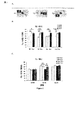
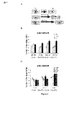
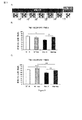
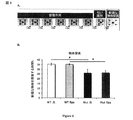
JP2013533287A - 嗅覚機能不全の処置のためのラサギリンの使用 - Google Patents
嗅覚機能不全の処置のためのラサギリンの使用 Download PDFInfo
- Publication number
- JP2013533287A JP2013533287A JP2013521949A JP2013521949A JP2013533287A JP 2013533287 A JP2013533287 A JP 2013533287A JP 2013521949 A JP2013521949 A JP 2013521949A JP 2013521949 A JP2013521949 A JP 2013521949A JP 2013533287 A JP2013533287 A JP 2013533287A
- Authority
- JP
- Japan
- Prior art keywords
- olfactory
- subject
- pharmaceutically acceptable
- acceptable salt
- propargyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/02—Nasal agents, e.g. decongestants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
Landscapes
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biomedical Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Epidemiology (AREA)
- Otolaryngology (AREA)
- Psychology (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- Pulmonology (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US40046410P | 2010-07-27 | 2010-07-27 | |
| US61/400,464 | 2010-07-27 | ||
| US201161437212P | 2011-01-28 | 2011-01-28 | |
| US61/437,212 | 2011-01-28 | ||
| PCT/US2011/045574 WO2012015950A1 (en) | 2010-07-27 | 2011-07-27 | Use of rasagiline for the treatment of olfactory dysfunction |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2013533287A true JP2013533287A (ja) | 2013-08-22 |
| JP2013533287A5 JP2013533287A5 (OSRAM) | 2014-10-16 |
Family
ID=45527351
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013521949A Ceased JP2013533287A (ja) | 2010-07-27 | 2011-07-27 | 嗅覚機能不全の処置のためのラサギリンの使用 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8569379B2 (OSRAM) |
| EP (1) | EP2603212A4 (OSRAM) |
| JP (1) | JP2013533287A (OSRAM) |
| AU (1) | AU2011282720B2 (OSRAM) |
| CA (1) | CA2806740A1 (OSRAM) |
| RU (1) | RU2587330C2 (OSRAM) |
| WO (1) | WO2012015950A1 (OSRAM) |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5701485B2 (ja) * | 2006-02-21 | 2015-04-15 | テバ ファーマシューティカル インダストリーズ リミティド | 多系統萎縮症の治療のためのラサギリンの使用 |
| MX2008012781A (es) | 2006-04-03 | 2008-10-14 | Teva Pharma | Uso de rasagilina para el tratamiento de sindrome de piernas inquietas. |
| PT2101569E (pt) * | 2006-12-14 | 2012-01-13 | Teva Pharma | Base de rasagilina sólida cristalina |
| EP1987816A1 (de) * | 2007-04-30 | 2008-11-05 | Ratiopharm GmbH | Adsorbate eines Rasagilinsalzes mit einem wasserlöslichen Hilfsstoff |
| EP2234478A4 (en) * | 2008-01-11 | 2013-01-23 | Teva Pharma | RASAGILINE FORMULATIONS, THEIR PREPARATION AND USE |
| US20100008983A1 (en) * | 2008-06-10 | 2010-01-14 | Muhammad Safadi | Rasagiline soft gelatin capsules |
| DK2451771T3 (da) | 2009-07-09 | 2014-09-08 | Ratiopharm Gmbh | Salte af rasagilin og farmaceutiske fremstillingsmåder heraf |
| EP2485722A1 (en) * | 2009-10-09 | 2012-08-15 | Teva Pharmaceutical Industries, Ltd. | Use of rasagiline for the treatment of progressive supranuclear palsy |
| EP2515891A4 (en) * | 2009-12-22 | 2013-06-05 | Teva Pharma | 3-keto-N-propargyl-1-aminoindan |
| CA2806737A1 (en) | 2010-07-27 | 2012-02-02 | Teva Pharmaceutical Industries Ltd. | Dispersions of rasagiline citrate |
| AU2012323349A1 (en) | 2011-10-10 | 2014-05-22 | Teva Pharmaceutical Industries Ltd. | R(+)-N-formyl-propargyl-aminoindan |
| BR112014008552A2 (pt) | 2011-10-10 | 2017-04-18 | Teva Pharma | r(+)-n-metil-propargilaminoindano |
| CA2882072A1 (en) | 2012-08-17 | 2014-02-20 | Teva Pharmaceutical Industries Ltd. | Parenteral formulations of rasagiline |
| RU2713786C1 (ru) * | 2019-05-24 | 2020-02-07 | Сергей Геннадиевич Вахрушев | Способ лечения пациентов с обонятельной дисфункцией при атрофическом рините |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009151625A1 (en) * | 2008-06-13 | 2009-12-17 | Teva Pharmaceutical Industries, Ltd. | Rasagiline for parkinson's disease modification |
Family Cites Families (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL92952A (en) | 1990-01-03 | 1994-06-24 | Teva Pharma | R-enantiomers of n-propargyl-1-aminoindan compounds, their preparation and pharmaceutical compositions containing them |
| US5744500A (en) | 1990-01-03 | 1998-04-28 | Teva Pharmaceutical Industries, Ltd. | Use of R-enantiomer of N-propargyl-1-aminoindan, salts, and compositions thereof |
| IL112819A (en) | 1991-10-16 | 2001-11-25 | Teva Pharma | Fluorinated 1-aminoindan compounds and a process for their preparation |
| IL115357A (en) | 1995-09-20 | 2000-01-31 | Teva Pharma | Stable compositions containing N-propargyl-1-aminoindan and polyhydric alcohols |
| IL118836A (en) | 1996-07-11 | 2001-01-11 | Teva Pharma | Pharmaceutical compositions comprising s-(-)-n-propargyl-1-aminoindan |
| DE69738275T2 (de) | 1996-12-18 | 2008-08-28 | Teva Pharmaceutical Industries Ltd. | Aminoindanderivate |
| US7326690B2 (en) | 2002-10-30 | 2008-02-05 | Bach Pharma, Inc. | Modulation of cell fates and activities by phthalazinediones |
| ES2588780T3 (es) | 2002-11-15 | 2016-11-04 | Teva Pharmaceutical Industries Limited | Uso de rasagilina con o sin riluzol para el tratamiento de la esclerosis lateral amiotrófica |
| CN101693695A (zh) * | 2004-03-30 | 2010-04-14 | 大正制药株式会社 | 嘧啶衍生物以及与其应用有关的治疗方法 |
| WO2006014973A2 (en) | 2004-07-26 | 2006-02-09 | Teva Pharmaceutical Industries, Ltd. | Pharmaceutical dosage forms including rasagiline |
| CN101098685A (zh) | 2004-11-24 | 2008-01-02 | 特瓦制药工业有限公司 | 雷沙吉兰经口崩解组合物 |
| CA2901244A1 (en) | 2005-02-23 | 2006-08-31 | Teva Pharmaceutical Industries, Ltd. | Rasagiline formulations of improved content uniformity |
| US7491847B2 (en) | 2005-11-17 | 2009-02-17 | Teva Pharmaceutical Industries, Ltd. | Methods for isolating propargylated aminoindans |
| US7572834B1 (en) | 2005-12-06 | 2009-08-11 | Teva Pharmaceutical Industries, Ltd. | Rasagiline formulations and processes for their preparation |
| JP5701485B2 (ja) | 2006-02-21 | 2015-04-15 | テバ ファーマシューティカル インダストリーズ リミティド | 多系統萎縮症の治療のためのラサギリンの使用 |
| MX2008012781A (es) | 2006-04-03 | 2008-10-14 | Teva Pharma | Uso de rasagilina para el tratamiento de sindrome de piernas inquietas. |
| EP1892233A1 (de) | 2006-08-18 | 2008-02-27 | Ratiopharm GmbH | Neue Salze des Wirkstoffs Rasagilin |
| NZ577623A (en) | 2006-12-14 | 2011-05-27 | Teva Pharma | Tannate salt of rasagiline |
| PT2101569E (pt) | 2006-12-14 | 2012-01-13 | Teva Pharma | Base de rasagilina sólida cristalina |
| EP1987816A1 (de) | 2007-04-30 | 2008-11-05 | Ratiopharm GmbH | Adsorbate eines Rasagilinsalzes mit einem wasserlöslichen Hilfsstoff |
| GB2463833B (en) | 2007-06-26 | 2012-02-08 | Parkinson S Inst | Compositions that prevent or reverse alpha-synuclein fibrillation for use in the treatment of neurological disorders |
| US20090062400A1 (en) | 2007-09-05 | 2009-03-05 | Laurence Oron | Method of treating glaucoma using rasagiline |
| US8188149B2 (en) | 2007-09-17 | 2012-05-29 | Teva Pharmaceutical Industries, Ltd. | Use of R(+)-N-propargy1-1-aminoindan to treat or prevent hearing loss |
| EP2234478A4 (en) | 2008-01-11 | 2013-01-23 | Teva Pharma | RASAGILINE FORMULATIONS, THEIR PREPARATION AND USE |
| US20100008983A1 (en) | 2008-06-10 | 2010-01-14 | Muhammad Safadi | Rasagiline soft gelatin capsules |
| NZ589547A (en) | 2008-06-19 | 2013-03-28 | Teva Pharma | Dried rasagiline base having a water content less than 0.5 % by weight |
| JP2011525489A (ja) | 2008-06-19 | 2011-09-22 | テバ ファーマシューティカル インダストリーズ リミティド | ラサリジン塩基の精製方法 |
| DE102008064061A1 (de) | 2008-12-19 | 2010-06-24 | Ratiopharm Gmbh | Feste Zusammensetzung mit dem Wirkstoff Rasagilin |
| US20100189791A1 (en) | 2009-01-23 | 2010-07-29 | Teva Pharmaceutical Industries, Ltd. | Delayed release rasagiline malate formulation |
| DK2451771T3 (da) | 2009-07-09 | 2014-09-08 | Ratiopharm Gmbh | Salte af rasagilin og farmaceutiske fremstillingsmåder heraf |
| EP2485722A1 (en) | 2009-10-09 | 2012-08-15 | Teva Pharmaceutical Industries, Ltd. | Use of rasagiline for the treatment of progressive supranuclear palsy |
| EP2515891A4 (en) | 2009-12-22 | 2013-06-05 | Teva Pharma | 3-keto-N-propargyl-1-aminoindan |
| CA2806737A1 (en) | 2010-07-27 | 2012-02-02 | Teva Pharmaceutical Industries Ltd. | Dispersions of rasagiline citrate |
| MX2013004598A (es) | 2010-10-26 | 2013-07-17 | Teva Pharma | Rasagilina enriquesida con deuterio. |
-
2011
- 2011-07-27 EP EP11813130.9A patent/EP2603212A4/en not_active Withdrawn
- 2011-07-27 AU AU2011282720A patent/AU2011282720B2/en not_active Ceased
- 2011-07-27 US US13/192,019 patent/US8569379B2/en not_active Expired - Fee Related
- 2011-07-27 CA CA2806740A patent/CA2806740A1/en not_active Abandoned
- 2011-07-27 WO PCT/US2011/045574 patent/WO2012015950A1/en not_active Ceased
- 2011-07-27 RU RU2013108258/15A patent/RU2587330C2/ru not_active IP Right Cessation
- 2011-07-27 JP JP2013521949A patent/JP2013533287A/ja not_active Ceased
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009151625A1 (en) * | 2008-06-13 | 2009-12-17 | Teva Pharmaceutical Industries, Ltd. | Rasagiline for parkinson's disease modification |
Non-Patent Citations (2)
| Title |
|---|
| JPN6015024065; Alvarez, M. V. et al., Movement Disorders, 2010, Vol.25, Supp. 2, p. S255, Abstract Number:211 * |
| JPN6015024066; Simuni, T. et al., European Neurology, 2009, Vol.61, No.4, p.206-215 * |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2011282720B2 (en) | 2016-12-22 |
| WO2012015950A1 (en) | 2012-02-02 |
| EP2603212A1 (en) | 2013-06-19 |
| RU2013108258A (ru) | 2014-09-10 |
| AU2011282720A1 (en) | 2013-03-14 |
| CA2806740A1 (en) | 2012-02-02 |
| EP2603212A4 (en) | 2014-01-08 |
| RU2587330C2 (ru) | 2016-06-20 |
| US20120029087A1 (en) | 2012-02-02 |
| US8569379B2 (en) | 2013-10-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2013533287A (ja) | 嗅覚機能不全の処置のためのラサギリンの使用 | |
| CN100450475C (zh) | 用于治疗注意力缺陷多动障碍及多发性硬化症疲劳的含有莫达非尼的组合物 | |
| JP5701485B2 (ja) | 多系統萎縮症の治療のためのラサギリンの使用 | |
| CN101442997B (zh) | 雷沙吉兰用于治疗多动腿综合征 | |
| US9980927B2 (en) | Use of tapentadol for inhibiting and/or treating depression and anxiety | |
| Nicolodi et al. | Fibromyalgia and migraine, two faces of the same mechanism: serotonin as the common clue for pathogenesis and therapy | |
| US20230310368A1 (en) | Mdma enantiomers | |
| US20250090637A1 (en) | Methods of inhibiting cerebral inflammation | |
| RU2464023C2 (ru) | Лекарственное средство для лечения фибромиалгии | |
| WO2010132128A1 (en) | Method of improving cognitve functions in individuals with down syndrome and/or alzheimer's disease | |
| US20230226020A1 (en) | Mdma enantiomers | |
| US20230226019A1 (en) | Mdma enantiomers | |
| JP7744419B2 (ja) | レット症候群を治療するためのプリドピジンまたはその類似体の使用 | |
| CN117396200A (zh) | 辅助性d-环丝氨酸对重度抑郁症的经颅磁刺激(tms)治疗的增效作用 | |
| US20120309840A1 (en) | Treatment of Pain Associated with Trigeminal Neuralgia | |
| CN115337307A (zh) | 布托啡诺或其盐在制备抗抑郁药物中的新用途 | |
| WO2024196772A2 (en) | Mdma enantiomers | |
| CA2575204A1 (en) | Methods of treating ophthalmic conditions | |
| WO2024253775A1 (en) | Mdma enantiomers | |
| US20190274974A1 (en) | Treatment of Pain Associated with Trigeminal Neuralgia | |
| Woodlee | Mechanisms underlying the dysregulation of postural stability in dopamine-depleted rats | |
| WO2015167567A1 (en) | Stat3 inhibitors for treatment of hyperglycemia and impaired spatial vision |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20140728 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140901 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20150623 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20150910 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20151224 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20160531 |
|
| A045 | Written measure of dismissal of application [lapsed due to lack of payment] |
Free format text: JAPANESE INTERMEDIATE CODE: A045 Effective date: 20180227 |