CN107710350B - 基于各向异性纳米结构的磁可调谐光子晶体 - Google Patents
基于各向异性纳米结构的磁可调谐光子晶体 Download PDFInfo
- Publication number
- CN107710350B CN107710350B CN201680037841.7A CN201680037841A CN107710350B CN 107710350 B CN107710350 B CN 107710350B CN 201680037841 A CN201680037841 A CN 201680037841A CN 107710350 B CN107710350 B CN 107710350B
- Authority
- CN
- China
- Prior art keywords
- anisotropic
- magnetic
- nanoparticles
- iron
- nanoellipsoids
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F41/00—Apparatus or processes specially adapted for manufacturing or assembling magnets, inductances or transformers; Apparatus or processes specially adapted for manufacturing materials characterised by their magnetic properties
- H01F41/02—Apparatus or processes specially adapted for manufacturing or assembling magnets, inductances or transformers; Apparatus or processes specially adapted for manufacturing materials characterised by their magnetic properties for manufacturing cores, coils, or magnets
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C1/00—Treatment of specific inorganic materials other than fibrous fillers; Preparation of carbon black
- C09C1/22—Compounds of iron
- C09C1/24—Oxides of iron
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F1/00—Metallic powder; Treatment of metallic powder, e.g. to facilitate working or to improve properties
- B22F1/05—Metallic powder characterised by the size or surface area of the particles
- B22F1/054—Nanosized particles
- B22F1/0551—Flake form nanoparticles
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G49/00—Compounds of iron
- C01G49/02—Oxides; Hydroxides
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C1/00—Treatment of specific inorganic materials other than fibrous fillers; Preparation of carbon black
- C09C1/0081—Composite particulate pigments or fillers, i.e. containing at least two solid phases, except those consisting of coated particles of one compound
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C3/00—Treatment in general of inorganic materials, other than fibrous fillers, to enhance their pigmenting or filling properties
- C09C3/06—Treatment with inorganic compounds
- C09C3/063—Coating
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F1/00—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties
- H01F1/0036—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties showing low dimensional magnetism, i.e. spin rearrangements due to a restriction of dimensions, e.g. showing giant magnetoresistivity
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F1/00—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties
- H01F1/0036—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties showing low dimensional magnetism, i.e. spin rearrangements due to a restriction of dimensions, e.g. showing giant magnetoresistivity
- H01F1/0045—Zero dimensional, e.g. nanoparticles, soft nanoparticles for medical/biological use
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F1/00—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties
- H01F1/01—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials
- H01F1/012—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials adapted for magnetic entropy change by magnetocaloric effect, e.g. used as magnetic refrigerating material
- H01F1/017—Compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F1/00—Metallic powder; Treatment of metallic powder, e.g. to facilitate working or to improve properties
- B22F1/05—Metallic powder characterised by the size or surface area of the particles
- B22F1/054—Nanosized particles
- B22F1/0547—Nanofibres or nanotubes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F2998/00—Supplementary information concerning processes or compositions relating to powder metallurgy
- B22F2998/10—Processes characterised by the sequence of their steps
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F2999/00—Aspects linked to processes or compositions used in powder metallurgy
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y20/00—Nanooptics, e.g. quantum optics or photonic crystals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y25/00—Nanomagnetism, e.g. magnetoimpedance, anisotropic magnetoresistance, giant magnetoresistance or tunneling magnetoresistance
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y40/00—Manufacture or treatment of nanostructures
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/01—Particle morphology depicted by an image
- C01P2004/04—Particle morphology depicted by an image obtained by TEM, STEM, STM or AFM
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/10—Particle morphology extending in one dimension, e.g. needle-like
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/10—Particle morphology extending in one dimension, e.g. needle-like
- C01P2004/16—Nanowires or nanorods, i.e. solid nanofibres with two nearly equal dimensions between 1-100 nanometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/80—Particles consisting of a mixture of two or more inorganic phases
- C01P2004/82—Particles consisting of a mixture of two or more inorganic phases two phases having the same anion, e.g. both oxidic phases
- C01P2004/84—Particles consisting of a mixture of two or more inorganic phases two phases having the same anion, e.g. both oxidic phases one phase coated with the other
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S977/00—Nanotechnology
- Y10S977/70—Nanostructure
- Y10S977/762—Nanowire or quantum wire, i.e. axially elongated structure having two dimensions of 100 nm or less
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S977/00—Nanotechnology
- Y10S977/70—Nanostructure
- Y10S977/81—Of specified metal or metal alloy composition
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S977/00—Nanotechnology
- Y10S977/84—Manufacture, treatment, or detection of nanostructure
- Y10S977/89—Deposition of materials, e.g. coating, cvd, or ald
- Y10S977/892—Liquid phase deposition
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S977/00—Nanotechnology
- Y10S977/84—Manufacture, treatment, or detection of nanostructure
- Y10S977/895—Manufacture, treatment, or detection of nanostructure having step or means utilizing chemical property
- Y10S977/896—Chemical synthesis, e.g. chemical bonding or breaking
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S977/00—Nanotechnology
- Y10S977/902—Specified use of nanostructure
- Y10S977/932—Specified use of nanostructure for electronic or optoelectronic application
- Y10S977/952—Display
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Inorganic Chemistry (AREA)
- Organic Chemistry (AREA)
- Power Engineering (AREA)
- Nanotechnology (AREA)
- Manufacturing & Machinery (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Composite Materials (AREA)
- Health & Medical Sciences (AREA)
- Compounds Of Iron (AREA)
- Pigments, Carbon Blacks, Or Wood Stains (AREA)
- Silicon Compounds (AREA)
- Hard Magnetic Materials (AREA)
- Optical Modulation, Optical Deflection, Nonlinear Optics, Optical Demodulation, Optical Logic Elements (AREA)
Abstract
公开了一种形成磁可调谐光子晶体的方法,其包括:合成一种或更多种具有各向异性形状的前体纳米颗粒;用二氧化硅涂覆该一种或更多种各向异性前体纳米颗粒,以形成复合结构;通过化学反应将该一种或更多种各向异性前体纳米颗粒转化成磁性纳米材料;和在溶剂中使各向异性磁性纳米颗粒组装成光子晶体。
Description
技术领域
本发明涉及形成基于各向异性纳米结构的磁可调谐光子晶体的方法。
背景技术
已广泛探索胶体组装体以通过操纵光与物理周期性结构的相互作用来产生人工结构颜色。在自然例如蛋白石、鸟类羽毛和蝴蝶鳞片中容易发现,结构颜色是灿烂的彩虹色、金属质感且不受光漂白,不像常规的颜料或染料那样。大多数胶体组装过程模拟蛋白石的形成并由单分散性胶体球产生密堆积结构。但是,生命系统常涉及各向异性基元(motif)(例如板和棒)的非密堆积有序组装体,从而展示显著更复杂的结构颜色响应,包括强角依赖性和偏振效应。因为预期在胶体组装体中采用各向异性构造单元可能产生光子结构,已作出努力来组织细长的颗粒例如聚苯乙烯和ZnS,尽管组装方法相当常规且所得的晶体接近密堆积并且与来自球形颗粒的那些相比不展现独特的光子响应。
各向异性颗粒的重要优势之一是它们常具有形状依赖性物理和化学性质,其可增加更多的自由度来操纵所得超结构的总体性质。这可能是制造场响应型胶体光子结构特别感兴趣的,其中静态或动态结构变化通常伴随着光子性质的转变。但是,沿着该方向的努力非常有限,主要归因于高质量各向异性构造单元的不可获得性以及组装和调谐有效机制的缺乏。
发明内容
公开了形成磁可调谐光子晶体的方法,其包括:合成一种或更多种具有各向异性形状的前体纳米颗粒;用二氧化硅涂覆该一种或更多种各向异性前体纳米颗粒,以形成复合结构;通过化学反应将所述一种或更多种各向异性前体纳米颗粒转化成磁性纳米材料;和在溶剂中使各向异性磁性纳米颗粒组装成光子晶体。
根据一个示例性实施方案,所述一种或更多种各向异性前体纳米颗粒是羟基氧化铁(FeOOH)纳米棒。
附图简述
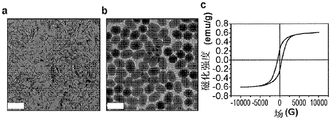
图1(a)-1(c)显示(a)典型的FeOOH纳米棒的TEM图像;(b)通过用二氧化硅涂覆这些FeOOH纳米棒并随后在H2中将它们还原获得的磁性纳米椭球的TEM图像;和(c)纳米椭球的对应的磁滞回线。比例尺:200nm。
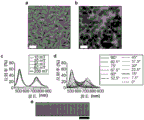
图2(a)-2(d)显示(a)纳米椭球在磁场下的自发排列的图;(b)在具有不同强度的垂直磁场和平行磁场下的光子结构的反射光谱;(c)在相对于光的方向具有不同方向的磁场下的光子结构的反射光谱;和(d)显示在非理想线性海尔贝克阵列下的包封在扁平玻璃管中的纳米椭球的光子响应的数码照片。比例尺:5mm。
图3(a)-3(c)显示(a)在不存在磁场的情况下,不同体积分数下的纳米椭球的胶体分散体的反射光谱;(b)具有体积分数梯度的玻璃毛细管中的纳米椭球的分散体的数码图像;和(c)在存在或不存在磁场的情况下,光子结构的反射波长对于纳米椭球的体积分数的依赖性。
图4(a)-4(e)显示(a)典型的FeOOH纳米棒的TEM图像;(b)通过用二氧化硅涂覆这些FeOOH纳米棒并随后在H2中将它们还原获得的磁性纳米椭球的TEM图像;比例尺:200nm;(c)在具有不同强度的垂直磁场和平行磁场下的光子结构的反射光谱;(d)在相对于光的方向具有不同方向的磁场下的光子结构的反射光谱;和(e)显示在非理想线性海尔贝克阵列下的包封在扁平玻璃管中的纳米椭球的光子响应的数码照片。(比例尺:5mm)。
详述
根据一个示例性实施方案,公开了一种组装体,其受益于针对高度均匀的各向异性胶体椭球的独特的合成。因为均匀的磁性椭球并非直接可获得的,所以采取间接方法,其通过首先合成均匀的羟基氧化铁(FeOOH)纳米棒(图1(a)),随后用二氧化硅来涂覆它们以形成复合椭球,以及最终通过还原将FeOOH转化成磁性金属铁。
根据一个示例性实施方案,二氧化硅涂层在此可起到多个重要作用。首先,二氧化硅涂层将FeOOH纳米棒的尺寸增加至适合于在可见光谱中产生光子响应的尺寸范围。第二,二氧化硅涂层提供在还原期间防止纳米棒崩解的保护机制。从FeOOH转化成Fe涉及脱水和还原反应,这两者均引起显著的形态变化。如图1(b)中所示,纳米棒在还原后主要发生收缩并崩解成小块。但是,总体的棒形状由于在二氧化硅壳内产生的椭球空间而很好地保持。最后,二氧化硅层起作将磁性纳米棒分开一定距离并限制其磁吸引的隔离体的作用,从而防止其胶体分散体聚集(尽管纳米棒具有铁磁性质)。
与球形构造单元相比较,各向异性纳米椭球的独特特征在于其组装体显示光子响应对场方向的强依赖性。除了对于描述来自球形构造单元的组装体通常考虑的位置序之外,当组装纳米椭球时,人们应考虑取向序。由于其中包埋的各向异性磁性核,可通过外部磁场容易地控制纳米椭球的取向。施加磁场之后,纳米椭球旋转并将其长轴与场方向平行排列,如图2(a)中图示。此旋转不仅引起取向序上的变化,而且影响位置序以及随后的组装体的光子性质。同时,还认为场强在排序的完善上起到重要作用,因为其决定纳米椭球可以排列的程度。
为了研究场强和场方向的效应,检查了由纳米椭球组装的结构在具有不同强度和方向的磁场下的光子性质。将纳米椭球的含水分散体浓缩至所需的体积分数,以允许其自发排序成胶体晶体。图2(b)显示了在与入射光方向平行和垂直的磁场下的光子结构的反射光谱。垂直于光的磁场也将纳米椭球排列成垂直取向,使得平面间间距主要由纳米椭球的短轴决定,导致在较短波长下的反射峰。当将场方向切换成与光平行时,平面间间距由纳米椭球的长轴决定,导致在较长波长下的反射峰。另一方面,对于两种样品,均发现场强影响反射的强度而不是波长。认为增加的反射强度由在较强场下的纳米椭球的较好的取向序引起:因为包埋在其内部的有限量的磁性物种,纳米椭球的旋转要求足够高的磁场以允许磁矩克服转动阻力。当固定场方向时,改变场强不会改变反射波长。纳米椭球之间的磁性相互作用对于组装体的平面间间距的贡献似乎是可忽略的,这主要是由于磁性纳米棒被相对厚的二氧化硅涂层有效分隔。
不同于使用球形颗粒组装的常规胶体晶体(其中仅可通过经由场强的变化控制平面间间距来实现光子性质的调谐),调谐场方向导致纳米椭球的同时旋转,其进一步导致组装体的平面间间距以及其光子性质的变化。根据一个示例性实施方案,在旋转磁场下研究由纳米椭球组装的结构的光子性质。如图2(c)中所示,反射峰的波长在场方向垂直于入射光时达到最小值,并且随着场从垂直于入射光切换成平行于入射光而逐渐红移。反射波长的这种移动即刻(在小于一秒内)响应于场方向的改变并且是完全可逆的。这种磁调谐的显著特征在于当磁场平行于入射光或垂直于入射光时在两个端点处实现最大强度。当场方向从这两个端点切换走时,相对强度减小并且在中点(距平行方向和垂直方向~45°)处达到最小值,产生大体U形的反射峰分布。当与之前报导的一维光子链(其中最强的反射仅可在中间场强下获得并且反射峰的分布是倒U形曲线)相比较时,这是一个主要的差别。
纳米椭球组装体的取向依赖性可在于具有不均匀的场方向的磁场下产生光子模式中找到直接用途。如图2(d)中所展示的,当经受由非理想线性海尔贝克阵列产生的复合磁场(其具有磁化强度的空间旋转模式)时,包封在扁平玻璃管中的纳米椭球的分散体展现包含蓝色和绿色条纹的多色图案。
除了取向之外,光子组装体的平面间间距还由纳米椭球的体积分数决定。随着体积分数降低,纳米椭球之间的距离增加,导致晶格膨胀以及反射波长红移。在不存在磁场时,记录不同体积分数下的光子组装体的反射光谱并随后展示在图3(a)中。随着体积分数从32%降低至10%,反射峰从425nm移动至660nm。一致地,在图3(b)中在具有体积分数梯度(其通过在3000rpm下离心5分钟实现)的纳米椭球分散体中观察到彩虹样颜色效应。
场方向和体积分数两者的协同作用允许光子组装体的光学性质的宽范围的调谐。图3(c)绘制了在平行磁场或垂直磁场或无磁场的情况下,峰位置随溶液的体积分数的变化的曲线。有趣的是,对于各体积分数,在平行磁场和垂直磁场下的光子组装体的峰位置的差异恒定保持在约105nm,如由黑色虚线所代表的。此峰位置差异归因于纳米椭球的长轴和短轴的差异,并因此不随着体积分数而改变。在不存在磁场的情况下,纳米椭球的取向较不均匀。组装体的平面间间距和反射峰的位置因此总是介于在平行磁场下的那些和在垂直磁场下的那些之间。在不存在外场的情况下,纳米椭球倾向于将其自身沿着样品容器的表面排列,其垂直于入射角,使得反射峰接近在垂直磁场情况下的反射峰,即使在相对低的体积分数下(图3(c))。随着体积分数增加,纳米椭球的取向序在不存在磁场的情况下增强并最终变得与通过磁场排列的情况相同,如由在~32%的高体积分数下峰位置的重合所证实的。增强的取向序可通过在较高体积分数下各纳米椭球的排除体积降低来解释。纳米椭球降低取向熵但增加平移熵以达到能量有利的结构,其中大多数纳米椭球平行排列。
根据一个示例性实施方案,公开了一新类磁响应光子晶体,其衍射性能可通过控制场方向来广泛调谐。与由球形颗粒制备的常规胶体晶体(其中光子性能的动态调谐主要通过控制场强来实现)形成对照,由高度均匀的形状和磁性各向异性纳米椭球组装的新颖胶体晶体在场方向垂直于入射角时衍射最小波长,并在场切换成平行于入射角时衍射最大波长。当场平行于入射光或垂直于入射光时,衍射强度达到最大值,并且当场方向切换成斜的(off-angle)时,衍射强度降低,展示独特的U形反射峰分布。响应于场方向变化的衍射移动是自发且完全可逆的。目前的系统不仅允许更多的研究形状和磁性各向异性纳米结构的组装体行为的机会,而且提供用于各种颜色呈现和显示应用的构建新颖有源光学部件的新平台。
在该方法中,起始材料不限于FeOOH纳米棒,并可扩展至其它金属氢氧化物,例如Co(OH)2、Ni(OH)2和Fe(OH)3。纳米颗粒的形态不限于椭球,并可扩展至棒、板、扁球等等。用于将纳米颗粒组装成光子结构的溶剂可为,但不限于水、乙醇、乙二醇(glycol)和其它极性或非极性溶剂。用于制备基于纳米椭球的光子结构的典型途径如下所列:
实施例1:
FeOOH纳米棒的合成:
根据一个示例性实施方案,该方法以FeOOH纳米棒的合成开始。在70nm FeOOH纳米棒的典型合成中,将FeCl3·6H2O溶解在40mL去离子水中,并将Fe3+的浓度调节至0.02M。在11000rpm下离心3分钟后丢弃未溶解的沉淀物。将上清液加入三颈烧瓶中并在81℃下在磁力搅拌下加热12小时。随后通过离心将颗粒分离,用水洗涤数次并分散在7.2mL水中。
FeOOH纳米棒的表面改性:
首先用聚丙烯酸(PAA)将如此合成的FeOOH纳米棒的表面功能化。通常将3mL上述FeOOH分散体和1mL PAA溶液(7.2mg/mL)在超声处理下加入16mL水中。随后将混合物搅拌12小时,以允许纳米棒的PAA功能化。此后,通过离心除去溶液中的过量PAA,并将FeOOH纳米棒重新分散在3mL水中。
FeOOH@SiO2纳米椭球的合成:
将PAA改性的FeOOH的3mL含水分散体加入20mL异丙醇中,随后添加1mL氢氧化铵(~28%wt)。对于70nm FeOOH纳米棒的二氧化硅涂层,每30分钟向以上混合物中添加400μL原硅酸四乙酯(TEOS),直至TEOS的总量达到2.4mL。在额外的1小时反应之后,通过离心将FeOOH@SiO2纳米椭球分离、用乙醇和水洗涤数次,并分散在乙醇中。
FeOOH@SiO2纳米椭球的还原:
将FeOOH@SiO2纳米椭球在N2保护下加热至500℃,并随后在该温度下通过纯H2还原2小时,以生产Fe@SiO2纳米椭球。
纳米椭球组装成光子结构:
通过超声处理30分钟将如此还原的纳米椭球重新分散在水中。将分散体加入三颈烧瓶中并在100℃下回流2小时。通过离心将纳米椭球分离,并用水洗涤数次。随后实施尺寸选择并通过在2000rpm下离心2分钟丢弃非分散性聚集体。将纳米椭球分散体首先浓缩至最大体积分数(超过该体积分数将形成聚集体),并随后将一定量的水加入分散体中以产生所需浓度。
实施例2:
还可合成具有较高长径比的纳米椭球。在110nm FeOOH纳米棒的典型合成中,将FeCl3·6H2O溶解在40mL去离子水中,并将Fe3+的浓度调节至0.1M。向溶液中添加1g CTAB。在11000rpm下离心3分钟后丢弃未溶解的沉淀物。将上清液加入三颈烧瓶中并在90℃下在磁力搅拌下加热18小时。随后通过离心将颗粒分离,用水洗涤数次并分散在36mL水中。
通过类似的程序用PAA将如此合成的FeOOH纳米棒进行改性。对于二氧化硅涂层,每30分钟向以上混合物中添加200μL TEOS,直至TEOS的总量达到1.2mL。在额外的1小时反应之后,通过离心将FeOOH@SiO2纳米椭球分离、用乙醇和水洗涤数次,并分散在乙醇中。随后与以上过程类似,通过H2还原纳米椭球、将其重新分散在水中,并组装成光子结构。如此组装的光子结构还显示角依赖性性质,并在将其置于非理想线性海尔贝克阵列下时展示彩虹样图案。
但是,本发明不限于上文所述和在附图中所示的实施方案和变型。在不偏离如所附权利要求中所定义的本发明的精神和范围的情况下,本领域技术人员可实现各种改变、修改和等同方案。明确期望落入权利要求书范围内的所有的此类改变、修改和等同方案被权利要求书所涵盖。
Claims (15)
1.一种形成磁可调谐光子晶体的方法,其包括:
合成具有各向异性形状的前体纳米颗粒;
用二氧化硅涂覆该各向异性前体纳米颗粒,以形成复合结构;
通过化学反应将所述各向异性前体纳米颗粒转化成磁性纳米材料;和
在溶剂中使所述各向异性磁性纳米颗粒组装成光子晶体,
其中所述各向异性前体纳米颗粒是羟基氧化铁纳米棒。
2.根据权利要求1所述的方法,其包括:
将原硅酸四乙酯加入蒸馏水、乙醇、氨和各向异性前体纳米颗粒的混合物中;和
通过离心分离该涂覆的复合结构。
3.根据权利要求1所述的方法,其包括:
加热该涂覆的复合结构;和
用还原剂还原所述涂覆的复合结构,以使得所述涂覆的复合结构为有磁性的。
4.根据权利要求1所述的方法,其包括:
通过超声处理将如此还原的磁性颗粒重新分散;
对所述颗粒实施尺寸选择;和
通过离心丢弃非分散性聚集体。
5.根据权利要求1所述的方法,其包含:
用于将纳米颗粒组装成光子结构的溶剂,所述溶剂为水、乙醇、乙二醇和其它极性或非极性溶剂。
6.根据权利要求1所述的方法,其中合成所述羟基氧化铁纳米棒的步骤包括:
各向异性前体纳米颗粒的基于溶液的合成;和
各向异性前体纳米颗粒的不含溶液的合成。
7.根据权利要求6所述的方法,其包括:
将铁盐溶解在去离子水中;
调节铁阳离子的浓度;
丢弃未溶解的沉淀物;和
加热上清液。
8.根据权利要求6所述的方法,其包括:
在高温下煅烧铁盐。
9.根据权利要求6所述的方法,其包括:
通过离心分离所述羟基氧化铁纳米棒。
10.根据权利要求6所述的方法,其包括:
用表面活性剂功能化羟基氧化铁纳米棒的表面。
11.根据权利要求7或权利要求8所述的方法,其中所述铁包含:
氯化铁、氮化铁、硫酸铁和/或其它可溶性铁化合物。
12.根据权利要求7所述的方法,其中所述上清液的加热温度包含:
在室温至2000°C范围内的温度。
13.根据权利要求10所述的方法,其中所述表面活性剂包含:
离子表面活性剂;和
非离子表面活性剂。
14.根据权利要求13所述的方法,其中所述离子表面活性剂包含:
聚丙烯酸或十六烷基三甲基溴化铵。
15.根据权利要求13所述的方法,其中所述离子表面活性剂包含:
聚乙烯吡咯烷酮。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010780714.4A CN111899973B (zh) | 2015-04-27 | 2016-04-27 | 基于各向异性纳米结构的磁可调谐光子晶体 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201562153228P | 2015-04-27 | 2015-04-27 | |
| US62/153228 | 2015-04-27 | ||
| PCT/US2016/029461 WO2016176267A1 (en) | 2015-04-27 | 2016-04-27 | Magnetically tunable photonic crystals based on anisotropic nanostructures |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010780714.4A Division CN111899973B (zh) | 2015-04-27 | 2016-04-27 | 基于各向异性纳米结构的磁可调谐光子晶体 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN107710350A CN107710350A (zh) | 2018-02-16 |
| CN107710350B true CN107710350B (zh) | 2020-09-01 |
Family
ID=57199401
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201680037841.7A Active CN107710350B (zh) | 2015-04-27 | 2016-04-27 | 基于各向异性纳米结构的磁可调谐光子晶体 |
| CN202010780714.4A Active CN111899973B (zh) | 2015-04-27 | 2016-04-27 | 基于各向异性纳米结构的磁可调谐光子晶体 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010780714.4A Active CN111899973B (zh) | 2015-04-27 | 2016-04-27 | 基于各向异性纳米结构的磁可调谐光子晶体 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US10796849B2 (zh) |
| EP (1) | EP3289596A4 (zh) |
| JP (1) | JP6776267B2 (zh) |
| KR (1) | KR102566859B1 (zh) |
| CN (2) | CN107710350B (zh) |
| WO (1) | WO2016176267A1 (zh) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108711480B (zh) * | 2018-04-03 | 2020-07-03 | 复旦大学 | 一种具有核壳结构磁性介孔二氧化硅纳米链及其制备方法 |
| CN109370263B (zh) * | 2018-11-13 | 2021-03-23 | 天津科技大学 | 一种全色可变结构生色材料的制备方法 |
| CN110194457A (zh) * | 2019-05-20 | 2019-09-03 | 重庆科技学院 | 一种SiO2中空纳米棒的制备方法和尺寸调控方法 |
| CN110223815A (zh) * | 2019-07-01 | 2019-09-10 | 苏州善恩纳米功能材料科技有限公司 | 一种高分散性磁性纳米棒合成的方法 |
| CN110526251B (zh) * | 2019-08-28 | 2023-01-24 | 贵州大学 | 一种锂电池二氧化硅负极材料的制备方法 |
| CN116106993B (zh) * | 2023-04-07 | 2023-07-18 | 武汉理工大学 | 一种磁响应性光子晶体基元的尺寸调控方法及应用 |
| CN117550651B (zh) * | 2024-01-10 | 2024-04-05 | 武汉理工大学 | 可组装磁性光子晶体的单分散纳米粒子的制备方法及应用 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102199302A (zh) * | 2010-03-22 | 2011-09-28 | 中国科学院化学研究所 | 具有各向异性结构的光子晶体薄膜的制备方法 |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4096292A (en) * | 1975-01-14 | 1978-06-20 | Montedison S.P.A. | Process for preparing ferrimagnetic acicular ferric oxide |
| US6813064B2 (en) * | 2000-07-24 | 2004-11-02 | Sajeev John | Electro-actively tunable photonic bandgap materials |
| CN1206065C (zh) * | 2003-06-27 | 2005-06-15 | 南京大学 | 高磁化强度、高稳定性、表面包覆SiO2的铁纳米颗粒的制备方法 |
| US10118834B2 (en) | 2007-04-27 | 2018-11-06 | The Regents Of The University Of California | Superparamagnetic colloidal photonic structures |
| US8409463B1 (en) * | 2007-07-16 | 2013-04-02 | University Of Central Florida Research Foundation, Inc. | Aqueous method for making magnetic iron oxide nanoparticles |
| RU2414417C2 (ru) | 2008-12-17 | 2011-03-20 | Учреждение Российской Академии Наук Институт Проблем Химической Физики Ран (Ипхф Ран) | Сверхрешетка нанокристаллов со скоррелированными кристаллографическими осями и способ ее изготовления |
| CA2753359C (en) * | 2009-02-23 | 2017-08-15 | The Regents Of The University Of California | Assembly of magnetically tunable photonic crystals in nonpolar solvents |
| ES2781572T3 (es) | 2009-04-14 | 2020-09-03 | Univ California | Procedimiento de creación de materiales coloreados fijando estructuras ordenadas de nanopartículas de magnetita dentro de un medio sólido |
| KR101332118B1 (ko) * | 2009-04-14 | 2013-11-21 | 서울대학교산학협력단 | 컬러 코드화된 자성 구조물 |
| US10013647B2 (en) | 2010-03-31 | 2018-07-03 | Quantamatrix Inc. | Method for magnetically controlling a magnetic structure |
| WO2013006207A1 (en) * | 2011-07-01 | 2013-01-10 | The Regents Of The University Of California | Direct assembly of hydrophobic nanoparticles to multifunction structures |
| WO2013112224A2 (en) * | 2011-11-09 | 2013-08-01 | The Regents Of The University Of California | Superparamagnetic colloids with enhanced charge stability for high quality magnetically tunable photonic structures |
| KR101466701B1 (ko) | 2012-03-20 | 2014-11-28 | 고려대학교 산학협력단 | 다양한 나노 구조를 갖는 헤마타이트 산화철의 제조방법 |
| CN102628188A (zh) | 2012-04-18 | 2012-08-08 | 上海应用技术学院 | 一种磁场导向下形貌各向异性的椭球状介孔二氧化硅光子晶体有序薄膜的组装方法 |
| CN103123836A (zh) | 2013-03-01 | 2013-05-29 | 东南大学 | 一种磁性各向异性胶体晶体微球及其制备方法 |
| EP3129828A4 (en) * | 2014-04-07 | 2017-11-29 | The Regents of The University of California | Highly tunable magnetic liquid crystals |
-
2016
- 2016-04-27 KR KR1020177033757A patent/KR102566859B1/ko active IP Right Grant
- 2016-04-27 CN CN201680037841.7A patent/CN107710350B/zh active Active
- 2016-04-27 US US15/569,912 patent/US10796849B2/en active Active
- 2016-04-27 JP JP2017556563A patent/JP6776267B2/ja active Active
- 2016-04-27 EP EP16787025.2A patent/EP3289596A4/en active Pending
- 2016-04-27 CN CN202010780714.4A patent/CN111899973B/zh active Active
- 2016-04-27 WO PCT/US2016/029461 patent/WO2016176267A1/en active Application Filing
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102199302A (zh) * | 2010-03-22 | 2011-09-28 | 中国科学院化学研究所 | 具有各向异性结构的光子晶体薄膜的制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US10796849B2 (en) | 2020-10-06 |
| CN111899973B (zh) | 2023-02-28 |
| JP2018525234A (ja) | 2018-09-06 |
| WO2016176267A1 (en) | 2016-11-03 |
| CN111899973A (zh) | 2020-11-06 |
| JP6776267B2 (ja) | 2020-10-28 |
| KR102566859B1 (ko) | 2023-08-11 |
| EP3289596A4 (en) | 2018-11-21 |
| EP3289596A1 (en) | 2018-03-07 |
| CN107710350A (zh) | 2018-02-16 |
| KR20180030466A (ko) | 2018-03-23 |
| US20180114637A1 (en) | 2018-04-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107710350B (zh) | 基于各向异性纳米结构的磁可调谐光子晶体 | |
| Wang et al. | Magnetic assembly and field‐tuning of ellipsoidal‐nanoparticle‐based colloidal photonic crystals | |
| US9916922B2 (en) | Iron oxide magnetic nanoparticle powder and method of producing the same, iron oxide magnetic nanoparticle thin film containing the iron oxide magnetic nanoparticle powder and method of producing the same | |
| Ding et al. | Fabrication of 3D photonic crystals of ellipsoids: convective self‐assembly in magnetic field | |
| Ge et al. | Magnetically responsive colloidal photonic crystals | |
| Nguyen et al. | Shape-and size-controlled synthesis of monoclinic ErOOH and cubic Er2O3 from micro-to nanostructures and their upconversion luminescence | |
| Dong et al. | Optical response of a disordered bicontinuous macroporous structure in the longhorn beetle Sphingnotus mirabilis | |
| CN107523139B (zh) | 一种光子晶体彩色墨水及应用 | |
| Wang et al. | Large-scale preparation of size-controlled Fe 3 O 4@ SiO 2 particles for electrophoretic display with non-iridescent structural colors | |
| Ma et al. | Photonic nanorods with magnetic responsiveness regulated by lattice defects | |
| KR101968634B1 (ko) | 고굴절률 나노 입자의 제조 방법, 이에 따라 제조된 나노 입자 및 나노 입자를 이용한 광결정 소자 | |
| Lee et al. | Synthesis and assembly of nonspherical hollow silica colloids under confinement | |
| Yang et al. | Gram-scale synthesis of superparamagnetic Fe 3 O 4 nanocrystal clusters with long-term charge stability for highly stable magnetically responsive photonic crystals | |
| Li et al. | High‐efficiency alignment of 3D biotemplated helices via rotating magnetic field for terahertz chiral metamaterials | |
| Chi et al. | Magnetically responsive colloidal crystals with angle-independent gradient structural colors in microfluidic droplet arrays | |
| CN103198913B (zh) | 银-四氧化三铁核壳纳米颗粒及其制备方法 | |
| Xiao et al. | Magnetic assembly and manipulation of Janus photonic crystal supraparticles from a colloidal mixture of spheres and ellipsoids | |
| Pan et al. | Robust synthesis of highly charged superparamagnetic Fe 3 O 4 colloidal nanocrystal clusters for magnetically responsive photonic crystals | |
| Ren et al. | Mie resonant structural colors based on ZnO spheres and their application in multi-color pattern: especially realization of red color | |
| Ahmed et al. | Facile synthesis of gold nanoworms with a tunable length and aspect ratio through oriented attachment of nanoparticles | |
| Peng et al. | Spectral selectivity of 3D magnetophotonic crystal film fabricated from single butterfly wing scales | |
| CN110655089B (zh) | 一种光学性质可调的分散液及其制备方法 | |
| CN110787743A (zh) | 一种磁响应性光子晶体、及其简单规模化制备方法与应用 | |
| Liu et al. | Facile synthesis of m-phenylenediamine-based nanospheres coated with poly (m-phenylenediamine) for building bright iridescent structural color | |
| CN115449899A (zh) | 一种磁响应光子晶体及其制备方法和观测方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |