WO2006003494A2 - Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides - Google Patents
Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides Download PDFInfo
- Publication number
- WO2006003494A2 WO2006003494A2 PCT/IB2005/002002 IB2005002002W WO2006003494A2 WO 2006003494 A2 WO2006003494 A2 WO 2006003494A2 IB 2005002002 W IB2005002002 W IB 2005002002W WO 2006003494 A2 WO2006003494 A2 WO 2006003494A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- optionally substituted
- alkyl
- alkoxy
- haloalkyl
- heteroaryl
- Prior art date
Links
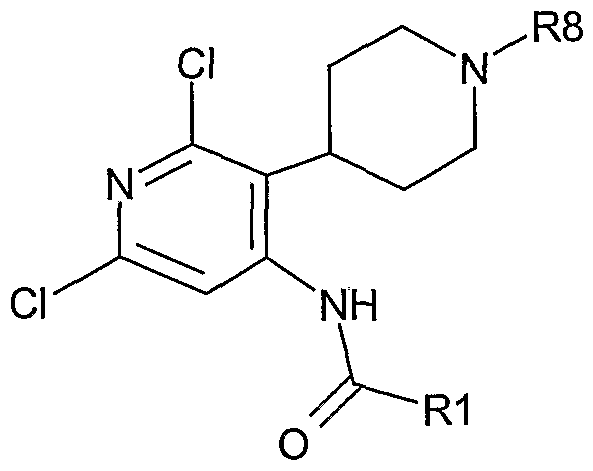
- 0 *C1=C(C2=CCN(*)CC2)C(NC(c2cc(Cl)nc(Cl)c2)=O)=C(*)**1 Chemical compound *C1=C(C2=CCN(*)CC2)C(NC(c2cc(Cl)nc(Cl)c2)=O)=C(*)**1 0.000 description 16
- BLSBQYUJENVWFW-UHFFFAOYSA-N CC(C)(C)OC(N(CC1)CC=C1S1OC(C)(C)C(C)(C)O1)=O Chemical compound CC(C)(C)OC(N(CC1)CC=C1S1OC(C)(C)C(C)(C)O1)=O BLSBQYUJENVWFW-UHFFFAOYSA-N 0.000 description 1
- GPPWROXEQAURAI-UHFFFAOYSA-N CC(C)(C)OC(N(CC1)CCC1[n]1nccc1N)=O Chemical compound CC(C)(C)OC(N(CC1)CCC1[n]1nccc1N)=O GPPWROXEQAURAI-UHFFFAOYSA-N 0.000 description 1
- MMWAHLXGFZBYQU-UHFFFAOYSA-N CC(C)(CC1)C1N Chemical compound CC(C)(CC1)C1N MMWAHLXGFZBYQU-UHFFFAOYSA-N 0.000 description 1
- QSYRYLLGMLINLH-FMIVXFBMSA-N CCCCN(C/C=C(\CC)/N)C(OC(C)(C)C)=O Chemical compound CCCCN(C/C=C(\CC)/N)C(OC(C)(C)C)=O QSYRYLLGMLINLH-FMIVXFBMSA-N 0.000 description 1
- VBUWHAJDOUIJMV-UHFFFAOYSA-N CCCNC(OC(C)(C)C)=O Chemical compound CCCNC(OC(C)(C)C)=O VBUWHAJDOUIJMV-UHFFFAOYSA-N 0.000 description 1
- KPMNBINFKBBHQJ-UHFFFAOYSA-N O=C(c1cc(Cl)ncc1)Nc(ccc(Cl)c1)c1C1=CCNCC1 Chemical compound O=C(c1cc(Cl)ncc1)Nc(ccc(Cl)c1)c1C1=CCNCC1 KPMNBINFKBBHQJ-UHFFFAOYSA-N 0.000 description 1
- VADDBZIXSXIABG-OWOJBTEDSA-N O=C(c1ccnc(Cl)c1)Nc(ccc(Cl)c1)c1C1=CCN(C/C=C/c(cc2)ccc2F)CC1 Chemical compound O=C(c1ccnc(Cl)c1)Nc(ccc(Cl)c1)c1C1=CCN(C/C=C/c(cc2)ccc2F)CC1 VADDBZIXSXIABG-OWOJBTEDSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/08—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms
- C07D211/18—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D211/34—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with substituted hydrocarbon radicals attached to ring carbon atoms with hydrocarbon radicals, substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/36—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D211/40—Oxygen atoms
- C07D211/44—Oxygen atoms attached in position 4
- C07D211/52—Oxygen atoms attached in position 4 having an aryl radical as the second substituent in position 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
Definitions
- the present invention relates to piperidine derivatives, to processes for preparing them, to insecticidal, acaricidal, molluscicidal and nematicidal compositions comprising them and to methods of using them to combat and control insect, acarine, mollusc and nematode pests.
- Piperidine derivatives with fungicidal properties are disclosed in for example in EP494717.
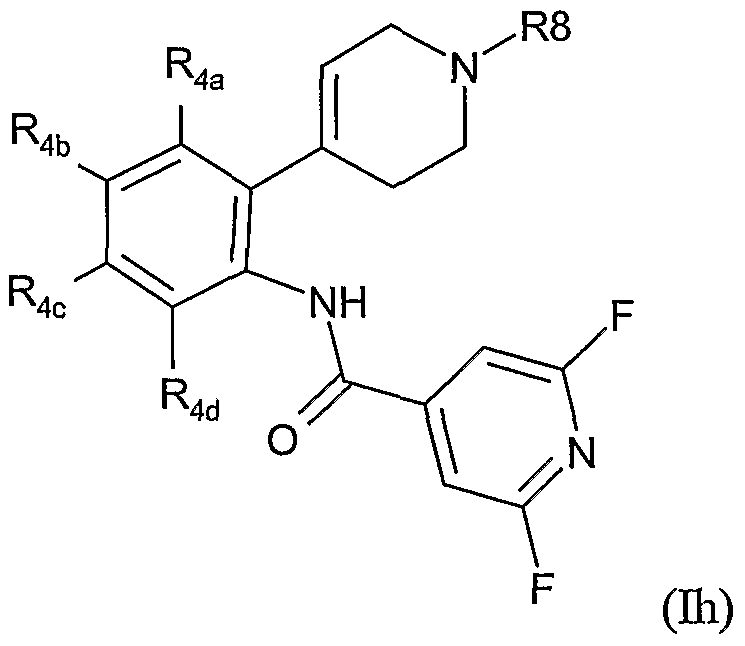
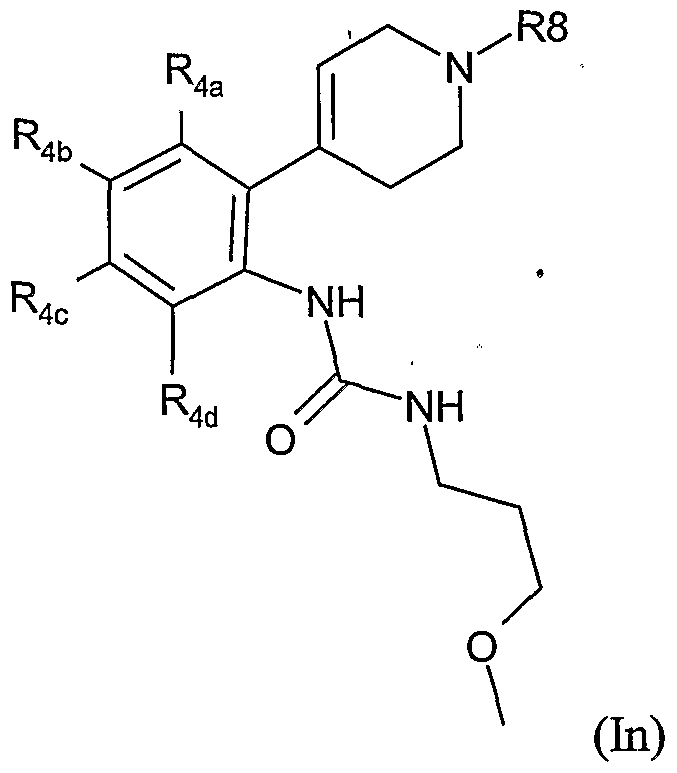
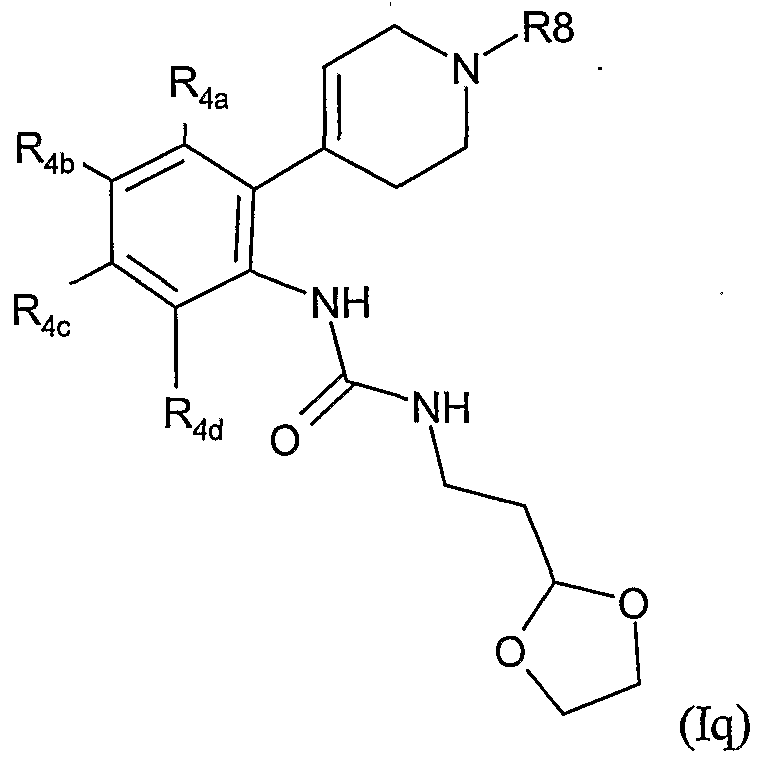
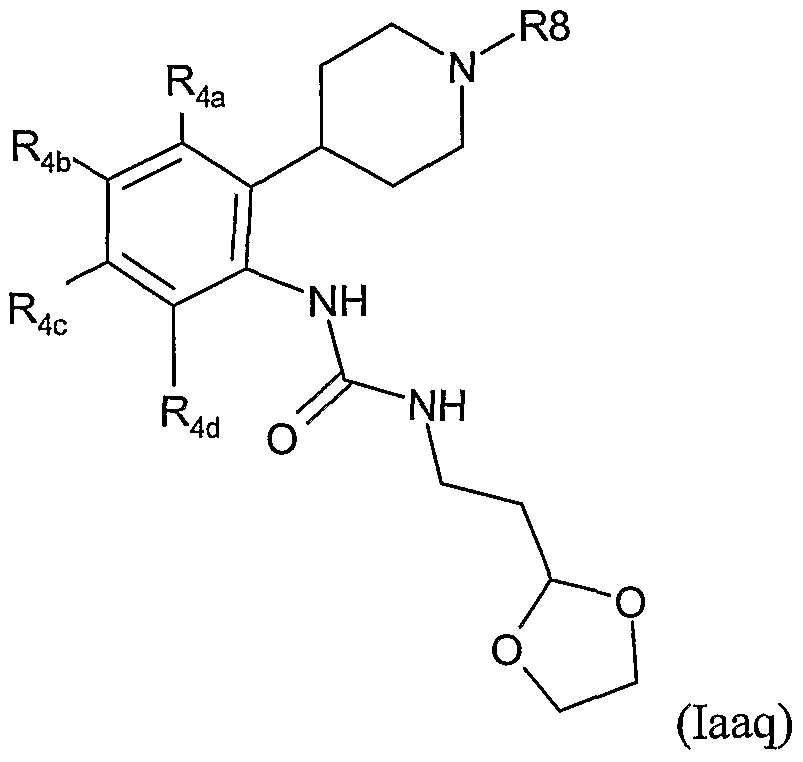
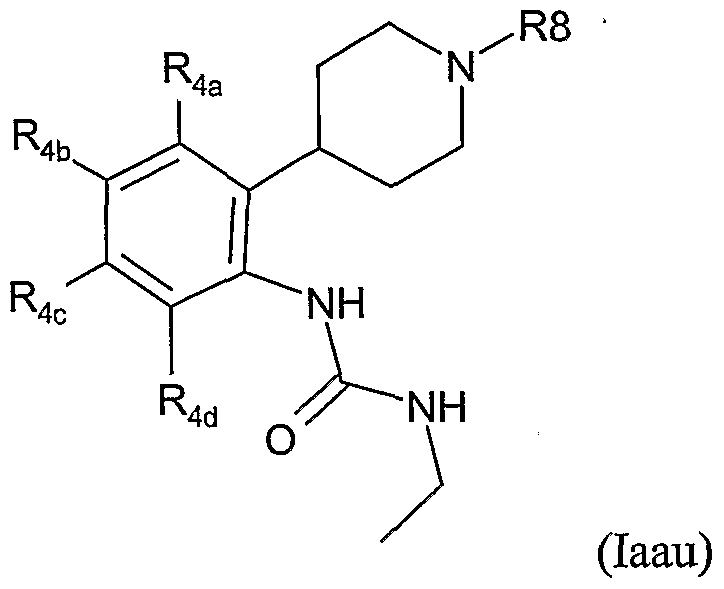
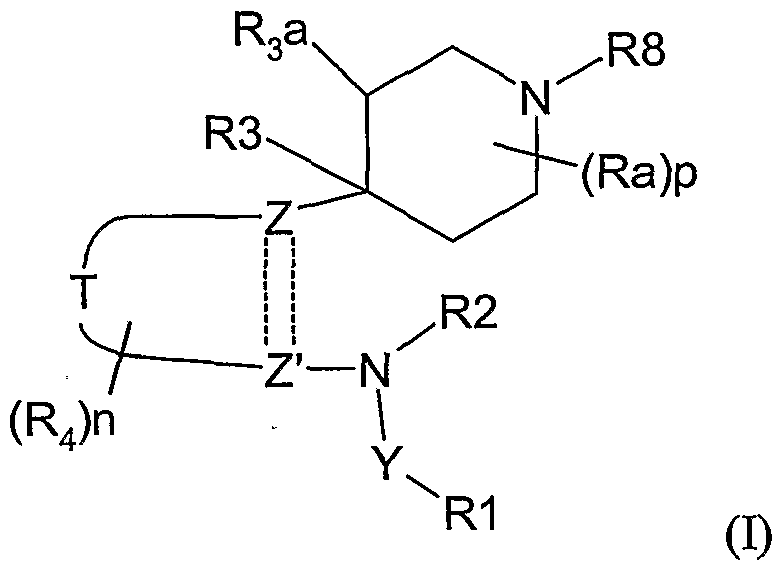
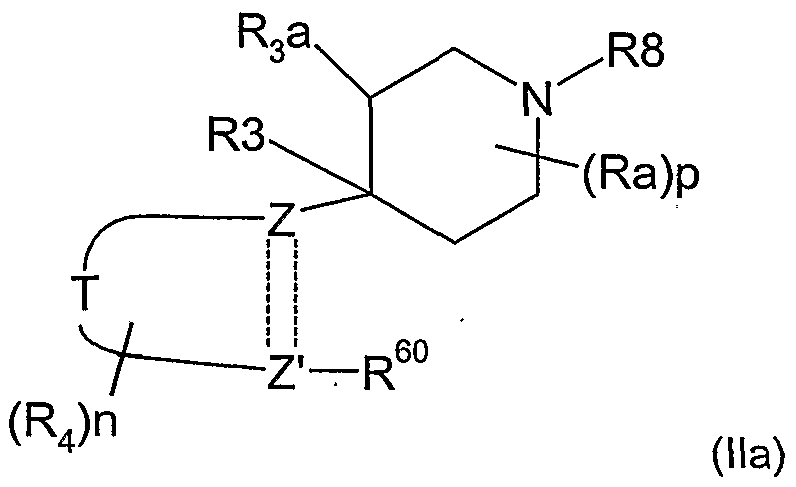
- the present invention therefore provides a method of combating and controlling insects, acarines, nematodes or molluscs which comprises applying to a pest, to a locus of a pest, or to a plant susceptible to attack by a pest an insecticidally, acaricidally, nematicidally or molluscicidally effective amount of a compound of formula (I):
- R 1 is hydrogen, optionally substituted alkyl, optionally substituted alkoxycarbonyl, optionally substituted alkylcarbonyl, aminocarbonyl, optionally substituted alkylaminocarbonyl, optionally substituted dialkylaminocarbonyl, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocyclyloxy, cyano, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted cycloalkyl, optionally substituted cycloalkenyl, formyl, optionally substituted heterocyclyl, optionally substituted alkylthio, NO or NR 13 R 14 where R 13 and R 14 are independently hydrogen, COR 15 , optionally substituted alkyl, optionally substituted aryl, optionally substituted heteroary
- R 2 is H, hydroxy, optionally substituted alkoxy or optionally substituted alkyl; or R 1 and R 2 together with the groups Y and N form a 5-or 6-membered heterocyclic ring which may optionally contain one further heteroatom selected from O, N or S and which may be optionally substituted by C 1-4 alkyl, C 1-4 haloalkyl or halogen;
- R 3 is H, OH, halogen or optionally substituted alkyl;
- R 3a is H or R 3 and R 3a together form a bond;
- each R 4 is independently halogen, nitro, cyano, optionally substituted C 1-8 alkyl, optionally substituted C 2-6 alkenyl, optionally substituted C 2-6 alkynyl, optionally substituted alkoxycarbonyl, optionally substituted alkylcarbonyl, optionally substituted alkylaminocarbonyl, optionally substituted dialkylaminocarbonyl, optionally substituted C 3-7 cycloalky
- R 8 is optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted cycloalkyl, optionally substituted aryl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted alkoxycarbonyl, optionally substituted alkylcarbonyl or optionally substituted alkenylcarbonyl; each Ra is independently halogen, hydroxy, cyano, optionally substituted C 1-8 alkyl, optionally substituted C 2-6 alkenyl, optionally substituted C 2-6 alkynyl, optionally substituted alkoxycarbonyl, optionally substituted alkylcarbonyl, optionally substituted alkylaminocarbonyl, optionally substituted dialkylaminocarbonyl, optionally substituted C 3-7 cycloalkyl, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocyclyl, optionally substituted alkoxy, optionally substituted aryloxy
- the compounds of formula (I) may exist in different geometric or optical isomers or tautomeric forms. This invention covers all such isomers and tautomers and mixtures thereof in all proportions as well as isotopic forms such as deuterated compounds.
- Each alkyl moiety either alone or as part of a larger group (such as alkoxy, alkoxycarbonyl, alkylcarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl) is a straight or branched chain and is, for example, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, iso- - A -
- alkyl groups are suitably C 1 to C 12 alkyl groups, but are preferably C 1 -C 10 , more preferably C 1 -C 8 , even more preferably preferably C 1 -C 6 and most preferably C 1 -C 4 alkyl groups.
- the optional substituents on an alkyl moiety include one or more of halogen, nitro, cyano, NCS-, C 3-7 cycloalkyl (itself optionally substituted with C 1-6 alkyl or halogen), C 5-7 cycloalkenyl (itself optionally substituted with C 1-6 alkyl or halogen), hydroxy, C 1-10 alkoxy, C 1-10 alkoxy(C 1-10 )alkoxy, tri(C 1-4 )alkylsilyl(C 1-6 )alkoxy, C 1-6 alkoxycarbonyl(C 1-10 )alkoxy, C 1-10 haloalkoxy, aryl(C 1-4 )- alkoxy (where the aryl group is optionally substituted), C 3-7 cycloalkyloxy
- Alkenyl and alkynyl moieties can be in the form of straight or branched chains, and the alkenyl moieties, where appropriate, can be of either the (E)- or ⁇ -configuration. Examples are vinyl, allyl and propargyl.
- the optional substituents on alkenyl or alkynyl include those optional substituents given above for an alkyl moiety.
- acyl is optionally substituted C 1-6 alkylcarbonyl (for example acetyl), optionally substituted C 2-6 alkenylcarbonyl, optionally substituted C 2-6 alkynylcarbonyl, optionally substituted arylcarbonyl (for example benzoyl) or optionally substituted heteroarylcarbonyl.
- Halogen is fluorine, chlorine, bromine or iodine.
- Haloalkyl groups are alkyl groups which are substituted with one or more of the same or different halogen atoms and are, for example, CF 3 , CF 2 Cl, CF 3 CH 2 or CHF 2 CH 2 .
- aryl refers to ring systems which may be mono-, bi- or tricyclic. Examples of such rings include phenyl, naphthalenyl, anthracenyl, indenyl or phenanthrenyl. A preferred aryl group is phenyl.
- heteroaryl refers to an aromatic ring system containing at least one heteroatom and consisting either of a single ring or of two or more fused rings.
- single rings will contain up to three and bicyclic systems up to four heteroatoms which will preferably be chosen from nitrogen, oxygen and sulphur.
- groups include furyl, thienyl, pyrrolyl, pyrazolyl, imidazolyl, 1,2,3-triazolyl, 1,2,4-triazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, 1,2,3-oxadiazolyl, 1,2,4-oxadiazolyl, 1,3,4-oxadiazolyl, 1,2,5-oxadiazolyl, 1,2,3-thiadiazolyl, 1,2,4-thiadiazolyl, 1,3,4-thiadiazolyl, 1,2,5-thiadiazolyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl, 1,2,3-triazinyl, 1,2,4-triazinyl, 1,3,5-tria
- heteroaromatic radicals include pyridyl, pyrimidyl, triazinyl, thienyl, furyl, oxazolyl, isoxazolyl, 2, 1 ,3 -benzoxadiazole and thiazolyl.
- heterocycle and heterocyclyl refer to a non-aromatic ring containing up to 10 atoms including one or more (preferably one or two) heteroatoms selected from O, S and N. Examples of such rings include 1,3-dioxolane, tetrahydrofuran and morpholine.
- heterocyclyl When present, the optional substituents on heterocyclyl include C 1-6 alkyl and C 1-6 haloalkyl as well as those optional substituents given above for an alkyl moiety.
- Cycloalkyl includes cyclopropyl, cyclopentyl and cyclohexyl.
- Cycloalkenyl includes cyclopentenyl and cyclohexenyl.
- cycloalkyl or cycloalkenyl include C 1-3 alkyl as well as those optional substituents given above for an alkyl moiety.
- Carbocyclic rings include aryl, cycloalkyl and cycloalkenyl groups.
- the optional substituents on aryl or heteroaryl are selected independently, from halogen, nitro, cyano, NCS-, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy- (C 1-6 )alkyl, C 2-6 alkenyl, C 2-6 haloalkenyl, C 2-6 alkynyl, C 3-7 cycloalkyl (itself optionally substituted with C 1-6 alkyl or halogen), C 5-7 cycloalkenyl (itself optionally substituted with C 1-6 alkyl or halogen), hydroxy, C 1-10 alkoxy, C 1-10 alkoxy(C 1-1 o)alkoxy, tri(C 1-4 )alkyl- silyl(C 1-6 )alkoxy, C 1-6 alkoxycarbonyl(C 1-10 )alkoxy, C 1-10 haloalkoxy, aryl(C 1-4 )alkoxy (where the aryl group is
- substituents are independently selected from halogen, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy(C 1-6 )alkyl, C 1-6 alkoxy, C 1-6 haloalkoxy, C 1-6 alkylthio, C 1-6 haloalkylthio, C 1-6 alkylsulfmyl, C 1-6 haloalkylsulfinyl, C 1-6 alkylsulfonyl, C 1-6 haloalkylsulfonyl, C 2-6 alkenyl, C 2-6 haloalkenyl, C 2-6 alkynyl, C 3-7 cycloalkyl, nitro, cyano, CO 2 H, " C 1-6 alkylcarbonyl, C 1-6 alkoxycarbonyl, R 25 R 26 N or R 27 R 28 NC(O); wherein R 25 R 26 N or R 27 R 28 NC(O); wherein R 25 R 26 N or R 27 R 28 NC(O
- Haloalkenyl groups are alkenyl groups which are substituted with one or more of the same or different halogen atoms. It is to be understood that dialkylamino substituents include those where the dialkyl groups together with the N atom to which they are attached form a five, six or seven- membered heterocyclic ring which may contain one or two further heteroatoms selected from O, N or S and which is optionally substituted by one or two independently selected (C 1-6 )alkyl groups. When heterocyclic rings are formed by joining two groups on an N atom, the resulting rings are suitably pyrrolidine, piperidine, thiomorpholine and morpholine each of which maybe substituted by one or two independently selected (C 1-6 ) alkyl groups.
- the optional substituents on an alkyl moiety include one or more of halogen, nitro, cyano, HO 2 C, C 1-10 alkoxy (itself optionally substituted by C 1-10 alkoxy), aryl(C 1-4 )alkoxy, C 1-10 alkylthio, C 1-10 alkylcarbonyl, C 1-10 alkoxycarbonyl, C 1-6 alkylaminocarbonyl, di(C 1-6 alkyl)aminocarbonyl, (C 1-6 )alkylcarbonyloxy, optionally substituted phenyl, heteroaryl, aryloxy, arylcarbonyloxy, heteroaryloxy, heterocyclyl, heterocyclyloxy, C 3-7 cycloalkyl (itself optionally substituted with (C 1-6 )alkyl or halogen), C 3- 7 cycloalkyloxy, C 5-7 cycloalkenyl, C 1-6 alkylsulfonyl, C 1-6 alkyl
- the optional substituents on alkenyl or alkynyl include one or more of halogen, aryl and C 3-7 cycloalkyl.
- a preferred optional substituent for heterocyclyl is C 1-6 alkyl.
- the optional substituents for cycloalkyl include halogen, cyano and C 1-3 alkyl.
- the optional substituents for cycloalkenyl include C 1-3 alkyl, halogen and cyano.
- Preferred groups for T, Y, Ra, R 1 , R 2 , R 3 , R 3a , R 4 and R 8 in any combination thereof are set out below.
- R 1 is hydrogen, C 1-6 alkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, C 3-7 cycloalky ⁇ C ⁇ alkyl, C 1-6 alkoxy(C 1-6 )alkyl, heteroaryl(C 1-6 )alkyl (wherein the heteroaryl group may be optionally substituted by halo, nitro, cyano, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy, C 1-6 haloalkoxy, C 1-6 alkylsulfonyl, C 1-6 alkylsulfinyl, C 1-6 alkylthio, C 1-6 alkoxycarbonyl, C 1-6 alkylcarbonylamino, arylcarbonyl, or two adjacent positions on the heteroaryl system may be cyclised to form a 5, 6 or 7 membered carbocyclic or heterocyclic ring, itself optionally substituted with
- R 1 is C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy(C 1-6 )alkyl, heteroary ⁇ Ci. 3 )alkyl (wherein the heteroaryl group may be optionally substituted by halo, nitro, cyano, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy, C 1-6 haloalkoxy, C 1-6 alkylsulfonyl, C 1-6 alkoxycarbonyl, or two adjacent positions on the heteroaryl system may be cyclised to form a 5, 6 or 7 membered carbocyclic or heterocyclic ring, itself optionally substituted with halogen), phenyl(C 1-3 )alkyl (wherein the phenyl group may be optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 ,
- R 1 is C 1-6 alkyl, C 1-6 haloalkyl, heteroaryl(C 1-3 )alkyl (wherein the heteroaryl group may be optionally substituted by halo, cyano, C 1-6 alkyl, C 1-6 haloalkyl and where the heteroaryl group is a thiazole, pyridine, pyrimidine, pyrazine or pyridazine ring), heteroaryl (optionally substituted by halo, cyano, C 1-6 alkyl, C 1-6 haloalkyl and where the heteroaryl group is a pyridine, pyrimidine, 2,1,3-benzoxadiazole, pyrazine or pyridazine ring), C 1-6 alkoxy, C 1-6 alkoxy(C 1-6 )alkyl, C 1-6 alkylamino, C 1-6 alkyoxy(C 1-6 )alkylamino or heteroaryl(C 1-3 )alkyl (where
- R 1 is pyridyl (optionally substituted by halo, C 1-3 alkyl or C 1-3 haloalkyl) especially halo-substituted pyridyl. It is preferred that : R R 2 is hydrogen, hydroxy, C 1-6 alkyl or C 1-6 haloalkyl. More preferably R 2 is hydrogen, C 1-4 alkyl or C 1-4 haloalkyl.
- R 2 is hydrogen or C 1-4 alkyl.
- R 2 is independently hydrogen or methyl.
- R 2 is hydrogen. It is preferred that R 3 is hydrogen, hydroxy, halogen, C 1-6 alkyl or C 1-6 haloalkyl.
- R 3 is hydrogen, hydroxy, halogen,C 1-4 alkyl or C 1-4 haloalkyl.
- R 3 is hydrogen or C 1-4 alkyl.
- R 3 is independently hydrogen or methyl.
- R 3 is hydrogen.
- R 3a is preferably hydrogen or R 3 and R 3a together form a double bond.
- each R 4 is independently halogen, cyano, C 1-8 alkyl, C 1-8 haloalkyl, cyanoalkyl, C 1-6 alkoxy(C 1-6 )alkyl, C 3-7 cycloalkyl(C 1-6 )alkyl, C 5-6 cycloalkenyl(C 1-6 )alkyl, C 3-6 alkenyloxy(C 1-6 )alkyl, C 3-6 alkynyloxy(C 1-6 )alkyl, aryloxy(C 1-6 )alkyl, C 1-6 carboxyalkyl, C 1-6 alkylcafbonyl(C 1-6 )alkyl, C 2-6 alkenylcarbonyl(C 1-6 )alkyl, C 2-6 alkynylcarbonyl(C 1-6 )- alkyl, C 1-6 alkoxycarbonyl(C 1-6 )alkyl, C 3-6 alkenyloxycarbonyl(C 1-6 )alkyl
- heterocyclyl group is optionally substituted by halo, nitro, cyano, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy or C 1-6 haloalkoxy
- C 2-6 alkenyl aminocarbonyl(C 2-6 )alkenyl, C 1-6 alkylaminocarbonyl(C 2-6 )alkenyl, di(C 1-6 )alkylaminocarbonyl(C 2-6 )alkenyl, phenyl(C 2-4 )- alkenyl, (wherein the phenyl group is optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , aryl, heteroaryl, amino or dialkylamino), C 2-6 alkynyl, trimethylsilyl(C 2-6 )alkynyl, aminocarbonyl(C
- each R 4 is independently halogen, cyano, C 1-8 alkyl, C 1-8 haloalkyl, C 1-8 cyanoalkyl, C 1-6 alkoxy(C 1-6 )alkyl, C 2-6 alkynyl, trimethylsilyl(C 2-6 )alkynyl, C 1-6 alkoxycarbonyl, C 3-7 cycloalkyl, C 1-3 alkyl (C 3-7 ) cycloalkyl, phenyl (optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , aryl, heteroaryl, amino or dialkylamino), heterocyclyl (optionally substituted by halo, nitro, cyano, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy or C 1-6 haloalkoxy), C 1-8 alk
- each R 4 is independently halogen, cyano, C 1-8 alkyl, C 1-8 haloalkyl, C 1-8 cyanoalkyl, C 1-6 alkoxy(C 1-6 )alkyl, C 2-6 alkynyl, heterocyclyl (optionally substituted by C 1-6 alkyl), C 1-8 alkoxy, C 1-6 haloalkoxy, phenoxy (optionally substituted by halo, cyano, C 1-3 alkyl or C 1-3 haloalkyl), heteroaryloxy (optionally substituted by halo, cyano, C 1-3 alkyl or C 1-3 haloalkyl), di(Ci -8 )alkylamino or 2 adjacent groups R 4 together with the carbon atoms to which they are attached form a 4, 5, 6 or 7 membered carbocylic or heterocyclic ring which may be optionally substituted by halogen; n is 0, 1, 2 or 3.
- each R 4 is independently fluoro, chloro, bromo, cyano, C 1-4 alkyl, C 1-4 haloalkyl, C 1-4 cyanoalkyl or C 1-3 alkoxy(C 1-3 )alkyl; n is 0, 1, 2 or 3, preferably 0, l or 2. Most preferably each R 4 is independently fluoro, chloro, bromo, C 1-4 alkyl or C 1-4 haloalkyl; n is 1, 2 or 3, preferably 1 or 2.
- R 8 is C 1-10 alkyl, C 1-10 haloalkyl, aryl(C 1-6 )alkyl (wherein the aryl group is optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , aryl, heteroaryl, amino or dialkylamino), heteroaryl(C 1-6 )alkyl (wherein the heteroaryl group is optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , aryl, heteroaryl, amino or dialkylamino), arylcarbonyl-(C 1-6 )alkyl (wherein the aryl group may be optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1- 4 haloalkyl
- R 8 is phenyl(C 1-4 )alkyl (wherein the phenyl group is optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , aryl, heteroaryl, amino or dialkylamino), heteroaryl(C 1-6 )alkyl (wherein the heteroaryl group is optionally substituted halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , aryl, heteroaryl, amino or dialkylamino), phenyl(C 2-6 )alkenyl (wherein the phenyl group is optionally substituted by halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , hal
- R 51 and R 52 are preferably hydrogen.
- R 53 and R 54 are preferably hydrogen or halogen, especially hydrogen.
- R 55 is preferably phenyl substituted with one to three substituents selected from halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 haloalkoxy, CN, NO 2 , aryl, heteroaryl, amino or dialkylamino.
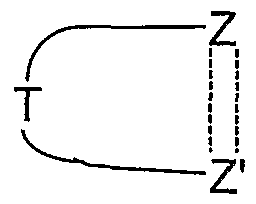
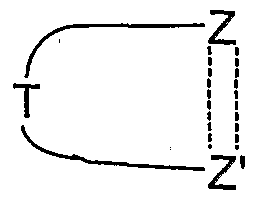
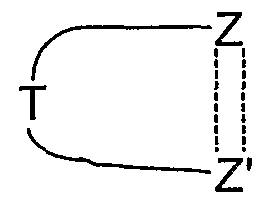
- ring members other than Z and Z' are each independently CH, S, N, NR 4 , O, or CR 4 provided that there are no more than one O or S atoms present in the ring. More preferably the ring
- benzene is a benzene, pyridine, pyrimidine, pyrazine, pyridazine, triazine, pyrrole, imidazole, quinoline, isoquinoline, thiophene, pyrazole, oxazole, thiazole, isoxazole, isothiazole, [l,2,3]triazole, [l,2,3]oxadiazole or [l,2,3]thiadiazole.
- ring is a benzene, pyridine, pyrimidine, pyrazine, pyridazine, triazine, pyrrole, imidazole, quinoline, isoquinoline, thiophene, pyrazole, oxazole, thiazole, isoxazole, isothiazole, [l,2,3]triazole, [l,2,3]oxadiazole or [l,2,3]thiadiazole.
- benzene is a benzene, pyridine, pyrimidine, pyrazine, thiophene or pyrazole ring, especially a benzene ring.
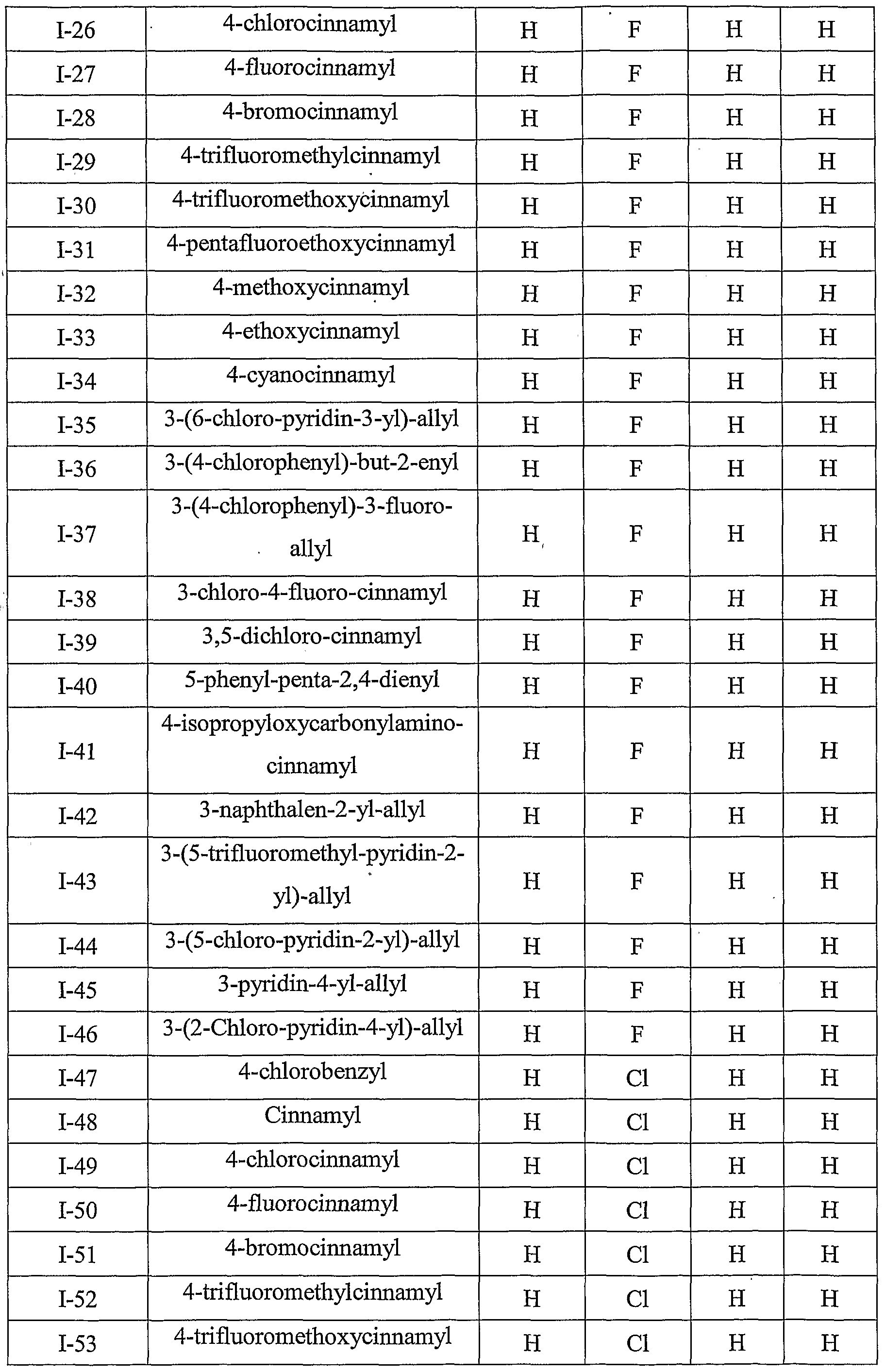
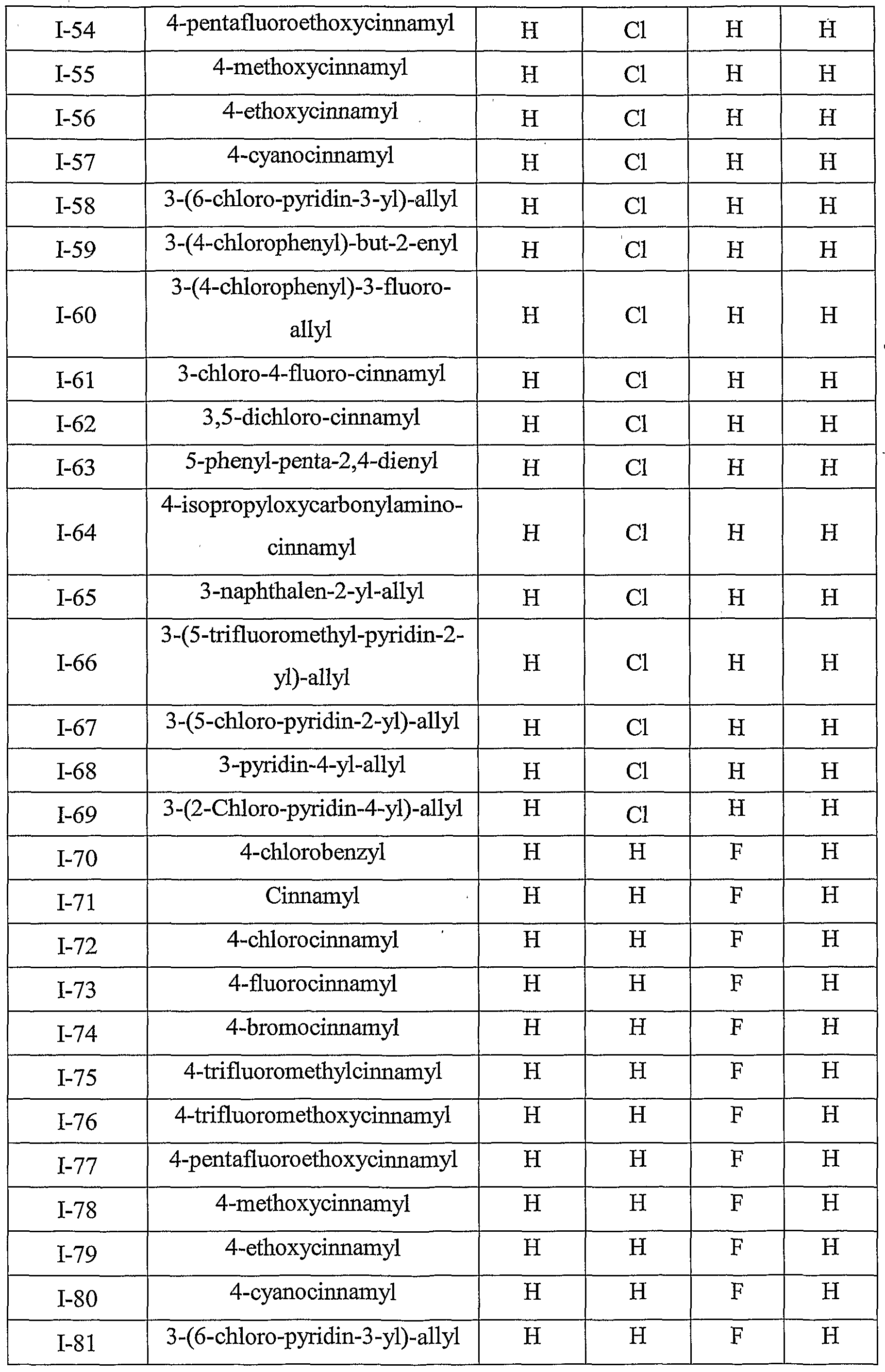
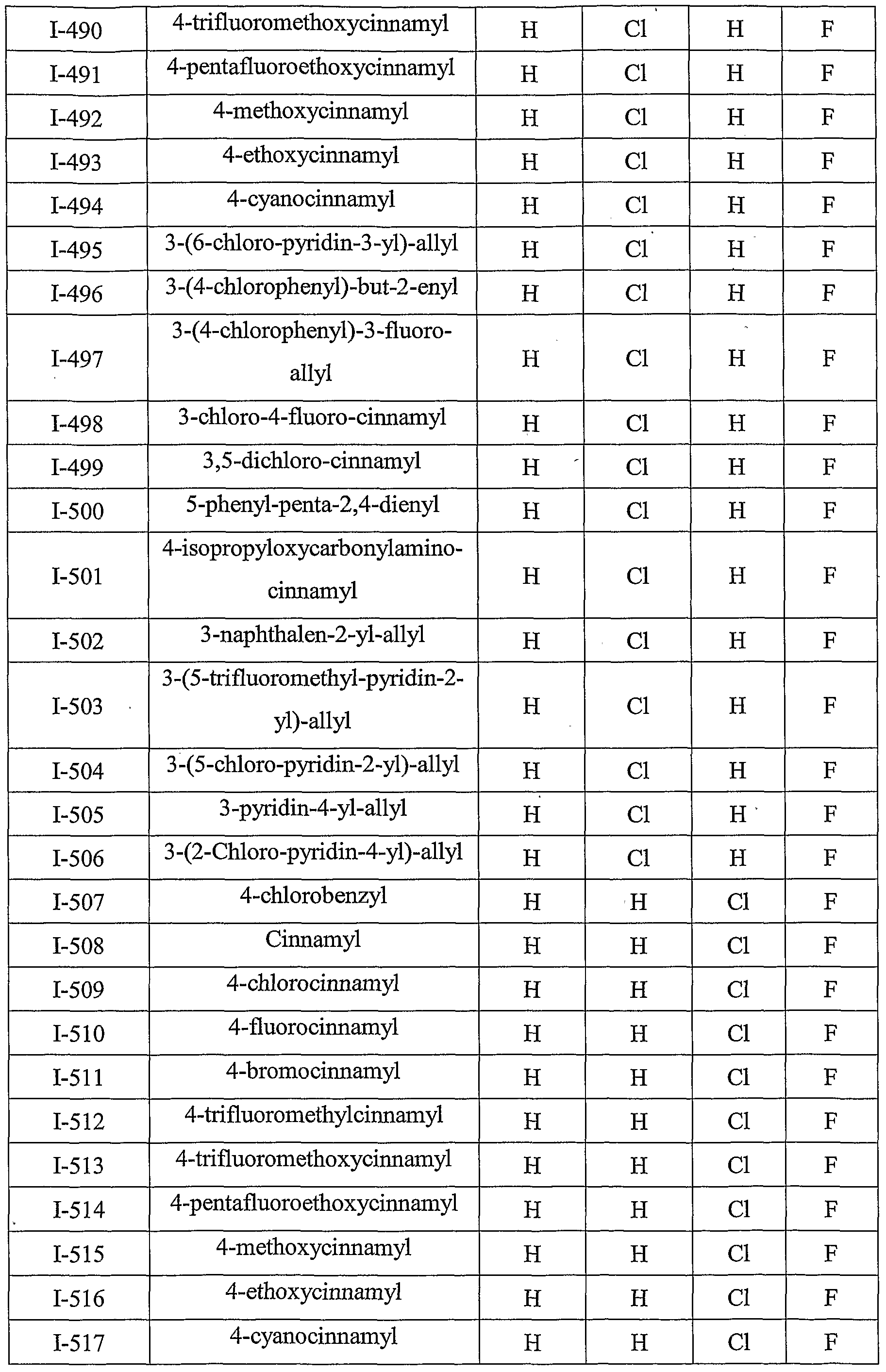
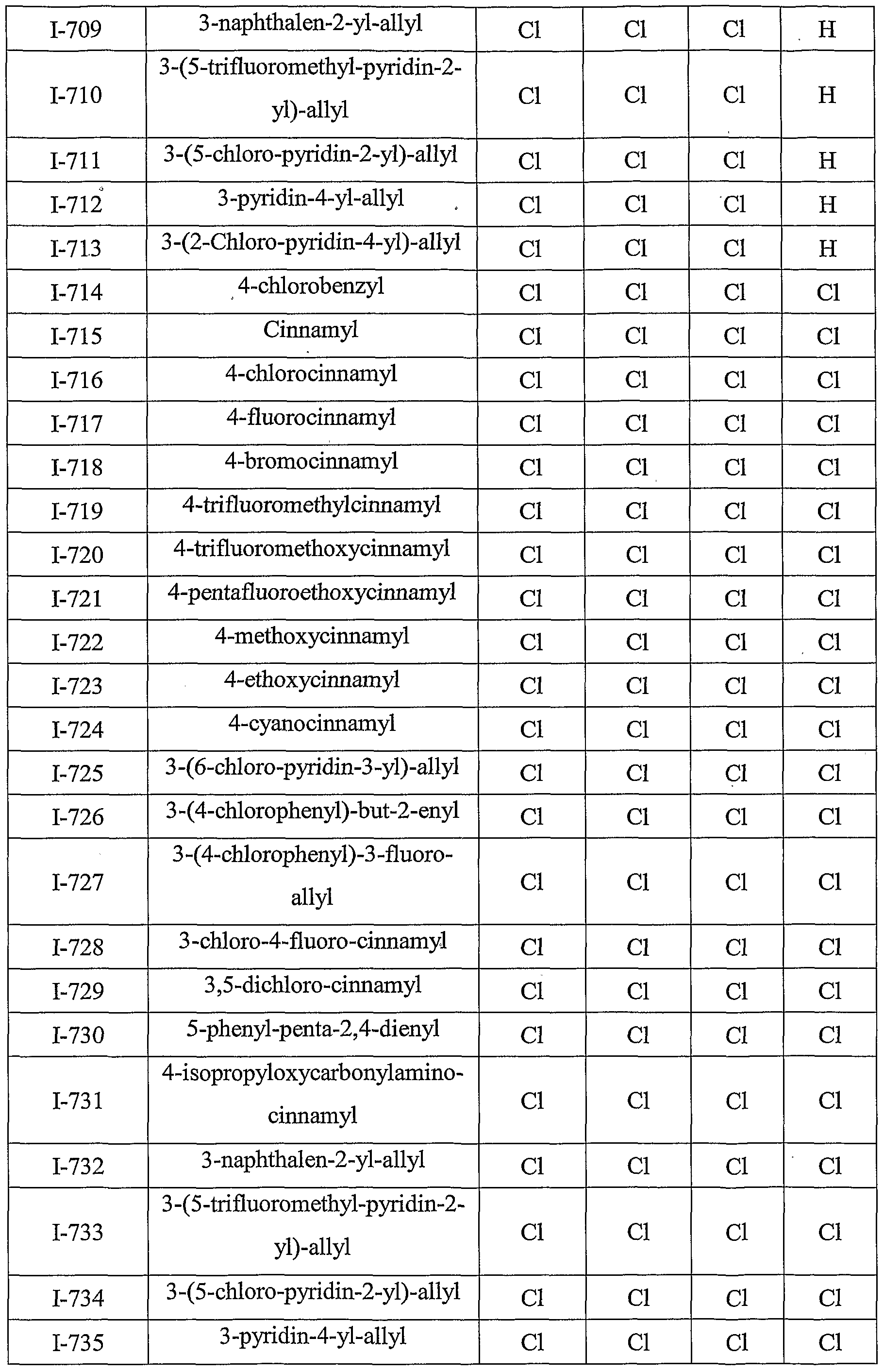
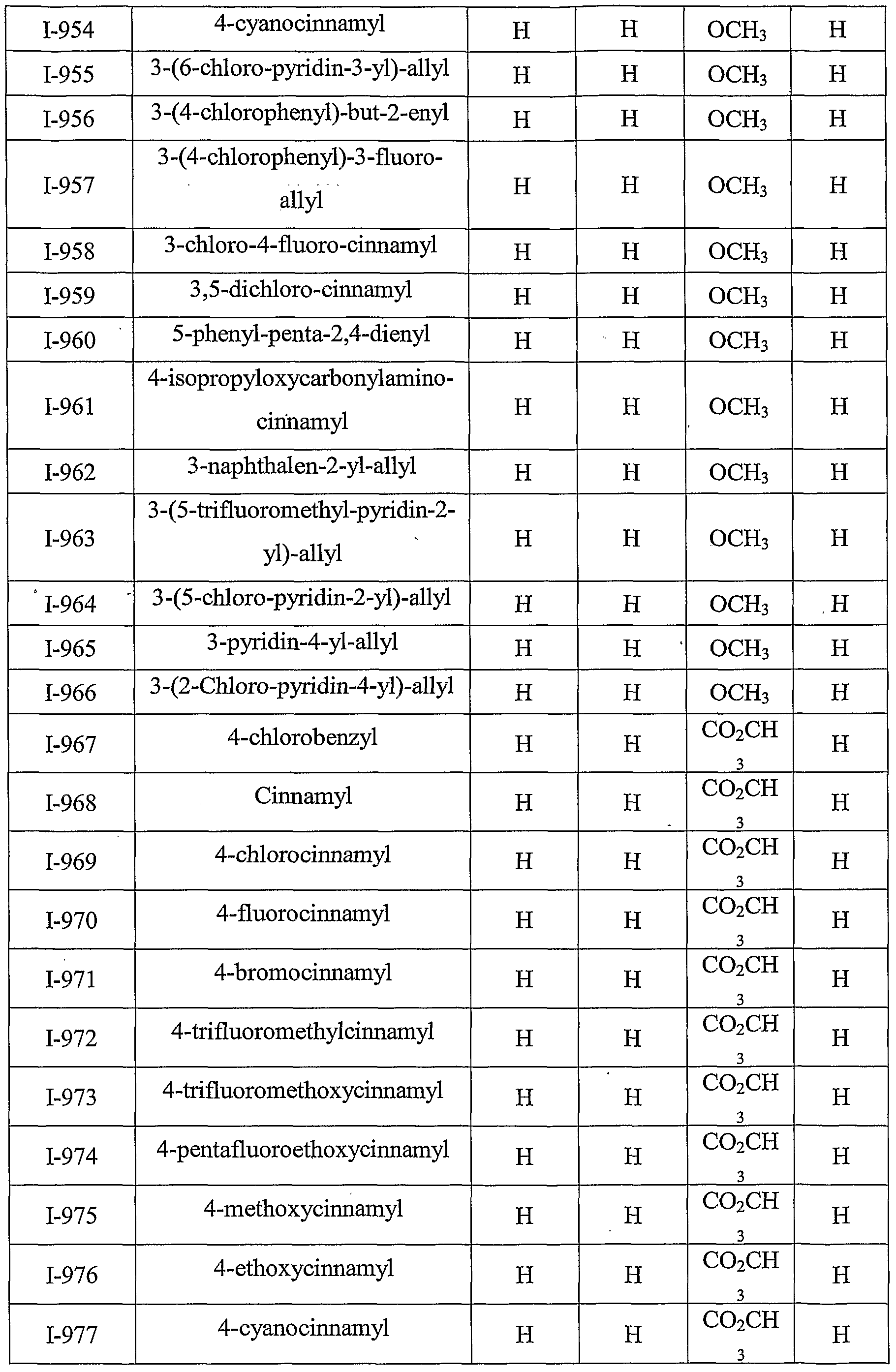
- Table III provides 1127 compounds of formula Ic wherein the values of R s , R > 4a , T Rj 4b , T RO 4 4 c C and R 4d ⁇ are given in Table 1
- Table VI provides 1127 compounds of formula If wherein the values of R s , R >4 4 a a , ⁇ Rj4b , r R>4 4 c C and R 4 w d are given in Table 1
- Table VII provides 1127 compounds of formula Ig
- Table IX provides 1127 compounds of formula Ii wherein the values of R 8 , R 4a , R 4b , R 4c and R 4d are given in Table 1.
- Table XII provides 1127 compounds of formula Il wherein the values of R ⁇ , R >4a , D R4b , - Rr,4 4 c 0 and R 4 4 d ⁇ are given in Table 1
- Table XIV provides 1127 compounds of formula hi
- Table XV provides 1127 compounds of formula Io wherein the values of R > 8 5 , - Rr>4a , r R>4b , ⁇ R>4 4 c C and R 4 4 d ⁇ are given in Table 1
- Table XVIII provides 1127 compounds of formula Lr wherein the values of R 8 , R 4a , R 4b , R 4c and R 4d are given in Table 1.
- Table XXI provides 1127 compounds of formula Iu wherein the values of R 8 , R >4a , ⁇ R>4b , ⁇ R>4 4 c C and R 4 ⁇ are given in Table 1
- Table XXVI provides 1127 compounds of formula lab
- Table XXVII provides 1127 compounds of formula lac wherein the values of R 8 , R , R 4b , T R 5 4 4 c C and R >4 4 d ⁇ are given in Table 1
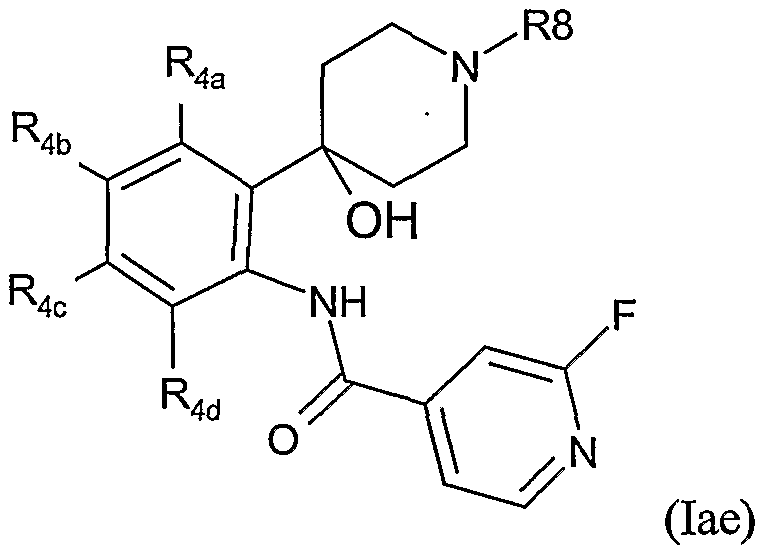
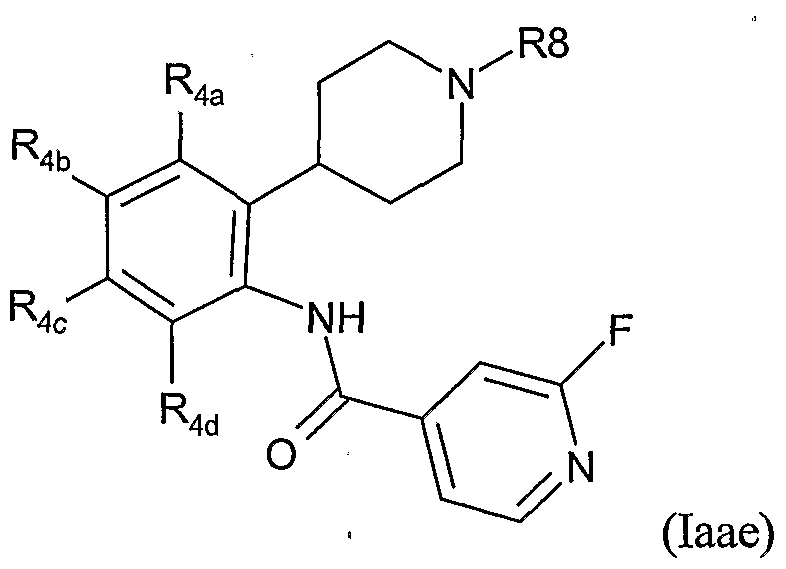
- Table XXX provides 1127 compounds of formula Iaf wherein the values of R 8 , R 4a , R 4b , R 4c and R 4d are given in Table 1
- Table XXXII provides 1127 compounds of formula Iah
- Table XXXIII provides 1127 compounds of formula Iai wherein the values of R 8 , R 4a , R 4b , R 4c and R 4d are given in Table 1.
- Table XXXTV provides 1127 compounds of formula Iaj
- Table XXXVI provides 1127 compounds of formula IaI wherein the values of R ⁇ R >4a, - Rr>4 4 b b , ⁇ R)4 4 c C and R 4 4 d ⁇ are given in Table 1
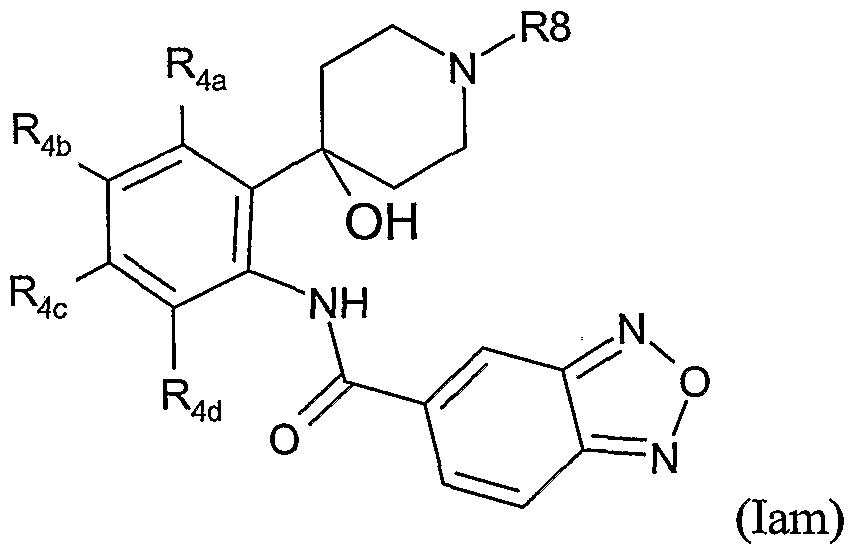
- Table XXXIX provides 1127 compounds of formula Iao wherein the values of R s , R >4a, ⁇ R>4b , T Rj 4 4 C C and R 4d are given in Table 1.
- Table XL provides 1127 compounds of formula lap
- Table XLII provides 1127 compounds of formula inquire wherein the values of R 8 , R )4a, ⁇ R > 4 4 b D , ⁇ R)4 4 c C and R 4 4 d ⁇ are given in Table 1.
- Table XLV provides 1127 compounds of formula Iau wherein the values of R 8 , R ,4a, r R>4 4 b 0 , ⁇ R>4 4 c C and ⁇ C 4d a are given in Table 1
- Table XLVI provides 1127 compounds of formula lav
- Table XLVII provides 1127 compounds of formula law
- Table XLVIII provides 1127 compounds of formula lax
- Table L provides 1127 compounds of formula Iaab
- Table LI provides 1127 compounds of formula Iaac wherein the values of R ⁇ R 4 4 a a , ⁇ R->4 4 b b , r R>4 4 o 0 and i ⁇ R>4 4 d ⁇ are given in Table 1
- Table LII provides 1127 compounds of formula Iaad
- Table LIV provides 1127 compounds of formula Iaaf wherein the values of R ⁇ R > 4a , R ⁇ 4b , R ⁇ 4 4 c C and R 4 ⁇ are given in Table 1
- Table LV provides 1127 compounds of formula Iaag
- Table LVII provides 1127 compounds of formula Iaai wherein the values of R 8 , R 4a , R 4b , R 4c and R 4d are given in Table 1.
- Table LVIII provides 1127 compounds of formula Iaaj
- Table LIX provides 1127 compounds of formula Iaak
- Table LX provides 1127 compounds of formula Iaal wherein the values of R 8 , R >4a , ⁇ R>4b , ⁇ R>4*c c and R 4 4 d are given in Table 1.
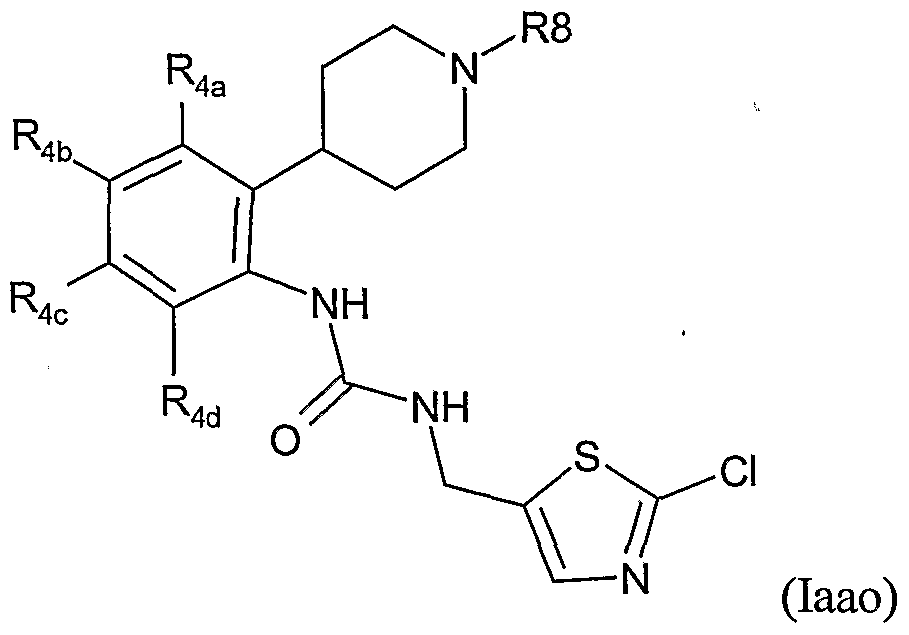
- Table LXIII provides 1127 compounds of formula Iaao wherein the values of R , R >4a , ⁇ R>4b , - R D 4 4 C C and R 4 4 d ⁇ are given in Table 1.
- Table LXV provides 1127 compounds of formula Iaaq
- Table LXVI provides 1127 compounds of formula Iaar wherein the values of R ⁇ R >4a , r R>4b , ⁇ R)4 4 c Q and R 4 ⁇ are given in Table 1.
- Table LXIX provides 1127 compounds of formula Iaau wherein the values of R 8 , R >4 w a, D R4b , ⁇ R>4 4 c C and R 4 4 d ⁇ are given in Table 1
- Table LXXII provides 1127 compounds of formula Iaax
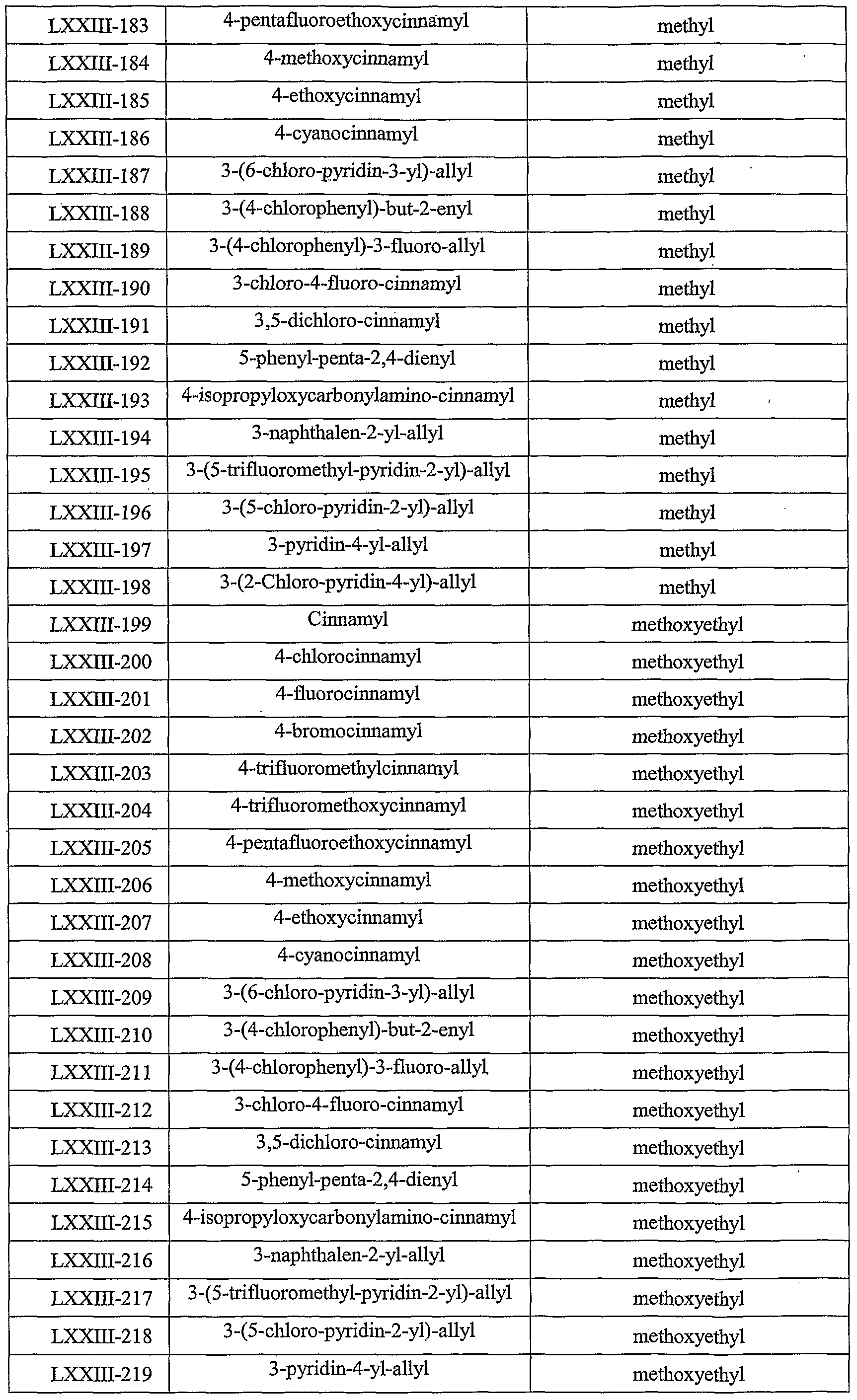
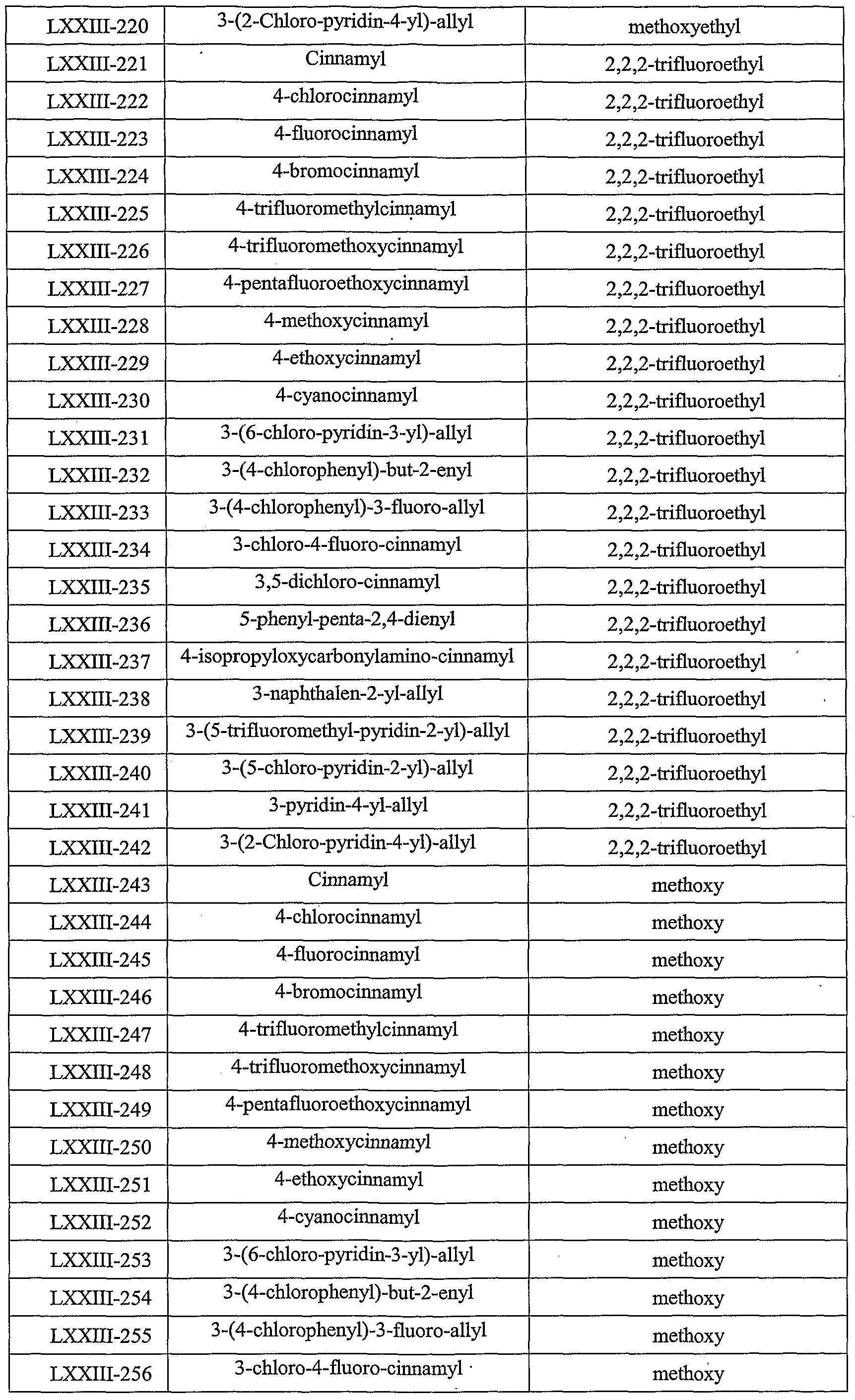
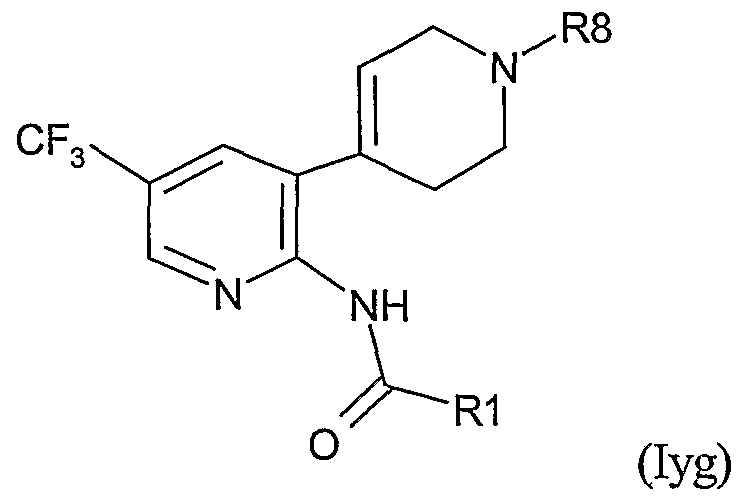
- Table LXXIII provides 506 compounds of formula Iy
- Table LXXIV provides 506 compounds of formula Iya
- Table XCIV provides 506 compounds of formula Iyu
- Table XCV provides 506 compounds of formula Iyv
- Pl is R 8 or is a suitable protective group for example a group such as BOC, benzyl or alkyl and S 1 is the group (R 4 )n.
- a compound of formula 1 maybe obtained from a compound of formula 2 by reaction with a suitable electrophilic species.
- Compounds of formula 1 where Y is a carbonyl group may be formed by the reaction of compounds of formula 2 with a carboxylic acid derivative of formula R ⁇ C(O)-Z' where Z' is chloride, hydroxy, alkoxy or acyloxy at a temperature between 0°C and 150°C optionally in an organic solvent such as dichloromethane, chloroform or 1 ,2-dichloroethane, optionally in the presence of a tertiary amine base such as triethylamine or diisopropylethylamine and optionally in the presence of a coupling agent such as dicyclohexylcarbodiimide.
- Compounds of formula 1 where Y is a group of formula S(O) m maybe formed from compounds of formula 2 by treatment with compounds of formula under similar conditions.
- compounds of formula 1 where Y is a thiocarbonyl group and R 1 is a carbon substituent may be formed by treatment of compounds of formula 1 where Y is a carbonyl group and R 1 is a carbon substituent with a suitable thionating agent such as Lawesson's reagent.
- a suitable thionating agent such as Lawesson's reagent.
- sulfur electrophiles of formula R ⁇ S(O) 1n -Cl are either known compounds or may be formed from known compounds by known methods by a person skilled in the art.
- Compounds of formula 2 may be prepared from compounds of formula 3 by cleavage of the amide bond, according to known methods by a person skilled in the art.
- Compounds of formula 3 may be obtained from compounds of formula 4 by reaction with an alkylating agent of the formula R 8 -L, where L is chloride, bromide, iodide or a sulfonate (e.g. mesylate or tosylate) or similar leaving group at a temperature of between ambient temperature and 100°C, typically 65 0 C, in an organic solvent such as dichloromethane, chloroform or 1,2-dichloroethane in the presence of a tertiary amine base such as triethylamine or diisopropylethylamine and optionally catalysed by halide salts such as sodium iodide, potassium iodide or tetrabutylammonium iodide.
- an alkylating agent of the formula R 8 -L where L is chloride, bromide, iodide or a sulfonate (e.g. mesylate or tosylate) or similar
- a compound of formula 4 may be reacted with an aldehyde of the formula R -CHO at a temperature between ambient temperature and 100°C in an organic solvent such as tetrahydrofuran or ethanol or mixtures of solvents in the presence of a reducing agent such as borane-pyridine complex, sodium borohydride, sodium (triacetoxy)borohydride, sodium cyanoborohydride or such like, to produce a compound of formula 3 where R is CH 2 -R.
- an organic solvent such as tetrahydrofuran or ethanol or mixtures of solvents
- a reducing agent such as borane-pyridine complex, sodium borohydride, sodium (triacetoxy)borohydride, sodium cyanoborohydride or such like
- Compounds of formula 4 may be prepared from compounds of formula 5 where Pl is benzyl or alkyl by a dealkylation reaction, according to known methods by a person skilled in the art. Compounds of formula 4 maybe prepared from compounds of formula 5 where Pl is
- compounds of formula 4 may be formed by the reaction of compounds of formula 6 where P 1 is BOC by treatment with HCl or H 2 SO 4 in AcOH at a temperature between 0°C and 15O 0 C optionally in an inert organic solvent.
- Compounds of formula 5 may be prepared from compounds of formula 6 where Pl is benzyl or alkyl by a H 2 O elimination reaction, according to known methods by a person skilled in the art. Most favourable is the treatment of a compound of formula 6 with cone. HCl or H 2 SO 4 in AcOH at a temperature between 0°C and 150°C.
- compounds of formula 5 maybe prepared from compounds of formula 6 by treatment with SOCl 2 , according to known methods by a person skilled in the art.
- compounds of formula 5 may be formed by the reaction of compounds of formula 9 with a carboxylic acid derivative of formula t-Bu-C(O)-Z" where Z" is chloride, hydroxy, alkoxy or acyloxy at a temperature between 0°C and 150°C optionally in an inert organic solvent.
- Compounds of formula 6 maybe prepared from compounds of formula 7 by treatment of lithiated compounds of formula 7 with a piperidinone at a temperature between - 10O 0 C and O 0 C optionally in an inert organic solvent, according to known methods by a person skilled in the art.
- compounds of formula 1 may be formed by alkylation of compounds of formula 11 as described above for compounds of formula 3.
- Compounds of formula 11 may be prepared from compounds of formula 10 where P 1 is benzyl or alkyl by a dealkylation reaction, according to known methods by a person skilled in the art.
- Compounds of formula 10 maybe prepared from compounds of formula 9 by methods described above for the conversion of compounds of formula 2 to compounds of formula 1.
- Compounds of formula 9 maybe prepared from compounds of formula 6 by a H 2 O elimination reaction, according to known methods by a person skilled in the art. Most favourable is the treatment of a compound of formula 6 with aqueous HCl or H 2 SO 4 in AcOH at a temperature between 0°C and 150°C or with a base in H 2 O and an appropriate solvent.
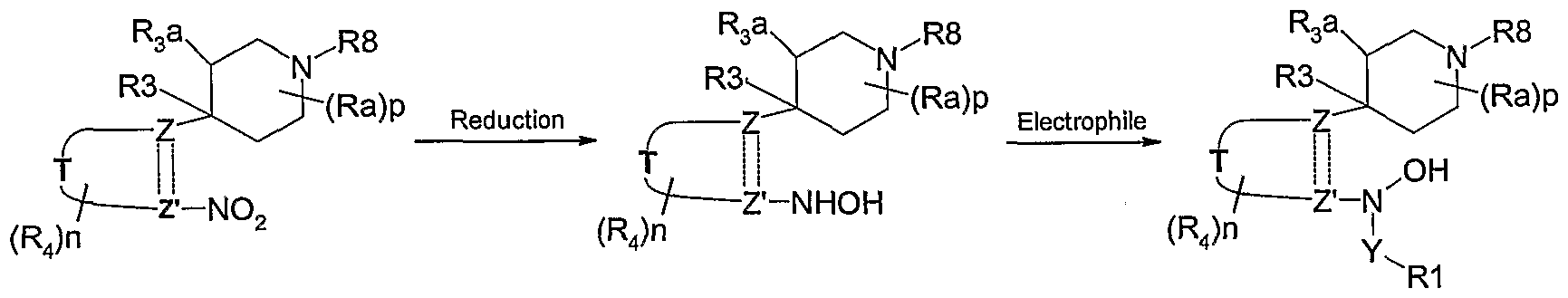
- 4-Hydroxy-piperidinyl compounds of the general formula 1 may be prepared according to the reactions of Scheme 2 using synthetic methodologies known by a person skilled in the art and as described above.
- Pl is R8 or is a suitable protective group for example a group such as BOC, benzyl or alkyl and S 1 is the group (R 4 )n.
- Piperidinyl compounds of the general formula 1 maybe prepared according to the reactions of Scheme 3 using synthetic methodologies known by a person skilled in the art and as described above.
- Pl is R8 or is a suitable protective group for example a group such as BOC, benzyl or alkyl and S 1 is the group (R 4 )n.
- Certain compounds of formula 25 are novel and as such form a further aspect of the invention.
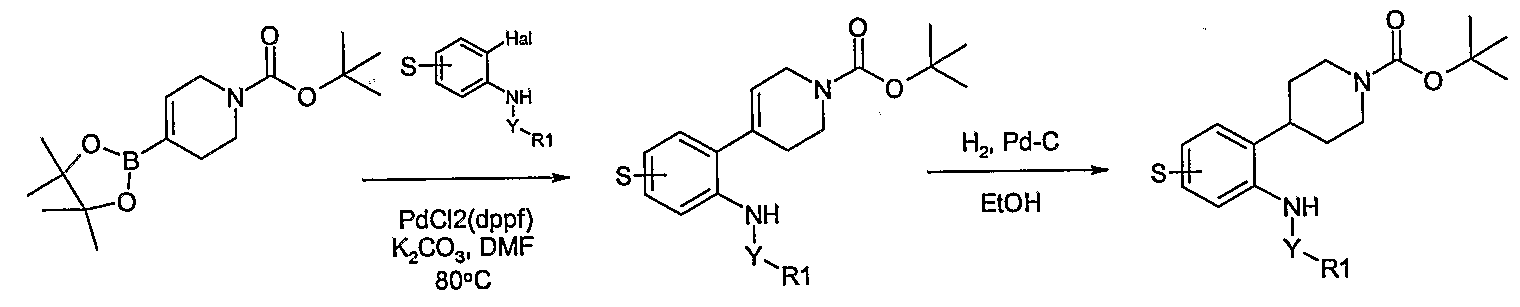
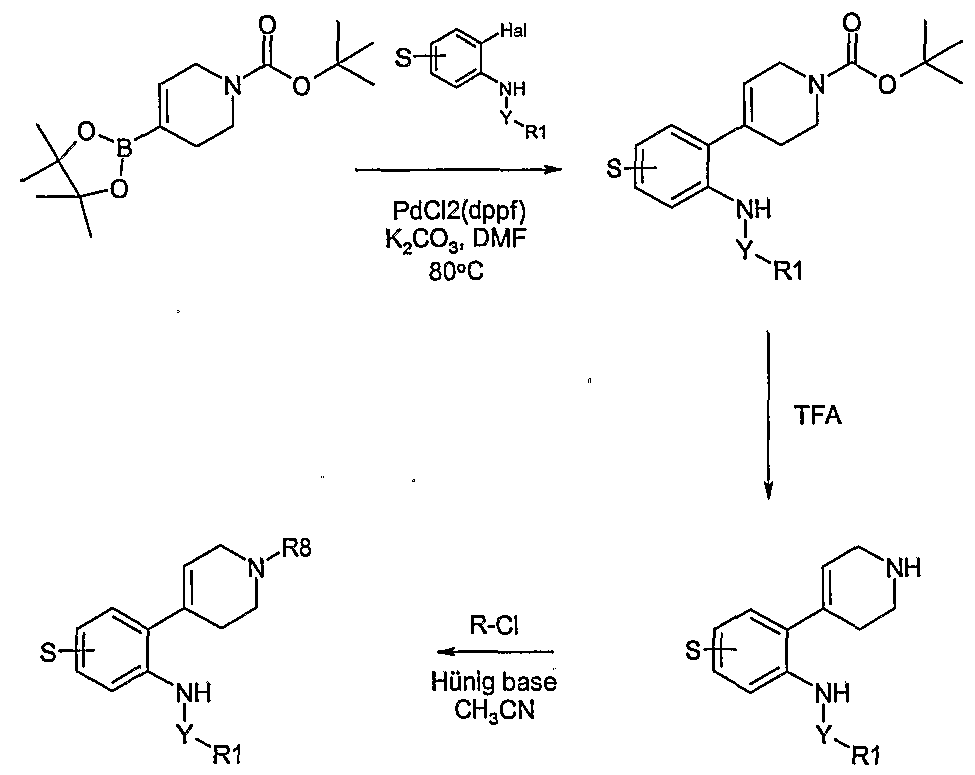
- piperidinyl-aniline derivatives of the general formula 1 may be prepared according to the reactions of Schemes 8 - 13 where S is the group (R 4 )n using synthetic methodologies known by a person skilled in the art and as described above.
- a key step in these synthetic routes is a Suzuki coupling reaction to prepare tetrahydro ⁇ yridin-4-yl-aniline derivatives.
- Other cross coupling reactions such as Stille and Negishi couplings, maybe applied as well.
- the boronate reagents may be prepared as described in the literature; for example P.R Eastwood, THL 41, 3705 (2000). Examples of coupling reactions are given in Examples 21-23 which describe the synthesis of the compounds in Tables EX23.1 - EX23.11.
- the compounds of formula (I) can be used to combat and control infestations of insect pests such as Lepidoptera, Diptera, Hemiptera, Thysanoptera, Orthoptera, Dictyoptera, Coleoptera, Siphonaptera, Hymenoptera and Isoptera and also other invertebrate pests, for example, acarine, nematode and mollusc pests. Insects, acarines, nematodes and molluscs are hereinafter collectively referred to as pests.
- the pests which may be combated and controlled by the use of the, invention compounds include those pests associated with agriculture (which term includes the growing of crops for food and fibre products), horticulture and animal husbandry, companion animals, forestry and the storage of products of vegetable origin (such as fruit, grain and timber); those pests associated with the damage of man-made structures and the transmission of diseases of man and animals; and also nuisance pests (such as flies).
- pest species which may be controlled by the compounds of formula (I) include: Myzus persicae (aphid), Aphis gossypii (aphid), Aphis fabae (aphid), Lygus spp. (capsids), Dysdercus spp. (capsids), Nilaparvata lugens (planthopper), Nephotettixc incticeps (leafhopper), Nezara spp. (stinkbugs), Euschistus spp. (stinkbugs), Leptocorisa spp. (stinkbugs), Frankliniella occidentalis (thrip), Thrips spp.
- the invention therefore provides a method of combating and controlling insects, acarines, nematodes or molluscs which comprises applying an insecticidally, acaricidally, nematicidally or molluscicidally effective amount of a compound of formula (T), or a composition containing a compound of formula (I), to a pest, a locus of pest, or to a plant susceptible to attack by a pest,
- the compounds of formula (I) are preferably used against insects, acarines or nematodes.
- a compound of formula (I) is usually formulated into a composition which includes, in addition to the compound of formula (I), a suitable inert diluent or carrier and, optionally, a surface active agent (SFA).
- a suitable inert diluent or carrier and, optionally, a surface active agent (SFA).
- SFA surface active agent
- SFAs are chemicals which are able to modify the properties of an interface (for example, liquid/solid, liquid/air or liquid/liquid interfaces) by lowering the interfacial tension and thereby leading to changes in other properties (for example dispersion, • emulsification and wetting).
- all compositions (both solid and liquid formulations) comprise, by weight, 0.0001 to 95%, more preferably 1 to 85%, for example 5 to 60%, of a compound of formula (I).
- the composition is generally used for the control of pests such that a compound of formula (I) is applied at a rate of from O.lg to 10kg per hectare, preferably from Ig to 6kg per hectare, more preferably from Ig to lkg per hectare.
- a compound of formula (I) When used in a seed dressing, a compound of formula (I) is used at a rate of O.OOOlg to 1Og (for example 0.00 Ig or 0.05g), preferably 0.005g to 1Og, more preferably 0.005g to 4g, per kilogram of seed.
- the present invention provides an insecticidal, acaricidal, nematicidal or molluscicidal composition comprising an insecticidally, acaricidally, nematicidally or molluscicidally effective amount of a compound of formula (I) and a suitable carrier or diluent therefor.
- the composition is preferably an insecticidal, acaricidal, nematicidal or molluscicidal composition.
- the invention provides a method of combating and controlling pests at a locus which comprises treating the pests or the locus of the pests with an insecticidally, acaricidally, nematicidally or molluscicidally effective amount of a composition comprising a compound of formula (I).
- the compounds of formula (I) are preferably used against insects, acarines or nematodes.
- compositions can be chosen from a number of formulation types, including dustable powders (DP), soluble powders (SP), water soluble granules (SG), water dispersible granules (WG), wettable powders (WP), granules (GR) (slow or fast release), soluble concentrates (SL), oil miscible liquids (OL), ultra low volume liquids (UL), emulsifiable concentrates (EC), dispersible concentrates (DC), emulsions (both oil in water (EW) and water in oil (EO)), micro-emulsions (ME), suspension concentrates (SC), aerosols, fogging/smoke formulations, capsule suspensions (CS) and seed treatment formulations.
- DP dustable powders
- SP soluble powders
- SG water soluble granules
- WP water dispersible granules
- GR granules
- SL soluble concentrates
- OL oil miscible liquids
- UL ultra
- Dustable powders may be prepared by mixing a compound of formula (I) with one or more solid diluents (for example natural clays, kaolin, pyrophyllite, bentonite, alumina, montmorillonite, kieselguhr, chalk, diatomaceous earths, calcium phosphates, calcium and magnesium carbonates, sulphur, lime, flours, talc and other organic and inorganic solid carriers) and mechanically grinding the mixture to a fine powder.
- solid diluents for example natural clays, kaolin, pyrophyllite, bentonite, alumina, montmorillonite, kieselguhr, chalk, diatomaceous earths, calcium phosphates, calcium and magnesium carbonates, sulphur, lime, flours, talc and other organic and inorganic solid carriers
- Soluble powders may be prepared by mixing a compound of formula (I) with one or more water-soluble inorganic salts (such as sodium bicarbonate, sodium carbonate or magnesium sulphate) or one or more water-soluble organic solids (such as a polysaccharide) and, optionally, one or more wetting agents, one or more dispersing agents or a mixture of said agents to improve water dispersibility/solubility. The mixture is then ground to a fine powder. Similar compositions may also be granulated to form water soluble granules (SG).
- water-soluble inorganic salts such as sodium bicarbonate, sodium carbonate or magnesium sulphate
- water-soluble organic solids such as a polysaccharide
- WP Wettable powders
- WG Water dispersible granules
- Granules may be formed either by granulating a mixture of a compound of formula (I) and one or more powdered solid diluents or carriers, or from pre-formed blank granules by absorbing a compound of formula (I) (or a solution thereof, in a suitable agent) in a porous granular material (such as pumice, attapulgite clays, fuller's earth, kieselguhr, diatomaceous earths or ground corn cobs) or by adsorbing a compound of formula (I) (or a solution thereof, in a suitable agent) on to a hard core material (such as sands, silicates, " mineral carbonates, sulphates or phosphates) and drying if necessary.
- a hard core material such as sands, silicates, " mineral carbonates, sulphates or phosphates
- Agents which are commonly used to aid absorption or adsorption include solvents (such as aliphatic and aromatic petroleum solvents, alcohols, ethers, ketones and esters) and sticking agents (such as polyvinyl acetates, polyvinyl alcohols, dextrins, sugars and vegetable oils).
- solvents such as aliphatic and aromatic petroleum solvents, alcohols, ethers, ketones and esters
- sticking agents such as polyvinyl acetates, polyvinyl alcohols, dextrins, sugars and vegetable oils.
- One or more other additives may also be included in granules (for example an emulsifying agent, wetting agent or dispersing agent).
- DC Dispersible Concentrates
- a compound of formula (I) may be prepared by dissolving a compound of formula (I) in water or an organic solvent, such as a ketone, alcohol or glycol ether. These solutions may contain a surface active agent (for example to improve water dilution or prevent crystallisation in a spray tank).
- Emulsifiable concentrates (EC) or oil-in-water emulsions (EW) may be prepared by dissolving a compound of formula (I) in an organic solvent (optionally containing one or more wetting agents, one or more emulsifying agents or a mixture of said agents).
- Suitable organic solvents for use in ECs include aromatic hydrocarbons (such as alkylbenzenes or alkylnaphthalenes, exemplified by SOLVESSO 100, SOLVESSO 150 and SOLVESSO 200; SOLVESSO is a Registered Trade Mark), ketones (such as cyclohexanone or methylcyclohexanone) and alcohols (such as benzyl alcohol, furfuryl alcohol or butanol), N-alkylpyrrolidones (such as N-methylpyrrolidone or N-octylpyrrolidone), dimethyl amides of fatty acids (such as C 8 -C 10 fatty acid dimethylamide) and chlorinated hydrocarbons.
- aromatic hydrocarbons such as alkylbenzenes or alkylnaphthalenes, exemplified by SOLVESSO 100, SOLVESSO 150 and SOLVESSO 200; SOLVESSO is a Registered Trade Mark
- ketones such as cycl
- An EC product may spontaneously emulsify on addition to water, to produce an emulsion with sufficient stability to allow spray application through appropriate equipment.
- Preparation of an EW involves obtaining a compound of formula (I) either as a liquid (if it is not a liquid at room temperature, it may be melted at a reasonable temperature, typically below 7O 0 C) or in solution (by dissolving it in an appropriate solvent) and then emulsifiying the resultant liquid or solution into water containing one or more SFAs, under high shear, to produce an emulsion.
- Suitable solvents for use in EWs include vegetable oils, chlorinated hydrocarbons (such as chlorobenzenes), aromatic solvents (such as alkylbenzenes or alkymaphthalenes) and other appropriate organic solvents which have a low solubility in water.
- Microemulsions may be prepared by mixing water with a blend of one or more solvents with one or more SFAs, to produce spontaneously a thermodynamically stable isotropic liquid formulation.
- a compound of formula (I) is present initially in either the water or the solvent/SFA blend.
- Suitable solvents for use in MEs include those hereinbefore described for use in in ECs or in EWs.
- An ME may be either an oil-in- water or a water-in-oil system (which system is present may be determined by conductivity measurements) and may be suitable for mixing water-soluble and oil-soluble pesticides in the same formulation.
- An ME is suitable for dilution into water, either remaining as a microemulsion or forming a conventional oil-in-water emulsion.
- SC Suspension concentrates
- SCs may comprise aqueous or non-aqueous suspensions of finely divided insoluble solid particles of a compound of formula (I).
- SCs may be prepared by ball or bead milling the solid compound of formula (I) in a suitable medium, optionally with one or more dispersing agents, to produce a fine particle suspension of the compound.
- One or more wetting agents may be included in the composition and a suspending agent may be included to reduce the rate at which the particles settle.
- a compound of formula (I) may be dry milled and added to water, containing agents hereinbefore described, to produce the desired end product.
- Aerosol formulations comprise a compound of formula (I) and a suitable propellant (for example n-butane).
- a compound of formula (I) may also be dissolved or dispersed in a suitable medium (for example water or a water miscible liquid, such as n-propanol) to provide compositions for use in non-pressurised, hand-actuated spray pumps.
- a compound of formula (I) may be mixed in the dry state with a pyrotechnic mixture to form a composition suitable for generating, in an enclosed space, a smoke containing the compound.
- Capsule suspensions (CS) may be prepared in a manner similar to the preparation of
- each oil droplet is encapsulated by a polymeric shell and contains a compound of formula (I) and, optionally, a carrier or diluent therefor.
- the polymeric shell may be produced by either an interfacial polycondensation reaction or by a coacervation procedure.
- the compositions may provide for controlled release of the compound of. formula (I) and they may be used for seed treatment.
- a compound of formula (I) may also be formulated in a biodegradable polymeric matrix to provide a slow, controlled release of the compound.
- a composition may include one or more additives to improve the biological performance of the composition (for example by improving wetting, retention or distribution on surfaces; resistance to rain on treated surfaces; or uptake or mobility of a compound of formula (I)).
- additives include surface active agents, spray additives based on oils, for example certain mineral oils or natural plant oils (such as soy bean and rape seed oil), and blends of these with other bio-enhancing adjuvants (ingredients which may aid or modify the action of a compound of formula (I)).
- a compound of formula (I) may also be formulated for use as a seed treatment, for example as a powder composition, including a powder for dry seed treatment (DS), a water soluble powder (SS) or a water dispersible powder for slurry treatment (WS), or as a liquid composition, including a flowable concentrate (FS), a solution (LS) or a capsule suspension (CS).
- DS powder for dry seed treatment
- SS water soluble powder
- WS water dispersible powder for slurry treatment
- CS capsule suspension
- the preparations of DS, SS, WS, FS and LS compositions are very similar to those of, respectively, DP, SP, WP, SC and DC compositions described above.
- Compositions for treating seed may include an agent for assisting the adhesion of the composition to the seed (for example a mineral oil or a film-forming barrier).
- Wetting agents, dispersing agents and emulsifying agents maybe surface SFAs of the cationic, anionic, amphoteric or non-ionic type.
- Suitable SFAs of the cationic type include quaternary ammonium compounds (for example cetyltrimethyl ammonium bromide), imidazolines and amine salts.
- Suitable anionic SFAs include alkali metals salts of fatty acids, salts of aliphatic monoesters of sulphuric acid (for example sodium lauryl sulphate), salts of sulphonated aromatic compounds (for example sodium dodecylbenzenesulphonate, calcium dodecylbenzenesulphonate, butylnaphthalene sulphonate and mixtures of sodium di- zsopropyl- and tri-wopropyl-naphthalene sulphonates), ether sulphates, alcohol ether sulphates (for example sodium laureth-3-sulphate), ether carboxylates (for example sodium laureth-3-carboxylate), phosphate esters (products from the reaction between one or more fatty alcohols and phosphoric acid (predominately mono-esters) or phosphorus pentoxide (predominately di-esters), for example the reaction between lauryl alcohol and tetraphosphoric acid
- Suitable SFAs of the non-ionic type include condensation products of alkylene oxides, such as ethylene oxide, propylene oxide, butylene oxide or mixtures thereof, with fatty alcohols (such as oleyl alcohol or cetyl alcohol) or with alkylphenols (such as octylphenol, nonylphenol or octylcresol); partial esters derived from long chain fatty acids or hexitol anhydrides; condensation products of said partial esters with ethylene oxide; block polymers (comprising ethylene oxide and propylene oxide); alkanolamides; simple esters (for example fatty acid polyethylene glycol esters); amine oxides (for example lauryl dimethyl amine oxide); and lecithins.
- alkylene oxides such as ethylene oxide, propylene oxide, butylene oxide or mixtures thereof
- fatty alcohols such as oleyl alcohol or cetyl alcohol
- alkylphenols such as octylphenol, nonyl
- Suitable suspending agents include hydrophilic colloids (such as polysaccharides, polyvinylpyrrolidone or sodium carboxymethylcellulose) and swelling clays (such as bentonite or attapulgite).
- hydrophilic colloids such as polysaccharides, polyvinylpyrrolidone or sodium carboxymethylcellulose
- swelling clays such as bentonite or attapulgite
- a compound of formula (I) may be applied by any of the known means of applying pesticidal compounds. For example, it may be applied, formulated or unformulated, to the pests or to a locus of the pests (such as a habitat of the pests, or a growing plant liable to infestation by the pests) or to any part of the plant, including the foliage, stems, branches or " roots, to the seed before it is planted or to other media in which plants are growing or are to be planted (such as soil surrounding the roots, the soil generally, paddy water or hydroponic culture systems), directly or it may be sprayed on, dusted on, applied by dipping, applied as a cream or paste formulation, applied as a vapour or applied through distribution or incorporation of a composition (such as a granular composition or a composition packed in a water-soluble bag) in soil or an aqueous environment.
- a locus of the pests such as a habitat of the pests, or a growing plant liable to infestation by the pests
- a compound of formula (I) may also be injected into plants or sprayed onto vegetation using electrodynamic spraying techniques or other low volume methods, or applied by land or aerial irrigation systems.
- Compositions for use as aqueous preparations are generally supplied in the form of a concentrate containing a high proportion of the active ingredient, the concentrate being added to water before use.
- These concentrates which may include DCs, SCs, ECs, EWs, MEs SGs, SPs, WPs, WGs and CSs, are often required to withstand storage for prolonged periods and, after such storage, to be capable of addition to water to form aqueous preparations which remain homogeneous for a sufficient time to enable them to be applied by conventional spray equipment.
- Such aqueous preparations may contain varying amounts of a compound of formula (I) (for example 0.0001 to 10%, by weight) depending upon the purpose for which they are to be used.
- a compound of formula (I) may be used in mixtures with fertilisers (for example nitrogen-, potassium- or phosphorus-containing fertilisers).
- fertilisers for example nitrogen-, potassium- or phosphorus-containing fertilisers.
- Suitable formulation types include granules of fertiliser.
- the mixtures suitably contain up to 25% by weight of the compound of formula (I) .
- the invention therefore also provides a fertiliser composition comprising a fertiliser and a compound of formula (I).
- compositions of this invention may contain other compounds having biological activity, for example micronutrients or compounds having fungicidal activity or which possess plant growth regulating, herbicidal, insecticidal, nematicidal or acaricidal activity.
- the compound of formula (I) may be the sole active ingredient of the composition or it may be admixed with one or more additional active ingredients such as a pesticide, fungicide, synergist, herbicide or plant growth regulator where appropriate.
- An additional active ingredient may: provide a composition having a broader spectrum of activity or increased persistence at a locus; synergise the activity or complement the activity (for example by increasing the speed of effect or overcoming repellency) of the compound of formula (I); or help to overcome or prevent the development of resistance to individual components.
- the particular additional active ingredient will depend upon the intended utility of the composition.
- Suitable pesticides include the following: a) Pyrethroids, such as permethrin, cypermethrin, fenvalerate, esfenvalerate, deltamethrin, cyhalothrin (in particular lambda-cyhalothrin), bifenthrin, fenpropathrin, cyfluthrin, tefluthrin, fish safe pyrethroids (for example ethofenprox), natural pyrethrin, tetramethrin, s-bioallethrin, fenfluthrin, prallethrin or 5-benzyl-3-furylmethyl-(E)-(lR,3S)-2,2-dimethyl- 3 -(2-oxothiolan-3 -ylidenemethyl)cyclopropane carboxylate; b) Organophosphates, such as, profenofos, sulprofos,
- Chloronicotinyl compounds such as imidacloprid, thiacloprid, acetamiprid, nitenpyram or thiamethoxam;
- Diacylhydrazines such as tebufenozide, chromafenozide or methoxyfenozide;
- Diphenyl ethers such as diofenolan or pyriproxifen; o) Indoxacarb; p) Chlorfenapyr; or q) Pymetrozine.
- pesticides having particular targets may be employed in the composition, if appropriate for the intended utility of the composition.
- selective insecticides for particular crops for example stemborer specific insecticides (such as cartap) or hopper specific insecticides (such as buprofezin) for use in rice may be employed.
- insecticides or acaricides specific for particular insect species/stages may also be included in the compositions (for example acaricidal ovo-larvicides, such as clofentezine, flubenzimine, hexythiazox or tetradifon; acaricidal motilicides, such as dicofol or propargite; acaricides, such as bromopropylate or chlorobenzilate; or growth regulators, such as hydramethylnon, cyromazine, methoprene, chlorfluazuron or diflubenzuron).
- acaricidal ovo-larvicides such as clofentezine, flubenzimine, hexythiazox or tetradifon
- acaricidal motilicides such as dicofol or propargite
- acaricides such as bromopropylate or chlorobenzilate
- growth regulators such
- fungicidal compounds which may be included in the composition of the invention are (E)-N-methyl-2-[2-(2,5-dimethylphenoxymethyl)phenyl]-2-methoxy- iminoacetamide (SSF-129), 4-bromo-2-cyano-N,N-dimethyl-6-trifluoromethylbenzimidazole- 1 -sulphonamide, ⁇ -[N-(3-chloro-2,6-xylyl)-2-methoxyacetamido]- ⁇ -butyrolactone, 4-chloro- 2-cyano-N,N-dimethyl-5-/>-tolylimidazole- 1 -sulfonamide (IKF-916, cyamidazosulfamid), 3 -5-dichloro-N-(3 -chloro- 1 -ethyl- 1 -methyl-2-oxopropyl)-4-methylbenzamide (RH-7281 , zoxamide
- the compounds of formula (I) may be mixed with soil, peat or other rooting media for the protection of plants against seed-borne, soil-borne or foliar fungal diseases.
- synergists for use in the compositions include piperonyl butoxide, sesamex, safroxan and dodecyl imidazole.
- Suitable herbicides and plant-growth regulators for inclusion in the compositions will depend upon the intended target and the effect required.
- a rice selective herbicide which may be included is propanil.
- An example of a plant growth regulator for use in cotton is PIXTM.
- Some mixtures may comprise active ingredients which have significantly different physical, chemical or biological properties such that they do not easily lend themselves to the same conventional formulation type. In these circumstances other formulation types may be prepared. For example, where one active ingredient is a water insoluble solid and the other a water insoluble liquid, it may nevertheless be possible to disperse each active ingredient in the same continuous aqueous phase by dispersing the solid active ingredient as a suspension (using a preparation analogous to that of an SC) but dispersing the liquid active ingredient as an emulsion (using a preparation analogous to that of an EW).
- SE suspoemulsion
- Step A Preparation of N-(4-Chloro-phenyl)-2,2-dimethyl-propiffnide
- 4-chloroaniline 25.51 g
- triethylamine 69.73 ml
- chloroform 350 ml
- 2,2-dimethyl-propionyl chloride 25.32 g
- the resulting solution was stirred at r.t. for 1 hour, then water was added and the mixture extracted three times with ethyl acetate.
- the combined organic layers were dried over sodium sulfate and concentrated in vacuo to afford 35.8 g of product.
- M.p. 149-15O 0 C Retention Time HPLC 2.83 min
- Step B Preparation of N-(4-Chloro-2- ⁇ l-[(E)-3-(4-chloro-phenyl)-allyl]-4-hydroxy- piperidin-4-yl ⁇ -phenyl)-2,2-dimethyl-propionamide
- This Example illustrates the preparation of 4-Chloro-2- ⁇ l-[(E)-3-(4-chloro-phenyl)-allyl]- 1 ,2,3 ,6-tetrahydro-pyridin-4-yl ⁇ -phenylamine and 4-(2-Amino-5-chloro-phenyl)- 1 -[(E)-3 -(4- chloro-phenyl)-allyl]-piperidin-4-ol.
- This Example illustrates the preparation of 2-(l-Benzyl-l,2,3,6-tetrahydro-pyridin-4-yl)-4- chloro-phenylamine and 4-(2-Amino-5-chloro-phenyl)-l-benzyl-piperidin-4-ol.
- Step A Preparation of 4- ⁇ 5-Chloro-2-[(2-chloro-pyridine-4-carbonyl)- amino]phenyl ⁇ -3,6-dihydro ⁇ 2H-pyridine-l-carboxylic acid 1-chloro-ethyl ester
- Step A Preparation of N-(4-Fluoro-phenyl)-2,2-dimethyl-propionamide
- 4-fluoroaniline 50.0 g
- triethylamine 157 ml
- 2,2-dimethyl-propionyl chloride 58.0 ml
- the resulting solution was stirred at r.t. for 2 hour, then water was added and the mixture extracted three times with ethyl acetate.
- the combined organic layers were dried over sodium sulfate and concentrated in vacuo to afford 86.0 g of the title compound.
- M.p. 124-125 0 C Retention Time HPLC 2.57 min
- Step B Preparation of N-(4-Fluoro-2- ⁇ l-[(E)-3-(4-chloro-phenyl)-allyl]-4-hydroxy- piperidin-4-yl ⁇ -phenyl)-2,2-dimethyl-propionamide
- N-(4-Fluoro-2-piperidin-4-yl-phenyl)-2,2-dimethyl-propionamide (380 mg) was dissolved in CHCl 3 (20 ml) and treated with triethyl amine (0.260 mg). Then, a solution of l-((E)-3- chloro-propenyl)-4-chloro-benzene (255 mg) was added. After stirring for 16 hours at r.t. the mixture was filtrated and concentrated in vacuo. The residue was subjected to silica gel chromatography (hexane:ethyl acetate:triethyl amine 74:24:2) to afford the title product (380 mg). M.p. 174-176 0 C; Retention Time HPLC 2.37 min; MS (ES+) 4.29 (M+H + ).
- Triethylamine (2.8 ml) was added to a stirred solution of the compound obtained in example 18 (2.66 g) in dichloromethane (100 ml); the solution was cooled to 0°C and 2- chloroisonicotinoyl chloride (prepared from 2.05 g of 2-chloroisonicotinic acid and 1.46 ml of oxalyl chloride in 50 ml dichloromethane) was added. The resulting mixture was stirred at room temperature for 12 hours, poured into water, extracted two times with dichloromethane; the combined organic layers were dried over sodium sulfate and concentrated in vacuo.
- 2- chloroisonicotinoyl chloride prepared from 2.05 g of 2-chloroisonicotinic acid and 1.46 ml of oxalyl chloride in 50 ml dichloromethane
- Step A l-(t-Butoxycarbonyl)-4-tributylstannyl-l,2,3,6-tetrahydropyridine (2.12 g, prepared in 2 steps from l-(t-butoxycarbonyl)-piperidin-4-one according to WO 0123381) was dissolved in toluene (45 ml) in a dried, nitrogen-flushed flask. 2-Chloro-3-nitropyridine (712 mg) and palladium tetrakis(triphenylphosphine) (130 mg) were added and the solution was heated at 110°C for 16 hours.
- Step B Hydrazine monohydrate (0.4 ml) was added to a suspension of Raney nickel (50% slurry in water, 200 mg) and the product obtained in Step A (240 mg) in ethanol (10 ml). After 4 hours stirring, the reaction mixture was filtered over Hyfio and the solvent removed in vacuo. The residue was dissolved in ethyl acetate, dried over sodium sulfate, filtered and concentrated in vacuo to afford 3-amino-3',6'-dihydro-2'H-[2,4']bipyridinyl-r-carboxylic acid tert-butyl ester (200 mg) as white cystals. M.p.
- Step C The product obtained in Step B (815 mg) was reduced by transfer hydrogenation using 10% Pd/C (200 mg) and ammonium formate (935 mg) in ethanol (40 ml) at 60°C for 45 min.
- Step D sodium bicarbonate (714 mg) was added to a stirred solution of the compound obtained in Step C (785 mg) in dichloromethane (30 ml); the solution was then treated with 2-chloro-isonicotinoyl chloride (500 mg) and the resulting mixture was stirred at room temperature for 1 hour, poured into water, extracted two times with dichloromethane, the combined organic layers were dried over sodium sulfate and concentrated in vacuo to afford 3-[(2-Chloro-pyridine-4-carbonyl)-amino]-3',4',5 t ,6'-tetrahydro-2 ⁇ -[2,4']bipyridinyl-r- carboxylic acid tert-butyl ester (1.2 g).
- Step E A solution of the compound obtained in Step D (834 mg) in dichloromethane (40 ml) was treated with trifiuoroacetic acid (4 ml) for 5 hours at room temperature. The reaction mixture was concentrated in vacuo and then dried under high vacuum for 1 hour. The residue was dissolved in acetonitrile (40 ml), diisopropylethylamine (1.8 ml) and 4- chlorocinnamyl chloride (380 mg) were added. The solution was stirred 20 hours at room temperature, the solvent was removed in vacuo and the residue was subjected to silica gel chromatography (ethyl acetate:methanol 95:5) to afford the title product (409 mg) as a yellow solid. M.p.
- This Example illustrates the preparation of 2-Chloro-N- ⁇ 5-chloro-l'-[(E)-3-(4-chloro- ⁇ henyl)-allyl]-r,2 l ,3 l ,4',5',6 l -hexahydro-[2,4']bi ⁇ yridinyl-3-yl ⁇ -isonicotinamide.
- Cotton leaf discs were placed on agar in a 24- well microtiter plate and sprayed with test solutions at an application rate of 200 ppm. After drying, the leaf discs were infested with 5 L 1 larvae. The samples were checked for mortality, repellent effect, feeding behaviour, and growth regulation 3 days after treatment (DAT). The following compounds gave at least 80% control of Spodoptera littoralis: Iaaa-3 and Iaaa-49. Heliothis virescens ( Tobacco budworm):
- Aedes aegypti Yellow fever mosquito
- Aedes larvae (L2) together with a nutrition mixture are placed in 96-well microtiter plates. Test solutions at an application rate of 2ppm are pipetted into the wells. 2 days later, insects were checked for mortality and growth inhibition. The following compounds gave at least 80% control of Aedes aegypti: Ia-53, Iaaa-3, Iaaa-26, Iaaa-49, Iaaa-52, Iaab-26, Iaac-26 and Iaai-26.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Dentistry (AREA)
- Wood Science & Technology (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Agronomy & Crop Science (AREA)
- General Health & Medical Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Hydrogenated Pyridines (AREA)
Abstract
Description
Claims
Priority Applications (20)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020067027660A KR101338876B1 (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
| CA2568808A CA2568808C (en) | 2004-06-28 | 2005-06-22 | Chemical compounds |
| BRPI0512659-2A BRPI0512659A (en) | 2004-06-28 | 2005-06-22 | chemical compounds |
| ES05757532T ES2408856T3 (en) | 2004-06-28 | 2005-06-22 | Chemical compounds |
| AU2005258905A AU2005258905B2 (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
| EP05757532A EP1763302B1 (en) | 2004-06-28 | 2005-06-22 | Chemical compounds |
| PL05757532T PL1763302T3 (en) | 2004-06-28 | 2005-06-22 | Chemical compounds |
| NZ551629A NZ551629A (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
| AP2008004646A AP2008004646A0 (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
| JP2007517523A JP5043653B2 (en) | 2004-06-28 | 2005-06-22 | Compound |
| MXPA06014005A MXPA06014005A (en) | 2004-06-28 | 2005-06-22 | Chemical compounds. |
| US11/571,303 US8129534B2 (en) | 2004-06-28 | 2005-06-22 | Chemical compounds |
| KR1020127019520A KR101268288B1 (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
| EA200602170A EA014686B1 (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives, compositions based thereon methods for use thereof |
| AP2006003830A AP1970A (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
| KR1020137008545A KR101396174B1 (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
| IL179745A IL179745A (en) | 2004-06-28 | 2006-11-30 | Method for combating and controlling insects, piperidine derivatives and insecticidal and acaricidal compositions comprising said derivatives |
| TNP2006000442A TNSN06442A1 (en) | 2004-06-28 | 2006-12-27 | Chemical compounds |
| US13/371,002 US8546569B2 (en) | 2004-06-28 | 2012-02-10 | Chemical compounds |
| US13/949,803 US9045422B2 (en) | 2004-06-28 | 2013-07-24 | Piperidine derivatives with pesticidal properties |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GBGB0414438.2A GB0414438D0 (en) | 2004-06-28 | 2004-06-28 | Chemical compounds |
| GB0414438.2 | 2004-06-28 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/571,303 A-371-Of-International US8129534B2 (en) | 2004-06-28 | 2005-06-22 | Chemical compounds |
| US13/371,002 Division US8546569B2 (en) | 2004-06-28 | 2012-02-10 | Chemical compounds |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2006003494A2 true WO2006003494A2 (en) | 2006-01-12 |
| WO2006003494A3 WO2006003494A3 (en) | 2006-06-15 |
Family
ID=32800314
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2005/002002 WO2006003494A2 (en) | 2004-06-28 | 2005-06-22 | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides |
Country Status (26)
| Country | Link |
|---|---|
| US (3) | US8129534B2 (en) |
| EP (1) | EP1763302B1 (en) |
| JP (1) | JP5043653B2 (en) |
| KR (3) | KR101268288B1 (en) |
| CN (2) | CN103214460A (en) |
| AP (2) | AP1970A (en) |
| AR (1) | AR049556A1 (en) |
| AU (1) | AU2005258905B2 (en) |
| BR (1) | BRPI0512659A (en) |
| CA (1) | CA2568808C (en) |
| CR (1) | CR8790A (en) |
| EA (1) | EA014686B1 (en) |
| EC (1) | ECSP067112A (en) |
| ES (1) | ES2408856T3 (en) |
| GB (1) | GB0414438D0 (en) |
| IL (1) | IL179745A (en) |
| MA (1) | MA28678B1 (en) |
| MX (1) | MXPA06014005A (en) |
| NZ (1) | NZ551629A (en) |
| PL (1) | PL1763302T3 (en) |
| PT (1) | PT1763302E (en) |
| TN (1) | TNSN06442A1 (en) |
| TW (1) | TWI379636B (en) |
| UA (1) | UA89788C2 (en) |
| WO (1) | WO2006003494A2 (en) |
| ZA (1) | ZA200609687B (en) |
Cited By (308)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007072143A2 (en) * | 2005-12-21 | 2007-06-28 | Syngenta Participations Ag | Chemical compounds |
| WO2009138219A2 (en) * | 2008-05-15 | 2009-11-19 | Syngenta Participations Ag | Insecticidal compounds |
| WO2010009968A1 (en) * | 2008-07-22 | 2010-01-28 | Syngenta Participations Ag | Insecticidal phenyl- or pyridyl-piperidine compounds |
| WO2011003684A1 (en) * | 2009-07-06 | 2011-01-13 | Syngenta Participations Ag | Insecticidal compounds |
| US8114868B2 (en) | 2008-07-25 | 2012-02-14 | Boehringer Ingelheim International Gmbh | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8138178B2 (en) | 2008-05-01 | 2012-03-20 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8202857B2 (en) | 2008-02-11 | 2012-06-19 | Vitae Pharmaceuticals, Inc. | 1,3-oxazepan-2-one and 1,3-diazepan-2-one inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8242111B2 (en) | 2008-05-01 | 2012-08-14 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8329897B2 (en) | 2007-07-26 | 2012-12-11 | Vitae Pharmaceuticals, Inc. | Synthesis of inhibitors of 11β-hydroxysteroid dehydrogenase type 1 |
| US8440658B2 (en) | 2007-12-11 | 2013-05-14 | Vitae Pharmaceuticals, Inc. | Cyclic urea inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8569292B2 (en) | 2008-05-01 | 2013-10-29 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| WO2013164954A1 (en) * | 2012-05-01 | 2013-11-07 | 住友化学株式会社 | Piperidine compound and pest-control use therefore |
| US8592410B2 (en) | 2008-05-01 | 2013-11-26 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11BETA-hydroxysteroid dehydrogenase 1 |
| US8592409B2 (en) | 2008-01-24 | 2013-11-26 | Vitae Pharmaceuticals, Inc. | Cyclic carbazate and semicarbazide inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8598160B2 (en) | 2008-02-15 | 2013-12-03 | Vitae Pharmaceuticals, Inc. | Cycloalkyl lactame derivatives as inhibitors of 11-beta-hydroxysteroid dehydrogenase 1 |
| WO2014005982A1 (en) | 2012-07-05 | 2014-01-09 | Bayer Cropscience Ag | Insecticide and fungicide active ingredient combinations |
| US8637505B2 (en) | 2009-02-04 | 2014-01-28 | Boehringer Ingelheim International Gmbh | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8680093B2 (en) | 2009-04-30 | 2014-03-25 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8680281B2 (en) | 2008-01-07 | 2014-03-25 | Vitae Pharmaceuticals, Inc. | Lactam inhibitors of 11-β-hydroxysteroid dehydrogenase 1 |
| US8765744B2 (en) | 2010-06-25 | 2014-07-01 | Boehringer Ingelheim International Gmbh | Azaspirohexanones |
| US8835426B2 (en) | 2007-02-26 | 2014-09-16 | Vitae Pharmaceuticals, Inc. | Cyclic urea and carbamate inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8846613B2 (en) | 2010-11-02 | 2014-09-30 | Boehringer Ingelheim International Gmbh | Pharmaceutical combinations for the treatment of metabolic disorders |
| US8846668B2 (en) | 2008-07-25 | 2014-09-30 | Vitae Pharmaceuticals, Inc. | Inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8883778B2 (en) | 2009-07-01 | 2014-11-11 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11 beta-hydroxysteroid dehydrogenase 1 |
| US8927539B2 (en) | 2009-06-11 | 2015-01-06 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 based on the 1,3-oxazinan-2-one structure |
| US8933072B2 (en) | 2010-06-16 | 2015-01-13 | Vitae Pharmaceuticals, Inc. | Substituted 5-,6- and 7-membered heterocycles, medicaments containing such compounds, and their use |
| WO2015055554A1 (en) | 2013-10-14 | 2015-04-23 | Bayer Cropscience Ag | Active substance for treating seed and soil |
| WO2015059088A1 (en) | 2013-10-23 | 2015-04-30 | Bayer Cropscience Ag | Substituted quinoxaline derivatives as pest control agent |
| WO2015101622A1 (en) | 2014-01-03 | 2015-07-09 | Bayer Cropscience Ag | Novel pyrazolyl-heteroarylamides as pesticides |
| US9079861B2 (en) | 2007-11-07 | 2015-07-14 | Vitae Pharmaceuticals, Inc. | Cyclic urea inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| WO2015107133A1 (en) | 2014-01-20 | 2015-07-23 | Bayer Cropscience Ag | Quinoline derivatives as insecticides and acaricides |
| EP2910126A1 (en) | 2015-05-05 | 2015-08-26 | Bayer CropScience AG | Active compound combinations having insecticidal properties |
| WO2015150300A1 (en) | 2014-04-02 | 2015-10-08 | Bayer Cropscience Ag | N-(1-(hetero)aryl-1h-pyrazol-4-yl)-(hetero)arylamide derivatives and use thereof as pesticides |
| WO2015169776A1 (en) | 2014-05-08 | 2015-11-12 | Bayer Cropscience Ag | Pyrazolopyridine sulfonamides as nematicides |
| WO2015185531A1 (en) | 2014-06-05 | 2015-12-10 | Bayer Cropscience Aktiengesellschaft | Bicyclic compounds as pesticides |
| WO2016001119A1 (en) | 2014-07-01 | 2016-01-07 | Bayer Cropscience Aktiengesellschaft | Insecticide and fungicide active ingredient combinations |
| WO2016008830A1 (en) | 2014-07-15 | 2016-01-21 | Bayer Cropscience Aktiengesellschaft | Aryl-triazolyl pyridines as pest control agents |
| WO2016055096A1 (en) | 2014-10-07 | 2016-04-14 | Bayer Cropscience Ag | Method for treating rice seed |
| WO2016091857A1 (en) | 2014-12-11 | 2016-06-16 | Bayer Cropscience Aktiengesellschaft | Five-membered c-n bonded aryl sulphide and aryl sulphoxide derivatives as pest control agents |
| WO2016124563A1 (en) | 2015-02-05 | 2016-08-11 | Bayer Cropscience Aktiengesellschsaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2016124557A1 (en) | 2015-02-05 | 2016-08-11 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2016128298A1 (en) | 2015-02-09 | 2016-08-18 | Bayer Cropscience Aktiengesellschaft | Substituted 2-thioimidazolyl-carboxamides as pest control agents |
| WO2016142394A1 (en) | 2015-03-10 | 2016-09-15 | Bayer Animal Health Gmbh | Pyrazolyl-derivatives as pest control agents |
| WO2016162318A1 (en) | 2015-04-08 | 2016-10-13 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents and intermediate product |
| EP3081085A1 (en) | 2015-04-14 | 2016-10-19 | Bayer CropScience AG | Method for improving earliness in cotton |
| WO2016174049A1 (en) | 2015-04-30 | 2016-11-03 | Bayer Animal Health Gmbh | Anti-parasitic combinations including halogen-substituted compounds |
| WO2016180802A1 (en) | 2015-05-13 | 2016-11-17 | Bayer Cropscience Aktiengesellschaft | Insecticidal arylpyrrolidines, method for synthesizing same, and use thereof as agents for controlling animal pests |
| WO2017005717A1 (en) | 2015-07-06 | 2017-01-12 | Bayer Cropscience Aktiengesellschaft | Heterocyclic compounds as pesticides |
| WO2017072039A1 (en) | 2015-10-26 | 2017-05-04 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093180A1 (en) | 2015-12-01 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093214A1 (en) | 2015-12-03 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Mesoionic halogenated 3-(acetyl)-1-[(1,3-thiazol-5-yl)methyl]-1h-imidazo[1,2-a]pyridin-4-ium-2-olate derivatives and related compounds as insecticides |
| WO2017137339A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituted 2-oxyimidazolyl-carboxamides as pest control agents |
| WO2017137338A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituierted 2-(het)aryl-imidazolyl-carboxyamides as pest control agents |
| EP3210468A1 (en) | 2016-02-26 | 2017-08-30 | Bayer CropScience Aktiengesellschaft | Solvent-free formulations of low-melting point agents |
| WO2017144341A1 (en) | 2016-02-23 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017157735A1 (en) | 2016-03-15 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2017157885A1 (en) | 2016-03-16 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | N-(cyanobenzyl)-6-(cyclopropyl-carbonylamino)-4-(phenyl)-pyridine-2-carboxamide derivatives and related compounds as pesticides and plant protection agents |
| US9776967B2 (en) | 2013-10-14 | 2017-10-03 | Bayer Animal Health Gmbh | Carboxamide derivatives as pesticidal compounds |
| WO2017174414A1 (en) | 2016-04-05 | 2017-10-12 | Bayer Cropscience Aktiengesellschaft | Naphthaline-derivatives as pest control agents |
| WO2017178416A1 (en) | 2016-04-15 | 2017-10-19 | Bayer Animal Health Gmbh | Pyrazolopyrimidine derivatives |
| WO2017186536A1 (en) | 2016-04-25 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Substituted 2-alkylimidazolyl-carboxamides as pest control agents |
| EP3241830A1 (en) | 2016-05-04 | 2017-11-08 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pesticides |
| EP3245865A1 (en) | 2016-05-17 | 2017-11-22 | Bayer CropScience Aktiengesellschaft | Method for increasing yield in brassicaceae |
| WO2017198451A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in small grain cereals such as wheat and rice |
| WO2017198452A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in soybean |
| WO2017198455A2 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in beta spp. plants |
| WO2017198453A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in potato, tomato or alfalfa |
| WO2017198449A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in brassicaceae |
| WO2017198454A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in cotton |
| WO2017198450A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in maize |
| WO2018011111A1 (en) | 2016-07-12 | 2018-01-18 | Bayer Cropscience Aktiengesellschaft | Bicyclic compounds as pest control agents |
| WO2018013380A1 (en) | 2016-07-11 | 2018-01-18 | Covestro Llc, Et Al. | Methods for treating seeds with an aqueous compostion and seeds treated therewith |
| WO2018013382A1 (en) | 2016-07-11 | 2018-01-18 | Covestro Llc, Et Al | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| WO2018013381A1 (en) | 2016-07-11 | 2018-01-18 | Covestro Llc, Et Al. | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| WO2018015289A1 (en) | 2016-07-19 | 2018-01-25 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018019937A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Formulation comprising a beneficial p. bilaii strain and talc for use in seed treatment |
| WO2018029102A1 (en) | 2016-08-10 | 2018-02-15 | Bayer Cropscience Aktiengesellschaft | Substituted 2-heterocyclyl imidazolyl-carboxamides as pest control agents |
| EP3284739A1 (en) | 2017-07-19 | 2018-02-21 | Bayer CropScience Aktiengesellschaft | Substituted (het) aryl compounds as pesticides |
| WO2018033455A1 (en) | 2016-08-15 | 2018-02-22 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018050825A1 (en) | 2016-09-19 | 2018-03-22 | Bayer Cropscience Aktiengesellschaft | Pyrazolo [1,5-a]pyridine derivatives and their use as pesticides |
| EP3305786A2 (en) | 2018-01-22 | 2018-04-11 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pesticides |
| WO2018065288A1 (en) | 2016-10-07 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-[2-phenyl-1-(sulfonyl-methyl)-vinyl]-imidazo-[4,5-b] pyridine derivatives and related compounds as pesticides in plant protection |
| WO2018065292A1 (en) | 2016-10-06 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018069841A1 (en) | 2016-10-14 | 2018-04-19 | Pi Industries Ltd | 4-substituted phenylamine derivatives and their use to protect crops by fighting undesired phytopathogenic micoorganisms |
| WO2018069842A1 (en) | 2016-10-14 | 2018-04-19 | Pi Industries Ltd | 4-amino substituted phenylamidine derivatives and their use to protect crops by fighting undesired phytopathogenic micoorganisms |
| WO2018083288A1 (en) | 2016-11-07 | 2018-05-11 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2018087036A1 (en) | 2016-11-11 | 2018-05-17 | Bayer Animal Health Gmbh | New anthelmintic quinoline-3-carboxamide derivatives |
| WO2018095953A1 (en) | 2016-11-23 | 2018-05-31 | Bayer Cropscience Aktiengesellschaft | 2-[3-(alkylsulfonyl)-2h-indazol-2-yl]-3h-imidazo[4,5-b]pyridine derivatives and similar compounds as pesticides |
| WO2018104500A1 (en) | 2016-12-09 | 2018-06-14 | Bayer Cropscience Aktiengesellschaft | Plant health effect of purpureocillium lilacinum |
| WO2018108791A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Thiadiazole derivatives as pesticides |
| WO2018108730A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018116073A1 (en) | 2016-12-21 | 2018-06-28 | Pi Industries Ltd. | 1, 2, 3-thiadiazole compounds and their use as crop protecting agent |
| WO2018116072A1 (en) | 2016-12-20 | 2018-06-28 | Pi Industries Ltd. | Heterocyclic compounds |
| WO2018130437A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018130443A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018138050A1 (en) | 2017-01-26 | 2018-08-02 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclene derivatives as pest control agents |
| WO2018141954A1 (en) | 2017-02-06 | 2018-08-09 | Bayer Aktiengesellschaft | Aryl or heteroaryl-substituted imidazo pyridine derivatives and their use as pesticides |
| EP3369320A1 (en) | 2017-03-02 | 2018-09-05 | Bayer CropScience Aktiengesellschaft | Agent for controlling bugs |
| WO2018177993A1 (en) | 2017-03-31 | 2018-10-04 | Bayer Cropscience Aktiengesellschaft | Pyrazoles for controlling arthropods |
| WO2018177995A1 (en) | 2017-03-31 | 2018-10-04 | Bayer Cropscience Aktiengesellschaft | Tricyclic carboxamides for controlling arthropods |
| WO2018189077A1 (en) | 2017-04-12 | 2018-10-18 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018193385A1 (en) | 2017-04-20 | 2018-10-25 | Pi Industries Ltd. | Novel phenylamine compounds |
| WO2018192872A1 (en) | 2017-04-21 | 2018-10-25 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2018197257A1 (en) | 2017-04-24 | 2018-11-01 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic-compound derivatives as pest control agents |
| WO2018197401A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Animal Health Gmbh | New bicyclic pyrazole derivatives |
| WO2018197692A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Aktiengesellschaft | Heteroarylphenylaminoquinolines and analogues |
| WO2018202712A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylphenoxyquinolines and analogues |
| WO2018202494A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202706A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylheteroaryloxyquinolines and analogues |
| WO2018202501A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202525A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | Phenoxyethanamine derivatives for controlling pests |
| WO2018202715A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylbenzylbenzimidazoles and analogues |
| WO2018202524A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | 2-{[2-(phenyloxymethyl)pyridin-5-yl]oxy}-ethanamin-derivatives and related compounds as pest-control agents e.g. for the protection of plants |
| WO2019002132A1 (en) | 2017-06-30 | 2019-01-03 | Bayer Animal Health Gmbh | New azaquinoline derivatives |
| WO2019007888A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insecticidal active ingredient combinations |
| WO2019007887A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insecticide and fungicide active ingredient combinations |
| WO2019007891A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insecticidal active ingredient combinations |
| WO2019025341A1 (en) | 2017-08-04 | 2019-02-07 | Bayer Animal Health Gmbh | Quinoline derivatives for treating infections with helminths |
| WO2019035881A1 (en) | 2017-08-17 | 2019-02-21 | Bayer Cropscience Lp | Liquid fertilizer-dispersible compositions and methods thereof |
| WO2019038195A1 (en) | 2017-08-22 | 2019-02-28 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019059412A1 (en) | 2017-09-20 | 2019-03-28 | Mitsui Chemicals Agro, Inc. | Prolonged ectoparasite-controlling agent for animal |
| WO2019068572A1 (en) | 2017-10-04 | 2019-04-11 | Bayer Aktiengesellschaft | Derivatives of heterocyclic compounds as pest control agents |
| EP3473100A1 (en) | 2017-10-18 | 2019-04-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3473103A1 (en) | 2017-10-17 | 2019-04-24 | Bayer AG | Aqueous suspension concentrates based on 2- [(2,4-dichlorophenyl) -methyl] -4,4 '-dimethyl-3-isoxazolidinone |
| WO2019076749A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076754A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076751A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076750A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076752A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019092086A1 (en) | 2017-11-13 | 2019-05-16 | Bayer Aktiengesellschaft | Tetrazolylpropyl derivatives and their use as fungicides |
| WO2019105875A1 (en) | 2017-11-28 | 2019-06-06 | Bayer Aktiengesellschaft | Heterocyclic compounds as pesticides |
| WO2019105871A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Aktiengesellschaft | Nitrogenous heterocycles as a pesticide |
| WO2019122319A1 (en) | 2017-12-21 | 2019-06-27 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylheteroaryloxyquinolines and analogues |
| WO2019123196A1 (en) | 2017-12-20 | 2019-06-27 | Pi Industries Ltd. | Fluoralkenyl compounds, process for preparation and use thereof |
| WO2019150311A1 (en) | 2018-02-02 | 2019-08-08 | Pi Industries Ltd. | 1-3 dithiol compounds and their use for the protection of crops from phytopathogenic microorganisms |
| WO2019155066A1 (en) | 2018-02-12 | 2019-08-15 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2019162228A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | 1-(5-substituted imidazol-1-yl)but-3-en derivatives and their use as fungicides |
| WO2019162174A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019170626A1 (en) | 2018-03-08 | 2019-09-12 | Bayer Aktiengesellschaft | Use of heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides in plant protection |
| WO2019175046A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| EP3545764A1 (en) | 2019-02-12 | 2019-10-02 | Bayer AG | Crystal form of 2-({2-fluoro-4-methyl-5-[(r)-(2,2,2-trifluoroethyl)sulfinyl]phenyl}imino)-3-(2,2,2- trifluoroethyl)-1,3-thiazolidin-4-one |
| WO2019197468A1 (en) | 2018-04-12 | 2019-10-17 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| WO2019197615A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with fungicides, insecticides and acaricidal properties |
| WO2019197623A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, fungicidal and acaricidal properties |
| WO2019197371A1 (en) | 2018-04-10 | 2019-10-17 | Bayer Aktiengesellschaft | Oxadiazoline derivatives |
| WO2019201835A1 (en) | 2018-04-17 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019201921A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019202077A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019206799A1 (en) | 2018-04-25 | 2019-10-31 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| EP3564225A1 (en) | 2019-03-21 | 2019-11-06 | Bayer Aktiengesellschaft | Crystalline form of spiromesifen |
| WO2019215182A1 (en) | 2018-05-09 | 2019-11-14 | Bayer Animal Health Gmbh | New quinoline derivatives |
| WO2019224143A1 (en) | 2018-05-24 | 2019-11-28 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, nematicidal and acaricidal properties |
| EP3586630A1 (en) | 2018-06-28 | 2020-01-01 | Bayer AG | Active compound combinations having insecticidal/acaricidal properties |
| WO2020005678A1 (en) | 2018-06-25 | 2020-01-02 | Bayer Cropscience Lp | Seed treatment method |
| WO2020002189A1 (en) | 2018-06-27 | 2020-01-02 | Bayer Aktiengesellschaft | Active substance combinations |
| WO2020007905A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020020813A1 (en) | 2018-07-25 | 2020-01-30 | Bayer Aktiengesellschaft | Fungicidal active compound combinations |
| WO2020020816A1 (en) | 2018-07-26 | 2020-01-30 | Bayer Aktiengesellschaft | Novel triazole derivatives |
| WO2020021082A1 (en) | 2018-07-27 | 2020-01-30 | Bayer Aktiengesellschaft | Controlled release formulations for agrochemicals |
| WO2020025650A1 (en) | 2018-07-31 | 2020-02-06 | Bayer Aktiengesellschaft | Controlled release formulations with lignin for agrochemicals |
| EP3608311A1 (en) | 2019-06-28 | 2020-02-12 | Bayer AG | Crystalline form a of n-[4-chloro-3-[(1-cyanocyclopropyl)carbamoyl]phenyl]-2-methyl-4-methylsulfonyl-5-(1,1,2,2,2-pentafluoroethyl)pyrazole-3-carboxamide |
| WO2020035826A1 (en) | 2018-08-17 | 2020-02-20 | Pi Industries Ltd. | 1,2-dithiolone compounds and use thereof |
| WO2020043650A1 (en) | 2018-08-29 | 2020-03-05 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3620052A1 (en) | 2018-12-12 | 2020-03-11 | Bayer Aktiengesellschaft | Use of phenoxypyridinyl-substituted (1h-1,2,4-triazol-1-yl)alcohols for controlling fungicidal diseases in maize |
| WO2020053282A1 (en) | 2018-09-13 | 2020-03-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2020057939A1 (en) | 2018-09-17 | 2020-03-26 | Bayer Aktiengesellschaft | Use of the fungicide isoflucypram for controlling claviceps purpurea and reducing sclerotia in cereals |
| WO2020070050A1 (en) | 2018-10-01 | 2020-04-09 | Bayer Aktiengesellschaft | Fungicidal 5-substituted imidazol-1-yl carbinol derivatives |
| EP3636644A1 (en) | 2018-10-11 | 2020-04-15 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2020079173A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Pyridylphenylaminoquinolines and analogues |
| WO2020079232A1 (en) | 2018-10-20 | 2020-04-23 | Bayer Aktiengesellschaft | Oxetanylphenoxyquinolines and analogues |
| WO2020079167A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Heteroarylaminoquinolines and analogues |
| WO2020078839A1 (en) | 2018-10-16 | 2020-04-23 | Bayer Aktiengesellschaft | Active substance combinations |
| EP3643711A1 (en) | 2018-10-24 | 2020-04-29 | Bayer Animal Health GmbH | New anthelmintic compounds |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020114934A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicide compositions |
| WO2020114932A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP3669652A1 (en) | 2018-12-21 | 2020-06-24 | Bayer AG | Active compound combination |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| WO2020127974A1 (en) | 2018-12-21 | 2020-06-25 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as new antifungal agents |
| WO2020126980A1 (en) | 2018-12-18 | 2020-06-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3679793A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679791A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679789A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679792A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679790A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3701796A1 (en) | 2019-08-08 | 2020-09-02 | Bayer AG | Active compound combinations |
| WO2020173861A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2020173860A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Fused bicyclic heterocycle derivatives as pesticides |
| WO2020178067A1 (en) | 2019-03-01 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020178307A1 (en) | 2019-03-05 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combination |
| EP3708565A1 (en) | 2020-03-04 | 2020-09-16 | Bayer AG | Pyrimidinyloxyphenylamidines and the use thereof as fungicides |
| WO2020182929A1 (en) | 2019-03-13 | 2020-09-17 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| WO2020187656A1 (en) | 2019-03-15 | 2020-09-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3725788A1 (en) | 2019-04-15 | 2020-10-21 | Bayer AG | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020225438A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High uptake and rainfastness ulv formulations |
| WO2020225242A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020231751A1 (en) | 2019-05-10 | 2020-11-19 | Bayer Cropscience Lp | Active compound combinations |
| WO2020229398A1 (en) | 2019-05-14 | 2020-11-19 | Bayer Aktiengesellschaft | (1-alkenyl)-substituted pyrazoles and triazoles as pest control agents |
| EP3750888A1 (en) | 2019-06-12 | 2020-12-16 | Bayer Aktiengesellschaft | Crystalline form a of 1,4-dimethyl-2-[2-(pyridin-3-yl)-2h-indazol-5-yl]-1,2,4-triazolidine-3,5-dione |
| WO2020254487A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254493A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Thienylhydroxyisoxazolines and derivatives thereof |
| WO2020254486A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254490A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Phenoxyphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254494A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2020254488A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and use thereof as fungicides |
| WO2020254492A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254489A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Benzylphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020263812A1 (en) | 2019-06-24 | 2020-12-30 | Auburn University | A bacillus strain and methods of its use for plant growth promotion |
| WO2021001331A1 (en) | 2019-07-03 | 2021-01-07 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides and derivatives thereof as microbicides |
| WO2021001273A1 (en) | 2019-07-04 | 2021-01-07 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2021013720A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013719A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013721A1 (en) | 2019-07-22 | 2021-01-28 | Bayer Aktiengesellschaft | 5-amino substituted pyrazoles and triazoles as pest control agents |
| EP3771714A1 (en) | 2019-07-30 | 2021-02-03 | Bayer AG | Nitrogen-containing heterocycles as pesticides |
| WO2021018839A1 (en) | 2019-07-30 | 2021-02-04 | Bayer Animal Health Gmbh | Isoquinoline derivatives and their use for the treatment of parasitic infections |
| WO2021048188A1 (en) | 2019-09-11 | 2021-03-18 | Bayer Aktiengesellschaft | Highly effective formulations on the basis of 2-[(2,4-dichlorphenyl)-methyl]-4,4'-dimethyl-3-isoxazolidinones and preemergence herbicides |
| WO2021058659A1 (en) | 2019-09-26 | 2021-04-01 | Bayer Aktiengesellschaft | Rnai-mediated pest control |
| WO2021069575A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Animal Health Gmbh | Heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021069567A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069569A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021089673A1 (en) | 2019-11-07 | 2021-05-14 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2021097162A1 (en) | 2019-11-13 | 2021-05-20 | Bayer Cropscience Lp | Beneficial combinations with paenibacillus |
| WO2021099303A1 (en) | 2019-11-18 | 2021-05-27 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021105091A1 (en) | 2019-11-25 | 2021-06-03 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| WO2021122986A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Thienyloxazolones and analogues |
| EP3845304A1 (en) | 2019-12-30 | 2021-07-07 | Bayer AG | Capsule suspension concentrates based on polyisocyanates and biodegradable amine based cross-linker |
| EP3868207A1 (en) | 2020-02-24 | 2021-08-25 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved activity in soil and leaf applications |
| WO2021165195A1 (en) | 2020-02-18 | 2021-08-26 | Bayer Aktiengesellschaft | Heteroaryl-triazole compounds as pesticides |
| WO2021204930A1 (en) | 2020-04-09 | 2021-10-14 | Bayer Animal Health Gmbh | Substituted condensed azines as anthelmintic compounds |
| EP3896065A1 (en) | 2015-08-07 | 2021-10-20 | Bayer CropScience Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pesticides |
| WO2021209363A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209365A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209490A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Cyclaminephenylaminoquinolines as fungicides |
| WO2021209364A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209368A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209366A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021213978A1 (en) | 2020-04-21 | 2021-10-28 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocyclic derivatives as pest control agents |
| WO2021224220A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Pyridine (thio)amides as fungicidal compounds |
| WO2021224323A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021228734A1 (en) | 2020-05-12 | 2021-11-18 | Bayer Aktiengesellschaft | Triazine and pyrimidine (thio)amides as fungicidal compounds |
| WO2021233861A1 (en) | 2020-05-19 | 2021-11-25 | Bayer Aktiengesellschaft | Azabicyclic(thio)amides as fungicidal compounds |
| EP3915971A1 (en) | 2020-12-16 | 2021-12-01 | Bayer Aktiengesellschaft | Phenyl-s(o)n-phenylamidines and the use thereof as fungicides |
| EP3915371A1 (en) | 2020-11-04 | 2021-12-01 | Bayer AG | Active compound combinations and fungicide compositions comprising those |
| WO2021245087A1 (en) | 2020-06-04 | 2021-12-09 | Bayer Aktiengesellschaft | Heterocyclyl pyrimidines and triazines as novel fungicides |
| WO2021249995A1 (en) | 2020-06-10 | 2021-12-16 | Bayer Aktiengesellschaft | Azabicyclyl-substituted heterocycles as fungicides |
| WO2021255071A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | 3-(pyridazin-4-yl)-5,6-dihydro-4h-1,2,4-oxadiazine derivatives as fungicides for crop protection |
| WO2021255170A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255169A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255089A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines and 1,3,4-oxadiazole pyridines as fungicides |
| WO2021255091A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as fungicides |
| EP3929189A1 (en) | 2020-06-25 | 2021-12-29 | Bayer Animal Health GmbH | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021260017A1 (en) | 2020-06-26 | 2021-12-30 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates comprising biodegradable ester groups |
| WO2022002818A1 (en) | 2020-07-02 | 2022-01-06 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2022033991A1 (en) | 2020-08-13 | 2022-02-17 | Bayer Aktiengesellschaft | 5-amino substituted triazoles as pest control agents |
| WO2022053453A1 (en) | 2020-09-09 | 2022-03-17 | Bayer Aktiengesellschaft | Azole carboxamide as pest control agents |
| WO2022058327A1 (en) | 2020-09-15 | 2022-03-24 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| EP3974414A1 (en) | 2020-09-25 | 2022-03-30 | Bayer AG | 5-amino substituted pyrazoles and triazoles as pesticides |
| EP3994995A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994986A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| EP3994992A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| EP3994991A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| EP3994989A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, rainfastness and uptake properties |
| EP3994988A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and rainfastness properties |
| EP3994990A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994985A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| EP3994993A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| EP3994994A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| WO2022096687A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and uptake properties |
| WO2022129188A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | 1,2,4-oxadiazol-3-yl pyrimidines as fungicides |
| WO2022129196A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | Heterobicycle substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129190A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | (hetero)aryl substituted 1,2,4-oxadiazoles as fungicides |
| WO2022152728A1 (en) | 2021-01-15 | 2022-07-21 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP4036083A1 (en) | 2021-02-02 | 2022-08-03 | Bayer Aktiengesellschaft | 5-oxy substituted heterocycles as pesticides |
| WO2022162129A1 (en) | 2021-01-28 | 2022-08-04 | Rhodia Operations | Method for treating rice seed with improved retention of agrochemical, micronutrient and colorant |
| WO2022207496A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022207494A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022233777A1 (en) | 2021-05-06 | 2022-11-10 | Bayer Aktiengesellschaft | Alkylamide substituted, annulated imidazoles and use thereof as insecticides |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2022238391A1 (en) | 2021-05-12 | 2022-11-17 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pest control agents |
| WO2023017120A1 (en) | 2021-08-13 | 2023-02-16 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2023025682A1 (en) | 2021-08-25 | 2023-03-02 | Bayer Aktiengesellschaft | Novel pyrazinyl-triazole compounds as pesticides |
| EP4144739A1 (en) | 2021-09-02 | 2023-03-08 | Bayer Aktiengesellschaft | Anellated pyrazoles as parasiticides |
| EP4148052A1 (en) | 2021-09-09 | 2023-03-15 | Bayer Animal Health GmbH | New quinoline derivatives |
| WO2023078915A1 (en) | 2021-11-03 | 2023-05-11 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether (thio)amides as fungicidal compounds |
| WO2023092050A1 (en) | 2021-11-20 | 2023-05-25 | Bayer Cropscience Lp | Beneficial combinations with recombinant bacillus cells expressing a serine protease |
| WO2023099445A1 (en) | 2021-11-30 | 2023-06-08 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether oxadiazines as fungicidal compounds |
| WO2023110656A1 (en) | 2021-12-15 | 2023-06-22 | Bayer Aktiengesellschaft | Spectroscopic solution for non-destructive quantification of one or more chemical substances in a matrix comprising coating and bulk material in a sample, such as coated seeds, using multivariate data analysis |
| EP4265110A1 (en) | 2022-04-20 | 2023-10-25 | Bayer AG | Water dispersible granules with low melting active ingredients prepared by extrusion |
| WO2023205602A1 (en) | 2022-04-18 | 2023-10-26 | Basf Corporation | High-load agricultural formulations and methods of making same |
| WO2023213626A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Use of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine for controlling unwanted microorganisms |
| WO2023213670A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Crystalline forms of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine |
| WO2023217619A1 (en) | 2022-05-07 | 2023-11-16 | Bayer Aktiengesellschaft | Low drift aqueous liquid formulations for low, medium, and high spray volume application |
| WO2023237444A1 (en) | 2022-06-06 | 2023-12-14 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295683A1 (en) | 2022-06-21 | 2023-12-27 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295688A1 (en) | 2022-09-28 | 2023-12-27 | Bayer Aktiengesellschaft | Active compound combination |
| WO2024013016A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024013015A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024068517A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068520A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068518A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-heteroaryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068473A1 (en) | 2022-09-27 | 2024-04-04 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2024068519A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| EP4353082A1 (en) | 2022-10-14 | 2024-04-17 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024104643A1 (en) | 2022-11-17 | 2024-05-23 | Bayer Aktiengesellschaft | Use of isotianil for controlling plasmodiophora brassica |
| WO2024155864A1 (en) * | 2023-01-20 | 2024-07-25 | Allianthera (Suzhou) Biopharmaceutical Co., Ltd. | Sprk1 inhibitors and methods of use |
| WO2024170472A1 (en) | 2023-02-16 | 2024-08-22 | Bayer Aktiengesellschaft | Herbicidal mixtures |
| WO2024213752A1 (en) | 2023-04-14 | 2024-10-17 | Elanco Animal Health Gmbh | Long-term prevention and/or treatment of a disease by slo-1 inhibitors |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EA024939B1 (en) * | 2012-02-21 | 2016-11-30 | Ацьенде Кимике Рьюните Анджелини Франческо А.К.Р.А.Ф. С.П.А. | 1h-indazole-3-carboxamide compounds as glycogen synthase kinase 3 beta inhibitors |
| JPWO2015068719A1 (en) * | 2013-11-07 | 2017-03-09 | 日本曹達株式会社 | Pyridine compounds and pest control agents |
| AU2015218402B2 (en) | 2014-02-14 | 2019-07-11 | Takeda Pharmaceutical Company Limited | Pyrazines modulators of GPR6 |
| CN107987012A (en) * | 2018-01-29 | 2018-05-04 | 田元强 | A kind of 4- benzyl piepridines class amide compound and its application in plant nematode diseases are prevented |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL253320A (en) * | 1959-07-03 | |||
| FR1365661A (en) | 1963-07-24 | 1964-07-03 | Sandoz Sa | New pyrazole derivatives and their preparation |
| NL286158A (en) | 1962-12-03 | |||
| GB9100505D0 (en) * | 1991-01-10 | 1991-02-20 | Shell Int Research | Piperidine derivatives |
| US6977264B2 (en) | 2001-07-25 | 2005-12-20 | Amgen Inc. | Substituted piperidines and methods of use |
| GB0808888D0 (en) * | 2008-05-15 | 2008-06-25 | Syngenta Participations Ag | Insecticidal compounds |
| GB0813436D0 (en) * | 2008-07-22 | 2008-08-27 | Syngenta Participations Ag | Insecticidal compounds |
| CA2765012A1 (en) * | 2009-07-06 | 2011-01-13 | Syngenta Participations Ag | Insecticidal compounds |
-
2004
- 2004-06-28 GB GBGB0414438.2A patent/GB0414438D0/en not_active Ceased
-
2005
- 2005-06-14 TW TW094119610A patent/TWI379636B/en not_active IP Right Cessation
- 2005-06-22 MX MXPA06014005A patent/MXPA06014005A/en active IP Right Grant
- 2005-06-22 KR KR1020127019520A patent/KR101268288B1/en not_active IP Right Cessation
- 2005-06-22 PL PL05757532T patent/PL1763302T3/en unknown
- 2005-06-22 US US11/571,303 patent/US8129534B2/en active Active
- 2005-06-22 ES ES05757532T patent/ES2408856T3/en active Active
- 2005-06-22 AU AU2005258905A patent/AU2005258905B2/en not_active Ceased
- 2005-06-22 CN CN201310146631XA patent/CN103214460A/en active Pending
- 2005-06-22 EA EA200602170A patent/EA014686B1/en not_active IP Right Cessation
- 2005-06-22 CN CNA2005800218477A patent/CN1976584A/en active Pending
- 2005-06-22 KR KR1020137008545A patent/KR101396174B1/en not_active IP Right Cessation
- 2005-06-22 WO PCT/IB2005/002002 patent/WO2006003494A2/en active Application Filing
- 2005-06-22 PT PT57575326T patent/PT1763302E/en unknown
- 2005-06-22 CA CA2568808A patent/CA2568808C/en not_active Expired - Fee Related
- 2005-06-22 JP JP2007517523A patent/JP5043653B2/en not_active Expired - Fee Related
- 2005-06-22 EP EP05757532A patent/EP1763302B1/en active Active
- 2005-06-22 AP AP2006003830A patent/AP1970A/en active
- 2005-06-22 NZ NZ551629A patent/NZ551629A/en not_active IP Right Cessation
- 2005-06-22 UA UAA200700618A patent/UA89788C2/en unknown
- 2005-06-22 AP AP2008004646A patent/AP2008004646A0/en unknown
- 2005-06-22 KR KR1020067027660A patent/KR101338876B1/en not_active IP Right Cessation
- 2005-06-22 BR BRPI0512659-2A patent/BRPI0512659A/en active Search and Examination
- 2005-06-24 AR ARP050102615A patent/AR049556A1/en unknown
-
2006
- 2006-11-21 ZA ZA200609687A patent/ZA200609687B/en unknown
- 2006-11-30 IL IL179745A patent/IL179745A/en not_active IP Right Cessation
- 2006-12-05 CR CR8790A patent/CR8790A/en not_active Application Discontinuation
- 2006-12-25 MA MA29552A patent/MA28678B1/en unknown
- 2006-12-26 EC EC2006007112A patent/ECSP067112A/en unknown
- 2006-12-27 TN TNP2006000442A patent/TNSN06442A1/en unknown
-
2012
- 2012-02-10 US US13/371,002 patent/US8546569B2/en not_active Expired - Fee Related
-
2013
- 2013-07-24 US US13/949,803 patent/US9045422B2/en not_active Expired - Fee Related
Non-Patent Citations (1)
| Title |
|---|
| None |
Cited By (378)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007072143A3 (en) * | 2005-12-21 | 2007-12-06 | Syngenta Participations Ag | Chemical compounds |
| WO2007072143A2 (en) * | 2005-12-21 | 2007-06-28 | Syngenta Participations Ag | Chemical compounds |
| US8338443B2 (en) | 2005-12-21 | 2012-12-25 | Syngenta Crop Protection Llc | Chemical compounds |
| US8835426B2 (en) | 2007-02-26 | 2014-09-16 | Vitae Pharmaceuticals, Inc. | Cyclic urea and carbamate inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8329897B2 (en) | 2007-07-26 | 2012-12-11 | Vitae Pharmaceuticals, Inc. | Synthesis of inhibitors of 11β-hydroxysteroid dehydrogenase type 1 |
| US8575156B2 (en) | 2007-07-26 | 2013-11-05 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US9079861B2 (en) | 2007-11-07 | 2015-07-14 | Vitae Pharmaceuticals, Inc. | Cyclic urea inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8748444B2 (en) | 2007-12-11 | 2014-06-10 | Vitae Pharmaceuticals, Inc. | Cyclic urea inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8440658B2 (en) | 2007-12-11 | 2013-05-14 | Vitae Pharmaceuticals, Inc. | Cyclic urea inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8680281B2 (en) | 2008-01-07 | 2014-03-25 | Vitae Pharmaceuticals, Inc. | Lactam inhibitors of 11-β-hydroxysteroid dehydrogenase 1 |
| US8592409B2 (en) | 2008-01-24 | 2013-11-26 | Vitae Pharmaceuticals, Inc. | Cyclic carbazate and semicarbazide inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8202857B2 (en) | 2008-02-11 | 2012-06-19 | Vitae Pharmaceuticals, Inc. | 1,3-oxazepan-2-one and 1,3-diazepan-2-one inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8598160B2 (en) | 2008-02-15 | 2013-12-03 | Vitae Pharmaceuticals, Inc. | Cycloalkyl lactame derivatives as inhibitors of 11-beta-hydroxysteroid dehydrogenase 1 |
| US8592410B2 (en) | 2008-05-01 | 2013-11-26 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11BETA-hydroxysteroid dehydrogenase 1 |
| US8242111B2 (en) | 2008-05-01 | 2012-08-14 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8673899B2 (en) | 2008-05-01 | 2014-03-18 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8569292B2 (en) | 2008-05-01 | 2013-10-29 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8138178B2 (en) | 2008-05-01 | 2012-03-20 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| WO2009138219A3 (en) * | 2008-05-15 | 2010-01-21 | Syngenta Participations Ag | Insecticidal compounds |
| US8193362B2 (en) | 2008-05-15 | 2012-06-05 | Syngenta Crop Protection Llc | Insecticidal compounds |
| EA019508B1 (en) * | 2008-05-15 | 2014-04-30 | Зингента Партисипейшнс Аг | Insecticidal compounds |
| WO2009138219A2 (en) * | 2008-05-15 | 2009-11-19 | Syngenta Participations Ag | Insecticidal compounds |
| CN103396403B (en) * | 2008-05-15 | 2015-09-09 | 先正达参股股份有限公司 | Pesticidal compound |
| CN103396403A (en) * | 2008-05-15 | 2013-11-20 | 先正达参股股份有限公司 | Insecticidal compound |
| CN102105461A (en) * | 2008-07-22 | 2011-06-22 | 先正达参股股份有限公司 | Insecticidal phenyl- or pyridyl-piperidine compounds |
| US8518971B2 (en) | 2008-07-22 | 2013-08-27 | Syngenta Crop Protection, Inc. | Insecticidal phenyl- or pyridyl-piperdine compounds |
| US10034476B2 (en) | 2008-07-22 | 2018-07-31 | Syngenta Participations Ag | Insecticidal phenyl-or pyridyl-piperdine compounds |
| US9808006B2 (en) | 2008-07-22 | 2017-11-07 | Syngenta Participations Ag | Insecticidal phenyl-or pyridyl-piperdine compounds |
| WO2010009968A1 (en) * | 2008-07-22 | 2010-01-28 | Syngenta Participations Ag | Insecticidal phenyl- or pyridyl-piperidine compounds |
| AU2009273368B2 (en) * | 2008-07-22 | 2013-09-26 | Syngenta Participations Ag | Insecticidal phenyl- or pyridyl-piperidine compounds |
| EA017523B1 (en) * | 2008-07-22 | 2013-01-30 | Зингента Партисипейшнс Аг | Insecticidal phenyl- or pyridyl-piperidine compounds |
| CN102105461B (en) * | 2008-07-22 | 2013-07-10 | 先正达参股股份有限公司 | Insecticidal phenyl- or pyridyl-piperidine compounds |
| US8754076B2 (en) | 2008-07-25 | 2014-06-17 | Vitae Pharmaceuticals, Inc./Boehringer-Ingelheim | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8846668B2 (en) | 2008-07-25 | 2014-09-30 | Vitae Pharmaceuticals, Inc. | Inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8114868B2 (en) | 2008-07-25 | 2012-02-14 | Boehringer Ingelheim International Gmbh | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 |
| US8637505B2 (en) | 2009-02-04 | 2014-01-28 | Boehringer Ingelheim International Gmbh | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8680093B2 (en) | 2009-04-30 | 2014-03-25 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11beta-hydroxysteroid dehydrogenase 1 |
| US8927539B2 (en) | 2009-06-11 | 2015-01-06 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11β-hydroxysteroid dehydrogenase 1 based on the 1,3-oxazinan-2-one structure |
| US8883778B2 (en) | 2009-07-01 | 2014-11-11 | Vitae Pharmaceuticals, Inc. | Cyclic inhibitors of 11 beta-hydroxysteroid dehydrogenase 1 |
| WO2011003684A1 (en) * | 2009-07-06 | 2011-01-13 | Syngenta Participations Ag | Insecticidal compounds |
| AU2010270464B2 (en) * | 2009-07-06 | 2015-01-22 | Syngenta Participations Ag | Insecticidal compounds |
| EA019857B1 (en) * | 2009-07-06 | 2014-06-30 | Зингента Партисипейшнс Аг | Insecticidal compounds |
| US8586593B2 (en) | 2009-07-06 | 2013-11-19 | Syngenta Crop Protection, Inc. | Insecticidal compounds |
| US8933072B2 (en) | 2010-06-16 | 2015-01-13 | Vitae Pharmaceuticals, Inc. | Substituted 5-,6- and 7-membered heterocycles, medicaments containing such compounds, and their use |
| US9090605B2 (en) | 2010-06-16 | 2015-07-28 | Vitae Pharmaceuticals, Inc. | Substituted 5-,6- and 7-membered heterocycles, medicaments containing such compounds, and their use |
| US8765744B2 (en) | 2010-06-25 | 2014-07-01 | Boehringer Ingelheim International Gmbh | Azaspirohexanones |
| US8846613B2 (en) | 2010-11-02 | 2014-09-30 | Boehringer Ingelheim International Gmbh | Pharmaceutical combinations for the treatment of metabolic disorders |
| CN104302623A (en) * | 2012-05-01 | 2015-01-21 | 住友化学株式会社 | Piperidine compound and pest-control use therefore |
| WO2013164954A1 (en) * | 2012-05-01 | 2013-11-07 | 住友化学株式会社 | Piperidine compound and pest-control use therefore |
| WO2014005982A1 (en) | 2012-07-05 | 2014-01-09 | Bayer Cropscience Ag | Insecticide and fungicide active ingredient combinations |
| US9776967B2 (en) | 2013-10-14 | 2017-10-03 | Bayer Animal Health Gmbh | Carboxamide derivatives as pesticidal compounds |
| WO2015055554A1 (en) | 2013-10-14 | 2015-04-23 | Bayer Cropscience Ag | Active substance for treating seed and soil |
| WO2015059088A1 (en) | 2013-10-23 | 2015-04-30 | Bayer Cropscience Ag | Substituted quinoxaline derivatives as pest control agent |
| WO2015101622A1 (en) | 2014-01-03 | 2015-07-09 | Bayer Cropscience Ag | Novel pyrazolyl-heteroarylamides as pesticides |
| WO2015107133A1 (en) | 2014-01-20 | 2015-07-23 | Bayer Cropscience Ag | Quinoline derivatives as insecticides and acaricides |
| WO2015150300A1 (en) | 2014-04-02 | 2015-10-08 | Bayer Cropscience Ag | N-(1-(hetero)aryl-1h-pyrazol-4-yl)-(hetero)arylamide derivatives and use thereof as pesticides |
| WO2015169776A1 (en) | 2014-05-08 | 2015-11-12 | Bayer Cropscience Ag | Pyrazolopyridine sulfonamides as nematicides |
| WO2015185531A1 (en) | 2014-06-05 | 2015-12-10 | Bayer Cropscience Aktiengesellschaft | Bicyclic compounds as pesticides |
| WO2016001119A1 (en) | 2014-07-01 | 2016-01-07 | Bayer Cropscience Aktiengesellschaft | Insecticide and fungicide active ingredient combinations |
| WO2016008830A1 (en) | 2014-07-15 | 2016-01-21 | Bayer Cropscience Aktiengesellschaft | Aryl-triazolyl pyridines as pest control agents |
| WO2016055096A1 (en) | 2014-10-07 | 2016-04-14 | Bayer Cropscience Ag | Method for treating rice seed |
| WO2016091857A1 (en) | 2014-12-11 | 2016-06-16 | Bayer Cropscience Aktiengesellschaft | Five-membered c-n bonded aryl sulphide and aryl sulphoxide derivatives as pest control agents |
| WO2016124563A1 (en) | 2015-02-05 | 2016-08-11 | Bayer Cropscience Aktiengesellschsaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2016124557A1 (en) | 2015-02-05 | 2016-08-11 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2016128298A1 (en) | 2015-02-09 | 2016-08-18 | Bayer Cropscience Aktiengesellschaft | Substituted 2-thioimidazolyl-carboxamides as pest control agents |
| WO2016142394A1 (en) | 2015-03-10 | 2016-09-15 | Bayer Animal Health Gmbh | Pyrazolyl-derivatives as pest control agents |
| WO2016162318A1 (en) | 2015-04-08 | 2016-10-13 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents and intermediate product |
| EP3081085A1 (en) | 2015-04-14 | 2016-10-19 | Bayer CropScience AG | Method for improving earliness in cotton |
| WO2016174049A1 (en) | 2015-04-30 | 2016-11-03 | Bayer Animal Health Gmbh | Anti-parasitic combinations including halogen-substituted compounds |
| EP2910126A1 (en) | 2015-05-05 | 2015-08-26 | Bayer CropScience AG | Active compound combinations having insecticidal properties |
| WO2016180802A1 (en) | 2015-05-13 | 2016-11-17 | Bayer Cropscience Aktiengesellschaft | Insecticidal arylpyrrolidines, method for synthesizing same, and use thereof as agents for controlling animal pests |
| WO2017005717A1 (en) | 2015-07-06 | 2017-01-12 | Bayer Cropscience Aktiengesellschaft | Heterocyclic compounds as pesticides |
| EP3896065A1 (en) | 2015-08-07 | 2021-10-20 | Bayer CropScience Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pesticides |
| EP3896066A2 (en) | 2015-08-07 | 2021-10-20 | Bayer CropScience Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pesticides |
| WO2017072039A1 (en) | 2015-10-26 | 2017-05-04 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093180A1 (en) | 2015-12-01 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093214A1 (en) | 2015-12-03 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Mesoionic halogenated 3-(acetyl)-1-[(1,3-thiazol-5-yl)methyl]-1h-imidazo[1,2-a]pyridin-4-ium-2-olate derivatives and related compounds as insecticides |
| WO2017137338A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituierted 2-(het)aryl-imidazolyl-carboxyamides as pest control agents |
| WO2017137339A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituted 2-oxyimidazolyl-carboxamides as pest control agents |
| WO2017144341A1 (en) | 2016-02-23 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017144497A1 (en) | 2016-02-26 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Solvent-free formulations of low-melting active substances |
| EP3210468A1 (en) | 2016-02-26 | 2017-08-30 | Bayer CropScience Aktiengesellschaft | Solvent-free formulations of low-melting point agents |
| WO2017157735A1 (en) | 2016-03-15 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2017157885A1 (en) | 2016-03-16 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | N-(cyanobenzyl)-6-(cyclopropyl-carbonylamino)-4-(phenyl)-pyridine-2-carboxamide derivatives and related compounds as pesticides and plant protection agents |
| WO2017174414A1 (en) | 2016-04-05 | 2017-10-12 | Bayer Cropscience Aktiengesellschaft | Naphthaline-derivatives as pest control agents |
| WO2017178416A1 (en) | 2016-04-15 | 2017-10-19 | Bayer Animal Health Gmbh | Pyrazolopyrimidine derivatives |
| WO2017186536A1 (en) | 2016-04-25 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Substituted 2-alkylimidazolyl-carboxamides as pest control agents |
| EP3241830A1 (en) | 2016-05-04 | 2017-11-08 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pesticides |
| WO2017198449A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in brassicaceae |
| WO2017198450A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in maize |
| WO2017198452A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in soybean |
| WO2017198453A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in potato, tomato or alfalfa |
| WO2017198451A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in small grain cereals such as wheat and rice |
| WO2017198455A2 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in beta spp. plants |
| WO2017198454A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in cotton |
| EP3245865A1 (en) | 2016-05-17 | 2017-11-22 | Bayer CropScience Aktiengesellschaft | Method for increasing yield in brassicaceae |
| WO2018013382A1 (en) | 2016-07-11 | 2018-01-18 | Covestro Llc, Et Al | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| US10653135B2 (en) | 2016-07-11 | 2020-05-19 | Covestro Llc | Methods for treating seeds with an aqueous composition and seeds treated therewith |
| US10653136B2 (en) | 2016-07-11 | 2020-05-19 | Covestro Llc | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| US10750750B2 (en) | 2016-07-11 | 2020-08-25 | Covestro Llc | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| WO2018013381A1 (en) | 2016-07-11 | 2018-01-18 | Covestro Llc, Et Al. | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| WO2018013380A1 (en) | 2016-07-11 | 2018-01-18 | Covestro Llc, Et Al. | Methods for treating seeds with an aqueous compostion and seeds treated therewith |
| WO2018011111A1 (en) | 2016-07-12 | 2018-01-18 | Bayer Cropscience Aktiengesellschaft | Bicyclic compounds as pest control agents |
| WO2018015289A1 (en) | 2016-07-19 | 2018-01-25 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018019937A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Formulation comprising a beneficial p. bilaii strain and talc for use in seed treatment |
| WO2018029102A1 (en) | 2016-08-10 | 2018-02-15 | Bayer Cropscience Aktiengesellschaft | Substituted 2-heterocyclyl imidazolyl-carboxamides as pest control agents |
| WO2018033455A1 (en) | 2016-08-15 | 2018-02-22 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018050825A1 (en) | 2016-09-19 | 2018-03-22 | Bayer Cropscience Aktiengesellschaft | Pyrazolo [1,5-a]pyridine derivatives and their use as pesticides |
| WO2018065292A1 (en) | 2016-10-06 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018065288A1 (en) | 2016-10-07 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-[2-phenyl-1-(sulfonyl-methyl)-vinyl]-imidazo-[4,5-b] pyridine derivatives and related compounds as pesticides in plant protection |
| WO2018069841A1 (en) | 2016-10-14 | 2018-04-19 | Pi Industries Ltd | 4-substituted phenylamine derivatives and their use to protect crops by fighting undesired phytopathogenic micoorganisms |
| US11155517B2 (en) | 2016-10-14 | 2021-10-26 | Pi Industries Ltd. | 4-substituted phenylamine derivatives and their use to protect crops by fighting undesired phytopathogenic micoorganisms |
| WO2018069842A1 (en) | 2016-10-14 | 2018-04-19 | Pi Industries Ltd | 4-amino substituted phenylamidine derivatives and their use to protect crops by fighting undesired phytopathogenic micoorganisms |
| WO2018083288A1 (en) | 2016-11-07 | 2018-05-11 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2018087036A1 (en) | 2016-11-11 | 2018-05-17 | Bayer Animal Health Gmbh | New anthelmintic quinoline-3-carboxamide derivatives |
| US11505545B2 (en) | 2016-11-11 | 2022-11-22 | Bayer Animal Health Gmbh | Anthelmintic quinoline-3-carboxamide derivatives |
| US10889573B2 (en) | 2016-11-11 | 2021-01-12 | Bayer Animal Health Gmbh | Anthelmintic quinoline-3-carboxamide derivatives |
| WO2018095953A1 (en) | 2016-11-23 | 2018-05-31 | Bayer Cropscience Aktiengesellschaft | 2-[3-(alkylsulfonyl)-2h-indazol-2-yl]-3h-imidazo[4,5-b]pyridine derivatives and similar compounds as pesticides |
| WO2018104500A1 (en) | 2016-12-09 | 2018-06-14 | Bayer Cropscience Aktiengesellschaft | Plant health effect of purpureocillium lilacinum |
| WO2018108730A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018108791A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Thiadiazole derivatives as pesticides |
| WO2018116072A1 (en) | 2016-12-20 | 2018-06-28 | Pi Industries Ltd. | Heterocyclic compounds |
| WO2018116073A1 (en) | 2016-12-21 | 2018-06-28 | Pi Industries Ltd. | 1, 2, 3-thiadiazole compounds and their use as crop protecting agent |
| WO2018130437A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018130443A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018138050A1 (en) | 2017-01-26 | 2018-08-02 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclene derivatives as pest control agents |
| WO2018141954A1 (en) | 2017-02-06 | 2018-08-09 | Bayer Aktiengesellschaft | Aryl or heteroaryl-substituted imidazo pyridine derivatives and their use as pesticides |
| EP3369320A1 (en) | 2017-03-02 | 2018-09-05 | Bayer CropScience Aktiengesellschaft | Agent for controlling bugs |
| WO2018177993A1 (en) | 2017-03-31 | 2018-10-04 | Bayer Cropscience Aktiengesellschaft | Pyrazoles for controlling arthropods |
| WO2018177995A1 (en) | 2017-03-31 | 2018-10-04 | Bayer Cropscience Aktiengesellschaft | Tricyclic carboxamides for controlling arthropods |
| WO2018189077A1 (en) | 2017-04-12 | 2018-10-18 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| US11524934B2 (en) | 2017-04-20 | 2022-12-13 | Pi Industries Ltd | Phenylamine compounds |
| WO2018193385A1 (en) | 2017-04-20 | 2018-10-25 | Pi Industries Ltd. | Novel phenylamine compounds |
| WO2018192872A1 (en) | 2017-04-21 | 2018-10-25 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2018197257A1 (en) | 2017-04-24 | 2018-11-01 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic-compound derivatives as pest control agents |
| WO2018197401A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Animal Health Gmbh | New bicyclic pyrazole derivatives |
| US11130768B2 (en) | 2017-04-27 | 2021-09-28 | Bayer Animal Health Gmbh | Bicyclic pyrazole derivatives |
| WO2018197692A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Aktiengesellschaft | Heteroarylphenylaminoquinolines and analogues |
| WO2018202501A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202494A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202715A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylbenzylbenzimidazoles and analogues |
| WO2018202706A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylheteroaryloxyquinolines and analogues |
| WO2018202712A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylphenoxyquinolines and analogues |
| WO2018202525A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | Phenoxyethanamine derivatives for controlling pests |
| US11827616B2 (en) | 2017-05-04 | 2023-11-28 | Discovery Purchaser Corporation | Heterocyclic compounds as pesticides |
| WO2018202524A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | 2-{[2-(phenyloxymethyl)pyridin-5-yl]oxy}-ethanamin-derivatives and related compounds as pest-control agents e.g. for the protection of plants |
| EP3400801A1 (en) | 2017-05-10 | 2018-11-14 | Bayer CropScience Aktiengesellschaft | Plant health effect of purpureocillium lilacinum |
| WO2019002132A1 (en) | 2017-06-30 | 2019-01-03 | Bayer Animal Health Gmbh | New azaquinoline derivatives |
| WO2019007888A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insecticidal active ingredient combinations |
| WO2019007887A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insecticide and fungicide active ingredient combinations |
| WO2019007891A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insecticidal active ingredient combinations |
| EP3284739A1 (en) | 2017-07-19 | 2018-02-21 | Bayer CropScience Aktiengesellschaft | Substituted (het) aryl compounds as pesticides |
| WO2019025341A1 (en) | 2017-08-04 | 2019-02-07 | Bayer Animal Health Gmbh | Quinoline derivatives for treating infections with helminths |
| WO2019035881A1 (en) | 2017-08-17 | 2019-02-21 | Bayer Cropscience Lp | Liquid fertilizer-dispersible compositions and methods thereof |
| WO2019038195A1 (en) | 2017-08-22 | 2019-02-28 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019059412A1 (en) | 2017-09-20 | 2019-03-28 | Mitsui Chemicals Agro, Inc. | Prolonged ectoparasite-controlling agent for animal |
| WO2019068572A1 (en) | 2017-10-04 | 2019-04-11 | Bayer Aktiengesellschaft | Derivatives of heterocyclic compounds as pest control agents |
| WO2019076744A1 (en) | 2017-10-17 | 2019-04-25 | Bayer Aktiengesellschaft | Aqueous [2-(2,4-dichlorophenyl)methyl]-4,4-dimethyl-3-isoxazolidinone-based suspension concentrates |
| EP3473103A1 (en) | 2017-10-17 | 2019-04-24 | Bayer AG | Aqueous suspension concentrates based on 2- [(2,4-dichlorophenyl) -methyl] -4,4 '-dimethyl-3-isoxazolidinone |
| WO2019076752A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076750A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076751A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076754A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076749A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3473100A1 (en) | 2017-10-18 | 2019-04-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019092086A1 (en) | 2017-11-13 | 2019-05-16 | Bayer Aktiengesellschaft | Tetrazolylpropyl derivatives and their use as fungicides |
| WO2019105875A1 (en) | 2017-11-28 | 2019-06-06 | Bayer Aktiengesellschaft | Heterocyclic compounds as pesticides |
| WO2019105871A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Aktiengesellschaft | Nitrogenous heterocycles as a pesticide |
| WO2019123196A1 (en) | 2017-12-20 | 2019-06-27 | Pi Industries Ltd. | Fluoralkenyl compounds, process for preparation and use thereof |
| WO2019122319A1 (en) | 2017-12-21 | 2019-06-27 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylheteroaryloxyquinolines and analogues |
| EP3305786A2 (en) | 2018-01-22 | 2018-04-11 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pesticides |
| WO2019150311A1 (en) | 2018-02-02 | 2019-08-08 | Pi Industries Ltd. | 1-3 dithiol compounds and their use for the protection of crops from phytopathogenic microorganisms |
| WO2019155066A1 (en) | 2018-02-12 | 2019-08-15 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2019162174A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019162228A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | 1-(5-substituted imidazol-1-yl)but-3-en derivatives and their use as fungicides |
| WO2019170626A1 (en) | 2018-03-08 | 2019-09-12 | Bayer Aktiengesellschaft | Use of heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides in plant protection |
| WO2019175046A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019175045A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019197371A1 (en) | 2018-04-10 | 2019-10-17 | Bayer Aktiengesellschaft | Oxadiazoline derivatives |
| EP3904349A2 (en) | 2018-04-12 | 2021-11-03 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}heterocyclyl amide derivatives and similar compounds as pesticides |
| EP3904350A1 (en) | 2018-04-12 | 2021-11-03 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| WO2019197468A1 (en) | 2018-04-12 | 2019-10-17 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| WO2019197623A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, fungicidal and acaricidal properties |
| WO2019197615A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with fungicides, insecticides and acaricidal properties |
| WO2019201835A1 (en) | 2018-04-17 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| EP4039682A1 (en) | 2018-04-17 | 2022-08-10 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019202077A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019201921A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| EP3919486A1 (en) | 2018-04-25 | 2021-12-08 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019206799A1 (en) | 2018-04-25 | 2019-10-31 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019215182A1 (en) | 2018-05-09 | 2019-11-14 | Bayer Animal Health Gmbh | New quinoline derivatives |
| WO2019224143A1 (en) | 2018-05-24 | 2019-11-28 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, nematicidal and acaricidal properties |
| WO2020005678A1 (en) | 2018-06-25 | 2020-01-02 | Bayer Cropscience Lp | Seed treatment method |
| WO2020002189A1 (en) | 2018-06-27 | 2020-01-02 | Bayer Aktiengesellschaft | Active substance combinations |
| EP3586630A1 (en) | 2018-06-28 | 2020-01-01 | Bayer AG | Active compound combinations having insecticidal/acaricidal properties |
| WO2020007905A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020007904A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020007902A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| US11884643B2 (en) | 2018-07-05 | 2024-01-30 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| US11952359B2 (en) | 2018-07-05 | 2024-04-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020020813A1 (en) | 2018-07-25 | 2020-01-30 | Bayer Aktiengesellschaft | Fungicidal active compound combinations |
| WO2020020816A1 (en) | 2018-07-26 | 2020-01-30 | Bayer Aktiengesellschaft | Novel triazole derivatives |
| WO2020021082A1 (en) | 2018-07-27 | 2020-01-30 | Bayer Aktiengesellschaft | Controlled release formulations for agrochemicals |
| WO2020025650A1 (en) | 2018-07-31 | 2020-02-06 | Bayer Aktiengesellschaft | Controlled release formulations with lignin for agrochemicals |
| WO2020035826A1 (en) | 2018-08-17 | 2020-02-20 | Pi Industries Ltd. | 1,2-dithiolone compounds and use thereof |
| WO2020043650A1 (en) | 2018-08-29 | 2020-03-05 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020053282A1 (en) | 2018-09-13 | 2020-03-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2020057939A1 (en) | 2018-09-17 | 2020-03-26 | Bayer Aktiengesellschaft | Use of the fungicide isoflucypram for controlling claviceps purpurea and reducing sclerotia in cereals |
| WO2020070050A1 (en) | 2018-10-01 | 2020-04-09 | Bayer Aktiengesellschaft | Fungicidal 5-substituted imidazol-1-yl carbinol derivatives |
| EP3636644A1 (en) | 2018-10-11 | 2020-04-15 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2020078839A1 (en) | 2018-10-16 | 2020-04-23 | Bayer Aktiengesellschaft | Active substance combinations |
| WO2020079173A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Pyridylphenylaminoquinolines and analogues |
| WO2020079167A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Heteroarylaminoquinolines and analogues |
| WO2020079232A1 (en) | 2018-10-20 | 2020-04-23 | Bayer Aktiengesellschaft | Oxetanylphenoxyquinolines and analogues |
| EP3643711A1 (en) | 2018-10-24 | 2020-04-29 | Bayer Animal Health GmbH | New anthelmintic compounds |
| WO2020083971A2 (en) | 2018-10-24 | 2020-04-30 | Bayer Animal Health Gmbh | New anthelmintic compounds |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020114932A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2020114934A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicide compositions |
| EP3620052A1 (en) | 2018-12-12 | 2020-03-11 | Bayer Aktiengesellschaft | Use of phenoxypyridinyl-substituted (1h-1,2,4-triazol-1-yl)alcohols for controlling fungicidal diseases in maize |
| WO2020126980A1 (en) | 2018-12-18 | 2020-06-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| WO2020127974A1 (en) | 2018-12-21 | 2020-06-25 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as new antifungal agents |
| EP3669652A1 (en) | 2018-12-21 | 2020-06-24 | Bayer AG | Active compound combination |
| EP3679791A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679792A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679793A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679790A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679789A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3545764A1 (en) | 2019-02-12 | 2019-10-02 | Bayer AG | Crystal form of 2-({2-fluoro-4-methyl-5-[(r)-(2,2,2-trifluoroethyl)sulfinyl]phenyl}imino)-3-(2,2,2- trifluoroethyl)-1,3-thiazolidin-4-one |
| WO2020173860A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Fused bicyclic heterocycle derivatives as pesticides |
| WO2020173861A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2020178067A1 (en) | 2019-03-01 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020178307A1 (en) | 2019-03-05 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020182929A1 (en) | 2019-03-13 | 2020-09-17 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| WO2020187656A1 (en) | 2019-03-15 | 2020-09-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3564225A1 (en) | 2019-03-21 | 2019-11-06 | Bayer Aktiengesellschaft | Crystalline form of spiromesifen |
| EP3725788A1 (en) | 2019-04-15 | 2020-10-21 | Bayer AG | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020212235A1 (en) | 2019-04-15 | 2020-10-22 | Bayer Animal Health Gmbh | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020225428A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading ulv formulations for insecticides |
| WO2020225438A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High uptake and rainfastness ulv formulations |
| WO2020225434A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading ulv formulations for agrochemical compounds ii |
| WO2020225440A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading and rainfastness ulv formulations |
| WO2020225242A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020225437A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Ulv formulations with enhanced uptake |
| WO2020225439A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Ulv formulations with enhanced rainfastness |
| WO2020225435A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading and uptake ulv formulations |
| WO2020225436A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading, uptake and rainfastness ulv formulations |
| WO2020231751A1 (en) | 2019-05-10 | 2020-11-19 | Bayer Cropscience Lp | Active compound combinations |
| WO2020229398A1 (en) | 2019-05-14 | 2020-11-19 | Bayer Aktiengesellschaft | (1-alkenyl)-substituted pyrazoles and triazoles as pest control agents |
| EP3750888A1 (en) | 2019-06-12 | 2020-12-16 | Bayer Aktiengesellschaft | Crystalline form a of 1,4-dimethyl-2-[2-(pyridin-3-yl)-2h-indazol-5-yl]-1,2,4-triazolidine-3,5-dione |
| WO2020254493A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Thienylhydroxyisoxazolines and derivatives thereof |
| WO2020254487A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254489A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Benzylphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254492A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254488A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and use thereof as fungicides |
| WO2020254494A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2020254490A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Phenoxyphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254486A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020263812A1 (en) | 2019-06-24 | 2020-12-30 | Auburn University | A bacillus strain and methods of its use for plant growth promotion |
| EP3608311A1 (en) | 2019-06-28 | 2020-02-12 | Bayer AG | Crystalline form a of n-[4-chloro-3-[(1-cyanocyclopropyl)carbamoyl]phenyl]-2-methyl-4-methylsulfonyl-5-(1,1,2,2,2-pentafluoroethyl)pyrazole-3-carboxamide |
| WO2021001331A1 (en) | 2019-07-03 | 2021-01-07 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides and derivatives thereof as microbicides |
| WO2021001273A1 (en) | 2019-07-04 | 2021-01-07 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP4378314A2 (en) | 2019-07-04 | 2024-06-05 | Bayer AG | Herbicidal compositions |
| WO2021013721A1 (en) | 2019-07-22 | 2021-01-28 | Bayer Aktiengesellschaft | 5-amino substituted pyrazoles and triazoles as pest control agents |
| WO2021013719A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013720A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021018839A1 (en) | 2019-07-30 | 2021-02-04 | Bayer Animal Health Gmbh | Isoquinoline derivatives and their use for the treatment of parasitic infections |
| EP3771714A1 (en) | 2019-07-30 | 2021-02-03 | Bayer AG | Nitrogen-containing heterocycles as pesticides |
| EP3701796A1 (en) | 2019-08-08 | 2020-09-02 | Bayer AG | Active compound combinations |
| WO2021048188A1 (en) | 2019-09-11 | 2021-03-18 | Bayer Aktiengesellschaft | Highly effective formulations on the basis of 2-[(2,4-dichlorphenyl)-methyl]-4,4'-dimethyl-3-isoxazolidinones and preemergence herbicides |
| WO2021058659A1 (en) | 2019-09-26 | 2021-04-01 | Bayer Aktiengesellschaft | Rnai-mediated pest control |
| WO2021069567A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069569A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069575A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Animal Health Gmbh | Heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021089673A1 (en) | 2019-11-07 | 2021-05-14 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2021097162A1 (en) | 2019-11-13 | 2021-05-20 | Bayer Cropscience Lp | Beneficial combinations with paenibacillus |
| WO2021099303A1 (en) | 2019-11-18 | 2021-05-27 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021105091A1 (en) | 2019-11-25 | 2021-06-03 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021122986A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Thienyloxazolones and analogues |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| WO2021136758A1 (en) | 2019-12-30 | 2021-07-08 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates based on polyurea shell material containing polyfunctional aminocarboxylic esters |
| EP3845304A1 (en) | 2019-12-30 | 2021-07-07 | Bayer AG | Capsule suspension concentrates based on polyisocyanates and biodegradable amine based cross-linker |
| WO2021165195A1 (en) | 2020-02-18 | 2021-08-26 | Bayer Aktiengesellschaft | Heteroaryl-triazole compounds as pesticides |
| WO2021170527A1 (en) | 2020-02-24 | 2021-09-02 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved effictiveness in soil and leaf applications |
| EP3868207A1 (en) | 2020-02-24 | 2021-08-25 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved activity in soil and leaf applications |
| EP3708565A1 (en) | 2020-03-04 | 2020-09-16 | Bayer AG | Pyrimidinyloxyphenylamidines and the use thereof as fungicides |
| WO2021204930A1 (en) | 2020-04-09 | 2021-10-14 | Bayer Animal Health Gmbh | Substituted condensed azines as anthelmintic compounds |
| WO2021209490A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Cyclaminephenylaminoquinolines as fungicides |
| WO2021209366A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209368A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209364A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209365A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209363A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021213978A1 (en) | 2020-04-21 | 2021-10-28 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocyclic derivatives as pest control agents |
| WO2021224323A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021224220A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Pyridine (thio)amides as fungicidal compounds |
| WO2021228734A1 (en) | 2020-05-12 | 2021-11-18 | Bayer Aktiengesellschaft | Triazine and pyrimidine (thio)amides as fungicidal compounds |
| WO2021233861A1 (en) | 2020-05-19 | 2021-11-25 | Bayer Aktiengesellschaft | Azabicyclic(thio)amides as fungicidal compounds |
| WO2021245087A1 (en) | 2020-06-04 | 2021-12-09 | Bayer Aktiengesellschaft | Heterocyclyl pyrimidines and triazines as novel fungicides |
| WO2021249995A1 (en) | 2020-06-10 | 2021-12-16 | Bayer Aktiengesellschaft | Azabicyclyl-substituted heterocycles as fungicides |
| WO2021255071A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | 3-(pyridazin-4-yl)-5,6-dihydro-4h-1,2,4-oxadiazine derivatives as fungicides for crop protection |
| WO2021255089A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines and 1,3,4-oxadiazole pyridines as fungicides |
| WO2021255091A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as fungicides |
| WO2021255169A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255170A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| EP3929189A1 (en) | 2020-06-25 | 2021-12-29 | Bayer Animal Health GmbH | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021259997A1 (en) | 2020-06-25 | 2021-12-30 | Bayer Animal Health Gmbh | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021260017A1 (en) | 2020-06-26 | 2021-12-30 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates comprising biodegradable ester groups |
| WO2022002818A1 (en) | 2020-07-02 | 2022-01-06 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2022033991A1 (en) | 2020-08-13 | 2022-02-17 | Bayer Aktiengesellschaft | 5-amino substituted triazoles as pest control agents |
| WO2022053453A1 (en) | 2020-09-09 | 2022-03-17 | Bayer Aktiengesellschaft | Azole carboxamide as pest control agents |
| WO2022058327A1 (en) | 2020-09-15 | 2022-03-24 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| EP3974414A1 (en) | 2020-09-25 | 2022-03-30 | Bayer AG | 5-amino substituted pyrazoles and triazoles as pesticides |
| EP3915371A1 (en) | 2020-11-04 | 2021-12-01 | Bayer AG | Active compound combinations and fungicide compositions comprising those |
| EP3994985A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| WO2022096690A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994988A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and rainfastness properties |
| EP3994990A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994991A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| EP3994993A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| EP3994994A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| WO2022096695A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| WO2022096688A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading and rainfastness properties |
| WO2022096692A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| WO2022096685A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| WO2022096686A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| WO2022096691A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, uptake and rainfastness properties |
| EP3994989A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, rainfastness and uptake properties |
| WO2022096687A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and uptake properties |
| WO2022096693A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| WO2022096694A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| WO2022096696A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994995A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994986A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| EP3994992A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| EP3915971A1 (en) | 2020-12-16 | 2021-12-01 | Bayer Aktiengesellschaft | Phenyl-s(o)n-phenylamidines and the use thereof as fungicides |
| WO2022129190A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | (hetero)aryl substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129196A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | Heterobicycle substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129188A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | 1,2,4-oxadiazol-3-yl pyrimidines as fungicides |
| WO2022152728A1 (en) | 2021-01-15 | 2022-07-21 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2022162129A1 (en) | 2021-01-28 | 2022-08-04 | Rhodia Operations | Method for treating rice seed with improved retention of agrochemical, micronutrient and colorant |
| EP4036083A1 (en) | 2021-02-02 | 2022-08-03 | Bayer Aktiengesellschaft | 5-oxy substituted heterocycles as pesticides |
| WO2022207494A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022207496A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022233777A1 (en) | 2021-05-06 | 2022-11-10 | Bayer Aktiengesellschaft | Alkylamide substituted, annulated imidazoles and use thereof as insecticides |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2022238391A1 (en) | 2021-05-12 | 2022-11-17 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pest control agents |
| WO2023017120A1 (en) | 2021-08-13 | 2023-02-16 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2023025682A1 (en) | 2021-08-25 | 2023-03-02 | Bayer Aktiengesellschaft | Novel pyrazinyl-triazole compounds as pesticides |
| EP4144739A1 (en) | 2021-09-02 | 2023-03-08 | Bayer Aktiengesellschaft | Anellated pyrazoles as parasiticides |
| WO2023036821A1 (en) | 2021-09-09 | 2023-03-16 | Bayer Animal Health Gmbh | New quinoline derivatives |
| EP4148052A1 (en) | 2021-09-09 | 2023-03-15 | Bayer Animal Health GmbH | New quinoline derivatives |
| WO2023078915A1 (en) | 2021-11-03 | 2023-05-11 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether (thio)amides as fungicidal compounds |
| WO2023092050A1 (en) | 2021-11-20 | 2023-05-25 | Bayer Cropscience Lp | Beneficial combinations with recombinant bacillus cells expressing a serine protease |
| WO2023099445A1 (en) | 2021-11-30 | 2023-06-08 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether oxadiazines as fungicidal compounds |
| WO2023110656A1 (en) | 2021-12-15 | 2023-06-22 | Bayer Aktiengesellschaft | Spectroscopic solution for non-destructive quantification of one or more chemical substances in a matrix comprising coating and bulk material in a sample, such as coated seeds, using multivariate data analysis |
| WO2023205602A1 (en) | 2022-04-18 | 2023-10-26 | Basf Corporation | High-load agricultural formulations and methods of making same |
| WO2023203009A1 (en) | 2022-04-20 | 2023-10-26 | Bayer Aktiengesellschaft | Water dispersible granules with low melting active ingredients prepared by extrusion |
| EP4265110A1 (en) | 2022-04-20 | 2023-10-25 | Bayer AG | Water dispersible granules with low melting active ingredients prepared by extrusion |
| WO2023213670A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Crystalline forms of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine |
| WO2023213626A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Use of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine for controlling unwanted microorganisms |
| WO2023217619A1 (en) | 2022-05-07 | 2023-11-16 | Bayer Aktiengesellschaft | Low drift aqueous liquid formulations for low, medium, and high spray volume application |
| WO2023237444A1 (en) | 2022-06-06 | 2023-12-14 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295683A1 (en) | 2022-06-21 | 2023-12-27 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| WO2024013016A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024013015A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024068473A1 (en) | 2022-09-27 | 2024-04-04 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2024068519A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068518A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-heteroaryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068520A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| EP4295688A1 (en) | 2022-09-28 | 2023-12-27 | Bayer Aktiengesellschaft | Active compound combination |
| WO2024068517A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| EP4353082A1 (en) | 2022-10-14 | 2024-04-17 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024104643A1 (en) | 2022-11-17 | 2024-05-23 | Bayer Aktiengesellschaft | Use of isotianil for controlling plasmodiophora brassica |
| WO2024155864A1 (en) * | 2023-01-20 | 2024-07-25 | Allianthera (Suzhou) Biopharmaceutical Co., Ltd. | Sprk1 inhibitors and methods of use |
| WO2024170472A1 (en) | 2023-02-16 | 2024-08-22 | Bayer Aktiengesellschaft | Herbicidal mixtures |
| WO2024213752A1 (en) | 2023-04-14 | 2024-10-17 | Elanco Animal Health Gmbh | Long-term prevention and/or treatment of a disease by slo-1 inhibitors |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2006003494A2 (en) | Piperidine derivatives and their use as insecticides, acaricides, molluscicides or nematicides | |
| EP1515969B2 (en) | Spiroindolinepiperidine derivatives | |
| EP1755396A1 (en) | Piperazin derivatives and their use in controlling pests | |
| US8193362B2 (en) | Insecticidal compounds | |
| EP1979354A1 (en) | Diaza-spiro-[4, 5]-decanes useful as pesticides | |
| EP1732385A1 (en) | Use of (3-(1-(3-phenyl-propenyl)-piperidin-4-yl)-2,3-dihydro-indol-1-yl)-(pyridin-4-yl)-methanone derivatives and related compounds as insecticides | |
| EP1694676A1 (en) | Spiroindoline derivatives having insecticidal properties | |
| EP1694677A1 (en) | Spiropiperidine derivatives for controlling pests | |
| TWI466881B (en) | Insecticidal compounds | |
| EP1697376A1 (en) | Spiro-condensed indoline derivatives as pesticides | |
| EP2207421B1 (en) | Haloalkylsubstituted aryloxyalkylimidazolines for use as pesticides | |
| EP1965651A2 (en) | Chemical compounds | |
| EP1697327A1 (en) | Insecticidal spiroindane derivatives | |
| WO2007060541A2 (en) | Piperazine derivatives and their use as insecticides, acaricides, molluscicides, and nematicides |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12006502277 Country of ref document: PH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200609687 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005757532 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AP/P/2006/003830 Country of ref document: AP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 551629 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2006/014005 Country of ref document: MX Ref document number: 179745 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2568808 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2006-008790 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005258905 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007517523 Country of ref document: JP Ref document number: 06129099 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020067027660 Country of ref document: KR Ref document number: 200580021847.7 Country of ref document: CN Ref document number: 4783/CHENP/2006 Country of ref document: IN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2005258905 Country of ref document: AU Date of ref document: 20050622 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005258905 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: DZP2007000065 Country of ref document: DZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200602170 Country of ref document: EA |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067027660 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005757532 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11571303 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0512659 Country of ref document: BR |