JP7282079B2 - センサが使用可能な陰圧創傷監視および療法装置のph感知 - Google Patents
センサが使用可能な陰圧創傷監視および療法装置のph感知 Download PDFInfo
- Publication number
- JP7282079B2 JP7282079B2 JP2020516581A JP2020516581A JP7282079B2 JP 7282079 B2 JP7282079 B2 JP 7282079B2 JP 2020516581 A JP2020516581 A JP 2020516581A JP 2020516581 A JP2020516581 A JP 2020516581A JP 7282079 B2 JP7282079 B2 JP 7282079B2
- Authority
- JP
- Japan
- Prior art keywords
- wound
- layer
- dressing
- sensor
- negative pressure
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000012544 monitoring process Methods 0.000 title claims description 37
- 238000002560 therapeutic procedure Methods 0.000 title description 30
- 239000000463 material Substances 0.000 claims description 187
- 239000006260 foam Substances 0.000 claims description 69
- 230000003287 optical effect Effects 0.000 claims description 64
- 210000000416 exudates and transudate Anatomy 0.000 claims description 60
- 239000012925 reference material Substances 0.000 claims description 49
- 239000000853 adhesive Substances 0.000 claims description 39
- 230000001070 adhesive effect Effects 0.000 claims description 38
- 230000008859 change Effects 0.000 claims description 30
- 239000004814 polyurethane Substances 0.000 claims description 18
- 229920002635 polyurethane Polymers 0.000 claims description 18
- 230000004044 response Effects 0.000 claims description 10
- 229920001477 hydrophilic polymer Polymers 0.000 claims description 6
- 206010052428 Wound Diseases 0.000 description 613
- 208000027418 Wounds and injury Diseases 0.000 description 613
- 239000010410 layer Substances 0.000 description 424
- 239000012530 fluid Substances 0.000 description 106
- 239000002250 absorbent Substances 0.000 description 96
- 230000002745 absorbent Effects 0.000 description 96
- 238000000034 method Methods 0.000 description 52
- 210000001519 tissue Anatomy 0.000 description 51
- 239000007788 liquid Substances 0.000 description 34
- 238000011282 treatment Methods 0.000 description 34
- 239000000835 fiber Substances 0.000 description 33
- 239000004744 fabric Substances 0.000 description 26
- 239000000499 gel Substances 0.000 description 23
- 238000009581 negative-pressure wound therapy Methods 0.000 description 21
- 239000010408 film Substances 0.000 description 19
- 229920000642 polymer Polymers 0.000 description 19
- 125000006850 spacer group Chemical group 0.000 description 18
- 210000000056 organ Anatomy 0.000 description 17
- 230000029663 wound healing Effects 0.000 description 15
- 230000035876 healing Effects 0.000 description 14
- 239000000203 mixture Chemical class 0.000 description 14
- -1 propylcellulose Polymers 0.000 description 14
- 230000005540 biological transmission Effects 0.000 description 13
- 229920002678 cellulose Polymers 0.000 description 13
- 239000001913 cellulose Substances 0.000 description 13
- 230000001684 chronic effect Effects 0.000 description 13
- 238000001139 pH measurement Methods 0.000 description 13
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 13
- 239000000975 dye Substances 0.000 description 12
- 229920000728 polyester Polymers 0.000 description 12
- 238000007789 sealing Methods 0.000 description 12
- 238000007906 compression Methods 0.000 description 11
- 230000006835 compression Effects 0.000 description 11
- 230000002209 hydrophobic effect Effects 0.000 description 11
- 238000005259 measurement Methods 0.000 description 11
- 208000004210 Pressure Ulcer Diseases 0.000 description 10
- 239000012790 adhesive layer Substances 0.000 description 10
- 239000012528 membrane Substances 0.000 description 10
- 239000000758 substrate Substances 0.000 description 10
- 230000008901 benefit Effects 0.000 description 9
- 230000006378 damage Effects 0.000 description 9
- 238000005516 engineering process Methods 0.000 description 9
- 239000000945 filler Substances 0.000 description 9
- 230000001965 increasing effect Effects 0.000 description 9
- 210000004379 membrane Anatomy 0.000 description 9
- 239000011148 porous material Substances 0.000 description 9
- 230000008569 process Effects 0.000 description 9
- 230000017531 blood circulation Effects 0.000 description 8
- 239000011248 coating agent Substances 0.000 description 8
- 238000000576 coating method Methods 0.000 description 8
- 239000007789 gas Substances 0.000 description 8
- 229920001296 polysiloxane Polymers 0.000 description 8
- 229920006264 polyurethane film Polymers 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 7
- 239000004820 Pressure-sensitive adhesive Substances 0.000 description 7
- 238000003491 array Methods 0.000 description 7
- 230000001580 bacterial effect Effects 0.000 description 7
- 238000005286 illumination Methods 0.000 description 7
- 208000015181 infectious disease Diseases 0.000 description 7
- 238000012545 processing Methods 0.000 description 7
- 238000001356 surgical procedure Methods 0.000 description 7
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 230000003187 abdominal effect Effects 0.000 description 6
- 230000002378 acidificating effect Effects 0.000 description 6
- 239000003570 air Substances 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 208000014674 injury Diseases 0.000 description 6
- 238000009434 installation Methods 0.000 description 6
- 238000012856 packing Methods 0.000 description 6
- 230000002829 reductive effect Effects 0.000 description 6
- 239000004698 Polyethylene Substances 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- 230000001154 acute effect Effects 0.000 description 5
- 150000008052 alkyl sulfonates Chemical class 0.000 description 5
- 230000004888 barrier function Effects 0.000 description 5
- 230000000903 blocking effect Effects 0.000 description 5
- 230000008878 coupling Effects 0.000 description 5
- 238000010168 coupling process Methods 0.000 description 5
- 238000005859 coupling reaction Methods 0.000 description 5
- 238000013480 data collection Methods 0.000 description 5
- 239000008367 deionised water Substances 0.000 description 5
- 229910021641 deionized water Inorganic materials 0.000 description 5
- 238000013461 design Methods 0.000 description 5
- 238000010586 diagram Methods 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 239000011159 matrix material Substances 0.000 description 5
- 229920000573 polyethylene Polymers 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 238000012546 transfer Methods 0.000 description 5
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 4
- 206010011985 Decubitus ulcer Diseases 0.000 description 4
- 239000003522 acrylic cement Substances 0.000 description 4
- 239000008280 blood Substances 0.000 description 4
- 210000004369 blood Anatomy 0.000 description 4
- 239000001768 carboxy methyl cellulose Substances 0.000 description 4
- 210000004027 cell Anatomy 0.000 description 4
- 238000004891 communication Methods 0.000 description 4
- 238000005538 encapsulation Methods 0.000 description 4
- 210000003414 extremity Anatomy 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 150000004676 glycans Chemical class 0.000 description 4
- 239000007943 implant Substances 0.000 description 4
- 230000035699 permeability Effects 0.000 description 4
- 229920001282 polysaccharide Polymers 0.000 description 4
- 239000005017 polysaccharide Substances 0.000 description 4
- 239000013464 silicone adhesive Substances 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 102000004127 Cytokines Human genes 0.000 description 3
- 108090000695 Cytokines Proteins 0.000 description 3
- 206010063560 Excessive granulation tissue Diseases 0.000 description 3
- 206010030113 Oedema Diseases 0.000 description 3
- 239000004952 Polyamide Substances 0.000 description 3
- 229920005830 Polyurethane Foam Polymers 0.000 description 3
- 208000025865 Ulcer Diseases 0.000 description 3
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 125000000217 alkyl group Chemical group 0.000 description 3
- 239000003242 anti bacterial agent Substances 0.000 description 3
- 229940088710 antibiotic agent Drugs 0.000 description 3
- 239000004599 antimicrobial Substances 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- 230000009286 beneficial effect Effects 0.000 description 3
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 3
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 3
- 239000002131 composite material Substances 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 230000002526 effect on cardiovascular system Effects 0.000 description 3
- 210000001126 granulation tissue Anatomy 0.000 description 3
- 230000003993 interaction Effects 0.000 description 3
- 230000000670 limiting effect Effects 0.000 description 3
- 150000002632 lipids Chemical class 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000001537 neural effect Effects 0.000 description 3
- 230000000399 orthopedic effect Effects 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 229920002647 polyamide Polymers 0.000 description 3
- 239000004810 polytetrafluoroethylene Substances 0.000 description 3
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 3
- 239000011496 polyurethane foam Substances 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 230000003595 spectral effect Effects 0.000 description 3
- 208000037816 tissue injury Diseases 0.000 description 3
- 230000008733 trauma Effects 0.000 description 3
- 231100000397 ulcer Toxicity 0.000 description 3
- UDOQBLACUKZOKY-UHFFFAOYSA-N 2-[[4-(2-hydroxyethylsulfonyl)phenyl]diazenyl]-4-methylphenol Chemical compound OCCS(=O)(=O)C1=CC=C(C=C1)N=NC1=C(C=CC(=C1)C)O UDOQBLACUKZOKY-UHFFFAOYSA-N 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 2
- 208000034656 Contusions Diseases 0.000 description 2
- 206010056340 Diabetic ulcer Diseases 0.000 description 2
- 206010014989 Epidermolysis bullosa Diseases 0.000 description 2
- 239000004593 Epoxy Substances 0.000 description 2
- 108091005804 Peptidases Proteins 0.000 description 2
- 239000004642 Polyimide Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- 239000004372 Polyvinyl alcohol Chemical class 0.000 description 2
- 239000004365 Protease Substances 0.000 description 2
- 229920000297 Rayon Polymers 0.000 description 2
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 208000002847 Surgical Wound Diseases 0.000 description 2
- 208000000558 Varicose Ulcer Diseases 0.000 description 2
- 210000001015 abdomen Anatomy 0.000 description 2
- 229920006243 acrylic copolymer Polymers 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 229940045714 alkyl sulfonate alkylating agent Drugs 0.000 description 2
- 125000005227 alkyl sulfonate group Chemical group 0.000 description 2
- 239000012080 ambient air Substances 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 210000000988 bone and bone Anatomy 0.000 description 2
- QDHFHIQKOVNCNC-UHFFFAOYSA-N butane-1-sulfonic acid Chemical group CCCCS(O)(=O)=O QDHFHIQKOVNCNC-UHFFFAOYSA-N 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000005253 cladding Methods 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- 238000007405 data analysis Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 230000023753 dehiscence Effects 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 239000002657 fibrous material Substances 0.000 description 2
- 239000000834 fixative Substances 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- 230000004054 inflammatory process Effects 0.000 description 2
- 238000003475 lamination Methods 0.000 description 2
- 238000002386 leaching Methods 0.000 description 2
- 230000031700 light absorption Effects 0.000 description 2
- 230000014759 maintenance of location Effects 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 210000003205 muscle Anatomy 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 238000010606 normalization Methods 0.000 description 2
- 238000006213 oxygenation reaction Methods 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 210000003200 peritoneal cavity Anatomy 0.000 description 2
- 229920001721 polyimide Polymers 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 229920002451 polyvinyl alcohol Chemical class 0.000 description 2
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 238000007639 printing Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- KCXFHTAICRTXLI-UHFFFAOYSA-N propane-1-sulfonic acid Chemical group CCCS(O)(=O)=O KCXFHTAICRTXLI-UHFFFAOYSA-N 0.000 description 2
- 238000002106 pulse oximetry Methods 0.000 description 2
- 238000011084 recovery Methods 0.000 description 2
- 239000011540 sensing material Substances 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- 230000000638 stimulation Effects 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 230000000699 topical effect Effects 0.000 description 2
- 230000000472 traumatic effect Effects 0.000 description 2
- 238000002604 ultrasonography Methods 0.000 description 2
- 238000011179 visual inspection Methods 0.000 description 2
- FKOZPUORKCHONH-UHFFFAOYSA-N 2-methylpropane-1-sulfonic acid Chemical compound CC(C)CS(O)(=O)=O FKOZPUORKCHONH-UHFFFAOYSA-N 0.000 description 1
- OURSFPZPOXNNKX-UHFFFAOYSA-N 3-sulfopropanoic acid Chemical compound OC(=O)CCS(O)(=O)=O OURSFPZPOXNNKX-UHFFFAOYSA-N 0.000 description 1
- XURABDHWIADCPO-UHFFFAOYSA-N 4-prop-2-enylhepta-1,6-diene Chemical compound C=CCC(CC=C)CC=C XURABDHWIADCPO-UHFFFAOYSA-N 0.000 description 1
- 206010058808 Abdominal compartment syndrome Diseases 0.000 description 1
- 208000019901 Anxiety disease Diseases 0.000 description 1
- 208000035143 Bacterial infection Diseases 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- 229920003043 Cellulose fiber Polymers 0.000 description 1
- 108010035532 Collagen Chemical class 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 229920001651 Cyanoacrylate Polymers 0.000 description 1
- 208000008960 Diabetic foot Diseases 0.000 description 1
- 229920006347 Elastollan Polymers 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- KIWBPDUYBMNFTB-UHFFFAOYSA-N Ethyl hydrogen sulfate Chemical compound CCOS(O)(=O)=O KIWBPDUYBMNFTB-UHFFFAOYSA-N 0.000 description 1
- 208000035874 Excoriation Diseases 0.000 description 1
- 208000003790 Foot Ulcer Diseases 0.000 description 1
- 108010010803 Gelatin Chemical class 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 102000001554 Hemoglobins Human genes 0.000 description 1
- 108010054147 Hemoglobins Proteins 0.000 description 1
- 239000004831 Hot glue Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 208000002623 Intra-Abdominal Hypertension Diseases 0.000 description 1
- 208000034693 Laceration Diseases 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 208000005230 Leg Ulcer Diseases 0.000 description 1
- 208000018501 Lymphatic disease Diseases 0.000 description 1
- MWCLLHOVUTZFKS-UHFFFAOYSA-N Methyl cyanoacrylate Chemical compound COC(=O)C(=C)C#N MWCLLHOVUTZFKS-UHFFFAOYSA-N 0.000 description 1
- 208000004221 Multiple Trauma Diseases 0.000 description 1
- 206010028885 Necrotising fasciitis Diseases 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 239000002033 PVDF binder Substances 0.000 description 1
- CYTYCFOTNPOANT-UHFFFAOYSA-N Perchloroethylene Chemical group ClC(Cl)=C(Cl)Cl CYTYCFOTNPOANT-UHFFFAOYSA-N 0.000 description 1
- 208000005764 Peripheral Arterial Disease Diseases 0.000 description 1
- 208000030831 Peripheral arterial occlusive disease Diseases 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- 238000012274 Preoperative evaluation Methods 0.000 description 1
- 229920001247 Reticulated foam Polymers 0.000 description 1
- 206010040047 Sepsis Diseases 0.000 description 1
- 206010040943 Skin Ulcer Diseases 0.000 description 1
- 206010048625 Skin maceration Diseases 0.000 description 1
- 206010044546 Traumatic ulcer Diseases 0.000 description 1
- 210000000683 abdominal cavity Anatomy 0.000 description 1
- 210000003815 abdominal wall Anatomy 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- 239000011358 absorbing material Substances 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- 230000001133 acceleration Effects 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 230000009692 acute damage Effects 0.000 description 1
- 210000000577 adipose tissue Anatomy 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 150000008051 alkyl sulfates Chemical group 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000036506 anxiety Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 208000022362 bacterial infectious disease Diseases 0.000 description 1
- 238000003287 bathing Methods 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- 239000001045 blue dye Substances 0.000 description 1
- 238000009529 body temperature measurement Methods 0.000 description 1
- 208000034526 bruise Diseases 0.000 description 1
- DQXBYHZEEUGOBF-UHFFFAOYSA-N but-3-enoic acid;ethene Chemical compound C=C.OC(=O)CC=C DQXBYHZEEUGOBF-UHFFFAOYSA-N 0.000 description 1
- BRXCDHOLJPJLLT-UHFFFAOYSA-N butane-2-sulfonic acid Chemical compound CCC(C)S(O)(=O)=O BRXCDHOLJPJLLT-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000003990 capacitor Substances 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 210000000748 cardiovascular system Anatomy 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 238000010909 chemical acidification Methods 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 230000009693 chronic damage Effects 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000012459 cleaning agent Substances 0.000 description 1
- 239000011247 coating layer Substances 0.000 description 1
- 229920001436 collagen Chemical class 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 230000009519 contusion Effects 0.000 description 1
- 239000013039 cover film Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000006837 decompression Effects 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000005108 dry cleaning Methods 0.000 description 1
- 230000005684 electric field Effects 0.000 description 1
- 238000002567 electromyography Methods 0.000 description 1
- 210000002889 endothelial cell Anatomy 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 239000005038 ethylene vinyl acetate Substances 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 229920005570 flexible polymer Polymers 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 229920002313 fluoropolymer Polymers 0.000 description 1
- 239000004811 fluoropolymer Substances 0.000 description 1
- 239000006261 foam material Substances 0.000 description 1
- 210000002683 foot Anatomy 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 239000008273 gelatin Chemical class 0.000 description 1
- 229920000159 gelatin Chemical class 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 238000005469 granulation Methods 0.000 description 1
- 230000003179 granulation Effects 0.000 description 1
- 230000037313 granulation tissue formation Effects 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 238000003306 harvesting Methods 0.000 description 1
- 229920001903 high density polyethylene Polymers 0.000 description 1
- 239000004700 high-density polyethylene Substances 0.000 description 1
- 239000012943 hotmelt Substances 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 239000000416 hydrocolloid Substances 0.000 description 1
- 229920001600 hydrophobic polymer Polymers 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 150000003949 imides Chemical class 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 230000002458 infectious effect Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 239000000976 ink Substances 0.000 description 1
- 239000012784 inorganic fiber Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 229920000592 inorganic polymer Polymers 0.000 description 1
- 238000005304 joining Methods 0.000 description 1
- 210000003127 knee Anatomy 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 238000000608 laser ablation Methods 0.000 description 1
- 210000002414 leg Anatomy 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 229910001416 lithium ion Inorganic materials 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 210000003141 lower extremity Anatomy 0.000 description 1
- 208000018555 lymphatic system disease Diseases 0.000 description 1
- 238000002803 maceration Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 239000012569 microbial contaminant Substances 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 230000004220 muscle function Effects 0.000 description 1
- 230000004092 musculoskeletal function Effects 0.000 description 1
- 229920005615 natural polymer Polymers 0.000 description 1
- 201000007970 necrotizing fasciitis Diseases 0.000 description 1
- 230000007383 nerve stimulation Effects 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 238000002640 oxygen therapy Methods 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 230000010412 perfusion Effects 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000002572 peristaltic effect Effects 0.000 description 1
- 206010034674 peritonitis Diseases 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 238000000554 physical therapy Methods 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 229920006112 polar polymer Polymers 0.000 description 1
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 1
- 229920002401 polyacrylamide Chemical class 0.000 description 1
- 229920000058 polyacrylate Chemical class 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 239000011112 polyethylene naphthalate Substances 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229920001289 polyvinyl ether Chemical class 0.000 description 1
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Chemical class 0.000 description 1
- 239000001267 polyvinylpyrrolidone Chemical class 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 230000030393 positive regulation of fibroblast proliferation Effects 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000000541 pulsatile effect Effects 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000001044 red dye Substances 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 230000002940 repellent Effects 0.000 description 1
- 239000005871 repellent Substances 0.000 description 1
- 230000037390 scarring Effects 0.000 description 1
- 230000009919 sequestration Effects 0.000 description 1
- 230000037067 skin hydration Effects 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000008259 solid foam Substances 0.000 description 1
- 238000007711 solidification Methods 0.000 description 1
- 230000008023 solidification Effects 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 238000011477 surgical intervention Methods 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 210000004243 sweat Anatomy 0.000 description 1
- 230000001360 synchronised effect Effects 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 229920005613 synthetic organic polymer Polymers 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229950011008 tetrachloroethylene Drugs 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 230000017423 tissue regeneration Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000005068 transpiration Effects 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 230000003966 vascular damage Effects 0.000 description 1
- 208000037997 venous disease Diseases 0.000 description 1
- 230000035899 viability Effects 0.000 description 1
- 210000001835 viscera Anatomy 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 238000004078 waterproofing Methods 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 238000003466 welding Methods 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 230000010388 wound contraction Effects 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/0059—Measuring for diagnostic purposes; Identification of persons using light, e.g. diagnosis by transillumination, diascopy, fluorescence
- A61B5/0075—Measuring for diagnostic purposes; Identification of persons using light, e.g. diagnosis by transillumination, diascopy, fluorescence by spectroscopy, i.e. measuring spectra, e.g. Raman spectroscopy, infrared absorption spectroscopy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/103—Detecting, measuring or recording devices for testing the shape, pattern, colour, size or movement of the body or parts thereof, for diagnostic purposes
- A61B5/1032—Determining colour for diagnostic purposes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/145—Measuring characteristics of blood in vivo, e.g. gas concentration, pH value; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid, cerebral tissue
- A61B5/14539—Measuring characteristics of blood in vivo, e.g. gas concentration, pH value; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid, cerebral tissue for measuring pH
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/145—Measuring characteristics of blood in vivo, e.g. gas concentration, pH value; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid, cerebral tissue
- A61B5/1455—Measuring characteristics of blood in vivo, e.g. gas concentration, pH value; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid, cerebral tissue using optical sensors, e.g. spectral photometrical oximeters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/44—Detecting, measuring or recording for evaluating the integumentary system, e.g. skin, hair or nails
- A61B5/441—Skin evaluation, e.g. for skin disorder diagnosis
- A61B5/445—Evaluating skin irritation or skin trauma, e.g. rash, eczema, wound, bed sore
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/68—Arrangements of detecting, measuring or recording means, e.g. sensors, in relation to patient
- A61B5/6801—Arrangements of detecting, measuring or recording means, e.g. sensors, in relation to patient specially adapted to be attached to or worn on the body surface
- A61B5/683—Means for maintaining contact with the body
- A61B5/6832—Means for maintaining contact with the body using adhesives
- A61B5/6833—Adhesive patches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/00051—Accessories for dressings
- A61F13/00059—Accessories for dressings provided with visual effects, e.g. printed or colored
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/00051—Accessories for dressings
- A61F13/00063—Accessories for dressings comprising medicaments or additives, e.g. odor control, PH control, debriding, antimicrobic
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/02—Adhesive plasters or dressings
- A61F13/0203—Adhesive plasters or dressings having a fluid handling member
-
- A61F13/05—
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/90—Negative pressure wound therapy devices, i.e. devices for applying suction to a wound to promote healing, e.g. including a vacuum dressing
- A61M1/91—Suction aspects of the dressing
- A61M1/912—Connectors between dressing and drainage tube
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/90—Negative pressure wound therapy devices, i.e. devices for applying suction to a wound to promote healing, e.g. including a vacuum dressing
- A61M1/91—Suction aspects of the dressing
- A61M1/912—Connectors between dressing and drainage tube
- A61M1/913—Connectors between dressing and drainage tube having a bridging element for transferring the reduced pressure from the connector to the dressing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B2562/00—Details of sensors; Constructional details of sensor housings or probes; Accessories for sensors
- A61B2562/06—Arrangements of multiple sensors of different types
- A61B2562/066—Arrangements of multiple sensors of different types in a matrix array
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B2562/00—Details of sensors; Constructional details of sensor housings or probes; Accessories for sensors
- A61B2562/16—Details of sensor housings or probes; Details of structural supports for sensors
- A61B2562/164—Details of sensor housings or probes; Details of structural supports for sensors the sensor is mounted in or on a conformable substrate or carrier
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B2562/00—Details of sensors; Constructional details of sensor housings or probes; Accessories for sensors
- A61B2562/16—Details of sensor housings or probes; Details of structural supports for sensors
- A61B2562/166—Details of sensor housings or probes; Details of structural supports for sensors the sensor is mounted on a specially adapted printed circuit board
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/48—Other medical applications
- A61B5/4836—Diagnosis combined with treatment in closed-loop systems or methods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F2013/00361—Plasters
- A61F2013/00902—Plasters containing means
- A61F2013/0094—Plasters containing means for sensing physical parameters
- A61F2013/00948—Ph, e.g. acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/75—General characteristics of the apparatus with filters
- A61M2205/7518—General characteristics of the apparatus with filters bacterial
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/75—General characteristics of the apparatus with filters
- A61M2205/7536—General characteristics of the apparatus with filters allowing gas passage, but preventing liquid passage, e.g. liquophobic, hydrophobic, water-repellent membranes
Description
本出願は、2017年9月27日に米国仮特許出願第62/564,126号で出願された、名称「PH SENGING FOR SENSOR ENABLED NEGATIVE PRESSURE WOUND MONITORING AND THERAPY APPARATUSES」の利益を主張し、その開示は、参照によりその全体が本明細書に組み込まれる。
創傷または創傷を囲む皮膚のうちの少なくとも一方を、創傷または創傷を囲む皮膚と接触して位置決めされた創傷被覆材を用いて監視することであって、創傷被覆材は、創傷のpHの変化に応答して色を変化させるように構成されたpH感受性材料と、pH感受性材料の色変化を検出するように構成された光学センサと、を含む、監視することと、
光学センサからの検出された色変化に基づいて、プロセッサでpH値を計算することと、を含んでもよい。
本明細書に開示するいくつかの実施形態は、ヒトまたは動物の身体に対する創傷療法に関する。そのため、本明細書における創傷へのいかなる言及も、ヒトまたは動物の身体上の創傷を指すことができ、本明細書における身体へのいかなる言及も、ヒトまたは動物の身体を指し得る。開示技術実施形態は、生理学的組織または生体組織への損傷の防止または最小化、または例えば、陰圧源および創傷被覆材コンポーネントおよび装置などを含む、減圧を伴うまたは伴わない損傷した組織(例えば、本明細書で説明した創傷など)の治療に関連し得る。創傷オーバーレイおよびパッキング材料、または、存在する場合には内層を備える、装置およびコンポーネントは、時に総称して本明細書では被覆材と呼ばれる。いくつかの実施形態では、創傷被覆材は、減圧せずに利用されるように提供され得る。
いくつかの実施形態では、創傷の治癒を容易にするおよび促進するために、創傷に被覆材に減圧または陰圧を適用することができる、陰圧創傷療法を使用してそのような創傷の治療を実施することができる。本明細書に開示される創傷被覆材および方法は、身体の他の部分に適用されてもよく、創傷の治療に必ずしも限定されないことも、理解されるであろう。
いくつかの実施形態では、創傷被覆材装置は、1つ以上のナノテクノロジー対応センサ(ナノセンサとも称される)を組み込むか、またはそれを含むことができる。ナノセンサを利用して、体内の細胞の体積、濃度、変位および速度、重力、電気、および磁力、圧力、または温度の変化を測定することができる。ナノセンサは、体内の特定の場所に薬を送達するか、または開発を監視するために、特定の細胞を分子レベルで識別または認識できる場合がある。ナノセンサは、例えば、創傷を監視し、傷がどれほど良く治癒しているかに基づいて治療計画を推奨するために使用できる創傷の特性を検出することができる。一組のナノセンサは、集合コミュニティとして機能することができる。例えば、ナノセンサはネットワークとして通信することができ、基材(例えば、創傷空洞内に設置できる発泡体または創傷充填材)に配合することができる。
センサまたはセンサアレイの正確な設置は、創傷の効果的な治療、または効果的なデータ収集に重要であり得る。例えば、創傷のおよびその周囲の異なる場所は、劇的に異なる特性を有し得る。センサがどこに位置付けられているかを知らなければ(例えば、創傷、他のセンサ、患者などに対して)、測定されたデータを誤解または不正確なものにする恐れがあり、それにより、患者に効果的な治療を提供することが困難である。したがって、いくつかの実施形態では、センサデータの精度の向上を支援するために、1つ以上の技術を利用する。例えば、不完全または不正確な設置の可能性を低減するために、1つ以上の技術が提供されている。加えて、不完全または不正確な設置にもかかわらず、センサデータの精度を向上させるために、1つ以上の技術が提供されている。同様に、正確な情報を収集するために、センサの特定で正確な設置を必要としない1つ以上の技術が提供されている。
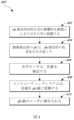
図4は、いくつかの実施形態による、創傷被覆材上のpH感受性材料を利用する創傷治療方法400を図示する。特定の実施形態では、pH感受性材料を含む創傷被覆材または創傷パッキング材料は、創傷402上またはその中に設置され得る。pH感受性材料には、pH感受性染料、pH感受性顔料、pH感受性インク、pH感受性超吸収材、pH感受性接着ゲルまたは非接着性ゲル、pH感受性接着性発泡体または非接着性発泡体、pH感受性親水性ポリマー、pH感受性疎水性ポリマー、またはその他の同様の材料を含み得る。例えば、Smith&NephewによるCUTINOVA(商標)水力被覆材に見出されるものなどのポリウレタンゲルマトリクスは、多少の変更を加えたpH感受性材料として好適であり得る。こうしたpH感受性材料は、異なるpH値で色を変化させるであろう要素(染料分子など)を組み込む。当業者には理解されるように、pH感受性要素は、化学結合(イオン、共有結合、および/または極性共有結合など)を介してポリマーの骨格に直接取り付けられるか、ポリマーに吸収されるか、ポリマーに接着されるか、または他の好適な手段を介して取り付けられ得る。さらに、当業者には理解されるように、pH感知材料またはpH感受性材料は、本明細書全体で同義的に使用されてもよい。
1.2-[4(2-ヒドロキシエチルスルホニル)-フェニル]ジアゼニル-4-メチルフェノール(GJM514)(32mg)および4-[4-(2-ヒドロキシエチルスルホニル)-フェニルアゾ]-2、6-ジメトキシフェノール(GJM534)(18mg)(1:0.5)を計量し、これに280μlの硫酸(濃縮物)を加え、30分間反応させる
2.500mlのメスフラスコに446mlの脱イオン水を入れ、これに染料溶液を加える(30分後)
3.これに4.0mlの水酸化ナトリウム(10mlの脱イオン水中3.2gの水酸化ナトリウムペレットなどの32%w/v溶液)を加える
4.次に、50mlの炭酸ナトリウム溶液(2.36M)を加え、脱イオン水を500mlのマークまで追加する
5.ポリウレタンサンプルEU33(BASF Elastogran SP9109 ポリウレタン)をビーカーに入れる
6.ポリウレタンサンプルに染料溶液を加え、攪拌しながら2時間反応させる
7.染料溶液を除去し、次に、脱イオン水(250ml)で洗浄し、穏やかに短時間攪拌した後、洗浄液を除去する
8.水に染料が出なくなるまで、この洗浄ステップを繰り返す
9.最後に脱イオン水(250ml)でサンプルを洗浄する
10.その後、サンプルは酸性溶液、次に塩基溶液に曝露され、色変化に注意することにより評価される
様々な因子は、時間と共にpHセンサ測定値の変動性を異なる創傷内で増大させ得る。例えば、異なる外部光源を介してなどでの照明の変動、pH感受性材料の色特性の変化、pH検出センサの変化、および周囲のその他の態様およびシステム自体は、センサ測定値の変動性を増大させ得る。こうした変動性は、色変化測定値の精度を低下させ得る。したがって、特定の実施形態では、上に記載したものなどの光学pH検出センサは、上に記載したような創傷の実際のpH特性以外の因子によって生じる光学センサの読み取り値の変動性を回避するために、基準材料に正規化/較正される必要があり得る。当業者であれば、正規化および較正が本開示内で互換的に使用され得ることを理解するであろう。
いくつかの実施形態では、1つ以上の電子部品は、創傷に面する側の反対側の創傷接触層の側に位置決めされ得る。本明細書に記載するシステムおよび方法は、そのような創傷接触層に等しく適用可能である。本明細書に記載する創傷被覆材の実施形態は、その他の記載された創傷被覆材の実施形態のいずれかの特徴を含み得る。同様に、本明細書に記載する任意のコントローラは、その他の記載された創傷被覆材の実施形態のいずれかの特徴を含み得る。さらに、特定の実施形態に記載した任意のデバイス、コンポーネント、またはモジュールは、その他の記載されたデバイス、コンポーネント、またはモジュールの実施形態のいずれかの特徴を含み得る。
[付記項1]
創傷監視システムであって、
創傷と接触して位置決めされるように構成された創傷被覆材であって、色を測定するように構成された光学センサを備える、創傷被覆材と、
前記創傷被覆材の底面上に位置決めされたpH感受性材料であって、前記創傷のpHの変化に応答して色を変化させるように構成された、pH感受性材料と、を備え、
前記光学センサが、前記pH感受性材料の前記色変化の検出に基づいて、前記創傷の前記pHを検出するようにさらに構成されている、創傷監視システム。
[付記項2]
前記pH感受性材料が、親水性ポリマー、ゲル、または発泡体を含む、付記項1に記載のシステム。
[付記項3]
前記pH感受性材料には、pH感受性要素が散在している、付記項1~2のいずれか一項に記載のシステム。
[付記項4]
前記pH感受性要素が、不均一に分散されている、付記項3に記載のシステム。
[付記項5]
前記pH感受性要素が、均一に分散されている、付記項3に記載のシステム。
[付記項6]
前記pH感受性材料が、接着材料を含む、付記項1~5のいずれか一項に記載のシステム。
[付記項7]
前記pH感受性材料が、ポリウレタンを含む、付記項1~6のいずれか一項に記載のシステム。
[付記項8]
前記創傷被覆材の前記底面上に位置決めされた非pH感受性材料をさらに含み、前記非pH感受性材料が、創傷滲出液を前記pH感受性材料に誘導するように構成されている、付記項1~7のいずれか一項に記載のシステム。
[付記項9]
前記非pH感受性材料が、親水性ポリマー、ゲル、または発泡体を含む、付記項8に記載のシステム。
[付記項10]
前記非pH感受性材料が、前記創傷被覆材の前記底面上に1つまたは複数のチャネルとして配設されている、付記項8または9に記載のシステム。
[付記項11]
前記1つまたは複数のチャネルが、前記pH感受性材料から前記被覆材の縁まで延在している、付記項10に記載のシステム。
[付記項12]
前記非pH感受性材料が、接着材料を含む、付記項8~11のいずれか一項に記載のシステム。
[付記項13]
コントローラをさらに備え、前記コントローラが、前記光学センサによって測定された前記色をpH値に変換するように構成されている、付記項1~12のいずれか一項に記載のシステム。
[付記項14]
前記コントローラが、前記pH値の表示をユーザに提供するように構成されている、付記項13に記載のシステム。
[付記項15]
前記コントローラが、前記pH値を表示するようにさらに構成されている、付記項14に記載のシステム。
[付記項16]
基準材料をさらに含み、前記基準材料が、安定した色を維持するように構成されている、付記項1~15のいずれか一項に記載のシステム。
[付記項17]
前記基準材料が、前記創傷被覆材に組み込まれている、付記項16に記載のシステム。
[付記項18]
創傷のpHを監視する方法であって、
創傷または創傷を囲む皮膚のうちの少なくとも一方を、前記創傷または前記創傷を囲む皮膚と接触して位置決めされた創傷被覆材を用いて監視することであって、前記創傷被覆材は、前記創傷のpHの変化に応答して色を変化するように構成されたpH感受性材料と、前記pH感受性材料の色変化を検出するように構成された光学センサと、を含む、監視することと、
前記光学センサからの前記検出された色変化に基づいて、プロセッサでpH値を計算することと、を含む、方法。
[付記項19]
前記光学センサが、基準材料の色値を検出するように構成されており、前記pH感受性材料の色値を前記基準材料に正規化することをさらに含む、付記項18に記載の方法。
[付記項20]
前記基準材料が、安定した色を維持するように構成されている、付記項19に記載の方法。
[付記項21]
付記項1~17のいずれか一項に記載のシステムを使用する方法。
[付記項22]
実質的に示されおよび/または記載されるような装置。
[付記項23]
実質的に示されおよび/または記載されるような方法。
[付記項24]
実質的に示されおよび/または記載されるようなシステム。
Claims (15)
- 創傷と接触して位置決めされるように構成された創傷被覆材(500、604)であって、色を測定するように構成された光学センサ(504、608)を備える、創傷被覆材と、
前記創傷被覆材の底面に位置決めされたpH感受性材料であって、前記創傷のpHの変化に応答して色を変化させるように構成され、親水性ポリマー、親水性ゲル、または親水性発泡体を含む、pH感受性材料と、を備え、
前記光学センサが、前記pH感受性材料の色変化の検出に基づいて、前記創傷の前記pHを検出するように構成されており、
前記創傷被覆材の前記底面に位置決めされた非pH感受性材料をさらに含み、前記非pH感受性材料が、創傷滲出液を前記pH感受性材料に誘導するようにさらに構成されている、創傷監視システム。 - 前記pH感受性材料には、pH感受性要素が散在している、請求項1に記載のシステム。
- 前記pH感受性要素が、不均一に分散されている、請求項2に記載のシステム。
- 前記pH感受性要素が、均一に分散されている、請求項2に記載のシステム。
- 前記pH感受性材料が、接着材料を含む、請求項1~4のいずれか一項に記載のシステム。
- 前記pH感受性材料が、ポリウレタンを含む、請求項1~5のいずれか一項に記載のシステム。
- 前記非pH感受性材料が、親水性ポリマー、ゲル、または発泡体を含む、請求項1~6のいずれか一項に記載のシステム。
- 前記非pH感受性材料が、前記創傷被覆材の前記底面に1以上のチャネルとして配設されている、請求項1~7のいずれか一項に記載のシステム。
- 1以上の前記チャネルが、前記pH感受性材料から前記創傷被覆材の縁まで延在している、請求項8に記載のシステム。
- 前記非pH感受性材料が、接着材料を含む、請求項1~9のいずれか一項に記載のシステム。
- コントローラをさらに備え、前記コントローラが、前記光学センサによって測定された前記色をpH値に変換するように構成されている、請求項1~10のいずれか一項に記載のシステム。
- 前記コントローラが、前記pH値の表示をユーザに提供するように構成されている、請求項11に記載のシステム。
- 前記コントローラが、前記pH値を表示するようにさらに構成されている、請求項12に記載のシステム。
- 基準材料をさらに含み、前記基準材料が、安定した色を維持するように構成されている、請求項1~13のいずれか一項に記載のシステム。
- 前記基準材料が、前記創傷被覆材に組み込まれている、請求項14に記載のシステム。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762564126P | 2017-09-27 | 2017-09-27 | |
| US62/564,126 | 2017-09-27 | ||
| PCT/EP2018/075802 WO2019063481A1 (en) | 2017-09-27 | 2018-09-24 | PH DETECTION FOR NEGATIVE PRESSURE WOUND THERAPY SURVEILLANCE AND THERAPY APPARATUS |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2020537553A JP2020537553A (ja) | 2020-12-24 |
| JP2020537553A5 JP2020537553A5 (ja) | 2021-11-04 |
| JP7282079B2 true JP7282079B2 (ja) | 2023-05-26 |
Family
ID=63722366
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020516581A Active JP7282079B2 (ja) | 2017-09-27 | 2018-09-24 | センサが使用可能な陰圧創傷監視および療法装置のph感知 |
Country Status (5)
| Country | Link |
|---|---|
| US (2) | US11596553B2 (ja) |
| EP (1) | EP3687380A1 (ja) |
| JP (1) | JP7282079B2 (ja) |
| CN (1) | CN111132605B (ja) |
| WO (1) | WO2019063481A1 (ja) |
Families Citing this family (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018162736A1 (en) | 2017-03-09 | 2018-09-13 | Smith & Nephew Plc | Wound dressing, patch member and method of sensing one or more wound parameters |
| US11324424B2 (en) | 2017-03-09 | 2022-05-10 | Smith & Nephew Plc | Apparatus and method for imaging blood in a target region of tissue |
| SG11201909449TA (en) | 2017-04-11 | 2019-11-28 | Smith & Nephew | Component positioning and stress relief for sensor enabled wound dressings |
| CN110832598B (zh) | 2017-05-15 | 2024-03-15 | 史密夫及内修公开有限公司 | 伤口分析装置和方法 |
| AU2018288530B2 (en) | 2017-06-23 | 2024-03-28 | Smith & Nephew Plc | Positioning of sensors for sensor enabled wound monitoring or therapy |
| GB201804502D0 (en) | 2018-03-21 | 2018-05-02 | Smith & Nephew | Biocompatible encapsulation and component stress relief for sensor enabled negative pressure wound therapy dressings |
| GB201809007D0 (en) | 2018-06-01 | 2018-07-18 | Smith & Nephew | Restriction of sensor-monitored region for sensor-enabled wound dressings |
| US11925735B2 (en) | 2017-08-10 | 2024-03-12 | Smith & Nephew Plc | Positioning of sensors for sensor enabled wound monitoring or therapy |
| GB201804971D0 (en) | 2018-03-28 | 2018-05-09 | Smith & Nephew | Electrostatic discharge protection for sensors in wound therapy |
| JP2020533093A (ja) | 2017-09-10 | 2020-11-19 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | 封入を検査するためのシステムおよび方法、ならびにセンサを装備した創傷被覆材内の構成要素 |
| GB201718870D0 (en) | 2017-11-15 | 2017-12-27 | Smith & Nephew Inc | Sensor enabled wound therapy dressings and systems |
| GB201718859D0 (en) | 2017-11-15 | 2017-12-27 | Smith & Nephew | Sensor positioning for sensor enabled wound therapy dressings and systems |
| US11596553B2 (en) | 2017-09-27 | 2023-03-07 | Smith & Nephew Plc | Ph sensing for sensor enabled negative pressure wound monitoring and therapy apparatuses |
| EP3687396A1 (en) | 2017-09-28 | 2020-08-05 | Smith & Nephew plc | Neurostimulation and monitoring using sensor enabled wound monitoring and therapy apparatus |
| JP2021502845A (ja) | 2017-11-15 | 2021-02-04 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | 統合センサ対応型創傷モニタリングおよび/または治療被覆材ならびにシステム |
| GB2592508B (en) | 2018-09-12 | 2022-08-31 | Smith & Nephew | Device, apparatus and method of determining skin perfusion pressure |
| WO2021074856A1 (en) * | 2019-10-18 | 2021-04-22 | Kci Licensing, Inc. | Wound therapy system and wound dressing with ph sensor |
| US11399750B2 (en) | 2020-06-18 | 2022-08-02 | Covidien Lp | Hydrophobic materials in a medical sensor |
| WO2022266070A1 (en) | 2021-06-14 | 2022-12-22 | Preh Holding, Llc | Connected body surface care module |
| KR102564069B1 (ko) * | 2021-11-17 | 2023-08-04 | 성균관대학교산학협력단 | 환부 상태 모니터링 장치 및 이를 포함하는 환부 상태 모니터링용 드레싱 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005179314A (ja) | 2003-12-24 | 2005-07-07 | Life Kea Giken Kk | 皮膚検査用貼付剤 |
| US20130217983A1 (en) | 2011-12-29 | 2013-08-22 | Wellsense, Inc. | Analyte sensor |
| US20160015962A1 (en) | 2014-07-16 | 2016-01-21 | Mehdi Shokoueinejad Maragheh | Smart Patch For Wound Management |
| JP2017500167A (ja) | 2013-10-08 | 2017-01-05 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | pHインジケータードレッシング |
| WO2017041386A1 (zh) | 2015-09-12 | 2017-03-16 | 深圳市前海颐老科技有限公司 | 伤口感染程度监测系统及方法 |
| JP2017510319A5 (ja) | 2015-01-20 | 2018-02-01 |
Family Cites Families (441)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3896802A (en) | 1974-04-19 | 1975-07-29 | American Cyanamid Co | Flexible flocked dressing |
| US4334530A (en) | 1980-09-12 | 1982-06-15 | Hassell Donald S | Indicia-bearing adhesive bandages |
| AU7316287A (en) * | 1986-06-02 | 1987-12-03 | Vincent L. Vaillancourt | Naso-gastric intubation system having a ph indicator |
| US5090410A (en) | 1989-06-28 | 1992-02-25 | Datascope Investment Corp. | Fastener for attaching sensor to the body |
| US5642096A (en) | 1992-03-20 | 1997-06-24 | Paromed Medizintechnik Gmbh | Device for prevention of ulcers in the feet of diabetes patients |
| SE500972C2 (sv) | 1992-03-30 | 1994-10-10 | Moelnlycke Ab | Förfarande och anordning för tillverkning av sårförband samt ett sårförband tillverkat medelst förfarandet |
| US5253654A (en) | 1992-04-30 | 1993-10-19 | Thomas Berten R | Orthopedic weight monitor |
| EP0614652B1 (en) | 1993-03-10 | 2001-07-18 | Nichiban Co. Ltd. | Sticking material for hemostasis |
| US6178342B1 (en) | 1993-09-09 | 2001-01-23 | Vasamedics | Surface perfusion pressure monitoring system |
| US5678448A (en) | 1994-01-14 | 1997-10-21 | Fullen Systems, Inc. | System for continuously measuring forces applied by the foot |
| GB9523640D0 (en) | 1995-11-18 | 1996-01-17 | Bristol Myers Squibb Co | Bandaging system |
| US5703225A (en) | 1995-12-13 | 1997-12-30 | Kimberly-Clark Worldwide, Inc. | Sulfonated cellulose having improved absorbent properties |
| JPH09187431A (ja) * | 1996-01-12 | 1997-07-22 | Hitachi Ltd | テレメータ端末 |
| GB9615895D0 (en) | 1996-07-29 | 1996-09-11 | Thames Medical Ltd | Pulse oximeter |
| US20020099318A1 (en) | 1997-05-16 | 2002-07-25 | Susan Suehr | Multi-layer compression bandage |
| DE19722075C1 (de) | 1997-05-27 | 1998-10-01 | Wilhelm Dr Med Fleischmann | Vorrichtung zur Applikation von Wirkstoffen an einer Wundoberfläche |
| US7206623B2 (en) | 2000-05-02 | 2007-04-17 | Sensys Medical, Inc. | Optical sampling interface system for in vivo measurement of tissue |
| US5836990A (en) | 1997-09-19 | 1998-11-17 | Medtronic, Inc. | Method and apparatus for determining electrode/tissue contact |
| US6381482B1 (en) | 1998-05-13 | 2002-04-30 | Georgia Tech Research Corp. | Fabric or garment with integrated flexible information infrastructure |
| US6095992A (en) | 1998-04-06 | 2000-08-01 | Augustine Medical, Inc. | Wound treatment apparatus for normothermic treatment of wounds |
| US6343224B1 (en) | 1998-10-15 | 2002-01-29 | Sensidyne, Inc. | Reusable pulse oximeter probe and disposable bandage apparatus |
| FR2785544B1 (fr) | 1998-11-09 | 2001-01-05 | Lhd Lab Hygiene Dietetique | Electrode de transfert d'un courant electrique traversant la peau d'un patient |
| WO2000043046A2 (en) | 1999-01-21 | 2000-07-27 | Rhoda Zione | Wound management system and wound dressing |
| US7047054B2 (en) | 1999-03-12 | 2006-05-16 | Cas Medical Systems, Inc. | Laser diode optical transducer assembly for non-invasive spectrophotometric blood oxygenation monitoring |
| US6856821B2 (en) | 2000-05-26 | 2005-02-15 | Kci Licensing, Inc. | System for combined transcutaneous blood gas monitoring and vacuum assisted wound closure |
| US6669663B1 (en) | 1999-04-30 | 2003-12-30 | Medtronic, Inc. | Closed loop medicament pump |
| US20030208148A1 (en) | 1999-11-01 | 2003-11-06 | Sullivan John Patrick | Adhesive bandage with soft, three-dimensional toy figure |
| US6517484B1 (en) | 2000-02-28 | 2003-02-11 | Wilk Patent Development Corporation | Ultrasonic imaging system and associated method |
| US6639674B2 (en) | 2000-03-28 | 2003-10-28 | Board Of Regents, The University Of Texas System | Methods and apparatus for polarized reflectance spectroscopy |
| AU5359901A (en) | 2000-04-17 | 2001-10-30 | Vivometrics Inc | Systems and methods for ambulatory monitoring of physiological signs |
| US7520875B2 (en) | 2001-04-06 | 2009-04-21 | Mattioli Engineering Ltd. | Method and apparatus for skin absorption enhancement and transdermal drug delivery |
| CA2452376A1 (en) | 2001-07-02 | 2003-01-16 | Battelle Memorial Institute | Intelligent microsensor module |
| DE60315805D1 (de) | 2002-01-23 | 2007-10-04 | Bang & Olufsen Medicom As | Blutdruckmessvorrichtung mit manschette aus zwei öffnenden konkaven schalenteilen |
| US7169107B2 (en) | 2002-01-25 | 2007-01-30 | Karen Jersey-Willuhn | Conductivity reconstruction based on inverse finite element measurements in a tissue monitoring system |
| GB0202654D0 (en) | 2002-02-06 | 2002-03-20 | Univ Nottingham | Examination of superficial regions of a body |
| US7158660B2 (en) | 2002-05-08 | 2007-01-02 | Gee Jr James W | Method and apparatus for detecting structures of interest |
| US20070100666A1 (en) | 2002-08-22 | 2007-05-03 | Stivoric John M | Devices and systems for contextual and physiological-based detection, monitoring, reporting, entertainment, and control of other devices |
| US7846141B2 (en) | 2002-09-03 | 2010-12-07 | Bluesky Medical Group Incorporated | Reduced pressure treatment system |
| US8111165B2 (en) | 2002-10-02 | 2012-02-07 | Orthocare Innovations Llc | Active on-patient sensor, method and system |
| GB0224986D0 (en) | 2002-10-28 | 2002-12-04 | Smith & Nephew | Apparatus |
| JP2004158605A (ja) | 2002-11-06 | 2004-06-03 | Konica Minolta Holdings Inc | プリント配線基板、及びプリント配線基板の導電性筐体への取付方法 |
| GB0228375D0 (en) | 2002-12-05 | 2003-01-08 | Innovation And Entpr Off Of | Wound mapping |
| DE602004026280D1 (de) | 2003-02-07 | 2010-05-12 | Alfred E Mann Inst Biomed Eng | Chirurgischer drain mit sensoren zur überwachung des internen gewebezustands und zur überwachung von flüssigkeit in lumen |
| GB0303797D0 (en) | 2003-02-19 | 2003-03-26 | Huntleigh Technology Plc | Blood assessment |
| US7706862B2 (en) | 2003-04-17 | 2010-04-27 | Research Foundation Of The City University Of New York | Detecting human cancer through spectral optical imaging using key water absorption wavelengths |
| US20060181791A1 (en) | 2003-07-31 | 2006-08-17 | Van Beek Michael C | Method and apparatus for determining a property of a fluid which flows through a biological tubular structure with variable numerical aperture |
| WO2005023361A2 (en) | 2003-09-10 | 2005-03-17 | Power Paper Ltd. | Disposable electric bandage |
| WO2005027730A2 (en) | 2003-09-19 | 2005-03-31 | The General Hospital Corporation | Fluorescence polarization imaging devices and methods |
| TWI220787B (en) | 2003-10-24 | 2004-09-01 | Asustek Comp Inc | Electric device with electrostatic discharge protection structure thereof |
| WO2005044090A2 (en) | 2003-11-04 | 2005-05-19 | General Hospital Corporation | Respiration motion detection and health state assessment system |
| WO2005057658A1 (en) | 2003-12-15 | 2005-06-23 | Semiconductor Energy Laboratory Co., Ltd. | Method for manufacturing thin film integrated circuit device, noncontact thin film integrated circuit device and method for manufacturing the same, and idtag and coin including the noncontact thin film integrated circuit device |
| DK1718257T3 (da) | 2004-02-13 | 2010-10-18 | Convatec Technologies Inc | Flerlaget sårforbinding |
| US7904133B2 (en) | 2004-02-27 | 2011-03-08 | Koninklijke Philips Electronics N.V. | Wearable wireless device for monitoring, analyzing and communicating physiological status |
| WO2005084537A1 (en) | 2004-03-08 | 2005-09-15 | Medicus Engineering Aps | A method and an instrument for measuring of physiological parameters |
| CA2560323C (en) | 2004-03-22 | 2014-01-07 | Bodymedia, Inc. | Non-invasive temperature monitoring device |
| US7884258B2 (en) | 2004-04-13 | 2011-02-08 | Boehringer Technologies, L.P. | Wound contact device |
| GB0408492D0 (en) | 2004-04-16 | 2004-05-19 | Univ Strathclyde | Performance measurement of wound dressings |
| US7753894B2 (en) | 2004-04-27 | 2010-07-13 | Smith & Nephew Plc | Wound cleansing apparatus with stress |
| GB0508528D0 (en) | 2005-04-27 | 2005-06-01 | Smith & Nephew | SAI with macrostress |
| US7201063B2 (en) | 2004-04-30 | 2007-04-10 | Taylor Geoffrey L | Normal force gradient/shear force sensors and method of measuring internal biological tissue stress |
| US7521292B2 (en) | 2004-06-04 | 2009-04-21 | The Board Of Trustees Of The University Of Illinois | Stretchable form of single crystal silicon for high performance electronics on rubber substrates |
| US20050280531A1 (en) | 2004-06-18 | 2005-12-22 | Fadem Kalford C | Device and method for transmitting physiologic data |
| SE0401632D0 (sv) | 2004-06-24 | 2004-06-24 | Innovation Team Ab | Medel och sätt att detektera blodläckage från sår |
| US20060058593A1 (en) | 2004-09-02 | 2006-03-16 | Drinan Darrel D | Monitoring platform for detection of hypovolemia, hemorrhage and blood loss |
| US20060058690A1 (en) | 2004-09-10 | 2006-03-16 | Optical Sensors, Inc. | Method and instrument for automated measurement of skin perfusion pressure |
| EP1799139B1 (en) | 2004-10-05 | 2008-12-24 | Koninklijke Philips Electronics N.V. | Skin treatment device with radiation emission protection |
| US8498681B2 (en) | 2004-10-05 | 2013-07-30 | Tomophase Corporation | Cross-sectional mapping of spectral absorbance features |
| NL1027236C2 (nl) | 2004-10-13 | 2006-04-18 | Innova Medical V O F | Verbandmiddel voor een gekwetst lichaamsdeel. |
| US20060142560A1 (en) | 2004-12-29 | 2006-06-29 | Glasser Wolfgang G | Sulfoalkylated cellulose |
| US9597024B2 (en) | 2005-02-09 | 2017-03-21 | Medici Instruments Llc | Methods and apparatuses for noninvasive determinations of analytes |
| US20080287747A1 (en) | 2005-02-28 | 2008-11-20 | Michael Mestrovic | Flexible Electronic Device |
| US20060241495A1 (en) | 2005-03-23 | 2006-10-26 | Eastman Kodak Company | Wound healing monitoring and treatment |
| EP1709947A1 (en) | 2005-04-08 | 2006-10-11 | 3M Innovative Properties Company | Compression bandage system |
| EP2420185A3 (en) | 2005-04-14 | 2012-09-05 | Hidalgo Limited | Apparatus and system for monitoring |
| US8060174B2 (en) | 2005-04-15 | 2011-11-15 | Dexcom, Inc. | Analyte sensing biointerface |
| CN101257875A (zh) | 2005-09-06 | 2008-09-03 | 泰科保健集团有限合伙公司 | 具有微型泵的独立伤口敷料 |
| EP1933697A4 (en) | 2005-09-06 | 2011-05-25 | Vaesamed Inc | SYSTEM FOR THE AUTOMATIC MEASUREMENT OF SKIN PERFUSION PRESSURE |
| MX2008002880A (es) | 2005-09-07 | 2008-03-27 | Tyco Healthcare | Aparato de aposito independiente de herida. |
| EP1922095A2 (en) | 2005-09-07 | 2008-05-21 | Tyco Healthcare Group LP | Wound dressing with vacuum reservoir |
| GB0519836D0 (en) | 2005-09-29 | 2005-11-09 | Smartlife Technology Ltd | Contact sensors |
| US8032210B2 (en) | 2005-10-06 | 2011-10-04 | Spinematrix, Inc. | EMG diagnostic system and method |
| US7420472B2 (en) | 2005-10-16 | 2008-09-02 | Bao Tran | Patient monitoring apparatus |
| US7877866B1 (en) | 2005-10-26 | 2011-02-01 | Second Sight Medical Products, Inc. | Flexible circuit electrode array and method of manufacturing the same |
| US8333874B2 (en) | 2005-12-09 | 2012-12-18 | Flexible Medical Systems, Llc | Flexible apparatus and method for monitoring and delivery |
| CN101341424B (zh) | 2005-12-22 | 2012-07-18 | 皇家飞利浦电子股份有限公司 | 磁感应断层成像系统和方法 |
| US7816577B2 (en) | 2006-02-13 | 2010-10-19 | Aalnex, Inc. | Wound shield |
| CA2538940A1 (en) | 2006-03-03 | 2006-06-22 | James W. Haslett | Bandage with sensors |
| DE102006045138A1 (de) | 2006-03-27 | 2007-11-15 | Siemens Ag | Vorrichtung, Sensor, Sensorelement sowie Verfahren zur Vermessung des Wirbelsäulenverlaufs und von Verlaufsänderungen der Wirbelsäule |
| US7607243B2 (en) | 2006-05-03 | 2009-10-27 | Nike, Inc. | Athletic or other performance sensing systems |
| US8644911B1 (en) | 2006-06-30 | 2014-02-04 | Hypermed Imaging, Inc. | OxyVu-1 hyperspectral tissue oxygenation (HTO) measurement system |
| GB2439750A (en) | 2006-07-06 | 2008-01-09 | Wound Solutions Ltd | Monitoring a limb wound |
| WO2008006150A1 (en) | 2006-07-11 | 2008-01-17 | Citech Research Ip Pty Ltd | Bio-activity data capture and transmission |
| WO2008010604A1 (fr) | 2006-07-19 | 2008-01-24 | School Juridical Person Kitasato Gakuen | Dispositif d'imagerie des vaisseaux sanguins et système pour analyser une distribution des vaisseaux sanguins |
| US9254220B1 (en) | 2006-08-29 | 2016-02-09 | Vasamed, Inc. | Method and system for assessing severity and stage of peripheral arterial disease and lower extremity wounds using angiosome mapping |
| US7890153B2 (en) | 2006-09-28 | 2011-02-15 | Nellcor Puritan Bennett Llc | System and method for mitigating interference in pulse oximetry |
| GB0620061D0 (en) | 2006-10-10 | 2006-11-22 | Medical Device Innovations Ltd | Oesophageal treatment apparatus and method |
| US7785301B2 (en) | 2006-11-28 | 2010-08-31 | Vadim V Yuzhakov | Tissue conforming microneedle array and patch for transdermal drug delivery or biological fluid collection |
| US8019401B1 (en) | 2006-12-04 | 2011-09-13 | Smithmarks, Inc. | Stretchable electrode and method of making physiologic measurements |
| NZ551819A (en) | 2006-12-04 | 2009-03-31 | Zephyr Technology Ltd | Impact detection system |
| US8238996B2 (en) | 2006-12-05 | 2012-08-07 | Tyco Healthcare Group Lp | Electrode array |
| US8100834B2 (en) | 2007-02-27 | 2012-01-24 | J&M Shuler, Inc. | Method and system for monitoring oxygenation levels of a compartment for detecting conditions of a compartment syndrome |
| US7687678B2 (en) | 2007-05-10 | 2010-03-30 | Cisco Technology, Inc. | Electronic bandage with flexible electronic controller |
| ATE524108T1 (de) | 2007-06-19 | 2011-09-15 | Biocompatibles Uk Ltd | Verfahren und vorrichtung zum messen der hauttextur |
| CA2696932A1 (en) | 2007-08-22 | 2009-02-26 | Commonwealth Scientific And Industrial Research Organisation | A system, garment and method |
| WO2009036313A1 (en) | 2007-09-14 | 2009-03-19 | Corventis, Inc. | Adherent device with multiple physiological sensors |
| WO2009052607A1 (en) | 2007-10-24 | 2009-04-30 | Perceptronix Medical Inc. | Method and apparatus for microvascular oxygenation imaging |
| US8116838B2 (en) | 2007-11-27 | 2012-02-14 | Carnegie Mellon University | Medical device for diagnosing pressure ulcers |
| GB0723898D0 (en) | 2007-12-06 | 2008-01-16 | Wound Solutions Ltd | Wound treatment device and method |
| CA2708497A1 (en) | 2007-12-10 | 2009-06-18 | Isis Biopolymer Llc | Iontophoretic drug delivery device and software application |
| US9357944B2 (en) | 2008-01-08 | 2016-06-07 | Cardiac Pacemakers, Inc. | Impedance measurement and demodulation using implantable device |
| ES2382595T3 (es) | 2008-01-08 | 2012-06-11 | Bluesky Medical Group Inc. | Tratamiento de heridas mediante presión negativa variable y sostenida y método para controlar el mismo |
| US20090177051A1 (en) | 2008-01-09 | 2009-07-09 | Heal-Ex, Llc | Systems and methods for providing sub-dressing wound analysis and therapy |
| GB0801264D0 (en) | 2008-01-24 | 2008-02-27 | Univ Ulster | Electrically enhances wound healing system and method |
| WO2009103034A2 (en) | 2008-02-13 | 2009-08-20 | Board Of Regents, The University Of Texas System | System, method and apparatus for an amorphous iridium oxide film ph sensor |
| WO2009114624A2 (en) | 2008-03-12 | 2009-09-17 | Bluesky Medical Group Inc. | Negative pressure dressing and method of using same |
| US8161826B1 (en) | 2009-03-05 | 2012-04-24 | Stryker Corporation | Elastically stretchable fabric force sensor arrays and methods of making |
| JP2009225863A (ja) | 2008-03-19 | 2009-10-08 | Nemoto Kyorindo:Kk | 剥離シート付き両面粘着シート |
| WO2009120951A2 (en) | 2008-03-28 | 2009-10-01 | Nordson Corporation | Automated conformal coating inspection system and methods of use |
| US7792334B2 (en) | 2008-03-31 | 2010-09-07 | Immersion Corporation | Locating blood vessels |
| ES2715633T3 (es) * | 2008-05-20 | 2019-06-05 | Univ Health Network | Dispositivo y método para formación de imágenes y supervisión por fluorescencia |
| WO2009141777A1 (en) | 2008-05-23 | 2009-11-26 | Koninklijke Philips Electronics N.V. | A substrate layer adapted to carry sensors, actuators or electrical components |
| BRPI0908620A2 (pt) | 2008-05-23 | 2016-06-14 | Koninkl Philips Electronics Nv | conjunto de sensores adaptado para ser fixado a uma superfície do corpo de um ser humano ou animal ou uma espécie biológica e adaptado para medir uma temperatura de corpo do núcleo e método para manufaturar um conjunto de sensores |
| CA2765419C (en) | 2008-06-13 | 2017-10-24 | Premco Medical Systems, Inc. | Wound treatment apparatus and method |
| EP2312998B1 (en) | 2008-07-18 | 2018-12-05 | Flexcon Company, Inc. | High impedance signal detection systems and methods for use in electrocardiogram detection systems |
| US20100022990A1 (en) | 2008-07-25 | 2010-01-28 | Boehringer Technologies, L.P. | Pump system for negative pressure wound therapy and improvements thereon |
| WO2010020919A1 (en) | 2008-08-22 | 2010-02-25 | Koninklijke Philips Electronics N.V. | Monitoring of a fluid accumulation in a body of a person |
| US8389862B2 (en) | 2008-10-07 | 2013-03-05 | Mc10, Inc. | Extremely stretchable electronics |
| US9221963B2 (en) | 2008-11-27 | 2015-12-29 | Speciality Fibres And Materials Ltd. | Absorbent material |
| US8503712B2 (en) | 2008-12-31 | 2013-08-06 | Motorola Mobility Llc | Method and apparatus for determining blood oxygenation using a mobile communication device |
| FR2940904B1 (fr) | 2009-01-13 | 2012-08-31 | Urgo Laboratoires | Systeme de mesure de pression d'interface |
| DE102009008885A1 (de) | 2009-02-14 | 2010-08-26 | Fresenius Medical Care Deutschland Gmbh | Vorrichtung zum Detektieren von Feuchtigkeit für eine Vorrichtung zur Überwachung eines Zugangs zu einem Patienten, insbesondere zur Überwachung des Gefäßzugangs bei einer extrakkorporalen Blutbehandlung |
| WO2010105053A2 (en) | 2009-03-13 | 2010-09-16 | Corventis, Inc. | Acute patient management for military and emergency applications |
| US11278237B2 (en) | 2010-04-22 | 2022-03-22 | Leaf Healthcare, Inc. | Devices, systems, and methods for preventing, detecting, and treating pressure-induced ischemia, pressure ulcers, and other conditions |
| US8823934B2 (en) | 2009-03-27 | 2014-09-02 | Brightex Bio-Photonics Llc | Methods and systems for imaging and modeling skin using polarized lighting |
| US8332053B1 (en) | 2009-04-28 | 2012-12-11 | Hrl Laboratories, Llc | Method for fabrication of a stretchable electronic skin |
| US20110004088A1 (en) | 2009-05-13 | 2011-01-06 | Kurt Paul Grossman | The ecg shirt |
| GB0912009D0 (en) | 2009-07-10 | 2009-08-19 | Univ Strathclyde | Sensor |
| US8535282B2 (en) | 2009-07-14 | 2013-09-17 | Southwest Research Institute | Wound healing sensor techniques |
| WO2011043863A2 (en) | 2009-08-13 | 2011-04-14 | Michael Simms Shuler | Methods and dressing systems for promoting healing of injured tissue |
| JP5774590B2 (ja) | 2009-08-17 | 2015-09-09 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | 表面および表面下の生物医学的構造および状態を評価するための分散型内外部無線式センサシステム |
| US20110218756A1 (en) | 2009-10-01 | 2011-09-08 | Mc10, Inc. | Methods and apparatus for conformal sensing of force and/or acceleration at a person's head |
| GB0919659D0 (en) | 2009-11-10 | 2009-12-23 | Convatec Technologies Inc | A component for a wound dressing |
| GB0921477D0 (en) | 2009-12-08 | 2010-01-20 | Moor Instr Ltd | Apparatus for measuring blood parameters |
| US10441185B2 (en) | 2009-12-16 | 2019-10-15 | The Board Of Trustees Of The University Of Illinois | Flexible and stretchable electronic systems for epidermal electronics |
| ES2731200T3 (es) | 2009-12-22 | 2019-11-14 | Smith & Nephew Inc | Aparatos para terapia de herida por presión negativa |
| WO2011079390A1 (en) | 2009-12-30 | 2011-07-07 | Societe De Commercialisation Des Produits De La Recherche Appliquee - Socpra-Sciences Et Genie S.E.C. | Carbon nanotubes based sensing elements and system for monitoring and mapping force, strain and stress |
| EP2521931A4 (en) | 2010-01-04 | 2018-01-03 | John Stephan | Illuminatable apparatus and method of manufacturing same |
| FR2955763B1 (fr) | 2010-02-02 | 2012-03-09 | Commissariat Energie Atomique | Sonde optique peroperatoire bi-spectrale |
| EP2538841A2 (en) | 2010-02-26 | 2013-01-02 | Myskin, Inc. | Analytic methods of tissue evaluation |
| JP5663900B2 (ja) | 2010-03-05 | 2015-02-04 | セイコーエプソン株式会社 | 分光センサー装置及び電子機器 |
| WO2011113070A1 (en) | 2010-03-07 | 2011-09-15 | Centauri Medical, INC. | Systems, devices and methods for preventing, detecting, and treating pressure-induced ischemia, pressure ulcers, and other conditions |
| US9427506B2 (en) | 2010-03-31 | 2016-08-30 | Kci Licensing, Inc. | System and method for locating fluid leaks at a drape using sensing techniques |
| US8553223B2 (en) | 2010-03-31 | 2013-10-08 | Covidien Lp | Biodegradable fibers for sensing |
| US9061095B2 (en) | 2010-04-27 | 2015-06-23 | Smith & Nephew Plc | Wound dressing and method of use |
| US20130317367A1 (en) | 2010-05-04 | 2013-11-28 | Michael Simms Shuler | Method and system for providing versatile nirs sensors |
| BR112012028410A2 (pt) | 2010-05-08 | 2016-11-16 | Univ California | sistema de registro de imagens e método de registro de imagens |
| HUE059711T2 (hu) | 2010-05-08 | 2022-12-28 | Univ California | Készülék fekély korai érzékelésére szubepidermális nedvesség szkennelésével |
| US8182425B2 (en) | 2010-05-18 | 2012-05-22 | Johnson & Johnson Consumer Companies, Inc. | Method for measuring skin hydration |
| US8525340B2 (en) | 2010-06-11 | 2013-09-03 | Premitec, Inc. | Flexible electronic devices and related methods |
| US8783948B2 (en) | 2010-06-29 | 2014-07-22 | Indian Institute Of Technology Kanpur | Flexible temperature sensor and sensor array |
| US9314175B2 (en) | 2010-07-08 | 2016-04-19 | TCI3—Pressure Applications, LLC | Compartment syndrome monitoring systems and methods |
| US20120029307A1 (en) | 2010-07-27 | 2012-02-02 | Carefusion 303, Inc. | Vital-signs monitor with spaced electrodes |
| US20120029306A1 (en) | 2010-07-27 | 2012-02-02 | Carefusion 303, Inc. | Vital-signs monitor with encapsulation arrangement |
| US9585620B2 (en) | 2010-07-27 | 2017-03-07 | Carefusion 303, Inc. | Vital-signs patch having a flexible attachment to electrodes |
| EP2702969B1 (de) * | 2010-08-19 | 2014-10-08 | Paul Hartmann AG | Wundauflage enthaltend schaumstoff und salbengrundlage zur unterdrucktherapie, und vorrichtung zur unterdrucktherapie |
| US20120316486A1 (en) | 2010-08-20 | 2012-12-13 | Andrew Cheung | Surgical Component Navigation Systems And Methods |
| US9000251B2 (en) | 2010-08-26 | 2015-04-07 | Combat Medical Systems, Llc | Draining wound dressing |
| US8579872B2 (en) | 2010-10-27 | 2013-11-12 | Kci Licensing, Inc. | Reduced-pressure systems, dressings, and methods employing a wireless pump |
| RU2570283C2 (ru) | 2010-11-17 | 2015-12-10 | Смарт Солюшнз Текнолоджиз, С.Л. | Датчик для получения физиологических сигналов |
| DE102010051459A1 (de) | 2010-11-17 | 2012-05-24 | Harald Pötzschke | Wundüberwachung mit textilen Messgrößenwandler-Systemen |
| US20130261409A1 (en) | 2010-11-30 | 2013-10-03 | Srikant Pathak | Sensing Patch Applications |
| CN103347561B (zh) | 2010-12-08 | 2016-09-07 | 康沃特克科技公司 | 用于评估伤口分泌液的集成系统 |
| US20120165717A1 (en) | 2010-12-22 | 2012-06-28 | Convatec Technologies Inc. | Medical compression product, system utilizing such product, and program for use therewith |
| AU2012207287B2 (en) | 2011-01-19 | 2015-12-17 | The Regents Of The University Of California | Apparatus, systems, and methods for tissue oximetry and perfusion imaging |
| GB2487758A (en) | 2011-02-03 | 2012-08-08 | Isansys Lifecare Ltd | Health monitoring electrode assembly |
| WO2012109661A2 (en) | 2011-02-13 | 2012-08-16 | Cas Medical Systems, Inc. | Nirs sensor assembly including electrically conductive and optically transparent emi shielding |
| EP2683291B1 (en) | 2011-03-11 | 2019-07-31 | Proteus Digital Health, Inc. | Wearable personal body associated device with various physical configurations |
| US8818478B2 (en) | 2011-03-31 | 2014-08-26 | Adidas Ag | Sensor garment |
| US8838228B2 (en) | 2011-04-15 | 2014-09-16 | Arthur Beisang, III | Systems and methods for reducing the proliferation of microorganisms |
| US20120271265A1 (en) | 2011-04-20 | 2012-10-25 | Frederick Michael Langdon | Zero-Strain Stretch Laminate with Enhanced Strength, Appearance and Tactile Features, and Absorbent Articles Having Components Formed Therefrom |
| KR20120119523A (ko) | 2011-04-21 | 2012-10-31 | 방부복 | 혈관 위치 탐지용 장치 |
| US10117705B2 (en) | 2011-05-16 | 2018-11-06 | Covidien Lp | Optical recognition of tissue and vessels |
| WO2012167096A2 (en) | 2011-06-03 | 2012-12-06 | The Board Of Trustees Of The University Of Illinois | Conformable actively multiplexed high-density surface electrode array for brain interfacing |
| US20120316518A1 (en) * | 2011-06-09 | 2012-12-13 | Pierre Croizat | Wound dressing, containing nonwoven fabric and ointment base for negative pressure therapy |
| WO2012178161A1 (en) | 2011-06-24 | 2012-12-27 | Kci Licensing, Inc. | Medical drapes, devices, and systems employing a holographically-formed polymer dispersed liquid crystal (h-pdlc) device |
| CN105963074B (zh) | 2011-07-14 | 2020-01-17 | 史密夫及内修公开有限公司 | 伤口敷料和治疗方法 |
| JP5450527B2 (ja) | 2011-08-10 | 2014-03-26 | 富士フイルム株式会社 | 内視鏡装置 |
| WO2013023071A1 (en) | 2011-08-11 | 2013-02-14 | Reflectance Medical, Inc. | Patient interface for reusable optical sensor |
| WO2013026999A1 (en) | 2011-08-19 | 2013-02-28 | Pulse Innovate Ltd | A wound management system |
| EP2745771B1 (en) | 2011-08-19 | 2020-11-25 | Murata Manufacturing Co., Ltd. | Living organism sensor |
| WO2013028785A1 (en) | 2011-08-22 | 2013-02-28 | Ceramatec, Inc. | Osmotic wound vacuum system |
| US9023002B2 (en) | 2011-08-31 | 2015-05-05 | Timothy Mark Robinson | Reduced-pressure interfaces, systems, and methods employing a Coanda device |
| WO2013033724A1 (en) | 2011-09-01 | 2013-03-07 | Mc10, Inc. | Electronics for detection of a condition of tissue |
| EP2565630B1 (en) | 2011-09-02 | 2015-08-19 | CSEM Centre Suisse D'electronique Et De Microtechnique SA | Dye-doped gelatin-coated optical fibers for in situ monitoring of protease activity in wounds |
| JP2014526314A (ja) | 2011-09-08 | 2014-10-06 | インディケーター システムズ インターナショナル, インコーポレイテッド | 感染により活性化される創傷ケア組成物およびデバイス |
| WO2013044226A2 (en) | 2011-09-24 | 2013-03-28 | President And Fellows Of Harvard College | Artificial skin and elastic strain sensor |
| JP2014532178A (ja) | 2011-09-28 | 2014-12-04 | エムシー10 インコーポレイテッドMc10,Inc. | 表面の特性を検出するための電子機器 |
| WO2013066775A1 (en) | 2011-10-31 | 2013-05-10 | Smith & Nephew, Inc. | Apparatuses and methods for detecting leaks in a negative pressure wound therapy system |
| US9393354B2 (en) | 2011-11-01 | 2016-07-19 | J&M Shuler Medical, Inc. | Mechanical wound therapy for sub-atmospheric wound care system |
| AU2012335000B2 (en) | 2011-11-11 | 2017-10-05 | Kci Licensing, Inc. | Dressings and systems for treating a wound on a patients limb employing liquid control |
| WO2013086426A1 (en) | 2011-12-07 | 2013-06-13 | Kci Licensing, Inc. | Synthetic granulating gauze for use with reduced-pressure treatment systems |
| US9569566B2 (en) | 2011-12-12 | 2017-02-14 | Zam Research Llc | Simulation and control system and method using contact, pressure waves and factor controls for cell regeneration, tissue closure and related applications |
| US9603560B2 (en) | 2012-01-26 | 2017-03-28 | The University Of Akron | Flexible electrode for detecting changes in temperature, humidity, and sodium ion concentration in sweat |
| FR2986151B1 (fr) | 2012-01-30 | 2014-03-14 | Commissariat Energie Atomique | Pansement muni d'un systeme de detection. |
| ES2618728T3 (es) | 2012-01-30 | 2017-06-22 | Sensoria Inc. | Sensores, interfaces y sistemas de sensores para la recolección de data y la integración del monitoreo remoto de condiciones cercanas o sobre las superficies del cuerpo |
| WO2016109744A1 (en) | 2014-12-31 | 2016-07-07 | Sensoria Inc. | Sensors, interfaces and sensor systems for data collection and integrated monitoring of conditions at or near body surfaces |
| WO2013116013A1 (en) | 2012-02-02 | 2013-08-08 | The United States Government, As Represented By The Department Of Veterans Affairs | Integrated surface stimulation device for pain management and wound therapy |
| US9282897B2 (en) | 2012-02-13 | 2016-03-15 | MedHab, LLC | Belt-mounted movement sensor system |
| AU2013234034B2 (en) | 2012-03-12 | 2017-03-30 | Smith & Nephew Plc | Reduced pressure apparatus and methods |
| US8887313B2 (en) | 2012-03-30 | 2014-11-18 | Aaron McGuin | Wrap for human appendage |
| US9554484B2 (en) | 2012-03-30 | 2017-01-24 | The Board Of Trustees Of The University Of Illinois | Appendage mountable electronic devices conformable to surfaces |
| US10265219B2 (en) | 2012-04-12 | 2019-04-23 | Elwha Llc | Wound dressing monitoring systems including appurtenances for wound dressings |
| US9084530B2 (en) | 2012-04-12 | 2015-07-21 | Elwha Llc | Computational methods and systems for reporting information regarding appurtenances to wound dressings |
| US10158928B2 (en) | 2012-04-12 | 2018-12-18 | Elwha Llc | Appurtenances for reporting information regarding wound dressings |
| US10130518B2 (en) | 2012-04-12 | 2018-11-20 | Elwha Llc | Appurtenances including sensors for reporting information regarding wound dressings |
| US9332918B1 (en) | 2012-05-07 | 2016-05-10 | Jill Buckley | Patient monitoring devices and systems |
| CN104661601B (zh) | 2012-05-22 | 2018-06-22 | 史密夫及内修公开有限公司 | 用于伤口治疗的设备和方法 |
| EP3650055A1 (en) | 2012-05-23 | 2020-05-13 | Smith & Nephew plc | Apparatuses and methods for negative pressure wound therapy |
| US9226402B2 (en) | 2012-06-11 | 2015-12-29 | Mc10, Inc. | Strain isolation structures for stretchable electronics |
| US9582072B2 (en) | 2013-09-17 | 2017-02-28 | Medibotics Llc | Motion recognition clothing [TM] with flexible electromagnetic, light, or sonic energy pathways |
| DE102012211015A1 (de) * | 2012-06-27 | 2014-01-02 | Robert Bosch Gmbh | Wundverbandvorrichtung |
| EP2866851B1 (en) | 2012-06-28 | 2019-09-11 | KCI Licensing, Inc. | Wound connection pad with rfid and integrated strain gauge pressure sensor |
| CA2819803C (en) | 2012-07-06 | 2017-08-29 | Covidien Lp | Angiosome-based perfusion monitoring system |
| EP2872877B1 (en) | 2012-07-10 | 2023-05-24 | The General Hospital Corporation | System and method for monitoring and treating a surface of a subject |
| US20140018637A1 (en) | 2012-07-12 | 2014-01-16 | Oakwell - Cayman Company | Cloud-Based Monitoring of Medical Devices |
| RU2015106112A (ru) | 2012-08-01 | 2016-09-20 | СМИТ ЭНД НЕФЬЮ ПиЭлСи | Раневая повязка |
| CN104411236B (zh) | 2012-08-14 | 2017-05-24 | 泰尔茂株式会社 | 体内水分计 |
| KR20140024743A (ko) | 2012-08-21 | 2014-03-03 | 삼성전기주식회사 | 터치패널 제조용 지그 |
| KR101224629B1 (ko) | 2012-09-05 | 2013-01-22 | 주식회사 뉴피아 | 광을 이용한 피부 패치 |
| AT513325B1 (de) * | 2012-09-06 | 2014-09-15 | Ima Integrated Microsystems Austria Gmbh | Verfahren und Vorrichtung zur Überwachung der Wundheilung |
| US9811901B2 (en) | 2012-09-07 | 2017-11-07 | Massachusetts Institute Of Technology | Linear-based Eulerian motion modulation |
| US8948839B1 (en) | 2013-08-06 | 2015-02-03 | L.I.F.E. Corporation S.A. | Compression garments having stretchable and conductive ink |
| US8997588B2 (en) | 2012-09-29 | 2015-04-07 | Stryker Corporation | Force detecting mat with multiple sensor types |
| US20140107495A1 (en) | 2012-10-17 | 2014-04-17 | Nokia Corporation | Wearable Apparatus and Associated Methods |
| US20140107498A1 (en) | 2012-10-17 | 2014-04-17 | Nokia Corporation | Wearable Apparatus and Associated Methods |
| US9797791B2 (en) | 2012-10-27 | 2017-10-24 | President And Fellows Of Harvard College | Multi-axis force sensing soft artificial skin |
| KR101402925B1 (ko) | 2012-11-06 | 2014-06-02 | 주식회사 오라컴 | 플렉시블 회로기판 진공 흡착 지그 장치 |
| US9968488B2 (en) | 2012-11-12 | 2018-05-15 | Kci Usa, Inc. | Externally-applied patient interface system and method |
| US20150265191A1 (en) | 2012-11-16 | 2015-09-24 | Philip Harding | Electrochemical determination of infection |
| US20140147611A1 (en) | 2012-11-23 | 2014-05-29 | Sure Flash Llc | Conformable Preconditioned Adhesive Sealing Tape |
| PL2941195T3 (pl) | 2013-01-02 | 2017-06-30 | Fibrotx Oü | Urządzenie do pomiaru analitów w skórze |
| JP2016506281A (ja) | 2013-01-08 | 2016-03-03 | エムシー10 インコーポレイテッドMc10,Inc. | 表面の特性のモニタリングの応用 |
| GB201309369D0 (en) | 2013-05-24 | 2013-07-10 | Smith & Nephew | Moisture indicating system |
| GB201300470D0 (en) | 2013-01-11 | 2013-02-27 | Smith & Nephew | Moisture indicator dressing |
| GB201317746D0 (en) | 2013-10-08 | 2013-11-20 | Smith & Nephew | PH indicator |
| GB201401112D0 (en) | 2014-01-23 | 2014-03-12 | Smith & Nephew | Systems and methods for wound monitoring |
| WO2014113770A1 (en) * | 2013-01-21 | 2014-07-24 | Cornell University | Smartphone-based apparatus and method for obtaining repeatable, quantitative colorimetric measurement |
| US10583037B2 (en) | 2013-01-23 | 2020-03-10 | Transqtronics, Llc. | Heating device using exothermic chemical reaction |
| CA2900138A1 (en) | 2013-02-01 | 2014-08-07 | Daniel L. Farkas | Method and system for characterizing tissue in three dimensions using multimode optical measurements |
| US9613911B2 (en) | 2013-02-06 | 2017-04-04 | The Board Of Trustees Of The University Of Illinois | Self-similar and fractal design for stretchable electronics |
| US9026053B2 (en) | 2013-02-17 | 2015-05-05 | Fitbit, Inc. | System and method for wireless device pairing |
| US20140243709A1 (en) | 2013-02-28 | 2014-08-28 | Hill-Rom Services, Inc. | Pressure Sensing Pad, Method of Making the Same, Pressure Sensing System, and Pressure Map Display |
| US9042075B2 (en) | 2013-03-04 | 2015-05-26 | Nokia Technologies Oy | Apparatus and method for water protection of an electronic device |
| US20160022223A1 (en) | 2013-03-13 | 2016-01-28 | The Regents Of The University Of California | Multi-modal depth-resolved tissue status monitor |
| US11433254B2 (en) | 2013-03-15 | 2022-09-06 | Pavel V. Efremkin | Apparatus and method for treatment of wounds and skin medical conditions at a predetermined skin area |
| US9398880B2 (en) | 2013-03-20 | 2016-07-26 | Kelly Annette Vanscoy Barnett | Plurality of lamination for soft tissue compression support, protection and bracing; intelligent textile for equine and equestrian sports or activities |
| US20140296749A1 (en) | 2013-03-26 | 2014-10-02 | Lawrence G. Reid, Jr. | Body Monitoring System and Method |
| US9494474B2 (en) | 2013-04-03 | 2016-11-15 | Texavie Technologies Inc. | Core-shell nanofiber textiles for strain sensing, and methods of their manufacture |
| US20160074234A1 (en) | 2013-04-16 | 2016-03-17 | Drexel University | Radial compression utilizing a shape-memory alloy |
| MX2015015153A (es) | 2013-05-02 | 2016-08-11 | Vomaris Innovations Inc | Metodos y dispositivos para activacion celular. |
| US9706647B2 (en) | 2013-05-14 | 2017-07-11 | Mc10, Inc. | Conformal electronics including nested serpentine interconnects |
| US10314506B2 (en) | 2013-05-15 | 2019-06-11 | Polar Electro Oy | Heart activity sensor structure |
| US10166402B2 (en) | 2013-05-16 | 2019-01-01 | Excelitas Technologies Corp. | Visible light photo-disinfection patch |
| JP6420321B2 (ja) | 2013-05-21 | 2018-11-07 | オーピクス・メディカル・テクノロジーズ・インコーポレイテッド | 圧力データ取得アセンブリ |
| US10206604B2 (en) | 2013-05-23 | 2019-02-19 | Cutosense Oy | Arrangement for facilitating wound healing, a method for measuring wound healing and a wound dressing |
| US10166387B2 (en) | 2013-05-23 | 2019-01-01 | Cutosense Oy | Arrangement for facilitating wound healing, a method for measuring wound healing and a wound dressing |
| WO2014190534A1 (zh) | 2013-05-31 | 2014-12-04 | 东莞宇龙通信科技有限公司 | 移动终端、可穿戴设备和设备配对方法 |
| US20150335288A1 (en) | 2013-06-06 | 2015-11-26 | Tricord Holdings, Llc | Modular physiologic monitoring systems, kits, and methods |
| US10182757B2 (en) | 2013-07-22 | 2019-01-22 | The Rockefeller University | System and method for optical detection of skin disease |
| EP3030873A4 (en) | 2013-08-05 | 2017-07-05 | Mc10, Inc. | Flexible temperature sensor including conformable electronics |
| DE102013013013A1 (de) | 2013-08-06 | 2015-02-12 | Daniel Scharfen | Vorrichtung und Verfahren eines Signalisierungskanals basierend auf Vibrationsübertragung zur Paarung drahtloser Geräte |
| EP3052017B1 (en) | 2013-10-02 | 2019-12-11 | The Board of Trustees of the University of Illionis | Organ mounted electronics |
| JP2016532468A (ja) | 2013-10-07 | 2016-10-20 | エムシー10 インコーポレイテッドMc10,Inc. | 検知および分析のためのコンフォーマルセンサシステム |
| GB2519987B (en) | 2013-11-04 | 2021-03-03 | Imperial College Innovations Ltd | Biomechanical activity monitoring |
| US9418279B2 (en) | 2013-11-19 | 2016-08-16 | Qualcomm Incorporated | Detection of an object's varying features with a non-stationary device |
| KR102365120B1 (ko) | 2013-11-22 | 2022-02-18 | 메디데이타 솔루션즈, 인코포레이티드 | 심장 활동 감지 및 분석용 등각 센서 시스템 |
| US9192531B2 (en) | 2013-11-27 | 2015-11-24 | Chuan-Shih Wu | Intelligent sensing device with warning function |
| WO2015085240A1 (en) | 2013-12-05 | 2015-06-11 | Veriskin Llc | Skin perfusion monitoring device |
| US20160302729A1 (en) | 2013-12-11 | 2016-10-20 | The Board Of Regents Of The University Of Texas System | Devices and methods for parameter measurement |
| US9867571B2 (en) | 2014-01-06 | 2018-01-16 | Interaxon Inc. | Wearable apparatus for brain sensors |
| WO2015112095A1 (en) | 2014-01-23 | 2015-07-30 | Agency For Science, Technology And Research | Smart belt for breathing and heart rate monitoring |
| US9301691B2 (en) | 2014-02-21 | 2016-04-05 | Covidien Lp | Instrument for optically detecting tissue attributes |
| KR20160127021A (ko) | 2014-02-27 | 2016-11-02 | 쓰리엠 이노베이티브 프로퍼티즈 캄파니 | 유연한 센서 패치 및 이를 이용하는 방법 |
| US9706922B2 (en) | 2014-03-12 | 2017-07-18 | Zansors Llc | Wireless ECG acquisition and monitoring device and system |
| AT515656B1 (de) | 2014-03-17 | 2016-01-15 | Ait Austrian Inst Technology | Vorrichtung für die Ermittlung des Zustandes der Haut einer Person |
| US9526427B2 (en) | 2014-03-21 | 2016-12-27 | Hypermed Imaging, Inc. | Compact light sensors with symmetrical lighting |
| CA2945011A1 (en) | 2014-04-07 | 2015-10-15 | North Carolina State University | Electrodes and sensors having nanowires |
| US10973462B2 (en) | 2014-05-04 | 2021-04-13 | Scott J. Rapp | Fiber optic based devices and methods for monitoring soft tissue |
| US9402988B2 (en) | 2014-05-06 | 2016-08-02 | West Affum Holdings Corp. | Wearable medical system with stretch-cable assembly |
| WO2015168720A1 (en) | 2014-05-07 | 2015-11-12 | University Of South Australia | Wound sensor, system and method |
| US20150327777A1 (en) | 2014-05-14 | 2015-11-19 | Stryker Corporation | Tissue monitoring apparatus and system |
| US20170086519A1 (en) | 2014-05-15 | 2017-03-30 | Sensoria, Inc. | Gloves with sensors for monitoring and analysis of position, pressure and movement |
| US11207002B2 (en) | 2014-05-19 | 2021-12-28 | The Regents Of The University Of California | Fetal health monitor |
| US9592007B2 (en) | 2014-05-23 | 2017-03-14 | Samsung Electronics Co., Ltd. | Adjustable wearable system having a modular sensor platform |
| US11026847B2 (en) | 2014-06-02 | 2021-06-08 | Zdzislaw Harry Piotrowski | Systems and methods for wound healing |
| WO2015195720A1 (en) | 2014-06-16 | 2015-12-23 | The Regents Of The University Of California | Methods and apparatus for monitoring wound healing using impedance spectroscopy |
| US20150359485A1 (en) | 2014-06-17 | 2015-12-17 | MAD Apparel, Inc. | Biometric signal conduction system and method of manufacture |
| CA3177264A1 (en) | 2014-06-18 | 2015-12-23 | Innopix, Inc. | Spectral imaging system for remote and noninvasive detection of target substances using spectral filter arrays and image capture arrays |
| US10383550B2 (en) | 2014-07-17 | 2019-08-20 | Elwha Llc | Monitoring body movement or condition according to motion regimen with conformal electronics |
| US10152789B2 (en) | 2014-07-25 | 2018-12-11 | Covidien Lp | Augmented surgical reality environment |
| US10758133B2 (en) | 2014-08-07 | 2020-09-01 | Apple Inc. | Motion artifact removal by time domain projection |
| US20160038083A1 (en) | 2014-08-08 | 2016-02-11 | Orn, Inc. | Garment including integrated sensor components and feedback components |
| EP3179902B1 (en) | 2014-08-11 | 2020-10-14 | The Board of Trustees of the University of Illionis | Epidermal device for analysis of temperature and thermal transport characteristics |
| US20160051147A1 (en) | 2014-08-21 | 2016-02-25 | Irmed | System and method for noninvasive analysis of subcutaneous tissue |
| EP3185757B1 (en) | 2014-08-25 | 2023-06-07 | Bainisha CVBA | Elastic sensor |
| US11219413B2 (en) | 2014-08-26 | 2022-01-11 | Dexcom, Inc. | Systems and methods for securing a continuous analyte sensor to a host |
| US9993203B2 (en) | 2014-09-05 | 2018-06-12 | VivaLnk, Inc. | Electronic stickers with modular structures |
| US9572507B2 (en) | 2014-09-10 | 2017-02-21 | Dymedix Corporation | Combination physiologic sensor |
| US10258267B2 (en) | 2014-09-22 | 2019-04-16 | Capsule Technologies, Inc. | Pulse oximeter with an accelerometer |
| WO2016057633A1 (en) | 2014-10-08 | 2016-04-14 | Revealix, Inc. | Automated systems and methods for skin assessment and early detection of a latent pathogenic bio-signal anomaly |
| US20170311874A1 (en) | 2014-10-17 | 2017-11-02 | Andmine Pty Ltd. | Improvements to positional feedback devices |
| US20160317057A1 (en) | 2014-10-22 | 2016-11-03 | VivaLnk, Inc. | Compliant wearable patch capable of measuring electrical signals |
| EP3240471A4 (en) | 2014-11-05 | 2018-10-03 | The Regents of the University of California | Telemedical wearable sensing system for management of chronic venous disorders |
| US10245614B2 (en) | 2014-11-11 | 2019-04-02 | The Regents Of The University Of California | Imprinter for conformal coating of three-dimensional surfaces |
| US10512420B2 (en) | 2014-11-21 | 2019-12-24 | Elwha Llc | Systems to monitor body portions for injury after impact |
| WO2016083483A1 (en) | 2014-11-27 | 2016-06-02 | Koninklijke Philips N.V. | Imaging device and method for generating an image of a patient |
| US9380698B1 (en) | 2014-12-05 | 2016-06-28 | VivaLnk, Inc. | Stretchable electronic patch having a foldable circuit layer |
| US9378450B1 (en) | 2014-12-05 | 2016-06-28 | Vivalnk, Inc | Stretchable electronic patch having a circuit layer undulating in the thickness direction |
| US20160157779A1 (en) | 2014-12-08 | 2016-06-09 | Intel Corporation | Wearable sensor apparatus with multiple flexible substrates |
| US9483726B2 (en) | 2014-12-10 | 2016-11-01 | VivaLnk Inc. | Three dimensional electronic patch |
| KR101709344B1 (ko) | 2014-12-11 | 2017-03-08 | 한국과학기술원 | 종이기판 및 그 제조방법과 종이기판을 이용하는 센서 및 그 제조방법 |
| EP3235353B1 (en) | 2014-12-15 | 2022-09-21 | Robert Bosch GmbH | Modular deformable platform |
| EP3034054B1 (en) | 2014-12-16 | 2021-01-20 | Absorbest AB | Wound dressing with a sensor and method for manufacturing the same |
| JP2018510035A (ja) | 2014-12-29 | 2018-04-12 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | 陰圧閉鎖治療器及び器具を動作させる方法 |
| US10695555B2 (en) | 2015-01-08 | 2020-06-30 | Ecole Polytechnique Federale De Lausanne (Epfl) | Synthetic skin for recording and modulating physiological activities |
| AU2016200113B2 (en) | 2015-01-21 | 2019-10-31 | Covidien Lp | Wirelessly detectable objects for use in medical procedures and methods of making same |
| US10039466B2 (en) | 2015-01-28 | 2018-08-07 | City University Of Hong Kong | Apparatus for detection of electrical signals of a biological subject and electrode thereof, and method of manufacture thereof |
| US20160228049A1 (en) * | 2015-02-06 | 2016-08-11 | Nxp B.V. | Wound monitoring |
| KR102350499B1 (ko) | 2015-02-17 | 2022-01-14 | 삼성전자주식회사 | 전자장치용 전자기 쉴드 구조 |
| US10022073B2 (en) | 2015-03-20 | 2018-07-17 | Intel Corproation | Wearable apparatus with a stretch sensor |
| US10485481B2 (en) | 2015-03-20 | 2019-11-26 | The Trustees Of Dartmouth College | Systems and methods for enhancing uptake of therapeutic agent from bloodstream into disease site |
| WO2016154507A1 (en) | 2015-03-25 | 2016-09-29 | Son Jae S | Apparatuses, devices, and methods for measuring fluid pressure variation in an insole |
| EP3283029A1 (en) | 2015-04-15 | 2018-02-21 | King Abdullah University Of Science And Technology | Wound dressing with reusable electronics for wireless monitoring |
| US10182740B2 (en) | 2015-04-24 | 2019-01-22 | Bruin Biometrics, Llc | Apparatus and methods for determining damaged tissue using sub-epidermal moisture measurements |
| DK3288508T3 (da) | 2015-04-27 | 2020-03-09 | Smith & Nephew | Anordninger med reduceret tryk |
| KR101572119B1 (ko) | 2015-04-30 | 2015-11-26 | 주식회사 오라컴 | 플렉시블 피씨비 장착용 진공 지그 시스템 |
| US20160331263A1 (en) | 2015-05-13 | 2016-11-17 | Ep Solutions Sa | Customizable Electrophysiological Mapping Electrode Patch Systems, Devices, Components and Methods |
| WO2016187136A1 (en) | 2015-05-15 | 2016-11-24 | Veriskin, Inc. | Cutaneous blood flow monitoring device |
| US10076594B2 (en) | 2015-05-18 | 2018-09-18 | Smith & Nephew Plc | Fluidic connector for negative pressure wound therapy |
| US20160338591A1 (en) | 2015-05-21 | 2016-11-24 | Hill-Rom Services, Inc. | Systems and methods for mitigating tissue breakdown |
| US10835181B2 (en) | 2015-06-16 | 2020-11-17 | Fossil Group, Inc. | Apparatuses, methods, and systems for measuring insole deformation |
| WO2016205881A1 (en) | 2015-06-23 | 2016-12-29 | Ti2 Medical Pty Ltd | Anisotropically conductive material for use with a biological surface |
| AU2016282079A1 (en) | 2015-06-26 | 2017-12-14 | Impedimed Limited | Impedance methods and apparatuses using arrays of bipolar electrodes |
| US20170007853A1 (en) | 2015-07-10 | 2017-01-12 | Medtronic, Inc. | Physiological monitoring for ultrasound therapy |
| EP3117807A1 (en) | 2015-07-16 | 2017-01-18 | Carag AG | Multifunctional wound treatment dressing |
| DE102015010189A1 (de) | 2015-08-04 | 2017-02-09 | Infineon Technologies Ag | Körperparameterüberwachungsvorrichtung |
| WO2017021965A2 (en) | 2015-08-06 | 2017-02-09 | Upright Technologies Ltd. | Body movement feedback system and method |
| CN107926117B (zh) | 2015-08-21 | 2020-08-14 | 阿莫绿色技术有限公司 | 可穿戴柔性印刷电路板、其制造方法及利用其的可穿戴智能装置 |
| WO2017033058A1 (en) | 2015-08-24 | 2017-03-02 | L.I.F.E. Corporation S.A. | Physiological monitoring garments with enhanced sensor stabilization |
| MX2018001293A (es) | 2015-08-26 | 2018-05-17 | Kimberly Clark Co | Dispositivos portatiles para tomografia por induccion magnetica. |
| WO2017041014A1 (en) | 2015-09-02 | 2017-03-09 | The General Hospital Corporation | Electroencephalogram monitoring system and method of use of the same |
| CN108601524A (zh) | 2015-09-04 | 2018-09-28 | 福法斯哈比斯英国有限公司 | 组合的织物压力和光学传感器 |
| CN105250075A (zh) | 2015-09-12 | 2016-01-20 | 深圳市前海安测信息技术有限公司 | 伤口愈合程度监测系统及方法 |
| CN105105716A (zh) | 2015-09-12 | 2015-12-02 | 深圳市前海安测信息技术有限公司 | 用于检测伤口愈合程度的智能传感器及其制备方法 |
| KR102556007B1 (ko) | 2015-10-07 | 2023-07-17 | 삼성전자주식회사 | 생체신호 측정장치 및 방법 |
| CN105232229B (zh) | 2015-10-19 | 2018-07-24 | 中国人民解放军第四军医大学 | 一种可实时监测创面愈合的智能无线传感敷料 |
| US9484209B1 (en) | 2015-11-20 | 2016-11-01 | International Business Machines Corporation | Flexible and stretchable sensors formed by patterned spalling |
| CN105395184B (zh) | 2015-12-04 | 2018-05-08 | 华中科技大学 | 生物组织血流、血氧和血容量的多参数检测方法及装置 |
| US20170156594A1 (en) | 2015-12-07 | 2017-06-08 | Bodymedia, Inc. | Systems, methods, and devices to determine and predict physilogical states of individuals and to administer therapy, reports, notifications, and the like therefor |
| US11478178B2 (en) | 2015-12-08 | 2022-10-25 | Carnegie Mellon University | Electronic structures on swollen hydrogels |
| US11141100B2 (en) | 2015-12-23 | 2021-10-12 | Coloplast A/S | Moisture assessment system and method for wound care |
| EP3400032B1 (en) | 2016-01-06 | 2020-03-18 | KCI Licensing, Inc. | System for the treatment of wounds with dressing having closed cells |
| US20170202711A1 (en) | 2016-01-19 | 2017-07-20 | Andrei Cernasov | Wound treatment system and method |
| EP3208700B1 (en) | 2016-02-05 | 2020-04-01 | Lg Electronics Inc. | Iot device, mobile terminal and method for controlling the iot device with vibration pairing |
| US10426396B2 (en) | 2016-02-10 | 2019-10-01 | Hill-Rom Services, Inc. | Pressure ulcer detection systems and methods |
| CN114053031A (zh) | 2016-03-07 | 2022-02-18 | 史密夫及内修公开有限公司 | 利用整合到伤口敷料中的负压源的伤口治疗设备和方法 |
| WO2017158428A1 (en) | 2016-03-14 | 2017-09-21 | Smith & Nephew Plc | Wound dressing apparatus with flexible display |
| EP3231478A1 (en) | 2016-04-13 | 2017-10-18 | Oncotherm Kft. | Radiofrequency hyperthermia device with double impedance matching system |
| CN114469523A (zh) | 2016-04-26 | 2022-05-13 | 史密夫及内修公开有限公司 | 与具有流体侵入抑制部件的一体化负压源一起使用的伤口敷料和方法 |
| EP3454809B8 (en) | 2016-05-10 | 2020-12-30 | 3M Innovative Properties Company | Flexible means for determining the extent of debridement required to remove non-viable tissue |
| JP2019527566A (ja) | 2016-05-13 | 2019-10-03 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | センサが使用可能な創傷監視および治療装置 |
| WO2017205728A1 (en) | 2016-05-26 | 2017-11-30 | Wound Care And Rehab Medicine Llc | Pressure and vacuum sensors, systems, and associated methods |
| WO2017214188A1 (en) | 2016-06-06 | 2017-12-14 | University Of Massachusetts | Systems and methods for prevention of pressure ulcers |
| US20170367644A1 (en) | 2016-06-27 | 2017-12-28 | Claris Healthcare Inc. | Apparatus and Method for Monitoring Rehabilitation from Joint Surgery |
| US11229553B2 (en) | 2016-08-16 | 2022-01-25 | Synaptive Medical Inc. | Dressing apparatus and methods for facilitating healing |
| WO2018035612A1 (en) | 2016-08-24 | 2018-03-01 | Mimosa Diagnostics Inc. | Multispectral mobile tissue assessment |
| US20180056087A1 (en) | 2016-08-26 | 2018-03-01 | Adolfo Ribeiro | Wearable Micro-LED Healing Bandage |
| US10426672B2 (en) | 2016-08-26 | 2019-10-01 | Vener8 Technologies | Moisture detection and notification system |
| US10122184B2 (en) | 2016-09-15 | 2018-11-06 | Blackberry Limited | Application of modulated vibrations in docking scenarios |
| AU2017335637B2 (en) | 2016-09-29 | 2023-01-19 | Smith & Nephew Plc | Protection of electronics in negative pressure wound therapy systems |
| WO2018064569A1 (en) | 2016-09-30 | 2018-04-05 | The Regents Of The University Of California | Multi-modal depth-resolved tissue status and contact pressure monitor |
| EP3519001A1 (en) | 2016-09-30 | 2019-08-07 | Smith & Nephew PLC | Negative pressure wound treatment apparatuses and methods with integrated electronics |
| TWI647967B (zh) | 2016-11-04 | 2019-01-11 | 宏達國際電子股份有限公司 | 藉由振動建立無線連接的方法、電子裝置和記錄媒體 |
| EP3558184A1 (en) | 2016-12-22 | 2019-10-30 | Fleming Medical Ltd. | A dressing system |
| KR20230003447A (ko) | 2017-02-03 | 2023-01-05 | 브루인 바이오메트릭스, 엘엘씨 | 당뇨병성 족부 궤양에 대한 감수성의 측정 |
| US11304652B2 (en) | 2017-02-03 | 2022-04-19 | Bbi Medical Innovations, Llc | Measurement of tissue viability |
| AU2018217182B2 (en) | 2017-02-03 | 2019-12-12 | Bruin Biometrics, Llc | Bisymmetric comparison of sub-epidermal moisture values |
| KR20220074988A (ko) | 2017-02-03 | 2022-06-03 | 브루인 바이오메트릭스, 엘엘씨 | 부종의 측정 |
| US10638944B2 (en) | 2017-02-22 | 2020-05-05 | Covidien Lp | Methods of determining tissue viability |
| US11324424B2 (en) | 2017-03-09 | 2022-05-10 | Smith & Nephew Plc | Apparatus and method for imaging blood in a target region of tissue |
| WO2018162728A2 (en) | 2017-03-09 | 2018-09-13 | Smith & Nephew Plc | Device, apparatus and method of determining skin perfusion pressure |
| GB201703769D0 (en) | 2017-03-09 | 2017-04-26 | Smith & Nephew | Imaging apparatus and method of imaging |
| WO2018162736A1 (en) | 2017-03-09 | 2018-09-13 | Smith & Nephew Plc | Wound dressing, patch member and method of sensing one or more wound parameters |
| RU2745731C1 (ru) | 2017-04-04 | 2021-03-31 | Ф. Хоффманн-Ля Рош Аг | Носимое на теле медицинское устройство |
| GB201800057D0 (en) | 2018-01-03 | 2018-02-14 | Smith & Nephew Inc | Component Positioning And stress Relief For Sensor Enabled Wound Dressings |
| WO2018209090A1 (en) | 2017-05-11 | 2018-11-15 | Kent State University | Microcirculation assessment device |
| US20200078499A1 (en) | 2017-05-15 | 2020-03-12 | Smith & Nephew Plc | Negative pressure wound therapy system using eulerian video magnification |
| CN110832598B (zh) | 2017-05-15 | 2024-03-15 | 史密夫及内修公开有限公司 | 伤口分析装置和方法 |
| US20200188180A1 (en) * | 2017-05-17 | 2020-06-18 | Uvic Industry Partnerships Inc. | Wound covering for wound monitoring and therapeutic agent delivery |
| EP3409190A1 (en) | 2017-05-31 | 2018-12-05 | CutoSense Oy | Measuring wound healing |
| GB2563602B (en) | 2017-06-19 | 2022-06-08 | Middlesex Univ Higher Education Corporation | Method and apparatus for imaging |
| AU2018288530B2 (en) | 2017-06-23 | 2024-03-28 | Smith & Nephew Plc | Positioning of sensors for sensor enabled wound monitoring or therapy |
| GB201809007D0 (en) | 2018-06-01 | 2018-07-18 | Smith & Nephew | Restriction of sensor-monitored region for sensor-enabled wound dressings |
| GB201803496D0 (en) | 2018-03-05 | 2018-04-18 | Smith & Nephew | Skewing pads for impedance measurement |
| GB201804502D0 (en) | 2018-03-21 | 2018-05-02 | Smith & Nephew | Biocompatible encapsulation and component stress relief for sensor enabled negative pressure wound therapy dressings |
| US11925735B2 (en) | 2017-08-10 | 2024-03-12 | Smith & Nephew Plc | Positioning of sensors for sensor enabled wound monitoring or therapy |
| US20190060126A1 (en) | 2017-08-30 | 2019-02-28 | Hill-Rom Services, Inc. | Systems for monitoring wounds and wound dressing status and systems for protecting wounds |
| JP2020533093A (ja) | 2017-09-10 | 2020-11-19 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | 封入を検査するためのシステムおよび方法、ならびにセンサを装備した創傷被覆材内の構成要素 |
| GB201804971D0 (en) | 2018-03-28 | 2018-05-09 | Smith & Nephew | Electrostatic discharge protection for sensors in wound therapy |
| GB201718870D0 (en) | 2017-11-15 | 2017-12-27 | Smith & Nephew Inc | Sensor enabled wound therapy dressings and systems |
| US11160491B2 (en) | 2017-09-12 | 2021-11-02 | Hill-Rom Services, Inc. | Devices, systems, and methods for monitoring wounds |
| GB201718859D0 (en) | 2017-11-15 | 2017-12-27 | Smith & Nephew | Sensor positioning for sensor enabled wound therapy dressings and systems |
| US11596553B2 (en) | 2017-09-27 | 2023-03-07 | Smith & Nephew Plc | Ph sensing for sensor enabled negative pressure wound monitoring and therapy apparatuses |
| EP3687396A1 (en) | 2017-09-28 | 2020-08-05 | Smith & Nephew plc | Neurostimulation and monitoring using sensor enabled wound monitoring and therapy apparatus |
| US11266537B2 (en) | 2017-09-29 | 2022-03-08 | Kci Licensing, Inc. | Dressing exhibiting low tissue ingrowth and negative-pressure treatment method |
| GB201718851D0 (en) | 2017-11-15 | 2017-12-27 | Smith & Nephew | Flocked conformable circuit boards for sensor enabled wound therapy dressings and systems |
| CA3079131A1 (en) | 2017-11-09 | 2019-05-16 | 11 Health and Technologies Inc. | Ostomy monitoring system and method |
| JP2021502845A (ja) | 2017-11-15 | 2021-02-04 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | 統合センサ対応型創傷モニタリングおよび/または治療被覆材ならびにシステム |
| AU2018380431A1 (en) | 2017-12-07 | 2020-06-18 | Bruin Biometrics, Llc | SEM trend analysis |
| US10080524B1 (en) | 2017-12-08 | 2018-09-25 | VivaLnk, Inc. | Wearable thermometer patch comprising a temperature sensor array |
| EP3499510A1 (en) | 2017-12-14 | 2019-06-19 | Koninklijke Philips N.V. | System and method for monitoring wound healing |
| EP3727236A1 (en) | 2017-12-22 | 2020-10-28 | Coloplast A/S | Sensor assembly part and a base plate for an ostomy appliance and a method for manufacturing a sensor assembly part and a base plate |
| EP4238594A3 (en) | 2018-01-15 | 2023-11-08 | 3M Innovative Properties Company | Wound sensor and diagnostics system for wound therapy applications |
| US11511033B2 (en) | 2018-01-15 | 2022-11-29 | Kci Licensing, Inc. | Systems and methods for controlling negative pressure therapy with fluid instillation therapy |
| EP3740259A1 (en) | 2018-01-15 | 2020-11-25 | 3M Innovative Properties Company | Systems and methods for controlling negative pressure therapy using properties of fluids from a tissue site |
| EP3740258A1 (en) | 2018-01-15 | 2020-11-25 | 3M Innovative Properties Company | Systems and methods for sensing properties of wound exudates |
| KR102067501B1 (ko) | 2018-03-06 | 2020-01-17 | 에릭스바이오(주) | 광 펄스를 적용한 항균 드레싱 밴드 |
| GB201805584D0 (en) | 2018-04-05 | 2018-05-23 | Smith & Nephew | Negative pressure wound treatment apparatuses and methods with integrated electronics |
| CA3097113C (en) | 2018-05-08 | 2023-07-04 | Mark D. Edmundson | Flexible printed circuits for dermal applications |
| US20210205378A1 (en) | 2018-05-31 | 2021-07-08 | Hokukon Co., Ltd. | Lactic acid bacteria and use thereof |
| US11583449B2 (en) | 2018-06-06 | 2023-02-21 | Hill-Rom Services, Inc. | Systems for recognizing temperature as a sign of surgical infection and methods of using the same |
| US20210205138A1 (en) | 2018-06-15 | 2021-07-08 | Coloplast A/S | Wound dressing and method of manufacturing a wound dressing |
| US20210205140A1 (en) | 2018-06-15 | 2021-07-08 | Coloplast A/S | Accessory device of a wound dressing system, and related methods for communicating operating state |
| US11633304B2 (en) | 2018-06-15 | 2023-04-25 | Coloplast A/S | Wound dressing comprising a monitor interface |
| EP3806716B1 (en) | 2018-06-15 | 2023-06-07 | Coloplast A/S | Wound dressing system, monitor device and related method |
| CN112261923B (zh) | 2018-06-15 | 2022-09-06 | 科洛普拉斯特公司 | 用于伤口护理的水分评估系统和方法 |
| WO2019238181A1 (en) | 2018-06-15 | 2019-12-19 | Coloplast A/S | Moisture sensing wound dressing |
| US20210251565A1 (en) | 2018-06-15 | 2021-08-19 | Coloplast A/S | Data collection schemes for a wound dressing and related methods |
| US11938236B2 (en) | 2018-08-17 | 2024-03-26 | Seoul Viosys Co., Ltd. | Medical dressing |
| US11109765B2 (en) | 2018-08-20 | 2021-09-07 | VivaLnk, Inc. | Wearable thermometer patch comprising a temperature sensor array |
| GB201814011D0 (en) | 2018-08-29 | 2018-10-10 | Smith & Nephew | Componet positioning and encapsulation for sensor enabled wound dressings |
-
2018
- 2018-09-24 US US16/649,340 patent/US11596553B2/en active Active
- 2018-09-24 CN CN201880060704.4A patent/CN111132605B/zh active Active
- 2018-09-24 JP JP2020516581A patent/JP7282079B2/ja active Active
- 2018-09-24 EP EP18781990.9A patent/EP3687380A1/en active Pending
- 2018-09-24 WO PCT/EP2018/075802 patent/WO2019063481A1/en unknown
-
2023
- 2023-02-17 US US18/111,158 patent/US20230277381A1/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005179314A (ja) | 2003-12-24 | 2005-07-07 | Life Kea Giken Kk | 皮膚検査用貼付剤 |
| US20130217983A1 (en) | 2011-12-29 | 2013-08-22 | Wellsense, Inc. | Analyte sensor |
| JP2017500167A (ja) | 2013-10-08 | 2017-01-05 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | pHインジケータードレッシング |
| US20160015962A1 (en) | 2014-07-16 | 2016-01-21 | Mehdi Shokoueinejad Maragheh | Smart Patch For Wound Management |
| JP2017510319A5 (ja) | 2015-01-20 | 2018-02-01 | ||
| WO2017041386A1 (zh) | 2015-09-12 | 2017-03-16 | 深圳市前海颐老科技有限公司 | 伤口感染程度监测系统及方法 |
Non-Patent Citations (1)
| Title |
|---|
| PUCHBERGER-ENENGL, D., et al.,Organically modified silicate film pH sensor for continuous wound monitoring,SENSORS, 2011 IEEE,2011年10月28日,pp.679-682,<DOI: 10.1109/ICSENS.2011.6127220>, <Date of Conference: 28-31 October 2011>, <Date Added to IEEE Xplore: 12 January 2012> |
Also Published As
| Publication number | Publication date |
|---|---|
| US20230277381A1 (en) | 2023-09-07 |
| WO2019063481A1 (en) | 2019-04-04 |
| JP2020537553A (ja) | 2020-12-24 |
| CN111132605A (zh) | 2020-05-08 |
| EP3687380A1 (en) | 2020-08-05 |
| CN111132605B (zh) | 2023-05-16 |
| US11596553B2 (en) | 2023-03-07 |
| US20200289329A1 (en) | 2020-09-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7282079B2 (ja) | センサが使用可能な陰圧創傷監視および療法装置のph感知 | |
| JP7234200B2 (ja) | センサ対応創傷被覆材のセンサ監視領域の制限 | |
| JP7354098B2 (ja) | センサ対応型創傷療法被覆材およびシステムのためのセンサ位置決めおよび光学検知 | |
| EP3659409B1 (en) | Biocompatible encapsulation and component stress relief for sensor enabled negative pressure wound therapy dressings | |
| JP7286626B2 (ja) | センサ対応の創傷治療被覆材およびサイバーセキュリティを実施するシステム | |
| EP3641627B1 (en) | Positioning of sensors for sensor enabled wound monitoring or therapy | |
| CN111093726B (zh) | 实施传感器的伤口监测或治疗的传感器定位 | |
| US20210267806A1 (en) | Component positioning and encapsulation for sensor enabled wound dressings | |
| WO2019020551A1 (en) | ASYMMETRIC ELECTRODES FOR MEASURING IMPEDANCE | |
| US20210378876A1 (en) | Systems and method for applying biocompatible encapsulation to sensor enabled wound monitoring and therapy dressings |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20200420 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20200424 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210921 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20210921 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20220928 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20221107 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20230126 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20230417 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20230516 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7282079 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |