WO2014157165A1 - Steel plate with excellent hydrogen-induced cracking resistance and toughness, and line pipe steel tube - Google Patents
Steel plate with excellent hydrogen-induced cracking resistance and toughness, and line pipe steel tube Download PDFInfo
- Publication number
- WO2014157165A1 WO2014157165A1 PCT/JP2014/058222 JP2014058222W WO2014157165A1 WO 2014157165 A1 WO2014157165 A1 WO 2014157165A1 JP 2014058222 W JP2014058222 W JP 2014058222W WO 2014157165 A1 WO2014157165 A1 WO 2014157165A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- less
- toughness
- steel
- steel plate
- amount
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0247—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the heat treatment
- C21D8/0263—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the heat treatment following hot rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/46—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for sheet metals
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/001—Ferrous alloys, e.g. steel alloys containing N
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/002—Ferrous alloys, e.g. steel alloys containing In, Mg, or other elements not provided for in one single group C22C38/001 - C22C38/60
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/005—Ferrous alloys, e.g. steel alloys containing rare earths, i.e. Sc, Y, Lanthanides
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/06—Ferrous alloys, e.g. steel alloys containing aluminium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/08—Ferrous alloys, e.g. steel alloys containing nickel
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/12—Ferrous alloys, e.g. steel alloys containing tungsten, tantalum, molybdenum, vanadium, or niobium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/14—Ferrous alloys, e.g. steel alloys containing titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/16—Ferrous alloys, e.g. steel alloys containing copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/26—Ferrous alloys, e.g. steel alloys containing chromium with niobium or tantalum
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/28—Ferrous alloys, e.g. steel alloys containing chromium with titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/42—Ferrous alloys, e.g. steel alloys containing chromium with nickel with copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/44—Ferrous alloys, e.g. steel alloys containing chromium with nickel with molybdenum or tungsten
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/48—Ferrous alloys, e.g. steel alloys containing chromium with nickel with niobium or tantalum
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/50—Ferrous alloys, e.g. steel alloys containing chromium with nickel with titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/54—Ferrous alloys, e.g. steel alloys containing chromium with nickel with boron
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0221—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the working steps
- C21D8/0226—Hot rolling
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Heat Treatment Of Steel (AREA)
Abstract
Description
C:0.02~0.15%(%は質量%の意味。以下同じ)、
Si:0.02~0.50%、
Mn:0.6~2.0%、
P:0%超0.030%以下、
S:0%超0.003%以下、
Al:0.010~0.08%、
Ca:0.0003~0.0060%、
N:0.001~0.01%、および
O(酸素):0%超0.0045%以下を満たし、残部が鉄および不可避不純物からなり、
前記Caと前記Sの比(Ca/S)が2.0以上であり、かつ
板厚方向に表面から深さ5mmまでの領域の最大Ca濃度(Cmax)と該領域の平均Ca濃度(Cave)との比(Cmax/Cave)が1.20以下であるところに特徴を有する。 The steel sheet excellent in hydrogen-induced crack resistance and toughness of the present invention that has solved the above problems is
C: 0.02 to 0.15% (% means mass%, the same applies hereinafter)
Si: 0.02 to 0.50%,
Mn: 0.6 to 2.0%,
P: more than 0% and 0.030% or less,
S: more than 0% and 0.003% or less,
Al: 0.010 to 0.08%,
Ca: 0.0003 to 0.0060%,
N: 0.001 to 0.01%, and O (oxygen): more than 0% and 0.0045% or less, with the balance consisting of iron and inevitable impurities,
The ratio of Ca to S (Ca / S) is 2.0 or more, and the maximum Ca concentration (Cmax) in the region from the surface to the depth of 5 mm in the plate thickness direction and the average Ca concentration (Cave) in the region And the ratio (Cmax / Cave) is 1.20 or less.
(a)B:0%超0.005%以下、
V:0%超0.1%以下、
Cu:0%超1.5%以下、
Ni:0%超1.5%以下、
Cr:0%超1.5%以下、
Mo:0%超1.5%以下、および
Nb:0%超0.06%以下よりなる群
(b)Ti:0%超0.03%以下、
Mg:0%超0.01%以下、
REM:0%超0.02%以下、および
Zr:0%超0.010%以下よりなる群 The steel sheet may further contain one or more elements selected from at least one of the following groups (a) and (b) as other elements.
(A) B: more than 0% and 0.005% or less,
V: more than 0% and 0.1% or less,
Cu: more than 0% and 1.5% or less,
Ni: more than 0% and 1.5% or less,
Cr: more than 0% and 1.5% or less,
Mo: more than 0% and not more than 1.5%, and Nb: more than 0% and not more than 0.06% (b) Ti: more than 0% and not more than 0.03%,
Mg: more than 0% and 0.01% or less,
REM: a group consisting of more than 0% and 0.02% or less, and Zr: more than 0% and 0.010% or less
[C:0.02~0.15%]
Cは、母材および溶接部の強度を確保するために必要不可欠な元素であり、0.02%以上含有させる必要がある。C量は、好ましくは0.03%以上であり、より好ましくは0.05%以上である。一方、C量が多すぎるとHAZ靭性と溶接性が劣化する。またC量が過剰であると、HICの起点や破壊進展経路となるNbCや島状マルテンサイトが生成しやすくなる。よってC量は0.15%以下とする必要がある。C量は、好ましくは0.12%以下、より好ましくは0.10%以下である。 (Component composition)
[C: 0.02 to 0.15%]
C is an indispensable element for securing the strength of the base material and the welded portion, and needs to be contained by 0.02% or more. The amount of C is preferably 0.03% or more, and more preferably 0.05% or more. On the other hand, if the amount of C is too large, the HAZ toughness and weldability deteriorate. On the other hand, if the amount of C is excessive, NbC and island-shaped martensite that become the starting point of HIC and the fracture propagation path are likely to be generated. Therefore, the C amount needs to be 0.15% or less. The amount of C is preferably 0.12% or less, more preferably 0.10% or less.
Siは、脱酸作用を有する上に、母材および溶接部の強度向上に有効な元素である。これらの効果を得るため、Si量を0.02%以上とする。Si量は、好ましくは0.05%以上であり、より好ましくは0.15%以上である。しかし、Si量が多すぎると溶接性や靭性が劣化する。またSi量が過剰であると、島状マルテンサイトが生じてHICが発生・進展する。よってSi量は、0.50%以下に抑える必要がある。Si量は、好ましくは0.45%以下、より好ましくは0.35%以下である。 [Si: 0.02 to 0.50%]
Si is an element that has a deoxidizing action and is effective in improving the strength of the base material and the welded portion. In order to obtain these effects, the Si content is set to 0.02% or more. The amount of Si is preferably 0.05% or more, and more preferably 0.15% or more. However, if the amount of Si is too large, weldability and toughness deteriorate. If the amount of Si is excessive, island martensite is generated and HIC is generated and progresses. Therefore, the amount of Si needs to be suppressed to 0.50% or less. The amount of Si is preferably 0.45% or less, more preferably 0.35% or less.
Mnは、母材および溶接部の強度向上に有効な元素であり、本発明では0.6%以上含有させる。Mn量は、好ましくは0.8%以上であり、より好ましくは1.0%以上である。しかし、Mn量が多すぎると、MnSが生成されて耐水素誘起割れ性が劣化するだけでなくHAZ靭性や溶接性も劣化する。よってMn量の上限を2.0%以下とする。好ましくは1.8%以下であり、より好ましくは1.5%以下、さらに好ましくは1.2%以下である。 [Mn: 0.6 to 2.0%]
Mn is an element effective for improving the strength of the base material and the welded portion, and is contained in an amount of 0.6% or more in the present invention. The amount of Mn is preferably 0.8% or more, and more preferably 1.0% or more. However, if the amount of Mn is too large, not only MnS is produced and the hydrogen-induced cracking resistance deteriorates, but also the HAZ toughness and weldability deteriorate. Therefore, the upper limit of the amount of Mn is 2.0% or less. Preferably it is 1.8% or less, More preferably, it is 1.5% or less, More preferably, it is 1.2% or less.
Pは、鋼材中に不可避的に含まれる元素であり、P量が0.030%を超えると母材やHAZ部の靭性劣化が著しく、耐水素誘起割れ性も劣化する。よって本発明ではP量を0.030%以下に抑える。P量は、好ましくは0.020%以下、より好ましくは0.010%以下である。 [P: more than 0% and 0.030% or less]
P is an element inevitably contained in the steel material. If the amount of P exceeds 0.030%, the toughness of the base material and the HAZ part is significantly deteriorated, and the resistance to hydrogen-induced cracking is also deteriorated. Therefore, in the present invention, the amount of P is suppressed to 0.030% or less. The amount of P is preferably 0.020% or less, more preferably 0.010% or less.
Sは、多すぎるとMnSを多量に生成し耐水素誘起割れ性を著しく劣化させる元素であるため、本発明ではS量の上限を0.003%とする。S量は、好ましくは0.002%以下であり、より好ましくは0.0015%以下、更に好ましくは0.0010%以下である。この様に耐水素誘起割れ性向上の観点からは少ない方が望ましい。 [S: more than 0% and 0.003% or less]
If S is too much, it is an element that produces a large amount of MnS and significantly deteriorates the resistance to hydrogen-induced cracking. Therefore, in the present invention, the upper limit of the amount of S is set to 0.003%. The amount of S is preferably 0.002% or less, more preferably 0.0015% or less, and still more preferably 0.0010% or less. Thus, the smaller one is desirable from the viewpoint of improving hydrogen-induced crack resistance.
Alは強脱酸元素であり、Al量が少ないと、酸化物中のCa濃度が上昇、即ち、Ca系介在物が鋼板表層部に形成されやすくなり微細なHICが発生する。よって本発明では、Alを0.010%以上とする必要がある。Al量は、好ましくは0.020%以上、より好ましくは0.030%以上である。一方、Al含有量が多すぎると、Alの酸化物がクラスター状に生成し水素誘起割れの起点となる。よってAl量は0.08%以下とする必要がある。Al量は、好ましくは0.06%以下であり、より好ましくは0.05%以下である。 [Al: 0.010 to 0.08%]
Al is a strong deoxidizing element. When the amount of Al is small, the Ca concentration in the oxide increases, that is, Ca inclusions are easily formed in the surface layer of the steel sheet, and fine HIC is generated. Therefore, in the present invention, Al needs to be 0.010% or more. The amount of Al is preferably 0.020% or more, more preferably 0.030% or more. On the other hand, when there is too much Al content, the oxide of Al will produce | generate in cluster shape and will become the starting point of a hydrogen induced crack. Therefore, the Al amount needs to be 0.08% or less. The amount of Al is preferably 0.06% or less, and more preferably 0.05% or less.
Caは、硫化物の形態を制御する作用があり、CaSを形成することによってMnSの形成を抑制する効果がある。この効果を得るには、Ca量を0.0003%以上とする必要がある。Ca量は、好ましくは0.0005%以上であり、より好ましくは0.0010%以上である。一方、Ca量が0.0060%を超えると、Ca系介在物を起点にHICが多く発生する。よって本発明では、Ca量の上限を0.0060%とする。Ca量は、好ましくは0.0045%以下であり、より好ましくは0.0035%以下、さらに好ましくは0.0025%以下である。 [Ca: 0.0003 to 0.0060%]
Ca has the effect | action which controls the form of sulfide, and there exists an effect which suppresses formation of MnS by forming CaS. In order to obtain this effect, the Ca content needs to be 0.0003% or more. The Ca content is preferably 0.0005% or more, and more preferably 0.0010% or more. On the other hand, when the Ca content exceeds 0.0060%, a large amount of HIC is generated starting from Ca-based inclusions. Therefore, in the present invention, the upper limit of the Ca amount is set to 0.0060%. The Ca content is preferably 0.0045% or less, more preferably 0.0035% or less, and still more preferably 0.0025% or less.
Nは、鋼組織中にTiNとして析出し、HAZ部のオーステナイト粒の粗大化を抑制し、さらにフェライト変態を促進させて、HAZ部の靭性を向上させる元素である。この効果を得るにはNを0.001%以上含有させる必要がある。N量は、好ましくは0.003%以上であり、より好ましくは0.0040%以上である。しかし、N量が多すぎると、固溶Nの存在によりHAZ靭性がかえって劣化するため、N量は、0.01%以下とする必要がある。好ましくは0.008%以下であり、より好ましくは0.0060%以下である。 [N: 0.001 to 0.01%]
N is an element that precipitates as TiN in the steel structure, suppresses coarsening of the austenite grains in the HAZ part, further promotes ferrite transformation, and improves the toughness of the HAZ part. In order to acquire this effect, it is necessary to contain N 0.001% or more. The N amount is preferably 0.003% or more, and more preferably 0.0040% or more. However, if the amount of N is too large, the HAZ toughness deteriorates due to the presence of solute N, so the amount of N needs to be 0.01% or less. Preferably it is 0.008% or less, More preferably, it is 0.0060% or less.
O(酸素)は、清浄度向上の観点から低いほうが望ましく、Oが多量に含まれる場合、靭性が劣化することに加え、酸化物を起点にHICが発生し、耐水素誘起割れ性が劣化する。この観点から、O量は0.0045%以下とする必要があり、好ましくは0.0030%以下、より好ましくは0.0020%以下である。 [O: more than 0% and 0.0045% or less]
O (oxygen) is preferably low from the viewpoint of improving cleanliness. When a large amount of O is contained, in addition to deterioration of toughness, HIC is generated starting from oxide, and resistance to hydrogen-induced cracking is deteriorated. . From this viewpoint, the amount of O needs to be 0.0045% or less, preferably 0.0030% or less, more preferably 0.0020% or less.
Caに対してSが過剰となる場合、板厚中央部を中心にMnSが生成し、MnSを起点にHICが発生する。これを抑制するためにはCa/Sを2.0以上とする必要があり、好ましくは2.5以上、より好ましくは3.0以上である。尚、本発明で規定するCa量とS量からCa/Sの上限は15程度となる。 [Ca / S (mass ratio): 2.0 or more]
When S is excessive with respect to Ca, MnS is generated around the center of the plate thickness, and HIC is generated starting from MnS. In order to suppress this, Ca / S needs to be 2.0 or more, preferably 2.5 or more, more preferably 3.0 or more. The upper limit of Ca / S is about 15 from the Ca amount and S amount specified in the present invention.
(a)下記量のB、V、Cu、Ni、Cr、Mo、およびNbよりなる群から選択される1種類以上の元素を含有させて、強度や靭性をより高めることや、
(b)下記量のTi、Mg、REM、およびZrよりなる群から選択される1種類以上の元素を含有させて、HAZ靭性をより高めるとともに、脱硫を促進させ耐HIC性をより改善することができる。以下、これらの元素について詳述する。 The components of the steel material (steel plate, steel pipe) of the present invention are as described above, and the balance consists of iron and inevitable impurities. In addition to the above elements,
(A) Inclusion of one or more elements selected from the group consisting of the following amounts of B, V, Cu, Ni, Cr, Mo, and Nb to further increase strength and toughness,
(B) To contain one or more elements selected from the group consisting of Ti, Mg, REM, and Zr in the following amounts to increase HAZ toughness and promote desulfurization to further improve HIC resistance. Can do. Hereinafter, these elements will be described in detail.
Bは、焼入れ性を高め、母材および溶接部の強度を高めるとともに、溶接時に、加熱されたHAZ部が冷却する過程でNと結合してBNを析出し、オーステナイト粒内からのフェライト変態を促進するため、HAZ靭性を向上させる。この効果を得るためには、B量を0.0002%以上含有させることが好ましい。より好ましくは0.0005%以上であり、更に好ましくは0.0010%以上である。しかし、B含有量が過多になると、母材とHAZ部の靭性が劣化したり、溶接性の劣化を招くため、B含有量は0.005%以下とするのが好ましい。より好ましくは0.004%以下、更に好ましくは0.0030%以下である。 [B: more than 0% and 0.005% or less]
B enhances hardenability, increases the strength of the base metal and the welded portion, and bonds with N during the process of cooling the heated HAZ portion during precipitation, thereby precipitating BN and causing ferrite transformation from within the austenite grains. In order to promote, HAZ toughness is improved. In order to acquire this effect, it is preferable to contain B amount 0.0002% or more. More preferably, it is 0.0005% or more, More preferably, it is 0.0010% or more. However, if the B content is excessive, the toughness between the base material and the HAZ part deteriorates or weldability deteriorates, so the B content is preferably 0.005% or less. More preferably, it is 0.004% or less, More preferably, it is 0.0030% or less.
Vは、強度の向上に有効な元素であり、この効果を得るには0.003%以上含有させることが好ましい。より好ましくは0.010%以上である。一方、V含有量が0.1%を超えると溶接性と母材靭性が劣化する。よってV量は0.1%以下とすることが好ましく、より好ましくは0.08%以下である。 [V: more than 0% and 0.1% or less]
V is an element effective for improving the strength. To obtain this effect, V is preferably contained in an amount of 0.003% or more. More preferably, it is 0.010% or more. On the other hand, if the V content exceeds 0.1%, weldability and base metal toughness deteriorate. Therefore, the V amount is preferably 0.1% or less, and more preferably 0.08% or less.
Cuは、焼入れ性を向上させて強度を高めるのに有効な元素である。この効果を得るにはCuを0.01%以上含有させることが好ましい。Cu量は、より好ましくは0.05%以上、更に好ましくは0.10%以上である。しかし、Cu含有量が1.5%を超えると靭性が劣化するため、1.5%以下とすることが好ましい。Cu量は、より好ましくは1.0%以下、更に好ましくは0.50%以下である。 [Cu: more than 0% and 1.5% or less]
Cu is an element effective for improving the hardenability and increasing the strength. In order to acquire this effect, it is preferable to contain 0.01% or more of Cu. The amount of Cu is more preferably 0.05% or more, and still more preferably 0.10% or more. However, if the Cu content exceeds 1.5%, the toughness deteriorates, so it is preferable to set it to 1.5% or less. The amount of Cu is more preferably 1.0% or less, still more preferably 0.50% or less.
Niは、母材および溶接部の強度と靭性の向上に有効な元素である。この効果を得るためには、Ni量を0.01%以上とすることが好ましい。Ni量は、より好ましくは0.05%以上、更に好ましくは0.10%以上である。しかしNiが多量に含まれると、構造用鋼材として極めて高価となるため、経済的な観点からNi量は1.5%以下とすることが好ましい。Ni量は、より好ましくは1.0%以下、更に好ましくは0.50%以下である。 [Ni: more than 0% and 1.5% or less]
Ni is an element effective for improving the strength and toughness of the base material and the welded portion. In order to obtain this effect, the Ni content is preferably 0.01% or more. The amount of Ni is more preferably 0.05% or more, and still more preferably 0.10% or more. However, if Ni is contained in a large amount, it becomes extremely expensive as a structural steel material. Therefore, the Ni content is preferably 1.5% or less from an economical viewpoint. The amount of Ni is more preferably 1.0% or less, and still more preferably 0.50% or less.
Crは、強度の向上に有効な元素であり、この効果を得るには0.01%以上含有させることが好ましい。Cr量は、より好ましくは0.05%以上、更に好ましくは0.10%以上である。一方、Cr量が1.5%を超えるとHAZ靭性が劣化する。よってCr量は1.5%以下とすることが好ましい。Cr量は、より好ましくは1.0%以下、更に好ましくは0.50%以下である。 [Cr: more than 0% and 1.5% or less]
Cr is an element effective for improving the strength, and in order to obtain this effect, it is preferable to contain 0.01% or more. The amount of Cr is more preferably 0.05% or more, and still more preferably 0.10% or more. On the other hand, if the Cr content exceeds 1.5%, the HAZ toughness deteriorates. Therefore, the Cr content is preferably 1.5% or less. The amount of Cr is more preferably 1.0% or less, and still more preferably 0.50% or less.
Moは、母材の強度と靭性の向上に有効な元素である。この効果を得るには、Mo量を0.01%以上とすることが好ましい。Mo量は、より好ましくは0.05%以上、更に好ましくは0.10%以上である。しかし、Mo量が1.5%を超えるとHAZ靭性および溶接性が劣化する。よってMo量は1.5%以下とすることが好ましく、より好ましくは1.0%以下、更に好ましくは0.50%以下である。 [Mo: more than 0% and 1.5% or less]
Mo is an element effective for improving the strength and toughness of the base material. In order to obtain this effect, the Mo amount is preferably 0.01% or more. The amount of Mo is more preferably 0.05% or more, and still more preferably 0.10% or more. However, if the Mo amount exceeds 1.5%, the HAZ toughness and weldability deteriorate. Therefore, the Mo amount is preferably 1.5% or less, more preferably 1.0% or less, and still more preferably 0.50% or less.
Nbは、溶接性を劣化させることなく強度と母材靭性を高めるのに有効な元素である。この効果を得るには、Nb量を0.002%以上とすることが好ましい。Nb量は、より好ましくは0.010%以上、更に好ましくは0.020%以上である。しかし、Nb量が0.06%を超えると母材とHAZの靭性が劣化する。よって、本発明ではNb量の上限を0.06%とすることが好ましい。Nb量は、より好ましくは0.050%以下、更に好ましくは0.040%以下、より更に好ましくは0.030%以下である。 [Nb: more than 0% and 0.06% or less]
Nb is an element effective for increasing strength and base metal toughness without degrading weldability. In order to obtain this effect, the Nb content is preferably 0.002% or more. The Nb amount is more preferably 0.010% or more, and still more preferably 0.020% or more. However, if the Nb content exceeds 0.06%, the toughness of the base material and the HAZ deteriorates. Therefore, in the present invention, the upper limit of the Nb amount is preferably 0.06%. The Nb amount is more preferably 0.050% or less, still more preferably 0.040% or less, and still more preferably 0.030% or less.
Tiは、鋼中にTiNとして析出することで、溶接時のHAZ部でのオーステナイト粒の粗大化を防止しかつフェライト変態を促進するため、HAZ部の靭性を向上させるのに必要な元素である。さらにTiは、脱硫作用を示すため耐HIC性の向上にも有効な元素である。これらの効果を得るには、Tiを0.003%以上含有させることが好ましい。Ti量は、より好ましくは0.005%以上、更に好ましくは0.010%以上である。一方、Ti含有量が過多になると、Tiの固溶やTiCの析出により母材とHAZ部の靭性が劣化するため、0.03%以下とすることが好ましい。Ti量は、より好ましくは0.02%以下である。 [Ti: more than 0% and 0.03% or less]
Ti is an element necessary for improving the toughness of the HAZ part in order to prevent coarsening of the austenite grains in the HAZ part during welding and promote ferrite transformation by precipitating as TiN in the steel. . Further, Ti is an element effective for improving the HIC resistance since it exhibits a desulfurization action. In order to obtain these effects, it is preferable to contain 0.003% or more of Ti. The amount of Ti is more preferably 0.005% or more, and still more preferably 0.010% or more. On the other hand, if the Ti content is excessive, the toughness of the base material and the HAZ part deteriorates due to solid solution of Ti and precipitation of TiC, so 0.03% or less is preferable. The amount of Ti is more preferably 0.02% or less.
Mgは、結晶粒の微細化を通じて靭性の向上に有効な元素であり、また脱硫作用を示すため耐HIC性の向上にも有効な元素である。この効果を得るためには、Mgを0.0003%以上含有させることが好ましい。Mg量は、より好ましくは0.001%以上である。一方、Mgを、過剰に含有させても効果が飽和するため、Mg量の上限は0.01%とすることが好ましい。Mg量は、より好ましくは0.005%以下である。 [Mg: more than 0% and 0.01% or less]
Mg is an element effective for improving toughness through refinement of crystal grains, and is an element effective for improving HIC resistance since it exhibits a desulfurization action. In order to acquire this effect, it is preferable to contain Mg 0.0003% or more. The amount of Mg is more preferably 0.001% or more. On the other hand, since the effect is saturated even if Mg is excessively contained, the upper limit of the Mg content is preferably 0.01%. The amount of Mg is more preferably 0.005% or less.
REM(希土類元素)は、脱硫作用によりMnSの生成を抑制し耐水素誘起割れ性を高めるのに有効な元素である。このような効果を発揮させるには、REMを0.0002%以上含有させることが好ましい。REM量は、より好ましくは0.0005%以上、更に好ましくは0.0010%以上である。一方、REMを多量に含有させても効果が飽和する。よってREM量の上限は0.02%とすることが好ましい。鋳造時の浸漬ノズルの閉塞を抑えて生産性を高める観点からは、REM量を0.015%以下とすることがより好ましく、更に好ましくは0.010%以下、より更に好ましくは0.0050%以下である。尚、本発明において、上記REMとは、ランタノイド元素(LaからLuまでの15元素)とSc(スカンジウム)およびYを意味する。 [REM: more than 0% and 0.02% or less]
REM (rare earth element) is an element effective for suppressing the formation of MnS by the desulfurization action and enhancing the resistance to hydrogen-induced cracking. In order to exhibit such an effect, it is preferable to contain REM 0.0002% or more. The amount of REM is more preferably 0.0005% or more, and further preferably 0.0010% or more. On the other hand, the effect is saturated even if a large amount of REM is contained. Therefore, the upper limit of the REM amount is preferably 0.02%. From the viewpoint of increasing productivity by suppressing the clogging of the immersion nozzle during casting, the REM content is more preferably 0.015% or less, still more preferably 0.010% or less, and still more preferably 0.0050%. It is as follows. In the present invention, the REM means a lanthanoid element (15 elements from La to Lu), Sc (scandium) and Y.
Zrは、脱硫作用により耐HIC性の向上に寄与するとともに、酸化物を形成し微細に分散することでHAZ靭性の向上にも寄与する元素である。これらの効果を発揮させるには、Zr量を0.0003%以上とすることが好ましい。Zr量は、より好ましくは0.0005%以上、更に好ましくは0.0010%以上、より更に好ましくは0.0015%以上である。一方、Zrを過剰に添加すると粗大な介在物を形成して耐水素誘起割れ性および母材靭性を劣化させる。よってZr量は0.010%以下とすることが好ましい。Zr量は、より好ましくは0.0070%以下、更に好ましくは0.0050%以下、より更に好ましくは0.0030%以下である。 [Zr: more than 0% and 0.010% or less]
Zr is an element that contributes to improvement of HIC resistance by desulfurization and also contributes to improvement of HAZ toughness by forming an oxide and finely dispersing it. In order to exert these effects, the Zr content is preferably 0.0003% or more. The amount of Zr is more preferably 0.0005% or more, still more preferably 0.0010% or more, and still more preferably 0.0015% or more. On the other hand, when Zr is added excessively, coarse inclusions are formed and the hydrogen-induced crack resistance and the base metal toughness are deteriorated. Therefore, the Zr content is preferably 0.010% or less. The amount of Zr is more preferably 0.0070% or less, still more preferably 0.0050% or less, and still more preferably 0.0030% or less.
上記成分組成となるように溶製した後、溶鋼は、取鍋、タンディッシュを経て鋳型に注入されるが、本発明で規定の鋼板表層部を有する鋼板を得るには、上記タンディッシュに溶鋼を注入し連続鋳造を行う工程において、下記(1)~(3)の全てを満たすことが推奨される。
(1)タンディッシュにおいて、取鍋からの溶鋼注入位置での流路断面積よりも、鋳型への溶鋼注入位置における流路断面積が大きくなるようにする。具体的には、各流路断面積がこの様に設計されたタンディッシュを用いる。
(2)注入ノズルの吐出孔上部から50mm以上の位置からArを0.04~9.7L(リットル)/t(ton)の流量で吹き込みつつ、鋳造する。
(3)鋳型内溶鋼のメニスカス位置から引き抜き方向に向かって1~3mの位置の凝固速度を0.26mm/s以下とする。 〔Production method〕
After melting to have the above component composition, the molten steel is poured into a mold through a ladle and tundish, but in order to obtain a steel sheet having a specified steel sheet surface layer portion in the present invention, the molten steel is poured into the tundish. It is recommended to satisfy all of the following (1) to (3) in the process of injecting and continuously casting.
(1) In the tundish, the flow path cross-sectional area at the molten steel injection position into the mold is made larger than the flow path cross-sectional area at the molten steel injection position from the ladle. Specifically, a tundish in which each channel cross-sectional area is designed in this way is used.
(2) Casting while blowing Ar at a flow rate of 0.04 to 9.7 L (liter) / t (ton) from a position of 50 mm or more from the upper part of the discharge hole of the injection nozzle.
(3) The solidification rate at a position of 1 to 3 m from the meniscus position of the molten steel in the mold in the drawing direction is set to 0.26 mm / s or less.
(1)流路断面積
Ca系介在物は高融点であり、溶鋼との接触角が大きいため凝集合体を形成しやすく粗大な介在物となりやすい。よってこのCa系介在物をタンディッシュ内部で十分に浮上分離させる必要がある。この浮上分離が不十分な場合には、例えば連続鋳造時の湾曲部で上記粗大なCa系介在物が浮上し、表層に集積しやすくなる。タンディッシュ内で上記介在物を十分に浮上分離させるには、タンディッシュ内での溶鋼平均流速を小さくするのがよい。溶鋼平均流速を小さくすることによって、浮上時間を長時間化でき、また取鍋注入時の乱流により浮上分離を促進させることができる。タンディッシュ内での溶鋼平均流速を小さくするには、タンディッシュにおける鋳型への溶鋼注入位置における流路断面積が、取鍋からの溶鋼注入位置での流路断面積よりも大きいタンディッシュを用いる。(鋳型への溶鋼注入位置における流路断面積)/(取鍋からの溶鋼注入位置での流路断面積)で表される比が1.00超であればよいが、上記比は好ましくは1.50以上である。尚、上記比の上限は5.0程度である。 The conditions (1) to (3) will be described in order below.
(1) Channel cross-sectional area Ca-based inclusions have a high melting point and have a large contact angle with molten steel, so that agglomerates are easily formed and coarse inclusions are easily formed. Therefore, it is necessary to sufficiently float and separate this Ca-based inclusion inside the tundish. If this floating separation is insufficient, for example, the coarse Ca-based inclusion floats at the curved portion during continuous casting, and tends to accumulate on the surface layer. In order to sufficiently float and separate the inclusions in the tundish, it is preferable to reduce the average molten steel flow velocity in the tundish. By reducing the average molten steel flow velocity, the ascent time can be extended, and levitation separation can be promoted by turbulent flow during ladle injection. To reduce the average molten steel flow velocity in the tundish, use a tundish where the cross-sectional area at the pouring position of the molten steel into the mold in the tundish is larger than the cross-sectional area at the pouring position of the molten steel from the ladle. . The ratio represented by (flow path cross-sectional area at the molten steel injection position into the mold) / (flow path cross-sectional area at the molten steel injection position from the ladle) may be more than 1.00, but the ratio is preferably 1.50 or more. The upper limit of the ratio is about 5.0.
ノズル内の溶鋼が非充満となる吐出孔上部から50mm以上の位置でArを吹込みつつ鋳造を行うことで、ノズルおよび鋳型内でCa系介在物とAr気泡を合体させ浮上分離を促進させることができる。この効果を得るには、Ar流量を0.04L/t以上とするのが好ましい。前記Ar流量は、より好ましくは0.10L/t以上、更に好ましくは0.20L/t以上である。一方、Ar流量が9.7L/tを上回る場合、鋼片表層にAr気泡が残存し、鋼板に欠陥として残存しやすくなる。よってAr流量は、9.7L/t以下とすることが好ましく、より好ましくは9.0L/t以下、更に好ましくは8.0L/t以下である。 (2) Ar blowing By casting while blowing Ar at a position of 50 mm or more from the upper part of the discharge hole where the molten steel in the nozzle is not filled, the Ca inclusions and Ar bubbles are combined in the nozzle and the mold. Floating separation can be promoted. In order to obtain this effect, the Ar flow rate is preferably 0.04 L / t or more. The Ar flow rate is more preferably 0.10 L / t or more, and still more preferably 0.20 L / t or more. On the other hand, when the Ar flow rate exceeds 9.7 L / t, Ar bubbles remain in the steel slab surface layer and easily remain as defects in the steel sheet. Therefore, the Ar flow rate is preferably 9.7 L / t or less, more preferably 9.0 L / t or less, and still more preferably 8.0 L / t or less.
一般に、凝固速度が大きい場合は、凝固界面近傍に存在する介在物が界面に取り込まれやすく、凝固速度が小さい場合は、介在物の一部が凝固界面から未凝固の中央部に押し出される。本発明では凝固速度を小さくすることによって、鋼板表層部に介在物が集積しないようにする。具体的には、本発明で対象とする「表面から深さ5mmまでの領域」が凝固する、鋳型内溶鋼のメニスカス位置から引き抜き方向に向かって1~3mの位置の、凝固速度を0.26mm/s以下とする。凝固速度は好ましくは0.22mm/s以下、より好ましくは0.18mm/s以下である。尚、凝固速度の下限値は、生産性等の観点からおおよそ0.05mm/sとなる。上記凝固速度は、冷却水の水量密度や鋳造速度の制御によって調整することができる。 (3) Solidification rate In general, when the solidification rate is high, inclusions existing in the vicinity of the solidification interface are easily taken into the interface, and when the solidification rate is low, some of the inclusions are unsolidified from the solidification interface to the central part. Extruded. In the present invention, by reducing the solidification rate, inclusions are prevented from accumulating in the steel sheet surface layer. Specifically, the solidification rate is 0.26 mm at a position 1 to 3 m from the meniscus position of the molten steel in the mold where the “region from the surface to a depth of 5 mm”, which is the object of the present invention, solidifies. / S or less. The solidification rate is preferably 0.22 mm / s or less, more preferably 0.18 mm / s or less. The lower limit of the solidification rate is approximately 0.05 mm / s from the viewpoint of productivity and the like. The solidification speed can be adjusted by controlling the water density of the cooling water and the casting speed.
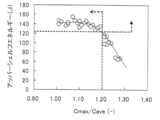
鋼板の板厚方向に表面から深さ5mmまでの領域のCa濃度の分布を蛍光分光分析により測定した。具体的には、最初に鋼板のスケール層を剥離するため鋼板表面から0.5mmまでを研削し、鋼板の表面に相当する該研削面のCa濃度を測定した。次いで、板厚方向に0.5mm研削してから該研削面のCa濃度測定を行った。これを板厚方向に0.5mmピッチで繰り返し行い、表面から板厚方向に深さ5mmまでの計10断面のCa濃度を測定した。そして10断面におけるCa濃度の最大値をCmax、10断面のCa濃度の平均値をCaveとし、Cmax/Caveを求めた。 [Measurement of Cmax / Cave]
The distribution of Ca concentration in the region from the surface to a depth of 5 mm in the plate thickness direction of the steel plate was measured by fluorescence spectroscopic analysis. Specifically, in order to first peel off the scale layer of the steel sheet, 0.5 mm from the steel sheet surface was ground, and the Ca concentration of the ground surface corresponding to the steel sheet surface was measured. Next, after grinding 0.5 mm in the plate thickness direction, the Ca concentration of the ground surface was measured. This was repeated at a pitch of 0.5 mm in the plate thickness direction, and the Ca concentration of 10 cross sections from the surface to the depth of 5 mm was measured. Cmax / Cave was determined by setting Cmax as the maximum value of Ca concentration in 10 cross sections and Cave as the average value of Ca concentration in 10 cross sections.
HIC試験は、NACE standard TM0284-2003に従って実施・評価した。詳細には、各鋼板の幅方向における1/4W位置と1/2W位置から、それぞれ3本、計6本の試験片(サイズ:板厚×(幅)100mm×(圧延方向)20mm)を採取した。そして該試験片を、1atmの硫化水素を飽和させた25℃の0.5%NaClと0.5%酢酸を含む混合水溶液中に96時間浸漬し、断面評価をNACE standard TM0284-2003 FIGURE3に従って行い、CLR(Crack Length Ratio、試験片幅に対する割れ長さ合計の割合(%)、割れ長さ率)を測定した。そして、前記CLRが3%以下の場合を耐HIC性に優れる(○)と評価し、CLRが3%超の場合を耐HIC性に劣る(×)と評価した。 [HIC test (NACE test)]
The HIC test was performed and evaluated according to NACE standard TM0284-2003. Specifically, three test pieces (size: plate thickness x (width) 100 mm x (rolling direction) 20 mm) were collected from each 1/4 W position and 1/2 W position in the width direction of each steel sheet. did. Then, the test piece was immersed in a mixed aqueous solution containing 0.5% NaCl and 0.5% acetic acid at 25 ° C. saturated with 1 atm of hydrogen sulfide for 96 hours, and the cross section was evaluated according to NACE standard TM0284-2003 FIGURE3. , CLR (Crack Length Ratio, ratio (%) of crack length to crack width, crack length ratio) was measured. And when the said CLR was 3% or less, it evaluated that it was excellent in HIC resistance ((circle)), and the case where CLR was over 3% was evaluated as inferior to HIC resistance (x).
NACE試験後、試験片の表面直下よりASTM A370に従い、板厚方向5mm×圧延方向10mmのシャルピー試験片を圧延方向に垂直な方向で3本採取し、鋼板の板厚方向にノッチを施した。シャルピー衝撃試験はASTM A370に従い実施し、試験温度は0℃~80℃まで種々変化させ、脆性破面率が0%でのシャルピー吸収エネルギー、つまりアッパーシェルフエネルギーを求めた。そして、このアッパーシェルフエネルギーが125J以上の場合を靭性に優れると評価した。 [Charpy impact test]
After the NACE test, three Charpy test pieces having a thickness of 5 mm and a rolling direction of 10 mm were sampled in a direction perpendicular to the rolling direction from just below the surface of the test piece in accordance with ASTM A370, and notched in the thickness direction of the steel sheet. The Charpy impact test was carried out according to ASTM A370, and the test temperature was changed in various ways from 0 ° C. to 80 ° C., and the Charpy absorbed energy, that is, the upper shelf energy when the brittle fracture surface ratio was 0% was determined. And it evaluated that the case where this upper shelf energy was 125J or more was excellent in toughness.
Claims (5)
- C:0.02~0.15%(%は質量%の意味。以下同じ)、
Si:0.02~0.50%、
Mn:0.6~2.0%、
P:0%超0.030%以下、
S:0%超0.003%以下、
Al:0.010~0.08%、
Ca:0.0003~0.0060%、
N:0.001~0.01%、および
O(酸素):0%超0.0045%以下を満たし、残部が鉄および不可避不純物からなり、
前記Caと前記Sの比(Ca/S)が2.0以上であり、かつ
板厚方向に表面から深さ5mmまでの領域の最大Ca濃度(Cmax)と該領域の平均Ca濃度(Cave)との比(Cmax/Cave)が1.20以下であることを特徴とする耐水素誘起割れ性と靭性に優れた鋼板。 C: 0.02 to 0.15% (% means mass%, the same applies hereinafter)
Si: 0.02 to 0.50%,
Mn: 0.6 to 2.0%,
P: more than 0% and 0.030% or less,
S: more than 0% and 0.003% or less,
Al: 0.010 to 0.08%,
Ca: 0.0003 to 0.0060%,
N: 0.001 to 0.01%, and O (oxygen): more than 0% and 0.0045% or less, with the balance consisting of iron and inevitable impurities,
The ratio of Ca to S (Ca / S) is 2.0 or more, and the maximum Ca concentration (Cmax) in the region from the surface to the depth of 5 mm in the plate thickness direction and the average Ca concentration (Cave) in the region A steel sheet excellent in hydrogen-induced crack resistance and toughness, wherein the ratio (Cmax / Cave) is 1.20 or less. - 更に他の元素として、以下の(a)、(b)の少なくともいずれかの群から選択される1種以上の元素を含有する請求項1に記載の鋼板。
(a)B:0%超0.005%以下、
V:0%超0.1%以下、
Cu:0%超1.5%以下、
Ni:0%超1.5%以下、
Cr:0%超1.5%以下、
Mo:0%超1.5%以下、および
Nb:0%超0.06%以下よりなる群
(b)Ti:0%超0.03%以下、
Mg:0%超0.01%以下、
REM:0%超0.02%以下、および
Zr:0%超0.010%以下よりなる群 Furthermore, the steel plate of Claim 1 which contains 1 or more types of elements selected from the group of at least any one of the following (a) and (b) as another element.
(A) B: more than 0% and 0.005% or less,
V: more than 0% and 0.1% or less,
Cu: more than 0% and 1.5% or less,
Ni: more than 0% and 1.5% or less,
Cr: more than 0% and 1.5% or less,
Mo: more than 0% and not more than 1.5%, and Nb: more than 0% and not more than 0.06% (b) Ti: more than 0% and not more than 0.03%,
Mg: more than 0% and 0.01% or less,
REM: a group consisting of more than 0% and 0.02% or less, and Zr: more than 0% and 0.010% or less - ラインパイプ用である請求項1または2に記載の鋼板。 The steel plate according to claim 1 or 2, which is for line pipes.
- 圧力容器用である請求項1または2に記載の鋼板。 The steel plate according to claim 1 or 2, which is for a pressure vessel.
- 請求項1または2に記載の鋼板を用いて製造されるラインパイプ用鋼管。 A steel pipe for a line pipe manufactured using the steel sheet according to claim 1 or 2.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020157026518A KR101709034B1 (en) | 2013-03-29 | 2014-03-25 | Steel plate with excellent hydrogen-induced cracking resistance and toughness, and steel pipe for line pipe |
| CN201480017953.7A CN105102652B (en) | 2013-03-29 | 2014-03-25 | The steel plate and line-pipes steel pipe of hydrogen induced cracking resistance and tenacity excellent |
| EP14773706.8A EP2980238B1 (en) | 2013-03-29 | 2014-03-25 | Steel plate with excellent hydrogen-induced cracking resistance and toughness, and line pipe steel tube |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013073310 | 2013-03-29 | ||
| JP2013-073310 | 2013-03-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014157165A1 true WO2014157165A1 (en) | 2014-10-02 |
Family
ID=51624136
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/058222 WO2014157165A1 (en) | 2013-03-29 | 2014-03-25 | Steel plate with excellent hydrogen-induced cracking resistance and toughness, and line pipe steel tube |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP2980238B1 (en) |
| JP (1) | JP6169025B2 (en) |
| KR (1) | KR101709034B1 (en) |
| CN (1) | CN105102652B (en) |
| WO (1) | WO2014157165A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016104529A1 (en) * | 2014-12-26 | 2016-06-30 | 株式会社神戸製鋼所 | Steel plate having excellent resistance to hydrogen-induced cracking, and steel pipe for line pipe |
| WO2016104528A1 (en) * | 2014-12-26 | 2016-06-30 | 株式会社神戸製鋼所 | Steel plate having excellent toughness and resistance to hydrogen-induced cracking, and steel pipe for line pipe |
| JP2016125140A (en) * | 2014-12-26 | 2016-07-11 | 株式会社神戸製鋼所 | Steel sheet and steel pipe for line pipe excellent in hydrogen-induced crack resistance and toughness |
| JP2016125139A (en) * | 2014-12-26 | 2016-07-11 | 株式会社神戸製鋼所 | Steel sheet and steel pipe for line pipe excellent in hydrogen-induced crack resistance |
| CN106636958A (en) * | 2015-07-16 | 2017-05-10 | 中国科学院金属研究所 | Cu-containing pipeline steel and enhanced heat treatment process thereof |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6211296B2 (en) * | 2013-04-30 | 2017-10-11 | 株式会社神戸製鋼所 | Steel plate with excellent sour resistance and HAZ toughness |
| JP6343472B2 (en) * | 2014-03-28 | 2018-06-13 | 株式会社神戸製鋼所 | Steel sheets for high-strength line pipes and steel pipes for high-strength line pipes with excellent low-temperature toughness |
| JP6584912B2 (en) * | 2014-12-26 | 2019-10-02 | 株式会社神戸製鋼所 | Steel plate and line pipe steel pipe with excellent hydrogen-induced crack resistance |
| JP2016125137A (en) * | 2014-12-26 | 2016-07-11 | 株式会社神戸製鋼所 | Steel sheet and steel pipe for line pipe excellent in hydrogen-induced crack resistance |
| CN105886909B (en) * | 2016-04-19 | 2017-08-11 | 江阴兴澄特种钢铁有限公司 | A kind of resistance against hydrogen cracking steel plate of pressure vessel and its manufacture method |
| JP6798565B2 (en) * | 2017-10-19 | 2020-12-09 | Jfeスチール株式会社 | High-strength steel sheets for sour-resistant pipes and high-strength steel pipes using them |
| CN109694991A (en) * | 2017-10-20 | 2019-04-30 | 鞍钢股份有限公司 | A kind of tank plate that hydrogen induced cracking resistance can be excellent |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS55128536A (en) * | 1979-03-28 | 1980-10-04 | Sumitomo Metal Ind Ltd | Preparation of steel plate with excellent resistance against hydrogen-induced cracking |
| JPH06136440A (en) | 1992-10-28 | 1994-05-17 | Nippon Steel Corp | Production of high strength steel sheet excellent in sour resistance |
| JP2000109947A (en) * | 1998-10-02 | 2000-04-18 | Nippon Steel Corp | Sour resistant steel plate excellent in toughness of heat affected zone and its production |
| JP2002348609A (en) * | 2001-03-23 | 2002-12-04 | Nkk Corp | Method for manufacturing steel with high strength for line pipe superior in hic resistance |
| JP2009136909A (en) * | 2007-12-10 | 2009-06-25 | Jfe Steel Corp | Method for producing continuously cast slab |
| JP2010209461A (en) | 2009-02-12 | 2010-09-24 | Nippon Steel Corp | High-strength steel sheet and high-strength steel pipe having excellent hydrogen-induced cracking resistance for use in line pipe |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5492511A (en) * | 1977-12-29 | 1979-07-21 | Kawasaki Steel Co | Steel having good hydrogenninduced crack resistivity |
| JPS607686B2 (en) * | 1978-02-16 | 1985-02-26 | 住友金属工業株式会社 | Manufacturing method for line pipe steel with excellent resistance to hydrogen-induced cracking |
| JPS55134155A (en) * | 1979-04-03 | 1980-10-18 | Nippon Steel Corp | Steel plate with superior hydrogen-induced crack resistance |
| JP2896198B2 (en) * | 1990-06-12 | 1999-05-31 | 川崎製鉄株式会社 | Casting method for steel with excellent resistance to hydrogen-induced cracking |
| JPH1121613A (en) * | 1997-05-07 | 1999-01-26 | Nippon Steel Corp | Production of steel excellent in toughness at low temperature and sour resistance |
| CN101082383A (en) * | 2006-05-29 | 2007-12-05 | 宝山钢铁股份有限公司 | Sulfurated hydrogen corrosion proof seamless pipe for oil-gas gathering and transporting |
| JP4725437B2 (en) * | 2006-06-30 | 2011-07-13 | 住友金属工業株式会社 | Continuous cast slab for thick steel plate, method for producing the same, and thick steel plate |
| JP5262075B2 (en) * | 2007-11-14 | 2013-08-14 | 新日鐵住金株式会社 | Method for producing steel for pipes with excellent sour resistance |
| JP2010116611A (en) * | 2008-11-13 | 2010-05-27 | Kobe Steel Ltd | Method for manufacturing low-sulfur thick steel plate excellent in haz toughness at the time of inputting large amount of heat |
| JP5540982B2 (en) * | 2010-08-09 | 2014-07-02 | 新日鐵住金株式会社 | Steel with excellent resistance to hydrogen-induced cracking |
| CN102719744B (en) * | 2012-06-25 | 2014-03-19 | 宝山钢铁股份有限公司 | Steel for low-temperature structures and manufacture method of steel |
-
2014
- 2014-03-14 JP JP2014052390A patent/JP6169025B2/en not_active Expired - Fee Related
- 2014-03-25 EP EP14773706.8A patent/EP2980238B1/en not_active Not-in-force
- 2014-03-25 CN CN201480017953.7A patent/CN105102652B/en not_active Expired - Fee Related
- 2014-03-25 WO PCT/JP2014/058222 patent/WO2014157165A1/en active Application Filing
- 2014-03-25 KR KR1020157026518A patent/KR101709034B1/en active IP Right Grant
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS55128536A (en) * | 1979-03-28 | 1980-10-04 | Sumitomo Metal Ind Ltd | Preparation of steel plate with excellent resistance against hydrogen-induced cracking |
| JPH06136440A (en) | 1992-10-28 | 1994-05-17 | Nippon Steel Corp | Production of high strength steel sheet excellent in sour resistance |
| JP2000109947A (en) * | 1998-10-02 | 2000-04-18 | Nippon Steel Corp | Sour resistant steel plate excellent in toughness of heat affected zone and its production |
| JP2002348609A (en) * | 2001-03-23 | 2002-12-04 | Nkk Corp | Method for manufacturing steel with high strength for line pipe superior in hic resistance |
| JP2009136909A (en) * | 2007-12-10 | 2009-06-25 | Jfe Steel Corp | Method for producing continuously cast slab |
| JP2010209461A (en) | 2009-02-12 | 2010-09-24 | Nippon Steel Corp | High-strength steel sheet and high-strength steel pipe having excellent hydrogen-induced cracking resistance for use in line pipe |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016104529A1 (en) * | 2014-12-26 | 2016-06-30 | 株式会社神戸製鋼所 | Steel plate having excellent resistance to hydrogen-induced cracking, and steel pipe for line pipe |
| WO2016104528A1 (en) * | 2014-12-26 | 2016-06-30 | 株式会社神戸製鋼所 | Steel plate having excellent toughness and resistance to hydrogen-induced cracking, and steel pipe for line pipe |
| JP2016125140A (en) * | 2014-12-26 | 2016-07-11 | 株式会社神戸製鋼所 | Steel sheet and steel pipe for line pipe excellent in hydrogen-induced crack resistance and toughness |
| JP2016125139A (en) * | 2014-12-26 | 2016-07-11 | 株式会社神戸製鋼所 | Steel sheet and steel pipe for line pipe excellent in hydrogen-induced crack resistance |
| CN107109595A (en) * | 2014-12-26 | 2017-08-29 | 株式会社神户制钢所 | The steel plate and line-pipes steel pipe of hydrogen induced cracking resistance and tenacity excellent |
| CN107109594A (en) * | 2014-12-26 | 2017-08-29 | 株式会社神户制钢所 | The excellent steel plate of hydrogen induced cracking resistance and line-pipes steel pipe |
| CN106636958A (en) * | 2015-07-16 | 2017-05-10 | 中国科学院金属研究所 | Cu-containing pipeline steel and enhanced heat treatment process thereof |
| CN106636958B (en) * | 2015-07-16 | 2018-09-04 | 中国科学院金属研究所 | One kind pipe line steel containing Cu and its reinforcing heat treatment process |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101709034B1 (en) | 2017-02-21 |
| JP6169025B2 (en) | 2017-07-26 |
| JP2014208892A (en) | 2014-11-06 |
| EP2980238A1 (en) | 2016-02-03 |
| EP2980238A4 (en) | 2016-11-23 |
| KR20150121193A (en) | 2015-10-28 |
| EP2980238B1 (en) | 2018-02-14 |
| CN105102652A (en) | 2015-11-25 |
| CN105102652B (en) | 2017-11-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6169025B2 (en) | Steel plates and line pipe steel pipes with excellent hydrogen-induced crack resistance and toughness | |
| JP6165088B2 (en) | Steel sheets and line pipe steel pipes with excellent resistance to hydrogen-induced cracking and toughness of weld heat affected zone | |
| JP5824401B2 (en) | Steel sheet with excellent resistance to hydrogen-induced cracking and method for producing the same | |
| JP6872616B2 (en) | Steel materials for pressure vessels with excellent hydrogen-induced cracking resistance and their manufacturing methods | |
| JP5618036B1 (en) | Thick steel plate excellent in multi-layer welded joint CTOD characteristics and method for producing the same | |
| JP4725437B2 (en) | Continuous cast slab for thick steel plate, method for producing the same, and thick steel plate | |
| JP6316548B2 (en) | Steel plates and line pipe steel pipes with excellent hydrogen-induced crack resistance and toughness | |
| CN109628841B (en) | Corrosion-resistant steel with yield strength of 350MPa for coal transportation open wagon and manufacturing method thereof | |
| JP5618037B1 (en) | Thick steel plate excellent in multi-layer welded joint CTOD characteristics and method for producing the same | |
| JP5445723B1 (en) | Ultra high strength steel plate for welding | |
| WO2017107779A1 (en) | Thick steel plate for high heat input welding and having great heat-affected area toughness and manufacturing method therefor | |
| US10837089B2 (en) | Thick steel plate for high heat input welding and having great heat-affected area toughness and manufacturing method therefor | |
| CN107287520A (en) | A kind of Ultra-low carbon composite rolling steel and sheet blank continuous casting production method | |
| KR102226990B1 (en) | Steel pipe for steel plate and line pipe, and manufacturing method thereof | |
| JP4959402B2 (en) | High strength welded structural steel with excellent surface cracking resistance and its manufacturing method | |
| JP4959401B2 (en) | High strength welded structural steel with excellent surface cracking resistance and its manufacturing method | |
| JP5857693B2 (en) | Steel plate for large heat input and manufacturing method thereof | |
| WO2018092605A1 (en) | Steel sheet, steel pipe for line pipe, and production method therefor | |
| JP6086036B2 (en) | Steel plate with excellent weld heat-affected zone toughness and its melting method | |
| CN116179952A (en) | Thick-specification phosphorus-containing hot-rolled weather-resistant steel plate and manufacturing method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480017953.7 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14773706 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20157026518 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014773706 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |