RU2570730C2 - Стабильные иммуногенные композиции антигенов staphylococcus aureus - Google Patents
Стабильные иммуногенные композиции антигенов staphylococcus aureus Download PDFInfo
- Publication number
- RU2570730C2 RU2570730C2 RU2013128277/15A RU2013128277A RU2570730C2 RU 2570730 C2 RU2570730 C2 RU 2570730C2 RU 2013128277/15 A RU2013128277/15 A RU 2013128277/15A RU 2013128277 A RU2013128277 A RU 2013128277A RU 2570730 C2 RU2570730 C2 RU 2570730C2
- Authority
- RU
- Russia
- Prior art keywords
- immunogenic composition
- clfa
- protein
- lyophilized
- aureus
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 350
- 230000002163 immunogen Effects 0.000 title claims abstract description 254
- 241000191967 Staphylococcus aureus Species 0.000 title claims abstract description 24
- 108091007433 antigens Proteins 0.000 title abstract description 98
- 102000036639 antigens Human genes 0.000 title abstract description 98
- 239000000427 antigen Substances 0.000 title abstract description 97
- 229920001282 polysaccharide Polymers 0.000 claims abstract description 184
- 239000005017 polysaccharide Substances 0.000 claims abstract description 184
- 108090000765 processed proteins & peptides Proteins 0.000 claims abstract description 136
- 102000004196 processed proteins & peptides Human genes 0.000 claims abstract description 131
- 229920001184 polypeptide Polymers 0.000 claims abstract description 125
- 239000000872 buffer Substances 0.000 claims abstract description 66
- 230000004523 agglutinating effect Effects 0.000 claims abstract description 56
- 150000004676 glycans Chemical class 0.000 claims abstract description 5
- 108090000623 proteins and genes Proteins 0.000 claims description 231
- 102000004169 proteins and genes Human genes 0.000 claims description 226
- 229930006000 Sucrose Natural products 0.000 claims description 112
- 239000005720 sucrose Substances 0.000 claims description 112
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 claims description 106
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 claims description 103
- 229920000053 polysorbate 80 Polymers 0.000 claims description 76
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 claims description 74
- 229940068968 polysorbate 80 Drugs 0.000 claims description 74
- 239000007788 liquid Substances 0.000 claims description 71
- 230000027455 binding Effects 0.000 claims description 47
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 42
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 39
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims description 36
- 239000003085 diluting agent Substances 0.000 claims description 36
- 229930195725 Mannitol Natural products 0.000 claims description 35
- 239000000594 mannitol Substances 0.000 claims description 35
- 235000010355 mannitol Nutrition 0.000 claims description 35
- -1 polyoxyethylene Polymers 0.000 claims description 35
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 claims description 34
- 239000004067 bulking agent Substances 0.000 claims description 30
- 108010049003 Fibrinogen Proteins 0.000 claims description 26
- 102000008946 Fibrinogen Human genes 0.000 claims description 26
- 229940012952 fibrinogen Drugs 0.000 claims description 26
- 239000004094 surface-active agent Substances 0.000 claims description 24
- 150000001413 amino acids Chemical group 0.000 claims description 20
- 108090000056 Complement factor B Proteins 0.000 claims description 18
- 102000003712 Complement factor B Human genes 0.000 claims description 18
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 claims description 18
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 18
- 239000000600 sorbitol Substances 0.000 claims description 18
- 235000010356 sorbitol Nutrition 0.000 claims description 18
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 claims description 15
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 claims description 15
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 claims description 15
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 claims description 14
- 239000002671 adjuvant Substances 0.000 claims description 14
- 108010059574 C5a peptidase Proteins 0.000 claims description 10
- 238000006467 substitution reaction Methods 0.000 claims description 10
- 239000004471 Glycine Substances 0.000 claims description 9
- HNDVDQJCIGZPNO-RXMQYKEDSA-N D-histidine Chemical compound OC(=O)[C@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-RXMQYKEDSA-N 0.000 claims description 8
- 230000002829 reductive effect Effects 0.000 claims description 8
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 7
- 125000000185 sucrose group Chemical group 0.000 claims description 6
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 5
- 229930195729 fatty acid Natural products 0.000 claims description 5
- 239000000194 fatty acid Substances 0.000 claims description 5
- 229920001983 poloxamer Polymers 0.000 claims description 5
- RVGRUAULSDPKGF-UHFFFAOYSA-N Poloxamer Chemical compound C1CO1.CC1CO1 RVGRUAULSDPKGF-UHFFFAOYSA-N 0.000 claims description 3
- 150000005215 alkyl ethers Chemical class 0.000 claims description 3
- 229960000502 poloxamer Drugs 0.000 claims description 3
- 239000002775 capsule Substances 0.000 abstract description 43
- 239000003814 drug Substances 0.000 abstract description 41
- 230000001965 increasing effect Effects 0.000 abstract description 30
- 239000000126 substance Substances 0.000 abstract description 24
- 230000000694 effects Effects 0.000 abstract description 19
- 239000003795 chemical substances by application Substances 0.000 abstract description 13
- 235000018102 proteins Nutrition 0.000 description 216
- 150000004804 polysaccharides Chemical class 0.000 description 181
- 238000011969 continuous reassessment method Methods 0.000 description 164
- 238000002360 preparation method Methods 0.000 description 121
- 238000000034 method Methods 0.000 description 105
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 92
- 101150044687 crm gene Proteins 0.000 description 81
- 238000004108 freeze drying Methods 0.000 description 75
- 102000014914 Carrier Proteins Human genes 0.000 description 59
- 235000002639 sodium chloride Nutrition 0.000 description 57
- 108010078791 Carrier Proteins Proteins 0.000 description 56
- 235000018977 lysine Nutrition 0.000 description 54
- 238000013456 study Methods 0.000 description 52
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 48
- 239000011780 sodium chloride Substances 0.000 description 47
- 210000004027 cell Anatomy 0.000 description 45
- 208000015181 infectious disease Diseases 0.000 description 41
- 229940079593 drug Drugs 0.000 description 40
- 239000000047 product Substances 0.000 description 38
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 38
- 230000006870 function Effects 0.000 description 37
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 33
- 235000014304 histidine Nutrition 0.000 description 33
- 241000894006 Bacteria Species 0.000 description 32
- 229940126534 drug product Drugs 0.000 description 32
- 239000000825 pharmaceutical preparation Substances 0.000 description 31
- 229960005486 vaccine Drugs 0.000 description 31
- 150000001720 carbohydrates Chemical class 0.000 description 29
- 230000028993 immune response Effects 0.000 description 28
- 230000000890 antigenic effect Effects 0.000 description 27
- 238000002474 experimental method Methods 0.000 description 27
- 238000009472 formulation Methods 0.000 description 27
- 238000000113 differential scanning calorimetry Methods 0.000 description 26
- 238000004458 analytical method Methods 0.000 description 25
- 239000012634 fragment Substances 0.000 description 25
- 241001465754 Metazoa Species 0.000 description 23
- 230000008859 change Effects 0.000 description 22
- 238000000354 decomposition reaction Methods 0.000 description 22
- 235000001014 amino acid Nutrition 0.000 description 21
- 238000002983 circular dichroism Methods 0.000 description 21
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 21
- 238000001035 drying Methods 0.000 description 21
- 125000000539 amino acid group Chemical group 0.000 description 20
- 201000010099 disease Diseases 0.000 description 20
- 239000000499 gel Substances 0.000 description 20
- 238000004519 manufacturing process Methods 0.000 description 20
- 230000008569 process Effects 0.000 description 20
- 238000004128 high performance liquid chromatography Methods 0.000 description 19
- 238000002955 isolation Methods 0.000 description 19
- 238000000746 purification Methods 0.000 description 18
- 229940024606 amino acid Drugs 0.000 description 17
- 230000008014 freezing Effects 0.000 description 17
- 238000007710 freezing Methods 0.000 description 17
- 238000004007 reversed phase HPLC Methods 0.000 description 17
- 230000032683 aging Effects 0.000 description 16
- 238000003556 assay Methods 0.000 description 16
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 15
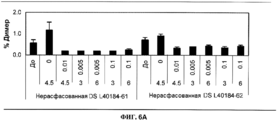
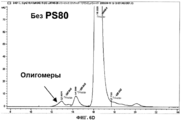
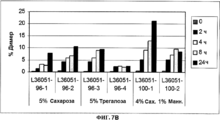
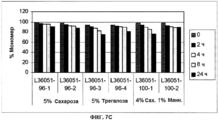
- 239000000539 dimer Substances 0.000 description 15
- 230000003053 immunization Effects 0.000 description 15
- 238000004153 renaturation Methods 0.000 description 15
- 230000001580 bacterial effect Effects 0.000 description 14
- 239000000463 material Substances 0.000 description 14
- 239000008186 active pharmaceutical agent Substances 0.000 description 13
- 230000002776 aggregation Effects 0.000 description 13
- 238000004220 aggregation Methods 0.000 description 13
- 239000007864 aqueous solution Substances 0.000 description 13
- 238000002649 immunization Methods 0.000 description 13
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 12
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 12
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 12
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 12
- 125000003275 alpha amino acid group Chemical group 0.000 description 12
- 230000000875 corresponding effect Effects 0.000 description 12
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 11
- 239000002253 acid Substances 0.000 description 11
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 10
- 241000699670 Mus sp. Species 0.000 description 10
- 229910019142 PO4 Inorganic materials 0.000 description 10
- 238000006640 acetylation reaction Methods 0.000 description 10
- 239000004615 ingredient Substances 0.000 description 10
- 239000000155 melt Substances 0.000 description 10
- 239000000178 monomer Substances 0.000 description 10
- 239000002245 particle Substances 0.000 description 10
- 235000021317 phosphate Nutrition 0.000 description 10
- 239000003755 preservative agent Substances 0.000 description 10
- 150000003839 salts Chemical class 0.000 description 10
- 238000001228 spectrum Methods 0.000 description 10
- 238000003860 storage Methods 0.000 description 10
- 229910017119 AlPO Inorganic materials 0.000 description 9
- 108060003951 Immunoglobulin Proteins 0.000 description 9
- 239000004472 Lysine Substances 0.000 description 9
- 238000003776 cleavage reaction Methods 0.000 description 9
- 230000007423 decrease Effects 0.000 description 9
- 230000036039 immunity Effects 0.000 description 9
- 102000018358 immunoglobulin Human genes 0.000 description 9
- 238000000338 in vitro Methods 0.000 description 9
- 238000002347 injection Methods 0.000 description 9
- 239000007924 injection Substances 0.000 description 9
- 238000004255 ion exchange chromatography Methods 0.000 description 9
- 238000002844 melting Methods 0.000 description 9
- 230000008018 melting Effects 0.000 description 9
- 238000012986 modification Methods 0.000 description 9
- 238000007911 parenteral administration Methods 0.000 description 9
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 9
- 239000010452 phosphate Substances 0.000 description 9
- 239000000523 sample Substances 0.000 description 9
- 230000007017 scission Effects 0.000 description 9
- 235000000346 sugar Nutrition 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 238000010257 thawing Methods 0.000 description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 8
- 210000001744 T-lymphocyte Anatomy 0.000 description 8
- 230000015556 catabolic process Effects 0.000 description 8
- 230000021615 conjugation Effects 0.000 description 8
- 230000006240 deamidation Effects 0.000 description 8
- 238000011161 development Methods 0.000 description 8
- 230000018109 developmental process Effects 0.000 description 8
- 239000000839 emulsion Substances 0.000 description 8
- 239000000945 filler Substances 0.000 description 8
- 239000011159 matrix material Substances 0.000 description 8
- 230000004048 modification Effects 0.000 description 8
- 230000001681 protective effect Effects 0.000 description 8
- 238000011084 recovery Methods 0.000 description 8
- 239000008362 succinate buffer Substances 0.000 description 8
- 230000004083 survival effect Effects 0.000 description 8
- 235000002374 tyrosine Nutrition 0.000 description 8
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 8
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Natural products OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 7
- 241000191963 Staphylococcus epidermidis Species 0.000 description 7
- 210000004369 blood Anatomy 0.000 description 7
- 239000008280 blood Substances 0.000 description 7
- 230000001413 cellular effect Effects 0.000 description 7
- 238000006731 degradation reaction Methods 0.000 description 7
- 239000003599 detergent Substances 0.000 description 7
- 229940088679 drug related substance Drugs 0.000 description 7
- 238000001914 filtration Methods 0.000 description 7
- 239000011521 glass Substances 0.000 description 7
- 239000003446 ligand Substances 0.000 description 7
- 108091033319 polynucleotide Proteins 0.000 description 7
- 102000040430 polynucleotide Human genes 0.000 description 7
- 239000002157 polynucleotide Substances 0.000 description 7
- 230000009467 reduction Effects 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 238000011282 treatment Methods 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 6
- 208000031729 Bacteremia Diseases 0.000 description 6
- 101710087657 Manganese ABC transporter substrate-binding lipoprotein Proteins 0.000 description 6
- 241000283973 Oryctolagus cuniculus Species 0.000 description 6
- 206010040047 Sepsis Diseases 0.000 description 6
- 238000004847 absorption spectroscopy Methods 0.000 description 6
- 238000010171 animal model Methods 0.000 description 6
- 239000012736 aqueous medium Substances 0.000 description 6
- 239000002738 chelating agent Substances 0.000 description 6
- 150000001875 compounds Chemical class 0.000 description 6
- 230000001419 dependent effect Effects 0.000 description 6
- 239000012530 fluid Substances 0.000 description 6
- 230000000670 limiting effect Effects 0.000 description 6
- 230000001404 mediated effect Effects 0.000 description 6
- 239000012528 membrane Substances 0.000 description 6
- 210000004379 membrane Anatomy 0.000 description 6
- 239000002773 nucleotide Substances 0.000 description 6
- 125000003729 nucleotide group Chemical group 0.000 description 6
- 230000008520 organization Effects 0.000 description 6
- 230000003647 oxidation Effects 0.000 description 6
- 238000007254 oxidation reaction Methods 0.000 description 6
- 239000012071 phase Substances 0.000 description 6
- 239000006187 pill Substances 0.000 description 6
- 229920000136 polysorbate Polymers 0.000 description 6
- 150000003254 radicals Chemical class 0.000 description 6
- 230000004044 response Effects 0.000 description 6
- 241000894007 species Species 0.000 description 6
- 230000007704 transition Effects 0.000 description 6
- YYGNTYWPHWGJRM-UHFFFAOYSA-N (6E,10E,14E,18E)-2,6,10,15,19,23-hexamethyltetracosa-2,6,10,14,18,22-hexaene Chemical compound CC(C)=CCCC(C)=CCCC(C)=CCCC=C(C)CCC=C(C)CCC=C(C)C YYGNTYWPHWGJRM-UHFFFAOYSA-N 0.000 description 5
- FWEOQOXTVHGIFQ-UHFFFAOYSA-M 8-anilinonaphthalene-1-sulfonate Chemical compound C=12C(S(=O)(=O)[O-])=CC=CC2=CC=CC=1NC1=CC=CC=C1 FWEOQOXTVHGIFQ-UHFFFAOYSA-M 0.000 description 5
- 101710146739 Enterotoxin Proteins 0.000 description 5
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 5
- 241000282412 Homo Species 0.000 description 5
- 102000008133 Iron-Binding Proteins Human genes 0.000 description 5
- 108010035210 Iron-Binding Proteins Proteins 0.000 description 5
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 5
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 5
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 5
- 108700018351 Major Histocompatibility Complex Proteins 0.000 description 5
- MSFSPUZXLOGKHJ-UHFFFAOYSA-N Muraminsaeure Natural products OC(=O)C(C)OC1C(N)C(O)OC(CO)C1O MSFSPUZXLOGKHJ-UHFFFAOYSA-N 0.000 description 5
- 108010013639 Peptidoglycan Proteins 0.000 description 5
- 108010076504 Protein Sorting Signals Proteins 0.000 description 5
- 241000700159 Rattus Species 0.000 description 5
- BHEOSNUKNHRBNM-UHFFFAOYSA-N Tetramethylsqualene Natural products CC(=C)C(C)CCC(=C)C(C)CCC(C)=CCCC=C(C)CCC(C)C(=C)CCC(C)C(C)=C BHEOSNUKNHRBNM-UHFFFAOYSA-N 0.000 description 5
- 102100022203 Tumor necrosis factor receptor superfamily member 25 Human genes 0.000 description 5
- 239000004480 active ingredient Substances 0.000 description 5
- 239000003963 antioxidant agent Substances 0.000 description 5
- 235000006708 antioxidants Nutrition 0.000 description 5
- 108091008324 binding proteins Proteins 0.000 description 5
- 235000014633 carbohydrates Nutrition 0.000 description 5
- 210000002421 cell wall Anatomy 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N dodecahydrosqualene Natural products CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 description 5
- 239000003937 drug carrier Substances 0.000 description 5
- 239000000147 enterotoxin Substances 0.000 description 5
- 231100000655 enterotoxin Toxicity 0.000 description 5
- 235000011187 glycerol Nutrition 0.000 description 5
- 210000000987 immune system Anatomy 0.000 description 5
- 230000005847 immunogenicity Effects 0.000 description 5
- 238000001727 in vivo Methods 0.000 description 5
- 201000007119 infective endocarditis Diseases 0.000 description 5
- 210000003734 kidney Anatomy 0.000 description 5
- 229930182817 methionine Natural products 0.000 description 5
- 238000002156 mixing Methods 0.000 description 5
- 238000012544 monitoring process Methods 0.000 description 5
- 230000035772 mutation Effects 0.000 description 5
- 238000004848 nephelometry Methods 0.000 description 5
- 230000000625 opsonophagocytic effect Effects 0.000 description 5
- 230000001717 pathogenic effect Effects 0.000 description 5
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 229940031439 squalene Drugs 0.000 description 5
- TUHBEKDERLKLEC-UHFFFAOYSA-N squalene Natural products CC(=CCCC(=CCCC(=CCCC=C(/C)CCC=C(/C)CC=C(C)C)C)C)C TUHBEKDERLKLEC-UHFFFAOYSA-N 0.000 description 5
- 230000020382 suppression by virus of host antigen processing and presentation of peptide antigen via MHC class I Effects 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 210000001519 tissue Anatomy 0.000 description 5
- 239000008215 water for injection Substances 0.000 description 5
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 4
- 101710092462 Alpha-hemolysin Proteins 0.000 description 4
- 239000004475 Arginine Substances 0.000 description 4
- 102000004127 Cytokines Human genes 0.000 description 4
- 108090000695 Cytokines Proteins 0.000 description 4
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 4
- 241000588724 Escherichia coli Species 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 4
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 4
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 4
- 102000018697 Membrane Proteins Human genes 0.000 description 4
- 108010052285 Membrane Proteins Proteins 0.000 description 4
- 108010062010 N-Acetylmuramoyl-L-alanine Amidase Proteins 0.000 description 4
- 206010057249 Phagocytosis Diseases 0.000 description 4
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- 101710124951 Phospholipase C Proteins 0.000 description 4
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 241001147693 Staphylococcus sp. Species 0.000 description 4
- 230000004913 activation Effects 0.000 description 4
- 235000004279 alanine Nutrition 0.000 description 4
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 description 4
- 230000005875 antibody response Effects 0.000 description 4
- PYMYPHUHKUWMLA-UHFFFAOYSA-N arabinose Natural products OCC(O)C(O)C(O)C=O PYMYPHUHKUWMLA-UHFFFAOYSA-N 0.000 description 4
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 4
- 235000009697 arginine Nutrition 0.000 description 4
- 108010056458 bacterial fibronectin-binding proteins Proteins 0.000 description 4
- SRBFZHDQGSBBOR-UHFFFAOYSA-N beta-D-Pyranose-Lyxose Natural products OC1COC(O)C(O)C1O SRBFZHDQGSBBOR-UHFFFAOYSA-N 0.000 description 4
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 4
- 210000004899 c-terminal region Anatomy 0.000 description 4
- 239000000969 carrier Substances 0.000 description 4
- 150000001768 cations Chemical class 0.000 description 4
- 239000012707 chemical precursor Substances 0.000 description 4
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- 230000000295 complement effect Effects 0.000 description 4
- 230000000959 cryoprotective effect Effects 0.000 description 4
- 230000006378 damage Effects 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- 229910001873 dinitrogen Inorganic materials 0.000 description 4
- 239000012636 effector Substances 0.000 description 4
- 206010014665 endocarditis Diseases 0.000 description 4
- 238000010353 genetic engineering Methods 0.000 description 4
- 235000013922 glutamic acid Nutrition 0.000 description 4
- 239000004220 glutamic acid Substances 0.000 description 4
- 210000002216 heart Anatomy 0.000 description 4
- 230000002209 hydrophobic effect Effects 0.000 description 4
- 229940072221 immunoglobulins Drugs 0.000 description 4
- 239000002955 immunomodulating agent Substances 0.000 description 4
- 229940121354 immunomodulator Drugs 0.000 description 4
- 230000006872 improvement Effects 0.000 description 4
- 230000003834 intracellular effect Effects 0.000 description 4
- 230000002147 killing effect Effects 0.000 description 4
- 239000008101 lactose Substances 0.000 description 4
- 210000000265 leukocyte Anatomy 0.000 description 4
- 230000021633 leukocyte mediated immunity Effects 0.000 description 4
- 150000002632 lipids Chemical class 0.000 description 4
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 4
- 229940126601 medicinal product Drugs 0.000 description 4
- 244000005700 microbiome Species 0.000 description 4
- 108020004707 nucleic acids Proteins 0.000 description 4
- 102000039446 nucleic acids Human genes 0.000 description 4
- 150000007523 nucleic acids Chemical class 0.000 description 4
- 229920001542 oligosaccharide Polymers 0.000 description 4
- 150000002482 oligosaccharides Chemical class 0.000 description 4
- 230000008782 phagocytosis Effects 0.000 description 4
- 230000002335 preservative effect Effects 0.000 description 4
- 238000011160 research Methods 0.000 description 4
- 229930182490 saponin Natural products 0.000 description 4
- 150000007949 saponins Chemical class 0.000 description 4
- 235000017709 saponins Nutrition 0.000 description 4
- 238000012216 screening Methods 0.000 description 4
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 4
- 238000002255 vaccination Methods 0.000 description 4
- 239000000304 virulence factor Substances 0.000 description 4
- 230000007923 virulence factor Effects 0.000 description 4
- MQWZJOSNNICZJE-JWXFUTCRSA-N (2s,3s,4r,5s)-5-acetamido-2,3,4-trihydroxy-6-oxohexanoic acid Chemical compound CC(=O)N[C@H](C=O)[C@@H](O)[C@H](O)[C@H](O)C(O)=O MQWZJOSNNICZJE-JWXFUTCRSA-N 0.000 description 3
- DRHZYJAUECRAJM-DWSYSWFDSA-N (2s,3s,4s,5r,6r)-6-[[(3s,4s,4ar,6ar,6bs,8r,8ar,12as,14ar,14br)-8a-[(2s,3r,4s,5r,6r)-3-[(2s,3r,4s,5r,6s)-5-[(2s,3r,4s,5r)-4-[(2s,3r,4r)-3,4-dihydroxy-4-(hydroxymethyl)oxolan-2-yl]oxy-3,5-dihydroxyoxan-2-yl]oxy-3,4-dihydroxy-6-methyloxan-2-yl]oxy-5-[(3s,5s, Chemical compound O([C@H]1[C@H](O)[C@H](O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO)O1)O)O[C@H]1CC[C@]2(C)[C@H]3CC=C4[C@@H]5CC(C)(C)CC[C@@]5([C@@H](C[C@@]4(C)[C@]3(C)CC[C@H]2[C@@]1(C=O)C)O)C(=O)O[C@@H]1O[C@H](C)[C@@H]([C@@H]([C@H]1O[C@H]1[C@@H]([C@H](O)[C@@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@](O)(CO)CO3)O)[C@H](O)CO2)O)[C@H](C)O1)O)O)OC(=O)C[C@@H](O)C[C@H](OC(=O)C[C@@H](O)C[C@@H]([C@@H](C)CC)O[C@H]1[C@@H]([C@@H](O)[C@H](CO)O1)O)[C@@H](C)CC)C(O)=O)[C@@H]1OC[C@@H](O)[C@H](O)[C@H]1O DRHZYJAUECRAJM-DWSYSWFDSA-N 0.000 description 3
- 102000019034 Chemokines Human genes 0.000 description 3
- 108010012236 Chemokines Proteins 0.000 description 3
- SRBFZHDQGSBBOR-IOVATXLUSA-N D-xylopyranose Chemical compound O[C@@H]1COC(O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-IOVATXLUSA-N 0.000 description 3
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 3
- IAJILQKETJEXLJ-UHFFFAOYSA-N Galacturonsaeure Natural products O=CC(O)C(O)C(O)C(O)C(O)=O IAJILQKETJEXLJ-UHFFFAOYSA-N 0.000 description 3
- 241000606768 Haemophilus influenzae Species 0.000 description 3
- 101000679903 Homo sapiens Tumor necrosis factor receptor superfamily member 25 Proteins 0.000 description 3
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 3
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 3
- 108010076876 Keratins Proteins 0.000 description 3
- 102000011782 Keratins Human genes 0.000 description 3
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 3
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 3
- 241000699666 Mus <mouse, genus> Species 0.000 description 3
- 206010031252 Osteomyelitis Diseases 0.000 description 3
- 108010081690 Pertussis Toxin Proteins 0.000 description 3
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 3
- 206010035664 Pneumonia Diseases 0.000 description 3
- 229920001214 Polysorbate 60 Polymers 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 206010041925 Staphylococcal infections Diseases 0.000 description 3
- 241000191940 Staphylococcus Species 0.000 description 3
- 102100040112 Tumor necrosis factor receptor superfamily member 10B Human genes 0.000 description 3
- UZQJVUCHXGYFLQ-AYDHOLPZSA-N [(2s,3r,4s,5r,6r)-4-[(2s,3r,4s,5r,6r)-4-[(2r,3r,4s,5r,6r)-4-[(2s,3r,4s,5r,6r)-3,5-dihydroxy-6-(hydroxymethyl)-4-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxan-2-yl]oxy-3,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-3,5-dihydroxy-6-(hy Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O)O[C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O)O[C@H]1CC[C@]2(C)[C@H]3CC=C4[C@@]([C@@]3(CC[C@H]2[C@@]1(C=O)C)C)(C)CC(O)[C@]1(CCC(CC14)(C)C)C(=O)O[C@H]1[C@@H]([C@@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O[C@H]4[C@@H]([C@@H](O[C@H]5[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O5)O)[C@H](O)[C@@H](CO)O4)O)[C@H](O)[C@@H](CO)O3)O)[C@H](O)[C@@H](CO)O2)O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O UZQJVUCHXGYFLQ-AYDHOLPZSA-N 0.000 description 3
- 238000007792 addition Methods 0.000 description 3
- 239000000853 adhesive Substances 0.000 description 3
- 230000001070 adhesive effect Effects 0.000 description 3
- 235000010443 alginic acid Nutrition 0.000 description 3
- 239000000783 alginic acid Substances 0.000 description 3
- 229920000615 alginic acid Polymers 0.000 description 3
- 229960001126 alginic acid Drugs 0.000 description 3
- 150000004781 alginic acids Chemical class 0.000 description 3
- ILRRQNADMUWWFW-UHFFFAOYSA-K aluminium phosphate Chemical compound O1[Al]2OP1(=O)O2 ILRRQNADMUWWFW-UHFFFAOYSA-K 0.000 description 3
- DIZPMCHEQGEION-UHFFFAOYSA-H aluminium sulfate (anhydrous) Chemical compound [Al+3].[Al+3].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O DIZPMCHEQGEION-UHFFFAOYSA-H 0.000 description 3
- 230000003078 antioxidant effect Effects 0.000 description 3
- 235000010323 ascorbic acid Nutrition 0.000 description 3
- 239000011668 ascorbic acid Substances 0.000 description 3
- 229940009098 aspartate Drugs 0.000 description 3
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 3
- 230000033228 biological regulation Effects 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 238000005277 cation exchange chromatography Methods 0.000 description 3
- 230000001276 controlling effect Effects 0.000 description 3
- 238000012217 deletion Methods 0.000 description 3
- 230000037430 deletion Effects 0.000 description 3
- 230000029087 digestion Effects 0.000 description 3
- 206010013023 diphtheria Diseases 0.000 description 3
- 229920001971 elastomer Polymers 0.000 description 3
- 239000002158 endotoxin Substances 0.000 description 3
- 238000011049 filling Methods 0.000 description 3
- 238000001506 fluorescence spectroscopy Methods 0.000 description 3
- 238000005227 gel permeation chromatography Methods 0.000 description 3
- 230000009477 glass transition Effects 0.000 description 3
- 239000008103 glucose Substances 0.000 description 3
- 229940047650 haemophilus influenzae Drugs 0.000 description 3
- 210000002443 helper t lymphocyte Anatomy 0.000 description 3
- 150000002411 histidines Chemical class 0.000 description 3
- 230000007062 hydrolysis Effects 0.000 description 3
- 238000006460 hydrolysis reaction Methods 0.000 description 3
- 230000001939 inductive effect Effects 0.000 description 3
- 239000003112 inhibitor Substances 0.000 description 3
- 238000007912 intraperitoneal administration Methods 0.000 description 3
- 229920006008 lipopolysaccharide Polymers 0.000 description 3
- 238000012792 lyophilization process Methods 0.000 description 3
- 238000013507 mapping Methods 0.000 description 3
- 239000008194 pharmaceutical composition Substances 0.000 description 3
- 229920005862 polyol Polymers 0.000 description 3
- 150000003077 polyols Chemical class 0.000 description 3
- 229950008882 polysorbate Drugs 0.000 description 3
- 235000004252 protein component Nutrition 0.000 description 3
- 239000010453 quartz Substances 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 210000002966 serum Anatomy 0.000 description 3
- 238000003998 size exclusion chromatography high performance liquid chromatography Methods 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 238000010561 standard procedure Methods 0.000 description 3
- 230000001954 sterilising effect Effects 0.000 description 3
- 230000035882 stress Effects 0.000 description 3
- 150000008163 sugars Chemical class 0.000 description 3
- 230000032258 transport Effects 0.000 description 3
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 3
- 230000000007 visual effect Effects 0.000 description 3
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 2
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 2
- QCDWFXQBSFUVSP-UHFFFAOYSA-N 2-phenoxyethanol Chemical compound OCCOC1=CC=CC=C1 QCDWFXQBSFUVSP-UHFFFAOYSA-N 0.000 description 2
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 2
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 2
- 108010006533 ATP-Binding Cassette Transporters Proteins 0.000 description 2
- 102000005416 ATP-Binding Cassette Transporters Human genes 0.000 description 2
- 241000251468 Actinopterygii Species 0.000 description 2
- 101710197219 Alpha-toxin Proteins 0.000 description 2
- 108090000254 Aureolysin Proteins 0.000 description 2
- 101710154607 Azurocidin Proteins 0.000 description 2
- 208000035143 Bacterial infection Diseases 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- 241000282836 Camelus dromedarius Species 0.000 description 2
- 206010007882 Cellulitis Diseases 0.000 description 2
- 101000597253 Clostridium novyi Phospholipase C Proteins 0.000 description 2
- 101000822677 Clostridium perfringens (strain 13 / Type A) Small, acid-soluble spore protein 1 Proteins 0.000 description 2
- 101000822695 Clostridium perfringens (strain 13 / Type A) Small, acid-soluble spore protein C1 Proteins 0.000 description 2
- 108091026890 Coding region Proteins 0.000 description 2
- 241000759568 Corixa Species 0.000 description 2
- 101710112752 Cytotoxin Proteins 0.000 description 2
- WQZGKKKJIJFFOK-WHZQZERISA-N D-aldose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-WHZQZERISA-N 0.000 description 2
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 2
- 108020004414 DNA Proteins 0.000 description 2
- 108010041986 DNA Vaccines Proteins 0.000 description 2
- 229940021995 DNA vaccine Drugs 0.000 description 2
- 229940032046 DTaP vaccine Drugs 0.000 description 2
- 108050002483 DltD Proteins 0.000 description 2
- 102000016942 Elastin Human genes 0.000 description 2
- 108010014258 Elastin Proteins 0.000 description 2
- 101000867232 Escherichia coli Heat-stable enterotoxin II Proteins 0.000 description 2
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 2
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 2
- 229920002444 Exopolysaccharide Polymers 0.000 description 2
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 2
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 2
- 108010067306 Fibronectins Proteins 0.000 description 2
- 102000016359 Fibronectins Human genes 0.000 description 2
- 241000192125 Firmicutes Species 0.000 description 2
- 102000004269 Granulocyte Colony-Stimulating Factor Human genes 0.000 description 2
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 description 2
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 2
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 2
- SQUHHTBVTRBESD-UHFFFAOYSA-N Hexa-Ac-myo-Inositol Natural products CC(=O)OC1C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C1OC(C)=O SQUHHTBVTRBESD-UHFFFAOYSA-N 0.000 description 2
- 101710141457 Histidine-binding periplasmic protein Proteins 0.000 description 2
- 101000610604 Homo sapiens Tumor necrosis factor receptor superfamily member 10B Proteins 0.000 description 2
- 102000014150 Interferons Human genes 0.000 description 2
- 108010050904 Interferons Proteins 0.000 description 2
- 102000015696 Interleukins Human genes 0.000 description 2
- 108010063738 Interleukins Proteins 0.000 description 2
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 2
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 2
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 2
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 2
- 241000270322 Lepidosauria Species 0.000 description 2
- 108090001030 Lipoproteins Proteins 0.000 description 2
- 102000004895 Lipoproteins Human genes 0.000 description 2
- 102000007651 Macrophage Colony-Stimulating Factor Human genes 0.000 description 2
- 108010046938 Macrophage Colony-Stimulating Factor Proteins 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 2
- 101001026869 Mus musculus F-box/LRR-repeat protein 3 Proteins 0.000 description 2
- XOCCAGJZGBCJME-IANFNVNHSA-N N-Acetyl-D-fucosamine Chemical compound C[C@H]1OC(O)[C@H](NC(C)=O)[C@@H](O)[C@H]1O XOCCAGJZGBCJME-IANFNVNHSA-N 0.000 description 2
- XOCCAGJZGBCJME-VAYLDTTESA-N N-Acetyl-L-Fucosamine Chemical compound C[C@@H]1OC(O)[C@@H](NC(C)=O)[C@H](O)[C@@H]1O XOCCAGJZGBCJME-VAYLDTTESA-N 0.000 description 2
- 108700020354 N-acetylmuramyl-threonyl-isoglutamine Proteins 0.000 description 2
- 239000005642 Oleic acid Substances 0.000 description 2
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 2
- 108091034117 Oligonucleotide Proteins 0.000 description 2
- 102000004264 Osteopontin Human genes 0.000 description 2
- 108010081689 Osteopontin Proteins 0.000 description 2
- 101710116435 Outer membrane protein Proteins 0.000 description 2
- 239000002033 PVDF binder Substances 0.000 description 2
- 108010078471 Panton-Valentine leukocidin Proteins 0.000 description 2
- 102000035195 Peptidases Human genes 0.000 description 2
- 108091005804 Peptidases Proteins 0.000 description 2
- 208000005228 Pericardial Effusion Diseases 0.000 description 2
- 241000404883 Pisa Species 0.000 description 2
- 229920001213 Polysorbate 20 Polymers 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- 101710092628 Probable histidine-binding protein Proteins 0.000 description 2
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 2
- 239000004365 Protease Substances 0.000 description 2
- 108010066124 Protein S Proteins 0.000 description 2
- 229940096437 Protein S Drugs 0.000 description 2
- 102000029301 Protein S Human genes 0.000 description 2
- 101000576806 Protobothrops flavoviridis Small serum protein 1 Proteins 0.000 description 2
- 101000576807 Protobothrops flavoviridis Small serum protein 2 Proteins 0.000 description 2
- 206010037596 Pyelonephritis Diseases 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- 101150036293 Selenop gene Proteins 0.000 description 2
- 206010040070 Septic Shock Diseases 0.000 description 2
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 2
- 101100227488 Staphylococcus aureus (strain USA300) fnbB gene Proteins 0.000 description 2
- 101000794214 Staphylococcus aureus Toxic shock syndrome toxin-1 Proteins 0.000 description 2
- 241000193998 Streptococcus pneumoniae Species 0.000 description 2
- 241000282887 Suidae Species 0.000 description 2
- 108010031318 Vitronectin Proteins 0.000 description 2
- 102100035140 Vitronectin Human genes 0.000 description 2
- 238000009825 accumulation Methods 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 239000002776 alpha toxin Substances 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- PYMYPHUHKUWMLA-WDCZJNDASA-N arabinose Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)C=O PYMYPHUHKUWMLA-WDCZJNDASA-N 0.000 description 2
- 210000003567 ascitic fluid Anatomy 0.000 description 2
- 229960005070 ascorbic acid Drugs 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- 208000022362 bacterial infectious disease Diseases 0.000 description 2
- 244000052616 bacterial pathogen Species 0.000 description 2
- UREZNYTWGJKWBI-UHFFFAOYSA-M benzethonium chloride Chemical compound [Cl-].C1=CC(C(C)(C)CC(C)(C)C)=CC=C1OCCOCC[N+](C)(C)CC1=CC=CC=C1 UREZNYTWGJKWBI-UHFFFAOYSA-M 0.000 description 2
- 229960001950 benzethonium chloride Drugs 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 210000000988 bone and bone Anatomy 0.000 description 2
- 229940098773 bovine serum albumin Drugs 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 239000007853 buffer solution Substances 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 230000007969 cellular immunity Effects 0.000 description 2
- 210000001175 cerebrospinal fluid Anatomy 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 238000002512 chemotherapy Methods 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 239000000084 colloidal system Substances 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 238000011109 contamination Methods 0.000 description 2
- 239000002577 cryoprotective agent Substances 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 235000018417 cysteine Nutrition 0.000 description 2
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 2
- 231100000599 cytotoxic agent Toxicity 0.000 description 2
- 239000002619 cytotoxin Substances 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 231100000517 death Toxicity 0.000 description 2
- 238000006471 dimerization reaction Methods 0.000 description 2
- PSLWZOIUBRXAQW-UHFFFAOYSA-M dimethyl(dioctadecyl)azanium;bromide Chemical compound [Br-].CCCCCCCCCCCCCCCCCC[N+](C)(C)CCCCCCCCCCCCCCCCCC PSLWZOIUBRXAQW-UHFFFAOYSA-M 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 229920002549 elastin Polymers 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 230000005284 excitation Effects 0.000 description 2
- 230000007717 exclusion Effects 0.000 description 2
- 101150098467 fnbA gene Proteins 0.000 description 2
- 239000000174 gluconic acid Substances 0.000 description 2
- 235000012208 gluconic acid Nutrition 0.000 description 2
- 229960002442 glucosamine Drugs 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 239000003102 growth factor Substances 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- 230000028996 humoral immune response Effects 0.000 description 2
- 150000002454 idoses Chemical class 0.000 description 2
- 239000005414 inactive ingredient Substances 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 210000005007 innate immune system Anatomy 0.000 description 2
- CDAISMWEOUEBRE-GPIVLXJGSA-N inositol Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@@H]1O CDAISMWEOUEBRE-GPIVLXJGSA-N 0.000 description 2
- 229960000367 inositol Drugs 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 229940047124 interferons Drugs 0.000 description 2
- 229940047122 interleukins Drugs 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 238000010255 intramuscular injection Methods 0.000 description 2
- 239000007927 intramuscular injection Substances 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 2
- 108020001756 ligand binding domains Proteins 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 210000001165 lymph node Anatomy 0.000 description 2
- RLSSMJSEOOYNOY-UHFFFAOYSA-N m-cresol Chemical compound CC1=CC=CC(O)=C1 RLSSMJSEOOYNOY-UHFFFAOYSA-N 0.000 description 2
- 229910052748 manganese Inorganic materials 0.000 description 2
- 239000011572 manganese Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 239000004005 microsphere Substances 0.000 description 2
- JMUHBNWAORSSBD-WKYWBUFDSA-N mifamurtide Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@@H](OC(=O)CCCCCCCCCCCCCCC)COP(O)(=O)OCCNC(=O)[C@H](C)NC(=O)CC[C@H](C(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](C)O[C@H]1[C@H](O)[C@@H](CO)OC(O)[C@@H]1NC(C)=O JMUHBNWAORSSBD-WKYWBUFDSA-N 0.000 description 2
- 229960005225 mifamurtide Drugs 0.000 description 2
- 229940035032 monophosphoryl lipid a Drugs 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- 239000007764 o/w emulsion Substances 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 2
- 230000001662 opsonic effect Effects 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 238000005457 optimization Methods 0.000 description 2
- 210000001672 ovary Anatomy 0.000 description 2
- 210000000496 pancreas Anatomy 0.000 description 2
- 244000052769 pathogen Species 0.000 description 2
- 210000004912 pericardial fluid Anatomy 0.000 description 2
- 229960003742 phenol Drugs 0.000 description 2
- 229960005323 phenoxyethanol Drugs 0.000 description 2
- 229940068196 placebo Drugs 0.000 description 2
- 239000000902 placebo Substances 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 229920003023 plastic Polymers 0.000 description 2
- 210000004910 pleural fluid Anatomy 0.000 description 2
- 108010040473 pneumococcal surface protein A Proteins 0.000 description 2
- 229920000747 poly(lactic acid) Polymers 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 2
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 2
- 229940068965 polysorbates Drugs 0.000 description 2
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 230000000069 prophylactic effect Effects 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 235000019419 proteases Nutrition 0.000 description 2
- 230000007026 protein scission Effects 0.000 description 2
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 238000001881 scanning electron acoustic microscopy Methods 0.000 description 2
- CDAISMWEOUEBRE-UHFFFAOYSA-N scyllo-inosotol Natural products OC1C(O)C(O)C(O)C(O)C1O CDAISMWEOUEBRE-UHFFFAOYSA-N 0.000 description 2
- 230000036303 septic shock Effects 0.000 description 2
- 206010040872 skin infection Diseases 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 238000010183 spectrum analysis Methods 0.000 description 2
- 210000000952 spleen Anatomy 0.000 description 2
- 230000006641 stabilisation Effects 0.000 description 2
- 238000013112 stability test Methods 0.000 description 2
- 238000011105 stabilization Methods 0.000 description 2
- 238000010972 statistical evaluation Methods 0.000 description 2
- 230000004936 stimulating effect Effects 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 229940031000 streptococcus pneumoniae Drugs 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 238000000859 sublimation Methods 0.000 description 2
- 230000008022 sublimation Effects 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 150000003890 succinate salts Chemical class 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-N succinic acid Chemical compound OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 2
- 150000005846 sugar alcohols Chemical class 0.000 description 2
- 230000001629 suppression Effects 0.000 description 2
- 210000001179 synovial fluid Anatomy 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 230000009885 systemic effect Effects 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 238000001757 thermogravimetry curve Methods 0.000 description 2
- RTKIYNMVFMVABJ-UHFFFAOYSA-L thimerosal Chemical compound [Na+].CC[Hg]SC1=CC=CC=C1C([O-])=O RTKIYNMVFMVABJ-UHFFFAOYSA-L 0.000 description 2
- 229940033663 thimerosal Drugs 0.000 description 2
- 238000004448 titration Methods 0.000 description 2
- 239000003053 toxin Substances 0.000 description 2
- 231100000765 toxin Toxicity 0.000 description 2
- 108700012359 toxins Proteins 0.000 description 2
- 150000004043 trisaccharides Chemical class 0.000 description 2
- 235000017103 tryptophane Nutrition 0.000 description 2
- 150000003654 tryptophanes Chemical class 0.000 description 2
- TVTGZVYLUHVBAJ-VFUOTHLCSA-N (2R,3R,4R,5R,6R)-3-amino-6-methyloxane-2,4,5-triol Chemical compound C[C@H]1O[C@@H](O)[C@H](N)[C@@H](O)[C@H]1O TVTGZVYLUHVBAJ-VFUOTHLCSA-N 0.000 description 1
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical class OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- OEANUJAFZLQYOD-CXAZCLJRSA-N (2r,3s,4r,5r,6r)-6-[(2r,3r,4r,5r,6r)-5-acetamido-3-hydroxy-2-(hydroxymethyl)-6-methoxyoxan-4-yl]oxy-4,5-dihydroxy-3-methoxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](OC)O[C@H](CO)[C@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](OC)[C@H](C(O)=O)O1 OEANUJAFZLQYOD-CXAZCLJRSA-N 0.000 description 1
- ZFTFOHBYVDOAMH-XNOIKFDKSA-N (2r,3s,4s,5r)-5-[[(2r,3s,4s,5r)-5-[[(2r,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)oxolan-2-yl]oxymethyl]-3,4-dihydroxy-2-(hydroxymethyl)oxolan-2-yl]oxymethyl]-2-(hydroxymethyl)oxolane-2,3,4-triol Chemical class O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)OC[C@@H]1[C@@H](O)[C@H](O)[C@](CO)(OC[C@@H]2[C@H]([C@H](O)[C@@](O)(CO)O2)O)O1 ZFTFOHBYVDOAMH-XNOIKFDKSA-N 0.000 description 1
- YHQZWWDVLJPRIF-JLHRHDQISA-N (4R)-4-[[(2S,3R)-2-[acetyl-[(3R,4R,5S,6R)-3-amino-4-[(1R)-1-carboxyethoxy]-5-hydroxy-6-(hydroxymethyl)oxan-2-yl]amino]-3-hydroxybutanoyl]amino]-5-amino-5-oxopentanoic acid Chemical compound C(C)(=O)N([C@@H]([C@H](O)C)C(=O)N[C@H](CCC(=O)O)C(N)=O)C1[C@H](N)[C@@H](O[C@@H](C(=O)O)C)[C@H](O)[C@H](O1)CO YHQZWWDVLJPRIF-JLHRHDQISA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- WPRAXAOJIODQJR-UHFFFAOYSA-N 1-(3,4-dimethylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(C)C(C)=C1 WPRAXAOJIODQJR-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- IDOQDZANRZQBTP-UHFFFAOYSA-N 2-[2-(2,4,4-trimethylpentan-2-yl)phenoxy]ethanol Chemical compound CC(C)(C)CC(C)(C)C1=CC=CC=C1OCCO IDOQDZANRZQBTP-UHFFFAOYSA-N 0.000 description 1
- NLMKTBGFQGKQEV-UHFFFAOYSA-N 2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-(2-hexadecoxyethoxy)ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethanol Chemical compound CCCCCCCCCCCCCCCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCO NLMKTBGFQGKQEV-UHFFFAOYSA-N 0.000 description 1
- IEQAICDLOKRSRL-UHFFFAOYSA-N 2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-[2-(2-dodecoxyethoxy)ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethanol Chemical compound CCCCCCCCCCCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCO IEQAICDLOKRSRL-UHFFFAOYSA-N 0.000 description 1
- MSWZFWKMSRAUBD-GASJEMHNSA-N 2-amino-2-deoxy-D-galactopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@H](O)[C@@H]1O MSWZFWKMSRAUBD-GASJEMHNSA-N 0.000 description 1
- QCVGEOXPDFCNHA-UHFFFAOYSA-N 5,5-dimethyl-2,4-dioxo-1,3-oxazolidine-3-carboxamide Chemical compound CC1(C)OC(=O)N(C(N)=O)C1=O QCVGEOXPDFCNHA-UHFFFAOYSA-N 0.000 description 1
- QWVRTSZDKPRPDF-UHFFFAOYSA-N 5-(piperidin-1-ylmethyl)-3-pyridin-3-yl-5,6-dihydro-2h-1,2,4-oxadiazine Chemical compound C1CCCCN1CC(N=1)CONC=1C1=CC=CN=C1 QWVRTSZDKPRPDF-UHFFFAOYSA-N 0.000 description 1
- 208000030507 AIDS Diseases 0.000 description 1
- 206010000060 Abdominal distension Diseases 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- 101710126837 Alpha-2-macroglobulin receptor-associated protein Proteins 0.000 description 1
- 229920000945 Amylopectin Polymers 0.000 description 1
- 229920000856 Amylose Polymers 0.000 description 1
- 235000002198 Annona diversifolia Nutrition 0.000 description 1
- 241000272517 Anseriformes Species 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- 241000271566 Aves Species 0.000 description 1
- 241000193738 Bacillus anthracis Species 0.000 description 1
- 101000743092 Bacillus spizizenii (strain DSM 15029 / JCM 12233 / NBRC 101239 / NRRL B-23049 / TU-B-10) tRNA3(Ser)-specific nuclease WapA Proteins 0.000 description 1
- 101000743093 Bacillus subtilis subsp. natto (strain BEST195) tRNA(Glu)-specific nuclease WapA Proteins 0.000 description 1
- 108010071023 Bacterial Outer Membrane Proteins Proteins 0.000 description 1
- 231100000699 Bacterial toxin Toxicity 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- 102100023995 Beta-nerve growth factor Human genes 0.000 description 1
- 241000157302 Bison bison athabascae Species 0.000 description 1
- 102000004506 Blood Proteins Human genes 0.000 description 1
- 108010017384 Blood Proteins Proteins 0.000 description 1
- 241000588807 Bordetella Species 0.000 description 1
- 241001416153 Bos grunniens Species 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 241000269420 Bufonidae Species 0.000 description 1
- 102100021943 C-C motif chemokine 2 Human genes 0.000 description 1
- 101710155857 C-C motif chemokine 2 Proteins 0.000 description 1
- 102100032367 C-C motif chemokine 5 Human genes 0.000 description 1
- 108700012434 CCL3 Proteins 0.000 description 1
- 108010029697 CD40 Ligand Proteins 0.000 description 1
- 101150013553 CD40 gene Proteins 0.000 description 1
- 102100032937 CD40 ligand Human genes 0.000 description 1
- 108010084313 CD58 Antigens Proteins 0.000 description 1
- 101100289995 Caenorhabditis elegans mac-1 gene Proteins 0.000 description 1
- 101100065878 Caenorhabditis elegans sec-10 gene Proteins 0.000 description 1
- 101100172879 Caenorhabditis elegans sec-5 gene Proteins 0.000 description 1
- 101100172886 Caenorhabditis elegans sec-6 gene Proteins 0.000 description 1
- 101100172892 Caenorhabditis elegans sec-8 gene Proteins 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 102000011727 Caspases Human genes 0.000 description 1
- 108010076667 Caspases Proteins 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 241000282994 Cervidae Species 0.000 description 1
- 102000000013 Chemokine CCL3 Human genes 0.000 description 1
- 102000001326 Chemokine CCL4 Human genes 0.000 description 1
- 108010055165 Chemokine CCL4 Proteins 0.000 description 1
- 108010055166 Chemokine CCL5 Proteins 0.000 description 1
- 241001227713 Chiron Species 0.000 description 1
- 229920002101 Chitin Polymers 0.000 description 1
- 108010049048 Cholera Toxin Proteins 0.000 description 1
- 102000009016 Cholera Toxin Human genes 0.000 description 1
- 241000251730 Chondrichthyes Species 0.000 description 1
- 229920002567 Chondroitin Polymers 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 241000193163 Clostridioides difficile Species 0.000 description 1
- 108010065152 Coagulase Proteins 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 108091035707 Consensus sequence Proteins 0.000 description 1
- 241000186216 Corynebacterium Species 0.000 description 1
- 241000186227 Corynebacterium diphtheriae Species 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 206010011409 Cross infection Diseases 0.000 description 1
- YTBSYETUWUMLBZ-UHFFFAOYSA-N D-Erythrose Natural products OCC(O)C(O)C=O YTBSYETUWUMLBZ-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-CBPJZXOFSA-N D-Gulose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@H](O)[C@H]1O WQZGKKKJIJFFOK-CBPJZXOFSA-N 0.000 description 1
- WQZGKKKJIJFFOK-IVMDWMLBSA-N D-allopyranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@H](O)[C@@H]1O WQZGKKKJIJFFOK-IVMDWMLBSA-N 0.000 description 1
- YTBSYETUWUMLBZ-IUYQGCFVSA-N D-erythrose Chemical compound OC[C@@H](O)[C@@H](O)C=O YTBSYETUWUMLBZ-IUYQGCFVSA-N 0.000 description 1
- DSLZVSRJTYRBFB-LLEIAEIESA-N D-glucaric acid Chemical compound OC(=O)[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O DSLZVSRJTYRBFB-LLEIAEIESA-N 0.000 description 1
- MNQZXJOMYWMBOU-VKHMYHEASA-N D-glyceraldehyde Chemical compound OC[C@@H](O)C=O MNQZXJOMYWMBOU-VKHMYHEASA-N 0.000 description 1
- CIWBSHSKHKDKBQ-DUZGATOHSA-N D-isoascorbic acid Chemical compound OC[C@@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-DUZGATOHSA-N 0.000 description 1
- SHZGCJCMOBCMKK-UHFFFAOYSA-N D-mannomethylose Natural products CC1OC(O)C(O)C(O)C1O SHZGCJCMOBCMKK-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- AEMOLEFTQBMNLQ-VANFPWTGSA-N D-mannopyranuronic acid Chemical compound OC1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@@H]1O AEMOLEFTQBMNLQ-VANFPWTGSA-N 0.000 description 1
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 1
- ZAQJHHRNXZUBTE-NQXXGFSBSA-N D-ribulose Chemical compound OC[C@@H](O)[C@@H](O)C(=O)CO ZAQJHHRNXZUBTE-NQXXGFSBSA-N 0.000 description 1
- ZAQJHHRNXZUBTE-UHFFFAOYSA-N D-threo-2-Pentulose Natural products OCC(O)C(O)C(=O)CO ZAQJHHRNXZUBTE-UHFFFAOYSA-N 0.000 description 1
- YTBSYETUWUMLBZ-QWWZWVQMSA-N D-threose Chemical compound OC[C@@H](O)[C@H](O)C=O YTBSYETUWUMLBZ-QWWZWVQMSA-N 0.000 description 1
- 102100037840 Dehydrogenase/reductase SDR family member 2, mitochondrial Human genes 0.000 description 1
- 229920000045 Dermatan sulfate Polymers 0.000 description 1
- 101710088341 Dermatopontin Proteins 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- 108010053187 Diphtheria Toxin Proteins 0.000 description 1
- 102000016607 Diphtheria Toxin Human genes 0.000 description 1
- 108010024212 E-Selectin Proteins 0.000 description 1
- 102100023471 E-selectin Human genes 0.000 description 1
- 238000012286 ELISA Assay Methods 0.000 description 1
- 102000002322 Egg Proteins Human genes 0.000 description 1
- 108010000912 Egg Proteins Proteins 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 102400001368 Epidermal growth factor Human genes 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 241000283074 Equus asinus Species 0.000 description 1
- 241001331845 Equus asinus x caballus Species 0.000 description 1
- 206010056474 Erythrosis Diseases 0.000 description 1
- 101710082714 Exotoxin A Proteins 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 108010073385 Fibrin Proteins 0.000 description 1
- 102000009123 Fibrin Human genes 0.000 description 1
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 1
- 108050007372 Fibroblast Growth Factor Proteins 0.000 description 1
- 102000018233 Fibroblast Growth Factor Human genes 0.000 description 1
- 229920002670 Fructan Polymers 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 229920000855 Fucoidan Polymers 0.000 description 1
- PNNNRSAQSRJVSB-SLPGGIOYSA-N Fucose Natural products C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C=O PNNNRSAQSRJVSB-SLPGGIOYSA-N 0.000 description 1
- BXEARCKJAZWJTJ-IJCVXDJZSA-N Galactocarolose Natural products OC[C@H](O)[C@@H]1O[C@@H](O[C@H](CO)[C@@H]2O[C@@H](O[C@H](CO)[C@@H]3O[C@@H](O)[C@H](O)[C@@H]3O)[C@H](O)[C@@H]2O)[C@H](O)[C@@H]1O BXEARCKJAZWJTJ-IJCVXDJZSA-N 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 241000237858 Gastropoda Species 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 241000699694 Gerbillinae Species 0.000 description 1
- 229920001503 Glucan Polymers 0.000 description 1
- 108010024636 Glutathione Proteins 0.000 description 1
- 229920002527 Glycogen Polymers 0.000 description 1
- 206010053240 Glycogen storage disease type VI Diseases 0.000 description 1
- 239000006173 Good's buffer Substances 0.000 description 1
- 108060003393 Granulin Proteins 0.000 description 1
- 102100031573 Hematopoietic progenitor cell antigen CD34 Human genes 0.000 description 1
- 101000777663 Homo sapiens Hematopoietic progenitor cell antigen CD34 Proteins 0.000 description 1
- 101000934346 Homo sapiens T-cell surface antigen CD2 Proteins 0.000 description 1
- 101100100117 Homo sapiens TNFRSF10B gene Proteins 0.000 description 1
- 101000679921 Homo sapiens Tumor necrosis factor receptor superfamily member 21 Proteins 0.000 description 1
- IMQLKJBTEOYOSI-GPIVLXJGSA-N Inositol-hexakisphosphate Chemical compound OP(O)(=O)O[C@H]1[C@H](OP(O)(O)=O)[C@@H](OP(O)(O)=O)[C@H](OP(O)(O)=O)[C@H](OP(O)(O)=O)[C@@H]1OP(O)(O)=O IMQLKJBTEOYOSI-GPIVLXJGSA-N 0.000 description 1
- 102100025323 Integrin alpha-1 Human genes 0.000 description 1
- 102100022339 Integrin alpha-L Human genes 0.000 description 1
- 108010041341 Integrin alpha1 Proteins 0.000 description 1
- 108010055795 Integrin alpha1beta1 Proteins 0.000 description 1
- 108010064593 Intercellular Adhesion Molecule-1 Proteins 0.000 description 1
- 108010064600 Intercellular Adhesion Molecule-3 Proteins 0.000 description 1
- 102100037877 Intercellular adhesion molecule 1 Human genes 0.000 description 1
- 102100037872 Intercellular adhesion molecule 2 Human genes 0.000 description 1
- 101710148794 Intercellular adhesion molecule 2 Proteins 0.000 description 1
- 102100037871 Intercellular adhesion molecule 3 Human genes 0.000 description 1
- 229920001202 Inulin Polymers 0.000 description 1
- 208000007976 Ketosis Diseases 0.000 description 1
- LKDRXBCSQODPBY-AMVSKUEXSA-N L-(-)-Sorbose Chemical compound OCC1(O)OC[C@H](O)[C@@H](O)[C@@H]1O LKDRXBCSQODPBY-AMVSKUEXSA-N 0.000 description 1
- PWKSKIMOESPYIA-BYPYZUCNSA-N L-N-acetyl-Cysteine Chemical compound CC(=O)N[C@@H](CS)C(O)=O PWKSKIMOESPYIA-BYPYZUCNSA-N 0.000 description 1
- 108010092694 L-Selectin Proteins 0.000 description 1
- WQZGKKKJIJFFOK-VSOAQEOCSA-N L-altropyranose Chemical compound OC[C@@H]1OC(O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-VSOAQEOCSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- SHZGCJCMOBCMKK-DHVFOXMCSA-N L-fucopyranose Chemical compound C[C@@H]1OC(O)[C@@H](O)[C@H](O)[C@@H]1O SHZGCJCMOBCMKK-DHVFOXMCSA-N 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- 102100033467 L-selectin Human genes 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- 241000282838 Lama Species 0.000 description 1
- 108010085895 Laminin Proteins 0.000 description 1
- 102000007547 Laminin Human genes 0.000 description 1
- 108010000851 Laminin Receptors Proteins 0.000 description 1
- 102000002297 Laminin Receptors Human genes 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 238000003231 Lowry assay Methods 0.000 description 1
- 238000009013 Lowry's assay Methods 0.000 description 1
- 108010064548 Lymphocyte Function-Associated Antigen-1 Proteins 0.000 description 1
- 102000008072 Lymphokines Human genes 0.000 description 1
- 108010074338 Lymphokines Proteins 0.000 description 1
- 241000282560 Macaca mulatta Species 0.000 description 1
- 229920000057 Mannan Polymers 0.000 description 1
- 108010060408 Member 25 Tumor Necrosis Factor Receptors Proteins 0.000 description 1
- RJQXTJLFIWVMTO-TYNCELHUSA-N Methicillin Chemical compound COC1=CC=CC(OC)=C1C(=O)N[C@@H]1C(=O)N2[C@@H](C(O)=O)C(C)(C)S[C@@H]21 RJQXTJLFIWVMTO-TYNCELHUSA-N 0.000 description 1
- 241000282339 Mustela Species 0.000 description 1
- 241000187479 Mycobacterium tuberculosis Species 0.000 description 1
- QPCDCPDFJACHGM-UHFFFAOYSA-N N,N-bis{2-[bis(carboxymethyl)amino]ethyl}glycine Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(=O)O)CCN(CC(O)=O)CC(O)=O QPCDCPDFJACHGM-UHFFFAOYSA-N 0.000 description 1
- GXCLVBGFBYZDAG-UHFFFAOYSA-N N-[2-(1H-indol-3-yl)ethyl]-N-methylprop-2-en-1-amine Chemical compound CN(CCC1=CNC2=C1C=CC=C2)CC=C GXCLVBGFBYZDAG-UHFFFAOYSA-N 0.000 description 1
- 125000003047 N-acetyl group Chemical group 0.000 description 1
- OVRNDRQMDRJTHS-FMDGEEDCSA-N N-acetyl-beta-D-glucosamine Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-FMDGEEDCSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- 108010025020 Nerve Growth Factor Proteins 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 108010035766 P-Selectin Proteins 0.000 description 1
- 102100023472 P-selectin Human genes 0.000 description 1
- 101150044441 PECAM1 gene Proteins 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 229920002230 Pectic acid Polymers 0.000 description 1
- 102000007079 Peptide Fragments Human genes 0.000 description 1
- 108010033276 Peptide Fragments Proteins 0.000 description 1
- 201000005702 Pertussis Diseases 0.000 description 1
- 241000286209 Phasianidae Species 0.000 description 1
- IMQLKJBTEOYOSI-UHFFFAOYSA-N Phytic acid Natural products OP(O)(=O)OC1C(OP(O)(O)=O)C(OP(O)(O)=O)C(OP(O)(O)=O)C(OP(O)(O)=O)C1OP(O)(O)=O IMQLKJBTEOYOSI-UHFFFAOYSA-N 0.000 description 1
- 208000000474 Poliomyelitis Diseases 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920000954 Polyglycolide Polymers 0.000 description 1
- 229920001219 Polysorbate 40 Polymers 0.000 description 1
- 102000017033 Porins Human genes 0.000 description 1
- 108010013381 Porins Proteins 0.000 description 1
- 101710188053 Protein D Proteins 0.000 description 1
- 102100036352 Protein disulfide-isomerase Human genes 0.000 description 1
- 241000589516 Pseudomonas Species 0.000 description 1
- 241000589517 Pseudomonas aeruginosa Species 0.000 description 1
- 208000028017 Psychotic disease Diseases 0.000 description 1
- 229920001218 Pullulan Polymers 0.000 description 1
- 239000004373 Pullulan Substances 0.000 description 1
- 102100028688 Putative glycosylation-dependent cell adhesion molecule 1 Human genes 0.000 description 1
- 206010037660 Pyrexia Diseases 0.000 description 1
- 206010037742 Rabies Diseases 0.000 description 1
- 241000283011 Rangifer Species 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 208000035415 Reinfection Diseases 0.000 description 1
- 108091081062 Repeated sequence (DNA) Proteins 0.000 description 1
- 101710132893 Resolvase Proteins 0.000 description 1
- 108010073443 Ribi adjuvant Proteins 0.000 description 1
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 1
- 240000005499 Sasa Species 0.000 description 1
- 108090000184 Selectins Proteins 0.000 description 1
- 102000003800 Selectins Human genes 0.000 description 1
- VBKBDLMWICBSCY-IMJSIDKUSA-N Ser-Asp Chemical group OC[C@H](N)C(=O)N[C@H](C(O)=O)CC(O)=O VBKBDLMWICBSCY-IMJSIDKUSA-N 0.000 description 1
- 241000270295 Serpentes Species 0.000 description 1
- 102100027287 Serpin H1 Human genes 0.000 description 1
- 108050008290 Serpin H1 Proteins 0.000 description 1
- 101710084578 Short neurotoxin 1 Proteins 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- PRXRUNOAOLTIEF-ADSICKODSA-N Sorbitan trioleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@@H](OC(=O)CCCCCCC\C=C/CCCCCCCC)[C@H]1OC[C@H](O)[C@H]1OC(=O)CCCCCCC\C=C/CCCCCCCC PRXRUNOAOLTIEF-ADSICKODSA-N 0.000 description 1
- 101100532872 Staphylococcus aureus (strain MSSA476) sdrD gene Proteins 0.000 description 1
- 101100532873 Staphylococcus aureus (strain MW2) sdrD gene Proteins 0.000 description 1
- 101100532870 Staphylococcus aureus (strain Mu50 / ATCC 700699) sdrD gene Proteins 0.000 description 1
- 101100532867 Staphylococcus aureus (strain NCTC 8325 / PS 47) sdrD gene Proteins 0.000 description 1
- 101100532866 Staphylococcus aureus (strain USA300) sdrD gene Proteins 0.000 description 1
- 241000191965 Staphylococcus carnosus Species 0.000 description 1
- 241000751182 Staphylococcus epidermidis ATCC 12228 Species 0.000 description 1
- 241000495427 Staphylococcus haemolyticus JCSC1435 Species 0.000 description 1
- 241001147691 Staphylococcus saprophyticus Species 0.000 description 1
- 241000191973 Staphylococcus xylosus Species 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- 241000194017 Streptococcus Species 0.000 description 1
- 241000193991 Streptococcus parasanguinis Species 0.000 description 1
- 108010008038 Synthetic Vaccines Proteins 0.000 description 1
- 230000024932 T cell mediated immunity Effects 0.000 description 1
- 108091008874 T cell receptors Proteins 0.000 description 1
- 230000005867 T cell response Effects 0.000 description 1
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 1
- 102100025237 T-cell surface antigen CD2 Human genes 0.000 description 1
- 241000270666 Testudines Species 0.000 description 1
- 206010043376 Tetanus Diseases 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- 101710182223 Toxin B Proteins 0.000 description 1
- 101710182532 Toxin a Proteins 0.000 description 1
- 102000010912 Transferrin-Binding Proteins Human genes 0.000 description 1
- 108010062476 Transferrin-Binding Proteins Proteins 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 239000013504 Triton X-100 Substances 0.000 description 1
- 229920004890 Triton X-100 Polymers 0.000 description 1
- 229920004929 Triton X-114 Polymers 0.000 description 1
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 1
- 108060008683 Tumor Necrosis Factor Receptor Proteins 0.000 description 1
- 102100022205 Tumor necrosis factor receptor superfamily member 21 Human genes 0.000 description 1
- 102100040245 Tumor necrosis factor receptor superfamily member 5 Human genes 0.000 description 1
- 208000037386 Typhoid Diseases 0.000 description 1
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 1
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 description 1
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 1
- 102100039037 Vascular endothelial growth factor A Human genes 0.000 description 1
- 241001416177 Vicugna pacos Species 0.000 description 1
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 229960004308 acetylcysteine Drugs 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 230000002730 additional effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000004931 aggregating effect Effects 0.000 description 1
- 150000001294 alanine derivatives Chemical class 0.000 description 1
- IAJILQKETJEXLJ-RSJOWCBRSA-N aldehydo-D-galacturonic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-RSJOWCBRSA-N 0.000 description 1
- IAJILQKETJEXLJ-QTBDOELSSA-N aldehydo-D-glucuronic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-QTBDOELSSA-N 0.000 description 1
- 150000001323 aldoses Chemical class 0.000 description 1
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- SRBFZHDQGSBBOR-STGXQOJASA-N alpha-D-lyxopyranose Chemical compound O[C@@H]1CO[C@H](O)[C@@H](O)[C@H]1O SRBFZHDQGSBBOR-STGXQOJASA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 229940024545 aluminum hydroxide Drugs 0.000 description 1
- 229940024546 aluminum hydroxide gel Drugs 0.000 description 1
- SMYKVLBUSSNXMV-UHFFFAOYSA-K aluminum;trihydroxide;hydrate Chemical compound O.[OH-].[OH-].[OH-].[Al+3] SMYKVLBUSSNXMV-UHFFFAOYSA-K 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 125000004103 aminoalkyl group Chemical group 0.000 description 1
- 238000012870 ammonium sulfate precipitation Methods 0.000 description 1
- 230000003466 anti-cipated effect Effects 0.000 description 1
- 230000010056 antibody-dependent cellular cytotoxicity Effects 0.000 description 1
- 210000001765 aortic valve Anatomy 0.000 description 1
- 238000000149 argon plasma sintering Methods 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 229940072107 ascorbate Drugs 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- WXNRAKRZUCLRBP-UHFFFAOYSA-N avridine Chemical compound CCCCCCCCCCCCCCCCCCN(CCCN(CCO)CCO)CCCCCCCCCCCCCCCCCC WXNRAKRZUCLRBP-UHFFFAOYSA-N 0.000 description 1
- 229950010555 avridine Drugs 0.000 description 1
- 239000000688 bacterial toxin Substances 0.000 description 1
- 229960001212 bacterial vaccine Drugs 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 229960004365 benzoic acid Drugs 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 238000002306 biochemical method Methods 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 230000031018 biological processes and functions Effects 0.000 description 1
- 229960000074 biopharmaceutical Drugs 0.000 description 1
- 238000013378 biophysical characterization Methods 0.000 description 1
- OWMVSZAMULFTJU-UHFFFAOYSA-N bis-tris Chemical compound OCCN(CCO)C(CO)(CO)CO OWMVSZAMULFTJU-UHFFFAOYSA-N 0.000 description 1
- 208000024330 bloating Diseases 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 230000005587 bubbling Effects 0.000 description 1
- 235000019846 buffering salt Nutrition 0.000 description 1
- 229940041514 candida albicans extract Drugs 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 239000000679 carrageenan Substances 0.000 description 1
- 229940113118 carrageenan Drugs 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 238000001516 cell proliferation assay Methods 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000002144 chemical decomposition reaction Methods 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- 108010031071 cholera toxoid Proteins 0.000 description 1
- DLGJWSVWTWEWBJ-HGGSSLSASA-N chondroitin Chemical compound CC(O)=N[C@@H]1[C@H](O)O[C@H](CO)[C@H](O)[C@@H]1OC1[C@H](O)[C@H](O)C=C(C(O)=O)O1 DLGJWSVWTWEWBJ-HGGSSLSASA-N 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 238000000978 circular dichroism spectroscopy Methods 0.000 description 1
- 238000001142 circular dichroism spectrum Methods 0.000 description 1
- 101150085107 clfA gene Proteins 0.000 description 1
- 238000003759 clinical diagnosis Methods 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 238000012937 correction Methods 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 238000003869 coulometry Methods 0.000 description 1
- 230000009260 cross reactivity Effects 0.000 description 1
- 230000016396 cytokine production Effects 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 238000011266 cytolytic assay Methods 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 238000007405 data analysis Methods 0.000 description 1
- 238000013480 data collection Methods 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 238000011118 depth filtration Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 238000011026 diafiltration Methods 0.000 description 1
- 238000002405 diagnostic procedure Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 230000001079 digestive effect Effects 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 150000002016 disaccharides Chemical class 0.000 description 1
- 231100000676 disease causative agent Toxicity 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- PZZHMLOHNYWKIK-UHFFFAOYSA-N eddha Chemical compound C=1C=CC=C(O)C=1C(C(=O)O)NCCNC(C(O)=O)C1=CC=CC=C1O PZZHMLOHNYWKIK-UHFFFAOYSA-N 0.000 description 1
- 235000014103 egg white Nutrition 0.000 description 1
- 210000000969 egg white Anatomy 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 238000001663 electronic absorption spectrum Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 238000000295 emission spectrum Methods 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 230000009088 enzymatic function Effects 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 235000010350 erythorbic acid Nutrition 0.000 description 1
- UQPHVQVXLPRNCX-UHFFFAOYSA-N erythrulose Chemical compound OCC(O)C(=O)CO UQPHVQVXLPRNCX-UHFFFAOYSA-N 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 210000002744 extracellular matrix Anatomy 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 229950003499 fibrin Drugs 0.000 description 1
- 229940126864 fibroblast growth factor Drugs 0.000 description 1
- 102000036072 fibronectin binding proteins Human genes 0.000 description 1
- 108091010988 fibronectin binding proteins Proteins 0.000 description 1
- 239000013020 final formulation Substances 0.000 description 1
- 235000013312 flour Nutrition 0.000 description 1
- 238000012921 fluorescence analysis Methods 0.000 description 1
- 238000002189 fluorescence spectrum Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 238000005187 foaming Methods 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 229940097043 glucuronic acid Drugs 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 235000004554 glutamine Nutrition 0.000 description 1
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 description 1
- YQEMORVAKMFKLG-UHFFFAOYSA-N glycerine monostearate Natural products CCCCCCCCCCCCCCCCCC(=O)OC(CO)CO YQEMORVAKMFKLG-UHFFFAOYSA-N 0.000 description 1
- SVUQHVRAGMNPLW-UHFFFAOYSA-N glycerol monostearate Natural products CCCCCCCCCCCCCCCCC(=O)OCC(O)CO SVUQHVRAGMNPLW-UHFFFAOYSA-N 0.000 description 1
- 229940096919 glycogen Drugs 0.000 description 1
- 150000002337 glycosamines Chemical class 0.000 description 1
- 229930182470 glycoside Natural products 0.000 description 1
- 150000002338 glycosides Chemical class 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- 238000009499 grossing Methods 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 210000005003 heart tissue Anatomy 0.000 description 1
- 108060003552 hemocyanin Proteins 0.000 description 1
- 108010037896 heparin-binding hemagglutinin Proteins 0.000 description 1
- 150000002386 heptoses Chemical class 0.000 description 1
- 150000002402 hexoses Chemical class 0.000 description 1
- 230000004727 humoral immunity Effects 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- 210000004408 hybridoma Anatomy 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 230000002519 immonomodulatory effect Effects 0.000 description 1
- 230000017555 immunoglobulin mediated immune response Effects 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 238000010324 immunological assay Methods 0.000 description 1
- 230000002584 immunomodulator Effects 0.000 description 1
- 238000001114 immunoprecipitation Methods 0.000 description 1
- 230000003308 immunostimulating effect Effects 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 206010022000 influenza Diseases 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 102000006495 integrins Human genes 0.000 description 1
- 108010044426 integrins Proteins 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 229940029339 inulin Drugs 0.000 description 1
- JYJIGFIDKWBXDU-MNNPPOADSA-N inulin Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)OC[C@]1(OC[C@]2(OC[C@]3(OC[C@]4(OC[C@]5(OC[C@]6(OC[C@]7(OC[C@]8(OC[C@]9(OC[C@]%10(OC[C@]%11(OC[C@]%12(OC[C@]%13(OC[C@]%14(OC[C@]%15(OC[C@]%16(OC[C@]%17(OC[C@]%18(OC[C@]%19(OC[C@]%20(OC[C@]%21(OC[C@]%22(OC[C@]%23(OC[C@]%24(OC[C@]%25(OC[C@]%26(OC[C@]%27(OC[C@]%28(OC[C@]%29(OC[C@]%30(OC[C@]%31(OC[C@]%32(OC[C@]%33(OC[C@]%34(OC[C@]%35(OC[C@]%36(O[C@@H]%37[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O%37)O)[C@H]([C@H](O)[C@@H](CO)O%36)O)[C@H]([C@H](O)[C@@H](CO)O%35)O)[C@H]([C@H](O)[C@@H](CO)O%34)O)[C@H]([C@H](O)[C@@H](CO)O%33)O)[C@H]([C@H](O)[C@@H](CO)O%32)O)[C@H]([C@H](O)[C@@H](CO)O%31)O)[C@H]([C@H](O)[C@@H](CO)O%30)O)[C@H]([C@H](O)[C@@H](CO)O%29)O)[C@H]([C@H](O)[C@@H](CO)O%28)O)[C@H]([C@H](O)[C@@H](CO)O%27)O)[C@H]([C@H](O)[C@@H](CO)O%26)O)[C@H]([C@H](O)[C@@H](CO)O%25)O)[C@H]([C@H](O)[C@@H](CO)O%24)O)[C@H]([C@H](O)[C@@H](CO)O%23)O)[C@H]([C@H](O)[C@@H](CO)O%22)O)[C@H]([C@H](O)[C@@H](CO)O%21)O)[C@H]([C@H](O)[C@@H](CO)O%20)O)[C@H]([C@H](O)[C@@H](CO)O%19)O)[C@H]([C@H](O)[C@@H](CO)O%18)O)[C@H]([C@H](O)[C@@H](CO)O%17)O)[C@H]([C@H](O)[C@@H](CO)O%16)O)[C@H]([C@H](O)[C@@H](CO)O%15)O)[C@H]([C@H](O)[C@@H](CO)O%14)O)[C@H]([C@H](O)[C@@H](CO)O%13)O)[C@H]([C@H](O)[C@@H](CO)O%12)O)[C@H]([C@H](O)[C@@H](CO)O%11)O)[C@H]([C@H](O)[C@@H](CO)O%10)O)[C@H]([C@H](O)[C@@H](CO)O9)O)[C@H]([C@H](O)[C@@H](CO)O8)O)[C@H]([C@H](O)[C@@H](CO)O7)O)[C@H]([C@H](O)[C@@H](CO)O6)O)[C@H]([C@H](O)[C@@H](CO)O5)O)[C@H]([C@H](O)[C@@H](CO)O4)O)[C@H]([C@H](O)[C@@H](CO)O3)O)[C@H]([C@H](O)[C@@H](CO)O2)O)[C@@H](O)[C@H](O)[C@@H](CO)O1 JYJIGFIDKWBXDU-MNNPPOADSA-N 0.000 description 1
- 229940026239 isoascorbic acid Drugs 0.000 description 1
- 229960000310 isoleucine Drugs 0.000 description 1
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 1
- BJHIKXHVCXFQLS-PQLUHFTBSA-N keto-D-tagatose Chemical compound OC[C@@H](O)[C@H](O)[C@H](O)C(=O)CO BJHIKXHVCXFQLS-PQLUHFTBSA-N 0.000 description 1
- BQINXKOTJQCISL-GRCPKETISA-N keto-neuraminic acid Chemical compound OC(=O)C(=O)C[C@H](O)[C@@H](N)[C@@H](O)[C@H](O)[C@H](O)CO BQINXKOTJQCISL-GRCPKETISA-N 0.000 description 1
- 150000002584 ketoses Chemical class 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 231100000636 lethal dose Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- AIHDCSAXVMAMJH-GFBKWZILSA-N levan Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)OC[C@@H]1[C@@H](O)[C@H](O)[C@](CO)(CO[C@@H]2[C@H]([C@H](O)[C@@](O)(CO)O2)O)O1 AIHDCSAXVMAMJH-GFBKWZILSA-N 0.000 description 1
- 238000012417 linear regression Methods 0.000 description 1
- GZQKNULLWNGMCW-PWQABINMSA-N lipid A (E. coli) Chemical class O1[C@H](CO)[C@@H](OP(O)(O)=O)[C@H](OC(=O)C[C@@H](CCCCCCCCCCC)OC(=O)CCCCCCCCCCCCC)[C@@H](NC(=O)C[C@@H](CCCCCCCCCCC)OC(=O)CCCCCCCCCCC)[C@@H]1OC[C@@H]1[C@@H](O)[C@H](OC(=O)C[C@H](O)CCCCCCCCCCC)[C@@H](NC(=O)C[C@H](O)CCCCCCCCCCC)[C@@H](OP(O)(O)=O)O1 GZQKNULLWNGMCW-PWQABINMSA-N 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 239000012669 liquid formulation Substances 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 239000012263 liquid product Substances 0.000 description 1
- 239000006193 liquid solution Substances 0.000 description 1
- 239000006194 liquid suspension Substances 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 210000002751 lymph Anatomy 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 238000002794 lymphocyte assay Methods 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229960002160 maltose Drugs 0.000 description 1
- LUEWUZLMQUOBSB-GFVSVBBRSA-N mannan Chemical class O[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@@H](O[C@@H]2[C@H](O[C@@H](O[C@H]3[C@H](O[C@@H](O)[C@@H](O)[C@H]3O)CO)[C@@H](O)[C@H]2O)CO)[C@H](O)[C@H]1O LUEWUZLMQUOBSB-GFVSVBBRSA-N 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- 210000002418 meninge Anatomy 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 229960003085 meticillin Drugs 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- PJUIMOJAAPLTRJ-UHFFFAOYSA-N monothioglycerol Chemical compound OCC(O)CS PJUIMOJAAPLTRJ-UHFFFAOYSA-N 0.000 description 1
- 210000000214 mouth Anatomy 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 125000001446 muramyl group Chemical group N[C@@H](C=O)[C@@H](O[C@@H](C(=O)*)C)[C@H](O)[C@H](O)CO 0.000 description 1
- 229950006780 n-acetylglucosamine Drugs 0.000 description 1
- 229940053128 nerve growth factor Drugs 0.000 description 1
- CERZMXAJYMMUDR-UHFFFAOYSA-N neuraminic acid Natural products NC1C(O)CC(O)(C(O)=O)OC1C(O)C(O)CO CERZMXAJYMMUDR-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 238000006384 oligomerization reaction Methods 0.000 description 1
- 230000014207 opsonization Effects 0.000 description 1
- 230000003571 opsonizing effect Effects 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 150000003961 organosilicon compounds Chemical class 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000006174 pH buffer Substances 0.000 description 1
- 239000006179 pH buffering agent Substances 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 230000009589 pathological growth Effects 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 150000002972 pentoses Chemical class 0.000 description 1
- 230000002688 persistence Effects 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 229940124531 pharmaceutical excipient Drugs 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 229940068041 phytic acid Drugs 0.000 description 1
- 238000002264 polyacrylamide gel electrophoresis Methods 0.000 description 1
- 229920000447 polyanionic polymer Polymers 0.000 description 1
- 239000000249 polyoxyethylene sorbitan monopalmitate Substances 0.000 description 1
- 235000010483 polyoxyethylene sorbitan monopalmitate Nutrition 0.000 description 1
- 239000001818 polyoxyethylene sorbitan monostearate Substances 0.000 description 1
- 235000010989 polyoxyethylene sorbitan monostearate Nutrition 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229940068977 polysorbate 20 Drugs 0.000 description 1
- 229940101027 polysorbate 40 Drugs 0.000 description 1
- 229940113124 polysorbate 60 Drugs 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 230000008092 positive effect Effects 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- OTYBMLCTZGSZBG-UHFFFAOYSA-L potassium sulfate Chemical compound [K+].[K+].[O-]S([O-])(=O)=O OTYBMLCTZGSZBG-UHFFFAOYSA-L 0.000 description 1
- 229910052939 potassium sulfate Inorganic materials 0.000 description 1
- 235000011151 potassium sulphates Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 229940071643 prefilled syringe Drugs 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- AAEVYOVXGOFMJO-UHFFFAOYSA-N prometryn Chemical compound CSC1=NC(NC(C)C)=NC(NC(C)C)=N1 AAEVYOVXGOFMJO-UHFFFAOYSA-N 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 230000002633 protecting effect Effects 0.000 description 1
- 230000004845 protein aggregation Effects 0.000 description 1
- 238000002731 protein assay Methods 0.000 description 1
- 108020003519 protein disulfide isomerase Proteins 0.000 description 1
- 230000012846 protein folding Effects 0.000 description 1
- 230000006318 protein oxidation Effects 0.000 description 1
- 230000006432 protein unfolding Effects 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000005180 public health Effects 0.000 description 1
- 235000019423 pullulan Nutrition 0.000 description 1
- 230000002685 pulmonary effect Effects 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 239000002510 pyrogen Substances 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 230000002285 radioactive effect Effects 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 230000001718 repressive effect Effects 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 238000004366 reverse phase liquid chromatography Methods 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 238000013374 right angle light scattering Methods 0.000 description 1
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical class O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 208000013223 septicemia Diseases 0.000 description 1
- 238000002864 sequence alignment Methods 0.000 description 1
- 238000012163 sequencing technique Methods 0.000 description 1
- 238000013458 shaking study Methods 0.000 description 1
- 206010040560 shock Diseases 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 229920002545 silicone oil Polymers 0.000 description 1
- 238000002741 site-directed mutagenesis Methods 0.000 description 1
- 235000020183 skimmed milk Nutrition 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 235000011083 sodium citrates Nutrition 0.000 description 1
- RYYKJJJTJZKILX-UHFFFAOYSA-M sodium octadecanoate Chemical compound [Na+].CCCCCCCCCCCCCCCCCC([O-])=O RYYKJJJTJZKILX-UHFFFAOYSA-M 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- GNBVPFITFYNRCN-UHFFFAOYSA-M sodium thioglycolate Chemical compound [Na+].[O-]C(=O)CS GNBVPFITFYNRCN-UHFFFAOYSA-M 0.000 description 1
- 229940046307 sodium thioglycolate Drugs 0.000 description 1
- 235000019832 sodium triphosphate Nutrition 0.000 description 1
- 235000019337 sorbitan trioleate Nutrition 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 230000009870 specific binding Effects 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 239000001384 succinic acid Substances 0.000 description 1
- 108010012704 sulfated glycoprotein p50 Proteins 0.000 description 1
- DHCDFWKWKRSZHF-UHFFFAOYSA-N sulfurothioic S-acid Chemical compound OS(O)(=O)=S DHCDFWKWKRSZHF-UHFFFAOYSA-N 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 229960000814 tetanus toxoid Drugs 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 229920002725 thermoplastic elastomer Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 150000003573 thiols Chemical group 0.000 description 1
- 229940048910 thiosulfate Drugs 0.000 description 1
- 235000008521 threonine Nutrition 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 230000037317 transdermal delivery Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000009261 transgenic effect Effects 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- UNXRWKVEANCORM-UHFFFAOYSA-I triphosphate(5-) Chemical compound [O-]P([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O UNXRWKVEANCORM-UHFFFAOYSA-I 0.000 description 1
- 125000000430 tryptophan group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C2=C([H])C([H])=C([H])C([H])=C12 0.000 description 1
- 229960001005 tuberculin Drugs 0.000 description 1
- 102000003390 tumor necrosis factor Human genes 0.000 description 1
- 102000003298 tumor necrosis factor receptor Human genes 0.000 description 1
- 201000008297 typhoid fever Diseases 0.000 description 1
- 238000000108 ultra-filtration Methods 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- 229940125575 vaccine candidate Drugs 0.000 description 1
- 229940124856 vaccine component Drugs 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 230000006444 vascular growth Effects 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 238000002424 x-ray crystallography Methods 0.000 description 1
- 229920001221 xylan Polymers 0.000 description 1
- 150000004823 xylans Chemical class 0.000 description 1
- 239000012138 yeast extract Substances 0.000 description 1
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/02—Bacterial antigens
- A61K39/085—Staphylococcus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/60—Medicinal preparations containing antigens or antibodies characteristics by the carrier linked to the antigen
- A61K2039/6031—Proteins
- A61K2039/6037—Bacterial toxins, e.g. diphteria toxoid [DT], tetanus toxoid [TT]
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Immunology (AREA)
- Epidemiology (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- General Chemical & Material Sciences (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Medicinal Preparation (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Gastroenterology & Hepatology (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201061426476P | 2010-12-22 | 2010-12-22 | |
| US61/426,476 | 2010-12-22 | ||
| PCT/IB2011/055883 WO2012085872A2 (en) | 2010-12-22 | 2011-12-21 | Stable immunogenic compositions of staphylococcus aureus antigens |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2013128277A RU2013128277A (ru) | 2015-01-27 |
| RU2570730C2 true RU2570730C2 (ru) | 2015-12-10 |
Family
ID=45444677
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2013128277/15A RU2570730C2 (ru) | 2010-12-22 | 2011-12-21 | Стабильные иммуногенные композиции антигенов staphylococcus aureus |
Country Status (18)
| Country | Link |
|---|---|
| US (1) | US20130259896A1 (cg-RX-API-DMAC7.html) |
| EP (2) | EP2654784B1 (cg-RX-API-DMAC7.html) |
| JP (1) | JP6097478B2 (cg-RX-API-DMAC7.html) |
| KR (2) | KR101808398B1 (cg-RX-API-DMAC7.html) |
| CN (1) | CN103826656B (cg-RX-API-DMAC7.html) |
| AU (1) | AU2011346535B2 (cg-RX-API-DMAC7.html) |
| BR (1) | BR112013016153A2 (cg-RX-API-DMAC7.html) |
| CA (1) | CA2819120C (cg-RX-API-DMAC7.html) |
| DK (2) | DK3150222T3 (cg-RX-API-DMAC7.html) |
| ES (2) | ES2769425T3 (cg-RX-API-DMAC7.html) |
| HU (2) | HUE033072T2 (cg-RX-API-DMAC7.html) |
| IL (1) | IL226793B (cg-RX-API-DMAC7.html) |
| MX (1) | MX350170B (cg-RX-API-DMAC7.html) |
| PL (2) | PL2654784T3 (cg-RX-API-DMAC7.html) |
| PT (2) | PT3150222T (cg-RX-API-DMAC7.html) |
| RU (1) | RU2570730C2 (cg-RX-API-DMAC7.html) |
| SI (2) | SI2654784T1 (cg-RX-API-DMAC7.html) |
| WO (1) | WO2012085872A2 (cg-RX-API-DMAC7.html) |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2893938B1 (en) | 2004-09-22 | 2019-03-06 | GlaxoSmithKline Biologicals SA | Immunogenic composition for use in vaccination against Staphylococcei |
| PE20110023A1 (es) | 2009-06-22 | 2011-01-31 | Wyeth Llc | Composiciones inmunogenicas de antigenos de staphylococcus aureus |
| EP3461496B1 (en) | 2009-06-22 | 2023-08-23 | Wyeth LLC | Compositions and methods for preparing staphylococcus aureus serotype 5 and 8 capsular polysaccharide conjugate immunogenic compositions |
| MX2012013664A (es) * | 2010-06-04 | 2013-01-24 | Wyeth Llc | Formulaciones de vacuna. |
| AU2012266880A1 (en) * | 2011-03-16 | 2013-10-03 | Regents Of The University Of Minnesota | Compositions and methods for inducing immune responses against bacteria in the genus staphylococcus |
| GB201310008D0 (en) * | 2013-06-05 | 2013-07-17 | Glaxosmithkline Biolog Sa | Immunogenic composition for use in therapy |
| JP2016540764A (ja) * | 2013-12-04 | 2016-12-28 | グリコヴァキシン アーゲー | 大腸菌で合成された糖タンパク質ワクチンによる黄色ブドウ球菌感染の予防 |
| IL310015B1 (en) * | 2013-12-31 | 2025-10-01 | Access To Advanced Health Inst | Single vial formulation |
| US20170100476A1 (en) * | 2014-04-10 | 2017-04-13 | Asit Biotech S.A. | Hsp-free allergen preparation |
| KR20200040812A (ko) * | 2017-08-16 | 2020-04-20 | 머크 샤프 앤드 돔 코포레이션 | 폐렴구균 접합체 백신 제제 |
| GB201802339D0 (en) | 2018-02-13 | 2018-03-28 | Glaxosmithkline Biologicals Sa | Immunogenic composition |
| KR102649069B1 (ko) | 2018-05-31 | 2024-03-19 | 주식회사 엑소코바이오 | 줄기세포 유래의 엑소좀을 유효성분으로 포함하는 안면 홍조 개선용 조성물 |
| CN112261871B (zh) | 2018-07-28 | 2022-05-17 | 韩国外泌体生技有限公司 | 用于使外排体冻干的方法 |
| KR102163806B1 (ko) | 2018-07-30 | 2020-10-07 | 주식회사 엑소코바이오 | 줄기세포 유래의 엑소좀을 유효성분으로 포함하는 피지분비 감소용 조성물 |
| US20230151102A1 (en) * | 2020-01-13 | 2023-05-18 | Aptevo Research And Development Llc | Methods and compositions for preventing adsorption of therapeutic proteins to drug delivery system components |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006032472A2 (en) * | 2004-09-22 | 2006-03-30 | Glaxosmithkline Biologicals S.A. | Immunogenic composition for use in vaccination against staphylococcei |
| WO2007113222A2 (en) * | 2006-03-30 | 2007-10-11 | Glaxosmithkline Biologicals S.A. | Immunogenic composition |
| WO2007113223A2 (en) * | 2006-03-30 | 2007-10-11 | Glaxosmithkline Biologicals S.A. | Immunogenic composition |
| WO2008057550A2 (en) * | 2006-11-07 | 2008-05-15 | Sanofi Pasteur Biologics Co. | Stabilization of vaccines by lyophilization |
Family Cites Families (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4902506A (en) | 1983-07-05 | 1990-02-20 | The University Of Rochester | Immunogenic conjugates |
| US4673574A (en) | 1981-08-31 | 1987-06-16 | Anderson Porter W | Immunogenic conjugates |
| EP0138854B1 (en) | 1983-03-08 | 1992-11-04 | Chiron Mimotopes Pty. Ltd. | Antigenically active amino acid sequences |
| SE8405493D0 (sv) | 1984-11-01 | 1984-11-01 | Bror Morein | Immunogent komplex samt sett for framstellning derav och anvendning derav som immunstimulerande medel |
| US5078996A (en) | 1985-08-16 | 1992-01-07 | Immunex Corporation | Activation of macrophage tumoricidal activity by granulocyte-macrophage colony stimulating factor |
| US5057540A (en) | 1987-05-29 | 1991-10-15 | Cambridge Biotech Corporation | Saponin adjuvant |
| US4912094B1 (en) | 1988-06-29 | 1994-02-15 | Ribi Immunochem Research Inc. | Modified lipopolysaccharides and process of preparation |
| HU212924B (en) | 1989-05-25 | 1996-12-30 | Chiron Corp | Adjuvant formulation comprising a submicron oil droplet emulsion |
| IT1253009B (it) | 1991-12-31 | 1995-07-10 | Sclavo Ricerca S R L | Mutanti immunogenici detossificati della tossina colerica e della tossina lt, loro preparazione ed uso per la preparazione di vaccini |
| US5571515A (en) | 1994-04-18 | 1996-11-05 | The Wistar Institute Of Anatomy & Biology | Compositions and methods for use of IL-12 as an adjuvant |
| US6207646B1 (en) | 1994-07-15 | 2001-03-27 | University Of Iowa Research Foundation | Immunostimulatory nucleic acid molecules |
| US6008341A (en) | 1994-08-22 | 1999-12-28 | The Provost, Fellows And Scholars Of The College Of The Holy And Undivided Trinity Of Queen Elizabeth Near Dublin | S. aureus fibrinogen binding protein gene |
| BE1008978A5 (fr) | 1994-12-27 | 1996-10-01 | Solvay | Adjuvants pour vaccins. |
| WO1997014800A1 (en) | 1995-10-16 | 1997-04-24 | Smithkline Beecham Plc | Novel saliva binding protein |
| GB9622159D0 (en) | 1996-10-24 | 1996-12-18 | Solvay Sociutu Anonyme | Polyanionic polymers as adjuvants for mucosal immunization |
| GB9622660D0 (en) | 1996-10-31 | 1997-01-08 | Biocine Spa | Immunogenic detoxified mutant toxin |
| US6113918A (en) | 1997-05-08 | 2000-09-05 | Ribi Immunochem Research, Inc. | Aminoalkyl glucosamine phosphate compounds and their use as adjuvants and immunoeffectors |
| AU1537699A (en) | 1997-11-26 | 1999-06-15 | Bioresearch Ireland a division of Eolas - The Irish Science and Technology Agency | Extracellular matrix-binding proteins from (staphylococcus aureus) |
| US6680195B1 (en) | 1997-11-26 | 2004-01-20 | Inhibitex, Inc. | Extracellular matrix-binding proteins from staphylococcus aureus |
| MXPA01002119A (es) * | 1998-08-31 | 2003-03-27 | Inhibitex Inc | Vacunas multicomponentes. |
| US6936258B1 (en) | 1999-03-19 | 2005-08-30 | Nabi Biopharmaceuticals | Staphylococcus antigen and vaccine |
| US7115730B1 (en) | 1999-04-27 | 2006-10-03 | Chiron Srl | Immunogenic detoxified mutant E. coli LT-A-toxin |
| US7384640B1 (en) | 1999-09-30 | 2008-06-10 | Wyeth Holdings Corporation | Mutant cholera holotoxin as an adjuvant |
| BRPI0111639B8 (pt) | 2000-06-08 | 2021-05-25 | Intercell Ag | uso de uma molécula de ácido oligodeoxinucléico imunoestimulatório e composição farmacêutica. |
| AT410635B (de) | 2000-10-18 | 2003-06-25 | Cistem Biotechnologies Gmbh | Vakzin-zusammensetzung |
| JP2004535187A (ja) | 2001-06-07 | 2004-11-25 | ワイス・ホールデイングス・コーポレーシヨン | アジュバントとしてのコレラホロトキシンの突然変異形 |
| WO2002098369A2 (en) | 2001-06-07 | 2002-12-12 | Wyeth Holdings Corporation | Mutant forms of cholera holotoxin as an adjuvant |
| EP2339344A1 (en) | 2001-06-15 | 2011-06-29 | Inhibitex, Inc. | Cross-reactive monoclonal and polyclonal antibodies which recognize surface proteins from coagulase-negative staphylococci and staphylococcus aureus |
| GB0118249D0 (en) * | 2001-07-26 | 2001-09-19 | Chiron Spa | Histidine vaccines |
| NZ561879A (en) | 2003-03-07 | 2009-05-31 | Wyeth Corp | Polysaccharide - staphylococcal surface adhesin carrier protein conjugates for immunization against nosocomial infections |
| US20060251675A1 (en) | 2003-03-17 | 2006-11-09 | Michael Hagen | Mutant cholera holotoxin as an adjuvant and an antigen carrier protein |
| GB0323103D0 (en) | 2003-10-02 | 2003-11-05 | Chiron Srl | De-acetylated saccharides |
| GB0606416D0 (en) * | 2006-03-30 | 2006-05-10 | Glaxosmithkline Biolog Sa | Immunogenic composition |
| WO2007127668A2 (en) | 2006-04-26 | 2007-11-08 | Wyeth | Novel processes for coating container means which inhibit precipitation of polysaccharide-protein conjugate formulations |
| ES2615390T3 (es) | 2008-03-05 | 2017-06-06 | Sanofi Pasteur | Proceso para estabilizar una composición de vacuna que contiene adyuvante |
| PE20110023A1 (es) * | 2009-06-22 | 2011-01-31 | Wyeth Llc | Composiciones inmunogenicas de antigenos de staphylococcus aureus |
-
2011
- 2011-12-21 WO PCT/IB2011/055883 patent/WO2012085872A2/en not_active Ceased
- 2011-12-21 HU HUE11804822A patent/HUE033072T2/hu unknown
- 2011-12-21 US US13/993,887 patent/US20130259896A1/en not_active Abandoned
- 2011-12-21 DK DK16193095.3T patent/DK3150222T3/da active
- 2011-12-21 PL PL11804822T patent/PL2654784T3/pl unknown
- 2011-12-21 SI SI201131064A patent/SI2654784T1/sl unknown
- 2011-12-21 KR KR1020157031951A patent/KR101808398B1/ko not_active Expired - Fee Related
- 2011-12-21 EP EP11804822.2A patent/EP2654784B1/en active Active
- 2011-12-21 PT PT161930953T patent/PT3150222T/pt unknown
- 2011-12-21 EP EP16193095.3A patent/EP3150222B1/en active Active
- 2011-12-21 PL PL16193095T patent/PL3150222T3/pl unknown
- 2011-12-21 AU AU2011346535A patent/AU2011346535B2/en not_active Ceased
- 2011-12-21 MX MX2013006960A patent/MX350170B/es active IP Right Grant
- 2011-12-21 PT PT118048222T patent/PT2654784T/pt unknown
- 2011-12-21 CA CA2819120A patent/CA2819120C/en active Active
- 2011-12-21 SI SI201131825T patent/SI3150222T1/sl unknown
- 2011-12-21 DK DK11804822.2T patent/DK2654784T3/en active
- 2011-12-21 BR BR112013016153A patent/BR112013016153A2/pt not_active Application Discontinuation
- 2011-12-21 ES ES16193095T patent/ES2769425T3/es active Active
- 2011-12-21 ES ES11804822.2T patent/ES2614815T3/es active Active
- 2011-12-21 HU HUE16193095A patent/HUE047263T2/hu unknown
- 2011-12-21 RU RU2013128277/15A patent/RU2570730C2/ru active IP Right Revival
- 2011-12-21 CN CN201180068105.5A patent/CN103826656B/zh not_active Expired - Fee Related
- 2011-12-21 KR KR1020137018851A patent/KR20130114210A/ko not_active Ceased
- 2011-12-22 JP JP2011280781A patent/JP6097478B2/ja not_active Expired - Fee Related
-
2013
- 2013-06-06 IL IL226793A patent/IL226793B/en active IP Right Grant
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006032472A2 (en) * | 2004-09-22 | 2006-03-30 | Glaxosmithkline Biologicals S.A. | Immunogenic composition for use in vaccination against staphylococcei |
| WO2007113222A2 (en) * | 2006-03-30 | 2007-10-11 | Glaxosmithkline Biologicals S.A. | Immunogenic composition |
| WO2007113223A2 (en) * | 2006-03-30 | 2007-10-11 | Glaxosmithkline Biologicals S.A. | Immunogenic composition |
| WO2008057550A2 (en) * | 2006-11-07 | 2008-05-15 | Sanofi Pasteur Biologics Co. | Stabilization of vaccines by lyophilization |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2570730C2 (ru) | Стабильные иммуногенные композиции антигенов staphylococcus aureus | |
| EP2445522B1 (en) | Immunogenic compositions of staphylococcus aureus antigens | |
| TWI505834B (zh) | 組合物及製備金黃色葡萄球菌血清型5及8莢膜多醣結合物之免疫原性組合物之方法 | |
| IL305459A (en) | Immunogenic polysaccharide protein conjugated comprising a polysaccharide derived from b streptococcus gbs | |
| HK1197370B (en) | Stable immunogenic compositions of staphylococcus aureus antigens | |
| HK1171653A (en) | Immunogenic compositions of staphylococcus aureus antigens |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| HE4A | Change of address of a patent owner | ||
| MM4A | The patent is invalid due to non-payment of fees |
Effective date: 20161222 |
|
| NF4A | Reinstatement of patent |
Effective date: 20171121 |
|
| RH4A | Copy of patent granted that was duplicated for the russian federation |
Effective date: 20171201 |
|
| PD4A | Correction of name of patent owner |