WO2004029033A1 - バレロラクトン化合物及び香料組成物 - Google Patents
バレロラクトン化合物及び香料組成物 Download PDFInfo
- Publication number
- WO2004029033A1 WO2004029033A1 PCT/JP2003/012341 JP0312341W WO2004029033A1 WO 2004029033 A1 WO2004029033 A1 WO 2004029033A1 JP 0312341 W JP0312341 W JP 0312341W WO 2004029033 A1 WO2004029033 A1 WO 2004029033A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- methyl
- fragrance composition
- valerolactone
- group
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D315/00—Heterocyclic compounds containing rings having one oxygen atom as the only ring hetero atom according to more than one of groups C07D303/00 - C07D313/00
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L27/00—Spices; Flavouring agents or condiments; Artificial sweetening agents; Table salts; Dietetic salt substitutes; Preparation or treatment thereof
- A23L27/20—Synthetic spices, flavouring agents or condiments
- A23L27/205—Heterocyclic compounds
- A23L27/2052—Heterocyclic compounds having oxygen or sulfur as the only hetero atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D263/00—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings
- C07D263/52—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings condensed with carbocyclic rings or ring systems
- C07D263/54—Benzoxazoles; Hydrogenated benzoxazoles
- C07D263/58—Benzoxazoles; Hydrogenated benzoxazoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D309/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings
- C07D309/16—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D309/28—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D309/30—Oxygen atoms, e.g. delta-lactones
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11B—PRODUCING, e.g. BY PRESSING RAW MATERIALS OR BY EXTRACTION FROM WASTE MATERIALS, REFINING OR PRESERVING FATS, FATTY SUBSTANCES, e.g. LANOLIN, FATTY OILS OR WAXES; ESSENTIAL OILS; PERFUMES
- C11B9/00—Essential oils; Perfumes
- C11B9/0069—Heterocyclic compounds
- C11B9/0073—Heterocyclic compounds containing only O or S as heteroatoms
- C11B9/008—Heterocyclic compounds containing only O or S as heteroatoms the hetero rings containing six atoms
Definitions
- the present invention relates to a valerolactone compound and a method for producing the same, and a fragrance composition containing the valerolactone compound.
- Sandals (Japanese name: sandalwood) are prized as Buddha statues and various sculpture materials for crafts in the Orient, because they are mainly produced in eastern India and have a noble and unique aroma.
- the essential oil obtained by steam distillation from its core and roots has long been used as a fragrance.
- This sandalwood oil has a soft and sweet pedi fragrance and a balsamic fragrance, and is characterized by its high persistence. It is widely used in oriental-type compounded fragrances, perfumes, cosmetics, stones, incense sticks, etc. (For example, see Motoki Nakajima, “Basic Knowledge of Fragrances and Incense,” Sangyo Tosho Co., Ltd., First Edition, Third Edition, 2000, p. 322-233).
- esters for example, 7-pentyl
- 7-Putyrolactone also known as ⁇ -nonalactone
- d-pentyloo (5-valerola lactone also known as 5-decalactone
- 7-Ethyl—7-1 butenyl-5-valerolactone (see, for example, Japanese Patent Application Laid-Open No. 53-84975) is a Costas-like lactone, saturated 7 "-ethyl-7-.
- Butyl- ⁇ 5-valerolactone (eg, manufactured by PFW, trade name: C0STAU and ON) is a Costas-like and Peddy-like lactone, and a lactone compound that can be used as a perfume is 4-cyclohexyl pen.
- Tanolide see, for example, Japanese Patent Publication No. 62-51223
- —alkyl-1-alkyl-5-alkyl-5-valerolactone are known (see, for example, US Pat. No. 3,380,380). 4 5 7 Specification See).
- the present invention provides a compound of formula (I):
- R 1 and R 2 each independently represent a hydrogen atom, a methyl group or an ethyl group, except when R 1 and R 2 are simultaneously a hydrogen atom.
- R 3 is a hydrogen atom or a methyl group
- R 4 represents a propyl group, a 1-propenyl group or a phenyl group
- the present invention provides a valerolactone compound which emits a scent of lactone having a coumarin-like sweetness reminiscent of eddy aroma and tonto beans, and has an excellent residual odor, and a valerolactone compound described above in a perfume composition.
- the present invention relates to a fragrance composition capable of exhibiting the characteristics of natural sand beads.
- valerolactone compound represented by the formula (I) described below emits a peculiar fragrance which itself has a sweet scent such as Puddy scent and coumarin, and the scent is harmonized with Peddi scent. It can produce a sweet scent, especially when combined with a sandalwood-based synthetic fragrance, and can easily exhibit the characteristics of natural sandals that were insufficient for synthetic products. Found
- the valerolactone compound represented by the formula (II) of the present invention can be produced by reacting the obtained reaction solution with an acid after allowing a salt to act on propanal. Further, the valerolactone compound represented by the formula (II) of the present invention can be produced by treating 2-methyl-2-pentenal with a base and treating the resulting reaction solution with an acid. It is possible.
- the present invention has made it possible to provide a fragrance composition having a sweet scent in harmony with eddy scent by including a valerolactone compound represented by the formula (I) in the composition. Furthermore, the present invention provides a sweet scent that matches a scent that is closer to a natural sandalwood by adding such a fragrance composition to household products, cosmetics products, environmental hygiene products, beverages and foods. Can be assigned.
- R 1 and R 2 each independently represent a hydrogen atom, a methyl group or an ethyl group, except when R 1 and R 2 are simultaneously a hydrogen atom.
- R 3 is a hydrogen atom or a methyl group
- R 4 represents a propyl group, a 1-propenyl group or a phenyl group
- valerolactone compound according to the present invention has the formula (II):
- valerolactone compound according to the present invention is obtained by allowing a base to act on propanal.
- a valerolactone compound represented by the formula (II) obtained by treating the obtained reaction solution with an acid.
- the production method according to the present invention is a method for producing a valerolactone compound represented by the formula (II), wherein a base is allowed to act on 2-methyl-2-pentenal, and the resulting reaction solution is treated with an acid. Is the law.
- the fragrance composition according to the present invention is a fragrance composition containing a valerolactone compound represented by the formula (I).
- the fragrance composition according to the present invention is a fragrance composition containing a valerolactone compound represented by the formula (II).
- a fragrance composition according to the present invention is a fragrance composition containing the valerolactone compound according to (3).
- fragrance composition according to the present invention is a compound represented by the following formulas (III) to (VI I):
- a fragrance composition comprising one or more compounds selected from the group consisting of:
- the fragrance composition according to the present invention comprises the above-mentioned (6) to (6), which contains two or more compounds selected from the group consisting of the compounds represented by the formulas (III) to (VII). 8) Perfume set described It is an adult.
- the fragrance composition according to the present invention comprises: Component (A): 2-ethyl-4-1- (2,2,3-trimethyl-3-cyclopentene-1-yl) -12-butene-1-ol , 3-Methyl-1-5- (2,2,3-trimethyl-1-cyclopentene-1-yl) -pentan-2-ol, 2-methyl-1-4-1 (2,2,3-trimethyl-3-cyclopentene-1-1-1 1) Butanol, 3,3-dimethyl-5_ (2,2,
- fragrance composition according to any one of the above (6) to (8), comprising a kind or two or more kinds of compounds.
- the present invention is a.
- FIG. 1 is a 1 ⁇ - ⁇ MR (400 MHz) chart of 1 -methyl-7-methyl-1-7-1-propenylvalerolactone.
- Figure 2 shows the red color of 1-methyl-17-methyl-1-propenylvalerolactone.
- 6 is a graph showing the results of determining the absorption spectrum.
- FIG. 3 is a 1 H-N MR (400 Mz) chart of ethyl 7-methyl-111-propenyl valerolactone.
- FIG. 4 is a graph showing the measurement results of the infrared absorption spectrum of 1-ethyl-17-methyl-7-1-propenylvalerolactone.
- FIG. 5 is a 1 H-NMR (400 MHz) chart of 1-methyl-1-ethyl-methyl-1-phenylvalerolactone.
- FIG. 6 is a graph showing the measurement results of the infrared absorption spectrum of ⁇ -methyl-1-ethyl-methyl-17-phenylvalerolactone.
- FIG. 7 is a graph showing the results of measurement of the infrared absorption spectrum of permethyl-ethyl-7 * -methyl-1-propenyl-5-valerolactone obtained in Example 4 of the present invention. is there.
- FIG. 9 is a 1 H-MR (400 Hz) chart of ⁇ -methyl- ⁇ -ethyl-7-methyl-1- ⁇ -propyl-15-valerolactone.
- FIG. 10 is a graph showing the measurement results of the infrared absorption spectrum of 1-methyl-1-ethyl- ⁇ -methyl-7-propyl-1 valerolactone.
- the valerolactone compound of the present invention is represented by the formula (I) and has a sweet scent useful as a fragrance substance.
- valerolactone compounds represented by the formula (I) the compound represented by the formula (II) —methyl-ethyl- ⁇ - ⁇ -methyl- ⁇ - 1-probelule 5-valerolactone is itself. Especially preferred because it has a sweet scent like coumarin reminiscent of Peddy incense and toniki beans, and is easy to harmonize with Peddy flavors. That is, represented by equation (II) When a fragrance composition is prepared by combining a valerolactone compound and a peddy-based fragrance, a soft and sweet milk-like scent in harmony with the peddy scent can be developed.
- valerolactone compound represented by the formula (II) when the valerolactone compound represented by the formula (II) is combined with a sandalwood-based synthetic fragrance, it is in harmony with the sandalwood-based synthetic fragrance of the fragrance, soft and sweet, and has a milk-like fragrance and pulsamic. A natural fragrance can be easily obtained, and the characteristics of natural sandal wood can be easily exhibited. In addition, this valerolactone compound also has the advantage of having high residual odor.
- the valerolactone compound represented by the formula (I) may have a position other than the 5-position, a ⁇ -position or
- valerolactones generally used as fragrances have an alkyl substituent at the 5-position, the odor is sweet and coconut-like, and the milk-like characteristics become stronger as the carbon number increases. Due to its nature, it is often used for food flavors.
- the valerolactone compound represented by the formula (I) has the same sweetness as the alkyl substituted at the 5-position, despite having no alkyl substituent at the ⁇ 5-position, and at the same time, Various fragrances, for example, household products such as detergents for clothing, softeners for clothing, stoneware, body wash, hair cosmetics, cosmetics, perfumes, etc. It has the advantage of being in harmony with the fragrances used in products or in environmentally friendly products such as fragrances, deodorants, incense sticks and candles.
- the compound of formula (II) has a combination of eddy and spice odors, and has a new strong odor reminiscent of Costas and Iris.
- the compound represented by the formula (IV) has both a peddy smell and a floral smell; Has a soft new odor compared to the compound represented by
- the compound represented by the formula (V) has a seddy wood-like odor and a coumarin-like odor.
- the compound represented by the formula ( ⁇ ) has a combination of a peddy smell and a green scent, and has a novel odor that is slightly weaker than the compound represented by the formula (II) but is pronounced of a sandalwood.
- the valerolactone compound represented by the formula (I) can be contained alone in the fragrance composition, or two or more of these valerolactone compounds can be used in combination.
- a compound represented by the formula (II), a compound represented by the formula (III), a compound represented by the formula (IV), a compound represented by the formula (V), a compound represented by the formula (VI) Combining two or more selected from the group consisting of compounds and compounds represented by formula (VI I) to create a variety of fragrances and intensities of peddy, floral, spice, and kossu It has the advantage that it can be more easily harmonized with the fragrance for products than when these are used alone.
- the compound represented by the formula (V) has a novel odor with a scent of eddy and coumarin.
- the combination of the compound represented by the formula (V) and the compound represented by the formula (II) is
- valerolactone compound represented by the formula (I) can be synthesized, for example, by the route shown in Scheme 1.
- an alkali such as sodium methoxide
- an acrylonitrile derivative and a malonic ester derivative are subjected to an all-condensation reaction, and then the obtained compound ⁇ is converted into lithium borohydride (LiBH 4 ) can be converted to compound B with a hydride reducing reagent such as Thereafter, under alkaline conditions (such as aqueous sodium hydroxide solution), the nitrile moiety is hydrolyzed, and hydroxyvalerolactone C can be synthesized with an acid such as sulfuric acid.
- the hydroxy group is oxidized with an oxidizing agent such as pyridinium dichromate (PDC) to form an aldehyde D, and the carbon is increased by the wittig reaction.
- PDC pyridinium dichromate
- Compound F can be synthesized by isomerization of Olefin.
- the compound F ′ can be easily synthesized by using a hydrogenation catalyst such as hydrogen and palladium monocarbon (Pd / C) as the olefin in the ⁇ -position side chain.
- a hydrogenation catalyst such as hydrogen and palladium monocarbon (Pd / C) as the olefin in the ⁇ -position side chain.
- a compound in which R 4 is a phenyl group can be synthesized by the route shown in Scheme 2. More specifically, compound G can be synthesized by performing an alkali condensation reaction of an acrolein derivative with an aldehyde and acidifying it.
- X represents a hydrogen atom or a metal atom.
- a potassium atom As the metal atom, a potassium atom
- alkali metal atoms such as sodium atom and alkaline earth metal atoms such as calcium and magnesium.
- X in the compound represented by the formula (L) is XZ 2.
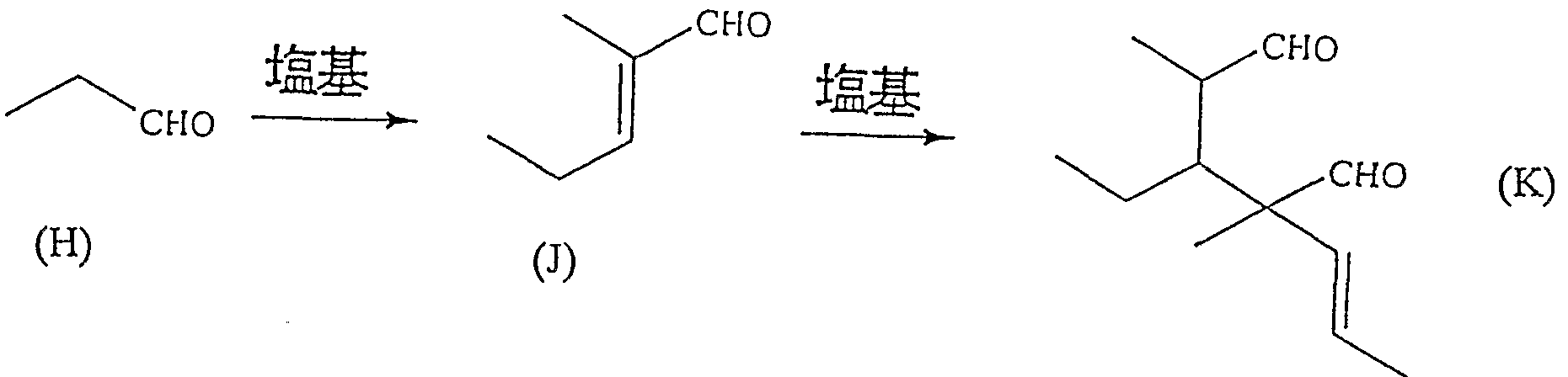
- the compound represented by the formula (K) is obtained by allowing a base to act on propanal represented by the formula (H) or 2-methyl-2-pentenal represented by the formula (J),
- the compound represented by the formula (L) is sequentially formed, and the resulting reaction solution is treated with an acid, whereby the -C00X group and the -OH group of the compound represented by the formula (L) react to close the ring.
- a valerolactone compound represented by the formula (N) is produced.
- a base is allowed to act on propanal or 2-methyl-2-pentenal.
- the base can be made to act on propanal or 2-methyl-2-pentenal in a solvent.
- a polar solvent is preferable from the viewpoint of enhancing the reactivity, and for example, a lower alcohol having 1 to 3 carbon atoms such as methanol, ethanol, and isopropanol, and water are preferable. Among them, a combination of water and a lower alcohol is preferred.
- the amount of the solvent is preferably from 0 to 1,000 parts by mass, more preferably from 50 to 700 parts by mass, based on 100 parts by mass of propanal or 2-methyl-2-pentenal, from the viewpoint of improving the yield and increasing the reactivity. Parts, more preferably 100 to 500 parts by mass.
- Examples of the base include those having 1 to 3 carbon atoms, such as alkali metal hydroxides such as potassium hydroxide and sodium hydroxide, alkaline earth metal hydroxides such as calcium hydroxide and magnesium hydroxide, and potassium methoxide and sodium methoxide.
- alkali metal salts of lower alcohols include one or more selected from the group consisting of potassium titanium oxide, sodium hydroxide, potassium methoxide and sodium methoxide are preferred.
- the amount of the base is preferably from 0.001 to 10 mol, more preferably from 0.005 to 5 mol, and still more preferably from 0.01 to 1 mol, per mol of propanal or 2-methyl-2- ⁇ tennal. Is a mole.
- a method of mixing propanal or 2-pentanal with a base for example, dissolving a base in a solvent in advance and then preparing a solution of the solution
- the method includes adding propanal or 2-methyl-2-pentenal to a solvent, dissolving propanal or 2-methyl-2-pentenal in a solvent, and adding a base or the solution to the solution.
- the atmosphere in which the base is allowed to act on propanal or 2-methyl-2-entenal is not particularly limited, but is preferably an inert gas atmosphere such as nitrogen gas or argon gas.
- the temperature at which the base is allowed to act on propanal or 2-methyl-2-pentenal is not particularly limited. Usually, such a temperature may be 20 to 60 ° C.
- the end point when a base is allowed to act on propanal or 2-methyl-2-pentenal is, for example, the point when propanal or 2-methyl-2-pentenal disappears by gas chromatography, thin-layer liquid chromatography, etc. It can be.
- the time required for the base to act on propanal or 2-methyl-2-pentenal varies depending on the reaction conditions and cannot be unconditionally determined, and is generally about 1 to 48 hours.
- the valerolactone compound can be obtained by treating the obtained reaction solution with an acid.
- a general-purpose ether such as Jetil ether should be used as the ether.
- the amounts of water and ether are not particularly limited, but are usually preferably 50 to 200 parts by mass with respect to 100 parts by mass of the reaction solution.
- the acid examples include inorganic acids such as hydrochloric acid and phosphoric acid, and organic acids such as acetic acid, citric acid and tartaric acid. Of these, inorganic acids are preferred.
- the treatment of the reaction solution with an acid is performed by adding an acid to the reaction solution (aqueous layer) so that the pH of the reaction solution (aqueous layer) is 1 to 6, preferably 2 to 4, to make the reaction solution acidic.
- the temperature of the reaction solution at this time is not particularly limited, but usually may be about 5 to 40 ° C.
- the end point of the acid treatment is determined when pH is measured using a pH test paper (for example, Merck, trade name: Acilit pH 0 to 6) and the pH reaches the aforementioned pH. be able to.
- the reaction mixture thus obtained by the treatment with an acid contains the valerolactone compound of the present invention.
- ether is added to the reaction mixture, and the mixture is extracted, and then purified by silica gel column chromatography or the like, thereby isolating the relolactone compound.
- the fragrance composition of the present invention contains a valerolactone compound represented by the formula (I). Since the valerolactone compound represented by the formula (I) has a novel scent, a new scent can be easily produced by combining it with various scents.
- fragrance substances that can be used in combination with the valerolactone compound of the present invention include the following.
- Hydrocarbons such as limonene, ⁇ -bine, human ⁇ -binene, terbinene, cedrene, longifolene, valencene;
- Phenols such as eugenol, thymol and vanillin;
- Methylheptenone 4-methylene-1,3,5,6,6-tetramethyl-1-heptanone, amylcyclopentene, 3-methyl-2- (cis-1-pentene-11-yl) 1-2-cyclopentene 1-one, methylcyclopentenolone, rose ketone, 7-methylyonone, hyonynon, carvone, mentone, show brain, acetylcedrene, isolongiforanone, nootkatone, benzylacetone, anicilacetone, methyl ⁇ naphthylketone , 2,5-Dimethyl-1-hydroxy-3 (2 ⁇ ) -ketones such as furanone, maltol, muscone, cibetone, cyclopentadecanone, cyclohexadecene;
- Acetals such as formaldehyde cyclododecylethyl acetal, acetoaldehyde ethyl phenylpropyl acetal, citral cetyl acetal, phenylacetaldehyde glycerin acetal, ethyl acetoacetate ethylene glycol ketal, and ketals;
- 7-nonalactone, 7-dedecalactone, 5-decalactone, 7-jasmolactone, coumarin, cyclopentene decanolide, cyclohexadecanolide, amplettrid, ethylene brasilate, and 11-loxa Lactones such as oxadecanolide, orange, lemon, bergamot, mandarin, peppermint, spearmint, lavender, chamomil, rosemary, and u
- natural essential oils and extracts such as potash, sage, basil, rose, geranium, jasmine, ylang-ylang, anis, clove, ginger, nutmeg, cardamom, seda, hinoki, vetiver, pachiyori, rabudanam, etc. It is possible.
- the valerolactone compound represented by the formula (I) of the present invention can be arbitrarily combined with the fragrance substance.
- the content of the valerolactone compound in the fragrance composition is not particularly limited, but is usually 0.001% by mass or more, preferably 0.01% by mass or more, and more preferably 0.2% by mass, from the viewpoint of providing a unique sweetness and peddy aroma. It is more preferably at least 0.3% by mass, even more preferably at least 0.4% by mass, particularly preferably at least 0.5% by mass.
- a fragrance having a fragrance like sandalwood is effective as a fragrance in combination with the valerolactone compound represented by the formula (II), and a fragrance composition having a high natural sandalwood texture can be obtained.
- fragrance having a sandalwood-like fragrance examples include the following artificial fragrances (B).
- Fragrance mainly composed of; isocampylsix hexanols; 3,7-dimethyl-7-methoxyoctane-1-ol;
- One or more compounds selected from optical isomers and the like of the compound may be mentioned.
- the valerolactone compound represented by the formula (I) and the perfume having a sandalwood-like fragrance can be arbitrarily combined.
- the mass ratio of the valerolactone compound to the fragrance (B) having a sandalwood-like scent is not particularly limited, but is preferably 1: 100000 or more from the viewpoint of providing a soft and sweet milk-like scent unique to natural sandalwood. It is more preferably at least 1: 10,000, further preferably at least 1: 1000. Further, the mass ratio is preferably 1: 100000 to 1: 1, more preferably 1: 1000 0 to 3: 7, further preferably 1: 1000 to 2: 8, and particularly preferably 1: 500 to 1: 9. , 1: 350 to 1: 9.
- the fragrance composition of the present invention can be produced.
- the fragrance composition containing the valerolactone compound can be blended or applied to various forms of aromatic products. Its application fields include, for example, household products, cosmetic products, and environmental hygiene products. Household products are products for maintaining the function or cleanliness of various articles such as dwellings and household products necessary for home life.Specifically, detergents for clothing, fabric softeners, Examples include pastes for clothing, detergents for houses, detergents for baths, detergents for dishes, bleaching agents, mold removers, waxes for floors and the like.
- the fragrance composition of the present invention can be added to these product groups in any amount, but is usually 0.001 to 2% by mass, preferably 0.01 to 1% by mass.
- Cosmetic products are products that cleanse or beautify humans. Specifically, stone products, body cleansers, hair cleansers, hair cosmetics, and cosmetics (for example, skin cosmetics, Finishing cosmetics, etc.), perfumes, colons, antiperspirants, deodorants, bath agents and the like.
- the fragrance composition of the present invention can be added to these product groups in any amount.
- the content of the fragrance composition of the present invention in the product group is 0.0001 to 50% by mass, preferably 1 to 40% by mass in the case of perfume and colon, stones other than perfume and colon, body washing
- the amount is 0.001 to 2% by mass.
- An environmental hygiene product is a product for adjusting the environment to a predetermined state or atmosphere, and in particular, a product that can control the scent drifting in the environment by applying a fragrance composition.
- the fragrance composition of the present invention can be added to these product groups in any amount.
- the content of the fragrance composition of the present invention in the product group is 0.01 to 80% by mass, preferably 0.1 to 70% by mass.
- the valerolactone compound of the present invention can be added to flavors used for beverages and foods.
- the valerolactone compound can be used as an oral product (eg, toothpaste, mouthwash, etc.) and a fragrance for tobacco.
- fragrance composition There are various ways to use products to which the fragrance composition is applied.
- a method of generating odors by applying it to a predetermined site such as perfume or cosmetics, or a rinsing agent, such as a cleaning agent How to leave an odor in the area, volatilize it in space like a fragrance
- a fragrance composition optionally containing a sandal-based synthetic fragrance and containing a valerolactone compound in an amount of 1% by mass is added to the fabric softener in an amount of 0.1% by mass, so that natural garments can be added to clothing. It brings out the soft and sweet scent peculiar to sandals and makes it possible to maintain a clear scent.
- FIG. 1 shows a 1 H-NMR (400 MHz) chart of methyl-7-methyl-1-7-1-propenylvalerolactone.
- Figure 2 shows a chart of the infrared absorption spectrum (IR) of 1-methyl-1- ⁇ -methyl-7-1-propenyl valerolactone.
- Figure 3 shows the 1 H-NMR (400 MHz) chart of ⁇ ethyl-7-methyl-7-1-propenylvalerolactone.
- Fig. 4 shows the infrared absorption spectrum (IR) of / S-ethyl-1 7-methyl-17 "-1-propenylvalerolactone.
- Example 7 [Preparation of sandalwood type fragrance composition containing valerolactone compound (I)] 2-Methyl-4-1- (2,2,3-trimethyl-3-cyclopentene-11-yl) -1-2-butene-1-ol as a fragrance substance having a fragrance of sandalwood (trade name, manufactured by Kao Corporation) : Sandal mysole core] to prepare a fragrance composition A,, B i, C i and D, respectively, at a mass ratio of fragrances A, B, C and D in Table 1 below, and use fragrance E as a comparative product. The odor was compared and evaluated by specialized panelists. As a result, as shown below, it was recognized that the blending of the valerolactone compound represented by the formula (II) further enhanced the quality of the fragrance of the sandalwood. table 1
- the sandalwood feeling of the sandal mysole core was enhanced as compared with the comparative fragrance E.
- the fragrance composition was found to have a sweetness and a natural sandalpad feel as compared with the comparative fragrance E.
- the fragrance composition C had a milky sweetness and a stronger volume than the comparative fragrance E, and was found to have the effect of enhancing the natural sandalwood likeness.
- the sweetness and natural sandalwood-like qualities were further enhanced as compared with the comparative fragrance E.
- Perfume composition D 2 the effect of Uddi feeling is enhanced as compared with the perfume E as a comparative product was observed.
- 3-Methyl-5- (2,2,3-trimethyl-13-cyclopenten-1-yl) -pentan-2-ol is used as a perfume substance having a sandalwood scent, and the mass ratio of perfume D described in Table 1 is used.
- the perfume composition D 3 were prepared and comparative evaluation of odor was the perfume E as a comparative product.
- Perfume composition D 3 are characteristic of powdery natural Sandaruu' de compared with the perfume E as a comparative product endowed became soft odor.
- Perfume composition D 4 was observed to smell with sweetness and volume compared with the perfume E as a comparative product.
- perfume substances having a sandalwood scent shown in Table 1 include 2-methyl-4- (2,2,3-trimethyl-3-cyclopentene-11-yl) -butanol, 3,3-Dimethyl-1-5- (2,2,3-trimethyl-3-cyclopentene-1-yl) -14-penten-1-ol, etc.
- Example 8 Perfume composition for perfume
- Example 8 A mixture of 99 parts by mass of the fragrance composition obtained in Example 8 (Table 2) and 1 part by mass of 7-ethyl-butyl- ⁇ 5-valerolactone has no sweetness and has a strong Costas odor. There was no softness like sandals, and the smell was unbalanced. Comparative Example 2
- Example 9 Frarance composition of detergent for clothing
- Example 15 (Fragrance composition for incense sticks)
- Example 16 0.19 parts by mass of the fragrance composition of Example 16 (flavor) is blended with 99.9 parts by mass of a milk coffee composition having the composition shown in Table 11 below, and has a plump sweetness and moderate astringency. A milk coffee composition was obtained.
- the valerolactone compound can be optionally combined with various flavors.
- the fragrance composition containing the valerolactone compound can be blended or applied to various forms of aromatic products in the fields of household products, fragrance products, environmental hygiene products, and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nutrition Science (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Fats And Perfumes (AREA)
- Cosmetics (AREA)
- Pyrane Compounds (AREA)
- General Preparation And Processing Of Foods (AREA)
- Seasonings (AREA)
- Non-Alcoholic Beverages (AREA)
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2003272896A AU2003272896A1 (en) | 2002-09-27 | 2003-09-26 | Valerolactone compounds and perfume composition |
| EP03753957A EP1555261B1 (en) | 2002-09-27 | 2003-09-26 | Valerolactone compounds and perfume composition |
| DE60334012T DE60334012D1 (de) | 2002-09-27 | 2003-09-26 | Valerolactonverbindungen und parf mzusammensetzung |
| US10/529,290 US7491833B2 (en) | 2002-09-27 | 2003-09-26 | Valerolactone compounds and perfume composition |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002282675 | 2002-09-27 | ||
| JP2002-282675 | 2002-09-27 | ||
| JP2002308952 | 2002-10-23 | ||
| JP2002-308952 | 2002-10-23 | ||
| JP2003-323125 | 2003-09-16 | ||
| JP2003323125A JP4323271B2 (ja) | 2002-09-27 | 2003-09-16 | バレロラクトン化合物及び香料組成物 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004029033A1 true WO2004029033A1 (ja) | 2004-04-08 |
Family
ID=32045736
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2003/012341 WO2004029033A1 (ja) | 2002-09-27 | 2003-09-26 | バレロラクトン化合物及び香料組成物 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US7491833B2 (ja) |
| EP (1) | EP1555261B1 (ja) |
| JP (1) | JP4323271B2 (ja) |
| CN (1) | CN100400533C (ja) |
| AU (1) | AU2003272896A1 (ja) |
| DE (1) | DE60334012D1 (ja) |
| WO (1) | WO2004029033A1 (ja) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7964220B2 (en) | 2002-02-04 | 2011-06-21 | ALFAMA—Investigação e Desenvolvimento de Produtos Farmacêuticos, Lda. | Method for treating a mammal by administration of a compound having the ability to release CO |
| US7968605B2 (en) | 2002-02-04 | 2011-06-28 | ALFAMA—Investigação e Desenvolvimento de Produtos Farmacêuticos, Lda. | Methods for treating inflammatory disease by administering aldehydes and derivatives thereof |
| US7989650B2 (en) | 2002-11-20 | 2011-08-02 | Hemocorm Limited | Therapeutic delivery of carbon monoxide to extracorporeal and isolated organs |
| US8236339B2 (en) | 2001-05-15 | 2012-08-07 | Hemocorm Limited | Therapeutic delivery of carbon monoxide |
| US8389572B2 (en) | 2006-01-24 | 2013-03-05 | Hemocorm Limited | Therapeutic delivery of carbon monoxide |
| US9062089B2 (en) | 2011-07-21 | 2015-06-23 | Alfama, Inc. | Ruthenium carbon monoxide releasing molecules and uses thereof |
| US9163044B2 (en) | 2011-04-19 | 2015-10-20 | Alfama, Inc. | Carbon monoxide releasing molecules and uses thereof |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5137249B2 (ja) * | 2002-09-27 | 2013-02-06 | 花王株式会社 | 香料組成物 |
| WO2010052636A1 (en) * | 2008-11-07 | 2010-05-14 | Firmenich Sa | Perfuming ingredients of the floral and/or anis type |
| WO2014169003A2 (en) * | 2013-04-09 | 2014-10-16 | Arch Chemicals, Inc. | Multi-functional composition for cosmetic formulations |
| WO2018050658A1 (en) * | 2016-09-16 | 2018-03-22 | Firmenich Sa | Perfuming composition |
| WO2018050655A1 (en) * | 2016-09-16 | 2018-03-22 | Firmenich Sa | Perfuming composition |
| WO2022085515A1 (ja) * | 2020-10-20 | 2022-04-28 | アサヒグループホールディングス株式会社 | ビールテイスト飲料用香味改善剤及びビールテイスト飲料 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3825572A (en) | 1972-05-31 | 1974-07-23 | Sun Research Development | Epimeric mixtures of the lactone 3,6-dihydro-4,5-dimethyl-5-phenyl-alpha-pyrone |
| JPS50101563A (ja) * | 1974-01-24 | 1975-08-12 | ||
| GB2024208A (en) * | 1978-06-26 | 1980-01-09 | Dragoco Gerberding Co Gmbh | Polysubstituted cyclopentene derivatives |
| US4288350A (en) | 1976-12-23 | 1981-09-08 | Polak's Frutal Works B.V. | Perfume compositions containing dialkyl delta-lactones |
| JPS60109537A (ja) * | 1983-11-18 | 1985-06-15 | T Hasegawa Co Ltd | シクロペンテン誘導体 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3380457A (en) | 1965-08-02 | 1968-04-30 | Reynolds Tobacco Co R | Tobacco product |
| EP0054316A1 (fr) | 1980-12-09 | 1982-06-23 | Firmenich Sa | Nouveaux composés lactoniques, procédé pour leur préparation et leur utilisation à titre d'ingrédients parfumants ou aromatisants |
| JPS625123A (ja) | 1985-07-01 | 1987-01-12 | Hitachi Ltd | ナビゲ−シヨンシステム |
| US5231192A (en) * | 1989-02-24 | 1993-07-27 | Henkel Corporation | Process for the preparation of alpha-alkyl lactones |
| EP0513627B1 (de) | 1991-05-15 | 1996-02-28 | Givaudan-Roure (International) S.A. | Tetrahydro-alpha-pyronderivat, Verfahren zu seiner Herstellung und dieses enthaltende Riech- und/oder Geschmackstoffkompositionen |
| JPH07502285A (ja) | 1992-09-29 | 1995-03-09 | ジボーダン − ルール (アンテルナシヨナル)ソシエテ アノニム | ラクトン |
| US6117835A (en) | 1998-04-23 | 2000-09-12 | International Flavors & Fragrances Inc | Process for preparing saturated lactones, products produced therefrom and organoleptic uses of said products |
-
2003
- 2003-09-16 JP JP2003323125A patent/JP4323271B2/ja not_active Expired - Fee Related
- 2003-09-26 AU AU2003272896A patent/AU2003272896A1/en not_active Abandoned
- 2003-09-26 EP EP03753957A patent/EP1555261B1/en not_active Expired - Lifetime
- 2003-09-26 WO PCT/JP2003/012341 patent/WO2004029033A1/ja active Application Filing
- 2003-09-26 CN CNB038230852A patent/CN100400533C/zh not_active Expired - Fee Related
- 2003-09-26 US US10/529,290 patent/US7491833B2/en active Active
- 2003-09-26 DE DE60334012T patent/DE60334012D1/de not_active Expired - Lifetime
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3825572A (en) | 1972-05-31 | 1974-07-23 | Sun Research Development | Epimeric mixtures of the lactone 3,6-dihydro-4,5-dimethyl-5-phenyl-alpha-pyrone |
| JPS50101563A (ja) * | 1974-01-24 | 1975-08-12 | ||
| US4288350A (en) | 1976-12-23 | 1981-09-08 | Polak's Frutal Works B.V. | Perfume compositions containing dialkyl delta-lactones |
| GB2024208A (en) * | 1978-06-26 | 1980-01-09 | Dragoco Gerberding Co Gmbh | Polysubstituted cyclopentene derivatives |
| JPS60109537A (ja) * | 1983-11-18 | 1985-06-15 | T Hasegawa Co Ltd | シクロペンテン誘導体 |
Non-Patent Citations (5)
| Title |
|---|
| IHARA, M. ET AL., J. ORG. CHEM., vol. 61, 1996, pages 667 |
| IHARA, MASATAKA, ET AL: "Synthesis of Six-Membered Compounds by Environmentally Friendly Cyclization Using Indirect Electrolysis.", J. ORG. CHEM., vol. 61, 1996, pages 677 - 684, XP002974955 * |
| See also references of EP1555261A4 |
| TSUNOI, S ET AL: "New Strategies in Carbonylation Chemistry : The Synthesis of delta-lactones fromsatured alcohols and CO.", J. AM. CHEM.SOC., vol. 120, 1998, pages 8692 - 8701, XP002974954 * |
| TSUNOI, S. ET AL., J. AM. CHEM. SOC., vol. 120, 1998, pages 8692 |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8236339B2 (en) | 2001-05-15 | 2012-08-07 | Hemocorm Limited | Therapeutic delivery of carbon monoxide |
| US7964220B2 (en) | 2002-02-04 | 2011-06-21 | ALFAMA—Investigação e Desenvolvimento de Produtos Farmacêuticos, Lda. | Method for treating a mammal by administration of a compound having the ability to release CO |
| US7968605B2 (en) | 2002-02-04 | 2011-06-28 | ALFAMA—Investigação e Desenvolvimento de Produtos Farmacêuticos, Lda. | Methods for treating inflammatory disease by administering aldehydes and derivatives thereof |
| US9023402B2 (en) | 2002-02-04 | 2015-05-05 | ALFAMA—Investigação e Desenvolvimento de Produtos Farmacêuticos, Lda. | Method for treating a mammal by administration of a compound having the ability to release CO |
| US7989650B2 (en) | 2002-11-20 | 2011-08-02 | Hemocorm Limited | Therapeutic delivery of carbon monoxide to extracorporeal and isolated organs |
| US8389572B2 (en) | 2006-01-24 | 2013-03-05 | Hemocorm Limited | Therapeutic delivery of carbon monoxide |
| US9163044B2 (en) | 2011-04-19 | 2015-10-20 | Alfama, Inc. | Carbon monoxide releasing molecules and uses thereof |
| US9062089B2 (en) | 2011-07-21 | 2015-06-23 | Alfama, Inc. | Ruthenium carbon monoxide releasing molecules and uses thereof |
| US9611286B2 (en) | 2011-07-21 | 2017-04-04 | Alfama, Inc. | Ruthenium carbon monoxide releasing molecules and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1555261A4 (en) | 2007-05-02 |
| EP1555261B1 (en) | 2010-09-01 |
| AU2003272896A1 (en) | 2004-04-19 |
| JP2004161746A (ja) | 2004-06-10 |
| DE60334012D1 (de) | 2010-10-14 |
| US7491833B2 (en) | 2009-02-17 |
| US20060258559A1 (en) | 2006-11-16 |
| CN1684949A (zh) | 2005-10-19 |
| CN100400533C (zh) | 2008-07-09 |
| JP4323271B2 (ja) | 2009-09-02 |
| EP1555261A1 (en) | 2005-07-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109310602B (zh) | 香料材料 | |
| CN109310601B (zh) | 香料材料 | |
| WO2004029033A1 (ja) | バレロラクトン化合物及び香料組成物 | |
| JP5137249B2 (ja) | 香料組成物 | |
| JP4589967B2 (ja) | 香料組成物 | |
| JP6202679B2 (ja) | 4,8−ジメチル−4,9−デカジエンニトリル | |
| JP2008308456A (ja) | 新規香料成分及び香料組成物 | |
| WO2019194186A1 (ja) | ムスク様組成物 | |
| JP5501524B1 (ja) | ニトリル化合物 | |
| JP6054157B2 (ja) | 香料組成物 | |
| MX2014008704A (es) | Compuestos organolepticos novedosos. | |
| JP5808704B2 (ja) | ジメチルシクロヘキセニルアルケノン | |
| CN102007204B (zh) | 作为加香成分的酮 | |
| JP4041238B2 (ja) | 香料中で、およびフレーバリング成分としての2,5,6−トリメチル−2−ヘプタノールの使用 | |
| WO2007013394A1 (ja) | 香料組成物 | |
| JP5912943B2 (ja) | 3,7−ジメチル−2−メチレン−6−オクテンニトリル | |
| ES2351210T3 (es) | Compuestos de valerolactona y composición de perfume. | |
| JP5930467B2 (ja) | ニトリル化合物 | |
| JP6137681B2 (ja) | ニトリル化合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS KE KG KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 483/CHENP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006258559 Country of ref document: US Ref document number: 10529290 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20038230852 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003753957 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003753957 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 10529290 Country of ref document: US |