EP0903770A2 - Beleuchtungssystem - Google Patents
Beleuchtungssystem Download PDFInfo
- Publication number
- EP0903770A2 EP0903770A2 EP98111187A EP98111187A EP0903770A2 EP 0903770 A2 EP0903770 A2 EP 0903770A2 EP 98111187 A EP98111187 A EP 98111187A EP 98111187 A EP98111187 A EP 98111187A EP 0903770 A2 EP0903770 A2 EP 0903770A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- lamp
- lighting system
- voltage
- mercury
- metal halide
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J61/00—Gas-discharge or vapour-discharge lamps
- H01J61/02—Details
- H01J61/12—Selection of substances for gas fillings; Specified operating pressure or temperature
- H01J61/18—Selection of substances for gas fillings; Specified operating pressure or temperature having a metallic vapour as the principal constituent
Definitions
- the invention is based on a lighting system according to the preamble of claim 1, consisting of lamp and ballast.
- lamps metal halide lamps with ceramic Discharge vessel inserted.
- mercury is intensified as an environmentally harmful and toxic substance viewed in modern mass products due to the environmental hazard Avoided during use, production and disposal shall be. Therefore, efforts are increasing to to develop mercury-free high-pressure discharge lamps.
- From DE-PS 40 35 561 is already a metal halide lamp with a ceramic Discharge vessel known, the mercury-free filling noble gas (Xenon) and a halide of lithium (or Na, Tl, In) for production an arc discharge.
- the filling also contains a substance which forms a halide complex, for example a halide of Aluminum or tin with the halides of sodium or lithium Forms complexes.
- From DE-PS 27 07 204 is a mercury-free filling with noble gases and Metal halides known to contain thallium, one or two rare earth metals (Dy, Ho) and / or an alkali metal (Na, Cs) and possibly indium.
- noble gases and Metal halides known to contain thallium, one or two rare earth metals (Dy, Ho) and / or an alkali metal (Na, Cs) and possibly indium.
- a metal halide lamp with high luminous efficacy which uses mercury as a buffer gas.
- An exemplary embodiment also shows a mercury-free filling for daylight use with a color temperature of 5350 K using HfBr 4 as the metal halide and addition of elemental tin.
- the xenon (cold filling pressure 1 bar) takes on the role of the buffer gas.
- these lamps have enormous re-ignition peaks of around 600 V and can therefore only be operated with complex circuitry.
- low-mercury or almost mercury-free fillings are mainly used for electrodeless high-pressure metal halide lamps, since the coupling of the electrical energy via electromagnetic waves decreases with increasing mercury density and is shielded in the outer plasma layers.
- xenon (Xe) or other noble gases are predominantly used as buffer gases or mercury is filled in in very small amounts ( ⁇ 1 mg / cm 3 , "essentially mercury-free").
- this technology is very complex and unsuitable for lamps with low power (below 250 W), since the luminous efficiency then drops drastically.
- the underlying task requires a substitute or a mixture of substitutes for mercury in high-pressure lamps with at the same time extensive Preservation of the lighting and electrical properties of the typical High pressure metal halide lamp.
- the discharge vessel can consist of quartz glass.
- a discharge vessel made of ceramic, transparent or translucent material that can withstand higher thermal loads is.
- This material can be made of monocrystalline metal oxide (e.g. sapphire), polycrystalline sintered metal oxide (e.g .: PCA: polycrystalline, densely sintered Aluminum oxide, yttrium aluminum garnet or yttrium oxide) or consist of polycrystalline non-oxidative material (e.g. AlN).
- Xe is mainly used as a substitute for Hg as a buffer gas heaviest of the stable noble gases used. It can be used when using Discharge vessels made of quartz glass can be filled in by freezing out, see above that the lamp filling contains the buffer gas in excess pressure. When using of ceramic bodies as a discharge vessel can this filling process because the resulting high temperature gradient along the discharge vessel lead to jumps and is therefore only with great effort and risk applicable.
- xenon only makes a small contribution as a buffer gas (10 to 20%) to the voltage gradient in the lamp.
- a mercury-free electrode-containing metal halide lamp with a ceramic discharge vessel in an evacuated outer bulb Quartz glass or hard glass with high luminous efficacy (typ.> 80 lm / W), and high Color rendering index (typically Ra> 80).
- the range can preferably be Realize warm white to neutral white color temperatures (typ. 3000 - 4500 K). It may be but also possible, daylight white color temperatures (um 5300 K) with high Ra (approx. 90).
- Halogen here and in the following is always iodine, bromine or chlorine, however not fluorine. The same applies to halides.
- first additional additives preferably metal halides, to improve the electrical Lamp properties and to influence the arc temperature profile used.
- Metals or metal compounds are particularly suitable for this, their excitation or ionization energies in the range of the above Metal halides lie and are preferably below.
- second additives preferably elemental metals
- elemental metals can be added to the filling, which reduce the re-ignition peaks by acting as a getter for free electronegative gas fractions.
- Their halides have lower enthalpies of formation than metal compounds, which can possibly be formed from the material of the electrodes and that of the current leads (W, Mo) in the lamp. They essentially serve to extend the life of the lamps and support an effective, stable chemical cycle.
- These are mostly elemental metals that are present in excess of the halides of these metals that have already been filled in, in particular aluminum, tin and magnesium. Good experiences have also been achieved with elementary tantalum.
- the maximum dosage of these metals is 10 mg / cm 3 in each case.
- discharge vessels are made of for the present invention Quartz glass can be used. However, preference is given to lamps with ceramic vessels which allow much higher wall temperatures. So one can clearly higher total pressure and partial vapor pressure as well as a higher particle density adjust the materials used to generate light. Also be the conditions for the possibility of metal halide complex formation and the possibility of the formation of supersaturated metal vapors to form metal-atom clusters by increasing the Wall temperature improved.
- the ratio of the total molar amount of all the metals introduced is preferably to the total molar amount of all filled-in halogens between 0.1 and 10.
- the lamps are operated on AC voltage in such a way that that the rate of change in lamp voltage is (in absolute terms voltage rise in negative or positive direction) occurs so quickly during the polarity change that re-ignition peaks can be greatly reduced over the course of the lamp voltage. Thereby extinguishing of the lamp is reliably prevented. These reignition tips arise from the extinction of the discharge arc when the polarity changes and by cooling the electrodes.
- the level of the still acceptable reignition peak is determined on the one hand after the open circuit voltage, i.e. after the supply voltage, the maximum is attainable, and on the other hand after the response voltage of an im Ignition device located voltage path, which is exceeded when a certain voltage level (just the response voltage) ignition pulses the lamp voltage generated.
- a faulty mode of operation with too high Re-ignition tip leads to overloading of the igniter and shortens it its lifespan.
- the Voltage change rate of the lamp voltage which is called the absolute value of the Voltage change divided by the duration of the voltage change is defined (therefore in the following it is often simplified as the voltage rise rate referred to), at least at 0.3 V / ⁇ s, particularly preferably at are at least 1 V / ⁇ s. Good results are achieved at around 3 V / ⁇ s.
- a sufficient rate of voltage rise can in principle be determined by a realize relatively high-frequency sinusoidal AC voltage (at least 1 kHz, preferably more than 250 kHz). In principle, they are suitable also other similar voltage forms (for example sawtooth shape) comparable duration of the half period.
- the latter corresponds to the usual mains voltage of 230 V eff .
- a medium-voltage mains voltage (approx. 110 V rms ) can of course also be used.
- Acceptable re-ignition peaks of the lamp voltage here the peak voltage is of primary interest and less the effective value of the voltage) must be significantly below the response voltage.
- a value of approximately 75% of the open circuit voltage is therefore acceptable for the re-ignition peak.
- this gives a value of 173 V eff i.e. a peak voltage of 244 V pk .
- Operation on an electronic ballast is particularly preferred with rectangular current injection, since this pulse shape is steep from the start Flanks guaranteed.
- a frequency of 50 Hz is sufficient to increase the voltage rise rate when changing polarity to the above set the range above 0.3 V / ⁇ s. This is due to the steepness the edges of the rectangle. But it is also a higher frequency operation (for example 120 Hz or more) possible.
- a period of time is advantageous Voltage rise of at most about 400 ⁇ s, in a particularly preferred one Embodiment, it is less than 100 microseconds.
- A is very suitable Value of about 10 to 50 ⁇ s.
- a suitable electronic ballast is in principle, for example, from the US Pat. No. 4,291,254 or DE-OS 44 00 093, both of which are known express reference is made. However, there is above all the aspect the increased luminous efficiency (up to 8%) due to the high operating frequency.
- a particular advantage of rectangular operation is that it provides the basis is created for stable continuous operation without acoustic resonances.
- high-frequency sinusoidal excitation is also possible when operating at frequencies> 1 kHz with sinusoidal voltage edges takes place, their time scale typically the steep edges in rectangular operation (Order of magnitude 10 to 100 ⁇ s).
- a high frequency > 250 kHz
- the voltage rise rate is such is set that re-ignition peaks that on the burning voltage of the Lamp are stamped, are suppressed as possible. Then also at sinusoidal AC voltage, stable operation possible.
- Another advantageous aspect of rectangular current operation is also that the performance of the lamp is constant to within a few percent can be maintained (constant wattage operation).
- the lamp should at least 50% during the first few minutes (preferably more than 60%) of the nominal power.
- Advantageous therefore electronic ballasts with rectangular operation are used, with which a "constant wattage" operation can be realized and the occurrence of high reignition peaks is reliably avoided.
- In principle is a circuit for operating a high-pressure discharge lamp with constant Performance known for example from EP-A 680 245.
- the approach according to the invention now consists in instead of xenon primarily iodides or bromides of easily evaporable metals use to generate a voltage gradient comparable to the mercury.
- Bromine and iodine (atomic or molecular) alone or in combination have a large cross section for electron capture. Thereby the operating voltage of a lamp is raised to form negative Ions or molecules.
- the concept of the voltage gradient generator can be modified accordingly that the metal halides alone do not perform this function, but a certain contribution to the voltage gradient (up to 40%) due to a correspondingly high xenon pressure (more than 500 mb cold filling pressure) is contributed.
- This allows a good coordination with regard to on the simplest possible filling systems in which a part of the Voltage gradient formers also used metal halides as Light formers act, for example halides of Al, In, Mg and before All of the part.
- the advantage of this concept is that when starting with high starting current (typically 2 A) the electrodes against excessive overheating be protected if xenon acts as an ignition gas and gradient generator.
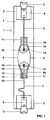
- a metal halide lamp with an output of 70 W is shown schematically in FIG. It consists of a cylindrical outer bulb 1 made of quartz glass which defines a lamp axis and is squeezed (2) and base (3) on two sides.
- the axially arranged discharge vessel 4 made of Al 2 O 3 ceramic is bulged in the middle 5 and has two cylindrical ends 6a and 6b.
- it can also be cylindrical with elongated capillary tubes as plugs, as is known, for example, from EP-A 587 238.
- the discharge vessel is held in the outer bulb 1 by means of two power supply lines 7, which are connected to the base parts 3 via foils 8.
- the power supply lines 7, one of which is a molybdenum band to compensate for the large expansion differences, are welded to bushings 9, 10, which are each fitted in an end plug 11 at the end of the discharge vessel.
- the bushings 9, 10 are, for example, molybdenum pins. Both executions 9, 10 are on the plug 11 on both sides and hold on the discharge side Electrodes 14, consisting of an electrode shaft 15 Tungsten and a helix 16 pushed on at the discharge end.
- the bushing 9, 10 is in each case with the electrode shaft 15 and with the outer power supply 7 butt welded.
- the end plugs 11 essentially consist of a cermet known per se with the ceramic component Al 2 O 3 and the metallic component tungsten or molybdenum.
- the filling of the discharge vessel consists of an inert ignition gas / buffer gas, here argon with 250 mbar cold filling pressure and from various additives on metal halides.
- TlJ has a dual function as a voltage gradient generator and photographers proven, in combination with others Stress gradient formers.
- Table 2 shows some fillings, with voltage gradient formers and light formers shown separately.
- the light color is in the warm white to neutral white range (3500 to 4250 K).
- the voltage gradient is usually in the order of 60 to 120 V / cm. Surprisingly, however, relatively low voltage gradients between 45 and 60 V / cm still lead to good lighting values.
- the voltage gradient in a conventional metal halide lamp with a mercury filling is approximately between 75 and 110 V / cm.
- the metal halide mixtures shown in Table 3 are used as light formers resorted to, with CsJ as an additional additive of the first Type is taken into account.
- a three-component mixture is particularly suitable as a light generator, consisting of thallium as the first component, Sodium and / or cerium as a second component and at least one rare earth metal as the third component.
- a lamp volume of 0.3 cm 3 was used for all fillings.
- the electrode gap is 9 mm.
- the specific wall load (defined as electrical power / inner surface) varies between 15 and 50 W / cm 2 . On average, it is 25 W / cm 2 .
- the specific electrical power density varies between 100 and 500 W / cm 3 . On average, it is 235 W / cm 3 .
- the lamps were each operated on an electronic ballast with rectangular current injection in a regulated power mode with I eff ⁇ 1.8 A.
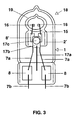
- the lamp is a metal halide lamp 18 with 70 W power, which is squeezed on one side, and also the discharge vessel 19 is a quartz glass bulb squeezed on one side. Closer Details of this can be found, for example, in US Pat. No. 4,717,852. Otherwise correspond to the same reference numerals for analog components as in FIG. 1. A getter 17 is also accommodated in the outer bulb 1.
- a neutral white filling was used for this, based on voltage gradient formers that form easily vaporizable halides (AlJ 3 , SnJ 4 , HfJ 4 ) and that approximate the voltage gradient of Hg.
- An Xe filling of 800 mbar was used as the starting gas.
- HfJ 4 filling shows the strongest voltage gradient due to its high vapor pressure, while AlJ 3 (symbolized as ⁇ ) and SnJ 4 (symbolized as ⁇ ) show approximately the same behavior, even with different dosage amounts.
- the lamps according to the invention should therefore preferably be operated with a rectangular electronic ballast in which the edges of the rectangular pulse are so steep (in the order of magnitude approx. 10 to 50 ⁇ sec) that no noticeable re-ignition peaks occur. Then, for example, the SnJ 4 dosage (11 mg) lowers the operating voltage from 92.8 V to 78.0 V, that is to say by 14.9 V (symbolized as a large ⁇ in FIG. 4). The associated re-ignition peak, which still had a value of 329 V during KVG operation, disappears almost completely (symbolized as a small ⁇ in Figure 4).
- the lamps initially only after taking over the discharge arc have a burning voltage of approx. 20 V (because no halides have yet evaporated are, the power at the KVG is only about 25-30 W, because the choke limited the current to just over 1 A. With this low performance the lamp remains so cold that the halides cannot evaporate and the lamp gets stuck in the start-up phase. For measurements at the KVG the lamp current was therefore started up by means of a control choke almost 2 A increased. This is sufficient for the evaporation of the halides then causes an increase in the burning voltage so that the current again can be withdrawn.
- argon with a cold filling pressure of 150 mbar was used as the starting gas.
- the voltage gradient formers AlJ 3 and SnJ 4 light additions of DyJ 3 and TmJ 3 (0.27 mg each) and TlJ (0.1 mg) and NaJ (0.4 mg) were used to reduce the emission to strengthen in the visible spectral region.
- the DyJ 3 is used as an additive to the AlJ 3 to achieve a better emission in the red.
- the TmJ 3 is used as an addition to the SnJ 4 to increase the emission in the blue and green.
- the AlJ 3 / DyJ 3 / NaJ / TlJ system achieved an operating voltage of 64.1 V.
- a very similar filling was used for a metal halide lamp with a ceramic discharge vessel.
- the filling consists of 5 mg AlJ 3 as a voltage gradient and the light formers DyJ 3 , TmJ 3 , TlJ, NaJ.
- the ceramic discharge vessel has a volume of 0.3 cm 3 and an electrode spacing of 9 mm. 51.2 V operating voltage was achieved with a very high luminous flux of 5 klm.
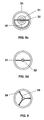
- Figure 5 is another embodiment of an inventive Metal halide lamp 20 shown with a power of 70 W.
- Figure 5a and 5b each show side views rotated by 90 °
- FIG. 5c shows a view of FIG above.
- FIG. 5d shows a section through a lamp corresponding to FIG. 5c.
- the holding frame 23 is also on the foils 24a, 24b of the outer bulb 25 squeezed on one side attached to a G12 ceramic base.
- the squeeze-close implementation 26 is via a short angled power supply 27 with one Foil 24a connected.
- the pinch-free bushing 28 is over a Conductor system with double symmetry and a short power supply 36 connected to the other film 24b.
- the conductor system consists of one semicircular arch 30, the level of the crushing implementation 26 in a plane transverse to the lamp axis on the inside of the wall of the outer bulb is led.
- a typical value for current 1 is 1 to 2 A.
- the force deflecting the discharge arc is proportional to I 2 and the effective length l of the return, which corresponds to the length of the arc, and inversely proportional to the distance r between the return and discharge arc:
- the returns (31; 38) with sleeves 39 made of suitable ones are advantageous Materials (quartz stocking, ceramic tube) coated in a manner known per se to avoid photo effects from UV radiation. More than four Returns (fourfold symmetry) lead to a noticeable Shading and are therefore, especially for cost reasons, less suitable.
- FIG 6 is a corresponding section through a lamp with three Symmetry shown.
- the three returns 38 decrease according to Eq. (1) the magnetic force to a ninth, compared to the magnetic By force of a single repatriation. They run at the end of the ceramic remote from the base Discharge vessel in a star shape for metallic passage together.
- the returns 38 are from ceramic sleeves 39 for Shield surrounded by UV radiation.
- the mercury-free filling for the lamp in FIGS. 5 and 6 consists of the voltage gradient formers InBr (2 mg) and TlJ and contains the filling MHS 8-6 (5 mg), see Table 3.
- 1 mg elemental indium is added. Indeed, it has been found that the addition of elemental metal further reduces the re-ignition spike.
- the electrode gap is 9 mm.
- the discharge volume is 0.3 cm 3 . The behavior regarding the reignition peak was investigated in detail on this system.
- the lamp voltage (in V) is a function of time (in milliseconds ms).
- the lamp was at a frequency of each 120 Hz either a sinusoidal AC voltage (curve A) or a rectangular AC voltage (curves B to E) is impressed.
- the amplitude of the operating voltage in the first half-wave is approximately 65 V.
- the corresponding voltage change rates can be calculated from FIG. 8, where the reignition peak voltage (in V) as a function of time the voltage change (in ⁇ s) is specified.
- the Voltage change rate is to be noted that in each case to the specified Measured value of the peak voltage in the area of the re-ignition peak or Base value of the burning voltage (denoted by x) from the previous one Half period (approx. -65 V) must be added.
- curve A correspond to a voltage change rate of 0.25 V / ⁇ s, this value is significantly higher for rectangular operation.
Landscapes
- Discharge Lamp (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Glass Compositions (AREA)
Abstract
- ein Puffergas, das auch als Startgas zur Zündung der Lampe wirkt,
- einen Spannungsgradientenbildner, bestehend aus mindestens einem Metallhalogenid, das leicht verdampft, und das hauptsächlich (zu mehr als 50%) dafür verantwortlich ist, einen Spannungsgradienten zu erzeugen, der in etwa dem von Quecksilber entspricht,
- ein Lichterzeuger, bestehend aus einem Metall und/oder Metallhalogeniden.
Description
- Figur 1
- eine Metallhalogenidlampe mit keramischem Entladungsgefäß;
- Figur 2
- ein Spektrum einer Metallhalogenidlampe;
- Figur 3
- eine Metallhalogenidlampe mit einem Entladungsgefäß aus Quarzglas;
- Figur 4
- ein Diagramm, das die Brennspannung und Wiederzünd-Spitzenspannung als Funktion der Füllmenge zeigt;
- Figur 5
- eine keramische Metallhalogenidlampe mit speziellem Haltegestell;
- Figur 6
- einen Schnitt durch eine Lampe mit dreizähliger Symmetrie;
- Figur 7
- eine Darstellung des Wiederzündverhaltens bei unterschiedlicher Flankensteilheit;
- Figur 8
- die Wiederzünd-Spitzenspannung für die verschiedenen Spannungsformen aus Figur 7;
- Figur 9
- eine Übersicht über Lichtausbeuten, Farbwiedergabeindices und Brennspannungen verschiedener Füllungen;
- Figur 10
- das Maintenance-Verhalten zweier Füllungen.
Claims (25)
- Beleuchtungssystem, umfassend eine quecksilberfreie Metallhalogenidlampe mit einer Lichtausbeute von mindestens 75 lm/W und einem Farbwiedergabeindex von mindestens 75 und ein elektronisches Vorschaltgerät, das Wechselspannung vermittelt, wobei die Lampe ein Entladungsgefäß umfaßt, in das Elektroden vakuumdicht eingeführt sind, dadurch gekennzeichnet, daß das elektronisches Vorschaltgerät der Lampe eine Änderung der Spannung während des Polaritätswechsels mit einer Spannungsänderungsrate von mindestens 0,3 V/µs, bevorzugt mindestens 1 V/µs, vermittelt, wobei die Füllung folgende Komponenten umfaßt:ein Puffergas, das auch als Startgas zur Zündung der Lampe wirkt,einen Spannungsgradientenbildner, bestehend zumindest aus einem Metallhalogenid, das leicht verdampft, und das hauptsächlich (zu mehr als 50%) dafür verantwortlich ist, einen Spannungsgradienten von mindestens 45 V/cm zu erzeugen, der bevorzugt in etwa dem von Quecksilber entspricht,ein Lichterzeuger, bestehend zumindest aus einem Metallhalogenid und/oder einem Metall.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß der Spannungsgradientenbildner ein Metalljodid und/oder Metallbromid ist, insbesondere mit einem Betriebsfülldruck von mindestens 0,5 bar.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß das elektronische Vorschaltgerät der Lampe eine Rechteck-Stromversorgung einprägt.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß das elektronische Vorschaltgerät die Leistung im Betrieb konstant hält.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß die Zeitdauer der Spannungsänderung während eines Polaritätswechsels so kurz ist, daß die Wiederzündspitze stark unterdrückt wird, wobei insbesondere diese Zeitspanne kleiner 1000 µs, vorteilhaft kleiner 100 µs, ist.
- Beleuchtungssystem nach Anspruch 5, dadurch gekennzeichnet, daß die Spannungsänderung in der Flanke eines Rechteckpulses realisiert wird.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß das Startgas ein Edelgas oder eine Mischung von Edelgasen mit einem Kaltfülldruck von mindestens 1 mbar ist.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß der Spannungsgradientenbildner mindestens ein Halogenid (außer Fluorid) folgender Metalle ist: Al, Bi, Hf, In, Mg, Sc, Sn, Tl, Zr, Zn, Sb, Ga.
- Beleuchtungssystem nach Anspruch 5, dadurch gekennzeichnet, daß der Spannungsgradientenbildner in einer Menge von 1 bis 200 µmol/cm3 im Entladungsgefäß vorliegt.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß der Lichtbildner mindestens eines der folgenden Metalle oder eine Verbindung dieses Metalls, insbesondere ein Halogenid davon, ist: Na, Pr, Nd, Ce, La, Dy, Ho, Tl, Sc, Hf, Zr, Tm.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß der Lichtbildner in einer Menge zwischen 1 und 30 mg/cm3 im Entladungsgefäß vorliegt.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß die Füllung Zusatzadditive enthält zur Verbesserung der elektrischen Lampeneigenschaften und zur Beeinflussung des Temperaturprofils des Bogens, insbesondere Metallhalogenide mit niedriger Anregungs- bzw. Ionisierungsenergie.
- Beleuchtungssystem nach Anspruch 12, dadurch gekennzeichnet, daß die Zusatzadditive Cäsium und evtl. Lithium (letzteres nur für den Fall, daß die Füllung kein Natrium aufweist) enthalten.
- Beleuchtungssystem nach Anspruch 12, dadurch gekennzeichnet, daß der Anteil der Zusatzadditive in der Größenordnung von 5 bis 50 Mol-%, verglichen mit dem Anteil der Lichtbildner, liegt.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß die Füllung elementare Metalle (im Überschuß) enthält, die die Wiederzündspitze verringern, insbesondere in einer Menge zwischen 1 und 10 mg/cm3.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß die Füllung elementares Ta oder In enthält.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß das Entladungsgefäß aus Keramik besteht.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß elementares Zn als ein Spannungsgradientenbildner enthalten ist.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß die Leistung der Lampe maximal 250 W beträgt.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß das Entladungsgefäß von einem evakuierten Außenkolben umgeben ist.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß die Farbtemperatur der Lampe zwischen 2800 und 4600 K beträgt.
- Beleuchtungssystem nach Anspruch 1, dadurch gekennzeichnet, daß die Farbtemperatur der Lampe etwa 5300 K beträgt.
- Quecksilberfreie Metallhalogenidlampe mit einer Lichtausbeute von mindestens 75 lm/W und einem Farbwiedergabeindex von mindestens 75 zum Betrieb an einem eine Wechselspannung vermittelnden elektronischen Vorschaltgerät, das einen Polaritätswechsel mit einer Spannungsänderungsrate von mindestens 0,3 V/µs vermittelt, wobei die Lampe ein Entladungsgefäß umfaßt, in das Elektroden vakuumdicht eingeführt sind, dadurch gekennzeichnet, daß die Füllung folgende Komponenten umfaßt:ein Puffergas, das auch als Startgas zur Zündung der Lampe wirkt,ein Spannungsgradientenbildner, bestehend zumindest aus einem Metallhalogenid, das leicht verdampft, und das hauptsächlich (zu mehr als 50%) dafür verantwortlich ist, einen Spannungsgradienten zu erzeugen, der in etwa dem von Quecksilber entspricht,ein Lichterzeuger, bestehend zumindest aus einem Metallhalogenid und/oder einem Metall.
- Metallhalogenidlampe nach Anspruch 23, dadurch gekennzeichnet, daß das Entladungsgefäß (21) in einem einseitig gequetschten Außenkolben (25) mittels eines Haltegestells (23) befestigt ist, wobei das Haltegestell eine rückführende Stromzuführung (31;38) mit mindestens zweizähliger Symmetrie besitzt.
- Quecksilberfreie Metallhalogenidlampe, wobei die Lampe ein Entladungsgefäß (21) umfaßt, in das Elektroden vakuumdicht eingeführt sind, wobei das Entladungsgefäß (21) in einem einseitig gequetschten Außenkolben (25) mittels eines Haltegestells (23) befestigt ist, dadurch gekennzeichnet, daß das Haltegestell (23) ein rückführendes Leitersystem aus mindestens drei Stromzuführungen (38) besitzt, die symmetrisch angeordnet sind.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19731168A DE19731168A1 (de) | 1997-07-21 | 1997-07-21 | Beleuchtungssystem |
| DE19731168 | 1997-07-21 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0903770A2 true EP0903770A2 (de) | 1999-03-24 |
| EP0903770A3 EP0903770A3 (de) | 1999-04-07 |
| EP0903770B1 EP0903770B1 (de) | 2004-08-18 |
Family
ID=7836341
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP98111187A Expired - Lifetime EP0903770B1 (de) | 1997-07-21 | 1998-06-18 | Metallhalogenidlampe |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US6069456A (de) |
| EP (1) | EP0903770B1 (de) |
| JP (1) | JP4335332B2 (de) |
| AT (1) | ATE274236T1 (de) |
| CA (1) | CA2243737C (de) |
| DE (2) | DE19731168A1 (de) |
| HU (1) | HU221394B1 (de) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6586891B2 (en) | 2000-06-06 | 2003-07-01 | Matsushita Electric Industrial Co., Ltd. | High-intensity discharge lamp and high-intensity discharge lamp operating apparatus |
| US6608444B2 (en) | 2000-05-26 | 2003-08-19 | Matsushita Electric Industrial Co., Ltd. | Mercury-free high-intensity discharge lamp operating apparatus and mercury-free metal halide lamp |
| EP1045622A3 (de) * | 1999-04-14 | 2003-10-29 | Osram Sylvania Inc. | Vorschaltgerät für eine Bogenlampe |
| EP1271612A3 (de) * | 2001-06-19 | 2006-04-19 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Hochdruckentladungslampe |
| EP1011126A3 (de) * | 1998-12-14 | 2006-08-23 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Metallhalogenidlampe |
| US7126281B2 (en) | 2003-03-19 | 2006-10-24 | Patent-Treuhand-Gesellschaft für elektrishe Glūhlampen mbH | High-pressure discharge lamp for vehicle headlights |
Families Citing this family (78)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6653801B1 (en) * | 1979-11-06 | 2003-11-25 | Matsushita Electric Industrial Co., Ltd. | Mercury-free metal-halide lamp |
| JPH11238488A (ja) | 1997-06-06 | 1999-08-31 | Toshiba Lighting & Technology Corp | メタルハライド放電ランプ、メタルハライド放電ランプ点灯装置および照明装置 |
| JP2002536786A (ja) * | 1999-01-28 | 2002-10-29 | コーニンクレッカ フィリップス エレクトロニクス エヌ ヴィ | メタルハライドランプ |
| US6392346B1 (en) * | 1999-04-14 | 2002-05-21 | Osram Sylvania Inc. | Chemical composition for mercury free metal halide lamp |
| US6731069B1 (en) * | 1999-04-14 | 2004-05-04 | Osram Sylvania Inc. | Mercury-free metal halide arc lamps |
| DE19923237A1 (de) * | 1999-05-20 | 2000-11-23 | Patent Treuhand Ges Fuer Elektrische Gluehlampen Mbh | Schaltungsanordnung, zugeordnetes elektrisches System sowie Entladungslampe mit derartiger Schaltungsanordnung und Verfahren zu ihrem Betrieb |
| DE19937312A1 (de) * | 1999-08-10 | 2001-02-15 | Patent Treuhand Ges Fuer Elektrische Gluehlampen Mbh | Quecksilberfreie Metallhalogenidlampe |
| CN1174464C (zh) * | 1999-11-11 | 2004-11-03 | 皇家菲利浦电子有限公司 | 高压气体放电灯 |
| US6498429B1 (en) * | 1999-11-15 | 2002-12-24 | General Electric Company | Sodium-xenon lamp with improved characteristics at end-of-life |
| JP3415533B2 (ja) * | 2000-01-12 | 2003-06-09 | エヌイーシーマイクロ波管株式会社 | 高圧放電灯 |
| WO2001080606A1 (en) * | 2000-04-14 | 2001-10-25 | Macquarie Research Ltd | Methods and systems for providing emission of incoherent radiation and uses therefor |
| US6639343B2 (en) | 2000-07-14 | 2003-10-28 | Matsushita Electric Industrial Co., Ltd. | Mercury-free metal halide lamp |
| CN1333547A (zh) * | 2000-07-14 | 2002-01-30 | 松下电器产业株式会社 | 无水银金属卤化物灯 |
| US6573656B2 (en) | 2000-07-14 | 2003-06-03 | Matsushita Electric Industrial Co., Ltd. | High-pressure discharge lamp and method for producing the same |
| DE10044562A1 (de) * | 2000-09-08 | 2002-03-21 | Philips Corp Intellectual Pty | Niederdruckgasentladungslampe mit quecksilberfreier Gasfüllung |
| WO2002091429A1 (en) * | 2001-05-10 | 2002-11-14 | Koninklijke Philips Electronics N.V. | High-pressure gas discharge lamp |
| DE60206215T2 (de) * | 2001-06-27 | 2006-05-04 | Matsushita Electric Industrial Co., Ltd., Kadoma | Metall-Halogen-Lampe |
| JP2003016998A (ja) * | 2001-06-28 | 2003-01-17 | Matsushita Electric Ind Co Ltd | メタルハライドランプ |
| JP3990582B2 (ja) * | 2001-06-29 | 2007-10-17 | 松下電器産業株式会社 | メタルハライドランプ |
| EP1288998A1 (de) | 2001-08-24 | 2003-03-05 | Stanley Electric Co., Ltd. | Quecksilberfreie Metallhalogenidlampe, darin enthaltene Füllung und widerstandsbedingte Leistungssteuerung |
| JP2003151787A (ja) | 2001-08-29 | 2003-05-23 | Harison Toshiba Lighting Corp | 高圧放電ランプ点灯装置および自動車用ヘッドライト装置 |
| KR20030019167A (ko) * | 2001-08-30 | 2003-03-06 | 마쯔시다덴기산교 가부시키가이샤 | 고압방전 램프 및 그 제조방법 |
| JP3701222B2 (ja) * | 2001-09-14 | 2005-09-28 | 松下電器産業株式会社 | 高圧放電ランプ及びこれを用いた高圧放電ランプシステム |
| JP2003100251A (ja) * | 2001-09-27 | 2003-04-04 | Koito Mfg Co Ltd | 放電ランプ装置用水銀フリーアークチューブ |
| US6844676B2 (en) * | 2001-10-01 | 2005-01-18 | Koninklijke Philips Electronics N.V. | Ceramic HID lamp with special frame wire for stabilizing the arc |
| DE10163584C1 (de) * | 2001-11-26 | 2003-04-17 | Philips Corp Intellectual Pty | Verfahren und Vorrichtung zur Herstellung von Lampenkolben mit nicht-rotationssymmetrischer und/oder konkaver innerer und/oder äußerer Form |
| US6979958B2 (en) | 2002-01-31 | 2005-12-27 | Matsushita Electric Industrial Co., Ltd. | High efficacy metal halide lamp with praseodymium and sodium halides in a configured chamber |
| US7091673B2 (en) | 2002-07-02 | 2006-08-15 | Mitsubishi Denki Kabushiki Kaisha | Discharge lamp lighting device |
| JP2004063158A (ja) * | 2002-07-25 | 2004-02-26 | Koito Mfg Co Ltd | 放電バルブ |
| DE10242740A1 (de) * | 2002-09-13 | 2004-03-18 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Hochdruckentladungslampe für Kraftfahrzeugscheinwerfer |
| US7215081B2 (en) * | 2002-12-18 | 2007-05-08 | General Electric Company | HID lamp having material free dosing tube seal |
| US7132797B2 (en) * | 2002-12-18 | 2006-11-07 | General Electric Company | Hermetical end-to-end sealing techniques and lamp having uniquely sealed components |
| US7839089B2 (en) * | 2002-12-18 | 2010-11-23 | General Electric Company | Hermetical lamp sealing techniques and lamp having uniquely sealed components |
| JP4553736B2 (ja) * | 2002-12-20 | 2010-09-29 | コーニンクレッカ フィリップス エレクトロニクス エヌ ヴィ | 高圧ガス放電ランプ及び高圧ガス放電ランプを備えた照明ユニット |
| JP4342810B2 (ja) | 2003-02-25 | 2009-10-14 | ハリソン東芝ライティング株式会社 | 高圧金属蒸気放電ランプ点灯装置および自動車用前照灯装置 |
| US20050104501A1 (en) * | 2003-04-04 | 2005-05-19 | Transworld Lighting, Inc. | High efficiency gas discharge lamps |
| JP4317908B2 (ja) * | 2003-11-07 | 2009-08-19 | ハリソン東芝ライティング株式会社 | 自動車前照灯・赤外暗視装置兼用メタルハライドランプおよびメタルハライドランプ点灯装置 |
| JP4086158B2 (ja) | 2003-12-22 | 2008-05-14 | 株式会社小糸製作所 | 放電ランプ装置用水銀フリーアークチューブ |
| CN1918687A (zh) * | 2004-02-12 | 2007-02-21 | 株式会社杰士汤浅 | 陶瓷金属卤化物灯、其使用方法及照明器具 |
| DE102004024063A1 (de) * | 2004-05-13 | 2005-12-01 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Hochdruckentladungslampe |
| EP1754247A2 (de) * | 2004-05-27 | 2007-02-21 | Philips Intellectual Property & Standards GmbH | Niederdruck-entladungslampe mit einem entladungsaufrechterhaltungsmittel |
| JP2006120599A (ja) * | 2004-09-21 | 2006-05-11 | Osram Melco Toshiba Lighting Kk | 金属蒸気放電ランプおよび金属蒸気放電ランプ点灯装置 |
| JP4488856B2 (ja) * | 2004-09-27 | 2010-06-23 | スタンレー電気株式会社 | 水銀フリーメタルハライドランプ |
| US7358666B2 (en) | 2004-09-29 | 2008-04-15 | General Electric Company | System and method for sealing high intensity discharge lamps |
| WO2006046704A1 (ja) * | 2004-10-29 | 2006-05-04 | Toshiba Lighting & Technology Corporation | メタルハライドランプおよび照明装置 |
| US7256546B2 (en) * | 2004-11-22 | 2007-08-14 | Osram Sylvania Inc. | Metal halide lamp chemistries with magnesium and indium |
| CN101438094A (zh) * | 2004-12-17 | 2009-05-20 | 皇家飞利浦电子股份有限公司 | 多用途照明单元 |
| US20060132043A1 (en) * | 2004-12-20 | 2006-06-22 | Srivastava Alok M | Mercury-free discharge compositions and lamps incorporating gallium |
| US7944148B2 (en) * | 2004-12-20 | 2011-05-17 | General Electric Company | Mercury free tin halide compositions and radiation sources incorporating same |
| US7825598B2 (en) * | 2004-12-20 | 2010-11-02 | General Electric Company | Mercury-free discharge compositions and lamps incorporating Titanium, Zirconium, and Hafnium |
| US7847484B2 (en) * | 2004-12-20 | 2010-12-07 | General Electric Company | Mercury-free and sodium-free compositions and radiation source incorporating same |
| JP2006244735A (ja) * | 2005-02-28 | 2006-09-14 | Toshiba Lighting & Technology Corp | 高圧放電ランプおよび光学機器 |
| US7245075B2 (en) * | 2005-04-11 | 2007-07-17 | Osram Sylvania Inc. | Dimmable metal halide HID lamp with good color consistency |
| US7615929B2 (en) | 2005-06-30 | 2009-11-10 | General Electric Company | Ceramic lamps and methods of making same |
| US7432657B2 (en) * | 2005-06-30 | 2008-10-07 | General Electric Company | Ceramic lamp having shielded niobium end cap and systems and methods therewith |
| US7852006B2 (en) | 2005-06-30 | 2010-12-14 | General Electric Company | Ceramic lamp having molybdenum-rhenium end cap and systems and methods therewith |
| ITTO20050585A1 (it) * | 2005-08-23 | 2007-02-24 | Space Cannon Vh Srl | Lampada a scarica, in particolare alimentata da corrente continua |
| JP2007115653A (ja) * | 2005-09-22 | 2007-05-10 | Toshiba Lighting & Technology Corp | 高圧放電ランプ、高圧放電ランプ点灯装置および照明装置 |
| JP2007115652A (ja) * | 2005-09-22 | 2007-05-10 | Toshiba Lighting & Technology Corp | 高圧放電ランプおよび照明装置 |
| JP2007134086A (ja) * | 2005-11-08 | 2007-05-31 | Sony Corp | 高圧放電ランプ |
| JP2009516331A (ja) * | 2005-11-14 | 2009-04-16 | コーニンクレッカ フィリップス エレクトロニクス エヌ ヴィ | メタルハライドランプに関するループ型フレームアーク管取り付けアセンブリ及びランプ |
| US7378799B2 (en) * | 2005-11-29 | 2008-05-27 | General Electric Company | High intensity discharge lamp having compliant seal |
| US7633228B2 (en) * | 2005-11-30 | 2009-12-15 | General Electric Company | Mercury-free metal halide discharge lamp |
| DE102006048983A1 (de) | 2006-10-17 | 2008-04-24 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Niederdruckentladungslampe |
| JP5045065B2 (ja) * | 2006-11-06 | 2012-10-10 | 岩崎電気株式会社 | セラミックメタルハライドランプ |
| US20080211971A1 (en) * | 2007-01-08 | 2008-09-04 | Luxim Corporation | Color balancing systems and methods |
| US8299709B2 (en) * | 2007-02-05 | 2012-10-30 | General Electric Company | Lamp having axially and radially graded structure |
| US20100109522A1 (en) * | 2007-04-05 | 2010-05-06 | Koninklijke Philips Electronics N.V. | Mercury-free high intensity gas-discharge lamp |
| US7868553B2 (en) * | 2007-12-06 | 2011-01-11 | General Electric Company | Metal halide lamp including a source of available oxygen |
| US20090153053A1 (en) * | 2007-12-18 | 2009-06-18 | General Electric Company | Low mercury ceramic metal halide lamp |
| US7777418B2 (en) * | 2008-04-08 | 2010-08-17 | General Electric Company | Ceramic metal halide lamp incorporating a metallic halide getter |
| JP2009289518A (ja) * | 2008-05-28 | 2009-12-10 | Koito Mfg Co Ltd | 自動車用水銀フリー放電バルブ |
| JP5242433B2 (ja) * | 2009-01-29 | 2013-07-24 | 株式会社小糸製作所 | 放電ランプ装置用水銀フリーアークチューブ |
| DE102009009890A1 (de) * | 2009-02-20 | 2010-08-26 | Osram Gesellschaft mit beschränkter Haftung | Hochdruckentladungslampe |
| DE102010039221A1 (de) * | 2010-08-11 | 2012-02-16 | Osram Ag | Verfahren zum Betreiben einer Hochdruckentladungslampe außerhalb ihres nominalen Leistungsbereiches |
| US8497633B2 (en) | 2011-07-20 | 2013-07-30 | General Electric Company | Ceramic metal halide discharge lamp with oxygen content and metallic component |
| JP5874589B2 (ja) * | 2012-09-18 | 2016-03-02 | 岩崎電気株式会社 | セラミックメタルハライドランプ |
| RU208591U1 (ru) * | 2020-12-02 | 2021-12-24 | Игорь Георгиевич Рудой | Газоразрядная лампа ультрафиолетового диапазона спектра |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| HU172230B (hu) * | 1976-04-07 | 1978-07-28 | Egyesuelt Izzolampa | Razrjadnyj istochnik sveta vysokogo davlenija s metallo-galogennoj dobavkoj |
| DE2909605A1 (de) * | 1979-03-12 | 1980-09-25 | Patra Patent Treuhand | Vorschaltanordnung zum betreiben einer entladungslampe |
| US4373146A (en) * | 1980-10-20 | 1983-02-08 | Gte Products Corporation | Method and circuit for operating discharge lamp |
| US4647821A (en) * | 1984-09-04 | 1987-03-03 | Gte Laboratories Incorporated | Compact mercury-free fluorescent lamp |
| JPH03152852A (ja) * | 1989-11-08 | 1991-06-28 | Matsushita Electric Works Ltd | 高輝度放電ランプ及び無電極放電灯装置 |
| JPH04303549A (ja) * | 1991-03-30 | 1992-10-27 | Toshiba Lighting & Technol Corp | 高周波点灯式放電ランプ |
| DE4132530A1 (de) * | 1991-09-30 | 1993-04-01 | Patent Treuhand Ges Fuer Elektrische Gluehlampen Mbh | Hochdruckentladungslampe kleiner leistung |
| US5491387A (en) * | 1992-06-29 | 1996-02-13 | Kansei Corporation | Discharge lamp lighting circuit for increasing electric power fed in initial lighting of the lamp |
| EP0609477B1 (de) * | 1993-02-05 | 1999-05-06 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Keramisches Entladungsgefäss für Hochdruckentladungslampe und Herstellungsverfahren derselben und damit verbundene Dichtungsmaterialien |
| KR950001852A (ko) * | 1993-06-01 | 1995-01-04 | 에프.제이.스미트 | 고압금속 할로겐 램프 |
| US5323090A (en) * | 1993-06-02 | 1994-06-21 | Lestician Ballast, Inc. | Lighting system with variable control current sensing ballast |
| DE4334074A1 (de) * | 1993-10-06 | 1995-04-13 | Patent Treuhand Ges Fuer Elektrische Gluehlampen Mbh | Metallhalogenidentladungslampe |
| TW323379B (de) * | 1994-01-18 | 1997-12-21 | Patent Treuhand Ges Fuer Elektrische Gluehlampen Mbh | |
| US5565741A (en) * | 1994-03-16 | 1996-10-15 | Osram Sylvania Inc. | Method of operating a neon discharge lamp particularly useful on a vehicle |
| DE69713239T2 (de) * | 1996-03-22 | 2003-01-09 | Koninklijke Philips Electronics N.V., Eindhoven | Hochdruck metallhalogenidlampe |
-
1997
- 1997-07-21 DE DE19731168A patent/DE19731168A1/de not_active Withdrawn
-
1998
- 1998-06-18 EP EP98111187A patent/EP0903770B1/de not_active Expired - Lifetime
- 1998-06-18 AT AT98111187T patent/ATE274236T1/de not_active IP Right Cessation
- 1998-06-18 DE DE59811826T patent/DE59811826D1/de not_active Expired - Lifetime
- 1998-07-17 US US09/118,491 patent/US6069456A/en not_active Expired - Lifetime
- 1998-07-20 CA CA002243737A patent/CA2243737C/en not_active Expired - Fee Related
- 1998-07-20 HU HU9801641A patent/HU221394B1/hu not_active IP Right Cessation
- 1998-07-21 JP JP20567698A patent/JP4335332B2/ja not_active Expired - Fee Related
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1011126A3 (de) * | 1998-12-14 | 2006-08-23 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Metallhalogenidlampe |
| EP1045622A3 (de) * | 1999-04-14 | 2003-10-29 | Osram Sylvania Inc. | Vorschaltgerät für eine Bogenlampe |
| US6608444B2 (en) | 2000-05-26 | 2003-08-19 | Matsushita Electric Industrial Co., Ltd. | Mercury-free high-intensity discharge lamp operating apparatus and mercury-free metal halide lamp |
| US6586891B2 (en) | 2000-06-06 | 2003-07-01 | Matsushita Electric Industrial Co., Ltd. | High-intensity discharge lamp and high-intensity discharge lamp operating apparatus |
| EP1271612A3 (de) * | 2001-06-19 | 2006-04-19 | Patent-Treuhand-Gesellschaft für elektrische Glühlampen mbH | Hochdruckentladungslampe |
| US7126281B2 (en) | 2003-03-19 | 2006-10-24 | Patent-Treuhand-Gesellschaft für elektrishe Glūhlampen mbH | High-pressure discharge lamp for vehicle headlights |
Also Published As
| Publication number | Publication date |
|---|---|
| HU9801641D0 (en) | 1998-09-28 |
| HUP9801641A2 (hu) | 1999-04-28 |
| EP0903770B1 (de) | 2004-08-18 |
| CA2243737A1 (en) | 1999-01-21 |
| US6069456A (en) | 2000-05-30 |
| CA2243737C (en) | 2006-11-28 |
| HUP9801641A3 (en) | 2001-02-28 |
| JP4335332B2 (ja) | 2009-09-30 |
| ATE274236T1 (de) | 2004-09-15 |
| DE19731168A1 (de) | 1999-01-28 |
| DE59811826D1 (de) | 2004-09-23 |
| JPH1186795A (ja) | 1999-03-30 |
| EP0903770A3 (de) | 1999-04-07 |
| HU221394B1 (en) | 2002-09-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0903770B1 (de) | Metallhalogenidlampe | |
| EP0314732B1 (de) | Xenon-kurzbogen-entlandungslampe | |
| EP0834905B1 (de) | Natriumhochdrucklampe kleiner Leistung | |
| DE69324047T2 (de) | Elektrodenlose Entladungslampe | |
| DE19857585A1 (de) | Metallhalogenidlampe | |
| DE1464181B2 (de) | Elektrische hochdruck dampfentladungslampe | |
| DE1589171B1 (de) | Natriumdampflampe hoher intensitaet mit quecksilber | |
| EP0733266A1 (de) | Verfahren zum betreiben einer inkohärent emittierenden strahlungsquelle | |
| DE19801485A1 (de) | Halogen-Metalldampflampe | |
| DE10243867A1 (de) | Quecksilberfreie Bogenentladungsröhre für Entladungslampeneinheit | |
| DE19901987A1 (de) | Metallhalogenidlampe mit Zündhilfe | |
| DE2930328C2 (de) | Verwendung eines Zündgases aus einer Penning-Mischung | |
| DE2359138A1 (de) | Quecksilber-metallhalogenid-entladungslampen | |
| EP2499657B1 (de) | Quecksilberfreie hochdruckentladungslampe mit reduziertem zinkhalogenidanteil | |
| DE69402107T2 (de) | Hochdruckmetallhalogenidentladungslampe | |
| EP2347430B1 (de) | Quecksilberfreie entladungslampe | |
| DE10209424A1 (de) | Quecksilber-Kurzbogenlampe | |
| DE68916346T2 (de) | Metallhalogenidentladungslampe mit verbesserter Farbwiedergabe. | |
| EP0269958A2 (de) | Einseitig gequetschte Hochdruckentladungslampe | |
| DE1639113B1 (de) | Dampfentladungslampe fuer photochemische Zwecke | |
| DE3044121A1 (de) | Natriumhochdrucklampe | |
| DE3687667T2 (de) | Hochdrucknatriumdampflampe und ternaeres amalgam dafuer. | |
| DE748762C (de) | Elektrische Hochdruckentladungslampe mit flachem Entladungsrohr | |
| DE1464181C (de) | Elektrische Hochdruck Dampfentladungs lampe | |
| DE1589171C (de) | Natriumdampflampe hoher Intensität mit Quecksilber |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE CH DE FR GB IT LI NL |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| 17P | Request for examination filed |
Effective date: 19990505 |
|
| AKX | Designation fees paid |
Free format text: AT BE CH DE FR GB IT LI NL |
|
| 17Q | First examination report despatched |
Effective date: 20021030 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| RTI1 | Title (correction) |
Free format text: METAL HALIDE LAMP |
|
| RTI1 | Title (correction) |
Free format text: METAL HALIDE LAMP |
|
| RTI1 | Title (correction) |
Free format text: METAL HALIDE LAMP |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE FR GB IT LI NL |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: SIEMENS SCHWEIZ AG Ref country code: CH Ref legal event code: EP |
|
| REF | Corresponds to: |
Ref document number: 59811826 Country of ref document: DE Date of ref document: 20040923 Kind code of ref document: P |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 20041209 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20050519 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20060517 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20060912 Year of fee payment: 9 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070630 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070630 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20110630 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20110617 Year of fee payment: 14 Ref country code: GB Payment date: 20110613 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20110627 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20110819 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20110713 Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 59811826 Country of ref document: DE Owner name: OSRAM GMBH, DE Free format text: FORMER OWNER: OSRAM GESELLSCHAFT MIT BESCHRAENKTER HAFTUNG, 81543 MUENCHEN, DE Effective date: 20111130 |
|
| BERE | Be: lapsed |
Owner name: *PATENT-TREUHAND-G.- FUR ELEKTRISCHE GLUHLAMPEN M. Effective date: 20120630 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20130101 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20120618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120618 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 59811826 Country of ref document: DE Owner name: OSRAM GMBH, DE Free format text: FORMER OWNER: OSRAM AG, 81543 MUENCHEN, DE Effective date: 20130205 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20130228 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 59811826 Country of ref document: DE Effective date: 20130101 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120702 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120630 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120618 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130101 Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130101 |