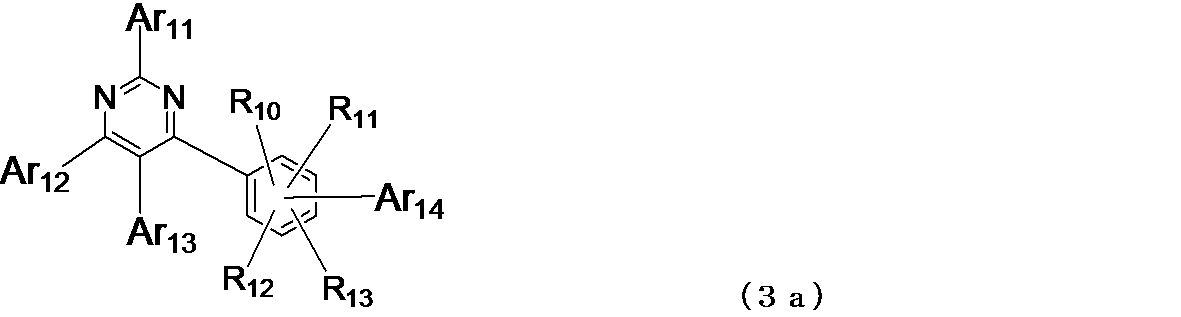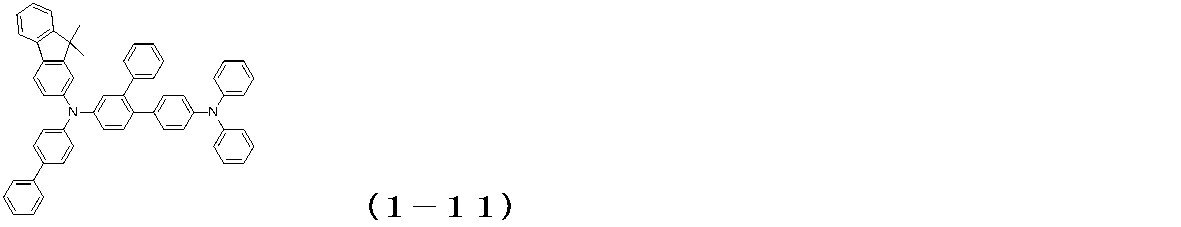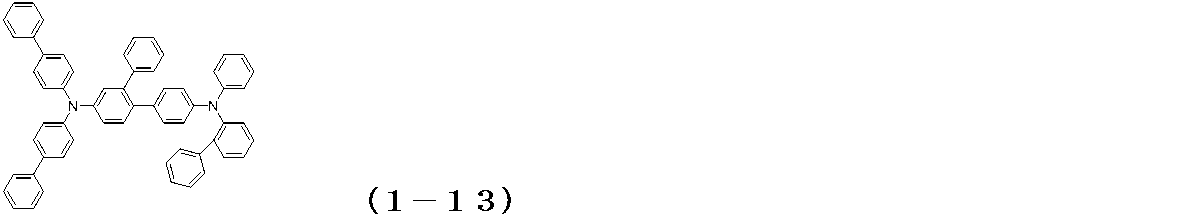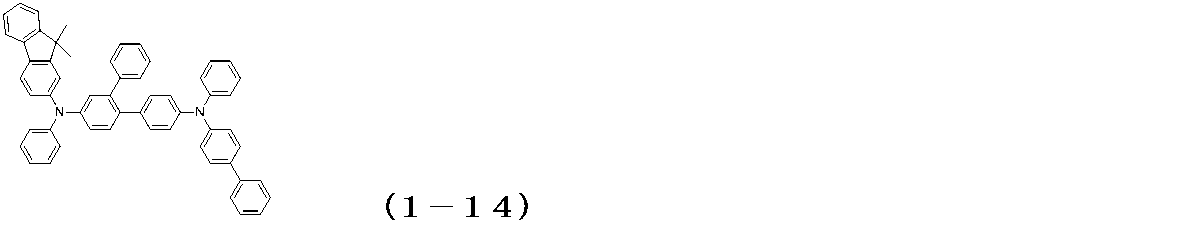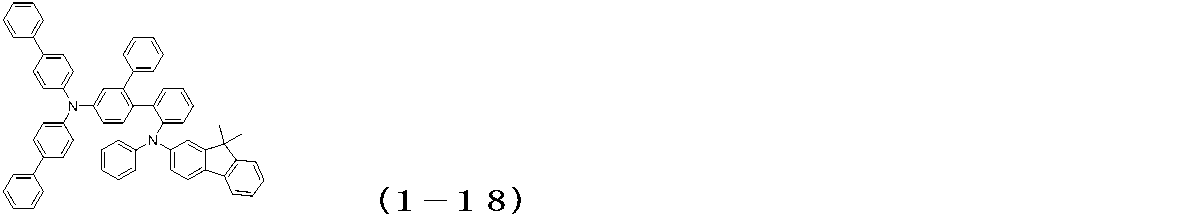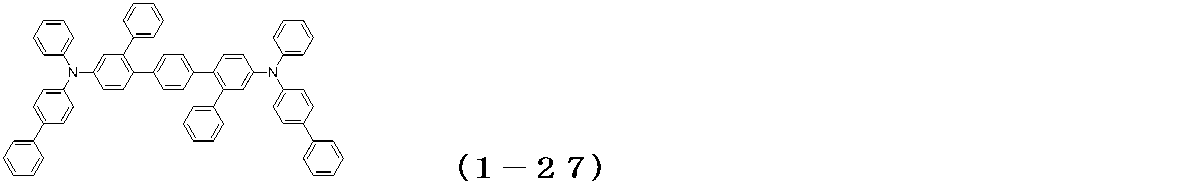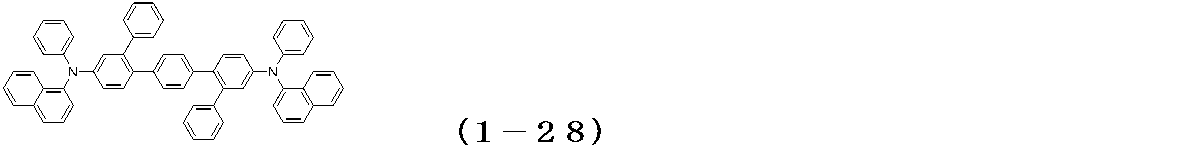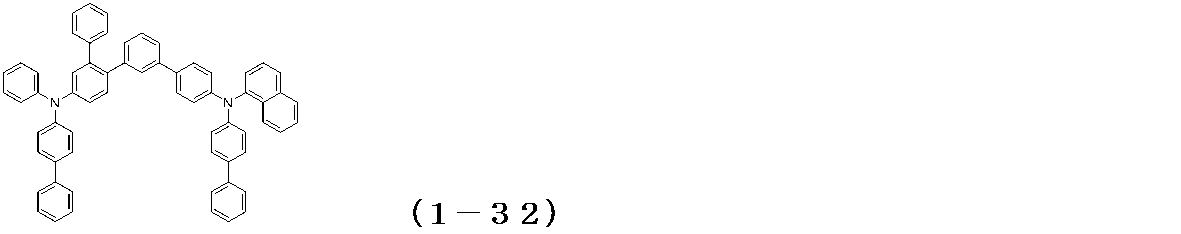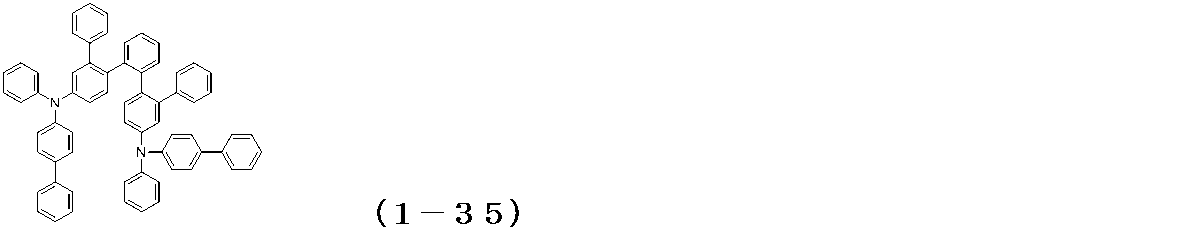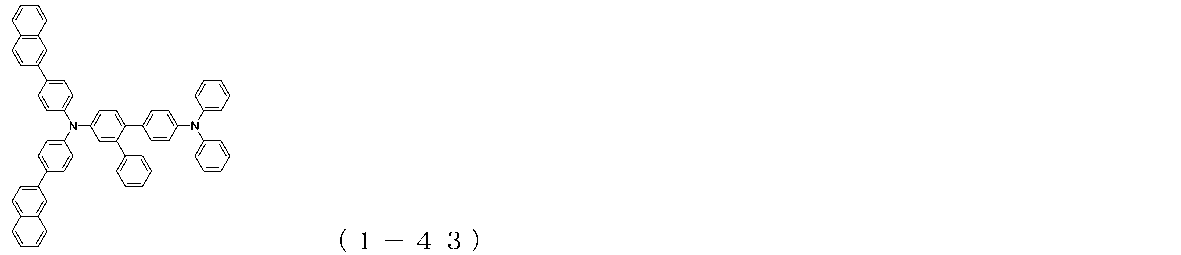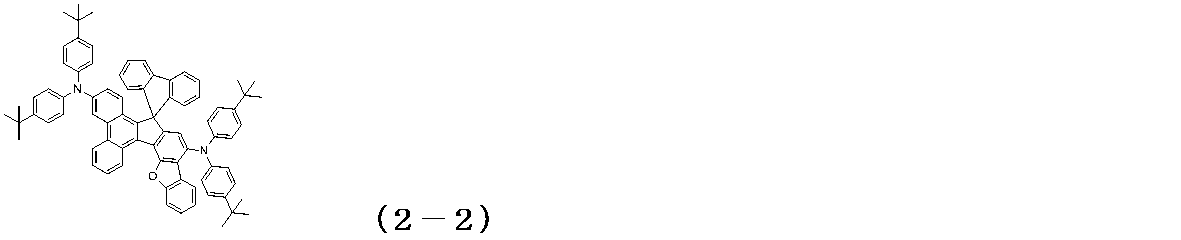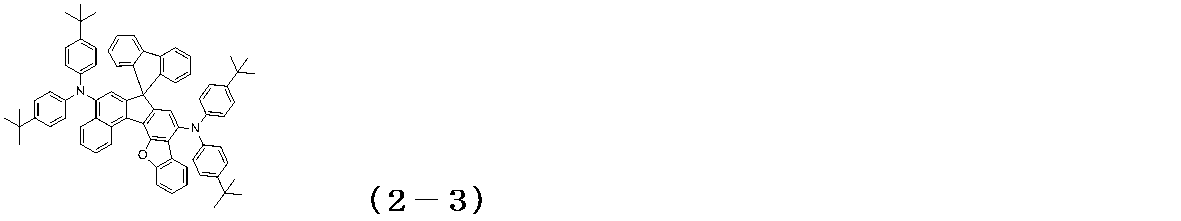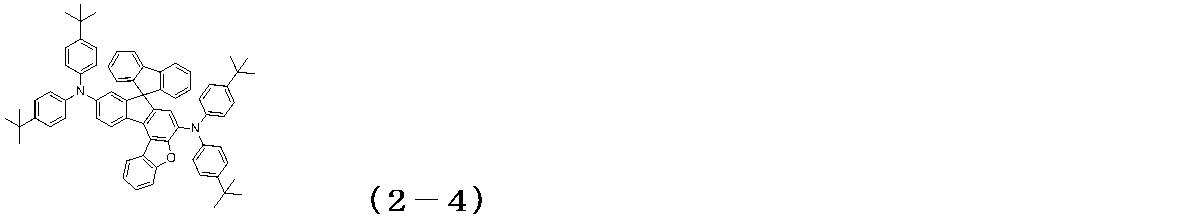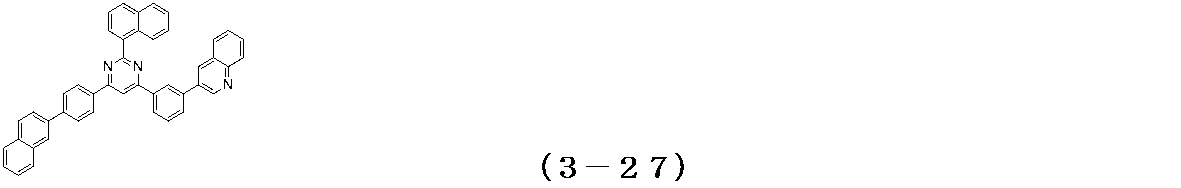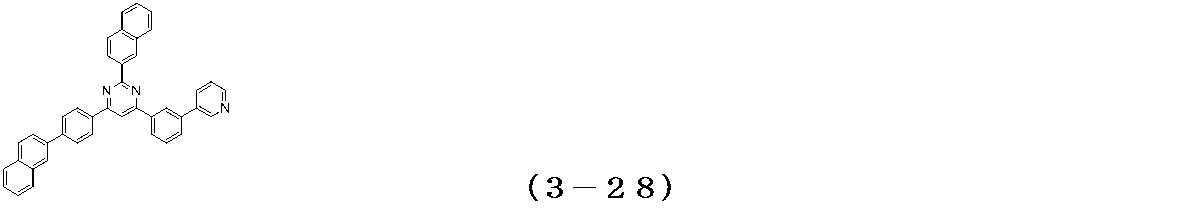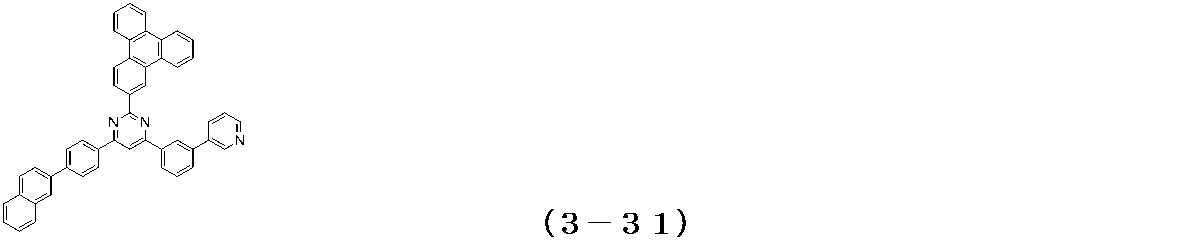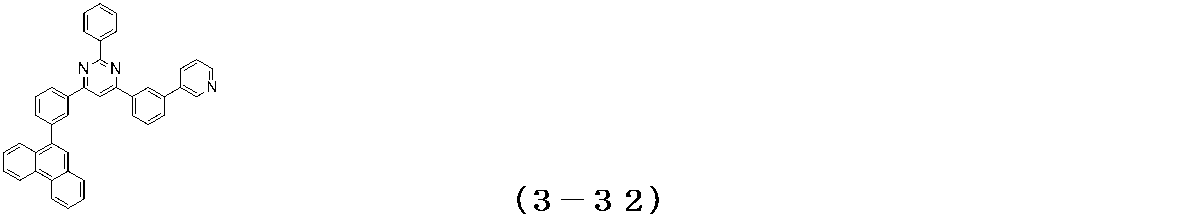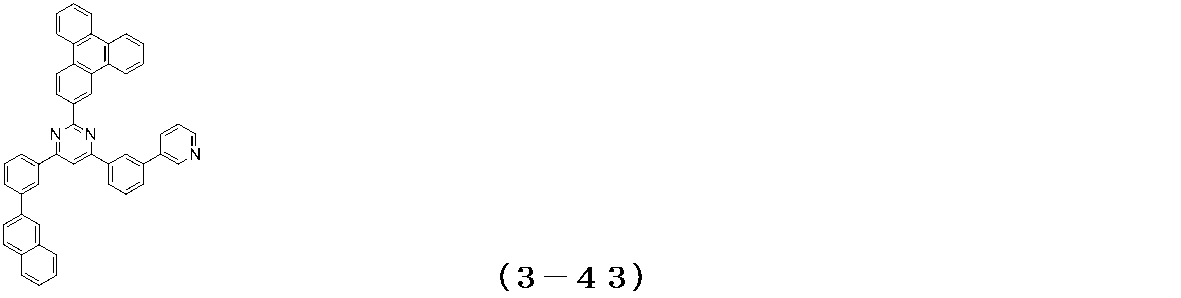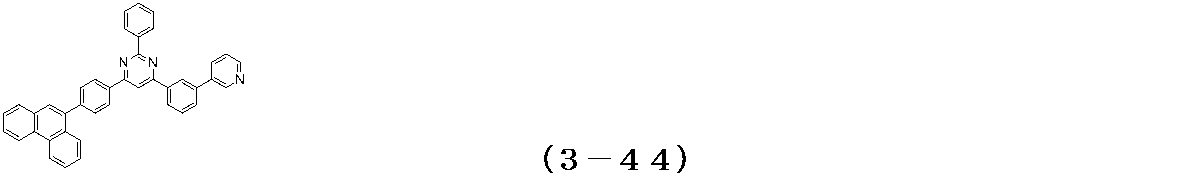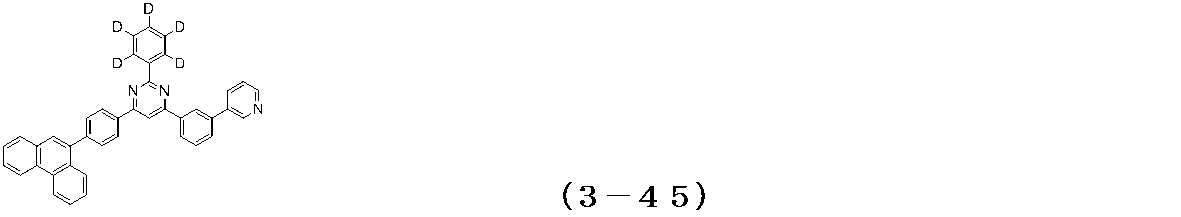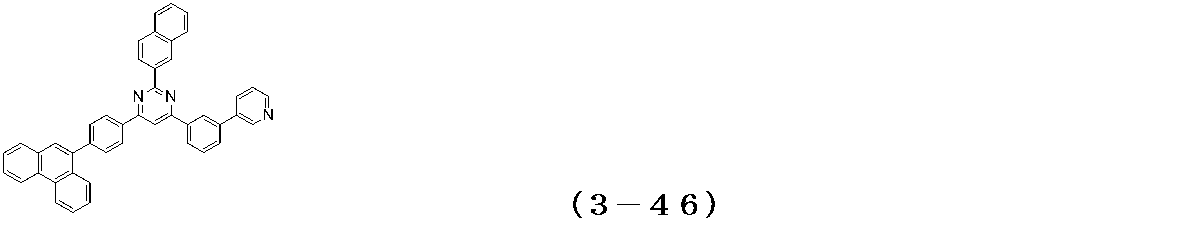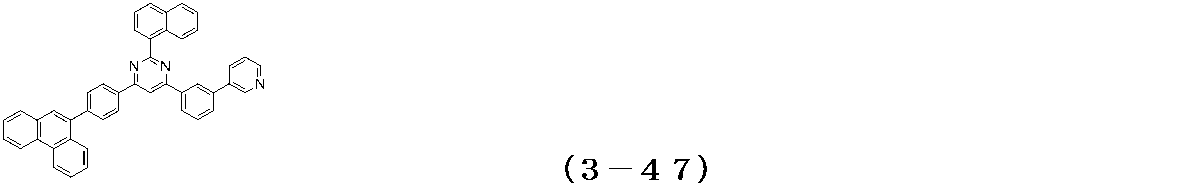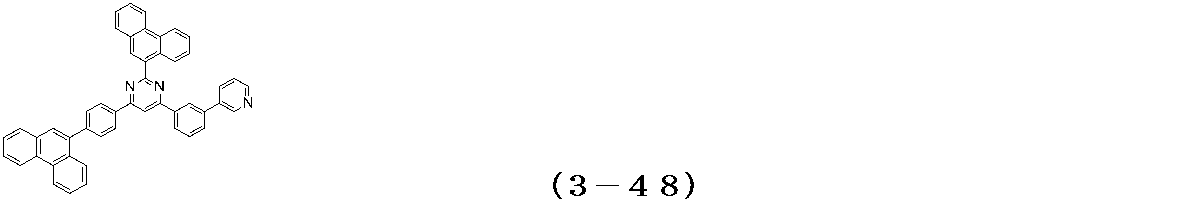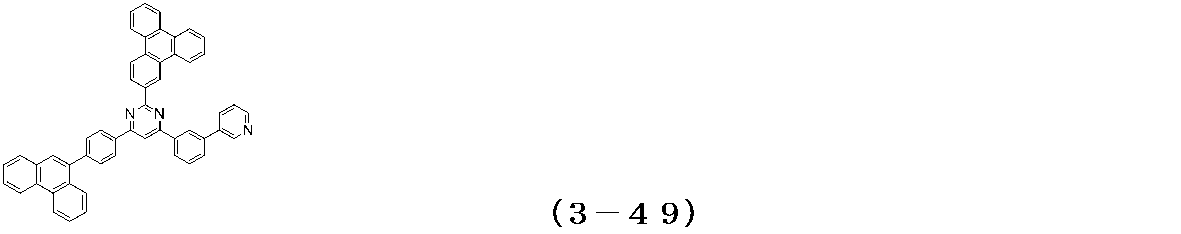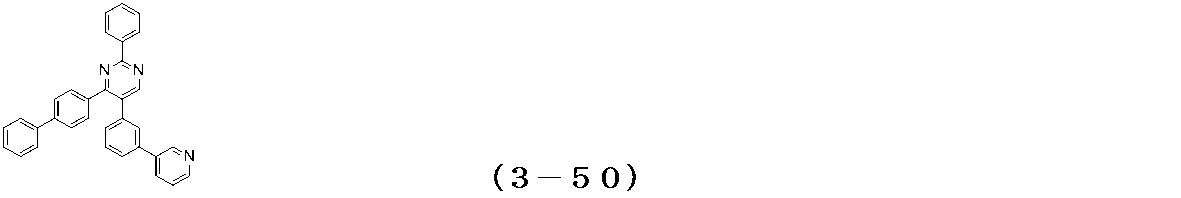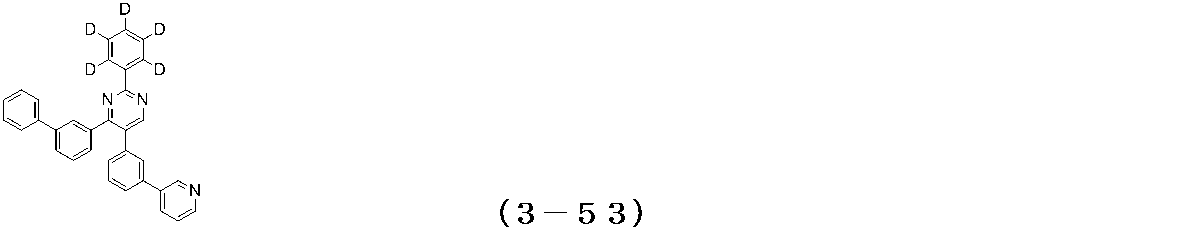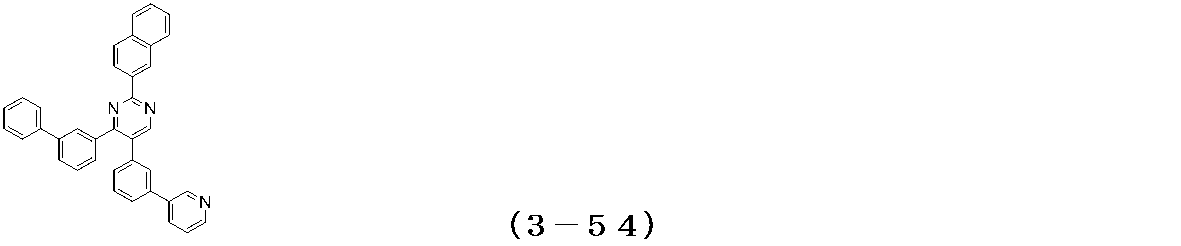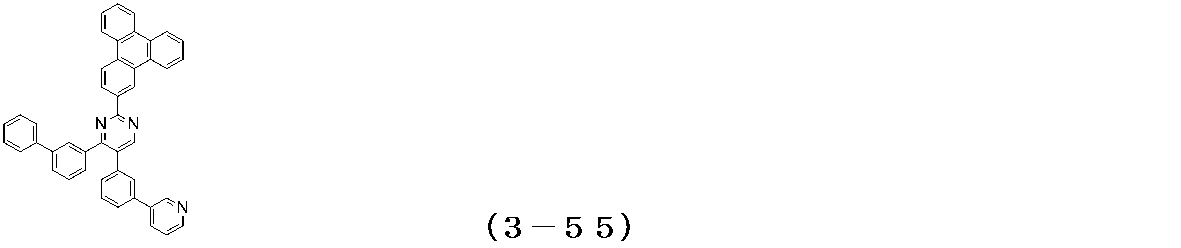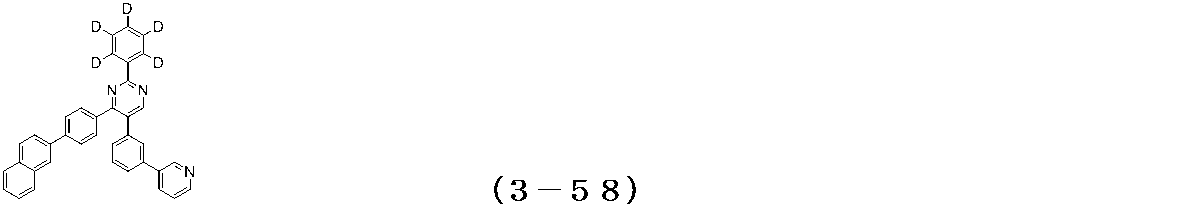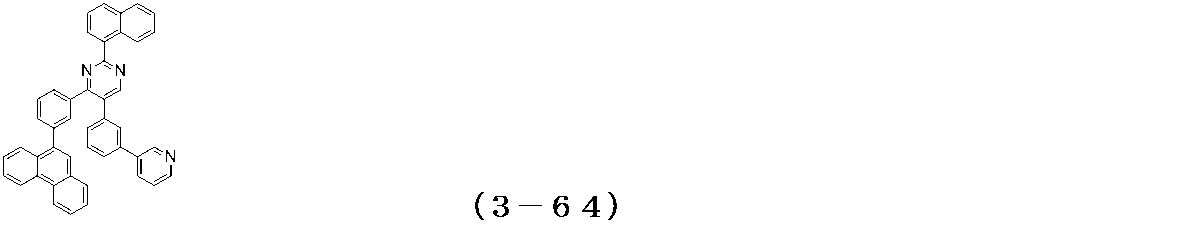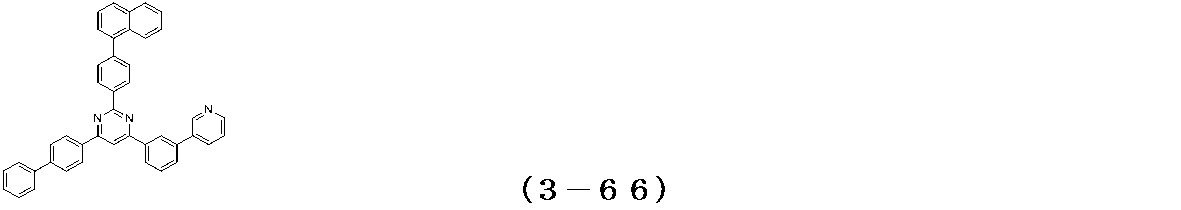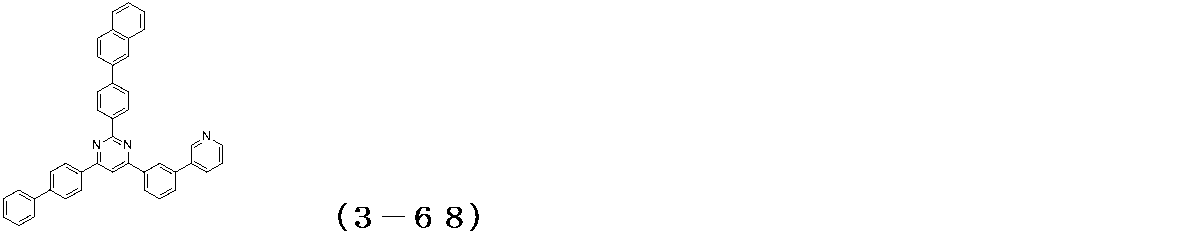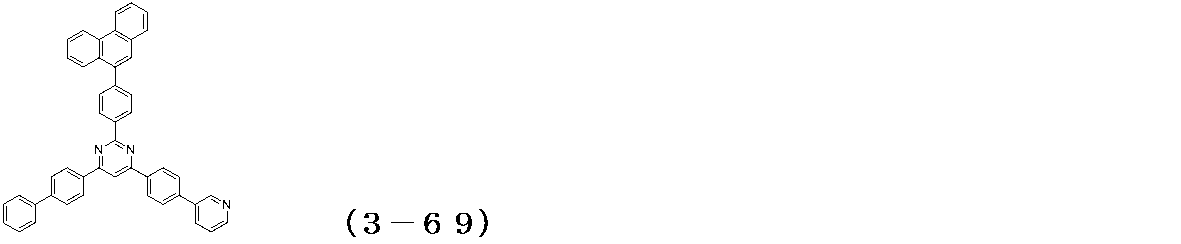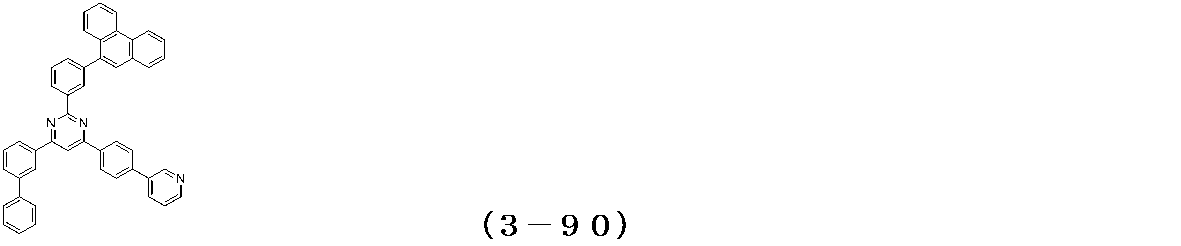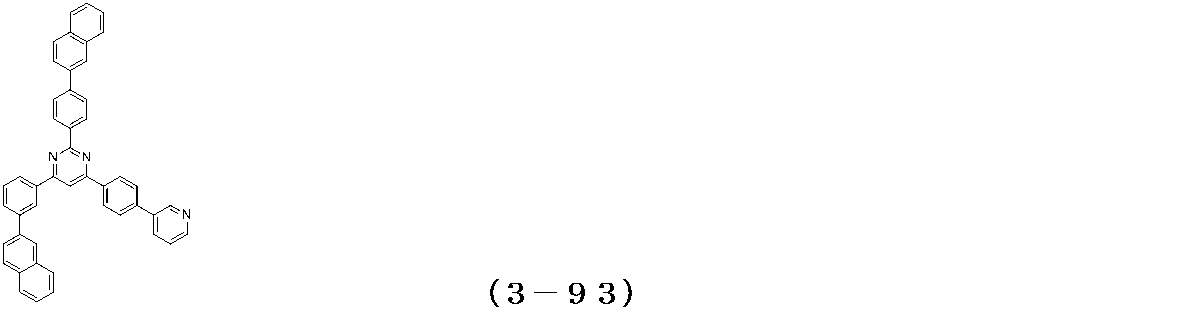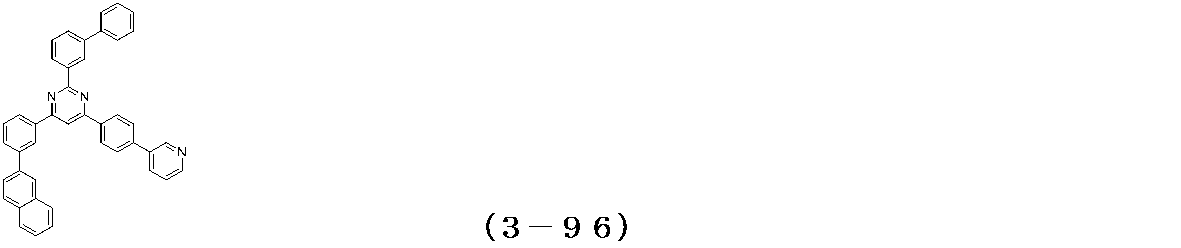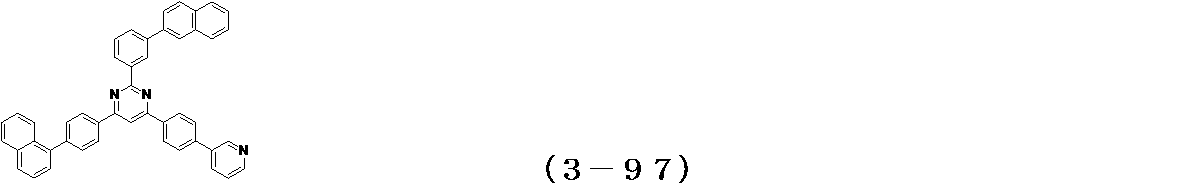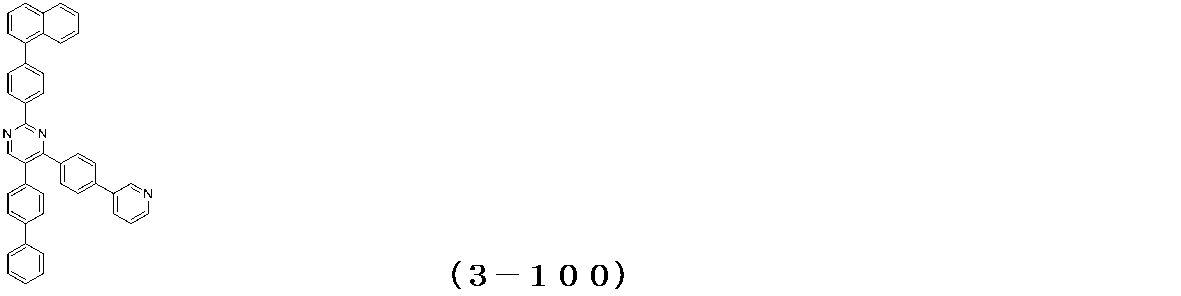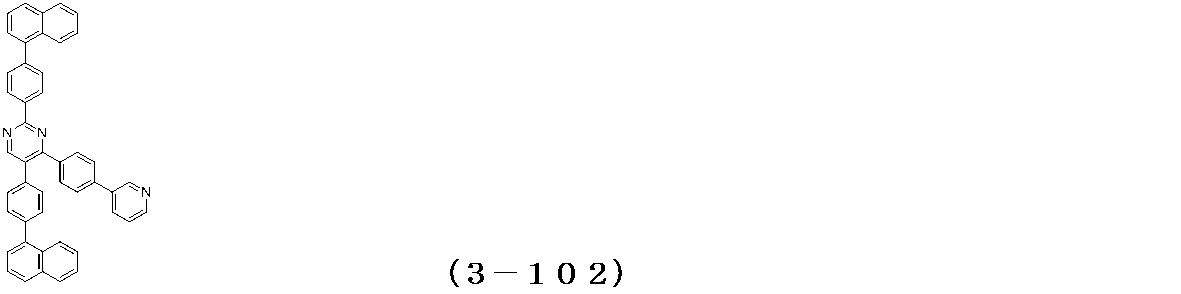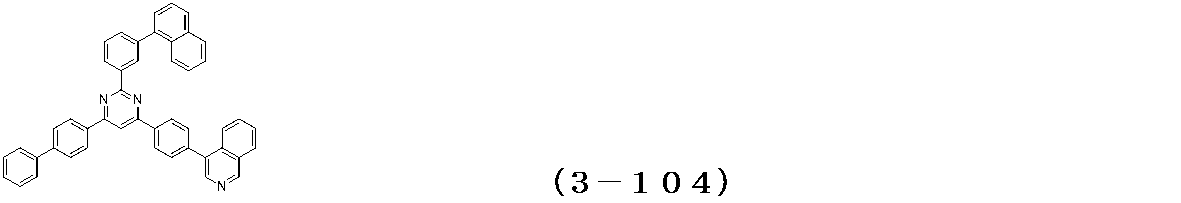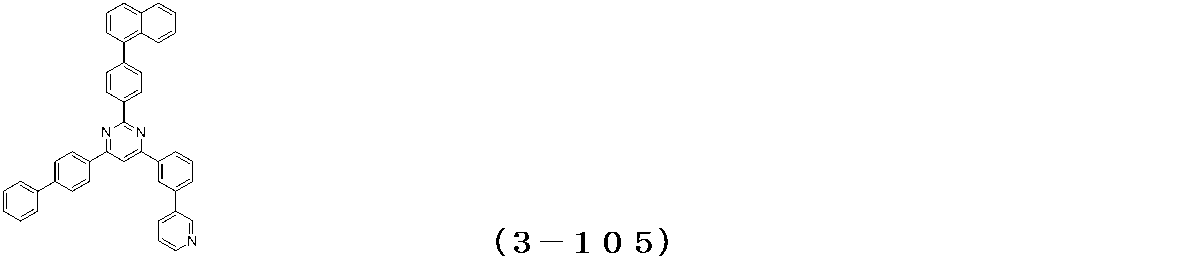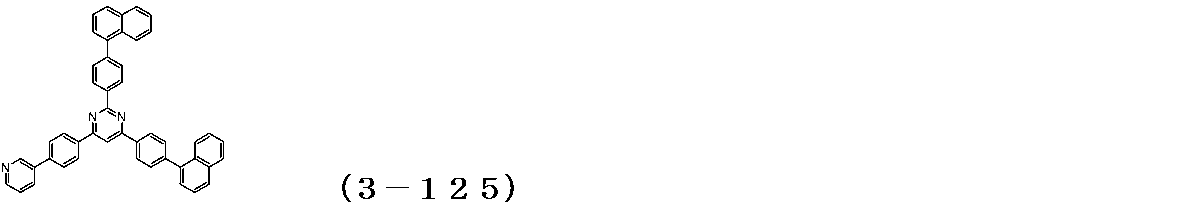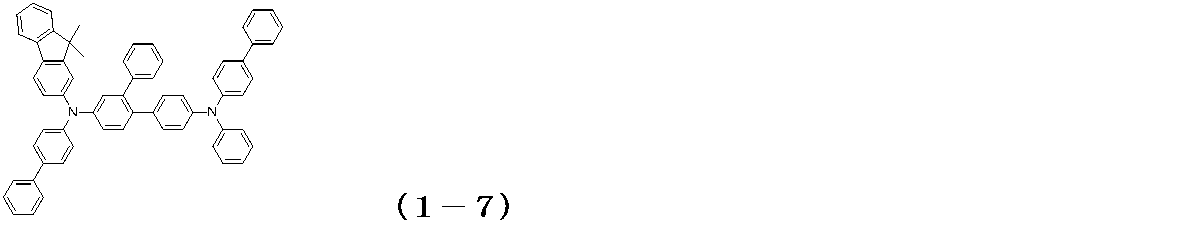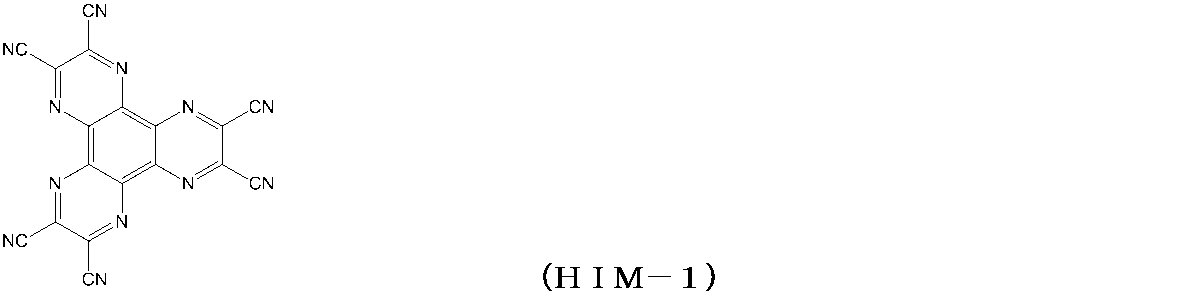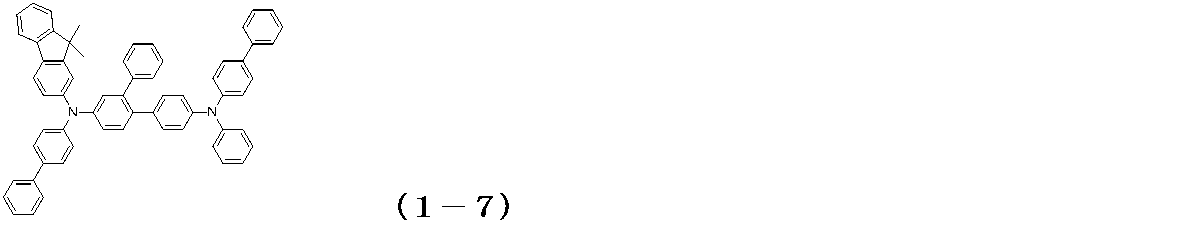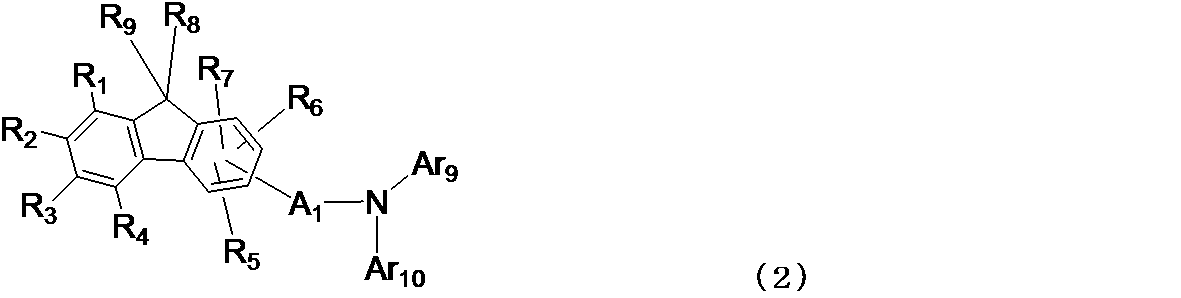WO2017122813A1 - 有機エレクトロルミネッセンス素子 - Google Patents
有機エレクトロルミネッセンス素子 Download PDFInfo
- Publication number
- WO2017122813A1 WO2017122813A1 PCT/JP2017/001112 JP2017001112W WO2017122813A1 WO 2017122813 A1 WO2017122813 A1 WO 2017122813A1 JP 2017001112 W JP2017001112 W JP 2017001112W WO 2017122813 A1 WO2017122813 A1 WO 2017122813A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituted
- unsubstituted
- atom
- substituent
- Prior art date
Links
- 0 *c1nc(N)ncc1 Chemical compound *c1nc(N)ncc1 0.000 description 7
- XOFHQYLNSNZGKK-UHFFFAOYSA-N C(C1)C(C2NC(C3=C(C=CC=C4)C4=CCC3)=NC=C2c2cc(C3C=NC=CC3)ccc2)C=CC1c1ccccc1 Chemical compound C(C1)C(C2NC(C3=C(C=CC=C4)C4=CCC3)=NC=C2c2cc(C3C=NC=CC3)ccc2)C=CC1c1ccccc1 XOFHQYLNSNZGKK-UHFFFAOYSA-N 0.000 description 1
- MRSBHAJTOPJTIH-UHFFFAOYSA-N C(C1)C=Cc2c1c(CCC(c1nc(C(C3)CC=CC3c3cccnc3)cc(C(C=C3)=CCC3c3ccccc3)n1)=C1)c1c1c2cccc1 Chemical compound C(C1)C=Cc2c1c(CCC(c1nc(C(C3)CC=CC3c3cccnc3)cc(C(C=C3)=CCC3c3ccccc3)n1)=C1)c1c1c2cccc1 MRSBHAJTOPJTIH-UHFFFAOYSA-N 0.000 description 1
- ZESNSYHSBUGMPB-UHFFFAOYSA-N C(CC(c1ccccc1)=C1)C=C1c1nc(C2=C3C=CC=C(C4C=CC=CC44)C3C4C=C2)nc(-c2cc(-c3cccnc3)ccc2)c1 Chemical compound C(CC(c1ccccc1)=C1)C=C1c1nc(C2=C3C=CC=C(C4C=CC=CC44)C3C4C=C2)nc(-c2cc(-c3cccnc3)ccc2)c1 ZESNSYHSBUGMPB-UHFFFAOYSA-N 0.000 description 1
- ZHSPYIPHUUHQNY-UHFFFAOYSA-N C(CC=C1)C2C1=CN=CC2c1cccc(-c2cnc(-c3cccc4c3cccc4)nc2-c2cc(-c3cc4ccccc4cc3)ccc2)c1 Chemical compound C(CC=C1)C2C1=CN=CC2c1cccc(-c2cnc(-c3cccc4c3cccc4)nc2-c2cc(-c3cc4ccccc4cc3)ccc2)c1 ZHSPYIPHUUHQNY-UHFFFAOYSA-N 0.000 description 1
- LFZOLXBLWKKHMF-UHFFFAOYSA-N C1C=CC(c2cccnc2)=CC1c1cc(-c2cccc(-c3cc4ccccc4cc3)c2)nc(-c2cc3ccccc3cc2)n1 Chemical compound C1C=CC(c2cccnc2)=CC1c1cc(-c2cccc(-c3cc4ccccc4cc3)c2)nc(-c2cc3ccccc3cc2)n1 LFZOLXBLWKKHMF-UHFFFAOYSA-N 0.000 description 1
- XGTPEGYYLKTICU-BRJRFNKRSA-N CC1[C@H](I)[IH]CC1 Chemical compound CC1[C@H](I)[IH]CC1 XGTPEGYYLKTICU-BRJRFNKRSA-N 0.000 description 1
- RTGBYKKJNBCIDZ-ZOSDIUFLSA-N CCC(C(CC12[C@H](C)CC3C1CC3)N)[C@@H]2I Chemical compound CCC(C(CC12[C@H](C)CC3C1CC3)N)[C@@H]2I RTGBYKKJNBCIDZ-ZOSDIUFLSA-N 0.000 description 1
- YXFVVABEGXRONW-UHFFFAOYSA-N Cc1ccccc1 Chemical compound Cc1ccccc1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B57/00—Other synthetic dyes of known constitution
- C09B57/008—Triarylamine dyes containing no other chromophores
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/54—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to two or three six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/57—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton
- C07C211/61—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton with at least one of the condensed ring systems formed by three or more rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/80—[b, c]- or [b, d]-condensed
- C07D209/82—Carbazoles; Hydrogenated carbazoles
- C07D209/86—Carbazoles; Hydrogenated carbazoles with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the ring system
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
- H10K50/12—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers comprising dopants
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/622—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing four rings, e.g. pyrene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/636—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising heteroaromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/06—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members
- C07C2603/10—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings
- C07C2603/12—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings only one five-membered ring
- C07C2603/18—Fluorenes; Hydrogenated fluorenes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1014—Carbocyclic compounds bridged by heteroatoms, e.g. N, P, Si or B
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2102/00—Constructional details relating to the organic devices covered by this subclass
- H10K2102/301—Details of OLEDs
- H10K2102/351—Thickness
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/15—Hole transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/16—Electron transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K71/00—Manufacture or treatment specially adapted for the organic devices covered by this subclass
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K71/00—Manufacture or treatment specially adapted for the organic devices covered by this subclass
- H10K71/10—Deposition of organic active material
- H10K71/16—Deposition of organic active material using physical vapour deposition [PVD], e.g. vacuum deposition or sputtering
- H10K71/164—Deposition of organic active material using physical vapour deposition [PVD], e.g. vacuum deposition or sputtering using vacuum deposition
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
Definitions
- the present invention relates to an organic electroluminescence element which is a self-luminous element suitable for various display devices, and more specifically, a specific arylamine compound and the compound (and a compound having a pyrimidine ring structure having a specific structure).
- the present invention relates to the used organic electroluminescence element (hereinafter abbreviated as an organic EL element).
- the organic EL element is a self-luminous element, it has been actively researched because it is brighter and more visible than a liquid crystal element and can be clearly displayed.
- Non-Patent Document 2 An element utilizing light emission by thermally activated delayed fluorescence (TADF) has also been developed.
- TADF thermally activated delayed fluorescence
- the light emitting layer can be prepared by doping a charge transporting compound generally called a host material with a fluorescent compound, a phosphorescent compound or a material emitting delayed fluorescence.
- a charge transporting compound generally called a host material with a fluorescent compound, a phosphorescent compound or a material emitting delayed fluorescence.
- selection of an organic material in the organic EL element greatly affects various characteristics such as efficiency and durability of the element (for example, see Non-patent document 2).
- the light injected from both electrodes is recombined in the light emitting layer to obtain light emission, but it is important how efficiently both holes and electrons are transferred to the light emitting layer. It is necessary to make the device excellent in carrier balance.
