EP1335722B1 - Derivés indole-sulfoniques pour le traitement de troubles du snc - Google Patents
Derivés indole-sulfoniques pour le traitement de troubles du snc Download PDFInfo
- Publication number
- EP1335722B1 EP1335722B1 EP01997304A EP01997304A EP1335722B1 EP 1335722 B1 EP1335722 B1 EP 1335722B1 EP 01997304 A EP01997304 A EP 01997304A EP 01997304 A EP01997304 A EP 01997304A EP 1335722 B1 EP1335722 B1 EP 1335722B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- indole
- chloro
- piperazin
- chlorobenzenesulfonyl
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 238000011282 treatment Methods 0.000 title claims abstract description 21
- 238000000034 method Methods 0.000 claims abstract description 43
- 238000002360 preparation method Methods 0.000 claims abstract description 21
- 150000001875 compounds Chemical class 0.000 claims description 130
- 150000003839 salts Chemical class 0.000 claims description 28
- -1 C1-6alkoxyC1-6alkoxy Chemical group 0.000 claims description 24
- 229910052739 hydrogen Inorganic materials 0.000 claims description 15
- 229910052757 nitrogen Inorganic materials 0.000 claims description 15
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 12
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 10
- 125000001072 heteroaryl group Chemical group 0.000 claims description 9
- 125000001041 indolyl group Chemical group 0.000 claims description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 9
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 9
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 8
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Chemical group BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 8
- 229910052794 bromium Inorganic materials 0.000 claims description 8
- 239000000460 chlorine Substances 0.000 claims description 8
- 229910052736 halogen Inorganic materials 0.000 claims description 8
- 150000002367 halogens Chemical group 0.000 claims description 8
- 125000004193 piperazinyl group Chemical group 0.000 claims description 8
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 7
- 239000005864 Sulphur Chemical group 0.000 claims description 7
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 7
- 229910052801 chlorine Inorganic materials 0.000 claims description 7
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 7
- 125000005842 heteroatom Chemical group 0.000 claims description 7
- 239000001257 hydrogen Substances 0.000 claims description 7
- 125000001624 naphthyl group Chemical group 0.000 claims description 7
- 229910052760 oxygen Chemical group 0.000 claims description 7
- 239000001301 oxygen Chemical group 0.000 claims description 7
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 claims description 7
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 6
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 6
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 6
- 125000005960 1,4-diazepanyl group Chemical group 0.000 claims description 5
- 125000004793 2,2,2-trifluoroethoxy group Chemical group FC(CO*)(F)F 0.000 claims description 5
- 125000003341 7 membered heterocyclic group Chemical group 0.000 claims description 5
- 125000000217 alkyl group Chemical group 0.000 claims description 5
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 5
- 125000006239 protecting group Chemical group 0.000 claims description 5
- 208000006096 Attention Deficit Disorder with Hyperactivity Diseases 0.000 claims description 4
- 208000036864 Attention deficit/hyperactivity disease Diseases 0.000 claims description 4
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 4
- 239000008194 pharmaceutical composition Substances 0.000 claims description 4
- 208000024827 Alzheimer disease Diseases 0.000 claims description 3
- 208000019901 Anxiety disease Diseases 0.000 claims description 3
- 208000026139 Memory disease Diseases 0.000 claims description 3
- 230000007000 age related cognitive decline Effects 0.000 claims description 3
- 230000036506 anxiety Effects 0.000 claims description 3
- 125000002618 bicyclic heterocycle group Chemical group 0.000 claims description 3
- 208000010877 cognitive disease Diseases 0.000 claims description 3
- 230000001149 cognitive effect Effects 0.000 claims description 3
- 230000008878 coupling Effects 0.000 claims description 3
- 238000010168 coupling process Methods 0.000 claims description 3
- 238000005859 coupling reaction Methods 0.000 claims description 3
- 239000003814 drug Substances 0.000 claims description 3
- 125000005843 halogen group Chemical group 0.000 claims description 3
- 208000027061 mild cognitive impairment Diseases 0.000 claims description 3
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 3
- 201000000980 schizophrenia Diseases 0.000 claims description 3
- WTVXIBRMWGUIMI-UHFFFAOYSA-N trifluoro($l^{1}-oxidanylsulfonyl)methane Chemical group [O]S(=O)(=O)C(F)(F)F WTVXIBRMWGUIMI-UHFFFAOYSA-N 0.000 claims description 3
- 125000004454 (C1-C6) alkoxycarbonyl group Chemical group 0.000 claims description 2
- 125000004890 (C1-C6) alkylamino group Chemical group 0.000 claims description 2
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 claims description 2
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 2
- CMLZQBIEVUEKBH-ZDUSSCGKSA-N 5-chloro-1-(3-chlorophenyl)sulfonyl-4-[(3s)-3-methylpiperazin-1-yl]indole Chemical compound C1CN[C@@H](C)CN1C1=C(Cl)C=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 CMLZQBIEVUEKBH-ZDUSSCGKSA-N 0.000 claims description 2
- 239000003937 drug carrier Substances 0.000 claims description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 150000004885 piperazines Chemical group 0.000 claims description 2
- IAOGIUYLCUOQQA-UHFFFAOYSA-N 5-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 IAOGIUYLCUOQQA-UHFFFAOYSA-N 0.000 claims 2
- JBGSBTWMZFFAMS-UHFFFAOYSA-N 1-(2-bromophenyl)sulfonyl-5-chloro-4-piperazin-1-ylindole Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C(=CC=CC=3)Br)C=CC2=C1N1CCNCC1 JBGSBTWMZFFAMS-UHFFFAOYSA-N 0.000 claims 1
- FHVSPMZBCRDYNJ-UHFFFAOYSA-N 1-(3-bromophenyl)sulfonyl-5-chloro-4-piperazin-1-ylindole Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C=C(Br)C=CC=3)C=CC2=C1N1CCNCC1 FHVSPMZBCRDYNJ-UHFFFAOYSA-N 0.000 claims 1
- UHNQCIMNUOOBKH-UHFFFAOYSA-N 1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole-5-carbonitrile Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=CC=C(C(N4CCNCC4)=C3C=C2)C#N)=C1 UHNQCIMNUOOBKH-UHFFFAOYSA-N 0.000 claims 1
- BACXNNZPTRPOBE-UHFFFAOYSA-N 1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole-7-carbonitrile Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=C(C#N)C=CC(=C3C=C2)N2CCNCC2)=C1 BACXNNZPTRPOBE-UHFFFAOYSA-N 0.000 claims 1
- LNOAWMRNRLHSQE-UHFFFAOYSA-N 1-(benzenesulfonyl)-5-chloro-4-piperazin-1-ylindole Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C=CC=CC=3)C=CC2=C1N1CCNCC1 LNOAWMRNRLHSQE-UHFFFAOYSA-N 0.000 claims 1
- JUDHAQDVLAMRDF-UHFFFAOYSA-N 3,5-dichloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound C=12C(Cl)=CN(S(=O)(=O)C=3C=C(Cl)C=CC=3)C2=CC=C(Cl)C=1N1CCNCC1 JUDHAQDVLAMRDF-UHFFFAOYSA-N 0.000 claims 1
- UEBZCZBLGRXLNI-UHFFFAOYSA-N 3-(5-chloro-4-piperazin-1-ylindol-1-yl)sulfonylbenzonitrile Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C=C(C=CC=3)C#N)C=CC2=C1N1CCNCC1 UEBZCZBLGRXLNI-UHFFFAOYSA-N 0.000 claims 1
- JQVJNMUSHFGIKK-UHFFFAOYSA-N 3-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound C=12C(Cl)=CN(S(=O)(=O)C=3C=C(Cl)C=CC=3)C2=CC=CC=1N1CCNCC1 JQVJNMUSHFGIKK-UHFFFAOYSA-N 0.000 claims 1
- PFUNHIUMWXXZFZ-UHFFFAOYSA-N 5,7-dichloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=C(Cl)C=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 PFUNHIUMWXXZFZ-UHFFFAOYSA-N 0.000 claims 1
- LRRSSRBVSLJELN-UHFFFAOYSA-N 5-bromo-2-(5-chloro-4-piperazin-1-ylindol-1-yl)sulfonylbenzonitrile Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C(=CC(Br)=CC=3)C#N)C=CC2=C1N1CCNCC1 LRRSSRBVSLJELN-UHFFFAOYSA-N 0.000 claims 1
- SOAKKJXXLGTXKS-UHFFFAOYSA-N 5-chloro-1-(2,5-difluorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound FC1=CC=C(F)C(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 SOAKKJXXLGTXKS-UHFFFAOYSA-N 0.000 claims 1
- ZVCMMJUNXXHZKA-UHFFFAOYSA-N 5-chloro-1-(2,6-dichlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C(=CC=CC=3Cl)Cl)C=CC2=C1N1CCNCC1 ZVCMMJUNXXHZKA-UHFFFAOYSA-N 0.000 claims 1
- XGSJFSTWFVCSJX-UHFFFAOYSA-N 5-chloro-1-(2,6-difluorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound FC1=CC=CC(F)=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 XGSJFSTWFVCSJX-UHFFFAOYSA-N 0.000 claims 1
- NPQVMTFUOLJIOP-UHFFFAOYSA-N 5-chloro-1-(2-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=CC=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 NPQVMTFUOLJIOP-UHFFFAOYSA-N 0.000 claims 1
- KZTDWKHSRMXEJU-UHFFFAOYSA-N 5-chloro-1-(3,5-dichloro-2-methoxyphenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound COC1=C(Cl)C=C(Cl)C=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 KZTDWKHSRMXEJU-UHFFFAOYSA-N 0.000 claims 1
- GBYSRFIPDOENBL-UHFFFAOYSA-N 5-chloro-1-(3,5-dichlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC(Cl)=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 GBYSRFIPDOENBL-UHFFFAOYSA-N 0.000 claims 1
- XQTVHIVCOXSHNT-UHFFFAOYSA-N 5-chloro-1-(3-chlorophenyl)sulfonyl-4-(1,4-diazepan-1-yl)indole Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCCC4)=C3C=C2)=C1 XQTVHIVCOXSHNT-UHFFFAOYSA-N 0.000 claims 1
- CMLZQBIEVUEKBH-CYBMUJFWSA-N 5-chloro-1-(3-chlorophenyl)sulfonyl-4-[(3r)-3-methylpiperazin-1-yl]indole Chemical compound C1CN[C@H](C)CN1C1=C(Cl)C=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 CMLZQBIEVUEKBH-CYBMUJFWSA-N 0.000 claims 1
- FXFREJZOVPDACV-UHFFFAOYSA-N 5-chloro-1-(3-methylphenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound CC1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 FXFREJZOVPDACV-UHFFFAOYSA-N 0.000 claims 1
- WVOCARZSOYXWIZ-UHFFFAOYSA-N 5-chloro-1-(4-fluorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound C1=CC(F)=CC=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 WVOCARZSOYXWIZ-UHFFFAOYSA-N 0.000 claims 1
- ZGXQWBKTMYXZPG-UHFFFAOYSA-N 5-chloro-1-naphthalen-1-ylsulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C4=CC=CC=C4C=CC=3)C=CC2=C1N1CCNCC1 ZGXQWBKTMYXZPG-UHFFFAOYSA-N 0.000 claims 1
- XHQNNCMNVAMRHZ-UHFFFAOYSA-N 5-chloro-1-naphthalen-2-ylsulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3C=C4C=CC=CC4=CC=3)C=CC2=C1N1CCNCC1 XHQNNCMNVAMRHZ-UHFFFAOYSA-N 0.000 claims 1
- XWEXGSBUDIXVNZ-UHFFFAOYSA-N 5-chloro-4-piperazin-1-yl-1-(1,3,5-trimethylpyrazol-4-yl)sulfonylindole Chemical compound CC1=NN(C)C(C)=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 XWEXGSBUDIXVNZ-UHFFFAOYSA-N 0.000 claims 1
- FTFDDINXLNNJHI-UHFFFAOYSA-N 5-chloro-4-piperazin-1-yl-1-[3-(trifluoromethyl)phenyl]sulfonylindole Chemical compound FC(F)(F)C1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 FTFDDINXLNNJHI-UHFFFAOYSA-N 0.000 claims 1
- YKJAYWYRGOVBDE-UHFFFAOYSA-N 5-chloro-4-piperazin-1-yl-1-pyridin-2-ylsulfonylindole Chemical compound ClC1=CC=C2N(S(=O)(=O)C=3N=CC=CC=3)C=CC2=C1N1CCNCC1 YKJAYWYRGOVBDE-UHFFFAOYSA-N 0.000 claims 1
- NCCITMOIKYJTCT-UHFFFAOYSA-N 6-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=CC(Cl)=CC(=C3C=C2)N2CCNCC2)=C1 NCCITMOIKYJTCT-UHFFFAOYSA-N 0.000 claims 1
- DCTVKLQQPFXPSC-UHFFFAOYSA-N 7-bromo-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=C(Br)C=CC(=C3C=C2)N2CCNCC2)=C1 DCTVKLQQPFXPSC-UHFFFAOYSA-N 0.000 claims 1
- MINAGCAGXAFKFT-UHFFFAOYSA-N 7-chloro-1-(3-chlorophenyl)sulfonyl-4-(1,4-diazepan-1-yl)indole Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=C(Cl)C=CC(=C3C=C2)N2CCNCCC2)=C1 MINAGCAGXAFKFT-UHFFFAOYSA-N 0.000 claims 1
- BJWONGXVVAAEEW-UHFFFAOYSA-N 7-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=C(Cl)C=CC(=C3C=C2)N2CCNCC2)=C1 BJWONGXVVAAEEW-UHFFFAOYSA-N 0.000 claims 1
- 208000020401 Depressive disease Diseases 0.000 claims 1
- 239000000203 mixture Substances 0.000 abstract description 72
- 208000015114 central nervous system disease Diseases 0.000 abstract description 3
- 150000002475 indoles Chemical class 0.000 abstract description 3
- 230000000144 pharmacologic effect Effects 0.000 abstract description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 130
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 79
- 239000000243 solution Substances 0.000 description 65
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 54
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 51
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 38
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 37
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 31
- 238000004440 column chromatography Methods 0.000 description 31
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 30
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 26
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 24
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 24
- 239000007787 solid Substances 0.000 description 23
- 239000000741 silica gel Substances 0.000 description 22
- 229910002027 silica gel Inorganic materials 0.000 description 22
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 21
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 20
- 239000003921 oil Substances 0.000 description 20
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 20
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 19
- 239000012267 brine Substances 0.000 description 19
- 238000003756 stirring Methods 0.000 description 19
- 239000000725 suspension Substances 0.000 description 19
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 18
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 18
- 229920006395 saturated elastomer Polymers 0.000 description 18
- JRNVZBWKYDBUCA-UHFFFAOYSA-N N-chlorosuccinimide Chemical compound ClN1C(=O)CCC1=O JRNVZBWKYDBUCA-UHFFFAOYSA-N 0.000 description 16
- 230000015572 biosynthetic process Effects 0.000 description 16
- ZFKPTSLREZFGAO-UHFFFAOYSA-N tert-butyl 4-(5-chloro-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=C(Cl)C=CC2=C1C=CN2 ZFKPTSLREZFGAO-UHFFFAOYSA-N 0.000 description 16
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 15
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 15
- 150000002148 esters Chemical class 0.000 description 14
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 14
- 239000011541 reaction mixture Substances 0.000 description 13
- 229910052786 argon Inorganic materials 0.000 description 12
- 238000010511 deprotection reaction Methods 0.000 description 12
- 239000012044 organic layer Substances 0.000 description 12
- 238000000746 purification Methods 0.000 description 12
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 12
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 11
- 238000001704 evaporation Methods 0.000 description 11
- 230000008020 evaporation Effects 0.000 description 11
- 239000000463 material Substances 0.000 description 11
- 239000011347 resin Substances 0.000 description 11
- 229920005989 resin Polymers 0.000 description 11
- VBHPUAZPKSHQCK-UHFFFAOYSA-N tert-butyl 4-[5-chloro-1-(3-chlorophenyl)sulfonylindol-4-yl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=C(Cl)C=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 VBHPUAZPKSHQCK-UHFFFAOYSA-N 0.000 description 11
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 10
- 239000012074 organic phase Substances 0.000 description 10
- 235000017557 sodium bicarbonate Nutrition 0.000 description 10
- 239000012312 sodium hydride Substances 0.000 description 10
- 229910000104 sodium hydride Inorganic materials 0.000 description 10
- 239000002904 solvent Substances 0.000 description 10
- 238000003786 synthesis reaction Methods 0.000 description 10
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 9
- 238000002953 preparative HPLC Methods 0.000 description 9
- OINWZUJVEXUHCC-UHFFFAOYSA-N 3-chlorobenzenesulfonyl chloride Chemical compound ClC1=CC=CC(S(Cl)(=O)=O)=C1 OINWZUJVEXUHCC-UHFFFAOYSA-N 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 8
- QCQCHGYLTSGIGX-GHXANHINSA-N 4-[[(3ar,5ar,5br,7ar,9s,11ar,11br,13as)-5a,5b,8,8,11a-pentamethyl-3a-[(5-methylpyridine-3-carbonyl)amino]-2-oxo-1-propan-2-yl-4,5,6,7,7a,9,10,11,11b,12,13,13a-dodecahydro-3h-cyclopenta[a]chrysen-9-yl]oxy]-2,2-dimethyl-4-oxobutanoic acid Chemical compound N([C@@]12CC[C@@]3(C)[C@]4(C)CC[C@H]5C(C)(C)[C@@H](OC(=O)CC(C)(C)C(O)=O)CC[C@]5(C)[C@H]4CC[C@@H]3C1=C(C(C2)=O)C(C)C)C(=O)C1=CN=CC(C)=C1 QCQCHGYLTSGIGX-GHXANHINSA-N 0.000 description 7
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 7
- 208000035475 disorder Diseases 0.000 description 7
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 7
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 7
- 239000003208 petroleum Substances 0.000 description 7
- FVYWXNHMPHKXQS-UHFFFAOYSA-N tert-butyl 4-[1-(3-chlorophenyl)sulfonylindol-4-yl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 FVYWXNHMPHKXQS-UHFFFAOYSA-N 0.000 description 7
- 239000003981 vehicle Substances 0.000 description 7
- 239000003039 volatile agent Substances 0.000 description 7
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 238000001816 cooling Methods 0.000 description 6
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 6
- 235000019345 sodium thiosulphate Nutrition 0.000 description 6
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 6
- OEAMZRWFIQSVJQ-UHFFFAOYSA-N tert-butyl 4-(1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=CC2=C1C=CN2 OEAMZRWFIQSVJQ-UHFFFAOYSA-N 0.000 description 6
- CTZQQLPXRKSALV-UHFFFAOYSA-N tert-butyl 4-(7-bromo-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=C(Br)C2=C1C=CN2 CTZQQLPXRKSALV-UHFFFAOYSA-N 0.000 description 6
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 6
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 6
- BFXFOOXEHHXWEY-UHFFFAOYSA-N 4-(4-benzylpiperazin-1-yl)-1-[(5-chloro-3-methyl-1-benzothiophen-2-yl)sulfonyl]indole Chemical compound S1C2=CC=C(Cl)C=C2C(C)=C1S(=O)(=O)N(C1=CC=C2)C=CC1=C2N(CC1)CCN1CC1=CC=CC=C1 BFXFOOXEHHXWEY-UHFFFAOYSA-N 0.000 description 5
- UKKDPMZQOAIVAD-UHFFFAOYSA-N 4-bromo-2-cyanobenzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=C(Br)C=C1C#N UKKDPMZQOAIVAD-UHFFFAOYSA-N 0.000 description 5
- XTYSXGIKUCWFNC-UHFFFAOYSA-N 4-chloro-2-(trifluoromethoxy)benzenesulfonyl chloride Chemical compound FC(F)(F)OC1=CC(Cl)=CC=C1S(Cl)(=O)=O XTYSXGIKUCWFNC-UHFFFAOYSA-N 0.000 description 5
- HNQLFSIYLOLPLG-UHFFFAOYSA-N 5-methoxy-1-(4-methylphenyl)sulfonyl-4-nitroindole Chemical compound C1=CC2=C([N+]([O-])=O)C(OC)=CC=C2N1S(=O)(=O)C1=CC=C(C)C=C1 HNQLFSIYLOLPLG-UHFFFAOYSA-N 0.000 description 5
- NERFNHBZJXXFGY-UHFFFAOYSA-N [4-[(4-methylphenyl)methoxy]phenyl]methanol Chemical group C1=CC(C)=CC=C1COC1=CC=C(CO)C=C1 NERFNHBZJXXFGY-UHFFFAOYSA-N 0.000 description 5
- 239000000706 filtrate Substances 0.000 description 5
- 239000006260 foam Substances 0.000 description 5
- 235000010193 gold Nutrition 0.000 description 5
- 239000004333 gold (food color) Substances 0.000 description 5
- 125000000623 heterocyclic group Chemical group 0.000 description 5
- 239000004179 indigotine Substances 0.000 description 5
- 235000012738 indigotine Nutrition 0.000 description 5
- 239000010410 layer Substances 0.000 description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 5
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 102000005962 receptors Human genes 0.000 description 5
- 108020003175 receptors Proteins 0.000 description 5
- 239000011369 resultant mixture Substances 0.000 description 5
- MTOBQHFPPWMIMQ-UHFFFAOYSA-N tert-butyl 4-(1h-indol-4-yl)-1,4-diazepane-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCCN1C1=CC=CC2=C1C=CN2 MTOBQHFPPWMIMQ-UHFFFAOYSA-N 0.000 description 5
- HELDIERIJDINPF-UHFFFAOYSA-N tert-butyl 4-(3-methyl-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C=12C(C)=CNC2=CC=CC=1N1CCN(C(=O)OC(C)(C)C)CC1 HELDIERIJDINPF-UHFFFAOYSA-N 0.000 description 5
- OUKVYWPPZYKPFT-UHFFFAOYSA-N tert-butyl 4-(6-chloro-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC(Cl)=CC2=C1C=CN2 OUKVYWPPZYKPFT-UHFFFAOYSA-N 0.000 description 5
- CTUVFDVKGGTUOC-UHFFFAOYSA-N tert-butyl 4-[1-(3-chlorophenyl)sulfonyl-7-cyanoindol-4-yl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=C(C#N)C2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 CTUVFDVKGGTUOC-UHFFFAOYSA-N 0.000 description 5
- BSMWNBACTOSKGS-UHFFFAOYSA-N tert-butyl 4-[3-methyl-1-tri(propan-2-yl)silylindol-4-yl]piperazine-1-carboxylate Chemical compound C1=CC=C2N([Si](C(C)C)(C(C)C)C(C)C)C=C(C)C2=C1N1CCN(C(=O)OC(C)(C)C)CC1 BSMWNBACTOSKGS-UHFFFAOYSA-N 0.000 description 5
- JLVOSNUBRZHLNW-UHFFFAOYSA-N tert-butyl 4-[4-[(2-methylpropan-2-yl)oxycarbonyl]-1,4-diazepan-1-yl]indole-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCCN1C1=CC=CC2=C1C=CN2C(=O)OC(C)(C)C JLVOSNUBRZHLNW-UHFFFAOYSA-N 0.000 description 5
- ZAJRKTXYJQFAQW-UHFFFAOYSA-N tert-butyl 4-[6-chloro-1-(3-chlorophenyl)sulfonylindol-4-yl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC(Cl)=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 ZAJRKTXYJQFAQW-UHFFFAOYSA-N 0.000 description 5
- IEASWPUEHCBDKO-UHFFFAOYSA-N tert-butyl 4-[7-bromo-1-(3-chlorophenyl)sulfonylindol-4-yl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=C(Br)C2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 IEASWPUEHCBDKO-UHFFFAOYSA-N 0.000 description 5
- OLWQSPISEHDEAR-UHFFFAOYSA-N 5-chloro-1-(3,5-dichloro-2-methoxyphenyl)sulfonyl-4-piperazin-1-ylindole;2,2,2-trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.COC1=C(Cl)C=C(Cl)C=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 OLWQSPISEHDEAR-UHFFFAOYSA-N 0.000 description 4
- OTSNHBVJFREMOF-UHFFFAOYSA-N 5-chloro-1-naphthalen-2-ylsulfonyl-4-piperazin-1-ylindole;2,2,2-trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.ClC1=CC=C2N(S(=O)(=O)C=3C=C4C=CC=CC4=CC=3)C=CC2=C1N1CCNCC1 OTSNHBVJFREMOF-UHFFFAOYSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 4
- 239000003875 Wang resin Substances 0.000 description 4
- 229960000583 acetic acid Drugs 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 125000003118 aryl group Chemical group 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 4
- 229960004756 ethanol Drugs 0.000 description 4
- 239000000284 extract Substances 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 239000002480 mineral oil Substances 0.000 description 4
- 235000010446 mineral oil Nutrition 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 125000004076 pyridyl group Chemical group 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- 229940124530 sulfonamide Drugs 0.000 description 4
- 150000003456 sulfonamides Chemical class 0.000 description 4
- YSUUMEYVEBHJRZ-UHFFFAOYSA-N tert-butyl 4-[1-(3-chlorophenyl)sulfonyl-5-cyanoindol-4-yl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=C(C#N)C=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 YSUUMEYVEBHJRZ-UHFFFAOYSA-N 0.000 description 4
- RZPFWTIIYOBHMB-UHFFFAOYSA-N tert-butyl 4-[1-(3-chlorophenyl)sulfonylindol-4-yl]-1,4-diazepane-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCCN1C1=CC=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 RZPFWTIIYOBHMB-UHFFFAOYSA-N 0.000 description 4
- HBYHSIGLNBVLKP-UHFFFAOYSA-N tert-butyl 4-[3-[(dimethylamino)methyl]-1-tri(propan-2-yl)silylindol-4-yl]piperazine-1-carboxylate Chemical compound C1=CC=C2N([Si](C(C)C)(C(C)C)C(C)C)C=C(CN(C)C)C2=C1N1CCN(C(=O)OC(C)(C)C)CC1 HBYHSIGLNBVLKP-UHFFFAOYSA-N 0.000 description 4
- VDZOOKBUILJEDG-UHFFFAOYSA-M tetrabutylammonium hydroxide Chemical compound [OH-].CCCC[N+](CCCC)(CCCC)CCCC VDZOOKBUILJEDG-UHFFFAOYSA-M 0.000 description 4
- 239000004108 vegetable carbon Substances 0.000 description 4
- 235000012712 vegetable carbon Nutrition 0.000 description 4
- HEERNCIUTDNAOD-UHFFFAOYSA-N 1-[(5-chloro-3-methyl-1-benzothiophen-2-yl)sulfonyl]-4-piperazin-1-ylindole;oxalic acid Chemical compound OC(=O)C(O)=O.S1C2=CC=C(Cl)C=C2C(C)=C1S(=O)(=O)N(C1=CC=C2)C=CC1=C2N1CCNCC1 HEERNCIUTDNAOD-UHFFFAOYSA-N 0.000 description 3
- GUGXBGRCDWRZCS-UHFFFAOYSA-N 2,3,5-trichlorobenzenesulfonyl chloride Chemical compound ClC1=CC(Cl)=C(Cl)C(S(Cl)(=O)=O)=C1 GUGXBGRCDWRZCS-UHFFFAOYSA-N 0.000 description 3
- MCSXGCZMEPXKIW-UHFFFAOYSA-N 3-hydroxy-4-[(4-methyl-2-nitrophenyl)diazenyl]-N-(3-nitrophenyl)naphthalene-2-carboxamide Chemical compound Cc1ccc(N=Nc2c(O)c(cc3ccccc23)C(=O)Nc2cccc(c2)[N+]([O-])=O)c(c1)[N+]([O-])=O MCSXGCZMEPXKIW-UHFFFAOYSA-N 0.000 description 3
- LNBPBQCQMJPTQS-UHFFFAOYSA-N 4-(4-methylpiperazin-1-yl)-1h-indole Chemical compound C1CN(C)CCN1C1=CC=CC2=C1C=CN2 LNBPBQCQMJPTQS-UHFFFAOYSA-N 0.000 description 3
- CNDVIIPWSVCCIZ-UHFFFAOYSA-N 5-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;2,2,2-trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.ClC1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 CNDVIIPWSVCCIZ-UHFFFAOYSA-N 0.000 description 3
- WQESCKJRCSIUGY-UHFFFAOYSA-N 6-chloro-1h-indol-4-amine Chemical compound NC1=CC(Cl)=CC2=C1C=CN2 WQESCKJRCSIUGY-UHFFFAOYSA-N 0.000 description 3
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 3
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- MUALRAIOVNYAIW-UHFFFAOYSA-N binap Chemical compound C1=CC=CC=C1P(C=1C(=C2C=CC=CC2=CC=1)C=1C2=CC=CC=C2C=CC=1P(C=1C=CC=CC=1)C=1C=CC=CC=1)C1=CC=CC=C1 MUALRAIOVNYAIW-UHFFFAOYSA-N 0.000 description 3
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 3
- 229910000024 caesium carbonate Inorganic materials 0.000 description 3
- VTYYLEPIZMXCLO-UHFFFAOYSA-L calcium carbonate Substances [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 3
- 235000010216 calcium carbonate Nutrition 0.000 description 3
- 150000001721 carbon Chemical group 0.000 description 3
- 238000011097 chromatography purification Methods 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 3
- 125000002971 oxazolyl group Chemical group 0.000 description 3
- YJVFFLUZDVXJQI-UHFFFAOYSA-L palladium(ii) acetate Chemical compound [Pd+2].CC([O-])=O.CC([O-])=O YJVFFLUZDVXJQI-UHFFFAOYSA-L 0.000 description 3
- 238000011321 prophylaxis Methods 0.000 description 3
- 125000003226 pyrazolyl group Chemical group 0.000 description 3
- 239000004248 saffron Substances 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- VIOPRZBSGLPGJQ-UHFFFAOYSA-N tert-butyl 4-(7-chloro-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=C(Cl)C2=C1C=CN2 VIOPRZBSGLPGJQ-UHFFFAOYSA-N 0.000 description 3
- ZYVJUKUGSVFWAH-UHFFFAOYSA-N tert-butyl 4-[1-(3-chlorophenyl)sulfonyl-3-methylindol-4-yl]piperazine-1-carboxylate Chemical compound C=12C(C)=CN(S(=O)(=O)C=3C=C(Cl)C=CC=3)C2=CC=CC=1N1CCN(C(=O)OC(C)(C)C)CC1 ZYVJUKUGSVFWAH-UHFFFAOYSA-N 0.000 description 3
- IMBLWFGDUGKHOL-UHFFFAOYSA-N tert-butyl 4-[1-(3-chlorophenyl)sulfonyl-5-methoxyindol-4-yl]piperazine-1-carboxylate Chemical compound COC1=CC=C2N(S(=O)(=O)C=3C=C(Cl)C=CC=3)C=CC2=C1N1CCN(C(=O)OC(C)(C)C)CC1 IMBLWFGDUGKHOL-UHFFFAOYSA-N 0.000 description 3
- ZILAHWVMZRPTEY-UHFFFAOYSA-N tert-butyl 4-[7-chloro-1-(3-chlorophenyl)sulfonylindol-4-yl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=C(Cl)C2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 ZILAHWVMZRPTEY-UHFFFAOYSA-N 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- 0 *S([n](ccc1c(cc2)N3CC[N+]CC3)c1c2Cl)(=O)=O Chemical compound *S([n](ccc1c(cc2)N3CC[N+]CC3)c1c2Cl)(=O)=O 0.000 description 2
- LEBUUXDRJSXXQV-UHFFFAOYSA-N 2-chloro-3,5-difluorobenzenesulfonyl chloride Chemical compound FC1=CC(F)=C(Cl)C(S(Cl)(=O)=O)=C1 LEBUUXDRJSXXQV-UHFFFAOYSA-N 0.000 description 2
- LUNUNJFSHKSXGQ-UHFFFAOYSA-N 4-Aminoindole Chemical class NC1=CC=CC2=C1C=CN2 LUNUNJFSHKSXGQ-UHFFFAOYSA-N 0.000 description 2
- YZKSXUIOKWQABW-UHFFFAOYSA-N 4-piperazin-1-yl-1h-indole Chemical compound C1CNCCN1C1=CC=CC2=C1C=CN2 YZKSXUIOKWQABW-UHFFFAOYSA-N 0.000 description 2
- OERSHGZPYWYISS-UHFFFAOYSA-N 5,7-dichloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=CC(S(=O)(=O)N2C3=C(Cl)C=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 OERSHGZPYWYISS-UHFFFAOYSA-N 0.000 description 2
- FPPGTUQPALMUQU-UHFFFAOYSA-N 5-chloro-1-(2,6-dichlorophenyl)sulfonyl-4-piperazin-1-ylindole;2,2,2-trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.ClC1=CC=C2N(S(=O)(=O)C=3C(=CC=CC=3Cl)Cl)C=CC2=C1N1CCNCC1 FPPGTUQPALMUQU-UHFFFAOYSA-N 0.000 description 2
- ZKNDRTCWQYPHET-UHFFFAOYSA-N 5-chloro-1-(3,5-dichlorophenyl)sulfonyl-4-piperazin-1-ylindole;2,2,2-trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.ClC1=CC(Cl)=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 ZKNDRTCWQYPHET-UHFFFAOYSA-N 0.000 description 2
- CVICEEPAFUYBJG-UHFFFAOYSA-N 5-chloro-2,2-difluoro-1,3-benzodioxole Chemical group C1=C(Cl)C=C2OC(F)(F)OC2=C1 CVICEEPAFUYBJG-UHFFFAOYSA-N 0.000 description 2
- UKQHGVZROAGWTC-UHFFFAOYSA-N 5-chloro-4-piperazin-1-yl-1-pyridin-2-ylsulfonylindole;hydrochloride Chemical compound Cl.ClC1=CC=C2N(S(=O)(=O)C=3N=CC=CC=3)C=CC2=C1N1CCNCC1 UKQHGVZROAGWTC-UHFFFAOYSA-N 0.000 description 2
- FGINWIULZMWURR-UHFFFAOYSA-N 7-chloro-1-(3,5-dichlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC(Cl)=CC(S(=O)(=O)N2C3=C(Cl)C=CC(=C3C=C2)N2CCNCC2)=C1 FGINWIULZMWURR-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical class [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 229910021592 Copper(II) chloride Inorganic materials 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 2
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical compound O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 239000001654 beetroot red Substances 0.000 description 2
- 235000012677 beetroot red Nutrition 0.000 description 2
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 2
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 2
- 239000004126 brilliant black BN Substances 0.000 description 2
- 235000012709 brilliant black BN Nutrition 0.000 description 2
- 239000001678 brown HT Substances 0.000 description 2
- 235000012670 brown HT Nutrition 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- KQIADDMXRMTWHZ-UHFFFAOYSA-N chloro-tri(propan-2-yl)silane Chemical compound CC(C)[Si](Cl)(C(C)C)C(C)C KQIADDMXRMTWHZ-UHFFFAOYSA-N 0.000 description 2
- ZPUCINDJVBIVPJ-LJISPDSOSA-N cocaine Chemical compound O([C@H]1C[C@@H]2CC[C@@H](N2C)[C@H]1C(=O)OC)C(=O)C1=CC=CC=C1 ZPUCINDJVBIVPJ-LJISPDSOSA-N 0.000 description 2
- 239000013058 crude material Substances 0.000 description 2
- 239000012259 ether extract Substances 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000012458 free base Substances 0.000 description 2
- 239000012362 glacial acetic acid Substances 0.000 description 2
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 2
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 2
- 239000012442 inert solvent Substances 0.000 description 2
- 208000002551 irritable bowel syndrome Diseases 0.000 description 2
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 2
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 2
- GBMDVOWEEQVZKZ-UHFFFAOYSA-N methanol;hydrate Chemical compound O.OC GBMDVOWEEQVZKZ-UHFFFAOYSA-N 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 229910017604 nitric acid Inorganic materials 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- WRIKHQLVHPKCJU-UHFFFAOYSA-N sodium bis(trimethylsilyl)amide Chemical compound C[Si](C)(C)N([Na])[Si](C)(C)C WRIKHQLVHPKCJU-UHFFFAOYSA-N 0.000 description 2
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- YBBRCQOCSYXUOC-UHFFFAOYSA-N sulfuryl dichloride Chemical compound ClS(Cl)(=O)=O YBBRCQOCSYXUOC-UHFFFAOYSA-N 0.000 description 2
- XCDOWLDSMCYDCB-UHFFFAOYSA-N tert-butyl 4-(5-methoxy-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound COC1=CC=C2NC=CC2=C1N1CCN(C(=O)OC(C)(C)C)CC1 XCDOWLDSMCYDCB-UHFFFAOYSA-N 0.000 description 2
- YJAWZICAYTVBIZ-UHFFFAOYSA-N tert-butyl 4-(7-methoxy-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C1=2C=CNC=2C(OC)=CC=C1N1CCN(C(=O)OC(C)(C)C)CC1 YJAWZICAYTVBIZ-UHFFFAOYSA-N 0.000 description 2
- OZZYFAQEQFBTRO-UHFFFAOYSA-N tert-butyl 4-[1-(3-chlorophenyl)sulfonyl-2-methylindol-4-yl]piperazine-1-carboxylate Chemical compound CC1=CC2=C(N3CCN(CC3)C(=O)OC(C)(C)C)C=CC=C2N1S(=O)(=O)C1=CC=CC(Cl)=C1 OZZYFAQEQFBTRO-UHFFFAOYSA-N 0.000 description 2
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- 125000000335 thiazolyl group Chemical group 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- SNICXCGAKADSCV-JTQLQIEISA-N (-)-Nicotine Chemical compound CN1CCC[C@H]1C1=CC=CN=C1 SNICXCGAKADSCV-JTQLQIEISA-N 0.000 description 1
- HFVMEOPYDLEHBR-UHFFFAOYSA-N (2-fluorophenyl)-phenylmethanol Chemical compound C=1C=CC=C(F)C=1C(O)C1=CC=CC=C1 HFVMEOPYDLEHBR-UHFFFAOYSA-N 0.000 description 1
- JOMNTHCQHJPVAZ-RXMQYKEDSA-N (2r)-2-methylpiperazine Chemical compound C[C@@H]1CNCCN1 JOMNTHCQHJPVAZ-RXMQYKEDSA-N 0.000 description 1
- JOMNTHCQHJPVAZ-YFKPBYRVSA-N (2s)-2-methylpiperazine Chemical compound C[C@H]1CNCCN1 JOMNTHCQHJPVAZ-YFKPBYRVSA-N 0.000 description 1
- MAYZWDRUFKUGGP-VIFPVBQESA-N (3s)-1-[5-tert-butyl-3-[(1-methyltetrazol-5-yl)methyl]triazolo[4,5-d]pyrimidin-7-yl]pyrrolidin-3-ol Chemical compound CN1N=NN=C1CN1C2=NC(C(C)(C)C)=NC(N3C[C@@H](O)CC3)=C2N=N1 MAYZWDRUFKUGGP-VIFPVBQESA-N 0.000 description 1
- NXLNNXIXOYSCMB-UHFFFAOYSA-N (4-nitrophenyl) carbonochloridate Chemical compound [O-][N+](=O)C1=CC=C(OC(Cl)=O)C=C1 NXLNNXIXOYSCMB-UHFFFAOYSA-N 0.000 description 1
- ZGYIXVSQHOKQRZ-COIATFDQSA-N (e)-n-[4-[3-chloro-4-(pyridin-2-ylmethoxy)anilino]-3-cyano-7-[(3s)-oxolan-3-yl]oxyquinolin-6-yl]-4-(dimethylamino)but-2-enamide Chemical compound N#CC1=CN=C2C=C(O[C@@H]3COCC3)C(NC(=O)/C=C/CN(C)C)=CC2=C1NC(C=C1Cl)=CC=C1OCC1=CC=CC=N1 ZGYIXVSQHOKQRZ-COIATFDQSA-N 0.000 description 1
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 1
- KASJZXHXXNEULX-UHFFFAOYSA-N 1,5,6,7-tetrahydroindol-4-one Chemical compound O=C1CCCC2=C1C=CN2 KASJZXHXXNEULX-UHFFFAOYSA-N 0.000 description 1
- APWRZPQBPCAXFP-UHFFFAOYSA-N 1-(1-oxo-2H-isoquinolin-5-yl)-5-(trifluoromethyl)-N-[2-(trifluoromethyl)pyridin-4-yl]pyrazole-4-carboxamide Chemical compound O=C1NC=CC2=C(C=CC=C12)N1N=CC(=C1C(F)(F)F)C(=O)NC1=CC(=NC=C1)C(F)(F)F APWRZPQBPCAXFP-UHFFFAOYSA-N 0.000 description 1
- VQNQRZSQBXCWHP-UHFFFAOYSA-N 1-(2-bromophenyl)sulfonyl-5-chloro-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=C2N(S(=O)(=O)C=3C(=CC=CC=3)Br)C=CC2=C1N1CCNCC1 VQNQRZSQBXCWHP-UHFFFAOYSA-N 0.000 description 1
- MQSGMUAWESCQHU-UHFFFAOYSA-N 1-(3-bromophenyl)sulfonyl-5-chloro-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=C2N(S(=O)(=O)C=3C=C(Br)C=CC=3)C=CC2=C1N1CCNCC1 MQSGMUAWESCQHU-UHFFFAOYSA-N 0.000 description 1
- HOVJBPKYXLHHRR-UHFFFAOYSA-N 1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole-7-carbonitrile;hydrochloride Chemical compound Cl.ClC1=CC=CC(S(=O)(=O)N2C3=C(C#N)C=CC(=C3C=C2)N2CCNCC2)=C1 HOVJBPKYXLHHRR-UHFFFAOYSA-N 0.000 description 1
- MADREAMCRBZQQC-UHFFFAOYSA-N 1-(3-chlorophenyl)sulfonyl-6,7-dihydro-5h-indol-4-one Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=C(C(CCC3)=O)C=C2)=C1 MADREAMCRBZQQC-UHFFFAOYSA-N 0.000 description 1
- RWJLLFCQKGWGEA-UHFFFAOYSA-N 1-(benzenesulfonyl)-5-chloro-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=C2N(S(=O)(=O)C=3C=CC=CC=3)C=CC2=C1N1CCNCC1 RWJLLFCQKGWGEA-UHFFFAOYSA-N 0.000 description 1
- PVOAHINGSUIXLS-UHFFFAOYSA-N 1-Methylpiperazine Chemical compound CN1CCNCC1 PVOAHINGSUIXLS-UHFFFAOYSA-N 0.000 description 1
- SWMZRWDZOSSJIW-UHFFFAOYSA-N 1-[4-bromo-2-(trifluoromethoxy)phenyl]sulfonyl-5-chloro-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.FC(F)(F)OC1=CC(Br)=CC=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 SWMZRWDZOSSJIW-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- MOTBXEPLFOLWHZ-UHFFFAOYSA-N 2,3,5-trichloroaniline Chemical compound NC1=CC(Cl)=CC(Cl)=C1Cl MOTBXEPLFOLWHZ-UHFFFAOYSA-N 0.000 description 1
- OATYCBHROMXWJO-UHFFFAOYSA-N 2-amino-5-bromobenzonitrile Chemical compound NC1=CC=C(Br)C=C1C#N OATYCBHROMXWJO-UHFFFAOYSA-N 0.000 description 1
- XDHOQZCRADUDRI-UHFFFAOYSA-N 2-chloro-3,5-difluoroaniline Chemical compound NC1=CC(F)=CC(F)=C1Cl XDHOQZCRADUDRI-UHFFFAOYSA-N 0.000 description 1
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 description 1
- VSCBATMPTLKTOV-UHFFFAOYSA-N 2-tert-butylimino-n,n-diethyl-1,3-dimethyl-1,3,2$l^{5}-diazaphosphinan-2-amine Chemical compound CCN(CC)P1(=NC(C)(C)C)N(C)CCCN1C VSCBATMPTLKTOV-UHFFFAOYSA-N 0.000 description 1
- XSKDWODPDJLNED-UHFFFAOYSA-N 3,5-dichloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.C=12C(Cl)=CN(S(=O)(=O)C=3C=C(Cl)C=CC=3)C2=CC=C(Cl)C=1N1CCNCC1 XSKDWODPDJLNED-UHFFFAOYSA-N 0.000 description 1
- RJSQINMKOSOUGT-UHFFFAOYSA-N 3,5-dichlorobenzenesulfonyl chloride Chemical compound ClC1=CC(Cl)=CC(S(Cl)(=O)=O)=C1 RJSQINMKOSOUGT-UHFFFAOYSA-N 0.000 description 1
- NOUOUGDQNNWARY-UHFFFAOYSA-N 3-(5-chloro-4-piperazin-1-ylindol-1-yl)sulfonylbenzonitrile;hydrochloride Chemical compound Cl.ClC1=CC=C2N(S(=O)(=O)C=3C=C(C=CC=3)C#N)C=CC2=C1N1CCNCC1 NOUOUGDQNNWARY-UHFFFAOYSA-N 0.000 description 1
- WNEODWDFDXWOLU-QHCPKHFHSA-N 3-[3-(hydroxymethyl)-4-[1-methyl-5-[[5-[(2s)-2-methyl-4-(oxetan-3-yl)piperazin-1-yl]pyridin-2-yl]amino]-6-oxopyridin-3-yl]pyridin-2-yl]-7,7-dimethyl-1,2,6,8-tetrahydrocyclopenta[3,4]pyrrolo[3,5-b]pyrazin-4-one Chemical compound C([C@@H](N(CC1)C=2C=NC(NC=3C(N(C)C=C(C=3)C=3C(=C(N4C(C5=CC=6CC(C)(C)CC=6N5CC4)=O)N=CC=3)CO)=O)=CC=2)C)N1C1COC1 WNEODWDFDXWOLU-QHCPKHFHSA-N 0.000 description 1
- SRVXSISGYBMIHR-UHFFFAOYSA-N 3-[3-[3-(2-amino-2-oxoethyl)phenyl]-5-chlorophenyl]-3-(5-methyl-1,3-thiazol-2-yl)propanoic acid Chemical compound S1C(C)=CN=C1C(CC(O)=O)C1=CC(Cl)=CC(C=2C=C(CC(N)=O)C=CC=2)=C1 SRVXSISGYBMIHR-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- MWGLHLZQWAHHMA-UHFFFAOYSA-N 3-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.C=12C(Cl)=CN(S(=O)(=O)C=3C=C(Cl)C=CC=3)C2=CC=CC=1N1CCNCC1 MWGLHLZQWAHHMA-UHFFFAOYSA-N 0.000 description 1
- JLZIBIQTDGMAFZ-UHFFFAOYSA-N 4-(4-benzylpiperazin-1-yl)-1h-indole Chemical compound C1CN(C=2C=3C=CNC=3C=CC=2)CCN1CC1=CC=CC=C1 JLZIBIQTDGMAFZ-UHFFFAOYSA-N 0.000 description 1
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 1
- QISOBCMNUJQOJU-UHFFFAOYSA-N 4-bromo-1h-pyrazole-5-carboxylic acid Chemical compound OC(=O)C=1NN=CC=1Br QISOBCMNUJQOJU-UHFFFAOYSA-N 0.000 description 1
- UCFTYLMKCJPWBX-UHFFFAOYSA-N 4-chloro-2-(trifluoromethoxy)aniline Chemical compound NC1=CC=C(Cl)C=C1OC(F)(F)F UCFTYLMKCJPWBX-UHFFFAOYSA-N 0.000 description 1
- NLMQHXUGJIAKTH-UHFFFAOYSA-N 4-hydroxyindole Chemical compound OC1=CC=CC2=C1C=CN2 NLMQHXUGJIAKTH-UHFFFAOYSA-N 0.000 description 1
- ZEZXLZGFTFMUFQ-UHFFFAOYSA-N 5,5-dichloro-1-(3-chlorophenyl)sulfonyl-6,7-dihydroindol-4-one Chemical compound ClC1=CC=CC(S(=O)(=O)N2C3=C(C(C(Cl)(Cl)CC3)=O)C=C2)=C1 ZEZXLZGFTFMUFQ-UHFFFAOYSA-N 0.000 description 1
- IRPVABHDSJVBNZ-RTHVDDQRSA-N 5-[1-(cyclopropylmethyl)-5-[(1R,5S)-3-(oxetan-3-yl)-3-azabicyclo[3.1.0]hexan-6-yl]pyrazol-3-yl]-3-(trifluoromethyl)pyridin-2-amine Chemical compound C1=C(C(F)(F)F)C(N)=NC=C1C1=NN(CC2CC2)C(C2[C@@H]3CN(C[C@@H]32)C2COC2)=C1 IRPVABHDSJVBNZ-RTHVDDQRSA-N 0.000 description 1
- PXRKCOCTEMYUEG-UHFFFAOYSA-N 5-aminoisoindole-1,3-dione Chemical compound NC1=CC=C2C(=O)NC(=O)C2=C1 PXRKCOCTEMYUEG-UHFFFAOYSA-N 0.000 description 1
- FWXCITOZPUNHSK-UHFFFAOYSA-N 5-bromo-2-(5-chloro-4-piperazin-1-ylindol-1-yl)sulfonylbenzonitrile;hydrochloride Chemical compound Cl.ClC1=CC=C2N(S(=O)(=O)C=3C(=CC(Br)=CC=3)C#N)C=CC2=C1N1CCNCC1 FWXCITOZPUNHSK-UHFFFAOYSA-N 0.000 description 1
- YGMCLSFAJTXGJX-UHFFFAOYSA-N 5-chloro-1-(2,5-difluorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.FC1=CC=C(F)C(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 YGMCLSFAJTXGJX-UHFFFAOYSA-N 0.000 description 1
- XSHPQFNCXSEOFL-UHFFFAOYSA-N 5-chloro-1-(2,6-difluorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.FC1=CC=CC(F)=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 XSHPQFNCXSEOFL-UHFFFAOYSA-N 0.000 description 1
- ORTZHJBTFPFSHL-UHFFFAOYSA-N 5-chloro-1-(2-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=CC=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 ORTZHJBTFPFSHL-UHFFFAOYSA-N 0.000 description 1
- AFMBAINXIYTSCB-UHFFFAOYSA-N 5-chloro-1-(3-chlorophenyl)sulfonyl-4-(1,4-diazepan-1-yl)indole;hydrochloride Chemical compound Cl.ClC1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCCC4)=C3C=C2)=C1 AFMBAINXIYTSCB-UHFFFAOYSA-N 0.000 description 1
- ZUQXYIRPWFOKSR-UHFFFAOYSA-N 5-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 ZUQXYIRPWFOKSR-UHFFFAOYSA-N 0.000 description 1
- PBAMYTWQYQDZKK-UHFFFAOYSA-N 5-chloro-1-(3-chlorophenyl)sulfonylindol-4-ol Chemical compound C1=CC=2C(O)=C(Cl)C=CC=2N1S(=O)(=O)C1=CC=CC(Cl)=C1 PBAMYTWQYQDZKK-UHFFFAOYSA-N 0.000 description 1
- PNFBEVDONTVADC-UHFFFAOYSA-N 5-chloro-1-(3-methylphenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.CC1=CC=CC(S(=O)(=O)N2C3=CC=C(Cl)C(N4CCNCC4)=C3C=C2)=C1 PNFBEVDONTVADC-UHFFFAOYSA-N 0.000 description 1
- ZDXIUXIHQMFACR-UHFFFAOYSA-N 5-chloro-1-(4-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.C1=CC(Cl)=CC=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 ZDXIUXIHQMFACR-UHFFFAOYSA-N 0.000 description 1
- QUPWFQXZCVTGQW-UHFFFAOYSA-N 5-chloro-1-[4-chloro-2-(trifluoromethoxy)phenyl]sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.FC(F)(F)OC1=CC(Cl)=CC=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 QUPWFQXZCVTGQW-UHFFFAOYSA-N 0.000 description 1
- DVQPOKFIHHCCLM-UHFFFAOYSA-N 5-chloro-1-naphthalen-1-ylsulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=C2N(S(=O)(=O)C=3C4=CC=CC=C4C=CC=3)C=CC2=C1N1CCNCC1 DVQPOKFIHHCCLM-UHFFFAOYSA-N 0.000 description 1
- KEPIENQOWOJEBQ-UHFFFAOYSA-N 5-chloro-2-methyl-1,3-dinitrobenzene Chemical compound CC1=C([N+]([O-])=O)C=C(Cl)C=C1[N+]([O-])=O KEPIENQOWOJEBQ-UHFFFAOYSA-N 0.000 description 1
- JLTUANWNGKWRQO-UHFFFAOYSA-N 5-chloro-3-methyl-1-benzothiophene-2-sulfonyl chloride Chemical compound C1=C(Cl)C=C2C(C)=C(S(Cl)(=O)=O)SC2=C1 JLTUANWNGKWRQO-UHFFFAOYSA-N 0.000 description 1
- VTMOFCYBJDCWNR-UHFFFAOYSA-N 5-chloro-4-piperazin-1-yl-1-(1,3,5-trimethylpyrazol-4-yl)sulfonylindole;hydrochloride Chemical compound Cl.CC1=NN(C)C(C)=C1S(=O)(=O)N1C2=CC=C(Cl)C(N3CCNCC3)=C2C=C1 VTMOFCYBJDCWNR-UHFFFAOYSA-N 0.000 description 1
- QFSZPNUHXFOHIA-UHFFFAOYSA-N 5-methoxy-1-(4-methylphenyl)sulfonylindol-4-amine Chemical compound C1=CC2=C(N)C(OC)=CC=C2N1S(=O)(=O)C1=CC=C(C)C=C1 QFSZPNUHXFOHIA-UHFFFAOYSA-N 0.000 description 1
- SWJXHHMYYCPWLX-UHFFFAOYSA-N 5-methoxy-1-(4-methylphenyl)sulfonylindole Chemical compound C1=CC2=CC(OC)=CC=C2N1S(=O)(=O)C1=CC=C(C)C=C1 SWJXHHMYYCPWLX-UHFFFAOYSA-N 0.000 description 1
- YTRLGXQPTYRYKK-UHFFFAOYSA-N 5-methoxy-1h-indol-4-amine Chemical compound COC1=CC=C2NC=CC2=C1N YTRLGXQPTYRYKK-UHFFFAOYSA-N 0.000 description 1
- RLEGGEKTUQDHMA-UHFFFAOYSA-N 6-chloro-4-(4-methylpiperazin-1-yl)-1h-indole Chemical compound C1CN(C)CCN1C1=CC(Cl)=CC2=C1C=CN2 RLEGGEKTUQDHMA-UHFFFAOYSA-N 0.000 description 1
- WGYNPEYJDCCEJS-UHFFFAOYSA-N 7-bromo-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=CC(S(=O)(=O)N2C3=C(Br)C=CC(=C3C=C2)N2CCNCC2)=C1 WGYNPEYJDCCEJS-UHFFFAOYSA-N 0.000 description 1
- LYRUJARAXUQUMK-UHFFFAOYSA-N 7-chloro-1-(3-chlorophenyl)sulfonyl-4-(1,4-diazepan-1-yl)indole;hydrochloride Chemical compound Cl.ClC1=CC=CC(S(=O)(=O)N2C3=C(Cl)C=CC(=C3C=C2)N2CCNCCC2)=C1 LYRUJARAXUQUMK-UHFFFAOYSA-N 0.000 description 1
- DPYBOLIVHIACPX-UHFFFAOYSA-N 7-chloro-1-(3-chlorophenyl)sulfonyl-4-piperazin-1-ylindole;hydrochloride Chemical compound Cl.ClC1=CC=CC(S(=O)(=O)N2C3=C(Cl)C=CC(=C3C=C2)N2CCNCC2)=C1 DPYBOLIVHIACPX-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- 244000144730 Amygdalus persica Species 0.000 description 1
- 208000032841 Bulimia Diseases 0.000 description 1
- 206010006550 Bulimia nervosa Diseases 0.000 description 1
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- YSNVKDGEALPJGC-UHFFFAOYSA-N Cc(cc(cc1)F)c1F Chemical compound Cc(cc(cc1)F)c1F YSNVKDGEALPJGC-UHFFFAOYSA-N 0.000 description 1
- RYMMNSVHOKXTNN-UHFFFAOYSA-N Cc1cc(Cl)cc(Cl)c1 Chemical compound Cc1cc(Cl)cc(Cl)c1 RYMMNSVHOKXTNN-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 229910021591 Copper(I) chloride Inorganic materials 0.000 description 1
- 206010013654 Drug abuse Diseases 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 240000007002 Eucalyptus tereticornis Species 0.000 description 1
- 239000004214 Fast Green FCF Substances 0.000 description 1
- 208000019454 Feeding and Eating disease Diseases 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 206010019196 Head injury Diseases 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 208000019695 Migraine disease Diseases 0.000 description 1
- ZSXGLVDWWRXATF-UHFFFAOYSA-N N,N-dimethylformamide dimethyl acetal Chemical compound COC(OC)N(C)C ZSXGLVDWWRXATF-UHFFFAOYSA-N 0.000 description 1
- IDRGFNPZDVBSSE-UHFFFAOYSA-N OCCN1CCN(CC1)c1ccc(Nc2ncc3cccc(-c4cccc(NC(=O)C=C)c4)c3n2)c(F)c1F Chemical compound OCCN1CCN(CC1)c1ccc(Nc2ncc3cccc(-c4cccc(NC(=O)C=C)c4)c3n2)c(F)c1F IDRGFNPZDVBSSE-UHFFFAOYSA-N 0.000 description 1
- 208000021384 Obsessive-Compulsive disease Diseases 0.000 description 1
- 206010033664 Panic attack Diseases 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 235000006040 Prunus persica var persica Nutrition 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric Acid Chemical compound [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- OKJPEAGHQZHRQV-UHFFFAOYSA-N Triiodomethane Natural products IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 1
- 125000002777 acetyl group Chemical class [H]C([H])([H])C(*)=O 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 230000029936 alkylation Effects 0.000 description 1
- 238000005804 alkylation reaction Methods 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 238000010976 amide bond formation reaction Methods 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 239000003708 ampul Substances 0.000 description 1
- 230000003444 anaesthetic effect Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 208000022531 anorexia Diseases 0.000 description 1
- 229940054051 antipsychotic indole derivative Drugs 0.000 description 1
- 239000003420 antiserotonin agent Substances 0.000 description 1
- 125000004391 aryl sulfonyl group Chemical group 0.000 description 1
- 125000004421 aryl sulphonamide group Chemical class 0.000 description 1
- 238000011914 asymmetric synthesis Methods 0.000 description 1
- 208000015802 attention deficit-hyperactivity disease Diseases 0.000 description 1
- 125000003725 azepanyl group Chemical group 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 229940049706 benzodiazepine Drugs 0.000 description 1
- 125000003310 benzodiazepinyl group Chemical class N1N=C(C=CC2=C1C=CC=C2)* 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000004109 brown FK Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001589 carboacyl group Chemical group 0.000 description 1
- 125000002843 carboxylic acid group Chemical group 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- QOPVNWQGBQYBBP-UHFFFAOYSA-N chloroethyl chloroformate Chemical compound CC(Cl)OC(Cl)=O QOPVNWQGBQYBBP-UHFFFAOYSA-N 0.000 description 1
- 239000001752 chlorophylls and chlorophyllins Substances 0.000 description 1
- 235000012698 chlorophylls and chlorophyllins Nutrition 0.000 description 1
- WRJWRGBVPUUDLA-UHFFFAOYSA-N chlorosulfonyl isocyanate Chemical compound ClS(=O)(=O)N=C=O WRJWRGBVPUUDLA-UHFFFAOYSA-N 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 1
- 230000027288 circadian rhythm Effects 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 229960003920 cocaine Drugs 0.000 description 1
- 239000004121 copper complexes of chlorophylls and chlorophyllins Substances 0.000 description 1
- 235000012700 copper complexes of chlorophylls and chlorophyllins Nutrition 0.000 description 1
- OXBLHERUFWYNTN-UHFFFAOYSA-M copper(I) chloride Chemical compound [Cu]Cl OXBLHERUFWYNTN-UHFFFAOYSA-M 0.000 description 1
- DOBRDRYODQBAMW-UHFFFAOYSA-N copper(i) cyanide Chemical compound [Cu+].N#[C-] DOBRDRYODQBAMW-UHFFFAOYSA-N 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 239000010779 crude oil Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 206010061428 decreased appetite Diseases 0.000 description 1
- 125000005959 diazepanyl group Chemical group 0.000 description 1
- 239000002027 dichloromethane extract Substances 0.000 description 1
- GLUUGHFHXGJENI-UHFFFAOYSA-N diethylenediamine Natural products C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000008157 edible vegetable oil Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000007345 electrophilic aromatic substitution reaction Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 206010015037 epilepsy Diseases 0.000 description 1
- VVDUZZGYBOWDSQ-UHFFFAOYSA-M eschenmoser's salt Chemical compound [I-].C[N+](C)=C VVDUZZGYBOWDSQ-UHFFFAOYSA-M 0.000 description 1
- 238000010931 ester hydrolysis Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 239000004120 green S Substances 0.000 description 1
- 235000012701 green S Nutrition 0.000 description 1
- 235000003528 grey gum Nutrition 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 208000003906 hydrocephalus Diseases 0.000 description 1
- 208000013403 hyperactivity Diseases 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 208000035231 inattentive type attention deficit hyperactivity disease Diseases 0.000 description 1
- JFDDFGLNZWNJTK-UHFFFAOYSA-N indole-4-carbaldehyde Chemical compound O=CC1=CC=CC2=C1C=CN2 JFDDFGLNZWNJTK-UHFFFAOYSA-N 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 125000000468 ketone group Chemical group 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- XGZVUEUWXADBQD-UHFFFAOYSA-L lithium carbonate Chemical compound [Li+].[Li+].[O-]C([O-])=O XGZVUEUWXADBQD-UHFFFAOYSA-L 0.000 description 1
- 229910052808 lithium carbonate Inorganic materials 0.000 description 1
- UBJFKNSINUCEAL-UHFFFAOYSA-N lithium;2-methylpropane Chemical compound [Li+].C[C-](C)C UBJFKNSINUCEAL-UHFFFAOYSA-N 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 206010027599 migraine Diseases 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 125000000896 monocarboxylic acid group Chemical group 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical compound C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- 229960002715 nicotine Drugs 0.000 description 1
- SNICXCGAKADSCV-UHFFFAOYSA-N nicotine Natural products CN1CCCC1C1=CC=CN=C1 SNICXCGAKADSCV-UHFFFAOYSA-N 0.000 description 1
- 125000006574 non-aromatic ring group Chemical group 0.000 description 1
- 239000002687 nonaqueous vehicle Substances 0.000 description 1
- 238000007339 nucleophilic aromatic substitution reaction Methods 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 150000003891 oxalate salts Chemical class 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 150000002940 palladium Chemical class 0.000 description 1
- 208000019906 panic disease Diseases 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 239000003444 phase transfer catalyst Substances 0.000 description 1
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 1
- XKJCHHZQLQNZHY-UHFFFAOYSA-N phthalimide Chemical compound C1=CC=C2C(=O)NC(=O)C2=C1 XKJCHHZQLQNZHY-UHFFFAOYSA-N 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- JQJOGAGLBDBMLU-UHFFFAOYSA-N pyridine-2-sulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=N1 JQJOGAGLBDBMLU-UHFFFAOYSA-N 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 239000002464 receptor antagonist Substances 0.000 description 1
- 229940044551 receptor antagonist Drugs 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000006722 reduction reaction Methods 0.000 description 1
- 230000007017 scission Effects 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 239000000952 serotonin receptor agonist Substances 0.000 description 1
- 208000019116 sleep disease Diseases 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 235000010288 sodium nitrite Nutrition 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 230000000707 stereoselective effect Effects 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 239000012258 stirred mixture Substances 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 208000011117 substance-related disease Diseases 0.000 description 1
- 238000006277 sulfonation reaction Methods 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000002278 tabletting lubricant Substances 0.000 description 1
- WDPWEXWMQDRXAL-UHFFFAOYSA-N tert-butyl 1,4-diazepane-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CCCNCC1 WDPWEXWMQDRXAL-UHFFFAOYSA-N 0.000 description 1
- UOUFRTFWWBCVPV-UHFFFAOYSA-N tert-butyl 4-(2,4-dioxo-1H-thieno[3,2-d]pyrimidin-3-yl)piperidine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CCC(CC1)n1c(=O)[nH]c2ccsc2c1=O UOUFRTFWWBCVPV-UHFFFAOYSA-N 0.000 description 1
- HKIBHZQWOOLFIE-UHFFFAOYSA-N tert-butyl 4-(3-chloro-1h-indol-4-yl)piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C1=CC=CC2=C1C(Cl)=CN2 HKIBHZQWOOLFIE-UHFFFAOYSA-N 0.000 description 1
- AOBNKHCADIZMCX-UHFFFAOYSA-N tert-butyl 4-[5-chloro-1-(3-chlorophenyl)sulfonylindol-4-yl]-1,4-diazepane-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCCN1C1=C(Cl)C=CC2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 AOBNKHCADIZMCX-UHFFFAOYSA-N 0.000 description 1
- FMKSDQTTZCNWCG-UHFFFAOYSA-N tert-butyl 4-[7-chloro-1-(3-chlorophenyl)sulfonylindol-4-yl]-1,4-diazepane-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCCN1C1=CC=C(Cl)C2=C1C=CN2S(=O)(=O)C1=CC=CC(Cl)=C1 FMKSDQTTZCNWCG-UHFFFAOYSA-N 0.000 description 1
- 125000004853 tetrahydropyridinyl group Chemical group N1(CCCC=C1)* 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 150000003555 thioacetals Chemical class 0.000 description 1
- YONPGGFAJWQGJC-UHFFFAOYSA-K titanium(iii) chloride Chemical compound Cl[Ti](Cl)Cl YONPGGFAJWQGJC-UHFFFAOYSA-K 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 125000002088 tosyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])S(*)(=O)=O 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 125000002221 trityl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C([*])(C1=C(C(=C(C(=C1[H])[H])[H])[H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 238000009827 uniform distribution Methods 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
Claims (10)
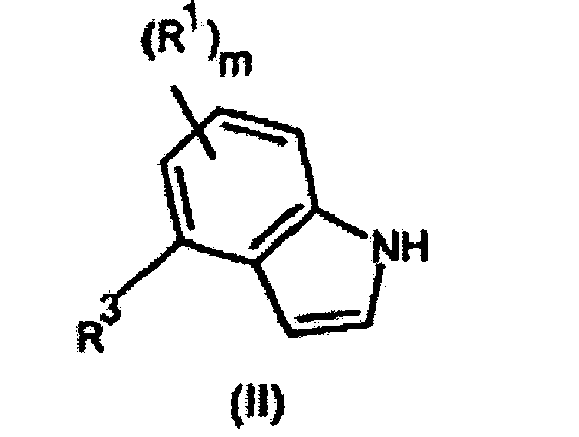
- Composé de formule (I) ou un de ses sels pharmaceutiquement acceptables :
- Composé suivant la revendication 1, dans lequel R3 représente un noyau pipérazine non substitué.
- Composé suivant la revendication 1 ou 2, dans lequel m est égal à 1 et R1 représente un groupe chloro en position 5 du noyau indole.
- Composé suivant l'une quelconque des revendications 1 à 3, dans lequel m est égal à 2 et R1 représente un groupe 5,7-dichloro.
- Composé suivant l'une quelconque des revendications 1 à 4, dans lequel P représente un groupe phényle.
- Composé suivant l'une quelconque des revendications 1 à 5, dans lequel n est égal à 1 et R2 représente le chlore.
- Composé suivant la revendication 1, qui est le 5-chloro-1-(3-chloro-benzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(3,5-dichlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(2,6-dichlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(naphtalène-2-sulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(3,5-dichloro-2-méthoxybenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(3-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
1-(benzènesulfonyl)-5-chloro-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(2,6-difluorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
1-(2-bromobenzènesulfonyl)-5-chloro-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(2-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(3-trifluorométhyl-benzènesulfonyl)-.4-pipérazine-1-yl-lh-indole ;
5-chloro-1-(3-méthylbenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
1-(3-bromobenzènesulfonyl)-5-chloro-4-pipérazine-1-yl-1H-indole ;
1-(4-bromo-2-trifluorométhoxybenzènesulfonyl)-5-chloro-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(3-cyanobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(1-naphtalènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(2,5-difluorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(4-fluorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(4-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(1,3,5-triméthyl-1H-pyrazole-4-sulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(pyridine-2-sulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(2,5-diméthylbenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
3-chloro-1-(3-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
7-chloro-1-(3-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
7-chloro-1-(3,5-dichlorobenzènesulfonyl).-4-pipérazine-1-yl-1H-indole ;
1-benzènesulfonyl-7-chloro-4-pipérazine-1-yl-1H-indole ;
1-(4-bromo-2-trifluorométhoxybenzènesulfonyl)-7-chloro-4-pipérazine-1-yl-1H-indole ;
7-bromo-1-(3-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
7-cyano-1-(3-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
1-(3-chlorobenzènesulfonyl)-3,5-dichloro-4-pipérazine-1-yl-1H-indole ;
1-(3-chlorobenzènesulfonyl)-5,7-dichloro-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(3-chlorobenzènesulfonyl)-4-[1,4]diazépan-1-yl-1H-indole ;
5-chloro-1-(3-chlorobenzènesulfonyl)-4-((R)-3-méthylpipérazine-1-yl)-1H-indole ;
5-chloro-1-(3-chlorobenzènesulfonyl)-4-((S)-3-méthylpipérazine-1-yl)-1H-indole ;
1-(3-chlorobenzènesulfonyl)-5-cyano-4-pipérazine-1-yl-1H-indole ;
6-chloro-1-(3-chlorobenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
5-chloro-1-(4-chloro-2-trifluorométhoxybenzènesulfonyl)-4-pipérazine-1-yl-1H-indole ;
1-(4-bromo-2-cyanobenzènesulfonyl)-5-chloro-4-pipérazine-1-yl-1H-indole ;
7-chloro-1-(3-chlorobenzènesulfonyl)-4-[1,4]diazépan-1-yl-1H-indole ;
ou un de ses sels pharmaceutiquement acceptables. - Composition pharmaceutique comprenant un composé de formule (I) répondant à la définition figurant dans l'une quelconque des revendications 1 à 7 ou un de ses sels pharmaceutiquement acceptables et un support ou excipient pharmaceutiquement acceptable.
- Utilisation d'un composé de formule (I) répondant à la définition figurant dans l'une quelconque des revendications 1 à 7 dans la production d'un médicament destiné au traitement de la dépression, de l'anxiété, de troubles cognitifs de la mémoire, de la maladie d'Alzheimer, du déclin cognitif dû à l'âge, de l'altération cognitive faible, du syndrome de déficit d'attention/ hyperactivité et/ou de la schizophrénie.
- Procédé pour la préparation d'un composé de formule (I) répondant à la définition figurant dans la revendication 1 ou d'un de ses sels pharmaceutiquement acceptables, procédé qui comprend(a) le couplage d'un composé de formule (II) :
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0028708 | 2000-11-24 | ||
| GB0028708A GB0028708D0 (en) | 2000-11-24 | 2000-11-24 | Novel compounds |
| GB0113517A GB0113517D0 (en) | 2001-06-04 | 2001-06-04 | Novel Compounds |
| GB0113517 | 2001-06-04 | ||
| PCT/EP2001/013411 WO2002041889A2 (fr) | 2000-11-24 | 2001-11-16 | Composes utiles pour le traitement de troubles du snc |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1335722A2 EP1335722A2 (fr) | 2003-08-20 |
| EP1335722B1 true EP1335722B1 (fr) | 2006-08-30 |
Family
ID=26245327
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP01997304A Expired - Lifetime EP1335722B1 (fr) | 2000-11-24 | 2001-11-16 | Derivés indole-sulfoniques pour le traitement de troubles du snc |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20050176705A1 (fr) |
| EP (1) | EP1335722B1 (fr) |
| JP (1) | JP2004517072A (fr) |
| AT (1) | ATE337780T1 (fr) |
| AU (1) | AU2002220715A1 (fr) |
| DE (1) | DE60122767T2 (fr) |
| ES (1) | ES2271124T3 (fr) |
| WO (1) | WO2002041889A2 (fr) |
Families Citing this family (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1326830A1 (fr) | 2000-10-20 | 2003-07-16 | Biovitrum Ab | N1-(benzensulfonyl)indoles substitues en 2, 3, 4 ou 5 et leur utilisation a des fins de traitement |
| BR0210411A (pt) * | 2001-06-15 | 2004-08-17 | Hoffmann La Roche | Derivados 4-piperazinilindol com afinidade para o receptor 5-ht6 |
| AU2003208711A1 (en) * | 2002-02-12 | 2003-09-04 | Akzo Nobel N.V. | 1-arylsulfonyl-3-substituted indole and indoline derivatives useful in the treatment of central nervous system disorders |
| PL209872B1 (pl) | 2002-03-27 | 2011-10-31 | Glaxo Group Ltd | Pochodna chinolinowa, sposób jej wytwarzania i jej zastosowanie oraz zawierająca ją farmaceutyczna kompozycja |
| MXPA04012122A (es) * | 2002-06-05 | 2005-04-19 | Hoffmann La Roche | Derivados de 1-sulfonil-4-aminoalcoxi indol como moduladores del receptor 5-hidroxitriptamina 6 (5-5-ht6) para tratamiento de trastornos del sistema nervioso central (snc). |
| MXPA05002836A (es) * | 2002-09-17 | 2005-05-27 | Hoffmann La Roche | Indoles 2,4-sustituidos y su uso como moduladores 5-ht6. |
| PL376479A1 (en) * | 2002-10-18 | 2005-12-27 | F.Hoffmann-La Roche Ag | 4-piperazinyl benzenesulfonyl indoles with 5-ht6 receptor affinity |
| KR100818508B1 (ko) * | 2002-11-28 | 2008-03-31 | 수벤 라이프 사이언시스 리미티드 | 세로토닌 수용체 친화력을 가지는 엔-아릴술포닐-3-치환인돌, 그 제조방법 및 그를 포함하는 약제학적 조성물 |
| TWI289141B (en) * | 2003-03-11 | 2007-11-01 | Hoffmann La Roche F. Ag. | Quinolinone derivatives and uses thereof |
| DE602004000260T2 (de) | 2003-07-22 | 2006-08-24 | Arena Pharmaceuticals, Inc., San Diego | Diaryl- und arylheteroarylharnstoffderivate als modulatoren des 5-ht2a-serotoninrezeptors, die sich zur prophylaxe und behandlung von damit im zusammenhang stehenden erkrankungen eignen |
| SE0302760D0 (sv) * | 2003-10-20 | 2003-10-20 | Biovitrum Ab | New compounds |
| JP3997255B2 (ja) | 2004-02-13 | 2007-10-24 | ワーナー−ランバート カンパニー リミテッド ライアビリティー カンパニー | アンドロゲンレセプターモジュレーター |
| US7507860B2 (en) | 2004-04-13 | 2009-03-24 | Pfizer Inc. | Androgen modulators |
| WO2005102990A1 (fr) | 2004-04-22 | 2005-11-03 | Warner-Lambert Company Llc | Modulateurs des androgenes |
| TW200724139A (en) | 2005-05-05 | 2007-07-01 | Warner Lambert Co | Androgen modulators |
| US7696229B2 (en) * | 2006-02-17 | 2010-04-13 | Memory Pharmaceuticals Corporation | Compounds having 5-HT6 receptor affinity |
| ES2398299T3 (es) | 2006-07-03 | 2013-03-15 | Proximagen Ltd. | Indoles como moduladores de 5-HT6 |
| CA2674087A1 (fr) * | 2007-02-16 | 2008-08-21 | Memory Pharmaceuticals Corporation | Composes substitues en 6 presentant une affinite avec le recepteur 5-ht6 |
| WO2008113818A1 (fr) * | 2007-03-21 | 2008-09-25 | Glaxo Group Limited | Utilisation de composés pour le traitement de la douleur et du syndrome du côlon irritable |
| AU2008256859A1 (en) * | 2007-05-24 | 2008-12-04 | Memory Pharmaceuticals Corporation | 4' substituted compounds having 5-HT6 receptor affinity |
| MX2010001576A (es) * | 2007-08-15 | 2010-09-14 | Memory Pharm Corp | Compuestos 3´sustituidos que tienen afinidad con el receptor 5-hidroxitriptamina 6 (5-ht6). |
| EP2508177A1 (fr) | 2007-12-12 | 2012-10-10 | Glaxo Group Limited | Associations contenant de la 3-phenylsulfonyl-8-piperazinyl-1yl-quinoline |
| US20110021538A1 (en) | 2008-04-02 | 2011-01-27 | Arena Pharmaceuticals, Inc. | Processes for the preparation of pyrazole derivatives useful as modulators of the 5-ht2a serotonin receptor |
| US20100016297A1 (en) * | 2008-06-24 | 2010-01-21 | Memory Pharmaceuticals Corporation | Alkyl-substituted 3' compounds having 5-ht6 receptor affinity |
| US20100022581A1 (en) * | 2008-07-02 | 2010-01-28 | Memory Pharmaceuticals Corporation | Pyrrolidine-substituted azaindole compounds having 5-ht6 receptor affinity |
| US20100029629A1 (en) * | 2008-07-25 | 2010-02-04 | Memory Pharmaceuticals Corporation | Acyclic compounds having 5-ht6 receptor affinity |
| US20100056531A1 (en) * | 2008-08-22 | 2010-03-04 | Memory Pharmaceuticals Corporation | Alkyl-substituted 3' compounds having 5-ht6 receptor affinity |
| WO2010062321A1 (fr) | 2008-10-28 | 2010-06-03 | Arena Pharmaceuticals, Inc. | Procédés utiles pour la préparation de 1-[3-(4-bromo-2-méthyl-2h-pyrazol-3-yl)-4-méthoxy-phényl]-3-(2,4-difluoro‑phényl)-urée, et formes cristallines associées |
| WO2011088836A1 (fr) | 2010-01-25 | 2011-07-28 | H. Lundbeck A/S | Nouveaux dérivés de 4-(aryl-4-sulfonyl)-6,6a,7,8,9,10-hexahydro-4h-4,8,10a-triaza-acéphénanthrylène et 3-arylsulfonyl-6,6a,7,8,9,10-hexahydro-3h-3,8,10a-triaza-cyclopenta[c]fluorène en tant que ligands de sérotonine 5-ht6 |
| JP6454346B2 (ja) | 2013-12-20 | 2019-01-16 | サンシャイン・レイク・ファーマ・カンパニー・リミテッドSunshine Lake Pharma Co.,Ltd. | 芳香族複素環式化合物及び医薬におけるその応用 |
| CA2953004C (fr) * | 2014-07-08 | 2023-02-21 | Sunshine Lake Pharma Co., Ltd. | Derives heterocycliques aromatiques et leurs applications pharmaceutiques |
| JP2018516992A (ja) | 2015-06-12 | 2018-06-28 | アクソファント サイエンシーズ ゲーエムベーハーAxovant Sciences Gmbh | レム睡眠行動障害の予防および処置のために有用なジアリールおよびアリールヘテロアリール尿素誘導体 |
| RU2018103338A (ru) | 2015-07-15 | 2019-08-15 | Аксовант Сайенсиз Гмбх | Производные диарил- и арилгетероарилмочевины для профилактики и лечения галлюцинаций, ассоциированных с нейродегенеративным заболеванием |
| WO2017052394A1 (fr) | 2015-09-23 | 2017-03-30 | Uniwersytet Jagielloński | Composés d'imidazopyridine et leur utilisation comme ligands du récepteur 5-ht6 |
| CN111362859B (zh) * | 2020-03-16 | 2021-05-11 | 东莞市东阳光新药研发有限公司 | 芳杂环类衍生物的盐及其用途 |
| CN111362858B (zh) * | 2020-03-16 | 2021-05-11 | 东莞市东阳光新药研发有限公司 | 芳杂环类衍生物的盐及其用途 |
| CN111233737B (zh) * | 2020-03-16 | 2021-05-11 | 东莞市东阳光新药研发有限公司 | 芳杂环类衍生物的盐及其用途 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4808581A (en) * | 1986-03-17 | 1989-02-28 | Glaxo Group Limited | Imidazolyl- indolylpropanones as 5-HT3 receptor antagonists |
| GB8623819D0 (en) * | 1986-10-03 | 1986-11-05 | Glaxo Group Ltd | Heterocyclic compounds |
| US4859662A (en) * | 1986-11-28 | 1989-08-22 | Glaxo Group Limited | Tetrahydro-imidazolylmethylcarbazolones and analogs thereof for treating 5-HT function disturbances |
| GB8720693D0 (en) * | 1987-09-03 | 1987-10-07 | Glaxo Group Ltd | Chemical compounds |
| US4985422A (en) * | 1988-04-27 | 1991-01-15 | Glaxo Group Limited | Lactam derivatives |
| GB8904551D0 (en) * | 1989-02-28 | 1989-04-12 | Glaxo Group Ltd | Chemical compounds |
| NZ233285A (en) * | 1989-04-18 | 1992-06-25 | Duphar Int Res | Imidazole-substituted carbamoyl-indoles and condensed analogues thereof and pharmaceutical compositions |
| US5225407A (en) * | 1990-02-22 | 1993-07-06 | Glaxo Group Limited | 5-HT3 receptor antagonists for the treatment of autism |
| FR2665444B1 (fr) * | 1990-08-06 | 1992-11-27 | Sanofi Sa | Derives d'amino-benzofuranne, benzothiophene ou indole, leur procede de preparation ainsi que les compositions les contenant. |
| US5965574A (en) * | 1996-08-13 | 1999-10-12 | Chen; Yuhpyng Liang | Heteroaryl amines as novel acetylcholinesterase inhibitors |
| CA2194984C (fr) * | 1994-07-26 | 2002-07-02 | John Eugene Macor | Derives 4-indole utilises comme agonistes et antagonistes de la serotonine |
| US6100291A (en) * | 1998-03-16 | 2000-08-08 | Allelix Biopharmaceuticals Inc. | Pyrrolidine-indole compounds having 5-HT6 affinity |
| US6818639B2 (en) * | 2000-07-21 | 2004-11-16 | Biovitrum Ab | Pharmaceutical combination formulation and method of treatment with the combination |
| EP1326830A1 (fr) * | 2000-10-20 | 2003-07-16 | Biovitrum Ab | N1-(benzensulfonyl)indoles substitues en 2, 3, 4 ou 5 et leur utilisation a des fins de traitement |
-
2001
- 2001-11-16 US US10/432,471 patent/US20050176705A1/en not_active Abandoned
- 2001-11-16 DE DE60122767T patent/DE60122767T2/de not_active Expired - Fee Related
- 2001-11-16 AT AT01997304T patent/ATE337780T1/de not_active IP Right Cessation
- 2001-11-16 JP JP2002544068A patent/JP2004517072A/ja active Pending
- 2001-11-16 WO PCT/EP2001/013411 patent/WO2002041889A2/fr active IP Right Grant
- 2001-11-16 AU AU2002220715A patent/AU2002220715A1/en not_active Abandoned
- 2001-11-16 ES ES01997304T patent/ES2271124T3/es not_active Expired - Lifetime
- 2001-11-16 EP EP01997304A patent/EP1335722B1/fr not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| DE60122767T2 (de) | 2007-09-13 |
| WO2002041889A2 (fr) | 2002-05-30 |
| AU2002220715A1 (en) | 2002-06-03 |
| WO2002041889A3 (fr) | 2003-04-10 |
| ES2271124T3 (es) | 2007-04-16 |
| EP1335722A2 (fr) | 2003-08-20 |
| ATE337780T1 (de) | 2006-09-15 |
| US20050176705A1 (en) | 2005-08-11 |
| JP2004517072A (ja) | 2004-06-10 |
| DE60122767D1 (de) | 2006-10-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1335722B1 (fr) | Derivés indole-sulfoniques pour le traitement de troubles du snc | |
| EP1401812B1 (fr) | Derives de 4-piperazinylindole a affinite avec les recepteurs 5-ht6 | |
| US6316450B1 (en) | Compounds | |
| EP1401813B1 (fr) | Nouveaux derives de l'indole a affinite pour le recepteur 5-ht6 | |
| US6730695B2 (en) | 1,3-dihydro-2h-indol-2-one derivatives and their use as ligands for V1b and V1a arginine-vasopressin receptors | |
| US20040242589A1 (en) | 3-arylsulfonyl-7-piperzinyl-indoles-benzofurans and -benzothiophenes with 5-ht6 receptor affinity for treating cns disorders | |
| US20050090496A1 (en) | Sulphonyl compounds with 5-ht6 receptor affinity | |
| EP1392316B1 (fr) | Derives de benzo[d]azepine utiles comme antagonistes du recepteur 5-ht6 | |
| JP2009185031A (ja) | 2−、3−、4−、または5−置換−n1−(ベンゼンスルホニル)インドールおよび治療におけるそれらの使用 | |
| US6849644B2 (en) | Isoquinoline derivatives useful in the treatment of CNS disorders | |
| US6660751B1 (en) | Sulfonamide derivatives as 5-HT7 receptor antagonists | |
| NZ286090A (en) | 1,2,5-thiadiazole derivatives of indolylalkyl-pyridinyl- and pyrimidinylpiperazine, preparation and pharmaceutical compositions thereof | |
| ZA200309258B (en) | 4-Piperazinylindole derivatives with 5-HT6 receptor affinity. | |
| CZ200082A3 (cs) | Sulfonamidové derváty, antagonisté receptoru 5- HT6, a způsob jejich přípravy | |
| AU2002345587A1 (en) | 4-Piperazinylindole derivatives with 5-HT6 receptor affinity |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20030522 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK RO SI |
|
| 17Q | First examination report despatched |
Effective date: 20031103 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| RTI1 | Title (correction) |
Free format text: INDOLYL-SULFONYL- COMPOUNDS USEFUL IN THE TREATMENT OF CNS DISORDERS |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE TR |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060830 Ref country code: CH Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060830 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20060830 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060830 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060830 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060830 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20060920 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20061004 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20061005 Year of fee payment: 6 |
|
| REF | Corresponds to: |
Ref document number: 60122767 Country of ref document: DE Date of ref document: 20061012 Kind code of ref document: P |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20061024 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20061031 Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20061116 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20061130 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20061130 Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20061130 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20061130 Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070212 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2271124 Country of ref document: ES Kind code of ref document: T3 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20070531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20061201 |
|
| BERE | Be: lapsed |
Owner name: *SMITHKLINE BEECHAM P.L.C. Effective date: 20071130 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20071116 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060830 Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20061116 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080603 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20080930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060830 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071116 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20071117 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071117 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071116 |