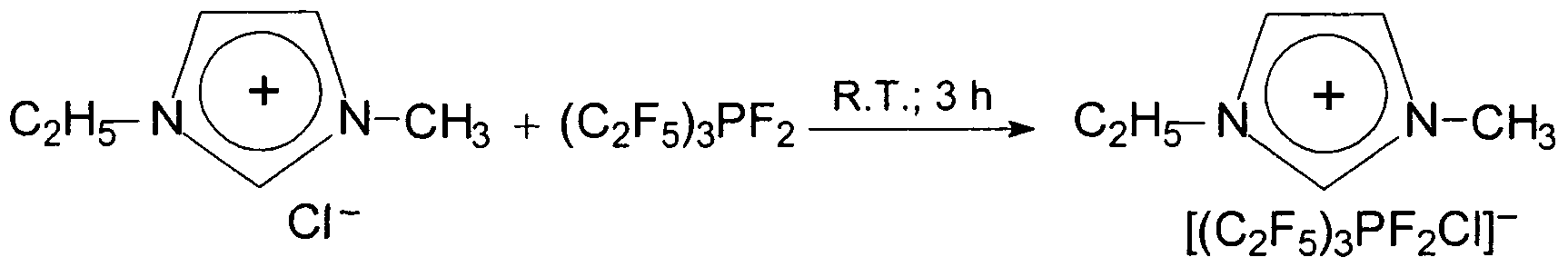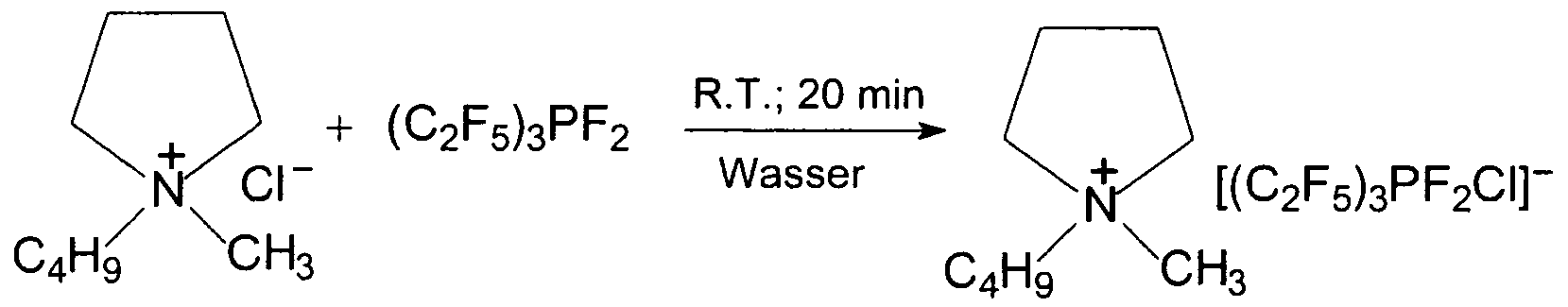WO2008092489A1 - Verbindungen enthaltend organofluorochlorophosphatanionen - Google Patents
Verbindungen enthaltend organofluorochlorophosphatanionen Download PDFInfo
- Publication number
- WO2008092489A1 WO2008092489A1 PCT/EP2007/010646 EP2007010646W WO2008092489A1 WO 2008092489 A1 WO2008092489 A1 WO 2008092489A1 EP 2007010646 W EP2007010646 W EP 2007010646W WO 2008092489 A1 WO2008092489 A1 WO 2008092489A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- atoms
- partially
- straight
- chain
- compound according
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/54—Quaternary phosphonium compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/02—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms containing only hydrogen and carbon atoms in addition to the ring hetero elements
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D521/00—Heterocyclic compounds containing unspecified hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/535—Organo-phosphoranes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K5/00—Heat-transfer, heat-exchange or heat-storage materials, e.g. refrigerants; Materials for the production of heat or cold by chemical reactions other than by combustion
- C09K5/08—Materials not undergoing a change of physical state when used
- C09K5/10—Liquid materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M6/00—Primary cells; Manufacture thereof
- H01M6/14—Cells with non-aqueous electrolyte
- H01M6/145—Cells with non-aqueous electrolyte containing ammonia
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/22—Fuel cells in which the fuel is based on materials comprising carbon or oxygen or hydrogen and other elements; Fuel cells in which the fuel is based on materials comprising only elements other than carbon, oxygen or hydrogen
- H01M8/222—Fuel cells in which the fuel is based on compounds containing nitrogen, e.g. hydrazine, ammonia
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/002—Inorganic electrolyte
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the present invention relates to compounds with Organofluorochlorophosphatanionen, their preparation and their use, in particular as ionic liquids.
- Ionic liquids or liquid salts are ionic species consisting of an organic cation and a generally inorganic anion. They contain no neutral molecules and usually have melting points less than 373 K.
- ionic liquids eg melting point, thermal and electrochemical stability, viscosity
- properties of ionic liquids are strongly influenced by the nature of the anion.
- the polarity and hydrophilicity or lipophilicity can be varied by the appropriate choice of the cation / anion pair.
- EP 0 929 558, WO 02/085919 and EP 1 162 204 disclose salts with perfluoroalkyl fluorophosphate anions (FAP anions for short). These salts are characterized by a high electrochemical and thermal stability and at the same time have a low viscosity. Salts based on FAP anions are largely inert and have a larger
- the object of the present invention is accordingly the provision of novel compounds which are suitable, for example, as ionic liquids.
- a first subject of the present invention are thus compounds containing Organofluorochlorophosphatanionen, preferably salts with Organofluorochlorophosphatanionen.
- organo means any organic radical, for example aliphatic or aromatic organo radicals, which in turn may themselves be substituted, for example with further organo radicals or radicals containing heteroatoms Invention no limitation.
- the compounds according to the present invention are those of the formula (I)
- y stands for the number of Cl anions present in the anion.
- the number z stands for the degree of charge of the cation and thus for the number of anions present in the compounds according to the invention. Overall, the electroneutrality of the compounds should be ensured.
- Very particularly preferred in the context of the present invention are the compounds selected from the group comprising [Kt] 2+ z [(C 2 F 5 ) 3 PCIF 2 ] " , [Kt] 2+ Zt (C 3 F 7 ) S PCIF 2 ] " or [Kt] 2+ Zt (C 4 Fg) 3 PCIF 2 ] -.
- the [Kt] 2+ may be an inorganic or organic cation.
- Ammonium cations can be exemplified by the formula (1) [NR 4 J + (D, where R are each independently H, OR ', NR' 2 , with the proviso that at most one substituent R in formula (1)
- NR ' 2 is straight-chain or branched alkyl having 1-20 C atoms, straight-chain or branched alkenyl having 2-20 C atoms and one or more double bonds, straight-chain or branched alkynyl having 2-20 C atoms and a or more triple bonds, saturated, partially or fully unsaturated cycloalkyl having 3-7 C atoms, which may be substituted by alkyl groups having 1-6 C atoms, wherein one or more R partially or completely halogens, especially -F and / or -Cl, or partially with -OH, -OR 1 , - NR ' 2 , -CN 1 -C (O) OH, -C (O) NR' 2 , -SO 2 NR ' 2 , -C (O ) X, -SO 2 OH, -SO 2 X, -SR ', - S (O) R', -SO 2 R ', - NO 2
- Phosphonium cations can be exemplified by the formula (2)
- R 2 are each, independently of one another, H, OR 'or NR' 2 straight-chain or branched alkyl having 1-20 C atoms, straight-chain or branched alkenyl having 2-20 C atoms and one or more double bonds, straight-chain or branched alkynyl having 2-20 C atoms and one or more triple bonds, saturated, partially or completely unsaturated cycloalkyl having 3-7 C
- Atoms which may be substituted by alkyl groups having 1-6 carbon atoms, where one or more R 2 partially or completely with halogens, in particular -F and / or -Cl 1 or partially with -OH, -OR ', - NR ' 2 , -CN 1 -C (O) OH 1 -C (O) NR' 2l -SO 2 NR ' 2 , -C (O) X, -SO 2 OH, -SO 2 X, -SR', -S (O) R ', -SO 2 R', -NO 2 , and wherein one or two non-adjacent and non- ⁇ -carbon atoms of R 2 , by atoms and / or atomic groups selected from the group O-, -S-, -S (O) -, -SO 2 -, -SO 2 O-, -C (O) -, -C (O) O-, -N + R '
- cations of the formulas (1) and (2) in which all four or three substituents R and R 2 are completely substituted by halogens are excluded, for example the tris (trifluoromethyl) methylammonium cation, the tetra (trifluoromethyl) ammonium cation or the tetra (nonafluorobutyl) ammonium cation.
- Uronium cations can be represented, for example, by the formula (3)
- R 3 to R 7 are each independently H, where H is excluded for R 5 , straight-chain or branched alkyl having 1 to 20 C atoms, straight-chain or branched alkenyl having 2-20 C atoms and one or more double bonds, straight-chain or branched alkynyl having 2-20 C atoms and one or more triple bonds, saturated, partial or fully unsaturated cycloalkyl having 3-7 C atoms, which may be substituted by alkyl groups having 1-6 C atoms, wherein one or more of the substituents R 3 to R 7 partially or completely with halogens, in particular -F and / or -Cl, or partially with -OH, -OR ', -NR' 2 , -CN, -C (O) OH, -C (O) NR ' 2 , -SO 2 NR' 2 , -C (O) X , -SO 2 OH, -SO 2 X, -SR ',
- Guanidinium cations can be represented by the formula (5)
- R 8 to R 13 are each independently
- HetN] 2+ (6) where HetN z + is a heterocyclic cation selected from the group
- R 1 'to R 4 ' are each independently
- substituents R 1 , R 2 , R 3 and / or R 4 may together also form a ring system, one or more substituents R 1 'to R 4 ' being partially or completely halogenated, in particular -F and / or Cl, or -OH, -OR ', -NR' 2 , -CN, -C (O) OH, -C (O) NR ' 2 , -SO 2 NR' 2 , -C (O) X, -SO 2 OH, -SO 2 X, -SR 1 , -S (O) R 1 , -SO 2 R ', -NO 2 , may be substituted, but not at the same time R 1 and R 4 may be completely substituted with halogens, and wherein one or two non-adjacent and non-bonded to the heteroatom carbon atoms of the substituents R 1 'to R 4 ', by atoms and / or atomic groups selected from the
- suitable compounds according to the invention are preferably: C r to C 2O- , in particular C 1 - to C- ⁇ -alkyl groups, and saturated or unsaturated, ie also aromatic, C 3 to C 7 -cycloalkyl groups, which may be substituted with C r to C ⁇ alkyl groups, especially phenyl.
- the substituents R and R 2 in the compounds of formula (1) or (2) may be the same or different. Preferably, the substituents R and R 2 are different.
- the substituents R and R 2 are particularly preferably methyl, ethyl, isopropyl, propyl, butyl, sec-butyl, pentyl, hexyl, octyl, decyl or tetradecyl.
- the substituents R 8 to R 10 and R 13 may have a previously given or particularly preferred meaning.
- the carbocycles or heterocycles of the above guanidinium cations can still by C r to C ⁇ -alkyl, Cr to C ⁇ -alkenyl, -CN, -NO 2 , F, Cl 1 Br, I, -OH, -CrC 6 alkoxy , -NR ' 2 , -SR', -S (O) R ', -SO 2 R', -COOH, -SO 2 NR ' 2) -SO 2 X' or -SO 3 H be substituted, wherein X and R 1 have the meaning given above, be substituted or unsubstituted phenyl or unsubstituted or substituted heterocycle substituted.
- substituents R 3 , R 5 and R 6 may have a previously stated or particularly preferred meaning.
- carbocycles or heterocycles of the abovementioned cations can also be replaced by Cr to C ⁇ -alkyl, C 1 to C 6 -alkenyl, -CN, -NO 2 , F, Cl, Br, I, -OH, -C 1 -C 6 - AIkOXy, -NR ' 2 , -SR', -S (O) R ',
- the substituents R 3 to R 13 are each, independently of one another, preferably a straight-chain or branched alkyl group having 1 to 16 C atoms.
- the substituents R 3 and R 4 , R 6 and R 7 , R 8 and R 9 , R 10 and R 11 and R 12 and R 13 in compounds of the formulas (3) to (5) may be the same or different.
- R 3 to R 13 are each independently methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, sec-butyl, phenyl, hexyl or cyclohexyl, most preferably methyl, ethyl, n-propyl , Isopropyl, n-butyl or hexyl.
- substituents R 1 to R 4 of compounds of the formula (6) are according to the invention
- the substituents R 1 and R 4 are each, independently of one another, particularly preferably methyl, ethyl, isopropyl, propyl, butyl, sec-butyl, tert-butyl, pentyl, hexyl, octyl, decyl, cyclohexyl, phenyl or benzyl. They are very particularly preferably methyl, ethyl, n-butyl or hexyl. In pyrrolidinium, piperidinium or indolinium compounds, the two substituents R 1 and R 4 are preferably different.
- R 2 or R 3 is in each case independently of one another in particular H, methyl, ethyl, isopropyl, propyl, butyl, sec-butyl, tert-butyl, cyclohexyl, phenyl or benzyl.
- R 2 is particularly preferably H, methyl, ethyl, isopropyl, propyl, butyl or sec-butyl.
- R 2 ' and R 3 are H.
- the C 1 -C 2 -alkyl group is, for example, methyl, ethyl, isopropyl, propyl, butyl, sec-butyl or tert-butyl, and also pentyl, 1-, 2- or 3-methylbutyl, 1, 1, 1, 2 or 2,2-dimethylpropyl, 1-ethylpropyl, hexyl, heptyl, octyl, nonyl, decyl, undecyl or dodecyl.
- a straight-chain or branched alkenyl having 2 to 20 C atoms, wherein several double bonds may also be present, is, for example, allyl, 2- or 3-butenyl, isobutenyl, sec-butenyl, furthermore 4-pentenyl, isopentenyl, hexenyl, heptenyl, octenyl, -CgHi 7, -C 10 H 19 to C 2O H 39; preferably allyl, 2- or 3-butenyl, isobutenyl, sec-butenyl, furthermore preferred is 4-pentenyl, iso-pentenyl or hexenyl.
- a straight-chain or branched alkynyl having 2 to 20 C atoms, wherein a plurality of triple bonds may also be present, is, for example, ethynyl, 1- or 2-propynyl, 2- or 3-butynyl, furthermore 4-pentynyl, 3-pentynyl, hexynyl, Heptynyl, octynyl, -C 9 H 15 , -Ci 0 H 17 to -C 2 oH 37 , preferably ethynyl, 1- or 2-propynyl, 2- or 3-butynyl, 4-pentynyl, 3-pentynyl or hexynyl.
- Aryl-C 1 -C 6 -alkyl is, for example, benzyl, phenylethyl, phenylpropyl, phenylbutyl, phenylpentyl or phenylhexyl, where both the phenyl ring and the alkylene chain, as described above, partially or completely with halogens, in particular -F and / or -Cl, or partially with -OH, -OR ', -NR' 2 , -CN, -C (O) OH, -C (O) NR ' 2 , -SO 2 NR' 2 , -C (O) X, -SO 2 OH, -SO 2 X, -SR ",
- Unsubstituted saturated or partially or fully unsaturated cycloalkyl groups having 3-7 C atoms are therefore cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclopentenyl, cyclopenta-1, 3-dienyl, cyclohexenyl, cyclohexa-1,3-dienyl, cyclohexa-1 4-dienyl, phenyl, cycloheptenyl, cyclohepta-1,3-dienyl, cyclohepta-1,4-dienyl or cyclohepta-1,5-dienyl, which may be substituted by Cr to C 6 alkyl groups, again the cycloalkyl group or the C 6 to C r - alkyl-substituted cycloalkyl group with halogen atoms such as F, Cl, Br or I, particularly F or Cl
- C 3 - to C 7 -cycloalkyl is, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or cycloheptyl.
- R ' substituted phenyl, by Cr to C 6 alkyl, C 1 to C 6 alkenyl, -CN, -NO 2 , F, Cl, Br 1 I 1 -OH, -C 1 -C 6 -Akoxy , NR " 2 , -COOH, -SO 2 X ', -SR", -S (O) R ", -SO 2 R", SO 2 NR “ 2 or SO 3 H substituted phenyl, wherein X 1 F, Cl or Br and R "is a non, partially or perfluorinated C 1 - to C 6 -alkyl or C 3 - to C 7 -cycloalkyl as defined for R ', for example, o-, m- or p-methylphenyl, o-, m- or p-ethylphenyl, o-, m- or p-propylphenyl, o-, m- or p-isopropyl
- R 1 to R 4 as heteroaryl is a saturated or unsaturated mono- or bicyclic heterocyclic radical having 5 to 13 ring members understood, wherein 1, 2 or 3 N and / or 1 or 2 S or O atoms may be present and the heterocyclic radical may be mono- or polysubstituted by C 1 - to C ⁇ alkyl, C 1 - to C 6 alkenyl, -CN, -NO 2, F, Cl, Br, I, -OH, -NR "2, -C 1 - C 6 alkoxy, -COOH, -SO 2 X 1 , -SO 2 NFT 2 , -SR ", -S (O) R", -SO 2 R "or SO 3 H may be substituted, wherein X 'and R "have a previously given meaning.
- the heterocyclic radical is preferably substituted or unsubstituted 2- or 3-furyl, 2- or 3-thienyl, 1-, 2- or 3-pyrrolyl, 1-,
- Benzopyrazolyl 2-, 4-, 5-, 6- or 7-benzoxazolyl, 3-, A-, 5-, 6- or 7- benzisoxazolyl, 2-, 4-, 5-, 6- or 7-benzthiazolyl, 2-, 4-, 5-, 6- or 7- benzisothiazolyl, A-, 5-, 6- or 7-benz-2,1,3-oxadiazolyl, 1-, 2-, 3-, 4-, 5 -, 6-, 7- or 8-quinolinyl, 1-, 3-, A-, 5-, 6-, 7- or 8-isoquinolinyl, 1-, 2-, 3-, A- or 9-carbazolyl, 1-, 2-, 3-, 4-, 5-, 6-, 7-, 8- or 9-acridinyl, 3-, A-, 5-, 6-, 7- or 8-cinnolinyl, 2-, 4-, 5-, 6-, 7- or 8-quinazolinyl or 1-, 2- or 3-pyrrolidinyl.
- Heteroaryl-C r C 6 -alkyl is now in analogy to aryl-CrC 6 -alkyl, for example, pyridinyl-methyl, pyridinyl-ethyl, pyridinyl-propyl, pyridinyl-butyl, pyridinyl-pentyl, pyridinyl-hexyl understood, wherein further heterocycles described in this way can be linked to the alkylene chain.
- HetN z + is preferred - -
- the cation [Kt] 2+ may be a [(R 3 ) 3 O] + cation or a [(R 3 ) 3 S] + cation, wherein R 0 denotes straight-chain or branched alkyl groups 1-8 C-atoms or unsubstituted phenyl or with R 0 , OR 0 , N (R °) 2 , CN or halogen-substituted phenyl.
- R 0 of the [(R °) 3 O] + cation or [(R °) 3 S] + cation is preferably straight-chain alkyl having 1-8 C atoms, preferably having 1-4 C atoms, in particular methyl or Ethyl, most preferably ethyl.
- the cation [Kt] z + may also be inorganic, in particular it is a metal cation.
- the metal cation may comprise metals of groups 1 to 12 of the periodic table, in particular alkali metals.
- the metal is selected from the group comprising Li, K, Rb, Cs.
- the cations of the compounds according to the invention are ammonium, phosphonium, guanidinium or heterocyclic cations, more preferably heterocyclic cations (HetN z + ).
- HetN z + is particularly preferably imidazolium, pyrrolidinium or pyridinium, as defined above, wherein the substituents R 1 to R 4 each independently have a meaning as described above.
- HetN 2+ is very particularly preferably imidazolium, where the substituents R 1 to R 4 each independently of one another have the meaning described above.
- the compounds of the formula (I) are very particularly preferably imidazolium, pyrrolidinium, pyridinium, ammonium, phosphonium, guanidinium salts with [(C 2 Fs) 3 PCIF 2 ] -, [( C 3 F 7 ) SPCIF 2 ] " - or [(C 4 Fg) 3 PCIF 2 ] - anions.
- processes for the preparation of compounds with organofluorochlorophosphate anions comprising the reaction of a chloride salt with organofluorophosphoranes.
- the processes according to the invention are preferably those for preparing a compound of the formula (I) as described above, comprising the reaction of [Kt] z + z [CI] " with a compound of the general formula (M)
- the inventive method can be carried out at temperatures of -40 to 180 ° C, preferably, the reaction is carried out at 0 to 5O 0 C and most preferably at room temperature.
- reaction takes place in particular in a solvent, but can also be carried out in the absence of a solvent.
- Suitable solvents are those selected from the group of nitriles, dialkyl carbonates, glymes, dialkyl ethers, cyclic ethers, dimethylfomamides, dimethyl sulfoxides, dichloromethanes, water or mixtures thereof.
- Preferred examples of suitable solvents are acetonitrile and glymes.
- the reaction time is usually 5 minutes to 24 hours, preferably 1 hour to 10 hours.
- the compounds of the invention can be worked up and purified in a manner known to those skilled in the art, for example by removing volatile components in vacuo and drying, optionally further purification steps can follow.
- the present invention furthermore relates to the use of the compounds according to the invention as solvent or solvent additive, as phase transfer catalyst, as extractant, as heat transfer medium, as surface-active substance, as plasticizer, as flame retardant, as additive or as conducting salt and catalysts in chemical processes.
- said compounds are suitable in any type of reaction known to those skilled in the art, e.g. for transition metal- or enzyme-catalyzed reactions, such as hydroformylation reactions, Friedel-Crafts reactions, oligomerization reactions, esterifications or isomerization reactions, the said list being not exhaustive.
- the compounds for the separation of reaction products but also for the separation of impurities can be used, depending on how the solubility of the respective component in the compound of the invention used is.
- the compounds of the formula (I) can also serve as release agents in the separation of a plurality of components, for example in the distillative separation of a plurality of components of a mixture.
- Electrolyte ⁇ , electrochemical cells, capacitors or fuel cells containing at least one compound of the general formula (I) according to the invention are also the subject of the present invention Invention.
- the compounds according to the invention can be used in combination with all materials and additives known to the person skilled in the art, without the need for an inventive step by the person skilled in the art.
- the compounds of the invention are suitable for the preparation of organophosphinates.
- the organofluoro chlorophosphates that initially form may continue to react with water to form corresponding organophosphinates.
- the organofluoro chlorophosphates that initially form may continue to react with water to form corresponding organophosphinates.
- Organophosphinates are also prepared in which the Organofluorochlorophosphate according to the invention are dissolved in water. Accordingly, the organofluorochlorophosphates according to the invention are suitable for use in the preparation of organophosphinates. These organophosphinates themselves represent valuable compounds that can be used, for example, as ionic liquids. Corresponding organophosphinates are known, for example, from WO 2003/087110, the disclosure of which is hereby incorporated by reference. Processes according to the invention for the preparation of organophosphinates comprise the reaction of a compound according to the present invention with water or water-containing solvents or solvent mixtures. This reaction can take place at temperatures of -40 to 200 ° C., at atmospheric pressure, up to pressures of 100 bar. Preferably, the reaction is carried out with water at room temperature and atmospheric pressure.
- Tris (pentafluoroethyl) difluorophosphoran and acetonitrile) are removed in vacuo and the residue is dried in vacuo for two hours at 7 Pa and at 40-50 0 C (temperature of the oil bath). 17.0 g of a viscous, oily material are obtained.
- the yield of tetrabutylammonium tris (pentafluoroethyl) difluoro-chlorophosphate is 95.5%, based on the tetrabutylammonium chloride used.
- the compound is analyzed by NMR spectroscopy.
- Tris (pentafluoroethyl) difluorophosphorane and acetonitrile) are removed in vacuo and the residue is added in vacuo for two hours
- Benzylthethylammonium tris (pentafluoroethyl) difluorochlorophosphate is 98.2%, based on the benzyltriethylammonium chloride used.
- the compound is analyzed by NMR spectroscopy.
- the yield of 1-ethyl-3-methylimidazolium tris (pentafluoroethyl) difluorochlorophosphat is 99.2%, based on the 1-ethyl-3-methylimidazoliumchlorid used.
- the compound is analyzed by NMR spectroscopy.
- Trihexyltetradecylphosphonium tris (pentafluoroethyl) difluorochlorophosphat is 99.6%, based on the Trihexyltetradecyl- phosphonium chloride.
- the compound is analyzed by NMR spectroscopy.
- Tetrakis (dimethylamino) ethylidenium di [tris (pentafluoroethyl) difluorochlorophosphate] is 99.7%, based on the tetrakis (dimethylamino) ethylidenium dichloride used.
- the compound is analyzed by NMR spectroscopy.
- Example 8 Tetramethylammonium tris (pentafluoroethyl) difluorochlorophosphate - -
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Manufacturing & Machinery (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Electrochemistry (AREA)
- Physics & Mathematics (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Thermal Sciences (AREA)
- Materials Engineering (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- General Physics & Mathematics (AREA)
- Inorganic Chemistry (AREA)
- Combustion & Propulsion (AREA)
- Sustainable Development (AREA)
- Sustainable Energy (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Polymers With Sulfur, Phosphorus Or Metals In The Main Chain (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Catalysts (AREA)
- Secondary Cells (AREA)
- Electric Double-Layer Capacitors Or The Like (AREA)
- Inert Electrodes (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009547540A JP5484915B2 (ja) | 2007-01-31 | 2007-12-07 | オルガノフルオロクロロホスフェートアニオンを含む化合物 |
| US12/525,099 US8211277B2 (en) | 2007-01-31 | 2007-12-07 | Compounds containing organofluorochlorophosphate anions |
| AT07856451T ATE483720T1 (de) | 2007-01-31 | 2007-12-07 | Verbindungen enthaltend organofluorochlorophosphatanionen |
| EP07856451A EP2114965B1 (de) | 2007-01-31 | 2007-12-07 | Verbindungen enthaltend organofluorochlorophosphatanionen |
| CN200780050725XA CN101595114B (zh) | 2007-01-31 | 2007-12-07 | 含有机氟氯磷酸根阴离子的化合物 |
| DE502007005301T DE502007005301D1 (de) | 2007-01-31 | 2007-12-07 | Verbindungen enthaltend organofluorochlorophosphatanionen |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102007004698.9 | 2007-01-31 | ||
| DE102007004698A DE102007004698A1 (de) | 2007-01-31 | 2007-01-31 | Verbindungen enthaltend Organofluorochlorophosphatanionen |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008092489A1 true WO2008092489A1 (de) | 2008-08-07 |
Family
ID=39092342
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2007/010646 WO2008092489A1 (de) | 2007-01-31 | 2007-12-07 | Verbindungen enthaltend organofluorochlorophosphatanionen |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US8211277B2 (de) |
| EP (1) | EP2114965B1 (de) |
| JP (1) | JP5484915B2 (de) |
| CN (1) | CN101595114B (de) |
| AT (1) | ATE483720T1 (de) |
| DE (2) | DE102007004698A1 (de) |
| RU (1) | RU2465278C2 (de) |
| WO (1) | WO2008092489A1 (de) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2408051A1 (de) * | 2009-04-22 | 2012-01-18 | Central Glass Company, Limited | Elektrolyt für eine elektrochemische vorrichtung, elektrolytlösung damit und batterie mit nichtwässrigem elektrolyt |
| WO2012041431A1 (de) | 2010-09-27 | 2012-04-05 | Merck Patent Gmbh | Fluoralkylfluorphosphoran-addukte |
| JP2013514288A (ja) * | 2009-12-18 | 2013-04-25 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | (パーフルオロアルキル)フルオロリン酸水素アニオンを有する化合物 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8846922B2 (en) | 2010-09-27 | 2014-09-30 | Merck Patent Gmbh | Functionalized fluoroalkyl fluorophosphate salts |
| JP6501252B2 (ja) * | 2015-03-20 | 2019-04-17 | 東洋合成工業株式会社 | アンモニウムカチオン含有有機塩 |
| JP7012660B2 (ja) | 2016-04-01 | 2022-02-14 | ノームズ テクノロジーズ インコーポレイテッド | リン含有修飾イオン性液体 |
| JP7296893B2 (ja) | 2017-07-17 | 2023-06-23 | ノームズ テクノロジーズ インコーポレイテッド | リン含有電解質 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0929558B1 (de) * | 1996-10-05 | 2001-01-10 | MERCK PATENT GmbH | Lithiumfluorphosphate sowie deren verwendung als leitsalze |
| EP1162204A1 (de) * | 2000-06-09 | 2001-12-12 | MERCK PATENT GmbH | Ionische Flüssigkeiten |
| US6340716B1 (en) * | 1997-07-25 | 2002-01-22 | Acep Inc. | Ionic compounds with delocalized anionic charge, and their use as ion conducting components or as catalysts |
| WO2002085919A1 (de) * | 2001-04-20 | 2002-10-31 | Merck Patent Gmbh | Verfahren zur herstellung von fluoralkylphosphaten |
| WO2006128563A1 (de) * | 2005-06-02 | 2006-12-07 | Merck Patent Gmbh | Ionische flüssigkeiten mit niedriger viskosität |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19846636A1 (de) * | 1998-10-09 | 2000-04-13 | Merck Patent Gmbh | Elektrochemische Synthese von Perfluoralkylfluorophosphoranen |
| DE10038858A1 (de) * | 2000-08-04 | 2002-02-14 | Merck Patent Gmbh | Fluoralkylphosphate zur Anwendung in elektrochemischen Zellen |
| DE10216997A1 (de) | 2002-04-16 | 2003-10-30 | Merck Patent Gmbh | Verfahren zur Herstellung von Bis(perfluoralkyl)phosphinsäuren und deren Salze |
| CN1850592A (zh) * | 2006-05-30 | 2006-10-25 | 天津化工研究设计院 | 六氟磷酸锂的制备方法 |
-
2007
- 2007-01-31 DE DE102007004698A patent/DE102007004698A1/de not_active Withdrawn
- 2007-12-07 JP JP2009547540A patent/JP5484915B2/ja not_active Expired - Fee Related
- 2007-12-07 AT AT07856451T patent/ATE483720T1/de active
- 2007-12-07 US US12/525,099 patent/US8211277B2/en not_active Expired - Fee Related
- 2007-12-07 DE DE502007005301T patent/DE502007005301D1/de active Active
- 2007-12-07 RU RU2009132609/04A patent/RU2465278C2/ru not_active IP Right Cessation
- 2007-12-07 CN CN200780050725XA patent/CN101595114B/zh not_active Expired - Fee Related
- 2007-12-07 EP EP07856451A patent/EP2114965B1/de not_active Not-in-force
- 2007-12-07 WO PCT/EP2007/010646 patent/WO2008092489A1/de active Application Filing
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0929558B1 (de) * | 1996-10-05 | 2001-01-10 | MERCK PATENT GmbH | Lithiumfluorphosphate sowie deren verwendung als leitsalze |
| US6340716B1 (en) * | 1997-07-25 | 2002-01-22 | Acep Inc. | Ionic compounds with delocalized anionic charge, and their use as ion conducting components or as catalysts |
| EP1162204A1 (de) * | 2000-06-09 | 2001-12-12 | MERCK PATENT GmbH | Ionische Flüssigkeiten |
| WO2002085919A1 (de) * | 2001-04-20 | 2002-10-31 | Merck Patent Gmbh | Verfahren zur herstellung von fluoralkylphosphaten |
| WO2006128563A1 (de) * | 2005-06-02 | 2006-12-07 | Merck Patent Gmbh | Ionische flüssigkeiten mit niedriger viskosität |
Non-Patent Citations (1)
| Title |
|---|
| A. N. IVANOV, V. B. SOKOLOV, T. V. GOREVA AND I. V. MARTYNOV: "2,2,3,3-Tetrafluoropropyldichlorophosphite in the Allen reaction with 1,1-dichloro-1-nitrosoalkanes", JOURNAL OF FLUORINE CHEMISTRY, vol. 58, no. 2-3, 1992, pages 374, XP002470493 * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2408051A1 (de) * | 2009-04-22 | 2012-01-18 | Central Glass Company, Limited | Elektrolyt für eine elektrochemische vorrichtung, elektrolytlösung damit und batterie mit nichtwässrigem elektrolyt |
| CN102414902A (zh) * | 2009-04-22 | 2012-04-11 | 中央硝子株式会社 | 电化学设备用电解质、使用其的电解液及非水电解液电池 |
| EP2408051A4 (de) * | 2009-04-22 | 2012-09-19 | Central Glass Co Ltd | Elektrolyt für eine elektrochemische vorrichtung, elektrolytlösung damit und batterie mit nichtwässrigem elektrolyt |
| US8841034B2 (en) | 2009-04-22 | 2014-09-23 | Central Glass Company, Limited | Electrolyte for electrochemical device, electrolyte solution using same, and non-aqueous electrolyte battery |
| JP2013514288A (ja) * | 2009-12-18 | 2013-04-25 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | (パーフルオロアルキル)フルオロリン酸水素アニオンを有する化合物 |
| WO2012041431A1 (de) | 2010-09-27 | 2012-04-05 | Merck Patent Gmbh | Fluoralkylfluorphosphoran-addukte |
| US8829188B2 (en) | 2010-09-27 | 2014-09-09 | Merck Patent Gmbh | Fluoroalkylfluorophosphorane adducts |
Also Published As
| Publication number | Publication date |
|---|---|
| DE502007005301D1 (de) | 2010-11-18 |
| RU2465278C2 (ru) | 2012-10-27 |
| US20100004461A1 (en) | 2010-01-07 |
| RU2009132609A (ru) | 2011-03-10 |
| JP2010516791A (ja) | 2010-05-20 |
| JP5484915B2 (ja) | 2014-05-07 |
| ATE483720T1 (de) | 2010-10-15 |
| US8211277B2 (en) | 2012-07-03 |
| DE102007004698A1 (de) | 2008-08-07 |
| CN101595114A (zh) | 2009-12-02 |
| EP2114965A1 (de) | 2009-11-11 |
| EP2114965B1 (de) | 2010-10-06 |
| CN101595114B (zh) | 2012-09-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2006128563A1 (de) | Ionische flüssigkeiten mit niedriger viskosität | |
| EP1711458B1 (de) | Verfahren zur herstellung von guanidinium-salzen | |
| EP2114965B1 (de) | Verbindungen enthaltend organofluorochlorophosphatanionen | |
| EP1768962B1 (de) | Verfahren zur herstellung von onium-salzen mit geringem chlorid-gehalt | |
| DE102005035103A1 (de) | Verfahren zur Herstellung von Onium-Salzen mit Tetrafluoroborat-Anion mit reduziertem Halogenid-Gehalt | |
| WO2004106288A2 (de) | Ionische flüssigkeiten mit guanidinium-kationen | |
| EP2180002B1 (de) | Verfahren zur Herstellung von Onium-Salzen mit Diakylphosphinat Anionen mit geringem Halogenid-Gehalt | |
| WO2015049034A1 (de) | Perfluoralkylfluor- oder perfluoralkylchlorgermanate | |
| EP1979361B1 (de) | Oxonium- und sulfoniumsalze | |
| EP1730157B1 (de) | Ionische flüssigkeiten mit fluoralkyltriffluorborat-anionen | |
| EP1628952B1 (de) | Ionische flüssigkeiten mit uronium- oder thiouronium-kationen | |
| EP1773848B1 (de) | Salze mit alkoxytris(fluoralkyl)borat-anionen | |
| EP2621939B1 (de) | Funktionalisierte fluoralkylfluorphosphat-salze | |
| EP1685095B1 (de) | Verfahren zur herstellung von organischen salzen mit bis(perfluoralkyl)phosphinat-anionen | |
| EP1831179B1 (de) | Verfahren zur herstellung von onium-salzen mit tetrafluoroborat-anion mit reduziertem halogenid-gehalt | |
| EP2513123B1 (de) | Verbindungen mit (perfluoralkyl)fluorohydrogenphosphat-anionen | |
| DE102010013971A1 (de) | Fluorierungsreagenzien mit (Perfluoralkyl) fluorophosphat-Anion | |
| DE102016010488A1 (de) | Verbindungen mit ein-, bis- und mehrfach geladenen schwach koordinierenden Anionen | |
| DE102004060073A1 (de) | Verfahren zur Hertellung von Onium-Salzen mit Tetrafluoroborat-Anion mit reduziertem Halogenid-Gehalt | |
| DE102004036299A1 (de) | Salze mit Alkoxytris(fluoralkyl)borat-Anionen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780050725.X Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07856451 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007856451 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2009547540 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12525099 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2009132609 Country of ref document: RU Kind code of ref document: A |