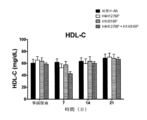
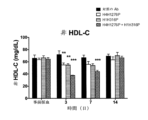
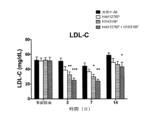
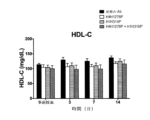
JP7541810B2 - Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 - Google Patents
Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 Download PDFInfo
- Publication number
- JP7541810B2 JP7541810B2 JP2018545828A JP2018545828A JP7541810B2 JP 7541810 B2 JP7541810 B2 JP 7541810B2 JP 2018545828 A JP2018545828 A JP 2018545828A JP 2018545828 A JP2018545828 A JP 2018545828A JP 7541810 B2 JP7541810 B2 JP 7541810B2
- Authority
- JP
- Japan
- Prior art keywords
- antibody
- angptl3
- patient
- pcsk9
- antigen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/22—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against growth factors ; against growth regulators
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/40—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
- A61K2039/507—Comprising a combination of two or more separate antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/545—Medicinal preparations containing antigens or antibodies characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Immunology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dermatology (AREA)
- Epidemiology (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Diabetes (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2022040874A JP2022078306A (ja) | 2016-03-03 | 2022-03-16 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
| JP2024025078A JP2024059787A (ja) | 2016-03-03 | 2024-02-22 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201662302907P | 2016-03-03 | 2016-03-03 | |
| US62/302,907 | 2016-03-03 | ||
| PCT/US2017/020221 WO2017151783A1 (en) | 2016-03-03 | 2017-03-01 | Methods for treating patients with hyperlipidemia by administering a pcsk9 inhibitor in combination with an angptl3 inhibitor |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022040874A Division JP2022078306A (ja) | 2016-03-03 | 2022-03-16 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2019512472A JP2019512472A (ja) | 2019-05-16 |
| JP2019512472A5 JP2019512472A5 (OSRAM) | 2020-03-05 |
| JP7541810B2 true JP7541810B2 (ja) | 2024-08-29 |
Family
ID=58361088
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018545828A Active JP7541810B2 (ja) | 2016-03-03 | 2017-03-01 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
| JP2022040874A Pending JP2022078306A (ja) | 2016-03-03 | 2022-03-16 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
| JP2024025078A Pending JP2024059787A (ja) | 2016-03-03 | 2024-02-22 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022040874A Pending JP2022078306A (ja) | 2016-03-03 | 2022-03-16 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
| JP2024025078A Pending JP2024059787A (ja) | 2016-03-03 | 2024-02-22 | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 |
Country Status (13)
| Country | Link |
|---|---|
| US (2) | US20170253666A1 (OSRAM) |
| EP (1) | EP3423157A1 (OSRAM) |
| JP (3) | JP7541810B2 (OSRAM) |
| KR (1) | KR102456731B1 (OSRAM) |
| CN (1) | CN109069868B (OSRAM) |
| AU (1) | AU2017227713B2 (OSRAM) |
| CA (1) | CA3016764A1 (OSRAM) |
| EA (1) | EA201891979A1 (OSRAM) |
| IL (1) | IL261420A (OSRAM) |
| MA (1) | MA43734A (OSRAM) |
| MX (1) | MX2018010401A (OSRAM) |
| WO (1) | WO2017151783A1 (OSRAM) |
| ZA (1) | ZA201805403B (OSRAM) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AR087329A1 (es) | 2011-06-17 | 2014-03-19 | Regeneron Pharma | Anticuerpos humanos contra proteina 3 de tipo angiopoietina humana |
| CA3016764A1 (en) | 2016-03-03 | 2017-09-08 | Regeneron Pharmaceuticals, Inc. | Methods for treating patients with hyperlipidemia by administering a pcsk9 inhibitor in combination with an angptl3 inhibitor |
| MX2018012741A (es) * | 2016-04-28 | 2019-05-16 | Regeneron Pharma | Metodos para el tratamiento de pacientes con hipercolesterolemia familiar. |
| CN110464842B (zh) * | 2018-05-11 | 2022-10-14 | 信达生物制药(苏州)有限公司 | 包含抗pcsk9抗体的制剂及其用途 |
| WO2021001804A1 (en) * | 2019-07-04 | 2021-01-07 | Cadila Healthcare Limited | Angptl3 based vaccine for the treatment of liver disease |
| AU2021320417A1 (en) * | 2020-08-07 | 2023-03-30 | Regeneron Pharmaceuticals, Inc. | Methods for treating refractory hypercholesterolemia involving an ANGPTL3 inhibitor |
| WO2022187353A1 (en) * | 2021-03-05 | 2022-09-09 | Anji Pharmaceuticals Inc. | Methods and compositions for treating sepsis |
| CN117535353A (zh) * | 2023-10-11 | 2024-02-09 | 首都医科大学附属北京潞河医院 | 一种糖尿病视网膜病变模型的建立方法及应用 |
| WO2025185644A1 (zh) * | 2024-03-05 | 2025-09-12 | 江苏恒瑞医药股份有限公司 | 抗angptl3抗体治疗高血脂症的用途及方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012511913A (ja) | 2008-12-15 | 2012-05-31 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | Pcsk9に対する高親和性ヒト抗体 |
| JP2014523739A (ja) | 2011-06-17 | 2014-09-18 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | 抗angptl3抗体及びその使用 |
| WO2016011256A1 (en) | 2014-07-16 | 2016-01-21 | Sanofi Biotechnology | METHODS FOR TREATING PATIENTS WITH HETEROZYGOUS FAMILIAL HYPERCHOLESTEROLEMIA (heFH) |
Family Cites Families (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0747045B2 (ja) | 1986-10-15 | 1995-05-24 | 株式会社大協精工 | 積層した注射器用滑栓 |
| JP3100727B2 (ja) | 1992-01-23 | 2000-10-23 | 株式会社大協精工 | 変性ポリシロキサン組成物及び該組成物を被覆した衛生ゴム製品 |
| JP3172057B2 (ja) | 1995-04-05 | 2001-06-04 | 株式会社大協精工 | ラミネートゴム栓 |
| US20020035058A1 (en) | 1996-05-15 | 2002-03-21 | The University Of Sheffield | Isopentenyl pyrophosphate isomerase (IPI) and/or prenyl transferase inhibitors |
| US6030831A (en) | 1997-09-19 | 2000-02-29 | Genetech, Inc. | Tie ligand homologues |
| US6172071B1 (en) | 1998-07-30 | 2001-01-09 | Hughes Institute | Lipid-lowering quinazoline derivative |
| JP3512349B2 (ja) | 1999-01-29 | 2004-03-29 | 株式会社大協精工 | 柱状ゴム要素の成形型 |
| EP1151092B1 (en) | 1999-02-12 | 2008-12-31 | President And Fellows Of Harvard College | Inhibiting formation of atherosclerotic lesions with afabp antisens nucleic acids |
| US6659982B2 (en) | 2000-05-08 | 2003-12-09 | Sterling Medivations, Inc. | Micro infusion drug delivery device |
| US6629949B1 (en) | 2000-05-08 | 2003-10-07 | Sterling Medivations, Inc. | Micro infusion drug delivery device |
| US6596541B2 (en) | 2000-10-31 | 2003-07-22 | Regeneron Pharmaceuticals, Inc. | Methods of modifying eukaryotic cells |
| JP2002209975A (ja) | 2001-01-19 | 2002-07-30 | Daikyo Seiko Ltd | 医薬バイアル用ラミネートゴム栓 |
| ATE357216T1 (de) | 2001-02-22 | 2007-04-15 | Jagotec Ag | Fibrat-statin kombinationen mit verminderten von der nahrungsaufnahme abhängigen auswirkungen |
| CA2464542C (en) | 2001-11-16 | 2015-01-20 | Genentech, Inc. | Composition comprising and method of using angiopoietin-like protein 3 angptl3 |
| EP2322200A3 (en) | 2002-10-29 | 2011-07-27 | Genentech, Inc. | Compositions and methods for the treatment of immune related diseases |
| JP2005080508A (ja) | 2003-09-04 | 2005-03-31 | Sankyo Co Ltd | 脂質代謝改善剤の試験方法 |
| US20110097330A1 (en) | 2005-03-11 | 2011-04-28 | Genentech, Inc. | Novel Gene Disruptions, Compostitions and Methods Relating Thereto |
| MX2009006082A (es) | 2006-12-08 | 2009-08-18 | Lexicon Pharmaceuticals Inc | Anticuerpos monoclonales contra la proteina 3 similar a angiopoyetina (angptl3). |
| AR070315A1 (es) | 2008-02-07 | 2010-03-31 | Merck & Co Inc | Anticuerpos 1b20 antagonistas de pcsk9 |
| US20130064834A1 (en) * | 2008-12-15 | 2013-03-14 | Regeneron Pharmaceuticals, Inc. | Methods for treating hypercholesterolemia using antibodies to pcsk9 |
| EP2216016A1 (en) | 2009-02-06 | 2010-08-11 | LEK Pharmaceuticals d.d. | Process for the preparation of a pharmaceutical composition comprising ezetimibe |
| CN102292088B (zh) | 2009-02-23 | 2014-01-29 | 奈米瑞斯公司 | 普利醇纳米粒子 |
| CN101852805B (zh) | 2009-03-31 | 2015-04-01 | 浙江大学 | Angptl3作为卵巢癌的诊断标记物的用途 |
| KR20180000337A (ko) | 2009-07-14 | 2018-01-02 | 노파르티스 아게 | 중간엽 줄기 세포 분화 |
| CN111700901A (zh) | 2010-01-08 | 2020-09-25 | Ionis制药公司 | 血管生成素样3表达的调节 |
| WO2012168491A1 (en) | 2011-06-10 | 2012-12-13 | Novartis Ag | Pharmaceutical formulations of pcsk9 antagonists |
| JP6110372B2 (ja) | 2011-06-21 | 2017-04-05 | アルナイラム ファーマシューティカルズ, インコーポレイテッドAlnylam Pharmaceuticals, Inc. | アンジオポエチン様3(ANGPTL3)iRNA組成物及びその使用方法 |
| EP2735315B1 (en) | 2011-07-19 | 2019-10-02 | Chugai Seiyaku Kabushiki Kaisha | Stable protein-containing preparation containing argininamide or valinamide |
| AR087305A1 (es) | 2011-07-28 | 2014-03-12 | Regeneron Pharma | Formulaciones estabilizadas que contienen anticuerpos anti-pcsk9, metodo de preparacion y kit |
| EA039663B1 (ru) | 2012-05-03 | 2022-02-24 | Амген Инк. | Применение антитела против pcsk9 для снижения сывороточного холестерина лпнп и лечения связанных с холестерином расстройств |
| KR20150070384A (ko) | 2012-10-25 | 2015-06-24 | 메디뮨 엘엘씨 | 안정한 저점도 항체 제제 |
| JP6570511B2 (ja) | 2013-03-14 | 2019-09-04 | ユーエイビー リサーチ ファンデーション | アポリポタンパク質模倣体及びその使用 |
| US10111953B2 (en) * | 2013-05-30 | 2018-10-30 | Regeneron Pharmaceuticals, Inc. | Methods for reducing remnant cholesterol and other lipoprotein fractions by administering an inhibitor of proprotein convertase subtilisin kexin-9 (PCSK9) |
| CN105705521A (zh) | 2013-06-07 | 2016-06-22 | 再生元制药公司 | 通过施用pcsk9抑制剂抑制动脉粥样硬化的方法 |
| WO2015077154A1 (en) | 2013-11-20 | 2015-05-28 | Cymabay Therapeutics, Inc. | Treatment of homozygous familial hypercholesterolemia |
| KR102298476B1 (ko) | 2013-12-24 | 2021-09-03 | 아이오니스 파마수티컬즈, 인코포레이티드 | 안지오포이에틴-유사 3 발현의 조절 |
| EP3137605B1 (en) | 2014-05-01 | 2020-10-28 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating angiopoietin-like 3 expression |
| KR20230007538A (ko) | 2014-07-16 | 2023-01-12 | 사노피 바이오테크놀로지 | 고콜레스테롤혈증이 있는 심혈관 위험이 높은 환자를 치료하는 방법 |
| EP3201225B1 (en) | 2014-10-03 | 2020-08-19 | NGM Biopharmaceuticals, Inc. | Angptl8 polypeptide and its use for treatment of conditions associated with elevated triglycerides |
| TW201713690A (zh) | 2015-08-07 | 2017-04-16 | 再生元醫藥公司 | 抗angptl8抗體及其用途 |
| WO2017142832A1 (en) | 2016-02-17 | 2017-08-24 | Regeneron Pharmaceuticals, Inc. | Methods for treating or preventing atherosclerosis by administering an inhibitor of angptl3 |
| CA3016764A1 (en) | 2016-03-03 | 2017-09-08 | Regeneron Pharmaceuticals, Inc. | Methods for treating patients with hyperlipidemia by administering a pcsk9 inhibitor in combination with an angptl3 inhibitor |
| MX386832B (es) | 2016-04-08 | 2025-03-19 | Regeneron Pharma | Inhibidor angptl8 e inhibidor angptl3 para usarse en el tratamiento de hiperlipidemia. |
| MX2018012741A (es) | 2016-04-28 | 2019-05-16 | Regeneron Pharma | Metodos para el tratamiento de pacientes con hipercolesterolemia familiar. |
| US11603407B2 (en) | 2017-04-06 | 2023-03-14 | Regeneron Pharmaceuticals, Inc. | Stable antibody formulation |
| US20200369760A1 (en) | 2019-05-24 | 2020-11-26 | Regeneron Pharmaceuticals, Inc. | Stabilized formulations containing anti-angptl3 antibodies |
| AU2021320417A1 (en) | 2020-08-07 | 2023-03-30 | Regeneron Pharmaceuticals, Inc. | Methods for treating refractory hypercholesterolemia involving an ANGPTL3 inhibitor |
-
2017
- 2017-03-01 CA CA3016764A patent/CA3016764A1/en active Pending
- 2017-03-01 AU AU2017227713A patent/AU2017227713B2/en active Active
- 2017-03-01 MX MX2018010401A patent/MX2018010401A/es unknown
- 2017-03-01 EA EA201891979A patent/EA201891979A1/ru unknown
- 2017-03-01 EP EP17712283.5A patent/EP3423157A1/en active Pending
- 2017-03-01 US US15/446,720 patent/US20170253666A1/en not_active Abandoned
- 2017-03-01 MA MA043734A patent/MA43734A/fr unknown
- 2017-03-01 CN CN201780014857.0A patent/CN109069868B/zh active Active
- 2017-03-01 WO PCT/US2017/020221 patent/WO2017151783A1/en not_active Ceased
- 2017-03-01 JP JP2018545828A patent/JP7541810B2/ja active Active
- 2017-03-01 KR KR1020187028068A patent/KR102456731B1/ko active Active
-
2018
- 2018-08-14 ZA ZA2018/05403A patent/ZA201805403B/en unknown
- 2018-08-28 IL IL261420A patent/IL261420A/en unknown
-
2020
- 2020-03-03 US US16/808,058 patent/US12281173B2/en active Active
-
2022
- 2022-03-16 JP JP2022040874A patent/JP2022078306A/ja active Pending
-
2024
- 2024-02-22 JP JP2024025078A patent/JP2024059787A/ja active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012511913A (ja) | 2008-12-15 | 2012-05-31 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | Pcsk9に対する高親和性ヒト抗体 |
| JP2014523739A (ja) | 2011-06-17 | 2014-09-18 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | 抗angptl3抗体及びその使用 |
| WO2016011256A1 (en) | 2014-07-16 | 2016-01-21 | Sanofi Biotechnology | METHODS FOR TREATING PATIENTS WITH HETEROZYGOUS FAMILIAL HYPERCHOLESTEROLEMIA (heFH) |
Non-Patent Citations (1)
| Title |
|---|
| Hypercholesterolemia,2015年,Chapter 10,Resistance of Statin Therapy, and Methods for its Influence,pp. 185-202,http://dx.doi.org/10.5772/60004 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2017151783A1 (en) | 2017-09-08 |
| JP2022078306A (ja) | 2022-05-24 |
| MA43734A (fr) | 2018-11-28 |
| CN109069868A (zh) | 2018-12-21 |
| CN109069868B (zh) | 2023-06-06 |
| EP3423157A1 (en) | 2019-01-09 |
| CA3016764A1 (en) | 2017-09-08 |
| JP2019512472A (ja) | 2019-05-16 |
| JP2024059787A (ja) | 2024-05-01 |
| KR102456731B1 (ko) | 2022-10-24 |
| IL261420A (en) | 2018-10-31 |
| AU2017227713B2 (en) | 2022-12-22 |
| KR20180118181A (ko) | 2018-10-30 |
| MX2018010401A (es) | 2019-03-06 |
| US12281173B2 (en) | 2025-04-22 |
| ZA201805403B (en) | 2019-08-28 |
| EA201891979A1 (ru) | 2019-01-31 |
| US20200199253A1 (en) | 2020-06-25 |
| AU2017227713A1 (en) | 2018-09-13 |
| US20170253666A1 (en) | 2017-09-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7404471B2 (ja) | 家族性高コレステロール血症を有する患者を処置するための方法 | |
| US20220127348A1 (en) | Methods for treating hyperlipidemia with an angptl8 inhibitor and an angptl3 inhibitor | |
| JP7541810B2 (ja) | Angptl3阻害剤と組み合わせてpcsk9阻害剤を投与することにより高脂血症を有する患者を処置するための方法 | |
| JP7648335B2 (ja) | Angptl3の阻害剤を投与することによるアテローム性動脈硬化症の処置又は予防のための方法 | |
| US20240197870A1 (en) | Methods for treating patients with refractory hypercholesterolemia | |
| EA043821B1 (ru) | Способы лечения пациентов с гиперлипидемией введением ингибитора pcsk9 в комбинации с ингибитором angptl3 | |
| EA042876B1 (ru) | Способы лечения пациентов с семейной гиперхолестеринемией | |
| HK40003056B (en) | Methods for treating patients with familial hypercholesterolemia | |
| HK40003056A (en) | Methods for treating patients with familial hypercholesterolemia | |
| EA040975B1 (ru) | Способы лечения или предотвращения атеросклероза путем введения ингибитора angptl3 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200123 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20200123 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210126 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20210426 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210720 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20211116 |
|
| C60 | Trial request (containing other claim documents, opposition documents) |
Free format text: JAPANESE INTERMEDIATE CODE: C60 Effective date: 20220316 |
|
| C116 | Written invitation by the chief administrative judge to file amendments |
Free format text: JAPANESE INTERMEDIATE CODE: C116 Effective date: 20220329 |
|
| C22 | Notice of designation (change) of administrative judge |
Free format text: JAPANESE INTERMEDIATE CODE: C22 Effective date: 20220329 |
|
| C22 | Notice of designation (change) of administrative judge |
Free format text: JAPANESE INTERMEDIATE CODE: C22 Effective date: 20220412 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20231228 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240131 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20240819 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7541810 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |