CN110251663B - 一种具有抗衰老作用的外泌体-超氧化物歧化酶纳米制剂及其制备方法 - Google Patents
一种具有抗衰老作用的外泌体-超氧化物歧化酶纳米制剂及其制备方法 Download PDFInfo
- Publication number
- CN110251663B CN110251663B CN201910593342.1A CN201910593342A CN110251663B CN 110251663 B CN110251663 B CN 110251663B CN 201910593342 A CN201910593342 A CN 201910593342A CN 110251663 B CN110251663 B CN 110251663B
- Authority
- CN
- China
- Prior art keywords
- exosome
- sod
- superoxide dismutase
- preparation
- nano
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 41
- 230000003712 anti-aging effect Effects 0.000 title abstract description 9
- 210000001808 exosome Anatomy 0.000 claims abstract description 57
- 239000001397 quillaja saponaria molina bark Substances 0.000 claims abstract description 25
- 229930182490 saponin Natural products 0.000 claims abstract description 25
- 150000007949 saponins Chemical class 0.000 claims abstract description 25
- 238000011068 loading method Methods 0.000 claims abstract description 24
- 239000012528 membrane Substances 0.000 claims abstract description 18
- 238000001125 extrusion Methods 0.000 claims abstract description 12
- 102000019197 Superoxide Dismutase Human genes 0.000 claims description 106
- 108010012715 Superoxide dismutase Proteins 0.000 claims description 71
- 210000004027 cell Anatomy 0.000 claims description 21
- 239000006228 supernatant Substances 0.000 claims description 21
- 238000000034 method Methods 0.000 claims description 14
- 102000004169 proteins and genes Human genes 0.000 claims description 11
- 108090000623 proteins and genes Proteins 0.000 claims description 11
- 238000000108 ultra-filtration Methods 0.000 claims description 8
- 239000000872 buffer Substances 0.000 claims description 7
- 239000002245 particle Substances 0.000 claims description 7
- 238000005119 centrifugation Methods 0.000 claims description 3
- 210000001985 kidney epithelial cell Anatomy 0.000 claims description 3
- 239000002502 liposome Substances 0.000 claims description 3
- 229920000515 polycarbonate Polymers 0.000 claims description 3
- 239000004417 polycarbonate Substances 0.000 claims description 3
- 239000000047 product Substances 0.000 claims description 3
- 239000012141 concentrate Substances 0.000 claims 3
- 150000002148 esters Chemical class 0.000 claims 1
- 239000013618 particulate matter Substances 0.000 claims 1
- 239000003814 drug Substances 0.000 abstract description 24
- 229940079593 drug Drugs 0.000 abstract description 21
- 230000000694 effects Effects 0.000 abstract description 19
- 102000004190 Enzymes Human genes 0.000 abstract description 11
- 108090000790 Enzymes Proteins 0.000 abstract description 11
- 230000005847 immunogenicity Effects 0.000 abstract description 5
- 239000000203 mixture Substances 0.000 abstract description 5
- 241000244203 Caenorhabditis elegans Species 0.000 abstract description 4
- 230000008901 benefit Effects 0.000 abstract description 4
- 230000003197 catalytic effect Effects 0.000 abstract description 4
- 238000009472 formulation Methods 0.000 abstract description 4
- 230000035699 permeability Effects 0.000 abstract description 3
