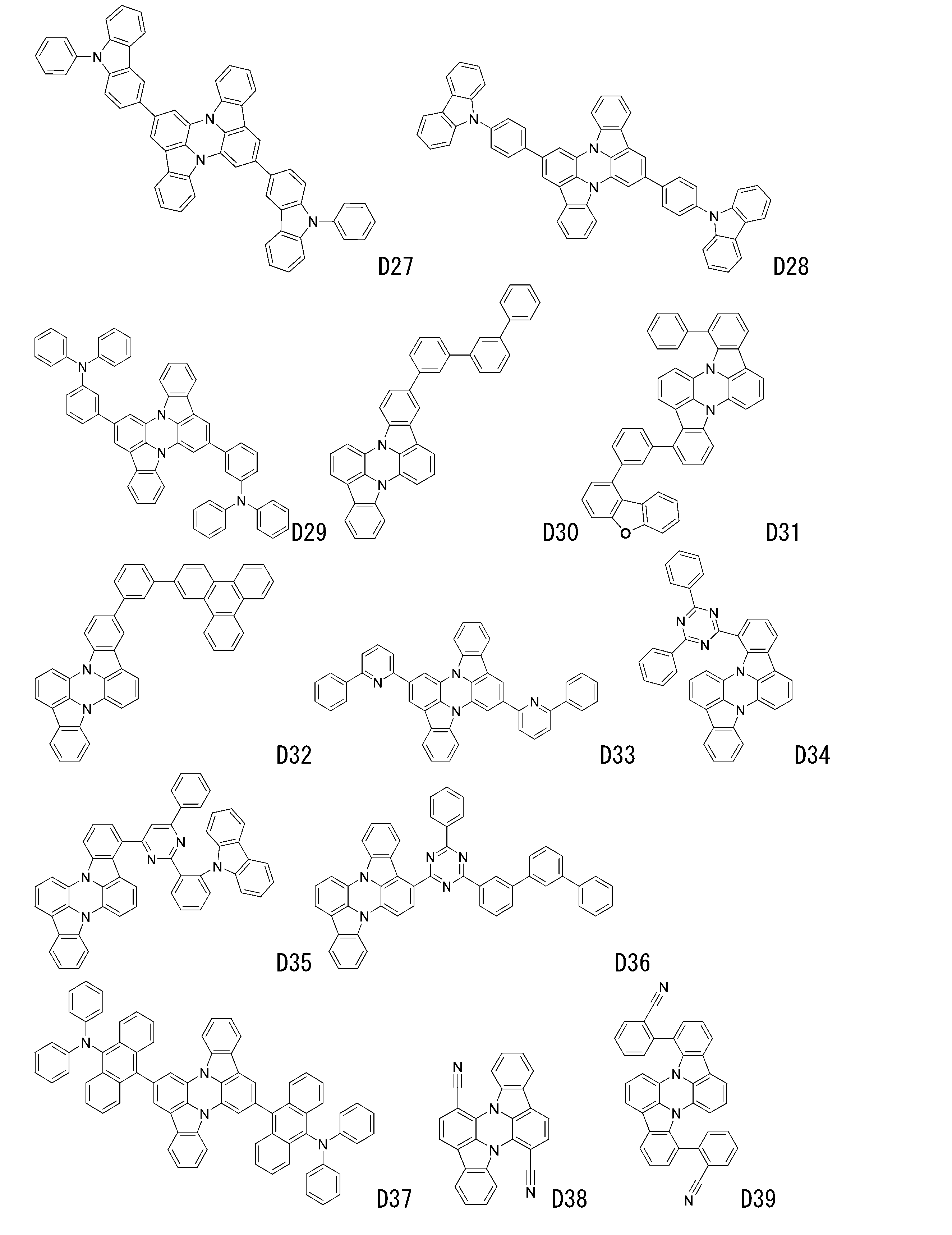WO2022215580A1 - 発光材料、及び有機電界発光素子 - Google Patents
発光材料、及び有機電界発光素子 Download PDFInfo
- Publication number
- WO2022215580A1 WO2022215580A1 PCT/JP2022/015058 JP2022015058W WO2022215580A1 WO 2022215580 A1 WO2022215580 A1 WO 2022215580A1 JP 2022015058 W JP2022015058 W JP 2022015058W WO 2022215580 A1 WO2022215580 A1 WO 2022215580A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- substituted
- unsubstituted
- group
- aromatic
- carbon atoms
- Prior art date
Links
- 239000000463 material Substances 0.000 title claims abstract description 95
- 125000003118 aryl group Chemical group 0.000 claims abstract description 133
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 20
- 125000000623 heterocyclic group Chemical group 0.000 claims abstract description 18
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 7
- 229910052760 oxygen Inorganic materials 0.000 claims abstract description 6
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 6
- 229910052711 selenium Inorganic materials 0.000 claims abstract description 4
- 125000004432 carbon atom Chemical group C* 0.000 claims description 137
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 claims description 65
- 125000006615 aromatic heterocyclic group Chemical group 0.000 claims description 52
- 125000001931 aliphatic group Chemical group 0.000 claims description 37
- 229910052739 hydrogen Inorganic materials 0.000 claims description 37
- 239000001257 hydrogen Substances 0.000 claims description 37
- 150000002431 hydrogen Chemical class 0.000 claims description 30
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 26
- 229910052805 deuterium Inorganic materials 0.000 claims description 26
- 125000004986 diarylamino group Chemical group 0.000 claims description 24
- 125000005240 diheteroarylamino group Chemical group 0.000 claims description 22
- 150000001721 carbon Chemical class 0.000 claims description 10
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 9
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 claims description 5
- 238000005401 electroluminescence Methods 0.000 claims description 3
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 claims 1
- 125000005241 heteroarylamino group Chemical group 0.000 claims 1
- 239000010410 layer Substances 0.000 description 96
- 150000001875 compounds Chemical class 0.000 description 47
- -1 polycyclic aromatic compound Chemical class 0.000 description 33
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 30
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 27
- XSCHRSMBECNVNS-UHFFFAOYSA-N quinoxaline Chemical compound N1=CC=NC2=CC=CC=C21 XSCHRSMBECNVNS-UHFFFAOYSA-N 0.000 description 27
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 24
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 22
- UJOBWOGCFQCDNV-UHFFFAOYSA-N 9H-carbazole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=C1 UJOBWOGCFQCDNV-UHFFFAOYSA-N 0.000 description 20
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical compound C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 20
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 20
- VLLMWSRANPNYQX-UHFFFAOYSA-N thiadiazole Chemical compound C1=CSN=N1.C1=CSN=N1 VLLMWSRANPNYQX-UHFFFAOYSA-N 0.000 description 19
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical compound C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 18
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical compound N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 description 18
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 18
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 18
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 18
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 18
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 18
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 18
- CUFNKYGDVFVPHO-UHFFFAOYSA-N azulene Chemical compound C1=CC=CC2=CC=CC2=C1 CUFNKYGDVFVPHO-UHFFFAOYSA-N 0.000 description 18
- 230000000903 blocking effect Effects 0.000 description 18
- TXCDCPKCNAJMEE-UHFFFAOYSA-N dibenzofuran Chemical compound C1=CC=C2C3=CC=CC=C3OC2=C1 TXCDCPKCNAJMEE-UHFFFAOYSA-N 0.000 description 18
- IYYZUPMFVPLQIF-UHFFFAOYSA-N dibenzothiophene Chemical compound C1=CC=C2C3=CC=CC=C3SC2=C1 IYYZUPMFVPLQIF-UHFFFAOYSA-N 0.000 description 18
- IQZZFVDIZRWADY-UHFFFAOYSA-N isocoumarin Chemical compound C1=CC=C2C(=O)OC=CC2=C1 IQZZFVDIZRWADY-UHFFFAOYSA-N 0.000 description 18
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 18
- 239000000203 mixture Substances 0.000 description 17
- 238000002347 injection Methods 0.000 description 16
- 239000007924 injection Substances 0.000 description 16
- YNPNZTXNASCQKK-UHFFFAOYSA-N phenanthrene Chemical compound C1=CC=C2C3=CC=CC=C3C=CC2=C1 YNPNZTXNASCQKK-UHFFFAOYSA-N 0.000 description 16
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 13
- 238000000034 method Methods 0.000 description 13
- DXBHBZVCASKNBY-UHFFFAOYSA-N 1,2-Benz(a)anthracene Chemical compound C1=CC=C2C3=CC4=CC=CC=C4C=C3C=CC2=C1 DXBHBZVCASKNBY-UHFFFAOYSA-N 0.000 description 12
- WDECIBYCCFPHNR-UHFFFAOYSA-N chrysene Chemical compound C1=CC=CC2=CC=C3C4=CC=CC=C4C=CC3=C21 WDECIBYCCFPHNR-UHFFFAOYSA-N 0.000 description 12
- BBEAQIROQSPTKN-UHFFFAOYSA-N pyrene Chemical compound C1=CC=C2C=CC3=CC=CC4=CC=C1C2=C43 BBEAQIROQSPTKN-UHFFFAOYSA-N 0.000 description 12
- 229910052751 metal Inorganic materials 0.000 description 11
- 239000002184 metal Substances 0.000 description 11
- 239000010409 thin film Substances 0.000 description 11
- 229930192474 thiophene Natural products 0.000 description 11
- 238000007740 vapor deposition Methods 0.000 description 11
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical compound C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 10
- 125000001424 substituent group Chemical group 0.000 description 10
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical compound C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 9
- CSNIZNHTOVFARY-UHFFFAOYSA-N 1,2-benzothiazole Chemical compound C1=CC=C2C=NSC2=C1 CSNIZNHTOVFARY-UHFFFAOYSA-N 0.000 description 9
- BAXOFTOLAUCFNW-UHFFFAOYSA-N 1H-indazole Chemical compound C1=CC=C2C=NNC2=C1 BAXOFTOLAUCFNW-UHFFFAOYSA-N 0.000 description 9
- ZPSJGADGUYYRKE-UHFFFAOYSA-N 2H-pyran-2-one Chemical compound O=C1C=CC=CO1 ZPSJGADGUYYRKE-UHFFFAOYSA-N 0.000 description 9
- GOLORTLGFDVFDW-UHFFFAOYSA-N 3-(1h-benzimidazol-2-yl)-7-(diethylamino)chromen-2-one Chemical compound C1=CC=C2NC(C3=CC4=CC=C(C=C4OC3=O)N(CC)CC)=NC2=C1 GOLORTLGFDVFDW-UHFFFAOYSA-N 0.000 description 9
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 9
- VOLMSPGWNYJHQQ-UHFFFAOYSA-N Pyranone Natural products CC1=C(O)C(=O)C(O)CO1 VOLMSPGWNYJHQQ-UHFFFAOYSA-N 0.000 description 9
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 9
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 9
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 9
- RFRXIWQYSOIBDI-UHFFFAOYSA-N benzarone Chemical compound CCC=1OC2=CC=CC=C2C=1C(=O)C1=CC=C(O)C=C1 RFRXIWQYSOIBDI-UHFFFAOYSA-N 0.000 description 9
- QRUDEWIWKLJBPS-UHFFFAOYSA-N benzotriazole Chemical compound C1=CC=C2N[N][N]C2=C1 QRUDEWIWKLJBPS-UHFFFAOYSA-N 0.000 description 9
- 239000012964 benzotriazole Substances 0.000 description 9
- OTAFHZMPRISVEM-UHFFFAOYSA-N chromone Chemical compound C1=CC=C2C(=O)C=COC2=C1 OTAFHZMPRISVEM-UHFFFAOYSA-N 0.000 description 9
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 9
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 9
- ZLTPDFXIESTBQG-UHFFFAOYSA-N isothiazole Chemical compound C=1C=NSC=1 ZLTPDFXIESTBQG-UHFFFAOYSA-N 0.000 description 9
- CTAPFRYPJLPFDF-UHFFFAOYSA-N isoxazole Chemical compound C=1C=NOC=1 CTAPFRYPJLPFDF-UHFFFAOYSA-N 0.000 description 9
- LFSXCDWNBUNEEM-UHFFFAOYSA-N phthalazine Chemical compound C1=NN=CC2=CC=CC=C21 LFSXCDWNBUNEEM-UHFFFAOYSA-N 0.000 description 9
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical compound C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 9
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 9
- JWVCLYRUEFBMGU-UHFFFAOYSA-N quinazoline Chemical compound N1=CN=CC2=CC=CC=C21 JWVCLYRUEFBMGU-UHFFFAOYSA-N 0.000 description 9
- 239000000243 solution Substances 0.000 description 9
- 150000003536 tetrazoles Chemical class 0.000 description 9
- 150000003852 triazoles Chemical class 0.000 description 9
- FNQJDLTXOVEEFB-UHFFFAOYSA-N 1,2,3-benzothiadiazole Chemical compound C1=CC=C2SN=NC2=C1 FNQJDLTXOVEEFB-UHFFFAOYSA-N 0.000 description 8
- CYSGHNMQYZDMIA-UHFFFAOYSA-N 1,3-Dimethyl-2-imidazolidinon Chemical compound CN1CCN(C)C1=O CYSGHNMQYZDMIA-UHFFFAOYSA-N 0.000 description 8
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 8
- 239000005964 Acibenzolar-S-methyl Substances 0.000 description 8
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 8
- 229910052782 aluminium Inorganic materials 0.000 description 8
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 8
- 239000010408 film Substances 0.000 description 8
- 239000011777 magnesium Substances 0.000 description 8
- 229910052749 magnesium Inorganic materials 0.000 description 8
- 239000000758 substrate Substances 0.000 description 8
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 7
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- HXGDTGSAIMULJN-UHFFFAOYSA-N acetnaphthylene Natural products C1=CC(C=C2)=C3C2=CC=CC3=C1 HXGDTGSAIMULJN-UHFFFAOYSA-N 0.000 description 6
- 230000003111 delayed effect Effects 0.000 description 6
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthrene Natural products C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 6
- RMBPEFMHABBEKP-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2C3=C[CH]C=CC3=CC2=C1 RMBPEFMHABBEKP-UHFFFAOYSA-N 0.000 description 6
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N o-biphenylenemethane Natural products C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 5
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 5
- DHFABSXGNHDNCO-UHFFFAOYSA-N dibenzoselenophene Chemical compound C1=CC=C2C3=CC=CC=C3[se]C2=C1 DHFABSXGNHDNCO-UHFFFAOYSA-N 0.000 description 5
- 239000002019 doping agent Substances 0.000 description 5
- 230000005525 hole transport Effects 0.000 description 5
- VVVPGLRKXQSQSZ-UHFFFAOYSA-N indolo[3,2-c]carbazole Chemical compound C1=CC=CC2=NC3=C4C5=CC=CC=C5N=C4C=CC3=C21 VVVPGLRKXQSQSZ-UHFFFAOYSA-N 0.000 description 5
- 230000007246 mechanism Effects 0.000 description 5
- 125000005580 triphenylene group Chemical group 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical compound C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 239000007772 electrode material Substances 0.000 description 4
- 238000000295 emission spectrum Methods 0.000 description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 4
- 239000012299 nitrogen atmosphere Substances 0.000 description 4
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 239000002244 precipitate Substances 0.000 description 4
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 238000006862 quantum yield reaction Methods 0.000 description 4
- MCJGNVYPOGVAJF-UHFFFAOYSA-N quinolin-8-ol Chemical class C1=CN=C2C(O)=CC=CC2=C1 MCJGNVYPOGVAJF-UHFFFAOYSA-N 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- 238000005215 recombination Methods 0.000 description 4
- 230000006798 recombination Effects 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- 238000001269 time-of-flight mass spectrometry Methods 0.000 description 4
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 4
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 3
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 3
- SLGBZMMZGDRARJ-UHFFFAOYSA-N Triphenylene Natural products C1=CC=C2C3=CC=CC=C3C3=CC=CC=C3C2=C1 SLGBZMMZGDRARJ-UHFFFAOYSA-N 0.000 description 3
- 150000004982 aromatic amines Chemical class 0.000 description 3
- 150000001716 carbazoles Chemical class 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 229910052802 copper Inorganic materials 0.000 description 3
- 239000010949 copper Substances 0.000 description 3
- 230000005284 excitation Effects 0.000 description 3
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 3
- 229910052738 indium Inorganic materials 0.000 description 3
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 3
- 229910052744 lithium Inorganic materials 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 150000004866 oxadiazoles Chemical class 0.000 description 3
- 238000005424 photoluminescence Methods 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 239000010453 quartz Substances 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- 238000004544 sputter deposition Methods 0.000 description 3
- RMVRSNDYEFQCLF-UHFFFAOYSA-N thiophenol Chemical compound SC1=CC=CC=C1 RMVRSNDYEFQCLF-UHFFFAOYSA-N 0.000 description 3
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N N-phenyl amine Natural products NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- 229910045601 alloy Inorganic materials 0.000 description 2
- 239000000956 alloy Substances 0.000 description 2
- 150000001454 anthracenes Chemical class 0.000 description 2
- 125000005110 aryl thio group Chemical group 0.000 description 2
- 125000004104 aryloxy group Chemical group 0.000 description 2
- 239000010406 cathode material Substances 0.000 description 2
- 238000010549 co-Evaporation Methods 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 150000008376 fluorenones Chemical class 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 125000001072 heteroaryl group Chemical group 0.000 description 2
- 150000002460 imidazoles Chemical class 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 239000012044 organic layer Substances 0.000 description 2
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 2
- 229960003540 oxyquinoline Drugs 0.000 description 2
- 125000001791 phenazinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3N=C12)* 0.000 description 2
- 150000004986 phenylenediamines Chemical class 0.000 description 2
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical class N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 2
- 150000004033 porphyrin derivatives Chemical class 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 150000003222 pyridines Chemical class 0.000 description 2
- 150000003230 pyrimidines Chemical class 0.000 description 2
- 238000001226 reprecipitation Methods 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 239000004332 silver Substances 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical class C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 description 2
- 239000012780 transparent material Substances 0.000 description 2
- 150000003918 triazines Chemical class 0.000 description 2
- 229910000404 tripotassium phosphate Inorganic materials 0.000 description 2
- 235000019798 tripotassium phosphate Nutrition 0.000 description 2
- 238000001771 vacuum deposition Methods 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- UWRZIZXBOLBCON-VOTSOKGWSA-N (e)-2-phenylethenamine Chemical class N\C=C\C1=CC=CC=C1 UWRZIZXBOLBCON-VOTSOKGWSA-N 0.000 description 1
- VERMWGQSKPXSPZ-BUHFOSPRSA-N 1-[(e)-2-phenylethenyl]anthracene Chemical compound C=1C=CC2=CC3=CC=CC=C3C=C2C=1\C=C\C1=CC=CC=C1 VERMWGQSKPXSPZ-BUHFOSPRSA-N 0.000 description 1
- MVWPVABZQQJTPL-UHFFFAOYSA-N 2,3-diphenylcyclohexa-2,5-diene-1,4-dione Chemical class O=C1C=CC(=O)C(C=2C=CC=CC=2)=C1C1=CC=CC=C1 MVWPVABZQQJTPL-UHFFFAOYSA-N 0.000 description 1
- ITNMDRWLTGIOCF-UHFFFAOYSA-N 2-ethoxy-3-methoxyphenol Chemical compound CCOC1=C(O)C=CC=C1OC ITNMDRWLTGIOCF-UHFFFAOYSA-N 0.000 description 1
- ZYASLTYCYTYKFC-UHFFFAOYSA-N 9-methylidenefluorene Chemical class C1=CC=C2C(=C)C3=CC=CC=C3C2=C1 ZYASLTYCYTYKFC-UHFFFAOYSA-N 0.000 description 1
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical class N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 description 1
- HAKMCEOODDDIRS-UHFFFAOYSA-N CCSC(C(SC)=C(C(OC1=CC=CC=C1)=C1)OC2=CC=CC=C2)=C1S Chemical compound CCSC(C(SC)=C(C(OC1=CC=CC=C1)=C1)OC2=CC=CC=C2)=C1S HAKMCEOODDDIRS-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 229910000799 K alloy Inorganic materials 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- 229920000265 Polyparaphenylene Chemical class 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 239000010405 anode material Substances 0.000 description 1
- 150000008425 anthrones Chemical class 0.000 description 1
- 229940027991 antiseptic and disinfectant quinoline derivative Drugs 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 150000001556 benzimidazoles Chemical class 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 150000001718 carbodiimides Chemical class 0.000 description 1
- PJVZQNVOUCOJGE-CALCHBBNSA-N chembl289853 Chemical compound N1([C@H]2CC[C@H](O2)N2[C]3C=CC=CC3=C3C2=C11)C2=CC=C[CH]C2=C1C1=C3C(=O)N(C)C1=O PJVZQNVOUCOJGE-CALCHBBNSA-N 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 229920001940 conductive polymer Polymers 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- ZBJBRUSGEJORQL-UHFFFAOYSA-N diphenan Chemical compound C1=CC(OC(=O)N)=CC=C1CC1=CC=CC=C1 ZBJBRUSGEJORQL-UHFFFAOYSA-N 0.000 description 1
- 229950002048 diphenan Drugs 0.000 description 1
- LTYMSROWYAPPGB-UHFFFAOYSA-N diphenyl sulfide Chemical group C=1C=CC=CC=1SC1=CC=CC=C1 LTYMSROWYAPPGB-UHFFFAOYSA-N 0.000 description 1
- DKHNGUNXLDCATP-UHFFFAOYSA-N dipyrazino[2,3-f:2',3'-h]quinoxaline-2,3,6,7,10,11-hexacarbonitrile Chemical compound C12=NC(C#N)=C(C#N)N=C2C2=NC(C#N)=C(C#N)N=C2C2=C1N=C(C#N)C(C#N)=N2 DKHNGUNXLDCATP-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 150000007857 hydrazones Chemical class 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 1
- 150000002475 indoles Chemical class 0.000 description 1
- 229960005544 indolocarbazole Drugs 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 229940079865 intestinal antiinfectives imidazole derivative Drugs 0.000 description 1
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 238000009740 moulding (composite fabrication) Methods 0.000 description 1
- KKFHAJHLJHVUDM-UHFFFAOYSA-N n-vinylcarbazole Chemical class C1=CC=C2N(C=C)C3=CC=CC=C3C2=C1 KKFHAJHLJHVUDM-UHFFFAOYSA-N 0.000 description 1
- 238000006902 nitrogenation reaction Methods 0.000 description 1
- 150000007978 oxazole derivatives Chemical class 0.000 description 1
- 150000002916 oxazoles Chemical class 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- MPQXHAGKBWFSNV-UHFFFAOYSA-N oxidophosphanium Chemical class [PH3]=O MPQXHAGKBWFSNV-UHFFFAOYSA-N 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 230000001443 photoexcitation Effects 0.000 description 1
- 238000000206 photolithography Methods 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920003227 poly(N-vinyl carbazole) Polymers 0.000 description 1
- 229920000553 poly(phenylenevinylene) Chemical class 0.000 description 1
- 229920002098 polyfluorene Chemical class 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920000123 polythiophene Polymers 0.000 description 1
- BITYAPCSNKJESK-UHFFFAOYSA-N potassiosodium Chemical compound [Na].[K] BITYAPCSNKJESK-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000010298 pulverizing process Methods 0.000 description 1
- 150000003248 quinolines Chemical class 0.000 description 1
- 229910052761 rare earth metal Inorganic materials 0.000 description 1
- 150000002910 rare earth metals Chemical class 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 238000000859 sublimation Methods 0.000 description 1
- 230000008022 sublimation Effects 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229940042055 systemic antimycotics triazole derivative Drugs 0.000 description 1
- IBBLKSWSCDAPIF-UHFFFAOYSA-N thiopyran Chemical compound S1C=CC=C=C1 IBBLKSWSCDAPIF-UHFFFAOYSA-N 0.000 description 1
- 238000002834 transmittance Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains three hetero rings
- C07D487/16—Peri-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/22—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains four or more hetero rings
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1059—Heterocyclic compounds characterised by ligands containing three nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1074—Heterocyclic compounds characterised by ligands containing more than three nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1074—Heterocyclic compounds characterised by ligands containing more than three nitrogen atoms as heteroatoms
- C09K2211/1077—Heterocyclic compounds characterised by ligands containing more than three nitrogen atoms as heteroatoms with oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1074—Heterocyclic compounds characterised by ligands containing more than three nitrogen atoms as heteroatoms
- C09K2211/1081—Heterocyclic compounds characterised by ligands containing more than three nitrogen atoms as heteroatoms with sulfur
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
Definitions
- the present invention relates to a luminescent material and an organic electroluminescence device (referred to as an organic EL device) using the luminescent material in the luminescent layer.
- Patent Literature 1 discloses an organic EL element that utilizes a TTF (Triplet-Triplet Fusion) mechanism, which is one mechanism of delayed fluorescence.
- TTF Triplet-Triplet Fusion
- the TTF mechanism utilizes a phenomenon in which singlet excitons are generated by the collision of two triplet excitons, and is theoretically believed to increase the internal quantum efficiency to 40%.
- the efficiency is lower than that of phosphorescent organic EL devices, there is a demand for further improvement in efficiency.
- Patent Document 2 discloses an organic EL element using a thermally activated delayed fluorescence (TADF) mechanism.
- the TADF mechanism utilizes the phenomenon of reverse intersystem crossing from triplet excitons to singlet excitons in materials with a small energy difference between the singlet and triplet levels. It is believed that it can be increased to 100%.
- Patent Literature 2 discloses a thermally activated delayed fluorescence material comprising an indolocarbazole compound.
- Patent Documents 3, 4, and 5 disclose a material composed of a polycyclic aromatic compound containing an indolophenadine skeleton and an organic EL device using the material.
- an organic EL device using a material composed of a polycyclic aromatic compound containing an indolofenadine skeleton as a light-emitting material.
- Non-Patent Document 1 discloses a material composed of a polycyclic aromatic compound containing a carbazole skeleton, which is a partial skeleton of an indolofenadine skeleton, and a blue organic EL device using the material as a light-emitting material.
- a material composed of a polycyclic aromatic compound containing a carbazole skeleton which is a partial skeleton of an indolofenadine skeleton
- a blue organic EL device using the material as a light-emitting material.
- it does not have a luminous efficiency that can withstand practical use.
- the present invention has been made in view of such circumstances, and provides a light-emitting material capable of obtaining a practically useful organic EL device that emits light with high efficiency and has high driving stability, and the same.
- An object of the present invention is to provide an organic EL device using the method.
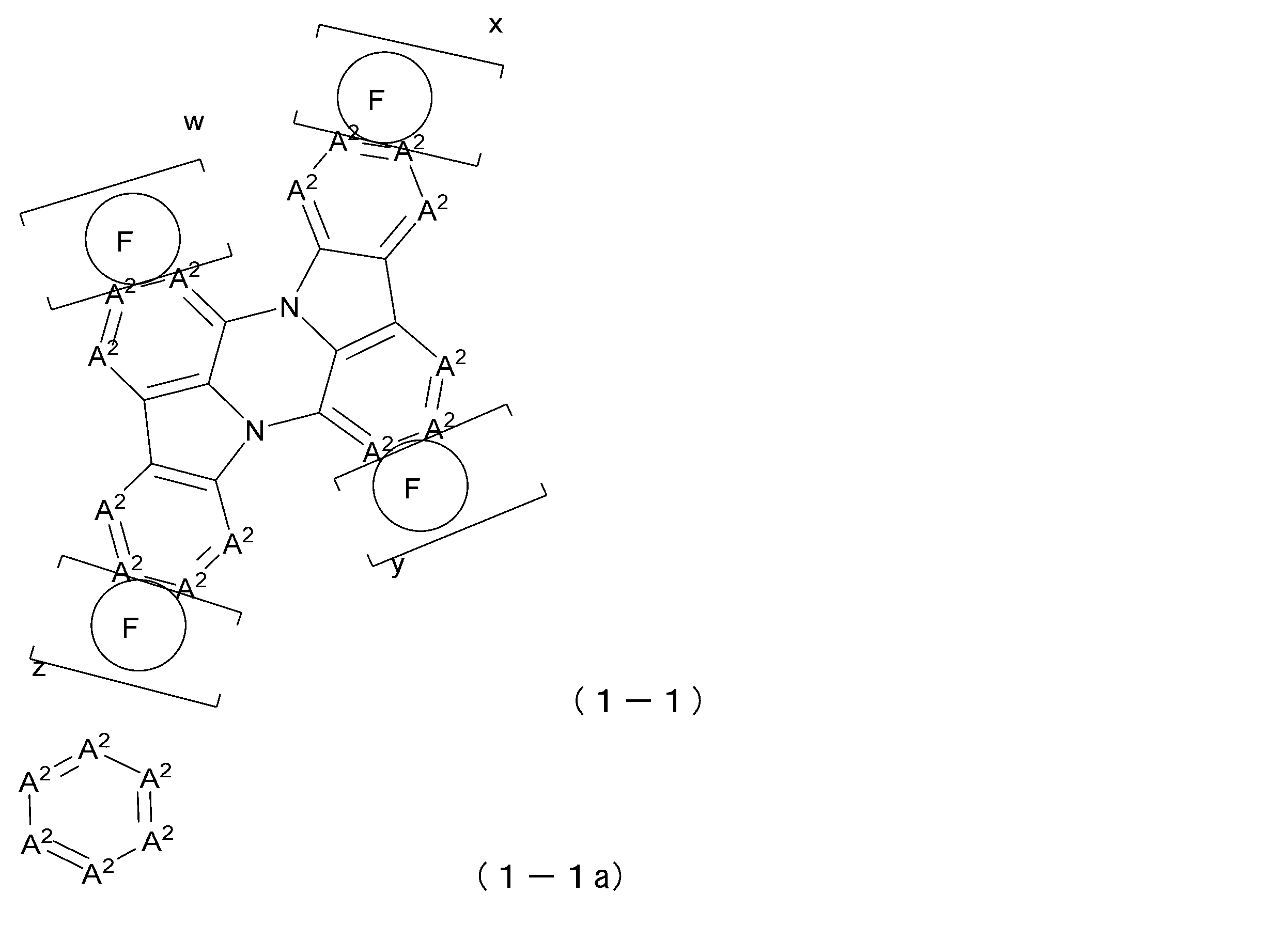
- the present invention is a luminescent material represented by the following general formula (1) or (1-1).
- each A 1 is independently CR 1 , C or N. However, the number of N present in one six-membered ring containing A 1 in general formula (1) is 2 or less.
- Each R 1 is independently hydrogen, a cyano group, deuterium, a substituted or unsubstituted diarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted arylheteroarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted diheteroarylamino group having 12 to 44 carbon atoms, aliphatic hydrocarbon group having 1 to 10 carbon atoms, substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, substituted or unsubstituted carbon 3 to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- R 1 When R 1 is a substituted or unsubstituted diarylamino group, arylheteroarylamino group, diheteroarylamino group, aromatic hydrocarbon group, or substituted or unsubstituted aromatic heterocyclic group, R 1 has the formula directly with the aromatic ring having A 1 to which R 1 is attached in (1), or —O—, —S—, —Si(R a ) 2 —, —C(R a ) 2 —, or —NR One or more bonds may be formed through a— to form a condensed ring.
- Each R a is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- Each ring E is independently a heterocyclic ring represented by formula (1a), and the ring E is fused with the adjacent ring at any position.
- Each X 1 is independently a group represented by Si(R d ) 2 , C(R d ) 2 , O, S, Se, or NL 1 -(Ar 1 ) e .
- Each R d is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- Each L 1 is independently a substituted or unsubstituted C 6-30 aromatic hydrocarbon group or a substituted or unsubstituted C 3-30 aromatic heterocyclic group
- each Ar 1 is independently , a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 30 carbon atoms, or 2 to 9 of these aromatic rings are linked is a substituted or unsubstituted linked aromatic group consisting of L 1 is directly connected to the aromatic ring adjacent to the heterocyclic ring to which L 1 is attached, or —O—, —S—, —Si(R c ) 2 —, —C(R c ) 2 —, or —NR
- One bond may be formed through c- to form a condensed ring.
- Each R c is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- At least one R 1 or L 1 is directly with the aromatic having A 1 to which R 1 is attached in formula (1) or the aromatic ring to which L 1 is adjacent, or —O—, —S—, —Si One or more bonds may be formed via (R a ) 2 —, —C(R a ) 2 —, or —NR a — to form a condensed ring. Also, at least one L1 may be directly bonded to an adjacent aromatic ring to form a condensed ring.
- At least one R 1 is a substituted or unsubstituted carbazolyl group, and the aromatic ring to which the carbazolyl group is bonded directly or —O—, —S—, —Si(R a ) 2 —, —C(R a ) 2- or -NR a - may form one or more bonds to form a condensed ring.
- Each A 2 is independently CR 2 , C or N; However, the number of N present in one six-membered ring containing A 2 in general formula (1-1) is 2 or less.
- Each R 2 is independently hydrogen, a cyano group, deuterium, a substituted or unsubstituted diarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted arylheteroarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted diheteroarylamino group having 12 to 44 carbon atoms, aliphatic hydrocarbon group having 1 to 10 carbon atoms, substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, substituted or unsubstituted carbon 3 to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- R 2 When R 2 is a substituted or unsubstituted diarylamino group, arylheteroarylamino group, diheteroarylamino group, aromatic hydrocarbon group, or substituted or unsubstituted aromatic heterocyclic group, R 2 has the formula directly with the aromatic ring having A 2 to which R 2 is attached in (1-1), or —O—, —S—, —Si(R b ) 2 —, —C(R b ) 2 —, or One or more bonds may be formed through —NR b — to form a condensed ring.
- Each R b is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- Each ring F is independently an aromatic ring represented by formula (1-1a), and the ring F is fused with an adjacent ring at any position.
- x, y, w, and z each independently represent 0 or 1;
- a+b+c+d it is preferable that a+b+c+d ⁇ 2. Also, b and c may be 1, or a and d may be 1.
- x+y+z+w it is preferable that x+y+z+w ⁇ 2. Also, x and z may be 1, or y and w may be 1.
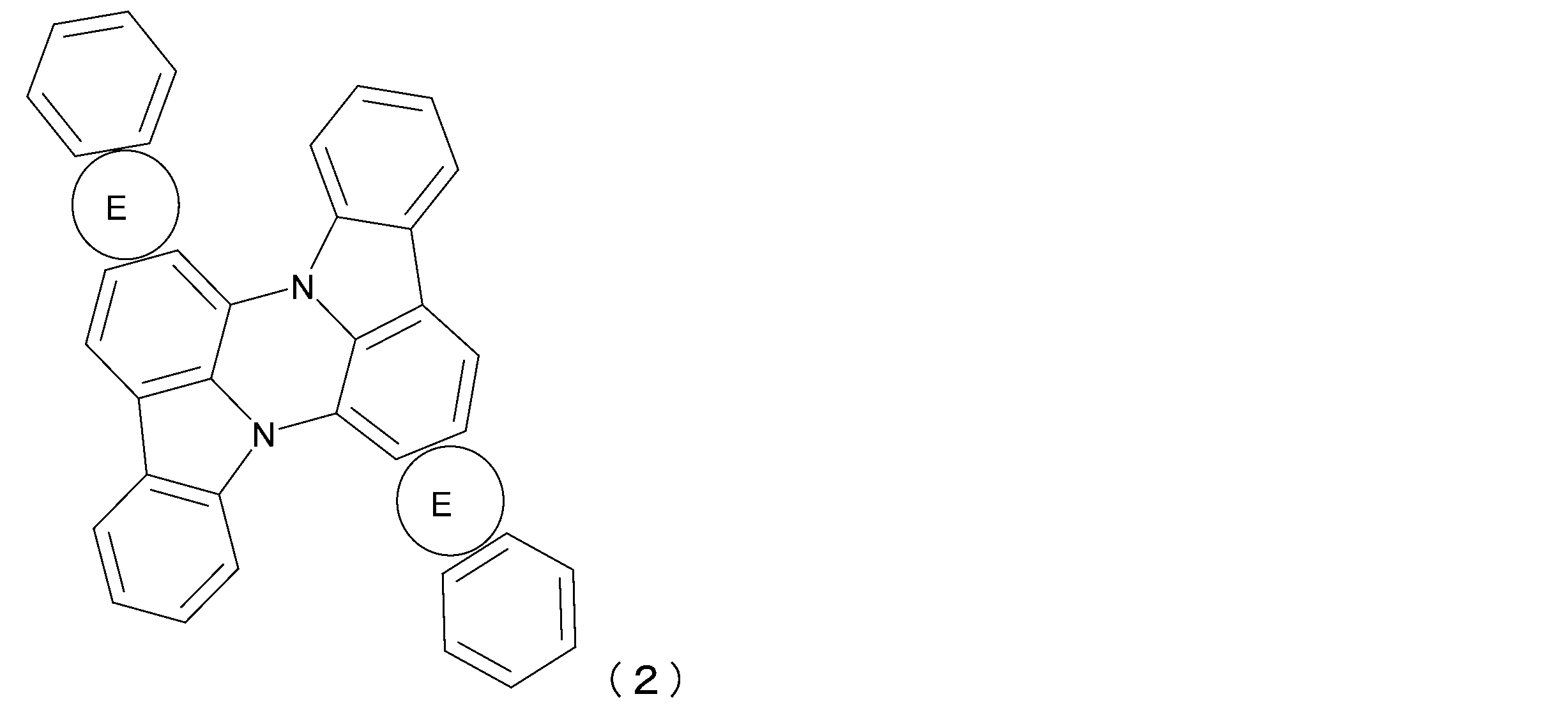
- the luminescent material represented by the general formula (1) can be a luminescent material represented by the following general formula (2) or (3).
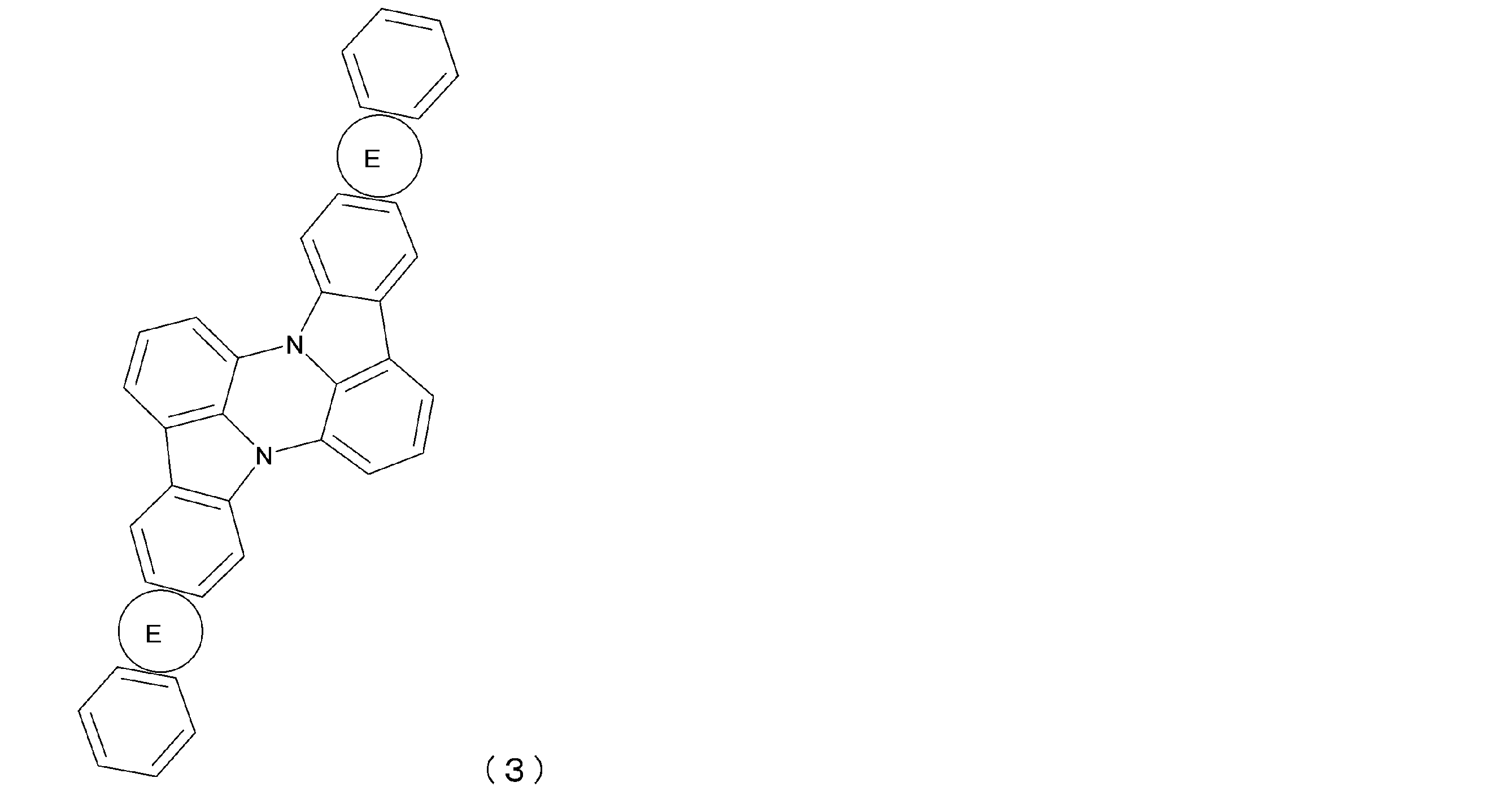
- ring E is synonymous with general formula (1).
- each of the above rings E is independently a group represented by NL 1 -(Ar 1 ) e for X 1 in formula (1a).
- the light-emitting material represented by the general formula (1) or general formula (1-1) can be a light-emitting material represented by the following general formula (4).
- each A 1 is independently CR 1 or N.
- R 1 is the same as R 1 in general formula (1).
- the number of N present in one six-membered ring containing A 1 in general formula (4) is 2 or less.
- the light emitting material represented by the general formula (1) or general formula (1-1) has a difference ( ⁇ EST) between the excited singlet energy (S1) and the excited triplet energy (T1) of 0.40 eV or less. is desirable.
- the present invention also provides an organic electroluminescence device comprising one or more light-emitting layers between an anode and a cathode facing each other, wherein at least one light-emitting layer contains the above-described light-emitting material. is.
- the luminescent material of the present invention it is possible to obtain a practically useful organic EL device that emits light with high efficiency and has high driving stability.
- the luminescent material of the invention also exhibits a maximum wavelength in the blue, light blue or green spectral region. This luminescent material exhibits a wavelength maximum in particular between 410 nm and 550 nm, preferably between 430 nm and 495 nm.
- the photoluminescence quantum yield of the luminescent material of the present invention can be 40% or more. Use of the luminescent materials of the present invention results in more efficient devices. Further, an organic EL device having a light-emitting layer containing this has high luminous efficiency and color.
- FIG. 1 is a schematic cross-sectional view showing a structural example of an organic EL device used in the present invention.
- the luminescent material of the present invention is represented by the general formula (1) or general formula (1-1).
- it is a luminescent material represented by the general formula (2), general formula (3), or general formula (4).
- the organic EL device of the present invention has one or more light-emitting layers between an anode and a cathode facing each other, and at least one of the light-emitting layers is represented by general formula (1) or general formula (1-1). It contains the represented compound as a light-emitting material.
- This organic EL device has a plurality of layers between an anode and a cathode facing each other. At least one of the layers is a light-emitting layer, and the light-emitting layer can contain a host material if necessary.
- General formula (1) will be described below.
- the compound represented by general formula (1) typically has a structure in which a plurality of indole rings are condensed to a phenazine ring or a structure similar thereto.
- a 1 is CR 1 , N, or a carbon atom.
- the number of N in A 1 present in one six-membered ring containing A 1 in general formula (1) is 2 or less.
- This six-membered ring containing A 1 may be fused with the adjacent ring E, in which case two of A 1 are carbon atoms, which are shared with ring E.
- Each R 1 is independently hydrogen, a cyano group, deuterium, a substituted or unsubstituted diarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted arylheteroarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted diheteroarylamino group having 12 to 44 carbon atoms, aliphatic hydrocarbon group having 1 to 10 carbon atoms, substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, substituted or unsubstituted carbon 3 to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- R 1 represents an unsubstituted diarylamino group, an unsubstituted arylheteroarylamino group, an unsubstituted diheteroarylamino group, or an aliphatic hydrocarbon group
- specific examples include diphenylamino, dibiphenylamino, Phenylbiphenylamino, naphthylphenylamino, dinaphthylamino, dianthranylamino, diphenanthrenylamino, dibenzofuranylphenylamino, dibenzofuranylbiphenylamino, bisdibenzofuranylamino, methyl, ethyl, propyl, butyl, pentyl , hexyl, heptyl, octyl, nonyl and decyl.
- R 1 being an unsubstituted aromatic hydrocarbon group, an aromatic heterocyclic group, or a linked aromatic group
- R 1 being an unsubstituted aromatic hydrocarbon group, an aromatic heterocyclic group, or a linked aromatic group
- benzene naphthalene, acenaphthene, acenaphthylene, azulene, anthracene, chrysene, pyrene, phenanthrene, triphenylene, fluorene, benzo[a]anthracene, pyridine, pyrimidine, triazine, thiophene, isothiazole, thiazole, pyridazine, pyrrole, pyrazole, imidazole, triazole, thiadiazole, pyrazine, furan, isoxazole, quinoline, isoquinoline, quinoxaline, quinazoline, thiadiazole , phthalazine, t
- aromatic hydrocarbon group the aromatic heterocyclic group, or the linked aromatic group may each have a substituent.
- the substituent is a cyano group, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a diarylamino group having 12 to 30 carbon atoms, an arylheteroarylamino group having 12 to 30 carbon atoms, and 12 carbon atoms. diheteroarylamino groups of up to 30, alkoxy groups of 1 to 10 carbon atoms, aryloxy groups of 6 to 18 carbon atoms, alkylthio groups of 1 to 10 carbon atoms, and arylthio groups of 6 to 18 carbon atoms.
- the substituent when it is an aliphatic hydrocarbon group having 1 to 10 carbon atoms, it may be linear, branched or cyclic.
- the diarylamino group, arylheteroarylamino group, diheteroarylamino group, aryloxy group, and arylthio group are the aromatic hydrocarbon group, the aromatic heterocyclic group, the aromatic ring of the linked aromatic group, Alternatively, when substituting an aryl group or heteroaryl group contained in a diarylamino group, an arylheteroarylamino group, or a diheteroarylamino group, nitrogen and carbon, oxygen and carbon, or sulfur and carbon are bonded with a single bond.
- the number of substituents is 0-5, preferably 0-2.
- the number of carbon atoms in the substituent is not included in the calculation of the number of carbon atoms. However, it is preferable that the total carbon number including the carbon number of the substituent satisfies the above range.
- substituents include cyano, methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, diphenylamino, naphthylphenylamino, dinaphthylamino, dianthranylamino, and diphenane.
- Threnylamino dibenzofuranylphenylamino, dibenzofuranylbiphenylamino, bisdibenzofuranylamino, methoxy, ethoxy, phenol, diphenyloxy, methylthio, ethylthio, thiophenol, or diphenylthio.
- cyano methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl, diphenylamino, naphthylphenylamino, dinaphthylamino, dibenzofuranylphenylamino, dibenzofuranylbiphenylamino, bisdibenzofuranylamino , phenol, or thiophenol.
- a linked aromatic group refers to an aromatic group in which the carbon atoms of the aromatic rings of the aromatic group are linked by single bonds.
- An aromatic group is an aromatic group in which two or more are linked, and these may be linear or branched.
- the aromatic group may be an aromatic hydrocarbon group or an aromatic heterocyclic group, and plural aromatic groups may be the same or different.
- Aromatic groups that correspond to linking aromatic groups are different from substituted aromatic groups.
- hydrogen may be deuterium. That is, in the general formulas (1) to (4), (1-1), etc., part or all of H in a skeleton such as carbazole and a substituent such as R 1 and Ar 1 is deuterium. good too.
- Each ring E is independently a heterocyclic ring represented by formula (1a), and ring E is fused to an adjacent ring at any position.
- each X 1 is independently a divalent group represented by Si(R d ) 2 , C(R d ) 2 , O, S, Se, or NL 1 -(Ar 1 ) e ; O, S, or NL 1 -(Ar 1 ) e is preferred, and NL 1 -(Ar 1 ) e is more preferred.
- Each R d is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- an aliphatic hydrocarbon group having 1 to 4 carbon atoms More preferably, hydrogen, an aliphatic hydrocarbon group having 1 to 4 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 10 carbon atoms, a substituted or unsubstituted aromatic heterocyclic ring having 2 to 12 carbon atoms or a substituted or unsubstituted linked aromatic group formed by linking 2 to 4 of these aromatic groups.
- R d is an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 30 carbon atoms, or Specific examples of substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked are the same as in the case of R 1 .
- Each L 1 independently represents a substituted or unsubstituted C 6-30 aromatic hydrocarbon group or a substituted or unsubstituted C 3-30 aromatic heterocyclic group. Preferably, it represents a substituted or unsubstituted C6-C20 aromatic hydrocarbon group or a substituted or unsubstituted C2-C20 aromatic heterocyclic group. More preferably, it represents a substituted or unsubstituted C6-C10 aromatic hydrocarbon group or a substituted or unsubstituted C2-C12 aromatic heterocyclic group.
- unsubstituted L 1 examples include benzene, naphthalene, acenaphthene, acenaphthylene, azulene, anthracene, chrysene, pyrene, phenanthrene, triphenylene, fluorene, benzo[a]anthracene, pyridine, pyrimidine, triazine, thiophene, isothiazole, Thiazole, pyridazine, pyrrole, pyrazole, imidazole, triazole, thiadiazole, pyrazine, furan, isoxazole, quinoline, isoquinoline, quinoxaline, quinazoline, thiadiazole, phthalazine, tetrazole, indole, benzofuran, benzothiophene, benzoxazole, benzothiazole, indazole, benz A group obtained by removing 1+e
- Ar 1 is a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 30 carbon atoms, or the aromatic hydrocarbon group and the aromatic heterocyclic group. It represents a substituted or unsubstituted linked aromatic group composed of linked aromatic rings of 2 to 9 aromatic groups selected from cyclic groups.
- a substituted or unsubstituted aromatic hydrocarbon group having 6 to 20 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 20 carbon atoms, or the aromatic hydrocarbon group and the aromatic heterocyclic ring represents a substituted or unsubstituted linked aromatic group formed by connecting 2 to 6 aromatic rings of an aromatic group selected from groups.
- a substituted or unsubstituted aromatic hydrocarbon group having 6 to 10 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 12 carbon atoms, or the aromatic hydrocarbon group and the aromatic heterocyclic group It represents a substituted or unsubstituted linked aromatic group formed by linking 2 to 4 aromatic rings of an aromatic group selected from cyclic groups.
- aromatic hydrocarbon group, aromatic heterocyclic group and linked aromatic group are also referred to as aromatic groups.
- unsubstituted aromatic groups include benzene, naphthalene, acenaphthene, acenaphthylene, azulene, anthracene, chrysene, pyrene, phenanthrene, triphenylene, fluorene, benzo[a]anthracene, pyridine, pyrimidine, triazine, thiophene, iso Thiazole, thiazole, pyridazine, pyrrole, pyrazole, imidazole, triazole, thiadiazole, pyrazine, furan, isoxazole, quinoline, isoquinoline, quinoxaline, quinazoline, thiadiazole, phthalazine, tetrazole, indole, benzofuran, benzothiophene, benzoxazole, benzothiazole, indazole , benzimidazole, benzo
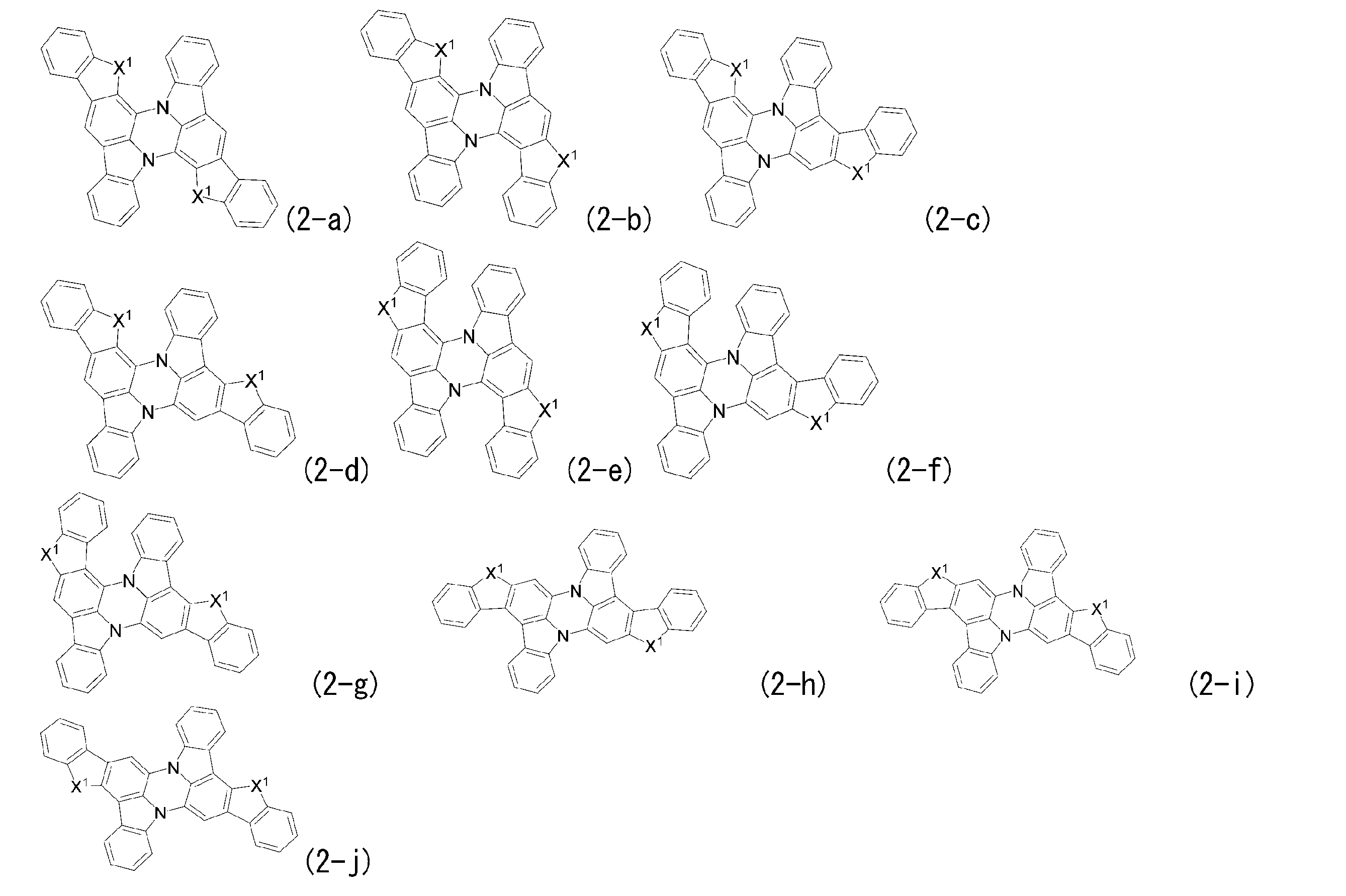
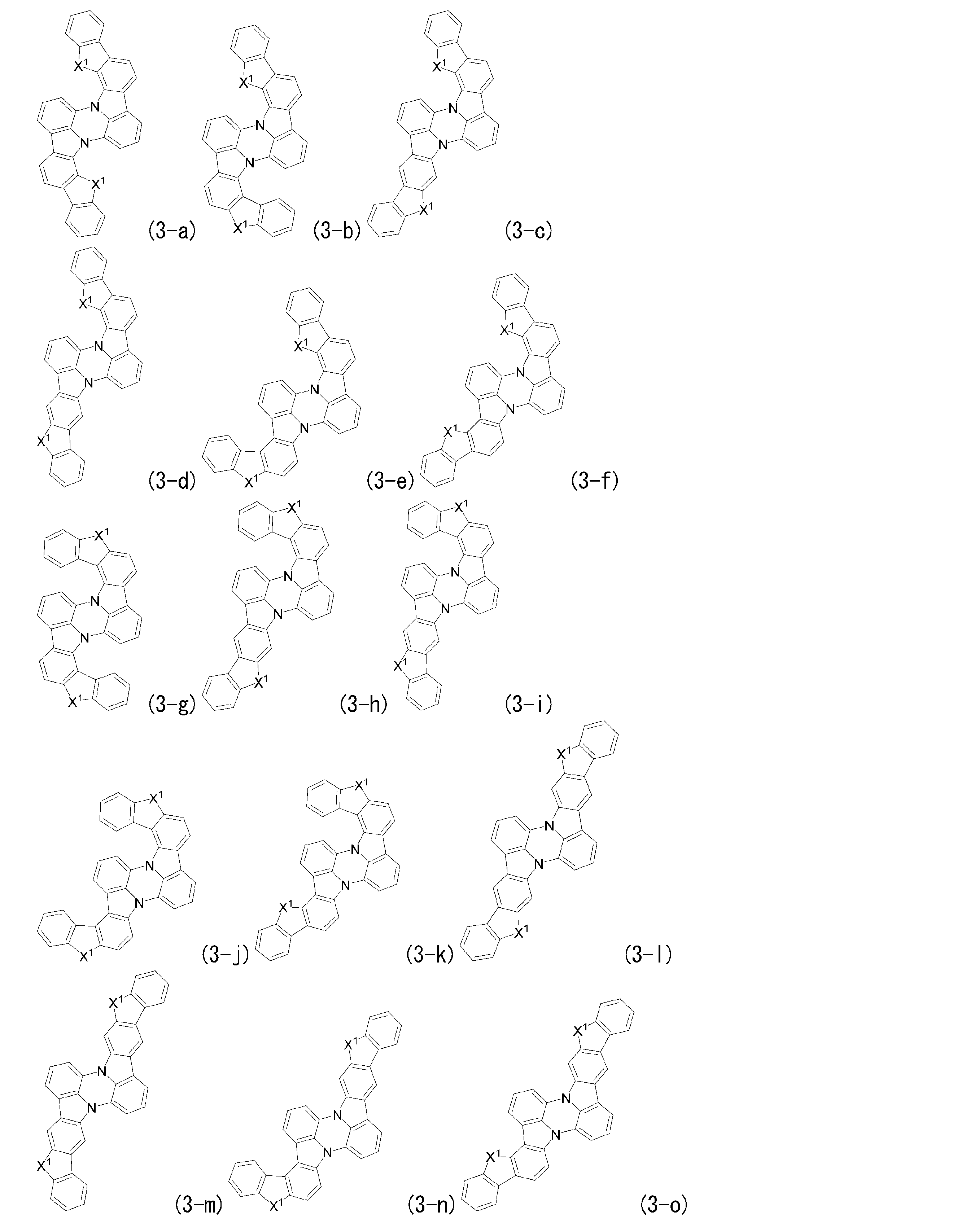
- Preferred embodiments of general formula (1) include general formula (2), general formula (3), and general formula (4).
- general formulas (1) to (4) common symbols have the same meanings.
- General formula (2) corresponds to a structure in which b and c are both 1 and a and d are 0 in general formula (1).
- Ring E has the same definition as in general formula (1).
- General formula (3) corresponds to a structure in which both a and d are 1 and b and c are 0 in general formula (1).
- Ring E has the same definition as in general formula (1).
- the general formula (4) corresponds to a structure in which a, b, c and d are all 0 in the general formula (1).
- each A 1 is CR 1 or N independently.
- R 1 is the same as the general formula (1).
- the number of N in A 1 present in one six-membered ring containing A 1 in general formula (4) is 2 or less.
- R 1 is a substituted or unsubstituted diarylamino group, arylheteroarylamino group, diheteroarylamino group, aromatic hydrocarbon group, or substituted or unsubstituted aromatic
- R 1 is directly with the aromatic ring having A 1 to which R 1 is attached in formula (1), or —O—, —S—, —Si(R a ) 2 —,
- One or more bonds may be formed via —C(R a ) 2 — or —NR a — to form a condensed ring.
- Each R a is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- an aliphatic hydrocarbon group having 1 to 4 carbon atoms More preferably, hydrogen, an aliphatic hydrocarbon group having 1 to 4 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 10 carbon atoms, or a substituted or unsubstituted aromatic heterocyclic ring having 3 to 12 carbon atoms or a substituted or unsubstituted linked aromatic group formed by linking 2 to 4 of these aromatic groups.
- R a is an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 30 carbon atoms, or Specific examples of substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked are the same as in the case of R 1 .
- R 1 is a substituted or unsubstituted diarylamino group, arylheteroarylamino group, diheteroarylamino group, aromatic hydrocarbon group, or substituted or unsubstituted aromatic a heterocyclic group directly with the aromatic ring to which R 1 is bonded, or —O—, —S—, —Si(R a ) 2 —, —C(R a ) 2 —, or —NR a —
- structures such as the following exemplary compounds (D226) to (D236) can be obtained.
- R 1 is a substituted or unsubstituted diarylamino group, arylheteroarylamino group, diheteroarylamino group, aromatic hydrocarbon group, or substituted or unsubstituted aromatic a heterocyclic group directly with the aromatic ring to which R 1 is bonded, or —O—, —S—, —Si(R a ) 2 —, —C(R a ) 2 —, or —NR a —
- structures such as the following exemplary compounds (D237) to (D240) can be obtained.
- Each R c is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- an aliphatic hydrocarbon group having 1 to 4 carbon atoms More preferably, hydrogen, an aliphatic hydrocarbon group having 1 to 4 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 10 carbon atoms, or a substituted or unsubstituted aromatic heterocyclic ring having 3 to 12 carbon atoms or a substituted or unsubstituted linked aromatic group formed by linking 2 to 4 of these aromatic groups.
- R c is an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 30 carbon atoms, or Specific examples of substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked are the same as in the case of R 1 .
- the general formula (1-1) will be explained below.
- the compound represented by general formula (1-1) typically has a structure in which a plurality of benzene rings are condensed to a phenazine ring or a structure similar thereto.
- One of preferred embodiments is represented by general formula (4).
- a 2 is CR 2 , N, or a carbon atom.
- the number of N in A 2 present in one six-membered ring containing A 2 in general formula (1-1) is 2 or less.
- This six-membered ring containing A2 may be fused to the adjacent ring F, in which case two of A2 are carbon atoms, which are shared with ring F.
- Each R 2 is independently hydrogen, a cyano group, deuterium, a substituted or unsubstituted diarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted arylheteroarylamino group having 12 to 44 carbon atoms, a substituted or unsubstituted diheteroarylamino group having 12 to 44 carbon atoms, aliphatic hydrocarbon group having 1 to 10 carbon atoms, substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, substituted or unsubstituted carbon 3 to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- R 2 Specific examples of unsubstituted R 2 are the same as those of R 1 .
- R 2 is a substituted or unsubstituted diarylamino group, arylheteroarylamino group, diheteroarylamino group, aromatic hydrocarbon group, or substituted or unsubstituted aromatic heterocyclic ring.
- R 2 is a substituted or unsubstituted diarylamino group, arylheteroarylamino group, diheteroarylamino group, aromatic hydrocarbon group, or substituted or unsubstituted aromatic heterocyclic ring.
- R 2 is directly with the aromatic ring having A 2 to which R 2 is attached in formula (1-1), or —O—, —S—, —Si(R b ) 2 —,
- the structure can be as shown in the following exemplary compound (D169).
- Each R b is independently hydrogen, deuterium, an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted 3 carbon atoms to 30 aromatic heterocyclic groups, or substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked.
- an aliphatic hydrocarbon group having 1 to 4 carbon atoms More preferably, hydrogen, an aliphatic hydrocarbon group having 1 to 4 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 10 carbon atoms, a substituted or unsubstituted aromatic heterocyclic ring having 2 to 12 carbon atoms or a substituted or unsubstituted linked aromatic group formed by linking 2 to 4 of these aromatic groups.
- R b is an aliphatic hydrocarbon group having 1 to 10 carbon atoms, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 3 to 30 carbon atoms, or Specific examples of substituted or unsubstituted linked aromatic groups in which 2 to 9 of these aromatic groups are linked are the same as in the case of R 1 .
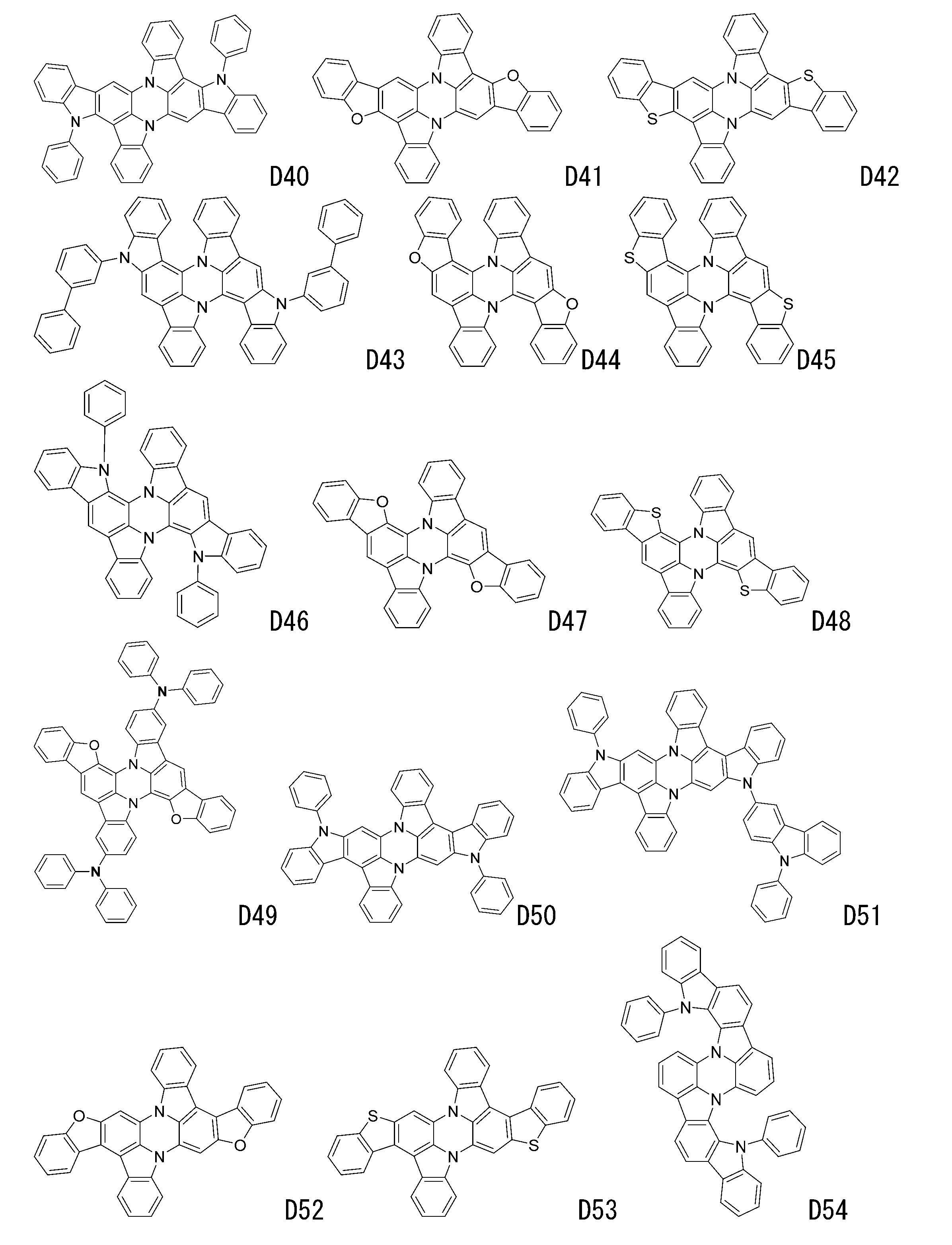
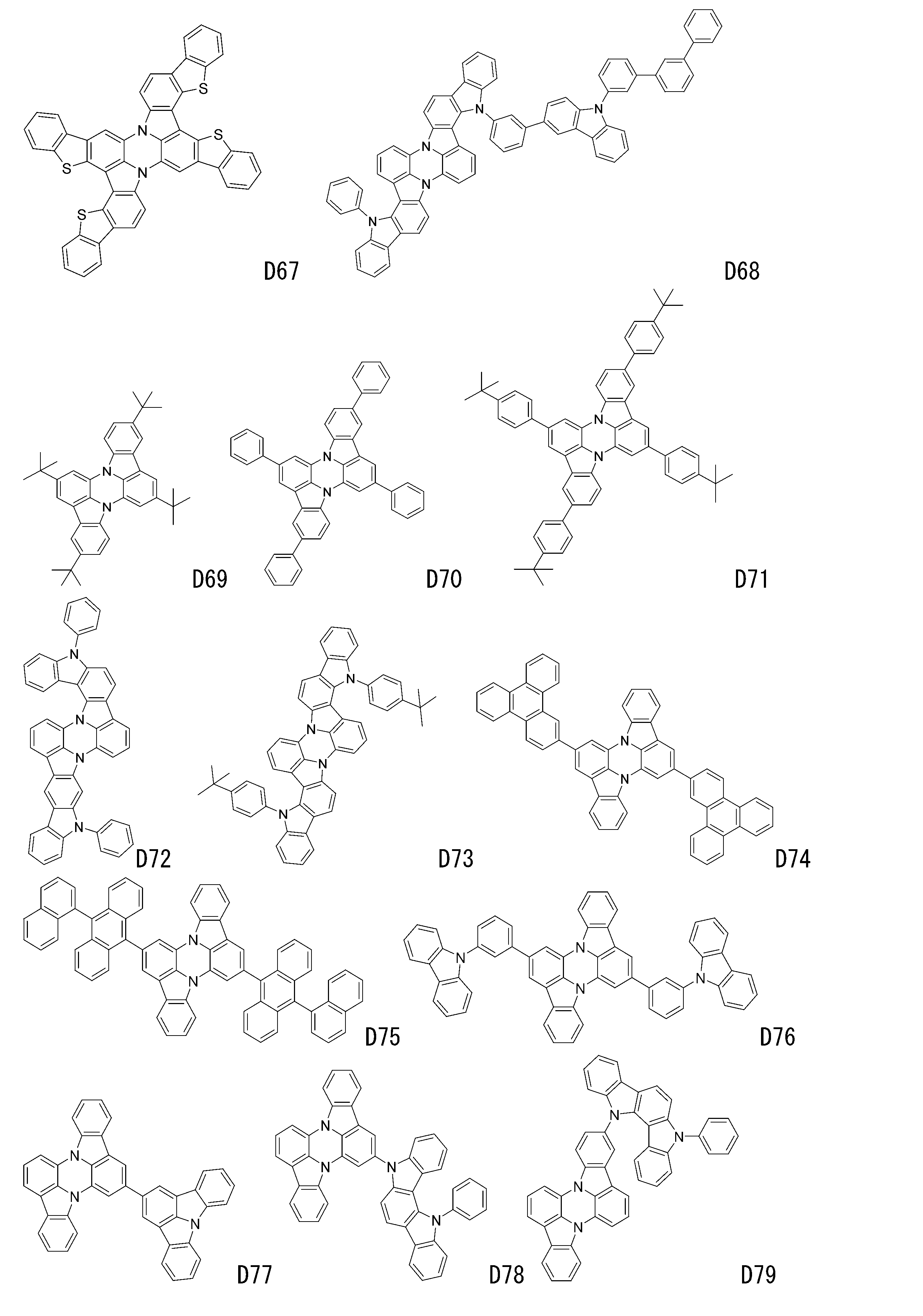
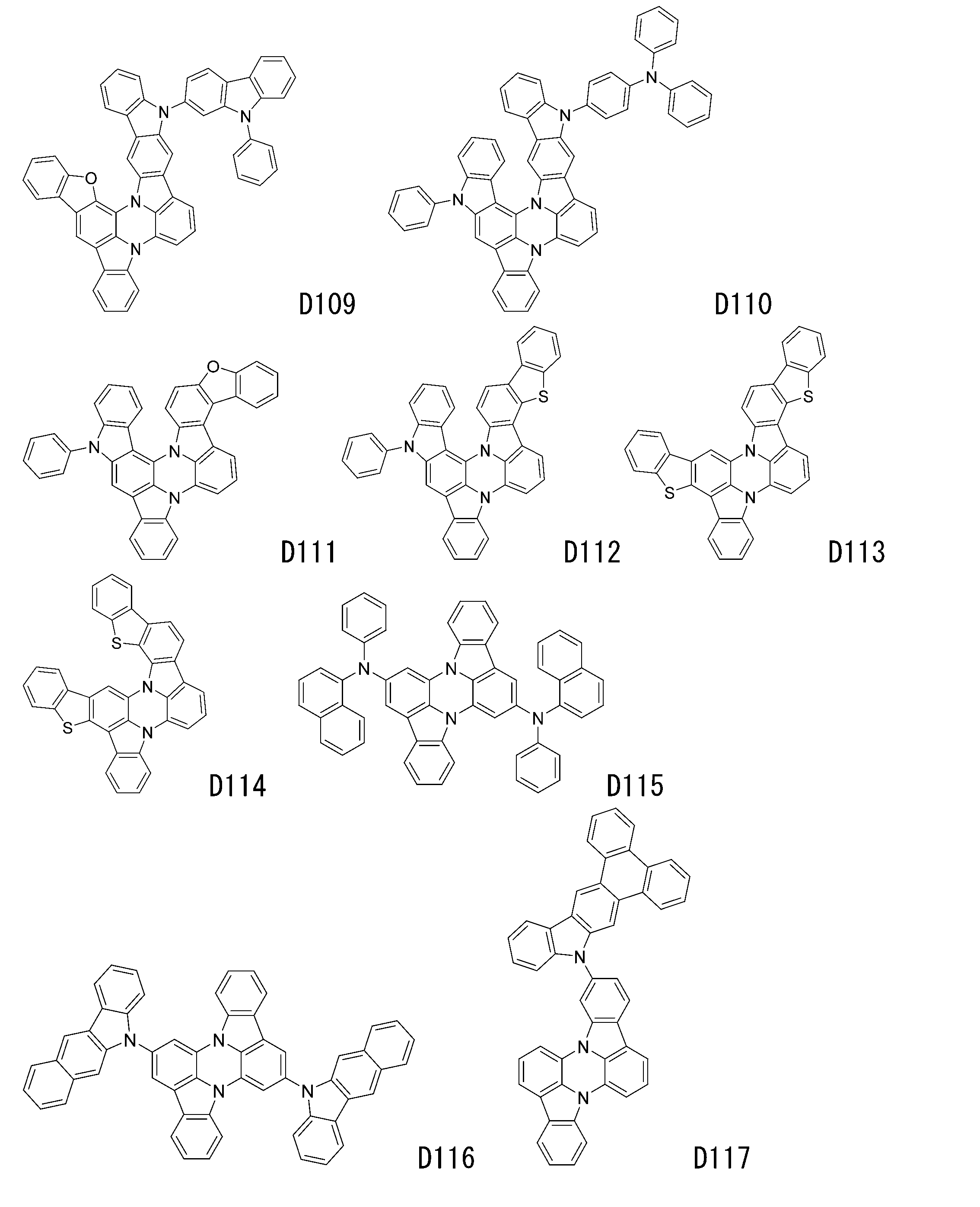
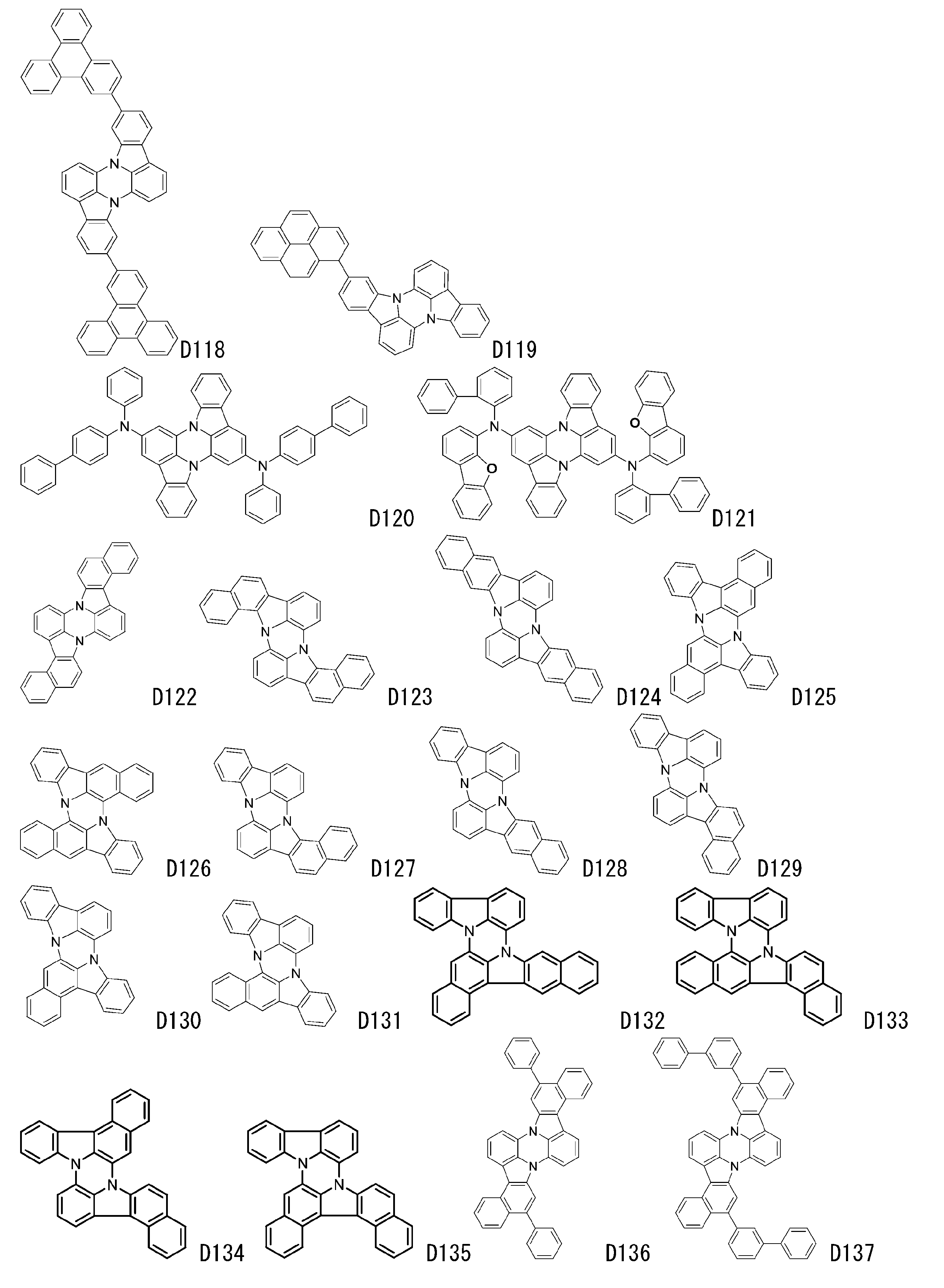
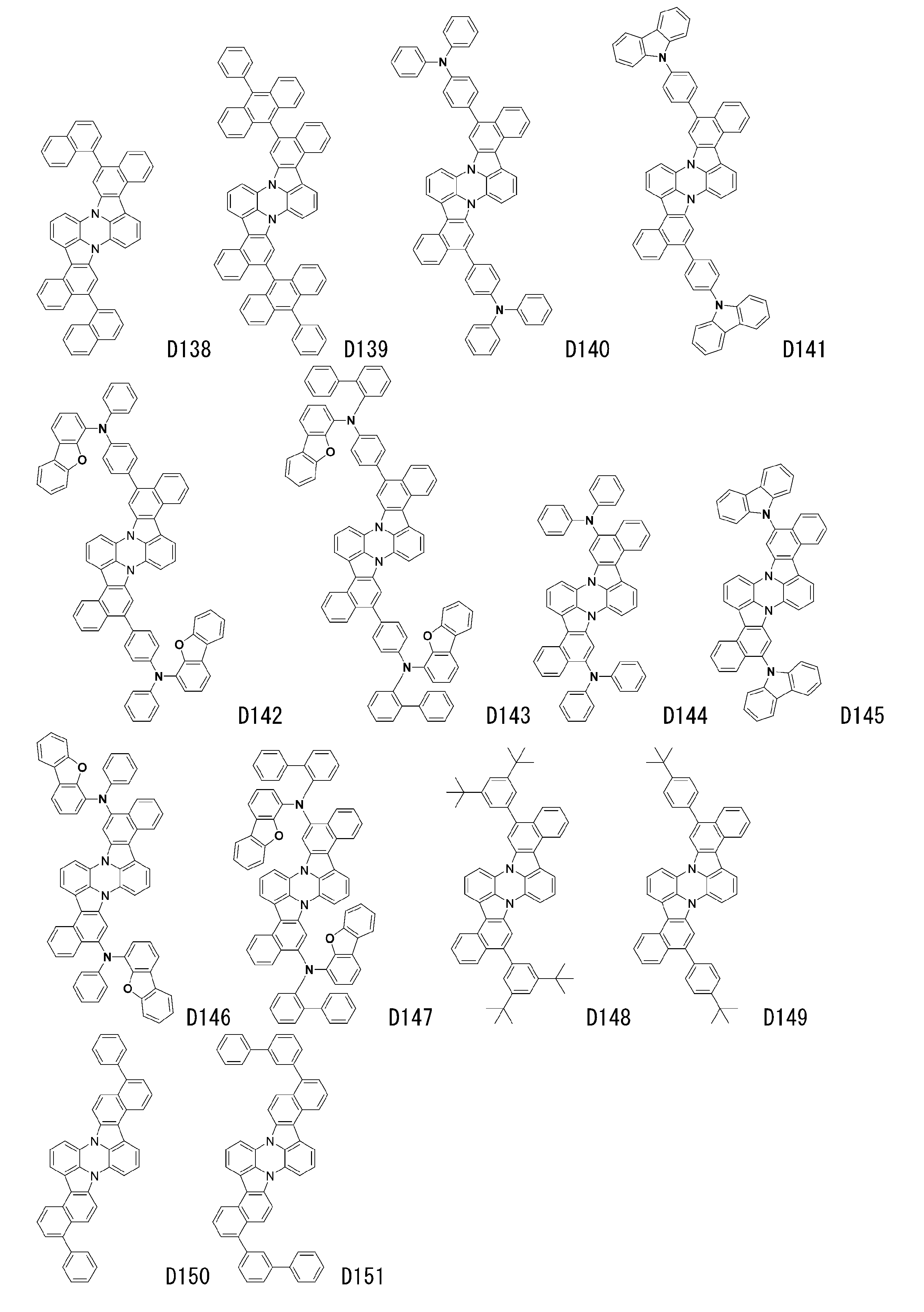
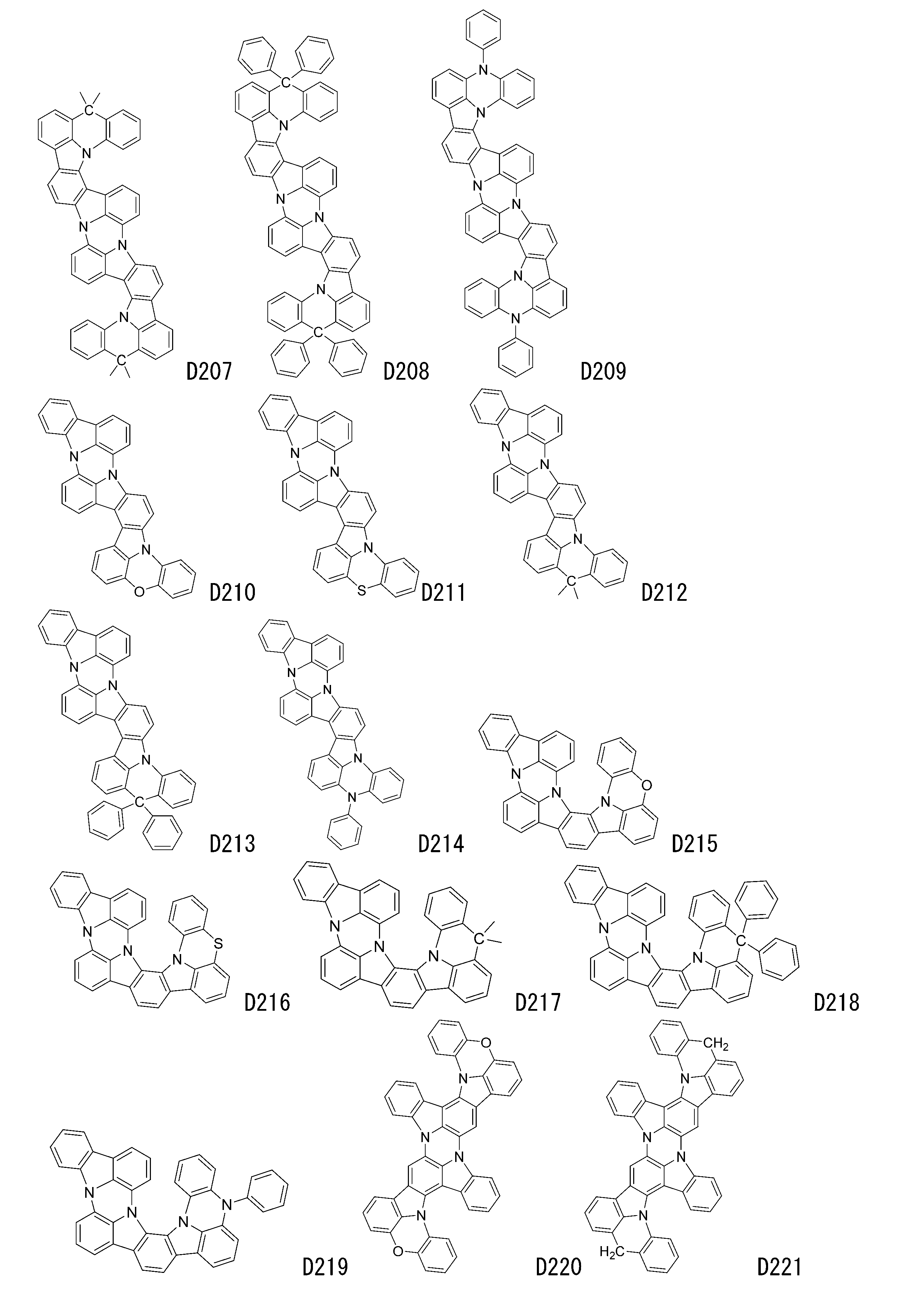
- luminescent materials represented by general formulas (1) to (4) and (1-1) are shown below, but are not limited to these exemplary compounds.
- the organic EL By including the light emitting material represented by the general formula (1) or general formula (1-1) in the light emitting layer, the organic EL emits light with high efficiency and has high driving stability and is excellent in practical use. element.
- FIG. 1 is a cross-sectional view showing a structural example of a general organic EL device used in the present invention, wherein 1 is a substrate, 2 is an anode, 3 is a hole injection layer, 4 is a hole transport layer, and 5 is a light emitting layer. , 6 represents the electron transport layer and 7 represents the cathode.
- the organic EL device of the present invention may have an exciton blocking layer adjacent to the light emitting layer, or may have an electron blocking layer between the light emitting layer and the hole injection layer.
- the exciton blocking layer can be inserted either on the cathode side or the anode side of the light-emitting layer, or both can be inserted at the same time.
- the organic EL device of the present invention has an anode, a light-emitting layer, and a cathode as essential layers. It is preferable to have a hole blocking layer between the transport layers.
- the hole injection transport layer means either or both of the hole injection layer and the hole transport layer
- the electron injection transport layer means either or both of the electron injection layer and the electron transport layer.
- the organic EL element As described above, the layers constituting the laminated structure on the substrate other than the electrodes such as the anode and the cathode are sometimes collectively referred to as the organic layer.
- the organic EL device of the present invention is preferably supported by a substrate.
- the substrate is not particularly limited as long as it is conventionally used in organic EL elements, and can be made of, for example, glass, transparent plastic, quartz, or the like.
- anode material in the organic EL element a material having a large work function (4 eV or more) metal, alloy, electrically conductive compound, or a mixture thereof is preferably used.
- electrode materials include metals such as Au, conductive transparent materials such as CuI, indium tin oxide (ITO), SnO 2 and ZnO.
- a material such as IDIXO (In 2 O 3 —ZnO) that is amorphous and capable of forming a transparent conductive film may also be used.
- the anode may be formed by forming a thin film of these electrode materials by a method such as vapor deposition or sputtering, and forming a pattern of a desired shape by photolithography.
- a pattern may be formed through a mask having a desired shape during vapor deposition or sputtering of the electrode material.
- a coatable substance such as an organic conductive compound
- a wet film forming method such as a printing method or a coating method may be used.
- the transmittance is desirably higher than 10%, and the sheet resistance of the anode is preferably several hundred ⁇ / ⁇ or less.
- the film thickness depends on the material, it is usually selected in the range of 10 to 1000 nm, preferably 10 to 200 nm.
- a cathode material a material composed of a metal having a small work function (4 eV or less) (referred to as an electron-injecting metal), an alloy, an electrically conductive compound, or a mixture thereof is used.
- electrode materials include sodium, sodium-potassium alloys, magnesium, lithium, magnesium/copper mixtures, magnesium/silver mixtures, magnesium/aluminum mixtures, magnesium/indium mixtures, aluminum/aluminum oxide (Al 2 O 3 ) mixtures, indium, lithium/aluminum mixtures, rare earth metals and the like.
- a mixture of an electron injection metal and a second metal that has a higher work function and is more stable such as a magnesium/silver mixture, magnesium /aluminum mixtures, magnesium/indium mixtures, aluminum/aluminum oxide (Al 2 O 3 ) mixtures, lithium/aluminum mixtures, aluminum and the like are suitable.
- the cathode can be produced by forming a thin film of these cathode materials by a method such as vapor deposition or sputtering.
- the sheet resistance of the cathode is preferably several hundred ⁇ / ⁇ or less, and the film thickness is usually selected in the range of 10 nm to 5 ⁇ m, preferably 50 to 200 nm.
- the film thickness is usually selected in the range of 10 nm to 5 ⁇ m, preferably 50 to 200 nm.
- a transparent or translucent cathode can be produced by forming the above metal in a thickness of 1 to 20 nm on the cathode and then forming the conductive transparent material mentioned in the explanation of the anode thereon. By applying this, it is possible to fabricate a device in which both the anode and the cathode are transparent.
- the light-emitting layer is a layer that emits light after recombination of holes and electrons injected from the anode and the cathode to generate excitons.
- a light-emitting material represented by general formula (1) or general formula (1-1) may be used alone, or this light-emitting material may be used together with a host material. When a light-emitting material is used with a host material, the light-emitting material is responsible for light emission in the device.
- the content of the luminescent material is preferably 0.1 to 50 wt%, more preferably 0.1 to 40 wt%, relative to the host material.
- host materials used in phosphorescent light-emitting devices and fluorescent light-emitting devices can be used.
- Known host materials that can be used include compounds having hole-transporting ability and electron-transporting ability and having a high glass transition temperature, and the triplet excitation energy (T1 ), preferably have a triplet excitation energy (T1) greater than .
- T1 triplet excitation energy
- a TADF-active compound may be used as the host material. preferable.
- host materials are known from many patent documents, etc., and can be selected from them.
- Specific examples of the host material include, but are not limited to, indole compounds, carbazole compounds, indolocarbazole compounds, pyridine compounds, pyrimidine compounds, triazine compounds, triazole compounds, oxazole compounds, oxadiazole compounds, and imidazole compounds.
- Carbazole compounds, indolocarbazole compounds, pyridine compounds, pyrimidine compounds, triazine compounds, anthracene compounds, triphenylene compounds, carborane compounds, and porphyrin compounds are preferred.
- Only one type of host may be used in one light-emitting layer, or two or more types of hosts may be used.
- at least one is preferably an electron-transporting compound such as the triazine compound or anthracene compound described above, and the other hosts are carbazole compounds or indolocarbazole compounds. It is preferably a hole-transporting compound such as
- each host is vapor-deposited from a different vapor deposition source, or a pre-mixture is formed by pre-mixing before vapor deposition so that multiple types of hosts can be simultaneously vapor-deposited from one vapor deposition source. .
- the light-emitting material and the host material can be vapor-deposited from different vapor deposition sources, or the light-emitting material and the host material can be simultaneously vapor-deposited from one vapor deposition source by premixing them before vapor deposition to form a premix.
- premixing method a method that can mix uniformly as much as possible is desirable, and examples thereof include pulverization and mixing, heating and melting under reduced pressure or in an inert gas atmosphere such as nitrogen, and sublimation.
- the method is not limited.
- the form of the host and its preliminary mixture may be powder, stick, or granule.
- the injection layer is a layer provided between an electrode and an organic layer to reduce driving voltage and improve luminance. and between the cathode and the light-emitting layer or electron-transporting layer.
- An injection layer can be provided as required.
- the hole-blocking layer has the function of an electron-transporting layer. can improve the recombination probability of electrons and holes in the light-emitting layer.
- the hole blocking layer can be any known hole blocking material. Also, a plurality of types of hole blocking materials may be used in combination.
- Electron blocking layer has the function of a hole-transporting layer, and by blocking electrons while transporting holes, it is possible to improve the probability of recombination of electrons and holes in the light-emitting layer.
- a known electron blocking layer material can be used as the material for the electron blocking layer.
- Exciton blocking layer is a layer that prevents excitons generated by recombination of holes and electrons in the light-emitting layer from diffusing into the charge-transporting layer. It becomes possible to efficiently confine them in the light-emitting layer, and the light-emitting efficiency of the device can be improved.
- An exciton blocking layer can be inserted between two adjacent light-emitting layers in a device in which two or more light-emitting layers are adjacent to each other.
- a known exciton blocking layer material can be used as a material for such an exciton blocking layer.
- Layers adjacent to the light-emitting layer include a hole-blocking layer, an electron-blocking layer, an exciton-blocking layer, and the like. Become.
- the hole-transporting layer is made of a hole-transporting material having a function of transporting holes, and the hole-transporting layer can be provided as a single layer or multiple layers.
- the hole-transporting material has either hole injection or transport or electron barrier properties, and may be either organic or inorganic. Any compound can be selected from conventionally known compounds and used for the hole transport layer. Examples of such hole-transporting materials include porphyrin derivatives, arylamine derivatives, triazole derivatives, oxadiazole derivatives, imidazole derivatives, polyarylalkane derivatives, phenylenediamine derivatives, arylamine derivatives, amino-substituted chalcone derivatives, oxazole derivatives, styrylanthracene.
- arylamine derivatives fluorenone derivatives, hydrazone derivatives, stilbene derivatives, silazane derivatives, aniline copolymers, conductive polymer oligomers, especially thiophene oligomers, etc.
- porphyrin derivatives, arylamine derivatives and styrylamine derivatives can also be used.
- an arylamine compound is used.
- the electron transport layer is made of a material having a function of transporting electrons, and the electron transport layer can be provided as a single layer or multiple layers.
- the electron-transporting material (sometimes also serving as a hole-blocking material) should have the function of transmitting electrons injected from the cathode to the light-emitting layer.
- any compound can be selected and used from conventionally known compounds.
- each layer when producing the organic EL element of the present invention is not particularly limited, and it may be produced by either a dry process or a wet process.
- S1 and T1 are measured as follows.
- a sample compound thermalally activated delayed fluorescence material
- a quartz substrate by a vacuum vapor deposition method at a degree of vacuum of 10 ⁇ 4 Pa or less to form a vapor-deposited film with a thickness of 100 nm.
- S1 measures the emission spectrum of this deposited film, draws a tangent line to the rising edge of the emission spectrum on the short wavelength side, and obtains the wavelength value ⁇ edge [nm] at the intersection of the tangent line and the horizontal axis using the following formula (i). to calculate S1.
- S1[eV] 1239.85/ ⁇ edge (i)
- T1 measures the phosphorescent spectrum of the vapor deposited film, draws a tangent line to the rise on the short wavelength side of the phosphorescent spectrum, and calculates the wavelength value ⁇ edge [nm] at the intersection of the tangent line and the horizontal axis by the formula (ii). to calculate T1.
- T1[eV] 1239.85/ ⁇ edge (ii)
- Example 1 A thin film shown below was formed on a quartz substrate at a degree of vacuum of 4.0 ⁇ 10 ⁇ 5 Pa by a vacuum deposition method.
- BH-1 as a host and compound (D1) as a dopant were co-deposited from different deposition sources to form a light-emitting layer having a thickness of 100 nm. At this time, the co-evaporation was carried out under the condition that the concentration of the compound (D1) was 2% by mass.
- An organic thin film according to Example 1 was produced.
- the above organic thin film was subjected to photoluminescence quantum yield (PLQY) measurement using the Absolute PL Quantum Yield Measurement C9920-03G system (Hamamatsu Photonics Co., Ltd.).
- PLQY photoluminescence quantum yield
- C9920-03G Absolute PL Quantum Yield Measurement C9920-03G system
- the photoexcitation and emission spectrum of the organic thin film can be continuously measured, and the PLQY of the organic thin film can be calculated by calculating the energy balance at this time.
- Maximum emission wavelength, half-width, PLQY and CIE coordinates are determined using software U6039-05 version 3.6.0. Maximum emission wavelength and half-width are given in nm, PLQY in % and CIE coordinates as x,y values.
- the excitation wavelength in PLQY measurement was set to 340 nm.
- Example 2 An organic thin film according to Example 2 was produced in the same manner as in Example 1, except that the compound (D69) was used as the dopant.
- Example 3 A solution was prepared by adjusting the concentration of compound (D41) to 5 ⁇ 10 ⁇ 6 M in toluene. For this solution, the maximum emission wavelength, half width, PLQY and CIE coordinates were determined in the same manner as in Example 1.
- Example 4 A solution was prepared by adjusting the concentration of compound (D122) to 5 ⁇ 10 ⁇ 6 M in toluene. For this solution, the maximum emission wavelength, half width, PLQY and CIE coordinates were determined in the same manner as in Example 1.
- Comparative example 1 An organic thin film was prepared in the same manner as in Example 1, except that BH-1 was used as the dopant.
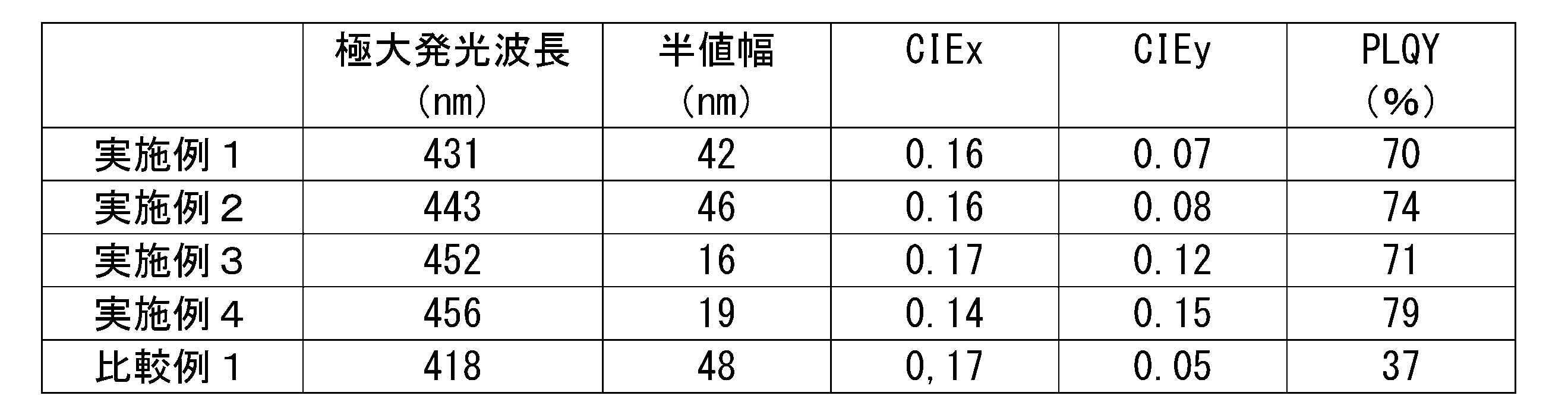
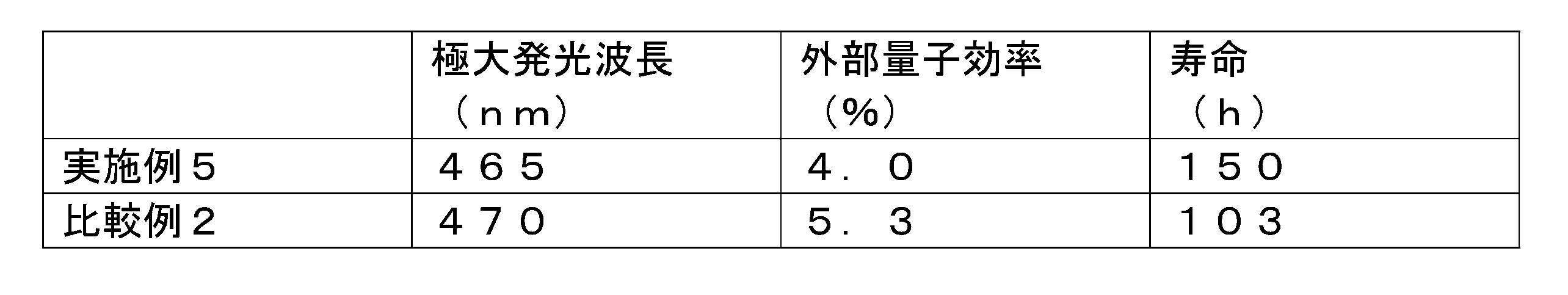
- Table 1 shows the results of measuring the maximum emission wavelength, half width, chromaticity (CIEx, CIEy) and PLQY of the emission spectrum of the produced organic thin film and solution.
- the luminescent material of the present invention has highly efficient characteristics, and it can be seen from the maximum emission wavelength that it emits blue light.
- Example 5 Each thin film shown below was laminated at a degree of vacuum of 4.0 ⁇ 10 ⁇ 5 Pa by a vacuum deposition method on a glass substrate on which an anode made of ITO with a film thickness of 70 nm was formed.
- HAT-CN shown above was formed to a thickness of 10 nm as a hole injection layer on ITO, and then HT-1 was formed to a thickness of 25 nm as a hole transport layer.
- HT-2 was formed with a thickness of 5 nm as an electron blocking layer.
- BH-2 as a host and compound (D122) as a dopant were co-deposited from different vapor deposition sources to form a light-emitting layer having a thickness of 30 nm.
- Table 2 shows the maximum emission wavelength, external quantum efficiency, and lifetime of the produced organic EL device.
- the maximum emission wavelength and external quantum efficiency are values at a drive current density of 2.5 mA/cm 2 , and are initial characteristics.
- the lifetime was measured by measuring the time until the brightness decayed to 90% of the initial brightness when the driving current density was 40 mA/cm 2 .
- the organic EL device using the luminescent material of the present invention has high characteristics, and it can be seen from the maximum emission wavelength that it emits blue light. In addition, particularly excellent results were obtained with respect to life characteristics.
- the luminescent material of the present invention it is possible to obtain a practically useful organic EL device that emits light with high efficiency and has high driving stability.
- the luminescent material of the invention also exhibits a maximum wavelength in the blue, light blue or green spectral region. This luminescent material exhibits a wavelength maximum in particular between 410 nm and 550 nm, preferably between 430 nm and 495 nm.
- the photoluminescence quantum yield of the luminescent material of the present invention can be 40% or more. Use of the luminescent materials of the present invention results in more efficient devices. Further, an organic EL device having a light-emitting layer containing this has high luminous efficiency and color.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Electroluminescent Light Sources (AREA)
- Optics & Photonics (AREA)
Abstract
Description
環Eはそれぞれ独立して、式(1a)で表される複素環であり、環Eは隣接する環と任意の位置で縮合する。X1はそれぞれ独立して、Si(Rd)2、C(Rd)2、O、S、Se、又はN-L1-(Ar1)eで表される基である。Rdは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。L1はそれぞれ独立して、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、又は置換若しくは未置換の炭素数3~30の芳香族複素環基であり、Ar1はそれぞれ独立して、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれらの芳香族環が2~9個連結してなる置換若しくは未置換の連結芳香族基である。L1は、L1が結合する複素環と隣接する芳香族環と直接、又は―O―、―S―、―Si(Rc)2―、―C(Rc)2―、又は―NRc―を介して1つの結合を形成し、縮合環を形成してもよい。Rcは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。a、b、c、及びdはそれぞれ独立して、0又は1を表し、eはそれぞれ独立して、0~5の整数を表す。但し、R1又はL1が、R1又はL1が結合する芳香族環やその隣接する芳香族環と縮合環を形成しないとき、a+b+c+d=1であることはない。
A2はそれぞれ独立にCR2、C又はNである。但し、一般式(1-1)でA2を含んだ一つの六員環中に存在するNの数は2以下である。R2は、それぞれ独立に、水素、シアノ基、重水素、置換若しくは未置換の炭素数12~44のジアリールアミノ基、置換若しくは未置換の炭素数12~44のアリールヘテロアリールアミノ基、置換若しくは未置換の炭素数12~44のジヘテロアリールアミノ基、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。R2が置換若しくは未置換のジアリールアミノ基、アリールヘテロアリールアミノ基、ジヘテロアリールアミノ基、芳香族炭化水素基、又は置換若しくは未置換の芳香族複素環基であるとき、R2は、式(1-1)中でR2が結合するA2を有する芳香族環と直接、又は―O―、―S―、―Si(Rb)2―、―C(Rb)2―、又は―NRb―を介して1つ以上の結合を形成し、縮合環を形成してもよい。Rbは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。環Fはそれぞれ独立して、式(1-1a)で表される芳香族環であり、環Fは隣接する環と任意の位置で縮合する。
x、y、w、及びzはそれぞれ独立して、0又は1を表す。
ここで、A1はそれぞれ独立にCR1、又はNである。R1は一般式(1)のR1と同意である。但し、一般式(4)でA1を含んだ一つの六員環中に存在するNの数は2以下である。
ここで、X1は前記一般式(1)と同義である。好ましくは、式(2-a)、(2-e)、(2-h)、又は(2-j)である。
ここで、X1は前記一般式(1)と同義である。好ましくは、式(3-a)、(3-g)、(3-l)、(3-p)、(3-s)、及び(3-u)である。
Raは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。好ましくは、水素、重水素、炭素数1~8の脂肪族炭化水素基、置換若しくは未置換の炭素数6~20の芳香族炭化水素基、置換若しくは未置換の炭素数3~20の芳香族複素環基、又はこれら芳香族基が2~6個連結してなる置換若しくは未置換の連結芳香族基である。より好ましくは、水素、炭素数1~4の脂肪族炭化水素基、置換若しくは未置換の炭素数6~10の芳香族炭化水素基、置換若しくは未置換の炭素数3~12の芳香族複素環基、又はこれら芳香族基が2~4個連結してなる置換若しくは未置換の連結芳香族基である。
Rcは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。好ましくは、水素、重水素、炭素数1~8の脂肪族炭化水素基、置換若しくは未置換の炭素数6~20の芳香族炭化水素基、置換若しくは未置換の炭素数3~20の芳香族複素環基、又はこれら芳香族基が2~6個連結してなる置換若しくは未置換の連結芳香族基である。より好ましくは、水素、炭素数1~4の脂肪族炭化水素基、置換若しくは未置換の炭素数6~10の芳香族炭化水素基、置換若しくは未置換の炭素数3~12の芳香族複素環基、又はこれら芳香族基が2~4個連結してなる置換若しくは未置換の連結芳香族基である。
本発明の有機EL素子は、基板に支持されていることが好ましい。この基板については特に制限はなく、従来から有機EL素子に用いられているものであればよく、例えばガラス、透明プラスチック、石英等からなるものを用いることができる。
有機EL素子における陽極材料としては、仕事関数の大きい(4eV以上)金属、合金、電気伝導性化合物又はこれらの混合物からなる材料が好ましく用いられる。このような電極材料の具体例としてはAu等の金属、CuI、インジウムチンオキシド(ITO)、SnO2、ZnO等の導電性透明材料が挙げられる。また、IDIXO(In2O3-ZnO)等の非晶質で、透明導電膜を作製可能な材料を用いてもよい。陽極はこれらの電極材料を蒸着やスパッタリング等の方法により薄膜を形成させ、フォトリソグラフィー法で所望の形状のパターンを形成してもよく、あるいはパターン精度をあまり必要としない場合(100μm以上程度)は、上記電極材料の蒸着やスパッタリング時に所望の形状のマスクを介してパターンを形成してもよい。あるいは有機導電性化合物のような塗布可能な物質を用いる場合には印刷方式、コーティング方式等湿式成膜法を用いることもできる。この陽極より発光を取り出す場合には、透過率を10%より大きくすることが望ましく、また、陽極としてのシート抵抗は数百Ω/□以下が好ましい。膜厚は材料にもよるが、通常10~1000nm、好ましくは10~200nmの範囲で選ばれる。
一方、陰極材料としては仕事関数の小さい(4eV以下)金属(電子注入性金属と称する)、合金、電気伝導性化合物又はこれらの混合物からなる材料が用いられる。このような電極材料の具体例としては、ナトリウム、ナトリウム-カリウム合金、マグネシウム、リチウム、マグネシウム/銅混合物、マグネシウム/銀混合物、マグネシウム/アルミニウム混合物、マグネシウム/インジウム混合物、アルミニウム/酸化アルミニウム(Al2O3)混合物、インジウム、リチウム/アルミニウム混合物、希土類金属等が挙げられる。これらの中で、電子注入性及び酸化等に対する耐久性の点から、電子注入性金属とこれより仕事関数の値が大きく安定な金属である第二金属との混合物、例えばマグネシウム/銀混合物、マグネシウム/アルミニウム混合物、マグネシウム/インジウム混合物、アルミニウム/酸化アルミニウム(Al2O3)混合物、リチウム/アルミニウム混合物、アルミニウム等が好適である。陰極はこれらの陰極材料を蒸着やスパッタリング等の方法により薄膜を形成させることにより、作製することができる。また、陰極としてシート抵抗は数百Ω/□以下が好ましく、膜厚は通常10nm~5μm、好ましくは50~200nmの範囲で選ばれる。なお、発光した光を透過させるため、有機EL素子の陽極又は陰極のいずれか一方が透明又は半透明であれば発光輝度は向上し、好都合である。
発光層は陽極及び陰極のそれぞれから注入された正孔及び電子が再結合することにより励起子が生成した後、発光する層である。発光層には、一般式(1)又は一般式(1-1)で表される発光材料を単独で使用してもよいし、この発光材料をホスト材料と共に使用してもよい。発光材料をホスト材料と共に使用する場合、発光材料が素子中で発光を担う。
注入層とは、駆動電圧低下や発光輝度向上のために電極と有機層間に設けられる層のことで、正孔注入層と電子注入層があり、陽極と発光層又は正孔輸送層の間、及び陰極と発光層又は電子輸送層との間に存在させてもよい。注入層は必要に応じて設けることができる。
正孔阻止層とは広い意味では電子輸送層の機能を有し、電子を輸送する機能を有しつつ正孔を輸送する能力が著しく小さい正孔阻止材料からなり、電子を輸送しつつ正孔を阻止することで発光層中での電子と正孔の再結合確率を向上させることができる。正孔阻止層には、公知の正孔阻止材料をすることができる。また正孔阻止材料を複数種類併用して用いてもよい。
電子阻止層とは広い意味では正孔輸送層の機能を有し、正孔を輸送しつつ電子を阻止することで発光層中での電子と正孔が再結合する確率を向上させることができる。電子阻止層の材料としては、公知の電子阻止層材料を用いることができる。
励起子阻止層とは、発光層内で正孔と電子が再結合することにより生じた励起子が電荷輸送層に拡散することを阻止するための層であり、本層の挿入により励起子を効率的に発光層内に閉じ込めることが可能となり、素子の発光効率を向上させることができる。励起子阻止層は2つ以上の発光層が隣接する素子において、隣接する2つの発光層の間に挿入することができる。このような励起子阻止層の材料としては、公知の励起子阻止層材料を用いることができる。
正孔輸送層とは正孔を輸送する機能を有する正孔輸送材料からなり、正孔輸送層は単層又は複数層設けることができる。
電子輸送層とは電子を輸送する機能を有する材料からなり、電子輸送層は単層又は複数層設けることができる。
S1[eV] = 1239.85/λedge (i)
T1[eV] = 1239.85/λedge (ii)
窒素雰囲気下、原料(A)3.0g、銅 0.78g、炭酸カリウム 3.5g、8-キノリノール(8HQ) 0.36g、1,3-ジメチル-2-イミダゾリジノン(DMI) 6.0mlを三口フラスコに入れ、190℃で72時間撹拌した。反応溶液を室温に戻し、メタノールを少しずつ添加して得られた沈降物を濾取した。濾取物をシリカゲルカラムクロマトグラフィーで精製した。その後メタノールで洗浄し、得られた固体を減圧乾燥することで化合物(D1) 0.15g(収率:7.2%)を得た。
APCI-TOFMS m/z 331[M+1]
合成例2
窒素雰囲気下、原料(B)3.0g、銅 0.53g、炭酸カリウム 2.4g、8-キノリノール(8HQ) 0.25g、1,3-ジメチル-2-イミダゾリジノン(DMI) 4.0mlを三口フラスコに入れ、190℃で72時間撹拌した。反応溶液を室温に戻し、メタノールを少しずつ添加して得られた沈降物を濾取した。濾取物をシリカゲルカラムクロマトグラフィーで精製した。その後メタノールで洗浄し、得られた固体を減圧乾燥することで化合物(D69) 0.4g(収率:9.2%)を得た。
APCI-TOFMS m/z 554[M+1]
合成例3
窒素雰囲気下、原料(C)5.6g、リン酸三カリウム 12.9g、1,3-ジメチル-2-イミダゾリジノン(DMI) 48.0mlを三口フラスコに入れ、230℃で14日間撹拌した。反応溶液を室温に戻し、メタノールを少しずつ添加して得られた沈降物を濾取した。濾取物をテトラヒドロフラン、キシレンで再沈殿することで精製した。その後メタノールで洗浄し、得られた固体を減圧乾燥することで化合物(D41) 0.4g(収率:4.0%)を得た。
APCI-TOFMS m/z 510[M+1]
合成例4
窒素雰囲気下、原料(D)7.8g、リン酸三カリウム 21.1g、1,3-ジメチル-2-イミダゾリジノン(DMI) 83.0mlを三口フラスコに入れ、230℃で10日間撹拌した。反応溶液を室温に戻し、メタノールを少しずつ添加して得られた沈降物を濾取した。濾取物をテトラヒドロフラン、キシレンで再沈殿することで精製した。その後メタノールで洗浄し、得られた固体を減圧乾燥することで化合物(D122) 0.5g(収率:3.5%)を得た。
APCI-TOFMS m/z 430[M+1]
石英基板上に、以下に示す薄膜を真空蒸着法にて、真空度4.0×10-5Paで製膜した。ホストとしてBH-1を、ドーパントとして化合物(D1)をそれぞれ異なる蒸着源から共蒸着し、100nmの厚さを有する発光層を形成した。この時、化合物(D1)の濃度が2質量%となる蒸着条件で共蒸着した。実施例1に係る有機薄膜を作製した。
ドーパントとして化合物(D69)を使用した以外は実施例1と同様にして、実施例2に係る有機薄膜を作製した。
化合物(D41)をトルエンに5×10-6Mの濃度で調整した溶液を作製した。この溶液について実施例1と同様にして極大発光波長、半値幅、PLQY及びCIE座標を決定した。
化合物(D122)をトルエンに5×10-6Mの濃度で調整した溶液を作製した。この溶液について実施例1と同様にして極大発光波長、半値幅、PLQY及びCIE座標を決定した。
ドーパントをBH-1とした以外は実施例1と同様にして、有機薄膜を作製した。
膜厚70nmのITOからなる陽極が形成されたガラス基板上に、以下に示す各薄膜を真空蒸着法にて、真空度4.0×10-5Paで積層した。先ず、ITO上に正孔注入層として先に示したHAT-CNを10nmの厚さに形成し、次に、正孔輸送層としてHT-1を25nmの厚さに形成した。次に、電子阻止層としてHT-2を5nmの厚さに形成した。そして、ホストとしてBH-2を、ドーパントとして化合物(D122)をそれぞれ異なる蒸着源から共蒸着し、30nmの厚さを有する発光層を形成した。この時、化合物(D122)の濃度1質量%となる蒸着条件で共蒸着した。次に、正孔阻止層としてBH-2を5nmの厚さに形成した。次に電子輸送層としてALQ3を40nmの厚さに形成した。更に、電子輸送層上に電子注入層としてフッ化リチウム(LiF)を1nmの厚さに形成した。最後に、電子注入層上に陰極としてアルミニウム(Al)を70nmの厚さに形成し、実施例5に係る有機EL素子を作製した。
比較例2
ドーパントをv-DABNAとした以外は実施例5と同様にして、有機EL素子を作製した。
Claims (13)
- 下記一般式(1)又は下記一般式(1-1)で表される発光材料。
(ここで、A1はそれぞれ独立にCR1、C又はNである。但し、一般式(1)でA1を含んだ一つの六員環中に存在するNの数は2以下である。R1は、それぞれ独立に、水素、シアノ基、重水素、置換若しくは未置換の炭素数12~44のジアリールアミノ基、置換若しくは未置換の炭素数12~44のアリールヘテロアリールアミノ基、置換若しくは未置換の炭素数12~44のジヘテロアリールアミノ基、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。R1が置換若しくは未置換のジアリールアミノ基、アリールヘテロアリールアミノ基、ジヘテロアリールアミノ基、芳香族炭化水素基、又は置換若しくは未置換の芳香族複素環基であるとき、R1は、式(1)中でR1が結合するA1を有する芳香族環と直接、又は―O―、―S―、―Si(Ra)2―、―C(Ra)2―、又は―NRa―を介して1つ以上の結合を形成し、縮合環を形成してもよい。Raは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。環Eはそれぞれ独立して、式(1a)で表される複素環であり、環Eは隣接する環と任意の位置で縮合する。X1はそれぞれ独立して、Si(Rd)2、C(Rd)2、O、S、Se、又はN-L1-(Ar1)eで表される基である。Rdは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。L1はそれぞれ独立して、置換若しくは未置換の炭素数6~30の芳香族炭化水素基又は置換若しくは未置換の炭素数3~30の芳香族複素環基であり、Ar1はそれぞれ独立して、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれらの芳香族環が2~9個連結してなる置換若しくは未置換の連結芳香族基である。L1は、L1が結合する複素環と隣接する芳香族環と直接、又は―O―、―S―、―Si(Rc)2―、―C(Rc)2―、又は―NRc―を介して1つの結合を形成し、縮合環を形成してもよい。Rcは、それぞれ独立に、水素、重水素、炭素数1~10の脂肪族炭化水素基、置換若しくは未置換の炭素数6~30の芳香族炭化水素基、置換若しくは未置換の炭素数3~30の芳香族複素環基、又はこれら芳香族基が2~9個連結してなる置換若しくは未置換の連結芳香族基である。a、b、c、及びdはそれぞれ独立して、0又は1を表し、eはそれぞれ独立して、0~5の整数を表す。但し、R1又はL1が隣接する芳香族環と縮合環を形成しないとき、a+b+c+d=1であることはない。)
- a+b+c+d≧2であることを特徴とする請求項1に記載の発光材料。
- b及びc、又はa及びdが、1であることを特徴とする請求項2に記載の発光材料。
- 前記一般式(1)において、環Eのそれぞれが独立して式(1a)中のX1がN-L1-(Ar1)eで表される基であることを特徴とする請求項1に記載の発光材料。
- 励起一重項エネルギー(S1)と励起三重項エネルギー(T1)の差(ΔEST)が0.40eV以下であることを特徴とする請求項1に記載の発光材料。
- 少なくとも一つのR1又はL1が、式(1)中でR1が結合するA1を有する芳香族環若しくはL1が結合する複素環と隣接する芳香族環と直接、又は―O―、―S―、―Si(Ra)2―、―C(Ra)2―、又は―NRa―を介して1つ以上の結合を形成し、縮合環を形成することを特徴とする一般式(1)で表される請求項1に記載の発光材料。
- 少なくとも一つのL1が、L1が結合する複素環と隣接する芳香族環と直接結合し縮合環を形成することを特徴とする一般式(1)で表される請求項9に記載の発光材料。
- 少なくとも一つのR1が置換若しくは未置換のカルバゾリル基であり、該カルバゾリル基が式(1)中でR1が結合するA1を有する芳香族環と直接、又は―O―、―S―、―Si(Ra)2―、―C(Ra)2―、又は―NRa―を介して1つ以上の結合を形成し、縮合環を形成することを特徴とする一般式(1)で表される請求項9に記載の発光材料。
- x+y+w+z≧2であることを特徴とする一般式(1-1)で表される請求項1に記載の発光材料。
- 対向する陽極と陰極の間に1つ以上の発光層を含む有機電界発光素子において、少なくとも1つの発光層が、請求項1~12のいずれかに記載の発光材料を含有することを特徴とする有機電界発光素子。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202280023607.4A CN117083276A (zh) | 2021-04-08 | 2022-03-28 | 发光材料、及有机电场发光元件 |
| KR1020237030947A KR20230167351A (ko) | 2021-04-08 | 2022-03-28 | 발광 재료, 및 유기 전계 발광 소자 |
| EP22784564.1A EP4322236A1 (en) | 2021-04-08 | 2022-03-28 | Light-emitting material, and organic electroluminescent element |
| JP2023512953A JPWO2022215580A1 (ja) | 2021-04-08 | 2022-03-28 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-066013 | 2021-04-08 | ||
| JP2021066013 | 2021-04-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022215580A1 true WO2022215580A1 (ja) | 2022-10-13 |
Family
ID=83545484
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/015058 WO2022215580A1 (ja) | 2021-04-08 | 2022-03-28 | 発光材料、及び有機電界発光素子 |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP4322236A1 (ja) |
| JP (1) | JPWO2022215580A1 (ja) |
| KR (1) | KR20230167351A (ja) |
| CN (1) | CN117083276A (ja) |
| WO (1) | WO2022215580A1 (ja) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010134350A1 (ja) | 2009-05-22 | 2010-11-25 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2011070963A1 (ja) | 2009-12-07 | 2011-06-16 | 新日鐵化学株式会社 | 有機発光材料及び有機発光素子 |
| JP2012234873A (ja) | 2011-04-28 | 2012-11-29 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子材料、有機エレクトロルミネッセンス素子、表示装置および照明装置 |
| WO2014008967A2 (de) | 2012-07-10 | 2014-01-16 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| KR20180008283A (ko) * | 2016-07-14 | 2018-01-24 | 주식회사 엘지화학 | 화합물 및 이를 포함하는 유기 전자 소자 |
| CN110759925A (zh) * | 2018-12-06 | 2020-02-07 | 广州华睿光电材料有限公司 | 含氮稠环有机化合物及其应用 |
| CN111892607A (zh) | 2020-08-26 | 2020-11-06 | 上海天马有机发光显示技术有限公司 | 一种n杂联苯类有机化合物及其应用 |
| US20210098715A1 (en) * | 2019-09-30 | 2021-04-01 | Samsung Display Co., Ltd. | Organic electroluminescence device and polycyclic compound for organic electroluminescence device |
| KR20210096958A (ko) * | 2020-01-29 | 2021-08-06 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물, 이를 이용한 유기전기소자 및 그 전자 장치 |
-
2022
- 2022-03-28 JP JP2023512953A patent/JPWO2022215580A1/ja active Pending
- 2022-03-28 CN CN202280023607.4A patent/CN117083276A/zh active Pending
- 2022-03-28 KR KR1020237030947A patent/KR20230167351A/ko unknown
- 2022-03-28 WO PCT/JP2022/015058 patent/WO2022215580A1/ja active Application Filing
- 2022-03-28 EP EP22784564.1A patent/EP4322236A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010134350A1 (ja) | 2009-05-22 | 2010-11-25 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2011070963A1 (ja) | 2009-12-07 | 2011-06-16 | 新日鐵化学株式会社 | 有機発光材料及び有機発光素子 |
| JP2012234873A (ja) | 2011-04-28 | 2012-11-29 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子材料、有機エレクトロルミネッセンス素子、表示装置および照明装置 |
| WO2014008967A2 (de) | 2012-07-10 | 2014-01-16 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| KR20180008283A (ko) * | 2016-07-14 | 2018-01-24 | 주식회사 엘지화학 | 화합물 및 이를 포함하는 유기 전자 소자 |
| CN110759925A (zh) * | 2018-12-06 | 2020-02-07 | 广州华睿光电材料有限公司 | 含氮稠环有机化合物及其应用 |
| US20210098715A1 (en) * | 2019-09-30 | 2021-04-01 | Samsung Display Co., Ltd. | Organic electroluminescence device and polycyclic compound for organic electroluminescence device |
| KR20210096958A (ko) * | 2020-01-29 | 2021-08-06 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물, 이를 이용한 유기전기소자 및 그 전자 장치 |
| CN111892607A (zh) | 2020-08-26 | 2020-11-06 | 上海天马有机发光显示技术有限公司 | 一种n杂联苯类有机化合物及其应用 |
Non-Patent Citations (1)
| Title |
|---|
| JOURNAL OF MATERIAL CHEMISTRY C, vol. 5, no. 3, 2017, pages 709 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20230167351A (ko) | 2023-12-08 |
| JPWO2022215580A1 (ja) | 2022-10-13 |
| EP4322236A1 (en) | 2024-02-14 |
| CN117083276A (zh) | 2023-11-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2018173598A1 (ja) | 有機電界発光素子 | |
| KR102659607B1 (ko) | 유기 전계 발광 소자 | |
| TW201940664A (zh) | 有機電場發光元件 | |
| JP7456997B2 (ja) | 有機電界発光素子用溶融混合物、及び有機電界発光素子 | |
| WO2021200252A1 (ja) | 有機電界発光素子 | |
| EP4083033A1 (en) | Organic electroluminescent element | |
| JP7198804B2 (ja) | 有機電界発光素子 | |
| WO2021200250A1 (ja) | 有機電界発光素子 | |
| WO2022215580A1 (ja) | 発光材料、及び有機電界発光素子 | |
| WO2024058146A1 (ja) | 発光材料、及び有機電界発光素子 | |
| JP2020105341A (ja) | 熱活性化遅延蛍光発光材料、及び有機電界発光素子 | |
| WO2023157629A1 (ja) | 発光材料、及び有機電界発光素子 | |
| WO2022264638A1 (ja) | 有機電界発光素子用材料、及び有機電界発光素子 | |
| WO2023162701A1 (ja) | 有機電界発光素子用材料、及び有機電界発光素子 | |
| WO2022085590A1 (ja) | 発光材料、及び有機電界発光素子 | |
| WO2024048535A1 (ja) | 有機電界発光素子用ホスト材料及び予備混合物並びに有機電界発光素子 | |
| US20240179929A1 (en) | Light-emitting material, and organic electroluminescent element | |
| WO2024048536A1 (ja) | 有機電界発光素子 | |
| EP4130192A1 (en) | Organic electroluminescent device | |
| WO2024019072A1 (ja) | 有機電界発光素子 | |
| WO2022255241A1 (ja) | 重水素化物及び有機電界発光素子 | |
| WO2022255242A1 (ja) | 重水素化物及び有機電界発光素子 | |
| WO2022009883A1 (ja) | 熱活性化遅延蛍光材料、及び有機電界発光素子 | |
| WO2022255243A1 (ja) | 重水素化物及び有機電界発光素子 | |
| WO2023008501A1 (ja) | 有機電界発光素子 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22784564 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18279313 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023512953 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280023607.4 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022784564 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022784564 Country of ref document: EP Effective date: 20231108 |