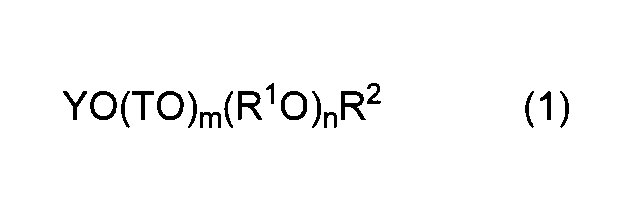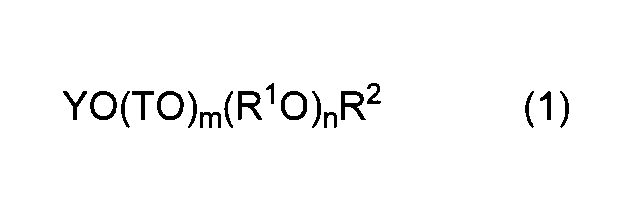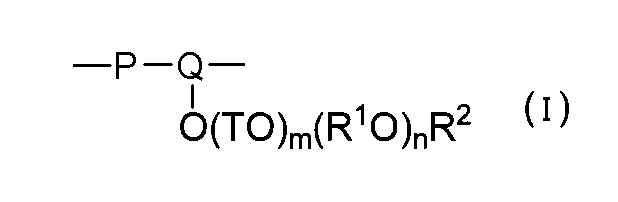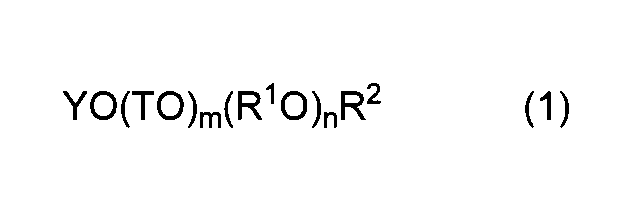WO2009139328A1 - ポリカルボン酸系共重合体の製造方法およびセメント混和剤用共重合体組成物 - Google Patents
ポリカルボン酸系共重合体の製造方法およびセメント混和剤用共重合体組成物 Download PDFInfo
- Publication number
- WO2009139328A1 WO2009139328A1 PCT/JP2009/058672 JP2009058672W WO2009139328A1 WO 2009139328 A1 WO2009139328 A1 WO 2009139328A1 JP 2009058672 W JP2009058672 W JP 2009058672W WO 2009139328 A1 WO2009139328 A1 WO 2009139328A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- copolymer
- monomer
- acid
- group
- general formula
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B24/00—Use of organic materials as active ingredients for mortars, concrete or artificial stone, e.g. plasticisers
- C04B24/16—Sulfur-containing compounds
- C04B24/161—Macromolecular compounds comprising sulfonate or sulfate groups
- C04B24/163—Macromolecular compounds comprising sulfonate or sulfate groups obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C04B24/165—Macromolecular compounds comprising sulfonate or sulfate groups obtained by reactions only involving carbon-to-carbon unsaturated bonds containing polyether side chains
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B24/00—Use of organic materials as active ingredients for mortars, concrete or artificial stone, e.g. plasticisers
- C04B24/24—Macromolecular compounds
- C04B24/26—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C04B24/2641—Polyacrylates; Polymethacrylates
- C04B24/2647—Polyacrylates; Polymethacrylates containing polyether side chains
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F216/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical
- C08F216/12—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical by an ether radical
- C08F216/14—Monomers containing only one unsaturated aliphatic radical
- C08F216/1416—Monomers containing oxygen in addition to the ether oxygen, e.g. allyl glycidyl ether
- C08F216/1425—Monomers containing side chains of polyether groups
- C08F216/1433—Monomers containing side chains of polyethylene oxide groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F283/00—Macromolecular compounds obtained by polymerising monomers on to polymers provided for in subclass C08G
- C08F283/06—Macromolecular compounds obtained by polymerising monomers on to polymers provided for in subclass C08G on to polyethers, polyoxymethylenes or polyacetals
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/40—Redox systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D4/00—Coating compositions, e.g. paints, varnishes or lacquers, based on organic non-macromolecular compounds having at least one polymerisable carbon-to-carbon unsaturated bond ; Coating compositions, based on monomers of macromolecular compounds of groups C09D183/00 - C09D183/16
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2103/00—Function or property of ingredients for mortars, concrete or artificial stone
- C04B2103/40—Surface-active agents, dispersants
- C04B2103/408—Dispersants
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2111/00—Mortars, concrete or artificial stone or mixtures to prepare them, characterised by specific function, property or use
- C04B2111/60—Flooring materials
- C04B2111/62—Self-levelling compositions
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/04—Acids; Metal salts or ammonium salts thereof
- C08F220/06—Acrylic acid; Methacrylic acid; Metal salts or ammonium salts thereof
Definitions
- the present invention relates to a method for producing a polycarboxylic acid copolymer. Specifically, the present invention relates to a method for producing a polycarboxylic acid copolymer suitable for a cement admixture. The present invention also relates to a copolymer composition for a cement admixture. In detail, it is related with the copolymer composition suitable as a cement admixture.
- cement admixtures are widely used in cement compositions such as cement paste, mortar and concrete.
- the fluidity of the cement composition can be increased, and the cement composition can be reduced in water. This water reduction can improve the strength and durability of the cured product.
- a cement admixture mainly composed of a polycarboxylic acid copolymer has been proposed as a cement admixture.
- a cement admixture mainly composed of a polycarboxylic acid copolymer can exhibit high water reduction performance.
- the unsaturated polyalkylene glycol ether monomer has a problem of low copolymerization compared with, for example, a corresponding ester monomer. Therefore, when producing a polycarboxylic acid-based copolymer comprising a structural unit derived from an unsaturated polyalkylene glycol ether monomer and a structural unit derived from an unsaturated carboxylic acid monomer, the desired copolymer is produced.
- a polycarboxylic acid-based copolymer having a proportion there is a problem that it is not obtained at all, and even if it is obtained, the purity of the polymer is lowered and the quality of the polymer is deteriorated.
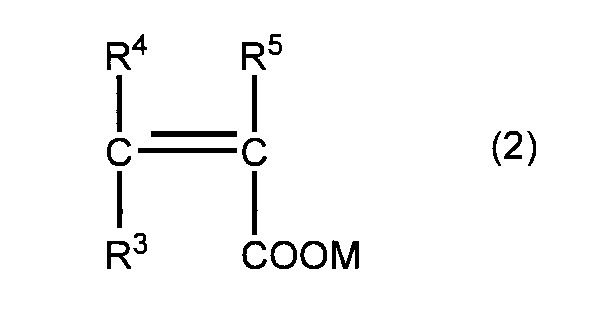
- the method for producing the polycarboxylic acid copolymer of the present invention comprises: Structural unit (I) derived from unsaturated polyalkylene glycol ether monomer (a) represented by general formula (1) and unsaturated carboxylic acid monomer (b) represented by general formula (2)
- a method for producing a polycarboxylic acid-based copolymer containing the derived structural unit (II) Polymerization of the monomer component containing the monomer (a) and the monomer (b) is being performed in the presence of a pH adjuster using a peroxide and a reducing agent in combination as a polymerization initiator. The pH is controlled to 3 or less.
- Y represents an alkenyl group having 2 to 8 carbon atoms.
- T is the same or different and represents an alkylene group having 1 to 5 carbon atoms or an aryl group having 6 to 9 carbon atoms.
- R 1 O represents one or more oxyalkylene groups having 2 to 18 carbon atoms
- m represents 0 or 1
- n represents the average number of moles added of the oxyalkylene group
- R 2 represents a hydrogen atom or an alkyl group having 1 to 5 carbon atoms.
- R 3 , R 4 , and R 5 are the same or different and each represents a hydrogen atom, a methyl group, or a —COOM group.
- M represents a hydrogen atom, a monovalent metal atom, or a divalent metal. Represents an atom, an ammonium group, or an organic amine group.
- the pH adjuster is an organic sulfonic acid and / or a salt thereof.
- the peroxide is hydrogen peroxide and the reducing agent is L-ascorbic acid.
- the copolymer is a cement admixture copolymer.
- the copolymer composition for a cement admixture of the present invention is composed of the structural unit (I) derived from the unsaturated polyalkylene glycol ether monomer (a) represented by the general formula (1) and the general formula (2).
- Y represents an alkenyl group having 2 to 8 carbon atoms.
- T is the same or different and represents an alkylene group having 1 to 5 carbon atoms or an aryl group having 6 to 9 carbon atoms.
- R 1 O represents one or more oxyalkylene groups having 2 to 18 carbon atoms
- m represents 0 or 1
- n represents the average number of moles added of the oxyalkylene group
- n is 1 to 500.
- R 2 represents a hydrogen atom or an alkyl group having 1 to 5 carbon atoms.
- R 3 , R 4 , and R 5 are the same or different and each represents a hydrogen atom, a methyl group, or a —COOM group.
- M represents a hydrogen atom, a monovalent metal atom, or a divalent metal. Represents an atom, an ammonium group, or an organic amine group.
- the said organic sulfonic acid is a compound represented by General formula (3).
- R 6- (Ph) p-SO 3 H (3) (In the general formula (3), R 6 represents a hydrogen atom or an alkyl group having 1 to 6 carbon atoms. Ph represents a phenylene group. P represents 0 or 1.)
- the compound represented by the general formula (3) is a low molecular compound having a molecular weight of 250 or less.
- R 6 in the general formula (3) is an alkyl group having 1 or 2 carbon atoms.
- Y in the general formula (1) is an alkenyl group having 4 or 5 carbon atoms.
- a method for producing a polycarboxylic acid copolymer comprising a structural unit derived from an unsaturated polyalkylene glycol ether monomer and a structural unit derived from an unsaturated carboxylic acid monomer.
- Another object of the present invention is to provide a method for producing a polycarboxylic acid copolymer, which can reduce the production cost of the copolymer and can provide an unprecedented high-performance cement admixture.
- a copolymer composition for a cement admixture comprising a structural unit derived from an unsaturated polyalkylene glycol ether monomer and a structural unit derived from an unsaturated carboxylic acid monomer,
- a copolymer composition for cement admixture that can provide an unprecedented high performance cement admixture can be provided.
- the method for producing the polycarboxylic acid copolymer of the present invention comprises: The structural unit (I) derived from the unsaturated polyalkylene glycol ether monomer (a) represented by the general formula (1) and the unsaturated carboxylic acid monomer represented by the general formula (2) ( This is a method for producing a polycarboxylic acid-based copolymer containing the structural unit (II) derived from b).
- the unsaturated polyalkylene glycol ether monomer (a) may be used alone or in combination of two or more.
- the unsaturated carboxylic acid monomer (b) may be used alone or in combination of two or more.
- the total content of the structural unit (I) and the structural unit (II) in the polycarboxylic acid copolymer obtained by the production method of the present invention is preferably 10 to 100% by mass, more preferably It is 20 to 100% by mass, more preferably 30 to 100% by mass. If the total content of the structural unit (I) and the structural unit (II) in the polycarboxylic acid copolymer obtained by the production method of the present invention is within the above range, a high-performance cement admixture It is possible to provide a polycarboxylic acid copolymer that can provide
- the content ratio of the structural unit (I) in the polycarboxylic acid copolymer obtained by the production method of the present invention is preferably 10 to 99% by mass, more preferably 20 to 99% by mass, and still more preferably 30%. ⁇ 99% by mass. If the content of the structural unit (I) in the polycarboxylic acid copolymer obtained by the production method of the present invention is within the above range, the polycarboxylic acid copolymer can provide a high-performance cement admixture. Coalescence can be provided.
- the content ratio of the structural unit (II) in the polycarboxylic acid copolymer obtained by the production method of the present invention is preferably 1 to 90% by mass, more preferably 1 to 80% by mass, and still more preferably 1 -70% by mass. If the content ratio of the structural unit (II) in the polycarboxylic acid copolymer obtained by the production method of the present invention is within the above range, the polycarboxylic acid copolymer can provide a high-performance cement admixture. Coalescence can be provided.
- Y represents an alkenyl group having 2 to 8 carbon atoms.
- Y is preferably an alkenyl group having 2 to 5 carbon atoms.
- Examples of Y include, for example, vinyl group, allyl group, methallyl group, 3-butenyl group, 3-methyl-3-butenyl group, 3-methyl-2-butenyl group, 2-methyl-3-butenyl group, 2-methyl
- Examples include -2-butenyl group and 1,1-dimethyl-2-propenyl group. Among these, an allyl group, a methallyl group, and a 3-methyl-3-butenyl group are preferable.
- T is the same or different and represents an alkylene group having 1 to 5 carbon atoms or an aryl group having 6 to 9 carbon atoms.
- m 0 or 1.
- R 1 O represents one or more oxyalkylene groups having 2 to 18 carbon atoms.
- R 1 O is preferably one or more oxyalkylene groups having 2 to 8 carbon atoms, more preferably one or more oxyalkylene groups having 2 to 4 carbon atoms.
- R 1 O include an oxyethylene group, an oxypropylene group, an oxybutylene group, and an oxystyrene group.
- Examples of the addition format of R 1 O include random addition, block addition, and alternate addition.
- an oxyethylene group contains an oxyethylene group as an essential component. More specifically, 50 mol% or more is more preferably an oxyethylene group, and more preferably 90 mol% or more is an oxyethylene group with respect to 100 mol% of all oxyalkylene groups.
- n is the average number of moles added of the oxyalkylene group, and n is 1 to 500.
- n is preferably 2 to 300, more preferably 5 to 300, still more preferably 10 to 300, particularly preferably 15 to 300, and most preferably 20 to 300.
- the smaller n is, the lower the hydrophilicity of the resulting polymer and the lower the dispersion performance.
- n exceeds 500, copolymerization reactivity may be reduced.
- R 2 represents a hydrogen atom or an alkyl group having 1 to 5 carbon atoms.
- R 3 , R 4 , and R 5 are the same or different and each represents a hydrogen atom, a methyl group, or a —COOM group.
- the above M represents a hydrogen atom, a monovalent metal atom, a divalent metal atom, an ammonium group, or an organic amine group.
- Any appropriate monovalent metal atom can be adopted as the monovalent metal atom.
- the monovalent metal atom For example, lithium, sodium, and potassium are mentioned.
- divalent metal atom any appropriate divalent metal atom can be adopted as the divalent metal atom.
- divalent metal atoms such as alkaline earth metal atoms such as calcium and magnesium.
- organic amine group any appropriate organic amine group can be adopted as long as it is a protonated organic amine.
- examples of the organic amine group include alkanolamine groups such as ethanolamine group, diethanolamine group, and triethanolamine group, and triethylamine group.
- Examples of the unsaturated polyalkylene glycol ether monomer (a) include 3-methyl-3-buten-1-ol, 3-methyl-2-buten-1-ol, 2-methyl-3-butene- Examples thereof include compounds obtained by adding 1 to 500 moles of alkylene oxide to unsaturated alcohols such as 2-ol, 2-methyl-2-buten-1-ol, and 2-methyl-3-buten-1-ol.
- unsaturated polyalkylene glycol ether monomer (a) examples include polyethylene glycol mono (3-methyl-3-butenyl) ether and polyethylene glycol mono (3-methyl-2-butenyl) ether.
- Examples of the unsaturated carboxylic acid monomer (b) include an unsaturated monocarboxylic acid monomer (b-1) and an unsaturated dicarboxylic acid monomer (b-2).
- the unsaturated carboxylic acid monomer (b) is preferably an unsaturated monocarboxylic acid monomer (b-1).
- the unsaturated monocarboxylic acid monomer (b-1) is preferably a (meth) acrylic acid monomer. Specific examples include acrylic acid, methacrylic acid, crotonic acid, and monovalent metal salts, divalent metal salts, ammonium salts, and organic amine salts thereof.
- the unsaturated monocarboxylic acid monomer (b-1) is more preferably (meth) acrylic acid and / or a salt thereof (monovalent metal salt, divalent metal salt, Ammonium salts, organic amine salts, and the like, and more preferably acrylic acid and / or salts thereof (monovalent metal salts, divalent metal salts, ammonium salts, organic amine salts, etc.).
- any appropriate unsaturated dicarboxylic acid monomer can be adopted as the unsaturated dicarboxylic acid monomer (b-2).
- Specific examples of the unsaturated dicarboxylic acid monomer (b-2) include maleic acid, maleic anhydride, fumaric acid, itaconic acid, citraconic acid, and monovalent metal salts and divalent metals thereof. Mention may be made of salts, ammonium salts and organic amine salts.
- unsaturated dicarboxylic acid monomer (b-2) maleic acid, maleic anhydride, fumaric acid, itaconic acid, citraconic acid, and salts thereof (monovalent metal salt, divalent metal salt, Ammonium salts, organic amine salts, etc., more preferably maleic acid, maleic anhydride, fumaric acid, citraconic acid, and salts thereof (monovalent metal salts, divalent metal salts, ammonium salts, organic amine salts) ⁇ , ⁇ -unsaturated dicarboxylic acid-based monomers.
- the monomer component used for the polymerization is other than the unsaturated polyalkylene glycol ether monomer (a) and the unsaturated carboxylic acid monomer (b). Further, any appropriate monomer (a) and another monomer (c) copolymerizable with the monomer (b) may be included. Other monomers (c) may be used alone or in combination of two or more.
- the other monomer (c) include half esters and diesters of the unsaturated dicarboxylic acid monomer (b-2) and an alcohol having 1 to 30 carbon atoms; Half amides and diamides of unsaturated dicarboxylic acid monomers (b-2) and amines having 1 to 30 carbon atoms; alkyl (poly) alkylene glycols and unsaturated dicarboxylic acid monomers (b-2) Half-esters and diesters; half of the unsaturated dicarboxylic acid monomer (b-2) and a glycol having 2 to 18 carbon atoms or a polyalkylene glycol having 2 to 500 moles of addition of these glycols Esters, diesters; methyl (meth) acrylate, ethyl (meth) acrylate, propyl (meth) acrylate, glycidyl (meth) acrylate, methyl Esters of unsaturated monocarboxylic acid monomers (b-1) such as crot
- Saturated amides Saturated amides; Unsaturated cyanides such as (meth) acrylonitrile and ⁇ -chloroacrylonitrile; Acetic acid Nyl, unsaturated esters such as vinyl propionate; aminoethyl (meth) acrylate, methylaminoethyl (meth) acrylate, dimethylaminoethyl (meth) acrylate, dimethylaminopropyl (meth) acrylate, (meth) Unsaturated amines such as dibutylaminoethyl acrylate and vinyl pyridine; divinyl aromatics such as divinylbenzene; cyanurates such as triallyl cyanurate; allyls such as (meth) allyl alcohol and glycidyl (meth) allyl ether; Unsaturated amino compounds such as dimethylaminoethyl (meth) acrylate; methoxypolyethylene glycol monovinyl ether
- polymerization of a monomer component containing the monomer (a) and the monomer (b) is started, and polymerization of a peroxide and a reducing agent is started.
- the pH during polymerization is controlled to 3 or less in the presence of a pH adjuster.
- Polymerization of the monomer component can be performed by any appropriate method. Examples thereof include solution polymerization and bulk polymerization. Examples of the solution polymerization method include a batch method and a continuous method. Solvents that can be used in solution polymerization include water; alcohols such as methyl alcohol, ethyl alcohol, and isopropyl alcohol; aromatic or aliphatic hydrocarbons such as benzene, toluene, xylene, cyclohexane, and n-hexane; esters such as ethyl acetate Compounds; ketone compounds such as acetone and methyl ethyl ketone; cyclic ether compounds such as tetrahydrofuran and dioxane; and the like.
- Solvents that can be used in solution polymerization include water; alcohols such as methyl alcohol, ethyl alcohol, and isopropyl alcohol; aromatic or aliphatic hydrocarbons such as benzene, toluen
- a chain transfer agent can be used.
- the molecular weight of the resulting copolymer can be easily adjusted.
- chain transfer agent Any appropriate chain transfer agent can be adopted as the chain transfer agent. Specifically, for example, mercaptoethanol, thioglycerol, thioglycolic acid, 2-mercaptopropionic acid, 3-mercaptopropionic acid, thiomalic acid, octyl thioglycolate, octyl 3-mercaptopropionate, 2-mercaptoethanesulfonic acid Thiol chain transfer agents such as n-dodecyl mercaptan, octyl mercaptan, butyl thioglycolate; halides such as carbon tetrachloride, methylene chloride, bromoform, bromotrichloroethane; secondary alcohols such as isopropanol; phosphorous acid, next Phosphorous acid and its salts (sodium hypophosphite, potassium hypophosphite, etc.), sulfurous acid, hydrogen sulfite, dithionic
- a peroxide and a reducing agent are used in combination as a polymerization initiator.
- peroxide any appropriate peroxide can be adopted as the peroxide.
- persulfates such as ammonium persulfate, sodium persulfate, potassium persulfate; hydrogen peroxide; peroxides such as benzoyl peroxide, lauroyl peroxide, sodium peroxide, t-butyl hydroperoxide, cumene hydroperoxide; Etc.
- any appropriate reducing agent can be adopted as the reducing agent.
- salts of metals in a low valence state such as iron (II), tin (II), titanium (III), chromium (II), V (II), Cu (II), etc.
- Amine compounds such as monoethanolamine, diethanolamine, triethanolamine, hydroxylamine, hydroxylamine hydrochloride, hydrazine and their salts; sodium dithionite, sodium formaldehyde sulfoxylate, sodium hydroxymethanesulfinate dihydrate; Organic compounds containing —SH group, —SO 2 H group, —NHNH 2 group, —COCH (OH) — group and salts thereof; alkali metal sulfites such as sodium sulfite, sodium bisulfite and metabisulfite; Phosphoric acid, sodium hypophosphite, sodium hydrosulfite, sodium hyponitrite Lower oxides such as D-fructose and D-glucose; invert sugars such as thiourea and thiourea dioxide; L-ascorbic acid (salt), L-ascorbic acid ester, erythorbic acid (salt), Erythorbic acid ester; and the like.
- the combination of the peroxide and the reducing agent is preferably a combination of a water-soluble peroxide and a reducing agent, such as a combination of hydrogen peroxide and L-ascorbic acid, hydrogen peroxide and erythorbic acid, and the like.
- a combination of hydrogen peroxide and a mole salt, and a combination of sodium persulfate and sodium bisulfite is a particularly preferable combination.
- a particularly preferable combination is a combination of hydrogen peroxide and L-ascorbic acid in that the effects of the present invention can be expressed more effectively.
- the amount of the peroxide used is preferably 0.01 to 30 mol%, more preferably 0.1 to 20 mol%, and still more preferably 0.5 to 10 mol, based on the total amount of the monomer components. %.
- an unreacted monomer may increase that the usage-amount of the said peroxide is less than 0.01 mol% with respect to the total amount of a monomer component.
- the amount of the peroxide used exceeds 30 mol% with respect to the total amount of the monomer components, a polycarboxylic acid having a large amount of oligomers may be obtained.
- the amount of the reducing agent used is preferably 0.1 to 500 mol%, more preferably 1 to 200 mol%, still more preferably 10 to 100 mol% with respect to the peroxide.
- the amount of the reducing agent used is less than 0.1 mol% with respect to the peroxide, active radicals are not sufficiently generated, and the amount of unreacted monomers may increase.
- the amount of the reducing agent used exceeds 500 mol% with respect to the peroxide, there is a risk that the reducing agent remaining without reacting with hydrogen peroxide will increase.
- the peroxide and the reducing agent are always present in the reaction system. Specifically, it is preferable that the peroxide and the reducing agent are not added simultaneously. If peroxide and reducing agent are simultaneously added at once, the peroxide and reducing agent react rapidly, generating a large amount of reaction heat immediately after the addition, making reaction control difficult, and then radical concentration Therefore, a large amount of unreacted monomer components may remain. Furthermore, the radical concentration with respect to the monomer component is extremely different between the initial and second half of the reaction, so the molecular weight distribution becomes extremely large, and the performance when the resulting copolymer is used as a cement admixture may be reduced. There is.
- the time from when one of the peroxide and the reducing agent is charged to when the other is started is preferably within 5 hours, more preferably within 3 hours.
- the polymerization reaction temperature is preferably 30 to 90 ° C, more preferably 35 to 85 ° C, and further preferably 40 to 80 ° C. If the polymerization reaction temperature is out of the above range, the polymerization rate may be lowered or the productivity may be lowered.
- the polymerization time is preferably 0.5 to 10 hours, more preferably 0.5 to 8 hours, and further preferably 1 to 6 hours. If the polymerization time is out of the above range, the polymerization rate may be lowered or the productivity may be lowered.
- Any appropriate method can be adopted as a method for charging the monomer component into the reaction vessel.
- a method in which the entire amount is initially charged into the reaction vessel a method in which the entire amount is divided or continuously charged into the reaction vessel, a method in which a part is initially charged in the reaction vessel and the remainder is divided or continuously charged into the reaction vessel, etc.
- a method in which a part is initially charged in the reaction vessel and the remainder is divided or continuously charged into the reaction vessel etc.
- a method in which the total amount of monomer (a) and the total amount of monomer (b) are continuously charged into the reaction vessel, a part of monomer (a) is initially charged into the reaction vessel A method in which the remainder of the monomer (a) and the entire amount of the monomer (b) are continuously charged into the reaction vessel, a part of the monomer (a) and a part of the monomer (b) in the reaction vessel In the initial stage, and the remainder of the monomer (a) and the remainder of the monomer (b) are alternately charged into the reaction vessel in several batches.
- the polymerization of the monomer component is carried out in the presence of a pH adjuster while controlling the pH during polymerization to 3 or less.
- the polymerization is carried out by controlling the pH to 2 to 3.
- Sufficient copolymerization of unsaturated polyalkylene glycol ether monomers can be easily expressed by performing polymerization of the above monomer components in the presence of a pH adjuster while controlling the pH during polymerization to 3 or less.
- the production cost of the polycarboxylic acid copolymer to be produced can be reduced, and a polycarboxylic acid copolymer that can provide an unprecedented high-performance cement admixture can be produced.
- Examples of the pH adjusting agent include phosphoric acid and / or a salt thereof, organic sulfonic acid and / or a salt thereof, hydrochloric acid and / or a salt thereof, nitric acid and / or a salt thereof, sulfuric acid and / or a salt thereof.
- at least one selected from phosphoric acid and / or a salt thereof and organic sulfonic acid and / or a salt thereof is preferable, and an organic sulfonic acid and / or a salt thereof is more preferable in that the addition amount can be reduced.
- any appropriate salt can be adopted.
- examples thereof include alkali metal salts, alkaline earth metal salts, ammonium salts, and organic ammonium salts. Only 1 type of pH adjuster may be used and it may use 2 or more types together.
- organic sulfonic acid and / or salt thereof examples include p-toluenesulfonic acid and / or a hydrate thereof, methanesulfonic acid and / or a salt thereof, and the like.
- the amount of the pH adjuster used is preferably 0.01 to 5% by mass, more preferably 0.05 to 4% by mass, and still more preferably 0.05 to 2.5% by mass, based on the total amount of the monomer components. %. If the amount of the pH adjuster used is too large, the pH during the polymerization will be too low, which may lead to inappropriate polymerization conditions. Moreover, the ratio of the usage-amount of the pH adjuster with respect to the total amount of said monomer component is substantially the same as the mass ratio of the pH adjuster with respect to the mass of the copolymer in the composition obtained. Therefore, the mass ratio of the pH adjuster to the mass of the copolymer in the composition obtained is preferably 0.01 to 5% by mass, more preferably 0.05 to 4% by mass, and still more preferably 0.05. ⁇ 2.5% by mass.
- the polymerization of the monomer component is performed in the presence of a pH adjuster while controlling the pH during the polymerization to 3 or less.
- the pH is adjusted to any appropriate pH. good.
- the pH is preferably adjusted to 4-7 after polymerization.
- the polycarboxylic acid copolymer obtained by the production method of the present invention has a mass average molecular weight (Mw) of preferably 10,000 to 300,000, more preferably 10,000 to 100,000, and still more preferably 10,000 to 80,000.
- Mw mass average molecular weight
- a high-performance cement admixture can be provided.
- the polycarboxylic acid copolymer obtained by the production method of the present invention can be suitably used as a copolymer for cement admixture.
- the content ratio of the polycarboxylic acid copolymer in the obtained cement admixture is preferably It is 5 to 100% by mass, more preferably 10 to 100% by mass, and further preferably 15 to 100% by mass. This is because the effects of the present invention can be sufficiently exhibited.
- the above cement admixture may contain any appropriate other component in addition to the polycarboxylic acid copolymer obtained by the production method of the present invention.
- the above cement admixture can contain one or more kinds of any appropriate cement dispersant.
- the blending mass ratio of the polycarboxylic acid copolymer obtained by the production method of the present invention to the cement dispersant cement admixture of the present invention / cement dispersant
- the mass ratio (mass%) in terms of solid content is preferably 1 to 99/99 to 1, more preferably Is from 5 to 95/95 to 5, more preferably from 10 to 90/90 to 10.
- cement dispersant examples include the following cement dispersants.
- Polyalkylaryl sulfonate system such as naphthalene sulfonic acid formaldehyde condensate, methyl naphthalene sulfonic acid formaldehyde condensate, anthracene sulfonic acid formaldehyde condensate; melamine formalin resin sulfonate system such as melamine sulfonic acid formaldehyde condensate; amino Aromatic amino sulfonates such as aryl sulfonic acid-phenol-formaldehyde condensates; lignin sulfonates such as lignin sulfonates and modified lignin sulfonates; polystyrene sulfonates; Various sulfonic acid-based dispersants having a sulfonic acid group.
- Polyalkylene glycol mono (meth) acrylic acid ester monomers, (meth) acrylic acid monomers, and single amounts thereof described in JP-B-59-18338 and JP-A-7-223852 A copolymer obtained by polymerizing a monomer copolymerizable with a polymer; a polyether compound described in JP-A-7-53645, JP-A-8-208769, JP-A-8-208770; Various polycarboxylic acid dispersants having a (poly) oxyalkylene group and a carboxyl group in the molecule, such as a hydrophilic graft polymer obtained by graft polymerization of an unsaturated carboxylic acid monomer.
- cement additive any appropriate cement additive (cement additive).
- cement additive water-soluble polymer substances, polymer emulsions, curing retarders, early strengthening agents / accelerators, antifoaming agents, AE agents, waterproofing agents, rust prevention agents, crack reducing agents, swelling agents, cement wetting agents, thickening agents Agents, separation reducing agents, flocculants, drying shrinkage reducing agents, strength enhancers, self-leveling agents, coloring agents, antifungal agents and the like.
- cement additive only one kind of cement additive (cement additive) as mentioned above may be used, or two or more kinds may be used in combination.
- the following (1) to (7) are particularly preferred embodiments of the cement admixture.
- ⁇ 1> A combination of the above-mentioned cement admixture and ⁇ 2> oxyalkylene-based antifoaming agent, in which two components are essential.
- the oxyalkylene-based antifoaming agent include polyoxyalkylenes, polyoxyalkylene alkyl ethers, polyoxyalkylene acetylene ethers and polyoxyalkylene alkylamines, preferably polyoxyalkylene alkylamines.
- the blending mass ratio of ⁇ 2> oxyalkylene-based antifoaming agent is preferably in the range of 0.01 to 20% by mass with respect to ⁇ 1> the cement admixture.
- ⁇ 1> A combination of the above-mentioned cement admixture, ⁇ 2> oxyalkylene-based antifoaming agent, and ⁇ 3> AE agent, which essentially comprises three components.
- the oxyalkylene-based antifoaming agent include polyoxyalkylenes, polyoxyalkylene alkyl ethers, polyoxyalkylene acetylene ethers and polyoxyalkylene alkylamines, preferably polyoxyalkylene alkylamines. .
- the blending mass ratio of ⁇ 2> oxyalkylene-based antifoaming agent is preferably in the range of 0.01 to 20% by mass with respect to ⁇ 1> the cement admixture.
- the blending mass ratio of ⁇ 3> AE agent is preferably in the range of 0.001 to 2% by mass with respect to ⁇ 1> the cement admixture.
- Examples of the oxyalkylene-based antifoaming agent include polyoxyalkylenes, polyoxyalkylene alkyl ethers, polyoxyalkylene acetylene ethers and polyoxyalkylene alkylamines, preferably polyoxyalkylene alkylamines.
- the mixing ratio of ⁇ 1> the cement admixture and ⁇ 2> copolymer is ⁇ 1> the mass ratio of the cement admixture / ⁇ 2> copolymer, preferably 5/95 to 95/5. More preferably, it is 10/90 to 90/10.
- the blending mass ratio of the ⁇ 3> oxyalkylene-based antifoaming agent is preferably in the range of 0.01 to 20% by mass with respect to the total amount of ⁇ 1> the cement admixture and ⁇ 2> copolymer.
- sulfonic acid-based dispersant having a sulfonic acid group in the molecule, which essentially includes two components.
- the sulfonic acid-based dispersant include amino sulfonic acid-based compounds such as lignin sulfonate, naphthalene sulfonic acid formalin condensate, melamine sulfonic acid formalin condensate, polystyrene sulfonate, aminoaryl sulfonic acid-phenol-formaldehyde condensate, and the like.
- a dispersing agent is mentioned.
- the mixing ratio of ⁇ 1> the cement admixture and ⁇ 2> the sulfonic acid dispersant is ⁇ 1> the mass ratio of the cement admixture / ⁇ 2> the sulfonic acid dispersant, preferably 5/95 to 95/5, more preferably 10/90 to 90/10.
- a combination of the above-mentioned cement admixture and ⁇ 2> material separation reducing agent which essentially includes two components.
- the material separation reducing agent include various thickeners such as nonionic cellulose ethers, a hydrophobic substituent composed of a hydrocarbon chain having 4 to 30 carbon atoms as a partial structure, and an alkylene oxide having 2 to 18 carbon atoms. Examples thereof include compounds having a polyoxyalkylene chain added in an average addition mole number of 2 to 300.
- the mixing ratio of ⁇ 1> the cement admixture and ⁇ 2> the material separation reducing agent is a mass ratio of ⁇ 1> the cement admixture / ⁇ 2> the material separation reducing agent, preferably 10/90 to 99.99. 99 / 0.01, more preferably 50/50 to 99.9 / 0.1.
- the cement admixture in this combination is suitable as high fluidity concrete, self-filling concrete, and self-leveling agent.
- the retarder include oxycarboxylic acids such as gluconic acid (salt) and citric acid (salt), sugars such as glucose, sugar alcohols such as sorbitol, and phosphonic acids such as aminotri (methylenephosphonic acid), Oxycarboxylic acids are preferred.
- the mixing ratio of ⁇ 1> the cement admixture and ⁇ 2> retarder is the mass ratio of ⁇ 1> the cement admixture / ⁇ 2> retarder, preferably 50/50 to 99.9 / 0. 1, more preferably 70/30 to 99/1.
- ⁇ 1> A combination of the above-mentioned cement admixture and ⁇ 2> accelerator that essentially requires two components.
- the accelerator include soluble calcium salts such as calcium chloride, calcium nitrite and calcium nitrate, chlorides such as iron chloride and magnesium chloride, formates such as thiosulfate, formic acid and calcium formate, and the like.
- the mixing ratio of ⁇ 1> the cement admixture and ⁇ 2> accelerator is a mass ratio of ⁇ 1> the cement admixture / ⁇ 2> accelerator, preferably 10/90 to 99.9 / 0. 1, more preferably 20/80 to 99/1.
- the cement admixture using the polycarboxylic acid copolymer obtained by the production method of the present invention can be used by adding to a cement composition such as cement paste, mortar or concrete.
- cement composition Any appropriate cement composition can be adopted as the cement composition.
- what contains cement, water, an aggregate, and an antifoamer is mentioned.
- any appropriate cement can be adopted as the cement.
- Portland cement ordinary, early strength, very early strength, moderate heat, sulfate-resistant and low alkali type of each
- various mixed cements blast furnace cement, silica cement, fly ash cement
- white Portland cement alumina cement
- grout cement oil well cement
- low exothermic cement low exothermic blast furnace cement, fly ash mixed low exothermic blast furnace cement, belite High-content cement
- ultra-high-strength cement cement-based solidified material
- eco-cement cement produced from one or more of municipal waste incineration ash and sewage sludge incineration ash
- fine powder such as blast furnace slag, fly ash, cinder ash, clinker ash, husk ash, silica powder, limestone powder, or gypsum may be added.
- any appropriate aggregate can be adopted as the aggregate. Examples include gravel, crushed stone, granulated slag, and recycled aggregate.
- refractory aggregates such as siliceous, clay, zircon, high alumina, silicon carbide, graphite, chromium, chromic, magnesia, etc. can be used.
- antifoaming agent Any appropriate antifoaming agent can be adopted as the antifoaming agent.
- antifoaming agents described in paragraphs 0041 to 0042 of Japanese Patent No. 3683176 can be mentioned.
- the blending amount and unit water amount per 1 m 3 of concrete are preferably 100 to 185 kg / m 3 per unit water amount, water /
- the cement ratio is 10 to 70% by mass, more preferably the unit water amount is 120 to 175 kg / m 3 and the water / cement ratio is 20 to 65% by mass.
- the amount of the cement admixture added to the cement composition is preferably 0.01 to 10% by mass, more preferably 0.05 to 8% by mass, when the total amount of cement is 100% by mass. More preferably, the content is 0.1 to 5% by mass. There exists a possibility that it may be inferior to the performance as a cement composition as the said addition amount is less than 0.01 mass%. If the amount added exceeds 10% by mass, the economy may be inferior.
- the above cement composition may be prepared by blending the above components by any appropriate method. For example, the method of kneading in a mixer is mentioned.
- copolymer composition for cement admixture comprises the structural unit (I) derived from the unsaturated polyalkylene glycol ether monomer (a) represented by the general formula (1) and the general formula (2). And a polycarboxylic acid copolymer comprising a structural unit (II) derived from an unsaturated carboxylic acid monomer (b), a non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or its Including salt.
- the polycarboxylic acid copolymer contained in the cement admixture copolymer composition of the present invention is derived from the unsaturated polyalkylene glycol ether monomer (a) represented by the general formula (1). 1 type of structural unit (I) may be included, or 2 or more types may be included.
- the polycarboxylic acid copolymer in the present invention contains only one type of structural unit (II) derived from the unsaturated carboxylic acid monomer (b) represented by the general formula (2). Or two or more of them may be included.
- the structural unit (I) is a structural unit in which a polymerizable unsaturated double bond of Y in the general formula (1) is cleaved by polymerization to form a single bond.
- the structural unit (I) derived from the unsaturated polyalkylene glycol ether monomer (a) represented by the general formula (1) is represented by the general formula (I). expressed.
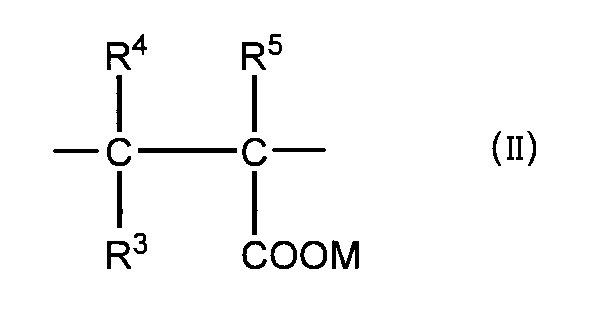
- (II) is represented by general formula (II).
- R 3 , R 4 , and R 5 are the same or different and each represents a hydrogen atom, a methyl group, or a —COOM group.
- M represents a hydrogen atom, a monovalent metal atom, or a divalent metal. Represents an atom, an ammonium group, or an organic amine group.
- the total content of the structural unit (I) and the structural unit (II) in the polycarboxylic acid copolymer contained in the cement admixture copolymer composition of the present invention is preferably 10 to The amount is 100% by mass, more preferably 20 to 100% by mass, and still more preferably 30 to 100% by mass. If the total content of the structural unit (I) and the structural unit (II) in the polycarboxylic acid copolymer in the present invention is within the above range, a poly which can provide a high-performance cement admixture. Carboxylic acid copolymers can be provided.
- the content ratio of the structural unit (I) in the polycarboxylic acid copolymer contained in the cement admixture copolymer composition of the present invention is preferably 10 to 99% by mass, more preferably 20 to 99%. % By mass, more preferably 30 to 99% by mass. If the content ratio of the structural unit (I) in the polycarboxylic acid copolymer in the present invention is within the above range, a polycarboxylic acid copolymer capable of providing a high-performance cement admixture can be provided. .
- the content ratio of the structural unit (II) in the polycarboxylic acid copolymer contained in the cement admixture copolymer composition of the present invention is preferably 1 to 90% by mass, more preferably 1 to 80%. % By mass, more preferably 1 to 70% by mass. If the content ratio of the structural unit (II) in the polycarboxylic acid copolymer in the present invention is within the above range, a polycarboxylic acid copolymer capable of providing a high-performance cement admixture can be provided. .
- Y, T, m, R 1 O, n, and R 2 in the general formula (1) are the above ⁇ 1. This is the same as described in the item “ Production method of polycarboxylic acid-based copolymer” .
- R 3 R 4, R 5 in the general formula (2), of the ⁇ 1. This is the same as described in the item “ Production method of polycarboxylic acid-based copolymer” .
- the monomer components used for the polymerization are unsaturated polyalkylene glycol ether monomer (a) and unsaturated carboxylic acid monomer (b).
- any appropriate monomer (a) and another monomer (c) copolymerizable with the monomer (b) may be included.
- Other monomers (c) may be used alone or in combination of two or more.
- the polycarboxylic acid copolymer contained in the cement admixture copolymer composition of the present invention has a mass average molecular weight (Mw) of preferably 10,000 to 300,000, more preferably 10,000 to 100,000, and still more preferably 10,000 to 10,000. 80000.
- Mw mass average molecular weight
- a high-performance copolymer composition for cement admixture can be provided.
- the copolymer composition for cement admixture of the present invention contains a non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof.
- a non-polymerizable organic sulfonic acid and / or salt thereof having a molecular weight of 300 or less any appropriate non-polymerizable organic sulfonic acid and / or salt thereof having a molecular weight of 300 or less can be adopted.
- Any appropriate salt can be adopted as the salt. Examples thereof include alkali metal salts, alkaline earth metal salts, ammonium salts, and organic ammonium salts. Only one non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof may be used, or two or more thereof may be used in combination.
- the organic sulfonic acid is preferably a compound represented by the general formula (3).
- R 6- (Ph) p-SO 3 H (3) (In the general formula (3), R 6 represents a hydrogen atom or an alkyl group having 1 to 6 carbon atoms. Ph represents a phenylene group. P represents 0 or 1.)
- the compound represented by the general formula (3) is preferably a low molecular compound having a molecular weight of 250 or less. More preferably, it is a low molecular weight compound having a molecular weight of 200 or less.
- R 6 in the general formula (3) is preferably an alkyl group having 1 or 2 carbon atoms. Specific examples include a methyl group and an ethyl group.
- Y in the general formula (1) is an alkenyl group having 4 or 5 carbon atoms.
- non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof include paratoluenesulfonic acid and / or a hydrate thereof, methanesulfonic acid and / or a salt thereof.
- the content ratio of the non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof in the copolymer composition for cement admixture of the present invention is contained in the copolymer composition for cement admixture of the present invention.
- the content is preferably 0.01 to 5% by mass, more preferably 0.05 to 4% by mass, and still more preferably 0.05 to 2.5% by mass with respect to the polycarboxylic acid copolymer.
- a copolymer composition for a cement admixture that can provide an agent can be provided.
- the copolymer composition for cement admixture of the present invention can be produced by any appropriate method.
- polymerization of a monomer component including the monomer (a) and the monomer (b) is used in combination with a peroxide and a reducing agent as a polymerization initiator, and a non-polymerization having a molecular weight of 300 or less.
- a pH adjuster that essentially contains a functional organic sulfonic acid and / or salt thereof, the pH during polymerization is controlled to 3 or less.
- the polymerization of the monomer component can be performed by any appropriate method.
- Examples thereof include the method described in the item “ Production method of polycarboxylic acid-based copolymer” .
- a chain transfer agent can be used.
- the molecular weight of the resulting copolymer can be easily adjusted.
- chain transfer agent Any appropriate chain transfer agent can be adopted as the chain transfer agent. Specifically, for example, the above ⁇ 1. Examples include the chain transfer agent described in the item of “Production Method of Polycarboxylic Acid Copolymer” >> .
- the copolymer composition for a cement admixture of the present invention it is preferable to use a peroxide and a reducing agent in combination as a polymerization initiator.
- any appropriate peroxide can be adopted as the peroxide.
- any appropriate reducing agent can be adopted as the reducing agent.
- the reducing agent demonstrated in the item of the manufacturing method >> of a polycarboxylic acid-type copolymer is mentioned.
- Any appropriate method can be adopted as a method of charging the monomer component into the reaction vessel.
- the polymerization of the monomer component is carried out by adjusting pH which essentially contains a non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof. It is preferable to carry out by controlling the pH during polymerization to 3 or less in the presence of an agent. Preferably, the polymerization is carried out by controlling the pH to 2 to 3. Sufficient copolymerization of unsaturated polyalkylene glycol ether monomers can be easily expressed by performing polymerization of the above monomer components in the presence of a pH adjuster while controlling the pH during polymerization to 3 or less. The production cost of the polycarboxylic acid copolymer to be produced can be reduced, and a copolymer composition for a cement admixture that can provide an unprecedented high performance cement admixture can be produced.
- any appropriate pH adjustor can be adopted as long as it is a pH adjuster that essentially contains a non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof.
- the non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof include the compounds described above.
- Examples of the pH adjuster other than the non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or its salt include, for example, phosphoric acid and / or its salt, hydrochloric acid and / or its salt, nitric acid and / or its salt, sulfuric acid and And / or a salt thereof. Any appropriate salt can be adopted as the salt. Examples thereof include alkali metal salts, alkaline earth metal salts, ammonium salts, and organic ammonium salts. Only 1 type of pH adjuster may be used and it may use 2 or more types together.
- the pH adjuster essentially comprises non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof for the polymerization of the monomer component.
- the pH is preferably adjusted to 4-7 after polymerization.
- the copolymer composition for a cement admixture of the present invention is a composition other than a polycarboxylic acid copolymer and a non-polymerizable organic sulfonic acid having a molecular weight of 300 or less and / or a salt thereof, as long as the effects of the present invention are not impaired. Any suitable component may be included.
- copolymer composition for a cement admixture of the present invention can be used as a cement admixture together with any appropriate component as necessary.
- components that can be used as a cement admixture in combination with the copolymer composition for cement admixture of the present invention include a cement dispersant.
- the blending mass ratio of the copolymer composition for a cement admixture of the present invention and the cement dispersant is:
- the mass ratio (mass%) in terms of solid content is preferably 1 to 99/99 to 1, More preferably, it is 5 to 95/95 to 5, and still more preferably 10 to 90/90 to 10. Only one cement dispersant may be used, or two or more cement dispersants may be used in combination.
- cement dispersant examples include the above ⁇ 1.
- cement dispersants examples include the cement dispersants described in the section “ Production method of polycarboxylic acid-based copolymer” .
- cement additives examples of the components that can be used as a cement admixture in combination with the copolymer composition for cement admixture of the present invention include water-soluble polymer substances, polymer emulsions, curing retarders, early-stage additives, and the like. Strong agent / accelerator, antifoaming agent, AE agent, waterproofing agent, rust preventive agent, crack reducing agent, swelling agent, cement wetting agent, thickener, separation reducing agent, flocculant, drying shrinkage reducing agent, strength enhancer And cement additives (cement additives) such as self-leveling agents, colorants, and fungicides.
- cement additives cement additives
- cement additive only one kind of cement additive (cement additive) as mentioned above may be used, or two or more kinds may be used in combination.
- the cement admixture obtained from the copolymer composition for cement admixture of the present invention can be used by adding to a cement composition such as cement paste, mortar or concrete.
- cement composition Any appropriate cement composition can be adopted as the cement composition.
- what contains cement, water, an aggregate, and an antifoamer is mentioned.
- any appropriate cement can be adopted as the cement.
- any appropriate aggregate can be adopted as the aggregate.
- antifoaming agent Any appropriate antifoaming agent can be adopted as the antifoaming agent.
- antifoaming agents described in paragraphs 0041 to 0042 of Japanese Patent No. 3683176 can be mentioned.
- the compounding amount and unit water content per concrete 1 m 3, of the ⁇ 1. This is the same as described in the item “ Production method of polycarboxylic acid-based copolymer” .
- the above cement composition may be prepared by blending the above components by any appropriate method. For example, the method of kneading in a mixer is mentioned.
- Fine aggregate and cement were put into a mixer and kneaded for 10 seconds. Then, water containing cement admixture and coarse aggregate were added and kneaded for 90 seconds, and then the concrete was discharged.
- the slump value, slump flow value, and air amount of the obtained concrete were measured in accordance with Japanese Industrial Standards (JIS A1101, 1128, 6204).
- the resulting copolymer composition (1) contained a copolymer (1P) and a non-polymerizable organic sulfonic acid (salt) (1S).
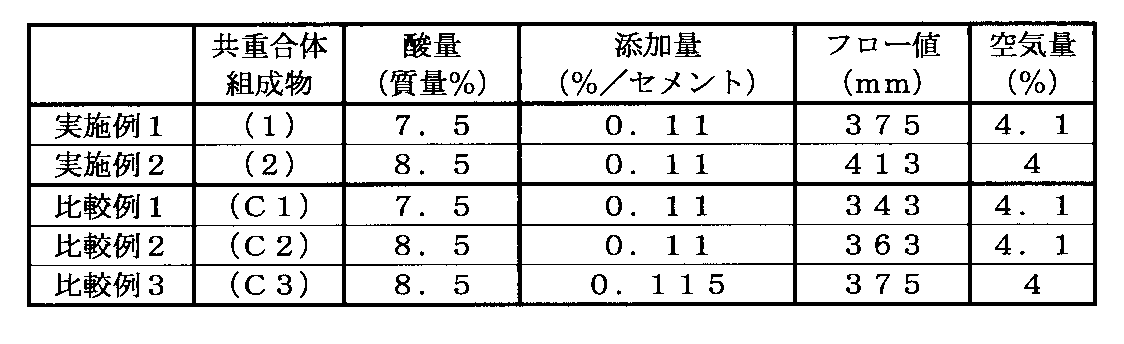
- the results are shown in Table 1.
- a concrete test was performed using the obtained copolymer composition (1) as a cement admixture. The results are shown in Table 2.
- the obtained copolymer composition (2) contained a copolymer (2P) and a non-polymerizable organic sulfonic acid (salt) (2S).
- the results are shown in Table 1.
- a concrete test was performed using the obtained copolymer composition (2) as a cement admixture. The results are shown in Table 2.
- the obtained copolymer composition (3) contained a copolymer (3P) and a non-polymerizable organic sulfonic acid (salt) (3S).
- the results are shown in Table 1.
- the resulting copolymer composition (4) contained a copolymer (4P) and a non-polymerizable organic sulfonic acid (salt) (4S).
- the results are shown in Table 1.
- the resulting copolymer composition (5) contained a copolymer (5P) and a non-polymerizable organic sulfonic acid (salt) (5S).
- a copolymer 5P
- a non-polymerizable organic sulfonic acid salt
- the results are shown in Table 1.
- a pH adjusting agent pH adjusting agent
- the obtained copolymer composition (6) contained the copolymer (6P) and non-polymerizable organic sulfonic acid (salt) (6S).
- the results are shown in Table 1.
- the obtained copolymer composition (7) contained the copolymer (7P) and non-polymerizable organic sulfonic acid (salt) (7S).
- the results are shown in Table 1.
- the obtained copolymer composition (C1) contained a copolymer (C1P) but did not contain a non-polymerizable organic sulfonic acid (salt).
- the results are shown in Table 1.
- a concrete test was performed using the obtained copolymer composition (C1) as a cement admixture. The results are shown in Table 2.
- the obtained copolymer composition (C2) contained a copolymer (C2P) but did not contain a non-polymerizable organic sulfonic acid (salt).
- the results are shown in Table 1.
- a concrete test was performed using the obtained copolymer composition (C2) as a cement admixture. The results are shown in Table 2.
- the obtained copolymer composition (C3) contained a copolymer (C3P) but did not contain a non-polymerizable organic sulfonic acid (salt).
- the results are shown in Table 1.
- a concrete test was performed using the obtained copolymer composition (C3) as a cement admixture. The results are shown in Table 2.
- the obtained copolymer composition (C4) contained a copolymer (C4P) but did not contain a non-polymerizable organic sulfonic acid (salt).
- the results are shown in Table 1.
- the obtained copolymer composition (C5) contained a copolymer (C5P) but did not contain a non-polymerizable organic sulfonic acid (salt).
- the results are shown in Table 1.
- the acid amount in Table 2 is a numerical value obtained by converting charged acrylic acid into sodium acrylate.
- Example 1 From the results of Example 1 and Comparative Example 1, the flow value is higher in Comparative Example 1 than in Comparative Example 1 in Example 1 where the polymer content is the same and the polymer content is high. From this, the copolymer composition (1) obtained in Example 1 is more fluid than the copolymer composition (C1) obtained in Comparative Example 1 when used as a cement admixture. I understand that it is expensive.
- Example 2 and Comparative Examples 2 and 3 From the results of Example 2 and Comparative Examples 2 and 3, the flow value is higher in Comparative Example 2 and 3 than in Comparative Example 2 and 3 in Example 2 where the polymer content is the same in the same acid amount. From this, the copolymer composition (2) obtained in Example 2 was used as a cement admixture compared with the copolymer compositions (C2) and (C3) obtained in Comparative Examples 2 and 3. It can be seen that the fluidity is high. Moreover, it turns out that the addition amount reduction
- the polycarboxylic acid copolymer obtained by the production method of the present invention is suitably used as a cement admixture.
- the cement admixture is suitably used for cement compositions such as cement paste, mortar, and concrete.
- the copolymer composition for a cement admixture of the present invention is suitably used for a cement admixture.
- the cement admixture is suitably used for cement compositions such as cement paste, mortar, and concrete.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Ceramic Engineering (AREA)
- Materials Engineering (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Structural Engineering (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Curing Cements, Concrete, And Artificial Stone (AREA)
- Macromonomer-Based Addition Polymer (AREA)
Abstract
Description
一般式(1)で表される不飽和ポリアルキレングリコールエーテル系単量体(a)由来の構造単位(I)と一般式(2)で表される不飽和カルボン酸系単量体(b)由来の構造単位(II)とを含むポリカルボン酸系共重合体の製造方法であって、
該単量体(a)と該単量体(b)とを含む単量体成分の重合を、過酸化物と還元剤を重合開始剤として併用して、pH調整剤の存在下で重合中のpHを3以下に制御して行う。
R6-(Ph)p-SO3H (3)
(一般式(3)中、R6は水素原子または炭素数1~6のアルキル基を表す。Phはフェニレン基を表す。pは0または1を表す。)
本発明によれば、不飽和ポリアルキレングリコールエーテル系単量体由来の構造単位と不飽和カルボン酸系単量体由来の構造単位とを含むセメント混和剤用の共重合体組成物であって、従来にない高性能のセメント混和剤を提供しうる、セメント混和剤用共重合体組成物を提供することができる。
本発明のポリカルボン酸系共重合体の製造方法は、
上記一般式(1)で表される不飽和ポリアルキレングリコールエーテル系単量体(a)由来の構造単位(I)と上記一般式(2)で表される不飽和カルボン酸系単量体(b)由来の構造単位(II)とを含むポリカルボン酸系共重合体の製造方法である。本発明において、不飽和ポリアルキレングリコールエーテル系単量体(a)は1種のみ用いても良いし、2種以上を併用しても良い。本発明において、不飽和カルボン酸系単量体(b)は1種のみ用いても良いし、2種以上を併用しても良い。
本発明のセメント混和剤用共重合体組成物は、上記一般式(1)で表される不飽和ポリアルキレングリコールエーテル系単量体(a)由来の構造単位(I)と上記一般式(2)で表される不飽和カルボン酸系単量体(b)由来の構造単位(II)とを含むポリカルボン酸系共重合体と、分子量300以下の非重合性の有機スルホン酸および/またはその塩とを含む。
R6-(Ph)p-SO3H (3)
(一般式(3)中、R6は水素原子または炭素数1~6のアルキル基を表す。Phはフェニレン基を表す。pは0または1を表す。)
装置:Waters Alliance(2695)
解析ソフト:Waters社製 Empowerプロフェッショナル+GPCオプション
カラム:TSKgel ガードカラム(内径6.0×40mm)+G4000SWXL+G3000SWXL+G2000SWXL(各内径7.8×300mm)
検出器:示差屈折率計(RI)検出器(Waters 2414)、多波長可視紫外(PDA)検出器(Waters 2996)
溶離液:アセトニトリル/酢酸ナトリウム(50mM)イオン交換水溶液=40/60(容積%)の混合溶液に酢酸を加えてpH6.0に調整したもの
流量:1.0ml/分
カラム・検出器温度:40℃
測定時間:45分
試料液注入量:100μl(試料濃度0.5質量%の溶離液溶液)
GPC標準サンプル:東ソー(株)製ポリエチレングリコール Mp=272500、219300、107000、50000、24000、11840、6450、4250、1470の9点を使用
検量線:上記ポリエチレングリコールのMp値を用いて3次式で作成
(1)使用材料
セメント:太平洋セメント
粗骨材:青梅産砕石
細骨材:小笠山産/千葉県君津産山砂
(2)単位量(kg/m3)
W/C=52
s/a=49.0
空気=45.0
水=166.0
セメント=320.0
石=942.0
砂=846
(3)使用ミキサー:太平洋機工、TM55(55リットル強制練パン型ミキサー)、練り量30リットル
(4)試験方法
AE剤としてMA202(ポゾリス物産)をセメントに対し0.0015%配合した。細骨材とセメントをミキサーに投入し、10秒間空練りを行い、次いで、セメント混和剤込みの水、粗骨材を投入し、90秒間混練を行った後、コンクリートを排出した。得られたコンクリートのスランプ値、スランプフロー値、空気量は、日本工業規格(JIS A1101、1128、6204)に準拠して測定した。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水343.1g、3-メチル-3-ブテン-1-オールのエチレンオキシド50モル付加体(ポリエチレングリコールを6.7質量%含む)857.12g、pH調整剤として15%のパラトルエンスルホン酸1水和物水溶液30.89gを仕込み(pH=2.7、21℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水37.9gを投入した。温度が58℃で安定した後、アクリル酸53.3gを水13.3gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.0g、2-メルカプトプロピオン酸1.9gを水161.6gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3以下を維持していた。そして、冷却後(pH=2.8、21.4℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(1)は、共重合体(1P)と非重合性の有機スルホン酸(塩)(1S)を含んでいた。共重合体(1P)のGPCを測定したところ、モノマー(Mw=2289)相当分のピークを除いたポリマー分の質量平均分子量は34000、ポリマー分の含有割合は82.9%であった。結果を表1に示す。
得られた共重合体組成物(1)をセメント混和剤として用い、コンクリート試験を行った。結果を表2に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水339.9g、3-メチル-3-ブテン-1-オールのエチレンオキシド50モル付加体(ポリエチレングリコールを6.7質量%含む)849.9g、pH調整剤として15%のパラトルエンスルホン酸1水和物水溶液30.93gを仕込み(pH=2.7、20℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水41.2gを投入した。温度が58℃で安定した後、アクリル酸60.5gを水15.1gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.1g、2-メルカプトプロピオン酸2.9gを水159.2gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3以下を維持していた。そして、冷却後(pH=2.7、21.6℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(2)は、共重合体(2P)と非重合性の有機スルホン酸(塩)(2S)を含んでいた。共重合体(2P)のGPCを測定したところ、モノマー(Mw=2289)相当分のピークを除いたポリマー分の質量平均分子量は37000、ポリマー分の含有割合は84.3%であった。結果を表1に示す。
得られた共重合体組成物(2)をセメント混和剤として用い、コンクリート試験を行った。結果を表2に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水343.1g、3-メチル-3-ブテン-1-オールのエチレンオキシド50モル付加体(ポリエチレングリコールを6.7質量%含む)829.7g、pH調整剤として15%のパラトルエンスルホン酸1水和物水溶液31.2gを仕込み(pH=2.6、21℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水50.4gを投入した。温度が58℃で安定した後、アクリル酸80.6gを水20.1gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.3g、2-メルカプトプロピオン酸2.7gを水156.9gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3以下を維持していた。そして、冷却後(pH=2.6、26.0℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(3)は、共重合体(3P)と非重合性の有機スルホン酸(塩)(3S)を含んでいた。共重合体(3P)のGPCを測定したところ、モノマー(Mw=2289)相当分のピークを除いたポリマー分の質量平均分子量は36500、ポリマー分の含有割合は88.7%であった。結果を表1に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水339.9g、2-メチル-2-プロペン-1-オールのエチレンオキシド50モル付加体849.9g、pH調整剤として15%のパラトルエンスルホン酸1水和物水溶液30.93gを仕込み(pH=2.4、27℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水41.2gを投入した。温度が58℃で安定した後、アクリル酸60.5gを水15.1gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.1g、2-メルカプトプロピオン酸2.9gを水159.2gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3以下を維持していた。そして、冷却後(pH=2.7、21℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(4)は、共重合体(4P)と非重合性の有機スルホン酸(塩)(4S)を含んでいた。共重合体(4P)のGPCを測定したところ、モノマー(Mw=2274)相当分のピークを除いたポリマー分の質量平均分子量は36500、ポリマー分の含有割合は83.7%であった。結果を表1に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水294.1g、アリルアルコールのエチレンオキシド50モル付加体(ポリエチレングリコールを5.0質量%含む)605.3g、pH調整剤として15%のパラトルエンスルホン酸1水和物水溶液30.0gを仕込み(pH=2.5、27℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水39.0gを投入した。温度が58℃で安定した後、アクリル酸63.3gを水15.8gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.0g、2-メルカプトプロピオン酸2.2gを水156.4gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3以下を維持していた。そして、冷却後(pH=2.6、21.4℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(5)は、共重合体(5P)と非重合性の有機スルホン酸(塩)(5S)を含んでいた。共重合体(5P)のGPCを測定したところ、モノマー(Mw=2260)相当分のピークを除いたポリマー分の質量平均分子量は37500、ポリマー分の含有割合は67.8%であった。結果を表1に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水343.1g、3-メチル-3-ブテン-1-オールのエチレンオキシド50モル付加体(ポリエチレングリコールを6.7質量%含む)857.12g、pH調整剤として7.5%のメタンスルホン酸水溶液30.89gを仕込み(pH=2.5、27℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水37.9gを投入した。温度が58℃で安定した後、アクリル酸53.3gを水13.3gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.0g、2-メルカプトプロピオン酸1.9gを水161.6gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3以下を維持していた。そして、冷却後(pH=2.7、21.4℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(6)は、共重合体(6P)と非重合性の有機スルホン酸(塩)(6S)を含んでいた。共重合体(6P)のGPCを測定したところ、モノマー(Mw=2289)相当分のピークを除いたポリマー分の質量平均分子量は34500、ポリマー分の含有割合は82.5%であった。結果を表1に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水58.7g、2-メチル-2-プロペン-1-オールのエチレンオキシド150モル付加体234.8g、pH調整剤として15%のパラトルエンスルホン酸1水和物水溶液11.0gを仕込み(pH=2.5、27℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水16.8gを投入した。温度が58℃で安定した後、アクリル酸12.7gを水7.2gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸0.9g、2-メルカプトプロピオン酸0.8gを水43.3gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3以下を維持していた。そして、冷却後(pH=2.7、20.5℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(7)は、共重合体(7P)と非重合性の有機スルホン酸(塩)(7S)を含んでいた。共重合体(7P)のGPCを測定したところ、モノマー(Mw=6680)相当分のピークを除いたポリマー分の質量平均分子量は48040、ポリマー分の含有割合は83.9%であった。結果を表1に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水343.1g、3-メチル-3-ブテン-1-オールのエチレンオキシド50モル付加体(ポリエチレングリコールを6.7質量%含む)857.12gを仕込み(pH=7.4、23℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水37.9gを投入した。温度が58℃で安定した後、アクリル酸53.3gを水13.3gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.0g、2-メルカプトプロピオン酸1.9gを水161.6gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3を超えていた。そして、冷却後(pH=5.1、21.4℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(C1)は、共重合体(C1P)を含んでいたが、非重合性の有機スルホン酸(塩)は含んでいなかった。共重合体(C1P)のGPCを測定したところ、モノマー(Mw=2289)相当分のピークを除いたポリマー分の質量平均分子量は34000、ポリマー分の含有割合は74.7%であった。結果を表1に示す。
得られた共重合体組成物(C1)をセメント混和剤として用い、コンクリート試験を行った。結果を表2に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水339.9g、3-メチル-3-ブテン-1-オールのエチレンオキシド50モル付加体(ポリエチレングリコールを6.7質量%含む)849.9gを仕込み(pH=7.2、28℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水41.2gを投入した。温度が58℃で安定した後、アクリル酸60.5gを水15.1gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.1g、2-メルカプトプロピオン酸2.9gを水159.2gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3を超えていた。そして、冷却後(pH=5.0、21.6℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(C2)は、共重合体(C2P)を含んでいたが、非重合性の有機スルホン酸(塩)は含んでいなかった。共重合体(C2P)のGPCを測定したところ、モノマー(Mw=2289)相当分のピークを除いたポリマー分の質量平均分子量は33400、ポリマー分の含有割合は77.2%であった。結果を表1に示す。
得られた共重合体組成物(C2)をセメント混和剤として用い、コンクリート試験を行った。結果を表2に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水343.1g、3-メチル-3-ブテン-1-オールのエチレンオキシド50モル付加体(ポリエチレングリコールを6.7質量%含む)829.7gを仕込み(pH=7.3、23℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水50.4gを投入した。温度が58℃で安定した後、アクリル酸80.6gを水20.1gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.3g、2-メルカプトプロピオン酸2.7gを水156.9gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3を超えていた。そして、冷却後(pH=4.95、26.0℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(C3)は、共重合体(C3P)を含んでいたが、非重合性の有機スルホン酸(塩)は含んでいなかった。共重合体(C3P)のGPCを測定したところ、モノマー(Mw=2289)相当分のピークを除いたポリマー分の質量平均分子量は36000、ポリマー分の含有割合は81.9%であった。結果を表1に示す。
得られた共重合体組成物(C3)をセメント混和剤として用い、コンクリート試験を行った。結果を表2に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水339.9g、2-メチル-2-プロペン-1-オールのエチレンオキシド50モル付加体849.9gを仕込み(pH=7.4、21℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水41.2gを投入した。温度が58℃で安定した後、アクリル酸60.5gを水15.1gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.1g、2-メルカプトプロピオン酸2.9gを水159.2gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3を超えていた。そして、冷却後(pH=4.95、21.6℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(C4)は、共重合体(C4P)を含んでいたが、非重合性の有機スルホン酸(塩)は含んでいなかった。共重合体(C4P)のGPCを測定したところ、モノマー(Mw=2274)相当分のピークを除いたポリマー分の質量平均分子量は36200、ポリマー分の含有割合は76.4%であった。結果を表1に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水313.9g、アリルアルコールのエチレンオキシド50モル付加体(ポリエチレングリコールを5.0質量%含む)608.4g、アクリル酸1.1gを仕込み(pH=5.5、27℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水39.6gを投入した。温度が58℃で安定した後、アクリル酸62.5gを水24.8gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸1.0g、2-メルカプトプロピオン酸2.2gを水146.8gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3を超えていた。そして、冷却後(pH=5.0、20.1℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(C5)は、共重合体(C5P)を含んでいたが、非重合性の有機スルホン酸(塩)は含んでいなかった。共重合体(C5P)のGPCを測定したところ、モノマー(Mw=2260)相当分のピークを除いたポリマー分の質量平均分子量は36000、ポリマー分の含有割合は64.5%であった。結果を表1に示す。
温度計、攪拌機、滴下装置、窒素導入管、還流冷却器を備えたガラス製反応容器に、水58.7g、2-メチル-2-プロペン-1-オールのエチレンオキシド150モル付加体234.8g、アクリル酸0.4gを仕込み(pH=5.5、27℃)、撹拌下に反応容器内を窒素置換し、窒素雰囲気下で58℃に昇温した後、2%過酸化水素水18.0gを投入した。温度が58℃で安定した後、アクリル酸12.3gを水7.2gに溶解させた水溶液を3時間かけて滴下した。アクリル酸水溶液を滴下し始めると同時に、L-アスコルビン酸0.9g、2-メルカプトプロピオン酸0.7gを水43.4gに溶解させた水溶液を3.5時間かけて滴下した。その後、1時間引き続き58℃を維持し、重合反応を完結させた。重合反応中のpHは3を超えていた。そして、冷却後(pH=5.0、21.6℃)、30%NaOH水溶液でpH=6まで中和した。
得られた共重合体組成物(C6)は、共重合体(C6P)を含んでいたが、非重合性の有機スルホン酸(塩)は含んでいなかった。共重合体(C6P)のGPCを測定したところ、モノマー(Mw=6680)相当分のピークを除いたポリマー分の質量平均分子量は50670、ポリマー分の含有割合は80.9%であった。結果を表1に示す。
Claims (9)
- 一般式(1)で表される不飽和ポリアルキレングリコールエーテル系単量体(a)由来の構造単位(I)と一般式(2)で表される不飽和カルボン酸系単量体(b)由来の構造単位(II)とを含むポリカルボン酸系共重合体の製造方法であって、
該単量体(a)と該単量体(b)とを含む単量体成分の重合を、過酸化物と還元剤を重合開始剤として併用して、pH調整剤の存在下で重合中のpHを3以下に制御して行う、
ポリカルボン酸系共重合体の製造方法。
- 前記pH調整剤が、有機スルホン酸および/またはその塩である、請求項1に記載の製造方法。
- 前記過酸化物が過酸化水素であり、前記還元剤がL-アスコルビン酸である、請求項1または2に記載の製造方法。
- 前記共重合体が、セメント混和剤用共重合体である、請求項1から3までのいずれかに記載の製造方法。
- 一般式(1)で表される不飽和ポリアルキレングリコールエーテル系単量体(a)由来の構造単位(I)と一般式(2)で表される不飽和カルボン酸系単量体(b)由来の構造単位(II)とを含むポリカルボン酸系共重合体と、分子量300以下の非重合性の有機スルホン酸および/またはその塩とを含む、セメント混和剤用共重合体組成物。
- 前記有機スルホン酸が、一般式(3)で表される化合物である、請求項5に記載のセメント混和剤用共重合体組成物。
R6-(Ph)p-SO3H (3)
(一般式(3)中、R6は水素原子または炭素数1~6のアルキル基を表す。Phはフェニレン基を表す。pは0または1を表す。) - 前記一般式(3)で表される化合物が分子量250以下の低分子化合物である、請求項6に記載のセメント混和剤用共重合体組成物。
- 前記一般式(3)中のR6が炭素数1または2のアルキル基である、請求項6または7に記載のセメント混和剤用共重合体組成物。
- 前記一般式(1)中のYが炭素数4または5のアルケニル基である、請求項5から8までのいずれかに記載のセメント混和剤用共重合体組成物。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN200980117278.4A CN102027028B (zh) | 2008-05-14 | 2009-05-08 | 制造聚羧酸共聚物的方法和水泥掺合剂用共聚物组合物 |
| US12/992,502 US9079797B2 (en) | 2008-05-14 | 2009-05-08 | Method for producing polycarboxylic acid copolymer and copolymer composition for cement admixture |
| EP09746535.5A EP2277932B1 (en) | 2008-05-14 | 2009-05-08 | Method for producing polycarboxylic acid copolymer and copolymer composition for cement admixture |
| KR1020107027251A KR101707243B1 (ko) | 2008-05-14 | 2009-05-08 | 폴리카르복시산계 공중합체의 제조 방법 및 시멘트 혼화제용 공중합체 조성물 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008-126684 | 2008-05-14 | ||
| JP2008126684 | 2008-05-14 | ||
| JP2008128104 | 2008-05-15 | ||
| JP2008-128104 | 2008-05-15 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009139328A1 true WO2009139328A1 (ja) | 2009-11-19 |
Family
ID=41318698
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/058672 WO2009139328A1 (ja) | 2008-05-14 | 2009-05-08 | ポリカルボン酸系共重合体の製造方法およびセメント混和剤用共重合体組成物 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US9079797B2 (ja) |
| EP (1) | EP2277932B1 (ja) |
| KR (1) | KR101707243B1 (ja) |
| CN (1) | CN102027028B (ja) |
| WO (1) | WO2009139328A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101906193A (zh) * | 2010-08-06 | 2010-12-08 | 天津市飞龙砼外加剂有限公司 | 一种聚羧酸减水剂的合成方法 |
| WO2012043395A1 (ja) * | 2010-09-30 | 2012-04-05 | 株式会社日本触媒 | 不飽和ポリアルキレングリコールエーテル系単量体を含む単量体組成物、該組成物の製造方法、該組成物を用いて得られる重合体および該重合体の製造方法 |
| CN106380551A (zh) * | 2016-09-13 | 2017-02-08 | 科之杰新材料集团有限公司 | 一种低敏感抗泥型聚羧酸减水剂及其制备方法 |
| CN110423317A (zh) * | 2019-09-10 | 2019-11-08 | 成都鑫源祥辰建材有限公司 | 一种聚羧酸减水剂及其制备方法和应用 |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MX2014000469A (es) | 2011-07-12 | 2014-10-17 | Maraxi Inc | Metodos y composiciones para combustibles. |
| US20140220217A1 (en) | 2011-07-12 | 2014-08-07 | Maraxi, Inc. | Method and compositions for consumables |
| US10039306B2 (en) | 2012-03-16 | 2018-08-07 | Impossible Foods Inc. | Methods and compositions for consumables |
| CN102911323B (zh) * | 2012-11-01 | 2016-06-08 | 三门峡市八四八化工厂 | 一种聚羧酸高性能减水剂的低温合成方法 |
| DK2943078T3 (da) | 2013-01-11 | 2021-06-14 | Impossible Foods Inc | Fremgangsmåder og sammensætninger til føde- og drikkevarer |
| BR112015016651A2 (pt) | 2013-01-11 | 2017-07-11 | Impossible Foods Inc | réplica de queijo não láctea compreendendo um coacervado |
| FR3012808B1 (fr) * | 2013-11-07 | 2017-09-01 | Kerneos | Composition fluidifiante sous forme de poudre et son procede de preparation |
| MX2016012817A (es) | 2014-03-31 | 2017-01-13 | Impossible Foods Inc | Replicas de carne molida. |
| CN103926902B (zh) * | 2014-04-24 | 2016-08-24 | 山东国元新材料有限公司 | 用于水泥及商砼外加剂生产的多功能自动化控制装备 |
| KR101717176B1 (ko) * | 2014-11-11 | 2017-03-16 | 주식회사 엘지화학 | 폴리카르본산계 공중합체를 포함하는 시멘트 조성물 첨가제 및 이의 제조방법 |
| CN107245130B (zh) * | 2017-06-08 | 2018-06-26 | 武汉理工大学 | 一种银杏酸接枝改性的聚羧酸减水剂及其制备方法 |
| US11168402B2 (en) | 2018-02-16 | 2021-11-09 | Nippon Shokubai Co., Ltd. | Metal corrosion inhibitor |
| CN109021180B (zh) * | 2018-08-16 | 2020-12-29 | 科之杰新材料集团有限公司 | 一种低敏感减缩型聚羧酸减水剂及其制备方法 |
| CN109102919B (zh) * | 2018-08-24 | 2020-12-01 | 山东圣泉新材料股份有限公司 | 一种复合导电膜及其制备方法和应用 |
| CN111171247B (zh) * | 2020-01-09 | 2022-05-27 | 詹烨 | 一种混凝土聚羧酸保坍剂的制备方法 |
| CN115215566A (zh) * | 2021-09-28 | 2022-10-21 | 鲁昌宝 | 一种液体水泥金属离子去除剂及其制备方法 |
Citations (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5918338B2 (ja) | 1981-10-30 | 1984-04-26 | 株式会社日本触媒 | セメント分散剤 |
| JPH0753645A (ja) | 1993-08-18 | 1995-02-28 | Nippon Shokubai Co Ltd | 水溶性グラフト重合体の製造方法 |
| JPH07223852A (ja) | 1993-12-14 | 1995-08-22 | Kao Corp | コンクリート混和剤 |
| JPH08208770A (ja) | 1995-02-07 | 1996-08-13 | Nippon Shokubai Co Ltd | 水溶性グラフト重合体、その製造方法及び用途 |
| JPH08208769A (ja) | 1995-02-03 | 1996-08-13 | Nippon Shokubai Co Ltd | 水溶性グラフト重合体、その製法および用途 |
| WO2001014438A1 (fr) | 1999-08-23 | 2001-03-01 | Kao Corporation | Procede de production de polymeres (meth)acryliques |
| JP2001220417A (ja) | 1999-11-29 | 2001-08-14 | Nippon Shokubai Co Ltd | セメント混和剤用共重合体、その製造方法およびその用途 |
| JP2002121055A (ja) | 2000-08-11 | 2002-04-23 | Nippon Shokubai Co Ltd | セメント分散剤およびこれを用いたセメント組成物 |
| WO2003040194A1 (fr) | 2001-11-06 | 2003-05-15 | Nippon Shokubai Co., Ltd. | Polymere d'acide (meth)acrylique (sel) et procede de production associe |
| JP2003221266A (ja) * | 2002-01-30 | 2003-08-05 | Nippon Shokubai Co Ltd | セメント混和剤 |
| JP2004067934A (ja) * | 2002-08-08 | 2004-03-04 | Nippon Shokubai Co Ltd | セメント添加剤用共重合体 |
| JP2006248889A (ja) | 2005-02-14 | 2006-09-21 | Nippon Shokubai Co Ltd | セメント混和剤およびその製造方法 |
| WO2006129883A1 (en) | 2005-06-01 | 2006-12-07 | Nippon Shokubai Co., Ltd. | Powdery cement dispersant |
| JP2007119337A (ja) | 2005-09-28 | 2007-05-17 | Nippon Shokubai Co Ltd | セメント混和剤及びセメント組成物 |
| JP2007327067A (ja) | 1996-12-20 | 2007-12-20 | Basf Ag | カルボキシル基及びポリアルキレンオキシドエーテル−側鎖を含有するポリマー |
| JP2008274258A (ja) * | 2007-04-05 | 2008-11-13 | Nippon Shokubai Co Ltd | ポリカルボン酸系共重合体の製造方法 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5362323A (en) * | 1992-02-14 | 1994-11-08 | W. R. Grace & Co. Conn. | Cement admixture composition |
| US6166112A (en) * | 1997-03-10 | 2000-12-26 | Nippon Shokubai Co., Ltd. | Cement admixture and cement composition |
| US20020052232A1 (en) * | 2000-06-28 | 2002-05-02 | Kaminkow James E. | Apparatus and method for modifying generated values to determine an award in a gaming device |
| JP4947856B2 (ja) | 2000-08-11 | 2012-06-06 | 株式会社日本触媒 | セメント分散剤およびこれを用いたセメント組成物 |
| SG101990A1 (en) * | 2000-08-11 | 2004-02-27 | Nippon Catalytic Chem Ind | Cement dispersant and cement composition comprising this |
| WO2004089845A1 (en) | 2003-04-08 | 2004-10-21 | Nippon Shokubai Co., Ltd. | Cement admixture and cement composition |
| JP2007302519A (ja) | 2006-05-12 | 2007-11-22 | Nippon Shokubai Co Ltd | セメント分散剤 |
| EP2152771B2 (en) | 2007-04-05 | 2021-03-10 | Nippon Shokubai Co., Ltd. | Production method of unsaturated (poly)alkylene glycol ether monomer and production method of (poly)alkylene glycol chain-containing polymer |
-
2009
- 2009-05-08 WO PCT/JP2009/058672 patent/WO2009139328A1/ja active Application Filing
- 2009-05-08 KR KR1020107027251A patent/KR101707243B1/ko active IP Right Grant
- 2009-05-08 CN CN200980117278.4A patent/CN102027028B/zh active Active
- 2009-05-08 US US12/992,502 patent/US9079797B2/en active Active
- 2009-05-08 EP EP09746535.5A patent/EP2277932B1/en active Active
Patent Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5918338B2 (ja) | 1981-10-30 | 1984-04-26 | 株式会社日本触媒 | セメント分散剤 |
| JPH0753645A (ja) | 1993-08-18 | 1995-02-28 | Nippon Shokubai Co Ltd | 水溶性グラフト重合体の製造方法 |
| JPH07223852A (ja) | 1993-12-14 | 1995-08-22 | Kao Corp | コンクリート混和剤 |
| JPH08208769A (ja) | 1995-02-03 | 1996-08-13 | Nippon Shokubai Co Ltd | 水溶性グラフト重合体、その製法および用途 |
| JPH08208770A (ja) | 1995-02-07 | 1996-08-13 | Nippon Shokubai Co Ltd | 水溶性グラフト重合体、その製造方法及び用途 |
| JP2007327067A (ja) | 1996-12-20 | 2007-12-20 | Basf Ag | カルボキシル基及びポリアルキレンオキシドエーテル−側鎖を含有するポリマー |
| WO2001014438A1 (fr) | 1999-08-23 | 2001-03-01 | Kao Corporation | Procede de production de polymeres (meth)acryliques |
| JP3683176B2 (ja) | 1999-11-29 | 2005-08-17 | 株式会社日本触媒 | セメント混和剤およびセメント組成物 |
| JP2001220417A (ja) | 1999-11-29 | 2001-08-14 | Nippon Shokubai Co Ltd | セメント混和剤用共重合体、その製造方法およびその用途 |
| JP2002121055A (ja) | 2000-08-11 | 2002-04-23 | Nippon Shokubai Co Ltd | セメント分散剤およびこれを用いたセメント組成物 |
| WO2003040194A1 (fr) | 2001-11-06 | 2003-05-15 | Nippon Shokubai Co., Ltd. | Polymere d'acide (meth)acrylique (sel) et procede de production associe |
| JP2003221266A (ja) * | 2002-01-30 | 2003-08-05 | Nippon Shokubai Co Ltd | セメント混和剤 |
| JP2004067934A (ja) * | 2002-08-08 | 2004-03-04 | Nippon Shokubai Co Ltd | セメント添加剤用共重合体 |
| JP2006248889A (ja) | 2005-02-14 | 2006-09-21 | Nippon Shokubai Co Ltd | セメント混和剤およびその製造方法 |
| WO2006129883A1 (en) | 2005-06-01 | 2006-12-07 | Nippon Shokubai Co., Ltd. | Powdery cement dispersant |
| JP2007119337A (ja) | 2005-09-28 | 2007-05-17 | Nippon Shokubai Co Ltd | セメント混和剤及びセメント組成物 |
| JP2008274258A (ja) * | 2007-04-05 | 2008-11-13 | Nippon Shokubai Co Ltd | ポリカルボン酸系共重合体の製造方法 |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101906193A (zh) * | 2010-08-06 | 2010-12-08 | 天津市飞龙砼外加剂有限公司 | 一种聚羧酸减水剂的合成方法 |
| WO2012043395A1 (ja) * | 2010-09-30 | 2012-04-05 | 株式会社日本触媒 | 不飽和ポリアルキレングリコールエーテル系単量体を含む単量体組成物、該組成物の製造方法、該組成物を用いて得られる重合体および該重合体の製造方法 |
| US20130184420A1 (en) * | 2010-09-30 | 2013-07-18 | Nippon Shokubai Co., Ltd. | Monomer composition containing unsaturated polyalkylene glycol ether-based monomer, method for producing composition thereof, polymer obtained using composition thereof, and method for producing polymer thereof |
| US9850378B2 (en) | 2010-09-30 | 2017-12-26 | Nippon Shokubai Co., Ltd. | Monomer composition containing unsaturated polyalkylene glycol ether-based monomer, method for producing composition thereof, polymer obtained using composition thereof, and method for producing polymer thereof |
| US10208203B2 (en) | 2010-09-30 | 2019-02-19 | Nippon Shokubai Co., Ltd. | Monomer composition containing unsaturated polyalkylene glycol ether-based monomer, method for producing composition thereof, polymer obtained using composition thereof, and method for producing polymer thereof |
| CN106380551A (zh) * | 2016-09-13 | 2017-02-08 | 科之杰新材料集团有限公司 | 一种低敏感抗泥型聚羧酸减水剂及其制备方法 |
| CN110423317A (zh) * | 2019-09-10 | 2019-11-08 | 成都鑫源祥辰建材有限公司 | 一种聚羧酸减水剂及其制备方法和应用 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20110065847A1 (en) | 2011-03-17 |
| EP2277932A1 (en) | 2011-01-26 |
| CN102027028B (zh) | 2014-11-26 |
| EP2277932A4 (en) | 2013-06-05 |
| CN102027028A (zh) | 2011-04-20 |
| EP2277932B1 (en) | 2018-01-10 |
| KR20110016911A (ko) | 2011-02-18 |
| US9079797B2 (en) | 2015-07-14 |
| KR101707243B1 (ko) | 2017-02-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2009139328A1 (ja) | ポリカルボン酸系共重合体の製造方法およびセメント混和剤用共重合体組成物 | |
| JP4233294B2 (ja) | セメント混和剤、セメント組成物及びセメント混和剤の製造方法 | |
| JP2004519406A (ja) | セメント混和剤及びセメント組成物 | |
| JP6188412B2 (ja) | 共重合体とその用途 | |
| EP2465836B1 (en) | Polymer composition for cement dispersant and method for producing same | |
| JP6012139B2 (ja) | セメント混和剤及びセメント組成物 | |
| JP2017186232A (ja) | 水硬性材料用減水剤の製造方法 | |
| JP5909359B2 (ja) | セメント混和剤用共重合体、その製造方法および当該共重合体を含むセメント混和剤 | |
| JP5306040B2 (ja) | セメント混和剤用共重合体組成物 | |
| JP5584638B2 (ja) | セメント混和剤およびセメント組成物 | |
| JP5137368B2 (ja) | セメント混和剤及びセメント組成物 | |
| JP5707165B2 (ja) | セメント混和剤及びこれを含むセメント組成物 | |
| JP2002121056A (ja) | セメント分散剤およびこれを用いたセメント組成物 | |
| JP6249630B2 (ja) | 共重合体とその用途 | |
| JP5561953B2 (ja) | ポリカルボン酸系共重合体の製造方法 | |
| JP5912964B2 (ja) | セメント混和剤用ポリカルボン酸系共重合体 | |
| JP6747962B2 (ja) | セメント用添加剤およびセメント組成物 | |
| JP2016145126A (ja) | セメント混和剤およびこれを含有するセメント組成物 | |
| JP6109240B2 (ja) | セメント混和剤及びセメント組成物 | |
| JP2014189756A (ja) | セメント混和剤用ポリカルボン酸系共重合体 | |
| JP4822613B2 (ja) | セメント混和剤及びセメント組成物 | |
| JP2014031296A (ja) | セメント混和剤用ポリカルボン酸系共重合体 | |
| JP5422105B2 (ja) | コンクリート混和剤用ポリカルボン酸系共重合体及び共重合体組成物 | |
| JP2017122026A (ja) | 水硬性組成物用分散剤の製造方法 | |
| JP5649610B2 (ja) | セメント混和剤及びセメント組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980117278.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09746535 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009746535 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12992502 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20107027251 Country of ref document: KR Kind code of ref document: A |