EP2470681B1 - Acier inoxydable martensique - Google Patents
Acier inoxydable martensique Download PDFInfo
- Publication number
- EP2470681B1 EP2470681B1 EP10751807.8A EP10751807A EP2470681B1 EP 2470681 B1 EP2470681 B1 EP 2470681B1 EP 10751807 A EP10751807 A EP 10751807A EP 2470681 B1 EP2470681 B1 EP 2470681B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- chromium steel
- steel according
- chromium
- steel
- tungsten
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 229910000734 martensite Inorganic materials 0.000 title claims abstract description 21
- 229910001220 stainless steel Inorganic materials 0.000 title claims abstract description 17
- 229910000831 Steel Inorganic materials 0.000 title abstract description 40
- 239000010959 steel Substances 0.000 title abstract description 40
- 239000010935 stainless steel Substances 0.000 title 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims abstract description 20
- 229910052804 chromium Inorganic materials 0.000 claims abstract description 20
- 239000011651 chromium Substances 0.000 claims abstract description 20
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims abstract description 17
- 239000000314 lubricant Substances 0.000 claims abstract description 16
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 claims abstract description 14
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 14
- 229910052721 tungsten Inorganic materials 0.000 claims abstract description 13
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 12
- 229910052720 vanadium Inorganic materials 0.000 claims abstract description 12
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims abstract description 11
- 239000000463 material Substances 0.000 claims abstract description 11
- 229910052758 niobium Inorganic materials 0.000 claims abstract description 11
- 239000010955 niobium Substances 0.000 claims abstract description 11
- 229910052710 silicon Inorganic materials 0.000 claims abstract description 11
- 239000010936 titanium Substances 0.000 claims abstract description 11
- 229910052719 titanium Inorganic materials 0.000 claims abstract description 11
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 10
- 239000010703 silicon Substances 0.000 claims abstract description 10
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims abstract description 10
- 239000010937 tungsten Substances 0.000 claims abstract description 10
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 claims abstract description 9
- 229910052750 molybdenum Inorganic materials 0.000 claims abstract description 9
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims abstract description 8
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims abstract description 8
- 239000011733 molybdenum Substances 0.000 claims abstract description 8
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 claims abstract description 8
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 claims abstract description 8
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims abstract description 6
- 229910017052 cobalt Inorganic materials 0.000 claims abstract description 6
- 239000010941 cobalt Substances 0.000 claims abstract description 6
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 claims abstract description 6
- 229910052802 copper Inorganic materials 0.000 claims abstract description 6
- 239000010949 copper Substances 0.000 claims abstract description 6
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims abstract description 5
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 5
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims abstract description 5
- 229910052796 boron Inorganic materials 0.000 claims abstract description 5
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims abstract description 4
- 229910052742 iron Inorganic materials 0.000 claims abstract description 4
- 239000012535 impurity Substances 0.000 claims abstract description 3
- 239000005864 Sulphur Substances 0.000 claims abstract 2
- 239000004411 aluminium Substances 0.000 claims abstract 2
- 229910052748 manganese Inorganic materials 0.000 claims description 10
- 239000011572 manganese Substances 0.000 claims description 10
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 9
- 238000005299 abrasion Methods 0.000 claims description 9
- 238000001816 cooling Methods 0.000 claims description 9
- 229910052751 metal Inorganic materials 0.000 claims description 8
- 239000002184 metal Substances 0.000 claims description 8
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 claims description 7
- 239000000446 fuel Substances 0.000 claims description 5
- 238000002347 injection Methods 0.000 claims description 2
- 239000007924 injection Substances 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims 1
- 238000007790 scraping Methods 0.000 claims 1
- 238000007789 sealing Methods 0.000 claims 1
- 230000007797 corrosion Effects 0.000 abstract description 11
- 238000005260 corrosion Methods 0.000 abstract description 11
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 abstract description 2
- 238000009628 steelmaking Methods 0.000 abstract 1
- 238000012360 testing method Methods 0.000 description 18
- 239000002244 precipitate Substances 0.000 description 16
- 230000015572 biosynthetic process Effects 0.000 description 8
- 238000010438 heat treatment Methods 0.000 description 8
- 150000001247 metal acetylides Chemical class 0.000 description 8
- GVEHJMMRQRRJPM-UHFFFAOYSA-N chromium(2+);methanidylidynechromium Chemical compound [Cr+2].[Cr]#[C-].[Cr]#[C-] GVEHJMMRQRRJPM-UHFFFAOYSA-N 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- 229910003470 tongbaite Inorganic materials 0.000 description 7
- 238000009736 wetting Methods 0.000 description 7
- 239000013078 crystal Substances 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- 238000001556 precipitation Methods 0.000 description 6
- 229910001566 austenite Inorganic materials 0.000 description 5
- 230000002349 favourable effect Effects 0.000 description 5
- -1 chromium carbides Chemical class 0.000 description 4
- 239000011159 matrix material Substances 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 239000007921 spray Substances 0.000 description 4
- 229910052717 sulfur Inorganic materials 0.000 description 4
- 239000011593 sulfur Substances 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 238000002425 crystallisation Methods 0.000 description 3
- 230000008025 crystallization Effects 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 150000002430 hydrocarbons Chemical class 0.000 description 3
- 239000000523 sample Substances 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 238000005496 tempering Methods 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 238000000137 annealing Methods 0.000 description 2
- 239000003599 detergent Substances 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 230000006911 nucleation Effects 0.000 description 2
- 238000010899 nucleation Methods 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 239000005069 Extreme pressure additive Substances 0.000 description 1
- 239000005662 Paraffin oil Substances 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000002551 biofuel Substances 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- 229910021386 carbon form Inorganic materials 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- RKTYLMNFRDHKIL-UHFFFAOYSA-N copper;5,10,15,20-tetraphenylporphyrin-22,24-diide Chemical compound [Cu+2].C1=CC(C(=C2C=CC([N-]2)=C(C=2C=CC=CC=2)C=2C=CC(N=2)=C(C=2C=CC=CC=2)C2=CC=C3[N-]2)C=2C=CC=CC=2)=NC1=C3C1=CC=CC=C1 RKTYLMNFRDHKIL-UHFFFAOYSA-N 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 239000004519 grease Substances 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 150000002611 lead compounds Chemical class 0.000 description 1
- 239000010687 lubricating oil Substances 0.000 description 1
- 238000005461 lubrication Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 230000036316 preload Effects 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000009958 sewing Methods 0.000 description 1
- 238000010583 slow cooling Methods 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000002604 ultrasonography Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 229910000859 α-Fe Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/22—Ferrous alloys, e.g. steel alloys containing chromium with molybdenum or tungsten
Definitions
- the invention relates to a stainless martensitic chromium steel and its use.
- Such steels are known in large numbers and are suitable depending on their composition for a very different range of uses.
- German patent specification describes 100 27 049 B4 a martensitic chromium steel with 0.4 to 0.75% carbon, up to 0.7% silicon, up to 0.2% nickel, 0.4 to 1.6% manganese, 0.02 to 0.15% sulfur, 12 to 19% chromium, 0.5 to 1.5% molybdenum, up to 1.5% tungsten, up to 0.1% nitrogen and 0.05 to 0.3% vanadium, titanium and niobium singly or side by side and up to 0.008% boron.
- This steel has good processability, corrosion resistance and low plastic deformability and high wear and abrasion resistance; it is therefore suitable without a galvanic coating as a material for industrial needles and in particular allows a high sewing speed.
- the material is not very suitable for use, the characteristic feature of which is a sliding contact metal / metal in the presence of a lubricant film.
- Crucial here is especially in the case of systems for conveying or compressing fuels or in piston rings and wiper seals and dosing or valve needles for chemical or pharmaceutical mixtures, the stability and the adhesion of the wear-reducing surface layer.
- stabilization is not possible in various systems, such as with new methanol and ethanol containing fuels.
- the invention is therefore based on the problem of finding a stainless martensitic chromium steel, which has better wettability due to its chemical affinity and strong adhesion forces and forms a stable lubricant film, which is much more difficult to disturb or displace than conventional steels of this type and consequently causes less wear.
- the solution to this problem consists in a martensitic chromium steel with 0.40 up to 0.80% carbon 0.20 up to 1.50% silicon 0.15 up to 1.00% nickel 0.30 up to 1.00% manganese 0,015 up to 0.035% sulfur 16 up to 18% Chrome, 1.25 up to 1.50% molybdenum up to 0.8% tungsten 0.04 up to 0.08% nitrogen 0.15 up to 0.20% vanadium, up to 0.05% titanium up to 0.05% niobium 0.001 up to 0.03% aluminum 0.02 up to 0.5% copper up to 0.5% cobalt up to 0.004% Boron, Remaining iron, including impurities caused by melting.
- the steel contains each individually or side by side 0.55 up to 0.75% Carbon, up to 0.65% Silicon, up to 0.8% manganese at least 0.001% Tungsten.
- the carbon is austenite and therefore stabilizes the austenitic crystal lattice.
- carbon together with the carbide formers, also contributes to carbide precipitations, which increase the hardness and abrasion resistance of the steel.
- carbide precipitations which increase the hardness and abrasion resistance of the steel.
- the steel therefore contains 0.40 to 0.80%, preferably 0.55 to 0.75% carbon.
- Silicon serves as a deoxidizer. However, higher contents can lead to the formation of intermetallic phases. On the other hand, silicon is also a ferrite former. The steel therefore contains 0.2 to 1.5% silicon, preferably up to 0.65% silicon.
- Nickel belongs to the austenite blidners, but the austenite part in the microstructure carries the risk of a deterioration of the wear properties. On the other hand, nickel is also an advantage as part of the crystal lattice with its influence on the c / a ratio of the martensite. The steel therefore contains 0.15 to 1% nickel.
- Manganese stabilizes the austenite and advantageously shifts the martensite formation to lower temperatures.
- the maximum level of manganese is therefore 1%, but a minimum content of 0.30% should not be undercut, because manganese simultaneously alters the c / a ratio of the newly formed martensite and advantageously influences the precipitation behavior of the fine precipitates in the austenitic region.
- the sulfur content is limited to a maximum of 0.035%, since at higher sulfur levels, troublesome sulfidic precipitates may occur.
- Chromium is required to ensure the corrosion resistance of the steel in combination with its molybdenum content of 1.25 to 1.50%, in particular adequate resistance to pitting corrosion.
- the chromium content is therefore at least 16%.
- its content is limited to 18%.
- the steel contains from 0.001 to 0.8% tungsten, preferably at least 0.001% tungsten, which together with the iron and molybdenum forms mixed carbides, which contribute significantly to the hot strength of the steel and lead to secondary carbide secondary precipitation during tempering.
- the nitrogen together with the carbon forms carbonitrides, but also worsens the wettability of the steel to hydrocarbon lubricants; the upper content limit for nitrogen is therefore 0.08%.
- the steel contains vanadium, niobium and titanium as carbide formers with the advantage that they form nucleation nuclei for the formation of chromium carbides even at very high temperatures in view of their high affinity for carbon.
- the vanadium content is therefore 0.15 to 0.20% at titanium and niobium contents of up to 0.05%.
- the steel also contains 0.001 to 0.03% aluminum as a deoxidizer, but not more, because higher aluminum contents have an embrittling effect.
- the maximum copper content is 0.5% and, especially during tempering, leads to fine-grained secondary precipitations, which together with other precipitates improve the wettability of the steel for oils or hydrocarbons.
- cobalt promotes the formation of ⁇ -carbides and other fine precipitates; it improves the hot strength of the steel in this way. For cost reasons, however, a cobalt content of 0.5% should not be exceeded.
- the cooling rate should not exceed 50 ° C / sec (heat treatment A), so as not to affect the carbide formation and to prevent part of the reactants from remaining metastable at interstitial sites when the carbides are formed.
- martensite spontaneously forms below the MS temperature from the cubic body-centered crystal lattice, with the result that the previously formed carbides are incorporated in the martensite, but the stress state of the matrix is lower, the more finely distributed the carbides are. This condition greatly enhances the lubricant or oil wetting of the surface of the steel.
- the martensite from the austenite transformation has a tetragonal distorted crystal lattice with a ratio of the crystal axes a / c over 1.
- the martensite formed during the conversion can be influenced by a heat treatment in the temperature range below 550 ° C. following martensite formation in such a way that the martensite Crystal axis ratio s / c reduced, which has an extremely beneficial effect on the material properties.
- the nucleation is less favorable, so that the chromium carbide can be subject to crystallization inhibition and crystallized later. Associated with this is a coarser grained chromium carbide with an unfavorable distribution in the matrix.
- coarse MC carbides of niobium, titanium and vanadium may be formed instead of fine primary carbides. Thus, the effect of the aforementioned elements is lost as carbide crystallization nuclei for the chromium carbides.
- the heat treatment B consists in a starter annealing at 100 to 550 ° C, preferably at least 200 ° C instead and leads to the formation of fine precipitates in the stabilized in the previous heat treatment A in the temperature range of 1100 to 900 ° C martensite.
- Table I below shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five
- Table II below shows the cumulative values for K1 to K3 resulting from the analyzes. ⁇ b> Table II ⁇ / b> K1 K2 K3 0.12 1.35 0.18 0.10 0.93 0.18 0.18 6.96 1.46 0.13 3.80 0.36 0.36 8.53 0.25 0.19 0.32 0.59 0.17 0.17 0.82 0.19 0.43 1.35
- Austenitizing annealing indicated by A in Table III took place at 1020 ° C or 1050 ° C, followed by rapid cooling at a cooling rate of at least 50 ° C / sec to 800 ° C followed by cooling within 5 min. to 300 ° C and a slow cooling to room temperature.
- the samples were finally heated according to the test series B to a temperature of 100 to 530 ° C and cooled at a rate of 100 ° C / h to room temperature.
- the samples were then ground and polished, cleaned in an aqueous ultrasonic bath at 50 ° C, with hot distilled water under the action of ultrasound for another 20 min. freed of detergent residues and then dried.
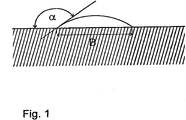
- 10 ⁇ l of paraffin oil were then applied to each sample by means of microdosing, and the oil droplets then forming were measured with respect to their width B, as shown schematically in FIG Fig. 1 results.
- the measurement results found are recorded together with the respective austenitizing temperature in the following Table III. ⁇ b> Table III ⁇ / b> Verse no. Leg.
- the abrasion and wear resistance was determined by means of a modified "pin on disk” test *, whereby the cylindrical samples were first ground flat, then cleaned, clamped in a holder and then correspondingly Fig. 2 Under a rotating steel shaft with an eccentric carbide ball under load and spring preload dynamically loaded. During the experiment, the contact zone between the sliding hard metal ball and the sample surface was steadily lubricated by the dripping of lubricating oil. At the end of the test period, the average width R of the sliding or wear track was then measured under a microscope at four points offset by ninety degrees from each other, and from these four measured values in each case the mean value R (see Table IV). Fig. 3 ) educated. In this case, a wide wear track or a large R value indicates that the steel ball has penetrated deeper and accordingly with greater width into the samples and that the sample material accordingly has a lower seal strength than those samples with a small wear track width R.
- the result of a salt spray test can serve as an indicator of the size and distribution of the chromium carbide precipitates.
- the samples were therefore subjected to 120 hours of corrosion testing as part of a modified salt spray test with a 3% NaCl solution and 5% alcohol.
- test results are listed in column 7 of Table III .
- the results of the salt spray test are an indicator of the size and distribution of chromium carbide precipitates.
- the test results show that the wetting behavior of the inventive martensitic chromium steels for lubricants is significantly better than that of the comparative steels.
- the good wettability results in less adhesive wear in the presence of lubrication.
- Not only the chemical composition of the steel is of crucial importance.
- a significant influence on the wettability is also exerted by a heat treatment of the samples. This is shown by the larger C values and the smaller R values of the samples according to the invention of experiments 9 to 17.
- the factors K1 and K3 also show that the favorable test results are based on a more favorable precipitation of the carbides and other phases as well as the basic structure.
Claims (13)
- Acier inoxydable martensitique comprenant :
de 0,4 à 0,8 % de carbone, de 0,2 à 1,5 % de silicium, de 0,15 à 1,0 % de nickel, de 0,3 à 1,00 % de manganèse, de 0,015 à 0,035 % de soufre, de 16 à 18 % de chrome, de 1,25 à 1,50 % de molybdène, jusqu'à 0,8 % de tungstène, de 0,04 % à 0,08 % d'azote, de 0,15 à 0,20 % de vanadium, jusqu'à 0,05 % de titane, jusqu'à 0,05 % de niobium, de 0,001 à 0,03 % d'aluminium, de 0,02 à 0,5 % de cuivre, jusqu'à 0,5 % de cobalt, jusqu'à 0,040 % de bore, le reste étant du fer II avec des impuretés consécutives à la production d'acier. - Acier selon la revendication 1, qui comprend à l'état élémentaire ou combiné
de 0,55 à 0,75 % de carbone, jusqu'à 0,65 % de silicium, jusqu'à 0,8 % de manganèse, au moins 0,001 % de tungstène. - Acier selon l'une des revendications 1 à 6, caractérisé en ce qu'il subit une calcination en passant de 1 020 à 1 050°C pour passer à la phase austénitique, puis est rapidement refroidi selon une vitesse de refroidissement d'au moins 50 °C/s jusqu'à atteindre une température 800 °C, puis est refroidi en cinq minutes jusqu'à atteindre une température 300 °C à l'aide flux d'air à la température ambiante.
- Acier selon la revendication 7, qui est à nouveau chauffé de 100 à 530 °C puis est refroidi lentement jusqu'à atteindre la température ambiante selon une vitesse de refroidissement de 100 °C/h.
- Utilisation d'un acier selon l'une des revendications 1 à 8, en tant que matière première résistant aux piqûres de corrosion.
- Utilisation d'un acier selon l'une des revendications 1 à 8, en tant que matière première résistant à l'usure par frottement.
- Utilisation selon la revendication 9 ou 10 avec un film intermédiaire lubrifiant.
- Utilisation selon la revendication 11 dans un contact à frottement métal/métal avec une interface intermédiaire lubrifiante.
- Utilisation d'un acier selon l'une des revendications 1 à 7 en tant matière première pour un fabricant de tiges de soupape, d'aiguilles de commande et de dosage, de
douilles de guidage, de composants fonctionnels de systèmes d'injection de carburant, de segments de piston pour organes plongeurs et moteurs ainsi que de bagues d'étanchéité et de segments racleurs pour compresseurs.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PL10751807T PL2470681T3 (pl) | 2009-08-24 | 2010-08-18 | Nierdzewna martenzytyczna stal chromowa |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102009038382A DE102009038382A1 (de) | 2009-08-24 | 2009-08-24 | Rostfreier martensitischer Chromstahl |
| PCT/EP2010/005067 WO2011023326A1 (fr) | 2009-08-24 | 2010-08-18 | Acier chromé martensitique inoxydable |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2470681A1 EP2470681A1 (fr) | 2012-07-04 |
| EP2470681B1 true EP2470681B1 (fr) | 2013-11-20 |
Family
ID=42937637
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP10751807.8A Active EP2470681B1 (fr) | 2009-08-24 | 2010-08-18 | Acier inoxydable martensique |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP2470681B1 (fr) |
| DE (1) | DE102009038382A1 (fr) |
| ES (1) | ES2446716T3 (fr) |
| PL (1) | PL2470681T3 (fr) |
| WO (1) | WO2011023326A1 (fr) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102011102293A1 (de) | 2011-02-24 | 2012-08-30 | Stahlwerk Ergste Westig Gmbh | Verwendung eines Chromstahls mit martensitischem Gefüge und karbidischen Einschlüssen |
| DE102017003965B4 (de) * | 2017-04-25 | 2019-12-12 | Zapp Precision Metals Gmbh | Martensitischer Chromstahl, Stahlfolie, perforierte und/oder gelochte Komponente aus einer Stahlfolie, Verfahren zum Herstellen einer Stahlfolie |
| CN115109891B (zh) * | 2022-07-14 | 2022-12-20 | 中北大学 | 一种高碳高铬含氮马氏体不锈钢及其碳化物细化方法 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SE404029B (sv) * | 1968-05-31 | 1978-09-18 | Uddeholms Ab | Forfarande for framstellning av emnen for tunna eggverktyg av herdat stal |
| US3990892A (en) * | 1972-03-28 | 1976-11-09 | Kabushiki Kaisha Fujikoshi | Wear resistant and heat resistant alloy steels |
| AT401387B (de) * | 1994-06-29 | 1996-08-26 | Boehler Ybbstalwerke | Korrosionsbeständige legierung und verfahren zur herstellung korrosionsbeständiger schneidwaren |
| DE19808276C2 (de) * | 1998-02-27 | 2003-12-24 | Stahlwerk Ergste Westig Gmbh | Stahllegierung für Gleitelemente |
| JP2000337389A (ja) * | 1999-03-19 | 2000-12-05 | Nsk Ltd | 転がり軸受 |
| DE10027049B4 (de) | 2000-01-17 | 2004-09-16 | Stahlwerk Ergste Westig Gmbh | Verwendung einer Chrom-Stahllegierung |
| DE102004051629B4 (de) * | 2004-10-23 | 2006-08-24 | Stahlwerk Ergste Westig Gmbh | Rostfreier martensitischer Chromstahl |
| JP2008291307A (ja) * | 2007-05-24 | 2008-12-04 | Daido Steel Co Ltd | 金型の製造方法、金型用鋼材の製造方法、及び、金型用鋼材を用いた金型の製造方法 |
| DE102007025758A1 (de) * | 2007-06-01 | 2008-12-04 | Mahle International Gmbh | Dichtring |
-
2009
- 2009-08-24 DE DE102009038382A patent/DE102009038382A1/de not_active Withdrawn
-
2010
- 2010-08-18 EP EP10751807.8A patent/EP2470681B1/fr active Active
- 2010-08-18 PL PL10751807T patent/PL2470681T3/pl unknown
- 2010-08-18 WO PCT/EP2010/005067 patent/WO2011023326A1/fr active Application Filing
- 2010-08-18 ES ES10751807.8T patent/ES2446716T3/es active Active
Also Published As
| Publication number | Publication date |
|---|---|
| PL2470681T3 (pl) | 2014-05-30 |
| EP2470681A1 (fr) | 2012-07-04 |
| WO2011023326A1 (fr) | 2011-03-03 |
| DE102009038382A1 (de) | 2011-03-03 |
| ES2446716T3 (es) | 2014-03-10 |
| DE102009038382A8 (de) | 2011-06-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AT502397B1 (de) | Legierung für wälzlager | |
| DE60214456T2 (de) | Martensitischer rostfreier Stahl mit hoher Härte und guter Korrosionsbeständigkeit | |
| DE69604341T3 (de) | Martensitischer. rostfreier stahl mit guter beständigkeit gegen lochfrasskorrosion und mit hoher härte | |
| DE60215655T2 (de) | Martensitischer nichtrostender stahl | |
| EP3228724A1 (fr) | Procédé de réglage de la conductivité thermique d'un acier, acier à outil, en particulier acier pour travail à chaud et objet en acier | |
| DE2040308A1 (de) | Austenitischer rostfreier Stahl | |
| DE3041565C2 (fr) | ||
| DE112019006482T5 (de) | Karbonitrierte lagerkomponente | |
| AT409389B (de) | Pm-schnellarbeitsstahl mit hoher warmfestigkeit | |
| EP1233080B1 (fr) | Acier pour moules à matières plastiques et son traitement thermique | |
| EP2470681B1 (fr) | Acier inoxydable martensique | |
| DE2447137B2 (de) | Gegen gruebchenkorrosion bestaendige stahllegierung | |
| DE60030175T2 (de) | Automatenlegierung | |
| DE112019006504T5 (de) | Stahlmaterial als ausgangsmaterial für karbonitrierte lagerkomponente | |
| AT410550B (de) | Reaktionsträger werkstoff mit erhöhter härte für thermisch beanspruchte bauteile | |
| DE112008003146T5 (de) | Stahl für eine Induktionshärtung mit einer hervorragenden Kaltverarbeitungsfähigkeit, Rollglied, das aus einem derartigen Stahl ausgebildet ist, und Bewegungsführungsvorrichtung, die ein derartiges Rollglied verwendet | |
| DE60011115T2 (de) | Stahlmaterial, dessen verwendung und herstellung | |
| EP3061838B1 (fr) | Produit longitudinal bainitique nu et son procédé de fabrication | |
| DE69909940T2 (de) | Teile aus martensitischem rostfreiem Stahl und Verfahren zu ihrer Herstellung | |
| WO2022049282A1 (fr) | Produit plat en acier laminé à chaud et procédé de production d'un produit plat en acier laminé à chaud | |
| EP3781719B1 (fr) | Alliage de cuivre, zinc, nickel et manganèse | |
| EP1471160B1 (fr) | Objet en acier travaillé à froid | |
| DE60100730T2 (de) | Austenitischer stahl | |
| EP1445339B1 (fr) | Alliage et article à haute résistance thermique et à haute stabilité thermique | |
| EP2453027B1 (fr) | Produit déformé à chaud et son procédé de fabrication |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20120320 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| DAX | Request for extension of the european patent (deleted) | ||
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20130918 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 641717 Country of ref document: AT Kind code of ref document: T Effective date: 20131215 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: E. BLUM AND CO. AG PATENT- UND MARKENANWAELTE , CH |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 502010005436 Country of ref document: DE Effective date: 20140116 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2446716 Country of ref document: ES Kind code of ref document: T3 Effective date: 20140310 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: PC2A Owner name: ZAPP PRECISION METALS GMBH Effective date: 20140409 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140220 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140320 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| RAP2 | Party data changed (patent owner data changed or rights of a patent transferred) |
Owner name: ZAPP PRECISION METALS GMBH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: ZAPP PRECISION METALS GMBH, DE Free format text: FORMER OWNER: STAHLWERK ERGSTE WESTIG GMBH, DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: PL Ref legal event code: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140320 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 502010005436 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| 26N | No opposition filed |
Effective date: 20140821 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 502010005436 Country of ref document: DE Representative=s name: KOENIG SZYNKA TILMANN VON RENESSE PATENTANWAEL, DE |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 502010005436 Country of ref document: DE Effective date: 20140821 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 502010005436 Country of ref document: DE Owner name: ZAPP PRECISION METALS GMBH, DE Free format text: FORMER OWNER: STAHLWERK ERGSTE WESTIG GMBH, 58239 SCHWERTE, DE Effective date: 20141104 Ref country code: DE Ref legal event code: R082 Ref document number: 502010005436 Country of ref document: DE Representative=s name: KOENIG SZYNKA TILMANN VON RENESSE PATENTANWAEL, DE Effective date: 20141104 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: LU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140818 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20140818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140831 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140221 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20100818 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 7 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 641717 Country of ref document: AT Kind code of ref document: T Effective date: 20150818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150818 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230513 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: TR Payment date: 20230811 Year of fee payment: 14 Ref country code: IT Payment date: 20230831 Year of fee payment: 14 Ref country code: ES Payment date: 20230918 Year of fee payment: 14 Ref country code: CH Payment date: 20230902 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PL Payment date: 20230804 Year of fee payment: 14 Ref country code: FR Payment date: 20230821 Year of fee payment: 14 Ref country code: DE Payment date: 20230919 Year of fee payment: 14 |