- 230000007246 mechanism Effects 0.000 abstract description 2
- 241000244206 Nematoda Species 0.000 description 47
- 239000000243 solution Substances 0.000 description 18
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 14
- 239000007853 buffer solution Substances 0.000 description 13
- 150000003254 radicals Chemical class 0.000 description 10
- 230000032683 aging Effects 0.000 description 8
- 238000006701 autoxidation reaction Methods 0.000 description 7
- 229940079877 pyrogallol Drugs 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 238000006243 chemical reaction Methods 0.000 description 6
- VSIVTUIKYVGDCX-UHFFFAOYSA-M sodium;4-[2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)tetrazol-2-ium-5-yl]benzene-1,3-disulfonate Chemical compound [Na+].COC1=CC([N+]([O-])=O)=CC=C1[N+]1=NC(C=2C(=CC(=CC=2)S([O-])(=O)=O)S([O-])(=O)=O)=NN1C1=CC=C([N+]([O-])=O)C=C1 VSIVTUIKYVGDCX-UHFFFAOYSA-M 0.000 description 6
- 230000004083 survival effect Effects 0.000 description 6
- 239000002033 PVDF binder Substances 0.000 description 5
- 239000010949 copper Substances 0.000 description 5
- 238000005538 encapsulation Methods 0.000 description 5
- 230000006870 function Effects 0.000 description 5
- 239000000499 gel Substances 0.000 description 5
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- KQPYUDDGWXQXHS-UHFFFAOYSA-N juglone Chemical compound O=C1C=CC(=O)C2=C1C=CC=C2O KQPYUDDGWXQXHS-UHFFFAOYSA-N 0.000 description 4
- 230000004792 oxidative damage Effects 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 239000001301 oxygen Substances 0.000 description 4
- 229920001213 Polysorbate 20 Polymers 0.000 description 3
- OUUQCZGPVNCOIJ-UHFFFAOYSA-M Superoxide Chemical compound [O-][O] OUUQCZGPVNCOIJ-UHFFFAOYSA-M 0.000 description 3
- 238000002835 absorbance Methods 0.000 description 3
- 238000012258 culturing Methods 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 235000013601 eggs Nutrition 0.000 description 3
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 3
- 230000036541 health Effects 0.000 description 3
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 3
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 3
- 230000009758 senescence Effects 0.000 description 3
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 2
- 108010093894 Xanthine oxidase Proteins 0.000 description 2
- 102100033220 Xanthine oxidase Human genes 0.000 description 2
- 239000000427 antigen Substances 0.000 description 2
- 102000036639 antigens Human genes 0.000 description 2
- 108091007433 antigens Proteins 0.000 description 2
- VYTBDSUNRJYVHL-UHFFFAOYSA-N beta-Hydrojuglone Natural products O=C1CCC(=O)C2=C1C=CC=C2O VYTBDSUNRJYVHL-UHFFFAOYSA-N 0.000 description 2
- 230000027455 binding Effects 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 238000012377 drug delivery Methods 0.000 description 2
- 239000012091 fetal bovine serum Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- 238000009396 hybridization Methods 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000003550 marker Substances 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 210000002487 multivesicular body Anatomy 0.000 description 2
- VMGAPWLDMVPYIA-HIDZBRGKSA-N n'-amino-n-iminomethanimidamide Chemical compound N\N=C\N=N VMGAPWLDMVPYIA-HIDZBRGKSA-N 0.000 description 2
- 230000009871 nonspecific binding Effects 0.000 description 2
- 230000036542 oxidative stress Effects 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- BOLDJAUMGUJJKM-LSDHHAIUSA-N renifolin D Natural products CC(=C)[C@@H]1Cc2c(O)c(O)ccc2[C@H]1CC(=O)c3ccc(O)cc3O BOLDJAUMGUJJKM-LSDHHAIUSA-N 0.000 description 2
- 230000002000 scavenging effect Effects 0.000 description 2
- -1 superoxide anion free radical Chemical class 0.000 description 2
- 230000001360 synchronised effect Effects 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 101000836247 Aquifex pyrophilus Superoxide dismutase [Fe] Proteins 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- 102100025222 CD63 antigen Human genes 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 108020004414 DNA Proteins 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 101000934368 Homo sapiens CD63 antigen Proteins 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- HLFSDGLLUJUHTE-SNVBAGLBSA-N Levamisole Chemical compound C1([C@H]2CN3CCSC3=N2)=CC=CC=C1 HLFSDGLLUJUHTE-SNVBAGLBSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 102000008221 Superoxide Dismutase-1 Human genes 0.000 description 1
- 108010021188 Superoxide Dismutase-1 Proteins 0.000 description 1
- 238000003917 TEM image Methods 0.000 description 1
- 108700030796 Tsg101 Proteins 0.000 description 1
- 101150072717 Tsg101 gene Proteins 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 230000002238 attenuated effect Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 230000034303 cell budding Effects 0.000 description 1
- 230000007910 cell fusion Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 238000004737 colorimetric analysis Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 238000007323 disproportionation reaction Methods 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 210000001163 endosome Anatomy 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 230000003203 everyday effect Effects 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 238000010812 external standard method Methods 0.000 description 1
- 210000002744 extracellular matrix Anatomy 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- ODKNJVUHOIMIIZ-RRKCRQDMSA-N floxuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ODKNJVUHOIMIIZ-RRKCRQDMSA-N 0.000 description 1
- 238000002073 fluorescence micrograph Methods 0.000 description 1
- 238000001506 fluorescence spectroscopy Methods 0.000 description 1
- 230000009931 harmful effect Effects 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 229960001614 levamisole Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 230000003859 lipid peroxidation Effects 0.000 description 1
- 239000012160 loading buffer Substances 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000017448 oviposition Effects 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 238000005502 peroxidation Methods 0.000 description 1
- 210000001539 phagocyte Anatomy 0.000 description 1
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 210000003370 receptor cell Anatomy 0.000 description 1
- 238000006479 redox reaction Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 239000012488 sample solution Substances 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 235000020183 skimmed milk Nutrition 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229940126585 therapeutic drug Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/43—Enzymes; Proenzymes; Derivatives thereof
- A61K38/44—Oxidoreductases (1)
- A61K38/446—Superoxide dismutase (1.15)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5063—Compounds of unknown constitution, e.g. material from plants or animals
- A61K9/5068—Cell membranes or bacterial membranes enclosing drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
- A61P39/06—Free radical scavengers or antioxidants
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0603—Embryonic cells ; Embryoid bodies
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0684—Cells of the urinary tract or kidneys
- C12N5/0686—Kidney cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y115/00—Oxidoreductases acting on superoxide as acceptor (1.15)
- C12Y115/01—Oxidoreductases acting on superoxide as acceptor (1.15) with NAD or NADP as acceptor (1.15.1)
- C12Y115/01001—Superoxide dismutase (1.15.1.1)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2509/00—Methods for the dissociation of cells, e.g. specific use of enzymes
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Zoology (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Biochemistry (AREA)
- Biotechnology (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- General Engineering & Computer Science (AREA)
- Public Health (AREA)
- Cell Biology (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Urology & Nephrology (AREA)
- Microbiology (AREA)
- Developmental Biology & Embryology (AREA)
- General Chemical & Material Sciences (AREA)
- Reproductive Health (AREA)
- Gynecology & Obstetrics (AREA)
- Gastroenterology & Hepatology (AREA)
- Toxicology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Immunology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Virology (AREA)
- Botany (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Enzymes And Modification Thereof (AREA)
Abstract
一种具有抗衰老作用的外泌体‑超氧化物歧化酶(SOD)纳米制剂及其制备方法,属于生物技术领域。本发明通过挤出法结合皂苷辅助的策略将SOD酶分子载入到外泌体中,并以制备的SOD@EXO纳米制剂为纳米药物,以N2野生型秀丽隐杆线虫为模式生物,系统评价了该纳米制剂在抗衰老方面的作用效果与机制。本发明将SOD装载进入外泌体,不仅保留了天然酶的催化活性,同时较天然SOD具有更加显著的优势,如延长半衰期、降低免疫原性和增加膜通透性等。该纳米制剂具有较高的载药量,同时能够实现稳定的药物释放,最终构建低免疫原性及高催化活力的外泌体‑超氧化物歧化酶纳米制剂,极大提升SOD分子的半衰期和抗衰老治疗效果。
Description
技术领域
本发明属于生物技术领域,具体涉及一种具有抗衰老作用的外泌体-超氧化物歧化酶(SOD)纳米制剂及其制备方法。
背景技术
衰老(senescence)是生物体细胞、器官以及组织的功能随时间而逐渐丧失的渐进过程。现代衰老学说认为,机体的衰老过程与自由基及所诱导的脂质过氧化对细胞和机体的损伤有关。自由基及其衍生物是指具有高度活性的不配对电子的原子团,具有强氧化性,是人体氧化还原反应的重要反应成分。适量的自由基能够促进细胞增殖,刺激吞噬细胞杀灭细菌。但过量的自由基对脂质、DNA等物质产生伤害作用,造成生物膜的损伤,使机体处于不正常的状态,从而加速衰老。
超氧化物歧化酶(Superoxide Dismutase,EC1.15.1.1)简称SOD,是一种酸性金属酶,广泛存在于动物、植物、微生物体内。根据酶分子中所含金属辅基的不同,超氧化物歧化酶主要可分为Cu/Zn-SOD,Mn-SOD,Fe-SOD等类型,其中Cu/Zn-SOD研究的最多。SOD能催化超氧阴离子自由基O2·-发生歧化反应,在生物体防御氧化损伤的过程中,SOD起到清除和减少人体内过多的自由基、减轻细胞组织过氧化损伤、提高机体免疫力、延缓衰老的作用。此外,人体内的SOD可有效地通过清除超氧阴离子自由基最终能达到抑制癌细胞的效果,Mn-SOD表达效果更为突出。SOD也具有极强的抗炎症作用,有歧化活性氧、消除炎症、保护关节的作用,在类风湿性关节炎患者中,血清中SOD水平显著低于健康人。但是SOD的免疫原性、不稳定性、以及在体内的半衰期较短等缺点,限制了其在临床和保健方面的应用。
外泌体(Exosome,EXO)是由多种活细胞分泌的30~150nm的纳米级囊泡,来源于晚期核内体,是由细胞内多泡体(MVBs)出芽形成,再与细胞膜融合后释放到细胞外基质中,几乎所有类型的细胞都能分泌,广泛存在于各种体液中,其功能主要为在细胞间转运相关物质从而实现供受体细胞之间的物质和信息交流。外泌体作为药物的天然内源性载体具有独特优势,如良好的生物相容性、细胞运输药物的效率高和更强的靶部位识别能力等,因此外泌体有望可以成为良好的蛋白质药物递送载体,为相关疾病的治疗提供一个良好的纳米药物平台,具有广阔的应用前景。
发明内容
本发明通过挤出法结合皂苷辅助的策略将SOD酶分子载入到外泌体中,构建了一种外泌体-超氧化物歧化酶纳米制剂(SOD@EXO纳米制剂),并以制备的SOD@EXO纳米制剂为纳米药物,以N2野生型秀丽隐杆线虫为模式生物,系统评价了该纳米制剂在抗衰老方面的作用效果与机制。本发明将SOD装载进入外泌体,不仅保留了天然酶的催化活性,同时较天然SOD具有更加显著的优势,如延长半衰期、降低免疫原性和增加膜通透性等。该纳米制剂具有较高的载药量,同时能够实现稳定的药物释放,能显著抵抗线虫的氧化应激状态,降低线虫体内活性氧水平,提高线虫健康状况,延长线虫寿命。最终构建了低免疫原性及高催化活力的外泌体-超氧化物歧化酶纳米制剂,极大提升SOD分子的半衰期和抗衰老治疗效果。因此,该纳米制剂在清除自由基、延缓衰老方面具有良好的应用前景,成为一类具有良好抗衰老效果的纳米制剂。
本发明中所涉及的SOD来源于牛血,属于Cu/Zn-SOD,能够清除机体代谢过程中产生的过量超氧阴离子自由基,延缓由于自由基侵害而出现的衰老现象。
本发明所述的一种外泌体-超氧化物歧化酶纳米制剂的制备方法,其步骤如下:
(1)获得外泌体供体细胞的上清液400~600mL,然后在200~400g条件下离心10~20min去除悬浮细胞,再在2000~4000g条件下离心10~20min去除细胞碎片,最后在8000~12000g条件下离心20~40min去除较大的颗粒状物质;
(2)将步骤(1)获得的上清液经0.22μm的滤膜过滤,去除大于220nm的颗粒,再将上清液经过100kDa截留分子量的超滤离心管进行浓缩,收集超滤离心管上层浓缩液;
(3)将步骤(2)得到的浓缩液在90000~110000g条件下离心1~2h,去除上清液,保留沉淀外泌体;然后用PBS缓冲液重悬此沉淀外泌体,在相同的条件下再次离心,去除上清液,保留沉淀外泌体;最后再以1~2mL的PBS缓冲液重悬此沉淀外泌体,其中外泌体的浓度为1~3mg/mL;
(4)将超氧化物歧化酶(SOD)和皂素加入到步骤(3)得到的溶有外泌体的PBS缓冲液中,然后装入基于注射器的脂质体挤出器(Avanti Polar Lipids),挤压通过100nm聚碳酸酯膜20~30次;挤压过程中,外泌体膜被破坏,与超氧化物歧化酶(SOD)剧烈混合;外泌体载药后通过90000~110000g离心除去游离的SOD和皂素,离心产物以PBS缓冲液重悬后得到本发明所述的外泌体-超氧化物歧化酶纳米制剂(SOD@EXO);
上述方法中,外泌体供体细胞为人胚肾上皮细胞(HEK293),外泌体蛋白质量与超氧化物歧化酶(SOD)的质量比为1:0.2~0.8,皂素在PBS缓冲液中的浓度为1~2mg/mL。
本发明中所采用的挤出法结合皂苷辅助的策略,将SOD担载进入外泌体,相较于游离SOD,具有如下优点:合成条件温和,生物相容性好,对酶活性影响小,装载量高,具有良好的膜通透性,释放时间长的特点。
本发明所构建的SOD@EXO纳米制剂基于超氧化物歧化酶抗自由基的活性,以N2野生型秀丽隐杆线虫为衰老模型,进行给药,SOD@EXO可以明显提高线虫抵抗外界不良环境的能力,延长线虫的寿命。
综上,本发明通过挤出法结合皂苷辅助将SOD分子担载进外泌体之中,合成了SOD@EXO纳米制剂。以N2野生型秀丽隐杆线虫为模式生物,证明SOD@EXO纳米制剂能显著抵抗线虫的氧化应激状态,降低线虫体内活性氧水平,提高线虫健康状况,延长线虫寿命。因此,该纳米制剂在清除自由基、延缓衰老方面具有很好的应用前景,在制备治疗抗衰老的药物方面可以得到应用,成为一类具有良好抗衰老效果的纳米制剂。
附图说明
图1:实施例1合成的EXO、SOD@EXO的透射电镜图。
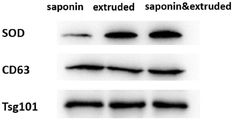
图2:SOD@EXO中外泌体标志蛋白及SOD的鉴定图。
图3:游离SOD与SOD@EXO的相对酶活性柱形图。
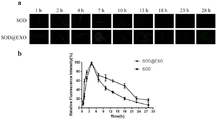
图4:(a)为线虫给药后不同时间的荧光显微镜图,b)为线虫给药后不同时间由ImageJ软件计算得到的荧光数据曲线,线虫摄取时间4h。
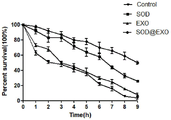
图5:SOD@EXO对线虫氧化损伤能力的存活率曲线。横坐标为时间,纵坐标为线虫存活率。
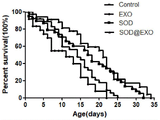
图6:SOD@EXO抵抗线虫的衰老曲线。图中横坐标为线虫寿命,纵坐标为线虫存活率。
具体实施方式
下面给出的实施例子是对本发明作进一步说明,以便于本专业技术人员更全面地理解本发明。但所给出的实施例不能理解为对本发明保护范围的限制,因而该专业的技术人员根据上述发明内容所做出的非本质的改进和调整也应属于本发明保护范围。
实施例1
首先将人胚肾上皮细胞(HEK293)培养于DMEM培养基中,DMEM培养基中含有10%(体积百分数)去除外泌体的胎牛血清(FBS),培养条件为37℃,5%(体积百分数)二氧化碳环境,待细胞融合度达90%后,获得HEK293细胞上清液500mL。
将上述HEK293细胞上清通过300g离心10min去除悬浮细胞,2000g离心10min去除细胞碎片,10000g离心30min去除较大的颗粒状物质;将获得的上清液经0.22μm的滤膜过滤,去除大于220nm的颗粒,再将上清液经过100kDa截留分子量的超滤离心管进行浓缩,取超滤离心管上层浓缩清液在超速离心管中以100000g离心1.5h,去除上清液,保留沉淀外泌体;用PBS缓冲液重悬沉淀外泌体,在相同的离心条件下再次离心,去除上清液,保留沉淀外泌体;最后以1mL的PBS缓冲液重悬得到外泌体溶液(Exosome),即空载外泌体(Exosome)溶液,其中外泌体的浓度是2mg/mL;
将SOD(来源于牛血)和皂素加入到上述1mL溶有外泌体的PBS缓冲液中,皂素浓度为1mg/mL,外泌体蛋白质量与SOD质量比为1:0.5,上述混合物装入基于注射器的脂质体挤出器并通过100nm聚碳酸酯膜21次。挤压过程中,外泌体膜被破坏,与药物剧烈混合。载药后通过100000g离心除去游离的SOD和皂素,最后再以1mL PBS缓冲液重悬即得到本发明所述的外泌体-超氧化物歧化酶纳米制剂(SOD@EXO)溶液。
另外,在上述方法中,加皂素而不采取挤压载药方式,可以制备得到外泌体-超氧化物歧化酶纳米制剂,记为SOD@EXO,saponin;不加皂素而采取挤压载药方式,可以制备得到外泌体-超氧化物歧化酶纳米制剂,记为SOD@EXO,extruded;加皂素并采取挤压载药方式制备得到的外泌体-超氧化物歧化酶纳米制剂,记为SOD@EXO saponin&extruded。
实施例2
将实施例1合成的空载外泌体溶液及3种外泌体-超氧化物歧化酶纳米制剂(SOD@EXO)的PBS重悬液再分别用PBS缓冲液稀释至1mg/mL,涂于300目碳膜覆盖的铜网上,室温自然风干。将涂好纳米制剂的铜网用透射电镜检测,加速电压200kV。如图1所示,可以清楚地观察到担载SOD分子的外泌体呈球形。
实施例3
将实施例1得到的载外泌体溶液及3种外泌体-超氧化物歧化酶纳米制剂(SOD@EXO)的PBS重悬液再分别用PBS缓冲液稀释至1mg/mL,再分别溶于经0.22μm滤膜过滤过的PBS缓冲液中,最终制备成10μg/mL溶液,分别用Zetasizer Nano ZS90粒度电位仪测试样品的粒径。如表1所示,外泌体装载SOD前后粒径在100nm-200nm之间。
表1:实施例1合成的空载外泌体及3种纳米制剂的粒径表征数据
实施例4
将实施例1合成的3种SOD@EXO纳米制剂的PBS重悬液以PBS缓冲液稀释至5mg/mL,直接加入5×SDS上样缓冲液,100℃煮样15min,煮样后冷却至室温后分装放入-20℃冰箱保存。配制分离胶浓度12%(体积百分数)的SDS-PAGE凝胶并且80V预电泳5min,上样时,每孔100μg蛋白量的Exosome或SOD@EXO,80V电压20min将蛋白样品压缩至浓缩胶/分离胶界面,更改电压至120V,约1h后电泳至凝胶下缘。以300mA恒流冰浴2h将细胞全蛋白转至聚偏二氟乙烯膜(PVDF)上。将膜取出晾干后,将PVDF膜按照不同分子量(上样时有marker条带显示分子量)分割至合适大小,放入至5%(质量体积比)脱脂奶粉/0.1%(体积百分数)Tween 20/PBS溶液,在常温下封闭PVDF膜2h,去除膜的蛋白非特异结合。取出后以滤纸吸干,放入SOD蛋白与外泌体蛋白标志物CD63、Tsg101的抗体于杂交袋中,抗体以3%(质量与体积比)BSA/PBS溶液稀释至适当比例(CD63 1:500;SOD 1:5000;Tsg101 1:1000)加入杂交袋中,在4℃摇动孵育过夜。0.1%(体积百分数)Tween20/PBS清洗三次,每次10min以除去抗体的非特异性结合,加入3%(质量与体积比)BSA/PBS溶液稀释相应的二抗(一抗是能和非抗体性抗原(特异性抗原)特异性结合的蛋白。二抗是能和抗体结合,即抗体的抗体,其主要作用是检测抗体的存在,放大一抗的信号。1:5000)。0.1%(体积与体积比)Tween20/PBS清洗三次,每次10min以除去二抗的非特异性结合,最后用ECL显色试剂盒曝光显影。如图2所示,挤出法结合皂苷辅助担载SOD分子的外泌体担载量最高,同时也说明了外泌体对SOD的成功装载。
实施例5
制备SOD@EXO前用FITC标记SOD(以SOD和FITC 2:1的质量比,在10mL的PBS里室温过夜。通过10kDa超滤管去除反应液中游离的FITC,4000g离心30min,离心约10次,直到超滤管下层滤出液为无色)。外标法检测SOD-FITC@EXO中的载药量与包封率。标准曲线的建立过程如下:在1mL PBS体系中,分别溶解0、5、10、20、40、80ng的SOD-FITC,以激发波长495nm,发射波长为519nm测定荧光强度。三份平行样的上清液检测荧光强度,用于测定SOD的载药量与包封率(通过测定上清的荧光强度,得到上清未包载的游离SOD的含量,包封率为外泌体内包载的SOD与总投入的SOD的比值;载药量是外泌体内包载的SOD的量与(外泌体包载SOD量+外泌体蛋白质量之和)的比值。通过荧光强度对照标准曲线得到上清未包载的游离SOD浓度,进而得到含量)。如表2所示(EE是包封率,DLC是载药量),挤出法结合皂苷辅助的SOD@EXO的包封率为38.67±1.11%,载药量为16.20±0.40%,满足后续实验的基本要求。
表2:实施例1合成的3种纳米制剂的包载率和载药量数据
实施例6
采用邻苯三酚自氧化法和WST-8法检测游离SOD和SOD@EXO saponin&extruded的抗氧化活性。邻苯三酚自氧化法:将990μL Tris-HCl缓冲液,在25℃条件下孵育20min后,加入10μL的邻苯三酚溶液启动反应,连续测定1min以内325nm处吸光度的变化。
样品组和对照组测定如下:在比色杯中分别加入750μL和760μL Tris-HCl缓冲液,然后加入240μL相应的SOD或SOD@EXO saponin&extruded样品液,样品组测定加入10μL的邻苯三酚溶液,之后在325nm波长下连续监测1min内吸光度的变化。邻苯三酚自氧化抑制率=(&自氧化-&样品)/&自氧化×100%,其中&自氧化、&样品分别为1min后监测的吸光度。SOD酶活性通过邻苯三酚自氧化抑制率计算得出,定义为将邻苯三酚自氧化速率抑制50%的酶量为一个活力单位。WST-8法:本专利对SOD酶活性检测使用的是“总SOD活性检测试剂盒(WST-8法)”(碧云天),试剂盒含有SOD样品制备液、SOD检测缓冲液、WST-8、酶溶液、反应启动液(40×)。WST-8可以和黄嘌呤氧化酶(Xanthine Oxidase,XO)催化产生的超氧化物阴离子(O2·-)反应产生水溶性的甲臜染料(formazan dye),由于SOD能催化超氧化物阴离子发生歧化作用,所以该反应步骤可以被SOD所抑制,因此SOD的活性与甲臜染料的生成量成负相关,从而通过对WST-8产物的比色分析即可计算SOD的酶活力。如图3所示,当SOD分子经过外泌体包载后,仍具有很强的酶活性,其活性约为游离SOD分子的78%。
实施例7
选用刚进入成虫期(L4期)的N2野生型线虫进行实验,线虫共分为2组,分别为SOD组和SOD@EXO saponin&extruded组。将成虫转移至给药板,每组100只,给药时间4h,4h后转移至新的板中。从开始给药起,每隔一定时间用左旋咪唑麻醉线虫,置于3%(质量体积比)琼脂覆盖的载玻片上,使用奥林巴斯公司的IX73P1F荧光显微镜进行拍照,Image JSoftware测定荧光强度。如图4(b)所示,线虫给药后,荧光强度升高,4h后,体内荧光衰减,游离SOD半衰期7.42h,SOD@EXO半衰期19.32h,可见游离药包载于外泌体后释放时间更长。
实施例8
选用刚进入成虫期(L4期)的N2野生型线虫进行实验,选用胡桃醌刺激线虫使线虫体内过量产生活性氧,从而对线虫造成氧化损伤。线虫共分为4组,分别为Control组、SOD组、SOD@EXO saponin&extruded组及EXO组。用含有相应药物的OP50线虫培养基培养线虫(SOD、SOD@EXO的酶当量为1μM,Control组不给药),48h后,换成含有胡桃醌(500μM)的OP50在20℃条件下培养线虫,每小时通过显微镜统计死亡的线虫的数量(线虫对铂丝的刺激无反应,认为线虫已经死亡),存活率为剩余存活线虫数与总线虫数的比值。从图5可以发现,随着时间延长,SOD@EXO组线虫的存活率明显高于SOD组。虽然游离SOD组的线虫也有较高的存活率,但是仍低于SOD@EXO组。以上研究表明,SOD@EXO纳米制剂能够显著增强线虫对氧化损伤的抵抗能力。
实施例9
选用刚进入成虫期(L4期)的N2野生型秀丽隐杆线虫进行实验。线虫寿命实验需要同期化的线虫,线虫同期化的方法为:选取L4期即将产卵的线虫,接种到涂有OP50的NGM培养基上,在20℃环境下使线虫产卵,然后进行同期化处理。将同期化线虫共分为4组,分别为Control组、SOD组、SOD@EXO saponin&extruded组及EXO组。同期化的线虫生长到L4期产卵之前,将其转移到含有产卵抑制剂FUDR(80μM)和相应的治疗药物(SOD、SOD@EXO的酶当量为1μM)的培养板上,并将这一天记为给药第一天。接着,每隔一天把线虫转移到新鲜的含有相应药物的培养板上,连续转移5次,期间每天观察并纪录线虫的死亡情况,如图6所示。SOD@EXO组的线虫在平均寿命、最大寿命方面都明显优于Control组和EXO组的线虫,说明SOD@EXO纳米制剂能够有效延缓线虫衰老,延长线虫寿命。
Claims (4)
1.一种外泌体-超氧化物歧化酶纳米制剂的制备方法,其步骤如下:
(1)获得外泌体供体细胞的上清液400~600mL,然后在200~400g条件下离心10~20min去除悬浮细胞,再在2000~4000g条件下离心10~20min去除细胞碎片,最后在8000~12000g条件下离心20~40min去除较大的颗粒状物质;
(2)将步骤(1)获得的上清液经0.22μm的滤膜过滤,去除大于220nm的颗粒,再将上清液经过100kDa截留分子量的超滤离心管进行浓缩,收集超滤离心管上层浓缩液;
(3)将步骤(2)得到的浓缩液在90000~110000g条件下离心1~2h,去除上清液,保留沉淀外泌体;然后用PBS缓冲液重悬此沉淀外泌体,在相同的条件下再次离心,去除上清液,保留沉淀外泌体;最后再以1~2mL的PBS缓冲液重悬此沉淀外泌体,其中外泌体的浓度为1~3mg/mL;
(4)将超氧化物歧化酶和皂素加入到步骤(3)得到的溶有外泌体的PBS缓冲液中,然后装入基于注射器的脂质体挤出器,挤压通过100nm聚碳酸酯膜20~30次;挤压过程中,外泌体膜被破坏,与超氧化物歧化酶剧烈混合;外泌体载药后通过90000~110000g离心除去游离的超氧化物歧化酶和皂素,离心产物以PBS缓冲液重悬后得到外泌体-超氧化物歧化酶纳米制剂。
2.如权利要求1所述的一种外泌体-超氧化物歧化酶纳米制剂的制备方法,其特征在于:外泌体供体细胞为人胚肾上皮细胞HEK293。
3.如权利要求1所述的一种外泌体-超氧化物歧化酶纳米制剂的制备方法,其特征在于:外泌体蛋白质量与超氧化物歧化酶的质量比为1:0.2~0.8,皂素在PBS缓冲液中的浓度为1~2mg/mL。
4.一种外泌体-超氧化物歧化酶纳米制剂,其特征在于:是由权利要求1~3任何一项所述的方法制备得到。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910593342.1A CN110251663B (zh) | 2019-07-03 | 2019-07-03 | 一种具有抗衰老作用的外泌体-超氧化物歧化酶纳米制剂及其制备方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910593342.1A CN110251663B (zh) | 2019-07-03 | 2019-07-03 | 一种具有抗衰老作用的外泌体-超氧化物歧化酶纳米制剂及其制备方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110251663A CN110251663A (zh) | 2019-09-20 |
| CN110251663B true CN110251663B (zh) | 2022-04-01 |
Family
ID=67924027
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910593342.1A Active CN110251663B (zh) | 2019-07-03 | 2019-07-03 | 一种具有抗衰老作用的外泌体-超氧化物歧化酶纳米制剂及其制备方法 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110251663B (zh) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112021558B (zh) * | 2020-09-11 | 2023-08-04 | 福州大学 | 一种自组装Cu/Zn-SOD纳米颗粒及其应用 |
| WO2022108424A1 (ko) * | 2020-11-23 | 2022-05-27 | 주식회사 제노포커스 | 호흡기 바이러스 감염의 치료 또는 예방을 위한 수퍼옥시드 디스무타아제 활성을 갖는 폴리펩티드 및 세포외 소포체의 용도 |
| CN113234677A (zh) * | 2021-04-27 | 2021-08-10 | 西南医科大学附属医院 | 一种从体外肿瘤组织中提取外泌体的方法 |
| CN114831894A (zh) * | 2021-12-17 | 2022-08-02 | 广西萌大夫生物技术有限公司 | 一种含nmn的氧化敏感纳米粒子及其制备方法和用途 |
| CN118947903B (zh) * | 2024-07-25 | 2025-09-02 | 陕西科技大学 | 一种抗氧化应激制剂及其制备方法与应用 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015161184A1 (en) * | 2014-04-18 | 2015-10-22 | University Of Massachusetts | Exosomal loading using hydrophobically modified oligonucleotides |
| CN105535022A (zh) * | 2016-01-12 | 2016-05-04 | 浙江生创精准医疗科技有限公司 | 外泌体在制备治疗急性肝衰竭的药物中的用途和药物组合物 |
| WO2017173034A1 (en) * | 2016-03-30 | 2017-10-05 | The University Of North Carolina At Chapel Hill | Biological agent-exosome compositions and uses thereof |
| WO2018214694A1 (zh) * | 2017-05-23 | 2018-11-29 | 北京希诺赛尔健康科技推广有限公司 | 外泌体在皮肤美白制剂中的应用 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017526388A (ja) * | 2014-09-05 | 2017-09-14 | エクサーカイン コーポレイションExerkine Corporation | エキソソームの単離 |
-
2019
- 2019-07-03 CN CN201910593342.1A patent/CN110251663B/zh active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015161184A1 (en) * | 2014-04-18 | 2015-10-22 | University Of Massachusetts | Exosomal loading using hydrophobically modified oligonucleotides |
| CN105535022A (zh) * | 2016-01-12 | 2016-05-04 | 浙江生创精准医疗科技有限公司 | 外泌体在制备治疗急性肝衰竭的药物中的用途和药物组合物 |
| WO2017173034A1 (en) * | 2016-03-30 | 2017-10-05 | The University Of North Carolina At Chapel Hill | Biological agent-exosome compositions and uses thereof |
| WO2018214694A1 (zh) * | 2017-05-23 | 2018-11-29 | 北京希诺赛尔健康科技推广有限公司 | 外泌体在皮肤美白制剂中的应用 |
Non-Patent Citations (2)
| Title |
|---|
| Exosome-dependent and independent mechanisms are involved in prion-like transmission of propagated Cu/Zn superoxide dismutase misfolding;Leslie I Grad等;《Prion》;20141231;第331-335页 * |
| 外泌体作为药物递送载体的研究进展;张盈盈;《药学学报》;20190419;第54卷(第6期);第1010-1016页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN110251663A (zh) | 2019-09-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN110251663B (zh) | 一种具有抗衰老作用的外泌体-超氧化物歧化酶纳米制剂及其制备方法 | |
| Dou et al. | Chimeric apoptotic bodies functionalized with natural membrane and modular delivery system for inflammation modulation | |
| JP6463493B2 (ja) | 細胞の脂質膜に由来するナノ小胞及びその用途 | |
| AU2007305076B2 (en) | Immune modulators, preparations and compositions including immune modulators, tests for evaluating the activity of immune modulators and preparations and compositions including the same, and methods | |
| JP2728166B2 (ja) | 導入抗原タンパク質を有する動物由来細胞 | |
| CN111467483A (zh) | 一种包裹肿瘤细胞膜的磁性纳米微载体的制备方法和应用 | |
| US20230002743A1 (en) | Self-assembled catalase nanoparticle and preparation method therefor and use thereof | |
| Saldanha et al. | Antifungal activity of amphotericin B conjugated to nanosized magnetite in the treatment of paracoccidioidomycosis | |
| CN108815520A (zh) | 一种仿生二元协同纳米载体及其制备方法与应用 | |
| CN117138042B (zh) | 一种二价无机金属离子/光敏剂蛋白纳米粒及制备和应用 | |
| CN114404571A (zh) | 一种装载化疗药物且tigit过表达的工程化载药细胞膜囊泡以及制备方法和应用 | |
| CN112791061B (zh) | 一种具有靶向长循环的多级仿生纳米药物载体的制备方法 | |
| CN109568269B (zh) | 一种具有诊疗功能外泌体及其制备方法 | |
| CN116606766A (zh) | 一种食淀粉乳杆菌胞外囊泡的制备方法及其应用 | |
| Espe et al. | Hydrolyzed fish proteins reduced activation of caspase-3 in H2O2 induced oxidative stressed liver cells isolated from Atlantic salmon (Salmo salar) | |
| CN116251062B (zh) | 一种细菌膜-脂质体载药系统的制备方法及其应用 | |
| CN118121569A (zh) | 装载Gli1-shRNA核酸序列靶向肝星状细胞防治肝纤维化的LNPs纳米粒 | |
| CN117771206B (zh) | Sting激动剂仿生纳米递送系统及其制备方法和应用 | |
| CN115353550B (zh) | 一种靶向血管细胞黏附分子-1的自组装硒肽、硒肽纳米药物及其制备方法和应用 | |
| CN115137723B (zh) | 醋酸钙梯度主动载药法制备维a酸外泌体模拟物 | |
| CN118892465A (zh) | 一种用于三阴性乳腺癌的纳米药物及其制备方法 | |
| Wang et al. | Neutrophil-targeted Mn3O4 nanozyme treats myocardial ischemia reperfusion injury by scavenging reactive oxygen species | |
| CN114134065B (zh) | 生物胞膜系统及其制备方法、应用 | |
| CN119499213A (zh) | 一种应用于敌草快急性中毒的仿生纳米制剂及其制备方法和应用 | |
| CN118079012B (zh) | 一种调控肿瘤脂代谢重编程协同增效铁死亡的多功能纳米囊泡及其制备方法和应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |