WO2006090920A1 - ペルオキシソーム増殖剤活性化受容体δの活性化剤 - Google Patents
ペルオキシソーム増殖剤活性化受容体δの活性化剤 Download PDFInfo
- Publication number
- WO2006090920A1 WO2006090920A1 PCT/JP2006/304193 JP2006304193W WO2006090920A1 WO 2006090920 A1 WO2006090920 A1 WO 2006090920A1 JP 2006304193 W JP2006304193 W JP 2006304193W WO 2006090920 A1 WO2006090920 A1 WO 2006090920A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- carbon atoms
- group
- alkyl group
- compound
- salt
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/32—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/325—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms with substituted hydrocarbon radicals directly attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/32—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/33—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms with substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/333—Radicals substituted by oxygen or sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/32—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/33—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms with substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/337—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/14—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D231/18—One oxygen or sulfur atom
- C07D231/20—One oxygen atom attached in position 3 or 5
- C07D231/22—One oxygen atom attached in position 3 or 5 with aryl radicals attached to ring nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/34—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D307/38—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D307/40—Radicals substituted by oxygen atoms
- C07D307/42—Singly bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/34—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D307/38—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D307/40—Radicals substituted by oxygen atoms
- C07D307/46—Doubly bound oxygen atoms, or two oxygen atoms singly bound to the same carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/14—Radicals substituted by singly bound hetero atoms other than halogen
- C07D333/16—Radicals substituted by singly bound hetero atoms other than halogen by oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/22—Radicals substituted by doubly bound hetero atoms, or by two hetero atoms other than halogen singly bound to the same carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/24—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/04—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
Definitions
- the present invention relates to an activator of peroxisome proliferator activated receptor (PPAR) ⁇ .
- PPAR peroxisome proliferator activated receptor
- Peroxisome proliferator-activated receptor pe r o x i s ome p ro l i f e r a t o r r a c t i v a t e d r e c e p t to r: P PAR) f
- Patent Document 1 describes the use of L 1 1 6504 1 (Merck) as an anti-obesity agent for diabetes
- Patent Document 2 describes that YM-1 6 6 3 8 (Yamayuki) has a serum cholesterol lowering action and an LD L-cholesterol lowering action
- Patent Document 2 describes the use of a biaryl derivative as a drug that increases blood HD L
- Patent Document 4 GW-5 907 3 5, GSK
- WO 2004/6 3 1 6 6 Patent Document 5: Pyrazole Derivative, Lily
- Patent Document 6 Thiophene Derivative, GS K)
- Patent Document 8
- GW- 5 0 1 5 1 6 represented by is currently being developed as a lipid metabolism improving agent.
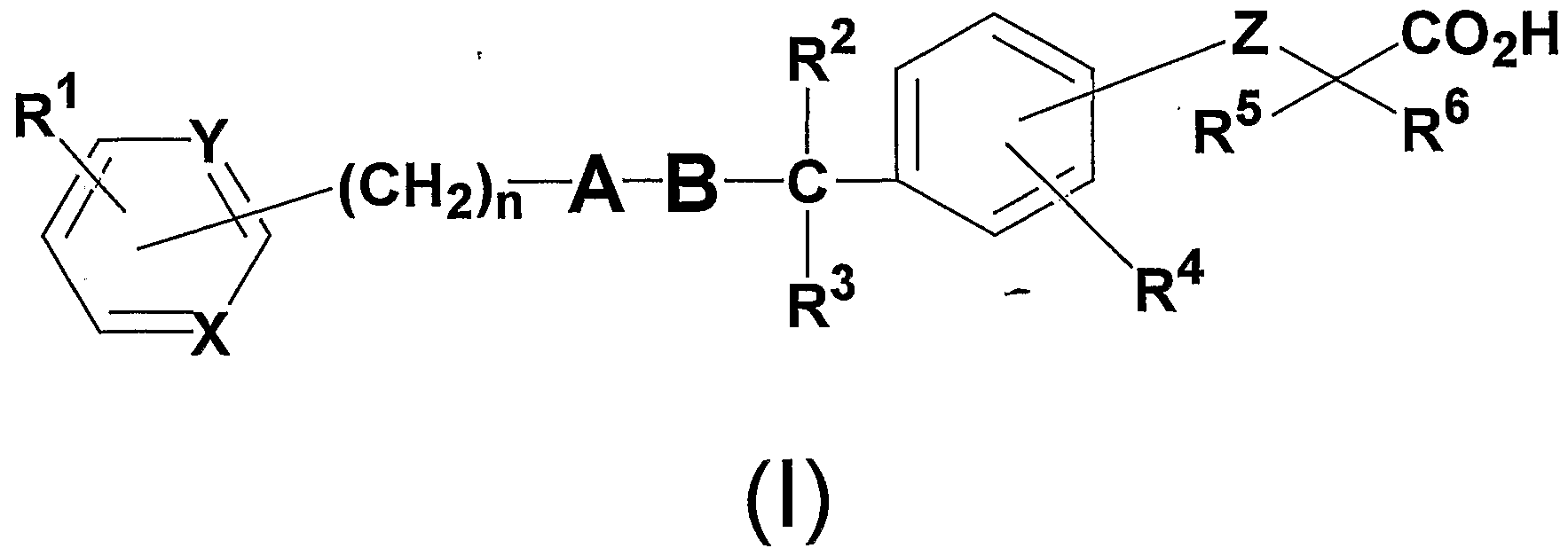
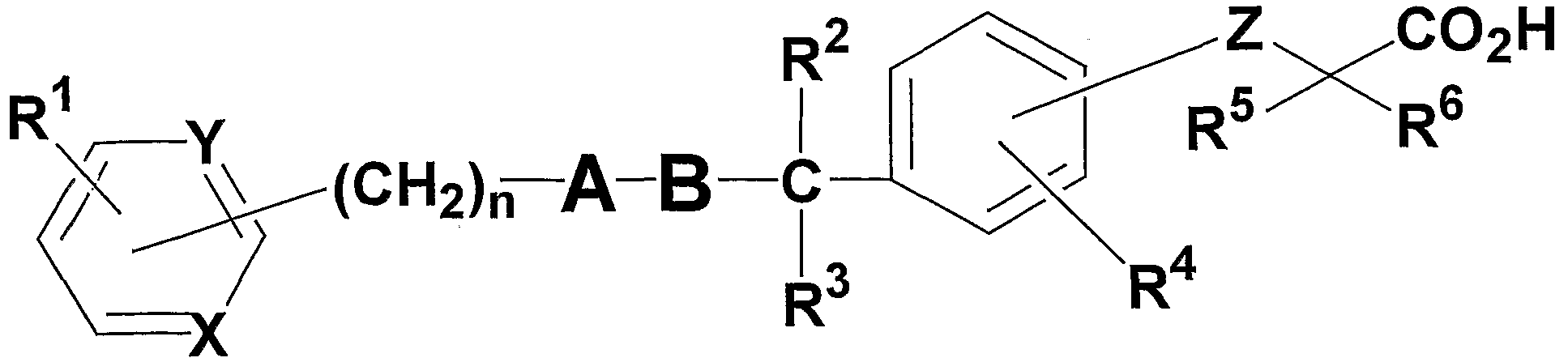
- An object of the present invention is to provide a compound represented by the following general formulas (I), (II) and ( ⁇ ) having an activating action on a peroxisome proliferator-activated receptor.
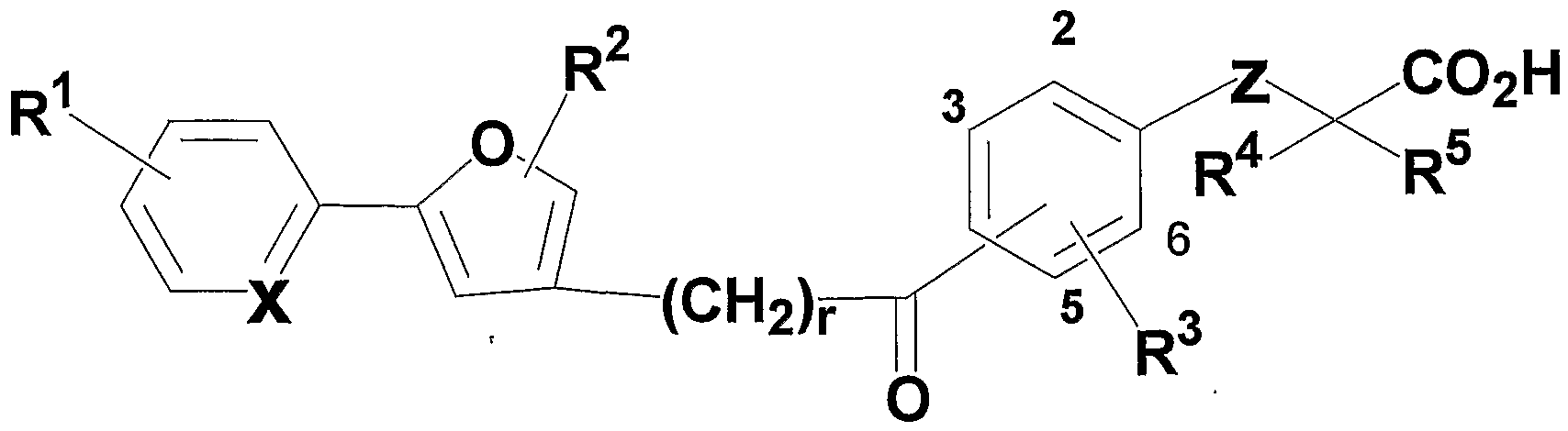
- R 1 and R 4 may be the same or different, a hydrogen atom, carbon number 1 to 8
- R 2 represents a hydrogen atom
- R 3 represents an alkyl group having 1 to 8 carbon atoms
- R 5 and R 6 may be the same or different and each represents a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom,
- X and Y may be the same or different and represent C H or N;
- Z represents an oxygen atom or a sulfur atom
- A is an alkyl group having 1 to 8 carbon atoms, a 3- to 7-membered ring alkyl group, an alkenyl group having 2 to 8 carbon atoms, an alkyl group having 2 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms.
- 1 to 8 carbon atoms substituted with a 3- to 7-membered alkyl group 1 to 8 carbon atoms substituted with a halogen atom, carbon substituted with a halogen atom
- An alkoxy group having 1 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, a 5- or 6-membered heterocyclic group, and an aralkyl group (the carbon number of the aryl moiety is 6 to 10 carbon atoms in the alkylene moiety) The number is selected from pyrazole, thiophene, furan or pyrrole, which may have 1 to 8) or an alkyl group having 1 to 8 carbon atoms substituted with a 5- or 6-membered heterocyclic group. Represents a hetero five-membered ring,
- B is an alkyl group having 1 to 8 carbon atoms, a 3 to 7-membered cycloalkyl group, an alkenyl group having 2 to 8 carbon atoms, an alkynyl group having 2 to 8 carbon atoms, or an alkoxy group having 1 to 8 carbon atoms.
- a halogen atom, an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom, or an alkoxy group having 1 to 8 carbon atoms substituted with a halogen atom may have 1 to 6 carbon atoms Represents an alkylene chain (in the case of 2 to 6 carbon atoms, it may have a double bond), N represents an integer of 0 to 5. )
- the present invention relates to the following compounds or salts thereof:
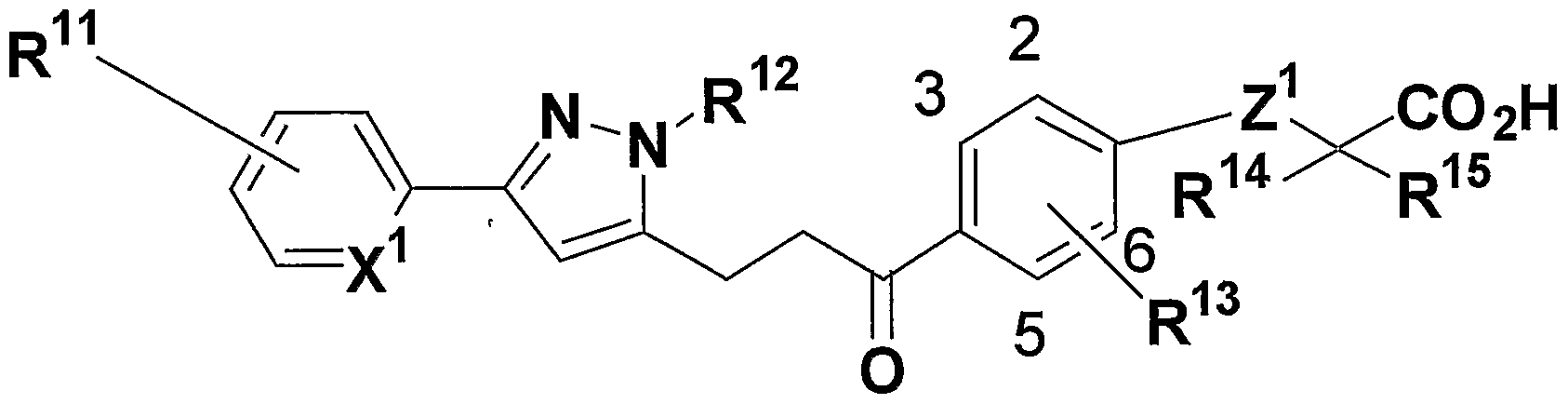
- R 1 1 and R 13 may be the same or different, and are a hydrogen atom, an alkynole group having 1 to 8 carbon atoms, an alkyl group having 2 to 8 carbon atoms, or an alkyl having 2 to 8 carbon atoms.
- R 12 is a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, a cycloalkyl group having 3 to 7 members, an alkenyl group having 2 to 8 carbon atoms, an alkynyl group having 2 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms.
- An alkoxy group an alkyl group having 1 to 8 carbon atoms substituted with a 3- to 7-membered cycloalkyl group, an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom, or a carbon atom substituted with a halogen atom 1 ⁇ 8 alkoxy group, C6-C10 aryl group, 5- or 6-membered heterocyclic group, aralkyl group (Aryl moiety has 6-10 carbon atoms, carbon of alkylene part The number represents 1-8) or a C1-C8 alkyl group substituted with a 5- or 6-membered heterocyclic group,
- R 14 and R 15 may be the same or different and each represents a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom,
- X 1 represents CH or N
- Z 1 represents an oxygen atom or a sulfur atom
- W 1 represents an oxygen atom or CH 2 ,
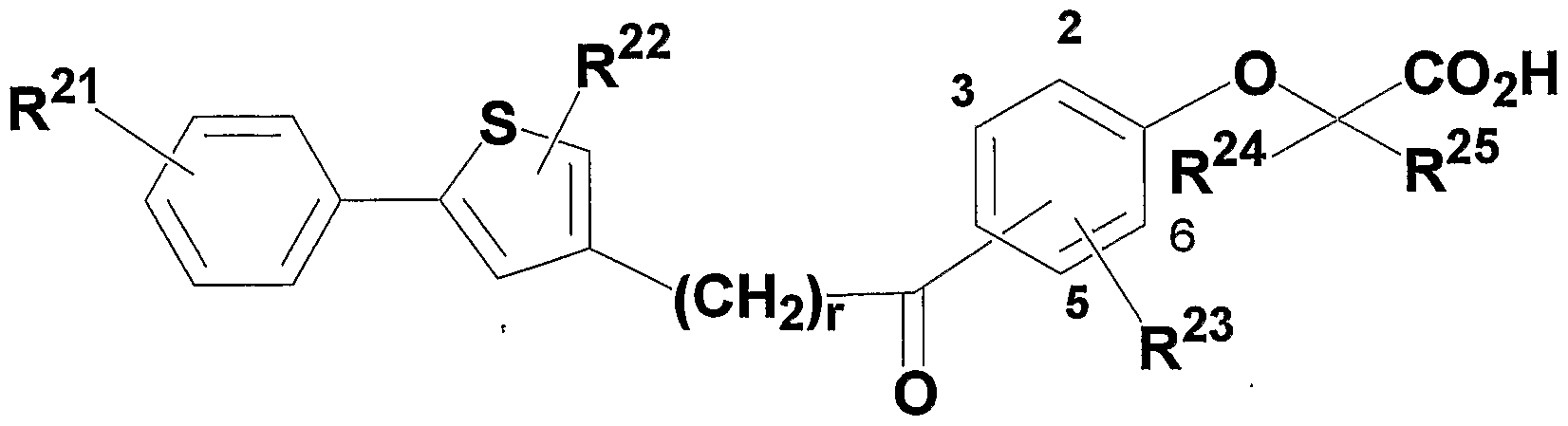
- R 2 1 and R 2 3 may be the same or different, a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, an alkynyl group having 2 to 8 carbon atoms, C1-C8 alkoxy group, halogen atom, C1-C8 alkyl group substituted with halogen atom, C1-C8 alkoxy group substituted with halogen atom, hydroxyl group, nitrogen group, carbon number Represents a 2 to 8 acyl group, a 6 to 10 carbon aryl group, or a 5- or 6-membered heterocyclic group,
- R 2 2 is a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, a cycloalkyl group having 3 to 7 members, an alkenyl group having 2 to 8 carbon atoms, an alkynyl group having 2 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms.
- An alkoxy group an alkyl group having 1 to 8 carbon atoms substituted with a 3- to 7-membered cycloalkyl group, an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom, or a carbon atom substituted with a halogen atom 1 ⁇ 8 alkoxy group, 6-.10 carbon aryl group, 5- or 6-membered heterocyclic group, aralkyl group (the aryl group has 6-10 carbon atoms, the carbon of the alkylene part The number represents 1 to 8) or an alkyl group having 1 to 8 carbon atoms substituted with a 5- or 6-membered heterocyclic group,
- R 24 and R 25 may be the same or different and each represents a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom,
- X 2 represents CH or N; 2 2 represents an oxygen atom or a sulfur atom,
- W 2 represents an oxygen atom or CH 2 ,
- the present invention relates to an activator of peroxisome proliferator-activated receptor ⁇ containing the compound represented by the above general formula (I), (II) or (III) or a salt thereof as an active ingredient.
- the alkyl group having 1 to 8 carbon atoms of the substituent that the alkylene chain of 2 to 6 may have include a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, an i-butyl group, a t-group. Examples include a methyl group, a pentyl group, and a hexyl group.
- R 1 and R 4 and the hetero ring of A which may have a 5-membered ring, and the substituent which the alkylene chain of B 2 to 6 carbon atoms may have 2 to 8 carbon atoms
- Examples of the alkenyl group include a bur group and an aryl group.
- R 1 and R 4 and the hetero ring of A which may have a 5-membered ring, and the substituent which the alkylene chain of B 2 to 6 carbon atoms may have 2 to 8 carbon atoms
- Examples of the alkynyl group include a propargyl group.
- R 1 and R 4 and the hetero ring of A which may have a 5-membered ring, and the carbon number of the substituent which the alkylene chain of 2 to 6 carbon atoms may have 1 to 8
- the alkoxy group include a methoxy group, an ethoxy group, a propoxy group, an isopropoxy group, a butoxy group, an i-butoxy group, a t-butoxy group, a pentyloxy group, and a hexyloxy group.
- Examples of the halogen atom of the substituent that the alkylene chain of 2 to 6 carbon atoms of R 1 , R 4 and B may have include a fluorine atom, a chlorine atom, or a bromine atom.
- Examples of the alkyl group having 1 to 8 carbon atoms substituted with a halogen atom of 1 to 3 include a methyl group, an ethyl group, and a propyl group substituted with a halogen atom such as 1 to 3 fluorine atoms, chlorine atoms, or bromine atoms.
- R 1 , R 4 , and a hetero ring of A which may have a 5-membered ring and a halogen atom of a substituent which may have an alkylene chain of 2 to 6 carbon atoms of B
- the alkoxy group having 1 to 8 carbon atoms include a methoxy group, an ethoxy group, a propoxy group, an isopropyloxy group, a ptyloxy group substituted with 1 to 3 fluorine atoms, a chlorine atom or a halogen atom such as a fluorine atom, or t-Butyloxy group, etc., preferably trifluoromethyloxy group, chloromethyloxy group, 2-chloroethyloxy group, 2-promoethyloxy group or 2-fluoroethyloxy group And the like.
- Examples of the acyl group having 2 to 8 carbon atoms of R 1 and R 4 include a acetyl group or a propionyl group.
- R 1 and R 4 as well as A hetero carbon group of the substituent which the 5-membered ring may have 6 to 1
- Examples of the 0 aryl group include a phenyl group.
- Examples of the 5- or 6-membered heterocyclic group of the substituent which the hetero ring of R 1 and R 4 and A may have include a pyridyl group.
- alkyl group having 1 to 8 carbon atoms that is substituted with the 3- to 7-membered ring alkyl group of the substituent which the 5-membered ring of A may have, a cyclopropyl group, cyclopentyl And a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, an i-butyl group, a t-butyl group, a pentyl group, or a hexyl group substituted with a group or a cyclohexyl group.
- a heteroalkyl group of A which may have a 5-membered ring is a aralkyl group (where the carbon number of the aryl moiety is 6 to 10 and the carbon number of the alkylene part is 1 to 8). Groups and the like.
- the alkyl group having 1 to 8 carbon atoms substituted by the 5- or 6-membered heterocyclic group which may be substituted on the 5-membered ring includes methyl substituted with a pyridyl group or the like.
- the alkoxy group, the acyl group having 2 to 8 carbon atoms, the aryl group having 6 to 10 carbon atoms, and the 5- or 6-membered heterocyclic group include R 1 or R 4 in the general formula (I). The same as those exemplified in the above.
- the alkyl group having 1 to 8 carbon atoms substituted with a heterocyclic group of the ring are
- R 1 in the general formula (I), for the R 2 1 in the general formula (II) R 1 1 and the general formula in (II I) in, on the benzene ring or the like R 1 or the like is substituted There may be 1 to 3 of the same or different.
- R 4 in the above general formula (I), R 13 in general formula (II) and R 2 3 in general formula (III) are the same as the benzene ring substituted by R 4 etc. Or 1 to 3 different things may be present.
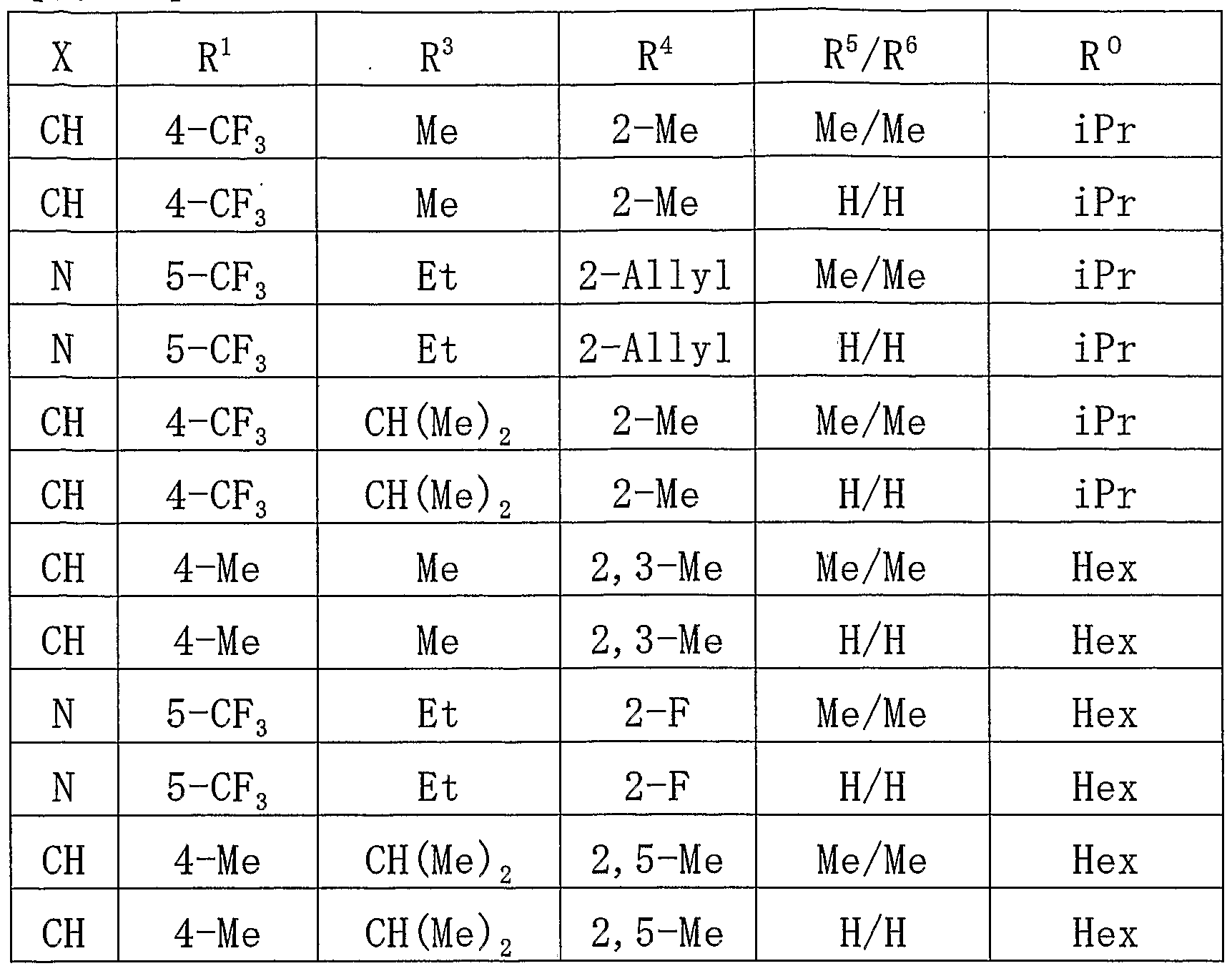
- the compound of the present invention is preferably a compound represented by the above general formula (I) wherein A is pyrazole or a salt thereof.
- the compound of the present invention is preferably a compound in which — (CH 2 ) n is bonded at the 1-position of pyrazole in the above (1) or a salt thereof.
- the compound of the present invention is preferably a compound in which 1 (CH 2 ) n — is bonded at the 3-position of pyrazole or a salt thereof in the above (1).
- the compound of the present invention is preferably a compound or a salt thereof in which (B) is bonded at the 4 or 5 position of pyrazole in the above (2) or (3).
- the compound of the present invention is preferably a compound represented by the above general formula (I), wherein A is thiophene, furan or pyrrole, or a salt thereof.
- the compound of the present invention is preferably a compound or a salt thereof in which (1) (CH 2 ) n _ is bonded at the 2-position of the hetero 5-membered ring.
- the compound of the present invention is preferably a compound represented by the above general formula (I) wherein A is thiophene or a salt thereof.
- the compound of the present invention is preferably a compound represented by the above general formula (I) or a compound represented by the above (1) to (8), wherein n is 0 or a salt thereof.
- the compound of the present invention is preferably a compound represented by the above general formula (I) or a compound of the above (1) to (9), wherein X and Y are both CH or a salt thereof. .
- the compound or salt thereof is preferred.
- the compound of the present invention is a compound represented by the above general formula (I) or a compound of the above (1) to (1 1), wherein B is an alkyl group having 1 to 8 carbon atoms or halogen as a substituent.
- B is an alkyl group having 1 to 8 carbon atoms or halogen as a substituent.
- a compound or a salt thereof which is an alkylene chain having 2 to 4 carbon atoms which may have an alkyl group having 1 to 8 carbon atoms substituted with an atom is preferable.
- the compound of the present invention is preferably a compound represented by the above general formula (I) or a compound of the above (1) to (12), wherein B is an ethylene chain, or a salt thereof.
- the compound of the present invention is a compound represented by the above general formula (I) or a compound of the above (1) to (13), wherein R 1 and R 4 may be the same or different, and hydrogen An atom, an alkyl group having 1 to 8 carbon atoms, an alkoxy group having 1 to 8 carbon atoms, a halogen atom, an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom, or a carbon atom having 1 to 8 carbon atoms substituted with a halogen atom A compound having 8 alkoxy groups or a salt thereof is preferred.
- the compound of the present invention is a compound represented by the above general formula (I) or a compound of the above (1) to (14), wherein R 5 and R 6 may be the same or different.
- a compound or a salt thereof is preferred with an atom or an alkyl group having 1 to 8 carbon atoms.
- the compound of the present invention is a compound represented by the above general formula (I) or a compound of the above (1) to (15), which may be substituted on the hetero 5-membered ring of A
- a compound or a salt thereof in which the group is an alkyl group having 1 to 8 carbon atoms or an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom is preferable.
- the compound of the present invention is preferably a compound represented by the above general formula (II) wherein X 1 is CH or a salt thereof.
- the compound of the present invention is a compound represented by the above general formula (II), wherein R 1 1 monophenyl or R 1 1 monopyridyl is bonded at the 1-position of pyrazole or a salt thereof Is preferred.
- the compound of the present invention is a compound represented by the above general formula (II), wherein R 1 1 monophenyl or R 1 1 monopyridyl is bonded at the 3-position of pyrazole or a salt thereof. Is preferred.
- the compound of the present invention is a compound represented by the above general formula (II) or the compounds of the above (17) to (2 1), wherein R 11 and R 13 are the same or different. Also well, a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, an alkoxy group having 1 to 8 carbon atoms, a halogen atom, a carbon atom having 1 to 8 carbon atoms substituted by a halogen atom, or a carbon atom substituted by a halogen atom A compound having 1 to 8 alkoxy groups or a salt thereof is preferred. '.
- the compound of the present invention includes the compound represented by the above general formula (II) or the compounds of the above (17) to (2 1), wherein R 11 and R 13 are the same or A compound or a salt thereof, which may be different, is an alkyl group having 1 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom.
- the compound of the present invention is a compound represented by the above general formula (II) or the compounds of the above (17) to (23), wherein R 14 and R 15 are the same or different.
- a compound having a hydrogen atom or an alkyl group having 1 to 8 carbon atoms or a salt thereof is preferred.
- a compound or a salt thereof is preferably an alkoxy group having 1 to 8 carbon atoms, an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom, or an alkoxy group having 1 to 8 carbon atoms substituted with a halogen atom.
- the compound of the present invention is preferably a compound represented by the above general formula (III) wherein X 2 is CH or a salt thereof.
- the compound of the present invention is a compound represented by the above general formula (III), wherein R 2 1 monophenyl or R 2 1 monopyridyl is bonded at the 2-position of thiophene or The salt is preferred.
- the compound of the present invention includes the compound represented by the above general formula (III) or the above A compound represented by the formula (2 8) or (2 9), wherein W 2 is an oxygen atom, or a salt thereof is preferred.
- R 2 1 ⁇ Pi R 2 3 are the same or different Substituted with a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, an alkoxy group having 1 to 8 carbon atoms, a halogen atom, an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom, or a halogen atom.
- a compound having a C 1-8 alkoxy group or a salt thereof is preferred.
- R 2 1 ⁇ Pi R 2 3 is may be the same or different, A compound or a salt thereof which is an alkyl group having 1 to 8 carbon atoms, or an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom is preferable.
- R 2 4 and 2 15 may be the same or different in the compound represented by the above general formula (III) or the above (2 8) to (3 2)
- a compound which is a hydrogen atom or an alkyl group having 1 to 8 carbon atoms or a salt thereof is preferable.
- R 2 2 is a hydrogen atom, an alkyl group of 1-8 carbon atoms, carbon atoms
- a compound or a salt thereof which is an alkoxy group having 1 to 8 carbon atoms, an alkyl group having 1 to 8 carbon atoms substituted with a halogen atom, or an alkoxy group having 1 to 8 carbon atoms substituted with a halogen atom is preferable.
- the compound of the present invention is a compound represented by the above general formula (III) or the above (28) to (3 3), wherein R 2 2 is an alkyl group having 1 to 8 carbon atoms or a halogen atom.
- R 2 2 is an alkyl group having 1 to 8 carbon atoms or a halogen atom.
- a compound or a salt thereof which is a substituted alkyl group having 1 to 8 carbon atoms is preferable.
- the compound of the present invention is preferably a compound represented by the above general formula (III), a compound represented by the above (28) to (35), wherein r is 2, or a salt thereof.
- the compounds represented by the above general formulas (I), (II) and (III) may be pharmacologically acceptable salts.
- alkali metal salts such as sodium, potassium and lithium may be used. Can be mentioned.
- the compounds of the present invention include optically active isomers, racemic and other optical isomers, cis and trans Geometric isomers and the like may exist, but both are included in the present invention.
- optically active isomers racemic and other optical isomers
- cis and trans Geometric isomers and the like may exist, but both are included in the present invention.
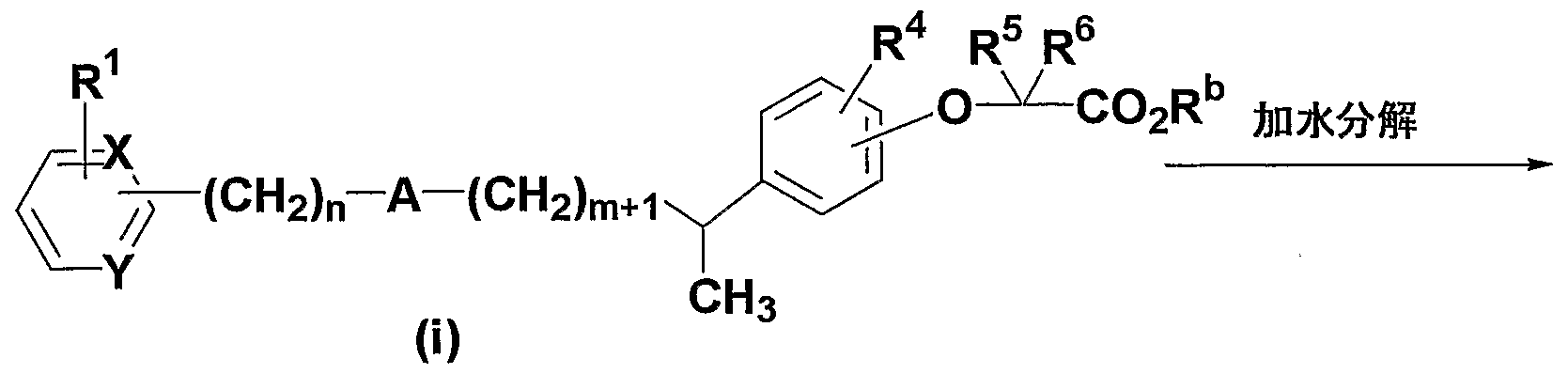
- the ester compound represented by the general formula (i) is the above synthesis method 2 Can be obtained by subjecting the exomethylene compound represented by the general formula (g) described in 1 to a catalytic hydrogenation reaction using palladium-carbon.
- the bur compound represented by the general formula (m) is composed of the aldehyde compound represented by the general formula (k) and the general formula.
- the acetophenone compound represented by (1) can be obtained by an aldol condensation reaction in the presence of a base.
- the phenol compound represented by the general formula (n) is obtained by reducing the reaction of the olefin moiety with the bur compound represented by the general formula (m). It can be obtained by subjecting to reaction and debenzylation reaction.
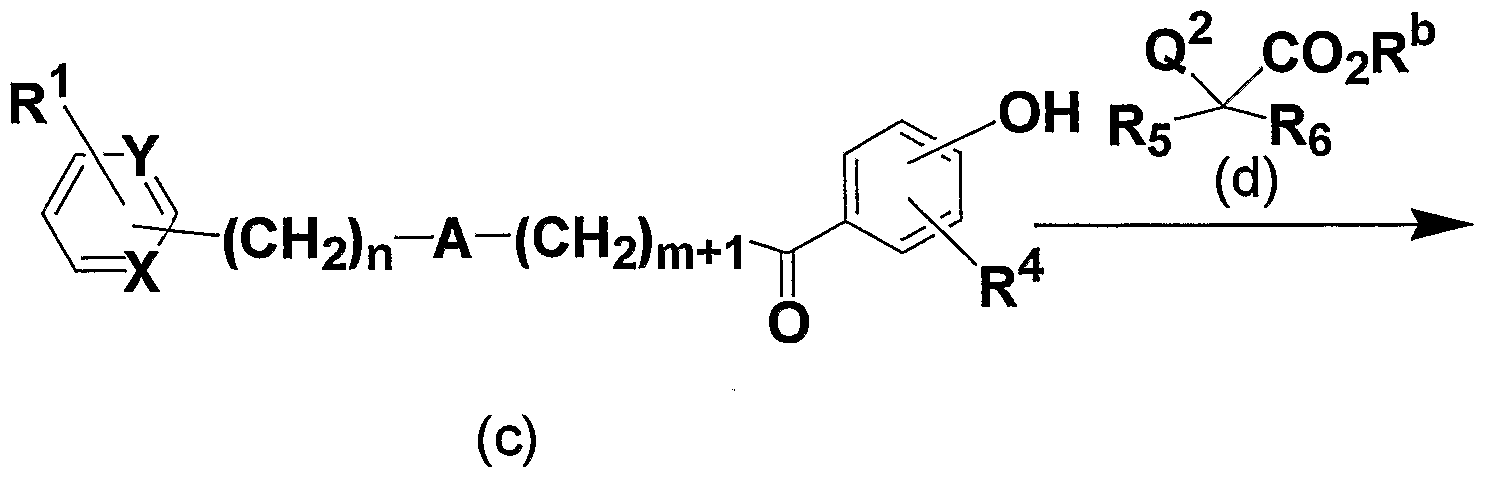
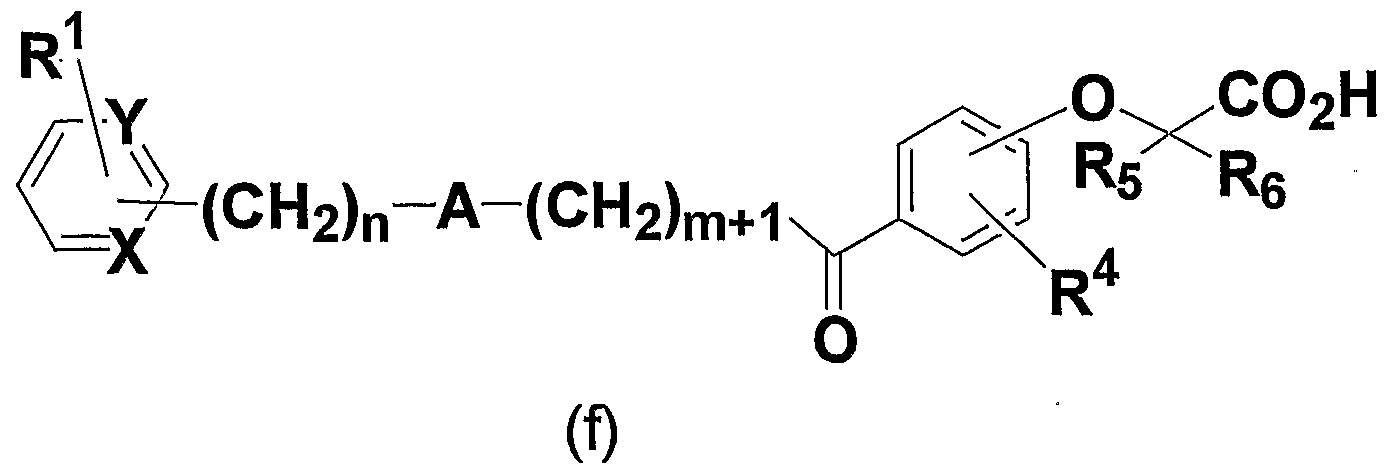
- ester compound represented by the general formula (p) can be obtained by reacting the phenol compound represented by the general formula (n) with the compound represented by the general formula (o).
- the present formula represented by the general formula (s) Inventive compounds can be obtained.
- the compound of the present invention represented by the above general formula (I) can also be produced with reference to the above-mentioned patent documents and publicly known documents in addition to the examples described below. Examples of the compounds of the present invention thus obtained are shown in the table.
- the P PAR ⁇ activation action of the compound of the present invention was measured as follows for the test compound (Example compound).
- Receptor expression plasmid (p SG 5— GAL 4— h PPAR aor ⁇ or ⁇ (LBD), luciferase expression plasmid ( ⁇ UC 8— ⁇ 1 00 ⁇ , 4— ⁇ ) 1 luc) and] 3 galactosidase expression plasmids (p CMX—; 3-GAL) (K liewer, SA et. Al. F (1 9 9 2) Nature, 3 5 8: 7 7 1- 7 74) Introduced the gene into cells using Lipofection Reagent DMR IE-C or Lipofectamin 2000 (I 11 vitrogen), and then cultured in the presence of the test compound for 42 hours.
- the compounds represented by the general formulas (I), (II) and (III) of the present invention have an excellent PPAR ⁇ activation action, they are hypoglycemic agents, obesity, syndrome X, high cholesterol.
- disorders metabolic disorders such as hyperlipoproteinemia, hyperlipidemia, arteriosclerosis, cardiovascular disease, bulimia, ischemic disease, lung cancer, breast cancer, colon cancer, large intestine cancer, ovarian cancer
- malignant tumors such as Alzheimer's disease and inflammatory diseases.
- the compound of the present invention can be administered to humans by an appropriate administration method such as general oral administration or parenteral administration.
- a dosage form such as tablets, granules, powders, capsules, suspensions, injections, suppositories and the like by conventional methods in the technical field of preparations.
- excipients include lactose, D-mannitol, crystalline cellulose, and glucose
- disintegrants include starch, carbo'xymethyl cellulose and calcium (CMC-Ca), and lubricants.
- binder examples include magnesium stearate and talc
- examples of the binder include hydroxypropylcellulose (HP C), gelatin, and polybutylpyrrolidone (PVP).
- the dose is usually about 0.1 mg / day to LO Omg of the compound of the present invention which is an active ingredient in an injection for oral administration, and 1 mg to 2000 mg / day orally, depending on age, symptoms, etc. It can be increased or decreased.
- the reaction mixture was concentrated under reduced pressure, and ice water and 1 M aqueous hydrochloric acid solution were added to the obtained residue to adjust to pH 3.
- ethyl acetate 5 O m LX 2
- the organic layer was washed with water (3 O mL) and saturated brine (3 O mL), and dried over anhydrous sodium sulfate.
- the solvent was distilled off under reduced pressure to obtain 12 1 mg (yield 76%) of the title compound as a white amorphous.
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0609178A BRPI0609178A2 (pt) | 2005-02-28 | 2006-02-28 | composto ou um sal do mesmo, e, ativador para o receptor ativado do proliferador de peroxissoma delta |
| CA002599454A CA2599454A1 (en) | 2005-02-28 | 2006-02-28 | Activator for peroxisome proliferator activating receptor d |
| JP2007504849A JPWO2006090920A1 (ja) | 2005-02-28 | 2006-02-28 | ペルオキシソーム増殖剤活性化受容体δの活性化剤 |
| EP06715252A EP1854784A1 (en) | 2005-02-28 | 2006-02-28 | Activator for peroxisome proliferator activating receptor |
| AU2006217682A AU2006217682A1 (en) | 2005-02-28 | 2006-02-28 | Activator for peroxisome proliferator activating receptor delta |
| IL185536A IL185536A0 (en) | 2005-02-28 | 2007-08-27 | Activator for peroxisome proliferator activating receptor delta |
| NO20074738A NO20074738L (no) | 2005-02-28 | 2007-09-17 | Aktivator for peroksisom-proliferator som aktiverer reseptor delta |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005052762 | 2005-02-28 | ||
| JP2005-052762 | 2005-02-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006090920A1 true WO2006090920A1 (ja) | 2006-08-31 |
Family
ID=36927539
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2006/304193 WO2006090920A1 (ja) | 2005-02-28 | 2006-02-28 | ペルオキシソーム増殖剤活性化受容体δの活性化剤 |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US20090240058A1 (ja) |
| EP (1) | EP1854784A1 (ja) |
| JP (1) | JPWO2006090920A1 (ja) |
| KR (1) | KR20070113253A (ja) |
| CN (1) | CN101166720A (ja) |
| AU (1) | AU2006217682A1 (ja) |
| BR (1) | BRPI0609178A2 (ja) |
| CA (1) | CA2599454A1 (ja) |
| IL (1) | IL185536A0 (ja) |
| MX (1) | MX2007010511A (ja) |
| NO (1) | NO20074738L (ja) |
| RU (1) | RU2007135745A (ja) |
| WO (1) | WO2006090920A1 (ja) |
| ZA (1) | ZA200707899B (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2910894A1 (fr) * | 2006-12-29 | 2008-07-04 | Genfit Sa | Derives de 3-phenyl-1-(phenylthienyl)propan-1-one et de 3-phenyl-1-(phenylfuranyl)propan-1-one substitues, preparation et utilisation. |
| CN104672220A (zh) * | 2008-04-15 | 2015-06-03 | 日本化学医药株式会社 | 过氧化物酶体增殖剂活化受体的活化剂 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008154023A1 (en) * | 2007-06-11 | 2008-12-18 | Cerenis Therapeutics S.A. | Novel uses of ppar delta agonists |
| US20110092517A1 (en) * | 2009-08-14 | 2011-04-21 | Cerenis Therapeutics S.A. & Nippon Chemiphar Co., Ltd. | Use of PPAR Delta Ligands for the Treatment or Prevention of Inflammation or Energy Metabolism/Production Related Diseases |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0680630A (ja) * | 1990-02-13 | 1994-03-22 | Bristol Myers Squibb Co | 複素環カルボン酸およびエステル |
| WO2002014291A1 (en) * | 2000-08-11 | 2002-02-21 | Nippon Chemiphar Co.,Ltd. | PPARδ ACTIVATORS |
| WO2003016291A1 (fr) * | 2001-08-10 | 2003-02-27 | Nippon Chemiphar Co., Ltd. | Activateur du recepteur $g(d) sensible au proliferateur de peroxysome |
| JP2003292439A (ja) * | 2002-02-04 | 2003-10-15 | Sumitomo Pharmaceut Co Ltd | 新規ピロール誘導体からなるppar活性化剤 |
| WO2004022551A1 (ja) * | 2002-09-06 | 2004-03-18 | Takeda Pharmaceutical Company Limited | フランまたはチオフェン誘導体およびその医薬用途 |
| WO2004063166A1 (en) * | 2003-01-06 | 2004-07-29 | Eli Lilly And Company | Heterocyclic ppar modulators |
| JP2004534035A (ja) * | 2001-05-11 | 2004-11-11 | グラクソ グループ リミテッド | ヒト・ペルオキシソーム増殖因子活性化受容体を活性化するフランおよびチオフェン誘導体 |
| WO2005049578A1 (en) * | 2003-11-17 | 2005-06-02 | Smithkline Beecham Corporation | Substituted pyrazoles as ppar agonists |
| WO2005054213A1 (ja) * | 2003-12-02 | 2005-06-16 | Shionogi & Co., Ltd. | ペルオキシソーム増殖活性化受容体アゴニスト活性を有するイソキサゾール誘導体 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4956379A (en) * | 1990-02-13 | 1990-09-11 | Bristol-Myers Squibb Company | Pyrazole carboxylic acids and esters and inhibition of blood platelet aggregation therewith |
-
2006
- 2006-02-28 MX MX2007010511A patent/MX2007010511A/es not_active Application Discontinuation
- 2006-02-28 EP EP06715252A patent/EP1854784A1/en not_active Withdrawn
- 2006-02-28 RU RU2007135745/04A patent/RU2007135745A/ru not_active Application Discontinuation
- 2006-02-28 US US11/885,265 patent/US20090240058A1/en not_active Abandoned
- 2006-02-28 KR KR1020077021867A patent/KR20070113253A/ko not_active Application Discontinuation
- 2006-02-28 ZA ZA200707899A patent/ZA200707899B/xx unknown
- 2006-02-28 BR BRPI0609178A patent/BRPI0609178A2/pt not_active IP Right Cessation
- 2006-02-28 CN CNA2006800145540A patent/CN101166720A/zh active Pending
- 2006-02-28 AU AU2006217682A patent/AU2006217682A1/en not_active Abandoned
- 2006-02-28 WO PCT/JP2006/304193 patent/WO2006090920A1/ja active Application Filing
- 2006-02-28 JP JP2007504849A patent/JPWO2006090920A1/ja active Pending
- 2006-02-28 CA CA002599454A patent/CA2599454A1/en not_active Abandoned
-
2007
- 2007-08-27 IL IL185536A patent/IL185536A0/en unknown
- 2007-09-17 NO NO20074738A patent/NO20074738L/no not_active Application Discontinuation
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0680630A (ja) * | 1990-02-13 | 1994-03-22 | Bristol Myers Squibb Co | 複素環カルボン酸およびエステル |
| WO2002014291A1 (en) * | 2000-08-11 | 2002-02-21 | Nippon Chemiphar Co.,Ltd. | PPARδ ACTIVATORS |
| JP2004534035A (ja) * | 2001-05-11 | 2004-11-11 | グラクソ グループ リミテッド | ヒト・ペルオキシソーム増殖因子活性化受容体を活性化するフランおよびチオフェン誘導体 |
| WO2003016291A1 (fr) * | 2001-08-10 | 2003-02-27 | Nippon Chemiphar Co., Ltd. | Activateur du recepteur $g(d) sensible au proliferateur de peroxysome |
| JP2003292439A (ja) * | 2002-02-04 | 2003-10-15 | Sumitomo Pharmaceut Co Ltd | 新規ピロール誘導体からなるppar活性化剤 |
| WO2004022551A1 (ja) * | 2002-09-06 | 2004-03-18 | Takeda Pharmaceutical Company Limited | フランまたはチオフェン誘導体およびその医薬用途 |
| WO2004063166A1 (en) * | 2003-01-06 | 2004-07-29 | Eli Lilly And Company | Heterocyclic ppar modulators |
| WO2005049578A1 (en) * | 2003-11-17 | 2005-06-02 | Smithkline Beecham Corporation | Substituted pyrazoles as ppar agonists |
| WO2005054213A1 (ja) * | 2003-12-02 | 2005-06-16 | Shionogi & Co., Ltd. | ペルオキシソーム増殖活性化受容体アゴニスト活性を有するイソキサゾール誘導体 |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2910894A1 (fr) * | 2006-12-29 | 2008-07-04 | Genfit Sa | Derives de 3-phenyl-1-(phenylthienyl)propan-1-one et de 3-phenyl-1-(phenylfuranyl)propan-1-one substitues, preparation et utilisation. |
| WO2008087366A2 (fr) | 2006-12-29 | 2008-07-24 | Genfit | Derives de 3-phenyl-1-(phenylthienyl)propan-1-one et de 3-phenyl-1-(phenylfuranyl)propan-1-one substitues, preparation et utilisation |
| WO2008087366A3 (fr) * | 2006-12-29 | 2008-10-23 | Genfit | Derives de 3-phenyl-1-(phenylthienyl)propan-1-one et de 3-phenyl-1-(phenylfuranyl)propan-1-one substitues, preparation et utilisation |
| JP2010514745A (ja) * | 2006-12-29 | 2010-05-06 | ジェンフィット | 置換3−フェニル−1−(フェニルチエニル)プロパン−1−オンおよび3−フェニル−1−(フェニルフラニル)プロパン−1−オンの誘導体、その調製および使用 |
| US8088819B2 (en) | 2006-12-29 | 2012-01-03 | Delhomel Jean-Francois | Derivatives of substituted 3-phenyl-1-(phenylthienyl)propan-1-ones and of 3-phenyl-1-(phenylfuranyl) propan-1-ones, preparation and use |
| EA017449B1 (ru) * | 2006-12-29 | 2012-12-28 | Жанфит | Производные замещенных 3-фенил-1-(фенилтиенил)пропан-1-онов и 3-фенил-1-(фенилфуранил)пропан-1-онов, их получение и применение |
| AU2007344310B2 (en) * | 2006-12-29 | 2013-08-15 | Genfit | Substituted 3-phenyl-1-(phenylthienyl)propan-1-one and 3-phenyl-1-(phenylfuranyl)propan-1-one derivatives, and preparation and use of same |
| CN101605775B (zh) * | 2006-12-29 | 2014-11-12 | 基恩菲特公司 | 取代的3-苯基-1-(苯基噻吩基)丙-1-酮类以及3-苯基-1-(苯基呋喃基)丙-1-酮类的衍生物、制备以及用途 |
| KR101505577B1 (ko) * | 2006-12-29 | 2015-03-30 | 장피트 | 치환된 3-페닐-1-(페닐티에닐)프로판-1-온 및 3-페닐-1-(페닐푸라닐)프로판-1-온의 유도체, 그 제조 및 용도 |
| CN104672220A (zh) * | 2008-04-15 | 2015-06-03 | 日本化学医药株式会社 | 过氧化物酶体增殖剂活化受体的活化剂 |
| CN104672220B (zh) * | 2008-04-15 | 2017-09-22 | 日本化学医药株式会社 | 过氧化物酶体增殖剂活化受体的活化剂 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1854784A1 (en) | 2007-11-14 |
| ZA200707899B (en) | 2008-12-31 |
| AU2006217682A1 (en) | 2006-08-31 |
| CN101166720A (zh) | 2008-04-23 |
| KR20070113253A (ko) | 2007-11-28 |
| BRPI0609178A2 (pt) | 2016-08-23 |
| RU2007135745A (ru) | 2009-04-10 |
| NO20074738L (no) | 2007-11-08 |
| MX2007010511A (es) | 2007-11-07 |
| JPWO2006090920A1 (ja) | 2008-07-24 |
| CA2599454A1 (en) | 2006-08-31 |
| IL185536A0 (en) | 2008-01-06 |
| US20090240058A1 (en) | 2009-09-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2006059744A1 (ja) | ペルオキシソーム増殖剤活性化受容体δの活性化剤 | |
| JP4790969B2 (ja) | ペルオキシソーム増殖剤応答性受容体δの活性化剤 | |
| KR101507173B1 (ko) | 페록시좀 증식제 활성화 수용체 δ 의 활성화제 | |
| WO2001079197A1 (en) | ACTIVATORS FOR PEROXISOME PROLIFERATOR ACTIVATED RECEPTOR δ (PPARδ) | |
| JP5474769B2 (ja) | ペルオキシソーム増殖剤活性化受容体の活性化剤 | |
| AU2013341945A1 (en) | GPR40 receptor agonist, methods of preparing the same, and pharmaceutical compositions containing the same as an active ingredient | |
| WO2002076957A1 (fr) | Activateur de recepteur active par les proliferateurs du peroxysome | |
| WO2004024705A1 (ja) | 5員複素環化合物 | |
| EP2480530A1 (en) | Alkene oxindole derivatives and their uses to treat obesity, diabetes and hyperlipidemia | |
| IE910766A1 (en) | Novel pyrazole derivatives which are angiotensin ii¹receptor antagonists; their methods of preparation¹and pharmaceutical compositions in which they are present | |
| WO2002046154A1 (en) | Peroxisome proliferator activated receptor d activators | |
| WO2006090920A1 (ja) | ペルオキシソーム増殖剤活性化受容体δの活性化剤 | |
| KR20050042130A (ko) | Cck-1 수용체 조절제 | |
| WO2007004733A1 (ja) | ペルオキシソーム増殖剤活性化受容体δの活性化剤 | |
| WO2006041197A1 (ja) | ペルオキシソーム増殖剤活性化受容体δの活性化剤 | |
| WO2015162452A1 (en) | Substituted pyrazole compounds as cb1 receptor antagonists and uses thereof | |
| JP2005179281A (ja) | ビフェニル化合物 | |
| WO2002085851A1 (fr) | Derive de type pyrrole | |
| Dabak et al. | Synthesis of 1-vinyl 1, 2, 3-triazole derivatives | |
| WO2001030771A1 (fr) | Derives de thiazolidinedione | |
| US20030114456A1 (en) | Novel heterocylic compounds with anti-inflamatory activity | |
| WO2004048341A1 (ja) | 新規ヘテロアリール誘導体 | |
| JP2006036730A (ja) | 新規ピロール誘導体 | |
| EP2639212A1 (en) | Enantioselective organic anhydride reactions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200680014554.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2007504849 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 185536 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2599454 Country of ref document: CA Ref document number: MX/a/2007/010511 Country of ref document: MX Ref document number: 12007501843 Country of ref document: PH |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 561396 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006715252 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020077021867 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006217682 Country of ref document: AU Ref document number: 4285/CHENP/2007 Country of ref document: IN Ref document number: 1200701975 Country of ref document: VN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007135745 Country of ref document: RU |
|
| ENP | Entry into the national phase |
Ref document number: 2006217682 Country of ref document: AU Date of ref document: 20060228 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2006217682 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2006715252 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: PI0609178 Country of ref document: BR Kind code of ref document: A2 Effective date: 20070828 |