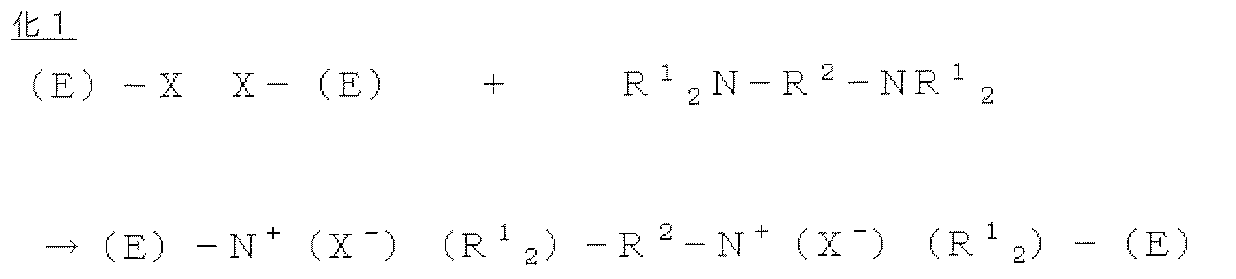WO2013129478A1 - 触媒電極層、及びその製造方法 - Google Patents
触媒電極層、及びその製造方法 Download PDFInfo
- Publication number
- WO2013129478A1 WO2013129478A1 PCT/JP2013/055141 JP2013055141W WO2013129478A1 WO 2013129478 A1 WO2013129478 A1 WO 2013129478A1 JP 2013055141 W JP2013055141 W JP 2013055141W WO 2013129478 A1 WO2013129478 A1 WO 2013129478A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- catalyst electrode
- quaternizing agent
- electrode layer
- copolymer
- catalyst
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/04—Processes of manufacture in general
- H01M4/0402—Methods of deposition of the material
- H01M4/0404—Methods of deposition of the material by coating on electrode collectors
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/8663—Selection of inactive substances as ingredients for catalytic active masses, e.g. binders, fillers
- H01M4/8673—Electrically conductive fillers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/8663—Selection of inactive substances as ingredients for catalytic active masses, e.g. binders, fillers
- H01M4/8668—Binders
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/88—Processes of manufacture
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/88—Processes of manufacture
- H01M4/8825—Methods for deposition of the catalytic active composition
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/88—Processes of manufacture
- H01M4/8825—Methods for deposition of the catalytic active composition
- H01M4/8828—Coating with slurry or ink
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/88—Processes of manufacture
- H01M4/8878—Treatment steps after deposition of the catalytic active composition or after shaping of the electrode being free-standing body
- H01M4/8892—Impregnation or coating of the catalyst layer, e.g. by an ionomer
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1004—Fuel cells with solid electrolytes characterised by membrane-electrode assemblies [MEA]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M2008/1095—Fuel cells with polymeric electrolytes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0065—Solid electrolytes
- H01M2300/0082—Organic polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/86—Inert electrodes with catalytic activity, e.g. for fuel cells
- H01M4/90—Selection of catalytic material
- H01M4/92—Metals of platinum group
- H01M4/925—Metals of platinum group supported on carriers, e.g. powder carriers
- H01M4/926—Metals of platinum group supported on carriers, e.g. powder carriers on carbon or graphite
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
Definitions
- the present invention relates to a novel catalyst electrode layer and a method for producing the catalyst electrode layer.
- the present invention also relates to a novel laminate having the catalyst electrode layer, and a novel polymer electrolyte fuel cell including the laminate.
- a fuel cell is a power generation system that takes out chemical energy of fuel as electric power, and several types of fuel cells such as alkaline type, phosphoric acid type, molten carbonate type, solid electrolyte type, and solid polymer type have been proposed and studied. Has been. Among these, the polymer electrolyte fuel cell is expected to be a small and medium-sized low-temperature operation fuel cell such as a stationary power source and an in-vehicle application because the operation temperature is particularly low.
- This solid polymer fuel cell is a fuel cell using a solid polymer such as an ion exchange resin as an electrolyte.
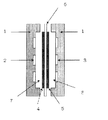
- the solid polymer fuel cell has a space in the battery partition wall 1 having a fuel flow hole 2 and an oxidant gas flow hole 3 communicating with the outside, respectively, in the solid polymer electrolyte membrane 6.
- the fuel chamber side catalyst electrode layer 4 and the oxidant chamber side catalyst electrode layer 5 are respectively joined to both sides by a joined body, and the fuel chamber 7 communicated with the outside through the fuel circulation hole 2 and the outside through the oxidant gas circulation hole 3. It has a basic structure in which an oxidant chamber 8 communicating with the oxidant chamber 8 is formed.
- fuel made of liquid such as hydrogen gas or alcohol is supplied to the fuel chamber 7 through the fuel flow hole 2 and the oxidant gas flow hole is supplied to the oxidant chamber 8.
- an oxygen-containing gas such as pure oxygen or air as an oxidant through 3 and connecting an external load circuit between the fuel chamber side catalyst electrode layer and the oxidant chamber side catalyst electrode layer, the following mechanism is used. Electric energy is generated.
- the solid polymer electrolyte membrane 6 use of an anion exchange membrane has been studied in that the reaction field becomes alkaline and a metal other than a noble metal can be used.
- hydrogen or alcohol or the like is supplied to the fuel chamber, and oxygen and water are supplied to the oxidant chamber, so that the catalyst contained in the electrode and the oxygen and water are contained in the oxidant chamber side catalyst electrode layer 5.
- hydroxide ions are produced.
- the hydroxide ions are transferred to the fuel chamber 7 through the solid polymer electrolyte membrane 6 (anion exchange membrane) made of the anion exchange membrane, and react with the fuel in the fuel chamber side catalyst electrode layer 4. Water is generated, and accompanying this, the electrons generated in the fuel chamber side catalyst electrode layer 4 are transferred to the oxidant chamber side catalyst electrode layer 5 through an external load circuit, and the energy of this reaction is transferred. Use as electrical energy.
- a catalyst electrode layer having a cross-linked structure is formed from a composition for forming a catalyst electrode including a precursor of an ion exchange resin into which an organic group having a halogen atom is introduced, a polyfunctional quaternizing agent, and a catalyst for an electrode. And a method of reacting the halogen atom with a polyfunctional quaternizing agent to form a catalyst electrode layer having a crosslinked structure.
- Patent Document 2 discloses that this method is used to bond an ion exchange membrane and a catalyst electrode layer with a crosslinked structure. According to these methods, it is possible to obtain a bonded body having excellent durability and excellent durability between the catalyst electrode layer and the ion exchange membrane.
- the degree of cross-linking necessarily depends on the blending amount of the polyfunctional quaternizing agent contained in the catalyst electrode forming composition. End up. That is, since the degree of crosslinking is determined when forming the catalyst electrode layer, in order to form catalyst electrode layers having various degrees of crosslinking, the catalyst electrode in which the blending amount of the polyfunctional quaternizing agent is changed each time. There was a need to prepare a forming composition.
- the polymer electrolyte fuel cell is expected to be used in various fields in the future. Since the operating conditions of the polymer electrolyte fuel cell naturally vary depending on the application, it is necessary to form a polymer electrolyte fuel cell (catalyst electrode layer) that is optimal for the operating conditions. Therefore, if it is possible to easily produce catalyst electrode layers having different degrees of crosslinking, it is possible to simplify the production of polymer electrolyte fuel cells suitable for various applications.
- an object of the present invention is to provide a catalyst electrode layer that is excellent in durability and that allows easy adjustment of the degree of crosslinking. Furthermore, the present invention provides a method for forming the catalyst electrode layer, a laminate in which the catalyst electrode layer and an anion exchange membrane are joined, a method for producing the laminate, and a polymer electrolyte fuel cell including the laminate. is there.
- the present inventors have intensively studied to solve the above problems. As a result, in consideration of the balance between durability and productivity, it has been found that a catalyst electrode layer composed of an anion conductive elastomer having a specific composition and a crosslinked structure, and a catalyst for an electrode can solve the above problems. It was.
- the present inventors when forming the catalyst electrode layer, the present inventors not only form a crosslinked structure simultaneously with the formation of the catalyst electrode layer, but also use a multifunctional quaternizing agent later to form the precursor layer of the catalyst electrode layer. It was considered that productivity could be improved if cross-linking (hereinafter, this cross-linking may be referred to as “post-crosslinking”). Then, the types of the polymer material, the cross-linking agent, the ion exchange group, etc. that become the matrix of the catalyst electrode layer were examined, and an anion conductive elastomer precursor in which a halogen atom-containing group was introduced into a copolymer having a specific composition. It has been found that the above-mentioned problems can be solved by bringing a polyfunctional quaternizing agent into contact with a catalyst electrode layer precursor comprising a body and a catalyst for an electrode, and the present invention has been completed.
- the first aspect of the present invention is that a double bond in the main chain is partially or by hydrogenating a copolymer of an aromatic vinyl compound and a conjugated diene compound, or a conjugated diene portion of the copolymer.
- a quaternary base type anion exchange group is introduced into at least a part of the aromatic ring of the all-saturated copolymer, and at least a part of the quaternary base type anion exchange group forms a crosslinked structure.
- a catalyst electrode layer comprising an anion conductive elastomer and an electrode catalyst.
- the ratio of the aromatic vinyl compound in the copolymer is 5 to 80 mass. % Is preferred.
- the quaternary base type anion exchange group which forms a crosslinked structure has a quaternary ammonium group and an alkylene group.
- the water content of the ion conductive elastomer at a temperature of 40 ° C. and a humidity of 90% RH is preferably 1 to 90%.
- the second aspect of the present invention is a laminate formed by forming the catalyst electrode layer described above on a gas diffusion layer or an anion exchange membrane
- the third aspect of the present invention is a polymer electrolyte fuel cell having the laminate.
- the fourth aspect of the present invention relates to a copolymer of an aromatic vinyl compound and a conjugated diene compound, or a double bond in the main chain partially or hydrogenated by hydrogenating the conjugated diene portion of the copolymer.
- the anionic conductive elastomer precursor is converted into quaternary by contacting the "functional group capable of reacting with quaternizing agent" with the polyfunctional quaternizing agent by contacting with the polyfunctional quaternizing agent.
- the catalyst electrode layer is produced by crosslinking with a base type anion exchange group.
- the “group capable of reacting with the quaternizing agent” introduced into the aromatic ring of the anion conductive elastomer precursor reacts with the polyfunctional quaternizing agent to thereby form an anion exchange group. And form a cross-linked structure.
- an anion conductive elastomer precursor having a “group capable of reacting with a quaternizing agent” and a polyfunctional quaternizing agent are compounds having one halogen atom at the terminal and the other corresponding organic group.
- a compound having an atom having a lone electron pair As a compound having an atom having a lone electron pair. And both form an anion exchange group in both the said atoms, and while forming an anion exchange group, a crosslinked structure is formed between both anion exchange groups.
- the anion conductive elastomer precursor and the polyfunctional quaternizing agent are bonded to each other to form an ion exchange group, both have different atoms. It is necessary. Therefore, when the anion conductive elastomer has a halogen atom at the terminal, the polyfunctional quaternizing agent needs to be a compound having an atom having a lone pair as a corresponding functional group. On the contrary, when the anion conductive elastomer precursor is a compound having an atom having a lone electron pair, the polyfunctional quaternizing agent needs to have a halogen atom as a corresponding functional group.
- the group capable of reacting with the quaternizing agent introduced into the aromatic ring is preferably a halogen atom-containing group, and the polyfunctional quaternizing agent is preferably an alkylenediamine compound.
- the catalyst electrode layer of the present invention has excellent catalytic performance, durability, and bondability as a catalyst electrode layer of a polymer electrolyte fuel cell. Therefore, the polymer electrolyte fuel cell provided with the catalyst electrode layer of the present invention is excellent in durability and can be used at a higher temperature.
- the catalyst electrode layer can be formed by post-crosslinking the catalyst electrode layer precursor, catalyst electrode layers having different degrees of crosslinking can be easily formed. As a result, it becomes possible to efficiently produce a polymer electrolyte fuel cell applied to various uses, and its industrial utility value is very high.
- This figure is a diagram showing an example of the structure of a polymer electrolyte fuel cell.
- the present invention relates to a copolymer in which a double bond in a main chain is partially or completely saturated by hydrogenating a copolymer of an aromatic vinyl compound and a conjugated diene compound, or a conjugated diene portion of the copolymer.
- a quaternary base type anion exchange group is introduced into at least a part of the aromatic ring of a polymer (hereinafter, these copolymers may be referred to as “styrene elastomer”), and the quaternary base type anion is introduced.
- a catalyst electrode layer comprising an anion conductive elastomer in which at least a part of exchange groups forms a crosslinked structure, and a catalyst for an electrode.
- the catalyst electrode layer of the present invention contains the anion conductive elastomer and an electrode catalyst. And this anion conductive elastomer partially bonds a double bond in the main chain by hydrogenating a copolymer of an aromatic vinyl compound and a conjugated diene compound or a conjugated diene portion of the copolymer.
- a quaternary base type anion exchange group is introduced into at least a part of the aromatic ring of the saturated copolymer, and at least a part of the quaternary base type anion exchange group forms a crosslinked structure. Become.
- the quaternary base type anion exchange group forms a cross-linked structure means that the aromatic rings of the styrene elastomer are bonded to each other with the quaternary base type anion exchange group.
- This anionic conductive elastomer is a copolymer of an aromatic vinyl compound and a conjugated diene compound, or by partially hydrogenating the double bond in the main chain by hydrogenating the conjugated diene portion of the copolymer. It can be obtained by reacting an anion conductive elastomer precursor having a halogen atom-containing group introduced into at least a part of the aromatic ring of the saturated copolymer and a polyfunctional quaternizing agent.
- a copolymer (styrene elastomer) that is a base resin will be described.
- the copolymer constituting the anion conductive elastomer is obtained by hydrogenating a copolymer of an aromatic vinyl compound and a conjugated diene compound, or a conjugated diene portion of the copolymer, to form a double chain in the main chain.
- the copolymer is not particularly limited, but is preferably a flexible polymer having a Young's modulus of 1 to 300 MPa, preferably 20 to 250 MPa, more preferably 30 to 200 MPa.
- this copolymer may be collectively used as a styrene elastomer.
- This Young's modulus is a value at 25 ° C. measured with a viscoelasticity measuring apparatus.
- the styrene elastomer may be a random copolymer of an aromatic vinyl compound and a conjugated diene compound, or may be a block copolymer.
- a block copolymer is preferable when post-crosslinking described later is performed.
- examples of the block form include a diblock copolymer, a triblock copolymer, and a multiblock copolymer. Among these, a triblock copolymer is preferable.
- Examples of the aromatic vinyl compound constituting the styrene elastomer include styrene, ⁇ -methylstyrene, chloromethylstyrene, bromobutylstyrene, vinylpyridine, vinylimidazole, vinyloxazoline, vinylbenzyldimethylamine, vinylnaphthalene and the like.
- chloromethyl styrene, bromobutyl styrene, vinyl pyridine, vinyl imidazole, vinyl oxazoline, vinyl benzyl dimethylamine it can be used as an anion conductive elastomer precursor by copolymerizing with conjugated diene. .
- halogen atom-containing group when the halogen atom-containing group is introduced later, considering the ease of introduction of the halogen atom-containing group, the ease of reaction of the halogen atom-containing group and the polyfunctional quaternizing agent, etc. Styrene and ⁇ -methylstyrene are preferred.
- conjugated diene compound examples include butadiene, isoprene, chloroprene, 1,3-pentadiene, 2,3-dimethyl-1,3-butadiene and the like.
- the content of the aromatic vinyl compound unit is not particularly limited, but is preferably 5 to 80% by mass, more preferably 10 to 50% by mass.
- the content of the aromatic vinyl compound unit satisfies the above range, it becomes easy to introduce a polyfunctional quaternizing agent described in detail below.
- monomers other than the aromatic vinyl compound and the conjugated diene compound can be blended within a range that does not impair the effects of the present invention.
- the number average molecular weight of the styrene elastomer is preferably from 5,000 to 300,000, more preferably from 10,000 to 200,000, particularly preferably from 2 to 150,000, and most preferably from 3 to 130,000.
- the hydrogenation rate is preferably 80% or more, particularly preferably 90% or more and 100% or less.
- the styrenic elastomer is obtained by copolymerizing an aromatic vinyl compound and a conjugated diene compound by a known method such as anionic polymerization, cationic polymerization, coordination polymerization, or radical polymerization.
- a styrenic elastomer produced by living anionic polymerization is preferred.
- Specific examples of the styrene elastomer include polystyrene-polybutadiene-polystyrene triblock copolymer (SBS) and polystyrene-polyisoprene-polystyrene triblock copolymer (SIS).
- polystyrene-poly (ethylene-butylene) -polystyrene triblock copolymer (SEBS) and polystyrene-poly (ethylene-propylene) -polystyrene triblock (SEPS) copolymer obtained by hydrogenation of SBS, SIS, etc., respectively. It is done.
- the anion conductive elastomer precursor is a polymer having a “group capable of reacting with a quaternizing agent” on the aromatic ring of the styrene elastomer.
- the anion conductive elastomer precursor can be classified into two types depending on the type of “group capable of reacting with a quaternizing agent”. One is a halogen atom-containing elastomer in which the “group capable of reacting with a quaternizing agent” is a halogen atom, and the other is an atom having a lone electron pair in which the “group capable of reacting with a quaternizing agent”. It is a lone electron pair-containing elastomer.
- Halogen atom-containing elastomer When a monomer having a halogen atom, for example, chloromethylstyrene is used as a monomer when polymerizing a styrene-based elastomer, a halogen atom-containing elastomer is an anion conductive elastomer precursor (halogen atom) as it is. Containing elastomer). Moreover, when the monomer which does not have a halogen atom is used, it can be set as an anion conductive elastomer precursor by introduce
- the method for introducing a halogen atom-containing group into the styrene elastomer is not particularly limited, and a known method may be employed. Specifically, a method in which the aromatic ring of styrene is reacted with formaldehyde and then halogenated, a method in which the aromatic ring of styrene is reacted with halogenomethyl ether, and an alkyl group is provided by a Grineer reaction after halogenating the aromatic ring of styrene. And a method of halogenating the alkyl chain terminal.
- the proportion of the halogen atom-containing group introduced into the styrene-based elastomer may be appropriately determined according to the desired ion exchange capacity and degree of crosslinking (density) of the anion conductive elastomer.
- a styrene-based elastomer having an aromatic vinyl compound unit content of 5 to 80% by mass, preferably 10 to 50% by mass is used, 50 to 100% by mol, more preferably 80%, of the aromatic ring.
- Halogen atoms are preferably introduced at ⁇ 100 mol%.
- two or more halogen atom-containing groups may be introduced into one aromatic ring.
- an anion conductive elastomer precursor corresponding to the structure can be produced.
- lone electron pair containing elastomer In the lone electron pair-containing elastomer, at least one organic residue is bonded to an atom having a lone pair such as nitrogen, sulfur, oxygen, phosphorus, selenium, tin, iodine, antimony, etc. present in the molecule.
- a cation type atom or atomic group is coordinated to form a cation (onium ion), and various types can be used.
- the atom having a lone pair of electrons is preferably nitrogen, phosphorus, or sulfur from the viewpoint of the usefulness of the ion exchange resin to be formed.
- nitrogen is preferable, and a high crosslinking density is obtained.
- This lone electron pair-containing elastomer is obtained by polymerizing the styrene-based elastomer, when vinyl pyridine, vinyl imidazole, vinyl oxazoline, vinyl benzyl dimethylamine is used as a monomer, the resulting polymer is an anion conductive elastomer as it is. It becomes a precursor (lone electron pair-containing elastomer). When a monomer having no lone pair is used, a substituent having an lone pair may be introduced into the resulting styrene elastomer.
- two or more atoms having a lone electron pair may be introduced in one aromatic ring.
- a monomer containing a group having two or more lone electron pairs in the aromatic ring as a starting material, an anion conductive elastomer precursor corresponding to the structure can be produced.
- An anion conductive elastomer can be obtained by crosslinking an anion conductive elastomer precursor such as the halogen atom-containing elastomer or the lone electron pair-containing elastomer with a polyfunctional quaternizing agent.
- the catalyst electrode layer of the present invention can be composed of an anion conductive elastomer precursor, a polyfunctional quaternizing agent, and an electrode catalyst.
- the anion conductive elastomer having a cross-linked structure can be synthesized by reacting the “group capable of reacting with a quaternizing agent” of the anion conductive elastomer precursor with a polyfunctional quaternizing agent. That is, an anion in which a “group capable of reacting with a quaternizing agent” of an anion conductive elastomer precursor and a polyfunctional quaternizing agent react to form a crosslinked structure by a quaternary base type anion exchange group.
- a conductive elastomer can be obtained.
- the polyfunctional quaternizing agent is a group that reacts with a group capable of reacting with the quaternizing agent (a halogen atom-containing group or a group having an atom having a lone electron pair) to form an anion exchange group.
- a group capable of reacting with the quaternizing agent a halogen atom-containing group or a group having an atom having a lone electron pair
- E is an anion conductive elastomer (halogen atom-containing elastomer)
- X is a halogen atom bonded to the aromatic ring of E
- R 1 is an alkyl group
- R 2 is an alkylene group
- N is a nitrogen atom.
- the polyfunctional quaternizing agent is different depending on whether the anion conductive elastomer is a halogen atom-containing elastomer or a lone electron pair-containing elastomer.
- the polyfunctional quaternizing agent used when the anion conductive elastomer is a halogen atom-containing elastomer will be described.
- Multifunctional quaternizing agent for halogen atom-containing elastomer As this polyfunctional quaternizing agent, a nitrogen-containing compound having two or more amino groups, and a phosphorus-containing compound having two or more phosphino groups such as bis (dimethylphosphino) propane and bis (diphenylphosphino) propane And compounds having two or more thio groups such as bis (methylthio) methane and bis (phenylthio) methane as the sulfur-containing compound, preferably diamine, triamine, tetraamine, particularly preferably diamine. .
- polyamine compound of diamine, triamine, and tetraamine for example, a compound described in Citation 2 (International Publication No. WO2007 / 072842 pamphlet) can be used.
- polyamine compounds it is preferable to use an alkylenediamine compound because it has good chemical stability after formation of a crosslinked structure and has an appropriate flexibility.
- alkylene diamine compound examples include compounds represented by the following formula (1).
- each of R 3 , R 4 , R 5 , and R 6 is an alkyl group having 1 to 4 carbon atoms, preferably a methyl group, n is an integer of 1 to 15, preferably an integer of 2 to 8.
- alkylenediamine compound examples include the compounds described in Cited Document 2 (International Publication No. WO2007 / 072842 pamphlet).
- these compounds in particular, N, N, N ′, N′-tetramethyl-1,4-butanediamine, N, N, N ′, N′-tetramethyl-1,6-hexanediamine, N, N , N ′, N′-tetramethyl-1,8-octanediamine, N, N, N ′, N′-tetramethyl 1,10-decanediamine can be preferably used.
- the polyfunctional quaternizing agent used when the anion conductive elastomer is a lone electron pair-containing elastomer will be described.
- Multifunctional quaternizing agent for lone electron pair-containing elastomer As the polyfunctional quaternizing agent, a compound having two or more halogeno groups, preferably a dihalogeno compound, a trihalogeno compound or a tetrahalogeno compound, particularly preferably a dihalogeno compound is used.
- polyfunctional quaternizing agents examples include the compounds described in Citation 2 (International Publication No. WO2007 / 072842 pamphlet).
- the anion conductive elastomer precursor can be crosslinked only with the polyfunctional quaternizing agent, but the monofunctional quaternizing agent and the polyfunctional quaternizing agent can also be combined.
- This monofunctional quaternizing agent is a group that reacts with a group capable of reacting with the quaternizing agent (a halogen atom-containing group or a group having an atom having a lone electron pair) to form an anion exchange group. Compound.
- this monofunctional quaternizing agent is also different between the one used for the halogen atom-containing elastomer and the one used for the lone electron pair-containing elastomer.
- a tertiary alkyl trialkylamine, aromatic amine or the like is used as the monofunctional quaternizing agent.
- a trialkylamine having an alkyl group having 1 to 4 carbon atoms or an aromatic amine having a phenyl group is preferable to use a trialkylamine having an alkyl group having 1 to 4 carbon atoms or an aromatic amine having a phenyl group.
- Specific examples include trimethylamine, triethylamine, tripropylamine, tributylamine, diethylmethylamine, dipropylmethylamine, dibutylmethylamine, phenyldimethylamine, and phenyldiethylamine.
- Examples of the monofunctional quaternizing agent include alkyl halogen compounds. Of these, alkyl halogen compounds having an alkyl group having 1 to 2 carbon atoms are preferred. Specifically, alkyl halogen compounds such as methyl iodide, methyl bromide, methyl chloride, ethyl iodide, ethyl bromide, and ethyl chloride are preferred. Can be mentioned. A halogen compound having an aromatic group such as benzyl chloride can also be used.
- the anion conductive elastomer is obtained by reacting the anion conductive elastomer precursor, the polyfunctional quaternizing agent, and if necessary, the monofunctional quaternizing agent. is there.
- This anion conductive elastomer has a quaternary base type anion exchange group introduced into an aromatic ring in the molecule, and at least a part of the quaternary base type anion exchange group forms a crosslinked structure.
- a quaternary base type anion exchange group a quaternary ammonium group or a quaternary pyridinium group which is a strongly basic group in anion conductivity is preferable.
- a group that can react with a quaternizing agent that forms a quaternary base type anion exchange group, or a group having an atom having a lone pair of electrons is a primary to tertiary amino group, a pyridyl group, an imidazole group, a phosphonium group. And sulfonium groups, but primary to tertiary amino groups and pyridyl groups are preferred.
- this anion conductive elastomer has a moisture content at a temperature of 40 ° C. and a humidity of 90% RH in the moisture content measurement method described in detail in the following examples. It is preferably 1 to 90%, more preferably 10 to 60%.
- This water content has a correlation with the degree of cross-linking, and is low when the degree of cross-linking is high and high when the degree of cross-linking is low.
- the degree of crosslinking can be determined by measuring the moisture content of the catalyst electrode layer at a temperature of 40 ° C. and a humidity of 90% RH. Therefore, even when the catalyst electrode layer is formed by the post-crosslinking method described below, the degree of crosslinking (polyfunctional quaternary quaternary is determined by measuring the moisture content of the catalyst electrode layer at a temperature of 40 ° C. and a humidity of 90% RH. It is also possible to determine the use ratio of the agent. That is, since the electrode catalyst contained in the catalyst electrode layer hardly affects the moisture content, the degree of crosslinking can be determined by measuring the moisture content of the catalyst electrode layer.
- the degree of crosslinking determined from such a viewpoint is based on the total number of moles (Bm) of groups capable of reacting with the quaternizing agent in the anion conductive elastomer precursor.
- the ratio (Fm / Bm) of the total number of moles (Fm) of the group reacting with the group is preferably 0.005 to 0.30.
- the degree of crosslinking (Fm / Bm) is more preferably 0.01 to 0.25, and preferably 0.01 to 0.20. Further preferred is 0.02 to 0.20.
- the anion conductive elastomer used in the present invention has a partially or fully saturated conjugated diene compound portion, and this portion maintains hydrophobicity. Therefore, the anion conductive elastomer is hardly soluble in water even if the degree of cross-linking is low, so there is no risk of outflow or dropout due to water. Therefore, the anion conductive elastomer can maintain flexibility by reducing the degree of crosslinking, and can maintain physical strength.
- the anion conductive elastomer is not particularly limited, but has an ion exchange capacity of 0.5 to 10 mmol / g in order to impart good ion conductivity and further increase electrical efficiency. More preferably, it is preferably 1 to 8 mmol / g. This anion exchange capacity can be determined by measurement from the formed catalyst electrode layer.
- the water content and ion exchange capacity can be adjusted by the types and blending ratios of the polyfunctional quaternizing agent and the monofunctional quaternizing agent.
- the degree of crosslinking is preferably 0.005 to 0.30, more preferably 0.01 to 0.25, and more preferably 0.01 to 0.20. Is more preferable, and 0.02 to 0.20 is particularly preferable.
- Catalyst for electrode A known catalyst can be used as the electrode catalyst.
- metal particles such as platinum, gold, silver, palladium, iridium, rhodium, ruthenium, tin, iron, cobalt, nickel, molybdenum, tungsten, vanadium, or alloys thereof that promote hydrogen oxidation reaction and oxygen reduction reaction
- the particle size of the metal particles used as the catalyst is usually 0.1 to 100 nm, more preferably 0.5 to 10 nm. The smaller the particle size, the higher the catalyst performance. However, it is difficult to produce a material having a particle size of less than 0.5 nm.
- These catalysts may be used after being supported on a conductive agent in advance.
- the conductive agent is not particularly limited as long as it is an electronically conductive substance. For example, carbon black such as furnace black and acetylene black, activated carbon, graphite and the like are generally used alone or in combination. .
- the content of these catalysts is usually 0.01 to 10 mg / cm 2 , more preferably 0.1 to 5.0 mg / cm 2 in terms of metal weight per unit area when the catalyst electrode layer is in a sheet form. .
- thermoplastic resins are generally used, and examples of thermoplastic resins that can be suitably used include polytetrafluoroethylene, polyvinylidene fluoride, tetrafluoroethylene. -Perfluoroalkyl vinyl ether copolymer, polyether ether ketone, polyether sulfone, styrene / butadiene copolymer, acrylonitrile / butadiene copolymer and the like.
- the content of the binder is preferably 5 to 25% by weight of the electrode catalyst layer.
- a binder may be used independently and may mix and use 2 or more types.
- the catalyst electrode layer introduces a quaternary base type anion exchange group into the aromatic ring of the styrenic elastomer, and at least a part of the quaternary base type anion exchange group forms a crosslinked structure.
- An anion conductive elastomer, and a catalyst for the electrode are examples of the catalyst electrode layer.
- the catalyst electrode layer can be directly produced from a composition for forming a catalyst electrode layer containing an anion conductive elastomer precursor, a polyfunctional quaternizing agent, and an electrode catalyst.
- the catalyst electrode layer can be formed by roll molding or extrusion molding the catalyst electrode layer forming composition.
- the catalyst electrode layer-forming composition further containing a solvent is applied on some base material (for example, a gas diffusion layer made of an anion exchange membrane or porous carbon paper), and the solvent is dried to obtain a catalyst electrode. Layers can also be formed.
- the thickness of the catalyst electrode layer is not particularly limited, and may be determined as appropriate according to the intended use. In general, the thickness is preferably 0.1 to 50 ⁇ m, and more preferably 0.5 to 20 ⁇ m.
- the catalyst electrode layer can also be produced by the following method. Specifically, by forming a catalyst electrode precursor layer containing the anion conductive elastomer precursor and an electrode catalyst, and contacting the catalyst electrode precursor layer with a polyfunctional quaternizing agent The anion conductive elastomer precursor can be crosslinked with a quaternary base type anion exchange group to form a catalyst electrode layer.
- this method post-crosslinking method
- many catalyst electrode precursor layers of the same type are produced, and liquids containing polyfunctional quaternizing agents having different concentrations are brought into contact with the respective catalyst electrode layers.
- various catalyst electrode layers having different degrees of crosslinking can be formed.
- the catalyst electrode layer of the present invention can be formed by the above method. That is, it is considered that the composition of the styrenic elastomer used enables post-crosslinking. Since the styrenic elastomer is composed of a copolymer of an aromatic vinyl compound and a conjugated diene compound, the styrene elastomer has a flexible and good mobility portion in the polymer chain. Therefore, the polyfunctional quaternizing agent is easily brought into contact with the halogen atom-containing group of the aromatic ring.
- the styrenic elastomer is preferably a block copolymer
- the content of the aromatic vinyl compound unit is preferably 5 to 80% by mass, and 30 to 70%. A mass %% is more preferred.
- the matrix resin having a conventional cross-linked structure formed most of the polymer chain with the aromatic vinyl compound. Therefore, when the catalyst electrode layer is formed, that is, when the composition for forming a catalyst electrode does not contain an anion conductive polymer precursor (polymer having a halogen atom-containing group) and a polyfunctional quaternizing agent, a crosslinked structure Is considered to have failed to form.
- a catalyst electrode precursor layer can be formed by roll molding or extrusion molding the composition for a catalyst electrode precursor layer containing the anion conductive elastomer precursor and a catalyst for an electrode.
- the catalyst electrode precursor layer can also be formed by applying a catalyst electrode precursor layer composition further containing a solvent to the substrate and drying the solvent.
- the solvent is not particularly limited, and a single solution or a mixed solution of tetrahydrofuran, chloroform, dichloromethane, dimethylformamide, dimethyl sulfoxide, 1-propanol, toluene, benzene, ethyl acetate, acetone and the like can be used.
- composition for the catalyst electrode precursor layer containing the solvent may be formulated so that the layer can be formed by applying to a certain substrate.
- the total solid content of the anion conductive elastomer precursor and the electrode catalyst is preferably 100 parts by mass, and the solvent is preferably 1 to 20 parts by mass.
- the substrate on which the composition for the catalyst electrode precursor layer is applied is not particularly limited.
- the catalyst electrode precursor layer composition can be applied on a substrate made of an inorganic material such as glass.
- the base material and the catalyst electrode precursor layer may be separated, the obtained catalyst electrode precursor layer may be laminated on the anion exchange membrane, pressed, etc., and bonded.
- the catalyst electrode precursor layer composition can be applied on an anion exchange membrane, or a support layer that supports the catalyst electrode layer, for example, a porous layer. It can also be applied on a porous substrate. What was apply
- anion exchange membrane When using an anion exchange membrane as a substrate, a known anion exchange membrane can be used. Among these, it is preferable to use a hydrocarbon-based anion exchange membrane. Specifically, a chloromethylstyrene-divinylbenzene copolymer, a vinylpyridine-divinylbenzene copolymer or the like is filled with an ion exchange resin into which a desired anion exchange group has been introduced by treatment such as amination or alkylation. A membrane is mentioned. These anion exchange resin membranes are generally supported by a woven fabric, nonwoven fabric, porous membrane, etc. made of thermoplastic resin by a base material, but have low gas permeability and can be thinned.
- the base material is made of a thermoplastic resin such as a polyolefin resin such as polyethylene, polypropylene, or polymethylpentene, or a fluorine resin such as polytetrafluoroethylene, poly (tetrafluoroethylene-hexafluoropropylene), or polyvinylidene fluoride. It is preferable to use a substrate made of a film. Further, the film thickness of these hydrocarbon-based anion exchange membranes is preferably from 5 to 200 ⁇ m, more preferably from the viewpoint of keeping electric resistance low and imparting mechanical strength necessary as a support membrane. Has a thickness of 8 to 150 ⁇ m.
- a bonding layer made of the anion conductive elastomer may be laminated on the anion exchange membrane.
- the thickness of the bonding layer is preferably 0.1 to 10 ⁇ m.
- a porous membrane that supports the catalyst electrode layer and can be used as a gas diffusion layer can also be used.
- the porous substrate is not particularly limited, but is preferably a carbon porous membrane.
- carbon fiber woven fabric, carbon paper, or the like can be used.
- the thickness of the support layer is preferably 50 to 300 ⁇ m, and the porosity is preferably 50 to 90%.
- this carbon porous membrane is preferably used when the catalyst electrode layer is formed by post-crosslinking. The reason is that after the catalyst electrode precursor layer is formed, the catalyst electrode precursor layer is brought into contact with the polyfunctional quaternizing agent, but the carbon porous membrane does not undergo deformation such as swelling. Because.
- the thickness of the catalyst electrode precursor layer is not particularly limited, and may be adjusted so as to be the thickness of the target catalyst electrode layer. Since the thicknesses of the catalyst electrode layer and the catalyst electrode precursor layer are substantially the same, the thickness of the catalyst electrode precursor layer is preferably 0.1 to 50 ⁇ m, and more preferably 0.5 to 20 ⁇ m. Is preferred.
- the catalyst electrode precursor layer composition is applied onto a support layer (carbon porous membrane) functioning as a gas diffusion layer or a solid polymer electrolyte membrane (anion exchange membrane).
- a support layer carbon porous membrane
- anion exchange membrane organic solvent
- the solvent is dried to form a catalyst electrode precursor layer on the carbon porous membrane.
- the anion conductive elastomer precursor in the catalyst electrode precursor layer is further crosslinked with a quaternary base type anion exchange group by bringing the catalyst electrode precursor layer into contact with the polyfunctional quaternizing agent. .
- the material to be used it is preferable to use the catalyst electrode precursor layer composition and the polyfunctional quaternizing agent as described above. Especially, it is preferable to make a catalyst electrode precursor layer and a polyfunctional quaternizing agent contact with the following mixture ratios.
- the contact between the catalyst electrode precursor layer and the polyfunctional quaternizing agent is not particularly limited, and the catalyst electrode precursor layer is placed in the polyfunctional quaternizing agent diluted in a solvent as necessary.
- Examples thereof include a dipping method and a method of spraying a polyfunctional quaternizing agent on the catalyst electrode precursor layer. Among these, it is preferable to employ a method of dipping.
- the conditions for the immersion may be appropriately determined according to the styrene-based elastomer to be used, the polyfunctional quaternizing agent, and the degree of crosslinking.
- the immersion may be performed at 10 to 50 ° C. for 4 to 48 hours.
- a solvent can be used for the immersion, and as this solvent, a solvent that does not react with a group capable of reacting with a quaternizing agent or a group having an atom having a lone pair of electrons, such as tetrahydrofuran, acetone, And toluene.
- the polyfunctional quaternizing agent is a liquid, it is not necessary to use a solvent.
- a monofunctional quaternizing agent can also be used when contacting the catalyst electrode precursor layer and the polyfunctional quaternizing agent.
- the amount of polyfunctional quaternizing agent used is the polyfunctional quaternizing agent used, the type of “group capable of reacting with quaternizing agent”, and the desired degree of crosslinking. What is necessary is just to determine suitably according to an ion exchange capacity.
- the anion conductive elastomer precursor has 4 N1 is preferably 0.01 to 10 mol, more preferably 0.01 to 2 mol, per 1 mol of the group capable of reacting with the classifier.
- the use amount (preparation amount) of the polyfunctional quaternizing agent is as described above in consideration of the reactivity between the polyfunctional quaternizing agent used and the “group capable of reacting with the quaternizing agent”. It is preferable to adjust appropriately so as to satisfy the degree of crosslinking (Fm / Bm).
- a method of bringing a mixture of a polyfunctional quaternizing agent and a monofunctional quaternizing agent into contact with the catalyst electrode precursor layer A method of contacting a monofunctional quaternizing agent after contacting the monofunctional quaternizing agent, and a method of contacting a monofunctional quaternizing agent after contacting the multifunctional quaternizing agent Any of these methods may be adopted.
- the amount of the monofunctional quaternizing agent used may be determined in consideration of the ratio with the polyfunctional quaternizing agent used together depending on the desired degree of crosslinking.
- the degree of crosslinking when actually reacted is the number of functional groups contained in the polyfunctional quaternizing agent, the difference in reactivity between the monofunctional quaternizing agent and the polyfunctional quaternizing agent, etc. Therefore, in order to obtain a desired degree of crosslinking, it is preferable to determine after a few trials by changing the ratio of the monofunctional quaternizing agent to the polyfunctional quaternizing agent.
- the formed catalyst electrode layer contains a catalyst for the electrode.
- the amount of the catalyst is known, the water content of the anion conductive elastomer is obtained by taking the amount of the catalyst into consideration.
- the use ratio of the polyfunctional quaternizing agent can be estimated.
- the blending amount of the catalyst is unknown, the ash content of the catalyst electrode layer coincides with the blending amount of the catalyst, so that the ash content may be measured.
- the usage amount of the polyfunctional quaternizing agent and the monofunctional quaternizing agent used as necessary reacts with the quaternizing agent possessed by the anion conductive elastomer precursor. It is preferable that the amount is equal to or greater than the group to be obtained.
- the catalyst electrode layer (anion conductive elastomer) at a temperature of 40 ° C. and a humidity of 90% RH as described in the section of the anion conductive elastomer.
- the water content is preferably 1 to 90%, more preferably 10 to 60%.
- the ion exchange capacity of the catalyst electrode layer (anionic conductive elastomer) is preferably 0.5 to 10 mmol / g, more preferably 1 to 8 mmol / g. In order to satisfy these requirements, it is preferable to appropriately adjust the types and usage ratios of the polyfunctional quaternizing agent and the monofunctional quaternizing agent.
- the excess polyfunctional quaternizing agent may be removed by a washing operation.
- the counter ion when it is a halogen atom, it can be converted into a hydroxide ion, bicarbonate ion, carbonate ion or the like.
- the conversion method is not particularly limited, and a known method can be employed. After conversion of the counter ions, excess ions may be removed by washing.
- a polymer electrolyte fuel cell can be assembled with the configuration shown in FIG.
- a catalyst electrode layer is formed on a support layer that functions as a gas diffusion layer, two of them are used to sandwich an ion exchange membrane on the side on which the catalyst electrode layer is formed. As a result, a state in which 4, 5, and 6 in FIG. 1 are combined can be realized.
- the support carbon porous membrane
- the support functions as it is or as a gas diffusion layer in order to make the gas diffusivity good. Can be used by overlaying on top of each other.
- FIG. 1 the configuration of FIG. 1 is taken as an example, and the case of hydrogen fuel is taken as an example.
- Electric power can be generated by supplying humidified hydrogen gas to the fuel chamber side and humidified oxygen or air to the air electrode side. Since there is an optimum value for each flow rate, it is possible to measure the voltage value or the current value when a certain load is applied, and set them so that they are the largest. Humidification is performed to prevent the ion exchange membrane and the catalyst electrode layer from drying out and lowering the ionic conductivity, which can also be optimized in the same manner. The higher the reaction temperature in the fuel cell, the higher the output. However, the higher the temperature, the more the deterioration of the ion exchange membrane and the catalyst electrode layer is promoted.
- the catalyst electrode layer includes a catalyst and an ion conductive resin.
- the ion conductive resin is also swelled and deformed when heat is applied in the presence of moisture, which causes a problem that the output of the fuel cell is lowered in order to suppress diffusion of fuel and oxidant gas.
- the anion conductive elastomers are cross-linked, it is considered that such swelling deformation hardly occurs. Therefore, it has the characteristic that it can be used without degrading output performance even at high temperatures.
- Styrene elastomer which is a polystyrene-poly (ethylene-butylene) -polystyrene copolymer (Young's modulus at 25 ° C. 30 MPa, number average molecular weight 30,000, aromatic (styrene) content 30% by mass, hydrogenation rate 99%) 20 g was dissolved in 1000 ml of chloroform, 100 g of chloromethyl ethyl ether and 100 g of anhydrous tin chloride SnCl 4 were added under ice cooling, and the mixture was reacted at 100 ° C. for 3 hours. Next, after a polymer was precipitated using a large amount of methanol, it was separated and vacuum-dried to obtain an anion conductive elastomer precursor 1 that was chloromethylated.
- N, N, N ', N'-tetramethyl-1,6-butanediamine is used as the multifunctional quaternizing agent
- trimethylamine 13C isotope
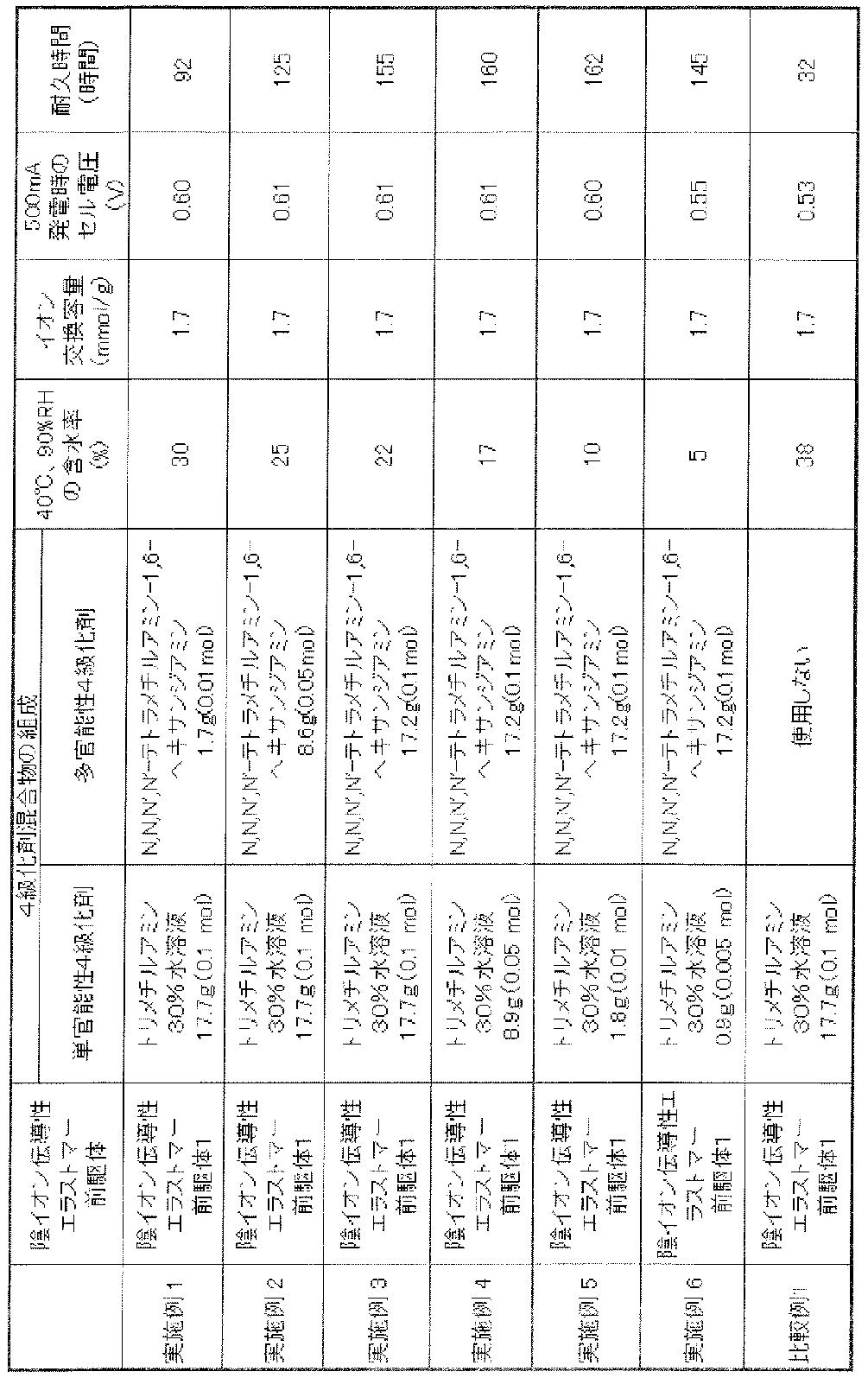
- the anion conductive elastomer precursor was immersed in a mixed solution in which the mixing ratio of these quaternizing agents was changed as shown in Table 1, and various catalyst electrode layers for preparing a calibration curve were prepared. The results are shown in Table 1.
- the measurement of the moisture content and the ion exchange capacity was performed by the same method as when the following laminate was measured.
- the degree of crosslinking can be known from the 13 C-NMR spectrum of the produced catalyst electrode layer.
- trimethylamine containing a 13 C isotope was used as the monofunctional quaternizing agent, so that the amount of trimethylamine contained by the 13 C-NMR spectrum can be quantified. It was obtained when the peak area of trimethylamine obtained from a catalyst electrode layer prepared using only trimethylamine ( 13 C isotope), which is a monofunctional quaternizing agent, was 1, and each catalyst electrode layer was measured. If the peak area is P, the degree of crosslinking can be calculated as 1 ⁇ P ( 13 C derived from other carbon atoms is a very small amount, and is set to 0 here). The obtained degree of crosslinking is also shown in Table 1.
- Example 1 (Laminated body production method: a method of laminating a catalyst electrode precursor layer on an ion exchange membrane and then forming a laminated body) 1 g of anion conductive elastomer precursor 1 is dissolved in 100 ml of chloroform and dispersed by adding 2 g of catalyst (platinum particles having a primary particle diameter of 30 to 50 nm supported on platinum particles having a particle diameter of 2 to 10 nm). To prepare a catalyst electrode precursor layer composition. This is 23 mm square (about 5 cm 2 ) on an anion exchange membrane (anion exchange capacity is 1.8 mmol / g, moisture content at 25 ° C. is 25% by mass, dry film thickness is 28 ⁇ m, outer size is 40 mm square).
- the membrane electrode assembly intermediate (anion exchange membrane / catalyst electrode precursor layer laminate structure) was obtained.
- the membrane electrode assembly intermediate was mixed with a monofunctional quaternizing agent (triethylamine 30% aqueous solution 17.7 g (triethylamine 0.1 mol)) and a polyfunctional quaternizing agent (N, N, N ′, N′—). It was immersed in a mixed solution of tetramethylamine-1,6-hexanediamine (1.7 g (0.01 mol)). After 48 hours, it was taken out, washed and dried to obtain a laminate. In the obtained laminate, the thickness of the catalyst electrode layer was 5 ⁇ m.
- an anion exchange membrane having the same area as that of the laminate is immersed in a 1 (mol / l) HCl aqueous solution for 10 hours or more to obtain a chlorine ion type, and then a nitrate ion type in a 1 (mol / l) NaNO 3 aqueous solution.
- the released chlorine ions were quantified by ion chromatography (ICS-2000, manufactured by Nippon Dionex Co., Ltd.). Let the quantitative value at this time be B (mol).
- the fuel cell was supplied with 100 ml / min of hydrogen humidified to 50 ° C. and 100% RH as the fuel gas and 200 ml / min of air humidified to 50 ° C. and 100% RH as the oxidant gas.
- the temperature of the fuel cell was 50 ° C.
- the voltage value when a current of 500 mA was taken out from this cell was measured.
- Table 2 shows the results of the above measurement results (water content, ion exchange capacity, power generation output test (cell voltage), power generation durability test (endurance time)).
- Example 2 The same operation as in Example 1 was performed except that a laminate was produced using the anion conductive elastomer precursor 1 and the monofunctional quaternizing agent and the polyfunctional quaternizing agent shown in Table 2. It was.
- the thickness of the catalyst electrode layer in the laminate was the same as in Example 1. After measuring the moisture content of the obtained laminate, it was incorporated into a fuel cell, and an output test and an endurance test were evaluated in the same manner as in Example 1. These measurement results are shown in Table 2.
- the water content was changed by changing the ratio of the quaternizing agent, and the value became smaller as the multifunctional quaternizing agent was increased. This is because the degree of cross-linking of the anion conductive elastomer increases as the polyfunctional quaternizing agent increases.
- the cell voltage during power generation was higher when the crosslinked structure was formed than when the crosslinked structure was not formed (see Comparative Example 1). This is considered to be because the formation of a crosslinked structure suppresses the swelling of the catalyst electrode layer, so that the fuel gas or oxidant gas easily reaches the catalyst surface, and the reaction necessary for power generation proceeds well. It is done. However, when the degree of crosslinking becomes too high, the value is higher than that of the non-crosslinked one (Comparative Example 1), but the cell voltage tends to decrease slightly (Examples 1 to 5 and Example 6). Comparison).
- Comparative Example 1 The same operation as in Example 1 was performed using the anion conductive elastomer precursor and the monofunctional quaternizing agent shown in Table 2. The results are shown in Table 2. The thickness of the layer containing the anion conductive elastomer comprising the anion conductive elastomer precursor and the monofunctional quaternizing agent and the catalyst was the same as in Example 1.
- the water content was higher than when using a multifunctional quaternizing agent. This is probably because it does not have a crosslinked structure.
- the cell voltage when generating power at 500 mA was smaller than in Examples 1-6. This is presumably because the absence of the cross-linking structure caused the elastomer to swell and the fuel gas or oxidant gas from reaching the catalyst surface, thereby suppressing the progress of the reaction necessary for power generation.
- Comparative Example 1 having no cross-linked structure the durability time was shorter than in Examples 1 to 6 having a cross-linked structure.
- water is supplied to the catalyst electrode layer, which is considered to swell.
- it does not have a cross-linked structure, it is considered that the catalyst electrode layer is deformed due to swelling, and the fuel gas or the oxidant gas hardly reaches the catalyst surface.
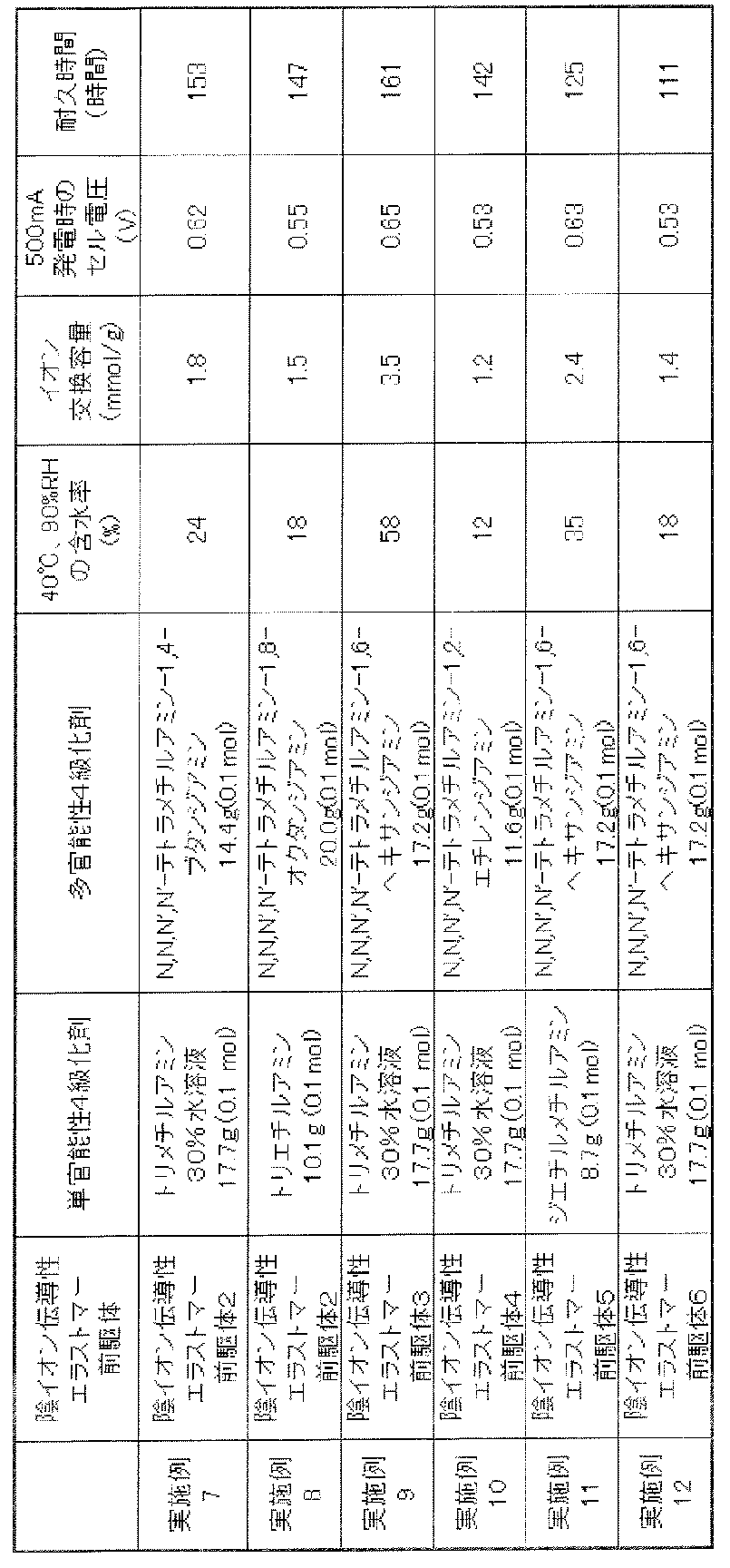
- Examples 7-12 1 g of the anion conductive elastomer precursor shown in Table 4 was dissolved in 100 ml of chloroform, and 2 g of catalyst (platinum particles having a particle diameter of 2 to 10 nm supported on carbon particles having a primary particle diameter of 30 to 50 nm) was added and dispersed to prepare a catalyst electrode precursor layer composition. This was coated on two carbon porous membranes (HGP-H-060 manufactured by Toray Industries, Inc., thickness: 200 ⁇ m: gas diffusion layer) cut to an outer size of 23 mm square, and then dried to provide a gas diffusion electrode intermediate. (Carbon porous membrane / laminated structure of catalyst electrode precursor layer) was obtained.
- HGP-H-060 manufactured by Toray Industries, Inc., thickness: 200 ⁇ m: gas diffusion layer
- the gas diffusion electrode intermediate was immersed in 10 ml of a mixture of monofunctional quaternizing agent and polyfunctional quaternizing agent shown in Table 4. After 48 hours, it was taken out, washed and dried to obtain a laminate. The thickness of the catalyst electrode layer in the laminate was the same as in Example 1.
- the same procedure as the above moisture content measurement method was used except that the carbon porous membrane (gas diffusion layer) used here was used, and the moisture content of the resulting laminate was measured. It was.
- the carbon porous membrane (gas diffusion layer) used here was used instead of the anion exchange membrane, the same operation as the above ion exchange capacity measurement method was performed, and the ion exchange capacity of the obtained laminate was determined. It was measured.
- the anion exchange membrane and the electrode assembly were sandwiched with the side where the anion conductive elastomer and the catalyst existed inside, and incorporated into the fuel cell.
- Performance evaluation was performed according to the above power generation output test method and power generation durability test method. The results are shown in Table 4.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Life Sciences & Earth Sciences (AREA)
- Sustainable Development (AREA)
- Sustainable Energy (AREA)
- Inert Electrodes (AREA)
- Fuel Cell (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
Abstract
Description
第三の本発明は、前記積層体を有する固体高分子型燃料電池である。
先ず、ベース樹脂である共重合体(スチレン系エラストマー)について説明する。
この陰イオン導電性エラストマーを構成する前記共重合体は、芳香族ビニル化合物と共役ジエン化合物との共重合体、又は該共重合体の共役ジエン部分を水素添加することによって主鎖中の2重結合を部分的に或いは全て飽和させた共重合体である。
本発明において、陰イオン伝導性エラストマー前駆体とは、前記スチレン系エラストマーの芳香環に「4級化剤と反応し得る基」を有するポリマーである。そして、この陰イオン伝導性エラストマー前駆体は、「4級化剤と反応し得る基」の種類によって2種類に分けることができる。1つは、「4級化剤と反応し得る基」がハロゲン原子であるハロゲン原子含有エラストマーであり、他方は、「4級化剤と反応し得る基」が孤立電子対を持つ原子である孤立電子対含有エラストマーである。
ハロゲン原子含有エラストマーは、スチレン系エラストマーを重合する際にハロゲン原子を有するモノマー、例えば、クロロメチルスチレンをモノマーに使用した場合には、得られる重合体がそのまま陰イオン伝導性エラストマー前駆体(ハロゲン原子含有エラストマー)となる。また、ハロゲン原子を有さないモノマーを使用した場合には、得られるスチレン系エラストマーにハロゲン原子含有基を導入することにより陰イオン伝導性エラストマー前駆体とすることができる。
孤立電子対含有エラストマーは、分子内に少なくとも一つ存在する窒素、硫黄、酸素、リン、セレン、錫、ヨウ素、アンチモン等の孤立電子対を持つ原子に少なくとも一つの有機残基が結合し、該原子に陽イオン型の原子又は原子団が配位してカチオン(オニウムイオン)を形成するものであれば特に限定されることはなく、各種のものが使用できる。
架橋構造を有する陰イオン伝導性エラストマーは、前記陰イオン伝導性エラストマー前駆体の「4級化剤と反応し得る基」と多官能性4級化剤とを反応させることにより合成できる。つまり、陰イオン伝導性エラストマー前駆体の「4級化剤と反応し得る基」と多官能性4級化剤とが反応し、4級塩基型陰イオン交換基による架橋構造を形成した陰イオン伝導性エラストマーを得ることができる。この多官能性4級化剤とは、4級化剤と反応し得る基(ハロゲン原子含有基、又は孤立電子対を持つ原子を有する基)と反応して陰イオン交換基を形成する基を複数有する化合物である。
この多官能性4級化剤としては、含窒素化合物として2以上のアミノ基を有する化合物、含リン化合物としてビス(ジメチルホスフィノ)プロパンやビス(ジフェニルホスフィノ)プロパン等の2以上のホスフィノ基を有する化合物、含硫黄化合物としてビス(メチルチオ)メタンやビス(フェニルチオ)メタン等の2以上のチオ基等を有する化合物が挙げられるが、好ましくはジアミン、トリアミン、テトラアミン、特に好ましくはジアミンが用いられる。
nは1~15の整数であり、好ましくは2~8の整数である。
次に、陰イオン伝導性エラストマーが孤立電子対含有エラストマーである場合に使用する多官能性4級化剤について説明する。
この多官能性4級化剤としては、2以上のハロゲノ基を有する化合物、好ましくはジハロゲノ化合物、トリハロゲノ化合物、テトラハロゲノ化合物、特に好ましくはジハロゲノ化合物が用いられる。
本発明においては、多官能性4級化剤のみで陰イオン伝導性エラストマー前駆体を架橋することもできるが、単官能性4級化剤と多官能性4級化剤とを組み合わせることもできる。この単官能性4級化剤は、4級化剤と反応し得る基(ハロゲン原子含有基、又は孤立電子対を持つ原子を有する基)と反応して陰イオン交換基を形成する基を1つ有する化合物である。この単官能性4級化剤も、当然のことながら、ハロゲン原子含有エラストマー用に使用するものと、孤立電子対含有エラストマー用に使用するものとで異なる。
この単官能性4級化剤としては、3級アミンであるトリアルキルアミン、芳香族アミン等が用いられる。中でも、炭素数が1~4のアルキル基を有するトリアルキルアミン、フェニル基を有する芳香族アミンを使用することが好ましい。具体的には、トリメチルアミン、トリエチルアミン、トリプロピルアミン、トリブチルアミン、ジエチルメチルアミン、ジプロピルメチルアミン、ジブチルメチルアミン、フェニルジメチルアミン、フェニルジエチルアミン等が挙げられる。
この単官能性4級化剤としては、アルキルハロゲン化合物が挙げられる。中でも、炭素数1~2のアルキル基を有するアルキルハロゲン化合物が好ましく、具体的には、ヨウ化メチル、臭化メチル、塩化メチル、ヨウ化エチル、臭化エチル、塩化エチル等のアルキルハロゲン化合物が挙げられる。また、ベンジルクロライドのような芳香族基を有するハロゲン化合物も使用できる。
本発明において、陰イオン伝導性エラストマーは、前記陰イオン伝導性エラストマー前駆体、前記多官能性4級化剤、及び必要に応じて前記単官能性4級化剤を反応させて得られるものである。
この電極用の触媒は、公知の触媒を使用することができる。例えば、水素の酸化反応及び酸素の還元反応を促進する白金、金、銀、パラジウム、イリジウム、ロジウム、ルテニウム、スズ、鉄、コバルト、ニッケル、モリブデン、タングステン、バナジウム、あるいはそれらの合金等の金属粒子が制限なく使用できるが、触媒活性が優れていることから白金族触媒を用いるのが好適である。
本発明において、触媒電極層は、前記スチレン系エラストマーの芳香環に4級塩基型陰イオン交換基を導入し、かつ、少なくとも一部の該4級塩基型陰イオン交換基が架橋構造を形成してなる陰イオン伝導性エラストマー、及び電極用の触媒を含んでなる。
また、本発明の方法によれば、触媒電極層は、以下の方法で製造することもできる。具体的には、前記陰イオン伝導性エラストマー前駆体、及び電極用の触媒を含む触媒電極前駆体層を形成し、該触媒電極前駆体層と多官能性4級化剤とを接触させることにより、該陰イオン伝導性エラストマー前駆体を4級塩基型陰イオン交換基により架橋して触媒電極層を形成することができる。この方法(後架橋方法)によれば、同じ種類の触媒電極前駆体層を多く製造し、それぞれの触媒電極層に濃度の異なる多官能性4級化剤含有液を接触させ、触媒電極層中の多官能性4級化剤の含有量を変えることにより、架橋度の異なる様々な触媒電極層を形成することが可能となる。
後架橋により触媒電極層を形成するためには、以下の方法を採用すればよい。先ず、触媒電極前駆体層を形成する方法について説明する。例えば、前記陰イオン伝導性エラストマー前駆体、電極用の触媒を含む触媒電極前駆体層用組成物をロール成形や押出成形することにより、触媒電極前駆体層を形成することができる。
先ず、ガス拡散層として機能する支持層(カーボン製多孔質膜)、または固体高分子電解質膜(陰イオン交換膜)上に、前記触媒電極前駆体層用組成物を塗布する。次いで、溶媒を乾燥し、カーボン製多孔質膜上に触媒電極前駆体層を形成する。さらに、該触媒電極前駆体層と多官能性4級化剤とを接触させることにより、触媒電極前駆体層中の陰イオン伝導性エラストマー前駆体を4級塩基型陰イオン交換基でより架橋する。
上記のようにして作製した、ガス拡散層として機能する支持層(カーボン製多孔質膜)、又は固体高分子電解質膜(陰イオン交換膜)の上に形成された触媒電極層を用いれば、例えば図1に示す構成にて固体高分子型燃料電池を組み立てることができる。
ポリスチレン-ポリ(エチレン-ブチレン)-ポリスチレン共重合体(25℃におけるヤング率 30MPa、数平均分子量30、000、芳香族(スチレン)含有量30質量%、水素添加率99%)であるスチレン系エラストマー20gを1000mlのクロロホルムに溶解し、クロルメチルエチルエーテル100g、無水塩化スズSnCl4 100gを氷冷下で添加した後、100℃で3時間反応させた。次に、大量のメタノールを用いて重合物を沈殿させた後に分離し、真空乾燥によりクロルメチル化された陰イオン伝導性エラストマー前駆体1を得た。
多官能性4級化剤としてN,N,N',N'-テトラメチル-1,6-ブタンジアミンを使用し、単官能性4級化剤としてトリメチルアミン(13C同位体のもの)を使用した。これら4級化剤の配合割合を表1のように変えた混合溶液中に、前記陰イオン伝導性エラストマー前駆体を浸漬させて、検量線作成用の触媒電極層を種々作製した。その結果を表1に示す。なお、含水率、イオン交換容量の測定は、下記の積層体を測定した際と同様の方法で実施した。
(積層体の作製方法:イオン交換膜上に触媒電極前駆体層を積層した後、積層体とする方法)
陰イオン伝導性エラストマー前駆体1を1gとりクロロホルム100mlに溶解し、触媒(一次粒径30~50nmのカーボン粒子上に、粒径2~10nmの白金粒子が担持されたもの)2gを加えて分散させ、触媒電極前駆体層用組成物を調製した。これを、陰イオン交換膜(陰イオン交換容量が1.8mmol/g、25℃における含水率が25質量%、乾燥膜厚は28μm、外寸40mm角)の上に23mm四方(約5cm2)にて塗布後、乾燥させ膜電極接合体中間体(陰イオン交換膜/触媒電極前駆体層の積層構造)を得た。該膜電極接合体中間体を、単官能性4級化剤(トリエチルアミン 30%水溶液 17.7g(トリエチルアミン0.1mol))と多官能性4級化剤(N,N,N’,N’-テトラメチルアミン-1,6-ヘキサンジアミン 1.7g(0.01mol))の混合溶液中に浸漬した。48時間後に取り出し、洗浄、乾燥し積層体を得た。得られた積層体において、触媒電極層の厚みは、5μmであった。
得られた積層体をバキュームオーブンに入れ、50℃、10mmHgの減圧下にて12時間乾燥し、その重量を測定した(W1とする)。さらにこのガス拡散電極を40℃、90%RHに調湿したグローブボックス中に12時間静置して水分を吸収させた後に、その重量を測定した(W2とする)。また、同様の操作で、積層体と同一面積の陰イオン交換膜のみの減圧乾燥後の重量(W3とする)、90%RH調湿後の重量(W4)を測定した。
このとき、含水率は、次式により求めた。
含水率 = (W2-W4-(W1-W3))÷ (W1-W3)
作製した積層体を1(mol/l)のHCl水溶液に10時間以上浸漬して塩素イオン型とした後、1(mol/l)のNaNO3 水溶液で硝酸イオン型に置換させ、遊離した塩素イオンをイオンクロマトグラフ(ICS-2000、日本ダイオネクス社製)で定量した。分析条件は以下のとおりである。
分析カラム:IonPac AS-17(日本ダイオネクス社製)
溶離液: 35(mmol/L) KOH水溶液 1ml/min
カラム温度: 35℃
この時の定量値をA(mol)とする。次に、同じ積層体を1(mol/l)HCl水溶液に4時間以上浸漬し、60℃で5時間減圧乾燥させその重量を測定した。この時の重量をW2(g)とする。
上記測定値に基づいて、イオン交換容量は次式により求めた。
イオン交換容量=(A―B)×1000/(W1-W2)[mmol/g-乾燥重量]
23mm角(約5cm2)に切断したカーボン製多孔質膜(東レ株式会社製 HGP-H-060、厚みは200μm)を2枚用いて、上記積層体の両面にある触媒電極層に一枚ずつ積層し、膜電極接合体(MEA)とした。このMEAを図1に示す燃料電池セルに組み込んだ。
燃料ガスとして50℃、100%RHに加湿した水素 100ml/minを、酸化剤ガスとして50℃、100%RHに加湿した空気 200ml/minを、燃料電池セルに供給した。燃料電池セルの温度は50℃とした。このセルから500mAの電流を取り出したときの電圧値を測定した。
燃料ガスとして80℃、100%RHに加湿した水素 100ml/minを、酸化剤ガスとして80℃、100%RHに加湿した空気 200ml/minを、燃料電池セルに供給した。燃料電池セルの温度は80℃とした。また、この状態で、電圧値が初期の1/2となるまでの時間を測定した。
陰イオン伝導性エラストマー前駆体1および、表2に示した単官能性4級化剤、多官能性4級化剤を用いて積層体を作製した以外は、実施例1と同様の操作を行った。積層体における触媒電極層の厚みは、実施例1と同じであった。得られた積層体の含水率を測定後、燃料電池セルに組み込み、出力試験、耐久試験を実施例1と同様の方法で評価を行った。これら測定結果を表2に示す。
表2に示した陰イオン伝導性エラストマー前駆体、及び単官能性4級化剤を用い、実施例1と同様の操作を行なった。その結果を表2に示した。陰イオン伝導性エラストマー前駆体と単官能性4級化剤とからなる陰イオン伝導性エラストマーと触媒とを含む層の厚みは、実施例1と同じであった。
表3に示したスチレン系エラストマーを用いて、陰イオン伝導性エラストマー前駆体1の合成方法と同じ操作を行い、陰イオン伝導性エラストマー前駆体2~6を得た。
表4に示した陰イオン伝導性エラストマー前駆体を1gとりクロロホルム100mlに溶解し、触媒(一次粒径30~50nmのカーボン粒子上に、粒径2~10nmの白金粒子が担持されたもの)2gを加えて分散させ、触媒電極前駆体層用組成物を調製した。これを、外寸23mm角に切断した2枚のカーボン製多孔質膜(東レ株式会社製 HGP-H-060、厚みは200μm:ガス拡散層)の上に塗布後、乾燥させガス拡散電極中間体(カーボン製多孔質膜/触媒電極前駆体層の積層構造)を得た。ガス拡散電極中間体を、表4に示した単官能性4級化剤および多官能性4級化剤の混合物10ml中に浸漬した。48時間後に取り出し、洗浄、乾燥し積層体を得た。積層体における触媒電極層の厚みは、実施例1と同じであった。
上記発電出力試験方法、発電耐久性試験方法にしたがい、性能評価を行なった。その結果をあわせて表4に示した。
2;燃料流通孔
3;酸化剤ガス流通孔
4;燃料室側触媒電極層(ガス拡散層を含む)
5;酸化剤室側触媒電極層(ガス拡散層を含む)
6;固体高分子電解質(陰イオン交換膜)
7;アノード室
8;カソード室
Claims (10)
- 芳香族ビニル化合物と共役ジエン化合物との共重合体、又は該共重合体の共役ジエン部分を水素添加することによって主鎖中の2重結合を部分的に或いは全て飽和させた共重合体の少なくとも一部の芳香環に4級塩基型陰イオン交換基を導入し、かつ、少なくとも一部の該4級塩基型陰イオン交換基が架橋構造を形成してなる陰イオン伝導性エラストマー、及び
電極用の触媒
を含むことを特徴とする触媒電極層。 - 前記共重合体における前記芳香族ビニル化合物の割合が、5~80質量%である請求項1に記載の触媒電極層。
- 温度40℃、湿度90%RHにおける前記陰イオン伝導性エラストマーの含水率が、1~90%である請求項1に記載の触媒電極層。
- 架橋構造を形成する4級塩基型陰イオン交換基が、第4級アンモニウム基、及びアルキレン基を有することを特徴とする請求項1に記載の触媒電極層。
- ガス拡散層上、又は陰イオン交換膜上に請求項1に記載の触媒電極層を形成してなる積層体。
- 請求項5に記載の積層体を有する固体高分子型燃料電池。
- 芳香族ビニル化合物と共役ジエン化合物との共重合体、又は該共重合体の共役ジエン部分を水素添加することによって主鎖中の2重結合を部分的に或いは全て飽和させた共重合体の少なくとも一部の芳香環に4級化剤と反応し得る基を導入した陰イオン伝導性エラストマー前駆体、及び電極用の触媒を含む触媒電極前駆体層と、多官能性4級化剤とを接触させることにより、4級化剤と反応し得る基と多官能性4級化剤とを反応させて該陰イオン伝導性エラストマー前駆体を4級塩基型陰イオン交換基により架橋することを特徴とする請求項1に記載の触媒電極層の製造方法。
- 前記芳香族ビニル化合物の割合が、5~70質量%である請求項7に記載の触媒電極層の製造方法。
- 芳香環に導入する4級化剤と反応し得る基がハロゲン原子含有基であり、多官能性4級化剤がアルキレンジアミン化合物であることを特徴とする請求項7に記載の触媒電極層の製造方法。
- 芳香族ビニル化合物と共役ジエン化合物との共重合体、又は該共重合体の共役ジエン部分を水素添加することによって主鎖中の2重結合を部分的に或いは全て飽和させた共重合体の少なくとも一部の芳香環に4級化剤と反応し得る基を導入した陰イオン伝導性エラストマー前駆体、及び電極用の触媒を含む触媒電極前駆体層を、ガス拡散層上、又は陰イオン交換膜上に形成した後、該触媒電極前駆体層と多官能性4級化剤とを接触させることを特徴とする請求項5に記載の積層体の製造方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/380,766 US20150017566A1 (en) | 2012-02-29 | 2013-02-27 | Catalyst Electrode Layer and Method for Producing Same |
| KR1020147020068A KR20140143134A (ko) | 2012-02-29 | 2013-02-27 | 촉매 전극층 및 그 제조 방법 |
| CN201380006848.9A CN104081569A (zh) | 2012-02-29 | 2013-02-27 | 催化电极层及其制造方法 |
| IN1765KON2014 IN2014KN01765A (ja) | 2012-02-29 | 2013-02-27 | |
| EP13755212.1A EP2822070A4 (en) | 2012-02-29 | 2013-02-27 | CATALYST ELECTRODE LAYER AND MANUFACTURING METHOD THEREFOR |
| JP2014502312A JP6066989B2 (ja) | 2012-02-29 | 2013-02-27 | 触媒電極層、及びその製造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012044209 | 2012-02-29 | ||
| JP2012-044209 | 2012-02-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013129478A1 true WO2013129478A1 (ja) | 2013-09-06 |
Family
ID=49082672
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/055141 WO2013129478A1 (ja) | 2012-02-29 | 2013-02-27 | 触媒電極層、及びその製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20150017566A1 (ja) |
| EP (1) | EP2822070A4 (ja) |
| JP (1) | JP6066989B2 (ja) |
| KR (1) | KR20140143134A (ja) |
| CN (1) | CN104081569A (ja) |
| IN (1) | IN2014KN01765A (ja) |
| WO (1) | WO2013129478A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014013879A1 (ja) * | 2012-07-20 | 2014-01-23 | 株式会社トクヤマ | 陰イオン交換膜型燃料電池用の触媒層及び膜-電極接合体並びにこれらを用いる陰イオン交換膜型燃料電池及びその運転方法 |
| WO2015122320A1 (ja) * | 2014-02-14 | 2015-08-20 | 株式会社トクヤマ | 部分四級化スチレン系共重合体、イオン伝導性付与剤、触媒電極層、膜-電極接合体及びその製造方法、ガス拡散電極及びその製造方法、並びに陰イオン交換膜型燃料電池 |
| JP2023521327A (ja) * | 2020-04-07 | 2023-05-24 | エナプター エス.アール.エル. | イオン交換膜およびイオン交換膜の製造方法 |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3221912B1 (en) | 2014-11-18 | 2024-04-03 | Rensselaer Polytechnic Institute | Novel polymers and methods for their manufacture |
| CN105702970B (zh) * | 2014-11-27 | 2019-07-02 | 中国科学院大连化学物理研究所 | 碱性阴离子交换膜燃料电池用立体化树脂溶液的制备 |
| CN106328958A (zh) * | 2015-07-06 | 2017-01-11 | 中国科学院大连化学物理研究所 | 一种碱性阴离子交换膜燃料电池膜电极的制备方法 |
| CN106887607A (zh) * | 2015-12-15 | 2017-06-23 | 中国科学院大连化学物理研究所 | 一种化学分子修饰的Pt基电催化剂及其制备和应用 |
| US10840521B2 (en) * | 2015-12-30 | 2020-11-17 | Mann+Hummel Gmbh | Humidifier, for example for a fuel cell |
| US20200238272A1 (en) | 2017-07-06 | 2020-07-30 | Rensselaer Polytechnic Institute | Ionic functionalization of aromatic polymers for ion exchange membranes |
| US20190060842A1 (en) | 2017-08-22 | 2019-02-28 | Daniel M. Knauss | Functionalized poly(diallylpiperidinium) and its copolymers for use in ion conducting applications |
| US10836874B2 (en) * | 2017-10-02 | 2020-11-17 | Colorado School Of Mines | High performance cross-linked triblock cationic functionalized polymer for electrochemical applications, methods of making and methods of using |
| CN112512668A (zh) * | 2018-04-24 | 2021-03-16 | 伦斯勒理工学院 | 用于阴离子交换膜的芳香族聚合物的交联 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH09223513A (ja) * | 1996-02-19 | 1997-08-26 | Kashimakita Kyodo Hatsuden Kk | 液循環式電池 |
| JPH11273695A (ja) * | 1998-03-19 | 1999-10-08 | Asahi Glass Co Ltd | 固体高分子電解質型メタノール燃料電池 |
| JP2002367626A (ja) * | 2001-06-12 | 2002-12-20 | Tokuyama Corp | ガス拡散電極用イオン伝導性付与剤 |
| JP2003086193A (ja) | 2001-09-13 | 2003-03-20 | Tokuyama Corp | ガス拡散電極の製造方法 |
| WO2007072842A1 (ja) | 2005-12-20 | 2007-06-28 | Tokuyama Corporation | 固体高分子型燃料電池用電解質膜-電極膜接合体及びその製造方法、並びにそれを具備した燃料電池 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7300719B2 (en) * | 2004-08-05 | 2007-11-27 | General Motors Corporation | Porous diffusion media and bipolar plate assembly with anion exchange resin and devices incorporating same |
| US8242042B2 (en) * | 2007-06-05 | 2012-08-14 | Tokuyama Corporation | OH-type anion-exchange hydrocarbon-based elastomer, use and production method thereof |
-
2013
- 2013-02-27 WO PCT/JP2013/055141 patent/WO2013129478A1/ja active Application Filing
- 2013-02-27 KR KR1020147020068A patent/KR20140143134A/ko not_active Application Discontinuation
- 2013-02-27 EP EP13755212.1A patent/EP2822070A4/en not_active Withdrawn
- 2013-02-27 CN CN201380006848.9A patent/CN104081569A/zh active Pending
- 2013-02-27 US US14/380,766 patent/US20150017566A1/en not_active Abandoned
- 2013-02-27 IN IN1765KON2014 patent/IN2014KN01765A/en unknown
- 2013-02-27 JP JP2014502312A patent/JP6066989B2/ja not_active Expired - Fee Related
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH09223513A (ja) * | 1996-02-19 | 1997-08-26 | Kashimakita Kyodo Hatsuden Kk | 液循環式電池 |
| JPH11273695A (ja) * | 1998-03-19 | 1999-10-08 | Asahi Glass Co Ltd | 固体高分子電解質型メタノール燃料電池 |
| JP2002367626A (ja) * | 2001-06-12 | 2002-12-20 | Tokuyama Corp | ガス拡散電極用イオン伝導性付与剤 |
| JP2003086193A (ja) | 2001-09-13 | 2003-03-20 | Tokuyama Corp | ガス拡散電極の製造方法 |
| WO2007072842A1 (ja) | 2005-12-20 | 2007-06-28 | Tokuyama Corporation | 固体高分子型燃料電池用電解質膜-電極膜接合体及びその製造方法、並びにそれを具備した燃料電池 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2822070A4 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014013879A1 (ja) * | 2012-07-20 | 2014-01-23 | 株式会社トクヤマ | 陰イオン交換膜型燃料電池用の触媒層及び膜-電極接合体並びにこれらを用いる陰イオン交換膜型燃料電池及びその運転方法 |
| WO2015122320A1 (ja) * | 2014-02-14 | 2015-08-20 | 株式会社トクヤマ | 部分四級化スチレン系共重合体、イオン伝導性付与剤、触媒電極層、膜-電極接合体及びその製造方法、ガス拡散電極及びその製造方法、並びに陰イオン交換膜型燃料電池 |
| CN105980418A (zh) * | 2014-02-14 | 2016-09-28 | 德山株式会社 | 部分季铵化苯乙烯系共聚物、离子传导性赋予剂、催化剂电极层、膜-电极组件及其制造方法、气体扩散电极及其制造方法以及阴离子交换膜型燃料电池 |
| KR20160122703A (ko) | 2014-02-14 | 2016-10-24 | 가부시키가이샤 도쿠야마 | 부분 4급화 스티렌계 공중합체, 이온전도성 부여제, 촉매전극층, 막-전극 접합체 및 그의 제조방법, 가스확산 전극 및 그의 제조방법, 및 음이온 교환막형 연료전지 |
| EP3106476A4 (en) * | 2014-02-14 | 2017-07-12 | Tokuyama Corporation | Partially quaternized styrene-based copolymer, ionic-conductivity imparter, catalytic electrode layer, membrane/electrode assembly and process for producing same, gas diffusion electrode and process for producing same, and fuel cell of anion exchange membrane type |
| JP2023521327A (ja) * | 2020-04-07 | 2023-05-24 | エナプター エス.アール.エル. | イオン交換膜およびイオン交換膜の製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2822070A1 (en) | 2015-01-07 |
| EP2822070A4 (en) | 2015-10-21 |
| IN2014KN01765A (ja) | 2015-10-23 |
| CN104081569A (zh) | 2014-10-01 |
| US20150017566A1 (en) | 2015-01-15 |
| KR20140143134A (ko) | 2014-12-15 |
| JPWO2013129478A1 (ja) | 2015-07-30 |
| JP6066989B2 (ja) | 2017-01-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6066989B2 (ja) | 触媒電極層、及びその製造方法 | |
| JP6077774B2 (ja) | イオン伝導性付与剤並びにカソード触媒層並びに該触媒層を用いて形成する膜−電極接合体並びに陰イオン交換膜型燃料電池及びその運転方法 | |
| WO2015122320A1 (ja) | 部分四級化スチレン系共重合体、イオン伝導性付与剤、触媒電極層、膜-電極接合体及びその製造方法、ガス拡散電極及びその製造方法、並びに陰イオン交換膜型燃料電池 | |
| EP1965456B1 (en) | Electrolyte membrane-electrode membrane assembly for solid polymer fuel cell, process for producing the same, and fuel cell comprising said assembly | |
| KR101590651B1 (ko) | 고체 고분자형 연료 전지용 격막, 및 격막-촉매 전극 접합체 | |
| JP2002367626A (ja) | ガス拡散電極用イオン伝導性付与剤 | |
| TW201019523A (en) | Method for producing anion exchange membrane for solid polymer electrolyte-type fuel cell | |
| JP4849893B2 (ja) | 陰イオン交換樹脂膜形成用4級化剤、ガス拡散電極膜、固体電解質膜及びそれを具備する固体高分子型燃料電池 | |
| WO2017022775A1 (ja) | 陰イオン交換体、イオン伝導性付与剤、触媒電極層、膜-電極接合体、陰イオン交換膜型燃料電池および陰イオン交換膜型水電解装置 | |
| JP2008004312A (ja) | イオン伝導性バインダー、膜−電極接合体及び燃料電池 | |
| WO2009148051A1 (ja) | 陰イオン交換膜型燃料電池における触媒電極層用イオン伝導性付与剤の製造方法 | |
| JP4749630B2 (ja) | ガス拡散電極の製造方法 | |
| JP2007194048A (ja) | イオン伝導性バインダー、膜−電極接合体及び燃料電池 | |
| JP2007042573A (ja) | 固体高分子型燃料電池用高分子電解質膜、膜−電極接合体及び燃料電池 | |
| JP5535724B2 (ja) | 触媒電極層用電解質材料及び電解質膜―触媒電極層接合体 | |
| JP2014044940A (ja) | イオン伝導性付与剤、アノード触媒層、該触媒層を用いて形成する膜−電極接合体、並びに陰イオン交換膜型燃料電池及びその運転方法 | |
| JP2008153175A (ja) | 固体高分子型燃料電池用膜−電極接合体の製造方法 | |
| JP2009158127A (ja) | 膜電極接合体 | |
| JPWO2007046239A1 (ja) | 燃料電池用電解質組成物、及びにそれを用いた燃料電池用膜−電極接合体、並びに燃料電池 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13755212 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20147020068 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2014502312 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14380766 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 234311 Country of ref document: IL Ref document number: 2013755212 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |