JP6067567B2 - カテーテル組立体 - Google Patents
カテーテル組立体 Download PDFInfo
- Publication number
- JP6067567B2 JP6067567B2 JP2013536153A JP2013536153A JP6067567B2 JP 6067567 B2 JP6067567 B2 JP 6067567B2 JP 2013536153 A JP2013536153 A JP 2013536153A JP 2013536153 A JP2013536153 A JP 2013536153A JP 6067567 B2 JP6067567 B2 JP 6067567B2
- Authority
- JP
- Japan
- Prior art keywords
- catheter assembly
- outer needle
- proximal end
- plug
- needle hub
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000003780 insertion Methods 0.000 claims description 31
- 230000037431 insertion Effects 0.000 claims description 31
- 230000000903 blocking effect Effects 0.000 claims description 23
- 239000012528 membrane Substances 0.000 claims description 20
- 239000012530 fluid Substances 0.000 claims description 9
- 239000007788 liquid Substances 0.000 claims description 5
- 230000023597 hemostasis Effects 0.000 description 78
- 238000001802 infusion Methods 0.000 description 44
- 239000008280 blood Substances 0.000 description 32
- 210000004369 blood Anatomy 0.000 description 32
- 230000002093 peripheral effect Effects 0.000 description 17
- 230000002439 hemostatic effect Effects 0.000 description 15
- 239000000463 material Substances 0.000 description 14
- 239000003795 chemical substances by application Substances 0.000 description 13
- 230000000052 comparative effect Effects 0.000 description 13
- 210000003462 vein Anatomy 0.000 description 10
- 229920005989 resin Polymers 0.000 description 8
- 239000011347 resin Substances 0.000 description 8
- 210000004204 blood vessel Anatomy 0.000 description 7
- 230000017531 blood circulation Effects 0.000 description 5
- 238000004891 communication Methods 0.000 description 5
- 239000000470 constituent Substances 0.000 description 5
- 239000013013 elastic material Substances 0.000 description 5
- 238000006073 displacement reaction Methods 0.000 description 4
- 238000007789 sealing Methods 0.000 description 4
- 239000004698 Polyethylene Substances 0.000 description 3
- 230000036772 blood pressure Effects 0.000 description 3
- 229920003049 isoprene rubber Polymers 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 230000000149 penetrating effect Effects 0.000 description 3
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 3
- 229920002647 polyamide Polymers 0.000 description 3
- 229920000728 polyester Polymers 0.000 description 3
- -1 polyethylene Polymers 0.000 description 3
- 238000003825 pressing Methods 0.000 description 3
- 230000001012 protector Effects 0.000 description 3
- 239000004952 Polyamide Substances 0.000 description 2
- 239000005062 Polybutadiene Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 229920001971 elastomer Polymers 0.000 description 2
- 230000004927 fusion Effects 0.000 description 2
- 239000003978 infusion fluid Substances 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 230000035515 penetration Effects 0.000 description 2
- 229920002857 polybutadiene Polymers 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 229920005672 polyolefin resin Polymers 0.000 description 2
- 229920002635 polyurethane Polymers 0.000 description 2
- 239000004814 polyurethane Substances 0.000 description 2
- 239000005060 rubber Substances 0.000 description 2
- 229910000838 Al alloy Inorganic materials 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- 229920000459 Nitrile rubber Polymers 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 239000004721 Polyphenylene oxide Substances 0.000 description 1
- 229910001069 Ti alloy Inorganic materials 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 229920006311 Urethane elastomer Polymers 0.000 description 1
- 229920000800 acrylic rubber Polymers 0.000 description 1
- 239000002390 adhesive tape Substances 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- AYJRCSIUFZENHW-DEQYMQKBSA-L barium(2+);oxomethanediolate Chemical compound [Ba+2].[O-][14C]([O-])=O AYJRCSIUFZENHW-DEQYMQKBSA-L 0.000 description 1
- 229940036348 bismuth carbonate Drugs 0.000 description 1
- 229920005549 butyl rubber Polymers 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 239000002872 contrast media Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- GMZOPRQQINFLPQ-UHFFFAOYSA-H dibismuth;tricarbonate Chemical compound [Bi+3].[Bi+3].[O-]C([O-])=O.[O-]C([O-])=O.[O-]C([O-])=O GMZOPRQQINFLPQ-UHFFFAOYSA-H 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 239000005038 ethylene vinyl acetate Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920005606 polypropylene copolymer Polymers 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 239000004945 silicone rubber Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 229920003048 styrene butadiene rubber Polymers 0.000 description 1
- 229920002725 thermoplastic elastomer Polymers 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000012780 transparent material Substances 0.000 description 1
- CMPGARWFYBADJI-UHFFFAOYSA-L tungstic acid Chemical compound O[W](O)(=O)=O CMPGARWFYBADJI-UHFFFAOYSA-L 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0067—Catheters; Hollow probes characterised by the distal end, e.g. tips
- A61M25/0074—Dynamic characteristics of the catheter tip, e.g. openable, closable, expandable or deformable
- A61M25/0075—Valve means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/06—Body-piercing guide needles or the like
- A61M25/0606—"Over-the-needle" catheter assemblies, e.g. I.V. catheters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0067—Catheters; Hollow probes characterised by the distal end, e.g. tips
- A61M25/0082—Catheter tip comprising a tool
- A61M25/0084—Catheter tip comprising a tool being one or more injection needles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M39/00—Tubes, tube connectors, tube couplings, valves, access sites or the like, specially adapted for medical use
- A61M39/02—Access sites
- A61M39/06—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof
- A61M2039/0633—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof the seal being a passive seal made of a resilient material with or without an opening
- A61M2039/064—Slit-valve
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M39/00—Tubes, tube connectors, tube couplings, valves, access sites or the like, specially adapted for medical use
- A61M39/02—Access sites
- A61M39/06—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof
- A61M2039/0633—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof the seal being a passive seal made of a resilient material with or without an opening
- A61M2039/0653—Perforated disc
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biophysics (AREA)
- Pulmonology (AREA)
- Engineering & Computer Science (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Hematology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Infusion, Injection, And Reservoir Apparatuses (AREA)
- Media Introduction/Drainage Providing Device (AREA)
Description
Claims (6)
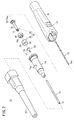
- 管状の挿入部(12)と、
前記挿入部(12)の基端側に接続され、該挿入部(12)内に連通する流路(26)を内部に有する保持部(14)と、
前記流路(26)上に配設され、流体の流通を遮断する閉塞膜(58)、及び該閉塞膜(58)を部分的に開閉可能な開閉部(60)を有する弁体(46)と、
前記閉塞膜(58)よりも基端側に配置され、先端方向への移動によって前記開閉部(60)を挿通する挿通部材(50)と、を備えるカテーテル組立体(10)であって、
前記開閉部(60)は、前記閉塞膜(58)に1本のスリット状に貫通形成されたものであり、且つ前記開閉部(60)の、前記挿通部材(50)が挿出される先端口部(62)の線長をLf、前記開閉部(60)の、前記挿通部材(50)が挿入される基端口部(64)の線長をLrとした場合に、Lf<Lrの関係で形成され、
且つ前記弁体(46)の側面断面視で、前記先端口部(62)及び前記基端口部(64)を上底及び下底とする等脚台形形状に形成される
ことを特徴とするカテーテル組立体(10)。 - 請求項1記載のカテーテル組立体(10)において、
前記弁体(46)の基端側には、気体を通過させる一方で、液体を通過させ難いシール部材(48)が前記流路(26)を塞ぐように配置される
ことを特徴とするカテーテル組立体(10)。 - 請求項1又は2記載のカテーテル組立体(10)において、
前記挿入部(12)内に挿通される内針(16)と、
前記内針(16)の基端部に固着され、前記保持部(14)の基端側と接続可能な内針ハブ(18)と、を有する
ことを特徴とするカテーテル組立体(10)。 - 請求項1〜3のいずれか一項に記載のカテーテル組立体(10)において、
前記基端口部(64)の線長Lrは、前記先端口部(62)の線長Lfよりも30%以上大きい
ことを特徴とするカテーテル組立体(10)。 - 請求項1〜4のいずれか一項に記載のカテーテル組立体(10)において、
前記閉塞膜(58)は、平坦状に形成され、前記保持部(14)の軸方向と直交する方向に配置されている
ことを特徴とするカテーテル組立体(10)。 - 請求項5記載のカテーテル組立体(10)において、
前記弁体(46)は、
前記閉塞膜(58)の周縁に連結して該閉塞膜(58)を支持し、前記保持部(14)の軸方向に延在する側壁(56)と、
前記閉塞膜(58)の基端側で前記側壁(56)によって囲まれ、前記挿通部材(50)を移動自在に配置可能な空洞と、を有する
ことを特徴とするカテーテル組立体(10)。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013536153A JP6067567B2 (ja) | 2011-09-28 | 2012-09-12 | カテーテル組立体 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011212694 | 2011-09-28 | ||
| JP2011212694 | 2011-09-28 | ||
| JP2013536153A JP6067567B2 (ja) | 2011-09-28 | 2012-09-12 | カテーテル組立体 |
| PCT/JP2012/073328 WO2013047205A1 (ja) | 2011-09-28 | 2012-09-12 | カテーテル組立体 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2013047205A1 JPWO2013047205A1 (ja) | 2015-03-26 |
| JP6067567B2 true JP6067567B2 (ja) | 2017-01-25 |
Family
ID=47995239
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013536153A Active JP6067567B2 (ja) | 2011-09-28 | 2012-09-12 | カテーテル組立体 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9095679B2 (ja) |
| EP (1) | EP2762193B1 (ja) |
| JP (1) | JP6067567B2 (ja) |
| CN (1) | CN103842021B (ja) |
| AU (1) | AU2012317947B2 (ja) |
| WO (1) | WO2013047205A1 (ja) |
Families Citing this family (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10089443B2 (en) | 2012-05-15 | 2018-10-02 | Baxter International Inc. | Home medical device systems and methods for therapy prescription and tracking, servicing and inventory |
| US8323249B2 (en) | 2009-08-14 | 2012-12-04 | The Regents Of The University Of Michigan | Integrated vascular delivery system |
| US8814833B2 (en) | 2010-05-19 | 2014-08-26 | Tangent Medical Technologies Llc | Safety needle system operable with a medical device |
| US8771230B2 (en) | 2010-05-19 | 2014-07-08 | Tangent Medical Technologies, Llc | Integrated vascular delivery system |
| CA2891255C (en) | 2012-11-12 | 2022-10-04 | Icu Medical, Inc. | Medical connector |
| US9592367B2 (en) * | 2013-07-30 | 2017-03-14 | Becton, Dickinson And Company | Blood control catheter valve employing actuator with flexible retention arms |
| JP6461174B2 (ja) | 2014-02-04 | 2019-01-30 | アイシーユー・メディカル・インコーポレーテッド | 自己プライミングシステムおよび自己プライミング方法 |
| JPWO2015141366A1 (ja) * | 2014-03-20 | 2017-04-06 | テルモ株式会社 | カテーテル組立体 |
| WO2015141365A1 (ja) * | 2014-03-20 | 2015-09-24 | テルモ株式会社 | カテーテル組立体 |
| CN112675412B (zh) | 2014-04-18 | 2023-01-13 | 贝克顿·迪金森公司 | 用于导管的针捕获安全互锁设备 |
| ES2880104T3 (es) | 2015-08-18 | 2021-11-23 | Braun Melsungen Ag | Dispositivos de catéter con válvulas |
| DE102016106626A1 (de) | 2016-04-11 | 2017-10-12 | Joline Gmbh & Co. Kg | Einführkatheter mit einem Ventilkörper |
| US11213656B2 (en) | 2016-04-15 | 2022-01-04 | Becton, Dickinson And Company | Medical device with anti-rotation push tab |
| SG10202100465YA (en) | 2016-04-15 | 2021-02-25 | Becton Dickinson Co | Medical device with anti-rotation push tab |
| CA3032639A1 (en) * | 2016-08-04 | 2018-02-08 | Nipro Corporation | Valved needle assembly and indwelling needle assembly |
| USD858755S1 (en) | 2017-01-25 | 2019-09-03 | Becton, Dickinson And Company | Catheter assembly and needle cover |
| USD859651S1 (en) | 2017-01-25 | 2019-09-10 | Becton, Dickinson And Company | Catheter hub |
| USD821574S1 (en) | 2017-01-25 | 2018-06-26 | Becton, Dickinson And Company | Needle hub |
| USD865956S1 (en) | 2017-01-25 | 2019-11-05 | Becton, Dickinson And Company | Catheter assembly |
| US10569072B2 (en) * | 2017-03-13 | 2020-02-25 | Boston Scientific Limited | Hemostasis valves and methods for making and using hemostasis valves |
| US11147957B2 (en) | 2017-07-19 | 2021-10-19 | Becton, Dickinson And Company | Systems and methods to improve instrument guidance within an intravenous catheter assembly |
| CA3071818A1 (en) * | 2017-08-02 | 2019-02-07 | Nipro Corporation | Hemostasis valve-equipped indwelling needle and indwelling needle assembly |
| EP3576829B1 (en) * | 2018-03-05 | 2021-01-06 | Poly Medicure Limited | Intravenous catheter assembly |
| US11097083B2 (en) | 2018-07-17 | 2021-08-24 | Becton, Dickinson And Company | Systems and methods to improve instrument guidance within an intravenous catheter assembly |
| WO2020127328A1 (en) | 2018-12-17 | 2020-06-25 | B. Braun Melsungen Ag | Over-the-needle catheter assemblies and related manufacturing method |
| JPWO2021210484A1 (ja) * | 2020-04-17 | 2021-10-21 |
Family Cites Families (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61131723A (ja) * | 1984-10-05 | 1986-06-19 | オリンパス光学工業株式会社 | 内視鏡用鉗子栓 |
| US5149327A (en) | 1989-09-05 | 1992-09-22 | Terumo Kabushiki Kaisha | Medical valve, catheter with valve, and catheter assembly |
| JP2510887B2 (ja) * | 1989-09-29 | 1996-06-26 | テルモ株式会社 | 医療用弁体及びその製法並びに弁体付カテ―テル |
| US5053014A (en) * | 1990-02-01 | 1991-10-01 | Critikon, Inc. | Catheter with controlled valve |
| US5195980A (en) * | 1992-01-03 | 1993-03-23 | Thomas Medical Products, Inc. | Hemostatic valve |
| JPH07148265A (ja) * | 1993-11-29 | 1995-06-13 | Nippon Zeon Co Ltd | 止血弁を有する医療器具 |
| US5911710A (en) * | 1997-05-02 | 1999-06-15 | Schneider/Namic | Medical insertion device with hemostatic valve |
| JP3991438B2 (ja) * | 1998-03-31 | 2007-10-17 | 日本ゼオン株式会社 | 医療用弁体および医療用挿入補助具 |
| US20050187524A1 (en) * | 2000-12-19 | 2005-08-25 | Willis Allan F. | Sealing valve assembly for medical products |
| CA2495203C (en) * | 2002-08-12 | 2008-07-29 | Jms Co., Ltd. | Needleless port and method of manufacturing the same |
| US8034035B2 (en) * | 2004-01-29 | 2011-10-11 | Navilyst Medical, Inc. | Pressure activated safety valve with high flow slit |
| US20070196414A1 (en) * | 2006-02-23 | 2007-08-23 | Ola Hammarsten | Device to prevent blood leakage using intravenous catheters |
| US8074338B2 (en) * | 2006-11-06 | 2011-12-13 | Becton, Dickinson And Company | Vascular access devices including a tear-resistant septum |
| US20080132833A1 (en) * | 2006-11-06 | 2008-06-05 | Becton, Dickinson And Company | Vascular access devices including a tear-resistant septum |
| CN201033193Y (zh) * | 2007-05-15 | 2008-03-12 | 上海林华医疗器械有限公司 | 静脉留置针止血密封塞 |
| WO2009002828A2 (en) * | 2007-06-22 | 2008-12-31 | Medical Components, Inc. | Tearaway sheath assembly with hemostasis valve |
| US9101748B2 (en) * | 2008-05-08 | 2015-08-11 | Becton, Dickinson And Company | Push-button blood control |
| CN201286901Y (zh) * | 2008-09-12 | 2009-08-12 | 吴林元 | 静脉留置针隔离塞 |
| JP5321957B2 (ja) * | 2008-10-03 | 2013-10-23 | ニプロ株式会社 | 留置針組立体 |
| US8469928B2 (en) * | 2009-02-11 | 2013-06-25 | Becton, Dickinson And Company | Systems and methods for providing a flushable catheter assembly |
| US8388583B2 (en) * | 2009-08-20 | 2013-03-05 | Becton, Dickinson And Company | Systems and methods for providing a flushable catheter assembly |
| DE202009013213U1 (de) * | 2009-10-01 | 2009-12-17 | B. Braun Melsungen Ag | Kathetereinführvorrichtung |
| CN201612944U (zh) * | 2009-12-08 | 2010-10-27 | 上海锦泽夏本医疗器械有限公司 | 一种逆流防止阀以及采用该防止阀的留置针 |
| JP5785951B2 (ja) * | 2009-12-15 | 2015-09-30 | クック・メディカル・テクノロジーズ・リミテッド・ライアビリティ・カンパニーCook Medical Technologies Llc | 低丈ワンウェイバルブ |
| CN201862061U (zh) * | 2010-09-07 | 2011-06-15 | 陈尊孔 | 蝶式双安全留置针 |
-
2012
- 2012-09-12 CN CN201280043941.2A patent/CN103842021B/zh active Active
- 2012-09-12 JP JP2013536153A patent/JP6067567B2/ja active Active
- 2012-09-12 AU AU2012317947A patent/AU2012317947B2/en active Active
- 2012-09-12 EP EP12835725.8A patent/EP2762193B1/en active Active
- 2012-09-12 WO PCT/JP2012/073328 patent/WO2013047205A1/ja active Application Filing
-
2014
- 2014-03-27 US US14/227,941 patent/US9095679B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| AU2012317947A1 (en) | 2014-03-20 |
| EP2762193B1 (en) | 2016-10-26 |
| WO2013047205A1 (ja) | 2013-04-04 |
| EP2762193A1 (en) | 2014-08-06 |
| JPWO2013047205A1 (ja) | 2015-03-26 |
| US9095679B2 (en) | 2015-08-04 |
| CN103842021A (zh) | 2014-06-04 |
| US20140213987A1 (en) | 2014-07-31 |
| CN103842021B (zh) | 2017-06-27 |
| EP2762193A4 (en) | 2015-08-19 |
| AU2012317947B2 (en) | 2015-06-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6067567B2 (ja) | カテーテル組立体 | |
| JP5877196B2 (ja) | カテーテル組立体 | |
| US7740614B2 (en) | Indwelling needle assembly | |
| US8951230B2 (en) | Indwelling needle and indwelling needle assembly | |
| JP7309024B2 (ja) | カテーテル組立体 | |
| US8192404B2 (en) | Indwelling needle assembly | |
| JP2013070870A (ja) | カテーテル組立体 | |
| JP5253385B2 (ja) | 留置針組立体 | |
| EP1944049A1 (en) | Needle-stent device | |
| JP4812472B2 (ja) | 留置針組立体 | |
| US20220203073A1 (en) | Catheter assembly | |
| WO2015141365A1 (ja) | カテーテル組立体 | |
| JP2007125126A (ja) | 留置針組立体 | |
| JP6978885B2 (ja) | カテーテル組立体 | |
| JP4966860B2 (ja) | 留置針組立体 | |
| JP2007312925A (ja) | 留置針組立体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20150319 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20160119 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160311 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20160712 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160909 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20161122 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20161221 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6067567 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |