JP6946261B2 - Asicを用いない内部電子機器を有する埋め込み可能神経刺激装置および方法 - Google Patents
Asicを用いない内部電子機器を有する埋め込み可能神経刺激装置および方法 Download PDFInfo
- Publication number
- JP6946261B2 JP6946261B2 JP2018500525A JP2018500525A JP6946261B2 JP 6946261 B2 JP6946261 B2 JP 6946261B2 JP 2018500525 A JP2018500525 A JP 2018500525A JP 2018500525 A JP2018500525 A JP 2018500525A JP 6946261 B2 JP6946261 B2 JP 6946261B2
- Authority
- JP
- Japan
- Prior art keywords
- current
- differential amplifier
- conductor
- source
- voltage
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 210000005036 nerve Anatomy 0.000 title claims description 112
- 238000000034 method Methods 0.000 title claims description 89
- 239000004020 conductor Substances 0.000 claims description 163
- 230000000638 stimulation Effects 0.000 claims description 63
- WABPQHHGFIMREM-UHFFFAOYSA-N lead(0) Chemical compound [Pb] WABPQHHGFIMREM-UHFFFAOYSA-N 0.000 claims description 12
- 238000012384 transportation and delivery Methods 0.000 claims description 6
- 238000012358 sourcing Methods 0.000 claims description 4
- 230000007383 nerve stimulation Effects 0.000 description 46
- 238000011282 treatment Methods 0.000 description 43
- 230000001537 neural effect Effects 0.000 description 32
- 210000001519 tissue Anatomy 0.000 description 28
- 210000003205 muscle Anatomy 0.000 description 24
- 238000002567 electromyography Methods 0.000 description 22
- 230000027939 micturition Effects 0.000 description 22
- 230000008569 process Effects 0.000 description 20
- 230000004044 response Effects 0.000 description 19
- 230000004064 dysfunction Effects 0.000 description 16
- 238000002513 implantation Methods 0.000 description 16
- 238000012360 testing method Methods 0.000 description 14
- 239000000919 ceramic Substances 0.000 description 13
- 208000024891 symptom Diseases 0.000 description 13
- 238000002560 therapeutic procedure Methods 0.000 description 12
- 208000002193 Pain Diseases 0.000 description 11
- 230000001225 therapeutic effect Effects 0.000 description 11
- 238000004891 communication Methods 0.000 description 10
- 206010046555 Urinary retention Diseases 0.000 description 9
- 230000001953 sensory effect Effects 0.000 description 8
- 230000002485 urinary effect Effects 0.000 description 8
- 206010036018 Pollakiuria Diseases 0.000 description 7
- 238000013459 approach Methods 0.000 description 7
- 210000000056 organ Anatomy 0.000 description 7
- 230000036407 pain Effects 0.000 description 7
- 210000000278 spinal cord Anatomy 0.000 description 7
- 206010046543 Urinary incontinence Diseases 0.000 description 6
- 239000000853 adhesive Substances 0.000 description 6
- 230000001070 adhesive effect Effects 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 238000013461 design Methods 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 230000004936 stimulating effect Effects 0.000 description 6
- 230000008901 benefit Effects 0.000 description 5
- 230000001276 controlling effect Effects 0.000 description 5
- 238000004146 energy storage Methods 0.000 description 5
- 230000006698 induction Effects 0.000 description 5
- 208000034347 Faecal incontinence Diseases 0.000 description 4
- 238000003491 array Methods 0.000 description 4
- 230000001684 chronic effect Effects 0.000 description 4
- 230000008878 coupling Effects 0.000 description 4
- 238000010168 coupling process Methods 0.000 description 4
- 238000005859 coupling reaction Methods 0.000 description 4
- 238000002695 general anesthesia Methods 0.000 description 4
- 108010024001 incobotulinumtoxinA Proteins 0.000 description 4
- 238000002690 local anesthesia Methods 0.000 description 4
- 230000007774 longterm Effects 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 238000012544 monitoring process Methods 0.000 description 4
- 206010010774 Constipation Diseases 0.000 description 3
- 206010020853 Hypertonic bladder Diseases 0.000 description 3
- 206010021639 Incontinence Diseases 0.000 description 3
- 229940121948 Muscarinic receptor antagonist Drugs 0.000 description 3
- 208000009722 Overactive Urinary Bladder Diseases 0.000 description 3
- 108010057266 Type A Botulinum Toxins Proteins 0.000 description 3
- 230000004913 activation Effects 0.000 description 3
- 230000009286 beneficial effect Effects 0.000 description 3
- 230000033228 biological regulation Effects 0.000 description 3
- 229940089093 botox Drugs 0.000 description 3
- 238000005219 brazing Methods 0.000 description 3
- 239000000812 cholinergic antagonist Substances 0.000 description 3
- 230000013872 defecation Effects 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 230000001976 improved effect Effects 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 210000002414 leg Anatomy 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000003227 neuromodulating effect Effects 0.000 description 3
- 208000020629 overactive bladder Diseases 0.000 description 3
- 210000000578 peripheral nerve Anatomy 0.000 description 3
- 210000000954 sacrococcygeal region Anatomy 0.000 description 3
- 238000004088 simulation Methods 0.000 description 3
- 230000007704 transition Effects 0.000 description 3
- 229910000859 α-Fe Inorganic materials 0.000 description 3
- 208000000094 Chronic Pain Diseases 0.000 description 2
- 208000027534 Emotional disease Diseases 0.000 description 2
- 208000016285 Movement disease Diseases 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 210000001015 abdomen Anatomy 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 230000005540 biological transmission Effects 0.000 description 2
- RYYVLZVUVIJVGH-UHFFFAOYSA-N caffeine Chemical compound CN1C(=O)N(C)C(=O)C2=C1N=CN2C RYYVLZVUVIJVGH-UHFFFAOYSA-N 0.000 description 2
- 239000003990 capacitor Substances 0.000 description 2
- 210000004027 cell Anatomy 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 230000008030 elimination Effects 0.000 description 2
- 238000003379 elimination reaction Methods 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 230000004907 flux Effects 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 239000007943 implant Substances 0.000 description 2
- 230000001939 inductive effect Effects 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000009434 installation Methods 0.000 description 2
- 210000002988 lumbosacral plexus Anatomy 0.000 description 2
- 230000036453 micturition reflex Effects 0.000 description 2
- 230000000414 obstructive effect Effects 0.000 description 2
- 208000035824 paresthesia Diseases 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 230000011514 reflex Effects 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 230000008672 reprogramming Effects 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 210000002972 tibial nerve Anatomy 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 238000012549 training Methods 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N Alumina Chemical compound [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 206010069632 Bladder dysfunction Diseases 0.000 description 1
- 108030001720 Bontoxilysin Proteins 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 208000006561 Cluster Headache Diseases 0.000 description 1
- 208000023890 Complex Regional Pain Syndromes Diseases 0.000 description 1
- 208000013586 Complex regional pain syndrome type 1 Diseases 0.000 description 1
- 208000014094 Dystonic disease Diseases 0.000 description 1
- 206010017076 Fracture Diseases 0.000 description 1
- 229910000575 Ir alloy Inorganic materials 0.000 description 1
- LPHGQDQBBGAPDZ-UHFFFAOYSA-N Isocaffeine Natural products CN1C(=O)N(C)C(=O)C2=C1N(C)C=N2 LPHGQDQBBGAPDZ-UHFFFAOYSA-N 0.000 description 1
- 206010027566 Micturition urgency Diseases 0.000 description 1
- 208000019430 Motor disease Diseases 0.000 description 1
- 208000021384 Obsessive-Compulsive disease Diseases 0.000 description 1
- 206010033799 Paralysis Diseases 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- 229910001260 Pt alloy Inorganic materials 0.000 description 1
- 201000001947 Reflex Sympathetic Dystrophy Diseases 0.000 description 1
- 206010040021 Sensory abnormalities Diseases 0.000 description 1
- 208000027520 Somatoform disease Diseases 0.000 description 1
- 206010041349 Somnolence Diseases 0.000 description 1
- 229910001069 Ti alloy Inorganic materials 0.000 description 1
- 208000000323 Tourette Syndrome Diseases 0.000 description 1
- 208000016620 Tourette disease Diseases 0.000 description 1
- 206010066901 Treatment failure Diseases 0.000 description 1
- 206010044565 Tremor Diseases 0.000 description 1
- 208000032159 Vaginal inflammation Diseases 0.000 description 1
- 201000008100 Vaginitis Diseases 0.000 description 1
- 206010047513 Vision blurred Diseases 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 208000005298 acute pain Diseases 0.000 description 1
- 239000000048 adrenergic agonist Substances 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 238000004873 anchoring Methods 0.000 description 1
- 206010003074 arachnoiditis Diseases 0.000 description 1
- 230000003542 behavioural effect Effects 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- 239000000560 biocompatible material Substances 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 229940053031 botulinum toxin Drugs 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 210000000133 brain stem Anatomy 0.000 description 1
- 210000001217 buttock Anatomy 0.000 description 1
- 229960001948 caffeine Drugs 0.000 description 1
- VJEONQKOZGKCAK-UHFFFAOYSA-N caffeine Natural products CN1C(=O)N(C)C(=O)C2=C1C=CN2C VJEONQKOZGKCAK-UHFFFAOYSA-N 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 229910010293 ceramic material Inorganic materials 0.000 description 1
- 208000018912 cluster headache syndrome Diseases 0.000 description 1
- 230000000112 colonic effect Effects 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 230000010485 coping Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 206010013781 dry mouth Diseases 0.000 description 1
- 208000010118 dystonia Diseases 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 238000002594 fluoroscopy Methods 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 210000004392 genitalia Anatomy 0.000 description 1
- 239000012212 insulator Substances 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 210000005072 internal anal sphincter Anatomy 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000002085 irritant Substances 0.000 description 1
- 231100000021 irritant Toxicity 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 230000004807 localization Effects 0.000 description 1
- 230000033001 locomotion Effects 0.000 description 1
- 210000005230 lumbar spinal cord Anatomy 0.000 description 1
- 210000004705 lumbosacral region Anatomy 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 210000000663 muscle cell Anatomy 0.000 description 1
- 230000004118 muscle contraction Effects 0.000 description 1
- 210000004126 nerve fiber Anatomy 0.000 description 1
- 230000008904 neural response Effects 0.000 description 1
- 208000004296 neuralgia Diseases 0.000 description 1
- 230000002232 neuromuscular Effects 0.000 description 1
- 208000021722 neuropathic pain Diseases 0.000 description 1
- 206010029446 nocturia Diseases 0.000 description 1
- 238000011369 optimal treatment Methods 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 208000027753 pain disease Diseases 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 210000003903 pelvic floor Anatomy 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 208000033808 peripheral neuropathy Diseases 0.000 description 1
- 230000037081 physical activity Effects 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 238000012797 qualification Methods 0.000 description 1
- 238000004451 qualitative analysis Methods 0.000 description 1
- 230000007115 recruitment Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 230000035807 sensation Effects 0.000 description 1
- 206010040872 skin infection Diseases 0.000 description 1
- 210000002460 smooth muscle Anatomy 0.000 description 1
- 230000000392 somatic effect Effects 0.000 description 1
- 210000005070 sphincter Anatomy 0.000 description 1
- 210000001032 spinal nerve Anatomy 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 229910002082 tetragonal zirconia polycrystal Inorganic materials 0.000 description 1
- 210000000115 thoracic cavity Anatomy 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 208000019206 urinary tract infection Diseases 0.000 description 1
- 238000007794 visualization technique Methods 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/3605—Implantable neurostimulators for stimulating central or peripheral nerve system
- A61N1/36125—Details of circuitry or electric components
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
- A61N1/0551—Spinal or peripheral nerve electrodes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/36007—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation of urogenital or gastrointestinal organs, e.g. for incontinence control
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/3605—Implantable neurostimulators for stimulating central or peripheral nerve system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/3605—Implantable neurostimulators for stimulating central or peripheral nerve system
- A61N1/36128—Control systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/3605—Implantable neurostimulators for stimulating central or peripheral nerve system
- A61N1/36128—Control systems
- A61N1/36135—Control systems using physiological parameters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/372—Arrangements in connection with the implantation of stimulators
- A61N1/375—Constructional arrangements, e.g. casings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/372—Arrangements in connection with the implantation of stimulators
- A61N1/375—Constructional arrangements, e.g. casings
- A61N1/3752—Details of casing-lead connections
- A61N1/3754—Feedthroughs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/372—Arrangements in connection with the implantation of stimulators
- A61N1/378—Electrical supply
- A61N1/3787—Electrical supply from an external energy source
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Radiology & Medical Imaging (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Gastroenterology & Hepatology (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Biophysics (AREA)
- Physiology (AREA)
- Electrotherapy Devices (AREA)
- Pain & Pain Management (AREA)
- Hospice & Palliative Care (AREA)
Description
本願は、米国仮出願第62/191,134号(2015年7月10日出願、名称「IMPLANTABLE NERVE STIMULATOR HAVING INTERNAL ELECTRONICS WITHOUT ASIC AND METHODS OF USE」)の利益を主張し、上記出願の全体は、参照により本明細書に引用される。本願は、以下の出願に関連する:米国仮出願第62/038,122号(2014年8月15日出願、名称「Devices and Methods for Anchoring of Neurostimulation Leads」)、米国仮出願第62/038,131号(2014年8月15日出願、名称「External Pulse Generator Device and Associated Methods for Trial Nerve Stimulation」、代理人事件番号97672−001100US)、米国仮出願第62/041,611号(2014年8月25日出願、名称「Electromyographic Lead Positioning and Stimulation Titration in a Nerve Stimulation System for Treatment of Overactive Bladder,Pain and Other Indicators」、代理人事件番号97672−001200US)、米国仮出願第62/110,274号(2015年1月30日出願、名称「Implantable Lead Affixation Structure for Nerve Stimulation to Alleviate Bladder Dysfunction and Other Indications」、代理人事件番号97672−001010US)、および、米国仮出願第62/101,888号(名称「Electromyographic Lead Positioning and Stimulation Titration in a Nerve Stimulation System for Treatment of Overactive Bladder」、代理人事件番号97672−001210US)、米国仮出願第62,101,899号(名称「Integrated Electromyographic Clinician Programmer For Use With an Implantable Neurostimulator」、代理人事件番号97672−001220US)、米国仮出願第62/101,897号(名称「Systems and Methods for Neurostimulation Electrode Configurations Based on Neural Localization」、代理人事件番号97672−001230US)、米国仮出願第62/101,666号(名称「Patient Remote and Associated Methods of Use With aNerve Stimulation System」、代理人事件番号97672−001400US)、米国仮出願第62/101,884号(名称「Attachment Devices and Associated Methods of Use With aNerve Stimulation Charging Device」、代理人事件番号97672−001500US)、米国仮出願第62/101,782号(名称「Improved Antenna and Methods of Use For an Implantable Nerve Stimulator」、代理人事件番号97672− 001600US)(以上の全ては2015年1月9日出願)。
上記出願の各々は、出願人に譲渡され、あらゆる目的のためにその全体が参照により本明細書に引用される。
本発明は、例えば、以下を提供する。
(項目1)
1つ以上の電気パルスを患者の体内の標的領域に送達するための埋め込み可能神経刺激装置システムであって、
複数の電極を備えている埋め込み可能導線であって、前記複数の電極は、前記導線の遠位端上に位置しており、前記電極は、患者の体内の標的領域に近接して位置付け可能であり、電気刺激を前記標的領域に提供する、埋め込み可能導線と、
前記導線の近位端に電気的に結合されている埋め込み可能パルス発生器と
を備え、
前記埋め込み可能パルス発生器は、
密封してシールされた内部容積を画定する生体適合性筐体と、
前記生体適合性筐体の前記密封してシールされた内部容積内に配置されている再充電可能電力供給源と、
前記生体適合性筐体の前記密封してシールされた内部容積内に配置されている回路と
を備え、
前記回路は、前記再充電可能電力供給源に電気的に結合され、1つ以上の電気パルスを生成するように構成され、前記回路は、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている第1の差動増幅器を備えている、
埋め込み可能神経刺激装置システム。
(項目2)
前記回路は、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている第2の差動増幅器をさらに備えている、項目1に記載の埋め込み可能神経刺激装置システム。
(項目3)
前記第2の差動増幅器は、前記第1の差動増幅器が、電流を前記導線上に位置している前記複数の電極のうちの少なくとも1つにソースするために選択されている場合、電流を前記導線上に位置している前記複数の電極のうちの少なくとも別の1つからシンクするために選択される、項目2に記載の埋め込み可能神経刺激装置システム。
(項目4)
前記第2の差動増幅器は、前記第1の差動増幅器が、電流を前記導線上に位置している前記複数の電極のうちの少なくとも1つからシンクするために選択されている場合、電流を前記導線上に位置している前記複数の電極のうちの少なくとも別の1つにソースするために選択される、項目3に記載の埋め込み可能神経刺激装置システム。
(項目5)
前記第1の差動増幅器は、電流コマンドに結合されている非反転入力を含み、前記電流コマンドは、第1の範囲内の電圧を前記非反転入力に供給するように構成され、前記第1の差動増幅器は、仮想接地に結合されている反転入力を含む、項目4に記載の埋め込み可能神経刺激装置システム。
(項目6)
供給される電圧の前記第1の範囲は、最大電圧および最小電圧を有し、前記第1の範囲の前記最大電圧と前記最小電圧との間の電圧の差異は、少なくとも5ボルトである、項目5に記載の埋め込み可能神経刺激装置システム。
(項目7)
前記仮想接地は、前記供給される電圧の前記最大電圧と前記最小電圧との間の接地電圧を備えている、項目6に記載の埋め込み可能神経刺激装置システム。
(項目8)
前記第1の差動増幅器の反転入力に結合されている仮想接地は、前記回路が定常状態で動作しているときの前記仮想接地の接地電圧と等しい前記反転入力における電圧を有する、項目7に記載の埋め込み可能神経刺激装置システム。
(項目9)
前記第1の差動増幅器は、前記第1の差動増幅器の非反転入力に印加される前記供給される電圧が前記仮想接地の接地電圧を上回る場合、電流をソースするために構成可能である、項目8に記載の埋め込み可能神経刺激装置システム。
(項目10)
前記第1の差動増幅器は、前記第1の差動増幅器の非反転入力に印加される前記供給される電圧が前記仮想接地の接地電圧未満である場合、電流をシンクするために構成可能である、項目9に記載の埋め込み可能神経刺激装置システム。
(項目11)
複数の電極を備えている埋め込み可能導線を用いて1つ以上の電気パルスを患者の体内の標的領域に送達するための埋め込み可能神経刺激装置であって、前記複数の電極は、前記標的領域に近接して位置付け可能であり、前記標的領域に電気的に統合可能であり、
前記埋め込み可能神経刺激装置は、
患者の体内に埋め込まれるように構成されている密封してシールされた内部容積を画定する生体適合性筐体と、
前記生体適合性筐体の前記密封してシールされた内部容積内に配置されている再充電可能バッテリと、
前記生体適合性筐体の前記密封してシールされた内部容積内に配置されている回路と
を備え、
前記回路は、前記再充電可能電力供給源に電気的に結合され、1つ以上の電気パルスを生成するように構成され、
前記回路は、
第1の電流制御モジュールと、
第2の電流制御モジュールと
を備え、
前記第1の電流制御モジュールおよび前記第2の電流制御モジュールの各々は、
非反転入力、反転入力、および出力を有する電流駆動差動増幅器と、
前記非反転入力に結合されている電流制御であって、前記電流制御は、第1の範囲内の電圧を前記非反転入力に供給するように構成され、前記第1の範囲は、最小電圧と最大電圧との間である、電流制御と、
前記反転入力に結合されている仮想接地であって、前記仮想接地は、前記最小電圧と前記最大電圧との間の接地電圧を有する、仮想接地と、
前記電流駆動差動増幅器の出力を前記導線に選択的に結合している負荷経路であって、前記負荷経路は、前記電流駆動差動増幅器の出力と前記導線との間に位置する感知抵抗器を備えている、負荷経路と
を備えている、埋め込み可能神経刺激装置。
(項目12)
前記第1および第2の電流制御モジュールのうちの少なくとも1つは、感知非反転入力、感知反転入力、および感知出力を備えている電流感知差動増幅器を備えている、項目11に記載の埋め込み可能神経刺激装置。
(項目13)
前記感知非反転入力は、第1の抵抗を有する第1の抵抗器を介して、前記感知抵抗器と前記電流駆動差動増幅器の出力との間で前記負荷経路に接続され、前記感知反転入力は、前記第1の抵抗を有する第2の抵抗器を介して、前記感知抵抗器と前記導線との間で前記負荷経路に接続されている、項目12に記載の埋め込み可能神経刺激装置。
(項目14)
前記感知非反転入力は、第2の抵抗を有する第3の抵抗器を介して、前記仮想接地に接続され、前記感知反転入力は、前記第2の抵抗を有する第4の抵抗器を介して、前記感知出力に接続されている、項目13に記載の埋め込み可能神経刺激装置。
(項目15)
前記第2の抵抗は、前記第1の抵抗の少なくとも2倍である、項目14に記載の埋め込み可能神経刺激装置。
(項目16)
前記第2の抵抗は、前記第1の抵抗の少なくとも10倍である、項目15に記載の埋め込み可能神経刺激装置。
(項目17)
前記感知抵抗器を横断する電圧降下を測定するように構成されている電圧センサをさらに備えている、項目14に記載の埋め込み可能神経刺激装置。
(項目18)
前記感知抵抗器を横断する電圧降下は、前記接地電圧と前記電流感知差動増幅器の出力との間の差異を決定することによって測定される、項目17に記載の埋め込み可能神経刺激装置。
(項目19)
前記第1の制御モジュールは、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている、項目18に記載の埋め込み可能神経刺激装置。
(項目20)
前記第2の制御モジュールは、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている、項目19に記載の埋め込み可能神経刺激装置。
(項目21)
前記電流駆動差動増幅器の反転入力に結合されている前記仮想接地は、前記第1の制御モジュールおよび前記第2の制御モジュールのうちの少なくとも1つが定常状態で動作しているとき、前記仮想接地の接地電圧と等しい前記反転入力における電圧を有する、項目20に記載の埋め込み可能神経刺激装置。
(項目22)
前記電流駆動差動増幅器は、前記電流駆動差動増幅器の非反転入力に印加される供給される電圧が前記仮想接地の接地電圧を上回る場合、電流をソースするために構成可能である、項目21に記載の埋め込み可能神経刺激装置。
(項目23)
前記電流駆動差動増幅器は、前記電流駆動差動増幅器の非反転入力に印加される前記供給される電圧が前記仮想接地の接地電圧未満である場合、電流をシンクするために構成可能である、項目22に記載の埋め込み可能神経刺激装置。
(項目24)
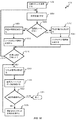
埋め込み可能導線に対して電流を選択的にソースまたはシンクするように埋め込み可能神経刺激装置の回路の作動方法であって、前記埋め込み可能導線は、前記回路に電気的に結合され、前記埋め込み可能導線は、電気刺激を前記標的領域に送達するために患者の体内の標的領域に近接して位置付け可能である複数の電極を備え、
前記方法は、
第1の差動増幅器の所望の動作を識別することであって、前記第1の差動増幅器は、生体適合性筐体の密封してシールされた内部容積内に位置し、前記第1の差動増幅器は、電流を導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている、ことと、
第1の制御信号を生成することであって、前記第1の制御信号は、前記第1の差動増幅器に、電流を前記導線にソースすることまたは電流を前記導線からシンクすることのいずれかを行わせるように構成されている、ことと
前記第1の制御信号を前記第1の差動増幅器に提供することと
を含む、方法。
(項目25)
前記第1の制御信号を生成することは、前記第1の差動増幅器の所望の動作が電流を前記導線にソースすることであるか、または電流を前記導線からシンクすることであるかを決定することを含む、項目24に記載の方法。
(項目26)
前記第1の制御信号を生成することは、電流を前記第1の差動増幅器からソースすべきか、または電流を前記第1の差動増幅器に選択的にシンクすべきかを決定することを含む、項目25に記載の方法。
(項目27)
前記第1の制御信号を生成することは、電流を前記第1の差動増幅器からソースすべきことが決定された場合、仮想接地電圧を上回る電圧を有する前記第1の制御信号を生成することをさらに含む、項目26に記載の方法。
(項目28)
前記制御信号を生成することは、電流を前記第1の差動増幅器にシンクすべきことが決定された場合、仮想接地電圧未満の電圧を有する前記第1の制御信号を生成することをさらに含む、項目26に記載の方法。
(項目29)
第2の差動増幅器の所望の動作を識別することであって、前記第2の差動増幅器は、生体適合性筐体の密封してシールされた内部容積内に位置し、前記第2の差動増幅器は、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている、ことと、
第2の制御信号を生成することであって、前記第2の制御信号は、前記第2の差動増幅器に、電流を前記導線にソースするか、または電流を前記導線からシンクするかのいずれかを行わせるように構成されている、ことと、
前記第2の制御信号を前記第2の差動増幅器に提供することと
をさらに含む、
項目24に記載の方法。
(項目30)
前記第1の制御信号が前記第1の差動増幅器に電流を前記導線にソースさせる場合、前記第2の信号は、前記第2の差動増幅器に電流を前記導線からシンクさせるために生成される、項目29に記載の方法。
(項目31)
前記第1の制御信号が前記第1の差動増幅器に電流を前記導線からシンクさせる場合、前記第2の信号は、前記第2の差動増幅器に電流を前記導線にソースさせるために生成される、項目29に記載の方法。
(項目32)
前記第1の差動増幅器は、電流を前記導線の前記複数の電極のうちの第1の少なくとも1つに選択的にソースし、電流を前記導線の前記複数の電極のうちの前記第1の少なくとも1つから選択的にシンクするように構成され、前記第2の差動増幅器は、電流を前記導線の前記複数の電極のうちの第2の少なくとも1つに選択的にソースし、電流を前記導線の前記複数の電極のうちの前記第2の少なくとも1つから選択的にシンクするように構成されている、項目29に記載の方法。
(項目33)
前記導線の前記複数の電極のうちの前記第1の少なくとも1つおよび前記導線の前記複数の電極のうちの前記第2の少なくとも1つは、前記患者の体内の標的領域を通して回路を完成するように選択される、項目32に記載の方法。
(項目34)
前記患者の身体の標的領域は、仙骨組織を備えている、項目33に記載の方法。
(項目35)
前記第1の差動増幅器および前記第2の差動増幅器は、電流をソースまたは電流をシンクし、1つまたはいくつかの電気パルスを生成するように構成されている、項目29に記載の方法。
(項目36)
前記1つまたはいくつかの電気パルスは、前記電気パルスのパラメータを規定するパルスプログラムに従って生成される、項目35に記載の方法。
(項目37)
前記1つまたはいくつかの電気パルスは、単極であることができる、項目36に記載の方法。
(項目38)
前記1つまたはいくつかの電気パルスは、双極であることができる、項目36に記載の方法。
(項目39)
埋め込み可能導線を用いて1つ以上の電気パルスを患者の体内の標的領域に送達するための埋め込み可能神経刺激装置であって、前記埋め込み可能導線は、前記標的領域に近接して位置付け可能であり、前記標的領域に電気的に統合可能な複数の電極を備え、
前記埋め込み可能導線は、
無ASIC電流ソース/シンク発生器であって、前記発生器は、
電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている第1の差動増幅器と、
電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている第2の差動増幅器と
を備えている、無ASIC電流ソース/シンク発生器と、
刺激コントローラと
を備え、
前記刺激コントローラは、
前記第1の差動増幅器のために、電流をソースすることまたはシンクすることのうちの一方を選択するための第1の電気信号を生成することと、
前記第2の差動増幅器のために、電流をソースすることまたはシンクすることのうちの他方を選択するための第2の電気信号を生成することと
を行うように構成されている、埋め込み可能神経刺激装置。
(項目40)
前記第1の差動増幅器は、仮想接地に接続されている反転入力および第1の電流コマンドに接続されている非反転入力を備え、前記第2の差動増幅器は、前記仮想接地に接続されている反転入力および第2の電流コマンドに接続されている非反転入力を備えている、項目39に記載の埋め込み可能神経刺激装置。
(項目41)
前記仮想接地は、第1の電圧を前記第1の差動増幅器の反転入力に供給し、前記第1の電圧を前記第2の差動増幅器の反転入力に供給する、項目40に記載の埋め込み可能神経刺激装置。
(項目42)
前記第1の電流コマンドは、電流をソースするために前記第1の差動増幅器を選択する場合、前記供給される第1の電圧を上回る第2の電圧を供給し、前記第1の電流コマンドは、電流をシンクするために前記第1の差動増幅器を選択する場合、前記供給される第1の電圧未満の第2の電圧を供給する、項目41に記載の埋め込み可能神経刺激装置。
(項目43)
前記第2の電流コマンドは、電流をソースするために前記第2の差動増幅器を選択する場合、前記供給される第1の電圧を上回る第3の電圧を供給し、前記第2の電流コマンドは、電流をシンクするために前記第2の差動増幅器を選択する場合、前記供給される第1の電圧未満の第3の電圧を供給する、項目42に記載の埋め込み可能神経刺激装置。
本明細書に説明されるもののうちのいずれか等の神経刺激(または以降で同義的に使用され得るような神経調節)治療システムは、急性疼痛障害、運動障害、情動障害、ならびに膀胱関連機能不全等の種々の病気および関連症状を治療するために使用されることができる。神経刺激によって治療され得る疼痛障害の例は、脊椎手術後疼痛症候群、反射性交感神経性ジストロフィまたは複合性局所疼痛症候群、灼熱痛、クモ膜炎、および末梢神経障害を含む。運動障害は、筋麻痺、振戦、ジストニア、およびパーキンソン病を含む。情動障害は、うつ病、強迫性障害、群発頭痛、トゥレット障害、およびあるタイプの慢性疼痛を含む。膀胱関連機能不全は、OAB、切迫尿失禁、切迫頻尿、および尿閉を含むが、それらに限定されない。OABは、単独で、または組み合わせて、切迫尿失禁および切迫頻尿を含むことができる。切迫尿失禁は、突然の強い尿意(切迫性)に関連付けられる失禁である。切迫頻尿は、多くの場合、非常に少量の排尿をもたらす(頻尿)、頻繁で多くの場合は制御不能な尿意切迫感(切迫性)である。尿閉は、膀胱を空にすることができない状態である。神経刺激治療は、その状態もしくは関連症状に関連付けられる感覚および/または運動制御に関する標的神経組織の神経刺激を達成することによって、特定の状態に対処するように構成されることができる。
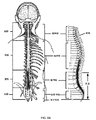
SNMは、切迫尿失禁、切迫頻尿、および非閉塞性尿閉の管理のための安全、効果的、可逆的、かつ長期継続的な治療選択肢を提供する確立された療法である。SNM療法は、下背に位置する仙骨神経を刺激するために、弱電気パルスの使用を伴う。電極は、電極導線を仙骨の対応する孔の中に挿入することによって、仙骨神経に隣接して、通常、S3レベルに設置される。電極は、皮下に挿入され、後に、本明細書では「埋め込み可能神経刺激装置」または「神経刺激装置」とも称される、埋め込み可能パルス発生器(IPG)に取り付けられる。切迫尿失禁および切迫頻尿の両方の患者のために、5年の耐久性を含む、OABの治療のためのSNMの安全性ならびに有効性が、複数の研究によって支持され、十分に証明されている。SNMは、より保守的治療に失敗した患者、またはその候補ではない患者における慢性便失禁を治療するためにも是認されている。
現在、SNM資格は、試験段階を油脂、成功した場合、恒久的埋め込みが続く。試験段階は、療法が効果的であるかどうかを患者が評価することを可能にされる試験刺激期間である。典型的には、試験刺激を行うために利用される、2つの技法がある。第1は、経皮的神経評価(PNE)と称される通院ベースの手技であり、他方は、段階的試験である。
従来のアプローチは、膀胱関連機能不全の治療において有効性を示しているが、神経刺激導線の位置付けおよび導線の試験的位置と恒久的埋め込み位置との間の一貫性を向上させる必要が存在する。神経刺激は、1つ以上の神経刺激電極を介して、パルス発生器から特定の神経もしくは標的領域に治療用刺激を一貫して送達することに依拠する。神経刺激電極は、患者組織内に形成されるトンネルを通して前進させられることができる、埋め込み可能導線の遠位端上に提供される。埋め込み可能神経刺激システムは、患者に大幅な自由度および移動性を提供するが、そのようなシステムの神経刺激電極が外科的に埋め込まれる前に、それらを調節することがより容易であり得る。医師は、IPGを埋め込む前に、患者が所望の運動および/または感覚反応を有することを確認することが望ましい。少なくともいくつかの治療(少なくともいくつかの形態の排尿および/または排便機能不全の治療を含む)に対して、適切な運動反応の実証は、感覚反応が要求されないことも、利用可能でないこともあるが(例えば、患者が全身麻酔下にある)、正確かつ客観的な導線設置のために高度に有益であり得る。
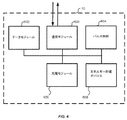
図1は、本発明の側面による、試験的神経刺激システム200および恒久的埋め込み式神経刺激システム100の両方を含む、例示的神経刺激システムを概略的に図示する。EPG80およびIPG10の各々は、臨床医用プログラム装置(CP)60および患者遠隔装置70との適合性があり、無線で通信し、臨床医用プログラム装置60および患者遠隔装置70は、試験的神経刺激システム200および/または試験成功後の恒久的埋め込み式システム100を位置付けることおよび/またはプログラムすることにおいて使用される。上で議論されるように、臨床医用プログラム装置は、導線設置、プログラミング、再プログラミング、刺激制御、および/またはパラメータ設定に役立つように、特殊ソフトウェア、特殊ハードウェア、ならびに/もしくは両方を含むことができる。加えて、IPGおよびEPGの各々は、患者が、患者遠隔装置を用いて、刺激の少なくとも一部を制御し(例えば、事前に設定されたプログラムを開始すること、刺激を増加または減少させること)、および/またはバッテリステータスを監視することを可能にする。このアプローチは、試験的システムと恒久的システムとの間のほぼシームレスな遷移も可能にする。
Claims (23)
- 1つ以上の電気パルスを患者の体内の標的領域に送達するための埋め込み可能神経刺激装置システムであって、
複数の電極を備えている埋め込み可能導線であって、前記複数の電極は、前記導線の遠位端上に位置しており、前記電極は、患者の体内の標的領域に近接して位置付け可能であり、電気刺激を前記標的領域に提供する、埋め込み可能導線と、
前記導線の近位端に電気的に結合されている埋め込み可能パルス発生器と
を備え、
前記埋め込み可能パルス発生器は、
密封してシールされた内部容積を画定する生体適合性筐体と、
前記生体適合性筐体の前記密封してシールされた内部容積内に配置されている再充電可能電力供給源と、
前記生体適合性筐体の前記密封してシールされた内部容積内に配置されている回路と
を備え、
前記回路は、前記再充電可能電力供給源に電気的に結合され、1つ以上の電気パルスを生成するように構成され、前記回路は、電流コマンドおよび一定電圧源に結合される入力を有する第1の差動増幅器であって、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている第1の差動増幅器を備えており、
前記第1の差動増幅器の前記入力は、前記電流コマンドに結合されている非反転入力を含み、前記電流コマンドは、第1の範囲内の電圧を前記非反転入力に供給するように構成され、前記第1の差動増幅器の前記入力は、前記一定電圧源に結合されている反転入力を含む、
埋め込み可能神経刺激装置システム。 - 前記回路は、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている第2の差動増幅器をさらに備えている、請求項1に記載の埋め込み可能神経刺激装置システム。
- 前記第2の差動増幅器は、前記第1の差動増幅器が、電流を前記導線上に位置している前記複数の電極のうちの少なくとも1つにソースするために選択されている場合、電流を前記導線上に位置している前記複数の電極のうちの少なくとも別の1つからシンクするために選択される、請求項2に記載の埋め込み可能神経刺激装置システム。
- 前記第2の差動増幅器は、前記第1の差動増幅器が、電流を前記導線上に位置している前記複数の電極のうちの少なくとも1つからシンクするために選択されている場合、電流を前記導線上に位置している前記複数の電極のうちの少なくとも別の1つにソースするために選択される、請求項2に記載の埋め込み可能神経刺激装置システム。
- 供給される電圧の前記第1の範囲は、最大電圧および最小電圧を有し、前記第1の範囲の前記最大電圧と前記最小電圧との間の電圧の差異は、少なくとも5ボルトである、請求項1に記載の埋め込み可能神経刺激装置システム。
- 前記一定電圧源は、前記供給される電圧の前記最大電圧と前記最小電圧との間の電圧を備えている、請求項5に記載の埋め込み可能神経刺激装置システム。
- 前記第1の差動増幅器の反転入力に結合されている前記一定電圧源は、前記回路が定常状態で動作しているときの前記一定電圧源の電圧と等しい前記反転入力における電圧を有する、請求項6に記載の埋め込み可能神経刺激装置システム。
- 前記第1の差動増幅器は、前記第1の差動増幅器の非反転入力に印加される前記供給される電圧が前記一定電圧源の電圧を上回る場合、電流をソースするために構成可能である、請求項7に記載の埋め込み可能神経刺激装置システム。
- 前記第1の差動増幅器は、前記第1の差動増幅器の非反転入力に印加される前記供給される電圧が前記一定電圧源の電圧未満である場合、電流をシンクするために構成可能である、請求項8に記載の埋め込み可能神経刺激装置システム。
- 埋め込み可能導線に対して電流を選択的にソースまたはシンクするための埋め込み可能神経刺激装置の回路の作動方法であって、前記埋め込み可能導線は、前記回路に電気的に結合され、前記埋め込み可能導線は、電気刺激を標的領域に送達するために患者の体内の前記標的領域に近接して位置付け可能である複数の電極を備え、
前記方法は、
第1の差動増幅器の所望の動作を識別することであって、前記第1の差動増幅器は、一定電圧源に結合される入力を有し、前記第1の差動増幅器は、生体適合性筐体の密封してシールされた内部容積内に位置し、前記第1の差動増幅器は、電流を導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている、ことと、
第1の制御信号を生成することであって、前記第1の制御信号は、前記第1の差動増幅器に、電流を前記導線にソースすることまたは電流を前記導線からシンクすることのいずれかを行わせるように構成されており、前記第1の制御信号を生成することは、電流を前記第1の差動増幅器からソースすべきことが決定された場合、前記一定電圧源の電圧を上回る電圧を有する前記第1の制御信号を生成することを含む、ことと、
前記第1の制御信号を前記第1の差動増幅器に提供することと
を含む、方法。 - 前記第1の制御信号を生成することは、前記第1の差動増幅器の所望の動作が電流を前記導線にソースすることであるか、または電流を前記導線からシンクすることであるかを決定することを含む、請求項10に記載の方法。
- 前記第1の制御信号を生成することは、電流を前記第1の差動増幅器からソースすべきか、または電流を前記第1の差動増幅器に選択的にシンクすべきかを決定することを含む、請求項11に記載の方法。
- 前記制御信号を生成することは、電流を前記第1の差動増幅器にシンクすべきことが決定された場合、前記一定電圧源の電圧未満の電圧を有する前記第1の制御信号を生成することをさらに含む、請求項12に記載の方法。
- 第2の差動増幅器の所望の動作を識別することであって、前記第2の差動増幅器は、生体適合性筐体の密封してシールされた内部容積内に位置し、前記第2の差動増幅器は、電流を前記導線に選択的にソースし、電流を前記導線から選択的にシンクするように構成されている、ことと、
第2の制御信号を生成することであって、前記第2の制御信号は、前記第2の差動増幅器に、電流を前記導線にソースするか、または電流を前記導線からシンクするかのいずれかを行わせるように構成されている、ことと、
前記第2の制御信号を前記第2の差動増幅器に提供することと
をさらに含む、
請求項10に記載の方法。 - 前記第1の制御信号が前記第1の差動増幅器に電流を前記導線にソースさせる場合、前記第2の信号は、前記第2の差動増幅器に電流を前記導線からシンクさせるために生成される、請求項14に記載の方法。
- 前記第1の制御信号が前記第1の差動増幅器に電流を前記導線からシンクさせる場合、前記第2の信号は、前記第2の差動増幅器に電流を前記導線にソースさせるために生成される、請求項14に記載の方法。
- 前記第1の差動増幅器は、電流を前記導線の前記複数の電極のうちの第1の少なくとも1つに選択的にソースし、電流を前記導線の前記複数の電極のうちの前記第1の少なくとも1つから選択的にシンクするように構成され、前記第2の差動増幅器は、電流を前記導線の前記複数の電極のうちの第2の少なくとも1つに選択的にソースし、電流を前記導線の前記複数の電極のうちの前記第2の少なくとも1つから選択的にシンクするように構成されている、請求項14に記載の方法。
- 前記導線の前記複数の電極のうちの前記第1の少なくとも1つおよび前記導線の前記複数の電極のうちの前記第2の少なくとも1つは、前記患者の体内の前記標的領域を通して回路を完成するように選択される、請求項17に記載の方法。
- 前記患者の身体の前記標的領域は、仙骨組織を備えている、請求項18に記載の方法。
- 前記第1の差動増幅器および前記第2の差動増幅器は、電流をソースまたは電流をシンクし、1つまたはいくつかの電気パルスを生成するように構成されている、請求項14に記載の方法。
- 前記1つまたはいくつかの電気パルスは、前記電気パルスのパラメータを規定するパルスプログラムに従って生成される、請求項20に記載の方法。
- 前記1つまたはいくつかの電気パルスは、単極であることができる、請求項21に記載の方法。
- 前記1つまたはいくつかの電気パルスは、双極であることができる、請求項21に記載の方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201562191134P | 2015-07-10 | 2015-07-10 | |
| US62/191,134 | 2015-07-10 | ||
| PCT/US2016/041508 WO2017011305A1 (en) | 2015-07-10 | 2016-07-08 | Implantable nerve stimulator having internal electronics without asic and methods of use |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2018519127A JP2018519127A (ja) | 2018-07-19 |
| JP2018519127A5 JP2018519127A5 (ja) | 2019-08-15 |
| JP6946261B2 true JP6946261B2 (ja) | 2021-10-06 |
Family
ID=57730694
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018500525A Active JP6946261B2 (ja) | 2015-07-10 | 2016-07-08 | Asicを用いない内部電子機器を有する埋め込み可能神経刺激装置および方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (3) | US11766568B2 (ja) |
| EP (1) | EP3319683A4 (ja) |
| JP (1) | JP6946261B2 (ja) |
| CN (2) | CN107847731B (ja) |
| AU (1) | AU2016291554B2 (ja) |
| CA (1) | CA2991903C (ja) |
| WO (1) | WO2017011305A1 (ja) |
Families Citing this family (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11413458B2 (en) | 2011-05-19 | 2022-08-16 | Neuros Medical, Inc. | Nerve cuff electrode for neuromodulation in large human nerve trunks |
| US20150018728A1 (en) | 2012-01-26 | 2015-01-15 | Bluewind Medical Ltd. | Wireless neurostimulators |
| US9861812B2 (en) | 2012-12-06 | 2018-01-09 | Blue Wind Medical Ltd. | Delivery of implantable neurostimulators |
| KR101656723B1 (ko) * | 2015-06-30 | 2016-09-12 | 재단법인 오송첨단의료산업진흥재단 | 피드스루 제조방법 |
| CA2991903C (en) | 2015-07-10 | 2023-10-17 | Axonics Modulation Technologies, Inc. | Implantable nerve stimulator having internal electronics without asic and methods of use |
| US10105540B2 (en) | 2015-11-09 | 2018-10-23 | Bluewind Medical Ltd. | Optimization of application of current |
| AU2017315473B2 (en) * | 2016-08-26 | 2022-09-29 | Spr Therapeutics, Inc. | Devices and methods for delivery of electrical current for pain relief |
| US10124178B2 (en) | 2016-11-23 | 2018-11-13 | Bluewind Medical Ltd. | Implant and delivery tool therefor |
| US20180353764A1 (en) | 2017-06-13 | 2018-12-13 | Bluewind Medical Ltd. | Antenna configuration |
| WO2019034223A1 (en) * | 2017-08-18 | 2019-02-21 | Innocon Medical Aps | SYSTEM FOR ELECTRICAL STIMULATION OF NERFS |
| EP3668586B1 (en) * | 2017-08-18 | 2021-06-02 | Innocon Medical ApS | System for electrical stimulation of nerves |
| EP3700617A4 (en) | 2017-10-25 | 2021-08-04 | Epineuron Technologies Inc. | SYSTEMS AND METHODS FOR DELIVERY OF ANTI-NEURODEGENERATIVE THERAPY |
| US10589089B2 (en) | 2017-10-25 | 2020-03-17 | Epineuron Technologies Inc. | Systems and methods for delivering neuroregenerative therapy |
| EP3723840B1 (en) | 2017-12-13 | 2022-11-30 | Neuros Medical, Inc. | Nerve cuff deployment devices |
| IT201800002962A1 (it) | 2018-02-22 | 2019-08-22 | Newronika Srl | Apparato per il trattamento di disordini neurologici mediante elettrostimolazione e metodo di elaborazione del segnale neurologico raccolto da detto apparato |
| AU2019253298B2 (en) | 2018-04-09 | 2024-06-27 | Neuros Medical, Inc. | Apparatuses and methods for setting an electrical dose |
| AU2019255377A1 (en) * | 2018-04-19 | 2020-11-26 | Iota Biosciences, Inc. | Implants using ultrasonic communication for neural sensing and stimulation |
| US20200368534A1 (en) * | 2019-05-23 | 2020-11-26 | Axonics Modulation Technologies, Inc. | Low energy implantable devices and methods of use |
| US11848090B2 (en) | 2019-05-24 | 2023-12-19 | Axonics, Inc. | Trainer for a neurostimulator programmer and associated methods of use with a neurostimulation system |
| US11247043B2 (en) | 2019-10-01 | 2022-02-15 | Epineuron Technologies Inc. | Electrode interface devices for delivery of neuroregenerative therapy |
| AU2021219722A1 (en) | 2020-02-11 | 2022-09-08 | Neuros Medical, Inc. | System and method for quantifying qualitative patient-reported data sets |
| TW202202091A (zh) * | 2020-03-25 | 2022-01-16 | 美商感應學公司 | 具有嵌入式電源的無線可植入設備的外殼 |
| US11400299B1 (en) | 2021-09-14 | 2022-08-02 | Rainbow Medical Ltd. | Flexible antenna for stimulator |
Family Cites Families (480)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3057356A (en) | 1960-07-22 | 1962-10-09 | Wilson Greatbatch Inc | Medical cardiac pacemaker |
| US3348548A (en) | 1965-04-26 | 1967-10-24 | William M Chardack | Implantable electrode with stiffening stylet |
| US3646940A (en) | 1969-07-15 | 1972-03-07 | Univ Minnesota | Implantable electronic stimulator electrode and method |
| US3888260A (en) | 1972-06-28 | 1975-06-10 | Univ Johns Hopkins | Rechargeable demand inhibited cardiac pacer and tissue stimulator |
| US3825015A (en) | 1972-12-14 | 1974-07-23 | American Optical Corp | Single catheter for atrial and ventricular stimulation |
| US3824129A (en) | 1973-03-14 | 1974-07-16 | Mallory & Co Inc P R | Heart pacer rechargeable cell and protective control system |
| US3902501A (en) | 1973-06-21 | 1975-09-02 | Medtronic Inc | Endocardial electrode |
| US3970912A (en) | 1973-08-28 | 1976-07-20 | Hoffman Philip A | Battery charging circuit |
| US3942535A (en) | 1973-09-27 | 1976-03-09 | G. D. Searle & Co. | Rechargeable tissue stimulating system |
| US3939843A (en) | 1974-03-04 | 1976-02-24 | Medtronic, Inc. | Transvenous electrode |
| US3995623A (en) | 1974-12-23 | 1976-12-07 | American Hospital Supply Corporation | Multipurpose flow-directed catheter |
| US4019518A (en) | 1975-08-11 | 1977-04-26 | Medtronic, Inc. | Electrical stimulation system |
| US4044774A (en) | 1976-02-23 | 1977-08-30 | Medtronic, Inc. | Percutaneously inserted spinal cord stimulation lead |
| US4082097A (en) | 1976-05-20 | 1978-04-04 | Pacesetter Systems Inc. | Multimode recharging system for living tissue stimulators |
| US4141365A (en) | 1977-02-24 | 1979-02-27 | The Johns Hopkins University | Epidural lead electrode and insertion needle |
| US4166469A (en) | 1977-12-13 | 1979-09-04 | Littleford Philip O | Apparatus and method for inserting an electrode |
| US4340062A (en) | 1978-11-06 | 1982-07-20 | Medtronic, Inc. | Body stimulator having selectable stimulation energy levels |
| US4285347A (en) | 1979-07-25 | 1981-08-25 | Cordis Corporation | Stabilized directional neural electrode lead |
| US4269198A (en) | 1979-12-26 | 1981-05-26 | Medtronic, Inc. | Body implantable lead |
| DE3015260A1 (de) | 1980-04-21 | 1981-10-22 | Siemens AG, 1000 Berlin und 8000 München | Endocard-elektrodenanordnung |
| US4379462A (en) | 1980-10-29 | 1983-04-12 | Neuromed, Inc. | Multi-electrode catheter assembly for spinal cord stimulation |
| US4721118A (en) | 1981-04-20 | 1988-01-26 | Cordis Leads, Inc. | Pervenous electrical pacing lead with foldable fins |
| US4468723A (en) | 1981-04-24 | 1984-08-28 | Hewlett-Packard Company | Magnetically regulated power supply |
| US4437475A (en) | 1981-08-28 | 1984-03-20 | Medtronic, Inc. | Transvenous cardiovascular integrated lead anchoring sleeve, protector, and permanent lead introducer stop gap |
| DE3146182C2 (de) | 1981-11-21 | 1984-03-29 | Peter Dr. 7889 Grenzach-Wyhlen Osypka | Transvenös ins Herz einführbare Elektrodenanordnung für einen Herzschrittmacher |
| US4512351A (en) | 1982-11-19 | 1985-04-23 | Cordis Corporation | Percutaneous lead introducing system and method |
| US4961422A (en) | 1983-01-21 | 1990-10-09 | Marchosky J Alexander | Method and apparatus for volumetric interstitial conductive hyperthermia |
| US4558702A (en) | 1983-01-21 | 1985-12-17 | Cordis Corporation | Cardiac pacer having input/output circuit programmable for use with unipolar and bipolar pacer leads |
| CH673768A5 (ja) | 1983-01-21 | 1990-04-12 | Ramm Associates | |
| US4800898A (en) | 1983-10-07 | 1989-01-31 | Cordis Corporation | Neural stimulator electrode element and lead |
| US4654880A (en) | 1983-12-09 | 1987-03-31 | Minnesota Mining And Manufacturing Company | Signal transmission system |
| US4550731A (en) | 1984-03-07 | 1985-11-05 | Cordis Corporation | Acquisition circuit for cardiac pacer |
| US4662382A (en) | 1985-01-16 | 1987-05-05 | Intermedics, Inc. | Pacemaker lead with enhanced sensitivity |
| US4722353A (en) | 1985-09-16 | 1988-02-02 | Intermedics, Inc. | Stabilizer for implantable electrode |
| US4673867A (en) | 1986-06-30 | 1987-06-16 | Motorola, Inc. | Current mirror circuit and method for providing zero temperature coefficient trimmable current ratios |
| US4848352A (en) | 1987-02-13 | 1989-07-18 | Telectronics, N.V. | Method for cardiac pacing and sensing using combination of electrodes |
| US4744371A (en) | 1987-04-27 | 1988-05-17 | Cordis Leads, Inc. | Multi-conductor lead assembly for temporary use |
| US4957118A (en) | 1988-01-15 | 1990-09-18 | Jay Erlebacher | Electrode lead |
| US4860446A (en) | 1988-02-16 | 1989-08-29 | Medtronic, Inc. | Medical electrical lead and method of manufacture |
| US5052407A (en) | 1988-04-14 | 1991-10-01 | Mieczyslaw Mirowski | Cardiac defibrillation/cardioversion spiral patch electrode |
| DE3914662A1 (de) | 1989-05-03 | 1990-11-08 | Alt Eckhard | Vorrichtung zum uebertragen elektrischer signale zwischen einem implantierbaren medizinischen geraet und elektrisch erregbarem menschlichen gewebe |
| US4989617A (en) | 1989-07-14 | 1991-02-05 | Case Western Reserve University | Intramuscular electrode for neuromuscular stimulation system |
| US5012176A (en) | 1990-04-03 | 1991-04-30 | Baxter International, Inc. | Apparatus and method for calorimetrically determining battery charge state |
| CA2038160C (en) | 1991-03-13 | 1996-10-22 | Jiri K. Nor | Charging circuits for rechargeable batteries and cells |
| US5255691A (en) | 1991-11-13 | 1993-10-26 | Medtronic, Inc. | Percutaneous epidural lead introducing system and method |
| US5193539A (en) | 1991-12-18 | 1993-03-16 | Alfred E. Mann Foundation For Scientific Research | Implantable microstimulator |
| GB9211085D0 (en) | 1992-05-23 | 1992-07-08 | Tippey Keith E | Electrical stimulation |
| US5257634A (en) | 1992-07-16 | 1993-11-02 | Angeion Corporation | Low impedence defibrillation catheter electrode |
| US5676651A (en) | 1992-08-06 | 1997-10-14 | Electric Boat Corporation | Surgically implantable pump arrangement and method for pumping body fluids |
| US5342408A (en) | 1993-01-07 | 1994-08-30 | Incontrol, Inc. | Telemetry system for an implantable cardiac device |
| JP3384027B2 (ja) | 1993-05-14 | 2003-03-10 | ソニー株式会社 | 充電方法および充電器 |
| US5864220A (en) | 1993-09-16 | 1999-01-26 | Chartec Laboratories A/S | Method and apparatus for controlling the charging of a rechargeable battery to ensure that full charge is achieved without damaging the battery |
| US5439485A (en) | 1993-09-24 | 1995-08-08 | Ventritex, Inc. | Flexible defibrillation electrode of improved construction |
| US5484445A (en) | 1993-10-12 | 1996-01-16 | Medtronic, Inc. | Sacral lead anchoring system |
| SE9304031D0 (sv) | 1993-12-03 | 1993-12-03 | Siemens Elema Ab | Elektrodsystem |
| JPH07222370A (ja) | 1994-01-28 | 1995-08-18 | Sanyo Electric Co Ltd | 温度センサーを有する充電器 |
| US6249703B1 (en) | 1994-07-08 | 2001-06-19 | Medtronic, Inc. | Handheld patient programmer for implantable human tissue stimulator |
| US5571148A (en) | 1994-08-10 | 1996-11-05 | Loeb; Gerald E. | Implantable multichannel stimulator |
| US6035237A (en) | 1995-05-23 | 2000-03-07 | Alfred E. Mann Foundation | Implantable stimulator that prevents DC current flow without the use of discrete output coupling capacitors |
| US5690693A (en) | 1995-06-07 | 1997-11-25 | Sulzer Intermedics Inc. | Transcutaneous energy transmission circuit for implantable medical device |
| US5702431A (en) | 1995-06-07 | 1997-12-30 | Sulzer Intermedics Inc. | Enhanced transcutaneous recharging system for battery powered implantable medical device |
| US5712795A (en) | 1995-10-02 | 1998-01-27 | Alaris Medical Systems, Inc. | Power management system |
| US5877472A (en) | 1996-02-22 | 1999-03-02 | Pacesetter, Inc. | System for laser-welding components of an implantable device |
| US6898454B2 (en) | 1996-04-25 | 2005-05-24 | The Johns Hopkins University | Systems and methods for evaluating the urethra and the periurethral tissues |
| DE19623788A1 (de) | 1996-06-04 | 1997-12-11 | Biotronik Mess & Therapieg | Implantierbares Stimulationsgerät |
| US6609031B1 (en) | 1996-06-07 | 2003-08-19 | Advanced Neuromodulation Systems, Inc. | Multiprogrammable tissue stimulator and method |
| US5733313A (en) | 1996-08-01 | 1998-03-31 | Exonix Corporation | RF coupled, implantable medical device with rechargeable back-up power source |
| US5713939A (en) | 1996-09-16 | 1998-02-03 | Sulzer Intermedics Inc. | Data communication system for control of transcutaneous energy transmission to an implantable medical device |
| SE9604143D0 (sv) | 1996-11-13 | 1996-11-13 | Pacesetter Ab | Implanterbar elektrodkabel |
| US5741316A (en) | 1996-12-02 | 1998-04-21 | Light Sciences Limited Partnership | Electromagnetic coil configurations for power transmission through tissue |
| US5735887A (en) | 1996-12-10 | 1998-04-07 | Exonix Corporation | Closed-loop, RF-coupled implanted medical device |
| JP3954177B2 (ja) | 1997-01-29 | 2007-08-08 | 日本碍子株式会社 | 金属部材とセラミックス部材との接合構造およびその製造方法 |
| US7114502B2 (en) | 1997-02-26 | 2006-10-03 | Alfred E. Mann Foundation For Scientific Research | Battery-powered patient implantable device |
| US8555894B2 (en) | 1997-02-26 | 2013-10-15 | Alfred E. Mann Foundation For Scientific Research | System for monitoring temperature |
| US7460911B2 (en) | 1997-02-26 | 2008-12-02 | Alfred E. Mann Foundation For Scientific Research | System and method suitable for treatment of a patient with a neurological deficit by sequentially stimulating neural pathways using a system of discrete implantable medical devices |
| US6164284A (en) | 1997-02-26 | 2000-12-26 | Schulman; Joseph H. | System of implantable devices for monitoring and/or affecting body parameters |
| AU6667698A (en) | 1997-02-26 | 1998-09-18 | Alfred E. Mann Foundation For Scientific Research | Battery-powered patient implantable device |
| US6208894B1 (en) | 1997-02-26 | 2001-03-27 | Alfred E. Mann Foundation For Scientific Research And Advanced Bionics | System of implantable devices for monitoring and/or affecting body parameters |
| US8684009B2 (en) | 1997-02-26 | 2014-04-01 | Alfred E. Mann Foundation For Scientific Research | System for determining relative distance(s) and/or angle(s) between at least two points |
| US5957965A (en) | 1997-03-03 | 1999-09-28 | Medtronic, Inc. | Sacral medical electrical lead |
| CA2230930A1 (en) | 1997-04-25 | 1998-10-25 | Dale Gathergood | Exterior rear view mirror integral warning light |
| US5871513A (en) | 1997-04-30 | 1999-02-16 | Medtronic Inc. | Centerless ground feedthrough pin for an electrical power source in an implantable medical device |
| US6191365B1 (en) | 1997-05-02 | 2001-02-20 | General Science And Technology Corp | Medical devices incorporating at least one element made from a plurality of twisted and drawn wires |
| US5871532A (en) | 1997-05-22 | 1999-02-16 | Sulzer Intermedics Inc. | Epicardial lead for minimally invasive implantation |
| IT1292016B1 (it) | 1997-05-28 | 1999-01-25 | Valerio Cigaina | Dispositivo di impianto particolarmente per elettrostimolazione e/o elettroregistrazione di visceri endoaddominali |
| ATE277672T1 (de) | 1997-08-01 | 2004-10-15 | Mann Alfred E Found Scient Res | Implantierbare einrichtung mit verbesserter anordnung zur ladung der batterie und zur energiezufuhr |
| US5991665A (en) | 1997-09-18 | 1999-11-23 | Sulzer Intermedics Inc. | Self-cooling transcutaneous energy transfer system for battery powered implantable device |
| US6138681A (en) | 1997-10-13 | 2000-10-31 | Light Sciences Limited Partnership | Alignment of external medical device relative to implanted medical device |
| US6427086B1 (en) | 1997-10-27 | 2002-07-30 | Neuropace, Inc. | Means and method for the intracranial placement of a neurostimulator |
| WO1999027603A1 (en) | 1997-11-20 | 1999-06-03 | Seiko Epson Corporation | Electronic device |
| US6306100B1 (en) | 1997-12-16 | 2001-10-23 | Richard L. Prass | Intraoperative neurophysiological monitoring system |
| US6169387B1 (en) | 1997-12-22 | 2001-01-02 | Lifecor, Inc. | Battery management apparatus for portable electronic devices |
| DE19800416C2 (de) | 1998-01-08 | 2002-09-19 | Storz Karl Gmbh & Co Kg | Vorrichtung zur Behandlung von Körpergewebe, insbesondere von oberflächennahem Weichgewebe, mittels Ultraschall |
| US6081097A (en) | 1998-01-19 | 2000-06-27 | Matsushita Electric Industrial Co., Ltd. | Method for charging lithium secondary battery |
| US6305381B1 (en) | 1998-02-02 | 2001-10-23 | Medtronic Inc. | System for locating implantable medical device |
| EP1056510B1 (en) | 1998-02-23 | 2005-05-11 | Medtronic, Inc. | Rf coupled, implantable medical device with rechargeable back-up power source |
| US5902331A (en) | 1998-03-10 | 1999-05-11 | Medtronic, Inc. | Arrangement for implanting an endocardial cardiac lead |
| US6316909B1 (en) | 1998-03-24 | 2001-11-13 | Seiko Epson Corporation | Electronic device, control method for electronic device, recharge-rate estimating method for secondary battery, and charging control method for secondary battery |
| US6402793B1 (en) | 1998-04-03 | 2002-06-11 | Medtronic, Inc. | Implantable medical device having flat electrolytic capacitor with cathode/case electrical connections |
| US6221513B1 (en) | 1998-05-12 | 2001-04-24 | Pacific Coast Technologies, Inc. | Methods for hermetically sealing ceramic to metallic surfaces and assemblies incorporating such seals |
| US6243608B1 (en) | 1998-06-12 | 2001-06-05 | Intermedics Inc. | Implantable device with optical telemetry |
| US6735474B1 (en) | 1998-07-06 | 2004-05-11 | Advanced Bionics Corporation | Implantable stimulator system and method for treatment of incontinence and pain |
| US6941171B2 (en) | 1998-07-06 | 2005-09-06 | Advanced Bionics Corporation | Implantable stimulator methods for treatment of incontinence and pain |
| US6027456A (en) | 1998-07-10 | 2000-02-22 | Advanced Neuromodulation Systems, Inc. | Apparatus and method for positioning spinal cord stimulation leads |
| US6104960A (en) | 1998-07-13 | 2000-08-15 | Medtronic, Inc. | System and method for providing medical electrical stimulation to a portion of the nervous system |
| US6178353B1 (en) | 1998-07-27 | 2001-01-23 | Advanced Bionics Corporation | Laminated magnet keeper for implant device |
| US7231254B2 (en) | 1998-08-05 | 2007-06-12 | Bioneuronics Corporation | Closed-loop feedback-driven neuromodulation |
| US6083247A (en) | 1998-08-10 | 2000-07-04 | Medtronic, Inc. | Perpendicular atrial fixation/stimulation loop |
| US6104957A (en) | 1998-08-21 | 2000-08-15 | Alo; Kenneth M. | Epidural nerve root stimulation with lead placement method |
| DE19838137A1 (de) | 1998-08-21 | 2000-03-02 | Implex Hear Tech Ag | Vorrichtung und Verfahren zum Laden von wiederaufladbaren Akkumulatoren von Implantaten |
| US6212431B1 (en) | 1998-09-08 | 2001-04-03 | Advanced Bionics Corporation | Power transfer circuit for implanted devices |
| US7142925B1 (en) | 1998-09-16 | 2006-11-28 | Axon Engineering, Inc. | Combined stimulation of ventral and dorsal sacral roots for control of bladder function |
| IL127481A (en) * | 1998-10-06 | 2004-05-12 | Bio Control Medical Ltd | Urine excretion prevention device |
| DE69935727T2 (de) | 1998-10-06 | 2007-12-27 | Bio Control Medical, Ltd. | Vorrichtung zur behandlung von inkontinenz |
| US6208895B1 (en) | 1998-10-13 | 2001-03-27 | Physio-Control Manufacturing Corporation | Circuit for performing external pacing and biphasic defibrillation |
| US5948006A (en) | 1998-10-14 | 1999-09-07 | Advanced Bionics Corporation | Transcutaneous transmission patch |
| US6275737B1 (en) | 1998-10-14 | 2001-08-14 | Advanced Bionics Corporation | Transcutaneous transmission pouch |
| US7076307B2 (en) | 2002-05-09 | 2006-07-11 | Boveja Birinder R | Method and system for modulating the vagus nerve (10th cranial nerve) with electrical pulses using implanted and external components, to provide therapy neurological and neuropsychiatric disorders |
| US20030212440A1 (en) | 2002-05-09 | 2003-11-13 | Boveja Birinder R. | Method and system for modulating the vagus nerve (10th cranial nerve) using modulated electrical pulses with an inductively coupled stimulation system |
| WO2000025859A1 (en) | 1998-10-30 | 2000-05-11 | Aalborg University | A method to control an overactive bladder |
| US5949632A (en) | 1998-12-01 | 1999-09-07 | Exonix Corporation | Power supply having means for extending the operating time of an implantable medical device |
| US6393325B1 (en) | 1999-01-07 | 2002-05-21 | Advanced Bionics Corporation | Directional programming for implantable electrode arrays |
| US6052624A (en) | 1999-01-07 | 2000-04-18 | Advanced Bionics Corporation | Directional programming for implantable electrode arrays |
| US7555346B1 (en) | 1999-01-07 | 2009-06-30 | Boston Scientific Neuromodulation Corporation | Implantable pulse generator having current steering means |
| US6321118B1 (en) | 1999-01-28 | 2001-11-20 | Advanced Bionics Corporation | Method and apparatus for power link detection with implantable medical devices |
| AU772100B2 (en) | 1999-02-08 | 2004-04-08 | Cochlear Limited | Offset coils for radio frequency transcutaneous links |
| US6172556B1 (en) | 1999-03-04 | 2001-01-09 | Intersil Corporation, Inc. | Feedback-controlled low voltage current sink/source |
| US8666495B2 (en) | 1999-03-05 | 2014-03-04 | Metacure Limited | Gastrointestinal methods and apparatus for use in treating disorders and controlling blood sugar |
| US7131996B2 (en) | 1999-03-18 | 2006-11-07 | Helmut Wasserman | Artificial urinary diversion device |
| EP2275167A3 (en) | 1999-03-24 | 2014-04-30 | Second Sight Medical Products, Inc. | Visual prosthesis |
| WO2000056677A1 (en) | 1999-03-24 | 2000-09-28 | Alfred E. Mann Foundation | Method and apparatus of a strong metal-ceramic braze bond |
| US6166518A (en) | 1999-04-26 | 2000-12-26 | Exonix Corporation | Implantable power management system |
| US6181105B1 (en) | 1999-04-26 | 2001-01-30 | Exonix Corporation | Self contained transportable power source maintenance and charge |
| US6055456A (en) | 1999-04-29 | 2000-04-25 | Medtronic, Inc. | Single and multi-polar implantable lead for sacral nerve electrical stimulation |
| US6212430B1 (en) | 1999-05-03 | 2001-04-03 | Abiomed, Inc. | Electromagnetic field source with detection of position of secondary coil in relation to multiple primary coils |
| US6505075B1 (en) | 1999-05-29 | 2003-01-07 | Richard L. Weiner | Peripheral nerve stimulation method |
| US6516227B1 (en) | 1999-07-27 | 2003-02-04 | Advanced Bionics Corporation | Rechargeable spinal cord stimulator system |
| US7177690B2 (en) | 1999-07-27 | 2007-02-13 | Advanced Bionics Corporation | Implantable system having rechargeable battery indicator |
| US7295878B1 (en) | 1999-07-30 | 2007-11-13 | Advanced Bionics Corporation | Implantable devices using rechargeable zero-volt technology lithium-ion batteries |
| US6553263B1 (en) | 1999-07-30 | 2003-04-22 | Advanced Bionics Corporation | Implantable pulse generators using rechargeable zero-volt technology lithium-ion batteries |
| US7047082B1 (en) | 1999-09-16 | 2006-05-16 | Micronet Medical, Inc. | Neurostimulating lead |
| US6654642B2 (en) | 1999-09-29 | 2003-11-25 | Medtronic, Inc. | Patient interactive neurostimulation system and method |
| US6381496B1 (en) | 1999-10-01 | 2002-04-30 | Advanced Bionics Corporation | Parameter context switching for an implanted device |
| US6442434B1 (en) | 1999-10-19 | 2002-08-27 | Abiomed, Inc. | Methods and apparatus for providing a sufficiently stable power to a load in an energy transfer system |
| US6466817B1 (en) | 1999-11-24 | 2002-10-15 | Nuvasive, Inc. | Nerve proximity and status detection system and method |
| WO2001037728A1 (en) | 1999-11-24 | 2001-05-31 | Nuvasive, Inc. | Electromyography system |
| US6438423B1 (en) | 2000-01-20 | 2002-08-20 | Electrocore Technique, Llc | Method of treating complex regional pain syndromes by electrical stimulation of the sympathetic nerve chain |
| US6473652B1 (en) | 2000-03-22 | 2002-10-29 | Nac Technologies Inc. | Method and apparatus for locating implanted receiver and feedback regulation between subcutaneous and external coils |
| US6662051B1 (en) | 2000-03-31 | 2003-12-09 | Stephen A. Eraker | Programmable pain reduction device |
| JP2003530960A (ja) | 2000-04-20 | 2003-10-21 | コックレア リミティド | 人工内耳経皮電力最適化回路 |
| US7167756B1 (en) | 2000-04-28 | 2007-01-23 | Medtronic, Inc. | Battery recharge management for an implantable medical device |
| US6453198B1 (en) | 2000-04-28 | 2002-09-17 | Medtronic, Inc. | Power management for an implantable medical device |
| US6327504B1 (en) | 2000-05-10 | 2001-12-04 | Thoratec Corporation | Transcutaneous energy transfer with circuitry arranged to avoid overheating |
| US6505077B1 (en) | 2000-06-19 | 2003-01-07 | Medtronic, Inc. | Implantable medical device with external recharging coil electrical connection |
| US6456220B1 (en) | 2000-06-19 | 2002-09-24 | Cygnal Integrated Products, Inc. | Analog-to-digital converter for processing differential and single-ended inputs |
| US7305268B2 (en) | 2000-07-13 | 2007-12-04 | Northstar Neurscience, Inc. | Systems and methods for automatically optimizing stimulus parameters and electrode configurations for neuro-stimulators |
| ES2318238T3 (es) | 2000-07-26 | 2009-05-01 | Boston Scientific Neuromodulation Corporation | Sistema de estimulacion. |
| IT1316598B1 (it) | 2000-08-07 | 2003-04-24 | Caen Microelettronica E Sistem | Manufatto tessile con fibre illuminate, capo di abbigliamento daquesto ottenuto e metodo di produzione del manufatto. |
| US6510347B2 (en) | 2000-08-17 | 2003-01-21 | William N. Borkan | Spinal cord stimulation leads |
| US7054689B1 (en) | 2000-08-18 | 2006-05-30 | Advanced Bionics Corporation | Fully implantable neurostimulator for autonomic nerve fiber stimulation as a therapy for urinary and bowel dysfunction |
| DE10041727C2 (de) | 2000-08-25 | 2003-04-10 | Cochlear Ltd | Implantierbares hermetisch dichtes Gehäuse für eine implantierbare medizinische Vorrichtung |
| DE10041728A1 (de) | 2000-08-25 | 2002-03-21 | Implex Hear Tech Ag | Implantierbare medizinische Vorrichtung mit einem hermetisch dichten Gehäuse |
| US6591138B1 (en) | 2000-08-31 | 2003-07-08 | Neuropace, Inc. | Low frequency neurostimulator for the treatment of neurological disorders |
| US6864755B2 (en) | 2000-10-06 | 2005-03-08 | Alfred E. Mann Institute For Biomedical Engineering At The University Of Southern California | Switched reactance modulated E-class oscillator design |
| US6745077B1 (en) | 2000-10-11 | 2004-06-01 | Advanced Bionics Corporation | Electronic impedance transformer for inductively-coupled load stabilization |
| US6542846B1 (en) | 2000-11-09 | 2003-04-01 | Koninklijke Philips Electronics N.V. | Thermal management system for a portable ultrasound imaging device |
| US6847849B2 (en) | 2000-11-15 | 2005-01-25 | Medtronic, Inc. | Minimally invasive apparatus for implanting a sacral stimulation lead |
| US6971393B1 (en) | 2000-11-15 | 2005-12-06 | George Mamo | Minimally invasive method for implanting a sacral stimulation lead |
| US6600954B2 (en) | 2001-01-25 | 2003-07-29 | Biocontrol Medical Bcm Ltd. | Method and apparatus for selective control of nerve fibers |
| US6609945B2 (en) | 2001-02-08 | 2003-08-26 | Plexus, Inc. | Radio-controlled toy blimp with infrared beam weapons for staging a gun battle |
| US7069081B2 (en) | 2001-02-08 | 2006-06-27 | Wilson Greatbatch Ltd. | One piece header assembly for an implantable medical device |
| US6901287B2 (en) | 2001-02-09 | 2005-05-31 | Medtronic, Inc. | Implantable therapy delivery element adjustable anchor |
| US6597953B2 (en) | 2001-02-20 | 2003-07-22 | Neuropace, Inc. | Furcated sensing and stimulation lead |
| US6708065B2 (en) | 2001-03-02 | 2004-03-16 | Cardiac Pacemakers, Inc. | Antenna for an implantable medical device |
| CA2442412A1 (en) | 2001-03-30 | 2002-10-10 | Case Western Reserve University | Systems and methods for selectively stimulating components in, on, or near the pudendal nerve or its branches to achieve selective physiologic responses |
| US6584355B2 (en) | 2001-04-10 | 2003-06-24 | Cardiac Pacemakers, Inc. | System and method for measuring battery current |
| US8145324B1 (en) | 2001-04-13 | 2012-03-27 | Greatbatch Ltd. | Implantable lead bandstop filter employing an inductive coil with parasitic capacitance to enhance MRI compatibility of active medical devices |
| US20070088416A1 (en) | 2001-04-13 | 2007-04-19 | Surgi-Vision, Inc. | Mri compatible medical leads |
| US6892098B2 (en) | 2001-04-26 | 2005-05-10 | Biocontrol Medical Ltd. | Nerve stimulation for treating spasticity, tremor, muscle weakness, and other motor disorders |
| KR100606307B1 (ko) | 2001-05-23 | 2006-07-28 | 안태영 | 인체 이식 기구용 무접촉식 동력 전달 장치 |
| US6521350B2 (en) | 2001-06-18 | 2003-02-18 | Alfred E. Mann Foundation For Scientific Research | Application and manufacturing method for a ceramic to metal seal |
| JP4295086B2 (ja) | 2001-07-11 | 2009-07-15 | ヌバシブ, インコーポレイテッド | 手術の間の神経近接度、神経の方向、および病理学を決定するシステムおよび方法 |
| JP2003047179A (ja) | 2001-07-26 | 2003-02-14 | Matsushita Electric Works Ltd | 非接触電力伝達装置 |
| US6456256B1 (en) | 2001-08-03 | 2002-09-24 | Cardiac Pacemakers, Inc. | Circumferential antenna for an implantable medical device |
| US7151914B2 (en) | 2001-08-21 | 2006-12-19 | Medtronic, Inc. | Transmitter system for wireless communication with implanted devices |
| US6999819B2 (en) | 2001-08-31 | 2006-02-14 | Medtronic, Inc. | Implantable medical electrical stimulation lead fixation method and apparatus |
| US7734355B2 (en) | 2001-08-31 | 2010-06-08 | Bio Control Medical (B.C.M.) Ltd. | Treatment of disorders by unidirectional nerve stimulation |
| EP1435828A4 (en) | 2001-09-25 | 2009-11-11 | Nuvasive Inc | SYSTEM AND METHODS FOR EVALUATIONS AND SURGICAL ACTS |
| AU2002334749A1 (en) | 2001-09-28 | 2003-04-07 | Northstar Neuroscience, Inc. | Methods and implantable apparatus for electrical therapy |
| US7187978B2 (en) | 2001-11-01 | 2007-03-06 | Medtronic, Inc. | Method and apparatus for programming an implantable medical device |
| US6894456B2 (en) | 2001-11-07 | 2005-05-17 | Quallion Llc | Implantable medical power module |
| US6721603B2 (en) | 2002-01-25 | 2004-04-13 | Cyberonics, Inc. | Nerve stimulation as a treatment for pain |
| US8494625B2 (en) | 2002-02-04 | 2013-07-23 | Cerephex Corporation | Methods and apparatus for electrical stimulation of tissues using signals that minimize the effects of tissue impedance |
| US7317948B1 (en) | 2002-02-12 | 2008-01-08 | Boston Scientific Scimed, Inc. | Neural stimulation system providing auto adjustment of stimulus output as a function of sensed impedance |
| JP3731881B2 (ja) | 2002-05-23 | 2006-01-05 | 有限会社ティーエム | 人工臓器用非侵襲式充電システム、並びにこのシステムに用いる蓄電装置、および給電装置 |
| US7860570B2 (en) | 2002-06-20 | 2010-12-28 | Boston Scientific Neuromodulation Corporation | Implantable microstimulators and methods for unidirectional propagation of action potentials |
| US7582058B1 (en) | 2002-06-26 | 2009-09-01 | Nuvasive, Inc. | Surgical access system and related methods |
| JP4411205B2 (ja) | 2002-06-28 | 2010-02-10 | ボストン サイエンティフィック ニューロモデュレイション コーポレイション | 内蔵型電源及び双方向性テレメトリ・システムを有する微小刺激装置 |
| US8386048B2 (en) | 2002-06-28 | 2013-02-26 | Boston Scientific Neuromodulation Corporation | Systems and methods for communicating with or providing power to an implantable stimulator |
| AU2002951217A0 (en) | 2002-09-04 | 2002-09-19 | Cochlear Limited | Method and apparatus for measurement of transmitter/receiver separation |
| US7328068B2 (en) | 2003-03-31 | 2008-02-05 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by means of electrical stimulation of the pudendal and associated nerves, and the optional delivery of drugs in association therewith |
| US7369894B2 (en) | 2002-09-06 | 2008-05-06 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by electrical stimulation of the sacral and/or pudendal nerves |
| US20040055610A1 (en) | 2002-09-25 | 2004-03-25 | Peter Forsell | Detection of implanted wireless energy receiving device |
| AU2002951738A0 (en) | 2002-09-30 | 2002-10-17 | Cochlear Limited | Feedthrough with extended conductive pathways |
| US7127298B1 (en) | 2002-10-18 | 2006-10-24 | Advanced Bionics Corporation | Switched-matrix output for multi-channel implantable stimulator |
| US7146219B2 (en) | 2002-10-31 | 2006-12-05 | Medtronic, Inc. | Applying filter information to identify combinations of electrodes |
| AU2003285078A1 (en) | 2002-10-31 | 2004-06-07 | Medtronic, Inc. | Distributed system for neurostimulation therapy programming |
| US7933655B2 (en) | 2002-10-31 | 2011-04-26 | Medtronic, Inc. | Neurostimulation therapy manipulation |
| CA2448376A1 (en) | 2002-11-05 | 2004-05-05 | Wilson Greatbatch Technologies, Inc. | One piece header assembly for an implantable medical device |
| US6990376B2 (en) | 2002-12-06 | 2006-01-24 | The Regents Of The University Of California | Methods and systems for selective control of bladder function |
| US7952349B2 (en) | 2002-12-09 | 2011-05-31 | Ferro Solutions, Inc. | Apparatus and method utilizing magnetic field |
| TR200202651A2 (tr) | 2002-12-12 | 2004-07-21 | Met�N�Tulgar | VücutÁdışındanÁdirekÁtedaviÁsinyaliÁtransferliÁÁbeyinÁpili |
| US6685638B1 (en) | 2002-12-23 | 2004-02-03 | Codman & Shurtleff, Inc. | Acoustic monitoring system |
| US7742821B1 (en) | 2003-06-11 | 2010-06-22 | Boston Scientific Neutomodulation Corporation | Remote control for implantable medical device |
| AU2004226596C1 (en) | 2003-04-02 | 2010-09-16 | Neurostream Technologies General Partnership | Implantable nerve signal sensing and stimulation device for treating foot drop and other neurological disorders |
| US9446229B2 (en) | 2003-04-08 | 2016-09-20 | Omar Omar-Pasha | Catheter |
| US7463928B2 (en) | 2003-04-25 | 2008-12-09 | Medtronic, Inc. | Identifying combinations of electrodes for neurostimulation therapy |
| US20050187590A1 (en) | 2003-05-11 | 2005-08-25 | Boveja Birinder R. | Method and system for providing therapy for autism by providing electrical pulses to the vagus nerve(s) |
| US7444184B2 (en) | 2003-05-11 | 2008-10-28 | Neuro And Cardial Technologies, Llc | Method and system for providing therapy for bulimia/eating disorders by providing electrical pulses to vagus nerve(s) |
| US7317947B2 (en) | 2003-05-16 | 2008-01-08 | Medtronic, Inc. | Headset recharger for cranially implantable medical devices |
| US20040267137A1 (en) | 2003-06-27 | 2004-12-30 | Michael Peszynski | Apparatus and method for IC-based ultrasound transducer temperature sensing |
| US7617002B2 (en) | 2003-09-15 | 2009-11-10 | Medtronic, Inc. | Selection of neurostimulator parameter configurations using decision trees |
| US7286880B2 (en) | 2003-10-02 | 2007-10-23 | Medtronic, Inc. | System and method for transcutaneous energy transfer achieving high efficiency |
| US7225032B2 (en) | 2003-10-02 | 2007-05-29 | Medtronic Inc. | External power source, charger and system for an implantable medical device having thermal characteristics and method therefore |
| US8140168B2 (en) | 2003-10-02 | 2012-03-20 | Medtronic, Inc. | External power source for an implantable medical device having an adjustable carrier frequency and system and method related therefore |
| US7515967B2 (en) | 2003-10-02 | 2009-04-07 | Medtronic, Inc. | Ambulatory energy transfer system for an implantable medical device and method therefore |
| US20050075696A1 (en) | 2003-10-02 | 2005-04-07 | Medtronic, Inc. | Inductively rechargeable external energy source, charger, system and method for a transcutaneous inductive charger for an implantable medical device |
| US8265770B2 (en) | 2003-10-02 | 2012-09-11 | Medtronic, Inc. | Driver circuitry switchable between energy transfer and telemetry for an implantable medical device |
| US7286881B2 (en) | 2003-10-02 | 2007-10-23 | Medtronic, Inc. | External power source having an adjustable magnetic core and method of use |
| US6989200B2 (en) | 2003-10-30 | 2006-01-24 | Alfred E. Mann Foundation For Scientific Research | Ceramic to noble metal braze and method of manufacture |
| US6986453B2 (en) | 2003-11-13 | 2006-01-17 | Alfred E. Mann Foundation For Scientific Research | Manufacturing method for a ceramic to metal seal |
| US8467875B2 (en) | 2004-02-12 | 2013-06-18 | Medtronic, Inc. | Stimulation of dorsal genital nerves to treat urologic dysfunctions |
| US7120499B2 (en) | 2004-02-12 | 2006-10-10 | Ndi Medical, Llc | Portable percutaneous assemblies, systems and methods for providing highly selective functional or therapeutic neuromuscular stimulation |
| US20080161874A1 (en) | 2004-02-12 | 2008-07-03 | Ndi Medical, Inc. | Systems and methods for a trial stage and/or long-term treatment of disorders of the body using neurostimulation |
| US8086318B2 (en) | 2004-02-12 | 2011-12-27 | Ndi Medical, Llc | Portable assemblies, systems, and methods for providing functional or therapeutic neurostimulation |
| US20080132969A1 (en) | 2004-02-12 | 2008-06-05 | Ndi Medical, Inc. | Systems and methods for bilateral stimulation of left and right branches of the dorsal genital nerves to treat urologic dysfunctions |
| JP2007531562A (ja) | 2004-02-12 | 2007-11-08 | エヌディーアイ メディカル, エルエルシー | 機能的または治療的神経筋刺激を提供するための携帯式アセンブリ、システムおよび方法 |
| WO2005082453A1 (en) | 2004-02-25 | 2005-09-09 | Advanced Neuromodulation Systems, Inc. | System and method for neurological stimulation of peripheral nerves to treat low back pain |
| US7738963B2 (en) | 2004-03-04 | 2010-06-15 | Advanced Neuromodulation Systems, Inc. | System and method for programming an implantable pulse generator |
| US7844343B2 (en) | 2004-03-30 | 2010-11-30 | Medtronic, Inc. | MRI-safe implantable medical device |
| US7212110B1 (en) | 2004-04-19 | 2007-05-01 | Advanced Neuromodulation Systems, Inc. | Implantable device and system and method for wireless communication |
| US7532936B2 (en) | 2004-04-20 | 2009-05-12 | Advanced Neuromodulation Systems, Inc. | Programmable switching device for implantable device |
| US7245972B2 (en) | 2004-04-29 | 2007-07-17 | Alfred E. Mann Foundation For Scientific Research | Electrical treatment to treat shoulder subluxation |
| US7359751B1 (en) | 2004-05-05 | 2008-04-15 | Advanced Neuromodulation Systems, Inc. | Clinician programmer for use with trial stimulator |
| US7539538B2 (en) | 2004-05-28 | 2009-05-26 | Boston Science Neuromodulation Corporation | Low power loss current digital-to-analog converter used in an implantable pulse generator |
| US7225028B2 (en) | 2004-05-28 | 2007-05-29 | Advanced Bionics Corporation | Dual cochlear/vestibular stimulator with control signals derived from motion and speech signals |
| US7450991B2 (en) | 2004-05-28 | 2008-11-11 | Advanced Neuromodulation Systems, Inc. | Systems and methods used to reserve a constant battery capacity |
| US9205255B2 (en) | 2004-06-10 | 2015-12-08 | Medtronic Urinary Solutions, Inc. | Implantable pulse generator systems and methods for providing functional and/or therapeutic stimulation of muscles and/or nerves and/or central nervous system tissue |
| US7283867B2 (en) | 2004-06-10 | 2007-10-16 | Ndi Medical, Llc | Implantable system and methods for acquisition and processing of electrical signals from muscles and/or nerves and/or central nervous system tissue |
| US9308382B2 (en) | 2004-06-10 | 2016-04-12 | Medtronic Urinary Solutions, Inc. | Implantable pulse generator systems and methods for providing functional and/or therapeutic stimulation of muscles and/or nerves and/or central nervous system tissue |
| US7761167B2 (en) | 2004-06-10 | 2010-07-20 | Medtronic Urinary Solutions, Inc. | Systems and methods for clinician control of stimulation systems |
| DE602005020965D1 (de) | 2004-07-20 | 2010-06-10 | Medtronic Inc | Lokalisierung eines implantierten objekts auf basis einer externen antennenladung |
| US7061780B2 (en) | 2004-09-09 | 2006-06-13 | System General Corp. | Switching control circuit with variable switching frequency for primary-side-controlled power converters |
| US7771838B1 (en) | 2004-10-12 | 2010-08-10 | Boston Scientific Neuromodulation Corporation | Hermetically bonding ceramic and titanium with a Ti-Pd braze interface |
| US7578819B2 (en) | 2005-05-16 | 2009-08-25 | Baxano, Inc. | Spinal access and neural localization |
| US8768452B2 (en) | 2005-02-23 | 2014-07-01 | Medtronic, Inc. | Implantable neurostimulator supporting trial and chronic modes |
| US7515965B2 (en) | 2005-02-23 | 2009-04-07 | Medtronic, Inc. | Implantable medical device providing adaptive neurostimulation therapy for incontinence |
| US8774912B2 (en) | 2005-02-23 | 2014-07-08 | Medtronic, Inc. | Implantable neurostimulator supporting trial and chronic modes |
| US20060200205A1 (en) | 2005-03-01 | 2006-09-07 | Haller Matthew I | Systems and methods for treating a patient with multiple stimulation therapies |
| US7979119B2 (en) | 2005-04-26 | 2011-07-12 | Boston Scientific Neuromodulation Corporation | Display graphics for use in stimulation therapies |
| US7406351B2 (en) | 2005-04-28 | 2008-07-29 | Medtronic, Inc. | Activity sensing for stimulator control |
| US7774069B2 (en) | 2005-04-29 | 2010-08-10 | Medtronic, Inc. | Alignment indication for transcutaneous energy transfer |
| WO2006124068A1 (en) | 2005-05-13 | 2006-11-23 | Ndi Medical, Llc | Systems for electrical stimulation of nerves in adipose tissue regions |
| US8244360B2 (en) | 2005-06-09 | 2012-08-14 | Medtronic, Inc. | Regional therapies for treatment of pain |
| ATE537870T1 (de) | 2005-06-09 | 2012-01-15 | Medtronic Inc | Periphere nervenfeldstimulation und rückenmarksstimulation |
| US8620436B2 (en) | 2005-07-08 | 2013-12-31 | Boston Scientific Neuromodulation Corporation | Current generation architecture for an implantable stimulator device having coarse and fine current control |
| KR100792311B1 (ko) | 2005-07-30 | 2008-01-07 | 엘에스전선 주식회사 | 충전전력 공급장치, 충전 장치, 배터리 장치, 무접점 충전 시스템 및 무접점 충전 방법 |
| US8175717B2 (en) | 2005-09-06 | 2012-05-08 | Boston Scientific Neuromodulation Corporation | Ultracapacitor powered implantable pulse generator with dedicated power supply |
| US7640059B2 (en) * | 2005-09-08 | 2009-12-29 | Medtronic, Inc. | External presentation of electrical stimulation parameters |
| US7551960B2 (en) | 2005-09-08 | 2009-06-23 | Medtronic, Inc. | External presentation of electrical stimulation parameters |
| US7650192B2 (en) | 2005-12-02 | 2010-01-19 | Medtronic, Inc. | Passive charge of implantable medical device utilizing external power source and method |
| US7444181B2 (en) | 2005-12-14 | 2008-10-28 | Boston Scientific Neuromodulation Corporation | Techniques for sensing and adjusting a compliance voltage in an implantable stimulator device |
| CN101495174A (zh) * | 2005-12-22 | 2009-07-29 | 德州系统大学董事会 | 用于测定患者的心功能的方法和装置 |
| US8706219B2 (en) | 2005-12-22 | 2014-04-22 | Board Of Regents, The University Of Texas System | Method and apparatus for monitoring an organ of a patient |
| US7720547B2 (en) | 2006-01-04 | 2010-05-18 | Kenergy, Inc. | Extracorporeal power supply with a wireless feedback system for an implanted medical device |
| US7809443B2 (en) | 2006-01-31 | 2010-10-05 | Medtronic, Inc. | Electrical stimulation to alleviate chronic pelvic pain |
| US8019423B2 (en) | 2006-02-17 | 2011-09-13 | Marc Possover | Laparoscopic implantation of neurostimulators |
| US7747330B2 (en) | 2006-03-09 | 2010-06-29 | Medtronic, Inc. | Global parameter adjustment for multiple stimulation programs |
| US8447402B1 (en) | 2006-03-31 | 2013-05-21 | Alfred E. Mann Foundation For Scientific Research | Zirconia to platinum assembly using a titanium connector |
| US7738965B2 (en) | 2006-04-28 | 2010-06-15 | Medtronic, Inc. | Holster for charging pectorally implanted medical devices |
| US7761166B2 (en) | 2006-04-28 | 2010-07-20 | Medtronic, Inc. | Electrical stimulation of iliohypogastric nerve to alleviate chronic pelvic pain |
| US8892214B2 (en) | 2006-04-28 | 2014-11-18 | Medtronic, Inc. | Multi-electrode peripheral nerve evaluation lead and related system and method of use |
| US8219202B2 (en) | 2006-04-28 | 2012-07-10 | Medtronic, Inc. | Electrical stimulation of ilioinguinal nerve to alleviate chronic pelvic pain |
| US7715920B2 (en) | 2006-04-28 | 2010-05-11 | Medtronic, Inc. | Tree-based electrical stimulator programming |
| US20070265675A1 (en) | 2006-05-09 | 2007-11-15 | Ams Research Corporation | Testing Efficacy of Therapeutic Mechanical or Electrical Nerve or Muscle Stimulation |
| US9480846B2 (en) | 2006-05-17 | 2016-11-01 | Medtronic Urinary Solutions, Inc. | Systems and methods for patient control of stimulation systems |
| EP2032203B1 (en) | 2006-06-05 | 2013-10-23 | AMS Research Corporation | Electrical muscle stimulation to treat fecal incontinence and/or pelvic prolapse |
| US20070282376A1 (en) | 2006-06-06 | 2007-12-06 | Shuros Allan C | Method and apparatus for neural stimulation via the lymphatic system |
| US8116862B2 (en) | 2006-06-08 | 2012-02-14 | Greatbatch Ltd. | Tank filters placed in series with the lead wires or circuits of active medical devices to enhance MRI compatibility |
| AU2007284422B2 (en) | 2006-08-18 | 2011-06-02 | Second Sight Medical Products, Inc. | Package for an implantable neural stimulation device |
| EP2069011B1 (en) * | 2006-10-06 | 2013-11-20 | Neurostream Technologies General Partnership | Implantable pulse generator |
| US7979126B2 (en) | 2006-10-18 | 2011-07-12 | Boston Scientific Neuromodulation Corporation | Orientation-independent implantable pulse generator |
| US20100076534A1 (en) | 2006-10-25 | 2010-03-25 | William Alan Mock | Malleable needle having a plurality of electrodes for facilitating implantation of stimulation lead and method of implanting an electrical stimulation lead |
| US9643004B2 (en) | 2006-10-31 | 2017-05-09 | Medtronic, Inc. | Implantable medical elongated member with adhesive elements |
| US20080103572A1 (en) | 2006-10-31 | 2008-05-01 | Medtronic, Inc. | Implantable medical lead with threaded fixation |
| US9713706B2 (en) | 2006-10-31 | 2017-07-25 | Medtronic, Inc. | Implantable medical elongated member including intermediate fixation |
| US9827415B2 (en) | 2006-11-09 | 2017-11-28 | Greatbatch Ltd. | Implantable lead having multi-planar spiral inductor filter |
| US7857819B2 (en) | 2006-11-30 | 2010-12-28 | Boston Scientific Neuromodulation Corporation | Implant tool for use with a microstimulator |
| US8010205B2 (en) | 2007-01-11 | 2011-08-30 | Boston Scientific Neuromodulation Corporation | Multiple telemetry and/or charging coil configurations for an implantable medical device system |
| JP5562648B2 (ja) | 2007-01-29 | 2014-07-30 | スパイナル・モデュレーション・インコーポレイテッド | 非縫合の先頭保持機構 |
| US9615744B2 (en) | 2007-01-31 | 2017-04-11 | Medtronic, Inc. | Chopper-stabilized instrumentation amplifier for impedance measurement |
| US7391257B1 (en) | 2007-01-31 | 2008-06-24 | Medtronic, Inc. | Chopper-stabilized instrumentation amplifier for impedance measurement |
| US8549015B2 (en) | 2007-05-01 | 2013-10-01 | Giancarlo Barolat | Method and system for distinguishing nociceptive pain from neuropathic pain |
| US7898823B2 (en) | 2007-05-08 | 2011-03-01 | Leadtrend Technology Corp. | Quasi-resonant fly-back converter without auxiliary winding |
| US7932696B2 (en) | 2007-05-14 | 2011-04-26 | Boston Scientific Neuromodulation Corporation | Charger alignment indicator with adjustable threshold |
| US9427573B2 (en) | 2007-07-10 | 2016-08-30 | Astora Women's Health, Llc | Deployable electrode lead anchor |
| US20090076580A1 (en) | 2007-09-13 | 2009-03-19 | Medtronic, Inc. | Medical electrical lead |
| US8290596B2 (en) | 2007-09-26 | 2012-10-16 | Medtronic, Inc. | Therapy program selection based on patient state |
| WO2009051965A1 (en) | 2007-10-14 | 2009-04-23 | Board Of Regents, The University Of Texas System | A wireless neural recording and stimulating system for pain management |
| ES2900264T3 (es) | 2007-10-16 | 2022-03-16 | Implantica Patent Ltd | Procedimiento de control de suministro de energía a un dispositivo médico implantable |
| US8121694B2 (en) * | 2007-10-16 | 2012-02-21 | Medtronic, Inc. | Therapy control based on a patient movement state |
| US8244367B2 (en) | 2007-10-26 | 2012-08-14 | Medtronic, Inc. | Closed loop long range recharging |
| US8362742B2 (en) | 2007-10-26 | 2013-01-29 | Medtronic, Inc. | Method and apparatus for dynamic adjustment of recharge parameters |
| NZ565234A (en) | 2008-01-18 | 2010-11-26 | Telemetry Res Ltd | Selectable resonant frequency transcutaneous energy transfer system |
| US9199075B1 (en) | 2008-02-07 | 2015-12-01 | Respicardia, Inc. | Transvascular medical lead |
| US8332040B1 (en) | 2008-03-10 | 2012-12-11 | Advanced Neuromodulation Systems, Inc. | External charging device for charging an implantable medical device and methods of regulating duty of cycle of an external charging device |
| EP2269200B1 (en) | 2008-03-20 | 2014-09-24 | Greatbatch Ltd. | Shielded three-terminal flat-through emi/energy dissipating filter |
| US8019443B2 (en) | 2008-04-01 | 2011-09-13 | Boston Scientific Neuromodulation Corporation | Anchoring units for leads of implantable electric stimulation systems and methods of making and using |
| US8509909B2 (en) | 2008-04-10 | 2013-08-13 | Medtronic, Inc. | Using telemetry coupling as a surrogate for recharger coupling |
| WO2009126872A2 (en) | 2008-04-11 | 2009-10-15 | Bal Seal Engineering | Connector cartridge stack for electrical transmission |
| US9238135B2 (en) | 2008-04-30 | 2016-01-19 | Medtronic, Inc. | Flagging of electrodes of an implantable medical device, controller, system and method therefore |
| US8314594B2 (en) | 2008-04-30 | 2012-11-20 | Medtronic, Inc. | Capacity fade adjusted charge level or recharge interval of a rechargeable power source of an implantable medical device, system and method |
| US8103360B2 (en) | 2008-05-09 | 2012-01-24 | Foster Arthur J | Medical lead coil conductor with spacer element |
| US7890182B2 (en) | 2008-05-15 | 2011-02-15 | Boston Scientific Neuromodulation Corporation | Current steering for an implantable stimulator device involving fractionalized stimulation pulses |
| EP2138203B1 (en) | 2008-06-26 | 2013-01-30 | Greatbatch Ltd. | Stimulation lead design |
| WO2010011721A1 (en) | 2008-07-24 | 2010-01-28 | Boston Scientific Neuromodulation Corporation | System and method for maintaining a distribution of currents in an electrode array using independent voltage sources |
| US20120119698A1 (en) | 2008-09-27 | 2012-05-17 | Aristeidis Karalis | Wireless energy transfer for vehicles |
| WO2010042057A1 (en) | 2008-10-10 | 2010-04-15 | Milux Holding S.A. | Charger for implant |
| WO2010042056A1 (en) | 2008-10-10 | 2010-04-15 | Milux Holding S.A. | Charger for implant with means for indicating alignment between charger and implant |
| JP5643764B2 (ja) | 2008-10-27 | 2014-12-17 | スパイナル・モデュレーション・インコーポレイテッドSpinal Modulation Inc. | 選択的刺激システムおよび医学的状態の信号パラメータ |
| US9987493B2 (en) | 2008-10-28 | 2018-06-05 | Medtronic, Inc. | Medical devices and methods for delivery of current-based electrical stimulation therapy |
| US8219196B2 (en) | 2008-10-31 | 2012-07-10 | Medtronic, Inc. | Determination of stimulation output capabilities throughout power source voltage range |
| US8311639B2 (en) | 2009-07-08 | 2012-11-13 | Nevro Corporation | Systems and methods for adjusting electrical therapy based on impedance changes |
| US8255057B2 (en) | 2009-01-29 | 2012-08-28 | Nevro Corporation | Systems and methods for producing asynchronous neural responses to treat pain and/or other patient conditions |
| US8538530B1 (en) | 2008-11-19 | 2013-09-17 | Advanced Bionics | Hermetically sealed feedthrough case |
| US20110278948A1 (en) | 2008-11-21 | 2011-11-17 | Milux Holdings SA | System for supplying energy |
| US9694188B2 (en) | 2008-11-26 | 2017-07-04 | Medtronic, Inc. | Automated verification of MRI compatibility of active implantable medical device |
| US8355797B2 (en) | 2009-02-10 | 2013-01-15 | Nevro Corporation | Systems and methods for delivering neural therapy correlated with patient status |
| JP5350016B2 (ja) | 2009-02-27 | 2013-11-27 | 国立大学法人 東京医科歯科大学 | 顎運動測定システム |
| US9561366B2 (en) | 2009-03-27 | 2017-02-07 | Medtronic, Inc. | Conditional electrical stimulation |
| US9764147B2 (en) | 2009-04-24 | 2017-09-19 | Medtronic, Inc. | Charge-based stimulation intensity programming with pulse amplitude and width adjusted according to a function |
| US9155885B2 (en) | 2009-04-24 | 2015-10-13 | Medtronic, Inc. | Incontinence therapy |
| US8214042B2 (en) | 2009-05-26 | 2012-07-03 | Boston Scientific Neuromodulation Corporation | Techniques for controlling charging of batteries in an external charger and an implantable medical device |
| US8498710B2 (en) | 2009-07-28 | 2013-07-30 | Nevro Corporation | Linked area parameter adjustment for spinal cord stimulation and associated systems and methods |
| US9468755B2 (en) | 2009-09-30 | 2016-10-18 | Respicardia, Inc. | Medical lead with preformed bias |
| US8571677B2 (en) | 2009-10-21 | 2013-10-29 | Medtronic, Inc. | Programming techniques for stimulation with utilization of case electrode |
| US8457756B2 (en) | 2009-11-11 | 2013-06-04 | Boston Scientific Neuromodulation Corporation | Using the case of an implantable medical device to broaden communication bandwidth |
| US8577474B2 (en) | 2009-11-11 | 2013-11-05 | Boston Scientific Neuromodulation Corporation | Minimizing interference between charging and telemetry coils in an implantable medical device |
| US9123470B2 (en) | 2009-12-18 | 2015-09-01 | Cardiac Pacemakers, Inc. | Implantable energy storage device including a connection post to connect multiple electrodes |
| EP3636314B1 (en) | 2009-12-23 | 2021-09-08 | Setpoint Medical Corporation | Neural stimulation devices and systems for treatment of chronic inflammation |
| US20110184482A1 (en) | 2010-01-24 | 2011-07-28 | Kevin Wilmot Eberman | Non-rechargeable batteries and implantable medical devices |
| DE102010006837B4 (de) | 2010-02-03 | 2013-01-17 | Heraeus Precious Metals Gmbh & Co. Kg | (Meth)acrylsäurealkylester aufweisende elektrische Durchführung |
| AU2011224323B2 (en) | 2010-03-11 | 2016-06-23 | Mainstay Medical Limited | Modular stimulator for treatment of back pain, implantable RF ablation system and methods of use |
| US9314616B2 (en) | 2010-04-14 | 2016-04-19 | Medtronic, Inc. | Temporary implantable medical electrical leads |
| US9901284B2 (en) | 2010-04-16 | 2018-02-27 | Medtronic, Inc. | Coordination of functional MRI scanning and electrical stimulation therapy |
| US9020589B2 (en) | 2010-04-27 | 2015-04-28 | Medtronic, Inc. | Electrical stimulator with voltage mode emulation using regulated current |
| US9242104B2 (en) | 2010-05-11 | 2016-01-26 | Cardiac Pacemakers, Inc. | Systems for patient control of implantable medical device therapy |
| WO2011156286A2 (en) | 2010-06-07 | 2011-12-15 | Medtronic, Inc. | Stimulation therapy for bladder dysfunction |
| US8700165B2 (en) | 2010-07-16 | 2014-04-15 | Boston Scientific Neuromodulation Corporation | System and method for estimating lead configuration from neighboring relationship between electrodes |
| US9155891B2 (en) | 2010-09-20 | 2015-10-13 | Neuropace, Inc. | Current management system for a stimulation output stage of an implantable neurostimulation system |
| CA2813061C (en) * | 2010-10-13 | 2016-05-10 | Boston Scientific Neuromodulation Corporation | Architectures for an implantable medical device system having daisy-chained electrode-driver integrated circuits |
| US9867990B2 (en) | 2010-10-29 | 2018-01-16 | Medtronic, Inc. | Determination of dipole for tissue conductance communication |
| US9293741B1 (en) | 2010-12-29 | 2016-03-22 | Greatbatch Ltd. | Mechanical conditioning by bead blasting lithium iodine cell case |
| EP3685880B1 (en) | 2011-01-28 | 2021-03-24 | Stimwave Technologies Incorporated | Neural stimulator system |
| US9168374B2 (en) | 2011-01-28 | 2015-10-27 | Medtronic, Inc. | Intra-burst pulse variation for stimulation therapy |
| US9931514B2 (en) | 2013-06-30 | 2018-04-03 | Greatbatch Ltd. | Low impedance oxide resistant grounded capacitor for an AIMD |
| US9757558B2 (en) | 2011-03-01 | 2017-09-12 | Greatbatch Ltd. | RF filter for an active medical device (AMD) for handling high RF power induced in an associated implanted lead from an external RF field |
| US8543223B2 (en) | 2011-03-11 | 2013-09-24 | Greatbach Ltd. | Implantable lead with braided conductors |
| US9166321B2 (en) | 2011-03-22 | 2015-10-20 | Greatbatch Ltd. | Thin profile stacked layer contact |
| CN103608069B (zh) | 2011-03-24 | 2017-03-29 | 加利福尼亚理工学院 | 神经刺激器 |
| US9931513B2 (en) | 2011-03-29 | 2018-04-03 | Nuvectra Corporation | Feed-through connector assembly for implantable pulse generator and method of use |
| US9656076B2 (en) | 2011-04-07 | 2017-05-23 | Nuvectra Corporation | Arbitrary waveform generator and neural stimulation application with scalable waveform feature and charge balancing |
| US8738141B2 (en) | 2011-04-07 | 2014-05-27 | Greatbatch, Ltd. | Contact assembly for implantable pulse generator and method of use |
| US8996117B2 (en) | 2011-04-07 | 2015-03-31 | Greatbatch, Ltd. | Arbitrary waveform generator and neural stimulation application with scalable waveform feature |
| US9623257B2 (en) | 2011-04-18 | 2017-04-18 | Medtronic, Inc. | Recharge tuning techniques for an implantable device |
| US9136728B2 (en) | 2011-04-28 | 2015-09-15 | Medtronic, Inc. | Implantable medical devices and systems having inductive telemetry and recharge on a single coil |
| US9789307B2 (en) | 2011-04-29 | 2017-10-17 | Medtronic, Inc. | Dual prophylactic and abortive electrical stimulation |
| US8515545B2 (en) | 2011-04-29 | 2013-08-20 | Greatbatch Ltd. | Current steering neurostimulator device with unidirectional current sources |
| US9089712B2 (en) | 2011-04-29 | 2015-07-28 | Cyberonics, Inc. | Implantable medical device without antenna feedthrough |
| US9265958B2 (en) | 2011-04-29 | 2016-02-23 | Cyberonics, Inc. | Implantable medical device antenna |
| US9259582B2 (en) | 2011-04-29 | 2016-02-16 | Cyberonics, Inc. | Slot antenna for an implantable device |
| US10448889B2 (en) | 2011-04-29 | 2019-10-22 | Medtronic, Inc. | Determining nerve location relative to electrodes |
| US9240630B2 (en) | 2011-04-29 | 2016-01-19 | Cyberonics, Inc. | Antenna shield for an implantable medical device |
| US9592389B2 (en) | 2011-05-27 | 2017-03-14 | Boston Scientific Neuromodulation Corporation | Visualization of relevant stimulation leadwire electrodes relative to selected stimulation information |
| US9144680B2 (en) | 2011-05-31 | 2015-09-29 | Greatbatch Ltd. | System and method of establishing a protocol for providing electrical stimulation with a stimulation system to treat a patient |
| US9375574B2 (en) | 2011-05-31 | 2016-06-28 | Nuvectra Corporation | System and method of providing computer assisted stimulation programming (CASP) |
| US8954148B2 (en) | 2011-06-28 | 2015-02-10 | Greatbatch, Ltd. | Key fob controller for an implantable neurostimulator |
| US20130006330A1 (en) | 2011-06-28 | 2013-01-03 | Greatbatch, Ltd. | Dual patient controllers |
| US8571667B2 (en) | 2011-07-01 | 2013-10-29 | Greatbatch Ltd. | Active current control using the enclosure of an implanted pulse generator |
| US9399135B2 (en) | 2011-07-12 | 2016-07-26 | Astora Women's Health, Llc | Electronic stimulator device pulse generator circuit |
| US9675809B2 (en) | 2011-07-14 | 2017-06-13 | Cyberonics, Inc. | Circuit, system and method for far-field radiative powering of an implantable medical device |
| US9492678B2 (en) | 2011-07-14 | 2016-11-15 | Cyberonics, Inc. | Far field radiative powering of implantable medical therapy delivery devices |
| US8700175B2 (en) | 2011-07-19 | 2014-04-15 | Greatbatch Ltd. | Devices and methods for visually indicating the alignment of a transcutaneous energy transfer device over an implanted medical device |
| US9393433B2 (en) | 2011-07-20 | 2016-07-19 | Boston Scientific Neuromodulation Corporation | Battery management for an implantable medical device |
| US9517333B2 (en) | 2011-08-31 | 2016-12-13 | Nuvectra Corporation | Lead identification system |
| US9446254B2 (en) | 2011-10-13 | 2016-09-20 | Boston Scientific Neuromodulation Corporation | Charger alignment in an implantable medical device system employing reflected impedance modulation |
| ES2971060T3 (es) | 2011-11-04 | 2024-06-03 | Nevro Corp | Conjuntos de carga y comunicación para dispositivos médicos para el uso con generadores de señal implantables |
| US9814884B2 (en) | 2011-11-04 | 2017-11-14 | Nevro Corp. | Systems and methods for detecting faults and/or adjusting electrical therapy based on impedance changes |
| WO2013070490A1 (en) | 2011-11-10 | 2013-05-16 | Medtronic, Inc. | Introduction and anchoring tool for an implantable medical device element |
| WO2013082283A1 (en) | 2011-11-30 | 2013-06-06 | Medtronic, Inc. | Medical electrical stimulation lead including expandable coiled fixation element |
| US8971471B2 (en) | 2011-12-07 | 2015-03-03 | Imagine Communications Corp. | Predictable coding delay over communications networks |
| EP2628504A1 (en) | 2012-01-16 | 2013-08-21 | Greatbatch Ltd. | EMI filtered co-connected hermetic feedthrough, feedthrough capacitor and leadwire assembly for an active implantable medical device |
| US9889306B2 (en) | 2012-01-16 | 2018-02-13 | Greatbatch Ltd. | Hermetically sealed feedthrough with co-fired filled via and conductive insert for an active implantable medical device |
| US9270134B2 (en) | 2012-01-27 | 2016-02-23 | Medtronic, Inc. | Adaptive rate recharging system |
| US9981137B2 (en) | 2012-01-27 | 2018-05-29 | Nuvectra Corporation | Heat dispersion for implantable medical devices |
| US9974108B2 (en) | 2012-02-06 | 2018-05-15 | Nuvectra Corporation | Paired communication between an implanted medical device and an external control device |
| WO2013141844A1 (en) | 2012-03-20 | 2013-09-26 | International Truck Intellectual Property Company, Llc | Roof assembly |
| US9522282B2 (en) | 2012-03-29 | 2016-12-20 | Cyberonics, Inc. | Powering multiple implantable medical therapy delivery devices using far field radiative powering at multiple frequencies |
| US10086191B2 (en) | 2012-04-19 | 2018-10-02 | Medtronic, Inc. | Medical leads having a distal body and an openly coiled filar |
| US9653935B2 (en) | 2012-04-20 | 2017-05-16 | Medtronic, Inc. | Sensing temperature within medical devices |
| US9436481B2 (en) | 2012-04-23 | 2016-09-06 | Medtronic, Inc. | Restoration of medical device programming |
| EP2841151B1 (en) | 2012-04-26 | 2019-11-20 | Medtronic, Inc. | Trial stimulation systems |
| EP2841156A1 (en) | 2012-04-26 | 2015-03-04 | Medtronic, Inc. | Trial stimulation systems |
| US10369370B2 (en) | 2012-04-26 | 2019-08-06 | Medtronic, Inc. | Trial stimulation systems |
| US9149635B2 (en) | 2012-04-27 | 2015-10-06 | Medtronic, Inc. | Stimulation waveform generator for an implantable medical device |
| US9174051B2 (en) * | 2012-04-29 | 2015-11-03 | Boston Scientific Neuromodulation Corporation | Real time compliance voltage generation for an implantable stimulator |
| US9358039B2 (en) | 2012-05-08 | 2016-06-07 | Greatbatch Ltd. | Transseptal needle apparatus |
| WO2013169896A2 (en) | 2012-05-08 | 2013-11-14 | The Cleveland Clinic Foundation | Implantable pressure sensor |
| US10195419B2 (en) | 2012-06-13 | 2019-02-05 | Mainstay Medical Limited | Electrode leads for use with implantable neuromuscular electrical stimulator |
| US9833614B1 (en) | 2012-06-22 | 2017-12-05 | Nevro Corp. | Autonomic nervous system control via high frequency spinal cord modulation, and associated systems and methods |
| US9089693B2 (en) | 2012-06-29 | 2015-07-28 | Greatbatch Ltd. | Lead positioning and finned fixation system |
| US9427571B2 (en) | 2012-06-29 | 2016-08-30 | Nuvectra Corporation | Dynamic coil for implantable stimulation leads |
| WO2014036081A1 (en) | 2012-08-28 | 2014-03-06 | Boston Scientific Neuromodulation Corporation | Capture and visualization of clinical effects data in relation to a lead and/or locus of stimulation |
| US9259577B2 (en) | 2012-08-31 | 2016-02-16 | Greatbatch Ltd. | Method and system of quick neurostimulation electrode configuration and positioning |
| US9507912B2 (en) | 2012-08-31 | 2016-11-29 | Nuvectra Corporation | Method and system of simulating a pulse generator on a clinician programmer |
| US8761897B2 (en) | 2012-08-31 | 2014-06-24 | Greatbatch Ltd. | Method and system of graphical representation of lead connector block and implantable pulse generators on a clinician programmer |
| US9471753B2 (en) | 2012-08-31 | 2016-10-18 | Nuvectra Corporation | Programming and virtual reality representation of stimulation parameter Groups |
| US8903496B2 (en) | 2012-08-31 | 2014-12-02 | Greatbatch Ltd. | Clinician programming system and method |
| US9375582B2 (en) | 2012-08-31 | 2016-06-28 | Nuvectra Corporation | Touch screen safety controls for clinician programmer |
| US9767255B2 (en) | 2012-09-05 | 2017-09-19 | Nuvectra Corporation | Predefined input for clinician programmer data entry |
| US9209634B2 (en) | 2012-09-07 | 2015-12-08 | Greatbatch Ltd. | Method of improving battery recharge efficiency by statistical analysis |
| US9225190B2 (en) | 2012-09-07 | 2015-12-29 | Manufacturers And Traders Trust Company | Implant current controlled battery charging based on temperature |
| US9861812B2 (en) | 2012-12-06 | 2018-01-09 | Blue Wind Medical Ltd. | Delivery of implantable neurostimulators |
| WO2014089392A1 (en) | 2012-12-07 | 2014-06-12 | Medtronic, Inc. | Minimally invasive implantable neurostimulation system |
| US9308022B2 (en) | 2012-12-10 | 2016-04-12 | Nevro Corporation | Lead insertion devices and associated systems and methods |
| WO2014093178A2 (en) | 2012-12-14 | 2014-06-19 | Boston Scientific Neuromodulation Corporation | Method for automation of therapy-based programming in a tissue stimulator user interface |
| US9352148B2 (en) | 2013-02-27 | 2016-05-31 | Greatbatch Ltd. | Header block for an AIMD with an abandoned lead connector cavity |
| US20140275968A1 (en) | 2013-03-13 | 2014-09-18 | Greatbatch Ltd. | Surrogate implanted medical device for energy dissipation of existing implanted leads during mri scans |
| US9472916B2 (en) | 2013-03-14 | 2016-10-18 | Medtronic, Inc. | Distal connector assemblies for medical lead extensions |
| US9002447B2 (en) | 2013-03-14 | 2015-04-07 | Medtronic, Inc. | Implantable medical device having power supply for generating a regulated power supply |
| JP6298145B2 (ja) * | 2013-03-15 | 2018-03-20 | アルフレッド イー. マン ファウンデーション フォー サイエンティフィック リサーチ | 高速のターンオン時間をもつ電流検出複数出力電流刺激装置 |
| EP2991723A4 (en) | 2013-05-03 | 2017-02-01 | Nevro Corporation | Molded headers for implantable signal generators, and associated systems and methods |
| US9610449B2 (en) | 2013-05-16 | 2017-04-04 | Nuvectra Corporation | Method and apparatus for displaying a graphical impedance history for output channels of a lead |
| US9265935B2 (en) | 2013-06-28 | 2016-02-23 | Nevro Corporation | Neurological stimulation lead anchors and associated systems and methods |
| US20150018911A1 (en) | 2013-07-02 | 2015-01-15 | Greatbatch Ltd. | Apparatus, system, and method for minimized energy in peripheral field stimulation |
| JP6781865B2 (ja) | 2013-07-29 | 2020-11-11 | アルフレッド イー. マン ファウンデーション フォー サイエンティフィック リサーチ | マイクロプロセッサ制御されたeクラスドライバ |
| EP3038704B1 (en) * | 2013-08-29 | 2018-10-03 | Boston Scientific Neuromodulation Corporation | System for adjusting the compliance voltage in a neuromodulation device |
| US9068587B2 (en) | 2013-09-20 | 2015-06-30 | Greatbach Ltd. | Set screw apparatus |
| US9205258B2 (en) | 2013-11-04 | 2015-12-08 | ElectroCore, LLC | Nerve stimulator system |
| US9511230B2 (en) | 2013-11-08 | 2016-12-06 | Nuvectra Corporation | Implantable medical lead for stimulation of multiple nerves |
| US9502754B2 (en) | 2014-01-24 | 2016-11-22 | Medtronic, Inc. | Implantable medical devices having cofire ceramic modules and methods of fabricating the same |
| WO2015134327A2 (en) | 2014-03-03 | 2015-09-11 | Boston Scientific Neuromodulation Corporation | Electrical stimulation leads with multiple anchoring units and methods of making and using |
| US9457188B2 (en) | 2014-03-03 | 2016-10-04 | Medtronic, Inc. | Therapeutic window determination |
| US9757555B2 (en) | 2014-04-24 | 2017-09-12 | Medtronic, Inc. | Pre-molded sub-assemblies for implantable medical leads |
| US9981121B2 (en) | 2014-04-28 | 2018-05-29 | Medtronic, Inc. | Implantable medical devices, systems and components thereof |
| EP3137162A4 (en) | 2014-05-02 | 2018-01-24 | Nevro Corporation | Mri compatible medical devices |
| US9409029B2 (en) | 2014-05-12 | 2016-08-09 | Micron Devices Llc | Remote RF power system with low profile transmitting antenna |
| EP3145582B1 (en) | 2014-05-20 | 2020-10-21 | Nevro Corporation | Implanted pulse generators with reduced power consumption via signal strength/duration characteristics, and associated systems |
| US9775984B2 (en) | 2014-08-01 | 2017-10-03 | Nuvectra Corporation | Apparatus with unencapsulated reinforcement |
| US9924904B2 (en) | 2014-09-02 | 2018-03-27 | Medtronic, Inc. | Power-efficient chopper amplifier |
| AU2015336218B2 (en) | 2014-10-22 | 2020-07-23 | Nevro Corp. | Systems and methods for extending the life of an implanted pulse generator battery |
| US9597507B2 (en) | 2014-10-31 | 2017-03-21 | Medtronic, Inc. | Paired stimulation pulses based on sensed compound action potential |
| US9498628B2 (en) | 2014-11-21 | 2016-11-22 | Medtronic, Inc. | Electrode selection for electrical stimulation therapy |
| US10095837B2 (en) | 2014-11-21 | 2018-10-09 | Medtronic, Inc. | Real-time phase detection of frequency band |
| US9907955B2 (en) | 2014-11-25 | 2018-03-06 | Medtronic Bakken Research Center B.V. | Disturbing magnetic resonance imaging (MRI) images using implantable medical device |
| US10183162B2 (en) | 2015-01-02 | 2019-01-22 | Greatbatch Ltd. | Coiled, closed-loop RF current attenuator configured to be placed about an implantable lead conductor |
| US10277055B2 (en) | 2015-02-26 | 2019-04-30 | Greatbatch Ltd. | Battery wireless charging system |
| US10226636B2 (en) | 2015-04-24 | 2019-03-12 | Medtronic, Inc. | Managing recharge power for implantable medical devices |
| US10052490B2 (en) | 2015-06-09 | 2018-08-21 | Nuvectra Corporation | Systems, methods, and devices for performing electronically controlled test stimulation |
| US10076667B2 (en) | 2015-06-09 | 2018-09-18 | Nuvectra Corporation | System and method of performing computer assisted stimulation programming (CASP) with a non-zero starting value customized to a patient |
| US9872988B2 (en) | 2015-06-09 | 2018-01-23 | Nuvectra Corporation | Systems, methods, and devices for evaluating lead placement based on patient physiological responses |
| CA2991903C (en) | 2015-07-10 | 2023-10-17 | Axonics Modulation Technologies, Inc. | Implantable nerve stimulator having internal electronics without asic and methods of use |
| US9974949B2 (en) | 2015-10-16 | 2018-05-22 | Cyberonics, Inc. | MRI-safe implantable lead assembly |
| EP4079369A1 (en) | 2015-12-31 | 2022-10-26 | Nevro Corporation | Controller for nerve stimulation circuit and associated systems and methods |
| WO2017124082A1 (en) | 2016-01-15 | 2017-07-20 | Richard Lebaron | An implantable relay module |
| US10244956B2 (en) | 2016-02-12 | 2019-04-02 | Nuvectra Corporation | Stimulation needle apparatus and method |
| US10236709B2 (en) | 2016-05-05 | 2019-03-19 | Greatbatch Ltd. | Apparatus, system, and method for wireless charging of a device within a sterilizable vessel |
| US10109844B2 (en) | 2016-11-02 | 2018-10-23 | Greatbatch Ltd. | Dual weld plug for an electrochemical cell |
| US10493287B2 (en) | 2017-02-27 | 2019-12-03 | Medtronic, Inc. | Facilitating trusted pairing of an implantable device and an external device |
| AT520440B1 (de) | 2018-01-31 | 2019-04-15 | Ing Josef Scharmueller | Niederhalter |
-
2016
- 2016-07-08 CA CA2991903A patent/CA2991903C/en active Active
- 2016-07-08 EP EP16824936.5A patent/EP3319683A4/en active Pending
- 2016-07-08 CN CN201680040653.XA patent/CN107847731B/zh active Active
- 2016-07-08 AU AU2016291554A patent/AU2016291554B2/en active Active
- 2016-07-08 WO PCT/US2016/041508 patent/WO2017011305A1/en active Application Filing
- 2016-07-08 US US15/205,613 patent/US11766568B2/en active Active
- 2016-07-08 JP JP2018500525A patent/JP6946261B2/ja active Active
- 2016-07-08 CN CN201910475952.1A patent/CN110101968B/zh active Active
-
2017
- 2017-01-24 US US15/414,531 patent/US9925381B2/en active Active
-
2018
- 2018-02-06 US US15/889,844 patent/US10850104B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP3319683A4 (en) | 2018-12-26 |
| CA2991903C (en) | 2023-10-17 |
| US20170007836A1 (en) | 2017-01-12 |
| WO2017011305A1 (en) | 2017-01-19 |
| AU2016291554B2 (en) | 2021-01-07 |
| US9925381B2 (en) | 2018-03-27 |
| CA2991903A1 (en) | 2017-01-19 |
| US20180333581A1 (en) | 2018-11-22 |
| US10850104B2 (en) | 2020-12-01 |
| AU2016291554A1 (en) | 2018-01-18 |
| US20170128728A1 (en) | 2017-05-11 |
| CN107847731B (zh) | 2019-06-28 |
| CN107847731A (zh) | 2018-03-27 |
| EP3319683A1 (en) | 2018-05-16 |
| CN110101968A (zh) | 2019-08-09 |
| CN110101968B (zh) | 2023-09-08 |
| US11766568B2 (en) | 2023-09-26 |
| JP2018519127A (ja) | 2018-07-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6946261B2 (ja) | Asicを用いない内部電子機器を有する埋め込み可能神経刺激装置および方法 | |
| CN107073258B (zh) | 用于基于神经定位来进行神经刺激电极配置的系统和方法 | |
| CN106999709B (zh) | 用于与可植入神经刺激器一起使用的集成肌电图临床医生程控器 | |
| JP6791857B2 (ja) | 埋込可能神経刺激装置のための改良されたアンテナおよび使用方法 | |
| CN107073257B (zh) | 在用于治疗膀胱过度活动症的神经刺激系统中的肌电图引线定位和刺激滴定 | |
| US12083349B2 (en) | Methods and systems for frequency adjustment to optimize charging of implantable neurostimulator | |
| US20230080703A1 (en) | Low energy implantable devices and methods of use | |
| US20200289832A1 (en) | Charging device with off-center coil |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190705 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20190705 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20200529 |
|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20200617 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20200710 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20200727 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20201027 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210208 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210507 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20210820 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20210915 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6946261 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |