the following recurring units:
- R and R ' are aliphatic, cycloaliphatic or arylaliphatic hydrocarbon radicals, advantageously aliphatic or cycloaliphatic, preferably comprising between 2 and 18 carbon atoms
- R " is a hydrocarbon radical preferably comprising between 2 and 18 carbon atoms
- Y is a trivalent aromatic hydrocarbon radical
- Y ' is a tetravalent aromatic hydrocarbon radical
- polyamideimide comprising the following recurring units:
- Y is a trivalent hydrocarbon radical, aromatic or aliphatic, preferably aromatic
- Y ' is a tetravalent, aromatic or aliphatic hydrocarbon radical, preferably aromatic
- the invention provides a thermoplastic polymer composition comprising the semi-aromatic polyamideimide of the invention described above or obtained by the method of the invention described above.
- the invention finally relates, in a fourth object, the articles obtained by shaping the composition of the invention.
- the process for the preparation of polyamideimide of the invention uses as monomer at least one compound a) organic, preferably aromatic.
- the compound a) has at least three functional groups selected from carboxyl groups and amino groups.
- the compound a) comprises three or four carboxyl groups, preferably three or four carboxylic acid functions.
- the compound a) may for example comprise at least one pair of carboxyl groups ortho position relative to each other.
- the compound a) is of formula (I) below:
- Z may be a trivalent radical of benzene, naphthalene, biphenyl, diphenyl ether, diphenyl sulfide, diphenyl sulfone, ditolyl ether, and the like.
- Advantageously Z has between 6 and 18 carbon atoms.
- This particular embodiment of the process of the invention uses carboxylic acids, which are generally less toxic than the equivalent carboxylic anhydrides.
- the compound a) is preferably chosen from trimellitic acid, pyromellitic acid, their anhydrides, their esters or their amides.
- the compound a) does not include an imide function.
- the compound a) may also be a compound having an amino group and two carboxyl groups.
- examples of such compounds include aspartic acid, 3-aminophthalic acid or 4-aminophthalic acid.
- mixtures of different compounds a) can be implemented.
- the polyamideimide preparation process of the invention uses as monomer at least one compound b) diamine.
- the diamines useful in the present invention advantageously have the formula H 2 NR-NH 2 (II) in which R is a divalent hydrocarbon radical, in particular an aliphatic, aromatic or arylaliphatic diradical or a substituted derivative of these diradicals.
- R is a divalent hydrocarbon radical, in particular an aliphatic, aromatic or arylaliphatic diradical or a substituted derivative of these diradicals.
- the radical R advantageously comprises between 2 and 18 carbon atoms.
- arylaliphatic diamine means a diamine at least one of the amine functions is not attached to a carbon atom forming part of an aromatic ring.
- Suitable aliphatic diamines include straight-chain aliphatic diamines, such as 1,10-diaminodecane, branched-chain aliphatic diamines, such as 2-methyl-1,6-diaminohexane, and cycloaliphatic diamines, such as than di (aminomethyl) cyclohexanediamines.
- the aliphatic chain may contain hetero atoms such as sulfur or oxygen, as represented by 3,3'-ethylenedioxybis (propylamine), and may also carry substituents, such as carbon atoms. halogen, which do not react under the polymerization conditions.
- Suitable aromatic diamines in the present invention include diamines wherein R in the general formula is phenylene group, a condensed aromatic group, such as naphthylene group, or two (or more) linked aromatic rings, as shown by bisphenylene, bisphenylenemethane, bisphenylenepropane, bisphenylenesulfone, bisphenylene ether and the like.
- any of the aromatic groups may carry one or more substituents on the ring, such as lower alkyl groups or halogen atoms, which do not react under the polymerization conditions.
- the diamine preferably contains 2 to 18 carbon atoms, more preferably 4 to 12 carbon atoms.
- diamines include diamines of the homologous series H 2 N (CH 2 ) InNH 2 in which m is an integer of 2 to 12, preferably 4 to 8, and diamines of the general formula H 2 N ( CH 2 ) p Z (CH 2 ) qNH 2 wherein Z is phenylene and p and q are independently 1, 2 or 3.
- the compound b) is an aliphatic diamine.
- the diamines may for example be chosen from hexamethylenediamine; butane diamine; 2-methyl pentamethylene diamine; 2-methyl hexamethylenediamine; 3-methyl hexamethylene diamine; 2,5-dimethyl hexamethylenediamine; 2,2-dimethylpentamethylene diamine; nonane diamine; 5-methylnonanediamine; dodecamethylene diamine; 2,2,4- and 2,4,4-trimethyl hexamethylenediamine; 2,2,7,7-tetramethyl octamethylene diamine; meta-xylylenediamine; paraxylylene diamine; isophorone diamine; diaminodicyclohexyl methane and C2-C16 aliphatic diamines which may be substituted by one or more alkyl groups.
- the preferred diamine is hexamethylenediamine. Mixtures of diamines can also be used in the present invention to produce polymers having recurring units wherein the group represented by R in the
- At least 45 mol%, preferably at least 50 mol% of the diamine compound is an aliphatic, cycloaliphatic or arylaliphatic diamine.
- the polyamideimide preparation process of the invention also optionally performs as monomer at least one compound c) diacid.
- the compound c) has the following formula (III):
- R ' is a divalent aliphatic, cycloaliphatic, arylaliphatic or aromatic hydrocarbon radical.
- the radical R ' preferably has between 2 and 18 carbon atoms.
- arylaliphatic diacid means a diacid which at least one of the acid functions is not attached to a carbon atom forming part of an aromatic ring.
- the compound c) is an aliphatic diacid.
- the aliphatic acid may for example be chosen from oxalic, maleic, succinic, pimelic and azelaic acid. It may also include unsaturations, this is the case, for example, of maleic or fumaric acid.
- the dicarboxylic acids may also be chosen from glutaric acid, adipic acid, suberic acid, sebacic acid, dodecanedioic acid; 1,2-or 1,3-cyclohexane dicarboxylic acid; 1,2-or 1,3-phenylene diacetic acid; 1, 2-or 1,3-cyclohexane diacetic acid; isophthalic acid; terephthalic acid; 4,4'-benzophenone acid dicarboxylic acid; 2,5-naphthalene dicarboxylic acid; and pt-butyl isophthalic acid.
- the preferred dicarboxylic acid is adipic acid.
- mixtures of different compounds c) can be implemented.
- the process of the invention does not comprise lactam monomers or aliphatic amino acids, preferably no lactams or amino acids, such as caprolactam, 6-aminohexanoic acid, 5-aminopentanoic acid, the acid. 7-aminoheptanoic acid, aminoundecanoic acid, dodecanolactam.
- the method of the invention does not comprise diol monomers. Indeed the presence of such monomers may for example result in low viscosity of the polymers obtained, which is not desirable.
- the process for preparing the polyamideimide of the invention comprises a melt polymerization of the monomers a), b) and optionally c).
- melt polymerization is meant that the polymerization is carried out in the liquid state, and that the polymerization medium contains no solvent other than water, optionally.
- the polymerization is carried out in a continuous liquid phase.
- the polymerization medium may for example be an aqueous solution comprising the monomers, or a liquid comprising the monomers.
- the polymerization medium comprises water as a solvent. This facilitates the agitation of the medium, and therefore its homogeneity.
- the polymerization medium may also include additives such as chain limiters.
- the polyamideimide of the invention is generally obtained by polycondensation between the compound a), the compound b) and optionally the compound c) to form polyamideimide chains, with formation of the elimination product, in particular water, some of which can vaporize.
- the polyamideimide of the invention is generally obtained by heating at high temperature and pressure, for example an aqueous solution comprising the monomers, or a liquid comprising the monomers, to evaporate the elimination product, in particularly water (initially present in the polymerization medium and / or formed during the polycondensation) while avoiding any solid phase formation to prevent caking.
- the polycondensation reaction is generally carried out at a pressure of approximately 0.5-2.5 MPa (0.5-3.5 MPa) at a temperature of approximately 215-300 ° C. (180-320 ° C.). VS).
- the polycondensation is generally continued in the melt phase at atmospheric pressure or reduced so as to reach the desired degree of advancement.
- the polycondensation product is a polymer or molten prepolymer. It may comprise a vapor phase consisting essentially of vapor of the elimination product, in particular water, which may have been formed and / or vaporized.
- This product can be subjected to vapor phase separation and finishing steps to achieve the desired degree of polycondensation.
- the separation of the vapor phase may for example be carried out in a cyclone device. Such devices are known.
- the finish consists in maintaining the polycondensation product in the molten state, under a pressure close to atmospheric pressure or under reduced pressure, for a time sufficient to reach the desired degree of advancement. Such an operation is known to those skilled in the art.
- the temperature of the finishing step is advantageously greater than or equal to 200 ° C. and in all cases greater than the solidification temperature of the polymer.
- the residence time in the finishing device is preferably greater than or equal to 5 minutes.
- the polycondensation product may also undergo a post-condensation phase in solid phase. This step is known to the man of and increases the degree of polycondensation to a desired value.
- the process of the invention is similar in its conditions to the conventional polyamide preparation process of the type of those obtained from dicarboxylic acids and diamines, in particular the process for producing polyamide 66 from acid. adipic and hexamethylene diamine.
- This method of manufacturing polyamide 66 is known to those skilled in the art.
- the process for producing polyamide of the type of those obtained from dicarboxylic acids and diamines generally uses as raw material, a salt obtained by a mixture in stoichiometric quantity, generally in a solvent such as water, a diacid with a diamine.
- adipic acid is mixed with hexamethylene diamine generally in water to obtain hexamethylene diammonium adipate better known as nylon salt or " Salt N ".
- this compound c) and the compound b) diamine can be introduced, at least in part, in the form of a salt of compound c ) and compound b).
- these compounds can be introduced at least partly in the form of N salt. This makes it possible to have a stoichiometric equilibrium.
- the process of the invention generally leads to a random polymer.
- the semi-aromatic polyamideimide obtained by the process of the invention comprises the recurring units indicated above.
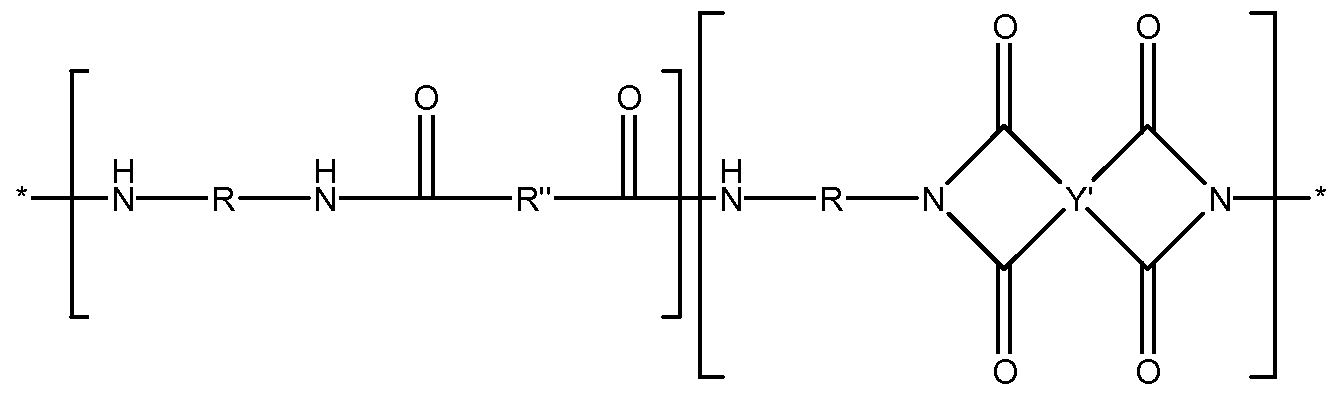
- the polyamideimide obtained may also comprise the following bisimide repeating units:
- the process of the invention is a simple process and easy to implement, which does not implement organic solvents that can be toxic.
- the polyamideimide obtained according to the process of the invention can be used directly, without additional step for example to recover the polymer, as is the case for example in solution preparation processes.
- the polyamideimide obtained at the end of the finishing step can be cooled and put into the form of granules.
- the polyamideimide obtained by the process of the invention in molten form can be directly shaped or extruded and granulated, for subsequent shaping after melting.
- the polyamideimide can be used for a large number of applications, in particular for the manufacture of yarns, fibers or filaments, films, or for shaping articles by injection molding, extrusion, extrusion blow molding. It can especially be used in technical plastic compositions.
- the invention relates in a second object, a polyamideimide comprising the following recurring units:
- R and R ' are aliphatic, cycloaliphatic or arylaliphatic hydrocarbon radicals, advantageously aliphatic or cycloaliphatic, preferably comprising between 2 and 18 carbon atoms
- R " is a hydrocarbon radical preferably comprising between 2 and 18 carbon atoms
- Y is a trivalent aromatic hydrocarbon radical
- Y ' is a tetravalent aromatic hydrocarbon radical
- R, R 'and R " are hydrocarbon radicals preferably comprising between 2 and 18 carbon atoms
- R '" is an aromatic hydrocarbon radical, preferably comprising between 2 and 18 carbon atoms
- Y is a trivalent hydrocarbon radical, aromatic or aliphatic, preferably aromatic
- Y ' is a tetravalent, aromatic or aliphatic hydrocarbon radical, preferably aromatic
- such a polyamideimide does not comprise a unit derived from a diol monomer.
- the polyamideimide advantageously comprises at least 60 mol%, preferably at least 80 mol% of recurring units A.
- the polyamideimides of the invention have the advantage of being easily converted into the molten route, such as aliphatic polyamides, for example, which facilitates their shaping.
- aliphatic polyamides for example, which facilitates their shaping.
- thermomechanical properties in particular the HDT property "Heat Distortion Temperature”
- they show better water absorption properties compared to aliphatic polyamides, and a reduced thermal expansion compared with aliphatic polyamides.
- the polyamideimide of the invention may be used alone or as part of a composition. It can particularly be used as an additive in thermoplastic polymer compositions comprising a thermoplastic matrix. He is involved in the composition as a reinforcing agent.
- the thermoplastic matrix is a thermoplastic polymer.
- polymers which may be suitable, mention may be made of: polylactones such as poly (pivalolactone), poly (caprolactone) and polymers of the same family; polyurethanes obtained by reaction between diisocyanates such as 1,5-naphthalene diisocyanate; p-phenylene diisocyanate, m-phenylene diisocyanate, 2,4-toluene diisocyanate, 4,4'-diphenylmethane diisocyanate, 3,3'-dimethyl-4,4'-diphenylmethane diisocyanate, 3, 3-dimethyl-4,4'-biphenyl diisocyanate, 4,4'-diphenylisopropylidene diisocyanate, 3,3'-dimethyl-4,4'-diphenyl diisocyanate, 3,3'-dimethyl-4,4 ' diphenylmethane diisocyanate, 3,3'-dimethoxy-4
- the thermoplastic matrix is a polymer comprising star or H macromolecular chains, and optionally linear macromolecular chains.
- the polymers comprising such star or H macromolecular chains are for example described in the documents FR 2 743 077, FR 2 779 730, US 5 959 069, EP 0 632 703, EP 0 682 057 and EP 0 832 149.
- thermoplastic matrix of the invention is a random tree type polymer, preferably a copolyamide having a random tree structure.
- a copolyamide having a random tree structure are described in particular in WO 99/03909.
- thermoplastic matrix of the invention may also be a composition comprising a linear thermoplastic polymer and a thermoplastic star, H and / or tree polymer as described above.
- compositions of the invention may also comprise a hyperbranched copolyamide of the type of those described in WO 00/68298.
- compositions of the invention may also comprise any combination of thermoplastic star polymer, H, tree, hyperbranched copolyamide described above.
- thermostable polymers these polymers are preferably infusible or have a softening point greater than 180 ° C., preferably> 200 0 C, or higher.
- thermostable polymers may for example be chosen from aromatic polyamides, polyamide imides such as polytrimellamide imides, or polyimides such as the polyimides obtained according to document EP 0119185, known commercially under the trademark P84.
- aromatic polyamides may be as described in patent EP 0360707. They may be obtained according to the process described in patent EP 0360707.
- polymeric matrix there may also be mentioned viscose, cellulose, cellulose acetate and so on.
- the polymer matrix of the invention may also be of the type of polymers used in adhesives, such as copolymers of plastisol vinyl acetates, acrylic latices, urethane latices, PVC plastisol, etc.
- semicrystalline polyamides such as polyamide 6, polyamide 6.6, polyamide 11, polyamide 12, polyamide 4, polyamides 4-6, 6-10, are particularly preferred.
- 6-12, 6-36, 12-12, semi-aromatic polyamides obtained from terephthalic and / or isophthalic acid such as the polyamide marketed under the trade name AMODEL; polyesters such as PET, PBT, PTT; polyolefins such as polypropylene, polyethylene; aromatic polyamides, polyamide imides or polyimides; latex such as acrylic latex and urethane; PVC, viscose, cellulose, cellulose acetate; their copolymers and alloys.
- compositions may contain any other additives that may be used, for example reinforcing fillers, flame retardants, UV stabilizers, heat, mattifying agents such as titanium dioxide.
- compositions according to the invention are preferably obtained by melt blending of the thermoplastic polymer and the polyamideimide.
- the mixture can for example be made using an extrusion device, for example a single screw or double screw mixer.
- the weight ratio of polyamideimide in the composition is advantageously between 1 and 99%, preferably between 5 and 30%.
- compositions according to the invention can be used as raw material in the field of engineering plastics, for example for the production of injection molded articles or by injection / blow molding, extruded by conventional extrusion or by extrusion blow molding, or movies.
- compositions according to the invention can also be put into the form of son, fibers, filaments by melt-spinning.
- Absolute molar mass determined by gel permeation chromatography (GPC) in dichloromethane (+ trifluoroacetic anhydride), then by triple detection by refractometry R1, UV absorption and viscosimethe.
- Imide cyclization rate determined by 1 H NMR at 300 K in deuterated formic acid, using a 300 MHz Bruker DRX apparatus
- T f Melting temperature and associated enthalpy ( ⁇ Hf), glass transition temperature (T 9 ), cooling crystallization temperature (T c ): determined by differential scanning calorimetry (DSC) using a Perkin Elmer Pyris 1 device, at a rate of 10 ° C / min.
- Acid and amino terminal group contents assayed by potentiometry
- Viscosity index (IV) measured according to ISO EN 307 Example 1 Preparation of a Polyamide Imide PAI 66 / 6TMA 90/10
- the polyamideimide is manufactured according to a standard polyamide 66 polymerization process.
- the polymer obtained is cast in the form of rod, cooled and granulated by cutting rushes.
- Tg 63 ° C.
- Tc 204.2 ° C.
- mp 247.2 ° C.
- Examples 2 to 7 are prepared, according to a standard method of polymerization of polyamide 66 type, as in Example 1, polyamideimides of PAI 66 / 6TMA type of molar composition 80/20 (Example 2), 67/33 ( Example 3), 60/40 (Example 4), 50/50 (Example 5), 30/70 (Example 6),
- compositions (all compositions comprise 2 g of antifoaming agent) and characterizations of these polymers are mentioned in Table 1 below.
- Table 1 The compositions (all compositions comprise 2 g of antifoaming agent) and characterizations of these polymers are mentioned in Table 1 below.
- trimellitic anhydride is used in place of trimellitic acid.
- the polyamideimide is manufactured according to a standard polyamide 66 polymerization process, as in Example 1.
- the polymer obtained is cast in the form of rod, cooled and granulated by cutting rushes.
- the analyzes do not show any major differences between the polymers synthesized with trimellitic anhydride or with trimellitic acid.
- the polyamideimide is manufactured according to a standard polyamide 66 polymerization process, as in Example 1.
- the polymer obtained is cast in the form of rod, cooled and granulated by cutting rushes.
- the polymer is amorphous.
- the polyamideimide is manufactured according to a standard polyamide 66 polymerization process, as in Example 1.
- the polymer obtained is cast in the form of rod, cooled and granulated by cutting rushes.
- Tc 220.2 ° C.
- Tm 255.1 ° C.
- N salt (1: 1 salt of hexamethylene diamine and adipic acid)
- 6T salt (1: 1 salt)
- hexamethylene diamine and terephthalic acid 5.84 g of trimellitic acid
- TMLA hexamethylenediamine
- HMD hexamethylenediamine
- the polyamideimide is manufactured according to a standard polyamide 66 polymerization process.
- the polymer obtained is cast in the form of rod, cooled and granulated by cutting rushes.
- Examples 12 to 17 Preparation of compositions comprising a polyamide and a polyamideimide according to the invention.
- compositions comprising a polyamide PA 66 and a polyamideimide PAI 6TMA were prepared in a micro-extruder ("micro-compounder")
- the polyamideimide used is polyamidimide of Example 9.
- the polyamide PA66 and the polyamideimide PAI 6TMA are mixed in the micro-extruder at a temperature of 275 ° C., and injected into a mold at 70 ° C. (injection module) in the form of bars of dimensions 62 ⁇ 12 ⁇ 4 mm 3 .
- N.M. No Measurable: existence of a Ta but it can not be measured.
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09765750A EP2294113A2 (fr) | 2008-06-12 | 2009-06-05 | Procede de preparation d'un polyamideimide, polyamideimide et composition comprenant ce polyamideimide |
| CN2009801282568A CN102099398B (zh) | 2008-06-12 | 2009-06-05 | 聚酰胺酰亚胺的制备方法、聚酰胺酰亚胺以及包含该聚酰胺酰亚胺的组合物 |
| KR1020107027871A KR101308319B1 (ko) | 2008-06-12 | 2009-06-05 | 폴리아미드 이미드의 제조 방법, 폴리아미드 이미드 및 폴리아미드 이미드를 포함하는 조성물 |
| BRPI0909912A BRPI0909912A2 (pt) | 2008-06-12 | 2009-06-05 | "processo de preparação de poliamidaimida semiaromática, poliamidaimida, composição polimérica termoplástica e artigo" |
| US12/997,208 US20110160407A1 (en) | 2008-06-12 | 2009-06-05 | Novel polyamideimides and preparation and compositions comprised thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0803263A FR2932486B1 (fr) | 2008-06-12 | 2008-06-12 | Procede de preparation d'un polyamideimide, polyamideimide et composition comprenant ce polyamideimide. |
| FR08/03263 | 2008-06-12 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2009153170A2 true WO2009153170A2 (fr) | 2009-12-23 |
| WO2009153170A3 WO2009153170A3 (fr) | 2010-02-18 |
Family
ID=40043051
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/056915 WO2009153170A2 (fr) | 2008-06-12 | 2009-06-05 | Procede de preparation d'un polyamideimide, polyamideimide et composition comprenant ce polyamideimide |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20110160407A1 (fr) |
| EP (1) | EP2294113A2 (fr) |
| KR (1) | KR101308319B1 (fr) |
| CN (1) | CN102099398B (fr) |
| BR (1) | BRPI0909912A2 (fr) |
| FR (1) | FR2932486B1 (fr) |
| WO (1) | WO2009153170A2 (fr) |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102532545B (zh) * | 2011-12-29 | 2013-11-06 | 东莞市信诺橡塑工业有限公司 | 一种长碳链半芳香族聚酰胺酰亚胺及其合成方法 |
| CN102585223B (zh) * | 2011-12-29 | 2013-08-28 | 东莞市信诺橡塑工业有限公司 | 一种长碳链半芳香族聚酰胺酰亚胺及其合成方法 |
| CN102702526B (zh) * | 2012-06-21 | 2014-04-02 | 东莞市信诺橡塑工业有限公司 | 松香基半脂环族聚酰胺酰亚胺共聚物及其合成方法 |
| EP2708569B1 (fr) * | 2012-09-12 | 2018-05-23 | Ems-Patent Ag | Polyamide-imides transparentes |
| JP6913346B2 (ja) * | 2016-04-05 | 2021-08-04 | ユニチカ株式会社 | ポリアミドイミド前駆体溶液 |
| KR20200053302A (ko) * | 2018-11-08 | 2020-05-18 | 삼성전자주식회사 | 폴리(아미드-이미드) 코폴리머, 폴리(아미드-이미드) 코폴리머 제조용 조성물, 폴리(아미드-이미드) 코폴리머를 포함하는 성형품 및 표시 장치 |
| CN110437445B (zh) * | 2019-08-21 | 2021-11-16 | 中仑塑业(福建)有限公司 | 一种聚酰胺树脂及其制备方法和用途 |
| JP2022546854A (ja) | 2019-09-09 | 2022-11-09 | ソルベイ スペシャルティ ポリマーズ ユーエスエー, エルエルシー | ポリアミド-イミドポリマー及びその製造方法 |
| WO2021048074A1 (fr) | 2019-09-09 | 2021-03-18 | Solvay Specialty Polymers Usa, Llc | Polymère de polyamide-imide et son procédé de fabrication |
| CN110734545A (zh) * | 2019-10-28 | 2020-01-31 | 中仑塑业(福建)有限公司 | 一种聚酰亚胺树脂及其制备方法和用途 |
| CN112321829B (zh) * | 2020-10-29 | 2023-01-06 | 山东君昊高性能聚合物有限公司 | 水相合成聚酰胺-酰亚胺树脂的方法 |
| CN114716678A (zh) * | 2020-12-22 | 2022-07-08 | 成都肆零壹科技有限公司 | 一种酰亚胺结构聚合物的制备方法 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5550208A (en) | 1983-02-16 | 1996-08-27 | Amoco Corporation | Polycondensation process |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BE756218A (fr) * | 1969-09-16 | 1971-03-01 | Bayer Ag | Procede de production d'acides imidocarboxyliques |
| US3961009A (en) * | 1970-04-22 | 1976-06-01 | Toray Industries, Inc. | Process for the production of a shaped article of a heat resistant polymer |
| US4070524A (en) * | 1976-02-23 | 1978-01-24 | Standard Oil Company (Indiana) | Self-bonding varnish for magnetic wire comprising polyalkylenetrimellitate imide polyalkylenetrimellitate ester |
| JPS58162637A (ja) * | 1982-03-19 | 1983-09-27 | Nitto Electric Ind Co Ltd | ポリアミドイミド樹脂の製造方法 |
| US5395870A (en) * | 1988-12-28 | 1995-03-07 | Asahi Kasei Kogyo Kabushiki Kaisha | Vinyl resin composition |
| DE4025346A1 (de) * | 1990-08-10 | 1992-02-13 | Bayer Ag | Transparente, thermoplastische polyetheresteramidimid-elastomere sowie ihre verwendung zur herstellung von medizinischen gebrauchsartikeln |
-
2008
- 2008-06-12 FR FR0803263A patent/FR2932486B1/fr not_active Expired - Fee Related
-
2009
- 2009-06-05 CN CN2009801282568A patent/CN102099398B/zh not_active Expired - Fee Related
- 2009-06-05 BR BRPI0909912A patent/BRPI0909912A2/pt not_active IP Right Cessation
- 2009-06-05 WO PCT/EP2009/056915 patent/WO2009153170A2/fr active Application Filing
- 2009-06-05 US US12/997,208 patent/US20110160407A1/en not_active Abandoned
- 2009-06-05 KR KR1020107027871A patent/KR101308319B1/ko not_active IP Right Cessation
- 2009-06-05 EP EP09765750A patent/EP2294113A2/fr not_active Withdrawn
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5550208A (en) | 1983-02-16 | 1996-08-27 | Amoco Corporation | Polycondensation process |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101308319B1 (ko) | 2013-09-17 |
| CN102099398B (zh) | 2013-11-20 |
| FR2932486B1 (fr) | 2012-12-07 |
| BRPI0909912A2 (pt) | 2015-10-20 |
| CN102099398A (zh) | 2011-06-15 |
| WO2009153170A3 (fr) | 2010-02-18 |
| FR2932486A1 (fr) | 2009-12-18 |
| KR20110013472A (ko) | 2011-02-09 |
| US20110160407A1 (en) | 2011-06-30 |
| EP2294113A2 (fr) | 2011-03-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2009153170A2 (fr) | Procede de preparation d'un polyamideimide, polyamideimide et composition comprenant ce polyamideimide | |
| EP0550314B1 (fr) | Polyamides et objets obtenus à partir de ceux-ci | |
| EP2729516B1 (fr) | Nouveau polyamide, procede de preparation et utilisations | |
| US9376535B2 (en) | Branched polyamide with different blocks | |
| EP2326681B1 (fr) | Polyamide modifie, procede de preparation, article obtenu a partir de ce polyamide | |
| US9670319B2 (en) | Polyamide, preparation process therefor and uses thereof | |
| CH615204A5 (fr) | ||
| KR100683995B1 (ko) | 테트라메틸렌 테레프탈아미드 및 헥사메틸렌 테레프탈아미드계 코폴리아미드 | |
| KR20160013995A (ko) | Me-bht를 포함하는 폴리아미드, 이와 같은 폴리아미드를 포함하는 조성물, 이와 같은 폴리아미드 또는 이와 같은 조성물을 포함하는 성형품 | |
| CA2700334A1 (fr) | Polyamide de haute fluidite | |
| FR2952062A1 (fr) | Procede de preparation de poudre recyclable a base de polyamide | |
| US9321888B2 (en) | Branched polyamide | |
| FR2632958A1 (fr) | Polyamides amorphes transparents ayant une temperature de transition vitreuse elevee | |
| FR2632959A1 (fr) | Polyamides semi-aromatiques cristallises ayant une temperature de fusion inferieure a 290 oc | |
| WO2019121824A1 (fr) | Copolyamide semi-aromatique contenant de la pipéridine | |
| EP2448998B1 (fr) | Polyamide modifie, procede de preparation, article obtenu a partir de ce polyamide | |
| JPH02115226A (ja) | 固相後縮合による分枝鎖状(コ)ポリアミド類の製造方法 | |
| US9534083B2 (en) | Production of polyamides by polycondensation | |
| JPH02115227A (ja) | 高分子量(コ)ポリアミド類およびそれらの製造方法 | |
| FR3008984A1 (fr) | Articles obtenus a partir d'une composition polymerique, procede de preparation et utilisations | |
| JPH0218421A (ja) | ポリアミドイミドの製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980128256.8 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09765750 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009765750 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20107027871 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: PI0909912 Country of ref document: BR Kind code of ref document: A2 Effective date: 20101210 |