WO2007013588A1 - カワラタケ株及びその抽出物及びその利用 - Google Patents
カワラタケ株及びその抽出物及びその利用 Download PDFInfo
- Publication number
- WO2007013588A1 WO2007013588A1 PCT/JP2006/314978 JP2006314978W WO2007013588A1 WO 2007013588 A1 WO2007013588 A1 WO 2007013588A1 JP 2006314978 W JP2006314978 W JP 2006314978W WO 2007013588 A1 WO2007013588 A1 WO 2007013588A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- extract
- power
- strain
- oyster mushroom
- test
- Prior art date
Links
- 235000001674 Agaricus brunnescens Nutrition 0.000 title claims abstract description 10
- 239000000284 extract Substances 0.000 title claims description 138
- 241000222355 Trametes versicolor Species 0.000 title abstract description 4
- 108010012236 Chemokines Proteins 0.000 claims abstract description 32
- 102000019034 Chemokines Human genes 0.000 claims abstract description 30
- 230000014509 gene expression Effects 0.000 claims abstract description 23
- 230000002790 anti-mutagenic effect Effects 0.000 claims abstract description 17
- 208000010110 spontaneous platelet aggregation Diseases 0.000 claims abstract description 14
- 241000221198 Basidiomycota Species 0.000 claims abstract description 13
- 240000001462 Pleurotus ostreatus Species 0.000 claims description 76
- 235000001603 Pleurotus ostreatus Nutrition 0.000 claims description 76
- 239000000203 mixture Substances 0.000 claims description 39
- 239000004480 active ingredient Substances 0.000 claims description 7
- 206010050661 Platelet aggregation inhibition Diseases 0.000 claims description 5
- 230000008859 change Effects 0.000 claims description 4
- 235000013305 food Nutrition 0.000 claims description 4
- 230000005764 inhibitory process Effects 0.000 claims description 4
- 229940122498 Gene expression inhibitor Drugs 0.000 claims description 3
- 239000002246 antineoplastic agent Substances 0.000 claims description 3
- 101000783577 Dendroaspis angusticeps Thrombostatin Proteins 0.000 claims description 2
- 101000783578 Dendroaspis jamesoni kaimosae Dendroaspin Proteins 0.000 claims description 2
- 208000007536 Thrombosis Diseases 0.000 claims description 2
- 229940030600 antihypertensive agent Drugs 0.000 claims description 2
- 239000002220 antihypertensive agent Substances 0.000 claims description 2
- 239000002592 antimutagenic agent Substances 0.000 claims description 2
- 229940127218 antiplatelet drug Drugs 0.000 claims description 2
- 239000002955 immunomodulating agent Substances 0.000 claims description 2
- 230000002584 immunomodulator Effects 0.000 claims description 2
- 229940121354 immunomodulator Drugs 0.000 claims description 2
- 239000008194 pharmaceutical composition Substances 0.000 claims description 2
- 239000000106 platelet aggregation inhibitor Substances 0.000 claims description 2
- 239000003146 anticoagulant agent Substances 0.000 claims 2
- 229940127219 anticoagulant drug Drugs 0.000 claims 2
- 241000282994 Cervidae Species 0.000 claims 1
- 241000406668 Loxodonta cyclotis Species 0.000 claims 1
- 230000007886 mutagenicity Effects 0.000 claims 1
- 231100000299 mutagenicity Toxicity 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 31
- 230000002401 inhibitory effect Effects 0.000 abstract description 26
- 206010028980 Neoplasm Diseases 0.000 abstract description 14
- 230000000259 anti-tumor effect Effects 0.000 abstract description 11
- 230000012010 growth Effects 0.000 abstract description 10
- 230000003276 anti-hypertensive effect Effects 0.000 abstract description 8
- 201000010099 disease Diseases 0.000 abstract description 8
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 8
- 206010061218 Inflammation Diseases 0.000 abstract description 7
- 230000004054 inflammatory process Effects 0.000 abstract description 7
- 230000006872 improvement Effects 0.000 abstract description 6
- 239000002537 cosmetic Substances 0.000 abstract description 5
- 230000035790 physiological processes and functions Effects 0.000 abstract description 5
- 230000002519 immonomodulatory effect Effects 0.000 abstract description 4
- 230000002265 prevention Effects 0.000 abstract description 4
- 208000026935 allergic disease Diseases 0.000 abstract description 3
- 206010020751 Hypersensitivity Diseases 0.000 abstract description 2
- 230000007815 allergy Effects 0.000 abstract description 2
- 230000004071 biological effect Effects 0.000 abstract description 2
- 230000002411 adverse Effects 0.000 abstract 1
- 235000013361 beverage Nutrition 0.000 abstract 1
- 230000002035 prolonged effect Effects 0.000 abstract 1
- 238000012360 testing method Methods 0.000 description 85
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 69
- 239000000843 powder Substances 0.000 description 41
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 33
- 239000002609 medium Substances 0.000 description 28
- 230000036772 blood pressure Effects 0.000 description 20
- 241000195649 Chlorella <Chlorellales> Species 0.000 description 19
- 235000013399 edible fruits Nutrition 0.000 description 19
- 238000002360 preparation method Methods 0.000 description 18
- 240000000599 Lentinula edodes Species 0.000 description 17
- 235000001715 Lentinula edodes Nutrition 0.000 description 17
- 238000011282 treatment Methods 0.000 description 15
- 210000004027 cell Anatomy 0.000 description 14
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 13
- 238000000605 extraction Methods 0.000 description 13
- 210000003491 skin Anatomy 0.000 description 13
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 12
- 230000035488 systolic blood pressure Effects 0.000 description 12
- 229920001817 Agar Polymers 0.000 description 11
- 239000008272 agar Substances 0.000 description 11
- 210000004369 blood Anatomy 0.000 description 11
- 239000008280 blood Substances 0.000 description 11
- 238000009472 formulation Methods 0.000 description 11
- 239000000047 product Substances 0.000 description 11
- 206010020772 Hypertension Diseases 0.000 description 9
- 201000011510 cancer Diseases 0.000 description 9
- 239000007788 liquid Substances 0.000 description 9
- 238000000034 method Methods 0.000 description 8
- 210000001938 protoplast Anatomy 0.000 description 8
- 239000012488 sample solution Substances 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- 108090000790 Enzymes Proteins 0.000 description 7
- 102000004190 Enzymes Human genes 0.000 description 7
- 208000006268 Sarcoma 180 Diseases 0.000 description 7
- 230000009471 action Effects 0.000 description 7
- 229940088598 enzyme Drugs 0.000 description 7
- 230000002757 inflammatory effect Effects 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 240000005893 Pteridium aquilinum Species 0.000 description 6
- 235000009936 Pteridium aquilinum Nutrition 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 description 6
- -1 hemisenseolase Proteins 0.000 description 6
- 238000001727 in vivo Methods 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 230000001629 suppression Effects 0.000 description 6
- 238000010998 test method Methods 0.000 description 6
- 241000699670 Mus sp. Species 0.000 description 5
- 241000237502 Ostreidae Species 0.000 description 5
- 102100028255 Renin Human genes 0.000 description 5
- 108090000783 Renin Proteins 0.000 description 5
- 210000001744 T-lymphocyte Anatomy 0.000 description 5
- 208000010668 atopic eczema Diseases 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 239000003205 fragrance Substances 0.000 description 5
- 230000001631 hypertensive effect Effects 0.000 description 5
- 230000003505 mutagenic effect Effects 0.000 description 5
- 210000000822 natural killer cell Anatomy 0.000 description 5
- 235000020636 oyster Nutrition 0.000 description 5
- 238000012545 processing Methods 0.000 description 5
- 208000017520 skin disease Diseases 0.000 description 5
- 231100000456 subacute toxicity Toxicity 0.000 description 5
- FUFLCEKSBBHCMO-UHFFFAOYSA-N 11-dehydrocorticosterone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)C(=O)CO)C4C3CCC2=C1 FUFLCEKSBBHCMO-UHFFFAOYSA-N 0.000 description 4
- 101800000734 Angiotensin-1 Proteins 0.000 description 4
- 102400000344 Angiotensin-1 Human genes 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- MFYSYFVPBJMHGN-ZPOLXVRWSA-N Cortisone Chemical compound O=C1CC[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 MFYSYFVPBJMHGN-ZPOLXVRWSA-N 0.000 description 4
- MFYSYFVPBJMHGN-UHFFFAOYSA-N Cortisone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)(O)C(=O)CO)C4C3CCC2=C1 MFYSYFVPBJMHGN-UHFFFAOYSA-N 0.000 description 4
- 108010029541 Laccase Proteins 0.000 description 4
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 4
- 229930006000 Sucrose Natural products 0.000 description 4
- 230000002776 aggregation Effects 0.000 description 4
- 238000004220 aggregation Methods 0.000 description 4
- 230000000172 allergic effect Effects 0.000 description 4
- ORWYRWWVDCYOMK-HBZPZAIKSA-N angiotensin I Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC(C)C)C(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](N)CC(O)=O)C(C)C)C1=CC=C(O)C=C1 ORWYRWWVDCYOMK-HBZPZAIKSA-N 0.000 description 4
- 238000007796 conventional method Methods 0.000 description 4
- 229960004544 cortisone Drugs 0.000 description 4
- 238000009402 cross-breeding Methods 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 230000037406 food intake Effects 0.000 description 4
- 238000007710 freezing Methods 0.000 description 4
- 230000008014 freezing Effects 0.000 description 4
- 235000011187 glycerol Nutrition 0.000 description 4
- 230000028644 hyphal growth Effects 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 239000003471 mutagenic agent Substances 0.000 description 4
- 231100000707 mutagenic chemical Toxicity 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 229960004793 sucrose Drugs 0.000 description 4
- 229940058015 1,3-butylene glycol Drugs 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 241000190633 Cordyceps Species 0.000 description 3
- 229920000858 Cyclodextrin Polymers 0.000 description 3
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 102000004890 Interleukin-8 Human genes 0.000 description 3
- 108090001007 Interleukin-8 Proteins 0.000 description 3
- 230000002159 abnormal effect Effects 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 235000019437 butane-1,3-diol Nutrition 0.000 description 3
- 229940041514 candida albicans extract Drugs 0.000 description 3
- 238000005119 centrifugation Methods 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 239000007799 cork Substances 0.000 description 3
- 238000012258 culturing Methods 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 230000000593 degrading effect Effects 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 230000035487 diastolic blood pressure Effects 0.000 description 3
- 239000012153 distilled water Substances 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 3
- 210000004623 platelet-rich plasma Anatomy 0.000 description 3
- 150000003180 prostaglandins Chemical class 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 230000001954 sterilising effect Effects 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- 239000006228 supernatant Substances 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- 230000002195 synergetic effect Effects 0.000 description 3
- 230000036962 time dependent Effects 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- 239000012138 yeast extract Substances 0.000 description 3
- PJVXUVWGSCCGHT-ZPYZYFCMSA-N (2r,3s,4r,5r)-2,3,4,5,6-pentahydroxyhexanal;(3s,4r,5r)-1,3,4,5,6-pentahydroxyhexan-2-one Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O.OC[C@@H](O)[C@@H](O)[C@H](O)C(=O)CO PJVXUVWGSCCGHT-ZPYZYFCMSA-N 0.000 description 2
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 108010059892 Cellulase Proteins 0.000 description 2
- 108010022172 Chitinases Proteins 0.000 description 2
- 102000012286 Chitinases Human genes 0.000 description 2
- 206010012438 Dermatitis atopic Diseases 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 241000233866 Fungi Species 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 239000004166 Lanolin Substances 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- 108010059820 Polygalacturonase Proteins 0.000 description 2
- 201000004681 Psoriasis Diseases 0.000 description 2
- 241000831652 Salinivibrio sharmensis Species 0.000 description 2
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 2
- 240000006794 Volvariella volvacea Species 0.000 description 2
- 244000085595 Zizania latifolia Species 0.000 description 2
- 235000004259 Zizania latifolia Nutrition 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 231100000215 acute (single dose) toxicity testing Toxicity 0.000 description 2
- 231100000403 acute toxicity Toxicity 0.000 description 2
- 230000007059 acute toxicity Effects 0.000 description 2
- 238000005276 aerator Methods 0.000 description 2
- 229940114079 arachidonic acid Drugs 0.000 description 2
- 235000021342 arachidonic acid Nutrition 0.000 description 2
- 201000008937 atopic dermatitis Diseases 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 239000003130 blood coagulation factor inhibitor Substances 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 210000002421 cell wall Anatomy 0.000 description 2
- 229940106157 cellulase Drugs 0.000 description 2
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 2
- 230000001684 chronic effect Effects 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 235000008504 concentrate Nutrition 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 230000002354 daily effect Effects 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 229960001617 ethyl hydroxybenzoate Drugs 0.000 description 2
- 235000010228 ethyl p-hydroxybenzoate Nutrition 0.000 description 2
- 239000004403 ethyl p-hydroxybenzoate Substances 0.000 description 2
- NUVBSKCKDOMJSU-UHFFFAOYSA-N ethylparaben Chemical compound CCOC(=O)C1=CC=C(O)C=C1 NUVBSKCKDOMJSU-UHFFFAOYSA-N 0.000 description 2
- 108010093305 exopolygalacturonase Proteins 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 230000001605 fetal effect Effects 0.000 description 2
- 210000002950 fibroblast Anatomy 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 235000013355 food flavoring agent Nutrition 0.000 description 2
- 150000004676 glycans Chemical class 0.000 description 2
- 235000012907 honey Nutrition 0.000 description 2
- 210000000987 immune system Anatomy 0.000 description 2
- 210000004969 inflammatory cell Anatomy 0.000 description 2
- 208000027866 inflammatory disease Diseases 0.000 description 2
- XKTZWUACRZHVAN-VADRZIEHSA-N interleukin-8 Chemical compound C([C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@@H](NC(C)=O)CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CCSC)C(=O)N1[C@H](CCC1)C(=O)N1[C@H](CCC1)C(=O)N[C@@H](C)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC=1C=CC(O)=CC=1)C(=O)N[C@H](CO)C(=O)N1[C@H](CCC1)C(N)=O)C1=CC=CC=C1 XKTZWUACRZHVAN-VADRZIEHSA-N 0.000 description 2
- 229940096397 interleukin-8 Drugs 0.000 description 2
- 238000005342 ion exchange Methods 0.000 description 2
- 238000009533 lab test Methods 0.000 description 2
- 229940039717 lanolin Drugs 0.000 description 2
- 235000019388 lanolin Nutrition 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 230000007721 medicinal effect Effects 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- 231100000219 mutagenic Toxicity 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 2
- 230000003204 osmotic effect Effects 0.000 description 2
- 239000002304 perfume Substances 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920001282 polysaccharide Polymers 0.000 description 2
- 239000005017 polysaccharide Substances 0.000 description 2
- 230000003449 preventive effect Effects 0.000 description 2
- 238000010298 pulverizing process Methods 0.000 description 2
- 239000008213 purified water Substances 0.000 description 2
- LXNHXLLTXMVWPM-UHFFFAOYSA-N pyridoxine Chemical compound CC1=NC=C(CO)C(CO)=C1O LXNHXLLTXMVWPM-UHFFFAOYSA-N 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 210000001626 skin fibroblast Anatomy 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 2
- 238000011699 spontaneously hypertensive rat Methods 0.000 description 2
- 238000004659 sterilization and disinfection Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 150000003595 thromboxanes Chemical class 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 230000004614 tumor growth Effects 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 238000003809 water extraction Methods 0.000 description 2
- 239000002023 wood Substances 0.000 description 2
- PFTAWBLQPZVEMU-DZGCQCFKSA-N (+)-catechin Chemical compound C1([C@H]2OC3=CC(O)=CC(O)=C3C[C@@H]2O)=CC=C(O)C(O)=C1 PFTAWBLQPZVEMU-DZGCQCFKSA-N 0.000 description 1
- RMBLTWUTZAFABA-XVSDJDOKSA-N (5z,8z,11z,14z)-icosa-5,8,11,14-tetraenoic acid;sodium Chemical compound [Na].CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O RMBLTWUTZAFABA-XVSDJDOKSA-N 0.000 description 1
- FFJCNSLCJOQHKM-CLFAGFIQSA-N (z)-1-[(z)-octadec-9-enoxy]octadec-9-ene Chemical compound CCCCCCCC\C=C/CCCCCCCCOCCCCCCCC\C=C/CCCCCCCC FFJCNSLCJOQHKM-CLFAGFIQSA-N 0.000 description 1
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Chemical compound FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 1
- CMCBDXRRFKYBDG-UHFFFAOYSA-N 1-dodecoxydodecane Chemical compound CCCCCCCCCCCCOCCCCCCCCCCCC CMCBDXRRFKYBDG-UHFFFAOYSA-N 0.000 description 1
- FDCJDKXCCYFOCV-UHFFFAOYSA-N 1-hexadecoxyhexadecane Chemical compound CCCCCCCCCCCCCCCCOCCCCCCCCCCCCCCCC FDCJDKXCCYFOCV-UHFFFAOYSA-N 0.000 description 1
- HMFKFHLTUCJZJO-UHFFFAOYSA-N 2-{2-[3,4-bis(2-hydroxyethoxy)oxolan-2-yl]-2-(2-hydroxyethoxy)ethoxy}ethyl dodecanoate Chemical compound CCCCCCCCCCCC(=O)OCCOCC(OCCO)C1OCC(OCCO)C1OCCO HMFKFHLTUCJZJO-UHFFFAOYSA-N 0.000 description 1
- DAUAQNGYDSHRET-UHFFFAOYSA-N 3,4-dimethoxybenzoic acid Chemical compound COC1=CC=C(C(O)=O)C=C1OC DAUAQNGYDSHRET-UHFFFAOYSA-N 0.000 description 1
- ARZWATDYIYAUTA-UHFFFAOYSA-N 3-methyl-3H-imidazo[4,5-f]quinolin-2-amine Chemical compound C1=CC2=NC=CC=C2C2=C1N(C)C(N)=N2 ARZWATDYIYAUTA-UHFFFAOYSA-N 0.000 description 1
- 101150044182 8 gene Proteins 0.000 description 1
- 241001327634 Agaricus blazei Species 0.000 description 1
- 108010064733 Angiotensins Proteins 0.000 description 1
- 102000015427 Angiotensins Human genes 0.000 description 1
- 235000017166 Bambusa arundinacea Nutrition 0.000 description 1
- 235000017491 Bambusa tulda Nutrition 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 241000195940 Bryophyta Species 0.000 description 1
- 235000002568 Capsicum frutescens Nutrition 0.000 description 1
- 241000218645 Cedrus Species 0.000 description 1
- 244000249214 Chlorella pyrenoidosa Species 0.000 description 1
- 235000007091 Chlorella pyrenoidosa Nutrition 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- 101710088194 Dehydrogenase Proteins 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- 206010012442 Dermatitis contact Diseases 0.000 description 1
- 206010013786 Dry skin Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 101710121765 Endo-1,4-beta-xylanase Proteins 0.000 description 1
- 239000001116 FEMA 4028 Substances 0.000 description 1
- 241001070947 Fagus Species 0.000 description 1
- 241001138420 Fagus crenata Species 0.000 description 1
- 235000010099 Fagus sylvatica Nutrition 0.000 description 1
- 244000307700 Fragaria vesca Species 0.000 description 1
- 235000016623 Fragaria vesca Nutrition 0.000 description 1
- 235000011363 Fragaria x ananassa Nutrition 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 102100031181 Glyceraldehyde-3-phosphate dehydrogenase Human genes 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 206010022489 Insulin Resistance Diseases 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- GXCLVBGFBYZDAG-UHFFFAOYSA-N N-[2-(1H-indol-3-yl)ethyl]-N-methylprop-2-en-1-amine Chemical compound CN(CCC1=CNC2=C1C=CC=C2)CC=C GXCLVBGFBYZDAG-UHFFFAOYSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 241000123107 Phellinus Species 0.000 description 1
- 244000082204 Phyllostachys viridis Species 0.000 description 1
- 235000015334 Phyllostachys viridis Nutrition 0.000 description 1
- 241000222341 Polyporaceae Species 0.000 description 1
- 101710093543 Probable non-specific lipid-transfer protein Proteins 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- 241000607142 Salmonella Species 0.000 description 1
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 1
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 1
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 1
- 102100040247 Tumor necrosis factor Human genes 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- GPVDHNVGGIAOQT-UHFFFAOYSA-N Veratric acid Natural products COC1=CC=C(C(O)=O)C(OC)=C1 GPVDHNVGGIAOQT-UHFFFAOYSA-N 0.000 description 1
- 210000001015 abdomen Anatomy 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 231100000460 acute oral toxicity Toxicity 0.000 description 1
- 238000011047 acute toxicity test Methods 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N alpha-methyl toluene Natural products CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 230000002019 anti-mutation Effects 0.000 description 1
- 230000002421 anti-septic effect Effects 0.000 description 1
- 230000002785 anti-thrombosis Effects 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 229940064004 antiseptic throat preparations Drugs 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 239000011425 bamboo Substances 0.000 description 1
- 239000003788 bath preparation Substances 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- WHGYBXFWUBPSRW-FOUAGVGXSA-N beta-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO WHGYBXFWUBPSRW-FOUAGVGXSA-N 0.000 description 1
- 235000011175 beta-cyclodextrine Nutrition 0.000 description 1
- 229960004853 betadex Drugs 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000010876 biochemical test Methods 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 238000005422 blasting Methods 0.000 description 1
- 239000007844 bleaching agent Substances 0.000 description 1
- 230000023555 blood coagulation Effects 0.000 description 1
- 230000004531 blood pressure lowering effect Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- ADRVNXBAWSRFAJ-UHFFFAOYSA-N catechin Natural products OC1Cc2cc(O)cc(O)c2OC1c3ccc(O)c(O)c3 ADRVNXBAWSRFAJ-UHFFFAOYSA-N 0.000 description 1
- 235000005487 catechin Nutrition 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000007705 chemical test Methods 0.000 description 1
- 231100000045 chemical toxicity Toxicity 0.000 description 1
- 229950001002 cianidanol Drugs 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 239000013065 commercial product Substances 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 235000009508 confectionery Nutrition 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 208000010247 contact dermatitis Diseases 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 235000013681 dietary sucrose Nutrition 0.000 description 1
- 235000015872 dietary supplement Nutrition 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 235000011869 dried fruits Nutrition 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000000797 effect on infection Effects 0.000 description 1
- 230000002124 endocrine Effects 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 210000003979 eosinophil Anatomy 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- 238000002481 ethanol extraction Methods 0.000 description 1
- 239000000469 ethanolic extract Substances 0.000 description 1
- 229940071106 ethylenediaminetetraacetate Drugs 0.000 description 1
- 230000003203 everyday effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000012091 fetal bovine serum Substances 0.000 description 1
- 235000013312 flour Nutrition 0.000 description 1
- 239000006562 flour medium Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 235000013376 functional food Nutrition 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 108020004445 glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 229940094952 green tea extract Drugs 0.000 description 1
- 235000020688 green tea extract Nutrition 0.000 description 1
- 230000009422 growth inhibiting effect Effects 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 239000011121 hardwood Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 235000013402 health food Nutrition 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 210000002865 immune cell Anatomy 0.000 description 1
- 230000004957 immunoregulator effect Effects 0.000 description 1
- 230000003308 immunostimulating effect Effects 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 238000011081 inoculation Methods 0.000 description 1
- 239000002054 inoculum Substances 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- 239000008274 jelly Substances 0.000 description 1
- 210000002510 keratinocyte Anatomy 0.000 description 1
- 239000004816 latex Substances 0.000 description 1
- 229920000126 latex Polymers 0.000 description 1
- 150000002617 leukotrienes Chemical class 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000013028 medium composition Substances 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 239000004200 microcrystalline wax Substances 0.000 description 1
- 235000019808 microcrystalline wax Nutrition 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 235000011929 mousse Nutrition 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 231100000957 no side effect Toxicity 0.000 description 1
- 108010090409 novozym 234 Proteins 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 238000011580 nude mouse model Methods 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- DXGLGDHPHMLXJC-UHFFFAOYSA-N oxybenzone Chemical compound OC1=CC(OC)=CC=C1C(=O)C1=CC=CC=C1 DXGLGDHPHMLXJC-UHFFFAOYSA-N 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 210000002381 plasma Anatomy 0.000 description 1
- 108010001062 polysaccharide-K Proteins 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 238000012802 pre-warming Methods 0.000 description 1
- 210000000229 preadipocyte Anatomy 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 150000003138 primary alcohols Chemical class 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 239000003223 protective agent Substances 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- RADKZDMFGJYCBB-UHFFFAOYSA-N pyridoxal hydrochloride Natural products CC1=NC=C(CO)C(C=O)=C1O RADKZDMFGJYCBB-UHFFFAOYSA-N 0.000 description 1
- 238000003753 real-time PCR Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 238000011076 safety test Methods 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 238000010187 selection method Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000001593 sorbitan monooleate Substances 0.000 description 1
- 235000011069 sorbitan monooleate Nutrition 0.000 description 1
- 229940035049 sorbitan monooleate Drugs 0.000 description 1
- 230000002269 spontaneous effect Effects 0.000 description 1
- 238000013300 spontaneous hypertensive rat model Methods 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 239000003270 steroid hormone Substances 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 238000011410 subtraction method Methods 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 235000012976 tarts Nutrition 0.000 description 1
- 235000013616 tea Nutrition 0.000 description 1
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- RZWIIPASKMUIAC-VQTJNVASSA-N thromboxane Chemical compound CCCCCCCC[C@H]1OCCC[C@@H]1CCCCCCC RZWIIPASKMUIAC-VQTJNVASSA-N 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 1
- 230000036269 ulceration Effects 0.000 description 1
- 238000003828 vacuum filtration Methods 0.000 description 1
- 239000011726 vitamin B6 Substances 0.000 description 1
- 235000019158 vitamin B6 Nutrition 0.000 description 1
- 229940011671 vitamin b6 Drugs 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L17/00—Food-from-the-sea products; Fish products; Fish meal; Fish-egg substitutes; Preparation or treatment thereof
- A23L17/60—Edible seaweed
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L31/00—Edible extracts or preparations of fungi; Preparation or treatment thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L5/00—Preparation or treatment of foods or foodstuffs, in general; Food or foodstuffs obtained thereby; Materials therefor
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/06—Fungi, e.g. yeasts
- A61K36/07—Basidiomycota, e.g. Cryptococcus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9706—Algae
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9728—Fungi, e.g. yeasts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/16—Emollients or protectives, e.g. against radiation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/14—Fungi; Culture media therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/14—Fungi; Culture media therefor
- C12N1/145—Fungal isolates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P39/00—Processes involving microorganisms of different genera in the same process, simultaneously
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L21/00—Marmalades, jams, jellies or the like; Products from apiculture; Preparation or treatment thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/125—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives containing carbohydrate syrups; containing sugars; containing sugar alcohols; containing starch hydrolysates
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12R—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES C12C - C12Q, RELATING TO MICROORGANISMS
- C12R2001/00—Microorganisms ; Processes using microorganisms
- C12R2001/645—Fungi ; Processes using fungi
Definitions
- the present invention relates to a power oyster mushroom strain, an extract thereof, and use thereof, and in particular, organisms such as a platelet aggregation inhibitory action, a chemokine gene expression inhibitory action, an antimutagenic action, an anticancer action, and an antihypertensive action.
- the present invention relates to a power oyster mushroom strain excellent in biological activity.
- mushrooms are used not only for food but also for medicinal purposes, and mushrooms having various medicinal effects are known.
- Rikitake is called “unshi” in Chinese medicine and is said to have antitumor activity.
- an antitumor agent (generic name: PSK, trade name: Krestin), which has been developed for the protein polysaccharide strength derived from the power oyster mushroom, is used.
- Patent Document 3 In recent years, selective binding to TGF- ⁇ and PDGF (patent document 1), immune system preventive effect on cancer and infection by administration to immature animals (patent document 2), suppression of preadipocyte differentiation Effects (Patent Document 3) have been reported!
- Patent Document 1 JP-A-8-113540
- Patent Document 2 JP-A-11-60495
- Patent Document 3 Japanese Patent Laid-Open No. 2004-75640
- the present invention has been made in view of the above-mentioned problems of the prior art, and an object of the present invention is to provide a novel power oyster mushroom strain that is extremely excellent in useful activity and use thereof. Means for solving the problem
- the present inventors have conducted extensive research on cross-breeding for selling or wild power oyster mushrooms.
- the present inventors have studied platelet aggregation inhibition, chemokine gene expression inhibition, antimutation
- the present inventors have also found that the action is synergistically improved by further combining microalgae and other basidiomycetes with this power oyster, and the present invention has been completed.
- the power oyster mushroom strain used in the present invention is a power oyster mushroom strain having a deposit number power SFERM BP-10633.
- the platelet aggregation inhibitor, the chemokine gene expression inhibitor, the antimutagenic agent, the antitumor agent, the antihypertensive agent, or the immunomodulator of the present invention contains the above-described power oyster mushroom and Z or an extract thereof as active ingredients.
- the above-described power oyster mushrooms and Z or an extract thereof can be blended in oral compositions such as foods and drinks and pharmaceutical compositions.
- the power oyster mushroom and Z or an extract thereof can be blended in a composition for external use of skin such as cosmetics and external preparations for pharmaceutical use.
- microalgae and other basidiomycetes and their collective strength as well as the above-described power oyster mushroom and Z or an extract thereof.
- the specific power oyster mushroom and Z or an extract thereof of the present invention have excellent platelet aggregation inhibitory action, chemokine gene expression inhibitory action, antimutagenic action, antitumor action, antihypertensive action, immunomodulatory action. Have Furthermore, the combination of microalgae and other basidiomycetes will synergistically improve their action.
- the power oyster mushroom of the present invention is highly safe and has a low possibility of causing side effects even after long-term use. Food and drink, cosmetics for the purpose of prevention and improvement of inflammation, allergies, tumors and other diseases, Useful for pharmaceuticals.
- the power oyster mushroom strain of the present invention has a very high hyphal growth and physiological function compared to conventional varieties, so that it can be used stably.
- FIG. 1 Power Strains of the present invention (Test strain 1, FERM BP-10633), other produced hybrid strains (Test strains 2 to 4), parent strain A and parent strain B used for test strain production, conventional It is the figure which compared the platelet aggregation, the chemokine gene expression inhibitory effect, and the antimutagenic action of the fruit body hot water extract about the cultivars, Kojiwaratake I and Kishiwaratake mushroom (control is DMSO).
- FIG. 2 Platelet aggregation inhibitory action, chemokine gene expression inhibitory action, antimutagenic action of hot water extract and hydrous ethanol extract of fruiting body of power oyster mushroom strain (Test strain 1, FERM BP-10633) of the present invention (Control is DMSO).
- FIG. 3 Chloroella and Z or Shiitake fruiting body hot water extract mixed with the hot water extract of the power oyster mushroom strain (Test strain 1, FERM BP-10633) of the present invention
- FIG. 5 is a diagram showing a synergistic effect on platelet aggregation inhibitory action, chemokine gene expression inhibitory action, and antimutagenic action (control is DMSO).
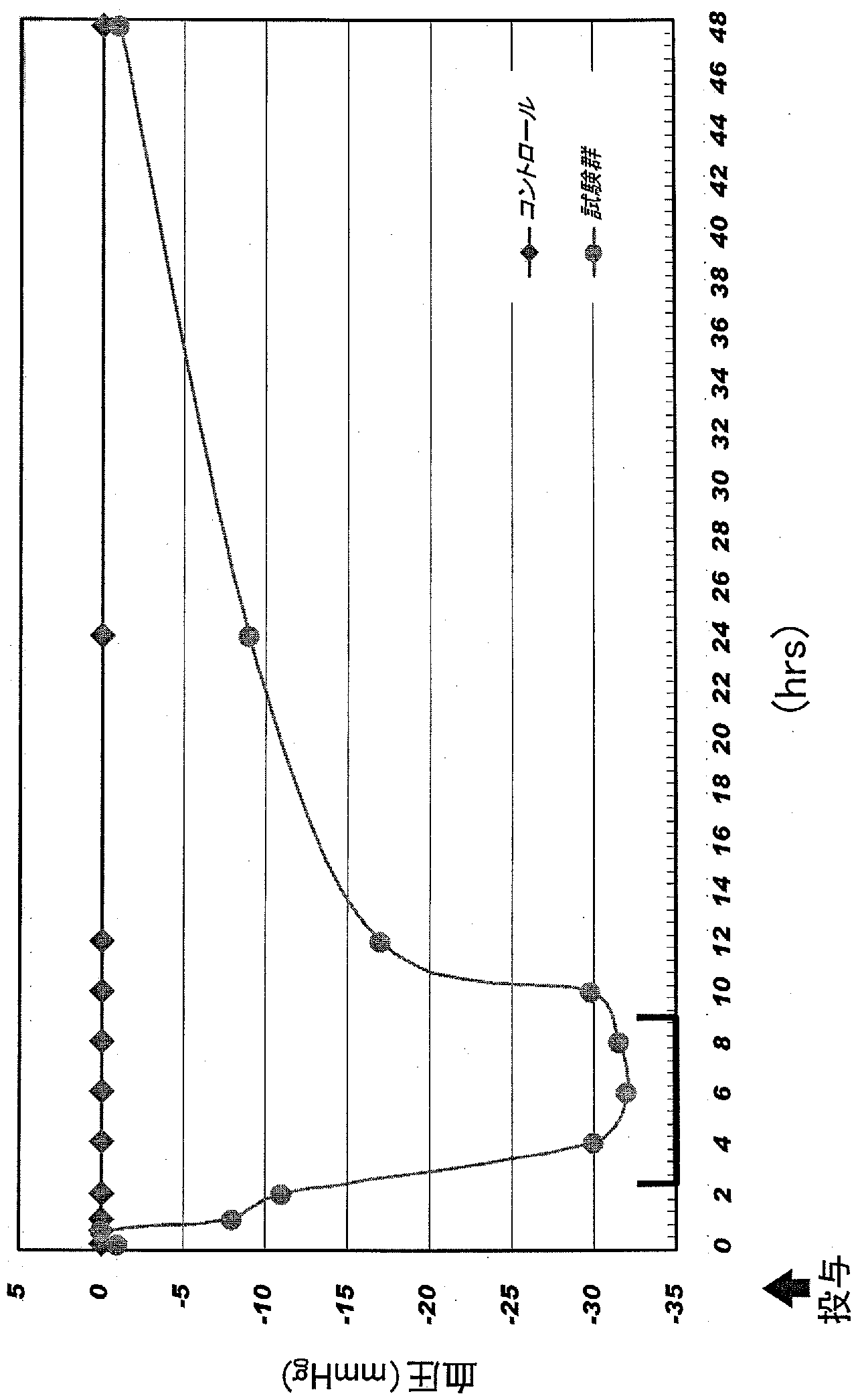
- FIG. 4 Contrast with control (non-administration) after a single administration to SHR of a composition containing a hot water extract of the fruiting body of the power oyster mushroom strain of the present invention (test strain 1, FERM BP-10633). It is a figure which shows the time-dependent change of systolic blood pressure.
- FIG. 5 A composition containing a hot water extract of the fruit body of the power oyster mushroom strain of the present invention (test strain 1, FERM BP-10633) when administered chronically to SHR and when not administered (control) It is a figure which shows the time-dependent change of systolic blood pressure.
- FIG. 6 (a) Expansion when a composition containing a hot water extract of the fruit body of the power oyster mushroom strain of the present invention (test strain 1, FERM BP-10633) is chronically administered to hypertensive patients. It is a figure which shows the time-dependent change of a systolic blood pressure, and (b) systolic blood pressure.
- FIG. 7 (a) Blood renin concentration when a composition containing a hot water extract of the fruit body of the power oyster mushroom strain of the present invention (test strain 1, FERM BP-10633) is chronically administered to hypertensive patients. (B) and (b) are diagrams showing the time course of blood angiotensin I concentration.
- FIG. 8 A composition containing a hot water extract of the fruit body of the power oyster mushroom strain of the present invention (test strain 1, FERM BP-10633) was chronically administered to 30 healthy individuals for 1 month and then for 2 months.
- FIG. 4 is a graph showing changes over time in the number of T cells and the number of NK cells when administration is stopped.
- the power oyster mushroom used in the present invention is Trametes versicolor (L .: Fr.) Pilat belonging to the genus Corpioaceae (Polyporaceae), and is a patent biological deposit center of the National Institute of Advanced Industrial Science and Technology. It is a power waratake strain (trade name: Tanaka Yoshiho strain) with a deposit number of FERM BP-10633, which was deposited internationally on June 28, 2006. This was transferred from the deposit number FERM P-20377 (deposited on January 26, 2005) deposited at the center. All were deposited by Fumiyo Eguchi, one of the inventors.
- the main power oyster mushroom strain is a new power oyster mushroom strain obtained by cross-breeding using wild protoplasty technology from wild power oyster mushrooms that grew naturally in Kumamoto Prefecture, compared to conventional varieties and parental strains. It has the highest activity in hyphal growth and physiological functions, platelet aggregation inhibitory action, chemokine gene expression inhibitory action, antimutagenic action, and antitumor action. It also has high activity in antihypertensive action and immunosuppressive action. The production method, selection method and effects of this strain are described in detail in the examples.
- the power oyster mushroom may be a fruit body, an extract of the fruit body, or a dried product of the extract, and the fruit body may be either raw or dried. From the standpoints of handling, storage, extraction efficiency, etc., dried fruit bodies are desirable. In order to efficiently ingest an effective amount, it is preferable to use a dry powder of fruit body or an extract thereof, and the use of the extract is particularly preferable.
- fruit bodies are preferably used, but the effect can be expected for the mycelium.
- mycelium live or dry mycelium obtained by culturing inoculum in a medium containing a carbon source and a nitrogen source can be used. The dried one is convenient.
- a known cell wall degrading enzyme or polysaccharide degrading enzyme can be used.
- 8-Dalkanase, etc. can be used alone or in combination of two or more kinds.
- Enzymatic treatment of force oyster mushroom is added to force oyster mushroom that has been appropriately shredded or crushed, as an aqueous solution.
- an extraction solvent is added to the oyster mushroom that has been subjected to the tissue destruction treatment as described above, and the mixture is appropriately stirred and extracted. What is necessary is just to process.
- the extraction solvent is not particularly limited, and examples thereof include primary alcohols such as water, methanol and ethanol, polyhydric alcohols such as 1,3-butylene glycol and propylene glycol, lower alkyl esters such as ethyl acetate, benzene and hexane. And hydrocarbons such as ethyl ether, acetone and the like. These may be used in combination of two or more.
- Preferred extraction solvents include water, methanol, ethanol, 1,3-butylene glycol, or a mixed solvent thereof. In particular, a product obtained by extracting dry powder of power oyster mushroom fruit body with hot water is preferable.
- the extraction operation may be performed under normal pressure and at room temperature, but may also be performed under a pressure of 1 to 5 atmospheres, a pressure of 60 to 150 ° C, and heating. Specifically, for example, after extraction with hot water or the like, it is preferable to separate into an extract and a residue by centrifugal separation or vacuum filtration.
- the extraction residue may be re-extracted under the same conditions as described above, and this extraction operation may be repeated several times.
- the extract may be an extract or a dried product thereof.
- the extract is removed by subjecting the extract to treatments such as vacuum concentration, sterilization, freeze drying, and spray drying.
- Mycelium elongation is the best at around 27 ° C, and low temperature treatment of 10-12 ° C is suitable for fruit body development.
- the cultivation period takes 20 to 25 days for the hyphae to spread, and about 20 days for subsequent fruiting. 2.
- a yield of about 200g can be expected with 5kg capacity bag cultivation.
- Culture medium The base sawdust is suitable for most hardwoods, and cedar hydrolyzed can be used.
- the power oyster mushroom strain of the present invention is excellent in platelet aggregation inhibitory action and chemokine gene expression inhibitory action, and thus is useful for preventing thrombus formation 'improvement, improving blood flow, preventing inflammatory or allergic diseases' etc. .
- a preventive / ameliorating effect can be expected for inflammatory or allergic skin diseases.
- the power oyster mushroom strain of the present invention is excellent in antimutagenic action and antitumor action.
- the power oyster mushroom strain of the present invention capable of suppressing the activity of the mutagen is useful for the prevention of such diseases. It is also useful for inhibiting the growth of tumor cells.
- the power oyster mushroom strain of the present invention has an antihypertensive action and can improve hypertension.
- the power oyster mushroom strain of the present invention has an immunomodulatory action, and therefore can contribute to disease prevention and health maintenance by adjusting the immune system in vivo.
- the force oyster mushroom extract dry mass
- the force oyster mushroom extract is an adult (60 kg body weight) per day.
- Chlorella spp. can be preferably used, and in particular, Chlorella pyrenoidaosa can be preferably used.
- the present invention is not limited to this.
- basidiomycetes at least one selected from agaritas (Agaricus blazei), mesimacob (Phellinu s linteus), shitake (Lentinus edodes) and cordyceps sinesis power can be preferably used.
- microalgae and basidiomycetes As for the extraction method of these microalgae and basidiomycetes, it can be carried out by the extraction method of early oyster mushrooms or other known methods. [0025] Some of the above-mentioned microalgae and basidiomycetes have been reported to have antitumor activity, but when used in combination with the power oyster mushroom of the present invention, the activity is higher than when they are used alone. Synergistically improve. Also, a synergistic improvement effect is observed in the platelet aggregation inhibitory action, chemokine gene expression inhibitory action, and antimutagenic action.
- the mixing ratio of the two is not particularly limited, but the ratio of the power oyster mushroom and Z or its extract to the microalgae, other basidiomycetes and the total amount of these extracts is from 1: 1 to 1 in mass ratio.
- the range is preferably 25: 1, more preferably 5: 3 to 15: 1.
- an oral composition is prepared using the power oyster mushroom or extract thereof according to the present invention
- powders, tablets, capsules, granules, teas, suspending agents, fluid extracts are prepared by conventional methods. , Liquids, syrups and the like.
- normal formulation carriers such as excipients, binders, disintegrants, lubricants, coloring agents, flavoring agents, and flavoring agents are used as necessary. be able to. If necessary, apply a coating with an appropriate coating agent.
- a power oyster extract may be blended with a generally known skin external preparation base.
- the external preparation for skin of the present invention is usually used for external preparations for skin, such as cosmetics and pharmaceuticals, for example, whitening agents, moisturizers, antioxidants, antiseptics, oily components, ultraviolet absorption. Agents, surfactants, thickeners, alcohols, powder ingredients, coloring materials, fragrances, aqueous ingredients, water, polymer compounds, chelating agents, pH adjusters, vitamins, amino acids, various skin nutrients Can be appropriately blended as necessary.
- the topical skin preparation can be applied as pharmaceuticals, quasi drugs, and cosmetics, and the dosage forms are ointments, creams, emulsions, lotions, knocks, bath preparations, sheet preparations, mousses, sprays, sticks If it is conventionally used for skin external preparations, it will not be restrict
- the amount of oyster mushroom compounded in these oral compositions or external skin compositions can be determined appropriately according to the purpose and application, etc., but as a normal oyster mushroom extract (dry mass), 0001% by mass or more. Preferably 0.001 to 20% by weight, Is 0.01 to 10 mass 0/0.
- the power oyster mushroom strain FERM BP-10633 is a physico-biochemical test and a pharmacological activity test among test strains obtained by the cross breeding method using protoplast technology. It was selected and adopted as the most superior strain (test strain 1) compared to conventional varieties and parent strains.
- the binuclear mycelium obtained by the general cross breeding method of mushrooms is examined under a microscope. Observed to obtain a plurality of test strains (hybrid strains).
- the binuclear mycelium was aseptically isolated from the wild power oyster mushroom fruit body B that had grown naturally in the Kikuchi basin in Kumamoto Prefecture.
- This mycelium was cultured for 7 days in SMY agar medium (containing 1% sucrose, 1% malt extract, 0.4% yeast extract, 2% agar).
- the obtained mycelium was a cork borer with an inner diameter of 5 mm. I punched it with.
- This was inoculated into 40 mL of SMY liquid medium (containing 1% sucrose, 1% malt extract, and 0.4% yeast extract) in an lOOmL Erlenmeyer flask. After stationary culture for 7 days, the mycelium was ground with a magnetic stirrer and a stirrer to prepare a mycelium suspension.
- the mycelium for preparing protoplasts was collected by filtration through a nylon mesh (pore size 10 X 10 m).
- the obtained mycelium 200 mg was mixed with 2% Novozym—234 (Novo Industry AZS), 0.5% Zymolyase—20T (Seikagaku Corporation), 0.2% Chitinase (Sigma Chemical Co., ) And 0.5 molZL of mannitol dissolved in 0.05 mol / L maleate buffer (pH 6.0), shaken for 1 hour at a shaking width of 40 mm, shaking frequency of 70 Zmin, shaking temperature of 28 ° C. Then, the enzyme treatment (protoplastization treatment) was performed.
- the treatment solution was filtered through Miracloth (Calbiochem corporation), and the filtrate was centrifuged at 1100 g x 10 minutes.
- the obtained precipitate (protoplast) was washed three times by repeating the centrifugation operation using 0.05 mol ZL maleic acid buffer (PH 6.0, containing an osmotic pressure regulator) to obtain a purified protoplast.
- This purified protoplast was cultured on a multi-layered agar medium, and mononuclear hyphae were screened from the regenerated colonies to obtain a hybrid strain (parent strain B1).
- test method is as follows.
- Each mycelium was cultured on SMY agar medium for 7 days, and the obtained mycelium was punched with a cork borer having an inner diameter of 5 mm. This was inoculated onto 25 mL of fresh SM Y agar medium dispensed into a petri dish having an inner diameter of 90 mm.
- the mycelial density is evaluated by “High: + + + +”, “Normal: + + +”, “Low: + +”, and “Very low: +”.
- the mycelium was inoculated into SMY liquid medium containing ImM veratric acid and cultured for 13 days at a culture temperature of 26 ⁇ 2 ° C and a relative humidity of 80% or higher.
- the laccase activity (n katZL) of the filtrate was measured using a spectrophotometer by a conventional method (see Journal of the Wood Society, 40 (1), 107-110 (1994), etc.).
- Table 1 shows the test results.
- Parent strain A1 and parent strain B1 are mononuclear mycelia obtained as described above.
- Parent strain A2 and parent strain B2 are binuclear mycelia, and subculture the protozoa that had been isolated from power bracken fruit body A and power bracken fruit body B before creating parent strain A1 and parent strain B1, respectively. It is obtained.
- Rikiwaratake I and Rikiwaratake II are isolates (binuclear hyphae) obtained from wild-strength Waratatake mushrooms collected in Takasaki, Gunma Prefecture and Okutama-cho, Tokyo, respectively.
- test strain 1 is superior to parent strains and conventional varieties with higher hyphal growth and physiological functions. But it was very good.
- Test strain 1 85.6 ⁇ 3.1 ++++ 351 ⁇ 21 1 89.7
- Test strain 3 85.8 ⁇ 2.1 +++ 297 ⁇ 18 1 55.4
- 40g of fine powder of dried oyster mushrooms was mixed with 750ml of distilled water and extracted with stirring at 90 ° C for 1-2 hours. Thereafter, the mixture was centrifuged (3000 rpm, 15 minutes), and the collected supernatant was concentrated (about 10 times) with an evaporator. The concentrated solution was frozen at ⁇ 80 ° C. and freeze-dried to recover 3.5 g of a power extract of hot straw mushroom hot water, which was used as a sample for testing.
- Human blood was centrifuged at 1 lOOrpm for 20 minutes at room temperature to collect platelet-rich plasma (PRP), and then centrifuged at 3000 rpm for 5 minutes to collect platelet poor plasma (PPP).
- PRP platelet-rich plasma
- PPP platelet poor plasma
- the induced aggregation was measured using an aggregometer (manufactured by MCM Hematracer MCM Medical Co., Ltd.). By comparing the maximum aggregation rate of the test sample (the maximum value obtained from the aggregation curve of the test sample with a PPP value of 100) with the maximum aggregation rate of the control, The inhibitory action was evaluated.
- DMEM medium Dulbecco's modified Eagle's medium
- 10% ushi fetal serum 10% ushi fetal serum
- test medium To the test medium was added power bracken hot water extract extract powder (dissolved in an appropriate amount of DMSO), DMS o (negative control), or hyidocortisone (positive control). Each 0.01% final concentration of force Waratake hot water extract powder and DMSO (dry weight), the final concentration of Nono id port cortisone was 10 _ 7 M.
- tumor necrosis factor TNF-a (IngZml), which is known to promote chemokine gene expression, was added and cultured at 37 ° C for 6 hours.
- RNA was isolated from the cells according to a conventional method, cDNA was synthesized, and the expression level of IL 8 gene was measured by quantitative PCR (TaqMan PCR).
- GAPDH Denriceraldehyde 3-phosphate dehydrogenase
- Human skin fibroblasts can be obtained as a commercial product, for example, from Kurashiki Spinning Co., Ltd.
- human fibroblasts are cultured according to a conventional method for culturing animal cells.
- DMEM medium containing 10% ushi fetal serum is particularly preferred.
- Salmonella TA98 solution His + back mutation, supplemented with DMSO to 5-7% concentration, 7% in the chart below
- DMSO fetal sulfate
- Antimutagenicity (%) [(NC—NB) — (NS—NB)] Z (NC—NB) X 100
- NC Number of colonies when DMSO is obtained instead of sample solution (control)
- NB Number of colonies when DMSO is obtained instead of sample solution and mutagen (blank)
- test strain 1 has a very high activity regardless of platelet aggregation inhibitory activity, chemokine (IL 8) gene expression level inhibitory activity, and antimutagenic activity. Compared with other test strains, parent strains and conventional varieties.
- arachidonic acid and its metabolites leukotrienes, thromboxane and prostaglandins are involved in the development of various inflammatory and allergic skin diseases and rough skin.
- psoriasis which is an inflammatory abnormal keratotic disease
- arachidonic acid metabolites are high in the affected epidermis
- allergic skin diseases such as atopic dermatitis and contact skin
- arachidonic acid metabolites are also involved in inflammation and eczema.
- prostaglandins and thromboxanes aggregate platelets, compounds that also inhibit the synthesis of arachidonic acid power into prostaglandins and thromboxanes have an antithrombotic effect, that is, an improvement in blood flow. It is considered a thing.
- chemokines that control the migration and activation of inflammatory cells are strongly involved in the development of various inflammatory and allergic skin diseases.
- Chemokine is a generic name for basic proteins with a molecular weight of 8 to: LOkDa heparin-binding, and acts mainly on the migration and activation of various cells involved in inflammation in the living body. Play an important role It is considered a thing.
- inflammatory cells such as neutrophils and eosinophils migrate and become active at the site of inflammation such as contact dermatitis and atopic dermatitis, and are involved in amplification of inflammation generation.
- chemokines such as interleukin-8 (IL-8, interleukin-8) are produced from cells such as skin keratinocytes and fibroblasts.
- psoriasis which is an inflammatory abnormal keratotic disease, is known to have a large amount of IL 8 in the affected skin area.
- chemokines such as IL 8 are considered effective in improving or preventing these inflammatory or allergic disease symptoms.
- test strain 1 In order to investigate the safety of the test strain 1 with the highest growth potential, physiological functionality, and activity based on the above tests, an acute toxicity test and a subacute toxicity test were conducted.
- OECD Guidelines for Toxicity of Chemical Substances (1987) As a sample, the above-described power oyster hot water extract extract powder was used. Single doses were administered at doses of 200, 400, 600, 800 and 2000 mgZkg. Individual observation was performed until 24 days after administration.
- the doses were 200, 400, 600, 800, and 2000 mgZkg ', and were administered once a day for 28 days.
- Test strain 1 has been confirmed to be highly safe in acute toxicity studies, even in the case of an oral dose of 2000 mgZkg! .
- test strain 1 was confirmed to be extremely safe, with no degeneration in the dead individuals and organs even in the oral dose of 2000 mgZkg!
- Hyde mouth cortisone has a strong action as a chemokine gene expression inhibitor! Even in the above chemokine gene expression suppression test, the gene expression level of Hyde mouth cortisone was as high as 0.13, showing a high suppression effect. However, long-term use of steroid hormones causes side effects such as inflammation, tissue damage, and ulceration. On the other hand, although test strain 1 has a stronger inhibitory effect than Hyde Cortisone, it can be used for a long period of time because it is safe with no side effects.
- test strain 1 counterculture was performed on parent strain A, parent strain B, and conventional varieties.
- the homology of both can be easily determined by observing the presence or absence of band lines (zone lines) at the contact surfaces of the opposing fungus cells (colony). The test was conducted as follows.
- test strain 1 formed a band line for any parent strain and conventional varieties, and thus the homology with them was low, and it was considered that they were different varieties.
- FIG. 2 shows the results of the pharmacological activity test of a power oyster mushroom extract extract powder prepared by the same method as described above except that the extraction solvent was hydrous ethanol and the extraction temperature was room temperature (25 ° C.). As can be seen from Fig. 2, the highest activity was obtained with hot water extraction. The activity was also observed with hydrous ethanol extraction. In addition, hot water extraction tended to be preferable in terms of the recovery rate of the extracted extract powder.
- MEM Earl medium glucose-free, multi-layered: INC Biomedicals Inc. 87%, fetal bovine serum (inactivated for 30-60 minutes at 56 ° C: GIBCO) 10%, 2 OOmM gnoretamine A mixed solution (29.23 mg / mL: INC Biomedicals Inc.) 2% and penicillin'streptomycin (10,000 units / mL: GIBCO) 1% was used as a test medium.
- a solution prepared by diluting force extract of hot water extract (FERM BP-10633) with hot water 1000 times with PBS and sterilizing with an autoclave was used as a sample solution.
- FERM BP-10633 A solution prepared by diluting force extract of hot water extract (FERM BP-10633) with hot water 1000 times with PBS and sterilizing with an autoclave was used as a sample solution.
- a solution in which PBS alone was similarly sterilized was used as a control.
- test medium was dispensed into each 96-well plate by 180 ⁇ L, and sarcoma 180 cancer cell line 10 ⁇ L and sample solution 10 ⁇ L were added.
- the number of sarcoma 180 cancer cells was 5 ⁇ loScells / well ⁇ OO / z L). Number of cells after standing at 37 ° C, 5% CO for 48 hours
- mice Twenty-four weeks old female mice were used as test animals. From 5 weeks of age, sarcoma 180 cancer cells were transplanted into the left abdomen of mice at 1 x 10 6 cells Z0. 05 ml and administered in the following 3 groups.
- Control group Forced oral administration of water (non-administration of power bracken fruit body hot water extract extract powder)
- 500mgZkg administration group 500mgZkg per day (20mgZ0.2ml per 40g mouse) of 5 weeks-old force-waratake fruiting body hot water extract powder was orally administered daily.
- tumor growth was significantly suppressed in the test group (administered group) compared to the control group (non-administered group).
- the power oyster mushroom strain FERM BP-10633 which is effective in the present invention, has very high activity, but it can be used in combination with microalgae and other basidiomycetes for the purpose of further improving the activity. After all, it was examined.
- Chlorella pyrenoidosa 40 kg and distilled water (750 L) were placed in a chlorella extraction tank, heated to 90 ° C, and extracted for 30 minutes. Thereafter, the supernatant was collected by centrifugation to obtain a chlorella extract. Concentrate this extract to about 45 L (about 18 times) with an evaporator, add 40 kg of
- shitake extract tank 40 kg of dried shiitake (Lentinus edodes) chips and 750 L of distilled water were placed, heated to 85 ° C, and extracted for 60 minutes. Thereafter, the extract was collected while squeezing with a shitake squeezer, and the supernatant was further collected by centrifugation to obtain a shiitake extract.
- the chlorella extract 650 L and the shiitake extract 620 L were mixed and concentrated to 70 L with an evaporator. Add 27.5 kg of ⁇ -cyclodextrin and stir with a kneader to dry the drum (drum surface temperature approximately 150 ° C, reaction temperature approximately 90 ° C) until the water content is 5% or less. Dried. The chip-like dried product generated in the dried product was pulverized by a pulverizer to obtain about 40 kg of chlorella shitake extract powder.
- a drink was prepared according to the following Formulation Examples 1 and 2, and the test was performed in the same manner as described above. As a result, as shown in Table 4, all of the drinks showed very high activity in the platelet aggregation inhibitory action, chemokine gene inhibitory action, and antimutagenic action.
- Formulation example 2 72.9 0.1 8 82.1 Formulation Example 1
- Functional drink (Contain the following ingredients in 50 mL of purified water.) Power oyster mushroom (FERM BP-10633) Hot water extract extract powder 200 mg Agaritas hot water extract 10 Shiitake mycelium hot water extract 10 Meshimakob mycelium heat Water extract 20 Cordyceps mycelium hot water extract 10 Chlorella extract 200 Carotec 1.
- the test was conducted in vivo using spontaneously hypertensive rats (Spontaneously Hypertensive Rat: SHR (4 weeks old), obtained from Japan chili Sliver Co., Ltd.).
- SHR is 7-15 weeks of age and 100% of individuals develop spontaneous hypertension.
- the blood pressure averages about 171 mmHg at 10 weeks of age, and the maximum blood pressure reaches about 200 mmHg or more.
- SHR mice also show abnormal cholesterol metabolism and insulin resistance.
- the mean systolic blood pressure of the control group (non-administered group) is zero, and the mean systolic blood pressure of the test group (administered group) is plotted.
- Blood pressure began to decrease more than control at 30 minutes, and blood pressure decreased most at 4 to: LO time after administration. After that, the increase in blood pressure gradually increased, and the increase in power was slow. It took about 48 hours to reach the same blood pressure as the control.
- FIG. 5 shows changes in mean systolic blood pressure for each week in the control group (non-administration group) and the test group (administration group).
- control group blood pressure gradually increased with increasing age, and blood pressure exceeded 220 mmHg at 16 weeks of age (11 weeks of administration).
- test group the blood pressure gradually increased until 8 weeks of age (3 weeks of administration), and then remained stable in the range of 170 to 180 mmHg.
- test patients were 10 hypertensive patients (7 males and 3 females) whose systolic blood pressure was constantly around 150 to 170 mmZHg.
- the subjects had a mean systolic blood pressure of 162 mmHg and a mean diastolic blood pressure of 96 mmHg, which were almost the same for 10 days before the start of treatment.
- the subject patient was orally ingested the drink of Prescription Example 2 once a day for 70 days from the administration start date (day zero).
- the intake was 50 mL per day (200 mg as a power extract of hot straw mushroom hot water).
- blood pressure was measured immediately before administration.
- the patient's blood was collected in a blood collection tube containing a coagulation inhibitor, and the blood renin concentration and blood angiotensin I concentration were also measured based on general laboratory test methods.
- composition containing power oyster mushroom extract of the present invention lowers the blood pressure of hypertensive patients, but there is very little concern that the blood pressure will be excessively lowered even when chronically ingested.
- Fig. 7 shows changes in blood renin concentration and blood angiotensin I concentration in the subjects. As the number of administration days increased, renin concentration increased, while angiotensin I concentration decreased. This suggests that blood pressure can be controlled without excessive secretion or excessive suppression of endocrine renin, angiotensin and other hormones.
- the test patients were 30 healthy adults.
- the test patient was orally ingested the drink of Formulation Example 2 once a day for one month every day.
- the intake was 50 mL per day (200 mg as a power extract of hot oyster mushroom extract). Thereafter, intake was stopped for 2 months.
- blood of the test patient is collected into a blood collection tube containing a blood coagulation inhibitor, and T cells and NK cells based on general laboratory test methods The percentage of immune cells was measured.
- FIG. 8 shows the number of T cells (%) and the number of NK cells (%) during the administration period (1 month) and the subsequent administration suspension period (2 months).
- the number of T cells and NK cells both increased as the number of administration days increased.
- both the number of NK cells and the number of T cells began to decrease, but the decrease was slow, and it took two months to return to the level before the start of administration.
- the composition containing the oyster mushroom extract has an immunostimulatory effect, and the sustained effect is high by continuous ingestion.
- further examples of the composition containing the extract of power oyster mushroom (FERM BP-10633) of the present invention are further omitted.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Biotechnology (AREA)
- Mycology (AREA)
- Microbiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Zoology (AREA)
- Medicinal Chemistry (AREA)
- Botany (AREA)
- Wood Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Food Science & Technology (AREA)
- Nutrition Science (AREA)
- Genetics & Genomics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biochemistry (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Epidemiology (AREA)
- General Engineering & Computer Science (AREA)
- Immunology (AREA)
- Birds (AREA)
- Virology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Biomedical Technology (AREA)
- Dermatology (AREA)
- Medical Informatics (AREA)
- Alternative & Traditional Medicine (AREA)
- Marine Sciences & Fisheries (AREA)
- Hematology (AREA)
- Cardiology (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/996,961 US7790175B2 (en) | 2005-07-28 | 2006-07-28 | Strain of turkey tail mushroom, extract from the same, and use of the same |
| CN2006800276532A CN101233225B (zh) | 2005-07-28 | 2006-07-28 | 云芝菌株、其提取物及其应用 |
| JP2006529405A JP4044599B2 (ja) | 2005-07-28 | 2006-07-28 | カワラタケ株及びその抽出物及びその利用 |
| AU2006273203A AU2006273203B2 (en) | 2005-07-28 | 2006-07-28 | Strain of turkey tail mushroom, extract from the same, and use of the same |
| EP06781888.0A EP1918363B1 (en) | 2005-07-28 | 2006-07-28 | Strain of turkey tail mushroom, extract from the same, and use of the same |
| IL189030A IL189030A (en) | 2005-07-28 | 2008-01-24 | A strain of turkey tail fungus, its extraction and use |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005-218554 | 2005-07-28 | ||
| JP2005218554 | 2005-07-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007013588A1 true WO2007013588A1 (ja) | 2007-02-01 |
Family
ID=37683475
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2006/314978 WO2007013588A1 (ja) | 2005-07-28 | 2006-07-28 | カワラタケ株及びその抽出物及びその利用 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US7790175B2 (ja) |
| EP (1) | EP1918363B1 (ja) |
| JP (1) | JP4044599B2 (ja) |
| CN (1) | CN101233225B (ja) |
| AU (1) | AU2006273203B2 (ja) |
| IL (1) | IL189030A (ja) |
| TW (1) | TWI386485B (ja) |
| WO (1) | WO2007013588A1 (ja) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009085472A1 (en) | 2007-12-19 | 2009-07-09 | Elc Management Llc | Compositions and methods for treating skin with extract from trametes |
| JP2010030955A (ja) * | 2008-07-29 | 2010-02-12 | Bhn Kk | 抗変異原性剤、その製造方法並びに利用 |
| JP5518223B1 (ja) * | 2013-02-20 | 2014-06-11 | 暁酵素産業株式会社 | 液状組成物およびその製造方法 |
| JP2015514129A (ja) * | 2012-04-11 | 2015-05-18 | バギ リサーチ リミテッド | カワラタケ抽出物、生物学的に活性な化合物を単離する方法、およびその使用 |
| WO2023135949A1 (ja) * | 2022-01-11 | 2023-07-20 | 一丸ファルコス株式会社 | プロテオグリカンを含む免疫応答調節剤 |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7829098B2 (en) | 2005-11-08 | 2010-11-09 | Vita Green Health Products Co., Ltd | Herbal powder extracts and methods of preparing and using the same |
| KR101479816B1 (ko) * | 2012-09-12 | 2015-01-06 | 강동영 | 상황버섯 자실체 추출물을 유효성분으로 함유하는 혈전증 예방 또는 치료용 약학적 조성물 및 건강 기능 식품 |
| US10092609B2 (en) | 2015-01-16 | 2018-10-09 | James A. Wieser | Process for preparing medicinal mycological preparations |
| CN107090020B (zh) * | 2017-06-09 | 2020-10-16 | 福建省平潭县水产良种实验有限公司 | 食用菌凝集素及其在抑制棕囊藻生长中的应用 |
| AU2021246519A1 (en) | 2020-04-03 | 2022-11-17 | Turtle Bear Holdings, Llc | Compositions and methods for modulating inflammatory response |
| CA3173635A1 (en) * | 2020-04-03 | 2021-10-07 | Paul E. STAMETS | Compositions and methods for modulating inflammatory response |
| CN111727809B (zh) * | 2020-07-27 | 2021-12-24 | 平泉市希才应用菌科技发展有限公司 | 一种香菇菌株及其培育方法和应用 |
| CN111727808B (zh) * | 2020-07-27 | 2022-02-01 | 平泉市希才应用菌科技发展有限公司 | 一种香菇菌株及其培育方法和应用 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6045531A (ja) * | 1983-08-11 | 1985-03-12 | Kureha Chem Ind Co Ltd | 抗高脂血症剤 |
| JPH1160495A (ja) * | 1997-08-20 | 1999-03-02 | Kureha Chem Ind Co Ltd | 免疫系未成熟動物用の疾病予防剤 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3429551A1 (de) * | 1983-08-11 | 1985-02-21 | Kureha Kagaku Kogyo K.K., Nihonbashi, Tokio/Tokyo | Pharmazeutische zubereitung, die ein glycoprotein enthaelt |
| JP2711201B2 (ja) * | 1992-10-06 | 1998-02-10 | 東洋製薬株式会社 | 連鎖球菌抗菌剤 |
| US20020176796A1 (en) * | 2000-06-20 | 2002-11-28 | Purepulse Technologies, Inc. | Inactivation of microbes in biological fluids |
| CN1364572A (zh) * | 2001-01-16 | 2002-08-21 | 杨孟君 | 纳米云芝菌制剂药物及其制备方法 |
| CN100363384C (zh) * | 2001-12-29 | 2008-01-23 | 福州绿谷生物药业技术研究所 | 草栽灵芝多糖肽及其提取方法 |
| WO2005021703A1 (ja) * | 2003-08-29 | 2005-03-10 | Yamaha Hatsudoki Kabushiki Kaisha | 微生物の培養装置、培養装置への袋の挿入方法 |
| JP4618770B2 (ja) * | 2004-02-12 | 2011-01-26 | 株式会社クレハ | 新規な血圧降下剤および食品 |
| CN1634121A (zh) * | 2004-10-15 | 2005-07-06 | 北京正大绿洲医药科技有限公司 | 灵芝多糖、云芝多糖滴丸 |
-
2006
- 2006-07-28 WO PCT/JP2006/314978 patent/WO2007013588A1/ja active Application Filing
- 2006-07-28 CN CN2006800276532A patent/CN101233225B/zh not_active Expired - Fee Related
- 2006-07-28 AU AU2006273203A patent/AU2006273203B2/en not_active Ceased
- 2006-07-28 EP EP06781888.0A patent/EP1918363B1/en not_active Not-in-force
- 2006-07-28 TW TW095127693A patent/TWI386485B/zh not_active IP Right Cessation
- 2006-07-28 US US11/996,961 patent/US7790175B2/en not_active Expired - Fee Related
- 2006-07-28 JP JP2006529405A patent/JP4044599B2/ja active Active
-
2008
- 2008-01-24 IL IL189030A patent/IL189030A/en active IP Right Grant
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6045531A (ja) * | 1983-08-11 | 1985-03-12 | Kureha Chem Ind Co Ltd | 抗高脂血症剤 |
| JPH1160495A (ja) * | 1997-08-20 | 1999-03-02 | Kureha Chem Ind Co Ltd | 免疫系未成熟動物用の疾病予防剤 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1918363A4 * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009085472A1 (en) | 2007-12-19 | 2009-07-09 | Elc Management Llc | Compositions and methods for treating skin with extract from trametes |
| JP2011507867A (ja) * | 2007-12-19 | 2011-03-10 | イーエルシー マネージメント エルエルシー | トラメテス属の抽出物を用いた皮膚処置用組成物および方法 |
| AU2008343599B2 (en) * | 2007-12-19 | 2013-04-11 | Elc Management Llc | Compositions and methods for treating skin with extract from trametes |
| US8956624B2 (en) | 2007-12-19 | 2015-02-17 | Elc Management, Llc | Compositions and methods for treating skin with extract from Trametes |
| JP2010030955A (ja) * | 2008-07-29 | 2010-02-12 | Bhn Kk | 抗変異原性剤、その製造方法並びに利用 |
| JP2015514129A (ja) * | 2012-04-11 | 2015-05-18 | バギ リサーチ リミテッド | カワラタケ抽出物、生物学的に活性な化合物を単離する方法、およびその使用 |
| JP5518223B1 (ja) * | 2013-02-20 | 2014-06-11 | 暁酵素産業株式会社 | 液状組成物およびその製造方法 |
| JP2014159387A (ja) * | 2013-02-20 | 2014-09-04 | Akatsuki Koso Sangyo Kk | 液状組成物およびその製造方法 |
| WO2023135949A1 (ja) * | 2022-01-11 | 2023-07-20 | 一丸ファルコス株式会社 | プロテオグリカンを含む免疫応答調節剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| US7790175B2 (en) | 2010-09-07 |
| EP1918363A1 (en) | 2008-05-07 |
| AU2006273203A1 (en) | 2007-02-01 |
| AU2006273203B2 (en) | 2011-07-28 |
| JP4044599B2 (ja) | 2008-02-06 |
| CN101233225A (zh) | 2008-07-30 |
| IL189030A0 (en) | 2008-08-07 |
| JPWO2007013588A1 (ja) | 2009-02-12 |
| CN101233225B (zh) | 2012-03-14 |
| EP1918363A4 (en) | 2011-02-02 |
| EP1918363B1 (en) | 2016-09-28 |
| TWI386485B (zh) | 2013-02-21 |
| US20100047269A1 (en) | 2010-02-25 |
| TW200736389A (en) | 2007-10-01 |
| IL189030A (en) | 2011-12-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4044599B2 (ja) | カワラタケ株及びその抽出物及びその利用 | |
| EP2474237B1 (en) | Equol-containing fermentation product of soybean embryonic axis, and method for production thereof | |
| KR100962587B1 (ko) | 천연 식물소재 및 한약재의 발효방법, 상기 방법에 의해서제조된 발효물 및 이를 함유하는 약학 조성물, 화장품조성물 및 식품 조성물 | |
| KR100988072B1 (ko) | 황칠나무 발효물 및 그것을 포함하는 약학 조성물 | |
| JP6684966B2 (ja) | 新規なラクトバチルス・サケイ及びそれを含む組成物 | |
| NZ586941A (en) | Fermentation with Pantoea agglomerans | |
| KR101914344B1 (ko) | 발효보리, 발효배즙, 발효콩 및 발효석류 추출물을 유효성분으로 하는 피부 보습력과 주름 개선 효능을 가지는 화장료 조성물 | |
| KR100445685B1 (ko) | 버섯 분말 또는 버섯 추출물을 포함하는 유산균배지조성물, 상기 배지조성물을 이용한 버섯 유산발효 조성물 및 상기 조성물의 제조방법 | |
| KR101368293B1 (ko) | 참다래 과실 발효물을 이용한 항염증제 조성물 및 피부 미백제 조성물 | |
| KR101333130B1 (ko) | 발모 촉진 또는 탈모 방지용 조성물 | |
| KR102247810B1 (ko) | 락토바실러스 속 sdcm 1003 및 sdcm 1105의 혼합 균주, 그 배양액, 또는 이의 추출물을 유효성분으로 포함하는 조성물 | |
| KR101367961B1 (ko) | 아토피성 피부염 개선제 조성물 | |
| KR20180042936A (ko) | 치마버섯 분리 균사체 배양액을 유효성분으로 함유하는 숙취해소 및 간질환의 예방 및 치료용 약학조성물 | |
| KR20180098888A (ko) | 땅콩새싹 추출물을 유효성분으로 포함하는 면역력 증강용 조성물 | |
| KR20110044966A (ko) | 항산화 조성물 | |
| KR101999374B1 (ko) | 감 발효물을 이용한 아토피성 피부염 개선용 조성물 | |
| KR20100101257A (ko) | 자외선 차단, 항염증, 항산화 및 항노화 조성물 | |
| KR100697655B1 (ko) | 미생물 발효에 의해 안전성이 증진된 발효 구아바 조성물 | |
| KR101894874B1 (ko) | 스트렙토마이세스 프라질리스를 이용한 항염증 조성물 | |
| CN112423770A (zh) | 包含酵母提取物的改善糖尿病或增强抗氧化活性的组合物及制备酵母提取物的方法 | |
| KR102244812B1 (ko) | 락토바실러스 플란타룸 아종 플란타룸 sdcm 1002 및 사카로마에세스 세르비지에 sdcm 3017의 혼합 균주, 그 배양액, 또는 이의 추출물을 유효성분으로 포함하는 조성물 | |
| KR20230030552A (ko) | 더위지기 추출물을 포함하는 면역증강용 조성물 | |
| KR101866850B1 (ko) | 세리포리아 락세라타 균사체 배양액을 유효성분으로 함유하는 항염증용 조성물 | |
| KR20180053278A (ko) | 감 발효물을 이용한 아토피성 피부염 개선용 조성물 | |
| KR20150017206A (ko) | 고체 발효에 의한 황칠 발효 대사체 및 그 제조방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200680027653.2 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006529405 Country of ref document: JP |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 189030 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11996961 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2006781888 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006781888 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006273203 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2006273203 Country of ref document: AU Date of ref document: 20060728 Kind code of ref document: A |