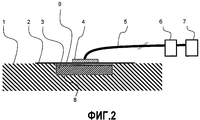
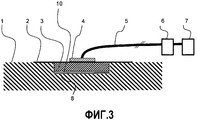
RU2530054C2 - Устройство для вакуум-терапии ран - Google Patents
Устройство для вакуум-терапии ран Download PDFInfo
- Publication number
- RU2530054C2 RU2530054C2 RU2012129974/05A RU2012129974A RU2530054C2 RU 2530054 C2 RU2530054 C2 RU 2530054C2 RU 2012129974/05 A RU2012129974/05 A RU 2012129974/05A RU 2012129974 A RU2012129974 A RU 2012129974A RU 2530054 C2 RU2530054 C2 RU 2530054C2
- Authority
- RU
- Russia
- Prior art keywords
- wound
- vacuum
- foam
- wounds
- polyorganosiloxane
- Prior art date
Links
- 208000027418 Wounds and injury Diseases 0.000 title claims abstract description 216
- 206010052428 Wound Diseases 0.000 title claims abstract description 214
- 238000002560 therapeutic procedure Methods 0.000 title claims abstract description 51
- 239000000463 material Substances 0.000 claims abstract description 66
- 239000000203 mixture Substances 0.000 claims abstract description 26
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims abstract description 18
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims abstract description 11
- 239000003054 catalyst Substances 0.000 claims abstract description 9
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims abstract description 6
- 239000007788 liquid Substances 0.000 claims abstract description 6
- 239000006260 foam Substances 0.000 claims description 83
- 239000011248 coating agent Substances 0.000 claims description 56
- 238000000576 coating method Methods 0.000 claims description 56
- 125000003118 aryl group Chemical group 0.000 claims description 7
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 6
- 230000035699 permeability Effects 0.000 claims description 6
- 239000004604 Blowing Agent Substances 0.000 claims description 5
- 125000004737 (C1-C6) haloalkoxy group Chemical group 0.000 claims description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 4
- 229910052739 hydrogen Inorganic materials 0.000 claims description 4
- 239000001257 hydrogen Substances 0.000 claims description 4
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 4
- 125000002524 organometallic group Chemical group 0.000 claims description 4
- 230000037431 insertion Effects 0.000 claims 2
- 238000003780 insertion Methods 0.000 claims 2
- 239000011148 porous material Substances 0.000 abstract description 9
- 239000000126 substance Substances 0.000 abstract description 5
- 238000006243 chemical reaction Methods 0.000 abstract description 4
- 230000000694 effects Effects 0.000 abstract description 2
- 239000006261 foam material Substances 0.000 abstract 3
- 239000003814 drug Substances 0.000 abstract 1
- 210000004027 cell Anatomy 0.000 description 22
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 19
- 229920001296 polysiloxane Polymers 0.000 description 18
- -1 polypropylene Polymers 0.000 description 17
- 239000000853 adhesive Substances 0.000 description 13
- 230000008901 benefit Effects 0.000 description 13
- 239000004372 Polyvinyl alcohol Substances 0.000 description 9
- 239000003570 air Substances 0.000 description 9
- 150000001298 alcohols Chemical class 0.000 description 9
- 210000000416 exudates and transudate Anatomy 0.000 description 9
- 239000010408 film Substances 0.000 description 9
- 229920002451 polyvinyl alcohol Polymers 0.000 description 9
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 8
- 230000001070 adhesive effect Effects 0.000 description 8
- 238000009826 distribution Methods 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- 229910052709 silver Inorganic materials 0.000 description 7
- 239000004332 silver Substances 0.000 description 7
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 6
- 238000000034 method Methods 0.000 description 6
- 229920000642 polymer Polymers 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 5
- 239000012530 fluid Substances 0.000 description 5
- 229910052697 platinum Inorganic materials 0.000 description 5
- 229920003176 water-insoluble polymer Polymers 0.000 description 5
- 230000029663 wound healing Effects 0.000 description 5
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 4
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 4
- 238000004026 adhesive bonding Methods 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 239000003292 glue Substances 0.000 description 4
- ZSIAUFGUXNUGDI-UHFFFAOYSA-N hexan-1-ol Chemical compound CCCCCCO ZSIAUFGUXNUGDI-UHFFFAOYSA-N 0.000 description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 4
- 239000004814 polyurethane Substances 0.000 description 4
- 230000001681 protective effect Effects 0.000 description 4
- 238000007789 sealing Methods 0.000 description 4
- 238000009489 vacuum treatment Methods 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- BTANRVKWQNVYAZ-UHFFFAOYSA-N butan-2-ol Chemical compound CCC(C)O BTANRVKWQNVYAZ-UHFFFAOYSA-N 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 238000010276 construction Methods 0.000 description 3
- 239000002657 fibrous material Substances 0.000 description 3
- 238000005755 formation reaction Methods 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 244000005700 microbiome Species 0.000 description 3
- 125000004430 oxygen atom Chemical group O* 0.000 description 3
- 230000000704 physical effect Effects 0.000 description 3
- 229920000728 polyester Polymers 0.000 description 3
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 238000002054 transplantation Methods 0.000 description 3
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 2
- BBMCTIGTTCKYKF-UHFFFAOYSA-N 1-heptanol Chemical compound CCCCCCCO BBMCTIGTTCKYKF-UHFFFAOYSA-N 0.000 description 2
- 206010011985 Decubitus ulcer Diseases 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical group [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical group [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 208000004210 Pressure Ulcer Diseases 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 239000002313 adhesive film Substances 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- 125000004429 atom Chemical group 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 2
- 210000002421 cell wall Anatomy 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000007795 chemical reaction product Substances 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 238000004132 cross linking Methods 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- BITPLIXHRASDQB-UHFFFAOYSA-N ethenyl-[ethenyl(dimethyl)silyl]oxy-dimethylsilane Chemical compound C=C[Si](C)(C)O[Si](C)(C)C=C BITPLIXHRASDQB-UHFFFAOYSA-N 0.000 description 2
- 239000004744 fabric Substances 0.000 description 2
- QNVRIHYSUZMSGM-UHFFFAOYSA-N hexan-2-ol Chemical compound CCCCC(C)O QNVRIHYSUZMSGM-UHFFFAOYSA-N 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 230000007794 irritation Effects 0.000 description 2
- 229910052753 mercury Inorganic materials 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 239000000565 sealant Substances 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 230000003068 static effect Effects 0.000 description 2
- 230000001954 sterilising effect Effects 0.000 description 2
- 238000004659 sterilization and disinfection Methods 0.000 description 2
- 229920000247 superabsorbent polymer Polymers 0.000 description 2
- 239000004753 textile Substances 0.000 description 2
- 239000012780 transparent material Substances 0.000 description 2
- 238000009827 uniform distribution Methods 0.000 description 2
- 229920002554 vinyl polymer Polymers 0.000 description 2
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 description 1
- 125000006729 (C2-C5) alkenyl group Chemical group 0.000 description 1
- QNVRIHYSUZMSGM-LURJTMIESA-N 2-Hexanol Natural products CCCC[C@H](C)O QNVRIHYSUZMSGM-LURJTMIESA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical group [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229920005830 Polyurethane Foam Polymers 0.000 description 1
- 229910008051 Si-OH Inorganic materials 0.000 description 1
- 229920002323 Silicone foam Polymers 0.000 description 1
- 229910006358 Si—OH Inorganic materials 0.000 description 1
- 206010072170 Skin wound Diseases 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000002390 adhesive tape Substances 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 239000012080 ambient air Substances 0.000 description 1
- 230000036770 blood supply Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 210000003850 cellular structure Anatomy 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical group [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 238000003869 coulometry Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- XCIXKGXIYUWCLL-UHFFFAOYSA-N cyclopentanol Chemical compound OC1CCCC1 XCIXKGXIYUWCLL-UHFFFAOYSA-N 0.000 description 1
- 239000000645 desinfectant Substances 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000000383 hazardous chemical Substances 0.000 description 1
- 230000036074 healthy skin Effects 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 208000030159 metabolic disease Diseases 0.000 description 1
- QQZOPKMRPOGIEB-UHFFFAOYSA-N n-butyl methyl ketone Natural products CCCCC(C)=O QQZOPKMRPOGIEB-UHFFFAOYSA-N 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 150000002902 organometallic compounds Chemical class 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 229920006264 polyurethane film Polymers 0.000 description 1
- 239000011496 polyurethane foam Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000013514 silicone foam Substances 0.000 description 1
- 239000011343 solid material Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/26—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/425—Porous materials, e.g. foams or sponges
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/90—Negative pressure wound therapy devices, i.e. devices for applying suction to a wound to promote healing, e.g. including a vacuum dressing
- A61M1/91—Suction aspects of the dressing
- A61M1/915—Constructional details of the pressure distribution manifold
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/02—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof using blowing gases generated by the reacting monomers or modifying agents during the preparation or modification of macromolecules
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/90—Negative pressure wound therapy devices, i.e. devices for applying suction to a wound to promote healing, e.g. including a vacuum dressing
- A61M1/98—Containers specifically adapted for negative pressure wound therapy
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2201/00—Foams characterised by the foaming process
- C08J2201/02—Foams characterised by the foaming process characterised by mechanical pre- or post-treatments
- C08J2201/024—Preparation or use of a blowing agent concentrate, i.e. masterbatch in a foamable composition
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2205/00—Foams characterised by their properties
- C08J2205/04—Foams characterised by their properties characterised by the foam pores
- C08J2205/05—Open cells, i.e. more than 50% of the pores are open
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2207/00—Foams characterised by their intended use
- C08J2207/10—Medical applications, e.g. biocompatible scaffolds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2383/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen, or carbon only; Derivatives of such polymers
- C08J2383/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L83/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers
- C08L83/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L83/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers
- C08L83/14—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers in which at least two but not all the silicon atoms are connected by linkages other than oxygen atoms
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Hematology (AREA)
- Materials Engineering (AREA)
- Heart & Thoracic Surgery (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Vascular Medicine (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Dispersion Chemistry (AREA)
- Organic Chemistry (AREA)
- Polymers & Plastics (AREA)
- Medicinal Chemistry (AREA)
- Materials For Medical Uses (AREA)
- Medicinal Preparation (AREA)
- Manufacture Of Porous Articles, And Recovery And Treatment Of Waste Products (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09015561.5A EP2335747B1 (de) | 2009-12-16 | 2009-12-16 | Vorrichtung zur Unterdrucktherapie von Wunden |
| EP09015561.5 | 2009-12-16 | ||
| PCT/EP2010/007620 WO2011072840A1 (de) | 2009-12-16 | 2010-12-14 | Vorrichtung zur unterdrucktherapie von wunden |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2012129974A RU2012129974A (ru) | 2014-01-27 |
| RU2530054C2 true RU2530054C2 (ru) | 2014-10-10 |
Family
ID=42122859
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2012129974/05A RU2530054C2 (ru) | 2009-12-16 | 2010-12-14 | Устройство для вакуум-терапии ран |
Country Status (5)
| Country | Link |
|---|---|
| EP (2) | EP2335747B1 (enExample) |
| JP (1) | JP5723386B2 (enExample) |
| CN (1) | CN102740901B (enExample) |
| RU (1) | RU2530054C2 (enExample) |
| WO (1) | WO2011072840A1 (enExample) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2771915C1 (ru) * | 2021-04-20 | 2022-05-13 | Алексей Андреевич Евсюков | Устройство для создания среды с заданными характеристиками в ранах мягких тканей и повязка для реализации устройства |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0224986D0 (en) | 2002-10-28 | 2002-12-04 | Smith & Nephew | Apparatus |
| GB201011173D0 (en) | 2010-07-02 | 2010-08-18 | Smith & Nephew | Provision of wound filler |
| BR112013012785A2 (pt) * | 2010-11-25 | 2020-10-20 | Bluestar Silicones France Sas | composição i - ii e produtos e usos das mesmas |
| GB201020005D0 (en) * | 2010-11-25 | 2011-01-12 | Smith & Nephew | Composition 1-1 |
| US20150159066A1 (en) | 2011-11-25 | 2015-06-11 | Smith & Nephew Plc | Composition, apparatus, kit and method and uses thereof |
| US20160120706A1 (en) | 2013-03-15 | 2016-05-05 | Smith & Nephew Plc | Wound dressing sealant and use thereof |
| EP2968647B1 (en) | 2013-03-15 | 2022-06-29 | Smith & Nephew plc | Wound dressing sealant and use thereof |
| DE102013008964A1 (de) | 2013-05-22 | 2014-11-27 | Gmbu E.V., Fachsektion Dresden | Vorrichtung zur Behandlung von Problemwunden |
| DE102014106518A1 (de) | 2014-05-09 | 2015-11-12 | Paul Hartmann Ag | Schaumwundauflage für die Unterdrucktherapie |
| DE102014116912A1 (de) | 2014-11-19 | 2016-05-19 | Paul Hartmann Ag | Flüssigkeitsindikator für Unterdrucktherapie-Vorrichtung |
| DE102014116910A1 (de) | 2014-11-19 | 2016-05-19 | Paul Hartmann Ag | Elektronischer Flüssigkeitssensor für Unterdrucktherapie-Vorrichtung |
| DE102015007622A1 (de) | 2015-06-16 | 2016-12-22 | Paul Hartmann Ag | Vorrichtung zur Unterdrucktherapie von Wunden enthaltend siliconhaltigen Polyurethanschaumstoff |
| DE102016114817A1 (de) | 2016-08-10 | 2018-02-15 | Paul Hartmann Ag | Medizinisches Kit zur Verwendung bei der Unterdrucktherapie von endoluminalen Wundstellen im Bereich des Gastrointestinaltraktes |
| DE102016114786A1 (de) | 2016-08-10 | 2018-02-15 | Paul Hartmann Ag | Fassschlaufe für Saugkörper zur endoluminalen Unterdrucktherapie |
| DE102016114819A1 (de) | 2016-08-10 | 2018-02-15 | Paul Hartmann Ag | Saugkörper zur endoluminalen Unterdrucktherapie |
| GB201904402D0 (en) | 2019-03-29 | 2019-05-15 | Trio Healthcare Ltd | Foamed skin compatible silicone composition |
| CN120168707A (zh) * | 2025-04-07 | 2025-06-20 | 安徽省小山卫生材料有限公司 | 一种软聚硅酮泡沫敷料及其生产方法 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0691113A1 (en) * | 1994-06-08 | 1996-01-10 | Seton Healthcare Group plc | Wound dressings |
| RU22749U1 (ru) * | 2001-02-06 | 2002-04-27 | Закрытое акционерное общество "МедСил" | Резиновая медицинская силиконовая трубка |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3775452A (en) | 1971-04-28 | 1973-11-27 | Gen Electric | Platinum complexes of unsaturated siloxanes and platinum containing organopolysiloxanes |
| US4808634A (en) * | 1988-03-24 | 1989-02-28 | General Electric Company | Low density silicon foam |
| US5645081A (en) | 1991-11-14 | 1997-07-08 | Wake Forest University | Method of treating tissue damage and apparatus for same |
| JP3500992B2 (ja) * | 1997-11-19 | 2004-02-23 | 信越化学工業株式会社 | オルガノポリシロキサン組成物 |
| CA2409907A1 (en) | 2000-05-22 | 2001-11-29 | Arthur C. Coffey | Combination sis and vacuum bandage and method |
| JP3647389B2 (ja) * | 2000-08-01 | 2005-05-11 | ジーイー東芝シリコーン株式会社 | ポリオルガノシロキサン発泡材、発泡体およびその製造方法 |
| US7700819B2 (en) * | 2001-02-16 | 2010-04-20 | Kci Licensing, Inc. | Biocompatible wound dressing |
| GB2415382A (en) | 2004-06-21 | 2005-12-28 | Johnson & Johnson Medical Ltd | Wound dressings for vacuum therapy |
| GB2415908A (en) | 2004-07-09 | 2006-01-11 | Ethicon Inc | Vacuum wound dressings |
| DE202004017052U1 (de) | 2004-11-02 | 2005-06-09 | Riesinger, Birgit | Vorrichtung zur Wundbehandlung unter Einsatz von Unterdruck |
| ES2901826T3 (es) | 2004-11-05 | 2022-03-23 | Convatec Technologies Inc | Módulo para el cuidado de heridas al vacío |
| FR2902107A1 (fr) * | 2006-06-07 | 2007-12-14 | Rhodia Recherches & Tech | Composition organopolysiloxane pour mousse elastomere |
| US7842848B2 (en) * | 2006-11-13 | 2010-11-30 | Ossur Hf | Absorbent structure in an absorbent article |
| JP2008150447A (ja) * | 2006-12-15 | 2008-07-03 | Shin Etsu Chem Co Ltd | 発泡性オルガノポリシロキサン組成物およびシリコーンゴムスポンジ |
| US8377016B2 (en) * | 2007-01-10 | 2013-02-19 | Wake Forest University Health Sciences | Apparatus and method for wound treatment employing periodic sub-atmospheric pressure |
| JP2008214440A (ja) * | 2007-03-01 | 2008-09-18 | Shin Etsu Chem Co Ltd | シリコーンゴムスポンジ組成物 |
| RU2010135033A (ru) * | 2008-03-05 | 2012-04-10 | КейСиАй Лайсензинг Инк. (US) | Повязка и способ приложения пониженного давления к участку ткани и сбора и хранения текучей среды от участка ткани |
-
2009
- 2009-12-16 EP EP09015561.5A patent/EP2335747B1/de active Active
-
2010
- 2010-12-14 WO PCT/EP2010/007620 patent/WO2011072840A1/de not_active Ceased
- 2010-12-14 JP JP2012543519A patent/JP5723386B2/ja not_active Expired - Fee Related
- 2010-12-14 EP EP10795947A patent/EP2512544A1/de not_active Withdrawn
- 2010-12-14 CN CN201080057146.XA patent/CN102740901B/zh active Active
- 2010-12-14 RU RU2012129974/05A patent/RU2530054C2/ru active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0691113A1 (en) * | 1994-06-08 | 1996-01-10 | Seton Healthcare Group plc | Wound dressings |
| RU22749U1 (ru) * | 2001-02-06 | 2002-04-27 | Закрытое акционерное общество "МедСил" | Резиновая медицинская силиконовая трубка |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2771915C1 (ru) * | 2021-04-20 | 2022-05-13 | Алексей Андреевич Евсюков | Устройство для создания среды с заданными характеристиками в ранах мягких тканей и повязка для реализации устройства |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2013514094A (ja) | 2013-04-25 |
| WO2011072840A1 (de) | 2011-06-23 |
| CN102740901B (zh) | 2015-11-25 |
| RU2012129974A (ru) | 2014-01-27 |
| EP2512544A1 (de) | 2012-10-24 |
| CN102740901A (zh) | 2012-10-17 |
| EP2335747A1 (de) | 2011-06-22 |
| JP5723386B2 (ja) | 2015-05-27 |
| EP2335747B1 (de) | 2016-12-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2530054C2 (ru) | Устройство для вакуум-терапии ран | |
| US9452247B2 (en) | Device for negative pressure wound therapy | |
| US11931226B2 (en) | Wound dressing sealant and use thereof | |
| AU2014229779B2 (en) | Wound dressing sealant and use thereof | |
| RU2597393C2 (ru) | Композиция i-ii, содержащие ее изделия и ее применения | |
| RU2609993C2 (ru) | Композиция, приспособление, набор и способ и их использование | |
| ES2582564T3 (es) | Apósito para heridas con margen adhesivo | |
| EP2670369A1 (en) | Silicone wound dressing laminate and method for making the same | |
| US20240139035A1 (en) | Wound dressing sealant and use thereof | |
| CN103596527A (zh) | 用于减压疗法的包含泡沫材料和药膏基质和膨胀材料的伤口敷料 | |
| HK1165981A1 (en) | Film for covering a wound | |
| HK1165981B (en) | Film for covering a wound |