CN1361663A - 杀真菌活性化合物组合物 - Google Patents
杀真菌活性化合物组合物 Download PDFInfo
- Publication number
- CN1361663A CN1361663A CN00810584A CN00810584A CN1361663A CN 1361663 A CN1361663 A CN 1361663A CN 00810584 A CN00810584 A CN 00810584A CN 00810584 A CN00810584 A CN 00810584A CN 1361663 A CN1361663 A CN 1361663A
- Authority
- CN
- China
- Prior art keywords
- active compound
- group
- following formula
- weight ratio
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
- A01N43/42—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings condensed with carbocyclic rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/18—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof
- A01N37/22—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof the nitrogen atom being directly attached to an aromatic ring system, e.g. anilides
- A01N37/24—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof the nitrogen atom being directly attached to an aromatic ring system, e.g. anilides containing at least one oxygen or sulfur atom being directly attached to the same aromatic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/88—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms six-membered rings with three ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/02—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom
- A01N47/04—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom containing >N—S—C≡(Hal)3 groups
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/12—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing a —O—CO—N< group, or a thio analogue thereof, neither directly attached to a ring nor the nitrogen atom being a member of a heterocyclic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/12—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing a —O—CO—N< group, or a thio analogue thereof, neither directly attached to a ring nor the nitrogen atom being a member of a heterocyclic ring
- A01N47/14—Di-thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N2300/00—Combinations or mixtures of active ingredients covered by classes A01N27/00 - A01N65/48 with other active or formulation relevant ingredients, e.g. specific carrier materials or surfactants, covered by classes A01N25/00 - A01N65/48
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
本发明涉及含有下述成分的新的活性化合物组合物,右式的5,7-二氯-4-(4-氟-苯氧基)-喹啉和说明书中所列的组(1)-(8)的活性物质。这些活性物质的新组合物具有非常好的杀真菌活性。
Description
本发明涉及新的活性化合物组合物,其中一方面含有已知的5,7-二氯-4-(4-氟-苯氧基)-喹啉,另一方面含有其它已知的杀真菌活性化合物,并且上述组合物特别适用于控制植物病原真菌。
已知5,7-二氯-4-(4-氟-苯氧基)-喹啉具有杀真菌活性(参见EP-A 0 326 330)。虽然该物质具有很好的杀菌活性;然而在低施用量时,某些情况下其效果则不能令人满意。
而且,已知5,7-二氯-4-(4-氟-苯氧基)-喹啉与其它已知杀真菌活性化合物的某些组合物可用于控制真菌(参见EP-A 0 326 330,WO96/18 299和WO98/51 146)。然而,上述混合物的活性也不总是能令人满意。
目前已发现新的活性化合物组合物具有很好的活性,这些组合物包括
和
(1)下式的三唑衍生物
其中
X代表氯或苯基,
或
或
或
或
(5)下式化合物
或
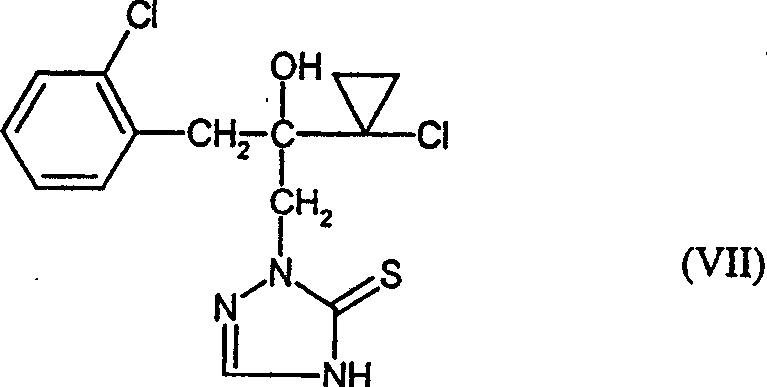
(6)下式的2-[2-(1-氯环丙基)-3-(2-氯苯基)-2-羟基丙基]-2,4-二氢-[1,2,4]-三唑-3-硫酮
或
或
令人惊奇地,本发明活性化合物组合物的杀真菌活性显著大于每一种活性化合物的活性总和。因此,获得意想不到的真正的增效作用,而不仅仅是加合作用。
式(I)的5,7-二氯-4-(4-氟-苯氧基)-喹啉是已知的(参见EP-A 0326 330)。
式(II)包括下述化合物
下式的1-(4-氯-苯氧基)-3,3-二甲基-1-(1,2,4-三唑-1-基)-丁-2-醇
和
为简便起见,每种情况下所示的仅是“硫羰”形式。
除了式(I)的5,7-二氯-4-(4-氟-苯氧基)-喹啉外,本发明活性化合物组合物中存在的其它杀真菌组分也是已知的。具体地,这些活性化合物公开在下述出版物中:
(1)式(II)化合物
DE-A 2 324 010
(2)式(III)化合物
Pesticide Manual,第11版,第1208页
(3)式(IV)化合物
Pesticide Manual,第11版,第608页
(4)式(V)化合物
EP-A 0 339 418
(5)式(VI)化合物
EP-A 0 472 996
(6)式(VII)化合物
WO96/16048
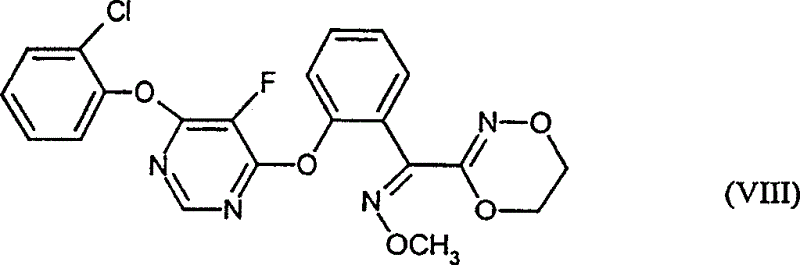
(7)式(VIII)化合物
EP-A 0 882 043
(8)式(IX)化合物
WO97/06171
除式(I)活性化合物以外,本发明活性化合物组合物包含选自组(1)-(8)化合物中的至少一种活性化合物。
如果在本发明活性化合物组合物中,活性化合物以一定的重量比例存在,则增效作用特别明显。但是,活性化合物组合物中活性化合物的重量比可在相对宽的范围内变化。通常,
每重量份的式(I)活性化合物,使用
0.1-10重量份,优选0.2-5重量份的组(1)活性化合物,
1-50重量份,优选2-10重量份的组(2)活性化合物,
1-50重量份,优选2-10重量份的组(3)活性化合物,
0.1-10重量份,优选0.2-5重量份的组(4)活性化合物,
0.1-10重量份,优选0.2-5重量份的组(5)活性化合物,
0.1-10重量份,优选0.2-5重量份的组(6)活性化合物,
0.05-10重量份,优选0.1-5重量份的组(7)活性化合物和/或
0.05-10重量份,优选0.1-5重量份的组(8)活性化合物。
本发明的活性化合物组合物具有非常好的杀真菌活性,并可用来控制植物病原真菌,如根肿菌纲、卵菌纲、壶菌纲、接合菌纲、子囊菌纲、担子菌纲、半知菌纲等。
本发明的活性化合物组合物特别适于防治,如白粉菌属,柄锈菌属和壳针孢属真菌引起的禾谷类作物病害,以及用于防治葡萄栽培中发生的由如钩丝壳霉属,单轴霉属和葡萄孢属真菌引起的病害,还可用于防治双子叶作物的白粉病和霜霉病以及叶斑病的致病微生物。
植物对控制植物病害所需浓度的活性化合物组合物的良好的耐受性,使得用其处理植物的地上部分、植物的繁殖根茎和种子,以及植物生长的土壤成为可能。本发明活性化合物组合物可作为叶面施用,还可用于拌种。
本发明活性化合物组合物可被转化成为常规的制剂,如液剂,乳剂,悬浮剂,粉剂,泡沫剂,糊剂,颗粒剂,气雾剂,在聚合物中的微细胶囊和种子包衣组合物,以及超低容量弥雾剂(UIV-Formulieren)。
这些制剂是以已知方法生产的,例如,通过将活性化合物或活性化合物组合物与填充剂,即液体溶剂,加压液化气体,和/或固体载体混合而生产,制剂中可任意选择的使用表面活性剂,即乳化剂和/或分散剂,和/或成泡剂。在使用水作为填充剂的情况下,例如,也可使用有机溶剂作为助溶剂。适当的液体溶剂主要有:芳香烃类,如二甲苯,甲苯或烷基萘,氯代芳烃类和氯代脂肪烃类,如氯苯,二氯乙烷或二氯甲烷,脂肪烃类,如环己烷或链烷烃,例如石油馏份,醇类,如丁醇或乙二醇及其醚和酯类,酮类,如丙酮,甲基乙基酮,甲基异丁基酮或环己酮,强极性溶剂如二甲基甲酰胺和二甲基亚砜以及水。液化气体填充剂或载体中液体意为在室温和大气压下是气体的液体,例如气雾推进剂,如丁烷,丙烷,氮和二氧化碳。适当的固体载体有:例如,天然矿物粉末如高岭土,粘土,滑石,白垩,石英,硅镁土,蒙脱土或硅藻土,和合成矿物粉末如高分散二氧化硅,氧化铝和硅酸盐。适合颗粒剂的固体载体有:例如粉碎和分级的天然岩石如方解石,大理石,浮石,海泡石和白云石,以及无机和有机粉末的合成颗粒,和有机材料的颗粒如锯末,坚果壳,玉米穗茎和烟草茎。适当的乳化剂和/或成泡剂有:例如非离子和阴离子乳化剂,如聚氧乙烯脂肪酸酯,聚氧乙烯脂肪醇醚,例如烷基芳基聚乙二醇醚,烷基磺酸盐,烷基硫酸盐,芳基磺酸盐以及蛋白水解产物。适合的分散剂有:例如木质素亚硫酸盐废液和甲基纤维素。
在制剂中还可使用粘着剂如羧甲基纤维素和粉末、颗粒或胶乳状天然和合成聚合物,如阿拉伯树胶,聚乙烯醇和聚乙酸乙烯酯,以及天然磷脂,如脑磷脂和卵磷脂,和合成磷脂。其它的添加剂可以是矿物油和植物油。
可以使用的着色剂如无机颜料,例如氧化铁,氧化钛和普鲁士兰,和有机染料,如茜素染料,偶氮染料和金属酞菁染料,和痕量营养物,例如铁、锰、硼、铜、钴、钼和锌的盐。
制剂中通常含有按重量计0.1%-95%,优选按重量计0.5%-90%的活性化合物。
制剂中,本发明的活性化合物组合物可以与其它已知活性化合物的混合物的形式存在,其它已知活性化合物如杀真菌剂,杀虫剂,杀螨剂和除草剂,以及与肥料和植物生长调节剂的混合物。
本发明活性化合物组合物可以其本身或其制剂形式使用,或以其制备形式使用,如现混现用溶液,乳油,乳液,悬浮液,可湿性粉剂,可溶粉和颗粒剂。以常规方法使用本发明组合物,例如泼浇,喷雾,弥雾,撒播,涂抹,干式包衣,微湿包衣,湿式包衣,浆液包衣或包覆。
使用本发明活性化合物组合物时,根据施用方式施用量可在较宽的范围内变化。在处理植物体时,活性化合物组合物的施用量通常为0.1至10,000g/ha,优选10至1000g/ha。在处理种子时,活性化合物组合物的施用量通常为每公斤种子需要0.001至50g,优选0.01至10g的活性化合物组合物。在处理土壤时,活性化合物组合物的施用量通常为0.1至10,000g/ha,优选1至5000g/ha。
从下述实施例中可以看到本发明活性化合物组合物的良好的杀真菌活性。在单独的活性化合物显示出弱的杀真菌活性的同时,组合物的活性超过了化合物活性的简单加合。
如果,活性化合物组合物的杀真菌活性大于单独使用时的活性化合物的活性总和,则认为杀菌剂总具有增效作用。
两种活性化合物的特定组合物的预期活性可通过S.R.Colby公式如下计算(“Calculating Synergistic and Antagonistic Responsesof Herbicide Combinations”,Weeds 15,(1967),20-22)
如果
X 表示活性化合物A在mg/ha施用量时的活性
Y 表示活性化合物B在ng/ha施用量时的活性
E 表示活性化合物A和B在m和ng/ha施用量时的活性
则
这里,活性用%表示。0%表示相当于对照的活性,而100%活性表示未观察到侵染。
如果实际观测的杀真菌活性超出计算值,则组合物的活性是超加合的,即存在增效作用。在这种情况下,实际观测的活性必须大于由上述公式计算的预期活性(E)的数值。
用下述实施例解释本发明。
实施例
实施例1
杀单丝壳菌属真菌(Sphaerotheca)试验(黄瓜)/保护作用
溶剂: 47重量份丙酮
乳化剂: 3重量份烷基-芳基聚乙二醇醚
为了制备活性化合物的适合的制剂,将1重量份的活性化合物或活性化合物组合物与预定量的溶剂和乳化剂混合,并将浓缩物用水稀释到所需浓度,或将活性化合物或活性化合物组合物的商品制剂用水稀释到所需浓度。
为了试验保护活性,用预定施用量的活性化合物制剂喷雾幼苗。喷雾层干燥后,用苍耳单丝壳菌(Sphaerotheca fuliginea)的孢子水悬液接种植物。然后将植物放置在约23℃和相对大气湿度约70%的温室中。
接种10天后进行评价。在评价中,0%表示活性水平与对照相同,100%活性表示未观察到侵染。
实施例2白粉菌的杀菌试验(大麦)/治疗作用溶剂: 10重量份N-甲基吡咯烷酮乳化剂: 0.6重量份烷基芳基聚乙二醇醚
为了制备活性化合物的适合的制剂,将1重量份的活性化合物或活性化合物组合物与预定量的溶剂和乳化剂混合,并将浓缩物用水稀释到所需浓度,或将活性化合物或活性化合物组合物的商品制剂用水稀释到所需浓度。
为了试验治疗活性,用禾白粉菌(Erysiphe graminisf.sp.hordei)的孢子向幼苗喷粉。接种48小时后,用活性化合物制剂以预定施用量喷雾植物。
将植物放置在约20℃和相对大气湿度约80%的温室中,以促进白粉菌孢子的发育。
接种7天后进行评价。在评价中,0%表示活性水平与对照相同,100%活性表示未观察到侵染。
实施例3
疫霉属真菌试验(番茄)/保护作用
溶剂: 47重量份丙酮
乳化剂: 3重量份烷基-芳基聚乙二醇醚
为了制备活性化合物的适合的制剂,将1重量份的活性化合物或活性化合物组合物与预定量的溶剂和乳化剂混合,并将浓缩物用水稀释到所需浓度,或将活性化合物或活性化合物组合物的商品制剂用水稀释到所需浓度。
为了试验保护活性,用预定施用量的活性化合物制剂喷雾幼苗。喷雾层干燥后,用蔓延疫霉(Phytophthora infestans)的孢子水悬液接种植物。然后将植物放置在约20℃和相对大气湿度100%的培育室中。
接种3天后进行评价。在评价中,0%表示活性水平与对照相同,100%活性表示未观察到侵染。
Claims (5)
2.根据权利要求1的组合物,其特征在于,在活性化合物组合物中,式(I)活性化合物与下述化合物的重量比为
-与组(1)活性化合物的重量比为1∶0.1-1∶10,
-与组(2)活性化合物的重量比为1∶1-1∶50,
-与组(3)活性化合物的重量比为1∶1-1∶50,
-与组(4)活性化合物的重量比为1∶0.1-1∶10,
-与组(5)活性化合物的重量比为1∶0.1-1∶10,
-与组(6)活性化合物的重量比为1∶0.1-1∶10,
-与组(7)活性化合物的重量比为1∶0.5-1∶10,以及
-与组(8)活性化合物的重量比为1∶0.5-1∶10。
3.防治真菌的方法,其特征在于将权利要求1的活性化合物组合物作用于真菌和/或其生长环境。
4.权利要求1活性化合物组合物防治真菌的用途。
5.制备杀真菌组合物的方法,其特征在于将根据权利要求1的活性化合物组合物与填充剂和/或表面活性物质混合。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19933938A DE19933938A1 (de) | 1999-07-20 | 1999-07-20 | Fungizide Wirkstoffkombinationen |
| DE19933938.4 | 1999-07-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN1361663A true CN1361663A (zh) | 2002-07-31 |
Family
ID=7915383
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN00810584A Pending CN1361663A (zh) | 1999-07-20 | 2000-07-07 | 杀真菌活性化合物组合物 |
Country Status (16)
| Country | Link |
|---|---|
| US (1) | US6620822B1 (zh) |
| EP (2) | EP1202627B1 (zh) |
| JP (1) | JP2003504382A (zh) |
| KR (1) | KR20020020752A (zh) |
| CN (1) | CN1361663A (zh) |
| AT (1) | ATE242967T1 (zh) |
| AU (1) | AU5983500A (zh) |
| BR (1) | BR0012573A (zh) |
| DE (2) | DE19933938A1 (zh) |
| ES (1) | ES2195918T3 (zh) |
| HU (1) | HUP0201986A3 (zh) |
| MX (1) | MXPA02000675A (zh) |
| PL (1) | PL353484A1 (zh) |
| PT (1) | PT1202627E (zh) |
| WO (1) | WO2001005228A2 (zh) |
| ZA (1) | ZA200110383B (zh) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103444768A (zh) * | 2012-06-02 | 2013-12-18 | 陕西韦尔奇作物保护有限公司 | 一种含苯氧喹啉与三唑类的高效杀菌组合物 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19716257A1 (de) | 1997-04-18 | 1998-10-22 | Bayer Ag | Fungizide Wirkstoffkombination |
| FR2832031A1 (fr) * | 2001-11-14 | 2003-05-16 | Aventis Cropscience Sa | Composition fongicide a base d'au moins un derive de pyridylmethylbenzamide et d'au moins un derive de type valinamide |
| PT2080433E (pt) * | 2002-03-21 | 2011-10-03 | Basf Se | Misturas fungicidas |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2324010C3 (de) | 1973-05-12 | 1981-10-08 | Bayer Ag, 5090 Leverkusen | 1-Substituierte 2-Triazolyl-2-phenoxyäthanol-Verbindungen, Verfahren zu ihrer Herstellung und ihre Verwendung zur Bekämpfung von Pilzen |
| GB2110934A (en) * | 1981-11-17 | 1983-06-29 | Ici Plc | Fungicidal compositions |
| DE3234624A1 (de) * | 1982-09-18 | 1984-03-22 | Bayer Ag, 5090 Leverkusen | Fungizide mittel |
| DE3234625A1 (de) * | 1982-09-18 | 1984-03-22 | Bayer Ag, 5090 Leverkusen | Fungizide mittel |
| DE3700923A1 (de) * | 1987-01-15 | 1988-07-28 | Bayer Ag | Fungizide wirkstoffkombination |
| IL89029A (en) * | 1988-01-29 | 1993-01-31 | Lilly Co Eli | Fungicidal quinoline and cinnoline derivatives, compositions containing them, and fungicidal methods of using them |
| DE3814505A1 (de) | 1988-04-29 | 1989-11-09 | Bayer Ag | Substituierte cycloalkyl- bzw. heterocyclyl-carbonsaeureanilide |
| US5453531A (en) | 1990-08-25 | 1995-09-26 | Bayer Aktiengesellschaft | Substituted valinamide derivatives |
| DE4139637A1 (de) * | 1991-12-02 | 1993-06-03 | Bayer Ag | Fungizide wirkstoffkombinationen |
| DE19528046A1 (de) | 1994-11-21 | 1996-05-23 | Bayer Ag | Triazolyl-Derivate |
| DE4444911A1 (de) | 1994-12-16 | 1996-06-27 | Basf Ag | Fungizide Mischung |
| GB9709907D0 (en) | 1997-05-15 | 1997-07-09 | Ciba Geigy Ag | Novel combinations |
| GB9711127D0 (en) * | 1997-05-29 | 1997-07-23 | Leo Pharm Prod Ltd | Novel cyanoguanidines |
| DE19939841A1 (de) * | 1998-11-20 | 2000-05-25 | Bayer Ag | Fungizide Wirkstoffkombinationen |
-
1999
- 1999-07-20 DE DE19933938A patent/DE19933938A1/de not_active Withdrawn
-
2000
- 2000-07-07 US US10/031,175 patent/US6620822B1/en not_active Expired - Fee Related
- 2000-07-07 AU AU59835/00A patent/AU5983500A/en not_active Abandoned
- 2000-07-07 ES ES00945897T patent/ES2195918T3/es not_active Expired - Lifetime
- 2000-07-07 KR KR1020017016618A patent/KR20020020752A/ko not_active Ceased
- 2000-07-07 JP JP2001510306A patent/JP2003504382A/ja active Pending
- 2000-07-07 PL PL00353484A patent/PL353484A1/xx unknown
- 2000-07-07 PT PT00945897T patent/PT1202627E/pt unknown
- 2000-07-07 EP EP00945897A patent/EP1202627B1/de not_active Expired - Lifetime
- 2000-07-07 WO PCT/EP2000/006470 patent/WO2001005228A2/de not_active Ceased
- 2000-07-07 EP EP02014967A patent/EP1247452A3/de not_active Withdrawn
- 2000-07-07 DE DE50002602T patent/DE50002602D1/de not_active Expired - Fee Related
- 2000-07-07 BR BR0012573-3A patent/BR0012573A/pt not_active Application Discontinuation
- 2000-07-07 MX MXPA02000675A patent/MXPA02000675A/es active IP Right Grant
- 2000-07-07 HU HU0201986A patent/HUP0201986A3/hu unknown
- 2000-07-07 CN CN00810584A patent/CN1361663A/zh active Pending
- 2000-07-07 AT AT00945897T patent/ATE242967T1/de not_active IP Right Cessation
-
2001
- 2001-12-19 ZA ZA200110383A patent/ZA200110383B/xx unknown
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103444768A (zh) * | 2012-06-02 | 2013-12-18 | 陕西韦尔奇作物保护有限公司 | 一种含苯氧喹啉与三唑类的高效杀菌组合物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1202627B1 (de) | 2003-06-18 |
| WO2001005228A2 (de) | 2001-01-25 |
| ATE242967T1 (de) | 2003-07-15 |
| ES2195918T3 (es) | 2003-12-16 |
| EP1202627A2 (de) | 2002-05-08 |
| BR0012573A (pt) | 2002-04-16 |
| PL353484A1 (en) | 2003-11-17 |
| AU5983500A (en) | 2001-02-05 |
| DE50002602D1 (de) | 2003-07-24 |
| PT1202627E (pt) | 2003-11-28 |
| JP2003504382A (ja) | 2003-02-04 |
| WO2001005228A3 (de) | 2001-08-02 |
| EP1247452A3 (de) | 2003-03-19 |
| EP1247452A2 (de) | 2002-10-09 |
| MXPA02000675A (es) | 2002-08-30 |
| KR20020020752A (ko) | 2002-03-15 |
| HUP0201986A2 (en) | 2002-09-28 |
| ZA200110383B (en) | 2002-12-19 |
| DE19933938A1 (de) | 2001-01-25 |
| HUP0201986A3 (en) | 2005-01-28 |
| US6620822B1 (en) | 2003-09-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP3682988B2 (ja) | 殺菌・殺カビ組成物 | |
| JP4094067B2 (ja) | 殺菌・殺カビ剤活性物質の組み合わせ | |
| JP4680189B2 (ja) | 殺真菌性三元活性成分組み合わせ | |
| RU2331192C2 (ru) | Фунгицидные комбинации активных веществ | |
| JP4177899B2 (ja) | 殺菌・殺カビ活性物質組合せ物 | |
| JP2530590B2 (ja) | 殺菌・殺カビ剤 | |
| US20060014738A1 (en) | Fungicidal combination of active substances | |
| CN85108922A (zh) | 杀真菌的吡咯化合物的制备方法 | |
| CN1245015A (zh) | 杀菌剂组合物及其使用方法 | |
| JP2006528149A (ja) | 殺菌剤の組み合わせ | |
| JPS6150921B2 (zh) | ||
| CN1140976A (zh) | 含吡咯基团杀菌剂与含吡唑、吡咯或苯基咪唑基团杀虫剂的复配物 | |
| KR20020093063A (ko) | 살진균성 활성제 배합물 | |
| CN1220433C (zh) | 增效除草混合物 | |
| CN1066610C (zh) | 杀微生物剂 | |
| RU2366177C2 (ru) | Фунгицидные комбинации биологически активных веществ, содержащие спироксамин, протиоконазол и тебуконазол | |
| JPH05194111A (ja) | 殺菌剤 | |
| HU203270B (en) | Herbicide compositions containing substituted 2-phenyl-imino-oxazolidines as active components and process for producing the active components | |
| CN100387128C (zh) | N-(4,6-二甲基嘧啶-2-基)苯胺杀菌组合物 | |
| CN1106148C (zh) | 杀真菌混合物 | |
| CN1361663A (zh) | 杀真菌活性化合物组合物 | |
| JP2858915B2 (ja) | 殺菌剤 | |
| CN1216442A (zh) | 杀真菌混合物 | |
| CN1212767C (zh) | 杀真菌组合物 | |
| CN1190868A (zh) | 杀真菌活性化合物的组合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C06 | Publication | ||
| PB01 | Publication | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |