RU2696071C2 - Получение плюрипотентных клеток de novo - Google Patents
Получение плюрипотентных клеток de novo Download PDFInfo
- Publication number
- RU2696071C2 RU2696071C2 RU2014147093A RU2014147093A RU2696071C2 RU 2696071 C2 RU2696071 C2 RU 2696071C2 RU 2014147093 A RU2014147093 A RU 2014147093A RU 2014147093 A RU2014147093 A RU 2014147093A RU 2696071 C2 RU2696071 C2 RU 2696071C2
- Authority
- RU
- Russia
- Prior art keywords
- cells
- cell
- pluripotent
- stap
- stem
- Prior art date
Links
- 210000004027 cell Anatomy 0.000 claims abstract description 1395
- 238000000034 method Methods 0.000 claims abstract description 253
- 210000001082 somatic cell Anatomy 0.000 claims abstract description 72
- 101150086694 SLC22A3 gene Proteins 0.000 claims abstract description 21
- 239000002243 precursor Substances 0.000 claims abstract description 12
- 101150033839 4 gene Proteins 0.000 claims abstract 2
- 230000035882 stress Effects 0.000 claims description 126
- 210000001519 tissue Anatomy 0.000 claims description 115
- 210000000130 stem cell Anatomy 0.000 claims description 110
- 108090000623 proteins and genes Proteins 0.000 claims description 68
- 210000000805 cytoplasm Anatomy 0.000 claims description 47
- 238000000338 in vitro Methods 0.000 claims description 28
- 210000004962 mammalian cell Anatomy 0.000 claims description 18
- -1 transcript Proteins 0.000 claims description 14
- 210000002308 embryonic cell Anatomy 0.000 claims description 7
- 230000007910 cell fusion Effects 0.000 claims description 4
- 210000004700 fetal blood Anatomy 0.000 claims description 4
- 210000005260 human cell Anatomy 0.000 claims description 4
- 102000007999 Nuclear Proteins Human genes 0.000 claims description 3
- 108010089610 Nuclear Proteins Proteins 0.000 claims description 3
- 230000008650 pH stress Effects 0.000 claims 1
- 239000000126 substance Substances 0.000 abstract description 30
- 238000004519 manufacturing process Methods 0.000 abstract description 29
- 230000000694 effects Effects 0.000 abstract description 28
- 210000001161 mammalian embryo Anatomy 0.000 abstract description 10
- 108020004414 DNA Proteins 0.000 description 108
- 101000738771 Homo sapiens Receptor-type tyrosine-protein phosphatase C Proteins 0.000 description 95
- 102100037422 Receptor-type tyrosine-protein phosphatase C Human genes 0.000 description 95
- 230000014509 gene expression Effects 0.000 description 92
- 230000004069 differentiation Effects 0.000 description 69
- 241000699670 Mus sp. Species 0.000 description 64
- 238000004458 analytical method Methods 0.000 description 63
- 239000002609 medium Substances 0.000 description 63
- 210000003470 mitochondria Anatomy 0.000 description 51
- 239000003795 chemical substances by application Substances 0.000 description 48
- 101710126211 POU domain, class 5, transcription factor 1 Proteins 0.000 description 44
- 102100035423 POU domain, class 5, transcription factor 1 Human genes 0.000 description 42
- 210000002459 blastocyst Anatomy 0.000 description 42
- 239000000275 Adrenocorticotropic Hormone Substances 0.000 description 36
- 102400000739 Corticotropin Human genes 0.000 description 36
- 101800000414 Corticotropin Proteins 0.000 description 36
- IDLFZVILOHSSID-OVLDLUHVSA-N corticotropin Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)NC(=O)[C@@H](N)CO)C1=CC=C(O)C=C1 IDLFZVILOHSSID-OVLDLUHVSA-N 0.000 description 36
- 229960000258 corticotropin Drugs 0.000 description 36
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 35
- 210000001778 pluripotent stem cell Anatomy 0.000 description 34
- 239000000203 mixture Substances 0.000 description 32
- 238000011282 treatment Methods 0.000 description 32
- 230000001973 epigenetic effect Effects 0.000 description 30
- 238000011161 development Methods 0.000 description 29
- 230000008672 reprogramming Effects 0.000 description 29
- 238000012258 culturing Methods 0.000 description 28
- 230000018109 developmental process Effects 0.000 description 28
- 210000001671 embryonic stem cell Anatomy 0.000 description 28
- 210000002257 embryonic structure Anatomy 0.000 description 26
- 230000015572 biosynthetic process Effects 0.000 description 25
- 241000699666 Mus <mouse, genus> Species 0.000 description 24
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 23
- 230000000670 limiting effect Effects 0.000 description 23
- 239000003550 marker Substances 0.000 description 23
- 108010075210 streptolysin O Proteins 0.000 description 23
- 241001465754 Metazoa Species 0.000 description 22
- 230000008859 change Effects 0.000 description 21
- 210000001654 germ layer Anatomy 0.000 description 21
- 206010020649 Hyperkeratosis Diseases 0.000 description 20
- 239000000243 solution Substances 0.000 description 20
- 230000007423 decrease Effects 0.000 description 19
- 238000001727 in vivo Methods 0.000 description 19
- 241000196324 Embryophyta Species 0.000 description 18
- 230000006378 damage Effects 0.000 description 18
- 239000001963 growth medium Substances 0.000 description 18
- 238000002347 injection Methods 0.000 description 18
- 239000007924 injection Substances 0.000 description 18
- 210000004698 lymphocyte Anatomy 0.000 description 18
- 230000035899 viability Effects 0.000 description 18
- 239000002771 cell marker Substances 0.000 description 17
- 201000010099 disease Diseases 0.000 description 17
- 208000035199 Tetraploidy Diseases 0.000 description 16
- 238000004113 cell culture Methods 0.000 description 16
- 230000003169 placental effect Effects 0.000 description 16
- 102100028072 Fibroblast growth factor 4 Human genes 0.000 description 15
- 101001060274 Homo sapiens Fibroblast growth factor 4 Proteins 0.000 description 15
- 238000005516 engineering process Methods 0.000 description 15
- 210000004379 membrane Anatomy 0.000 description 15
- 239000012528 membrane Substances 0.000 description 15
- 102000000905 Cadherin Human genes 0.000 description 14
- 108050007957 Cadherin Proteins 0.000 description 14
- 230000011987 methylation Effects 0.000 description 14
- 238000007069 methylation reaction Methods 0.000 description 14
- 206010021143 Hypoxia Diseases 0.000 description 13
- 102000004142 Trypsin Human genes 0.000 description 13
- 108090000631 Trypsin Proteins 0.000 description 13
- 230000001413 cellular effect Effects 0.000 description 13
- 238000006243 chemical reaction Methods 0.000 description 13
- 230000006870 function Effects 0.000 description 13
- 210000004602 germ cell Anatomy 0.000 description 13
- 230000001965 increasing effect Effects 0.000 description 13
- 235000018102 proteins Nutrition 0.000 description 13
- 102000004169 proteins and genes Human genes 0.000 description 13
- 230000000392 somatic effect Effects 0.000 description 13
- 239000012588 trypsin Substances 0.000 description 13
- 241001529936 Murinae Species 0.000 description 12
- 238000002659 cell therapy Methods 0.000 description 12
- 239000002085 irritant Substances 0.000 description 12
- 231100000021 irritant Toxicity 0.000 description 12
- 210000003205 muscle Anatomy 0.000 description 12
- 208000024891 symptom Diseases 0.000 description 12
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 11
- 241000124008 Mammalia Species 0.000 description 11
- 210000000170 cell membrane Anatomy 0.000 description 11
- 239000012894 fetal calf serum Substances 0.000 description 11
- 108090000765 processed proteins & peptides Proteins 0.000 description 11
- 230000008707 rearrangement Effects 0.000 description 11
- 210000000952 spleen Anatomy 0.000 description 11
- 208000002720 Malnutrition Diseases 0.000 description 10
- 238000003556 assay Methods 0.000 description 10
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 10
- 210000004369 blood Anatomy 0.000 description 10
- 239000008280 blood Substances 0.000 description 10
- 150000001875 compounds Chemical class 0.000 description 10
- 230000007246 mechanism Effects 0.000 description 10
- 150000007523 nucleic acids Chemical class 0.000 description 10
- 229910052760 oxygen Inorganic materials 0.000 description 10
- 239000001301 oxygen Substances 0.000 description 10
- 230000004083 survival effect Effects 0.000 description 10
- 208000011580 syndromic disease Diseases 0.000 description 10
- 230000007067 DNA methylation Effects 0.000 description 9
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 9
- 101100247004 Rattus norvegicus Qsox1 gene Proteins 0.000 description 9
- 210000001185 bone marrow Anatomy 0.000 description 9
- 210000004556 brain Anatomy 0.000 description 9
- 230000007812 deficiency Effects 0.000 description 9
- 230000007613 environmental effect Effects 0.000 description 9
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 9
- 210000004185 liver Anatomy 0.000 description 9
- 210000002894 multi-fate stem cell Anatomy 0.000 description 9
- 230000008569 process Effects 0.000 description 9
- 102000004196 processed proteins & peptides Human genes 0.000 description 9
- 230000002829 reductive effect Effects 0.000 description 9
- 230000033458 reproduction Effects 0.000 description 9
- LAQPKDLYOBZWBT-NYLDSJSYSA-N (2s,4s,5r,6r)-5-acetamido-2-{[(2s,3r,4s,5s,6r)-2-{[(2r,3r,4r,5r)-5-acetamido-1,2-dihydroxy-6-oxo-4-{[(2s,3s,4r,5s,6s)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}hexan-3-yl]oxy}-3,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy}-4-hydroxy-6-[(1r,2r)-1,2,3-trihydrox Chemical compound O[C@H]1[C@H](O)[C@H](O)[C@H](C)O[C@H]1O[C@H]([C@@H](NC(C)=O)C=O)[C@@H]([C@H](O)CO)O[C@H]1[C@H](O)[C@@H](O[C@]2(O[C@H]([C@H](NC(C)=O)[C@@H](O)C2)[C@H](O)[C@H](O)CO)C(O)=O)[C@@H](O)[C@@H](CO)O1 LAQPKDLYOBZWBT-NYLDSJSYSA-N 0.000 description 8
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 8
- 108700021430 Kruppel-Like Factor 4 Proteins 0.000 description 8
- 101710150336 Protein Rex Proteins 0.000 description 8
- 230000001747 exhibiting effect Effects 0.000 description 8
- 230000012010 growth Effects 0.000 description 8
- 208000014674 injury Diseases 0.000 description 8
- 210000004072 lung Anatomy 0.000 description 8
- 239000000463 material Substances 0.000 description 8
- 102000039446 nucleic acids Human genes 0.000 description 8
- 108020004707 nucleic acids Proteins 0.000 description 8
- 235000018343 nutrient deficiency Nutrition 0.000 description 8
- 230000005855 radiation Effects 0.000 description 8
- 239000003642 reactive oxygen metabolite Substances 0.000 description 8
- 238000003757 reverse transcription PCR Methods 0.000 description 8
- 210000003491 skin Anatomy 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- 238000001369 bisulfite sequencing Methods 0.000 description 7
- 239000003814 drug Substances 0.000 description 7
- 239000012634 fragment Substances 0.000 description 7
- 230000006698 induction Effects 0.000 description 7
- 210000003716 mesoderm Anatomy 0.000 description 7
- 230000035755 proliferation Effects 0.000 description 7
- 230000004044 response Effects 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 208000002491 severe combined immunodeficiency Diseases 0.000 description 7
- 206010063601 Exposure to extreme temperature Diseases 0.000 description 6
- 238000011579 SCID mouse model Methods 0.000 description 6
- 206010043276 Teratoma Diseases 0.000 description 6
- 230000004913 activation Effects 0.000 description 6
- 210000004102 animal cell Anatomy 0.000 description 6
- 238000010171 animal model Methods 0.000 description 6
- 230000032459 dedifferentiation Effects 0.000 description 6
- 208000035475 disorder Diseases 0.000 description 6
- 238000010494 dissociation reaction Methods 0.000 description 6
- 230000005593 dissociations Effects 0.000 description 6
- 239000003102 growth factor Substances 0.000 description 6
- 238000012744 immunostaining Methods 0.000 description 6
- 230000002438 mitochondrial effect Effects 0.000 description 6
- 210000001178 neural stem cell Anatomy 0.000 description 6
- 210000004940 nucleus Anatomy 0.000 description 6
- 210000002826 placenta Anatomy 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 230000001225 therapeutic effect Effects 0.000 description 6
- 230000008733 trauma Effects 0.000 description 6
- 210000002993 trophoblast Anatomy 0.000 description 6
- 210000004881 tumor cell Anatomy 0.000 description 6
- 102000007469 Actins Human genes 0.000 description 5
- 108010085238 Actins Proteins 0.000 description 5
- 101150072501 Klf2 gene Proteins 0.000 description 5
- 108091023040 Transcription factor Proteins 0.000 description 5
- 102000040945 Transcription factor Human genes 0.000 description 5
- 210000004504 adult stem cell Anatomy 0.000 description 5
- 230000000735 allogeneic effect Effects 0.000 description 5
- 230000005540 biological transmission Effects 0.000 description 5
- 230000005779 cell damage Effects 0.000 description 5
- 230000024245 cell differentiation Effects 0.000 description 5
- 208000037887 cell injury Diseases 0.000 description 5
- 210000000349 chromosome Anatomy 0.000 description 5
- 230000017858 demethylation Effects 0.000 description 5
- 238000010520 demethylation reaction Methods 0.000 description 5
- 239000000975 dye Substances 0.000 description 5
- 210000001900 endoderm Anatomy 0.000 description 5
- 230000004049 epigenetic modification Effects 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- 235000019688 fish Nutrition 0.000 description 5
- 238000003205 genotyping method Methods 0.000 description 5
- 239000008187 granular material Substances 0.000 description 5
- 238000002513 implantation Methods 0.000 description 5
- 230000002779 inactivation Effects 0.000 description 5
- 230000001939 inductive effect Effects 0.000 description 5
- 235000000824 malnutrition Nutrition 0.000 description 5
- 230000001071 malnutrition Effects 0.000 description 5
- 239000013642 negative control Substances 0.000 description 5
- 208000015380 nutritional deficiency disease Diseases 0.000 description 5
- 210000003463 organelle Anatomy 0.000 description 5
- 230000003204 osmotic effect Effects 0.000 description 5
- 210000005059 placental tissue Anatomy 0.000 description 5
- 230000008929 regeneration Effects 0.000 description 5
- 238000011069 regeneration method Methods 0.000 description 5
- 230000008439 repair process Effects 0.000 description 5
- 238000011160 research Methods 0.000 description 5
- 230000002441 reversible effect Effects 0.000 description 5
- 230000000638 stimulation Effects 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- 108091035539 telomere Proteins 0.000 description 5
- 210000003411 telomere Anatomy 0.000 description 5
- 102000055501 telomere Human genes 0.000 description 5
- 238000002560 therapeutic procedure Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 206010068051 Chimerism Diseases 0.000 description 4
- 230000008836 DNA modification Effects 0.000 description 4
- 230000033616 DNA repair Effects 0.000 description 4
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 4
- 108091092584 GDNA Proteins 0.000 description 4
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 4
- 239000012981 Hank's balanced salt solution Substances 0.000 description 4
- 210000001744 T-lymphocyte Anatomy 0.000 description 4
- 101150111019 Tbx3 gene Proteins 0.000 description 4
- 229920004890 Triton X-100 Polymers 0.000 description 4
- 239000013504 Triton X-100 Substances 0.000 description 4
- 230000002378 acidificating effect Effects 0.000 description 4
- 102000013529 alpha-Fetoproteins Human genes 0.000 description 4
- 108010026331 alpha-Fetoproteins Proteins 0.000 description 4
- 235000001014 amino acid Nutrition 0.000 description 4
- 150000001413 amino acids Chemical class 0.000 description 4
- 239000000427 antigen Substances 0.000 description 4
- 108091007433 antigens Proteins 0.000 description 4
- 102000036639 antigens Human genes 0.000 description 4
- 238000000149 argon plasma sintering Methods 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 201000011510 cancer Diseases 0.000 description 4
- 239000000969 carrier Substances 0.000 description 4
- 238000012512 characterization method Methods 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 210000001612 chondrocyte Anatomy 0.000 description 4
- 230000004064 dysfunction Effects 0.000 description 4
- 210000003981 ectoderm Anatomy 0.000 description 4
- 230000006353 environmental stress Effects 0.000 description 4
- 210000003754 fetus Anatomy 0.000 description 4
- 210000002950 fibroblast Anatomy 0.000 description 4
- 238000007710 freezing Methods 0.000 description 4
- 230000008014 freezing Effects 0.000 description 4
- 230000002068 genetic effect Effects 0.000 description 4
- 239000007943 implant Substances 0.000 description 4
- 210000002660 insulin-secreting cell Anatomy 0.000 description 4
- 238000002372 labelling Methods 0.000 description 4
- 208000032839 leukemia Diseases 0.000 description 4
- 210000000265 leukocyte Anatomy 0.000 description 4
- 238000004020 luminiscence type Methods 0.000 description 4
- 230000031864 metaphase Effects 0.000 description 4
- 239000003068 molecular probe Substances 0.000 description 4
- 230000001537 neural effect Effects 0.000 description 4
- 230000036961 partial effect Effects 0.000 description 4
- 239000008194 pharmaceutical composition Substances 0.000 description 4
- 230000009894 physiological stress Effects 0.000 description 4
- 238000007747 plating Methods 0.000 description 4
- 239000011148 porous material Substances 0.000 description 4
- 230000001902 propagating effect Effects 0.000 description 4
- 230000001105 regulatory effect Effects 0.000 description 4
- 230000035939 shock Effects 0.000 description 4
- 238000010186 staining Methods 0.000 description 4
- 238000012546 transfer Methods 0.000 description 4
- 230000001960 triggered effect Effects 0.000 description 4
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 3
- 241000282472 Canis lupus familiaris Species 0.000 description 3
- 238000001712 DNA sequencing Methods 0.000 description 3
- 108010010803 Gelatin Proteins 0.000 description 3
- 108700039691 Genetic Promoter Regions Proteins 0.000 description 3
- 102100031181 Glyceraldehyde-3-phosphate dehydrogenase Human genes 0.000 description 3
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 description 3
- 102000004269 Granulocyte Colony-Stimulating Factor Human genes 0.000 description 3
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 3
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 3
- 102000006947 Histones Human genes 0.000 description 3
- 108010033040 Histones Proteins 0.000 description 3
- 102000007651 Macrophage Colony-Stimulating Factor Human genes 0.000 description 3
- 108010046938 Macrophage Colony-Stimulating Factor Proteins 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 241000283973 Oryctolagus cuniculus Species 0.000 description 3
- 229930040373 Paraformaldehyde Natural products 0.000 description 3
- 108010056995 Perforin Proteins 0.000 description 3
- 102000004503 Perforin Human genes 0.000 description 3
- 241000288906 Primates Species 0.000 description 3
- 238000011529 RT qPCR Methods 0.000 description 3
- 101100127230 Rattus norvegicus Khdc3 gene Proteins 0.000 description 3
- 101150037203 Sox2 gene Proteins 0.000 description 3
- 108700042077 T-Cell Receptor beta Genes Proteins 0.000 description 3
- 102000004243 Tubulin Human genes 0.000 description 3
- 108090000704 Tubulin Proteins 0.000 description 3
- 208000027418 Wounds and injury Diseases 0.000 description 3
- 210000001766 X chromosome Anatomy 0.000 description 3
- 101100425550 Xenopus laevis esg1 gene Proteins 0.000 description 3
- 230000021736 acetylation Effects 0.000 description 3
- 238000006640 acetylation reaction Methods 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 239000000853 adhesive Substances 0.000 description 3
- 230000001070 adhesive effect Effects 0.000 description 3
- 210000000577 adipose tissue Anatomy 0.000 description 3
- 238000003149 assay kit Methods 0.000 description 3
- 210000004204 blood vessel Anatomy 0.000 description 3
- 210000004413 cardiac myocyte Anatomy 0.000 description 3
- 238000011109 contamination Methods 0.000 description 3
- 229920001577 copolymer Polymers 0.000 description 3
- 230000000875 corresponding effect Effects 0.000 description 3
- 210000004748 cultured cell Anatomy 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 230000019975 dosage compensation by inactivation of X chromosome Effects 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 235000013399 edible fruits Nutrition 0.000 description 3
- 235000013601 eggs Nutrition 0.000 description 3
- 230000013020 embryo development Effects 0.000 description 3
- 238000010195 expression analysis Methods 0.000 description 3
- 230000006355 external stress Effects 0.000 description 3
- 230000001605 fetal effect Effects 0.000 description 3
- 238000000684 flow cytometry Methods 0.000 description 3
- 239000008273 gelatin Substances 0.000 description 3
- 229920000159 gelatin Polymers 0.000 description 3
- 235000019322 gelatine Nutrition 0.000 description 3
- 235000011852 gelatine desserts Nutrition 0.000 description 3
- 108020004445 glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 3
- 238000000227 grinding Methods 0.000 description 3
- 238000000265 homogenisation Methods 0.000 description 3
- 230000007954 hypoxia Effects 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 239000003112 inhibitor Substances 0.000 description 3
- 238000007912 intraperitoneal administration Methods 0.000 description 3
- 238000002955 isolation Methods 0.000 description 3
- 201000005992 juvenile myelomonocytic leukemia Diseases 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 230000003211 malignant effect Effects 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 210000002569 neuron Anatomy 0.000 description 3
- 229920002866 paraformaldehyde Polymers 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 238000003752 polymerase chain reaction Methods 0.000 description 3
- 230000002062 proliferating effect Effects 0.000 description 3
- 238000001959 radiotherapy Methods 0.000 description 3
- 238000010839 reverse transcription Methods 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 210000001988 somatic stem cell Anatomy 0.000 description 3
- 210000004989 spleen cell Anatomy 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- 239000003053 toxin Substances 0.000 description 3
- 231100000765 toxin Toxicity 0.000 description 3
- 230000009466 transformation Effects 0.000 description 3
- 230000001052 transient effect Effects 0.000 description 3
- 238000002054 transplantation Methods 0.000 description 3
- 230000001573 trophoblastic effect Effects 0.000 description 3
- NNJPGOLRFBJNIW-HNNXBMFYSA-N (-)-demecolcine Chemical compound C1=C(OC)C(=O)C=C2[C@@H](NC)CCC3=CC(OC)=C(OC)C(OC)=C3C2=C1 NNJPGOLRFBJNIW-HNNXBMFYSA-N 0.000 description 2
- XYJODUBPWNZLML-UHFFFAOYSA-N 5-ethyl-6-phenyl-6h-phenanthridine-3,8-diamine Chemical compound C12=CC(N)=CC=C2C2=CC=C(N)C=C2N(CC)C1C1=CC=CC=C1 XYJODUBPWNZLML-UHFFFAOYSA-N 0.000 description 2
- 108010059616 Activins Proteins 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 241000193830 Bacillus <bacterium> Species 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 108010083123 CDX2 Transcription Factor Proteins 0.000 description 2
- 102000006277 CDX2 Transcription Factor Human genes 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- 208000018380 Chemical injury Diseases 0.000 description 2
- 206010010356 Congenital anomaly Diseases 0.000 description 2
- NNJPGOLRFBJNIW-UHFFFAOYSA-N Demecolcine Natural products C1=C(OC)C(=O)C=C2C(NC)CCC3=CC(OC)=C(OC)C(OC)=C3C2=C1 NNJPGOLRFBJNIW-UHFFFAOYSA-N 0.000 description 2
- 101100010303 Drosophila melanogaster PolG1 gene Proteins 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000009024 Epidermal Growth Factor Human genes 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- 102000003974 Fibroblast growth factor 2 Human genes 0.000 description 2
- 102100024785 Fibroblast growth factor 2 Human genes 0.000 description 2
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 2
- 102100039289 Glial fibrillary acidic protein Human genes 0.000 description 2
- 101710193519 Glial fibrillary acidic protein Proteins 0.000 description 2
- 102400000321 Glucagon Human genes 0.000 description 2
- 108060003199 Glucagon Proteins 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 102100026818 Inhibin beta E chain Human genes 0.000 description 2
- 102100032832 Integrin alpha-7 Human genes 0.000 description 2
- 102100023974 Keratin, type II cytoskeletal 7 Human genes 0.000 description 2
- 108010070507 Keratin-7 Proteins 0.000 description 2
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 2
- 241000282553 Macaca Species 0.000 description 2
- 108020005196 Mitochondrial DNA Proteins 0.000 description 2
- 208000008955 Mucolipidoses Diseases 0.000 description 2
- 206010072928 Mucolipidosis type II Diseases 0.000 description 2
- 208000002678 Mucopolysaccharidoses Diseases 0.000 description 2
- 208000037538 Myelomonocytic Juvenile Leukemia Diseases 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 2
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 description 2
- 101150078890 POLG gene Proteins 0.000 description 2
- 208000000733 Paroxysmal Hemoglobinuria Diseases 0.000 description 2
- 102100036050 Phosphatidylinositol N-acetylglucosaminyltransferase subunit A Human genes 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- 241000283984 Rodentia Species 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 208000006011 Stroke Diseases 0.000 description 2
- 108091008874 T cell receptors Proteins 0.000 description 2
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 2
- 101150080431 Tfam gene Proteins 0.000 description 2
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 2
- 239000003929 acidic solution Substances 0.000 description 2
- 239000000488 activin Substances 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 230000003321 amplification Effects 0.000 description 2
- 210000000612 antigen-presenting cell Anatomy 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 238000011130 autologous cell therapy Methods 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- 210000000227 basophil cell of anterior lobe of hypophysis Anatomy 0.000 description 2
- 239000012620 biological material Substances 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 210000001772 blood platelet Anatomy 0.000 description 2
- 229940022399 cancer vaccine Drugs 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 210000003855 cell nucleus Anatomy 0.000 description 2
- 230000036755 cellular response Effects 0.000 description 2
- 230000007960 cellular response to stress Effects 0.000 description 2
- 210000003169 central nervous system Anatomy 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical compound NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 238000009795 derivation Methods 0.000 description 2
- 238000003745 diagnosis Methods 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 2
- 230000008995 epigenetic change Effects 0.000 description 2
- 238000005562 fading Methods 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 230000004720 fertilization Effects 0.000 description 2
- 230000008175 fetal development Effects 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 238000000799 fluorescence microscopy Methods 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 230000030279 gene silencing Effects 0.000 description 2
- 238000010353 genetic engineering Methods 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 210000005046 glial fibrillary acidic protein Anatomy 0.000 description 2
- MASNOZXLGMXCHN-ZLPAWPGGSA-N glucagon Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)C(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 MASNOZXLGMXCHN-ZLPAWPGGSA-N 0.000 description 2
- 229960004666 glucagon Drugs 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 229910052737 gold Inorganic materials 0.000 description 2
- 239000010931 gold Substances 0.000 description 2
- 230000005283 ground state Effects 0.000 description 2
- UYTPUPDQBNUYGX-UHFFFAOYSA-N guanine Chemical compound O=C1NC(N)=NC2=C1N=CN2 UYTPUPDQBNUYGX-UHFFFAOYSA-N 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- 230000002440 hepatic effect Effects 0.000 description 2
- 210000003494 hepatocyte Anatomy 0.000 description 2
- JYGXADMDTFJGBT-VWUMJDOOSA-N hydrocortisone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 JYGXADMDTFJGBT-VWUMJDOOSA-N 0.000 description 2
- 230000001146 hypoxic effect Effects 0.000 description 2
- 238000003364 immunohistochemistry Methods 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 210000004263 induced pluripotent stem cell Anatomy 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 108010024084 integrin alpha7 Proteins 0.000 description 2
- 230000001788 irregular Effects 0.000 description 2
- 230000000366 juvenile effect Effects 0.000 description 2
- 210000000629 knee joint Anatomy 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 230000007257 malfunction Effects 0.000 description 2
- 230000008774 maternal effect Effects 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 2
- 229940126619 mouse monoclonal antibody Drugs 0.000 description 2
- 208000020460 mucolipidosis II alpha/beta Diseases 0.000 description 2
- 206010028093 mucopolysaccharidosis Diseases 0.000 description 2
- 208000011045 mucopolysaccharidosis type 3 Diseases 0.000 description 2
- 208000025919 mucopolysaccharidosis type 7 Diseases 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 206010028537 myelofibrosis Diseases 0.000 description 2
- 210000005036 nerve Anatomy 0.000 description 2
- 210000000653 nervous system Anatomy 0.000 description 2
- 238000003199 nucleic acid amplification method Methods 0.000 description 2
- 239000002773 nucleotide Substances 0.000 description 2
- 125000003729 nucleotide group Chemical group 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- 230000005868 ontogenesis Effects 0.000 description 2
- 210000000056 organ Anatomy 0.000 description 2
- 229960003104 ornithine Drugs 0.000 description 2
- 239000012188 paraffin wax Substances 0.000 description 2
- 239000006201 parenteral dosage form Substances 0.000 description 2
- 201000003045 paroxysmal nocturnal hemoglobinuria Diseases 0.000 description 2
- 230000008775 paternal effect Effects 0.000 description 2
- 239000008188 pellet Substances 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920001184 polypeptide Polymers 0.000 description 2
- 238000004321 preservation Methods 0.000 description 2
- 230000000750 progressive effect Effects 0.000 description 2
- 230000004952 protein activity Effects 0.000 description 2
- 238000003753 real-time PCR Methods 0.000 description 2
- 230000001172 regenerating effect Effects 0.000 description 2
- 230000010076 replication Effects 0.000 description 2
- 230000008458 response to injury Effects 0.000 description 2
- 238000012552 review Methods 0.000 description 2
- 150000003384 small molecules Chemical group 0.000 description 2
- 210000002460 smooth muscle Anatomy 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 238000009168 stem cell therapy Methods 0.000 description 2
- 238000009580 stem-cell therapy Methods 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- 238000002604 ultrasonography Methods 0.000 description 2
- 210000001835 viscera Anatomy 0.000 description 2
- YFGBQHOOROIVKG-BHDDXSALSA-N (2R)-2-[[(2R)-2-[[2-[[2-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]acetyl]amino]acetyl]amino]-3-phenylpropanoyl]amino]-4-methylsulfanylbutanoic acid Chemical compound C([C@H](C(=O)N[C@H](CCSC)C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=CC=C1 YFGBQHOOROIVKG-BHDDXSALSA-N 0.000 description 1
- BSDCIRGNJKZPFV-GWOFURMSSA-N (2r,3s,4r,5r)-2-(hydroxymethyl)-5-(2,5,6-trichlorobenzimidazol-1-yl)oxolane-3,4-diol Chemical group O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C2=CC(Cl)=C(Cl)C=C2N=C1Cl BSDCIRGNJKZPFV-GWOFURMSSA-N 0.000 description 1
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- WVHGJJRMKGDTEC-WCIJHFMNSA-N 2-[(1R,4S,8R,10S,13S,16S,27R,34S)-34-[(2S)-butan-2-yl]-8,22-dihydroxy-13-[(2R,3S)-3-hydroxybutan-2-yl]-2,5,11,14,27,30,33,36,39-nonaoxo-27lambda4-thia-3,6,12,15,25,29,32,35,38-nonazapentacyclo[14.12.11.06,10.018,26.019,24]nonatriaconta-18(26),19(24),20,22-tetraen-4-yl]acetamide Chemical compound CC[C@H](C)[C@@H]1NC(=O)CNC(=O)[C@@H]2Cc3c([nH]c4cc(O)ccc34)[S@](=O)C[C@H](NC(=O)CNC1=O)C(=O)N[C@@H](CC(N)=O)C(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H]([C@@H](C)[C@H](C)O)C(=O)N2 WVHGJJRMKGDTEC-WCIJHFMNSA-N 0.000 description 1
- JKYKXTRKURYNGW-UHFFFAOYSA-N 3,4-dihydroxy-9,10-dioxo-9,10-dihydroanthracene-2-sulfonic acid Chemical compound O=C1C2=CC=CC=C2C(=O)C2=C1C(O)=C(O)C(S(O)(=O)=O)=C2 JKYKXTRKURYNGW-UHFFFAOYSA-N 0.000 description 1
- NMUSYJAQQFHJEW-UHFFFAOYSA-N 5-Azacytidine Natural products O=C1N=C(N)N=CN1C1C(O)C(O)C(CO)O1 NMUSYJAQQFHJEW-UHFFFAOYSA-N 0.000 description 1
- NMUSYJAQQFHJEW-KVTDHHQDSA-N 5-azacytidine Chemical compound O=C1N=C(N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 NMUSYJAQQFHJEW-KVTDHHQDSA-N 0.000 description 1
- 230000002407 ATP formation Effects 0.000 description 1
- 102100024643 ATP-binding cassette sub-family D member 1 Human genes 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 206010000830 Acute leukaemia Diseases 0.000 description 1
- 208000024893 Acute lymphoblastic leukemia Diseases 0.000 description 1
- 208000031261 Acute myeloid leukaemia Diseases 0.000 description 1
- 208000016585 Acute panmyelosis with myelofibrosis Diseases 0.000 description 1
- 201000011452 Adrenoleukodystrophy Diseases 0.000 description 1
- 241000607528 Aeromonas hydrophila Species 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- VWEWCZSUWOEEFM-WDSKDSINSA-N Ala-Gly-Ala-Gly Chemical compound C[C@H](N)C(=O)NCC(=O)N[C@@H](C)C(=O)NCC(O)=O VWEWCZSUWOEEFM-WDSKDSINSA-N 0.000 description 1
- 231100000729 Amatoxin Toxicity 0.000 description 1
- 241001083548 Anemone Species 0.000 description 1
- 208000032467 Aplastic anaemia Diseases 0.000 description 1
- 108091023037 Aptamer Proteins 0.000 description 1
- 241000239223 Arachnida Species 0.000 description 1
- 206010003594 Ataxia telangiectasia Diseases 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- 206010003694 Atrophy Diseases 0.000 description 1
- 241000972773 Aulopiformes Species 0.000 description 1
- 208000023275 Autoimmune disease Diseases 0.000 description 1
- 229930192334 Auxin Natural products 0.000 description 1
- 101150076489 B gene Proteins 0.000 description 1
- 208000010839 B-cell chronic lymphocytic leukemia Diseases 0.000 description 1
- 208000032791 BCR-ABL1 positive chronic myelogenous leukemia Diseases 0.000 description 1
- 208000035462 Biphenotypic Acute Leukemia Diseases 0.000 description 1
- 241000157302 Bison bison athabascae Species 0.000 description 1
- 208000008720 Bone Marrow Neoplasms Diseases 0.000 description 1
- 108010049955 Bone Morphogenetic Protein 4 Proteins 0.000 description 1
- 102100024505 Bone morphogenetic protein 4 Human genes 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 101710183816 Brevinin-2E Proteins 0.000 description 1
- FWPKHBSTLJXXIA-CATQOQJWSA-N CC[C@H](C)[C@H](NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCSC)[C@@H](C)CC)[C@@H](C)CC)[C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(O)=O Chemical compound CC[C@H](C)[C@H](NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCSC)[C@@H](C)CC)[C@@H](C)CC)[C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(O)=O FWPKHBSTLJXXIA-CATQOQJWSA-N 0.000 description 1
- 241000282461 Canis lupus Species 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 206010007558 Cardiac failure chronic Diseases 0.000 description 1
- 208000004918 Cartilage-hair hypoplasia Diseases 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 241000282994 Cervidae Species 0.000 description 1
- 208000031404 Chromosome Aberrations Diseases 0.000 description 1
- 208000031879 Chédiak-Higashi syndrome Diseases 0.000 description 1
- 102000018156 Claudin-7 Human genes 0.000 description 1
- 108050007296 Claudin-7 Proteins 0.000 description 1
- 241000193468 Clostridium perfringens Species 0.000 description 1
- 206010010099 Combined immunodeficiency Diseases 0.000 description 1
- 206010053138 Congenital aplastic anaemia Diseases 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 102100025621 Cytochrome b-245 heavy chain Human genes 0.000 description 1
- 230000004543 DNA replication Effects 0.000 description 1
- 102000016928 DNA-directed DNA polymerase Human genes 0.000 description 1
- 108010014303 DNA-directed DNA polymerase Proteins 0.000 description 1
- 101150003195 DNMT3L gene Proteins 0.000 description 1
- 101150063564 DPPA3 gene Proteins 0.000 description 1
- 244000000626 Daucus carota Species 0.000 description 1
- 235000002767 Daucus carota Nutrition 0.000 description 1
- 101710121036 Delta-hemolysin Proteins 0.000 description 1
- 102100036912 Desmin Human genes 0.000 description 1
- 108010044052 Desmin Proteins 0.000 description 1
- 102100037124 Developmental pluripotency-associated 5 protein Human genes 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- QRLVDLBMBULFAL-UHFFFAOYSA-N Digitonin Natural products CC1CCC2(OC1)OC3C(O)C4C5CCC6CC(OC7OC(CO)C(OC8OC(CO)C(O)C(OC9OCC(O)C(O)C9OC%10OC(CO)C(O)C(OC%11OC(CO)C(O)C(O)C%11O)C%10O)C8O)C(O)C7O)C(O)CC6(C)C5CCC4(C)C3C2C QRLVDLBMBULFAL-UHFFFAOYSA-N 0.000 description 1
- 102000016607 Diphtheria Toxin Human genes 0.000 description 1
- 108010053187 Diphtheria Toxin Proteins 0.000 description 1
- 101150006195 Dppa4 gene Proteins 0.000 description 1
- 241000271571 Dromaius novaehollandiae Species 0.000 description 1
- 108010042407 Endonucleases Proteins 0.000 description 1
- 102000004533 Endonucleases Human genes 0.000 description 1
- 241000204733 Entamoeba dispar Species 0.000 description 1
- 101710146739 Enterotoxin Proteins 0.000 description 1
- 101710201246 Eomesodermin Proteins 0.000 description 1
- 102100030751 Eomesodermin homolog Human genes 0.000 description 1
- 102400001368 Epidermal growth factor Human genes 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- 101150099612 Esrrb gene Proteins 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 208000006168 Ewing Sarcoma Diseases 0.000 description 1
- 101150092822 FGF5 gene Proteins 0.000 description 1
- 201000004939 Fanconi anemia Diseases 0.000 description 1
- RZSYLLSAWYUBPE-UHFFFAOYSA-L Fast green FCF Chemical compound [Na+].[Na+].C=1C=C(C(=C2C=CC(C=C2)=[N+](CC)CC=2C=C(C=CC=2)S([O-])(=O)=O)C=2C(=CC(O)=CC=2)S([O-])(=O)=O)C=CC=1N(CC)CC1=CC=CC(S([O-])(=O)=O)=C1 RZSYLLSAWYUBPE-UHFFFAOYSA-L 0.000 description 1
- 241000282324 Felis Species 0.000 description 1
- 102000018233 Fibroblast Growth Factor Human genes 0.000 description 1
- 108050007372 Fibroblast Growth Factor Proteins 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 208000015872 Gaucher disease Diseases 0.000 description 1
- 208000013607 Glanzmann thrombasthenia Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 102100035364 Growth/differentiation factor 3 Human genes 0.000 description 1
- 108010004889 Heat-Shock Proteins Proteins 0.000 description 1
- 102000002812 Heat-Shock Proteins Human genes 0.000 description 1
- 108010006464 Hemolysin Proteins Proteins 0.000 description 1
- 208000031220 Hemophilia Diseases 0.000 description 1
- 208000009292 Hemophilia A Diseases 0.000 description 1
- 101150064358 Hif3a gene Proteins 0.000 description 1
- 208000032672 Histiocytosis haematophagic Diseases 0.000 description 1
- 108090000252 Histolysain Proteins 0.000 description 1
- 101000881848 Homo sapiens Developmental pluripotency-associated 5 protein Proteins 0.000 description 1
- 101001023986 Homo sapiens Growth/differentiation factor 3 Proteins 0.000 description 1
- 101001139146 Homo sapiens Krueppel-like factor 2 Proteins 0.000 description 1
- 101000615488 Homo sapiens Methyl-CpG-binding domain protein 2 Proteins 0.000 description 1
- 101000590492 Homo sapiens Nuclear fragile X mental retardation-interacting protein 1 Proteins 0.000 description 1
- 101000908580 Homo sapiens Spliceosome RNA helicase DDX39B Proteins 0.000 description 1
- 206010061598 Immunodeficiency Diseases 0.000 description 1
- 208000029462 Immunodeficiency disease Diseases 0.000 description 1
- 208000026350 Inborn Genetic disease Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 108010002350 Interleukin-2 Proteins 0.000 description 1
- 101100288095 Klebsiella pneumoniae neo gene Proteins 0.000 description 1
- 102100020675 Krueppel-like factor 2 Human genes 0.000 description 1
- 201000005099 Langerhans cell histiocytosis Diseases 0.000 description 1
- 229930190887 Leptomycin Natural products 0.000 description 1
- 201000001779 Leukocyte adhesion deficiency Diseases 0.000 description 1
- 108700027766 Listeria monocytogenes hlyA Proteins 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 208000035809 Lymphohistiocytosis Diseases 0.000 description 1
- 208000030289 Lymphoproliferative disease Diseases 0.000 description 1
- 102000004083 Lymphotoxin-alpha Human genes 0.000 description 1
- 108090000542 Lymphotoxin-alpha Proteins 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 208000015439 Lysosomal storage disease Diseases 0.000 description 1
- 241000282560 Macaca mulatta Species 0.000 description 1
- 108060003100 Magainin Proteins 0.000 description 1
- 206010064912 Malignant transformation Diseases 0.000 description 1
- 108010036176 Melitten Proteins 0.000 description 1
- 108010049137 Member 1 Subfamily D ATP Binding Cassette Transporter Proteins 0.000 description 1
- 102400000988 Met-enkephalin Human genes 0.000 description 1
- 201000011442 Metachromatic leukodystrophy Diseases 0.000 description 1
- 108010042237 Methionine Enkephalin Proteins 0.000 description 1
- 102100021299 Methyl-CpG-binding domain protein 2 Human genes 0.000 description 1
- 206010056893 Mucopolysaccharidosis VII Diseases 0.000 description 1
- 208000034578 Multiple myelomas Diseases 0.000 description 1
- 101100224389 Mus musculus Dppa5a gene Proteins 0.000 description 1
- 101100446513 Mus musculus Fgf4 gene Proteins 0.000 description 1
- 101100369076 Mus musculus Tdgf1 gene Proteins 0.000 description 1
- 101100107167 Mus musculus Znf296 gene Proteins 0.000 description 1
- 241000282339 Mustela Species 0.000 description 1
- 206010028561 Myeloid metaplasia Diseases 0.000 description 1
- 208000014767 Myeloproliferative disease Diseases 0.000 description 1
- 102000003505 Myosin Human genes 0.000 description 1
- 108060008487 Myosin Proteins 0.000 description 1
- 101150012532 NANOG gene Proteins 0.000 description 1
- 101150063042 NR0B1 gene Proteins 0.000 description 1
- 108091061960 Naked DNA Proteins 0.000 description 1
- 208000012902 Nervous system disease Diseases 0.000 description 1
- 206010029260 Neuroblastoma Diseases 0.000 description 1
- 208000025966 Neurological disease Diseases 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 206010029719 Nonspecific reaction Diseases 0.000 description 1
- 102000014736 Notch Human genes 0.000 description 1
- 108010070047 Notch Receptors Proteins 0.000 description 1
- 102100032428 Nuclear fragile X mental retardation-interacting protein 1 Human genes 0.000 description 1
- 108091028043 Nucleic acid sequence Proteins 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 208000022873 Ocular disease Diseases 0.000 description 1
- 238000012408 PCR amplification Methods 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 241000282579 Pan Species 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 108010067902 Peptide Library Proteins 0.000 description 1
- KHGNFPUMBJSZSM-UHFFFAOYSA-N Perforine Natural products COC1=C2CCC(O)C(CCC(C)(C)O)(OC)C2=NC2=C1C=CO2 KHGNFPUMBJSZSM-UHFFFAOYSA-N 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 208000013544 Platelet disease Diseases 0.000 description 1
- 101710183389 Pneumolysin Proteins 0.000 description 1
- 208000008601 Polycythemia Diseases 0.000 description 1
- 229920000954 Polyglycolide Polymers 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 208000006265 Renal cell carcinoma Diseases 0.000 description 1
- 108700008625 Reporter Genes Proteins 0.000 description 1
- 101150071661 SLC25A20 gene Proteins 0.000 description 1
- 101150052594 SLC2A3 gene Proteins 0.000 description 1
- 241000277331 Salmonidae Species 0.000 description 1
- 241000555745 Sciuridae Species 0.000 description 1
- 229920005654 Sephadex Polymers 0.000 description 1
- 239000012507 Sephadex™ Substances 0.000 description 1
- 229920002684 Sepharose Polymers 0.000 description 1
- 201000001828 Sly syndrome Diseases 0.000 description 1
- 241000191967 Staphylococcus aureus Species 0.000 description 1
- 241000272534 Struthio camelus Species 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 208000000392 Thrombasthenia Diseases 0.000 description 1
- 208000005485 Thrombocytosis Diseases 0.000 description 1
- 102000004338 Transferrin Human genes 0.000 description 1
- 108090000901 Transferrin Proteins 0.000 description 1
- 108700019146 Transgenes Proteins 0.000 description 1
- 208000030886 Traumatic Brain injury Diseases 0.000 description 1
- 108010067973 Valinomycin Proteins 0.000 description 1
- 241000607626 Vibrio cholerae Species 0.000 description 1
- 241000607272 Vibrio parahaemolyticus Species 0.000 description 1
- 241000282485 Vulpes vulpes Species 0.000 description 1
- 208000016025 Waldenstroem macroglobulinemia Diseases 0.000 description 1
- 208000033559 Waldenström macroglobulinemia Diseases 0.000 description 1
- 208000026589 Wolman disease Diseases 0.000 description 1
- 206010052428 Wound Diseases 0.000 description 1
- 240000001781 Xanthosoma sagittifolium Species 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 238000002679 ablation Methods 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 208000036677 acute biphenotypic leukemia Diseases 0.000 description 1
- 208000036676 acute undifferentiated leukemia Diseases 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 201000009628 adenosine deaminase deficiency Diseases 0.000 description 1
- 238000004115 adherent culture Methods 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 108010014387 aerolysin Proteins 0.000 description 1
- 239000011543 agarose gel Substances 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 108010014709 amatoxin Proteins 0.000 description 1
- 125000000539 amino acid group Chemical group 0.000 description 1
- 229940009444 amphotericin Drugs 0.000 description 1
- 206010002026 amyotrophic lateral sclerosis Diseases 0.000 description 1
- 210000003484 anatomy Anatomy 0.000 description 1
- 230000003042 antagnostic effect Effects 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 230000002917 arthritic effect Effects 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 230000037444 atrophy Effects 0.000 description 1
- 230000002238 attenuated effect Effects 0.000 description 1
- 208000036556 autosomal recessive T cell-negative B cell-negative NK cell-negative due to adenosine deaminase deficiency severe combined immunodeficiency Diseases 0.000 description 1
- 239000002363 auxin Substances 0.000 description 1
- 229960002756 azacitidine Drugs 0.000 description 1
- 102100021298 b(0,+)-type amino acid transporter 1 Human genes 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 239000007640 basal medium Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 208000036815 beta tubulin Diseases 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 210000001109 blastomere Anatomy 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 201000006491 bone marrow cancer Diseases 0.000 description 1
- 210000002798 bone marrow cell Anatomy 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000007975 buffered saline Substances 0.000 description 1
- 101150102633 cact gene Proteins 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 229910002091 carbon monoxide Inorganic materials 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 210000003321 cartilage cell Anatomy 0.000 description 1
- 241001233037 catfish Species 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 230000022131 cell cycle Effects 0.000 description 1
- 230000030833 cell death Effects 0.000 description 1
- 230000011712 cell development Effects 0.000 description 1
- 230000003915 cell function Effects 0.000 description 1
- 230000006727 cell loss Effects 0.000 description 1
- 239000006285 cell suspension Substances 0.000 description 1
- 230000010307 cell transformation Effects 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 230000007248 cellular mechanism Effects 0.000 description 1
- 230000033077 cellular process Effects 0.000 description 1
- 230000008668 cellular reprogramming Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 206010008129 cerebral palsy Diseases 0.000 description 1
- 150000005829 chemical entities Chemical class 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 230000002759 chromosomal effect Effects 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 208000016532 chronic granulomatous disease Diseases 0.000 description 1
- 208000024207 chronic leukemia Diseases 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 230000007012 clinical effect Effects 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 238000007621 cluster analysis Methods 0.000 description 1
- 230000005757 colony formation Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- FCFNRCROJUBPLU-UHFFFAOYSA-N compound M126 Natural products CC(C)C1NC(=O)C(C)OC(=O)C(C(C)C)NC(=O)C(C(C)C)OC(=O)C(C(C)C)NC(=O)C(C)OC(=O)C(C(C)C)NC(=O)C(C(C)C)OC(=O)C(C(C)C)NC(=O)C(C)OC(=O)C(C(C)C)NC(=O)C(C(C)C)OC1=O FCFNRCROJUBPLU-UHFFFAOYSA-N 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 210000002808 connective tissue Anatomy 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000001054 cortical effect Effects 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 150000003983 crown ethers Chemical class 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000012136 culture method Methods 0.000 description 1
- 210000001771 cumulus cell Anatomy 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- 150000001945 cysteines Chemical class 0.000 description 1
- 229940104302 cytosine Drugs 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 230000008260 defense mechanism Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 210000005045 desmin Anatomy 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
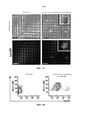
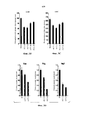
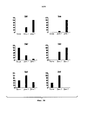
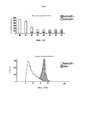
- 238000010586 diagram Methods 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- UVYVLBIGDKGWPX-KUAJCENISA-N digitonin Chemical compound O([C@@H]1[C@@H]([C@]2(CC[C@@H]3[C@@]4(C)C[C@@H](O)[C@H](O[C@H]5[C@@H]([C@@H](O)[C@@H](O[C@H]6[C@@H]([C@@H](O[C@H]7[C@@H]([C@@H](O)[C@H](O)CO7)O)[C@H](O)[C@@H](CO)O6)O[C@H]6[C@@H]([C@@H](O[C@H]7[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O7)O)[C@@H](O)[C@@H](CO)O6)O)[C@@H](CO)O5)O)C[C@@H]4CC[C@H]3[C@@H]2[C@@H]1O)C)[C@@H]1C)[C@]11CC[C@@H](C)CO1 UVYVLBIGDKGWPX-KUAJCENISA-N 0.000 description 1
- UVYVLBIGDKGWPX-UHFFFAOYSA-N digitonine Natural products CC1C(C2(CCC3C4(C)CC(O)C(OC5C(C(O)C(OC6C(C(OC7C(C(O)C(O)CO7)O)C(O)C(CO)O6)OC6C(C(OC7C(C(O)C(O)C(CO)O7)O)C(O)C(CO)O6)O)C(CO)O5)O)CC4CCC3C2C2O)C)C2OC11CCC(C)CO1 UVYVLBIGDKGWPX-UHFFFAOYSA-N 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 1
- 210000001163 endosome Anatomy 0.000 description 1
- 230000003511 endothelial effect Effects 0.000 description 1
- 239000000147 enterotoxin Substances 0.000 description 1
- 231100000655 enterotoxin Toxicity 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 230000006718 epigenetic regulation Effects 0.000 description 1
- 206010015037 epilepsy Diseases 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 230000002392 erythrophagocytic effect Effects 0.000 description 1
- VNUQOJUAIRBYQO-PPAUOFQQSA-N esculentin Chemical compound O([C@H]1[C@@H](OC)C[C@@H](O[C@@H]1C)O[C@@H]1[C@@H](C)O[C@@H](O[C@@H]2CC3=CC[C@@]4(O)[C@@]5(O)CC[C@](O)([C@@]5(C)[C@H](O)[C@@H](O)C4[C@@]3(C)CC2)C(C)=O)C[C@H]1OC)[C@@H]1O[C@H](C)[C@@H](O)[C@H](OC)[C@H]1O VNUQOJUAIRBYQO-PPAUOFQQSA-N 0.000 description 1
- VNUQOJUAIRBYQO-UHFFFAOYSA-N esculentin Natural products COC1CC(OC2CCC3(C)C4C(O)C(O)C5(C)C(O)(CCC5(O)C4(O)CC=C3C2)C(=O)C)OC(C)C1OC6CC(OC)C(OC7OC(C)C(O)C(OC)C7O)C(C)O6 VNUQOJUAIRBYQO-UHFFFAOYSA-N 0.000 description 1
- 210000003527 eukaryotic cell Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 210000001808 exosome Anatomy 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 210000000646 extraembryonic cell Anatomy 0.000 description 1
- 210000003414 extremity Anatomy 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 230000035558 fertility Effects 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 229940126864 fibroblast growth factor Drugs 0.000 description 1
- 229940093334 flomax Drugs 0.000 description 1
- 230000002538 fungal effect Effects 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 210000000973 gametocyte Anatomy 0.000 description 1
- 230000007045 gastrulation Effects 0.000 description 1
- 101150110946 gatC gene Proteins 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000011223 gene expression profiling Methods 0.000 description 1
- 208000016361 genetic disease Diseases 0.000 description 1
- 238000003144 genetic modification method Methods 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 238000011134 hematopoietic stem cell transplantation Methods 0.000 description 1
- 210000000777 hematopoietic system Anatomy 0.000 description 1
- 239000003228 hemolysin Substances 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 235000014304 histidine Nutrition 0.000 description 1
- 150000002411 histidines Chemical class 0.000 description 1
- 239000003276 histone deacetylase inhibitor Substances 0.000 description 1
- 230000013632 homeostatic process Effects 0.000 description 1
- 229960000890 hydrocortisone Drugs 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 238000007031 hydroxymethylation reaction Methods 0.000 description 1
- 239000000815 hypotonic solution Substances 0.000 description 1
- 238000010191 image analysis Methods 0.000 description 1
- 208000026278 immune system disease Diseases 0.000 description 1
- 230000007813 immunodeficiency Effects 0.000 description 1
- 238000003125 immunofluorescent labeling Methods 0.000 description 1
- 238000012151 immunohistochemical method Methods 0.000 description 1
- 230000004968 inflammatory condition Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 208000030603 inherited susceptibility to asthma Diseases 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 230000003426 interchromosomal effect Effects 0.000 description 1
- 210000004347 intestinal mucosa Anatomy 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 238000011031 large-scale manufacturing process Methods 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 230000004807 localization Effects 0.000 description 1
- 230000007762 localization of cell Effects 0.000 description 1
- 210000003738 lymphoid progenitor cell Anatomy 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 210000003712 lysosome Anatomy 0.000 description 1
- 230000001868 lysosomic effect Effects 0.000 description 1
- 239000003120 macrolide antibiotic agent Substances 0.000 description 1
- 229940041033 macrolides Drugs 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 208000002780 macular degeneration Diseases 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 230000036212 malign transformation Effects 0.000 description 1
- 210000003519 mature b lymphocyte Anatomy 0.000 description 1
- VDXZNPDIRNWWCW-JFTDCZMZSA-N melittin Chemical compound NCC(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(N)=O)CC1=CNC2=CC=CC=C12 VDXZNPDIRNWWCW-JFTDCZMZSA-N 0.000 description 1
- 210000002901 mesenchymal stem cell Anatomy 0.000 description 1
- 210000001704 mesoblast Anatomy 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 208000030159 metabolic disease Diseases 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 230000000116 mitigating effect Effects 0.000 description 1
- IKEOZQLIVHGQLJ-UHFFFAOYSA-M mitoTracker Red Chemical compound [Cl-].C1=CC(CCl)=CC=C1C(C1=CC=2CCCN3CCCC(C=23)=C1O1)=C2C1=C(CCC1)C3=[N+]1CCCC3=C2 IKEOZQLIVHGQLJ-UHFFFAOYSA-M 0.000 description 1
- 230000004898 mitochondrial function Effects 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 230000000877 morphologic effect Effects 0.000 description 1
- 210000000472 morula Anatomy 0.000 description 1
- 201000002273 mucopolysaccharidosis II Diseases 0.000 description 1
- 208000005340 mucopolysaccharidosis III Diseases 0.000 description 1
- 208000022018 mucopolysaccharidosis type 2 Diseases 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- 210000000663 muscle cell Anatomy 0.000 description 1
- 210000001665 muscle stem cell Anatomy 0.000 description 1
- 208000025113 myeloid leukemia Diseases 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 230000008689 nuclear function Effects 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 210000001706 olfactory mucosa Anatomy 0.000 description 1
- 210000000287 oocyte Anatomy 0.000 description 1
- 210000001328 optic nerve Anatomy 0.000 description 1
- 230000033667 organ regeneration Effects 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 230000000399 orthopedic effect Effects 0.000 description 1
- 210000004409 osteocyte Anatomy 0.000 description 1
- 208000002865 osteopetrosis Diseases 0.000 description 1
- 230000000242 pagocytic effect Effects 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 239000000123 paper Substances 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 238000002161 passivation Methods 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 239000000816 peptidomimetic Substances 0.000 description 1
- 229930192851 perforin Natural products 0.000 description 1
- 108010029039 phallolysine Proteins 0.000 description 1
- 108010045867 phallotoxin Proteins 0.000 description 1
- RLZZZVKAURTHCP-UHFFFAOYSA-N phenanthrene-3,4-diol Chemical compound C1=CC=C2C3=C(O)C(O)=CC=C3C=CC2=C1 RLZZZVKAURTHCP-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 238000005375 photometry Methods 0.000 description 1
- 239000003375 plant hormone Substances 0.000 description 1
- 210000004180 plasmocyte Anatomy 0.000 description 1
- 239000002985 plastic film Substances 0.000 description 1
- 229920006255 plastic film Polymers 0.000 description 1
- 239000004633 polyglycolic acid Substances 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 230000035935 pregnancy Effects 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- XJMOSONTPMZWPB-UHFFFAOYSA-M propidium iodide Chemical compound [I-].[I-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CCC[N+](C)(CC)CC)=C1C1=CC=CC=C1 XJMOSONTPMZWPB-UHFFFAOYSA-M 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 238000003908 quality control method Methods 0.000 description 1
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 238000006479 redox reaction Methods 0.000 description 1
- 230000003938 response to stress Effects 0.000 description 1
- 108091008146 restriction endonucleases Proteins 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 201000007153 reticular dysgenesis Diseases 0.000 description 1
- 230000002207 retinal effect Effects 0.000 description 1
- 210000003705 ribosome Anatomy 0.000 description 1
- 239000011435 rock Substances 0.000 description 1
- 235000019515 salmon Nutrition 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 230000007017 scission Effects 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 238000005204 segregation Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000012163 sequencing technique Methods 0.000 description 1
- 235000004400 serine Nutrition 0.000 description 1
- 150000003355 serines Chemical class 0.000 description 1
- 239000012679 serum free medium Substances 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 210000002027 skeletal muscle Anatomy 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000021595 spermatogenesis Effects 0.000 description 1
- 208000020431 spinal cord injury Diseases 0.000 description 1
- 230000002269 spontaneous effect Effects 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 210000002536 stromal cell Anatomy 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 125000000547 substituted alkyl group Chemical group 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 230000008093 supporting effect Effects 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 210000002437 synoviocyte Anatomy 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- ZZIZZTHXZRDOFM-XFULWGLBSA-N tamsulosin hydrochloride Chemical compound [H+].[Cl-].CCOC1=CC=CC=C1OCCN[C@H](C)CC1=CC=C(OC)C(S(N)(=O)=O)=C1 ZZIZZTHXZRDOFM-XFULWGLBSA-N 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 230000004797 therapeutic response Effects 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 235000008521 threonine Nutrition 0.000 description 1
- 150000003588 threonines Chemical class 0.000 description 1
- 206010043554 thrombocytopenia Diseases 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 230000017423 tissue regeneration Effects 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 238000011200 topical administration Methods 0.000 description 1
- 210000003014 totipotent stem cell Anatomy 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 239000012581 transferrin Substances 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 230000009529 traumatic brain injury Effects 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- RTKIYFITIVXBLE-QEQCGCAPSA-N trichostatin A Chemical compound ONC(=O)/C=C/C(/C)=C/[C@@H](C)C(=O)C1=CC=C(N(C)C)C=C1 RTKIYFITIVXBLE-QEQCGCAPSA-N 0.000 description 1
- 210000000251 trophoblastic cell Anatomy 0.000 description 1
- 235000002374 tyrosine Nutrition 0.000 description 1
- 150000003668 tyrosines Chemical class 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 230000003827 upregulation Effects 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- FCFNRCROJUBPLU-DNDCDFAISA-N valinomycin Chemical compound CC(C)[C@@H]1NC(=O)[C@H](C)OC(=O)[C@@H](C(C)C)NC(=O)[C@@H](C(C)C)OC(=O)[C@H](C(C)C)NC(=O)[C@H](C)OC(=O)[C@@H](C(C)C)NC(=O)[C@@H](C(C)C)OC(=O)[C@H](C(C)C)NC(=O)[C@H](C)OC(=O)[C@@H](C(C)C)NC(=O)[C@@H](C(C)C)OC1=O FCFNRCROJUBPLU-DNDCDFAISA-N 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 229940118696 vibrio cholerae Drugs 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 230000036266 weeks of gestation Effects 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
- 210000005253 yeast cell Anatomy 0.000 description 1
- 210000001325 yolk sac Anatomy 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0607—Non-embryonic pluripotent stem cells, e.g. MASC
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/14—Drugs for dermatological disorders for baldness or alopecia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/02—Muscle relaxants, e.g. for tetanus or cramps
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/06—Antianaemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/04—Inotropic agents, i.e. stimulants of cardiac contraction; Drugs for heart failure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0696—Artificially induced pluripotent stem cells, e.g. iPS
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/02—Atmosphere, e.g. low oxygen conditions
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/60—Buffer, e.g. pH regulation, osmotic pressure
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/11—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from blood or immune system cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2521/00—Culture process characterised by the use of hydrostatic pressure, flow or shear forces
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2523/00—Culture process characterised by temperature
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Immunology (AREA)
- Cell Biology (AREA)
- Developmental Biology & Embryology (AREA)
- Hematology (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Diabetes (AREA)
- General Engineering & Computer Science (AREA)
- Pulmonology (AREA)
- Urology & Nephrology (AREA)
- Cardiology (AREA)
- Physical Education & Sports Medicine (AREA)
- Transplantation (AREA)
- Virology (AREA)
- Epidemiology (AREA)
- Molecular Biology (AREA)
- Neurology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Obesity (AREA)
- Heart & Thoracic Surgery (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261637631P | 2012-04-24 | 2012-04-24 | |
| US61/637,631 | 2012-04-24 | ||
| US201361779533P | 2013-03-13 | 2013-03-13 | |
| US61/779,533 | 2013-03-13 | ||
| PCT/US2013/037996 WO2013163296A1 (en) | 2012-04-24 | 2013-04-24 | Generating pluripotent cells de novo |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2014147093A RU2014147093A (ru) | 2016-06-10 |
| RU2696071C2 true RU2696071C2 (ru) | 2019-07-30 |
Family
ID=49483852
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2014147093A RU2696071C2 (ru) | 2012-04-24 | 2013-04-24 | Получение плюрипотентных клеток de novo |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US11963977B2 (enExample) |
| EP (2) | EP3483261B1 (enExample) |
| JP (5) | JP2015516812A (enExample) |
| KR (2) | KR20200084916A (enExample) |
| CN (2) | CN112048466A (enExample) |
| AU (2) | AU2013251649B2 (enExample) |
| CA (1) | CA2885576A1 (enExample) |
| ES (1) | ES2972525T3 (enExample) |
| RU (1) | RU2696071C2 (enExample) |
| SG (2) | SG11201407768QA (enExample) |
| WO (1) | WO2013163296A1 (enExample) |
Families Citing this family (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8497124B2 (en) | 2011-12-05 | 2013-07-30 | Factor Bioscience Inc. | Methods and products for reprogramming cells to a less differentiated state |
| DK3260140T3 (da) | 2011-12-05 | 2021-04-19 | Factor Bioscience Inc | Fremgangsmåder og produkter til transficering af celler |
| BR122019025681B1 (pt) | 2012-11-01 | 2023-04-18 | Factor Bioscience Inc | Método para inserir uma sequência de ácido nucleico em uma localização segura de um genoma de uma célula |
| CA2895791A1 (en) | 2013-01-25 | 2014-07-31 | Xcell Biosciences, Inc. | Methods, compositions, kits, and systems for selective enrichment of target cells |
| JP6495658B2 (ja) | 2013-02-08 | 2019-04-03 | 国立大学法人京都大学 | 巨核球及び血小板の製造方法 |
| US11241460B2 (en) | 2013-03-15 | 2022-02-08 | Astellas Institute For Regenerative Medicine | Photoreceptors and photoreceptor progenitors produced from pluripotent stem cells |
| WO2014201254A1 (en) | 2013-06-12 | 2014-12-18 | University Of Washington Through Its Center For Commercialization | Methods for maturing cardiomyocytes and uses thereof |
| WO2015117021A1 (en) | 2014-01-31 | 2015-08-06 | Factor Bioscience Inc. | Methods and products for nucleic acid production and delivery |
| RU2711942C2 (ru) * | 2014-03-19 | 2020-01-23 | Виселл Терапеутикс, Инк. | Способы, относящиеся к плюрипотентным клеткам |
| JP6986968B2 (ja) * | 2014-12-04 | 2021-12-22 | ステモン・インコーポレイテッド | エネルギーを利用した多能性細胞誘導装置及び方法 |
| WO2016089178A1 (ko) * | 2014-12-04 | 2016-06-09 | 가톨릭관동대학교산학협력단 | 에너지를 이용한 다능성 세포 유도 장치 및 방법 |
| EP3543339A1 (en) | 2015-02-13 | 2019-09-25 | Factor Bioscience Inc. | Nucleic acid products and methods of administration thereof |
| WO2016203477A1 (en) * | 2015-06-18 | 2016-12-22 | Yeda Research And Development Co. Ltd. | Conditioning protocols and use of same for tissue regeneration |
| KR102050852B1 (ko) * | 2016-03-11 | 2019-12-03 | 주식회사 스템온 | 세포 리프로그래밍 장치 |
| KR101855967B1 (ko) * | 2016-03-11 | 2018-05-10 | 가톨릭관동대학교산학협력단 | 물리적 자극에 의한 환경유입을 이용한 세포 리프로그래밍 방법 |
| WO2017155167A1 (ko) * | 2016-03-11 | 2017-09-14 | 가톨릭관동대학교산학협력단 | 세포 리프로그래밍 장치 |
| WO2017155166A1 (ko) * | 2016-03-11 | 2017-09-14 | 가톨릭관동대학교기술지주 주식회사 | 물리적 자극에 의한 환경유입을 이용한 세포 리프로그래밍 방법 |
| US10456423B2 (en) | 2016-06-13 | 2019-10-29 | SMART SURGICAL, Inc. | Compositions for biological systems and methods for preparing and using the same |
| US10426796B2 (en) | 2016-06-13 | 2019-10-01 | SMART SURGICAL, Inc. | Compositions for biological systems and methods for preparing and using the same |
| CN109803977B (zh) | 2016-08-17 | 2023-03-17 | 菲克特生物科学股份有限公司 | 核酸产品及其施用方法 |
| US20180127738A1 (en) * | 2016-11-07 | 2018-05-10 | BiomediStem, LLC | Production and therapeutic uses of epinul pluripotent cells and differentiated cells derived therefrom |
| EP3601530A1 (en) * | 2017-03-20 | 2020-02-05 | IFOM Fondazione Istituto Firc di Oncologia Molecolare | Method of generating 2 cell-like stem cells |
| WO2018226435A1 (en) * | 2017-06-10 | 2018-12-13 | SMART SURGICAL, Inc. | Compositions for biological systems and methods for preparing and using the same |
| CN107729932B (zh) * | 2017-10-10 | 2019-07-26 | 杭州智微信息科技有限公司 | 骨髓细胞标记方法和系统 |
| KR102416171B1 (ko) | 2018-10-02 | 2022-07-06 | 주식회사 스템온 | 세포의 리프로그래밍을 유도하는 리프로좀 및 그 제조 방법 |
| WO2021003462A1 (en) | 2019-07-03 | 2021-01-07 | Factor Bioscience Inc. | Cationic lipids and uses thereof |
| US10501404B1 (en) | 2019-07-30 | 2019-12-10 | Factor Bioscience Inc. | Cationic lipids and transfection methods |
| JP2022546578A (ja) * | 2019-09-04 | 2022-11-04 | ブイセル セラピューティックス,インコーポレイテッド | 癌を処置するためのワクチンおよびストレスリプログラミングによるその作製方法 |
| AU2021218762A1 (en) | 2020-02-14 | 2022-10-06 | Hackensack Meridian Health, Inc. | Platelet-derived mitochondria treatment and method of generating multipotent cells |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2215539C2 (ru) * | 1995-02-02 | 2003-11-10 | ДХУТ Гхази Джасвиндер | Способ получения недифференцированной клетки |
| US20070190646A1 (en) * | 2006-02-10 | 2007-08-16 | The Trustees Of The University Of Pennsylvania | Regulating stem cell differentiation by controlling matrix elasticity |
| WO2008038148A2 (en) * | 2006-05-11 | 2008-04-03 | Andrew Craig Boquest | Stem cells and methods of making and using stem cells |
| US20110070647A1 (en) * | 2009-07-15 | 2011-03-24 | Mari Dezawa | Pluripotent stem cell that can be isolated from body tissue |
Family Cites Families (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4963489A (en) | 1987-04-14 | 1990-10-16 | Marrow-Tech, Inc. | Three-dimensional cell and tissue culture system |
| FR2687404B1 (fr) | 1992-02-19 | 1994-05-20 | Centre Nal Recherc Scientifique | Peptides immunologiquement apparentes aux proteines d'un agent viral et leurs applications biologiques. |
| AU4543193A (en) | 1992-06-22 | 1994-01-24 | Henry E. Young | Scar inhibitory factor and use thereof |
| US5672346A (en) | 1992-07-27 | 1997-09-30 | Indiana University Foundation | Human stem cell compositions and methods |
| CA2146747C (en) | 1992-10-09 | 2006-12-19 | Brian A. Naughton | Liver reserve cells |
| US5753431A (en) * | 1993-10-13 | 1998-05-19 | Northeastern Ohio University | Cholesterol 7 α-hydroxdylase gene regulatory elements and transcription factors |
| US5843780A (en) | 1995-01-20 | 1998-12-01 | Wisconsin Alumni Research Foundation | Primate embryonic stem cells |
| WO2002057428A1 (en) | 2000-10-30 | 2002-07-25 | University Of Massachusetts | Isolation of spore-like cells from tissues exposed to extreme conditions |
| GB0113118D0 (en) * | 2001-05-31 | 2001-07-18 | Intercytex Ltd | Stem Cells |
| US7575321B2 (en) | 2003-10-30 | 2009-08-18 | Welch Allyn, Inc. | Apparatus and method of diagnosis of optically identifiable ophthalmic conditions |
| JP4676442B2 (ja) * | 2003-12-02 | 2011-04-27 | セラヴィー バイオサイエンシズ エルエルシー | 神経前駆細胞を増殖させるための組成物および方法 |
| EP1789540B9 (en) * | 2004-09-03 | 2012-02-22 | Moraga Biotechnology Inc. | Non-embryonic totipotent blastomer-like stem cells and methods therefor |
| US20060084167A1 (en) | 2004-10-16 | 2006-04-20 | Cohenford Menashi A | Formation of Hybrid Cells by Fusion of Lineage Committed Cells with Stem Cells |
| US20090298095A1 (en) * | 2005-04-15 | 2009-12-03 | The John Hopkins University | Immortalization of cells including neuronal cells |
| FR2885368A1 (fr) * | 2005-05-04 | 2006-11-10 | Fred Zacouto | Systemes et procedes de regeneration cellulaire et utilisations de telles cellules regenerees |
| CA2621161A1 (en) * | 2005-09-08 | 2007-03-15 | The Government Of The United States Of America As Represented By The Sec Retary Of The Department Of Health And Human Services | Methods for promoting stem cell proliferation and survival |
| US20070087437A1 (en) * | 2005-10-14 | 2007-04-19 | Jifan Hu | Methods for rejuvenating cells in vitro and in vivo |
| US8278104B2 (en) * | 2005-12-13 | 2012-10-02 | Kyoto University | Induced pluripotent stem cells produced with Oct3/4, Klf4 and Sox2 |
| US20100135970A1 (en) * | 2006-10-27 | 2010-06-03 | Caritas St. Elizabeth Medical Center Of Boston, In | Methods for Reprogramming Adult Somatic Cells and Uses Thereof |
| CN105861443A (zh) * | 2007-04-07 | 2016-08-17 | 怀特黑德生物医学研究所 | 体细胞重编程 |
| CA2704220A1 (en) * | 2007-10-30 | 2009-05-07 | Le Centre Nationale De La Recherche Scientifique | Method for conducting an assay for neutralizing antibodies |
| KR101564044B1 (ko) | 2007-10-31 | 2015-10-28 | 고쿠리츠 다이가쿠 호진 교토 다이가쿠 | 핵초기화 방법 |
| US20090280518A1 (en) * | 2008-05-12 | 2009-11-12 | Massachusetts Institute Of Technology | System for high throughput measurement of mechanical properties of cells |
| EP2492715B1 (en) * | 2009-10-06 | 2019-01-09 | Canon Kabushiki Kaisha | Rear attachment lens, imaging optical system, and image pickup apparatus |
| JPWO2011125948A1 (ja) * | 2010-04-02 | 2013-07-11 | 独立行政法人理化学研究所 | Es細胞の製造方法 |
| KR20120002134A (ko) | 2010-06-30 | 2012-01-05 | 서울대학교산학협력단 | 지방 기질 세포의 역분화를 유도하는 방법 |
| ES3005334T3 (en) | 2010-07-09 | 2025-03-14 | Gid Bio Inc | Apparatus and methods relating to collecting and processing human biological material containing adipose |
-
2013
- 2013-04-24 WO PCT/US2013/037996 patent/WO2013163296A1/en not_active Ceased
- 2013-04-24 EP EP18203532.9A patent/EP3483261B1/en active Active
- 2013-04-24 ES ES18203532T patent/ES2972525T3/es active Active
- 2013-04-24 CA CA2885576A patent/CA2885576A1/en active Pending
- 2013-04-24 JP JP2015509109A patent/JP2015516812A/ja active Pending
- 2013-04-24 KR KR1020207019169A patent/KR20200084916A/ko not_active Ceased
- 2013-04-24 US US14/397,080 patent/US11963977B2/en active Active
- 2013-04-24 KR KR1020147032963A patent/KR20150045935A/ko not_active Ceased
- 2013-04-24 CN CN202010907822.3A patent/CN112048466A/zh active Pending
- 2013-04-24 CN CN201380033613.9A patent/CN104718283A/zh active Pending
- 2013-04-24 AU AU2013251649A patent/AU2013251649B2/en active Active
- 2013-04-24 SG SG11201407768QA patent/SG11201407768QA/en unknown
- 2013-04-24 RU RU2014147093A patent/RU2696071C2/ru active
- 2013-04-24 EP EP13780959.6A patent/EP2844738A4/en not_active Ceased
- 2013-04-24 SG SG10201809901SA patent/SG10201809901SA/en unknown
-
2018
- 2018-06-20 JP JP2018117481A patent/JP2018183145A/ja not_active Withdrawn
-
2019
- 2019-06-05 AU AU2019203943A patent/AU2019203943B2/en active Active
-
2020
- 2020-07-16 JP JP2020122035A patent/JP2020182480A/ja not_active Withdrawn
-
2022
- 2022-11-01 JP JP2022175459A patent/JP2023002805A/ja active Pending
-
2025
- 2025-04-03 JP JP2025061922A patent/JP2025106394A/ja active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2215539C2 (ru) * | 1995-02-02 | 2003-11-10 | ДХУТ Гхази Джасвиндер | Способ получения недифференцированной клетки |
| US20070190646A1 (en) * | 2006-02-10 | 2007-08-16 | The Trustees Of The University Of Pennsylvania | Regulating stem cell differentiation by controlling matrix elasticity |
| WO2008038148A2 (en) * | 2006-05-11 | 2008-04-03 | Andrew Craig Boquest | Stem cells and methods of making and using stem cells |
| US20110070647A1 (en) * | 2009-07-15 | 2011-03-24 | Mari Dezawa | Pluripotent stem cell that can be isolated from body tissue |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2885576A1 (en) | 2013-10-31 |
| ES2972525T3 (es) | 2024-06-13 |
| RU2014147093A (ru) | 2016-06-10 |
| EP3483261B1 (en) | 2023-11-29 |
| JP2020182480A (ja) | 2020-11-12 |
| CN104718283A (zh) | 2015-06-17 |
| KR20200084916A (ko) | 2020-07-13 |
| EP2844738A4 (en) | 2015-12-23 |
| US11963977B2 (en) | 2024-04-23 |
| SG10201809901SA (en) | 2018-12-28 |
| KR20150045935A (ko) | 2015-04-29 |
| JP2015516812A (ja) | 2015-06-18 |
| SG11201407768QA (en) | 2015-01-29 |
| WO2013163296A1 (en) | 2013-10-31 |
| AU2013251649A1 (en) | 2014-11-06 |
| JP2023002805A (ja) | 2023-01-10 |
| JP2018183145A (ja) | 2018-11-22 |
| US20150110749A1 (en) | 2015-04-23 |
| EP2844738A1 (en) | 2015-03-11 |
| AU2019203943B2 (en) | 2021-10-07 |
| AU2013251649B2 (en) | 2019-04-04 |
| EP3483261A1 (en) | 2019-05-15 |
| AU2019203943A1 (en) | 2019-06-27 |
| CN112048466A (zh) | 2020-12-08 |
| JP2025106394A (ja) | 2025-07-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2696071C2 (ru) | Получение плюрипотентных клеток de novo | |
| AU2021286294B2 (en) | Methods relating to pluripotent cells | |
| HK40008457A (en) | Generating pluripotent cells de novo | |
| HK40041588A (en) | Generating pluripotent cells de novo | |
| HK40043770A (en) | Methods relating to pluripotent cells |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| HZ9A | Changing address for correspondence with an applicant | ||
| HC9A | Changing information about inventors |