- the probability of recombination of holes and electrons is improved by improving the hole injection property and blocking the electrons injected from the cathode, and further excitons generated in the light emitting layer. By confining, high luminous efficiency can be obtained. Therefore, the role of the hole transport material is important, and there is a demand for a hole transport material that has high hole injectability, high hole mobility, high electron blocking properties, and high durability against electrons. ing.
- the heat resistance and amorphous nature of the material are important for the lifetime of the element.
- thermal decomposition occurs even at a low temperature due to heat generated when the element is driven, and the material is deteriorated.
- the thin film is crystallized even in a short time, and the element is deteriorated. For this reason, the material used is required to have high heat resistance and good amorphous properties.
- NPD N, N′-diphenyl-N, N′-di ( ⁇ -naphthyl) benzidine (NPD) and various aromatic amine derivatives are known as hole transport materials that have been used in organic EL devices so far.
- NPD has a good hole transport capability, but its glass transition point (Tg), which is an index of heat resistance, is as low as 96 ° C., and device characteristics are degraded due to crystallization under high temperature conditions (for example, Non-patent document 4).
- Tg glass transition point
- arylamine compounds having a substituted carbazole structure have been proposed as compounds with improved characteristics such as heat resistance and hole injection properties (see, for example, Patent Document 4 and Patent Document 5), these compounds are injected with holes.
- the elements used for the layer or the hole transport layer have been improved in heat resistance, luminous efficiency, etc., they are still not sufficient, and further lower driving voltage and higher luminous efficiency are required. .
- JP-A-8-048656 Japanese Patent No. 3194657 Japanese Patent No. 4943840 JP 2006-151979 A International Publication No. 2008/62636 Korean Open Patent No. 10-2015-0130206 Korean Published Patent No. 10-2013-0060157
- the object of the present invention is as a material for an organic EL device having high efficiency and high durability, and for an organic EL device excellent in hole injection / transport performance, electron blocking capability, stability in a thin film state, and durability.
- the materials and various materials for organic EL elements having excellent hole and electron injection / transport performance, electron blocking ability, stability in a thin film state, and durability It is an object of the present invention to provide an organic EL element with high efficiency, low driving voltage, and long life by combining them so that the characteristics of these materials can be effectively expressed.
- the physical properties that the organic compound to be provided by the present invention should have include (1) good hole injection properties, (2) high hole mobility, and (3) a thin film state. It can be mentioned that it is stable and (4) it has excellent heat resistance. Further, the physical characteristics to be provided by the organic EL element to be provided by the present invention are (1) high luminous efficiency and power efficiency, (2) low emission start voltage, and (3) practical driving. Low voltage and (4) long life can be mentioned.
- arylamine-based materials are excellent in hole injection and transport capability, thin film stability and durability, and synthesized various compounds.
- an arylamine compound in which a specific position is substituted with an allyl group can efficiently inject and transport holes to the light emitting layer.
- an arylamine compound having a specific position substituted with an allyl group and a pyrimidine having a specific structure A compound having a ring structure is selected so that holes and electrons can be efficiently injected and transported to a light-emitting layer containing a specific light-emitting material (dopant), and a positive structure having a specific structure is provided so that carrier balance can be achieved.
- Various organic EL devices were prepared by combining a hole transport material, a specific light emitting material (dopant), and an electron transport material having a specific structure, and the characteristics of the device were intensively evaluated. As a result, the present invention has been completed.
- the following organic EL elements are provided.
- the hole transport layer contains an arylamine compound represented by the following general formula (1).
- An organic EL element characterized by the above.
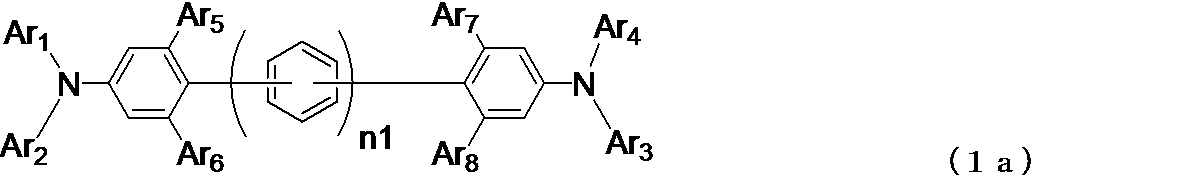
- Ar 1 ⁇ Ar 5 may be the same or different, a substituted or unsubstituted aromatic hydrocarbon group, a substituted or unsubstituted aromatic heterocyclic group, or a substituted or unsubstituted condensed polycyclic, Represents an aromatic group, and Ar 6 to Ar 8 may be the same as or different from each other, and represent a hydrogen atom, a substituted or unsubstituted aromatic hydrocarbon group, a substituted or unsubstituted aromatic heterocyclic group, or a substituted or unsubstituted group. Represents a substituted condensed polycyclic aromatic group, and n1 represents 0, 1 or 2.
- Ar 3 and Ar 4 are a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted group.
- a ring may be bonded to each other via a linking group such as an amino group, and Ar 3 or Ar 4 may be a single bond, substituted or unsubstituted, and a benzene ring to which —NAr 3 Ar 4 group is bonded.
- Methylene group, oxygen atom, sulfur It may be bonded to each other via a linking group such as a yellow atom or a monosubstituted amino group to form a ring.
- Ar 1 to Ar 5 may be the same or different from each other, and are a substituted or unsubstituted aromatic hydrocarbon group, a substituted or unsubstituted aromatic heterocyclic group, or a substituted or unsubstituted condensed polycyclic ring
- Ar 6 to Ar 8 may be the same as or different from each other, and represent a hydrogen atom, a substituted or unsubstituted aromatic hydrocarbon group, a substituted or unsubstituted aromatic heterocyclic group, or a substituted or unsubstituted group.
- n1 represents 0, 1 or 2.
- Ar 3 and Ar 4 are a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted group.
- a ring may be bonded to each other via a linking group such as an amino group, and Ar 3 or Ar 4 may be a single bond, substituted or unsubstituted, and a benzene ring to which —NAr 3 Ar 4 group is bonded.
- Methylene group, oxygen atom, sulfur It may be bonded to each other via a linking group such as a yellow atom or a monosubstituted amino group to form a ring.
- a 1 is a divalent group of a substituted or unsubstituted aromatic hydrocarbon, a divalent group of a substituted or unsubstituted aromatic heterocyclic ring, a substituted or unsubstituted condensed polycyclic aromatic divalent group, Or a single bond
- Ar 9 and Ar 10 may be the same or different from each other, and are substituted or unsubstituted aromatic hydrocarbon groups, substituted or unsubstituted aromatic heterocyclic groups, substituted or unsubstituted condensed poly a ring aromatic group, a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, one via a linking group such as a substituted amino group may be bonded to each other to form a ring .
- R 1 R 4 may be the same or different from each other, and may be a hydrogen atom, a deuterium atom, a fluorine atom, a chlorine atom, a
- Te single bond in each group to each other, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, may be bonded to each other to form a ring through a linking group such as monosubstituted amino group, R 5 ⁇
- a benzene ring to which R 7 is bonded may be bonded to each other via a linking group such as a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring.
- R 8 and R 9 may be the same or different from each other, and may have a substituent, a linear or branched alkyl group having 1 to 6 carbon atoms or a substituent.
- a ring may be formed by bonding to each other via a linking group such as an oxygen atom, a sulfur atom, or a monosubstituted amino group.
- Ar 11 represents a substituted or unsubstituted aromatic hydrocarbon group, or a substituted or unsubstituted condensed polycyclic aromatic group
- Ar 12 and Ar 13 may be the same as or different from each other
- Ar 14 represents a substituted or unsubstituted aromatic heterocyclic group
- R 10 to R 13 May be the same as or different from each other, a hydrogen atom, a deuterium atom, a fluorine atom, a chlorine atom, a cyano group, a trifluoromethyl group, a linear or branched alkyl group having 1 to 6 carbon atoms, a substituted or Represents an unsubstituted aromatic hydrocarbon group, a substituted or unsubstituted aromatic heterocyclic group, or a substituted or unsubstituted conden
- the “aromatic hydrocarbon group”, “aromatic heterocyclic group” or “fused polycyclic aromatic group” in the “unsubstituted fused polycyclic aromatic group” specifically includes a phenyl group, a biphenylyl group, a terphenylyl group.
- Ar 3 and Ar 4 may combine with each other via a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring
- Ar 3 and Ar 4 are bonded to each other via a benzene ring to which a —NAr 3 Ar 4 group is bonded and a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, and a monosubstituted amino group. They may combine to form a ring.
- substituted aromatic hydrocarbon group “substituted aromatic heterocyclic group” or “substituted condensed polycyclic aromatic group” represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)
- substituted aromatic hydrocarbon group “substituted aromatic heterocyclic group” or “substituted condensed polycyclic aromatic group” represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)
- substituents include deuterium atom, cyano group, nitro group; halogen atom such as fluorine atom, chlorine atom, bromine atom, iodine atom; methyl group, ethyl group, n-propyl group, isopropyl group
- a linear or branched alkyl group having 1 to 6 carbon atoms such as n-butyl group, isobutyl group, tert-butyl group, n-pentyl group, isopentyl group, neopentyl group,
- An aromatic vinyl group such as a styryl group and a naphthyl vinyl group; and a group such as an acyl group such as an acetyl group and a benzoyl group. May be substituted.
- these substituents may be bonded to each other via a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring.
- Specific examples of the “aromatic hydrocarbon”, “aromatic heterocycle” or “fused polycyclic aromatic” include benzene, biphenyl, terphenyl, tetrakisphenyl, styrene, naphthalene, anthracene, acenaphthalene, fluorene, phenanthrene, Indane, pyrene, triphenylene, pyridine, pyrimidine, triazine,
- a divalent group of a substituted or unsubstituted aromatic hydrocarbon “a divalent group of a substituted or unsubstituted aromatic heterocycle” represented by A 1 in the general formula (2), or “substituted or The “unsubstituted fused polycyclic aromatic divalent group” is a divalent group formed by removing two hydrogen atoms from the above “aromatic hydrocarbon”, “aromatic heterocycle” or “fused polycyclic aromatic”. To express.
- these divalent groups may have a substituent.
- the “substituted aromatic carbonization represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)” is used.
- Substituted or unsubstituted aromatic hydrocarbon group “substituted or unsubstituted aromatic heterocyclic group” represented by Ar 9 or Ar 10 in the general formula (2), or “substituted or unsubstituted condensed poly group”
- the “aromatic hydrocarbon group”, “aromatic heterocyclic group” or “condensed polycyclic aromatic group” in the “ring aromatic group” includes Ar 1 to Ar in the general formula (1) and general formula (1a).
- Ar 8 substituted or unsubstituted aromatic hydrocarbon group "," aromatic in the "substituted or unsubstituted aromatic heterocyclic group” or “substituted or unsubstituted condensed polycyclic aromatic group” hydrocarbon Examples thereof include the same as those described for the “hydrogen group”, “aromatic heterocyclic group” or “fused polycyclic aromatic group”, and Ar 9 and Ar 10 are single bonds, substituted or unsubstituted methylene groups. Oxygen atom, sulfur atom, monosubstituted amino group, etc. It may be bonded to each other to form a ring via Yuimoto.
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)” may be used.
- a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)” may be used.
- “Substituted aromatic heterocyclic group ”or“ Substituted condensed polycyclic aromatic group ” can be the same as those shown for“ Substituent ”, and possible embodiments are also the same. Can do.
- a linear or branched alkyl group having 1 to 6 carbon atoms which may have a substituent represented by R 1 to R 7 in the general formula (2), “having a substituent In the “cycloalkyl group having 5 to 10 carbon atoms” or “straight or branched alkenyl group having 2 to 6 carbon atoms which may have a substituent”.
- Examples of “straight or branched alkyl group of 6”, “cycloalkyl group of 5 to 10 carbon atoms” or “straight chain or branched alkenyl group of 2 to 6 carbon atoms” specifically include , Methyl group, ethyl group, n-propyl group, isopropyl group, n-butyl group, isobutyl group, tert-butyl group, n-pentyl group, isopentyl group, neopentyl group, n-hexyl group, cyclopentyl group, cyclo A xyl group, a 1-adamantyl group, a 2-adamantyl group, a vinyl group, an allyl group, an isopropenyl group, a 2-butenyl group, and the like.
- These groups are a single bond, a substituted or unsubstituted methylene group, These groups (R 1 to R 7 ) and these groups (R 1 to R 7 ) may be bonded to each other through a linking group such as an oxygen atom, a sulfur atom or a monosubstituted amino group. And a benzene ring to which are directly bonded, may be bonded to each other via a linking group such as a substituted or unsubstituted methylene group, oxygen atom, sulfur atom or monosubstituted amino group to form a ring.
- a linear or branched alkyl group having 1 to 6 carbon atoms having a substituent represented by R 1 to R 7 in the general formula (2), “5 to 10 carbon atoms having a substituent”
- Specific examples of the “substituent” in the “cycloalkyl group of” or “straight-chain or branched alkenyl group having 2 to 6 carbon atoms having a substituent” include a deuterium atom, a cyano group, and a nitro group; Halogen atoms such as fluorine, chlorine, bromine and iodine; linear or branched alkyloxy having 1 to 6 carbon atoms such as methyloxy, ethyloxy and propyloxy; vinyl and allyl Alkenyl groups such as phenyl groups; aryloxy groups such as phenyloxy groups and tolyloxy groups; arylalkyloxy groups such as benzyloxy groups and phenethyloxy groups; Group, biphenylyl
- substituents may be bonded to each other via a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring.
- a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring.
- these groups (R 1 to R 7 ) and the benzene ring to which these groups (R 1 to R 7 ) are directly bonded are substituted or unsubstituted methylene groups, oxygen atoms, sulfur atoms, monosubstituted
- a ring may be formed by bonding to each other via a linking group such as an amino group.
- these groups may have a substituent, and the substituent is represented by R 1 to R 7 in the general formula (2), which is a straight chain having 1 to 6 carbon atoms having a substituent.
- Substituted or unsubstituted aromatic hydrocarbon group “substituted or unsubstituted aromatic heterocyclic group” represented by R 1 to R 7 in formula (2), or “substituted or unsubstituted condensed hydrocarbon group”
- the “aromatic hydrocarbon group”, “aromatic heterocyclic group” or “condensed polycyclic aromatic group” in the “ring aromatic group” includes Ar 1 to Ar in the general formula (1) and general formula (1a).
- aryl vinyl groups such as styryl groups and naphthyl vinyl groups
- acyl groups such as acetyl groups and benzoyl groups
- silyl groups such as trimethylsilyl groups and triphenylsilyl groups
- aromatic hydrocarbons such as diphenylamino groups and dinaphthylamino groups
- a disubstituted amino group substituted with a group or a condensed polycyclic aromatic group a disubstituted amino group substituted with an aromatic heterocyclic group such as a dipyridylamino group or a dithienylamino group
- an aromatic hydrocarbon group, a condensed polycycle examples thereof include a group such as a disubstituted amino group substituted with a substituent selected from an aromatic group or an aromatic heterocyclic group, and these substituents are further substituted by the above-exemplified substituents.
- substituents may be bonded to each other via a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring.
- a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring.
- aryloxy group in the “substituted or unsubstituted aryloxy group” represented by R 1 to R 7 in the general formula (2) include a phenyloxy group, a biphenylyloxy group, a terphenyl group.
- Examples include ryloxy group, naphthyloxy group, anthracenyloxy group, phenanthrenyloxy group, fluorenyloxy group, indenyloxy group, pyrenyloxy group, perylenyloxy group, and the like.
- a bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, a monosubstituted amino group or the like may be bonded to each other to form a ring, and these groups (R 1 to R 7 ) and in the benzene ring of these groups (R 1 ⁇ R 7) is directly bonded, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, such as monosubstituted amino group It may be bonded to each other to form a ring via Yuimoto.
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group” represented by R 1 to R 7 in the general formula (2), “substituted aromatic”
- R 1 to R 7 in the general formula (2) “substituted aromatic”
- Disubstituted amino group substituted by a group selected from an aromatic hydrocarbon group, an aromatic heterocyclic group or a condensed polycyclic aromatic group represented by R 1 to R 4 in the general formula (2)
- the “aromatic hydrocarbon group”, “aromatic heterocyclic group” or “fused polycyclic aromatic group” is represented by Ar 1 to Ar 8 in the above general formula (1) and general formula (1a).
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group” represented by R 1 to R 7 in the general formula (2), “substituted aromatic”
- R 1 to R 7 in the general formula (2) “substituted aromatic”
- the “disubstituted amino group substituted by a group selected from an aromatic hydrocarbon group, an aromatic heterocyclic group or a condensed polycyclic aromatic group” represented by R 1 to R 4 in the general formula (2) is: between these groups (R 1 ⁇ R 4) is, these groups (R 1 ⁇ R 4) has the "aromatic hydrocarbon group", the "aromatic heterocyclic group” or “condensed polycyclic aromatic group” May be bonded to each other through a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group, and these groups (R 1- R 4 ) and a benzene ring to which these groups (R 1 to R 4 ) are directly bonded, and these groups (R 1 to R 4 ) have an “aromatic hydrocarbon group” and “aromatic heterocycle” Group “or" condensed polycyclic aromatic group ", substituted or unsubsti
- a linear or branched alkyl group having 1 to 6 carbon atoms which may have a substituent represented by R 8 or R 9 in the general formula (2), “having a substituent In the “cycloalkyl group having 5 to 10 carbon atoms” or “straight or branched alkenyl group having 2 to 6 carbon atoms which may have a substituent”.
- the “6 linear or branched alkyl group”, “C 5 -C 10 cycloalkyl group” or “C 2 -C 6 linear or branched alkenyl group” includes the above general formula “A linear or branched alkyl group having 1 to 6 carbon atoms which may have a substituent” represented by R 1 to R 7 in (2), “having a substituent; May be a cycloalkyl group having 5 to 10 carbon atoms "or “Straight or branched alkyl group having 1 to 6 carbon atoms” in “optionally substituted linear or branched alkenyl group having 2 to 6 carbon atoms”, “carbon atom” Examples thereof are the same as those shown for the “cycloalkyl group of 5 to 10” or “straight or branched alkenyl group of 2 to 6 carbon atoms”, and these groups are single bonds, A ring may be formed by bonding to each other via a linking group such as a substituted or unsubstituted methylene group
- these groups may have a substituent, and the substituent is represented by R 1 to R 7 in the general formula (2), which is a straight chain having 1 to 6 carbon atoms having a substituent.
- R 1 to R 7 in the general formula (2), which is a straight chain having 1 to 6 carbon atoms having a substituent.
- Substituted or unsubstituted aromatic hydrocarbon group “substituted or unsubstituted aromatic heterocyclic group” represented by R 8 or R 9 in general formula (2), or “substituted or unsubstituted condensed hydrocarbon group”
- the “aromatic hydrocarbon group”, “aromatic heterocyclic group” or “condensed polycyclic aromatic group” in the “ring aromatic group” includes Ar 1 to Ar in the general formula (1) and general formula (1a).
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group” represented by R 1 to R 7 in the general formula (2), “substituted aromatic”
- R 1 to R 7 in the general formula (2) “substituted aromatic”
- the “aryloxy group” in the “substituted or unsubstituted aryloxy group” represented by R 8 and R 9 in the general formula (2) is represented by R 1 to R 7 in the general formula (2).
- lifted These groups are a single bond, a substituted or unsubstituted methylene group, oxygen atom , A sulfur atom, a monosubstituted amino group and the like may be bonded to each other to form a ring.
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group” represented by R 1 to R 7 in the general formula (2), “substituted aromatic”
- R 1 to R 7 in the general formula (2) “substituted aromatic”
- the “substituent” in the linking group “monosubstituted amino group” in formula (2) may have a “substituent” represented by R 1 to R 7 in formula (2).
- a linear or branched alkyl group having 1 to 6 carbon atoms in “group”, “substituted or unsubstituted aromatic heterocyclic group” or “substituted or unsubstituted condensed polycyclic aromatic group”, " Examples thereof include those similar to those shown for the “cycloalkyl group having 5 to 10 carbon atoms”, “aromatic hydrocarbon group”, “aromatic heterocyclic group” or “fused polycyclic aromatic group”. .
- these groups may have a substituent, and the substituent is represented by R 1 to R 7 in the general formula (2), which is a straight chain having 1 to 6 carbon atoms having a substituent.
- Specific examples of the ⁇ hydrogen group '' or ⁇ fused polycyclic aromatic group '' include phenyl group, biphenylyl group, terphenylyl group, quarterphenyl group, styryl group, naphthyl group, anthracenyl group, acenaphthenyl group, phenanthrenyl group, fluorenyl group, Examples thereof include an indenyl group, a pyrenyl group, a perylenyl group, a fluoranthenyl group, and a triphenylenyl group.
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)” may be used.
- a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)” may be used.
- “Substituted aromatic heterocyclic group ”or“ Substituted condensed polycyclic aromatic group ” can be the same as those shown for“ Substituent ”, and possible embodiments are also the same. Can do.
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)” may be used.
- a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) and general formula (1a)” may be used.
- “Substituted aromatic heterocyclic group ”or“ Substituted condensed polycyclic aromatic group ” can be the same as those shown for“ Substituent ”, and possible embodiments are also the same. Can do.
- linear or branched alkyl group having 1 to 6 carbon atoms represented by R 10 to R 13 in the general formula (3)
- R 10 to R 13 in the general formula (3)
- Specific examples of the “linear or branched alkyl group having 1 to 6 carbon atoms” represented by R 10 to R 13 in the general formula (3) include a methyl group, an ethyl group, and n-propyl.
- Group, i-propyl group, n-butyl group, 2-methylpropyl group, t-butyl group, n-pentyl group, 3-methylbutyl group, tert-pentyl group, n-hexyl group, iso-hexyl group and tert- A hexyl group can be mentioned.
- aromatic hydrocarbon group “aromatic heterocyclic group” or “fused polycyclic aromatic group” in the “ring aromatic group”
- a phenyl group a biphenylyl group, a terphenylyl group, a quarterphenyl group, Styryl, naphthyl, anthracenyl, acenaphthenyl, phenanthrenyl, fluorenyl, indenyl, pyrenyl, perylenyl, fluoranthenyl, triphenylenyl, triazinyl, pyridyl, pyrimidinyl, furyl, pyrrolyl, Thienyl, quinoly
- these groups may have a substituent, and as the substituent, a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) or general formula (1a)” may be used.
- a “substituted aromatic hydrocarbon group represented by Ar 1 to Ar 8 in the general formula (1) or general formula (1a)” may be used.
- “Substituted aromatic heterocyclic group ”or“ Substituted condensed polycyclic aromatic group ” can be the same as those shown for“ Substituent ”, and possible embodiments are also the same. Can do.
- n1 represents 0 or 1 to 2, and when n1 is 0, it means that two diarylaminobenzene rings are directly bonded (single bond).
- n1 When n1 is 1, it indicates that two diarylaminobenzene rings are bonded via one phenylene group. When n1 is 2, two diarylaminobenzene rings are two phenylene groups. It shows bonding through a group (biphenylene group).
- n1 In general formula (1) and general formula (1a), it is preferable that n1 is 0, that is, two diarylaminobenzene rings are directly bonded (single bond).
- Ar 3 or Ar 4 is bonded to —NAr 3 Ar 4 group (Ar 3 , Ar 4 and a diarylamino group composed of a nitrogen atom to which Ar 4 is bonded).
- a ring is bonded to each other via a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group is also preferable.
- the bonding position on the benzene ring is preferably a position adjacent to the —NAr 3 Ar 4 group.
- a 1 in the general formula (2) is preferably a “divalent group of a substituted or unsubstituted aromatic hydrocarbon” or a single bond, which is formed by removing two hydrogen atoms from benzene, biphenyl or naphthalene.
- a valent group or a single bond is more preferable, and a single bond is particularly preferable.
- Ar 9 and Ar 10 in the general formula (2) are preferably a phenyl group, a biphenylyl group, a naphthyl group, a fluorenyl group, an indenyl group, a pyridyl group, a dibenzofuranyl group, and a pyridobenzofuranyl group.
- Ar 9 and Ar 10 in the general formula (2) are directly or via a substituent of those groups, such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, and a monosubstituted amino group.
- a ring may be formed by bonding to each other via a linking group.
- At least one of R 1 to R 4 in the general formula (2) is “a disubstituted amino group substituted by a group selected from an aromatic hydrocarbon group, an aromatic heterocyclic group or a condensed polycyclic aromatic group”.
- the “aromatic hydrocarbon group”, “aromatic heterocyclic group”, and “condensed polycyclic aromatic group” include a phenyl group, a biphenylyl group, a naphthyl group, a fluorenyl group, and an indenyl group.
- a pyridyl group, a dibenzofuranyl group, and a pyridobenzofuranyl group are preferable.
- the two adjacent R 1 to R 4 or all are vinyl groups, and the two adjacent vinyl groups are bonded to each other through a single bond to form a condensed ring, Also preferred is an embodiment in which a naphthalene ring or a phenanthrene ring is formed together with the benzene ring to which R 1 to R 4 are bonded.
- any one of R 1 to R 4 is an “aromatic hydrocarbon group”, a benzene ring to which R 1 to R 4 are bonded, a substituted or unsubstituted methylene group,
- An embodiment in which a ring is bonded to each other via a linking group such as an oxygen atom, a sulfur atom, or a monosubstituted amino group is preferred.
- the “aromatic hydrocarbon group” is a phenyl group and is bonded to each other via a benzene ring to which R 1 to R 4 are bonded and a linking group such as an oxygen atom, a sulfur atom, or a monosubstituted amino group.
- any one of R 5 to R 7 is an “aromatic hydrocarbon group”, a benzene ring to which R 5 to R 7 are bonded, a substituted or unsubstituted methylene group,
- a linking group such as an oxygen atom, a sulfur atom, or a monosubstituted amino group is preferred.
- the “aromatic hydrocarbon group” is a phenyl group and is bonded to each other via a benzene ring to which R 5 to R 7 are bonded and a linking group such as an oxygen atom, a sulfur atom, or a monosubstituted amino group.
- a ring is formed, that is, an embodiment in which a dibenzofuran ring or a dibenzothiophene ring is formed with a benzene ring to which R 5 to R 7 are bonded is particularly preferable.
- R 8 and R 9 are “substituted or unsubstituted aromatic hydrocarbon group”, “substituted or unsubstituted oxygen-containing aromatic heterocyclic group” or “substituted or unsubstituted condensed group”.
- a “polycyclic aromatic group” is preferable, and a phenyl group, a naphthyl group, a phenanthrenyl group, a pyridyl group, a quinolyl group, an isoquinolyl group, and a dibenzofuranyl group are more preferable, and a phenyl group is particularly preferable.
- R 8 and R 9 are bonded to each other via a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring is preferable.
- a linking group such as a single bond, a substituted or unsubstituted methylene group, an oxygen atom, a sulfur atom, or a monosubstituted amino group to form a ring is preferable.
- a ring is bonded to each other through a bond is particularly preferable.
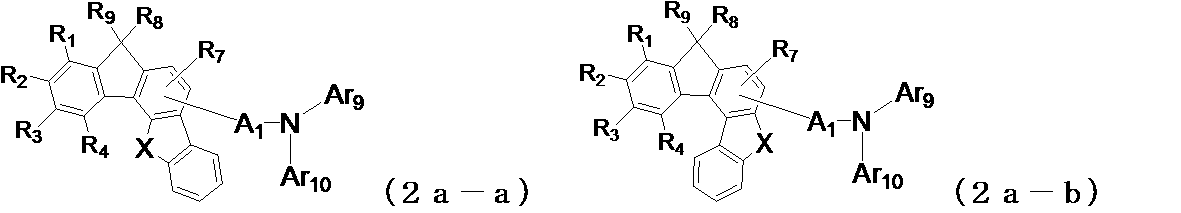
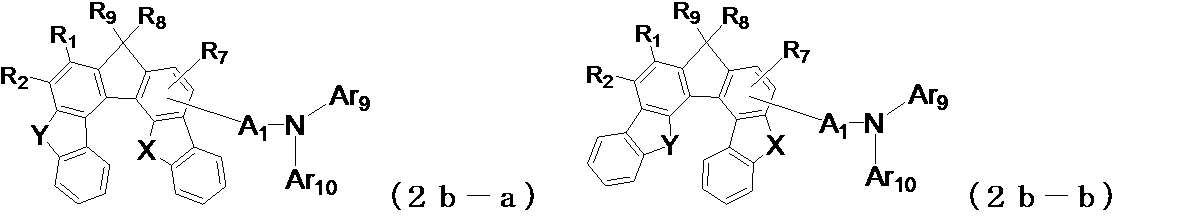
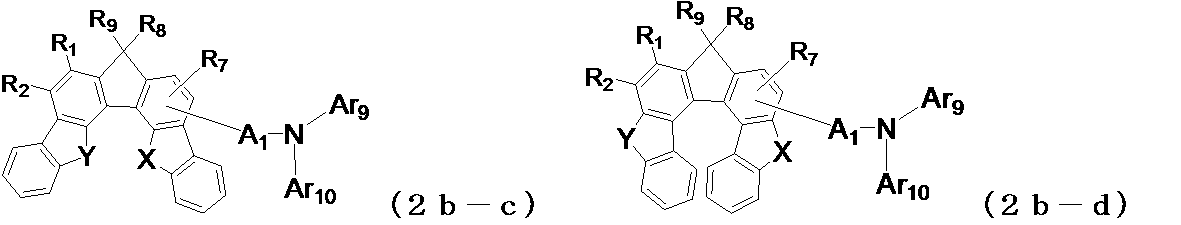
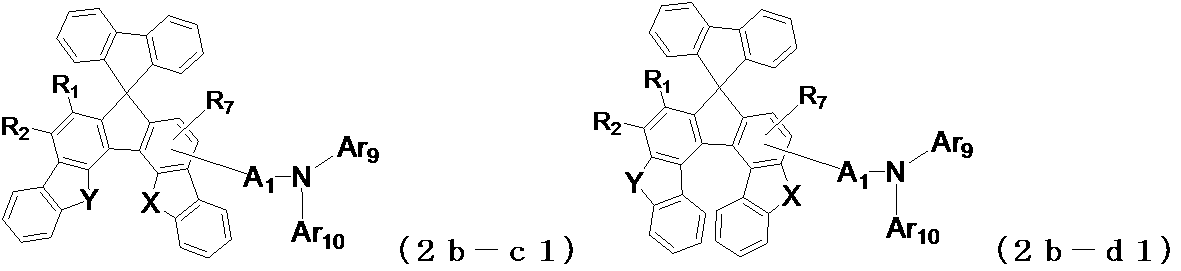
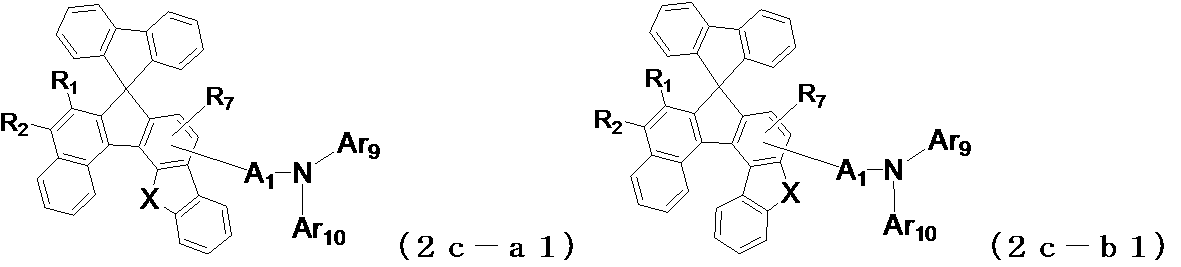
- the following general formula (2a-a1) , (2a-b1), (2b-a1), (2b-b1), (2b-c1), (2b-d1), (2c-a1) or (2c-b1) is preferably used. It is done.
- X and Y may be the same or different from each other, and represent an oxygen atom or a sulfur atom, and A 1 , Ar 9 , Ar 10 , R 1 to R 4 , R 7 are the same as those in the general formula (2) (Indicates the meaning as indicated by.)
- Ar 11 in the general formula (3) includes phenyl group, biphenylyl group, naphthyl group, anthracenyl group, acenaphthenyl group, phenanthrenyl group, fluorenyl group, indenyl group, pyrenyl group, perylenyl group, fluoranthenyl group, triphenylenyl group.
- a phenyl group, a biphenylyl group, a naphthyl group, an anthracenyl group, a phenanthrenyl group, a pyrenyl group, a fluoranthenyl group, and a triphenylenyl group are more preferable.
- the phenyl group preferably has a substituted or unsubstituted condensed polycyclic aromatic group as a substituent, and is selected from a naphthyl group, anthracenyl group, phenanthrenyl group, pyrenyl group, fluoranthenyl group, and triphenylenyl group. It is more preferable to have a substituent.
- Ar 12 in the general formula (3) is preferably a phenyl group having a substituent.
- the substituent includes an aromatic hydrocarbon group such as a phenyl group, a biphenylyl group, and a terphenyl group, a naphthyl group, and an anthracenyl group.
- Ar 13 in the general formula (3) is preferably a phenyl group having a substituent.
- the substituent is an aromatic hydrocarbon group such as a phenyl group, a biphenylyl group or a terphenyl group, a naphthyl group, or an anthracenyl group.
- aromatic hydrocarbon group such as a phenyl group, a biphenylyl group or a terphenyl group, a naphthyl group, or an anthracenyl group.
- Group, acenaphthenyl group, phenanthrenyl group, fluorenyl group, indenyl group, pyrenyl group, perylenyl group, fluoranthenyl group, triphenylenyl group and other condensed polycyclic aromatic groups are preferable, phenyl group, naphthyl group, anthracenyl group, phenanthrenyl group, A pyrenyl group, a fluoranthenyl group, and a triphenylenyl group are more preferable.
- Ar 11 and Ar 12 are preferably not the same from the viewpoint of the stability of the thin film.
- Ar 11 and Ar 12 may be different substituents or different substitution positions.
- Ar 12 and Ar 13 may be the same group, but may be easily crystallized by improving the symmetry of the whole molecule.
- Ar 12 And Ar 13 are preferably different groups, and Ar 12 and Ar 13 are not simultaneously hydrogen atoms.
- Ar 14 in the general formula (3) is a triazinyl group, pyridyl group, pyrimidinyl group, pyrrolyl group, quinolyl group, isoquinolyl group, indolyl group, carbazolyl group, benzoxazolyl group, benzothiazolyl group, quinoxalinyl group, benzoimidazolyl group , Pyrazolyl group, naphthyridinyl group, phenanthrolinyl group, acridinyl group, carbolinyl group and other nitrogen-containing heterocyclic groups are preferable, triazinyl group, pyridyl group, pyrimidinyl group, quinolyl group, isoquinolyl group, indolyl group, quinoxalinyl group, benzoimidazolyl group Group, naphthyridinyl group, phenanthrolinyl group, and acridinyl group are more preferable, pyridyl group
- Examples of the compound having a pyrimidine ring structure represented by the general formula (3) include compounds having a pyrimidine ring structure represented by the following general formulas (3a) and (3b), which have different substituent bonding modes. .
- the arylamine compound represented by the general formula (1) or general formula (1a) that is preferably used in the organic EL device of the present invention is a hole injection layer, an electron blocking layer, or a hole transport layer of the organic EL device. It can be used as a constituent material. It is a compound that has a high hole mobility and is preferable as a material for the hole injection layer or the hole transport layer. In addition, it has a high electron blocking performance and is a preferred compound as a material for the electron blocking layer.
- the amine derivative having a condensed ring structure represented by the general formula (2) which is preferably used in the organic EL device of the present invention, can be used as a constituent material of the light emitting layer of the organic EL device. It is excellent in luminous efficiency as compared with conventional materials and is a preferred compound as a dopant material for the light emitting layer.
- the compound having a pyrimidine ring structure represented by the general formula (3) that is preferably used in the organic EL device of the present invention can be used as a constituent material of the electron transport layer of the organic EL device.
- the compound having a pyrimidine ring structure represented by the general formula (3) is excellent in electron injection and transport capability, and further excellent in stability and durability of a thin film, and is a preferable compound as a material for an electron transport layer. .
- the organic EL device of the present invention is a material for an organic EL device excellent in hole and electron injection / transport performance, thin film stability and durability, and matches the characteristics of the material of the light emitting layer having a specific structure.
- the hole transport efficiency from the hole transport layer to the light-emitting layer is improved, and the electron transport efficiency from the electron transport layer to the light-emitting layer is also improved.
- the light emission efficiency is improved and the driving voltage is lowered, so that the durability of the organic EL element can be improved. It has become possible to realize a long-life organic EL element with high luminous efficiency.
- the organic EL device of the present invention is excellent in hole and electron injection / transport performance, thin film stability and durability, and has an arylamine compound having a specific structure capable of effectively expressing the role of hole injection / transport By selecting, holes can be efficiently injected and transported to the light emitting layer, and an organic EL element with high efficiency, low drive voltage, and long life can be realized.
- an arylamine compound having a specific structure is selected and combined with a specific electron transport material so as to achieve a carrier balance that matches the characteristics of the material of the light-emitting layer having the specific structure, resulting in high efficiency and low driving voltage.
- a long-life organic EL element can be realized. According to the present invention, the luminous efficiency and durability of a conventional organic EL device can be improved.
- FIG. 3 is a diagram showing organic EL element configurations of Examples 14 to 15 and Comparative Examples 1 and 2.
- the amine derivative having the above-mentioned condensed ring structure can be synthesized according to a method known per se (see, for example, Patent Document 6).
- the compound having the pyrimidine ring structure described above can be synthesized according to a method known per se (see, for example, Patent Document 7).
- Purification of the arylamine compounds represented by the general formulas (1) and (1a) is performed by purification using column chromatography, adsorption purification using silica gel, activated carbon, activated clay, etc., recrystallization or crystallization using a solvent, sublimation purification, etc. went.
- the compound was identified by NMR analysis.
- melting point, glass transition point (Tg) and work function were measured.
- the melting point is an index of vapor deposition
- the glass transition point (Tg) is an index of stability in a thin film state
- the work function is an index of hole transportability and hole blocking property.
- the compound used in the organic EL device of the present invention is purified by column chromatography, purification by adsorption using silica gel, activated carbon, activated clay, etc., recrystallization using a solvent, crystallization method, sublimation purification method, etc. Finally, the product purified by the sublimation purification method was used.
- Tg Melting point and glass transition point (Tg) were measured with a high sensitivity differential scanning calorimeter (Bruker AXS, DSC3100SA) using powder.
- the work function was determined using an ionization potential measurement device (PYS-202, manufactured by Sumitomo Heavy Industries, Ltd.) after forming a 100 nm thin film on the ITO substrate.
- PYS-202 manufactured by Sumitomo Heavy Industries, Ltd.
- the substrate is composed of an anode, a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer, and a cathode sequentially on the substrate. And those having a hole blocking layer between the light emitting layer and the electron transport layer, and those having an electron injection layer between the electron transport layer and the cathode.
- a structure that serves as a hole injection layer and a hole transport layer or a structure that serves as an electron injection layer and an electron transport layer. And so on.
- the hole transport layer has a structure in which two layers of a first hole transport layer and a second hole transport layer are laminated.
- an electrode material having a large work function such as ITO or gold is used.
- the hole injection layer of the organic EL device of the present invention in addition to the arylamine compound represented by the general formula (1), materials such as starburst type triphenylamine derivatives and various triphenylamine tetramers; A porphyrin compound typified by copper phthalocyanine; an acceptor heterocyclic compound such as hexacyanoazatriphenylene, a coating-type polymer material, or the like can be used. These materials can be formed into a thin film by a known method such as a spin coating method or an ink jet method in addition to a vapor deposition method.
- the arylamine compound represented by the general formula (1) is used as the hole transport layer of the organic EL device of the present invention. These may be formed alone, or may be used as a single layer formed by mixing with other hole transporting materials, or the layers formed separately may be mixed to form a film. A stacked structure of layers formed by mixing with each other, or a layer formed by mixing with a single layer formed may be employed. These materials can be formed into a thin film by a known method such as a spin coating method or an ink jet method in addition to a vapor deposition method.
- N, N′-diphenyl-N, N′-di (m-tolyl) benzidine (TPD) is used.
- N, N′-diphenyl-N, N′-di ( ⁇ -naphthyl) benzidine (NPD) is used.
- N, N′-diphenyl-N, N′-di ( ⁇ -naphthyl) benzidine (NPD) is used.
- benzidine derivatives such as N, N, N ′, N′-tetrabiphenylylbenzidine, 1,1-bis [ 4- (di-4-tolylamino) phenyl] cyclohexane (TAPC), a triphenylamine derivative having two triphenylamine skeletons as a whole molecule, and a triphenylamine derivative having four triphen
- trisbromophenylamine hexachloroantimony, a radialene derivative (see, for example, International Publication No. 2014/009310) or the like is further added to the material normally used for the layer.
- a doped compound or a polymer compound having a partial structure of a benzidine derivative such as TPD can be used.
- These may be formed alone, but may be used as a single layer formed by mixing with other materials, layers formed alone, mixed layers formed, or A stacked structure of layers formed by mixing with a layer formed alone may be used.
- These materials can be formed into a thin film by a known method such as a spin coating method or an ink jet method in addition to the vapor deposition method.
- various amine complexes and pyrene derivatives having a condensed ring structure represented by the general formula (2) as well as various metal complexes of quinolinol derivatives such as Alq 3 can be used.
- Metal complexes, anthracene derivatives, bisstyrylbenzene derivatives, oxazole derivatives, polyparaphenylene vinylene derivatives, and the like can be used.
- the light-emitting layer may be composed of a host material and a dopant material.
- a thiazole derivative, a benzimidazole derivative, a polydialkylfluorene derivative, or the like can be used in addition to the light-emitting material.
- the dopant material in addition to amine derivatives and pyrene derivatives having a condensed ring structure represented by the general formula (2), quinacridone, coumarin, rubrene, perylene, and derivatives thereof, benzopyran derivatives, indenophenanthrene derivatives, Rhodamine derivatives, aminostyryl derivatives, and the like can be used.
- an amine derivative or pyrene derivative having a condensed ring structure represented by the general formula (2) is preferably used as a dopant material, and is represented by the general formula (2). It is more preferable to use an amine derivative having a condensed ring structure as a dopant material.
- a phosphorescent emitter As the phosphorescent emitter, a phosphorescent emitter of a metal complex such as iridium or platinum can be used. Green phosphorescent emitters such as Ir (ppy) 3 , blue phosphorescent emitters such as FIrpic and FIr6, red phosphorescent emitters such as Btp 2 Ir (acac), and the like are used as the host material. As the hole injecting / transporting host material, carbazole derivatives such as 4,4′-di (N-carbazolyl) biphenyl (CBP), TCTA, and mCP can be used.
- CBP 4,4′-di (N-carbazolyl) biphenyl
- CBP 4,4′-di (N-carbazolyl) biphenyl
- TCTA TCTA
- mCP mCP
- p-bis (triphenylsilyl) benzene (UGH2) and 2,2 ′, 2 ′′-(1,3,5-phenylene) -tris (1-phenyl-1H-benzimidazole) ) (TPBI) or the like, and a high-performance organic EL element can be manufactured.
- the phosphorescent light-emitting material into the host material by co-evaporation in the range of 1 to 30 weight percent with respect to the entire light-emitting layer.
- Non-Patent Document 3 a material that emits delayed fluorescence such as CDCB derivatives such as PIC-TRZ, CC2TA, PXZ-TRZ, and 4CzIPN as the light emitting material (see, for example, Non-Patent Document 3).
- CDCB derivatives such as PIC-TRZ, CC2TA, PXZ-TRZ, and 4CzIPN
- These materials can be formed into a thin film by a known method such as a spin coating method or an ink jet method in addition to a vapor deposition method.
- a phenanthroline derivative such as bathocuproine (BCP) or aluminum (III) bis (2-methyl-8-quinolinato) -4-phenylphenolate (hereinafter abbreviated as BAlq).
- BCP bathocuproine
- BAlq aluminum (III) bis (2-methyl-8-quinolinato) -4-phenylphenolate
- compounds having a hole blocking action such as various rare earth complexes, triazole derivatives, triazine derivatives, and oxadiazole derivatives can be used. These materials may also serve as the material for the electron transport layer.
- These may be formed alone, but may be used as a single layer formed by mixing with other materials, layers formed alone, mixed layers formed, or A stacked structure of layers formed by mixing with a layer formed alone may be used.
- These materials can be formed into a thin film by a known method such as a spin coating method or an ink jet method in addition to a vapor deposition method.
- a compound having a pyrimidine ring structure represented by the general formula (3) is preferably used. These may be formed alone, or may be used as a single layer formed by mixing with other electron transporting materials, or may be formed by mixing layers formed alone. A layered structure of layers formed by mixing layers with each other or a layer formed by itself may be used. These materials can be formed into a thin film by a known method such as a spin coating method or an ink jet method in addition to a vapor deposition method.
- Examples of the electron transporting material that can be mixed or simultaneously used with the compound having the pyrimidine ring structure represented by the general formula (3) include metal complexes of quinolinol derivatives such as Alq 3 and BAlq, various metal complexes, and triazoles. Derivatives, triazine derivatives, oxadiazole derivatives, pyridine derivatives, pyrimidine derivatives, benzimidazole derivatives, thiadiazole derivatives, anthracene derivatives, carbodiimide derivatives, quinoxaline derivatives, pyridoindole derivatives, phenanthroline derivatives, silole derivatives, and the like.
- an alkali metal salt such as lithium fluoride and cesium fluoride
- an alkaline earth metal salt such as magnesium fluoride
- a metal oxide such as aluminum oxide
- an electrode material having a low work function such as aluminum or an alloy having a lower work function such as a magnesium silver alloy, a magnesium indium alloy, or an aluminum magnesium alloy is used as the electrode material.
- the arylamine compound represented by the general formula (1) has a glass transition point of 100 ° C. or higher, which indicates that the thin film state is stable.
- a deposited film having a film thickness of 100 nm was prepared on the ITO substrate, and an ionization potential measuring device (PYS-202, manufactured by Sumitomo Heavy Industries, Ltd.) was used. The work function was measured. Work function Compound of Example 1 5.63 eV Compound of Example 2 5.62 eV Compound of Example 3 5.62 eV Compound of Example 4 5.65 eV Compound of Example 5 5.57 eV Compound of Example 6 5.56 eV Compound of Example 7 5.60 eV Compound of Example 8 5.70 eV Compound of Example 9 5.74 eV Compound of Example 10 5.79 eV Compound of Example 11 5.67 eV
- the arylamine compound represented by the general formula (1) exhibits a suitable energy level as compared with a work function of 5.4 eV possessed by a general hole transport material such as NPD or TPD. It can be seen that it has a hole transport capability.
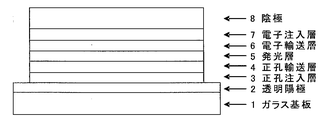
- the organic EL element has a hole injection layer 3, a hole transport layer 4, a light emitting layer 5, an electron transport layer on a glass substrate 1 on which an ITO electrode is previously formed as a transparent anode 2. 6, an electron injection layer 7 and a cathode (aluminum electrode) 8 were deposited in this order.
- the glass substrate 1 formed with ITO having a film thickness of 150 nm was subjected to ultrasonic cleaning in isopropyl alcohol for 20 minutes, and then dried on a hot plate heated to 200 ° C. for 10 minutes. Then, after performing UV ozone treatment for 15 minutes, this glass substrate 1 with ITO was attached in the vacuum evaporation machine, and pressure was reduced to 0.001 Pa or less. Subsequently, a compound (HIM-1) having the following structural formula was formed to a thickness of 5 nm as a hole injection layer 3 so as to cover the transparent anode 2. On the hole injection layer 3, the compound (1-7) of Example 5 was formed as the hole transport layer 4 so as to have a film thickness of 65 nm.
- Binary vapor deposition was performed at a vapor deposition rate of 95 to form a film thickness of 20 nm.
- a compound (3-125) having the following structural formula and a compound (ETM-1) having the following structural formula are deposited at a deposition rate ratio of 3-125:
- ETM-1 50: Binary vapor deposition was performed at a vapor deposition rate of 50 to form a film thickness of 30 nm.
- An organic EL element was produced under the same conditions except that the thickness was 20 nm. About the produced organic EL element, the characteristic measurement was performed at normal temperature in air
- Example 14 For comparison, in Example 14, a compound (HTM-1) having the following structural formula was used instead of the compound (1-7) of Example 5 as the material of the hole transport layer 4, so that the film thickness was 65 nm.
- An organic EL element was produced under the same conditions except that the above was formed. About the produced organic EL element, the characteristic measurement was performed at normal temperature in air
- Example 15 For comparison, in Example 15, the compound (HTM-1) having the above structural formula was used instead of the compound (1-7) of Example 5 as the material of the hole transport layer 4, so that the film thickness was 65 nm.
- An organic EL element was produced under the same conditions except that the above was formed. About the produced organic EL element, the characteristic measurement was performed at normal temperature in air
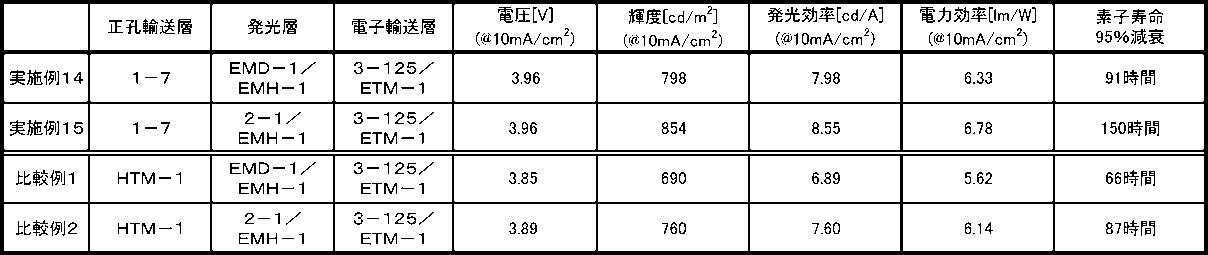
- Table 1 summarizes the results of measuring the device lifetime using the organic EL devices prepared in Examples 14 to 15 and Comparative Examples 1 and 2.
- the element lifetime corresponds to 95% of the emission brightness of 1900 cd / m 2 (when the initial brightness is 100%) when the constant current drive is performed with the emission brightness (initial brightness) at the start of light emission being 2000 cd / m 2 : It was measured as the time to decay to 95% decay.
- Example 14 in comparison between Example 14 and Comparative Example 1 in which the materials of the light emitting layer are the same combination, the luminance when a current having a current density of 10 mA / cm 2 was passed was the organic EL of Comparative Example 1 Compared to 690 cd / m 2 of the element, the organic EL element of Example 14 had a high luminance of 798 cd / m 2 . Further, the luminous efficiency when a current having a current density of 10 mA / cm 2 was passed was as high as 7.98 cd / A for the organic EL element of Example 14 compared to 6.89 cd / A for the organic EL element of Comparative Example 1. It was efficiency.
- the organic EL element of Example 14 was as high as 6.33 lm / W compared to 5.62 lm / W of the organic EL element of Comparative Example 1. Furthermore, in the element lifetime (95% attenuation), it can be seen that the organic EL element of Example 14 has a long lifetime of 91 hours compared to 66 hours of the organic EL element of Comparative Example 1. Similarly, in comparison between Example 15 and Comparative Example 2 in which the materials of the light emitting layer are the same combination, the luminance when a current having a current density of 10 mA / cm 2 was passed was 760 cd / d of the organic EL element of Comparative Example 2.
- the organic EL element of example 15 was a high luminance and 854cd / m 2.
- the luminous efficiency when a current density of 10 mA / cm 2 was passed was high at 8.55 cd / A in the organic EL element of Example 15 compared to 7.60 cd / A in the organic EL element in Comparative Example 2. It was efficiency.
- the power efficiency of the organic EL element of Example 15 was 6.78 lm / W, which was higher than that of the organic EL element of Comparative Example 2, which was 6.14 lm / W.
- the element lifetime (95% attenuation) it can be seen that the lifetime of the organic EL element of Example 15 is increased to 150 hours compared to 87 hours of the organic EL element of Comparative Example 2.
- the organic EL device of the present invention has a carrier balance inside the organic EL device by combining an arylamine compound having a specific structure and an amine derivative having a specific condensed ring structure (and a compound having a specific pyrimidine ring structure). Improved and combined with a carrier balance that matches the characteristics of the light-emitting material, it is possible to realize an organic EL element with higher luminous efficiency and longer life than conventional organic EL elements. I understood.
- the organic EL device of the present invention which combines an arylamine compound having a specific structure and an amine derivative having a specific condensed ring structure (and a compound having a specific pyrimidine ring structure), has improved luminous efficiency and organic EL.
- the durability of the element can be improved. For example, it can be applied to household appliances and lighting applications.
Abstract
Description
そして、熱活性化遅延蛍光(TADF)による発光を利用する素子も開発されている。2011年に九州大学の安達らは、熱活性化遅延蛍光材料を用いた素子によって5.3%の外部量子効率を実現させた(例えば、非特許文献3参照)。
ここで、Ar3とAr4は、単結合、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成してもよく、Ar3またはAr4は-NAr3Ar4基が結合しているベンゼン環と、単結合、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成してもよい。
そして、一般式(2)中のA1で表される「置換もしくは無置換の芳香族炭化水素の2価基」、「置換もしくは無置換の芳香族複素環の2価基」または「置換もしくは無置換の縮合多環芳香族の2価基」は、上記「芳香族炭化水素」、「芳香族複素環」または「縮合多環芳香族」から水素原子を2個取り除いてできる2価基を表す。
また、これらの2価基は置換基を有していてもよく、置換基として、前記一般式(1)、一般式(1a)中のAr1~Ar8で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(1)、一般式(1a)中のAr1~Ar8で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(2)中のR1~R7で表される「置換基を有する炭素原子数1ないし6の直鎖状もしくは分岐状のアルキル基」、「置換基を有する炭素原子数5ないし10のシクロアルキル基」または「置換基を有する炭素原子数2ないし6の直鎖状もしくは分岐状のアルケニル基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、一般式(2)中のR1~R7で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」としては、具体的に、重水素原子、シアノ基、ニトロ基;フッ素原子、塩素原子、臭素原子、ヨウ素原子などのハロゲン原子;メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基、イソブチル基、tert-ブチル基、n-ペンチル基、イソペンチル基、ネオペンチル基、n-ヘキシル基などの炭素原子数1ないし6の直鎖状もしくは分岐状のアルキル基;メチルオキシ基、エチルオキシ基、プロピルオキシ基などの炭素原子数1ないし6の直鎖状もしくは分岐状のアルキルオキシ基;ビニル基、アリル基などのアルケニル基;フェニルオキシ基、トリルオキシ基などのアリールオキシ基;ベンジルオキシ基、フェネチルオキシ基などのアリールアルキルオキシ基;フェニル基、ビフェニリル基、ターフェニリル基、ナフチル基、アントラセニル基、フェナントレニル基、フルオレニル基、インデニル基、ピレニル基、ペリレニル基、フルオランテニル基、トリフェニレニル基などの芳香族炭化水素基もしくは縮合多環芳香族基;ピリジル基、ピリミジニル基、トリアジニル基、チエニル基、フリル基、ピロリル基、キノリル基、イソキノリル基、ベンゾフラニル基、ベンゾチエニル基、インドリル基、カルバゾリル基、ベンゾオキサゾリル基、ベンゾチアゾリル基、キノキサリニル基、ベンゾイミダゾリル基、ピラゾリル基、ジベンゾフラニル基、ジベンゾチエニル基、カルボリニル基などの芳香族複素環基;スチリル基、ナフチルビニル基などのアリールビニル基;アセチル基、ベンゾイル基などのアシル基;トリメチルシリル基、トリフェニルシリル基などのシリル基;ジフェニルアミノ基、ジナフチルアミノ基などの芳香族炭化水素基もしくは縮合多環芳香族基で置換されたジ置換アミノ基;ジピリジルアミノ基、ジチエニルアミノ基などの芳香族複素環基で置換されたジ置換アミノ基;芳香族炭化水素基、縮合多環芳香族基もしくは芳香族複素環基から選択される置換基で置換されたジ置換アミノ基のような基をあげることができ、これらの置換基は、さらに前記例示した置換基が置換していてもよい。また、これらの置換基同士が単結合、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成していてもよい。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(2)中のR1~R7で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(2)中のR1~R7で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
一般式(2)中のR1~R4で表される「芳香族炭化水素基、芳香族複素環基もしくは縮合多環芳香族基から選ばれる基によって置換されたジ置換アミノ基」は、これらの基(R1~R4)同士が、これらの基(R1~R4)が有する「芳香族炭化水素基」、「芳香族複素環基」または「縮合多環芳香族基」を介しつつ、単結合、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成してもよく、これらの基(R1~R4)とこれらの基(R1~R4)が直接結合しているベンゼン環とで、これらの基(R1~R4)が有する「芳香族炭化水素基」、「芳香族複素環基」または「縮合多環芳香族基」を介しつつ、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成してもよい。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(2)中のR1~R7で表される「置換基を有する炭素原子数1ないし6の直鎖状もしくは分岐状のアルキル基」、「置換基を有する炭素原子数5ないし10のシクロアルキル基」、「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(2)中のR1~R7で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(2)中のR1~R7で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(2)中のR1~R7で表される「置換基を有する炭素原子数1ないし6の直鎖状もしくは分岐状のアルキル基」、「置換基を有する炭素原子数5ないし10のシクロアルキル基」、「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(1)、一般式(1a)中のAr1~Ar8で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(1)、一般式(1a)中のAr1~Ar8で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
また、これらの基は置換基を有していてもよく、置換基として、前記一般式(1)、一般式(1a)において、Ar1~Ar8で表される「置換芳香族炭化水素基」、「置換芳香族複素環基」または「置換縮合多環芳香族基」における「置換基」に関して示したものと同様のものをあげることができ、とりうる態様も、同様のものをあげることができる。
一般式(1)、一般式(1a)において、n1は0または1~2を表すが、n1が0の時は、2つのジアリールアミノベンゼン環が直接(単結合で)結合していることを示し、n1が1の時は、2つのジアリールアミノベンゼン環が1個のフェニレン基を介して結合していることを示し、n1が2の時は、2つのジアリールアミノベンゼン環が2個のフェニレン基(ビフェニレン基)を介して結合していることを示す。
一般式(1)、一般式(1a)において、n1は0であること、すなわち2つのジアリールアミノベンゼン環が直接(単結合で)結合していることが好ましい。
一般式(1)、一般式(1a)において、Ar3またはAr4が-NAr3Ar4基(Ar3、Ar4とそれらが結合する窒素原子からなるジアリールアミノ基)が結合しているベンゼン環と、単結合、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成する態様も好ましい。この場合のベンゼン環における結合位置としては、-NAr3Ar4基と隣接する位置が好ましい。
一般式(2)中のAr9、Ar10としては、フェニル基、ビフェニリル基、ナフチル基、フルオレニル基、インデニル基、ピリジル基、ジベンゾフラニル基、ピリドベンゾフラニル基が好ましい。
一般式(2)中のAr9とAr10は直接、またはそれらの基が有する置換基を介しつつ、単結合、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成してもよい。
一般式(2)中のR1~R4において、少なくともひとつが、「芳香族炭化水素基、芳香族複素環基もしくは縮合多環芳香族基から選ばれる基によって置換されたジ置換アミノ基」であることが好ましく、この場合の「芳香族炭化水素基」、「芳香族複素環基」、「縮合多環芳香族基」としては、フェニル基、ビフェニリル基、ナフチル基、フルオレニル基、インデニル基、ピリジル基、ジベンゾフラニル基、ピリドベンゾフラニル基が好ましい。
一般式(2)において、R1~R4のいずれかひとつが「芳香族炭化水素基」であって、R1~R4が結合しているベンゼン環と、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成する態様が好ましい。この場合の「芳香族炭化水素基」がフェニル基であって、R1~R4が結合しているベンゼン環と、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成する態様、すなわち、R1~R4が結合しているベンゼン環とともにジベンゾフラン環、またはジベンゾチオフェン環を形成する態様が特に好ましい。
一般式(2)において、R5~R7のいずれかひとつが「芳香族炭化水素基」であって、R5~R7が結合しているベンゼン環と、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成する態様が好ましい。この場合の「芳香族炭化水素基」がフェニル基であって、R5~R7が結合しているベンゼン環と、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成する態様、すなわちR5~R7が結合しているベンゼン環とともにジベンゾフラン環、またはジベンゾチオフェン環を形成する態様が特に好ましい。
以上のように、一般式(2)で表される縮合環構造を有するアミン誘導体の中で、R1~R7がこれらの基同士で互いに結合して環を形成する態様、または、R1~R7とR1~R7が結合しているベンゼン環とが互いに結合して環を形成する態様として、下記一般式(2a-a)、(2a-b)、(2b-a)、(2b-b)、(2b-c)、(2b-d)、(2c-a)または(2c-b)で表される態様が好ましく用いられる。
そして、R8とR9が、単結合、置換もしくは無置換のメチレン基、酸素原子、硫黄原子、一置換アミノ基などの連結基を介して互いに結合して環を形成する態様が好ましく、単結合を介して互いに結合して環を形成する態様が特に好ましい。
以上のように、一般式(2)で表される縮合環構造を有するアミン誘導体の中で、R8とR9が互いに結合して環を形成する態様として、下記一般式(2a-a1)、(2a-b1)、(2b-a1)、(2b-b1)、(2b-c1)、(2b-d1)、(2c-a1)または(2c-b1)で表される態様が好ましく用いられる。
一般式(3)中のAr12としては、置換基を有するフェニル基が好ましく、この場合の置換基としては、フェニル基、ビフェニリル基、ターフェニル基などの芳香族炭化水素基、ナフチル基、アントラセニル基、アセナフテニル基、フェナントレニル基、フルオレニル基、インデニル基、ピレニル基、ペリレニル基、フルオランテニル基、トリフェニレニル基などの縮合多環芳香族基が好ましく、フェニル基、ナフチル基、アントラセニル基、フェナントレニル基、ピレニル基、フルオランテニル基、トリフェニレニル基がより好ましい。
一般式(3)中のAr13としては、置換基を有するフェニル基が好ましく、この場合の置換基としては、フェニル基、ビフェニリル基、ターフェニル基などの芳香族炭化水素基、ナフチル基、アントラセニル基、アセナフテニル基、フェナントレニル基、フルオレニル基、インデニル基、ピレニル基、ペリレニル基、フルオランテニル基、トリフェニレニル基などの縮合多環芳香族基が好ましく、フェニル基、ナフチル基、アントラセニル基、フェナントレニル基、ピレニル基、フルオランテニル基、トリフェニレニル基がより好ましい。
一般式(3)において、Ar11とAr12が同一ではないことが、薄膜の安定性の観点から好ましい。ここで、Ar11とAr12が同一ではない場合とは、異なる置換基であってもよいし、または、異なる置換位置であってもよい。
一般式(3)において、Ar12とAr13は同一の基であってもよいが、分子全体の対称性がよくなることによって結晶化し易くなる恐れがあり、薄膜の安定性の観点から、Ar12とAr13は異なる基であることが好ましく、Ar12とAr13が同時に水素原子とはなることはないものとする。
一般式(3)において、Ar12とAr13の一方が水素原子であることが好ましい。
一般式(3)において、ベンゼン環におけるAr14の結合位置は、ピリミジン環との結合位置に対し、メタ位であることが、薄膜の安定性の観点から好ましい。
前記一般式(3)で表される、ピリミジン環構造を有する化合物は、電子注入および輸送能力に優れており、さらに薄膜の安定性や耐久性に優れ、電子輸送層の材料として好ましい化合物である。
高発光効率であって、長寿命の有機EL素子を実現することが可能となった。
本発明によれば、従来の有機EL素子の発光効率および耐久性を改良することができる。
その他、本発明の有機EL素子に用いられる化合物は、カラムクロマトグラフによる精製、シリカゲル、活性炭、活性白土などによる吸着精製、溶媒による再結晶や晶析法、昇華精製法などによって精製を行った後、最後に昇華精製法によって精製したものを用いた。
前記一般式(1)で表されるアリールアミン化合物と混合または同時に使用できる、正孔輸送性の材料としては、N,N’-ジフェニル-N,N’-ジ(m-トリル)ベンジジン(TPD)、N,N’-ジフェニル-N,N’-ジ(α-ナフチル)ベンジジン(NPD)、N,N,N’,N’-テトラビフェニリルベンジジンなどのベンジジン誘導体、1,1-ビス[4-(ジ-4-トリルアミノ)フェニル]シクロヘキサン(TAPC)、分子全体としてトリフェニルアミン骨格を2個有しているトリフェニルアミン誘導体、分子全体としてトリフェニルアミン骨格を4個有しているトリフェニルアミン誘導体、分子全体としてトリフェニルアミン骨格を3個有しているトリフェニルアミン誘導体などをあげることができる。
本発明の有機EL素子の発光層としては、前記一般式(2)で表される縮合環構造を有するアミン誘導体またはピレン誘導体をドーパント材料として用いることが好ましく、前記一般式(2)で表される縮合環構造を有するアミン誘導体をドーパント材料として用いることがより好ましい。
前記一般式(3)で表されるピリミジン環構造を有する化合物と混合または同時に使用できる、電子輸送性の材料としては、Alq3、BAlqをはじめとするキノリノール誘導体の金属錯体、各種金属錯体、トリアゾール誘導体、トリアジン誘導体、オキサジアゾール誘導体、ピリジン誘導体、ピリミジン誘導体、ベンズイミダゾール誘導体、チアジアゾール誘導体、アントラセン誘導体、カルボジイミド誘導体、キノキサリン誘導体、ピリドインドール誘導体、フェナントロリン誘導体、シロール誘導体などをあげることができる。
窒素置換した反応容器に、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミン10.0g、4-{(ビフェニル-4-イル)-フェニルアミノ}フェニルボロン酸7.9g、テトラキストリフェニルホスフィンパラジウム(0)0.60g、炭酸カリウム5.0g、トルエン80ml、エタノール40ml、水30mlを加えて加熱し、100℃で一晩撹拌した。冷却し、分液操作により有機層を採取した後、濃縮し、カラムクロマトグラフ(担体:シリカゲル、溶離液:ジクロロメタン/ヘプタン)によって精製することにより、4-ビス(ビフェニル-4-イル)アミノ-4’-{(ビフェニル-4-イル)-フェニルアミノ}-2-フェニル-ビフェニル(化合物1-1)の白色粉体5.30g(収率37%)を得た。
1H-NMR(CDCl3)で以下の44個の水素のシグナルを検出した。
δ(ppm)=7.65-7.60(5H)、7.59-7.53(5H)、7.52-7.40(9H)、7.39-7.21(15H)、7.20-7.10(5H)、7.90-6.91(5H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)-(4-ナフタレン-1-イル-フェニル)アミンを用い、同様の条件で反応を行うことによって4-{(ビフェニル-4-イル)-(4-ナフタレン-1-イル-フェニル)アミノ}-4’-{(ビフェニル-4-イル)-フェニルアミノ}-2-フェニル-ビフェニル(化合物1-3)の類白色粉体9.70g(収率69%)を得た。
1H-NMR(CDCl3)で以下の46個の水素のシグナルを検出した。
δ(ppm)= 8.07(1H)、7.93(1H)、7.87(1H)、7.67-7.54(7H)、7.54-7.11(31H)、7.69-6.92(5H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)-(p-ターフェニル-4-イル)アミンを用い、同様の条件で反応を行うことによって4-{(ビフェニル-4-イル)-(p-ターフェニル-4-イル)アミノ}-4’-{(ビフェニル-4-イル)-フェニルアミノ}-2-フェニル-ビフェニル(化合物1-5)の白色粉体6.76g(収率57%)を得た。
1H-NMR(CDCl3)で以下の48個の水素のシグナルを検出した。
δ(ppm)=7.71-7.10(43H)、7.08-6.93(5H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、ビス(4-ナフタレン-1-イル-フェニル)-(6-ブロモ-ビフェニル-3-イル)アミンを用い、同様の条件で反応を行うことによって4-{(ビフェニル-4-イル)-フェニルアミノ}-4’-{ビス(4-ナフタレン-1-イル-フェニル)アミノ}-2’-フェニル-ビフェニル(化合物1-6)の黄白色粉体10.0g(収率73%)を得た。
1H-NMR(CDCl3)で以下の48個の水素のシグナルを検出した。
δ(ppm)=8.08(1H)、7.94(1H)、7.88(1H)、7.63-7.20(40H)、7.19-6.92(5H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、(9,9-ジメチルフルオレン-2-イル)-(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンを用い、同様の条件で反応を行うことによって4-{(9,9-ジメチルフルオレン-2-イル)-(ビフェニル-4-イル)アミノ}-4’-(ビフェニル-4-イル-フェニルアミノ)-2-フェニル-ビフェニル(化合物1-7)の白色粉体8.30g(収率49%)を得た。
1H-NMR(CDCl3)で以下の48個の水素のシグナルを検出した。
δ(ppm)=7.72-7.60(2H)、7.59-7.52(2H)、7.51-7.10(35H)、7.09-6.90(3H)、1.56(6H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、(9,9-ジメチルフルオレン-2-イル)-(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンを用い、4-{(ビフェニル-4-イル)-フェニルアミノ}フェニルボロン酸に代えて(9-フェニルカルバゾール-3-イル)ボロン酸を用い同様の条件で反応を行うことによって4-{(9,9-ジメチルフルオレン-2-イル)-(ビフェニル-4-イル)アミノ}-4’-(9-フェニルカルバゾール-3-イル)-2-フェニル-ビフェニル(化合物1-8)の白色粉体17.4g(収率85%)を得た。
1H-NMR(CDCl3)で以下の42個の水素のシグナルを検出した。
δ(ppm)=8.05(2H)、7.72-7.10(34H)、1.52(6H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、(6-ブロモ-1,1’-ビフェニル-3-イル)-{(ナフタレン-2-イル)フェニル-4-イル}(ビフェニル-4-イル)アミンを用い同様の条件で反応を行うことによって4-{(ナフタレン-2-イル)フェニル-4-イル}(ビフェニル-4-イル)アミノ-4’-{(ビフェニル-4-イル)-フェニルアミノ}-2-フェニル-1,1’-ビフェニル(化合物1-4)の白色粉体6.1g(収率58%)を得た。
1H-NMR(CDCl3)で以下の46個の水素のシグナルを検出した。
δ(ppm)=8.07(1H)、7.95-7.76(4H)、7.68-6.98(41H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、(6-ブロモ-1,1’-ビフェニル-3-イル)-(1,1’-ビフェニル-4-イル)フェニルアミンを用い同様の条件で反応を行うことによって4,4’’-ビス{(ビフェニル-4-イル)-フェニルアミノ}-2-フェニル-1,1’;4’,1’’-ターフェニル(化合物1-19)の白色粉体12.9g(収率43%)を得た。
1H-NMR(CDCl3)で以下の44個の水素のシグナルを検出した。
δ(ppm)=7.65-7.61(4H)、7.57-7.07(40H)。
窒素置換した反応容器に、4,4’’-ビス{(ビフェニル-4-イル)-アミノ}-3,3’’-ジフェニル-1,1’;4’,1’’-ターフェニル16.3g、ヨードベンゼン18.6g、銅粉0.29g、炭酸カリウム9.61g、3,5-ジ-tert-ブチルサリチル酸1.85g、亜硫酸水素ナトリウム0.47g、ドデシルベンゼン20mlを加えて加熱し、190~200℃で17時間撹拌した。冷却し、トルエン1500ml、シリカゲル40g、活性白土20gを添加した後、撹拌した。ろ過によって不溶物を除いた後濃縮し、クロロベンゼンを用いた再結晶を繰り返すことによって、4,4’’-ビス{(ビフェニル-4-イル)-フェニルアミノ}-3,3’’-ジフェニル-1,1’;4’,1’’-ターフェニル(化合物1-27)の白色粉体9.65g(収率49%)を得た。
1H-NMR(CDCl3)で以下の48個の水素のシグナルを検出した。
δ(ppm)=7.62(4H)、7.52(4H)、7.45(4H)、7.36-7.04(32H)、6.99(4H)。
窒素置換した反応容器に、4-{(ビフェニル-4-イル)-フェニルアミノ}-4’’-{(ビフェニル-4-イル)-アミノ}-3-フェニル-1,1’;3’,1’’-ターフェニル17.0g、ブロモベンゼン4.12g、酢酸パラジウム0.13g、トリ-tert-ブチルホスフィンの50%(w/v)トルエン溶液0.33ml、tert-ブトキシナトリウム2.73g、トルエン190mlを加えて加熱し、80℃で3時間撹拌した。冷却し、ろ過によって不溶物を除いた後濃縮し、カラムクロマトグラフ(担体:シリカゲル、溶離液:トルエン/n-ヘキサン)によって精製した。アセトンを加えることによって析出する固体を集めることによって、4,4’’-ビス{(ビフェニル-4-イル)-フェニルアミノ}-3-フェニル-1,1’;3’,1’’-ターフェニル(化合物1-30)の白色粉体13.29g(収率71%)を得た。
1H-NMR(CDCl3)で以下の44個の水素のシグナルを検出した。
δ(ppm)=7.62-7.58(4H)、7.55-7.49(4H)、7.48-7.38(6H)、7.37-7.05(30H)。
実施例1において、ビス(ビフェニル-4-イル)-(6-ブロモビフェニル-3-イル)アミンに代えて、4-ビス(ビフェニル-4-イル)アミノ-2,6-ジフェニル-ブロモベンゼンを用い、同様の条件で反応を行うことによって4-ビス(ビフェニル-4-イル)アミノ-4’-{(ビフェニル-4-イル)-フェニルアミノ)}-2,6-ジフェニル-ビフェニル(化合物1-41)の白色粉体12.7g(収率57%)を得た。
1H-NMR(CDCl3)で以下の48個の水素のシグナルを検出した。
δ(ppm)=7.65-7.53(8H)、7.48-6.97(36H)、6.79-6.73(4H)。
融点 ガラス転移点
実施例1の化合物 観測されず 118℃
実施例2の化合物 観測されず 121℃
実施例3の化合物 観測されず 125℃
実施例4の化合物 観測されず 125℃
実施例5の化合物 観測されず 125℃
実施例6の化合物 観測されず 139℃
実施例7の化合物 観測されず 121℃
実施例8の化合物 観測されず 120℃
実施例9の化合物 263℃ 124℃
実施例10の化合物 観測されず 117℃
実施例11の化合物 238℃ 126℃
仕事関数
実施例1の化合物 5.63eV
実施例2の化合物 5.62eV
実施例3の化合物 5.62eV
実施例4の化合物 5.65eV
実施例5の化合物 5.57eV
実施例6の化合物 5.56eV
実施例7の化合物 5.60eV
実施例8の化合物 5.70eV
実施例9の化合物 5.74eV
実施例10の化合物 5.79eV
実施例11の化合物 5.67eV
比較のために、実施例14において、正孔輸送層4の材料として実施例5の化合物(1-7)に代えて下記構造式の化合物(HTM-1)を用い、膜厚65nmとなるように形成した以外は同様の条件で有機EL素子を作製した。作製した有機EL素子について、大気中、常温で特性測定を行った。作製した有機EL素子に直流電圧を印加したときの発光特性の測定結果を表1にまとめて示した。
比較のために、実施例15において、正孔輸送層4の材料として実施例5の化合物(1-7)に代えて上記構造式の化合物(HTM-1)を用い、膜厚65nmとなるように形成した以外は同様の条件で有機EL素子を作製した。作製した有機EL素子について、大気中、常温で特性測定を行った。作製した有機EL素子に直流電圧を印加したときの発光特性の測定結果を表1にまとめて示した。
同様に、発光層の材料が同じ組合せである実施例15と比較例2との比較において、電流密度10mA/cm2の電流を流したときの輝度は、比較例2の有機EL素子の760cd/m2に対し、実施例15の有機EL素子では854cd/m2と高輝度であった。また、電流密度10mA/cm2の電流を流したときの発光効率は、比較例2の有機EL素子の7.60cd/Aに対し、実施例15の有機EL素子では8.55cd/Aと高効率であった。また、電力効率においても、比較例2の有機EL素子の6.14lm/Wに対し、実施例15の有機EL素子では6.78lm/Wと高効率であった。さらに、素子寿命(95%減衰)においては、比較例2の有機EL素子の87時間に対し、実施例15の有機EL素子では150時間と長寿命化していることが分かる。
2 透明陽極
3 正孔注入層
4 正孔輸送層
5 発光層
6 電子輸送層
7 電子注入層
8 陰極
Claims (8)
- 少なくとも陽極、正孔輸送層、発光層、電子輸送層および陰極をこの順に有する有機エレクトロルミネッセンス素子において、前記正孔輸送層が下記一般式(1)で表されるアリールアミン化合物を含有することを特徴とする有機エレクトロルミネッセンス素子。
- 前記アリールアミン化合物が、下記一般式(1a)で表されるアリールアミン化合物である、請求項1記載の有機エレクトロルミネッセンス素子。
- 前記発光層が、青色発光性ドーパントを含有する、請求項1記載の有機エレクトロルミネッセンス素子。
- 前記青色発光性ドーパントが、ピレン誘導体である、請求項3記載の有機エレクトロルミネッセンス素子。
- 前記青色発光性ドーパントが、下記一般式(2)で表される縮合環構造を有するアミン誘導体である、請求項3記載の有機エレクトロルミネッセンス素子。
- 前記電子輸送層が、下記一般式(3)で表されるピリミジン環構造を有する化合物を含有する、請求項1~5のいずれか一項に記載の有機エレクトロルミネッセンス素子。
- 前記発光層が、アントラセン誘導体を含有する、請求項1~6のいずれか一項に記載の有機エレクトロルミネッセンス素子。
- 前記発光層が、アントラセン誘導体であるホスト材料を含有する、請求項7記載の有機エレクトロルミネッセンス素子。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017561204A JP6810710B2 (ja) | 2016-01-14 | 2017-01-13 | 有機エレクトロルミネッセンス素子 |
| CN201780006469.8A CN108475737B (zh) | 2016-01-14 | 2017-01-13 | 有机电致发光元件 |
| EP17738565.5A EP3404735B1 (en) | 2016-01-14 | 2017-01-13 | Organic electroluminescent device |
| US16/069,218 US20190006596A1 (en) | 2016-01-14 | 2017-01-13 | Organic electroluminescent device |
| KR1020187022569A KR20180102113A (ko) | 2016-01-14 | 2017-01-13 | 유기 일렉트로루미네선스 소자 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016-004843 | 2016-01-14 | ||
| JP2016004843 | 2016-01-14 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017122813A1 true WO2017122813A1 (ja) | 2017-07-20 |
Family
ID=59311249
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/001112 WO2017122813A1 (ja) | 2016-01-14 | 2017-01-13 | 有機エレクトロルミネッセンス素子 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20190006596A1 (ja) |
| EP (1) | EP3404735B1 (ja) |
| JP (1) | JP6810710B2 (ja) |
| KR (1) | KR20180102113A (ja) |
| CN (1) | CN108475737B (ja) |
| TW (1) | TWI740881B (ja) |
| WO (1) | WO2017122813A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107652223A (zh) * | 2017-10-10 | 2018-02-02 | 上海道亦化工科技有限公司 | 一种咔唑类有机电致发光化合物及其有机电致发光器件 |
| WO2019054233A1 (ja) * | 2017-09-13 | 2019-03-21 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2019059334A1 (ja) * | 2017-09-25 | 2019-03-28 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102002031B1 (ko) * | 2015-06-12 | 2019-07-23 | 에스에프씨주식회사 | 고효율을 갖는 유기 발광 소자 |
| JP7417350B2 (ja) * | 2017-03-28 | 2024-01-18 | キヤノン株式会社 | 光学素子、光学材料、光学機器及びトリアリールアミン化合物 |
| EP4095216A4 (en) * | 2020-01-22 | 2024-04-03 | Hodogaya Chemical Co Ltd | ORGANIC ELECTROLUMINESCENT ELEMENT |
| CN117730636A (zh) | 2021-09-07 | 2024-03-19 | 保土谷化学工业株式会社 | 有机电致发光元件 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002356462A (ja) * | 2001-03-30 | 2002-12-13 | Konica Corp | アリールベンジジン誘導体化合物、有機エレクトロルミネッセンス素子材料、および有機エレクトロルミネッセンス素子 |
| JP2004253298A (ja) * | 2003-02-21 | 2004-09-09 | Konica Minolta Holdings Inc | 白色発光有機エレクトロルミネッセンス素子 |
| US20050236976A1 (en) * | 2003-12-31 | 2005-10-27 | Ritdisplay Corporation | Organic electroluminescent device |
| KR20110084798A (ko) * | 2010-01-18 | 2011-07-26 | 덕산하이메탈(주) | 다양한 치환기를 갖는 방향족 아민 화합물 및 이를 이용한 유기전자소자, 그 단말 |
| JP2013080961A (ja) * | 2009-04-24 | 2013-05-02 | Idemitsu Kosan Co Ltd | 芳香族アミン誘導体及びアントラセン誘導体を用いた有機エレクトロルミネッセンス素子 |
| KR20150130206A (ko) * | 2014-05-13 | 2015-11-23 | 에스에프씨 주식회사 | 방향족 아민기를 포함하는 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| CN105566123A (zh) * | 2014-10-13 | 2016-05-11 | 江苏和成新材料有限公司 | 芳香族胺衍生物及其有机电致发光器件 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS4943840A (ja) | 1973-07-06 | 1974-04-25 | ||
| JP3194657B2 (ja) | 1993-11-01 | 2001-07-30 | 松下電器産業株式会社 | 電界発光素子 |
| JP3828595B2 (ja) | 1994-02-08 | 2006-10-04 | Tdk株式会社 | 有機el素子 |
| US6998487B2 (en) * | 2001-04-27 | 2006-02-14 | Lg Chem, Ltd. | Double-spiro organic compounds and organic electroluminescent devices using the same |
| KR100787425B1 (ko) | 2004-11-29 | 2007-12-26 | 삼성에스디아이 주식회사 | 페닐카바졸계 화합물 및 이를 이용한 유기 전계 발광 소자 |
| JP2007126439A (ja) * | 2005-10-07 | 2007-05-24 | Toyo Ink Mfg Co Ltd | カルバゾール含有アミン化合物およびその用途 |
| WO2007043484A1 (ja) * | 2005-10-07 | 2007-04-19 | Toyo Ink Manufacturing Co., Ltd. | カルバゾール含有アミン化合物及びその用途 |
| KR101370183B1 (ko) | 2006-11-24 | 2014-03-05 | 이데미쓰 고산 가부시키가이샤 | 방향족 아민 유도체 및 그것을 이용한 유기 전기발광 소자 |
| KR20100007639A (ko) * | 2008-07-14 | 2010-01-22 | 덕산하이메탈(주) | 신규한 페닐-플루오렌 유도체 및 이를 포함하는 유기전계발광 소자 |
| KR102013399B1 (ko) | 2011-11-29 | 2019-08-22 | 에스에프씨 주식회사 | 안트라센 유도체 및 이를 포함하는 유기전계발광소자 |
| JP5749870B1 (ja) * | 2013-07-03 | 2015-07-15 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| KR102137429B1 (ko) * | 2013-07-11 | 2020-07-24 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물을 이용한 유기전기소자 및 그 전자 장치 |
| KR102072756B1 (ko) * | 2013-09-17 | 2020-02-03 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물을 이용한 유기전기소자 및 그 전자 장치 |
| JP2016111099A (ja) * | 2014-12-03 | 2016-06-20 | 三星ディスプレイ株式會社Samsung Display Co.,Ltd. | 有機電界発光素子 |
-
2017
- 2017-01-13 WO PCT/JP2017/001112 patent/WO2017122813A1/ja active Application Filing
- 2017-01-13 CN CN201780006469.8A patent/CN108475737B/zh active Active
- 2017-01-13 JP JP2017561204A patent/JP6810710B2/ja active Active
- 2017-01-13 US US16/069,218 patent/US20190006596A1/en active Pending
- 2017-01-13 EP EP17738565.5A patent/EP3404735B1/en active Active
- 2017-01-13 KR KR1020187022569A patent/KR20180102113A/ko not_active Application Discontinuation
- 2017-01-16 TW TW106101328A patent/TWI740881B/zh active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002356462A (ja) * | 2001-03-30 | 2002-12-13 | Konica Corp | アリールベンジジン誘導体化合物、有機エレクトロルミネッセンス素子材料、および有機エレクトロルミネッセンス素子 |
| JP2004253298A (ja) * | 2003-02-21 | 2004-09-09 | Konica Minolta Holdings Inc | 白色発光有機エレクトロルミネッセンス素子 |
| US20050236976A1 (en) * | 2003-12-31 | 2005-10-27 | Ritdisplay Corporation | Organic electroluminescent device |
| JP2013080961A (ja) * | 2009-04-24 | 2013-05-02 | Idemitsu Kosan Co Ltd | 芳香族アミン誘導体及びアントラセン誘導体を用いた有機エレクトロルミネッセンス素子 |
| KR20110084798A (ko) * | 2010-01-18 | 2011-07-26 | 덕산하이메탈(주) | 다양한 치환기를 갖는 방향족 아민 화합물 및 이를 이용한 유기전자소자, 그 단말 |
| KR20150130206A (ko) * | 2014-05-13 | 2015-11-23 | 에스에프씨 주식회사 | 방향족 아민기를 포함하는 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| CN105566123A (zh) * | 2014-10-13 | 2016-05-11 | 江苏和成新材料有限公司 | 芳香族胺衍生物及其有机电致发光器件 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3404735A4 * |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3683852A4 (en) * | 2017-09-13 | 2021-06-23 | Hodogaya Chemical Co., Ltd. | ORGANIC ELECTROLUMINESCENT ELEMENT |
| WO2019054233A1 (ja) * | 2017-09-13 | 2019-03-21 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| CN111095588A (zh) * | 2017-09-13 | 2020-05-01 | 保土谷化学工业株式会社 | 有机电致发光元件 |
| JPWO2019054233A1 (ja) * | 2017-09-13 | 2020-08-27 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| TWI790280B (zh) * | 2017-09-13 | 2023-01-21 | 日商保土谷化學工業股份有限公司 | 有機電致發光元件 |
| US11578065B2 (en) | 2017-09-13 | 2023-02-14 | Hodogaya Chemical Co., Ltd. | Organic electroluminescence device |
| JP7227141B2 (ja) | 2017-09-13 | 2023-02-21 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| CN111095588B (zh) * | 2017-09-13 | 2023-11-17 | 保土谷化学工业株式会社 | 有机电致发光元件 |
| WO2019059334A1 (ja) * | 2017-09-25 | 2019-03-28 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| JPWO2019059334A1 (ja) * | 2017-09-25 | 2020-11-05 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| US11605785B2 (en) | 2017-09-25 | 2023-03-14 | Hodogaya Chemical Co., Ltd. | Organic electroluminescence device |
| JP7250683B2 (ja) | 2017-09-25 | 2023-04-03 | 保土谷化学工業株式会社 | 有機エレクトロルミネッセンス素子 |
| CN107652223A (zh) * | 2017-10-10 | 2018-02-02 | 上海道亦化工科技有限公司 | 一种咔唑类有机电致发光化合物及其有机电致发光器件 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN108475737B (zh) | 2020-12-01 |
| US20190006596A1 (en) | 2019-01-03 |
| TW201736337A (zh) | 2017-10-16 |
| JPWO2017122813A1 (ja) | 2018-11-08 |
| JP6810710B2 (ja) | 2021-01-06 |
| KR20180102113A (ko) | 2018-09-14 |
| CN108475737A (zh) | 2018-08-31 |
| TWI740881B (zh) | 2021-10-01 |
| EP3404735B1 (en) | 2023-05-10 |
| EP3404735A1 (en) | 2018-11-21 |
| EP3404735A4 (en) | 2019-08-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6329937B2 (ja) | 有機エレクトロルミネッセンス素子 | |
| JP6815326B2 (ja) | 有機エレクトロルミネッセンス素子 | |
| JP7406486B2 (ja) | トリアリールアミン構造を有する化合物および有機エレクトロルミネッセンス素子 | |
| JP6901977B2 (ja) | 有機エレクトロルミネッセンス素子 | |
| JP6735743B2 (ja) | アリールアミン化合物および有機エレクトロルミネッセンス素子 | |
| JP2015155378A (ja) | トリフェニレン環構造を有する化合物および有機エレクトロルミネッセンス素子 | |
| WO2017122813A1 (ja) | 有機エレクトロルミネッセンス素子 | |
| JP2011178742A (ja) | フェノキサジン環構造またはフェノチアジン環構造を有する化合物および有機エレクトロルミネッセンス素子 | |
| JP7018895B2 (ja) | 有機エレクトロルミネッセンス素子 | |
| JP6251675B2 (ja) | アクリダン環構造を有する化合物および有機エレクトロルミネッセンス素子 | |
| WO2019159919A1 (ja) | 有機エレクトロルミネッセンス素子 | |
| WO2020184219A1 (ja) | 有機エレクトロルミネッセンス素子 | |
| WO2017033927A1 (ja) | 有機エレクトロルミネッセンス素子 | |
| TW201920126A (zh) | 含有具有苯并咪唑環結構的化合物之有機電致發光元件 | |
| JP7149263B2 (ja) | 有機エレクトロルミネッセンス素子 | |
| JP7481268B2 (ja) | 有機エレクトロルミネッセンス素子 | |
| TW201805292A (zh) | 具有苯并唑環結構之化合物及有機電致發光元件 | |
| JP2016086147A (ja) | 有機エレクトロルミネッセンス素子 | |
| WO2014042006A1 (ja) | 新規なチエノインドール誘導体及び該誘導体を用いた有機エレクトロルミネッセンス素子 | |
| WO2021149735A1 (ja) | アリールアミン化合物および有機エレクトロルミネッセンス素子 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17738565 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017561204 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20187022569 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020187022569 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2017738565 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2017738565 Country of ref document: EP Effective date: 20180814 |