WO2010052161A2 - Herbicidal compositions - Google Patents
Herbicidal compositions Download PDFInfo
- Publication number
- WO2010052161A2 WO2010052161A2 PCT/EP2009/064214 EP2009064214W WO2010052161A2 WO 2010052161 A2 WO2010052161 A2 WO 2010052161A2 EP 2009064214 W EP2009064214 W EP 2009064214W WO 2010052161 A2 WO2010052161 A2 WO 2010052161A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- crc
- alkyl
- methyl
- Prior art date
Links
- 0 *C(*)(CC(CC1(*)*O2)(*(C3=O)I)C2=C3c2c(*)cc(*)c(*)c2*)*1O* Chemical compound *C(*)(CC(CC1(*)*O2)(*(C3=O)I)C2=C3c2c(*)cc(*)c(*)c2*)*1O* 0.000 description 1
- CTVLTMSKUZLVII-UHFFFAOYSA-N CCON(CC1)CCC1(C(OC)=O)NC(Cc1c(C)cc(C)cc1C)=O Chemical compound CCON(CC1)CCC1(C(OC)=O)NC(Cc1c(C)cc(C)cc1C)=O CTVLTMSKUZLVII-UHFFFAOYSA-N 0.000 description 1
- KICSUORQCGEXIL-UHFFFAOYSA-N CON(CC1)CCC1(C#N)N Chemical compound CON(CC1)CCC1(C#N)N KICSUORQCGEXIL-UHFFFAOYSA-N 0.000 description 1
- HQWDZXPJBOITFK-UHFFFAOYSA-N Cc(c(C(C(NC1(CC2)CCN2OC)=O)=C1O)c1)cc(F)c1Br Chemical compound Cc(c(C(C(NC1(CC2)CCN2OC)=O)=C1O)c1)cc(F)c1Br HQWDZXPJBOITFK-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/02—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom
- A01N47/06—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom containing —O—CO—O— groups; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/18—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing a —O—CO—N< group, or a thio analogue thereof, directly attached to a heterocyclic or cycloaliphatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/10—Spiro-condensed systems
Definitions
- the present invention relates to novel herbicidal compositions containing as active ingredient spiroheterocyclic pyrrolidine diones or derivatives thereof and to the use of these compounds in controlling grasses and weeds, especially in crops of useful plants, or in inhibiting undesired plant growth.
- Spiroheterocyclic pyrrolidine dione compounds having herbicidal action are described, for example, in WO9501358.
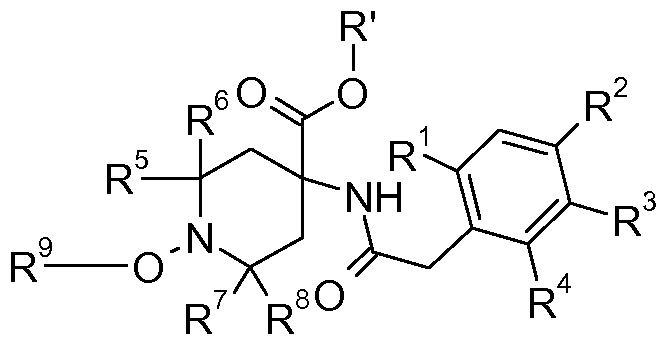
- the present invention accordingly relates to novel herbicidal compositions containg in addition to comprising formulation adjuvants, a herbicidally effective amount of a compound of formula I
- G is hydrogen or an agriculturally acceptable metal, sulfonium, ammonium or latentiating group
- R 1 is hydrogen, methyl, ethyl, n-propyl, iso-propyl, halomethyl, haloethyl, halogen, vinyl, ethynyl, methoxy, ethoxy, halomethoxy or haloethoxy, cyclopropyl or halogenocyclopropyl
- R 2 and R 3 are independently hydrogen, halogen, d-C ⁇ alkyl, d-C ⁇ haloalkyl, d-C ⁇ alkoxy, d- C 6 haloalkoxy, C 2 -C 6 alkenyl, C 2 -C 6 haloalkenyl, C 2 -C 6 alkynyl, C 2 -C 6 haloalkynyl, C 3 -C 6 alkenyloxy, Cs-C ⁇ haloalkenyloxy,
- R 4 is hydrogen, methyl, ethyl, n-propyl, iso-propyl, halomethyl, haloethyl, halogen, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, halomethoxy or haloethoxy,
- R 5 , R 6 , R 7 and R 8 independently of each other, are hydrogen or methyl
- R 9 is hydrogen, d-C ⁇ alkyl, d-C ⁇ haloalkyl, d-C ⁇ cyanoalkyl, benzyl, d-dalkoxy (d-d)alkyl, d- dalkoxy (Ci-d)alkoxy(Ci-d)alkyl or a group selected from G, or an agrochemically acceptable salt or N-oxide thereof.
- each alkyl moiety either alone or as part of a larger group is a straight or branched chain and is, for example, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, isopropyl, n-butyl, sec-butyl, isobutyl, fert-butyl or neopentyl.
- the alkyl groups are suitably d-C 6 alkyl groups, but are preferably d-dalkyl or d-C 3 alkyl groups, and, more preferably, d-C 2 alkyl groups.
- Alkenyl and alkynyl moieties can be in the form of straight or branched chains, and the alkenyl moieties, where appropriate, can be of either the (E)- or (Z)-configu ration. Examples are vinyl, allyl and propargyl. Alkenyl and alkynyl moieties can contain one or more double and/or triple bonds in any combination. It is understood, that allenyl and alkylinylalkenyl are included in these terms.
- Halogen is fluorine, chlorine, bromine or iodine.
- Haloalkyl groups are alkyl groups which are substituted with one or more of the same or different halogen atoms and are, for example, CF 3 , CF 2 CI, CF 2 H, CCI 2 H, FCH 2 , CICH 2 , BrCH 2 , CH 3 CHF, (CHs) 2 CF, CF 3 CH 2 or CHF 2 CH 2 .
- Cycloalkyl includes preferably cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl.
- heteroaryl preferably refers to an aromatic ring system containing at least one heteroatom and consisting either of a single ring or of two or more fused rings.
- single rings will contain up to three and bicyclic systems up to four heteroatoms which will preferably be chosen from nitrogen, oxygen and sulphur.
- Examples of such groups include furyl, thienyl, pyrrolyl, pyrazolyl, imidazolyl, 1 ,2,3-triazolyl, 1 ,2,4-triazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, 1 ,2,4-oxadiazolyl, 1 ,3,4-oxadiazolyl, 1 ,2,5-oxadiazolyl, 1 ,2,3-thiadiazolyl, 1 ,2,4-thiadiazolyl, 1 ,3,4-thiadiazolyl, 1 ,2,5-thiadiazolyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl, 1 ,2,3-triazinyl, 1 ,2,4-triazinyl, 1 ,3,5-triazinyl, benzofuryl, benzisofuryl, benzothienyl,
- heteroaromatic radicals include pyridyl, pyrimidinyl, triazinyl, thienyl, furyl, oxazolyl, isoxazolyl, 2,1 ,3-benzoxadiazolyl and thiazolyl.
- Another group of preferred heteroaryls comprises furyl, thienyl, pyrazolyl, 1 ,2,3-triazolyl, 1 ,2,4-triazolyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinazolinyl or quinoxalinyl.
- the invention relates also to herbicidal compositions containing the agriculturally acceptable salts which the compounds of formula I are able to form with transition metal, alkali metal and alkaline earth metal bases, amines, quaternary ammonium bases or tertiary sulfonium bases.
- transition metal alkali metal and alkaline earth metal salt formers
- hydroxides of copper, iron, lithium, sodium, potassium, magnesium and calcium special mention should be made of the hydroxides of copper, iron, lithium, sodium, potassium, magnesium and calcium, and preferably the hydroxides, bicarbonates and carbonates of sodium and potassium.
- amines suitable for ammonium salt formation include ammonia as well as primary, secondary and tertiary Ci-Cisalkylamines, Ci-C 4 hydroxyalkylamines and C 2 -C 4 alkoxyalkylamines, for example methylamine, ethylamine, n-propylamine, isopropylamine, the four butylamine isomers, n-amylamine, isoamylamine, hexylamine, heptylamine, octylamine, nonylamine, decylamine, pentadecylamine, hexadecylamine, heptadecylamine, octadecylamine, methylethylamine, methylisopropylamine, methylhexylamine, methylnonylamine, methylpentadecylamine, methyloctadecylamine, ethylbutyl
- piperidine, pyrrolidine, indoline, quinuclidine and azepine primary arylamines, for example anilines, methoxyanilines, ethoxyanilines, o-, m- and p-toluidines, phenylenediamines, benzidines, naphthylamines and o-, m- and p-chloroanilines; but especially triethylamine, isopropylamine and diisopropylamine.
- Preferred quaternary ammonium bases suitable for salt formation correspond, for example, to the formula [N(R 3 R b R c R d )]OH, wherein R 3 , R b , R c and R d are each independently of the others CrC 4 alkyl.
- Further suitable tetraalkylammonium bases with other anions can be obtained, for example, by anion exchange reactions.
- Preferred tertiary sulfonium bases suitable for salt formation correspond, for example, to the formula [SR e R f R g ]OH, wherein R e , R f and R 9 are each independently of the others C 1 -C 4 alkyl.
- Trimethylsulfonium hydroxide is especially preferred.
- Suitable sulfonium bases may be obtained from the reaction of thioethers, in particular dialkylsulfides, with alkylhalides, followed by conversion to a suitable base, for example a hydroxide, by anion exchange reactions.
- inventively used compounds of formula I also include hydrates which may be formed during the salt formation.
- the latentiating groups G are selected to allow its removal by one or a combination of biochemical, chemical or physical processes to afford compounds of formula I where G is H before, during or following application to the treated area or plants. Examples of these processes include enzymatic cleavage, chemical hydrolysis and photoloysis. Compounds bearing such groups G may offer certain advantages, such as improved penetration of the cuticula of the plants treated, increased tolerance of crops, improved compatibility or stability in formulated mixtures containing other herbicides, herbicide safeners, plant growth regulators, fungicides or insecticides, or reduced leaching in soils.
- the latentiating group G is preferably selected from the groups C-i-Csalkyl, C 2 -Cshaloalkyl, phenylCrC 8 alkyl (wherein the phenyl may optionally be substituted by CrC 3 alkyl, CrC 3 haloalkyl, CrCsalkoxy, C-i-Cshaloalkoxy, C-i-Csalkylthio, CrCsalkylsulfinyl, d-Csalkylsulfonyl, halogen, cyano or by nitro), heteroarylC-i-Csalkyl (wherein the heteroaryl may optionally be substituted by CrC 3 alkyl, C r C 3 haloalkyl, C r C 3 alkoxy, C r C 3 haloalkoxy, C r C 3 alkylthio, C r C 3 alkylsulfinyl, C 1 -C 3 alkylsulf
- R a is H, Ci-Ci 8 alkyl, C 2 -Ci 8 alkenyl, C 2 -Ci 8 alkynyl, Ci-Ciohaloalkyl, Ci-Ciocyanoalkyl, d- Ci 0 nitroalkyl, Ci-Ci 0 aminoalkyl, Ci-C 5 alkylaminoCi-C 5 alkyl, C 2 -C 8 dialkylaminoCi-C 5 alkyl, C 3 - C 7 cycloalkylCrC 5 alkyl, Ci-CsalkoxyCi-Csalkyl, drCsalkenyloxyd-Csalkyl, C 3 -C 5 alkynylC r Csoxyalkyl, Ci-CsalkylthioCi-Csalkyl, Ci-CsalkylsulfinylCi-Csalkyl, Ci-CsalkylsulfonylCi-
- R e is Ci-Cioalkyl, C 2 -Cioalkenyl, C 2 -Cioalkynyl, d-dohaloalkyl, Ci-Ciocyanoalkyl, d- Cionitroalkyl, d-Cioaminoalkyl, Ci-CsalkylaminoCi-Csalkyl, C 2 -C 8 dialkylaminoCrC 5 alkyl, C 3 - C 7 cycloalkylCrC 5 alkyl, d-CsalkoxyCrCsalkyl, Cs-CsalkenyloxyCrCsalkyl, C 3 -C 5 alkynyloxyd- C 5 alkyl, Ci-CsalkylthioCi-Csalkyl, Ci-CsalkylsulfinylCi-Csalkyl, Ci-CsalkylsulfonylCi-Csalky
- R h is CrCioalkyl, C 3 -Cioalkenyl, C 3 -Cioalkynyl, Ci-Ciohaloalkyl, Ci-Ciocyanoalkyl, Cr Ci 0 nitroalkyl, C 2 -Ci 0 aminoalkyl, Ci-C 5 alkylaminoCi-C 5 alkyl, C 2 -C 8 dialkylaminoCi-C 5 alkyl, C 3 - C 7 cycloalkylCi-C 5 alkyl, Ci-C 5 alkoxyCi-C 5 alkyl, Cs-CsalkenyloxyC-i-Csalkyl, C 3 -C 5 alkynyloxyCr C 5 alkyl, Ci-CsalkylthioCi-Csalkyl, Ci-CsalkylsulfinylCi-Csalkyl, d-CsalkylsulfonylCi-
- the latentiating group G is a group -C(X a )-R a or -C(X b )-X c -R b , and the meanings of X a , R a , X b , X c and R b are as defined above.
- G is hydrogen, an alkali metal or alkaline earth metal, where hydrogen is especially preferred.
- inventively used compounds of formula I may exist in different geometric or optical isomers or different tautomeric forms.
- One or more centres of chirality may be present, in which case compounds of the formula I may be present as pure enantiomers, mixtures of enantiomers, pure diastereomers or mixtures of diastereomers. Centres of tautomerisation may be present.
- This invention covers the herbicidal use of all such isomers and tautomers and mixtures thereof in all proportions as well as isotopic forms such as deuterated compounds.
- Preferred herbicidal compositions according to the invention are those wherein in the compounds of formula I R 1 methyl, ethyl, n-propyl, vinyl, ethynyl, halogen, methoxy, ethoxy, halomethoxy, haloethoxy, R 2 is methyl, halogen, methoxy, ethoxy, halomethoxy, haloethoxy, phenyl or phenyl substituted by CrC 4 alkyl, d-Cshaloalkyl, d-Csalkoxy, C-i-Cshaloalkoxy, cyano, nitro, halogen, CrCsalkylthio, C-i-Csalkylsulfinyl or d-Csalkylsulfonyl, R 3 is hydrogen, R 4 is methyl ethyl, n- propyl, vinyl or ethynyl, R 5 , R 6 , R 7 and
- inventively used compounds may be made by a variety of methods.
- the compounds of formula I wherein the substituents have the meanings assigned to them above, can be prepared by means of processes known per se, e.g. by treating compounds of formula (A) with an alkylating, acylating, phosphorylating or sulfonylating agent G-Q in the presence of at least one equivalent of a base, where G is the alkyl, acyl, phosphoryl or sulfonyl group to be incorporated and Q is a nucleofuge:
- compounds of formula (A) are treated with an acylating agent such as an acid halide (especially acid chloride), acid anhydride, haloformate (especially chloroformate), halothioformate (especially chlorothioformate), isocyanate, isothiocycanate, carbamoyl halide (especially carbamoyl chloride) or thiocarbamoyl chloride (especially thiocarbamoyl chloride) in the presence of at least one equivalent of a suitable base, optionally in the presence of a suitable solvent.
- an acylating agent such as an acid halide (especially acid chloride), acid anhydride, haloformate (especially chloroformate), halothioformate (especially chlorothioformate), isocyanate, isothiocycanate, carbamoyl halide (especially carbamoyl chloride) or thiocarbamoyl chloride (especially thiocarbamoyl chlor
- the base may be inorganic such as an alkali metal carbonate or hydroxide or a metal hydride, or an organic base such as a tertiary amine or metal alkoxide.
- suitable inorganic bases include sodium carbonate, sodium or potassium hydroxide, sodium hydride
- suitable organic bases include trialkylamines such as trimethylamine and triethylamine, pyridines or other amine bases such as 1 ,4- diazobicyclo[2.2.2]octane and 1 ,8-diazabicyclo[5.4.0]undec-7-ene.
- Preferred bases include triethylamine and pyridine.
- Suitable solvents for this reaction are selected to be compatible with the reagents and include ethers such as tetrahydrofuran and 1 ,2-dimethoxyethane and halogenated solvents such as dichloromethane and chloroform. Certain bases, such as pyridine and triethylamine, may be employed successfully as both base and solvent.
- acylation is preferably effected in the presence of a coupling agent such as 2-chloro-1-methylpyridinium iodide, ⁇ /, ⁇ / -dicyclohexycarbodiimide, 1-(3- dimethylaminopropyl)-3-ethylcarbodiimide and ⁇ /, ⁇ / -carbodiimidazole, and a base such as triethylamine or pyridine in a suitable solvent such as tetrahydrofuran, dichloromethane and acetonitrile.
- a coupling agent such as 2-chloro-1-methylpyridinium iodide, ⁇ /, ⁇ / -dicyclohexycarbodiimide, 1-(3- dimethylaminopropyl)-3-ethylcarbodiimide and ⁇ /, ⁇ / -carbodiimidazole
- a base such as triethylamine or pyridine
- suitable solvent such as t
- Compounds of formula I, wherein G is a latentiating group of the formula C(X b )-X c -R b , C(X d )- N(R C )-R d may be also be prepared by treating compounds of formula Il with phosgene or a phosgene equivalent, optionally in the presence of a solvent such as toluene or ethyl acetate, and a base and reacting the resultant chloroformate, or equivalent, with an alcohol, thiol or amine under known conditions, as described, for example, in US 6774133, US6555567 and US6479489.
- Compounds of formula I, wherein G is a latentiating group of the formula -S ⁇ 2 -R e may be prepared by reaction of compounds of formula Il with an alkyl or aryl sulfonyl halide, preferably in the presence of at least one equivalent of base.
- Compounds of formula I wherein G is CrC ⁇ alkyl, C 2 -C 6 alkenyl, Cs-C ⁇ alkynyl or a latentiating group of the formula CH 2 -X f -R h , may be prepared by treatment of a compound of formula Il with a compound of formula G-Y wherein Y is a halogen (especially bromine or iodine), sulfonate (especially mesylate or tosylate) or a sulfate preferably in the presence of a base, under known conditions.
- Y is a halogen (especially bromine or iodine), sulfonate (especially mesylate or tosylate) or a sulfate preferably in the presence of a base, under known conditions.
- Suitable conditions are the same as described above for the conversion of compounds of formula (A) to compounds of formula I.
- Compounds of formula (A) may be prepared via the cyclisation of compounds of Formula (C), preferably in the presence of base, and optionally in the presence of a suitable solvent, by methods described, for example, in US6774133, US6555567 and US6479489.
- Compounds of formula (C) may be prepared by reacting amino acid derivatives of formula (D) with phenylacetyl halides of formula (E), preferably in the presence of base in a suitable solvent by known methods described, for example, in US6774133, US6555567 and US6479489.
- Phenylacetyl halides of formula (E), wherein Hal is Cl or Br are known compounds or can be prepared by known methods as described, for example, in US6774133, US6555567 and US6479489.
- Amino acid esters of formula (D), wherein R is CrC 4 alkyl can be prepared by known methods from amino acids of formula (F). These compounds can be isolated as free amines or amine salts.
- Amino acids of formula (F) can be prepared from ketons of formula (J) by means of Strecker Synthesis via amino nitriles of formula (G). Altematively, amino acids of formula (F) can be prepared from ketones of formula (J) by means of Bucherer-Bergs reaction via hydantoins of formula (H).
- compounds of formula (C) may be prepared by subjecting derivatives of formula (K) to alcoholysis with ROH, preferably in in acidic media by known methods described, for example, in US6774133, US6555567 and US6479489.
- Compounds of formula (K) may be themselves prepared by reacting amino nitriles of formula (G) with phenylacetyl halides of formula (E), preferably in the presence of base in a suitable solvent by known methods described, for example, in US6774133, US6555567 and US6479489.
- the starting compounds and intermediates of the reaction schemes are known or can be prepared according to methods known to a person skilled in the art.
- the reactants can be reacted in the presence of a base.
- suitable bases are alkali metal or alkaline earth metal hydroxides, alkali metal or alkaline earth metal hydrides, alkali metal or alkaline earth metal amides, alkali metal or alkaline earth metal alkoxides, alkali metal or alkaline earth metal acetates, alkali metal or alkaline earth metal carbonates, alkali metal or alkaline earth metal dialkylamides or alkali metal or alkaline earth metal alkylsilylamides, alkylamines, alkylenediamines, free or N-alkylated saturated or unsaturated cycloalkylamines, basic heterocycles, ammonium hydroxides and carbocyclic amines.
- Examples which may be mentioned are sodium hydroxide, sodium hydride, sodium amide, sodium methoxide, sodium acetate, sodium carbonate, potassium tert-butoxide, potassium hydroxide, potassium carbonate, potassium hydride, lithium diisopropylamide, potassium bis(trimethylsilyl)amide, calcium hydride, triethylamine, diisopropylethylamine, triethylenediamine, cyclohexylamine, N-cyclohexyl-N,N- dimethylamine, N,N-diethylaniline, pyridine, 4-(N,N-dimethylamino)pyridine, quinuclidine, N- methylmorpholine, benzyltrimethylammonium hydroxide and 1 ,8-diazabicyclo[5.4.0]undec-7-ene (DBU).
- the reactants can be reacted with each other as such, i.e. without adding a solvent or diluent. In most cases, however, it is advantageous to add an inert solvent or diluent or a mixture of these. If the reaction is carried out in the presence of a base, bases which are employed in excess, such as triethylamine, pyridine, N-methylmorpholine or N,N-diethylaniline, may also act as solvents or diluents.
- the reaction is advantageously carried out in a temperature range from approximately -80 0 C to approximately +140 0 C, preferably from approximately -30 0 C to approximately +100 0 C, in many cases in the range between ambient temperature and approximately +80 0 C.
- a compound I can be converted in a manner known per se into another compound I by replacing one or more substituents of the starting compound I in the customary manner by (an)other substituent(s) according to the invention.
- Salts of compounds I can be prepared in a manner known per se.
- acid addition salts of compounds I are obtained by treatment with a suitable acid or a suitable ion exchanger reagent and salts with bases are obtained by treatment with a suitable base or with a suitable ion exchanger reagent.
- Salts of compounds I can be converted in the customary manner into the free compounds I, acid addition salts, for example, by treatment with a suitable basic compound or with a suitable ion exchanger reagent and salts with bases, for example, by treatment with a suitable acid or with a suitable ion exchanger reagent.
- Salts of compounds I can be converted in a manner known per se into other salts of compounds I, acid addition salts, for example, into other acid addition salts, for example by treatment of a salt of inorganic acid such as hydrochloride with a suitable metal salt such as a sodium, barium or silver salt, of an acid, for example with silver acetate, in a suitable solvent in which an inorganic salt which forms, for example silver chloride, is insoluble and thus precipitates from the reaction mixture.
- a salt of inorganic acid such as hydrochloride
- a suitable metal salt such as a sodium, barium or silver salt
- the compounds I which have salt-forming properties can be obtained in free form or in the form of salts.
- the compounds I and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can be present in the form of one of the isomers which are possible or as a mixture of these, for example in the form of pure isomers, such as antipodes and/or diastereomers, or as isomer mixtures, such as enantiomer mixtures, for example racemates, diastereomer mixtures or racemate mixtures, depending on the number, absolute and relative configuration of asymmetric carbon atoms which occur in the molecule and/or depending on the configuration of non-aromatic double bonds which occur in the molecule; the invention relates to the pure isomers and also to all isomer mixtures which are possible and is to be understood in each case in this sense hereinabove and hereinbelow, even when stereochemical details are not mentioned specifically in each case.
- Diastereomer mixtures or racemate mixtures of compounds I, in free form or in salt form, which can be obtained depending on which starting materials and procedures have been chosen can be separated in a known manner into the pure diasteromers or racemates on the basis of the physicochemical differences of the components, for example by fractional crystallization, distillation and/or chromatography.
- Enantiomer mixtures such as racemates, which can be obtained in a similar manner can be resolved into the optical antipodes by known methods, for example by recrystallization from an optically active solvent, by chromatography on chiral adsorbents, for example high-performance liquid chromatography (HPLC) on acetyl celulose, with the aid of suitable microorganisms, by cleavage with specific, immobilized enzymes, via the formation of inclusion compounds, for example using chiral crown ethers, where only one enantiomer is complexed, or by conversion into diastereomeric salts, for example by reacting a basic end-product racemate with an optically active acid, such as a carboxylic acid, for example camphor, tartaric or malic acid, or sulfonic acid, for example camphorsulfonic acid, and separating the diastereomer mixture which can be obtained in this manner, for example by fractional crystallization based on their differing solubilities, to give the
- Pure diastereomers or enantiomers can be obtained according to the invention not only by separating suitable isomer mixtures, but also by generally known methods of diastereoselective or enantioselective synthesis, for example by carrying out the process according to the invention with starting materials of a suitable stereochemistry.
- the compounds I and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can, if appropriate, also be obtained in the form of hydrates and/or include other solvents, for example those which may have been used for the crystallization of compounds which are present in solid form.
- inventively used compounds of formula I can be used as herbicides in unmodified form, as obtained in the synthesis, but they are generally formulated into herbicidal compositions in a variety of ways using formulation adjuvants, such as carriers, solvents and surface-active substances.
- the formulations can be in various physical forms, for example in the form of dusting powders, gels, wettable powders, water-dispersible granules, water-dispersible tablets, effervescent compressed tablets, emulsifiable concentrates, microemulsifiable concentrates, oil- in-water emulsions, oil flowables, aqueous dispersions, oily dispersions, suspoemulsions, capsule suspensions, emulsifiable granules, soluble liquids, water-soluble concentrates (with water or a water-miscible organic solvent as carrier), impregnated polymer films or in other forms known, for example, from the Manual on Development and Use of FAO Specifications for Plant Protection Products, 5th Edition, 1999.
- Such formulations can either be used directly or are diluted prior to use.
- Diluted formulations can be prepared, for example, with water, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
- the formulations can be prepared, for example, by mixing the active ingredient with formulation adjuvants in order to obtain compositions in the form of finely divided solids, granules, solutions, dispersions or emulsions.
- the active ingredients can also be formulated with other adjuvants, for example finely divided solids, mineral oils, vegetable oils, modified vegetable oils, organic solvents, water, surface-active substances or combinations thereof.
- the active ingredients can also be contained in very fine microcapsules consisting of a polymer. Microcapsules contain the active ingredients in a porous carrier. This enables the active ingredients to be released into their surroundings in controlled amounts (e.g. slow release). Microcapsules usually have a diameter of from 0.1 to 500 microns.
- the active ingredients contain active ingredients in an amount of about from 25 to 95 % by weight of the capsule weight.
- the active ingredients can be present in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution.
- the encapsulating membranes comprise, for example, natural and synthetic gums, cellulose, styrene-butadiene copolymers, polyacrylonitrile, polyacrylate, polyester, polyamides, polyureas, polyurethane or chemically modified polymers and starch xanthates or other polymers that are known to the person skilled in the art in this connection.
- the formulation adjuvants suitable for the preparation of the compositions according to the invention are known perse.
- liquid carriers there may be used: water, toluene, xylene, petroleum ether, vegetable oils, acetone, methyl ethyl ketone, cyclohexanone, acid anhydrides, acetonitrile, acetophenone, amyl acetate, 2-butanone, butylenes carbonate, chlorobenzene, cyclo- hexane, cyclohexanol, alkyl esters of acetic acid, diacetone alcohol, 1 ,2-dichloropropane, diethanolamine, p-diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, N,N-dimethylformamide, dimethyl sulfoxide, 1 ,4-dioxan
- Water is generally the carrier of choice for the dilution of the concentrates.
- Suitable solid carriers are, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, limestone, calcium carbonate, bentonite, calcium montomorillonite, cottonseed husks, wheatmeal, soybean flour, pumice, wood flour, ground walnut shells, lignin and similar materials, as described, for example, in CFR 180.1001. (c) & (d).
- a large number of surface-active substances can advantageously be used both in solid and in liquid formulations, especially in those formulations which can be diluted with a carrier prior to use.
- Surface-active substances may be anionic, cationic, non-ionic or polymeric and they may be used as emulsifiying, wetting or suspending agents or for other purposes.
- Typical surface-active substances include, for example, salts of alkyl sulfates, such as diethanolammonium lauryl sulfate; salts of alkylarylsulfonates, such as calcium dodecylbenzenesulfonate; alkylphenol- alkylene oxide addition products, such as nonylphenol ethoxylate; alcohol-alkylene oxide addition products, such as tridecyl alcohol ethoxylate; soaps, such as sodium stearate; salts of alkylnaphthalenesulfonates, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2-ethylhexyl)sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryl trimethylammonium chloride, polyethylene glycol esters of fatty acids,
- Further adjuvants which can usually be used in pesticidal formulations include crystallisation inhibitors, viscosity-modifying substances, suspending agents, dyes, anti-oxidants, foaming agents, light absorbers, mixing aids, anti-foams, complexing agents, neutralising or pH-modifying substances and buffers, corrosion-inhibitors, fragrances, wetting agents, absorption improvers, micronutrients, plasticisers, glidants, lubricants, dispersants, thickeners, anti-freezes, microbiocides, and also liquid and solid fertilisers.
- the formulations may also comprise additional active substances, for example further herbicides, herbicide safeners, plant growth regulators, fungicides or insecticides
- compositions according to the invention can additionally include an additive comprising an oil of vegetable or animal origin, a mineral oil, alkyl esters of such oils or mixtures of such oils and oil derivatives.
- the amount of oil additive used in the composition according to the invention is generally from 0.01 to 10 %, based on the spray mixture.
- the oil additive can be added to the spray tank in the desired concentration after the spray mixture has been prepared.
- Preferred oil additives comprise mineral oils or an oil of vegetable origin, for example rapeseed oil, olive oil or sunflower oil, emulsified vegetable oil, such as AMIGO® (Rh ⁇ ne-Poulenc Canada Inc.), alkyl esters of oils of vegetable origin, for example the methyl derivatives, or an oil of animal origin, such as fish oil or beef tallow.
- a preferred additive contains, for example, as active components essentially 80 % by weight alkyl esters of fish oils and 15 % by weight methylated rapeseed oil, and also 5 % by weight of customary emulsifiers and pH modifiers.
- Especially preferred oil additives comprise alkyl esters of C 8 -C 22 fatty acids, especially the methyl derivatives of C 12 -C 18 fatty acids, for example the methyl esters of lauric acid, palmitic acid and oleic acid, being important.
- Those esters are known as methyl laurate (CAS-1 1 1-82-0), methyl palmitate (CAS- 1 12-39-0) and methyl oleate (CAS-1 12-62-9).
- a preferred fatty acid methyl ester derivative is Emery ⁇ 2230 and 2231 (Cognis GmbH).
- Those and other oil derivatives are also known from the Compendium of Herbicide Adjuvants, 5th Edition, Southern Illinois University, 2000.
- the application and action of the oil additives can be further improved by combining them with surface-active substances, such as non-ionic, anionic or cationic surfactants.
- surface-active substances such as non-ionic, anionic or cationic surfactants.
- suitable anionic, non-ionic and cationic surfactants are listed on pages 7 and 8 of WO97/34485.
- Preferred surface-active substances are anionic surfactants of the dodecylbenzylsulfonate type, especially the calcium salts thereof, and also non-ionic surfactants of the fatty alcohol ethoxylate type. Special preference is given to ethoxylated C 12 -C 22 fatty alcohols having a degree of ethoxylation of from 5 to 40.
- Examples of commercially available surfactants are the Genapol types (Clariant AG).
- silicone surfactants especially polyalkyl-oxide-modified heptamethyltrisiloxanes, which are commercially available e.g. as Silwet L-77®, and also perfluorinated surfactants.
- concentration of surface-active substances in relation to the total additive is generally from 1 to 30 % by weight.
- oil additives that consist of mixtures of oils or mineral oils or derivatives thereof with surfactants are Edenor ME SU®, Turbocharge® (Syngenta AG, CH) and Actipron® (BP Oil UK Limited, GB).
- the said surface-active substances may also be used in the formulations alone, that is to say without oil additives.
- oil additive/surfactant mixture can contribute to a further enhancement of action.
- Suitable solvents are, for example, Solvesso® (ESSO) and Aromatic Solvent® (Exxon Corporation).
- the concentration of such solvents can be from 10 to 80 % by weight of the total weight.
- Such oil additives which may be in admixture with solvents, are described, for example, in US4834908.
- a commercially available oil additive disclosed therein is known by the name MERGE® (BASF Corporation).
- Further oil additives that are preferred according to the invention are SCORE® (Syngenta Crop Protection Canada) and Adigor® (Syngenta Crop Protection Canada).
- alkylpyrrolidones e.g. Agrimax®
- formulations of alkylpyrrolidones such as, for example, Agrimax®
- synthetic latices such as, for example, polyacrylamide, polyvinyl compounds or poly-1-p-menthene (e.g. Bond®, Courier® or Emerald®)
- propionic acid for example Eurogkem Pen-e-trate®
- the herbicidal formulations generally contain from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, of a compound of formula I and from 1 to 99.9 % by weight of a formulation adjuvant, which preferably includes from 0 to 25 % by weight of a surface-active substance. Whereas commercial products will preferably be formulated as concentrates, the end user will normally employ dilute formulations.
- the rate of application of the compounds of formula I may vary within wide limits and depends upon the nature of the soil, the method of application (pre- or post-emergence; seed dressing; application to the seed furrow; no tillage application etc.), the crop plant, the weed or grass to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop.
- the compounds of formula I are generally applied at a rate of 1- 2000 g/ha, preferably 1- 1000 g / ha and most preferably at 1- 500 g / ha.
- Emulsifiable concentrates active ingredient: 1 to 95 %, preferably 60 to 90 % surface-active agent: 1 to 30 %, preferably 5 to 20 % liquid carrier: 1 to 80 %, preferably 1 to 35 %
- Dusts active ingredient: 0.1 to 10 %, preferably 0.1 to 5 % solid carrier: 99.9 to 90 %, preferably 99.9 to 99 %
- Suspension concentrates active ingredient: 5 to 75 %, preferably 10 to 50 % water: 94 to 24 %, preferably 88 to 30 % surface-active agent: 1 to 40 %, preferably 2 to 30 %
- Wettable powders active ingredient: 0.5 to 90 %, preferably 1 to 80 % surface-active agent: 0.5 to 20 %, preferably 1 to 15 % solid carrier: 5 to 95 %, preferably 15 to 90 %
- Granules active ingredient: 0.1 to 30 %, preferably 0.1 to 15 % solid carrier: 99.5 to 70 %, preferably 97 to 85 %
- Emulsifiable concentrates a) b) c) d) active ingredient 5 % 10 % 25 % 50 % calcium dodecylbenzene- sulfonate 6 % 8 % 6 % 8 % castor oil polyglycol ether 4 % - 4 % 4 %
- Emulsions of any desired concentration can be prepared from such concentrates by dilution with water.
- the solutions are suitable for application in the form of microdrops.
- Wettable powders a) b) c) d) active ingredient 5% 25% 50% 80% sodium lignosulfonate 4 % - 3 % - sodium lauryl sulfate 2 % 3 % - 4 % sodium diisobutylnaphthalene- sulfonate _ 6 % 5% 6 % octylphenol polyglycol ether - 1 % 2 % -
- the active ingredient is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, yielding wettable powders which can be diluted with water to give suspensions of any desired concentration.
- the active ingredient is dissolved in methylene chloride, the solution is sprayed onto the carrier and the solvent is subsequently evaporated off in vacuo.
- F5. Coated granules a) b) c) active ingredient 0.1 % 5% 15 % polyethylene glycol MW 200 1.0 % 2% 3 % highly disperse silicic acid 0.9 % 1 % 2 % inorg. carrier 98. 0% 92% 80 %
- the finely ground active ingredient is applied uniformly, in a mixer, to the carrier moistened with polyethylene glycol. Non-dusty coated granules are obtained in this manner.
- the active ingredient is mixed and ground with the adjuvants and the mixture is moistened with water.
- the resulting mixture is extruded and then dried in a stream of air.
- Ready-to-use dusts are obtained by mixing the active ingredient with the carriers and grinding the mixture in a suitable mill.
- Suspension concentrates a) b) c) d) active ingredient 3 % 10% 25% 50% ethylene glycol 5% 5% 5% nonylphenol polyglycol ether - 1 % 2 % -
- the finely ground active ingredient is intimately mixed with the adjuvants, yielding a suspension concentrate from which suspensions of any desired concentration can be prepared by dilution with water.
- the invention relates also to a method for the selective control of grasses and weeds in crops of useful plants, and for non-selective weed control, which comprises treating the useful plants or the area under cultivation or the locus thereof with a compound of formula I.
- Crops of useful plants in which the compositions according to the invention can be used include especially cereals, in particular wheat and barley, rice, corn, rape, sugarbeet, sugarcane, soybean, cotton, sunflower, peanut and plantation crops.
- crops is to be understood as also including crops that have been rendered tolerant to herbicides or classes of herbicides (for example ALS, GS, EPSPS, PPO and HPPD inhibitors) as a result of conventional methods of breeding or genetic engineering.
- herbicides or classes of herbicides for example ALS, GS, EPSPS, PPO and HPPD inhibitors
- An example of a crop that has been rendered tolerant e.g. to imidazolinones, such as imazamox, by conventional methods of breeding is Clearfield® summer rape (Canola).
- crops that have been rendered tolerant to herbicides by genetic engineering methods include e.g. glyphosate- and glufosinate- resistant maize varieties commercially available under the trade names RoundupReady ⁇ and LibertyLink®.
- the weeds to be controlled may be both monocotyledonous and dicotyledonous weeds, such as, for example, Stellaria, Nasturtium, Agrostis, Digitaria, Avena, Setaria, Sinapis, Lolium, Solanum, Echinochloa, Scirpus, Monochoria, Sagittaria, Bromus, Alopecurus, Sorghum, Rottboellia, Cyperus, Abutilon, Sida, Xanthium, Amaranthus, Chenopodium, Ipomoea, Chrysanthemum, Galium, Viola and Veronica. Control of monocotyledonous weeds, in particular Agrostis, Avena, Setaria, Lolium, Echinochloa, Bromus, Alopecurus and Sorghum is very extensive.
- Crops are also to be understood as being those which have been rendered resistant to harmful insects by genetic engineering methods, for example Bt maize (resistant to European corn borer), Bt cotton (resistant to cotton boll weevil) and also Bt potatoes (resistant to Colorado beetle).
- Bt maize are the Bt-176 maize hybrids of NK® (Syngenta Seeds).
- the Bt toxin is a protein that is formed naturally by Bacillus thuringiensis soil bacteria. Examples of toxins and transgenic plants able to synthesise such toxins are described in EP-A-451 878, EP-A-374 753, WO 93/07278, WO 95/34656, WO 03/052073 and EP-A-427 529.
- transgenic plants that contain one or more genes which code for an insecticidal resistance and express one or more toxins are KnockOut® (maize), Yield Gard® (maize), NuCOTIN33B® (cotton), Bollgard® (cotton), NewLeaf® (potatoes), NatureGard® and Protexcta®.
- Plant crops and their seed material can be resistant to herbicides and at the same time also to insect feeding ("stacked" transgenic events). Seed can, for example, have the ability to express an insecticidally active Cry3 protein and at the same time be glyphosate-tolerant.
- the term "crops" is to be understood as also including crops obtained as a result of conventional methods of breeding or genetic engineering which contain so-called output traits (e.g. improved flavour, storage stability, nutritional content).
- Areas under cultivation are to be understood as including land where the crop plants are already growing as well as land intended for the cultivation of those crop plants.
- the compounds of formula I used according to the invention can also be used in combination with further herbicides.
- the compound of the formula I is one of those compounds listed in Tables 1 to 22 below.
- the following mixtures of the compound of formula I are preferred: compound of formula I + aclonifen, compound of formula I + amidosulfuron, compound of formula I + aminopyralid, compound of formula I + azimsulfuron, compound of formula I + beflubutamid, compound of formula I + benfluralin, compound of formula I + bensulfuron, compound of formula I + bensulfuron-methyl, compound of formula I + benzobicyclon, compound of formula I + bifenox, compound of formula I + bispyribac, compound of formula I + bispyribac-sodium, compound of formula I + bromoxynil, compound of formula I + butachlor, compound of formula I + butafenacil, compound of formula I + cafenstrole, compound of formula I + carbetamide,
- the mixing partners for the compound of formula I may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, 12th Edition (BCPC) 2000.
- the compounds of formula I used according to the invention can also be used in combination with safeners.
- the compound of the formula I is one of those compounds listed in Tables 1 to 22 below.
- Benoxacor, cloquintocet-mexyl, cyprosulfamide, mefenpyr-diethyl and N-(2-methoxybenzoyl)-4- [(methylaminocarbonyl)amino]benzenesulfonamide are especially preferred, where cloquintocet- mexyl is particularly valuable.
- Step 1
- Step 1
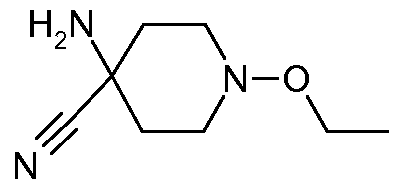
- 4-Amino-1-ethoxy-piperidine-4-carbonitrile is prepared analogously to the synthesis of 4-amino-1- methoxy-piperidine-4-carbonitrile (preparation example 1 , Step 1 ) starting from N-ethoxy-4- piperidone (Journal of Organic Chemistry (1961 ), 26, 1867-74).
- Step 1
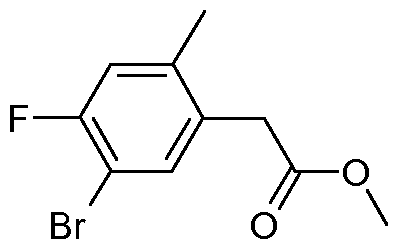
- reaction mixture is filtered, the solvent removed in vacuo and the residue is subjected to silica gel chromatography (isohexane/diethyl ether 2:1 ) to yield 2.5 g of (5-cyclopropyl-2,4-dimethyl- phenyl)-acetic acid methyl ester.
- Step 1
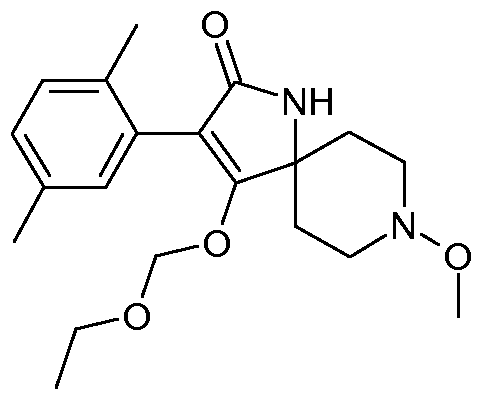
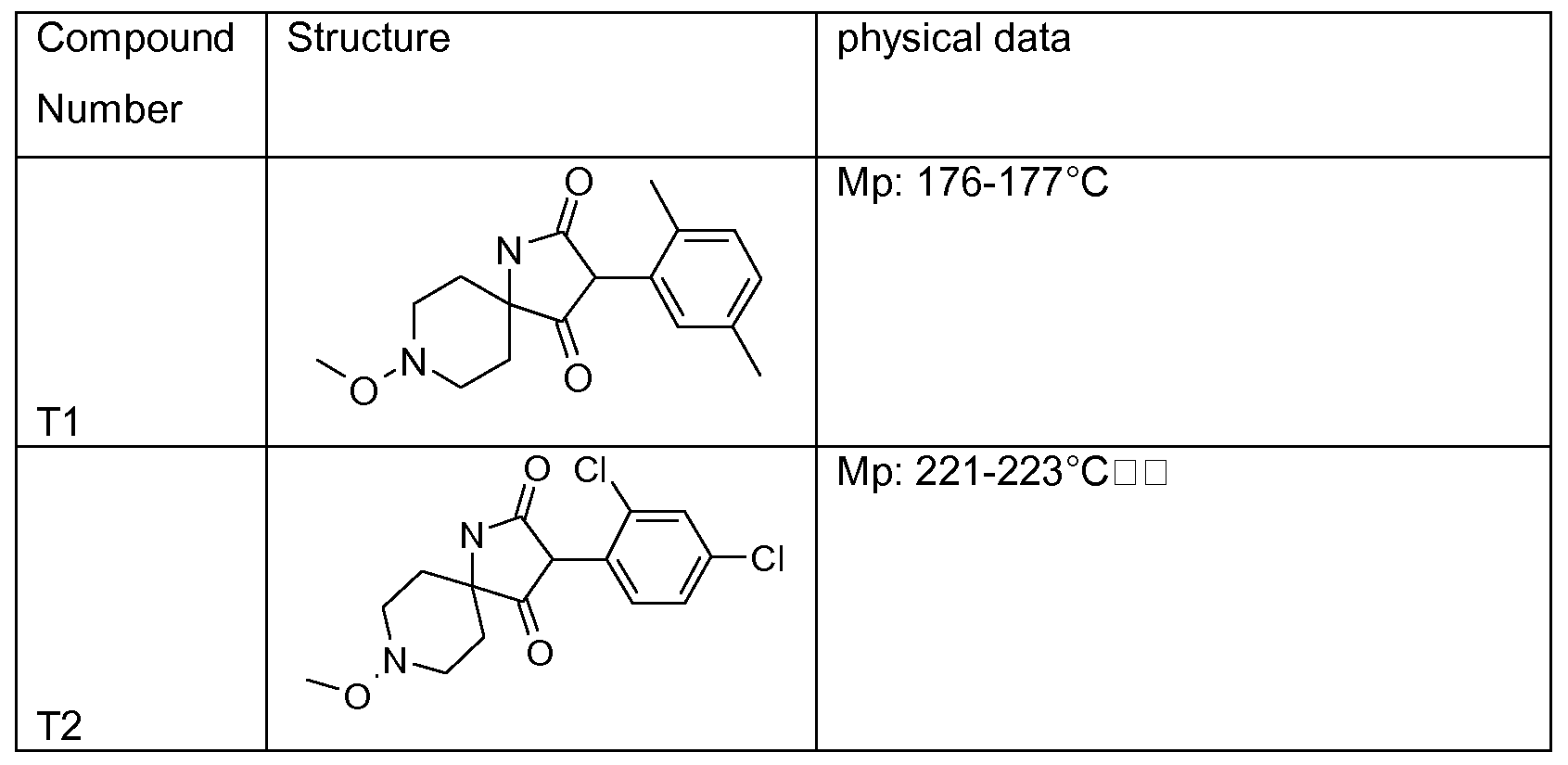
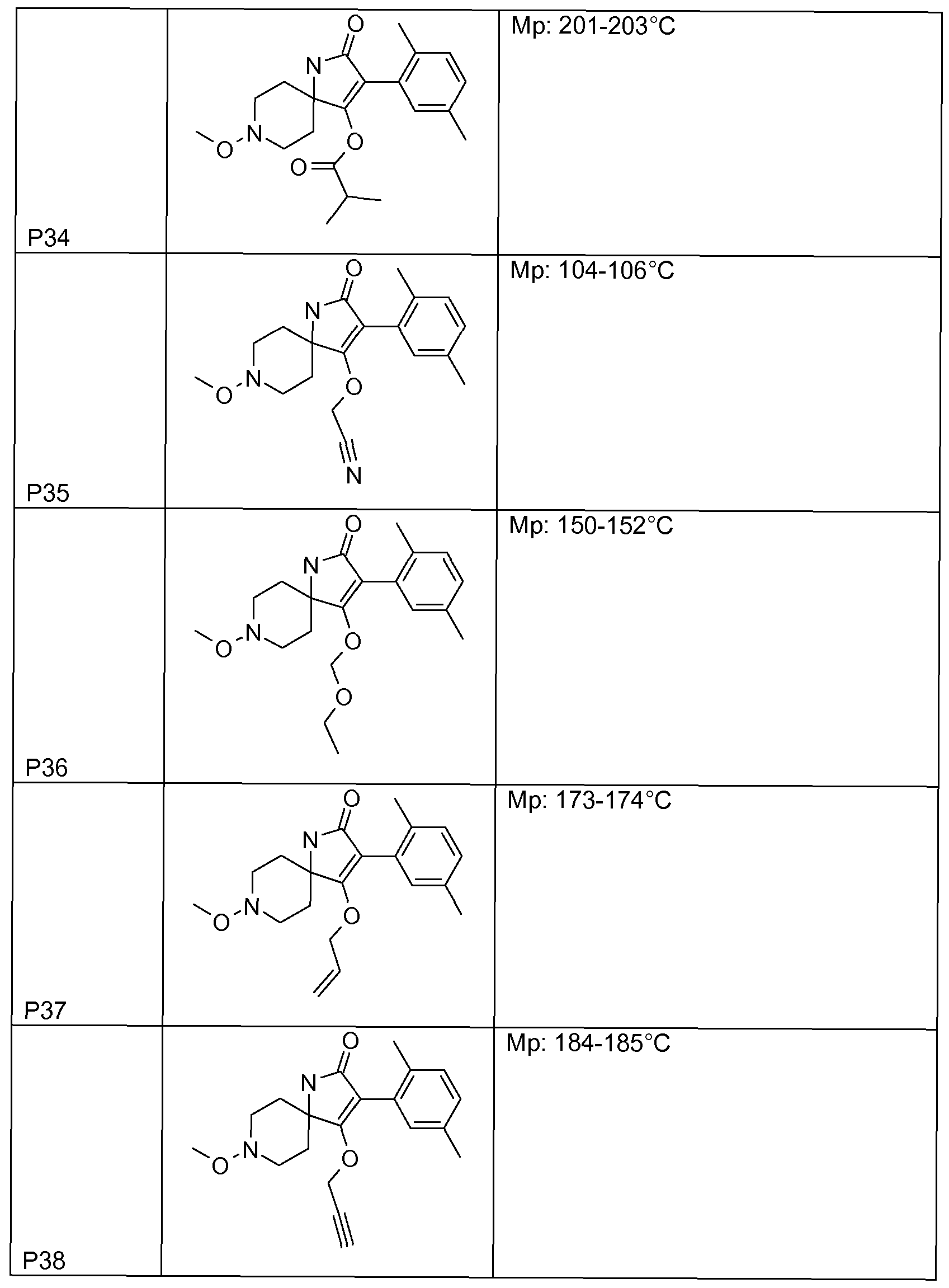
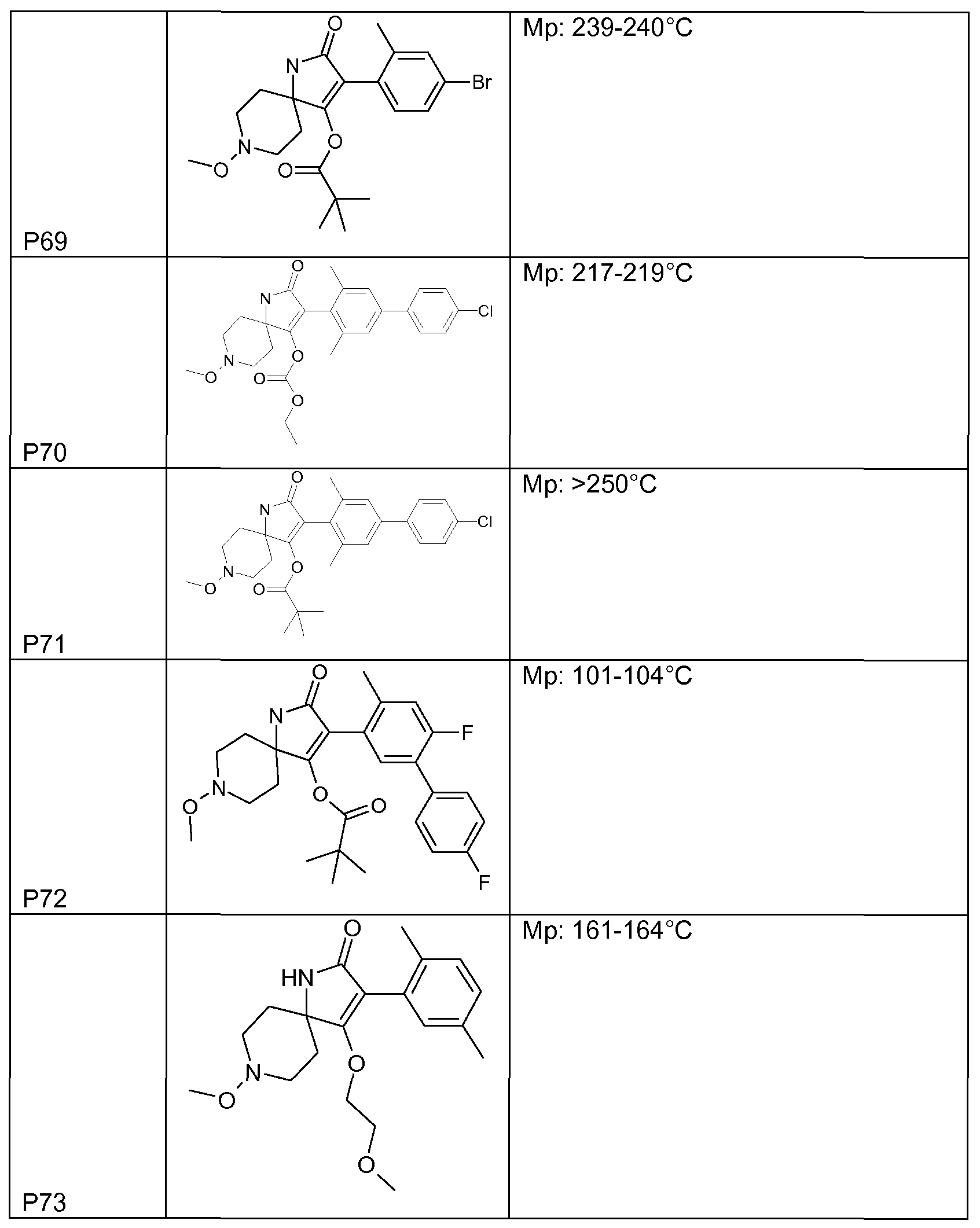
- Tables 1 to 22 below illustrate the compounds used according to the invention.
- Table 1 This table discloses the 262 compounds of the formula I:
- R ⁇ M 9 i s, CH 3 , R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined below:
- Table 2 This table discloses the 262 compounds of the formula I, wherein R 9 is CH 2 CH 3 , R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 3 This table discloses the 262 compounds of the formula I, wherein R 9 is H-CsH 7 , R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 4 discloses the 262 compounds of the formula I, wherein R 9 is /-C 3 H 7 , R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 5 This table discloses the 262 compounds of the formula I, wherein R 9 is allyl, R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 6 This table discloses the 262 compounds of the formula I, wherein R 9 is benzyl, R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 16 This table discloses the 262 compounds of the formula I, wherein R 5 , R 6 , R 7 , R 8 and R 9 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 17 This table discloses the 262 compounds of the formula I, wherein R 9 is CH 2 -O-CH 3 , R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 18 discloses the 262 compounds of the formula I, wherein R 9 is CH 2 -O-C 2 H 5 , R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 19 discloses the 262 compounds of the formula I, wherein R 9 is CH 2 -O-C 2 H 4 -O- CH 3 , R 5 , R 6 , R 7 and R 8 are hydrogen, G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 20 This table discloses the 262 compounds of the formula I, wherein R 9 is hydrogen, R 5 , R 6 , R 7 and R 8 are CH 3 , G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 21 This table discloses the 262 compounds of the formula I, wherein R 9 is CH 3 , R 5 , R 6 , R 7 and R 8 are CH 3 , G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Table 22 This table discloses the 262 compounds of the formula I, wherein R 9 is C 2 H 5 , R 5 , R 6 , R 7 and R 8 are CH 3 , G is hydrogen and R 1 , R 2 , R 3 and R 4 are as defined in Table 1.
- Alopecurus myosuroides Alopecurus myosuroides (ALOMY), Avena fatua (AVEFA), Lolium perenne (LOLPE), Setaria faberi (SETFA), Digitaria sanguinalis (DIGSA), Echinochloa crus-galli (ECHCG)
- Seeds of a variety of test species are sown in standard soil in pots. After cultivation for one day
- aqueous spray solution derived from the formulation of the technical active ingredient in acetone / water (50:50) solution containing 0.5% Tween 20 (polyoxyethelyene sorbitan monolaurate, CAS
- test plants are then grown in a glasshouse under controlled conditions in a glasshouse (at
- Setaria faberi (SETFA), Alopecurus myosuroides (ALOMY), Echinochloa crus-galli (ECHCG), Avena fatua (AVEFA)
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2742552A CA2742552A1 (en) | 2008-11-06 | 2009-10-28 | Herbicidal compositions |
| US13/128,133 US8722578B2 (en) | 2008-11-06 | 2009-10-28 | Spiroheterocyclic pyrrolidinedione herbicidal compositions |
| AU2009312909A AU2009312909A1 (en) | 2008-11-06 | 2009-10-28 | Herbicidal compositions |
| CN2009801441973A CN102209469A (en) | 2008-11-06 | 2009-10-28 | Herbicidal compositions |
| EP09740705.0A EP2352375B1 (en) | 2008-11-06 | 2009-10-28 | Herbicidal compositions |
| BRPI0921411-9A BRPI0921411A2 (en) | 2008-11-06 | 2009-10-28 | Herbicidal composition, and method for controlling grass and weeds in useful plant crops. |
| JP2011533709A JP5592386B2 (en) | 2008-11-06 | 2009-10-28 | Herbicidal composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GBGB0820344.0A GB0820344D0 (en) | 2008-11-06 | 2008-11-06 | Herbicidal compositions |
| GB0820344.0 | 2008-11-06 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010052161A2 true WO2010052161A2 (en) | 2010-05-14 |
| WO2010052161A3 WO2010052161A3 (en) | 2010-12-23 |
Family
ID=40139509
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/064214 WO2010052161A2 (en) | 2008-11-06 | 2009-10-28 | Herbicidal compositions |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US8722578B2 (en) |
| EP (1) | EP2352375B1 (en) |
| JP (1) | JP5592386B2 (en) |
| CN (1) | CN102209469A (en) |
| AU (1) | AU2009312909A1 (en) |
| BR (1) | BRPI0921411A2 (en) |
| CA (1) | CA2742552A1 (en) |
| GB (1) | GB0820344D0 (en) |
| WO (1) | WO2010052161A2 (en) |
Cited By (250)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011067131A1 (en) * | 2009-12-04 | 2011-06-09 | Syngenta Participations Ag | Spiroheterocyclic dione derivatives used as pesticides |
| WO2011098440A2 (en) | 2010-02-10 | 2011-08-18 | Bayer Cropscience Ag | Biphenyl substituted cyclical keto-enols |
| WO2011098443A1 (en) | 2010-02-10 | 2011-08-18 | Bayer Cropscience Ag | Spiroheterocyclical substituted tetramic acid derivatives |
| WO2011098433A1 (en) | 2010-02-15 | 2011-08-18 | Bayer Schering Pharma Aktiengesellschaft | Cyclic keto-enols for therapy |
| WO2011131623A1 (en) | 2010-04-20 | 2011-10-27 | Bayer Cropscience Ag | Insecticidal and/or herbicidal composition having improved activity on the basis of spiro-heterocyclically substituted tetramic acid derivatives |
| WO2011151199A1 (en) * | 2010-05-31 | 2011-12-08 | Syngenta Participations Ag | Spiroheterocyclic pyrrolidine derivatives based pesticides |
| WO2011151248A1 (en) * | 2010-05-31 | 2011-12-08 | Syngenta Participations Ag | Pesticidal compositions |
| WO2011151247A3 (en) * | 2010-05-31 | 2012-03-01 | Syngenta Participations Ag | Pesticidal compositions |
| JP2012511541A (en) * | 2008-12-12 | 2012-05-24 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | Spiroheterocyclic N-oxypiperidine as a pesticide |
| US20120190865A1 (en) * | 2011-01-25 | 2012-07-26 | Bayer Cropscience Ag | Process for the Preparation of 1-H-pyrrolidine-2,4-dione Derivatives |
| DE102011011040A1 (en) | 2011-02-08 | 2012-08-09 | Bayer Pharma Aktiengesellschaft | (5s, 8s) -3- (4'-chloro-3'-fluoro-4-methylbiphenyl-3-yl) -4-hydroxy-8-methoxy-1-azaspiro [4.5] dec-3-en-2- on (compound A) for therapy |
| WO2012110519A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Cropscience Ag | Substituted 3-(biphenyl-3-yl)-8,8-difluoro-hydroxy-1-azaspiro[4.5]dec-3-en-2-ones for therapy and halogen-substituted spirocyclic ketoenols |
| WO2012116960A1 (en) | 2011-03-01 | 2012-09-07 | Bayer Cropscience Ag | 2-acyloxy-pyrrolin-4-ones |
| DE102011080406A1 (en) | 2011-08-04 | 2013-02-07 | Bayer Pharma AG | Substituted 3- (biphenyl-3-yl) -4-hydroxy-8-methoxy-1-azaspiro8 [4.5] dec-3-ene-2-ones |
| DE102011080405A1 (en) | 2011-08-04 | 2013-02-07 | Bayer Pharma AG | New substituted 3-biphenyl-3-yl-8,8-difluoro-4-hydroxy-1-azaspiro(4.5)dec-3-en-2-one derivatives useful for prophylaxis or therapy of tumor diseases comprising breast cancer, prostate cancer, colorectal cancer or non-small cell lung cancer |
| WO2013079672A1 (en) * | 2011-11-30 | 2013-06-06 | Syngenta Limited | Herbicidal uses of substituted spiroheterocyclic pyrrolidine dione derivatives, and herbicidal compositions comprising them |
| JP2013527208A (en) * | 2010-05-31 | 2013-06-27 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | How to grow crops |
| KR20140098226A (en) * | 2011-11-30 | 2014-08-07 | 신젠타 파티서페이션즈 아게 | Pesticidal mixtures including spiroheterocyclic pyrrolidine diones |
| WO2017072039A1 (en) | 2015-10-26 | 2017-05-04 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093180A1 (en) | 2015-12-01 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093214A1 (en) | 2015-12-03 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Mesoionic halogenated 3-(acetyl)-1-[(1,3-thiazol-5-yl)methyl]-1h-imidazo[1,2-a]pyridin-4-ium-2-olate derivatives and related compounds as insecticides |
| WO2017121699A1 (en) | 2016-01-15 | 2017-07-20 | Bayer Cropscience Aktiengesellschaft | Method for the preparation of substituted 2-aryl-ethanols |
| WO2017137338A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituierted 2-(het)aryl-imidazolyl-carboxyamides as pest control agents |
| WO2017137339A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituted 2-oxyimidazolyl-carboxamides as pest control agents |
| WO2017144341A1 (en) | 2016-02-23 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017157735A1 (en) | 2016-03-15 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2017157885A1 (en) | 2016-03-16 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | N-(cyanobenzyl)-6-(cyclopropyl-carbonylamino)-4-(phenyl)-pyridine-2-carboxamide derivatives and related compounds as pesticides and plant protection agents |
| WO2017174414A1 (en) | 2016-04-05 | 2017-10-12 | Bayer Cropscience Aktiengesellschaft | Naphthaline-derivatives as pest control agents |
| WO2017178416A1 (en) | 2016-04-15 | 2017-10-19 | Bayer Animal Health Gmbh | Pyrazolopyrimidine derivatives |
| WO2017186536A1 (en) | 2016-04-25 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Substituted 2-alkylimidazolyl-carboxamides as pest control agents |
| EP3241830A1 (en) | 2016-05-04 | 2017-11-08 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pesticides |
| EP3245865A1 (en) | 2016-05-17 | 2017-11-22 | Bayer CropScience Aktiengesellschaft | Method for increasing yield in brassicaceae |
| WO2017198449A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in brassicaceae |
| WO2017198451A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in small grain cereals such as wheat and rice |
| WO2017198452A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in soybean |
| WO2017198453A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in potato, tomato or alfalfa |
| WO2017198450A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in maize |
| WO2017198454A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in cotton |
| WO2017198455A2 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in beta spp. plants |
| WO2018015289A1 (en) | 2016-07-19 | 2018-01-25 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018019937A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Formulation comprising a beneficial p. bilaii strain and talc for use in seed treatment |
| WO2018029102A1 (en) | 2016-08-10 | 2018-02-15 | Bayer Cropscience Aktiengesellschaft | Substituted 2-heterocyclyl imidazolyl-carboxamides as pest control agents |
| EP3284739A1 (en) | 2017-07-19 | 2018-02-21 | Bayer CropScience Aktiengesellschaft | Substituted (het) aryl compounds as pesticides |
| WO2018033455A1 (en) | 2016-08-15 | 2018-02-22 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018050825A1 (en) | 2016-09-19 | 2018-03-22 | Bayer Cropscience Aktiengesellschaft | Pyrazolo [1,5-a]pyridine derivatives and their use as pesticides |
| EP3305786A2 (en) | 2018-01-22 | 2018-04-11 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pesticides |
| WO2018065292A1 (en) | 2016-10-06 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018065288A1 (en) | 2016-10-07 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-[2-phenyl-1-(sulfonyl-methyl)-vinyl]-imidazo-[4,5-b] pyridine derivatives and related compounds as pesticides in plant protection |
| WO2018083288A1 (en) | 2016-11-07 | 2018-05-11 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2018087036A1 (en) | 2016-11-11 | 2018-05-17 | Bayer Animal Health Gmbh | New anthelmintic quinoline-3-carboxamide derivatives |
| WO2018095953A1 (en) | 2016-11-23 | 2018-05-31 | Bayer Cropscience Aktiengesellschaft | 2-[3-(alkylsulfonyl)-2h-indazol-2-yl]-3h-imidazo[4,5-b]pyridine derivatives and similar compounds as pesticides |
| WO2018108730A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018108791A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Thiadiazole derivatives as pesticides |
| WO2018130437A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018130443A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018138050A1 (en) | 2017-01-26 | 2018-08-02 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclene derivatives as pest control agents |
| WO2018141954A1 (en) | 2017-02-06 | 2018-08-09 | Bayer Aktiengesellschaft | Aryl or heteroaryl-substituted imidazo pyridine derivatives and their use as pesticides |
| EP3369320A1 (en) | 2017-03-02 | 2018-09-05 | Bayer CropScience Aktiengesellschaft | Agent for controlling bugs |
| WO2018189077A1 (en) | 2017-04-12 | 2018-10-18 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018192872A1 (en) | 2017-04-21 | 2018-10-25 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2018197257A1 (en) | 2017-04-24 | 2018-11-01 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic-compound derivatives as pest control agents |
| WO2018197692A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Aktiengesellschaft | Heteroarylphenylaminoquinolines and analogues |
| WO2018197401A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Animal Health Gmbh | New bicyclic pyrazole derivatives |
| WO2018202501A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202715A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylbenzylbenzimidazoles and analogues |
| WO2018202706A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylheteroaryloxyquinolines and analogues |
| WO2018202494A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202712A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylphenoxyquinolines and analogues |
| WO2018202524A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | 2-{[2-(phenyloxymethyl)pyridin-5-yl]oxy}-ethanamin-derivatives and related compounds as pest-control agents e.g. for the protection of plants |
| WO2018202525A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | Phenoxyethanamine derivatives for controlling pests |
| WO2019002132A1 (en) | 2017-06-30 | 2019-01-03 | Bayer Animal Health Gmbh | New azaquinoline derivatives |
| WO2019025341A1 (en) | 2017-08-04 | 2019-02-07 | Bayer Animal Health Gmbh | Quinoline derivatives for treating infections with helminths |
| WO2019035881A1 (en) | 2017-08-17 | 2019-02-21 | Bayer Cropscience Lp | Liquid fertilizer-dispersible compositions and methods thereof |
| WO2019038195A1 (en) | 2017-08-22 | 2019-02-28 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019059412A1 (en) | 2017-09-20 | 2019-03-28 | Mitsui Chemicals Agro, Inc. | Prolonged ectoparasite-controlling agent for animal |
| WO2019068572A1 (en) | 2017-10-04 | 2019-04-11 | Bayer Aktiengesellschaft | Derivatives of heterocyclic compounds as pest control agents |
| EP3473100A1 (en) | 2017-10-18 | 2019-04-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076749A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076752A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076751A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076754A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076750A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019092086A1 (en) | 2017-11-13 | 2019-05-16 | Bayer Aktiengesellschaft | Tetrazolylpropyl derivatives and their use as fungicides |
| WO2019105875A1 (en) | 2017-11-28 | 2019-06-06 | Bayer Aktiengesellschaft | Heterocyclic compounds as pesticides |
| WO2019105871A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Aktiengesellschaft | Nitrogenous heterocycles as a pesticide |
| WO2019122319A1 (en) | 2017-12-21 | 2019-06-27 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylheteroaryloxyquinolines and analogues |
| WO2019155066A1 (en) | 2018-02-12 | 2019-08-15 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2019162174A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019162228A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | 1-(5-substituted imidazol-1-yl)but-3-en derivatives and their use as fungicides |
| WO2019170626A1 (en) | 2018-03-08 | 2019-09-12 | Bayer Aktiengesellschaft | Use of heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides in plant protection |
| WO2019175046A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| EP3545764A1 (en) | 2019-02-12 | 2019-10-02 | Bayer AG | Crystal form of 2-({2-fluoro-4-methyl-5-[(r)-(2,2,2-trifluoroethyl)sulfinyl]phenyl}imino)-3-(2,2,2- trifluoroethyl)-1,3-thiazolidin-4-one |
| WO2019197468A1 (en) | 2018-04-12 | 2019-10-17 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| WO2019197615A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with fungicides, insecticides and acaricidal properties |
| WO2019197652A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Solid formulation of insecticidal mixtures |
| WO2019197371A1 (en) | 2018-04-10 | 2019-10-17 | Bayer Aktiengesellschaft | Oxadiazoline derivatives |
| WO2019197612A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Cropscience Aktiengesellschaft | Use of tetramic acid derivatives for controlling pests by watering or droplet application |
| WO2019197620A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Cropscience Aktiengesellschaft | Use of tetramic acid derivatives for controlling specific insects |
| WO2019197623A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, fungicidal and acaricidal properties |
| WO2019197617A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Cropscience Aktiengesellschaft | Use of tetramic acid derivatives for controlling animal pests by watering, drip application plant hole treatment or furrow application |
| WO2019201835A1 (en) | 2018-04-17 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019202077A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019201921A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019206799A1 (en) | 2018-04-25 | 2019-10-31 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| EP3564225A1 (en) | 2019-03-21 | 2019-11-06 | Bayer Aktiengesellschaft | Crystalline form of spiromesifen |
| WO2019215182A1 (en) | 2018-05-09 | 2019-11-14 | Bayer Animal Health Gmbh | New quinoline derivatives |
| WO2019224143A1 (en) | 2018-05-24 | 2019-11-28 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, nematicidal and acaricidal properties |
| EP3586630A1 (en) | 2018-06-28 | 2020-01-01 | Bayer AG | Active compound combinations having insecticidal/acaricidal properties |
| WO2020005678A1 (en) | 2018-06-25 | 2020-01-02 | Bayer Cropscience Lp | Seed treatment method |
| WO2020007905A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020020816A1 (en) | 2018-07-26 | 2020-01-30 | Bayer Aktiengesellschaft | Novel triazole derivatives |
| WO2020020813A1 (en) | 2018-07-25 | 2020-01-30 | Bayer Aktiengesellschaft | Fungicidal active compound combinations |
| EP3608311A1 (en) | 2019-06-28 | 2020-02-12 | Bayer AG | Crystalline form a of n-[4-chloro-3-[(1-cyanocyclopropyl)carbamoyl]phenyl]-2-methyl-4-methylsulfonyl-5-(1,1,2,2,2-pentafluoroethyl)pyrazole-3-carboxamide |
| WO2020043650A1 (en) | 2018-08-29 | 2020-03-05 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3620052A1 (en) | 2018-12-12 | 2020-03-11 | Bayer Aktiengesellschaft | Use of phenoxypyridinyl-substituted (1h-1,2,4-triazol-1-yl)alcohols for controlling fungicidal diseases in maize |
| WO2020053282A1 (en) | 2018-09-13 | 2020-03-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2020057939A1 (en) | 2018-09-17 | 2020-03-26 | Bayer Aktiengesellschaft | Use of the fungicide isoflucypram for controlling claviceps purpurea and reducing sclerotia in cereals |
| WO2020070050A1 (en) | 2018-10-01 | 2020-04-09 | Bayer Aktiengesellschaft | Fungicidal 5-substituted imidazol-1-yl carbinol derivatives |
| EP3636644A1 (en) | 2018-10-11 | 2020-04-15 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2020079232A1 (en) | 2018-10-20 | 2020-04-23 | Bayer Aktiengesellschaft | Oxetanylphenoxyquinolines and analogues |
| WO2020079173A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Pyridylphenylaminoquinolines and analogues |
| WO2020079167A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Heteroarylaminoquinolines and analogues |
| WO2020078839A1 (en) | 2018-10-16 | 2020-04-23 | Bayer Aktiengesellschaft | Active substance combinations |
| EP3643711A1 (en) | 2018-10-24 | 2020-04-29 | Bayer Animal Health GmbH | New anthelmintic compounds |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020114932A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2020114934A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicide compositions |
| EP3669652A1 (en) | 2018-12-21 | 2020-06-24 | Bayer AG | Active compound combination |
| WO2020127974A1 (en) | 2018-12-21 | 2020-06-25 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as new antifungal agents |
| WO2020126980A1 (en) | 2018-12-18 | 2020-06-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| EP3679790A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679792A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679793A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679789A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679791A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3701796A1 (en) | 2019-08-08 | 2020-09-02 | Bayer AG | Active compound combinations |
| WO2020173860A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Fused bicyclic heterocycle derivatives as pesticides |
| WO2020173861A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2020178067A1 (en) | 2019-03-01 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020178307A1 (en) | 2019-03-05 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combination |
| EP3708565A1 (en) | 2020-03-04 | 2020-09-16 | Bayer AG | Pyrimidinyloxyphenylamidines and the use thereof as fungicides |
| WO2020182929A1 (en) | 2019-03-13 | 2020-09-17 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| WO2020187656A1 (en) | 2019-03-15 | 2020-09-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3725788A1 (en) | 2019-04-15 | 2020-10-21 | Bayer AG | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020225434A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading ulv formulations for agrochemical compounds ii |
| WO2020225242A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020229398A1 (en) | 2019-05-14 | 2020-11-19 | Bayer Aktiengesellschaft | (1-alkenyl)-substituted pyrazoles and triazoles as pest control agents |
| WO2020231751A1 (en) | 2019-05-10 | 2020-11-19 | Bayer Cropscience Lp | Active compound combinations |
| EP3750888A1 (en) | 2019-06-12 | 2020-12-16 | Bayer Aktiengesellschaft | Crystalline form a of 1,4-dimethyl-2-[2-(pyridin-3-yl)-2h-indazol-5-yl]-1,2,4-triazolidine-3,5-dione |
| WO2020254487A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254488A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and use thereof as fungicides |
| WO2020254489A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Benzylphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254486A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254492A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254493A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Thienylhydroxyisoxazolines and derivatives thereof |
| WO2020254490A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Phenoxyphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254494A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2020263812A1 (en) | 2019-06-24 | 2020-12-30 | Auburn University | A bacillus strain and methods of its use for plant growth promotion |
| WO2021001273A1 (en) | 2019-07-04 | 2021-01-07 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2021001331A1 (en) | 2019-07-03 | 2021-01-07 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides and derivatives thereof as microbicides |
| WO2021009229A1 (en) | 2019-07-16 | 2021-01-21 | Syngenta Crop Protection Ag | Herbicidal compounds |
| WO2021013719A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013720A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013721A1 (en) | 2019-07-22 | 2021-01-28 | Bayer Aktiengesellschaft | 5-amino substituted pyrazoles and triazoles as pest control agents |
| EP3771714A1 (en) | 2019-07-30 | 2021-02-03 | Bayer AG | Nitrogen-containing heterocycles as pesticides |
| WO2021018839A1 (en) | 2019-07-30 | 2021-02-04 | Bayer Animal Health Gmbh | Isoquinoline derivatives and their use for the treatment of parasitic infections |
| WO2021058659A1 (en) | 2019-09-26 | 2021-04-01 | Bayer Aktiengesellschaft | Rnai-mediated pest control |
| WO2021069567A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069575A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Animal Health Gmbh | Heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021069569A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021089673A1 (en) | 2019-11-07 | 2021-05-14 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2021097162A1 (en) | 2019-11-13 | 2021-05-20 | Bayer Cropscience Lp | Beneficial combinations with paenibacillus |
| WO2021099303A1 (en) | 2019-11-18 | 2021-05-27 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021105091A1 (en) | 2019-11-25 | 2021-06-03 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| WO2021122986A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Thienyloxazolones and analogues |
| EP3845304A1 (en) | 2019-12-30 | 2021-07-07 | Bayer AG | Capsule suspension concentrates based on polyisocyanates and biodegradable amine based cross-linker |
| EP3868207A1 (en) | 2020-02-24 | 2021-08-25 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved activity in soil and leaf applications |
| WO2021165195A1 (en) | 2020-02-18 | 2021-08-26 | Bayer Aktiengesellschaft | Heteroaryl-triazole compounds as pesticides |
| WO2021204930A1 (en) | 2020-04-09 | 2021-10-14 | Bayer Animal Health Gmbh | Substituted condensed azines as anthelmintic compounds |
| WO2021209363A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209490A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Cyclaminephenylaminoquinolines as fungicides |
| WO2021209364A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209365A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209366A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209368A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021213978A1 (en) | 2020-04-21 | 2021-10-28 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocyclic derivatives as pest control agents |
| WO2021224323A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021224220A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Pyridine (thio)amides as fungicidal compounds |
| WO2021228734A1 (en) | 2020-05-12 | 2021-11-18 | Bayer Aktiengesellschaft | Triazine and pyrimidine (thio)amides as fungicidal compounds |
| WO2021233861A1 (en) | 2020-05-19 | 2021-11-25 | Bayer Aktiengesellschaft | Azabicyclic(thio)amides as fungicidal compounds |
| EP3915971A1 (en) | 2020-12-16 | 2021-12-01 | Bayer Aktiengesellschaft | Phenyl-s(o)n-phenylamidines and the use thereof as fungicides |
| EP3915371A1 (en) | 2020-11-04 | 2021-12-01 | Bayer AG | Active compound combinations and fungicide compositions comprising those |
| WO2021245087A1 (en) | 2020-06-04 | 2021-12-09 | Bayer Aktiengesellschaft | Heterocyclyl pyrimidines and triazines as novel fungicides |
| WO2021249995A1 (en) | 2020-06-10 | 2021-12-16 | Bayer Aktiengesellschaft | Azabicyclyl-substituted heterocycles as fungicides |
| WO2021255071A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | 3-(pyridazin-4-yl)-5,6-dihydro-4h-1,2,4-oxadiazine derivatives as fungicides for crop protection |
| WO2021255170A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255169A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255089A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines and 1,3,4-oxadiazole pyridines as fungicides |
| WO2021255091A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as fungicides |
| EP3929189A1 (en) | 2020-06-25 | 2021-12-29 | Bayer Animal Health GmbH | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021260017A1 (en) | 2020-06-26 | 2021-12-30 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates comprising biodegradable ester groups |
| WO2022002818A1 (en) | 2020-07-02 | 2022-01-06 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2022033991A1 (en) | 2020-08-13 | 2022-02-17 | Bayer Aktiengesellschaft | 5-amino substituted triazoles as pest control agents |
| WO2022053453A1 (en) | 2020-09-09 | 2022-03-17 | Bayer Aktiengesellschaft | Azole carboxamide as pest control agents |
| WO2022058327A1 (en) | 2020-09-15 | 2022-03-24 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| EP3974414A1 (en) | 2020-09-25 | 2022-03-30 | Bayer AG | 5-amino substituted pyrazoles and triazoles as pesticides |
| EP3994994A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994990A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994991A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| EP3994986A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| EP3994989A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, rainfastness and uptake properties |
| EP3994987A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift and uptake properties |
| EP3994985A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| EP3994995A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994993A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| EP3994992A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| EP3994988A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and rainfastness properties |
| WO2022129188A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | 1,2,4-oxadiazol-3-yl pyrimidines as fungicides |
| WO2022129190A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | (hetero)aryl substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129196A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | Heterobicycle substituted 1,2,4-oxadiazoles as fungicides |
| WO2022152728A1 (en) | 2021-01-15 | 2022-07-21 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP4036083A1 (en) | 2021-02-02 | 2022-08-03 | Bayer Aktiengesellschaft | 5-oxy substituted heterocycles as pesticides |
| WO2022207496A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022207494A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022233777A1 (en) | 2021-05-06 | 2022-11-10 | Bayer Aktiengesellschaft | Alkylamide substituted, annulated imidazoles and use thereof as insecticides |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2022238391A1 (en) | 2021-05-12 | 2022-11-17 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pest control agents |
| WO2023017120A1 (en) | 2021-08-13 | 2023-02-16 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2023025682A1 (en) | 2021-08-25 | 2023-03-02 | Bayer Aktiengesellschaft | Novel pyrazinyl-triazole compounds as pesticides |
| EP4144739A1 (en) | 2021-09-02 | 2023-03-08 | Bayer Aktiengesellschaft | Anellated pyrazoles as parasiticides |
| EP4148052A1 (en) | 2021-09-09 | 2023-03-15 | Bayer Animal Health GmbH | New quinoline derivatives |
| EP4265110A1 (en) | 2022-04-20 | 2023-10-25 | Bayer AG | Water dispersible granules with low melting active ingredients prepared by extrusion |
| WO2023205602A1 (en) | 2022-04-18 | 2023-10-26 | Basf Corporation | High-load agricultural formulations and methods of making same |
| WO2023213626A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Use of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine for controlling unwanted microorganisms |
| WO2023213670A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Crystalline forms of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine |
| WO2023217619A1 (en) | 2022-05-07 | 2023-11-16 | Bayer Aktiengesellschaft | Low drift aqueous liquid formulations for low, medium, and high spray volume application |
| WO2023237444A1 (en) | 2022-06-06 | 2023-12-14 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295683A1 (en) | 2022-06-21 | 2023-12-27 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295688A1 (en) | 2022-09-28 | 2023-12-27 | Bayer Aktiengesellschaft | Active compound combination |
| WO2024013015A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024013016A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024068518A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-heteroaryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068520A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068519A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068473A1 (en) | 2022-09-27 | 2024-04-04 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2024068517A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| EP4353082A1 (en) | 2022-10-14 | 2024-04-17 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024104643A1 (en) | 2022-11-17 | 2024-05-23 | Bayer Aktiengesellschaft | Use of isotianil for controlling plasmodiophora brassica |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017060204A1 (en) * | 2015-10-06 | 2017-04-13 | Bayer Cropscience Aktiengesellschaft | New alkynyl-substituted 3-phenylpyrrolidine-2,4-diones and use thereof as herbicides |
| GB201803736D0 (en) * | 2018-03-08 | 2018-04-25 | Syngenta Participations Ag | Improvements in or relating to organic compounds |
| GB201804002D0 (en) | 2018-03-13 | 2018-04-25 | Syngenta Participations Ag | Improvements in or relating to organic compounds |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0365484A1 (en) | 1988-10-20 | 1990-04-25 | Ciba-Geigy Ag | Sulfamoyl phenyl ureas |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995001358A1 (en) | 1993-07-02 | 1995-01-12 | Bayer Aktiengesellschaft | Substituted spiroheterocyclic 1h-3-arylpyrrolidine-2,4-dione derivatives, methods of preparing them and their use as pest-control agents |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU666040B2 (en) * | 1992-10-28 | 1996-01-25 | Bayer Aktiengesellschaft | Substituted 1-H-3-aryl-pyrrolidine-2,4-dione derivatives |
| DE19749720A1 (en) * | 1997-11-11 | 1999-05-12 | Bayer Ag | New substituted phenylketoenols |
| DE10016544A1 (en) * | 2000-04-03 | 2001-10-11 | Bayer Ag | New phenyl-substituted cyclic keto-enol compounds useful e.g. as insecticides, acaricides, nematocides, acaricides, herbicides, ectoparasiticides, antifouling agents or intermediates |
| DE10354629A1 (en) * | 2003-11-22 | 2005-06-30 | Bayer Cropscience Ag | 2-ethyl-4,6-dimethyl-phenyl substituted spirocyclic tetramic acid derivatives |
| DE102004014620A1 (en) * | 2004-03-25 | 2005-10-06 | Bayer Cropscience Ag | 2,4,6-phenyl-substituted cyclic ketoenols |
| GB0720126D0 (en) * | 2007-10-15 | 2007-11-28 | Syngenta Participations Ag | Chemical compounds |
-
2008
- 2008-11-06 GB GBGB0820344.0A patent/GB0820344D0/en not_active Ceased
-
2009
- 2009-10-28 JP JP2011533709A patent/JP5592386B2/en not_active Expired - Fee Related
- 2009-10-28 US US13/128,133 patent/US8722578B2/en active Active
- 2009-10-28 CA CA2742552A patent/CA2742552A1/en not_active Abandoned
- 2009-10-28 WO PCT/EP2009/064214 patent/WO2010052161A2/en active Application Filing
- 2009-10-28 CN CN2009801441973A patent/CN102209469A/en active Pending
- 2009-10-28 AU AU2009312909A patent/AU2009312909A1/en not_active Abandoned
- 2009-10-28 EP EP09740705.0A patent/EP2352375B1/en active Active
- 2009-10-28 BR BRPI0921411-9A patent/BRPI0921411A2/en not_active IP Right Cessation
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0365484A1 (en) | 1988-10-20 | 1990-04-25 | Ciba-Geigy Ag | Sulfamoyl phenyl ureas |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995001358A1 (en) | 1993-07-02 | 1995-01-12 | Bayer Aktiengesellschaft | Substituted spiroheterocyclic 1h-3-arylpyrrolidine-2,4-dione derivatives, methods of preparing them and their use as pest-control agents |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
Non-Patent Citations (2)
| Title |
|---|
| "Pesticide Manual", 2000, BRITISH CROP PROTECTION COUNCIL, pages: R-29148 |
| P.B. GOLDSBROUGH ET AL., PLANT PHYSIOLOGY, vol. 130, 2002, pages 1497 - 1505 |
Cited By (310)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012511541A (en) * | 2008-12-12 | 2012-05-24 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | Spiroheterocyclic N-oxypiperidine as a pesticide |
| WO2011067131A1 (en) * | 2009-12-04 | 2011-06-09 | Syngenta Participations Ag | Spiroheterocyclic dione derivatives used as pesticides |
| US9809542B2 (en) | 2010-02-10 | 2017-11-07 | Bayer Intellectual Property Gmbh | Spiroheterocyclically substituted tetramic acid derivatives |
| WO2011098440A2 (en) | 2010-02-10 | 2011-08-18 | Bayer Cropscience Ag | Biphenyl substituted cyclical keto-enols |
| WO2011098443A1 (en) | 2010-02-10 | 2011-08-18 | Bayer Cropscience Ag | Spiroheterocyclical substituted tetramic acid derivatives |
| US8901038B2 (en) | 2010-02-10 | 2014-12-02 | Bayer Cropscience Ag | Biphenyl-substituted cyclic ketoenols |
| JP2013519643A (en) * | 2010-02-10 | 2013-05-30 | バイエル・インテレクチユアル・プロパテイー・ゲー・エム・ベー・ハー | Spiroheterocyclic substituted tetramic acid derivatives |
| WO2011098433A1 (en) | 2010-02-15 | 2011-08-18 | Bayer Schering Pharma Aktiengesellschaft | Cyclic keto-enols for therapy |
| WO2011131623A1 (en) | 2010-04-20 | 2011-10-27 | Bayer Cropscience Ag | Insecticidal and/or herbicidal composition having improved activity on the basis of spiro-heterocyclically substituted tetramic acid derivatives |
| US8889592B2 (en) | 2010-04-20 | 2014-11-18 | Bayer Cropscience Ag | Insecticidal and/or herbicidal composition having improved activity based on spiroheterocyclically substituted tetramic acid derivatives |
| CN103002740A (en) * | 2010-05-31 | 2013-03-27 | 先正达参股股份有限公司 | Pesticidal compositions |
| US20130150394A1 (en) * | 2010-05-31 | 2013-06-13 | Syngenta Crop Protection Llc | Pesticidal compositions |
| WO2011151199A1 (en) * | 2010-05-31 | 2011-12-08 | Syngenta Participations Ag | Spiroheterocyclic pyrrolidine derivatives based pesticides |
| US9462808B2 (en) | 2010-05-31 | 2016-10-11 | Syngenta Participants Ag | Pesticidal compositions |
| CN103002731B (en) * | 2010-05-31 | 2015-12-02 | 先正达参股股份有限公司 | Pesticidal combination |
| US9018135B2 (en) | 2010-05-31 | 2015-04-28 | Syngenta Participations Ag | Pesticidal compositions |
| WO2011151248A1 (en) * | 2010-05-31 | 2011-12-08 | Syngenta Participations Ag | Pesticidal compositions |
| WO2011151247A3 (en) * | 2010-05-31 | 2012-03-01 | Syngenta Participations Ag | Pesticidal compositions |
| CN103002731A (en) * | 2010-05-31 | 2013-03-27 | 先正达参股股份有限公司 | Pesticidal compositions |
| CN103003239A (en) * | 2010-05-31 | 2013-03-27 | 先正达参股股份有限公司 | Spiroheterocyclic pyrrolidine derivatives based pesticides |
| JP2013528609A (en) * | 2010-05-31 | 2013-07-11 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | Agrochemical composition |
| JP2013527208A (en) * | 2010-05-31 | 2013-06-27 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | How to grow crops |
| JP2013527210A (en) * | 2010-05-31 | 2013-06-27 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | Agrochemical composition |
| US9272997B2 (en) | 2011-01-25 | 2016-03-01 | Bayer Intellectual Property Gmbh | Process for the preparation of 1-H-pyrrolidine-2,4-dione derivatives |
| US20120190865A1 (en) * | 2011-01-25 | 2012-07-26 | Bayer Cropscience Ag | Process for the Preparation of 1-H-pyrrolidine-2,4-dione Derivatives |
| WO2012101047A1 (en) | 2011-01-25 | 2012-08-02 | Bayer Cropscience Ag | Method for producing 1-h-pyrrolidine-2,4-dione derivatives |
| EP3372580A1 (en) | 2011-01-25 | 2018-09-12 | Bayer CropScience Aktiengesellschaft | Method for the preparation of 1-h-pyrrolidin-2,4-dione-derivatives |
| US8859782B2 (en) | 2011-01-25 | 2014-10-14 | Bayer Cropscience Ag | Process for the preparation of 1-H-pyrrolidine-2,4-dione derivatives |
| DE102011011040A1 (en) | 2011-02-08 | 2012-08-09 | Bayer Pharma Aktiengesellschaft | (5s, 8s) -3- (4'-chloro-3'-fluoro-4-methylbiphenyl-3-yl) -4-hydroxy-8-methoxy-1-azaspiro [4.5] dec-3-en-2- on (compound A) for therapy |
| WO2012110518A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Pharma Aktiengesellschaft | Substituted 3-(biphenyl-3-yl)-8,8-difluoro-4-hydroxy-1-azaspiro[4.5]dec-3-en-2-ones for therapy |
| WO2012110519A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Cropscience Ag | Substituted 3-(biphenyl-3-yl)-8,8-difluoro-hydroxy-1-azaspiro[4.5]dec-3-en-2-ones for therapy and halogen-substituted spirocyclic ketoenols |
| US8946124B2 (en) | 2011-02-17 | 2015-02-03 | Bayer Intellectual Property Gmbh | Substituted 3-(biphenyl-3-yl)-8,8-difluoro-4-hydroxy-1-azaspiro[4.5]dec-3-en-2-ones for therapy and halogen-substituted spirocyclic ketoenols |
| WO2012116960A1 (en) | 2011-03-01 | 2012-09-07 | Bayer Cropscience Ag | 2-acyloxy-pyrrolin-4-ones |
| US9204640B2 (en) | 2011-03-01 | 2015-12-08 | Bayer Intellectual Property Gmbh | 2-acyloxy-pyrrolin-4-ones |
| DE102011080406A1 (en) | 2011-08-04 | 2013-02-07 | Bayer Pharma AG | Substituted 3- (biphenyl-3-yl) -4-hydroxy-8-methoxy-1-azaspiro8 [4.5] dec-3-ene-2-ones |
| WO2013017600A1 (en) | 2011-08-04 | 2013-02-07 | Bayer Intellectual Property Gmbh | Substituted 3-(biphenyl-3-yl)-4-hydroxy-8-methoxy-1-azaspiro[4.5]dec-3-en-2-one |
| DE102011080405A1 (en) | 2011-08-04 | 2013-02-07 | Bayer Pharma AG | New substituted 3-biphenyl-3-yl-8,8-difluoro-4-hydroxy-1-azaspiro(4.5)dec-3-en-2-one derivatives useful for prophylaxis or therapy of tumor diseases comprising breast cancer, prostate cancer, colorectal cancer or non-small cell lung cancer |
| WO2013079672A1 (en) * | 2011-11-30 | 2013-06-06 | Syngenta Limited | Herbicidal uses of substituted spiroheterocyclic pyrrolidine dione derivatives, and herbicidal compositions comprising them |
| KR101993420B1 (en) | 2011-11-30 | 2019-06-26 | 신젠타 파티서페이션즈 아게 | Pesticidal mixtures including spiroheterocyclic pyrrolidine diones |
| KR20140098226A (en) * | 2011-11-30 | 2014-08-07 | 신젠타 파티서페이션즈 아게 | Pesticidal mixtures including spiroheterocyclic pyrrolidine diones |
| WO2017072039A1 (en) | 2015-10-26 | 2017-05-04 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093180A1 (en) | 2015-12-01 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093214A1 (en) | 2015-12-03 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Mesoionic halogenated 3-(acetyl)-1-[(1,3-thiazol-5-yl)methyl]-1h-imidazo[1,2-a]pyridin-4-ium-2-olate derivatives and related compounds as insecticides |
| WO2017121699A1 (en) | 2016-01-15 | 2017-07-20 | Bayer Cropscience Aktiengesellschaft | Method for the preparation of substituted 2-aryl-ethanols |
| US10519085B2 (en) | 2016-01-15 | 2019-12-31 | Bayer Cropscience Aktiengesellschaft | Process for preparing substituted 2-arylethanols |
| WO2017137338A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituierted 2-(het)aryl-imidazolyl-carboxyamides as pest control agents |
| WO2017137339A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituted 2-oxyimidazolyl-carboxamides as pest control agents |
| WO2017144341A1 (en) | 2016-02-23 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017157735A1 (en) | 2016-03-15 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2017157885A1 (en) | 2016-03-16 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | N-(cyanobenzyl)-6-(cyclopropyl-carbonylamino)-4-(phenyl)-pyridine-2-carboxamide derivatives and related compounds as pesticides and plant protection agents |
| WO2017174414A1 (en) | 2016-04-05 | 2017-10-12 | Bayer Cropscience Aktiengesellschaft | Naphthaline-derivatives as pest control agents |
| WO2017178416A1 (en) | 2016-04-15 | 2017-10-19 | Bayer Animal Health Gmbh | Pyrazolopyrimidine derivatives |
| WO2017186536A1 (en) | 2016-04-25 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Substituted 2-alkylimidazolyl-carboxamides as pest control agents |
| EP3241830A1 (en) | 2016-05-04 | 2017-11-08 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pesticides |
| WO2017198450A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in maize |
| WO2017198449A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in brassicaceae |
| WO2017198452A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in soybean |
| WO2017198453A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in potato, tomato or alfalfa |
| WO2017198451A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in small grain cereals such as wheat and rice |
| WO2017198455A2 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in beta spp. plants |
| WO2017198454A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in cotton |
| EP3245865A1 (en) | 2016-05-17 | 2017-11-22 | Bayer CropScience Aktiengesellschaft | Method for increasing yield in brassicaceae |
| WO2018015289A1 (en) | 2016-07-19 | 2018-01-25 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018019937A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Formulation comprising a beneficial p. bilaii strain and talc for use in seed treatment |
| WO2018029102A1 (en) | 2016-08-10 | 2018-02-15 | Bayer Cropscience Aktiengesellschaft | Substituted 2-heterocyclyl imidazolyl-carboxamides as pest control agents |
| WO2018033455A1 (en) | 2016-08-15 | 2018-02-22 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018050825A1 (en) | 2016-09-19 | 2018-03-22 | Bayer Cropscience Aktiengesellschaft | Pyrazolo [1,5-a]pyridine derivatives and their use as pesticides |
| WO2018065292A1 (en) | 2016-10-06 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018065288A1 (en) | 2016-10-07 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-[2-phenyl-1-(sulfonyl-methyl)-vinyl]-imidazo-[4,5-b] pyridine derivatives and related compounds as pesticides in plant protection |
| WO2018083288A1 (en) | 2016-11-07 | 2018-05-11 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| US11505545B2 (en) | 2016-11-11 | 2022-11-22 | Bayer Animal Health Gmbh | Anthelmintic quinoline-3-carboxamide derivatives |
| US10889573B2 (en) | 2016-11-11 | 2021-01-12 | Bayer Animal Health Gmbh | Anthelmintic quinoline-3-carboxamide derivatives |
| WO2018087036A1 (en) | 2016-11-11 | 2018-05-17 | Bayer Animal Health Gmbh | New anthelmintic quinoline-3-carboxamide derivatives |
| WO2018095953A1 (en) | 2016-11-23 | 2018-05-31 | Bayer Cropscience Aktiengesellschaft | 2-[3-(alkylsulfonyl)-2h-indazol-2-yl]-3h-imidazo[4,5-b]pyridine derivatives and similar compounds as pesticides |
| WO2018108730A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018108791A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Thiadiazole derivatives as pesticides |
| WO2018130437A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018130443A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018138050A1 (en) | 2017-01-26 | 2018-08-02 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclene derivatives as pest control agents |
| WO2018141954A1 (en) | 2017-02-06 | 2018-08-09 | Bayer Aktiengesellschaft | Aryl or heteroaryl-substituted imidazo pyridine derivatives and their use as pesticides |
| EP3369320A1 (en) | 2017-03-02 | 2018-09-05 | Bayer CropScience Aktiengesellschaft | Agent for controlling bugs |
| WO2018189077A1 (en) | 2017-04-12 | 2018-10-18 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018192872A1 (en) | 2017-04-21 | 2018-10-25 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2018197257A1 (en) | 2017-04-24 | 2018-11-01 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic-compound derivatives as pest control agents |
| WO2018197401A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Animal Health Gmbh | New bicyclic pyrazole derivatives |
| WO2018197692A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Aktiengesellschaft | Heteroarylphenylaminoquinolines and analogues |
| US11130768B2 (en) | 2017-04-27 | 2021-09-28 | Bayer Animal Health Gmbh | Bicyclic pyrazole derivatives |
| WO2018202501A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202494A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202715A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylbenzylbenzimidazoles and analogues |
| WO2018202706A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylheteroaryloxyquinolines and analogues |
| WO2018202712A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylphenoxyquinolines and analogues |
| WO2018202524A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | 2-{[2-(phenyloxymethyl)pyridin-5-yl]oxy}-ethanamin-derivatives and related compounds as pest-control agents e.g. for the protection of plants |
| WO2018202525A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | Phenoxyethanamine derivatives for controlling pests |
| US11827616B2 (en) | 2017-05-04 | 2023-11-28 | Discovery Purchaser Corporation | Heterocyclic compounds as pesticides |
| WO2019002132A1 (en) | 2017-06-30 | 2019-01-03 | Bayer Animal Health Gmbh | New azaquinoline derivatives |
| EP3284739A1 (en) | 2017-07-19 | 2018-02-21 | Bayer CropScience Aktiengesellschaft | Substituted (het) aryl compounds as pesticides |
| WO2019025341A1 (en) | 2017-08-04 | 2019-02-07 | Bayer Animal Health Gmbh | Quinoline derivatives for treating infections with helminths |
| WO2019035881A1 (en) | 2017-08-17 | 2019-02-21 | Bayer Cropscience Lp | Liquid fertilizer-dispersible compositions and methods thereof |
| WO2019038195A1 (en) | 2017-08-22 | 2019-02-28 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019059412A1 (en) | 2017-09-20 | 2019-03-28 | Mitsui Chemicals Agro, Inc. | Prolonged ectoparasite-controlling agent for animal |
| WO2019068572A1 (en) | 2017-10-04 | 2019-04-11 | Bayer Aktiengesellschaft | Derivatives of heterocyclic compounds as pest control agents |
| WO2019076754A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3473100A1 (en) | 2017-10-18 | 2019-04-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076751A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076749A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076750A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076752A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019092086A1 (en) | 2017-11-13 | 2019-05-16 | Bayer Aktiengesellschaft | Tetrazolylpropyl derivatives and their use as fungicides |
| WO2019105875A1 (en) | 2017-11-28 | 2019-06-06 | Bayer Aktiengesellschaft | Heterocyclic compounds as pesticides |
| WO2019105871A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Aktiengesellschaft | Nitrogenous heterocycles as a pesticide |
| WO2019122319A1 (en) | 2017-12-21 | 2019-06-27 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylheteroaryloxyquinolines and analogues |
| EP3305786A2 (en) | 2018-01-22 | 2018-04-11 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pesticides |
| WO2019155066A1 (en) | 2018-02-12 | 2019-08-15 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2019162174A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019162228A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | 1-(5-substituted imidazol-1-yl)but-3-en derivatives and their use as fungicides |
| WO2019170626A1 (en) | 2018-03-08 | 2019-09-12 | Bayer Aktiengesellschaft | Use of heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides in plant protection |
| WO2019175045A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019175046A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019197371A1 (en) | 2018-04-10 | 2019-10-17 | Bayer Aktiengesellschaft | Oxadiazoline derivatives |
| EP3904349A2 (en) | 2018-04-12 | 2021-11-03 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}heterocyclyl amide derivatives and similar compounds as pesticides |
| WO2019197468A1 (en) | 2018-04-12 | 2019-10-17 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| WO2019197623A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, fungicidal and acaricidal properties |
| WO2019197620A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Cropscience Aktiengesellschaft | Use of tetramic acid derivatives for controlling specific insects |
| WO2019197652A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Solid formulation of insecticidal mixtures |
| WO2019197612A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Cropscience Aktiengesellschaft | Use of tetramic acid derivatives for controlling pests by watering or droplet application |
| WO2019197617A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Cropscience Aktiengesellschaft | Use of tetramic acid derivatives for controlling animal pests by watering, drip application plant hole treatment or furrow application |
| WO2019197615A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with fungicides, insecticides and acaricidal properties |
| EP4039682A1 (en) | 2018-04-17 | 2022-08-10 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019201835A1 (en) | 2018-04-17 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019202077A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019201921A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019206799A1 (en) | 2018-04-25 | 2019-10-31 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| EP3919486A1 (en) | 2018-04-25 | 2021-12-08 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019215182A1 (en) | 2018-05-09 | 2019-11-14 | Bayer Animal Health Gmbh | New quinoline derivatives |
| WO2019224143A1 (en) | 2018-05-24 | 2019-11-28 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, nematicidal and acaricidal properties |
| WO2020005678A1 (en) | 2018-06-25 | 2020-01-02 | Bayer Cropscience Lp | Seed treatment method |
| EP3586630A1 (en) | 2018-06-28 | 2020-01-01 | Bayer AG | Active compound combinations having insecticidal/acaricidal properties |
| WO2020007904A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020007902A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| US11952359B2 (en) | 2018-07-05 | 2024-04-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020007905A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| US11884643B2 (en) | 2018-07-05 | 2024-01-30 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020020813A1 (en) | 2018-07-25 | 2020-01-30 | Bayer Aktiengesellschaft | Fungicidal active compound combinations |
| WO2020020816A1 (en) | 2018-07-26 | 2020-01-30 | Bayer Aktiengesellschaft | Novel triazole derivatives |
| WO2020043650A1 (en) | 2018-08-29 | 2020-03-05 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020053282A1 (en) | 2018-09-13 | 2020-03-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2020057939A1 (en) | 2018-09-17 | 2020-03-26 | Bayer Aktiengesellschaft | Use of the fungicide isoflucypram for controlling claviceps purpurea and reducing sclerotia in cereals |
| WO2020070050A1 (en) | 2018-10-01 | 2020-04-09 | Bayer Aktiengesellschaft | Fungicidal 5-substituted imidazol-1-yl carbinol derivatives |
| EP3636644A1 (en) | 2018-10-11 | 2020-04-15 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2020078839A1 (en) | 2018-10-16 | 2020-04-23 | Bayer Aktiengesellschaft | Active substance combinations |
| WO2020079167A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Heteroarylaminoquinolines and analogues |
| WO2020079173A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Pyridylphenylaminoquinolines and analogues |
| WO2020079232A1 (en) | 2018-10-20 | 2020-04-23 | Bayer Aktiengesellschaft | Oxetanylphenoxyquinolines and analogues |
| EP3643711A1 (en) | 2018-10-24 | 2020-04-29 | Bayer Animal Health GmbH | New anthelmintic compounds |
| WO2020083971A2 (en) | 2018-10-24 | 2020-04-30 | Bayer Animal Health Gmbh | New anthelmintic compounds |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020114934A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicide compositions |
| WO2020114932A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP3620052A1 (en) | 2018-12-12 | 2020-03-11 | Bayer Aktiengesellschaft | Use of phenoxypyridinyl-substituted (1h-1,2,4-triazol-1-yl)alcohols for controlling fungicidal diseases in maize |
| WO2020126980A1 (en) | 2018-12-18 | 2020-06-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| EP3669652A1 (en) | 2018-12-21 | 2020-06-24 | Bayer AG | Active compound combination |
| WO2020127974A1 (en) | 2018-12-21 | 2020-06-25 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as new antifungal agents |
| EP3679790A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679793A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679789A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679791A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679792A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3545764A1 (en) | 2019-02-12 | 2019-10-02 | Bayer AG | Crystal form of 2-({2-fluoro-4-methyl-5-[(r)-(2,2,2-trifluoroethyl)sulfinyl]phenyl}imino)-3-(2,2,2- trifluoroethyl)-1,3-thiazolidin-4-one |
| WO2020173860A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Fused bicyclic heterocycle derivatives as pesticides |
| WO2020173861A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2020178067A1 (en) | 2019-03-01 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020178307A1 (en) | 2019-03-05 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020182929A1 (en) | 2019-03-13 | 2020-09-17 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| WO2020187656A1 (en) | 2019-03-15 | 2020-09-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3564225A1 (en) | 2019-03-21 | 2019-11-06 | Bayer Aktiengesellschaft | Crystalline form of spiromesifen |
| WO2020212235A1 (en) | 2019-04-15 | 2020-10-22 | Bayer Animal Health Gmbh | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| EP3725788A1 (en) | 2019-04-15 | 2020-10-21 | Bayer AG | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020225440A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading and rainfastness ulv formulations |
| WO2020225438A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High uptake and rainfastness ulv formulations |
| WO2020225242A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020225437A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Ulv formulations with enhanced uptake |
| WO2020225428A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading ulv formulations for insecticides |
| WO2020225435A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading and uptake ulv formulations |
| WO2020225436A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading, uptake and rainfastness ulv formulations |
| WO2020225439A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Ulv formulations with enhanced rainfastness |
| WO2020225434A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading ulv formulations for agrochemical compounds ii |
| WO2020231751A1 (en) | 2019-05-10 | 2020-11-19 | Bayer Cropscience Lp | Active compound combinations |
| WO2020229398A1 (en) | 2019-05-14 | 2020-11-19 | Bayer Aktiengesellschaft | (1-alkenyl)-substituted pyrazoles and triazoles as pest control agents |
| EP3750888A1 (en) | 2019-06-12 | 2020-12-16 | Bayer Aktiengesellschaft | Crystalline form a of 1,4-dimethyl-2-[2-(pyridin-3-yl)-2h-indazol-5-yl]-1,2,4-triazolidine-3,5-dione |
| WO2020254488A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and use thereof as fungicides |
| WO2020254493A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Thienylhydroxyisoxazolines and derivatives thereof |
| WO2020254490A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Phenoxyphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254494A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2020254492A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254486A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254487A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254489A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Benzylphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020263812A1 (en) | 2019-06-24 | 2020-12-30 | Auburn University | A bacillus strain and methods of its use for plant growth promotion |
| EP3608311A1 (en) | 2019-06-28 | 2020-02-12 | Bayer AG | Crystalline form a of n-[4-chloro-3-[(1-cyanocyclopropyl)carbamoyl]phenyl]-2-methyl-4-methylsulfonyl-5-(1,1,2,2,2-pentafluoroethyl)pyrazole-3-carboxamide |
| WO2021001331A1 (en) | 2019-07-03 | 2021-01-07 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides and derivatives thereof as microbicides |
| WO2021001273A1 (en) | 2019-07-04 | 2021-01-07 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2021009229A1 (en) | 2019-07-16 | 2021-01-21 | Syngenta Crop Protection Ag | Herbicidal compounds |
| WO2021013721A1 (en) | 2019-07-22 | 2021-01-28 | Bayer Aktiengesellschaft | 5-amino substituted pyrazoles and triazoles as pest control agents |
| WO2021013719A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013720A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021018839A1 (en) | 2019-07-30 | 2021-02-04 | Bayer Animal Health Gmbh | Isoquinoline derivatives and their use for the treatment of parasitic infections |
| EP3771714A1 (en) | 2019-07-30 | 2021-02-03 | Bayer AG | Nitrogen-containing heterocycles as pesticides |
| EP3701796A1 (en) | 2019-08-08 | 2020-09-02 | Bayer AG | Active compound combinations |
| WO2021058659A1 (en) | 2019-09-26 | 2021-04-01 | Bayer Aktiengesellschaft | Rnai-mediated pest control |
| WO2021069567A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069569A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069575A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Animal Health Gmbh | Heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021089673A1 (en) | 2019-11-07 | 2021-05-14 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2021097162A1 (en) | 2019-11-13 | 2021-05-20 | Bayer Cropscience Lp | Beneficial combinations with paenibacillus |
| WO2021099303A1 (en) | 2019-11-18 | 2021-05-27 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021105091A1 (en) | 2019-11-25 | 2021-06-03 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| WO2021122986A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Thienyloxazolones and analogues |
| WO2021136758A1 (en) | 2019-12-30 | 2021-07-08 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates based on polyurea shell material containing polyfunctional aminocarboxylic esters |
| EP3845304A1 (en) | 2019-12-30 | 2021-07-07 | Bayer AG | Capsule suspension concentrates based on polyisocyanates and biodegradable amine based cross-linker |
| WO2021165195A1 (en) | 2020-02-18 | 2021-08-26 | Bayer Aktiengesellschaft | Heteroaryl-triazole compounds as pesticides |
| WO2021170527A1 (en) | 2020-02-24 | 2021-09-02 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved effictiveness in soil and leaf applications |
| EP3868207A1 (en) | 2020-02-24 | 2021-08-25 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved activity in soil and leaf applications |
| EP3708565A1 (en) | 2020-03-04 | 2020-09-16 | Bayer AG | Pyrimidinyloxyphenylamidines and the use thereof as fungicides |
| WO2021204930A1 (en) | 2020-04-09 | 2021-10-14 | Bayer Animal Health Gmbh | Substituted condensed azines as anthelmintic compounds |
| WO2021209364A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209368A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209366A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209365A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209490A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Cyclaminephenylaminoquinolines as fungicides |
| WO2021209363A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021213978A1 (en) | 2020-04-21 | 2021-10-28 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocyclic derivatives as pest control agents |
| WO2021224323A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021224220A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Pyridine (thio)amides as fungicidal compounds |
| WO2021228734A1 (en) | 2020-05-12 | 2021-11-18 | Bayer Aktiengesellschaft | Triazine and pyrimidine (thio)amides as fungicidal compounds |
| WO2021233861A1 (en) | 2020-05-19 | 2021-11-25 | Bayer Aktiengesellschaft | Azabicyclic(thio)amides as fungicidal compounds |
| WO2021245087A1 (en) | 2020-06-04 | 2021-12-09 | Bayer Aktiengesellschaft | Heterocyclyl pyrimidines and triazines as novel fungicides |
| WO2021249995A1 (en) | 2020-06-10 | 2021-12-16 | Bayer Aktiengesellschaft | Azabicyclyl-substituted heterocycles as fungicides |
| WO2021255071A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | 3-(pyridazin-4-yl)-5,6-dihydro-4h-1,2,4-oxadiazine derivatives as fungicides for crop protection |
| WO2021255089A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines and 1,3,4-oxadiazole pyridines as fungicides |
| WO2021255091A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as fungicides |
| WO2021255170A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255169A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| EP3929189A1 (en) | 2020-06-25 | 2021-12-29 | Bayer Animal Health GmbH | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021259997A1 (en) | 2020-06-25 | 2021-12-30 | Bayer Animal Health Gmbh | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021260017A1 (en) | 2020-06-26 | 2021-12-30 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates comprising biodegradable ester groups |
| WO2022002818A1 (en) | 2020-07-02 | 2022-01-06 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2022033991A1 (en) | 2020-08-13 | 2022-02-17 | Bayer Aktiengesellschaft | 5-amino substituted triazoles as pest control agents |
| WO2022053453A1 (en) | 2020-09-09 | 2022-03-17 | Bayer Aktiengesellschaft | Azole carboxamide as pest control agents |
| WO2022058327A1 (en) | 2020-09-15 | 2022-03-24 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| EP3974414A1 (en) | 2020-09-25 | 2022-03-30 | Bayer AG | 5-amino substituted pyrazoles and triazoles as pesticides |
| EP3915371A1 (en) | 2020-11-04 | 2021-12-01 | Bayer AG | Active compound combinations and fungicide compositions comprising those |
| EP3994995A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| WO2022096693A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| EP3994985A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| EP3994989A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, rainfastness and uptake properties |
| EP3994993A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| EP3994992A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| EP3994988A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and rainfastness properties |
| WO2022096687A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and uptake properties |
| WO2022096694A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| WO2022096691A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, uptake and rainfastness properties |
| WO2022096690A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994987A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift and uptake properties |
| WO2022096686A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| WO2022096692A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| WO2022096688A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading and rainfastness properties |
| WO2022096696A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| WO2022096685A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| WO2022096695A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994994A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994986A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| EP3994991A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| EP3994990A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3915971A1 (en) | 2020-12-16 | 2021-12-01 | Bayer Aktiengesellschaft | Phenyl-s(o)n-phenylamidines and the use thereof as fungicides |
| WO2022129190A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | (hetero)aryl substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129196A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | Heterobicycle substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129188A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | 1,2,4-oxadiazol-3-yl pyrimidines as fungicides |
| WO2022152728A1 (en) | 2021-01-15 | 2022-07-21 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP4036083A1 (en) | 2021-02-02 | 2022-08-03 | Bayer Aktiengesellschaft | 5-oxy substituted heterocycles as pesticides |
| WO2022207496A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022207494A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022233777A1 (en) | 2021-05-06 | 2022-11-10 | Bayer Aktiengesellschaft | Alkylamide substituted, annulated imidazoles and use thereof as insecticides |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2022238391A1 (en) | 2021-05-12 | 2022-11-17 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pest control agents |
| WO2023017120A1 (en) | 2021-08-13 | 2023-02-16 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2023025682A1 (en) | 2021-08-25 | 2023-03-02 | Bayer Aktiengesellschaft | Novel pyrazinyl-triazole compounds as pesticides |
| EP4144739A1 (en) | 2021-09-02 | 2023-03-08 | Bayer Aktiengesellschaft | Anellated pyrazoles as parasiticides |
| WO2023036821A1 (en) | 2021-09-09 | 2023-03-16 | Bayer Animal Health Gmbh | New quinoline derivatives |
| EP4148052A1 (en) | 2021-09-09 | 2023-03-15 | Bayer Animal Health GmbH | New quinoline derivatives |
| WO2023205602A1 (en) | 2022-04-18 | 2023-10-26 | Basf Corporation | High-load agricultural formulations and methods of making same |
| WO2023203009A1 (en) | 2022-04-20 | 2023-10-26 | Bayer Aktiengesellschaft | Water dispersible granules with low melting active ingredients prepared by extrusion |
| EP4265110A1 (en) | 2022-04-20 | 2023-10-25 | Bayer AG | Water dispersible granules with low melting active ingredients prepared by extrusion |
| WO2023213626A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Use of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine for controlling unwanted microorganisms |
| WO2023213670A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Crystalline forms of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine |
| WO2023217619A1 (en) | 2022-05-07 | 2023-11-16 | Bayer Aktiengesellschaft | Low drift aqueous liquid formulations for low, medium, and high spray volume application |
| WO2023237444A1 (en) | 2022-06-06 | 2023-12-14 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295683A1 (en) | 2022-06-21 | 2023-12-27 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| WO2024013015A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024013016A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024068473A1 (en) | 2022-09-27 | 2024-04-04 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| EP4295688A1 (en) | 2022-09-28 | 2023-12-27 | Bayer Aktiengesellschaft | Active compound combination |
| WO2024068518A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-heteroaryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068520A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068519A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068517A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| EP4353082A1 (en) | 2022-10-14 | 2024-04-17 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024104643A1 (en) | 2022-11-17 | 2024-05-23 | Bayer Aktiengesellschaft | Use of isotianil for controlling plasmodiophora brassica |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102209469A (en) | 2011-10-05 |
| WO2010052161A3 (en) | 2010-12-23 |
| CA2742552A1 (en) | 2010-05-14 |
| JP5592386B2 (en) | 2014-09-17 |
| GB0820344D0 (en) | 2008-12-17 |
| US8722578B2 (en) | 2014-05-13 |
| JP2012507488A (en) | 2012-03-29 |
| AU2009312909A1 (en) | 2010-05-14 |
| EP2352375A2 (en) | 2011-08-10 |
| BRPI0921411A2 (en) | 2015-08-25 |
| EP2352375B1 (en) | 2015-03-25 |
| US20120004105A1 (en) | 2012-01-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2352375B1 (en) | Herbicidal compositions | |
| AU2008226027B2 (en) | Novel herbicides | |
| EP2134699B1 (en) | 2-aryl-5-heterocyclyl-cyclohexane-1,3-dione compounds and their use as herbicides | |
| EP2313370B1 (en) | 5-heterocyclylalkyl-3-hydroxy-2-phenylcyclopent-2-enones as herbicides | |
| EP2185564B1 (en) | Herbicidally active cyclopentanedione compounds and derivatives thereof | |
| US20050090399A1 (en) | Phenyl substituted heterocyclic compounds useful as herbicides | |
| CA2964831C (en) | 2-substituted phenyl-cyclopent-2-enone or -cyclopentane-1,3-dione compounds and their use as herbicides | |
| EP2350034B1 (en) | Novel herbicides | |
| US20130137573A1 (en) | Herbicidal compositions comprising, and methods of use of, herbicidally active pyrandiones | |
| WO2011073615A2 (en) | Herbicidal composition comprising a pyrandione herbicide and a co-herbicide | |
| AU2015335174B2 (en) | Herbicidal compounds | |
| EP2454224A1 (en) | Novel herbicides | |
| WO2010057880A1 (en) | Novel herbicides | |
| BRPI0921411B1 (en) | METHOD FOR CONTROLLING WEEDS IN CULTURES OF USEFUL PLANTS. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980144197.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09740705 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2573/DELNP/2011 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009740705 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2009312909 Country of ref document: AU Date of ref document: 20091028 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011533709 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2742552 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13128133 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0921411 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110506 |