WO2021070874A1 - 血管内皮幹細胞の製造方法 - Google Patents
血管内皮幹細胞の製造方法 Download PDFInfo
- Publication number
- WO2021070874A1 WO2021070874A1 PCT/JP2020/038029 JP2020038029W WO2021070874A1 WO 2021070874 A1 WO2021070874 A1 WO 2021070874A1 JP 2020038029 W JP2020038029 W JP 2020038029W WO 2021070874 A1 WO2021070874 A1 WO 2021070874A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cells
- vascular endothelial
- neonatal
- stem cell
- liver
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/069—Vascular Endothelial cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/069—Vascular Endothelial cells
- C12N5/0692—Stem cells; Progenitor cells; Precursor cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/44—Vessels; Vascular smooth muscle cells; Endothelial cells; Endothelial progenitor cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/50—Cell markers; Cell surface determinants
- C12N2501/599—Cell markers; Cell surface determinants with CD designations not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2502/00—Coculture with; Conditioned medium produced by
- C12N2502/14—Coculture with; Conditioned medium produced by hepatocytes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/28—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from vascular endothelial cells
Definitions
- the present invention relates to a method for producing vascular endothelial stem cells.
- vascular diseases Diseases related to blood vessels are collectively called vascular diseases, and there are many diseases such as malignant tumors, retinopathy, chronic inflammatory diseases, arteriosclerosis, and ischemic diseases.
- diseases such as malignant tumors, retinopathy, chronic inflammatory diseases, arteriosclerosis, and ischemic diseases.
- the development of therapeutic methods for improving the diseases by controlling angioplasty is being actively carried out.
- the treatment that improves the disease by regenerating blood vessels is called therapeutic angiogenesis (therapeutic angiogenesis), and for diseases in which the number of blood vessels decreases or the blood vessel function deteriorates due to various causes, the blood vessels are regenerated.
- It is a treatment method for improving the pathological condition. For example, 1) a treatment method for inducing new blood vessels to myocardial ischemia, cerebral ischemia, lower limb ischemia, etc.
- ischemia that is, a treatment method for improving blood transport, which is the original function of blood vessels, and 2) normal Diseases in which factors secreted from vascular endothelial cells are suppressed by genetic factors or acquired factors, leading to disease (for example, production of coagulation factors in vascular endothelial cells)
- Treatment method for vascular regeneration for hemophilia, etc. that develops due to a decrease in blood vessels, etc. Blood vessel regeneration for renal dysfunction, etc.).
- cell therapy there is a method of regenerating blood vessels using vascular endothelial progenitor cells existing in bone marrow.
- progenitor cells are not stem cells, it is considered that the blood vessels formed by these progenitor cells are not maintained for a long period of time.
- mesenchymal stem cells such as adipose tissue to induce angiogenesis.
- This method is considered to be a method of regenerating blood vessels by proliferating vascular endothelial cells of existing blood vessels by a substance that induces angiogenesis secreted from mesenchymal stem cells.
- it cannot be used for patients with diminished ability to regenerate blood vessels.
- VEGF Vascular Endothelial Growth Factor
- HGF hepatocyte growth factor
- Non-Patent Documents 1 and 2). are cells that can contribute as vascular endothelial cells for a long period of time, unlike vascular endothelial progenitor cells existing in bone marrow.
- the present inventors have found that such cells having an extremely high ability to produce vascular endothelial cells exist in existing blood vessels as vascular endothelial cells having a high drug excretion ability.
- the present inventors isolated the surface marker expressed on the vascular endothelial cell. It was found that CD157 and CD200 are expressed in vascular endothelial stem cells (Non-Patent Document 2 and Patent Document 1). The CD200-positive CD157-positive vascular endothelial cell population was capable of maintaining undifferentiated state and dividing (self-replicating), and finally producing a large amount of CD200-negative CD157-negative differentiated vascular endothelial cells. ..
- vascular endothelial stem cells the blood vessels once regenerated by the vascular endothelial cells are maintained as blood vessels for a long period of time without retraction. Specifically, not only the blood-carrying function but also the long-term production function of molecules to be secreted from vascular endothelial cells (for example, coagulation factor VIII) was maintained.
- vascular endothelial stem cells the vascular endothelial cells having stem cell properties discovered by the present inventors will be referred to as "vascular endothelial stem cells”.
- vascular endothelial stem cells When inducing angiogenesis, it is necessary to regenerate blood vessels by vascular endothelial cells with such stem cell properties, and transplanting vascular endothelial stem cells has a therapeutic effect even on patients who do not have the reserve for revascularization. It can be a treatment to bring.
- vascular endothelial stem cells there are very few vascular endothelial stem cells in the living body, and it is very inefficient to collect the patient's own vascular endothelial stem cells for each patient who undergoes revascularization. It is also possible that the patient's vascular endothelial stem cell function is impaired. Therefore, it is desired to develop a method for artificially producing vascular endothelial stem cells from some cells.
- An object of the present invention is to provide a method for artificially producing vascular endothelial stem cells from cells other than vascular endothelial stem cells.
- a method for producing vascular endothelial stem cells which comprises a step of contacting a vascular endothelial cell having no stem cell property with a factor secreted from a mammalian organ in a neonatal period or an immature stage.
- the vascular endothelial cells having no stem cell property are cultured.
- [3] The production method according to the above [1] or [2], wherein the neonatal or juvenile mammal is a neonatal mammal.
- [4] The production method according to any one of [1] to [3] above, wherein the vascular endothelial cells having no stem cell property are cells obtained from a neonatal or juvenile mammal.
- [5] The production method according to the above [4], wherein the vascular endothelial cells having no stem cell property are cells obtained from a mammal in the neonatal period.
- [6] The production method according to any one of [1] to [5] above, wherein the organ is the liver.
- a vascular endothelial stem cell inducer containing a factor secreted from a neonatal or juvenile mammalian organ [8] A culture supernatant of cells prepared from a neonatal or juvenile mammalian organ containing the factor, or a neonatal or juvenile mammalian organ extract containing the factor. The vascular endothelial stem cell inducer according to the above [7]. [9] The vascular endothelial stem cell inducer according to the above [7] or [8], wherein the neonatal or juvenile mammal is a neonatal mammal. [10] The vascular endothelial stem cell inducer according to any one of [7] to [9] above, wherein the organ is the liver.
- FIG. 1 It is a figure which shows the result of culturing the vascular endothelial cell (CD31 positive CD45 negative cell) obtained from the living body of a mouse by the culture system using OP9 stromal cell as a feeder cell
- (A) is the embryonic day 15
- (B) is the result of vascular endothelial cells obtained from the liver of an 8-week-old adult mouse. Livers were removed from fetal mice on the 15th day of fetal life and neonatal to juvenile mice on the 1st, 7th, 14th, and 21st days after birth, and frozen sections were prepared using anti-CD31 antibody. It is a figure which shows the result of immunostaining.
- Vascular endothelial cells obtained by flow cytometry from the livers of fetal mice on the 15th day of embryonic development and neonatal to juvenile mice on the 7th, 14th, 21st, and 28th days after birth. It is a figure which shows the result of the flow cytometry analysis which performed Hoechst stain, (A) is the flow cytometry analysis result of Hoechst stain, and (B) is a fraction with high drug excretion ability (SP cell fraction). It is a graph showing the ratio of.
- (B) is the result of analyzing the presence of CD31-positive GFP-positive cells in the liver of (A) by flow cytometry
- (C) is the SP cells in the CD31-positive GFP-positive cells of (B).
- This is the result of analyzing the existence of.
- CD200-negative CD157-negative vascular endothelial cells obtained from the liver of green mice (C57BL / 6-Tg (CAG-EGFP)) on the 15th day of fetal life were transferred to the liver of mice on the 7th day after birth.
- A is the result of observing the opened liver with a fluorescent substance microscope
- B is the CD31 positive in the liver of (A). It is a result of analysis of the presence of CD200-positive CD157-positive cells in CD45-negative GFP-positive cells by flow cytometry.
- the present invention provides a method for producing vascular endothelial stem cells.
- the method for producing vascular endothelial stem cells of the present invention includes vascular endothelial cells having no stem cell property and factors secreted from neonatal or juvenile mammalian organs. It is characterized by including a step of contacting with.
- the neonatal or juvenile mammalian organ may be a neonatal mammalian organ.
- vascular endothelial stem cells mean cells having the ability to divide in an undifferentiated state (self-renewal ability) and the ability to differentiate into vascular endothelial cells.
- the fact that vascular endothelial stem cells were obtained by the production method of the present invention will be clarified by confirming the existence of vascular endothelial cells capable of forming colonies from a single cell. That is, it can be determined that the vascular endothelial cells having a high colony forming ability are vascular endothelial stem cells.
- the cells obtained by the production method of the present invention have at least one of the characteristics of being a cell having a high drug excretion ability and being a CD200-positive and CD157-positive cell. May be good.
- a Hoechst stain method or the like can be used for confirmation of the drug excretion ability.
- Whether or not the cells are CD200-positive and CD157-positive can be determined by staining the cells with an anti-CD200 antibody and an anti-CD157 antibody.
- Endothelial cells are single-layer flat cells that line the vascular lumen and can be identified as CD31-positive and CD45-negative cells.
- the vascular endothelial cells obtained as CD31-positive and CD45-negative cells by a conventional method can be suitably used as vascular endothelial cells having no stem cell property to be used in the production method of the present invention.
- the vascular endothelial cells that do not have stem cell properties may be mammalian vascular endothelial cells. Mammals are not particularly limited, and examples thereof include humans, monkeys, cows, pigs, sheep, goats, dogs, cats, mice, rats, and rabbits. When the mammal is a human, the vascular endothelial stem cells produced by the production method of the present invention can be safely used for human cell therapy and the like.
- vascular endothelial cells that do not have stem cell properties can be prepared from any organ.
- the organ may be, for example, liver, retina, brain, heart, skin, muscle (skeletal muscle), lung, kidney, placenta, umbilical cord, adipose tissue and the like.
- the liver and umbilical cord are preferable.
- the method for preparing vascular endothelial cells having no stem cell property is not particularly limited.
- an isolated organ is digested and dispersed with a commercially available cell dispersion reagent to prepare a cell suspension, and this cell suspension is prepared. Examples thereof include a method of recovering CD31-positive and CD45-negative cells by using flow cytometry technique after staining with anti-CD31 antibody and anti-CD45 antibody.
- the vascular endothelial cells that do not have stem cell properties may be vascular endothelial cells acquired from mammals, and the acquisition time is not particularly limited. It may be a vascular endothelial cell obtained from a neonatal or juvenile mammal, preferably a vascular endothelial cell obtained from a neonatal mammal.
- the neonatal period means from birth to weaning
- the infancy means from weaning to the acquisition of fertility.
- neonatal vascular endothelial cells are more likely to be reprogrammed into vascular endothelial stem cells than adult mouse vascular endothelial cells.
- Non-human mammals are not particularly limited, and examples thereof include monkeys, cows, pigs, sheep, goats, dogs, cats, mice, rats, and rabbits. Preferably, it is a pig or a sheep.
- the organ is not particularly limited, and examples thereof include liver, retina, brain, heart, skin, muscle (skeletal muscle), lung, kidney, placenta, and adipose tissue. The liver is preferred. Transplantation can be performed, for example, by laparotomy of a host animal under anesthesia and inoculating a target organ with vascular endothelial cells having no stem cell property by injection or the like.
- the step of contacting non-stem cell vascular endothelial cells with factors secreted by neonatal or juvenile mammalian organs is, for example, secreted from neonatal or juvenile mammalian organs. It may be carried out by culturing vascular endothelial cells having no stem cell property using a culture system containing a factor. Specifically, cells prepared from neonatal or juvenile mammalian organs may be co-cultured with non-stem cell vascular endothelial cells. Examples of the preparation of cells from an organ include a method of digesting and dispersing an isolated organ with a commercially available cell dispersion reagent.
- vascular endothelial cells having no stem cell property and cells prepared from organs may be cultured in the same culture vessel.
- a device such as a culture insert may be attached to the culture vessel for co-culture so that the vascular endothelial cells having no stem cell property and the cells prepared from the organ do not come into direct contact with each other.
- cells prepared from neonatal or juvenile mammalian organs may be cultured in an appropriate medium, and the culture supernatant may be collected and added to a medium of vascular endothelial cells having no stem cell property.
- an organ extract of a neonatal or juvenile mammal may be prepared and the extract may be added to a medium of vascular endothelial cells having no stem cell property.
- an organ extract for neonatal or juvenile mammals for example, an organ homogenate supernatant can be preferably used.
- the period of contact between non-stem cell vascular endothelial cells and factors secreted from neonatal or juvenile mammalian organs is not particularly limited, and an appropriate period for obtaining the desired vascular endothelial stem cells is set. , It may be set by preliminary examination. For example, when transplanting into a neonatal or juvenile non-human mammalian organ, it may be 1 day or more, 2 days or more, 3 days or more, 4 days or more, 100 days or less, 50 days or less, 10 days or less. It may be less than a day and less than 5 days.
- the co-cultured with cells prepared from neonatal or juvenile non-human mammalian organs it may be 1 day or more, 2 days or more, 3 days or more, 4 days or more, 100 days or less, It may be 50 days or less, 10 days or less, and 5 days or less.
- non-stem cell vascular endothelial cells By contacting non-stem cell vascular endothelial cells with factors secreted from neonatal or juvenile mammalian organs for a period of time, non-stem cell vascular endothelial cells are reprogrammed and vascular endothelial stem cells. Changes to.
- a cell suspension When manufactured by a method of transplantation into a non-human mammalian organ, a cell suspension is prepared by excising the transplantation site and digesting / dispersing with a commercially available cell dispersion reagent, and has a high colony-forming ability.
- vascular endothelial cells CD31-positive and CD45-negative cells
- Endothelial endothelial cells (CD31-positive, CD45-negative cells) with high colony-forming ability in the cells after culture when produced by a method using a culture system containing factors secreted from neonatal or juvenile mammalian organs. ) Is present, it can be determined that the vascular endothelial stem cells have been produced. Furthermore, it may be confirmed that the vascular endothelial cells having a high colony forming ability are cells having a high drug excretion ability and / or CD200-positive and CD157-positive cells. The obtained vascular endothelial stem cells can be recovered, for example, by isolating and proliferating the cells that have formed colonies. In addition, cells having high drug excretion ability or CD200-positive CD157-positive cells may be used as an index for recovery.
- the present invention provides vascular endothelial stem cell inducers containing factors secreted by neonatal or juvenile mammalian organs.
- the neonatal or juvenile mammalian organ may be a neonatal mammalian organ.
- Mammals are not particularly limited, and examples thereof include humans, monkeys, cows, pigs, sheep, goats, dogs, cats, mice, rats, and rabbits.
- the mammal may be human.
- the organ is not particularly limited, and examples thereof include liver, retina, brain, heart, skin, muscle (skeletal muscle), lung, kidney, placenta, and fat.
- the organ may be the liver.
- the vascular endothelial stem cell inducer of the present invention may be in any form as long as it contains a factor secreted from a neonatal or juvenile mammalian organ.
- the vascular endothelial stem cell inducer of the present invention may contain, for example, a culture supernatant of cells prepared from a neonatal or juvenile mammalian organ, and is a neonatal or juvenile mammal. It may contain the organ extract of.
- Such culture supernatants and organ extracts contain factors secreted by the organs.
- the culture supernatant can be obtained, for example, by culturing the cells obtained by digesting and dispersing the isolated organ with a commercially available cell dispersion reagent using an appropriate medium.
- the organ extract can be obtained, for example, by homogenizing and centrifuging the organ.
- the vascular endothelial stem cell inducer of the present invention can be used by adding it to a medium when culturing vascular endothelial cells having no stem cell property. It is recommended that the amount to be added be appropriately set according to the culture conditions and the like.
- the present invention includes the following inventions.
- a method for inducing vascular endothelial stem cells which comprises the step of culturing vascular endothelial cells having no stem cell property using a medium containing a factor secreted from a neonatal or juvenile mammalian organ.
- Use of factors secreted by neonatal or juvenile mammalian organs to induce vascular endothelial stem cells Use of culture supernatants of cells prepared from neonatal or juvenile mammalian organs to induce vascular endothelial stem cells.
- vascular endothelial stem cell Use of factors secreted by neonatal or juvenile mammalian organs to produce vascular endothelial stem cell inducers. Use of culture supernatants of cells prepared from neonatal or juvenile mammalian organs to produce vascular endothelial stem cell inducers. Use of neonatal or juvenile mammalian organ extracts to produce vascular endothelial stem cell inducers.
- Example 1 Examination of stem cell properties in fetal and postnatal vascular endothelial cells
- E15 15th day of fetal life
- 8W 8th week after birth
- E15 fetal whole foetation
- 8W mouse excised liver
- chopped as much as possible with ophthalmic scissors chopped tissue Dispase II (Roche Applied Science)
- collagenase Wako
- type II The extracellular matrix was digested by immersing in a mixed solution of collagenase (manufactured by Worthington Biochemical) and shaking at 37 ° C.
- the solution containing the digested cells was passed through a filter having a pore size of 40 ⁇ m to obtain a dispersed cell suspension.
- Erythrocytes were hemolyzed with ACK (Ammonium-Chloride-Potassium) solution (0.15M NH 4 Cl, 10mM KHCO 3 , and 0.1mM Na 2- EDTA), and the remaining cells were subjected to the following experiments.
- ACK Ammonium-Chloride-Potassium
- the prepared cells were immunofluorescently stained and flow cytometric analysis was performed.
- an anti-CD31 antibody (clone MEC13.3, manufactured by BD Biosciences) and an anti-CD45 antibody (Clone 30-F11, manufactured by BD Biosciences) were used.
- Propidium iodide (PI, 2 ⁇ g / mL, manufactured by Sigma-Aldrich) was added to the stained cells, and the nuclei of dead cells were stained to remove dead cells.
- SOAP FACSAria manufactured by BD Bioscience
- FlowJo Software (manufactured by Treestar Software) were used for flow cytometry analysis.
- CD31-positive and CD45-negative vascular endothelial cells obtained by flow cytometry were seeded in a culture system using OP9 stromal cells (RIKEN cell bank) as feeder cells, and 5000 cells were seeded on a culture plate with a diameter of 3 cm for 7 days. It was cultured.
- Fig. 1 The results are shown in Fig. 1.
- A is the result of culturing vascular endothelial cells obtained from fetal mice
- B is the result of culturing vascular endothelial cells obtained from the liver of adult mice.
- the vascular endothelial cells obtained from the foetation contained a small number of cells capable of proliferating and forming colonies.
- the vascular endothelial cells collected from the adult liver contain vascular endothelial cells that form very large colonies. From this result, it was considered that vascular endothelial stem cells do not exist in the fetal period and may develop after birth.
- Example 2 Examination of formation of sinusoidal blood vessels in the liver
- Liver of C57BL / 6 mice (Japan SLC) from fetal mice on the 15th day of fetal life and neonatal to juvenile mice on the 1st, 7th, 14th and 21st days after birth, respectively. It was excised and frozen sections of the liver were prepared according to a conventional method. Frozen sections were immunostained with an anti-CD31 antibody (clone MEC13.3, manufactured by BD Biosciences). PE-labeled anti-rat IgG (manufactured by BD Biosciences) was used as the secondary antibody.
- Fig. 2 Results The results are shown in Fig. 2.
- sinusoidal blood vessels which are vascular structures peculiar to the liver, are induced by vascular endothelial stem cells (Wakabayashi T. et al., Cell Stem Cell. 2018 Mar 1; 22 (3): 384-397), vascular endothelial stem cells were absent during the fetal period and may occur after birth.
- Example 3 Analysis of development mode of fraction (SP cell fraction) containing vascular endothelial stem cells that can be isolated by utilizing high drug excretion ability]
- 3-1 Experimental method Fetal mice on the 15th day of fetal life (E15) of C57BL / 6 mice (Nippon SLC), 1st day (P1), 7th day (P7), 14th day (P14), 21 after birth Livers were removed from neonatal to juvenile mouse mice on day (P21) and day 28 (P28), and adult mice at week 8 (8W), respectively.
- the excised liver is chopped as much as possible using ophthalmic scissors, and the chopped tissue is Dispase II (Roche Applied Science), collagenase (Wako) and type II collagenase (Worthington).
- the extracellular matrix was digested by immersing it in a mixed solution (manufactured by Biochemical) and shaking it at 37 ° C.
- the solution containing the digested cells was passed through a filter having a pore size of 40 ⁇ m to obtain a dispersed cell suspension.
- the obtained cell suspension was stained with Hoechst.
- Hoechst stain is performed on 1 ⁇ 10 6 cells in 1 ml of Hoechst-containing medium (2% FBS (Sigma-Aldrich), 1 mM HEPES (Gibco), 5 ⁇ g / mL Hoechst33342 (Sigma-Aldrich) -containing DMEM (Sigma-Aldrich)). ), And performed at 37 ° C. for 90 minutes.
- the same anti-CD31 antibody and anti-CD45 antibody as in Example 1 were used for immunofluorescent staining.
- Propidium iodide PI, 2 ⁇ g / mL, manufactured by Sigma-Aldrich
- FIG. 3 (A) is a graph showing typical flow cytometry analysis results of fetal period and newborn to juvenile period, and (B) is a graph showing the ratio of SP cell fraction.
- SP cells SP cell fraction cells
- MP cells MP cell fraction cells
- Example 4 MP cells are transformed into SP cells in the liver environment
- 4-1 Experimental method C57BL / 6 mice and C57BL / 6-Tg (CAG-EGFP) mice (hereinafter referred to as "green mice") were purchased from Japan SLC and used. Livers were removed from green mice 1 day (1 day old) and 8 weeks (8 weeks old) after birth, and cell suspensions were prepared in the same manner as in Example 3. Hoechst staining and immunofluorescent staining (anti-CD31 antibody, anti-CD45 antibody, PI) were performed in the same manner as in Example 3, and MP cells of vascular endothelial cells were collected by flow cytometry. The recovered MP cells (1 ⁇ 10 5 cells) were transplanted into liver injury model mice.
- mice were intraperitoneally administered with monocotalin (Sigma-Aldrich) at a dose of 300 mg / kg to C57BL / 6 mice 7 days (7 days old) and 8 weeks (8 weeks old) after birth. Then, on the same day, it was prepared by total body irradiation of 30 rads / g. Two months after the MP cell transplantation, the mice were laparotomized under anesthesia, and the liver was observed with a fluorescent stereomicroscope (manufactured by Leica).
- monocotalin Sigma-Aldrich
- liver was excised, a cell suspension was prepared by the same method as in Example 3, Hoechst staining and immunofluorescent staining (anti-CD31 antibody, anti-CD45 antibody, PI) were performed, and SP cells were subjected to flow cytometry. The existence was analyzed.
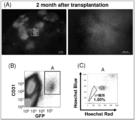
- FIG. 4-2 Results Figure 4 shows the results of transplantation of MP cells from 1-day-old green mouse liver into 7-day-old liver injury model mice.
- A is the result of observing the liver of a liver disorder model mouse 2 months after transplantation
- B is the result of analyzing the presence of CD31-positive GFP-positive cells in the mouse liver of (A) by flow cytometry.
- C are the results of analysis of the presence of SP cells in the CD31-positive GFP-positive cells of (B) by flow cytometry.
- GFP-positive cells derived from green mice transplanted into the liver of a liver disorder model mouse were observed.
- Example 5 Analysis of developmental process of CD157-positive and CD200-positive vascular endothelial stem cells
- 5-1 Experimental method Fetal mice on the 13th day (E13), 15th day (E15), and 18th day (E18) of C57BL / 6 mice (Nippon SLC), 1st day after birth (P1), 4 The livers were removed from the neonatal mice on the day (P4), the 7th day (P7), the 14th day (P14), and the adult mice on the 8th week (8W), respectively, and the cells were suspended in the same manner as in Examples 1 and 3. A turbid solution was prepared.
- Anti-CD31 antibody (clone MEC13.3, manufactured by BD Biosciences), anti-CD45 antibody (Clone 30-F11, manufactured by BD Biosciences), anti-CD157 antibody (clone BP3, manufactured by Biolegend), anti-CD200 antibody (clone) as monoclonal antibodies OX90, manufactured by Biolegend) was used.
- Propidium iodide (PI, 2 ⁇ g / mL, manufactured by Sigma-Aldrich) was added to the stained cells, and the nuclei of dead cells were stained to remove dead cells.
- SOAP FACSAria manufactured by BD Bioscience
- FlowJo Software (manufactured by Treestar Software) were used for flow cytometry analysis.
- CD200-positive CD157-positive cells At P4, a few CD200-positive CD157-positive cells appear, at P7 and P14, CD200-positive CD157-positive cells gradually increase, and in 8W adult mice, 4 to 6% of all vascular endothelial cells are CD200-positive CD157-positive cells. It became. Although no data are shown, vascular endothelial stem cells of CD200-positive and CD157-positive cells were present in the liver at about the same rate thereafter. This result was the same as that of vascular endothelial stem cells in SP cells by the Hoechst exclusion method of Example 3.
- vascular endothelial stem cells there are no vascular endothelial stem cells in the fetal liver, and some vascular endothelial progenitor cells that have some stem cell characteristics immediately before birth but are more differentiated than stem cells appear, and in the liver after birth. It became clear that vascular endothelial stem cells were generated in.
- Example 6 CD157-negative CD200-negative cells Vascular endothelial cells are transformed into vascular endothelial stem cells in the neonatal hepatic environment.
- 6-1 Experimental method On day 15 of embryonic development (E15), the liver of a green mouse was removed, a cell suspension was prepared by the same method as in Example 3, and the same anti-CD31 antibody, anti-CD45 antibody, and anti-CD157 as in Example 5 were prepared. The cells were stained with an antibody and an anti-CD200 antibody and subjected to flow cytometry, and CD200-negative CD157-negative vascular endothelial cells (CD31-positive CD45-negative cells) were collected.
- the collected cells were transplanted into the liver of a liver disorder model mouse (C57BL / 6) 7 days after birth (7 days old) in the same manner as in Example 4.
- the liver disorder model mouse was prepared by the same method as in Example 4. Two months after the transplantation, the mice were opened under anesthesia, and the liver was observed with a fluorescent stereomicroscope (manufactured by Leica). Further, the liver was excised, a cell suspension was prepared by the same method as in Example 3, and stained with the same anti-CD31 antibody, anti-CD45 antibody, and anti-CD157 antibody as in Example 5, and flow cytometric analysis was performed.
- Example 7 Change from human vascular endothelial cells to vascular endothelial stem cells
- HUVEC Human umbilical vein endothelial cells
- HuMedia-EG2 HuMedia-EG2 (KURABO, Osaka, Japan) culture medium.
- HUVEC was sown on a 6-well plate at 1.0 ⁇ 10 5 pcs / well and culture inserts (Corning, NY, USA) with a pore size of 1.0 ⁇ m were set in the wells.
- the liver of a newborn mouse (C57BL / 6) 4 days after birth was excised, and a cell suspension was prepared by the same method as in Example 1.
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biomedical Technology (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Vascular Medicine (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Developmental Biology & Embryology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Description
[1]幹細胞性を有しない血管内皮細胞と、新生児期または幼若期の哺乳動物の臓器から分泌される因子とを接触させる工程を含むことを特徴とする、血管内皮幹細胞の製造方法。
[2]前記工程が、以下の(1)または(2)のいずれかにより行われることを特徴とする、前記[1]に記載の製造方法:
(1)前記幹細胞性を有しない血管内皮細胞を非ヒト哺乳動物の臓器に移植する、または
(2)新生児期または幼若期の哺乳動物の臓器から分泌される因子を含む培養系を用いて前記幹細胞性を有しない血管内皮細胞を培養する。
[3]前記新生児期または幼若期の哺乳動物が、新生児期の哺乳動物である、前記[1]または[2]に記載の製造方法。
[4]前記幹細胞性を有しない血管内皮細胞が、新生児期または幼若期の哺乳動物から取得した細胞である、前記[1]~[3]のいずれかに記載の製造方法。
[5]前記幹細胞性を有しない血管内皮細胞が、新生児期の哺乳動物から取得した細胞である、前記[4]に記載の製造方法。
[6]前記臓器が肝臓である、前記[1]~[5]のいずれかに記載の製造方法。
[7]新生児期または幼若期の哺乳動物の臓器から分泌される因子を含有する、血管内皮幹細胞誘導剤。
[8]前記因子を含む新生児期もしくは幼若期の哺乳動物の臓器から調製した細胞の培養上清、または、前記因子を含む新生児期もしくは幼若期の哺乳動物の臓器抽出液を含有する、前記[7]に記載の血管内皮幹細胞誘導剤。
[9]前記新生児期または幼若期の哺乳動物が、新生児期の哺乳動物である、前記[7]または[8]に記載の血管内皮幹細胞誘導剤。
[10]前記臓器が肝臓である、前記[7]~[9]のいずれかに記載の血管内皮幹細胞誘導剤。
本発明は、血管内皮幹細胞の製造方法を提供する。本発明の血管内皮幹細胞の製造方法(以下、「本発明の製造方法」と記す)は、幹細胞性を有しない血管内皮細胞と、新生児期または幼若期の哺乳動物の臓器から分泌される因子とを接触させる工程を含むことを特徴とする。新生児期または幼若期の哺乳動物の臓器は、新生児期の哺乳動物の臓器であってもよい。
本発明は、新生児期または幼若期の哺乳動物の臓器から分泌される因子を含有する血管内皮幹細胞誘導剤を提供する。新生児期または幼若期の哺乳動物の臓器は、新生児期の哺乳動物の臓器であってもよい。
新生児期または幼若期の哺乳動物の臓器から分泌される因子を含む培地を用いて、幹細胞性を有しない血管内皮細胞を培養する工程を含む、血管内皮幹細胞の誘導方法。
血管内皮幹細胞を誘導するための、新生児期または幼若期の哺乳動物の臓器から分泌される因子の使用。
血管内皮幹細胞を誘導するための、新生児期または幼若期の哺乳動物の臓器から調製した細胞の培養上清の使用。
血管内皮幹細胞を誘導するための、新生児期または幼若期の哺乳動物の臓器抽出液の使用。
血管内皮幹細胞誘導剤を製造するための、新生児期または幼若期の哺乳動物の臓器から分泌される因子の使用。
血管内皮幹細胞誘導剤を製造するための、新生児期または幼若期の哺乳動物の臓器から調製した細胞の培養上清の使用。
血管内皮幹細胞誘導剤を製造するための、新生児期または幼若期の哺乳動物の臓器抽出液の使用。
1-1 実験方法
C57BL/6マウス(日本エスエルシー)の胎生15日目(E15)の胎児マウスと、出生後8週目(8W)の成体マウスを用いた。E15胎児は全胎児を、8Wマウスは摘出した肝臓を、眼科用はさみを用いてできる限り細切し、細切組織をDispase II(Roche Applied Science社製)、collagenase(Wako社製)およびtype II collagenase(Worthington Biochemical社製)の混合溶液に浸漬し、37℃で振盪して細胞外マトリックスを消化した。消化後の細胞を含む溶液を孔径40μmのフィルターに通し、分散した細胞懸濁液を得た。ACK(Ammonium-Chloride-Potassium)溶液(0.15M NH4Cl, 10mM KHCO3, and 0.1mM Na2-EDTA)で赤血球を溶血させ、残りの細胞を以下の実験に供した。
結果を図1に示した。(A)は胎児マウスから得られた血管内皮細胞を培養した結果、(B)は成体マウスの肝臓から得られた血管内皮細胞を培養した結果である。胎児から得られた血管内皮細胞には、増殖してコロニーを形成する能力がある細胞が少数存在していた。一方、成体肝臓から回収した血管内皮細胞には、非常に大きなコロニーを形成する血管内皮細胞が存在することが判明した。この結果から、血管内皮幹細胞は胎児期には存在せず、出生後に発生する可能性が考えられた。
2-1 実験方法
C57BL/6マウス(日本エスエルシー)の胎生15日目の胎児マウス、出生後1日目、7日目、14日目および21日目の新生児~幼若マウスからそれぞれ肝臓を摘出し、定法に従い肝臓の凍結切片を作製した。凍結切片に抗CD31抗体(clone MEC13.3, BD Biosciences社製)を用いて免疫染色を行った。2次抗体としてPEラベルした抗ラットIgG(BD Biosciences社製)を用いた。
結果を図2に示した。出生後21日目の幼若マウスの肝臓では、中心静脈を中心にして、そこから放射状に類洞血管が形成されていた。この放射状に形成される肝臓特異的な血管構造は、胎児期のマウスの肝臓内では形成されておらず、出生後に徐々に形成されてくることが判明した。本発明者らは、肝臓に特有の血管構造である類洞血管は、血管内皮幹細胞により誘導されることを解明していることから(Wakabayashi T. et al., Cell Stem Cell. 2018 Mar 1;22(3):384-397)、血管内皮幹細胞は胎児期には存在せず、出生後に発生する可能性が考えられた。
3-1 実験方法
C57BL/6マウス(日本エスエルシー)の胎生15日目(E15)の胎児マウス、出生後1日目(P1)、7日目(P7)、14日目(P14)、21日目(P21)、28日目(P28)の新生児~幼若マウスマウス、および8週目(8W)の成体マウスから、それぞれ肝臓を摘出した。実施例1と同様に、摘出した肝臓を、眼科用はさみを用いてできる限り細切し、細切組織をDispase II(Roche Applied Science社製)、collagenase(Wako社製)およびtype II collagenase(Worthington Biochemical社製)の混合溶液に浸漬し、37℃で振盪して細胞外マトリックスを消化した。消化後の細胞を含む溶液を孔径40μmのフィルターに通し、分散した細胞懸濁液を得た。得られた細胞懸濁液にヘキスト染色を行った。ヘキスト染色は、1×106個の細胞を、1mlのヘキスト含有培地(2% FBS (Sigma-Aldrich), 1mM HEPES (Gibco), 5μg/mL Hoechst33342 (Sigma-Aldrich)含有DMEM (Sigma-Aldrich))に懸濁して、37℃で90分間行った。免疫蛍光染色には、実施例1と同じ抗CD31抗体および抗CD45抗体を使用した。染色した細胞にPropidium iodide(PI, 2μg/mL, Sigma-Aldrich社製)を加え、死細胞の核を染色して死細胞を除去した。フローサイトメトリーにより、CD31陽性CD45陰性PI陰性細胞(死細胞を除去した血管内皮細胞)におけるヘキスト染色解析を行った。フローサイトメトリー解析には、SOAP FACSAria(BD Bioscience社製)およびFlowJo Software(Treestar Software社製)を使用した。
結果を図3に示した。(A)は胎児期、新生児~幼若期の代表的なフローサイトトリー解析結果を示す図であり、(B)はSP細胞画分の割合を表したグラフである。胎児期の血管内皮細胞には血管内皮幹細胞を含むSP細胞画分の細胞(SP細胞)が観察されず、非血管内皮幹細胞であるMP細胞画分の細胞(MP細胞)のみが観察された。SP細胞は出生後に徐々に出現し、21日齢(P21)の幼若マウスでは、SP細胞の割合が8週齢(8W)の成体マウスに匹敵する約1%程度になることが判明した。この結果から、血管内皮幹細胞は、出生後の体内環境中で発生することが示唆された。
4-1 実験方法
C57BL/6マウスおよびC57BL/6-Tg(CAG-EGFP)マウス(以下「グリーンマウス」と記す)を日本エスエルシーから購入して使用した。出生後1日目(1日齢)および8週目(8週齢)のグリーンマウスから肝臓を摘出し、実施例3と同じ方法で細胞懸濁液を調製した。実施例3と同じ方法でヘキスト染色および免疫蛍光染色(抗CD31抗体、抗CD45抗体、PI)を行い、フローサイトメトリーにより、血管内皮細胞のMP細胞を回収した。回収したMP細胞(1×105個)を肝障害モデルマウスに移植した。肝血管障害モデルマウスは、出生後7日(7日齢)および8週(8週齢)のC57BL/6マウスに、モノクロタリン(Sigma-Aldrich 社製)を300mg/kgの用量で腹腔内投与し、同日に30rads/gの放射線を全身照射して作製した。MP細胞の移植から2か月後に、麻酔下でマウスを開腹し、肝臓を蛍光実体顕微鏡(Leica社製)で観察した。さらに、肝臓を摘出して実施例3と同じ方法で細胞懸濁液を調製し、ヘキスト染色および免疫蛍光染色(抗CD31抗体、抗CD45抗体、PI)を行い、フローサイトメトリーにより、SP細胞の存在を解析した。
1日齢のグリーンマウス肝臓のMP細胞を7日齢の肝臓障害モデルマウスに移植した結果を図4に示した。(A)は移植2ヶ月後の肝臓障害モデルマウスの肝臓を観察した結果であり、(B)は(A)のマウス肝臓におけるCD31陽性GFP陽性細胞の存在をフローサイトメトリーにより解析した結果であり、(C)は(B)のCD31陽性GFP陽性細胞におけるSP細胞の存在をフローサイトメトリーにより解析した結果である。(A)に示されるように、肝臓障害モデルマウスの肝臓中に移植されたグリーンマウス由来のGFP陽性細胞が観察された。また、(B)に示されるように、多数のCD31陽性GFP陽性細胞が検出されたことから、グリーンマウスの血管内皮細胞がホストの肝臓で血管を形成していることが判明した。さらに、(C)に示されるように、CD31陽性GFP陽性細胞中にSP細胞が1.5%存在することが判明した。この結果は、MP細胞は適切な環境下でSP細胞にリプログラミングできることを示す。
5-1 実験方法
C57BL/6マウス(日本エスエルシー)の胎生13日目(E13)、15日目(E15)、18日目(E18)の胎児マウス、出生後1日目(P1)、4日目(P4)、7日目(P7)、14日目(P14)の新生児マウス、および8週目(8W)の成体マウスからそれぞれ肝臓を摘出し、実施例1、3と同様に細胞懸濁液を調製した。モノクローナル抗体として抗CD31抗体(clone MEC13.3, BD Biosciences社製)、抗CD45抗体(Clone 30-F11, BD Biosciences社製)、抗CD157抗体(clone BP3, Biolegend社製)、抗CD200抗体(clone OX90, Biolegend社製)を使用した。染色した細胞にPropidium iodide(PI, 2μg/mL, Sigma-Aldrich社製)を加え、死細胞の核を染色して死細胞を除去した。フローサイトメトリー解析には、SOAP FACSAria(BD Bioscience社製)およびFlowJo Software(Treestar Software社製)を使用した。
結果を図5に示した。肝臓内のCD31陽性CD45陰性細胞を肝臓の血管内皮細胞としてゲートし、CD200およびCD157の発現をフローサイトメトリーで解析すると、E15までは血管内皮細胞はすべてCD200陰性CD157陰性の細胞であるのに対して、E18でわずかにCD200陽性CD157陰性細胞が出現した。出生後になると、P1でCD200陽性CD157陰性細胞集団がCD200陰性CD157陰性細胞集団ときれいに分画され始めたが、CD200陽性CD157陽性細胞集団はほとんど現れていない。P4になるとCD200陽性CD157陽性細胞がわずかに現れ、P7、P14ではCD200陽性CD157陽性細胞が徐々に増加し、8Wの成体マウスでは全体の血管内皮細胞中の4~6%がCD200陽性CD157陽性細胞となった。データを示していないが、それ以降はほぼ同じ割合でCD200陽性CD157陽性細胞の血管内皮幹細胞が肝臓内に存在した。この結果は、実施例3のヘキスト排除法によるSP細胞中の血管内皮幹細胞と同じであった。すなわち、胎児期の肝臓内には血管内皮幹細胞は存在せず、出生直前に幹細胞性を一部持ち合わせているが幹細胞より分化の進んだ血管内皮前駆細胞が一部出現し、出生後の肝臓内で血管内皮幹細胞が発生してくることが明らかになった。
6-1 実験方法
胎生15日目(E15)グリーンマウスの肝臓を摘出し、実施例3と同じ方法で細胞懸濁液を調製し、実施例5と同じ抗CD31抗体、抗CD45抗体、抗CD157抗体、抗CD200抗体で染色してフローサイトメトリーに供し、CD200陰性CD157陰性の血管内皮細胞(CD31陽性CD45陰性細胞)を回収した。回収した細胞を実施例4と同様に、出生後7日目(7日齢)の肝障害モデルマウス(C57BL/6)の肝臓に移植した。肝障害モデルマウスは、実施例4と同じ方法で作製した。移植から2か月後に麻酔下でマウスを開腹し、肝臓を蛍光実体顕微鏡(Leica社製)で観察した。さらに、肝臓を摘出して実施例3と同じ方法で細胞懸濁液を調製し、実施例5と同じ抗CD31抗体、抗CD45抗体、抗CD157抗体で染色してフローサイトメトリー解析を行った。
結果を図6に示した。(A)は移植2ヶ月後のホストマウスの肝臓を観察した結果であり、(B)は(A)のホストマウス肝臓におけるCD31陽性CD45陰性GFP陽性細胞中のCD200陽性CD157陽性細胞の存在をフローサイトメトリーにより解析した結果である。(A)に示されるように、ホストマウスの肝臓中に、グリーンマウス由来のGFP陽性細胞が多数存在することが観察され、グリーンマウス胎児のCD200陰性CD157陰性の血管内皮細胞(CD31陽性CD45陰性GFP陽性細胞)は、新生児の肝臓環境下で類洞血管への貢献性を獲得することが判明した。また、(B)に示されるように、移植されたグリーンマウス胎児のCD200陰性CD157陰性血管内皮細胞の一部が、CD200陽性CD157陽性の血管内皮幹細胞にリプログラミングしていることが判明した。
7-1 実験方法
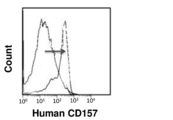
出生直後のヒトの血管内皮細胞として、ヒト臍帯静脈血管内皮細胞(human umbilical vein endothelial cells; HUVEC)(ロンザ社)を使用した。HUVECはHuMedia-EG2(KURABO, Osaka, Japan)培養液を用いて培養した。HUVECを6ウェルプレートに1.0×105個/ウェルで播種し、ポアサイズ1.0μmのカルチャーインサート(Corning, NY, USA)をウェルにセットした。出生後4日目の新生児マウス(C57BL/6)の肝臓を摘出し、実施例1と同じ方法で細胞懸濁液を調製した。細胞懸濁液中の細胞2.0×106個をカルチャーインサートに播種し、HUVECと新生児マウス由来細胞が直接接触しない状態で5日間共培養した。共培養後のHUVECを回収し、CD157抗体で染色した後にフローサイトメトリーで解析した。
結果を図7に示した。共培養前のHUVECはCD157陰性であったが、新生児マウス肝臓由来の細胞との共培養により、CD157の発現が陽性に誘導された。この結果から、新生児の肝臓内の細胞には、CD157を誘導し、内皮細胞から内皮幹細胞に分化誘導する能力があることが実証され、また肝臓から分泌される液性因子がこの分化転換に関わっていることが示唆された。
Claims (10)
- 幹細胞性を有しない血管内皮細胞と、新生児期または幼若期の哺乳動物の臓器から分泌される因子とを接触させる工程を含むことを特徴とする、血管内皮幹細胞の製造方法。
- 前記工程が、以下の(1)または(2)のいずれかにより行われることを特徴とする、請求項1に記載の製造方法:
(1)前記幹細胞性を有しない血管内皮細胞を非ヒト哺乳動物の臓器に移植する、または
(2)新生児期または幼若期の哺乳動物の臓器から分泌される因子を含む培養系を用いて前記幹細胞性を有しない血管内皮細胞を培養する。 - 前記新生児期または幼若期の哺乳動物が、新生児期の哺乳動物である、請求項1または2に記載の製造方法。
- 前記幹細胞性を有しない血管内皮細胞が、新生児期または幼若期の哺乳動物から取得した細胞である、請求項1~3のいずれかに記載の製造方法。
- 前記幹細胞性を有しない血管内皮細胞が、新生児期の哺乳動物から取得した細胞である、請求項4に記載の製造方法。
- 前記臓器が肝臓である、請求項1~5のいずれかに記載の製造方法。
- 新生児期または幼若期の哺乳動物の臓器から分泌される因子を含有する、血管内皮幹細胞誘導剤。
- 前記因子を含む新生児期もしくは幼若期の哺乳動物の臓器から調製した細胞の培養上清、または、前記因子を含む新生児期もしくは幼若期の哺乳動物の臓器抽出液を含有する、請求項7に記載の血管内皮幹細胞誘導剤。
- 前記新生児期または幼若期の哺乳動物が、新生児期の哺乳動物である、請求項7または8に記載の血管内皮幹細胞誘導剤。
- 前記臓器が肝臓である、請求項7~9のいずれかに記載の血管内皮幹細胞誘導剤。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP20875184.2A EP4043554A4 (en) | 2019-10-09 | 2020-10-07 | METHOD FOR PRODUCING A VASCULAR ENDOTHELIAL STEM CELL |
| CN202080061207.3A CN114341347A (zh) | 2019-10-09 | 2020-10-07 | 血管内皮干细胞的制造方法 |
| US17/638,026 US20220275343A1 (en) | 2019-10-09 | 2020-10-07 | Method for producing vascular endothelial stem cell |
| CA3151911A CA3151911A1 (en) | 2019-10-09 | 2020-10-07 | Method for producing vascular endothelial stem cell |
| JP2021551691A JPWO2021070874A1 (ja) | 2019-10-09 | 2020-10-07 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019186035 | 2019-10-09 | ||
| JP2019-186035 | 2019-10-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021070874A1 true WO2021070874A1 (ja) | 2021-04-15 |
Family
ID=75437220
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2020/038029 WO2021070874A1 (ja) | 2019-10-09 | 2020-10-07 | 血管内皮幹細胞の製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20220275343A1 (ja) |
| EP (1) | EP4043554A4 (ja) |
| JP (1) | JPWO2021070874A1 (ja) |
| CN (1) | CN114341347A (ja) |
| CA (1) | CA3151911A1 (ja) |
| WO (1) | WO2021070874A1 (ja) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008066630A2 (en) * | 2006-10-27 | 2008-06-05 | Caritas St. Elizabeth Medical Center Of Boston, Inc. | Methods for reprogramming adult somatic cells and uses thereof |
| JP2008528059A (ja) * | 2005-02-02 | 2008-07-31 | プライムジェン バイオテック エルエルシー | 分化細胞を治療用に再プログラム化するための核の材料の使用 |
| WO2019098264A1 (ja) | 2017-11-17 | 2019-05-23 | 国立大学法人大阪大学 | Cd31陽性cd45陰性cd200陽性の哺乳動物細胞からなる細胞集団、およびその利用 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11326150B2 (en) * | 2011-09-27 | 2022-05-10 | Public University Corporation Yokohama City University | Method for producing tissue and organ |
| CN103860597A (zh) * | 2014-03-03 | 2014-06-18 | 奥思达干细胞有限公司 | 一种治疗缺血性心肌病的干细胞制剂及其制备方法 |
| WO2017014165A1 (ja) * | 2015-07-17 | 2017-01-26 | 国立大学法人京都大学 | 血管内皮細胞の誘導方法 |
| BR112018012070A2 (ja) * | 2015-12-24 | 2018-11-27 | Public University Corporation Yokohama City University | A manufacturing method of an organization and internal organs using blood cells |
| CN108251352B (zh) * | 2018-02-01 | 2021-07-06 | 伯仕利生物科技发展(盐城)有限公司 | 一种血管内皮干细胞的培养方法 |
-
2020
- 2020-10-07 WO PCT/JP2020/038029 patent/WO2021070874A1/ja unknown
- 2020-10-07 CN CN202080061207.3A patent/CN114341347A/zh active Pending
- 2020-10-07 EP EP20875184.2A patent/EP4043554A4/en active Pending
- 2020-10-07 CA CA3151911A patent/CA3151911A1/en active Pending
- 2020-10-07 JP JP2021551691A patent/JPWO2021070874A1/ja active Pending
- 2020-10-07 US US17/638,026 patent/US20220275343A1/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008528059A (ja) * | 2005-02-02 | 2008-07-31 | プライムジェン バイオテック エルエルシー | 分化細胞を治療用に再プログラム化するための核の材料の使用 |
| WO2008066630A2 (en) * | 2006-10-27 | 2008-06-05 | Caritas St. Elizabeth Medical Center Of Boston, Inc. | Methods for reprogramming adult somatic cells and uses thereof |
| WO2019098264A1 (ja) | 2017-11-17 | 2019-05-23 | 国立大学法人大阪大学 | Cd31陽性cd45陰性cd200陽性の哺乳動物細胞からなる細胞集団、およびその利用 |
Non-Patent Citations (5)
| Title |
|---|
| NAITO H ET AL.: "Identification and characterization of a resident vascular stem/progenitor cell population in preexisting blood vessels", EMBO J, vol. 31, no. 4, 15 February 2012 (2012-02-15), pages 842 - 55, XP055609361, DOI: 10.1038/emboj.2011.465 |
| TAKAKURA, NOBUYUKI: "JST Strategic Basic Research Programs, CREST Research area, 'Basic medical technology such as production and control of induced pluripotent stem cells (iPS cells)', research subject Structural elucidation of Physiological cellular reprogramming and its application", RESEARCH COMPLETION REPORTS, 19 November 2017 (2017-11-19), pages 1 - 21, XP055817460, Retrieved from the Internet <URL:https://web.archive.org/web/20171119013145/https://www.jst.go.jp/kisoken/crest/research/s-houkoku/JST_1111057_09154542_EE.pdf> [retrieved on 20201028] * |
| TATEDA, TOMOMI: "Effect of microenvironment on dedifferentiation of vascular endothelial cells -A study using human umbilical vein endothelial cells (HUVEC) and placenta extract", HUMAN SCIENCES, no. 14, 30 November 2010 (2010-11-30), pages 88 - 90, XP009534339, ISSN: 1344-6126 * |
| WAKABAYASHI T ET AL., CELL STEM CELL, vol. 22, no. 3, 1 March 2018 (2018-03-01), pages 384 - 397 |
| WAKABAYASHI T ET AL.: "CD157 Marks Tissue-Resident Endothelial Stem Cells with Homeostatic and Regenerative Properties", CELL STEM CELL, vol. 22, no. 3, 1 March 2018 (2018-03-01), pages 384 - 397, XP055609373, DOI: 10.1016/j.stem.2018.01.010 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20220275343A1 (en) | 2022-09-01 |
| CA3151911A1 (en) | 2021-04-15 |
| EP4043554A1 (en) | 2022-08-17 |
| JPWO2021070874A1 (ja) | 2021-04-15 |
| CN114341347A (zh) | 2022-04-12 |
| EP4043554A4 (en) | 2022-11-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP3934539B2 (ja) | 胎盤等由来の成体又は生後組織の前駆細胞 | |
| Wang et al. | Cell-to-cell contact induces mesenchymal stem cell to differentiate into cardiomyocyte and smooth muscle cell | |
| Kitamura et al. | Establishment and characterization of renal progenitor like cells from S3 segment of nephron in rat adult kidney | |
| US8309352B2 (en) | Human cord blood as a source of neural tissue repair of the brain and spinal cord | |
| AU2001243464A1 (en) | Human cord blood as a source of neural tissue for repair of the brain and spinal cord | |
| RU2665364C2 (ru) | Улучшение органов для трансплантации | |
| US20240279616A1 (en) | Cell populations of cd31-positive, cd45-negative, cd200-positive mammalian cells and use thereof | |
| KR102464099B1 (ko) | 다능성 간세포에 의한 허혈재관류 폐장애의 경감 및 치료 | |
| JP2021045168A (ja) | 腎臓の製造方法 | |
| WO2005123904A1 (ja) | 霊長類動物胚性幹細胞からの血管内皮細胞の製造方法 | |
| JPWO2004101775A1 (ja) | 新規な成体組織由来の幹細胞およびその用途 | |
| CN107714726A (zh) | 一种重塑骨髓微环境的方法 | |
| WO2021070874A1 (ja) | 血管内皮幹細胞の製造方法 | |
| WO2021201286A1 (ja) | ハイポテンシャル多能性幹細胞 | |
| KR20050086606A (ko) | 장기의 재생 방법 | |
| JP2007202512A (ja) | 腎臓幹/前駆細胞の分離方法及び腎臓幹/前駆細胞、並びに腎疾患治療剤 | |
| EP3888751B1 (en) | Cell product for use in treating a peripheral blood flow disorder | |
| WO2021029346A1 (ja) | 脳血管性認知症の治療または予防剤 | |
| JPWO2018003997A1 (ja) | 臓器線維症の予防または治療剤 | |
| Sanberg et al. | Human cord blood as a source of neural tissue repair of the brain and spinal cord | |
| Bojin et al. | Epithelization of skin lesions in animal model treated with mesenchymal stem cells and derivatives | |
| Watanabe et al. | Articles in PresS. Am J Physiol Gastrointest Liver Physiol (October 1, 2009). doi: 10.1152/ajpgi. 00123.2009 | |
| Howell | Identification of a common pluripotent stem cell population derived from multiple murine tissues | |
| Hongbao et al. | Bone Marrow Stem Cell |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20875184 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2021551691 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 3151911 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2020875184 Country of ref document: EP Effective date: 20220509 |