WO2015100873A1 - 无铅无铋无硅黄铜 - Google Patents
无铅无铋无硅黄铜 Download PDFInfo
- Publication number
- WO2015100873A1 WO2015100873A1 PCT/CN2014/074942 CN2014074942W WO2015100873A1 WO 2015100873 A1 WO2015100873 A1 WO 2015100873A1 CN 2014074942 W CN2014074942 W CN 2014074942W WO 2015100873 A1 WO2015100873 A1 WO 2015100873A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- free
- alloy
- lead
- silicon
- brass
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C9/00—Alloys based on copper
- C22C9/04—Alloys based on copper with zinc as the next major constituent
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22D—CASTING OF METALS; CASTING OF OTHER SUBSTANCES BY THE SAME PROCESSES OR DEVICES
- B22D21/00—Casting non-ferrous metals or metallic compounds so far as their metallurgical properties are of importance for the casting procedure; Selection of compositions therefor
- B22D21/002—Castings of light metals
- B22D21/005—Castings of light metals with high melting point, e.g. Be 1280 degrees C, Ti 1725 degrees C
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C1/00—Making non-ferrous alloys
- C22C1/02—Making non-ferrous alloys by melting
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22F—CHANGING THE PHYSICAL STRUCTURE OF NON-FERROUS METALS AND NON-FERROUS ALLOYS
- C22F1/00—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working
- C22F1/08—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working of copper or alloys based thereon
Definitions
- the present invention relates to an environmentally friendly brass alloy, and more particularly to a brass alloy material that is easy to cut and dezincification resistant.
- silicon appears in the Y phase of the alloy metallographic structure (sometimes a ⁇ phase), and at this time, silicon can replace the role of lead in the alloy to some extent, and improve the machinability of the alloy.
- the machinability of the alloy increases with the increase of silicon content, but the melting point of silicon is high, the specific gravity is low, and it is easy to be oxidized. Therefore, after the silicon monomer is added into the furnace during the melting of the alloy, silicon floats on the surface of the alloy, when the alloy is molten. Silicon is oxidized to silicon oxide or other oxides, making it difficult to produce a copper-containing copper alloy, and if silicon is added as a Cu-Si alloy, the economic cost is high.
- niobium to lead can be used as a cutting break in the alloy structure to increase the machinability.
- the niobium content is too high, hot cracking is likely to occur during forging, which is not conducive to production.
- the object of the present invention is to provide a brass alloy excellent in tensile strength, elongation, dezincification resistance and machinability, and is suitable as a processed product requiring high strength and abrasion resistance, and forged products and A constituent material such as a cast product is used. It can safely replace alloy copper containing a large amount of lead, and fully meets the requirements of human society for the restriction of lead-containing products.
- a lead-free, silicon-free, silicon-free machinable brass alloy (hereinafter referred to as Invention 1), comprising: 60-65 wt% copper, 0.01-0.15 wt% bismuth and 0.1-0.5 wt% of the total weight of the brass alloy % magnesium, the remainder being zinc.
- the present invention 1 controls the copper content to be 60-65 wt%, and adds a small amount of bismuth and magnesium to form an intermetallic compound with copper to increase the machinability of the alloy. It also contributes to the dezincification resistance of the alloy. In other words, the addition of yttrium and magnesium to the Y phase of Invention 1 improves the machinability.
- the metallographic structure of the alloy mainly includes ⁇ phase, ⁇ phase, Y phase, and soft and brittle intermetallic compounds distributed in the grain boundary or grain, wherein copper and zinc constitute the main component of the brass alloy, and the addition of bismuth and In addition to improving the machinability of the alloy, magnesium also contributes to dezincification resistance.
- the formed alloy cannot achieve the basic machinability required in industrial production.
- the machinability of the alloy increases with the increase of the content of niobium and magnesium, but when the niobium content in the alloy is 0.15 wt% and the magnesium content is 0.5%, The improvement of the machinability of gold is saturated.
- a lead-free, silicon-free, silicon-free machinable brass alloy (hereinafter referred to as Invention 2), comprising: 60-65 wt% of copper, based on the total weight of the brass alloy, 0.01-0.15 wt% of bismuth, 0.1-0.5 wt. % of magnesium, and 0.05 to 0.3% by weight of phosphorus and/or 0.05 to 0.5% by weight of manganese, based on the total weight of the brass alloy, the remainder being zinc.
- the inventive article 2 further adds 0.05 to 0.3% by weight of phosphorus and/or 0.05 to 0.5% by weight of manganese based on the total weight of the brass alloy on the basis of the invention 1.
- phosphorus cannot form the Y phase, phosphorus has a function of forming a Y phase distribution of bismuth and magnesium, thereby improving the machinability of the alloy.
- the Y phase will disperse the crystal grains of the main ⁇ phase, which improves the casting properties and corrosion resistance of the alloy.
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as Invention 3), comprising: copper in an amount of 60-65 wt% based on the total weight of the brass alloy, 0.01-0.15 wt% of bismuth, 0.1-0.5 wt. % magnesium, and 0.05-0.5 wt% manganese, 0.1-0.7 wt% aluminum, 0.05-0.5 wt% tin, 0.05-0.3 wt% phosphorus, and/or 0.001-0.01 wt%, based on the total weight of the brass alloy Boron, the remainder is zinc.
- Inventive 3 further adds 0.05-0.5 wt% of manganese, 0.1-0.7 wt% of aluminum, 0.05-0.5 wt% of tin, 0.05-0.3 wt% of phosphorus, based on the total weight of the brass alloy. And / or 0.001-0.01wt% addition of tin in the alloy is also to form the Y phase, improve the machinability of the alloy, and the addition of tin significantly improves the strength of the alloy, and improves its plasticity and corrosion resistance. However, considering the addition of tin, the cost is high. Therefore, adding aluminum while adding tin can improve the alloy's machinability, and also improve alloy strength, wear resistance, casting fluidity and high temperature oxidation resistance.
- the content range of phosphorus and manganese is the same as that of the invention 2, and the reason is the same as that of the invention 2.
- the addition of bismuth, magnesium, aluminum, tin, phosphorus, manganese and/or boron is selected according to the difference in machinability requirements of different products.
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as Invention 4), comprising: 60-65 wt% of copper, based on the total weight of the brass alloy, 0.01-0.15 wt% of bismuth, 0.1-0.5 wt.
- % magnesium and 0.05-0.5 wt% manganese, 0.1-0.7 wt% aluminum, 0.05-0.5 wt% tin, 0.05-0.3 wt% phosphorus, and/or 0.001-0.01 wt%, based on the total weight of the brass alloy
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as Invention 5), comprising: 60-65 wt% of copper, based on the total weight of the brass alloy, 0.01-0.15 wt% of bismuth, 0.1-0.5 wt.
- % magnesium and 0.05-0.5 wt% manganese, 0.1-0.7 wt% aluminum, 0.05-0.5 wt% tin, 0.05-0.3 wt% phosphorus, and/or 0.001-0.01 wt%, based on the total weight of the brass alloy Boron, the remainder being zinc, wherein the total content of manganese, aluminum, tin, phosphorus and/or boron is from 0.2 to 2% by weight based on the total weight of the brass alloy.
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as Invention 6), comprising: 60-65 wt% of copper, based on the total weight of the brass alloy, 0.01-0.15 wt% of bismuth, 0.1-0.5 wt.
- the invention 6 includes on the basis of the invention 3 some unavoidable impurities, namely mechanical impurities nickel, chromium and/or iron.
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as Invention 7), comprising: 60-65 wt% of copper, 0.05-0.5 wt% of tin, and more than two Selected from brass
- the total weight of gold is 0.1-0.7 wt% aluminum, 0.05-0.3 wt% phosphorus and 0.05-0.5 wt% manganese, and the remainder is zinc.
- the addition of 0.05-0.5wt% tin, based on the total weight of the alloy can also meet the need for machinability in industrial production, and the content is taken from the range of the invention 3, and the reason is also The reasons stated are the same; the addition of aluminum, phosphorus and manganese is selected according to the requirements of different products for machinability, and the content of the content is the same as that of the invention 3, and the reason is also explained by the invention 3 The reason is the same.
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as the invention 8) includes: copper, which is 60-65 wt% of the total weight of the brass alloy, and 0.05-0.5 wt% of tin, two or more selected
- the total weight of the brass alloy is 0.1-0.7 wt% aluminum, 0.05-0.3 wt% phosphorus and 0.05-0.5 wt% manganese element, and 0.01-0.15 wt% ⁇ , 0.1-0.5, based on the total weight of the brass alloy.
- the addition of strontium, magnesium, aluminum, tin, phosphorus, manganese and/or boron is selected according to the requirements of different products for the machinability.
- the content of the content is the same as the invention, and the reason and invention The reason stated in item 3 is the same.
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as the invention 9), comprising: copper, which is 60-65 wt% of the total weight of the brass alloy, 0.05-0.5 wt% of tin, and two or more
- the total weight of the brass alloy is 0.1-0.7 wt% aluminum, 0.05-0.3 wt% phosphorus and 0.05-0.5 wt% manganese element, and 0.01-0.15 wt% ⁇ , 0.1-0.5, based on the total weight of the brass alloy.
- Wwt% of magnesium and/or 0.001-0.01 wt% of boron the remainder being zinc and unavoidable impurities, wherein the unavoidable impurities include: nickel of less than 0.25 wt% of the total weight of the brass alloy, less than 0.15 wt% Chromium and/or iron below 0.25 wt%.
- the invention 9 includes on the basis of the invention 8 some unavoidable impurities, namely mechanical impurities nickel, chromium and/or iron.
- a lead-free, non-silicon-free, highly machinable brass alloy (hereinafter referred to as the invention 10), comprising: copper, which accounts for 60-65 wt% of the total weight of the brass alloy, 0.01-0.15 wt% of bismuth and 0.1-0.5 And wt% of magnesium, and one or more selected from the group consisting of aluminum in an amount of 0.1-0.7% by weight based on the total weight of the brass alloy, 0.05-0.5% by weight of tin, 0.05-0.3 wt% of phosphorus, 0.05-0.5 wt% of manganese and 0.001-0.01 wt% of boron, the remainder being zinc.
- the addition or not of aluminum, tin, phosphorus, manganese and/or boron is selected according to the requirements of different products for the machinability.
- the content of the content is the same as the invention 3, and the reason and the invention The reasons stated in 3 are the same.
- a lead-free, non-silicon-free, machinable brass alloy (hereinafter referred to as the invention 11), comprising: copper in an amount of 60-65 wt% of the total weight of the brass alloy, 0.01-0.15 wt% of bismuth and 0.1-0.5 The wt% of magnesium, and one or more selected from the group consisting of 0.1-0.7 wt% of aluminum, 0.05-0.5 wt% of tin, 0.05-0.3 wt% of phosphorus, 0.05-0.5 wt% of manganese, and 0.001 - 0.01 wt% of the element of boron, the remainder being zinc and unavoidable impurities, wherein the unavoidable impurities include: nickel of less than 0.25 wt% of the total weight of the brass alloy, chromium of 0.15 wt% or less and/or 0.25 wt Below the iron.
- the invention 11 includes on the basis of the invention 10 some unavoidable impurities, namely mechanical impurities nickel, chromium and/or iron.
- the present invention further provides a method for producing a brass alloy, and taking an example of the invention 3 as an example, comprising the following steps:
- a copper-manganese alloy is provided as a source of copper and manganese elements.
- the melting furnace used is a high-frequency melting furnace, and the high-frequency melting furnace is lined with graphite crucible.
- the high frequency melting furnace has the characteristics of fast melting rate, fast heating, clean and pollution-free, and self-stirring in the melting process (ie, affected by magnetic lines of force).
- the lead-free and antimony-free silicon-free brass alloy described in the present invention is added to a certain proportion in various proportions, and then subjected to a high-frequency melting furnace to produce a mechanical processing property comparable to known lead-containing brass, and is good. It has good tensile strength, elongation, and anti-zinc resistance, and is lead-free. It is suitable for use as an alloy material for replacing known lead-containing brass, such as faucets or bathroom accessories.
- Fig. 1 is a flow chart showing a method of manufacturing one of the inventions 3.
- the anti-dezincification corrosion resistance test referred to in this paper is carried out in the form of as-cast according to the AS-2345-2006 specification. 12.8g of copper chloride is added in 1000C.C deionized water, and the measured object is placed therein for 24h. , to determine the depth of dezincification.
- ⁇ represents dezincification depth less than ⁇ ⁇ ⁇ ;
- ⁇ represents dezincification depth between 100 ⁇ m and 200 ⁇ m; and represents dezincification depth greater than 200 ⁇ m.
- ⁇ represents a relative cutting rate greater than 85%; ⁇ represents a relative cutting rate greater than 70%.
- the tensile strength and elongation tests referred to herein were tested in the as-cast condition at room temperature.
- the comparative sample is a lead-containing yellow brass of the same specification and the same specification, that is, a C36000 alloy.
- composition ratio of C36000 alloy material is as follows, the unit is weight percentage (wt%): 1 is a flow chart of a manufacturing method of an embodiment of the invention 3, comprising the following steps: Step S100: Providing copper and manganese. In this step, a copper-manganese alloy can be provided as a source for providing the copper and manganese elements.
- Step S102 heating the copper-manganese mother alloy to a temperature between 1000 and 1050 ° C to form a copper-manganese alloy melt into a copper-manganese alloy.
- the copper-manganese alloy can be added to a high-frequency melting furnace, and the melting temperature is raised in the melting furnace to raise the temperature to between 1000-1050 ° C, even up to 1100 ° C, and the process lasts for 5-10 minutes. , melting the copper-manganese alloy into a copper-manganese alloy melt.
- the above action can prevent the liquid melted by copper and manganese from absorbing a large amount of external gas due to the temperature being too high, resulting in cracking of the formed alloy material.
- Step S104 Lowering the temperature of the copper-manganese alloy melt to between 950 and 1000 °C. In this step, when the temperature in the melting furnace is raised to between 1000 and 1050 ° C, when it lasts for 5-10 minutes, the power of the high frequency melting furnace is turned off, and the temperature in the melting furnace is lowered to 950-1000 ° C, and The copper-manganese alloy melt is also kept molten.
- Step S106 covering the glass slagging agent on the surface of the copper-manganese alloy melt.
- the glass will be The glass slag agent covers the surface of the copper-manganese alloy melt at 950-1000 ° C.
- This step can effectively block the contact between the liquid and the air, and prevent the zinc added in the next step from melting at 950-1000 ° C due to high temperature melting. Boiling volatile.
- Step S108 adding zinc to the copper-manganese alloy melt to form a copper-manganese-zinc melt.
- zinc is added to the melting furnace, and the copper-manganese alloy melt is sunk, and the zinc and the copper-manganese alloy melt are sufficiently melted to form a copper-manganese-zinc melt.
- Step S110 removing slag from the copper manganese zinc melt.
- the copper manganese zinc melt can be stirred and mixed by the action of high-frequency induction, and then the slag-forming agent is picked up.
- the slag remover is then used for slag removal.
- Step S112 adding lanthanum, aluminum, tin, magnesium to the copper manganese zinc melt to form a molten metal.
- a copper beryllium mother alloy, a copper aluminum mother alloy, a copper tin alloy, and a copper-magnesium alloy may be added to the copper manganese zinc melt.
- Step S114 Raising the temperature of the molten metal to between 1000 and 1050 ° C, and adding a copper boron alloy and a phosphor bronze alloy to form a lead-free, non-silicone-free brass alloy melt.
- Step S116 The brass alloy melt is cast out to form a brass alloy.
- the furnace temperature is controlled between 1000-1050 ° C, and finally the brass alloy melt is discharged to produce lead-free, silicon-free, silicon-free, and has good processing performance.
- the invention 1 of five different components prepared according to the above process is numbered 1001-1005, and each component is in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- Example 2 Table 2-1 shows Invention 2 of five different components prepared according to the above process, numbered 2001-2005, and each component is in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification. C36000 alloy.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows:
- Table 3-1 there are 8 different components of the invention 3 prepared according to the above process, numbered 3001-3008, and each component is in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate
- Table 4-1 shows the inventive compositions 4 of 8 different compositions prepared according to the above process, numbered 4001-4008, and the components are in weight percent (wt%). Table 4-1
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate (N/mm 2 )
- Table 5-1 shows the inventive compositions 5 of 8 different compositions prepared according to the above process, numbered 5001-5008, and the components are in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- Table 6-1 shows the inventive compositions of eight different components prepared according to the above process, numbered 6001-6008, and the components are in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate
- Example 7 In Table 7-1, the inventions 7 of the eight different components prepared according to the above process are numbered 7001-7008, and the components are in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate
- Table 8-1 shows the eight different compositions of the invention 8 obtained according to the above process, numbered 8001-8008, and each component is in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate
- Example 9 C36000 Alloy 394 9 ⁇ Example 9
- Table 9-1 the inventions 9 of the eight different components prepared according to the above process are numbered 9001-9008, and the components are in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate
- Table 10-1 shows the inventive compositions 10 of 8 different compositions prepared according to the above process, numbered 10001-10008, and each component is in weight percent (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate
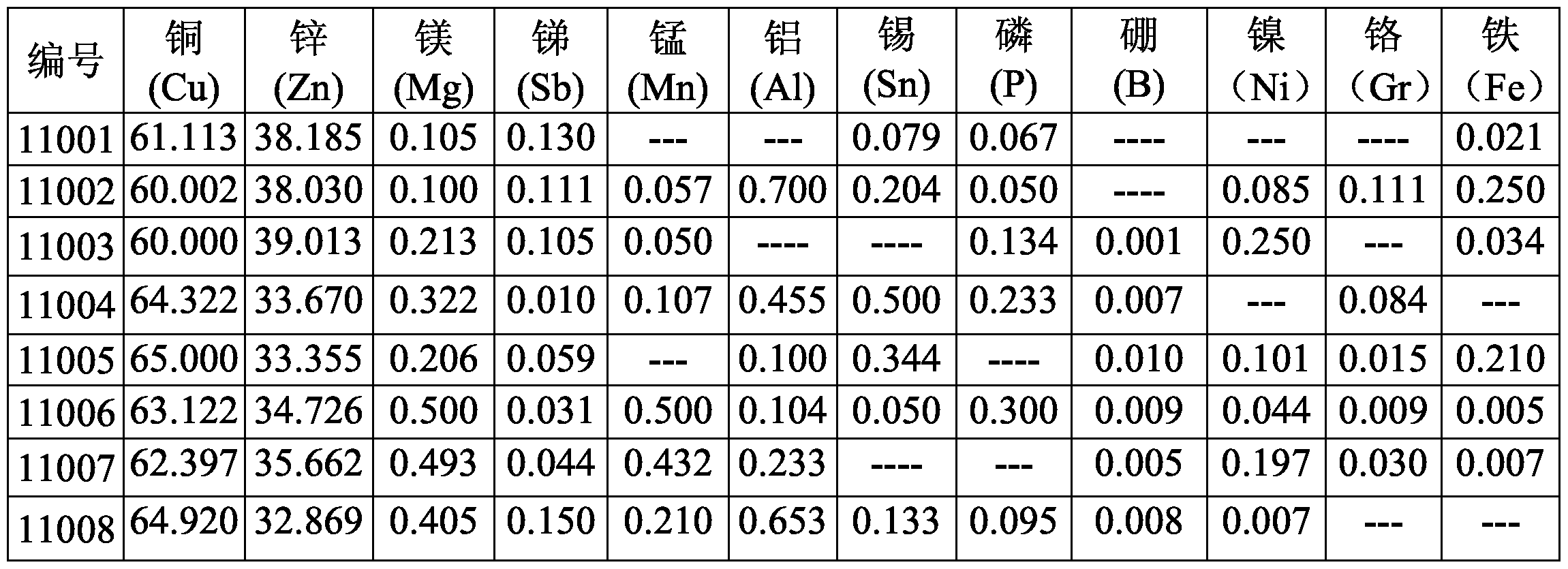
- Example 11 shows the inventive articles 11 of 8 different compositions prepared according to the above process, numbered 11001-11008, and the units of each component are percentage by weight (wt%).
- the alloys of the above components are tested in the as-cast form at room temperature for cutting performance, dezincification resistance, tensile strength and elongation.
- the comparative samples are lead-containing brass of the same specification and the same specification.
- the tensile strength, elongation, cutting performance and resistance to dezincification corrosion are as follows: No. Tensile strength elongation (%) Dezincification layer relative cutting rate
- a high-frequency melting furnace is used to produce a mechanical processing property comparable to known lead-containing brass, and good tensile strength, elongation, and dezincification resistance. Excellent, easy to cut, and lead free, suitable for use as an alloying material for known lead-containing brass, such as faucets or accessories for bathroom products.
- the present invention has been disclosed in the above embodiments, and is not intended to limit the present invention. Any person skilled in the art can make various modifications and refinements without departing from the spirit and scope of the present invention. The scope of protection is subject to the terms of the claims.
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015555583A JP6057109B2 (ja) | 2014-01-03 | 2014-04-09 | 鉛、ビスマスおよびケイ素を含まない黄銅 |
| US14/354,958 US20160362767A1 (en) | 2014-01-03 | 2014-04-09 | Lead-free bismuth-free silicon-free brass |
| AU2014202540A AU2014202540B2 (en) | 2014-01-03 | 2014-04-09 | Lead-free bismuth-free silicon-free brass |
| CN201480000087.0A CN103946402B (zh) | 2014-01-03 | 2014-04-09 | 无铅无铋无硅黄铜 |
| KR1020147015581A KR20150093100A (ko) | 2014-01-03 | 2014-04-09 | 리드, 비스무트 및 규소를 함유하지 않는 황동 |
| EP14725347.0A EP2913415A4 (en) | 2014-01-03 | 2014-04-09 | STAINLESS, INSULATED, SILICONE-FREE BRASS |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201410003372.X | 2014-01-03 | ||
| CN201410003372 | 2014-01-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015100873A1 true WO2015100873A1 (zh) | 2015-07-09 |
Family
ID=53493076
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2014/074942 WO2015100873A1 (zh) | 2014-01-03 | 2014-04-09 | 无铅无铋无硅黄铜 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20160362767A1 (zh) |
| EP (1) | EP2913415A4 (zh) |
| JP (1) | JP6057109B2 (zh) |
| KR (1) | KR20150093100A (zh) |
| AU (1) | AU2014202540B2 (zh) |
| TW (1) | TWI550105B (zh) |
| WO (1) | WO2015100873A1 (zh) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101838173B1 (ko) | 2016-11-29 | 2018-03-13 | 한국생산기술연구원 | 쾌삭 황동합금 및 그 제조방법 |
| CN107385273B (zh) * | 2017-07-07 | 2019-03-01 | 路达(厦门)工业有限公司 | 一种铸造用环保黄铜合金及其制造方法 |
| DE102021119474A1 (de) | 2021-07-27 | 2023-02-02 | Diehl Brass Solutions Stiftung & Co. Kg | Blei- und Antimonfreie Messinglegierung |
| DE102022120122A1 (de) | 2022-08-10 | 2024-02-15 | Diehl Brass Solutions Stiftung & Co. Kg | Bleifreie Messinglegierung |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0397817A (ja) * | 1989-09-08 | 1991-04-23 | Furukawa Electric Co Ltd:The | ワイヤ放電加工用電極線 |
| CN101298643A (zh) * | 2008-06-30 | 2008-11-05 | 中铝洛阳铜业有限公司 | 一种环保易切削铜合金材料及加工工艺 |
| CN101423905A (zh) * | 2008-12-05 | 2009-05-06 | 浙江科宇金属材料有限公司 | 一种无铅易切削锑镁黄铜合金 |
| TW201100564A (en) * | 2009-06-26 | 2011-01-01 | Chan Wen Copper Industry Co Ltd | Lead free copper zinc alloy |
| US20110129384A1 (en) * | 2009-11-27 | 2011-06-02 | Chan Wen Copper Industry Co., Ltd. | Copper-zinc alloy |
| CN103205597A (zh) * | 2012-01-16 | 2013-07-17 | 摩登岛股份有限公司 | 无铅的锑镍黄铜合金 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5934222B2 (ja) * | 1981-11-13 | 1984-08-21 | 日本鉱業株式会社 | ラジエ−タ−用銅合金 |
| JPS6086233A (ja) * | 1983-10-14 | 1985-05-15 | Nippon Mining Co Ltd | 高力導電銅合金 |
| JPH01272734A (ja) * | 1988-04-22 | 1989-10-31 | Kobe Steel Ltd | 熱間加工用耐食性銅合金 |
| DE10308779B8 (de) * | 2003-02-28 | 2012-07-05 | Wieland-Werke Ag | Bleifreie Kupferlegierung und deren Verwendung |
| CN1291051C (zh) * | 2004-01-15 | 2006-12-20 | 宁波博威集团有限公司 | 无铅易切削锑黄铜合金 |
| US20100303667A1 (en) * | 2009-03-09 | 2010-12-02 | Lazarus Norman M | Novel lead-free brass alloy |
| US20110129385A1 (en) * | 2009-11-27 | 2011-06-02 | Chan Wen Copper Industry Co., Ltd. | Copper-zinc alloy |
| CN102477497B (zh) * | 2010-11-30 | 2013-08-21 | 逢威工业(惠阳)有限公司 | 无铅铋易切削锑黄铜合金 |
-
2014
- 2014-04-09 US US14/354,958 patent/US20160362767A1/en not_active Abandoned
- 2014-04-09 AU AU2014202540A patent/AU2014202540B2/en active Active
- 2014-04-09 WO PCT/CN2014/074942 patent/WO2015100873A1/zh active Application Filing
- 2014-04-09 EP EP14725347.0A patent/EP2913415A4/en not_active Withdrawn
- 2014-04-09 JP JP2015555583A patent/JP6057109B2/ja active Active
- 2014-04-09 KR KR1020147015581A patent/KR20150093100A/ko not_active Application Discontinuation
- 2014-04-23 TW TW103114727A patent/TWI550105B/zh active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0397817A (ja) * | 1989-09-08 | 1991-04-23 | Furukawa Electric Co Ltd:The | ワイヤ放電加工用電極線 |
| CN101298643A (zh) * | 2008-06-30 | 2008-11-05 | 中铝洛阳铜业有限公司 | 一种环保易切削铜合金材料及加工工艺 |
| CN101423905A (zh) * | 2008-12-05 | 2009-05-06 | 浙江科宇金属材料有限公司 | 一种无铅易切削锑镁黄铜合金 |
| TW201100564A (en) * | 2009-06-26 | 2011-01-01 | Chan Wen Copper Industry Co Ltd | Lead free copper zinc alloy |
| US20110129384A1 (en) * | 2009-11-27 | 2011-06-02 | Chan Wen Copper Industry Co., Ltd. | Copper-zinc alloy |
| CN103205597A (zh) * | 2012-01-16 | 2013-07-17 | 摩登岛股份有限公司 | 无铅的锑镍黄铜合金 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20160362767A1 (en) | 2016-12-15 |
| EP2913415A1 (en) | 2015-09-02 |
| TW201527558A (zh) | 2015-07-16 |
| EP2913415A4 (en) | 2016-10-26 |
| AU2014202540A1 (en) | 2015-07-23 |
| AU2014202540B2 (en) | 2016-07-07 |
| JP2016507655A (ja) | 2016-03-10 |
| KR20150093100A (ko) | 2015-08-17 |
| JP6057109B2 (ja) | 2017-01-11 |
| TWI550105B (zh) | 2016-09-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI550106B (zh) | Low lead free bismuth no silicon brass alloy | |
| CA2639394C (en) | Tin-free lead-free free-cutting magnesium brass alloy and its manufacturing method | |
| JP6335194B2 (ja) | 良好な熱成形性を有する、無鉛の、切断が容易な、耐腐食性真鍮合金 | |
| CA2639301C (en) | Lead-free free-cutting phosphorous brass alloy and its manufacturing method | |
| JP2007517981A (ja) | アンチモンを含む無鉛快削性黄銅合金 | |
| WO2006136065A1 (fr) | Nouvel alliage de laiton écologique sans plomb à découpe libre qui présente une faible teneur en antimoine et bismuth, et sa préparation | |
| CN102618747A (zh) | 易切削的黄铜合金 | |
| CN103509967A (zh) | 一种重力铸造专用dzr环保黄铜合金锭及其制作工艺 | |
| TWI550105B (zh) | Lead - free bismuth - free silicon - brass alloy | |
| CN107974573B (zh) | 一种含锰易切削硅黄铜合金及其制备方法和应用 | |
| CN110747369A (zh) | 一种无铅易切削硅镁钙黄铜合金及其制备方法 | |
| CN103946402B (zh) | 无铅无铋无硅黄铜 | |
| CN101921926B (zh) | 一种低钙易切削硅黄铜合金及其制备方法 | |
| WO2011067682A1 (en) | Low lead brass alloy | |
| CN104726743B (zh) | 黄铜合金及其制造方法 | |
| CN101423905A (zh) | 一种无铅易切削锑镁黄铜合金 | |
| CN103194643A (zh) | 一种含硅与铝的铜基合金管及其制备方法 | |
| CA2687452C (en) | Brass alloy | |
| CN110938761B (zh) | 一种低铅易切削镁黄铜合金及其制备方法 | |
| KR101194218B1 (ko) | 금형주조용 무연청동 합금 및 그 제조방법 | |
| TWI392751B (zh) | 低鉛銅合金 | |
| CN101514410A (zh) | 一种无铅易切削镁锑黄铜合金 | |
| TWI500783B (zh) | Brass alloy and its manufacturing method | |
| TW201533252A (zh) | 環保抗鋅氧化黃銅合金配方及其製造方法 | |
| CA2718613A1 (en) | Dezincification resistant brass alloy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014202540 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2015555583 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2014725347 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014725347 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20147015581 Country of ref document: KR Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14725347 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14354958 Country of ref document: US |