WO2012117909A1 - O/w乳化組成物の製造方法 - Google Patents
O/w乳化組成物の製造方法 Download PDFInfo
- Publication number
- WO2012117909A1 WO2012117909A1 PCT/JP2012/054239 JP2012054239W WO2012117909A1 WO 2012117909 A1 WO2012117909 A1 WO 2012117909A1 JP 2012054239 W JP2012054239 W JP 2012054239W WO 2012117909 A1 WO2012117909 A1 WO 2012117909A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- emulsified
- oil
- aqueous phase
- emulsion
- emulsion composition
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/06—Emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/06—Emulsions
- A61K8/062—Oil-in-water emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/042—Gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/342—Alcohols having more than seven atoms in an unbroken chain
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/361—Carboxylic acids having more than seven carbon atoms in an unbroken chain; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/37—Esters of carboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/37—Esters of carboxylic acids
- A61K8/375—Esters of carboxylic acids the alcohol moiety containing more than one hydroxy group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/39—Derivatives containing from 2 to 10 oxyalkylene groups
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/44—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof
- A61K8/442—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof substituted by amido group(s)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/8105—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- A61K8/8111—Homopolymers or copolymers of aliphatic olefines, e.g. polyethylene, polyisobutene; Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/86—Polyethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/89—Polysiloxanes
- A61K8/891—Polysiloxanes saturated, e.g. dimethicone, phenyl trimethicone, C24-C28 methicone or stearyl dimethicone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/92—Oils, fats or waxes; Derivatives thereof, e.g. hydrogenation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/14—Esters of carboxylic acids, e.g. fatty acid monoglycerides, medium-chain triglycerides, parabens or PEG fatty acid esters
Definitions
- the present invention relates to a method for producing an O / W emulsified composition, and particularly to a production method excellent in simplicity and economy.
- O / W emulsification using ⁇ -gel formed by higher fatty alcohol and polyoxyethylene nonionic surfactant for the purpose of maintaining the emulsification stability of skin preparations for cosmetics, quasi-drugs, and pharmaceuticals.
- Compositions are used, and are particularly known for use as cosmetics.
- an essential component is added to a water phase in which a humectant and a hydrophilic polyoxyethylene nonionic surfactant are dissolved in water and heated to around 70 ° C.
- a method has been used in which an oil phase in which an oil and a higher alcohol are made uniform at around 70 ° C. is added while stirring with a homogenizer, and the emulsified product is cooled to around 35 ° C. with a cooler such as an onlator (for example, Non-patent document 1).
- the method of cooling after emulsification at around 70 ° C. which has been conventionally performed, is not only wasteful because it requires energy by heating and the use of a heat exchanger, but also for washing after using a cooler such as an on-lator. A large amount of water will be used, which will increase the environmental impact.
- Patent Document 1 includes (A) a hydrophilic nonionic surfactant, (B) a linear higher alcohol having 16 or more carbon atoms, (C) an oil component, and (D) an aqueous solvent that dissolves in water.
- an O / W fine emulsion preparation step of adding water or an aqueous formulation while mixing and stirring to invert the phase into an O / W fine emulsion, and an O / W with an emulsified particle size of 50 to 500 nm A method for producing a fine emulsion external preparation is described.
- the method of Patent Document 1 it is possible to easily produce an O / W fine emulsion that is difficult to prepare by a conventional method using a cooler such as an onlator without using an onlator.
- the O / W fine emulsion obtained by the method of Patent Document 1 is used more refreshingly than the O / W emulsion of the same prescription produced by a conventional method using a cooler such as an onlator. It has sex.
- Patent Document 1 since the method of Patent Document 1 is a method in which a W / O emulsion is once prepared with a specific composition and is converted into a fine O / W emulsion by phase inversion, the composition may be limited. Patent Document 1 does not describe a method for producing an O / W emulsion without going through phase inversion from the W / O emulsion.
- the present invention has been made in view of the circumstances of the background art described above, and a problem to be solved is to provide a method for producing an O / W emulsified composition excellent in simplicity and economy.
- the present inventors have conducted intensive research, and as a result, a straight chain having 16 or more carbon atoms capable of forming an ⁇ gel in water together with a specific nonionic surfactant and the nonionic surfactant.
- O / W emulsified composition having a feeling of use equivalent to that of a conventional method using an onlator or the like by using a part of the aqueous phase for cooling if the amount of the aqueous higher alcohol is controlled and the amount of the aqueous solvent is controlled.
- the inventors have found that it can be manufactured, and have completed the present invention.
- the method for producing an O / W emulsion composition includes: (A) a mono-branched fatty acid POE (0-60) glycerin ester; (B) a linear higher alcohol having 16 or more carbon atoms capable of forming an ⁇ gel in water together with (A), (C) oil and An oil phase containing, (D) A part of the aqueous phase containing water (first aqueous phase) is emulsified at a temperature of 70 ° C. or higher to prepare an emulsified part that is an O / W emulsion, Cooling the emulsified part by mixing with the remaining main aqueous phase (second aqueous phase) at 10-35 ° C. with stirring; The aqueous solvent in the emulsified part is 15% by mass or less.
- the present invention also provides a method for producing an O / W emulsion composition, characterized in that, in the above method, the aqueous solvent is a divalent to trivalent polyol. Further, the present invention provides the method for producing an O / W emulsion composition according to any one of the above, wherein the main aqueous phase is 50 to 75% by mass in the total amount of the O / W emulsion composition. provide.
- the present invention provides the method according to any one of the above, wherein the total amount of (A) mono-branched fatty acid POE (0-60) glycerin ester and (B) a linear higher alcohol having 16 or more carbon atoms is (C )
- a method for producing an O / W emulsified composition characterized in that it is 0.5 to 10 parts by mass with respect to 10 parts by mass of oil.
- the method for producing an O / W emulsified composition according to the present invention it can be easily produced with low energy without using a cooler such as an onlator, and thus it is excellent in economic efficiency. Furthermore, the O / W emulsion composition produced by the production method according to the present invention is equivalent in use to the O / W emulsion composition of the same formulation produced using a cooler such as an onlator. It has a feeling and is excellent in storage stability.
- the method for producing an O / W emulsified composition comprises (A) a monobranched fatty acid POE (0-60) glycerin ester and (B) a carbon number of 16 that can form an ⁇ gel in water together with (A).
- An oil phase containing the above linear higher alcohol, (C) an oil component, and (D) a part of the water phase containing water are emulsified at a temperature of 70 ° C. or higher to provide a high concentration O / W emulsification.
- the emulsified part is prepared and cooled by mixing the emulsified part with the remaining main aqueous phase at 10 to 35 ° C.
- the amount is 15% by mass or less, and an ⁇ -gel is formed at the emulsified particle interface of the obtained O / W emulsion composition.
- the ⁇ -gel refers to an aggregate consisting of a lamellar bimolecular film formed by a surfactant with a linear higher alcohol having 16 or more carbon atoms or an unneutralized fatty acid in the presence of water. .
- (A) Mono-branched fatty acid POE (0-60) glycerin ester used in the present invention is a nonionic surfactant capable of forming an ⁇ gel with (B) a higher alcohol in the aqueous phase.
- (A) a mono-branched fatty acid POE (0-60) glycerin ester is not used and another nonionic surfactant is used, even if an ⁇ gel is formed, preparation of an emulsified part In this case, sufficient emulsification cannot be performed, making it difficult to prepare the emulsified part itself, and the desired O / W emulsified composition may not be obtained.
- the (A) monobranched fatty acid POE (0-60) glycerin ester (also referred to as “POE (0-60) glyceryl monobranched fatty acid ester”) used in the present invention has an average addition mole number of oxyethylene groups of 0 to 60.
- the branched fatty acid monoester of polyoxyethylene glyceryl ether is a saturated branched fatty acid having 16 to 24 carbon atoms, preferably 16 to 20 carbon atoms.
- the branch position is not particularly limited.
- POE means a polyoxyethylene group, and the average added mole number thereof is 0 to 60, preferably 5 to 60.
- one or more (A) mono-branched fatty acid POE (0-60) glycerol esters can be used.
- suitable (A) mono-branched fatty acid POE (0-60) glycerin esters include, for example, those of the following general formula (1).
- R is a residue obtained by removing an OH group from a saturated branched fatty acid having 16 to 24 carbon atoms, preferably 16 to 20 carbon atoms.
- Mono-branched fatty acid POE (0-60) glycerin ester can be easily synthesized by a known method, but it is convenient to use a commercially available nonionic surfactant, for example, EAMALEX GWIS-100 series And Nippon Emulsion Co., Ltd. Further, the mono-branched fatty acid POE (0-60) glycerin ester is preferably hydrophilic, and particularly preferably HLB 6 or higher. In the present invention, a linear fatty acid type nonionic surfactant such as monostearic acid POE glycerin ester can be used in combination with mono-branched fatty acid POE (0-60) glycerin ester.
- the straight chain higher alcohol having 16 or more carbon atoms used in the present invention can form an ⁇ gel in water together with the (A) mono-branched fatty acid POE (0-60) glycerin ester, It is not particularly limited as long as it can be used in the fields of pharmaceuticals, quasi drugs and the like, and examples thereof include cetyl alcohol, stearyl alcohol, behenyl alcohol and the like. A linear saturated higher alcohol having 16 to 24 carbon atoms is preferred. In the present invention, (B) one or more linear higher alcohols having 16 or more carbon atoms can be used. In addition, it is preferable to add batyl alcohol, monoglyceride, etc. on formation of (alpha) gel.
- the mass ratio of (A) monobranched fatty acid POE (0-60) glycerin ester used in the present invention to (B) a straight chain higher alcohol having 16 or more carbon atoms is set within a range not impairing the effects of the present invention. There is no particular limitation. Usually, the mass ratio of (A) :( B) is 1: 2 to 10: 1 in the O / W emulsified part.
- the concentration of (A) mono-branched fatty acid POE (0-60) glycerin ester and (B) straight chain higher alcohol having 16 or more carbon atoms is not particularly limited, but in the O / W emulsion composition,
- the total amount of A) mono-branched fatty acid POE (0-60) glycerin ester and (B) straight chain higher alcohol having 16 or more carbon atoms is (C) 0.5 to 10 parts by mass with respect to 10 parts by mass of oil. It is preferable to do.
- the amount is less than 0.5 parts by mass, since the total amount of (A) and (B) is small, a highly stable O / W emulsion composition may not be obtained. Since there is too much total amount of (A) and (B), there exists a tendency which is unpreferable at the point of a usability
- the oil component (C) used in the present invention is not particularly limited, and any oil component can be used as long as it is usually used in cosmetics, pharmaceuticals and the like.
- the solid oil include oils that are solid at room temperature, which are generally used for cosmetics and external preparations for skin.
- liquid oil examples include oils that are liquid at room temperature, which are generally used for cosmetics and skin external preparations.
- avocado oil evening primrose oil, camellia oil, turtle oil, macadamia nut oil, sunflower oil, almond oil, corn oil, mink oil, olive oil, rapeseed oil, egg yolk oil, sesame oil, persic oil, Wheat germ oil, sasanqua oil, castor oil, flaxseed oil, safflower oil, cottonseed oil, eno oil, soybean oil, peanut oil, teaseed oil, kaya oil, rice bran oil, cinnabari oil, Japanese kiri oil, jojoba oil, germ oil Liquid oils such as cetyl octoate, cetyl 2-ethylhexanoate, hexyl decyl dimethyl octanoate, ethyl laurate, hexyl laurate, isopropyl myri
- (C) one or more higher fatty acids are blended in the oil.
- the emulsified particles are further refined, and a more stable O / W emulsion composition is obtained.
- Higher fatty acids are preferably those having 16 to 24 carbon atoms, such as unsaturated fatty acids such as oleic acid, linoleic acid, and linolenic acid, isostearic acid, isopalmitic acid, isomyristic acid, behenic acid, stearic acid, palmitic acid. An acid, myristic acid, etc. are mentioned.
- the higher fatty acid is preferably 0.1 to 3% by mass, more preferably 0.2 to 1.5% by mass in the O / W emulsion composition.
- the aqueous phase containing water (D) used in the present invention contains water as a main medium, and usually contains hydrophilic components used in cosmetics, pharmaceuticals, etc. within a range that does not impair the effects of the present invention. It doesn't matter.
- the total amount of water in the O / W emulsion composition of the present invention is not particularly limited, but is generally 40 to 95% by weight in the total amount of the O / W emulsion composition. preferable.
- the aqueous phase of the O / W emulsion composition of the present invention can contain an aqueous solvent.
- concentration of the aqueous solvent is too high in the emulsified part of the present invention, the preparation of the emulsified part itself cannot be performed, or even if the emulsified part can be prepared, a conventional O / W emulsified composition obtained using an onlator or the like O / W emulsion composition having a feeling of use equivalent to that of the product may not be obtained.
- the storage stability may be insufficient.
- the concentration of the aqueous solvent in the emulsified part is preferably 15% by mass or less, more preferably 3% by mass or less, and particularly preferably 1% by mass or less. It is. In cosmetics and external preparations for skin, a large amount of an aqueous solvent is often blended, but even if the amount of the aqueous solvent in the emulsified part of the present invention is limited as described above, it is for mixing with the emulsified part.
- the amount of the aqueous solvent in the main aqueous phase (second aqueous phase) is not particularly limited. Therefore, the amount of the aqueous solvent in the finally obtained O / W emulsion composition can be freely designed.
- aqueous solvent whose composition is limited in the aqueous phase for preparing emulsified parts
- examples of the aqueous solvent whose composition is limited in the aqueous phase for preparing emulsified parts include, for example, an aqueous solvent that is liquid at room temperature and is compatible with water having 0 to 3 hydroxyl groups in the molecule.
- glycol (divalent polyol) has a particularly significant effect on the preparation of emulsified parts.
- the main aqueous phase (second aqueous phase) contains (D) water, and can optionally contain the above-mentioned aqueous solvent and other aqueous solvents.
- the component used for a pharmaceutical etc. can also be included in the range by which the effect of this invention is not impaired.
- the main aqueous phase is preferably 50 to 75% by mass, more preferably 55 to 70% by mass, based on the total amount of the O / W emulsion composition. If the main water phase is less than 50% by mass, a sufficient cooling effect cannot be obtained, and extra cooling energy is required accordingly, resulting in poor economic efficiency. Moreover, when it exceeds 75 mass%, since the water phase of the O / W emulsification part prepared at 70 degreeC or more decreases, it exists in the tendency for preparation of a stable O / W emulsification composition to become difficult.
- the temperature of the main aqueous phase added later is preferably 10 to 35 ° C., particularly preferably 15 to 30 ° C. If it is less than 10 ° C., excessive energy for cooling the main water phase is required, which tends to be inferior in economic efficiency. Moreover, when it exceeds 35 degreeC, since sufficient cooling effect is not acquired and extra cooling energy is needed by that much, it is inferior to economical efficiency.
- exothermic peak temperature was measured by using DSC (Q-2000, TA Instruments, USA) and decreasing the temperature at 1 ° C./min.
- the peak temperature of the exothermic peak measured by DSC is considered to correspond to the melting point of the emulsified part in the production method of the present invention.
- the emulsified part of 70 ° C. or higher and the low temperature main water phase of 10 to 35 ° C. are mixed with stirring, but the temperature of the mixture immediately after the mixing of the low temperature main water phase is high, for example, 40 ° C. or higher.
- variation in viscosity of the obtained O / W emulsion composition may be increased.
- the temperature of the mixture immediately after the mixing of the main aqueous phase and the emulsified part is preferably 35 ° C. ⁇ 2 ° C. or lower. This can be easily controlled by the amount and temperature of the main aqueous phase.
- the O / W emulsified composition produced by the production method of the present invention has a monobranched fatty acid POE (0-60) glycerin ester and a higher alcohol at the interface of emulsified particles composed of an oil phase in the presence of water. In this state, there are aggregates formed of lamellar bimolecular films to be formed, so-called ⁇ gels.
- an emulsified part emulsified at a high concentration using a part of the aqueous phase is prepared at 70 ° C. or higher (preferably 70 to 80 ° C., more preferably 70 to 75 ° C.), The emulsified part and the remaining aqueous phase (main aqueous phase) at 10 to 35 ° C. are gradually mixed with stirring.
- the above-described high-temperature emulsified part is diluted and cooled at once, and an O / W emulsified composition in which an ⁇ -gel is present at the emulsion particle interface is prepared.
- an O / W emulsified composition in which an ⁇ -gel is present at the emulsion particle interface is prepared.
- An apparatus used for emulsification, stirring and the like may be appropriately selected from those normally used, and the apparatus used so far can be used as it is, and no special equipment is required.
- the mixing order of the emulsified part and the main aqueous phase is not particularly limited.
- the O / W emulsion composition obtained by the above production method has a sufficiently small particle diameter of 1 to 7.5 ⁇ m and can exist stably over a wide temperature range for a long time.
- an oil, a higher alcohol, and a hydrophilic nonionic surfactant are added to an aqueous phase in which water, a humectant, and a thickener are dissolved in advance and heated to around 70 ° C.
- a method has been used in which an oil phase obtained by homogenizing an agent at around 70 ° C. and emulsified with stirring by a homogenizer is rapidly cooled to around 35 ° C. using an onlator.
- a lot of energy is wasted due to heating and the use of a heat exchanger, and the consumption of water used in the cooler is increased, resulting in a high environmental load.
- the production method of the present invention it is not necessary to heat a large amount of an aqueous phase without using a cooler such as an onlator during emulsification, so that O / W emulsification can be easily performed with low energy.
- a composition can be produced.
- it is substantially emulsified only by the nonionic surfactant with comparatively small irritation with respect to a human body, it is excellent in safety.
- the method for producing an O / W emulsified composition according to the present invention can be achieved by simply adding an aqueous phase at 10 to 35 ° C. to an emulsified part previously produced at 70 to 80 ° C. Since a good O / W emulsified composition that does not change can be obtained, the conventionally used production process can be greatly simplified.
- the O / W emulsified composition according to the present invention can be suitably applied to, for example, an external composition that can be applied to the body such as skin and hair in cosmetics, medicines, and quasi drugs.
- an external composition that can be applied to the body
- it can be used for skin cosmetics, hair cleansing materials, skin cleansing materials, hair styling products and the like.
- components generally used in cosmetics, pharmaceuticals, and the like can be blended in the O / W emulsion composition according to the present invention within a range that does not impair the effects of the present invention. Examples of such components include the following.
- Thickeners such as methylcellulose, ethylcellulose, gum arabic, and polyvinyl alcohol.
- Organic solvents such as ethanol.
- Antioxidants such as butylhydroxytoluene, tocopherol and phytic acid.
- Antibacterial preservatives such as benzoic acid, salicylic acid, sorbic acid, paraoxybenzoic acid esters (such as ethylparaben and butylparaben), and hexachlorophene.
- Amino acids and salts thereof such as glycine, alanine, valine, leucine, serine, threonine, phenylalanine, tyrosine, aspartic acid, asparagine, glutamine, taurine, arginine, histidine.
- Organic acids such as acyl sarcosine acid (eg, lauroyl sarcosine sodium), glutathione, citric acid, malic acid, tartaric acid, lactic acid, and salts thereof.
- Vitamin A and its derivatives Vitamin A and its derivatives, vitamin B6 hydrochloride, vitamin B6 tripalmitate, vitamin B6 dioctanoate, vitamin B2 and its derivatives, vitamin B such as vitamin B12, vitamin B15 and its derivatives, ascorbic acid, ascorbic acid phosphate ( Salt), vitamin C such as ascorbic acid dipalmitate, ⁇ -tocopherol, ⁇ -tocopherol, ⁇ -tocopherol, vitamin E acetate, vitamin E nicotinate and other vitamin E, vitamin D, vitamin H, pantothenic acid, pantethine, etc. Vitamins.
- Nicotinamide benzyl nicotinate, ⁇ -oryzanol, allantoin, glycyrrhizic acid (salt), glycyrrhetinic acid and its derivatives, hinokitiol, mucidin, bisabolol, eucalyptol, thymol, inositol, saponins (psychosaponin, carrot saponin, loofah)
- Various drugs such as saponin, muclodisaponin, etc.), pantothenyl ethyl ether, ethinyl estradiol, tranexamic acid, cephalanthin, placenta extract.
- Cationic surfactants such as stearyltrimethylammonium chloride, benzalkonium chloride, and laurylamine oxide.
- Metal sequestering agents such as disodium edetate, trisodium edetate, sodium citrate, sodium polyphosphate, sodium metaphosphate and gluconic acid.
- Neutralizing agents such as potassium hydroxide, sodium hydroxide and triethanolamine.
- fragrances, scrub agents, powders, coloring materials, whitening agents, UV protection agents such as UV absorbers and UV scattering agents, and the like can be appropriately blended as long as stability is not impaired.
- Manufacturing method B The oil phase and the first aqueous phase were emulsified at 70 ° C. using a homogenizer to obtain an O / W emulsion (emulsified part). This emulsified part was mixed with the main aqueous phase (second aqueous phase) at 25 ° C. while stirring with a propeller stirrer (250 rpm) to obtain an O / W emulsion composition at 35 ° C. ⁇ 2 ° C.
- Production Example 1-a is an O / W emulsion composition (conventional product) by a conventional method (Production Method A) using an onlator.
- Production Examples 1-1 to 1-5 have the same composition as Production Example 1-a (conventional product), but all are cooled by mixing the low-temperature main water phase without using an onlator (production method) It is an O / W emulsion composition manufactured by B), and the amount of the aqueous solvent (dipropylene glycol) in the emulsified part is different.
- the emulsified parts of Production Examples 1-1 to 1-5 formed an ⁇ gel.
- the aqueous solvent in the emulsified part is 15% by mass or less, further 3% by mass or less, particularly 1% by mass or less. It was considered suitable. Also, from the viewpoint of storage stability, it was considered suitable to adjust the aqueous solvent concentration in the emulsified part as described above.
- Table 2 shows the results when glycerin is used as the aqueous solvent.
- the O / W emulsified compositions obtained in Production Examples 2-1 and 2-2 are all produced by a method (Production Method B) in which cooling is performed by mixing a low-temperature main aqueous phase without using an onlator. Yes, the emulsified part formed an alpha gel.
- the equivalence with the conventional product was evaluated based on the O / W emulsion composition (conventional product) obtained by the method (production method A) using the onlator with the same composition.
- Production Example 2-1 an O / W emulsion composition having the same usability as conventional products and good storage stability was obtained.
- the aqueous solvent in the emulsified part was used. If the concentration was too high, the emulsified part could not be prepared.
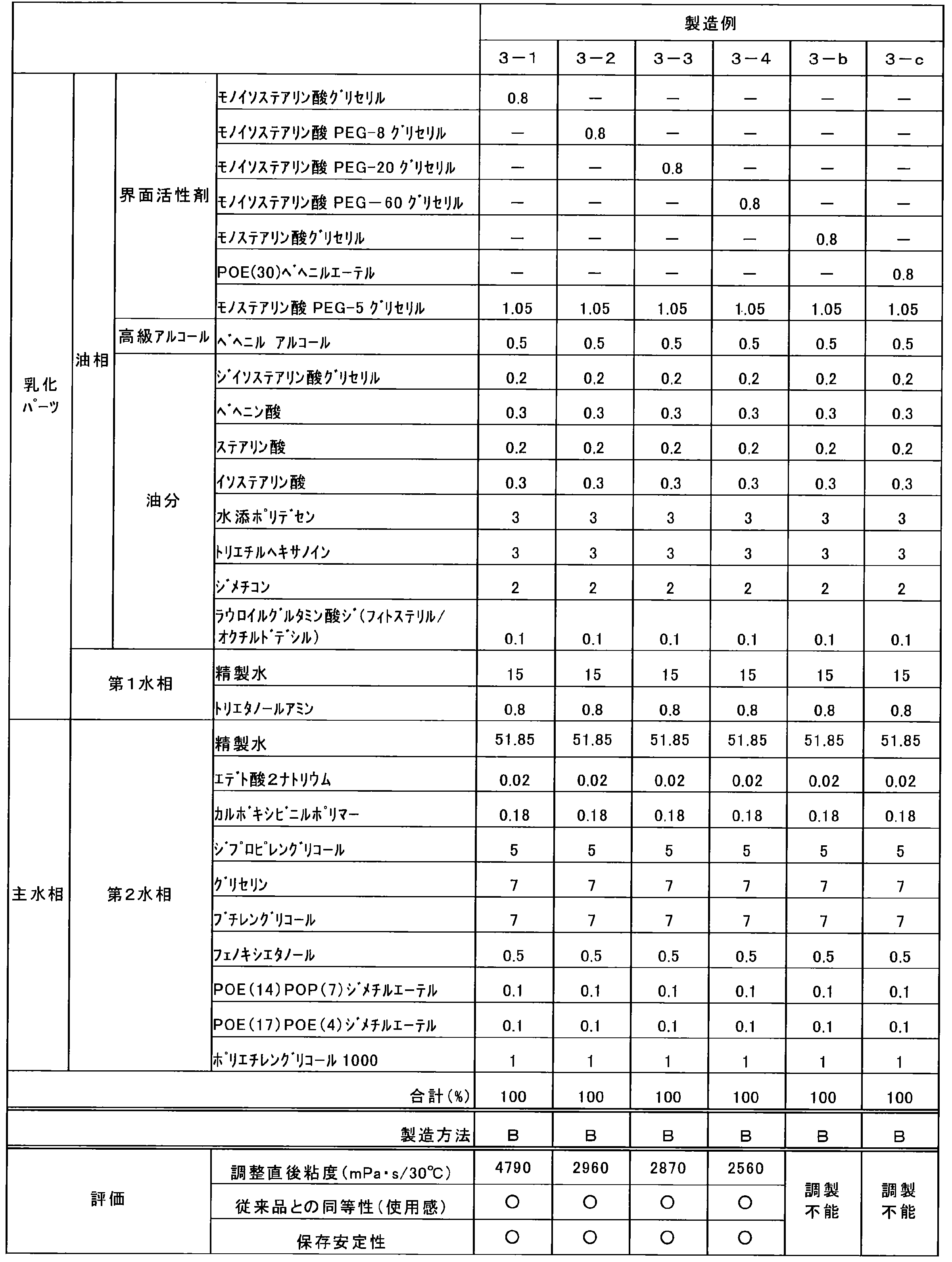
- Table 3 shows the results of investigations with different types of nonionic surfactants.
- an O / W emulsion composition was produced by a method of cooling by mixing with a low-temperature main aqueous phase (Production Method B). All the emulsified parts in Table 3 formed ⁇ -gel.
- the evaluation of the equivalence with the conventional product was evaluated based on the O / W emulsion composition (conventional product) produced according to the production method A with the same composition.
- Table 3 when monobranched fatty acid POE (0-60) glycerin ester is used, an O / W emulsified composition having a feeling of use equivalent to that of a conventional product can be obtained. (Production Examples 3-1 to 3-4) were also excellent.
- O / W emulsion was obtained by a method according to Production Method B.
- the viscosity of the obtained emulsion was 4,370 mPa ⁇ s / 30 ° C.
- This emulsion had the same feeling of use as a conventional product having the same composition (O / W emulsion obtained by production method A cooled with an onlator or the like) and was excellent in storage stability.
- Example 2 Emulsion ⁇ Second Aqueous Phase (Main Aqueous Phase)> Purified water to 100 mass% Potassium 4-methoxysalicylate 1.0 Edetic acid disodium 0.05 Glycerin 7.0 Dipropylene glycol 7.0 Butylene glycol 5.0 Succinoglycan 0.04 Carboxyvinyl polymer 0.15 Ethanol 1.0 Phenoxyethanol 0.5 Dipotassium glycyrrhizinate 0.05 ⁇ Oil phase> Triethylhexanoin 4.0 Hydrogenated polydecene 6.0 PEG-60 glyceryl monoisostearate 1.2 PEG-5 glyceryl monostearate 0.8 Behenic acid 0.45 Stearic acid 0.6 Isostearic acid 0.45 Behenyl alcohol 0.95 Batyl alcohol 0.25 Vaseline 2.0 Glyceryl diisostearate 2.0 Lauroyl glutamate di (phytosteryl / octyldodecyl) 0.1
- the viscosity of the obtained emulsion was 3,690 mPa ⁇ s / 30 ° C.
- This emulsion had the same feeling of use as a conventional product having the same composition (O / W emulsion obtained by production method A cooled with an onlator or the like) and was excellent in storage stability.
- Example 3 Emulsion ⁇ Second Aqueous Phase (Main Aqueous Phase)> Purified water to 100 mass% Sodium metaphosphate 0.01 Xylitol 3.0 Glycerin 4.0 Dipropylene glycol 5.0 Xanthan gum 0.1 Carboxyvinyl polymer 0.18 Ethanol 4.0 Phenoxyethanol 0.5 ⁇ Oil phase> Cetyl ethylhexanoate 3.0 Hydrogenated polydecene 3.5 Dimethicone 2.0 PEG-60 glyceryl monoisostearate 1.3 PEG-8 glyceryl monoisostearate 0.7 Behenic acid 0.3 Stearic acid 0.4 Isostearic acid 0.3 Behenyl alcohol 0.3 Batyl alcohol 0.15 ⁇ First aqueous phase (for preparing emulsified parts)> Potassium hydroxide 0.14 Purified water 15.0
- O / W emulsion was obtained by a method according to Production Method B.
- the viscosity of the obtained emulsion was 3,260 mPa ⁇ s / 30 ° C.
- This emulsion had the same feeling of use as a conventional product having the same composition (O / W emulsion obtained by production method A cooled with an onlator or the like) and was excellent in storage stability.
Abstract
Description
(A)モノ分岐脂肪酸POE(0-60)グリセリンエステルと、
(B)前記(A)とともに水中でαゲルを形成し得る炭素数16以上の直鎖状高級アルコールと、
(C)油分と、
を含む油相と、
(D)水
を含む水相の一部(第1水相)とを、70℃以上の温度で乳化してO/W乳化物である乳化パーツを調製し、
この乳化パーツを10~35℃の残りの主水相(第2水相)と攪拌しながら混合することにより冷却することを含み、
前記乳化パーツ中における水性溶媒が15質量%以下であることを特徴とする。
また、本発明は、前記何れかに記載の方法において、前記主水相がO/W乳化組成物全量中50~75質量%であることを特徴とするO/W乳化組成物の製造方法を提供する。
好適な(A)モノ分岐脂肪酸POE(0-60)グリセリンエステルの一例としては、例えば、下記一般式(1)のものが挙げられる。
また、モノ分岐鎖脂肪酸POE(0-60)グリセリンエステルとしては親水性であるものが好ましく、特にHLB6以上のものが好ましい。なお、本発明においては、モノ分岐脂肪酸POE(0-60)グリセリンエステルとともに、モノステアリン酸POEグリセリンエステルなどの、直鎖脂肪酸型非イオン性界面活性剤を組み合わせて使用することができる。
なお、αゲルの形成の上では、バチルアルコール、モノグリセライド等を添加することが好ましい。
本発明で用いる(A)モノ分岐脂肪酸POE(0-60)グリセリンエステルと(B)炭素数16以上の直鎖状高級アルコールとの質量比は、本発明の効果が損なわれない範囲で設定すればよく、特に制限されるものではない。通常は(A):(B)の質量比はO/W乳化パーツ中1:2~10:1である。
固形油分としては、一般に化粧料や皮膚外用剤に用いられる室温において固体の油分が挙げられる。具体的に示すとすれば、例えば、カカオ脂、ヤシ油、馬脂、硬化ヤシ油、パーム油、牛脂、羊脂、硬化牛脂、パーム核油、豚脂、牛骨脂、モクロウ核油、硬化脂、牛脚脂、モクロウ、硬化ヒマシ油等の固体油脂;ミツロウ、キャンデリラロウ、綿ロウ、カルナウバロウ、ベイベリーロウ、イボタロウ、鯨ロウ、モンタンロウ、ヌカロウ、ラノリン、カポックロウ、酢酸ラノリン、サトウキビロウ、ラノリン脂肪酸イソプロピル、ラウリン酸ヘキシル、還元ラノリン、ジョジョバロウ、硬質ラノリン、セラックロウ、POEラノリンアルコールエーテル、POEラノリンアルコールアセテート、POEコレステロールエーテル、POE水素添加ラノリンアルコールエーテル等のロウ類;ポリエチレンワックス、パラフィンワックス、セレシン、ワセリン、マイクロクリスタリンワックス、ルナセラ、オゾケライト等の炭化水素系ワックス;モノステアリルグリセリンエーテル(バチルアルコール)等の脂肪酸グリセリルエーテル;アセトグリセライド、トリ-2-ヘプチルウンデカン酸グリセライド等の脂肪酸グリセリド等が挙げられる。これらの固形油分は、それぞれ単独ないしは2種以上を組み合わせて配合することができる。
なお、本発明のO/W乳化組成物における水の総配合量は、特に限定されるものではないが、一般的には、O/W乳化組成物全量中40~95重量%であることが好ましい。
従って、乳化パーツ中において水性溶媒を含まないことが好ましく、含む場合には乳化パーツ中における水性溶媒の濃度は15質量%以下、さらには3質量%以下、特に1質量%以下とすることが好適である。
なお、化粧料や皮膚外用剤では、水性溶媒が多量に配合されることが多いが、本発明の乳化パーツ中の水性溶媒量は上記のように制限されても、乳化パーツとの混合用の主水相(第2水相)中の水性溶媒量は特に制限されない。よって、最終的に得られるO/W乳化組成物中の水性溶媒量は自由に設計することができる。
これらのうち、グリコール(2価ポリオール)は乳化パーツ調製に及ぼす影響が特に著しい。
該主水相は、O/W乳化組成物全量中50~75質量%が好ましく、55~70質量%がより好ましい。主水相が、50質量%未満であると十分な冷却効果が得られず、その分余計な冷却エネルギーを必要とするため、経済性に劣る。また、75質量%を超えると70℃以上で調製するO/W乳化パーツの水相が少なくなるために、安定なO/W乳化組成物の調製が困難になる傾向にある。
なお、DSC測定において、αゲル形成による発熱ピークの他にも発熱ピークが検出されることがある。これは、乳化パーツ中に含まれる成分の単なる凝固による発熱ピークであり、αゲル形成による発熱ピークはこれら凝固による発熱ピークとは異なる。
なお、本発明において発熱ピーク温度の測定は、DSC(Q-2000,TA Instruments,USA)を用い、1℃/分で降温させて測定した。
概念
(A)モノ分岐脂肪酸POE(0-60)グリセリンエステルと(B)前記(A)とともに水中でαゲルを形成し得る炭素数16以上の直鎖状高級アルコールと(C)油分とを含む油相と、(D)水を含む水相とを70℃以上の温度で乳化し、これを冷却する工程によりO/W乳化組成物が得られる。
よって、上記問題を解消するために、水相の一部を用いて高濃度に乳化された乳化パーツを70℃以上(好ましくは70~80℃、さらに好ましくは70~75℃)で調製し、上記乳化パーツと10~35℃の残りの水相(主水相)とを攪拌しながら徐々に混合する。この低温主水相の混合により、上述の高温乳化パーツが希釈されるとともに一気に冷却され、αゲルが乳化粒子界面に存在するO/W乳化組成物が調製される。ただし、このような方法において乳化パーツ中における水性溶媒濃度が高すぎると使用性や保存安定性に優れるO/W乳化組成物が得られない。よって、本発明においては、水性溶媒を乳化パーツ中15質量%以下とすることが必要である。乳化、攪拌などに用いる装置は通常使用されているものから適宜選択すればよく、それまで使用していた装置をそのまま利用することができ、特別の設備は不要である。乳化パーツと主水相との混合順序も特に制限されない。
また、本発明にかかるO/W乳化組成物には、上記必須成分の他に、通常、化粧品、医薬品等に用いられる成分を本発明の効果を損なわない範囲で配合することができる。かかる成分としては、例えば次のようなものが挙げられる。
また、その他、香料、スクラブ剤、粉末、色材、美白剤、紫外線吸収剤や紫外線散乱剤などの紫外線防御剤なども、安定性などを損なわない範囲で適宜配合することができる。
まず、本発明で用いた試験法・評価法について説明する。
B型粘度計(ローター番号3番、ローター回転数12rpm)を用い、30℃における粘度を測定した。
調製したO/W乳化組成物を、0℃、25℃、50℃の各温度で1か月間保存した後、製造直後の粘度との差が最大となる温度条件において、下記の評価基準により評価した。
○:粘度変化が10%未満
△:粘度変化が10%以上20%未満
×:粘度変化が20%以上
なお、△以上の評価を合格とした。
10名のパネルにより試験を行い、使用感(のび、なじみ、べたつき、みずみずしさ、こく)の総合評価として従来品(同一組成でオンレータを用いて製造したO/W乳化組成物)と同等であると回答した人数が、
○:8名以上
△:5名以上7名以下
×:4名以下
とし、△以上の評価を合格とした。
油相をホモゲナイザーで攪拌しながら第1水相及び第2水相を加え、70℃にて乳化し調製したO/Wエマルションをオンレーターに通過させて35℃まで冷却し、その後室温まで放冷してO/W乳化組成物を得た。
油相及び第1水相をホモゲナイザーを用いて70℃で乳化してO/Wエマルション(乳化パーツ)を得た。この乳化パーツを25℃の主水相(第2水相)にプロペラ攪拌機(250rpm)で攪拌しながら混合し、35℃±2℃のO/W乳化組成物を得た。
製造例1-aはオンレータを用いた従来法(製造方法A)によるO/W乳化組成物(従来品)である。
一方、製造例1-1~1-5は、製造例1-a(従来品)と同一組成であるものの、何れもオンレータを使用せずに低温主水相の混合により冷却する方法(製造方法B)により製造されたO/W乳化組成物であり、乳化パーツ中の水性溶媒(ジプロピレングリコール)の量がそれぞれ異なっている。製造例1-1~1-5の乳化パーツは、αゲルを形成した。
このようなことから、従来品と同等の使用感のO/W乳化組成物を得るためには、乳化パーツ中の水性溶媒を15質量%以下、さらには3質量%以下、特に1質量%以下とすることが好適であると考えられた。また、保存安定性の点からも、上記のように乳化パーツ中の水性溶媒濃度を調整することが好適であると考えられた。
製造例2-1では、従来品と同等の使用感を有し、保存安定性も良好なO/W乳化組成物が得られたが、製造例2-2のように乳化パーツ中の水性溶媒濃度が高すぎると、乳化パーツの調製ができなかった。
表3のように、モノ分岐脂肪酸POE(0-60)グリセリンエステルを用いた場合には、従来品と同等の使用感を有するO/W乳化組成物が得られ、またこれらは保存安定性にも優れていた(製造例3-1~3-4)。
このようなことから、本発明においては、モノ分岐脂肪酸POE(0-60)グリセリンエステルを用いることが重要であることが理解される。
<第2水相(主水相)>
精製水 to 100 質量%
トラネキサム酸 2.0
エデト酸2ナトリウム 0.01
キシリトール 3.0
グリセリン 3.0
ジプロピレングリコール 7.0
キサンタンガム 0.05
カルボキシビニルポリマー 0.1
エタノール 3.0
フェノキシエタノール 0.5
<油相>
エチルヘキサン酸セチル 4.0
流動パラフィン 3.5
モノイソステアリン酸PEG-60グリセリル 1.0
モノステアリン酸PEG-5グリセリル 1.0
ベヘニン酸 0.3
ステアリン酸 0.4
イソステアリン酸 0.3
ベヘニルアルコール 0.3
バチルアルコール 0.15
ワセリン 0.5
ジメチコン 1.0
<第1水相(乳化パーツ調製用)>
水酸化カリウム 0.08
精製水 15.0
製造方法Bに準じた方法により、O/W乳液を得た。得られた乳液の粘度は4,370mPa・s/30℃であった。この乳液は、同じ組成の従来品(オンレータ等で冷却する製造方法Aで得られたO/W乳液)と同等の使用感を有し、また保存安定性にも優れていた。
<第2水相(主水相)>
精製水 to 100 質量%
4-メトキシサリチル酸カリウム 1.0
エデト酸2ナトリウム 0.05
グリセリン 7.0
ジプロピレングリコール 7.0
ブチレングリコール 5.0
サクシノグリカン 0.04
カルボキシビニルポリマー 0.15
エタノール 1.0
フェノキシエタノール 0.5
グリチルリチン酸ジカリウム 0.05
<油相>
トリエチルヘキサノイン 4.0
水添ポリデセン 6.0
モノイソステアリン酸PEG-60グリセリル 1.2
モノステアリン酸PEG-5グリセリル 0.8
ベヘニン酸 0.45
ステアリン酸 0.6
イソステアリン酸 0.45
ベヘニルアルコール 0.95
バチルアルコール 0.25
ワセリン 2.0
ジイソステアリン酸グリセリル 2.0
ラウロイルグルタミン酸
ジ(フィトステリル/オクチルドデシル) 0.1
ジメチコン 2.0
メチルフェニルポリシロキサン 1.5
<第1水相(乳化パーツ調製用)>
トリエタノールアミン 1.5
精製水 15.0
<製造方法>
製造方法Bに準じた方法により、O/W乳液を得た。得られた乳液の粘度は3,690mPa・s/30℃であった。この乳液は、同じ組成の従来品(オンレータ等で冷却する製造方法Aで得られたO/W乳液)と同等の使用感を有し、また保存安定性にも優れていた。
<第2水相(主水相)>
精製水 to 100 質量%
メタリン酸ナトリウム 0.01
キシリトール 3.0
グリセリン 4.0
ジプロピレングリコール 5.0
キサンタンガム 0.1
カルボキシビニルポリマー 0.18
エタノール 4.0
フェノキシエタノール 0.5
<油相>
エチルヘキサン酸セチル 3.0
水添ポリデセン 3.5
ジメチコン 2.0
モノイソステアリン酸PEG-60グリセリル 1.3
モノイソステアリン酸PEG-8グリセリル 0.7
ベヘニン酸 0.3
ステアリン酸 0.4
イソステアリン酸 0.3
ベヘニルアルコール 0.3
バチルアルコール 0.15
<第1水相(乳化パーツ調製用)>
水酸化カリウム 0.14
精製水 15.0
製造方法Bに準じた方法により、O/W乳液を得た。得られた乳液の粘度は3,260mPa・s/30℃であった。この乳液は、同じ組成の従来品(オンレータ等で冷却する製造方法Aで得られたO/W乳液)と同等の使用感を有し、また保存安定性にも優れていた。
Claims (4)
- (A)モノ分岐脂肪酸POE(0-60)グリセリンエステルと、
(B)前記(A)とともに水中でαゲルを形成し得る炭素数16以上の直鎖状高級アルコールと、
(C)油分と、
を含む油相と、
(D)水
を含む水相の一部(第1水相)とを、70℃以上の温度で乳化してO/W乳化物である乳化パーツを調製し、
この乳化パーツを10~35℃の残りの主水相(第2水相)と攪拌しながら混合することにより冷却することを含み、
前記乳化パーツ中における水性溶媒が15質量%以下であることを特徴とするO/W乳化組成物の製造方法。 - 請求項1記載の方法において、水性溶媒が2~3価のポリオールであることを特徴とするO/W乳化組成物の製造方法。
- 請求項1又は2記載の方法において、前記主水相がO/W乳化組成物全量中50~75質量%であることを特徴とするO/W乳化組成物の製造方法。
- 請求項1~4の何れかに記載の方法において、(A)モノ分岐脂肪酸POE(0-60)グリセリンエステルと(B)炭素数16以上の直鎖状高級アルコールの合計量が、(C)油分10質量部に対して0.5~10質量部であることを特徴とするO/W乳化組成物の製造方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020137025384A KR20140012695A (ko) | 2011-03-01 | 2012-02-22 | O/w 유화조성물의 제조방법 |
| US14/001,680 US20130331468A1 (en) | 2011-03-01 | 2012-02-22 | Production Method Of O/W Emulsion Composition |
| EP12752276.1A EP2682094A4 (en) | 2011-03-01 | 2012-02-22 | METHOD OF MANUFACTURING OIL-IN-WATER EMULSION COMPOSITION |
| CN201280011063.6A CN103458855B (zh) | 2011-03-01 | 2012-02-22 | O/w乳化组合物的制备方法 |
| BR112013022072A BR112013022072A2 (pt) | 2011-03-01 | 2012-02-22 | método de produção de composição de emulsão o/a |
| HK14101241.9A HK1188133A1 (zh) | 2011-03-01 | 2014-02-11 | 乳化組合物的製備方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011043593 | 2011-03-01 | ||
| JP2011-043593 | 2011-03-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012117909A1 true WO2012117909A1 (ja) | 2012-09-07 |
Family
ID=46757848
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/054239 WO2012117909A1 (ja) | 2011-03-01 | 2012-02-22 | O/w乳化組成物の製造方法 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20130331468A1 (ja) |
| EP (1) | EP2682094A4 (ja) |
| JP (1) | JP5244989B2 (ja) |
| KR (1) | KR20140012695A (ja) |
| CN (1) | CN103458855B (ja) |
| BR (1) | BR112013022072A2 (ja) |
| HK (1) | HK1188133A1 (ja) |
| TW (1) | TWI532502B (ja) |
| WO (1) | WO2012117909A1 (ja) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015227305A (ja) * | 2014-05-30 | 2015-12-17 | 花王株式会社 | 皮膚化粧料 |
| WO2015190305A1 (ja) * | 2014-06-13 | 2015-12-17 | 株式会社 資生堂 | αゲル中間体組成物、及び該組成物を用いたαゲル含有O/W乳化化粧料の製造方法 |
| CN106604714B (zh) * | 2014-06-13 | 2018-10-12 | 株式会社资生堂 | α凝胶中间体组合物和使用该组合物的含有α凝胶的O/W乳化化妆品的制备方法 |
| JP6548265B2 (ja) * | 2015-03-06 | 2019-07-24 | 国立大学法人信州大学 | エマルション |
| ITUB20154642A1 (it) * | 2015-10-13 | 2017-04-13 | Bakel Srl | Metodo di preparazione di un prodotto in crema |
| WO2017081725A1 (ja) * | 2015-11-09 | 2017-05-18 | 花王株式会社 | 口腔用組成物 |
| HUE047368T2 (hu) * | 2017-07-17 | 2020-04-28 | Dr August Wolff Gmbh & Co Kg Arzneimittel | Olaj-a-vízben emulzió |
| CN109464302B (zh) * | 2018-12-29 | 2021-11-23 | 上海家化联合股份有限公司 | 含甘草酸二钾的组合物 |
| WO2020179947A1 (ko) * | 2019-03-06 | 2020-09-10 | 코스맥스 주식회사 | 입자 형태의 유상을 가지는 화장료 조성물을 제조하는 방법 및 그에 의해 제조된 화장료 조성물 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008044866A (ja) * | 2006-08-11 | 2008-02-28 | Shiseido Co Ltd | クリーム組成物 |
| WO2008117481A1 (ja) * | 2007-03-22 | 2008-10-02 | Noevir Co., Ltd. | 皮膚外用剤及び飲食品 |
| WO2010082602A1 (ja) | 2009-01-14 | 2010-07-22 | 株式会社 資生堂 | O/w微細エマルション外用剤の製造方法 |
| WO2011027811A1 (ja) * | 2009-09-04 | 2011-03-10 | 株式会社 資生堂 | O/w乳化組成物の製造方法 |
| WO2011027812A1 (ja) * | 2009-09-04 | 2011-03-10 | 株式会社 資生堂 | 高粘度o/wクリームの製造方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100678364B1 (ko) * | 1999-07-27 | 2007-02-05 | 가부시키가이샤 시세이도 | 수중유형 유화조성물 |
-
2012
- 2012-02-22 BR BR112013022072A patent/BR112013022072A2/pt not_active IP Right Cessation
- 2012-02-22 EP EP12752276.1A patent/EP2682094A4/en not_active Withdrawn
- 2012-02-22 JP JP2012035831A patent/JP5244989B2/ja active Active
- 2012-02-22 KR KR1020137025384A patent/KR20140012695A/ko not_active Application Discontinuation
- 2012-02-22 CN CN201280011063.6A patent/CN103458855B/zh active Active
- 2012-02-22 US US14/001,680 patent/US20130331468A1/en not_active Abandoned
- 2012-02-22 WO PCT/JP2012/054239 patent/WO2012117909A1/ja active Application Filing
- 2012-02-24 TW TW101106182A patent/TWI532502B/zh not_active IP Right Cessation
-
2014
- 2014-02-11 HK HK14101241.9A patent/HK1188133A1/zh not_active IP Right Cessation
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008044866A (ja) * | 2006-08-11 | 2008-02-28 | Shiseido Co Ltd | クリーム組成物 |
| WO2008117481A1 (ja) * | 2007-03-22 | 2008-10-02 | Noevir Co., Ltd. | 皮膚外用剤及び飲食品 |
| WO2010082602A1 (ja) | 2009-01-14 | 2010-07-22 | 株式会社 資生堂 | O/w微細エマルション外用剤の製造方法 |
| WO2011027811A1 (ja) * | 2009-09-04 | 2011-03-10 | 株式会社 資生堂 | O/w乳化組成物の製造方法 |
| WO2011027812A1 (ja) * | 2009-09-04 | 2011-03-10 | 株式会社 資生堂 | 高粘度o/wクリームの製造方法 |
Non-Patent Citations (2)
| Title |
|---|
| MANABU SENOO: "Chemistry and Application of Surface Activity", 1995, DAINIPPON TOSHO CO., LTD., pages: 159 - 160 |
| See also references of EP2682094A4 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN103458855B (zh) | 2016-03-30 |
| TWI532502B (zh) | 2016-05-11 |
| TW201240675A (en) | 2012-10-16 |
| HK1188133A1 (zh) | 2014-04-25 |
| KR20140012695A (ko) | 2014-02-03 |
| JP2012193168A (ja) | 2012-10-11 |
| EP2682094A4 (en) | 2014-09-10 |
| EP2682094A1 (en) | 2014-01-08 |
| BR112013022072A2 (pt) | 2016-08-09 |
| CN103458855A (zh) | 2013-12-18 |
| US20130331468A1 (en) | 2013-12-12 |
| JP5244989B2 (ja) | 2013-07-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4709320B2 (ja) | O/w乳化組成物の製造方法 | |
| JP5244989B2 (ja) | O/w乳化組成物の製造方法 | |
| JP5047369B2 (ja) | O/w微細エマルション外用剤の製造方法 | |
| JP4729126B2 (ja) | 高粘度o/wクリームの製造方法 | |
| US7763237B2 (en) | Ultraviolet protective preparation and cosmetics containing the same | |
| JP6281744B2 (ja) | 水中油型乳化化粧料 | |
| JP2009203200A (ja) | 水中油型乳化組成物 | |
| WO2012090581A1 (ja) | 水中油型乳化日焼け止め化粧料 | |
| JP6133618B2 (ja) | 油中水型乳化組成物 | |
| JP2010222317A (ja) | 水中油型乳化化粧料 | |
| JPH11228378A (ja) | 乳化化粧料 | |
| JP2021123590A (ja) | 油中水型乳化化粧料 | |
| JP4039542B2 (ja) | O/w/o型複合エマルジョン | |
| JP5824323B2 (ja) | 油中水型乳化組成物 | |
| JPH11286418A (ja) | 水中油型毛髪化粧料 | |
| JP2007269668A (ja) | 水中油型美白化粧料 | |
| JP2012072064A (ja) | 油中水型乳化組成物 | |
| JP2024011463A (ja) | 水中油型乳化化粧料 | |
| JP2007169174A (ja) | O/w型乳化組成物 | |
| JP2023157265A (ja) | 乳化化粧料 | |
| JP2013124240A (ja) | 水中油型乳化皮膚外用剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201280011063.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12752276 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14001680 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012752276 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012752276 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20137025384 Country of ref document: KR Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013022072 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013022072 Country of ref document: BR Kind code of ref document: A2 Effective date: 20130829 |