WO2020116625A1 - 水添共重合体組成物、粘接着材組成物及び粘接着フィルム - Google Patents
水添共重合体組成物、粘接着材組成物及び粘接着フィルム Download PDFInfo
- Publication number
- WO2020116625A1 WO2020116625A1 PCT/JP2019/047864 JP2019047864W WO2020116625A1 WO 2020116625 A1 WO2020116625 A1 WO 2020116625A1 JP 2019047864 W JP2019047864 W JP 2019047864W WO 2020116625 A1 WO2020116625 A1 WO 2020116625A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- copolymer composition
- hydrogenated copolymer
- mass
- component
- hydrogenated
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 250
- 229920001577 copolymer Polymers 0.000 title claims abstract description 217
- 239000000853 adhesive Substances 0.000 title claims description 89
- 230000001070 adhesive effect Effects 0.000 title claims description 84
- 239000002313 adhesive film Substances 0.000 title claims description 25
- 239000000463 material Substances 0.000 title description 26
- 229920000642 polymer Polymers 0.000 claims abstract description 141
- 239000000178 monomer Substances 0.000 claims abstract description 118
- 229920002554 vinyl polymer Polymers 0.000 claims abstract description 70
- 150000001993 dienes Chemical class 0.000 claims abstract description 45
- 239000000155 melt Substances 0.000 claims abstract description 8
- 229920005989 resin Polymers 0.000 claims description 45
- 239000011347 resin Substances 0.000 claims description 45
- 230000000903 blocking effect Effects 0.000 claims description 18
- 239000007822 coupling agent Substances 0.000 claims description 14
- 238000006886 vinylation reaction Methods 0.000 claims description 12
- 239000012790 adhesive layer Substances 0.000 abstract description 30
- 230000005764 inhibitory process Effects 0.000 abstract 2
- 238000004519 manufacturing process Methods 0.000 description 134
- -1 vinyl aromatic compound Chemical class 0.000 description 83
- 238000000034 method Methods 0.000 description 63
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 50
- 229920003048 styrene butadiene rubber Polymers 0.000 description 49
- 239000002174 Styrene-butadiene Substances 0.000 description 44
- 238000000465 moulding Methods 0.000 description 44
- 239000011115 styrene butadiene Substances 0.000 description 44
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 41
- 238000005984 hydrogenation reaction Methods 0.000 description 39
- 239000004743 Polypropylene Substances 0.000 description 33
- 229920001971 elastomer Polymers 0.000 description 33
- 229920001155 polypropylene Polymers 0.000 description 32
- 239000010410 layer Substances 0.000 description 29
- 239000011342 resin composition Substances 0.000 description 27
- 238000006116 polymerization reaction Methods 0.000 description 26
- 239000005060 rubber Substances 0.000 description 20
- 230000001629 suppression Effects 0.000 description 20
- 239000004820 Pressure-sensitive adhesive Substances 0.000 description 19
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 description 17
- 239000002585 base Substances 0.000 description 17
- 239000003054 catalyst Substances 0.000 description 17
- 238000006243 chemical reaction Methods 0.000 description 17
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 14
- 239000003795 chemical substances by application Substances 0.000 description 14
- 239000008188 pellet Substances 0.000 description 14
- 239000000806 elastomer Substances 0.000 description 13
- 239000000243 solution Substances 0.000 description 13
- 229910052783 alkali metal Inorganic materials 0.000 description 12
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical class C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 12
- 238000005259 measurement Methods 0.000 description 12
- 238000011156 evaluation Methods 0.000 description 11
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 10
- 239000004902 Softening Agent Substances 0.000 description 10
- BOTDANWDWHJENH-UHFFFAOYSA-N Tetraethyl orthosilicate Chemical compound CCO[Si](OCC)(OCC)OCC BOTDANWDWHJENH-UHFFFAOYSA-N 0.000 description 10
- 238000002156 mixing Methods 0.000 description 9
- 239000002904 solvent Substances 0.000 description 9
- UREWAKSZTRITCZ-UHFFFAOYSA-N buta-1,3-diene;styrene Chemical compound C=CC=C.C=CC=C.C=CC1=CC=CC=C1.C=CC1=CC=CC=C1 UREWAKSZTRITCZ-UHFFFAOYSA-N 0.000 description 8
- 125000004432 carbon atom Chemical group C* 0.000 description 8
- 239000004711 α-olefin Substances 0.000 description 8
- 239000000654 additive Substances 0.000 description 7
- 150000002642 lithium compounds Chemical class 0.000 description 7
- 238000004806 packaging method and process Methods 0.000 description 7
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 7
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 6
- 239000005977 Ethylene Substances 0.000 description 6
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 6
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 6
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 6
- 238000013461 design Methods 0.000 description 6
- 238000009826 distribution Methods 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- MTZQAGJQAFMTAQ-UHFFFAOYSA-N ethyl benzoate Chemical compound CCOC(=O)C1=CC=CC=C1 MTZQAGJQAFMTAQ-UHFFFAOYSA-N 0.000 description 6
- 238000005227 gel permeation chromatography Methods 0.000 description 6
- 239000003112 inhibitor Substances 0.000 description 6
- 150000002900 organolithium compounds Chemical class 0.000 description 6
- 239000003505 polymerization initiator Substances 0.000 description 6
- 238000005160 1H NMR spectroscopy Methods 0.000 description 5
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 5
- 239000004698 Polyethylene Substances 0.000 description 5
- 230000000996 additive effect Effects 0.000 description 5
- 239000003963 antioxidant agent Substances 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- 125000000524 functional group Chemical group 0.000 description 5
- 229930195733 hydrocarbon Natural products 0.000 description 5
- 239000001257 hydrogen Substances 0.000 description 5
- 229910052739 hydrogen Inorganic materials 0.000 description 5
- 230000006872 improvement Effects 0.000 description 5
- 238000001802 infusion Methods 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 230000000704 physical effect Effects 0.000 description 5
- 229920003023 plastic Polymers 0.000 description 5
- 239000004033 plastic Substances 0.000 description 5
- 229920002647 polyamide Polymers 0.000 description 5
- 229920000728 polyester Polymers 0.000 description 5
- 229920000573 polyethylene Polymers 0.000 description 5
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 5
- 239000003381 stabilizer Substances 0.000 description 5
- 150000003440 styrenes Chemical class 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 229910001868 water Inorganic materials 0.000 description 5
- MYRTYDVEIRVNKP-UHFFFAOYSA-N 1,2-Divinylbenzene Chemical compound C=CC1=CC=CC=C1C=C MYRTYDVEIRVNKP-UHFFFAOYSA-N 0.000 description 4
- VXNZUUAINFGPBY-UHFFFAOYSA-N 1-Butene Chemical compound CCC=C VXNZUUAINFGPBY-UHFFFAOYSA-N 0.000 description 4
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- 244000043261 Hevea brasiliensis Species 0.000 description 4
- 239000004952 Polyamide Substances 0.000 description 4
- 239000004793 Polystyrene Substances 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 229910000831 Steel Inorganic materials 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 4
- 150000001412 amines Chemical class 0.000 description 4
- 230000008859 change Effects 0.000 description 4
- 239000011248 coating agent Substances 0.000 description 4
- 238000000576 coating method Methods 0.000 description 4
- 239000005038 ethylene vinyl acetate Substances 0.000 description 4
- 238000001125 extrusion Methods 0.000 description 4
- 239000004088 foaming agent Substances 0.000 description 4
- 150000002430 hydrocarbons Chemical class 0.000 description 4
- 238000001746 injection moulding Methods 0.000 description 4
- 239000011256 inorganic filler Substances 0.000 description 4
- 229910003475 inorganic filler Inorganic materials 0.000 description 4
- 238000010030 laminating Methods 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 239000002480 mineral oil Substances 0.000 description 4
- 235000010446 mineral oil Nutrition 0.000 description 4
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 4
- 229920003052 natural elastomer Polymers 0.000 description 4
- 229920001194 natural rubber Polymers 0.000 description 4
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 4
- 239000005022 packaging material Substances 0.000 description 4
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 4
- 230000000379 polymerizing effect Effects 0.000 description 4
- 229920002223 polystyrene Polymers 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 229910052708 sodium Inorganic materials 0.000 description 4
- 239000011734 sodium Substances 0.000 description 4
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 4
- 239000010959 steel Substances 0.000 description 4
- 150000003505 terpenes Chemical class 0.000 description 4
- 235000007586 terpenes Nutrition 0.000 description 4
- 229920005992 thermoplastic resin Polymers 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 3
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 150000001339 alkali metal compounds Chemical class 0.000 description 3
- 125000000217 alkyl group Chemical group 0.000 description 3
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 3
- 238000011088 calibration curve Methods 0.000 description 3
- 238000005520 cutting process Methods 0.000 description 3
- KTQYJQFGNYHXMB-UHFFFAOYSA-N dichloro(methyl)silicon Chemical compound C[Si](Cl)Cl KTQYJQFGNYHXMB-UHFFFAOYSA-N 0.000 description 3
- 239000012530 fluid Substances 0.000 description 3
- 235000013305 food Nutrition 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 238000004898 kneading Methods 0.000 description 3
- 239000004611 light stabiliser Substances 0.000 description 3
- 239000005048 methyldichlorosilane Substances 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- SSDSCDGVMJFTEQ-UHFFFAOYSA-N octadecyl 3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)CCC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 SSDSCDGVMJFTEQ-UHFFFAOYSA-N 0.000 description 3
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 3
- YWAKXRMUMFPDSH-UHFFFAOYSA-N pentene Chemical compound CCCC=C YWAKXRMUMFPDSH-UHFFFAOYSA-N 0.000 description 3
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 3
- 229920000139 polyethylene terephthalate Polymers 0.000 description 3
- 239000005020 polyethylene terephthalate Substances 0.000 description 3
- 239000004926 polymethyl methacrylate Substances 0.000 description 3
- 229920000098 polyolefin Polymers 0.000 description 3
- 229920005629 polypropylene homopolymer Polymers 0.000 description 3
- 229910052700 potassium Inorganic materials 0.000 description 3
- 239000011591 potassium Substances 0.000 description 3
- 230000035484 reaction time Effects 0.000 description 3
- 230000003014 reinforcing effect Effects 0.000 description 3
- 239000002356 single layer Substances 0.000 description 3
- 229920003051 synthetic elastomer Polymers 0.000 description 3
- 229920003002 synthetic resin Polymers 0.000 description 3
- 239000000057 synthetic resin Substances 0.000 description 3
- 239000005061 synthetic rubber Substances 0.000 description 3
- 239000000454 talc Substances 0.000 description 3
- 229910052623 talc Inorganic materials 0.000 description 3
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 3
- JSNRRGGBADWTMC-UHFFFAOYSA-N (6E)-7,11-dimethyl-3-methylene-1,6,10-dodecatriene Chemical compound CC(C)=CCCC(C)=CCCC(=C)C=C JSNRRGGBADWTMC-UHFFFAOYSA-N 0.000 description 2
- ZGEGCLOFRBLKSE-UHFFFAOYSA-N 1-Heptene Chemical compound CCCCCC=C ZGEGCLOFRBLKSE-UHFFFAOYSA-N 0.000 description 2
- IANQTJSKSUMEQM-UHFFFAOYSA-N 1-benzofuran Chemical compound C1=CC=C2OC=CC2=C1 IANQTJSKSUMEQM-UHFFFAOYSA-N 0.000 description 2
- AFFLGGQVNFXPEV-UHFFFAOYSA-N 1-decene Chemical compound CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 2
- CRSBERNSMYQZNG-UHFFFAOYSA-N 1-dodecene Chemical compound CCCCCCCCCCC=C CRSBERNSMYQZNG-UHFFFAOYSA-N 0.000 description 2
- GQEZCXVZFLOKMC-UHFFFAOYSA-N 1-hexadecene Chemical compound CCCCCCCCCCCCCCC=C GQEZCXVZFLOKMC-UHFFFAOYSA-N 0.000 description 2
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 2
- HFDVRLIODXPAHB-UHFFFAOYSA-N 1-tetradecene Chemical compound CCCCCCCCCCCCC=C HFDVRLIODXPAHB-UHFFFAOYSA-N 0.000 description 2
- SDJHPPZKZZWAKF-UHFFFAOYSA-N 2,3-dimethylbuta-1,3-diene Chemical compound CC(=C)C(C)=C SDJHPPZKZZWAKF-UHFFFAOYSA-N 0.000 description 2
- HIXDQWDOVZUNNA-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-hydroxy-7-methoxychromen-4-one Chemical compound C=1C(OC)=CC(O)=C(C(C=2)=O)C=1OC=2C1=CC=C(OC)C(OC)=C1 HIXDQWDOVZUNNA-UHFFFAOYSA-N 0.000 description 2
- GTEXIOINCJRBIO-UHFFFAOYSA-N 2-[2-(dimethylamino)ethoxy]-n,n-dimethylethanamine Chemical compound CN(C)CCOCCN(C)C GTEXIOINCJRBIO-UHFFFAOYSA-N 0.000 description 2
- RCJMVGJKROQDCB-UHFFFAOYSA-N 2-methylpenta-1,3-diene Chemical compound CC=CC(C)=C RCJMVGJKROQDCB-UHFFFAOYSA-N 0.000 description 2
- YHQXBTXEYZIYOV-UHFFFAOYSA-N 3-methylbut-1-ene Chemical compound CC(C)C=C YHQXBTXEYZIYOV-UHFFFAOYSA-N 0.000 description 2
- JLBJTVDPSNHSKJ-UHFFFAOYSA-N 4-Methylstyrene Chemical compound CC1=CC=C(C=C)C=C1 JLBJTVDPSNHSKJ-UHFFFAOYSA-N 0.000 description 2
- VXEGSRKPIUDPQT-UHFFFAOYSA-N 4-[4-(4-methoxyphenyl)piperazin-1-yl]aniline Chemical compound C1=CC(OC)=CC=C1N1CCN(C=2C=CC(N)=CC=2)CC1 VXEGSRKPIUDPQT-UHFFFAOYSA-N 0.000 description 2
- WSSSPWUEQFSQQG-UHFFFAOYSA-N 4-methyl-1-pentene Chemical compound CC(C)CC=C WSSSPWUEQFSQQG-UHFFFAOYSA-N 0.000 description 2
- RSWGJHLUYNHPMX-UHFFFAOYSA-N Abietic-Saeure Natural products C12CCC(C(C)C)=CC2=CCC2C1(C)CCCC2(C)C(O)=O RSWGJHLUYNHPMX-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 229920000089 Cyclic olefin copolymer Polymers 0.000 description 2
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N Ethylbenzene Chemical compound CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 description 2
- 239000002879 Lewis base Substances 0.000 description 2
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- KHPCPRHQVVSZAH-HUOMCSJISA-N Rosin Natural products O(C/C=C/c1ccccc1)[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 KHPCPRHQVVSZAH-HUOMCSJISA-N 0.000 description 2
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical compound ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 239000006096 absorbing agent Substances 0.000 description 2
- 229920006243 acrylic copolymer Polymers 0.000 description 2
- 229920000800 acrylic rubber Polymers 0.000 description 2
- 150000001340 alkali metals Chemical class 0.000 description 2
- 150000001336 alkenes Chemical class 0.000 description 2
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 125000003368 amide group Chemical group 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 230000003078 antioxidant effect Effects 0.000 description 2
- 239000010426 asphalt Substances 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 2
- 239000012965 benzophenone Substances 0.000 description 2
- QRUDEWIWKLJBPS-UHFFFAOYSA-N benzotriazole Chemical compound C1=CC=C2N[N][N]C2=C1 QRUDEWIWKLJBPS-UHFFFAOYSA-N 0.000 description 2
- 239000012964 benzotriazole Substances 0.000 description 2
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 2
- 229920001400 block copolymer Polymers 0.000 description 2
- 238000000071 blow moulding Methods 0.000 description 2
- 125000005620 boronic acid group Chemical group 0.000 description 2
- 239000004566 building material Substances 0.000 description 2
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 2
- 239000008116 calcium stearate Substances 0.000 description 2
- 235000013539 calcium stearate Nutrition 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 239000004927 clay Substances 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 238000012937 correction Methods 0.000 description 2
- 238000005859 coupling reaction Methods 0.000 description 2
- DMEGYFMYUHOHGS-UHFFFAOYSA-N cycloheptane Chemical compound C1CCCCCC1 DMEGYFMYUHOHGS-UHFFFAOYSA-N 0.000 description 2
- NJLLQSBAHIKGKF-UHFFFAOYSA-N dipotassium dioxido(oxo)titanium Chemical compound [K+].[K+].[O-][Ti]([O-])=O NJLLQSBAHIKGKF-UHFFFAOYSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 150000002170 ethers Chemical class 0.000 description 2
- 238000007667 floating Methods 0.000 description 2
- 238000005187 foaming Methods 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 239000012943 hotmelt Substances 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- NNPPMTNAJDCUHE-UHFFFAOYSA-N isobutane Chemical compound CC(C)C NNPPMTNAJDCUHE-UHFFFAOYSA-N 0.000 description 2
- 150000007527 lewis bases Chemical class 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- WGOPGODQLGJZGL-UHFFFAOYSA-N lithium;butane Chemical compound [Li+].CC[CH-]C WGOPGODQLGJZGL-UHFFFAOYSA-N 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- QPJVMBTYPHYUOC-UHFFFAOYSA-N methyl benzoate Chemical compound COC(=O)C1=CC=CC=C1 QPJVMBTYPHYUOC-UHFFFAOYSA-N 0.000 description 2
- GDOPTJXRTPNYNR-UHFFFAOYSA-N methylcyclopentane Chemical compound CC1CCCC1 GDOPTJXRTPNYNR-UHFFFAOYSA-N 0.000 description 2
- VAMFXQBUQXONLZ-UHFFFAOYSA-N n-alpha-eicosene Natural products CCCCCCCCCCCCCCCCCCC=C VAMFXQBUQXONLZ-UHFFFAOYSA-N 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 2
- CCCMONHAUSKTEQ-UHFFFAOYSA-N octadec-1-ene Chemical compound CCCCCCCCCCCCCCCCC=C CCCMONHAUSKTEQ-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 239000012188 paraffin wax Substances 0.000 description 2
- 238000005453 pelletization Methods 0.000 description 2
- 229920001568 phenolic resin Polymers 0.000 description 2
- 239000005011 phenolic resin Substances 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 229920001084 poly(chloroprene) Polymers 0.000 description 2
- 229920000058 polyacrylate Polymers 0.000 description 2
- 229920002857 polybutadiene Polymers 0.000 description 2
- 229920001707 polybutylene terephthalate Polymers 0.000 description 2
- 229920001195 polyisoprene Polymers 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 229920005604 random copolymer Polymers 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 239000005049 silicon tetrachloride Substances 0.000 description 2
- CGRKYEALWSRNJS-UHFFFAOYSA-N sodium;2-methylbutan-2-olate Chemical compound [Na+].CCC(C)(C)[O-] CGRKYEALWSRNJS-UHFFFAOYSA-N 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- KHPCPRHQVVSZAH-UHFFFAOYSA-N trans-cinnamyl beta-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OCC=CC1=CC=CC=C1 KHPCPRHQVVSZAH-UHFFFAOYSA-N 0.000 description 2
- 239000001993 wax Substances 0.000 description 2
- CXENHBSYCFFKJS-UHFFFAOYSA-N (3E,6E)-3,7,11-Trimethyl-1,3,6,10-dodecatetraene Natural products CC(C)=CCCC(C)=CCC=C(C)C=C CXENHBSYCFFKJS-UHFFFAOYSA-N 0.000 description 1
- AHAREKHAZNPPMI-AATRIKPKSA-N (3e)-hexa-1,3-diene Chemical compound CC\C=C\C=C AHAREKHAZNPPMI-AATRIKPKSA-N 0.000 description 1
- PMJHHCWVYXUKFD-SNAWJCMRSA-N (E)-1,3-pentadiene Chemical compound C\C=C\C=C PMJHHCWVYXUKFD-SNAWJCMRSA-N 0.000 description 1
- PRJNEUBECVAVAG-UHFFFAOYSA-N 1,3-bis(ethenyl)benzene Chemical compound C=CC1=CC=CC(C=C)=C1 PRJNEUBECVAVAG-UHFFFAOYSA-N 0.000 description 1
- UYMQPNRUQXPLCY-UHFFFAOYSA-N 1-(2-piperidin-1-ylethyl)piperidine Chemical compound C1CCCCN1CCN1CCCCC1 UYMQPNRUQXPLCY-UHFFFAOYSA-N 0.000 description 1
- 229940106006 1-eicosene Drugs 0.000 description 1
- FIKTURVKRGQNQD-UHFFFAOYSA-N 1-eicosene Natural products CCCCCCCCCCCCCCCCCC=CC(O)=O FIKTURVKRGQNQD-UHFFFAOYSA-N 0.000 description 1
- KGRVJHAUYBGFFP-UHFFFAOYSA-N 2,2'-Methylenebis(4-methyl-6-tert-butylphenol) Chemical compound CC(C)(C)C1=CC(C)=CC(CC=2C(=C(C=C(C)C=2)C(C)(C)C)O)=C1O KGRVJHAUYBGFFP-UHFFFAOYSA-N 0.000 description 1
- QLZJUIZVJLSNDD-UHFFFAOYSA-N 2-(2-methylidenebutanoyloxy)ethyl 2-methylidenebutanoate Chemical compound CCC(=C)C(=O)OCCOC(=O)C(=C)CC QLZJUIZVJLSNDD-UHFFFAOYSA-N 0.000 description 1
- BIOCRZSYHQYVSG-UHFFFAOYSA-N 2-(4-ethenylphenyl)-n,n-diethylethanamine Chemical compound CCN(CC)CCC1=CC=C(C=C)C=C1 BIOCRZSYHQYVSG-UHFFFAOYSA-N 0.000 description 1
- OHDSHGBRKMRPHC-UHFFFAOYSA-N 2-(4-ethenylphenyl)-n,n-dimethylethanamine Chemical compound CN(C)CCC1=CC=C(C=C)C=C1 OHDSHGBRKMRPHC-UHFFFAOYSA-N 0.000 description 1
- VSKJLJHPAFKHBX-UHFFFAOYSA-N 2-methylbuta-1,3-diene;styrene Chemical compound CC(=C)C=C.C=CC1=CC=CC=C1.C=CC1=CC=CC=C1 VSKJLJHPAFKHBX-UHFFFAOYSA-N 0.000 description 1
- GPNYZBKIGXGYNU-UHFFFAOYSA-N 2-tert-butyl-6-[(3-tert-butyl-5-ethyl-2-hydroxyphenyl)methyl]-4-ethylphenol Chemical compound CC(C)(C)C1=CC(CC)=CC(CC=2C(=C(C=C(CC)C=2)C(C)(C)C)O)=C1O GPNYZBKIGXGYNU-UHFFFAOYSA-N 0.000 description 1
- BPINJMQATUWTID-UHFFFAOYSA-N 3,3-dimethylpentane-2,2-diamine Chemical compound CCC(C)(C)C(C)(N)N BPINJMQATUWTID-UHFFFAOYSA-N 0.000 description 1
- LDTAOIUHUHHCMU-UHFFFAOYSA-N 3-methylpent-1-ene Chemical compound CCC(C)C=C LDTAOIUHUHHCMU-UHFFFAOYSA-N 0.000 description 1
- UWSMKYBKUPAEJQ-UHFFFAOYSA-N 5-Chloro-2-(3,5-di-tert-butyl-2-hydroxyphenyl)-2H-benzotriazole Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O UWSMKYBKUPAEJQ-UHFFFAOYSA-N 0.000 description 1
- UYQYTUYNNYZATF-UHFFFAOYSA-N 6-methyl-4,6-bis(octylsulfanylmethyl)cyclohexa-1,3-dien-1-ol Chemical compound CCCCCCCCSCC1=CC=C(O)C(C)(CSCCCCCCCC)C1 UYQYTUYNNYZATF-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- NLZUEZXRPGMBCV-UHFFFAOYSA-N Butylhydroxytoluene Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NLZUEZXRPGMBCV-UHFFFAOYSA-N 0.000 description 1
- XMSPJSVNIFERFH-UHFFFAOYSA-N C(CCCCCCCCCCC)CCC(=S)OCC(COC(CCCCCCCCCCCCCC)=S)(COC(CCCCCCCCCCCCCC)=S)COC(CCCCCCCCCCCCCC)=S.S(CCC(=O)OCCCCCCCCCCCCCCCCCC)CCC(=O)OCCCCCCCCCCCC Chemical compound C(CCCCCCCCCCC)CCC(=S)OCC(COC(CCCCCCCCCCCCCC)=S)(COC(CCCCCCCCCCCCCC)=S)COC(CCCCCCCCCCCCCC)=S.S(CCC(=O)OCCCCCCCCCCCCCCCCCC)CCC(=O)OCCCCCCCCCCCC XMSPJSVNIFERFH-UHFFFAOYSA-N 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- GHKOFFNLGXMVNJ-UHFFFAOYSA-N Didodecyl thiobispropanoate Chemical compound CCCCCCCCCCCCOC(=O)CCSCCC(=O)OCCCCCCCCCCCC GHKOFFNLGXMVNJ-UHFFFAOYSA-N 0.000 description 1
- 239000003508 Dilauryl thiodipropionate Substances 0.000 description 1
- BRLQWZUYTZBJKN-UHFFFAOYSA-N Epichlorohydrin Chemical compound ClCC1CO1 BRLQWZUYTZBJKN-UHFFFAOYSA-N 0.000 description 1
- 239000004593 Epoxy Substances 0.000 description 1
- JIGUQPWFLRLWPJ-UHFFFAOYSA-N Ethyl acrylate Chemical compound CCOC(=O)C=C JIGUQPWFLRLWPJ-UHFFFAOYSA-N 0.000 description 1
- 229920000181 Ethylene propylene rubber Polymers 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- VHOQXEIFYTTXJU-UHFFFAOYSA-N Isobutylene-isoprene copolymer Chemical group CC(C)=C.CC(=C)C=C VHOQXEIFYTTXJU-UHFFFAOYSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- HTLZVHNRZJPSMI-UHFFFAOYSA-N N-ethylpiperidine Chemical compound CCN1CCCCC1 HTLZVHNRZJPSMI-UHFFFAOYSA-N 0.000 description 1
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 1
- 229920000459 Nitrile rubber Polymers 0.000 description 1
- 229920000571 Nylon 11 Polymers 0.000 description 1
- 229920000299 Nylon 12 Polymers 0.000 description 1
- 229920002292 Nylon 6 Polymers 0.000 description 1
- 229920002302 Nylon 6,6 Polymers 0.000 description 1
- 229920000572 Nylon 6/12 Polymers 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 239000005662 Paraffin oil Substances 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- JKIJEFPNVSHHEI-UHFFFAOYSA-N Phenol, 2,4-bis(1,1-dimethylethyl)-, phosphite (3:1) Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC=C1OP(OC=1C(=CC(=CC=1)C(C)(C)C)C(C)(C)C)OC1=CC=C(C(C)(C)C)C=C1C(C)(C)C JKIJEFPNVSHHEI-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical group OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229920002319 Poly(methyl acrylate) Polymers 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- 229920002367 Polyisobutene Polymers 0.000 description 1
- 229920012196 Polyoxymethylene Copolymer Polymers 0.000 description 1
- 229920009382 Polyoxymethylene Homopolymer Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 229920010524 Syndiotactic polystyrene Polymers 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 229920006311 Urethane elastomer Polymers 0.000 description 1
- 229920002978 Vinylon Polymers 0.000 description 1
- 239000011954 Ziegler–Natta catalyst Substances 0.000 description 1
- STLLXWLDRUVCHL-UHFFFAOYSA-N [2-[1-[2-hydroxy-3,5-bis(2-methylbutan-2-yl)phenyl]ethyl]-4,6-bis(2-methylbutan-2-yl)phenyl] prop-2-enoate Chemical compound CCC(C)(C)C1=CC(C(C)(C)CC)=CC(C(C)C=2C(=C(C=C(C=2)C(C)(C)CC)C(C)(C)CC)OC(=O)C=C)=C1O STLLXWLDRUVCHL-UHFFFAOYSA-N 0.000 description 1
- IORUEKDKNHHQAL-UHFFFAOYSA-N [2-tert-butyl-6-[(3-tert-butyl-2-hydroxy-5-methylphenyl)methyl]-4-methylphenyl] prop-2-enoate Chemical compound CC(C)(C)C1=CC(C)=CC(CC=2C(=C(C=C(C)C=2)C(C)(C)C)OC(=O)C=C)=C1O IORUEKDKNHHQAL-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 125000004018 acid anhydride group Chemical group 0.000 description 1
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 1
- 239000003522 acrylic cement Substances 0.000 description 1
- XECAHXYUAAWDEL-UHFFFAOYSA-N acrylonitrile butadiene styrene Chemical compound C=CC=C.C=CC#N.C=CC1=CC=CC=C1 XECAHXYUAAWDEL-UHFFFAOYSA-N 0.000 description 1
- 229920000122 acrylonitrile butadiene styrene Polymers 0.000 description 1
- 239000004676 acrylonitrile butadiene styrene Substances 0.000 description 1
- 229920001893 acrylonitrile styrene Polymers 0.000 description 1
- 238000003915 air pollution Methods 0.000 description 1
- 125000003172 aldehyde group Chemical group 0.000 description 1
- 229920006271 aliphatic hydrocarbon resin Polymers 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 1
- 150000004703 alkoxides Chemical class 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000005234 alkyl aluminium group Chemical group 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 239000002216 antistatic agent Substances 0.000 description 1
- 229920006231 aramid fiber Polymers 0.000 description 1
- 229920006272 aromatic hydrocarbon resin Polymers 0.000 description 1
- 239000010692 aromatic oil Substances 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- 238000012662 bulk polymerization Methods 0.000 description 1
- 229920005549 butyl rubber Polymers 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- 235000012241 calcium silicate Nutrition 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- UBAZGMLMVVQSCD-UHFFFAOYSA-N carbon dioxide;molecular oxygen Chemical compound O=O.O=C=O UBAZGMLMVVQSCD-UHFFFAOYSA-N 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 125000003262 carboxylic acid ester group Chemical group [H]C([H])([*:2])OC(=O)C([H])([H])[*:1] 0.000 description 1
- 229910052570 clay Inorganic materials 0.000 description 1
- 238000004581 coalescence Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000000748 compression moulding Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- MROCJMGDEKINLD-UHFFFAOYSA-N dichlorosilane Chemical compound Cl[SiH2]Cl MROCJMGDEKINLD-UHFFFAOYSA-N 0.000 description 1
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical compound C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 1
- 235000019304 dilauryl thiodipropionate Nutrition 0.000 description 1
- JJQZDUKDJDQPMQ-UHFFFAOYSA-N dimethoxy(dimethyl)silane Chemical compound CO[Si](C)(C)OC JJQZDUKDJDQPMQ-UHFFFAOYSA-N 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 229940069096 dodecene Drugs 0.000 description 1
- MCPKSFINULVDNX-UHFFFAOYSA-N drometrizole Chemical compound CC1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 MCPKSFINULVDNX-UHFFFAOYSA-N 0.000 description 1
- 238000009820 dry lamination Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 229920005558 epichlorohydrin rubber Polymers 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- BXOUVIIITJXIKB-UHFFFAOYSA-N ethene;styrene Chemical compound C=C.C=CC1=CC=CC=C1 BXOUVIIITJXIKB-UHFFFAOYSA-N 0.000 description 1
- 229920006244 ethylene-ethyl acrylate Polymers 0.000 description 1
- 239000005042 ethylene-ethyl acrylate Substances 0.000 description 1
- BLHLJVCOVBYQQS-UHFFFAOYSA-N ethyllithium Chemical compound [Li]CC BLHLJVCOVBYQQS-UHFFFAOYSA-N 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 229930009668 farnesene Natural products 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000003063 flame retardant Substances 0.000 description 1
- 229920001973 fluoroelastomer Polymers 0.000 description 1
- 239000005003 food packaging material Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000010528 free radical solution polymerization reaction Methods 0.000 description 1
- 235000013611 frozen food Nutrition 0.000 description 1
- 235000012055 fruits and vegetables Nutrition 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 238000012685 gas phase polymerization Methods 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 125000002795 guanidino group Chemical group C(N)(=N)N* 0.000 description 1
- 150000004820 halides Chemical group 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 229920002681 hypalon Polymers 0.000 description 1
- 125000005462 imide group Chemical group 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 239000012770 industrial material Substances 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 239000001282 iso-butane Substances 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- 229920003049 isoprene rubber Polymers 0.000 description 1
- ZBKFYXZXZJPWNQ-UHFFFAOYSA-N isothiocyanate group Chemical group [N-]=C=S ZBKFYXZXZJPWNQ-UHFFFAOYSA-N 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 238000003475 lamination Methods 0.000 description 1
- 239000010985 leather Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- UBJFKNSINUCEAL-UHFFFAOYSA-N lithium;2-methylpropane Chemical compound [Li+].C[C-](C)C UBJFKNSINUCEAL-UHFFFAOYSA-N 0.000 description 1
- SZAVVKVUMPLRRS-UHFFFAOYSA-N lithium;propane Chemical compound [Li+].C[CH-]C SZAVVKVUMPLRRS-UHFFFAOYSA-N 0.000 description 1
- XBEREOHJDYAKDA-UHFFFAOYSA-N lithium;propane Chemical compound [Li+].CC[CH2-] XBEREOHJDYAKDA-UHFFFAOYSA-N 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 239000002923 metal particle Substances 0.000 description 1
- 239000012968 metallocene catalyst Substances 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 229940095102 methyl benzoate Drugs 0.000 description 1
- BFXIKLCIZHOAAZ-UHFFFAOYSA-N methyltrimethoxysilane Chemical compound CO[Si](C)(OC)OC BFXIKLCIZHOAAZ-UHFFFAOYSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 239000004200 microcrystalline wax Substances 0.000 description 1
- 235000019808 microcrystalline wax Nutrition 0.000 description 1
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 description 1
- 125000002560 nitrile group Chemical group 0.000 description 1
- 239000002667 nucleating agent Substances 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 239000010690 paraffinic oil Substances 0.000 description 1
- PNJWIWWMYCMZRO-UHFFFAOYSA-N pent‐4‐en‐2‐one Natural products CC(=O)CC=C PNJWIWWMYCMZRO-UHFFFAOYSA-N 0.000 description 1
- 238000009512 pharmaceutical packaging Methods 0.000 description 1
- 239000002530 phenolic antioxidant Substances 0.000 description 1
- NHKJPPKXDNZFBJ-UHFFFAOYSA-N phenyllithium Chemical compound [Li]C1=CC=CC=C1 NHKJPPKXDNZFBJ-UHFFFAOYSA-N 0.000 description 1
- ODOPKAJVFRHHGM-UHFFFAOYSA-N phenyltin Chemical group [Sn]C1=CC=CC=C1 ODOPKAJVFRHHGM-UHFFFAOYSA-N 0.000 description 1
- 125000002270 phosphoric acid ester group Chemical group 0.000 description 1
- OJMIONKXNSYLSR-UHFFFAOYSA-N phosphorous acid Chemical compound OP(O)O OJMIONKXNSYLSR-UHFFFAOYSA-N 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- DOIRQSBPFJWKBE-UHFFFAOYSA-N phthalic acid di-n-butyl ester Natural products CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 description 1
- 238000000053 physical method Methods 0.000 description 1
- PMJHHCWVYXUKFD-UHFFFAOYSA-N piperylene Natural products CC=CC=C PMJHHCWVYXUKFD-UHFFFAOYSA-N 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920001083 polybutene Polymers 0.000 description 1
- 229920005668 polycarbonate resin Polymers 0.000 description 1
- 239000004431 polycarbonate resin Substances 0.000 description 1
- 229920001225 polyester resin Polymers 0.000 description 1
- 239000004645 polyester resin Substances 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 230000037048 polymerization activity Effects 0.000 description 1
- 229920005672 polyolefin resin Polymers 0.000 description 1
- 229920006324 polyoxymethylene Polymers 0.000 description 1
- 229920003246 polypentenamer Polymers 0.000 description 1
- 229920005606 polypropylene copolymer Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920003225 polyurethane elastomer Polymers 0.000 description 1
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 1
- ZRLVQFQTCMUIRM-UHFFFAOYSA-N potassium;2-methylbutan-2-olate Chemical compound [K+].CCC(C)(C)[O-] ZRLVQFQTCMUIRM-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- SCUZVMOVTVSBLE-UHFFFAOYSA-N prop-2-enenitrile;styrene Chemical compound C=CC#N.C=CC1=CC=CC=C1 SCUZVMOVTVSBLE-UHFFFAOYSA-N 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 238000001175 rotational moulding Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 238000010008 shearing Methods 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 239000004945 silicone rubber Substances 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 229920000468 styrene butadiene styrene block copolymer Polymers 0.000 description 1
- 229920001935 styrene-ethylene-butadiene-styrene Polymers 0.000 description 1
- 229920003047 styrene-styrene-butadiene-styrene Polymers 0.000 description 1
- 125000002130 sulfonic acid ester group Chemical group 0.000 description 1
- 125000000542 sulfonic acid group Chemical group 0.000 description 1
- 238000009864 tensile test Methods 0.000 description 1
- LFQCEHFDDXELDD-UHFFFAOYSA-N tetramethyl orthosilicate Chemical compound CO[Si](OC)(OC)OC LFQCEHFDDXELDD-UHFFFAOYSA-N 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 125000002813 thiocarbonyl group Chemical group *C(*)=S 0.000 description 1
- AWIJRPNMLHPLNC-UHFFFAOYSA-N thiocarboxylic acid group Chemical group C(=S)O AWIJRPNMLHPLNC-UHFFFAOYSA-N 0.000 description 1
- 125000005068 thioepoxy group Chemical group S(O*)* 0.000 description 1
- 125000000101 thioether group Chemical group 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- ZWYDDDAMNQQZHD-UHFFFAOYSA-L titanium(ii) chloride Chemical compound [Cl-].[Cl-].[Ti+2] ZWYDDDAMNQQZHD-UHFFFAOYSA-L 0.000 description 1
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 1
- JLTRXTDYQLMHGR-UHFFFAOYSA-N trimethylaluminium Chemical compound C[Al](C)C JLTRXTDYQLMHGR-UHFFFAOYSA-N 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- XSQUKJJJFZCRTK-UHFFFAOYSA-N urea group Chemical group NC(=O)N XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 1
- 238000004073 vulcanization Methods 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
- 239000013585 weight reducing agent Substances 0.000 description 1
- 238000009816 wet lamination Methods 0.000 description 1
- 238000004804 winding Methods 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- XOOUIPVCVHRTMJ-UHFFFAOYSA-L zinc stearate Chemical compound [Zn+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O XOOUIPVCVHRTMJ-UHFFFAOYSA-L 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L53/02—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers of vinyl-aromatic monomers and conjugated dienes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J153/00—Adhesives based on block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Adhesives based on derivatives of such polymers
- C09J153/02—Vinyl aromatic monomers and conjugated dienes
- C09J153/025—Vinyl aromatic monomers and conjugated dienes modified
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F297/00—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer
- C08F297/02—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type
- C08F297/04—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type polymerising vinyl aromatic monomers and conjugated dienes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F297/00—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer
- C08F297/02—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type
- C08F297/04—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type polymerising vinyl aromatic monomers and conjugated dienes
- C08F297/044—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type polymerising vinyl aromatic monomers and conjugated dienes using a coupling agent
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L53/02—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers of vinyl-aromatic monomers and conjugated dienes
- C08L53/025—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers of vinyl-aromatic monomers and conjugated dienes modified
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J11/00—Features of adhesives not provided for in group C09J9/00, e.g. additives
- C09J11/08—Macromolecular additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J153/00—Adhesives based on block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Adhesives based on derivatives of such polymers
- C09J153/02—Vinyl aromatic monomers and conjugated dienes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J7/00—Adhesives in the form of films or foils
- C09J7/30—Adhesives in the form of films or foils characterised by the adhesive composition
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J7/00—Adhesives in the form of films or foils
- C09J7/30—Adhesives in the form of films or foils characterised by the adhesive composition
- C09J7/38—Pressure-sensitive adhesives [PSA]
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J7/00—Adhesives in the form of films or foils
- C09J7/30—Adhesives in the form of films or foils characterised by the adhesive composition
- C09J7/38—Pressure-sensitive adhesives [PSA]
- C09J7/381—Pressure-sensitive adhesives [PSA] based on macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- C09J7/387—Block-copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
- C08L2205/025—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group containing two or more polymers of the same hierarchy C08L, and differing only in parameters such as density, comonomer content, molecular weight, structure
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/03—Polymer mixtures characterised by other features containing three or more polymers in a blend
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2453/00—Presence of block copolymer
Definitions
- the present invention relates to a hydrogenated copolymer composition, an adhesive composition and an adhesive film.
