WO2012021046A2 - 요오드를 함유한 방사형상의 고분자 화합물, 이의 제조방법 및 이를 포함하는 ct용 조영제 조성물 - Google Patents
요오드를 함유한 방사형상의 고분자 화합물, 이의 제조방법 및 이를 포함하는 ct용 조영제 조성물 Download PDFInfo
- Publication number
- WO2012021046A2 WO2012021046A2 PCT/KR2011/006002 KR2011006002W WO2012021046A2 WO 2012021046 A2 WO2012021046 A2 WO 2012021046A2 KR 2011006002 W KR2011006002 W KR 2011006002W WO 2012021046 A2 WO2012021046 A2 WO 2012021046A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- iodine
- compound
- acid
- polymer compound
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/06—Macromolecular compounds, carriers being organic macromolecular compounds, i.e. organic oligomeric, polymeric, dendrimeric molecules
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/06—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations
- A61K49/08—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations characterised by the carrier
- A61K49/10—Organic compounds
- A61K49/12—Macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/4035—Isoindoles, e.g. phthalimide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/04—X-ray contrast preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/04—X-ray contrast preparations
- A61K49/0433—X-ray contrast preparations containing an organic halogenated X-ray contrast-enhancing agent
- A61K49/0442—Polymeric X-ray contrast-enhancing agent comprising a halogenated group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
- C08L101/02—Compositions of unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups
- C08L101/04—Compositions of unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups containing halogen atoms
Definitions
- the present invention relates to a radial polymer compound containing iodine which can be used as an active ingredient of a CT contrast agent, a method for preparing the same, and a CT contrast agent composition comprising the same.
- Computed tomography is an imaging technique in which the target area of the human body is irradiated from various directions and collected by a penetrating X-ray detector, and the computer reconstructs the hop aberration difference of the area using a mathematical technique.
- the technique In other words, the camera rotates 360 ° around the body, and is an imaging technique that recombines numerous 2D X-ray images taken from different angles into computer 3D images. Therefore, such X-ray-based images generally represent bone-based images.
- the degree of X-ray hops ( ⁇ ) is proportional to the atomic number ( ⁇ and the wavelength of the X-rays ( ⁇ ), ie the degree of X-ray hops ( ⁇ ) is inversely proportional to the energy of X-rays. do.
- CT provides anatomical information in addition to magnetic resonance imaging (MRI), which differentiates it from PEKpositron emission tomography (SPK) or single photon emission computed tomography (SPECT) imaging, which provides information about physiological and biochemical functions. do.
- MRI magnetic resonance imaging
- SPK PEKpositron emission tomography
- SPECT single photon emission computed tomography
- CT imaging time is about one tenth or less than MRI, which is considered to be an indispensable diagnostic device for patients with fatal damage to the brain area, especially those who have to make a quick diagnosis.
- iodine-containing low-molecular CT contrast agents are divided into ionic and nonionic materials.
- the nonionic material since the nonionic material has much less side effects than the ionic material, most of the patients are currently administered a nonionic CT contrast agent.
- a nonionic CT contrast agent shares 2-3 conventional low molecular nonionic CT contrast agents containing iodine with one benzene ring.
- the molecular weight can be increased to extend the residence time slightly compared to conventional single benzene ring-based nonionic contrast agents.
- these low-molecular CT contrast agents containing iodine generally have a very short residence time (half-life), so they can be administered an excess of 80-90 g in excess of 80-90 g depending on the weight of an adult in one CT scan to obtain an appropriate CT image.
- a low molecular iodine compound or a derivative thereof may be covalently substituted with a low molecular iodine compound in a polymer skeleton, or a commercially available low molecule iodine in a polymer compound skeleton such as liposomes, micelles and other nanoparticles.
- a commercially available low molecule iodine in a polymer compound skeleton such as liposomes, micelles and other nanoparticles.
- Non-Patent Document 2 [A. Sachse, et al. , Invest. Radiol. 1997, 32, 44-50]
- Non-Patent Document 3 [E. Samei, et al. , Int. J. Nanomedicine 2009, 4, 277-282 and Non-Patent Document 4
- C.-Y. Kao, et al. , Acad. Radiol. 2003, 10, 475-483 discloses a method for containing a polymer compound-based liposome containing a low molecular CT contrast agent compound or a derivative thereof containing commercially available iodine.
- Non-Patent Document 5 VS Trubetskoy, et al., J.
- Non-Patent Document 6 [VP Torchilin, et al. , Acad. Radiol. 1999, 6, 61-65 discloses a method for containing a high molecular compound-based micelle containing a low molecular CT contrast agent compound or a derivative thereof containing commercially available iodine.
- Non-Patent Document 7 [F. Hyafil, et al. , Nat. Med. 2007, 13, 636-641 discloses a method for containing a low molecular CT contrast agent compound or derivative thereof containing commercially available iodine in polymer compound-based nanoparticles.
- the carboxylic acid terminal of the iodine compound was capped with an alkyl group to increase hydrophobicity, thereby making it well contained in the nanoparticles.
- these nanoparticles containing iodine are injected into blood vessels, they can be easily absorbed into macrophage and increase the circulation time in the body. As time passes, inflammation, cancer and arteries are relatively distributed. Accumulation in areas such as hardening and consequently facilitated diagnosis of related diseases through CT imaging.
- Patent document 1 discloses a contrast agent for CT based on gold (Au) nanoparticles whose surface is substituted with PEG.
- Non-Patent Document 8 [I.-C. Sun, et al., Chem. Eur. J. 2009, 15, 13341-1334 discloses a contrast agent for CT based on gold nanoparticles coated with heparin.
- Non Patent Literature 9 [MH Oh et al., J. Am. Chem. Soc. 2011, 133, 5508-5515 discloses a contrast agent for CT based on tantalum oxide nanoparticles of uniform size substituted with PEG and fluorescent material.
- CT contrast agent based on the metal nanoparticles itself acts as a contrast agent (absorbs X-rays), and the surface of the nanoparticles is coated with PEG or heparin to improve biocompatibility and smooth CT imaging or liver by increasing the circulation time in the body, respectively. It was intended to increase the contrast effect of specific sites through the accumulation of.
- the various polymer-based contrast agents described above are not only expensive but also have limitations of safety due to long-term accumulation in vivo (ie, half-lives of more than a few decades) without the smooth excretion, or stability of the compound structure, ie
- the self-assembled structure will be difficult to secure the reproducibility to maintain the original structure stably even in the body environment and to obtain the same performance by obtaining a polymer compound showing the same molecular weight distribution at the time of manufacture. It is expected.
- the material for human administration based on such a polymer compound there are various challenges to commercialization through practical clinical trials since the toxicity test standards have not yet been established worldwide.
- CT angiographic contrast agents containing low molecular contrast media containing iodine in liposome form include Fenestra (ART of Canada) and eXIA160 (Binitio Biomedical of Canada), both for animal use only. It is sold.
- the liposome CT angiography contrast agent is a very expensive price for a single vial (2.5 mL) to be injected into a mouse or rat 5-10 times. The injection of contrast medium through the tail vein of such a small animal is very expensive. It is not easy for basic medical researchers who need to take micro CT scans.
- a small molecule contrast medium for human body is administered to a micro CT scan using a small animal, it may take at least 1-3 minutes for the animal to be moved to the CT scan immediately after the injection of the contrast medium and the software is operated to start the CT scan. In the meantime, most of the low-molecular contrast agent is discharged into the bladder through the kidneys, so that an adequate level of contrast is not practical. Therefore, in addition to the diagnosis of cardiovascular diseases in which the low-molecular CT contrast agent containing the above-mentioned iodine is excessively administered to humans to obtain an appropriate level of CT images, it is necessary to properly conduct experiments using small animals.
- Dendrimers are relatively small, treelike or radial-shape polymers, typically less than 10 nm in diameter, that are pure monolecules obtained through stepwise iterative organic synthesis and purification. to be. Unlike most other typical polymers (polydisperse mixtures), dendrimers are always constant and somewhat predictable in certain environments (solvent, pH, temperature, etc.), especially in solution of polymer-based particles It is very advantageous as it can be selectively adapted to various research fields that are sensitive to hydrodynamic diameter.
- dendrimers include the integrity of chemical structures, the ability to make up the dendrimer, and the subsequent modifications of physicochemical properties, depending on the organic synthesis method applied, and various functional units ( For example, low molecular weight drugs, target materials, surface modifiers, etc.) can be easily substituted into and inside dendrimers, and in biological applications, there is little potential for significant enzymatic degradation in vivo. None can turn etc.
- Examples of the application of the dendrimer to the biopharmaceutical field include the use of polycationic dendrimers to form a charge complex with anionic genes and to efficiently carry it into cells (gene transfection); In the manner in which a dendrimer used as a drug delivery carrier carries a drug, the drug molecule is physically stored in a porous space in the dendrimer such that the drug is released only in the affected area by a specific stimulus (pH, light, enzyme, etc.).
- Covalent substitution of the dendrimer in the form of physical encapsulation or prodrug Delivery of the drug at a targeted or controlled release rate through structural modification of the dendrimer carrier; Using a multivalent effect to significantly increase binding affinity upon interaction ions in the extracellular membrane between proteins and carbohydrate ligands; Examples of polysubstituting a small molecule compound based imaging agent into a dendrimer backbone to amplify the signal to enable more effective medical diagnosis; Biocompatible and biodegradation Examples include the manufacture and application of artificial tissue engineering based on biodegradable dendrimer skeletons.
- Dendrimers were first described as polylysine dendrimers synthesized by Denkewalter in the late 1970s, and their molecular design and synthesis, their physicochemical properties, and basic use cases based on them (eg, self-assembly, Biomimetic systems, etc.), its materials and applications in the medical field have been actively studied.
- Examples of polylysine dendrimer structures synthesized by Denkewalter are as follows (Patent 2 [US 4,410,688]).
- PAMAM poly (amidoamine) dendrimers among the types of dendrimers are It was developed by Donald A. Tomalia at Dow Chemical Company in the 1980s, and its internal structure is composed of aliphatic amine group or aliphatic amide group, and its surface is composed of amine group, carboxyl group, and hydroxyl group. It is a functional group.
- the structure of the 1-4 generation (Gl, G2, G3, and G4) PAMAM dendrimers whose core is ethylenediamine and whose surface is composed of amine groups is as follows.
- G2 PA A endnmer (2a)
- PAMAM dendrimers are known to be suitable for in vivo applications among dendrimers and are widely applied in the biopharmaceutical field.
- dendrimers synthesized through stepwise organic synthesis and purification should in principle be a single molecule
- commercially available PAMAM dendrimers manufactured by Dendritech, sold by Sigma-Aldrich
- reactants reagent
- divergent synthetic method a divergent synthetic method.
- the mass production of very simple purification only by dialysis method, and relatively undesired side-products similar to the target are produced by the residual reaction product, resulting in incomplete and heterogeneity. Because of the problem of polydispersity index (PDI), ca.
- PDI polydispersity index
- Patent Document 3 [ PCT / EP94 / 04245], Patent Document 4 [PCT / EP96 / 02450], Patent Document 5 [PCT / FR92 / 01135], Patent Document 6 [PCT / EP94 / 648203], Patent Document 7 [PCT / EP95 / 730573] , Patent Document 8 [PCT / EP95 / 78263], Non-Patent Document 11 [AT Yordanov, et al., Nano Lett. 2002, 2, 595-599], Non-Patent Document 12 [Y. Fu, et al., Biocon jugate Chem. 2006, 17, 1043-1056).
- Gd-DTPA gadopentetate dimeglumine
- Magnevist a representative single low molecular weight compound MRI angiography currently administered to patients in hospitals.
- Bayer Schering the first company to develop an angiography contrast agent for MRI based on a dendrimer and filed a patent (EP 430863, W0 97/02051, W0 98/24775, W0 98/24774, US 5911971). 17 “(black is called” Gd-DTPA-17 “or” SH L 643 A "; K. Nael, et al., J. Magn. Reson.
- the radial polymer compound containing iodine characterized in that the biocompatible polymer forms a protective film surrounding the core and the iodine-containing compound substituted therein Biocompatible polymer chains with relatively long lengths of iodine-containing compounds, which significantly improve the duration of contrasting effects and cause toxicities and various adverse reactions substituted near the surface of the core, are significantly improved compared to the conventional low-molecular contrast agent-containing compounds. It forms a protective layer surrounded by layers to prevent their exposure to the external environment, which not only reduces the toxicity in the body, but also prolongs the circulation time by preventing the rapid absorption by macrophages.
- An object of the present invention is to provide a radial polymer compound containing iodine.
- Another object of the present invention is to provide a method for producing a radial polymer compound containing the iodine.
- Still another object of the present invention is to provide a contrast agent composition for CT, comprising the radial polymer compound containing the iodine.
- the present invention is a central core consisting of a circular or spherical symmetrical low molecular compound, or a radial polymer compound; An iodine-containing compound bonded directly or through a peptide to the core core; And a biocompatible polymer coupled directly or via a peptide to the core, or directly to an iodine-containing compound bound to the core, wherein the biocompatible is such that the core and iodine-containing compound are not exposed in vivo.
- a radial polymer compound containing iodine having a structure in which a polymer forms a protective film.
- the present invention comprises the steps of reacting the biocompatible polymer to the portion of the surface functional group of the core consisting of a circular or spherical symmetrical low molecular compound, or a radial high molecular compound (step 1); And reacting the iodine-containing compound to the unreacted surface functional group in the core of step 1 such that the iodine-containing compound is bonded directly or through a temper (step 2). do.
- the present invention comprises the steps of reacting the iodine-containing compound to the portion of the surface functional group of the core consisting of a circular or spherical symmetrical low molecular compound, or a radial polymer compound (step 1); And a step (step 2) of reacting the biocompatible polymer to the iodine-containing compound bonded to the core in step 1 to provide a method for preparing the radial polymer compound containing the iodine.
- the present invention comprises the step of reacting the iodine-containing compound is bound through the biocompatible polymer and peptide (step 1); And reacting the iodine-containing compound bound to the biocompatible polymer through the peptide in Step 1 so as to bind to the surface functional group of the core, which is composed of a circular or spherical symmetrical low molecular compound or a radial polymer compound (step 2).
- Radial polymerization containing the iodine containing Provided are methods for preparing the compound.
- the present invention provides a contrast agent composition for CT comprising the radial polymer compound containing the iodine as an active ingredient.
- Radial polymer compound containing iodine according to the present invention is significantly improved the duration of contrast effect compared to the conventional low molecular contrast agent containing iodine, and may cause toxicity and various adverse reactions substituted near the surface of the core
- a protective film that encloses iodine-containing compounds in a relatively long layer of biocompatible polymer chains prevents their exposure to the external environment, thereby reducing toxicity in the body and preventing rapid absorption by macrophages, thereby circulating in the body
- extracorporeal discharge is made after proper time elapses after injection into the body, and the production method and purification method is very simple, so that mass production is possible with high yield and low cost, and the polydispersity is very low, making and effect Highly reproducible in terms of radial high iodine content Since all of the structure of the chair consists of a compound covalent bond structurally very stable and can be useful in the preparation of contrast agents for CT.
- Example 1 is a graph measuring cytotoxicity with a compound according to an embodiment of the present invention.
- A Example 1, b: Example 2, c: Example 3, d: Example 4
- Figure 2 is a 2D X-ray radiograph of the CT contrast agent containing a compound according to an embodiment of the present invention.
- Figure 3 is an image taken by using a micro CT at 0-4 hours intervals the coronal surface of the rat administered a CT contrast agent containing a compound according to an embodiment of the present invention.
- Figure 4 is an image taken using a micro CT at 8-48 hours intervals of the coronal surface of the rat administered a CT contrast agent containing a compound according to an embodiment of the present invention.
- FIG. 5 is an image taken using a micro CT at 0-4 hours intervals of the sagittal surface of the rat administered a CT contrast agent containing a compound according to an embodiment of the present invention.
- FIG. 6 is an image taken using a micro CT at 8-48 hour intervals of the sagittal surface of the rat administered a CT contrast agent containing a compound according to an embodiment of the present invention.
- FIG. 7 is an image taken by using a micro CT of the coronal plane of the chest and abdomen of the rat administered the CT contrast agent containing the compound according to an embodiment of the present invention, vena (vena cava) and the image for comparing the trend of signal strength in the liver (liver) region.
- 8 is an image taken by using a micro CT of the coronal plane of the chest and abdomen of the rat to which the CT contrast agent containing the compound according to an embodiment of the present invention, the trend of the signal intensity in the heart (heart) region Image for comparing.
- FIG. 9 is an image taken using a micro CT of the coronal plane of the brain region of the rat administered a CT contrast agent containing a compound according to an embodiment of the present invention.
- FIG. 10 is an image taken by using a micro CT of the sagittal plane of the brain region of the rat administered a CT contrast agent containing a compound according to an embodiment of the present invention.
- the present invention is a core or core consisting of a circular or spherical symmetrical low molecular compound, or a radial polymer compound;
- Iodine-containing compounds bonded directly to or through a peptide of the core; And a biocompatible polymer that is directly linked to the core or through a peptide or directly to an iodine containing compound bonded to the core.
- a radial high molecular compound containing iodine having a structure in which the biocompatible polymer forms a protective film so that the core and the iodine-containing compound are not exposed in vivo.
- the circular or spherical symmetrical low molecular compound constituting the core is ⁇ -, ⁇ - and cyclodextrin, glucose, galactose, mannose, and derivatives thereof.
- Radial polymer compounds constituting the core include polyamidoamine (PAMAM) dendrimers, polylysine dendrimers, polypropyleneimine (PPI) dendrimers, polyester dendrimers, polyglutamic acid dendrimers, polyaspartic acid dendrimers, and polyglycerides Any one kind of dendrimers selected from the group consisting of dreamers and polymelamine dendrimers, polylysine, polyester, polyglutamic acid, polyaspartic acid, and polyglycene; Any one star-shaped polymer selected from the group consisting of a hyperbranched polymer, polyethylene glycol (PEG), and a copolymer derivative thereof, and derivatives thereof can be used.
- PEG polyethylene glycol
- a copolymer derivative thereof and derivatives thereof can be used.
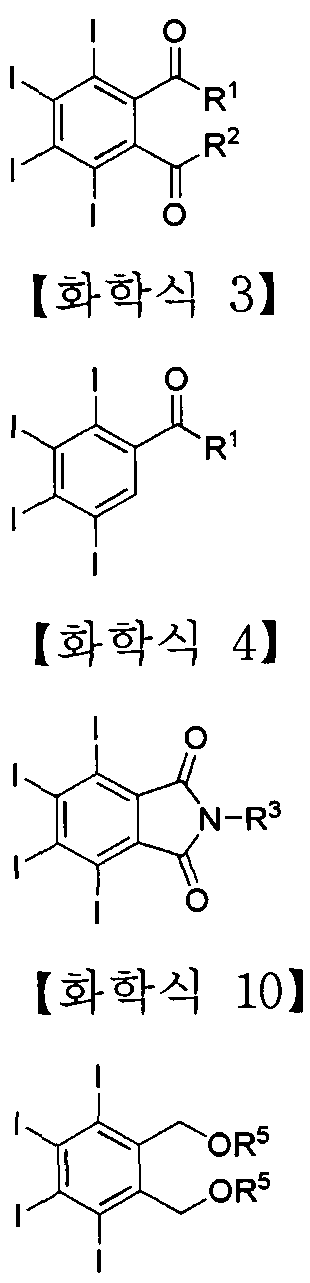
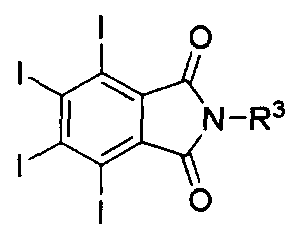
- a compound represented by the following Chemical Formulas 2, 3, 4 and 10 may be used as a preferred example of the iodine-containing compound.
- R 1 , R and R are each independently or optionally selected from -NH 2 , -NHR 4 , -NR 4 2 , -NHNH 2 ( -NHNHR 4 , -NHNR 4 2) -OH, -OR 4 , -SH or -SR 4 ,
- R 4 is —H, —Boc, 6 unsubstituted or substituted straight or branched chain alkyl or hetero Aryl or aryl of 7 to Tero, -alkyl, or unsubstituted or substituted C 5
- the iodine-containing compounds of Formulas 2, 3 and 4 may be prepared by using the iodine compound represented by the following Formula 1 and Formula 9 as a precursor.
- the biocompatible polymer may be polyethylene glycol (PEG), hyaluronic acid, heparin and derivatives thereof.
- the peptide is dilysine, trilysine, tetralysine, diglutamic acid, triglutamic acid, tetraglutamic acid, diaspartic acid, triaspartic acid, tetraaspartic acid And a peptide having 2-4 repeating units selected from the group consisting of dicysteine, tricysteine, tetracysteine, dieserine, triserine, and tetraserine.
- both ends of the connecting portion may be each independently or selectively.
- A, - (CH 2) m - , - (CH 2) m (C 0) -, (: three eu 20 cycloalkyl - ((: 3 ⁇ 4) "- , C 4 - 20 aryl - (CH 2 ) m -and
- the core is an amine group, hydroxy group, hydroxyamine (hydroxyamine) which can be combined with a peptide, an iodine-containing compound or a biocompatible polymer.
- Carboxyl group carboxyhydrazide group, hydrazine group, thiol group, azide group, alkynyl group, halogen group , aldehyde (aldehyde) group eu ketone (ketone) group, an epoxy (epoxy) group, 3-carboxylic bome rock
- aldehyde (aldehyde) group eu ketone (ketone) group
- an epoxy (epoxy) group 3-carboxylic bome rock
- non-piperidinyl (3-carbomethoxypyrroHdinone) group a tri- (alkoxy of d- 4) - silyl (tri- ( It has one or more surface functional groups, such as a d- 4 alkoxy) -silyl) group.
- the radial polymer compound containing iodine according to the present invention preferably has an average molecular weight of 8,000-150,000 Da. If the average molecular weight is less than 8,000 Da, the long-term circulation in the blood vessel is not sufficient, and similarly to the low molecular weight compound, it may be discharged relatively quickly through the kidney. Difficulties in infusion and increase in the likelihood of abnormal reaction (shock, allergy, etc.) in the body, increased accumulation in the liver due to high toxicity, difficulty in extracorporeal discharge (very long half-life).
- the contrast agent solution was a little thicker, and even more difficult than compounds lc, 2c, and 3c to dissolve in the complete solution at the concentration of the contrast agent.
- the actual G5 PAMAM dendrimer-based materials were prepared, synthesized and analyzed. However, they were difficult to dissolve at the concentrations in the sediment for use as contrast agents, and thus could not be used in the actual injection experiments. Also, the PDKpolydispersity index was relatively Very high compound uniformity was found to be not very useful.
- Preferred examples of the radial polymer compound containing iodine according to the present invention may be a compound represented by the following formulas (5) to (8).
- the core is any one cyclic hydrocarbon, porphyrin and derivatives thereof selected from the group consisting of ⁇ - , ⁇ - and ⁇ -cyclodextrin, glucose, galactose and mannose, D0TA (1, 4,7, 10—tetraazacyclododecane- 1,4,7, 10-tetraacetic acid) and derivatives thereof, at least one peptide selected from the group consisting of lysine, aspartic acid, glutamic acid, serine, cysteine and tyrosine 2-4 Cyclic peptides, polyamidoamine (PAMAM) dendrimers, polylysine dendrimers, polypropyleneimine (PPI) dendrimers, polyester dendrimers, polyglutamic acid dendrimers, polyaspartic acid dendrimers, polyglycerol dendrimers and polymelamines Any one dendrimer, polylysine or polyester selected from the group consisting of dendrimers From the group consist
- T and T are iodine-containing compounds
- R and R are each independently or alternatively
- D is a peptide such as lysine, trilysine, tetralysine digglutamic acid, triglutamic acid, tetraglutamic acid, diaspartic acid, triaspartic acid, tetraaspartic acid, discysteine, tricysteine, tetracysteine, diserine, triglycerine , And a peptide having 2-4 repeating units selected from the group consisting of tetraserine,
- P is polyethylene glycol (PEG), hyaluronic acid, heparin or derivatives thereof
- X is a surface functional group of the core, amine group, hydroxy group, hydroxyamine group, carboxyl (carboxyl) group, carboxyhydrazide group, hydrazine group, thiol group, azide group, alkynyl group, halogen group, aldehyde group Group, ketone group, epoxy group, 3-carbomethoxypyrrolidinone group, tri- (Ci- 4 alkoxy) -silyl (tri-CCw alkoxy) -si lyl )
- a and b are each independently or optionally an integer of 0 or 1
- c is an integer from 2-4
- d, e, f, g, h and i are each independently or alternatively an integer from 2 to 60, j is an integer from 1 to 10.
- j is an integer from 1 to 10.
- an alkoxy group of the d- 4 is the terminal facing outward from the core.
- the compound of formula 5 is imaged by combining the iodine-containing compound (red small circle) and the biocompatible polymer (long green wavy line) to the core (gray large circle), respectively.
- the compound of formula 6 is imaged by binding a biocompatible polymer to an iodine-containing compound bonded to the core.
- the compound of Formula 7 is imaged by independently binding a peptide (blue short wavy line) and a biocompatible polymer in which 2-4 iodine-containing compounds are bundled into a core,
- 2-4 iodine-containing compounds bound to the core are imaged of the compound of Formula 8 by binding the biocompatible polymer to the peptide substituted by the bundle.
- Radial polymer compound containing iodine according to the present invention is significantly improved the duration of contrast effect compared to the conventional low molecular contrast agent containing iodine, and may cause toxicity and various adverse reactions substituted near the surface of the core Iodine It forms a protective film that surrounds oil compounds with a relatively long biocompatible polymer chain layer to prevent their exposure to the external environment, thereby reducing toxicity in the body as well as preventing rapid absorption by macrophages, thereby reducing the body's circulation time.
- the present invention comprises the steps of reacting the biocompatible polymer to the portion of the surface functional group of the core consisting of a circular or spherical symmetrical low molecular compound, or a radial high molecular compound (step 1); And
- step 2 Providing a method for producing a radial polymer compound containing the iodine comprising the step (step 2) of reacting the iodine-containing compound directly or through the temper group to the unreacted surface functional group in the core of step 1 do.
- the present invention comprises the steps of reacting the iodine-containing compound to a portion of the surface functional group of the core consisting of a circular or spherical symmetrical low molecular compound, or a radial polymer compound (step 1);
- step 2 provides a method for producing a radial-phase polymer compound containing the iodine comprising the step (step 2) of reacting such that the biocompatible polymer is bonded to the iodine-containing compound bound to the core in step 1 above.
- the present invention comprises the steps of reacting the iodine-containing compound to be bonded through a biocompatible polymer and a peptide (step 1);
- Step 2 reacting the iodine-containing compound bound to the biocompatible polymer through the peptide in Step 1 to bind to a surface functional group of the core, which is composed of a circular or spherical symmetrical low molecular compound or a radial polymer compound (step 2). It provides a method for producing a radial polymer compound containing the iodine.
- the circular or spherical symmetrical low molecular compound, the radial polymer compound, Table Examples of the surface functional group, the iodine-containing compound, the peptide, and the biocompatible polymer are as described above.
- the surface functional group of the core remaining without the reaction by administering an excess of iodine-containing compound
- the iodine content in the radial high molecular compound can be adjusted to about 15-50% by binding to.
- the present invention also provides a contrast agent composition for CT containing the radial polymer compound containing the iodine as an active ingredient.
- the radial polymer compound containing iodine preferably has an iodine content of 15-50%. If the iodine content is less than 15% of the radial polymer compound containing iodine, the contrast effect is not sufficient, so an excessive contrast agent should be administered. If exceeded, there is a problem that it is difficult to use for in vivo injection because of increased toxicity.
- Triethylamine (0.82 mL, 5.88 ⁇ ol) was added to a solution of compound 60 ⁇ , 3.84 g, 5.89 mmol) and compound 9 (ethane to amine, 300 mg, 4.91 ⁇ ol) in DMS0 (8 mL), Heated to 50 ° C and stirred for 19 h. SEC column using DMF as mobile phase to remove DMS0 solvent from the reaction mixture (Model: Bio-Beads S-Xl, H
- Step 2 Preparation of Gl PAMAM-mPEG ⁇ -TIPAA Binder (lc) Compound lb solution (5.65 g) obtained in step 1 and 4,5,6,7-tetraiodobene
- reaction mixture After filling with argon gas, the reaction mixture was stirred at room temperature for 2 days, and then added thereto.
- TIPN (3.00 g, 4.60 mmol) was added all at once, and the reaction mixture was stirred at room temperature for 24 hours.
- the crude product obtained was purified by SEC (Model: Bio-Beads S-Xl, H 40 cm X0.D. 4.5 cm,
- Iodine Content (Inductively coupled plasma mass spectrometry, I CP-MS) :
- G2 PAMAM dendrimer (2a) purchased from Aldrich was dried in vacuo to remove methane.
- the compound 2a (1.32 g, 0.405 mmol) was dissolved in DMS0 (90 mL) and mPEG carbonate compound 5 (3.94 g, 1.81 dl) was added to the stirring solution in one portion.
- the reaction mixture was stirred for 2 days at room temperature, followed by dialysis under stirring for 6 hours in methanol (Model: Spectra / Por Regenerated Cel lulose (RC) membrane, MWCO 3500, manufacturer: Spectrum Laboratories) ) was performed twice.
- methanol Model: Spectra / Por Regenerated Cel lulose (RC) membrane, MWCO 3500, manufacturer: Spectrum Laboratories
- Step 2 Preparation of G2 PAMAM-mPEGpnoo-TIPAA conjugate (2c) Dissolve in solution 2b solution (5.20 g) and TIPN (6) OL89 g, 2.90 mmol) ol DMS0 (94 mL) obtained in step 1, here (0.80 mL, 4.6 ⁇ l ol) was added. After filling the argon gas in a semi-quilter, the semi-ung mixed mixture was stirred for 2 days at room temperature, and additionally, TIPN (990 mg, 1.52) ol) was added at a time, and the semi-ung mixed mixture was then heated at room temperature. Stir for 24 hours.
- the reaction mixture was subjected to dialysis (Model: Spectra / Por Regenerated Cel lulose (RC) membrane, MWCO 8000, Spectrum Laboratories) twice with stirring for 6 hours on methanol.
- the crude product obtained by removing the solvent under reduced pressure was purified by SEC (Model: Bio-Beads S-Xl, H 40 cm x O.D. 4.5 cm, Manufacturer: Bio-Rad).
- SEC Model: Bio-Beads S-Xl, H 40 cm x O.D. 4.5 cm, Manufacturer: Bio-Rad.
- the product purified above was combined and filtered through a SEC column (Sephadex LH-20, H 40 cm x O.D.
- Iodine Content (Inductively coupled plasma mass spectrometry, I CP-MS) :
- G3 PAMAM dendrimer (3a) purchased from Aldrich was dried in vacuo to remove methanol.
- the reaction mixture was stirred at room temperature for 2 days, and then stirred for 6 hours on methanol (model name: Spectra / Por Regenerated Cel lulose (RC) membrane, MWCO 1000, manufactured by Spectrum Laboratories). Was carried out twice.
- Step 2 Preparation of G3 PAMAM-mPEG ⁇ n-TIPAA conjugate (3c) Dissolve the compound 3b solution (3.24 g) and TIPN (6) (3.86 g, 5.92 ⁇ ol) obtained in step 1 above in 100 mL DMSC To this was added 50 mL of DIEAU, 8.61 ⁇ l). After filling the reaction vessel with argon gas, the reaction mixture was stirred at room temperature for 2 days, and further TIPNU.93 g, 2.96 ⁇ ol) was added at a time, and the reaction mixture was stirred at room temperature for 24 hours.
- the reaction mixture was subjected to dialysis (Model: Spectra / Por Regenerated Cel lulose (RC) membrane, MWCO 1000, Spectrum Laboratories) twice with stirring for 6 hours on methanol.
- the crude product obtained by removing the solvent under reduced pressure was purified by SEC (Model: Bio— Beads S-Xl, H 40 cm ⁇ O.D. 4.5 cm, Manufacturer: Bio-Rad).
- SEC Model: Bio— Beads S-Xl, H 40 cm ⁇ O.D. 4.5 cm, Manufacturer: Bio-Rad.
- the product purified above was combined and filtered through a SEC column (Sephadex LH-20, H 40 cm x O.D.
- G4 PAMAM dendrimer (4a) purchased from Aldrich was dried under vacuum to remove methanol.
- the compound 4a (579 mg, 0.0407 mmol) was dissolved in DMS0 (27 mL), and mPEG carbonate compound 5 (1.94 g, 0.890 ⁇ l) was added to the stirring solution in one portion.
- the reaction mixture was stirred for 2 days at room temperature, followed by dialysis with stirring for 6 hours on methane (Model: Spectra / Por Regenerated Cel lulose (RC) membrane, MWCO 1000, manufacturer: Spectrum Laboratories) was performed twice.
- methane Model: Spectra / Por Regenerated Cel lulose (RC) membrane, MWCO 1000, manufacturer: Spectrum Laboratories
- Step 2 Preparation of G4 PAMAM-mPEGgnm-TIPAA conjugate (4c) Dissolve the compound 4b solution (1.63 g) and TIPN (6) (1.35 g, 2.07 mmol) obtained in step 1 above in DMS0 (32 mL), DIEA (0.54 mL, 3.10 t ol) was added.
- the semi-ungmul mixture was stirred at room temperature for 2 days, and additionally, TIPN (677 mg, 1.04 ⁇ l) was added at a time, and the reaction mixture was stirred at room temperature for 24 hours. .
- the reaction mixture was subjected to dialysis (Model: Spectra / Por Regenerated Cel lulose (RC) membrane, MWC0 1000, manufactured by Spectrum Laboratories) twice with stirring for 6 hours on methanol.
- the crude product obtained by removing the solvent under reduced pressure was purified by SEC (Model: Bio-Beads S-Xl, H 40 cm x O.D. 4.5 cm, Manufacturer: Bio-Rad).
- the product purified above was combined and filtered through a SEC column (Sephadex LH-20, H 40 cm x O.D. 3.0 cm) to remove the remaining DMF, and dissolved in ethanol (80 mL) for a syringe filter (Model: Puradisc 25 Syringe filter, pore size 0.45 im, PTFE, manufacturer: Whatman) sterilization-filtered and dried under vacuum for 3 days to obtain the target compound 4c (1.27 g).
- Iodine content (Induct ively coupled plasma mass spectrometry, I CP-MS) ⁇ '
- HeLa cell lines used for cytotoxicity test were purchased from American Type Culture Collection (ATCC), DMEM (Dulbecco) supplemented with 10% fetal bovine serum (FBS), 50 U / mL penicillin and 50 ⁇ g / mL streptomycin.
- s modified eagle medium (Gibco) was stored at 37 ° C and 5% carbon dioxide (C0 2 ).
- Preparation step preparation of contrast agent composition for CT
- Table 1 summarizes the viscosity (viscosity, 30 ° C) and osmolality measurement results of the CT contrast agent of the present invention.
- CT contrast agent 250 mg / mL
- CT contrast agent 250 mg / mL
- their X-ray hops were checked by preliminary inspection.
- the positive control group is "Iobrix" (Taejun Pharmaceuticals), a low-molecular-weight compound-based CT contrast agent for rapid release during vascular injection, and an emulsion nanoparticle-based veterinary CT for long-term circulation during intravascular injection.
- Contrast agent "Fenestra VC" (ART, Canada) was used.
- each 250 mL tube contains four CT contrast agents prepared in the above preparation step and 20 mM Tris complete solution (pH 7.4, 0.1 mg / mL EDTA) as "Fenestra VC", "Iobrix” and negative control. Filled in each, and also an empty tube prepared as a negative control.
- the eight tubes were placed side by side on a photographing bed of an NFR Polaris-G90 in vivo micro-CT scanner (manufacturer: NanoFocusRay), and 2D X-ray images were taken [Image parameters: 60 kVp]. ,
- FIG. 2 X-ray transmittance of all CT contrast agents compared to 20 mM Tris buffer (X-ray transmission signal strength: 1359.5) and vacuo (X-ray transmission signal strength: 1676.5) used as a negative control ( Inversely proportional to X-ray absorbance) was much lower, and among them, the rapid X-ray absorbance (Iobrix), a CT contrast medium for humans, was the highest (X-ray transmission signal strength: 198.0).
- CT contrast agents containing 2c, 3c, and 4c (X-ray transmission signal strength: 904.5, 838.0, 826.5, and 827.0 in sequence) were similar in all in vitro tube 2D X-ray tomography images. Levels of X-ray absorbance are shown. Therefore, it can be seen that the contrast agent for CT containing the radial polymer compound containing iodine according to the present invention has a striking contrast effect.
- the micro animal CT device was used as follows. Experiment was as follows.
- mice 15-22 g of 6-8 weeks old healthy male C57BL / 6 mice (Orient Bio, Korea) were used as experimental animals. Mice were not fed with water and rice from 12 hours before contrast injection to 4 hours after injection, and immediately after CT imaging after 4 hours after contrast injection.
- the mouse was placed in an induction chamber in an oxygen atmosphere containing 33 ⁇ 4 isoflurane for 5 minutes without injecting the contrast agent, followed by anesthesia, and the mouse was subjected to NFR Polaris-G90 in vivo micro-CT.
- the scanner bed (NanoFocusRay, Korea) was raised with the back facing up, and a baseline (ie precontrast) CT scan was performed while maintaining anesthesia under an oxygen atmosphere containing 1.5% isoflurane.
- Image parameters 60 kVp, 60 ⁇ , 500 ms exposure time, 360 ° scan angle, 600 scan number, 512
- si ices i.e., image number
- FOV 80.00 83.71 mm for low resolution whole-
- CT scans of each mouse were performed 1, 2, 4, 8, 24, and 48 hours after contrast injection, without additional contrast injection.
- each mouse was transferred to an individual cage in a thoroughly controlled thermo-hygrostat environment (22-25 ° C and 50-60% humidity) for recovery.
- Whole-body CT scans were performed at the time of post-infusion described above using 3-6 mice for each contrast medium, and one CT scan was performed 7 days after injection for each contrast medium. This was done to confirm that most of the contrast agent was discharged in vitro.
- CT scans on the chest, mid resolution and brain area using the 1-2 mice for CT contrast media according to the present invention were performed at the imaging parameters described above.
- the contrast effect of the CT contrast agent of the present invention at the resolution was examined.
- the raw image file obtained after each CT scan was reassembled into 1 3 ⁇ 4 of DICOMCdigital imaging and communications in medicine (512X512 pixels) file immediately after the scan to check for cross-sectional errors.
- 3D CT images were obtained from DIC0M files using Lucion software Clnfinitt Healthcare, Korea).
- Coronal and sagittal planes showing several sites of interest taken over time after injection of CT contrast agents and "Fenestra VC" and "Iobrix" used as positive controls through the tail vein of mice
- Coronal and sagittal planes showing several sites of interest taken over time after injection of CT contrast agents and "Fenestra VC" and "Iobrix" used as positive controls through the tail vein of mice
- Whole body CT cross-sectional images of (sagittal) are shown in FIGS. 3-4 (coronal plane) and 5-6 (sagittal plane), respectively.
- CT cross-sectional images of the chest, the abdomen, and the brain region (high resolution) taken over time are shown in FIGS. And abdomen) and 9-10 (brain region), respectively.
- the blood vessel region of the mouse injected with CT contrast medium appeared light gray (bones appear as a very bright white area).
- "Iobrix" a low-molecular-weight compound-based CT contrast agent for humans, used as a positive control group, can be seen to be excreted through the kidney at a very high rate within minutes (see CT scan 0 hours after injection).
- “Fenestra VC” which is an emulsion nanoparticle-based contrast agent for CT for animals, contains a contrast agent for CT containing the compound prepared in Examples 1-4 of the present invention. Similarly, until 2-4 hours after contrast injection, the biodistribution was shown in a similar manner in the early stages, such as an intravascular contrast effect of the same intensity as immediately after injection, but 24 hours after the injection, the present invention Compared with CT contrast agent, the distribution of liver is much higher than that of CT.
- the rats did not die during the CT scan by injecting the CT contrast agent of the present invention, the autopsy results after the CT scan was confirmed that no damage to the liver, kidneys and other organs. Therefore, it can be seen that the CT contrast agent containing the radial polymer compound containing iodine according to the present invention has a high contrast effect, a long contrast time, and an adequate in vitro discharge.
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/816,846 US9387267B2 (en) | 2010-08-13 | 2011-08-16 | Iodine-containing radial-shaped macromolecular compounds, preparation method thereof and contrast media compositions for CT comprising the same |
| JP2013524056A JP5674941B2 (ja) | 2010-08-13 | 2011-08-16 | ヨードを含有した放射形状の高分子化合物、その製造方法及びそれを含有するct用造影剤組成物 |
| CN201180049142.1A CN103282056B (zh) | 2010-08-13 | 2011-08-16 | 含碘放射状大分子化合物及其制备方法及包含该化合物的ct造影介质组合物 |
| EP11816662.8A EP2604289B1 (en) | 2010-08-13 | 2011-08-16 | Radial shape of polymer compound containing iodine, preparation method thereof, and ct contrast medium composition containing same |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2010-0078060 | 2010-08-13 | ||
| KR20100078060 | 2010-08-13 | ||
| KR10-2011-0080912 | 2011-08-12 | ||
| KR1020110080912A KR101334780B1 (ko) | 2010-08-13 | 2011-08-12 | 요오드를 함유한 방사형상의 고분자 화합물, 이의 제조방법 및 이를 포함하는 ct용 조영제 조성물 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2012021046A2 true WO2012021046A2 (ko) | 2012-02-16 |
| WO2012021046A3 WO2012021046A3 (ko) | 2012-05-10 |
| WO2012021046A9 WO2012021046A9 (ko) | 2012-07-05 |
Family
ID=45838548
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2011/006002 WO2012021046A2 (ko) | 2010-08-13 | 2011-08-16 | 요오드를 함유한 방사형상의 고분자 화합물, 이의 제조방법 및 이를 포함하는 ct용 조영제 조성물 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9387267B2 (ko) |
| EP (1) | EP2604289B1 (ko) |
| JP (1) | JP5674941B2 (ko) |
| KR (1) | KR101334780B1 (ko) |
| CN (1) | CN103282056B (ko) |
| WO (1) | WO2012021046A2 (ko) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013112588A1 (en) * | 2012-01-23 | 2013-08-01 | Duke University | System and method for remote image organization and analysis |
| US10898593B2 (en) * | 2014-05-13 | 2021-01-26 | Yale University | Compositions for nanoconfinement induced contrast enhancement and methods of making and using thereof |
| CN105968376B (zh) * | 2016-06-03 | 2019-02-05 | 浙江大学 | 一种树枝状大分子及其制备方法和应用 |
| KR102121606B1 (ko) | 2017-06-02 | 2020-06-10 | 경북대학교 산학협력단 | 엽산 수용체 타겟팅 종양 진단 방사성 프로브의 개발 및 응용 |
| CN107308455B (zh) * | 2017-06-27 | 2021-02-12 | 南京成至诚医药科技有限公司 | 环糊精在制备放射性碘标金丝桃类药物制剂中的应用 |
| CA3201017A1 (en) * | 2020-12-28 | 2022-07-07 | Boston Scientific Scimed, Inc. | Polysaccharides having improved radiocontrast properties |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4410688A (en) | 1981-04-29 | 1983-10-18 | Allied Corporation | Macromolecular highly branched homogeneous compound |
| EP0430863A2 (de) | 1989-11-21 | 1991-06-05 | Schering Aktiengesellschaft | Kaskadenpolymer-gebundene Komplexbildner, deren Komplexe und Konjugate, Verfahren zu ihrer Herstellung und diese enthaltende pharmazeutische Mittel |
| WO1997002051A2 (de) | 1995-07-04 | 1997-01-23 | Schering Aktiengesellschaft | Kaskaden-polymer-komplexe, verfahren zu ihrer herstellung und diese enthaltende pharmazeutische mittel |
| WO1998024775A1 (de) | 1996-12-04 | 1998-06-11 | Schering Aktiengesellschaft | Verfahren zur herstellung von metallkomplexcarbonsäureamiden |
| WO1998024774A1 (de) | 1996-12-04 | 1998-06-11 | Schering Aktiengesellschaft | Macrocyclische metallkomplexcarbonsäuren, ihre verwendung sowie verfahren zu ihrer herstellung |
| KR20060003452A (ko) | 2004-07-06 | 2006-01-11 | 주식회사 디테일베이스 | 가전기기의 광고영상 출력장치 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR970011150B1 (ko) * | 1987-10-02 | 1997-07-07 | 더 다우 케미칼 캄파니 | 스타버스트 콘쥬게이트 |
| US5871713A (en) * | 1991-12-04 | 1999-02-16 | Guerbet S.A. | Macromolecular polyamine iodine-containing compound, process for its preparation and its use as a contrast agent |
| US5342605A (en) * | 1992-05-01 | 1994-08-30 | Sterling Winthrop Inc. | Polymeric X-ray compositions containing iodinated polymeric beads |
| US5326553A (en) * | 1993-02-02 | 1994-07-05 | Sterling Winthrop Inc. | Compositions of iodophenoxy alkanes and iodophenyl ethers in film-forming materials for visualization of the gastrointestinal tract |
| GB9407812D0 (en) * | 1994-04-20 | 1994-06-15 | Nycomed Salutar Inc | Compounds |
| AU5871798A (en) * | 1997-01-29 | 1998-08-18 | Nycomed Imaging As | Polymers |
| EP2257280A4 (en) | 2008-02-26 | 2015-09-09 | Aparna Biosciences | MODIFIED ADJUSTABLE NANOPARTICLES FOR THE DELIVERY OF THERAPEUTIC SUBSTANCES, DIAGNOSTIC PRODUCTS AND EXPERIMENTAL COMPOUNDS, AND RELATED COMPOSITIONS FOR THERAPEUTIC USE |
-
2011
- 2011-08-12 KR KR1020110080912A patent/KR101334780B1/ko active IP Right Grant
- 2011-08-16 CN CN201180049142.1A patent/CN103282056B/zh not_active Expired - Fee Related
- 2011-08-16 WO PCT/KR2011/006002 patent/WO2012021046A2/ko active Application Filing
- 2011-08-16 EP EP11816662.8A patent/EP2604289B1/en not_active Not-in-force
- 2011-08-16 US US13/816,846 patent/US9387267B2/en active Active
- 2011-08-16 JP JP2013524056A patent/JP5674941B2/ja active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4410688A (en) | 1981-04-29 | 1983-10-18 | Allied Corporation | Macromolecular highly branched homogeneous compound |
| EP0430863A2 (de) | 1989-11-21 | 1991-06-05 | Schering Aktiengesellschaft | Kaskadenpolymer-gebundene Komplexbildner, deren Komplexe und Konjugate, Verfahren zu ihrer Herstellung und diese enthaltende pharmazeutische Mittel |
| US5911971A (en) | 1989-11-21 | 1999-06-15 | Schering Aktiengesellschaft | Cascade polymer bound complexing compounds for MRI |
| WO1997002051A2 (de) | 1995-07-04 | 1997-01-23 | Schering Aktiengesellschaft | Kaskaden-polymer-komplexe, verfahren zu ihrer herstellung und diese enthaltende pharmazeutische mittel |
| WO1998024775A1 (de) | 1996-12-04 | 1998-06-11 | Schering Aktiengesellschaft | Verfahren zur herstellung von metallkomplexcarbonsäureamiden |
| WO1998024774A1 (de) | 1996-12-04 | 1998-06-11 | Schering Aktiengesellschaft | Macrocyclische metallkomplexcarbonsäuren, ihre verwendung sowie verfahren zu ihrer herstellung |
| KR20060003452A (ko) | 2004-07-06 | 2006-01-11 | 주식회사 디테일베이스 | 가전기기의 광고영상 출력장치 |
Non-Patent Citations (16)
| Title |
|---|
| A. SACHSE ET AL., INVEST. RADIOL., vol. 32, 1997, pages 44 - 50 |
| A. T. YORDANOV ET AL., NANO LETT., vol. 2, 2002, pages 595 - 599 |
| B. MISSELWITZ ET AL., MAGN. RESON. MATER. PHYS. BIOL. MED., vol. 12, 2001, pages 128 - 134 |
| C. Y. KAO ET AL., ACAD. RADIOL., vol. 10, 2003, pages 475 - 483 |
| C., SUN ET AL., CHEM. EUR. J., vol. 15, 2009, pages 13341 - 13347 |
| E. SAMEI ET AL., INT. J. NANOMEDICINE, vol. 4, 2009, pages 277 - 282 |
| F. HYAFIL ET AL., NAT. MED., vol. 13, 2007, pages 636 - 641 |
| J.ZHENG ET AL., MOL. PHARMACEUTICS, vol. 6, 2009, pages 571 - 580 |
| K. NAEL ET AL., J. MAGN. RESON. IMAGING, vol. 25, 2007, pages 66 - 72 |
| M. H. OH ET AL., J. AM. CHEM. SOC., vol. 133, 2011, pages 5508 - 5515 |
| MOSMANN, T., J. IMMUNOL. METHODS, vol. 65, 1983, pages 55 - 63 |
| See also references of EP2604289A4 |
| V. P. TORCHILIN ET AL., ACAD. RADIOL., vol. 6, 1999, pages 61 - 65 |
| V. S. TRUBETSKOY ET AL., J. DRUG TARGET., vol. 4, 1997, pages 381 - 388 |
| W. KRAUSE ET AL., TOP. CURR. CHEM., vol. 210, 2000, pages 261 - 308 |
| Y. FU ET AL., BIOCONJUGATE CHEM., vol. 17, 2006, pages 1043 - 1056 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2013535560A (ja) | 2013-09-12 |
| EP2604289A4 (en) | 2015-04-29 |
| EP2604289A2 (en) | 2013-06-19 |
| CN103282056A (zh) | 2013-09-04 |
| US20130150529A1 (en) | 2013-06-13 |
| KR20120016032A (ko) | 2012-02-22 |
| CN103282056B (zh) | 2016-03-16 |
| KR101334780B1 (ko) | 2013-12-02 |
| WO2012021046A9 (ko) | 2012-07-05 |
| JP5674941B2 (ja) | 2015-02-25 |
| WO2012021046A3 (ko) | 2012-05-10 |
| US9387267B2 (en) | 2016-07-12 |
| EP2604289B1 (en) | 2017-08-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4936312B2 (ja) | 新規な両親媒性物質、それを用いた薬剤搬送システム及び分子イメージングシステム | |
| US20190105412A1 (en) | Novel molecular assembly, molecular probe for molecular imaging and molecular probe for drug delivery system using the same, and molecular imaging system and drug delivery system | |
| ES2199226T3 (es) | Polimeros biocompatibles que contienen mitades diagnosticas o terapeuticas. | |
| US8697031B2 (en) | Dual function polymer micelles | |
| JP4758346B2 (ja) | 高分子ミセル型mri造影剤 | |
| KR101334780B1 (ko) | 요오드를 함유한 방사형상의 고분자 화합물, 이의 제조방법 및 이를 포함하는 ct용 조영제 조성물 | |
| CN107789632A (zh) | 一种t7肽修饰的主动脑靶向纳米递药系统及其制备方法 | |
| US20150320890A1 (en) | Nanoparticles for brain tumor imaging | |
| WO2011058776A1 (ja) | ブロックコポリマー、ブロックコポリマー-金属錯体複合体、及びそれを用いた中空構造体キャリア | |
| EP2228074A1 (en) | A tumor targeting protein conjugate and a method for preparing the same | |
| KR20080019507A (ko) | 근적외선 형광체가 결합된 양친성 고분자의 나노 입자를포함하는 암 진단용 조영제 | |
| CN115252877A (zh) | 一种碘代聚乙二醇-聚乳酸羟基乙酸-聚赖氨酸显影多孔微球、制备方法和应用 | |
| KR102014159B1 (ko) | 요오드를 포함하는 생체적합성 단분자 담도 컴퓨터 단층촬영용 조영제 및 이의 제조방법 | |
| US20150010476A1 (en) | Rare Earth Oxide Particles and Use Thereof in Particular Imaging | |
| KR20130092330A (ko) | 요오드를 함유한 방사형상의 고분자 화합물을 함유하는 조영제 | |
| KR100943043B1 (ko) | 산화철 나노입자가 봉입된 수용성 키토산-소수성 리놀레산복합체 자기-조립 나노입자, 이의 제조방법 및 이를포함하는 간질환 진단용 조영제 | |
| Hajfathalian et al. | Polyphosphazene-Based Nanoparticles as Contrast Agents | |
| CN107337653B (zh) | 四碘甲腺原氨酸-n-羧基内酸酐、聚四碘甲腺原氨酸及其制备方法与应用 | |
| US20170266325A1 (en) | Compositions for nanoconfinement induced contrast enhancement and methods of making and using thereof | |
| JP7156665B2 (ja) | 医薬、並びにその製造方法 | |
| Jeong et al. | Facile Hydrothermal Synthesis of an Iodine-Doped Computed Tomography Contrast Agent Using Insoluble Triiodobenzene | |
| JP5320569B2 (ja) | 高分子ミセル及びそれを有効成分として含有する固形がんの診断又は治療剤 | |
| WO2015181882A1 (ja) | 分岐型両親媒性ブロックポリマーを用いた分子集合体及び薬剤搬送システム |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180049142.1 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11816662 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2013524056 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13816846 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2011816662 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011816662 Country of ref document: EP |