JP2010006780A - フィルム状製剤及びフィルム状製剤の製造方法 - Google Patents
フィルム状製剤及びフィルム状製剤の製造方法 Download PDFInfo
- Publication number
- JP2010006780A JP2010006780A JP2008170683A JP2008170683A JP2010006780A JP 2010006780 A JP2010006780 A JP 2010006780A JP 2008170683 A JP2008170683 A JP 2008170683A JP 2008170683 A JP2008170683 A JP 2008170683A JP 2010006780 A JP2010006780 A JP 2010006780A
- Authority
- JP
- Japan
- Prior art keywords
- film
- preparation
- suspension
- oxide
- vapor deposition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 99
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 16
- 239000003814 drug Substances 0.000 claims abstract description 52
- 229940079593 drug Drugs 0.000 claims abstract description 51
- 239000000843 powder Substances 0.000 claims abstract description 44
- 238000007740 vapor deposition Methods 0.000 claims abstract description 27
- 239000000725 suspension Substances 0.000 claims abstract description 22
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 16
- 229940117841 methacrylic acid copolymer Drugs 0.000 claims abstract description 15
- 229920003145 methacrylic acid copolymer Polymers 0.000 claims abstract description 15
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims abstract description 14
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 claims abstract description 9
- 229920002678 cellulose Polymers 0.000 claims abstract description 9
- 239000001913 cellulose Substances 0.000 claims abstract description 9
- 229910052709 silver Inorganic materials 0.000 claims abstract description 9
- 239000004332 silver Substances 0.000 claims abstract description 9
- 238000005266 casting Methods 0.000 claims abstract description 8
- 238000001035 drying Methods 0.000 claims abstract description 8
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims abstract description 7
- 229920002472 Starch Polymers 0.000 claims abstract description 7
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 claims abstract description 7
- 229910052737 gold Inorganic materials 0.000 claims abstract description 7
- 239000010931 gold Substances 0.000 claims abstract description 7
- 229910052710 silicon Inorganic materials 0.000 claims abstract description 7
- 239000010703 silicon Substances 0.000 claims abstract description 7
- 235000019698 starch Nutrition 0.000 claims abstract description 7
- 239000008107 starch Substances 0.000 claims abstract description 7
- 229920001817 Agar Polymers 0.000 claims abstract description 6
- 102000008186 Collagen Human genes 0.000 claims abstract description 6
- 108010035532 Collagen Proteins 0.000 claims abstract description 6
- 229920001800 Shellac Polymers 0.000 claims abstract description 6
- 229920002494 Zein Polymers 0.000 claims abstract description 6
- 239000008272 agar Substances 0.000 claims abstract description 6
- 239000007864 aqueous solution Substances 0.000 claims abstract description 6
- 229920001436 collagen Polymers 0.000 claims abstract description 6
- 229960005188 collagen Drugs 0.000 claims abstract description 6
- 229910052697 platinum Inorganic materials 0.000 claims abstract description 6
- 239000004208 shellac Substances 0.000 claims abstract description 6
- 229940113147 shellac Drugs 0.000 claims abstract description 6
- ZLGIYFNHBLSMPS-ATJNOEHPSA-N shellac Chemical compound OCCCCCC(O)C(O)CCCCCCCC(O)=O.C1C23[C@H](C(O)=O)CCC2[C@](C)(CO)[C@@H]1C(C(O)=O)=C[C@@H]3O ZLGIYFNHBLSMPS-ATJNOEHPSA-N 0.000 claims abstract description 6
- 235000013874 shellac Nutrition 0.000 claims abstract description 6
- 239000005019 zein Substances 0.000 claims abstract description 6
- 229940093612 zein Drugs 0.000 claims abstract description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims abstract description 5
- 229910052814 silicon oxide Inorganic materials 0.000 claims abstract description 5
- 238000009472 formulation Methods 0.000 claims description 19
- 239000011159 matrix material Substances 0.000 claims description 19
- 239000000203 mixture Substances 0.000 claims description 19
- 239000000758 substrate Substances 0.000 claims description 17
- -1 acetal diethylaminoacetate Chemical class 0.000 claims description 9
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Natural products CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 claims description 7
- 229920000639 hydroxypropylmethylcellulose acetate succinate Polymers 0.000 claims description 7
- 229920002554 vinyl polymer Polymers 0.000 claims description 7
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 6
- 229920003144 amino alkyl methacrylate copolymer Polymers 0.000 claims description 6
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 claims description 6
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 6
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims description 5
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims description 5
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 5
- 229910052782 aluminium Inorganic materials 0.000 claims description 5
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 5
- 229910052802 copper Inorganic materials 0.000 claims description 5
- 239000010949 copper Substances 0.000 claims description 5
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 claims description 5
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 claims description 5
- 229910052749 magnesium Inorganic materials 0.000 claims description 5
- 239000011777 magnesium Substances 0.000 claims description 5
- 239000000395 magnesium oxide Substances 0.000 claims description 5
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 claims description 5
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 claims description 5
- 229910052719 titanium Inorganic materials 0.000 claims description 5
- 239000010936 titanium Substances 0.000 claims description 5
- 235000019640 taste Nutrition 0.000 abstract description 12
- 230000007794 irritation Effects 0.000 abstract description 9
- 229940023476 agar Drugs 0.000 abstract description 4
- 238000000151 deposition Methods 0.000 abstract description 4
- 229910052751 metal Inorganic materials 0.000 abstract description 4
- 239000002184 metal Substances 0.000 abstract description 4
- 150000002894 organic compounds Chemical class 0.000 abstract description 4
- 230000008447 perception Effects 0.000 abstract description 4
- 230000002496 gastric effect Effects 0.000 abstract description 3
- 229910044991 metal oxide Inorganic materials 0.000 abstract description 2
- 150000004706 metal oxides Chemical class 0.000 abstract description 2
- 239000000243 solution Substances 0.000 abstract description 2
- CRVGKGJPQYZRPT-UHFFFAOYSA-N diethylamino acetate Chemical compound CCN(CC)OC(C)=O CRVGKGJPQYZRPT-UHFFFAOYSA-N 0.000 abstract 1
- 229920000642 polymer Polymers 0.000 abstract 1
- 239000010408 film Substances 0.000 description 80
- 239000010410 layer Substances 0.000 description 33
- 210000000214 mouth Anatomy 0.000 description 24
- 239000002245 particle Substances 0.000 description 11
- 238000004544 sputter deposition Methods 0.000 description 11
- 239000011247 coating layer Substances 0.000 description 8
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 7
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 6
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 238000000034 method Methods 0.000 description 5
- 230000009747 swallowing Effects 0.000 description 5
- 235000019658 bitter taste Nutrition 0.000 description 4
- 230000000873 masking effect Effects 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 230000008021 deposition Effects 0.000 description 3
- 239000007884 disintegrant Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000010828 elution Methods 0.000 description 3
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 3
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 3
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 3
- 210000000936 intestine Anatomy 0.000 description 3
- 229960005489 paracetamol Drugs 0.000 description 3
- 210000002784 stomach Anatomy 0.000 description 3
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 2
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- LOUPRKONTZGTKE-WZBLMQSHSA-N Quinine Chemical compound C([C@H]([C@H](C1)C=C)C2)C[N@@]1[C@@H]2[C@H](O)C1=CC=NC2=CC=C(OC)C=C21 LOUPRKONTZGTKE-WZBLMQSHSA-N 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 238000005229 chemical vapour deposition Methods 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 2
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 230000007721 medicinal effect Effects 0.000 description 2
- 229920000609 methyl cellulose Polymers 0.000 description 2
- 239000001923 methylcellulose Substances 0.000 description 2
- 210000002200 mouth mucosa Anatomy 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- XNGIFLGASWRNHJ-UHFFFAOYSA-L phthalate(2-) Chemical compound [O-]C(=O)C1=CC=CC=C1C([O-])=O XNGIFLGASWRNHJ-UHFFFAOYSA-L 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- 239000004014 plasticizer Substances 0.000 description 2
- 229920002401 polyacrylamide Polymers 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 230000000638 stimulation Effects 0.000 description 2
- URAYPUMNDPQOKB-UHFFFAOYSA-N triacetin Chemical compound CC(=O)OCC(OC(C)=O)COC(C)=O URAYPUMNDPQOKB-UHFFFAOYSA-N 0.000 description 2
- LUEWUZLMQUOBSB-FSKGGBMCSA-N (2s,3s,4s,5s,6r)-2-[(2r,3s,4r,5r,6s)-6-[(2r,3s,4r,5s,6s)-4,5-dihydroxy-2-(hydroxymethyl)-6-[(2r,4r,5s,6r)-4,5,6-trihydroxy-2-(hydroxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-4,5-dihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol Chemical compound O[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@@H](O[C@@H]2[C@H](O[C@@H](OC3[C@H](O[C@@H](O)[C@@H](O)[C@H]3O)CO)[C@@H](O)[C@H]2O)CO)[C@H](O)[C@H]1O LUEWUZLMQUOBSB-FSKGGBMCSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- CIVCELMLGDGMKZ-UHFFFAOYSA-N 2,4-dichloro-6-methylpyridine-3-carboxylic acid Chemical compound CC1=CC(Cl)=C(C(O)=O)C(Cl)=N1 CIVCELMLGDGMKZ-UHFFFAOYSA-N 0.000 description 1
- KHOITXIGCFIULA-UHFFFAOYSA-N Alophen Chemical compound C1=CC(OC(=O)C)=CC=C1C(C=1N=CC=CC=1)C1=CC=C(OC(C)=O)C=C1 KHOITXIGCFIULA-UHFFFAOYSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 102000011632 Caseins Human genes 0.000 description 1
- 108010076119 Caseins Proteins 0.000 description 1
- 235000001258 Cinchona calisaya Nutrition 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 206010013911 Dysgeusia Diseases 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 229920002581 Glucomannan Polymers 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 1
- FFEARJCKVFRZRR-UHFFFAOYSA-N L-Methionine Natural products CSCCC(N)C(O)=O FFEARJCKVFRZRR-UHFFFAOYSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- 229930195722 L-methionine Natural products 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 229920001218 Pullulan Polymers 0.000 description 1
- 239000004373 Pullulan Substances 0.000 description 1
- 108010073771 Soybean Proteins Proteins 0.000 description 1
- DOOTYTYQINUNNV-UHFFFAOYSA-N Triethyl citrate Chemical compound CCOC(=O)CC(O)(C(=O)OCC)CC(=O)OCC DOOTYTYQINUNNV-UHFFFAOYSA-N 0.000 description 1
- UGXQOOQUZRUVSS-ZZXKWVIFSA-N [5-[3,5-dihydroxy-2-(1,3,4-trihydroxy-5-oxopentan-2-yl)oxyoxan-4-yl]oxy-3,4-dihydroxyoxolan-2-yl]methyl (e)-3-(4-hydroxyphenyl)prop-2-enoate Chemical compound OC1C(OC(CO)C(O)C(O)C=O)OCC(O)C1OC1C(O)C(O)C(COC(=O)\C=C\C=2C=CC(O)=CC=2)O1 UGXQOOQUZRUVSS-ZZXKWVIFSA-N 0.000 description 1
- 125000004103 aminoalkyl group Chemical group 0.000 description 1
- 229920000617 arabinoxylan Polymers 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 229960000503 bisacodyl Drugs 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 239000000679 carrageenan Substances 0.000 description 1
- 229940113118 carrageenan Drugs 0.000 description 1
- LOUPRKONTZGTKE-UHFFFAOYSA-N cinchonine Natural products C1C(C(C2)C=C)CCN2C1C(O)C1=CC=NC2=CC=C(OC)C=C21 LOUPRKONTZGTKE-UHFFFAOYSA-N 0.000 description 1
- AGOYDEPGAOXOCK-KCBOHYOISA-N clarithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@](C)([C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)OC)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 AGOYDEPGAOXOCK-KCBOHYOISA-N 0.000 description 1
- 229960002626 clarithromycin Drugs 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 238000005137 deposition process Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 229960000525 diphenhydramine hydrochloride Drugs 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- WHWZLSFABNNENI-UHFFFAOYSA-N epinastine Chemical compound C1C2=CC=CC=C2C2CN=C(N)N2C2=CC=CC=C21 WHWZLSFABNNENI-UHFFFAOYSA-N 0.000 description 1
- 229960003449 epinastine Drugs 0.000 description 1
- FCZCIXQGZOUIDN-UHFFFAOYSA-N ethyl 2-diethoxyphosphinothioyloxyacetate Chemical compound CCOC(=O)COP(=S)(OCC)OCC FCZCIXQGZOUIDN-UHFFFAOYSA-N 0.000 description 1
- 229940061262 ethyl cysteine hydrochloride Drugs 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 229940046240 glucomannan Drugs 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 239000001087 glyceryl triacetate Substances 0.000 description 1
- 235000013773 glyceryl triacetate Nutrition 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 229960003943 hypromellose Drugs 0.000 description 1
- 229960001680 ibuprofen Drugs 0.000 description 1
- 238000007733 ion plating Methods 0.000 description 1
- 229940031703 low substituted hydroxypropyl cellulose Drugs 0.000 description 1
- 229960003511 macrogol Drugs 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 229960001855 mannitol Drugs 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229960005321 mecobalamin Drugs 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 229960004452 methionine Drugs 0.000 description 1
- JEWJRMKHSMTXPP-BYFNXCQMSA-M methylcobalamin Chemical compound C[Co+]N([C@]1([H])[C@H](CC(N)=O)[C@]\2(CCC(=O)NC[C@H](C)OP(O)(=O)OC3[C@H]([C@H](O[C@@H]3CO)N3C4=CC(C)=C(C)C=C4N=C3)O)C)C/2=C(C)\C([C@H](C/2(C)C)CCC(N)=O)=N\C\2=C\C([C@H]([C@@]/2(CC(N)=O)C)CCC(N)=O)=N\C\2=C(C)/C2=N[C@]1(C)[C@@](C)(CC(N)=O)[C@@H]2CCC(N)=O JEWJRMKHSMTXPP-BYFNXCQMSA-M 0.000 description 1
- 235000007672 methylcobalamin Nutrition 0.000 description 1
- 239000011585 methylcobalamin Substances 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 231100000862 numbness Toxicity 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 229910003446 platinum oxide Inorganic materials 0.000 description 1
- 229920001495 poly(sodium acrylate) polymer Polymers 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229950008882 polysorbate Drugs 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 235000019423 pullulan Nutrition 0.000 description 1
- 238000010298 pulverizing process Methods 0.000 description 1
- 229960000948 quinine Drugs 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 229940080237 sodium caseinate Drugs 0.000 description 1
- NNMHYFLPFNGQFZ-UHFFFAOYSA-M sodium polyacrylate Chemical compound [Na+].[O-]C(=O)C=C NNMHYFLPFNGQFZ-UHFFFAOYSA-M 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 235000019710 soybean protein Nutrition 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 229960002622 triacetin Drugs 0.000 description 1
- 239000001069 triethyl citrate Substances 0.000 description 1
- 235000013769 triethyl citrate Nutrition 0.000 description 1
- VMYFZRTXGLUXMZ-UHFFFAOYSA-N triethyl citrate Natural products CCOC(=O)C(O)(C(=O)OCC)C(=O)OCC VMYFZRTXGLUXMZ-UHFFFAOYSA-N 0.000 description 1
- 238000005019 vapor deposition process Methods 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 1
Images
Landscapes
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
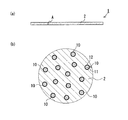
【解決手段】フィルム状製剤の製造方法は、薬物成分を含有する粉末母体の表面に、金、銀、白金等の金属、金属酸化物、ケイ素、酸化ケイ素、セルロース、コラーゲン、ツェイン、セラック、寒天、高分子デンプン、ポリビニルアセタールジエチルアミノアセテートに例示される胃溶性有機化合物、メタアクリル酸コポリマーLに例示される腸溶性有機化合物から選ばれる少なくとも一種を蒸着し、蒸着皮膜層が形成された粉末状製剤とする蒸着工程S1と、粉末状製剤が可食性の水溶性フィルム形成剤の水溶液に懸濁された懸濁液を調製する懸濁液調製工程S2と、懸濁液を基面上に流延し、乾燥させてフィルム化するフィルム化工程(流延工程S3,乾燥工程S4)とを具備する。
【選択図】図3
Description
真空度:0.8Pa
出力:0.5kW
スパッタリング時間:520秒
粉末状製剤:10質量%
HPMC:8.5質量%
結晶セルロース:63.2質量%
L−HPC:3.0質量%
その他:15.3質量%
2 フィルム基体
10 粉末状製剤
11 粉末母体
12 蒸着皮膜層
S1 蒸着工程
S2 懸濁液調製工程
S3 流延工程(フィルム化工程)
S4 乾燥工程(フィルム化工程)
Claims (2)
- 経口投与用の薬物成分を含有する粉末母体の表面に、金、銀、白金、アルミニウム、銅、チタン、マグネシウム、ケイ素、酸化ケイ素、酸化アルミニウム、酸化チタン、酸化マグネシウム、セルロース、コラーゲン、ツェイン、セラック、寒天、高分子デンプン、ポリビニルアセタールジエチルアミノアセテート、アミノアルキルメタアクリレートコポリマーE、メタアクリル酸コポリマーL、メタアクリル酸コポリマーLD、ヒドロキシプロピルメチルセルロースフタレート、ヒドロキシプロピルメチルセルロースアセテートサクシネートから選ばれる少なくとも一種が蒸着された蒸着皮膜層を備える粉末状製剤が、可食性の水溶性フィルム形成剤によりフィルム状に形成されたフィルム基体に含有されていることを特徴とするフィルム状製剤。
- 経口投与用の薬物成分を含有する粉末母体の表面に、金、銀、白金、アルミニウム、銅、チタン、マグネシウム、ケイ素、酸化ケイ素、酸化アルミニウム、酸化チタン、酸化マグネシウム、セルロース、コラーゲン、ツェイン、セラック、寒天、高分子デンプン、ポリビニルアセタールジエチルアミノアセテート、アミノアルキルメタアクリレートコポリマーE、メタアクリル酸コポリマーL、メタアクリル酸コポリマーLD、ヒドロキシプロピルメチルセルロースフタレート、ヒドロキシプロピルメチルセルロースアセテートサクシネートから選ばれる少なくとも一種を蒸着し、蒸着皮膜層が形成された粉末状製剤とする蒸着工程と、
前記粉末状製剤が可食性の水溶性フィルム形成剤の水溶液に懸濁された懸濁液を調製する懸濁液調製工程と、
前記懸濁液を基面上に流延し、乾燥させてフィルム化することにより、フィルム状製剤とするフィルム化工程と
を具備することを特徴とするフィルム状製剤の製造方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008170683A JP5466379B2 (ja) | 2008-06-30 | 2008-06-30 | フィルム状製剤及びフィルム状製剤の製造方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008170683A JP5466379B2 (ja) | 2008-06-30 | 2008-06-30 | フィルム状製剤及びフィルム状製剤の製造方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010006780A true JP2010006780A (ja) | 2010-01-14 |
| JP5466379B2 JP5466379B2 (ja) | 2014-04-09 |
Family
ID=41587677
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2008170683A Expired - Fee Related JP5466379B2 (ja) | 2008-06-30 | 2008-06-30 | フィルム状製剤及びフィルム状製剤の製造方法 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5466379B2 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10166198B2 (en) | 2013-05-24 | 2019-01-01 | Nanexa Ab | Solid nanoparticle with inorganic coating |
| WO2020080380A1 (ja) * | 2018-10-19 | 2020-04-23 | 東和薬品株式会社 | 被覆された固形製剤 |
| JPWO2021172395A1 (ja) * | 2020-02-26 | 2021-09-02 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63174657A (ja) * | 1986-12-29 | 1988-07-19 | ワーナー−ランバート・コンパニー | 可食性錠剤調製のための連続方法 |
| JPH06211649A (ja) * | 1992-11-21 | 1994-08-02 | Basf Ag | 水溶性の固形医薬有効物質の徐放型製剤およびその製造方法 |
| JP2003522139A (ja) * | 2000-02-10 | 2003-07-22 | ビーピーエスアイ ホールディングス,インコーポレーテッド | アクリル系の腸溶性コーティング組成物 |
| JP2004043316A (ja) * | 2002-07-09 | 2004-02-12 | Tsukioka:Kk | 金属膜を備えた薬または食品、その薬または食品の製造方法、および、その薬または食品を製造する、スパッタリング、真空蒸着、またはイオンプレーティング装置 |
| JP2005087101A (ja) * | 2003-09-17 | 2005-04-07 | Tsukioka:Kk | 箔押し材、および、この箔押し材を用いて箔押しされた食品、可食性フィルム等の可食物、または、水溶性の可食性フィルム、並びに、この箔押し材を用いて箔押しされた水溶性の可食性フィルムを使用した食品または飲物 |
| JP2005536443A (ja) * | 2001-10-12 | 2005-12-02 | コスモス・ファーマ | 味マスキング組成物を含む迅速溶解剤形用の均一フィルム |
| JP2008072195A (ja) * | 2006-09-12 | 2008-03-27 | Casio Hitachi Mobile Communications Co Ltd | 通話管理装置、通話管理システム、及びプログラム |
-
2008
- 2008-06-30 JP JP2008170683A patent/JP5466379B2/ja not_active Expired - Fee Related
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63174657A (ja) * | 1986-12-29 | 1988-07-19 | ワーナー−ランバート・コンパニー | 可食性錠剤調製のための連続方法 |
| JPH06211649A (ja) * | 1992-11-21 | 1994-08-02 | Basf Ag | 水溶性の固形医薬有効物質の徐放型製剤およびその製造方法 |
| JP2003522139A (ja) * | 2000-02-10 | 2003-07-22 | ビーピーエスアイ ホールディングス,インコーポレーテッド | アクリル系の腸溶性コーティング組成物 |
| JP2005536443A (ja) * | 2001-10-12 | 2005-12-02 | コスモス・ファーマ | 味マスキング組成物を含む迅速溶解剤形用の均一フィルム |
| JP2004043316A (ja) * | 2002-07-09 | 2004-02-12 | Tsukioka:Kk | 金属膜を備えた薬または食品、その薬または食品の製造方法、および、その薬または食品を製造する、スパッタリング、真空蒸着、またはイオンプレーティング装置 |
| JP2005087101A (ja) * | 2003-09-17 | 2005-04-07 | Tsukioka:Kk | 箔押し材、および、この箔押し材を用いて箔押しされた食品、可食性フィルム等の可食物、または、水溶性の可食性フィルム、並びに、この箔押し材を用いて箔押しされた水溶性の可食性フィルムを使用した食品または飲物 |
| JP2008072195A (ja) * | 2006-09-12 | 2008-03-27 | Casio Hitachi Mobile Communications Co Ltd | 通話管理装置、通話管理システム、及びプログラム |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10166198B2 (en) | 2013-05-24 | 2019-01-01 | Nanexa Ab | Solid nanoparticle with inorganic coating |
| US10478402B2 (en) | 2013-05-24 | 2019-11-19 | Nanexa Ab | Solid nanoparticle with inorganic coating |
| US10864171B2 (en) | 2013-05-24 | 2020-12-15 | Nanexa Ab | Solid nanoparticle with inorganic coating |
| US11717491B2 (en) | 2013-05-24 | 2023-08-08 | Nanexa Ab | Solid nanoparticle with inorganic coating |
| US12508236B2 (en) | 2013-05-24 | 2025-12-30 | Nanexa Ab | Solid nanoparticle with inorganic coating |
| WO2020080380A1 (ja) * | 2018-10-19 | 2020-04-23 | 東和薬品株式会社 | 被覆された固形製剤 |
| JPWO2020080380A1 (ja) * | 2018-10-19 | 2021-09-24 | 東和薬品株式会社 | 被覆された固形製剤 |
| JP7432209B2 (ja) | 2018-10-19 | 2024-02-16 | 東和薬品株式会社 | 被覆された固形製剤 |
| JPWO2021172395A1 (ja) * | 2020-02-26 | 2021-09-02 | ||
| WO2021172395A1 (ja) * | 2020-02-26 | 2021-09-02 | 東和薬品株式会社 | 被覆原薬粒子 |
| US20230094101A1 (en) * | 2020-02-26 | 2023-03-30 | Towa Pharmaceutical Co., Ltd. | Coated API Particles |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5466379B2 (ja) | 2014-04-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5871886B2 (ja) | 易服用性固形製剤 | |
| JP6105761B2 (ja) | 腸溶性被覆されたマルチ粒子徐放性ペパーミントオイル組成物及び関連方法 | |
| JP6025755B2 (ja) | 多粒子l−メントール製剤及びその関連する方法 | |
| JP2011513204A (ja) | 口腔内崩壊性固形製剤 | |
| CN102006862A (zh) | 包括弱碱性药物的组合物和控释剂型 | |
| JPWO2008081891A1 (ja) | 口腔内崩壊性固形製剤 | |
| CN103099799B (zh) | 复合膜状制剂及其制备方法 | |
| TWI835118B (zh) | 布瑞哌唑口溶膜組合物、其製備方法及用途 | |
| JP2005289868A (ja) | 経口投与剤 | |
| WO1994004135A1 (fr) | Preparation administree par voie orale et destinee a etre liberee dans le tube digestif inferieur | |
| JP4439499B2 (ja) | アムロジピン含有粒子およびそれからなる口腔内崩壊錠 | |
| WO2001010467A1 (fr) | Preparations solides contenant de la poudre de chitosane et procede de production associe | |
| JP5466379B2 (ja) | フィルム状製剤及びフィルム状製剤の製造方法 | |
| CN102178677B (zh) | 硝苯地平双层渗透泵药物组合物及其制备工艺 | |
| CN102933204A (zh) | 涂覆粒子及涂覆粒子的制造方法 | |
| CN101754754B (zh) | 含有包覆的含药物的颗粒的可分散药片及其制备方法 | |
| CN100393302C (zh) | 难溶性药物组合物渗透泵控释制剂 | |
| CN102406630A (zh) | 包被的药物递送制剂 | |
| JP2007254340A (ja) | 経口投与剤 | |
| TWI612978B (zh) | 口溶膜 | |
| JP6688819B2 (ja) | 易服用性固形製剤 | |
| JP3122478B2 (ja) | 下部消化管放出型経口製剤 | |
| JP2011093882A (ja) | 不快味マスキング粒子及びこれを含有する経口製剤 | |
| JP5241681B2 (ja) | アムロジピン含有粒子およびそれからなる口腔内崩壊錠 | |
| EP3622948A1 (en) | Multilayered formulations with dual release rate of one or more active principles |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110624 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20130207 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130212 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130410 Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20130410 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20131001 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20131129 Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20131129 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20140114 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20140124 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5466379 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |