WO2011024989A1 - グリセリン系固形物から液化物を製造する方法 - Google Patents
グリセリン系固形物から液化物を製造する方法 Download PDFInfo
- Publication number
- WO2011024989A1 WO2011024989A1 PCT/JP2010/064673 JP2010064673W WO2011024989A1 WO 2011024989 A1 WO2011024989 A1 WO 2011024989A1 JP 2010064673 W JP2010064673 W JP 2010064673W WO 2011024989 A1 WO2011024989 A1 WO 2011024989A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- glycerin
- liquefied product
- oil
- acid
- producing
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/02—Liquid carbonaceous fuels essentially based on components consisting of carbon, hydrogen, and oxygen only
- C10L1/026—Liquid carbonaceous fuels essentially based on components consisting of carbon, hydrogen, and oxygen only for compression ignition
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G1/00—Production of liquid hydrocarbon mixtures from oil-shale, oil-sand, or non-melting solid carbonaceous or similar materials, e.g. wood, coal
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G2300/00—Aspects relating to hydrocarbon processing covered by groups C10G1/00 - C10G99/00
- C10G2300/10—Feedstock materials
- C10G2300/1011—Biomass
- C10G2300/1014—Biomass of vegetal origin
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G2300/00—Aspects relating to hydrocarbon processing covered by groups C10G1/00 - C10G99/00
- C10G2300/10—Feedstock materials
- C10G2300/1011—Biomass
- C10G2300/1018—Biomass of animal origin
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G2300/00—Aspects relating to hydrocarbon processing covered by groups C10G1/00 - C10G99/00
- C10G2300/10—Feedstock materials
- C10G2300/1037—Hydrocarbon fractions
- C10G2300/1048—Middle distillates
- C10G2300/1051—Kerosene having a boiling range of about 180 - 230 °C
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G2300/00—Aspects relating to hydrocarbon processing covered by groups C10G1/00 - C10G99/00
- C10G2300/80—Additives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G2400/00—Products obtained by processes covered by groups C10G9/00 - C10G69/14
- C10G2400/04—Diesel oil
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E50/00—Technologies for the production of fuel of non-fossil origin
- Y02E50/10—Biofuels, e.g. bio-diesel
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P30/00—Technologies relating to oil refining and petrochemical industry
- Y02P30/20—Technologies relating to oil refining and petrochemical industry using bio-feedstock
Definitions
- the present invention relates to a method for producing a liquefied product by liquefying a glycerin solid.
- Biodiesel fuel (BDF) is obtained by utilizing an esterification reaction by adding alcohol and sodium hydroxide to vegetable oil such as jatropha oil, sunflower oil, rapeseed oil, olive oil and the like, and using this. At this time, glycerin is also produced as a byproduct (byproduct) together with the biodiesel fuel (see Patent Document 1). By-product glycerin left after biodiesel fuel (BDF) is separated from the product is mixed with BDF, ethanol, sodium hydroxide, etc. It was thrown away as a thing.
- the present invention has been made in view of such a technical background, and can be used to efficiently liquefy a glycerin solid to produce a liquefied material that can be used, for example, as a fuel. It aims at providing the manufacturing method of a thing.
- the present invention provides the following means.
- Glycerin-based solids produced as a by-product when producing biodiesel fuel from vegetable oil At least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil;
- a glycerin system characterized by producing a liquefied product by mixing a glycerin solid containing glycerin and an acid, and mixing the acid so that the liquefied product has a pH of 3 to 12.
- a method for producing a liquefied product from a solid characterized by producing a liquefied product by mixing a glycerin solid containing glycerin and an acid, and mixing the acid so that the liquefied product has a pH of 3 to 12.
- a glycerin solid containing glycerin An oxidizing agent, A liquefied product is produced by mixing at least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil, and the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12.
- a method for producing a liquefied product from a glycerin solid is
- a glycerin solid that is produced as a by-product when producing biodiesel fuel from vegetable oil;
- An oxidizing agent A liquefied product is produced by mixing at least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil, and the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12.
- a liquefied product is produced by mixing a glycerin solid containing glycerin and an oxidizing agent, and the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12.
- a method for producing a liquefied product from a glycerin solid is produced by mixing a glycerin solid containing glycerin and an oxidizing agent, and the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12.
- An oxidizing agent At least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil;
- a glycerin solid that is produced as a by-product when producing biodiesel fuel from vegetable oil;
- An oxidizing agent At least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil;
- the glycerin solid and the oxidizing agent are first mixed, then oil is mixed, and then the acid is mixed. How to manufacture.
- a liquefied product is produced by mixing a glycerin solid containing glycerin, an oxidizing agent and an acid, and the acid is mixed so that the liquefied product has a pH of 3 to 12.
- a method for producing a liquefied product from a glycerin solid is produced by mixing a glycerin solid containing glycerin, an oxidizing agent and an acid, and the acid is mixed so that the liquefied product has a pH of 3 to 12.
- [20] A method for producing a liquefied product from the glycerin solid according to any one of items 14 to 19, wherein acetic acid is used as the acid.
- a liquid glycerin-containing substance containing glycerin and an acid are mixed to produce a liquefied product having a freezing point lower than the freezing point of the liquid glycerin-based substance, and the liquefied product has a pH of 3 to 12.
- the said acid is mixed so that the manufacturing method of the liquefied material characterized by the above-mentioned.
- a liquid glycerin-containing substance containing glycerin and an oxidizing agent are mixed to produce a liquefied product having a freezing point lower than the freezing point of the liquid glycerin-based substance, and the liquefied product is adjusted to pH 3 to pH 12.
- a method for producing a liquefied product comprising mixing the oxidizing agent as described above.
- a liquid glycerin-containing substance containing glycerin, an acid, and an oxidizing agent are mixed to produce a liquefied product having a freezing point lower than the freezing point of the liquid glycerin-based substance.
- a method for producing a liquefied product comprising mixing the acid so as to have a pH of 3 to 12.
- At least one oil selected from the group consisting of glycerin-based solids (including solids composed only of glycerin), mineral oils, animal oils and vegetable oils, and acids, are mixed, so that the solid can be liquefied, that is, a liquefied product can be obtained. Further, since the acid is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify even after a lapse of time, and the liquefied product is stably in a liquid state over a long period of time. Maintained. The obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- an acid is mixed with a glycerin solid that is produced as a by-product when producing biodiesel fuel from vegetable oil, at least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil. Therefore, the solid can be liquefied, that is, a liquefied product can be obtained. Further, since the acid is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify even after a lapse of time, and the liquefied product is stably in a liquid state over a long period of time. Maintained. The obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel. Moreover, since the raw material glycerin-based solid is produced as a by-product when producing biodiesel fuel from vegetable oil, and it is used effectively, it can promote effective use of resources.
- the glycerin solid and the oil are mixed so that the content of the glycerin solid with respect to the total volume of the glycerin solid and the oil is 50 to 90% by volume. Since 2 to 10 parts by volume of the acid is mixed with respect to 100 parts by volume in total, glycerin can be sufficiently liquefied while being used in a high content ratio.
- the solid can be liquefied, that is, a liquefied product can be obtained.
- the acid is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify even after a lapse of time, and the liquefied product is stably in a liquid state over a long period of time. Maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- Can be liquefied that is, a liquefied product can be obtained.
- the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify over time, and the liquefied product is stably in a liquid state over a long period of time. Is maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify over time, and the liquefied product is stably in a liquid state over a long period of time. Is maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- the raw material glycerin-based solid is produced as a by-product when producing biodiesel fuel from vegetable oil, and it is used effectively, it can promote effective use of resources.
- the solid can be liquefied, that is, a liquefied product can be obtained.
- the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify over time, and the liquefied product is stably in a liquid state over a long period of time. Is maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- the acid are mixed, so that the solid can be liquefied, that is, a liquefied product can be obtained.
- the acid and the oxidizing agent are mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify over time, and the liquefied product is stable and liquid over a long period of time. State is maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- the glycerin solid and the oxidizing agent are first mixed, then the oil is further mixed, and then the acid is mixed, so that liquefaction can be efficiently performed.
- the solid can be liquefied, that is, a liquefied product can be obtained.
- the acid and the oxidizing agent are mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify over time, and the liquefied product is stable over a long period of time. A liquid state is maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- the acid is mixed so that the liquefied product has a pH of 4 to 11, so that the obtained liquefied product is stably maintained in a liquid state for a longer period of time.
- one or two or more kinds of petroleum selected from the group consisting of kerosene, light oil and heavy oil are used as the petroleum, and these are more flammable. Combustion performance can be further improved.
- the liquefied material obtained by the mixing is cooled to a temperature of 30 ° C. or lower, and then the precipitate is filtered to obtain a filtrate, and then the filtrate is distilled at a distillation temperature of 150 ° C. to 230 ° C. Since distillation is performed, colored components and the like in the liquefied product can be removed, and an almost colorless or colorless transparent liquefied product can be produced.
- the liquefied material obtained by the mixing is cooled to a temperature of 30 ° C. or lower, and then the precipitate is filtered to obtain a filtrate, and the filtrate is cooled to a temperature of 5 ° C. or lower to obtain the filtrate. Since a supernatant liquid is obtained from the cooling liquid, and then the supernatant liquid is distilled at a distillation temperature of 150 ° C. to 230 ° C., the colored components in the liquefied product can be sufficiently removed, and the almost colorless or colorless transparent liquefied product Can be manufactured.
- a liquefied product having a freezing point lower than the freezing point of the liquid glycerin-based substance can be produced.
- a liquefied product having a freezing point of ⁇ 30 ° C. can be produced from a liquid glycerin material having a freezing point of ⁇ 5 ° C.
- the acid is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify even after a lapse of time, and the liquefied product is stably in a liquid state over a long period of time. Maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- a liquefied product having a freezing point lower than the freezing point of the liquid glycerin-based substance can be produced.
- a liquefied product having a freezing point of ⁇ 30 ° C. can be produced from a liquid glycerin material having a freezing point of ⁇ 5 ° C.
- the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify over time, and the liquefied product is stably in a liquid state over a long period of time. Is maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- a liquefied product having a freezing point lower than the freezing point of the liquid glycerin-based substance can be produced.
- a liquefied product having a freezing point of ⁇ 30 ° C. can be produced from a liquid glycerin material having a freezing point of ⁇ 5 ° C.
- the acid and the oxidizing agent are mixed so that the liquefied product has a pH of 3 to 12, the liquefied product does not re-solidify over time, and the liquefied product is stable and liquid over a long period of time. State is maintained.
- the obtained liquefied product is very useful because it has excellent combustion performance and can be used, for example, as a fuel.
- the method for producing a liquefied product from a glycerin solid according to the first invention includes a glycerin solid containing glycerin, at least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil, and an acid. And the acid is mixed so that the liquefied product has a pH of 3 to 12.
- the method for producing a liquefied product from a glycerin solid according to the second invention is at least one selected from the group consisting of a glycerin solid containing glycerin, an oxidizing agent, mineral oil, animal oil and vegetable oil.
- a liquefied product is produced by mixing with an oil, and the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12.
- the method for producing a liquefied product from a glycerin solid according to the third invention is a glycerin solid containing glycerin, an oxidizing agent, and at least one selected from the group consisting of mineral oil, animal oil and vegetable oil.
- a liquefied product is produced by mixing oil and an acid, and the acid is mixed so that the liquefied product has a pH of 3 to 12.
- the glycerin-based solid includes a solid (including a viscous jelly-like material), glycerin, and other components (for example, water) Examples thereof include glycerin-based solids (including viscous jelly-like materials) containing sodium oxide and the like.
- Glycerin-based solids produced as a by-product (by-product) when producing biodiesel fuel from vegetable oils have not been used in the past because they are solid, and are waste.
- vegetable oils jatropha oil, sunflower oil, rapeseed oil, olive oil, etc.
- this when this is used as the glycerin-based solid (when liquefied), it can be used effectively, so resources can be used effectively.
- Glycerin is a viscous jelly-like property at room temperature, and at least does not become solid, but it is a by-product when producing biodiesel fuel from the above vegetable oil. As a result, the glycerin-containing material is solidified.
- biodiesel fuel BDF
- BDF biodiesel fuel
- the by-product glycerin remaining after separation of the biodiesel fuel from the product containing the biodiesel fuel is considered to contain free fatty acids and sodium hydroxide. It is presumed that, however, the reason for becoming a solid is not certain at present.
- the oil at least one oil selected from the group consisting of mineral oil, animal oil and vegetable oil is used.
- mineral oil For example, petroleum etc. are mentioned.
- animal oil For example, cow oil, pig oil, bird oil etc. are mentioned.
- vegetable oil For example, jatropha oil, sunflower oil, rapeseed oil, olive oil, coconut oil etc. are mentioned.
- petroleum has good combustibility, the liquefied product combustion performance can be further improved.
- the oil is not particularly limited, and examples thereof include kerosene, light oil, heavy oil, gasoline, BDF (biodiesel fuel), and kerosene (jet fuel, rocket fuel, etc.).
- kerosene a kind of petroleum selected from the group consisting of kerosene, light oil and heavy oil. Since these have good combustibility, the combustion performance of the liquefied product can be further improved.
- kerosene a kind of petroleum selected from the group consisting of kerosene, light oil and heavy oil.
- the combustion performance of the liquefied product can be further improved and the residue in the liquefied product can be reduced.
- light oil is preferred next to kerosene, and even in the case of light oil, the residue can be reduced to some extent.
- the oil is added in the solvent sense for the liquefaction reaction.
- the oil itself has good combustibility, there is an advantage that the combustion performance of the obtained liquefied product is not hindered.
- the following manufacturing method may be employed as a modification of the first, second, and third inventions.
- a glycerin-containing solid material containing glycerin is heated to form a liquid material, and then the liquid material is mixed with an acid to produce a liquefied material.
- the acid is mixed so as to have a pH of 12.
- a glycerin-containing solid material containing glycerin is heated to a liquid material, and then the liquid material and an oxidizing agent are mixed to produce a liquefied material.
- the oxidizing agent is mixed so as to have a pH of ⁇ 12.
- a glycerin-containing solid material containing glycerin is heated to form a liquid material, and then the liquid material, an oxidizing agent, and an acid are mixed to produce a liquefied material.
- the acid is mixed so that the pH is 3 to 12.
- inorganic acids such as hydrochloric acid and a sulfuric acid
- hydrochloric acid it is preferable to use an inorganic acid.
- liquefaction can be promoted.
- hydrochloric acid is particularly preferred.
- the mixing amount of acid and the concentration of acid used are set so that the liquefied product has a pH of 3 to 12.
- the alkalinity becomes strong beyond pH 12 the liquefied liquefied material is solidified again with time.
- the content of the glycerin solid is 50 to 90% by volume relative to the total volume of the glycerin solid and the oil (in other words, the oil content is 10 to 50% by volume).
- hydrochloric acid is used as the acid
- hydrochloric acid is mixed in a volume equivalent to 2 to 10 parts by volume in terms of 35% by mass of concentrated hydrochloric acid
- sulfuric acid is used as the acid
- sulfuric acid is converted to 90% by mass of concentrated sulfuric acid.
- the volume parts corresponding to 2 to 10 volume parts are mixed.
- the content of glycerin solids relative to the total volume of glycerin solids and oil is more preferably 60 to 85% by volume. Further, it is more preferable to mix 3 to 8 parts by volume of the acid with respect to 100 parts by volume in total of the glycerin solid and the oil.
- the mixed liquid in which the glycerin solid, oil and acid are mixed may be liquefied while being maintained at ordinary temperature, or may be liquefied by heating.
- the mixed solution is preferably liquefied while stirring.
- the oxidizing agent is not particularly limited, and examples thereof include hydrogen peroxide and ozone. Among them, it is preferable to use hydrogen peroxide as the oxidizing agent.
- hydrogen peroxide is used as the oxidizing agent, a liquefied product can be obtained with a high yield, that is, a liquefied product having a high combustion calorie can be obtained.
- the mixing amount of the oxidizing agent and the concentration of the oxidizing agent solution are set so that the liquefied product has a pH of 3 to 12.
- the alkalinity becomes strong beyond pH 12 the liquefied liquefied material is solidified again with time.
- it is easy to corrode containers such as a tank which mixes, on the acidic side from pH3.
- the volume of the oxidizer converted into an oxidizer solution having a concentration of 35% by mass with respect to 100 parts by volume of the glycerin-based solid is 0.1 to 20 parts by volume, and the oil is 0.1 to It is preferable to mix 40 parts by volume.
- glycerin can be sufficiently liquefied while being used in a high content ratio.
- the oil is further mixed after mixing the glycerin-based solid and the oxidizing agent before the mixing. In this case, liquefaction can be performed efficiently.
- the mixed liquid in which the glycerin solid, the oxidant and the oil are mixed may be liquefied while being maintained at ordinary temperature, or may be liquefied by heating.
- the mixed solution is preferably liquefied while stirring.
- the oxidizing agent is not particularly limited, and examples thereof include hydrogen peroxide and ozone. Among them, it is preferable to use hydrogen peroxide as the oxidizing agent.
- hydrogen peroxide is used as the oxidizing agent, a liquefied product can be obtained with a high yield, that is, a liquefied product having a high combustion calorie can be obtained.
- the acid is not particularly limited, and examples thereof include organic acids such as acetic acid and formic acid, and inorganic acids such as hydrochloric acid and sulfuric acid. Of these, organic acids such as acetic acid and formic acid are preferably used. In this case, there is an advantage that the kinematic viscosity can be lowered. Of the organic acids, acetic acid is particularly preferred.
- the mixing amount of the acid and the concentration of the acid used are set so that the liquefied product has a pH of 3 to 12.
- the alkalinity becomes strong beyond pH 12 the liquefied liquefied material is solidified again with time.
- the volume of the oxidizer converted into an oxidizer solution having a concentration of 5% by mass with respect to 100 parts by volume of the glycerin-based solid is 0.1 to 20 parts by volume, and the oil is 0.1 to It is preferable to mix 40 parts by volume and 0.1 to 10 parts by volume of acid.
- glycerin can be sufficiently liquefied while being used in a high content ratio.
- glycerin-based solid it is preferable to mix the glycerin-based solid and the oxidizing agent before mixing the oil, and then mix the oil and then the acid. In this case, liquefaction can be performed efficiently.
- the mixed liquid obtained by mixing the glycerin solid, the oxidizing agent, the oil and the acid may be liquefied while being maintained at ordinary temperature, or may be liquefied by heating.
- the mixed solution is preferably liquefied while stirring.
- the liquefied material obtained as described above can obtain a high temperature with a small amount of fuel at the time of combustion, and can be suitably used as, for example, a liquid fuel because of its excellent combustion performance.
- the liquefied product can be used for fuel as a mixed liquid in which oil such as petroleum (kerosene, light oil, heavy oil, etc.) is mixed.
- the liquefied product can be used as a liquid fuel as it is, but it may be used as a liquid fuel by removing solids by filtration and separating and removing water with a centrifuge.
- the liquefied product also contributes to environmental protection because it emits less NOx (nitrogen oxide), CO (carbon monoxide), CO 2 (carbon dioxide), SO 2 (sulfur dioxide), etc. during combustion. it can.
- NOx nitrogen oxide
- CO carbon monoxide
- CO 2 carbon dioxide
- SO 2 sulfur dioxide
- the liquefied material obtained as described above is cooled to a temperature of 30 ° C. or lower, and then the precipitate is filtered to obtain a filtrate, which is then distilled at a distillation temperature of 150 ° C. to 230 ° C. It is preferable to obtain a distillate by distillation.
- the distilled liquefied product thus obtained is free from colored components in the liquefied product, and an almost colorless or colorless transparent liquefied product can be produced.
- the distilled liquefied product can be suitably used as, for example, a liquid fuel.
- the liquefied product obtained as described above is cooled to a temperature of 30 ° C. or lower, and then a precipitate is filtered to obtain a filtrate.
- the filtrate is cooled to a temperature of 5 ° C. or lower ( ⁇ 5 ° C. It is preferable to cool to below, more preferably to -25 ° C to -10 ° C) to obtain a supernatant liquid, and then the supernatant liquid is distilled at a distillation temperature of 150 ° C to 230 ° C. It is particularly preferable to obtain a distilled liquefied product.
- the distilled liquefied product thus obtained has sufficiently removed the color components and the like in the liquefied product, and an almost colorless or colorless transparent liquefied product can be produced.
- the distilled liquefied product can be suitably used as, for example, a liquid fuel.
- the filtration of the precipitate can be performed using, for example, a filtration filter, a centrifuge, or the like.
- the distillation temperature is more preferably set to 160 ° C. to 200 ° C.
- the said distillation temperature exceeds 230 degreeC, since a coloring component will be partly distilled and it will become easy to distill, it is unpreferable.
- the said manufacturing method is applicable also to the liquid glycerin type substance formed by containing glycerin.
- a liquefied product having a freezing point lower than the freezing point of the liquid glycerin material can be produced.
- a liquefied product having a freezing point lower than the freezing point of the liquid glycerin material is produced by mixing a liquid glycerin material containing glycerin and an acid. And the acid is mixed so that the liquefied product has a pH of 3 to 12.
- the second method for producing a liquefied product of the present invention comprises mixing a glycerin-containing liquid glycerin substance and an oxidant to obtain a liquefied product having a freezing point lower than the freezing point of the liquid glycerin substance. And the oxidizing agent is mixed so that the liquefied product has a pH of 3 to 12.
- the third production method of the liquefied product of the present invention has a freezing point lower than the freezing point of the liquid glycerin substance by mixing a liquid glycerin substance containing glycerin, an acid, and an oxidizing agent. A liquefied product is produced, and the acid is mixed so that the liquefied product has a pH of 3 to 12.
- the first to third production methods are the production methods relating to the reaction steps in the first to third inventions (that is, after the glycerin solid is heated to become a liquid glycerin substance). It is also what prescribes the manufacturing method).
- a liquid substance can be used at room temperature (25 ° C.), or a solid glycerin solid at room temperature (25 ° C.) containing glycerin.
- a liquefied product can be used by heating (preferably heating temperature is 40 ° C. to 80 ° C .; particularly preferable heating temperature is 40 ° C. to 60 ° C.).
- Biodiesel fuel (fatty acid methyl ester) is prepared by mixing 75 parts by weight of vegetable oil obtained from Jatropha, 20 parts by weight of methanol, and 5 parts by weight of sodium hydroxide and holding it at room temperature for 8 hours while stirring. Etc.) was obtained. In this product, the content of biodiesel fuel was about 75% by mass, and the content of byproduct glycerin solids (solids containing glycerin) was about 25% by mass. Biodiesel fuel was separated from the product by precipitation separation.
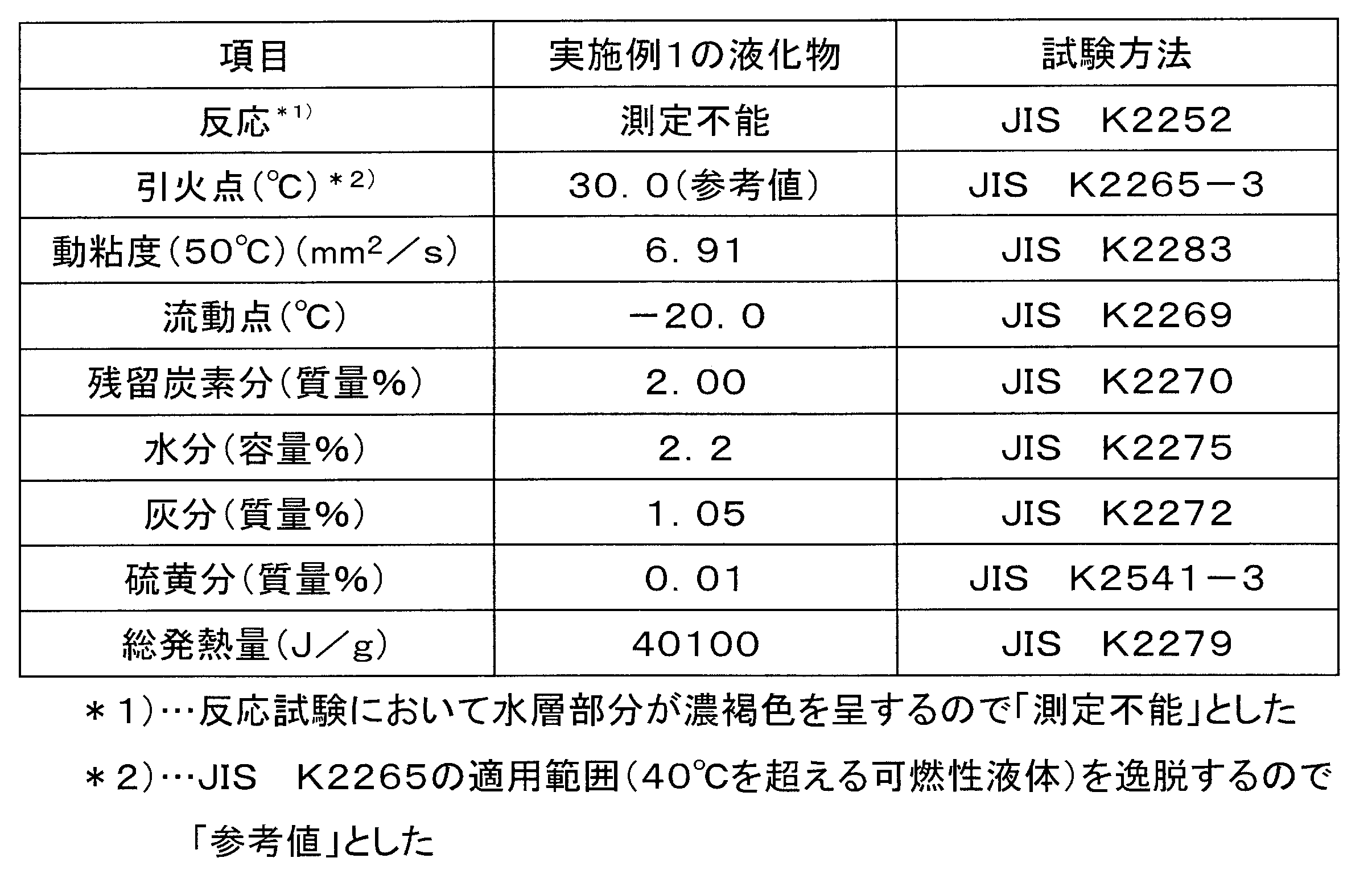
- the glycerin solid (about pH 14) remaining after the separation was liquefied. That is, first, 50 mL of concentrated hydrochloric acid (35% by mass concentrated hydrochloric acid) is mixed with 300 mL of kerosene in advance to obtain a mixture, and this mixture is mixed with 700 mL of the glycerin solid and stirred at room temperature for 30 minutes. Obtained. The obtained liquefied product was pH 7.
- Example 2 The glycerin solid (about pH 14) obtained in Example 1 was liquefied by the following method. First, 50 mL of concentrated hydrochloric acid (35% by mass concentrated hydrochloric acid) was mixed with 200 mL of light oil in advance to obtain a mixture, and this mixture was mixed with 800 mL of the glycerin solid and stirred at room temperature for 30 minutes to obtain a liquefied product. . The obtained liquefied product was pH 7.
- concentrated hydrochloric acid 35% by mass concentrated hydrochloric acid
- Example 3 The glycerin solid (about pH 14) obtained in Example 1 was liquefied by the following method. First, 50 mL of concentrated hydrochloric acid (35% by mass concentrated hydrochloric acid) was mixed with 200 mL of heavy oil in advance to obtain a mixture, and this mixture was mixed with 800 mL of the glycerin solid and stirred at room temperature for 30 minutes to obtain a liquefied product. . The obtained liquefied product was pH 7.
- concentrated hydrochloric acid 35% by mass concentrated hydrochloric acid
- Example 4 The glycerin solid (about pH 14) obtained in Example 1 was liquefied by the following method. First, 20 mL of concentrated hydrochloric acid (35% by mass concentrated hydrochloric acid) was mixed in advance with 300 mL of kerosene to obtain a mixture, and this mixture was mixed with 700 mL of the glycerin solid and stirred at room temperature for 30 minutes to obtain a liquefied product. . The obtained liquefied product was pH 10.
- concentrated hydrochloric acid 35% by mass concentrated hydrochloric acid
- Example 5 The glycerin solid (about pH 14) obtained in Example 1 was liquefied by the following method. First, 80 mL of concentrated hydrochloric acid (35% by mass concentrated hydrochloric acid) was mixed in advance with 300 mL of kerosene to obtain a mixture, and this mixture was mixed with 700 mL of the glycerin solid and stirred at room temperature for 30 minutes to obtain a liquefied product. . The obtained liquefied product was pH 5.
- concentrated hydrochloric acid 35% by mass concentrated hydrochloric acid
- Example 6 The glycerin solid (about pH 14) obtained in Example 1 was liquefied by the following method. First, 10 mL of concentrated sulfuric acid (90% by mass concentrated sulfuric acid) was mixed in advance with 300 mL of kerosene to obtain a mixture, and this mixture was mixed with 700 mL of the glycerin solid and stirred at room temperature for 30 minutes to obtain a liquefied product. . The obtained liquefied product was pH 8.
- Example 1 The glycerin solid (about pH 14) obtained in Example 1 was liquefied by the following method. First, 5 mL of concentrated hydrochloric acid (35% by mass concentrated hydrochloric acid) was mixed with 300 mL of kerosene in advance to obtain a mixture, and this mixture was mixed with 700 mL of the glycerin solid and stirred at room temperature for 30 minutes to obtain a liquefied product. . The obtained liquefied product was pH 13.
- concentrated hydrochloric acid 35% by mass concentrated hydrochloric acid
- Example 8> The liquefied product (pH 7, brown transparent liquid) obtained in Example 1 was further subjected to the following post-treatment. That is, the liquefied product (brown clear liquid) obtained in Example 1 was cooled to 20 ° C. and held at 20 ° C. for 1440 minutes, and then the precipitate present in the liquefied product at 20 ° C. was physically filtered with a filter. Thus, a filtrate was obtained, and then the filtrate was distilled at a distillation temperature of 200 ° C. to obtain a colorless and transparent distillate (distilled liquefied product).
- Example 9 Liquefaction of the glycerin solid obtained in Example 1 (about pH 14, freezing point 25 ° C.) was performed by the following method. A mixture of 700 mL of the glycerin-based solid and 30 mL of hydrogen peroxide (5% by mass hydrogen peroxide solution) was stirred at 60 ° C. for 20 minutes, then 300 mL of kerosene was added, and the mixture was stirred at 60 ° C. for 10 minutes. Further, 10 mL of acetic acid (purity 99% by mass) was added and stirred at 60 ° C. for 10 minutes to obtain a liquefied product (pH 7, brown transparent liquid).
- hydrogen peroxide 5% by mass hydrogen peroxide solution
- the liquefied product (brown transparent liquid) is cooled to 20 ° C. and held at 20 ° C. for 30 minutes, and then a precipitate present in the liquefied product at 20 ° C. is physically filtered with a filter to obtain a filtrate.
- the filtrate is cooled to ⁇ 10 ° C. and held at ⁇ 10 ° C. for 180 minutes, and then a supernatant is obtained from the coolant, and then the supernatant is distilled at a distillation temperature of 200 ° C. (Distilled liquefied product) was obtained.
- the thus obtained distilled liquefied product has a freezing point of ⁇ 30 ° C. or lower (not solidified at least at ⁇ 30 ° C.).
- Example 10 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 9 except that 300 mL of heavy oil was used instead of 300 mL of kerosene.
- Example 11 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 9 except that 300 mL of light oil was used instead of 300 mL of kerosene.
- Example 12 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 9 except that the amount of hydrogen peroxide (5% by mass hydrogen peroxide solution) was set to 10 mL.
- Example 13 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 9 except that the amount of hydrogen peroxide (5% by mass hydrogen peroxide solution) was set to 80 mL.
- Example 14 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 9 except that the amount of acetic acid used was set to 5 mL.
- Example 15 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 9 except that the amount of acetic acid used was set to 40 mL.
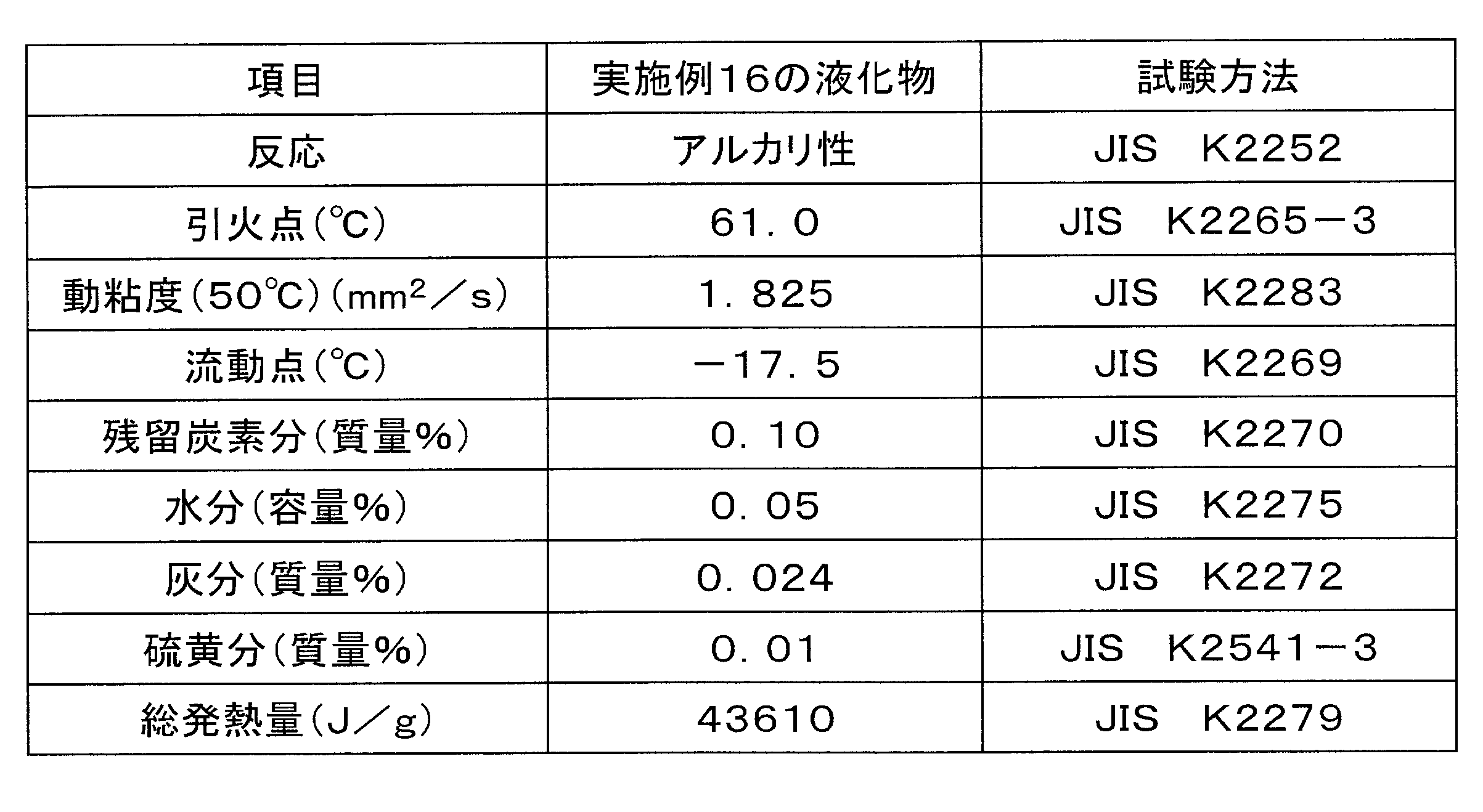
- Example 16 The glycerin solid (about pH 14) obtained in Example 1 was liquefied by the following method. A mixture of 700 mL of the glycerin solid and 30 mL of hydrogen peroxide (35% by mass hydrogen peroxide) was stirred at 60 ° C. for 10 minutes, and then 300 mL of kerosene was added and stirred at 60 ° C. for 10 minutes. As a result, a liquefied product (pH 7, clear brown liquid) was obtained.
- hydrogen peroxide 35% by mass hydrogen peroxide
- the liquefied product (brown transparent solution) is cooled to 20 ° C. and held at 20 ° C. for 60 minutes, and then the precipitate present in the liquefied product at 20 ° C. is physically filtered with a filter to obtain a filtrate.
- the filtrate is cooled to ⁇ 10 ° C. and held at ⁇ 10 ° C. for 1440 minutes, and then a supernatant is obtained from the cooling liquid, and then the supernatant is distilled at a distillation temperature of 200 ° C. (Distilled liquefied product) was obtained.
- Example 17 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 16 except that 300 mL of heavy oil was used instead of 300 mL of kerosene.
- Example 18 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 16 except that 300 mL of light oil was used instead of 300 mL of kerosene.
- Example 19 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 16 except that the amount of hydrogen peroxide (35 mass% hydrogen peroxide solution) was set to 10 mL.
- Example 20 A colorless and transparent distillate (distilled liquefied product) was obtained in the same manner as in Example 16 except that the amount of hydrogen peroxide (35% by mass hydrogen peroxide solution) was set to 70 mL.
- Example 21 Liquefaction of the glycerin solid obtained in Example 1 (about pH 14, freezing point 25 ° C.) was performed by the following method. 700 mL of the glycerin solid was heated to 60 ° C. to make a liquid, and then a mixture in which 10 mL of hydrogen peroxide (5 mass% hydrogen peroxide solution) was mixed with the liquid was stirred at 60 ° C. for 20 minutes. Thereafter, 20 mL of acetic acid (purity 99% by mass) was further added, followed by stirring at 60 ° C. for 10 minutes to obtain a liquefied product (pH 5, clear brown liquid).
- hydrogen peroxide 5 mass% hydrogen peroxide solution
- the liquefied product (brown transparent liquid) is cooled to 20 ° C. and held at 20 ° C. for 30 minutes, and then a precipitate present in the liquefied product at 20 ° C. is physically filtered with a filter to obtain a filtrate.
- the filtrate is cooled to ⁇ 10 ° C. and held at ⁇ 10 ° C. for 180 minutes, and then a supernatant is obtained from the coolant, and then the supernatant is distilled at a distillation temperature of 200 ° C. (Distilled liquefied product) was obtained.
- the thus obtained distilled liquefied product has a freezing point of ⁇ 30 ° C. or lower (not solidified at least at ⁇ 30 ° C.).
- Example 22 A mixture of 700 mL of a liquid glycerin-based material (freezing point—5 ° C.) mixed with 10 mL of hydrogen peroxide (5 mass% hydrogen peroxide solution) was stirred at 60 ° C. for 20 minutes, and then 300 mL of kerosene was added at 60 ° C. The mixture was stirred for 10 minutes, and 20 mL of acetic acid (purity 99% by mass) was further added, followed by stirring at 60 ° C. for 10 minutes to obtain a liquefied product (pH 5, brown transparent liquid).
- hydrogen peroxide 5 mass% hydrogen peroxide solution
- the liquefied product (brown transparent liquid) is cooled to 20 ° C. and held at 20 ° C. for 30 minutes, and then a precipitate present in the liquefied product at 20 ° C. is physically filtered with a filter to obtain a filtrate.
- the filtrate is cooled to ⁇ 10 ° C. and held at ⁇ 10 ° C. for 180 minutes, and then a supernatant is obtained from the coolant, and then the supernatant is distilled at a distillation temperature of 200 ° C. (Distilled liquefied product) was obtained.
- the thus obtained distilled liquefied product has a freezing point of ⁇ 30 ° C. or lower (not solidified at least at ⁇ 30 ° C.).
- Example 7 the combustion performance of the liquefied material obtained in Example 1 was evaluated.
- the liquefied material obtained in Example 1 was put into a boiler and the combustion performance was evaluated.
- the combustion performance of A heavy oil was also evaluated.
- the combustion performance of a mixed liquid (Example 7) obtained by mixing 50 parts by volume of A heavy oil with 50 parts by volume of the liquefied product of Example 1 was also evaluated.
- the liquefied product of Example 1 obtained by the production method of the present invention has a high temperature with a small amount of fuel compared to the A heavy oil of the reference example, and the combustion performance is improved. Are better. Further, the liquefied product of Example 1 is NOx (nitrogen oxide), CO (carbon monoxide), CO 2 (carbon dioxide), SO 2 (sulfur dioxide) during combustion, as compared with the heavy oil A of the reference example. Can contribute to environmental protection.
- NOx nitrogen oxide
- CO carbon monoxide
- CO 2 carbon dioxide
- SO 2 sulfur dioxide
- the mixed liquid of Example 7 has a high temperature with a small amount of fuel and is excellent in combustion performance as compared with the heavy oil A of the reference example. Further, the mixture of Example 7, compared to the A heavy oil of Reference Example, NOX during combustion (nitrogen oxides), CO (carbon monoxide), CO 2 (carbon dioxide), SO 2 (sulfur dioxide) Can contribute to environmental protection.
- Example 1 The analysis result of the liquefied product obtained in Example 1 is shown in Table 4, the analysis result of the liquefied product obtained in Example 4 is shown in Table 5, and the analysis result of the liquefied product obtained in Example 5 is shown in Table 5.
- Table 7 shows the analysis results of the liquefied product (distilled liquefied product) obtained in Example 9
- Table 8 shows the analysis results of the liquefied product (distilled liquefied product) obtained in Example 10.
- Table 9 shows the analysis results of the liquefied product (distilled liquefied product) obtained in Example 16.
- Example 21 the total calorific value of the liquefied product obtained in Example 21 was about 23500 J / g.
- the liquefied product produced by the production method of the present invention is excellent in combustion performance, and is therefore preferably used as, for example, a liquid fuel, but is not particularly limited to such use.
Abstract
Description
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸化剤を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸化剤を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
第1、第2、第3発明において、前記グリセリン系固形物としては、グリセリンのみからなる固形物(粘稠なゼリー状物を含む)、グリセリンを含有すると共にグリセリン以外の他の成分(例えば水酸化ナトリウム等)を含有してなるグリセリン系固形物(粘稠なゼリー状物を含む)等が挙げられる。
前記酸としては、特に限定されるものではないが、例えば塩酸、硫酸等の無機酸を例示できる。中でも、無機酸を用いるのが好ましく、この場合には液化を促進することができる。前記無機酸の中でも塩酸が特に好適である。
前記酸化剤としては、特に限定されるものではないが、例えば過酸化水素、オゾン等が挙げられる。中でも、前記酸化剤としては過酸化水素を用いるのが好ましい。酸化剤として過酸化水素を用いた場合には、高い収率で液化物を得ることができる、即ち燃焼カロリーの高い液化物を得ることができる。
前記酸化剤としては、特に限定されるものではないが、例えば過酸化水素、オゾン等が挙げられる。中でも、前記酸化剤としては過酸化水素を用いるのが好ましい。酸化剤として過酸化水素を用いた場合には、高い収率で液化物を得ることができる、即ち燃焼カロリーの高い液化物を得ることができる。
上記のようにして得られた液化物は、その燃焼時において、少ない燃料量で高い温度が得られるものであり、このように燃焼性能に優れているので、例えば液体燃料として好適に利用できる。また、前記液化物を石油(灯油、軽油、重油等)等の油と混合した混合液として燃料に利用することもできる。前記液化物は、そのまま液体燃料として利用できるが、濾過により固形分を除去し、遠心分離器で水分を分離除去して液体燃料として利用しても良い。
ジャトロファから得られた植物油75質量部、メタノール20質量部、水酸化ナトリウム5質量部を混合し、攪拌しながら常温で8時間保持することによってエステル化反応を行わせてバイオディーゼル燃料(脂肪酸メチルエステル等)を含む生成物を得た。この生成物において、バイオディーゼル燃料の含有率は約75質量%であり、副生グリセリン系固形物(グリセリンを含有してなる固形物)の含有率は約25質量%であった。前記生成物から沈殿分離法によりバイオディーゼル燃料を分離した。
前記実施例1で得られたグリセリン系固形物(約pH14)の液化を次の手法で行った。まず予め軽油200mLに濃塩酸(35質量%濃塩酸)を50mL混合して混合物を得、この混合物を前記グリセリン系固形物800mLに混合して常温で30分間攪拌を行うことによって液化物を得た。得られた液化物はpH7であった。
前記実施例1で得られたグリセリン系固形物(約pH14)の液化を次の手法で行った。まず予め重油200mLに濃塩酸(35質量%濃塩酸)を50mL混合して混合物を得、この混合物を前記グリセリン系固形物800mLに混合して常温で30分間攪拌を行うことによって液化物を得た。得られた液化物はpH7であった。
前記実施例1で得られたグリセリン系固形物(約pH14)の液化を次の手法で行った。まず予め灯油300mLに濃塩酸(35質量%濃塩酸)を20mL混合して混合物を得、この混合物を前記グリセリン系固形物700mLに混合して常温で30分間攪拌を行うことによって液化物を得た。得られた液化物はpH10であった。
前記実施例1で得られたグリセリン系固形物(約pH14)の液化を次の手法で行った。まず予め灯油300mLに濃塩酸(35質量%濃塩酸)を80mL混合して混合物を得、この混合物を前記グリセリン系固形物700mLに混合して常温で30分間攪拌を行うことによって液化物を得た。得られた液化物はpH5であった。
前記実施例1で得られたグリセリン系固形物(約pH14)の液化を次の手法で行った。まず予め灯油300mLに濃硫酸(90質量%濃硫酸)を10mL混合して混合物を得、この混合物を前記グリセリン系固形物700mLに混合して常温で30分間攪拌を行うことによって液化物を得た。得られた液化物はpH8であった。
前記実施例1で得られたグリセリン系固形物(約pH14)の液化を次の手法で行った。まず予め灯油300mLに濃塩酸(35質量%濃塩酸)を5mL混合して混合物を得、この混合物を前記グリセリン系固形物700mLに混合して常温で30分間攪拌を行うことによって液化物を得た。得られた液化物はpH13であった。
前記実施例1で得られた液化物(pH7、褐色透明液)に対して更に次のような後処理を実施した。即ち、実施例1で得られた液化物(褐色透明液)を20℃まで冷却して20℃で1440分間保持した後、該20℃の液化物中に存在する沈殿物をフィルターで物理濾過することによって濾液を得、次いで濾液を200℃の蒸留温度で蒸留することによって、無色透明の蒸留液(蒸留済み液化物)を得た。
前記実施例1で得られたグリセリン系固形物(約pH14、凝固点25℃)の液化を次の手法で行った。前記グリセリン系固形物700mLに過酸化水素(5質量%過酸化水素水)を30mL混合した混合物を60℃で20分間攪拌を行った後、さらに灯油300mLを加えて60℃で10分間攪拌し、さらに酢酸(純度99質量%)を10mL加えて60℃で10分間攪拌を行うことによって液化物(pH7、褐色透明液)を得た。
灯油300mLに代えて重油300mLを用いた以外は、実施例9と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
灯油300mLに代えて軽油300mLを用いた以外は、実施例9と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
過酸化水素(5質量%過酸化水素水)の使用量を10mLに設定した以外は、実施例9と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
過酸化水素(5質量%過酸化水素水)の使用量を80mLに設定した以外は、実施例9と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
酢酸の使用量を5mLに設定した以外は、実施例9と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
酢酸の使用量を40mLに設定した以外は、実施例9と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
前記実施例1で得られたグリセリン系固形物(約pH14)の液化を次の手法で行った。前記グリセリン系固形物700mLに過酸化水素(35質量%過酸化水素水)を30mL混合した混合物を60℃で10分間攪拌を行った後、さらに灯油300mLを加えて60℃で10分間攪拌を行うことによって液化物(pH7、褐色透明液)を得た。
灯油300mLに代えて重油300mLを用いた以外は、実施例16と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
灯油300mLに代えて軽油300mLを用いた以外は、実施例16と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
過酸化水素(35質量%過酸化水素水)の使用量を10mLに設定した以外は、実施例16と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
過酸化水素(35質量%過酸化水素水)の使用量を70mLに設定した以外は、実施例16と同様にして、無色透明の蒸留液(蒸留済み液化物)を得た。
前記実施例1で得られたグリセリン系固形物(約pH14、凝固点25℃)の液化を次の手法で行った。前記グリセリン系固形物700mLを60℃まで加熱して液状物にした後、該液状物に過酸化水素(5質量%過酸化水素水)を10mL混合した混合物を60℃で20分間攪拌を行った後、さらに酢酸(純度99質量%)を20mL加えて60℃で10分間攪拌を行うことによって液化物(pH5、褐色透明液)を得た。
液状グリセリン系物質(凝固点-5℃)700mLに過酸化水素(5質量%過酸化水素水)を10mL混合した混合物を60℃で20分間攪拌を行った後、さらに灯油300mLを加えて60℃で10分間攪拌し、さらに酢酸(純度99質量%)を20mL加えて60℃で10分間攪拌を行うことによって液化物(pH5、褐色透明液)を得た。
得られた液化物1000mLを約25℃の条件下で7日間放置した後、その液状状態を目視により観察した。観察結果を表1、2に示す。
得られた液化物を目視で観察し、下記判定基準に基づき液化物中の残渣の含有程度を評価した。評価結果を表1、2に示す。
(判定基準)
「◎」…液化物中の残渣の含有が非常に少ない
「○」…液化物中の残渣の含有が少ない
「△」…液化物中の残渣の含有が多い
「×」…液化物中の残渣の含有が非常に多い。
得られた液化物を目視で観察して液化物の色を評価すると共に、下記判定基準に基づき液化物の透明性を評価した。評価結果を表1、2に示す。
(判定基準)
「透明」…液化物は透明性に優れている
「半透明」…液化物は透明ではあるが、その透明度は高くない
「不透明」…液化物は不透明である。
Claims (30)
- グリセリンを含有してなるグリセリン系固形物と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。 - 植物油からバイオディーゼル燃料を生産する際に副生物として出るグリセリン系固形物と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。 - 前記混合の際に、油と酸とを混合した混合液と、グリセリン系固形物とを混合する請求項1または2に記載のグリセリン系固形物から液化物を製造する方法。
- 前記混合の際に、グリセリン系固形物と油の合計容量に対するグリセリン系固形物の含有率が50~90容量%になるように混合すると共に、グリセリン系固形物と油の合計100容量部に対して酸を2~10容量部混合することを特徴とする請求項1~3のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- グリセリンを含有してなるグリセリン系固形物と酸とを混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
- 前記酸として無機酸を用いる請求項1~5のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- 前記無機酸が塩酸である請求項6に記載のグリセリン系固形物から液化物を製造する方法。
- グリセリンを含有してなるグリセリン系固形物と、
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸化剤を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。 - 植物油からバイオディーゼル燃料を生産する際に副生物として出るグリセリン系固形物と、
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸化剤を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。 - 前記混合の際に、先にグリセリン系固形物と酸化剤とを混合した後、油を混合する請求項8または9に記載のグリセリン系固形物から液化物を製造する方法。
- 前記混合の際に、グリセリン系固形物100容量部に対し、酸化剤を35質量%濃度の酸化剤溶液に換算して0.1~20容量部、油を0.1~40容量部混合することを特徴とする請求項8~10のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- グリセリンを含有してなるグリセリン系固形物と酸化剤とを混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸化剤を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
- 前記酸化剤として過酸化水素を用いる請求項8~12のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- グリセリンを含有してなるグリセリン系固形物と、
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。 - 植物油からバイオディーゼル燃料を生産する際に副生物として出るグリセリン系固形物と、
酸化剤と、
鉱物油、動物油及び植物油からなる群より選ばれる少なくとも1種の油と、
酸と、を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。 - 前記混合の際に、先にグリセリン系固形物と酸化剤とを混合した後、さらに油を混合し、次いで酸を混合する請求項14または15に記載のグリセリン系固形物から液化物を製造する方法。
- 前記混合の際に、グリセリン系固形物100容量部に対し、酸化剤を5質量%濃度の酸化剤溶液に換算して0.1~20容量部、油を0.1~40容量部、酸を0.1~10容量部混合することを特徴とする請求項14~16のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- グリセリンを含有してなるグリセリン系固形物、酸化剤及び酸を混合することによって液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とするグリセリン系固形物から液化物を製造する方法。
- 前記酸化剤として過酸化水素を用いる請求項14~18のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- 前記酸として酢酸を用いる請求項14~19のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- 前記液化物がpH4~pH11になるように前記酸を混合する請求項14~20のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- 前記油として石油を用いる請求項1~21のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- 前記石油は、灯油、軽油及び重油からなる群より選ばれる1種または2種以上の石油である請求項22に記載のグリセリン系固形物から液化物を製造する方法。
- 前記石油は、灯油である請求項22に記載のグリセリン系固形物から液化物を製造する方法。
- 前記混合により得られた液化物を30℃以下の温度まで冷却した後、沈殿物を濾過することによって濾液を得、次いで前記濾液を150℃~230℃の蒸留温度で蒸留することを特徴とする請求項1~24のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- 前記混合により得られた液化物を30℃以下の温度まで冷却した後、沈殿物を濾過することによって濾液を得、前記濾液を5℃以下の温度まで冷却して該冷却液から上澄み液を得、次いで前記上澄み液を150℃~230℃の蒸留温度で蒸留することを特徴とする請求項1~24のいずれか1項に記載のグリセリン系固形物から液化物を製造する方法。
- グリセリンを含有してなる液状グリセリン系物質と酸とを混合することによって、前記液状グリセリン系物質の凝固点よりも低い凝固点を有する液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とする液化物の製造方法。
- グリセリンを含有してなる液状グリセリン系物質と酸化剤とを混合することによって、前記液状グリセリン系物質の凝固点よりも低い凝固点を有する液化物を製造し、該液化物がpH3~pH12になるように前記酸化剤を混合することを特徴とする液化物の製造方法。
- グリセリンを含有してなる液状グリセリン系物質と、酸と、酸化剤とを混合することによって、前記液状グリセリン系物質の凝固点よりも低い凝固点を有する液化物を製造し、該液化物がpH3~pH12になるように前記酸を混合することを特徴とする液化物の製造方法。
- 請求項1~29のいずれか1項に記載の製造方法によって製造された燃料用液化物。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201080046002.4A CN102892871B (zh) | 2009-08-31 | 2010-08-30 | 从甘油类固态物制造液化物的方法 |
| AU2010287437A AU2010287437A1 (en) | 2009-08-31 | 2010-08-30 | Process for production of liquefied material from glycerin-based solid material |
| US13/393,368 US8814960B2 (en) | 2009-08-31 | 2010-08-30 | Method for producing liquefied material from glycerin-based solid material |
| JP2011528888A JP5984390B2 (ja) | 2009-08-31 | 2010-08-30 | グリセリン系固形物から液化物を製造する方法 |
| EP10812035.3A EP2474597A4 (en) | 2009-08-31 | 2010-08-30 | PROCESS FOR PRODUCING LIQUEFIED MATERIAL FROM GLYCERINE-LIKE SOLID MATERIAL |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009199944 | 2009-08-31 | ||
| JP2009-199944 | 2009-08-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011024989A1 true WO2011024989A1 (ja) | 2011-03-03 |
Family
ID=43628080
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2010/064673 WO2011024989A1 (ja) | 2009-08-31 | 2010-08-30 | グリセリン系固形物から液化物を製造する方法 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US8814960B2 (ja) |
| EP (1) | EP2474597A4 (ja) |
| JP (1) | JP5984390B2 (ja) |
| KR (1) | KR20120083370A (ja) |
| CN (1) | CN102892871B (ja) |
| AU (1) | AU2010287437A1 (ja) |
| MY (1) | MY159310A (ja) |
| TW (1) | TW201124522A (ja) |
| WO (1) | WO2011024989A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013173826A (ja) * | 2012-02-24 | 2013-09-05 | Hiroshi Yamashita | 廃グリセリン再生燃料製造装置および方法 |
| CN104277857A (zh) * | 2014-09-25 | 2015-01-14 | 中国农业大学 | 一种藻类与粗甘油共液化制备的生物原油及方法 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101386077B1 (ko) * | 2013-02-25 | 2014-04-16 | 조선대학교산학협력단 | 액상 라디칼 반응에 의한 글리세린으로부터 메탄올을 주성분으로 하는 바이오연료 제조 방법 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004156836A (ja) * | 2002-11-07 | 2004-06-03 | Meidensha Corp | 廃植物油の再利用システム |
| JP2005060587A (ja) * | 2003-08-18 | 2005-03-10 | Daiki Co Ltd | 廃油脂のケミカルリサイクル方法 |
| WO2006129435A1 (ja) * | 2005-06-03 | 2006-12-07 | Lion Corporation | 脂肪酸低級アルキルエステルの製造方法および脂肪酸低級アルキルエステルならびに軽油代替燃料 |
| JP2006348191A (ja) | 2005-06-16 | 2006-12-28 | Chugoku Electric Power Co Inc:The | バイオマス循環システム |
| JP2008111098A (ja) * | 2006-08-15 | 2008-05-15 | Nova Biosource Technologies Llc | 高遊離脂肪酸原料からのバイオディーゼル及びグリセリンの生成 |
| JP2009199944A (ja) | 2008-02-22 | 2009-09-03 | Panasonic Electric Works Co Ltd | マグネット式コンセントアダプタ |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2194894A (en) * | 1936-02-20 | 1940-03-26 | Ellis Foster Co | Resinified soya bean oil product and process of making same |
| JP3530885B2 (ja) * | 1996-11-11 | 2004-05-24 | 箕口 新一 | 廃食油からのディーゼル燃料油の製造装置 |
| JP3530884B2 (ja) * | 1996-11-11 | 2004-05-24 | 箕口 新一 | 廃食油からのディーゼル燃料油の製造方法 |
| JP4854889B2 (ja) * | 2001-09-21 | 2012-01-18 | 株式会社レボインターナショナル | 廃食油からのディーゼル燃料油の製造方法 |
| JP2006348189A (ja) | 2005-06-16 | 2006-12-28 | Dynic Corp | 孔版印刷用インク |
| US20070113465A1 (en) * | 2005-11-21 | 2007-05-24 | Pech Craig W | Method for manufacture and use of the waste stream from biodiesel production (crude glycerin) as a commercial fuel |
| EP1918354A1 (de) * | 2006-10-13 | 2008-05-07 | Cognis Oleochemicals GmbH | Brennstoffzubereitungen die Glycerin enthalten |
| US8450370B2 (en) * | 2007-12-21 | 2013-05-28 | Old Dominion University Research Foundation | Production of glycerol-related products from a high temperature reaction |
| CN101481297A (zh) * | 2008-12-30 | 2009-07-15 | 浙江工业大学 | 一种生物柴油副产物甘油的精制方法 |
| US8648219B2 (en) * | 2010-08-20 | 2014-02-11 | James Cramer | Method for purifying glycerin and products obtained therefrom |
-
2010
- 2010-08-30 US US13/393,368 patent/US8814960B2/en not_active Expired - Fee Related
- 2010-08-30 EP EP10812035.3A patent/EP2474597A4/en not_active Withdrawn
- 2010-08-30 JP JP2011528888A patent/JP5984390B2/ja not_active Expired - Fee Related
- 2010-08-30 AU AU2010287437A patent/AU2010287437A1/en not_active Abandoned
- 2010-08-30 CN CN201080046002.4A patent/CN102892871B/zh not_active Expired - Fee Related
- 2010-08-30 TW TW099129077A patent/TW201124522A/zh unknown
- 2010-08-30 KR KR1020127008325A patent/KR20120083370A/ko not_active Application Discontinuation
- 2010-08-30 MY MYPI2012000864A patent/MY159310A/en unknown
- 2010-08-30 WO PCT/JP2010/064673 patent/WO2011024989A1/ja active Application Filing
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004156836A (ja) * | 2002-11-07 | 2004-06-03 | Meidensha Corp | 廃植物油の再利用システム |
| JP2005060587A (ja) * | 2003-08-18 | 2005-03-10 | Daiki Co Ltd | 廃油脂のケミカルリサイクル方法 |
| WO2006129435A1 (ja) * | 2005-06-03 | 2006-12-07 | Lion Corporation | 脂肪酸低級アルキルエステルの製造方法および脂肪酸低級アルキルエステルならびに軽油代替燃料 |
| JP2006348191A (ja) | 2005-06-16 | 2006-12-28 | Chugoku Electric Power Co Inc:The | バイオマス循環システム |
| JP2008111098A (ja) * | 2006-08-15 | 2008-05-15 | Nova Biosource Technologies Llc | 高遊離脂肪酸原料からのバイオディーゼル及びグリセリンの生成 |
| JP2009199944A (ja) | 2008-02-22 | 2009-09-03 | Panasonic Electric Works Co Ltd | マグネット式コンセントアダプタ |
Non-Patent Citations (2)
| Title |
|---|
| MARTA PANIAGUA ET AL., ENERGY & FUELS, vol. 21, no. 3, 2007, pages 1782 - 1791, XP008161060 * |
| See also references of EP2474597A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013173826A (ja) * | 2012-02-24 | 2013-09-05 | Hiroshi Yamashita | 廃グリセリン再生燃料製造装置および方法 |
| CN104277857A (zh) * | 2014-09-25 | 2015-01-14 | 中国农业大学 | 一种藻类与粗甘油共液化制备的生物原油及方法 |
| CN104277857B (zh) * | 2014-09-25 | 2016-04-06 | 中国农业大学 | 一种藻类与粗甘油共液化制备的生物原油及方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US8814960B2 (en) | 2014-08-26 |
| US20120186145A1 (en) | 2012-07-26 |
| EP2474597A1 (en) | 2012-07-11 |
| AU2010287437A1 (en) | 2012-04-12 |
| JP5984390B2 (ja) | 2016-09-06 |
| KR20120083370A (ko) | 2012-07-25 |
| EP2474597A4 (en) | 2013-04-24 |
| MY159310A (en) | 2016-12-30 |
| JPWO2011024989A1 (ja) | 2013-01-31 |
| TW201124522A (en) | 2011-07-16 |
| CN102892871B (zh) | 2015-04-08 |
| CN102892871A (zh) | 2013-01-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Shahbaz et al. | Eutectic solvents for the removal of residual palm oil-based biodiesel catalyst | |
| Kralova et al. | Biofuels–renewable energy sources: a review | |
| JP5757603B2 (ja) | 低温で安定なバイオジェット燃料の製造方法 | |
| US20070204506A1 (en) | Adjustable fuel power booster component composition | |
| US8142525B2 (en) | Preparation of fatty acid esters of glycerol formal and its use as biofuel | |
| CN101245271B (zh) | 车用高效生物柴油及其制备方法 | |
| CN101531930A (zh) | 一种耐低温醇类清洁燃料油及制备方法 | |
| JP5984390B2 (ja) | グリセリン系固形物から液化物を製造する方法 | |
| Forero | Biodiesel from castor oil: a promising fuel for cold weather | |
| WO2008135801A2 (en) | Adjustable fuel power booster component composition | |
| CN101333449B (zh) | 一种生物柴油制备方法 | |
| Kanthavelkumaran et al. | Recent trends and applications of bio diesel | |
| CN101812331A (zh) | 一种低硫酯基复合燃料油 | |
| CN106316727A (zh) | 一种多孔粒状铵油炸药及其制备方法 | |
| CN101508918A (zh) | 一种甲醇柴油及其制备方法 | |
| US10323199B2 (en) | Fuel supplement to reduce harmful emissions | |
| Shahbaz et al. | Removal of residual KOH from palm oil based biodiesel using deep eutectic solvents | |
| CN109609205A (zh) | 一种醇基柴油燃料及其制备方法 | |
| Zanzi et al. | Biodiesel fuel production | |
| US8974553B2 (en) | Miscible diesel fuel ethanol composition | |
| Isamotu et al. | Performance Evaluation of Biodiesel Sourced from Groundnut Oil for an IC Engine | |
| US10961472B2 (en) | Modification of fats and oils for fuel and lubricating applications | |
| Grai | Modelling of three-component systems in biodiesel production technology | |
| US20090038692A1 (en) | Modification of vegetable oils for fuel applications | |
| BRPI0703303B1 (pt) | processo de obtenção de biocombustíveis para motores diesel veiculares e estacionários |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080046002.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10812035 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011528888 Country of ref document: JP Ref document number: 12012500408 Country of ref document: PH |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1201000850 Country of ref document: TH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 678/MUMNP/2012 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010287437 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010812035 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20127008325 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13393368 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2010287437 Country of ref document: AU Date of ref document: 20100830 Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112012004553 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112012004553 Country of ref document: BR Kind code of ref document: A2 Effective date: 20120229 |