WO2009145171A1 - 新規なベンゾチオフェンオキシド誘導体またはその塩 - Google Patents
新規なベンゾチオフェンオキシド誘導体またはその塩 Download PDFInfo
- Publication number
- WO2009145171A1 WO2009145171A1 PCT/JP2009/059576 JP2009059576W WO2009145171A1 WO 2009145171 A1 WO2009145171 A1 WO 2009145171A1 JP 2009059576 W JP2009059576 W JP 2009059576W WO 2009145171 A1 WO2009145171 A1 WO 2009145171A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- salt
- benzothiophene
- oxide derivative
- acid
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the present invention relates to a benzothiophene oxide derivative or a salt thereof useful as a prodrug for a therapeutic agent for diseases of central and peripheral nerves.
- Benzothiophene derivatives represented by 1- (3- (2- (1-benzothiophen-5-yl) ethoxy) propyl) -3-azetidinol have neuroprotective action, nerve regeneration promoting action and neurite extension action. It is a compound useful as a therapeutic agent for diseases of central and peripheral nerves (Patent Document 1). However, the benzothiophene oxide derivatives described in the present application are not known at all.
- a compound that has a neuroprotective action, a nerve regeneration promoting action, and a neurite extension action and is useful as a therapeutic agent for diseases of the central and peripheral nerves is strongly desired.
- R 1 and R 2 are the same or different and are a hydrogen atom, a halogen atom, an optionally substituted alkyl, aryl, aralkyl, alkoxy, aryloxy, alkylthio, arylthio, alkenyl, alkenyloxy, amino One or more groups selected from an alkylsulfonyl, arylsulfonyl, carbamoyl or heterocyclic group, an optionally protected amino, hydroxyl or carboxyl group, a nitro group and an oxo group; R 3 is substituted A benzothiophene oxide derivative or a salt thereof, wherein an alkylamino group which may be protected, an amino or hydroxyl group which may be protected; and m and n are the same or different and each represents an integer of 1 to 6.
- R 1 , R 2 , R 3 , m and n have the same meanings as described above, and are converted into benzothiophene derivatives or salts thereof, which are useful as prodrugs of benzothiophene derivatives or salts thereof. I found out. Furthermore, administration of a benzothiophene oxide derivative or a salt thereof does not cause a rapid increase in the blood concentration of the benzothiophene derivative or a salt thereof, and the blood concentration of the benzothiophene derivative or a salt thereof is high over a long period of time. The present invention has been completed.
- the benzothiophene oxide derivative of the general formula [1] or a salt thereof is useful as a prodrug of the benzothiophene derivative or a salt thereof. Furthermore, the benzothiophene oxide derivative of the general formula [1] or a salt thereof is useful as a drug substance for a sustained-release preparation of the benzothiophene derivative or a salt thereof.
- a halogen atom is a fluorine atom, a chlorine atom, a bromine atom or an iodine atom
- an alkyl group is a straight chain such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl, heptyl and octyl
- a lower alkyl group is a linear or branched C 1 such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl and hexyl
- An alkenyl group means a C 2-12 alkenyl group such as vinyl, propenyl, butenyl, pentenyl, hexenyl, heptenyl and octenyl; a lower
- An alkoxy group is a linear or branched C 1-12 alkyloxy group such as methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy, tert-butoxy, pentyloxy, hexyloxy, heptyloxy and octyloxy
- a lower alkoxy group means a linear or branched C 1-6 alkyloxy group such as methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy, tert-butoxy, pentyloxy and hexyloxy;
- An oxy group means a C 2-12 alkenyloxy group such as vinyloxy, propenyloxy, butenyloxy, pentenyloxy, hexenyloxy, heptenyloxy and octenyloxy.
- the alkylthio group is a C 1-12 alkylthio group such as methylthio, ethylthio, propylthio, isopropylthio, butylthio, isobutylthio, tert-butylthio, pentylthio, hexylthio, heptylthio and octylthio;
- the lower alkylthio group is methylthio, ethylthio
- Means a C 1-6 alkylthio group such as propylthio, isopropylthio, butylthio, isobutylthio, tert-butylthio, pentylthio and hexylthio;
- Aryl groups are phenyl, naphthyl, indanyl and indenyl groups; aryloxy groups are phenyloxy, naphthyloxy, indanyloxy and indenyloxy groups; aralkyl groups are benzyl, diphenylmethyl, trityl and An ar C 1-6 alkyl group such as phenethyl; an arylthio group means a phenylthio, naphthylthio, indanylthio and indenylthio group.
- An acyl group is a formyl group, a C 2-12 alkanoyl group such as acetyl, isovaleyl, propionyl and pivaloyl, an aralkylcarbonyl group such as benzylcarbonyl, and an aroyl group such as benzoyl and naphthoyl; Linear or branched C 1-12 such as methoxycarbonyl, ethoxycarbonyl, 1,1-dimethylpropoxycarbonyl, isopropoxycarbonyl, 2-ethylhexyloxycarbonyl, tert-butoxycarbonyl and tert-pentyloxycarbonyl the alkyloxycarbonyl group; the aralkyl oxycarbonyl group, for example, Al C 1-6 Arukiruoki such as a benzyloxycarbonyl or phenethyloxycarbonyl group A carbonyl group; an aryloxycarbonyl group means a group such as phen
- alkylsulfonyl group methylsulfonyl, ethylsulfonyl, propylsulfonyl, isopropylsulfonyl, butylsulfonyl, iso-butylsulfonyl, sec- butylsulfonyl, tert- butylsulfonyl, pentylsulfonyl, hexylsulfonyl, C, such as heptyl sulphonyl and octylsulfonyl 1-
- a 12 alkylsulfonyl group; a lower alkylsulfonyl group for example, a C 1-6 alkylsulfonyl group such as methylsulfonyl, ethylsulfonyl and propylsulfonyl; an arylsulfonyl group, phenylsulfony
- alkylamino group is meant a mono- or di-C 1-6 alkylamino group such as methylamino, ethylamino, propylamino, isopropylamino, butylamino, dimethylamino, diethylamino, diisopropylamino and dibutylamino.
- Heterocyclic groups include pyrrolidinyl, piperidinyl, piperazinyl, homopiperazinyl, homopiperidinyl, morpholyl, thiomorpholyl, tetrahydroquinolinyl, tetrahydroisoquinolyl, quinuclidinyl, imidazolinyl, pyrrolyl, imidazolyl, pyrazolyl, pyridyl, pyridyl, quinodilyl, quinolyl, , Tetrazolyl, thiadiazolyl, pyrrolinyl, pyrazolinyl, pyrazolidinyl, purinyl, furyl, thienyl, benzothienyl, pyranyl, isobenzofuranyl, oxazolyl, isoxazolyl, benzofuranyl, indolyl, benzimidazolyl, benzoxazolyl, benzois
- the oxygen-containing heterocyclic group is, for example, a group such as 2-tetrahydropyranyl and 2-tetrahydrofuranyl;
- the sulfur-containing heterocyclic group is, for example, a group such as tetrahydrothiopyranyl;
- the alkylsilylalkyl group means, for example, a group such as 2- (trimethylsilyl) ethyl.
- the amino protecting group includes all groups that can be used as ordinary amino protecting groups. Greene et al., Protective Groups Organic Synthesis, 3rd edition, pages 494-615, 1999, John Wiley & Sons, INC.). Specifically, for example, acyl group, alkyloxycarbonyl group, aralkyloxycarbonyl group, aryloxycarbonyl group, aralkyl group, alkoxyalkyl group, aralkyloxyalkyl group, arylthio group, alkylsulfonyl group, arylsulfonyl group And substituted silyl groups.
- the hydroxyl protecting group includes all groups that can be used as protecting groups for ordinary hydroxyl groups. Greene et al., Protective Groups Organic Synthesis 3rd edition, pp. 17-245, 1999, John Wiley & Sons, INC.). Specifically, for example, acyl group, alkyloxycarbonyl group, aralkyloxycarbonyl group, heterocyclic oxycarbonyl group, alkyl group, alkenyl group, aralkyl group, oxygen-containing heterocyclic group, sulfur-containing heterocyclic group , An alkoxyalkyl group, an aralkyloxyalkyl group, an alkylsulfonyl group, an arylsulfonyl group, and a substituted silyl group.
- the carboxyl protecting group includes all groups that can be used as protecting groups for ordinary carboxyl groups. Greene et al., Protective Groups Organic Synthesis, 3rd edition, pp. 369-453, 1999, John Wiley & Sons (John Wiley & Sons, INC.).
- alkyl group alkenyl group, aryl group, aralkyl group, acylalkyl group, arylthioalkyl group, arylsulfonylalkyl group, oxygen-containing heterocyclic group, alkylsilylalkyl group, acyloxyalkyl group
- examples thereof include nitrogen-containing heterocyclic alkyl groups, cycloalkyl groups, alkoxyalkyl groups, aralkyloxyalkyl groups, alkylthioalkyl groups, and substituted silyl groups.
- substituent of the heterocyclic group and the alkylamino group in R 3 include a halogen atom, a lower alkyl group, a cycloalkyl group, an aryl group, a lower alkoxy group, an aryloxy group, a lower alkylthio group, an arylthio group, a lower alkenyl group, Selected from lower alkylsulfonyl group, arylsulfonyl group, alkylamino group, amino group which may be protected, hydroxyl group which may be protected, carboxyl group which may be protected, acyl group and hetero
- Examples of the salt of the compound of the general formula [1] include salts that are generally known in basic groups such as amino groups or acidic groups such as hydroxyl or carboxyl groups.
- Examples of salts in basic groups include salts with mineral acids such as hydrochloric acid, hydrobromic acid, nitric acid and sulfuric acid; formic acid, acetic acid, citric acid, oxalic acid, fumaric acid, maleic acid, succinic acid, malic acid, Salts with organic carboxylic acids such as tartaric acid, aspartic acid, trichloroacetic acid and trifluoroacetic acid; and salts with sulfonic acids such as methanesulfonic acid, benzenesulfonic acid, p-toluenesulfonic acid, mesitylenesulfonic acid and naphthalenesulfonic acid. Can be mentioned.
- salts in the acidic group include salts with alkali metals such as sodium and potassium; salts with alkaline earth metals such as calcium and magnesium; ammonium salts; and trimethylamine, triethylamine, tributylamine, pyridine, N, N— Nitrogen-containing organic bases such as dimethylaniline, N-methylpiperidine, N-methylmorpholine, diethylamine, dicyclohexylamine, procaine, dibenzylamine, N-benzyl- ⁇ -phenethylamine, 1-ephenamine and N, N′-dibenzylethylenediamine And a salt thereof.
- preferred salts include pharmacologically acceptable salts.
- the present invention encompasses all those isomers. And includes hydrates, solvates and all crystal forms.
- Preferred examples of the benzothiophene oxide derivative of the general formula [1] or a salt thereof include the following compounds.
- a compound in which R 1 is a hydrogen atom is preferable.
- a compound in which R 2 is a hydrogen atom, a halogen atom or an alkoxy group is preferable, and a compound in which R 2 is a hydrogen atom is more preferable.
- a compound in which R 3 is a hydroxyl group is preferable.
- a compound in which m is 2 is preferable.
- a compound in which n is 2 or 3 is preferred, and a compound in which n is 3 is more preferred.
- the benzothiophene oxide derivative of the general formula [1] can be produced, for example, by the following production method.
- a benzothiophene oxide derivative of the general formula [1] can be produced.
- the benzothiophene derivative of the general formula [2] can be produced, for example, by the method described in Patent Document 1.
- the solvent used in this reaction is not particularly limited as long as it does not adversely influence the reaction.
- oxidizing agent used in this reaction examples include hydrogen peroxide; peracids such as peracetic acid, perbenzoic acid and m-chloroperbenzoic acid; peroxides such as tert-butyl peroxide; and sodium metaperiodate. Is mentioned.
- the amount of the oxidizing agent used may be 1 to 50 times mol, preferably 1 to 10 times mol, of the benzothiophene derivative. This reaction is usually carried out at ⁇ 50 to 100 ° C., preferably ⁇ 20 to 20 ° C., for 30 minutes to 48 hours.
- the benzothiophene oxide derivative of the general formula [1] or a salt thereof is converted into a benzothiophene derivative or a salt thereof in vivo and is useful as a prodrug of the benzothiophene derivative or a salt thereof. Furthermore, administration of the benzothiophene oxide derivative of the general formula [1] or a salt thereof of the present invention does not increase the blood concentration of the benzothiophene derivative or a salt thereof rapidly, and the benzothiophene derivative or a salt thereof The blood concentration is maintained at a high concentration for a long time, and the benzothiophene oxide derivative or a salt thereof is useful as a drug substance for a sustained-release preparation of the benzothiophene derivative or a salt thereof.
- the benzothiophene oxide derivative of the general formula [1] or a salt thereof includes an excipient, a binder, a disintegrant, a disintegration inhibitor, a caking / adhesion preventing agent, a lubricant, an absorption / adsorption carrier, a solvent, a bulking agent, Isotonizing agents, solubilizers, emulsifiers, suspending agents, thickeners, coating agents, absorption accelerators, gelling / coagulation accelerators, light stabilizers, preservatives, moisture-proofing agents, emulsifying / suspending / Addition of various pharmaceuticals such as dispersion stabilizers, anti-coloring agents, deoxygenation / antioxidants, taste-masking / flavoring agents, coloring agents, foaming agents, antifoaming agents, soothing agents, antistatic agents, buffer / pH adjusting agents Products can be formulated into pharmaceutical preparations such as oral preparations (tablets, capsules, powders,
- Oral solid preparations such as tablets, powders and granules include, for example, lactose, sucrose, sodium chloride, glucose, starch, calcium carbonate, kaolin, crystalline cellulose, anhydrous dicalcium phosphate, partially pregelatinized starch, corn starch and alginic acid.
- Excipients simple syrup, glucose solution, starch solution, gelatin solution, polyvinyl alcohol, polyvinyl ether, polyvinyl pyrrolidone, carboxymethylcellulose, shellac, methylcellulose, ethylcellulose, sodium alginate, gum arabic, hydroxypropylmethylcellulose, hydroxypropylcellulose, water and Binders such as ethanol; dried starch, alginic acid, agar powder, starch, crosslinked polyvinylpyrrolidone, crosslinked carboxymethylcellulose sodium Disintegrants such as calcium carboxymethylcellulose and sodium starch glycolate; disintegration inhibitors such as stearyl alcohol, stearic acid, cocoa butter and hydrogenated oil; aluminum silicate, calcium hydrogen phosphate, magnesium oxide, talc, silicic anhydride, etc.
- Anti-caking / adhesion preventive agent carnauba wax, light anhydrous silicic acid, aluminum silicate, magnesium silicate, hardened oil, hardened vegetable oil derivative, sesame oil, white beeswax, titanium oxide, dry aluminum hydroxide gel, stearic acid, calcium stearate , Lubricants such as magnesium stearate, talc, calcium hydrogen phosphate, sodium lauryl sulfate and polyethylene glycol; absorption of quaternary ammonium salts, sodium lauryl sulfate, urea and enzymes Advancing agents; conventional methods using pharmaceutical additives for solid preparations such as absorption and adsorption carriers such as starch, lactose, kaolin, bentonite, silicic anhydride, hydrous silicon dioxide, magnesium aluminate metasilicate and colloidal silicic acid May be prepared.
- absorption and adsorption carriers such as starch, lactose, kaolin, bentonite,
- Injections include, for example, diluents such as water, ethyl alcohol, macrogol, propylene glycol, citric acid, acetic acid, phosphoric acid, lactic acid, sodium lactate, sulfuric acid and sodium hydroxide; sodium citrate, sodium acetate and sodium phosphate PH adjusters and buffers such as; sodium pyrosulfite, ethylenediaminetetraacetic acid, thioglycolic acid and thiolactic acid and other stabilizers; isotonic agents such as sodium chloride, glucose, mannitol or glycerin; sodium carboxymethylcellulose, propylene glycol, Solubilizers such as sodium benzoate, benzyl benzoate, urethane, ethanolamine, glycerin; soothing agents such as calcium gluconate, chlorobutanol, glucose, benzyl alcohol; and liquids such as local anesthetics
- diluents such as water, ethyl alcohol
- the administration method of the above preparation is not particularly limited, but is appropriately determined according to the form of the preparation, the patient's age, sex and other conditions, the degree of symptoms of the patient, and the like.
- the dosage of the active ingredient of the preparation of the present invention is appropriately selected according to the usage, patient age, sex, disease form and other conditions, but usually 0.1 to 1000 mg per day for an adult from a single dose. What is necessary is just to divide and administer in several times.
- Example 1 Preparation of Compound A Oxide Maleate 1- (3- (2- (1-benzothiophen-5-yl) ethoxy) propyl) azetidin-3-ol maleate 7.00 g water 35 mL and ethyl acetate 35 mL mixed with ice-cooled 5 mol / L water A sodium oxide aqueous solution was added to adjust the pH to 11.7. The organic layer was separated, washed successively with water and saturated brine, dried over anhydrous magnesium sulfate, and the solvent was distilled off under reduced pressure. To the obtained residue, 25 mL of methylene chloride was added, and 6.62 mL of trifluoroacetic acid was added dropwise at 6 to 7 ° C.
- Test example 1 The compound A administration solution was prepared by dissolving compound A ⁇ maleate in physiological saline and adjusting the concentration of compound A ⁇ maleate to 14 mg / mL.
- compound A oxide / maleate was dissolved in physiological saline, and the concentration of compound A oxide / maleate was adjusted to 14.54 mg / mL.
- Each administration solution was orally administered to rats (Crl: CD male rats, 8 weeks old) under non-fasting conditions. The dose was 56 mg / kg in the compound A administration group and 58 mg / kg in the compound A oxide administration group.
- the solid phase extraction plate was washed with 2 mL of distilled water and then eluted with 2 mL of acetonitrile.
- the eluate was concentrated to dryness, dissolved in mobile phase A solution, and centrifuged at about 1600 ⁇ g, 4 ° C. for 10 minutes.
- the obtained supernatant was subjected to a liquid chromatograph-tandem mass spectrometer, and the concentration of Compound A in the plasma of each group was determined.
- the isolated brain is homogenized with 25% aqueous ammonia / methanol (1:49) (volume 10 times the wet brain weight), and then 2 mL of the homogenate is centrifuged at approximately 1600 xg, 4 ° C for 10 minutes to separate the supernatant. I took it.
- 2 mL of 25% aqueous ammonia / methanol (1:49) was added and centrifuged at about 1600 ⁇ g, 4 ° C. for 10 minutes, and the supernatant was collected.
- These supernatants were combined, concentrated and dried, and then dissolved in 1 mL of distilled water containing 25 ng / mL of internal standard substance.
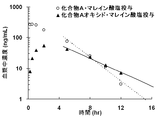
- Compound A oxide prolonged the residence time of Compound A in the body, and the plasma concentration and brain concentration were maintained at a high concentration over a long period of time.
- the benzothiophene oxide derivative of the general formula [1] or a salt thereof is useful as a prodrug of the benzothiophene derivative or a salt thereof. Furthermore, the benzothiophene oxide derivative of the general formula [1] or a salt thereof is useful as a drug substance for a sustained-release preparation of the benzothiophene derivative or a salt thereof.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
さらに、ベンゾチオフェンオキシド誘導体またはその塩を投与することによって、ベンゾチオフェン誘導体またはその塩の血中濃度が急激に上昇せず、かつ、ベンゾチオフェン誘導体またはその塩の血中濃度が長期間にわたって高濃度で維持されることを見出し、本発明を完成した。
本明細書において、特に断らない限り、各用語は、次の意味を有する。
化合物Aとは、「1-(3-(2-(1-ベンゾチオフェン-5-イル)エトキシ)プロピル)アゼチジン-3-オール」を意味する。
化合物Aオキシドとは、「1-(3-(2-(1-ベンゾチオフェン-5-イル)エトキシ)プロピル)アゼチジン-3-オール S-オキシド」を意味する。
塩基性基における塩としては、たとえば、塩酸、臭化水素酸、硝酸および硫酸などの鉱酸との塩;ギ酸、酢酸、クエン酸、シュウ酸、フマル酸、マレイン酸、コハク酸、リンゴ酸、酒石酸、アスパラギン酸、トリクロロ酢酸およびトリフルオロ酢酸などの有機カルボン酸との塩;ならびにメタンスルホン酸、ベンゼンスルホン酸、p-トルエンスルホン酸、メシチレンスルホン酸およびナフタレンスルホン酸などのスルホン酸との塩が挙げられる。
上記した塩の中で、好ましい塩としては、薬理学的に許容される塩が挙げられる。
R1が、水素原子である化合物が好ましい。
R2が、水素原子、ハロゲン原子またはアルコキシ基である化合物が好ましく、水素原子である化合物がさらに好ましい。
R3が、ヒドロキシル基である化合物が好ましい。
mが、2である化合物が好ましい。
nが、2または3である化合物が好ましく、3である化合物がさらに好ましい。
さらに、R1およびR2が、水素原子;R3が、ヒドロキシル基;mが、2;nが、3である化合物が最も好ましい。
一般式[2]のベンゾチオフェン誘導体は、たとえば、特許文献1に記載の方法で製造することができる。
この反応で使用される溶媒としては、反応に悪影響を及ぼさないものであれば特に限定されないが、たとえば、水;塩化メチレン、クロロホルムおよびジクロロエタンなどのハロゲン化炭化水素類;ヘキサン、シクロヘキサンなどの脂肪族炭化水素類;ならびにピリジンなどが挙げられ、これらは混合して使用してもよい。
この反応に用いられる酸化剤としては、たとえば、過酸化水素;過酢酸、過安息香酸およびm-クロロ過安息香酸などの過酸類;tert-ブチルペルオキシドなどのペルオキシド類;ならびにメタ過ヨウ素酸ナトリウムなどが挙げられる。酸化剤の使用量は、ベンゾチオフェン誘導体に対して1~50倍モル、好ましくは、1~10倍モルであればよい。
この反応は、通常、-50~100℃、好ましくは、-20~20℃で、30分間~48時間実施すればよい。
上記の各種製剤は、通常の方法により製剤化される。
錠剤は、必要に応じ、通常の剤皮を施した錠剤、たとえば、糖衣錠、ゼラチン被包錠、胃溶性被覆錠、腸溶性被覆錠および水溶性フィルムコーティング錠とすることができる。
カプセル剤は、上記で例示した各種の医薬品と混合し、硬質ゼラチンカプセルおよび軟質カプセルなどに充填して調製される。
これらの経口用固形製剤は、溶剤、増量剤、等張化剤、溶解補助剤、乳化剤、懸濁化剤、増粘剤などの上記した各種の液体製剤化用添加物を用い、常法により調製し、水性または油性の懸濁液、溶液、シロップおよびエリキシル剤とすることもできる。
本発明製剤の有効成分の投与量は、用法、患者の年齢、性別、疾患の形態およびその他の条件などに応じて適宜選択されるが、通常成人に対して1日0.1~1000mgを1回から数回に分割して投与すればよい。
実施例において略号は、以下の意味を有する。
DMSO-d6:重ジメチルスルホキシド
1-(3-(2-(1-ベンゾチオフェン-5-イル)エトキシ)プロピル)アゼチジン-3-オール・マレイン酸塩7.00gの水35mLおよび酢酸エチル35mL混液に氷冷下、5mol/L水酸化ナトリウム水溶液を加え、pH11.7に調整した。有機層を分取し、水および飽和食塩水で順次洗浄後、無水硫酸マグネシウムで乾燥し、減圧下で溶媒を留去した。得られた残留物に塩化メチレン25mLを加え、6~7℃でトリフルオロ酢酸6.62mLを滴下した。ついで、4~7℃でm-クロロ過安息香酸3.65gを分割添加し、同温度で2時間攪拌した。不溶物を濾去後、残渣を水で洗浄した。濾液および洗液を合わせ、水層を分取した。有機層を水で抽出し、合わせた水層を酢酸エチルで洗浄後、酢酸エチルを加え、炭酸カリウムでpH10.5に調整した。水層を分取し、クロロホルムで抽出後、無水硫酸マグネシウムで乾燥し、減圧下で16.1gまで濃縮した。
得られた溶液に氷冷下、マレイン酸1.20gのメタノール4.5mL溶液を滴下し、ついで酢酸エチル13.5mLを滴下し、同温度で1時間攪拌した。固形物を濾取し、1-(3-(2-(1-ベンゾチオフェン-5-イル)エトキシ)プロピル)アゼチジン-3-オール S-オキシド・マレイン酸塩3.36gを得た。
1H-NMR(DMSO-d6)δ値:1.74-1.63(m,2H),2.91(t,2H,J=6.5Hz),3.11(t,2H,J=7.6Hz),3.42(t,2H,J=5.9Hz),3.63(t,2H,J=6.5Hz),3.82-3.73(m,2H),4.28-4.15(m,2H),4.51-4.41(m,1H),6.03(s,2H),6.14-6.09(m,1H),7.43-7.38(m,1H),7.43(d,1H,J=6.1Hz),7.46(d,1H,J=6.1Hz),7.57-7.53(m,1H),7.90(d,1H,J=7.6Hz)
化合物A投与液は、化合物A・マレイン酸塩を生理食塩液に溶解し、化合物A・マレイン酸塩濃度を14mg/mLに調整した。化合物Aオキシド投与液は、化合物Aオキシド・マレイン酸塩を生理食塩液に溶解し、化合物Aオキシド・マレイン酸塩濃度を14.54mg/mLに調整した。
各々の投与液を非絶食下、ラット(Crl:CD系雄性ラット、8週齢)に経口投与した。投与量は、化合物A投与群で56mg/kg、化合物Aオキシド投与群で58mg/kgとした。
投与から0.25、0.5、1、2、5、8、10および12時間後、各群についてエーテル軽麻酔下で腹部大動脈より全血をヘパリン加採血した後、放血致死させ、脳を摘出した。
採取した血液を約1600×g、4℃、10分間遠心分離して血漿を得た。得られた血漿100μLに内標準物質25ng/mLを含む蒸留水900μLおよびリン酸25μLを加えて攪拌した後、メタノールおよび蒸留水でコンディショニングした固相抽出プレート(Oasis HLB 10mgまたは30mg、Waters社製)に添加した。蒸留水2mLで固相抽出プレートを洗浄した後、アセトニトリル2mLで溶出した。溶出液を濃縮乾固した後、移動相A液に溶解し、約1600×g、4℃、10分間遠心分離した。得られた上清を液体クロマトグラフ-タンデム質量分析計に付し、各群の血漿中の化合物A濃度を求めた。
摘出した脳を25%アンモニア水/メタノール(1:49)(脳湿重量の10倍容量)でホモジナイズした後、ホモジネート2mLを約1600×g、4℃、10分間遠心分離して上清を分取した。沈渣に25%アンモニア水/メタノール(1:49)2mLを加え、約1600×g、4℃、10分間遠心分離して上清を分取した。これら上清を合わせて濃縮乾固した後、内標準物質25ng/mLを含む蒸留水1mLに溶解した。得られた溶液にリン酸25μLを加えて攪拌した後、上記の血漿と同様の固相抽出操作を行った。得られた溶出液を濃縮乾固した後、移動相A液または50%メタノールに溶解し、約1600×g、4℃、10分間遠心分離操作した。得られた上清を液体クロマトグラフ-タンデム質量分析計に付し、各群の脳内の化合物A濃度を求めた。
結果を図1および図2に示す。
化合物A・マレイン酸塩および化合物Aオキシド・マレイン酸塩投与後の化合物Aの消失半減期(T1/2)および平均滞留時間(MRT)を表1に示す。
化合物A濃度の測定には、以下の機器および条件を使用した。
液体クロマトグラフ-タンデム質量分析計
液体クロマトグラフ装置:ACQUITY UPLCシステム(Waters)
質量分析計:API5000(Applied Biosystems/MDS SCIEX)
カラム:ACQUITY UPLC BEH C18 1.7μm、内径2.1mm×長さ100mm(Waters)
カラム温度:40℃
移動相
移動相A液:アセトニトリル/0.2mol/Lギ酸緩衝液(pH5.5)/蒸留水(10:5:85(v/v/v))
移動相B液:アセトニトリル/0.2mol/Lギ酸緩衝液(pH5.5)/蒸留水(90:5:5(v/v/v))
ステップグラジエント条件
0-3分:移動相A液95%、移動相B液5%
3-6分:移動相A液90%、移動相B液10%
6-9分:移動相A液85%、移動相B液15%
9-10分:移動相A液80%、移動相B液20%
10-11分:移動相A液25%、移動相B液75%
流速:0.4mL/分
内標準物質:2-[2-(ジエチルアミノ)エトキシ]-1-(1-ナフチル)エタノール塩酸塩
Claims (6)
- 一般式
- R1が、水素原子;R2が、水素原子、ハロゲン原子またはアルコキシ基である請求項1記載のベンゾチオフェンオキシド誘導体またはその塩。
- mが、2;nが、2または3である請求項1または2記載のベンゾチオフェンオキシド誘導体またはその塩。
- R2が、水素原子;R3が、ヒドロキシル基;nが、3である請求項1~3記載のベンゾチオフェンオキシド誘導体またはその塩。
- 1-(3-(2-(1-ベンゾチオフェン-5-イル)エトキシ)プロピル)アゼチジン-3-オール S-オキシドまたはその塩。
- 請求項1~5記載のベンゾチオフェンオキシド誘導体またはその塩を含有する医薬組成物。
Priority Applications (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0912266A BRPI0912266B8 (pt) | 2008-05-28 | 2009-05-26 | composto derivado de óxido de benzotiofeno, e, composição farmacêutica |
| US12/994,837 US8026232B2 (en) | 2008-05-28 | 2009-05-26 | Benzothiophene oxide derivative and salt thereof |
| JP2010514481A JP5552424B2 (ja) | 2008-05-28 | 2009-05-26 | 新規なベンゾチオフェンオキシド誘導体またはその塩 |
| EP09754679.0A EP2287160B1 (en) | 2008-05-28 | 2009-05-26 | Benzothiophene oxide derivative and salt thereof |
| ES09754679.0T ES2465005T3 (es) | 2008-05-28 | 2009-05-26 | Derivado de óxido de benzotiofeno y sus sales |
| NZ589448A NZ589448A (en) | 2008-05-28 | 2009-05-26 | Novel benzothiophene oxide derivative and salt thereof |
| MX2010012787A MX2010012787A (es) | 2008-05-28 | 2009-05-26 | Nuevo derivado de oxido de benzotiofeno y sal del mismo. |
| PL09754679T PL2287160T3 (pl) | 2008-05-28 | 2009-05-26 | Pochodna tlenku benzotiofenu i jej sól |
| CN200980119501.9A CN102046620B (zh) | 2008-05-28 | 2009-05-26 | 新的苯并噻吩氧化物衍生物及其盐 |
| DK09754679.0T DK2287160T3 (da) | 2008-05-28 | 2009-05-26 | Benzothiophenoxidderivat og salt deraf |
| AU2009252368A AU2009252368B2 (en) | 2008-05-28 | 2009-05-26 | Novel benzothiophene oxide derivative and salt thereof |
| CA2725307A CA2725307C (en) | 2008-05-28 | 2009-05-26 | Novel benzothiophene oxide derivative and salt thereof |
| SI200930987T SI2287160T1 (sl) | 2008-05-28 | 2009-05-26 | Derivat benzotiofenovega oksida in sol le-tega |
| IL209443A IL209443A (en) | 2008-05-28 | 2010-11-18 | Its zenothiofan oxide and its salt and a pharmaceutical preparation containing it |
| ZA2010/08354A ZA201008354B (en) | 2008-05-28 | 2010-11-22 | Novel benzothiophene oxide derivative and salt thereof |
| HRP20140495AT HRP20140495T1 (hr) | 2008-05-28 | 2014-05-29 | Derivat benzotiofen oksida i njegove soli |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008139007 | 2008-05-28 | ||
| JP2008-139007 | 2008-05-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009145171A1 true WO2009145171A1 (ja) | 2009-12-03 |
Family
ID=41377044
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/059576 WO2009145171A1 (ja) | 2008-05-28 | 2009-05-26 | 新規なベンゾチオフェンオキシド誘導体またはその塩 |
Country Status (20)
| Country | Link |
|---|---|
| US (1) | US8026232B2 (ja) |
| EP (1) | EP2287160B1 (ja) |
| JP (1) | JP5552424B2 (ja) |
| KR (1) | KR101615114B1 (ja) |
| CN (1) | CN102046620B (ja) |
| AU (1) | AU2009252368B2 (ja) |
| BR (1) | BRPI0912266B8 (ja) |
| CA (1) | CA2725307C (ja) |
| CY (1) | CY1115355T1 (ja) |
| DK (1) | DK2287160T3 (ja) |
| ES (1) | ES2465005T3 (ja) |
| HR (1) | HRP20140495T1 (ja) |
| IL (1) | IL209443A (ja) |
| MX (1) | MX2010012787A (ja) |
| NZ (1) | NZ589448A (ja) |
| PL (1) | PL2287160T3 (ja) |
| PT (1) | PT2287160E (ja) |
| SI (1) | SI2287160T1 (ja) |
| WO (1) | WO2009145171A1 (ja) |
| ZA (1) | ZA201008354B (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2002344107B2 (en) | 2001-10-19 | 2007-11-01 | Toyama Chemical Co., Ltd. | Alkyl ether derivatives or salts thereof |
| TR201907882T4 (tr) * | 2012-02-22 | 2019-06-21 | Fujifilm Toyama Chemical Co Ltd | 1-(3-(2-(1-Benzotiofen-5-il)etoksi)propil)azetidin-3-olü veya onun tuzunu içeren katı farmasötik bileşim |
| CN110139641B (zh) * | 2016-12-28 | 2022-09-09 | 富士胶片富山化学株式会社 | 外用组合物 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003035647A1 (fr) | 2001-10-19 | 2003-05-01 | Toyama Chemical Co., Ltd. | Derives ou sels d'ether d'alkyle |
| WO2007125913A1 (ja) * | 2006-04-26 | 2007-11-08 | Toyama Chemical Co., Ltd. | アルキルエーテル誘導体またはその塩を含有する神経細胞新生誘導剤および精神障害治療剤 |
| WO2008016107A1 (fr) * | 2006-08-04 | 2008-02-07 | Toyama Chemical Co., Ltd. | Amplificateur de l'activité de la protéine kinase c contenant un dérivé d'éther d'alkyle ou un sel de celui-ci |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2452513A1 (en) * | 2001-07-12 | 2003-01-23 | Pharmacia & Upjohn Company | Amide-containing compound having improved solubility and method of improving the solubility of an amide-containing compound |
| IL165589A (en) * | 2002-06-14 | 2012-04-30 | Toyama Chemical Co Ltd | Pharmaceutical preparation to improve brain function and use |
-
2009
- 2009-05-26 US US12/994,837 patent/US8026232B2/en active Active
- 2009-05-26 KR KR1020107026524A patent/KR101615114B1/ko active IP Right Grant
- 2009-05-26 WO PCT/JP2009/059576 patent/WO2009145171A1/ja active Application Filing
- 2009-05-26 AU AU2009252368A patent/AU2009252368B2/en active Active
- 2009-05-26 MX MX2010012787A patent/MX2010012787A/es active IP Right Grant
- 2009-05-26 ES ES09754679.0T patent/ES2465005T3/es active Active
- 2009-05-26 PL PL09754679T patent/PL2287160T3/pl unknown
- 2009-05-26 SI SI200930987T patent/SI2287160T1/sl unknown
- 2009-05-26 DK DK09754679.0T patent/DK2287160T3/da active
- 2009-05-26 PT PT97546790T patent/PT2287160E/pt unknown
- 2009-05-26 CN CN200980119501.9A patent/CN102046620B/zh active Active
- 2009-05-26 JP JP2010514481A patent/JP5552424B2/ja active Active
- 2009-05-26 NZ NZ589448A patent/NZ589448A/en unknown
- 2009-05-26 BR BRPI0912266A patent/BRPI0912266B8/pt active IP Right Grant
- 2009-05-26 CA CA2725307A patent/CA2725307C/en active Active
- 2009-05-26 EP EP09754679.0A patent/EP2287160B1/en active Active
-
2010
- 2010-11-18 IL IL209443A patent/IL209443A/en active IP Right Grant
- 2010-11-22 ZA ZA2010/08354A patent/ZA201008354B/en unknown
-
2014
- 2014-05-29 HR HRP20140495AT patent/HRP20140495T1/hr unknown
- 2014-07-01 CY CY20141100486T patent/CY1115355T1/el unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003035647A1 (fr) | 2001-10-19 | 2003-05-01 | Toyama Chemical Co., Ltd. | Derives ou sels d'ether d'alkyle |
| WO2007125913A1 (ja) * | 2006-04-26 | 2007-11-08 | Toyama Chemical Co., Ltd. | アルキルエーテル誘導体またはその塩を含有する神経細胞新生誘導剤および精神障害治療剤 |
| WO2008016107A1 (fr) * | 2006-08-04 | 2008-02-07 | Toyama Chemical Co., Ltd. | Amplificateur de l'activité de la protéine kinase c contenant un dérivé d'éther d'alkyle ou un sel de celui-ci |
Non-Patent Citations (5)
| Title |
|---|
| See also references of EP2287160A4 |
| W GREENE ET AL.: "Protective Groups in Organic Synthesis", 1999, JOHN WILEY & SONS, INC., pages: 17 - 245 |
| W GREENE ET AL.: "Protective Groups in Organic Synthesis", 1999, JOHN WILEY & SONS, INC., pages: 369 - 453 |
| W GREENE ET AL.: "Protective Groups in Organic Synthesis", 1999, JOHN WILEY & SONS, INC., pages: 494 - 615 |
| WERMUTH C.G.: "Translated under the supervision of Hiroshi NAGASE", SAISHIN SOYAKU KAGAKU, vol. LAST, 25 September 1999 (1999-09-25), pages 307 - 310, XP008147640 * |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2009252368A1 (en) | 2009-12-03 |
| PT2287160E (pt) | 2014-06-24 |
| CA2725307C (en) | 2015-12-22 |
| EP2287160B1 (en) | 2014-05-07 |
| DK2287160T3 (da) | 2014-06-10 |
| PL2287160T3 (pl) | 2014-09-30 |
| EP2287160A4 (en) | 2012-01-04 |
| CY1115355T1 (el) | 2017-01-04 |
| ZA201008354B (en) | 2012-02-29 |
| CA2725307A1 (en) | 2009-12-03 |
| EP2287160A1 (en) | 2011-02-23 |
| US8026232B2 (en) | 2011-09-27 |
| AU2009252368B2 (en) | 2013-10-03 |
| CN102046620B (zh) | 2014-04-09 |
| JP5552424B2 (ja) | 2014-07-16 |
| KR20110010741A (ko) | 2011-02-07 |
| SI2287160T1 (sl) | 2014-09-30 |
| IL209443A (en) | 2014-02-27 |
| BRPI0912266B8 (pt) | 2021-05-25 |
| IL209443A0 (en) | 2011-01-31 |
| BRPI0912266A2 (pt) | 2015-10-13 |
| ES2465005T3 (es) | 2014-06-04 |
| HRP20140495T1 (hr) | 2014-07-04 |
| US20110077413A1 (en) | 2011-03-31 |
| KR101615114B1 (ko) | 2016-04-25 |
| NZ589448A (en) | 2011-09-30 |
| CN102046620A (zh) | 2011-05-04 |
| MX2010012787A (es) | 2011-02-24 |
| JPWO2009145171A1 (ja) | 2011-10-13 |
| BRPI0912266B1 (pt) | 2019-07-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5695293B2 (ja) | アルキルエーテル誘導体またはその塩を含有する神経細胞新生誘導剤および精神障害治療剤 | |
| KR100659007B1 (ko) | 아미드 화합물 및 그 의약으로서의 용도 | |
| JP5255441B2 (ja) | アルキルエーテル誘導体またはその塩を含有するプロテインキナーゼc活性促進剤 | |
| JP4398247B2 (ja) | アルキルエーテル誘導体またはその塩 | |
| US8119625B2 (en) | Neurogenesis inducer or neuropathy therapeutic agent comprising alkyl ether derivative or salt thereof | |
| CN109153672B (zh) | Trpv4拮抗剂 | |
| US11548878B2 (en) | Emopamil binding protein binding agent and use thereof | |
| WO2016199878A1 (ja) | シグマ受容体結合剤 | |
| WO2004091605A1 (ja) | アルキルエーテル誘導体またはその塩を含有する網膜神経疾患の予防・治療剤 | |
| JP5552424B2 (ja) | 新規なベンゾチオフェンオキシド誘導体またはその塩 | |
| JP2003246732A (ja) | 1,2−エタンジオール誘導体またはその塩を含有する神経断裂治療剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980119501.9 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09754679 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010514481 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009252368 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2725307 Country of ref document: CA Ref document number: 8245/DELNP/2010 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 589448 Country of ref document: NZ Ref document number: MX/A/2010/012787 Country of ref document: MX Ref document number: 2009754679 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20107026524 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12994837 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2009252368 Country of ref document: AU Date of ref document: 20090526 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: PI0912266 Country of ref document: BR Kind code of ref document: A2 Effective date: 20101123 |