- a block copolymer composed of a vinyl aromatic monomer unit and a conjugated diene monomer unit has elasticity at room temperature similar to vulcanized natural rubber or synthetic rubber without vulcanization, Moreover, since it has the same molding processability as thermoplastic resins at high temperatures, it is used in the fields of footwear, plastic modification, asphalt modification, adhesives, etc., household products, home appliances, industrial parts, and other packaging materials. Widely used in toys, etc. Further, since the hydrogenated product of the block copolymer is excellent in weather resistance and heat resistance, it is widely used in automobile parts, medical instruments and the like in addition to the above-mentioned fields of application.
- tacky adhesive when it is used as a tacky adhesive, it is mainly used as a label, tape or surface protection film by applying hot melt for sanitary materials and building materials, or by providing an adhesive layer on the film surface.
- an adhesive/adhesive agent used for the adhesive layer of such a film conventionally, an acrylic adhesive/adhesive agent or a rubber-based adhesive/adhesive agent mainly containing rubber such as natural rubber and polyisobutylene has been mainly used.
- a method of applying these tacky adhesives to a predetermined support film a method of applying a tacky adhesive solution obtained by dissolving the tacky adhesive in a solvent using a roll, a spray or the like is used. ..
- These methods have the advantage of being able to coat the adhesive layer uniformly and thinly, but since they use a solvent, air pollution, fire, occupational safety and hygiene during manufacturing, economic efficiency, etc. There is a problem that it is not preferable from the viewpoint of.
- a coextrusion film in which a base material layer made of a polyolefin resin and a tacky adhesive layer containing a hydrogenated styrene elastomer or an olefin elastomer are integrated is preferably used. ing.
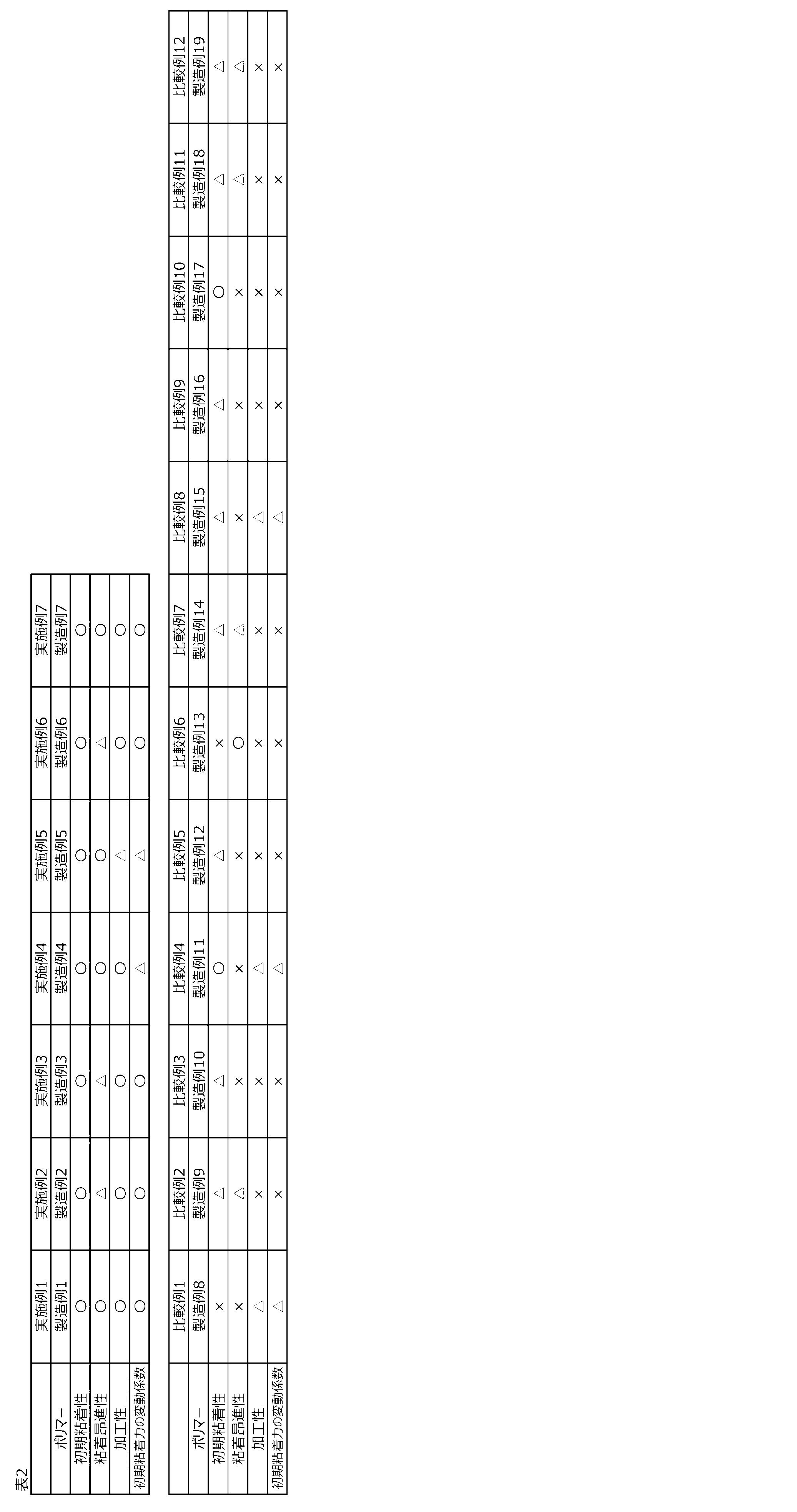
- Patent Document 1 describes an adhesive film aiming to achieve both moldability and adhesiveness without admixing a tackifier, but does not mention adhesiveness-promoting property, There is room for improvement.
- Patent Document 2 describes an adhesive film excellent in initial adhesiveness, low-temperature adhesiveness, feeding property, low adhesiveness-promoting property, and property balance, but does not mention moldability, There is room for improvement.
- Patent Document 3 describes a pressure-sensitive adhesive composition for the purpose of controlling initial tackiness, low tack-promoting property, surface contamination suppression, adhesive residue suppression, and zipping suppression, but use of a specific tacky resin compound. Therefore, there is still room for improvement in that these problems are essentially solved by the hydrogenated copolymer composition.
- the uneven thickness of the adhesive layer is not only regarded as a problem at the time of molding but also affects the adhesive strength and the adhesion promotion. Further, if there is unevenness in the thickness of the adhesive layer, floating of the film, adhesive residue, and zipping at the time of peeling occur, so suppression is particularly important. Conventional resin compositions still have room for improvement in terms of having a highly balanced balance of initial adhesive strength, suppression of adhesion promotion, and suppression of adhesive layer thickness unevenness.
- the present invention in view of the above-mentioned problems of the prior art, when used as a tacky adhesive, excellent initial pressure force, suppression of adhesion promotion, suppression of uneven thickness of the adhesive layer, a hydrogenated copolymer composition.
- the object is to provide a product, an adhesive composition, and an adhesive film.
- the present inventors have conducted extensive studies to solve the above-mentioned problems of the prior art, and as a result, a hydrogenated copolymer composition containing two components having a specific structure can effectively solve the above-mentioned problems.
- the present invention has been completed and the present invention has been completed.
- the present invention is as follows.
- a hydrogenated copolymer composition comprising: The component (a) has a peak top molecular weight of 50,000 to 120,000, The component (b) has a peak top molecular weight of 1.8 to 3.8 times the peak top molecular weight of the component (a), 81 mol% or more of the double bonds of the conjugated diene monomer unit contained in the hydrogenated copolymer composition are hydrogenated, The content of the vinyl aromatic monomer unit contained in the hydrogenated copolymer composition is 5 to 18% by mass based on the composition, The vinyl aromatic monomer unit contained in the hydrogenated copolymer composition has a blocking rate of 75% by mass
- the hydrogenated copolymer composition component (a) according to any one of [1] to [4], wherein the component (a) and the component (b) are each represented by the following formula: B2-A1- B1 and/or B2-A1-B1-X Component (b): (B2-A1-B1) n X [In the formula, A1 is a polymer block mainly composed of vinyl aromatic monomer units, B1 and B2 are each independently a polymer block mainly containing a conjugated diene monomer unit, n is an integer of 2 or more, X is the residue of the coupling agent].
- the initial adhesive force, suppression of adhesion promotion, and a highly excellent hydrogenated copolymer composition which is excellent in balance of suppression of thickness unevenness of the adhesive layer, an adhesive composition and An adhesive film is obtained.
- the present embodiment a mode for carrying out the present invention (hereinafter, referred to as “the present embodiment”) will be described in detail, but the present invention is not limited to the following description and various modifications are possible within the scope of the gist. It can be modified and implemented.
- the hydrogenated copolymer composition of the present embodiment Component (a) containing a polymer block mainly composed of vinyl aromatic monomer units and a polymer block mainly composed of conjugated diene monomer units; and mainly composed of vinyl aromatic monomer units
- the component (a) has a peak top molecular weight of 50,000 to 120,000
- the component (b) has a peak top molecular weight of 1.8 to 3.8 times the peak top molecular weight of the component (a), 81 mol% or more of the double bonds of the conjugated diene monomer unit contained in the hydrogenated copolymer composition are hydrogenated,
- the content of the vinyl aromatic monomer unit contained in the hydrogenated copolymer composition is 5 to 18% by mass based on the composition
- the vinyl aromatic monomer unit contained in the hydrogenated copolymer composition has a blocking rate of 75% by
- each monomer unit constituting the copolymer is named according to the name of the monomer from which the monomer unit is derived.
- vinyl aromatic monomer unit means a constitutional unit of a polymer, which is formed as a result of polymerizing a vinyl aromatic compound as a monomer.
- the vinyl aromatic monomer unit is a vinyl group of a vinyl aromatic compound and is bonded to another monomer unit.
- conjugated diene monomer unit means a constitutional unit of a polymer, which is generated as a result of polymerizing a conjugated diene compound which is a monomer.
- the conjugated diene monomer unit is bonded to another monomer unit on one of the two double bonds of the conjugated diene compound (1,2-bond or 3,4-bond), or the conjugated diene compound Both of the two double bonds are bound to other monomer units (1,4-bond).
- the hydrogenated copolymer composition of the present embodiment contains the component (a) and the component (b).
- the total content of the component (a) and the component (b) is preferably 90% by mass or more, more preferably 95% by mass or more, further preferably 100% by mass, based on the hydrogenated copolymer composition. %.
- the component (a) includes a polymer block A mainly containing vinyl aromatic monomer units and a polymer block B mainly containing conjugated diene monomer units.
- the number of the polymer blocks A and the polymer blocks B contained in the component (a) may be one or plural.
- the content of the polymer block A contained in the component (a) is preferably 4 to 16% by mass, more preferably 6 to 14% by mass, further preferably 8 to 10% by mass, based on the component (a). It is 12% by mass.
- the content of the polymer block B contained in the component (a) is preferably 84 to 96% by mass, more preferably 86 to 94% by mass, further preferably 88 to 96% by mass, based on the component (a). It is 92 mass %.
- the total content of the polymer block A and the polymer block B contained in the component (a) is preferably 90% by mass or more, more preferably 95% by mass or more, based on the component (a). It is preferably 100% by mass.
- the polymer block A contained in the component (a) is mainly composed of vinyl aromatic monomer units.
- the amount of the vinyl aromatic monomer unit contained in the polymer block A is 75% by mass or more, preferably 80% by mass or more, and more preferably 90% by mass or more, based on the polymer block A. is there.
- the polymer block B contained in the component (a) is mainly composed of a conjugated diene monomer unit.
- the amount of the conjugated diene monomer unit contained in the polymer block B is 90% by mass or more, preferably 96% by mass or more, and more preferably 99% by mass or more, based on the polymer block B. ..
- the component (b) contains a polymer block A mainly composed of vinyl aromatic monomer units and a polymer block B mainly composed of conjugated diene monomer units.
- the polymer block A and the polymer block B contained in the component (b) may be the same as or different from the polymer block A and the polymer block B contained in the component (a), respectively. Good.
- the number of polymer blocks A and polymer blocks B contained in the component (b) may be one or more.
- the content of the polymer block A contained in the component (b) is preferably 4 to 16% by mass, more preferably 6 to 14% by mass, further preferably 8 to 10% by mass, based on the component (b). It is 12% by mass.
- the content of the polymer block B contained in the component (b) is preferably 84 to 96% by mass, more preferably 86 to 94% by mass, further preferably 88 to 96% by mass, based on the component (b). It is 92 mass %.
- the total content of the polymer block A and the polymer block B contained in the component (b) is preferably 90% by mass or more, more preferably 95% by mass or more, based on the component (b). It is preferably 100% by mass.
- the polymer block A contained in the component (b) is mainly composed of vinyl aromatic monomer units.
- the amount of the vinyl aromatic monomer unit contained in the polymer block A is 75% by mass or more, preferably 80% by mass or more, and more preferably 90% by mass or more, based on the polymer block A. is there.
- the polymer block B contained in the component (b) is mainly composed of a conjugated diene monomer unit.
- the amount of the conjugated diene monomer unit contained in the polymer block B is 90% by mass or more, preferably 96% by mass or more, and more preferably 99% by mass or more, based on the polymer block B. ..
- Examples of the “vinyl aromatic compound” constituting the “vinyl aromatic monomer unit” include, but are not limited to, styrene, ⁇ -methylstyrene, p-methylstyrene, divinylbenzene, 1, Examples thereof include 1-diphenylethylene, N,N-dimethyl-p-aminoethylstyrene, N,N-diethyl-p-aminoethylstyrene and the like.
- styrene, ⁇ -methylstyrene and p-methylstyrene are preferable from the viewpoint of availability and productivity. Of these, styrene is particularly preferable. These may be used alone or in combination of two or more.
- the conjugated diene compound that constitutes the “conjugated diene monomer unit” is a diolefin having a pair of conjugated double bonds.
- the conjugated diene compound include, but are not limited to, 1,3-butadiene, 2-methyl-1,3-butadiene (isoprene), 2,3-dimethyl-1,3-butadiene, and 1,3-pentadiene. , 2-methyl-1,3-pentadiene, 1,3-hexadiene, and farnesene.
- Preferred diolefins include 1,3-butadiene and isoprene. These may be used alone or in combination of two or more.
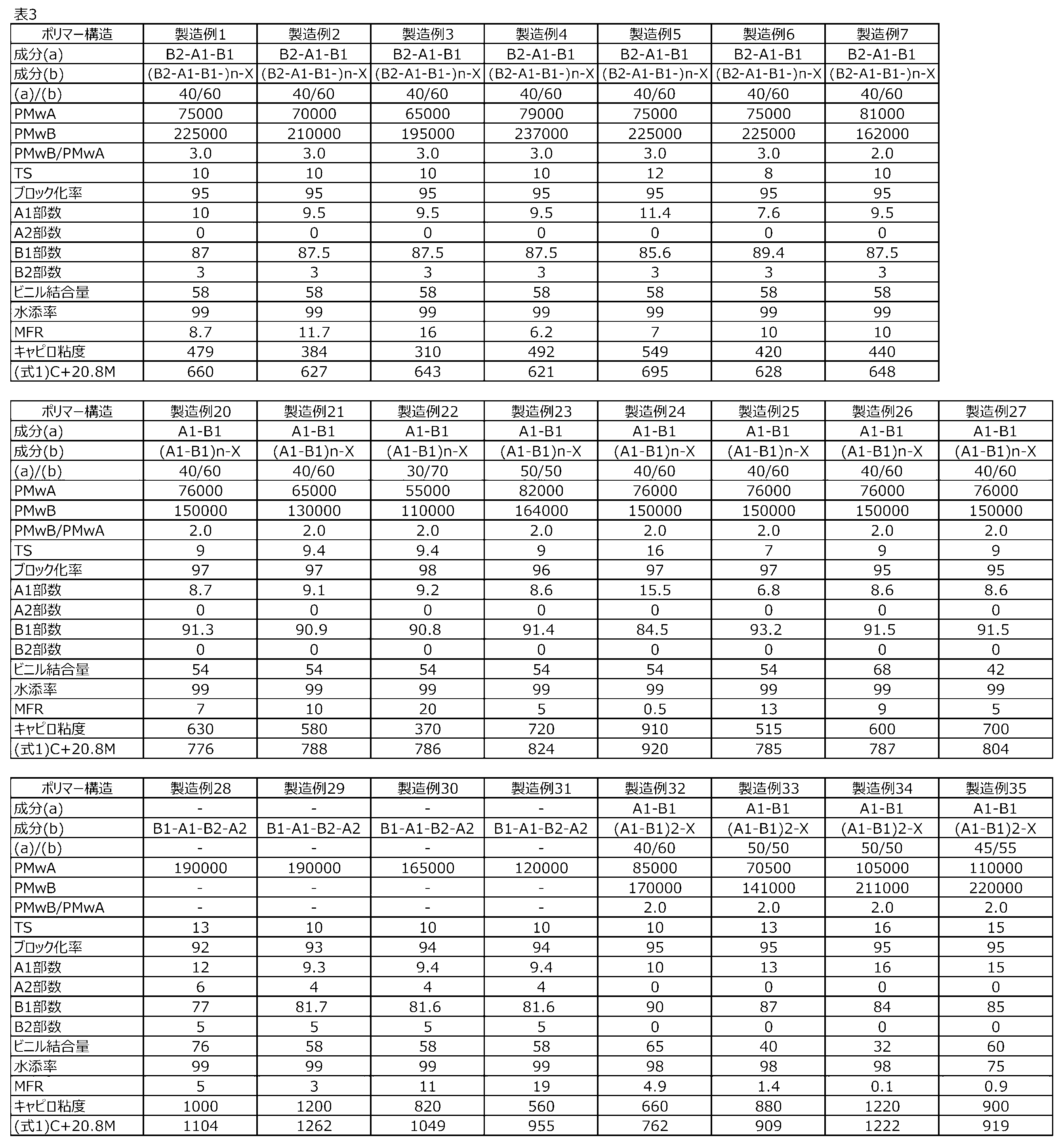
- the peak top molecular weight of the component (a) is 50,000 to 120,000, preferably 60,000 to 110,000, and more preferably 70,000 to 100,000. When the peak top molecular weight of the component (a) is 50,000 or more, it is excellent in suppression of adhesion promotion and molding processability. When the peak top molecular weight of the component (a) is 120,000 or less, the tackiness and molding processability are excellent.
- the peak top molecular weight can be determined by determining the molecular weight corresponding to the top of the peak obtained by gel permeation chromatography (GPC) (solvent: tetrahydrofuran, temperature: 40°C) from a standard polystyrene calibration curve.
- GPC gel permeation chromatography
- the peak top molecular weight of the component (b) is 1.8 to 3.8 times, preferably 1.9 to 3.7 times, and more preferably 2.0 times the peak top molecular weight of the component (a). It is from 2 times to 3.6 times, and more preferably from 2.1 times to 3.5 times. When the peak top molecular weight of the component (b) is within the above range, the moldability is excellent.
- the weight ratio of component (a)/component (b) is preferably 60/40 to 10/90, more preferably 55/45 to 20/80, and further preferably 50/50 to 30/70. Is. When the mass ratio of the component (b) is 40 or more, the adhesive strength and the suppression of the progress of adhesion are excellent. When the mass ratio of the component (b) is 90 mass or less, the moldability is excellent.
- the method for obtaining the hydrogenated copolymer composition containing the component (a) and the component (b) is not particularly limited.
- the component (a) and the component (b) may be polymerized and hydrogenated, respectively, and these may be solution blended at an appropriate ratio or dry blended.
- the component (a) is polymerized, a part of the component (a) is subjected to a coupling reaction to form the component (b), and a mixture of the component (a) and the component (b) is contained. It is preferred to obtain the polymer composition in one go and hydrogenate it.
- the coupling agent is not particularly limited as long as the above peak top molecular weights and mass ratios of the component (a) and the component (b) can be obtained, and examples thereof include polyalkenyl coupling agents.
- Suitable polyalkenyl coupling agents include, for example, divinylbenzene, with m-divinylbenzene being preferred.
- the coupling agent for example, tetraalkoxysilane such as tetraethoxysilane and tetramethoxysilane, alkyltrialkoxysilane such as methyltrimethoxysilane, dialkyldialkoxysilane such as dimethyldimethoxysilane, ethyl benzoate and methyl benzoate.
- diglycidyl aromatic epoxy compounds such as diglycidyl ether derived from the reaction of bisphenol A with epichlorohydrin.
- the components (a) and (b) are preferably each independently represented by the following formula.
- A1 and A2 are each independently a polymer block mainly composed of a vinyl aromatic monomer unit
- B1 and B2 are each independently a polymer block mainly containing a conjugated diene monomer unit
- n is an integer of 2 or more
- X is the residue of the coupling agent.
- the component (a) and/or the component (b) preferably have a coupling structure.
- the component (a) is B2-. It preferably has a structure represented by A1-B1 and/or B2-A1-B1-X, and the component (b) preferably has a structure represented by (B2-A1-B1) n X.
- N is preferably an integer of 2 to 4, and more preferably 3 or 4.
- n to 3 or 4 it is possible to control n to 3 or 4 by using tetraethoxysilane as a coupling agent and adjusting the stirring time at a temperature at the time of addition of 100° C. or less under the conditions of a stirrer such as a paddle blade. is there.
- the content of B2 is preferably 3 to 10 mass% based on the hydrogenated copolymer composition from the viewpoint of moldability. ..
- the content of B2 is 10% or less, it tends to be excellent in suppressing the progress of adhesion.
- the content of the vinyl aromatic monomer unit in the hydrogenated copolymer composition of the present embodiment is 5 to 18% by mass, preferably 6 to 17% by mass, based on the composition. It is more preferably 8 to 12% by mass.
- the content of the vinyl aromatic monomer unit is 18% by mass or less, the tackiness and molding processability will be better.
- the content of the vinyl aromatic monomer unit is 5% by mass or more, the adhesion promotion can be suppressed and the moldability tends to be good.
- the content of the vinyl aromatic monomer unit in the hydrogenated copolymer composition of the present embodiment can be measured by an ultraviolet spectrophotometer as described in Examples below. Since the content of the vinyl aromatic monomer unit is almost the same before and after the hydrogenation, it may be grasped by the content of the vinyl aromatic monomer unit in the copolymer before the hydrogenation.
- the content of the vinyl aromatic monomer unit in the hydrogenated copolymer composition can be controlled within the predetermined numerical range by adjusting the addition amount of the vinyl aromatic compound in the polymerization step.
- the content of the vinyl aromatic monomer block in the hydrogenated copolymer composition with respect to the content of all vinyl aromatic monomer units in the hydrogenated copolymer composition is defined as “vinyl aromatic monomer amount”. It is called a “body blocking rate”, or simply “blocking rate”.
- the vinyl aromatic monomer block is a set of three or more continuous vinyl aromatic monomer units, and its signal can be detected by 1 H-NMR.
- the blocking rate of the hydrogenated copolymer composition of the present embodiment is 75% by mass or more, preferably 80% by mass or more, and more preferably 90% by mass or more. When the blocking rate is 75% by mass or more, there is a tendency that the suppression of adhesion progress and the molding processability are excellent.
- the blocking rate can be measured by 1 H-NMR as described in Examples.
- the content of the conjugated diene monomer unit in the hydrogenated copolymer composition of the present embodiment is preferably 82 to 95% by mass, more preferably 83 to 94% by mass, based on the composition. Yes, and more preferably 88 to 92% by mass.
- the conjugated diene monomer unit is incorporated into the copolymer in the form of 1,2-bond, 3,4-bond, or 1,4-bond. ing.
- the total proportion of conjugated diene monomer units incorporated in the 1,2-bond or 3,4-bond mode is 1,2-bond, 3,4-bond, or 1,4-bond bond. It is preferably 40 to 70 mol %, more preferably 45 to 65 mol %, based on the total of conjugated diene monomer units incorporated in the manner.
- Conjugated diene monomer unit incorporated in 1,2-bond or 3,4-bond has a vinyl group, and thus is incorporated in 1,2-bond or 3,4-bond.
- the total proportion of conjugated diene monomer units is called the "vinylation rate".
- the vinylation rate is 40 mol% or more, the initial adhesive strength is excellent, the practically effective adhesiveness can be exhibited, and the moldability is excellent.
- the vinylation rate is 70 mol% or less, it tends to be excellent in suppressing the progress of adhesion.
- the vinylation rate can be controlled by adjusting the Lewis base (eg, ether, amine, etc.) as a vinylating agent, the amount used, and the polymerization temperature.
- the vinylation rate can be measured by nuclear magnetic resonance spectrum analysis ( 1 H-NMR) or the like.
- the vinylation rate may be measured from either the state before hydrogenation or the state after hydrogenation. Specifically, it can be measured by the method described in Examples described later.
- the double bond of the conjugated diene monomer unit contained in the hydrogenated copolymer composition of the present embodiment is hydrogenated.
- the proportion of double bonds in the hydrogenated conjugated diene monomer unit (hereinafter referred to as “hydrogenation rate”) is 81 mol% or more, preferably 85 mol% or more, and more preferably 90 mol% or more.
- hydrogenation rate is 81% or more, adhesion promotion can be suppressed, molding processability is excellent, deterioration during molding can be suppressed, and gel generation can be suppressed.
- the hydrogenation rate can be controlled, for example, by adjusting the catalyst amount and hydrogen feed amount during hydrogenation.
- the hydrogenation rate can be controlled, for example, by adjusting the catalyst amount, hydrogen feed amount, pressure and temperature during hydrogenation. Further, the hydrogenation rate can be measured by the method described in Examples described later.
- the melt flow rate of the hydrogenated copolymer composition of the present embodiment (hereinafter, also referred to as “MFR”; conforming to ISO 1133) is 4 to 20 g/10 minutes under the conditions of a temperature of 230° C. and a load of 2.16 kg. And preferably 5 to 18 g/10 minutes, more preferably 6 to 16 g/10 minutes.
- MFR is 20 g/10 minutes or less, the balance between the adhesive force and the suppression of adhesion promotion is excellent.
- the MFR is 4 g/10 minutes or more, the moldability is excellent.
- the MFR of the hydrogenated copolymer composition can be controlled by adjusting the polymerization conditions such as the amount of monomer added, the polymerization time, the temperature and the polymerization initiator, and is measured by the method described in Examples below. can do.
- the measured viscosity of the hydrogenated copolymer composition of the present embodiment (hereinafter, also referred to as “capillo viscosity”) is 300 to 600 Pa ⁇ s at a temperature of 230° C. and a shear rate of 121.6 sec ⁇ 1. Yes, it is preferably 350 to 580 Pa ⁇ s, and more preferably 370 to 560 Pa ⁇ s.
- This Capillo viscosity is a value measured according to ISO11443, but it is a so-called "apparent viscosity” that is not subjected to the Bargley correction or the Rabinovich correction, and the measured value greatly changes depending on the condition of the measuring device. Specify the conditions as follows. The accuracy of the following numerical values and the conditions not specified below comply with ISO11443. ⁇ Capillary die inner diameter: ⁇ 1.0 mm ⁇ Capillary die length: 10.0 mm ⁇ Inflow angle: 180° ⁇ Piston diameter: ⁇ 9.510mm ⁇ Furnace diameter: ⁇ 9.55
- capillo viscosity is 600 Pa ⁇ s or less, the moldability is excellent, and if it is 300 Pa ⁇ s or more, the balance between the adhesive force and the suppression of adhesion promotion is excellent.
- the capillo viscosity of the hydrogenated copolymer composition can be controlled by adjusting the amount of the monomer added, the polymerization time, the temperature, the polymerization conditions such as the polymerization initiator, and the method described in Examples below. Can be measured.
- Capillo viscosity (C) and MFR (M) of the hydrogenated copolymer composition of the present embodiment are represented by the following formula (1): C ⁇ 20.8 M+754.2 (Formula 1) Meet the relationship.
- Formula (1) is derived as a result of detailed examination of the relationship between the capillarity and MFR of a plurality of hydrogenated copolymer compositions and the moldability. Since both MFR and capillo viscosity are indices of viscosity, they naturally correlate, but MFR reflects low shear, and capillo viscosity is set to a constant condition (temperature 230°C, shear rate 121.6 sec-1). It is thought that this reflects the viscosity at high shear.
- the viscosity at low shear and the viscosity at high shear satisfy the preferable range as described above. Therefore, the molding processability of the hydrogenated copolymer composition and the adhesive film It has an excellent balance of adhesive strength and suppression of adhesion promotion. Specifically, for example, in coextrusion molding using a film extruder, thickness unevenness is unlikely to occur in the pressure-sensitive adhesive layer, and the pressure-sensitive adhesive force of the tacky-adhesive film and the balance between suppression of adhesion promotion are good.
- a hydrogenated copolymer composition comprising: The component (a) has a peak top molecular weight of 50,000 to 120,000, The component (b) has a peak top molecular weight of 1.8 to 3.8 times the peak top molecular weight of the component (a), 81 mol% or more of the double bonds of the conjugated diene monomer unit contained in the hydrogenated copolymer composition are hydrogenated, The content of the vinyl aromatic monomer unit contained in the hydrogenated copolymer composition is 5 to 18% by mass based on the composition, It is necessary to satisfy the requirement that the blocking ratio of the vinyl aromatic monomer unit
- the styrene block (large) has a molecular weight of about 10,000 to 20,000 and is set to have a molecular weight of 5 to 10 times that of the styrene block (small).
- the styrene block (small) has a molecular weight of about 10,000 to 20,000 and is set to have a molecular weight of 5 to 10 times that of the styrene block (small).
- Design example 2 Two or more kinds of polymers having different sizes of vinyl aromatic monomer blocks between different molecules are mixed. (The size of vinyl aromatic monomer block in the same molecular chain is the same) Similar to Design Example 1, due to the influence of the vinyl aromatic monomer blocks having different sizes, the effect of different viscosities at high shear and low shear can be expected.
- the layer thickness ratio with the base material layer will change, and the stress applied to the peeled portion will change during peeling, which will affect the adhesive strength.
- the layer thickness ratio with the base material layer changes, the force for holding the adhesive layer by the base material layer portion also changes, which affects the adhesion promotion. For other reasons, it also affects zipping, adhesive residue, and film floating.
- the hydrogenated copolymer composition of the present embodiment is produced, for example, by polymerizing in an organic solvent using an organic alkali metal compound as a polymerization initiator to obtain a copolymer and then performing a hydrogenation reaction. be able to.
- the mode of polymerization may be batch polymerization, continuous polymerization, or a combination thereof. From the viewpoint of obtaining a copolymer having a narrow molecular weight distribution, the batch polymerization method is preferable.
- the polymerization temperature is generally 0 to 150° C., preferably 20 to 120° C., more preferably 40 to 100° C., and further preferably 40 to 80° C.
- the polymerization time varies depending on the target polymer, but is usually within 24 hours, preferably 0.1 to 10 hours. From the viewpoint of obtaining a copolymer having a narrow molecular weight distribution and high strength, 0.5 to 3 hours is more preferable.
- the polymerization system atmosphere is not particularly limited as long as it is in a pressure range sufficient to maintain nitrogen and a solvent in a liquid phase.
- impurities such as water, oxygen, carbon dioxide, etc. that inactivate the polymerization initiator and the living polymer are not present in the polymerization system.
- the organic solvent is not particularly limited, and examples thereof include aliphatic hydrocarbons such as n-butane, isobutane, n-pentane, n-hexane, n-heptane, and n-octane; cyclohexane, cycloheptane, methylcyclopentane. And alicyclic hydrocarbons; aromatic hydrocarbons such as benzene, xylene, toluene, and ethylbenzene.
- Organolithium compounds are preferred as the organic alkali metal compound that is a polymerization initiator.
- an organic lithium compound an organic monolithium compound, an organic dilithium compound, or an organic polylithium compound can be used.
- organolithium compound include, but are not limited to, ethyllithium, n-propyllithium, isopropyllithium, n-butyllithium, sec-butyllithium, t-butyllithium, phenyllithium, hexamethylenediene.
- examples thereof include lithium, butadienyllithium, and isopropenyldilithium.
- n-butyllithium and sec-butyllithium are preferable from the viewpoint of polymerization activity.
- the amount of the organic alkali metal compound used as a polymerization initiator varies depending on the molecular weight of the target polymer, but is generally in the range of 0.01 to 0.5 phm (parts by mass relative to 100 parts by mass of monomer). Is more preferable, the range of 0.03 to 0.3 phm is more preferable, and the range of 0.05 to 0.15 phm is further preferable.
- the total amount of 1,2-bonds and 1,4-bonds of conjugated diene monomer units in the hydrogenated copolymer composition is controlled by using a Lewis base (eg, ether, amine, etc.) as a vinylating agent. it can.
- a Lewis base eg, ether, amine, etc.
- the amount of the vinylating agent used is adjusted according to the desired vinyl bond amount. Further, by adding a vinylating agent and a metal alkoxide described below under two or more conditions separately, it is possible to produce a polymer having a different vinyl bond amount in a polymer mainly containing a conjugated diene monomer unit. it can.
- Examples of the vinylating agent include, but are not limited to, ether compounds, ether compounds having two or more oxygen atoms, and tertiary amine compounds.
- Examples of the tertiary amine compound include, but are not limited to, pyridine, N,N,N′,N′-tetramethylethylenediamine, tributylamine, tetramethylpropanediamine and 1,2-diamine. Examples thereof include piperidinoethane and bis[2-(N,N-dimethylamino)ethyl]ether. These may be used alone or in combination of two or more.
- a compound having two amines is preferable. Further, among them, those having a structure showing symmetry in the molecule are more preferable, and N,N,N′,N′-tetramethylethylenediamine and bis[2-(N,N-dimethylamino)ethyl] Ether and 1,2-dipiperidinoethane are more preferred.
- the polymerization may be carried out in the coexistence of the above-mentioned vinylating agent, organic lithium compound and alkali metal alkoxide.
- the alkali metal alkoxide is a compound represented by the general formula MOR (in the formula, M is an alkali metal and R is an alkyl group).
- the alkali metal of the alkali metal alkoxide is preferably sodium or potassium from the viewpoint of a high vinyl bond amount, a narrow molecular weight distribution, a high polymerization rate, and a high blocking rate.
- alkali metal alkoxide examples include, but are not limited to, sodium alkoxide, lithium alkoxide, and potassium alkoxide having an alkyl group having 2 to 12 carbon atoms, and more preferably alkyl having 3 to 6 carbon atoms.
- Sodium alkoxide and potassium alkoxide having a group are more preferable, and sodium-t-butoxide, sodium-t-pentoxide, potassium-t-butoxide, and potassium-t-pentoxide are more preferable.
- sodium alkoxides such as sodium-t-butoxide and sodium-t-pentoxide are even more preferable.
- the molar ratio of the vinylating agent and the organolithium compound (vinyl It is preferable that the agent/organic lithium compound) and the molar ratio of the alkali metal alkoxide and the organic lithium compound (alkali metal alkoxide/organic lithium compound) coexist at the following molar ratios.
- Vinylating agent/organic lithium compound is 0.2 to less than 3.0 Alkali metal alkoxide/organic lithium compound is 0.3 or less
- the molar ratio of the vinylating agent/organolithium compound is more preferably 0.5 or more from the viewpoint of high vinyl bond amount and high polymerization rate, and 2.5 or less from the viewpoint of narrow molecular weight distribution and high hydrogenation activity. Is more preferable, and the range of 0.8 or more and 2.0 or less is further preferable.
- the molar ratio of the alkali metal alkoxide/organolithium compound is more preferably 0.2 or less, further preferably 0.1 or less, still more preferably 0.08 or less from the viewpoint of narrow molecular weight distribution and high hydrogenation activity.
- the molar ratio of alkali metal alkoxide/vinylating agent is more preferably 0.1 or less, further preferably 0.08 or less, and 0.06 or less from the viewpoint of achieving a narrow molecular weight distribution and obtaining high hydrogenation activity. Is even more preferable, and 0.05 or less is even more preferable.
- the amount of vinyl bond in the polymer mainly containing the conjugated diene monomer unit As a method of producing different polymers, a method of using a deactivator for the vinylating agent can also be mentioned.
- Deactivators include, but are not limited to, for example, alkyl metal compounds, such as alkylaluminums having from 1 to 20 carbon atoms per alkyl substituent, alkylzinc and alkylmagnesium, and It is selected from these mixtures.
- the method of hydrogenation is not particularly limited, for example, the copolymer obtained as described above, hydrogenation in the presence of a hydrogenation catalyst.
- the hydrogenated copolymer in which the double bond residue of the conjugated diene monomer unit is hydrogenated can be obtained.
- pellets of the hydrogenated copolymer composition can be produced by pelletizing the hydrogenated copolymer composition obtained as described above.
- a method of pelletizing for example, a method in which a hydrogenated copolymer is extruded in a strand form from a single-screw or twin-screw extruder and cut in water by a rotary blade installed in front of a die part; A method of extruding the hydrogenated copolymer composition in a strand form from a machine, water-cooling or air-cooling, and then cutting with a strand cutter; open roll, melt-mixed with a Banbury mixer, and then formed into a sheet with a roll, and further Examples include a method of cutting a sheet into strips and then cutting into cubic pellets with a pelletizer.
- the size and shape of the pellet molded product of the hydrogenated copolymer composition is not particularly limited.
- the pellets of the hydrogenated copolymer composition of the present embodiment may optionally contain a pellet blocking inhibitor for the purpose of preventing pellet blocking.
- pellet blocking inhibitor examples include, but are not limited to, calcium stearate, magnesium stearate, zinc stearate, polyethylene, polypropylene, ethylenebisstearylamide, talc, and amorphous silica. From the viewpoint of the tackiness of the tacky adhesive film obtained by molding the hydrogenated copolymer composition, the pellet blocking inhibitor is preferably polyethylene, ethylenebisstearylamide, or calcium stearate.
- the blending amount of the pellet blocking inhibitor is preferably 500 to 10000 ppm with respect to the hydrogenated copolymer composition, and more preferably 1000 to 7000 ppm with respect to the hydrogenated copolymer composition.
- the pellet blocking inhibitor is preferably blended in a state of being attached to the pellet surface, but it may be partly contained inside the pellet.
- the adhesive composition of this embodiment contains the hydrogenated copolymer composition of this embodiment.
- the adhesive composition of this embodiment may contain a tackifying resin.
- the tackifying resin is not particularly limited as long as it is a resin that can impart viscosity to the adhesive composition of the present embodiment, for example, rosin-based terpene-based resin, hydrogenated rosin-based terpene-based resin, Known tackifying resins such as coumarone resins, terpene resins, hydrogenated terpene resins, phenolic resins, terpene-phenolic resins, aromatic hydrocarbon resins and aliphatic hydrocarbon resins can be mentioned.
- the tackifier resins may be used alone or in combination of two or more.
- tackifying resin those described in "Rubber/Plastic Blended Chemicals" (edited by Rubber Digest) can be used. By using the tackifying resin, the adhesive strength can be improved.
- the content of the tackifying resin in the pressure-sensitive adhesive composition of the present embodiment is preferably 100 parts by mass or less, and 80 parts by mass or less, when the total mass of the hydrogenated copolymer composition is 100 parts by mass. Is more preferable and 60 parts by mass or less is further preferable. It is preferable that the content of the tackifying resin is 100 parts by mass or less, because there is a tendency that the adhesion promotion can be effectively suppressed.
- the adhesive composition of the present embodiment may further contain a hydrogenated styrene elastomer having a structure other than the hydrogenated copolymer composition of the present embodiment described above.
- hydrogenated styrene elastomer examples include, but are not limited to, for example, styrene-ethylene-butylene-styrene (styrene-butadiene-styrene (SBS), styrene-isoprene-styrene saturated by hydrogenation).
- SBS styrene-ethylene-butylene-styrene
- SEPS styrene-ethylene-propylene-styrene
- Other examples include elastomers having a structure of styrene-ethylene-butylene (SEB) and styrene-ethylene-propylene (SEP).
- a reactive elastomer having various functional groups added to the hydrogenated styrene elastomer may be used.
- Examples of the functional group include, but are not limited to, hydroxyl group, carboxyl group, carbonyl group, thiocarbonyl group, acid halide group, acid anhydride group, thiocarboxylic acid group, aldehyde group, thioaldehyde group.
- the adhesive composition of this embodiment may further contain an ethylene vinyl acetate copolymer.
- the ethylene-vinyl acetate copolymer is not particularly limited, but can be produced, for example, by radically copolymerizing ethylene and vinyl acetate under conditions of high temperature and high pressure.
- the ethylene vinyl acetate copolymer has different properties depending on the vinyl acetate content, but the vinyl acetate content is not particularly limited.
- the adhesive composition of this embodiment may further contain an acrylic copolymer.
- the acrylic copolymer is not limited to the following, for example, a copolymer of methyl acrylate, ethyl acrylate, methyl methacrylate, acrylonitrile and the like, vinyl acetate, vinyl chloride, styrene and the like. Can be mentioned.
- the adhesive composition of the embodiment may further contain a softening agent.
- the softening agent is not limited to the following, but examples thereof include a mineral oil softening agent and a synthetic resin softening agent.
- the mineral oil-based softener generally includes a mixture of aromatic hydrocarbons, naphthene-based hydrocarbons and paraffin-based hydrocarbons. It should be noted that paraffinic hydrocarbons having 50% or more of the total carbon atoms are called paraffinic oils, and naphthenic hydrocarbons having 30 to 45% carbon atoms are called naphthenic oils. In addition, aromatic hydrocarbons having 35% or more carbon atoms are called aromatic oils.
- Paraffin oil which is a softening agent for rubber, is preferable as the mineral oil-based softening agent, and polybutene, low molecular weight polybutadiene, etc. are preferable as the synthetic resin-based softening agent.
- the tackiness of the adhesive composition of the present embodiment tends to be further improved.
- the mass of the hydrogenated copolymer composition of the embodiment is 100 parts by mass, it is preferably 100 parts by mass or less, and more preferably 80 parts by mass or less.
- a stabilizer such as an antioxidant or a light stabilizer may be further added to the adhesive composition of the present embodiment.
- antioxidants examples include, but are not limited to, 2,6-di-t-butyl-4-methylphenol, n-octadecyl-3-(4′-hydroxy-3′,5′, and the like.
- -Di-t-butylphenyl)propionate 2,2'-methylenebis(4-methyl-6-t-butylphenol), 2,2'-methylenebis(4-ethyl-6-t-butylphenol), 2,4- Bis[(octylthio)methyl]-o-cresol, 2-t-butyl-6-(3-t-butyl-2-hydroxy-5-methylbenzyl)-4-methylphenyl acrylate, 2,4-di-t -Amyl-6-[1-(3,5-di-t-amyl-2-hydroxyphenyl)ethyl]phenyl acrylate, 2-[1-(2-hydroxy-3,5-di-tert-pentylphenyl) ] Hindered
- Examples of the light stabilizer include, but are not limited to, 2-(2′-hydroxy-5′-methylphenyl)benzotriazole and 2-(2′-hydroxy-3′,5′-t. -Butylphenyl)benzotriazole, 2-(2'-hydroxy-3',5'-di-t-butylphenyl)-5-chlorobenzotriazole and other benzotriazole-based UV absorbers and 2-hydroxy-4-methoxy Examples thereof include benzophenone-based ultraviolet absorbers such as benzophenone and hindered amine-based light stabilizers.
- various additives may be added to the adhesive composition of the present embodiment, if necessary.
- the additive examples include, but are not limited to, pigments such as red iron oxide and titanium dioxide; waxes such as paraffin wax, microcrystalline wax, and low molecular weight polyethylene wax; amorphous polyolefin, ethylene-ethyl acrylate.
- Polyolefin-based or low-molecular-weight vinyl aromatic thermoplastics such as copolymers; natural rubber; polyisoprene rubber, polybutadiene rubber, styrene-butadiene rubber, ethylene-propylene rubber, chloroprene rubber, acrylic rubber, isoprene-isobutylene rubber, Examples thereof include synthetic rubbers such as polypentenamer rubber.
- synthetic rubber examples include those described in “Rubber/Plastic Blended Chemicals” (edited by Rubber Digest).
- the method for producing the pressure-sensitive adhesive composition of the present embodiment is not particularly limited, and can be produced by a conventionally known method.
- a melt kneading method using a general kneader such as a Banbury mixer, a single screw extruder, a twin screw extruder, a co-kneader, and a multi-screw extruder
- a general kneader such as a Banbury mixer
- a single screw extruder such as a twin screw extruder
- a co-kneader a multi-screw extruder
- examples include a method of dissolving or dispersing and mixing each component, coating the base material film with a coater or the like, and then removing the solvent by heating.
- the adhesive composition of the present embodiment may be subjected to foaming treatment in order to reduce weight, soften, and improve adhesiveness.
- the foaming treatment method is not limited to the following, but includes, for example, a chemical method, a physical method, use of a thermal expansion type microballoon, and the like.
- Bubbles can be distributed inside the material by adding a chemical foaming agent such as an inorganic foaming agent or an organic foaming agent, a physical foaming agent or the like, or adding a thermal-thermal expansion type microballoon. Further, by adding a hollow filler (expanded balloon), weight reduction, softening, and improvement of adhesion may be achieved.
- the tacky-adhesive film of this embodiment contains the hydrogenated copolymer composition or the tacky-adhesive material composition of this embodiment.
- the pressure-sensitive adhesive film of the present embodiment has a configuration in which the hydrogenated copolymer composition or the pressure-sensitive adhesive material composition of the present embodiment described above is provided as a pressure-sensitive adhesive layer laminated on a predetermined base film. It is preferably one.
- the material of the base film is not particularly limited, and both nonpolar resin and polar resin can be used.
- the nonpolar resin is preferably polyethylene, homo- or block polypropylene
- the polar resin is a polyester resin such as polyethylene terephthalate or polybutylene terephthalate, a polyamide resin, an ethylene-vinyl acetate copolymer. Coalescence and its hydrolyzate etc. are preferable.
- the thickness of the pressure-sensitive adhesive layer is preferably 1.0 ⁇ m or more and 100 ⁇ m or less, and 2.0 ⁇ m or more and 100 ⁇ m or less. Is more preferable.
- the thickness of the adhesive layer is 100 ⁇ m or less, it tends to be more excellent in handleability, which is economically preferable. If it is 1 ⁇ m or more, the adhesion tends to be more excellent, and a uniform thickness tends to be easily obtained.
- the thickness of the substrate film is preferably 5.0 mm or less, more preferably 3.0 mm or less, still more preferably 1.0 mm or less, even more preferably 300 ⁇ m or less, still more preferably 10 ⁇ m or more and 200 ⁇ m or less.
- a sheet having a thickness of more than 300 ⁇ m is called a “sheet”, but in the present specification, these are also described as a film.
- the method for producing the tacky-adhesive film of the present embodiment is not limited to the following, for example, a solution or melt of the hydrogenated copolymer composition or the tacky-adhesive material composition of the present embodiment is used. Examples thereof include a method of coating on the predetermined base film and a method of coextruding the base layer and the adhesive layer using a film extruder.
- the solution or melt of the composition may be used alone, or the solution or melt of the hydrogenated copolymer composition may be mixed.
- the method of applying the solution of the hydrogenated copolymer composition or the adhesive composition of the present embodiment is not limited to the following, for example, these are dissolved in a solvent capable of being dissolved, and a coater or the like is used.
- a pressure-sensitive adhesive film can be produced by applying the adhesive film on a base film and heating and drying the solvent.
- the method is not limited to the following, for example, using a hot melt coater, the substrate A pressure-sensitive adhesive film can be produced by applying the hydrogenated copolymer composition or the pressure-sensitive adhesive composition of the present embodiment melted on the film.
- a substrate film having a glass transition temperature, melting point or softening point higher than the coating temperature it is preferable to use a substrate film having a glass transition temperature, melting point or softening point higher than the coating temperature.
- the components of the adhesive layer containing the hydrogenated copolymer composition or adhesive composition of the present embodiment and the substrate film layer The constituent components such as thermoplastic resin are made into two streams by a melt co-extruder, that is, the adhesive layer forming fluid and the base material film forming fluid are combined in a die port to form a single stream.
- a pressure-sensitive adhesive film can be produced by forming a fluid and extruding it to form a composite of the adhesive layer and the resin film layer.
- the pressure-sensitive adhesive composition of this embodiment is also produced by previously dry-blending each component of the hydrogenated copolymer composition or the pressure-sensitive adhesive composition. This is a method with excellent productivity because it can be performed.
- the tacky-adhesive film is produced by extrusion molding, the tacky-adhesive film tends to have particularly excellent adhesion and tackiness.
- thermoplastic resins such as polypropylene
- properties required for resin compositions with thermoplastic resins such as polypropylene include tensile strength. Elongation at break, low temperature impact resistance, low molding shrinkage, balance of the above-mentioned properties, and the like can be mentioned. Low molding shrinkage generally means that when a resin composition is molded, residual strain occurs and the strain relaxes over time, which causes dimensional change, but the change is small.
- the resin composition containing the hydrogenated copolymer composition of the present embodiment and the polypropylene resin tends to be excellent in tensile elongation at break, low temperature impact resistance, and low molding shrinkage.
- Polypropylene resin As the polypropylene resin, in addition to homopolypropylene, random polypropylene and block polypropylene can be used.
- Random in random polypropylene is a copolymer of propylene and a monomer other than propylene, and means that monomers other than propylene are randomly incorporated into the propylene chain, and monomers other than propylene are not substantially chained.
- the random polypropylene is not particularly limited as long as the content of propylene units is less than 99% by mass.
- Examples of the random polypropylene include a random copolymer of propylene and ethylene, a random copolymer of propylene and an ⁇ -olefin having 4 to 20 carbon atoms, and the like.
- ⁇ -olefins examples include, but are not limited to, ethylene, 1-butene, 1-pentene, 3-methyl-1-butene, 1-hexene, 3-methyl-1-pentene, 4-methyl-1- Examples include pentene, 1-heptene, 1-octene, 1-nonene, 1-decene, 1-dodecene, 1-tetradecene, 1-hexadecene, 1-octadecene, 1-eicosene and the like.
- Homopolypropylene and random polypropylene can also be used individually by 1 type or in combination of 2 or more types. Only one type of polypropylene may be used, or two or more types of polypropylene resins may be mixed. In the case of a mixture of two or more kinds of polypropylene resins, for example, a homopolypropylene obtained by polymerizing an ethylene-propylene copolymer by different processes and mixing different solid pellets may be used.
- the method for adjusting the polypropylene resin is not particularly limited, and for example, it can be adjusted by a known method.
- a known catalyst such as a Ziegler-Natta catalyst or a metallocene catalyst may be used.
- a method for adjusting the polypropylene resin for example, various processes such as one-stage or multi-stage may be adopted, and different polymers are polymerized in various processes such as slurry polymerization method, gas phase polymerization method, bulk polymerization method and solution polymerization. , May be mixed.
- the content of the polypropylene resin and the content of the hydrogenated copolymer composition is preferably in the range of 98/2 to 5/95, more preferably 90/10 to 10/90, and 85/ It is more preferably 15 to 15/85.
- the content of the polypropylene resin is preferably 5 mass% or more from the viewpoint of mechanical strength of the resin composition of the present embodiment, and is preferably 98 mass% or less from the viewpoint of low temperature impact resistance.
- an ethylene- ⁇ -olefin copolymer rubber (olefin elastomer) may be used in combination depending on the required performance.
- the ethylene- ⁇ -olefin copolymer rubber is a copolymer of ethylene and ⁇ -olefin. There is no particular limitation, and known ones may be used. Examples of the copolymer component include ethylene and one or more C3 to C20 ⁇ -olefins.
- thermoplastic resin other than polypropylene resin may be used in combination depending on the required performance.
- specific examples include polyesters such as polyethylene, polystyrene, polyamide, polycyclohexylethane, and polyethylene terephthalate, ethylene/styrene interpolymers, syndiotactic polystyrene, EPDM, and the like. These may be used alone or in combination of two or more.
- the resin composition of the present embodiment may be combined with other additives depending on the required performance.
- the additive is not particularly limited, and examples thereof include lubricants, flame retardants, stabilizers, colorants, pigments, antioxidants, antistatic agents, dispersants, flow enhancers, release agents such as stearic acid metal salts, and silicones.
- examples include oils, mineral oil-based softeners, synthetic resin-based softeners, copper damage inhibitors, crosslinking agents, nucleating agents, and the like.
- the resin composition of the present embodiment may use an inorganic filler in combination depending on the required performance.
- the inorganic filler is not particularly limited and may be a known one.
- the shape of the inorganic filler is not particularly limited, and may be scaly, spherical, granular, powder, irregular shape, or the like. These can be used alone or in combination.
- the resin composition of the present embodiment is a method of dry blending a hydrogenated copolymer and a polypropylene resin, and further other components, if necessary, in a blending amount according to the characteristics of each component, a conventional polymer material.
- a method of preparation is exemplified by an apparatus provided for mixing.
- the mixing device is not limited to the following, and examples thereof include kneading devices such as a Banbury mixer, a Labo Plastomill, a single-screw extruder, and a twin-screw extruder. It is preferable from the viewpoint of kneadability.
- the melting temperature at the time of kneading can be appropriately set, but is usually in the range of 130 to 300°C, preferably in the range of 150 to 250°C.
- the molded product of the present embodiment is a molded product of the resin composition of the present embodiment described above.
- a sheet-shaped molded product sheet, film
- tube, bag medical molded product
- medical tube medical tube
- medical film automobile member
- automobile interior skin material medical infusion bag
- packaging material examples thereof include food packaging materials and clothing packaging materials, but the molded body of the present embodiment is not limited to the above.
- the molded body of this embodiment can be molded by the method described below.
- the resin composition of the present embodiment can be molded by softening or melting with heat.
- conventional molding methods such as compression molding, injection molding, gas-assisted injection molding, hollow molding, sheet molding, rotational molding, laminate molding, vacuum molding, heat molding, and extrusion molding can be used. it can.
- These molding techniques may be used alone or in combination of two or more. Among these, injection molding is preferable from the viewpoint of productivity.
- the method for molding the tube is not particularly limited, for example, the resin composition of the present embodiment is charged into an extruder and melted, and this is tubed by passing through a die and water-cooled or air-cooled to form a tube. can do.
- a single-screw or multi-screw extruder can be used as the extruder, and a multi-layer extruded multilayer tube can be molded using a plurality of extruders. Alternatively, it can be molded as a tube directly from an extruder when producing a resin composition using a polypropylene resin.
- the tube that is a molded body may be a multilayer tube in which other polymers are laminated within a range that does not impair the purpose of the present embodiment.
- the above-mentioned other polymers may be used alone or in combination of two or more kinds, and may be used as a single layer or as a laminate of multiple layers which may be different in each layer.
- a pressure resistant tube in order to further improve pressure resistance and the like while suppressing an increase in wall thickness and maintaining flexibility, a pressure resistant tube (hose) can be formed by winding a braided reinforcing thread or a spiral reinforcing body. ..
- the braided reinforcing yarn is provided inside or between layers in the thickness direction, and vinylon, polyamide, polyester, aramid fiber, carbon fiber, metal wire or the like can be used, and the spiral reinforcement is provided on the outer periphery, metal, plastic, etc. Can be used.
- the method for producing the sheet-shaped molded article is not particularly limited, but examples of the molding method in which the resin composition is put into an extruder and extruded include a T-die method and an inflation method.
- inflation molding ordinary air-cooled inflation molding, air-cooled two-stage inflation molding, high-speed inflation molding, water-cooled inflation molding and the like can be adopted.
- a blow molding method such as direct blow or injection blow, or a press molding method may be employed.
- the sheet-shaped molded body may be a single-layer sheet, or may be a multi-layer sheet by laminating another polymer within a range not impairing the gist of the present embodiment.
- other polymers include, but are not limited to, olefin polymers such as polypropylene, polyethylene, ethylene-propylene copolymer rubber (EPM) and ethylene-propylene-non-conjugated diene copolymer rubber (EPDM); polyesters.
- polyester-based polymers such as polyethylene terephthalate and polybutylene terephthalate; polyamide-based resins such as polyamide 6, polyamide 6,6, polyamide 6,10, polyamide 11, polyamide 12, and polyamide 6,12; polymethyl acrylate and Acrylic resin such as polymethylmethacrylate; Polyoxymethylene resin such as polyoxymethylene homopolymer and polyoxymethylene copolymer; Styrene resin such as styrene homopolymer, acrylonitrile-styrene resin, and acrylonitrile-butadiene-styrene resin Polycarbonate resin; Styrene-based elastomers such as styrene-butadiene copolymer rubber and styrene-isoprene copolymer rubber and hydrogenated products or modified products thereof; Natural rubber; Synthetic isoprene rubber and liquid polyisoprene rubber and hydrogenated products thereof Or modified products; chloroprene rubber; acrylic rubber; butylene tere
- multi-layer T-die method In laminating with other polymers, multi-layer T-die method, multi-layer inflation method, co-extrusion molding method such as extrusion lamination method, general multilayer sheet or film molding method such as wet lamination, dry lamination and press molding. Blow molding methods such as multilayer injection blow such as coin injection blow and multilayer direct blow can be employed.
- the formed multilayer laminate may be unstretched or may be uniaxially or biaxially stretched.
- the bag means a bag-shaped molded product that can be molded from the sheet-shaped molded product.
- bags include food packaging bags, clothing packaging bags, medical bags such as medical infusion bags, and drug packaging bags.
- the resin composition of the present embodiment is excellent in tensile elongation at break and low temperature impact resistance, low molding shrinkage, and a good balance of the above properties, and is used without any particular limitation on the application. be able to.
- Materials for automotive interior and exterior parts such as skin materials, road pavement materials, waterproof materials, waterproof sheets, civil engineering packing, daily necessities, leisure goods, It can be suitably used for a wide range of applications such as toys, industrial products, furniture products, writing utensils, water supply members, transparent pockets, holders, stationery such as file back covers, medical devices such as infusion bags, and the like.
- the peak top molecular weight was measured by GPC [apparatus: manufactured by Waters]. In the GPC measurement, tetrahydrofuran was used as the solvent and the temperature was 40°C. In the chromatograph obtained by GPC measurement, the peak top molecular weights of the peak having the highest peak top (referred to as “first peak”) and the peak having the next highest peak top (referred to as “second peak”) were It was determined using a calibration curve (prepared using the peak molecular weight of standard polystyrene) obtained from the measurement of commercially available standard polystyrene. Of the first and second peaks, the one having a smaller peak top molecular weight was the peak corresponding to the component (a), and the one having a larger peak top molecular weight was the peak corresponding to the component (b).
- the inflection points between the peaks are the points where the peaks divided by the fitting process by the Gaussian approximation using the EcoSEC waveform separation software intersect.
- ⁇ Content of vinyl aromatic monomer unit (TS) (styrene content) of hydrogenated copolymer composition A certain amount of the hydrogenated copolymer composition was dissolved in chloroform and measured with an ultraviolet spectrophotometer (UV-2450, manufactured by Shimadzu Corp.). Based on the peak intensity of the absorption wavelength (262 nm) resulting from the vinyl aromatic compound (styrene), using a calibration curve, the content of the vinyl aromatic monomer unit in the hydrogenated copolymer composition (styrene content ) was calculated.
- ⁇ MFR (melt flow rate) of hydrogenated copolymer composition> According to ISO 1133, the MFR [g/10 min] of the hydrogenated copolymer composition was measured under the conditions of a temperature of 230° C. and a load of 2.16 kg.
- a “180 degree peeling test” was performed in a temperature of 23° C. ⁇ 50% relative humidity, and two types of initial tackiness and tackiness promotion were evaluated.
- the initial tackiness the initial tack strength was measured.
- the adhesion accelerating property the produced tacky-adhesive film was adhered by the above-mentioned method, then heated in a gear oven at 80° C. for 1 hour to promote adhesion, and left in a measurement environment for 30 minutes. Then, the adhesive strength was measured by the method described above.
- the initial tackiness and the tackiness enhancement were evaluated according to the following criteria.
- the "180 degree peeling test” was performed according to the JIS Z0237 standard. Tables 2 and 4 below show the results of evaluation of initial tackiness and tackiness.
- the thickness of the adhesive layer was measured to measure the thickness unevenness during molding.
- the thickness measurement sample was cut from a coextrudate [sample width (TD direction) 50 cm, sample length (MD direction) 2 m, base material layer thickness 50 ⁇ m, adhesive layer thickness 8 ⁇ m] with a single-edged razor. Specifically, the center of the coextruded product in the TD direction and three points 5 cm from both ends were cut out 6 times at equal intervals in the MD direction, and a total of 18 samples were prepared.
- the thickness of the adhesive layer was determined by standard deviation by measuring the cross section in the MD direction of the sample with a microscope "VHX-2000: Keyence Corporation".
- ⁇ : Standard deviation of adhesive layer thickness is 2.0 ⁇ m or less
- Standard deviation of adhesive layer thickness is over 2.0 and 4.0 ⁇ m or less
- Standard deviation of adhesive layer thickness is over 4.0 ⁇ m
- ⁇ Variation coefficient of initial adhesive strength> The sample whose thickness was measured by the evaluation of ⁇ molding workability> was attached to a PMMA plate (arithmetic mean roughness of surface: 0.1 ⁇ m) at a temperature of 23° C. and 50% relative humidity, and a sample having a weight of 2 kg was attached. A rubber roll (diameter 10 cm) was rolled and attached, left for 30 minutes, and then the peeling speed was set to 300 mm/min to measure the adhesive strength. The standard deviation of the adhesive strength was calculated and the coefficient of variation was calculated.
- ⁇ Coefficient of variation of adhesive strength is less than 0.01
- ⁇ Coefficient of variation of adhesive strength is 0.01 or more and less than 0.05 x: Coefficient of variation of adhesive strength is 0.05 or more
- the hydrogenation catalyst used for the hydrogenation reaction of the hydrogenated copolymer composition was prepared by the following method. In a reaction vessel purged with nitrogen, 1 liter of dried and purified cyclohexane was charged, 100 mmol of bis( ⁇ 5-cyclopentadienyl)titanium dichloride was added, and an n-hexane solution containing 200 mmol of trimethylaluminum was added with sufficient stirring. The hydrogenated catalyst was prepared by adding and reacting at room temperature for about 3 days.
- A1, A2, B1 and B2 described below respectively represent the following polymer blocks.
- B2 Polymer block mainly composed of butadiene
- the obtained copolymer composition was continuously hydrogenated at 80° C. to obtain a hydrogenated copolymer composition.
- the amount of catalyst was 100 ppm, and the hydrogen pressure in the hydrogenation polymerization vessel was 0.8 MPa.
- 0.25 part by mass of a stabilizer octadecyl-3-(3,5-di-t-butyl-4-hydroxyphenyl)propionate was added to 100 parts by mass of the hydrogenated copolymer composition. did.
- the obtained copolymer composition was continuously hydrogenated at 80° C. to obtain a hydrogenated copolymer composition.
- the amount of catalyst was 100 ppm, and the hydrogen pressure in the hydrogenation polymerization vessel was 0.8 MPa.
- 0.25 part by mass of a stabilizer octadecyl-3-(3,5-di-t-butyl-4-hydroxyphenyl)propionate was added to 100 parts by mass of the hydrogenated copolymer composition. did.
- Table 1 shows the structures of the components (a) and (b) contained in the hydrogenated copolymer compositions obtained in Production Examples 1 to 19 and 36 to 54. The physical properties of these hydrogenated copolymer compositions are also shown in Table 1.
- the hydrogenated copolymer composition constituting the adhesive layer and the tackifying resin (Yasuhara Chemical Co., Ltd., trade name "YS Polystar G150") (10 parts by mass with respect to 100 parts by mass of the hydrogenated copolymer composition) are homogeneous. Mixed in.
- a cohesive film having a substrate layer thickness of 50 ⁇ m and an adhesive layer thickness of 8 ⁇ m was produced by integrally coextruding both layers by a coextrusion method.
- the hydrogenated copolymer composition, the adhesive composition, the adhesive film and the resin composition of the present invention are footwear, plastic modified, asphalt modified, adhesive and the like fields, household products, Widely used in packaging materials for home appliances and industrial parts, toys, etc.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Inorganic Chemistry (AREA)
- Adhesives Or Adhesive Processes (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Graft Or Block Polymers (AREA)
- Adhesive Tapes (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
Abstract
Description
[1]
ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(a);及び
ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(b);
を含む、水添共重合体組成物であって、
前記成分(a)が、5万~12万のピークトップ分子量を有し、
前記成分(b)が、前記成分(a)のピークトップ分子量の1.8倍~3.8倍のピークトップ分子量を有し、
前記水添共重合体組成物に含まれる共役ジエン単量体単位の二重結合の81mol%以上が水素添加されており、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位の含有量が、前記組成物を基準として、5~18質量%であり、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位のブロック化率が75質量%以上であり、
前記水添共重合体組成物のメルトフローレート(MFR)が4~20g/10分であり、
前記水添共重合体組成物のキャピロ粘度が300~600Pa・sであり、
前記キャピロ粘度をC[Pa・s]とし、前記MFRをM[g/10分]とした場合、(式1):
C≦-20.8M+754.2 (式1)
の関係を満たす、
前記水添共重合体組成物。
[2]
前記成分(a)/前記成分(b)が質量比率で60/40~10/90である、[1]に記載の水添共重合体組成物。
[3]
前記水添共重合体組成物に含まれる共役ジエン単量体単位のビニル化率が40mol%~70mol%である、[1]又は[2]に記載の水添共重合体組成物。
[4]
前記ビニル化率が40mol%~61.9mol%である、[3]に記載の水添共重合体組成物。
[5]
前記成分(a)及び前記成分(b)がそれぞれ以下の式で表される、[1]~[4]のいずれかに記載の水添共重合体組成物
成分(a): B2-A1-B1及び/又はB2-A1-B1-X
成分(b):(B2-A1-B1)nX
[式中、
A1は、ビニル芳香族単量体単位を主体とする重合体ブロックであり、
B1及びB2は、それぞれ独立して、共役ジエン単量体単位を主体とする重合体ブロックであり、
nは、2以上の整数であり、
Xは、カップリング剤の残基である]。
[6]
前記水添共重合体組成物に含まれるB1の含有量が、前記組成物を基準として、3~10質量%である、[5]に記載の水添共重合体組成物。
[7]
[1]~[6]のいずれかに記載の水添共重合体組成物、及び粘着付与剤樹脂を含む、粘接着材組成物。
[8]
[7]に記載の粘接着材組成物を含む、粘接着フィルム。
本実施形態の水添共重合体組成物は、
ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(a);及び
ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(b);を含み、
前記成分(a)が、5万~12万のピークトップ分子量を有し、
前記成分(b)が、前記成分(a)のピークトップ分子量の1.8倍~3.8倍のピークトップ分子量を有し、
前記水添共重合体組成物に含まれる共役ジエン単量体単位の二重結合の81mol%以上が水素添加されており、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位の含有量が、前記組成物を基準として、5~18質量%であり、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位のブロック化率が75質量%以上であり、
前記水添共重合体組成物のメルトフローレート(MFR)が4~20g/10分であり、
前記水添共重合体組成物のキャピロ粘度が300~600Pa・sであり
前記キャピロ粘度をC[Pa・s]とし、前記MFRをM[g/10分]とした場合、(式1):
C≦-20.8M+754.2 (式1)
の関係を満たす。
成分(a)に含まれる重合体ブロックBの含有量は、成分(a)を基準として、好ましくは84~96質量%であり、より好ましくは86~94質量%であり、更に好ましくは88~92質量%である。
成分(a)に含まれる重合体ブロックA及び重合体ブロックBの合計含有量は、成分(a)を基準として、好ましくは90質量%以上であり、より好ましくは95質量%以上であり、更に好ましくは100質量%である。
成分(b)に含まれる重合体ブロックBの含有量は、成分(b)を基準として、好ましくは84~96質量%であり、より好ましくは86~94質量%であり、更に好ましくは88~92質量%である。
成分(b)に含まれる重合体ブロックA及び重合体ブロックBの合計含有量は、成分(b)を基準として、好ましくは90質量%以上であり、より好ましくは95質量%以上であり、更に好ましくは100質量%である。
A1-B1、B2-A1-B1、A1-B1-A2、B2-A1-B1-A2、B2-A1-B1-X、(A1-B1)nX、又は(B2-A1-B1)nX
[式中、
A1及びA2は、それぞれ独立して、ビニル芳香族単量体単位を主体とする重合体ブロックであり、
B1及びB2は、それぞれ独立して、共役ジエン単量体単位を主体とする重合体ブロックであり、
nは2以上の整数であり、
Xはカップリング剤の残基である。]
・キャピラリーダイ内径:φ1.0mm
・キャピラリーダイ長:10.0mm
・流入角:180°
・ピストン径:φ9.510mm
・炉体経:φ9.55mm
C≦-20.8M+754.2 (式1)
の関係を満たす。式(1)は、複数の水添共重合体組成物のキャピロ粘度及びMFRと成形加工性との関係を詳細に検討した結果として導き出されたものである。MFRとキャピロ粘度はどちらも粘度の指標であるので、当然に相関があるものの、MFRは低剪断を反映し、キャピロ粘度は一定の条件に設定(温度230℃、剪断速度121.6sec-1)することで高剪断時の粘度を反映すると考えられる。本発明は、組成物の成型加工時には高せん断が与えられることに着目し、キャピロ粘度を低くすることで成形加工性厚みムラを抑制しうると考えた。一方、フィルム成形後に使用される環境は、低剪断が与えられる条件に近く、低MFRほど粘着力が高く、粘着昂進も起こりにくいと考えた。式(1)の関係を満たすことは、静的な粘度の指標であるMFRに対し、動的な粘度の指標であるキャピロ粘度が従来の設計ではなし得ない程度に低いことを意味する。MFRと式(1)の関係を両立することで、低剪断時と高剪時の粘度が上述のように好ましい範囲を満たすため、水添共重合体組成物の成型加工性と粘接着フィルムの粘着力、粘着昂進抑制のバランスに優れる。具体的には、例えば、フィルム押出機を使用した共押出成型において、粘着層に厚みムラが生じにくく、粘接着フィルムの粘着力、粘着昂進抑制のバランスも良好である。
ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(a);及び
ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(b);
を含む、水添共重合体組成物であって、
前記成分(a)が、5万~12万のピークトップ分子量を有し、
前記成分(b)が、前記成分(a)のピークトップ分子量の1.8倍~3.8倍のピークトップ分子量を有し、
前記水添共重合体組成物に含まれる共役ジエン単量体単位の二重結合の81mol%以上が水素添加されており、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位の含有量が、前記組成物を基準として、5~18質量%であり、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位のブロック化率が75質量%以上であるという要件を満たす必要があるが、これらを兼ね備えても、従来の一般的な構造のポリマーでは、高剪断時と低剪断時の粘度のギャップを実現できないので、次のような観点で具体的な構造を設計するのが好ましい。
上記ビニル芳香族単量体量の範囲内で、分子内にビニル芳香族単量体ブロックを複数設け、かつ、ビニル芳香族単量体ブロックを異鎖長化する。
これにより、ビニル芳香族単量体ブロック(大)とビニル芳香族単量体ブロック(小)を同一分子鎖内に形成できる。MFR測定のような低せん断での粘度は、ビニル芳香族単量体ブロック(小)の寄与が大きいために高粘度を維持する。一方、キャピロのような高せん断での粘度は、ビニル芳香族単量体ブロック(小)の影響が相対的に小さくなり低粘度化する。この結果、MFRを維持したまま、キャピロ粘度のみを低下させることが期待できる。
分子量や水添率などにもよるが、スチレンブロック(大)は、10000~20000程度の分子量であり、かつ、スチレンブロック(小)と比較し、5倍~10倍の分子量を持つ程度に設定するのが好ましい。
異分子間のビニル芳香族単量体ブロックの大きさが異なる2種類以上のポリマーを混ぜる。(同一分子鎖内のビニル芳香族単量体ブロックの大きさは同じ)
設計例1と同様に、大きさの異なるビニル芳香族単量体ブロックの影響で、高剪断時と低剪断時の粘度が異なる効果を期待できる。
キャピロのような高せん断条件下では、一旦分子差が乖離(開裂)して、その後元に戻るような特殊な官能基を分子鎖に導入する。これにより、低剪断時の粘度を維持したまま、高剪断時の粘度のみを低下させる効果が期待できる。このような効果を奏し得る官能基の例として、非共有結合性の官能基が挙げられ、例えば、水酸基、アミノ基、カルボキシル基、アミド基、ウレア基、イミド基、グアニジノ基などが挙げられる。
本実施形態の水添共重合体組成物は、例えば、有機溶媒中で、有機アルカリ金属化合物を重合開始剤として重合を行い、共重合体を得た後、水素化反応を行うことにより製造することができる。
第3級アミン系化合物としては、以下に限定されるものではないが、例えば、ピリジン、N,N,N’,N’-テトラメチルエチレンジアミン、トリブチルアミン、テトラメチルプロパンジアミン、1,2-ジピペリジノエタン、ビス[2-(N,N-ジメチルアミノ)エチル]エーテル等が挙げられる。これらは、1種のみを単独で用いてもよく、2種以上を併用してもよい。
これらの中でも、ナトリウムアルコキシドであるナトリウム-t-ブトキシド、ナトリウム-t-ペントキシドがさらにより好ましい。
ビニル化剤/有機リチウム化合物が0.2~3.0未満
アルカリ金属アルコキシド/有機リチウム化合物が0.3以下
なお、水添共重合体組成物のペレット成形体の大きさ、形状は特に限定されない。
水添共重合体組成物を成形して得られる粘接着性フィルムの粘着性の観点から、ペレットブロッキング防止剤は、ポリエチレン、及びエチレンビスステアリルアミド、ステアリン酸カルシウムが好ましい。
本実施形態の粘接着材組成物は、本実施形態の水添共重合体組成物を含む。
粘着付与樹脂は1種のみを単独で使用してもよく、2種類以上を混合して使用してもよい。
粘着付与樹脂の含有量が100質量部以下であれば、粘着昂進を効果的に抑制できる傾向にあるため好ましい。
その他、スチレン-エチレン-ブチレン(SEB)、スチレン-エチレン-プロピレン(SEP)といった構造のエラストマーも挙げられる。
本実施形態の粘接着材組成物の製造方法としては、特に限定されず、従来公知の方法により製造できる。
本実施形態の粘接着フィルムは、本実施形態の水添共重合体組成物、又は粘接着材組成物を含む。
本実施形態の粘接着フィルムの製造方法としては、以下に限定されるものではないが、例えば、本実施形態の水添共重合体組成物又は粘接着材組成物の溶液又は溶融物を前記所定の基材フィルム上に塗工する方法、フィルム押出機を用いて基材層と粘着層とを共押し出しする方法等が挙げられる。
外装材、内装表皮材等の自動車部材や、輸液バッグ、輸液チューブ等の医療部材、食品、衣料用包装分野においては、ポリプロピレン等の熱可塑性樹脂との樹脂組成物に求められる特性としては、引張破断伸びと低温耐衝撃性、低成形収縮性、及び前記各特性のバランス等が挙げられる。低成形収縮性とは、一般的に樹脂組成物を成型すると残留歪が生じその歪が経時で緩和等することで寸法変化するが、その変化が少ないことを意味する。本実施形態の水添共重合体組成物とポリプロピレン樹脂を含有する樹脂組成物は、引張破断伸びと低温耐衝撃性、低成形収縮性に優れている傾向にある。
ポリプロピレン樹脂としては、ホモポリプロピレンの他、ランダムポリプロピレン、ブロックポリプロピレンも使用できる。
ランダムポリプロピレンとしては、例えば、プロピレンとエチレンのランダム共重合体又はプロピレンと炭素数4~20のα-オレフィンのランダム共重合体などが挙げられる。
ポリプロピレンは1種類のみでもよいし、2種類以上のポリプロピレン樹脂を混合してもよい。2種類以上のポリプロピレン樹脂の混合物である場合、例えば、ホモポリプロピレンにエチレン-プロピレン共重合体を別々のプロセスで重合し、別々の固形状ペレット等を混合したものを用いてもよい。
無機充填剤の形状は、特に限定されず、鱗片状、球状、粒状、粉体、不定形状等であってもよい。これらは単独又は複数を組み合わせて使用できる。
本実施形態の樹脂組成物は、水添共重合体及びポリプロピレン樹脂、さらに必要に応じて他の成分を、その各成分の特性に応じた配合量でドライブレンドする方法、通常の高分子材料の混合に供される装置によって調製する方法が挙げられる。
本実施形態の成形体は、上述した本実施形態の樹脂組成物の成形体である。
成形体としては、シート状成形体(シート、フィルム)、チューブ、バック、医療用成形体、例えば医療用チューブ、医療用フィルム、自動車部材、例えば自動車内装表皮材、医療用輸液バック、並びに包装材、例えば食品包装材、及び衣料包装材等が挙げられるが、本実施形態の成形体は、上記に限定されるものではない。
また、ダイレクトブロー、インジェクションブロー等のブロー成形法、プレス成形法を採用することもできる。
ピークトップ分子量を、GPC〔装置:ウォーターズ製〕で測定した。
GPC測定では、溶媒としてテトラヒドロフランを用い、温度を40℃とした。
GPC測定で得られたクロマトグラフにおいて、最も高いピークトップを有するピーク(「第1ピーク」という。)及び次に高いピークトップを有するピーク(「第2ピーク」という。)のピークトップ分子量を、市販の標準ポリスチレンの測定から求めた検量線(標準ポリスチレンのピーク分子量を使用して作成)を使用して、求めた。第1ピーク及び第2ピークのうち、より小さいピークトップ分子量を有するものを成分(a)に対応するピークとし、より大きいピークトップ分子量を有するものを成分(b)に対応するピークとした。
上記GPC測定で得られたクロマトグラフにおける各ピークを、各ピーク間曲線の変曲点で垂直分割した。ピークの総面積に対する、第1ピークの面積の比率、及び第2ピークの面積の比率をそれぞれ求めた。第1ピークの面積比率及び第2ピークの面積比率に基づき、成分(a)及び成分(b)の質量比率を求めた。
一定量の水添共重合体組成物をクロロホルムに溶解し、紫外分光光度計(島津製作所製、UV-2450)にて測定した。ビニル芳香族化合物(スチレン)に起因する吸収波長(262nm)のピーク強度に基づき、検量線を用いて、水添共重合体組成物中のビニル芳香族単量体単位の含有量(スチレン含有量)を算出した。
水添共重合体組成物中のブロック化率、ビニル化率、水添率を、核磁気共鳴スペクトル解析(NMR)により、下記の条件で測定した。
なお、測定においては、水添反応後の反応液を、大量のメタノール中に投入させることで、水添共重合体組成物を沈殿させて回収した。次いで、水添共重合体組成物をアセトンで抽出し、抽出液を真空乾燥し、1H-NMR測定のサンプルとして用いた。1H-NMR測定の条件を以下に記す。
測定機器 :JNM-LA400(JEOL製)
溶媒 :重水素化クロロホルム
測定サンプル :ポリマーを水素添加する前後の抜き取り品
サンプル濃度 :50mg/mL
観測周波数 :400MHz
化学シフト基準:CDCl3(重水素化クロロホルム)
パルスディレイ:2.904秒
スキャン回数 :256回
パルス幅 :45°
測定温度 :26℃
ISO 1133に準拠し、温度230℃、荷重2.16kgの条件で、水添共重合体組成物のMFR[g/10分]を測定した。
キャピラリーレオメーター(株式会社東洋精機製作所製、商品名:キャピログラフ1D、型式:PM-C)を用いて、温度を230℃、せん断速度を12.2sec-1~12160sec-1の条件で水添共重合体組成物を連続的に測定し、121.6sec-1での値を求めた。なお、単位はPa・sとした。
粘着強度の測定装置として、万能引張圧縮試験機「テクノグラフTGE-500N:(株)ミネベア製」を用いた。
温度23℃×50%相対湿度中で、下記のとおり作製した粘接着フィルムを25mm幅にしたものをPMMA板(表面の算術平均粗さ:0.1μm)に貼り付け、さらに重さ2kgのゴムロール(直径10cm)を転がして貼り付け、30分間放置し、その後、引き剥がし速度を300mm/分として粘着強度を測定した。
温度23℃×50%相対湿度中で「180度引き剥がし試験」を行い、初期粘着性と粘着昂進性の2種類について評価した。
なお、初期粘着性については初期の粘着強度を測定した。粘着昂進性については、作製した粘接着フィルムを上述した方法で貼り付けた後、ギアーオーブン内で80℃×1時間加熱して粘着を促進させ、30分間測定環境に放置した。その後、上述した方法により粘着強度を測定した。初期粘着性及び粘着昂進性はそれぞれ、下記の基準により評価した。なお、前記「180度引き剥がし試験」は、JIS Z0237の規格に準じて行った。下記表2及び表4において、初期粘着性、粘着昂進性の評価結果を示す。
○:400g以上/25mmの粘着強度
△:300以上400g未満/25mmの粘着強度
×:0以上300g未満/25mmの粘着強度
・粘着昂進性
○:初期粘着強度に対して1.5倍以内の粘着強度
△:初期粘着強度に対して1.5倍超2.0倍以内の粘着強度
×:初期粘着強度に対して2.0倍超の粘着強度
粘着層厚みを測定し、成形時の厚みムラを測定した。厚み測定用サンプルは、共押出物[サンプル幅(TD方向)50cm、サンプル長さ(MD方向)2m、基材層厚み50μm、粘着層厚み8μm]から片刃カミソリで切り出した。具体的には、共押出物のTD方向の中央、及び両端から5cmの3箇所を、MD方向に等間隔で6回切り出し、合計で18個のサンプルを作成した。マイクロスコープ「VHX-2000:(株)キーエンス」で、サンプルのMD方向の断面を計測することで、粘着層の厚みを標準偏差で求めた。
○:粘着層厚みの標準偏差が2.0μm以下
△:粘着層厚みの標準偏差が2.0超4.0μm以下
×:粘着層厚みの標準偏差が4.0μm超
上記<成形加工性>の評価で厚みを測定したサンプルを、温度23℃×50%相対湿度中で、PMMA板(表面の算術平均粗さ:0.1μm)に貼り付け、さらに重さ2kgのゴムロール(直径10cm)を転がして貼り付け、30分間放置し、その後、引き剥がし速度を300mm/分として粘着強度を測定した。粘着強度における標準偏差を求め、変動係数を求めた。
○:粘着強度の変動係数が0.01未満
△:粘着強度の変動係数が0.01以上0.05未満
×:粘着強度の変動係数が0.05以上
表5に示す配合割合(質量部)に基づき、二軸押出機(日本製鋼所製「TEX-30αII」、シリンダー口径30mm)によって、設定温度220℃で溶融混練してポリプロピレン樹脂組成物のペレットを得た。
〈射出成型板〉
射出成形温度220℃、金型温度40℃で成形し、ポリプロピレン樹脂組成物の成形体(厚み2.0mm)を得た。ポリプロピレン樹脂としては、PM970A(PP/サンアロマー製;MFR=30、ブロックポリプロピレン)、無機充填剤としてP‐3(日本タルク製)を使用した。
(ポリプロピレン樹脂組成物の物性評価)
<低温引張伸び(-30℃)>
JIS K6251に準拠し、恒温槽付き引張試験機(ミネベア、Tg-5kN)を用いて、-30℃、3号ダンベル、クロスヘッドスピード500mm/分で次のとおり引張試験を実施し、破断伸びを評価した。
3:破断伸びが500%以上
2:破断伸びが300%以上500%未満
1:破断伸びが300%未満
<低温耐衝撃性(-30℃)>
ISO 179-1に準拠して評価した。
3:シャルピー衝撃強度が8kJ/m2以上
2:シャルピー衝撃強度が3kJ/m2以上8kJ/m2未満
1:シャルピー衝撃強度が3kJ/m2未満
<成形収縮性>
ISO 294-4に準拠して評価した。
3:収縮率が0.4%未満
2:収縮率が0.4%以上0.8%未満
1:収縮率が0.8%以上
(水添触媒の調製)
水添共重合体組成物の水素添加反応に用いた水添触媒を下記の方法で調製した。
窒素置換した反応容器に、乾燥、精製したシクロヘキサン1リットルを仕込み、ビス(η5-シクロペンタジエニル)チタニウムジクロリド100ミリモルを添加し、十分に攪拌しながらトリメチルアルミニウム200ミリモルを含むn-ヘキサン溶液を添加して、室温にて約3日間反応させ、水添触媒を調製した。
以下に記載するA1、A2、B1、B2は、それぞれ下記の重合体ブロックを示す。
A1、A2:スチレンを主体とする重合体ブロック
B1、B2:ブタジエンを主体とする重合体ブロック
<ポリマー1:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
攪拌装置及びジャケット付き槽型反応器(内容積:100L)を使用して、バッチ重合を以下の方法で行った。
シクロヘキサン38L、及びブタジエンモノマー3.0質量部を含有するシクロヘキサン溶液(ブタジエンモノマー濃度40質量%)を張り込み、N,N,N’,N’-テトラメチルエチレンジアミン(以下「TMEDA」ともいう。)を、下記n-ブチルリチウム(以下「Bu-Li」ともいう。)1モルに対して、0.8モル添加した。その後、温度40℃に調整した後、本製造例で反応器に投入するブタジエンモノマー及びスチレンモノマーの総量(以下、全モノマーとする。)100質量部に対して、Bu-Liを0.149質量部添加し、60分間、反応器内温度を45℃調整しながら反応を継続させた。
スチレンモノマー10.0質量部を投入し、その後さらに20分間、反応器内温度を50℃に調整しながら反応を継続させた。
ブタジエンモノマー87.0質量部を含有するシクロヘキサン溶液(ブタジエンモノマー濃度40質量%)を投入し、その後さらに40分間、反応器内温度を80℃に調整しながら反応を継続させて、ブタジエン-スチレン-ブタジエン共重合体を得た。
カップリング剤のテトラエトキシシランを、Bu-Li1モルに対して、0.2モル添加し、ブタジエン-スチレン-ブタジエン共重合体の一部を架橋させた上、反応停止剤としてエタノールをBu-Li1モルに対して0.3モル添加し、ブタジエン-スチレン-ブタジエン共重合体とその架橋物との混合物(共重合体組成物)を得た。
上述のようにして調製した水添触媒を用いて、得られた共重合体組成物を80℃で連続的に水素添加し、水添共重合体組成物を得た。触媒量は100ppm、水添重合器内の水素圧は0.8MPaであった。反応終了後に、水添共重合体組成物100質量部に対して、安定剤(オクタデシル-3-(3,5-ジ-t-ブチル-4-ヒドロキシフェニル)プロピオネート)0.25質量部を添加した。
<ポリマー2:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.160質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー3:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.172質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー4:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.142質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー5:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.147質量部、第2ステップのスチレンモノマー投入量を12.0質量部、第3ステップのブタジエンモノマー投入量を85.0質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー6:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.152質量部、第2ステップのスチレンモノマー投入量を8.0質量部、第3ステップのブタジエンモノマー投入量を85.0質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー7:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.138質量部、第4ステップの添加剤を安息香酸エチルに変更し、安息香酸エチルの添加量をBu-Li1モルに対して0.30モルとしたこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー8:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第4ステップのテトラエトキシシランの添加量をBu-Li1モルに対して0.1モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー9:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第4ステップのテトラエトキシシランの添加量をBu-Li1モルに対して0.32モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー10:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップの反応時間を30分に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー11:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのブタジエンモノマー張り込み量を11.5質量部、第3ステップのブタジエンモノマー投入量を78.5質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー12:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.157質量部、第2ステップのスチレンモノマー投入量を4.0質量部、第3ステップのブタジエンモノマー投入量を93.0質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー13:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.138質量部、第2ステップのスチレンモノマー投入量を20.0質量部、第3ステップのブタジエンモノマー投入量を77.0質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー14:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.152質量部、TMEDAの添加量をBu-Li1モルに対して0.49モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー15:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.147質量部、TMEDAの添加量をBu-Li1モルに対して1.5モル、第3ステップの反応器内温度を45℃に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー16:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第5ステップの水添触媒量を50ppmに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー17:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.28質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー18:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.080質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー19:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第4ステップの添加剤を四塩化珪素に変更し、四塩化珪素の添加量をBu-Li1モルに対して0.15モルとしたこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー20:スチレン-ブタジエンの水添物(A1-B1)>
攪拌装置及びジャケット付き槽型反応器(内容積:100L)を使用して、バッチ重合を以下の方法で行った。
シクロヘキサン38L、及びスチレンモノマー9.0質量部を張り込み、TMEDAを、下記Bu-Li1モルに対して、0.8モル添加した。その後、温度40℃に調整した後、本製造例で反応器に投入するブタジエンモノマー及びスチレンモノマーの総量(以下、全モノマーとする。)100質量部に対して、Bu-Liを0.149質量部添加し、20分間、反応器内温度を50℃調整しながら反応を継続させた。
ブタジエンモノマー91.0質量部を含有するシクロヘキサン溶液(ブタジエンモノマー濃度40質量%)を投入し、その後さらに20分間、反応器内温度を80℃に調整しながら反応を継続させて、スチレン-ブタジエン共重合体を得た。
反応停止剤としてエタノールを、Bu-Li1モルに対して、0.4モル添加し、その後、カップリング剤のテトラエトキシシランを、Bu-Li1モルに対して、0.3モル添加し、スチレン-ブタジエン共重合体の一部を架橋させて、スチレン-ブタジエン共重合体とその架橋物との混合物(共重合体組成物)を得た。
上述のようにして調製した水添触媒を用いて、得られた共重合体組成物を80℃で連続的に水素添加し、水添共重合体組成物を得た。触媒量は100ppm、水添重合器内の水素圧は0.8MPaであった。反応終了後に、水添共重合体組成物100質量部に対して、安定剤(オクタデシル-3-(3,5-ジ-t-ブチル-4-ヒドロキシフェニル)プロピオネート)0.25質量部を添加した。
<ポリマー21:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのBu-Li添加量を0.174質量部に変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー22:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのBu-Li添加量を0.206質量部に変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー23:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのBu-Li添加量を0.138質量部に変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー24:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのスチレンモノマー張り込み量を16質量部に変更し、第2ステップのブタジエンモノマー投入量を84質量部に変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー25:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのスチレンモノマー張り込み量を7質量部に変更し、第2ステップのブタジエンモノマー投入量を93質量部に変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー26:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのTMEDAの添加量を、Bu-Li1モルに対して、1.5モルに変更し、第2ステップの反応器内温度を45℃に変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー27:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのTMEDAの添加量を、Bu-Li1モルに対して、0.5モルに変更し、第2ステップの反応器内温度を45℃に変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー28:ブタジエン-スチレン-ブタジエン-スチレンの水添物(B1-A1-B2-A2)>
攪拌装置及びジャケット付き槽型反応器(内容積:100L)を使用して、バッチ重合を以下の方法で行った。
シクロヘキサン38L、及びブタジエンモノマー5.0質量部を含有するシクロヘキサン溶液(ブタジエンモノマー濃度40質量%)を張り込み、TMEDAを、下記Bu-Li1モルに対して、1.5モル添加した。その後、温度40℃に調整した後、本製造例で反応器に投入するブタジエンモノマー及びスチレンモノマーの総量(以下、全モノマーとする。)100質量部に対して、Bu-Liを0.057質量部添加し、60分間、反応器内温度を45℃調整しながら反応を継続させた。
スチレンモノマー7.0質量部を投入し、その後さらに20分間、反応器内温度を50℃に調整しながら反応を継続させた。
ブタジエンモノマー82.0質量部を含有するシクロヘキサン溶液(ブタジエンモノマー濃度40質量%)を投入し、その後さらに40分間、反応器内温度を45℃に調整しながら反応を継続させた。
スチレンモノマー6質量部を投入し、65℃に調整しながら20分間反応させた後、反応停止剤としてエタノールを、Bu-Li1モルに対して、0.9モル添加し、ブタジエン-スチレン-ブタジエン-スチレン共重合体を得た。
上述のようにして調製した水添触媒を用いて、得られた共重合体を80℃で連続的に水素添加し、水添共重合体を得た。触媒量は100ppm、水添重合器内の水素圧は0.8MPaであった。反応終了後に、水添共重合体100質量部に対して、安定剤(オクタデシル-3-(3,5-ジ-t-ブチル-4-ヒドロキシフェニル)プロピオネート)0.25質量部を添加した。
<ポリマー29:ブタジエン-スチレン-ブタジエン-スチレンの水添物(B1-A1-B2-A2)>
第1ステップのBu-Li添加量を0.059質量部に変更し、TMEDAの添加量をBu-Li1モルに対して0.8モルに変更し、第2ステップのスチレンモノマー投入量を6質量部に変更し、第3ステップの反応器内温度を80℃に変更し、第4ステップのスチレンモノマー投入量を4質量部に変更し、第3ステップのブタジエンモノマー投入量を85質量部に変更したこと以外は製造例28と同様にして、水添共重合体を得た。
<ポリマー30:ブタジエン-スチレン-ブタジエン-スチレンの水添物(B1-A1-B2-A2)>
第1ステップのBu-Li添加量を0.068質量部に変更したこと以外は製造例29と同様にして、水添共重合体を得た。
<ポリマー31:ブタジエン-スチレン-ブタジエン-スチレンの水添物(B1-A1-B2-A2)>
第1ステップのBu-Li添加量を0.093質量部に変更したこと以外は製造例29と同様にして、水添共重合体を得た。
<ポリマー32:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのTMEDAの添加量をBu-Li1モルに対して1.40モルに変更し、Bu-Li添加量を0.131質量部に変更し、スチレンモノマー張り込み量を10質量部に変更し、第2ステップのブタジエンモノマー投入量を90質量部に変更し、反応器内温度を45℃に変更し、第3ステップのエタノールを添加せず、カップリング剤をメチルジクロロシランに変更し、メチルジクロロシランの添加量をBu-Li1モルに対して0.3モルに変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー33:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのTMEDAの添加量をBu-Li1モルに対して0.45モルに変更し、Bu-Li添加量を0.157質量部に変更し、スチレンモノマー張り込み量を13質量部に変更し、第2ステップのブタジエンモノマー投入量を87質量部に変更し、第3ステップのエタノールを添加せず、カップリング剤をメチルジクロロシランに変更し、メチルジクロロシランの添加量をBu-Li1モルに対して0.25モルに変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー34:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのTMEDAの添加量をBu-Li1モルに対して0.30モルに変更し、Bu-Li添加量を0.103質量部に変更し、スチレンモノマー張り込み量を16質量部に変更し、第2ステップのブタジエンモノマー投入量を84質量部に変更し、第3ステップのエタノールを添加せず、カップリング剤をエポトートZX-1059(新日鐵住金化学株式会社)に変更し、エポトートZX-1059の添加量をBu-Li1モルに対して0.25モルに変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー35:スチレン-ブタジエンの水添物(A1-B1)>
第1ステップのTMEDAの添加量をBu-Li1モルに対して1.10モルに変更し、Bu-Li添加量を0.098質量部に変更し、スチレンモノマー張り込み量を15質量部に変更し、第2ステップのブタジエンモノマー投入量を85質量部に変更し、第3ステップのエタノールを添加せず、カップリング剤をエポトートZX-1059(新日鐵住金化学株式会社)に変更し、エポトートZX-1059の添加量をBu-Li1モルに対して0.275モルに変更したこと以外は製造例20と同様にして、水添共重合体組成物を得た。
<ポリマー36:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第4ステップのテトラエトキシシランの添加量をBu-Li1モルに対して0.28モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー37:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第4ステップのテトラエトキシシランの添加量をBu-Li1モルに対して0.25モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー38:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第4ステップのテトラエトキシシランの添加量をBu-Li1モルに対して0.14モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー39:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第4ステップのテトラエトキシシランの添加量をBu-Li1モルに対して0.16モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー40:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.097質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー41:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.107質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー42:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.204質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー43:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.141質量部、第2ステップのスチレンモノマー投入量を17.5質量部、第3ステップのブタジエンモノマー投入量を79.5質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー44:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.144質量部、第2ステップのスチレンモノマー投入量を14.5質量部、第3ステップのブタジエンモノマー投入量を82.5質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー45:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.155質量部、第2ステップのスチレンモノマー投入量を5.5質量部、第3ステップのブタジエンモノマー投入量を91.5質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー46:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのBu-Li添加量を0.153質量部、第2ステップのスチレンモノマー投入量を7.0質量部、第3ステップのブタジエンモノマー投入量を90.0質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー47:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップの反応時間を37分に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー48:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップの反応時間を46.5分に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー49:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのブタジエンモノマー張り込み量を1.5質量部、第3ステップのブタジエンモノマー投入量を89.0質量部に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー50:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのTMEDAの添加量をBu-Li1モルに対して1.5モルに変更し、第3ステップの反応器内温度を45℃に変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー51:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第1ステップのTMEDAの添加量をBu-Li1モルに対して0.59モルに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー52:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第5ステップの水添触媒量を63ppmに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー53:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第5ステップの水添触媒量を71ppmに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
<ポリマー54:ブタジエン-スチレン-ブタジエンの水添物(B2-A1-B1)>
第5ステップの水添触媒量を78ppmに変更したこと以外は製造例1と同様にして、水添共重合体組成物を得た。
粘着層を構成する水添共重合体組成物、及び粘着付与樹脂(ヤスハラケミカル社製、商品名「YSポリスターG150」)(水添共重合体組成物100質量部に対して10質量部)を均一に混合した。粘着層を構成する前記混合物と、基材層を構成するポリプロピレン(サンアロマー社製、商品名「PC684S」、MFR(230℃、2.16kg荷重)=7.5g/10分)とを、Tダイ方式共押出法によって両層を一体化して共押出し、基材層厚さ50μm、粘着層厚さ8μmの粘接着フィルムを作製した。
粘着層を構成する水添共重合体組成物又は水添共重合体、及び粘着付与樹脂(ヤスハラケミカル社製、商品名「YSレジンPX1150N」)(水添共重合体組成物100質量部に対して10質量部)を均一に混合した。粘着層を構成する前記混合物と、基材層を構成するポリプロピレン(サンアロマー社製、商品名「PC684S」、MFR(230℃、2.16kg荷重)=7.5g/10分)とを、Tダイ方式共押出法によって両層を一体化して共押出し、横幅50cm、基材層厚さ50μm、粘着層厚さ8μmの粘接着フィルムを作製した。
比較例29~34においては、本発明の構成要件を満たさないので、樹脂組成物は、引張破断伸び、低温耐衝撃性、低成形収縮性及び各種性能バランスにおいて、点数「1」が存在し、劣ることが分かった。
Claims (8)
- ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(a);及び
ビニル芳香族単量体単位を主体とする重合体ブロックと、共役ジエン単量体単位を主体とする重合体ブロックと、を含む成分(b);
を含む、水添共重合体組成物であって、
前記成分(a)が、5万~12万のピークトップ分子量を有し、
前記成分(b)が、前記成分(a)のピークトップ分子量の1.8倍~3.8倍のピークトップ分子量を有し、
前記水添共重合体組成物に含まれる共役ジエン単量体単位の二重結合の81mol%以上が水素添加されており、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位の含有量が、前記組成物を基準として、5~18質量%であり、
前記水添共重合体組成物に含まれるビニル芳香族単量体単位のブロック化率が75質量%以上であり、
前記水添共重合体組成物のメルトフローレート(MFR)が4~20g/10分であり、
前記水添共重合体組成物のキャピロ粘度が300~600Pa・sであり、
前記キャピロ粘度をC[Pa・s]とし、前記MFRをM[g/10分]とした場合、(式1):
C≦-20.8M+754.2 (式1)
の関係を満たす、
前記水添共重合体組成物。 - 前記成分(a)/前記成分(b)が質量比率で60/40~10/90である、請求項1に記載の水添共重合体組成物。
- 前記水添共重合体組成物に含まれる共役ジエン単量体単位のビニル化率が40mol%~70mol%である、請求項1又は2に記載の水添共重合体組成物。
- 前記ビニル化率が40mol%~61.9mol%である、請求項3に記載の水添共重合体組成物。
- 前記成分(a)及び前記成分(b)がそれぞれ以下の式で表される、請求項1~4のいずれかに記載の水添共重合体組成物
成分(a): B2-A1-B1及び/又はB2-A1-B1-X
成分(b):(B2-A1-B1)nX
[式中、
A1は、ビニル芳香族単量体単位を主体とする重合体ブロックであり、
B1及びB2は、それぞれ独立して、共役ジエン単量体単位を主体とする重合体ブロックであり、
nは、2以上の整数であり、
Xは、カップリング剤の残基である]。 - 前記水添共重合体組成物に含まれるB2の含有量が、前記組成物を基準として、3~10質量%である、請求項5に記載の水添共重合体組成物。
- 請求項1~6のいずれかに記載の水添共重合体組成物、及び粘着付与剤樹脂を含む、粘接着材組成物。
- 請求項7に記載の粘接着材組成物を含む、粘接着フィルム。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201980079056.1A CN113166344B (zh) | 2018-12-07 | 2019-12-06 | 氢化共聚物组合物、粘着粘结材料组合物和粘着粘结膜 |
| JP2020560053A JP7079859B2 (ja) | 2018-12-07 | 2019-12-06 | 水添共重合体組成物、粘接着材組成物及び粘接着フィルム |
| EP19893664.3A EP3892658A4 (en) | 2018-12-07 | 2019-12-06 | HYDROGENATED COPOLYMER COMPOSITION, ADHESIVE COMPOSITION AND ADHESIVE FOIL |
| US17/299,399 US20220049137A1 (en) | 2018-12-07 | 2019-12-06 | Hydrogenated copolymer composition, adhesive material composition, and adhesive film |
| KR1020217016700A KR102543409B1 (ko) | 2018-12-07 | 2019-12-06 | 수소 첨가 공중합체 조성물, 점접착재 조성물 및 점접착 필름 |
| IL283681A IL283681A (en) | 2018-12-07 | 2021-06-03 | A hydrogenated copolymer preparation, an adhesive material preparation and an adhesive layer |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018-229520 | 2018-12-07 | ||
| JP2018229520 | 2018-12-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020116625A1 true WO2020116625A1 (ja) | 2020-06-11 |
Family
ID=70974259
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/047864 WO2020116625A1 (ja) | 2018-12-07 | 2019-12-06 | 水添共重合体組成物、粘接着材組成物及び粘接着フィルム |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20220049137A1 (ja) |
| EP (1) | EP3892658A4 (ja) |
| JP (1) | JP7079859B2 (ja) |
| KR (1) | KR102543409B1 (ja) |
| CN (1) | CN113166344B (ja) |
| IL (1) | IL283681A (ja) |
| TW (1) | TWI740296B (ja) |
| WO (1) | WO2020116625A1 (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20200216726A1 (en) * | 2019-01-04 | 2020-07-09 | Tsrc Corporation | Pressure sensitive adhesive composition and protective film employing the same |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001085818A1 (fr) * | 2000-05-09 | 2001-11-15 | Asahi Kasei Kabushiki Kaisha | Copolymere bloc et composition contenant ce copolymere |
| JP2010538145A (ja) * | 2007-09-04 | 2010-12-09 | クレイトン・ポリマーズ・ユー・エス・エル・エル・シー | 明確なイソプレンおよびブタジエンの中間ブロックを有するブロックコポリマー、これを製造する方法、およびかかるブロックコポリマーの使用 |
| JP5158151B2 (ja) | 2010-08-19 | 2013-03-06 | Jsr株式会社 | 粘着性フィルム |
| JP2014114343A (ja) * | 2012-12-07 | 2014-06-26 | Kraton Polymers Us Llc | 塗装面の保護フィルム用粘着剤組成物およびその調製方法 |
| JP2014114345A (ja) | 2012-12-07 | 2014-06-26 | Kraton Polymers Us Llc | 塗装面の保護フィルム用粘着剤組成物およびその調製方法 |
| WO2015111669A1 (ja) * | 2014-01-23 | 2015-07-30 | 旭化成ケミカルズ株式会社 | ブロック共重合体組成物及び粘接着剤組成物 |
| WO2015178259A1 (ja) * | 2014-05-19 | 2015-11-26 | 旭化成ケミカルズ株式会社 | 水素添加ブロック共重合体組成物及び粘接着剤組成物 |
| JP2016186049A (ja) * | 2015-03-27 | 2016-10-27 | 日本エラストマー株式会社 | ブロック共重合体組成物、粘接着剤組成物及びこれらの製造方法 |
| WO2017033987A1 (ja) * | 2015-08-24 | 2017-03-02 | 旭化成株式会社 | 水素化ブロック共重合体並びにこれを用いたポリプロピレン系樹脂組成物及びその成型体 |
| WO2017159800A1 (ja) * | 2016-03-16 | 2017-09-21 | 株式会社クラレ | 樹脂組成物、ヒートシール剤、液体包装容器用フィルム、液体包装容器、液体排出部材および医療容器 |
| JP2017197609A (ja) | 2016-04-25 | 2017-11-02 | 旭化成株式会社 | 粘着性フィルム |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7001956B2 (en) | 2002-06-04 | 2006-02-21 | Kraton Polymers U.S. Llc | Articles prepared from hydrogenated block copolymers |
| JP5710373B2 (ja) * | 2011-05-11 | 2015-04-30 | 旭化成ケミカルズ株式会社 | 粘着剤用ブロック共重合体、粘着剤組成物及び表面保護フィルム |
| JP6069004B2 (ja) | 2013-02-04 | 2017-01-25 | 旭化成株式会社 | 粘着剤組成物及び表面保護フィルム |
| MY174144A (en) * | 2014-01-23 | 2020-03-11 | Asahi Chemical Ind | Adhesive composition and adhesive tape |
| WO2017126469A1 (ja) * | 2016-01-19 | 2017-07-27 | 旭化成株式会社 | 水添共重合体、組成物及び成形体 |
| SG11201809320RA (en) * | 2016-04-25 | 2018-11-29 | Asahi Chemical Ind | Hydrogenated block copolymer, polypropylene resin composition, and molded body |
-
2019
- 2019-12-06 EP EP19893664.3A patent/EP3892658A4/en active Pending
- 2019-12-06 US US17/299,399 patent/US20220049137A1/en active Pending
- 2019-12-06 TW TW108144804A patent/TWI740296B/zh active
- 2019-12-06 CN CN201980079056.1A patent/CN113166344B/zh active Active
- 2019-12-06 KR KR1020217016700A patent/KR102543409B1/ko not_active Application Discontinuation
- 2019-12-06 JP JP2020560053A patent/JP7079859B2/ja active Active
- 2019-12-06 WO PCT/JP2019/047864 patent/WO2020116625A1/ja unknown
-
2021
- 2021-06-03 IL IL283681A patent/IL283681A/en unknown
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001085818A1 (fr) * | 2000-05-09 | 2001-11-15 | Asahi Kasei Kabushiki Kaisha | Copolymere bloc et composition contenant ce copolymere |
| JP2010538145A (ja) * | 2007-09-04 | 2010-12-09 | クレイトン・ポリマーズ・ユー・エス・エル・エル・シー | 明確なイソプレンおよびブタジエンの中間ブロックを有するブロックコポリマー、これを製造する方法、およびかかるブロックコポリマーの使用 |
| JP5158151B2 (ja) | 2010-08-19 | 2013-03-06 | Jsr株式会社 | 粘着性フィルム |
| JP2014114343A (ja) * | 2012-12-07 | 2014-06-26 | Kraton Polymers Us Llc | 塗装面の保護フィルム用粘着剤組成物およびその調製方法 |
| JP2014114345A (ja) | 2012-12-07 | 2014-06-26 | Kraton Polymers Us Llc | 塗装面の保護フィルム用粘着剤組成物およびその調製方法 |
| WO2015111669A1 (ja) * | 2014-01-23 | 2015-07-30 | 旭化成ケミカルズ株式会社 | ブロック共重合体組成物及び粘接着剤組成物 |
| WO2015178259A1 (ja) * | 2014-05-19 | 2015-11-26 | 旭化成ケミカルズ株式会社 | 水素添加ブロック共重合体組成物及び粘接着剤組成物 |
| JP2016186049A (ja) * | 2015-03-27 | 2016-10-27 | 日本エラストマー株式会社 | ブロック共重合体組成物、粘接着剤組成物及びこれらの製造方法 |
| WO2017033987A1 (ja) * | 2015-08-24 | 2017-03-02 | 旭化成株式会社 | 水素化ブロック共重合体並びにこれを用いたポリプロピレン系樹脂組成物及びその成型体 |
| WO2017159800A1 (ja) * | 2016-03-16 | 2017-09-21 | 株式会社クラレ | 樹脂組成物、ヒートシール剤、液体包装容器用フィルム、液体包装容器、液体排出部材および医療容器 |
| JP2017197609A (ja) | 2016-04-25 | 2017-11-02 | 旭化成株式会社 | 粘着性フィルム |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20210087068A (ko) | 2021-07-09 |
| KR102543409B1 (ko) | 2023-06-14 |
| JPWO2020116625A1 (ja) | 2021-09-30 |
| TW202028342A (zh) | 2020-08-01 |
| JP7079859B2 (ja) | 2022-06-02 |
| EP3892658A1 (en) | 2021-10-13 |
| IL283681A (en) | 2021-07-29 |
| US20220049137A1 (en) | 2022-02-17 |
| CN113166344A (zh) | 2021-07-23 |
| EP3892658A4 (en) | 2022-01-19 |
| TWI740296B (zh) | 2021-09-21 |
| CN113166344B (zh) | 2023-08-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6648145B2 (ja) | 水素化ブロック共重合体、ポリプロピレン樹脂組成物及び成形体 | |
| JP6716612B2 (ja) | 水添共重合体、組成物及び成形体 | |
| JP6703601B2 (ja) | 水素化ブロック共重合体、ポリプロピレン樹脂組成物及び成形体 | |
| JP6985395B2 (ja) | 感圧型接着剤用組成物、感圧型接着剤用材料、感圧型接着剤の形成方法、感圧型接着剤、多層フィルム及び包装体 | |
| RU2713173C1 (ru) | Композиция смолы и формованное изделие | |
| JP7079859B2 (ja) | 水添共重合体組成物、粘接着材組成物及び粘接着フィルム | |
| JP7195417B2 (ja) | 水添共重合体、粘着フィルム、樹脂組成物、及び成形体 | |
| JP2017197609A (ja) | 粘着性フィルム | |
| JP2022099692A (ja) | 水添共重合体、及び成形体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19893664 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020560053 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20217016700 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2019893664 Country of ref document: EP Effective date: 20210707 |