EP2470681B1 - Stainless steel martensitic steel - Google Patents
Stainless steel martensitic steel Download PDFInfo
- Publication number
- EP2470681B1 EP2470681B1 EP10751807.8A EP10751807A EP2470681B1 EP 2470681 B1 EP2470681 B1 EP 2470681B1 EP 10751807 A EP10751807 A EP 10751807A EP 2470681 B1 EP2470681 B1 EP 2470681B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- chromium steel
- steel according
- chromium
- steel
- tungsten
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 229910000734 martensite Inorganic materials 0.000 title claims abstract description 21
- 229910001220 stainless steel Inorganic materials 0.000 title claims abstract description 17
- 229910000831 Steel Inorganic materials 0.000 title abstract description 40
- 239000010959 steel Substances 0.000 title abstract description 40
- 239000010935 stainless steel Substances 0.000 title 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims abstract description 20
- 229910052804 chromium Inorganic materials 0.000 claims abstract description 20
- 239000011651 chromium Substances 0.000 claims abstract description 20
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims abstract description 17
- 239000000314 lubricant Substances 0.000 claims abstract description 16
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 claims abstract description 14
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 14
- 229910052721 tungsten Inorganic materials 0.000 claims abstract description 13
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 12
- 229910052720 vanadium Inorganic materials 0.000 claims abstract description 12
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims abstract description 11
- 239000000463 material Substances 0.000 claims abstract description 11
- 229910052758 niobium Inorganic materials 0.000 claims abstract description 11
- 239000010955 niobium Substances 0.000 claims abstract description 11
- 229910052710 silicon Inorganic materials 0.000 claims abstract description 11
- 239000010936 titanium Substances 0.000 claims abstract description 11
- 229910052719 titanium Inorganic materials 0.000 claims abstract description 11
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 10
- 239000010703 silicon Substances 0.000 claims abstract description 10
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims abstract description 10
- 239000010937 tungsten Substances 0.000 claims abstract description 10
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 claims abstract description 9
- 229910052750 molybdenum Inorganic materials 0.000 claims abstract description 9
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims abstract description 8
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims abstract description 8
- 239000011733 molybdenum Substances 0.000 claims abstract description 8
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 claims abstract description 8
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 claims abstract description 8
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims abstract description 6
- 229910017052 cobalt Inorganic materials 0.000 claims abstract description 6
- 239000010941 cobalt Substances 0.000 claims abstract description 6
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 claims abstract description 6
- 229910052802 copper Inorganic materials 0.000 claims abstract description 6
- 239000010949 copper Substances 0.000 claims abstract description 6
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims abstract description 5
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 5
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims abstract description 5
- 229910052796 boron Inorganic materials 0.000 claims abstract description 5
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims abstract description 4
- 229910052742 iron Inorganic materials 0.000 claims abstract description 4
- 239000012535 impurity Substances 0.000 claims abstract description 3
- 239000005864 Sulphur Substances 0.000 claims abstract 2
- 239000004411 aluminium Substances 0.000 claims abstract 2
- 229910052748 manganese Inorganic materials 0.000 claims description 10
- 239000011572 manganese Substances 0.000 claims description 10
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 9
- 238000005299 abrasion Methods 0.000 claims description 9
- 238000001816 cooling Methods 0.000 claims description 9
- 229910052751 metal Inorganic materials 0.000 claims description 8
- 239000002184 metal Substances 0.000 claims description 8
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 claims description 7
- 239000000446 fuel Substances 0.000 claims description 5
- 238000002347 injection Methods 0.000 claims description 2
- 239000007924 injection Substances 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims 1
- 238000007790 scraping Methods 0.000 claims 1
- 238000007789 sealing Methods 0.000 claims 1
- 230000007797 corrosion Effects 0.000 abstract description 11
- 238000005260 corrosion Methods 0.000 abstract description 11
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 abstract description 2
- 238000009628 steelmaking Methods 0.000 abstract 1
- 238000012360 testing method Methods 0.000 description 18
- 239000002244 precipitate Substances 0.000 description 16
- 230000015572 biosynthetic process Effects 0.000 description 8
- 238000010438 heat treatment Methods 0.000 description 8
- 150000001247 metal acetylides Chemical class 0.000 description 8
- GVEHJMMRQRRJPM-UHFFFAOYSA-N chromium(2+);methanidylidynechromium Chemical compound [Cr+2].[Cr]#[C-].[Cr]#[C-] GVEHJMMRQRRJPM-UHFFFAOYSA-N 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- 229910003470 tongbaite Inorganic materials 0.000 description 7
- 238000009736 wetting Methods 0.000 description 7
- 239000013078 crystal Substances 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- 238000001556 precipitation Methods 0.000 description 6
- 229910001566 austenite Inorganic materials 0.000 description 5
- 230000002349 favourable effect Effects 0.000 description 5
- -1 chromium carbides Chemical class 0.000 description 4
- 239000011159 matrix material Substances 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 239000007921 spray Substances 0.000 description 4
- 229910052717 sulfur Inorganic materials 0.000 description 4
- 239000011593 sulfur Substances 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 238000002425 crystallisation Methods 0.000 description 3
- 230000008025 crystallization Effects 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 150000002430 hydrocarbons Chemical class 0.000 description 3
- 239000000523 sample Substances 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 238000005496 tempering Methods 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 238000000137 annealing Methods 0.000 description 2
- 239000003599 detergent Substances 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 230000006911 nucleation Effects 0.000 description 2
- 238000010899 nucleation Methods 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 239000005069 Extreme pressure additive Substances 0.000 description 1
- 239000005662 Paraffin oil Substances 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000002551 biofuel Substances 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- 229910021386 carbon form Inorganic materials 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- RKTYLMNFRDHKIL-UHFFFAOYSA-N copper;5,10,15,20-tetraphenylporphyrin-22,24-diide Chemical compound [Cu+2].C1=CC(C(=C2C=CC([N-]2)=C(C=2C=CC=CC=2)C=2C=CC(N=2)=C(C=2C=CC=CC=2)C2=CC=C3[N-]2)C=2C=CC=CC=2)=NC1=C3C1=CC=CC=C1 RKTYLMNFRDHKIL-UHFFFAOYSA-N 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 239000004519 grease Substances 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 150000002611 lead compounds Chemical class 0.000 description 1
- 239000010687 lubricating oil Substances 0.000 description 1
- 238000005461 lubrication Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 230000036316 preload Effects 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000009958 sewing Methods 0.000 description 1
- 238000010583 slow cooling Methods 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000002604 ultrasonography Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 229910000859 α-Fe Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/22—Ferrous alloys, e.g. steel alloys containing chromium with molybdenum or tungsten
Definitions
- the invention relates to a stainless martensitic chromium steel and its use.
- Such steels are known in large numbers and are suitable depending on their composition for a very different range of uses.
- German patent specification describes 100 27 049 B4 a martensitic chromium steel with 0.4 to 0.75% carbon, up to 0.7% silicon, up to 0.2% nickel, 0.4 to 1.6% manganese, 0.02 to 0.15% sulfur, 12 to 19% chromium, 0.5 to 1.5% molybdenum, up to 1.5% tungsten, up to 0.1% nitrogen and 0.05 to 0.3% vanadium, titanium and niobium singly or side by side and up to 0.008% boron.
- This steel has good processability, corrosion resistance and low plastic deformability and high wear and abrasion resistance; it is therefore suitable without a galvanic coating as a material for industrial needles and in particular allows a high sewing speed.
- the material is not very suitable for use, the characteristic feature of which is a sliding contact metal / metal in the presence of a lubricant film.
- Crucial here is especially in the case of systems for conveying or compressing fuels or in piston rings and wiper seals and dosing or valve needles for chemical or pharmaceutical mixtures, the stability and the adhesion of the wear-reducing surface layer.
- stabilization is not possible in various systems, such as with new methanol and ethanol containing fuels.
- the invention is therefore based on the problem of finding a stainless martensitic chromium steel, which has better wettability due to its chemical affinity and strong adhesion forces and forms a stable lubricant film, which is much more difficult to disturb or displace than conventional steels of this type and consequently causes less wear.
- the solution to this problem consists in a martensitic chromium steel with 0.40 up to 0.80% carbon 0.20 up to 1.50% silicon 0.15 up to 1.00% nickel 0.30 up to 1.00% manganese 0,015 up to 0.035% sulfur 16 up to 18% Chrome, 1.25 up to 1.50% molybdenum up to 0.8% tungsten 0.04 up to 0.08% nitrogen 0.15 up to 0.20% vanadium, up to 0.05% titanium up to 0.05% niobium 0.001 up to 0.03% aluminum 0.02 up to 0.5% copper up to 0.5% cobalt up to 0.004% Boron, Remaining iron, including impurities caused by melting.
- the steel contains each individually or side by side 0.55 up to 0.75% Carbon, up to 0.65% Silicon, up to 0.8% manganese at least 0.001% Tungsten.
- the carbon is austenite and therefore stabilizes the austenitic crystal lattice.
- carbon together with the carbide formers, also contributes to carbide precipitations, which increase the hardness and abrasion resistance of the steel.
- carbide precipitations which increase the hardness and abrasion resistance of the steel.
- the steel therefore contains 0.40 to 0.80%, preferably 0.55 to 0.75% carbon.
- Silicon serves as a deoxidizer. However, higher contents can lead to the formation of intermetallic phases. On the other hand, silicon is also a ferrite former. The steel therefore contains 0.2 to 1.5% silicon, preferably up to 0.65% silicon.
- Nickel belongs to the austenite blidners, but the austenite part in the microstructure carries the risk of a deterioration of the wear properties. On the other hand, nickel is also an advantage as part of the crystal lattice with its influence on the c / a ratio of the martensite. The steel therefore contains 0.15 to 1% nickel.
- Manganese stabilizes the austenite and advantageously shifts the martensite formation to lower temperatures.
- the maximum level of manganese is therefore 1%, but a minimum content of 0.30% should not be undercut, because manganese simultaneously alters the c / a ratio of the newly formed martensite and advantageously influences the precipitation behavior of the fine precipitates in the austenitic region.
- the sulfur content is limited to a maximum of 0.035%, since at higher sulfur levels, troublesome sulfidic precipitates may occur.
- Chromium is required to ensure the corrosion resistance of the steel in combination with its molybdenum content of 1.25 to 1.50%, in particular adequate resistance to pitting corrosion.
- the chromium content is therefore at least 16%.
- its content is limited to 18%.
- the steel contains from 0.001 to 0.8% tungsten, preferably at least 0.001% tungsten, which together with the iron and molybdenum forms mixed carbides, which contribute significantly to the hot strength of the steel and lead to secondary carbide secondary precipitation during tempering.
- the nitrogen together with the carbon forms carbonitrides, but also worsens the wettability of the steel to hydrocarbon lubricants; the upper content limit for nitrogen is therefore 0.08%.
- the steel contains vanadium, niobium and titanium as carbide formers with the advantage that they form nucleation nuclei for the formation of chromium carbides even at very high temperatures in view of their high affinity for carbon.
- the vanadium content is therefore 0.15 to 0.20% at titanium and niobium contents of up to 0.05%.
- the steel also contains 0.001 to 0.03% aluminum as a deoxidizer, but not more, because higher aluminum contents have an embrittling effect.
- the maximum copper content is 0.5% and, especially during tempering, leads to fine-grained secondary precipitations, which together with other precipitates improve the wettability of the steel for oils or hydrocarbons.
- cobalt promotes the formation of ⁇ -carbides and other fine precipitates; it improves the hot strength of the steel in this way. For cost reasons, however, a cobalt content of 0.5% should not be exceeded.
- the cooling rate should not exceed 50 ° C / sec (heat treatment A), so as not to affect the carbide formation and to prevent part of the reactants from remaining metastable at interstitial sites when the carbides are formed.
- martensite spontaneously forms below the MS temperature from the cubic body-centered crystal lattice, with the result that the previously formed carbides are incorporated in the martensite, but the stress state of the matrix is lower, the more finely distributed the carbides are. This condition greatly enhances the lubricant or oil wetting of the surface of the steel.
- the martensite from the austenite transformation has a tetragonal distorted crystal lattice with a ratio of the crystal axes a / c over 1.
- the martensite formed during the conversion can be influenced by a heat treatment in the temperature range below 550 ° C. following martensite formation in such a way that the martensite Crystal axis ratio s / c reduced, which has an extremely beneficial effect on the material properties.
- the nucleation is less favorable, so that the chromium carbide can be subject to crystallization inhibition and crystallized later. Associated with this is a coarser grained chromium carbide with an unfavorable distribution in the matrix.
- coarse MC carbides of niobium, titanium and vanadium may be formed instead of fine primary carbides. Thus, the effect of the aforementioned elements is lost as carbide crystallization nuclei for the chromium carbides.
- the heat treatment B consists in a starter annealing at 100 to 550 ° C, preferably at least 200 ° C instead and leads to the formation of fine precipitates in the stabilized in the previous heat treatment A in the temperature range of 1100 to 900 ° C martensite.
- Table I below shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five conventional comparative steels V1 to V5 and three steels E1 to E3 which fall under the invention.
- Table I shows the analyzes of five
- Table II below shows the cumulative values for K1 to K3 resulting from the analyzes. ⁇ b> Table II ⁇ / b> K1 K2 K3 0.12 1.35 0.18 0.10 0.93 0.18 0.18 6.96 1.46 0.13 3.80 0.36 0.36 8.53 0.25 0.19 0.32 0.59 0.17 0.17 0.82 0.19 0.43 1.35
- Austenitizing annealing indicated by A in Table III took place at 1020 ° C or 1050 ° C, followed by rapid cooling at a cooling rate of at least 50 ° C / sec to 800 ° C followed by cooling within 5 min. to 300 ° C and a slow cooling to room temperature.
- the samples were finally heated according to the test series B to a temperature of 100 to 530 ° C and cooled at a rate of 100 ° C / h to room temperature.
- the samples were then ground and polished, cleaned in an aqueous ultrasonic bath at 50 ° C, with hot distilled water under the action of ultrasound for another 20 min. freed of detergent residues and then dried.
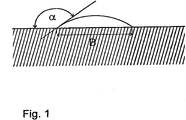
- 10 ⁇ l of paraffin oil were then applied to each sample by means of microdosing, and the oil droplets then forming were measured with respect to their width B, as shown schematically in FIG Fig. 1 results.
- the measurement results found are recorded together with the respective austenitizing temperature in the following Table III. ⁇ b> Table III ⁇ / b> Verse no. Leg.
- the abrasion and wear resistance was determined by means of a modified "pin on disk” test *, whereby the cylindrical samples were first ground flat, then cleaned, clamped in a holder and then correspondingly Fig. 2 Under a rotating steel shaft with an eccentric carbide ball under load and spring preload dynamically loaded. During the experiment, the contact zone between the sliding hard metal ball and the sample surface was steadily lubricated by the dripping of lubricating oil. At the end of the test period, the average width R of the sliding or wear track was then measured under a microscope at four points offset by ninety degrees from each other, and from these four measured values in each case the mean value R (see Table IV). Fig. 3 ) educated. In this case, a wide wear track or a large R value indicates that the steel ball has penetrated deeper and accordingly with greater width into the samples and that the sample material accordingly has a lower seal strength than those samples with a small wear track width R.
- the result of a salt spray test can serve as an indicator of the size and distribution of the chromium carbide precipitates.
- the samples were therefore subjected to 120 hours of corrosion testing as part of a modified salt spray test with a 3% NaCl solution and 5% alcohol.
- test results are listed in column 7 of Table III .
- the results of the salt spray test are an indicator of the size and distribution of chromium carbide precipitates.
- the test results show that the wetting behavior of the inventive martensitic chromium steels for lubricants is significantly better than that of the comparative steels.
- the good wettability results in less adhesive wear in the presence of lubrication.
- Not only the chemical composition of the steel is of crucial importance.
- a significant influence on the wettability is also exerted by a heat treatment of the samples. This is shown by the larger C values and the smaller R values of the samples according to the invention of experiments 9 to 17.
- the factors K1 and K3 also show that the favorable test results are based on a more favorable precipitation of the carbides and other phases as well as the basic structure.
Abstract
Description
Die Erfindung betrifft einen rostfreien martensitischen Chromstahl und dessen Verwendung.The invention relates to a stainless martensitic chromium steel and its use.
Derartige Stähle sind in großer Zahl bekannt und eignen sich je nach ihrer Zusammensetzung für ein sehr unterschiedliches Verwendungsspektrum.Such steels are known in large numbers and are suitable depending on their composition for a very different range of uses.
So beschreibt beispielsweise die deutsche Patentschrift
Wenig geeignet ist der Werkstoff jedoch für eine Verwendung, deren kennzeichnendes Merkmal ein reibender bzw. gleitender Kontakt Metall/Metall in Anwesenheit eines Schmiermittelfilms ist. Dies gilt insbesondere für Bauteile, die mit Treibstoffen, insbesondere Biokraftstoffen, bei denen es neben anderen Werkstoffeigenschaften sehr wesentlich auf eine gute Schmierfilmbildung bzw. -haftung ankommt, deren Lebensdauer mithin von dem Werkstoffabrieb bei einem Metall/Metall-Reibkontakt wie im Falle von Ventil- und Dosiernadeln sowie Abstreifringen von Kompressoren sehr wesentlich ankommt.However, the material is not very suitable for use, the characteristic feature of which is a sliding contact metal / metal in the presence of a lubricant film. This applies in particular to components that are fuel-based, in particular biofuels, where it is very important in addition to other material properties on a good film formation or adhesion, their life therefore of the material abrasion in a metal / metal frictional contact as in the case Of valve and dispensing needles and scraper rings of compressors is very important.
Ein derartiger, den Reibungsverschleiß herabsetzender Film erfordert jedoch nicht bei allen Verwendungsfällen ein Schmiermittel wie Öl und Fett, d.h. höhere molekulare Kohlenwasserstoffverbindungen, sondern die Teile können auch wie bei Einspritzsystemen oder Ölabstreifringen von dem Betriebsmittel selbst, beispielsweise Treibstoff, geschmiert werden. Entscheidend ist dabei immer das Entstehen eines verschieißhemmenden Films. Die Praxis verwendet jedenfalls eine Reihe von teils teuren, teils ökologisch bedenklichen Zusatzstoffen wie EP-Additive, Detergenzien, HD-Zusätze, Bleiverbindungen und chlorierte Diphenyle zur Beeinflussung, insbesondere zur Stabilisierung und zum Fixieren der verschleißhemmenden Schicht.However, such a frictional wear reducing film does not require a lubricant such as oil and grease in all uses. higher molecular weight hydrocarbon compounds, but the parts may also be lubricated by the resource itself, such as fuel, as in injection systems or oil control rings. The decisive factor is always the emergence of a deterrent film. In any case, the practice uses a number of partly expensive, partly ecologically questionable additives such as EP additives, detergents, HD additives, lead compounds and chlorinated diphenyls for influencing, in particular for stabilizing and fixing the wear-inhibiting layer.
Entscheidend dabei ist insbesondere im Falle von Systemen zum Fördern oder Verdichten von Kraftstoffen oder auch bei Kolbenringen und Abstreifdichtungen sowie Dosier- oder Ventilnadeln auch für chemische oder pharmazeutische Gemische die Stabilität und das Haftvermögen der verschleißmindernden Oberflächenschicht. Eine Stabilisierung ist jedoch in verschiedenen Systemen nicht möglich, so beispielsweise bei neuen, Methanol und Äthanol enthaltenden Kraftstoffen.Crucial here is especially in the case of systems for conveying or compressing fuels or in piston rings and wiper seals and dosing or valve needles for chemical or pharmaceutical mixtures, the stability and the adhesion of the wear-reducing surface layer. However, stabilization is not possible in various systems, such as with new methanol and ethanol containing fuels.
Hinzu kommt, dass der Gesetzgeber in zahlreichen Fällen die Verwendung von Hilfsstoffen und Zusätzen wie beispielsweise bleihaltige Verbindungen aus ökologischen Gründen untersagt.In addition, in many cases, the legislature prohibits the use of excipients and additives such as lead-containing compounds for environmental reasons.
Bei eingehenden Untersuchungen hat sich nun gezeigt, dass der Abrieb bei martensitischen Chromstählen sehr stark von der Benetzbarkeit der Oberfläche beeinflusst wird. So unterliegt ein derartiger Stahl auch bei hoher Festigkeit einem erhöhten Verschleiß, der sich auf eine unzureichende Benetzung der Stahloberfläche durch das Schmier-mittel zurückführen lässt. Unter Krafteinwirkung kann es nämlich zu einem örtlichen Verdrängen des Schmiermittels kommen, diese Gefahr ist insbesondere dort sehr groß, wo mikroskopisch kleine Oberflächenerhebungen unter entsprechend hohem spezifischem Druck stehen. Ursache dafür ist ein Verdrängen der Schmiermittelmoleküle an solchen Erhebungen und Spitzen, die infolge der dynamischen Beanspruchung getrennt und aufgerissen werden. Die Folge eines solchen Adhäsionsverschleißes ist eine erhöhte Rauhigkeit der Metalloberfläche und ein dadurch wiederum erhöhter Verschleiß.Extensive research has shown that the abrasion of martensitic chromium steels is strongly influenced by the wettability of the surface. Such a steel is subject to increased wear even at high strength, which can be attributed to insufficient wetting of the steel surface by the lubricant. Under the action of force, local displacement of the lubricant can occur, and this danger is particularly great where microscopically small surface elevations are under a correspondingly high specific pressure. The reason for this is a displacement of the lubricant molecules at such elevations and tips, which are separated and torn as a result of the dynamic stress. The consequence of such adhesive wear is an increased roughness of the metal surface and thereby in turn increased wear.
Der Erfindung liegt daher das Problem zugrunde, einen rostfreien martensitischen Chromstahl zu finden, der aufgrund seiner chemischen Affinität und starker Adhäsionskräfte eine bessere Benetzbarkeit besitzt und einen stabilen Schmiermittelfilm bildet, der sich weitaus schwieriger stören bzw. verdrängen lässt als bei herkömmlichen Stählen dieser Art und demzufolge einen geringeren Verschleiß bewirkt.The invention is therefore based on the problem of finding a stainless martensitic chromium steel, which has better wettability due to its chemical affinity and strong adhesion forces and forms a stable lubricant film, which is much more difficult to disturb or displace than conventional steels of this type and consequently causes less wear.
Die Lösung dieses Problems besteht in einem martensitischen Chromstahl mit
Vorzugsweise enthält der Stahl jeweils einzeln oder auch nebeneinander
Die Praxis hat sich bisher an Trockenverschleißversuchen, d.h. an den Ergebnissen von schmiermittelfreien Versuchen orientiert. Es hat sich daher gezeigt, dass der Reibverschleiß bei einem Kontakt auch unter Verwendung eines Schmiermittels erheblich war.Practice has hitherto been limited to dry wear tests, i. oriented on the results of lubricant-free tests. It has therefore been found that the frictional wear in a contact was also significant using a lubricant.
Der Kohlenstoff ist Austenitbildner und stabilisiert daher das austenitische Kristallgitter. Gleichzeitig trägt der Kohlenstoff aber auch zusammen mit den Karbidbildnem zu karbidischen Ausscheidungen bei, die eine Steigerung der Härte und Abriebfestigkeit des Stahls bewirken. Um ein grobkörniges und zeilenförmiges Ausscheiden von Chromkabiden zu vermeiden, empfiehlt sich ein Vorabausscheiden anderer Karbide im austenitischen Zustand, die eine bessere Karbidhomogenität bewirken. Der Stahl enthält daher 0,40 bis 0,80%, vorzugsweise 0,55 bis 0,75% Kohlenstoff.The carbon is austenite and therefore stabilizes the austenitic crystal lattice. At the same time, however, carbon, together with the carbide formers, also contributes to carbide precipitations, which increase the hardness and abrasion resistance of the steel. In order to avoid a coarse and line-like separation of chromium carbides, it is recommended to pre-precipitate other carbides in austenitic Condition that cause better carbide homogeneity. The steel therefore contains 0.40 to 0.80%, preferably 0.55 to 0.75% carbon.
Silizium dient als Desoxidationsmittel. Höhere Gehalte können jedoch zum Entstehen intermetallischer Phasen führen. Andererseits ist Silizium jedoch auch ein Ferritbildner. Der Stahl enthält daher 0,2 bis 1,5% Silizium, vorzugsweise bis 0,65% Silizium.Silicon serves as a deoxidizer. However, higher contents can lead to the formation of intermetallic phases. On the other hand, silicon is also a ferrite former. The steel therefore contains 0.2 to 1.5% silicon, preferably up to 0.65% silicon.
Nickel gehört zu den Austenitblidnern, jedoch bringt der Austenitanteil im Gefüge die Gefahr einer Verschlechterung der Verschleißeigenschaften mit sich. Andererseits ist das Nickel jedoch auch als Bestandteil des Kristallgitters mit seinem Einfluss auf das c/a-Verhältnis des Martensits von Vorteil. Der Stahl enthält daher 0,15 bis 1% Nickel.Nickel belongs to the austenite blidners, but the austenite part in the microstructure carries the risk of a deterioration of the wear properties. On the other hand, nickel is also an advantage as part of the crystal lattice with its influence on the c / a ratio of the martensite. The steel therefore contains 0.15 to 1% nickel.
Mangan stabilisiert den Austenit und verschiebt vorteilhafterweise die Martensitbildung zu tieferen Temperaturen. Der Höchstgehalt an Mangan beträgt daher 1%, jedoch sollte ein Mindestgehalt von 0,30% nicht unterschritten werden, weil Mangan gleichzeitig auch das c/a-Verhältnis des frisch entstandenen Martensits verändert und im austenitischen Bereich das Ausscheidungsverhalten der Feinausscheidungen vorteilhaft beeinflusst.Manganese stabilizes the austenite and advantageously shifts the martensite formation to lower temperatures. The maximum level of manganese is therefore 1%, but a minimum content of 0.30% should not be undercut, because manganese simultaneously alters the c / a ratio of the newly formed martensite and advantageously influences the precipitation behavior of the fine precipitates in the austenitic region.
Der Schwefelgehalt ist auf höchstens 0,035% begrenzt, da bei höheren Schwefelgehalten störende sulfidische Ausscheidungen entstehen können.The sulfur content is limited to a maximum of 0.035%, since at higher sulfur levels, troublesome sulfidic precipitates may occur.
Chrom ist erforderlich, um die Korrosionsbeständigkeit des Stahls in Kombination mit dessen Molybdängehalt von 1,25 bis 1,50% zu gewährleisten, insbesondere eine hinreichende Beständigkeit gegen Lochfraßkorrosion. Der Chromgehalt beträgt daher mindestens 16%. Im Hinblick auf die ferritisierende Wirkung des Chroms ist dessen Gehalt jedoch auf 18% begrenzt. Die synergistische Wirkung von Chrom und Molybdän im Hinblick auf die Lochfraßbeständigkeit ist insbesondere dann gewährleistet, wenn die Gehalte an Chrom, Molybdän und Wolfram der folgenden Gleichung genügen:
Der Stahl enthält 0,001 bis 0,8% Wolfram, vorzugsweise mindestens 0,001% Wolfram, das zusammen mit dem Eisen und Molybdän Mischkarbide bildet, die zur Warmfestigkeit des Stahls erheblich beitragen und bei einem Anlassglühen zum Entstehen von Sekundärausscheidungen in Form höherer Karbide führen.The steel contains from 0.001 to 0.8% tungsten, preferably at least 0.001% tungsten, which together with the iron and molybdenum forms mixed carbides, which contribute significantly to the hot strength of the steel and lead to secondary carbide secondary precipitation during tempering.
Der Stickstoff bildet zusammen mit dem Kohlenstoff Karbonitride, verschlechtert jedoch auch die Benetzbarkeit des Stahls für Kohlenwasserstoff-Schmiermittel; die obere Gehaltsgrenze für Stickstoff beträgt daher 0,08%.The nitrogen together with the carbon forms carbonitrides, but also worsens the wettability of the steel to hydrocarbon lubricants; the upper content limit for nitrogen is therefore 0.08%.
Der Stahl enthält Vanadium, Niob und Titan als Karbidbildner mit dem Vorteil, dass diese in Anbetracht der hohen Affinität zum Kohlenstoff bereits bei sehr hohen Temperaturen Kristallisationskeime für das Entstehen von Chromkarbiden bilden. Der Vanadiumgehalt beträgt daher 0,15 bis 0,20% bei Titan- und Niobgehalten von jeweils bis 0,05%. Besonders vorteilhaft ist dabei die folgende Wirksumme der Karbidbildner:
Der Stahl enthält des weiteren 0,001 bis 0,03% Aluminium als Desoxidationsmittel, jedoch nicht mehr, weil höhere Aluminiumgehalte versprödend wirken.The steel also contains 0.001 to 0.03% aluminum as a deoxidizer, but not more, because higher aluminum contents have an embrittling effect.
Der Kupfergehalt beträgt maximal 0,5% und führt insbesondere bei einem Anlassglühen zu feinstkörnigen Sekundärausscheidungen, die zusammen mit anderen Ausscheidungen die Benetzbarkeit des Stahls für Öle bzw. Kohlenwasserstoffe verbessem.The maximum copper content is 0.5% and, especially during tempering, leads to fine-grained secondary precipitations, which together with other precipitates improve the wettability of the steel for oils or hydrocarbons.
Kobalt schließlich fördert das Entstehen von ε-Karbiden und anderen Feinausscheidungen; es verbessert auf diese Weise die Warmfestigkeit des Stahls. Aus Kostengründen sollte jedoch ein Kobaltgehalt von 0,5% nicht überschritten werden.Finally, cobalt promotes the formation of ε-carbides and other fine precipitates; it improves the hot strength of the steel in this way. For cost reasons, however, a cobalt content of 0.5% should not be exceeded.
Da sich die für die Werkstoffeigenschaften entscheidenden Chromkarbide beim Abkühlen unter dem Einfluss von Kristallisationskeimen zu 90% im Temperaturbereich von 1100 bis 900 °C bilden, sollte die Abkühlungsgeschwindigkeit hier 50 °C/sec nicht übersteigen (Wärmebehandlung A), um die Karbidbildung nicht zu beeinträchtigen und zu verhindern, dass ein Teil der Reaktionspartner beim Entstehen der Karbide metastabil auf Zwischengitterplätzen verbleibt. Bei der weiteren Abkühlung bildet sich unterhalb der MS-Temperatur spontan aus dem kubisch raumzentrierten Kristallgitter Martensit mit der Folge, dass die zuvor entstandenen Karbide im Martensit eingelagert werden, der Spannungszustand der Matrix jedoch umso geringer ist, je feiner verteilt die Karbide vorliegen. Dieser Zustand fördert sehr wesentlich die Schmiermittel- bzw. Ölbenetzung der Oberfläche des Stahls.Since the decisive for the material properties of chromium carbides on cooling under the influence of crystallization nuclei form 90% in the temperature range of 1100 to 900 ° C, the cooling rate should not exceed 50 ° C / sec (heat treatment A), so as not to affect the carbide formation and to prevent part of the reactants from remaining metastable at interstitial sites when the carbides are formed. Upon further cooling, martensite spontaneously forms below the MS temperature from the cubic body-centered crystal lattice, with the result that the previously formed carbides are incorporated in the martensite, but the stress state of the matrix is lower, the more finely distributed the carbides are. This condition greatly enhances the lubricant or oil wetting of the surface of the steel.
Der Martensit aus der Austenit-Umwandlung besitzt ein tetragonal verzerrtes Kristallgitter mit einem Verhältnis der Kristallachsen a/c über 1. Dabei lässt sich der bei der Umwandlung entstandene Martensit durch eine der Martensitbidlung folgende Wärmebehandlung im Temperaturbereich unter 550 °C dahingehend beeinflussen, dass sich das Kristallachsenverhältnis s/c verringert, was sich außerordentlich vorteilhaft auf die Werkstoffeigenschaften auswirkt. Dieser Vorteil ergibt sich insbesondere, wenn der Gesamtgehalt an Niob, Titan und Vanadium folgender Bedingung genügt:
Unterhalb des Werts von 0,15 ist die Keimbildung weniger günstig, so dass das Chromkarbid einer Kristallisationshemmung unterliegen kann und später auskristallisiert. Damit verbunden ist ein gröberkörniges Chromkarbid mit einer ungünstigen Verteilung in der Matrix. Beim Überschreiten des oberen Grenzwerts können anstelle feiner Primärkarbide grobe MC-Karbide des Niobs, Titans und des Vanadiums entstehen. Damit geht die Wirkung der vorerwähnten Elemente als karbidische Kristallisationskeime für die Chromkarbide verloren.Below the value of 0.15, the nucleation is less favorable, so that the chromium carbide can be subject to crystallization inhibition and crystallized later. Associated with this is a coarser grained chromium carbide with an unfavorable distribution in the matrix. When the upper limit is exceeded, coarse MC carbides of niobium, titanium and vanadium may be formed instead of fine primary carbides. Thus, the effect of the aforementioned elements is lost as carbide crystallization nuclei for the chromium carbides.
Die Wärmebehandlung B besteht dabei in einem Anlaßglühen bei 100 bis 550 °C, vorzugsweise bei mindestens 200 °C statt und führt zum Entstehen von Feinausscheidungen in dem bei der vorausgegangenen Wärmebehandlung A im Temperaturbereich von 1100 bis 900 °C stabilisierten Martensit. In Lösung befindliche Atome wie die des Kupfers und des Kobalts sowie die karbidbildenden Elemente spielen hierbei eine wesentliche Rolle, da sie in die Feinausscheidungen eingehen, die insbesondere mit der gemäß
Von weiterem Vorteil ist die Einhaltung der Bedingung
Die Erfindung wird nachfolgend anhand von Ausführungsbeispielen und der Zeichnungen des näheren erläutert. In der Zeichnung zeigen:
- Fig. 1
- in schematischer Darstellung einen Öltropfen auf einer Stahloberfläche,
- Fig. 2
- eine Vorrichtung zum Bestimmen des Abriebverschleißes in schematischer Darstellung und
- Fig. 3
- die Spurbreite R einer Kugelschleifbahn nach
Fig. 2 als Maß für die Verschleißfestigkeit.
- Fig. 1
- a schematic representation of an oil drop on a steel surface,
- Fig. 2
- a device for determining the abrasion wear in a schematic representation and
- Fig. 3
- the track width R of a ball track after
Fig. 2 as a measure of wear resistance.
Aus der nachfolgenden Tabelle I sind die Analysen von fünf herkömmlichen Vergleichsstählen V1 bis V5 und dreier unter die Erfindung fallender Stähle E1 bis E3 ersichtlich.
Die nachfolgende Tabelle II gibt die sich aus den Analysen ergebenden Summenwerte für K1 bis K3 wieder.
Acht den Stand der Technik betreffende Proben 1 bis 8 und neun unter die Erfindung fallende Proben 9 bis 17 mit den sich aus Tabelle I ergebenden Zusammensetzungen wurden in einem Mittelfrequenzofen unter Schutzgas erschmolzen und in einer Kokille zu Probestäben vergossen sowie 30 min. bei 1200 °C ausgelagert. Danach wurden die Proben zu Stäben geschmiedet, von ihrer Zunderschicht befreit und mittels Hartmetallschneidplatten zu zylindrischen Probestäben abgedreht. Die Probestäbe besaßen einen Durchmesser von 15 mm und wurden verschiedenen Austenitisierungstemperaturen (A) und Anlaßtemperaturen (B) unterworfen, um schließlich die Qualität einer Ölbenetzung und den Abriebverschleiß zu bestimmen.Eight of the prior art samples 1 to 8 and nine falling under the invention samples 9 to 17 with the compositions resulting from Table I were melted in a medium frequency oven under inert gas and poured into test tubes in a mold and 30 min. outsourced at 1200 ° C. Thereafter, the samples were forged into rods, freed of their scale layer and turned off by means of carbide cutting plates to cylindrical test bars. The test bars had a diameter of 15 mm and had different austenitizing temperatures (A) and tempering temperatures (B) to finally determine the quality of oil wetting and abrasion wear.
Das in der Tabelle III mit A gekennzeichnete Austenitisierungsglühen fand bei 1020 °C oder 1050 °C statt, gefolgt von einem Schnellabkühlen mit einer Abkühlungsgeschwindigkeit von mindestens 50 °C/sec auf 800 °C und einem sich anschließenden Abkühlen innerhalb von 5 min. auf 300 °C sowie einem langsamen Abkühlen auf Raumtemperatur.Austenitizing annealing indicated by A in Table III took place at 1020 ° C or 1050 ° C, followed by rapid cooling at a cooling rate of at least 50 ° C / sec to 800 ° C followed by cooling within 5 min. to 300 ° C and a slow cooling to room temperature.
Die Proben wurden zudem abschließend entsprechend der Versuchsreihe B auf eine Temperatur von 100 bis 530 °C erwärmt und mit einer Geschwindigkeit von 100 °C/h auf Raumtemperatur abgekühlt.The samples were finally heated according to the test series B to a temperature of 100 to 530 ° C and cooled at a rate of 100 ° C / h to room temperature.
Zur Bestimmung der Benetzbarkeit wurden die Proben sodann geschliffen und poliert, in einem wässrigen Ultraschallbad bei 50 °C gereinigt, mit heißem destillierten Wasser unter Einwirkung von Ultraschall weitere 20 min. von Reinigungsmittelresten befreit und danach getrocknet. Zur Bestimmung der Benetzbarkeitskennzahl B wurden sodann mithilfe einer Mikrodosierung auf jede Probe 10 µl Parafinöl aufgetragen und der sich sodann bildende Öltropfen hinsichtlich seiner Breite B vermessen, wie sich das schematisch aus der Darstellung in
Die Abrieb- bzw. Verschleißfestigkeit wurde mit Hilfe eines modifizierten "Pin on Disk-Versuchs* bestimmt. Dabei wurden die zylindrischen Proben zunächst plangeschliffen, danach gereinigt, in eine Halterung eingespannt und sodann entsprechend
Die Ergebnisse sind eindeutig; die unter die Erfindung fallenden Proben 9 bis 17 besitzen eine deutlich bessere Verschleißfestigkeit als die Proben 1 bis 8 aus herkömmlichen Stählen. Die Bedeutung des K2-Wertes für eine weitere Verbesserung der Verschleißfestigkeit ergibt sich dabei aus einem Vergleich der Daten aus der nachfolgenden Tabelle IV mit den entsprechenden Daten der Spalte 6 von Tabelle III.
Da viele kleine Chromkarbidausscheidungen hinsichtlich der Korrosionsbeständigkeit wirkungsvoller sind als wenige grobe Ausscheidungen, kann das Ergebnis eines Salzsprühtests als Indikator für die Größe und die Verteilung der Chromkarbidausscheidungen dienen. Die Proben wurden daher 120 Stunden einem Korrosionsversuch im Rahmen eines modifizierten Salzsprühtests mit einer 3%-NaCl-Lösung und 5% Alkohol unterworfen.Since many small chromium carbide precipitates are more effective in corrosion resistance than a few coarse precipitates, the result of a salt spray test can serve as an indicator of the size and distribution of the chromium carbide precipitates. The samples were therefore subjected to 120 hours of corrosion testing as part of a modified salt spray test with a 3% NaCl solution and 5% alcohol.
Die Versuchsergebnisse sind in Spalte 7 der Tabelle III zusammengestellt.The test results are listed in column 7 of Table III .
Es ist allgemein bekannt, dass martensitische Chromstähle je nach der Größe und Dichte der Chromkarbidausscheidungen in chloridische Lösungen einer starken Lochfraßkorrosion unterliegen. Die Daten der Spalte 7 der Tabelle III bestätigen das für die herkömmlichen Vergleichsstähle 1 bis 8 im Vergleich mit den unter die Erfindung fallenden Stählen 9 bis 17.It is well known that martensitic chromium steels are subject to severe pitting corrosion, depending on the size and density of the chromium carbide precipitates in chloride solutions. The data of column 7 of Table III confirm that for the conventional comparative steels 1 to 8 in comparison with the steels 9 to 17 according to the invention.
Je nach Breite der Zone einer Chromverarmung kommt es zum Entstehen von Lochfraßkorrosion, woraus sich die Erkenntnis gibt, dass eine Vielzahl kleiner Chromkarbidausscheidungen hinsichtlich der Lochfraßkorrosion günstiger ist als eine geringere Anzahl großer Ausscheidungen. Insofern eignen sich die Ergebnisse des Salzsprühtest als Indikator für die Größe und die Verteilung der Chromkarbidausscheidungen.Depending on the width of the zone of a chromium depletion, pitting corrosion occurs, which leads to the finding that a large number of small chromium carbide precipitations is more favorable in terms of pitting corrosion than a smaller number of large precipitates. In this respect, the results of the salt spray test are an indicator of the size and distribution of chromium carbide precipitates.
Die Ergebnisse der Korrosionsversuche gemäß Tabelle III letzte Spalte wurden nach einer Güteskala von 0 bis 5 bewertet, wobei 0 für keinerlei Roststellen steht und 5 mindestens fünf Roststellen indiziert. Die Ergebnisse des Salzsprühtests sind in Tabelle III, letzte Spalte zusammengestellt.The results of the corrosion tests according to Table III last column were evaluated according to a quality scale of 0 to 5, where 0 stands for no rusting and 5 indicates at least five rust spots. The results of the salt spray test are summarized in Table III , last column.
Insgesamt zeigen die Versuchsergebnisse, dass sich das Benetzungsverhalten der erfindungsgemäßen martensitischen Chromstähle für Schmiermittel deutlich besser ist als das der Vergleichsstähle. Die gute Benetzbarkeit führt zu einem geringeren Adhäsionsverschleiß in Anwesenheit einer Schmierung. Dabei ist nicht nur die chemische Zusammensetzung des Stahls von entscheidender Bedeutung. Einen wesentlichen Einfluss auf die Benetzbarkeit übt auch eine Wärmebehandlung der Proben aus. Dies zeigt sich an den größeren C-Werten und den kleineren R-Werten der erfindungsgemäßen Proben der Versuche 9 bis 17.Overall, the test results show that the wetting behavior of the inventive martensitic chromium steels for lubricants is significantly better than that of the comparative steels. The good wettability results in less adhesive wear in the presence of lubrication. Not only the chemical composition of the steel is of crucial importance. A significant influence on the wettability is also exerted by a heat treatment of the samples. This is shown by the larger C values and the smaller R values of the samples according to the invention of experiments 9 to 17.
Maßgebend für die bessere Verschleißbeständigkeit R der erfindungsgemäßen Proben dürfte in erster Linie die Zusammensetzung des Stahls sein, wohin zu die zweistufige Wärmebehandlung zur Beeinflussung der Ausscheidungen kommt Insoweit bedingt nicht nur die spezielle Zusammensetzung des Stahls, sondern auch dessen Ausscheidungen im Gefüge die Materialeigenschaften. Dabei ist zu beachten, dass sich die Benetzbarkeit des Stahls für ein Schmiermittel besonders durch die Wärmebehandlungen A und B verbessern lässt. Das lässt den Schluss zu, dass die kohlenstoffhaltigen Feinausscheidungen im Martensit für die Schmiermittelbenetzung günstiger sind als stickstoffmodifizierte Karbonitride und eine stickstoffhaltige Matrix. Maßgebend ist somit der niedrige Stickstoffgehalt der Legierung ebenso wie der Faktor K2. Das belegen insbesondere die Vergleichsstähle V3 mit 0,25% Stickstoff und auch V5 mit 0,22% Stickstoff im Gegensatz zu den unter die Erfindung fallenden Stähle E1 bis E3 mit nur 0,05 bis 0,07% Stickstoff.Decisive for the better wear resistance R of the samples according to the invention should be primarily the composition of the steel, where the two-stage heat treatment for influencing the precipitates is due insofar not only the special composition of the steel, but also its precipitates in the structure the material properties. It should be noted that the wettability of the steel for a lubricant can be improved especially by the heat treatments A and B. This suggests that the carbonaceous fine precipitates in martensite are more favorable for lubricant wetting than nitrogen-modified carbonitrides and a nitrogen-containing matrix. The decisive factor is thus the low nitrogen content of the alloy as well as the factor K2. This is demonstrated in particular by the comparison steels V3 with 0.25% nitrogen and also V5 with 0.22% nitrogen, in contrast to the steels E1 to E3 covered by the invention with only 0.05 to 0.07% nitrogen.
Schließlich zeigen auch die Faktoren K1 und K3, dass die günstigen Versuchsergebnisse auf einer günstigeren Ausscheidung der Karbide und anderer Phasen sowie dem Grundgefüge basieren.Finally, the factors K1 and K3 also show that the favorable test results are based on a more favorable precipitation of the carbides and other phases as well as the basic structure.
Claims (13)
- Stainless martensitic chromium steel with:
0.4 to 0.8% carbon 0.2 to 1.5% silicon 0.15 to 1.0% nickel 0.3 to 1.00% manganese 0.015 to 0.035% sulphur 16 to 18% chromium 1.25 to 1.50% molybdenum up to 0.8% tungsten 0.04 to 0.08% nitrogen 0.15 to 0.20% vanadium up to 0.05% titanium up to 0.05% niobium 0.001 to 0.03% aluminium 0.02 to 0.5% copper up to 0.5% cobalt up to 0.040% boron, - Chromium steel according to claim 1, which contains individually or together:
0.55 to 0.75% carbon up to 0.65% silicon up to 0.8% manganese at least 0.001 % tungsten - Chromium steel according to any one of claims 1 to 6, characterised in that it is annealed austenitising at 1020 to 1050°C, then cooled rapidly with a cooling rate of at least 50°C/sec to 800°C, and then subjected to a five-minute cooling to 300°C with subsequent air cooling to room temperature.
- Chromium steel according to claim 7 which is reheated to 100 to 530°C and then cooled slowly to room temperature at a rate of 100°C/h.
- Use of a chromium steel according to any one of claims 1 to 8 as a pitting-resistant material.
- Use of a chromium steel according to any one of claims 1 to 8 as a material resistant to abrasion.
- Use according to claim 9 or 10 with a lubricant film.
- Use according to claim 11 in a metal/metal slip contact with a lubricant intermediate layer.
- Use of a chromium steel according to any one of claims 1 to 7 as a material for the production of valve stems, control and metering needles, guide sleeves, function components of fuel injection systems, piston rings for plungers and engines, and sealing and scraping rings for compressors.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PL10751807T PL2470681T3 (en) | 2009-08-24 | 2010-08-18 | Stainless steel martensitic steel |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102009038382A DE102009038382A1 (en) | 2009-08-24 | 2009-08-24 | Stainless martensitic chrome steel |
| PCT/EP2010/005067 WO2011023326A1 (en) | 2009-08-24 | 2010-08-18 | Stainless martensitic chromium steel |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2470681A1 EP2470681A1 (en) | 2012-07-04 |
| EP2470681B1 true EP2470681B1 (en) | 2013-11-20 |
Family
ID=42937637
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP10751807.8A Active EP2470681B1 (en) | 2009-08-24 | 2010-08-18 | Stainless steel martensitic steel |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP2470681B1 (en) |
| DE (1) | DE102009038382A1 (en) |
| ES (1) | ES2446716T3 (en) |
| PL (1) | PL2470681T3 (en) |
| WO (1) | WO2011023326A1 (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102011102293A1 (en) | 2011-02-24 | 2012-08-30 | Stahlwerk Ergste Westig Gmbh | Use of a chromium steel with a martensitic structure and carbide inclusions |
| DE102017003965B4 (en) * | 2017-04-25 | 2019-12-12 | Zapp Precision Metals Gmbh | Martensitic chrome steel, steel foil, perforated and / or perforated steel foil component, process for producing a steel foil |
| CN115109891B (en) * | 2022-07-14 | 2022-12-20 | 中北大学 | High-carbon high-chromium nitrogen-containing martensitic stainless steel and carbide refining method thereof |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SE404029B (en) * | 1968-05-31 | 1978-09-18 | Uddeholms Ab | PROCEDURE FOR THE PREPARATION OF THE SUBSTANCE FOR HERDAT STEEL THIN EGG TOOLS |
| US3990892A (en) * | 1972-03-28 | 1976-11-09 | Kabushiki Kaisha Fujikoshi | Wear resistant and heat resistant alloy steels |
| AT401387B (en) * | 1994-06-29 | 1996-08-26 | Boehler Ybbstalwerke | CORROSION RESISTANT ALLOY AND METHOD FOR PRODUCING CORROSION RESISTANT CUTTERS |
| DE19808276C2 (en) * | 1998-02-27 | 2003-12-24 | Stahlwerk Ergste Westig Gmbh | Steel alloy for sliding elements |
| JP2000337389A (en) * | 1999-03-19 | 2000-12-05 | Nsk Ltd | Rolling bearing |
| DE10027049B4 (en) | 2000-01-17 | 2004-09-16 | Stahlwerk Ergste Westig Gmbh | Use a chrome steel alloy |
| DE102004051629B4 (en) * | 2004-10-23 | 2006-08-24 | Stahlwerk Ergste Westig Gmbh | Stainless martensitic chrome steel |
| JP2008291307A (en) * | 2007-05-24 | 2008-12-04 | Daido Steel Co Ltd | Method for manufacturing die, method for manufacturing steel for die, and method for manufacturing die using steel for die |
| DE102007025758A1 (en) * | 2007-06-01 | 2008-12-04 | Mahle International Gmbh | seal |
-
2009
- 2009-08-24 DE DE102009038382A patent/DE102009038382A1/en not_active Withdrawn
-
2010
- 2010-08-18 ES ES10751807.8T patent/ES2446716T3/en active Active
- 2010-08-18 PL PL10751807T patent/PL2470681T3/en unknown
- 2010-08-18 EP EP10751807.8A patent/EP2470681B1/en active Active
- 2010-08-18 WO PCT/EP2010/005067 patent/WO2011023326A1/en active Application Filing
Also Published As
| Publication number | Publication date |
|---|---|
| WO2011023326A1 (en) | 2011-03-03 |
| PL2470681T3 (en) | 2014-05-30 |
| DE102009038382A1 (en) | 2011-03-03 |
| EP2470681A1 (en) | 2012-07-04 |
| ES2446716T3 (en) | 2014-03-10 |
| DE102009038382A8 (en) | 2011-06-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AT502397B1 (en) | Alloy used for preparing bearing component and bearing of roller for aircraft, contains carbon, manganese, silicon, chromium, molybdenum, vanadium, tungsten, niobium, tantalum, nickel, cobalt, aluminum, nitrogen and iron | |
| DE60214456T2 (en) | Martensitic stainless steel with high hardness and good corrosion resistance | |
| DE69604341T3 (en) | Martensitic. STAINLESS STEEL WITH GOOD RESISTANCE TO HOLE FRICTION CORROSION AND HIGH HARDENING | |
| DE60215655T2 (en) | MARTENSITIC STAINLESS STEEL | |
| EP3228724A1 (en) | Method for setting the thermal conductivity of a steel, tool steel, in particular hot-work steel, and steel object | |
| DE2040308A1 (en) | Austenitic stainless steel | |
| DE112019006482T5 (en) | CARBONITRATED BEARING COMPONENT | |
| EP1233080B1 (en) | Steel for plastic molds and process of heat treatment thereof | |
| AT393642B (en) | USE OF AN IRON BASED ALLOY FOR THE POWDER METALLURGICAL PRODUCTION OF PARTS WITH HIGH CORROSION RESISTANCE, HIGH WEAR RESISTANCE AND HIGH TENSITY AND PRESSURE STRENGTH, ESPECIALLY FOR THE PROCESS | |
| EP2470681B1 (en) | Stainless steel martensitic steel | |
| DE2447137B2 (en) | STEEL ALLOY RESISTANT AGAINST PITCH CORROSION | |
| DE60030175T2 (en) | cutting alloy | |
| DE112019006504T5 (en) | STEEL MATERIAL AS BASE MATERIAL FOR CARBONITRATED BEARING COMPONENTS | |
| AT410550B (en) | Material used as a tool material in the glass industry, especially as a molding material for machine pressed glass consists of an alloy containing carbon, silicon, chromium, nickel and nitrogen | |
| DE112008003146T5 (en) | Induction hardening steel having excellent cold workability, rolling member formed of such a steel, and motion guide apparatus using such a rolling member | |
| DE60011115T2 (en) | STEEL MATERIAL, ITS USE AND MANUFACTURE | |
| EP3061838B1 (en) | Blank bainite long product and method for producing the same | |
| DE69909940T2 (en) | Martensitic stainless steel parts and process for their manufacture | |
| EP4211279A1 (en) | Hot-rolled flat steel product and method for producing a hot-rolled flat steel product | |
| EP3781719B1 (en) | Copper-zinc-nickel-manganese alloy | |
| EP1471160B1 (en) | Cold-worked Steel Object | |
| DE60100730T2 (en) | AUSTENITIC STEEL | |
| EP1445339B1 (en) | Alloy and article with high heat resistance and high heat stability | |
| EP2453027B1 (en) | Thermoformed product and method for producing same | |
| EP1052304A1 (en) | Martensitic corrosion resistant chromium steel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20120320 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| DAX | Request for extension of the european patent (deleted) | ||
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20130918 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 641717 Country of ref document: AT Kind code of ref document: T Effective date: 20131215 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: E. BLUM AND CO. AG PATENT- UND MARKENANWAELTE , CH |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 502010005436 Country of ref document: DE Effective date: 20140116 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2446716 Country of ref document: ES Kind code of ref document: T3 Effective date: 20140310 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: PC2A Owner name: ZAPP PRECISION METALS GMBH Effective date: 20140409 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140220 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140320 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| RAP2 | Party data changed (patent owner data changed or rights of a patent transferred) |
Owner name: ZAPP PRECISION METALS GMBH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: ZAPP PRECISION METALS GMBH, DE Free format text: FORMER OWNER: STAHLWERK ERGSTE WESTIG GMBH, DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: PL Ref legal event code: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140320 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 502010005436 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| 26N | No opposition filed |
Effective date: 20140821 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 502010005436 Country of ref document: DE Representative=s name: KOENIG SZYNKA TILMANN VON RENESSE PATENTANWAEL, DE |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 502010005436 Country of ref document: DE Effective date: 20140821 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 502010005436 Country of ref document: DE Owner name: ZAPP PRECISION METALS GMBH, DE Free format text: FORMER OWNER: STAHLWERK ERGSTE WESTIG GMBH, 58239 SCHWERTE, DE Effective date: 20141104 Ref country code: DE Ref legal event code: R082 Ref document number: 502010005436 Country of ref document: DE Representative=s name: KOENIG SZYNKA TILMANN VON RENESSE PATENTANWAEL, DE Effective date: 20141104 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: LU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140818 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20140818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140831 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20140221 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20100818 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 7 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 641717 Country of ref document: AT Kind code of ref document: T Effective date: 20150818 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150818 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131120 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230513 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: TR Payment date: 20230811 Year of fee payment: 14 Ref country code: IT Payment date: 20230831 Year of fee payment: 14 Ref country code: ES Payment date: 20230918 Year of fee payment: 14 Ref country code: CH Payment date: 20230902 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PL Payment date: 20230804 Year of fee payment: 14 Ref country code: FR Payment date: 20230821 Year of fee payment: 14 Ref country code: DE Payment date: 20230919 Year of fee payment: 14 |