JP5509098B2 - 骨治療システムおよび方法 - Google Patents
骨治療システムおよび方法 Download PDFInfo
- Publication number
- JP5509098B2 JP5509098B2 JP2010548917A JP2010548917A JP5509098B2 JP 5509098 B2 JP5509098 B2 JP 5509098B2 JP 2010548917 A JP2010548917 A JP 2010548917A JP 2010548917 A JP2010548917 A JP 2010548917A JP 5509098 B2 JP5509098 B2 JP 5509098B2
- Authority
- JP
- Japan
- Prior art keywords
- bpo
- bone cement
- polymer particles
- cement
- component
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000000034 method Methods 0.000 title claims description 115
- 210000000988 bone and bone Anatomy 0.000 title description 77
- 239000002639 bone cement Substances 0.000 claims description 405
- 239000004342 Benzoyl peroxide Substances 0.000 claims description 254
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 claims description 254
- 235000019400 benzoyl peroxide Nutrition 0.000 claims description 254
- 239000000203 mixture Substances 0.000 claims description 210
- 239000002245 particle Substances 0.000 claims description 168
- 229920000642 polymer Polymers 0.000 claims description 166
- 238000002156 mixing Methods 0.000 claims description 116
- 239000007788 liquid Substances 0.000 claims description 109
- 229920003229 poly(methyl methacrylate) Polymers 0.000 claims description 96
- 239000004926 polymethyl methacrylate Substances 0.000 claims description 92
- 239000000843 powder Substances 0.000 claims description 88
- 239000000178 monomer Substances 0.000 claims description 58
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 claims description 26
- 238000006116 polymerization reaction Methods 0.000 claims description 23
- 239000010410 layer Substances 0.000 claims description 21
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 claims description 12
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 claims description 11
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 claims description 6
- GYVGXEWAOAAJEU-UHFFFAOYSA-N n,n,4-trimethylaniline Chemical compound CN(C)C1=CC=C(C)C=C1 GYVGXEWAOAAJEU-UHFFFAOYSA-N 0.000 claims description 6
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 claims description 6
- 239000002344 surface layer Substances 0.000 claims description 6
- -1 A powder component Chemical compound 0.000 claims description 4
- 239000004568 cement Substances 0.000 description 206
- 239000011324 bead Substances 0.000 description 57
- 238000002347 injection Methods 0.000 description 46
- 239000007924 injection Substances 0.000 description 46
- 239000000463 material Substances 0.000 description 35
- 238000006243 chemical reaction Methods 0.000 description 31
- 230000007246 mechanism Effects 0.000 description 26
- 239000002243 precursor Substances 0.000 description 26
- 238000009472 formulation Methods 0.000 description 22
- 230000008859 change Effects 0.000 description 20
- 238000010438 heat treatment Methods 0.000 description 15
- 230000014759 maintenance of location Effects 0.000 description 15
- 230000004044 response Effects 0.000 description 15
- 206010017076 Fracture Diseases 0.000 description 13
- 239000003999 initiator Substances 0.000 description 11
- 208000010392 Bone Fractures Diseases 0.000 description 10
- 239000012530 fluid Substances 0.000 description 10
- 206010010214 Compression fracture Diseases 0.000 description 9
- 208000002193 Pain Diseases 0.000 description 7
- 230000003321 amplification Effects 0.000 description 7
- 239000011248 coating agent Substances 0.000 description 7
- 238000000576 coating method Methods 0.000 description 7
- 230000007423 decrease Effects 0.000 description 7
- 238000011049 filling Methods 0.000 description 7
- 238000003199 nucleic acid amplification method Methods 0.000 description 7
- 239000002872 contrast media Substances 0.000 description 6
- 238000003384 imaging method Methods 0.000 description 6
- 230000036407 pain Effects 0.000 description 6
- 230000004913 activation Effects 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 229920001971 elastomer Polymers 0.000 description 5
- 230000000717 retained effect Effects 0.000 description 5
- 206010041569 spinal fracture Diseases 0.000 description 5
- 208000001132 Osteoporosis Diseases 0.000 description 4
- 238000013459 approach Methods 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 230000001054 cortical effect Effects 0.000 description 4
- 230000006378 damage Effects 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 239000000945 filler Substances 0.000 description 4
- 239000003094 microcapsule Substances 0.000 description 4
- 230000001009 osteoporotic effect Effects 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 239000003522 acrylic cement Substances 0.000 description 3
- 230000002146 bilateral effect Effects 0.000 description 3
- 230000006835 compression Effects 0.000 description 3
- 238000007906 compression Methods 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 230000009969 flowable effect Effects 0.000 description 3
- 238000002594 fluoroscopy Methods 0.000 description 3
- 229910052500 inorganic mineral Inorganic materials 0.000 description 3
- 239000011707 mineral Substances 0.000 description 3
- 230000001575 pathological effect Effects 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 230000035882 stress Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 206010020100 Hip fracture Diseases 0.000 description 2
- 208000001164 Osteoporotic Fractures Diseases 0.000 description 2
- 208000010378 Pulmonary Embolism Diseases 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 230000003213 activating effect Effects 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- 239000003828 free initiator Substances 0.000 description 2
- 230000000977 initiatory effect Effects 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 239000002861 polymer material Substances 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 238000011272 standard treatment Methods 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 230000000007 visual effect Effects 0.000 description 2
- 229920003026 Acene Polymers 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- 208000006386 Bone Resorption Diseases 0.000 description 1
- 206010065687 Bone loss Diseases 0.000 description 1
- 208000009079 Bronchial Spasm Diseases 0.000 description 1
- 208000014181 Bronchial disease Diseases 0.000 description 1
- 206010006482 Bronchospasm Diseases 0.000 description 1
- 208000000094 Chronic Pain Diseases 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 206010015866 Extravasation Diseases 0.000 description 1
- NNJVILVZKWQKPM-UHFFFAOYSA-N Lidocaine Chemical compound CCN(CC)CC(=O)NC1=C(C)C=CC=C1C NNJVILVZKWQKPM-UHFFFAOYSA-N 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- 208000031481 Pathologic Constriction Diseases 0.000 description 1
- 208000006735 Periostitis Diseases 0.000 description 1
- 241000219061 Rheum Species 0.000 description 1
- 206010039897 Sedation Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 238000007792 addition Methods 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 1
- 238000011882 arthroplasty Methods 0.000 description 1
- 239000012867 bioactive agent Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 230000037182 bone density Effects 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 230000024279 bone resorption Effects 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 230000001427 coherent effect Effects 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 238000004891 communication Methods 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 238000002591 computed tomography Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 230000006837 decompression Effects 0.000 description 1
- 238000007599 discharging Methods 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000013013 elastic material Substances 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 230000036251 extravasation Effects 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 238000002695 general anesthesia Methods 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 201000011066 hemangioma Diseases 0.000 description 1
- 210000004394 hip joint Anatomy 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 229960004194 lidocaine Drugs 0.000 description 1
- 239000003589 local anesthetic agent Substances 0.000 description 1
- 230000004807 localization Effects 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 230000000399 orthopedic effect Effects 0.000 description 1
- 230000011164 ossification Effects 0.000 description 1
- 210000000963 osteoblast Anatomy 0.000 description 1
- 210000002997 osteoclast Anatomy 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 210000003460 periosteum Anatomy 0.000 description 1
- 230000002572 peristaltic effect Effects 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 208000001685 postmenopausal osteoporosis Diseases 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000005086 pumping Methods 0.000 description 1
- 230000001698 pyrogenic effect Effects 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 238000010526 radical polymerization reaction Methods 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000036280 sedation Effects 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 230000036262 stenosis Effects 0.000 description 1
- 208000037804 stenosis Diseases 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 230000003685 thermal hair damage Effects 0.000 description 1
- 230000000472 traumatic effect Effects 0.000 description 1
- 210000005239 tubule Anatomy 0.000 description 1
- 238000002604 ultrasonography Methods 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L24/00—Surgical adhesives or cements; Adhesives for colostomy devices
- A61L24/0047—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material
- A61L24/0073—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material with a macromolecular matrix
- A61L24/0094—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material with a macromolecular matrix containing macromolecular fillers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/68—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin
- A61B17/70—Spinal positioners or stabilisers ; Bone stabilisers comprising fluid filler in an implant
- A61B17/7094—Solid vertebral fillers; devices for inserting such fillers
- A61B17/7095—Solid vertebral fillers; devices for inserting such fillers the filler comprising unlinked macroscopic particles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/88—Osteosynthesis instruments; Methods or means for implanting or extracting internal or external fixation devices
- A61B17/8802—Equipment for handling bone cement or other fluid fillers
- A61B17/8805—Equipment for handling bone cement or other fluid fillers for introducing fluid filler into bone or extracting it
- A61B17/8822—Equipment for handling bone cement or other fluid fillers for introducing fluid filler into bone or extracting it characterised by means facilitating expulsion of fluid from the introducer, e.g. a screw pump plunger, hydraulic force transmissions, application of vibrations or a vacuum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/88—Osteosynthesis instruments; Methods or means for implanting or extracting internal or external fixation devices
- A61B17/8802—Equipment for handling bone cement or other fluid fillers
- A61B17/8833—Osteosynthesis tools specially adapted for handling bone cement or fluid fillers; Means for supplying bone cement or fluid fillers to introducing tools, e.g. cartridge handling means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/88—Osteosynthesis instruments; Methods or means for implanting or extracting internal or external fixation devices
- A61B17/8802—Equipment for handling bone cement or other fluid fillers
- A61B17/8833—Osteosynthesis tools specially adapted for handling bone cement or fluid fillers; Means for supplying bone cement or fluid fillers to introducing tools, e.g. cartridge handling means
- A61B17/8836—Osteosynthesis tools specially adapted for handling bone cement or fluid fillers; Means for supplying bone cement or fluid fillers to introducing tools, e.g. cartridge handling means for heating, cooling or curing of bone cement or fluid fillers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/74—Synthetic polymeric materials
- A61K31/765—Polymers containing oxygen
- A61K31/78—Polymers containing oxygen of acrylic acid or derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L24/00—Surgical adhesives or cements; Adhesives for colostomy devices
- A61L24/001—Use of materials characterised by their function or physical properties
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L24/00—Surgical adhesives or cements; Adhesives for colostomy devices
- A61L24/0047—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material
- A61L24/0073—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material with a macromolecular matrix
- A61L24/0084—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material with a macromolecular matrix containing fillers of phosphorus-containing inorganic compounds, e.g. apatite
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L24/00—Surgical adhesives or cements; Adhesives for colostomy devices
- A61L24/0047—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material
- A61L24/0073—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material with a macromolecular matrix
- A61L24/0089—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material with a macromolecular matrix containing inorganic fillers not covered by groups A61L24/0078 or A61L24/0084
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L24/00—Surgical adhesives or cements; Adhesives for colostomy devices
- A61L24/04—Surgical adhesives or cements; Adhesives for colostomy devices containing macromolecular materials
- A61L24/06—Surgical adhesives or cements; Adhesives for colostomy devices containing macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/16—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/40—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material
- A61L27/44—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material having a macromolecular matrix
- A61L27/46—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material having a macromolecular matrix with phosphorus-containing inorganic fillers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B2017/00017—Electrical control of surgical instruments
- A61B2017/00022—Sensing or detecting at the treatment site
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2400/00—Materials characterised by their function or physical properties
- A61L2400/06—Flowable or injectable implant compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/02—Materials or treatment for tissue regeneration for reconstruction of bones; weight-bearing implants
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Surgery (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Medicinal Chemistry (AREA)
- Composite Materials (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Medical Informatics (AREA)
- Molecular Biology (AREA)
- Inorganic Chemistry (AREA)
- Dermatology (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Neurology (AREA)
- Pharmacology & Pharmacy (AREA)
- Materials For Medical Uses (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Prostheses (AREA)
- Surgical Instruments (AREA)
Description
本出願は、2008年2月1日に出願した米国特許出願第12/024969号(代理人参照番号:DFINE.054A)の一部継続出願である。本出願はさらに、2008年2月28日に出願した「Bone System Treatment Systems and Methods」という名称の米国特許仮出願第61/067479号(代理人参照番号:DFINE.068PR)、2008年2月28日に出願した「Bone System Treatment Systems and Methods」という名称の米国特許仮出願第61/067480号(代理人参照番号:DFINE.069PR)、2008年4月16日に出願した「Bone Treatment Systems and Methods」という名称の米国特許仮出願第61/124336号(代理人参照番号:DFINE.077PR)、2008年8月28日に出願した「Bone Treatment Systems and Methods」という名称の米国特許仮出願第61/190375号(代理人参照番号:DFINE.078PR)、および2008年4月16日に出願した「Bone Treatment Devices and Methods」という名称の米国特許仮出願第61/124338号(代理人参照番号:DFINE.074PR)の、米国特許法第119(e)条に基づく優先権の利益を主張するものであり、これらのいずれもの内容全体を参照により本明細書に組み込み、本明細書の一部と見なすべきである。
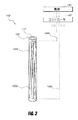
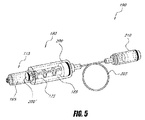
100 第1の構成要素、骨セメント注入器、注入器システム
105 第2の構成要素、セメント活性化構成要素、ハンドル部分
110 エミッタ
112 流れ流通路、チャネル、流れチャネル、流通路
113 ねじ部分
114 回転可能なねじ式取付具
115 シリンジ型の本体、シリンジ、骨セメント供給源
116 ねじ取付具
118 近位端
120 遠位端
122 流れの出口、注入器出口
124 延長部分
125 軸
130 骨セメント、骨セメントシステム、骨治療システム
132 チャンバ
140 電源、エネルギー源
145 コントローラ、コントローラアセンブリ
146 電気コネクタ
148 ケーブル
149a 導線
149b 導線
150 壁部分
155A 表面電極
155B 表面電極
162 油圧システム
163 取付具
164 取付具
165 セメント保持孔、セメント保持チャンバ、チャンバ
175 剛性プランジャ、アクチュエータ部材
176 Oリング、ゴムヘッド
180 加力および増幅構成要素
185 孔、チャンバ、加圧可能なチャンバ
186 近位端
187 Oリング、ガスケット
188 流動媒体
190 圧力機構、圧力
200 境界面
200’ 境界面
205 空気圧系または油圧系
210 シリンジポンプ、加圧機構、圧力源
211 電気モータ、モータ駆動システム
212 リモートスイッチ、作動スイッチ
214 ケーブル
240 時間−粘度曲線
250 時間−粘度曲線
260 修正された時間−粘度曲線、時間−粘度応答
270 センサ
272 温度センサ
275 混合デバイス、混合アセンブリ、セメント混合アセンブリ
276 温度センサ
280 包装
285 センサ、スイッチ、表示機構
288 センサ、スイッチ、表示機構
290 センサ
400 骨セメントシステム
405 粒子
410 第2のエミッタ
414a 電気コネクタ構成要素
414b 電気コネクタ構成要素
416 電線
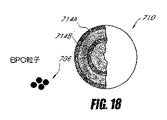
700 第1のポリマービーズ、粒子、第1の体積のポリマービーズ、
704A BPO
704B BPO
704C BPO粒子
705 第2のポリマービーズ、粒子、第2の体積のポリマービーズ
706 BPOの粒子
708 ポリマー粒子
710 ポリマー粒子
712 マイクロカプセル化されたBPO
714A 表層、表面のBPO層、BPO表面被覆
714B BPO層、内部のBPO層、内部層
716 ポリマー粒子
720 ポリマー粉末、粒子
722 BPO粒子
750 BPO利用可能率曲線
755A 領域
755B 領域
800 曲線
805A 第1の間隔
805B 第2の間隔
806 BPO利用可能率曲線
810 BPO利用可能率曲線
815A 第1の間隔
815B 第2の間隔
815C 第3の間隔
820 PMMA材料
820A PMMA層
820B PMMA材料
822 BPO粒子
824 PMMA材料
840 時間−粘度曲線
Claims (19)
- 骨セメント組成物であって、
メチルメタクリレート(MMA)を含むモノマー成分と、
ポリメチルメタクリレートポリマー(PMMA)を含むポリマー成分と、を含み、
前記ポリマー成分が、
平均直径が100μm以下である第1の体積のポリマー粒子であって、当該第1の体積のポリマー粒子が、第1のポリマー粒子の全重量に対して、0.9から1.1重量%の過酸化ベンゾイル(BPO)を含む、第1の体積のポリマー粒子と、
平均直径が100μm未満である第2の体積のポリマー粒子であって、当該第2の体積のポリマー粒子が、第2のポリマー粒子の全重量に対して、1.1から1.3重量%のBPOを含む、第2の体積のポリマー粒子と、
平均直径が100μm未満である第3の体積のポリマー粒子であって、当該第3の体積のポリマー粒子が、第3のポリマー粒子の全重量に対して、0.0重量%から0.1重量%のBPOを含む、第3の体積のポリマー粒子と、
を含み、
前記BPOが、前記ポリマー粒子に組み込まれたBPO粒子として提供され、前記ポリマー成分が、当該骨セメント組成物の重合中に時間の関数として前記モノマー成分に曝露されるBPOの量を制御することを特徴とする骨セメント組成物。 - 前記第1の体積のポリマー粒子が、150,000〜350,000の分子量をもつことを特徴とする請求項1に記載の骨セメント組成物。
- 前記第2の体積のポリマー粒子が、300,000〜500,000の分子量をもつことを特徴とする請求項1または2に記載の骨セメント組成物。
- 前記第3の体積のポリマー粒子が、250,000〜450,000の分子量をもつことを特徴とする請求項1から3のいずれか1項に記載の骨セメント組成物。
- 前記第1の体積のポリマー粒子が、100μm〜120μmの平均直径を有することを特徴とする請求項1から4のいずれか1項に記載の骨セメント組成物。
- 前記第2の体積のポリマー粒子が、70μm〜90μmの平均直径を有することを特徴とする請求項1から5のいずれか1項に記載の骨セメント組成物。
- 前記第3の体積のポリマー粒子が、前記ポリマー成分の全重量に対して、10%〜30%であることを特徴とする請求項1から6のいずれか1項に記載の骨セメント組成物。
- 前記第3の体積の粒子が、BPOを含まないことを特徴とする請求項1から7のいずれか1項に記載の骨セメント組成物。
- 骨セメント組成物を調製する方法であって、
液体成分と非液体成分とを混合して、重合可能な骨セメント組成物を知供する段階を含み、
前記液体成分が、少なくとも1つのモノマーを含み、
前記非液体成分が、過酸化ベンゾイル(BPO)を含む複数のポリマー粒子を含み、
前記複数のポリマー粒子が、
平均直径が100μm以下である第1の体積のポリマー粒子であって、当該第1の体積のポリマー粒子が、第1のポリマー粒子の全重量に対して、0.9から1.1重量%のBPOを含む、第1の体積のポリマー粒子と、
平均直径が100未満である第2の体積のポリマー粒子であって、当該第2の体積のポリマー粒子が、第2のポリマー粒子の全重量に対して、1.1から1.3重量%のBPOを含む、第2の体積のポリマー粒子と、
平均直径が100未満である第3の体積のポリマー粒子であって、当該第3の体積のポリマー粒子が、第3のポリマー粒子の全重量に対して、0.0重量%から0.1重量%のBPOを含む、第3の体積のポリマー粒子と、
を含み、
前記非液体成分が、前記骨セメント組成物の重合中に時間の関数として前記液体成分に曝露されるBPOの量を制御するように構成されており、
前記骨セメント組成物が、
ポリメチルメタクリレートポリマー(PMMA)を含む粉末成分と、
メチルメタクリレート(MMA)を含む液体成分と、
を含むことを特徴とする方法。 - 粉末成分であって、
45重量%〜55重量%のPMMAと、
55重量%〜45重量%の二酸化ジルコニウムまたは硫酸バリウムと、
過酸化ベンゾイル(BPO)と、
を含み、
当該粉末成分の量が、当該粉末成分の全重量に対してのものである、粉末成分と、
前記液体成分であって、
98.0重量%〜99.9重量%のMMAと、
0.15重量%〜0.95重量%のN,N−ジメチル−p−トルイジン(DMPT)と、
30ppm〜150ppmのヒドロキノン(HQ)と、
を含み、
当該液体成分の量が、当該液体成分の全重量に対してのものである、液体成分と、
を含むことを特徴とする請求項9に記載の方法。 - 前記第1の体積の粒子が、100μm〜120μmの平均直径を有することを特徴とする請求項9または10に記載の方法。
- 前記第2の体積の粒子が、70μm〜90μmの平均直径を有することを特徴とする請求項9から11のいずれか1項に記載の方法。
- 前記第1の体積のポリマーが、前記ポリマー粒子の全重量に対して前記ポリマー粒子の40%〜50%であることを特徴とする請求項9から12のいずれか1項に記載の方法。
- 前記第2の体積の粒子が、前記ポリマー粒子の全重量に対して前記ポリマー粒子の30%〜40%であることを特徴とする請求項9から13のいずれか1項に記載の方法。
- 前記第3の体積の粒子が、前記ポリマー粒子の全重量に対して前記ポリマー粒子の10%〜30%であることを特徴とする請求項9から14のいずれか1項に記載の方法。
- 前記BPOは、前記ポリマー粒子がBPOからなる少なくとも1つの層を含むよう構成され、このものにおいて、前記BPOは、前記ポリマー粒子の外面の少なくとも一部の上に存在する表層として構成されるか、または、前記BPOは、前記ポリマー粒子の内部に存在する層として構成されることを特徴とする請求項1から7のいずれか1項に記載の骨セメント組成物。
- 前記BPOは、前記ポリマー粒子がマイクロカプセル化されたBPOを含むよう構成され、このものにおいて、前記マイクロカプセル化されたBPOの少なくとも一部は、前記ポリマー粒子に組み込まれ、かつ、前記マイクロカプセル化されたBPOの少なくとも一部は、前記ポリマー粒子に組み込まれないことを特徴とする請求項1から7のいずれか1項に記載の骨セメント組成物。
- 前記BPOは、前記ポリマー粒子がBPOからなる少なくとも1つの層を含むよう構成され、このものにおいて、前記BPOは、前記ポリマー粒子の外面の少なくとも一部の上に存在する表層として構成されるか、または、前記BPOは、前記ポリマー粒子の内部に存在する層として構成されることを特徴とする請求項9から14のいずれか1項に記載の方法。
- 前記BPOは、前記ポリマー粒子がマイクロカプセル化されたBPOを含むよう構成され、このものにおいて、前記マイクロカプセル化されたBPOの少なくとも一部は、前記ポリマー粒子に組み込まれ、かつ、前記マイクロカプセル化されたBPOの少なくとも一部は、前記ポリマー粒子に組み込まれないことを特徴とする請求項9から14のいずれか1項に記載の方法。
Applications Claiming Priority (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US6747908P | 2008-02-28 | 2008-02-28 | |
| US6748008P | 2008-02-28 | 2008-02-28 | |
| US61/067,480 | 2008-02-28 | ||
| US61/067,479 | 2008-02-28 | ||
| US12433608P | 2008-04-16 | 2008-04-16 | |
| US12433808P | 2008-04-16 | 2008-04-16 | |
| US61/124,336 | 2008-04-16 | ||
| US61/124,338 | 2008-04-16 | ||
| US19037508P | 2008-08-28 | 2008-08-28 | |
| US61/190,375 | 2008-08-28 | ||
| PCT/US2009/035549 WO2009108893A2 (en) | 2008-02-28 | 2009-02-27 | Bone treatment systems and methods |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2011514818A JP2011514818A (ja) | 2011-05-12 |
| JP2011514818A5 JP2011514818A5 (ja) | 2012-03-22 |
| JP5509098B2 true JP5509098B2 (ja) | 2014-06-04 |
Family
ID=40873304
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2010548917A Active JP5509098B2 (ja) | 2008-02-28 | 2009-02-27 | 骨治療システムおよび方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (3) | US8487021B2 (ja) |
| EP (2) | EP2810664B1 (ja) |
| JP (1) | JP5509098B2 (ja) |
| ES (1) | ES2483996T3 (ja) |
| HK (1) | HK1205005A1 (ja) |
| WO (1) | WO2009108893A2 (ja) |
Families Citing this family (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8361067B2 (en) | 2002-09-30 | 2013-01-29 | Relievant Medsystems, Inc. | Methods of therapeutically heating a vertebral body to treat back pain |
| US7722620B2 (en) | 2004-12-06 | 2010-05-25 | Dfine, Inc. | Bone treatment systems and methods |
| US8777479B2 (en) | 2008-10-13 | 2014-07-15 | Dfine, Inc. | System for use in bone cement preparation and delivery |
| US8540723B2 (en) | 2009-04-14 | 2013-09-24 | Dfine, Inc. | Medical system and method of use |
| WO2008137428A2 (en) | 2007-04-30 | 2008-11-13 | Dfine, Inc. | Bone treatment systems and methods |
| US9597118B2 (en) | 2007-07-20 | 2017-03-21 | Dfine, Inc. | Bone anchor apparatus and method |
| US9445854B2 (en) | 2008-02-01 | 2016-09-20 | Dfine, Inc. | Bone treatment systems and methods |
| US20100030220A1 (en) * | 2008-07-31 | 2010-02-04 | Dfine, Inc. | Bone treatment systems and methods |
| US9161798B2 (en) | 2008-02-01 | 2015-10-20 | Dfine, Inc. | Bone treatment systems and methods |
| EP2810664B1 (en) | 2008-02-28 | 2019-05-29 | Dfine, Inc. | Bone Cement Composition |
| US9180416B2 (en) | 2008-04-21 | 2015-11-10 | Dfine, Inc. | System for use in bone cement preparation and delivery |
| US10028753B2 (en) | 2008-09-26 | 2018-07-24 | Relievant Medsystems, Inc. | Spine treatment kits |
| CA2772569A1 (en) * | 2009-09-01 | 2011-03-10 | Synthes Usa, Llc | Bone cement containing bone marrow |
| DE102009043550A1 (de) * | 2009-09-30 | 2011-05-19 | Aap Biomaterials Gmbh | Knochenzement und Verfahren zu dessen Herstellung |
| US8795369B1 (en) | 2010-07-16 | 2014-08-05 | Nuvasive, Inc. | Fracture reduction device and methods |
| ES2690276T3 (es) * | 2010-09-21 | 2018-11-20 | Spinewelding Ag | Dispositivo para reparar una articulación humana o animal |
| CA2845654C (en) * | 2011-08-18 | 2016-09-13 | Matthias Militz | Expansion device for bone expansion and medical apparatus for bone expansion |
| WO2013101772A1 (en) | 2011-12-30 | 2013-07-04 | Relievant Medsystems, Inc. | Systems and methods for treating back pain |
| EP2821086B1 (en) | 2012-02-29 | 2016-12-07 | Ishihara Sangyo Kaisha, Ltd. | Bone cement composition |
| US10588691B2 (en) | 2012-09-12 | 2020-03-17 | Relievant Medsystems, Inc. | Radiofrequency ablation of tissue within a vertebral body |
| EP2914186B1 (en) | 2012-11-05 | 2019-03-13 | Relievant Medsystems, Inc. | Systems for creating curved paths through bone and modulating nerves within the bone |
| TW201422268A (zh) * | 2012-12-07 | 2014-06-16 | Ind Tech Res Inst | 填充裝置及其加熱裝置 |
| US9724151B2 (en) | 2013-08-08 | 2017-08-08 | Relievant Medsystems, Inc. | Modulating nerves within bone using bone fasteners |
| GB201317299D0 (en) * | 2013-09-30 | 2013-11-13 | Lucite Int Uk Ltd | A hardenable multi-part acrylic composition |
| MX366896B (es) | 2013-09-30 | 2019-07-30 | Lucite International Speciality Polymers & Resins Ltd | Composición acrílica de múltiples partes endurecible. |
| EP3361974A4 (en) * | 2015-10-12 | 2019-06-12 | Dfine, Inc. | DEVICES FOR REALIZING A MEDICAL ACT IN AN APPROPRIATE INTERVAL, AND ASSOCIATED SYSTEMS AND METHODS |
| FR3053239B1 (fr) * | 2016-06-30 | 2022-02-25 | Teknimed | Susbtitut osseux et systeme d'injection autonome |
| CN106620841B (zh) * | 2016-12-22 | 2019-09-03 | 宁波华科润生物科技有限公司 | 低温可注射丙烯酸树脂骨水泥及其制备方法 |
| KR102394502B1 (ko) * | 2017-03-31 | 2022-05-03 | 미쓰이 가가쿠 가부시키가이샤 | 경조직 보수용 조성물 및 경조직 보수용 키트 |
| DE102017113126A1 (de) * | 2017-06-14 | 2018-12-20 | Heraeus Medical Gmbh | Knochenzementapplikator mit Leitungselement und Verschlussaufnahme |
| CN111867640B (zh) * | 2018-03-20 | 2022-04-08 | 三井化学株式会社 | 硬组织修补用组合物及硬组织修补用套装 |
| CN108714250A (zh) * | 2018-05-30 | 2018-10-30 | 上海尚融生物科技有限公司 | 一种凝胶状快速聚合骨填充材料及其制备方法 |
| WO2021050767A1 (en) | 2019-09-12 | 2021-03-18 | Relievant Medsystems, Inc. | Systems and methods for tissue modulation |
| CN111388759B (zh) * | 2020-04-28 | 2021-09-07 | 四川大学 | 一种骨水泥复合材料及其制备方法 |
Family Cites Families (201)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2496908A (en) | 1949-04-20 | 1950-02-07 | Alfred W Doux | Serving device |
| US3349840A (en) | 1965-04-05 | 1967-10-31 | Whirlpool Co | Fluid flow control apparatus |
| US3629187A (en) * | 1969-06-25 | 1971-12-21 | Dentsply Int Inc | Dental compositions containing adduct of 2 2' - propane bis 3-(4-phenoxy)-1 2-hydroxy propane - 1 - methacrylate and isocyanate |
| US4265618A (en) | 1977-09-09 | 1981-05-05 | Solar Energy Technology, Inc. | Electrically heated endodontic syringe for injecting thermoplastic material into a root canal cavity |
| DE2758374A1 (de) | 1977-12-28 | 1979-07-12 | Hilti Ag | Spreizduebel |
| US4294251A (en) | 1978-10-17 | 1981-10-13 | Greenwald A Seth | Method of suction lavage |
| US4281420A (en) | 1979-02-15 | 1981-08-04 | Raab S | Bone connective prostheses adapted to maximize strength and durability of prostheses-bone cement interface; and methods of forming same |
| US4250887A (en) | 1979-04-18 | 1981-02-17 | Dardik Surgical Associates, P.A. | Remote manual injecting apparatus |
| US4271839A (en) | 1979-07-25 | 1981-06-09 | Thomas J. Fogarty | Dilation catheter method and apparatus |
| US4338925A (en) | 1979-12-20 | 1982-07-13 | Jo Miller | Pressure injection of bone cement apparatus and method |
| US4377168A (en) | 1981-02-27 | 1983-03-22 | Wallach Surgical Instruments, Inc. | Cryosurgical instrument |
| EP0190504B1 (en) | 1984-12-28 | 1993-04-21 | Johnson Matthey Public Limited Company | Antimicrobial compositions |
| US4735625A (en) | 1985-09-11 | 1988-04-05 | Richards Medical Company | Bone cement reinforcement and method |
| JPS62182146A (ja) | 1985-10-11 | 1987-08-10 | 三井東圧化学株式会社 | 硬化性材料 |
| EP0290433B1 (en) | 1986-11-19 | 1990-09-19 | Laboratorium Für Experimentelle Chirurgie Forschungsinstitut Davos | Method and apparatus for preparing a self-curing two-component powder liquid bone cement |
| US4969906A (en) | 1987-07-28 | 1990-11-13 | Kronman Joseph H | Bone and bony tissue replacement |
| US4772287A (en) | 1987-08-20 | 1988-09-20 | Cedar Surgical, Inc. | Prosthetic disc and method of implanting |
| US4837279A (en) | 1988-02-22 | 1989-06-06 | Pfizer Hospital Products Corp, Inc. | Bone cement |
| US4910259A (en) | 1988-09-26 | 1990-03-20 | Wolff & Kaaber A/S | Bone cement |
| US4963151A (en) | 1988-12-28 | 1990-10-16 | Trustees Of The University Of Pennsylvania | Reinforced bone cement, method of production thereof and reinforcing fiber bundles therefor |
| US4969888A (en) | 1989-02-09 | 1990-11-13 | Arie Scholten | Surgical protocol for fixation of osteoporotic bone using inflatable device |
| DE3919534A1 (de) | 1989-06-15 | 1990-12-20 | Merck Patent Gmbh | Verfahren und einrichtung zum vorbereiten von knochenzement |
| US5037437B1 (en) | 1990-01-18 | 1998-04-14 | Univ Washington | Method of bone preparation for prosthetic fixation |
| US5130950A (en) | 1990-05-16 | 1992-07-14 | Schlumberger Technology Corporation | Ultrasonic measurement apparatus |
| US5292362A (en) | 1990-07-27 | 1994-03-08 | The Trustees Of Columbia University In The City Of New York | Tissue bonding and sealing composition and method of using the same |
| DE4033343A1 (de) | 1990-10-19 | 1992-04-23 | Draenert Klaus | Werkstoff als ausgangsmaterial zur herstellung von knochenzement und verfahren zu seiner herstellung |
| US5542928A (en) | 1991-05-17 | 1996-08-06 | Innerdyne, Inc. | Method and device for thermal ablation having improved heat transfer |
| DE4122950C2 (de) | 1991-07-11 | 1994-10-06 | Haerle Anton | Knochenimplantat mit einem Hohlraum |
| US5431654A (en) | 1991-09-30 | 1995-07-11 | Stryker Corporation | Bone cement injector |
| IT228979Y1 (it) | 1992-03-09 | 1998-06-05 | Giannini Sandro | Protesi biodegradabile per correzione retropiede pronato. |
| US5334626A (en) | 1992-07-28 | 1994-08-02 | Zimmer, Inc. | Bone cement composition and method of manufacture |
| WO1995001809A1 (en) | 1993-07-06 | 1995-01-19 | Earle Michael L | Bone cement delivery gun |
| US6241734B1 (en) | 1998-08-14 | 2001-06-05 | Kyphon, Inc. | Systems and methods for placing materials into bone |
| US20030032963A1 (en) | 2001-10-24 | 2003-02-13 | Kyphon Inc. | Devices and methods using an expandable body with internal restraint for compressing cancellous bone |
| ES2287635T3 (es) | 1994-01-26 | 2007-12-16 | Kyphon Inc. | Dispositivo inflamable mejorado para uso en metodos quirurgicos relacinados a la fijacion de hueso. |
| US6248110B1 (en) | 1994-01-26 | 2001-06-19 | Kyphon, Inc. | Systems and methods for treating fractured or diseased bone using expandable bodies |
| US7166121B2 (en) | 1994-01-26 | 2007-01-23 | Kyphon Inc. | Systems and methods using expandable bodies to push apart cortical bone surfaces |
| US20060100635A1 (en) | 1994-01-26 | 2006-05-11 | Kyphon, Inc. | Inflatable device for use in surgical protocol relating to fixation of bone |
| US7044954B2 (en) | 1994-01-26 | 2006-05-16 | Kyphon Inc. | Method for treating a vertebral body |
| US5507814A (en) | 1994-03-30 | 1996-04-16 | Northwestern University | Orthopedic implant with self-reinforced mantle |
| US5571189A (en) | 1994-05-20 | 1996-11-05 | Kuslich; Stephen D. | Expandable fabric implant for stabilizing the spinal motion segment |
| US6075067A (en) | 1994-08-15 | 2000-06-13 | Corpipharm Gmbh & Co | Cement for medical use, method for producing the cement, and use of the cement |
| DE4433201A1 (de) | 1994-09-17 | 1996-03-21 | Merck Patent Gmbh | Verfahren zur Herstellung von wirkstoffhaltigen Knochenzementen |
| WO1996011715A1 (fr) | 1994-10-13 | 1996-04-25 | Kuraray Co., Ltd. | Composition de reparation des tissus durs et son unite de distribution |
| AU713540B2 (en) | 1994-10-25 | 1999-12-02 | Osteonics Corp. | Interlocking structural elements and method for bone repair, augmentation and replacement |
| US6051751A (en) | 1995-01-20 | 2000-04-18 | Spire Corporation | Arthroplasty process for securely anchoring prostheses to bone, and arthroplasty products therefor |
| US5665122A (en) | 1995-01-31 | 1997-09-09 | Kambin; Parviz | Expandable intervertebral cage and surgical method |
| US6602248B1 (en) | 1995-06-07 | 2003-08-05 | Arthro Care Corp. | Methods for repairing damaged intervertebral discs |
| US5795922A (en) * | 1995-06-06 | 1998-08-18 | Clemson University | Bone cement composistion containing microencapsulated radiopacifier and method of making same |
| US6149688A (en) | 1995-06-07 | 2000-11-21 | Surgical Dynamics, Inc. | Artificial bone graft implant |
| US5648097A (en) | 1995-10-04 | 1997-07-15 | Biotek, Inc. | Calcium mineral-based microparticles and method for the production thereof |
| US6095149A (en) | 1996-08-13 | 2000-08-01 | Oratec Interventions, Inc. | Method for treating intervertebral disc degeneration |
| US5769880A (en) | 1996-04-12 | 1998-06-23 | Novacept | Moisture transport system for contact electrocoagulation |
| DE19618964C2 (de) | 1996-05-10 | 1999-12-16 | Implex Hear Tech Ag | Implantierbares Positionier- und Fixiersystem für aktorische und sensorische Implantate |
| US6620155B2 (en) | 1996-07-16 | 2003-09-16 | Arthrocare Corp. | System and methods for electrosurgical tissue contraction within the spine |
| US7357798B2 (en) | 1996-07-16 | 2008-04-15 | Arthrocare Corporation | Systems and methods for electrosurgical prevention of disc herniations |
| ATE260072T1 (de) | 1996-07-18 | 2004-03-15 | Implant Innovations Inc | Motorisch angetriebene osteotomiewerkzeuge zum verdichten von knochengewebe |
| FR2753368B1 (fr) | 1996-09-13 | 1999-01-08 | Chauvin Jean Luc | Cage d'osteosynthese expansive |
| EP0873145A2 (en) | 1996-11-15 | 1998-10-28 | Advanced Bio Surfaces, Inc. | Biomaterial system for in situ tissue repair |
| US5902839A (en) * | 1996-12-02 | 1999-05-11 | Northwestern University | Bone cement and method of preparation |
| DE19652608C1 (de) | 1996-12-18 | 1998-08-27 | Eska Implants Gmbh & Co | Prophylaxe-Implantat gegen Frakturen osteoporotisch befallener Knochensegmente |
| US5961554A (en) | 1996-12-31 | 1999-10-05 | Janson; Frank S | Intervertebral spacer |
| US5954716A (en) | 1997-02-19 | 1999-09-21 | Oratec Interventions, Inc | Method for modifying the length of a ligament |
| US6733515B1 (en) | 1997-03-12 | 2004-05-11 | Neomend, Inc. | Universal introducer |
| US5997580A (en) | 1997-03-27 | 1999-12-07 | Johnson & Johnson Professional, Inc. | Cement restrictor including shape memory material |
| US5972015A (en) | 1997-08-15 | 1999-10-26 | Kyphon Inc. | Expandable, asymetric structures for deployment in interior body regions |
| US6048346A (en) | 1997-08-13 | 2000-04-11 | Kyphon Inc. | Systems and methods for injecting flowable materials into bones |
| US6309420B1 (en) | 1997-10-14 | 2001-10-30 | Parallax Medical, Inc. | Enhanced visibility materials for implantation in hard tissue |
| AU1831999A (en) | 1997-12-18 | 1999-07-05 | Comfort Biomedical, Inc. | Bone augmentation for prosthetic implants and the like |
| US6236020B1 (en) | 1998-02-06 | 2001-05-22 | Joshua Friedman | Heating assembly for preheating dental materials |
| US6458375B1 (en) | 1998-02-27 | 2002-10-01 | Musculoskeletal Transplant Foundation | Malleable paste with allograft bone reinforcement for filling bone defects |
| US6348679B1 (en) | 1998-03-17 | 2002-02-19 | Ameritherm, Inc. | RF active compositions for use in adhesion, bonding and coating |
| US6719773B1 (en) | 1998-06-01 | 2004-04-13 | Kyphon Inc. | Expandable structures for deployment in interior body regions |
| WO1999062416A1 (en) | 1998-06-01 | 1999-12-09 | Kyphon Inc. | Expandable preformed structures for deployment in interior body regions |
| US7435247B2 (en) | 1998-08-11 | 2008-10-14 | Arthrocare Corporation | Systems and methods for electrosurgical tissue treatment |
| US6261289B1 (en) | 1998-10-26 | 2001-07-17 | Mark Levy | Expandable orthopedic device |
| CA2320097C (en) | 1998-12-09 | 2009-04-14 | Cook Incorporated | Hollow, curved, superelastic medical needle |
| US6709149B1 (en) | 1998-12-14 | 2004-03-23 | Ao Research Institute Davos | Method of bone cement preparation |
| US6436143B1 (en) | 1999-02-22 | 2002-08-20 | Anthony C. Ross | Method and apparatus for treating intervertebral disks |
| US6264659B1 (en) | 1999-02-22 | 2001-07-24 | Anthony C. Ross | Method of treating an intervertebral disk |
| US6395007B1 (en) | 1999-03-16 | 2002-05-28 | American Osteomedix, Inc. | Apparatus and method for fixation of osteoporotic bone |
| US6203844B1 (en) | 1999-04-01 | 2001-03-20 | Joon B. Park | Precoated polymeric prosthesis and process for making same |
| US6419704B1 (en) | 1999-10-08 | 2002-07-16 | Bret Ferree | Artificial intervertebral disc replacement methods and apparatus |
| ES2164548B1 (es) | 1999-08-05 | 2003-03-01 | Probitas Pharma Sa | Dispositivo para la dosificacion de masa fraguable para vertebroplastia y otros tratamientos oseos similares. |
| US6425919B1 (en) | 1999-08-18 | 2002-07-30 | Intrinsic Orthopedics, Inc. | Devices and methods of vertebral disc augmentation |
| CA2425951C (en) | 1999-08-18 | 2008-09-16 | Intrinsic Therapeutics, Inc. | Devices and method for nucleus pulposus augmentation and retention |
| US6649888B2 (en) | 1999-09-23 | 2003-11-18 | Codaco, Inc. | Radio frequency (RF) heating system |
| US6783515B1 (en) | 1999-09-30 | 2004-08-31 | Arthrocare Corporation | High pressure delivery system |
| US6458127B1 (en) | 1999-11-22 | 2002-10-01 | Csaba Truckai | Polymer embolic elements with metallic coatings for occlusion of vascular malformations |
| US6610079B1 (en) | 1999-12-14 | 2003-08-26 | Linvatec Corporation | Fixation system and method |
| US6872403B2 (en) | 2000-02-01 | 2005-03-29 | University Of Kentucky Research Foundation | Polymethylmethacrylate augmented with carbon nanotubes |
| US6740093B2 (en) | 2000-02-28 | 2004-05-25 | Stephen Hochschuler | Method and apparatus for treating a vertebral body |
| US6332894B1 (en) | 2000-03-07 | 2001-12-25 | Zimmer, Inc. | Polymer filled spinal fusion cage |
| US6425923B1 (en) | 2000-03-07 | 2002-07-30 | Zimmer, Inc. | Contourable polymer filled implant |
| US6447514B1 (en) | 2000-03-07 | 2002-09-10 | Zimmer | Polymer filled hip fracture fixation device |
| AR027685A1 (es) | 2000-03-22 | 2003-04-09 | Synthes Ag | Forma de tejido y metodo para realizarlo |
| ES2262642T3 (es) | 2000-04-05 | 2006-12-01 | Kyphon Inc. | Dispositivo para el tratamiento de huesos fracturados y/o enfermos. |
| US6821298B1 (en) | 2000-04-18 | 2004-11-23 | Roger P. Jackson | Anterior expandable spinal fusion cage system |
| CA2410637C (en) | 2000-05-31 | 2007-04-10 | Mnemoscience Gmbh | Shape memory polymers seeded with dissociated cells for tissue engineering |
| GB2363115A (en) | 2000-06-10 | 2001-12-12 | Secr Defence | Porous or polycrystalline silicon orthopaedic implants |
| US6964667B2 (en) | 2000-06-23 | 2005-11-15 | Sdgi Holdings, Inc. | Formed in place fixation system with thermal acceleration |
| US6899713B2 (en) | 2000-06-23 | 2005-05-31 | Vertelink Corporation | Formable orthopedic fixation system |
| US6620185B1 (en) | 2000-06-27 | 2003-09-16 | Smith & Nephew, Inc. | Surgical procedures and instruments |
| DE10032220A1 (de) | 2000-07-03 | 2002-01-24 | Sanatis Gmbh | Magnesium-ammonium-phosphat-Zemente, deren Herstellung und Verwendung |
| US6316885B1 (en) | 2000-07-18 | 2001-11-13 | General Electric Company | Single ballast for powering high intensity discharge lamps |
| ES2341641T3 (es) | 2000-07-21 | 2010-06-24 | The Spineology Group, Llc | Un dispositivo de bolsa de malla porosa expansible y su uso para cirugia osea. |
| NO312386B1 (no) | 2000-07-24 | 2002-04-29 | Abb Offshore Systems As | Arrangement og fremgangsmate for a installere en transformator pa sjobunnen |
| US6485436B1 (en) | 2000-08-10 | 2002-11-26 | Csaba Truckai | Pressure-assisted biopsy needle apparatus and technique |
| US20030069327A1 (en) | 2001-08-09 | 2003-04-10 | Uwe Walz | Dental compostions comprising bisacrylamides and use thereof |
| US6358254B1 (en) | 2000-09-11 | 2002-03-19 | D. Greg Anderson | Method and implant for expanding a spinal canal |
| US6312254B1 (en) | 2000-09-22 | 2001-11-06 | Joshua Friedman | Dispenser for heating and extruding dental material |
| US7045125B2 (en) | 2000-10-24 | 2006-05-16 | Vita Special Purpose Corporation | Biologically active composites and methods for their production and use |
| AU2002231355A1 (en) | 2000-10-24 | 2002-05-06 | Osteotech, Inc. | Injectable vertebral augmentation composition and method |
| EP1328203B1 (en) | 2000-10-25 | 2009-11-18 | Kyphon SÀRL | Systems for reducing fractured bone using a fracture reduction cannula |
| WO2002034378A2 (en) | 2000-10-25 | 2002-05-02 | Kyphon Inc. | Systems and methods for mixing and transferring flowable materials |
| US7306598B2 (en) | 2000-11-24 | 2007-12-11 | Dfine, Inc. | Polymer matrix devices for treatment of vascular malformations |
| US6958061B2 (en) | 2000-11-24 | 2005-10-25 | Csaba Truckai | Microspheres with sacrificial coatings for vaso-occlusive systems |
| JP4305594B2 (ja) | 2000-11-28 | 2009-07-29 | 株式会社トクヤマ | 歯科用接着キット |
| US6524102B2 (en) | 2000-12-08 | 2003-02-25 | Kerry N Davis | Method and apparatus for applying thermoplastic border molding to denture impression trays |
| US6622731B2 (en) | 2001-01-11 | 2003-09-23 | Rita Medical Systems, Inc. | Bone-treatment instrument and method |
| US6439439B1 (en) | 2001-01-12 | 2002-08-27 | Telios Orthopedic Systems, Inc. | Bone cement delivery apparatus and hand-held fluent material dispensing apparatus |
| ATE308278T1 (de) | 2001-01-26 | 2005-11-15 | Uab Research Foundation | Knochenzement |
| US6673113B2 (en) | 2001-10-18 | 2004-01-06 | Spinecore, Inc. | Intervertebral spacer device having arch shaped spring elements |
| US7008433B2 (en) | 2001-02-15 | 2006-03-07 | Depuy Acromed, Inc. | Vertebroplasty injection device |
| US6375659B1 (en) | 2001-02-20 | 2002-04-23 | Vita Licensing, Inc. | Method for delivery of biocompatible material |
| WO2002074195A2 (en) | 2001-03-19 | 2002-09-26 | Cambridge Polymer Group Inc. | System and methods for reducing interfacial porosity in cements |
| US20070191964A1 (en) | 2001-04-04 | 2007-08-16 | Arthrocare Corporation | Enhanced visibility materials for implantation in hard tissue |
| US20020147496A1 (en) | 2001-04-06 | 2002-10-10 | Integrated Vascular Systems, Inc. | Apparatus for treating spinal discs |
| US20040083002A1 (en) | 2001-04-06 | 2004-04-29 | Belef William Martin | Methods for treating spinal discs |
| US6632235B2 (en) | 2001-04-19 | 2003-10-14 | Synthes (U.S.A.) | Inflatable device and method for reducing fractures in bone and in treating the spine |
| US20020165582A1 (en) | 2001-04-26 | 2002-11-07 | Porter Christopher H. | Method and apparatus for delivering materials to the body |
| ATE419810T1 (de) | 2001-05-01 | 2009-01-15 | Amedica Corp | Röntgendurchlässiges knochentransplantat |
| WO2003002490A2 (en) | 2001-06-28 | 2003-01-09 | Wm. Marsh Rice University | Photocrosslinking of diethyl fumarate/poly(propylene fumarate) biomaterials |
| US6547432B2 (en) | 2001-07-16 | 2003-04-15 | Stryker Instruments | Bone cement mixing and delivery device for injection and method thereof |
| US6736815B2 (en) | 2001-09-06 | 2004-05-18 | Core Medical, Inc. | Apparatus and methods for treating spinal discs |
| US6706069B2 (en) | 2001-09-13 | 2004-03-16 | J. Lee Berger | Spinal grooved director with built in balloon |
| US20030130738A1 (en) | 2001-11-08 | 2003-07-10 | Arthrocare Corporation | System and method for repairing a damaged intervertebral disc |
| WO2003045274A2 (en) | 2001-11-21 | 2003-06-05 | Nuvasive, Inc. | Thermopolymer composition and related methods |
| US6780191B2 (en) | 2001-12-28 | 2004-08-24 | Yacmur Llc | Cannula system |
| US6723095B2 (en) | 2001-12-28 | 2004-04-20 | Hemodynamics, Inc. | Method of spinal fixation using adhesive media |
| CA2733282C (en) | 2002-01-22 | 2017-02-14 | Surgrx, Inc. | Electrosurgical instrument and method of use |
| ES2288578T3 (es) | 2002-05-29 | 2008-01-16 | Heraeus Kulzer Gmbh | Mezcla para cemento de huesos y agente de contraste de rayos x. |
| US7273523B2 (en) | 2002-06-07 | 2007-09-25 | Kyphon Inc. | Strontium-apatite-cement-preparations, cements formed therefrom, and uses thereof |
| WO2003105673A2 (en) | 2002-06-17 | 2003-12-24 | Trimedyne, Inc. | Devices and methods for minimally invasive treatment of degenerated spinal discs |
| GB0215916D0 (en) | 2002-07-10 | 2002-08-21 | Univ Dundee | Coatings |
| US7901407B2 (en) | 2002-08-02 | 2011-03-08 | Boston Scientific Scimed, Inc. | Media delivery device for bone structures |
| US6712852B1 (en) | 2002-09-30 | 2004-03-30 | Depuy Spine, Inc. | Laminoplasty cage |
| AU2003279506A1 (en) | 2002-11-12 | 2004-06-03 | Regenex Ltd. | Expandable devices and methods for tissue expansion, regenerationand fixation |
| US6979352B2 (en) | 2002-11-21 | 2005-12-27 | Depuy Acromed | Methods of performing embolism-free vertebroplasty and devices therefor |
| EP1592463B1 (en) | 2003-02-13 | 2006-08-16 | SYNTHES AG Chur | Injectable bone-replacement mixture |
| AU2004216181B2 (en) | 2003-02-21 | 2010-01-07 | Smith & Nephew, Inc. | Spinal fluid introduction |
| ATE488205T1 (de) | 2003-03-14 | 2010-12-15 | Depuy Spine Inc | Hydraulische vorrichtung zur knochenzementeinspritzung bei perkutaner vertebroplastie |
| US7824444B2 (en) | 2003-03-20 | 2010-11-02 | Spineco, Inc. | Expandable spherical spinal implant |
| US8066713B2 (en) | 2003-03-31 | 2011-11-29 | Depuy Spine, Inc. | Remotely-activated vertebroplasty injection device |
| GB2400935B (en) | 2003-04-26 | 2006-02-15 | Ibm | Configuring memory for a raid storage system |
| US20040267272A1 (en) | 2003-05-12 | 2004-12-30 | Henniges Bruce D | Bone cement mixing and delivery system |
| US7803395B2 (en) | 2003-05-15 | 2010-09-28 | Biomerix Corporation | Reticulated elastomeric matrices, their manufacture and use in implantable devices |
| US8415407B2 (en) | 2004-03-21 | 2013-04-09 | Depuy Spine, Inc. | Methods, materials, and apparatus for treating bone and other tissue |
| US20070032567A1 (en) * | 2003-06-17 | 2007-02-08 | Disc-O-Tech Medical | Bone Cement And Methods Of Use Thereof |
| US20050010231A1 (en) | 2003-06-20 | 2005-01-13 | Myers Thomas H. | Method and apparatus for strengthening the biomechanical properties of implants |
| US20050015148A1 (en) | 2003-07-18 | 2005-01-20 | Jansen Lex P. | Biocompatible wires and methods of using same to fill bone void |
| US7261717B2 (en) | 2003-09-11 | 2007-08-28 | Skeletal Kinetics Llc | Methods and devices for delivering orthopedic cements to a target bone site |
| WO2005039390A2 (en) | 2003-10-20 | 2005-05-06 | Arthrocare Corporation | Electrosurgical method and apparatus for removing tissue within a bone body |
| US6955691B2 (en) | 2003-11-21 | 2005-10-18 | Kyungwon Medical Co., Ltd. | Expandable interfusion cage |
| US20050113843A1 (en) | 2003-11-25 | 2005-05-26 | Arramon Yves P. | Remotely actuated system for bone cement delivery |
| US7189263B2 (en) | 2004-02-03 | 2007-03-13 | Vita Special Purpose Corporation | Biocompatible bone graft material |
| US8235256B2 (en) | 2004-02-12 | 2012-08-07 | Kyphon Sarl | Manual pump mechanism and delivery system |
| CN1988923B (zh) | 2004-04-27 | 2012-10-03 | 凯丰有限责任公司 | 骨替代组合物及其使用方法 |
| US20050245938A1 (en) | 2004-04-28 | 2005-11-03 | Kochan Jeffrey P | Method and apparatus for minimally invasive repair of intervertebral discs and articular joints |
| FR2870129A1 (fr) | 2004-05-14 | 2005-11-18 | Ceravic Sas Soc Par Actions Si | Ciment polymere pour la vertebroplastie percutanee |
| US20060095138A1 (en) | 2004-06-09 | 2006-05-04 | Csaba Truckai | Composites and methods for treating bone |
| CA2575699C (en) | 2004-07-30 | 2014-07-08 | Disc-O-Tech Medical Technologies Ltd. | Methods, materials and apparatus for treating bone and other tissue |
| US8038682B2 (en) | 2004-08-17 | 2011-10-18 | Boston Scientific Scimed, Inc. | Apparatus and methods for delivering compounds into vertebrae for vertebroplasty |
| US20080319445A9 (en) | 2004-08-17 | 2008-12-25 | Scimed Life Systems, Inc. | Apparatus and methods for delivering compounds into vertebrae for vertebroplasty |
| US20060106459A1 (en) | 2004-08-30 | 2006-05-18 | Csaba Truckai | Bone treatment systems and methods |
| DE602005025137D1 (de) | 2004-09-10 | 2011-01-13 | Kieran P Murphy | Zementabgabenadel |
| US20060229628A1 (en) | 2004-10-02 | 2006-10-12 | Csaba Truckai | Biomedical treatment systems and methods |
| US7678116B2 (en) | 2004-12-06 | 2010-03-16 | Dfine, Inc. | Bone treatment systems and methods |
| US7559932B2 (en) | 2004-12-06 | 2009-07-14 | Dfine, Inc. | Bone treatment systems and methods |
| US8048083B2 (en) | 2004-11-05 | 2011-11-01 | Dfine, Inc. | Bone treatment systems and methods |
| US7682378B2 (en) | 2004-11-10 | 2010-03-23 | Dfine, Inc. | Bone treatment systems and methods for introducing an abrading structure to abrade bone |
| US8562607B2 (en) | 2004-11-19 | 2013-10-22 | Dfine, Inc. | Bone treatment systems and methods |
| US8070753B2 (en) | 2004-12-06 | 2011-12-06 | Dfine, Inc. | Bone treatment systems and methods |
| US7722620B2 (en) | 2004-12-06 | 2010-05-25 | Dfine, Inc. | Bone treatment systems and methods |
| US20060122614A1 (en) | 2004-12-06 | 2006-06-08 | Csaba Truckai | Bone treatment systems and methods |
| US7717918B2 (en) | 2004-12-06 | 2010-05-18 | Dfine, Inc. | Bone treatment systems and methods |
| WO2006062916A2 (en) | 2004-12-06 | 2006-06-15 | Dfine, Inc. | Bone treatment systems and methods |
| ES2561493T3 (es) | 2005-02-22 | 2016-02-26 | Depuy Spine, Inc. | Materiales de tratamiento de hueso |
| EP2319440B1 (en) | 2005-02-22 | 2015-12-16 | Depuy Spine Inc. | Materials for treating bone |
| US7851189B2 (en) | 2005-03-07 | 2010-12-14 | Boston Scientific Scimed, Inc. | Microencapsulated compositions for endoluminal tissue engineering |
| US7959607B2 (en) | 2005-05-27 | 2011-06-14 | Stryker Corporation | Hand-held fluid delivery device with sensors to determine fluid pressure and volume of fluid delivered to intervertebral discs during discography |
| IL174347A0 (en) | 2005-07-31 | 2006-08-20 | Disc O Tech Medical Tech Ltd | Bone cement and methods of use thereof |
| US20070162043A1 (en) | 2005-09-01 | 2007-07-12 | Csaba Truckai | Methods for sensing retrograde flows of bone fill material |
| CA2637380A1 (en) | 2006-01-23 | 2007-08-02 | V-Bond Therapeutics, Llc, Known As Osseon Therapeutics, Inc. | Bone cement composite containing particles in a non-uniform spatial distribution and devices for implementation |
| US8496657B2 (en) | 2006-02-07 | 2013-07-30 | P Tech, Llc. | Methods for utilizing vibratory energy to weld, stake and/or remove implants |
| US20080103505A1 (en) | 2006-10-26 | 2008-05-01 | Hendrik Raoul Andre Fransen | Containment device for site-specific delivery of a therapeutic material and methods of use |
| WO2008097855A2 (en) | 2007-02-05 | 2008-08-14 | Dfine, Inc. | Bone treatment systems and methods |
| US9445854B2 (en) | 2008-02-01 | 2016-09-20 | Dfine, Inc. | Bone treatment systems and methods |
| EP2810664B1 (en) | 2008-02-28 | 2019-05-29 | Dfine, Inc. | Bone Cement Composition |
| US7968616B2 (en) | 2008-04-22 | 2011-06-28 | Kyphon Sarl | Bone cement composition and method |
| EP2402041B1 (en) | 2009-02-25 | 2017-10-18 | Kyoto University | Bone cement composition, bone cement composition kit, and method for forming bone cement cured body |
-
2009
- 2009-02-27 EP EP14173584.5A patent/EP2810664B1/en active Active
- 2009-02-27 JP JP2010548917A patent/JP5509098B2/ja active Active
- 2009-02-27 WO PCT/US2009/035549 patent/WO2009108893A2/en active Application Filing
- 2009-02-27 EP EP09715413.2A patent/EP2252336B1/en active Active
- 2009-02-27 ES ES09715413.2T patent/ES2483996T3/es active Active
- 2009-02-27 US US12/395,532 patent/US8487021B2/en active Active
-
2013
- 2013-06-19 US US13/921,479 patent/US9216195B2/en active Active
-
2015
- 2015-06-10 HK HK15105540.7A patent/HK1205005A1/xx unknown
- 2015-11-12 US US14/939,804 patent/US9821085B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP2810664A1 (en) | 2014-12-10 |
| WO2009108893A3 (en) | 2010-06-03 |
| EP2810664B1 (en) | 2019-05-29 |
| US20090247664A1 (en) | 2009-10-01 |
| US9216195B2 (en) | 2015-12-22 |
| ES2483996T3 (es) | 2014-08-08 |
| EP2252336B1 (en) | 2014-06-25 |
| US20160058906A1 (en) | 2016-03-03 |
| US9821085B2 (en) | 2017-11-21 |
| US8487021B2 (en) | 2013-07-16 |
| EP2252336A2 (en) | 2010-11-24 |
| JP2011514818A (ja) | 2011-05-12 |
| HK1205005A1 (en) | 2015-12-11 |
| US20140031450A1 (en) | 2014-01-30 |
| WO2009108893A2 (en) | 2009-09-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5509098B2 (ja) | 骨治療システムおよび方法 | |
| US10695117B2 (en) | Bone treatment systems and methods | |
| US11672579B2 (en) | Bone treatment systems and methods | |
| US20100030220A1 (en) | Bone treatment systems and methods | |
| US10080817B2 (en) | Bone treatment systems and methods | |
| US10278754B2 (en) | Bone treatment systems and methods | |
| US8562620B2 (en) | Bone treatment systems | |
| US8556910B2 (en) | Bone treatment systems and methods | |
| US20080188858A1 (en) | Bone treatment systems and methods |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120202 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20120202 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130910 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20131204 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20140224 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20140324 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5509098 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |