EP1887362A1 - Vorrichtung, verfahren, system, programm und aufzeichnungsmedium zur einstufung einer leberkrankheit - Google Patents
Vorrichtung, verfahren, system, programm und aufzeichnungsmedium zur einstufung einer leberkrankheit Download PDFInfo
- Publication number
- EP1887362A1 EP1887362A1 EP06746698A EP06746698A EP1887362A1 EP 1887362 A1 EP1887362 A1 EP 1887362A1 EP 06746698 A EP06746698 A EP 06746698A EP 06746698 A EP06746698 A EP 06746698A EP 1887362 A1 EP1887362 A1 EP 1887362A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- index
- hepatic
- fractional expression
- evaluating
- hepatic disease
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 208000019423 liver disease Diseases 0.000 title claims abstract description 227
- 238000000034 method Methods 0.000 title claims abstract description 37
- 230000014509 gene expression Effects 0.000 claims abstract description 152
- 150000001413 amino acids Chemical class 0.000 claims abstract description 106
- 206010019668 Hepatic fibrosis Diseases 0.000 claims abstract description 73
- 201000010099 disease Diseases 0.000 claims abstract description 65
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 65
- 238000011156 evaluation Methods 0.000 claims abstract description 54
- 238000004891 communication Methods 0.000 claims description 55
- 244000137850 Marrubium vulgare Species 0.000 claims description 43
- 208000006454 hepatitis Diseases 0.000 claims description 35
- 208000019425 cirrhosis of liver Diseases 0.000 claims description 27
- 231100000283 hepatitis Toxicity 0.000 claims description 21
- 206010008909 Chronic Hepatitis Diseases 0.000 claims description 14
- 102000014150 Interferons Human genes 0.000 abstract description 14
- 108010050904 Interferons Proteins 0.000 abstract description 14
- IWUCXVSUMQZMFG-AFCXAGJDSA-N Ribavirin Chemical compound N1=C(C(=O)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 IWUCXVSUMQZMFG-AFCXAGJDSA-N 0.000 abstract description 14
- 229940079322 interferon Drugs 0.000 abstract description 14
- 229960000329 ribavirin Drugs 0.000 abstract description 14
- HZCAHMRRMINHDJ-DBRKOABJSA-N ribavirin Natural products O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1N=CN=C1 HZCAHMRRMINHDJ-DBRKOABJSA-N 0.000 abstract description 14
- 229940024606 amino acid Drugs 0.000 description 88
- 235000001014 amino acid Nutrition 0.000 description 88
- 238000012545 processing Methods 0.000 description 49
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 30
- 230000001276 controlling effect Effects 0.000 description 20
- 230000000750 progressive effect Effects 0.000 description 17
- 210000004369 blood Anatomy 0.000 description 16
- 239000008280 blood Substances 0.000 description 16
- 238000003745 diagnosis Methods 0.000 description 16
- 230000005540 biological transmission Effects 0.000 description 12
- 238000010586 diagram Methods 0.000 description 12
- 208000005176 Hepatitis C Diseases 0.000 description 11
- 206010016654 Fibrosis Diseases 0.000 description 10
- 230000006870 function Effects 0.000 description 10
- 230000002440 hepatic effect Effects 0.000 description 10
- 230000035945 sensitivity Effects 0.000 description 9
- 238000004364 calculation method Methods 0.000 description 8
- 230000000875 corresponding effect Effects 0.000 description 8
- 230000004761 fibrosis Effects 0.000 description 8
- 241000711549 Hepacivirus C Species 0.000 description 7
- 238000001574 biopsy Methods 0.000 description 7
- 208000015181 infectious disease Diseases 0.000 description 7
- 230000002596 correlated effect Effects 0.000 description 5
- 230000010365 information processing Effects 0.000 description 5
- 210000002381 plasma Anatomy 0.000 description 5
- 208000006154 Chronic hepatitis C Diseases 0.000 description 4
- 210000001772 blood platelet Anatomy 0.000 description 4
- 238000004590 computer program Methods 0.000 description 4
- 230000002950 deficient Effects 0.000 description 4
- 238000002405 diagnostic procedure Methods 0.000 description 4
- 208000007386 hepatic encephalopathy Diseases 0.000 description 4
- 208000010710 hepatitis C virus infection Diseases 0.000 description 4
- 208000024891 symptom Diseases 0.000 description 4
- 238000012546 transfer Methods 0.000 description 4
- 238000004458 analytical method Methods 0.000 description 3
- 239000000306 component Substances 0.000 description 3
- 210000004185 liver Anatomy 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- 230000008520 organization Effects 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 2
- 108010088751 Albumins Proteins 0.000 description 2
- 102000009027 Albumins Human genes 0.000 description 2
- 108010044091 Globulins Proteins 0.000 description 2
- 102000006395 Globulins Human genes 0.000 description 2
- 206010061218 Inflammation Diseases 0.000 description 2
- 206010023126 Jaundice Diseases 0.000 description 2
- 108090000340 Transaminases Proteins 0.000 description 2
- 102000003929 Transaminases Human genes 0.000 description 2
- 230000000740 bleeding effect Effects 0.000 description 2
- 239000012503 blood component Substances 0.000 description 2
- 238000009534 blood test Methods 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 208000020403 chronic hepatitis C virus infection Diseases 0.000 description 2
- 230000007882 cirrhosis Effects 0.000 description 2
- 238000003759 clinical diagnosis Methods 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 210000003494 hepatocyte Anatomy 0.000 description 2
- 229920002674 hyaluronan Polymers 0.000 description 2
- 229960003160 hyaluronic acid Drugs 0.000 description 2
- 230000004054 inflammatory process Effects 0.000 description 2
- 238000002357 laparoscopic surgery Methods 0.000 description 2
- 230000017074 necrotic cell death Effects 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 238000013077 scoring method Methods 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- WXHLLJAMBQLULT-UHFFFAOYSA-N 2-[[6-[4-(2-hydroxyethyl)piperazin-1-yl]-2-methylpyrimidin-4-yl]amino]-n-(2-methyl-6-sulfanylphenyl)-1,3-thiazole-5-carboxamide;hydrate Chemical compound O.C=1C(N2CCN(CCO)CC2)=NC(C)=NC=1NC(S1)=NC=C1C(=O)NC1=C(C)C=CC=C1S WXHLLJAMBQLULT-UHFFFAOYSA-N 0.000 description 1
- 102100036475 Alanine aminotransferase 1 Human genes 0.000 description 1
- 108010082126 Alanine transaminase Proteins 0.000 description 1
- 108010003415 Aspartate Aminotransferases Proteins 0.000 description 1
- 102000004625 Aspartate Aminotransferases Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 1
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 1
- 208000004930 Fatty Liver Diseases 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 206010019708 Hepatic steatosis Diseases 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 108010026552 Proteome Proteins 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 206010047700 Vomiting Diseases 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 208000022531 anorexia Diseases 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 150000005693 branched-chain amino acids Chemical class 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 210000002808 connective tissue Anatomy 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 206010061428 decreased appetite Diseases 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 210000002744 extracellular matrix Anatomy 0.000 description 1
- 208000010706 fatty liver disease Diseases 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 208000027700 hepatic dysfunction Diseases 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 206010025482 malaise Diseases 0.000 description 1
- 230000003340 mental effect Effects 0.000 description 1
- 238000010295 mobile communication Methods 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- FEMOMIGRRWSMCU-UHFFFAOYSA-N ninhydrin Chemical compound C1=CC=C2C(=O)C(O)(O)C(=O)C2=C1 FEMOMIGRRWSMCU-UHFFFAOYSA-N 0.000 description 1
- 230000036285 pathological change Effects 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 238000003672 processing method Methods 0.000 description 1
- 230000001172 regenerating effect Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000000391 smoking effect Effects 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 231100000240 steatosis hepatitis Toxicity 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 230000008673 vomiting Effects 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/576—Immunoassay; Biospecific binding assay; Materials therefor for hepatitis
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6803—General methods of protein analysis not limited to specific proteins or families of proteins
- G01N33/6806—Determination of free amino acids
- G01N33/6812—Assays for specific amino acids
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/08—Hepato-biliairy disorders other than hepatitis
- G01N2800/085—Liver diseases, e.g. portal hypertension, fibrosis, cirrhosis, bilirubin
Definitions
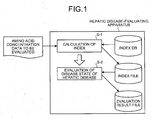
- the present invention relates to a hepatic disease-evaluating apparatus, a hepatic disease-evaluating method, a hepatic disease-evaluating system, and a hepatic disease-evaluating program and recording medium that calculate an index indicating the degree of hepatic fibrosis from amino acid concentration data to be evaluated based on an index formula and evaluate the disease state of the hepatic disease to be evaluated based on the index value.
- the Fischer ratio " (Leu+Val+Ile) ⁇ (Phe+Tyr)” based on blood amino acid concentration (see Nonpatent Literature 4) or the BTR (branched-chain amino acids and tyrosine ratio) ratio, which is a simplified Fischer ratio, " (Leu+Val+Ile) ⁇ Tyr” is used for diagnosis of hepatic encephalopathy in the patient with liver cirrhosis, as an index for use in clinical diagnosis of hepatic disease.
- An object of the present invention which was made to solve the problems above, is to provide a hepatic disease-evaluating apparatus, a hepatic disease-evaluating method, a hepatic disease-evaluating system, and a hepatic disease-evaluating program and a recording medium carrying the same that allow accurate evaluation of the progress of the disease state of hepatic disease and accurate determination, for example, of whether treatment with interferon/ribavirin combination is needed to patients with a hepatic disease.
- Another aspect of the present invention is the hepatic disease-evaluating system, wherein the index is the sum of two fractional expressions; the numerator in one fractional expression is any one of Phe and Tyr and the denominator in the one fractional expression is any one of Val, Leu, and Ile; and the numerator in the other fractional expression is the sum of at least one of Thr, Met and Orn and the denominator of the other fractional expression is the sum of at least one of Pro and Gly.
- hepatic fibrosis of a patient is in the stage of F0, F1, or F2 or in the stage of F3 or F4 more accurately. Specifically, it is also possible to determine whether the hepatic fibrosis of a patient is in the stage of F0, F1, F2 or F3 or in the stage of F4 more accurately.
- an index of the degree of hepatic fibrosis is calculated from previously obtained amino acid concentration data to be evaluated including amino acid concentration values, based on one or more indices (one index or combination of plurality of indices) (step S-1).
- the index is obtained by assigning the amino acid concentration values, based on the one or more indices.
- Data such as defective values and outliers may be removed from the amino acid concentration data before calculation of the index (data filtering.or data editing).
- the index used in step S-1 is a index newly generated by taking clinical need into consideration, and aimed, in particular, at discriminating two groups "F0, F1, or F2" and “F3 or F4" or two groups “F0, F1, F2, or F3” and "F4" in the progressive stages of hepatic fibrosis.
- An amino acid having smaller influence on the sum of Phe and Tyr, the sum of Thr, Met, and Orn, the sum of Val, Leu, and Ile, or the sum of Pro and Gly may be added as the variable in the index used in step S-1.
- the phrase "having smaller influence” means that addition of the amino acid does not deteriorate the determination (discrimination) efficiency of the index.
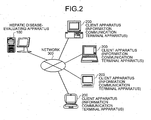
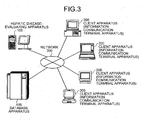
- the present system the configuration of the hepatic disease-evaluating system according to the present invention (hereinafter, referred to as the present system) will be described with reference to FIGS. 2 to 11 .
- the entire configuration of the present system will be described with reference to FIGS. 2 and 3 .
- FIG. 2 is a diagram showing an example of the entire configuration of the present system.
- FIG. 3 is a diagram showing another example of the entire configuration of the present system.
- the present system includes a hepatic disease-evaluating apparatus 100 which evaluates hepatic disease and client apparatuses 200 as information communication terminal apparatuses which provide the amino acid concentration data to be evaluated that are communicatively connected to each other via a network 300.
- a hepatic disease-evaluating apparatus 100 which evaluates hepatic disease and client apparatuses 200 as information communication terminal apparatuses which provide the amino acid concentration data to be evaluated that are communicatively connected to each other via a network 300.
- the user information file 106a stores information about users (user information).
- FIG. 5 is a chart showing an example of the information stored in the user information file 106a.
- the information stored in the user information file 106a includes user ID (identification) for identifying the user uniquely, user password for authentication of the user, user name, organization ID uniquely identifying the organization of the user, department ID for uniquely identifying the department of the user organization, department name, and electronic mail address of the user that are correlated to each other.
- the index database 106c stores the indices used in the index-calculating part 102g described below.
- FIG. 7 is a chart showing an example of the information stored in the index database 106c. As shown in FIG. 7 , the information stored in the index database 106c includes index number identifying each index uniquely and index of fractional expressions having amino acid concentrations as variables, that are correlated to each other.
- the index file 106d stores the index indicating the degree of hepatic fibrosis calculated in the index-calculating part 102g described below.
- FIG. 8 is a chart showing an example of the information stored in the index file 106d. As shown in FIG. 8 , the information stored in the index file 106d includes subject (sample) number of each subject to be evaluated, index number, and index value that are correlated to each other.
- it may store as needed sound files in the WAVE or AIFF (Audio Interchange File Format) Format for transmission to the client apparatuses 200 and image files of still image or motion picture in the JPEG (Joint Photographic Experts Group) or MPEG2 (Moving Picture Experts Group phase 2) format.
- WAVE Wireless Photographic Experts Group
- MPEG2 Motion Picture Experts Group phase 2
- the communication interface 104 allows communication between the hepatic disease-evaluating apparatus 100 and the network 300 (or communication apparatus such as router). Thus, the communication interface 104 has a function to communicate data via a communication line with other terminals.
- the input/output interface 108 is connected to the input device 112 and the output device 114.
- a monitor including home television), a speaker, or a printer may be used as the output device 114 (hereinafter, a monitor may be described as the output device 114).
- a keyboard, a mouse, a microphone, or a monitor functioning as a pointing device together with a mouse may be used as the input device 112.
- the controlling device 102 has an internal memory storing control programs such as OS (Operating System), programs for various processing procedures, and other needed data, and performs information processing for execution of various processings according to these programs.
- the controlling device 102 includes mainly an instruction-analyzing part 102a, a browsing processing part 102b, an authentication-processing part 102c, an electronic mail-generating part 102d, a Web page-generating part 102e, a receiving part 102f, an index-calculating part 102g, a disease state-evaluating part 102h, a result outputting part 102i, and a sending part 102j.
- the controlling device 102 performs data processing (data filtering or data editing) such as removal of data including defective values or many outliers and of variables for the defective value-including data in the amino acid concentration data obtained in the receiving part 102g described below.
- the disease state-evaluating part 102h may determine (discriminate) whether the progressive stage of hepatic fibrosis to be evaluated is in the stage of F0, F1, F2, or F3 or in the stage of F4, by comparing the index with a previously set particular threshold value (cutoff value).
- the client apparatus 200 may be realized, as peripheral parts such as printer, monitor, and image scanner connected as needed to information processing apparatus (such as known personal computer, workstation, family computer, Internet TV (Television), or the other information processing terminal (such as PHS (Personal Handyphone System) terminal, mobile phone terminal, mobile unit communication terminal or PDA (Personal Digital Assistants))), and also as software (including programs, data and others) for Web data-browsing function and electronic mail-processing function installed in the information processing apparatus.
- information processing apparatus such as known personal computer, workstation, family computer, Internet TV (Television), or the other information processing terminal (such as PHS (Personal Handyphone System) terminal, mobile phone terminal, mobile unit communication terminal or PDA (Personal Digital Assistants)
- PHS Personal Handyphone System
- PDA Personal Digital Assistants
- All or part of the controlling device 210 in the client apparatus 200 may be performed by a CPU and programs read and executed by the CPU.
- ROM 220 or HD 230 computer programs for giving instructions to the CPU and executing various processings together with the OS (Operating System) are recorded in the ROM 220 or HD 230.
- the computer programs may be stored in an application program server connected via any network to the client apparatus 200, and the client apparatus 200 may download all or part of them as needed. All or any part of the controlling device 210 may be substituted with hardware such as wired-logic.
- the network 300 has a function to connect the hepatic disease-evaluating apparatus 100, the client apparatuses 200, and the database apparatus 400 mutually, communicatively to each other, and is, for example, the Internet, intranet, or LAN (Local Area Network (both wired/wireless)).
- LAN Local Area Network (both wired/wireless)
- the network 300 may be VAN (Value Added Network), personal computer communication network, public telephone network (including both analog and digital), leased line network (including both analog and digital), CATV (Community Antenna Television) network, portable switched network or portable packet-switched network (including IMT2000 (International Mobile Telecommunication 2000) system, GSM (Global System for Mobile Communications) system, or PDC (Personal Digital Cellular)/PDC-P system), wireless calling network, local wireless network such as Bluetooth, PHS network, satellite communication network (including CS (Communication Satellite), BS (Broadcasting Satellite), and ISDB (Integrated Services Digital Broadcasting)), or the like.
- VAN Value Added Network
- personal computer communication network public telephone network (including both analog and digital), leased line network (including both analog and digital), CATV (Community Antenna Television) network, portable switched network or portable packet-switched network (including IMT2000 (International Mobile Telecommunication 2000) system, GSM (Global System for Mobile Communications) system, or PDC (Pers
- FIG. 11 is a block diagram showing an example of the configuration of the database apparatus 400 in the present system, showing conceptually only the region relevant to the present invention.
- the database apparatus 400 has functions to store, for example, the amino acid concentration data to be evaluated transmitted from the client apparatuses 200, the indices used in the hepatic disease-evaluating apparatus 100, and the evaluation results obtained in the hepatic disease-evaluating apparatus 100.
- the database apparatus 400 has mainly, a controlling device 402, such as CPU, which controls the entire database apparatus 400 integrally, a communication interface 404 connected to a communication apparatus such as router (not shown in the figure), for example, to a communication line, a memory device 406 storing various data, tables and others, and an input/output interface 408 connected to an input device 412 and an output device 414, and these parts are connected communicatively to each other via any communication channel.
- the database apparatus 400 is connected to the network 300 communicatively via a communication apparatus such as router and via a wired or wireless communication line such as private line.
- the memory device 406 is a storage means, and may be, for example, memory apparatus such as RAM or ROM, fixed disk drive such as hard' disk, flexible disk, optical disk, or the like. Various programs, tables, files, web-page files, and others used in various processings are stored in the memory device 406.
- the communication interface 404 allows communication between the database apparatus 400 and the network 300 (or communication apparatus such as router). Thus, the communication interface 404 has a function to communicate data with other terminal via a communication line.
- the input/output interface 408 is connected to the input device 412 and the output device 414.
- a monitor including home television), a speaker, or a printer may be used as the output device 414 (hereinafter, a monitor may be described as the output device 414).
- a keyboard, a mouse, a microphone, or a monitor functioning as a pointing device together with a mouse may be used as the input device 412.
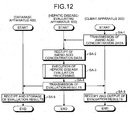
- the client apparatus 200 connects itself to the hepatic disease-evaluating apparatus 100 via the network 300, when the user specifies the Web site address (such as URL) provided from the hepatic disease-evaluating apparatus 100, via the input device 250 on the screen displaying Web browser 211.
- the Web browser 211 sends the Web site's URL using a particular protocol via the communication IF 280, transmits an instruction demanding transmission of the Web page corresponding to the amino acid concentration data transmission screen to the hepatic disease-evaluating apparatus 100 based on the routing of the URL.
- the authentication-processing part 102c examines the input user ID and password by comparing them with the user ID and user password stored in the user information file 106a for authentication, and the browsing processing part 102b sends the Web data to the client apparatus 200, only when the user is authenticated.
- the client apparatus 200 is identified with the IP (Internet Protocol) address transmitted from the client apparatus 200 together with the transmission instruction.
- IP Internet Protocol
- the client apparatus 200 receives in the receiving part 213 the Web data transmitted from the hepatic disease-evaluating apparatus 100 via the communication IF 280, examines the Web data with the Web browser 211, and displays the amino acid concentration data transmission screen on the monitor 261.
- the instruction demanding transmission of screen from the client apparatus 200 to the hepatic disease-evaluating apparatus 100, the transmission of the Web data from the hepatic disease-evaluating apparatus 100 to the client apparatus 200 and the display of the Web page in the client apparatus 200 are performed almost similarly, and thus, detailed description will be omitted below.
- the hepatic disease-evaluating apparatus 100 may analyze the identifier transmitted from the client apparatus 200 and the instruction from the client apparatus 200 in the instruction-analyzing part 102a and send an instruction demanding transmission of the index formula used in calculation of the index to the database apparatus 400; and the database apparatus 400 may analyze the instruction sent form the hepatic disease-evaluating apparatus 100 in the instruction-analyzing part 402a and send the index formula (specifically updated newest index) stored in a particular region of the memory device 406 to the hepatic disease-evaluating apparatus 100 via the communication interface 404.
- the hepatic disease-evaluating apparatus 100 then receives the amino acid concentration data transmitted from the client apparatuses 200 via the communication interface 104 in the receiving part 102f, stores the received amino acid concentration data in a particular region of the amino acid concentration data file 106b, and executes [3-2. hepatic disease evaluation processing] described below (step SA-3).
- the sending part 102f of the hepatic disease-evaluating apparatus 100 then sends the evaluation results (evaluation results concerning the disease state of the hepatic disease of the subject to be evaluated) obtained in step SA-3 to the client apparatus 200 that has sent the amino acid concentration data of the subject to be evaluated and the database apparatus 400 (step SA-4).
- the hepatic disease-evaluating apparatus 100 first generates a Web page for display of evaluation results in the Web page-generating part 102 and stores it in a particular memory region of the memory device 106.
- the user is authenticated as described above by inputting a predetermined URL (Uniform Resource Locator) into the Web browser 211 of the client apparatus 200 via the input device 250, and the client apparatus 200 sends a Web page browsing instruction to the hepatic disease-evaluating apparatus 100.
- the hepatic disease-evaluating apparatus 100 then examines the browsing instruction transmitted from the client apparatus 200 in the browsing processing part 102a and reads the Web page out of the memory device 106.
- the hepatic disease-evaluating apparatus 100 then sends Web data corresponding to the read-out Web page to the client apparatus 200 from the sending part 102f.
- the hepatic disease-evaluating apparatus 100 may send only the evaluation results or the data same as the Web data sent to the client apparatus 200 to the database apparatus 400.
- step SB-1 data editing
- stage F0, F1, or F2 and stage F3 or F4 For clinical purpose, there exists a need for a method of discriminating the progressive stage of fibrosis and determining whether the treatment with interferon/ribavirin combination is needed. In particular, there is a need for discriminating two groups: stage F0, F1, or F2 and stage F3 or F4, or stage F0, F1, F2, or F3 and stage F4.
- hepatic fibrosis of a patient is in the stage of F0, F1, or F2 or in the stage of F3 or F4 more accurately.
- the hepatic disease includes at least one of hepatitis, chronic hepatitis, hepatic fibrosis and liver cirrhosis, and thus, it is possible to apply the present invention suitably to clinically, .frequently required evaluation of the disease state of at least one of diseases such as hepatitis, chronic hepatitis, hepatic fibrosis and liver cirrhosis.
- the "program” is a data processing method written in any language or by any description method and may be of any format such as source code or binary code.
- the "program” may not be an independent program, and may be operated together with plurality of modules and libraries or with a different program such as OS (Operating System).
- the program is stored on a recording medium and read mechanically as needed by the hepatic disease-evaluating apparatus 100. Any well-known configuration or procedure may be used for reading the programs recorded on the recording medium in each apparatus and for reading procedure and installation of the procedure after reading.
- the “recording media” includes any “portable physical media”, “fixed physical media”, and “communication media”.
- Examples of the “portable physical media” include flexible disk, magnetic optical disk, ROM, EPROM (Erasable Programmable Read Only Memory), EEPROM (Electronically Erasable and Programmable Read Only Memory), CD-ROM (Compact Disk Read Only Memory), MO (Magneto-Optical disk), DVD (Digital Versatile Disk), and the like.
- Examples of the "fixed physical media” include various media installed in a computer system such as ROM, RAM, and HD.
- the “communication media” are, for example, media storing the program for a short period of time such as communication line and carrier wave when the program is transmitted via a network such as LAN (Local Area Network), WAN (Wide Area Network), or the Internet.
- the sensitivity was 89%; the specificity, 88%; the positive predictive value, 79%; and the negative predictive value, 88%, in discrimination of two groups "F0, F1, or F2" and "F3 or F4" by using the index 1, when the cutoff value was 0.95.
- the index 1 was found to be a useful index higher in diagnostic performance in discrimination of two groups "F0, F1, or F2" and "F3 or F4" (especially, superior in diagnostic performance to the Fischer ratio).
- the hepatic disease-evaluating apparatus, the hepatic disease-evaluating method, the hepatic disease-evaluating system, and the hepatic disease-evaluating program and recording medium according to the present invention are applicable to many industrial fields, especially in pharmaceutical, food, and medical fields, and are extremely useful particularly in the field of bioinformatics field of performing disease state prediction, disease risk prediction, proteome/metabolome analysis, and the like.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Immunology (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Physics & Mathematics (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- Cell Biology (AREA)
- Food Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- Pathology (AREA)
- Analytical Chemistry (AREA)
- Microbiology (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Communicable Diseases (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Bioinformatics & Computational Biology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005157802 | 2005-05-30 | ||
| PCT/JP2006/310164 WO2006129513A1 (ja) | 2005-05-30 | 2006-05-22 | 肝疾患評価装置、肝疾患評価方法、肝疾患評価システム、肝疾患評価プログラムおよび記録媒体 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1887362A1 true EP1887362A1 (de) | 2008-02-13 |
| EP1887362A4 EP1887362A4 (de) | 2010-01-27 |
Family
ID=37481442
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP06746698A Withdrawn EP1887362A4 (de) | 2005-05-30 | 2006-05-22 | Vorrichtung, verfahren, system, programm und aufzeichnungsmedium zur einstufung einer leberkrankheit |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US8244476B2 (de) |
| EP (1) | EP1887362A4 (de) |
| JP (1) | JPWO2006129513A1 (de) |
| WO (1) | WO2006129513A1 (de) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010106140A1 (en) | 2009-03-19 | 2010-09-23 | Universite D'angers | Non-invasive method for assessing liver fibrosis progression |
| CN102653552A (zh) * | 2011-03-02 | 2012-09-05 | 上海交通大学附属第一人民医院 | 慢性乙型肝炎肝纤维化的血清多肽标记物及其检测方法 |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008016111A1 (fr) | 2006-08-04 | 2008-02-07 | Ajinomoto Co., Inc. | Procédé permettant d'évaluer un cancer du poumon, dispositif d'évaluation d'un cancer du poumon, procédé d'évaluation d'un cancer du poumon, système d'évaluation d'un cancer du poumon, programme d'évaluation d' |
| EP2053405A4 (de) * | 2006-08-04 | 2009-11-11 | Ajinomoto Kk | Verfahren zur bewertung von stress, stressbewertungsvorrichtung, stressbewertungsverfahren, stressbewertungssystem, stressbewertungsprogramm und aufzeichnungsmedium |
| WO2008075663A1 (ja) | 2006-12-21 | 2008-06-26 | Ajinomoto Co., Inc. | 大腸癌の評価方法、ならびに大腸癌評価装置、大腸癌評価方法、大腸癌評価システム、大腸癌評価プログラムおよび記録媒体 |
| JP5746810B2 (ja) | 2006-12-21 | 2015-07-08 | 味の素株式会社 | 乳癌の評価方法、ならびに乳癌評価装置、乳癌評価方法、乳癌評価システム、乳癌評価プログラムおよび記録媒体 |
| JPWO2009001862A1 (ja) * | 2007-06-25 | 2010-08-26 | 味の素株式会社 | 内臓脂肪蓄積の評価方法 |
| JPWO2009054351A1 (ja) * | 2007-10-25 | 2011-03-03 | 味の素株式会社 | 生体状態評価装置、生体状態評価方法、生体状態評価システム、生体状態評価プログラムおよび記録媒体 |
| EP2209001A4 (de) * | 2007-10-25 | 2010-10-27 | Ajinomoto Kk | Verfahren zur beurteilung von gestörter glucosetoleranz |
| CN102066946B (zh) | 2008-06-20 | 2016-08-31 | 味之素株式会社 | 前列腺疾病的评价方法 |
| EP2380018A1 (de) * | 2008-12-18 | 2011-10-26 | University Of Utah | Systeme und verfahren zur klassifizierung und durchsuchung biologischer materialien zur verwendung für therapie- und/oder forschungszwecke |
| US20100280986A1 (en) * | 2009-05-04 | 2010-11-04 | Roche Palo Alto | Systems and methods for tailoring acute and chronic viral infection treatments to increase the probability of "cure" for a given subject |
| CA2799032A1 (en) * | 2010-06-10 | 2012-01-05 | Metanomics Health Gmbh | Methods to diagnose liver diseases |
| JP6260275B2 (ja) * | 2011-06-30 | 2018-01-17 | 味の素株式会社 | 脂肪肝の評価方法、脂肪肝評価装置、脂肪肝評価方法、脂肪肝評価プログラム、脂肪肝評価システム、および端末装置 |
| WO2013011919A1 (ja) * | 2011-07-15 | 2013-01-24 | 味の素株式会社 | Nashの評価方法、nash評価装置、nash評価方法、nash評価プログラム、nash評価システム、情報通信端末装置、およびnashの予防・改善物質の探索方法 |
| JP5234474B1 (ja) | 2012-01-30 | 2013-07-10 | 富山県 | 新規l−アミノ酸オキシダーゼ、l−リジンの測定方法、キット及び酵素センサ |
| KR102427924B1 (ko) * | 2016-10-04 | 2022-08-02 | 아지노모토 가부시키가이샤 | 췌장암의 평가 방법, 평가 장치, 평가 프로그램, 평가 시스템, 및 단말 장치 |
| KR102477319B1 (ko) * | 2016-12-01 | 2022-12-15 | 아지노모토 가부시키가이샤 | 암 모니터링의 방법, 산출 방법, 평가 장치, 산출 장치, 평가 프로그램, 산출 프로그램, 평가 시스템, 및 단말 장치 |
| WO2024237258A1 (ja) * | 2023-05-17 | 2024-11-21 | 株式会社島津製作所 | 非アルコール性脂肪肝疾患の識別方法、および、バイオマーカー |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61126472A (ja) * | 1984-11-24 | 1986-06-13 | Advance Res & Dev Co Ltd | 診断方法 |
| US20040022827A1 (en) * | 1997-09-30 | 2004-02-05 | Chugai Seiyaku Kabusiki Kaisha | Remedy for hepatopathy |
| JP2000298131A (ja) | 1999-04-14 | 2000-10-24 | Ryuzo Kawamori | 前糖尿病状態の検出方法 |
| JP3975279B2 (ja) | 1999-11-01 | 2007-09-12 | 旭化成ファーマ株式会社 | 糖尿病予備群の検査方法 |
| US6865415B2 (en) | 2000-04-18 | 2005-03-08 | Yamato Scale Co., Ltd. | Visceral fat determining device |
| US6631330B1 (en) | 2000-08-21 | 2003-10-07 | Assistance Publique-Hopitaux De Paris (Ap-Hp) | Diagnosis method of inflammatory, fibrotic or cancerous disease using biochemical markers |
| AU2003227237A1 (en) | 2002-03-29 | 2003-10-13 | Asahi Kasei Pharma Corporation | Method of detecting mild impaired glucose tolerance or insulin hyposecretion |
| AU2003289263A1 (en) | 2002-12-09 | 2004-06-30 | Ajinomoto Co., Inc. | Organism condition information processor, organism condition information processing method, organism condition information managing system, program, and recording medium |
| WO2006098192A1 (ja) | 2005-03-16 | 2006-09-21 | Ajinomoto Co., Inc. | 生体状態評価装置、生体状態評価方法、生体状態評価システム、生体状態評価プログラム、評価関数作成装置、評価関数作成方法、評価関数作成プログラムおよび記録媒体 |
| BRPI0617332A2 (pt) | 2005-10-11 | 2011-07-26 | Tethys Bioscience Inc | marcadores asoociados ao diabetes e mÉtodos de uso dos mesmos |
| JPWO2009001862A1 (ja) | 2007-06-25 | 2010-08-26 | 味の素株式会社 | 内臓脂肪蓄積の評価方法 |
-
2006
- 2006-05-22 EP EP06746698A patent/EP1887362A4/de not_active Withdrawn
- 2006-05-22 WO PCT/JP2006/310164 patent/WO2006129513A1/ja not_active Ceased
- 2006-05-22 JP JP2007518917A patent/JPWO2006129513A1/ja active Pending
-
2007
- 2007-11-29 US US11/987,415 patent/US8244476B2/en not_active Expired - Fee Related
Non-Patent Citations (2)
| Title |
|---|
| See also references of WO2006129513A1 * |
| ZHANG Q ET AL: "Plasma amino acid profiles applied for diagnosis of advanced liver fibrosis in patients with chronic hepatitis C infection" HEPATOLOGY RESEARCH, AMSTERDAM, NL, vol. 34, no. 3, 1 March 2006 (2006-03-01), pages 170-177, XP025177439 ISSN: 1386-6346 [retrieved on 2006-03-01] * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010106140A1 (en) | 2009-03-19 | 2010-09-23 | Universite D'angers | Non-invasive method for assessing liver fibrosis progression |
| US10861582B2 (en) | 2009-03-19 | 2020-12-08 | Centre Hospitalier Universitaire D'angers | Non-invasive method for assessing liver fibrosis progression |
| CN102653552A (zh) * | 2011-03-02 | 2012-09-05 | 上海交通大学附属第一人民医院 | 慢性乙型肝炎肝纤维化的血清多肽标记物及其检测方法 |
| CN102653552B (zh) * | 2011-03-02 | 2015-01-07 | 上海交通大学附属第一人民医院 | 慢性乙型肝炎肝纤维化的血清多肽标记物及其检测方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20080154515A1 (en) | 2008-06-26 |
| EP1887362A4 (de) | 2010-01-27 |
| JPWO2006129513A1 (ja) | 2008-12-25 |
| US20110282585A9 (en) | 2011-11-17 |
| WO2006129513A1 (ja) | 2006-12-07 |
| US8244476B2 (en) | 2012-08-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8244476B2 (en) | Hepatic disease-evaluating apparatus, hepatic disease-evaluating method, hepatic disease-evaluating system, hepatic disease-evaluating program and recording medium | |
| US9599618B2 (en) | Method, apparatus, system, program, and computer-readable recording medium for evaluating colorectal cancer | |
| EP1862797A1 (de) | Vorrichtung, verfahren, system und programm zur bewertung biologischer zustände, vorrichtung, verfahren und programm zur erzeugung einer bewertungsfunktion sowie aufzeichnungsmedium | |
| US8673647B2 (en) | Stress evaluating apparatus, method, system and program and recording medium therefor | |
| US9664681B2 (en) | Lung cancer evaluating apparatus, method, system, and program and recording medium therefor | |
| US9182407B2 (en) | Method of evaluating visceral fat accumulation, visceral fat accumulation-evaluating apparatus, visceral fat accumulation-evaluating method, visceral fat accumulation-evaluating system, visceral fat accumulation-evaluating program, recording medium, and method of searching for prophylactic/ameliorating substance for visceral fat accumulation | |
| EP2103941A1 (de) | Verfahren zur bewertung von krebs, krebsbewertungsvorrichtung, -verfahren, -system, -programm und aufzeichnungsmedium | |
| US20140127819A1 (en) | Method of evaluating nash, nash-evaluating apparatus, nash-evaluating method, nash-evaluating product, nash-evaluating system, information communication terminal apparatus, method of searching for preventing/ameliorating substance for nash | |
| EP2053395A1 (de) | Stoffwechselsyndrom-bewertungsverfahren, stoffwechselsyndrom-bewertungsvorrichtung, stoffwechselsyndrom-bewertungsverfahren, stoffwechselsyndrom-bewertungssystem, stoffwechselsyndrom-bewertungsprogramm und aufzeichnungsmedium sowie verfahren zur suche nach einer prophylaktischen/lindernden substanz für stoffwechselsyndrom | |
| US9459255B2 (en) | Method of evaluating breast cancer, breast cancer-evaluating apparatus, breast cancer-evaluating method, breast cancer-evaluating system, breast cancer-evaluating program and recording medium | |
| US20110035156A1 (en) | Method of evaluating gastric cancer, gastric cancer-evaluating apparatus, gastric cancer-evaluating method, gastric cancer-evaluating system, gastric cancer-evaluating program and recording medium | |
| US20110138889A1 (en) | Method of evaluating prostatic disease | |
| US9971866B2 (en) | Method of evaluating fatty liver related disease, fatty liver related disease-evaluating apparatus, fatty liver related disease-evaluating method, fatty liver related disease-evaluating program product, fatty liver related disease-evaluating system, information communication terminal apparatus, and method of searching for prophylactic/ameliorating substance for fatty liver related disease | |
| JPWO2009054351A1 (ja) | 生体状態評価装置、生体状態評価方法、生体状態評価システム、生体状態評価プログラムおよび記録媒体 | |
| EP2124059A1 (de) | Verfahren zur bewertung von ibd, aminosäuredatenverarbeitungsgerät, aminosäuredatenverarbeitungsverfahren, aminosäuredatenverarbeitungssystem, aminosäuredatenverarbeitungsprogramm und aufzeichnungsmedium | |
| EP2209001A1 (de) | Verfahren zur beurteilung von gestörter glucosetoleranz | |
| JP2013178239A (ja) | 早期腎症の評価方法、早期腎症評価装置、早期腎症評価方法、早期腎症評価プログラム、早期腎症評価システムおよび情報通信端末装置 | |
| KR20170132303A (ko) | 평가 방법, 평가 장치, 평가 프로그램 제품, 평가 시스템, 및 단말 장치 | |
| JP2013178238A (ja) | 心血管イベントの評価方法、心血管イベント評価装置、心血管イベント評価方法、心血管イベント評価プログラム、心血管イベント評価システムおよび情報通信端末装置 | |
| Dunphy et al. | Targeted Metabolomics Approach Identifies Alterations in the Plasma Metabolome of Multiple Myeloma Patients with and without Extramedullary Spread | |
| Vrana et al. | Diagnosis and classification of amyloidosis in abdominal subcutaneous fat aspiration specimens using mass spectrometry based proteomics | |
| States et al. | Pilot Phase Results of the Human Proteome Organization Plasma Proteome Project. | |
| Bergen III et al. | Monitoring Minimum Residual Disease In Multiple Myeloma Patients By LC-MS/MS | |
| Weissinger et al. | Prospective Evaluation of Graft-Versus-Host Disease-Specific Proteomics Pattern in Patients after Allogeneic Stem Cell Transplantation. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20071207 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| DAX | Request for extension of the european patent (deleted) | ||
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20091229 |
|
| 17Q | First examination report despatched |
Effective date: 20100414 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20111229 |