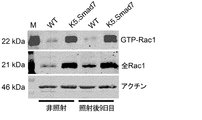
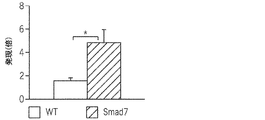
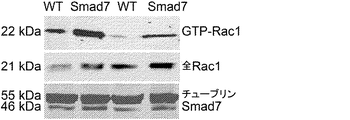
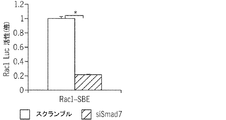
JP6576251B2 - PTD−Smad7薬物療法 - Google Patents
PTD−Smad7薬物療法 Download PDFInfo
- Publication number
- JP6576251B2 JP6576251B2 JP2015561737A JP2015561737A JP6576251B2 JP 6576251 B2 JP6576251 B2 JP 6576251B2 JP 2015561737 A JP2015561737 A JP 2015561737A JP 2015561737 A JP2015561737 A JP 2015561737A JP 6576251 B2 JP6576251 B2 JP 6576251B2
- Authority
- JP
- Japan
- Prior art keywords
- smad7
- protein
- tat
- oral
- cell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/18—Growth factors; Growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/162—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from virus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4702—Regulators; Modulating activity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4702—Regulators; Modulating activity
- C07K14/4703—Inhibitors; Suppressors
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/475—Growth factors; Growth regulators
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N7/00—Viruses; Bacteriophages; Compositions thereof; Preparation or purification thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/01—Fusion polypeptide containing a localisation/targetting motif
- C07K2319/10—Fusion polypeptide containing a localisation/targetting motif containing a tag for extracellular membrane crossing, e.g. TAT or VP22
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/20—Fusion polypeptide containing a tag with affinity for a non-protein ligand
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/20—Fusion polypeptide containing a tag with affinity for a non-protein ligand
- C07K2319/23—Fusion polypeptide containing a tag with affinity for a non-protein ligand containing a GST-tag
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2740/00—Reverse transcribing RNA viruses
- C12N2740/00011—Details
- C12N2740/10011—Retroviridae
- C12N2740/16011—Human Immunodeficiency Virus, HIV
- C12N2740/16311—Human Immunodeficiency Virus, HIV concerning HIV regulatory proteins
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2740/00—Reverse transcribing RNA viruses
- C12N2740/00011—Details
- C12N2740/10011—Retroviridae
- C12N2740/16011—Human Immunodeficiency Virus, HIV
- C12N2740/16311—Human Immunodeficiency Virus, HIV concerning HIV regulatory proteins
- C12N2740/16322—New viral proteins or individual genes, new structural or functional aspects of known viral proteins or genes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2800/00—Nucleic acids vectors
- C12N2800/22—Vectors comprising a coding region that has been codon optimised for expression in a respective host
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Gastroenterology & Hepatology (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Immunology (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Toxicology (AREA)
- Virology (AREA)
- Epidemiology (AREA)
- Wood Science & Technology (AREA)
- Dermatology (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- General Engineering & Computer Science (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361775252P | 2013-03-08 | 2013-03-08 | |
| US61/775,252 | 2013-03-08 | ||
| PCT/US2014/022052 WO2014138670A1 (en) | 2013-03-08 | 2014-03-07 | Ptd-smad7 therapeutics |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2016510596A JP2016510596A (ja) | 2016-04-11 |
| JP2016510596A5 JP2016510596A5 (enExample) | 2017-03-30 |
| JP6576251B2 true JP6576251B2 (ja) | 2019-09-18 |
Family
ID=51492023
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2015561737A Active JP6576251B2 (ja) | 2013-03-08 | 2014-03-07 | PTD−Smad7薬物療法 |
Country Status (8)
| Country | Link |
|---|---|
| US (5) | US10456448B2 (enExample) |
| EP (1) | EP2964774B1 (enExample) |
| JP (1) | JP6576251B2 (enExample) |
| CN (1) | CN105358704B (enExample) |
| CA (1) | CA2904329C (enExample) |
| IL (1) | IL241190B (enExample) |
| SG (1) | SG11201507045TA (enExample) |
| WO (1) | WO2014138670A1 (enExample) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3536337A1 (en) | 2010-09-22 | 2019-09-11 | The Regents of the University of Colorado, a body corporate | Therapeutic applications of smad7 |
| US10456448B2 (en) | 2013-03-08 | 2019-10-29 | The Regents Of The University Of Colorado, A Body Corporate | PTD-SMAD7 therapeutics |
| KR101853923B1 (ko) * | 2016-01-25 | 2018-05-02 | 연세대학교 산학협력단 | Smad 단백질을 포함하는 자가 면역 질환 치료용 조성물, Smad 단백질을 포함하는 융합 단백질, 이를 제조하기 위한 벡터, 및 이의 제조 방법 |
| US20200332302A1 (en) * | 2017-12-30 | 2020-10-22 | The Regents Of The University Of Colorado, A Body Corporate | Therapeutics of ptd-smad7 fusion proteins |
| JP7678483B2 (ja) * | 2021-03-10 | 2025-05-16 | 国立大学法人 熊本大学 | コロナウイルス感染を検出する方法 |
| WO2023044277A1 (en) * | 2021-09-15 | 2023-03-23 | The Regents Of The University Of Colorado, A Body Corporate | Smad7 polypeptide formulations |
| US20250269063A2 (en) * | 2022-02-07 | 2025-08-28 | Aavogen Inc. | Codon-optimized smad7 gene therapy to treat and prevent muscle wasting and to enhance muscle mass |
Family Cites Families (48)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL8200523A (nl) | 1982-02-11 | 1983-09-01 | Univ Leiden | Werkwijze voor het in vitro transformeren van planteprotoplasten met plasmide-dna. |
| US4879236A (en) | 1984-05-16 | 1989-11-07 | The Texas A&M University System | Method for producing a recombinant baculovirus expression vector |
| US4952500A (en) | 1988-02-01 | 1990-08-28 | University Of Georgia Research Foundation, Inc. | Cloning systems for Rhodococcus and related bacteria |
| US5703055A (en) | 1989-03-21 | 1997-12-30 | Wisconsin Alumni Research Foundation | Generation of antibodies through lipid mediated DNA delivery |
| US5302523A (en) | 1989-06-21 | 1994-04-12 | Zeneca Limited | Transformation of plant cells |
| US5550318A (en) | 1990-04-17 | 1996-08-27 | Dekalb Genetics Corporation | Methods and compositions for the production of stably transformed, fertile monocot plants and cells thereof |
| US7705215B1 (en) | 1990-04-17 | 2010-04-27 | Dekalb Genetics Corporation | Methods and compositions for the production of stably transformed, fertile monocot plants and cells thereof |
| US5322783A (en) | 1989-10-17 | 1994-06-21 | Pioneer Hi-Bred International, Inc. | Soybean transformation by microparticle bombardment |
| JPH05505102A (ja) | 1989-12-21 | 1993-08-05 | ホワイトヘツド・インスチチユート・フオー・バイオメデイカル・リサーチ | 真核細胞の中に分子を配達する方法 |
| US5484956A (en) | 1990-01-22 | 1996-01-16 | Dekalb Genetics Corporation | Fertile transgenic Zea mays plant comprising heterologous DNA encoding Bacillus thuringiensis endotoxin |
| US5670488A (en) | 1992-12-03 | 1997-09-23 | Genzyme Corporation | Adenovirus vector for gene therapy |
| US5384253A (en) | 1990-12-28 | 1995-01-24 | Dekalb Genetics Corporation | Genetic transformation of maize cells by electroporation of cells pretreated with pectin degrading enzymes |
| WO1993004169A1 (en) | 1991-08-20 | 1993-03-04 | Genpharm International, Inc. | Gene targeting in animal cells using isogenic dna constructs |
| US5610042A (en) | 1991-10-07 | 1997-03-11 | Ciba-Geigy Corporation | Methods for stable transformation of wheat |
| US5702932A (en) | 1992-07-20 | 1997-12-30 | University Of Florida | Microinjection methods to transform arthropods with exogenous DNA |
| JP2952041B2 (ja) | 1992-07-27 | 1999-09-20 | パイオニア ハイ−ブレッド インターナショナル,インコーポレイテッド | 培養ダイズ細胞のagrobacterium媒介形質転換の改良法 |
| ES2123062T3 (es) | 1992-08-21 | 1999-01-01 | Biogen Inc | Polipeptidos de transporte derivados de la proteina tat. |
| GB9222888D0 (en) | 1992-10-30 | 1992-12-16 | British Tech Group | Tomography |
| US5436222A (en) | 1993-03-15 | 1995-07-25 | The Research Foundation Of State University Of New York | Use of platelet factor 4 to treat inflammatory diseases |
| US5656610A (en) | 1994-06-21 | 1997-08-12 | University Of Southern California | Producing a protein in a mammal by injection of a DNA-sequence into the tongue |
| US5871986A (en) | 1994-09-23 | 1999-02-16 | The General Hospital Corporation | Use of a baculovirus to express and exogenous gene in a mammalian cell |
| US5736524A (en) | 1994-11-14 | 1998-04-07 | Merck & Co.,. Inc. | Polynucleotide tuberculosis vaccine |
| US5780448A (en) | 1995-11-07 | 1998-07-14 | Ottawa Civic Hospital Loeb Research | DNA-based vaccination of fish |
| US5945100A (en) | 1996-07-31 | 1999-08-31 | Fbp Corporation | Tumor delivery vehicles |
| US5981274A (en) | 1996-09-18 | 1999-11-09 | Tyrrell; D. Lorne J. | Recombinant hepatitis virus vectors |
| GB2320431B (en) * | 1996-12-20 | 2000-08-30 | Johnson & Johnson Medical | Compositions for the treatment of chronic wounds |
| WO1998053068A1 (en) * | 1997-05-20 | 1998-11-26 | Ludwig Institute For Cancer Research | Smad7 and uses thereof |
| US6891082B2 (en) | 1997-08-01 | 2005-05-10 | The Johns Hopkins University School Of Medicine | Transgenic non-human animals expressing a truncated activintype II receptor |
| US5994624A (en) | 1997-10-20 | 1999-11-30 | Cotton Incorporated | In planta method for the production of transgenic plants |
| AU3365299A (en) | 1998-03-27 | 1999-10-18 | Eli Lilly And Company | Treatment and prevention of vascular disease |
| US8466134B1 (en) | 1998-06-26 | 2013-06-18 | Athena Neurosciences, Inc. | Aqueous compositions containing corticosteroids for nasal and pulmonary delivery |
| US6998240B2 (en) | 2001-06-11 | 2006-02-14 | Wisconsin Alumni Research Foundation | Screen for selective inhibitors or activators of Smad protein function |
| AUPR638101A0 (en) | 2001-07-13 | 2001-08-09 | Bioa Pty Limited | Composition and method for treatment of disease |
| CA2496882C (en) | 2002-09-06 | 2011-11-01 | 3M Innovative Properties Company | Metering valve for a metered dose inhaler providing consistent delivery |
| KR100472938B1 (ko) | 2002-10-31 | 2005-03-11 | 학교법인 한림대학교 | 세포침투 효율을 향상시킨 수송도메인-목표단백질-수송도메인 융합단백질 및 그 용도 |
| KR20070030164A (ko) | 2003-10-16 | 2007-03-15 | 더 유니버시티 코트, 더 유니버시티 오브 에딘버러 | Es 세포 자가 재생 및 계통 지정의 조절, 및 이를 위한배지 |
| US9757461B2 (en) | 2005-01-14 | 2017-09-12 | Camurus Ab | GnRH analogue formulations |
| MX2007012344A (es) * | 2005-04-05 | 2007-12-13 | Pioneer Hi Bred Int | Metodos y composiciones para disenar moleculas de acido nucleico para la expresion de polipeptido en plantas usando la desviacion de codon de virus de planta. |
| CA2999420A1 (en) | 2005-09-27 | 2007-04-05 | Tissuetech, Inc. | Amniotic membrane preparations and purified compositions and methods of use |
| WO2008105925A2 (en) | 2006-09-01 | 2008-09-04 | Oregon Health & Science University | COMPOUNDS AND METHODS FOR MODULATING β-CATENIN/SMAD-7 INTERACTIONS |
| US20080114287A1 (en) | 2006-11-14 | 2008-05-15 | Kar Neng Lai | Ultrasound Microbubble Mediated Genes Delivery System |
| MX2010010165A (es) | 2008-03-17 | 2010-11-25 | Scripps Research Inst | Procedimientos quimicos y geneticos combinados para generacion de celulas madre pluripotentes inducidas. |
| US8591961B2 (en) | 2008-09-04 | 2013-11-26 | Allergan, Inc. | Aesthetic treatment of scars and aging skin |
| WO2009151600A2 (en) | 2008-06-10 | 2009-12-17 | Tufts University | Smad proteins control drosha-mediated mirna maturation |
| US20100184033A1 (en) | 2008-07-16 | 2010-07-22 | West Michael D | Methods to accelerate the isolation of novel cell strains from pluripotent stem cells and cells obtained thereby |
| EP3536337A1 (en) | 2010-09-22 | 2019-09-11 | The Regents of the University of Colorado, a body corporate | Therapeutic applications of smad7 |
| WO2013151665A2 (en) * | 2012-04-02 | 2013-10-10 | modeRNA Therapeutics | Modified polynucleotides for the production of proteins associated with human disease |
| US10456448B2 (en) | 2013-03-08 | 2019-10-29 | The Regents Of The University Of Colorado, A Body Corporate | PTD-SMAD7 therapeutics |
-
2014
- 2014-03-07 US US14/773,167 patent/US10456448B2/en active Active
- 2014-03-07 SG SG11201507045TA patent/SG11201507045TA/en unknown
- 2014-03-07 CA CA2904329A patent/CA2904329C/en active Active
- 2014-03-07 CN CN201480025596.9A patent/CN105358704B/zh active Active
- 2014-03-07 WO PCT/US2014/022052 patent/WO2014138670A1/en not_active Ceased
- 2014-03-07 US US14/201,488 patent/US9422352B2/en active Active
- 2014-03-07 EP EP14761076.0A patent/EP2964774B1/en active Active
- 2014-03-07 JP JP2015561737A patent/JP6576251B2/ja active Active
-
2015
- 2015-09-06 IL IL241190A patent/IL241190B/en active IP Right Grant
-
2016
- 2016-07-12 US US15/208,424 patent/US20170000851A1/en not_active Abandoned
-
2019
- 2019-09-26 US US16/584,608 patent/US20200023037A1/en not_active Abandoned
-
2022
- 2022-04-22 US US17/727,579 patent/US20220370560A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| IL241190A0 (en) | 2015-11-30 |
| US20170000851A1 (en) | 2017-01-05 |
| US9422352B2 (en) | 2016-08-23 |
| IL241190B (en) | 2019-09-26 |
| HK1219986A1 (zh) | 2017-04-21 |
| EP2964774A4 (en) | 2016-08-10 |
| US20200023037A1 (en) | 2020-01-23 |
| EP2964774A1 (en) | 2016-01-13 |
| EP2964774B1 (en) | 2020-05-06 |
| US20220370560A1 (en) | 2022-11-24 |
| CN105358704B (zh) | 2020-01-03 |
| US20160039894A1 (en) | 2016-02-11 |
| SG11201507045TA (en) | 2015-10-29 |
| CN105358704A (zh) | 2016-02-24 |
| CA2904329A1 (en) | 2014-09-12 |
| US20140288006A1 (en) | 2014-09-25 |
| US10456448B2 (en) | 2019-10-29 |
| CA2904329C (en) | 2021-11-30 |
| WO2014138670A1 (en) | 2014-09-12 |
| JP2016510596A (ja) | 2016-04-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6576251B2 (ja) | PTD−Smad7薬物療法 | |
| ES2281529T3 (es) | Peptidos eficaces en el tratamiento de tumores y otras afecciones que requieren la eliminacion o destruccion de celulas. | |
| US10350265B2 (en) | Therapeutic applications of Smad7 | |
| KR20130043242A (ko) | Sparc 혈관형성 영역과 사용방법 | |
| US20130101664A1 (en) | Muc1 ligand traps for use in treating cancers | |
| CN109937045A (zh) | 局部缺血/再灌注损伤 | |
| US20170298108A1 (en) | Compositions comprising grim-19 therapeutics and methods of use | |
| JP4252446B2 (ja) | Abinにより媒介される肝炎防護 | |
| US20200332302A1 (en) | Therapeutics of ptd-smad7 fusion proteins | |
| CN115605218A (zh) | 用于治疗大疱性表皮松解症的归巢肽引导的核心蛋白聚糖缀合物 | |
| HK1219986B (zh) | Ptd-smad7治疗剂 | |
| HK1223294B (en) | Therapeutic applications of smad7 | |
| CN121175330A (zh) | 溶瘤肽 | |
| JP2021534826A (ja) | がんの処置のためのペプチド治療薬およびその使用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20151130 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170223 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20170223 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20171228 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20171220 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180328 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20180730 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20181019 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20190304 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190621 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20190621 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20190710 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190731 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190820 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6576251 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: R3D03 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |