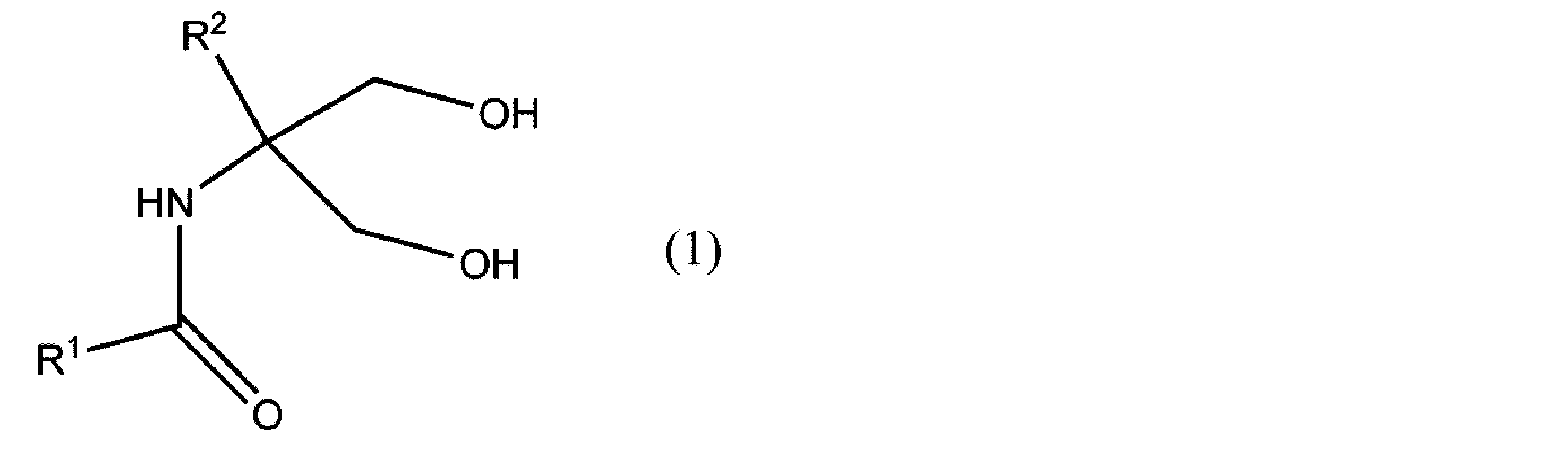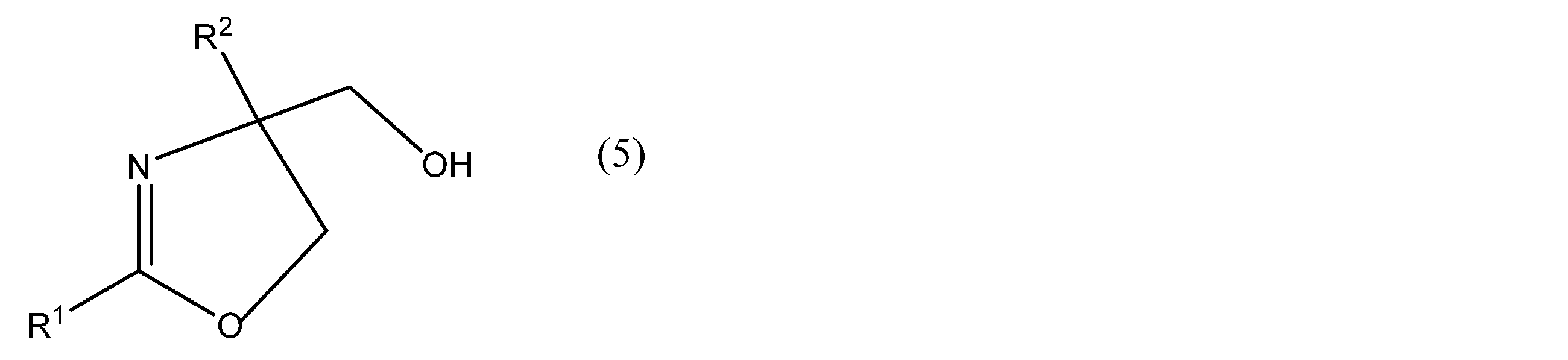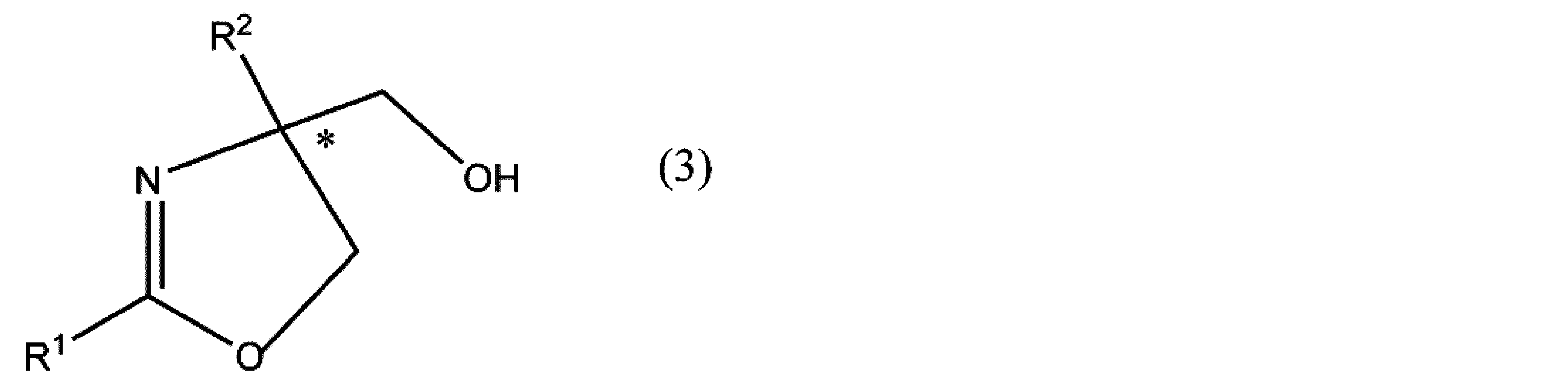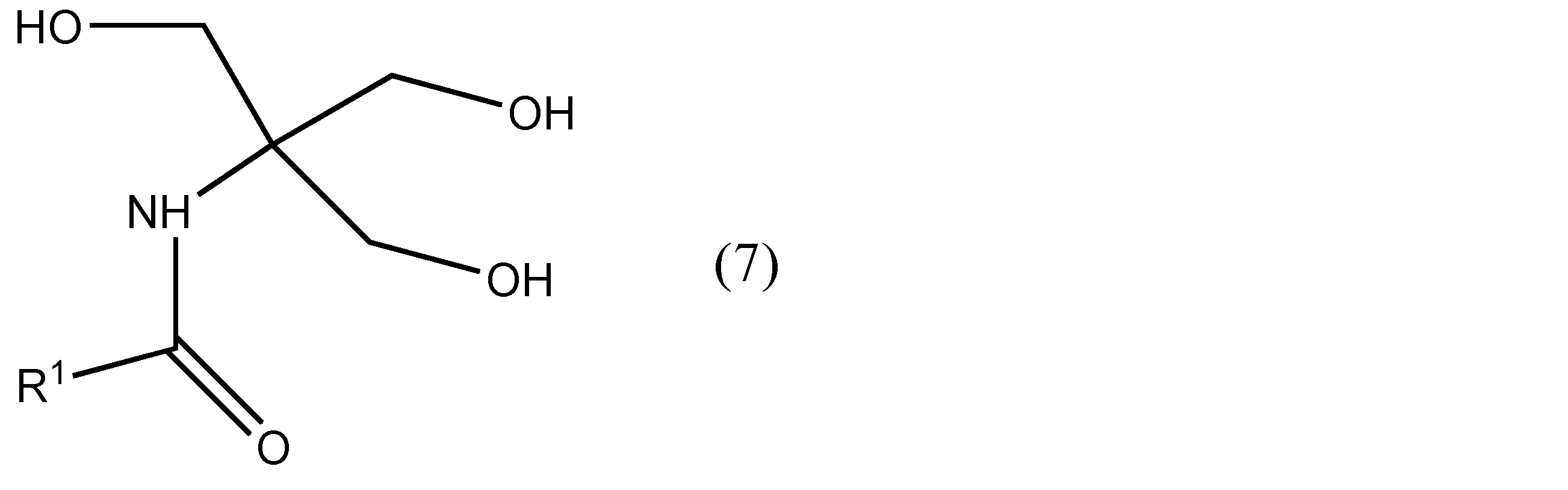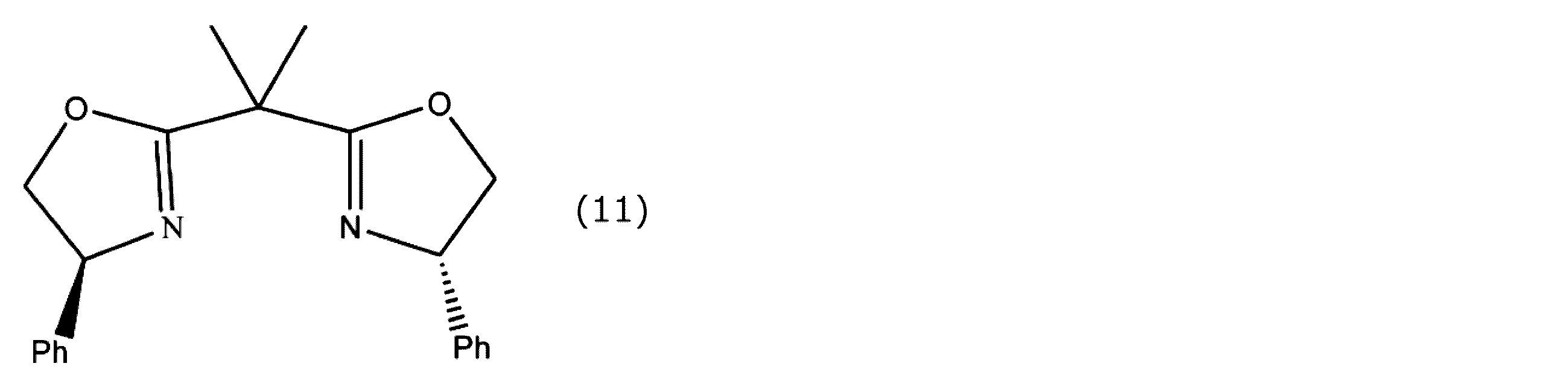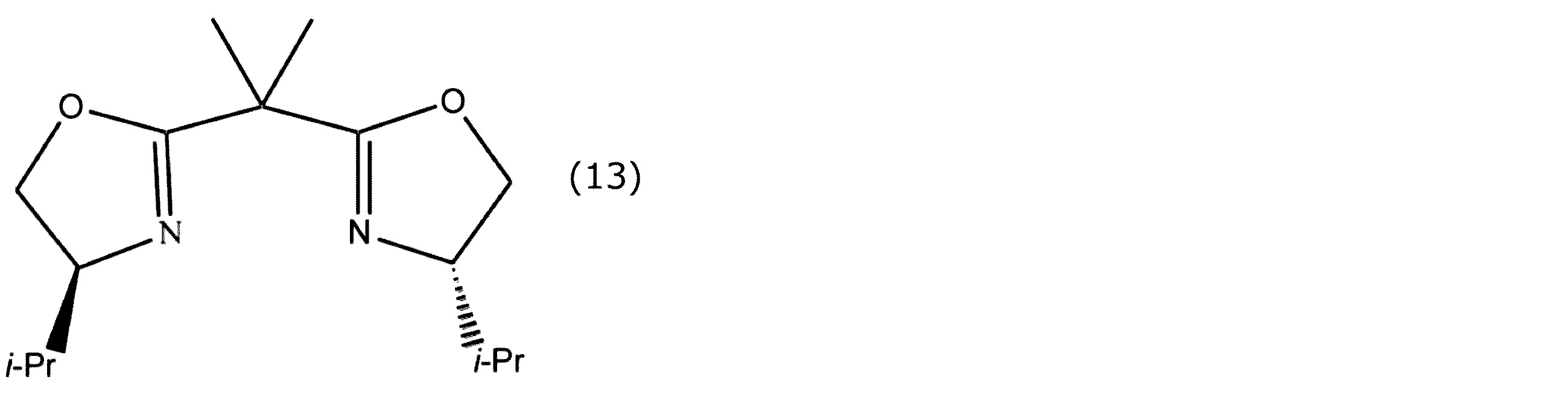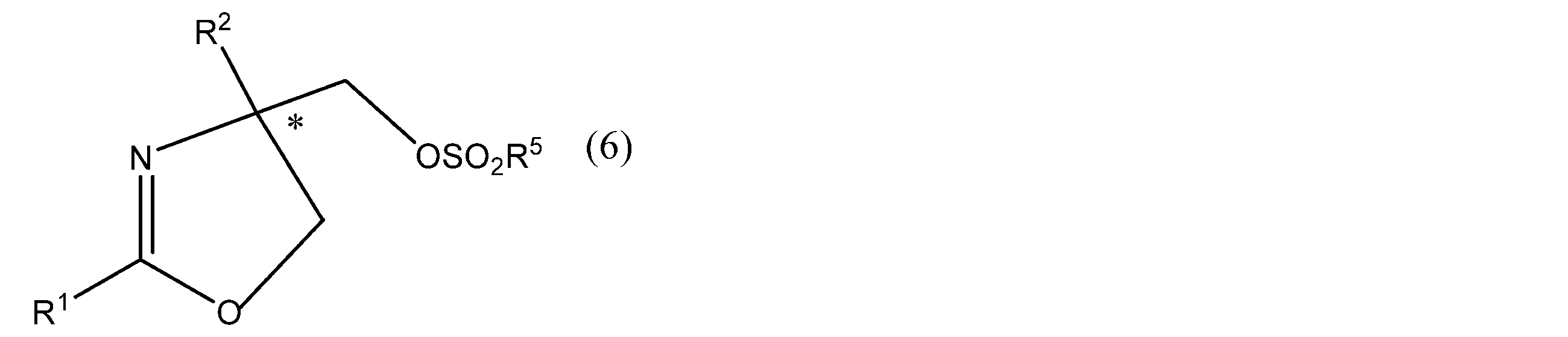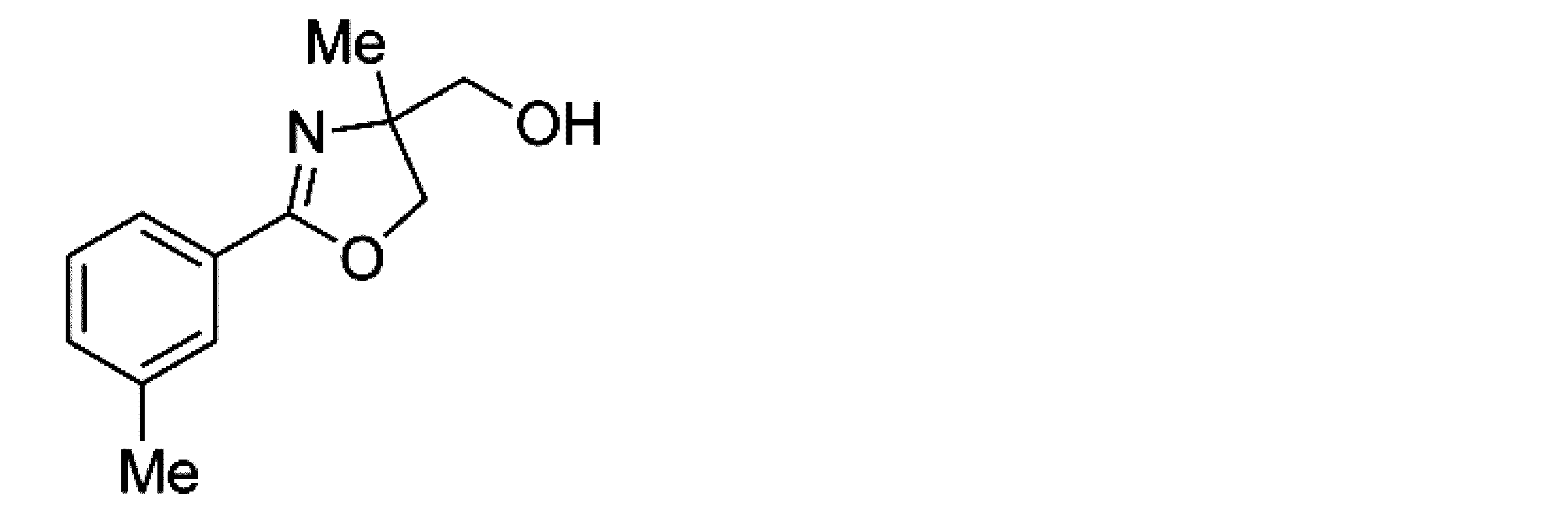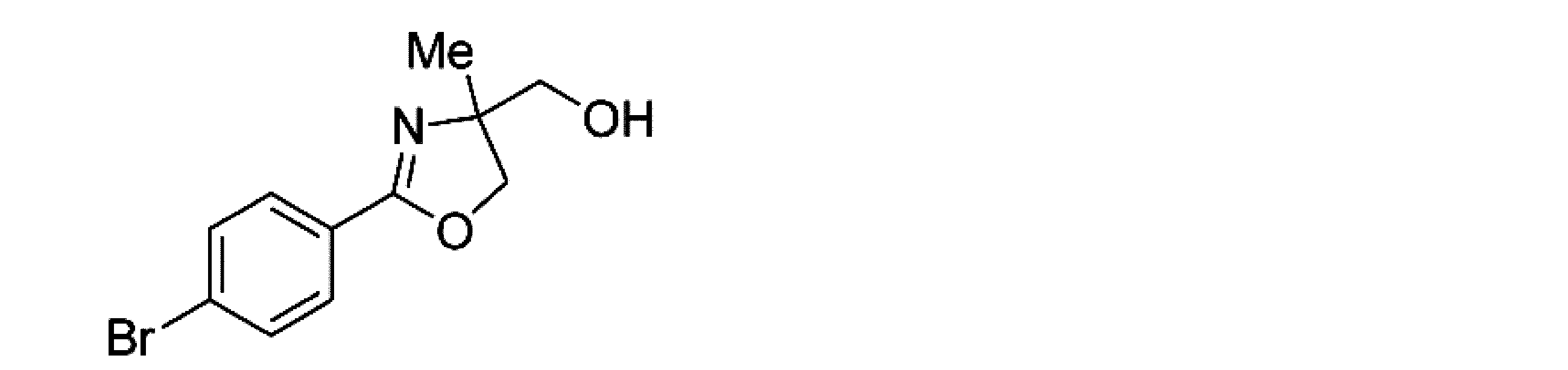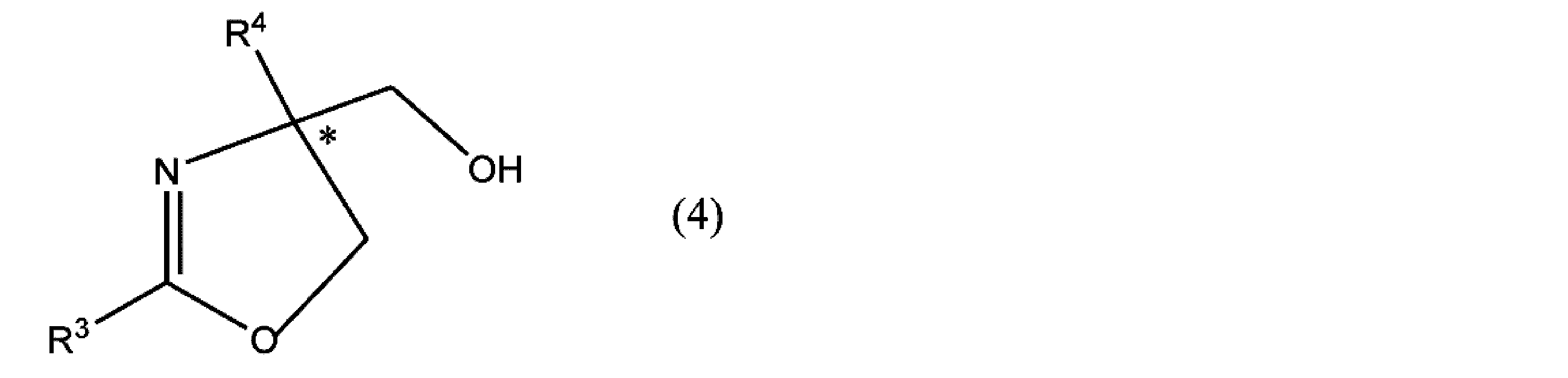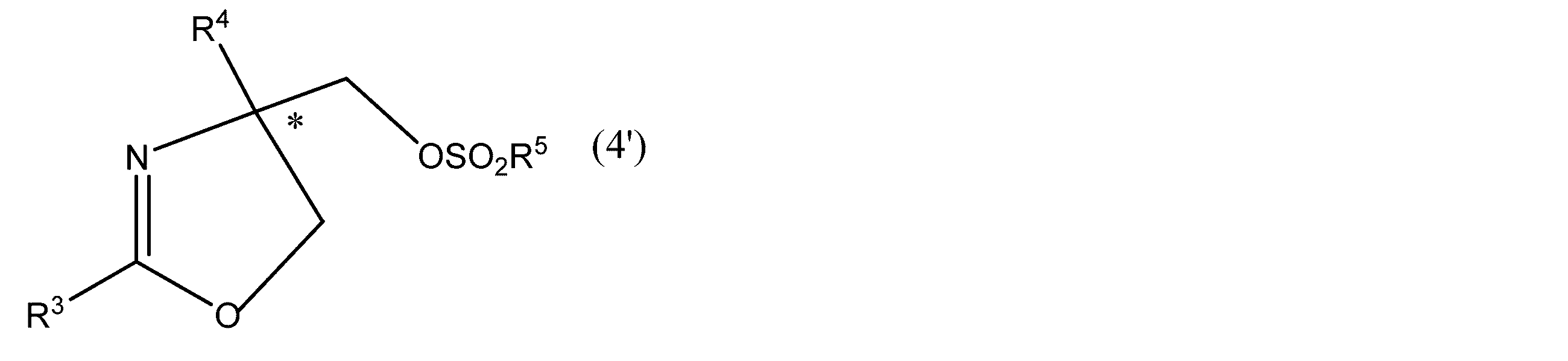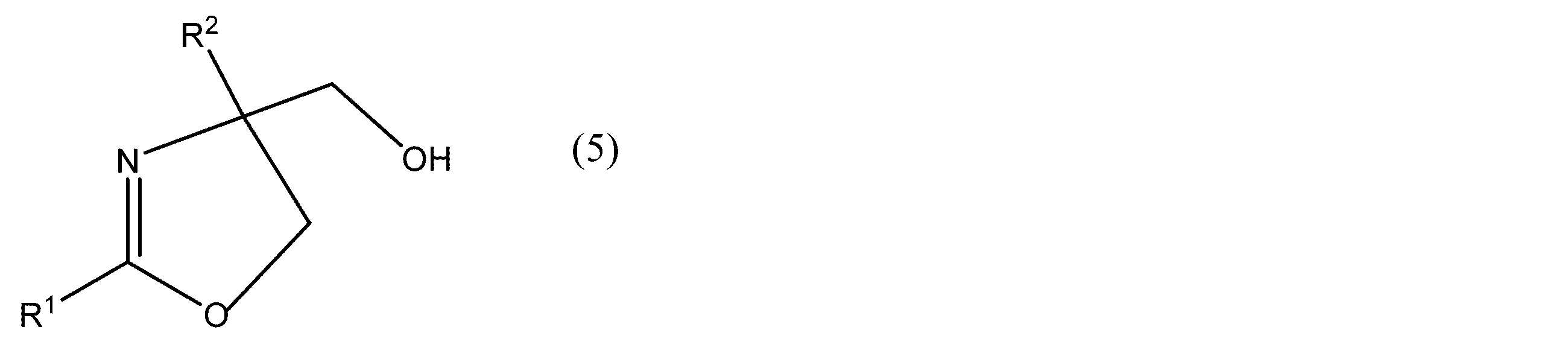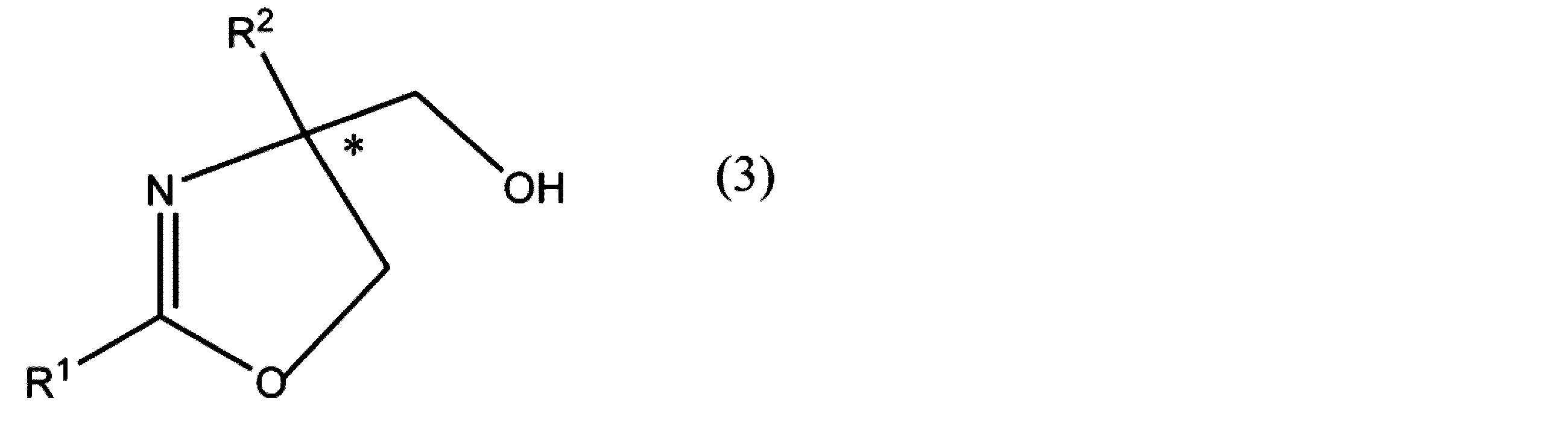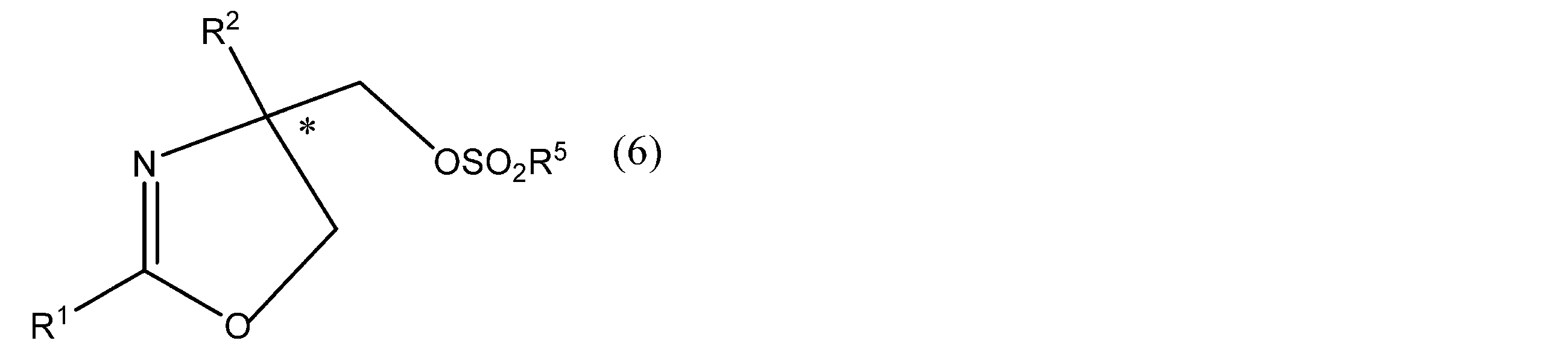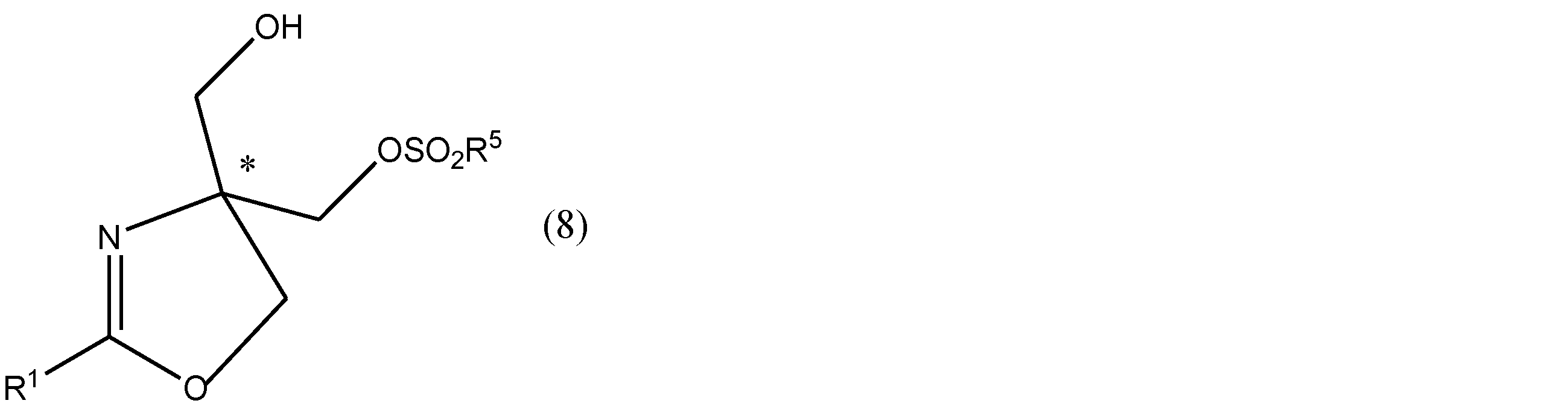WO2012105574A1 - 光学活性化合物又はその塩の製造方法 - Google Patents
光学活性化合物又はその塩の製造方法 Download PDFInfo
- Publication number
- WO2012105574A1 WO2012105574A1 PCT/JP2012/052176 JP2012052176W WO2012105574A1 WO 2012105574 A1 WO2012105574 A1 WO 2012105574A1 JP 2012052176 W JP2012052176 W JP 2012052176W WO 2012105574 A1 WO2012105574 A1 WO 2012105574A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- formula
- optically active
- compound
- atom
- Prior art date
Links
- 0 *CC1(*)N=C(*)OC1 Chemical compound *CC1(*)N=C(*)OC1 0.000 description 5
- LJXJINIRRQOTIO-UHFFFAOYSA-N CC1(CO)N=C(c2ccc[nH]2)OC1 Chemical compound CC1(CO)N=C(c2ccc[nH]2)OC1 LJXJINIRRQOTIO-UHFFFAOYSA-N 0.000 description 1
- IKNRMFZYJWKAMF-UHFFFAOYSA-N CC1(CO)N=C(c2ccc[o]2)OC1 Chemical compound CC1(CO)N=C(c2ccc[o]2)OC1 IKNRMFZYJWKAMF-UHFFFAOYSA-N 0.000 description 1
- BUQQXIUNDVIPQK-UHFFFAOYSA-N OCC(CO)(CO)NC(I)=O Chemical compound OCC(CO)(CO)NC(I)=O BUQQXIUNDVIPQK-UHFFFAOYSA-N 0.000 description 1
- DPDSPQXVICKZCL-UHFFFAOYSA-N OCC1(COS(c(cc2)ccc2Cl)(=O)=O)N=C(c2ccccc2)OC1 Chemical compound OCC1(COS(c(cc2)ccc2Cl)(=O)=O)N=C(c2ccccc2)OC1 DPDSPQXVICKZCL-UHFFFAOYSA-N 0.000 description 1
- SVKHMEAHRCGFDK-UHFFFAOYSA-N OCC1(COS(c2ccccc2)(=O)=O)N=C(c2ccccc2)OC1 Chemical compound OCC1(COS(c2ccccc2)(=O)=O)N=C(c2ccccc2)OC1 SVKHMEAHRCGFDK-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D263/00—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings
- C07D263/02—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings
- C07D263/08—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D263/10—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D263/14—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms with radicals substituted by oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B53/00—Asymmetric syntheses
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
Definitions
- the present invention relates to a method for producing an optically active oxazoline or a salt thereof useful as a synthetic intermediate for pharmaceuticals or agricultural chemicals or a synthetic intermediate for many chemical substances, an optically active oxazoline or a salt thereof produced by the above production method, and an oxazoline. It relates to optical resolution.
- optically active oxazolines are useful as building blocks and asymmetric ligands for organic synthesis, various production methods have been studied.
- a method for producing the optically active oxazoline for example, methods described in Non-Patent Documents 1 to 3 are known.
- any of the above methods needs to use a compound having optical activity as a raw material. That is, there is a problem that the cost of raw materials is high and the product price is increased. Moreover, since the said method requires a several process for manufacture, there existed a problem that a manufacturing facility became large, and it took time for separation, and the yield fell.
- the main object of the present invention is to provide a production method for synthesizing an optically active oxazoline with few production steps using a compound having no optical activity as a raw material compound.
- the present inventor has conducted research to solve the above-mentioned problems, and has found that the above-mentioned object can be solved at once by the following chemical reaction. Further, various studies have been made, and the present invention has been completed.
- the present invention includes the following contents [1] to [10], [1] Formula (1)
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent, and R 2 is a hydrogen atom or a group not involved in the reaction.)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 8 is an aryl group having 6 to 20 carbon atoms, 2 to 15 carbon atoms, and containing at least one hetero atom such as a nitrogen atom, an oxygen atom, or a sulfur atom as a hetero atom.
- a monocyclic heteroaryl group, a polycyclic or condensed ring heteroaryl group or a phenyl group substituted with an alkyl group, an alkenyl group, an alkoxy group, a halogen atom, a nitro group or an aryl group [1] Described manufacturing method, [3] The production method according to [1] or [2], wherein R 2 is hydrogen or a hydrocarbon group. [4] The production method according to any one of [1] to [3], wherein M m Z n is Cu (OTf) 2 .
- R 3 is an aryl group having 6 to 20 carbon atoms, 2 to 15 carbon atoms, and includes at least one hetero atom such as a nitrogen atom, an oxygen atom, or a sulfur atom as a hetero atom.
- R 3 and R 4 have the same meaning as in formula (4), R 5 is an optionally substituted alkyl group or phenyl group. * Represents asymmetricality.
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent, and R 2 is a hydrogen atom or a group not involved in the reaction.)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent.)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 1 has the same meaning as in formula (7), and R 5 is an optionally substituted alkyl group or phenyl group. * Represents asymmetric.
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent.)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 1 has the same meaning as in formula (9), and R 5 is an optionally substituted alkyl group or phenyl group. * Represents asymmetric.)
- the (R) body or (S) body of the optically active compound represented by these, or its salt is produced
- an optically active compound represented by the formula (3) or the formula (4) or a salt thereof which is a synthetic intermediate of various useful compounds, can be obtained with few steps by using a compound having no optical activity as a raw material compound Can be manufactured. More specifically, by using a compound having no optical activity represented by formula (1) as a raw material compound, the optically active compound represented by formula (3) or formula (4) or a salt thereof has excellent yield. And can be manufactured with excellent optical purity. Further, since asymmetric asymmetry and ring formation by ring closure can be performed simultaneously, it is industrially advantageous. Further, according to the present invention, it is possible to optically resolve the racemate of the compound represented by the formula (5) as a raw material with an excellent yield and an excellent optical purity.
- an optically active compound represented by the formula (8) or a salt thereof is obtained with an excellent yield and an excellent optical property, using a compound having no optical activity represented by the formula (7) as a raw material compound. It can be manufactured in purity. Moreover, the optically active compound represented by the formula (8) or a salt thereof is produced in an excellent yield and an excellent optical purity from the compound having no optical activity represented by the formula (9) according to the present invention as a raw material. Can be done.
- the present invention is a process for synthesizing optically active oxazolines, particularly compounds represented by the general formulas (3) and (4) or salts thereof with a small number of manufacturing steps, using a compound having no optical activity as a raw material compound.
- a method is provided.
- the present invention also provides a method for optical resolution of a racemate of oxazoline.
- this invention provides the manufacturing method which synthesize
- the manufacturing method of the compound or its salt represented by General formula (3) and (4), and the method of optically resolving the racemic body of an oxazoline are explained in full detail.
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent, and R 2 is a hydrogen atom or a group not involved in the reaction.) It is represented by
- the alkyl group represented by R 1 may be linear or branched, for example, a linear or branched alkyl group having 1 to 20 carbon atoms, such as a methyl group, an ethyl group, n -Propyl group, isopropyl group, n-butyl group, sec-butyl group, isobutyl group, tert-butyl group, n-pentyl group, neopentyl group, tert-pentyl group, isopentyl group, 2-methylbutyl group, 1-ethylpropyl Group, hexyl group, isohexyl group, 4-methylpentyl group, 3-methylpentyl group, 2-methylpentyl group, 1-methylpentyl group, 3,3-dimethylbutyl group, 2,2-dimethylbutyl group, 1, 1-dimethylbutyl group, 1,2-dimethylbutyl group, 1,3-dimethylbutyl group
- the alkynyl group represented by R 1 may be linear or branched, for example, an alkynyl group having 2 to 15 carbon atoms, preferably 2 to 10 carbon atoms, more preferably 2 to 6 carbon atoms, Specific examples include ethynyl group, 1-propynyl group, 2-propynyl group, 1-butynyl group, 3-butynyl group, pentynyl group, hexynyl group and the like.
- the alkenyl group represented by R 1 may be linear or branched, and examples thereof include alkenyl groups having 2 to 15 carbon atoms, preferably 2 to 10 carbon atoms, more preferably 2 to 6 carbon atoms, Specifically, vinyl group, 1-propenyl group, allyl group, 1-butenyl group, 2-butenyl group, 3-butenyl group, pentenyl group, hexenyl group, isopropenyl group, 2-methyl-2-propenyl group, 1 -Methyl-2-propenyl group, 2-methyl-1-propenyl group and the like.
- Examples of the aliphatic heterocyclic group represented by R 1 include, for example, 2 to 14 carbon atoms and at least one hetero atom, preferably 1 to 3 hetero atoms such as nitrogen atom, oxygen atom, sulfur atom and the like. Examples thereof include a 5- to 8-membered, preferably 5- or 6-membered monocyclic aliphatic heterocyclic group, polycyclic or condensed aliphatic heterocyclic group. Examples of the aliphatic heterocyclic group include pyrrolidyl-2-one group, piperidino group, piperazinyl group, morpholino group, tetrahydrofuryl group, tetrahydropyranyl group, tetrahydrothienyl group and the like.
- Examples of the cycloalkyl group represented by R 1 include a cycloalkyl group having 3 to 14 carbon atoms, preferably 5 to 12 carbon atoms, and more preferably 6 to 12 carbon atoms, specifically, a cyclopropyl group. , Cyclobutyl group, cyclopentyl group, methylcyclopentyl group, cyclohexyl group, cycloheptyl group, cyclooctyl group, 1,2-dimethylcyclopentyl group, 1,3-dimethylcyclopentyl group, 1-ethyl-2-methylcyclopentyl group, etc. It is done.
- Examples of the aryl group represented by R 1 include an aryl group having 6 to 20 carbon atoms, and specifically include a phenyl group, a 1-naphthyl group, a 2-naphthyl group, an anthryl group, a phenanthryl group, and 2-biphenyl. Group, 3-biphenyl group, 4-biphenyl group, terphenyl group and the like.
- Examples of the aralkyl group represented by R 1 include a group in which at least one hydrogen atom of the alkyl group is substituted with the aryl group, specifically, an aralkyl group having 7 to 18 carbon atoms.
- the aromatic heterocyclic group represented by R 1 includes, for example, 2 to 15 carbon atoms and at least one hetero atom, preferably 1 to 3 hetero atoms such as nitrogen atom, oxygen atom and sulfur atom. And a 5- to 8-membered, preferably 5- or 6-membered monocyclic heteroaryl group, or a polycyclic or fused-ring heteroaryl group.
- Examples of the 5- or 6-membered monocyclic heteroaryl group, polycyclic or condensed ring heteroaryl group include a furyl group, a thienyl group, a pyrrolyl group, a pyridyl group, a pyrimidyl group, a pyrazyl group, a pyridazyl group, and a pyrazolyl group.
- Imidazolyl group Imidazolyl group, oxazolyl group, thiazolyl group, benzofuryl group, benzothienyl group, quinolyl group, isoquinolyl group, quinoxalyl group, phthalazyl group, quinazolyl group, naphthyridyl group, cinnolyl group, benzoimidazolyl group, benzoxazolyl group, benzothiazolyl group, etc. Is mentioned.
- Any hydrogen atom of the alkyl group, alkynyl group, alkenyl group, aliphatic heterocyclic group, cycloalkyl group, aryl group, aralkyl group, or aromatic heterocyclic group may be substituted with a substituent.
- the substituent is not particularly limited.
- the number of the substituents is preferably 1 to 3, more preferably 1 or 2.
- the alkyl group as a substituent may be linear or branched, for example, a linear or branched alkyl group having 1 to 20 carbon atoms. Specifically, it is the same as the alkyl group as R 1 described above.
- the alkynyl group as a substituent may be linear or branched, and examples thereof include alkynyl groups having 2 to 15 carbon atoms, preferably 2 to 10 carbon atoms, and more preferably 2 to 6 carbon atoms. Specifically, it is the same as the alkynyl group as R 1 described above.
- the alkenyl group as a substituent may be linear or branched, and examples thereof include alkenyl groups having 2 to 15 carbon atoms, preferably 2 to 10 carbon atoms, more preferably 2 to 6 carbon atoms. Is the same as the alkenyl group as R 1 described above.
- Examples of the aliphatic heterocyclic group as a substituent include 2 to 14 carbon atoms and at least one hetero atom, preferably 1 to 3 hetero atoms such as a nitrogen atom, an oxygen atom, and a sulfur atom. And a 5- to 8-membered, preferably 5- or 6-membered monocyclic aliphatic heterocyclic group, and a polycyclic or condensed aliphatic heterocyclic group.
- the cycloalkyl group as the substituent for example 3 to 14 carbon atoms, preferably from 5 to 12 carbon atoms, more preferably be a cycloalkyl group having 6 to 12 carbon atoms, specifically as R 1 above Same as cycloalkyl group.
- Examples of the aryl group as a substituent include an aryl group having 6 to 20 carbon atoms. Specifically, it is the same as the aryl group as R 1 described above.
- Examples of the aryloxy group as a substituent include an aryloxy group having 6 to 14 carbon atoms, and specific examples include a phenoxy group, a tolyloxy group, a xylyloxy group, a naphthoxy group, and an anthryloxy group.
- Examples of the aralkyl group as a substituent include a group in which at least one hydrogen atom of the alkyl group is substituted with the aryl group. Specifically, it is the same as the aralkyl group as R 1 described above.
- the aromatic heterocyclic group as a substituent has, for example, 2 to 15 carbon atoms and contains at least one, preferably 1 to 3 hetero atoms such as nitrogen, oxygen and sulfur atoms as hetero atoms.
- hetero atoms such as nitrogen, oxygen and sulfur atoms as hetero atoms.
- examples thereof include a 5- to 8-membered, preferably 5- or 6-membered monocyclic heteroaryl group, and a polycyclic or fused-ring heteroaryl group. Specifically, it is the same as the aromatic heterocyclic group as R 1 described above.
- the alkoxy group as a substituent may be linear, branched or cyclic, and examples thereof include an alkoxy group having 1 to 6 carbon atoms, and specifically include a methoxy group, an ethoxy group, an n-propoxy group, an iso group.
- alkylenedioxy group as a substituent examples include an alkylenedioxy group having 1 to 3 carbon atoms, and specifically include a methylenedioxy group, an ethylenedioxy group, a trimethylenedioxy group, and a propylenedioxy group. And isopropylidenedioxy group.
- the alkylthio group as a substituent may be linear, branched or cyclic, and examples thereof include an alkylthio group having 1 to 6 carbon atoms. Specific examples include a methylthio group, an ethylthio group, an n-propylthio group, and an isopropyl group. Examples thereof include a thio group, an n-butylthio group, a 2-butylthio group, an isobutylthio group, a tert-butylthio group, a pentylthio group, a hexylthio group, and a cyclohexylthio group.
- Examples of the aralkyloxy group as a substituent include an aralkyloxy group having 7 to 12 carbon atoms, and specifically include a benzyloxy group, a 1-phenylethoxy group, a 2-phenylethoxy group, and a 1-phenylpropoxy group.
- Examples of the arylthio group as a substituent include an arylthio group having 6 to 14 carbon atoms, and specific examples include a phenylthio group, a tolylthio group, a xylylthio group, and a naphthylthio group.
- the heteroaryloxy group as a substituent includes, for example, at least one hetero atom, preferably 1 to 3 hetero atoms such as nitrogen atom, oxygen atom, sulfur atom and the like, having 2 to 14 carbon atoms. Examples include heteroaryloxy groups, and specific examples include 2-pyridyloxy group, 2-pyrazyloxy group, 2-pyrimidyloxy group, 2-quinolyloxy group and the like.
- Examples of the aralkylthio group as a substituent include an aralkylthio group having 7 to 12 carbon atoms, and specific examples include a benzylthio group and a 2-phenethylthio group.
- the heteroarylthio group for example, a heteroarylthio group having 2 to 14 carbon atoms containing at least one, preferably 1 to 3 heteroatoms such as nitrogen, oxygen and sulfur atoms as heteroatoms.
- Specific examples include 2-pyridylthio group, 4-pyridylthio group, 2-benzimidazolylthio group, 2-benzoxazolylthio group, 2-benzthiazolylthio group, and the like.
- Examples of the substituted amino group include an amino group in which one or two hydrogen atoms of the amino group are substituted with a substituent such as an alkyl group, an aryl group, or an aralkyl group.
- Examples of the amino group substituted with an alkyl group that is, an alkyl group-substituted amino group include an N-methylamino group, an N, N-dimethylamino group, an N, N-diethylamino group, an N, N-diisopropylamino group, an N- Examples thereof include mono- or dialkylamino groups such as a cyclohexylamino group.
- Examples of the amino group substituted with an aryl group that is, the aryl group-substituted amino group include N-phenylamino group, N, N-diphenylamino group, N, N-ditolylamino group, N-naphthylamino group, N-naphthyl- Examples thereof include mono- or diarylamino groups such as N-phenylamino group.
- Examples of the amino group substituted with an aralkyl group, that is, an aralkyl group-substituted amino group include mono- or diaralkylamino groups such as N-benzylamino group and N, N-dibenzylamino group.
- one or two or more hydrogen atoms may be substituted with a halogen atom.
- the halogen atom include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom.
- R 1 When the group represented by R 1 is a phenyl group substituted by a substituent, any of ortho-position, meta-position and para-position may be substituted, but from the viewpoint of reactivity and stereoselectivity, The para position is preferably substituted.
- one hydrogen atom may be substituted with a substituent, and two or more hydrogen atoms may be substituted with a substituent. It is preferable that one hydrogen atom is substituted with a substituent.
- R 1 is an aryl group having 6 to 20 carbon atoms, 2 to 15 carbon atoms, and containing at least one hetero atom such as a nitrogen atom, an oxygen atom, or a sulfur atom as a hetero atom.
- a cyclic heteroaryl group, a polycyclic or condensed cyclic heteroaryl group, or a phenyl group substituted with an alkyl group, an alkenyl group, an alkoxy group, a halogen atom, a nitro group, or an aryl group is preferable.
- the group that does not participate in the reaction represented by R 2 may be any group that does not participate in the reaction for producing the compounds represented by the above (3) and (4) or a salt thereof.
- a hydrocarbon group such as an aryl group, an aralkyl group, an alkenyl group, and an alkynyl group is preferable. Specific examples of the alkyl group, aryl group, aralkyl group, alkenyl group, and alkynyl group are the same as those described above for R 1 .
- These raw material compounds may be commercially available products, or may be easily produced by a known method or a method analogous thereto.
- the metal ion represented by M is not particularly limited.
- the metal ion represented by M is not particularly limited.
- Typical metal ions transition metal ions such as iron, copper, cobalt, nickel, mercury, palladium, zinc, silver, rhodium, platinum, scandium, yttrium, lanthanum, cerium, indium, ytterbium, and hafnium.
- transition metal ions are preferable, and iron ions, copper ions, cobalt ions, nickel ions, copper ions, zinc ions, silver ions, rhodium ions, palladium ions, and platinum ions are more preferable. It is more preferable that it is an ion, and it is especially preferable that it is a copper ion.
- M for example, fluorine, chlorine, bromine, halogen ions such as iodine, trifluoromethylsulfonyl ion (- OTf), boron tetrafluoride ion (- BF 4), hexafluorophosphate phosphorus (- PF 6), hexafluoroantimonate ion (- SbF 6), bis (trifluoromethylsulfonyl) imide ion (- NTf 2), acetate ion (- OAc), tri trifluoroacetate ion (- OCOCF 3), and the like.
- Z is fluoride ion, chloride ion, - OTf, - BF 4 or - is preferably PF 6, - and particularly preferably OTf.
- M and n are integers from 1 to 4.
- the above m and n take appropriate values according to the valence of M and the valence of Z so that the Lewis acid is electrically neutral.
- the Lewis acid represented by the above formula (2) may be a combination of those mentioned as M and Z.
- Cu (OTf) 2 , Zn (OTf) 2 , CuBr 2 , CuCl 2 , CuF 2 , Cu (BF 4 ) 2 , Cu (PF 6 ) 2 , Cu (SbF 6 ) 2, Cu (NTf 2 ) 2, PtCl 2 , CoCl 2 , PdCl 2 , Pd (OAc) 2, Pd (OCOCF 3 ) 2 Can be mentioned.
- Cu (OTf) 2 , Zn (OTf) 2 , CuCl 2 , CuF 2 , Cu (BF 4 ) 2 , Cu (PF 6 ) 2 , PtCl 2 , CoCl 2, PdCl 2 , Pd (OAc) 2, Pd ( OCOCF 3 ) 2 is preferable, and Cu (OTf) 2 is particularly preferable.
- the Lewis acid may be a commercially available product or may be produced by a known method.
- the amount of the Lewis acid used may be 0.001 to 1.00 mol, more preferably 0.005 mol to 0.50 mol, relative to 1 mol of the compound represented by the formula (1). Is particularly preferably from 0.01 to 0.20 mol.
- the asymmetric ligand having one or more coordination sites used in the ring closure reaction is not particularly limited, and examples thereof include an optically active diamine compound and an optically active oxazoline compound, and an optically active oxazoline compound is preferable.
- the bisoxazoline compound is referred to as Box.
- each of the oxazoline rings in the Box has a phenyl group bonded thereto (hereinafter referred to as Ph-Box), an isopropyl group bonded (hereinafter referred to as i-Pr-Box), and t-Bu bonded ( Hereinafter referred to as t-Bu-Box) and those having a benzyl group bonded thereto.
- the optically active oxazoline compound include those in which substituents having the same steric configuration are bonded to each of the oxazoline rings.
- part which the said asymmetric ligand has it is preferable that it is two or more.
- the amount of the asymmetric ligand used is 0.001 to 1.00 mol per mol of the compound represented by the formula (1), and 0.005 mol to 0.50 mol. More preferably, it is 0.01 to 0.20 mol.
- the optically active bisoxazoline compound a commercially available product can be used, or it can be produced by a method known per se. Examples of the production method include J. Org. Am. Chem. Soc. 113, 728-729 (1991).
- the asymmetric ligand forms a metal complex by coordination with the Lewis acid and acts as a catalyst in the production of an optically active compound.
- a sulfonyl halide having an optionally substituted alkyl group or phenyl group is used.
- the optionally substituted phenyl group include those in which a phenyl group or an arbitrary hydrogen atom thereof is substituted with a substituent.
- Specific examples of the phenyl group in the sulfonyl halide having an alkyl group or a phenyl group which may be substituted include those in which the phenyl group or an arbitrary hydrogen atom thereof is substituted with the substituent exemplified in R 1.
- C1-C4 alkylphenyl groups such as paramethylphenyl group, paraethylphenyl group, parapropylphenyl group, paraisopropylphenyl group, parabutylphenyl group; paramethoxyphenyl group, paraethoxyphenyl group, paraisopropoxyphenyl group
- An alkoxyphenyl group having 1 to 4 carbon atoms such as para n-butoxyphenyl group; a halophenyl group such as parafluorophenyl group, parachlorophenyl group, parabromophenyl group, paraiodophenyl group; paracarbamoylphenyl group, paraacylamino Phenyl group, para A midphenyl group, a paranitrophenyl group, and the like are preferable, and a paramethylphenyl group, a parachlorophenyl group, a paramethoxyphenyl group, and a paranitropheny
- one hydrogen atom may be substituted with a substituent, and two or more hydrogen atoms may be substituted with a substituent, but one hydrogen atom is a substituent. It is preferably substituted.
- the substitution site of the phenyl group which may be substituted is not particularly limited, and any of the ortho, meta, and para positions may be substituted, but the para position is preferably substituted.
- the alkyl group which may be substituted may be linear or branched, for example, a linear or branched alkyl group having 1 to 20 carbon atoms or an arbitrary hydrogen atom thereof substituted with a substituent. Is mentioned.
- linear or branched alkyl group examples include, for example, methyl group, ethyl group, n-propyl group, isopropyl group, n-butyl group, sec-butyl group, isobutyl group, tert-butyl group, n -Pentyl group, neopentyl group, tert-pentyl group, isopentyl group, 2-methylbutyl group, 1-ethylpropyl group, hexyl group, isohexyl group, 4-methylpentyl group, 3-methylpentyl group, 2-methylpentyl group, 1-methylpentyl group, 3,3-dimethylbutyl group, 2,2-dimethylbutyl group, 1,1-dimethylbutyl group, 1,2-dimethylbutyl group, 1,3-dimethylbutyl group, 2,3- Dimethylbutyl, 1-ethylbutyl, 2-ethylbutyl, 2-e
- the optionally substituted alkyl group preferably has 1 to 4 carbon atoms, and more preferably a methyl group or an ethyl group.
- one or more hydrogen atoms may be substituted with a substituent.
- the substitution site of the alkyl group which may be substituted is not particularly limited.
- the substituent the same groups as those exemplified as the substituent in the above (1) can be used.
- a halogen atom in the said sulfonyl halide For example, a fluorine atom, a chlorine atom, a bromine atom, an iodine atom etc.
- the sulfonyl halide is used in an amount of 0.1 to 10 mol, preferably 0.5 to 5.0 mol, based on 1 mol of the raw material represented by the formula (1), It is particularly preferably 8 to 4.0 mol.
- a compound having no optical activity can be used as a raw material compound, and an objective compound having optical activity can be produced with an excellent yield and an excellent optical purity.
- the catalyst may be a homogeneous catalyst or a heterogeneous catalyst.
- the catalyst may be produced according to a known method, or a commercially available one may be used. In addition to the above catalyst, other known catalysts may be used.
- solvent It does not specifically limit as a solvent used for the said reaction, Both an organic solvent and an inorganic solvent can be used.
- the organic solvent include aliphatic hydrocarbons such as pentane, hexane, heptane, octane, and cyclohexane; aromatic hydrocarbons such as benzene, toluene, and xylene; dichloromethane, 1,2-dichloroethane, chloroform, carbon tetrachloride, o -Halogenated hydrocarbons such as dichlorobenzene; alcohols such as methanol, ethanol, isopropyl alcohol, t-butyl alcohol, t-amyl alcohol; dimethyl ether, ethyl methyl ether, diethyl ether, diisopropyl ether, diglyme, tert-butyl methyl Ethers such as ether, dimethoxyethane, ethylene glycol diethyl
- inorganic solvents include acidic aqueous solutions such as hydrochloric acid, sulfuric acid, nitric acid, carbonic acid, and phosphoric acid; basic aqueous solutions such as sodium hydroxide, magnesium hydroxide, calcium hydroxide, potassium hydroxide, and sodium hydrogen carbonate; pure water and saline And neutral aqueous solutions.
- acidic aqueous solutions such as hydrochloric acid, sulfuric acid, nitric acid, carbonic acid, and phosphoric acid
- basic aqueous solutions such as sodium hydroxide, magnesium hydroxide, calcium hydroxide, potassium hydroxide, and sodium hydrogen carbonate
- pure water and saline And neutral aqueous solutions include water and saline And neutral aqueous solutions.
- the solvent is preferably an organic solvent, among which acetonitrile, propionitrile, dichloromethane, toluene, ethyl acetate, tetrahydrofuran, diethyl ether, dioxane, diglyme, N, N-dimethylformamide, methanol, ethanol, isopropanol, t-butanol, Particularly preferred are t-amyl alcohol and methyl isobutyl ketone.
- the said solvent may be used independently and may be used in combination of 2 or more type.
- the amount of the solvent used is not particularly limited, but may be an excess amount relative to the raw material, for example, 0.1 to 100 parts by weight with respect to 1 part by weight of the raw material represented by the formula (1). it can.
- base Preferable examples of the base that can be used in the reaction include inorganic bases such as sodium hydroxide, potassium hydroxide, potassium carbonate, sodium carbonate, cesium carbonate, and ammonia, and organic bases such as triethylamine, diisopropylethylamine, and pyridine.
- the amount of the base used may be 0.1 to 10 mol, more preferably 0.5 to 8.0 mol, more preferably 0.8 to 0.8 mol, relative to 1 mol of the raw material of formula (1). It is especially preferable that it is 5.0 mol.
- the compound of formula (1) is an asymmetric ligand having one or more coordination sites, Formula (2)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 1 and R 2 have the same meaning as in formula (1). * Represents asymmetric). Or a salt thereof.
- the above reaction is performed in two steps at once.
- the sulfonyl halide having an optionally substituted alkyl group or phenyl group is paratoluenesulfonyl halide
- the asymmetrically asymmetric formula (14) is used as an intermediate product.
- a compound having optical activity can be produced using a compound having no optical activity as a raw material. That is, since an inexpensive compound having no optical activity can be used as a raw material instead of an expensive compound having optical activity, the manufacturing cost can be reduced.
- the above reaction can be performed simultaneously with asymmetric asymmetry and ring formation by ring closure, it is possible to reduce the number of steps and synthesize an optically active compound with a smaller production facility. There are advantages such as being able to reduce the labor of separation of the compound, and preventing the yield from being lowered by the separation process.
- the oxazoline compound having an optical activity represented by the formula (3) or the formula (4) or a salt thereof is generated. It may be possible to use the present invention for the preparation of compounds.
- the optical activity of the oxazoline compound (formula (3)) produced is determined according to the stereoselectivity of the asymmetric ligand used. For example, when (R, R) -Ph-Box is used as the asymmetric ligand, the (R) oxazoline compound is preferentially produced as compared with the (S) oxazoline compound.
- the (S) oxazoline compound is also once generated in a certain amount.
- the hydrogen atom of the hydroxyl group of the (S) isomer is a group corresponding to the sulfonyl halide used, that is, from the sulfonyl halide to the halogen
- the hydrogen atom of the hydroxyl group of (S) is converted to paratoluenesulfonyl group (-Ts).
- the (S) oxazoline compound having a hydroxyl group is further reduced, and the stereoselectivity of the (R) isomer is increased.
- the oxazoline compound into which the sulfonyloxy group has been introduced is an (R) isomer in terms of the compound name, but the configuration of the asymmetric carbon is the same as the (S) oxazoline compound having a hydroxyl group. is there.
- optical activity means that it is not an equimolar mixture (for example, racemate) of isomers different in configuration at an asymmetric carbon, and one stereoisomer is present in excess. If so, it is defined as optical activity.
- the reaction temperature at which the above reaction is performed is not particularly limited.
- the reaction can be performed at a temperature of ⁇ 20 ° C. to 80 ° C., and it is preferably performed at a temperature of 5 ° C. to 40 ° C.
- a pressure which performs the said reaction Any of pressure reduction, atmospheric pressure, or pressurization may be sufficient.
- the reaction can be performed at a pressure of 0.1 to 20 Pa, preferably 0.7 to 1.2 Pa. That is, since this reaction can proceed under normal temperature and normal pressure, the production facility can be simplified, thereby contributing to the reduction of production cost.
- the reaction time for carrying out the above reaction is not particularly limited and can be, for example, 5 minutes to 72 hours, but preferably 10 minutes to 48 hours.
- the reaction can proceed by mixing the raw materials, preferably with stirring.
- R 3 is an aryl group having 6 to 20 carbon atoms, 2 to 15 carbon atoms, and includes at least one hetero atom such as a nitrogen atom, an oxygen atom, or a sulfur atom as a hetero atom.
- Examples of the aryl group having 6 to 20 carbon atoms represented by R 3 include phenyl group, 1-naphthyl group, 2-naphthyl group, anthryl group, phenanthryl group, 2-biphenyl group, 3-biphenyl group, 4-biphenyl. Group, terphenyl group and the like.
- Specific examples of the polycyclic or condensed cyclic heteroaryl group include, for example, furyl group, thienyl group, pyrrolyl group, pyridyl group, pyrimidyl group, pyrazyl group, pyridazyl group, pyrazolyl group, imidazolyl group, oxazolyl group, thiazolyl group Benzofuryl group, benzothienyl group, quinolyl group, isoquinolyl group, quinoxalyl group, phthalazyl group, quinazolyl group, naphthyridyl group, cinnolyl group, benzoimidazolyl group, benzoxazolyl group, benzothiazolyl group
- an alkyl group as a substituent an alkenyl group as a substituent, an alkoxy group as a substituent, a halogen atom as a substituent, a nitro group as a substituent, or an aryl group as a substituent include the above R 1.
- an alkyl group as a substituent an alkenyl group as a substituent, an alkoxy group as a substituent, a halogen atom as a substituent, a nitro group as a substituent, or an aryl group as a substituent.
- the group that does not participate in the reaction represented by R 4 in the formula (4) may be any group that does not participate in the reaction for producing the compound represented by the above (4) or a salt thereof.
- Hydrocarbon groups such as alkyl groups, aryl groups, and alkenyl groups are preferred.
- Specific examples of the alkyl group, aryl group, and alkenyl group are the same as those described for R 1 above.
- Specific examples of the compound represented by formula (4), R 1 from the compound represented by the above formula (3) is a phenyl group, except when R 2 is hydrogen atom or a methyl group Can be mentioned.
- the target compound represented by the formula (3) or (4) may be obtained alone or as a salt.
- the salt containing the target compound may be any salt as long as the ion derived from the target compound is ion-bonded to the counter ion.
- inorganic salts such as sodium salt, potassium salt, magnesium salt and calcium salt; angelic acid, lysine, ethanolamine, organic salts such as N, N'-dibenzylethylenediamine; and pharmaceutically acceptable salts such as triterpene alcohol or plant sterols.
- the target compound represented by the formula (3) or (4) generated by the above reaction may be isolated or purified, or may be subjected to the next reaction as a synthetic intermediate without being isolated or purified. Good.
- the means for isolation and purification is carried out by known means. As such means, known means such as distillation, concentration, extraction, crystallization, chromatography, filtration, dialysis, and centrifugation are conveniently employed.
- the target compound represented by the formula (3) or (4) is used for various purposes such as medical use, industrial use, and agricultural use. Specific uses include synthetic intermediates used for the synthesis of useful compounds, and can be used as, for example, building blocks for unnatural amino acid synthesis.
- the above target compounds can be used alone or in a mixed state. Further, the target compound may be used in any state of solid, liquid and gas.
- R 1 and R 2 have the same meanings as in formula (1), R 5 is an optionally substituted alkyl group or phenyl group. * Represents asymmetricality.) And more preferably the formula (4 ′)
- R 3 and R 4 have the same meaning as in formula (4), R 5 has the same meaning as in formula (6). * Represents asymmetric). Or a salt thereof is produced.
- R 1 and R 2 in the formula (6) include the same as those in the above formula (1).
- the alkyl group represented by R 5 in the formula (6) may be linear or branched, and examples thereof include linear or branched alkyl groups having 1 to 20 carbon atoms. Specifically, Is the same as the alkyl group as R 1 described above.
- the substituent for the alkyl group or phenyl group represented by R 5 in formula (6) include an alkyl group, an alkynyl group, an alkenyl group, an aryl group, an alkoxy group, an alkylenedioxy group, an aryloxy group, and an aralkyloxy group.
- heteroaryloxy group alkylthio group, cycloalkyl group, aliphatic heterocyclic group, arylthio group, aralkylthio group, heteroarylthio group, amino group, substituted amino group, cyano group, hydroxyl group, oxo group, nitro group , A mercapto group or a halogen atom, and specifically the same as the substituent for R 1 described above.
- R 3 and R 4 in formula (4 ′) include the same as in formula (4).
- R 5 in formula (4 ′) has the same meaning as in formula (6).
- the compound represented by the above formula (6) or formula (4 ′) can be converted into an oxazoline compound having an optical activity by hydrolyzing a sulfonyloxy group.
- the sulfonyloxy group is converted into an azide group, a cyano group, or an amino group. It can also be used as a synthetic intermediate for use in the synthesis of useful compounds after being converted to the above.
- the compound represented by the formula (6) or the formula (4 ′) may be obtained as a compound alone or as a salt.
- the salt containing the target compound may be any salt as long as the ion derived from the target compound is ion-bonded to the counter ion.
- inorganic salts such as sodium salt, potassium salt, magnesium salt and calcium salt; angelic acid, lysine, ethanolamine, organic salts such as N, N'-dibenzylethylenediamine; and pharmaceutically acceptable salts such as triterpene alcohol or plant sterols.
- the compound represented by the formula (6) or the formula (4 ′) may be isolated or purified, or may be subjected to the next reaction as a synthetic intermediate without being isolated or purified.
- the means for isolation and purification is carried out by known means. As such means, known means such as distillation, concentration, extraction, crystallization, chromatography, filtration, dialysis, and centrifugation are conveniently employed.
- the compound represented by Formula (6) or Formula (4 ′) is used for various purposes such as medical use, industrial use, and agricultural use. Specific uses include synthetic intermediates used in the synthesis of useful compounds. In this case, sulfonyloxy groups are hydrolyzed to form optically active oxazoline compounds, which can also be used as building blocks for unnatural amino acid synthesis.
- the compound represented by the formula (6) or the formula (4 ′) can be used as a synthetic intermediate used for the synthesis of useful compounds after converting the sulfonyloxy group to an azide group, a cyano group, an amino group, etc. These can be used alone or in a mixed state. Further, the target compound may be used in any state of solid, liquid and gas.
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent, and R 2 is a hydrogen atom or a group not involved in the reaction.)
- R 1 and R 2 in the compound represented by the formula (5) have the same meaning as in the formula (1). Since the raw material compound is a racemate (a substance obtained by mixing an equal amount of a pair of optical isomers whose chemical structures are mirror images), it does not have optical activity.
- a Lewis acid represented by Examples of the metal ion represented by M in the above formula (2), the counter anion of M represented by Z, and the Lewis acid are the same as those used in the method for producing the optically active oxazoline or a salt thereof. It is done.
- the amount of the Lewis acid used may be 0.001 to 1.00 mol with respect to 1 mol of the compound represented by the formula (5), and more preferably 0.005 mol to 0.50 mol. Is particularly preferably from 0.01 to 0.20 mol.
- Examples of the asymmetric ligand having one or more coordination sites used in the optical resolution include the same as those used in the method for producing the optically active oxazoline or a salt thereof.
- the amount of the asymmetric ligand used is 0.001 to 1.00 mol, and 0.005 mol to 0.50 mol per mol of the compound represented by the formula (5). Is more preferably 0.01 to 0.20 mol.
- Examples of the sulfonyl halide having an optionally substituted alkyl group or phenyl group used in the optical resolution include the same ones as used in the method for producing the optically active oxazoline or a salt thereof.
- the amount of the sulfonyl halide used may be 0.1 to 10 mol, more preferably 0.3 to 5.0 mol, relative to 1 mol of the raw material represented by the formula (5), Particularly preferred is 0.5 to 3.0 mol.
- solvent It does not specifically limit as a solvent used for the said reaction, although both an organic solvent and an inorganic solvent can be used, the same thing as what was used with the manufacturing method of the said optically active oxazoline or its salt is mentioned.
- the amount of the solvent used is not particularly limited, but may be an excess amount with respect to the raw material, for example, 0.1 to 100 parts by weight with respect to 1 part by weight of the raw material represented by the formula (5). it can.
- base examples of the base that can be used in the reaction include the same ones used in the above-described method for producing an optically active oxazoline or a salt thereof.
- the amount of the base used may be 0.1 to 10 mol, more preferably 0.3 to 8.0 mol, more preferably 0. 1 mol to 1 mol of the raw material represented by the formula (5). It is preferably 5 to 5.0 mol.
- reaction The present invention provides an asymmetric ligand having one or more coordination sites, the racemate of the compound of the above formula (5), Formula (2)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 1 and R 2 have the same meaning as in formula (5), R 5 is an optionally substituted alkyl group or phenyl group. * Represents asymmetricality.) Are optically resolved into the (R) -form or (S) -form of the optically active compound represented by
- an (R) isomer or (S) isomer of the compound having optical activity represented by formula (3) can be optically resolved into the compound (R) or (S) having optical activity.
- the optical activity of the oxazoline compound (formula (3)) to be produced is determined according to the stereoselectivity of the asymmetric ligand used. For example, when (R, R) -Ph-Box is used as the asymmetric ligand, an (R) oxazoline compound represented by the formula (3) is formed.
- the oxazoline compound represented by formula (3) in the (S) form is a group in which the hydrogen atom of the hydroxyl group corresponds to the sulfonyl halide used, that is, the sulfonyl group in which the halogen atom is removed from the sulfonyl halide. Therefore, it is considered that the oxazoline compound represented by the formula (3) in the (S) form is decreased, and the stereoselectivity of the (R) form is relatively increased.
- the oxazoline compound of formula (6) having a sulfonyloxy group introduced is in the (R) form on the notation of the compound name, but the asymmetric carbon configuration is the (S) form of the formula (3) ) Is the same as the oxazoline compound represented by
- reaction conditions examples include the same as those used in the method (1) for producing the optically active oxazoline or a salt thereof.
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent.)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 1 has the same meaning as in formula (7), and R 5 is an optionally substituted alkyl group or phenyl group. * Represents asymmetric.) The method of manufacturing the optically active compound shown by these, or its salt is included.
- R 1 examples of R 1 include the same as those described for the general formula (1) of the optically active oxazoline or a salt production method (1).
- Examples of the Lewis acid, the asymmetric ligand, the production method, the addition amount, and the like are the same as in the production method (1) of the optically active oxazoline or a salt thereof.
- Examples of the solvent include those similar to the method (1) for producing optically active oxazoline or a salt thereof, but MeCN (acetonitrile) is particularly preferable.
- Examples of the base include those similar to the method (1) for producing an optically active oxazoline or a salt thereof, but K 2 CO 3 or Na 2 CO 3 is particularly preferable.
- the amount of the base used may be 0.2 to 20 mol, more preferably 1.0 to 16.0 mol, with respect to 1 mol of the raw material represented by formula (7). Particularly preferred is .6 to 10.0 moles.
- Examples of the sulfonyl halide having an optionally substituted alkyl group or phenyl group include those similar to the production method (1) of optically active oxazoline or a salt thereof.
- the phenylsulfonyl halide or the 4-position is an alkyl group.
- phenylsulfonyl halide substituted with a halogen atom is particularly preferable.
- the amount of sulfonyl halide used may be 0.2 to 20 mol, preferably 1.0 to 10.0 mol, per 1 mol of the raw material represented by the formula (7). Particularly preferred is 6 to 8.0 moles.
- the reaction time for carrying out the above reaction is not particularly limited and can be, for example, 5 minutes to 96 hours, but preferably 10 minutes to 72 hours.
- a compound having optical activity represented by formula (8) can be produced using a cheap and readily available compound represented by formula (7) as a raw material.
- an oxazoline ring is formed by a ring-closing reaction following sulfonylation of one hydroxyl group of the raw material compound, and the following formula (9)
- the hydrogen atom of one hydroxyl group of the intermediate product is stereoselectively substituted with a group derived from the sulfonyl halide to be used (that is, a sulfonyl group obtained by removing a halogen atom from the sulfonyl halide).
- a group derived from the sulfonyl halide to be used that is, a sulfonyl group obtained by removing a halogen atom from the sulfonyl halide.
- R 1 represents an alkyl group, an alkynyl group, an alkenyl group, an aliphatic heterocyclic group, a cycloalkyl group, an aryl group, an aralkyl group, or an aromatic heterocyclic group, and any hydrogen atom in these groups is (It may be substituted with a substituent.)
- M is a metal ion
- Z is a counter anion of M
- m and n are integers of 1 to 4.
- R 1 has the same meaning as in formula (9), and R 5 is an optionally substituted alkyl group or phenyl group. * Represents asymmetric.)
- the (R) body or the (S) body of the optically active compound represented by these, or its salt is produced
- R 1 examples of R 1 include the same as those described for the general formula (1) of the optically active oxazoline or a salt production method (1).
- Illustrative examples of the Lewis acid, asymmetric ligand, production method, addition amount, and the like are the same as in production method (1) of optically active oxazoline or a salt thereof.
- the kind of solvent, the kind of base, the kind of sulfonyl halide having an alkyl group or phenyl group which may be substituted, and the reaction time are the same as in the production method (2) of optically active oxazoline or a salt thereof.
- a compound having optical activity represented by formula (8) can be produced using a compound represented by formula (9) as a raw material.
- High-performance liquid chromatography uses “LC-20AT” manufactured by Shimadzu Corporation as a liquid feed pump, “SPD-20A” manufactured by Shimadzu Corporation as a UV detector, and “C-R8A CHROMATAPAC” as a recording device.
- the measurement was performed using “CHIRALPAC” series manufactured by Daicel Chemical Industries, Ltd. as an optically active column. The optical purity was determined from the difference in retention time in HPLC.
- Mass spectrometry (MS) was measured using “JMS-700N” manufactured by JEOL. Column chromatography was measured using “Silica Gel 60, spherical, neutrality” manufactured by Nacalai Tesque. Unless otherwise stated, commercially available products and solvents were used for the reaction. Further, anhydrous sodium sulfate was used for drying the extraction solvent.
- Example 1 Production of Optically Active Oxazoline Compound (1) The experiment was performed at normal temperature and normal pressure. In 2 ml of tert-butanol, 0.1 mmol of Cu (OTf) 2 and 0.1 mmol of (R, R) -Ph-Box were mixed and stirred for 10 minutes. In this solution, 0.5 mmol of the following formula as a raw material
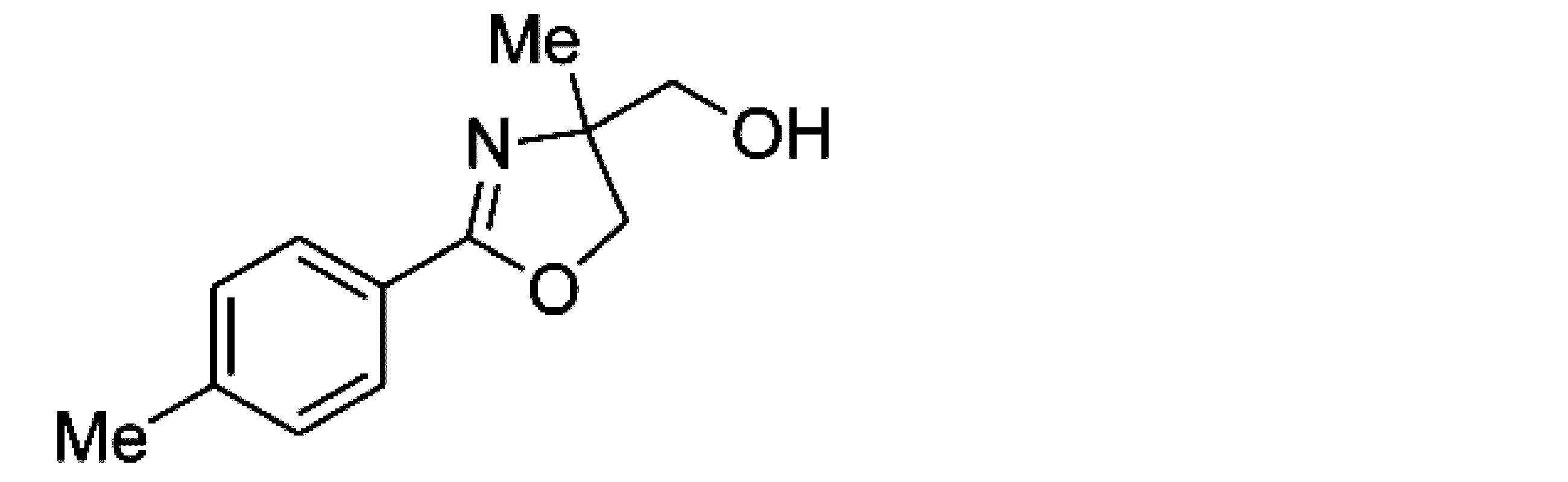
- Example 2 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was substituted with a methyl group, (+)-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (4-methylphenyl) oxazole was obtained. The yield was 89%. When the optical purity was measured, it was 94% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 3 When an experiment was conducted in the same manner as in Example 1 except that the 3-position of the phenyl group in the raw material of Example 1 was substituted with a methyl group, (-)-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (3-methylphenyl) oxazol was obtained. The yield was 73%. When the optical purity was measured, it was 99% or more ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 4 When an experiment was conducted in the same manner as in Example 1 except that the 2-position of the phenyl group in the raw material of Example 1 was substituted with a methyl group, (+)-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (2-methylphenyl) oxazol was obtained. The yield was 86%. When the optical purity was measured, it was 92% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
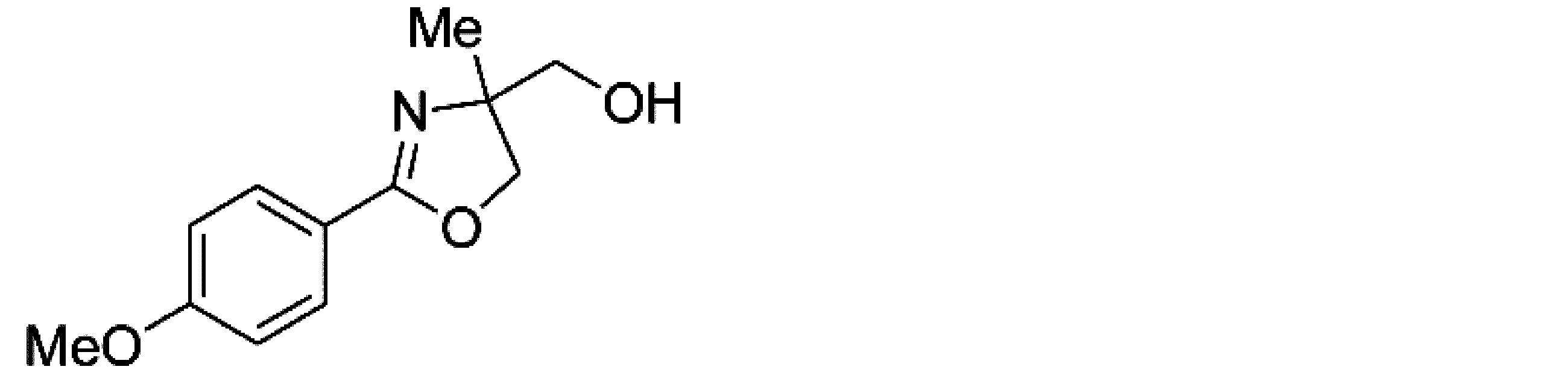
- Example 5 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was replaced with a methoxy group, (-)-4,5-dihydro-4-hydroxymethyl-2 -(4-methoxylphenyl) -4-methyloxazole was obtained. The yield was 64%. When the optical purity was measured, it was 94% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 6 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was replaced with fluorine, (-)-4,5-dihydro-4-hydroxymethyl-2- (4-fluorophenyl) -4-methyloxazole was obtained. The yield was 91%. When the optical purity was measured, it was 94% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
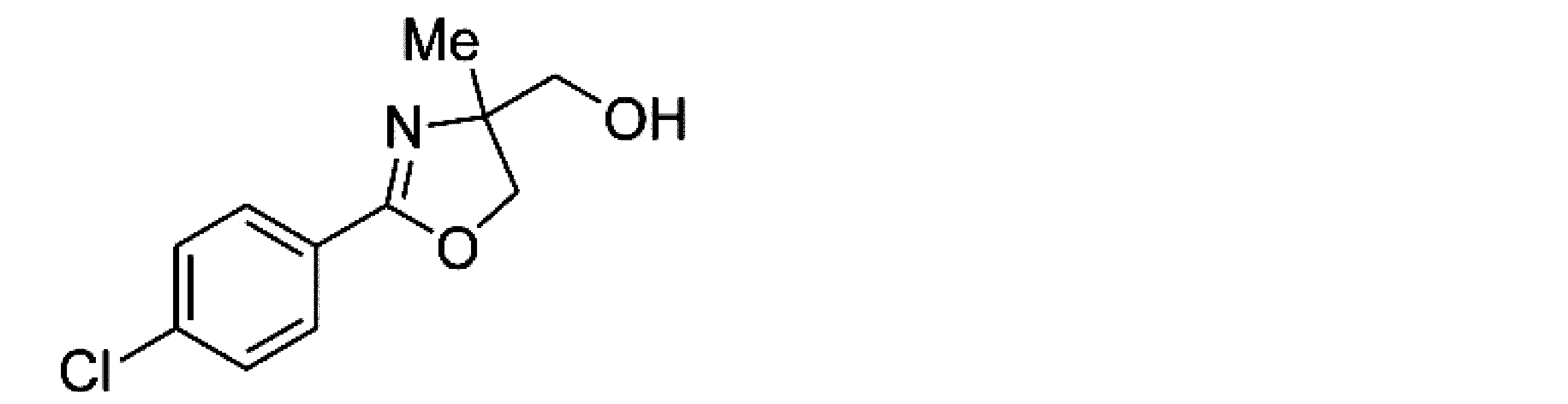
- Example 7 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was replaced with chlorine, (-)-2- (4-chlorophenyl) -4,5- Dihydro-4-hydroxymethyl-4-methyloxazole was obtained. The yield was 97%. When the optical purity was measured, it was 96% ee R-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 8 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was replaced with bromine, (-)-4- (4-bromophenyl) -4,5- Dihydro-2-hydroxymethyl-2-methyloxazole was obtained. The yield was 98%. When the optical purity was measured, it was 92% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
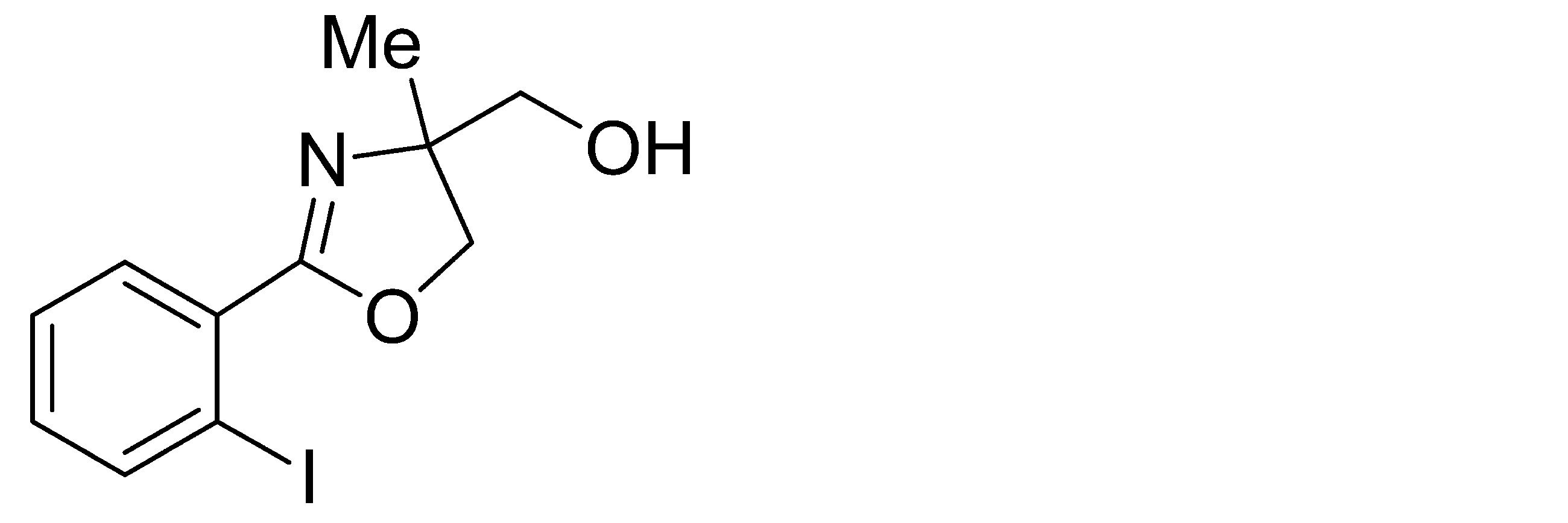
- Example 9 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was replaced by iodine, (-)-4,5-dihydro-4-hydroxymethyl-2- (4-iodophenyl) -4-methyloxazole was obtained. The yield was 97%. When the optical purity was measured, it was 87% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
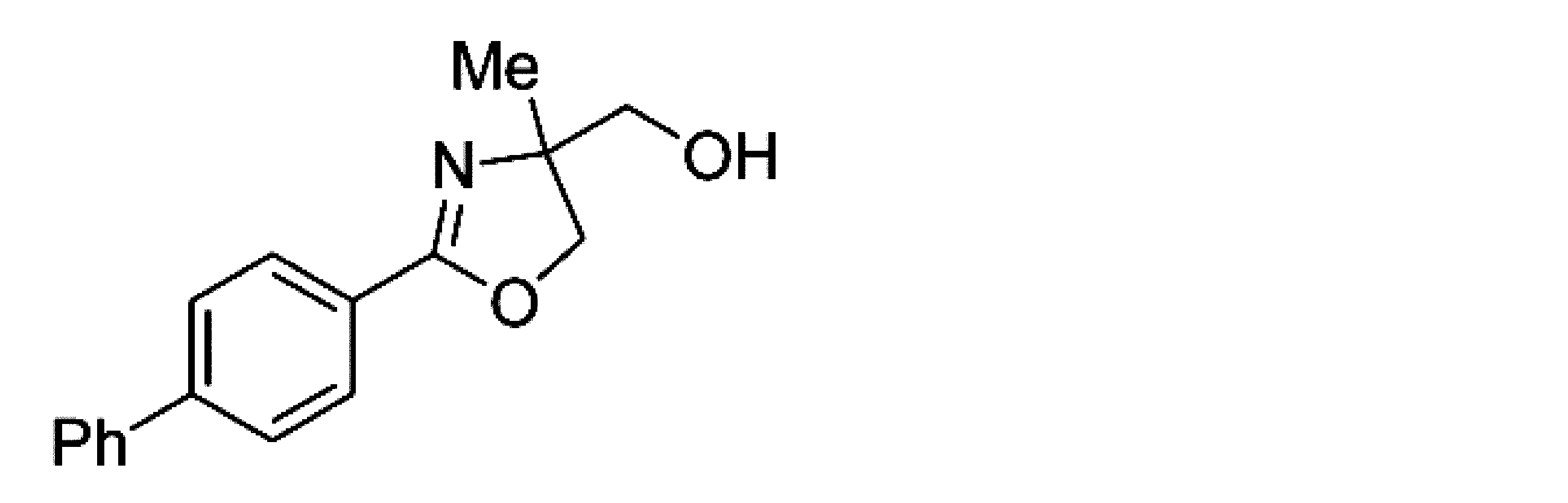
- Example 10 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was replaced with a phenyl group, (-)-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (4-phenylphenyl) oxazole was obtained. The yield was 91%. When the optical purity was measured, it was 91% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 11 When an experiment was conducted in the same manner as in Example 1 except that the 4-position of the phenyl group in the raw material of Example 1 was substituted with a nitro group, (-)-4,5-dihydro-4-hydroxymethyl-4- methyl-2- (4-nitrophenyl) oxazol was obtained. The yield was 91%. When the optical purity was measured, it was 93% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 12 When an experiment was conducted in the same manner as in Example 1 except that the methyl group in the raw material of Example 1 was replaced with a hydrogen atom, (+)-4,5-dihydro-4-hydroxymethyl-2-phenyloxazole was obtained. Obtained. The yield was 32%. When optical purity was measured, it was 89% ee R-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 13 When an experiment was conducted in the same manner as in Example 1 except that the methyl group in the raw material of Example 1 was replaced with an ethyl group, (+)-4-ethyl-4,5-dihydro-4-hydroxymethyl-2 -Phenyloxazole was obtained. The yield was 83%. When the optical purity was measured, it was 97% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 14 An experiment was conducted in the same manner as in Example 1 except that the methyl group in the raw material of Example 1 was substituted with an allyl group. As a result, (+)-4-allyl-4,5-dihydro-4-hydroxymethyl- 2-phenyloxazole was obtained. The yield was 71%. When the optical purity was measured, it was 92% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 15 Production of Optically Active Oxazoline Compound (1) The experiment was performed at normal temperature and normal pressure. In 2 ml of tert-butanol, 0.05 mmol of Cu (OTf) 2 and 0.05 mmol of (R, R) -Ph-Box were mixed and stirred for 10 minutes. In this solution, 0.5 mmol of the following formula as a raw material
- Example 16 An experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was substituted with a methyl group. As a result, (+)-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (4-methylphenyl) oxazole was obtained. The yield was 89.3%. When the optical purity was measured, it was 94.2% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 17 An experiment was conducted in the same manner as in Example 15 except that the 3-position of the phenyl group in the raw material of Example 15 was substituted with a methyl group. As a result, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (3-methylphenyl) oxazol was obtained. The yield was 72.9%. When the optical purity was measured, it was 99.9% or more ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 18 An experiment was conducted in the same manner as in Example 15 except that the 2-position of the phenyl group in the raw material of Example 15 was substituted with a methyl group. As a result, (+)-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (2-methylphenyl) oxazol was obtained. The yield was 85.8%. When the optical purity was measured, it was 91.9% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 19 When an experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was replaced with a methoxy group, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-2 -(4-methoxylphenyl) -4-methyloxazole was obtained. The yield was 63.7%. When the optical purity was measured, it was 93.8% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 20 An experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was substituted with fluorine. As a result, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-2- (4-fluorophenyl) -4-methyloxazole was obtained. The yield was 90.8%. When the optical purity was measured, it was 93.9% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 21 An experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was replaced with chlorine. As a result, ( ⁇ )-2- (4-chlorophenyl) -4,5- Dihydro-4-hydroxymethyl-4-methyloxazole was obtained. The yield was 97.3%. When the optical purity was measured, it was 95.6% ee R form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 22 An experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was substituted with bromine. As a result, ( ⁇ )-2- (4-bromophenyl) -4,5- Dihydro-4-hydroxymethyl-4-methyloxazole was obtained. The yield was 98.1%. When the optical purity was measured, it was 91.6% ee R form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 23 An experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was substituted with iodine. As a result, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-2- (4-iodophenyl) -4-methyloxazole was obtained. The yield was 96.9%. When the optical purity was measured, it was 86.5% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 24 An experiment was conducted in the same manner as in Example 15 except that the 2-position of the phenyl group in the raw material of Example 15 was replaced with iodine. As a result, (+)-4,5-dihydro-4-hydroxymethyl-2- (2-iodophenyl) -4-methyloxazole was obtained. The yield was 87.0%. When the optical purity was measured, it was 96.9% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 25 An experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was substituted with a phenyl group. As a result, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-4 -Methyl-2- (4-phenylphenyl) oxazole was obtained. The yield was 91.1%. When the optical purity was measured, it was 90.6% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 26 When an experiment was conducted in the same manner as in Example 15 except that the 4-position of the phenyl group in the raw material of Example 15 was replaced with a nitro group, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-4- methyl-2- (4-nitrophenyl) oxazole was obtained. The yield was 91.4%. When the optical purity was measured, it was 93.1% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 27 When an experiment was conducted in the same manner as in Example 15 except that the phenyl group in the raw material of Example 15 was replaced with a naphthyl group, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-4-methyl- 2- (2-naphthyl) oxazole was obtained. The yield was 90.0%. When the optical purity was measured, it was 99.2% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 28 When an experiment was conducted in the same manner as in Example 15 except that the phenyl group in the raw material of Example 15 was replaced with a styryl group, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-4-methyl- 2-styryloxazole was obtained. The yield was 20.1%. When the optical purity was measured, it was 94.3% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 29 An experiment was conducted in the same manner as in Example 15 except that the phenyl group in the raw material of Example 15 was substituted with a 2-furyl group. As a result, (+)-4,5-dihydro-2- (2-furyl ) -4-hydroxymethyl-4-methyloxazole was obtained. The yield was 69.9%. When the optical purity was measured, it was 99.0% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 30 An experiment was conducted in the same manner as in Example 15 except that the phenyl group in the raw material of Example 15 was substituted with a 2-thienyl group. As a result, (+)-4,5-dihydro-4-hydroxymethyl-4- methyl-2- (2-thienyl) oxazole was obtained. The yield was 66.8%. When the optical purity was measured, it was 94.0% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 31 When an experiment was conducted in the same manner as in Example 15 except that the phenyl group in the raw material of Example 15 was replaced with a 2-pyrrolyl group, ( ⁇ )-4,5-dihydro-4-hydroxymethyl-4- methyl-2- (2-pyrrolyl) oxazole was obtained. The yield was 28.1%. When the optical purity was measured, it was 89.2% ee ( ⁇ )-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 32 An experiment was conducted in the same manner as in Example 15 except that the phenyl group in the raw material of Example 15 was substituted with a 3-furyl group. As a result, (+)-4,5-dihydro-2- (3-furyl ) -4-hydroxymethyl-4-methyloxazole was obtained. The yield was 74.0%. When the optical purity was measured, it was 99.3% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 33 An experiment was conducted in the same manner as in Example 15 except that the methyl group in the raw material in Example 15 was replaced with a hydrogen atom. As a result, (+)-4,5-dihydro-4-hydroxymethyl-2-phenyloxazole was obtained. Obtained. The yield was 32.2%. When the optical purity was measured, it was an R form of 88.9% ee. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 34 An experiment was conducted in the same manner as in Example 15 except that the methyl group in the raw material of Example 15 was replaced with an ethyl group. (+)-4-ethyl-4,5-dihydro-4-hydroxymethyl-2 -Phenyloxazole was obtained. The yield was 82.6%. When the optical purity was measured, it was 97.1% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 35 An experiment was conducted in the same manner as in Example 15 except that the methyl group in the raw material of Example 15 was substituted with an allyl group. As a result, (+)-4-allyl-4,5-dihydro-4-hydroxymethyl- 2-phenyloxazole was obtained. The yield was 70.8%. When the optical purity was measured, it was 92.3% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- Example 36 An experiment was conducted in the same manner as in Example 15 except that the methyl group in the raw material of Example 15 was substituted with a benzyl group. As a result, (+)-4-benzyl-4,5-dihydro-4-hydroxymethyl- 2-phenyloxazole was obtained. The yield was 62.3%. When the optical purity was measured, it was 90.6% ee (+)-form. The structure and physical property data of the optically active oxazoline compound obtained by the above reaction are shown.
- (+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-phenyloxazole yield 40%, optical purity 95% ee
- (-)-4,5-dihydro-2-methyl -4-Phenyl-4-tosyloxymethyloxazole yield 60%, optical purity 60% ee
- Example 38 Production of optically active oxazoline compound (2) The experiment was performed at normal temperature and normal pressure. In 2 ml of acetonitrile, 0.05 mmol of Cu (OTf) 2 and 0.05 mmol of (R, R) -Ph-Box were mixed and stirred for 10 minutes. In this solution, 0.5 mmol of the following formula as a raw material
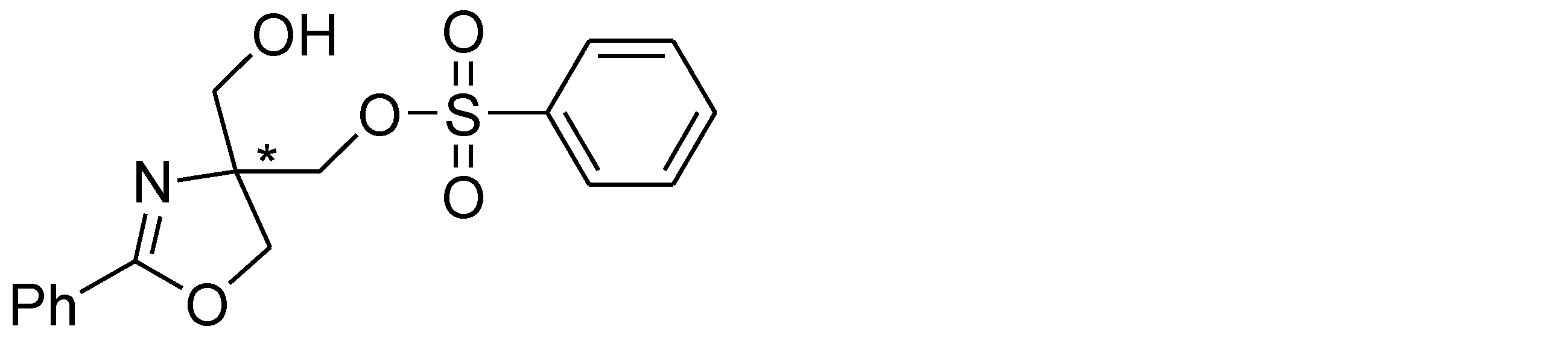
- Example 39 The experiment was conducted in the same manner as in Example 38 except that benzenesulfonyl chloride (1.25 mmol) was used instead of p-toluenesulfonyl chloride in the raw material of Example 38. The structure and physical property data of the product obtained by the above reaction are shown.
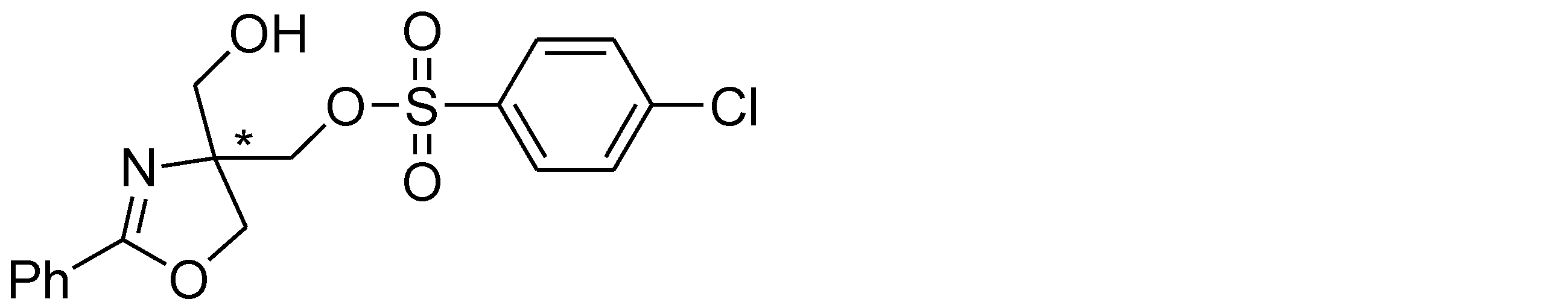
- Example 40 The experiment was conducted in the same manner as in Example 38 except that p-chlorobenzenesulfonyl chloride (1.25 mmol) was used instead of p-toluenesulfonyl chloride in the raw material of Example 38. The structure and physical property data of the product obtained by the above reaction are shown.
- Example 41 The experiment was conducted in the same manner as in Example 38, except that the phenyl group in the raw material of Example 38 was replaced with a 4-methylphenyl group. The structure and physical property data of the product obtained by the above reaction are shown.
- Example 42 The experiment was performed in the same manner as in Example 38, except that the phenyl group in the raw material of Example 38 was replaced with a 3-methylphenyl group. The structure and physical property data of the product obtained by the above reaction are shown.
- Example 43 The experiment was performed in the same manner as in Example 38, except that the phenyl group in the raw material of Example 38 was substituted with a 2-methylphenyl group. The structure and physical property data of the product obtained by the above reaction are shown.
- Example 44 The experiment was performed in the same manner as in Example 38 except that the phenyl group in the raw material of Example 38 was replaced with a 4-chlorophenyl group. The structure and physical property data of the product obtained by the above reaction are shown.
- Example 45 The experiment was conducted in the same manner as in Example 38, except that the phenyl group in the raw material of Example 38 was replaced with a 2-chlorophenyl group. The structure and physical property data of the product obtained by the above reaction are shown.
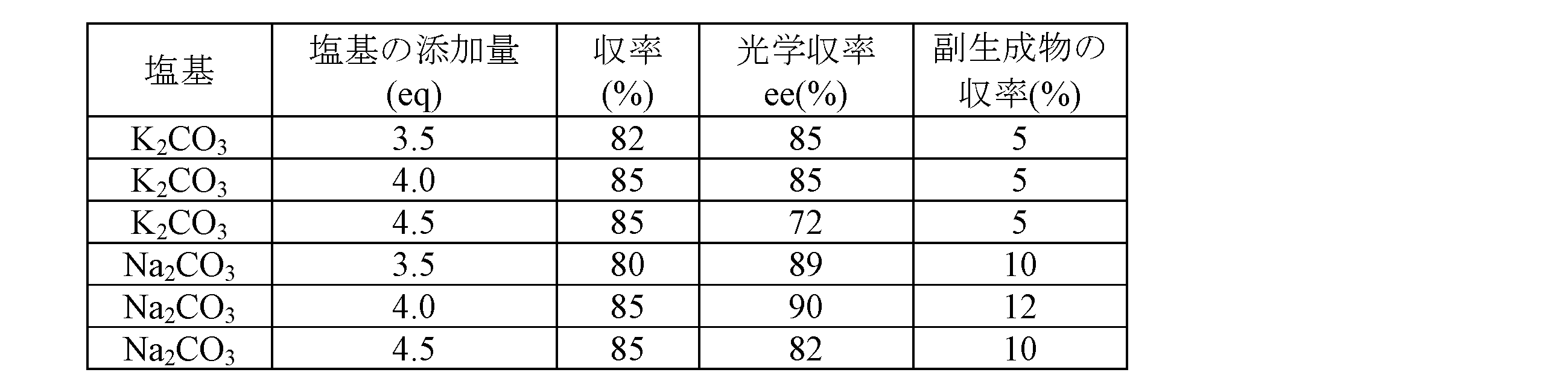
- optically active oxazoline compound 2 The amount of sulfonyl halide added is 2.5 mol per mol of the raw material compound, the base is replaced by potassium carbonate and sodium carbonate, and the type of solvent
- An optically active oxazoline compound was synthesized under the same conditions as in Example 38, except that was changed variously.
- the type of solvent, the yield of each reaction, and the optical purity are shown in Table 4 below.
- surface means the compound by which all the hydroxyl groups couple
- an optically active compound represented by the formula (3) or formula (4) or a salt thereof which is a synthetic intermediate of various useful compounds, can be obtained in a small number of steps using a compound having no optical activity as a raw material compound. Can be manufactured. Further, the optically active compound produced by the present invention is very useful as a synthetic intermediate for pharmaceuticals, agricultural chemicals, industrial reagents and the like. In addition, according to the present invention, an optically active compound represented by the formula (8) or a salt thereof is obtained with an excellent yield and an excellent optical property, using a compound having no optical activity represented by the formula (7) as a raw material compound. Can be produced in purity.
- optically active compound represented by the formula (8) or a salt thereof is produced in an excellent yield and an excellent optical purity using the compound having no optical activity represented by the formula (9) according to the present invention as a raw material. Can be done. Therefore, this invention can be used industrially usefully.
Abstract
Description
[1]式(1)
で表される化合物を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に閉環させることを特徴とする式(3)
で示される光学活性化合物又はその塩の製造方法、
[2]R1が炭素数6~20のアリール基、炭素数2~15で、異種原子として少なくとも1個の窒素原子、酸素原子、硫黄原子等の異種原子を含んでいる、5~8員の単環式ヘテロアリール基、多環式若しくは縮合環式のヘテロアリール基又はアルキル基、アルケニル基、アルコキシ基、ハロゲン原子、ニトロ基若しくはアリール基で置換されているフェニル基である[1]に記載の製造方法、
[3]R2が水素又は炭化水素基である[1]又は[2]に記載の製造方法、
[4]MmZnがCu(OTf)2である[1]~[3]のいずれかに記載の製造方法、
[5]さらに塩基の存在下に反応を行う[1]~[4]のいずれかに記載の製造方法、
[6]さらに有機溶媒の存在下に反応を行う[1]~[5]のいずれかに記載の製造方法、
[7]式(4)
R4は水素原子又は反応に関与しない基である。ただし、R3がフェニル基であって、R4が水素原子又はメチル基である場合を除く。*は不斉であることを表す。)
又は式(4’)
で表される化合物のラセミ体を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させ、式(3)
[9]式(7)
で表される化合物を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させることを特徴とする式(8)
で表される化合物を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させ、式(8)
以下に、一般式(3)及び(4)で表される化合物又はその塩の製造方法及びオキサゾリンのラセミ体を光学分割する方法を詳説する。
(原料化合物)
本発明で使用される原料化合物は、式(1)
で表される。
置換基としてのアルキニル基としては、直鎖状でも分岐状でもよい、例えば炭素数2~15、好ましくは炭素数2~10、より好ましくは炭素数2~6のアルキニル基が挙げられる。具体的には、上記R1としてのアルキニル基と同じである。
置換基としてのアルケニル基としては、直鎖状でも分岐状でもよい、例えば炭素数2~15、好ましくは炭素数2~10、より好ましくは炭素数2~6のアルケニル基が挙げられ、具体的には、上記R1としてのアルケニル基と同じである。
置換基としての脂肪族複素環基としては、例えば炭素数2~14で、異種原子として少なくとも1個、好ましくは1~3個の例えば窒素原子、酸素原子、硫黄原子等のヘテロ原子を含んでいる、5~8員、好ましくは5又は6員の単環の脂肪族複素環基、多環又は縮合環の脂肪族複素環基が挙げられる。具体的には、上記R1としての脂肪族複素環基と同じである。
置換基としてのシクロアルキル基としては、例えば炭素数3~14、好ましくは炭素数5~12、より好ましくは炭素数6~12のシクロアルキル基が挙げられ、具体的には上記R1としてのシクロアルキル基と同じである。
置換基としてのアリールオキシ基としては、例えば炭素数6~14のアリールオキシ基が挙げられ、具体的にはフェノキシ基、トリルオキシ基、キシリルオキシ基、ナフトキシ基、アントリルオキシ基等が挙げられる。
置換基としてのアラルキル基としては、上記アルキル基の少なくとも1個の水素原子が上記アリール基で置換された基が挙げられる。具体的には、上記R1としてのアラルキル基と同じである。
置換基としての芳香族複素環基としては、例えば炭素数2~15で、異種原子として少なくとも1個、好ましくは1~3個の窒素原子、酸素原子、硫黄原子等の異種原子を含んでいる、5~8員、好ましくは5又は6員の単環式ヘテロアリール基、多環式又は縮合環式のヘテロアリール基が挙げられる。具体的には、上記R1としての芳香族複素環基と同じである。
置換基としてのアラルキルオキシ基としては、例えば炭素数7~12のアラルキルオキシ基が挙げられ、具体的には、ベンジルオキシ基、1-フェニルエトキシ基、2-フェニルエトキシ基、1-フェニルプロポキシ基、2-フェニルプロポキシ基、3-フェニルプロポキシ基、1-フェニルブトキシ基、3-フェニルブトキシ基、4-フェニルブトキシ基、1-フェニルペンチルオキシ基、2-フェニルペンチルオキシ基、3-フェニルペンチルオキシ基、4-フェニルペンチルオキシ基、5-フェニルペンチルオキシ基、1-フェニルヘキシルオキシ基、2-フェニルヘキシルオキシ基、3-フェニルヘキシルオキシ基、4-フェニルヘキシルオキシ基、5-フェニルヘキシルオキシ基、6-フェニルヘキシルオキシ基等が挙げられる。
置換基としてのヘテロアリールオキシ基としては、例えば、異種原子として少なくとも1個、好ましくは1~3個の窒素原子、酸素原子、硫黄原子等の異種原子を含んでいる、炭素数2~14のヘテロアリールオキシ基が挙げられ、具体的には、2-ピリジルオキシ基、2-ピラジルオキシ基、2-ピリミジルオキシ基、2-キノリルオキシ基等が挙げられる。
ヘテロアリールチオ基としては、例えば異種原子として少なくとも1個、好ましくは1~3個の窒素原子、酸素原子、硫黄原子等の異種原子を含んでいる、炭素数2~14のヘテロアリールチオ基が挙げられ、具体的には、例えば2-ピリジルチオ基、4-ピリジルチオ基、2-ベンズイミダゾリルチオ基、2-ベンズオキサゾリルチオ基、2-ベンズチアゾリルチオ基等が挙げられる。
アルキル基で置換されたアミノ基、即ちアルキル基置換アミノ基としては、例えばN-メチルアミノ基、N,N-ジメチルアミノ基、N,N-ジエチルアミノ基、N,N-ジイソプロピルアミノ基、N-シクロヘキシルアミノ基等のモノ又はジアルキルアミノ基が挙げられる。
アリール基で置換されたアミノ基、即ちアリール基置換アミノ基としては、例えばN-フェニルアミノ基、N,N-ジフェニルアミノ基、N,N-ジトリルアミノ基、N-ナフチルアミノ基、N-ナフチル-N-フェニルアミノ基等のモノ又はジアリールアミノ基が挙げられる。
アラルキル基で置換されたアミノ基、即ちアラルキル基置換アミノ基としては、例えばN-ベンジルアミノ基、N,N-ジベンジルアミノ基等のモノ又はジアラルキルアミノ基が挙げられる。
上記閉環反応において、式(2)
上記式(2)中、Zで表されるMの対アニオンとしては特に限定されず、例えばフッ素、塩素、臭素、ヨウ素等のハロゲンイオン、トリフルオロメチルスルホニルイオン(-OTf)、四フッ化ホウ素イオン(-BF4)、六フッ化リンイオン(-PF6)、六フッ化アンチモンイオン(-SbF6)、ビス(トリフルオロメチルスルホニル)イミドイオン(-NTf2)、酢酸イオン(-OAc)、トリフルオロ酢酸イオン(-OCOCF3)等が挙げられる。中でもZがフッ化物イオン、塩化物イオン、-OTf、-BF4又は-PF6であることが好ましく、-OTfであることが特に好ましい。
なお上記ルイス酸は市販品を用いてもよいし、公知の方法により製造してもよい。
上記ルイス酸の使用量としては、式(1)で示される化合物1モルに対して0.001~1.00モルであればよく、0.005モル~0.50モルであることがより好ましく、0.01~0.20モルであることが特に好ましい。
ここで、ビスオキサゾリン化合物をBoxという。また、Box中のオキサゾリン環のそれぞれにフェニル基が結合したもの(以下Ph-Boxという。)、イソプロピル基が結合したもの(以下i-Pr-Boxという。)、t-Buが結合したもの(以下t-Bu-Boxという。)及びベンジル基が結合したもの等が挙げられる。
上記光学活性オキサゾリン化合物としては、オキサゾリン環のそれぞれに同一の立体配置を持つ置換基が結合しているものが挙げられ、例えば、下記式(10)
上記光学活性ビスオキサゾリン化合物は、市販のものを用いることもできるし、自体公知の方法で製造することもできる。当該製造方法としては、例えば、J.Am.Chem.Soc.,113,728-729(1991)記載の方法等が挙げられる。
上記置換されていてもよいフェニル基は一の水素原子が置換基で置換されていてもよく、二以上の水素原子が置換基で置換されていてもよいが、一の水素原子が置換基で置換されていることが好ましい。上記置換されていてもよいフェニル基の置換部位としては特に限定されず、オルト位、メタ位、パラ位のいずれが置換されていてもよいが、パラ位が置換されていることが好ましい。
上記置換されていてもよいアルキル基としては、直鎖状でも分岐状でもよい、例えば炭素数1~20の直鎖状又は分岐状のアルキル基又はその任意の水素原子を置換基で置換したものが挙げられる。直鎖状又は分岐状のアルキル基の具体例としては、例えば、メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基、sec-ブチル基、イソブチル基、tert-ブチル基、n-ペンチル基、ネオペンチル基、tert-ペンチル基、イソペンチル基、2-メチルブチル基、1-エチルプロピル基、ヘキシル基、イソヘキシル基、4-メチルペンチル基、3-メチルペンチル基、2-メチルペンチル基、1-メチルペンチル基、3,3-ジメチルブチル基、2,2-ジメチルブチル基、1,1-ジメチルブチル基、1,2-ジメチルブチル基、1,3-ジメチルブチル基、2,3-ジメチルブチル基、1-エチルブチル基、2-エチルブチル基、ヘプチル基、オクチル基、ノニル基、デシル基、セチル基、ステアリル基などの直鎖状又は分岐状のアルキル基などが挙げられる。上記置換されていてもよいアルキル基は、炭素数が1~4のものが好ましく、メチル基又はエチル基であることがより好ましい。
上記置換されていてもよいアルキル基は、一又は複数の水素原子が置換基で置換されていてもよい。上記置換されていてもよいアルキル基の置換部位としては特に限定されない。ここで置換基としては、上記(1)において置換基として例示された基と同じ基を使用することができる。
上記スルホニルハライドにおけるハロゲン原子としては特に限定されないが、例えばフッ素原子、塩素原子、臭素原子、ヨウ素原子等が挙げられ、中でも塩素原子又は臭素原子が好ましく、塩素原子が特に好ましい。
上記スルホニルハライドの使用量としては、式(1)で示される原料物質1モルに対して、0.1~10モルであればよく、0.5~5.0モルであることが好ましく、0.8~4.0モルであることが特に好ましい。
上記触媒は均一系触媒であっても不均一系触媒であってもよい。上記触媒は公知方法に従って製造してもよいし、市販のものを用いてもよい。また、上記の触媒に加え、公知の他の触媒を使用してもよい。
上記反応に用いる溶媒としては特に限定されず、有機溶媒、無機溶媒のいずれも用いることができる。
有機溶媒としては、ペンタン、ヘキサン、ヘプタン、オクタン、シクロヘキサン等の脂肪族炭化水素類;ベンゼン、トルエン、キシレン等の芳香族炭化水素類;ジクロロメタン、1,2-ジクロロエタン、クロロホルム、四塩化炭素、o-ジクロロベンゼン等のハロゲン化炭化水素類;メタノール、エタノール、イソプロピルアルコール、t-ブチルアルコール、t-アミルアルコール等のアルコール類;ジメチルエーテル、エチルメチルエーテル、ジエチルエーテル、ジイソプロピルエーテル、ジグリム、tert-ブチルメチルエーテル、ジメトキシエタン、エチレングリコールジエチルエーテル、テトラヒドロフラン、1,4-ジオキサン等のエーテル類;N,N-ジメチルホルムアミド、N,N-ジメチルアセトアミド、N-メチルピロリドン等のアミド類;ジメチルスルホキシド等のスルホキシド類;アセトニトリル、プロピオニトリル、ベンゾニトリル等のニトリル類、アセトン、メチルエチルケトン、メチルイソブチルケトン等のケトン類、酢酸メチル、酢酸エチル等のエステル化合物等が挙げられる。
無機溶媒としては、塩酸、硫酸、硝酸、炭酸、リン酸等の酸性水溶液;水酸化ナトリウム、水酸化マグネシウム、水酸化カルシウム、水酸化カリウム、炭酸水素ナトリウム等の塩基性水溶液;純水、食塩水等の中性水溶液等が挙げられる。
上記溶媒は単独で用いてもよいし、2種又はそれ以上を組み合わせて用いてもよい。溶媒の使用量は特に限定されないが、原料物質に対して過剰量であればよく、例えば式(1)で示される原料物質1重量部に対して、0.1~100重量部とすることができる。
反応に使用されうる塩基として、水酸化ナトリウム、水酸化カリウム、炭酸カリウム、炭酸ナトリウム、炭酸セシウム、アンモニア等の無機塩基、トリエチルアミン、ジイソプロピルエチルアミン、ピリジン等の有機塩基などが好適な例として挙げられる。塩基の使用量としては、式(1)の原料物質1モルに対して、0.1~10モルであればよく、0.5~8.0モルであることがより好ましく、0.8~5.0モルであることが特に好ましい。
本発明の反応は、上記式(1)の化合物を
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に閉環させることを特徴とする式(3)
さらに、当該反応により式(1)で表される非環状化合物が閉環し、式(3)若しくは式(4)で示される光学活性を有するオキサゾリン化合物又はその塩が生成するため、様々な有用な化合物の製造のために、本発明を用いることが可能となりうる。
上記反応では、使用される上記不斉配位子の立体選択性に応じて生成するオキサゾリン化合物(式(3))の光学活性が決まる。例えば、上記不斉配位子として(R,R)-Ph-Boxを使用したときは、(R)体のオキサゾリン化合物が(S)体のオキサゾリン化合物と比較して優先的に生成する。ここで、(S)体のオキサゾリン化合物もある程度の量で一旦生成すると考えられる。ところが、後述するオキサゾリンの光学分割反応に示されるように本発明の反応条件下では、(S)体の水酸基の水素原子が、使用される上記スルホニルハライドに対応する基、すなわち上記スルホニルハライドからハロゲン原子を除いたスルホニル基に変換されるため、例えば上記スルホニルハライドとして塩化パラトルエンスルホニルを使用した場合では(S)体の水酸基の水素原子はパラトルエンスルホニル基(-Ts)に変換されるため、水酸基を有する(S)体のオキサゾリン化合物がさらに減少し、(R)体の立体選択性が高くなると考えられる。
なお、スルホニルオキシ基が導入されたオキサゾリン化合物は、その化合物名称の表記の上では(R)体となるが、その不斉炭素の立体配置は水酸基を有する(S)体のオキサゾリン化合物と同じである。
上記反応を行う反応温度としては特に限定されず、例えば-20℃~80℃の条件下で行うことが可能であり、5℃~40℃の条件下で行うのが好ましい。上記反応を行う圧力としては特に限定されず、減圧下、大気圧下又は加圧下のいずれであってもよい。反応を行う圧力としては、例えば0.1~20Paで行うことが可能であり、0.7~1.2Paで行うことが好ましい。
すなわち本反応は常温常圧下において反応を進行することができるため、製造設備を簡略化することができ、もって製造コストの削減に寄与することが可能となる。
上記反応を行う反応時間は特に限定されず、例えば5分~72時間で行うことができるが、10分~48時間で行うことが好ましい。
上記反応は上記原料を、好ましくは撹拌下に混合することにより進行させることができる。
上記反応により生成する目的化合物は、式(3)
で表される。
R3で表される、炭素数2~15で、異種原子として少なくとも1個の窒素原子、酸素原子、硫黄原子等の異種原子を含んでいる、5~8員の単環式ヘテロアリール基、多環式若しくは縮合環式のヘテロアリール基の具体例としては、例えばフリル基、チエニル基、ピロリル基、ピリジル基、ピリミジル基、ピラジル基、ピリダジル基、ピラゾリル基、イミダゾリル基、オキサゾリル基、チアゾリル基、ベンゾフリル基、ベンゾチエニル基、キノリル基、イソキノリル基、キノキサリル基、フタラジル基、キナゾリル基、ナフチリジル基、シンノリル基、ベンゾイミダゾリル基、ベンゾオキサゾリル基、ベンゾチアゾリル基等が挙げられる。
置換基としてのアルキル基、置換基としてのアルケニル基、置換基としてのアルコキシ基、置換基としてのハロゲン原子、置換基としてのニトロ基若しくは置換基としてのアリール基の具体例としては、上記R1で挙げたものと同じものが挙げられる。
また、式(4)で表される化合物の具体例としては、上記式(3)で表される化合物よりR1がフェニル基であって、R2が水素原子又はメチル基である場合を除いたものが挙げられる。
上記反応によって生成する式(3)又は(4)で表される目的化合物は、単離若しくは精製されてもよいし、単離若しくは精製されることなく合成中間体として次の反応に供してもよい。単離精製のための手段は、公知手段により行われる。そのような手段としては、蒸留、濃縮、抽出、結晶化、クロマトグラフィー、濾過、透析、遠心分離等公知の手段が便宜に採用される。
式(3)又は(4)で表される目的化合物は、医療用、工業用、農業用等の種々の目的に用いられる。具体的な用途として、有用化合物の合成に用いる合成中間体が挙げられ、例えば非天然型アミノ酸合成のビルディングブロックとして用いることができる。
上記目的化合物は、単独で用いることもできるし、複数の化合物を混合した状態で用いることも可能である。また、上記目的化合物は、固体、液体及び気体のいずれの状態で用いてもよい。
上記反応により、式(6)
式(6)中のR5で表されるアルキル基としては、直鎖状でも、分岐状でもよい、例えば炭素数1~20の直鎖状又は分岐状のアルキル基が挙げられ、具体的には上記R1としてのアルキル基と同じものが挙げられる。
式(6)中のR5で表されるアルキル基又はフェニル基の置換基としては、例えばアルキル基、アルキニル基、アルケニル基、アリール基、アルコキシ基、アルキレンジオキシ基、アリールオキシ基、アラルキルオキシ基、ヘテロアリールオキシ基、アルキルチオ基、シクロアルキル基、脂肪族複素環基、アリールチオ基、アラルキルチオ基、ヘテロアリールチオ基、アミノ基、置換アミノ基、シアノ基、ヒドロキシル基、オキソ基、ニトロ基、メルカプト基又はハロゲン原子等が挙げられ、具体的には上記R1の置換基と同じものが挙げられる。
式(4’)中のR5は、式(6)と同じ意味である。
式(6)又は式(4’)で表される化合物は、単独で用いることもできるし、複数の化合物を混合した状態で用いることも可能である。また、上記目的化合物は、固体、液体及び気体のいずれの状態で用いてもよい。
(原料化合物)
本発明で使用される原料化合物は、式(5)
で表される化合物のラセミ体である。
式(5)で表される化合物におけるR1及びR2は、式(1)と同じ意味である。原料化合物はラセミ体(化学構造が鏡像の関係にある一対の光学異性体を等量混合した物質をいう。)であるため、光学活性を有さない。
上記光学分割において、式(2)
で表されるルイス酸を用いる。上記式(2)中のMで表される金属イオン、Zで表されるMの対アニオン及びルイス酸としては、上記光学活性なオキサゾリン又はその塩の製造方法で用いたものと同じものが挙げられる。
上記ルイス酸の使用量としては、式(5)で示される化合物1モルに対して0.001~1.00モルであればよく、0.005モル~0.50モルであることがより好ましく、0.01~0.20モルであることが特に好ましい。
上記不斉配位子の使用量としては、式(5)で示される化合物1モルに対して0.001~1.00モルであればよく、0.005モル~0.50モルであることがより好ましく、0.01~0.20モルであることが特に好ましい。
上記スルホニルハライドの使用量としては、式(5)で示される原料物質1モルに対して、0.1~10モルであればよく、0.3~5.0モルであることがより好ましく、0.5~3.0モルであることが特に好ましい。
上記反応に用いる溶媒としては特に限定されず、有機溶媒、無機溶媒のいずれも用いることができるが、上記光学活性なオキサゾリン又はその塩の製造方法で用いたものと同じものが挙げられる。
溶媒の使用量は特に限定されないが、原料物質に対して過剰量であればよく、例えば式(5)で示される原料物質1重量部に対して、0.1~100重量部とすることができる。
反応に使用されうる塩基としては、上記光学活性なオキサゾリン又はその塩の製造方法で用いたものと同じものが挙げられる。
塩基の使用量としては、式(5)で示される原料物質1モルに対して、0.1~10モルであればよく、0.3~8.0モルであることがより好ましく、0.5~5.0モルとであることが好ましい。
本発明は、上記式(5)の化合物のラセミ体を
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させ、式(3)
で表される光学活性化合物又はその塩の(R)体又は(S)体に光学分割するものである。
上記反応では、使用される上記不斉配位子の立体選択性に応じて、生成するオキサゾリン化合物(式(3))の光学活性が決まる。例えば、上記不斉配位子として(R,R)-Ph-Boxを使用したときは、(R)体の式(3)で表されるオキサゾリン化合物が生成する。ここで、(S)体の式(3)で表されるオキサゾリン化合物は、その水酸基の水素原子が、使用される上記スルホニルハライドに対応する基、すなわち上記スルホニルハライドからハロゲン原子を除いたスルホニル基に変換されるため、(S)体の式(3)で表されるオキサゾリン化合物は減少し、相対的に(R)体の立体選択性が高くなると考えられる。
なお、スルホニルオキシ基が導入された式(6)のオキサゾリン化合物は、その化合物名称の表記の上では(R)体となるが、その不斉炭素の立体配置は(S)体の式(3)で表されるオキサゾリン化合物と同じである。
本発明は、式(7)
で表される化合物を
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させ、式(8)
本発明において、原料化合物として上記式(7)で表す化合物を用いる。式中、R1としては、光学活性なオキサゾリン又はその塩の製造方法(1)の一般式(1)について説明したものと同じものが挙げられる。
溶媒は、光学活性なオキサゾリン又はその塩の製造方法(1)と同様のもの等が挙げられるが、MeCN(アセトニトリル)であることが特に好ましい。
塩基は、光学活性なオキサゾリン又はその塩の製造方法(1)と同様のもの等が挙げられるが、K2CO3又はNa2CO3であることが特に好ましい。また塩基の使用量としては、式(7)で示される原料物質1モルに対して、0.2~20モルであればよく、1.0~16.0モルであることがより好ましく、1.6~10.0モルであることが特に好ましい。
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドは、光学活性なオキサゾリン又はその塩の製造方法(1)と同様のもの等が挙げられるが、中でもフェニルスルホニルハライド又は4位がアルキル基又はハロゲン原子で置換されたフェニルスルホニルハライドが特に好ましい。またスルホニルハライドの使用量は、式(7)で示される原料物質1モルに対して、0.2~20モルであればよく、1.0~10.0モルであることが好ましく、1.6~8.0モルであることが特に好ましい。
上記反応を行う反応時間は特に限定されず、例えば5分~96時間で行うことができるが、10分~72時間で行うことが好ましい。
本反応において、生成物として上記式(8)で表される光学活性を有する化合物又はその塩が得られる。式中、R1としては、式(7)と同じものが挙げられ、R5としては、光学活性なオキサゾリン又はその塩の製造方法(1)の一般式(6)について説明したものと同じものが挙げられる。
本反応において、副生成物として式(15)
本発明により、式(7)で表される安価で入手容易な化合物を原料として、式(8)で表される光学活性を有する化合物を製造することができる。
具体的には、原料化合物のひとつの水酸基のスルホニル化に続く閉環反応によりオキサゾリン環が形成され、下記式(9)
その後、上記中間生成物の一方の水酸基の水素原子が、使用するスルホニルハライドに由来する基(すなわち、上記スルホニルハライドからハロゲン原子を除いたスルホニル基)で立体選択的に置換される。これにより上記式(8)で表される光学活性オキサゾリン化合物が生成すると考えられる。
本発明は、式(9)
で表される化合物を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させ、式(8)
で表される光学活性化合物又はその塩の(R)体又は(S)体を生成する製造方法である。
本発明において、原料化合物として上記式(9)で表す化合物を用いる。式中、R1としては、光学活性なオキサゾリン又はその塩の製造方法(1)の一般式(1)について説明したものと同じものが挙げられる。
ルイス酸、不斉配位子の例示、製造方法、添加量等は、光学活性なオキサゾリン又はその塩の製造方法(1)と同様である。
溶媒の種類、塩基の種類、置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの種類、反応時間は、光学活性なオキサゾリン又はその塩の製造方法(2)と同様である。
本反応において、生成物として上記式(8)で表される光学活性を有する化合物又はその塩が得られる。式中、R1としては、式(7)と同じものが挙げられ、R5としては、光学活性なオキサゾリン又はその塩の製造方法(1)の一般式(6)について説明したものと同じものが挙げられる。
本反応において、副生成物として式(15)
本発明により、式(9)で表される化合物を原料として、式(8)で表される光学活性を有する化合物を製造することができる。
融点は、Yanaco社製の「MICRO MELTING POINT APPARATUS」用いて測定した。測定値はすべて未補正である。
赤外吸収スペクトル(IR) は、島津製作所社製の「FTIR-8400S」を用いて測定した。
核磁気共鳴スペクトル(NMR) は、「VARIAN Gemini-300(300MHz)」を用いて測定した。内部標準物質としてTMS(Tetramethylsilane)を用い、測定溶媒としてCDCl3を用いて室温下で測定した。測定値はすべてδ(ppm)で示した。
旋光度は、日本分光社製の「DIP-1000」を用いて測定した。
高速液体クロマトグラフィー(HPLC)は、送液ポンプとして島津製作所社製の「LC-20AT」を、UV検出器として島津製作所社製の「SPD-20A」、記録装置として「C-R8A CHROMATOPAC」を、光学活性カラムとしてダイセル化学工業社製の「CHIRALPAC」シリーズを用いて測定した。HPLCにおける保持時間の差から、光学純度を決定した。
質量分析(MS)は、日本電子社製の「JMS-700N」を用いて測定した。
カラムクロマトグラフィーは、ナカライテスク社製の「Silica Gel 60,spherical,neutrality」を用いて測定した。
反応に用いた溶媒及び試薬は特記ない限り、市販品を使用した。また、抽出溶媒の乾燥には無水硫酸ナトリウムを用いた。
常温、常圧にて実験を行った。tert-ブタノール2ml中で、0.1mmolのCu(OTf)2及び0.1mmolの(R,R)-Ph-Boxを混合し、10分間撹拌した。この溶液に、原料として0.5mmolの下記式
これに1.5mmolの炭酸カリウムを加え、最後に塩化p-トルエンスルホニル(1mmol)を加え、12時間撹拌した。反応後、水を加え酢酸エチルで3回抽出した。有機層を無水硫酸ナトリウムで乾燥させて濾過した後、溶媒を減圧留去し、残渣をカラムクロマトグラフィー(展開溶液:n-ヘキサン:酢酸エチル=3:1(V/V)の混合溶液)で精製し、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-phenyloxazoleを得た。収率は85%であった。光学純度を測定したところ、95%eeのR体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 16 = +15.2 (c 0.10, CHCl3);
IR(neat): ν = 3171 (br), 2972, 2895, 2862, 1641, 1076 cm-1;
1H-NMR(CDCl3): δ = 1.32 (s, 3H), 3.49 (d, J = 11.4 Hz, 1H), 3.78 (d, J = 11.1 Hz, 1H), 4.09 (d, J = 8.1 Hz, 1H), 4.47 (d, J = 8.4 Hz, 1H), 7.33-7.41 (m, 2H), 7.43-7.50 (m, 1H), 7.84-7.91 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.1, 131.2, 128.2, 128.0, 127.1, 74.5, 71.9, 67.8, 23.7;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 10 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 15.3 min, 26.9 min, 95% ee;
HRMS(M+H, FAB): m/z calcd for C11H14NO2: 191.1024, found: 191.1038.
実施例1の原料中のフェニル基の4位をメチル基で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(4-methylphenyl)oxazoleを得た。収率は89%であった。光学純度を測定したところ、94%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 23 = +1.7 (c 0.50, CHCl3);
IR(neat): ν = 3192 (br), 2963, 2922, 2851, 1645, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.39 (s, 3H), 3.48 (d, J = 11.1 Hz, 1H), 3.76 (d, J = 11.4 Hz, 1H), 4.08 (d, J = 7.8 Hz, 1H), 4.44 (d, J = 8.1 Hz, 1H), 7.19 (d, J = 7.8 Hz, 2H), 7.79 (d, J = 8.1 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.3, 141.6, 128.8, 128.2, 124.4, 74.5, 71.8, 68.0, 23.7, 21.5;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 47.9 min, 62.2 min, 94% ee;
HRMS(M+H, FAB): m/z calcd for C12H16NO2: 206.1181, found: 206.1192.
実施例1の原料中のフェニル基の3位をメチル基で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(3-methylphenyl)oxazolを得た。収率は73%であった。光学純度を測定したところ、99%以上eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 16 = -5.6 (c 1.00, CHCl3);
IR(neat): ν = 3223 (br), 2967, 2924, 2862, 1645, 1072 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.37 (s, 3H), 3.49 (d, J = 11.1 Hz, 1H), 3.77 (d, J = 11.4 Hz, 1H), 4.09 (d, J = 8.1 Hz, 1H), 4.46 (d, J = 8.4 Hz, 1H), 7.20-7.34 (m, 2H), 7.66-7.78 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.2, 137.6, 131.9, 128.7, 127.9, 126.9, 125.2, 74.4, 71.9, 67.8, 23.7, 21.1;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 33.1 min, 73.4 min, >99% ee;
HRMS(M, EI): m/z calcd for C12H15NO2: 205.1098, found: 205.1100.
実施例1の原料中のフェニル基の2位をメチル基で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(2-methylphenyl)oxazolを得た。収率は86%であった。光学純度を測定したところ、92%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 21 = +14.2 (c 1.00, CHCl3);
IR(neat): ν = 3304 (br), 2967, 2926, 2870, 1639, 1045 cm-1;
1H-NMR(CDCl3): δ = 1.35 (s, 3H), 1.88 (br, 1H), 2.56 (s, 3H), 3.49 (d, J = 10.8 Hz, 1H), 3.75 (d, J = 11.1 Hz, 1H), 4.06 (d, J = 8.1 Hz, 1H), 4.40 (d, J = 8.1 Hz, 1H), 7.18-7.29 (m, 2H), 7.31-7.38 (m, 1H), 7.76 (d, J = 7.5 Hz, 1H);
13C-NMR(400MHz, CDCl3): δ = 165.1, 138.5, 131.1, 130.6, 129.8, 127.1, 125.5, 74.3, 72.1, 68.1, 23.7, 21.4;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 31.8 min, 40.5 min, 92% ee;
HRMS(M+H, EI): m/z calcd for C12H16NO2: 206.1181, found: 206.1171.
実施例1の原料中のフェニル基の4位をメトキシ基で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-2-(4-methoxylphenyl)-4-methyloxazoleを得た。収率は64%であった。光学純度を測定したところ、94%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 21 = -20.2 (c 1.00, CHCl3);
IR(neat): ν = 3208 (br), 2967, 2941, 2845, 1637, 1252, 1080 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 3.47 (d, J = 11.1 Hz, 1H), 3.75 (d, J = 11.4 Hz, 1H), 3.85 (s, 3H), 4.08 (d, J = 8.1 Hz, 1H), 4.44 (d, J = 8.1 Hz, 1H), 6.86-6.94 (m, 2H), 7.84-7.92 (m, 2H);
13C-NMR(400MHz, CDCl3) : δ = 164.0, 162.0, 130.0, 119.6, 113.4, 74.5, 71.7, 67.9, 55.3, 23.7;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 21.0 min, 37.5 min, 94% ee;
HRMS(M+H, FAB): m/z calcd for C12H16NO3: 222.1130, found: 222.1130.
実施例1の原料中のフェニル基の4位をフッ素で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-2-(4-fluorophenyl)-4-methyloxazoleを得た。収率は91%であった。光学純度を測定したところ、94%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 19 = -8.8 (c 1.00, CHCl3);
IR(neat): ν = 3184 (br), 2972, 2899, 2855, 1647, 1510, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.27 (br, 1H), 3.48 (d, J = 11.7 Hz, 1H), 3.76 (d, J = 11.4 Hz, 1H), 4.08 (d, J = 8.1 Hz, 1H), 4.46 (d, J = 7.8 Hz, 1H), 7.04-7.12 (m, 2H), 7.88-7.95 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 165.2, 163.2, 163.1, 130.4, 130.3, 123.2, 115.1, 114.9, 74.5, 72.0, 67.5, 23.6;
HPLC: OD-H column(4.6 mmφ, 500 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 62.9 min, 71.9 min, 94% ee;
HRMS(M+H, FAB): m/z calcd for C11H13FNO2: 210.0930, found: 210.0938.
実施例1の原料中のフェニル基の4位を塩素で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(-)-2-(4-chlorophenyl)-4,5-dihydro-4-hydroxymethyl-4-methyloxazoleを得た。収率は97%であった。光学純度を測定したところ、96%eeのR体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 21 = -32.2 (c 1.00, CHCl3);
IR(neat): ν = 3198 (br), 2967, 2897, 2860, 1651, 1492, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.27 (br, 1H), 3.49 (t, J = 7.8 Hz, 1H), 3.76 (d, J = 12.3 Hz, 1H), 4.09 (dd, J = 0.9, 8.1 Hz, 1H), 4.47 (d, J = 7.8 Hz, 1H), 7.35 (d, J = 8.7 Hz, 2H), 7.82 (d, J = 8.4 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 163.2, 137.5, 129.5, 128.3, 125.5, 74.6, 72.1, 67.7, 23.7;
HPLC: AY-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 20.8 min, 38.8 min, 96% ee;
HRMS(M+H, FAB): m/z calcd for C11H13ClNO2: 226.0635, found: 226.0627.
実施例1の原料中のフェニル基の4位を臭素で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(-)-4-(4-bromophenyl)-4,5-dihydro-2-hydroxymethyl-2-methyloxazoleを得た。収率は98%であった。光学純度を測定したところ、92%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 19 = -35.5 (c 1.00, CHCl3);
IR(neat): ν = 3219 (br), 2965, 2897, 2862, 1649, 1072 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.16 (br, 1H), 3.44-3.54 (m, 1H), 3.71-3.82 (m, 1H), 4.08 (dd, J = 0.6, 8.3 Hz, 1H), 4.54 (d, J = 8.1 Hz, 1H), 7.63 (d, J = 8.7 Hz, 2H), 7.76 (d, J = 8.7 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 163.3, 131.3, 129.7, 126.0, 125.0, 74.6, 72.1, 67.7, 23.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 200 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 93.4 min, 107.8 min, 92% ee;
HRMS(M+H, FAB): m/z calcd for C11H13BrNO2: 270.0130, found: 270.0153.
実施例1の原料中のフェニル基の4位をヨウ素で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-2-(4-iodophenyl)-4-methyloxazoleを得た。収率は97%であった。光学純度を測定したところ、87%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 18 = -41.4 (c 1.00, CHCl3);
IR(neat): ν = 3221 (br), 2972, 2932, 2854, 1636, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.32 (s, 3H), 2.17 (br, 1H), 3.42-3.56 (m, 1H), 3.79 (d, J = 11.1 Hz, 1H), 4.13 (dd, J = 0.6, 8.3 Hz, 1H), 4.49 (d, J = 8.1 Hz, 1H), 7.57-7.63 (m, 2H), 7.70-7.76 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 163.5, 137.3, 129.7, 126.5, 98.5, 74.6, 72.1, 67.7, 23.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 200 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 109.0 min, 135.1 min, 87% ee;
HRMS(M+H, EI): m/z calcd for C11H13INO2: 317.9991, found: 317.9986.
実施例1の原料中のフェニル基の4位をフェニル基で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(4-phenylphenyl)oxazoleを得た。収率は91%であった。光学純度を測定したところ、91%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 20 = -64.6 (c 1.00, CHCl3);
IR(neat): ν = 3221 (br), 2972, 2932, 2907, 1638, 1082 cm-1;
1H-NMR(CDCl3): δ = 1.31 (s, 3H), 2.32 (br, 1H), 3.48 (d, J = 11.4 Hz, 1H), 3.76 (d, J = 11.1 Hz, 1H), 4.13 (dd, J = 0.6, 8.3 Hz, 1H), 4.46 (d, J = 8.1 Hz, 1H), 7.21-7.28 (m, 2H), 7.34-7.50 (m, 2H), 7.62 (d, J = 7.8 Hz, 3H), 7.94-8.04 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.0, 143.9, 140.2, 128.8, 128.7, 127.9, 127.1, 126.7, 126.0, 74.5, 72.0, 67.9, 23.7;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 54.2 min, 72.1 min, 91% ee;
HRMS(M, EI): m/z calcd for C17H17NO2: 267.1259, found: 267.1258.
実施例1の原料中のフェニル基の4位をニトロ基で置換したものを用いる以外は実施例1と同様に実験を行ったところ(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(4-nitrophenyl)oxazolを得た。収率は91%であった。光学純度を測定したところ、93%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 22 = -9.4 (c 1.00, CHCl3); IR(neat): ν = 3225 (br), 2970, 2933, 2857, 1524, 1078 cm-1; 1H-NMR(CDCl3): δ = 1.36 (s, 3H), 1.60 (br, 1H), 3.48-3.57 (m, 1H), 3.83 (d, J = 12.0 Hz, 1H), 4.17 (dd, J = 0.9, 8.1 Hz, 1H), 4.48-4.56 (m, 1H), 7.94-8.30 (m, 4H);
13C-NMR(400MHz, CDCl3): δ = 162.2, 149.4, 132.9, 129.2, 123.3, 75.1, 72.6, 67.8, 23.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 52.6 min, 60.5 min, 93% ee;
HRMS(M, EI): m/z calcd for C11H12N2O4: 236.0797, found: 236.0798.
実施例1の原料中のメチル基を水素原子で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-2-phenyloxazoleを得た。収率は32%であった。光学純度を測定したところ、89%eeのR体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 22 =+64.2(c 0.77, CHCl3); IR(neat): ν = 3234 (br), 2910, 2866,1647, 1062 cm-1;
1H-NMR(CDCl3): δ = 3.67 (dd, J = 2.7 Hz, J = 11.7 Hz, 1H), 3.98 (dd, J = 3.0 Hz,J = 11.7 Hz, 1H), 4.30-4.54(m, 3H), 7.24-7.52 (m, 3H), 7.85-7.98 (m, 2H);
13C-NMR (400MHz,CDCl3): δ = 165.5, 131.4, 128.2, 128.2, 127.1, 69.1, 68.1, 63.7;
HPLC: AY-H column(4.6mmφ,250mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm,
flow rate: 1.0 mL/min, retention time: 18.4 min, 22.9 min, 89% ee;
HMRS(M+H, FAB): m/z calcd for C10H12NO2: 178.0868, found:178.0872.
実施例1の原料中のメチル基をエチル基で置換したものを用いる以外は実施例1と同様に実験を行ったところ(+)-4-ethyl-4,5-dihydro-4-hydroxymethyl-2-phenyloxazoleを得た。収率は83%であった。光学純度を測定したところ、97%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 14 = +6.2 (c 0.945, CHCl3);
IR(neat): ν=3248(br), 2967, 2928, 2858, 1645, 1067cm-1;
1H-NMR(CDCl3): δ=0.90 (t, J = 7.2 Hz, 3H), 1.45-1.80 (m, 2H), 3.50 (d, J = 11.7 Hz, 1H), 3.81 (d, J = 11.1 Hz, 1H), 4.20 (d, J = 8.1 Hz, 1H), 4.39 (d, J = 8.4 Hz, 1H), 7.32-7.50 (m, 3H), 7.87 (d, J = 8.4 Hz, 1H);
13C-NMR(400MHz, CDCl3): δ = 164.1, 131.0, 128.1, 127.9, 126.9, 75.3, 71.8, 66.6, 28.9, 7.4;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 16.0 min, 32.8 min, 97% ee;
HMRS(M+H, FAB): m/z calcd for C12H16NO2: 206.1181, found: 206.1180.
実施例1の原料中のメチル基をアリル基で置換したものを用いる以外は実施例1と同様に実験を行ったところ、(+)-4-allyl-4,5-dihydro-4-hydroxymethyl-2-phenyloxazoleを得た。収率は71%であった。光学純度を測定したところ、92%eeの(+)―体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 17 = +53.8 (c 0.975, CHCl3);
IR(neat): ν = 3238 (br), 3072, 2900, 2860, 1641, 1067 cm-1;
1H-NMR(CDCl3): δ = 1.23 (br, 1H), 2.20-2.50 (m, 2H), 3.54 (d, J = 11.7 Hz, 1H), 3.84 (d, J = 11.7 Hz, 1H), 4.24 (d, J = 8.7 Hz, 1H), 4.36 (d, J = 8.4 Hz, 1H), 5.05-5.20 (m, 2H), 5.60-5.82 (m, 1H), 7.22-7.50 (m, 3H), 7.85 (d, J = 7.8 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.6, 132.5, 131.3, 128.2, 128.0, 126.9, 119.0, 74.7, 71.9, 66.8, 40.9;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 22.5 min, 62.4 min, 92% ee;
HMRS(M, EI): m/z calcd for C13H15NO2: 217.1103, found: 217.1103.
常温、常圧にて実験を行った。tert-ブタノール2ml中で、0.05mmolのCu(OTf)2及び0.05mmolの(R,R)-Ph-Boxを混合し、10分間撹拌した。この溶液に、原料として0.5mmolの下記式
これに1.5mmolの炭酸カリウムを加え、最後に塩化p-トルエンスルホニル(1mmol)を加え、12時間撹拌した。反応後、水を加え酢酸エチルで3回抽出した。有機層を無水硫酸ナトリウムで乾燥させて濾過した後、溶媒を減圧留去し、残渣をカラムクロマトグラフィー(展開溶液:n-ヘキサン:酢酸エチル=1:1(V/V)の混合溶液)を用いた。)で精製し、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-phenyloxazoleを得た。収率は85.0%であった。光学純度を測定したところ、95.4%eeのR体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 16 = +15.2 (c 0.10, CHCl3);
IR(neat): ν = 3171 (br), 2972, 2895, 2862, 1641, 1076 cm-1;
1H-NMR(CDCl3): δ = 1.32 (s, 3H), 3.49 (d, J = 11.4 Hz, 1H), 3.78 (d, J = 11.1 Hz, 1H), 4.09 (d, J = 8.1 Hz, 1H), 4.47 (d, J = 8.4 Hz, 1H), 7.33-7.41 (m, 2H), 7.43-7.50 (m, 1H), 7.84-7.91 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.1, 131.2, 128.2, 128.0, 127.1, 74.5, 71.9, 67.8, 23.7;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 10 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 15.3 min, 26.9 min, 95.4% ee;
HRMS(M+H, FAB): m/z calcd for C11H14NO2: 191.1024, found: 191.1038.
実施例15の原料中のフェニル基の4位をメチル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(4-methylphenyl)oxazoleを得た。収率は89.3%であった。光学純度を測定したところ、94.2%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 23 = +1.7 (c 0.50, CHCl3);
IR(neat): ν = 3192 (br), 2963, 2922, 2851, 1645, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.39 (s, 3H), 3.48 (d, J = 11.1 Hz, 1H), 3.76 (d, J = 11.4 Hz, 1H), 4.08 (d, J = 7.8 Hz, 1H), 4.44 (d, J = 8.1 Hz, 1H), 7.19 (d, J = 7.8 Hz, 2H), 7.79 (d, J = 8.1 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.3, 141.6, 128.8, 128.2, 124.4, 74.5, 71.8, 68.0, 23.7, 21.5;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 47.9 min, 62.2 min, 94.2% ee;
HRMS(M+H, FAB): m/z calcd for C12H16NO2: 206.1181, found: 206.1192.
実施例15の原料中のフェニル基の3位をメチル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(3-methylphenyl)oxazolを得た。収率は72.9%であった。光学純度を測定したところ、99.9%以上eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 16 = -5.6 (c 1.00, CHCl3);
IR(neat): ν = 3223 (br), 2967, 2924, 2862, 1645, 1072 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.37 (s, 3H), 3.49 (d, J = 11.1 Hz, 1H), 3.77 (d, J = 11.4 Hz, 1H), 4.09 (d, J = 8.1 Hz, 1H), 4.46 (d, J = 8.4 Hz, 1H), 7.20-7.34 (m, 2H), 7.66-7.78 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.2, 137.6, 131.9, 128.7, 127.9, 126.9, 125.2, 74.4, 71.9, 67.8, 23.7, 21.1;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 33.1 min, 73.4 min, >99.9% ee;
HRMS(M, EI): m/z calcd for C12H15NO2: 205.1098, found: 205.1100.
実施例15の原料中のフェニル基の2位をメチル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(2-methylphenyl)oxazolを得た。収率は85.8%であった。光学純度を測定したところ、91.9%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 21 = +14.2 (c 1.00, CHCl3);
IR(neat): ν = 3304 (br), 2967, 2926, 2870, 1639, 1045 cm-1;
1H-NMR(CDCl3): δ = 1.35 (s, 3H), 1.88 (br, 1H), 2.56 (s, 3H), 3.49 (d, J = 10.8 Hz, 1H), 3.75 (d, J = 11.1 Hz, 1H), 4.06 (d, J = 8.1 Hz, 1H), 4.40 (d, J = 8.1 Hz, 1H), 7.18-7.29 (m, 2H), 7.31-7.38 (m, 1H), 7.76 (d, J = 7.5 Hz, 1H);
13C-NMR(400MHz, CDCl3): δ = 165.1, 138.5, 131.1, 130.6, 129.8, 127.1, 125.5, 74.3, 72.1, 68.1, 23.7, 21.4;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 31.8 min, 40.5 min, 91.9% ee;
HRMS(M+H, EI): m/z calcd for C12H16NO2: 206.1181, found: 206.1171.
実施例15の原料中のフェニル基の4位をメトキシ基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-2-(4-methoxylphenyl)-4-methyloxazoleを得た。収率は63.7%であった。光学純度を測定したところ、93.8%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 21 = -20.2 (c 1.00, CHCl3);
IR(neat): ν = 3208 (br), 2967, 2941, 2845, 1637, 1252, 1080 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 3.47 (d, J = 11.1 Hz, 1H), 3.75 (d, J = 11.4 Hz, 1H), 3.85 (s, 3H), 4.08 (d, J = 8.1 Hz, 1H), 4.44 (d, J = 8.1 Hz, 1H), 6.86-6.94 (m, 2H), 7.84-7.92 (m, 2H);
13C-NMR(400MHz, CDCl3) : δ = 164.0, 162.0, 130.0, 119.6, 113.4, 74.5, 71.7, 67.9, 55.3, 23.7;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 21.0 min, 37.5 min, 93.8% ee;
HRMS(M+H, FAB): m/z calcd for C12H16NO3: 222.1130, found: 222.1130.
実施例15の原料中のフェニル基の4位をフッ素で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-2-(4-fluorophenyl)-4-methyloxazoleを得た。収率は90.8%であった。光学純度を測定したところ、93.9%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 19 = -8.8 (c 1.00, CHCl3);
IR(neat): ν = 3184 (br), 2972, 2899, 2855, 1647, 1510, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.27 (br, 1H), 3.48 (d, J = 11.7 Hz, 1H), 3.76 (d, J = 11.4 Hz, 1H), 4.08 (d, J = 8.1 Hz, 1H), 4.46 (d, J = 7.8 Hz, 1H), 7.04-7.12 (m, 2H), 7.88-7.95 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 165.2, 163.2, 163.1, 130.4, 130.3, 123.2, 115.1, 114.9, 74.5, 72.0, 67.5, 23.6;
HPLC: OD-H column(4.6 mmφ, 500 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 62.9 min, 71.9 min, 93.9% ee;
HRMS(M+H, FAB): m/z calcd for C11H13FNO2: 210.0930, found: 210.0938.
実施例15の原料中のフェニル基の4位を塩素で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-2-(4-chlorophenyl)-4,5-dihydro-4-hydroxymethyl-4-methyloxazoleを得た。収率は97.3%であった。光学純度を測定したところ、95.6%eeのR体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 21 = -32.2 (c 1.00, CHCl3);
IR(neat): ν = 3198 (br), 2967, 2897, 2860, 1651, 1492, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.27 (br, 1H), 3.49 (t, J = 7.8 Hz, 1H), 3.76 (d, J = 12.3 Hz, 1H), 4.09 (dd, J = 0.9, 8.1 Hz, 1H), 4.47 (d, J = 7.8 Hz, 1H), 7.35 (d, J = 8.7 Hz, 2H), 7.82 (d, J = 8.4 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 163.2, 137.5, 129.5, 128.3, 125.5, 74.6, 72.1, 67.7, 23.7;
HPLC: AY-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 20.8 min, 38.8 min, 95.6% ee;
HRMS(M+H, FAB): m/z calcd for C11H13ClNO2: 226.0635, found: 226.0627.
実施例15の原料中のフェニル基の4位を臭素で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-2-(4-bromophenyl)-4,5-dihydro-4-hydroxymethyl-4-methyloxazoleを得た。収率は98.1%であった。光学純度を測定したところ、91.6%eeのR体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 19 = -35.5 (c 1.00, CHCl3);
IR(neat): ν = 3219 (br), 2965, 2897, 2862, 1649, 1072 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.16 (br, 1H), 3.44-3.54 (m, 1H), 3.71-3.82 (m, 1H), 4.08 (dd, J = 0.6, 8.3 Hz, 1H), 4.54 (d, J = 8.1 Hz, 1H), 7.63 (d, J = 8.7 Hz, 2H), 7.76 (d, J = 8.7 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 163.3, 131.3, 129.7, 126.0, 125.0, 74.6, 72.1, 67.7, 23.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 200 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 93.4 min, 107.8 min, 91.6% ee;
HRMS(M+H, FAB): m/z calcd for C11H13BrNO2: 270.0130, found: 270.0153.
実施例15の原料中のフェニル基の4位をヨウ素で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-2-(4-iodophenyl)-4-methyloxazoleを得た。収率は96.9%であった。光学純度を測定したところ、86.5%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 18 = -41.4 (c 1.00, CHCl3);
IR(neat): ν = 3221 (br), 2972, 2932, 2854, 1636, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.32 (s, 3H), 2.17 (br, 1H), 3.42-3.56 (m, 1H), 3.79 (d, J = 11.1 Hz, 1H), 4.13 (dd, J = 0.6, 8.3 Hz, 1H), 4.49 (d, J = 8.1 Hz, 1H), 7.57-7.63 (m, 2H), 7.70-7.76 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 163.5, 137.3, 129.7, 126.5, 98.5, 74.6, 72.1, 67.7, 23.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 200 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 109.0 min, 135.1 min, 86.5% ee;
HRMS(M+H, EI): m/z calcd for C11H13INO2: 317.9991, found: 317.9986.
実施例15の原料中のフェニル基の2位をヨウ素で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-2-(2-iodophenyl)-4-methyloxazoleを得た。収率は87.0%であった。光学純度を測定したところ、96.9%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 27 = +25.3 (c 0.86, CHCl3);
IR(neat): ν = 3292 (br), 2967, 2926, 2868, 1654, 1053 cm-1;
1H-NMR(CDCl3): δ = 1.38 (s, 3H), 3.51 (d, J = 11.7 Hz, 1H), 3.80 (d, J = 11.1 Hz, 1H), 4.13 (d, J = 8.1 Hz, 1H), 4.49 (d, J = 8.4 Hz, 1H), 7.13 (dt, J = 1.5, 7.5 Hz, 1H), 7.39 (dt, J = 1.2, 7.5 Hz, 1H), 7.56 (dd, J = 1.9, 7.8 Hz, 1H), 7.93 (dd, J = 0.9, 7.8 Hz, 1H);
13C-NMR(400MHz, CDCl3): δ = 164.8, 140.1, 133.5, 131.6, 130.3, 127.8, 94.7, 75.1, 72.4, 67.8, 23.3;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1 (+ 0.1% Et2NH), wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 23.4 min, 25.3 min, 96.9% ee;
HMRS(M, EI): m/z calcd for C11H12INO2: 317.9914, found: 317.9904.
実施例15の原料中のフェニル基の4位をフェニル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(4-phenylphenyl)oxazoleを得た。収率は91.1%であった。光学純度を測定したところ、90.6%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 20 = -64.6 (c 1.00, CHCl3);
IR(neat): ν = 3221 (br), 2972, 2932, 2907, 1638, 1082 cm-1;
1H-NMR(CDCl3): δ = 1.31 (s, 3H), 2.32 (br, 1H), 3.48 (d, J = 11.4 Hz, 1H), 3.76 (d, J = 11.1 Hz, 1H), 4.13 (dd, J = 0.6, 8.3 Hz, 1H), 4.46 (d, J = 8.1 Hz, 1H), 7.21-7.28 (m, 2H), 7.34-7.50 (m, 2H), 7.62 (d, J = 7.8 Hz, 3H), 7.94-8.04 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.0, 143.9, 140.2, 128.8, 128.7, 127.9, 127.1, 126.7, 126.0, 74.5, 72.0, 67.9, 23.7;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 100 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 54.2 min, 72.1 min, 90.6% ee;
HRMS(M, EI): m/z calcd for C17H17NO2: 267.1259, found: 267.1258.
実施例15の原料中のフェニル基の4位をニトロ基で置換したものを用いる以外は実施例15と同様に実験を行ったところ(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(4-nitrophenyl)oxazoleを得た。収率は91.4%であった。光学純度を測定したところ、93.1%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 22 = -9.4 (c 1.00, CHCl3);
IR(neat): ν = 3225 (br), 2970, 2933, 2857, 1524, 1078 cm-1;
1H-NMR(CDCl3): δ = 1.36 (s, 3H), 1.60 (br, 1H), 3.48-3.57 (m, 1H), 3.83 (d, J = 12.0 Hz, 1H), 4.17 (dd, J = 0.9, 8.1 Hz, 1H), 4.48-4.56 (m, 1H), 7.94-8.30 (m, 4H);
13C-NMR(400MHz, CDCl3): δ = 162.2, 149.4, 132.9, 129.2, 123.3, 75.1, 72.6, 67.8, 23.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 52.6 min, 60.5 min, 93.1% ee;
HRMS(M, EI): m/z calcd for C11H12N2O4: 236.0797, found: 236.0798.
実施例15の原料中のフェニル基をナフチル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(2-naphthyl)oxazoleを得た。収率は90.0%であった。光学純度を測定したところ、99.2%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 17 = -65.5 (c 1.00, CHCl3);
IR(neat): ν = 3207 (br), 2967, 2928, 2864, 1645, 1067 cm-1;
1H-NMR(CDCl3): δ = 1.26 (s, 3H), 1.60 (br, 1H), 3.54 (d, J = 11.7 Hz,1H), 3.83 (d, J = 10.8 Hz, 1H), 4.16 (d, J = 8.1 Hz, 1H), 4.55 (d, J = 7.8 Hz, 1H), 7.43-7.60 (m, 2H), 7.78-7.9 (m, 3H), 7.93-8.02 (m, 1H), 8.31-8.41 (m, 1H);
13C-NMR(400MHz, CDCl3): δ = 164.2, 134.5, 132.3, 128.8, 128.8, 127.6, 127.5, 127.4, 126.3, 124.5, 124.2, 74.5, 72.1, 67.9, 23.8;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 23.0 min, 35.4 min, 99.2% ee;
HMRS(M, EI): m/z calcd for C15H15NO2: 241.1103, found: 241.1081.
実施例15の原料中のフェニル基をスチリル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-styryloxazoleを得た。収率は20.1%であった。光学純度を測定したところ、94.3%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 16 = -43.0 (c 0.935, CHCl3);
IR(neat): ν = 3225 (br), 2970, 2933, 2857, 1524, 1078 cm-1;
1H-NMR(CDCl3): δ = 1.26 (br, 1H), 1.28 (s, 3H), 3.45 (d, J = 11.7 Hz, 1H), 3.74 (d, J = 11.7 Hz, 1H), 4.02 (d, J = 8.1 Hz, 1H), 4.46 (d, J = 7.8 Hz, 1H), 6.51 (d, J = 16.2 Hz, 1H), 7.19-7.38 (m, 6H);
13C-NMR(400MHz, CDCl3): δ = 164.1, 140.5, 134.9, 129.5, 128.7, 127.5, 114.4, 74.5, 71.6, 67.9, 23.5;
HPLC: AY-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 10 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 88.4 min, 102.4 min, 94.3% ee;
HMRS(M, EI): m/z calcd for C13H15NO2: 217.1103, found: 217.1083.
実施例15の原料中のフェニル基を2-フリル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4,5-dihydro-2-(2-furyl)-4-hydroxymethyl-4-methyloxazoleを得た。収率は69.9%であった。光学純度を測定したところ、99.0%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 18 = +6.0 (c 0.5, CHCl3);
IR(neat): ν = 3233 (br), 3130, 2968, 2928, 2866, 1670, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.64 (br, 1H), 3.48 (d, J = 11.7 Hz, 1H), 3.78 (d, J = 11.7 Hz, 1H), 4.07 (d, J = 8.4 Hz, 1H), 4.47 (d, J = 8.1 Hz, 1H), 6.42-6.50 (m, 1H), 6.92 (d, J = 3.0 Hz, 1H), 7.48-7.54 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 156.3, 145.1, 142.4, 114.6, 111.4, 74.5, 72.1, 67.7, 23.5;
HPLC: AY-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 24.7 min, 29.7 min, 99.0% ee;
HMRS(M, EI): m/z calcd for C9H11NO3: 181.0739, found: 181.0733.
実施例15の原料中のフェニル基を2-チエニル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(2-thienyl)oxazoleを得た。収率は66.8%であった。光学純度を測定したところ、94.0%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 20 = +3.5 (c 0.5, CHCl3);
IR(neat): ν = 3146 (br), 3090, 2976, 2855, 1638, 1078 cm-1;
1H-NMR(CDCl3): δ = 1.33 (s, 3H), 2.50 (br, 1H), 3.48 (d, J = 11.4 Hz, 1H), 3.77 (d, J = 11.4 Hz, 1H), 4.09 (d, J = 8.4 Hz, 1H), 4.48 (d, J = 8.4 Hz, 1H), 7.02-7.08 (m, 1H), 7.44 (dd, J = 1.2, 4.8 Hz, 1H), 7.58 (dd, J = 0.9, 3.6 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 159.7, 130.6, 129.9, 129.6, 127.4, 74.9, 72.3, 67.6, 23.6;
HPLC: AY-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 29.5 min, 37.4 min, 94.0% ee;
HMRS(M, EI): m/z calcd for C9H11NO2S: 197.0510, found: 197.0496.
実施例15の原料中のフェニル基を2-ピロリル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(-)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-(2-pyrrolyl)oxazoleを得た。収率は28.1%であった。光学純度を測定したところ、89.2%eeの(-)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 24 = -4.5 (c 1.04 CHCl3);
IR(neat): ν = 3217 (br), 3130, 2928, 2872, 1647, 1040 cm-1;
1H-NMR(CDCl3): δ = 1.38 (s, 3H), 3.54 (d, J = 12.3 Hz, 1H), 3.80 (d, J = 12.0 Hz, 1H), 4.24 (d, J = 8.4 Hz, 1H), 4.61 (d, J = 8.4 Hz, 1H), 6.42 (t, J = 2.4 Hz, 1H), 6.82 (d, J = 3.6 Hz, 1H), 7.05 (s, 1H);
13C-NMR(400MHz, CDCl3): δ = 160.9, 126.8, 118.0, 115.9, 111.1, 76.5, 69.1, 66.6, 22.8;
HPLC: AS column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 10 : 1 (+ 0.1% Et2NH), wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 11.7 min, 14.1 min, 89.2% ee;
HMRS(M, EI): m/z calcd for C9H12N2O2: 180.0899, found: 180.0895.
実施例15の原料中のフェニル基を3-フリル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4,5-dihydro-2-(3-furyl)-4-hydroxymethyl-4-methyloxazoleを得た。収率は74.0%であった。光学純度を測定したところ、99.3%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
m.p. 97-99 °C;
[α]D 24 = +15.1 (c 0.74, CHCl3); IR(neat): ν = 3184 (br), 3113, 2968, 2903, 2859, 1672, 1070 cm-1;
1H-NMR(CDCl3): δ = 1.26 (s, 3H), 3.44 (d, J = 11.7 Hz, 1H), 3.78 (d, J = 11.7 Hz, 1H), 4.00 (d, J = 8.1 Hz, 1H), 4.45 (d, J = 8.1 Hz, 1H), 6.69 (s, 1H), 7.33 (s, 1H), 7.72 (s, 1H);
13C-NMR(400MHz, CDCl3): δ = 159.6, 145.1, 143.4, 115.2, 109.3, 74.4, 71.8, 67.8, 23.6;
HPLC: AS column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 30 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 13.9 min, 24.2 min, 99.3% ee;
HMRS(M, EI): m/z calcd for C9H11NO3: 181.0739, found: 181.0733.
実施例15の原料中のメチル基を水素原子で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4,5-dihydro-4-hydroxymethyl-2-phenyloxazoleを得た。収率は32.2%であった。光学純度を測定したところ、88.9%eeのR体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 22 =+64.2(c 0.77, CHCl3); IR(neat): ν = 3234 (br), 2910, 2866, 1647, 1062 cm-1;
1H-NMR(CDCl3): δ = 3.67 (dd, J = 2.7 Hz, J = 11.7 Hz, 1H), 3.98 (dd, J = 3.0 Hz,J = 11.7 Hz, 1H), 4.30-4.54(m, 3H), 7.24-7.52 (m, 3H), 7.85-7.98 (m, 2H);
13C-NMR (400MHz,CDCl3): δ = 165.5, 131.4, 128.2, 128.2, 127.1, 69.1, 68.1, 63.7;
HPLC: AY-H column(4.6mmφ,250mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm,
flow rate: 1.0 mL/min, retention time: 18.4 min, 22.9 min, 88.9% ee;
HMRS(M+H, FAB): m/z calcd for C10H12NO2: 178.0868, found:178.0872.
実施例15の原料中のメチル基をエチル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ(+)-4-ethyl-4,5-dihydro-4-hydroxymethyl-2-phenyloxazoleを得た。収率は82.6%であった。光学純度を測定したところ、97.1%eeの(+)-体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 14 = +6.2 (c 0.945, CHCl3);
IR(neat): ν=3248(br), 2967, 2928, 2858, 1645, 1067cm-1;
1H-NMR(CDCl3): δ=0.90 (t, J = 7.2 Hz, 3H), 1.45-1.80 (m, 2H), 3.50 (d, J = 11.7 Hz, 1H), 3.81 (d, J = 11.1 Hz, 1H), 4.20 (d, J = 8.1 Hz, 1H), 4.39 (d, J = 8.4 Hz, 1H), 7.32-7.50 (m, 3H), 7.87 (d, J = 8.4 Hz, 1H);
13C-NMR(400MHz, CDCl3): δ = 164.1, 131.0, 128.1, 127.9, 126.9, 75.3, 71.8, 66.6, 28.9, 7.4;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 16.0 min, 32.8 min, 97.1% ee;
HMRS(M+H, FAB): m/z calcd for C12H16NO2: 206.1181, found: 206.1180.
実施例15の原料中のメチル基をアリル基で置換したものを用いる以外は実施例15と同様に実験を行ったところ、(+)-4-allyl-4,5-dihydro-4-hydroxymethyl-2-phenyloxazoleを得た。収率は70.8%であった。光学純度を測定したところ、92.3%eeの(+)―体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 17 = +53.8 (c 0.975, CHCl3);
IR(neat): ν = 3238 (br), 3072, 2900, 2860, 1641, 1067 cm-1;
1H-NMR(CDCl3): δ = 1.23 (br, 1H), 2.20-2.50 (m, 2H), 3.54 (d, J = 11.7 Hz, 1H), 3.84 (d, J = 11.7 Hz, 1H), 4.24 (d, J = 8.7 Hz, 1H), 4.36 (d, J = 8.4 Hz, 1H), 5.05-5.20 (m, 2H), 5.60-5.82 (m, 1H), 7.22-7.50 (m, 3H), 7.85 (d, J = 7.8 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.6, 132.5, 131.3, 128.2, 128.0, 126.9, 119.0, 74.7, 71.9, 66.8, 40.9;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 22.5 min, 62.4 min, 92.3% ee;
HMRS(M, EI): m/z calcd for C13H15NO2: 217.1103, found: 217.1103.
実施例15の原料中のメチル基をベンジル基で置換したものを用いる以外は実施例15 と同様に実験を行ったところ、(+)-4-benzyl-4,5-dihydro-4-hydroxymethyl-2-phenyloxazoleを得た。収率は62.3%であった。光学純度を測定したところ、90.6%eeの(+)―体であった。
上記反応により得られた光学活性オキサゾリン化合物の構造及び物性データを示す。
[α]D 17 = +101.4 (c 1.24, CHCl3);
IR(neat): ν = 3246 (br), 3062, 2920, 2858, 1645, 1066 cm-1;
1H-NMR(CDCl3): δ = 1.23 (br, 1H), 2.90 (dd, J = 33.9, 13.8 Hz, 2H), 3.56 (d, J = 11.7 Hz, 1H), 3.90 (d, J = 11.7 Hz, 1H), 4.28 (d, J = 7.2 Hz, 2H), 7.14-7.32 (m, 7H), 7.35-7.43 (m, 1H), 7.70 (d, J = 7.9 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.7, 136.0, 131.2, 130.5, 128.2, 128.1, 128.1, 127.0, 126.6, 75.5, 71.7, 67.1, 42.3;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 20 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 12.0 min, 27.1 min, 90.6% ee;
HMRS(M, EI): m/z calcd for C17H17NO2: 267.1260, found: 267.1271.
オキサゾリンの光学分割
常温、常圧にて実験を行った。tert-ブタノール2ml中で、0.03mmolのCu(OTf)2及び0.03mmolの(R,R)-Ph-Boxを混合し、10分間攪拌した。この溶液に、原料として0.3mmolの下記式
これに0.45mmolの炭酸カリウムを加え、最後に塩化p-トルエンスルホニル(0.36mmol)を加え、12時間撹拌した。反応後、水を加え酢酸エチルで3回抽出した。有機層を無水硫酸ナトリウムで乾燥させて濾過した後、溶媒を減圧留去し、残渣をカラムクロマトグラフィー(展開溶液:n-ヘキサン:酢酸エチル=1:1(V/V)の混合溶液)で精製し、(+)-4,5-dihydro-4-hydroxymethyl-4-methyl-2-phenyloxazole(収率40%、光学純度95%ee)及び(-)-4,5-dihydro-2-methyl-4-phenyl-4-tosyloxymethyloxazole(収率60%、光学純度60%ee)を得た。
上記反応により得られた生成物の構造及び物性データを示す。
[α]D 21 = +15.2 (c 0.10, CHCl3);
IR(neat): ν = 3171 (br), 2972, 2895, 2862, 1641, 1076 cm-1;
1H-NMR(CDCl3): δ = 1.32 (s, 3H), 3.49 (d, J = 11.4 Hz, 1H), 3.78 (d, J = 11.1 Hz, 1H), 4.09 (d, J = 8.1 Hz, 1H), 4.47 (d, J = 8.4 Hz, 1H), 7.33-7.41 (m, 2H), 7.43-7.50 (m, 1H), 7.84-7.91 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.1, 131.2, 128.2, 128.0, 127.1, 74.5, 71.9, 67.8, 23.7;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 10 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 15.3 min, 26.9 min, 95% ee;
HMRS(M+H, FAB): m/z calcd for C11H14NO2: 191.1024, found: 191.1038.
[α]D 19 = -35.4 (c 1.00, CHCl3);
IR(neat): ν = 3061, 2974, 2903, 2901, 1645, 1358, 1175 cm-1;
1H-NMR(CDCl3): δ = 1.36 (s, 3H), 2.42 (s, 1H), 3.94-4.06 (m, 3H), 4.43 (d, J = 9.0 Hz, 1H), 4.50 (d, J = 8.7 Hz, 1H), 7.20-7.52 (m, 5H), 7.72-7.78 (m, 2H), 7.82-7.88 (m, 2H);
13C-NMR(400MHz, CDCl3): δ = 164.5, 144.9, 132.5, 131.6, 129.8, 128.3, 128.2, 127.9, 127.1, 75.0, 74.2, 69.8, 23.6, 21.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Isopropanol = 10 : 1, wavelength : 254 nm, flow rate: 0.5 mL/min, retention time: 16.7min, 25.8 min, 60% ee;
HMRS(M, EI): m/z calcd for C18H19NO4S : 345.1035, found : 345.1038.
常温、常圧にて実験を行った。アセトニトリル2ml中で、0.05mmolのCu(OTf)2及び0.05mmolの(R,R)-Ph-Boxを混合し、10分間撹拌した。この溶液に、原料として0.5mmolの下記式
これに2.0mmolの炭酸ナトリウムを加え、最後に塩化p-トルエンスルホニル(1.5mmol)を加え、室温で15時間撹拌した。反応後、水を加え酢酸エチルで三回抽出した。有機層を無水硫酸ナトリウムで乾燥させて濾過した後、溶媒を減圧留去し、残渣をシリカゲルカラムクロマトグラフィー (展開溶媒 n-ヘキサン:酢酸エチル = 1:1(V/V)の混合溶液) で精製した。
上記反応により得られた生成物の構造及び物性データを示す。
m.p. 75-78 °C;
[α]D 21 = -36.1 (c 0.50, CHCl3);
IR(neat): ν = 3229 (br), 2957, 1626, 1358, 1169 cm-1;
1H-NMR(CDCl3): δ = 2.57 (s, 3H), 3.62 (d, J = 11.7 Hz, 1H), 3.73 (d, J = 11.7 Hz, 1H), 4.04 (d, J = 9.9 Hz, 1H), 4.17 (d, J = 9.9 Hz, 1H), 4.29 (d, J = 9.3 Hz, 1H), 4.39 (d, J = 9.0 Hz, 1H), 7.27-7.41 (m, 4H), 7.44-7.52 (m, 1H), 7.72-7.85 (m, 4H);
13C-NMR(400MHz, CDCl3): δ = 166.4, 145.1, 132.3, 132.0, 129.9, 128.5, 128.3, 128.0, 126.6, 74.4, 71.3, 71.0, 64.7, 21.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 10 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 12.4 min, 23.9 min, 96% ee;
HMRS(M+H, FAB): m/z calcd for C18H20NO5S: 362.1062, found: 362.1062.
m.p. 124-126 °C;
IR(neat): ν = 2955, 2926, 2857, 2361, 1643, 1362, 1177 cm-1;
1H-NMR(CDCl3): δ = 2.43 (s, 6H), 4.01 (d, J = 10.2 Hz, 2H), 4.12 (d, J = 9.9 Hz, 2H), 4.30 (s, 2H), 7.20-7.44 (m, 6H), 7.46-7.54 (m, 1H), 7.68-7.81 (m, 6H);
13C-NMR(400MHz, CDCl3): δ = 166.5, 145.2, 132.2, 132.1, 130.0, 128.6, 128.3, 128.0, 126.5, 72.5, 71.2, 71.5, 21.7;
HMRS(M+H, FAB): m/z calcd for C25H26NO7S2: 516.1151, found: 515.1158.
実施例38の原料中の塩化p-トルエンスルホニルに代えて塩化ベンゼンスルホニル(1.25mmol)を用いた以外は実施例38と同様に実験を行った。
上記反応により得られた生成物の構造及び物性データを示す。
m.p. 118-120 °C;
[α]D 24 = -31.1 (c 1.22, CHCl3);
IR(neat): ν = 3237 (br), 3067, 2905, 2870, 1636, 1360, 1186 cm-1;
1H-NMR(CDCl3): δ = 3.63 (d, J = 10.8 Hz, 1H), 3.74 (d, J = 11.4 Hz, 1H), 4.07 (d, J = 9.9 Hz, 1H), 4.17 (d, J = 9.9 Hz, 1H), 4.29 (d, J = 9.3 Hz, 1H), 4.41 (d, J = 9.0 Hz, 1H), 7.34-7.43 (m, 2H), 7.45-7.57 (m, 3H), 7.61-7.69 (m, 1H), 7.80-7.93 (m, 4H);
13C-NMR(400MHz, CDCl3): δ = 166.5, 135.3, 134.0, 131.9, 129.3, 128.4, 128.2, 127.9, 126.4, 74.3, 71.2, 71.1, 64.5;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 10 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 14.7 min, 29.7 min, 91% ee;
HMRS(M+H, FAB): m/z calcd for C17H18NO5S: 348.0906, found: 348.0891.
IR(neat): ν = 3069, 2924, 2855, 1640, 1362, 1188 cm-1;
1H-NMR(CDCl3): δ = 4.05 (d, J = 9.9 Hz, 2H), 4.15 (d, J = 9.9 Hz, 2H), 4.30 (s, 2H), 7.20-7.44 (m, 6H), 7.34-7.44 (m, 2H), 7.46-7.57 (m, 4H), 7.61-7.68 (m, 2H), 7.74-7.81 (m, 2H), 7.82-7.90 (m, 3H);
13C-NMR(400MHz, CDCl3): δ = 166.6, 135.2, 134.1, 132.2, 129.4, 128.6, 128.3, 128.0, 126.4, 72.5, 71.2, 70.7;
HMRS(M+H, FAB): m/z calcd for C23H22NO7S2: 488.0838, found: 488.0828.
実施例38の原料中の塩化p-トルエンスルホニルに代えて塩化p-クロロベンゼンスルホニル(1.25mmol)を用いた以外は実施例38と同様に実験を行った。
上記反応により得られた生成物の構造及び物性データを示す。
m.p. 121-122 °C;
[α]D 19 = -38.0 (c 1.695, CHCl3);
IR(neat): ν = 3254 (br), 2959, 1639, 1362, 1186 cm-1;
1H-NMR(CDCl3): δ = 3.63 (d, J = 10.8 Hz, 1H), 3.73 (d, J = 11.7 Hz, 1H), 4.12 (d, J = 8.1 Hz, 1H), 4.19-4.32 (m, 2H), 4.41 (d, J = 8.4 Hz, 1H), 7.35-7.55 (m, 5H), 7.74-7.87 (m, 4H);
13C-NMR(400MHz, CDCl3): δ = 166.5, 140.7, 133.9, 132.1, 129.6, 129.3, 128.4, 128.3, 126.4, 74.3, 71.4, 71.1, 64.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 10 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 13.1 min, 24.8 min, 75% ee;
HMRS(M+H, FAB): m/z calcd for C17H17ClNO5S: 382.0516, found: 382.0519.
m.p. 132-134 °C;
IR(neat): ν = 3094, 2963, 2909, 2857, 2357, 1643, 1364, 1186 cm-1;
1H-NMR(CDCl3): δ = 4.08 (d, J = 10.2 Hz, 2H), 4.12 (d, J = 10.2 Hz, 2H), 4.32 (s, 2H), 7.36-7.56 (m, 6H), 7.70-7.82 (m, 7H);
13C-NMR(400MHz, CDCl3): δ = 166.7, 140.9, 133.7, 132.3, 129.7, 129.4, 128.5, 128.4, 126.2, 72.5, 71.2, 70.8;
HMRS(M+H, FAB): m/z calcd for C23H20Cl2NO7S2: 556.0059, found: 556.0059.
実施例38の原料中のフェニル基を4-メチルフェニル基で置換したものを用いた以外は実施例38と同様に実験を行った。
上記反応により得られた生成物の構造及び物性データを示す。
m.p. 121-122 °C;
[α]D 16 = -36.2 (c 0.50, CHCl3);
IR(neat): ν = 3248 (br), 2949, 2924, 1638, 1358, 1173 cm-1;
1H-NMR(CDCl3): δ = 2.38 (s, 3H), 2.43 (s, 3H), 3.61 (d, J = 11.4 Hz, 1H), 3.71 (d, J = 11.7 Hz, 1H), 4.02 (d, J = 9.9 Hz, 1H), 4.16 (d, J = 9.6 Hz, 1H), 4.27 (d, J = 9.0 Hz, 1H), 4.37 (d, J = 9.0 Hz, 1H), 7.12 (d, J = 7.8 Hz, 2H), 7.31 (d, J = 8.1 Hz, 2H), 7.70 (d, J = 8.1 Hz, 2H), 7.76 (d, J = 8.4 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 166.6, 145.1, 142.5, 132.3, 129.9, 129.0, 128.5, 128.0, 123.8, 74.2, 71.2, 71.0, 65.0, 21.6, 21.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 10 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 12.7 min, 15.0 min, 86% ee;
HMRS(M+H, FAB): m/z calcd for C19H22NO5S: 376.1219, found: 376.1209.
m.p. 110-112 °C;
IR(neat): ν = 2955, 2924, 2361, 2342, 1641, 1360, 1175 cm-1;
1H-NMR(CDCl3): δ = 2.39 (s, 3H), 2.43 (s, 6H), 4.00 (d, J = 9.9 Hz, 2H), 4.11 (d, J = 9.9 Hz, 2H), 4.28 (s, 2H), 7.18 (d, J = 7.8 Hz, 2H), 7.30 (d, J = 8.4 Hz, 4H), 7.66 (d, J = 8.1 Hz, 2H), 7.72 (d, J = 8.4 Hz, 4H);
13C-NMR(400MHz, CDCl3): δ = 166.5, 145.2, 142.6, 132.1, 129.9, 129.0, 128.5, 127.9, 123.6, 72.4, 71.1, 70.6, 21.6, 21.6;
HMRS(M+H, FAB): m/z calcd for C26H28NO7S2: 530.1307, found: 530.1331.
実施例38の原料中のフェニル基を3-メチルフェニル基で置換したものを用いた以外は実施例38と同様に実験を行った。
上記反応により得られた生成物の構造及び物性データを示す。
[α]D 19 = -30.0 (c 0.50, CHCl3);
IR(neat): ν = 3244 (br), 2951, 2922, 2359, 2344, 1639, 1358, 1175 cm-1;
1H-NMR(CDCl3): δ = 2.33 (s, 3H), 2.43 (s, 3H), 3.61 (d, J = 11.7 Hz, 1H), 3.72 (d, J = 11.4 Hz, 1H), 4.01 (d, J = 9.9 Hz, 1H), 4.14 (d, J = 9.9 Hz, 1H), 4.29 (d, J = 9.0 Hz, 1H), 4.37 (d, J = 9.0 Hz, 1H), 7.18-7.34 (m, 4H), 7.53-7.61 (m, 2H), 7.72-7.78 (m, J = 8.1 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 166.6, 145.0, 137.9, 132.6, 132.2, 129.9, 128.9, 128.1, 128.0, 126.3, 125.5, 74.2, 71.1, 71.0, 64.4, 21.6, 21.1;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 10 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 11.3 min, 29.7 min, 97% ee;
HMRS(M+H, FAB): m/z calcd for C19H22NO5S: 376.1218, found: 376.1210.
m.p. 131-133 °C;
IR(neat): ν = 2953, 2924, 2853, 2363, 1643, 1360, 1175 cm-1;
1H-NMR(CDCl3): δ = 2.37 (s, 3H), 2.43 (s, 6H), 4.00 (d, J = 10.2 Hz, 2H), 4.11 (d, J = 10.2 Hz, 2H), 4.29 (s, 2H), 7.23-7.34 (m, 6H), 7.54-7.64 (m, 2H), 7.70-7.76 (m, 4H);
13C-NMR(400MHz, CDCl3): δ = 166.7, 145.2, 138.1, 132.9, 132.1, 129.9, 129.1, 128.2, 128.0, 126.3, 125.7, 72.5, 71.2, 70.6, 21.6, 21.2;
HMRS(M, FAB): m/z calcd for C26H27NO7S2: 529.1229, found: 529.1221.
実施例38の原料中のフェニル基を2-メチルフェニル基で置換したものを用いた以外は実施例38と同様に実験を行った。
上記反応により得られた生成物の構造及び物性データを示す。
m.p. 103-106 °C;
[α]D 17 = -31.0 (c 1.275, CHCl3);
IR(neat): ν = 3296 (br), 2957, 2926, 2363, 1638, 1358, 1175 cm-1;
1H-NMR(CDCl3): δ = 2.43 (s, 3H), 2.47 (s, 3H), 3.60 (d, J = 11.4 Hz, 1H), 3.71 (d, J = 11.1 Hz, 1H), 4.07 (d, J = 9.6 Hz, 1H), 4.14-4.25 (m, 2H), 4.34 (d, J = 8.7 Hz, 1H), 7.16-7.39 (m, 5H), 7.70 (d, J = 7.5 Hz , 1H), 7.77 (m, J = 8.4 Hz, 2H);
13C-NMR(400MHz, CDCl3): δ = 167.2, 145.1, 138.9, 132.4, 132., 131.1, 129.9, 129.9, 127.9, 126.2, 125.6, 74.6, 71.0, 70.7, 64.8, 21.7, 21.6;
HPLC: OD-H column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 30 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 38.5 min, 40.6 min, >99% ee;
HMRS(M+H, FAB): m/z calcd for C19H22NO5S: 376.1218, found: 376.1222.
m.p. 64-67 °C;
IR(neat): ν = 2956, 2922, 2853, 2361, 1636, 1362, 1177 cm-1;
1H-NMR(CDCl3): δ = 2.41 (s, 3H), 2.43 (s, 6H), 2.69 (s, 1H), 4.03 (d, J = 9.9 Hz, 2H), 4.12 (d, J = 9.9 Hz, 2H), 4.26 (s, 2H), 7.15-7.39 (m, 7H), 7.62-7.69 (m, 1H), 7.74 (d, J = 8.4 Hz, 4H);
13C-NMR(400MHz, CDCl3): δ = 167.2, 145.2, 139.3, 132.2, 131.3, 130.0, 129.6, 128.0, 127.8, 125.9, 125.5, 72.9, 70.6, 70.5, 21.8, 21.7;
HMRS(M+H, FAB): m/z calcd for C25H28NO7S2: 530.1307, found: 530.1319.
実施例38の原料中のフェニル基を4-クロロフェニル基で置換したものを用いた以外は実施例38と同様に実験を行った。
上記反応により得られた生成物の構造及び物性データを示す。
m.p. 128-130 °C;
[α]D 17 = -25.8 (c 0.50, CHCl3);
IR(neat): ν = 3331 (br), 3065, 2926, 2868, 2359, 1651, 1362, 1173 cm-1;
1H-NMR(CDCl3): δ = 2.09 (br, 1H), 2.44 (s, 3H), 3.60-3.78 (m, 2H), 4.05 (d, J = 9.6 Hz, 1H), 4.17 (d, J = 9.9 Hz, 1H), 4.29 (d, J = 9.0 Hz, 1H), 4.40 (d, J = 9.3 Hz, 1H), 7.23-7.40 (m, 4H), 7.73-7.82 (m, 4H);
13C-NMR(400MHz, CDCl3): δ = 165.5, 145.2, 138.3, 132.3, 129.9, 129.8, 128.6, 128.0, 125.1, 74.5, 71.5, 70.9, 64.7, 21.7;
HPLC: AY-H column(4.6 mmφ, 250 mm), n-Hexane : Ethanol = 10 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 31.2 min, 41.8 min, 68% ee;
HMRS(M+H, FAB): m/z calcd for C18H19ClNO5S: 398.0643, found: 398.0660.
IR(neat): ν = 2924, 2857, 2359, 1647, 1364, 1177 cm-1;
1H-NMR(CDCl3): δ = 2.44 (s, 3H), 4.01 (d, J = 9.9 Hz, 2H), 4.11 (d, J = 9.9 Hz, 2H), 4.31 (s, 2H), 7.23-7.39 (m, 6H), 7.68-7.75 (m, 6H);
13C-NMR(400MHz, CDCl3): δ = 165.6, 145.3, 138.4, 132.2, 130.0, 129.9, 128.6, 128.0, 124.9, 72.7, 71.4, 70.5, 21.7;
HMRS(M+H, FAB): m/z calcd for C25H25ClNO7S2: 552.0732, found: 552.0740.
実施例38の原料中のフェニル基を2-クロロフェニル基で置換したものを用いた以外は実施例38と同様に実験を行った。
上記反応により得られた生成物の構造及び物性データを示す。
[α]D 20 = -24.0 (c 1.41, CHCl3);
IR (neat): ν = 3314 (br), 3067, 2955, 2924, 1647, 1358, 1175 cm-1;
1H-NMR (CDCl3): δ = 2.43 (s, 3H), 2.57 (br, 1H), 3.61 (d, J = 11.4 Hz, 1H), 3.71 (d, J = 11.4 Hz, 1H), 4.12 (d, J = 9.9 Hz, 1H), 4.18 (d, J = 9.9 Hz, 1H), 4.29 (d, J = 9.0 Hz, 1H), 4.39 (d, J = 9.0 Hz, 1H),7.24-7.46 (m, 5H), 7.65 (dd, J = 1.5, 7.5 Hz, 1H), 7.78 (d, J = 8.4 Hz, 2H);
13C-NMR (400MHz, CDCl3): δ = 165.1, 145.1, 133.3, 132.3, 132.0, 131.2, 130.6, 129.9, 127.9, 126.6, 126.6, 74.9, 71.3, 70.8, 64.7, 21.6;
HPLC: AY-H column (4.6 mmφ, 250 mm), n-Hexane : Ethanol = 4 : 1, wavelength : 254 nm, flow rate: 1.0 mL/min, retention time: 14.6 min, 27.9 min, 96% ee;
HMRS (M+H, FAB): m/z calcd for C18H19ClNO5S: 398.0643, found: 398.0642.
IR (neat): ν = 3067, 2957, 2924, 2852, 1645, 1360, 1175 cm-1;
1H-NMR (CDCl3): δ = 2.44 (s, 3H), 4.06 (d, J = 10.2 Hz, 2H), 4.14 (d, J = 9.9 Hz, 2H), 4.34 (s, 2H), 7.23-7.45 (m, 7H), 7.79-7.64 (m, 1H), 7.72-7.80 (m, 4H);
13C-NMR (400MHz, CDCl3): δ = 165.4, 145.3, 133.5, 132.2, 132.1, 131.5, 130.7, 130.0, 128.0, 126.5, 126.3, 72.9, 71.3, 70.4, 21.7;
HMRS (M+H, FAB): m/z calcd for C25H25ClNO7S2: 552.0731, found: 552.0718.
実施例38、41~44における原料化合物の構造、各反応の収率及び光学純度を以下の表1に示す。表1において、R6は原料化合物中のオキサゾリン環の2位に結合した置換基である。
スルホニルハライドの種類及び添加量を種々変化させて、実施例38と同様の条件で光学活性オキサゾリン化合物を合成した。スルホニルハライドの構造、スルホニルハライドの添加量、各反応の収率、及び光学純度を以下の表2に示す。表2において、スルホニルハライドは一般式R7-Ph-SO2Clで表される化合物である。
なお、スルホニルハライドの添加量は、原料化合物1モルに対するモル等量である。また、表中の副生成物とは、4級炭素に結合する水酸基がいずれも同じ保護基で保護された化合物をいう。
スルホニルハライドの添加量を原料化合物1モルに対し2.5モルとし、塩基の種類及び添加量を種々変化させた以外は実施例38と同様の条件で、光学活性オキサゾリン化合物を合成した。塩基の種類、添加量、各反応の収率、及び光学純度を以下の表3に示す。
なお、塩基の添加量は、原料化合物1モルに対するモル等量である。また、表中の副生成物とは、4級炭素に結合する水酸基がいずれも同じ保護基で保護された化合物をいう。
スルホニルハライドの添加量を原料化合物1モルに対し2.5モルとし、塩基を炭酸ナトリウムに代えて炭酸カリウムとし、溶媒の種類を種々変化させた以外は実施例38と同様の条件で、光学活性オキサゾリン化合物を合成した。溶媒の種類、各反応の収率、及び光学純度を以下の表4に示す。
なお、表中の副生成物とは、4級炭素に結合する水酸基がいずれも同じ保護基で保護された化合物をいう。
スルホニルハライドの添加量を原料化合物1モルに対し2.5モルとし、溶媒及び塩基の種類を種々変化させた以外は実施例38と同様の条件で、光学活性オキサゾリン化合物を合成した。溶媒の種類、塩基の種類、各反応の収率、及び光学純度を以下の表5に示す。
なお、表中の副生成物とは、4級炭素に結合する水酸基がいずれも同じ保護基で保護された化合物をいう。
よって、本発明は産業上有用に用いることができる。
Claims (10)
- 式(1)
で表される化合物を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に閉環させることを特徴とする式(3)
で示される光学活性化合物又はその塩の製造方法。 - R1が炭素数6~20のアリール基、炭素数2~15で、異種原子として少なくとも1個の窒素原子、酸素原子、硫黄原子等の異種原子を含んでいる、5~8員の単環式ヘテロアリール基、多環式若しくは縮合環式のヘテロアリール基又はアルキル基、アルケニル基、アルコキシ基、ハロゲン原子、ニトロ基若しくはアリール基で置換されているフェニル基である請求項1に記載の製造方法。
- R2が水素又は炭化水素基である請求項1又は2に記載の製造方法。
- MmZnがCu(OTf)2である請求項1~3のいずれかに記載の製造方法。
- さらに塩基の存在下に反応を行う請求項1~4のいずれかに記載の製造方法。
- さらに有機溶媒の存在下に反応を行う請求項1~5のいずれかに記載の製造方法。
- 式(4)
又は式(4’)
で示される光学活性化合物又はその塩。 - 式(5)
で表される化合物のラセミ体を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させ、式(3)
で表される光学活性化合物又はその塩の(R)体又は(S)体及び式(6)
で表される光学活性化合物又はその塩の(R)体又は(S)体を光学選択的に生成する製造方法。 - 式(7)
で表される化合物を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させることを特徴とする式(8)
で示される光学活性化合物又はその塩の製造方法。 - 式(9)
で表される化合物を、
一又は二以上の配位部位を有する不斉配位子、
式(2)
で表されるルイス酸及び
置換されていてもよいアルキル基又はフェニル基を有するスルホニルハライドの存在下に反応させ、式(8)
で表される光学活性化合物又はその塩の(R)体又は(S)体を生成する製造方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/982,707 US8969585B2 (en) | 2011-01-31 | 2012-01-31 | Method for producing optically active compound or salt thereof |
| CN201280007276.1A CN103415511B (zh) | 2011-01-31 | 2012-01-31 | 光学活性化合物或其盐的制造方法 |
| JP2012555910A JPWO2012105574A1 (ja) | 2011-01-31 | 2012-01-31 | 光学活性化合物又はその塩の製造方法 |
| EP12742476.0A EP2671877A4 (en) | 2011-01-31 | 2012-01-31 | PROCESS FOR PRODUCING OPTICALLY ACTIVE COMPOUND OR SALT THEREOF |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011019287 | 2011-01-31 | ||
| JP2011-019287 | 2011-01-31 | ||
| JP2011-153342 | 2011-07-11 | ||
| JP2011153342 | 2011-07-11 | ||
| JP2011153692 | 2011-07-12 | ||
| JP2011-153692 | 2011-07-12 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012105574A1 true WO2012105574A1 (ja) | 2012-08-09 |
Family
ID=46602781
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/052176 WO2012105574A1 (ja) | 2011-01-31 | 2012-01-31 | 光学活性化合物又はその塩の製造方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US8969585B2 (ja) |
| EP (1) | EP2671877A4 (ja) |
| JP (1) | JPWO2012105574A1 (ja) |
| CN (1) | CN103415511B (ja) |
| WO (1) | WO2012105574A1 (ja) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006219464A (ja) * | 2005-02-14 | 2006-08-24 | Nagasaki Univ | 1,3−ジオール誘導体の製造方法 |
| JP2008037865A (ja) * | 2006-07-14 | 2008-02-21 | Nagasaki Univ | 光学活性ビスオキサゾリン−銅錯体を不斉触媒とするn−保護アミノアルコール化合物の不斉エステル化反応 |
| JP2008290981A (ja) * | 2007-05-25 | 2008-12-04 | Nagasaki Univ | 光学活性モノスルホネート化合物の製造方法 |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2556791A (en) | 1945-11-29 | 1951-06-12 | Billman | Alpha-amino-alpha-methylol malonic acid |
| JPH04273255A (ja) | 1991-02-28 | 1992-09-29 | Dainippon Ink & Chem Inc | 静電荷像現像用トナー |
| AU658955B2 (en) * | 1992-01-28 | 1995-05-04 | Sumitomo Chemical Company, Limited | An oxazoline derivative, its production and its use |
| US5352832A (en) * | 1992-12-18 | 1994-10-04 | Schering Corporation | Asymmetric process for preparing florfenicol, thiamphenicol chloramphenicol and oxazoline intermediates |
| JP2006515891A (ja) * | 2003-01-17 | 2006-06-08 | シーブイ・セラピューティクス・インコーポレイテッド | 循環器疾患の処置に有効な置換ヘテロ環化合物 |
| WO2005021503A1 (en) * | 2003-08-28 | 2005-03-10 | Novartis Ag | Aminopropanol derivatives |
| KR100854211B1 (ko) * | 2003-12-18 | 2008-08-26 | 동아제약주식회사 | 신규한 옥사졸리디논 유도체, 그의 제조방법 및 이를유효성분으로 하는 항생제용 약학 조성물 |
| TWI345568B (en) * | 2004-04-02 | 2011-07-21 | Mitsubishi Tanabe Pharma Corp | Tetrahydronaphthyridine derivatives and a process for preparing the same |
| US8889839B2 (en) * | 2008-05-07 | 2014-11-18 | Syracuse University | Pyridine-bis (oxazoline)(“pybox”) moiety as a chelator and sensitizer for lanthanide ion (Ln (III)) Luminescence |
| WO2010082601A1 (ja) * | 2009-01-16 | 2010-07-22 | 第一三共株式会社 | 新規2,5-二置換ピロール誘導体 |
-
2012
- 2012-01-31 JP JP2012555910A patent/JPWO2012105574A1/ja active Pending
- 2012-01-31 US US13/982,707 patent/US8969585B2/en not_active Expired - Fee Related
- 2012-01-31 WO PCT/JP2012/052176 patent/WO2012105574A1/ja active Application Filing
- 2012-01-31 EP EP12742476.0A patent/EP2671877A4/en not_active Withdrawn
- 2012-01-31 CN CN201280007276.1A patent/CN103415511B/zh not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006219464A (ja) * | 2005-02-14 | 2006-08-24 | Nagasaki Univ | 1,3−ジオール誘導体の製造方法 |
| JP2008037865A (ja) * | 2006-07-14 | 2008-02-21 | Nagasaki Univ | 光学活性ビスオキサゾリン−銅錯体を不斉触媒とするn−保護アミノアルコール化合物の不斉エステル化反応 |
| JP2008290981A (ja) * | 2007-05-25 | 2008-12-04 | Nagasaki Univ | 光学活性モノスルホネート化合物の製造方法 |
Non-Patent Citations (7)
| Title |
|---|
| A. L. BRAGA ET AL., TETRAHEDRON ASYMMETRY, vol. 14, 2003, pages 3291 - 3295 |
| DANIEL OBRECHT ET AL., J. ORG. CHEM., vol. 61, 1996, pages 4080 - 4086 |
| G. JONES; C. J. RICHARDS, TETRAHEDRON LETTERS, vol. 42, 2001, pages 5553 - 5555 |
| HONG, M. S. ET AL.: "Enantioselective formation of tert-alkylamines by desymmetrization of 2-substituted serinols", CHEMISTRY--A EUROPEAN JOURNAL, vol. 14, no. 11, 2008, pages 3290 - 3296, XP055077885 * |
| J. AM. CHEM. SOC., vol. 113, 1991, pages 728 - 729 |
| OSAMU ONOMURA ET AL.: "Shokubaiteki Fusai Hitaishokaho ni yoru Kogaku Kassei Oxazoline Gosei", 131ST ANNUAL MEETING OF PHARMACEUTICAL SOCIETY OF JAPAN (SHIZUOKA) HAPPYO YOSHI, vol. VIII-5-1, 1 February 2011 (2011-02-01), XP008169608, Retrieved from the Internet <URL:http://nenkai.pharm.or.jp/131/pc/ipdview.asp?i=293> * |
| See also references of EP2671877A4 * |
Also Published As
| Publication number | Publication date |
|---|---|
| US8969585B2 (en) | 2015-03-03 |
| CN103415511A (zh) | 2013-11-27 |
| EP2671877A1 (en) | 2013-12-11 |
| US20140012010A1 (en) | 2014-01-09 |
| JPWO2012105574A1 (ja) | 2014-07-03 |
| EP2671877A4 (en) | 2015-07-01 |
| CN103415511B (zh) | 2016-01-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4787021B2 (ja) | グアニジン化合物及びそれを用いる不斉反応 | |
| CN110099893B (zh) | 一种屈西多巴及其中间体的制备方法 | |
| CN109438205B (zh) | 一种2-甲基-2,3-二芳基丙醛衍生物的合成方法 | |
| CN111423394B (zh) | 1,3,4-恶二唑杂环化合物的合成方法 | |
| JP5417560B2 (ja) | 光学活性カルボン酸エステルを製造する方法 | |
| EP2139829A1 (en) | N-heterocyclic carbene (nhc) catalyzed synthesis of hydroxamic acids | |
| WO2012105574A1 (ja) | 光学活性化合物又はその塩の製造方法 | |
| JP6858993B1 (ja) | アミノアルコール−ボロン−バイノール複合体及びこれを用いた光学活性アミノアルコール誘導体の製造方法 | |
| EP3442957B1 (en) | "process for preparing intermediates useful in the synthesis of antifungal drugs" | |
| JP5408662B2 (ja) | ジスルホン酸化合物の製法、不斉マンニッヒ触媒、β−アミノカルボニル誘導体の製法及び新規なジスルホン酸塩 | |
| WO2011111762A1 (ja) | ジアリール誘導体の製造方法、新規ビナフチル誘導体、アレーン誘導体の製造方法、及び新規アレーン誘導体 | |
| JP4918736B2 (ja) | 新規光学活性ルテニウム錯体、その製法並びにそれを用いた光学活性化合物の製造方法 | |
| JP2007246517A (ja) | 光学活性な5−ヒドロキシ−3−ケトエステル化合物の製法 | |
| JP4869739B2 (ja) | 含フッ素ジヒドロキノリン化合物及び含フッ素キノリン化合物の製造方法 | |
| JP4546773B2 (ja) | シアノ化合物の製造法 | |
| JP4208174B2 (ja) | アミノ化合物類ならびにその立体選択的製造方法 | |
| US20210179536A1 (en) | Method of synthesizing (1r,2r)-nitroalcohol compound | |
| US8507715B2 (en) | Method for producing β-aminocarbonyl compound | |
| WO2010098193A1 (ja) | β-アミノカルボニル化合物の製法及びリチウムビナフトラート錯体 | |
| JP2013107844A (ja) | 光学活性アジリジン化合物の製造方法 | |
| JP2001252567A (ja) | キラル金属触媒とチオールの不斉マイケル付加反応方法 | |
| JP3012370B2 (ja) | 異性体分離法 | |
| Lee | New Complexity-Building Reactions of α-Keto Esters | |
| KR20220140556A (ko) | 비대칭 수소화를 사용하는 s-베플루부타미드의 합성 방법 | |
| Bartlett | New Complexity-Building Reactions of Alpha-Keto Esters |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12742476 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2012555910 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012742476 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012742476 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13982707 Country of ref document: US |