EP0752012B1 - Process for producing magnetic steel sheets with a glass coating - Google Patents
Process for producing magnetic steel sheets with a glass coating Download PDFInfo
- Publication number
- EP0752012B1 EP0752012B1 EP95912252A EP95912252A EP0752012B1 EP 0752012 B1 EP0752012 B1 EP 0752012B1 EP 95912252 A EP95912252 A EP 95912252A EP 95912252 A EP95912252 A EP 95912252A EP 0752012 B1 EP0752012 B1 EP 0752012B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- annealing separator
- additive
- mgo
- strip
- annealing
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment
- C21D8/12—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment
- C21D8/12—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties
- C21D8/1277—Modifying the physical properties of ferrous metals or ferrous alloys by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties involving a particular surface treatment
- C21D8/1283—Application of a separating or insulating coating
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22F—CHANGING THE PHYSICAL STRUCTURE OF NON-FERROUS METALS AND NON-FERROUS ALLOYS
- C22F1/00—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22F—CHANGING THE PHYSICAL STRUCTURE OF NON-FERROUS METALS AND NON-FERROUS ALLOYS
- C22F1/00—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working
- C22F1/10—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working of nickel or cobalt or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23D—ENAMELLING OF, OR APPLYING A VITREOUS LAYER TO, METALS
- C23D5/00—Coating with enamels or vitreous layers
- C23D5/02—Coating with enamels or vitreous layers by wet methods
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F1/00—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties
- H01F1/01—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials
- H01F1/03—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity
- H01F1/12—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials
- H01F1/14—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials metals or alloys
- H01F1/16—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials metals or alloys in the form of sheets
- H01F1/18—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials metals or alloys in the form of sheets with insulating coating
Definitions
- the invention relates to a method for producing Electrical sheets, especially grain-oriented ones Electrical sheets, with an evenly adhering Glass film and with improved magnetic properties, in which the hot strip initially produced and possibly annealed except for the final cold strip thickness with at least one Cold rolling stage is cold rolled, then to the an annealing separator rolled to the final thickness is applied and dried and then the cold-rolled strip coated in a high-temperature annealing is subjected, with an integral part of the Glow separator an aqueous magnesium oxide (MgO) dispersion and the glow separator is additionally at least one Has additive.
- MgO aqueous magnesium oxide
- decarburization annealing is carried out after rolling to the final thickness.
- the carbon is extracted from the material.
- An oxide layer forms as a base layer on the strip surface, the essential components of which are silicon dioxide (SiO 2 ) and fayalite (Fe 2 SiO 4 ).
- the strip is coated with a protective layer and subjected to a long-term annealing in the coil.
- the adhesive protection layer is intended to prevent the individual coil turns from sticking together during long-term annealing and, on the other hand, to form an insulation layer (glass film) with the base layer on the strip surface.
- the adhesive protection layer consists essentially of magnesium oxide (MgO).
- the MgO is slurried in water in the form of a powder, applied to the belt and dried. During this process, part of the magnesium oxide reacts with the water to form magnesium hydroxide (Mg (OH) 2 ). The amount of water bound to magnesium hydroxide, based on the total amount of oxide powder, is called the loss on ignition.
- Equation (I) shows the dehydration of the magnesium hydroxide, which starts at about 350 ° C. It is important for an optimally running process, both in terms of the insulation and the development of the magnetic properties, that the amount of water released is within certain limits.
- the water humidifies the predominantly hydrogen-containing annealing atmosphere and thus sets a corresponding oxidation potential.
- the annealing atmosphere must not be too dry because the glass film would be made too thin under such conditions. However, it must also not become too moist, because then it will be reoxidized too much and the glass film will have defects such as local flaking and poor adhesion.
- additives to MgO powder were introduced to improve the formation of the insulation layer and the magnetic properties of the finished product.
- These include titanium dioxide (TiO 2 ), boron compounds such as boron oxide (B 2 O 3 ) or sodium tetraborate (Na 2 B 4 O 7 ), as well as antimony compounds such as antimony sulfate (Sb 2 (SO 4 ) 3 ) in combination with a chloride, preferably antimony chloride SbCl 3 .
- TiO 2 titanium dioxide
- boron compounds such as boron oxide (B 2 O 3 ) or sodium tetraborate (Na 2 B 4 O 7 )
- antimony compounds such as antimony sulfate (Sb 2 (SO 4 ) 3 ) in combination with a chloride, preferably antimony chloride SbCl 3 .
- the additives used often also have disadvantages that reduce the product quality. Overall, the processing of such additives is cumbersome, since some of them have to be dissolved in previously
- the invention is based on the object of measures to meet, especially by modifying the Glow separators to the insulation properties and at the same time the magnetic properties of the Finished product to improve further.
- the Anti-adhesive layer can be applied more homogeneously quality-reducing phenomena, such as glow contours and to avoid local defects.
- one should easy handling can be guaranteed and the cost, am Standard measured, kept low.
- a readily water-soluble one Sodium phosphate compound is used.
- a readily water soluble sodium phosphate compound and a finely dispersed oxidic aluminum compound Glow separator can be added.
- the good water solubility of the sodium phosphate compound if necessary in combination with the finely dispersed distribution of the oxidic aluminum compound in the specified amounts ensure a homogeneous application of the adhesive protection, prevent coagulation within the aqueous Magnesium oxide dispersion and associated local Defects in the glass film and promote those in the Long-term annealing chemical reactions between the one on the belt surface Base layer and the adhesive protective layer to the glass film.
- a stronger than the standard Glass film formation that the interaction between the Annealing atmosphere and the strips are positively influenced the magnetic properties of the electrical sheets improved.
- a method with the generic measures has been known from EP 0 232 537 B1.
- a titanium compound such as TiO 2
- a boron compound such as B 2 O 3
- a sulfur compound such as SrS
- the MgO-based annealing separator is added to the MgO-based annealing separator as an additive, with the aim of improving the insulation properties, such as Adhesion and the appearance of the glass film to influence positively. This is achieved by hydrating the coating.
- the magnetic properties were also improved by the addition of such additives.
- JP-5-513 8021 describes a release agent based on MgO, which contains up to 50% by weight of Mg (OH) 2 and up to 5% by weight of Al (OH) 3 or Al (NO 3 ) 3 contains. It has been shown that the use of such a release agent for coating electrical sheets does not have an adverse effect on the magnetic properties of the product.
- JP-5-247 661 describes a process for the production of grain-oriented silicon steels which are coated with a glass layer.
- improvements in the magnetic properties and the surface properties of the coated steel are achieved by using release agent additives such as Sb 2 (SO 4 ) 3 , V 2 O s , SrS, Na 2 B 4 O 7 and Ca (H 2 PO 4 ) 2 .
- the positive influence on which the invention is based the magnetic properties is characteristic of the sodium phosphates.
- FIG. 1 shows the superiority of the samples produced by the method according to the invention with an MgO-based adhesive protection doped with sodium phosphate over other phosphate additives.
- HGO high permeability grain oriented tape samples were coated with MgO + 6% TiO 2 + the listed additives, dried and annealed.
- the sodium phosphates are readily water-soluble, allow thus an optimally homogeneous distribution within the Anti-adhesive layer.
- Sodium phosphates in the present case in particular using the example of Sodium pyrophosphate decahydrate are reported both the magnetic properties polarization and Magnetic loss, as well Isolation training improved.
- the Inhibitor test method is demonstrated that the Sodium pyrophosphate to a prematurely stronger one Glass film formation leads.
- the inhibitor test stops Process in which, in principle, high annealing certain annealing temperatures are canceled and the Samples can be assessed magnetically. In the present case the insulation training was also assessed.
- the magnetic properties of loss of magnetization P 1.7 and polarization J 800 were determined on the annealed strips.
- the aluminum compounds used as a further additive in addition to the sodium phosphate compound are aluminum oxides or hydroxides of the form Al 2 O 3 , AlO (OH) 3 and AL (OH), the effect of which is fully exploited when the corresponding particle sizes are small. The effect is particularly evident when the compounds are added in the form of sols (very fine particles / water mixtures).
- the average particle size should be less than 100 nm with the narrowest possible particle size distribution.
- the addition of these aluminum compounds leads to a considerable improvement in loss, similar to the case with the addition of titanium dioxide.
- the advantage of the aluminum compound as an additive over titanium dioxide is the lower dosage and the more homogeneous distribution of the particles. Another advantage lies in the fact that the aluminum compounds added also have the property of a ceramic binder, and the adhesive protective layer therefore adheres better to the tape.
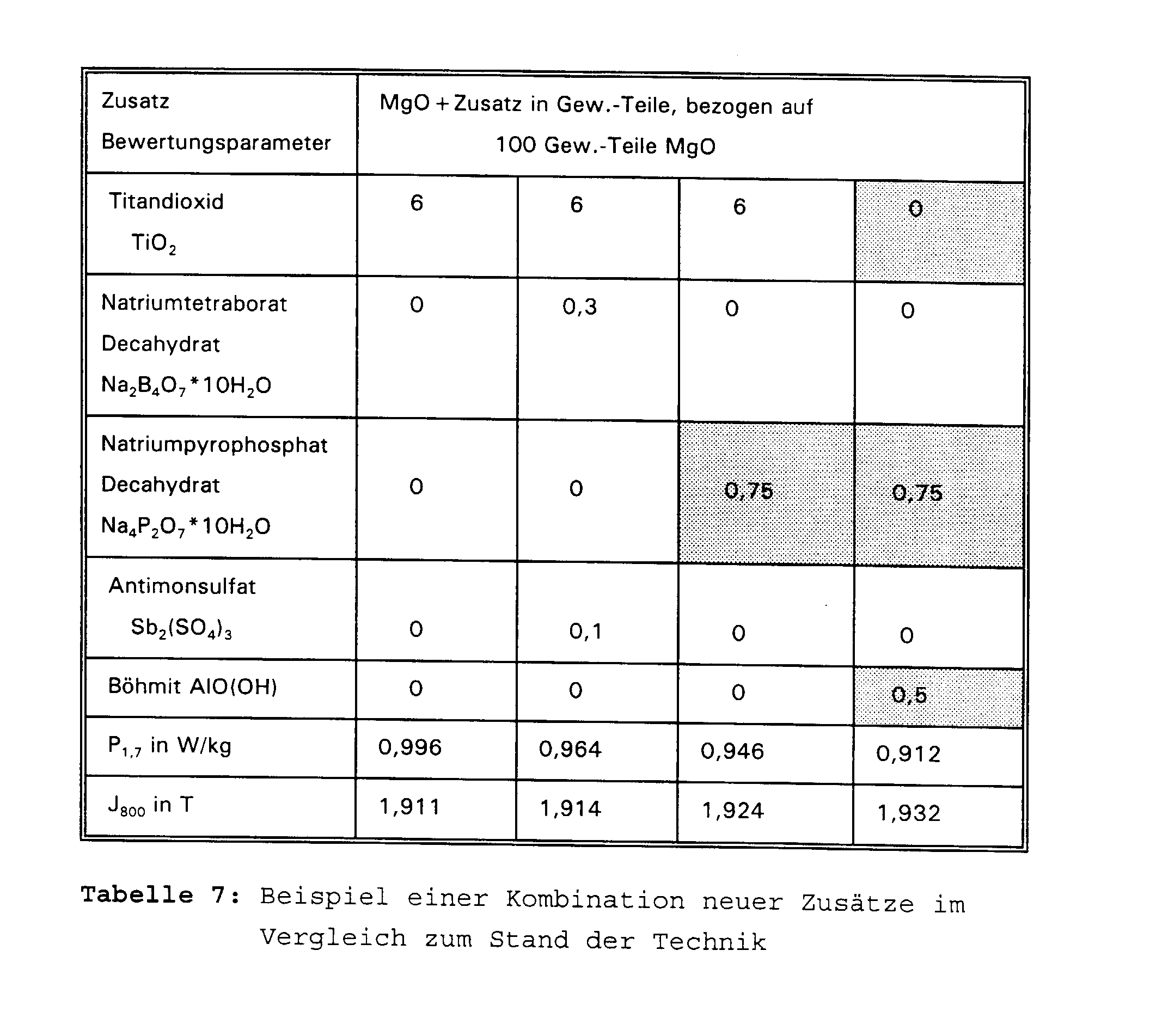
- Table 6 and Figure 3 show the influence of the selected aluminum compounds on the magnetic loss. Influence of different oxidic aluminum compounds on the magnetic properties and the glass film appearance
- Boehmite AIO (OH) 0 0.5 2nd Glass film appearance
- the effect of the above additives is optimized, if suitable combinations of additives are used. This also has positive effects in combination with additives already used, such as titanium dioxide, Antimony sulfate and sodium tetraborate achieved. Related to the slurry properties and thus the homogeneity of the MgO layer turns out to be a combination of one finely dispersed oxidic aluminum compound and one well water-soluble sodium phosphate as optimal because with these additives significantly fewer local defects to be observed.
- the magnetic properties of loss of magnetization P 1.7 and polarization J 800 were determined on the annealed strips.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Physics & Mathematics (AREA)
- Mechanical Engineering (AREA)
- Organic Chemistry (AREA)
- Metallurgy (AREA)
- Materials Engineering (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Electromagnetism (AREA)
- Manufacturing & Machinery (AREA)
- Power Engineering (AREA)
- Dispersion Chemistry (AREA)
- Chemical Treatment Of Metals (AREA)
- Soft Magnetic Materials (AREA)
- Manufacturing Of Steel Electrode Plates (AREA)
- Cell Separators (AREA)
- Glass Compositions (AREA)
- Laminated Bodies (AREA)
- Insulating Bodies (AREA)
- Inorganic Insulating Materials (AREA)
Abstract
Description
Die Erfindung betrifft ein Verfahren zur Herstellung von Elektroblechen, insbesondere von kornorientierten Elektroblechen, mit einem gleichmäßigen gut haftenden Glasfilm und mit verbesserten magnetischen Eigenschaften, bei dem das zunächst erzeugte und ggf. geglühte Warmband bis auf die Kaltband-Enddicke mit mindestens einer Kaltwalzstufe kaltgewalzt wird, anschließend auf das bis auf die Enddicke gewalzte Band ein Glühseparator aufgebracht und getrocknet wird und im Anschluß daran das so beschichtete Kaltband einer Hochtemperaturglühung unterworfen wird, wobei wesentlicher Bestandteil des Glühseparators eine wäßrige Magnesiumoxid(MgO)-Dispersion ist und der Glühseparator zusätzlich mindestens ein Additiv aufweist.The invention relates to a method for producing Electrical sheets, especially grain-oriented ones Electrical sheets, with an evenly adhering Glass film and with improved magnetic properties, in which the hot strip initially produced and possibly annealed except for the final cold strip thickness with at least one Cold rolling stage is cold rolled, then to the an annealing separator rolled to the final thickness is applied and dried and then the cold-rolled strip coated in a high-temperature annealing is subjected, with an integral part of the Glow separator an aqueous magnesium oxide (MgO) dispersion and the glow separator is additionally at least one Has additive.
Bei der Fertigung von kornorientiertem Elektroblech wird nach dem Walzen auf Enddicke eine Entkohlungsglühung durchgeführt. Dabei wird dem Werkstoff der Kohlenstoff entzogen. An der Bandoberfläche bildet sich dabei eine Oxidschicht als Grundschicht, deren wesentliche Bestandteile Siliziumdioxid (SiO2) und Fayalit (Fe2SiO4) sind. Im Anschluß an die Entkohlungsglühung wird das Band mit einer Klebschutzschicht beschichtet und im Coil einer Langzeitglühung unterzogen. Die Klebschutzschicht soll zum einen das Zusammenkleben der einzelnen Coilwindungen während der Langzeitglühung verhindern und zum anderen mit der Grundschicht auf der Bandoberfläche eine Isolationsschicht (Glasfilm) bilden. Die Klebschutzschicht besteht im wesentlichen aus Magnesiumoxid (MgO). Das MgO wird in Form eines Pulvers in Wasser aufgeschlämmt, auf das Band aufgetragen und getrocknet. Bei diesem Vorgang reagiert ein Teil des Magnesiumoxides mit dem Wasser zu Magnesiumhydroxid (Mg(OH)2). Die an Magnesiumhydroxid gebundene Menge Wasser, bezogen auf die Gesamtoxidpulvermenge, wird als Glühverlust bezeichnet.In the production of grain-oriented electrical sheet, decarburization annealing is carried out after rolling to the final thickness. The carbon is extracted from the material. An oxide layer forms as a base layer on the strip surface, the essential components of which are silicon dioxide (SiO 2 ) and fayalite (Fe 2 SiO 4 ). Following the decarburization annealing, the strip is coated with a protective layer and subjected to a long-term annealing in the coil. On the one hand, the adhesive protection layer is intended to prevent the individual coil turns from sticking together during long-term annealing and, on the other hand, to form an insulation layer (glass film) with the base layer on the strip surface. The adhesive protection layer consists essentially of magnesium oxide (MgO). The MgO is slurried in water in the form of a powder, applied to the belt and dried. During this process, part of the magnesium oxide reacts with the water to form magnesium hydroxide (Mg (OH) 2 ). The amount of water bound to magnesium hydroxide, based on the total amount of oxide powder, is called the loss on ignition.
Die auf die Isolation bezogenen wesentlichen Abläufe und Reaktionen zwischen Bandoberfläche und Klebschutzschicht während der Langzeitglühung sind nachfolgend vereinfacht zusammengefaßt:The essential processes and related to isolation Reactions between the tape surface and the protective layer during long-term annealing are simplified below summarized:
In der Vergangenheit wurden eine Reihe von Zusätzen zum MgO-Pulver eingeführt, die die Ausbildung der Isolationsschicht und die magnetischen Eigenschaften des Fertigproduktes verbessern sollen. Dazu zählen Titandioxid (TiO2), Borverbindungen, wie Boroxid (B2O3) oder Natriumtetraborat (Na2B4O7), sowie Antimonverbindungen, wie z.B. Antimonsulfat (Sb2(SO4)3) in Kombination mit einem Chlorid, vorzugsweise Antimonchlorid SbCl3. Die eingesetzten Zusätze weisen neben den positiven Einflüssen auf die jeweiligen Zielgrößen allerdings häufig auch Nachteile auf, die die Produktqualität herabsetzen. Insgesamt ist die Verarbeitung derartiger Zusätze umständlich, da diese z.T. in vorher erhitztem Wasser gelöst werden müssen. Besonders bei den schwer wasserlöslichen Salzen Natriumtetraborat und insbesondere Antimonsulfat führen nicht gelöste, grobe Partikel zu Inhomogenitäten in der Klebschutzschicht und nachfolgend zu lokalen Fehlstellen im Glasfilm. Bei Antimonsulfat kommt hinzu, daß die Verbindung teuer ist und in die Kategorie der "minder giftigen" Substanzen eingestuft wird. Eine inhomogene Verteilung von Titandioxid im Klebschutz führt zu Fehlstellen im Glasfilm.In the past, a number of additives to MgO powder were introduced to improve the formation of the insulation layer and the magnetic properties of the finished product. These include titanium dioxide (TiO 2 ), boron compounds such as boron oxide (B 2 O 3 ) or sodium tetraborate (Na 2 B 4 O 7 ), as well as antimony compounds such as antimony sulfate (Sb 2 (SO 4 ) 3 ) in combination with a chloride, preferably antimony chloride SbCl 3 . In addition to the positive influences on the respective target values, the additives used often also have disadvantages that reduce the product quality. Overall, the processing of such additives is cumbersome, since some of them have to be dissolved in previously heated water. Especially with the poorly water-soluble salts sodium tetraborate and in particular antimony sulfate, undissolved, coarse particles lead to inhomogeneities in the adhesive protective layer and subsequently to local defects in the glass film. In addition, with antimony sulfate, the compound is expensive and is classified in the category of "less toxic" substances. An inhomogeneous distribution of titanium dioxide in the adhesive protection leads to defects in the glass film.
Der Erfindung liegt nun die Aufgabe zugrunde, Maßnahmen zu treffen, insbesondere durch Modifizierung des Glühseparators, um die Isolationseigenschaften und gleichzeitig die magnetischen Eigenschaften des Fertigprodukts weiter zu verbessern. Dabei soll die Klebschutzschicht homogener aufgetragen werden können, um qualitätsmindernde Erscheinungen, wie Glühkonturen und lokale Fehlstellen, zu vermeiden. Daneben soll eine einfache Handhabung gewährleistet sein und die Kosten, am Standard gemessen, niedrig gehalten werden. The invention is based on the object of measures to meet, especially by modifying the Glow separators to the insulation properties and at the same time the magnetic properties of the Finished product to improve further. Thereby the Anti-adhesive layer can be applied more homogeneously quality-reducing phenomena, such as glow contours and to avoid local defects. In addition, one should easy handling can be guaranteed and the cost, am Standard measured, kept low.
Zur Lösung dieser Aufgabe wird bei dem gattungsgemäßen Verfahren erfindungsgemäß vorgeschlagen, daß als mindestens ein Additiv eine gut wasserlösliche Natriumphosphatverbindung verwendet wird. Gemäß einer weiteren vorteilhaften Ausgestaltung des erfindungsgemäßen Verfahrens können als weitere Additive eine gut wasserlösliche Natriumphosphatverbindung und eine feindisperse oxidische Aluminiumverbindung dem Glühseparator zugesetzt werden.To solve this problem is the generic Method proposed according to the invention that as at least one additive is a readily water-soluble one Sodium phosphate compound is used. According to one another advantageous embodiment of the The inventive method can be used as further additives a readily water soluble sodium phosphate compound and a finely dispersed oxidic aluminum compound Glow separator can be added.
Die gute Wasserlöslichkeit der Natriumphosphatverbindung, ggfs. in Kombination mit der feindispersen Verteilung der oxidischen Aluminiumverbindung in den angegebenen Mengen gewährleisten eine homogene Auftragung des Klebschutzes, verhindern Koagulationen innerhalb der wäßrigen Magnesiumoxiddispersion und damit verbundene lokale Fehlstellen im Glasfilm und fördern die in der Langzeitglühung ablaufenden chemischen Reaktionen zwischen der auf der Bandoberfläche befindlichen Grundschicht und der Klebschutzschicht zum Glasfilm. Durch eine gegenüber dem Standard stärker einsetzende Glasfilmbildung, die die Wechselwirkung zwischen der Glühatmosphäre und den Bändern positiv beeinflußt, werden die magnetischen Eigenschaften der Elektrobleche verbessert.The good water solubility of the sodium phosphate compound, if necessary in combination with the finely dispersed distribution of the oxidic aluminum compound in the specified amounts ensure a homogeneous application of the adhesive protection, prevent coagulation within the aqueous Magnesium oxide dispersion and associated local Defects in the glass film and promote those in the Long-term annealing chemical reactions between the one on the belt surface Base layer and the adhesive protective layer to the glass film. By using a stronger than the standard Glass film formation that the interaction between the Annealing atmosphere and the strips are positively influenced the magnetic properties of the electrical sheets improved.
Ein Verfahren mit den gattungsgemäßen Maßnahmen ist aus
der EP 0 232 537 B1 bekannt gewesen. Bei diesem bekannten
Verfahren wird dem Glühseparator auf MgO-Basis als
Additiv eine Titanverbindung, wie TiO2, und/oder eine
Borverbindung, wie B2O3, und/oder eine Schwefelverbindung,
wie SrS, mit dem Ziel zugesetzt, die Isolationseigenschaften,
wie Haftung und das Aussehen des Glasfilms,
positiv zu beeinflussen. Erreicht wird dies durch eine
Hydratation der Beschichtung. Auch die magnetischen
Eigenschaften wurden durch den Zusatz solcher Additive
verbessert.A method with the generic measures has been known from
In der JP-5-513 8021 wird ein Trennmittel auf der Basis von MgO beschrieben, welches als Zusätze bis zu 50 Gew.-% Mg(OH)2 sowie bis zu 5 Gew.-% Al(OH)3 oder Al(NO3)3 enthält. Es wurde gezeigt, daß sich die Verwendung eines solchen Trennmittels zur Beschichtung von Elektroblechen nicht ungünstig auf die magnetischen Eigenschaften des Produkts auswirken.JP-5-513 8021 describes a release agent based on MgO, which contains up to 50% by weight of Mg (OH) 2 and up to 5% by weight of Al (OH) 3 or Al (NO 3 ) 3 contains. It has been shown that the use of such a release agent for coating electrical sheets does not have an adverse effect on the magnetic properties of the product.
Die JP-5-247 661 beschreibt ein Verfahren zur Herstellung von kornorientierten Siliciumstählen, die mit einer Glasschicht beschichtet sind. Bei diesem Verfahren werden durch Verwendung von Trennmitteladditiven wie Sb2(SO4)3, V2Os, SrS, Na2B4O7 und Ca(H2PO4)2 Verbesserungen der magnetischen Eigenschaften sowie der Obenflächenbeschaffenheit des beschichteten Stahls erzielt.JP-5-247 661 describes a process for the production of grain-oriented silicon steels which are coated with a glass layer. In this process, improvements in the magnetic properties and the surface properties of the coated steel are achieved by using release agent additives such as Sb 2 (SO 4 ) 3 , V 2 O s , SrS, Na 2 B 4 O 7 and Ca (H 2 PO 4 ) 2 .
Der der Erfindung zugrunde liegende positive Einfluß auf die magnetischen Eigenschaften ist charakteristisch für die Natriumphosphate.The positive influence on which the invention is based the magnetic properties is characteristic of the sodium phosphates.
Fig. 1 zeigt die Überlegenheit der nach dem erfindungsgemäßen Verfahren hergestellten Proben mit einem Natriumphosphat dotierten Klebschutz auf MgO-Basis gegenüber anderen Phosphatzusätzen. Dabei wurden HGO (high permeability grain oriented) Bandproben mit MgO + 6% TiO2 + den aufgeführten Zusätzen beschichtet, getrocknet und hochgeglüht.FIG. 1 shows the superiority of the samples produced by the method according to the invention with an MgO-based adhesive protection doped with sodium phosphate over other phosphate additives. HGO (high permeability grain oriented) tape samples were coated with MgO + 6% TiO 2 + the listed additives, dried and annealed.
Die Natriumphosphate sind gut wasserlöslich, ermöglichen damit eine optimal homogene Verteilung innerhalb der Klebschutzschicht. Durch die bevorzugte Verwendung der Natriumphosphate, vorliegend insbesondere am Beispiel des Natriumpyrophosphat Decahydrat ausgewiesen, werden sowohl die magnetischen Eigenschaften Polarisation und Ummagnetisierungsverlust, als auch die Isolationsausbildung verbessert. Im Inhibitortestverfahren wird nachgewiesen, daß das Natriumpyrophosphat zu einer vorzeitig stärkeren Glasfilmbildung führt. Der Inhibitortest stellt ein Verfahren dar, bei dem prinzipiell Hochglühungen bei bestimmten Glühtemperaturen abgebrochen werden und die Proben magnetisch beurteilt werden. Im vorliegenden Fall wurden zusätzlich die Isolationsausbildungen bewertet.The sodium phosphates are readily water-soluble, allow thus an optimally homogeneous distribution within the Anti-adhesive layer. Through the preferred use of the Sodium phosphates, in the present case in particular using the example of Sodium pyrophosphate decahydrate are reported both the magnetic properties polarization and Magnetic loss, as well Isolation training improved. in the Inhibitor test method is demonstrated that the Sodium pyrophosphate to a prematurely stronger one Glass film formation leads. The inhibitor test stops Process in which, in principle, high annealing certain annealing temperatures are canceled and the Samples can be assessed magnetically. In the present case the insulation training was also assessed.
Jeweils 3 Bandproben aus 3 Bändern kornorientierten
Elektroblechs der Güte HGO (high permeability
grainoriented) und der Dicke 0,23 mm wurde zum einen mit
einer wäßrigen Magnesiumoxiddispersion und zum anderen
mit einer wäßrigen Magnesiumoxiddispersion, der 0,75 %
Natriumpyrophosphat Decahydrat, bezogen auf 100 %
Magnesiumoxid, zugesetzt wurde, beschichtet. Nachdem die
Bandproben entsprechend dem Stand der Technik hochgeglüht
wurden, wurden die magnetischen Kenngrößen bestimmt.
Tabelle 1 gibt die magnetischen Kenngrößen Polarisation
J800 und Ummagnetisierungsverlust P1,7 zum Vergleich der
beiden Beschichtungen wieder.
6 Bandproben aus kornorientiertem Elektroblech (HGO) der
Nenndicke 0,23 mm, deren chemische Zusammensetzung
willkürlich innerhalb des Analysenbereichs
29 Bandproben aus kornorientiertem Elektroblech (HGO) der
Nenndicke 0,23 mm, deren chemische Zusammensetzung
willkürlich innerhalb des Analysenbereichs
Elektroblechproben der Dicke 0,29 mm und der chemischen
Zusammensetzungen
%
%
%
%
%
%
%
%
%
%
%
%
%
Bänder aus kornorientiertem Elektroblech der Nenndicke
0,23 mm, die im Verfahren nach dem Stand der Technik bis
einschließlich der Entkohlung processiert wurden, wurden
mit einem Trennmittel auf der Basis Magnesiumoxid und
6 Gew.-Teilen Titandioxid, bezogen auf 100 Gew.-Teile
MgO, sowie den in Tabelle 5 aufgeführten Zusätzen
beschichtet und anschließend entsprechend dem Stand der
Technik hochgeglüht. An den hochgeglühten Bändern wurden
die magnetischen Eigenschaften Ummagnetisierungsverlust
P1,7 und Polarisation J800 bestimmt.
Die als weiteres Additiv neben der Natriumphosphatverbindung verwendeten Aluminiumverbindungen sind Aluminiumoxide bzw. -hydroxide der Form Al2O3, AlO(OH)3 und AL(OH) deren Wirkung dann voll ausgeschöpft wird, wenn die entsprechenden Partikelgrößen klein sind. Die Wirkung zeigt sich besonders deutlich, wenn die Verbindungen in Form von Solen (feinste Partikel/Wasser-Gemische) zugegeben werden. Die Partikelgröße sollte im Mittel kleiner als 100nm bei einer möglichst engen Partikelgrößenverteilung sein. Die Zugabe dieser Aluminiumverbindungen führt zu einer erheblichen Verlustverbesserung, ähnlich wie es bei der Zugabe von Titandioxid der Fall ist. Der Vorteil der Aluminiumverbindung als Zusatz gegenüber Titandioxid sind die geringer dosierten Zugaben und die homogenere Verteilung der Partikel. Ein weiterer Vorteil liegt in der Tatsache, daß die zugegebenen Aluminiumverbindungen auch die Eigenschaft eines keramischen Binders haben, die Klebschutzschicht demnach besser am Band haftet.The aluminum compounds used as a further additive in addition to the sodium phosphate compound are aluminum oxides or hydroxides of the form Al 2 O 3 , AlO (OH) 3 and AL (OH), the effect of which is fully exploited when the corresponding particle sizes are small. The effect is particularly evident when the compounds are added in the form of sols (very fine particles / water mixtures). The average particle size should be less than 100 nm with the narrowest possible particle size distribution. The addition of these aluminum compounds leads to a considerable improvement in loss, similar to the case with the addition of titanium dioxide. The advantage of the aluminum compound as an additive over titanium dioxide is the lower dosage and the more homogeneous distribution of the particles. Another advantage lies in the fact that the aluminum compounds added also have the property of a ceramic binder, and the adhesive protective layer therefore adheres better to the tape.
4 Bandproben aus kornorientiertem Elektroblech der
Nenndicke 0,23 mm, deren chemische Zusammensetzung
willkürlich innerhalb des Analysenbereichs
%
%
%
%
%
%
%
%
%
%
%
%
%
%
Die Wirkung der obengenannten Zusätze wird optimiert, wenn geeignete Kombinationen von Zusätzen benutzt werden. Dabei werden auch positive Effekte in Kombination mit bereits eingesetzten Zusätzen, wie Titandioxid, Antimonsulfat und Natriumtetraborat erzielt. Bezogen auf die Slurryeigenschaften und damit auf die Homogenität der MgO-Schicht erweist sich eine Kombination einer feindispersen oxidischen Aluminiumverbindung und eines gut wasserlöslichen Natriumphosphates als optimal, da mit diesen Zusätzen deutlich weniger lokale Fehlstellen beobachtet werden.The effect of the above additives is optimized, if suitable combinations of additives are used. This also has positive effects in combination with additives already used, such as titanium dioxide, Antimony sulfate and sodium tetraborate achieved. Related to the slurry properties and thus the homogeneity of the MgO layer turns out to be a combination of one finely dispersed oxidic aluminum compound and one well water-soluble sodium phosphate as optimal because with these additives significantly fewer local defects to be observed.
Proben aus einem Band aus kornorientiertem Elektroblech der Nenndicke 0,23 mm, die im Verfahren nach dem Stand der Technik bis einschließlich der Entkohlung processiert wurden, wurden mit einem Trennmittel auf der Basis Magnesiumoxid und den in Tabelle 7 aufgeführten Zusätzen beschichtet und anschließend entsprechend dem Stand der Technik hochgeglüht. An den hochgeglühten Bändern wurden die magnetischen Eigenschaften Ummagnetisierungsverlust P1,7 und Polarisation J800 bestimmt. Samples from a strip of grain-oriented electrical sheet with a nominal thickness of 0.23 mm, which were processed in the process according to the prior art up to and including decarburization, were coated with a release agent based on magnesium oxide and the additives listed in Table 7 and then in accordance with the status of technology glowed. The magnetic properties of loss of magnetization P 1.7 and polarization J 800 were determined on the annealed strips.
Claims (7)
- A method for producing electric sheets, in particular grain-oriented electric sheets, with an evenly well-adhering glass film and with improved magnetic properties, in which the hot rolled strip which is produced at first and is optionally annealed is cold-rolled up to the end thickness of the cold strip with at least one cold rolling stage, thereafter an annealing separator is applied to the strip which is rolled up to the end thickness, and is dried, and thereafter the cold strip which is thus coated is subjected to a high-temperature annealing, with a hydrous magnesium oxide (MgO) dispersion being an important component of the annealing separator and the annealing separator being provided with at least one additive, characterised in that a favourably water-soluble sodium phosphate compound is used as at least one additive.
- A method according to claim 1, characterised in that at least two additives are used, namely a favourably water-soluble sodium phosphate compound and a finely dispersed oxidic aluminium compound.
- A method according to claim 1 or 2, characterised in that 0.05 to 4.0 % sodium phosphate, relating to the MgO quantity, is added to the annealing separator as additive.
- A method according to claim 1 or 2, characterised in that 0.3 to 1.5 % of sodium pyrophosphate decahydrate, relating to the MgO quantity, is added to the annealing separator as additive.
- A method according to claims 2, 3 or 4, characterised in that 0.05 to 4.0 % of the finely dispersed oxidic aluminium compound, relating to the MgO quantity, is added to the annealing separator as additive.
- A method according to claims 2, 3, 4 or 5, characterised in that the oxidic aluminium compound is used with a particle size below 100 nm.
- A method according to claim 1 to 6, characterised in that further additives such as titanium dioxide, boron oxide, sodium tetraborate, antimony sulphate, metal chloride, preferably antimony chloride, are added to the annealing separator.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE4409691 | 1994-03-22 | ||
| DE4409691A DE4409691A1 (en) | 1994-03-22 | 1994-03-22 | Process for the production of electrical sheets with a glass coating |
| PCT/EP1995/001020 WO1995025820A1 (en) | 1994-03-22 | 1995-03-18 | Process for producing magnetic steel sheets with a glass coating |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0752012A1 EP0752012A1 (en) | 1997-01-08 |
| EP0752012B1 true EP0752012B1 (en) | 1998-08-26 |
Family
ID=6513410
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP95912252A Expired - Lifetime EP0752012B1 (en) | 1994-03-22 | 1995-03-18 | Process for producing magnetic steel sheets with a glass coating |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US5863356A (en) |
| EP (1) | EP0752012B1 (en) |
| JP (1) | JP3730254B2 (en) |
| KR (1) | KR100367985B1 (en) |
| AT (1) | ATE170226T1 (en) |
| CZ (1) | CZ292216B6 (en) |
| DE (2) | DE4409691A1 (en) |
| PL (1) | PL178890B1 (en) |
| RU (1) | RU2139945C1 (en) |
| WO (1) | WO1995025820A1 (en) |
Families Citing this family (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3475258B2 (en) * | 1994-05-23 | 2003-12-08 | 株式会社海水化学研究所 | Ceramic film forming agent and method for producing the same |
| DE19750066C1 (en) * | 1997-11-12 | 1999-08-05 | Ebg Elektromagnet Werkstoffe | Process for coating electrical steel strips with an annealing separator |
| US6835250B2 (en) * | 2000-05-01 | 2004-12-28 | Tateho Chemical Industries Co., Ltd. | Magnesium oxide particle aggregate |
| EP1342812B1 (en) * | 2000-10-25 | 2016-09-28 | Tateho Chemical Industries Co., Ltd. | Magnesium oxide particle aggregate |
| RU2298592C2 (en) * | 2002-03-28 | 2007-05-10 | Ниппон Стил Корпорейшн | Electrical-sheet steel with oriented grains possessing high adhesion of film and method of making such steel |
| DE102004014596A1 (en) * | 2004-03-23 | 2005-10-27 | Trithor Gmbh | Non-stick coating for the production of composite material wires |
| CN101541991B (en) * | 2006-11-22 | 2012-11-28 | 新日本制铁株式会社 | Grain-oriented electrical steel sheet excellent in film adhesion and manufacturing method thereof |
| JP5633178B2 (en) * | 2010-04-27 | 2014-12-03 | Jfeスチール株式会社 | Annealing separator for grain-oriented electrical steel sheet |
| DE102010038038A1 (en) * | 2010-10-07 | 2012-04-12 | Thyssenkrupp Electrical Steel Gmbh | Process for producing an insulation coating on a grain-oriented electro-steel flat product and electro-flat steel product coated with such an insulation coating |
| CN102453793B (en) * | 2010-10-25 | 2013-09-25 | 宝山钢铁股份有限公司 | Annealing isolation agent used for preparing mirror surface-oriented silicon steel with excellent magnetic property |
| PL2664689T4 (en) * | 2011-01-12 | 2019-09-30 | Nippon Steel & Sumitomo Metal Corp | Grain-oriented electrical steel sheet and manufacturing method thereof |
| JP5360272B2 (en) * | 2011-08-18 | 2013-12-04 | Jfeスチール株式会社 | Method for producing grain-oriented electrical steel sheet |
| IN2014MN00456A (en) * | 2011-10-04 | 2015-06-19 | Jfe Steel Corp | |
| DE102015114358B4 (en) | 2015-08-28 | 2017-04-13 | Thyssenkrupp Electrical Steel Gmbh | Method for producing a grain-oriented electrical strip and grain-oriented electrical strip |
| KR101909218B1 (en) | 2016-12-21 | 2018-10-17 | 주식회사 포스코 | Annealing separating agent composition for grain oriented electrical steel sheet, grain oriented electrical steel sheet, and method for manufacturing grain oriented electrical steel sheet |
| JP6939766B2 (en) | 2018-12-27 | 2021-09-22 | Jfeスチール株式会社 | Annealing separator for grain-oriented electrical steel sheets and manufacturing method of grain-oriented electrical steel sheets |
| JP6939767B2 (en) * | 2018-12-27 | 2021-09-22 | Jfeスチール株式会社 | Annealing separator for grain-oriented electrical steel sheets and manufacturing method of grain-oriented electrical steel sheets |
| CN111906142B (en) * | 2020-06-24 | 2022-08-16 | 浙江博星工贸有限公司 | Process for controlling mechanical property of cold-rolled stainless steel strip |
| CN114014529B (en) * | 2021-12-17 | 2023-02-21 | 中国建筑材料科学研究总院有限公司 | A kind of release agent for borosilicate glass bead fire throwing |
| CN114854960B (en) * | 2022-03-30 | 2023-09-05 | 武汉钢铁有限公司 | Annealing isolating agent for reducing surface defects of oriented silicon steel and use method thereof |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3151000A (en) * | 1959-08-28 | 1964-09-29 | Hooker Chemical Corp | Method of applying highly heat resistant protective coatings to metallic surfaces |
| US3151997A (en) * | 1961-09-29 | 1964-10-06 | United States Steel Corp | Separating-medium coating for preparation of electrical steel strip for annealing |
| US3615918A (en) * | 1969-03-28 | 1971-10-26 | Armco Steel Corp | Method of annealing with a magnesia separator containing a decomposable phosphate |
| SU569653A1 (en) * | 1976-01-04 | 1977-08-25 | Уральский научно-исследовательский институт черных металлов | Composition for thermoinsulating coatings |
| US4160681A (en) * | 1977-12-27 | 1979-07-10 | Allegheny Ludlum Industries, Inc. | Silicon steel and processing therefore |
| JPS55138021A (en) * | 1979-04-11 | 1980-10-28 | Nippon Steel Corp | Manufacture of annealing separation agent for electromagnetic steel plate |
| IT1127263B (en) * | 1978-11-28 | 1986-05-21 | Nippon Steel Corp | SEPARATION SUBSTANCE TO BE USED IN THE ANNEALING PHASE OF ORIENTED GRAINS OF SILICON STEEL |
| GB2130241B (en) * | 1982-09-24 | 1986-01-15 | Nippon Steel Corp | Method for producing a grain-oriented electrical steel sheet having a high magnetic flux density |
| US4582547A (en) * | 1984-05-07 | 1986-04-15 | Allegheny Ludlum Steel Corporation | Method for improving the annealing separator coating on silicon steel and coating therefor |
| JPS62156226A (en) * | 1985-12-27 | 1987-07-11 | Nippon Steel Corp | Production of grain oriented electrical steel sheet having uniform glass film and excellent magnetic characteristic |
| US4909864A (en) * | 1986-09-16 | 1990-03-20 | Kawasaki Steel Corp. | Method of producing extra-low iron loss grain oriented silicon steel sheets |
| US4740251A (en) * | 1986-12-22 | 1988-04-26 | Calgon Corporation | Method for improving magnesium oxide steel coatings |
| JPH0649949B2 (en) * | 1988-10-18 | 1994-06-29 | 新日本製鐵株式会社 | Method for producing grain-oriented electrical steel sheet having punching properties and metallic luster with excellent magnetic properties |
| EP0416420B1 (en) * | 1989-09-08 | 1994-12-14 | Armco Inc. | Magnesium oxide coating for electrical steels and the method of coating |
| JPH05247661A (en) * | 1992-03-04 | 1993-09-24 | Nippon Steel Corp | Method for producing grain-oriented electrical steel sheet having a uniform glass coating and excellent magnetic properties |
| EP0577124B1 (en) * | 1992-07-02 | 2002-10-16 | Nippon Steel Corporation | Grain oriented electrical steel sheet having high magnetic flux density and ultra low iron loss and process for producing the same |
-
1994
- 1994-03-22 DE DE4409691A patent/DE4409691A1/en not_active Withdrawn
-
1995
- 1995-03-18 US US08/704,579 patent/US5863356A/en not_active Expired - Lifetime
- 1995-03-18 WO PCT/EP1995/001020 patent/WO1995025820A1/en not_active Ceased
- 1995-03-18 JP JP52437895A patent/JP3730254B2/en not_active Expired - Lifetime
- 1995-03-18 AT AT95912252T patent/ATE170226T1/en not_active IP Right Cessation
- 1995-03-18 EP EP95912252A patent/EP0752012B1/en not_active Expired - Lifetime
- 1995-03-18 PL PL95316139A patent/PL178890B1/en unknown
- 1995-03-18 RU RU96119243A patent/RU2139945C1/en active
- 1995-03-18 DE DE59503345T patent/DE59503345D1/en not_active Expired - Lifetime
- 1995-03-18 CZ CZ19962738A patent/CZ292216B6/en not_active IP Right Cessation
- 1995-03-18 KR KR1019960705227A patent/KR100367985B1/en not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| EP0752012A1 (en) | 1997-01-08 |
| CZ273896A3 (en) | 1997-04-16 |
| CZ292216B6 (en) | 2003-08-13 |
| RU2139945C1 (en) | 1999-10-20 |
| WO1995025820A1 (en) | 1995-09-28 |
| DE59503345D1 (en) | 1998-10-01 |
| US5863356A (en) | 1999-01-26 |
| KR970701795A (en) | 1997-04-12 |
| PL178890B1 (en) | 2000-06-30 |
| DE4409691A1 (en) | 1995-09-28 |
| JP3730254B2 (en) | 2005-12-21 |
| ATE170226T1 (en) | 1998-09-15 |
| PL316139A1 (en) | 1996-12-23 |
| JPH09510503A (en) | 1997-10-21 |
| KR100367985B1 (en) | 2003-08-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0752012B1 (en) | Process for producing magnetic steel sheets with a glass coating | |
| DE69006946T2 (en) | Manufacture of grain-oriented silicon alloy thin sheets with an insulating layer produced on them. | |
| DE2637591C3 (en) | Method of forming a heat-resistant, insulating coating on an oriented silicon steel sheet | |
| EP0090241B1 (en) | Thermostable iron oxide pigments | |
| EP2252722B1 (en) | Method for producing a grain-oriented magnetic strip | |
| EP3921454B1 (en) | Aqueous composition for coating grain-oriented steel | |
| EP2586755B1 (en) | Hard magnetic La and Co doped hexagonal strontiumferrit | |
| DE2450850A1 (en) | METHOD AND MEANS OF FORMING AN INSULATING COATING ON AN ORIENTED SI STEEL SHEET | |
| DE2752803B2 (en) | Aqueous coating agent based on colloidal silicic acid anhydride | |
| DE2062290A1 (en) | Process for the production of an electrically insulating layer (Iso herschicht) on Sihziumstahlbleche | |
| EP4027358B1 (en) | Soft magnetic alloy and method for producing a soft magnetic alloy | |
| DE2947945A1 (en) | GLOW SEPARATOR FOR CORNORIENTED SILICON STEEL TAPES | |
| DE2917235A1 (en) | PROCESS FOR THE FORMATION OF FIXED AND UNIFORM INSULATION LAYERS ON GRAIN ORIENTED SILICON STEEL SHEET | |
| EP0199975A2 (en) | Red iron oxide pigments having improved colouring properties, process for their manufacture and their use | |
| DE68918836T2 (en) | Needle-shaped magnetic particles made of an iron alloy for magnetic recording and process for their production. | |
| DE3044771A1 (en) | MAGNETIC RECORDING MEDIUM AND PRODUCTION THEREOF | |
| DE69800332T2 (en) | Hematite particles and magnetic recording medium which uses the hematite particles as non-magnitic particles in a non-magnetic underlayer | |
| DE2810155A1 (en) | COATING SOLUTION FOR ELECTRIC STEEL SHEETS | |
| DE102018201618A1 (en) | Afterglow, but not nachglühpflichtiges electrical tape | |
| DE3218821A1 (en) | Stable slurry of inactive magnesium oxide, and process for the preparation thereof | |
| DE4293604C2 (en) | Soft magnetic steel material and process for its manufacture | |
| DE2856324A1 (en) | SILICON STEEL AND METHOD FOR PROCESSING IT | |
| DE3784473T2 (en) | RELEASE AGENT AND METHOD FOR GLOWING A SILICON STEEL. | |
| EP3720913A1 (en) | Chromium- and phosphate-free coating for electrically insulating an electric strip | |
| DE69513811T2 (en) | METHOD FOR PRODUCING A CORNORIENTED ELECTRIC STEEL SHEET WITH A MIRROR SURFACE AND WITH A LOW CORE LOSS |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19960821 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL PT SE |
|
| 17Q | First examination report despatched |
Effective date: 19970402 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL PT SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19980826 Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19980826 Ref country code: ES Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19980826 |
|
| REF | Corresponds to: |
Ref document number: 170226 Country of ref document: AT Date of ref document: 19980915 Kind code of ref document: T |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19980827 |
|
| REF | Corresponds to: |
Ref document number: 59503345 Country of ref document: DE Date of ref document: 19981001 |
|
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: GERMAN |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19981126 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19981126 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19981126 |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990318 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990318 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990331 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990331 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990504 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| BERE | Be: lapsed |
Owner name: EBG G.- FUR ELEKTROMAGNETISCHE WERKSTOFFE M.B.H. Effective date: 19990331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990930 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: CD |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20140319 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20140321 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20140318 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20140318 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R071 Ref document number: 59503345 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 Expiry date: 20150317 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20150317 |