WO2020004490A1 - コンドロイチン硫酸プロテオグリカン-5に結合する抗体 - Google Patents
コンドロイチン硫酸プロテオグリカン-5に結合する抗体 Download PDFInfo
- Publication number
- WO2020004490A1 WO2020004490A1 PCT/JP2019/025450 JP2019025450W WO2020004490A1 WO 2020004490 A1 WO2020004490 A1 WO 2020004490A1 JP 2019025450 W JP2019025450 W JP 2019025450W WO 2020004490 A1 WO2020004490 A1 WO 2020004490A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- amino acid
- acid sequence
- seq
- cspg5
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
- C07K16/3053—Skin, nerves, brain
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/08—Peptides, e.g. proteins, carriers being peptides, polyamino acids, proteins
- A61K51/10—Antibodies or immunoglobulins; Fragments thereof, the carrier being an antibody, an immunoglobulin or a fragment thereof, e.g. a camelised human single domain antibody or the Fc fragment of an antibody
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/10—Cells modified by introduction of foreign genetic material
- C12N5/12—Fused cells, e.g. hybridomas
- C12N5/16—Animal cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5044—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics involving specific cell types
- G01N33/5058—Neurological cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6854—Immunoglobulins
- G01N33/6857—Antibody fragments
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6893—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to diseases not provided for elsewhere
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
- C07K2317/522—CH1 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
- C07K2317/524—CH2 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
- C07K2317/526—CH3 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/77—Internalization into the cell
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/33—Fusion polypeptide fusions for targeting to specific cell types, e.g. tissue specific targeting, targeting of a bacterial subspecies
Definitions
- the present invention relates to, for example, an antibody that binds to chondroitin sulfate proteoglycan-5 (Chondroitin sulfate proteoglycan 5; CSPG5), the antibody fragment, a hybridoma that produces the antibody or the antibody fragment, a base sequence encoding the antibody or the antibody fragment.
- chondroitin sulfate proteoglycan-5 Chodroitin sulfate proteoglycan 5; CSPG5
- a transformed cell containing a vector containing the nucleic acid, a method for producing the antibody or the antibody fragment, a composition containing the antibody or the antibody fragment, an antigen present in the brain using the antibody or the antibody fragment The present invention relates to a method for detecting or measuring, a method for diagnosing or treating a brain disease, a method for improving the brain retention of antibodies, a method for increasing the amount of antibodies in the brain, and the like.
- muromonab-CD3 Since the mouse anti-CD3 antibody, muromonab-CD3 (OKT3) was approved by the FDA in 1986 as the first antibody drug, a number of antibody drugs have been developed. In 1994, a chimeric antibody abciximab in which the variable region of a mouse antibody and the constant region of a human antibody were linked was approved in order to reduce the antigenicity of the mouse antibody.
- CDR complementarity determining region
- Non-Patent Document 1 a fully human anti-TNF ⁇ antibody alimimumab was approved in 2002 as the first antibody obtained by phage display technology. More than 60 antibody drugs using CD20, CD52, TNF ⁇ , HER2, EGFR and the like as target antigens have already been approved (Non-Patent Document 1).
- antibodies are a widely recognized drug format.
- the majority of antibody drugs approved so far target cancer and immune disorders, accounting for more than 75% of the total.
- Non-Patent Document 2 In the treatment of central nervous system diseases, the importance of biologics such as antibodies is increasing. Monoclonal antibodies against amyloid ⁇ have been studied in Alzheimer's disease, and various neurotrophic factors having neuroprotective effects (brain-derived neurotrophic factors) have been studied. Factor (BDNF, glial-derived neurotrophic factor (GDNF)) has been reported in animal models to exhibit a neuroprotective effect in central nervous system diseases (Non-Patent Document 2).
- BDNF glial-derived neurotrophic factor
- BBB blood-brain barrier
- the blood-brain barrier has a physical / non-specific control mechanism by cell-cell junctions of vascular endothelial cells and a substrate-specific discharge mechanism by a discharge transporter, protecting the central nervous system from foreign substances or drugs, It plays an important role in maintaining homeostasis.
- Non-Patent Document 11 a method has been reported in which iduronate 2-sulfatase is administered into a patient's brain in order to prevent the progression of brain damage in patients with Hunter syndrome (mucopolysaccharidosis II) (Patent Document 1).
- direct administration into the intrathecal or brain is highly invasive (Non-Patent Document 11).
- Targeted cerebrovascular endothelial expression receptors include, for example, transferrin receptor, insulin receptor Body, insulin-like growth factor receptor, low density lipoprotein receptor family (LDLRf) and the like.
- anti-transferrin receptor antibodies include bispecific antibodies of anti-transferrin receptor antibody and anti-beta-secretase (BACE1) antibody (Patent Documents 2 and 3, and Non-Patent Documents 12 and 13), and anti-amyloid ⁇ antibody A fusion antibody (Patent Literature 4 and Non-Patent Literature 14) in which a monovalent antibody of an anti-transferrin receptor is fused to the carboxyl terminus side of is described.
- Non-Patent Document 13 Brain delivery of an anti-transferrin receptor antibody and an anti-BACE1 antibody by a bispecific antibody has been reported to increase the amount of antibody uptake in the brain by about 4 times that of a control when the antibody is administered at 20 mg / kg body weight in mice.
- Non-Patent Document 15 a technique has been reported in which a drug is encapsulated in a liposome having an anti-transferrin receptor antibody on the surface so that the drug passes through the blood-brain barrier. It has been reported that the fusion with an anti-rat transferrin receptor antibody and an immunomicelle increases the brain uptake in rats by about 2 to 5 times (Non-Patent Document 15).
- Non-patent Document 17 It has been reported that in rhesus monkeys, the amount of uptake in the brain 2 hours after administration of the fusion antibody of the labeled anti-human insulin receptor antibody and GDNF is about 15 times that of GDNF (Non-patent Document 17). ).
- Non-Patent Document 20 transferrin receptor and insulin receptor are expressed not only in cerebral vascular endothelial cells but also in the whole body such as the liver. Occurs (Non-Patent Document 20). Furthermore, since the antigen is expressed systemically, the half-life of the antibody in blood is short (Non-Patent Document 12).
- Fc5 is a heavy chain variable region (Variable ⁇ domain ⁇ of ⁇ Heavy ⁇ chain ⁇ of ⁇ Heavy ⁇ chain ⁇ antibody (hereinafter referred to as VHH)) antibody of a single-domain heavy chain antibody derived from a llama, and a fusion of Fc5 and human Fc with brain IgG is compared with control IgG. Is shown in the in vitro BBB model and the rat in vivo model.
- Non-patent Documents 24 and 25 fetal Fc receptor (neonal Fc receptor; FcRn)
- the half-life in the brain after intracerebral administration is as short as 48 minutes (Non-Patent Document 24).
- CSPG5 is a transmembrane chondroitin sulfate proteoglycan, and is mainly restricted to central nervous system tissues (Non-Patent Documents 26, 27 and 28). Immunohistochemical staining confirms staining for neurons (neural cells) and / or astrocytes, such as neuropils, dendrites and nerve fibers (Non-Patent Documents 28, 29, 30, and 36). Expression of CSPG5 in the rat central nervous system is observed from the fetal period, peaks at 3 weeks after birth, and decreases to about half of the peak level in the adult period (Non-patent Documents 26 and 30).
- CSPG5 exists in the form of proteoglycans in developing central nervous system tissues and in the form of non-proteoglycans in mature central nervous system tissues (NPLs 28 and 30).
- CSPG5 has a 120 kDa core protein.
- the core protein is divided into five distinct structures: an N-terminal domain to which chondroitin sulfate chains bind, an acidic amino acid cluster, a cysteine-rich domain containing an epidermal growth factor (EGF) -like module, a transmembrane segment and a cytoplasmic domain.
- EGF epidermal growth factor
- Non-Patent Documents 26 and 27 The extracellular region of CSPG5 binds to tenascin-C and tenascin-R via acidic amino acid clusters (Non-patent Documents 29, 32 and 33) and interacts with the ErbB3 fusion protein (Non-patent Document 34).
- Boado RJ. Methods Enzymology, 503, 269-292, 2012 Boado RJ., Etal., Drug Metab.Dispos., 37 (12), 2299-2304, 2009 Boado RJ., Et al., J. Pharmacol. Exp. Ther., 333 (3), 961-969, 2010 Boado RJ., Etal., Bioconjugate Chem., 1, 97-104, 2012 Yun Zhang.et al., J. Pharmacol. Exp.
- the present invention relates to, for example, a CSPG5-binding molecule that binds to CSPG5 and a method using the molecule.
- a CSPG5-binding molecule that binds to CSPG5 and a method using the molecule.
- an antibody that binds to CSPG5 or the antibody fragment a hybridoma that produces the antibody or the antibody fragment, a nucleic acid that includes a base sequence encoding the antibody or the antibody fragment, and a transformation that includes a vector that includes the nucleic acid Cell
- a method for producing the antibody or the antibody fragment a composition containing the antibody or the antibody fragment
- a method for detecting or measuring an antigen present in the brain using the antibody or the antibody fragment and diagnosing or treating a brain disease
- the present invention provides a CSPG5-binding molecule that binds to CSPG5 and a method using the molecule, specifically, an antibody that binds to CSPG5 or an antibody fragment thereof.
- the present invention relates to the following (1) to (23).
- CSPG5 chondroitin sulfate proteoglycan-5
- the antibody or the antibody fragment thereof according to any one of (1) to (3), wherein the antibody is 1 selected from the group consisting of the following (a) to (s); (A) the amino acid sequences of the complementarity determining regions (CDRs) 1-3 of the heavy chain variable region (VH) include the amino acid sequences shown in SEQ ID NOs: 3, 4 and 5, respectively, and the light chain variable region (VL )) Wherein the amino acid sequences of CDRs 1-3 comprise the amino acid sequences set forth in SEQ ID NOs: 8, 9 and 10, respectively.
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 13, 14 and 15, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 18, 19 and 20, respectively
- An antibody comprising the amino acid sequence described
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 23, 24 and 25, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 28, 29 and 30, respectively.
- an antibody comprising the amino acid sequence described (D) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 33, 34 and 35, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 38, 39 and 40, respectively
- An antibody comprising the amino acid sequence described (E) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 43, 44 and 45, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 48, 49 and 50, respectively
- An antibody comprising the amino acid sequence described (F) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOS: 53, 54 and 55, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 58, 59 and 60, respectively
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOS: 113, 114 and 115, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 118, 119 and 120, respectively.
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences described in SEQ ID NOs: 133, 134 and 135, respectively, and the amino acid sequences of CDRs 1-3 of VL are represented by SEQ ID NOs: 138, 139 and 140, respectively.
- An antibody comprising the amino acid sequence described, (O) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 143, 144 and 145, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 148, 149 and 150, respectively
- an antibody comprising an amino acid sequence having 85% or more homology with the amino acid sequence of the antibody.
- the antibody is one selected from the group consisting of the following (A) to (P);
- C an antibody wherein the amino acid sequence of VH comprises the amino acid sequence of SEQ ID NO: 22 and the amino acid sequence of VL comprises the amino acid sequence of SEQ ID NO: 27,
- E an antibody wherein the amino acid sequence of VH comprises the amino
- Antibody fragments are Fab, Fab ′, F (ab ′) 2 , single chain antibody (scFv), dimerized V region (diabody), disulfide stabilized V region (dsFv), heavy chain antibody
- scFv single chain antibody
- diabody dimerized V region
- dsFv disulfide stabilized V region
- VHH chain variable region
- CDR CDR
- the antibody and the antibody fragment according to any one of the above.
- the antibody, the antibody fragment, the fusion antibody or the hybridoma producing the fusion antibody fragment according to any one of (1) to (12).
- a nucleic acid comprising the antibody according to any one of (1) to (12), the antibody fragment, the fusion antibody, or a nucleotide sequence encoding the fusion antibody fragment.
- a transformed cell comprising the vector containing the nucleic acid according to (14).
- the hybridoma according to (13) or the transformed cell according to (15) is cultured, and the antibody, the antibody fragment, the fusion antibody or the antibody according to any one of (1) to (12) is cultured from the culture solution.
- a composition comprising the antibody according to any one of (1) to (12), the antibody fragment, the fusion antibody, or the fusion antibody fragment.
- (21) Diagnosis or treatment of a brain disease using the antibody according to any one of (1) to (12), the antibody fragment, the fusion antibody or the fusion antibody fragment, or the composition according to (17). how to.
- (22) An antibody, the antibody fragment, the antibody fragment, the fusion antibody or the fusion antibody fragment according to any one of (1) to (12), or the composition according to (17), A method for improving brain retention of a fusion antibody or the fusion antibody fragment.
- (23) Using the antibody according to any one of (1) to (12), the antibody fragment, the fusion antibody or the fusion antibody fragment, or the composition according to (17), the amount of antibody in the brain; A method of increasing the amount of the antibody fragment, the amount of the fusion antibody, or the amount of the fusion antibody fragment.
- the CSPG5 binding molecule of the present invention not only increases the brain retention of the binding molecule itself by specifically binding to CSPG5, but also modifies the other molecule of interest to a CSPG5 binding molecule to convert the molecule of interest into the brain. It can be applied to the treatment of brain diseases by transporting it to and retaining it.
- Specific CSPG5 binding molecules of the present invention include antibodies or antibody fragments thereof.
- the antibody or the antibody fragment of the present invention is an antibody or an antibody fragment having brain retention by binding to CSPG5 in the brain.
- the antibody or the antibody fragment of the present invention comprises a composition for detecting or measuring an antigen present in the brain (CSPG5, or CSPG5 and other antigens present in the brain), a composition for diagnosing a brain disease, And it can be used as a pharmaceutical composition for treating brain diseases.
- FIGS. 1A to 1D show the results of measuring the concentration of each antibody in the tissue.
- FIG. 1 (A) shows the antibody concentration in serum 3 days after antibody administration. The vertical axis indicates the antibody concentration (ng / mL), and the horizontal axis indicates the administered antibody.
- FIG. 1 (B) shows the antibody concentration in brain tissue three days after the antibody administration. The vertical axis indicates the antibody concentration (ng / g brain), and the horizontal axis indicates the administered antibody.
- FIG. 1 (C) shows the antibody concentration in serum 9 days after antibody administration. The vertical axis indicates the antibody concentration (ng / mL), and the horizontal axis indicates the administered antibody.
- FIG. 1 (D) shows the antibody concentration in brain tissue 9 days after antibody administration.
- FIGS. 2A and 2B show the results of measuring the concentration of each antibody in the tissue.
- FIG. 2 (A) shows the antibody concentration in serum 7 days after antibody administration.
- the vertical axis indicates the antibody concentration (ng / mL), and the horizontal axis indicates the administered antibody.
- FIG. 2 (B) shows the antibody concentration in brain tissue 7 days after antibody administration.
- the vertical axis indicates the amount of antibody eluted (ng / g brain), and the horizontal axis indicates the administered antibody.
- the antibody concentration is shown as a value converted from the molar concentration by the molecular weight (150 kDa) of the monoclonal antibody.
- FIGS. 3 (A) to 3 (D) show the results of measuring the concentration of each antibody in the tissue.
- FIGS. 3 (A) and 3 (C) show the antibody concentration in serum 7 days after antibody administration.
- the vertical axis indicates the antibody concentration (ng / mL), and the horizontal axis indicates the administered antibody.
- 3 (B) and (D) show the antibody concentration in brain tissue 7 days after antibody administration.
- the vertical axis indicates the amount of antibody eluted (ng / g brain), and the horizontal axis indicates the administered antibody.
- the antibody concentration is shown as a value converted from the molar concentration by the molecular weight (150 kDa) of the monoclonal antibody.
- FIGS. 4 (A) and (B) show the results of mouse brain migration imaging evaluation of each antibody.
- FIG. 4 (A) shows a brain imaging image 9 days after the antibody administration.
- FIG. 4 (B) shows the ratio of the amount of fluorescence in the brain corrected by the fluorescence intensity of the administered antibody to the ratio of the anti-AVM antibody.
- the vertical axis indicates the ratio of the anti-AVM antibody, and the horizontal axis indicates the administered antibody.
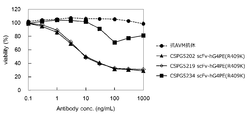
- FIG. 5 shows the results of internalization analysis of CSPG5202 @ scFv-hG4PE (R409K), CSPG5219 @ scFv-hG4PE (R409K), and CSPG5234 @ scFv-hG4PE (R409K) in hCSPG5 / L929 # 09.
- the horizontal axis indicates the antibody concentration (ng / mL), and the vertical axis indicates the cell viability (%).
- the dotted line shows the anti-AVM antibody as a negative control, and the solid line shows the sample.
- a black triangle marker ( ⁇ ) shows data of CSPG5202 scFv-hG4PE (R409K), a diamond-shaped marker ( ⁇ ) shows data of CSPG5219 scFv-hG4PE (R409K), and a black square marker ( ⁇ ) shows data of CSPG5234 scFv-hG4PE (R409K).
- FIG. 6 shows the results of internalization analysis of CSPG5202 @ scFv-hG4PE (R409K), CSPG5219 @ scFv-hG4PE (R409K), and CSPG5234 @ scFv-hG4PE (R409K) at IMR-32.
- the horizontal axis indicates the antibody concentration (ng / mL), and the vertical axis indicates the cell viability (%).
- the dotted line shows the anti-AVM antibody as a negative control, and the solid line shows the sample.
- a black triangle marker ( ⁇ ) shows data of CSPG5202 scFv-hG4PE (R409K), a diamond-shaped marker ( ⁇ ) shows data of CSPG5219 scFv-hG4PE (R409K), and a black square marker ( ⁇ ) shows data of CSPG5234 scFv-hG4PE (R409K).
- the present invention relates to an antigen binding molecule that binds to CSPG5. More specifically, the present invention relates to an antibody or an antibody fragment that binds to CSPG5.
- the CSPG5 binding molecule of the present invention may be in any molecular form as long as it specifically binds to CSPG5 and the molecule stays in the brain. Proteins, nucleic acids, organically synthesized low molecular weight compounds / high molecular weight compounds And any other molecule. Specifically, it may be any of a recombinant protein, an antibody, an aptamer, a low-molecular compound obtained by low-molecular screening, etc., but preferably an antibody and an antibody fragment thereof.
- the CSPG5 binding molecule is preferably a molecule that binds to the extracellular region of CSPG5.
- CSPG5 is a transmembrane chondroitin sulfate proteoglycan.
- the full length of human CSPG5 including the signal sequence is composed of 539 amino acids and is mainly present in central nervous system tissues, and is involved in maturation of cerebellar ⁇ -aminobutyric acid-operating synapses and molecular interactions during the development of central nervous tissues. Plays a role.
- the animal species of CSPG5 to which the CSPG5-binding molecule of the present invention binds include mouse, rat, rhesus monkey, and / or human, but are not particularly limited to these species. Animal species can be selected.
- the antibody of the present invention is used for human pharmaceutical use, the antibody is preferably an antibody that binds at least to human CSPG5.
- the human CSPG5 includes a polypeptide comprising the amino acid sequence of SEQ ID NO: 160 or the amino acid sequence of NCBI accession number NP — 00655.2, the amino acid sequence of SEQ ID NO: 160 or the polypeptide of NCBI accession number NP — 00655.2.
- a polypeptide having the amino acid sequence of SEQ ID NO: 160 or the amino acid sequence represented by NCBI accession number NP — 00655.2, in which one or more amino acids are deleted, substituted or added, can be obtained by site-directed mutagenesis.
- the number of amino acids to be deleted, substituted or added is not particularly limited, but is preferably 1 to several tens, for example, 1 to 20, more preferably 1 to several, for example, 1 to 5 amino acids. It is.
- examples of the gene encoding human CSPG5 include the nucleotide sequence of SEQ ID NO: 159 or the nucleotide sequence of NCBI accession number NM — 0065743.
- the nucleotide sequence of SEQ ID NO: 159 or the nucleotide sequence of NCBI accession number NM_0065743.3 which encodes a polypeptide having one or more bases deleted, substituted or added, and having a CSPG5 function.
- Including base sequence It consists hybridizing DNA with NA under stringent conditions and also such gene encoding a polypeptide having the function of CSPG5 included in the gene encoding the CSPG5 in the present invention.
- Examples of the DNA that hybridizes under stringent conditions include colony hybridization and plaque hybridization using a DNA containing the nucleotide sequence of SEQ ID NO: 159 or the nucleotide sequence of NCBI accession number NM — 00654.33 as a probe.
- a hybridizable DNA obtained by a hybridization method, a Southern blot hybridization method, a DNA microarray method, or the like.
- 0.7 to 1.0 mol / L of sodium chloride is present using a filter or a slide glass on which DNA from a hybridized colony or plaque, or a PCR product or oligo DNA having the sequence has been immobilized. Under the hybridization method at 65 ° C.
- CSPG5 includes maturation of cerebellar ⁇ -aminobutyric acid-operating synapses during molecular development of central nervous tissue and involvement in intermolecular interactions.
- Gene polymorphisms are often found in the nucleotide sequences of genes encoding eukaryotic proteins.
- a gene having a small mutation in the nucleotide sequence due to such polymorphism is also included in the gene encoding CSPG5 in the present invention.
- the numerical value of the homology in the present invention may be a numerical value calculated using a homology search program known to those skilled in the art, unless otherwise specified.
- BLAST J. Mol. Biol ., ⁇ 215, ⁇ 403 ⁇ (1990)
- amino acid sequences such as numerical values calculated using default parameters
- BLAST2 Nucleic Acids Res., 25, 3389 (1997), Genome Res., 7, 649 (1997)
- the default parameters are 5 if G (Cost @ to ⁇ open ⁇ gap) is a nucleotide sequence, 11 if an amino acid sequence, 2 if -E (Cost @ to ⁇ extend @ gap) is a nucleotide sequence, and 1 if -E (Cost @ to @ extend @ gap) is a nucleotide sequence.
- -Q Pulalty @ nucleotide @ mismatch
- -r forward @ nucleotide @ match
- -e exitpect @ value
- -W wordsize
- Is 3 20 when -y [Dropoff (X) for ⁇ blast ⁇ extensions ⁇ in ⁇ bits] is blastn, 7 when a program other than blastn is used, and -X (X ⁇ dropoff @ value @ for $).
- the applied @ alignment in bits is 15 and the -Z (final @ X @ dropoff @ value @ gapped @ alignment in bits) is 50 for blastn, and 25 for programs other than blastn (http://www.ncbi.nlm.com). nih.gov/blast/htmL/blastcgihelp.htmL).
- Polypeptides containing partial sequences of the above-mentioned various CSPG5 amino acid sequences can be prepared by methods known to those skilled in the art. Specifically, it can be produced by deleting a part of the DNA encoding the amino acid sequences of the above-mentioned various CSPG5, and culturing a transformant into which an expression vector containing the same has been introduced. Further, a polypeptide having an amino acid sequence in which one or more amino acids have been deleted, substituted or added in the amino acid sequence of various CSPG5 can be obtained by the same method as described above.
- a polypeptide comprising the amino acid sequence of various CSPG5, or a polypeptide having an amino acid sequence in which one or more amino acids are deleted, substituted or added in the amino acid sequence of various CSPG5 can be obtained by the fluorenylmethyloxycarbonyl (Fmoc) method. , T-butyloxycarbonyl (tBoc) method and the like.
- the extracellular region of human CSPG5 refers to the amino acid sequence from the 31st to the 423rd amino acid in the amino acid sequence described in SEQ ID NO: 160 or NCBI accession number NP — 00655.2.
- mouse CSPG5 refers to the 31st to 423th amino acid sequence in the amino acid sequence described in SEQ ID NO: 162 or NCBI accession number NP_038912.3.
- the extracellular region of rhesus monkey CSPG5 refers to the 31st to 414th amino acid sequence in the amino acid sequence described in SEQ ID NO: 164 or NCBI accession number AFE76329.1.
- the fact that the antibody of the present invention binds to the extracellular region of CSPG5 means that the binding of the antibody of the present invention to a CSPG5 expressing cell or a recombinant CSPG5 protein can be measured by Enzyme-Linked Immunosorbent Assay (ELISA), flow cytometry or surface plasmon resonance. It can be confirmed by measuring using a method or the like.
- ELISA Enzyme-Linked Immunosorbent Assay
- flow cytometry flow cytometry
- surface plasmon resonance It can be confirmed by measuring using a method or the like.
- known immunological detection methods (Monoclonal Antibodies-Principles and practice, Third Edition, Academic Press (1996), Antibodies-A Laboratory Manual, Cold Spring Harbor Laboratory (1988), Monoclonal Antibody Experiment Manual, Kodansha Scientific (1987)].
- the CSPG5-binding molecule of the present invention is a molecule having brain retention by specifically binding to CSPG5 in the brain.
- the antibody of the present invention has brain retention by binding to CSPG5 in the brain Antibodies.
- the antibody of the present invention when administered to the periphery of an animal, penetrates the blood through the blood-brain barrier of the brain from the periphery, migrates into the brain, and binds to CSPG5 in the brain. It is.
- the antibody of the present invention is preferably an antibody having excellent brain retention or an antibody having improved brain retention.
- the term “cerebral retention” refers to the property that a subject stays in the brain when the subject is administered to a test animal. That is, it is at least selected from an increase in translocation into the brain, an increase in accumulation in the brain, a decrease in translocation from the brain to the outside of the brain, a decrease in excretion from the brain to the outside of the brain, and a decrease in degradation in the brain. Either one means that the concentration of the subject in the brain (or the amount in the brain) increases, or that the concentration of the subject is detectably constant.
- brain retention is excellent, brain retention is high, or that the brain retention is improved, when the subject is administered to the test animal, compared to the control, compared to the control, the same day after administration Means that the concentration of the target in the brain (or the amount in the brain) increases, or that the target exists in the brain at a constant concentration (amount) such that it can be detected for a long period of time.
- excellent brain retention, high brain retention, or improved brain retention means, for example, that when a subject is administered to a test animal, the subject has 1 A high brain concentration (amount) of the subject for up to 10 days, preferably 2-10 days, 3-10 days, more preferably 4-10 days after administration, or a brain concentration (or The peak of (intracerebral volume) is 4 days or more after administration, preferably 5 days or more after administration, 6 days or more, 7 days or more, 8 days or more, 9 days or more, more preferably 10 days or more. And so on.
- Antibodies with high brain retention, high brain retention, or improved brain retention have higher antibody concentrations (antibody levels) in the brain than control antibodies, or have longer Any antibody can be used as long as it has characteristics that can be present.
- characteristics that are highly translocated into the brain and / or are accumulated in the brain characteristics that are translocated from the brain to outside the brain, excretion and / or degradability in the brain, and Antibodies having characteristics such as high translocation into the brain and / or accumulation in the brain as compared to translocation from the inside to the outside of the brain, excretion and / or degradability in the brain, and the like.
- the antibody or the antibody fragment of the present invention has a higher antibody concentration (or amount of antibody) in the brain after the same day from the administration, when the antibody or the antibody fragment is administered to an animal.
- the antibody fragment, or an antibody or the antibody fragment that can exist in the brain for a long period of time is administered to an animal.
- the change in the antibody concentration (or the amount of the antibody) in the brain may be any. For example, if the antibody concentration in the brain once peaks during the measurement period and then gradually decreases, After the antibody concentration reaches the peak, the case where the antibody concentration is maintained is maintained, or the case where the antibody concentration in the brain continues to increase after the administration of the antibody.
- Examples of the antibody or the antibody fragment of the present invention include an antibody having an antibody concentration or amount in the brain higher than that of a control antibody on the third or ninth day after administration to a mouse, and a third day after administration to a mouse.
- An antibody whose antibody concentration or amount in the brain is maintained or increases between day 9 and day 9 or an antibody whose presence in the brain can be clearly confirmed even after day 9 after administration to mice Etc., but not limited to these.
- control antibody any antibody may be used as long as it is an antibody of the same species or subclass as the test antibody.
- an anti-avermectin (AVM) antibody or the like can be used.
- examples of the inside of the brain include, but are not limited to, brain parenchyma, ventricle, and cerebrospinal fluid.
- CSPG5 binding molecule of the present invention it has nerve cell and / or astrocyte binding properties by specifically binding to CSPG5 on nerve cells and / or astrocytes, thereby improving brain retention.
- the antibody of the present invention includes, for example, an antibody that has binding to nerve cells and / or astrocytes by binding to CSPG5 on nerve cells and / or astrocytes and thereby has brain retention.
- examples of a method for administering an antibody to an animal include intravenous administration, intracerebroventricular administration, intraperitoneal administration, subcutaneous administration, intradermal administration, nasal administration, and intrathecal administration. The method is not limited.
- the brain tissue is collected several days after administering the antibody to an animal, homogenized, and the antibody concentration in the supernatant after centrifugation is measured.
- a method for calculating the amount of antibody per unit brain weight a method for detecting the presence of the antibody using a known immunological technique using the collected brain tissue, and administering a labeled antibody to an animal in vivo.
- the antibody of the present invention includes one antibody selected from the group consisting of the following (a) to (s).
- the antibody (e) is preferable from the viewpoints of brain retention and internalization ability of the antibody.
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 3, 4 and 5, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 8, 9 and 10, respectively;
- An antibody comprising the amino acid sequence described (B) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 13, 14 and 15, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 18, 19 and 20, respectively
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 23, 24 and 25, respectively, and the amino acid sequences of CDRs 1-3
- an antibody comprising the amino acid sequence described (D) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 33, 34 and 35, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 38, 39 and 40, respectively
- An antibody comprising the amino acid sequence described (E) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOs: 43, 44 and 45, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 48, 49 and 50, respectively
- An antibody comprising the amino acid sequence described (F) the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOS: 53, 54 and 55, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 58, 59 and 60, respectively
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences set forth in SEQ ID NOS: 113, 114 and 115, respectively, and the amino acid sequences of CDRs 1-3 of VL correspond to SEQ ID NOs: 118, 119 and 120, respectively.
- the amino acid sequences of CDRs 1-3 of VH include the amino acid sequences described in SEQ ID NOs: 133, 134 and 135, respectively, and the amino acid sequences of CDRs 1-3 of VL are represented by SEQ ID NOs: 138, 139 and 140, respectively.
- An antibody comprising the described amino acid sequence (p) an antibody that competes with at least one antibody of (a)-(o) for binding to CSPG5; (Q) an antibody that binds to an epitope including an epitope to which any one of the antibodies (a) to (o) binds, (R) an antibody that binds to the same epitope as the epitope to which any one of the antibodies (a) to (o) binds, (S) an antibody comprising an amino acid sequence having 85% or more homology with the amino acid sequence of any one of the above-mentioned antibodies (a) to (o).
- the antibody of the present invention may be at least 85%, preferably at least 90%, the amino acid sequence of CDR1-3 of VH and CDR1-3 of VL of any one of the above-mentioned antibodies (a) to (o).
- Antibodies having the amino acid sequences of CDRs 1-3 of VH and CDRs 1-3 of VL of antibodies exhibiting the above homology are included. More preferably, the homology of 90% or more includes 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% and 99% homology.
- one embodiment of the antibodies described in (a) to (o) above is a human anti-CSPG5 monoclonal antibody, CSPG5115 antibody, CSPG5120 antibody, CSPG5168 antibody, CSPG5201 antibody, CSPG5202 antibody, CSPG5205 antibody, Examples include CSPG5206, CSPG5207, CSPG5208, CSPG5214, CSPG5219, CSPG5222, CSPG5227, CSPG5230, and CSPG5234.
- the CSPG5202 antibody is preferred from the viewpoints of brain retention and internalization ability of the antibody.
- the antibody (p) is a second antibody that inhibits the binding between the first antibody and CSPG5 when any one of the antibodies (a) to (o) is used as the first antibody. Refers to an antibody.
- the antibody (q) refers to any one of the antibodies (a) to (o) described above as a first antibody and an epitope to which the first antibody binds as a first epitope. Refers to a second antibody that binds to a second epitope, including the first epitope.
- the antibody (r) of the present invention is defined as any one of the antibodies (a) to (o) described above as a first antibody and an epitope to which the first antibody binds as a first epitope. And a second antibody that binds to the first epitope.
- the antibody of the present invention include one antibody selected from the group consisting of the following (A) to (P).
- the antibody (E) is preferable from the viewpoints of brain retention and internalization ability of the antibody.
- C an antibody wherein the amino acid sequence of VH comprises the amino acid sequence of SEQ ID NO: 22 and the amino acid sequence of VL comprises the amino acid sequence of SEQ ID NO: 27,
- an antibody showing 85% or more, preferably 90% or more homology with the VH and VL amino acid sequences of any one of the antibodies described in (A) to (O) above, respectively. includes antibodies having VH and VL amino acid sequences. More preferably, the homology of 90% or more includes 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% and 99% homology.
- one embodiment of the antibodies described in (A) to (O) above is a CSPG5115 antibody, a CSPG5120 antibody, a CSPG5168 antibody, a CSPG5201 antibody, a CSPG5202 antibody, a CSPG5205 antibody, each of which is a human anti-CSPG5 monoclonal antibody.
- Examples include CSPG5206, CSPG5207, CSPG5208, CSPG5214, CSPG5219, CSPG5222, CSPG5227, CSPG5230, and CSPG5234.
- the CSPG5202 antibody is preferred from the viewpoints of brain retention and internalization ability of the antibody.
- the EU index refers to the position of an amino acid residue shown in the Sequence of Proteins of Immunological Interest, 5th edition (1991). The positions of amino acid residues shown below all indicate the positions of amino acid residues described in the EU index unless otherwise specified.
- An antibody molecule is also called an immunoglobulin (Ig), and its basic structure is a tetramer having two polypeptides called a heavy chain (Heavy chain; H chain) and two polypeptides called a light chain (Light chain; L chain). It is.
- Ig immunoglobulin
- the H chain is a variable region of the H chain (also denoted as VH) and a constant region of the H chain (also denoted as CH) from the N-terminal side
- the L chain is a variable region of the L chain (also denoted as VL) from the N-terminal side.
- L chain constant region also referred to as CL.
- CH is further composed of each domain of a CH1 domain, a hinge domain, a CH2 domain and a CH3 domain from the N-terminal side.
- the domain refers to a functional structural unit constituting each polypeptide of the antibody molecule.
- the CH2 domain and the CH3 domain are collectively referred to as an Fc (Fragment, crystallizable) region or simply Fc.
- CL is, C lambda chain and C kappa chain are known.
- IgA The subclasses of antibodies in which CH is the ⁇ , ⁇ , ⁇ , ⁇ , and ⁇ chains are called IgA, IgD, IgE, IgG, and IgM, respectively. Some subclasses of each antibody have isotypes depending on the animal. In humans, IgA has IgA1 and IgA2, and IgG has isotypes of IgG1, IgG2, IgG3 and IgG4.
- CH1 domain, hinge domain, CH2 domain, CH3 domain and Fc region in the present invention can be specified by the number of amino acid residues from the N-terminus by EU index.
- CH1 is the amino acid sequence of EU index 118 to 215
- hinge is the amino acid sequence of EU index 216 to 230
- CH2 is the amino acid sequence of EU index 231 to 340
- CH3 is the amino acid sequence of EU index 341 to 447.
- the amino acid sequence and the Fc region are specified as amino acid sequences of EU indexes 231 to 447, respectively.
- the antibodies of the present invention include any of polyclonal, monoclonal and oligoclonal antibodies.
- a polyclonal antibody refers to a population of antibody molecules secreted by antibody-producing cells of different clones.
- Monoclonal antibodies are antibodies that are secreted by a single clone of antibody-producing cells, recognize only one epitope (also called an antigenic determinant), and have a uniform amino acid sequence (primary sequence) that constitutes a monoclonal antibody.
- An oligoclonal antibody refers to a population of antibody molecules obtained by mixing a plurality of different monoclonal antibodies.
- the monoclonal antibody in the present invention includes an antibody produced by a hybridoma or a recombinant antibody produced by a transformant transformed with an expression vector containing an antibody gene.
- Epitope includes a single amino acid sequence, a three-dimensional structure consisting of an amino acid sequence, a post-translationally modified amino acid sequence, and a three-dimensional structure consisting of a post-translationally modified amino acid sequence, which are recognized and bound by a monoclonal antibody.
- amino acid sequence modified after translation examples include an O-linked sugar chain in which the sugar chain is bound to Tyr and Ser having an OH substituent, and an N-linked sugar chain in which the sugar chain is bound to Gln and Asn having an NH 2 substituent. , As well as tyrosine sulfated amino acid sequences in which a sulfuric acid molecule binds to Tyr having an OH substituent.
- the epitope of CSPG5 to which the antibody of the present invention binds is a deficient mutant in which a partial domain of CSPG5 is deleted, a mutant in which a partial domain of CSPG5 is substituted with a domain derived from another protein, a partial peptide of CSPG5 It can be determined by conducting an antibody binding experiment using fragments and the like. In addition, antibody binding experiments can also be performed using cells expressing the above-described defective or mutant.
- the epitope of CSPG5 to which the antibody of the present invention binds is also determined by adding the antibody of the present invention to a peptide fragment of CSPG5 digested with a protease and performing epitope mapping using a known mass spectrometry. can do.
- the antibody of the present invention includes a mouse antibody, a rat antibody, a hamster antibody, a rabbit antibody, a llama antibody, a camel antibody, an alpaca antibody, a chimeric antibody, a humanized antibody (also referred to as a “CDR-grafted antibody”) produced by a genetic recombination technique. ), And recombinant antibodies such as human antibodies.
- a chimeric antibody refers to an antibody in which VH and VL are different from CH and CL from animal species.
- Antibodies comprising antibodies VH and VL of non-human animals (non-human animals) and CH and CL of human antibodies are human-type chimeric antibodies, VH and VL of non-mouse antibodies, and CH and mouse antibodies.
- An antibody comprising CL is called a mouse-type chimeric antibody, and other chimeric antibodies are named in the same manner.
- any animal can be used, such as a mouse, a rat, a hamster, a rabbit, a llama, a camel, an alpaca, as long as it can produce a hybridoma or an antibody phage library.
- a hybridoma is a cell that produces a monoclonal antibody having a desired antigen specificity, obtained by cell fusion of a B cell obtained by immunizing a non-human animal with an antigen and a myeloma cell derived from a mouse or the like.
- Antibody phage library refers to a library prepared by cloning immunoglobulin variable region genes into phage and expressing antigen-binding molecules on the surface thereof.
- the phage to be used includes, but is not particularly limited to, M13 phage.
- the antigen-binding molecule displayed on the phage may be in any form, but is preferably an antibody fragment such as scFv, Fab, VHH or the like.
- the antibody phage library may be any one of an immune library, a naive library and a synthetic library.
- Immune library refers to an antibody phage library constructed based on antibody genes derived from lymphocytes of animals or patients immunized with the antigen.
- the naive library refers to an antibody phage library constructed based on antibody genes derived from lymphocytes of normal animals or healthy humans.
- a synthetic library refers to a library in which the CDRs of a V gene or a reconstructed functional V gene in genomic DNA have been replaced with oligonucleotides encoding random amino acid sequences of an appropriate length.
- a method for producing a chimeric antibody As a method for producing a chimeric antibody, a method for producing a human chimeric antibody is described below. Other chimeric antibodies can be prepared in the same manner.
- a human chimeric antibody is obtained by obtaining cDNAs encoding VH and VL from a hybridoma derived from a non-human animal cell that produces a monoclonal antibody, and converting the cDNA into an expression vector for animal cells having DNAs encoding CH and CL of a human antibody.
- a human-type chimeric antibody expression vector can be constructed by insertion, and can be expressed and produced by introducing it into animal cells.
- a human chimeric antibody expression vector is obtained by cloning genes encoding VH and VL from a non-human animal-derived antibody phage library and inserting them into animal cell expression vectors having DNAs encoding human antibody CH and CL, respectively. Can be expressed and produced by introducing it into animal cells.
- Humanized antibody refers to an antibody obtained by grafting the amino acid sequences of the CDRs of VH and VL of a non-human animal antibody to the corresponding CDRs of VH and VL of a human antibody.
- the regions other than the VH and VL CDRs are called FRs.
- the humanized antibody is a cDNA encoding a VH amino acid sequence consisting of the amino acid sequence of the CDR of VH of a non-human animal antibody and the amino acid sequence of FR of VH of any human antibody, and the amino acid sequence of the CDR of VL of a non-human animal antibody. And constructing a cDNA encoding the amino acid sequence of VL consisting of the amino acid sequence of FR of VL of any human antibody, inserting the cDNA into an expression vector for animal cells having DNAs encoding CH and CL of human antibody, and humanizing An antibody expression vector can be constructed and introduced into animal cells for expression and production.
- Human antibodies originally refer to antibodies naturally occurring in the human body, but also include antibodies obtained from human antibody phage libraries or human antibody-producing transgenic animals.
- the human antibody is obtained by immunizing a mouse (Tomizuka K. et al., Proc Natl Acad Sci U U S A. 97, 722-7, 2000.) carrying a human immunoglobulin gene with the desired antigen. Can be. Further, by selecting a human antibody having a desired binding activity using a phage display library obtained by amplifying an antibody gene from human-derived B cells, a human antibody can be obtained without performing immunization (Winter ⁇ G. et al., Annu Rev Immunol. 12: 433-55. 1994).
- the human antibody phage library is a phage library in which antibody fragments such as Fab, scFv, and VHH are expressed on the surface by inserting an antibody gene prepared from human (healthy or patient) lymphocytes into the phage gene. . From the library, phage expressing an antibody fragment having the desired antigen-binding activity can be recovered using the binding activity to the substrate on which the antigen is immobilized as an index. The antibody fragment can be further converted into a human antibody molecule consisting of two complete H chains and two complete L chains by genetic engineering techniques.
- a human antibody-producing transgenic animal refers to an animal in which the human antibody gene has been integrated into the chromosome of the host animal.
- a human antibody-producing transgenic animal can be produced by introducing a human antibody gene into mouse ES cells, transplanting the ES cells into an early embryo of another mouse, and then developing the embryo.
- Production of human antibodies from human antibody-producing transgenic animals is carried out by culturing human antibody-producing hybridomas obtained by a hybridoma production method performed in a normal non-human mammal to produce and accumulate human antibodies in the culture. And by purifying the antibody from the culture.
- the antibodies of the present invention include heavy chain antibodies composed only of heavy chains.
- Heavy chain antibodies refer to antibodies obtained from camelids such as llamas, camels, and alpacas, or recombinant antibodies produced based on the antibodies.
- an antibody fragment refers to an antibody fragment that has an antigen-binding activity.
- Fab, Fab ′, F (ab ′) 2 , scFv, diabody, dsFv, a peptide containing a plurality of CDRs, VHH and the like can be mentioned.
- the antibody fragment of the present invention includes an antibody fragment obtained by fusing the full length or a part of the constant region or Fc of the antibody to the antibody fragment, an antibody fragment containing the constant region or Fc, and the like. Any antibody fragment having a binding activity is included.
- Fab is a fragment obtained by treating an IgG antibody with the protease, papain (which is cleaved at the 224th amino acid residue of the H chain). About half of the N-terminal side of the H chain and the entire L chain are disulfide-bonded. (SS bond), which is an antibody fragment having a molecular weight of about 50,000 and having antigen-binding activity.
- SS bond is an antibody fragment having a molecular weight of about 50,000 and having antigen-binding activity.
- F (ab ') 2 is a fragment obtained by treating IgG with the protease pepsin (which is cleaved at amino acid residue 234 of the H chain). This is an antibody fragment having a molecular weight of about 100,000 and having an antigen-binding activity, which is slightly larger than that bound by the binding.
- Fab ' is an antibody fragment having a molecular weight of about 50,000 and having an antigen-binding activity in which the SS bond of the hinge region of F (ab') 2 is cleaved.
- the scFv uses an appropriate peptide linker (P) such as a linker peptide in which one VH and one VL are connected to an arbitrary number of linkers (G4S) consisting of four Gly and one Ser residue.
- P peptide linker
- G4S linkers
- Diabody is an antibody fragment in which scFvs having the same or different antigen-binding specificities form a dimer, and have a bivalent antigen-binding activity for the same antigen or a specific antigen-binding activity for a different antigen.
- dsFv is an antibody fragment having an antigen-binding activity in which a polypeptide in which one amino acid residue in each of VH and VL is substituted with a cysteine residue is bonded via an SS bond between the cysteine residues. It is.
- the peptide containing CDR comprises at least one region of CDR of VH or VL, and is an antibody fragment having antigen-binding activity.
- the CDRs can be linked to each other directly or via an appropriate peptide linker.
- the peptide containing the CDR of the present invention include a peptide containing 6 CDRs derived from the antibody of the present invention.
- the peptide containing the CDR is constructed by constructing a DNA encoding the VH and VL CDRs of the antibody of the present invention, inserting the DNA into a prokaryotic expression vector or a eukaryotic expression vector, and converting the expression vector into a prokaryotic expression vector. Alternatively, they can be expressed and produced by introducing them into eukaryotes. Further, the peptide containing the CDR can also be produced by a chemical synthesis method such as the Fmoc method or the tBoc method.
- VHH is a variable region of a heavy chain antibody, and is also referred to as nanobody.
- the antibody fragment of the present invention includes any antibody fragment containing any of the above-described antibody fragments or partial fragments thereof and having CSPG5 binding activity.
- an antibody having one antigen binding site or an antibody fragment thereof is referred to as a monovalent antibody.
- a monovalent antibody As the format of the monovalent antibody, the antigen binding site described in WO 2014/054804, WO 2011/090754, WO 2007/48037, WO 2012/116927, or the like can be used.
- One example is the format of the antibody or the antibody fragment.
- one molecule of an antibody or an antibody fragment that binds to three or more different antigens or epitopes is called a multispecific antibody.
- one molecule of an antibody or an antibody fragment that binds to two different antigens or epitopes is referred to as a bispecific antibody.
- bispecific antibody examples include the bispecific antibodies described below.
- Bispecific antibody in which an antibody fragment is fused to the N-terminus of the antibody.
- the bispecific antibody described in the above (1) is a bispecific antibody in which an antigen-binding site containing VH of heavy chain A binds to CSPG5 and an antigen-binding site containing VH of heavy chain B binds to an antigen present in the brain. Or vice versa.
- the bispecific antibody described in the above (2) includes, for example, both a bispecific antibody in which an antibody fragment is bound to one C-terminal of two heavy chains constituting an antibody, and two heavy chains constituting an antibody.
- Bispecific antibody in which an antibody fragment is bound to the C-terminus of the antibody Bispecific antibody in which an antibody fragment is bound to one of the two light chains constituting the antibody, and both light chains constituting the antibody
- a suitable linker may be present between the C-terminus of the antibody and the antibody fragment.
- the antibody fragment of the bispecific antibody described in (2) above is preferably scFv, Fab, VHH, etc., but is not particularly limited thereto.
- the bispecific antibody described in (2) above may be a bispecific antibody in which an N-terminal antigen-binding site binds to CSPG5 and a C-terminal antigen-binding site binds to an antigen present in the brain, or vice versa. .
- the bispecific antibody described in the above (3) refers to a bispecific antibody in which an antibody fragment is bound to the N-terminus of at least one of two heavy chains or light chains constituting the antibody. Further, a suitable linker may be present between the N-terminal of the heavy chain and / or light chain of the antibody and the antibody fragment.
- the antibody fragment of the bispecific antibody described in (3) above is preferably scFv, Fab, VHH or the like, but is not particularly limited thereto.
- the bispecific antibody described in the above (3) includes a bispecific antibody having a structure of VH 1 -CH1-VH 2 -CH1-hinge-CH2-CH3 from the N-terminus of the heavy chain, and the heavy chain structure described above.
- a, and the like bispecific antibody VH 1 and VH 2 forms a VL and antigen-binding sites, respectively, and the like.
- VL to VH 1 and VH 2 to form an antigen binding site can be the same amino acid sequence, it may be a different amino acid sequence.
- the multispecific antibody or the bispecific antibody may be any antibody as long as it is a multispecific antibody and a bispecific antibody that binds to CSPG5.
- a multispecific antibody or a bispecific antibody that binds to CSPG5 and an antigen present in the brain is preferable, and a multispecific antibody or a bispecific antibody containing an antigen binding site that binds to CSPG5 and an antigen that binds to an antigen present in the brain is preferred.
- Antibodies are more preferred.
- the antigens present in the brain include proteins, sugar chains, lipids and the like, and among them, proteins are preferable.
- proteins present in the brain include Prion, 5T4, AFP, ADAM10, ADAM12, ADAM17, AFP, AXL, BCAM, BSG, C5, C5R, CA9, CA72-4, CADM3, CCL11, CCL2, CCR1, CCR4, CCR5, CCR6, CD2, CD3E, CD4, CD5, CD6, CD8, CD11, CD18, CD19, CD20, CD22, CD24, CD25, CD29, CD30, CD32B, CD33, CD37, CD38, CD40, CD40LG, CD44, CD47, CD52, CD55SC1, CD56, CD66E, CD71, CD72, CD74, CD79a, CD79b, CD80, CD86, CD95, CD98, CD137, CD147, CD138, CD1 8, CD200, CD248, CD254, CD257, CDH2, CDH3, CEA, CEACAM1, CEACAM5, CEACAM6, CEACAM8, Claudin3, Claudin4, CSF-1, CSF2RA,
- sugar chains present in the brain include Lewis-x, Lewis-y, and CD15, but are not limited to these sugar chains.
- Examples of lipids present in the brain include, but are not limited to, GD1a, GD2, GD3, GM1, GM2, GM3, and Phosphatidylserine.
- the antibody or the antibody fragment of the present invention includes an antibody containing any amino acid modified after translation.
- post-translational modifications include deletion of a lysine residue at the C-terminus of the H chain [lysine clipping] and conversion of a glutamine residue at the N-terminus of the polypeptide to pyroglutamine (pyroGlu). [Beck et al, Analytical Chemistry, 85, 715-736 (2013)].
- the antibody or the antibody fragment of the present invention may have amino acid modification of the Fc region.
- the amino acid modification of the Fc region include an amino acid modification for stabilizing an antibody or controlling a half-life in blood.
- Specific examples of the amino acid modification of the Fc region include WO 2006/033386, WO 2006/075668, WO 2011/122011, and WO 2009/125825. .
- the antibody or the antibody fragment of the present invention also includes a fusion antibody or the fusion antibody fragment modified by binding a desired molecule to the antibody or the antibody fragment.
- the method for modifying the antibody is not particularly limited, and any method can be used as long as it can modify the desired amino acid residue and sugar chain.
- a chemical modification using a chemical reaction [Introduction to Antibody Engineering, Jinjinkan (1994); Kolb et al., Angew Chem Int Ed Engl. 40. 2004-21, 2001]; Modification by a genetic engineering technique for introducing a recombinant protein expression vector into an appropriate host cell and expressing the same is exemplified.
- the modification site when an antibody or an antibody fragment thereof is modified with another molecule by chemical modification, the modification site includes a constant region of the antibody or the antibody fragment, and particularly the Cys residue at the C-terminal or SS binding site. Groups are preferred.
- genetic engineering techniques it is also possible to introduce a chemically modifiable residue at an arbitrary position in the antibody or antibody fragment in advance.
- the modification site includes the N-terminus or C-terminus of the light or heavy chain of the antibody or antibody fragment.
- examples of the molecule that modifies the antibody or the antibody fragment include a hydrophilic polymer, an amphiphilic polymer, and a functional molecule.
- examples of the hydrophilic polymer or amphiphilic polymer include a molecule containing a polyoxyalkylene, a polyol, or a polysaccharide.
- polyoxyalkylene examples include linear or branched polyethylene glycol (PEG), polypropylene glycol, and polypropylene ethylene glycol.
- Examples of the molecule containing a polyol or a polysaccharide include a linear or branched polysaccharide obtained by polymerizing glucose such as amylose, dextran, pullulan, and glycogen. Further, the polysaccharide is not limited to the homopolysaccharide, and may be a heteropolysaccharide.
- the molecular weight of the molecule including the hydrophilic polymer or the amphiphilic polymer is not particularly limited, but is preferably 100 Da or more, for example, preferably 100 Da to 100 kDa.
- Functional molecules include, for example, antigen-binding molecules, fragments of antibody-binding molecules, drugs, bioactive peptides, bioactive proteins, nucleic acids, radiolabeled compounds, sugar chains, lipids, fluorescent compounds and the like.
- Molecules having dual specificity as a result of being modified with a functional molecule such as an antigen-binding molecule are bispecific antibodies.
- antigen-binding molecule examples include an antibody, a receptor, and a ligand.
- the fragment of the antigen-binding molecule may be any fragment of the antigen-binding molecule as long as it has an antigen-binding activity.
- an alkylating agent for example, an alkylating agent, a nitrosourea agent, an antimetabolite, an antiviral agent, an antibiotic, a plant alkaloid, a topoisomerase inhibitor, a tubulin polymerization inhibitor, a hormonal therapeutic agent, a hormone antagonist, an aromatase inhibitor,
- Anticancer agents such as P-glycoprotein inhibitors, platinum complex derivatives, M-phase inhibitors or kinase inhibitors [Clinical Oncology, Cancer and Chemotherapy (1996)], steroids, non-steroids, immunomodulators, immunosuppressants Or anti-inflammatory agents such as anti-histamines [Inflammation and anti-inflammatory therapy, Itoyaku Shuppan Co., Ltd. (1982)].
- a method for binding a drug and an antibody or an antibody fragment thereof in addition to the above-mentioned methods, a method for binding an amino group of a drug and an antibody via glutaraldehyde, or a method for binding an amino group of a drug and an antibody via a water-soluble carbodiimide and an antibody And a method of bonding a carboxyl group of the above.
- bioactive peptide or bioactive protein examples include interferon (IFN) - ⁇ , IFN- ⁇ , IFN- ⁇ , interleukin (IL) -2, IL-12, IL-15, IL-18, and IL-21.
- NK cells, macrophages, or neutrophils such as, IL-23, granulocyte colony stimulating factor (G-CSF), granulocyte / macrophage colony stimulating factor (GM-CSF), or macrophage colony stimulating factor (M-CSF)
- G-CSF granulocyte colony stimulating factor
- GM-CSF granulocyte / macrophage colony stimulating factor
- M-CSF macrophage colony stimulating factor
- cytokines or growth factors that activate immunocompetent cells proteolytic enzymes such as hydrolases, lyases and isomerases, enzymes such as acid sphingomyelinase, glucocerebrosidase, bacterial toxins such as lysine, diphtheria
- the nucleic acid may be any molecule as long as it is a molecule in which a nucleotide or a molecule having a function equivalent to the nucleotide is polymerized, and examples thereof include siRNA, microRNA, antisense RNA / DNA, and DNA aptamer.
- the radiolabeled compound may be a nuclide used for diagnostic or therapeutic purposes, and may be, for example, 3 H, 14 C, 32 P, 33 P, 35 S, 51 Cr, 57 CO, 18 F, 153 Gd. , 159 Gd, 64 Cu, 68 Ge, 166 Ho, 115 In, 113 In, 112 In, 111 In, 131 I, 125 I, 123 I, 121 I, 140 La, 177 Lu, 54 Mn, 99 Mo, 103 Pd, 142 Pr, 149 Pm, 186 Re, 188 Re, 211 At, 105 Rh, 97 Ru, 153 Sm, 47 Sc, 75 Se, 85 Sr, 99 Tc, 201 Ti, 113 Sn, 117 Sn, 133 Xe 169 Yb, 175 Yb, 90 Y and 65 Zn, etc., or including the nuclides described above Compounds.
- the radiolabeled compound can be directly bound to the antibody by the chloramine T method or the like. Further, a substance capable of chelating a radiolabeled compound may be bound to the antibody.
- the chelating agent include 1,4,7,10-tetraazacyclododecanetetraacetic acid (DOTA), 1- [2- (4-aminophenyl) ethyl] -1,4,7,10-tetraazacyclo Antibodies modified with a chelating agent, such as dodecanetetraacetic acid (PA-DOTA), 1,4,7,10-tetraazacyclotridecanetetraacetic acid (TRITA) and diethylenetriaminepentaacetic acid (DTPA), and a chelating agent
- PA-DOTA dodecanetetraacetic acid
- TRITA 1,4,7,10-tetraazacyclotridecanetetraacetic acid
- DTPA diethylenetriaminepentaacetic acid
- sugar chain examples include monosaccharides, disaccharides and oligosaccharides, and more specifically, for example, fucose, mannose, glucose, allose, artose, gulose, idose, galactose, talose, ribose, arabinose, Xylose, lyxose, erythose, erythrose, threose, cellobiose, maltose, isomaltose, lactose, lipoarabinomannan, Lewis X type trisaccharide, sialyl Lewis X type tetrasaccharide and the like.
- a natural product containing a sugar chain known as an immunoadjuvant may be used, and examples thereof include ⁇ (1 ⁇ 3) glucan (lentinan, schizophyllan) and ⁇ -galactosylceramide (KRN7000).
- lipids examples include esters of fatty acids with various alcohols and simple lipids (neutral lipids) that are analogs thereof.
- fatty acids eg, triacylglycerol
- waxes eg, fatty acid esters of higher alcohols
- sterol esters eg, cholesterol esters, fatty acid esters of vitamins, etc.
- Complex lipids having groups such as phospholipids (eg, glycerophospholipids and sphingolipids) and glycolipids (eg, glyceroglycolipids and glycosphingolipids), compounds formed by hydrolysis of simple lipids and complex lipids.
- derived lipids indicating fat-soluble ones, for example, fatty acids, higher alcohols, fat-soluble vitamins, steroids, hydrocarbons and the like can be mentioned.
- fluorescent compound examples include fluorescent dyes such as fluorescein series such as fluorescein isothiocyanate (FITC), rhodamine series such as rhodamine isothiocyanate (RITC), Cy3, Cy5, eosin series, alexafluoro series and NBD series, and acridinium.
- fluorescent dyes such as fluorescein series such as fluorescein isothiocyanate (FITC), rhodamine series such as rhodamine isothiocyanate (RITC), Cy3, Cy5, eosin series, alexafluoro series and NBD series, and acridinium.
- Luminescent substances such as esters or lophine
- fluorescent proteins such as green fluorescent protein (GFP).
- the antibody or the antibody fragment of the present invention can bind the above-mentioned hydrophilic polymer, amphiphilic polymer or functional molecule directly or via an appropriate linker.
- Linkers include, for example, esters, disulfides, hydrazones, dipeptides, and the like.
- the cDNA encoding the antibody is ligated to the cDNA encoding the protein, and the fusion antibody or fusion antibody is fused. Constructing a DNA encoding the antibody fragment, inserting the DNA into a prokaryotic or eukaryotic expression vector, and introducing the expression vector into a prokaryotic or eukaryotic organism to express the fusion antibody or fusion antibody fragment.
- a fusion antibody or a fusion antibody fragment can be prepared.
- composition of the present invention may be any composition as long as it contains the antibody or the antibody fragment of the present invention.
- Such a composition may contain, in addition to the antibody or the antibody fragment, additives such as a suitable carrier and a stabilizer.
- the composition of the present invention includes, for example, a composition for detection or measurement containing the antibody or the antibody fragment of the present invention.
- examples of the composition of the present invention include a pharmaceutical composition (therapeutic agent) containing the antibody of the present invention or the antibody fragment as an active ingredient, and the like, together with a pharmacologically acceptable carrier in a desired dosage form. Formulated.
- the composition for detection or measurement includes the antibody of the present invention or the antibody fragment thereof, and can detect or measure the antigen to which the antibody or the antibody fragment of the present invention specifically binds. Any composition may be used.
- the antigen to which the antibody or the antibody fragment of the present invention specifically binds include CSPG5, or antigens present in CSPG5 and brain.
- the antibody or the antibody fragment of the present invention has a property of binding to CSPG5 in the brain and staying in the brain. Therefore, by using a composition for detection or measurement containing the antibody or the antibody fragment, the antibody can be maintained in the brain or the antibody concentration in the brain can be increased, and CSPG5 or CSPG5 can be used for a long period of time. And antigens present in the brain and / or CSPG5, or antigens present in CSPG5 and the brain can also be detected or measured with high sensitivity.
- the composition for detection or measurement is a composition containing CSPG5 and a bispecific antibody that binds to an antigen present in the brain
- the CSPG5 to which the bispecific antibody binds and an antigen present in the brain are detected for a long period of time.
- measuring and / or detecting or measuring with high sensitivity CSPG5 and antigens present in the brain are measured for a long period of time.
- composition for detection or measurement is a fusion antibody that binds to CSPG5 labeled with a radiolabeled compound or a fluorescent dye or a composition containing the fusion antibody fragment
- CSPG5 can be detected or measured for a long period of time.
- / or CSPG5 can be detected or measured with high sensitivity.
- the pharmaceutical composition (therapeutic agent) containing the antibody or the antibody fragment of the present invention can be used as a therapeutic agent for any disease in which the antibody of the present invention or an antigen to which the antibody fragment specifically binds is expressed.
- a therapeutic agent for a brain disease is preferred.
- brain diseases for example, Alzheimer's disease, prodromal Alzheimer's disease, Huntington's disease, Parkinson's disease, brain tumor, multiple sclerosis, muscular dystrophy, amyotrophic lateral sclerosis, multiple system atrophy, progressive supranuclear palsy, Striatum substantia nigra, olive bridge cerebellar atrophy, spinobulbar muscular atrophy, spinocerebellar degeneration, cerebrovascular disorder, epilepsy, migraine, hyperactivity disorder, Creutzfeldt-Jakob disease, basal ganglia degeneration Disease, lysosomal disease, depression, and dystonia.
- the antibody or the antibody fragment of the present invention has a property of binding to CSPG5 in the brain when administered to an animal and remaining in the brain. Therefore, by using a therapeutic agent containing the antibody or the antibody fragment, the antibody or the antibody fragment in the brain can be maintained for a long period of time, and the antibody concentration in the brain can be increased. Can show therapeutic effect.
- the therapeutic agent when the therapeutic agent is a therapeutic agent containing the fusion antibody of the anti-CSPG5 antibody of the present invention, by delivering the fused molecule into the brain, the therapeutic effect of the molecule can be exerted.
- the therapeutic agent contains a fusion antibody obtained by fusing a drug or enzyme to an anti-CSPG5 antibody, the therapeutic effect can be exhibited by the drug or enzyme, and the therapeutic agent binds to CSPG5 and an antigen present in the brain.
- it when it is a therapeutic agent containing a bispecific antibody, it can exhibit a therapeutic effect on a brain disease associated with an antigen present in the brain to which the bispecific antibody binds.
- the therapeutic agent when the therapeutic agent is a fusion antibody or a fusion antibody fragment that binds to CSPG5 modified with a low-molecular-weight drug, the low-molecular-weight drug can exhibit a therapeutic effect on a target brain disease. At this time, it is preferable that the therapeutic effect is higher when the therapeutic agent of the present invention is used than when the low-molecular-weight drug is used alone.
- the therapeutic agent containing the antibody or the antibody fragment of the present invention may contain only the antibody or the antibody fragment as an active ingredient, but is usually combined with one or more pharmacologically acceptable carriers. Desirably, they are mixed together and provided as a pharmaceutical formulation prepared by any method known in the art of pharmacy.
- the administration route is preferably the one that is most effective in the treatment, for example, oral administration, or buccal, respiratory, intrarectal, subcutaneous, intradermal, intramuscular, intraventricular, intrathecal, intranasal Parenteral administration, such as intraperitoneal or intravenous administration, and particularly preferably intravenous or intraventricular administration.
- parenteral administration such as intraperitoneal or intravenous administration, and particularly preferably intravenous or intraventricular administration.
- the administration form include sprays, capsules, tablets, powders, granules, syrups, emulsions, suppositories, injections, ointments, tapes and the like.
- the dose or frequency of administration varies depending on the desired therapeutic effect, administration method, treatment period, age, body weight, etc., but is usually 10 ⁇ g / kg to 20 mg / kg per adult per day.
- the present invention provides a method for retaining an antibody in the brain using the antibody or the antibody fragment of the present invention, a method for improving the brain retention of the antibody, and a method for increasing the antibody concentration (or the amount of the antibody) in the brain. Is also included.
- the present invention provides a peptide that binds to CSPG5, a nucleic acid containing a nucleotide sequence encoding the peptide, a transformed cell containing a vector containing the nucleic acid, culturing the transformed cell, and collecting the peptide from the culture solution.
- the peptide of the present invention includes a fusion peptide in which the peptide is modified.
- CSPG5 Preparation of Antigen
- an expression vector containing cDNA encoding full-length or partial length of CSPG5 is introduced into Escherichia coli, yeast, insect cells, animal cells, or the like. Can be obtained at In addition, CSPG5 can also be obtained by purifying CSPG5 from various animal cell lines, animal cells, animal tissues, and the like that express CSPG5 in large amounts.
- these animal cell lines, animal cells, animal tissues, and the like can be used as they are as antigens.
- a synthetic peptide having a partial sequence of CSPG5 can be prepared by a chemical synthesis method such as the Fmoc method or the tBoc method and used as an antigen.
- a known tag such as FLAG or His may be added to the C-terminus or N-terminus of CSPG5 or a synthetic peptide having a partial sequence of CSPG5.
- CSPG5 used in the present invention is described in Molecular Cloning, A Laboratory Manual, Second Edition, Cold Spring Harbor Laboratory Press (1989) and Current Protocols In Molecular Molecular Biology, John Wiley & Sons (1987-1997) and the like.
- DNA encoding CSPG5 can be expressed in host cells and produced by the following method.
- a recombinant vector is prepared by inserting a full-length cDNA containing a portion encoding CSPG5 downstream of a promoter of an appropriate expression vector.
- a DNA fragment of an appropriate length containing a polypeptide-encoding portion prepared based on the full-length cDNA may be used.
- a transformant producing a polypeptide can be obtained by introducing the obtained recombinant vector into a host cell suitable for the expression vector.
- any vector can be used as long as it is capable of autonomous replication in a host cell to be used or integrated into a chromosome and contains a suitable promoter at a position where DNA encoding a polypeptide can be transcribed.
- a host cell any microorganism that belongs to the genus Escherichia, such as Escherichia coli, yeast, insect cells, or animal cells can be used as long as it can express the gene of interest.
- the expression vector When a prokaryote such as Escherichia coli is used as a host cell, the expression vector must be capable of autonomous replication in the prokaryote and, at the same time, contain a promoter, a ribosome binding sequence, DNA containing a portion encoding human CSPG5, and a transcription termination sequence. It is preferred that the vector contains the vector. In addition, a transcription termination sequence is not necessarily required in the expression vector, but it is preferable to arrange a transcription termination sequence immediately below a structural gene. Further, the recombinant vector may include a gene that controls a promoter.
- the expression vector it is preferable to use a plasmid in which the distance between the Shine-Dalgarno sequence (also called the SD sequence), which is a ribosome binding sequence, and the initiation codon is adjusted to an appropriate distance (for example, 6 to 18 bases).
- SD sequence also called the SD sequence
- the initiation codon is adjusted to an appropriate distance (for example, 6 to 18 bases).
- the base sequence of the DNA encoding CSPG5 can be substituted for a base so as to be an optimal codon for expression in a host, whereby the production rate of the target CSPG5 can be improved.
- Any expression vector can be used as long as it can exert its function in the host cell to be used.
- pBTrp2, pBTac1, pBTac2 all manufactured by Roche Diagnostics
- pKK233-2 Pharmacia
- pSE280 Invitrogen
- pGEMEX-1 Promega
- pQE-8 Qiagen
- pKYP10 JP-A-58-110600
- pKYP200 [Agricultural Biologic Chemistry, 48, 669 (1984)]
- pLSA1 Agric. Biol. Chem., 53, 277 (1989)]
- pGEL1 Proc. Natl. Acad. Sci.
- pBluescript II SK (- ) Manufactured by Stratagene
- pTrs30 Escherichia coli JM109 / pTrS30 (FERM @ BP-5407)
- PTrs32 prepared from Escherichia coli JM109 / pTrS32 (FERM @ BP-5408)
- pGHA2 prepared from Escherichia coli IGHA2 (FERM @ BP-400), Japanese Unexamined Patent Publication No. 60-221091]
- pGKA2 Escherichia coli IGKA2 (FERM) BP-6798
- the promoter may be any promoter as long as it can exert its function in the host cell used.
- a promoter derived from Escherichia coli or a phage such as a trp promoter (Ptrp), a lac promoter, a PL promoter, a PR promoter or a T7 promoter, may be mentioned.
- trp trp promoter
- lac lac promoter
- PL promoter PL promoter
- PR promoter a PR promoter
- T7 promoter a promoterp promoter
- artificially designed and modified promoters such as a tandem promoter, a tac promoter, a lacT7 promoter, and a let @ I promoter in which two Ptrps are connected in series may be mentioned.
- Examples of host cells include E. coli XL1-Blue, E. coli XL2-Blue, E. coli DH1, E. coli MC1000, E. coli KY3276, E. coli W1485, E. coli JM109, E. coli HB101, E. coli No. 49, Escherichia coli W3110, Escherichia coli NY49 or Escherichia coli DH5 ⁇ .
- any method for introducing a recombinant vector into a host cell any method can be used as long as DNA is introduced into a host cell to be used.
- a method using calcium ions Proc. Natl. Acad. Sci. USA , ⁇ 69, ⁇ 2110 ⁇ (1972), Gene, ⁇ 17, ⁇ 107 ⁇ (1982), Molecular & General Genetics, ⁇ 168, ⁇ 111 ⁇ (1979)].
- any expression vector can be used as long as it can exert a function in the animal cell.
- pcDNAI, pCDM8 manufactured by Funakoshi
- pAGE107 Japanese Unexamined Patent Publication No. -22979; Cytotechnology, ⁇ 3, ⁇ 133 ⁇ (1990)
- pAS3-3 Japanese Patent Application Laid-Open No.
- pCDM8 [Nature, ⁇ 329, ⁇ 840 ⁇ (1987)], pcDNAI / Amp (manufactured by Invitrogen) , PcDNA3.1 (Invitrogen), pREP4 (Invitrogen), pAGE103 [J. Biochemistry, 101, 1307 ⁇ (1987), pAGE210, pME18SFL3, pKANTEX93 (WO 97/10354), N5KG1 val (US patent). No. 6,001,358), IMPEP4 (Bi Made gen-IDEC, Inc.), pCI (Promega Corp.) and transposon vector (WO 2010/143698), and the like.
- Any promoter can be used as long as it can exert its function in animal cells, and examples thereof include a cytomegalovirus (CMV) immediate @ early (IE) gene promoter, an SV40 early promoter, and a retrovirus promoter. , Metallothionein promoter, heat shock promoter, SR ⁇ promoter or Moloney murine leukemia virus promoter or enhancer. Further, the enhancer of the IE gene of human CMV may be used together with the promoter.
- human leukemia cells Namalwa cells, monkey cells COS cells, Chinese hamster ovary cells CHO cells [Journal of Experimental Medicine, 108, 945 (1958); Proc. Natl. Acad. Sci. USA, 60] 1275 (1968); Genetics, 55, 513 (1968); Chromosoma, 41, 129 (1973); Methods in Cell Science, 18, 115 (1996); Radiation Research, 148, 260 (1997); Proc. Natl. Acad. Sci. USA, 77, 4216 (1980); Proc. Natl. Acad. Sci., 60, 1275 (1968); Cell, 6, 121 (1975); Molecular Cell Genetics, Appendix I, II (pp.
- CHO cells deficient in the dihydrofolate reductase gene CHO / DG44 cells [Proc. Natl. Acad. Sci. USA, 77, 4216 (1980)]; CHO-K1 (ATCC @ CCL-61). ), DUkXB11 (ATCC @ CCL-9096), Pro-5 (ATCC @ CCL-1781) ), CHO-S (Life @ Technologies, Cat # 11619), Pro-3, rat myeloma cells YB2 / 3HL. P2. G11.16Ag.
- mouse myeloma cell NS0 mouse myeloma cell SP2 / 0-Ag14
- Syrian hamster cell BHK or HBT5637 Japanese Patent Application Laid-Open No. 63-000299.
- any method for introducing DNA into animal cells can be used.
- an electroporation method [Cytotechnology, ⁇ 3, ⁇ 133 ⁇ (1990)]
- a calcium phosphate method Japanese Patent Application Laid-Open No. 2-227075
- the lipofection method [Proc. Natl. Acad. Sci. USA, 84, 7413 (1987)].
- a transformant derived from a microorganism or animal cell having an expression vector incorporating the DNA encoding CSPG5 obtained as described above is cultured in a medium, and the CSPG5 is produced and accumulated in a culture solution. By collecting from the culture solution, CSPG5 can be produced.
- the method for culturing the transformant in a medium can be performed according to a usual method used for culturing a host.
- CSPG5 When expressed in eukaryotic cells, CSPG5 to which a sugar or a sugar chain is added can be obtained.
- an inducer may be added to the medium as necessary.
- an inducer may be added to the culture medium such as indole acrylic acid.
- Examples of a medium for culturing a transformant obtained by using an animal cell as a host include, for example, a commonly used RPMI1640 medium [The Journal of the American Medical Association, 199, 519 (1967)], Eagle's MEM medium [Science , 122, 501 (1952)], Dulbecco's modified MEM medium [Virology, 8, 396 (1959)], 199 medium [Proc. Soc. Exp. Biol. Med., 73, 1 (1950)], or Iscove's Modified. Examples include Dulbecco's Medium (IMDM) medium or a medium obtained by adding fetal bovine serum (FBS) or the like to such a medium. The cultivation is usually performed for 1 to 7 days under conditions such as pH 6 to 8, 30 to 40 ° C., and 5% CO 2 . If necessary, an antibiotic such as kanamycin or penicillin may be added to the medium during the culture.
- RPMI1640 medium The Journal of the American Medical Association, 199, 519 (19
- Examples of the method of expressing the gene encoding CSPG5 include, in addition to direct expression, methods such as secretory production or fusion protein expression [Molecular Cloning, A Laboratory Laboratory Manual, Second Edition, Cold Spring Harbor Laboratory Press (1989)]. .
- Examples of the method of producing CSPG5 include a method of producing it in a host cell, a method of secreting it out of the host cell, and a method of producing it on the host cell outer membrane. By changing, an appropriate method can be selected.
- CSPG5 When CSPG5 is produced in the host cell or on the host cell outer membrane, the method of Paulson et al. [J. Biol. Chem., 264, 17619 ⁇ ⁇ (1989)] and the method of Lowe et al. [Proc. Natl. Acad. Sci. , USA, 86, 8227 (1989), Genes Develop., 4, 1288 (1990)], Japanese Patent Application Laid-Open No. 05-336963 or International Publication No. WO 94/23021. Can be actively secreted out of the host cell. Further, the production of CSPG5 can also be increased by using a gene amplification system using a dihydrofolate reductase gene or the like (Japanese Patent Application Laid-Open No. 2-227075).
- CSPG5 can be isolated and purified, for example, as follows.
- CSPG5 When CSPG5 is expressed in a dissolved state in the cells, the cells are collected by centrifugation after completion of the culture, suspended in an aqueous buffer, and then sonicated using a sonicator, French press, Mantongaulin homogenizer or Dynomill. The cells are disrupted to obtain a cell-free extract.
- a normal protein isolation and purification method that is, a solvent extraction method, a salting out method using ammonium sulfate, a desalting method, a precipitation method using an organic solvent, diethylamino Anion exchange chromatography using a resin such as ethyl (DEAE) -Sepharose and DIAION @ HPA-75 (manufactured by Mitsubishi Chemical Corporation), and cation exchange chromatography using a resin such as S-Sepharose @ FF (manufactured by Pharmacia) Method, hydrophobic chromatography using resins such as butyl sepharose and phenyl sepharose, gel filtration using molecular sieve, affinity chromatography, chromatofocusing, or electrophoresis such as isoelectric focusing Can be used alone or in combination It is possible.
- a resin such as ethyl (DEAE) -Sepharose and DIAION @ HPA-75 (manufactured by Mitsubishi Chemical Corporation)
- CSPG5 When CSPG5 is expressed by forming an insoluble substance in the cell, the cell is recovered and crushed in the same manner as described above, and centrifuged to collect the insoluble substance of CSPG5 as a precipitate fraction. The recovered insoluble form of CSPG5 is solubilized with a protein denaturant. After diluting or dialyzing the lysate to return the CSPG5 to a normal three-dimensional structure, a purified preparation of the polypeptide can be obtained by the same isolation and purification method as described above.
- the derivative such as CSPG5 or its modified sugar can be recovered from the culture supernatant.
- a soluble fraction is obtained by treating the culture by a technique such as centrifugation as described above, and a purified sample can be obtained from the soluble fraction by using the same isolation and purification method as described above. it can.
- CSPG5 used in the present invention can also be produced by a chemical synthesis method such as the Fmoc method or the tBoc method. Chemical synthesis using a peptide synthesizer manufactured by Advanced Chemtech, Perkin-Elmer, Pharmacia, Protein Technology Instrument, Synthcel-Vega, Perceptive or Shimadzu You can also.
- Immunization is performed by administering the antigen subcutaneously, intravenously or intraperitoneally to the animal, for example, with Freund's complete adjuvant, or with an appropriate adjuvant such as aluminum hydroxide gel and B. pertussis vaccine.
- a conjugate is prepared with a carrier protein such as bovine serum albumin (BSA) or Keyhole @ Limpet @ hemocyanin (KLH) and used as an immunogen.
- BSA bovine serum albumin
- KLH Keyhole @ Limpet @ hemocyanin
- the antigen is administered 5 to 10 times every 1 to 2 weeks after the first administration. Blood is collected from the fundus venous plexus 3 to 7 days after each administration, and the antibody titer of the serum is measured using an enzyme immunoassay [Antibodies II-A Laboratory Manual, Cold Spring Harbor Laboratory (1988)] or the like. An animal whose serum shows a sufficient antibody titer against the antigen used for immunization is used as a source of the antibody-producing cells for fusion.
- a tissue containing antibody-producing cells such as spleen is excised from the immunized animal, and the antibody-producing cells are collected.
- spleen cells When spleen cells are used, the spleen is shredded and loosened, then centrifuged, and the erythrocytes are removed to obtain antibody-producing cells for fusion.
- immunized animals can be immunized in the same manner to obtain antibody-producing cells.
- Appropriate conditions can be selected for the immunization interval and the period from the last immunization to the removal of the tissue according to the animal species of the immunized animal.
- myeloma cells cell lines obtained from mice were used.
- myeloma cell line P3-X63Ag8-U1 P3- U1 [Current Topics in Microbiology and Immunology, 18, 1 (1978)]
- P3-NS1 / 1-Ag41 P3-NS1 [European J. Immunology, 6, 511 (1976)]
- SP2 / 0-Ag14 SP-2 [Nature, 276, 269 (1978)]
- P3-X63-Ag8653 653
- J. Immunology, 123, 1548 (1979)] or P3-X63-Ag8 (X63) [Nature, 256, 495] (1975)].
- the myeloma cells were passaged in normal medium [RPMI 1640 medium supplemented with glutamine, 2-mercaptoethanol, gentamicin, FBS, and 8-azaguanine], and passaged to normal medium 3 to 4 days before cell fusion. On the day of the fusion, secure a cell count of 2 ⁇ 10 7 or more.
- MEM Minimu Essential Medium
- PBS phosphate buffered saline
- a mixed solution of polyethylene glycol-1000 (PEG-1000), MEM medium and dimethyl sulfoxide is added at 37 ° C. with stirring. Further, 1 to 2 mL of MEM medium is added several times every 1 to 2 minutes, and then MEM medium is added so that the total volume becomes 50 mL.
- HAT medium normal medium supplemented with hypoxanthine, thymidine and aminopterin. The suspension is cultured in a 5% CO 2 incubator at 37 ° C. for 7-14 days.
- a part of the culture supernatant is extracted, and a cell group that reacts with CSPG5 but does not react with an antigen other than CSPG5 is selected by a hybridoma selection method such as a binding assay described below.
- cloning is performed by the limiting dilution method, and those with stable and strong antibody titers are selected as monoclonal antibody-producing hybridomas.
- the supernatant is removed by centrifugation, suspended in Hybridoma SFM medium, and cultured for 3 to 7 days.
- the obtained cell suspension is centrifuged, and the obtained supernatant is purified by a protein A-column or a protein G-column, and an IgG fraction is collected to obtain a purified monoclonal antibody.
- 5% Daigo GF21 can be added to the Hybridoma @ SFM medium.
- the subclass of the antibody is determined by enzyme immunoassay using a subcluster typing kit.
- the quantification of the protein amount is calculated by the Lowry method or from the absorbance at 280 nm.
- the antibody is selected by measuring the binding of the antibody to cells expressing CSPG5 using flow cytometry, as described below.
- the CSPG5 expressing cell may be any cell as long as CSPG5 is expressed on the cell surface, and examples thereof include animal cells, animal cell lines, and the CSPG5 forced expression cell line obtained in (1).
- a test substance such as serum, hybridoma culture supernatant or purified antibody is dispensed as the first antibody and reacted.
- the cells are thoroughly washed with PBS containing 1 to 10% BSA (hereinafter referred to as BSA-PBS) or the like, and then an anti-immunoglobulin antibody labeled with a fluorescent reagent or the like is dispensed as a second antibody to perform the reaction. Let it. After thoroughly washing with BSA-PBS or the like, an antibody that specifically reacts with CSPG5 expressing cells is selected by measuring the amount of fluorescence of the labeled antibody using a flow cytometer.
- ⁇ Antibodies can also be selected by measuring the binding of the monoclonal antibody to CSPG5-expressing cells or CSPG5 protein using ELISA or surface plasmon resonance described below.
- the CSPG5 protein may be a protein comprising a partial domain of CSPG5 or a protein to which a tag such as GST has been added.
- ELISA In ELISA, after dispensing CSPG5-expressing cells or CSPG5 protein into a plate such as a 96-well plate, blocking is performed with BSA-PBS, and a test substance such as serum, a hybridoma culture supernatant or a purified antibody is dispensed as a first antibody. And react. Next, after thoroughly washing with PBS or the like, an anti-immunoglobulin antibody labeled with a fluorescent reagent or the like is dispensed as the second antibody and reacted.
- a test substance such as serum, a hybridoma culture supernatant or a purified antibody is dispensed as a first antibody. And react.
- an anti-immunoglobulin antibody labeled with a fluorescent reagent or the like is dispensed as the second antibody and reacted.
- Surface plasmon resonance can measure the affinity of an antibody that binds to CSPG5 by immobilizing the antibody on an appropriate sensor chip and using the CSPG5 protein as an analyte using a known protocol.
- An antibody having a desired affinity for CSPG5 protein can be selected from the obtained antibody affinity.
- CSPG5 protein can be immobilized on a sensor chip, and the affinity of an antibody that binds to CSPG5 can be measured using the antibody as an analyte.
- An antibody that binds to CSPG5 in competition with the antibody of the present invention can be obtained by adding a test antibody to the above-described measurement system using flow cytometry or ELISA and allowing it to react. That is, by screening for an antibody that inhibits the binding of the antibody of the present invention and CSPG5 when a test antibody is added, an antibody that competes with the antibody of the present invention for binding to the amino acid sequence of CSPG5 or its three-dimensional structure is determined. Can be obtained.
- an antibody that binds to an epitope including an epitope to which the antibody of the present invention binds can be obtained by identifying the epitope of the antibody obtained by the above-described screening method by a known method, and then synthesizing a peptide containing the identified epitope or a steric epitope. It can be obtained by preparing and immunizing a synthetic peptide or the like that mimics the structure.
- an epitope that binds to the same epitope as the epitope to which the antibody of the present invention binds identifies the epitope of the antibody obtained by the above-described screening method, and identifies a partial synthetic peptide of the identified epitope or the three-dimensional structure of the epitope. It can be obtained by preparing a synthetic peptide or the like mimicking the above and immunizing the same.
- the immunization library collects lymphocytes from animals or patients immunized by the same method as in the above (1), and the naive library collects lymphocytes from normal animals or healthy humans, extracts RNA, and performs reverse transcription reaction. To synthesize cDNA.
- the antibody gene fragment amplified by PCR using this cDNA as a template is inserted into a phagemid vector, and Escherichia coli is transformed with the phagemid vector.
- Escherichia coli is transformed with the phagemid vector.
- an antibody phage library in which an antibody gene has been libraryd can be obtained.
- the synthetic library is a phagemid vector in which the V gene in the genomic DNA or the CDR of the reconstructed functional V gene is replaced with an oligonucleotide encoding a random amino acid sequence of an appropriate length, and the V gene is inserted.
- an antibody phage library can be obtained.
- Lymphocyte-derived cDNA and antibody phage library may be commercially available.
- Phagemid vectors are pCANTABNT5E (Amersham Pharmacia), pUC118 / pUC119 vector (TaKaRa), pBlueScript II Phagemid Vector (Agilent Techn. 3), 205-215, 2015) can be used.
- Helper phage can be M13KO7 helper phage (Invitrogen), VCSM13 Interference Resistant Helper Phage (Agilent Technologies), R408 Interference Resistant HelgeAge, etc.
- a phage vector can also be used for phage display.
- Peptide phage libraries eg, New England Biolabs
- g3p of filamentous phage as a display molecule
- methods using g7p, g8p, and g9p as display molecules eg., New England Biolabs
- a phage display using T7 phage can be used.
- Examples of a display system for T7 phage include a T7Select vector (Novagen).
- Antibody phage clones can be selected from the antibody phage library prepared in (7-1) using the ELISA method described below.
- the antibody phage library prepared in (7-1) above is added to each well of the tube and reacted.
- the wells are washed, and a fluorescently labeled anti-phage antibody is added and reacted.
- the wells are washed again and a coloring solution is added. Thereafter, the color reaction is stopped with a reaction stop solution, and the absorbance in each well is measured with a microplate reader.
- an antibody phage clone that binds to CSPG5 is selected.
- Recombinant Antibody As an example of preparing a recombinant antibody, a method for preparing a human chimeric antibody and a humanized antibody is described below. Genetically modified mouse antibodies, rat antibodies, rabbit antibodies, hamster antibodies, camel antibodies, llama antibodies, alpaca antibodies, human antibodies, various chimeric antibodies, heavy chain antibodies, and the like can be prepared in the same manner.
- a vector for expressing a recombinant antibody is an expression vector for animal cells into which DNAs encoding human antibody CH and CL are incorporated. It can be constructed by cloning the DNAs encoding the antibody CH and CL, respectively.
- CH and CL of any human antibody can be used.
- CH of the ⁇ 1 subclass and CL of the ⁇ class of a human antibody are used.
- CDNA is used as the DNA encoding CH and CL of the human antibody, but chromosomal DNA consisting of exons and introns can also be used.
- Any expression vector for animal cells can be used as long as it can incorporate and express the gene encoding the C region of the human antibody.
- pAGE107 [Cytotechnol., ⁇ 3, ⁇ 133 ⁇ (1990)]
- pAGE103 [J. Biochemical., ⁇ 101, ⁇ 1307 ⁇ (1987)]
- pHSG274 [Gene, ⁇ 27, ⁇ 223 ⁇ (1984)]
- pKCR Proc. Sci. ⁇ USA, ⁇ 78, ⁇ 1527 ⁇ (1981)]
- pSG1bd2-4 [Cytotechnol., ⁇ 4, ⁇ 173 ⁇ (1990)]
- pSE1UK1Sed1-3 [Cytotechnol., ⁇ 13, ⁇ 79 ⁇ (1993)].
- the promoter and enhancer include the SV40 early promoter [J. Biochem., 101, 1307] (1987), Moloney murine leukemia virus LTR [Biochem. Biophys. Res. Commun., 149, 960]. (1987)] or an immunoglobulin heavy chain promoter [Cell, ⁇ 41, ⁇ 479 ⁇ (1985)], and an enhancer [Cell, ⁇ 33, ⁇ 717 ⁇ (1983)].
- the recombinant antibody expression vector balances the ease of construction of the recombinant antibody expression vector, the ease of introduction into animal cells, and the balance between the expression levels of antibody H chains and L chains in animal cells.
- a type (tandem type) expression vector for a recombinant antibody in which the antibody H chain and L chain are present on the same vector [J. Immunol. Methods, 167, 271 (1994)]
- pKANTEX93 WO 97/10354
- pEE18 Hybridoma, ⁇ 17, ⁇ 559 ⁇ (1998)] and the like are used.
- V region variable region of antibody derived from non-human animal and analysis of amino acid sequence Acquisition of cDNA encoding VH and VL of non-human antibody and analysis of amino acid sequence are as follows. You can do it.
- a recombinant phage or a recombinant plasmid having a cDNA encoding VH or VL is isolated from the library by using a DNA encoding the C region or V region of the non-human antibody as a probe.
- the entire nucleotide sequence of VH or VL of the target non-human antibody on the recombinant phage or recombinant plasmid is determined, and the entire amino acid sequence of VH or VL is deduced from the nucleotide sequence.
- mice For animals other than humans that produce non-human antibody-producing hybridoma cells, mice, rats, hamsters, rabbits, llamas, camels, or alpacas, etc. are used, provided that it is possible to produce hybridoma cells. Animals can also be used.
- RNA easy kit manufactured by Qiagen
- oligo (dT) -immobilized cellulose column method [Molecular Cloning, A Laboratory, Manual, Second Edition, Cold Spring, Harbor Laboratory Press] (1989), or Oligo-dT30 ⁇ Super> mRNA Purification ( A kit such as (registered trademark) Kit (manufactured by Takara Bio Inc.) is used.
- mRNA can be prepared from hybridoma cells using a kit such as Fast Track mRNA Isolation (registered trademark) Kit (manufactured by Invitrogen) or QuickPrep mRNA mRNA Purification (registered trademark) Kit (manufactured by Pharmacia).
- any vector can be used as a vector for incorporating a cDNA synthesized using mRNA extracted from hybridoma cells as a template, as long as the vector can incorporate the cDNA.
- ZAP Express [Strategies, 5, 58 58 (1992)], pBluescript II II SK (+) [Nucleic Acids Research, 17, 17, 9494 (1989)], ⁇ ZAPII (Stratagene), ⁇ gt10, ⁇ gt11 [DNA] Clon [DNA] Pract Approach, I, 49 (1985)], Lambda BlueMid (manufactured by Clonetech), EExCell, pT7T3-18U (manufactured by Pharmacia), pCD2 [Mol. Cell. Biol., 3, 280 (1983)] or pUC18 [Gene] , ⁇ 33, 103 ⁇ (1985)].
- ⁇ Escherichia coli to which a cDNA library constructed by a phage or a plasmid vector is to be introduced can be used as long as the cDNA library can be introduced, expressed and maintained.
- a cDNA clone encoding a non-human antibody VH or VL from a cDNA library can be performed by colony hybridization using an isotope or fluorescently labeled probe, or plaque hybridization [Molecular Cloning, A Laboratory Manual, Second Edition, Cold Spring Harbor Laboratory Press (1989)].
- primers were prepared and, using a cDNA or a cDNA library synthesized from mRNA as a template, a Polymerase Chain Reaction (PCR) method [Molecular Cloning, A Laboratory Laboratory, Second Edition, Cold Spring Harbor Laboratory Press (1989), Current Protocols] Biology, Supplement 1, John Wiley & Sons (1987-1997)] can also prepare a cDNA encoding VH or VL.
- PCR Polymerase Chain Reaction
- the cDNA is cloned into a plasmid such as pBluescript @ SK (-) (manufactured by Stratagene), and the nucleotide sequence of the cDNA is determined by a commonly used nucleotide sequence analysis method or the like.
- the nucleotide sequence analysis method includes, for example, a reaction such as the dideoxy method [Proc. Natl. Acad. Sci. USA, 74, 5463 (1977)], followed by ABI PRISM 3700 (manufactured by PE Biosystems) or A.I. L. F. An automatic base sequence analyzer such as a DNA sequencer (Pharmacia) is used.
- the entire amino acid sequences of VH and VL are deduced from the determined nucleotide sequence, and the entire amino acid sequences of VH and VL of a known antibody [Sequences of the Proteins of the Immunological Interest, US Dept. ⁇ Health ⁇ and ⁇ Human ⁇ Services ⁇ (1991)] to confirm whether the obtained cDNA encodes the complete amino acid sequence of VH and VL of the antibody including the secretory signal sequence.
- amino acid sequences of the CDRs of VH and VL are also found by comparing with the amino acid sequences of VH and VL of known antibodies [Sequences of Proteins of Immunology Interest, US Dept. Health and Human Services (1991)]. Can be.
- the base sequence of the connecting portion encodes an appropriate amino acid, and VH and VL cDNAs designed to have appropriate restriction enzyme recognition sequences are prepared.
- the prepared VH and VL cDNAs are respectively inserted into the recombinant antibody expression vector obtained in (1) such that they are expressed in an appropriate form upstream of the respective genes encoding CH or CL of the human antibody. Cloning is performed to construct a human chimeric antibody expression vector.
- the cDNA encoding the non-human antibody VH or VL is amplified by PCR using synthetic DNA having a recognition sequence for an appropriate restriction enzyme at both ends, and the recombinant antibody expression vector obtained in (1) is obtained. Can also be cloned.
- VH or VL FR amino acid sequence of the human antibody to transplant the VH or VL CDR amino acid sequence of the non-human antibody, respectively.
- Any amino acid sequence derived from a human antibody can be used for the selected amino acid sequence of FR.
- amino acid sequence of human antibody FR registered in a database such as Protein ⁇ Data ⁇ Bank, or the common amino acid sequence of each subgroup of human antibody FR [Sequences ⁇ of ⁇ Proteins ⁇ of ⁇ Immunological ⁇ Interest, ⁇ US ⁇ Dept. ⁇ Health ⁇ and ⁇ Human ⁇ Services ⁇ 1991)].
- an amino acid sequence of FR having the highest possible homology (at least 60% or more) to the amino acid sequence of VH or VL of the original antibody is selected.
- the amino acid sequence of the CDR of the original antibody is transplanted into the amino acid sequence of the FR of the VH or VL of the selected human antibody, respectively, and the amino acid sequence of the VH or VL of the humanized antibody is designed.
- the designed amino acid sequence is converted into a DNA sequence in consideration of the frequency of codon usage found in the nucleotide sequence of the antibody gene [Sequences of Proteins of Immunology Interest, US Dept. Health and Human Services (1991)], and the humanized antibody is converted.
- a DNA sequence encoding the amino acid sequence of VH or VL is designed, respectively.
- cDNA encoding the VH or VL of the humanized antibody can be obtained by the gene set obtained in (1). It can be easily cloned into a recombinant antibody expression vector.
- the amplified product was cloned into a plasmid such as pBluescript @ SK (-) (manufactured by Stratagene), the nucleotide sequence was determined by the same method as described in (2), and the desired humanized antibody was obtained.
- a plasmid having a DNA sequence encoding the amino acid sequence of VH or VL is obtained.
- one obtained by synthesizing the full length VH and the full length VL as one long chain DNA based on the designed DNA sequence can be used in place of the PCR amplification product.
- cDNA encoding the VH or VL of the humanized antibody can be easily inserted into the recombinant antibody expression vector obtained in (1). Can be cloned.
- amino acid residues directly involved in binding to the antigen amino acid residues that interact with amino acid residues of the CDR
- Reduced antigen binding by maintaining the steric structure, identifying amino acid residues that are indirectly involved in antigen binding, and replacing those amino acid residues with those of the original non-human antibody Activity can be increased.
- X-ray crystallography [J. Mol. Biol., 112, 535 (1977)] or computer modeling [Protein Engineering, 7, FR1501 (1994)] to identify amino acid residues of FRs involved in antigen-binding activity
- a humanized antibody having the required antigen-binding activity can be obtained by repeatedly preparing several kinds of variants for each antibody, examining the correlation with each antigen-binding activity, and conducting trial and error.
- VH and VL FR amino acid residues of the human antibody can be modified by performing the PCR reaction described in (4) using the synthetic DNA for modification.
- the nucleotide sequence of the amplified product after the PCR reaction is determined by the method described in (2) to confirm that the desired modification has been performed.
- each of the recombinant antibody expression vectors obtained in (1) is cloned so as to express them in an appropriate form upstream of each gene encoding CH or CL of the human antibody.
- Transient expression of the recombinant antibody is performed using the recombinant antibody expression vector obtained in (3) and (6) or an expression vector obtained by modifying the same.
- the antigen-binding activity of the various human chimeric antibodies and humanized antibodies thus prepared can be efficiently evaluated.
- any host cell that can express the recombinant antibody can be used.
- COS-7 cells American Type Culture Collection (ATCC) No .: CRL1651] [Methods in Nucleic Acids Res., CRC press, 283 (1991)].
- the expression level and antigen-binding activity of the recombinant antibody in the culture supernatant were determined by enzyme-linked immunosorbent assay (Monoclonal Antibodies-Principles and practice, Third Edition, Academic Press (1996), Antibodies-A Laboratory Manual, Cold Spring Harbor Laboratory (1988), monoclonal antibody experiment manual, Kodansha Scientific (1987)].
- any host cell that can express the recombinant antibody can be used.
- CHO-K1 ATCC @ CCL-61
- DUKXB11 ATCC @ CCL-9096
- Pro-5 ATCC @ CCL-1781
- CHO-S Life @ Technologies, Cat # 11619
- mice 20 (ATCC No .: CRL1662 or YB2 / 0), mouse myeloma cell NS0, mouse myeloma cell SP2 / 0-Ag14 (ATCC No .: CRL1581), mouse P3X63-Ag8.653 cell (ATCC No .: CRL1580), dhfr CHO cells (CHO / DG44 cells) [Proc. Natl. Acad. Sci. USA, 77, 4216 (1980)] or the like are used.
- a protein such as an enzyme involved in the synthesis of intracellular sugar nucleotide GDP-fucose; a sugar chain modification in which the 1-position of fucose is ⁇ -linked to the 6-position of N-acetylglucosamine at the reducing end of an N-glycoside-linked complex-type sugar chain.
- Cells that have reduced or deleted activities such as proteins involved in the transport of the intracellular sugar nucleotide GDP-fucose to the Golgi apparatus, for example, the ⁇ 1,6-fucose transferase gene is deficient.
- CHO cells WO 2005/035586, WO 02/31140
- Lec13 Somatic Cell and Molecular Genetics, ⁇ 12, ⁇ 55 ⁇ (1986)
- a transformant that stably expresses the recombinant antibody is selected by culturing in an animal cell culture medium containing a drug such as G418 sulfate (hereinafter, referred to as G418) (Japan). KOKAI Publication No. 2-25791).
- the culture medium for animal cells includes RPMI1640 medium (manufactured by Invitrogen), GIT medium (manufactured by Nippon Pharmaceutical), EX-CELL301 medium (manufactured by JR HR), IMDM medium (manufactured by Invitrogen) or Hybridoma-SFM (manufactured by Invitrogen) Or a medium in which various additives such as FBS are added to these media.
- the recombinant antibody is expressed and accumulated in the culture supernatant.
- the expression level and antigen-binding activity of the recombinant antibody in the culture supernatant can be measured by ELISA or the like. Further, the expression level of the recombinant antibody produced by the transformed strain can be improved by using a dhfr gene amplification system (Japanese Patent Application Laid-Open No. 2-25791).
- the recombinant antibody is purified from the culture supernatant of the transformant using a protein A-column [Monoclonal Antibodies-Principles and practice, Third Edition, Academic Press (1996), Antibodies-A Laboratory, Cold Spring Harbor Laboratory]. (1988)]. Also, methods used for protein purification such as gel filtration, ion exchange chromatography, and ultrafiltration can be combined.
- the molecular weight of the purified H chain, L chain or whole antibody molecule of the recombinant antibody can be determined by polyacrylamide gel electrophoresis [Nature, 227, 680 (1970)] or Western blotting [Monoclonal Antibodies-Principles and practice, Third] edition, Academic Press (1996), Antibodies-A Laboratory Manual, Cold Spring Harbor Laboratory (1988)] and the like.
- the antibody fragment of the present invention can be prepared according to a known method.
- the antibody fragment of the present invention may be produced by cleaving an antibody produced according to the method described in the above (1) to (8) with an enzyme or the like, or may be prepared by encoding a base sequence encoding a desired antibody fragment. It may be prepared and produced by a genetic engineering technique.
- bispecific antibody or multispecific antibody of the present invention can be produced according to the above-described method for producing an antibody.
- a bispecific antibody expression vector in which a scFv that binds to CSPG5 is fused to the C-terminus of an IgG antibody that binds to an antigen present in the brain can be prepared by the method described below.
- the bispecific antibody can be prepared according to the method and the antibody purification method.
- a bispecific antibody in which an antibody fragment is fused to the C-terminus of an antibody can be prepared in the same manner.
- the gene fragment of the CH1-Hinge-CH2-CH3-linker region is amplified by PCR using a synthetic gene of the heavy chain constant region of an IgG antibody that binds to an antigen present in the brain as a template.
- the base sequence of the antibody that binds to CSPG5 is prepared by PCR or the like.
- the above two regions are joined by a PCR method or the like, and the obtained gene fragment is inserted into an appropriate vector such as a pCI vector.
- a gene fragment of a light chain region (VL and CL) of an IgG antibody that binds to an antigen present in the brain and a gene fragment of a VH of the antibody are amplified by a PCR method using an appropriate template, respectively. Insert into the appropriate position.
- the bispecific antibody of the present invention can also be prepared by binding an antigen binding site containing an antibody fragment to an IgG antibody by a chemical technique.
- activity evaluation of Antibody or Antibody Fragment In the present invention, activity evaluation of the antibody or the antibody fragment can be performed as follows.
- Binding activity to CSPG5 The binding activity of the antibody or the antibody fragment of the present invention to CSPG5 is measured by using flow cytometry, ELISA, surface plasmon resonance detection and the like described in 1- (6) above. Alternatively, it can be measured using a fluorescent antibody method [Cancer Immunol. Immunother., 36, 373 (1993)].
- the antibody or the antibody fragment of the present invention is a monovalent antibody that binds to CSPG5
- the binding activity of the monovalent antibody to CSPG5 can be measured by the same method.
- the antibody or the antibody fragment of the present invention is a bispecific antibody or a multispecific antibody that binds to CSPG5 and an antigen present in the brain
- the bispecific antibody or the multispecific antibody is present in the CSPG5 or the brain in a similar manner. The activity of binding to an antigen can be measured.
- the brain tissue After several days after administering the antibody or the antibody fragment to the animal, the brain tissue is collected, the concentration of the antibody or the antibody fragment in the supernatant after homogenization and centrifugation is measured, and the antibody or the antibody per unit brain weight is measured. Examples include a method of calculating the amount of the antibody fragment, and a method of detecting the presence of the antibody or the antibody fragment by a known immunological technique using the collected brain tissue. In addition, a method of administering an antibody or an antibody fragment thereof labeled with a pharmacologically acceptable label to an animal and detecting the presence of the antibody or the antibody fragment over time using an in vivo imaging system, and the like can be mentioned.
- an appropriate animal can be selected according to the use of the antibody or the antibody fragment of the present invention.
- ADCC Antibody-Dependent Cytotoxic Activity
- CDC Complement-Dependent Cytotoxic Activity
- the effector activity refers to an antibody-dependent activity caused through an Fc region of an antibody or an antibody fragment thereof, and is dependent on ADCC, CDC, or phagocytic cells such as macrophages or dendritic cells.
- ADCC antibody-dependent activity caused through an Fc region of an antibody or an antibody fragment thereof
- phagocytic cells such as macrophages or dendritic cells.
- ADP Dependent phagocytosis
- a target cell for example, a human peripheral blood mononuclear cell (PBMC) as an effector, and a target cell-specific antibody or an antibody fragment thereof are mixed and incubated for about 4 hours. Lactate dehydrogenase (LDH) that has been released can be measured. Alternatively, the effector activity can be measured by a free 51 Cr method or a flow cytometry method.
- PBMC peripheral blood mononuclear cell
- LDH Lactate dehydrogenase
- the effector activity can be measured by a free 51 Cr method or a flow cytometry method.
- the effector activity of the antibody or the antibody fragment containing Fc can be increased or decreased by controlling the core fucose content of the N-linked complex type sugar chain of Fc of the antibody.
- a method for reducing the content of fucose bound to the N-linked complex-type sugar chain bound to Fc of the antibody or the antibody fragment a method for reducing the amount of fucose by using CHO cells lacking ⁇ 1,6-fucose transferase gene or By expressing the antibody fragment, an antibody to which fucose is not bound or the antibody fragment can be obtained.
- Antibodies or antibody fragments to which fucose is not bound have high ADCC.
- a method for increasing the content of fucose bound to the N-linked complex type sugar chain bound to Fc of the antibody or the antibody fragment a method of increasing the amount of fucose by using a host cell into which ⁇ 1,6-fucose transferase gene has been introduced
- an antibody to which fucose is bound or the antibody fragment can be obtained by expressing the antibody fragment.
- the antibody or antibody fragment to which fucose is bound has a lower ADCC than the antibody or antibody fragment to which fucose is not bound.
- AD ADCC or CDC can be increased or decreased by modifying amino acid residues in the Fc region of the antibody or the antibody fragment. For example, by using the amino acid sequence of the Fc region described in US Patent Application Publication No. 2007/0148165, CDC of an antibody or an antibody fragment thereof can be increased.
- ADCC or CDC can be obtained by amino acid modification described in US Pat. No. 6,737,056, US Pat. No. 7,297,775, or US Pat. No. 7,317,091. Can be increased or decreased.
- the antibody or the antibody fragment of the present invention may be modified, for example, according to Japanese Patent Application Publication No. 2013-165716 or Japanese Patent An antibody or an antibody fragment thereof in which the half-life in blood is controlled by controlling the reactivity to an Fc receptor by performing amino acid modification described in Japanese Patent Application Publication No. 2012-021004 or the like is also included.
- an antibody or an antibody fragment having a controlled effector activity or half-life in blood can be obtained.
- the antibody or the antibody fragment of the present invention can be used for treating a brain disease in an animal in which CSPG5 is expressed in the brain.
- brain diseases for example, Alzheimer's disease, prodromal Alzheimer's disease, Huntington's disease, Parkinson's disease, brain tumor, multiple sclerosis, muscular dystrophy, amyotrophic lateral sclerosis, multiple system atrophy, progressive supranuclear palsy, Striatum substantia nigra, olive bridge cerebellar atrophy, spinobulbar muscular atrophy, spinocerebellar degeneration, cerebrovascular disorder, epilepsy, migraine, hyperactivity disorder, Creutzfeldt-Jakob disease, basal ganglia degeneration Disease, lysosomal disease, depression, dystonia and the like.
- Brain diseases that can be treated by the antibody or the antibody fragment of the present invention include antigens to which the antibody or the antibody fragment of the present invention binds and types of molecules that modify the antibody or the antibody fragment in the fusion antibody or the fusion antibody fragment of the present invention. Depends on such factors.
- the therapeutic agent containing the antibody or the antibody fragment of the present invention may contain only the antibody or the antibody fragment as an active ingredient, but is usually combined with one or more pharmacologically acceptable carriers. They are mixed together and provided as pharmaceutical preparations prepared by methods known in the art of pharmacy.
- the administration route may be, for example, oral administration or non-administration such as intraoral, respiratory, rectal, subcutaneous, intramuscular, intraventricular, intraperitoneal, intradermal, intranasal, intrathecal or intravenous administration.
- Oral administration is included.
- Examples of the administration form include sprays, capsules, tablets, powders, granules, syrups, emulsions, suppositories, injections, ointments and tapes.
- Suitable formulations for oral administration include emulsions, syrups, capsules, tablets, powders or granules.
- Liquid preparations such as emulsions or syrups include water, sugars such as sucrose, sorbitol or fructose, glycols such as polyethylene glycol or propylene glycol, oils such as sesame oil, olive oil or soybean oil, p-hydroxybenzoic acid It is manufactured using preservatives such as esters or flavors such as strawberry flavor or peppermint as additives.
- Capsules, tablets, powders or granules include excipients such as lactose, glucose, sucrose or mannitol; disintegrants such as starch or sodium alginate; lubricants such as magnesium stearate or talc; polyvinyl alcohol; It is manufactured using a binder such as propylcellulose or gelatin, a surfactant such as a fatty acid ester, or a plasticizer such as glycerin as an additive.
- Suitable formulations for parenteral administration include injections, suppositories and sprays.
- the injection is prepared using a carrier comprising a salt solution or a glucose solution or a mixture of both.
- Suppositories are prepared using carriers such as cocoa butter, hydrogenated fats or carboxylic acids.
- Sprays are produced by using a carrier or the like which does not irritate the oral and respiratory tract mucous membranes of the recipient and disperses the antibody or the antibody fragment thereof of the present invention as fine particles to facilitate absorption.
- a carrier for example, lactose or glycerin is used. It can also be manufactured as an aerosol or dry powder. Further, also in the parenteral preparation, the components exemplified as additives in a preparation suitable for oral administration can be added.
- Method for detecting or measuring an antigen present in the brain using the antibody or the antibody fragment of the present invention, or a method for diagnosing disease Using the antibody or the antibody fragment of the present invention, CSPG5, or CSPG5 and an antigen present in the brain Can be detected or measured. Further, by detecting or measuring CSPG5 or CSPG5 and an antigen present in the brain, a brain disease of an animal in which CSPG5 is expressed in the brain can be diagnosed.
- brain diseases for example, Alzheimer's disease, prodromal Alzheimer's disease, Huntington's disease, Parkinson's disease, brain tumor, multiple sclerosis, muscular dystrophy, amyotrophic lateral sclerosis, multiple system atrophy, progressive supranuclear palsy, Striatum substantia nigra, olive bridge cerebellar atrophy, spinobulbar muscular atrophy, spinocerebellar degeneration, cerebrovascular disorder, epilepsy, migraine, hyperactivity disorder, Creutzfeldt-Jakob disease, basal ganglia degeneration Disease, lysosomal disease, depression, dystonia, etc., and brain diseases in which the antibody or the antibody fragment of the present invention can be diagnosed include the antigen to which the antibody or the antibody fragment of the present invention binds, and the fusion antibody or the present invention of the present invention.
- the fusion antibody fragment differs depending on the type of the antibody or the molecule modifying the antibody fragment.
- Diagnosis of a brain disease in an animal in which CSPG5 is expressed in the brain can be performed, for example, by detecting or measuring CSPG5 present in the brain of a patient or an affected animal by an immunological technique.
- diagnosis can be performed by detecting CSPG5 expressed or present in cells in the brain of the patient or diseased animal using an immunological technique such as flow cytometry.
- CSPG5 in the brain can be measured by the same method as described above.
- a bispecific antibody or a multispecific antibody that binds to CSPG5 and an antigen present in the brain is used as the antibody or the antibody fragment of the present invention, CSPG5 in the brain or an antigen present in the brain is detected or measured in the same manner as described above. can do.
- the immunological technique is a method for detecting or measuring the amount of an antibody or the amount of an antigen using a labeled antigen or antibody.
- a radioactive substance-labeled immunoantibody method an enzyme immunoassay, a fluorescence immunoassay, a luminescence immunoassay, a western blot method, a physicochemical technique, or the like is used.
- radioactive substance-labeled immunoantibody method for example, an antigen or a cell expressing the antigen is reacted with the antibody of the present invention or the antibody fragment, and further reacted with a radiolabeled anti-immunoglobulin antibody or the antibody fragment. Thereafter, measurement is performed using a scintillation counter or the like.
- an antigen or a cell expressing the antigen was reacted with the antibody or the antibody fragment of the present invention, and further reacted with an anti-immunoglobulin antibody or an antibody fragment labeled with an enzyme or the like. Thereafter, the substrate is added, and the absorbance of the reaction solution is measured with an absorptiometer. For example, a sandwich ELISA method or the like is used.
- a label used in the enzyme immunoassay a known enzyme label [enzyme immunoassay, Medical Shoin (1987)] can be used.
- the sandwich ELISA method is a method of binding an antibody to a solid phase, trapping an antigen to be detected or measured, and reacting the trapped antigen with a second antibody.
- the ELISA method two types of antibodies that recognize an antigen to be detected or measured and have different antigen recognition sites are prepared, and the first antibody is adsorbed on a plate (for example, a 96-well plate) in advance.
- the second antibody is labeled with a fluorescent substance such as FITC, an enzyme such as peroxidase, or biotin.
- the antigen concentration in the test sample is calculated from a calibration curve prepared by serially diluting an antigen having a known concentration.
- the antibody used in the sandwich ELISA method either a polyclonal antibody or a monoclonal antibody may be used. Further, an antibody fragment such as Fab, Fab ′ or F (ab) 2 may be used in place of the antibody.
- the combination of two types of antibodies used in the sandwich ELISA method may be a monoclonal antibody recognizing different epitopes or a combination of the antibody fragments, or a polyclonal antibody and a monoclonal antibody or a combination of those antibody fragments.
- the fluorescence immunoassay is carried out by a method described in the literature [Monoclonal Antibodies-Principles and practice, Third Edition, Academic Press (1996), Manual for Monoclonal Antibody Experiment, Kodansha Scientific (1987)].
- a label used in the fluorescent immunoassay a known fluorescent label [fluorescent antibody method, Soft Science Inc. (1983)] can be used.
- FITC or RITC is used.
- the luminescence immunoassay is measured by a method described in literature [Bioluminescence and chemiluminescence Clinical test 42, Hirokawa Shoten (1998)].
- Examples of the label used in the luminescence immunoassay include well-known luminescent labels, such as acridinium ester or lophine.
- antigens or cells expressing the antigens are fractionated by SDS (sodium dodecyl sulfate) -PAGE (polyacrylamide gel) [Antibodies-A Laboratory Manual Cold Spring Spring Harbor Laboratory (1988)], and then the gel is separated.
- SDS sodium dodecyl sulfate
- PAGE polyacrylamide gel
- the membrane was reacted with an antibody recognizing an antigen or the antibody fragment, and further subjected to a fluorescent substance such as FITC, an enzyme label such as peroxidase, or a biotin label.
- FITC fluorescent substance
- an enzyme label such as peroxidase
- biotin label such as peroxidase
- Cells or tissues expressing the polypeptide having the amino acid sequence of CSPG5 are lysed, and 0.1 to 30 ⁇ g of protein per lane is electrophoresed by SDS-PAGE under reducing conditions.
- the electrophoresed protein is transferred to a PVDF membrane and reacted with BSA-PBS at room temperature for 30 minutes to perform a blocking operation.
- the antibody of the present invention or the antibody fragment is reacted, and washed with PBS containing 0.05 to 0.1% of polyoxyethylene sorbitan monolaurate (Tween-20) (hereinafter referred to as Tween-PBS). Then, a peroxidase-labeled goat anti-mouse IgG is reacted at room temperature for 2 hours.
- PBS polyoxyethylene sorbitan monolaurate
- the polypeptide having the amino acid sequence of CSPG5 is detected by washing with Tween-PBS and detecting the band to which the antibody or the antibody fragment of the present invention is bound by using ECL Western Blotting Detection Reagents (manufactured by Amersham) or the like. .
- an antibody or an antibody fragment used for detection by Western blotting an antibody or an antibody fragment capable of binding to a polypeptide that does not retain a natural three-dimensional structure is used.
- the physicochemical method is performed by, for example, binding CSPG5 as an antigen to the antibody or the antibody fragment of the present invention to form an aggregate, and detecting the aggregate.
- a physicochemical method a capillary method, a one-dimensional immunodiffusion method, an immunoturbidimetric method, or a latex immunoturbidimetric method [Procedure of clinical test method, Kinbara Shuppan (1998)] can be used.
- a carrier such as polystyrene latex having a particle size of about 0.1 to 1 ⁇ m sensitized with an antibody or an antigen is used. Scattered light increases and transmitted light decreases. This change is detected as absorbance or turbidity of the integrating sphere to measure the antigen concentration in the test sample.
- CMOS complementary metal-oxide-semiconductor
- CMOS complementary metal-oxide-semiconductor
- immunoprecipitation immunocytostaining
- immunohistochemistry immunohistochemistry
- fluorescent antibody staining can be used.
- it is used.
- the immunoprecipitation method comprises reacting cells expressing CSPG5 or the like with the antibody or the antibody fragment of the present invention, and then adding a carrier having a specific binding ability to immunoglobulin such as protein G-Sepharose, and adding the antigen-antibody complex. To settle. Alternatively, it can be performed by the following method.
- the BSA-PBS is discarded, and after washing well with PBS, a lysate of cells or tissues expressing human CSPG5 is reacted.
- the immunoprecipitate is extracted from the well-washed plate with a sample buffer for SDS-PAGE, and detected by Western blotting as described above.
- cells or tissues expressing the antigen may be treated with a surfactant or methanol in order to improve antibody permeability, and then reacted with the antibody of the present invention.
- a fluorescent label such as FITC
- an enzyme label such as peroxidase or a biotin label, or the like, or an immunoglobulin antibody or a binding fragment thereof, the label is visualized, and then microscopically observed with a microscope.
- the formed antibody-antigen complex and free antibody not involved in the formation of the antibody-antigen complex are used.
- the antigen amount or the antibody amount can be measured without separating the antibody or the antigen.
- Example 1 Acquisition of anti-CSPG5 antibody (1) Acquisition of antibody using human antibody phage library From a cDNA derived from human PBMC, a VH gene fragment and a VL gene fragment were amplified by PCR. The VH gene fragment and the VL gene fragment were respectively inserted into a phagemid vector pCANTAB 5E (manufactured by Amersham Pharmacia), and Escherichia coli TG1 (manufactured by Lucigen) was transformed to obtain a plasmid. By infecting the obtained plasmid with M13KO7 Helper Page (manufactured by Invitrogen), a human antibody M13 phage library in which VH and VL genes were libraryd was obtained. In addition, a synthetic human antibody M13 phage library in which a random mutation was introduced into CDR3 was similarly prepared.
- pCANTAB 5E manufactured by Amersham Pharmacia
- Escherichia coli TG1 manufactured by Lucigen
- anti-CSPG5 monoclonal antibodies were obtained by the following phage display method.
- the human CSPG5-FLAG_Fc or the mouse CSPG5-FLAG_Fc of Example 4 described below was solid-phased in MAXISORP STARTUBE (manufactured by NUNC) and blocked using SuperBlock ⁇ Blockig ⁇ Buffer (manufactured by Thermo).
- the tube was reacted with a human antibody M13 phage library at room temperature for 1 hour, washed with PBS or PBS containing 0.1% @ Tween 20 (hereinafter referred to as PBS-T), and then washed with 0.1 mol / L glycine-HCl buffer.
- the phage was eluted with (Gly-HCl) (pH 2.2).
- the eluate was neutralized by adding Trishydroxymethylaminomethane-hydrochloric acid buffer (Tris-HCl) (pH 8.5).
- Tris-HCl Trishydroxymethylaminomethane-hydrochloric acid buffer
- the eluted phages were infected with TG1 competent cells, and the phages were amplified. Thereafter, the resultant was reacted again with human @ CSPG5-FLAG_Fc or mouse @ CSPG5-FLAG_Fc immobilized on MAXISORP @ STARTUBE to perform washing and
- This operation was repeated to concentrate phage displaying scFv that specifically bind to human CSPG5-FLAG_Fc and mouse CSPG5-FLAG_Fc.
- the concentrated phage was cloned, and clones having binding properties to human @ CSPG5-FLAG_Fc and mouse @ CSPG5-FLAG_Fc were selected by ELISA.
- human CSPG5-FLAG_Fc and mouse CSPG5-FLAG_Fc were immobilized on MAXISORP (manufactured by NUNC) and blocked using SuperBlock ⁇ Blockig ⁇ Buffer (manufactured by Thermo). A plate on which Fc was immobilized was also prepared as a negative control.
- Each phage clone was added to each well and reacted at room temperature for 30 minutes, and then each well was washed with PBS-T.
- a solution obtained by diluting an anti-M13 antibody (manufactured by GE Healthcare) labeled with horseradish peroxidase with PBS-T containing 10% Block Ace (manufactured by Dainippon Pharmaceutical Co., Ltd.) was added to each well. Incubated for minutes. After washing the microplate three times with PBS-T, 3,3 ', 5,5'-tetramethylbenzidine (TMB) chromogenic substrate solution (manufactured by DAKO) was added and incubated at room temperature. The coloring reaction was stopped by adding 0.5 mol / L sulfuric acid to each well, and the absorbance at a wavelength of 450 nm (reference wavelength 570 nm) was measured with a microplate reader.
- TMB 5,5'-tetramethylbenzidine
- human CSPG5-FLAG_Fc and mouse performs clones for sequence analysis bound to CSPG5-FLAG_Fc, as an anti CSPG5 antibody phagemid vector, pCANTAB_CSPG5115, pCANTAB_CSPG5120, pCANTAB_CSPG5168, pCANTAB_CSPG5201, pCANTAB_CSPG5202, pCANTAB_CSPG5205, pCANTAB_CSPG5206, pCANTAB_CSPG5207, pCANTAB_CSPG5208, pCANTAB_CSPG5214, pCANTAB_CSPG5219, pCANTAB_CSPG5222, pCANTAB_CSPG5227, pCANTAB_CSPG5230 and pCANTAB_C It has acquired the 15 types of PG5234.
- Table 1 shows the nucleotide sequences encoding VH and VL of various anti-CSPG5 antibodies, and the amino acid sequences deduced from the nucleotide sequences.
- Example 2 Production of antibody (1) Construction of CSPG5 scFv-hG4PE (R409K) expression vector Human IgG4 antibody containing amino acid residue substitutions of EU numbering S228P, L235E and R409K (hereinafter sometimes abbreviated as IgG4 variant) )), An expression vector was constructed to prepare an scFv-Fc antibody in which each anti-CSPG5 scFv antibody was bound to the Fc region. Using the phagemid vector pCANTAB_CSPG5115 as a template, the gene fragment of the scFv region was amplified by PCR.
- the gene fragment of the Hinge-CH2-CH3 region was amplified by PCR.
- the obtained gene fragment was inserted into a pCI vector (Promega) to prepare a pCI_CSPG5115 scFv-hG4PE (R409K) vector.
- antibody expression vectors into which gene fragments of the scFv regions of the various anti-CSPG5 antibodies shown in Table 1 were inserted were prepared, and the pCI_CSPG5120 @ scFv-hG4PE (R409K) vector, pCI_CSPG5168 @ scFv-hG4PE (R409K) vector, and pCI_CSPG5201 @ scFv, respectively.
- Vector pCI_CSPG5214 @ scFv-hG4PE (R409K) vector, pCI_CSPG5219 @ scFv-hG4PE (R409K) vector, pCI_CSPG5222 @ scFv-hG4PE (R409K) vector, pCI_CSPG5227 @ scFv-hG4PEC (R409KVPGCSPvCSGVPEGRPCRPGVPECRPCRPGVPECRPCPGRPC30GVPECRPCRPG30PEK). It was designated as hG4PE (R409K) vector.
- VL and VH gene fragments were amplified by PCR using the phagemid vector pCANTAB_CSPG5202 as a template. Using the synthetic gene as a template, CL and CH gene fragments were amplified by PCR. The obtained gene fragment was inserted into a pCI vector (Promega) to prepare a pCI_CSPG5202-hKG4PE (R409K) vector.
- variable region of the anti-AVM antibody as a template, the VL and VH gene fragments, the phagemid vector pCANTAB_CSPG5202 as a template, the CSPG5202 scFv region gene fragment, and the synthetic gene as a template for CL and CH1-Hinge-CH2-CH3-linker regions.
- the gene fragment was amplified by PCR.
- the obtained gene fragment was inserted into a pCI vector (Promega) to prepare a pCI_AVM-hLG4PE (R409K) -CSPG5202scFv vector.
- Table 2 shows the name of the antibody expression vector, the nucleotide sequence encoding the light or heavy chain, and the amino acid sequence deduced from the nucleotide sequence.
- the base sequence encoding the synthesized human IgG lambda chain constant region and the amplified variable region were inserted into a N5KG4PE vector (described in WO2002 / 088886) to prepare an expression vector N5LG4PE_AVM.
- the antibody expression plasmid vector was introduced into Expi293F cells (manufactured by Thermo Fisher Scientific) using Expi293 (trademark) Expression System (manufactured by Thermo Fisher Scientific), cultured, and cultured. Was expressed.
- the culture supernatant was collected 3 to 4 days after the introduction of the vector, and filtered with a membrane filter having a pore size of 0.22 ⁇ m (manufactured by Merck Millipore).
- the antibody protein in the culture supernatant was affinity-purified using Protein A resin (MabSelect SuRe, manufactured by GE Healthcare Bioscience). A phosphate buffer was used as a washing solution.
- the protein adsorbed on protein A was eluted with 20 mmol / L sodium citrate and 50 mmol / L NaCl buffer (pH 3.4) and collected in a tube containing 1 mol / L Tris-HCl (pH 8.0). .
- the solvent of the eluate was replaced with PBS by ultrafiltration using Amicon Ultra (manufactured by Merck Millipore) and NAP column (manufactured by GE Healthcare Bioscience).
- the obtained solution was sterilized by filtration through a membrane filter having a pore size of 0.22 ⁇ m (manufactured by Merck Millipore). The absorbance at 280 nm of the antibody solution was measured, and the concentration of the purified antibody was calculated.
- the anti-CSPG5 @ scFv-Fc antibody obtained by expressing the vector prepared in Example 2 (1) was used as CSPG5115 @ scFv-hG4PE (R409K), CSPG5120 @ scFv-hG4PE (R409K), CSPG5168 @ scFv-hG4PE (R409K), CSPG5201.
- scFv-hG4PE R409K
- CSPG5202 scFv-hG4PE R409K
- CSPG5205 scFv-hG4PE R409K
- CSPG5206 scFv-hG4PE R409K
- CSPG5207 scFv-hG4PE R409K4GPEV
- CSPG5F4G 4009K4
- hG4PE (R409K) CSPG5219 @ scFv-hG PE
- CSPG5222 scFv-hG4PE R409K
- CSPG5227 scFv-hG4PE R409K
- CSPG5230 scFv-hG4PE R409K
- CSPG5234 scFv-hG4PE R409K
- the anti-CSPG5-IgG4 antibody obtained by expressing the pCI_CSPG5202-hKG4PE (R409K) vector prepared in Example 2 (2) was expressed by expressing the CSPG5202 @ IgG4PE (R409K) and pCI_AVM-hLG4PE (R409K) -CSPG5202scFv vectors.
- the AVM-IgG4-CSPG5 dscFv bispecific antibody was named AVM IgG4PE (R409K) _CSPG5202 dscFv.
- the IgG4_AVM @ dscFv bispecific antibody was named an anti-AVM antibody, AVM @ IgG4PE (R409K) _AVM @ dscFv5, respectively.
- Example 3 Reactivity analysis to CSPG5-expressing cells
- the nucleotide sequence encoding human CSPG5 is shown in SEQ ID NO: 159
- the amino acid sequence deduced from the nucleotide sequence is shown in SEQ ID NO: 160
- the nucleotide sequence encoding mouse CSPG5 is shown in SEQ ID NO: 160.
- the amino acid sequence deduced from the nucleotide sequence is shown in SEQ ID NO: 162
- the nucleotide sequence encoding monkey CSPG5 is shown in SEQ ID NO: 163
- the amino acid sequence deduced from the nucleotide sequence is shown in SEQ ID NO: 164.
- the full-length gene sequences of human CSPG5, mouse CSPG5 and monkey CSPG5 are synthesized, and the respective gene sequences are inserted into the BamHI-NotI site of the pEF6 / V5-His (Thermo Fisher Scientific) vector to produce various CSPG5 membrane expressions.
- plasmid vectors pEF6_human ⁇ CSPG5, pEF6_mouse ⁇ CSPG5, and pEF6_monkey ⁇ CSPG5 were prepared.
- Expi293F cells human CSPG5 / Expi293F cells, mouse CSPG5 / Expi293F cells, and monkey CSPG5 / Expi293F cells were respectively suspended in 0.1% NaN 3 , 1% FBS-containing PBS in a staining buffer (SB), and a 96-well round bottom plate was used.
- SB staining buffer
- the supernatant was removed by centrifugation (2000 rpm, 4 ° C., 2 minutes), the pellet was washed with SB, and 1 ⁇ g / mL RPE fluorescence-labeled goat anti-human antibody (manufactured by Southern Biotech) was added. Incubated for minutes. After washing with SB, the cells were suspended in SB, and the fluorescence intensity of each cell was measured with a flow cytometer FACS CANTO II (manufactured by Becton Dickinson). In addition, 10 ⁇ g / mL anti-AVM antibody was used as a negative control.
- the detection results were analyzed, and the mean fluorescence intensity (MFI) was calculated using the geometric mean. Furthermore, the ratio (average fluorescence intensity ratio) of human CSPG5 / Expi293F cells to Expi293F cells (parent strain) was calculated for MFI at a concentration of 10 ⁇ g / mL for each antibody. In the same manner, the average fluorescence intensity ratio of the monkey CSPG5 / Expi293F cells and the mouse CSPG5 / Expi293F cells to the Expi293F cells (parent strain) was calculated, and the results are shown in Table 3.
- MFI mean fluorescence intensity
- the average fluorescence intensity ratio was increased relative to the anti-AVM antibody as a negative control, indicating that human CSPG5 / Expi293F cells, mouse CSPG5 / Expi293F cells, and monkey CSPG5 / Expi293F cells.
- the cells showed reactivity. Therefore, it was revealed that the anti-CSPG5 antibody recognizes and binds to all of human CSPG5, mouse CSPG5, and monkey CSPG5.
- CSPG5202 @ IgG4PE (R409K) CSPG5202 @ scFv-hG4PE (R409K) and AVM @ IgG4PE (R409K) _CSPG5202 @ dscFv, Expi293F cells, human CSPG5 / Expi293F cells, and monkey CSPG3F3F3 cells were also used for the same procedure.
- the average fluorescence intensity ratio was increased with respect to the anti-AVM antibody as a negative control, and reacted with human CSPG5 / Expi293F cells, mouse CSPG5 / Expi293F cells and monkey CSPG5 / Expi293F cells. It became clear.
- Example 4 Preparation of soluble CSPG5 antigen (1) Preparation of extracellular domain protein of CSPG5 bound to FLAG_Fc Extracellular domain of CSPG5 with FLAG_Fc added to the C-terminus as a soluble antigen of human CSPG5 and mouse CSPG5
- the protein was produced by the method described below. By inserting the synthetic gene of the extracellular domain of human and mouse CSPG5 and the synthetic gene of FLAG_Fc into a pCI vector (Promega), the extracellular domain of human and mouse CSPG5 with FLAG_Fc added to the C-terminal side is expressed.
- the plasmid vectors pCI-human CSPG5-FLAG_Fc and pCI-mouse CSPG5-FLAG_Fc were prepared.
- the nucleotide sequence of human @ CSPG5-FLAG_Fc is shown in SEQ ID NO: 165, the amino acid sequence deduced from the nucleotide sequence is shown in SEQ ID NO: 166, the nucleotide sequence of mouse @ CSPG5-FLAG_Fc is shown in SEQ ID NO: 167, and the amino acid deduced from the nucleotide sequence is shown in SEQ ID NO: 167.
- the sequence is shown in SEQ ID NO: 168.
- pCI-human ⁇ CSPG5-FLAG_Fc and pCI-mouse ⁇ CSPG5-FLAG_Fc were expressed using Expi293 (trademark) ⁇ Expression ⁇ System (manufactured by Thermo ⁇ Fisher ⁇ Scientific), and then transfected into Expi293F cells. It was expressed and purified in the same manner as in Example 2. The concentration of purified human and mouse CSPG5-FLAG-Fc protein in the solution was measured by absorbance at 280 nm.
- nucleotide sequence of human @ CSPG5-GST is shown in SEQ ID NO: 169
- amino acid sequence deduced from the nucleotide sequence is shown in SEQ ID NO: 170
- nucleotide sequence of mouse @ CSPG5-GST is shown in SEQ ID NO: 171
- amino acid deduced from the nucleotide sequence The sequence is shown in SEQ ID NO: 172.
- pCI-human @ CSPG5-GST and pCI-mouse @ CSPG5-GST were each introduced into Expi293F cells using Expi293 (registered trademark) ⁇ Expression ⁇ System (manufactured by Thermo ⁇ Fisher ⁇ Scientific), and the protein was transiently expressed. Was expressed.
- the culture supernatant was collected 3 to 4 days after the introduction of the vector, and filtered with a membrane filter having a pore size of 0.22 ⁇ m (manufactured by Merck Millipore).
- the protein in the culture supernatant was affinity-purified using Glutathione Sepharose 4B (manufactured by GE Healthcare Bioscience). A phosphate buffer was used as a washing solution. The protein adsorbed on Glutathione Sepharose 4B was eluted with 50 mmol / L Tris-HCl, 10 mmol / L reduced gluta- tion (pH 8.0).
- the solvent in the solution was replaced with PBS by ultrafiltration using Amicon Ultra (manufactured by Merck Millipore) and NAP column (manufactured by GE Healthcare Bioscience).
- the obtained solution was sterilized by filtration through a membrane filter having a pore size of 0.22 ⁇ m (manufactured by Merck Millipore).
- the concentration of purified human and mouse CSPG5-GST protein in the solution was measured by absorbance at 280 nm.
- Example 5 Evaluation of Binding Ability to CSPG5 by Surface Plasmon Resonance Detection
- the affinity of the anti-CSPG5 antibody, CSPG5202 IgG4PE (R409K) and AVM IgG4PE (R409K) _CSPG5202 dscFv prepared in Example 2 to human CSPG5 and mouse CSPG5 was determined by Biacore. The measurement was performed using T-100 (GE Healthcare).
- dissociation constant with mouse CSPG5 was 1 ⁇ 10 ⁇ 8 mol / L or less for all antibodies.
- a buffer solution was added to the collected brain tissue, homogenized, and after centrifugation, the antibody solution eluted in the supernatant was collected. The volume was measured and the antibody concentration was measured by AlphaLISA (manufactured by PerkinElmer) to calculate the amount of antibody per unit brain weight. In addition, the standard curve was created using the antibody attached to the kit.
- FIG. 1 (A) The serum antibody concentration 3 days after antibody administration is shown in FIG. 1 (A), the amount of antibody per unit brain weight in brain tissue is shown in FIG. 1 (B), and the serum antibody concentration 9 days after antibody administration is shown in FIG. ) And the amount of antibody per unit brain weight in brain tissue is shown in FIG. 1 (D).
- the anti-CSPG5 ⁇ scFv-Fc antibody had no difference in serum concentration 3 days and 9 days after administration of the antibody as compared with the negative control (anti-AVM-IgG4 antibody).
- the negative control anti-AVM-IgG4 antibody.
- FIG. 1 (B) it was shown that the amount of antibody in the brain of the anti-CSPG5 ⁇ scFv-Fc antibody was increased about 4 to 17 times as compared with the negative control. Further, as shown in FIG.
- each antibody was administered to the mouse at a tail vein (iv) at 35 nmol / kg body weight, and blood was collected 7 days later. After systemic perfusion under anesthesia on the same day as the blood collection, brain tissue was collected and weighed. Further, a buffer solution was added to the collected brain tissue, homogenized, and after centrifugation, the antibody solution eluted in the supernatant was collected. The volume was measured and the antibody concentration was measured by AlphaLISA (manufactured by PerkinElmer) to calculate the amount of antibody per unit brain weight. The antibody concentration was represented by a value obtained by converting the molar concentration into the molecular weight (150 kDa) of the monoclonal antibody. In addition, the standard curve was created using each antibody.
- FIG. 2 (A) shows the serum antibody concentration of ⁇ anti-CSPG5 ⁇ scFv-Fc antibody; CSPG5227 ⁇ scFv-hG4PE (R409K), and FIG. 2 (B) shows the amount of antibody per unit brain weight in brain tissue. It was shown that the amount of antibody in the brain increased as compared with the negative control (anti-AVM-IgG4 antibody).
- FIG. 3 (A) shows the serum antibody concentration of anti-CSPG5 scFv-Fc antibody; CSPG5202 scFv-hG4PE (R409K) and anti-CSPG5-IgG4 antibody; CSPG5202 IgG4PE (R409K), and the antibody per unit brain weight in brain tissue. The amounts are shown in FIG.
- the amount of anti-CSPG5 scFv-Fc antibody As shown in FIG. 3 (B), the amount of anti-CSPG5 scFv-Fc antibody; The CSPG5-IgG4 antibody; CSPG5202 IgG4PE (R409K) was shown to increase about 5-fold.
- anti-AVM-IgG4-AVM dscFv bispecific antibody AVM IgG4PE (R409K) _AVM dscFv5, and anti-AVM-IgG4-CSPG5 dscFv bispecific antibody; AVM IgG4PE (R409K) _CSPG5202 dscFv )
- AVM IgG4PE (R409K) _CSPG5202 dscFv the amount of antibody in brain tissue per unit brain weight is shown in FIG.
- the anti-AVM-IgG4-CSPG5 dscFv bispecific antibody As shown in FIG. 3 (D), the anti-AVM-IgG4-CSPG5 dscFv bispecific antibody; AVM IgG4PE (R409K) _CSPG5202 dscFv compared to AVM IgG4PE (R409K) _AVM dscFv5, which is a negative control of the bispecific antibody. It was shown that the amount of antibody in the cells increased. As described above, it was shown that the bispecific antibody that binds to CSPG5 can increase the amount of antibodies in the brain compared to the bispecific antibody that does not bind to CSPG5.
- FIG. 4B shows a negative control ratio of a value obtained by correcting the amount of fluorescence in the brain by the fluorescence intensity of the administered antibody.
- FIG. 4 (B) it was shown that the amount of antibody in the brain of each of the anti-CSPG5 scFv-Fc antibodies was several times higher than that of the negative control. Among them, in the case of the anti-CSPG5 scFv-Fc antibody; CSPG5202-scFv-hG4PE (R409K), the amount of antibody in the brain is increased about 20-fold as shown in FIG. The distribution of the antibody was shown to be throughout the brain.
- Example 7 Evaluation of antibody internalization
- the pEF6_human CSPG5 prepared in Example 3 was subjected to mouse connective tissue-derived fibroblast L929 [American Type Culture Collection (ATCC) No .: CCL-] using HilyMax (manufactured by Dojindo Chemical Co., Ltd.). 1].
- the cells after the gene transfer were selected using the antibiotic Blasticidin (manufactured by Invitrogen), cloned by limiting dilution, and L929 cells expressing human CSPG5 on the cell surface (hereinafter abbreviated as human CSPG5 / L929 # 09).
- human CSPG5 / L929 # 09 L929 cells expressing human CSPG5 on the cell surface
- the internalization ability of the antibody prepared in Example 2 was analyzed by the following method. 96well plates at 5 ⁇ 10 3 cells / well human CSPG5 / L929 # 09 or human abdominal neuroblastoma IMR-32 [The European Collection of Authenticated Cell Cultures (ECACC) ID: 86041809] were seeded, one 37 ° C. Glued overnight.
- ECACC European Collection of Authenticated Cell Cultures
- CSPG5202FscFv-hG4PE R409K
- CSPG5219 scFv-hG4PE R409K
- CSPG5234 scFv-hG4PE R409K
- the CSPG5 binding antibody binds to CSPG5 expressed on the cell membrane and is internalized in both the forced expression strain and the cell line.
- SEQ ID NO: 1 Description of Artificial Sequence: Base Sequence Encoding VH of CSPG5115 Excluding Signal Sequence
- SEQ ID NO: 2 Description of Artificial Sequence: Amino Acid Sequence of VH of CSPG5115 Excluding Signal Sequence
- SEQ ID NO: 3 Description of Artificial Sequence : Amino acid sequence of HCDR1 of CSPG5115 SEQ ID NO: 4-Description of artificial sequence: Amino acid sequence of HCDR2 of CSPG5115 SEQ ID NO: 5-Description of artificial sequence: Amino acid sequence of HCDR3 of CSPG5115 SEQ ID NO: 6-Description of artificial sequence: Excluding signal sequence Nucleotide sequence encoding CSPG5115 VL SEQ ID NO: 7-Description of artificial sequence: Amino acid sequence of CSPG5115 VL excluding signal sequence SEQ ID NO: 8-Description of artificial sequence: Amino acid sequence of LCDR1 of CSPG5115 SEQ ID NO
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Molecular Biology (AREA)
- Cell Biology (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Genetics & Genomics (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Neurology (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Physics & Mathematics (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Food Science & Technology (AREA)
- Analytical Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Neurosurgery (AREA)
- Biophysics (AREA)
- Epidemiology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- Toxicology (AREA)
- Tropical Medicine & Parasitology (AREA)
Abstract
Description
(1)コンドロイチン硫酸プロテオグリカン-5(CSPG5)に結合する抗体または該抗体断片。
(2)抗体が脳滞留性を有する(1)に記載の抗体または該抗体断片。
(3)抗体が神経細胞および/またはアストロサイト結合性を有する抗体である(2)に記載の抗体または該抗体断片。
(4)抗体が下記(a)~(s)からなる群より選ばれる1である、(1)~(3)のいずれか1に記載の抗体または該抗体断片;
(a)重鎖可変領域(VH)の相補性決定領域(CDR)1~3のアミノ酸配列が、それぞれ配列番号3、4および5に記載されるアミノ酸配列を含み、かつ軽鎖可変領域(VL)のCDR1~3のアミノ酸配列が、それぞれ配列番号8、9および10に記載されるアミノ酸配列を含む抗体、
(b)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号13、14および15に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号18、19および20に記載されるアミノ酸配列を含む抗体、
(c)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号23、24および25に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号28、29および30に記載されるアミノ酸配列を含む抗体、
(d)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号33、34および35に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号38、39および40に記載されるアミノ酸配列を含む抗体、
(e)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号43、44および45に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号48、49および50に記載されるアミノ酸配列を含む抗体、
(f)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号53、54および55に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号58、59および60に記載されるアミノ酸配列を含む抗体、
(g)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号63、64および65に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号68、69および70に記載されるアミノ酸配列を含む抗体、
(h)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号73、74および75に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号78、79および80に記載されるアミノ酸配列を含む抗体、
(i)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号83、84および85に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号88、89および90に記載されるアミノ酸配列を含む抗体、
(j)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号93、94および95に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号98、99および100に記載されるアミノ酸配列を含む抗体、
(k)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号103、104および105に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号108、109および110に記載されるアミノ酸配列を含む抗体、
(l)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号113、114および115に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号118、119および120に記載されるアミノ酸配列を含む抗体、
(m)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号123、124および125に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号128、129および130に記載されるアミノ酸配列を含む抗体、
(n)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号133、134および135に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号138、139および140に記載されるアミノ酸配列を含む抗体、
(o)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号143、144および145に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号148、149および150に記載されるアミノ酸配列を含む抗体、
(p)前記(a)~(o)に記載の少なくとも1つの抗体と、CSPG5への結合について競合する抗体、
(q)前記(a)~(o)に記載のいずれか1つの抗体が結合するエピトープを含むエピトープに結合する抗体、
(r)前記(a)~(o)に記載のいずれか1つの抗体が結合するエピトープと同じエピトープに結合する抗体、および
(s)前記(a)~(o)に記載のいずれか1つの抗体のアミノ酸配列と85%以上の相同性を有するアミノ酸配列を含む抗体。
(5)抗体が下記(A)~(P)からなる群より選ばれる1である、(1)~(4)のいずれか1に記載の抗体または該抗体断片;
(A)VHのアミノ酸配列が配列番号2に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号7に記載されるアミノ酸配列を含む抗体、
(B)VHのアミノ酸配列が配列番号12に記載されるアミノ酸配列含み、かつVLのアミノ酸配列が配列番号17に記載されるアミノ酸配列を含む抗体、
(C)VHのアミノ酸配列が配列番号22に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号27に記載されるアミノ酸配列を含む抗体、
(D)VHのアミノ酸配列が配列番号32に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号37に記載されるアミノ酸配列を含む抗体、
(E)VHのアミノ酸配列が配列番号42に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号47に記載されるアミノ酸配列を含む抗体、
(F)VHのアミノ酸配列が配列番号52に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号57に記載されるアミノ酸配列を含む抗体、
(G)VHのアミノ酸配列が配列番号62に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号67に記載されるアミノ酸配列を含む抗体、
(H)VHのアミノ酸配列が配列番号72に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号77に記載されるアミノ酸配列を含む抗体、
(I)VHのアミノ酸配列が配列番号82に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号87に記載されるアミノ酸配列を含む抗体、
(J)VHのアミノ酸配列が配列番号92に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号97に記載されるアミノ酸配列を含む抗体、
(K)VHのアミノ酸配列が配列番号102に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号107に記載されるアミノ酸配列を含む抗体、
(L)VHのアミノ酸配列が配列番号112に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号117に記載されるアミノ酸配列を含む抗体、
(M)VHのアミノ酸配列が配列番号122に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号127に記載されるアミノ酸配列を含む抗体、
(N)VHのアミノ酸配列が配列番号132に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号137に記載されるアミノ酸配列を含む抗体、
(O)VHのアミノ酸配列が配列番号142に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号147に記載されるアミノ酸配列を含む抗体、および
(P)前記(A)~(O)に記載のいずれか1つの抗体のアミノ酸配列と85%以上の相同性を有するアミノ酸配列を含む抗体。
(6)抗体または該抗体断片がバイスペシフィック抗体である、(1)~(5)のいずれか1に記載の抗体または該抗体断片。
(7)バイスペシフィック抗体がCSPG5および脳に存在する抗原に結合する、(6)に記載のバイスペシフィック抗体。
(8)バイスペシフィック抗体がCSPG5に結合する抗原結合部位および脳に存在する抗原に結合する抗原結合部位を含む、(6)または(7)に記載のバイスペシフィック抗体。
(9)抗体断片がFab、Fab’、F(ab’)2、一本鎖抗体(scFv)、二量体化V領域(diabody)、ジスルフィド安定化V領域(dsFv)、重鎖抗体の重鎖可変領域(VHH)およびCDRを含むペプチドからなる群より選ばれる1である、(1)~(8)のいずれか1に記載の抗体断片。
(10)抗体が遺伝子組換え抗体である、(1)~(9)のいずれか1に記載の抗体および該抗体断片。
(11)抗体がマウス抗体、ラット抗体、ラビット抗体、アルパカ抗体、ラクダ抗体、ラマ抗体、キメラ抗体、ヒト化抗体およびヒト抗体からなる群より選ばれる1である、(1)~(10)のいずれか1に記載の抗体および該抗体断片。
(12)(1)~(11)のいずれか1に記載のCSPG5に結合する抗体または該抗体断片に、下記(i)~(iii)からなる群より選ばれる少なくとも1つを結合させた融合抗体または該融合抗体断片;
(i)親水性高分子、
(ii)両親媒性高分子、および
(iii)機能性分子。
(13)(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片を産生するハイブリドーマ。
(14)(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片をコードする塩基配列を含む核酸。
(15)(14)に記載の核酸を含むベクターを含む形質転換細胞。
(16)(13)に記載のハイブリドーマまたは(15)に記載の形質転換細胞を培養し、培養液から(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片を採取することを含む、(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片の製造方法。
(17)(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片を含む、組成物。
(18)脳に存在する抗原の検出または測定用の組成物である、(17)に記載の組成物。
(19)脳疾患の診断または治療するための組成物である、(17)に記載の組成物。
(20)(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片、または(17)に記載の組成物を用いて、脳に存在する抗原を検出または測定する方法。
(21)(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片、または(17)に記載の組成物を用いて、脳疾患を診断または治療する方法。
(22)(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片、または(17)に記載の組成物を用いて、抗体、該抗体断片、融合抗体または該融合抗体断片の脳滞留性を向上させる方法。
(23)(1)~(12)のいずれか1に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片、または(17)に記載の組成物を用いて、脳内の抗体量、該抗体断片量、融合抗体量または該融合抗体断片量を増加させる方法。
(a)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号3、4および5に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号8、9および10に記載されるアミノ酸配列を含む抗体、
(b)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号13、14および15に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号18、19および20に記載されるアミノ酸配列を含む抗体、
(c)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号23、24および25に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号28、29および30に記載されるアミノ酸配列を含む抗体、
(d)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号33、34および35に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号38、39および40に記載されるアミノ酸配列を含む抗体、
(e)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号43、44および45に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号48、49および50に記載されるアミノ酸配列を含む抗体、
(f)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号53、54および55に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号58、59および60に記載されるアミノ酸配列を含む抗体、
(g)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号63、64および65に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号68、69および70に記載されるアミノ酸配列を含む抗体、
(h)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号73、74および75に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号78、79および80に記載されるアミノ酸配列を含む抗体、
(i)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号83、84および85に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号88、89および90に記載されるアミノ酸配列を含む抗体、
(j)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号93、94および95に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号98、99および100に記載されるアミノ酸配列を含む抗体、
(k)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号103、104および105に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号108、109および110に記載されるアミノ酸配列を含む抗体、
(l)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号113、114および115に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号118、119および120に記載されるアミノ酸配列を含む抗体、
(m)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号123、124および125に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号128、129および130に記載されるアミノ酸配列を含む抗体、
(n)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号133、134および135に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号138、139および140に記載されるアミノ酸配列を含む抗体、
(o)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号143、144および145に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号148、149および150に記載されるアミノ酸配列を含む抗体
(p)前記(a)~(o)に記載の少なくとも1つの抗体と、CSPG5への結合について競合する抗体、
(q)前記(a)~(o)に記載のいずれか1つの抗体が結合するエピトープを含むエピトープに結合する抗体、
(r)前記(a)~(o)に記載のいずれか1つの抗体が結合するエピトープと同じエピトープに結合する抗体、
(s)前記(a)~(o)に記載のいずれか1つの抗体のアミノ酸配列と85%以上の相同性を有するアミノ酸配列を含む抗体。
(A)VHのアミノ酸配列が配列番号2に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号7に記載されるアミノ酸配列を含む抗体、
(B)VHのアミノ酸配列が配列番号12に記載されるアミノ酸配列含み、かつVLのアミノ酸配列が配列番号17に記載されるアミノ酸配列を含む抗体、
(C)VHのアミノ酸配列が配列番号22に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号27に記載されるアミノ酸配列を含む抗体、
(D)VHのアミノ酸配列が配列番号32に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号37に記載されるアミノ酸配列を含む抗体、
(E)VHのアミノ酸配列が配列番号42に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号47に記載されるアミノ酸配列を含む抗体、
(F)VHのアミノ酸配列が配列番号52に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号57に記載されるアミノ酸配列を含む抗体、
(G)VHのアミノ酸配列が配列番号62に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号67に記載されるアミノ酸配列を含む抗体、
(H)VHのアミノ酸配列が配列番号72に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号77に記載されるアミノ酸配列を含む抗体、
(I)VHのアミノ酸配列が配列番号82に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号87に記載されるアミノ酸配列を含む抗体、
(J)VHのアミノ酸配列が配列番号92に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号97に記載されるアミノ酸配列を含む抗体、
(K)VHのアミノ酸配列が配列番号102に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号107に記載されるアミノ酸配列を含む抗体、
(L)VHのアミノ酸配列が配列番号112に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号117に記載されるアミノ酸配列を含む抗体、
(M)VHのアミノ酸配列が配列番号122に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号127に記載されるアミノ酸配列を含む抗体、
(N)VHのアミノ酸配列が配列番号132に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号137に記載されるアミノ酸配列を含む抗体、
(O)VHのアミノ酸配列が配列番号142に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号147に記載されるアミノ酸配列を含む抗体、
(P)前記(A)~(O)に記載のいずれか1つの抗体のアミノ酸配列と85%以上の相同性を有するアミノ酸配列を含む抗体。
(1)抗体の二つの重鎖のうち、一方の重鎖(重鎖A)のCH3にS354C/T366W、もう一方の重鎖(重鎖B)のCH3にY349C/T366S/L368A/Y407Vのアミノ酸改変を加えたバイスペシフィック抗体。
(2)抗体のC末端に抗体断片を融合させたバイスペシフィック抗体。
(3)抗体のN末端に抗体断片を融合させたバイスペシフィック抗体。
本発明において、抗体または該抗体断片に化学修飾によって他の分子を修飾する場合、その修飾部位としては、抗体または抗体断片の定常領域が挙げられ、特にC末端またはS-S結合部位のCys残基が好ましい。 遺伝子工学的手法により、後から化学修飾可能な残基を、予め抗体または抗体断片の任意の位置に導入することも可能である。
また、遺伝子工学的手法により、直接他の分子を修飾する場合、その修飾部位としては、抗体または抗体断片の軽鎖または重鎖のN末端またはC末端が挙げられる。
親水性高分子または両親媒性高分子としては、例えば、ポリオキシアルキレン、ポリオールまたは多糖を含む分子などが挙げられる。
(1)抗原の調製
抗原となるCSPG5またはCSPG5発現細胞は、CSPG5全長またはその部分長をコードするcDNAを含む発現ベクターを、大腸菌、酵母、昆虫細胞または動物細胞などに導入することで得ることができる。また、CSPG5は、CSPG5を多量に発現している各種動物細胞株、動物細胞および動物組織などからCSPG5を精製することによっても得ることができる。
3~20週令のマウス、ラット、ラビットまたはハムスターなどの動物に、(1)で得られる抗原を免疫して、その動物の脾、リンパ節、末梢血中の抗体産生細胞を採取する。また被免疫動物としてラマ、アルパカ、ラクダなどの動物を用いることもできる。
骨髄腫細胞としては、マウスから得られた株化細胞を用い、例えば、8-アザグアニン耐性マウス(BALB/c由来)骨髄腫細胞株P3-X63Ag8-U1(P3-U1)[Current Topics in Microbiology and Immunology, 18, 1 (1978)]、P3-NS1/1-Ag41(NS-1)[European J. Immunology, 6, 511 (1976)]、SP2/0-Ag14(SP-2)[Nature, 276, 269 (1978)]、P3-X63-Ag8653(653)[J. Immunology, 123, 1548 (1979)]またはP3-X63-Ag8(X63)[Nature, 256, 495 (1975)]などが用いられる。
(2)で得られる融合用抗体産生細胞と(3)で得られる骨髄腫細胞をMinimu Essential Medium(MEM)培地またはリン酸緩衝生理食塩水(PBS;リン酸二ナトリウム1.83g、リン酸一カリウム0.21g、食塩7.65g、蒸留水1リットル、pH7.2)でよく洗浄し、細胞数が、融合用抗体産生細胞:骨髄腫細胞=5~10:1になるよう混合し、遠心分離した後、上清を除く。
プリスタン処理[2,6,10,14-テトラメチルペンタデカン(Pristane)0.5mLを腹腔内投与し、2週間飼育する]した8~10週令のマウスまたはヌードマウスに、(4)で得られるモノクローナル抗体産生ハイブリドーマを腹腔内に注射する。10~21日でハイブリドーマは腹水癌化する。
抗体の選択は以下に示すように、フローサイトメトリーを用いて、CSPG5発現細胞への抗体の結合性を測定することなどにより行う。CSPG5発現細胞は、細胞表面上にCSPG5が発現していればいずれの細胞でもよく、例えば、動物細胞、動物細胞株および(1)で得られるCSPG5強制発現細胞株などが挙げられる。
(7-1)抗体ファージライブラリの作製方法
本発明において、抗体ファージライブラリは免疫ライブラリ、ナイーブライブラリおよび合成ライブラリを用いることができる。各ライブラリの作製方法を以下に記載する。
(7-1)で作製した抗体ファージライブラリからの抗体ファージクローンの選択は、以下に示すELISA法を用いて行うことができる。
遺伝子組換え抗体の作製例として、以下にヒト型キメラ抗体およびヒト化抗体の作製方法を示す。遺伝子組換えのマウス抗体、ラット抗体、ラビット抗体、ハムスター抗体、ラクダ抗体、ラマ抗体、アルパカ抗体、およびヒト抗体、各種キメラ抗体、ならびに重鎖抗体なども同様の方法で作製することができる。
遺伝子組換え抗体発現用ベクターは、ヒト抗体のCHおよびCLをコードするDNAが組み込まれた動物細胞用発現ベクターであり、動物細胞用発現ベクターにヒト抗体のCHおよびCLをコードするDNAをそれぞれクローニングすることにより構築することができる。
非ヒト抗体のVHおよびVLをコードするcDNAの取得およびアミノ酸配列の解析は以下のようにして行うことができる。
非ヒト抗体を産生するハイブリドーマ細胞よりmRNAを抽出し、cDNAを合成する。合成したcDNAをファージまたはプラスミドなどのベクターにクローニングしてcDNAライブラリを作製する。
選択したファージクローンのプラスミドベクターから、ベクター部分またはV領域部分をコードするDNAをプローブとして用い、VHまたはVLの全塩基配列をそれぞれ決定し、塩基配列よりVHまたはVLの全アミノ酸配列をそれぞれ推定することができる。
(1)で得られる遺伝子組換え抗体発現用ベクターのヒト抗体のCHまたはCLをコードするそれぞれの遺伝子の上流に、それぞれ非ヒト抗体のVHまたはVLをコードするcDNAをそれぞれクローニングすることで、ヒト型キメラ抗体発現ベクターを構築することができる。
ヒト化抗体のVHまたはVLをコードするcDNAは、以下のようにして構築することができる。
ヒト化抗体は、非ヒト抗体のVHおよびVLのCDRのみをヒト抗体のVHおよびVLのFRに移植しただけでは、その抗原結合活性は元の非ヒト抗体に比べて低下する[BIO/TECHNOLOGY, 9, 266 (1991)]。
(1)で得られる遺伝子組換え抗体発現用ベクターのヒト抗体のCHまたはCLをコードするそれぞれの遺伝子の上流に、構築した遺伝子組換え抗体のVHまたはVLをコードするcDNAをそれぞれクローニングし、ヒト化抗体発現ベクターを構築することができる。
(3)および(6)で得られる遺伝子組換え抗体発現ベクター、またはそれらを改変した発現ベクターを用いて遺伝子組換え抗体の一過性発現を行い、作製した多種類のヒト型キメラ抗体、ヒト化抗体の抗原結合活性を効率的に評価することができる。
(3)および(6)で得られた遺伝子組換え抗体発現ベクターを適当な宿主細胞に導入することにより遺伝子組換え抗体を安定に発現する形質転換株を得ることができる。
宿主細胞への発現ベクターの導入には、エレクトロポレーション法[日本国特開平2-257891号公報、Cytotechnology, 3, 133 (1990)]などを用いる。
本発明の抗体断片は、公知の方法に従い作製することができる。本発明の抗体断片は、上記(1)~(8)にて記載した方法に従い作製した抗体を、酵素などで切断することにより作製してもよいし、所望の抗体断片をコードする塩基配列を調製し、遺伝子工学的な手法で作製してもよい。
本発明において、一価抗体は、国際公開第2014/054804号、国際公開第2011/090754号、国際公開第2007/048037号、および国際公開第2012/116927号などに記載する方法などで作製することができる。
本発明のバイスペシフィック抗体またはマルチスペシフィック抗体は、上述した抗体の製造方法に準じて作製することができる。例えば、国際公開第2009/131239号、国際公開第2014/054804号、国際公開第01/077342号、米国特許出願公開第2007/0071675号明細書、国際公開第2007/024715、Wu et al.,[Nature Biotechnology,2007,25(11),p.1290-1297]、Labrijn et al.,[PNAS 2013, vol.110, no.13, p5145-5150]、Jong et al., [http://dx.doi.org/10.1371/journal.pbio.1002344]、Kontermann et al., [mAbs 2012, vol.4, issue2, p182-197]、Spiess et al., [Molecular Immunology 67 (2015) 95-106]、Ridgway et al., [Protein engineering, 1996 vol.9 no.7 pp617-621、国際公開第2009/080251、国際公開第2010/151792および国際公開第2014/033074などに記載される方法を用いて作製することができる。
本発明において、抗体または該抗体断片の活性評価は、以下のように行うことができる。
本発明の抗体または該抗体断片のCSPG5に対する結合活性は、前述の1-(6)に記載のフローサイトメトリー、ELISA、および表面プラズモン共鳴検出などを用いて測定する。また、蛍光抗体法[Cancer Immunol. Immunother., 36, 373 (1993)]を用いて測定することもできる。
本発明の抗体または該抗体断片の脳滞留性は以下に記載する方法で測定することができる。
ヒトCSPG5発現細胞、またはCSPG5および脳に存在する抗原が発現している細胞に対する本発明の抗体または該抗体断片のCDC、またはADCCは公知の測定方法[Cancer Immunol. Immunother., 36, 373(1993); Current protocols in Immunology, Chapter7. Immunologic studies in humans, Editor, John E, Coligan et al., John Wiley & Sons,Inc.,(1993)]により測定することができる。
本発明の抗体または該抗体断片のエフェクター活性を制御する方法としては、抗体またはFcを含む該抗体断片のFc領域の297番目のアスパラギン(Asn)に結合するN結合複合型糖鎖の還元末端に存在するN-アセチルグルコサミン(GlcNAc)にα1,6結合するフコース(コアフコースともいう)の量を制御する方法(国際公開第2005/035586号、国際公開第2002/31140号、国際公開第00/61739号)、または抗体若しくは該抗体断片のFc領域のアミノ酸残基を改変することで制御する方法などが知られている。本発明の抗体または該抗体断片にはいずれの方法を用いても、エフェクター活性を制御することができる。
本発明の抗体または該抗体断片は、脳内にCSPG5が発現している動物の脳疾患の治療に用いることができる。
本発明の抗体または該抗体断片を用いて、CSPG5、またはCSPG5と脳に存在する抗原とを検出または測定することができる。また、CSPG5、またはCSPG5と脳に存在する抗原とを検出または測定することにより、脳内にCSPG5が発現している動物の脳疾患を診断することができる。
(1)ヒト抗体ファージライブラリでの抗体の取得
ヒトPBMC由来のcDNAから、PCRにてVH遺伝子断片およびVL遺伝子断片を増幅させた。VH遺伝子断片およびVL遺伝子断片をファージミドベクターpCANTAB 5E(Amersham Pharmacia社製)にそれぞれ挿入し、大腸菌TG1(Lucigen社製)を形質転換してプラスミドを得た。得られたプラスミドをM13KO7 Helper Phage(Invitrogen社製)に感染させることで、VH遺伝子およびVL遺伝子がライブラリ化されたヒト抗体M13ファージライブラリを得た。
また、CDR3にランダム変異を導入した合成ヒト抗体M13ファージライブラリを同様に作製した。
(1)CSPG5 scFv-hG4PE(R409K)発現ベクターの構築
EUナンバリングS228P、L235EおよびR409Kのアミノ酸残基置換を含むヒトIgG4抗体(以下、IgG4改変体と略記する場合もある)のFc領域に、各抗CSPG5 scFv抗体を結合させたscFv-Fc抗体を作製するために発現ベクターを構築した。
ファージミドベクターpCANTAB_CSPG5115を鋳型として、PCRによりscFv領域の遺伝子断片を増幅した。重鎖定常領域の合成遺伝子を鋳型として、PCRによりHinge-CH2-CH3領域の遺伝子断片を増幅した。得られた遺伝子断片をpCIベクター(Promega社製)に挿入し、pCI_CSPG5115 scFv-hG4PE(R409K)ベクターを作製した。
抗CSPG5202 scFv抗体の抗体可変領域をそれぞれヒトIgG4改変体のCLおよびCHに結合させた抗CSPG5202-IgG4抗体、ならびに抗AVM-IgG4抗体のC末端側に2つの抗CSPG5202 scFv抗体を結合させた抗AVM-IgG4-CSPG5 dscFv抗体を作製するために発現ベクターを構築した。ファージミドベクターpCANTAB_CSPG5202を鋳型として、PCRによりVLおよびVHの遺伝子断片を増幅した。合成遺伝子を鋳型として、PCRによりCLおよびCHの遺伝子断片を増幅した。得られた遺伝子断片をpCIベクター(Promega社製)に挿入し、pCI_CSPG5202-hKG4PE(R409K)ベクターを作製した。
ネガティブコントロール抗体として、キメラ抗Avermectin(AVM)抗体を作製した。SDラットにAVMを免疫し、通常の方法で抗AVM抗体産生ハイブリドーマを樹立した。該ハイブリドーマ由来の可変領域を鋳型として、PCRによりVLおよびVH領域の遺伝子断片を増幅した。合成したヒトIgGのラムダ鎖定常領域をコードする塩基配列および増幅した可変領域をN5KG4PEベクター(国際公開第2002/088186号に記載)に挿入し、発現ベクターN5LG4PE_AVMを作製した。
抗体発現プラスミドベクターをExpi293(商標) Expression System(Thermo Fisher Scientific社製)を用いてExpi293F細胞(Thermo Fisher Scientific社製)に導入して培養し、一過性発現系で抗体を発現させた。ベクター導入3~4日後に培養上清を回収し、孔径0.22μmのメンブランフィルター(メルクミリポア社製)でろ過した。この培養上清中の抗体タンパク質をProteinA樹脂(MabSelect SuRe、GEヘルスケアバイオサイエンス社製)を用いてアフィニティー精製した。洗浄液としてリン酸緩衝液を用いた。プロテインAに吸着させたタンパク質を、20mmol/Lのクエン酸ナトリウムおよび50mmol/LのNaCl緩衝液(pH3.4)により溶出し、1mol/L Tris‐HCl(pH8.0)を含むチューブに回収した。次に、アミコンウルトラ(メルクミリポア社製)を用いた限外ろ過およびNAPカラム(GEヘルスケアバイオサイエンス社製)により、溶出液の溶媒をPBSに置換した。得られた溶液を孔径0.22μmのメンブランフィルター(メルクミリポア社製)でろ過滅菌した。抗体溶液の280nmの吸光度を測定し、精製抗体の濃度を算出した。
ヒトCSPG5をコードする塩基配列を配列番号159に、該塩基配列から推定されるアミノ酸配列を配列番号160に、マウスCSPG5をコードする塩基配列を配列番号161に、該塩基配列から推定されるアミノ酸配列を配列番号162に、サルCSPG5をコードする塩基配列を配列番号163に、該塩基配列から推定されるアミノ酸配列を配列番号164に示す。
(1)FLAG_Fcが結合したCSPG5の細胞外ドメインタンパク質の作製
ヒトCSPG5およびマウスCSPG5の可溶性抗原として、C末端にFLAG_Fcが付加されたCSPG5の細胞外ドメインタンパク質を以下に記載する方法で作製した。ヒトおよびマウスCSPG5の細胞外ドメインの合成遺伝子、およびFLAG_Fcの合成遺伝子をpCIベクター(Promega社製)に挿入することにより、C末端側にFLAG_Fcが付加されたヒトおよびマウスCSPG5の細胞外ドメインを発現するプラスミドベクターpCI-human CSPG5-FLAG_FcおよびpCI-mouse CSPG5-FLAG_Fcを作製した。
ヒトCSPG5およびマウスCSPG5の可溶性抗原として、C末端にGSTが付加されたCSPG5の細胞外ドメインタンパク質を以下に記載する方法で作製した。ヒトおよまたはマウスCSPG5の細胞外ドメインの合成遺伝子、およびGSTの合成遺伝子をpCIベクター(Promega社製)に挿入することにより、C末端側にGSTが付加されたヒトおよびマウスCSPG5の細胞外ドメインを発現するプラスミドベクターpCI-human CSPG5-GSTおよびpCI-mouse CSPG5-GSTをそれぞれ作製した。
実施例2で作製した抗CSPG5抗体、CSPG5202 IgG4PE(R409K)およびAVM IgG4PE(R409K)_CSPG5202 dscFvのヒトCSPG5およびマウスCSPG5へのアフィニティーをBiacore T-100(GE Healthcare)を用いて測定した。
(1)抗体量測定
マウスに各抗体を9mg/kg体重で尾静脈(i.v.)投与し、3日後および9日後に採血を行った。採血と同じ日に麻酔下にて全身灌流後、脳組織を回収してその重さを測定した。
抗CSPG5 scFv-Fc抗体およびネガティブコントロール(抗AVM-IgG4抗体)について、Alexa FluorR 488 Protein Labeling Kit(Molecular Probes社製)にて標識を行った。標識後の各抗体を9mg/kg体重でマウスに尾静脈(i.v.)投与し、9日後に採血を行った。
実施例3で作製したpEF6_human CSPG5を、HilyMax(同仁化学社製)を用いてマウス結合組織由来線維芽細胞L929 [American Type Culture Collection(ATCC)番号: CCL-1]に導入した。遺伝子導入後の細胞を抗生物質Blasticidin(Invitrogen社製)により選抜した後、限界希釈法によるクローニングを行い、human CSPG5を細胞表面に発現したL929細胞(以下、human CSPG5/L929#09と略記する)を作製した。
配列番号2-人工配列の説明:シグナル配列を除いたCSPG5115のVHのアミノ酸配列
配列番号3-人工配列の説明:CSPG5115のHCDR1のアミノ酸配列
配列番号4-人工配列の説明:CSPG5115のHCDR2のアミノ酸配列
配列番号5-人工配列の説明:CSPG5115のHCDR3のアミノ酸配列
配列番号6-人工配列の説明:シグナル配列を除いたCSPG5115のVLをコードする塩基配列
配列番号7-人工配列の説明:シグナル配列を除いたCSPG5115のVLのアミノ酸配列
配列番号8-人工配列の説明:CSPG5115のLCDR1のアミノ酸配列
配列番号9-人工配列の説明:CSPG5115のLCDR2のアミノ酸配列
配列番号10-人工配列の説明:CSPG5115のLCDR3のアミノ酸配列
配列番号11-人工配列の説明:シグナル配列を除いたCSPG5120のVHをコードする塩基配列
配列番号12-人工配列の説明:シグナル配列を除いたCSPG5120のVHのアミノ酸配列
配列番号13-人工配列の説明:CSPG5120のHCDR1のアミノ酸配列
配列番号14-人工配列の説明:CSPG5120のHCDR2のアミノ酸配列
配列番号15-人工配列の説明:CSPG5120のHCDR3のアミノ酸配列
配列番号16-人工配列の説明:シグナル配列を除いたCSPG5120のVHをコードする塩基配列
配列番号17-人工配列の説明:シグナル配列を除いたCSPG5120のVLのアミノ酸配列
配列番号18-人工配列の説明:CSPG5120のLCDR1のアミノ酸配列
配列番号19-人工配列の説明:CSPG5120のLCDR2のアミノ酸配列
配列番号20-人工配列の説明:CSPG5120のLCDR3のアミノ酸配列
配列番号21-人工配列の説明:シグナル配列を除いたCSPG5168のVHをコードする塩基配列
配列番号22-人工配列の説明:シグナル配列を除いたCSPG5168のVHのアミノ酸配列
配列番号23-人工配列の説明:CSPG5168のHCDR1のアミノ酸配列
配列番号24-人工配列の説明:CSPG5168のHCDR2のアミノ酸配列
配列番号25-人工配列の説明:CSPG5168のHCDR3のアミノ酸配列
配列番号26-人工配列の説明:シグナル配列を除いたCSPG5168のVLをコードする塩基配列
配列番号27-人工配列の説明:シグナル配列を除いたCSPG5168のVLのアミノ酸配列
配列番号28-人工配列の説明:CSPG5168のLCDR1のアミノ酸配列
配列番号29-人工配列の説明:CSPG5168のLCDR2のアミノ酸配列
配列番号30-人工配列の説明:CSPG5168のLCDR3のアミノ酸配列
配列番号31-人工配列の説明:シグナル配列を除いたCSPG5201のVHをコードする塩基配列
配列番号32-人工配列の説明:シグナル配列を除いたCSPG5201のVHのアミノ酸配列
配列番号33-人工配列の説明:CSPG5201のHCDR1のアミノ酸配列
配列番号34-人工配列の説明:CSPG5201のHCDR2のアミノ酸配列
配列番号35-人工配列の説明:CSPG5201のHCDR3のアミノ酸配列
配列番号36-人工配列の説明:シグナル配列を除いたCSPG5201のVLをコードする塩基配列
配列番号37-人工配列の説明:シグナル配列を除いたCSPG5201のVLのアミノ酸配列
配列番号38-人工配列の説明:CSPG5201のLCDR1のアミノ酸配列
配列番号39-人工配列の説明:CSPG5201のLCDR2のアミノ酸配列
配列番号40-人工配列の説明:CSPG5201のLCDR3のアミノ酸配列
配列番号41-人工配列の説明:シグナル配列を除いたCSPG5202のVHをコードする塩基配列
配列番号42-人工配列の説明:シグナル配列を除いたCSPG5202のVHのアミノ酸配列
配列番号43-人工配列の説明:CSPG5202のHCDR1のアミノ酸配列
配列番号44-人工配列の説明:CSPG5202のHCDR2のアミノ酸配列
配列番号45-人工配列の説明:CSPG5202のHCDR3のアミノ酸配列
配列番号46-人工配列の説明:シグナル配列を除いたCSPG5202のVLをコードする塩基配列
配列番号47-人工配列の説明:シグナル配列を除いたCSPG5202のVLのアミノ酸配列
配列番号48-人工配列の説明:CSPG5202のLCDR1のアミノ酸配列
配列番号49-人工配列の説明:CSPG5202のLCDR2のアミノ酸配列
配列番号50-人工配列の説明:CSPG5202のLCDR3のアミノ酸配列
配列番号51-人工配列の説明:シグナル配列を除いたCSPG5205のVHをコードする塩基配列
配列番号52-人工配列の説明:シグナル配列を除いたCSPG5205のVHのアミノ酸配列
配列番号53-人工配列の説明:CSPG5205のHCDR1のアミノ酸配列
配列番号54-人工配列の説明:CSPG5205のHCDR2のアミノ酸配列
配列番号55-人工配列の説明:CSPG5205のHCDR3のアミノ酸配列
配列番号56-人工配列の説明:シグナル配列を除いたCSPG5205のVLをコードする塩基配列
配列番号57-人工配列の説明:シグナル配列を除いたCSPG5205のVLのアミノ酸配列
配列番号58-人工配列の説明:CSPG5205のLCDR1のアミノ酸配列
配列番号59-人工配列の説明:CSPG5205のLCDR2のアミノ酸配列
配列番号60-人工配列の説明:CSPG5205のLCDR3のアミノ酸配列
配列番号61-人工配列の説明:シグナル配列を除いたCSPG5206のVHをコードする塩基配列
配列番号62-人工配列の説明:シグナル配列を除いたCSPG5206のVHのアミノ酸配列
配列番号63-人工配列の説明:CSPG5206のHCDR1のアミノ酸配列
配列番号64-人工配列の説明:CSPG5206のHCDR2のアミノ酸配列
配列番号65-人工配列の説明:CSPG5206のHCDR3のアミノ酸配列
配列番号66-人工配列の説明:シグナル配列を除いたCSPG5206のVLをコードする塩基配列
配列番号67-人工配列の説明:シグナル配列を除いたCSPG5206のVLのアミノ酸配列
配列番号68-人工配列の説明:CSPG5206のLCDR1のアミノ酸配列
配列番号69-人工配列の説明:CSPG5206のLCDR2のアミノ酸配列
配列番号70-人工配列の説明:CSPG5206のLCDR3のアミノ酸配列
配列番号71-人工配列の説明:シグナル配列を除いたCSPG5207のVHをコードする塩基配列
配列番号72-人工配列の説明:シグナル配列を除いたCSPG5207のVHのアミノ酸配列
配列番号73-人工配列の説明:CSPG5207のHCDR1のアミノ酸配列
配列番号74-人工配列の説明:CSPG5207のHCDR2のアミノ酸配列
配列番号75-人工配列の説明:CSPG5207のHCDR3のアミノ酸配列
配列番号76-人工配列の説明:シグナル配列を除いたCSPG5207のVLをコードする塩基配列
配列番号77-人工配列の説明:シグナル配列を除いたCSPG5207のVLのアミノ酸配列
配列番号78-人工配列の説明:CSPG5207のLCDR1のアミノ酸配列
配列番号79-人工配列の説明:CSPG5207のLCDR2のアミノ酸配列
配列番号80-人工配列の説明:CSPG5207のLCDR3のアミノ酸配列
配列番号81-人工配列の説明:シグナル配列を除いたCSPG5208のVHをコードする塩基配列
配列番号82-人工配列の説明:シグナル配列を除いたCSPG5208のVHのアミノ酸配列
配列番号83-人工配列の説明:CSPG5208のHCDR1のアミノ酸配列
配列番号84-人工配列の説明:CSPG5208のHCDR2のアミノ酸配列
配列番号85-人工配列の説明:CSPG5208のHCDR3のアミノ酸配列
配列番号86-人工配列の説明:シグナル配列を除いたCSPG5208のVLをコードする塩基配列
配列番号87-人工配列の説明:シグナル配列を除いたCSPG5208のVLのアミノ酸配列
配列番号88-人工配列の説明:CSPG5208のLCDR1のアミノ酸配列
配列番号89-人工配列の説明:CSPG5208のLCDR2のアミノ酸配列
配列番号90-人工配列の説明:CSPG5208のLCDR3のアミノ酸配列
配列番号91-人工配列の説明:シグナル配列を除いたCSPG5214のVHをコードする塩基配列
配列番号92-人工配列の説明:シグナル配列を除いたCSPG5214のVHのアミノ酸配列
配列番号93-人工配列の説明:CSPG5214のHCDR1のアミノ酸配列
配列番号94-人工配列の説明:CSPG5214のHCDR2のアミノ酸配列
配列番号95-人工配列の説明:CSPG5214のHCDR3のアミノ酸配列
配列番号96-人工配列の説明:シグナル配列を除いたCSPG5214のVLをコードする塩基配列
配列番号97-人工配列の説明:シグナル配列を除いたCSPG5214のVLのアミノ酸配列
配列番号98-人工配列の説明:CSPG5214のLCDR1のアミノ酸配列
配列番号99-人工配列の説明:CSPG5214のLCDR2のアミノ酸配列
配列番号100-人工配列の説明:CSPG5214のLCDR3のアミノ酸配列
配列番号101-人工配列の説明:シグナル配列を除いたCSPG5219のVHをコードする塩基配列
配列番号102-人工配列の説明:シグナル配列を除いたCSPG5219のVHのアミノ酸配列
配列番号103-人工配列の説明:CSPG5219のHCDR1のアミノ酸配列
配列番号104-人工配列の説明:CSPG5219のHCDR2のアミノ酸配列
配列番号105-人工配列の説明:CSPG5219のHCDR3のアミノ酸配列
配列番号106-人工配列の説明:シグナル配列を除いたCSPG5219のVLをコードする塩基配列
配列番号107-人工配列の説明:シグナル配列を除いたCSPG5219のVLのアミノ酸配列
配列番号108-人工配列の説明:CSPG5219のLCDR1のアミノ酸配列
配列番号109-人工配列の説明:CSPG5219のLCDR2のアミノ酸配列
配列番号110-人工配列の説明:CSPG5219のLCDR3のアミノ酸配列
配列番号111-人工配列の説明:シグナル配列を除いたCSPG5222のVHをコードする塩基配列
配列番号112-人工配列の説明:シグナル配列を除いたCSPG5222のVHのアミノ酸配列
配列番号113-人工配列の説明:CSPG5222のHCDR1のアミノ酸配列
配列番号114-人工配列の説明:CSPG5222のHCDR2のアミノ酸配列
配列番号115-人工配列の説明:CSPG5222のHCDR3のアミノ酸配列
配列番号116-人工配列の説明:シグナル配列を除いたCSPG5222のVLをコードする塩基配列
配列番号117-人工配列の説明:シグナル配列を除いたCSPG5222のVLのアミノ酸配列
配列番号118-人工配列の説明:CSPG5222のLCDR1のアミノ酸配列
配列番号119-人工配列の説明:CSPG5222のLCDR2のアミノ酸配列
配列番号120-人工配列の説明:CSPG5222のLCDR3のアミノ酸配列
配列番号121-人工配列の説明:シグナル配列を除いたCSPG5227のVHをコードする塩基配列
配列番号122-人工配列の説明:シグナル配列を除いたCSPG5227のVHのアミノ酸配列
配列番号123-人工配列の説明:CSPG5227のHCDR1のアミノ酸配列
配列番号124-人工配列の説明:CSPG5227のHCDR2のアミノ酸配列
配列番号125-人工配列の説明:CSPG5227のHCDR3のアミノ酸配列
配列番号126-人工配列の説明:シグナル配列を除いたCSPG5227のVLをコードする塩基配列
配列番号127-人工配列の説明:シグナル配列を除いたCSPG5227のVLのアミノ酸配列
配列番号128-人工配列の説明:CSPG5227のLCDR1のアミノ酸配列
配列番号129-人工配列の説明:CSPG5227のLCDR2のアミノ酸配列
配列番号130-人工配列の説明:CSPG5227のLCDR3のアミノ酸配列
配列番号131-人工配列の説明:シグナル配列を除いたCSPG5230のVHをコードする塩基配列
配列番号132-人工配列の説明:シグナル配列を除いたCSPG5230のVHのアミノ酸配列
配列番号133-人工配列の説明:CSPG5230のHCDR1のアミノ酸配列
配列番号134-人工配列の説明:CSPG5230のHCDR2のアミノ酸配列
配列番号135-人工配列の説明:CSPG5230のHCDR3のアミノ酸配列
配列番号136-人工配列の説明:シグナル配列を除いたCSPG5230のVLをコードする塩基配列
配列番号137-人工配列の説明:シグナル配列を除いたCSPG5230のVLのアミノ酸配列
配列番号138-人工配列の説明:CSPG5230のLCDR1のアミノ酸配列
配列番号139-人工配列の説明:CSPG5230のLCDR2のアミノ酸配列
配列番号140-人工配列の説明:CSPG5230のLCDR3のアミノ酸配列
配列番号141-人工配列の説明:シグナル配列を除いたCSPG5234のVHをコードする塩基配列
配列番号142-人工配列の説明:シグナル配列を除いたCSPG5234のVHのアミノ酸配列
配列番号143-人工配列の説明:CSPG5234のHCDR1のアミノ酸配列
配列番号144-人工配列の説明:CSPG5234のHCDR2のアミノ酸配列
配列番号145-人工配列の説明:CSPG5234のHCDR3のアミノ酸配列
配列番号146-人工配列の説明:シグナル配列を除いたCSPG5234のVLをコードする塩基配列
配列番号147-人工配列の説明:シグナル配列を除いたCSPG5234のVLのアミノ酸配列
配列番号148-人工配列の説明:CSPG5234のLCDR1のアミノ酸配列
配列番号149-人工配列の説明:CSPG5234のLCDR2のアミノ酸配列
配列番号150-人工配列の説明:CSPG5234のLCDR3のアミノ酸配列
配列番号151-人工配列の説明:pCI_CSPG5202-hKG4PE(R409K)の軽鎖(シグナル配列除く)をコードする塩基配列
配列番号152-人工配列の説明:pCI_CSPG5202-hKG4PE(R409K)の軽鎖(シグナル配列除く)のアミノ酸配列
配列番号153-人工配列の説明:pCI_CSPG5202-hKG4PE(R409K)の重鎖(シグナル配列除く)をコードする塩基配列
配列番号154-人工配列の説明:pCI_CSPG5202-hKG4PE(R409K)の重鎖(シグナル配列除く)のアミノ酸配列
配列番号155-人工配列の説明:pCI_AVM-hLG4PE(R409K)-CSPG5202scFvの軽鎖(シグナル配列除く)をコードする塩基配列
配列番号156-人工配列の説明:pCI_AVM-hLG4PE(R409K)-CSPG5202scFvの軽鎖(シグナル配列除く)のアミノ酸配列
配列番号157-人工配列の説明:pCI_AVM-hLG4PE(R409K)-CSPG5202scFvの重鎖(シグナル配列除く)をコードする塩基配列
配列番号158-人工配列の説明:pCI_AVM-hLG4PE(R409K)-CSPG5202scFvの重鎖(シグナル配列除く)のアミノ酸配列
配列番号159-人工配列の説明:ヒトCSPG5(シグナル配列を含む)をコードする塩基配列
配列番号160-人工配列の説明:ヒトCSPG5(シグナル配列を含む)のアミノ酸配列
配列番号161-人工配列の説明:マウスCSPG5(シグナル配列を含む)をコードする塩基配列
配列番号162-人工配列の説明:マウスCSPG5(シグナル配列を含む)のアミノ酸配列
配列番号163-人工配列の説明:サルCSPG5(シグナル配列を含む)をコードする塩基配列
配列番号164-人工配列の説明:サルCSPG5(シグナル配列を含む)のアミノ酸配列
配列番号165-human CSPG5-FLAG_Fc(シグナル配列を含む)をコードする塩基配列
配列番号166-人工配列の説明:human CSPG5-FLAG_Fc(シグナル配列を含む)のアミノ酸配列
配列番号167-人工配列の説明:mouse CSPG5-FLAG_Fc(シグナル配列を含む)をコードする塩基配列
配列番号168-人工配列の説明:mouse CSPG5-FLAG_Fc(シグナル配列を含む)のアミノ酸配列
配列番号169-人工配列の説明:human CSPG5-GST(シグナル配列を含む)をコードする塩基配列
配列番号170-人工配列の説明:human CSPG5-GST(シグナル配列を含む)のアミノ酸配列
配列番号171-人工配列の説明:mouse CSPG5-GST(シグナル配列を含む)をコードする塩基配列
配列番号172-人工配列の説明:mouse CSPG5-GST(シグナル配列を含む)のアミノ酸配列
Claims (23)
- コンドロイチン硫酸プロテオグリカン-5(CSPG5)に結合する抗体または該抗体断片。
- 抗体が脳滞留性を有する請求項1に記載の抗体または該抗体断片。
- 抗体が神経細胞および/またはアストロサイト結合性を有する抗体である請求項2に記載の抗体または該抗体断片。
- 抗体が下記(a)~(s)からなる群より選ばれる1である、請求項1~3のいずれか1項に記載の抗体または該抗体断片;
(a)重鎖可変領域(VH)の相補性決定領域(CDR)1~3のアミノ酸配列が、それぞれ配列番号3、4および5に記載されるアミノ酸配列を含み、かつ軽鎖可変領域(VL)のCDR1~3のアミノ酸配列が、それぞれ配列番号8、9および10に記載されるアミノ酸配列を含む抗体、
(b)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号13、14および15に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号18、19および20に記載されるアミノ酸配列を含む抗体、
(c)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号23、24および25に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号28、29および30に記載されるアミノ酸配列を含む抗体、
(d)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号33、34および35に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号38、39および40に記載されるアミノ酸配列を含む抗体、
(e)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号43、44および45に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号48、49および50に記載されるアミノ酸配列を含む抗体、
(f)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号53、54および55に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号58、59および60に記載されるアミノ酸配列を含む抗体、
(g)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号63、64および65に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号68、69および70に記載されるアミノ酸配列を含む抗体、
(h)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号73、74および75に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号78、79および80に記載されるアミノ酸配列を含む抗体、
(i)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号83、84および85に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号88、89および90に記載されるアミノ酸配列を含む抗体、
(j)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号93、94および95に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号98、99および100に記載されるアミノ酸配列を含む抗体、
(k)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号103、104および105に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号108、109および110に記載されるアミノ酸配列を含む抗体、
(l)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号113、114および115に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号118、119および120に記載されるアミノ酸配列を含む抗体、
(m)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号123、124および125に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号128、129および130に記載されるアミノ酸配列を含む抗体、
(n)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号133、134および135に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号138、139および140に記載されるアミノ酸配列を含む抗体、
(o)VHのCDR1~3のアミノ酸配列が、それぞれ配列番号143、144および145に記載されるアミノ酸配列を含み、かつVLのCDR1~3のアミノ酸配列が、それぞれ配列番号148、149および150に記載されるアミノ酸配列を含む抗体、
(p)前記(a)~(o)に記載の少なくとも1つの抗体と、CSPG5への結合について競合する抗体、
(q)前記(a)~(o)に記載のいずれか1つの抗体が結合するエピトープを含むエピトープに結合する抗体、
(r)前記(a)~(o)に記載のいずれか1つの抗体が結合するエピトープと同じエピトープに結合する抗体、および
(s)前記(a)~(o)に記載のいずれか1つの抗体のアミノ酸配列と85%以上の相同性を有するアミノ酸配列を含む抗体。 - 抗体が下記(A)~(P)からなる群より選ばれる1である、請求項1~4のいずれか1項に記載の抗体または該抗体断片;
(A)VHのアミノ酸配列が配列番号2に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号7に記載されるアミノ酸配列を含む抗体、
(B)VHのアミノ酸配列が配列番号12に記載されるアミノ酸配列含み、かつVLのアミノ酸配列が配列番号17に記載されるアミノ酸配列を含む抗体、
(C)VHのアミノ酸配列が配列番号22に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号27に記載されるアミノ酸配列を含む抗体、
(D)VHのアミノ酸配列が配列番号32に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号37に記載されるアミノ酸配列を含む抗体、
(E)VHのアミノ酸配列が配列番号42に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号47に記載されるアミノ酸配列を含む抗体、
(F)VHのアミノ酸配列が配列番号52に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号57に記載されるアミノ酸配列を含む抗体、
(G)VHのアミノ酸配列が配列番号62に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号67に記載されるアミノ酸配列を含む抗体、
(H)VHのアミノ酸配列が配列番号72に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号77に記載されるアミノ酸配列を含む抗体、
(I)VHのアミノ酸配列が配列番号82に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号87に記載されるアミノ酸配列を含む抗体、
(J)VHのアミノ酸配列が配列番号92に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号97に記載されるアミノ酸配列を含む抗体、
(K)VHのアミノ酸配列が配列番号102に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号107に記載されるアミノ酸配列を含む抗体、
(L)VHのアミノ酸配列が配列番号112に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号117に記載されるアミノ酸配列を含む抗体、
(M)VHのアミノ酸配列が配列番号122に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号127に記載されるアミノ酸配列を含む抗体、
(N)VHのアミノ酸配列が配列番号132に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号137に記載されるアミノ酸配列を含む抗体、
(O)VHのアミノ酸配列が配列番号142に記載されるアミノ酸配列を含み、かつVLのアミノ酸配列が配列番号147に記載されるアミノ酸配列を含む抗体、および
(P)前記(A)~(O)に記載のいずれか1つの抗体のアミノ酸配列と85%以上の相同性を有するアミノ酸配列を含む抗体。 - 抗体または該抗体断片がバイスペシフィック抗体である、請求項1~5のいずれか1項に記載の抗体または該抗体断片。
- バイスペシフィック抗体がCSPG5および脳に存在する抗原に結合する、請求項6に記載のバイスペシフィック抗体。
- バイスペシフィック抗体がCSPG5に結合する抗原結合部位および脳に存在する抗原に結合する抗原結合部位を含む、請求項6または7に記載のバイスペシフィック抗体。
- 抗体断片がFab、Fab’、F(ab’)2、一本鎖抗体(scFv)、二量体化V領域(diabody)、ジスルフィド安定化V領域(dsFv)、重鎖抗体の重鎖可変領域(VHH)およびCDRを含むペプチドからなる群より選ばれる1である、請求項1~8のいずれか1項に記載の抗体断片。
- 抗体が遺伝子組換え抗体である、請求項1~9のいずれか1項に記載の抗体および該抗体断片。
- 抗体がマウス抗体、ラット抗体、ラビット抗体、アルパカ抗体、ラクダ抗体、ラマ抗体、キメラ抗体、ヒト化抗体およびヒト抗体からなる群より選ばれる1である、請求項1~10のいずれか1項に記載の抗体および該抗体断片。
- 請求項1~11のいずれか1項に記載のCSPG5に結合する抗体または該抗体断片に、下記(i)~(iii)からなる群より選ばれる少なくとも1つを結合させた融合抗体または該融合抗体断片;
(i)親水性高分子、
(ii)両親媒性高分子、および
(iii)機能性分子。 - 請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片を産生するハイブリドーマ。
- 請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片をコードする塩基配列を含む核酸。
- 請求項14に記載の核酸を含むベクターを含む形質転換細胞。
- 請求項13に記載のハイブリドーマまたは請求項15に記載の形質転換細胞を培養し、培養液から請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片を採取することを含む、請求項1~12のいずれか1項に記載の抗体、融合抗体若しくは該融合抗体断片該抗体断片の製造方法。
- 請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片を含む、組成物。
- 脳に存在する抗原の検出または測定用の組成物である、請求項17に記載の組成物。
- 脳疾患の診断または治療するための組成物である、請求項17に記載の組成物。
- 請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片、または請求項17に記載の組成物を用いて、脳に存在する抗原を検出または測定する方法。
- 請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片、または請求項17に記載の組成物を用いて、脳疾患を診断または治療する方法。
- 請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは融合抗体断片、または請求項17に記載の組成物を用いて、抗体、該抗体断片、融合抗体若しくは該融合抗体断片の脳滞留性を向上させる方法。
- 請求項1~12のいずれか1項に記載の抗体、該抗体断片、融合抗体若しくは該融合抗体断片、または請求項17に記載の組成物を用いて、脳内の抗体量、該抗体断片量、融合抗体量若しくは該融合抗体断片量を増加させる方法。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP19826585.2A EP3816291A4 (en) | 2018-06-26 | 2019-06-26 | CHONDROITIN SULFATE PROTEOGLYCAN-5 BINDING ANTIBODIES |
| US17/255,692 US11965035B2 (en) | 2018-06-26 | 2019-06-26 | Antibody binding to chondroitin sulfate proteoglycan 5 |
| JP2020527592A JP7397444B2 (ja) | 2018-06-26 | 2019-06-26 | コンドロイチン硫酸プロテオグリカン-5に結合する抗体 |
| KR1020207037926A KR20210025023A (ko) | 2018-06-26 | 2019-06-26 | 콘드로이틴 황산 프로테오글리칸-5에 결합하는 항체 |
| CA3104997A CA3104997A1 (en) | 2018-06-26 | 2019-06-26 | Antibody binding to chondroitin sulfate proteoglycan 5 |
| CN201980043858.7A CN112424357A (zh) | 2018-06-26 | 2019-06-26 | 与硫酸软骨素蛋白聚糖5结合的抗体 |
| AU2019295277A AU2019295277A1 (en) | 2018-06-26 | 2019-06-26 | Antibody binding to chondroitin sulfate proteoglycan-5 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018120476 | 2018-06-26 | ||
| JP2018-120476 | 2018-06-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020004490A1 true WO2020004490A1 (ja) | 2020-01-02 |
Family
ID=68986614
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/025450 WO2020004490A1 (ja) | 2018-06-26 | 2019-06-26 | コンドロイチン硫酸プロテオグリカン-5に結合する抗体 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US11965035B2 (ja) |
| EP (1) | EP3816291A4 (ja) |
| JP (1) | JP7397444B2 (ja) |
| KR (1) | KR20210025023A (ja) |
| CN (1) | CN112424357A (ja) |
| AU (1) | AU2019295277A1 (ja) |
| CA (1) | CA3104997A1 (ja) |
| TW (1) | TW202012445A (ja) |
| WO (1) | WO2020004490A1 (ja) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109536506B (zh) * | 2018-12-10 | 2022-03-22 | 潍坊科技学院 | 一种调控日本沼虾生长发育的类胰岛素受体基因及其应用 |
| CN114957476A (zh) * | 2021-02-23 | 2022-08-30 | 复旦大学 | 一种半胱氨酸工程化的结合人5t4的全人源纳米抗体 |
| CN113929785B (zh) * | 2021-09-10 | 2023-05-23 | 钦元再生医学(珠海)有限公司 | 一种可自分泌pd1-trem2双特异性抗体的嵌合抗原受体免疫细胞及其应用 |
| CN114478802B (zh) * | 2022-01-28 | 2023-05-26 | 郑州大学 | 一种嵌合抗原受体及其应用 |
| WO2024029586A1 (en) * | 2022-08-04 | 2024-02-08 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecules with increased penetration into and retention in brain, and methods for use thereof |
Citations (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US160735A (en) | 1875-03-09 | Improvement in sash-holders | ||
| JPS58110600A (ja) | 1981-12-25 | 1983-07-01 | Kyowa Hakko Kogyo Co Ltd | ヒトβ型インタ−フエロン遺伝子を含む組みかえ体プラスミド |
| JPS60221091A (ja) | 1983-12-21 | 1985-11-05 | Kyowa Hakko Kogyo Co Ltd | 新規プロモ−タ− |
| JPS63299A (ja) | 1986-04-22 | 1988-01-05 | イミユネツクス・コ−ポレ−シヨン | ヒトg−csfタンパク質の発現 |
| US4939094A (en) | 1985-08-28 | 1990-07-03 | Kyowa Hakko Kogyo Co., Ltd. | Fused antigen polypeptide |
| JPH02227075A (ja) | 1988-09-29 | 1990-09-10 | Kyowa Hakko Kogyo Co Ltd | 新規ポリペプチド |
| JPH02257891A (ja) | 1989-03-31 | 1990-10-18 | Kyowa Hakko Kogyo Co Ltd | 組換え動物細胞による蛋白質の製造 |
| JPH0322979A (ja) | 1989-06-19 | 1991-01-31 | Kyowa Hakko Kogyo Co Ltd | 新規プラスミノーゲン活性化因子 |
| JPH05336963A (ja) | 1991-12-17 | 1993-12-21 | Kyowa Hakko Kogyo Co Ltd | 新規α2→3シアリルトランスフェラーゼ |
| WO1994023021A1 (en) | 1993-03-29 | 1994-10-13 | Kyowa Hakko Kogyo Co., Ltd. | α-1,3-FUCOSYLTRANSFERASE |
| WO1997010354A1 (en) | 1995-09-11 | 1997-03-20 | Kyowa Hakko Kogyo Co., Ltd. | ANTIBODY AGAINTS α-CHAIN OF HUMAN INTERLEUKIN 5 RECEPTOR |
| JPH09194502A (ja) * | 1995-11-13 | 1997-07-29 | Seikagaku Kogyo Co Ltd | 新規コンドロイチン硫酸プロテオグリカン、そのコア蛋白質、それをコードするdnaおよびそれに対する抗体 |
| US6001358A (en) | 1995-11-07 | 1999-12-14 | Idec Pharmaceuticals Corporation | Humanized antibodies to human gp39, compositions containing thereof |
| WO2000061739A1 (en) | 1999-04-09 | 2000-10-19 | Kyowa Hakko Kogyo Co., Ltd. | Method for controlling the activity of immunologically functional molecule |
| WO2001077342A1 (en) | 2000-04-11 | 2001-10-18 | Genentech, Inc. | Multivalent antibodies and uses therefor |
| WO2002031140A1 (fr) | 2000-10-06 | 2002-04-18 | Kyowa Hakko Kogyo Co., Ltd. | Cellules produisant des compositions d'anticorps |
| WO2002088186A1 (en) | 2001-04-27 | 2002-11-07 | Kirin Beer Kabushiki Kaisha | Anti-cd40 monoclonal antibody |
| JP2003079374A (ja) * | 2001-09-07 | 2003-03-18 | Seikagaku Kogyo Co Ltd | プロテオグリカンのコアタンパク質のポリペプチド、それをコードするdnaおよびそのプロモーターdna |
| US6737056B1 (en) | 1999-01-15 | 2004-05-18 | Genentech, Inc. | Polypeptide variants with altered effector function |
| WO2005035586A1 (ja) | 2003-10-08 | 2005-04-21 | Kyowa Hakko Kogyo Co., Ltd. | 融合蛋白質組成物 |
| WO2006033386A1 (ja) | 2004-09-22 | 2006-03-30 | Kirin Beer Kabushiki Kaisha | 安定化されたヒトIgG4抗体 |
| WO2006075668A1 (ja) | 2005-01-12 | 2006-07-20 | Kirin Beer Kabushiki Kaisha | 安定化されたヒトIgG2およびIgG3抗体 |
| WO2007024715A2 (en) | 2005-08-19 | 2007-03-01 | Abbott Laboratories | Dual variable domain immunoglobin and uses thereof |
| US20070071675A1 (en) | 2005-08-19 | 2007-03-29 | Chengbin Wu | Dual variable domain immunoglobulin and uses thereof |
| CA2623841A1 (en) | 2005-09-27 | 2007-04-05 | National Research Council Of Canada | Methods of identifying agents capable of tmem30a mediated transmigration across the blood brain barrier (bbb) |
| WO2007048037A2 (en) | 2005-10-21 | 2007-04-26 | Amgen Inc. | METHODS FOR GENERATING MONOVALENT IgG |
| US20070148165A1 (en) | 2005-07-22 | 2007-06-28 | Kyowa Hakko Kogyo Co., Ltd. | Recombinant antibody composition |
| US7297775B2 (en) | 1998-04-02 | 2007-11-20 | Genentech, Inc. | Polypeptide variants |
| US7317091B2 (en) | 2002-03-01 | 2008-01-08 | Xencor, Inc. | Optimized Fc variants |
| WO2009080251A1 (en) | 2007-12-21 | 2009-07-02 | F. Hoffmann-La Roche Ag | Bivalent, bispecific antibodies |
| WO2009125825A1 (ja) | 2008-04-11 | 2009-10-15 | 中外製薬株式会社 | 複数分子の抗原に繰り返し結合する抗原結合分子 |
| WO2009131239A1 (ja) | 2008-04-25 | 2009-10-29 | Kyowa Hakko Kirin Co Ltd | 安定な多価抗体 |
| WO2010143698A1 (ja) | 2009-06-11 | 2010-12-16 | 大学共同利用機関法人情報・システム研究機構 | タンパク質の生産方法 |
| WO2010151792A1 (en) | 2009-06-26 | 2010-12-29 | Regeneron Pharmaceuticals, Inc. | Readily isolated bispecific antibodies with native immunoglobulin format |
| WO2011090754A1 (en) | 2009-12-29 | 2011-07-28 | Emergent Product Development Seattle, Llc | Polypeptide heterodimers and uses thereof |
| WO2011122011A2 (en) | 2010-03-30 | 2011-10-06 | Chugai Seiyaku Kabushiki Kaisha | Antibodies with modified affinity to fcrn that promote antigen clearance |
| WO2012023623A2 (ja) | 2010-08-19 | 2012-02-23 | 日本ケミカルリサーチ株式会社 | ハンター症候群の治療剤 |
| WO2012116927A1 (en) | 2011-02-28 | 2012-09-07 | F. Hoffmann-La Roche Ag | Monovalent antigen binding proteins |
| JP2013165716A (ja) | 2006-03-31 | 2013-08-29 | Chugai Pharmaceut Co Ltd | 抗体の血中動態を制御する方法 |
| WO2014033074A1 (en) | 2012-08-29 | 2014-03-06 | F. Hoffmann-La Roche Ag | Blood brain barrier shuttle |
| WO2014054804A1 (ja) | 2012-10-05 | 2014-04-10 | 協和発酵キリン株式会社 | ヘテロダイマータンパク質組成物 |
| WO2016081643A1 (en) | 2014-11-19 | 2016-05-26 | Genentech, Inc. | Anti-transferrin receptor antibodies and methods of use |
| WO2016081640A1 (en) | 2014-11-19 | 2016-05-26 | Genentech, Inc. | Anti-transferrin receptor / anti-bace1 multispecific antibodies and methods of use |
| WO2016175307A1 (ja) | 2015-04-30 | 2016-11-03 | 東レ株式会社 | 癌の治療及び/又は予防用医薬組成物 |
| JP2018120476A (ja) | 2017-01-26 | 2018-08-02 | 富士ゼロックス株式会社 | 情報処理装置及びプログラム |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2623841A (en) | 1950-11-21 | 1952-12-30 | Schmid Inc Julius | Spermicidal compositions |
| AU2015357054A1 (en) * | 2014-12-04 | 2017-06-29 | Abruzzo Theranostic S.R.L. | Humanized anti-Trop-2 monoclonal antibodies and uses thereof |
| EA201891002A1 (ru) * | 2015-10-19 | 2018-11-30 | Дайэкс Корп. | Иммунологический анализ для определения расщепленного высокомолекулярного кининогена |
-
2019
- 2019-06-26 TW TW108122349A patent/TW202012445A/zh unknown
- 2019-06-26 KR KR1020207037926A patent/KR20210025023A/ko unknown
- 2019-06-26 CN CN201980043858.7A patent/CN112424357A/zh active Pending
- 2019-06-26 WO PCT/JP2019/025450 patent/WO2020004490A1/ja unknown
- 2019-06-26 JP JP2020527592A patent/JP7397444B2/ja active Active
- 2019-06-26 US US17/255,692 patent/US11965035B2/en active Active
- 2019-06-26 AU AU2019295277A patent/AU2019295277A1/en active Pending
- 2019-06-26 CA CA3104997A patent/CA3104997A1/en active Pending
- 2019-06-26 EP EP19826585.2A patent/EP3816291A4/en active Pending
Patent Citations (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US160735A (en) | 1875-03-09 | Improvement in sash-holders | ||
| JPS58110600A (ja) | 1981-12-25 | 1983-07-01 | Kyowa Hakko Kogyo Co Ltd | ヒトβ型インタ−フエロン遺伝子を含む組みかえ体プラスミド |
| US4686191A (en) | 1981-12-25 | 1987-08-11 | Hakko Kogyo Co., Ltd. Kyowa | Recombinant plasmid containing human interferon-beta gene |
| JPS60221091A (ja) | 1983-12-21 | 1985-11-05 | Kyowa Hakko Kogyo Co Ltd | 新規プロモ−タ− |
| US4939094A (en) | 1985-08-28 | 1990-07-03 | Kyowa Hakko Kogyo Co., Ltd. | Fused antigen polypeptide |
| JPS63299A (ja) | 1986-04-22 | 1988-01-05 | イミユネツクス・コ−ポレ−シヨン | ヒトg−csfタンパク質の発現 |
| JPH02227075A (ja) | 1988-09-29 | 1990-09-10 | Kyowa Hakko Kogyo Co Ltd | 新規ポリペプチド |
| JPH02257891A (ja) | 1989-03-31 | 1990-10-18 | Kyowa Hakko Kogyo Co Ltd | 組換え動物細胞による蛋白質の製造 |
| JPH0322979A (ja) | 1989-06-19 | 1991-01-31 | Kyowa Hakko Kogyo Co Ltd | 新規プラスミノーゲン活性化因子 |
| JPH05336963A (ja) | 1991-12-17 | 1993-12-21 | Kyowa Hakko Kogyo Co Ltd | 新規α2→3シアリルトランスフェラーゼ |
| WO1994023021A1 (en) | 1993-03-29 | 1994-10-13 | Kyowa Hakko Kogyo Co., Ltd. | α-1,3-FUCOSYLTRANSFERASE |
| WO1997010354A1 (en) | 1995-09-11 | 1997-03-20 | Kyowa Hakko Kogyo Co., Ltd. | ANTIBODY AGAINTS α-CHAIN OF HUMAN INTERLEUKIN 5 RECEPTOR |
| US6001358A (en) | 1995-11-07 | 1999-12-14 | Idec Pharmaceuticals Corporation | Humanized antibodies to human gp39, compositions containing thereof |
| JPH09194502A (ja) * | 1995-11-13 | 1997-07-29 | Seikagaku Kogyo Co Ltd | 新規コンドロイチン硫酸プロテオグリカン、そのコア蛋白質、それをコードするdnaおよびそれに対する抗体 |
| US7297775B2 (en) | 1998-04-02 | 2007-11-20 | Genentech, Inc. | Polypeptide variants |
| US6737056B1 (en) | 1999-01-15 | 2004-05-18 | Genentech, Inc. | Polypeptide variants with altered effector function |
| WO2000061739A1 (en) | 1999-04-09 | 2000-10-19 | Kyowa Hakko Kogyo Co., Ltd. | Method for controlling the activity of immunologically functional molecule |
| WO2001077342A1 (en) | 2000-04-11 | 2001-10-18 | Genentech, Inc. | Multivalent antibodies and uses therefor |
| WO2002031140A1 (fr) | 2000-10-06 | 2002-04-18 | Kyowa Hakko Kogyo Co., Ltd. | Cellules produisant des compositions d'anticorps |
| WO2002088186A1 (en) | 2001-04-27 | 2002-11-07 | Kirin Beer Kabushiki Kaisha | Anti-cd40 monoclonal antibody |
| JP2003079374A (ja) * | 2001-09-07 | 2003-03-18 | Seikagaku Kogyo Co Ltd | プロテオグリカンのコアタンパク質のポリペプチド、それをコードするdnaおよびそのプロモーターdna |
| US7317091B2 (en) | 2002-03-01 | 2008-01-08 | Xencor, Inc. | Optimized Fc variants |
| WO2005035586A1 (ja) | 2003-10-08 | 2005-04-21 | Kyowa Hakko Kogyo Co., Ltd. | 融合蛋白質組成物 |
| WO2006033386A1 (ja) | 2004-09-22 | 2006-03-30 | Kirin Beer Kabushiki Kaisha | 安定化されたヒトIgG4抗体 |
| WO2006075668A1 (ja) | 2005-01-12 | 2006-07-20 | Kirin Beer Kabushiki Kaisha | 安定化されたヒトIgG2およびIgG3抗体 |
| US20070148165A1 (en) | 2005-07-22 | 2007-06-28 | Kyowa Hakko Kogyo Co., Ltd. | Recombinant antibody composition |
| US20070071675A1 (en) | 2005-08-19 | 2007-03-29 | Chengbin Wu | Dual variable domain immunoglobulin and uses thereof |
| WO2007024715A2 (en) | 2005-08-19 | 2007-03-01 | Abbott Laboratories | Dual variable domain immunoglobin and uses thereof |
| CA2623841A1 (en) | 2005-09-27 | 2007-04-05 | National Research Council Of Canada | Methods of identifying agents capable of tmem30a mediated transmigration across the blood brain barrier (bbb) |
| WO2007048037A2 (en) | 2005-10-21 | 2007-04-26 | Amgen Inc. | METHODS FOR GENERATING MONOVALENT IgG |
| JP2013165716A (ja) | 2006-03-31 | 2013-08-29 | Chugai Pharmaceut Co Ltd | 抗体の血中動態を制御する方法 |
| WO2009080251A1 (en) | 2007-12-21 | 2009-07-02 | F. Hoffmann-La Roche Ag | Bivalent, bispecific antibodies |
| WO2009125825A1 (ja) | 2008-04-11 | 2009-10-15 | 中外製薬株式会社 | 複数分子の抗原に繰り返し結合する抗原結合分子 |
| JP2012021004A (ja) | 2008-04-11 | 2012-02-02 | Chugai Pharmaceut Co Ltd | 複数分子の抗原に繰り返し結合する抗原結合分子 |
| WO2009131239A1 (ja) | 2008-04-25 | 2009-10-29 | Kyowa Hakko Kirin Co Ltd | 安定な多価抗体 |
| WO2010143698A1 (ja) | 2009-06-11 | 2010-12-16 | 大学共同利用機関法人情報・システム研究機構 | タンパク質の生産方法 |
| WO2010151792A1 (en) | 2009-06-26 | 2010-12-29 | Regeneron Pharmaceuticals, Inc. | Readily isolated bispecific antibodies with native immunoglobulin format |
| WO2011090754A1 (en) | 2009-12-29 | 2011-07-28 | Emergent Product Development Seattle, Llc | Polypeptide heterodimers and uses thereof |
| WO2011122011A2 (en) | 2010-03-30 | 2011-10-06 | Chugai Seiyaku Kabushiki Kaisha | Antibodies with modified affinity to fcrn that promote antigen clearance |
| WO2012023623A2 (ja) | 2010-08-19 | 2012-02-23 | 日本ケミカルリサーチ株式会社 | ハンター症候群の治療剤 |
| WO2012116927A1 (en) | 2011-02-28 | 2012-09-07 | F. Hoffmann-La Roche Ag | Monovalent antigen binding proteins |
| WO2014033074A1 (en) | 2012-08-29 | 2014-03-06 | F. Hoffmann-La Roche Ag | Blood brain barrier shuttle |
| WO2014054804A1 (ja) | 2012-10-05 | 2014-04-10 | 協和発酵キリン株式会社 | ヘテロダイマータンパク質組成物 |
| WO2016081643A1 (en) | 2014-11-19 | 2016-05-26 | Genentech, Inc. | Anti-transferrin receptor antibodies and methods of use |
| WO2016081640A1 (en) | 2014-11-19 | 2016-05-26 | Genentech, Inc. | Anti-transferrin receptor / anti-bace1 multispecific antibodies and methods of use |
| WO2016175307A1 (ja) | 2015-04-30 | 2016-11-03 | 東レ株式会社 | 癌の治療及び/又は予防用医薬組成物 |
| JP2018120476A (ja) | 2017-01-26 | 2018-08-02 | 富士ゼロックス株式会社 | 情報処理装置及びプログラム |
Non-Patent Citations (123)
| Title |
|---|
| "Antibodies - A Laboratory Manual", 1988, COLD SPRING HARBOR LABORATORY |
| "Bioluminescence and Chemiluminescence", vol. 42, 1998, HIROKAWA-SHOTEN LTD., article "Clinical Test" |
| "Current protocols in Immunology", 1993, JOHN WILEY & SONS, INC., article "Immunologic studies in humans" |
| "DNA Cloning 1: Core Techniques, A Practical Approach", 1995, OXFORD UNIVERSITY |
| "Fluorescent Antibody Method", 1983, SOFT SCIENCE, INC. |
| "Methods in Nucleic Acids Res.", 1991, CRC PRESS, pages: 283 |
| "Monoclonal Antibodies - Principles and Practice", 1996, ACADEMIC PRESS |
| "Monoclonal Antibody Experimental Manual", 1987, KODANSHA SCIENTIFIC BOOKS |
| "NCBI", Database accession no. XM 015131074.1 |
| "Package insert of intravenous infusion 6 mg of Elaprase", July 2016 |
| ABULROB A. ET AL., J. NEUYROCHEM., vol. 95, no. 4, 2005, pages 1201 - 1214 |
| AGRIC. BIOL. CHEM., vol. 53, 1989, pages 277 |
| AGRICULTURAL BIOLOGICAL CHEMISTRY, vol. 48, 1984, pages 669 |
| AONO S. ET AL., J. BIOL. CHEM., vol. 275, 2000, pages 337 - 342 |
| AONO S. ET AL., J. NEUROSCI. RES., vol. 83, 2006, pages 110 - 118 |
| AONO SACHIKO ,TOKITA YOSHIHITO, YASUDA YOKO, HIRANO KANAKO, YAMAUCHI SHINOBU, SHUO TAKUYA, MATSUI FUMIKO, KEINO HIROOMI, KASHIWAI : "Expression and identification of a new splice variant of neuroglycan C, a transmembrane chondroitin sulfate proteoglycan, in the human brain", JOURNAL OF NEUROSCIENCE RESEARCH, vol. 83, no. 1, 1 January 2006 (2006-01-01), pages 110 - 118, XP055763205, ISSN: 0360-4012, DOI: 10.1002/jnr.20698 * |
| BECK ET AL., ANALYTICAL CHEMISTRY, vol. 85, 2013, pages 715 - 736 |
| BIO/TECHNOLOGY, vol. 9, 1991, pages 266 |
| BIOCHEM. BIOPHYS. RES. COMMUN., vol. 149, 1987, pages 960 |
| BOADO RJ. ET AL., BIOCONJUGATE CHEM., vol. 1, 2012, pages 97 - 104 |
| BOADO RJ. ET AL., DRUG METAB. DISPOS., vol. 37, no. 12, 2009, pages 2299 - 2304 |
| BOADO RJ. ET AL., J. PHARMACOL. EXP. THER., vol. 333, no. 3, 2010, pages 961 - 969 |
| BROOKS, D. A. ET AL., TRENDS MOL. MED., vol. 9, 2003, pages 450 - 453 |
| CANCER IMMUNOL. IMMUNOTHER., vol. 36, 1993, pages 373 |
| CELL, vol. 33, 1983, pages 717 |
| CELL, vol. 41, 1985, pages 4431 |
| CELL, vol. 6, 1975, pages 121 |
| CHROMOSOMA, vol. 41, 1973, pages 129 |
| CLINICAL ONCOLOGY, JAPANESE JOURNAL OF CANCER AND CHEMOTHERAPY, 1996 |
| COUCH JA. ET AL., SCIENCE TRANSLATIONAL MEDICINE, vol. 5, 2013, pages 183ra57 |
| CURRENT TOPICS IN MICROBIOLOGY AND IMMUNOLOGY, vol. 18, 1978, pages 1 |
| CYTOTECHNOL., vol. 13, 1993, pages 79 |
| CYTOTECHNOL., vol. 4, 1990, pages 173 |
| CYTOTECHNOLOGY, vol. 3, 1990, pages 133 |
| DNA CLONING: A PRACTICAL APPROACH, vol. 1, 1985, pages 49 |
| EUROPEAN J. IMMUNOLOGY, vol. 6, 1976, pages 511 |
| FARRINGTON GK. ET AL., FASEB J., vol. 28, 2014, pages 4764 - 4778 |
| GARGA. ET AL., AAPSJ., vol. 11, 2009, pages 553 - 557 |
| GENE, vol. 17, 1982, pages 107 |
| GENE, vol. 27, 1984, pages 223 |
| GENE, vol. 38, 1985, pages 275 |
| GENES DEVELOP., vol. 4, 1990, pages 1288 |
| GENETICS, vol. 39, 1954, pages 440 |
| GENETICS, vol. 55, 1968, pages 513 |
| GENOME RES., vol. 7, 1997, pages 649, Retrieved from the Internet <URL:http://www.ncbi.nlm.nih.gov/Education/BLASTinfo/information3.html> |
| HYBRIDOMA, vol. 17, 1998, pages 559 |
| INATANI M. ET AL., INVEST. OPHTHALMOL. VIS. SCI., vol. 41, 2000, pages 4338 - 46 |
| J. BACTERIOL., vol. 172, 1990, pages 2392 |
| J. BIOCHEM., vol. 101, 1987, pages 1307 |
| J. BIOCHEMISTRY, vol. 101, 1987, pages 1307 |
| J. IMMUNOL. METHODS, vol. 167, 1994, pages 271 |
| J. IMMUNOLOGY, vol. 123, 1979, pages 1548 |
| J. MOL. BIOL., vol. 112, 1977, pages 535 |
| J. MOL. BIOL., vol. 166, 1966, pages 118 |
| J. MOL. BIOL., vol. 215, 1990, pages 403 |
| JOURNAL OF EXPERIMENTAL MEDICINE, vol. 108, 1958, pages 945 |
| JUN Y. ET AL., MACROMOL. BIOSCI., vol. 12, 2012, pages 1209 - 1219 |
| JUTTNER R. ET AL., EUR. J. NEUROSCI., vol. 38, 2013, pages 3270 - 3280 |
| KAJ B. ET AL., ARCH. NEUROL., vol. 69, no. 8, 2012, pages 1002 - 1010 |
| KINUGASA Y. ET AL., BIOCHEM. BIOPH. RES. CO., vol. 321, 2004, pages 1045 - 1049 |
| KOLB, ANGEW CHEM INT ED ENGL, vol. 40, 2001, pages 2004 - 21 |
| KONTERMANN ET AL., MABS, vol. 4, 2012, pages 182 - 197 |
| KYLA RR.RICHARD CC., BIOTECHNOL ADV, vol. pii: S0734-9750, no. 16, 2016 |
| LABRIJN ET AL., PNAS, vol. 110, no. 13, 2013, pages 5145 - 5150 |
| LOWE ET AL., PROC. NATL. ACAD. SCI., USA, vol. 86, 1989, pages 8227 |
| MARK A. ET AL., NATURE, vol. 532, 2016, pages 195 - 200 |
| MASARU INATANI; HIDENOBU TANIHARA; ATSUHIKO OOHIRA; YASUMASA OTORI; AKIHIRO NISHIDA; MEGUMI HONJO; NORIAKI KIDO; YOSHIHITO HONDA: "Neuroglycan C, a neural tissue-specific transmembrane chondroitin sulfate proteoglycan, in retinal neural network formation", INVESTIGATIVE OPHTHALMOLOGY & VISUAL SCIENCE, vol. 41, no. 13, 2000, pages 4338 - 4346, XP055763211, ISSN: 0146-0404 * |
| METHODS IN CELL SCIENCE, vol. 18, 1996, pages 115 |
| METHODS IN ENZYMOL., vol. 154, 1987, pages 3 |
| MIYAZAKI ET AL., J. BIOCHEM., vol. 158, no. 3, 2015, pages 205 - 215 |
| MOL. CELL. BIOL., vol. 3, 1983, pages 280 |
| MOLECULAR & GENERAL GENETICS, vol. 168, 1979, pages 111 |
| MOLECULAR CELL GENETICS, pages 883 - 900 |
| MUENZER J. ET AL., GENET MED, vol. 8, no. 8, 2006, pages 465 - 473 |
| NATURE, vol. 227, 1970, pages 680 |
| NATURE, vol. 256, 1975, pages 495 |
| NATURE, vol. 276, 1978, pages 269 |
| NIEWOEHNER J. ET AL., NEURON, vol. 81, 2014, pages 49 - 60 |
| NOBUAKI TAKAHASHI, RINPEI NIWA , RYOUSUKE NAKANO , KAZUMA TOMIZUKA: "Strategy for technology development of antibody therapeutics", FOLIA PHARMACOLOGICA JAPONICA, vol. 147, no. 4, 1 January 2016 (2016-01-01), pages 235 - 240, XP055763221, ISSN: 0015-5691, DOI: 10.1254/fpj.147.235 * |
| NUCLEIC ACIDS RES., vol. 25, 1997, pages 3389 |
| NUCLEIC ACIDS RESEARCH, vol. 17, 1989, pages 9494 |
| PACKAGE INSERT OF INTRAVENOUS INFUSION 2.9 MG OF ALDURAZYME, July 2016 (2016-07-01) |
| PARDRIDGE WM., BIOCONJUGATE CHEM., vol. 19, 2008, pages 1327 - 1338 |
| PARDRIDGE WM.BOADO RJ., METHODS IN ENZYMOLOGY, vol. 503, pages 269 - 292 |
| PAULSON ET AL., J. BIOL. CHEM., vol. 264, 1989, pages 17619 |
| PHILIP RC. ET AL., BRAIN RESEARCH, vol. 1534, 2013, pages 13 - 21 |
| PROC. NATL. ACAD. SCI. USA, vol. 60, 1968, pages 1275 |
| PROC. NATL. ACAD. SCI. USA, vol. 69, 1972, pages 2110 |
| PROC. NATL. ACAD. SCI. USA, vol. 74, 1977, pages 5463 |
| PROC. NATL. ACAD. SCI. USA, vol. 77, 1980, pages 4216 |
| PROC. NATL. ACAD. SCI. USA, vol. 78, 1981, pages 1527 |
| PROC. NATL. ACAD. SCI. USA, vol. 82, 1985, pages 4306 |
| PROC. NATL. ACAD. SCI. USA, vol. 84, 1987, pages 7413 |
| PROC. SOC. EXP. BIOL. MED., vol. 73, 1950, pages 1 |
| PROTEIN ENGINEERING, vol. 7, 1994, pages 1501 |
| RADIATION RESEARCH, vol. 148, 1997, pages 260 |
| RIDGWAY ET AL., PROTEIN ENGINEERING, vol. 9, no. 7, 1996, pages 617 - 621 |
| ROSEN A. ET AL., NATURE, vol. 267, 1977, pages 52 - 54 |
| SCHUMACHER S. ET AL., J. BIOL. CHEM., vol. 276, 2001, pages 7337 - 7345 |
| SCHUMACHER S. ET AL., J. CELL BIOL., vol. 136, 1997, pages 895 - 906 |
| SCHUMACHER S.STUBE E. M., J. NEUROCHEM., vol. 87, 2003, pages 1213 - 1223 |
| SCIENCE, vol. 122, 1952, pages 501 |
| SCIENCE, vol. 222, 1983, pages 778 |
| See also references of EP3816291A4 |
| SOMATIC CELL AND MOLECULAR GENETICS, vol. 12, 1986, pages 55 |
| SORRENTINO NC. ET AL., PEDIATR ENDOCRINOL REV., vol. 1, 2016, pages 630 - 638 |
| SPIESS ET AL., MOLECULAR IMMUNOLOGY, vol. 67, 2015, pages 95 - 106 |
| STRATEGIES, vol. 5, 1992, pages 81 |
| TAKUYA SHUO, SACHIKO AONO, KEIKO NAKANISHI, YOSHIHITO TOKITA, YOSHIYUKI KURODA, MICHIRU IDA, FUMIKO MATSUI, HIROYO MARUYAMA, TOSHI: "Ectodomain shedding of neuroglycan C, a brain-specific chondroitin sulfate proteoglycan, by TIMP-2- and TIMP-3- sensitive proteolysis", JOURNAL OF NEUROCHEMISTRY, vol. 102, no. 5, 15 September 2007 (2007-09-15), pages 1561 - 1568, XP055763216, ISSN: 0022-3042, DOI: 10.1111/j.1471-4159.2007.04658.x * |
| THE JOURNAL OF THE AMERICAN MEDICAL ASSOCIATION, vol. 199, 1967, pages 519 |
| TOMIZUKA K. ET AL., PROC NATL ACAD SCI USA, vol. 97, 2000, pages 722 - 7 |
| VIROLOGY, vol. 8, 1959, pages 396 |
| WANG W. ET AL., CLIN. PHARMACOL. THER., vol. 84, 2008, pages 548 - 558 |
| WATANABE E. ET AL., J. BIOL. CHEM., vol. 270, 1995, pages 26876 - 26882 |
| WATANABE, E. ET AL.: "Neuroglycan C, a novel membrane-spanning chondroitin sulfate proteoglycan that is restricted to the brain", J. BIOL. CHEM., vol. 270, no. 45, 1995, pages 26876 - 26882, XP055326580, DOI: 10.1074/jbc.270.45.26876 * |
| WEBSTER CI. ET AL., FASEB J., vol. 30, 2016, pages 1927 - 1940 |
| WINTER G. ET AL., ANNU REV IMMUNOL, vol. 12, 1994, pages 433 - 55 |
| WRAITH JE. ET AL., J. PEDIATR., vol. 144, no. 5, 2004, pages 581 - 588 |
| WU ET AL., NATURE BIOTECHNOLOGY, vol. 25, no. 11, 2007, pages 1290 - 1297 |
| YASUDA Y. ET AL., NEUROSCI. RES., vol. 32, 1998, pages 313 - 322 |
| YU YJ. ET AL., SCIENCE TRANSLATIONAL MEDICINE, vol. 6, 2014, pages 261ra154 |
| YUN ZHANG. ET AL., J. PHARMACOL. EXP. THER., vol. 313, no. 3, 2005, pages 1075 - 1081 |
| ZHANG Y. ET AL., J. NEUROIMMUNOL., vol. 114, no. 1-2, 2001, pages 168 - 172 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3816291A4 (en) | 2022-03-16 |
| AU2019295277A1 (en) | 2021-01-21 |
| JP7397444B2 (ja) | 2023-12-13 |
| JPWO2020004490A1 (ja) | 2021-08-02 |
| CA3104997A1 (en) | 2020-01-02 |
| KR20210025023A (ko) | 2021-03-08 |
| TW202012445A (zh) | 2020-04-01 |
| EP3816291A1 (en) | 2021-05-05 |
| US20210269548A1 (en) | 2021-09-02 |
| CN112424357A (zh) | 2021-02-26 |
| US11965035B2 (en) | 2024-04-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240124577A1 (en) | Antibody which binds to myelin oligodendrocyte glycoprotein | |
| JP7397444B2 (ja) | コンドロイチン硫酸プロテオグリカン-5に結合する抗体 | |
| WO2011155579A1 (ja) | 抗Trop-2抗体 | |
| WO2011155607A1 (ja) | 抗tim-3抗体 | |
| WO2012176779A1 (ja) | 抗erbB3抗体 | |
| TWI811260B (zh) | 與CD40及EpCAM結合之雙專一性抗體 | |
| KR20120110141A (ko) | 항 IgA1 항체 | |
| JP7397445B2 (ja) | Cell Adhesion Molecule3に結合する抗体 | |
| WO2013005649A1 (ja) | 抗ヒトccr6抗体 | |
| JP6803231B2 (ja) | 抗ヒトGas6モノクローナル抗体 | |
| WO2016111344A1 (ja) | Trailr2とpsmaに結合するバイスペシフィック抗体 | |
| KR20200029469A (ko) | 항인간 ccr1 모노클로날 항체 | |
| WO2017126587A1 (ja) | 抗ヒトccr1モノクローナル抗体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19826585 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020527592 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 3104997 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2019295277 Country of ref document: AU Date of ref document: 20190626 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019826585 Country of ref document: EP Effective date: 20210126 |