EP1801262B1 - Procédé de traitement par carboxylatation de surfaces métalliques, utilisation de ce procédé pour la protection temporaire contre la corrosion, et procédé de fabrication d'une tôle mise en forme ainsi carboxylatée - Google Patents
Procédé de traitement par carboxylatation de surfaces métalliques, utilisation de ce procédé pour la protection temporaire contre la corrosion, et procédé de fabrication d'une tôle mise en forme ainsi carboxylatée Download PDFInfo
- Publication number
- EP1801262B1 EP1801262B1 EP05292773A EP05292773A EP1801262B1 EP 1801262 B1 EP1801262 B1 EP 1801262B1 EP 05292773 A EP05292773 A EP 05292773A EP 05292773 A EP05292773 A EP 05292773A EP 1801262 B1 EP1801262 B1 EP 1801262B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- process according
- bath
- acids
- mixture
- carboxylation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/48—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 not containing phosphates, hexavalent chromium compounds, fluorides or complex fluorides, molybdates, tungstates, vanadates or oxalates
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M173/00—Lubricating compositions containing more than 10% water
- C10M173/02—Lubricating compositions containing more than 10% water not containing mineral or fatty oils
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/48—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 not containing phosphates, hexavalent chromium compounds, fluorides or complex fluorides, molybdates, tungstates, vanadates or oxalates
- C23C22/50—Treatment of iron or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/48—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 not containing phosphates, hexavalent chromium compounds, fluorides or complex fluorides, molybdates, tungstates, vanadates or oxalates
- C23C22/53—Treatment of zinc or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/48—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 not containing phosphates, hexavalent chromium compounds, fluorides or complex fluorides, molybdates, tungstates, vanadates or oxalates
- C23C22/56—Treatment of aluminium or alloys based thereon
Definitions
- the invention relates to a method for the formation of conversion layers on a metal surface selected from zinc, iron, aluminum, copper, lead and their alloys, as well as galvanized, electrozinced, aluminized, coppered steels. , making it possible to produce at high speed conversion layers formed of very small crystals, from 1 to 20 ⁇ m.
- These different conversion treatments generally consist of anodic dissolution of the metal elements of the surface, followed by precipitation on this surface of the compounds formed by the reaction of the dissolved metal elements with the species present in the conversion bath.
- Dissolution requires the creation of oxidizing conditions vis-à-vis the metal surface and usually takes place in an acidic environment.
- the precipitation of the metal compounds to form the conversion layer requires a sufficiently high concentration and is favored by a medium that is locally less acidic under the effect of the dissolution of the metal. It is the nature and structure of the precipitated compounds on the treated surface that determine the degree of protection against corrosion, improvement of tribological properties and / or adhesion, as well as the other properties of the layer.
- the conversion baths essentially contain anions and cations capable of forming insoluble compounds with the dissolved metal of the surface.
- the main conversion treatments applied to the steels are thus chromating treatments on galvanized steel (by dip galvanizing or electrogalvanizing) or aluminized, phosphating on unalloyed bare steels or coated steels, or even oxalation on alloy steels such as as stainless steels, for example.
- the treated surface After being brought into contact with a conversion bath, the treated surface is generally rinsed to remove unreacted surface and / or treatment solution components, and this surface is dried, particularly to harden the coating layer. conversion and / or to improve its properties.
- the conversion treatment may itself be preceded by a pretreatment, generally consisting of a preliminary degreasing and rinsing of the surface followed by a so-called refining operation using a pretreatment solution adapted to create and / or promote germination sites on the surface to be treated.
- a pretreatment generally consisting of a preliminary degreasing and rinsing of the surface followed by a so-called refining operation using a pretreatment solution adapted to create and / or promote germination sites on the surface to be treated.
- sols or colloidal suspensions of titanium salts which allow the subsequent obtaining of a conversion layer having smaller crystals in a denser layer.
- a post-treatment can be carried out on a conversion layer obtained by phosphatation.
- conversion layers are formed by bringing the surface into contact with an aqueous, organic or hydro-organic bath comprising one or more carboxylic acids in solution or in emulsion at a concentration of at least 0.1 mol / liter, and this in oxidizing conditions vis-à-vis the metal surface.
- This or these acids are saturated or unsaturated aliphatic monocarboxylic or dicarboxylic acids.
- the object of the invention is to propose treatments by carboxylation of metal surfaces, in particular zinc and zinc alloy layers coating the galvanized and electrogalvanized steel sheets, solving better than the existing treatments the problems that we have just described. to quote.
- the respective proportions of the acids are x ⁇ 3% - y ⁇ 3%.
- Said oxidizing conditions can be created by the presence in the bath of an oxidizing compound for the metal surface.
- Said oxidizing compound may be hydrogen peroxide.
- Said oxidizing compound may be sodium perborate.
- Said oxidizing conditions can be created by the bath application of an electric current.
- the bath may be a hydro-organic bath and contain a co-solvent.
- This co-solvent may be chosen from 3-methoxy-3-methylbutan-1-ol, ethanol, n-propanol, dimethylsulfoxide, N-methyl-2-pyrrolidone and 4-hydroxy-4-methyl -2-pentanone, diacetone alcohol.
- Said bath may be an aqueous bath and include a surfactant and / or a dispersant.
- Said surfactant may be chosen from alkylpolyglycosides, ethoxylated fatty alcohols, ethoxylated fatty acids, ethoxylated oils, ethoxylated nonylphenols and ethoxylated sorbitan esters.
- Said dispersant may be chosen from high molecular weight polyols, carboxylic acid salts such as (meth) acrylic copolymers, polyamide derivatives such as polyamide waxes.
- Said saturated carboxylic acids may each have an even number of carbon atoms.
- Said saturated carboxylic acids may be lauric acid and palmitic acid.
- Said metal surface may be a galvanized steel sheet, and the bath may contain an Al 3+ complexing agent .
- the invention also relates to a use of the above method for the temporary protection against corrosion of said metal surface.
- the subject of the invention is also a process for producing a shaped sheet having a metal surface chosen from zinc, iron, aluminum, copper, lead, and their alloys as well as galvanized, aluminized, copper-plated steels, in which a carboxylation treatment of said sheet is carried out and is shaped, characterized in that said carboxylation treatment is performed by the above method.
- Said sheet may be steel coated with zinc or a zinc alloy and is shaped by stamping.
- the invention is based on the use, for composing the solution or the carboxylation emulsion, of a binary or ternary eutectic of saturated C 10 -C 18 linear fatty acids.
- the acids used are all even-numbered acids of carbon atoms.
- the binary eutectic of C 12 - C 16 acids is particularly preferred.
- the concentration of the eutectic in the carboxylate bath is greater than or equal to 20 g / l.
- eutectic refers to a mixture in the composition of the eutectic containing two or three C 10 -C 18 saturated linear fatty acids.
- the treatment bath may contain only the eutectic, a surfactant and water, if the necessary oxidizing conditions are obtained by electrochemical means.
- electrochemical means namely by adding an oxidizing compound, such as hydrogen peroxide.
- oxidizing compound such as hydrogen peroxide.
- the minimum concentration of 20 g / l of the eutectic is chosen because, below this limit, the rate of formation of the carboxylated layer is no longer sufficient to obtain an effective conversion layer with a duration of treatment compatible with industrial requirements.
- the ability of saturated linear aliphatic monocarboxylates to inhibit the aqueous corrosion of metals (Cu, Fe, Pb, Zn and Mg) in neutral and aerated solution has been widely demonstrated.
- the protection provided is due to the presence of a thin film consisting of crystals of metal soap and hydroxide of the treated metal.
- the protective layer is formed under oxidizing conditions and has a corrosion resistance closely dependent on the length of the carbon chain and the concentration of the carboxylate.
- a carboxylation bath contains a linear C n saturated carboxylic acid of general formula (CH 3 (CH 2 ) n-2 COOH), with n ⁇ 7, denoted HC n , dissolved in water or in a generally equivolumic water-non-aqueous solvent mixture (ethanol, etc.).
- An oxidant such as hydrogen peroxide or sodium perborate, is added to the bath to produce a sufficient amount of Zn ++ cations at the zinc / solution interface.
- the pH of the bath is close to 5.
- the oxidizing conditions producing the Zn ++ cations are obtained by circulating an electric current between the surface to be protected and a counter electrode immersed in the bath.
- the essential formation reaction of the carboxylated layer on the zinc surface is: Zn 2+ + 2 C n - ⁇ Zn (C n ) 2 ⁇
- the compounds that can be used in the context of the invention may be derived from products of the green chain, that is to say from agricultural production for non-food use (sunflower oils, flax, colza ). They advantageously replace the polluting mineral oils used for the lubrication of metal surfaces and the phosphating and chromating solutions used for the protection of these same surfaces against corrosion.
- the inventors have found that still improved results, both in terms of protection against corrosion and behavior of the carboxylate coating in use (reduction of dusting) could be obtained in the case where a mixture is used.
- saturated C 10 -C 18 fatty acids Such a mixture provides a significant improvement in corrosion protection compared to coatings obtained with a single acid or mixture of acids of composition not close to a eutectic.
- the lubricating properties of these coatings according to the invention are excellent. They make it possible to do without an oiling of the coated product during its shaping.
- saturated fatty acids those containing an even number of carbon atoms are preferred.
- the study of their binary mixtures makes it possible to highlight the existence of two particular proportions for which appear respectively an inflection and a minimum in the curve of the melting point.
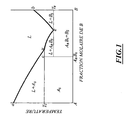
- the figure 1 schematizes the equilibrium diagram of fatty acid mixtures A and B as a function of temperature.
- the minimum e indicates the formation of a eutectic and the change of slope at point u is due, in general, to the existence of a defined molecular compound c of formula A m B n (m and n denote the molar fractions of A and B respectively).
- the figures 2b and 2d represent the HC 12 / HC 16 and HC 12 / HC 18 binary diagrams. It is found that the eutexte point e, as well as the point of inflection u corresponding to the complex, do not appear respectively at 25 and 50%, as is the case with acid mixtures whose lengths of chains differ only by two carbon atoms ( fig.2a for HC 10 / HC 12 and 2C for HC 16 / HC 18 ). Eutectic is shifted to higher molar concentrations of the shortest fatty acid.
- the shape of the binary diagram and the positions of the points u and e are a function of the more or less limited stability of the complex.
- Table 1 shows the compositions of the eutectics e of various binary mixtures and their melting points T f (e) .
- Table 1 The eutectic compositions given in Table 1 are approximate. According to the publications, they can vary from a few percent. These differences are due to the purity of the fatty acids used. Table 1 - Properties of the studied fatty acid mixtures HC blends n Composition e (% mol ) T f (e) (° C) HC 10 / HC 12 65/35 18 HC 12 / HC 14 69/31 34.2 HC 12 / HC 16 81/19 32.7 HC 12 / HC 18 81.5 / 18.5 37.0 HC 14 HC 16 58/42 42.6 HC 14 / HC 18 61/39 44.1 HC 16 / HC 18 72.5 / 27.5 51.1
- the sheets were degreased in an alkaline degreasing bath, similar to those used in industrial alkaline phosphating. They were then rinsed. Then the carboxylation treatment took place chemically (presence of an oxidant in the bath, such as hydrogen peroxide or a sodium perborate tetrahydrate) or electrochemical.
- an oxidant in the bath such as hydrogen peroxide or a sodium perborate tetrahydrate
- the oxidizing conditions allow a rapid reaction between Zn 2+ and C n - , providing fine crystals of Zn carboxylate.
- the concentration of H 2 O 2 in the solution is, for example, from 2 to 15 g / l. Below 2 g / l the medium is generally not enough oxidizing to form enough Zn 2+ in solution. The duration of the reaction may not be compatible with industrial requirements. Above 15 g / l, the medium is generally too oxidizing and the crystals are poorly formed. The optimum concentration is about 8 to 12 g / l H 2 O 2 in the solution.
- sodium perborate Compared to hydrogen peroxide, sodium perborate has the disadvantage of less solubility in water. The use of hydrogen peroxide thus provides greater flexibility in the choice of oxidant concentrations.
- the preferred co-solvent is 3-methoxy-3-methylbutan-1-ol (MMB). It is a green and biodegradable solvent. In addition, its flash point, which is the temperature from which it becomes flammable, is 71 ° C, compared for example with that of ethanol which is 12 ° C. MMB therefore provides better safety conditions than ethanol. It is also possible to use, in particular, ethanol, n-propanol, dimethylsulfoxide, N-methyl-2-pyrrolidone, 4-hydroxy-4-methyl-2-pentanone or diacetone alcohol.
- a first advantage is the lowering of the melting temperature compared to the use of a single fatty acid, as is apparent from the figure 2 . This keeps the carboxylate bath at a relatively low temperature of about 45 ° C in many cases.
- Eutectic is prepared by melting for several hours the mixture of fatty acids component. The mixture is then slowly cooled to room temperature.
- electrogalvanized steel sheets (thickness of the Zn layer: 7.5 ⁇ m) were treated to obtain a weight of carboxylated layer of between 1 and 2 g / m 2 , of which experience shows that it provides a maximum coverage rate of the sheet.
- the weight of the carboxylate layer is evaluated by measuring the difference in mass between the carboxylate substrate and the substrate etched with dichloroethane under ultrasound, which treatment causes the dissolution of the carboxylate layer.
- the aqueous corrosion resistance of the test samples was tested in a conventional three-electrode electrochemical cell, by monitoring the corrosion potential and measuring the polarization resistance.
- the electrolyte used is water according to ASTM D1384-87 (148 mg / l of Na 2 SO 4 , 138 mg / l of NaHCO 3 , 165 mg / l of NaCl, pH 7.8). This corrosive solution is usually used to evaluate the effectiveness of corrosion inhibitors in the laboratory.
- the resistance to atmospheric corrosion of 50 cm 2 samples was studied according to DIN 50017 using a climatic chamber where the samples were placed vertically and subjected to cycles of 24 h, each with successive exposure. from 8 h to 100% humidity (twice water at 40 ° C) and then to ambient air for 16 h.
- the degradation of the coating was estimated by visual observation and X-ray diffraction.
- Dusting of the samples was evaluated by measuring the mass difference of the substrate before and after successive passes between two squeezing rollers.
- the loss of mass thus measured can be related to the dusting tendency of the coating.
- Tribological tests were carried out to evaluate the lubricating properties of the coating during stamping. They were carried out on a plane / plane tribometer with tightening force control, by scrolling the sample of pressed sheet at a speed of 1 to 100 mm / s, and by measuring the evolution of the distance between the tools. planes ensuring the tightening of the sample. It is thus possible to determine the coefficient of friction as a function of the clamping pressure.
- compositions of the baths were as follows: medium 50% by volume of water and 50% by volume of 3-methoxy-3-methylbutan-1-ol (MMB); concentration of H 2 O 2 5 g / l; temperature 45 ° C .; compositions and concentrations of eutectics and duration of carboxylation according to Table 2: Table 2: compositions and concentrations of eutectics tested and duration of carboxylation Mixed % mol eutectic Concentration (g / l) Duration of carboxylation (s) HC 10 / HC 12 64/35 85 4 HC 12 / H 16 ; 81/19 55 4 HC 12 / HC 18 81.5 / 18.5 45 2
- the residence times of the sheet samples in the bath were determined to obtain a carboxylate layer weight of between 1 and 1.5 g / m 2 .
- the figure 3 shows the evolution over time of the polarization resistance Rp of the coatings, and the figure 4 shows this same development for the corrosion potential E corr in the corrosive water, for the three coatings tested previously defined and, by reference, for a galvanized coating EG not carboxylated.
- the coatings according to the invention have much better performances than coatings resulting from simple electrogalging.
- the polarization resistance is of the order of 2 k ⁇ .cm 2
- the carboxylation coatings usually made with water-solvent solutions based on a single fatty acid provide only one relatively weak improvement of this value (up to 15 k ⁇ .cm 2 ).
- the coatings according to the invention provide values of the order of 5 to 15 times higher than those observed for coatings electrozinced alone. Coatings obtained with HC 12 / HC 16 first, and with HC 12 / HC 18 second, provide the best results in absolute value and stability in time.
- those of the coatings according to the invention are 80 to 140 mV higher than the values obtained for the electrogalvanized coating.
- the HC 12 / HC 16 again gives the best result.
- the coatings obtained with a single fatty acid in a water-solvent medium usually provide corrosion potentials of the order of -1020 to -1080 mV, and therefore less favorable than those of the coatings according to the invention.
- the resistance to atmospheric corrosion was also estimated by observing the percentage of the surface of the corroded sample after 20 cycles of exposure, as defined above.
- Tribology tests were carried out on the coating formed with HC 12 / HC 16 as compared to electrogalvanized coating. The result is reported on the figure 5 which shows the coefficient of friction of the coating as a function of the contact pressure for the two coatings.
- the tribological behavior of uncoated electrogalvanized steel degrades substantially with the increase of the contact pressure, which is not the case with the coating according to the invention which constantly has a low coefficient of friction, of the same order of magnitude. larger than coatings formed with single fatty acids.
- This coating is well suited to be used as a lubricant during stamping of a steel sheet coated with zinc or zinc alloy.
- the carboxylation coatings obtained using binary mixtures of fatty acids with the composition of the eutectic have at least equal performances, and often superior in all points of view, to those of the coatings obtained at using unique fatty acids in water-solvent medium.
- the HC 12 / HC 16 mixture is the most satisfactory of those tested.
- composition difference (in mol%) with respect to the eutectic x% - y% should not exceed x ⁇ 5% - y ⁇ 5% and preferably x ⁇ 3% - y ⁇ 3%, for binary eutectics or x ⁇ 3%, y ⁇ 3% - z ⁇ 3% for ternary eutectics.
- dispersants it is possible to use especially high molecular weight polyols, carboxylic acid salts such as (meth) acrylic copolymers, polyamide derivatives such as polyamide waxes.
- the optimum for the oxygenated water concentration is between 2 and 8 g / l.
- carboxylation emulsions containing water, the surfactant APG 215 mentioned above and the mixture with the composition of the eutectic HC 12 / HC 16 at 81/19% were prepared.
- emulsion A with a low concentration of APG 215 makes it possible to release the fatty acids more rapidly.
- a coat weight of 1.2 g / m 2 is reached in 5 s, while 10 s are needed to achieve a layer weight comparable with other emulsions.
- contents of APG 215 of 1 to 3% no marked effect of the surfactant concentration is observed.
- the Oxidant concentration also has no very appreciable effect in the explored range.
- the size of the crystals does not seem to be related to the composition of the emulsion. Again, the product of the carboxylation is not well crystallized, and its composition is close to ZnC 12 C 16 .
- the performance of carboxylation coatings formed from eutectics in a water / organic solvent medium is generally greater than that of similar coatings formed by emulsions in a water / surfactant medium.
- the performance of the coatings formed without organic solvent are considered sufficient, for example because the coated products are not intended to remain long in a corrosive atmosphere, it is advantageous to use them because the toxicological risks are less for the manipulators and for the environment.
- their implementation does not require or little control and post-treatment effluents.
- the oxidizing conditions have been obtained using hydrogen peroxide. But, as is known, they could have been obtained with other oxidants, or by the application to the carboxylation bath of an electrical current of the order of intensity, for example, from 10 to 25 mA / cm. 2 .
- the invention is not limited to the examples that have been described.
- the eutectics of the other couples of saturated C 10 -C 18 linear fatty acids would be usable, whether these acids each have an even or odd number of carbon atoms.
- Eutectics of ternary mixtures of such fatty acids can also be used.
- even-numbered fatty acids which is the preferred embodiment of the invention. These even fatty acids are of plant origin and are generally derived from the green products sector, from renewable sources. Odd fatty acids do not exist in nature and must be synthesized. In addition, odd fatty acid eutectics require chemical treatments for their preparation.
- the conversion treatments according to the invention are applicable to other metal surfaces than galvanized steels. They may concern any metal surface susceptible to carboxylation, namely zinc, iron, aluminum, copper, lead and their alloys, aluminized or copper-coated steels.
Landscapes
- Chemical & Material Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Metallurgy (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Chemical Treatment Of Metals (AREA)
- Preventing Corrosion Or Incrustation Of Metals (AREA)
- Coating With Molten Metal (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Priority Applications (24)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT05292773T ATE415504T1 (de) | 2005-12-22 | 2005-12-22 | Behandlungsverfahren der metallischen oberflächen durch carboxilierung, benutzung dieses verfahrens für den zeitweiligen korrosionsschutz und verfahren zur herstellung eines geformten carboxylierten bleches |
| ES05292773T ES2318436T3 (es) | 2005-12-22 | 2005-12-22 | Procedimiento de tratamiento por carboxilacion de superficies metalicas, uso de este procedimiento para la proteccion temporal contra la corrosion y procedimiento de fabricacion de una chapa conformada asi carboxilada. |
| PL05292773T PL1801262T3 (pl) | 2005-12-22 | 2005-12-22 | Obróbka przez karboksylowanie powierzchni metalowych, zastosowanie tego procesu jako tymczasowej ochrony przeciw korozji oraz sposób wytwarzania blachy kształtowanej powleczonej karboksylowaną powłoką konwersyjną |
| DE602005011317T DE602005011317D1 (de) | 2005-12-22 | 2005-12-22 | Behandlungsverfahren der metallischen Oberflächen durch Carboxilierung, Benutzung dieses Verfahrens für den zeitweiligen Korrosionsschutz und Verfahren zur Herstellung eines geformten carboxylierten Bleches |
| EP05292773A EP1801262B1 (fr) | 2005-12-22 | 2005-12-22 | Procédé de traitement par carboxylatation de surfaces métalliques, utilisation de ce procédé pour la protection temporaire contre la corrosion, et procédé de fabrication d'une tôle mise en forme ainsi carboxylatée |
| EP06847093A EP1963545B1 (fr) | 2005-12-22 | 2006-12-20 | Procede de traitement par carboxylatation de surfaces metalliques, utilisation de ce procede pour la protection temporaire contre la corrosion, et procede de fabrication d'une tole mise en forme ainsi carboxylatee |
| RU2008130099/02A RU2384653C1 (ru) | 2005-12-22 | 2006-12-20 | Способ обработки металлических поверхностей карбоксилированием, применение этого способа для временной защиты против коррозии и способ изготовления деформированного и карбоксилированного листа |
| CA2632928A CA2632928C (fr) | 2005-12-22 | 2006-12-20 | Procede de traitement par carboxylatation de surfaces metalliques, utilisation de ce procede pour la protection temporaire contre la corrosion, et procede de fabrication d'une tole mise en forme ainsi carboxylatee |
| ES06847093T ES2355438T3 (es) | 2005-12-22 | 2006-12-20 | Procedimiento de tratamiento por carblxilación de superficies metálicas, uso de este procedimiento para la protección temporal contra la corrosión y procedimiento de fabricación de una chapa conformada así corboxilada. |
| BRPI0621113-5A BRPI0621113A2 (pt) | 2005-12-22 | 2006-12-20 | processo de conversão por carboxilação de uma superfìcie metálica, processo de proteção temporária contra a corrosão de uma superfìcie metálica e processo de fabricação de uma chapa moldada |
| PL06847093T PL1963545T3 (pl) | 2005-12-22 | 2006-12-20 | Sposób obróbki przez karboksylowanie powierzchni metalowych, zastosowanie tego sposobu celem zapewnienia czasowej ochrony przed korozją oraz sposób wytwarzania blachy kształtowej tak karboksylowanej |
| AT06847093T ATE488618T1 (de) | 2005-12-22 | 2006-12-20 | Behandlungsverfahren der metallischen oberflächen durch carboxilierung, benutzung dieses verfahrens für den zeitweiligen korrosionsschutz und verfahren zur herstellung eines geformten carboxylierten bleches |
| JP2008546519A JP4981062B2 (ja) | 2005-12-22 | 2006-12-20 | 金属表面のカルボキシル化処理方法、腐食に対する一時的な保護を提供するための前記方法の使用、およびこのようにカルボキシル化された成形鋼板を製造する方法 |
| DE602006018357T DE602006018357D1 (de) | 2005-12-22 | 2006-12-20 | Behandlungsverfahren der metallischen oberflächen durch carboxilierung, benutzung dieses verfahrens für den zeitweiligen korrosionsschutz und verfahren zur herstellung eines geformten carboxylierten bleches |
| PCT/FR2006/002814 WO2007077336A1 (fr) | 2005-12-22 | 2006-12-20 | Procede de traitement par carboxylatation de surfaces metalliques, utilisation de ce procede pour la protection temporaire contre la corrosion, et procede de fabrication d'une tole mise en forme ainsi carboxylatee |
| UAA200808303A UA86726C2 (ru) | 2005-12-22 | 2006-12-20 | Способ модифицирования поверхностей металлов, способ временной защиты от коррозии и способ изготовления формируемых изделий |
| MX2008007702A MX2008007702A (es) | 2005-12-22 | 2006-12-20 | Procedimiento de tratamiento por carboxilacion de superficies metalicas, uso de este procedimiento para la proteccion temporal contra la corrosion y procedimiento de fabricacion de una chapa formada de esta manera carboxilada. |
| KR1020087016185A KR101033913B1 (ko) | 2005-12-22 | 2006-12-20 | 카복실화에 의한 금속 표면 처리방법, 부식에 대한 일시적보호를 위한 상기 방법의 용도 및 이와 같이 카복실화된성형 금속 시트의 제조방법 |
| CN200680052258XA CN101448974B (zh) | 2005-12-22 | 2006-12-20 | 金属表面的羧化处理方法、这种方法用于临时性防腐蚀保护的用途以及如此羧化的成型板材的生产方法 |
| TW095148238A TWI376428B (en) | 2005-12-22 | 2006-12-21 | Method for treating metal surfaces by carboxylation, use of the method for temporary protection against corrosion, and method for manufacturing a shaped metal sheet thus carboxylated |
| ARP060105769A AR058727A1 (es) | 2005-12-22 | 2006-12-26 | Procedimiento de tratamiento por carboxilatacion de superficies metalicas, procedimiento para la proteccion temporaria contra la corrosion y procedimiento de fabricacion de una chapa formada carboxilatada |
| ZA200805274A ZA200805274B (en) | 2005-12-22 | 2008-06-18 | Method for the carboxylation treatment of metal surfaces, use of said method in order to provide temporary protection against corrosion and method for producing shaped sheet metal thus carboxylated |
| MA31067A MA30081B1 (fr) | 2005-12-22 | 2008-06-20 | Procede de traitement par carboxylation de surfaces metalliques, utilisation de ce procede pour la protection temporaire contre la corrosion, et procede de fabrication d'une tole mise en forme ainsi carboxylatee |
| US12/421,253 US8273189B2 (en) | 2005-12-22 | 2009-04-09 | Method for treating metal surfaces by carboxylation, use of the method for temporary protection against corrosion, and method for manufacturing a shaped metal sheet thus carboxylated |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP05292773A EP1801262B1 (fr) | 2005-12-22 | 2005-12-22 | Procédé de traitement par carboxylatation de surfaces métalliques, utilisation de ce procédé pour la protection temporaire contre la corrosion, et procédé de fabrication d'une tôle mise en forme ainsi carboxylatée |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1801262A1 EP1801262A1 (fr) | 2007-06-27 |
| EP1801262B1 true EP1801262B1 (fr) | 2008-11-26 |
Family
ID=36474726
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05292773A Expired - Lifetime EP1801262B1 (fr) | 2005-12-22 | 2005-12-22 | Procédé de traitement par carboxylatation de surfaces métalliques, utilisation de ce procédé pour la protection temporaire contre la corrosion, et procédé de fabrication d'une tôle mise en forme ainsi carboxylatée |
| EP06847093A Not-in-force EP1963545B1 (fr) | 2005-12-22 | 2006-12-20 | Procede de traitement par carboxylatation de surfaces metalliques, utilisation de ce procede pour la protection temporaire contre la corrosion, et procede de fabrication d'une tole mise en forme ainsi carboxylatee |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP06847093A Not-in-force EP1963545B1 (fr) | 2005-12-22 | 2006-12-20 | Procede de traitement par carboxylatation de surfaces metalliques, utilisation de ce procede pour la protection temporaire contre la corrosion, et procede de fabrication d'une tole mise en forme ainsi carboxylatee |
Country Status (19)
| Country | Link |
|---|---|
| US (1) | US8273189B2 (pl) |
| EP (2) | EP1801262B1 (pl) |
| JP (1) | JP4981062B2 (pl) |
| KR (1) | KR101033913B1 (pl) |
| CN (1) | CN101448974B (pl) |
| AR (1) | AR058727A1 (pl) |
| AT (2) | ATE415504T1 (pl) |
| BR (1) | BRPI0621113A2 (pl) |
| CA (1) | CA2632928C (pl) |
| DE (2) | DE602005011317D1 (pl) |
| ES (2) | ES2318436T3 (pl) |
| MA (1) | MA30081B1 (pl) |
| MX (1) | MX2008007702A (pl) |
| PL (2) | PL1801262T3 (pl) |
| RU (1) | RU2384653C1 (pl) |
| TW (1) | TWI376428B (pl) |
| UA (1) | UA86726C2 (pl) |
| WO (1) | WO2007077336A1 (pl) |
| ZA (1) | ZA200805274B (pl) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103422085A (zh) * | 2012-05-22 | 2013-12-04 | 广州市泓硕环保科技有限公司 | 一种改善铁或铝基材料对涂料附着力的处理方法及组合物 |
| FR3000103B1 (fr) | 2012-12-21 | 2015-04-03 | Total Raffinage Marketing | Composition lubrifiante a base d'ether de polyglycerol |
| BR112017022024A2 (pt) | 2015-04-15 | 2018-07-03 | Henkel Ag & Co. Kgaa | revestimentos protetores de corrosão fina incorporando polímeros de poliamidoamina |
| CN113106434B (zh) * | 2021-04-12 | 2022-01-04 | 南昌大学 | 一种环保型铝合金化学氧化液及化学氧化方法 |
| CN119374995B (zh) * | 2024-12-30 | 2025-03-18 | 河北燕山钢铁集团有限公司 | 一种冷轧金属材料金相试样制备方法 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB792813A (en) * | 1955-08-22 | 1958-04-02 | American Chem Paint Co | Improvements in or relating to the production of shaped metal articles |
| US4373050A (en) * | 1966-06-01 | 1983-02-08 | Amchem Products, Inc. | Process and composition for coating metals |
| BR7203791D0 (pt) * | 1971-06-14 | 1973-05-10 | Amchem Prod | Composicoes de revestimento e processo para aplica las a uma superficie metalica |
| ATE81528T1 (de) * | 1987-07-29 | 1992-10-15 | Ecoform Umformtechnik Gmbh | Schmiermittel fuer die kaltumformung metallischer werkstoffe und verfahren zu seiner herstellung. |
| GB2268512B (en) * | 1990-03-13 | 1994-09-28 | Henkel Corp | Compositions and processes for conditioning the surface of formed metal articles |
| JP3267853B2 (ja) * | 1996-01-12 | 2002-03-25 | ユシロ化学工業株式会社 | 水溶性潤滑剤組成物及び金属腐食防止方法 |
| FR2767079B1 (fr) * | 1997-08-11 | 1999-10-29 | Lorraine Laminage | Procede de traitement de surfaces de toles metalliques pour ameliorer leur aptitude au collage, a l'emboutissage et au degraissage |
| TW574353B (en) * | 2000-01-17 | 2004-02-01 | Nihon Parkerizing | Agents, liquid compositions and process for hydrophilization |
| FR2822852B1 (fr) | 2001-03-27 | 2003-12-12 | Usinor | Procede de traitement par carboxylatation de surfaces metalliques |
| RU2215766C2 (ru) * | 2001-08-07 | 2003-11-10 | Андреева Ольга Георгиевна | Состав для нанесения защитной молекулярной пленки |
| JP2005068473A (ja) * | 2003-08-21 | 2005-03-17 | Chubu Kiresuto Kk | 表面処理を施した銅または銅合金製品、銅または銅合金の表面処理方法、該表面処理に用いる表面処理剤、並びに該表面処理に用いる表面処理剤キット |
| US7223299B2 (en) * | 2003-09-02 | 2007-05-29 | Atotech Deutschland Gmbh | Composition and process for improving the adhesion of a siccative organic coating compositions to metal substrates |
-
2005
- 2005-12-22 EP EP05292773A patent/EP1801262B1/fr not_active Expired - Lifetime
- 2005-12-22 AT AT05292773T patent/ATE415504T1/de active
- 2005-12-22 ES ES05292773T patent/ES2318436T3/es not_active Expired - Lifetime
- 2005-12-22 PL PL05292773T patent/PL1801262T3/pl unknown
- 2005-12-22 DE DE602005011317T patent/DE602005011317D1/de not_active Expired - Lifetime
-
2006
- 2006-12-20 EP EP06847093A patent/EP1963545B1/fr not_active Not-in-force
- 2006-12-20 DE DE602006018357T patent/DE602006018357D1/de active Active
- 2006-12-20 BR BRPI0621113-5A patent/BRPI0621113A2/pt not_active IP Right Cessation
- 2006-12-20 AT AT06847093T patent/ATE488618T1/de active
- 2006-12-20 PL PL06847093T patent/PL1963545T3/pl unknown
- 2006-12-20 MX MX2008007702A patent/MX2008007702A/es active IP Right Grant
- 2006-12-20 RU RU2008130099/02A patent/RU2384653C1/ru not_active IP Right Cessation
- 2006-12-20 ES ES06847093T patent/ES2355438T3/es active Active
- 2006-12-20 KR KR1020087016185A patent/KR101033913B1/ko not_active Expired - Fee Related
- 2006-12-20 WO PCT/FR2006/002814 patent/WO2007077336A1/fr not_active Ceased
- 2006-12-20 CA CA2632928A patent/CA2632928C/fr not_active Expired - Fee Related
- 2006-12-20 JP JP2008546519A patent/JP4981062B2/ja not_active Expired - Fee Related
- 2006-12-20 CN CN200680052258XA patent/CN101448974B/zh not_active Expired - Fee Related
- 2006-12-20 UA UAA200808303A patent/UA86726C2/ru unknown
- 2006-12-21 TW TW095148238A patent/TWI376428B/zh not_active IP Right Cessation
- 2006-12-26 AR ARP060105769A patent/AR058727A1/es unknown
-
2008
- 2008-06-18 ZA ZA200805274A patent/ZA200805274B/xx unknown
- 2008-06-20 MA MA31067A patent/MA30081B1/fr unknown
-
2009
- 2009-04-09 US US12/421,253 patent/US8273189B2/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| PL1963545T3 (pl) | 2011-07-29 |
| EP1963545B1 (fr) | 2010-11-17 |
| US20090242079A1 (en) | 2009-10-01 |
| MX2008007702A (es) | 2008-09-12 |
| EP1963545A1 (fr) | 2008-09-03 |
| AR058727A1 (es) | 2008-02-20 |
| CA2632928C (fr) | 2011-08-09 |
| TW200728499A (en) | 2007-08-01 |
| EP1801262A1 (fr) | 2007-06-27 |
| ATE415504T1 (de) | 2008-12-15 |
| ATE488618T1 (de) | 2010-12-15 |
| TWI376428B (en) | 2012-11-11 |
| JP4981062B2 (ja) | 2012-07-18 |
| BRPI0621113A2 (pt) | 2011-11-29 |
| DE602005011317D1 (de) | 2009-01-08 |
| KR20080088596A (ko) | 2008-10-02 |
| ZA200805274B (en) | 2009-06-24 |
| ES2355438T3 (es) | 2011-03-25 |
| JP2009520879A (ja) | 2009-05-28 |
| PL1801262T3 (pl) | 2009-05-29 |
| WO2007077336A1 (fr) | 2007-07-12 |
| KR101033913B1 (ko) | 2011-05-11 |
| DE602006018357D1 (de) | 2010-12-30 |
| UA86726C2 (ru) | 2009-05-12 |
| US8273189B2 (en) | 2012-09-25 |
| CN101448974A (zh) | 2009-06-03 |
| RU2384653C1 (ru) | 2010-03-20 |
| CA2632928A1 (fr) | 2007-07-12 |
| MA30081B1 (fr) | 2008-12-01 |
| CN101448974B (zh) | 2011-09-14 |
| RU2008130099A (ru) | 2010-01-27 |
| ES2318436T3 (es) | 2009-05-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3017076B1 (fr) | Procede de traitement d'une tole pour reduire son noircissement ou son ternissement lors de son stockage et tole traitee par tel procede | |
| CA2864109A1 (fr) | Procede de traitement de surface de pieces en alliage d'aluminium ou de magnesium | |
| EP3250730A1 (fr) | Procédé de préparation d'une tôle revêtue comprenant l'application d'une solution aqueuse comprenant un aminoacide et utilisation associée pour améliorer la résistance à la corrosion | |
| FR3082528A1 (fr) | Composition aqueuse et procede de traitement de surface d'une piece en alliage d'aluminium mettant en œuvre une telle composition | |
| EP1801262B1 (fr) | Procédé de traitement par carboxylatation de surfaces métalliques, utilisation de ce procédé pour la protection temporaire contre la corrosion, et procédé de fabrication d'une tôle mise en forme ainsi carboxylatée | |
| EP1373597B1 (fr) | Procede de traitement par carboxylatation de surfaces metalliques | |
| CA2975360C (fr) | Traitement anti-corrosif d'une tole par solution d'acides amines | |
| EP0102284B1 (fr) | Solution et procédé pour la conversion chimique de substrats métalliques | |
| EP0517586B1 (fr) | Procédé pour former un revêtement anticorrosif à base de copolymère sur une surface métallique | |
| EP1252367B1 (fr) | Procede d'oxalatation de la surface zinguee d'une tole | |
| EP0298827A1 (fr) | Solution et procédé de phosphatation mixte | |
| EP3198054A1 (fr) | Traitement de surface de substrats métalliques | |
| WO2005038083A1 (fr) | Procede de traitement par sulfuration de pieces en alliage ferreux |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK YU |
|
| 17P | Request for examination filed |
Effective date: 20071201 |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: ARCELORMITTAL FRANCE Owner name: TOTAL FRANCE |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: TOTAL RAFFINAGE MARKETING Owner name: ARCELORMITTAL FRANCE |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: FRENCH |
|
| REF | Corresponds to: |
Ref document number: 602005011317 Country of ref document: DE Date of ref document: 20090108 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: RO Ref legal event code: EPE |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20090400542 Country of ref document: GR |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081126 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2318436 Country of ref document: ES Kind code of ref document: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081126 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081126 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20090326 |
|
| REG | Reference to a national code |
Ref country code: PL Ref legal event code: T3 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20090226 Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20081231 Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081126 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081126 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081126 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20090427 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| REG | Reference to a national code |
Ref country code: HU Ref legal event code: AG4A Ref document number: E005799 Country of ref document: HU |
|
| 26N | No opposition filed |
Effective date: 20090827 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081126 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091231 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091231 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: RO Payment date: 20101129 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20101123 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: ML Ref document number: 20090400542 Country of ref document: GR Effective date: 20120704 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120704 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 20121126 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: HU Payment date: 20121126 Year of fee payment: 8 Ref country code: CZ Payment date: 20121122 Year of fee payment: 8 Ref country code: DE Payment date: 20121207 Year of fee payment: 8 Ref country code: FI Payment date: 20121122 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20121219 Year of fee payment: 8 Ref country code: SK Payment date: 20121121 Year of fee payment: 8 Ref country code: IT Payment date: 20121213 Year of fee payment: 8 Ref country code: ES Payment date: 20121219 Year of fee payment: 8 Ref country code: PL Payment date: 20121219 Year of fee payment: 8 Ref country code: SE Payment date: 20121214 Year of fee payment: 8 Ref country code: TR Payment date: 20121219 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20121203 Year of fee payment: 8 Ref country code: NL Payment date: 20121122 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20121219 Year of fee payment: 8 Ref country code: FR Payment date: 20130114 Year of fee payment: 8 |
|
| BERE | Be: lapsed |
Owner name: ARCELORMITTAL FRANCE Effective date: 20131231 Owner name: TOTAL RAFFINAGE MARKETING Effective date: 20131231 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602005011317 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20140701 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: EUG |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 415504 Country of ref document: AT Kind code of ref document: T Effective date: 20131222 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20131222 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131223 Ref country code: RO Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: MM4A Ref document number: E 5240 Country of ref document: SK Effective date: 20131222 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602005011317 Country of ref document: DE Effective date: 20140701 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20140829 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140701 Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140701 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131223 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131231 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 Ref country code: SK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 |
|
| REG | Reference to a national code |
Ref country code: PL Ref legal event code: LAPE |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20150709 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131223 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131222 |