JP4383746B2 - 膨張性の糸状塞栓装置の製造方法 - Google Patents
膨張性の糸状塞栓装置の製造方法 Download PDFInfo
- Publication number
- JP4383746B2 JP4383746B2 JP2002592820A JP2002592820A JP4383746B2 JP 4383746 B2 JP4383746 B2 JP 4383746B2 JP 2002592820 A JP2002592820 A JP 2002592820A JP 2002592820 A JP2002592820 A JP 2002592820A JP 4383746 B2 JP4383746 B2 JP 4383746B2
- Authority
- JP
- Japan
- Prior art keywords
- carrier
- embolization
- polymer
- hydrogel
- dimensional shape
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 0 CC1S(CC2=*(C)*CC2(C)CI)**1C Chemical compound CC1S(CC2=*(C)*CC2(C)CI)**1C 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C70/00—Shaping composites, i.e. plastics material comprising reinforcements, fillers or preformed parts, e.g. inserts
- B29C70/68—Shaping composites, i.e. plastics material comprising reinforcements, fillers or preformed parts, e.g. inserts by incorporating or moulding on preformed parts, e.g. inserts or layers, e.g. foam blocks
- B29C70/74—Moulding material on a relatively small portion of the preformed part, e.g. outsert moulding
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
- A61B17/12113—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
- A61B17/12113—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm
- A61B17/12118—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm for positioning in conjunction with a stent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
- A61B17/12145—Coils or wires having a pre-set deployed three-dimensional shape
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
- A61B17/12154—Coils or wires having stretch limiting means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/12163—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a string of elements connected to each other
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/12181—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device formed by fluidized, gelatinous or cellular remodelable materials, e.g. embolic liquids, foams or extracellular matrices
- A61B17/1219—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device formed by fluidized, gelatinous or cellular remodelable materials, e.g. embolic liquids, foams or extracellular matrices expandable in contact with liquids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/02—Inorganic materials
- A61L31/022—Metals or alloys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/145—Hydrogels or hydrocolloids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/18—Materials at least partially X-ray or laser opaque
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B2017/00004—(bio)absorbable, (bio)resorbable, resorptive
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B2017/00526—Methods of manufacturing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B2017/1205—Introduction devices
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/36—Materials or treatment for tissue regeneration for embolization or occlusion, e.g. vaso-occlusive compositions or devices
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29L—INDEXING SCHEME ASSOCIATED WITH SUBCLASS B29C, RELATING TO PARTICULAR ARTICLES
- B29L2031/00—Other particular articles
- B29L2031/753—Medical equipment; Accessories therefor
- B29L2031/7542—Catheters
Description
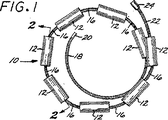
図1は、本発明の第1の好ましい具体例に従う脈管塞栓形成装置の立面図である。
図2は、図1の線2−2を切断した断面図である。
図3は、図2の線3−3を切断した断面図である。
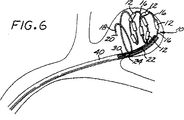
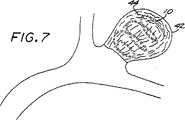
図4〜図7は、本発明の塞栓形成方法の一具体例に従って脈管部位(具体的には動脈瘤)を閉鎖させる方法の段階を示す概略図である。
図8は、本発明の塞栓形成装置が好ましくは配置器具の遠位末端部に装着される機構の詳細な透視図である。
図9は、図8と同様に、配置器具から分離された後の本発明の塞栓形成装置を示す詳細な透視図である。
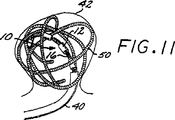
図10、11及び12は、図4〜7に例示されるものに加えて、本発明の塞栓形成方法の好ましい具体例に従って脈管部位を閉鎖させる方法を継続する段階を示す概略図である。
図13は、本発明の塞栓形成方法の別の具体例に従って脈管部位を閉鎖させる方法の段階を示す概略図である。
図14は、通常の又は膨張していない状態の装置を示す本発明の第2の好ましい具体例に従う塞栓性装置の部分断面正面図である。
図15は、図14の線15−15を切断した断面図である。
図16は、図14に示される装置の一部分の詳細な横断面図である。
図17は、脈管部位に配置された後の膨張した状態の図14の装置を示す図16と同様の図である。
図18は、脈管部位に配置された後の膨張した状態の図14の装置を示す図15と同様の図である。
図19は、通常の又は膨張していない状態の装置を示す本発明の第2の好ましい具体例に従う塞栓性装置の第1の改変型の部分横断面図である。
図20は、脈管部位内に置かれた後の膨張した状態の図19の装置を示す図19と同様の図である。
図21は、通常の又は膨張していない状態の装置を示す本発明の第2の好ましい具体例に従う塞栓性装置の第2の改変型の部分横断面図である。
図22は、脈管部位に配置された後の膨張した状態の図21の装置を示す図21と同様の図である。
図23は、本発明の第2の好ましい具体例に従う塞栓性装置の第3の改変型の詳細な横断面図である。
図24は、本発明に従う塞栓形成装置の第3の典型具体例を製造するための第1の典型方法に従う膨張した軟質親水性重合体塞栓形成要素の断面正面図である。
図25は、管状ホルダーに挿入された図24の塞栓形成要素の断面正面図である。
図26は、内腔心棒によって内部で支持された螺旋状キャリヤーによって同軸方向に串刺しにされつつある図25の塞栓形成要素の断面正面図である。
図27は、螺旋状キャリヤー及び内腔心棒によって完全に串刺しにされた後の図26の塞栓形成要素の断面正面図である。
図28は、本発明の塞栓形成装置の第3の典型具体例に従う未完成の塞栓形成装置を画定するために管状ホルダーから押し出されつつある図27の串刺しにされた塞栓形成要素の断面正面図である。
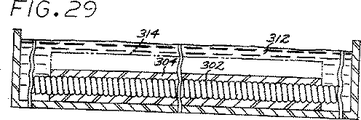
図29は、塞栓形成要素を収縮させるために乾燥浴中で脱水されつつある図28の塞栓形成装置の断面正面図である。
図30は、塞栓形成要素の水和速度を水性環境の物理的パラメーターのレベルに応じて調整するために酸浴中に浸漬された図29の塞栓形成装置の断面正面図である。
図31は、塞栓形成要素を乾燥させるためにオーブン中で加熱乾燥されつつある図30の塞栓形成装置の断面正面図である。
図32は、塞栓形成装置の第3の典型具体例に従う、図26の内腔心棒が所定位置に残った状態の完成塞栓形成装置の正面図である。
図33は、図32の塞栓形成装置の断面正面図である。
図34は、図33の線34−34を切断した断面によって明らかにされるような図33の塞栓形成装置の部分拡大断面図である。
図35は、塞栓形成装置から心棒を除去することによって該装置中に画定された軸方向溜めを示す図34と同様の図である。
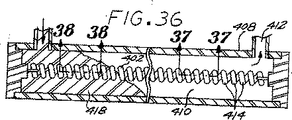
図36は、本発明に従う塞栓形成装置の製造方法の第2の典型具体例に従って成形されつつある本発明に従う塞栓形成装置の第4の典型具体例の断面正面図である。
図37は、図36の塞栓形成装置のキャリヤーを示す線37−37を切断した断面図によって明らかにされるような図36の初期の塞栓形成装置の部分拡大断面図である。
図38は、重合体中に包封されたキャリヤーを示す図37と同様の部分拡大断面図である。
図39は、塞栓形成装置の第4の典型具体例の一つの変形例の正面図である。
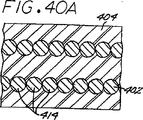
図40Aは、螺旋状に巻き付けられたキャリヤーのコイルが互いに隙間なく配置された塞栓形成装置の一つの見込まれる変形例を示す図39の線40−40を切断した断面によって明らかにされるような図39の塞栓形成装置の部分拡大断面図である。
図40Bは、キャリヤーのコイルが互いに間隔をあけて配置された別の変形例を示す図40Aと同様の図である。
図41は、塞栓形成装置のキャリヤー内の内腔心棒を示す塞栓装置の第4の典型具体例の別の変形例の正面図である。
図42Aは、螺旋状に巻き付けられたキャリヤーのコイルが互いに隙間なく配置され、しかも内腔心棒が取り除かれてキャリヤー内に軸上溜めが画定された塞栓形成装置の一つの見込まれる変形例を示す図41の線42−42を切断した断面によって明らかにされるような図41の塞栓形成装置の部分拡大断面図である。
図42Bは、キャリヤーのコイルが互いに間隔をあけて配置された別の変形例を示す図42Aと同様の図である。
図43は、塞栓形成装置の可撓性を測定するための方法及び装置の略正面図である。
図44は、図43中の丸で囲まれた細部44によって明らかにされるような図43で測定されている塞栓形成装置の部分拡大断面図である。
塞栓形成装置:第1の好ましい具体例
本発明の第1の好ましい具体例に従う脈管塞栓形成装置10を図1、2及び3に示す。この好ましい具体例では、塞栓形成装置10は複数の塞栓形成体を含み、それぞれは実質的に円筒形の「マイクロペレット」12として構成され、このマイクロペレット12は糸状キャリヤー14に沿って間隔をあけて設置されている。マイクロペレット12の数は、キャリヤー14の長さに依存して変化し、これは、同様に、閉鎖されるべき脈管部位の寸法に依存するであろう。大きな脈管部位については、例えば、8〜12個のマイクロペレットが使用できるが、必要ならばさらに多数を使用してもよい。いくらかの用途では(例えば非常に小さい動脈瘤)、1又は2個程度のマイクロペレットが使用できる。
図14〜23は、本発明の第2の好ましい具体例に従う塞栓形成装置を例示している。最初に図14〜17を参照すると、この第2の具体例に従う装置100は、細長く可撓性のある糸状キャリヤー102を含み、このものには膨張性塞栓形成要素104が取り外せないように保持されている。キャリヤー102は、好ましくは、白金、金、タングステン若しくはタンタルのような好適な金属又はステンレス鋼若しくはニチノールのような合金から作られた連続長の中空マイクロコイル106から形成される。これらの材料のうち、白金及びニチノールが好ましい。このマイクロコイルは、ぴったりと巻き付けられたコイルで形成されているため、隣接するコイルの間には間隔が殆ど又は全くない。また、キャリヤー102は、マイクロコイル106に沿って軸方向に延びる糸状心線108も含み得る。心線108は、好ましくはニチノールのような形状記憶金属から作られた薄い金属製ワイヤーである。装置100は、マイクロコイル106を同軸に包囲し且つ丸みのある遠位先端部112で終端する外側コイルからなる遠位部分を含む。上に記載され且つ図8及び9に例示されたタイプのヒドロゲル結合要素(示さない)は、有利には、キャリヤーの近位端部に備えられる。
体腔を閉塞させるための装置300の第3の典型具体例を図32〜35に例示し、装置300の第3の具体例を製造するための方法の第1の典型具体例を図24〜31に例示する。
体腔を閉塞させるための塞栓形成装置400の第4の典型具体例を図39〜42に例示し、装置400の第4の具体例を製造するための方法の第2の典型具体例を図36〜38に例示する。
塞栓形成装置10(第1の好ましい具体例)又は塞栓形成装置100(第2の好ましい具体例)のいずれかを使用して血管部位を閉鎖させる一方法を図4〜7に例示する。この方法を第1の好ましい具体例の塞栓装置10に関して記載するが、この方法は、第2の好ましい具体例の装置100にも同様に適用できることが分かるであろう。
12 マイクロペレット
14 キャリヤー
20 遠位保持部材
24 ヒドロゲル結合要素
30 配置器具
32 外側部分
34 ワイヤー心線
36 遠位部分
40 マイクロカテーテル
42 動脈瘤
50 血管塞栓性装置
60 血管内装置
100 塞栓形成装置
104 糸状キャリヤー
104 膨張性塞栓形成要素
106 中空マイクロコイル
108 糸状心線
101’ 塞栓装置
106' 中空マイクロコイル
108' 中央軸心線
100" 塞栓装置
300 塞栓形成装置
302 糸状キャリヤー
304 同軸重合体部材
306 軸方向溜め
308 管状ホルダー
316 支持心棒
320 オーブン
400 塞栓形成装置
402 糸状キャリヤー
404 同軸部材
408 型
410 空洞
422 第1水平表面
424 張り出し部分
426 第2水平表面
428 高さ
430 非支持末端部
432 水平距離
434 支持末端部
Claims (45)
- 次の工程:
拘束されていないときに三次元形状を形成する、細長く可撓性のある糸状キャリヤーを準備し、及び
該キャリヤーの全長の少なくとも一部分を膨張性の親水性重合体であってその膨張が周囲の生理学的pHにより誘発されるもの中に同軸に包封すること
を含む体腔閉塞装置の製造方法。 - キャリヤーの包封が、重合体の細長い部材を軟化された状態で準備し、及び該部材をキャリヤーで同軸方向に串刺しにすることからなる、請求項1に記載の方法。
- 細長い部材の準備が、該部材を管状ホルダーに該部材が径方向に閉じ込められ且つその中に同軸方向に拘束されるように挿入することからなる、請求項2に記載の方法。
- キャリヤーの包封が、中に細長い空洞を有する型を準備し、該型の空洞内にキャリヤーを同軸方向に配置し、及び該空洞に重合体を該重合体が該キャリヤーの全長の少なくとも一部分を同軸に包封する部材に成形されるように移すことからなる、請求項1に記載の方法。
- キャリヤーが軸方向内腔を含み、しかもキャリヤーの包封が該キャリヤーの包封部分の内腔を重合体で実質的に満たすことからなる、請求項1に記載の方法。
- キャリヤーが軸方向内腔を含み、しかも該内腔に軸方向溜めを形成させることをさらに含む、請求項1に記載の方法。
- 軸方向溜めの形成が、キャリヤーの内腔に細長い心棒を挿入してからこれを包封し、及びその包封後に該心棒をキャリヤーの内腔から取り出すことからなる、請求項6に記載の方法。
- 体腔を閉塞させるための装置であって、
拘束されていないときに三次元形状を形成する、細長く可撓性のある糸状キャリヤーと、
該キャリヤーの全長の少なくとも一部分を同軸に包封した膨張性の親水性重合体の部材
と
を含み、ここで、該親水性重合体は、周囲の生理学的pHレベルによりその膨張速度が変化するものである、体腔を閉塞させるための装置。 - キャリヤーが軸方向内腔を含み、しかも部材の重合体が該キャリヤーの包封部分の内腔
を実質的に満たす、請求項8に記載の装置。 - キャリヤーが中に軸方向溜めを有する軸方向内腔を含む請求項8に記載の装置。
- キャリヤーが可撓性ワイヤー、螺旋状コイル及びチューブよりなる群から選択される糸状要素を含む請求項8に記載の装置。
- 同軸部材が円筒形である請求項8に記載の装置。
- 同軸部材がヒドロゲルからなる請求項8に記載の装置。
- 同軸部材が生分解性及び生体吸収性のうちの少なくとも一つである請求項8に記載の装置。
- 同軸部材が乾燥したときよりも水和したときに実質的により大きい潤滑性を有する請求項8に記載の装置。
- 空洞を閉塞させるための装置であって、
生体適合性材料から形成された細長い糸状キャリヤーであって、該キャリヤーに三次元形状のバイアスをかける弾性記憶及び外部表面を有するものと、
該キャリヤーの外部表面のかなりの部分を覆うように該キャリヤー上に形成された膨張性ヒドロゲルの同軸部材と
を含み、
該装置が、該ヒドロゲルが脱水されたときよりも該ヒドロゲルが水和したときにより大きい可撓性及びより大きい潤滑性のうち少なくとも一つを有すること、及び
該ヒドロゲルが血液のpHレベルにより誘発される容積膨張を受けること
を特徴とする、空洞を閉塞させるための装置。 - 装置の一部分の非支持末端部が該部分の重量を受けて該部分の反対側の支持末端部に対して、
ヒドロゲルが乾燥状態にあって該部分の双方の末端部間の水平距離が2.25インチ(5 7.2mm)以上であるとき、
ヒドロゲルが中程度に水和した状態にあって該水平距離が1.5インチ(38.2mm)〜2.25インチ(57.2mm)の間にあるとき、及び
ヒドロゲルが完全に水和した状態にあって該水平距離が1.5インチ(38.2mm)未満であるときに
0.75インチ(19.1mm)下方に撓むことを特徴とする、請求項16に記載の装置。 - 体腔を閉塞させるための装置であって、
拘束されていないときに三次元形状をとる、外部表面を有する細長い糸状キャリヤーと、
該キャリヤーの外部表面のかなりの部分を覆う親水性重合体の同軸部材と
を含み、
該同軸部材の水性環境中での物理的性質が該環境中での時間及び該環境のpHレベルの関数である
ことを特徴とする、体腔を閉塞させるための装置。 - 物理的性質が部材の可撓性及び潤滑性のうちの少なくとも一つであり、しかも環境の物理的パラメーターが該環境の温度及びpHのうちの少なくとも一つである、請求項18に記載の装置。
- 遠位先端部及び外部表面を有する可撓性のある糸状キャリヤーであって拘束されていないときに三次元形状を形成するものと、該遠位先端部の近位に該キャリヤーの全長のかなりの部分に沿って該キャリヤーの該外部表面に取り外せないように固定された細長く連続した同軸塞栓形成要素であって血液にさらされたときに容積膨張を生じるプロトン化を受けるものとを含む脈管塞栓形成装置。
- 塞栓形成要素が耐伸長性の生体適合性重合体から形成された請求項20に記載の塞栓形成装置。
- 塞栓形成要素が膨張性の親水性重合体から形成された請求項20に記載の塞栓形成装置。
- キャリヤーがX線下で視覚化できる材料を含む請求項20に記載の塞栓形成装置。
- 重合体が環境に感受性のある重合体であって温度及びpHよりなる群から選択される環境パラメーターの変化に応答してその物理的特性を変化させるものである請求項22に記載の塞栓形成装置。
- 塞栓形成要素が2倍〜100倍の間の容積膨張係数によって膨張できる請求項22に記載の塞栓形成装置。
- 塞栓形成要素が放射線不透過性である請求項20に記載の塞栓形成装置。
- キャリヤーが拘束されていないときに三次元形状をとり得るマルチループ構造の形で配置された請求項20に記載の塞栓形成装置。
- 三次元形状が円形、螺旋形及び卵形よりなる群から選択される請求項27に記載の塞栓形成装置。
- 細長く可撓性のある糸状キャリヤーであって拘束されていないときに三次元形状を形成するものと、該キャリヤーに取り外せないように固定され、生理学的pHレベルに応答して増加する速度で膨張する膨張性重合体から形成された塞栓形成要素とを含む、脈管塞栓形成装置。
- 重合体が多孔性ヒドロゲルである請求項29に記載の塞栓形成装置。
- 塞栓形成要素がキャリヤーの外部表面に該キャリヤーの全長のかなりの部分に沿って取り外せないように固定された細長く連続した同軸塞栓形成要素からなる請求項29に記載の塞栓形成装置。
- 塞栓形成要素がキャリヤーに取り付けられた複数の膨張性重合体繊維からなる請求項29に記載の塞栓形成装置。
- 塞栓形成要素が放射線不透過性である請求項29に記載の塞栓形成装置。
- キャリヤーが拘束されていないときに三次元形状をとり得るマルチループ構造の形で配置された請求項29に記載の塞栓形成装置。
- 三次元形状が円形、螺旋形及び卵形よりなる群から選択される請求項34に記載の塞栓形成装置。
- 次の構成:
弾性記憶を有する所定の長さのワイヤーから形成され且つ最初に一部分がループ構造を形成する状態で配置され、それによって三次元形状をとる可撓性の糸状キャリヤーと、
該キャリヤーの周囲に同軸に配置され且つそれに取り外せないように取り付けられた塞栓形成要素であって、その少なくとも一部分が、周囲の生理学的pHレベルによって誘発される膨張を受ける親水性重合体から形成されているものと
を含む、糸状塞栓性装置。 - キャリヤーが連続長のマイクロコイルからなる請求項36に記載の装置。
- 塞栓形成要素がキャリヤーに塗布された被覆剤を含む請求項36に記載の装置。
- 被覆剤がキャリヤー全長の少なくとも一部分を包封する請求項38に記載の装置。
- 次の構成:
拘束されていないときに三次元形状をとる可撓性の糸状キャリヤーであって外部表面と遠位先端部とを有するものと、
該キャリヤーの外部表面にその近位先端部から近接した位置で取り外せないように固定された耐伸長性の塞栓形成要素であって、少なくとも一部分が、血液のpHを有する環境により誘発される膨張速度の変化を受ける親水性重合体から形成されたものと
を含む脈管塞栓形成装置。 - 次の構成:
可撓性のある重合体フィラメントと該フィラメントを同軸に取り囲むマイクロコイルとからなる所定の長さのキャリヤーであって拘束されていないときに三次元形状を形成するものと、
該キャリヤー上に同軸に配置され且つそれに取り外せないように取り付けられた重合体塞栓形成要素であって、該キャリヤーの全長の少なくとも一部分を事実上連続的に覆い、かつ、血液によりプロトン化されたときに膨張するものと
を含む、脈管部位を閉鎖させるための装置。 - 塞栓形成要素が親水性重合体からなる請求項41に記載の装置。
- マイクロコイルが少なくとも部分的に白金から作られた請求項41に記載の装置。
- キャリヤーが拘束されていないときに螺旋形、円形及び卵形よりなる群から選択される形状をとるループ構造に形成されている請求項41に記載の装置。
- 塞栓形成要素が耐伸長性である請求項41に記載の装置。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/867,340 US6602261B2 (en) | 1999-10-04 | 2001-05-29 | Filamentous embolic device with expansile elements |
| US10/157,621 US7014645B2 (en) | 1999-10-04 | 2002-05-29 | Method of manufacturing expansile filamentous embolization devices |
| PCT/US2002/016873 WO2002096302A1 (en) | 2001-05-29 | 2002-05-29 | Method of manufacturing expansile filamentous embolization devices |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2004527342A JP2004527342A (ja) | 2004-09-09 |
| JP2004527342A5 JP2004527342A5 (ja) | 2006-01-05 |
| JP4383746B2 true JP4383746B2 (ja) | 2009-12-16 |
Family
ID=26854315
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2002592820A Expired - Fee Related JP4383746B2 (ja) | 2001-05-29 | 2002-05-29 | 膨張性の糸状塞栓装置の製造方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (4) | US7014645B2 (ja) |
| EP (2) | EP2319430B1 (ja) |
| JP (1) | JP4383746B2 (ja) |
| AT (1) | ATE516759T1 (ja) |
| AU (3) | AU2002344223B2 (ja) |
| CA (2) | CA2449055C (ja) |
| ES (1) | ES2441341T3 (ja) |
| WO (1) | WO2002096302A1 (ja) |
Families Citing this family (222)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8795332B2 (en) | 2002-09-30 | 2014-08-05 | Ethicon, Inc. | Barbed sutures |
| US6176240B1 (en) * | 1995-06-07 | 2001-01-23 | Conceptus, Inc. | Contraceptive transcervical fallopian tube occlusion devices and their delivery |
| US6705323B1 (en) | 1995-06-07 | 2004-03-16 | Conceptus, Inc. | Contraceptive transcervical fallopian tube occlusion devices and methods |
| US5931855A (en) | 1997-05-21 | 1999-08-03 | Frank Hoffman | Surgical methods using one-way suture |
| US6015424A (en) * | 1998-04-28 | 2000-01-18 | Microvention, Inc. | Apparatus and method for vascular embolization |
| US7044134B2 (en) | 1999-11-08 | 2006-05-16 | Ev3 Sunnyvale, Inc | Method of implanting a device in the left atrial appendage |
| US7128073B1 (en) | 1998-11-06 | 2006-10-31 | Ev3 Endovascular, Inc. | Method and device for left atrial appendage occlusion |
| US6309384B1 (en) | 1999-02-01 | 2001-10-30 | Adiana, Inc. | Method and apparatus for tubal occlusion |
| US8702727B1 (en) | 1999-02-01 | 2014-04-22 | Hologic, Inc. | Delivery catheter with implant ejection mechanism |
| US6280457B1 (en) * | 1999-06-04 | 2001-08-28 | Scimed Life Systems, Inc. | Polymer covered vaso-occlusive devices and methods of producing such devices |
| US8048104B2 (en) | 2000-10-30 | 2011-11-01 | Dendron Gmbh | Device for the implantation of occlusion spirals |
| US8313504B2 (en) | 2000-09-18 | 2012-11-20 | Cordis Corporation | Foam matrix embolization device |
| US7033374B2 (en) * | 2000-09-26 | 2006-04-25 | Microvention, Inc. | Microcoil vaso-occlusive device with multi-axis secondary configuration |
| US6878384B2 (en) | 2001-03-13 | 2005-04-12 | Microvention, Inc. | Hydrogels that undergo volumetric expansion in response to changes in their environment and their methods of manufacture and use |
| US7056331B2 (en) | 2001-06-29 | 2006-06-06 | Quill Medical, Inc. | Suture method |
| US7572288B2 (en) * | 2001-07-20 | 2009-08-11 | Microvention, Inc. | Aneurysm treatment device and method of use |
| CN101919722A (zh) | 2002-07-31 | 2010-12-22 | 微温森公司 | 三部件同轴的血管闭塞装置 |
| US6773450B2 (en) | 2002-08-09 | 2004-08-10 | Quill Medical, Inc. | Suture anchor and method |
| US8100940B2 (en) | 2002-09-30 | 2012-01-24 | Quill Medical, Inc. | Barb configurations for barbed sutures |
| US20040088003A1 (en) * | 2002-09-30 | 2004-05-06 | Leung Jeffrey C. | Barbed suture in combination with surgical needle |
| US20040122349A1 (en) | 2002-12-20 | 2004-06-24 | Lafontaine Daniel M. | Closure device with textured surface |
| US8709038B2 (en) * | 2002-12-20 | 2014-04-29 | Boston Scientific Scimed, Inc. | Puncture hole sealing device |
| KR100721049B1 (ko) * | 2003-01-10 | 2007-05-23 | 가부시키가이샤 가네카 | 색전 형성용 체내 유치 코일 |
| JP2004261218A (ja) * | 2003-02-07 | 2004-09-24 | Yasuhiko Tabata | 血管塞栓用組成物 |
| US8043321B2 (en) * | 2003-07-24 | 2011-10-25 | Boston Scientific Scimed, Inc. | Embolic coil |
| US7371228B2 (en) * | 2003-09-19 | 2008-05-13 | Medtronic Vascular, Inc. | Delivery of therapeutics to treat aneurysms |
| US20050107867A1 (en) * | 2003-11-17 | 2005-05-19 | Taheri Syde A. | Temporary absorbable venous occlusive stent and superficial vein treatment method |
| US20080109057A1 (en) * | 2003-12-10 | 2008-05-08 | Calabria Marie F | Multiple point detacher system |
| DE102004003265A1 (de) | 2004-01-21 | 2005-08-11 | Dendron Gmbh | Vorrichtung zur Implantation von elektrisch isolierten Okklusionswendeln |
| US8048101B2 (en) | 2004-02-25 | 2011-11-01 | Femasys Inc. | Methods and devices for conduit occlusion |
| US8052669B2 (en) | 2004-02-25 | 2011-11-08 | Femasys Inc. | Methods and devices for delivery of compositions to conduits |
| US9238127B2 (en) | 2004-02-25 | 2016-01-19 | Femasys Inc. | Methods and devices for delivering to conduit |
| US8048086B2 (en) | 2004-02-25 | 2011-11-01 | Femasys Inc. | Methods and devices for conduit occlusion |
| US20050216049A1 (en) * | 2004-03-29 | 2005-09-29 | Jones Donald K | Vascular occlusive device with elastomeric bioresorbable coating |
| US7416757B2 (en) * | 2004-04-08 | 2008-08-26 | Cordis Neurovascular, Inc. | Method of making active embolic coil |
| US8801746B1 (en) | 2004-05-04 | 2014-08-12 | Covidien Lp | System and method for delivering a left atrial appendage containment device |
| JP5340593B2 (ja) | 2004-05-14 | 2013-11-13 | エシコン・エルエルシー | 縫合方法および装置 |
| US20050267510A1 (en) * | 2004-05-26 | 2005-12-01 | Nasser Razack | Device for the endovascular treatment of intracranial aneurysms |
| US7896879B2 (en) * | 2004-07-29 | 2011-03-01 | Vertos Medical, Inc. | Spinal ligament modification |
| US20060052822A1 (en) * | 2004-08-31 | 2006-03-09 | Mirizzi Michael S | Apparatus and material composition for permanent occlusion of a hollow anatomical structure |
| WO2006032291A1 (de) | 2004-09-22 | 2006-03-30 | Dendron Gmbh | Vorrichtung zur implantation von mikrowendeln |
| WO2006032289A1 (de) | 2004-09-22 | 2006-03-30 | Dendron Gmbh | Medizinisches implantat |
| US7201918B2 (en) * | 2004-11-16 | 2007-04-10 | Microvention, Inc. | Compositions, systems and methods for treatment of defects in blood vessels |
| US8425550B2 (en) * | 2004-12-01 | 2013-04-23 | Boston Scientific Scimed, Inc. | Embolic coils |
| US20060206139A1 (en) * | 2005-01-19 | 2006-09-14 | Tekulve Kurt J | Vascular occlusion device |
| US7972354B2 (en) | 2005-01-25 | 2011-07-05 | Tyco Healthcare Group Lp | Method and apparatus for impeding migration of an implanted occlusive structure |
| US20060178696A1 (en) * | 2005-02-04 | 2006-08-10 | Porter Stephen C | Macroporous materials for use in aneurysms |
| US20070001346A1 (en) | 2005-06-30 | 2007-01-04 | Murty Vyakarnam | Active embolization device |
| EP1912686B1 (en) * | 2005-07-11 | 2009-08-19 | DSMIP Assets B.V. | Coiled wire for the controlled release of dugs to the eye |
| US8696671B2 (en) | 2005-07-29 | 2014-04-15 | Vertos Medical Inc. | Percutaneous tissue excision devices |
| US7972359B2 (en) | 2005-09-16 | 2011-07-05 | Atritech, Inc. | Intracardiac cage and method of delivering same |
| US20070082021A1 (en) * | 2005-09-30 | 2007-04-12 | Bates Brian L | Coated vaso-occlusion device |
| US20070123890A1 (en) * | 2005-11-04 | 2007-05-31 | X-Sten, Corp. | Tissue retrieval devices and methods |
| US7927676B2 (en) * | 2006-03-31 | 2011-04-19 | Codman & Shurtleff, Inc. | Plasma-treated vascular occlusion devices and methods |
| US20070239194A1 (en) * | 2006-04-05 | 2007-10-11 | Boston Scientific Scimed, Inc. | Vaso-occlusive devices having expandable fibers |
| CN101448464B (zh) | 2006-04-17 | 2011-05-04 | 微治疗公司 | 用于以机械方式定位血管内植入物的系统和方法 |
| US8777979B2 (en) | 2006-04-17 | 2014-07-15 | Covidien Lp | System and method for mechanically positioning intravascular implants |
| US9017361B2 (en) | 2006-04-20 | 2015-04-28 | Covidien Lp | Occlusive implant and methods for hollow anatomical structure |
| US7942830B2 (en) | 2006-05-09 | 2011-05-17 | Vertos Medical, Inc. | Ipsilateral approach to minimally invasive ligament decompression procedure |
| US8377091B2 (en) * | 2006-06-15 | 2013-02-19 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US20070299461A1 (en) * | 2006-06-21 | 2007-12-27 | Boston Scientific Scimed, Inc. | Embolic coils and related components, systems, and methods |
| USD620593S1 (en) | 2006-07-31 | 2010-07-27 | Vertos Medical, Inc. | Tissue excision device |
| WO2008100906A2 (en) * | 2007-02-12 | 2008-08-21 | Vertos Medical, Inc. | Tissue excision devices and methods |
| US8801747B2 (en) * | 2007-03-13 | 2014-08-12 | Covidien Lp | Implant, a mandrel, and a method of forming an implant |
| KR20100015520A (ko) | 2007-03-13 | 2010-02-12 | 마이크로 테라퓨틱스 인코포레이티드 | 코일과 펴짐방지 부재를 포함하는 임플란트 |
| US20080255612A1 (en) * | 2007-04-13 | 2008-10-16 | Angiotech Pharmaceuticals, Inc. | Self-retaining systems for surgical procedures |
| WO2008147861A1 (en) * | 2007-05-22 | 2008-12-04 | Wilson-Cook Medical Inc. | Embolization coil delivery system |
| US20110022149A1 (en) | 2007-06-04 | 2011-01-27 | Cox Brian J | Methods and devices for treatment of vascular defects |
| US8100129B2 (en) * | 2007-08-28 | 2012-01-24 | Conceptus, Inc. | Methods and devices for occluding an ovarian pathway |
| EP2526975B1 (en) | 2007-09-27 | 2014-06-04 | Ethicon, LLC | Self-retaining sutures including tissue retainers having improved strength |
| WO2009052432A2 (en) | 2007-10-19 | 2009-04-23 | Coherex Medical, Inc. | Medical device for modification of left atrial appendange and related systems and methods |
| US20090118709A1 (en) * | 2007-11-06 | 2009-05-07 | Vertos Medical, Inc. A Delaware Corporation | Tissue Excision Tool, Kits and Methods of Using the Same |
| ES2397010T3 (es) * | 2007-12-03 | 2013-03-04 | Stryker Corporation | Dispositivo implantable con unión separable electrolíticamente que tiene múltiples cables delgados |
| US8916077B1 (en) | 2007-12-19 | 2014-12-23 | Ethicon, Inc. | Self-retaining sutures with retainers formed from molten material |
| JP5518737B2 (ja) | 2007-12-19 | 2014-06-11 | エシコン・エルエルシー | 熱接触媒介リテーナを備えた留置縫合糸 |
| US8118834B1 (en) | 2007-12-20 | 2012-02-21 | Angiotech Pharmaceuticals, Inc. | Composite self-retaining sutures and method |
| US8192480B2 (en) | 2007-12-21 | 2012-06-05 | Microvention, Inc. | System and method of detecting implant detachment |
| US8470035B2 (en) * | 2007-12-21 | 2013-06-25 | Microvention, Inc. | Hydrogel filaments for biomedical uses |
| AU2008340276B2 (en) | 2007-12-21 | 2014-08-07 | Microvention, Inc. | System and method for locating detachment zone of a detachable implant |
| WO2009097556A2 (en) | 2008-01-30 | 2009-08-06 | Angiotech Pharmaceuticals, Inc. | Appartaus and method for forming self-retaining sutures |
| US8615856B1 (en) | 2008-01-30 | 2013-12-31 | Ethicon, Inc. | Apparatus and method for forming self-retaining sutures |
| BRPI0907787B8 (pt) | 2008-02-21 | 2021-06-22 | Angiotech Pharm Inc | método para formar uma sutura de autorretenção e aparelho para elevar os retentores em um fio de sutura a um ângulo desejado |
| US8641732B1 (en) | 2008-02-26 | 2014-02-04 | Ethicon, Inc. | Self-retaining suture with variable dimension filament and method |
| US20090227976A1 (en) * | 2008-03-05 | 2009-09-10 | Calabria Marie F | Multiple biocompatible polymeric strand aneurysm embolization system and method |
| JP5346922B2 (ja) * | 2008-03-31 | 2013-11-20 | テルモ株式会社 | 医療用閉塞具 |
| US20100082056A1 (en) * | 2008-04-04 | 2010-04-01 | Akshay Mavani | Implantable fistula closure device |
| CA2720847C (en) | 2008-04-15 | 2016-06-28 | Angiotech Pharmaceuticals, Inc. | Self-retaining sutures with bi-directional retainers or uni-directional retainers |
| WO2009132045A2 (en) | 2008-04-21 | 2009-10-29 | Nfocus Neuromedical, Inc. | Braid-ball embolic devices and delivery systems |
| US10028747B2 (en) | 2008-05-01 | 2018-07-24 | Aneuclose Llc | Coils with a series of proximally-and-distally-connected loops for occluding a cerebral aneurysm |
| US10716573B2 (en) | 2008-05-01 | 2020-07-21 | Aneuclose | Janjua aneurysm net with a resilient neck-bridging portion for occluding a cerebral aneurysm |
| JP2011519300A (ja) * | 2008-05-01 | 2011-07-07 | アニュクローズ エルエルシー | 動脈瘤閉塞装置 |
| CA2722672C (en) | 2008-05-02 | 2019-10-22 | Sequent Medical Inc. | Filamentary devices for treatment of vascular defects |
| WO2009140437A1 (en) | 2008-05-13 | 2009-11-19 | Nfocus Neuromedical, Inc. | Braid implant delivery systems |
| WO2010022180A1 (en) | 2008-08-19 | 2010-02-25 | Micro Therapeutics, Inc. | Detachable tip microcatheter |
| WO2010028300A1 (en) * | 2008-09-04 | 2010-03-11 | Curaseal Inc. | Inflatable device for enteric fistula treatment |
| US9554826B2 (en) | 2008-10-03 | 2017-01-31 | Femasys, Inc. | Contrast agent injection system for sonographic imaging |
| US10070888B2 (en) | 2008-10-03 | 2018-09-11 | Femasys, Inc. | Methods and devices for sonographic imaging |
| USD611146S1 (en) | 2008-10-23 | 2010-03-02 | Vertos Medical, Inc. | Tissue modification device |
| USD619252S1 (en) | 2008-10-23 | 2010-07-06 | Vertos Medical, Inc. | Tissue modification device |
| USD635671S1 (en) | 2008-10-23 | 2011-04-05 | Vertos Medical, Inc. | Tissue modification device |
| USD621939S1 (en) | 2008-10-23 | 2010-08-17 | Vertos Medical, Inc. | Tissue modification device |
| USD619253S1 (en) | 2008-10-23 | 2010-07-06 | Vertos Medical, Inc. | Tissue modification device |
| USD610259S1 (en) | 2008-10-23 | 2010-02-16 | Vertos Medical, Inc. | Tissue modification device |
| US8932328B2 (en) | 2008-11-03 | 2015-01-13 | Ethicon, Inc. | Length of self-retaining suture and method and device for using the same |
| US20100131002A1 (en) * | 2008-11-24 | 2010-05-27 | Connor Robert A | Stent with a net layer to embolize and aneurysm |
| US8690911B2 (en) | 2009-01-08 | 2014-04-08 | Coherex Medical, Inc. | Medical device for modification of left atrial appendage and related systems and methods |
| US9566419B2 (en) * | 2009-04-08 | 2017-02-14 | Bayer Healthcare Llc | Body lumen occlusion device and method |
| AU2010236349B2 (en) * | 2009-04-15 | 2015-04-02 | Microvention, Inc. | Implant delivery system |
| EP2419166B1 (en) * | 2009-04-15 | 2017-11-22 | MicroVention, Inc. | Implant delivery system |
| EP2442728B1 (en) | 2009-06-17 | 2018-07-25 | Coherex Medical, Inc. | Medical device for modification of left atrial appendage |
| US10631969B2 (en) | 2009-06-17 | 2020-04-28 | Coherex Medical, Inc. | Medical device for modification of left atrial appendage and related systems and methods |
| US9693781B2 (en) | 2009-06-17 | 2017-07-04 | Coherex Medical, Inc. | Medical device for modification of left atrial appendage and related systems and methods |
| US10064628B2 (en) | 2009-06-17 | 2018-09-04 | Coherex Medical, Inc. | Medical device for modification of left atrial appendage and related systems and methods |
| US9351716B2 (en) | 2009-06-17 | 2016-05-31 | Coherex Medical, Inc. | Medical device and delivery system for modification of left atrial appendage and methods thereof |
| US9649115B2 (en) | 2009-06-17 | 2017-05-16 | Coherex Medical, Inc. | Medical device for modification of left atrial appendage and related systems and methods |
| EP2480166B1 (en) | 2009-09-24 | 2017-11-29 | Microvention, Inc. | Injectable hydrogel filaments for biomedical uses |
| US8434489B2 (en) | 2009-10-23 | 2013-05-07 | Conceptus, Inc. | Contraceptive devices and methods |
| EP2493367B1 (en) | 2009-10-26 | 2019-03-13 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| WO2011057002A2 (en) * | 2009-11-05 | 2011-05-12 | Sequent Medical Inc. | Multiple layer filamentary devices or treatment of vascular defects |
| WO2011057131A1 (en) | 2009-11-09 | 2011-05-12 | Spotlight Technology Partners Llc | Polysaccharide based hydrogels |
| NZ599527A (en) | 2009-11-09 | 2014-04-30 | Spotlight Technology Partners Llc | Fragmented hydrogels |
| US9358140B1 (en) | 2009-11-18 | 2016-06-07 | Aneuclose Llc | Stent with outer member to embolize an aneurysm |
| WO2011090628A2 (en) | 2009-12-29 | 2011-07-28 | Angiotech Pharmaceuticals, Inc. | Bidirectional self-retaining sutures with laser-marked and/or non-laser marked indicia and methods |
| US8906057B2 (en) * | 2010-01-04 | 2014-12-09 | Aneuclose Llc | Aneurysm embolization by rotational accumulation of mass |
| US8231619B2 (en) | 2010-01-22 | 2012-07-31 | Cytyc Corporation | Sterilization device and method |
| US20130042873A1 (en) * | 2010-03-09 | 2013-02-21 | Klaus Bechgaard | Contraceptive device |
| CN103037776B (zh) | 2010-04-14 | 2017-07-04 | 微排放器公司 | 植入物输送装置 |
| KR20170121318A (ko) | 2010-05-04 | 2017-11-01 | 에티컨, 엘엘씨 | 레이저 커팅된 리테이너를 갖는 자가-유지형 시스템 |
| US8550086B2 (en) | 2010-05-04 | 2013-10-08 | Hologic, Inc. | Radiopaque implant |
| WO2011156733A2 (en) | 2010-06-11 | 2011-12-15 | Angiotech Pharmaceuticals, Inc. | Suture delivery tools for endoscopic and robot-assisted surgery and methods |
| US9220506B2 (en) | 2010-06-16 | 2015-12-29 | DePuy Synthes Products, Inc. | Occlusive device with stretch resistant member and anchor filament |
| US9247942B2 (en) | 2010-06-29 | 2016-02-02 | Artventive Medical Group, Inc. | Reversible tubal contraceptive device |
| WO2012002944A1 (en) | 2010-06-29 | 2012-01-05 | Artventive Medical Group, Inc. | Reducing flow through a tubular structure |
| US8425548B2 (en) | 2010-07-01 | 2013-04-23 | Aneaclose LLC | Occluding member expansion and then stent expansion for aneurysm treatment |
| US9149277B2 (en) | 2010-10-18 | 2015-10-06 | Artventive Medical Group, Inc. | Expandable device delivery |
| NZ706725A (en) | 2010-11-03 | 2016-03-31 | Tissuegen Inc | Drug-eluting self-retaining sutures and methods relating thereto |
| KR101886614B1 (ko) | 2010-11-09 | 2018-08-09 | 에티컨, 엘엘씨 | 응급 자가-유지형 봉합재 및 패키징 |
| US8360765B2 (en) | 2011-01-07 | 2013-01-29 | Covidien Lp | Systems and method for forming a coaxial implant |
| MX347582B (es) | 2011-03-23 | 2017-05-02 | Ethicon Llc | Suturas de bucle variable de autoretención. |
| US20120253381A1 (en) | 2011-03-31 | 2012-10-04 | Codman & Shurtleff, Inc. | Occlusive device with porous structure and stretch resistant member |
| WO2012145431A2 (en) | 2011-04-18 | 2012-10-26 | Microvention, Inc. | Embolic devices |
| US20120283768A1 (en) * | 2011-05-05 | 2012-11-08 | Sequent Medical Inc. | Method and apparatus for the treatment of large and giant vascular defects |
| CA2835427A1 (en) | 2011-05-11 | 2012-11-15 | Microvention, Inc. | Device for occluding a lumen |
| US9138232B2 (en) | 2011-05-24 | 2015-09-22 | Aneuclose Llc | Aneurysm occlusion by rotational dispensation of mass |
| US20130172931A1 (en) | 2011-06-06 | 2013-07-04 | Jeffrey M. Gross | Methods and devices for soft palate tissue elevation procedures |
| EP2720625A4 (en) | 2011-06-16 | 2015-02-18 | Curaseal Inc | DEVICES FOR THE TREATMENT OF FISTLES AND CORRESPONDING METHODS |
| EP2720623A4 (en) | 2011-06-17 | 2015-04-22 | Curaseal Inc | METHOD AND DEVICES FOR TREATING FISTLES |
| US9579104B2 (en) | 2011-11-30 | 2017-02-28 | Covidien Lp | Positioning and detaching implants |
| US8758427B2 (en) | 2011-12-02 | 2014-06-24 | Vascular Solutions, Inc. | Elongated expandable member for occluding varicose veins |
| US9011480B2 (en) | 2012-01-20 | 2015-04-21 | Covidien Lp | Aneurysm treatment coils |
| WO2013119332A2 (en) | 2012-02-09 | 2013-08-15 | Stout Medical Group, L.P. | Embolic device and methods of use |
| US9687245B2 (en) | 2012-03-23 | 2017-06-27 | Covidien Lp | Occlusive devices and methods of use |
| US9011884B2 (en) | 2012-04-18 | 2015-04-21 | Microvention, Inc. | Embolic devices |
| EP2674114A1 (en) | 2012-06-11 | 2013-12-18 | Acandis GmbH & Co. KG | Implant for occlusion of vascular anomalies and method for producing such an implant |
| US10124087B2 (en) | 2012-06-19 | 2018-11-13 | Covidien Lp | Detachable coupling for catheter |
| US9326774B2 (en) | 2012-08-03 | 2016-05-03 | Covidien Lp | Device for implantation of medical devices |
| CN104918565B (zh) | 2012-11-13 | 2018-04-27 | 柯惠有限合伙公司 | 封堵装置 |
| US10342546B2 (en) | 2013-01-14 | 2019-07-09 | Microvention, Inc. | Occlusive device |
| US8984733B2 (en) | 2013-02-05 | 2015-03-24 | Artventive Medical Group, Inc. | Bodily lumen occlusion |
| US9095344B2 (en) | 2013-02-05 | 2015-08-04 | Artventive Medical Group, Inc. | Methods and apparatuses for blood vessel occlusion |
| US9662119B2 (en) * | 2013-03-13 | 2017-05-30 | Lawrence Livermore National Security, Llc | Shape-memory polymer foam device for treating aneurysms |
| US9877727B2 (en) | 2013-05-02 | 2018-01-30 | Empire Technology Development Llc | Diverticulum treating device |
| US9636116B2 (en) | 2013-06-14 | 2017-05-02 | Artventive Medical Group, Inc. | Implantable luminal devices |
| US10149968B2 (en) | 2013-06-14 | 2018-12-11 | Artventive Medical Group, Inc. | Catheter-assisted tumor treatment |
| US9737308B2 (en) | 2013-06-14 | 2017-08-22 | Artventive Medical Group, Inc. | Catheter-assisted tumor treatment |
| US9737306B2 (en) | 2013-06-14 | 2017-08-22 | Artventive Medical Group, Inc. | Implantable luminal devices |
| US10010328B2 (en) | 2013-07-31 | 2018-07-03 | NeuVT Limited | Endovascular occlusion device with hemodynamically enhanced sealing and anchoring |
| WO2015015314A2 (en) | 2013-07-31 | 2015-02-05 | EMBA Medical Limited | Methods and devices for endovascular embolization |
| US9078658B2 (en) | 2013-08-16 | 2015-07-14 | Sequent Medical, Inc. | Filamentary devices for treatment of vascular defects |
| US9955976B2 (en) | 2013-08-16 | 2018-05-01 | Sequent Medical, Inc. | Filamentary devices for treatment of vascular defects |
| CN108186074A (zh) | 2014-02-27 | 2018-06-22 | 因库麦迪斯有限公司 | 用于治疗血管疾病的构架微弹簧圈 |
| WO2015153996A1 (en) | 2014-04-03 | 2015-10-08 | Micro Vention, Inc. | Embolic devices |
| US9629635B2 (en) | 2014-04-14 | 2017-04-25 | Sequent Medical, Inc. | Devices for therapeutic vascular procedures |
| US9713475B2 (en) | 2014-04-18 | 2017-07-25 | Covidien Lp | Embolic medical devices |
| WO2015167752A1 (en) | 2014-04-29 | 2015-11-05 | Microvention, Inc. | Polymers including active agents |
| US10092663B2 (en) | 2014-04-29 | 2018-10-09 | Terumo Corporation | Polymers |
| US10363043B2 (en) | 2014-05-01 | 2019-07-30 | Artventive Medical Group, Inc. | Treatment of incompetent vessels |
| EP3148481A4 (en) | 2014-05-28 | 2017-05-03 | Stryker European Holdings I, LLC | Vaso-occlusive devices and methods of use |
| US9060777B1 (en) | 2014-05-28 | 2015-06-23 | Tw Medical Technologies, Llc | Vaso-occlusive devices and methods of use |
| WO2016014687A1 (en) * | 2014-07-22 | 2016-01-28 | Boston Scientific Scimed, Inc. | Expandable vaso-occlusive devices having shape memory and methods of using the same |
| US9814466B2 (en) | 2014-08-08 | 2017-11-14 | Covidien Lp | Electrolytic and mechanical detachment for implant delivery systems |
| US9808256B2 (en) | 2014-08-08 | 2017-11-07 | Covidien Lp | Electrolytic detachment elements for implant delivery systems |
| CN104188733B (zh) * | 2014-08-11 | 2016-07-06 | 中国人民解放军总医院 | 用于形成血管栓塞的装置、系统及其用途 |
| CN104398283A (zh) * | 2014-09-15 | 2015-03-11 | 北京泰杰伟业科技有限公司 | 一种带有可膨胀聚合物的栓塞器械 |
| TW201625754A (zh) * | 2014-11-21 | 2016-07-16 | 艾倫塔斯有限公司 | 單一成份、儲存穩定、可硬化之聚矽氧組成物 |
| US10857012B2 (en) | 2015-01-20 | 2020-12-08 | Neurogami Medical, Inc. | Vascular implant |
| US9999413B2 (en) | 2015-01-20 | 2018-06-19 | Neurogami Medical, Inc. | Micrograft for the treatment of intracranial aneurysms and method for use |
| US10736730B2 (en) | 2015-01-20 | 2020-08-11 | Neurogami Medical, Inc. | Vascular implant |
| US11484319B2 (en) | 2015-01-20 | 2022-11-01 | Neurogami Medical, Inc. | Delivery system for micrograft for treating intracranial aneurysms |
| US10925611B2 (en) | 2015-01-20 | 2021-02-23 | Neurogami Medical, Inc. | Packaging for surgical implant |
| CN107427304A (zh) * | 2015-03-26 | 2017-12-01 | 波士顿科学国际有限公司 | 基于生物学的可膨胀封堵器 |
| US9943314B2 (en) | 2015-04-14 | 2018-04-17 | Teleflex Innovations S.À.R.L. | Magnetically-driven delivery assembly and method |
| WO2016182949A1 (en) | 2015-05-08 | 2016-11-17 | Stryker European Holdings I, Llc | Vaso-occlusive devices |
| US9717503B2 (en) | 2015-05-11 | 2017-08-01 | Covidien Lp | Electrolytic detachment for implant delivery systems |
| WO2016201250A1 (en) | 2015-06-11 | 2016-12-15 | Microvention, Inc. | Expansile device for implantation |
| US10716915B2 (en) * | 2015-11-23 | 2020-07-21 | Mivi Neuroscience, Inc. | Catheter systems for applying effective suction in remote vessels and thrombectomy procedures facilitated by catheter systems |
| US10813644B2 (en) | 2016-04-01 | 2020-10-27 | Artventive Medical Group, Inc. | Occlusive implant and delivery system |
| JP6418613B2 (ja) * | 2016-05-31 | 2018-11-07 | 国立大学法人信州大学 | 塞栓コイル |
| US10828037B2 (en) | 2016-06-27 | 2020-11-10 | Covidien Lp | Electrolytic detachment with fluid electrical connection |
| US10828039B2 (en) | 2016-06-27 | 2020-11-10 | Covidien Lp | Electrolytic detachment for implantable devices |
| US11051822B2 (en) | 2016-06-28 | 2021-07-06 | Covidien Lp | Implant detachment with thermal activation |
| US10420563B2 (en) | 2016-07-08 | 2019-09-24 | Neurogami Medical, Inc. | Delivery system insertable through body lumen |
| US10595981B2 (en) * | 2016-08-16 | 2020-03-24 | Medtronic Vascular, Inc. | Coated sutures for reducing stent graft endoleaks |
| US10842607B2 (en) | 2016-10-14 | 2020-11-24 | Microvention, Inc. | Embolic coils |
| US10576099B2 (en) * | 2016-10-21 | 2020-03-03 | Covidien Lp | Injectable scaffold for treatment of intracranial aneurysms and related technology |
| WO2018200891A1 (en) | 2017-04-27 | 2018-11-01 | Boston Scientific Scimed, Inc. | Occlusive medical device with fabric retention barb |
| IT201800002332A1 (it) * | 2018-02-02 | 2019-08-02 | Amista Pietro | Dispositivo intraneurismatico per il trattamento di aneurismi |
| WO2019195540A2 (en) * | 2018-04-04 | 2019-10-10 | Incumedx, Inc. | Embolic device with improved neck coverage |
| DE102018113580A1 (de) * | 2018-06-07 | 2019-12-12 | Christoph Karl | Verfahren und vorrichtung zur herstellung eines implantats |
| EP3840670B1 (en) | 2018-08-21 | 2023-11-15 | Boston Scientific Scimed, Inc. | Projecting member with barb for cardiovascular devices |
| EP3908209A4 (en) | 2019-03-15 | 2022-10-19 | Sequent Medical, Inc. | FIBROUS DEVICES FOR TREATMENT OF VASCULAR DEFECTS |
| US11291453B2 (en) | 2019-03-15 | 2022-04-05 | Sequent Medical, Inc. | Filamentary devices having a flexible joint for treatment of vascular defects |
| US11317921B2 (en) | 2019-03-15 | 2022-05-03 | Sequent Medical, Inc. | Filamentary devices for treatment of vascular defects |
| US11369355B2 (en) | 2019-06-17 | 2022-06-28 | Coherex Medical, Inc. | Medical device and system for occluding a tissue opening and method thereof |
| WO2021011694A1 (en) | 2019-07-17 | 2021-01-21 | Boston Scientific Scimed, Inc. | Left atrial appendage implant with continuous covering |
| WO2021041831A1 (en) | 2019-08-30 | 2021-03-04 | Boston Scientific Scimed, Inc. | Left atrial appendage implant with sealing disk |
| US11484629B2 (en) | 2019-09-11 | 2022-11-01 | Stryker Corporation | Intravascular devices with high tungsten content |
| US11612678B2 (en) | 2019-09-11 | 2023-03-28 | Stryker Corporation | Intravascular devices |
| US11685007B2 (en) | 2019-11-04 | 2023-06-27 | Covidien Lp | Devices, systems, and methods for treatment of intracranial aneurysms |
| WO2021195085A1 (en) | 2020-03-24 | 2021-09-30 | Boston Scientific Scimed, Inc. | Medical system for treating a left atrial appendage |
| US11812969B2 (en) | 2020-12-03 | 2023-11-14 | Coherex Medical, Inc. | Medical device and system for occluding a tissue opening and method thereof |
| US20240016497A1 (en) * | 2022-07-12 | 2024-01-18 | Medtronic Inc. | Implantable embolization device |
Family Cites Families (94)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CS154391B1 (ja) * | 1970-09-10 | 1974-04-30 | ||
| US4509504A (en) * | 1978-01-18 | 1985-04-09 | Medline Ab | Occlusion of body channels |
| US4301803A (en) * | 1978-10-06 | 1981-11-24 | Kuraray Co., Ltd. | Balloon catheter |
| US4346712A (en) * | 1979-04-06 | 1982-08-31 | Kuraray Company, Ltd. | Releasable balloon catheter |
| SE419597B (sv) * | 1979-05-04 | 1981-08-17 | Medline Ab | Anordning for temporer eller permanent tillslutning av kroppskanaler eller halrum hos menniskor och djur |
| SE447446B (sv) * | 1979-12-05 | 1986-11-17 | Medline Ab | Anordning for tillforande av medicinskt aktiva substanser for att fremja en abort |
| HU184722B (en) | 1980-02-18 | 1984-10-29 | Laszlo Lazar | Therapeutically suitable silicone rubber mixture and therapeuticaid |
| US4529739A (en) * | 1984-07-24 | 1985-07-16 | The Dow Chemical Company | Foamed polymeric materials |
| JPH0678460B2 (ja) | 1985-05-01 | 1994-10-05 | 株式会社バイオマテリアル・ユニバース | 多孔質透明ポリビニルアルユールゲル |
| US4795741A (en) | 1987-05-06 | 1989-01-03 | Biomatrix, Inc. | Compositions for therapeutic percutaneous embolization and the use thereof |
| US4819637A (en) | 1987-09-01 | 1989-04-11 | Interventional Therapeutics Corporation | System for artificial vessel embolization and devices for use therewith |
| US4994069A (en) | 1988-11-02 | 1991-02-19 | Target Therapeutics | Vaso-occlusion coil and method |
| US5354290A (en) * | 1989-05-31 | 1994-10-11 | Kimberly-Clark Corporation | Porous structure of an absorbent polymer |
| US5122136A (en) | 1990-03-13 | 1992-06-16 | The Regents Of The University Of California | Endovascular electrolytically detachable guidewire tip for the electroformation of thrombus in arteries, veins, aneurysms, vascular malformations and arteriovenous fistulas |
| US5133731A (en) | 1990-11-09 | 1992-07-28 | Catheter Research, Inc. | Embolus supply system and method |
| US5129180A (en) | 1990-12-07 | 1992-07-14 | Landec Labs, Inc. | Temperature sensitive seed germination control |
| US5120349A (en) | 1990-12-07 | 1992-06-09 | Landec Labs, Inc. | Microcapsule having temperature-dependent permeability profile |
| DE4104702C2 (de) * | 1991-02-15 | 1996-01-18 | Malte Neuss | Implantate für Organwege in Wendelform |
| US5226911A (en) * | 1991-10-02 | 1993-07-13 | Target Therapeutics | Vasoocclusion coil with attached fibrous element(s) |
| US5304194A (en) * | 1991-10-02 | 1994-04-19 | Target Therapeutics | Vasoocclusion coil with attached fibrous element(s) |
| JP3356447B2 (ja) * | 1991-10-16 | 2002-12-16 | テルモ株式会社 | 乾燥高分子ゲルからなる血管病変塞栓材料 |
| US5258042A (en) * | 1991-12-16 | 1993-11-02 | Henry Ford Health System | Intravascular hydrogel implant |
| ES2118219T3 (es) * | 1991-12-20 | 1998-09-16 | Allied Signal Inc | Materiales de baja densidad que tienen alta superficie especifica, y articulos formados a partir de ellos para uso en la recuperacion de metales. |
| AU4926193A (en) * | 1992-09-21 | 1994-04-12 | Vitaphore Corporation | Embolization plugs for blood vessels |
| US5350397A (en) * | 1992-11-13 | 1994-09-27 | Target Therapeutics, Inc. | Axially detachable embolic coil assembly |
| US5312415A (en) | 1992-09-22 | 1994-05-17 | Target Therapeutics, Inc. | Assembly for placement of embolic coils using frictional placement |
| US5250071A (en) * | 1992-09-22 | 1993-10-05 | Target Therapeutics, Inc. | Detachable embolic coil assembly using interlocking clasps and method of use |
| US5382259A (en) | 1992-10-26 | 1995-01-17 | Target Therapeutics, Inc. | Vasoocclusion coil with attached tubular woven or braided fibrous covering |
| US5382260A (en) * | 1992-10-30 | 1995-01-17 | Interventional Therapeutics Corp. | Embolization device and apparatus including an introducer cartridge and method for delivering the same |
| US5690666A (en) | 1992-11-18 | 1997-11-25 | Target Therapeutics, Inc. | Ultrasoft embolism coils and process for using them |
| US5320639A (en) * | 1993-03-12 | 1994-06-14 | Meadox Medicals, Inc. | Vascular plug delivery system |
| AU705305B2 (en) * | 1994-03-18 | 1999-05-20 | Cook Medical Technologies Llc | Helical embolization coil |
| DE59500954D1 (de) * | 1994-04-18 | 1997-12-11 | Heinz Dr Seufert | Rückprojektionsmodul |
| US5939622A (en) | 1994-04-26 | 1999-08-17 | Cytec Technology Corp. | Settling process analysis method |
| US5573994A (en) * | 1994-05-13 | 1996-11-12 | University Of Cincinnati | Superabsorbent foams, and method for producing the same |
| JP2535785B2 (ja) | 1994-06-03 | 1996-09-18 | 工業技術院長 | 血管塞栓剤 |
| US5549624A (en) * | 1994-06-24 | 1996-08-27 | Target Therapeutics, Inc. | Fibered vasooclusion coils |
| DE19580865D2 (de) * | 1994-08-17 | 1998-03-19 | Boston Scient Corp | Implantat, Implantationsverfahren und Applikationsvorrichtung |
| US5570585A (en) | 1994-10-03 | 1996-11-05 | Vaynberg; Mikhail | Universal cooling system automatically configured to operate in compound or single compressor mode |
| US5690671A (en) * | 1994-12-13 | 1997-11-25 | Micro Interventional Systems, Inc. | Embolic elements and methods and apparatus for their delivery |
| US5578074A (en) | 1994-12-22 | 1996-11-26 | Target Therapeutics, Inc. | Implant delivery method and assembly |
| JP3625837B2 (ja) * | 1995-01-27 | 2005-03-02 | シメッド ライフ システムズ,インコーポレイテッド | 塞栓装置 |
| US5750585A (en) * | 1995-04-04 | 1998-05-12 | Purdue Research Foundation | Super absorbent hydrogel foams |
| US5645558A (en) * | 1995-04-20 | 1997-07-08 | Medical University Of South Carolina | Anatomically shaped vasoocclusive device and method of making the same |
| US5911731A (en) * | 1995-04-20 | 1999-06-15 | Target Therapeutics, Inc. | Anatomically shaped vasoocclusive devices |
| US6143007A (en) * | 1995-04-28 | 2000-11-07 | Target Therapeutics, Inc. | Method for making an occlusive device |
| US6103084A (en) | 1995-06-06 | 2000-08-15 | Eppendorf Netheler-Hinz Gmbh | Apparatus for electroporation |
| US5624461A (en) | 1995-06-06 | 1997-04-29 | Target Therapeutics, Inc. | Three dimensional in-filling vaso-occlusive coils |
| US5766160A (en) * | 1995-06-06 | 1998-06-16 | Target Therapeutics, Inc. | Variable stiffness coils |
| US6013084A (en) * | 1995-06-30 | 2000-01-11 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils (II) |
| US5853418A (en) * | 1995-06-30 | 1998-12-29 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils (II) |
| US5582619A (en) * | 1995-06-30 | 1996-12-10 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils |
| DK0754435T3 (da) * | 1995-06-30 | 2000-11-27 | Target Therapeutics Inc | Strækningsmodstandsdygtige karokklusionsspiraler |
| US5580568A (en) | 1995-07-27 | 1996-12-03 | Micro Therapeutics, Inc. | Cellulose diacetate compositions for use in embolizing blood vessels |
| EP0842209B1 (en) | 1995-07-28 | 2006-10-11 | Genzyme Corporation | Multiblock biodegradable hydrogels for use as controlled release agents for drugs and tissue treatment agents |
| DE29518932U1 (de) | 1995-11-29 | 1996-06-20 | Reul Juergen Dr Med | Kontrolliert ablösbare Embolisations-Kugelspirale |
| AU690862B2 (en) * | 1995-12-04 | 1998-04-30 | Target Therapeutics, Inc. | Fibered micro vaso-occlusive devices |
| US5658308A (en) | 1995-12-04 | 1997-08-19 | Target Therapeutics, Inc. | Bioactive occlusion coil |
| US5752974A (en) * | 1995-12-18 | 1998-05-19 | Collagen Corporation | Injectable or implantable biomaterials for filling or blocking lumens and voids of the body |
| US5749894A (en) * | 1996-01-18 | 1998-05-12 | Target Therapeutics, Inc. | Aneurysm closure method |
| US5898411A (en) * | 1996-02-26 | 1999-04-27 | Pacific Antenna Technologies, Inc. | Single-element, multi-frequency, dipole antenna |
| US5792154A (en) * | 1996-04-10 | 1998-08-11 | Target Therapeutics, Inc. | Soft-ended fibered micro vaso-occlusive devices |
| US6190402B1 (en) | 1996-06-21 | 2001-02-20 | Musc Foundation For Research Development | Insitu formable and self-forming intravascular flow modifier (IFM) and IFM assembly for deployment of same |
| US5980514A (en) * | 1996-07-26 | 1999-11-09 | Target Therapeutics, Inc. | Aneurysm closure device assembly |
| US5695480A (en) * | 1996-07-29 | 1997-12-09 | Micro Therapeutics, Inc. | Embolizing compositions |
| US5823198A (en) * | 1996-07-31 | 1998-10-20 | Micro Therapeutics, Inc. | Method and apparatus for intravasculer embolization |
| US5690667A (en) * | 1996-09-26 | 1997-11-25 | Target Therapeutics | Vasoocclusion coil having a polymer tip |
| GB9704288D0 (en) | 1997-03-01 | 1997-04-23 | Smith & Nephew | Hydrogels |
| US5911717A (en) * | 1997-03-17 | 1999-06-15 | Precision Vascular Systems, Inc. | Catheter deliverable thrombogenic apparatus and method |
| US5980554A (en) * | 1997-05-05 | 1999-11-09 | Micro Therapeutics, Inc. | Wire frame partial flow obstruction for aneurysm treatment |
| US6271278B1 (en) * | 1997-05-13 | 2001-08-07 | Purdue Research Foundation | Hydrogel composites and superporous hydrogel composites having fast swelling, high mechanical strength, and superabsorbent properties |
| US5891192A (en) * | 1997-05-22 | 1999-04-06 | The Regents Of The University Of California | Ion-implanted protein-coated intralumenal implants |
| US5916235A (en) * | 1997-08-13 | 1999-06-29 | The Regents Of The University Of California | Apparatus and method for the use of detachable coils in vascular aneurysms and body cavities |
| US6316522B1 (en) | 1997-08-18 | 2001-11-13 | Scimed Life Systems, Inc. | Bioresorbable hydrogel compositions for implantable prostheses |
| US6245090B1 (en) * | 1997-11-07 | 2001-06-12 | Salviac Limited | Transcatheter occluding implant |
| US6168570B1 (en) * | 1997-12-05 | 2001-01-02 | Micrus Corporation | Micro-strand cable with enhanced radiopacity |
| US5935145A (en) * | 1998-02-13 | 1999-08-10 | Target Therapeutics, Inc. | Vaso-occlusive device with attached polymeric materials |
| US6015424A (en) * | 1998-04-28 | 2000-01-18 | Microvention, Inc. | Apparatus and method for vascular embolization |
| US6113629A (en) * | 1998-05-01 | 2000-09-05 | Micrus Corporation | Hydrogel for the therapeutic treatment of aneurysms |
| US6463317B1 (en) | 1998-05-19 | 2002-10-08 | Regents Of The University Of Minnesota | Device and method for the endovascular treatment of aneurysms |
| US5980550A (en) | 1998-06-18 | 1999-11-09 | Target Therapeutics, Inc. | Water-soluble coating for bioactive vasoocclusive devices |
| US5935148A (en) * | 1998-06-24 | 1999-08-10 | Target Therapeutics, Inc. | Detachable, varying flexibility, aneurysm neck bridge |
| US6165193A (en) * | 1998-07-06 | 2000-12-26 | Microvention, Inc. | Vascular embolization with an expansible implant |
| US6093199A (en) * | 1998-08-05 | 2000-07-25 | Endovascular Technologies, Inc. | Intra-luminal device for treatment of body cavities and lumens and method of use |
| US6605294B2 (en) * | 1998-08-14 | 2003-08-12 | Incept Llc | Methods of using in situ hydration of hydrogel articles for sealing or augmentation of tissue or vessels |
| US6187024B1 (en) * | 1998-11-10 | 2001-02-13 | Target Therapeutics, Inc. | Bioactive coating for vaso-occlusive devices |
| US6280457B1 (en) | 1999-06-04 | 2001-08-28 | Scimed Life Systems, Inc. | Polymer covered vaso-occlusive devices and methods of producing such devices |
| US6312421B1 (en) * | 1999-07-23 | 2001-11-06 | Neurovasx, Inc. | Aneurysm embolization material and device |
| US6238403B1 (en) * | 1999-10-04 | 2001-05-29 | Microvention, Inc. | Filamentous embolic device with expansible elements |
| JP2003511188A (ja) * | 1999-10-04 | 2003-03-25 | マイクロ ベンション インコーポレイテッド | 拡開要素を備えたフィラメント状の塞栓装置 |
| EP1104681A1 (en) | 1999-12-03 | 2001-06-06 | Biomat B.V. | Wire, tube or catheter with hydrophilic coating |
| US6530934B1 (en) * | 2000-06-06 | 2003-03-11 | Sarcos Lc | Embolic device composed of a linear sequence of miniature beads |
| AU2001280618A1 (en) | 2000-07-18 | 2002-01-30 | George P. Teitelbaum | Biocompatible, expansile material and stent |
| US6723108B1 (en) | 2000-09-18 | 2004-04-20 | Cordis Neurovascular, Inc | Foam matrix embolization device |
-
2002
- 2002-05-29 CA CA002449055A patent/CA2449055C/en not_active Expired - Fee Related
- 2002-05-29 AU AU2002344223A patent/AU2002344223B2/en not_active Ceased
- 2002-05-29 US US10/157,621 patent/US7014645B2/en not_active Expired - Lifetime
- 2002-05-29 AT AT02752008T patent/ATE516759T1/de not_active IP Right Cessation
- 2002-05-29 CA CA2689598A patent/CA2689598A1/en not_active Abandoned
- 2002-05-29 ES ES10183747.4T patent/ES2441341T3/es not_active Expired - Lifetime
- 2002-05-29 JP JP2002592820A patent/JP4383746B2/ja not_active Expired - Fee Related
- 2002-05-29 EP EP10183747.4A patent/EP2319430B1/en not_active Expired - Lifetime
- 2002-05-29 WO PCT/US2002/016873 patent/WO2002096302A1/en active Application Filing
- 2002-05-29 EP EP02752008A patent/EP1401338B1/en not_active Expired - Lifetime
-
2003
- 2003-09-24 US US10/670,142 patent/US7491214B2/en not_active Expired - Fee Related
-
2006
- 2006-02-08 US US11/350,357 patent/US7842054B2/en not_active Expired - Lifetime
- 2006-10-06 AU AU2006225285A patent/AU2006225285B2/en not_active Ceased
-
2009
- 2009-01-29 US US12/362,466 patent/US8603128B2/en not_active Expired - Lifetime
-
2010
- 2010-01-15 AU AU2010200166A patent/AU2010200166A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| JP2004527342A (ja) | 2004-09-09 |
| US7491214B2 (en) | 2009-02-17 |
| EP1401338B1 (en) | 2011-07-20 |
| US20020177855A1 (en) | 2002-11-28 |
| CA2689598A1 (en) | 2002-12-05 |
| CA2449055C (en) | 2010-03-02 |
| EP1401338A1 (en) | 2004-03-31 |
| AU2010200166A1 (en) | 2010-02-04 |
| ATE516759T1 (de) | 2011-08-15 |
| US7014645B2 (en) | 2006-03-21 |
| US20060149299A1 (en) | 2006-07-06 |
| AU2002344223C1 (en) | 2002-12-09 |
| AU2006225285B2 (en) | 2009-10-29 |
| AU2002344223B2 (en) | 2006-07-06 |
| CA2449055A1 (en) | 2002-12-05 |
| US20040059370A1 (en) | 2004-03-25 |
| AU2006225285A1 (en) | 2006-10-26 |
| EP2319430A2 (en) | 2011-05-11 |
| US20090232869A1 (en) | 2009-09-17 |
| ES2441341T3 (es) | 2014-02-04 |
| US8603128B2 (en) | 2013-12-10 |
| EP2319430A3 (en) | 2011-09-28 |
| WO2002096302A1 (en) | 2002-12-05 |
| US7842054B2 (en) | 2010-11-30 |
| EP2319430B1 (en) | 2013-11-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4383746B2 (ja) | 膨張性の糸状塞栓装置の製造方法 | |
| US6238403B1 (en) | Filamentous embolic device with expansible elements | |
| US6602261B2 (en) | Filamentous embolic device with expansile elements | |
| US20190046210A1 (en) | Embolic Device With Shaped Wire | |
| CA2777171C (en) | Embolization device constructed from expansile polymer | |
| AU2002344223A1 (en) | Method of Manufacturing Expansile Filamentous Embolization Devices | |
| CA2385615C (en) | Filamentous embolic device with expansible elements |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20050527 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20050527 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20080708 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20081008 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20081008 |
|
| RD13 | Notification of appointment of power of sub attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7433 Effective date: 20081008 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20081008 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20081205 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090108 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20090217 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20090518 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090616 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20090804 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20090825 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20090924 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20121002 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20131002 Year of fee payment: 4 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |