KR20190039937A - 항-ApoC3 항체 및 이의 사용 방법 - Google Patents
항-ApoC3 항체 및 이의 사용 방법 Download PDFInfo
- Publication number
- KR20190039937A KR20190039937A KR1020197002789A KR20197002789A KR20190039937A KR 20190039937 A KR20190039937 A KR 20190039937A KR 1020197002789 A KR1020197002789 A KR 1020197002789A KR 20197002789 A KR20197002789 A KR 20197002789A KR 20190039937 A KR20190039937 A KR 20190039937A
- Authority
- KR
- South Korea
- Prior art keywords
- antibody
- ser
- apoc3
- gly
- thr
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 238000000034 method Methods 0.000 title claims abstract description 180
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 47
- 239000013604 expression vector Substances 0.000 claims abstract description 30
- 210000004027 cell Anatomy 0.000 claims description 132
- 150000001413 amino acids Chemical group 0.000 claims description 98
- 230000027455 binding Effects 0.000 claims description 91
- 150000002632 lipids Chemical class 0.000 claims description 69
- 108010062497 VLDL Lipoproteins Proteins 0.000 claims description 61
- 210000004369 blood Anatomy 0.000 claims description 51
- 239000008280 blood Substances 0.000 claims description 51
- 108091033319 polynucleotide Proteins 0.000 claims description 48
- 102000040430 polynucleotide Human genes 0.000 claims description 48
- 239000002157 polynucleotide Substances 0.000 claims description 48
- 108010027006 Apolipoproteins B Proteins 0.000 claims description 39
- 102000018616 Apolipoproteins B Human genes 0.000 claims description 39
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 32
- 230000000291 postprandial effect Effects 0.000 claims description 31
- 230000014509 gene expression Effects 0.000 claims description 25
- 210000003494 hepatocyte Anatomy 0.000 claims description 24
- 230000000694 effects Effects 0.000 claims description 21
- 230000001404 mediated effect Effects 0.000 claims description 21
- 102000004882 Lipase Human genes 0.000 claims description 20
- 108090001060 Lipase Proteins 0.000 claims description 20
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 claims description 20
- 239000003524 antilipemic agent Substances 0.000 claims description 17
- 208000024172 Cardiovascular disease Diseases 0.000 claims description 16
- 208000031226 Hyperlipidaemia Diseases 0.000 claims description 16
- 230000001965 increasing effect Effects 0.000 claims description 16
- 230000004130 lipolysis Effects 0.000 claims description 16
- 208000006575 hypertriglyceridemia Diseases 0.000 claims description 12
- 230000002401 inhibitory effect Effects 0.000 claims description 12
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 claims description 11
- 230000000295 complement effect Effects 0.000 claims description 11
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 claims description 11
- 108010004103 Chylomicrons Proteins 0.000 claims description 9
- OLNTVTPDXPETLC-XPWALMASSA-N ezetimibe Chemical compound N1([C@@H]([C@H](C1=O)CC[C@H](O)C=1C=CC(F)=CC=1)C=1C=CC(O)=CC=1)C1=CC=C(F)C=C1 OLNTVTPDXPETLC-XPWALMASSA-N 0.000 claims description 9
- 239000013078 crystal Substances 0.000 claims description 8
- 229960000815 ezetimibe Drugs 0.000 claims description 8
- 229940122392 PCSK9 inhibitor Drugs 0.000 claims description 7
- 239000003937 drug carrier Substances 0.000 claims description 6
- 230000004576 lipid-binding Effects 0.000 claims description 5
- 206010002383 Angina Pectoris Diseases 0.000 claims description 4
- 201000001320 Atherosclerosis Diseases 0.000 claims description 4
- 208000010125 myocardial infarction Diseases 0.000 claims description 4
- FJLGEFLZQAZZCD-MCBHFWOFSA-N (3R,5S)-fluvastatin Chemical compound C12=CC=CC=C2N(C(C)C)C(\C=C\[C@@H](O)C[C@@H](O)CC(O)=O)=C1C1=CC=C(F)C=C1 FJLGEFLZQAZZCD-MCBHFWOFSA-N 0.000 claims description 3
- XUKUURHRXDUEBC-KAYWLYCHSA-N Atorvastatin Chemical compound C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@@H](O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-KAYWLYCHSA-N 0.000 claims description 3
- XUKUURHRXDUEBC-UHFFFAOYSA-N Atorvastatin Natural products C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CCC(O)CC(O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-UHFFFAOYSA-N 0.000 claims description 3
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 claims description 3
- TUZYXOIXSAXUGO-UHFFFAOYSA-N Pravastatin Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(O)C=C21 TUZYXOIXSAXUGO-UHFFFAOYSA-N 0.000 claims description 3
- RYMZZMVNJRMUDD-UHFFFAOYSA-N SJ000286063 Natural products C12C(OC(=O)C(C)(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 RYMZZMVNJRMUDD-UHFFFAOYSA-N 0.000 claims description 3
- 229960005370 atorvastatin Drugs 0.000 claims description 3
- 238000012258 culturing Methods 0.000 claims description 3
- 229960003765 fluvastatin Drugs 0.000 claims description 3
- 229960004844 lovastatin Drugs 0.000 claims description 3
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 claims description 3
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 claims description 3
- 229960002797 pitavastatin Drugs 0.000 claims description 3
- VGYFMXBACGZSIL-MCBHFWOFSA-N pitavastatin Chemical compound OC(=O)C[C@H](O)C[C@H](O)\C=C\C1=C(C2CC2)N=C2C=CC=CC2=C1C1=CC=C(F)C=C1 VGYFMXBACGZSIL-MCBHFWOFSA-N 0.000 claims description 3
- 229960002965 pravastatin Drugs 0.000 claims description 3
- TUZYXOIXSAXUGO-PZAWKZKUSA-N pravastatin Chemical compound C1=C[C@H](C)[C@H](CC[C@@H](O)C[C@@H](O)CC(O)=O)[C@H]2[C@@H](OC(=O)[C@@H](C)CC)C[C@H](O)C=C21 TUZYXOIXSAXUGO-PZAWKZKUSA-N 0.000 claims description 3
- 229960000672 rosuvastatin Drugs 0.000 claims description 3
- BPRHUIZQVSMCRT-VEUZHWNKSA-N rosuvastatin Chemical compound CC(C)C1=NC(N(C)S(C)(=O)=O)=NC(C=2C=CC(F)=CC=2)=C1\C=C\[C@@H](O)C[C@@H](O)CC(O)=O BPRHUIZQVSMCRT-VEUZHWNKSA-N 0.000 claims description 3
- 229960002855 simvastatin Drugs 0.000 claims description 3
- RYMZZMVNJRMUDD-HGQWONQESA-N simvastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)C(C)(C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 RYMZZMVNJRMUDD-HGQWONQESA-N 0.000 claims description 3
- 241000282414 Homo sapiens Species 0.000 abstract description 155
- 150000007523 nucleic acids Chemical class 0.000 abstract description 26
- 102000039446 nucleic acids Human genes 0.000 abstract description 24
- 108020004707 nucleic acids Proteins 0.000 abstract description 24
- 235000001014 amino acid Nutrition 0.000 description 103
- 125000003275 alpha amino acid group Chemical group 0.000 description 94
- 229940024606 amino acid Drugs 0.000 description 90
- 108090000623 proteins and genes Proteins 0.000 description 78
- 108090000765 processed proteins & peptides Proteins 0.000 description 66
- 235000018102 proteins Nutrition 0.000 description 49
- 102000004169 proteins and genes Human genes 0.000 description 49
- 239000013598 vector Substances 0.000 description 47
- 239000000427 antigen Substances 0.000 description 44
- 108091007433 antigens Proteins 0.000 description 44
- 102000036639 antigens Human genes 0.000 description 44
- 241000880493 Leptailurus serval Species 0.000 description 43
- XKUKSGPZAADMRA-UHFFFAOYSA-N glycyl-glycyl-glycine Natural products NCC(=O)NCC(=O)NCC(O)=O XKUKSGPZAADMRA-UHFFFAOYSA-N 0.000 description 39
- 102000004196 processed proteins & peptides Human genes 0.000 description 38
- 238000003556 assay Methods 0.000 description 34
- 108010089804 glycyl-threonine Proteins 0.000 description 31
- 108060003951 Immunoglobulin Proteins 0.000 description 30
- 102000018358 immunoglobulin Human genes 0.000 description 30
- XXXAXOWMBOKTRN-XPUUQOCRSA-N Ser-Gly-Val Chemical compound [H]N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C(C)C)C(O)=O XXXAXOWMBOKTRN-XPUUQOCRSA-N 0.000 description 29
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 29
- 239000002953 phosphate buffered saline Substances 0.000 description 29
- 210000004408 hybridoma Anatomy 0.000 description 27
- 238000012360 testing method Methods 0.000 description 27
- 239000000872 buffer Substances 0.000 description 26
- PCDUALPXEOKZPE-DXCABUDRSA-N (2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-amino-3-hydroxypropanoyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxypropanoic acid Chemical compound OC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O PCDUALPXEOKZPE-DXCABUDRSA-N 0.000 description 25
- FITIQFSXXBKFFM-NRPADANISA-N Gln-Val-Ser Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O FITIQFSXXBKFFM-NRPADANISA-N 0.000 description 23
- 150000003626 triacylglycerols Chemical class 0.000 description 23
- 108010086434 alanyl-seryl-glycine Proteins 0.000 description 22
- 108010067216 glycyl-glycyl-glycine Proteins 0.000 description 22
- OWFGFHQMSBTKLX-UFYCRDLUSA-N Val-Tyr-Tyr Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)O)N OWFGFHQMSBTKLX-UFYCRDLUSA-N 0.000 description 21
- 239000000203 mixture Substances 0.000 description 21
- 108010073969 valyllysine Proteins 0.000 description 21
- 241000699666 Mus <mouse, genus> Species 0.000 description 20
- 241000699670 Mus sp. Species 0.000 description 20
- 239000000243 solution Substances 0.000 description 20
- 238000006467 substitution reaction Methods 0.000 description 20
- MIIVFRCYJABHTQ-ONGXEEELSA-N Gly-Leu-Val Chemical compound [H]NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(O)=O MIIVFRCYJABHTQ-ONGXEEELSA-N 0.000 description 19
- LCNASHSOFMRYFO-WDCWCFNPSA-N Leu-Thr-Gln Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@H](C(O)=O)CCC(N)=O LCNASHSOFMRYFO-WDCWCFNPSA-N 0.000 description 19
- QEDMOZUJTGEIBF-FXQIFTODSA-N Ser-Arg-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(O)=O QEDMOZUJTGEIBF-FXQIFTODSA-N 0.000 description 19
- YMTLKLXDFCSCNX-BYPYZUCNSA-N Ser-Gly-Gly Chemical compound OC[C@H](N)C(=O)NCC(=O)NCC(O)=O YMTLKLXDFCSCNX-BYPYZUCNSA-N 0.000 description 19
- 108010081404 acein-2 Proteins 0.000 description 19
- PZXPWHFYZXTFBI-YUMQZZPRSA-N Asp-Gly-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H](N)CC(O)=O PZXPWHFYZXTFBI-YUMQZZPRSA-N 0.000 description 18
- JSHWXQIZOCVWIA-ZKWXMUAHSA-N Asp-Ser-Val Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O JSHWXQIZOCVWIA-ZKWXMUAHSA-N 0.000 description 18
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 18
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 18
- PMGDADKJMCOXHX-UHFFFAOYSA-N L-Arginyl-L-glutamin-acetat Natural products NC(=N)NCCCC(N)C(=O)NC(CCC(N)=O)C(O)=O PMGDADKJMCOXHX-UHFFFAOYSA-N 0.000 description 18
- NRFGTHFONZYFNY-MGHWNKPDSA-N Leu-Ile-Tyr Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 NRFGTHFONZYFNY-MGHWNKPDSA-N 0.000 description 18
- QYSFWUIXDFJUDW-DCAQKATOSA-N Ser-Leu-Arg Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O QYSFWUIXDFJUDW-DCAQKATOSA-N 0.000 description 18
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 18
- JAWUQFCGNVEDRN-MEYUZBJRSA-N Thr-Tyr-Leu Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CC(C)C)C(=O)O)N)O JAWUQFCGNVEDRN-MEYUZBJRSA-N 0.000 description 18
- MBLJBGZWLHTJBH-SZMVWBNQSA-N Trp-Val-Arg Chemical compound C1=CC=C2C(C[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O)=CNC2=C1 MBLJBGZWLHTJBH-SZMVWBNQSA-N 0.000 description 18
- 108010008355 arginyl-glutamine Proteins 0.000 description 18
- 230000000903 blocking effect Effects 0.000 description 18
- 238000005516 engineering process Methods 0.000 description 18
- 239000012634 fragment Substances 0.000 description 18
- 239000002773 nucleotide Substances 0.000 description 18
- 125000003729 nucleotide group Chemical group 0.000 description 18
- 101100315624 Caenorhabditis elegans tyr-1 gene Proteins 0.000 description 17
- PDUHNKAFQXQNLH-ZETCQYMHSA-N Gly-Lys-Gly Chemical compound NCCCC[C@H](NC(=O)CN)C(=O)NCC(O)=O PDUHNKAFQXQNLH-ZETCQYMHSA-N 0.000 description 17
- AIMGJYMCTAABEN-GVXVVHGQSA-N Leu-Val-Glu Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O AIMGJYMCTAABEN-GVXVVHGQSA-N 0.000 description 17
- 229920001184 polypeptide Polymers 0.000 description 17
- 108010029020 prolylglycine Proteins 0.000 description 17
- YYSWCHMLFJLLBJ-ZLUOBGJFSA-N Ala-Ala-Ser Chemical compound C[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O YYSWCHMLFJLLBJ-ZLUOBGJFSA-N 0.000 description 16
- 101800001976 Apolipoprotein B-48 Proteins 0.000 description 16
- JBDLMLZNDRLDIX-HJGDQZAQSA-N Asn-Thr-Leu Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O JBDLMLZNDRLDIX-HJGDQZAQSA-N 0.000 description 16
- 108020004414 DNA Proteins 0.000 description 16
- 238000002965 ELISA Methods 0.000 description 16
- SOEGEPHNZOISMT-BYPYZUCNSA-N Gly-Ser-Gly Chemical compound NCC(=O)N[C@@H](CO)C(=O)NCC(O)=O SOEGEPHNZOISMT-BYPYZUCNSA-N 0.000 description 16
- JMVQDLDPDBXAAX-YUMQZZPRSA-N Pro-Gly-Gln Chemical compound NC(=O)CC[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1 JMVQDLDPDBXAAX-YUMQZZPRSA-N 0.000 description 16
- DJDSEDOKJTZBAR-ZDLURKLDSA-N Thr-Gly-Ser Chemical compound C[C@@H](O)[C@H](N)C(=O)NCC(=O)N[C@@H](CO)C(O)=O DJDSEDOKJTZBAR-ZDLURKLDSA-N 0.000 description 16
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 16
- 108010020755 prolyl-glycyl-glycine Proteins 0.000 description 16
- MRQQMVZUHXUPEV-IHRRRGAJSA-N Asp-Arg-Phe Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O MRQQMVZUHXUPEV-IHRRRGAJSA-N 0.000 description 15
- 102100035360 Cerebellar degeneration-related antigen 1 Human genes 0.000 description 15
- JXFLPKSDLDEOQK-JHEQGTHGSA-N Gln-Gly-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H](N)CCC(N)=O JXFLPKSDLDEOQK-JHEQGTHGSA-N 0.000 description 15
- FQCILXROGNOZON-YUMQZZPRSA-N Gln-Pro-Gly Chemical compound NC(=O)CC[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O FQCILXROGNOZON-YUMQZZPRSA-N 0.000 description 15
- 108010065920 Insulin Lispro Proteins 0.000 description 15
- AXZGZMGRBDQTEY-SRVKXCTJSA-N Leu-Gln-Met Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCSC)C(O)=O AXZGZMGRBDQTEY-SRVKXCTJSA-N 0.000 description 15
- KIZIOFNVSOSKJI-CIUDSAMLSA-N Leu-Ser-Cys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O)N KIZIOFNVSOSKJI-CIUDSAMLSA-N 0.000 description 15
- UIGMAMGZOJVTDN-WHFBIAKZSA-N Ser-Gly-Ser Chemical compound OC[C@H](N)C(=O)NCC(=O)N[C@@H](CO)C(O)=O UIGMAMGZOJVTDN-WHFBIAKZSA-N 0.000 description 15
- YUJLIIRMIAGMCQ-CIUDSAMLSA-N Ser-Leu-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(O)=O YUJLIIRMIAGMCQ-CIUDSAMLSA-N 0.000 description 15
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 15
- 108010063718 gamma-glutamylaspartic acid Proteins 0.000 description 15
- 108010078144 glutaminyl-glycine Proteins 0.000 description 15
- 108010018006 histidylserine Proteins 0.000 description 15
- 238000002493 microarray Methods 0.000 description 15
- SLKLLQWZQHXYSV-CIUDSAMLSA-N Asn-Ala-Lys Chemical compound NC(=O)C[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(O)=O SLKLLQWZQHXYSV-CIUDSAMLSA-N 0.000 description 14
- HPNDBHLITCHRSO-WHFBIAKZSA-N Asp-Ala-Gly Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](C)C(=O)NCC(O)=O HPNDBHLITCHRSO-WHFBIAKZSA-N 0.000 description 14
- MNQMTYSEKZHIDF-GCJQMDKQSA-N Asp-Thr-Ala Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(O)=O MNQMTYSEKZHIDF-GCJQMDKQSA-N 0.000 description 14
- 108091026890 Coding region Proteins 0.000 description 14
- BPLNJYHNAJVLRT-ACZMJKKPSA-N Glu-Ser-Ala Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(O)=O BPLNJYHNAJVLRT-ACZMJKKPSA-N 0.000 description 14
- FQZPTCNSNPWHLJ-AVGNSLFASA-N Leu-Gln-Lys Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCCN)C(O)=O FQZPTCNSNPWHLJ-AVGNSLFASA-N 0.000 description 14
- FEHQLKKBVJHSEC-SZMVWBNQSA-N Leu-Glu-Trp Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](N)CC(C)C)C(O)=O)=CNC2=C1 FEHQLKKBVJHSEC-SZMVWBNQSA-N 0.000 description 14
- GQZMPWBZQALKJO-UWVGGRQHSA-N Lys-Gly-Arg Chemical compound [H]N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(O)=O GQZMPWBZQALKJO-UWVGGRQHSA-N 0.000 description 14
- 241001465754 Metazoa Species 0.000 description 14
- UIPXCLNLUUAMJU-JBDRJPRFSA-N Ser-Ile-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O UIPXCLNLUUAMJU-JBDRJPRFSA-N 0.000 description 14
- 108010008685 alanyl-glutamyl-aspartic acid Proteins 0.000 description 14
- 238000002347 injection Methods 0.000 description 14
- 239000007924 injection Substances 0.000 description 14
- 238000004519 manufacturing process Methods 0.000 description 14
- 230000009467 reduction Effects 0.000 description 14
- CITHEXJVPOWHKC-UUWRZZSWSA-N 1,2-di-O-myristoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCC CITHEXJVPOWHKC-UUWRZZSWSA-N 0.000 description 13
- XMTDCXXLDZKAGI-ACZMJKKPSA-N Cys-Ala-Gln Chemical compound C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CS)N XMTDCXXLDZKAGI-ACZMJKKPSA-N 0.000 description 13
- WNZOCXUOGVYYBJ-CDMKHQONSA-N Gly-Phe-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=CC=C1)NC(=O)CN)O WNZOCXUOGVYYBJ-CDMKHQONSA-N 0.000 description 13
- PWWVAXIEGOYWEE-UHFFFAOYSA-N Isophenergan Chemical compound C1=CC=C2N(CC(C)N(C)C)C3=CC=CC=C3SC2=C1 PWWVAXIEGOYWEE-UHFFFAOYSA-N 0.000 description 13
- PNPYKQFJGRFYJE-GUBZILKMSA-N Lys-Ala-Glu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(O)=O PNPYKQFJGRFYJE-GUBZILKMSA-N 0.000 description 13
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 13
- KRPKYGOFYUNIGM-XVSYOHENSA-N Thr-Asp-Phe Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)O)N)O KRPKYGOFYUNIGM-XVSYOHENSA-N 0.000 description 13
- 210000002966 serum Anatomy 0.000 description 13
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 13
- QAPSNMNOIOSXSQ-YNEHKIRRSA-N 1-[(2r,4s,5r)-4-[tert-butyl(dimethyl)silyl]oxy-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O[Si](C)(C)C(C)(C)C)C1 QAPSNMNOIOSXSQ-YNEHKIRRSA-N 0.000 description 12
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 12
- FGWUALWGCZJQDJ-URLPEUOOSA-N Phe-Thr-Ile Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O FGWUALWGCZJQDJ-URLPEUOOSA-N 0.000 description 12
- 206010035226 Plasma cell myeloma Diseases 0.000 description 12
- RHAPJNVNWDBFQI-BQBZGAKWSA-N Ser-Pro-Gly Chemical compound OC[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O RHAPJNVNWDBFQI-BQBZGAKWSA-N 0.000 description 12
- XZKQVQKUZMAADP-IMJSIDKUSA-N Ser-Ser Chemical compound OC[C@H](N)C(=O)N[C@@H](CO)C(O)=O XZKQVQKUZMAADP-IMJSIDKUSA-N 0.000 description 12
- 229960003724 dimyristoylphosphatidylcholine Drugs 0.000 description 12
- 238000009396 hybridization Methods 0.000 description 12
- 201000000050 myeloid neoplasm Diseases 0.000 description 12
- 238000002823 phage display Methods 0.000 description 12
- 238000011282 treatment Methods 0.000 description 12
- LGFCAXJBAZESCF-ACZMJKKPSA-N Ala-Gln-Ala Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](C)C(O)=O LGFCAXJBAZESCF-ACZMJKKPSA-N 0.000 description 11
- MKJBPDLENBUHQU-CIUDSAMLSA-N Asn-Ser-Leu Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(O)=O MKJBPDLENBUHQU-CIUDSAMLSA-N 0.000 description 11
- OYTPNWYZORARHL-XHNCKOQMSA-N Gln-Ala-Pro Chemical compound C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCC(=O)N)N OYTPNWYZORARHL-XHNCKOQMSA-N 0.000 description 11
- LMDVGHQPPPLYAR-IHRRRGAJSA-N Leu-Val-His Chemical compound N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC1=CNC=N1)C(=O)O LMDVGHQPPPLYAR-IHRRRGAJSA-N 0.000 description 11
- WYBVBIHNJWOLCJ-UHFFFAOYSA-N N-L-arginyl-L-leucine Natural products CC(C)CC(C(O)=O)NC(=O)C(N)CCCN=C(N)N WYBVBIHNJWOLCJ-UHFFFAOYSA-N 0.000 description 11
- FMDHKPRACUXATF-ACZMJKKPSA-N Ser-Gln-Ser Chemical compound OC[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(O)=O FMDHKPRACUXATF-ACZMJKKPSA-N 0.000 description 11
- MECLEFZMPPOEAC-VOAKCMCISA-N Thr-Leu-Lys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)O)N)O MECLEFZMPPOEAC-VOAKCMCISA-N 0.000 description 11
- YOPQYBJJNSIQGZ-JNPHEJMOSA-N Thr-Tyr-Tyr Chemical compound C([C@H](NC(=O)[C@@H](N)[C@H](O)C)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(O)=O)C1=CC=C(O)C=C1 YOPQYBJJNSIQGZ-JNPHEJMOSA-N 0.000 description 11
- MNYNCKZAEIAONY-XGEHTFHBSA-N Thr-Val-Ser Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O MNYNCKZAEIAONY-XGEHTFHBSA-N 0.000 description 11
- BXJQKVDPRMLGKN-PMVMPFDFSA-N Tyr-Trp-Leu Chemical compound C([C@H](N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 BXJQKVDPRMLGKN-PMVMPFDFSA-N 0.000 description 11
- JSOXWWFKRJKTMT-WOPDTQHZSA-N Val-Val-Pro Chemical compound CC(C)[C@@H](C(=O)N[C@@H](C(C)C)C(=O)N1CCC[C@@H]1C(=O)O)N JSOXWWFKRJKTMT-WOPDTQHZSA-N 0.000 description 11
- 108010068265 aspartyltyrosine Proteins 0.000 description 11
- 201000010099 disease Diseases 0.000 description 11
- 108010050848 glycylleucine Proteins 0.000 description 11
- 230000005764 inhibitory process Effects 0.000 description 11
- 239000002502 liposome Substances 0.000 description 11
- 239000000463 material Substances 0.000 description 11
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 10
- OZHXXYOHPLLLMI-CIUDSAMLSA-N Cys-Lys-Ala Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(O)=O OZHXXYOHPLLLMI-CIUDSAMLSA-N 0.000 description 10
- HNAUFGBKJLTWQE-IFFSRLJSSA-N Gln-Val-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CCC(=O)N)N)O HNAUFGBKJLTWQE-IFFSRLJSSA-N 0.000 description 10
- LSIWVWRUTKPXDS-DCAQKATOSA-N Pro-Gln-Arg Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O LSIWVWRUTKPXDS-DCAQKATOSA-N 0.000 description 10
- 108010069020 alanyl-prolyl-glycine Proteins 0.000 description 10
- 238000006073 displacement reaction Methods 0.000 description 10
- 230000003993 interaction Effects 0.000 description 10
- 230000002829 reductive effect Effects 0.000 description 10
- -1 DMPC) Chemical compound 0.000 description 9
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 9
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 9
- CNGOEHJCLVCJHN-SRVKXCTJSA-N Lys-Pro-Glu Chemical compound NCCCC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O CNGOEHJCLVCJHN-SRVKXCTJSA-N 0.000 description 9
- 241001529936 Murinae Species 0.000 description 9
- 101100068676 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) gln-1 gene Proteins 0.000 description 9
- 238000002835 absorbance Methods 0.000 description 9
- 239000002299 complementary DNA Substances 0.000 description 9
- FSXRLASFHBWESK-UHFFFAOYSA-N dipeptide phenylalanyl-tyrosine Natural products C=1C=C(O)C=CC=1CC(C(O)=O)NC(=O)C(N)CC1=CC=CC=C1 FSXRLASFHBWESK-UHFFFAOYSA-N 0.000 description 9
- 239000003814 drug Substances 0.000 description 9
- 230000006870 function Effects 0.000 description 9
- 238000001727 in vivo Methods 0.000 description 9
- 235000012054 meals Nutrition 0.000 description 9
- 239000002609 medium Substances 0.000 description 9
- 108010070409 phenylalanyl-glycyl-glycine Proteins 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- 239000000523 sample Substances 0.000 description 9
- 239000003981 vehicle Substances 0.000 description 9
- ZDILXFDENZVOTL-BPNCWPANSA-N Ala-Val-Tyr Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O ZDILXFDENZVOTL-BPNCWPANSA-N 0.000 description 8
- GXMSVVBIAMWMKO-BQBZGAKWSA-N Asn-Arg-Gly Chemical compound NC(=O)C[C@H](N)C(=O)N[C@H](C(=O)NCC(O)=O)CCCN=C(N)N GXMSVVBIAMWMKO-BQBZGAKWSA-N 0.000 description 8
- ALMIMUZAWTUNIO-BZSNNMDCSA-N Asp-Tyr-Tyr Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O ALMIMUZAWTUNIO-BZSNNMDCSA-N 0.000 description 8
- 101100505161 Caenorhabditis elegans mel-32 gene Proteins 0.000 description 8
- 241000283707 Capra Species 0.000 description 8
- YXQCLIVLWCKCRS-RYUDHWBXSA-N Gln-Gly-Tyr Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)O)NC(=O)CNC(=O)[C@H](CCC(=O)N)N)O YXQCLIVLWCKCRS-RYUDHWBXSA-N 0.000 description 8
- LPIKVBWNNVFHCQ-GUBZILKMSA-N Gln-Ser-Leu Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(O)=O LPIKVBWNNVFHCQ-GUBZILKMSA-N 0.000 description 8
- UQJNXZSSGQIPIQ-FBCQKBJTSA-N Gly-Gly-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)CNC(=O)CN UQJNXZSSGQIPIQ-FBCQKBJTSA-N 0.000 description 8
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 8
- HPBCTWSUJOGJSH-MNXVOIDGSA-N Leu-Glu-Ile Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O HPBCTWSUJOGJSH-MNXVOIDGSA-N 0.000 description 8
- LINKCQUOMUDLKN-KATARQTJSA-N Leu-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CC(C)C)N)O LINKCQUOMUDLKN-KATARQTJSA-N 0.000 description 8
- MCIXMYKSPQUMJG-SRVKXCTJSA-N Phe-Ser-Ser Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O MCIXMYKSPQUMJG-SRVKXCTJSA-N 0.000 description 8
- NUQZCPSZHGIYTA-HKUYNNGSSA-N Tyr-Trp-Gly Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)NCC(=O)O)NC(=O)[C@H](CC3=CC=C(C=C3)O)N NUQZCPSZHGIYTA-HKUYNNGSSA-N 0.000 description 8
- 108010087924 alanylproline Proteins 0.000 description 8
- 108010069926 arginyl-glycyl-serine Proteins 0.000 description 8
- 239000012472 biological sample Substances 0.000 description 8
- 239000001963 growth medium Substances 0.000 description 8
- 238000011534 incubation Methods 0.000 description 8
- 108010034529 leucyl-lysine Proteins 0.000 description 8
- 108010090333 leucyl-lysyl-proline Proteins 0.000 description 8
- 210000004962 mammalian cell Anatomy 0.000 description 8
- 239000002245 particle Substances 0.000 description 8
- 238000003259 recombinant expression Methods 0.000 description 8
- 238000011160 research Methods 0.000 description 8
- 239000011780 sodium chloride Substances 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- 230000003612 virological effect Effects 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- ISCYZXFOCXWUJU-KZVJFYERSA-N Ala-Thr-Met Chemical compound [H]N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCSC)C(O)=O ISCYZXFOCXWUJU-KZVJFYERSA-N 0.000 description 7
- VAWNQIGQPUOPQW-ACZMJKKPSA-N Asp-Glu-Ala Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(O)=O VAWNQIGQPUOPQW-ACZMJKKPSA-N 0.000 description 7
- ZUNMTUPRQMWMHX-LSJOCFKGSA-N Asp-Val-Val Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(O)=O ZUNMTUPRQMWMHX-LSJOCFKGSA-N 0.000 description 7
- 238000012286 ELISA Assay Methods 0.000 description 7
- LGIKBBLQVSWUGK-DCAQKATOSA-N Gln-Leu-Gln Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O LGIKBBLQVSWUGK-DCAQKATOSA-N 0.000 description 7
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 7
- BYYNJRSNDARRBX-YFKPBYRVSA-N Gly-Gln-Gly Chemical compound NCC(=O)N[C@@H](CCC(N)=O)C(=O)NCC(O)=O BYYNJRSNDARRBX-YFKPBYRVSA-N 0.000 description 7
- ZLFNNVATRMCAKN-ZKWXMUAHSA-N Ile-Ser-Gly Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CO)C(=O)NCC(=O)O)N ZLFNNVATRMCAKN-ZKWXMUAHSA-N 0.000 description 7
- 206010028980 Neoplasm Diseases 0.000 description 7
- 108091028043 Nucleic acid sequence Proteins 0.000 description 7
- BIYWZVCPZIFGPY-QWRGUYRKSA-N Phe-Gly-Ser Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)NCC(=O)N[C@@H](CO)C(O)=O BIYWZVCPZIFGPY-QWRGUYRKSA-N 0.000 description 7
- IOVHBRCQOGWAQH-ZKWXMUAHSA-N Ser-Gly-Ile Chemical compound [H]N[C@@H](CO)C(=O)NCC(=O)N[C@@H]([C@@H](C)CC)C(O)=O IOVHBRCQOGWAQH-ZKWXMUAHSA-N 0.000 description 7
- JCLAFVNDBJMLBC-JBDRJPRFSA-N Ser-Ser-Ile Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O JCLAFVNDBJMLBC-JBDRJPRFSA-N 0.000 description 7
- PZTZYZUTCPZWJH-FXQIFTODSA-N Val-Ser-Ser Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)O)N PZTZYZUTCPZWJH-FXQIFTODSA-N 0.000 description 7
- 241000700605 Viruses Species 0.000 description 7
- 125000000539 amino acid group Chemical group 0.000 description 7
- 238000012512 characterization method Methods 0.000 description 7
- 239000000839 emulsion Substances 0.000 description 7
- 238000009472 formulation Methods 0.000 description 7
- 238000000338 in vitro Methods 0.000 description 7
- 239000003112 inhibitor Substances 0.000 description 7
- 239000008176 lyophilized powder Substances 0.000 description 7
- 235000013336 milk Nutrition 0.000 description 7
- 239000008267 milk Substances 0.000 description 7
- 210000004080 milk Anatomy 0.000 description 7
- 239000000546 pharmaceutical excipient Substances 0.000 description 7
- 108010051242 phenylalanylserine Proteins 0.000 description 7
- 108010070643 prolylglutamic acid Proteins 0.000 description 7
- 230000006798 recombination Effects 0.000 description 7
- 238000005215 recombination Methods 0.000 description 7
- 238000012216 screening Methods 0.000 description 7
- 239000007787 solid Substances 0.000 description 7
- 239000007790 solid phase Substances 0.000 description 7
- 229940124597 therapeutic agent Drugs 0.000 description 7
- 210000001519 tissue Anatomy 0.000 description 7
- 238000005406 washing Methods 0.000 description 7
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 6
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 6
- DNPCBMNFQVTHMA-DCAQKATOSA-N Glu-Leu-Gln Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O DNPCBMNFQVTHMA-DCAQKATOSA-N 0.000 description 6
- YWAQATDNEKZFFK-BYPYZUCNSA-N Gly-Gly-Ser Chemical compound NCC(=O)NCC(=O)N[C@@H](CO)C(O)=O YWAQATDNEKZFFK-BYPYZUCNSA-N 0.000 description 6
- YXTFLTJYLIAZQG-FJXKBIBVSA-N Gly-Thr-Arg Chemical compound NCC(=O)N[C@@H]([C@H](O)C)C(=O)N[C@H](C(O)=O)CCCN=C(N)N YXTFLTJYLIAZQG-FJXKBIBVSA-N 0.000 description 6
- 108010001336 Horseradish Peroxidase Proteins 0.000 description 6
- JMNRXRPBHFGXQX-GUBZILKMSA-N Lys-Ser-Glu Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCC(O)=O JMNRXRPBHFGXQX-GUBZILKMSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- BODDREDDDRZUCF-QTKMDUPCSA-N Pro-His-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CN=CN1)NC(=O)[C@@H]2CCCN2)O BODDREDDDRZUCF-QTKMDUPCSA-N 0.000 description 6
- UBRMZSHOOIVJPW-SRVKXCTJSA-N Ser-Leu-Lys Chemical compound OC[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(O)=O UBRMZSHOOIVJPW-SRVKXCTJSA-N 0.000 description 6
- NJGMALCNYAMYCB-JRQIVUDYSA-N Thr-Tyr-Asn Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC(N)=O)C(O)=O NJGMALCNYAMYCB-JRQIVUDYSA-N 0.000 description 6
- JWHOIHCOHMZSAR-QWRGUYRKSA-N Tyr-Asp-Gly Chemical compound OC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 JWHOIHCOHMZSAR-QWRGUYRKSA-N 0.000 description 6
- MWUYSCVVPVITMW-IGNZVWTISA-N Tyr-Tyr-Ala Chemical compound C([C@@H](C(=O)N[C@@H](C)C(O)=O)NC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=C(O)C=C1 MWUYSCVVPVITMW-IGNZVWTISA-N 0.000 description 6
- HQYVQDRYODWONX-DCAQKATOSA-N Val-His-Ser Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)N[C@@H](CO)C(=O)O)N HQYVQDRYODWONX-DCAQKATOSA-N 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- 108010077245 asparaginyl-proline Proteins 0.000 description 6
- 108010047857 aspartylglycine Proteins 0.000 description 6
- 238000001514 detection method Methods 0.000 description 6
- 238000010790 dilution Methods 0.000 description 6
- 239000012895 dilution Substances 0.000 description 6
- 235000021588 free fatty acids Nutrition 0.000 description 6
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 description 6
- VPZXBVLAVMBEQI-UHFFFAOYSA-N glycyl-DL-alpha-alanine Natural products OC(=O)C(C)NC(=O)CN VPZXBVLAVMBEQI-UHFFFAOYSA-N 0.000 description 6
- 229940028435 intralipid Drugs 0.000 description 6
- 238000010172 mouse model Methods 0.000 description 6
- 150000005830 nonesterified fatty acids Chemical class 0.000 description 6
- 239000004006 olive oil Substances 0.000 description 6
- 235000008390 olive oil Nutrition 0.000 description 6
- 238000003127 radioimmunoassay Methods 0.000 description 6
- 238000003786 synthesis reaction Methods 0.000 description 6
- 230000000699 topical effect Effects 0.000 description 6
- BUDNAJYVCUHLSV-ZLUOBGJFSA-N Ala-Asp-Ser Chemical compound C[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O BUDNAJYVCUHLSV-ZLUOBGJFSA-N 0.000 description 5
- IFTVANMRTIHKML-WDSKDSINSA-N Ala-Gln-Gly Chemical compound C[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)NCC(O)=O IFTVANMRTIHKML-WDSKDSINSA-N 0.000 description 5
- JPPLRQVZMZFOSX-UWJYBYFXSA-N Asn-Tyr-Ala Chemical compound NC(=O)C[C@H](N)C(=O)N[C@H](C(=O)N[C@@H](C)C(O)=O)CC1=CC=C(O)C=C1 JPPLRQVZMZFOSX-UWJYBYFXSA-N 0.000 description 5
- 108020004705 Codon Proteins 0.000 description 5
- 102000004190 Enzymes Human genes 0.000 description 5
- 108090000790 Enzymes Proteins 0.000 description 5
- 241000588724 Escherichia coli Species 0.000 description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- ZFBBMCKQSNJZSN-AUTRQRHGSA-N Gln-Val-Gln Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O ZFBBMCKQSNJZSN-AUTRQRHGSA-N 0.000 description 5
- SBCYJMOOHUDWDA-NUMRIWBASA-N Glu-Asp-Thr Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O SBCYJMOOHUDWDA-NUMRIWBASA-N 0.000 description 5
- YQPFCZVKMUVZIN-AUTRQRHGSA-N Glu-Val-Gln Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O YQPFCZVKMUVZIN-AUTRQRHGSA-N 0.000 description 5
- 108010091358 Hypoxanthine Phosphoribosyltransferase Proteins 0.000 description 5
- ASCFJMSGKUIRDU-ZPFDUUQYSA-N Ile-Arg-Gln Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(O)=O ASCFJMSGKUIRDU-ZPFDUUQYSA-N 0.000 description 5
- GQFDWEDHOQRNLC-QWRGUYRKSA-N Lys-Gly-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN GQFDWEDHOQRNLC-QWRGUYRKSA-N 0.000 description 5
- YTJFXEDRUOQGSP-DCAQKATOSA-N Lys-Pro-Ser Chemical compound [H]N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O YTJFXEDRUOQGSP-DCAQKATOSA-N 0.000 description 5
- 241000282567 Macaca fascicularis Species 0.000 description 5
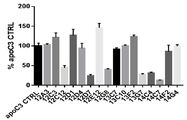
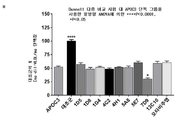
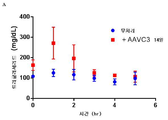
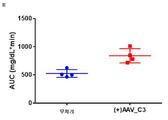
- 101100323491 Macaca fascicularis APOC3 gene Proteins 0.000 description 5
- 108091034117 Oligonucleotide Proteins 0.000 description 5
- 239000004698 Polyethylene Substances 0.000 description 5
- 239000002202 Polyethylene glycol Substances 0.000 description 5
- WTWGOQRNRFHFQD-JBDRJPRFSA-N Ser-Ala-Ile Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O WTWGOQRNRFHFQD-JBDRJPRFSA-N 0.000 description 5
- KDGARKCAKHBEDB-NKWVEPMBSA-N Ser-Gly-Pro Chemical compound C1C[C@@H](N(C1)C(=O)CNC(=O)[C@H](CO)N)C(=O)O KDGARKCAKHBEDB-NKWVEPMBSA-N 0.000 description 5
- YOOAQCZYZHGUAZ-KATARQTJSA-N Thr-Leu-Ser Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(O)=O YOOAQCZYZHGUAZ-KATARQTJSA-N 0.000 description 5
- MXDOAJQRJBMGMO-FJXKBIBVSA-N Thr-Pro-Gly Chemical compound C[C@@H](O)[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O MXDOAJQRJBMGMO-FJXKBIBVSA-N 0.000 description 5
- KZTLZZQTJMCGIP-ZJDVBMNYSA-N Thr-Val-Thr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O KZTLZZQTJMCGIP-ZJDVBMNYSA-N 0.000 description 5
- CPGJELLYDQEDRK-NAKRPEOUSA-N Val-Ile-Ala Chemical compound CC[C@H](C)[C@H](NC(=O)[C@@H](N)C(C)C)C(=O)N[C@@H](C)C(O)=O CPGJELLYDQEDRK-NAKRPEOUSA-N 0.000 description 5
- CKTMJBPRVQWPHU-JSGCOSHPSA-N Val-Phe-Gly Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)NCC(=O)O)N CKTMJBPRVQWPHU-JSGCOSHPSA-N 0.000 description 5
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 5
- 108010005233 alanylglutamic acid Proteins 0.000 description 5
- 108010047495 alanylglycine Proteins 0.000 description 5
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 239000012707 chemical precursor Substances 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- 239000003153 chemical reaction reagent Substances 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- 125000004122 cyclic group Chemical group 0.000 description 5
- 230000007423 decrease Effects 0.000 description 5
- 208000035475 disorder Diseases 0.000 description 5
- 238000010494 dissociation reaction Methods 0.000 description 5
- 230000005593 dissociations Effects 0.000 description 5
- 229940088598 enzyme Drugs 0.000 description 5
- 108010010147 glycylglutamine Proteins 0.000 description 5
- 239000013642 negative control Substances 0.000 description 5
- 239000013612 plasmid Substances 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 238000000159 protein binding assay Methods 0.000 description 5
- 238000000746 purification Methods 0.000 description 5
- 230000002285 radioactive effect Effects 0.000 description 5
- 108010026333 seryl-proline Proteins 0.000 description 5
- 108010071207 serylmethionine Proteins 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 108010072986 threonyl-seryl-lysine Proteins 0.000 description 5
- 238000013518 transcription Methods 0.000 description 5
- 230000035897 transcription Effects 0.000 description 5
- 238000013519 translation Methods 0.000 description 5
- 108010020532 tyrosyl-proline Proteins 0.000 description 5
- 108010003137 tyrosyltyrosine Proteins 0.000 description 5
- OBVSBEYOMDWLRJ-BFHQHQDPSA-N Ala-Gly-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)CNC(=O)[C@H](C)N OBVSBEYOMDWLRJ-BFHQHQDPSA-N 0.000 description 4
- 244000303258 Annona diversifolia Species 0.000 description 4
- 235000002198 Annona diversifolia Nutrition 0.000 description 4
- 208000002267 Anti-neutrophil cytoplasmic antibody-associated vasculitis Diseases 0.000 description 4
- QYRMBFWDSFGSFC-OLHMAJIHSA-N Asn-Thr-Asn Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CC(=O)N)N)O QYRMBFWDSFGSFC-OLHMAJIHSA-N 0.000 description 4
- QXHVOUSPVAWEMX-ZLUOBGJFSA-N Asp-Asp-Ser Chemical compound OC(=O)C[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O QXHVOUSPVAWEMX-ZLUOBGJFSA-N 0.000 description 4
- GCACQYDBDHRVGE-LKXGYXEUSA-N Asp-Thr-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H]([C@H](O)C)NC(=O)[C@@H](N)CC(O)=O GCACQYDBDHRVGE-LKXGYXEUSA-N 0.000 description 4
- AMRLSQGGERHDHJ-FXQIFTODSA-N Cys-Ala-Arg Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O AMRLSQGGERHDHJ-FXQIFTODSA-N 0.000 description 4
- SHZGCJCMOBCMKK-UHFFFAOYSA-N D-mannomethylose Natural products CC1OC(O)C(O)C(O)C1O SHZGCJCMOBCMKK-UHFFFAOYSA-N 0.000 description 4
- 101100228149 Drosophila melanogaster Trl gene Proteins 0.000 description 4
- PNNNRSAQSRJVSB-SLPGGIOYSA-N Fucose Natural products C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C=O PNNNRSAQSRJVSB-SLPGGIOYSA-N 0.000 description 4
- NNCSJUBVFBDDLC-YUMQZZPRSA-N Gly-Leu-Ser Chemical compound NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(O)=O NNCSJUBVFBDDLC-YUMQZZPRSA-N 0.000 description 4
- LCRDMSSAKLTKBU-ZDLURKLDSA-N Gly-Ser-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)CN LCRDMSSAKLTKBU-ZDLURKLDSA-N 0.000 description 4
- 101100370408 Helicobacter pylori (strain ATCC 700392 / 26695) trl gene Proteins 0.000 description 4
- LVWIJITYHRZHBO-IXOXFDKPSA-N His-Leu-Thr Chemical compound [H]N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O LVWIJITYHRZHBO-IXOXFDKPSA-N 0.000 description 4
- 102100029098 Hypoxanthine-guanine phosphoribosyltransferase Human genes 0.000 description 4
- JHNJNTMTZHEDLJ-NAKRPEOUSA-N Ile-Ser-Arg Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O JHNJNTMTZHEDLJ-NAKRPEOUSA-N 0.000 description 4
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 4
- SHZGCJCMOBCMKK-DHVFOXMCSA-N L-fucopyranose Chemical compound C[C@@H]1OC(O)[C@@H](O)[C@H](O)[C@@H]1O SHZGCJCMOBCMKK-DHVFOXMCSA-N 0.000 description 4
- VCHVSKNMTXWIIP-SRVKXCTJSA-N Leu-Lys-Ser Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(O)=O VCHVSKNMTXWIIP-SRVKXCTJSA-N 0.000 description 4
- SVBJIZVVYJYGLA-DCAQKATOSA-N Leu-Ser-Val Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O SVBJIZVVYJYGLA-DCAQKATOSA-N 0.000 description 4
- WXJKFRMKJORORD-DCAQKATOSA-N Lys-Arg-Ala Chemical compound NC(=N)NCCC[C@@H](C(=O)N[C@@H](C)C(O)=O)NC(=O)[C@@H](N)CCCCN WXJKFRMKJORORD-DCAQKATOSA-N 0.000 description 4
- WUGMRIBZSVSJNP-UHFFFAOYSA-N N-L-alanyl-L-tryptophan Natural products C1=CC=C2C(CC(NC(=O)C(N)C)C(O)=O)=CNC2=C1 WUGMRIBZSVSJNP-UHFFFAOYSA-N 0.000 description 4
- 101100342977 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) leu-1 gene Proteins 0.000 description 4
- 238000012408 PCR amplification Methods 0.000 description 4
- 206010033645 Pancreatitis Diseases 0.000 description 4
- YYKZDTVQHTUKDW-RYUDHWBXSA-N Phe-Gly-Gln Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)NCC(=O)N[C@@H](CCC(=O)N)C(=O)O)N YYKZDTVQHTUKDW-RYUDHWBXSA-N 0.000 description 4
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Natural products OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- ZSKJPKFTPQCPIH-RCWTZXSCSA-N Pro-Arg-Thr Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(O)=O ZSKJPKFTPQCPIH-RCWTZXSCSA-N 0.000 description 4
- AQSMZTIEJMZQEC-DCAQKATOSA-N Pro-His-Ser Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CC2=CN=CN2)C(=O)N[C@@H](CO)C(=O)O AQSMZTIEJMZQEC-DCAQKATOSA-N 0.000 description 4
- LEIKGVHQTKHOLM-IUCAKERBSA-N Pro-Pro-Gly Chemical compound OC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H]1NCCC1 LEIKGVHQTKHOLM-IUCAKERBSA-N 0.000 description 4
- 241000700159 Rattus Species 0.000 description 4
- 108020004511 Recombinant DNA Proteins 0.000 description 4
- LQESNKGTTNHZPZ-GHCJXIJMSA-N Ser-Ile-Asn Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(O)=O LQESNKGTTNHZPZ-GHCJXIJMSA-N 0.000 description 4
- GJFYFGOEWLDQGW-GUBZILKMSA-N Ser-Leu-Gln Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CO)N GJFYFGOEWLDQGW-GUBZILKMSA-N 0.000 description 4
- PPCZVWHJWJFTFN-ZLUOBGJFSA-N Ser-Ser-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(O)=O PPCZVWHJWJFTFN-ZLUOBGJFSA-N 0.000 description 4
- 108010090804 Streptavidin Proteins 0.000 description 4
- JEDIEMIJYSRUBB-FOHZUACHSA-N Thr-Asp-Gly Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(O)=O JEDIEMIJYSRUBB-FOHZUACHSA-N 0.000 description 4
- DKDHTRVDOUZZTP-IFFSRLJSSA-N Thr-Gln-Val Chemical compound CC(C)[C@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)[C@@H](C)O)C(O)=O DKDHTRVDOUZZTP-IFFSRLJSSA-N 0.000 description 4
- XVHAUVJXBFGUPC-RPTUDFQQSA-N Thr-Tyr-Phe Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O XVHAUVJXBFGUPC-RPTUDFQQSA-N 0.000 description 4
- CYCGARJWIQWPQM-YJRXYDGGSA-N Thr-Tyr-Ser Chemical compound C[C@@H](O)[C@H]([NH3+])C(=O)N[C@H](C(=O)N[C@@H](CO)C([O-])=O)CC1=CC=C(O)C=C1 CYCGARJWIQWPQM-YJRXYDGGSA-N 0.000 description 4
- UMSZZGTXGKHTFJ-SRVKXCTJSA-N Tyr-Ser-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 UMSZZGTXGKHTFJ-SRVKXCTJSA-N 0.000 description 4
- QHFQQRKNGCXTHL-AUTRQRHGSA-N Val-Gln-Glu Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(O)=O QHFQQRKNGCXTHL-AUTRQRHGSA-N 0.000 description 4
- GBIUHAYJGWVNLN-UHFFFAOYSA-N Val-Ser-Pro Natural products CC(C)C(N)C(=O)NC(CO)C(=O)N1CCCC1C(O)=O GBIUHAYJGWVNLN-UHFFFAOYSA-N 0.000 description 4
- DVLWZWNAQUBZBC-ZNSHCXBVSA-N Val-Thr-Pro Chemical compound C[C@H]([C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](C(C)C)N)O DVLWZWNAQUBZBC-ZNSHCXBVSA-N 0.000 description 4
- OFTXTCGQJXTNQS-XGEHTFHBSA-N Val-Thr-Ser Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](C(C)C)N)O OFTXTCGQJXTNQS-XGEHTFHBSA-N 0.000 description 4
- 239000000443 aerosol Substances 0.000 description 4
- 108010041407 alanylaspartic acid Proteins 0.000 description 4
- 201000011510 cancer Diseases 0.000 description 4
- 230000001413 cellular effect Effects 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 4
- 239000000460 chlorine Substances 0.000 description 4
- 210000001612 chondrocyte Anatomy 0.000 description 4
- 238000010367 cloning Methods 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- 239000008121 dextrose Substances 0.000 description 4
- 229910052731 fluorine Inorganic materials 0.000 description 4
- 108020001507 fusion proteins Proteins 0.000 description 4
- 102000037865 fusion proteins Human genes 0.000 description 4
- 208000015181 infectious disease Diseases 0.000 description 4
- 238000013507 mapping Methods 0.000 description 4
- 239000011159 matrix material Substances 0.000 description 4
- 230000007246 mechanism Effects 0.000 description 4
- 108020004999 messenger RNA Proteins 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 238000010369 molecular cloning Methods 0.000 description 4
- 230000036961 partial effect Effects 0.000 description 4
- 230000036470 plasma concentration Effects 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 230000002265 prevention Effects 0.000 description 4
- 108010087846 prolyl-prolyl-glycine Proteins 0.000 description 4
- 230000000069 prophylactic effect Effects 0.000 description 4
- 108010029895 rubimetide Proteins 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- 208000024891 symptom Diseases 0.000 description 4
- 238000011830 transgenic mouse model Methods 0.000 description 4
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 3
- MEFILNJXAVSUTO-JXUBOQSCSA-N Ala-Leu-Thr Chemical compound C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O MEFILNJXAVSUTO-JXUBOQSCSA-N 0.000 description 3
- NLOMBWNGESDVJU-GUBZILKMSA-N Ala-Met-Arg Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O NLOMBWNGESDVJU-GUBZILKMSA-N 0.000 description 3
- MAZZQZWCCYJQGZ-GUBZILKMSA-N Ala-Pro-Arg Chemical compound [H]N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(O)=O MAZZQZWCCYJQGZ-GUBZILKMSA-N 0.000 description 3
- INOIAEUXVVNJKA-XGEHTFHBSA-N Arg-Thr-Ser Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(O)=O INOIAEUXVVNJKA-XGEHTFHBSA-N 0.000 description 3
- VDCIPFYVCICPEC-FXQIFTODSA-N Asn-Arg-Ala Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(O)=O VDCIPFYVCICPEC-FXQIFTODSA-N 0.000 description 3
- HOIFSHOLNKQCSA-FXQIFTODSA-N Asn-Arg-Asp Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(O)=O HOIFSHOLNKQCSA-FXQIFTODSA-N 0.000 description 3
- VJTWLBMESLDOMK-WDSKDSINSA-N Asn-Gln-Gly Chemical compound NC(=O)C[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)NCC(O)=O VJTWLBMESLDOMK-WDSKDSINSA-N 0.000 description 3
- RCFGLXMZDYNRSC-CIUDSAMLSA-N Asn-Lys-Ala Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(O)=O RCFGLXMZDYNRSC-CIUDSAMLSA-N 0.000 description 3
- MYTHOBCLNIOFBL-SRVKXCTJSA-N Asn-Ser-Tyr Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O MYTHOBCLNIOFBL-SRVKXCTJSA-N 0.000 description 3
- WLVLIYYBPPONRJ-GCJQMDKQSA-N Asn-Thr-Ala Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(O)=O WLVLIYYBPPONRJ-GCJQMDKQSA-N 0.000 description 3
- QNMKWNONJGKJJC-NHCYSSNCSA-N Asp-Leu-Val Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(O)=O QNMKWNONJGKJJC-NHCYSSNCSA-N 0.000 description 3
- JDDYEZGPYBBPBN-JRQIVUDYSA-N Asp-Thr-Tyr Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O JDDYEZGPYBBPBN-JRQIVUDYSA-N 0.000 description 3
- 241000201370 Autographa californica nucleopolyhedrovirus Species 0.000 description 3
- 102100021277 Beta-secretase 2 Human genes 0.000 description 3
- 101710150190 Beta-secretase 2 Proteins 0.000 description 3
- 101100512078 Caenorhabditis elegans lys-1 gene Proteins 0.000 description 3
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 3
- 241000702421 Dependoparvovirus Species 0.000 description 3
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 3
- JSYULGSPLTZDHM-NRPADANISA-N Gln-Ala-Val Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(O)=O JSYULGSPLTZDHM-NRPADANISA-N 0.000 description 3
- JEFZIKRIDLHOIF-BYPYZUCNSA-N Gln-Gly Chemical compound NC(=O)CC[C@H](N)C(=O)NCC(O)=O JEFZIKRIDLHOIF-BYPYZUCNSA-N 0.000 description 3
- XZLLTYBONVKGLO-SDDRHHMPSA-N Gln-Lys-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(=O)N)N)C(=O)O XZLLTYBONVKGLO-SDDRHHMPSA-N 0.000 description 3
- HMIXCETWRYDVMO-GUBZILKMSA-N Gln-Pro-Glu Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O HMIXCETWRYDVMO-GUBZILKMSA-N 0.000 description 3
- VYOILACOFPPNQH-UMNHJUIQSA-N Gln-Val-Pro Chemical compound CC(C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCC(=O)N)N VYOILACOFPPNQH-UMNHJUIQSA-N 0.000 description 3
- BIYNPVYAZOUVFQ-CIUDSAMLSA-N Glu-Pro-Ser Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O BIYNPVYAZOUVFQ-CIUDSAMLSA-N 0.000 description 3
- RFTVTKBHDXCEEX-WDSKDSINSA-N Glu-Ser-Gly Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)NCC(O)=O RFTVTKBHDXCEEX-WDSKDSINSA-N 0.000 description 3
- HHSKZJZWQFPSKN-AVGNSLFASA-N Glu-Tyr-Asp Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC(O)=O)C(O)=O HHSKZJZWQFPSKN-AVGNSLFASA-N 0.000 description 3
- 108010024636 Glutathione Proteins 0.000 description 3
- WMGHDYWNHNLGBV-ONGXEEELSA-N Gly-Phe-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)CN)CC1=CC=CC=C1 WMGHDYWNHNLGBV-ONGXEEELSA-N 0.000 description 3
- RHRLHXQWHCNJKR-PMVVWTBXSA-N Gly-Thr-His Chemical compound NCC(=O)N[C@@H]([C@H](O)C)C(=O)N[C@H](C(O)=O)CC1=CN=CN1 RHRLHXQWHCNJKR-PMVVWTBXSA-N 0.000 description 3
- XHVONGZZVUUORG-WEDXCCLWSA-N Gly-Thr-Lys Chemical compound NCC(=O)N[C@@H]([C@H](O)C)C(=O)N[C@H](C(O)=O)CCCCN XHVONGZZVUUORG-WEDXCCLWSA-N 0.000 description 3
- KOYUSMBPJOVSOO-XEGUGMAKSA-N Gly-Tyr-Ile Chemical compound [H]NCC(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O KOYUSMBPJOVSOO-XEGUGMAKSA-N 0.000 description 3
- 239000004471 Glycine Substances 0.000 description 3
- 241000238631 Hexapoda Species 0.000 description 3
- CDGLBYSAZFIIJO-RCOVLWMOSA-N Ile-Gly-Gly Chemical compound CC[C@H](C)[C@H]([NH3+])C(=O)NCC(=O)NCC([O-])=O CDGLBYSAZFIIJO-RCOVLWMOSA-N 0.000 description 3
- FBGXMKUWQFPHFB-JBDRJPRFSA-N Ile-Ser-Cys Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O)N FBGXMKUWQFPHFB-JBDRJPRFSA-N 0.000 description 3
- RKQAYOWLSFLJEE-SVSWQMSJSA-N Ile-Thr-Cys Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)O)N RKQAYOWLSFLJEE-SVSWQMSJSA-N 0.000 description 3
- YBKKLDBBPFIXBQ-MBLNEYKQSA-N Ile-Thr-Gly Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)O)N YBKKLDBBPFIXBQ-MBLNEYKQSA-N 0.000 description 3
- NURNJECQNNCRBK-FLBSBUHZSA-N Ile-Thr-Thr Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O NURNJECQNNCRBK-FLBSBUHZSA-N 0.000 description 3
- NXRNRBOKDBIVKQ-CXTHYWKRSA-N Ile-Tyr-Tyr Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)O)N NXRNRBOKDBIVKQ-CXTHYWKRSA-N 0.000 description 3
- 108700005091 Immunoglobulin Genes Proteins 0.000 description 3
- RVVBWTWPNFDYBE-SRVKXCTJSA-N Leu-Glu-Arg Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O RVVBWTWPNFDYBE-SRVKXCTJSA-N 0.000 description 3
- JFSGIJSCJFQGSZ-MXAVVETBSA-N Leu-Ile-His Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)NC(=O)[C@H](CC(C)C)N JFSGIJSCJFQGSZ-MXAVVETBSA-N 0.000 description 3
- BRTVHXHCUSXYRI-CIUDSAMLSA-N Leu-Ser-Ser Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O BRTVHXHCUSXYRI-CIUDSAMLSA-N 0.000 description 3
- AIQWYVFNBNNOLU-RHYQMDGZSA-N Leu-Thr-Val Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O AIQWYVFNBNNOLU-RHYQMDGZSA-N 0.000 description 3
- UWKNTTJNVSYXPC-CIUDSAMLSA-N Lys-Ala-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCCCN UWKNTTJNVSYXPC-CIUDSAMLSA-N 0.000 description 3
- YKIRNDPUWONXQN-GUBZILKMSA-N Lys-Asn-Gln Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CCC(=O)N)C(=O)O)N YKIRNDPUWONXQN-GUBZILKMSA-N 0.000 description 3
- SBQDRNOLGSYHQA-YUMQZZPRSA-N Lys-Ser-Gly Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)NCC(O)=O SBQDRNOLGSYHQA-YUMQZZPRSA-N 0.000 description 3
- 241000699660 Mus musculus Species 0.000 description 3
- 101100491390 Mus musculus Apob gene Proteins 0.000 description 3
- 241000283973 Oryctolagus cuniculus Species 0.000 description 3
- NAXPHWZXEXNDIW-JTQLQIEISA-N Phe-Gly-Gly Chemical compound OC(=O)CNC(=O)CNC(=O)[C@@H](N)CC1=CC=CC=C1 NAXPHWZXEXNDIW-JTQLQIEISA-N 0.000 description 3
- YMIZSYUAZJSOFL-SRVKXCTJSA-N Phe-Ser-Asn Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(O)=O YMIZSYUAZJSOFL-SRVKXCTJSA-N 0.000 description 3
- BPCLGWHVPVTTFM-QWRGUYRKSA-N Phe-Ser-Gly Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)NCC(O)=O BPCLGWHVPVTTFM-QWRGUYRKSA-N 0.000 description 3
- JARJPEMLQAWNBR-GUBZILKMSA-N Pro-Asp-Arg Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O JARJPEMLQAWNBR-GUBZILKMSA-N 0.000 description 3
- HJSCRFZVGXAGNG-SRVKXCTJSA-N Pro-Gln-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H]1CCCN1 HJSCRFZVGXAGNG-SRVKXCTJSA-N 0.000 description 3
- KHRLUIPIMIQFGT-AVGNSLFASA-N Pro-Val-Leu Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O KHRLUIPIMIQFGT-AVGNSLFASA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 3
- VQBLHWSPVYYZTB-DCAQKATOSA-N Ser-Arg-His Chemical compound C1=C(NC=N1)C[C@@H](C(=O)O)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CO)N VQBLHWSPVYYZTB-DCAQKATOSA-N 0.000 description 3
- BNFVPSRLHHPQKS-WHFBIAKZSA-N Ser-Asp-Gly Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(O)=O BNFVPSRLHHPQKS-WHFBIAKZSA-N 0.000 description 3
- RQXDSYQXBCRXBT-GUBZILKMSA-N Ser-Met-Arg Chemical compound OC[C@H](N)C(=O)N[C@@H](CCSC)C(=O)N[C@H](C(O)=O)CCCN=C(N)N RQXDSYQXBCRXBT-GUBZILKMSA-N 0.000 description 3
- WLJPJRGQRNCIQS-ZLUOBGJFSA-N Ser-Ser-Asn Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(O)=O WLJPJRGQRNCIQS-ZLUOBGJFSA-N 0.000 description 3
- JURQXQBJKUHGJS-UHFFFAOYSA-N Ser-Ser-Ser-Ser Chemical compound OCC(N)C(=O)NC(CO)C(=O)NC(CO)C(=O)NC(CO)C(O)=O JURQXQBJKUHGJS-UHFFFAOYSA-N 0.000 description 3
- SNXUIBACCONSOH-BWBBJGPYSA-N Ser-Thr-Ser Chemical compound OC[C@H](N)C(=O)N[C@@H]([C@H](O)C)C(=O)N[C@@H](CO)C(O)=O SNXUIBACCONSOH-BWBBJGPYSA-N 0.000 description 3
- FZNNGIHSIPKFRE-QEJZJMRPSA-N Ser-Trp-Gln Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CCC(N)=O)C(O)=O FZNNGIHSIPKFRE-QEJZJMRPSA-N 0.000 description 3
- VVKVHAOOUGNDPJ-SRVKXCTJSA-N Ser-Tyr-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CO)C(O)=O VVKVHAOOUGNDPJ-SRVKXCTJSA-N 0.000 description 3
- VRUFCJZQDACGLH-UVOCVTCTSA-N Thr-Leu-Thr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O VRUFCJZQDACGLH-UVOCVTCTSA-N 0.000 description 3
- DIHPMRTXPYMDJZ-KAOXEZKKSA-N Thr-Tyr-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N2CCC[C@@H]2C(=O)O)N)O DIHPMRTXPYMDJZ-KAOXEZKKSA-N 0.000 description 3
- RERRMBXDSFMBQE-ZFWWWQNUSA-N Trp-Met-Gly Chemical compound CSCC[C@@H](C(=O)NCC(=O)O)NC(=O)[C@H](CC1=CNC2=CC=CC=C21)N RERRMBXDSFMBQE-ZFWWWQNUSA-N 0.000 description 3
- JQTYTBPCSOAZHI-FXQIFTODSA-N Val-Ser-Cys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O)N JQTYTBPCSOAZHI-FXQIFTODSA-N 0.000 description 3
- UQMPYVLTQCGRSK-IFFSRLJSSA-N Val-Thr-Gln Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](C(C)C)N)O UQMPYVLTQCGRSK-IFFSRLJSSA-N 0.000 description 3
- HTONZBWRYUKUKC-RCWTZXSCSA-N Val-Thr-Val Chemical compound CC(C)[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O HTONZBWRYUKUKC-RCWTZXSCSA-N 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 230000002378 acidificating effect Effects 0.000 description 3
- 230000003321 amplification Effects 0.000 description 3
- 238000010171 animal model Methods 0.000 description 3
- 239000003963 antioxidant agent Substances 0.000 description 3
- 235000006708 antioxidants Nutrition 0.000 description 3
- 230000002238 attenuated effect Effects 0.000 description 3
- 230000001580 bacterial effect Effects 0.000 description 3
- 229960002685 biotin Drugs 0.000 description 3
- 235000020958 biotin Nutrition 0.000 description 3
- 239000011616 biotin Substances 0.000 description 3
- 239000006172 buffering agent Substances 0.000 description 3
- 239000002738 chelating agent Substances 0.000 description 3
- 238000009232 chiropractic Methods 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 108010016616 cysteinylglycine Proteins 0.000 description 3
- 229910052805 deuterium Inorganic materials 0.000 description 3
- 239000000975 dye Substances 0.000 description 3
- 210000001671 embryonic stem cell Anatomy 0.000 description 3
- 239000003995 emulsifying agent Substances 0.000 description 3
- 239000000499 gel Substances 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 239000008103 glucose Substances 0.000 description 3
- 108010080575 glutamyl-aspartyl-alanine Proteins 0.000 description 3
- 229960003180 glutathione Drugs 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 230000036541 health Effects 0.000 description 3
- 108010025306 histidylleucine Proteins 0.000 description 3
- 238000002744 homologous recombination Methods 0.000 description 3
- 230000006801 homologous recombination Effects 0.000 description 3
- 230000001900 immune effect Effects 0.000 description 3
- 230000003053 immunization Effects 0.000 description 3
- 238000002649 immunization Methods 0.000 description 3
- 238000003018 immunoassay Methods 0.000 description 3
- 108010044374 isoleucyl-tyrosine Proteins 0.000 description 3
- 108010060857 isoleucyl-valyl-tyrosine Proteins 0.000 description 3
- 230000000670 limiting effect Effects 0.000 description 3
- 108010025153 lysyl-alanyl-alanine Proteins 0.000 description 3
- 239000003550 marker Substances 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 238000012544 monitoring process Methods 0.000 description 3
- 230000035772 mutation Effects 0.000 description 3
- 230000007935 neutral effect Effects 0.000 description 3
- 238000003199 nucleic acid amplification method Methods 0.000 description 3
- 238000007911 parenteral administration Methods 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 235000020183 skimmed milk Nutrition 0.000 description 3
- 230000009870 specific binding Effects 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 238000010186 staining Methods 0.000 description 3
- 238000007920 subcutaneous administration Methods 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 230000008685 targeting Effects 0.000 description 3
- 238000002560 therapeutic procedure Methods 0.000 description 3
- 108010038745 tryptophylglycine Proteins 0.000 description 3
- 241000701161 unidentified adenovirus Species 0.000 description 3
- 108010009962 valyltyrosine Proteins 0.000 description 3
- PKYCWFICOKSIHZ-UHFFFAOYSA-N 1-(3,7-dihydroxyphenoxazin-10-yl)ethanone Chemical compound OC1=CC=C2N(C(=O)C)C3=CC=C(O)C=C3OC2=C1 PKYCWFICOKSIHZ-UHFFFAOYSA-N 0.000 description 2
- JBGSZRYCXBPWGX-BQBZGAKWSA-N Ala-Arg-Gly Chemical compound OC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](N)C)CCCN=C(N)N JBGSZRYCXBPWGX-BQBZGAKWSA-N 0.000 description 2
- BLIMFWGRQKRCGT-YUMQZZPRSA-N Ala-Gly-Lys Chemical compound C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCCN BLIMFWGRQKRCGT-YUMQZZPRSA-N 0.000 description 2
- NIZKGBJVCMRDKO-KWQFWETISA-N Ala-Gly-Tyr Chemical compound C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 NIZKGBJVCMRDKO-KWQFWETISA-N 0.000 description 2
- HQJKCXHQNUCKMY-GHCJXIJMSA-N Ala-Ile-Asp Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)O)NC(=O)[C@H](C)N HQJKCXHQNUCKMY-GHCJXIJMSA-N 0.000 description 2
- RZZMZYZXNJRPOJ-BJDJZHNGSA-N Ala-Ile-Lys Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](C)N RZZMZYZXNJRPOJ-BJDJZHNGSA-N 0.000 description 2
- VNYMOTCMNHJGTG-JBDRJPRFSA-N Ala-Ile-Ser Chemical compound [H]N[C@@H](C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O VNYMOTCMNHJGTG-JBDRJPRFSA-N 0.000 description 2
- LNNSWWRRYJLGNI-NAKRPEOUSA-N Ala-Ile-Val Chemical compound C[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C(C)C)C(O)=O LNNSWWRRYJLGNI-NAKRPEOUSA-N 0.000 description 2
- AWNAEZICPNGAJK-FXQIFTODSA-N Ala-Met-Ser Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CO)C(O)=O AWNAEZICPNGAJK-FXQIFTODSA-N 0.000 description 2
- CQJHFKKGZXKZBC-BPNCWPANSA-N Ala-Pro-Tyr Chemical compound C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 CQJHFKKGZXKZBC-BPNCWPANSA-N 0.000 description 2
- OEVCHROQUIVQFZ-YTLHQDLWSA-N Ala-Thr-Ala Chemical compound C[C@H](N)C(=O)N[C@@H]([C@H](O)C)C(=O)N[C@@H](C)C(O)=O OEVCHROQUIVQFZ-YTLHQDLWSA-N 0.000 description 2
- AENHOIXXHKNIQL-AUTRQRHGSA-N Ala-Tyr-Ala Chemical compound [O-]C(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@@H]([NH3+])C)CC1=CC=C(O)C=C1 AENHOIXXHKNIQL-AUTRQRHGSA-N 0.000 description 2
- GIVATXIGCXFQQA-FXQIFTODSA-N Arg-Ala-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCCN=C(N)N GIVATXIGCXFQQA-FXQIFTODSA-N 0.000 description 2
- OTUQSEPIIVBYEM-IHRRRGAJSA-N Arg-Asn-Tyr Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O OTUQSEPIIVBYEM-IHRRRGAJSA-N 0.000 description 2
- GNYUVVJYGJFKHN-RVMXOQNASA-N Arg-Ile-Pro Chemical compound CC[C@H](C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCCN=C(N)N)N GNYUVVJYGJFKHN-RVMXOQNASA-N 0.000 description 2
- UVTGNSWSRSCPLP-UHFFFAOYSA-N Arg-Tyr Natural products NC(CCNC(=N)N)C(=O)NC(Cc1ccc(O)cc1)C(=O)O UVTGNSWSRSCPLP-UHFFFAOYSA-N 0.000 description 2
- IZSMEUDYADKZTJ-KJEVXHAQSA-N Arg-Tyr-Thr Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H]([C@@H](C)O)C(O)=O IZSMEUDYADKZTJ-KJEVXHAQSA-N 0.000 description 2
- CPTXATAOUQJQRO-GUBZILKMSA-N Arg-Val-Ser Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O CPTXATAOUQJQRO-GUBZILKMSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- BDMIFVIWCNLDCT-CIUDSAMLSA-N Asn-Arg-Glu Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(O)=O BDMIFVIWCNLDCT-CIUDSAMLSA-N 0.000 description 2
- POOCJCRBHHMAOS-FXQIFTODSA-N Asn-Arg-Ser Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(O)=O POOCJCRBHHMAOS-FXQIFTODSA-N 0.000 description 2
- DAPLJWATMAXPPZ-CIUDSAMLSA-N Asn-Asn-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CC(N)=O DAPLJWATMAXPPZ-CIUDSAMLSA-N 0.000 description 2
- OOWSBIOUKIUWLO-RCOVLWMOSA-N Asn-Gly-Val Chemical compound [H]N[C@@H](CC(N)=O)C(=O)NCC(=O)N[C@@H](C(C)C)C(O)=O OOWSBIOUKIUWLO-RCOVLWMOSA-N 0.000 description 2
- QEQVUHQQYDZUEN-GUBZILKMSA-N Asn-His-Glu Chemical compound C1=C(NC=N1)C[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)O)NC(=O)[C@H](CC(=O)N)N QEQVUHQQYDZUEN-GUBZILKMSA-N 0.000 description 2
- VWADICJNCPFKJS-ZLUOBGJFSA-N Asn-Ser-Asp Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(O)=O VWADICJNCPFKJS-ZLUOBGJFSA-N 0.000 description 2
- YHXNKGKUDJCAHB-PBCZWWQYSA-N Asn-Thr-His Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)NC(=O)[C@H](CC(=O)N)N)O YHXNKGKUDJCAHB-PBCZWWQYSA-N 0.000 description 2
- DPSUVAPLRQDWAO-YDHLFZDLSA-N Asn-Tyr-Val Chemical compound CC(C)[C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)[C@H](CC(=O)N)N DPSUVAPLRQDWAO-YDHLFZDLSA-N 0.000 description 2
- FAEIQWHBRBWUBN-FXQIFTODSA-N Asp-Arg-Ser Chemical compound C(C[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CC(=O)O)N)CN=C(N)N FAEIQWHBRBWUBN-FXQIFTODSA-N 0.000 description 2
- NYLBGYLHBDFRHL-VEVYYDQMSA-N Asp-Arg-Thr Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(O)=O NYLBGYLHBDFRHL-VEVYYDQMSA-N 0.000 description 2
- YNCHFVRXEQFPBY-BQBZGAKWSA-N Asp-Gly-Arg Chemical compound OC(=O)C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N YNCHFVRXEQFPBY-BQBZGAKWSA-N 0.000 description 2
- KHGPWGKPYHPOIK-QWRGUYRKSA-N Asp-Gly-Phe Chemical compound [H]N[C@@H](CC(O)=O)C(=O)NCC(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O KHGPWGKPYHPOIK-QWRGUYRKSA-N 0.000 description 2
- SVABRQFIHCSNCI-FOHZUACHSA-N Asp-Gly-Thr Chemical compound [H]N[C@@H](CC(O)=O)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(O)=O SVABRQFIHCSNCI-FOHZUACHSA-N 0.000 description 2
- TZOZNVLBTAFJRW-UGYAYLCHSA-N Asp-Ile-Asp Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)O)NC(=O)[C@H](CC(=O)O)N TZOZNVLBTAFJRW-UGYAYLCHSA-N 0.000 description 2
- IOXWDLNHXZOXQP-FXQIFTODSA-N Asp-Met-Ser Chemical compound CSCC[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CC(=O)O)N IOXWDLNHXZOXQP-FXQIFTODSA-N 0.000 description 2
- AHWRSSLYSGLBGD-CIUDSAMLSA-N Asp-Pro-Glu Chemical compound OC(=O)C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O AHWRSSLYSGLBGD-CIUDSAMLSA-N 0.000 description 2
- YFGUZQQCSDZRBN-DCAQKATOSA-N Asp-Pro-Leu Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(O)=O YFGUZQQCSDZRBN-DCAQKATOSA-N 0.000 description 2
- OZBXOELNJBSJOA-UBHSHLNASA-N Asp-Ser-Trp Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(=O)O)N OZBXOELNJBSJOA-UBHSHLNASA-N 0.000 description 2
- JJQGZGOEDSSHTE-FOHZUACHSA-N Asp-Thr-Gly Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(O)=O JJQGZGOEDSSHTE-FOHZUACHSA-N 0.000 description 2
- LTARLVHGOGBRHN-AAEUAGOBSA-N Asp-Trp-Gly Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)NCC(O)=O LTARLVHGOGBRHN-AAEUAGOBSA-N 0.000 description 2
- PLNJUJGNLDSFOP-UWJYBYFXSA-N Asp-Tyr-Ala Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](C)C(O)=O PLNJUJGNLDSFOP-UWJYBYFXSA-N 0.000 description 2
- HTSSXFASOUSJQG-IHPCNDPISA-N Asp-Tyr-Trp Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(O)=O HTSSXFASOUSJQG-IHPCNDPISA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 2
- 241000699800 Cricetinae Species 0.000 description 2
- 241000699802 Cricetulus griseus Species 0.000 description 2
- NOCCABSVTRONIN-CIUDSAMLSA-N Cys-Ala-Leu Chemical compound C[C@@H](C(=O)N[C@@H](CC(C)C)C(=O)O)NC(=O)[C@H](CS)N NOCCABSVTRONIN-CIUDSAMLSA-N 0.000 description 2
- HHABWQIFXZPZCK-ACZMJKKPSA-N Cys-Gln-Ser Chemical compound C(CC(=O)N)[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CS)N HHABWQIFXZPZCK-ACZMJKKPSA-N 0.000 description 2
- MXZYQNJCBVJHSR-KATARQTJSA-N Cys-Lys-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CS)N)O MXZYQNJCBVJHSR-KATARQTJSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical group [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- NPTGGVQJYRSMCM-GLLZPBPUSA-N Gln-Gln-Thr Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O NPTGGVQJYRSMCM-GLLZPBPUSA-N 0.000 description 2
- RWQCWSGOOOEGPB-FXQIFTODSA-N Gln-Ser-Glu Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(O)=O RWQCWSGOOOEGPB-FXQIFTODSA-N 0.000 description 2
- OSCLNNWLKKIQJM-WDSKDSINSA-N Gln-Ser-Gly Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(=O)NCC(O)=O OSCLNNWLKKIQJM-WDSKDSINSA-N 0.000 description 2
- ARYKRXHBIPLULY-XKBZYTNZSA-N Gln-Thr-Ser Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(O)=O ARYKRXHBIPLULY-XKBZYTNZSA-N 0.000 description 2
- HLRLXVPRJJITSK-IFFSRLJSSA-N Gln-Thr-Val Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O HLRLXVPRJJITSK-IFFSRLJSSA-N 0.000 description 2
- VPKBCVUDBNINAH-GARJFASQSA-N Glu-Arg-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CCC(=O)O)N)C(=O)O VPKBCVUDBNINAH-GARJFASQSA-N 0.000 description 2
- RDPOETHPAQEGDP-ACZMJKKPSA-N Glu-Asp-Ala Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C)C(O)=O RDPOETHPAQEGDP-ACZMJKKPSA-N 0.000 description 2
- IVGJYOOGJLFKQE-AVGNSLFASA-N Glu-Leu-Lys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CCC(=O)O)N IVGJYOOGJLFKQE-AVGNSLFASA-N 0.000 description 2
- GUOWMVFLAJNPDY-CIUDSAMLSA-N Glu-Ser-Met Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(O)=O GUOWMVFLAJNPDY-CIUDSAMLSA-N 0.000 description 2
- UZWUBBRJWFTHTD-LAEOZQHASA-N Glu-Val-Asn Chemical compound NC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)CCC(O)=O UZWUBBRJWFTHTD-LAEOZQHASA-N 0.000 description 2
- JVACNFOPSUPDTK-QWRGUYRKSA-N Gly-Asn-Phe Chemical compound NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 JVACNFOPSUPDTK-QWRGUYRKSA-N 0.000 description 2
- IWAXHBCACVWNHT-BQBZGAKWSA-N Gly-Asp-Arg Chemical compound NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CCCN=C(N)N IWAXHBCACVWNHT-BQBZGAKWSA-N 0.000 description 2
- XQHSBNVACKQWAV-WHFBIAKZSA-N Gly-Asp-Asn Chemical compound [H]NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O XQHSBNVACKQWAV-WHFBIAKZSA-N 0.000 description 2
- 108010050006 Gly-Asp-Gly-Arg Proteins 0.000 description 2
- CQZDZKRHFWJXDF-WDSKDSINSA-N Gly-Gln-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CN CQZDZKRHFWJXDF-WDSKDSINSA-N 0.000 description 2
- QITBQGJOXQYMOA-ZETCQYMHSA-N Gly-Gly-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)CNC(=O)CN QITBQGJOXQYMOA-ZETCQYMHSA-N 0.000 description 2
- LPCKHUXOGVNZRS-YUMQZZPRSA-N Gly-His-Ser Chemical compound [H]NCC(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CO)C(O)=O LPCKHUXOGVNZRS-YUMQZZPRSA-N 0.000 description 2
- DGKBSGNCMCLDSL-BYULHYEWSA-N Gly-Ile-Asn Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)CN DGKBSGNCMCLDSL-BYULHYEWSA-N 0.000 description 2
- XVYKMNXXJXQKME-XEGUGMAKSA-N Gly-Ile-Tyr Chemical compound NCC(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 XVYKMNXXJXQKME-XEGUGMAKSA-N 0.000 description 2
- IUZGUFAJDBHQQV-YUMQZZPRSA-N Gly-Leu-Asn Chemical compound NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(O)=O IUZGUFAJDBHQQV-YUMQZZPRSA-N 0.000 description 2
- FXGRXIATVXUAHO-WEDXCCLWSA-N Gly-Lys-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@@H](NC(=O)CN)CCCCN FXGRXIATVXUAHO-WEDXCCLWSA-N 0.000 description 2
- WCORRBXVISTKQL-WHFBIAKZSA-N Gly-Ser-Ser Chemical compound NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O WCORRBXVISTKQL-WHFBIAKZSA-N 0.000 description 2
- FFJQHWKSGAWSTJ-BFHQHQDPSA-N Gly-Thr-Ala Chemical compound [H]NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(O)=O FFJQHWKSGAWSTJ-BFHQHQDPSA-N 0.000 description 2
- ZZWUYQXMIFTIIY-WEDXCCLWSA-N Gly-Thr-Leu Chemical compound [H]NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O ZZWUYQXMIFTIIY-WEDXCCLWSA-N 0.000 description 2
- YGHSQRJSHKYUJY-SCZZXKLOSA-N Gly-Val-Pro Chemical compound CC(C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)CN YGHSQRJSHKYUJY-SCZZXKLOSA-N 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 241000701044 Human gammaherpesvirus 4 Species 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- 108700039609 IRW peptide Proteins 0.000 description 2
- NZOCIWKZUVUNDW-ZKWXMUAHSA-N Ile-Gly-Ala Chemical compound CC[C@H](C)[C@H](N)C(=O)NCC(=O)N[C@@H](C)C(O)=O NZOCIWKZUVUNDW-ZKWXMUAHSA-N 0.000 description 2
- KFVUBLZRFSVDGO-BYULHYEWSA-N Ile-Gly-Asp Chemical compound CC[C@H](C)[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CC(O)=O KFVUBLZRFSVDGO-BYULHYEWSA-N 0.000 description 2
- DZMWFIRHFFVBHS-ZEWNOJEFSA-N Ile-Tyr-Phe Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CC2=CC=CC=C2)C(=O)O)N DZMWFIRHFFVBHS-ZEWNOJEFSA-N 0.000 description 2
- RQZFWBLDTBDEOF-RNJOBUHISA-N Ile-Val-Pro Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](C(C)C)C(=O)N1CCC[C@@H]1C(=O)O)N RQZFWBLDTBDEOF-RNJOBUHISA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical class C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 102000009786 Immunoglobulin Constant Regions Human genes 0.000 description 2
- 108010009817 Immunoglobulin Constant Regions Proteins 0.000 description 2
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 2
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 2
- 150000008575 L-amino acids Chemical class 0.000 description 2
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 2
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 2
- CLVUXCBGKUECIT-HJGDQZAQSA-N Leu-Asp-Thr Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O CLVUXCBGKUECIT-HJGDQZAQSA-N 0.000 description 2
- VWHGTYCRDRBSFI-ZETCQYMHSA-N Leu-Gly-Gly Chemical compound CC(C)C[C@H](N)C(=O)NCC(=O)NCC(O)=O VWHGTYCRDRBSFI-ZETCQYMHSA-N 0.000 description 2
- HYIFFZAQXPUEAU-QWRGUYRKSA-N Leu-Gly-Leu Chemical compound CC(C)C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CC(C)C HYIFFZAQXPUEAU-QWRGUYRKSA-N 0.000 description 2
- HGFGEMSVBMCFKK-MNXVOIDGSA-N Leu-Ile-Glu Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(O)=O HGFGEMSVBMCFKK-MNXVOIDGSA-N 0.000 description 2
- DSFYPIUSAMSERP-IHRRRGAJSA-N Leu-Leu-Arg Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C(O)=O)CCCN=C(N)N DSFYPIUSAMSERP-IHRRRGAJSA-N 0.000 description 2
- FKQPWMZLIIATBA-AJNGGQMLSA-N Leu-Lys-Ile Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O FKQPWMZLIIATBA-AJNGGQMLSA-N 0.000 description 2
- ILDSIMPXNFWKLH-KATARQTJSA-N Leu-Thr-Ser Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(O)=O ILDSIMPXNFWKLH-KATARQTJSA-N 0.000 description 2
- NRQRKMYZONPCTM-CIUDSAMLSA-N Lys-Asp-Ser Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O NRQRKMYZONPCTM-CIUDSAMLSA-N 0.000 description 2
- YRNRVKTYDSLKMD-KKUMJFAQSA-N Lys-Ser-Tyr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O YRNRVKTYDSLKMD-KKUMJFAQSA-N 0.000 description 2
- RMOKGALPSPOYKE-KATARQTJSA-N Lys-Thr-Ser Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(O)=O RMOKGALPSPOYKE-KATARQTJSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- GHQFLTYXGUETFD-UFYCRDLUSA-N Met-Tyr-Tyr Chemical compound CSCC[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)O)N GHQFLTYXGUETFD-UFYCRDLUSA-N 0.000 description 2
- 102100031545 Microsomal triglyceride transfer protein large subunit Human genes 0.000 description 2
- 101710135898 Myc proto-oncogene protein Proteins 0.000 description 2
- 102100038895 Myc proto-oncogene protein Human genes 0.000 description 2
- SITLTJHOQZFJGG-UHFFFAOYSA-N N-L-alpha-glutamyl-L-valine Natural products CC(C)C(C(O)=O)NC(=O)C(N)CCC(O)=O SITLTJHOQZFJGG-UHFFFAOYSA-N 0.000 description 2
- PESQCPHRXOFIPX-UHFFFAOYSA-N N-L-methionyl-L-tyrosine Natural products CSCCC(N)C(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 PESQCPHRXOFIPX-UHFFFAOYSA-N 0.000 description 2
- XMBSYZWANAQXEV-UHFFFAOYSA-N N-alpha-L-glutamyl-L-phenylalanine Natural products OC(=O)CCC(N)C(=O)NC(C(O)=O)CC1=CC=CC=C1 XMBSYZWANAQXEV-UHFFFAOYSA-N 0.000 description 2
- KZNQNBZMBZJQJO-UHFFFAOYSA-N N-glycyl-L-proline Natural products NCC(=O)N1CCCC1C(O)=O KZNQNBZMBZJQJO-UHFFFAOYSA-N 0.000 description 2
- AJHCSUXXECOXOY-UHFFFAOYSA-N N-glycyl-L-tryptophan Natural products C1=CC=C2C(CC(NC(=O)CN)C(O)=O)=CNC2=C1 AJHCSUXXECOXOY-UHFFFAOYSA-N 0.000 description 2
- 108010079364 N-glycylalanine Proteins 0.000 description 2
- 238000005481 NMR spectroscopy Methods 0.000 description 2
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 2
- 108020005187 Oligonucleotide Probes Proteins 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- 206010033733 Papule Diseases 0.000 description 2
- 241001494479 Pecora Species 0.000 description 2
- ZENDEDYRYVHBEG-SRVKXCTJSA-N Phe-Asp-Asp Chemical compound OC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CC1=CC=CC=C1 ZENDEDYRYVHBEG-SRVKXCTJSA-N 0.000 description 2
- CSYVXYQDIVCQNU-QWRGUYRKSA-N Phe-Asp-Gly Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(O)=O CSYVXYQDIVCQNU-QWRGUYRKSA-N 0.000 description 2
- WPTYDQPGBMDUBI-QWRGUYRKSA-N Phe-Gly-Asn Chemical compound N[C@@H](Cc1ccccc1)C(=O)NCC(=O)N[C@@H](CC(N)=O)C(O)=O WPTYDQPGBMDUBI-QWRGUYRKSA-N 0.000 description 2
- WEDZFLRYSIDIRX-IHRRRGAJSA-N Phe-Ser-Arg Chemical compound NC(=N)NCCC[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC1=CC=CC=C1 WEDZFLRYSIDIRX-IHRRRGAJSA-N 0.000 description 2
- BONHGTUEEPIMPM-AVGNSLFASA-N Phe-Ser-Glu Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(O)=O BONHGTUEEPIMPM-AVGNSLFASA-N 0.000 description 2
- MSSXKZBDKZAHCX-UNQGMJICSA-N Phe-Thr-Val Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O MSSXKZBDKZAHCX-UNQGMJICSA-N 0.000 description 2
- 101710182846 Polyhedrin Proteins 0.000 description 2
- 241000288906 Primates Species 0.000 description 2
- OOLOTUZJUBOMAX-GUBZILKMSA-N Pro-Ala-Val Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(O)=O OOLOTUZJUBOMAX-GUBZILKMSA-N 0.000 description 2
- SGCZFWSQERRKBD-BQBZGAKWSA-N Pro-Asp-Gly Chemical compound OC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1 SGCZFWSQERRKBD-BQBZGAKWSA-N 0.000 description 2
- PULPZRAHVFBVTO-DCAQKATOSA-N Pro-Glu-Arg Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O PULPZRAHVFBVTO-DCAQKATOSA-N 0.000 description 2
- HAAQQNHQZBOWFO-LURJTMIESA-N Pro-Gly-Gly Chemical compound OC(=O)CNC(=O)CNC(=O)[C@@H]1CCCN1 HAAQQNHQZBOWFO-LURJTMIESA-N 0.000 description 2
- RMODQFBNDDENCP-IHRRRGAJSA-N Pro-Lys-Leu Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(O)=O RMODQFBNDDENCP-IHRRRGAJSA-N 0.000 description 2
- WFIVLLFYUZZWOD-RHYQMDGZSA-N Pro-Lys-Thr Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(O)=O WFIVLLFYUZZWOD-RHYQMDGZSA-N 0.000 description 2
- FDMKYQQYJKYCLV-GUBZILKMSA-N Pro-Pro-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H]1NCCC1 FDMKYQQYJKYCLV-GUBZILKMSA-N 0.000 description 2
- AJNGQVUFQUVRQT-JYJNAYRXSA-N Pro-Pro-Tyr Chemical compound C([C@@H](C(=O)O)NC(=O)[C@H]1N(CCC1)C(=O)[C@H]1NCCC1)C1=CC=C(O)C=C1 AJNGQVUFQUVRQT-JYJNAYRXSA-N 0.000 description 2
- QKWYXRPICJEQAJ-KJEVXHAQSA-N Pro-Tyr-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)[C@@H]2CCCN2)O QKWYXRPICJEQAJ-KJEVXHAQSA-N 0.000 description 2
- QMABBZHZMDXHKU-FKBYEOEOSA-N Pro-Tyr-Trp Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(O)=O QMABBZHZMDXHKU-FKBYEOEOSA-N 0.000 description 2
- 239000004365 Protease Substances 0.000 description 2
- 239000008156 Ringer's lactate solution Substances 0.000 description 2
- 241000283984 Rodentia Species 0.000 description 2
- PZZJMBYSYAKYPK-UWJYBYFXSA-N Ser-Ala-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O PZZJMBYSYAKYPK-UWJYBYFXSA-N 0.000 description 2
- QGMLKFGTGXWAHF-IHRRRGAJSA-N Ser-Arg-Phe Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O QGMLKFGTGXWAHF-IHRRRGAJSA-N 0.000 description 2
- NRCJWSGXMAPYQX-LPEHRKFASA-N Ser-Arg-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CO)N)C(=O)O NRCJWSGXMAPYQX-LPEHRKFASA-N 0.000 description 2
- VAIZFHMTBFYJIA-ACZMJKKPSA-N Ser-Asp-Gln Chemical compound OC[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CCC(N)=O VAIZFHMTBFYJIA-ACZMJKKPSA-N 0.000 description 2
- HMRAQFJFTOLDKW-GUBZILKMSA-N Ser-His-Glu Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CCC(O)=O)C(O)=O HMRAQFJFTOLDKW-GUBZILKMSA-N 0.000 description 2
- NLOAIFSWUUFQFR-CIUDSAMLSA-N Ser-Leu-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O NLOAIFSWUUFQFR-CIUDSAMLSA-N 0.000 description 2
- PJIQEIFXZPCWOJ-FXQIFTODSA-N Ser-Pro-Asp Chemical compound [H]N[C@@H](CO)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(O)=O)C(O)=O PJIQEIFXZPCWOJ-FXQIFTODSA-N 0.000 description 2
- AZWNCEBQZXELEZ-FXQIFTODSA-N Ser-Pro-Ser Chemical compound OC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O AZWNCEBQZXELEZ-FXQIFTODSA-N 0.000 description 2
- HHJFMHQYEAAOBM-ZLUOBGJFSA-N Ser-Ser-Ala Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(O)=O HHJFMHQYEAAOBM-ZLUOBGJFSA-N 0.000 description 2
- SRSPTFBENMJHMR-WHFBIAKZSA-N Ser-Ser-Gly Chemical compound OC[C@H](N)C(=O)N[C@@H](CO)C(=O)NCC(O)=O SRSPTFBENMJHMR-WHFBIAKZSA-N 0.000 description 2
- XQJCEKXQUJQNNK-ZLUOBGJFSA-N Ser-Ser-Ser Chemical compound OC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O XQJCEKXQUJQNNK-ZLUOBGJFSA-N 0.000 description 2
- DKGRNFUXVTYRAS-UBHSHLNASA-N Ser-Ser-Trp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(O)=O DKGRNFUXVTYRAS-UBHSHLNASA-N 0.000 description 2
- DYEGLQRVMBWQLD-IXOXFDKPSA-N Ser-Thr-Phe Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)O)NC(=O)[C@H](CO)N)O DYEGLQRVMBWQLD-IXOXFDKPSA-N 0.000 description 2
- ZKOKTQPHFMRSJP-YJRXYDGGSA-N Ser-Thr-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O ZKOKTQPHFMRSJP-YJRXYDGGSA-N 0.000 description 2
- PIQRHJQWEPWFJG-UWJYBYFXSA-N Ser-Tyr-Ala Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](C)C(O)=O PIQRHJQWEPWFJG-UWJYBYFXSA-N 0.000 description 2
- FHXGMDRKJHKLKW-QWRGUYRKSA-N Ser-Tyr-Gly Chemical compound OC[C@H](N)C(=O)N[C@H](C(=O)NCC(O)=O)CC1=CC=C(O)C=C1 FHXGMDRKJHKLKW-QWRGUYRKSA-N 0.000 description 2
- 108010003723 Single-Domain Antibodies Proteins 0.000 description 2
- 108091081024 Start codon Proteins 0.000 description 2
- 208000006011 Stroke Diseases 0.000 description 2
- RKSMVPNZHBRNNS-UHFFFAOYSA-N Succinobucol Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(SC(C)(C)SC=2C=C(C(OC(=O)CCC(O)=O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 RKSMVPNZHBRNNS-UHFFFAOYSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- MQCPGOZXFSYJPS-KZVJFYERSA-N Thr-Ala-Arg Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O MQCPGOZXFSYJPS-KZVJFYERSA-N 0.000 description 2
- UNURFMVMXLENAZ-KJEVXHAQSA-N Thr-Arg-Tyr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O UNURFMVMXLENAZ-KJEVXHAQSA-N 0.000 description 2
- OHAJHDJOCKKJLV-LKXGYXEUSA-N Thr-Asp-Ser Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O OHAJHDJOCKKJLV-LKXGYXEUSA-N 0.000 description 2
- DCLBXIWHLVEPMQ-JRQIVUDYSA-N Thr-Asp-Tyr Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 DCLBXIWHLVEPMQ-JRQIVUDYSA-N 0.000 description 2
- LIXBDERDAGNVAV-XKBZYTNZSA-N Thr-Gln-Ser Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(O)=O LIXBDERDAGNVAV-XKBZYTNZSA-N 0.000 description 2
- AQAMPXBRJJWPNI-JHEQGTHGSA-N Thr-Gly-Glu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CCC(O)=O)C(O)=O AQAMPXBRJJWPNI-JHEQGTHGSA-N 0.000 description 2
- KRGDDWVBBDLPSJ-CUJWVEQBSA-N Thr-His-Ser Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CO)C(O)=O KRGDDWVBBDLPSJ-CUJWVEQBSA-N 0.000 description 2
- RFKVQLIXNVEOMB-WEDXCCLWSA-N Thr-Leu-Gly Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)O)N)O RFKVQLIXNVEOMB-WEDXCCLWSA-N 0.000 description 2
- IJVNLNRVDUTWDD-MEYUZBJRSA-N Thr-Leu-Tyr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O IJVNLNRVDUTWDD-MEYUZBJRSA-N 0.000 description 2
- KZSYAEWQMJEGRZ-RHYQMDGZSA-N Thr-Leu-Val Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(O)=O KZSYAEWQMJEGRZ-RHYQMDGZSA-N 0.000 description 2
- KKPOGALELPLJTL-MEYUZBJRSA-N Thr-Lys-Tyr Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 KKPOGALELPLJTL-MEYUZBJRSA-N 0.000 description 2
- DXPURPNJDFCKKO-RHYQMDGZSA-N Thr-Lys-Val Chemical compound CC(C)[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)[C@@H](C)O)C(O)=O DXPURPNJDFCKKO-RHYQMDGZSA-N 0.000 description 2
- BIBYEFRASCNLAA-CDMKHQONSA-N Thr-Phe-Gly Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@H](C(=O)NCC(O)=O)CC1=CC=CC=C1 BIBYEFRASCNLAA-CDMKHQONSA-N 0.000 description 2
- NQQMWWVVGIXUOX-SVSWQMSJSA-N Thr-Ser-Ile Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O NQQMWWVVGIXUOX-SVSWQMSJSA-N 0.000 description 2
- IEZVHOULSUULHD-XGEHTFHBSA-N Thr-Ser-Val Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O IEZVHOULSUULHD-XGEHTFHBSA-N 0.000 description 2
- LXXCHJKHJYRMIY-FQPOAREZSA-N Thr-Tyr-Ala Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](C)C(O)=O LXXCHJKHJYRMIY-FQPOAREZSA-N 0.000 description 2
- LVRFMARKDGGZMX-IZPVPAKOSA-N Thr-Tyr-Thr Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)O)C(O)=O)CC1=CC=C(O)C=C1 LVRFMARKDGGZMX-IZPVPAKOSA-N 0.000 description 2
- IQFYYKKMVGJFEH-XLPZGREQSA-N Thymidine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 IQFYYKKMVGJFEH-XLPZGREQSA-N 0.000 description 2
- 241000723873 Tobacco mosaic virus Species 0.000 description 2
- 101710150448 Transcriptional regulator Myc Proteins 0.000 description 2
- GTNCSPKYWCJZAC-XIRDDKMYSA-N Trp-Asp-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CC1=CNC2=CC=CC=C21)N GTNCSPKYWCJZAC-XIRDDKMYSA-N 0.000 description 2
- SVGAWGVHFIYAEE-JSGCOSHPSA-N Trp-Gly-Gln Chemical compound C1=CC=C2C(C[C@H](N)C(=O)NCC(=O)N[C@@H](CCC(N)=O)C(O)=O)=CNC2=C1 SVGAWGVHFIYAEE-JSGCOSHPSA-N 0.000 description 2
- HWCBFXAWVTXXHZ-NYVOZVTQSA-N Trp-Ser-Trp Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC3=CNC4=CC=CC=C43)C(=O)O)N HWCBFXAWVTXXHZ-NYVOZVTQSA-N 0.000 description 2
- SUGLEXVWEJOCGN-ONUFPDRFSA-N Trp-Thr-Trp Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)O)NC(=O)[C@H](CC3=CNC4=CC=CC=C43)N)O SUGLEXVWEJOCGN-ONUFPDRFSA-N 0.000 description 2
- RWTFCAMQLFNPTK-UMPQAUOISA-N Trp-Val-Thr Chemical compound C1=CC=C2C(C[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)=CNC2=C1 RWTFCAMQLFNPTK-UMPQAUOISA-N 0.000 description 2
- LGEYOIQBBIPHQN-UWJYBYFXSA-N Tyr-Ala-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 LGEYOIQBBIPHQN-UWJYBYFXSA-N 0.000 description 2
- RCLOWEZASFJFEX-KKUMJFAQSA-N Tyr-Asp-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 RCLOWEZASFJFEX-KKUMJFAQSA-N 0.000 description 2
- AZGZDDNKFFUDEH-QWRGUYRKSA-N Tyr-Gly-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H](N)CC1=CC=C(O)C=C1 AZGZDDNKFFUDEH-QWRGUYRKSA-N 0.000 description 2
- FJBCEFPCVPHPPM-STECZYCISA-N Tyr-Ile-Val Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C(C)C)C(O)=O FJBCEFPCVPHPPM-STECZYCISA-N 0.000 description 2
- OGPKMBOPMDTEDM-IHRRRGAJSA-N Tyr-Met-Ser Chemical compound CSCC[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)N OGPKMBOPMDTEDM-IHRRRGAJSA-N 0.000 description 2
- ZPFLBLFITJCBTP-QWRGUYRKSA-N Tyr-Ser-Gly Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CO)C(=O)NCC(O)=O ZPFLBLFITJCBTP-QWRGUYRKSA-N 0.000 description 2
- ABZWHLRQBSBPTO-RNXOBYDBSA-N Tyr-Trp-Phe Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)O)NC(=O)[C@H](CC2=CNC3=CC=CC=C32)NC(=O)[C@H](CC4=CC=C(C=C4)O)N ABZWHLRQBSBPTO-RNXOBYDBSA-N 0.000 description 2
- ANHVRCNNGJMJNG-BZSNNMDCSA-N Tyr-Tyr-Cys Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)N[C@@H](CS)C(=O)O)N)O ANHVRCNNGJMJNG-BZSNNMDCSA-N 0.000 description 2
- UUJHRSTVQCFDPA-UFYCRDLUSA-N Tyr-Tyr-Val Chemical compound C([C@@H](C(=O)N[C@@H](C(C)C)C(O)=O)NC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=C(O)C=C1 UUJHRSTVQCFDPA-UFYCRDLUSA-N 0.000 description 2
- LABUITCFCAABSV-BPNCWPANSA-N Val-Ala-Tyr Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 LABUITCFCAABSV-BPNCWPANSA-N 0.000 description 2
- LABUITCFCAABSV-UHFFFAOYSA-N Val-Ala-Tyr Natural products CC(C)C(N)C(=O)NC(C)C(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 LABUITCFCAABSV-UHFFFAOYSA-N 0.000 description 2
- AGKDVLSDNSTLFA-UMNHJUIQSA-N Val-Gln-Pro Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N1CCC[C@@H]1C(=O)O)N AGKDVLSDNSTLFA-UMNHJUIQSA-N 0.000 description 2
- ROLGIBMFNMZANA-GVXVVHGQSA-N Val-Glu-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](C(C)C)N ROLGIBMFNMZANA-GVXVVHGQSA-N 0.000 description 2
- WNZSAUMKZQXHNC-UKJIMTQDSA-N Val-Ile-Gln Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](C(C)C)N WNZSAUMKZQXHNC-UKJIMTQDSA-N 0.000 description 2
- DJQIUOKSNRBTSV-CYDGBPFRSA-N Val-Ile-Val Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](C(C)C)C(=O)O)NC(=O)[C@H](C(C)C)N DJQIUOKSNRBTSV-CYDGBPFRSA-N 0.000 description 2
- UGFMVXRXULGLNO-XPUUQOCRSA-N Val-Ser-Gly Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](CO)C(=O)NCC(O)=O UGFMVXRXULGLNO-XPUUQOCRSA-N 0.000 description 2
- VHIZXDZMTDVFGX-DCAQKATOSA-N Val-Ser-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H](C(C)C)N VHIZXDZMTDVFGX-DCAQKATOSA-N 0.000 description 2
- JAIZPWVHPQRYOU-ZJDVBMNYSA-N Val-Thr-Thr Chemical compound C[C@H]([C@@H](C(=O)N[C@@H]([C@@H](C)O)C(=O)O)NC(=O)[C@H](C(C)C)N)O JAIZPWVHPQRYOU-ZJDVBMNYSA-N 0.000 description 2
- 238000002441 X-ray diffraction Methods 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 108010024078 alanyl-glycyl-serine Proteins 0.000 description 2
- 108010045350 alanyl-tyrosyl-alanine Proteins 0.000 description 2
- 108010011559 alanylphenylalanine Proteins 0.000 description 2
- 108010070783 alanyltyrosine Proteins 0.000 description 2
- 230000000692 anti-sense effect Effects 0.000 description 2
- 239000004599 antimicrobial Substances 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 239000008135 aqueous vehicle Substances 0.000 description 2
- 108010010430 asparagine-proline-alanine Proteins 0.000 description 2
- 108010092854 aspartyllysine Proteins 0.000 description 2
- 239000012131 assay buffer Substances 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 229960000686 benzalkonium chloride Drugs 0.000 description 2
- UREZNYTWGJKWBI-UHFFFAOYSA-M benzethonium chloride Chemical compound [Cl-].C1=CC(C(C)(C)CC(C)(C)C)=CC=C1OCCOCC[N+](C)(C)CC1=CC=CC=C1 UREZNYTWGJKWBI-UHFFFAOYSA-M 0.000 description 2
- 229960001950 benzethonium chloride Drugs 0.000 description 2
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 2
- 238000001574 biopsy Methods 0.000 description 2
- 229940098773 bovine serum albumin Drugs 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 239000005018 casein Substances 0.000 description 2
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 2
- 235000021240 caseins Nutrition 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- YCIMNLLNPGFGHC-UHFFFAOYSA-N catechol Chemical compound OC1=CC=CC=C1O YCIMNLLNPGFGHC-UHFFFAOYSA-N 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- 238000003776 cleavage reaction Methods 0.000 description 2
- 238000012411 cloning technique Methods 0.000 description 2
- 238000012875 competitive assay Methods 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 230000000875 corresponding effect Effects 0.000 description 2
- 230000008878 coupling Effects 0.000 description 2
- 238000010168 coupling process Methods 0.000 description 2
- 238000005859 coupling reaction Methods 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000012217 deletion Methods 0.000 description 2
- 230000037430 deletion Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 235000005911 diet Nutrition 0.000 description 2
- 230000037213 diet Effects 0.000 description 2
- 239000000539 dimer Substances 0.000 description 2
- 108010054812 diprotin A Proteins 0.000 description 2
- 239000002270 dispersing agent Substances 0.000 description 2
- 231100000673 dose–response relationship Toxicity 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 2
- 230000008030 elimination Effects 0.000 description 2
- 238000003379 elimination reaction Methods 0.000 description 2
- 230000002255 enzymatic effect Effects 0.000 description 2
- 235000019441 ethanol Nutrition 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 235000019197 fats Nutrition 0.000 description 2
- 229940125753 fibrate Drugs 0.000 description 2
- 239000012467 final product Substances 0.000 description 2
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 2
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 2
- 239000007850 fluorescent dye Substances 0.000 description 2
- 238000004108 freeze drying Methods 0.000 description 2
- 230000004927 fusion Effects 0.000 description 2
- 108010085059 glutamyl-arginyl-proline Proteins 0.000 description 2
- 108010049041 glutamylalanine Proteins 0.000 description 2
- 230000013595 glycosylation Effects 0.000 description 2
- 238000006206 glycosylation reaction Methods 0.000 description 2
- 108010000434 glycyl-alanyl-leucine Proteins 0.000 description 2
- 108010010096 glycyl-glycyl-tyrosine Proteins 0.000 description 2
- 108010028188 glycyl-histidyl-serine Proteins 0.000 description 2
- 108010050475 glycyl-leucyl-tyrosine Proteins 0.000 description 2
- 108010015792 glycyllysine Proteins 0.000 description 2
- 108010084389 glycyltryptophan Proteins 0.000 description 2
- 108010037850 glycylvaline Proteins 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 210000005260 human cell Anatomy 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- FDGQSTZJBFJUBT-UHFFFAOYSA-N hypoxanthine Chemical compound O=C1NC=NC2=C1NC=N2 FDGQSTZJBFJUBT-UHFFFAOYSA-N 0.000 description 2
- 230000016784 immunoglobulin production Effects 0.000 description 2
- 229940072221 immunoglobulins Drugs 0.000 description 2
- 238000001114 immunoprecipitation Methods 0.000 description 2
- 238000011065 in-situ storage Methods 0.000 description 2
- 229910052738 indium Inorganic materials 0.000 description 2
- 238000003780 insertion Methods 0.000 description 2
- 230000037431 insertion Effects 0.000 description 2
- 238000007912 intraperitoneal administration Methods 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 239000007951 isotonicity adjuster Substances 0.000 description 2
- 108010053037 kyotorphin Proteins 0.000 description 2
- 238000002372 labelling Methods 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000004811 liquid chromatography Methods 0.000 description 2
- 239000003589 local anesthetic agent Substances 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- 210000004698 lymphocyte Anatomy 0.000 description 2
- 230000002132 lysosomal effect Effects 0.000 description 2
- RLSSMJSEOOYNOY-UHFFFAOYSA-N m-cresol Chemical compound CC1=CC=CC(O)=C1 RLSSMJSEOOYNOY-UHFFFAOYSA-N 0.000 description 2
- 238000007726 management method Methods 0.000 description 2
- 238000004949 mass spectrometry Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 108010038232 microsomal triglyceride transfer protein Proteins 0.000 description 2
- 230000003278 mimic effect Effects 0.000 description 2
- 238000002703 mutagenesis Methods 0.000 description 2
- 231100000350 mutagenesis Toxicity 0.000 description 2
- 229960003512 nicotinic acid Drugs 0.000 description 2
- 235000001968 nicotinic acid Nutrition 0.000 description 2
- 239000011664 nicotinic acid Substances 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- 239000002751 oligonucleotide probe Substances 0.000 description 2
- 238000002515 oligonucleotide synthesis Methods 0.000 description 2
- 210000001672 ovary Anatomy 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- AQIXEPGDORPWBJ-UHFFFAOYSA-N pentan-3-ol Chemical compound CCC(O)CC AQIXEPGDORPWBJ-UHFFFAOYSA-N 0.000 description 2
- 108010083476 phenylalanyltryptophan Proteins 0.000 description 2
- 235000021317 phosphate Nutrition 0.000 description 2
- 239000004417 polycarbonate Substances 0.000 description 2
- 229920000136 polysorbate Polymers 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 108010004914 prolylarginine Proteins 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 238000010188 recombinant method Methods 0.000 description 2
- 230000000306 recurrent effect Effects 0.000 description 2
- GHMLBKRAJCXXBS-UHFFFAOYSA-N resorcinol Chemical compound OC1=CC=CC(O)=C1 GHMLBKRAJCXXBS-UHFFFAOYSA-N 0.000 description 2
- 230000007017 scission Effects 0.000 description 2
- 238000002741 site-directed mutagenesis Methods 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 238000002798 spectrophotometry method Methods 0.000 description 2
- 238000010561 standard procedure Methods 0.000 description 2
- 238000011146 sterile filtration Methods 0.000 description 2
- 239000008223 sterile water Substances 0.000 description 2
- 238000010254 subcutaneous injection Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 108010033670 threonyl-aspartyl-tyrosine Proteins 0.000 description 2
- 238000011200 topical administration Methods 0.000 description 2
- 230000009466 transformation Effects 0.000 description 2
- 230000001052 transient effect Effects 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- 229910052727 yttrium Inorganic materials 0.000 description 2
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 1
- OZFAFGSSMRRTDW-UHFFFAOYSA-N (2,4-dichlorophenyl) benzenesulfonate Chemical compound ClC1=CC(Cl)=CC=C1OS(=O)(=O)C1=CC=CC=C1 OZFAFGSSMRRTDW-UHFFFAOYSA-N 0.000 description 1
- DIGQNXIGRZPYDK-WKSCXVIASA-N (2R)-6-amino-2-[[2-[[(2S)-2-[[2-[[(2R)-2-[[(2S)-2-[[(2R,3S)-2-[[2-[[(2S)-2-[[2-[[(2S)-2-[[(2S)-2-[[(2R)-2-[[(2S,3S)-2-[[(2R)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[2-[[(2S)-2-[[(2R)-2-[[2-[[2-[[2-[(2-amino-1-hydroxyethylidene)amino]-3-carboxy-1-hydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1,5-dihydroxy-5-iminopentylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]hexanoic acid Chemical compound C[C@@H]([C@@H](C(=N[C@@H](CS)C(=N[C@@H](C)C(=N[C@@H](CO)C(=NCC(=N[C@@H](CCC(=N)O)C(=NC(CS)C(=N[C@H]([C@H](C)O)C(=N[C@H](CS)C(=N[C@H](CO)C(=NCC(=N[C@H](CS)C(=NCC(=N[C@H](CCCCN)C(=O)O)O)O)O)O)O)O)O)O)O)O)O)O)O)N=C([C@H](CS)N=C([C@H](CO)N=C([C@H](CO)N=C([C@H](C)N=C(CN=C([C@H](CO)N=C([C@H](CS)N=C(CN=C(C(CS)N=C(C(CC(=O)O)N=C(CN)O)O)O)O)O)O)O)O)O)O)O)O DIGQNXIGRZPYDK-WKSCXVIASA-N 0.000 description 1
- ICLYJLBTOGPLMC-KVVVOXFISA-N (z)-octadec-9-enoate;tris(2-hydroxyethyl)azanium Chemical compound OCCN(CCO)CCO.CCCCCCCC\C=C/CCCCCCCC(O)=O ICLYJLBTOGPLMC-KVVVOXFISA-N 0.000 description 1
- TZCPCKNHXULUIY-RGULYWFUSA-N 1,2-distearoyl-sn-glycero-3-phosphoserine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@H](N)C(O)=O)OC(=O)CCCCCCCCCCCCCCCCC TZCPCKNHXULUIY-RGULYWFUSA-N 0.000 description 1
- CMLUGNQVANVZHY-POURPWNDSA-N 2-[1-[2-[(3r,5s)-1-(3-acetyloxy-2,2-dimethylpropyl)-7-chloro-5-(2,3-dimethoxyphenyl)-2-oxo-5h-4,1-benzoxazepin-3-yl]acetyl]piperidin-4-yl]acetic acid Chemical compound COC1=CC=CC([C@@H]2C3=CC(Cl)=CC=C3N(CC(C)(C)COC(C)=O)C(=O)[C@@H](CC(=O)N3CCC(CC(O)=O)CC3)O2)=C1OC CMLUGNQVANVZHY-POURPWNDSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 1
- UAIUNKRWKOVEES-UHFFFAOYSA-N 3,3',5,5'-tetramethylbenzidine Chemical compound CC1=C(N)C(C)=CC(C=2C=C(C)C(N)=C(C)C=2)=C1 UAIUNKRWKOVEES-UHFFFAOYSA-N 0.000 description 1
- GOLORTLGFDVFDW-UHFFFAOYSA-N 3-(1h-benzimidazol-2-yl)-7-(diethylamino)chromen-2-one Chemical compound C1=CC=C2NC(C3=CC4=CC=C(C=C4OC3=O)N(CC)CC)=NC2=C1 GOLORTLGFDVFDW-UHFFFAOYSA-N 0.000 description 1
- ALEVUYMOJKJJSA-UHFFFAOYSA-N 4-hydroxy-2-propylbenzoic acid Chemical class CCCC1=CC(O)=CC=C1C(O)=O ALEVUYMOJKJJSA-UHFFFAOYSA-N 0.000 description 1
- XZIIFPSPUDAGJM-UHFFFAOYSA-N 6-chloro-2-n,2-n-diethylpyrimidine-2,4-diamine Chemical compound CCN(CC)C1=NC(N)=CC(Cl)=N1 XZIIFPSPUDAGJM-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 206010069754 Acquired gene mutation Diseases 0.000 description 1
- 102100029457 Adenine phosphoribosyltransferase Human genes 0.000 description 1
- 108010024223 Adenine phosphoribosyltransferase Proteins 0.000 description 1
- 241001164825 Adeno-associated virus - 8 Species 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- NXSFUECZFORGOG-CIUDSAMLSA-N Ala-Asn-Leu Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(O)=O NXSFUECZFORGOG-CIUDSAMLSA-N 0.000 description 1
- RXTBLQVXNIECFP-FXQIFTODSA-N Ala-Gln-Gln Chemical compound C[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O RXTBLQVXNIECFP-FXQIFTODSA-N 0.000 description 1
- KXEVYGKATAMXJJ-ACZMJKKPSA-N Ala-Glu-Asp Chemical compound C[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(O)=O KXEVYGKATAMXJJ-ACZMJKKPSA-N 0.000 description 1
- PUBLUECXJRHTBK-ACZMJKKPSA-N Ala-Glu-Ser Chemical compound C[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(O)=O PUBLUECXJRHTBK-ACZMJKKPSA-N 0.000 description 1
- ROLXPVQSRCPVGK-XDTLVQLUSA-N Ala-Glu-Tyr Chemical compound N[C@@H](C)C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O ROLXPVQSRCPVGK-XDTLVQLUSA-N 0.000 description 1
- WGDNWOMKBUXFHR-BQBZGAKWSA-N Ala-Gly-Arg Chemical compound C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N WGDNWOMKBUXFHR-BQBZGAKWSA-N 0.000 description 1
- PNALXAODQKTNLV-JBDRJPRFSA-N Ala-Ile-Ala Chemical compound C[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(O)=O PNALXAODQKTNLV-JBDRJPRFSA-N 0.000 description 1
- NYDBKUNVSALYPX-NAKRPEOUSA-N Ala-Ile-Arg Chemical compound C[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H](C(O)=O)CCCN=C(N)N NYDBKUNVSALYPX-NAKRPEOUSA-N 0.000 description 1
- IFKQPMZRDQZSHI-GHCJXIJMSA-N Ala-Ile-Asn Chemical compound [H]N[C@@H](C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(O)=O IFKQPMZRDQZSHI-GHCJXIJMSA-N 0.000 description 1
- GSHKMNKPMLXSQW-KBIXCLLPSA-N Ala-Ile-Gln Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](C)N GSHKMNKPMLXSQW-KBIXCLLPSA-N 0.000 description 1
- LXAARTARZJJCMB-CIQUZCHMSA-N Ala-Ile-Thr Chemical compound [H]N[C@@H](C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(O)=O LXAARTARZJJCMB-CIQUZCHMSA-N 0.000 description 1
- YHKANGMVQWRMAP-DCAQKATOSA-N Ala-Leu-Arg Chemical compound C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C(O)=O)CCCN=C(N)N YHKANGMVQWRMAP-DCAQKATOSA-N 0.000 description 1
- LBYMZCVBOKYZNS-CIUDSAMLSA-N Ala-Leu-Asp Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O LBYMZCVBOKYZNS-CIUDSAMLSA-N 0.000 description 1
- SOBIAADAMRHGKH-CIUDSAMLSA-N Ala-Leu-Ser Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(O)=O SOBIAADAMRHGKH-CIUDSAMLSA-N 0.000 description 1
- RGQCNKIDEQJEBT-CQDKDKBSSA-N Ala-Leu-Tyr Chemical compound C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 RGQCNKIDEQJEBT-CQDKDKBSSA-N 0.000 description 1
- QUIGLPSHIFPEOV-CIUDSAMLSA-N Ala-Lys-Ala Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(O)=O QUIGLPSHIFPEOV-CIUDSAMLSA-N 0.000 description 1
- AJBVYEYZVYPFCF-CIUDSAMLSA-N Ala-Lys-Asn Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(O)=O AJBVYEYZVYPFCF-CIUDSAMLSA-N 0.000 description 1
- SDZRIBWEVVRDQI-CIUDSAMLSA-N Ala-Lys-Asp Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(O)=O SDZRIBWEVVRDQI-CIUDSAMLSA-N 0.000 description 1
- PVQLRJRPUTXFFX-CIUDSAMLSA-N Ala-Met-Gln Chemical compound CSCC[C@H](NC(=O)[C@H](C)N)C(=O)N[C@@H](CCC(N)=O)C(O)=O PVQLRJRPUTXFFX-CIUDSAMLSA-N 0.000 description 1
- 108010011667 Ala-Phe-Ala Proteins 0.000 description 1
- IPZQNYYAYVRKKK-FXQIFTODSA-N Ala-Pro-Ala Chemical compound C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(O)=O IPZQNYYAYVRKKK-FXQIFTODSA-N 0.000 description 1
- BTRULDJUUVGRNE-DCAQKATOSA-N Ala-Pro-Lys Chemical compound C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(O)=O BTRULDJUUVGRNE-DCAQKATOSA-N 0.000 description 1
- GMGWOTQMUKYZIE-UBHSHLNASA-N Ala-Pro-Phe Chemical compound C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 GMGWOTQMUKYZIE-UBHSHLNASA-N 0.000 description 1
- VJVQKGYHIZPSNS-FXQIFTODSA-N Ala-Ser-Arg Chemical compound C[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCCN=C(N)N VJVQKGYHIZPSNS-FXQIFTODSA-N 0.000 description 1
- SYIFFFHSXBNPMC-UWJYBYFXSA-N Ala-Ser-Tyr Chemical compound C[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)N SYIFFFHSXBNPMC-UWJYBYFXSA-N 0.000 description 1
- ARHJJAAWNWOACN-FXQIFTODSA-N Ala-Ser-Val Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O ARHJJAAWNWOACN-FXQIFTODSA-N 0.000 description 1
- LFFOJBOTZUWINF-ZANVPECISA-N Ala-Trp-Gly Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@@H](N)C)C(=O)NCC(O)=O)=CNC2=C1 LFFOJBOTZUWINF-ZANVPECISA-N 0.000 description 1
- QDGMZAOSMNGBLP-MRFFXTKBSA-N Ala-Trp-Tyr Chemical compound C[C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)N[C@@H](CC3=CC=C(C=C3)O)C(=O)O)N QDGMZAOSMNGBLP-MRFFXTKBSA-N 0.000 description 1
- MTDDMSUUXNQMKK-BPNCWPANSA-N Ala-Tyr-Arg Chemical compound C[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)O)N MTDDMSUUXNQMKK-BPNCWPANSA-N 0.000 description 1
- PGNNQOJOEGFAOR-KWQFWETISA-N Ala-Tyr-Gly Chemical compound OC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](N)C)CC1=CC=C(O)C=C1 PGNNQOJOEGFAOR-KWQFWETISA-N 0.000 description 1
- QRIYOHQJRDHFKF-UWJYBYFXSA-N Ala-Tyr-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@@H](NC(=O)[C@@H](N)C)CC1=CC=C(O)C=C1 QRIYOHQJRDHFKF-UWJYBYFXSA-N 0.000 description 1
- 108700028369 Alleles Proteins 0.000 description 1
- 102000005666 Apolipoprotein A-I Human genes 0.000 description 1
- 108010059886 Apolipoprotein A-I Proteins 0.000 description 1
- 108010056301 Apolipoprotein C-III Proteins 0.000 description 1
- 102100030970 Apolipoprotein C-III Human genes 0.000 description 1
- 101001084702 Arabidopsis thaliana Histone H2B.10 Proteins 0.000 description 1
- XVLLUZMFSAYKJV-GUBZILKMSA-N Arg-Asp-Arg Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O XVLLUZMFSAYKJV-GUBZILKMSA-N 0.000 description 1
- PQWTZSNVWSOFFK-FXQIFTODSA-N Arg-Asp-Asn Chemical compound C(C[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC(=O)N)C(=O)O)N)CN=C(N)N PQWTZSNVWSOFFK-FXQIFTODSA-N 0.000 description 1
- HKRXJBBCQBAGIM-FXQIFTODSA-N Arg-Asp-Ser Chemical compound C(C[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CO)C(=O)O)N)CN=C(N)N HKRXJBBCQBAGIM-FXQIFTODSA-N 0.000 description 1
- YVTHEZNOKSAWRW-DCAQKATOSA-N Arg-Lys-Ala Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(O)=O YVTHEZNOKSAWRW-DCAQKATOSA-N 0.000 description 1
- HGKHPCFTRQDHCU-IUCAKERBSA-N Arg-Pro-Gly Chemical compound NC(N)=NCCC[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O HGKHPCFTRQDHCU-IUCAKERBSA-N 0.000 description 1
- ATABBWFGOHKROJ-GUBZILKMSA-N Arg-Pro-Ser Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O ATABBWFGOHKROJ-GUBZILKMSA-N 0.000 description 1
- KMFPQTITXUKJOV-DCAQKATOSA-N Arg-Ser-Leu Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(O)=O KMFPQTITXUKJOV-DCAQKATOSA-N 0.000 description 1
- FRBAHXABMQXSJQ-FXQIFTODSA-N Arg-Ser-Ser Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O FRBAHXABMQXSJQ-FXQIFTODSA-N 0.000 description 1
- BECXEHHOZNFFFX-IHRRRGAJSA-N Arg-Ser-Tyr Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O BECXEHHOZNFFFX-IHRRRGAJSA-N 0.000 description 1
- RYQSYXFGFOTJDJ-RHYQMDGZSA-N Arg-Thr-Leu Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O RYQSYXFGFOTJDJ-RHYQMDGZSA-N 0.000 description 1
- FSPQNLYOFCXUCE-BPUTZDHNSA-N Arg-Trp-Asn Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CCCN=C(N)N)N FSPQNLYOFCXUCE-BPUTZDHNSA-N 0.000 description 1
- NVPHRWNWTKYIST-BPNCWPANSA-N Arg-Tyr-Ala Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@H](C(=O)N[C@@H](C)C(O)=O)CC1=CC=C(O)C=C1 NVPHRWNWTKYIST-BPNCWPANSA-N 0.000 description 1
- CNBIWSCSSCAINS-UFYCRDLUSA-N Arg-Tyr-Tyr Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O CNBIWSCSSCAINS-UFYCRDLUSA-N 0.000 description 1
- QLSRIZIDQXDQHK-RCWTZXSCSA-N Arg-Val-Thr Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O QLSRIZIDQXDQHK-RCWTZXSCSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- YNDLOUMBVDVALC-ZLUOBGJFSA-N Asn-Ala-Ala Chemical compound C[C@@H](C(=O)N[C@@H](C)C(=O)O)NC(=O)[C@H](CC(=O)N)N YNDLOUMBVDVALC-ZLUOBGJFSA-N 0.000 description 1
- XYOVHPDDWCEUDY-CIUDSAMLSA-N Asn-Ala-Leu Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(O)=O XYOVHPDDWCEUDY-CIUDSAMLSA-N 0.000 description 1
- AKEBUSZTMQLNIX-UWJYBYFXSA-N Asn-Ala-Tyr Chemical compound C[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)NC(=O)[C@H](CC(=O)N)N AKEBUSZTMQLNIX-UWJYBYFXSA-N 0.000 description 1
- GMRGSBAMMMVDGG-GUBZILKMSA-N Asn-Arg-Arg Chemical compound C(C[C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)O)NC(=O)[C@H](CC(=O)N)N)CN=C(N)N GMRGSBAMMMVDGG-GUBZILKMSA-N 0.000 description 1
- QHBMKQWOIYJYMI-BYULHYEWSA-N Asn-Asn-Val Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C(C)C)C(O)=O QHBMKQWOIYJYMI-BYULHYEWSA-N 0.000 description 1
- QPTAGIPWARILES-AVGNSLFASA-N Asn-Gln-Phe Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O QPTAGIPWARILES-AVGNSLFASA-N 0.000 description 1
- NKLRWRRVYGQNIH-GHCJXIJMSA-N Asn-Ile-Ala Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(O)=O NKLRWRRVYGQNIH-GHCJXIJMSA-N 0.000 description 1
- ANPFQTJEPONRPL-UGYAYLCHSA-N Asn-Ile-Asp Chemical compound NC(=O)C[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(O)=O)C(O)=O ANPFQTJEPONRPL-UGYAYLCHSA-N 0.000 description 1
- SEKBHZJLARBNPB-GHCJXIJMSA-N Asn-Ile-Ser Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O SEKBHZJLARBNPB-GHCJXIJMSA-N 0.000 description 1
- JQBCANGGAVVERB-CFMVVWHZSA-N Asn-Ile-Tyr Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)NC(=O)[C@H](CC(=O)N)N JQBCANGGAVVERB-CFMVVWHZSA-N 0.000 description 1
- WIDVAWAQBRAKTI-YUMQZZPRSA-N Asn-Leu-Gly Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)NCC(O)=O WIDVAWAQBRAKTI-YUMQZZPRSA-N 0.000 description 1
- ICDDSTLEMLGSTB-GUBZILKMSA-N Asn-Met-Arg Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O ICDDSTLEMLGSTB-GUBZILKMSA-N 0.000 description 1
- YUUIAUXBNOHFRJ-IHRRRGAJSA-N Asn-Phe-Met Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CCSC)C(O)=O YUUIAUXBNOHFRJ-IHRRRGAJSA-N 0.000 description 1
- UYCPJVYQYARFGB-YDHLFZDLSA-N Asn-Phe-Val Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](C(C)C)C(O)=O UYCPJVYQYARFGB-YDHLFZDLSA-N 0.000 description 1
- PLTGTJAZQRGMPP-FXQIFTODSA-N Asn-Pro-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)CC(N)=O PLTGTJAZQRGMPP-FXQIFTODSA-N 0.000 description 1
- REQUGIWGOGSOEZ-ZLUOBGJFSA-N Asn-Ser-Asn Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(=O)N)C(=O)O)N)C(=O)N REQUGIWGOGSOEZ-ZLUOBGJFSA-N 0.000 description 1
- UGXYFDQFLVCDFC-CIUDSAMLSA-N Asn-Ser-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O UGXYFDQFLVCDFC-CIUDSAMLSA-N 0.000 description 1
- SNYCNNPOFYBCEK-ZLUOBGJFSA-N Asn-Ser-Ser Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O SNYCNNPOFYBCEK-ZLUOBGJFSA-N 0.000 description 1
- XIDSGDJNUJRUHE-VEVYYDQMSA-N Asn-Thr-Met Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCSC)C(O)=O XIDSGDJNUJRUHE-VEVYYDQMSA-N 0.000 description 1
- BCADFFUQHIMQAA-KKHAAJSZSA-N Asn-Thr-Val Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O BCADFFUQHIMQAA-KKHAAJSZSA-N 0.000 description 1
- DAYDURRBMDCCFL-AAEUAGOBSA-N Asn-Trp-Gly Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)NCC(=O)O)NC(=O)[C@H](CC(=O)N)N DAYDURRBMDCCFL-AAEUAGOBSA-N 0.000 description 1
- SKQTXVZTCGSRJS-SRVKXCTJSA-N Asn-Tyr-Asp Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)O)NC(=O)[C@H](CC(=O)N)N)O SKQTXVZTCGSRJS-SRVKXCTJSA-N 0.000 description 1
- DXHINQUXBZNUCF-MELADBBJSA-N Asn-Tyr-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CC2=CC=C(C=C2)O)NC(=O)[C@H](CC(=O)N)N)C(=O)O DXHINQUXBZNUCF-MELADBBJSA-N 0.000 description 1
- WQAOZCVOOYUWKG-LSJOCFKGSA-N Asn-Val-Val Chemical compound CC(C)[C@@H](C(=O)N[C@@H](C(C)C)C(=O)O)NC(=O)[C@H](CC(=O)N)N WQAOZCVOOYUWKG-LSJOCFKGSA-N 0.000 description 1
- PBVLJOIPOGUQQP-CIUDSAMLSA-N Asp-Ala-Leu Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(O)=O PBVLJOIPOGUQQP-CIUDSAMLSA-N 0.000 description 1
- ICTXFVKYAGQURS-UBHSHLNASA-N Asp-Asn-Trp Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(O)=O ICTXFVKYAGQURS-UBHSHLNASA-N 0.000 description 1
- RDRMWJBLOSRRAW-BYULHYEWSA-N Asp-Asn-Val Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C(C)C)C(O)=O RDRMWJBLOSRRAW-BYULHYEWSA-N 0.000 description 1
- VFUXXFVCYZPOQG-WDSKDSINSA-N Asp-Glu-Gly Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)NCC(O)=O VFUXXFVCYZPOQG-WDSKDSINSA-N 0.000 description 1
- SEMWSADZTMJELF-BYULHYEWSA-N Asp-Ile-Gly Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(O)=O SEMWSADZTMJELF-BYULHYEWSA-N 0.000 description 1
- YWLDTBBUHZJQHW-KKUMJFAQSA-N Asp-Lys-Phe Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(=O)O)N YWLDTBBUHZJQHW-KKUMJFAQSA-N 0.000 description 1
- RPUYTJJZXQBWDT-SRVKXCTJSA-N Asp-Phe-Ser Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CC(=O)O)N RPUYTJJZXQBWDT-SRVKXCTJSA-N 0.000 description 1
- KGHLGJAXYSVNJP-WHFBIAKZSA-N Asp-Ser-Gly Chemical compound OC(=O)C[C@H](N)C(=O)N[C@@H](CO)C(=O)NCC(O)=O KGHLGJAXYSVNJP-WHFBIAKZSA-N 0.000 description 1
- GHAHOJDCBRXAKC-IHPCNDPISA-N Asp-Trp-Tyr Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CC3=CC=C(C=C3)O)C(=O)O)NC(=O)[C@H](CC(=O)O)N GHAHOJDCBRXAKC-IHPCNDPISA-N 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- 238000000035 BCA protein assay Methods 0.000 description 1
- 235000014469 Bacillus subtilis Nutrition 0.000 description 1
- KPYSYYIEGFHWSV-UHFFFAOYSA-N Baclofen Chemical compound OC(=O)CC(CN)C1=CC=C(Cl)C=C1 KPYSYYIEGFHWSV-UHFFFAOYSA-N 0.000 description 1
- DWRXFEITVBNRMK-UHFFFAOYSA-N Beta-D-1-Arabinofuranosylthymine Natural products O=C1NC(=O)C(C)=CN1C1C(O)C(O)C(CO)O1 DWRXFEITVBNRMK-UHFFFAOYSA-N 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 101100348617 Candida albicans (strain SC5314 / ATCC MYA-2876) NIK1 gene Proteins 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 241000701489 Cauliflower mosaic virus Species 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 241000195597 Chlamydomonas reinhardtii Species 0.000 description 1
- 241000195628 Chlorophyta Species 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- TVYMKYUSZSVOAG-ZLUOBGJFSA-N Cys-Ala-Ala Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(O)=O TVYMKYUSZSVOAG-ZLUOBGJFSA-N 0.000 description 1
- PRXCTTWKGJAPMT-ZLUOBGJFSA-N Cys-Ala-Ser Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O PRXCTTWKGJAPMT-ZLUOBGJFSA-N 0.000 description 1
- SZQCDCKIGWQAQN-FXQIFTODSA-N Cys-Arg-Ala Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(O)=O SZQCDCKIGWQAQN-FXQIFTODSA-N 0.000 description 1
- ZEXHDOQQYZKOIB-ACZMJKKPSA-N Cys-Glu-Ser Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(O)=O ZEXHDOQQYZKOIB-ACZMJKKPSA-N 0.000 description 1
- NIXHTNJAGGFBAW-CIUDSAMLSA-N Cys-Lys-Ser Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CS)N NIXHTNJAGGFBAW-CIUDSAMLSA-N 0.000 description 1
- ZXCAQANTQWBICD-DCAQKATOSA-N Cys-Lys-Val Chemical compound CC(C)[C@@H](C(=O)O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CS)N ZXCAQANTQWBICD-DCAQKATOSA-N 0.000 description 1
- NDNZRWUDUMTITL-FXQIFTODSA-N Cys-Ser-Val Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O NDNZRWUDUMTITL-FXQIFTODSA-N 0.000 description 1
- LPBUBIHAVKXUOT-FXQIFTODSA-N Cys-Val-Ser Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CS)N LPBUBIHAVKXUOT-FXQIFTODSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- KDXKERNSBIXSRK-RXMQYKEDSA-N D-lysine group Chemical group N[C@H](CCCCN)C(=O)O KDXKERNSBIXSRK-RXMQYKEDSA-N 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- 101150074155 DHFR gene Proteins 0.000 description 1
- 230000004544 DNA amplification Effects 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 1
- 239000012591 Dulbecco’s Phosphate Buffered Saline Substances 0.000 description 1
- 241000283074 Equus asinus Species 0.000 description 1
- 108010074860 Factor Xa Proteins 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 108700028146 Genetic Enhancer Elements Proteins 0.000 description 1
- RZSLYUUFFVHFRQ-FXQIFTODSA-N Gln-Ala-Glu Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(O)=O RZSLYUUFFVHFRQ-FXQIFTODSA-N 0.000 description 1
- LPYPANUXJGFMGV-FXQIFTODSA-N Gln-Gln-Ala Chemical compound C[C@@H](C(=O)O)NC(=O)[C@H](CCC(=O)N)NC(=O)[C@H](CCC(=O)N)N LPYPANUXJGFMGV-FXQIFTODSA-N 0.000 description 1
- KVXVVDFOZNYYKZ-DCAQKATOSA-N Gln-Gln-Leu Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(O)=O KVXVVDFOZNYYKZ-DCAQKATOSA-N 0.000 description 1
- MADFVRSKEIEZHZ-DCAQKATOSA-N Gln-Gln-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)[C@H](CCC(=O)N)NC(=O)[C@H](CCC(=O)N)N MADFVRSKEIEZHZ-DCAQKATOSA-N 0.000 description 1
- VSXBYIJUAXPAAL-WDSKDSINSA-N Gln-Gly-Ala Chemical compound OC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](N)CCC(N)=O VSXBYIJUAXPAAL-WDSKDSINSA-N 0.000 description 1
- IKFZXRLDMYWNBU-YUMQZZPRSA-N Gln-Gly-Arg Chemical compound NC(=O)CC[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N IKFZXRLDMYWNBU-YUMQZZPRSA-N 0.000 description 1
- FGYPOQPQTUNESW-IUCAKERBSA-N Gln-Gly-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)CNC(=O)[C@H](CCC(=O)N)N FGYPOQPQTUNESW-IUCAKERBSA-N 0.000 description 1
- VGTDBGYFVWOQTI-RYUDHWBXSA-N Gln-Gly-Phe Chemical compound NC(=O)CC[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 VGTDBGYFVWOQTI-RYUDHWBXSA-N 0.000 description 1
- ZBKUIQNCRIYVGH-SDDRHHMPSA-N Gln-Leu-Pro Chemical compound CC(C)C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCC(=O)N)N ZBKUIQNCRIYVGH-SDDRHHMPSA-N 0.000 description 1
- MSHXWFKYXJTLEZ-CIUDSAMLSA-N Gln-Met-Asn Chemical compound CSCC[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CCC(=O)N)N MSHXWFKYXJTLEZ-CIUDSAMLSA-N 0.000 description 1
- YPFFHGRJCUBXPX-NHCYSSNCSA-N Gln-Pro-Val Chemical compound CC(C)[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)CCC(N)=O)C(O)=O YPFFHGRJCUBXPX-NHCYSSNCSA-N 0.000 description 1
- SYZZMPFLOLSMHL-XHNCKOQMSA-N Gln-Ser-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CO)NC(=O)[C@H](CCC(=O)N)N)C(=O)O SYZZMPFLOLSMHL-XHNCKOQMSA-N 0.000 description 1
- OTQSTOXRUBVWAP-NRPADANISA-N Gln-Ser-Val Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O OTQSTOXRUBVWAP-NRPADANISA-N 0.000 description 1
- VLOLPWWCNKWRNB-LOKLDPHHSA-N Gln-Thr-Pro Chemical compound C[C@H]([C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCC(=O)N)N)O VLOLPWWCNKWRNB-LOKLDPHHSA-N 0.000 description 1
- WZZSKAJIHTUUSG-ACZMJKKPSA-N Glu-Ala-Asp Chemical compound OC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCC(O)=O WZZSKAJIHTUUSG-ACZMJKKPSA-N 0.000 description 1
- LKDIBBOKUAASNP-FXQIFTODSA-N Glu-Ala-Glu Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(O)=O LKDIBBOKUAASNP-FXQIFTODSA-N 0.000 description 1
- LTUVYLVIZHJCOQ-KKUMJFAQSA-N Glu-Arg-Phe Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O LTUVYLVIZHJCOQ-KKUMJFAQSA-N 0.000 description 1
- VGUYMZGLJUJRBV-YVNDNENWSA-N Glu-Ile-Glu Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(O)=O VGUYMZGLJUJRBV-YVNDNENWSA-N 0.000 description 1
- INGJLBQKTRJLFO-UKJIMTQDSA-N Glu-Ile-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@@H](N)CCC(O)=O INGJLBQKTRJLFO-UKJIMTQDSA-N 0.000 description 1
- SUIAHERNFYRBDZ-GVXVVHGQSA-N Glu-Lys-Val Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(O)=O SUIAHERNFYRBDZ-GVXVVHGQSA-N 0.000 description 1
- MRWYPDWDZSLWJM-ACZMJKKPSA-N Glu-Ser-Asp Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(O)=O MRWYPDWDZSLWJM-ACZMJKKPSA-N 0.000 description 1
- BXSZPACYCMNKLS-AVGNSLFASA-N Glu-Ser-Phe Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O BXSZPACYCMNKLS-AVGNSLFASA-N 0.000 description 1
- YPHPEHMXOYTEQG-LAEOZQHASA-N Glu-Val-Asp Chemical compound OC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)CCC(O)=O YPHPEHMXOYTEQG-LAEOZQHASA-N 0.000 description 1
- 102100040890 Glucagon receptor Human genes 0.000 description 1
- 239000004366 Glucose oxidase Substances 0.000 description 1
- 108010015776 Glucose oxidase Proteins 0.000 description 1
- LJPIRKICOISLKN-WHFBIAKZSA-N Gly-Ala-Ser Chemical compound NCC(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O LJPIRKICOISLKN-WHFBIAKZSA-N 0.000 description 1
- KRRMJKMGWWXWDW-STQMWFEESA-N Gly-Arg-Phe Chemical compound NC(=N)NCCC[C@H](NC(=O)CN)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 KRRMJKMGWWXWDW-STQMWFEESA-N 0.000 description 1
- GWCRIHNSVMOBEQ-BQBZGAKWSA-N Gly-Arg-Ser Chemical compound [H]NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(O)=O GWCRIHNSVMOBEQ-BQBZGAKWSA-N 0.000 description 1
- XUORRGAFUQIMLC-STQMWFEESA-N Gly-Arg-Tyr Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)O)NC(=O)[C@H](CCCN=C(N)N)NC(=O)CN)O XUORRGAFUQIMLC-STQMWFEESA-N 0.000 description 1
- UXJHNZODTMHWRD-WHFBIAKZSA-N Gly-Asn-Ala Chemical compound [H]NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(O)=O UXJHNZODTMHWRD-WHFBIAKZSA-N 0.000 description 1
- LCNXZQROPKFGQK-WHFBIAKZSA-N Gly-Asp-Ser Chemical compound NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O LCNXZQROPKFGQK-WHFBIAKZSA-N 0.000 description 1
- PMNHJLASAAWELO-FOHZUACHSA-N Gly-Asp-Thr Chemical compound [H]NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O PMNHJLASAAWELO-FOHZUACHSA-N 0.000 description 1
- GNPVTZJUUBPZKW-WDSKDSINSA-N Gly-Gln-Ser Chemical compound [H]NCC(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(O)=O GNPVTZJUUBPZKW-WDSKDSINSA-N 0.000 description 1
- JSNNHGHYGYMVCK-XVKPBYJWSA-N Gly-Glu-Val Chemical compound [H]NCC(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(O)=O JSNNHGHYGYMVCK-XVKPBYJWSA-N 0.000 description 1
- CCQOOWAONKGYKQ-BYPYZUCNSA-N Gly-Gly-Ala Chemical compound OC(=O)[C@H](C)NC(=O)CNC(=O)CN CCQOOWAONKGYKQ-BYPYZUCNSA-N 0.000 description 1
- HQRHFUYMGCHHJS-LURJTMIESA-N Gly-Gly-Arg Chemical compound NCC(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N HQRHFUYMGCHHJS-LURJTMIESA-N 0.000 description 1
- KMSGYZQRXPUKGI-BYPYZUCNSA-N Gly-Gly-Asn Chemical compound NCC(=O)NCC(=O)N[C@H](C(O)=O)CC(N)=O KMSGYZQRXPUKGI-BYPYZUCNSA-N 0.000 description 1
- XPJBQTCXPJNIFE-ZETCQYMHSA-N Gly-Gly-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)CNC(=O)CN XPJBQTCXPJNIFE-ZETCQYMHSA-N 0.000 description 1
- AYBKPDHHVADEDA-YUMQZZPRSA-N Gly-His-Asn Chemical compound [H]NCC(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(N)=O)C(O)=O AYBKPDHHVADEDA-YUMQZZPRSA-N 0.000 description 1
- BHPQOIPBLYJNAW-NGZCFLSTSA-N Gly-Ile-Pro Chemical compound CC[C@H](C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)CN BHPQOIPBLYJNAW-NGZCFLSTSA-N 0.000 description 1
- LHYJCVCQPWRMKZ-WEDXCCLWSA-N Gly-Leu-Thr Chemical compound [H]NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O LHYJCVCQPWRMKZ-WEDXCCLWSA-N 0.000 description 1
- CLNSYANKYVMZNM-UWVGGRQHSA-N Gly-Lys-Arg Chemical compound NCCCC[C@H](NC(=O)CN)C(=O)N[C@H](C(O)=O)CCCN=C(N)N CLNSYANKYVMZNM-UWVGGRQHSA-N 0.000 description 1
- VDCRBJACQKOSMS-JSGCOSHPSA-N Gly-Phe-Val Chemical compound [H]NCC(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](C(C)C)C(O)=O VDCRBJACQKOSMS-JSGCOSHPSA-N 0.000 description 1
- OOCFXNOVSLSHAB-IUCAKERBSA-N Gly-Pro-Pro Chemical compound NCC(=O)N1CCC[C@H]1C(=O)N1[C@H](C(O)=O)CCC1 OOCFXNOVSLSHAB-IUCAKERBSA-N 0.000 description 1
- YOBGUCWZPXJHTN-BQBZGAKWSA-N Gly-Ser-Arg Chemical compound NCC(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCCN=C(N)N YOBGUCWZPXJHTN-BQBZGAKWSA-N 0.000 description 1
- WNGHUXFWEWTKAO-YUMQZZPRSA-N Gly-Ser-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)CN WNGHUXFWEWTKAO-YUMQZZPRSA-N 0.000 description 1
- POJJAZJHBGXEGM-YUMQZZPRSA-N Gly-Ser-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)[C@H](CO)NC(=O)CN POJJAZJHBGXEGM-YUMQZZPRSA-N 0.000 description 1
- TVTZEOHWHUVYCG-KYNKHSRBSA-N Gly-Thr-Thr Chemical compound [H]NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O TVTZEOHWHUVYCG-KYNKHSRBSA-N 0.000 description 1
- CUVBTVWFVIIDOC-YEPSODPASA-N Gly-Thr-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)CN CUVBTVWFVIIDOC-YEPSODPASA-N 0.000 description 1
- UMRIXLHPZZIOML-OALUTQOASA-N Gly-Trp-Phe Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)O)NC(=O)[C@H](CC2=CNC3=CC=CC=C32)NC(=O)CN UMRIXLHPZZIOML-OALUTQOASA-N 0.000 description 1
- IROABALAWGJQGM-OALUTQOASA-N Gly-Trp-Tyr Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CC3=CC=C(C=C3)O)C(=O)O)NC(=O)CN IROABALAWGJQGM-OALUTQOASA-N 0.000 description 1
- MREVELMMFOLESM-HOCLYGCPSA-N Gly-Trp-Val Chemical compound [H]NCC(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](C(C)C)C(O)=O MREVELMMFOLESM-HOCLYGCPSA-N 0.000 description 1
- UIQGJYUEQDOODF-KWQFWETISA-N Gly-Tyr-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)CN)CC1=CC=C(O)C=C1 UIQGJYUEQDOODF-KWQFWETISA-N 0.000 description 1
- GNNJKUYDWFIBTK-QWRGUYRKSA-N Gly-Tyr-Asp Chemical compound [H]NCC(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC(O)=O)C(O)=O GNNJKUYDWFIBTK-QWRGUYRKSA-N 0.000 description 1
- UVTSZKIATYSKIR-RYUDHWBXSA-N Gly-Tyr-Glu Chemical compound [H]NCC(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCC(O)=O)C(O)=O UVTSZKIATYSKIR-RYUDHWBXSA-N 0.000 description 1
- HQSKKSLNLSTONK-JTQLQIEISA-N Gly-Tyr-Gly Chemical compound OC(=O)CNC(=O)[C@@H](NC(=O)CN)CC1=CC=C(O)C=C1 HQSKKSLNLSTONK-JTQLQIEISA-N 0.000 description 1
- DNAZKGFYFRGZIH-QWRGUYRKSA-N Gly-Tyr-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@@H](NC(=O)CN)CC1=CC=C(O)C=C1 DNAZKGFYFRGZIH-QWRGUYRKSA-N 0.000 description 1
- GBYYQVBXFVDJPJ-WLTAIBSBSA-N Gly-Tyr-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)CN)O GBYYQVBXFVDJPJ-WLTAIBSBSA-N 0.000 description 1
- SBVMXEZQJVUARN-XPUUQOCRSA-N Gly-Val-Ser Chemical compound NCC(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O SBVMXEZQJVUARN-XPUUQOCRSA-N 0.000 description 1
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 1
- ZWZWYGMENQVNFU-UHFFFAOYSA-N Glycerophosphorylserin Natural products OC(=O)C(N)COP(O)(=O)OCC(O)CO ZWZWYGMENQVNFU-UHFFFAOYSA-N 0.000 description 1
- 201000005569 Gout Diseases 0.000 description 1
- JBCLFWXMTIKCCB-UHFFFAOYSA-N H-Gly-Phe-OH Natural products NCC(=O)NC(C(O)=O)CC1=CC=CC=C1 JBCLFWXMTIKCCB-UHFFFAOYSA-N 0.000 description 1
- HVLSXIKZNLPZJJ-TXZCQADKSA-N HA peptide Chemical compound C([C@@H](C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](C)C(O)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=C(O)C=C1 HVLSXIKZNLPZJJ-TXZCQADKSA-N 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- PROLDOGUBQJNPG-RWMBFGLXSA-N His-Arg-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC2=CN=CN2)N)C(=O)O PROLDOGUBQJNPG-RWMBFGLXSA-N 0.000 description 1
- VIVSWEBJUHXCDS-DCAQKATOSA-N His-Asn-Met Chemical compound [H]N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCSC)C(O)=O VIVSWEBJUHXCDS-DCAQKATOSA-N 0.000 description 1
- SKOKHBGDXGTDDP-MELADBBJSA-N His-Leu-Pro Chemical compound CC(C)C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CC2=CN=CN2)N SKOKHBGDXGTDDP-MELADBBJSA-N 0.000 description 1
- UXZMINKIEWBEQU-SZMVWBNQSA-N His-Trp-Gln Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CC3=CN=CN3)N UXZMINKIEWBEQU-SZMVWBNQSA-N 0.000 description 1
- UWNUQPZUSRFIIN-JUKXBJQTSA-N His-Tyr-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)[C@H](CC2=CN=CN2)N UWNUQPZUSRFIIN-JUKXBJQTSA-N 0.000 description 1
- CSTDQOOBZBAJKE-BWAGICSOSA-N His-Tyr-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)[C@H](CC2=CN=CN2)N)O CSTDQOOBZBAJKE-BWAGICSOSA-N 0.000 description 1
- HIJIJPFILYPTFR-ACRUOGEOSA-N His-Tyr-Tyr Chemical compound [H]N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O HIJIJPFILYPTFR-ACRUOGEOSA-N 0.000 description 1
- 101100323490 Homo sapiens APOC3 gene Proteins 0.000 description 1
- 101000793223 Homo sapiens Apolipoprotein C-III Proteins 0.000 description 1
- 101001040075 Homo sapiens Glucagon receptor Proteins 0.000 description 1
- 101100321817 Human parvovirus B19 (strain HV) 7.5K gene Proteins 0.000 description 1
- UGQMRVRMYYASKQ-UHFFFAOYSA-N Hypoxanthine nucleoside Natural products OC1C(O)C(CO)OC1N1C(NC=NC2=O)=C2N=C1 UGQMRVRMYYASKQ-UHFFFAOYSA-N 0.000 description 1
- AQCUAZTZSPQJFF-ZKWXMUAHSA-N Ile-Ala-Gly Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](C)C(=O)NCC(O)=O AQCUAZTZSPQJFF-ZKWXMUAHSA-N 0.000 description 1
- RPZFUIQVAPZLRH-GHCJXIJMSA-N Ile-Asp-Ala Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](C)C(=O)O)N RPZFUIQVAPZLRH-GHCJXIJMSA-N 0.000 description 1
- NKRJALPCDNXULF-BYULHYEWSA-N Ile-Asp-Gly Chemical compound [H]N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(O)=O NKRJALPCDNXULF-BYULHYEWSA-N 0.000 description 1
- YBJWJQQBWRARLT-KBIXCLLPSA-N Ile-Gln-Ser Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(O)=O YBJWJQQBWRARLT-KBIXCLLPSA-N 0.000 description 1
- LBRCLQMZAHRTLV-ZKWXMUAHSA-N Ile-Gly-Ser Chemical compound CC[C@H](C)[C@H](N)C(=O)NCC(=O)N[C@@H](CO)C(O)=O LBRCLQMZAHRTLV-ZKWXMUAHSA-N 0.000 description 1
- KEKTTYCXKGBAAL-VGDYDELISA-N Ile-His-Ser Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)N[C@@H](CO)C(=O)O)N KEKTTYCXKGBAAL-VGDYDELISA-N 0.000 description 1
- URWXDJAEEGBADB-TUBUOCAGSA-N Ile-His-Thr Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)N[C@@H]([C@@H](C)O)C(=O)O)N URWXDJAEEGBADB-TUBUOCAGSA-N 0.000 description 1
- DSDPLOODKXISDT-XUXIUFHCSA-N Ile-Leu-Val Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(O)=O DSDPLOODKXISDT-XUXIUFHCSA-N 0.000 description 1
- KCTIFOCXAIUQQK-QXEWZRGKSA-N Ile-Pro-Gly Chemical compound CC[C@H](C)[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O KCTIFOCXAIUQQK-QXEWZRGKSA-N 0.000 description 1
- CAHCWMVNBZJVAW-NAKRPEOUSA-N Ile-Pro-Ser Chemical compound CC[C@H](C)[C@@H](C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)O)N CAHCWMVNBZJVAW-NAKRPEOUSA-N 0.000 description 1
- PXKACEXYLPBMAD-JBDRJPRFSA-N Ile-Ser-Ser Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)O)N PXKACEXYLPBMAD-JBDRJPRFSA-N 0.000 description 1
- SAEWJTCJQVZQNZ-IUKAMOBKSA-N Ile-Thr-Asn Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(=O)N)C(=O)O)N SAEWJTCJQVZQNZ-IUKAMOBKSA-N 0.000 description 1
- ZGKVPOSSTGHJAF-HJPIBITLSA-N Ile-Tyr-Ser Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CO)C(=O)O)N ZGKVPOSSTGHJAF-HJPIBITLSA-N 0.000 description 1
- NGKPIPCGMLWHBX-WZLNRYEVSA-N Ile-Tyr-Thr Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)O)N NGKPIPCGMLWHBX-WZLNRYEVSA-N 0.000 description 1
- 108020005350 Initiator Codon Proteins 0.000 description 1
- 101710172072 Kexin Proteins 0.000 description 1
- 238000012449 Kunming mouse Methods 0.000 description 1
- HGCNKOLVKRAVHD-UHFFFAOYSA-N L-Met-L-Phe Natural products CSCCC(N)C(=O)NC(C(O)=O)CC1=CC=CC=C1 HGCNKOLVKRAVHD-UHFFFAOYSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- LHSGPCFBGJHPCY-UHFFFAOYSA-N L-leucine-L-tyrosine Natural products CC(C)CC(N)C(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 LHSGPCFBGJHPCY-UHFFFAOYSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- TYYLDKGBCJGJGW-UHFFFAOYSA-N L-tryptophan-L-tyrosine Natural products C=1NC2=CC=CC=C2C=1CC(N)C(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 TYYLDKGBCJGJGW-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- JUWJEAPUNARGCF-DCAQKATOSA-N Leu-Arg-Ala Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(O)=O JUWJEAPUNARGCF-DCAQKATOSA-N 0.000 description 1
- UCOCBWDBHCUPQP-DCAQKATOSA-N Leu-Arg-Ser Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(O)=O UCOCBWDBHCUPQP-DCAQKATOSA-N 0.000 description 1
- USTCFDAQCLDPBD-XIRDDKMYSA-N Leu-Asn-Trp Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)O)N USTCFDAQCLDPBD-XIRDDKMYSA-N 0.000 description 1
- XVSJMWYYLHPDKY-DCAQKATOSA-N Leu-Asp-Met Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCSC)C(O)=O XVSJMWYYLHPDKY-DCAQKATOSA-N 0.000 description 1
- MMEDVBWCMGRKKC-GARJFASQSA-N Leu-Asp-Pro Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N1CCC[C@@H]1C(=O)O)N MMEDVBWCMGRKKC-GARJFASQSA-N 0.000 description 1
- ZTLGVASZOIKNIX-DCAQKATOSA-N Leu-Gln-Glu Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CCC(=O)O)C(=O)O)N ZTLGVASZOIKNIX-DCAQKATOSA-N 0.000 description 1
- RSFGIMMPWAXNML-MNXVOIDGSA-N Leu-Gln-Ile Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O RSFGIMMPWAXNML-MNXVOIDGSA-N 0.000 description 1
- CQGSYZCULZMEDE-UHFFFAOYSA-N Leu-Gln-Pro Natural products CC(C)CC(N)C(=O)NC(CCC(N)=O)C(=O)N1CCCC1C(O)=O CQGSYZCULZMEDE-UHFFFAOYSA-N 0.000 description 1
- PRZVBIAOPFGAQF-SRVKXCTJSA-N Leu-Glu-Met Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCSC)C(O)=O PRZVBIAOPFGAQF-SRVKXCTJSA-N 0.000 description 1
- HVJVUYQWFYMGJS-GVXVVHGQSA-N Leu-Glu-Val Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(O)=O HVJVUYQWFYMGJS-GVXVVHGQSA-N 0.000 description 1
- BABSVXFGKFLIGW-UWVGGRQHSA-N Leu-Gly-Arg Chemical compound CC(C)C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCNC(N)=N BABSVXFGKFLIGW-UWVGGRQHSA-N 0.000 description 1
- HRTRLSRYZZKPCO-BJDJZHNGSA-N Leu-Ile-Ser Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O HRTRLSRYZZKPCO-BJDJZHNGSA-N 0.000 description 1
- FAELBUXXFQLUAX-AJNGGQMLSA-N Leu-Leu-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CC(C)C FAELBUXXFQLUAX-AJNGGQMLSA-N 0.000 description 1
- VULJUQZPSOASBZ-SRVKXCTJSA-N Leu-Pro-Glu Chemical compound [H]N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O VULJUQZPSOASBZ-SRVKXCTJSA-N 0.000 description 1
- RGUXWMDNCPMQFB-YUMQZZPRSA-N Leu-Ser-Gly Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CO)C(=O)NCC(O)=O RGUXWMDNCPMQFB-YUMQZZPRSA-N 0.000 description 1
- LJBVRCDPWOJOEK-PPCPHDFISA-N Leu-Thr-Ile Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O LJBVRCDPWOJOEK-PPCPHDFISA-N 0.000 description 1
- ODRREERHVHMIPT-OEAJRASXSA-N Leu-Thr-Phe Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 ODRREERHVHMIPT-OEAJRASXSA-N 0.000 description 1
- KLSUAWUZBMAZCL-RHYQMDGZSA-N Leu-Thr-Pro Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1CCC[C@H]1C(O)=O KLSUAWUZBMAZCL-RHYQMDGZSA-N 0.000 description 1
- HOMFINRJHIIZNJ-HOCLYGCPSA-N Leu-Trp-Gly Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)NCC(O)=O HOMFINRJHIIZNJ-HOCLYGCPSA-N 0.000 description 1
- RIHIGSWBLHSGLV-CQDKDKBSSA-N Leu-Tyr-Ala Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](C)C(O)=O RIHIGSWBLHSGLV-CQDKDKBSSA-N 0.000 description 1
- YIRIDPUGZKHMHT-ACRUOGEOSA-N Leu-Tyr-Tyr Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O YIRIDPUGZKHMHT-ACRUOGEOSA-N 0.000 description 1
- MVJRBCJCRYGCKV-GVXVVHGQSA-N Leu-Val-Gln Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O MVJRBCJCRYGCKV-GVXVVHGQSA-N 0.000 description 1
- QESXLSQLQHHTIX-RHYQMDGZSA-N Leu-Val-Thr Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O QESXLSQLQHHTIX-RHYQMDGZSA-N 0.000 description 1
- 229940127470 Lipase Inhibitors Drugs 0.000 description 1
- XFIHDSBIPWEYJJ-YUMQZZPRSA-N Lys-Ala-Gly Chemical compound OC(=O)CNC(=O)[C@H](C)NC(=O)[C@@H](N)CCCCN XFIHDSBIPWEYJJ-YUMQZZPRSA-N 0.000 description 1
- NFLFJGGKOHYZJF-BJDJZHNGSA-N Lys-Ala-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCCCN NFLFJGGKOHYZJF-BJDJZHNGSA-N 0.000 description 1
- KNKHAVVBVXKOGX-JXUBOQSCSA-N Lys-Ala-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O KNKHAVVBVXKOGX-JXUBOQSCSA-N 0.000 description 1
- SWWCDAGDQHTKIE-RHYQMDGZSA-N Lys-Arg-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(O)=O SWWCDAGDQHTKIE-RHYQMDGZSA-N 0.000 description 1
- AAORVPFVUIHEAB-YUMQZZPRSA-N Lys-Asp-Gly Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(O)=O AAORVPFVUIHEAB-YUMQZZPRSA-N 0.000 description 1
- LMVOVCYVZBBWQB-SRVKXCTJSA-N Lys-Asp-Lys Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CCCCN LMVOVCYVZBBWQB-SRVKXCTJSA-N 0.000 description 1
- ITWQLSZTLBKWJM-YUMQZZPRSA-N Lys-Gly-Ala Chemical compound OC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](N)CCCCN ITWQLSZTLBKWJM-YUMQZZPRSA-N 0.000 description 1
- KZJQUYFDSCFSCO-DLOVCJGASA-N Lys-His-Ala Chemical compound C[C@@H](C(=O)O)NC(=O)[C@H](CC1=CN=CN1)NC(=O)[C@H](CCCCN)N KZJQUYFDSCFSCO-DLOVCJGASA-N 0.000 description 1
- KNKJPYAZQUFLQK-IHRRRGAJSA-N Lys-His-Arg Chemical compound C1=C(NC=N1)C[C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)O)NC(=O)[C@H](CCCCN)N KNKJPYAZQUFLQK-IHRRRGAJSA-N 0.000 description 1
- SKRGVGLIRUGANF-AVGNSLFASA-N Lys-Leu-Glu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O SKRGVGLIRUGANF-AVGNSLFASA-N 0.000 description 1
- OIQSIMFSVLLWBX-VOAKCMCISA-N Lys-Leu-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O OIQSIMFSVLLWBX-VOAKCMCISA-N 0.000 description 1
- UDXSLGLHFUBRRM-OEAJRASXSA-N Lys-Phe-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=CC=C1)NC(=O)[C@H](CCCCN)N)O UDXSLGLHFUBRRM-OEAJRASXSA-N 0.000 description 1
- YSZNURNVYFUEHC-BQBZGAKWSA-N Lys-Ser Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CO)C(O)=O YSZNURNVYFUEHC-BQBZGAKWSA-N 0.000 description 1
- DNWBUCHHMRQWCZ-GUBZILKMSA-N Lys-Ser-Gln Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCC(N)=O DNWBUCHHMRQWCZ-GUBZILKMSA-N 0.000 description 1
- DIBZLYZXTSVGLN-CIUDSAMLSA-N Lys-Ser-Ser Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O DIBZLYZXTSVGLN-CIUDSAMLSA-N 0.000 description 1
- RQILLQOQXLZTCK-KBPBESRZSA-N Lys-Tyr-Gly Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)NCC(O)=O RQILLQOQXLZTCK-KBPBESRZSA-N 0.000 description 1
- SQRLLZAQNOQCEG-KKUMJFAQSA-N Lys-Tyr-Ser Chemical compound NCCCC[C@H](N)C(=O)N[C@H](C(=O)N[C@@H](CO)C(O)=O)CC1=CC=C(O)C=C1 SQRLLZAQNOQCEG-KKUMJFAQSA-N 0.000 description 1
- FPQMQEOVSKMVMA-ACRUOGEOSA-N Lys-Tyr-Tyr Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)O)NC(=O)[C@H](CCCCN)N)O FPQMQEOVSKMVMA-ACRUOGEOSA-N 0.000 description 1
- UGCIQUYEJIEHKX-GVXVVHGQSA-N Lys-Val-Glu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O UGCIQUYEJIEHKX-GVXVVHGQSA-N 0.000 description 1
- GILLQRYAWOMHED-DCAQKATOSA-N Lys-Val-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)CCCCN GILLQRYAWOMHED-DCAQKATOSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 241000282553 Macaca Species 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- PNDCUTDWYVKBHX-IHRRRGAJSA-N Met-Asp-Tyr Chemical compound CSCC[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 PNDCUTDWYVKBHX-IHRRRGAJSA-N 0.000 description 1
- LRALLISKBZNSKN-BQBZGAKWSA-N Met-Gly-Ser Chemical compound CSCC[C@H](N)C(=O)NCC(=O)N[C@@H](CO)C(O)=O LRALLISKBZNSKN-BQBZGAKWSA-N 0.000 description 1
- NDJSSFWDYDUQID-YTWAJWBKSA-N Met-Thr-Pro Chemical compound C[C@H]([C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCSC)N)O NDJSSFWDYDUQID-YTWAJWBKSA-N 0.000 description 1
- 102000003792 Metallothionein Human genes 0.000 description 1
- 108090000157 Metallothionein Proteins 0.000 description 1
- YBAFDPFAUTYYRW-UHFFFAOYSA-N N-L-alpha-glutamyl-L-leucine Natural products CC(C)CC(C(O)=O)NC(=O)C(N)CCC(O)=O YBAFDPFAUTYYRW-UHFFFAOYSA-N 0.000 description 1
- AUEJLPRZGVVDNU-UHFFFAOYSA-N N-L-tyrosyl-L-leucine Natural products CC(C)CC(C(O)=O)NC(=O)C(N)CC1=CC=C(O)C=C1 AUEJLPRZGVVDNU-UHFFFAOYSA-N 0.000 description 1
- 108091007491 NSP3 Papain-like protease domains Proteins 0.000 description 1
- BQVUABVGYYSDCJ-UHFFFAOYSA-N Nalpha-L-Leucyl-L-tryptophan Natural products C1=CC=C2C(CC(NC(=O)C(N)CC(C)C)C(O)=O)=CNC2=C1 BQVUABVGYYSDCJ-UHFFFAOYSA-N 0.000 description 1
- 229930193140 Neomycin Natural products 0.000 description 1
- 108090000526 Papain Proteins 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- 102000057297 Pepsin A Human genes 0.000 description 1
- 108090000284 Pepsin A Proteins 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 241000009328 Perro Species 0.000 description 1
- UHRNIXJAGGLKHP-DLOVCJGASA-N Phe-Ala-Ser Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O UHRNIXJAGGLKHP-DLOVCJGASA-N 0.000 description 1
- HTTYNOXBBOWZTB-SRVKXCTJSA-N Phe-Asn-Asn Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CC(=O)N)C(=O)O)N HTTYNOXBBOWZTB-SRVKXCTJSA-N 0.000 description 1
- DJPXNKUDJKGQEE-BZSNNMDCSA-N Phe-Asp-Phe Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O DJPXNKUDJKGQEE-BZSNNMDCSA-N 0.000 description 1
- QPVFUAUFEBPIPT-CDMKHQONSA-N Phe-Gly-Thr Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(O)=O QPVFUAUFEBPIPT-CDMKHQONSA-N 0.000 description 1
- ILGCZYGFYQLSDZ-KKUMJFAQSA-N Phe-Ser-His Chemical compound N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CO)C(=O)N[C@@H](Cc1cnc[nH]1)C(O)=O ILGCZYGFYQLSDZ-KKUMJFAQSA-N 0.000 description 1
- JXQVYPWVGUOIDV-MXAVVETBSA-N Phe-Ser-Ile Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O JXQVYPWVGUOIDV-MXAVVETBSA-N 0.000 description 1
- KLYYKKGCPOGDPE-OEAJRASXSA-N Phe-Thr-Leu Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O KLYYKKGCPOGDPE-OEAJRASXSA-N 0.000 description 1
- OLZVAVSJEUAOHI-UNQGMJICSA-N Phe-Thr-Met Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCSC)C(=O)O)NC(=O)[C@H](CC1=CC=CC=C1)N)O OLZVAVSJEUAOHI-UNQGMJICSA-N 0.000 description 1
- JLDZQPPLTJTJLE-IHPCNDPISA-N Phe-Trp-Asp Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CC2=CNC3=CC=CC=C32)C(=O)N[C@@H](CC(=O)O)C(=O)O)N JLDZQPPLTJTJLE-IHPCNDPISA-N 0.000 description 1
- ZVJGAXNBBKPYOE-HKUYNNGSSA-N Phe-Trp-Gly Chemical compound C([C@H](N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)NCC(O)=O)C1=CC=CC=C1 ZVJGAXNBBKPYOE-HKUYNNGSSA-N 0.000 description 1
- QTDBZORPVYTRJU-KKXDTOCCSA-N Phe-Tyr-Ala Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](C)C(O)=O QTDBZORPVYTRJU-KKXDTOCCSA-N 0.000 description 1
- 241000235648 Pichia Species 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- KIZQGKLMXKGDIV-BQBZGAKWSA-N Pro-Ala-Gly Chemical compound OC(=O)CNC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1 KIZQGKLMXKGDIV-BQBZGAKWSA-N 0.000 description 1
- SSSFPISOZOLQNP-GUBZILKMSA-N Pro-Arg-Asp Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(O)=O SSSFPISOZOLQNP-GUBZILKMSA-N 0.000 description 1
- VPVHXWGPALPDGP-GUBZILKMSA-N Pro-Asn-Arg Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O VPVHXWGPALPDGP-GUBZILKMSA-N 0.000 description 1
- FISHYTLIMUYTQY-GUBZILKMSA-N Pro-Gln-Gln Chemical compound NC(=O)CC[C@@H](C(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H]1CCCN1 FISHYTLIMUYTQY-GUBZILKMSA-N 0.000 description 1
- ZTVCLZLGHZXLOT-ULQDDVLXSA-N Pro-Glu-Trp Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CC2=CNC3=CC=CC=C32)C(=O)O ZTVCLZLGHZXLOT-ULQDDVLXSA-N 0.000 description 1
- AFXCXDQNRXTSBD-FJXKBIBVSA-N Pro-Gly-Thr Chemical compound [H]N1CCC[C@H]1C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(O)=O AFXCXDQNRXTSBD-FJXKBIBVSA-N 0.000 description 1
- FKVNLUZHSFCNGY-RVMXOQNASA-N Pro-Ile-Pro Chemical compound CC[C@H](C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@@H]2CCCN2 FKVNLUZHSFCNGY-RVMXOQNASA-N 0.000 description 1
- VTFXTWDFPTWNJY-RHYQMDGZSA-N Pro-Leu-Thr Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O VTFXTWDFPTWNJY-RHYQMDGZSA-N 0.000 description 1
- SPLBRAKYXGOFSO-UNQGMJICSA-N Pro-Phe-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=CC=C1)NC(=O)[C@@H]2CCCN2)O SPLBRAKYXGOFSO-UNQGMJICSA-N 0.000 description 1
- KDBHVPXBQADZKY-GUBZILKMSA-N Pro-Pro-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H]1NCCC1 KDBHVPXBQADZKY-GUBZILKMSA-N 0.000 description 1
- SVXXJYJCRNKDDE-AVGNSLFASA-N Pro-Pro-His Chemical compound C([C@@H](C(=O)O)NC(=O)[C@H]1N(CCC1)C(=O)[C@H]1NCCC1)C1=CN=CN1 SVXXJYJCRNKDDE-AVGNSLFASA-N 0.000 description 1
- POQFNPILEQEODH-FXQIFTODSA-N Pro-Ser-Ala Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(O)=O POQFNPILEQEODH-FXQIFTODSA-N 0.000 description 1
- PKHDJFHFMGQMPS-RCWTZXSCSA-N Pro-Thr-Arg Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O PKHDJFHFMGQMPS-RCWTZXSCSA-N 0.000 description 1
- YDTUEBLEAVANFH-RCWTZXSCSA-N Pro-Val-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H]1CCCN1 YDTUEBLEAVANFH-RCWTZXSCSA-N 0.000 description 1
- HCBIBCJNVBAKAB-UHFFFAOYSA-N Procaine hydrochloride Chemical compound Cl.CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 HCBIBCJNVBAKAB-UHFFFAOYSA-N 0.000 description 1
- 108010076504 Protein Sorting Signals Proteins 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 102220492414 Ribulose-phosphate 3-epimerase_H35A_mutation Human genes 0.000 description 1
- 241000235070 Saccharomyces Species 0.000 description 1
- 101100007329 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) COS1 gene Proteins 0.000 description 1
- 229920002684 Sepharose Polymers 0.000 description 1
- LVVBAKCGXXUHFO-ZLUOBGJFSA-N Ser-Ala-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(O)=O)C(O)=O LVVBAKCGXXUHFO-ZLUOBGJFSA-N 0.000 description 1
- HRNQLKCLPVKZNE-CIUDSAMLSA-N Ser-Ala-Leu Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(O)=O HRNQLKCLPVKZNE-CIUDSAMLSA-N 0.000 description 1
- YQHZVYJAGWMHES-ZLUOBGJFSA-N Ser-Ala-Ser Chemical compound OC[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O YQHZVYJAGWMHES-ZLUOBGJFSA-N 0.000 description 1
- HBOABDXGTMMDSE-GUBZILKMSA-N Ser-Arg-Val Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(O)=O HBOABDXGTMMDSE-GUBZILKMSA-N 0.000 description 1
- VAUMZJHYZQXZBQ-WHFBIAKZSA-N Ser-Asn-Gly Chemical compound OC[C@H](N)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(O)=O VAUMZJHYZQXZBQ-WHFBIAKZSA-N 0.000 description 1
- RDFQNDHEHVSONI-ZLUOBGJFSA-N Ser-Asn-Ser Chemical compound OC[C@H](N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(O)=O RDFQNDHEHVSONI-ZLUOBGJFSA-N 0.000 description 1
- UGJRQLURDVGULT-LKXGYXEUSA-N Ser-Asn-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O UGJRQLURDVGULT-LKXGYXEUSA-N 0.000 description 1
- ICHZYBVODUVUKN-SRVKXCTJSA-N Ser-Asn-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O ICHZYBVODUVUKN-SRVKXCTJSA-N 0.000 description 1
- OHKLFYXEOGGGCK-ZLUOBGJFSA-N Ser-Asp-Asn Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O OHKLFYXEOGGGCK-ZLUOBGJFSA-N 0.000 description 1
- MMAPOBOTRUVNKJ-ZLUOBGJFSA-N Ser-Asp-Ser Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CO)N)C(=O)O MMAPOBOTRUVNKJ-ZLUOBGJFSA-N 0.000 description 1
- XWCYBVBLJRWOFR-WDSKDSINSA-N Ser-Gln-Gly Chemical compound OC[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)NCC(O)=O XWCYBVBLJRWOFR-WDSKDSINSA-N 0.000 description 1
- HVKMTOIAYDOJPL-NRPADANISA-N Ser-Gln-Val Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(O)=O HVKMTOIAYDOJPL-NRPADANISA-N 0.000 description 1
- SMIDBHKWSYUBRZ-ACZMJKKPSA-N Ser-Glu-Ala Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(O)=O SMIDBHKWSYUBRZ-ACZMJKKPSA-N 0.000 description 1
- HJEBZBMOTCQYDN-ACZMJKKPSA-N Ser-Glu-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(O)=O HJEBZBMOTCQYDN-ACZMJKKPSA-N 0.000 description 1
- DSGYZICNAMEJOC-AVGNSLFASA-N Ser-Glu-Phe Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O DSGYZICNAMEJOC-AVGNSLFASA-N 0.000 description 1
- BPMRXBZYPGYPJN-WHFBIAKZSA-N Ser-Gly-Asn Chemical compound [H]N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(N)=O)C(O)=O BPMRXBZYPGYPJN-WHFBIAKZSA-N 0.000 description 1
- MUARUIBTKQJKFY-WHFBIAKZSA-N Ser-Gly-Asp Chemical compound [H]N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(O)=O MUARUIBTKQJKFY-WHFBIAKZSA-N 0.000 description 1
- CAOYHZOWXFFAIR-CIUDSAMLSA-N Ser-His-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CO)C(O)=O CAOYHZOWXFFAIR-CIUDSAMLSA-N 0.000 description 1
- BKZYBLLIBOBOOW-GHCJXIJMSA-N Ser-Ile-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(O)=O)C(O)=O BKZYBLLIBOBOOW-GHCJXIJMSA-N 0.000 description 1
- JIPVNVNKXJLFJF-BJDJZHNGSA-N Ser-Ile-Lys Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CO)N JIPVNVNKXJLFJF-BJDJZHNGSA-N 0.000 description 1
- MQQBBLVOUUJKLH-HJPIBITLSA-N Ser-Ile-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O MQQBBLVOUUJKLH-HJPIBITLSA-N 0.000 description 1
- XNCUYZKGQOCOQH-YUMQZZPRSA-N Ser-Leu-Gly Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)NCC(O)=O XNCUYZKGQOCOQH-YUMQZZPRSA-N 0.000 description 1
- VMLONWHIORGALA-SRVKXCTJSA-N Ser-Leu-Leu Chemical compound CC(C)C[C@@H](C([O-])=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H]([NH3+])CO VMLONWHIORGALA-SRVKXCTJSA-N 0.000 description 1
- MUJQWSAWLLRJCE-KATARQTJSA-N Ser-Leu-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O MUJQWSAWLLRJCE-KATARQTJSA-N 0.000 description 1
- LRZLZIUXQBIWTB-KATARQTJSA-N Ser-Lys-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(O)=O LRZLZIUXQBIWTB-KATARQTJSA-N 0.000 description 1
- IFLVBVIYADZIQO-DCAQKATOSA-N Ser-Met-Lys Chemical compound CSCC[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CO)N IFLVBVIYADZIQO-DCAQKATOSA-N 0.000 description 1
- HEYZPTCCEIWHRO-IHRRRGAJSA-N Ser-Met-Phe Chemical compound OC[C@H](N)C(=O)N[C@@H](CCSC)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 HEYZPTCCEIWHRO-IHRRRGAJSA-N 0.000 description 1
- RXSWQCATLWVDLI-XGEHTFHBSA-N Ser-Met-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H]([C@@H](C)O)C(O)=O RXSWQCATLWVDLI-XGEHTFHBSA-N 0.000 description 1
- AXOHAHIUJHCLQR-IHRRRGAJSA-N Ser-Met-Tyr Chemical compound CSCC[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)NC(=O)[C@H](CO)N AXOHAHIUJHCLQR-IHRRRGAJSA-N 0.000 description 1
- TVPQRPNBYCRRLL-IHRRRGAJSA-N Ser-Phe-Met Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CCSC)C(O)=O TVPQRPNBYCRRLL-IHRRRGAJSA-N 0.000 description 1
- NUEHQDHDLDXCRU-GUBZILKMSA-N Ser-Pro-Arg Chemical compound OC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(O)=O NUEHQDHDLDXCRU-GUBZILKMSA-N 0.000 description 1
- KQNDIKOYWZTZIX-FXQIFTODSA-N Ser-Ser-Arg Chemical compound OC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCCNC(N)=N KQNDIKOYWZTZIX-FXQIFTODSA-N 0.000 description 1
- BMKNXTJLHFIAAH-CIUDSAMLSA-N Ser-Ser-Leu Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(O)=O BMKNXTJLHFIAAH-CIUDSAMLSA-N 0.000 description 1
- PYTKULIABVRXSC-BWBBJGPYSA-N Ser-Ser-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(O)=O PYTKULIABVRXSC-BWBBJGPYSA-N 0.000 description 1
- VGQVAVQWKJLIRM-FXQIFTODSA-N Ser-Ser-Val Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O VGQVAVQWKJLIRM-FXQIFTODSA-N 0.000 description 1
- WUXCHQZLUHBSDJ-LKXGYXEUSA-N Ser-Thr-Asp Chemical compound OC[C@H](N)C(=O)N[C@@H]([C@H](O)C)C(=O)N[C@@H](CC(O)=O)C(O)=O WUXCHQZLUHBSDJ-LKXGYXEUSA-N 0.000 description 1
- SZRNDHWMVSFPSP-XKBZYTNZSA-N Ser-Thr-Gln Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CO)N)O SZRNDHWMVSFPSP-XKBZYTNZSA-N 0.000 description 1
- FLMYSKVSDVHLEW-SVSWQMSJSA-N Ser-Thr-Ile Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O FLMYSKVSDVHLEW-SVSWQMSJSA-N 0.000 description 1
- BDMWLJLPPUCLNV-XGEHTFHBSA-N Ser-Thr-Val Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O BDMWLJLPPUCLNV-XGEHTFHBSA-N 0.000 description 1
- FVFUOQIYDPAIJR-XIRDDKMYSA-N Ser-Trp-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CC1=CNC2=CC=CC=C21)NC(=O)[C@H](CO)N FVFUOQIYDPAIJR-XIRDDKMYSA-N 0.000 description 1
- FGBLCMLXHRPVOF-IHRRRGAJSA-N Ser-Tyr-Arg Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O FGBLCMLXHRPVOF-IHRRRGAJSA-N 0.000 description 1
- KIEIJCFVGZCUAS-MELADBBJSA-N Ser-Tyr-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CC2=CC=C(C=C2)O)NC(=O)[C@H](CO)N)C(=O)O KIEIJCFVGZCUAS-MELADBBJSA-N 0.000 description 1
- OSFZCEQJLWCIBG-BZSNNMDCSA-N Ser-Tyr-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O OSFZCEQJLWCIBG-BZSNNMDCSA-N 0.000 description 1
- LLSLRQOEAFCZLW-NRPADANISA-N Ser-Val-Gln Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O LLSLRQOEAFCZLW-NRPADANISA-N 0.000 description 1
- MFQMZDPAZRZAPV-NAKRPEOUSA-N Ser-Val-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CO)N MFQMZDPAZRZAPV-NAKRPEOUSA-N 0.000 description 1
- LGIMRDKGABDMBN-DCAQKATOSA-N Ser-Val-Lys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CO)N LGIMRDKGABDMBN-DCAQKATOSA-N 0.000 description 1
- JGUWRQWULDWNCM-FXQIFTODSA-N Ser-Val-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O JGUWRQWULDWNCM-FXQIFTODSA-N 0.000 description 1
- SIEBDTCABMZCLF-XGEHTFHBSA-N Ser-Val-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O SIEBDTCABMZCLF-XGEHTFHBSA-N 0.000 description 1
- ODRUTDLAONAVDV-IHRRRGAJSA-N Ser-Val-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O ODRUTDLAONAVDV-IHRRRGAJSA-N 0.000 description 1
- 108010071390 Serum Albumin Proteins 0.000 description 1
- 102000007562 Serum Albumin Human genes 0.000 description 1
- 241000700584 Simplexvirus Species 0.000 description 1
- 102220497176 Small vasohibin-binding protein_T47D_mutation Human genes 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- 241000256251 Spodoptera frugiperda Species 0.000 description 1
- 108090000787 Subtilisin Proteins 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- TYVAWPFQYFPSBR-BFHQHQDPSA-N Thr-Ala-Gly Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)NCC(O)=O TYVAWPFQYFPSBR-BFHQHQDPSA-N 0.000 description 1
- ZUXQFMVPAYGPFJ-JXUBOQSCSA-N Thr-Ala-Lys Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@H](C(O)=O)CCCCN ZUXQFMVPAYGPFJ-JXUBOQSCSA-N 0.000 description 1
- LVHHEVGYAZGXDE-KDXUFGMBSA-N Thr-Ala-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](C)C(=O)N1CCC[C@@H]1C(=O)O)N)O LVHHEVGYAZGXDE-KDXUFGMBSA-N 0.000 description 1
- DWYAUVCQDTZIJI-VZFHVOOUSA-N Thr-Ala-Ser Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O DWYAUVCQDTZIJI-VZFHVOOUSA-N 0.000 description 1
- DGDCHPCRMWEOJR-FQPOAREZSA-N Thr-Ala-Tyr Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 DGDCHPCRMWEOJR-FQPOAREZSA-N 0.000 description 1
- XSLXHSYIVPGEER-KZVJFYERSA-N Thr-Ala-Val Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(O)=O XSLXHSYIVPGEER-KZVJFYERSA-N 0.000 description 1
- JHBHMCMKSPXRHV-NUMRIWBASA-N Thr-Asn-Gln Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CCC(=O)N)C(=O)O)N)O JHBHMCMKSPXRHV-NUMRIWBASA-N 0.000 description 1
- OJRNZRROAIAHDL-LKXGYXEUSA-N Thr-Asn-Ser Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(O)=O OJRNZRROAIAHDL-LKXGYXEUSA-N 0.000 description 1
- OYTNZCBFDXGQGE-XQXXSGGOSA-N Thr-Gln-Ala Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](C)C(=O)O)N)O OYTNZCBFDXGQGE-XQXXSGGOSA-N 0.000 description 1
- GCXFWAZRHBRYEM-NUMRIWBASA-N Thr-Gln-Asn Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CC(=O)N)C(=O)O)N)O GCXFWAZRHBRYEM-NUMRIWBASA-N 0.000 description 1
- VUVCRYXYUUPGSB-GLLZPBPUSA-N Thr-Gln-Glu Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CCC(=O)O)C(=O)O)N)O VUVCRYXYUUPGSB-GLLZPBPUSA-N 0.000 description 1
- DIPIPFHFLPTCLK-LOKLDPHHSA-N Thr-Gln-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N1CCC[C@@H]1C(=O)O)N)O DIPIPFHFLPTCLK-LOKLDPHHSA-N 0.000 description 1
- QQWNRERCGGZOKG-WEDXCCLWSA-N Thr-Gly-Leu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CC(C)C)C(O)=O QQWNRERCGGZOKG-WEDXCCLWSA-N 0.000 description 1
- JKGGPMOUIAAJAA-YEPSODPASA-N Thr-Gly-Val Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](C(C)C)C(O)=O JKGGPMOUIAAJAA-YEPSODPASA-N 0.000 description 1
- AYCQVUUPIJHJTA-IXOXFDKPSA-N Thr-His-Leu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(C)C)C(O)=O AYCQVUUPIJHJTA-IXOXFDKPSA-N 0.000 description 1
- PAXANSWUSVPFNK-IUKAMOBKSA-N Thr-Ile-Asn Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H]([C@@H](C)O)N PAXANSWUSVPFNK-IUKAMOBKSA-N 0.000 description 1
- CRZNCABIJLRFKZ-IUKAMOBKSA-N Thr-Ile-Asp Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)O)NC(=O)[C@H]([C@@H](C)O)N CRZNCABIJLRFKZ-IUKAMOBKSA-N 0.000 description 1
- ZBKDBZUTTXINIX-RWRJDSDZSA-N Thr-Ile-Gln Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(N)=O)C(O)=O ZBKDBZUTTXINIX-RWRJDSDZSA-N 0.000 description 1
- FQPDRTDDEZXCEC-SVSWQMSJSA-N Thr-Ile-Ser Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O FQPDRTDDEZXCEC-SVSWQMSJSA-N 0.000 description 1
- IHAPJUHCZXBPHR-WZLNRYEVSA-N Thr-Ile-Tyr Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)NC(=O)[C@H]([C@@H](C)O)N IHAPJUHCZXBPHR-WZLNRYEVSA-N 0.000 description 1
- RRRRCRYTLZVCEN-HJGDQZAQSA-N Thr-Leu-Asp Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O RRRRCRYTLZVCEN-HJGDQZAQSA-N 0.000 description 1
- HOVLHEKTGVIKAP-WDCWCFNPSA-N Thr-Leu-Gln Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O HOVLHEKTGVIKAP-WDCWCFNPSA-N 0.000 description 1
- JLNMFGCJODTXDH-WEDXCCLWSA-N Thr-Lys-Gly Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)NCC(O)=O JLNMFGCJODTXDH-WEDXCCLWSA-N 0.000 description 1
- JWQNAFHCXKVZKZ-UVOCVTCTSA-N Thr-Lys-Thr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(O)=O JWQNAFHCXKVZKZ-UVOCVTCTSA-N 0.000 description 1
- PCMDGXKXVMBIFP-VEVYYDQMSA-N Thr-Met-Asn Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(O)=O PCMDGXKXVMBIFP-VEVYYDQMSA-N 0.000 description 1
- FDQXPJCLVPFKJW-KJEVXHAQSA-N Thr-Met-Tyr Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)N)O FDQXPJCLVPFKJW-KJEVXHAQSA-N 0.000 description 1
- WTMPKZWHRCMMMT-KZVJFYERSA-N Thr-Pro-Ala Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(O)=O WTMPKZWHRCMMMT-KZVJFYERSA-N 0.000 description 1
- FWTFAZKJORVTIR-VZFHVOOUSA-N Thr-Ser-Ala Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(O)=O FWTFAZKJORVTIR-VZFHVOOUSA-N 0.000 description 1
- STUAPCLEDMKXKL-LKXGYXEUSA-N Thr-Ser-Asn Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(O)=O STUAPCLEDMKXKL-LKXGYXEUSA-N 0.000 description 1
- IQPWNQRRAJHOKV-KATARQTJSA-N Thr-Ser-Lys Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCCCN IQPWNQRRAJHOKV-KATARQTJSA-N 0.000 description 1
- HUPLKEHTTQBXSC-YJRXYDGGSA-N Thr-Ser-Tyr Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 HUPLKEHTTQBXSC-YJRXYDGGSA-N 0.000 description 1
- YRJOLUDFVAUXLI-GSSVUCPTSA-N Thr-Thr-Asp Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@H](C(O)=O)CC(O)=O YRJOLUDFVAUXLI-GSSVUCPTSA-N 0.000 description 1
- UQCNIMDPYICBTR-KYNKHSRBSA-N Thr-Thr-Gly Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(O)=O UQCNIMDPYICBTR-KYNKHSRBSA-N 0.000 description 1
- XEVHXNLPUBVQEX-DVJZZOLTSA-N Thr-Trp-Gly Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)NCC(=O)O)N)O XEVHXNLPUBVQEX-DVJZZOLTSA-N 0.000 description 1
- BKVICMPZWRNWOC-RHYQMDGZSA-N Thr-Val-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)[C@@H](C)O BKVICMPZWRNWOC-RHYQMDGZSA-N 0.000 description 1
- 108090000190 Thrombin Proteins 0.000 description 1
- 102000006601 Thymidine Kinase Human genes 0.000 description 1
- 108020004440 Thymidine kinase Proteins 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- 108700019146 Transgenes Proteins 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- 101000980463 Treponema pallidum (strain Nichols) Chaperonin GroEL Proteins 0.000 description 1
- YZCKVEUIGOORGS-NJFSPNSNSA-N Tritium Chemical compound [3H] YZCKVEUIGOORGS-NJFSPNSNSA-N 0.000 description 1
- MHNHRNHJMXAVHZ-AAEUAGOBSA-N Trp-Asn-Gly Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)NCC(=O)O)N MHNHRNHJMXAVHZ-AAEUAGOBSA-N 0.000 description 1
- YRXXUYPYPHRJPB-RXVVDRJESA-N Trp-Gly-Trp Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)NCC(=O)N[C@@H](CC3=CNC4=CC=CC=C43)C(=O)O)N YRXXUYPYPHRJPB-RXVVDRJESA-N 0.000 description 1
- OFTGYORHQMSPAI-PJODQICGSA-N Trp-Met-Ala Chemical compound [H]N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](C)C(O)=O OFTGYORHQMSPAI-PJODQICGSA-N 0.000 description 1
- PKZIWSHDJYIPRH-JBACZVJFSA-N Trp-Tyr-Gln Chemical compound [H]N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCC(N)=O)C(O)=O PKZIWSHDJYIPRH-JBACZVJFSA-N 0.000 description 1
- XQMGDVVKFRLQKH-BBRMVZONSA-N Trp-Val-Gly Chemical compound C1=CC=C2C(C[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)NCC(O)=O)=CNC2=C1 XQMGDVVKFRLQKH-BBRMVZONSA-N 0.000 description 1
- IEESWNWYUOETOT-BVSLBCMMSA-N Trp-Val-Phe Chemical compound CC(C)[C@H](NC(=O)[C@@H](N)Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(O)=O IEESWNWYUOETOT-BVSLBCMMSA-N 0.000 description 1
- BURPTJBFWIOHEY-UWJYBYFXSA-N Tyr-Ala-Asp Chemical compound OC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 BURPTJBFWIOHEY-UWJYBYFXSA-N 0.000 description 1
- IELISNUVHBKYBX-XDTLVQLUSA-N Tyr-Ala-Glu Chemical compound OC(=O)CC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 IELISNUVHBKYBX-XDTLVQLUSA-N 0.000 description 1
- SDNVRAKIJVKAGS-LKTVYLICSA-N Tyr-Ala-His Chemical compound C[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)NC(=O)[C@H](CC2=CC=C(C=C2)O)N SDNVRAKIJVKAGS-LKTVYLICSA-N 0.000 description 1
- FBVGQXJIXFZKSQ-GMVOTWDCSA-N Tyr-Ala-Trp Chemical compound C[C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)O)NC(=O)[C@H](CC3=CC=C(C=C3)O)N FBVGQXJIXFZKSQ-GMVOTWDCSA-N 0.000 description 1
- IIJWXEUNETVJPV-IHRRRGAJSA-N Tyr-Arg-Ser Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CO)C(=O)O)N)O IIJWXEUNETVJPV-IHRRRGAJSA-N 0.000 description 1
- XMNDQSYABVWZRK-BZSNNMDCSA-N Tyr-Asn-Phe Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O XMNDQSYABVWZRK-BZSNNMDCSA-N 0.000 description 1
- NSTPFWRAIDTNGH-BZSNNMDCSA-N Tyr-Asn-Tyr Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O NSTPFWRAIDTNGH-BZSNNMDCSA-N 0.000 description 1
- QOIKZODVIPOPDD-AVGNSLFASA-N Tyr-Cys-Gln Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(O)=O QOIKZODVIPOPDD-AVGNSLFASA-N 0.000 description 1
- QUILOGWWLXMSAT-IHRRRGAJSA-N Tyr-Gln-Gln Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O QUILOGWWLXMSAT-IHRRRGAJSA-N 0.000 description 1
- GGXUDPQWAWRINY-XEGUGMAKSA-N Tyr-Ile-Gly Chemical compound OC(=O)CNC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 GGXUDPQWAWRINY-XEGUGMAKSA-N 0.000 description 1
- OHOVFPKXPZODHS-SJWGOKEGSA-N Tyr-Ile-Pro Chemical compound CC[C@H](C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CC2=CC=C(C=C2)O)N OHOVFPKXPZODHS-SJWGOKEGSA-N 0.000 description 1
- OLYXUGBVBGSZDN-ACRUOGEOSA-N Tyr-Leu-Tyr Chemical compound C([C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(O)=O)C1=CC=C(O)C=C1 OLYXUGBVBGSZDN-ACRUOGEOSA-N 0.000 description 1
- PGEFRHBWGOJPJT-KKUMJFAQSA-N Tyr-Lys-Ser Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(O)=O PGEFRHBWGOJPJT-KKUMJFAQSA-N 0.000 description 1
- UBKKNELWDCBNCF-STQMWFEESA-N Tyr-Met-Gly Chemical compound OC(=O)CNC(=O)[C@H](CCSC)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 UBKKNELWDCBNCF-STQMWFEESA-N 0.000 description 1
- VYQQQIRHIFALGE-UWJYBYFXSA-N Tyr-Ser-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 VYQQQIRHIFALGE-UWJYBYFXSA-N 0.000 description 1
- SYFHQHYTNCQCCN-MELADBBJSA-N Tyr-Ser-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CO)NC(=O)[C@H](CC2=CC=C(C=C2)O)N)C(=O)O SYFHQHYTNCQCCN-MELADBBJSA-N 0.000 description 1
- WYOBRXPIZVKNMF-IRXDYDNUSA-N Tyr-Tyr-Gly Chemical compound C([C@H](N)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)NCC(O)=O)C1=CC=C(O)C=C1 WYOBRXPIZVKNMF-IRXDYDNUSA-N 0.000 description 1
- FZADUTOCSFDBRV-RNXOBYDBSA-N Tyr-Tyr-Trp Chemical compound C([C@H](N)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(O)=O)C1=CC=C(O)C=C1 FZADUTOCSFDBRV-RNXOBYDBSA-N 0.000 description 1
- AEOFMCAKYIQQFY-YDHLFZDLSA-N Tyr-Val-Asn Chemical compound NC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 AEOFMCAKYIQQFY-YDHLFZDLSA-N 0.000 description 1
- 241000700618 Vaccinia virus Species 0.000 description 1
- IZFVRRYRMQFVGX-NRPADANISA-N Val-Ala-Gln Chemical compound C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](C(C)C)N IZFVRRYRMQFVGX-NRPADANISA-N 0.000 description 1
- ASQFIHTXXMFENG-XPUUQOCRSA-N Val-Ala-Gly Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](C)C(=O)NCC(O)=O ASQFIHTXXMFENG-XPUUQOCRSA-N 0.000 description 1
- AZSHAZJLOZQYAY-FXQIFTODSA-N Val-Ala-Ser Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O AZSHAZJLOZQYAY-FXQIFTODSA-N 0.000 description 1
- COYSIHFOCOMGCF-WPRPVWTQSA-N Val-Arg-Gly Chemical compound CC(C)[C@H](N)C(=O)N[C@H](C(=O)NCC(O)=O)CCCN=C(N)N COYSIHFOCOMGCF-WPRPVWTQSA-N 0.000 description 1
- COYSIHFOCOMGCF-UHFFFAOYSA-N Val-Arg-Gly Natural products CC(C)C(N)C(=O)NC(C(=O)NCC(O)=O)CCCN=C(N)N COYSIHFOCOMGCF-UHFFFAOYSA-N 0.000 description 1
- CVUDMNSZAIZFAE-TUAOUCFPSA-N Val-Arg-Pro Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1CCC[C@@H]1C(=O)O)N CVUDMNSZAIZFAE-TUAOUCFPSA-N 0.000 description 1
- OQWNEUXPKHIEJO-NRPADANISA-N Val-Glu-Ser Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CO)C(=O)O)N OQWNEUXPKHIEJO-NRPADANISA-N 0.000 description 1
- ZIGZPYJXIWLQFC-QTKMDUPCSA-N Val-His-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CN=CN1)NC(=O)[C@H](C(C)C)N)O ZIGZPYJXIWLQFC-QTKMDUPCSA-N 0.000 description 1
- BZMIYHIJVVJPCK-QSFUFRPTSA-N Val-Ile-Asn Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](C(C)C)N BZMIYHIJVVJPCK-QSFUFRPTSA-N 0.000 description 1
- BZWUSZGQOILYEU-STECZYCISA-N Val-Ile-Tyr Chemical compound CC(C)[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 BZWUSZGQOILYEU-STECZYCISA-N 0.000 description 1
- RWOGENDAOGMHLX-DCAQKATOSA-N Val-Lys-Ala Chemical compound C[C@@H](C(=O)O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C(C)C)N RWOGENDAOGMHLX-DCAQKATOSA-N 0.000 description 1
- SJRUJQFQVLMZFW-WPRPVWTQSA-N Val-Pro-Gly Chemical compound CC(C)[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O SJRUJQFQVLMZFW-WPRPVWTQSA-N 0.000 description 1
- RYHUIHUOYRNNIE-NRPADANISA-N Val-Ser-Gln Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(=O)N)C(=O)O)N RYHUIHUOYRNNIE-NRPADANISA-N 0.000 description 1
- GBIUHAYJGWVNLN-AEJSXWLSSA-N Val-Ser-Pro Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)N1CCC[C@@H]1C(=O)O)N GBIUHAYJGWVNLN-AEJSXWLSSA-N 0.000 description 1
- HWNYVQMOLCYHEA-IHRRRGAJSA-N Val-Ser-Tyr Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)N HWNYVQMOLCYHEA-IHRRRGAJSA-N 0.000 description 1
- NZYNRRGJJVSSTJ-GUBZILKMSA-N Val-Ser-Val Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O NZYNRRGJJVSSTJ-GUBZILKMSA-N 0.000 description 1
- PDDJTOSAVNRJRH-UNQGMJICSA-N Val-Thr-Phe Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)O)NC(=O)[C@H](C(C)C)N)O PDDJTOSAVNRJRH-UNQGMJICSA-N 0.000 description 1
- YLBNZCJFSVJDRJ-KJEVXHAQSA-N Val-Thr-Tyr Chemical compound CC(C)[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](Cc1ccc(O)cc1)C(O)=O YLBNZCJFSVJDRJ-KJEVXHAQSA-N 0.000 description 1
- QPJSIBAOZBVELU-BPNCWPANSA-N Val-Tyr-Ala Chemical compound C[C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)[C@H](C(C)C)N QPJSIBAOZBVELU-BPNCWPANSA-N 0.000 description 1
- YKZVPMUGEJXEOR-JYJNAYRXSA-N Val-Val-Tyr Chemical compound CC(C)[C@@H](C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)N YKZVPMUGEJXEOR-JYJNAYRXSA-N 0.000 description 1
- 238000012452 Xenomouse strains Methods 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- ATBOMIWRCZXYSZ-XZBBILGWSA-N [1-[2,3-dihydroxypropoxy(hydroxy)phosphoryl]oxy-3-hexadecanoyloxypropan-2-yl] (9e,12e)-octadeca-9,12-dienoate Chemical compound CCCCCCCCCCCCCCCC(=O)OCC(COP(O)(=O)OCC(O)CO)OC(=O)CCCCCCC\C=C\C\C=C\CCCCC ATBOMIWRCZXYSZ-XZBBILGWSA-N 0.000 description 1
- 230000001133 acceleration Effects 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 230000003044 adaptive effect Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 238000001042 affinity chromatography Methods 0.000 description 1
- 108010045023 alanyl-prolyl-tyrosine Proteins 0.000 description 1
- 108010070944 alanylhistidine Proteins 0.000 description 1
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 229940126575 aminoglycoside Drugs 0.000 description 1
- 239000012491 analyte Substances 0.000 description 1
- 238000000137 annealing Methods 0.000 description 1
- 230000003042 antagnostic effect Effects 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 210000000628 antibody-producing cell Anatomy 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 108010043240 arginyl-leucyl-glycine Proteins 0.000 description 1
- 108010059459 arginyl-threonyl-phenylalanine Proteins 0.000 description 1
- 108010060035 arginylproline Proteins 0.000 description 1
- 108010036533 arginylvaline Proteins 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 108010040443 aspartyl-aspartic acid Proteins 0.000 description 1
- 108010069205 aspartyl-phenylalanine Proteins 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- IQFYYKKMVGJFEH-UHFFFAOYSA-N beta-L-thymidine Natural products O=C1NC(=O)C(C)=CN1C1OC(CO)C(O)C1 IQFYYKKMVGJFEH-UHFFFAOYSA-N 0.000 description 1
- 229920000080 bile acid sequestrant Polymers 0.000 description 1
- 229940096699 bile acid sequestrants Drugs 0.000 description 1
- 238000001815 biotherapy Methods 0.000 description 1
- HUTDDBSSHVOYJR-UHFFFAOYSA-H bis[(2-oxo-1,3,2$l^{5},4$l^{2}-dioxaphosphaplumbetan-2-yl)oxy]lead Chemical compound [Pb+2].[Pb+2].[Pb+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O HUTDDBSSHVOYJR-UHFFFAOYSA-H 0.000 description 1
- 210000002459 blastocyst Anatomy 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- LRHPLDYGYMQRHN-UHFFFAOYSA-N butyl alcohol Substances CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 210000004899 c-terminal region Anatomy 0.000 description 1
- BMLSTPRTEKLIPM-UHFFFAOYSA-I calcium;potassium;disodium;hydrogen carbonate;dichloride;dihydroxide;hydrate Chemical compound O.[OH-].[OH-].[Na+].[Na+].[Cl-].[Cl-].[K+].[Ca+2].OC([O-])=O BMLSTPRTEKLIPM-UHFFFAOYSA-I 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 230000007211 cardiovascular event Effects 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 239000013553 cell monolayer Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- TZRFSLHOCZEXCC-HIVFKXHNSA-N chembl2219536 Chemical compound N1([C@H]2C[C@@H]([C@H](O2)COP(O)(=O)S[C@@H]2[C@H](O[C@H](C2)N2C3=C(C(NC(N)=N3)=O)N=C2)COP(O)(=O)S[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)S[C@@H]2[C@H](O[C@H](C2)N2C(N=C(N)C(C)=C2)=O)COP(O)(=O)S[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)S[C@@H]2[C@H](O[C@H](C2)N2C3=C(C(NC(N)=N3)=O)N=C2)COP(O)(=O)S[C@@H]2[C@H](O[C@H](C2)N2C3=NC=NC(N)=C3N=C2)COP(O)(=O)S[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=O)S[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=O)S[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=O)S[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=O)S[C@H]2[C@H]([C@@H](O[C@@H]2CO)N2C3=C(C(NC(N)=N3)=O)N=C2)OCCOC)N2C(N=C(N)C(C)=C2)=O)OCCOC)N2C(N=C(N)C(C)=C2)=O)OCCOC)N2C(NC(=O)C(C)=C2)=O)OCCOC)N2C(N=C(N)C(C)=C2)=O)OCCOC)SP(O)(=O)OC[C@H]2O[C@H](C[C@@H]2SP(O)(=O)OC[C@H]2O[C@H](C[C@@H]2SP(O)(=O)OC[C@H]2O[C@H](C[C@@H]2SP(O)(=O)OC[C@@H]2[C@H]([C@H]([C@@H](O2)N2C3=C(C(NC(N)=N3)=O)N=C2)OCCOC)SP(O)(=O)OC[C@H]2[C@@H]([C@@H]([C@H](O2)N2C(N=C(N)C(C)=C2)=O)OCCOC)SP(O)(=O)OC[C@H]2[C@@H]([C@@H]([C@H](O2)N2C3=NC=NC(N)=C3N=C2)OCCOC)SP(O)(=O)OC[C@H]2[C@@H]([C@@H]([C@H](O2)N2C(N=C(N)C(C)=C2)=O)OCCOC)SP(O)(=O)OC[C@H]2[C@H](O)[C@@H]([C@H](O2)N2C(N=C(N)C(C)=C2)=O)OCCOC)N2C(N=C(N)C(C)=C2)=O)N2C(NC(=O)C(C)=C2)=O)N2C(NC(=O)C(C)=C2)=O)C=C(C)C(N)=NC1=O TZRFSLHOCZEXCC-HIVFKXHNSA-N 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- 235000012000 cholesterol Nutrition 0.000 description 1
- 230000001906 cholesterol absorption Effects 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 210000000349 chromosome Anatomy 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 239000013599 cloning vector Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004737 colorimetric analysis Methods 0.000 description 1
- 238000007398 colorimetric assay Methods 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 230000009137 competitive binding Effects 0.000 description 1
- 230000006957 competitive inhibition Effects 0.000 description 1
- 230000001268 conjugating effect Effects 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- 208000029078 coronary artery disease Diseases 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 229930003836 cresol Natural products 0.000 description 1
- 238000012866 crystallographic experiment Methods 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 238000013016 damping Methods 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 239000008355 dextrose injection Substances 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- 238000002050 diffraction method Methods 0.000 description 1
- 239000013024 dilution buffer Substances 0.000 description 1
- 150000002016 disaccharides Chemical class 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000005553 drilling Methods 0.000 description 1
- 230000002526 effect on cardiovascular system Effects 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 230000005611 electricity Effects 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 239000008393 encapsulating agent Substances 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 210000003527 eukaryotic cell Anatomy 0.000 description 1
- 235000013861 fat-free Nutrition 0.000 description 1
- 150000004665 fatty acids Chemical group 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000001506 fluorescence spectroscopy Methods 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 239000012737 fresh medium Substances 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- 238000002825 functional assay Methods 0.000 description 1
- 230000005021 gait Effects 0.000 description 1
- 238000001502 gel electrophoresis Methods 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 238000012252 genetic analysis Methods 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 210000004602 germ cell Anatomy 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 229940116332 glucose oxidase Drugs 0.000 description 1
- 235000019420 glucose oxidase Nutrition 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 108010090037 glycyl-alanyl-isoleucine Proteins 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 102000057403 human APOC3 Human genes 0.000 description 1
- 210000005104 human peripheral blood lymphocyte Anatomy 0.000 description 1
- 229920001477 hydrophilic polymer Polymers 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 238000012872 hydroxylapatite chromatography Methods 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 230000000055 hyoplipidemic effect Effects 0.000 description 1
- 238000010191 image analysis Methods 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 230000000984 immunochemical effect Effects 0.000 description 1
- 238000003364 immunohistochemistry Methods 0.000 description 1
- 230000003308 immunostimulating effect Effects 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 208000027866 inflammatory disease Diseases 0.000 description 1
- 238000002664 inhalation therapy Methods 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 101150066555 lacZ gene Proteins 0.000 description 1
- 230000006651 lactation Effects 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 108010083708 leucyl-aspartyl-valine Proteins 0.000 description 1
- 108010044311 leucyl-glycyl-glycine Proteins 0.000 description 1
- 108010057821 leucylproline Proteins 0.000 description 1
- 108010012058 leucyltyrosine Proteins 0.000 description 1
- 239000006193 liquid solution Substances 0.000 description 1
- 239000006194 liquid suspension Substances 0.000 description 1
- 229960005015 local anesthetics Drugs 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- HWYHZTIRURJOHG-UHFFFAOYSA-N luminol Chemical compound O=C1NNC(=O)C2=C1C(N)=CC=C2 HWYHZTIRURJOHG-UHFFFAOYSA-N 0.000 description 1
- 210000001165 lymph node Anatomy 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 238000000691 measurement method Methods 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- 210000004379 membrane Anatomy 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Chemical class 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- HPNSFSBZBAHARI-UHFFFAOYSA-N micophenolic acid Natural products OC1=C(CC=C(C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-UHFFFAOYSA-N 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 238000000386 microscopy Methods 0.000 description 1
- 229960004778 mipomersen Drugs 0.000 description 1
- 108091060283 mipomersen Proteins 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 229960001521 motavizumab Drugs 0.000 description 1
- 210000004877 mucosa Anatomy 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- HPNSFSBZBAHARI-RUDMXATFSA-N mycophenolic acid Chemical compound OC1=C(C\C=C(/C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-RUDMXATFSA-N 0.000 description 1
- 229960000951 mycophenolic acid Drugs 0.000 description 1
- 239000006199 nebulizer Substances 0.000 description 1
- 229960004927 neomycin Drugs 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000002687 nonaqueous vehicle Substances 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 239000000346 nonvolatile oil Substances 0.000 description 1
- 238000007899 nucleic acid hybridization Methods 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 229940012843 omega-3 fatty acid Drugs 0.000 description 1
- 235000020660 omega-3 fatty acid Nutrition 0.000 description 1
- 239000006014 omega-3 oil Substances 0.000 description 1
- 238000001543 one-way ANOVA Methods 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- LXCFILQKKLGQFO-UHFFFAOYSA-N p-hydroxybenzoic acid methyl ester Natural products COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 1
- 238000010979 pH adjustment Methods 0.000 description 1
- 239000006179 pH buffering agent Substances 0.000 description 1
- 238000004091 panning Methods 0.000 description 1
- 229940055729 papain Drugs 0.000 description 1
- 235000019834 papain Nutrition 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- 229940111202 pepsin Drugs 0.000 description 1
- 238000010647 peptide synthesis reaction Methods 0.000 description 1
- 210000005105 peripheral blood lymphocyte Anatomy 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 229960003742 phenol Drugs 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 108010084572 phenylalanyl-valine Proteins 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 125000001095 phosphatidyl group Chemical group 0.000 description 1
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 1
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 1
- 150000003905 phosphatidylinositols Chemical class 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 229940068065 phytosterols Drugs 0.000 description 1
- 108010025488 pinealon Proteins 0.000 description 1
- 229920001983 poloxamer Polymers 0.000 description 1
- 230000008488 polyadenylation Effects 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 230000001323 posttranslational effect Effects 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 235000008476 powdered milk Nutrition 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 229960001309 procaine hydrochloride Drugs 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 108010093296 prolyl-prolyl-alanine Proteins 0.000 description 1
- 108010031719 prolyl-serine Proteins 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 235000019419 proteases Nutrition 0.000 description 1
- 230000009145 protein modification Effects 0.000 description 1
- 230000006337 proteolytic cleavage Effects 0.000 description 1
- 238000003908 quality control method Methods 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 238000004445 quantitative analysis Methods 0.000 description 1
- 239000000700 radioactive tracer Substances 0.000 description 1
- 239000008175 ready-to-use sterile solution Substances 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 1
- 238000007423 screening assay Methods 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 239000006152 selective media Substances 0.000 description 1
- 239000003352 sequestering agent Substances 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 229960004249 sodium acetate Drugs 0.000 description 1
- 239000007974 sodium acetate buffer Substances 0.000 description 1
- WBHQBSYUUJJSRZ-UHFFFAOYSA-M sodium bisulfate Chemical compound [Na+].OS([O-])(=O)=O WBHQBSYUUJJSRZ-UHFFFAOYSA-M 0.000 description 1
- 229910000342 sodium bisulfate Inorganic materials 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000008354 sodium chloride injection Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 239000008137 solubility enhancer Substances 0.000 description 1
- 230000037439 somatic mutation Effects 0.000 description 1
- 229940035044 sorbitan monolaurate Drugs 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 210000004989 spleen cell Anatomy 0.000 description 1
- 210000004988 splenocyte Anatomy 0.000 description 1
- 235000000891 standard diet Nutrition 0.000 description 1
- SFVFIFLLYFPGHH-UHFFFAOYSA-M stearalkonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)CC1=CC=CC=C1 SFVFIFLLYFPGHH-UHFFFAOYSA-M 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 229950009108 succinobucol Drugs 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 125000002653 sulfanylmethyl group Chemical group [H]SC([H])([H])[*] 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 238000007910 systemic administration Methods 0.000 description 1
- 229910052713 technetium Inorganic materials 0.000 description 1
- GKLVYJBZJHMRIY-UHFFFAOYSA-N technetium atom Chemical compound [Tc] GKLVYJBZJHMRIY-UHFFFAOYSA-N 0.000 description 1
- MPLHNVLQVRSVEE-UHFFFAOYSA-N texas red Chemical compound [O-]S(=O)(=O)C1=CC(S(Cl)(=O)=O)=CC=C1C(C1=CC=2CCCN3CCCC(C=23)=C1O1)=C2C1=C(CCC1)C3=[N+]1CCCC3=C2 MPLHNVLQVRSVEE-UHFFFAOYSA-N 0.000 description 1
- 238000011287 therapeutic dose Methods 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 229960004072 thrombin Drugs 0.000 description 1
- 229940104230 thymidine Drugs 0.000 description 1
- 229940098465 tincture Drugs 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 230000037317 transdermal delivery Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 229940117013 triethanolamine oleate Drugs 0.000 description 1
- 125000005457 triglyceride group Chemical group 0.000 description 1
- 229910052722 tritium Inorganic materials 0.000 description 1
- 108010044292 tryptophyltyrosine Proteins 0.000 description 1
- 108010005834 tyrosyl-alanyl-glycine Proteins 0.000 description 1
- 241000701447 unidentified baculovirus Species 0.000 description 1
- 241001515965 unidentified phage Species 0.000 description 1
- 238000011870 unpaired t-test Methods 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 238000011179 visual inspection Methods 0.000 description 1
- 239000011534 wash buffer Substances 0.000 description 1
- 239000008215 water for injection Substances 0.000 description 1
- 239000008136 water-miscible vehicle Substances 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/3955—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against proteinaceous materials, e.g. enzymes, hormones, lymphokines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/397—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having four-membered rings, e.g. azetidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/775—Apolipopeptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/22—Immunoglobulins specific features characterized by taxonomic origin from camelids, e.g. camel, llama or dromedary
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/34—Identification of a linear epitope shorter than 20 amino acid residues or of a conformational epitope defined by amino acid residues
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/51—Complete heavy chain or Fd fragment, i.e. VH + CH1
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Genetics & Genomics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Biochemistry (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Molecular Biology (AREA)
- Immunology (AREA)
- Biophysics (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Epidemiology (AREA)
- Microbiology (AREA)
- General Engineering & Computer Science (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Vascular Medicine (AREA)
- Urology & Nephrology (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Mycology (AREA)
- Endocrinology (AREA)
- Cell Biology (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201662360084P | 2016-07-08 | 2016-07-08 | |
| US62/360,084 | 2016-07-08 | ||
| US201762491591P | 2017-04-28 | 2017-04-28 | |
| US62/491,591 | 2017-04-28 | ||
| PCT/IB2017/054125 WO2018007999A1 (en) | 2016-07-08 | 2017-07-07 | Anti-apoc3 antibodies and methods of use thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20190039937A true KR20190039937A (ko) | 2019-04-16 |
Family
ID=59416751
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020197002789A Ceased KR20190039937A (ko) | 2016-07-08 | 2017-07-07 | 항-ApoC3 항체 및 이의 사용 방법 |
Country Status (9)
| Country | Link |
|---|---|
| US (3) | US11091539B2 (OSRAM) |
| EP (1) | EP3481864A1 (OSRAM) |
| JP (2) | JP2019525772A (OSRAM) |
| KR (1) | KR20190039937A (OSRAM) |
| CN (1) | CN109689685A (OSRAM) |
| AU (1) | AU2017292184A1 (OSRAM) |
| CA (1) | CA3030099A1 (OSRAM) |
| MA (1) | MA45602A (OSRAM) |
| WO (1) | WO2018007999A1 (OSRAM) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MA45602A (fr) | 2016-07-08 | 2019-05-15 | Staten Biotechnology B V | Anticorps anti-apoc3 et leurs méthodes d'utilisation |
| CN110506056A (zh) * | 2017-04-21 | 2019-11-26 | 斯塔滕生物技术有限公司 | 抗apoc3抗体和其使用方法 |
| MX2020004512A (es) | 2017-10-31 | 2020-08-13 | Anticuerpos anti apolipoproteina c-iii (anti-apoc3) y metodos de uso de los mismos. | |
| US10538583B2 (en) | 2017-10-31 | 2020-01-21 | Staten Biotechnology B.V. | Anti-APOC3 antibodies and compositions thereof |
| JP2021532167A (ja) | 2018-06-05 | 2021-11-25 | アンジ ファーマ(ユーエス)エルエルシー | 膵炎を治療するための組成物及び方法 |
| CN112969503A (zh) * | 2018-10-03 | 2021-06-15 | 斯塔滕生物技术有限公司 | 对人类和食蟹猕猴apoc3具有特异性的抗体和其使用方法 |
| CN115819556A (zh) * | 2022-11-29 | 2023-03-21 | 迪亚莱博(张家港)生物科技有限公司 | 一种apoc3重组蛋白及其制备方法 |
Family Cites Families (166)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1429184A (en) | 1972-04-20 | 1976-03-24 | Allen & Hanburys Ltd | Physically anti-inflammatory steroids for use in aerosols |
| US4044126A (en) | 1972-04-20 | 1977-08-23 | Allen & Hanburys Limited | Steroidal aerosol compositions and process for the preparation thereof |
| US4444887A (en) | 1979-12-10 | 1984-04-24 | Sloan-Kettering Institute | Process for making human antibody producing B-lymphocytes |
| US4716111A (en) | 1982-08-11 | 1987-12-29 | Trustees Of Boston University | Process for producing human antibodies |
| GB8308235D0 (en) | 1983-03-25 | 1983-05-05 | Celltech Ltd | Polypeptides |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US5807715A (en) | 1984-08-27 | 1998-09-15 | The Board Of Trustees Of The Leland Stanford Junior University | Methods and transformed mammalian lymphocyte cells for producing functional antigen-binding protein including chimeric immunoglobulin |
| US4970144A (en) | 1984-12-31 | 1990-11-13 | International Genetic Engineering | Peptide fragments of human apolipoprotein, type-specific antibodies and methods of use |
| DE3668186D1 (de) | 1985-04-01 | 1990-02-15 | Celltech Ltd | Transformierte myeloma-zell-linie und dieselbe verwendendes verfahren zur expression eines gens, das ein eukaryontisches polypeptid kodiert. |
| GB8601597D0 (en) | 1986-01-23 | 1986-02-26 | Wilson R H | Nucleotide sequences |
| GB8607679D0 (en) | 1986-03-27 | 1986-04-30 | Winter G P | Recombinant dna product |
| US5225539A (en) | 1986-03-27 | 1993-07-06 | Medical Research Council | Recombinant altered antibodies and methods of making altered antibodies |
| US5869620A (en) | 1986-09-02 | 1999-02-09 | Enzon, Inc. | Multivalent antigen-binding proteins |
| GB8717430D0 (en) | 1987-07-23 | 1987-08-26 | Celltech Ltd | Recombinant dna product |
| EP0436597B1 (en) | 1988-09-02 | 1997-04-02 | Protein Engineering Corporation | Generation and selection of recombinant varied binding proteins |
| US5223409A (en) | 1988-09-02 | 1993-06-29 | Protein Engineering Corp. | Directed evolution of novel binding proteins |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5413923A (en) | 1989-07-25 | 1995-05-09 | Cell Genesys, Inc. | Homologous recombination for universal donor cells and chimeric mammalian hosts |
| GB8928874D0 (en) | 1989-12-21 | 1990-02-28 | Celltech Ltd | Humanised antibodies |
| US5585112A (en) | 1989-12-22 | 1996-12-17 | Imarx Pharmaceutical Corp. | Method of preparing gas and gaseous precursor-filled microspheres |
| AU7247191A (en) | 1990-01-11 | 1991-08-05 | Molecular Affinities Corporation | Production of antibodies using gene libraries |
| US5780225A (en) | 1990-01-12 | 1998-07-14 | Stratagene | Method for generating libaries of antibody genes comprising amplification of diverse antibody DNAs and methods for using these libraries for the production of diverse antigen combining molecules |
| EP1690935A3 (en) | 1990-01-12 | 2008-07-30 | Abgenix, Inc. | Generation of xenogeneic antibodies |
| US6075181A (en) | 1990-01-12 | 2000-06-13 | Abgenix, Inc. | Human antibodies derived from immunized xenomice |
| IT1246382B (it) | 1990-04-17 | 1994-11-18 | Eurand Int | Metodo per la cessione mirata e controllata di farmaci nell'intestino e particolarmente nel colon |
| US5427908A (en) | 1990-05-01 | 1995-06-27 | Affymax Technologies N.V. | Recombinant library screening methods |
| GB9015198D0 (en) | 1990-07-10 | 1990-08-29 | Brien Caroline J O | Binding substance |
| US5625126A (en) | 1990-08-29 | 1997-04-29 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5661016A (en) | 1990-08-29 | 1997-08-26 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5633425A (en) | 1990-08-29 | 1997-05-27 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5545806A (en) | 1990-08-29 | 1996-08-13 | Genpharm International, Inc. | Ransgenic non-human animals for producing heterologous antibodies |
| JP2938569B2 (ja) | 1990-08-29 | 1999-08-23 | ジェンファーム インターナショナル,インコーポレイティド | 異種免疫グロブリンを作る方法及びトランスジェニックマウス |
| US5814318A (en) | 1990-08-29 | 1998-09-29 | Genpharm International Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5698426A (en) | 1990-09-28 | 1997-12-16 | Ixsys, Incorporated | Surface expression libraries of heteromeric receptors |
| US5543390A (en) | 1990-11-01 | 1996-08-06 | State Of Oregon, Acting By And Through The Oregon State Board Of Higher Education, Acting For And On Behalf Of The Oregon Health Sciences University | Covalent microparticle-drug conjugates for biological targeting |
| WO1992009690A2 (en) | 1990-12-03 | 1992-06-11 | Genentech, Inc. | Enrichment method for variant proteins with altered binding properties |
| EP1471142B1 (en) | 1991-04-10 | 2008-11-19 | The Scripps Research Institute | Heterodimeric receptor libraries using phagemids |
| EP0519596B1 (en) | 1991-05-17 | 2005-02-23 | Merck & Co. Inc. | A method for reducing the immunogenicity of antibody variable domains |
| CA2110799A1 (en) | 1991-06-14 | 1992-12-23 | Arnold H. Horwitz | Microbially-produced antibody fragments and their conjugates |
| CA2103059C (en) | 1991-06-14 | 2005-03-22 | Paul J. Carter | Method for making humanized antibodies |
| US5637481A (en) | 1993-02-01 | 1997-06-10 | Bristol-Myers Squibb Company | Expression vectors encoding bispecific fusion proteins and methods of producing biologically active bispecific fusion proteins in a mammalian cell |
| US5565332A (en) | 1991-09-23 | 1996-10-15 | Medical Research Council | Production of chimeric antibodies - a combinatorial approach |
| WO1993011236A1 (en) | 1991-12-02 | 1993-06-10 | Medical Research Council | Production of anti-self antibodies from antibody segment repertoires and displayed on phage |
| WO1993011794A1 (en) | 1991-12-13 | 1993-06-24 | Xoma Corporation | Methods and materials for preparation of modified antibody variable domains and therapeutic uses thereof |
| GB9203459D0 (en) | 1992-02-19 | 1992-04-08 | Scotgen Ltd | Antibodies with germ-line variable regions |
| US5733743A (en) | 1992-03-24 | 1998-03-31 | Cambridge Antibody Technology Limited | Methods for producing members of specific binding pairs |
| US6174666B1 (en) | 1992-03-27 | 2001-01-16 | The United States Of America As Represented By The Department Of Health And Human Services | Method of eliminating inhibitory/instability regions from mRNA |
| US6010715A (en) | 1992-04-01 | 2000-01-04 | Bertek, Inc. | Transdermal patch incorporating a polymer film incorporated with an active agent |
| US6024975A (en) | 1992-04-08 | 2000-02-15 | Americare International Diagnostics, Inc. | Method of transdermally administering high molecular weight drugs with a polymer skin enhancer |
| ATE427968T1 (de) | 1992-08-21 | 2009-04-15 | Univ Bruxelles | Immunoglobuline ohne leichtkette |
| US6005079A (en) | 1992-08-21 | 1999-12-21 | Vrije Universiteit Brussels | Immunoglobulins devoid of light chains |
| US5639641A (en) | 1992-09-09 | 1997-06-17 | Immunogen Inc. | Resurfacing of rodent antibodies |
| EP0672142B1 (en) | 1992-12-04 | 2001-02-28 | Medical Research Council | Multivalent and multispecific binding proteins, their manufacture and use |
| US6274552B1 (en) | 1993-03-18 | 2001-08-14 | Cytimmune Sciences, Inc. | Composition and method for delivery of biologically-active factors |
| US5523092A (en) | 1993-04-14 | 1996-06-04 | Emory University | Device for local drug delivery and methods for using the same |
| US5985307A (en) | 1993-04-14 | 1999-11-16 | Emory University | Device and method for non-occlusive localized drug delivery |
| ES2162863T3 (es) | 1993-04-29 | 2002-01-16 | Unilever Nv | Produccion de anticuerpos o fragmentos (funcionalizados) de los mismos derivados de inmunoglobulinas de cadena pesada de camelidae. |
| US6004534A (en) | 1993-07-23 | 1999-12-21 | Massachusetts Institute Of Technology | Targeted polymerized liposomes for improved drug delivery |
| AU696293B2 (en) | 1993-12-08 | 1998-09-03 | Genzyme Corporation | Process for generating specific antibodies |
| DK0744958T3 (da) | 1994-01-31 | 2003-10-20 | Univ Boston | Polyklonale antistofbiblioteker |
| US5516637A (en) | 1994-06-10 | 1996-05-14 | Dade International Inc. | Method involving display of protein binding pairs on the surface of bacterial pili and bacteriophage |
| US6107045A (en) | 1994-06-30 | 2000-08-22 | Oklahoma Medical Research Foundation | Antibodies to lipoproteins and apolipoproteins and methods of use thereof |
| US5759542A (en) | 1994-08-05 | 1998-06-02 | New England Deaconess Hospital Corporation | Compositions and methods for the delivery of drugs by platelets for the treatment of cardiovascular and other diseases |
| US5660854A (en) | 1994-11-28 | 1997-08-26 | Haynes; Duncan H | Drug releasing surgical implant or dressing material |
| US5983134A (en) | 1995-04-23 | 1999-11-09 | Electromagnetic Bracing Systems Inc. | Electrophoretic cuff apparatus drug delivery system |
| DE69637481T2 (de) | 1995-04-27 | 2009-04-09 | Amgen Fremont Inc. | Aus immunisierten Xenomäusen stammende menschliche Antikörper gegen IL-8 |
| EP0823941A4 (en) | 1995-04-28 | 2001-09-19 | Abgenix Inc | HUMAN ANTIBODIES DERIVED FROM IMMUNIZED XENO MOUSES |
| US6316652B1 (en) | 1995-06-06 | 2001-11-13 | Kosta Steliou | Drug mitochondrial targeting agents |
| US6167301A (en) | 1995-08-29 | 2000-12-26 | Flower; Ronald J. | Iontophoretic drug delivery device having high-efficiency DC-to-DC energy conversion circuit |
| GB9601081D0 (en) | 1995-10-06 | 1996-03-20 | Cambridge Antibody Tech | Specific binding members for human transforming growth factor beta;materials and methods |
| WO1997014719A1 (en) | 1995-10-16 | 1997-04-24 | Unilever N.V. | A bifunctional or bivalent antibody fragment analogue |
| US6039975A (en) | 1995-10-17 | 2000-03-21 | Hoffman-La Roche Inc. | Colon targeted delivery system |
| JP2978435B2 (ja) | 1996-01-24 | 1999-11-15 | チッソ株式会社 | アクリロキシプロピルシランの製造方法 |
| AU728657B2 (en) | 1996-03-18 | 2001-01-18 | Board Of Regents, The University Of Texas System | Immunoglobulin-like domains with increased half-lives |
| TW345603B (en) | 1996-05-29 | 1998-11-21 | Gmundner Fertigteile Gmbh | A noise control device for tracks |
| US6027947A (en) | 1996-08-20 | 2000-02-22 | Ramtron International Corporation | Partially or completely encapsulated top electrode of a ferroelectric capacitor |
| US5985317A (en) | 1996-09-06 | 1999-11-16 | Theratech, Inc. | Pressure sensitive adhesive matrix patches for transdermal delivery of salts of pharmaceutical agents |
| HUP0000116A3 (en) | 1996-10-01 | 2000-08-28 | Stanford Res Inst Int | Taste-masked microcapsule compositions and methods of manufacture |
| US5916771A (en) | 1996-10-11 | 1999-06-29 | Abgenix, Inc. | Production of a multimeric protein by cell fusion method |
| US6131570A (en) | 1998-06-30 | 2000-10-17 | Aradigm Corporation | Temperature controlling device for aerosol drug delivery |
| ES2301183T3 (es) | 1996-12-03 | 2008-06-16 | Amgen Fremont Inc. | Anticuerpo completamente humano que se une al receptor del egfr. |
| US5860957A (en) | 1997-02-07 | 1999-01-19 | Sarcos, Inc. | Multipathway electronically-controlled drug delivery system |
| US6277375B1 (en) | 1997-03-03 | 2001-08-21 | Board Of Regents, The University Of Texas System | Immunoglobulin-like domains with increased half-lives |
| US6120751A (en) | 1997-03-21 | 2000-09-19 | Imarx Pharmaceutical Corp. | Charged lipids and uses for the same |
| CN100387621C (zh) | 1997-04-14 | 2008-05-14 | 麦可麦脱股份公司 | 抗人抗原受体的新的生产方法及其用途 |
| US6060082A (en) | 1997-04-18 | 2000-05-09 | Massachusetts Institute Of Technology | Polymerized liposomes targeted to M cells and useful for oral or mucosal drug delivery |
| US20020062010A1 (en) | 1997-05-02 | 2002-05-23 | Genentech, Inc. | Method for making multispecific antibodies having heteromultimeric and common components |
| US7951917B1 (en) | 1997-05-02 | 2011-05-31 | Genentech, Inc. | Method for making multispecific antibodies having heteromultimeric and common components |
| US6235883B1 (en) | 1997-05-05 | 2001-05-22 | Abgenix, Inc. | Human monoclonal antibodies to epidermal growth factor receptor |
| US5948433A (en) | 1997-08-21 | 1999-09-07 | Bertek, Inc. | Transdermal patch |
| JP4395200B2 (ja) | 1997-10-28 | 2010-01-06 | バンドー化学株式会社 | 皮膚貼付薬シート及びそのための基材シートの製造方法 |
| US6048736A (en) | 1998-04-29 | 2000-04-11 | Kosak; Kenneth M. | Cyclodextrin polymers for carrying and releasing drugs |
| US6737056B1 (en) | 1999-01-15 | 2004-05-18 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US6271359B1 (en) | 1999-04-14 | 2001-08-07 | Musc Foundation For Research Development | Tissue-specific and pathogen-specific toxic agents and ribozymes |
| US6256533B1 (en) | 1999-06-09 | 2001-07-03 | The Procter & Gamble Company | Apparatus and method for using an intracutaneous microneedle array |
| ATE440111T1 (de) | 1999-11-29 | 2009-09-15 | Bac Ip B V | Immobilisierte antigenbindende moleküle aus einer domäne |
| US6261595B1 (en) | 2000-02-29 | 2001-07-17 | Zars, Inc. | Transdermal drug patch with attached pocket for controlled heating device |
| CZ20022971A3 (cs) | 2000-03-03 | 2003-02-12 | Smithkline Beecham Biologicals S. A. | Vakcinační prostředek vhodný pro léčbu aterosklerózy |
| GB0121171D0 (en) | 2001-08-31 | 2001-10-24 | Glaxosmithkline Biolog Sa | Vaccine |
| ES2649037T3 (es) | 2000-12-12 | 2018-01-09 | Medimmune, Llc | Moléculas con semividas prolongadas, composiciones y usos de las mismas |
| JP2005538706A (ja) | 2001-07-12 | 2005-12-22 | ジェファーソン フーテ, | スーパーヒト化抗体 |
| FR2829581A1 (fr) | 2001-09-07 | 2003-03-14 | Genfit S A | Methodes de criblage de molecules utiles pour la prevention ou le traitement du syndrome metabolique, des maladies cardiovasculaires et de l'atherosclerose |
| NZ532526A (en) | 2001-10-25 | 2007-01-26 | Genentech Inc | Compositions comprising a glycoprotein having a Fc region |
| US7662925B2 (en) | 2002-03-01 | 2010-02-16 | Xencor, Inc. | Optimized Fc variants and methods for their generation |
| US7217797B2 (en) | 2002-10-15 | 2007-05-15 | Pdl Biopharma, Inc. | Alteration of FcRn binding affinities or serum half-lives of antibodies by mutagenesis |
| US9068234B2 (en) | 2003-01-21 | 2015-06-30 | Ptc Therapeutics, Inc. | Methods and agents for screening for compounds capable of modulating gene expression |
| GB0305793D0 (en) | 2003-03-13 | 2003-04-16 | Glaxosmithkline Biolog Sa | Vaccine |
| GB0305790D0 (en) * | 2003-03-13 | 2003-04-16 | Glaxosmithkline Biolog Sa | Novel Composition |
| GB0305794D0 (en) * | 2003-03-13 | 2003-04-16 | Glaxosmithkline Biolog Sa | Vaccine |
| US9062306B2 (en) * | 2003-04-29 | 2015-06-23 | Biocrine Ab | Methods for identifying compounds for treating type 1 diabetes |
| GB2404981A (en) | 2003-08-15 | 2005-02-16 | Univ Geneve | Diagnostic method for stroke |
| US20050042664A1 (en) | 2003-08-22 | 2005-02-24 | Medimmune, Inc. | Humanization of antibodies |
| JP2007531707A (ja) | 2003-10-15 | 2007-11-08 | ピーディーエル バイオファーマ, インコーポレイテッド | IGの重鎖定常領域の位置250、314および/または428の変異誘発によるFc融合タンパク質血清半減期の改変 |
| AU2004290070A1 (en) | 2003-11-12 | 2005-05-26 | Biogen Idec Ma Inc. | Neonatal Fc receptor (FcRn)-binding polypeptide variants, dimeric Fc binding proteins and methods related thereto |
| CN102746404B (zh) | 2004-11-12 | 2016-01-20 | 赞科股份有限公司 | 对FcRn的结合被改变的Fc变体 |
| US8802820B2 (en) | 2004-11-12 | 2014-08-12 | Xencor, Inc. | Fc variants with altered binding to FcRn |
| US8367805B2 (en) | 2004-11-12 | 2013-02-05 | Xencor, Inc. | Fc variants with altered binding to FcRn |
| ES2523666T3 (es) | 2005-05-31 | 2014-11-28 | Board Of Regents, The University Of Texas System | Anticuerpos IgG1 con la parte Fc mutada para el aumento de unión al receptor FcRn y usos de los mismos |
| US8629245B2 (en) | 2007-05-01 | 2014-01-14 | Research Development Foundation | Immunoglobulin Fc libraries |
| US8227577B2 (en) | 2007-12-21 | 2012-07-24 | Hoffman-La Roche Inc. | Bivalent, bispecific antibodies |
| HRP20150279T1 (hr) | 2007-12-26 | 2015-05-08 | Xencor, Inc. | Fc inaäśice s promijenjenim vezanjem na fcrn |
| US8444976B2 (en) | 2008-07-02 | 2013-05-21 | Argen-X B.V. | Antigen binding polypeptides |
| GB2461546B (en) | 2008-07-02 | 2010-07-07 | Argen X Bv | Antigen binding polypeptides |
| US20170247470A9 (en) | 2008-09-17 | 2017-08-31 | Xencor, Inc. | Rapid clearance of antigen complexes using novel antibodies |
| EP3524620A1 (en) | 2008-10-14 | 2019-08-14 | Genentech, Inc. | Immunoglobulin variants and uses thereof |
| EP2233500A1 (en) | 2009-03-20 | 2010-09-29 | LFB Biotechnologies | Optimized Fc variants |
| JP2012530900A (ja) | 2009-06-17 | 2012-12-06 | メイン スタンダーズ カンパニー リミテッド ライアビリティ カンパニー | リポタンパク質特異的アポリポタンパク質を測定する方法 |
| ES2865648T3 (es) | 2009-06-26 | 2021-10-15 | Regeneron Pharma | Anticuerpos biespecíficos fácilmente aislados con formato de inmunoglobulina nativa |
| JP5683581B2 (ja) | 2009-06-30 | 2015-03-11 | リサーチ ディベロップメント ファウンデーション | 免疫グロブリンFcポリペプチド |
| GB2476681B (en) | 2010-01-04 | 2012-04-04 | Argen X Bv | Humanized camelid VH, VK and VL immunoglobulin domains |
| AU2011225716A1 (en) | 2010-03-11 | 2012-09-27 | Pfizer Inc. | Antibodies with pH dependent antigen binding |
| CN112812184A (zh) | 2011-02-25 | 2021-05-18 | 中外制药株式会社 | FcγRIIb特异性Fc抗体 |
| MX344807B (es) * | 2011-06-21 | 2017-01-09 | Alnylam Pharmaceuticals Inc | Composiciones y metodos para inhibicion de genes para alipoproteina c-iii (apoc3). |
| US8557513B2 (en) | 2011-06-27 | 2013-10-15 | Biocrine Ab | Methods for treating and/or limiting development of diabetes |
| WO2013040429A1 (en) * | 2011-09-14 | 2013-03-21 | Rana Therapeutics Inc. | Multimeric oligonucleotide compounds |
| EP2752200B1 (en) | 2011-09-30 | 2023-11-01 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule inducing immune response to target antigen |
| WO2013064700A2 (en) | 2011-11-03 | 2013-05-10 | Argen-X B.V. | Chimeric polypeptides and methods of use |
| WO2013074598A1 (en) | 2011-11-18 | 2013-05-23 | Merck Sharp & Dohme Corp. | Fc CONTAINING POLYPEPTIDES HAVING INCREASED ANTI-INFLAMMATORY PROPERTIES AND INCREASED FcRN BINDING |
| KR20230143201A (ko) | 2011-11-30 | 2023-10-11 | 추가이 세이야쿠 가부시키가이샤 | 면역 복합체를 형성하는 세포내로의 운반체(캐리어)를 포함하는 의약 |
| KR102041412B1 (ko) | 2011-12-30 | 2019-11-11 | 한미사이언스 주식회사 | 면역글로불린 Fc 단편 유도체 |
| CN104302170B (zh) | 2012-03-16 | 2016-09-28 | 瑞泽恩制药公司 | 生产具有ph依赖性结合特性的抗原结合蛋白的小鼠 |
| US20150166654A1 (en) | 2012-05-30 | 2015-06-18 | Chugai Seiyaku Kabushiki Kaisha | Target tissue-specific antigen-binding molecule |
| CN110256559B (zh) * | 2012-07-25 | 2023-05-26 | 塞尔德克斯医疗公司 | 抗kit抗体及其用途 |
| DK2890711T3 (en) | 2012-08-31 | 2017-02-27 | Argen-X Nv | Process for Preparation of Inter-species, Intra-Target Cross-Reactivity Antibody Molecules |
| CN104583236A (zh) | 2012-08-31 | 2015-04-29 | 阿尔金-X有限公司 | 高度多样的组合抗体文库 |
| US9452151B2 (en) * | 2013-02-06 | 2016-09-27 | Amarin Pharmaceuticals Ireland Limited | Methods of reducing apolipoprotein C-III |
| CA2901119C (en) | 2013-02-14 | 2022-10-18 | Isis Pharmaceuticals, Inc. | Modulation of apolipoprotein c-iii (apociii) expression in lipoprotein lipase deficient (lpld) populations |
| GB201302878D0 (en) | 2013-02-19 | 2013-04-03 | Argen X Bv | Modified igG molecules |
| WO2014131008A1 (en) * | 2013-02-25 | 2014-08-28 | Intrinsic Metabio Solutions, Llc | A polipoprotein c3 (apociii) antagonists and methods of their use to remove apociii inhibition of lipoprotein lipase (lpl) |
| US20150259416A1 (en) | 2014-03-12 | 2015-09-17 | Biocrine Ab | Methods for treating and/or limiting development of diabetes |
| US20140294812A1 (en) | 2013-03-15 | 2014-10-02 | Xencor, Inc. | Fc variants that improve fcrn binding and/or increase antibody half-life |
| CN105164157B (zh) | 2013-04-29 | 2024-05-28 | 豪夫迈·罗氏有限公司 | Fc-受体结合的修饰的非对称抗体及使用方法 |
| GB2517953A (en) | 2013-09-05 | 2015-03-11 | Argen X Bv | Antibodies to complex targets |
| CA2934644C (en) | 2013-12-24 | 2023-11-21 | Argen-X N.V. | Fcrn antagonists and methods of use |
| GB201414823D0 (en) | 2014-08-20 | 2014-10-01 | Argen X Bv | Multispecific antibodies |
| US20160264669A1 (en) | 2015-03-09 | 2016-09-15 | Argen-X N.V. | Methods of reducing serum levels of fc-containing agents using fcrn antagonists |
| TW201713690A (zh) | 2015-08-07 | 2017-04-16 | 再生元醫藥公司 | 抗angptl8抗體及其用途 |
| WO2017055627A1 (en) | 2015-10-02 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Apoc3 mutations for the diagnosis and therapy of hereditary renal amyloidosis disease |
| MX2018005540A (es) | 2015-11-06 | 2018-11-09 | Gemphire Therapeutics Inc | Tratamiento de dislipemia mixta. |
| MA45602A (fr) | 2016-07-08 | 2019-05-15 | Staten Biotechnology B V | Anticorps anti-apoc3 et leurs méthodes d'utilisation |
| GB201617270D0 (en) | 2016-10-11 | 2016-11-23 | Argen-X N V And Fairjourney Biologics | Triple vector for expressing antibody molecules in full therapeutic format |
| CN110506056A (zh) | 2017-04-21 | 2019-11-26 | 斯塔滕生物技术有限公司 | 抗apoc3抗体和其使用方法 |
| GB201707484D0 (en) | 2017-05-10 | 2017-06-21 | Argenx Bvba | Method of preparing ph-dependent antibodies |
| US10538583B2 (en) | 2017-10-31 | 2020-01-21 | Staten Biotechnology B.V. | Anti-APOC3 antibodies and compositions thereof |
| MX2020004512A (es) | 2017-10-31 | 2020-08-13 | Anticuerpos anti apolipoproteina c-iii (anti-apoc3) y metodos de uso de los mismos. | |
| CN112969503A (zh) | 2018-10-03 | 2021-06-15 | 斯塔滕生物技术有限公司 | 对人类和食蟹猕猴apoc3具有特异性的抗体和其使用方法 |
-
2017
- 2017-07-07 MA MA045602A patent/MA45602A/fr unknown
- 2017-07-07 WO PCT/IB2017/054125 patent/WO2018007999A1/en not_active Ceased
- 2017-07-07 EP EP17745513.6A patent/EP3481864A1/en not_active Withdrawn
- 2017-07-07 CA CA3030099A patent/CA3030099A1/en active Pending
- 2017-07-07 CN CN201780055144.9A patent/CN109689685A/zh active Pending
- 2017-07-07 JP JP2019521522A patent/JP2019525772A/ja active Pending
- 2017-07-07 KR KR1020197002789A patent/KR20190039937A/ko not_active Ceased
- 2017-07-07 AU AU2017292184A patent/AU2017292184A1/en not_active Abandoned
-
2019
- 2019-01-07 US US16/241,407 patent/US11091539B2/en not_active Expired - Fee Related
-
2021
- 2021-07-07 US US17/305,413 patent/US20220025027A1/en not_active Abandoned
- 2021-07-07 US US17/305,412 patent/US20220025026A1/en not_active Abandoned
-
2022
- 2022-07-07 JP JP2022109837A patent/JP2022137159A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| US20190241648A1 (en) | 2019-08-08 |
| MA45602A (fr) | 2019-05-15 |
| US11091539B2 (en) | 2021-08-17 |
| JP2019525772A (ja) | 2019-09-12 |
| EP3481864A1 (en) | 2019-05-15 |
| WO2018007999A1 (en) | 2018-01-11 |
| AU2017292184A1 (en) | 2019-02-07 |
| CN109689685A (zh) | 2019-04-26 |
| US20220025026A1 (en) | 2022-01-27 |
| JP2022137159A (ja) | 2022-09-21 |
| US20220025027A1 (en) | 2022-01-27 |
| CA3030099A1 (en) | 2018-01-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11091539B2 (en) | Anti-ApoC3 antibodies and methods of use thereof | |
| AU2013370009B2 (en) | Anti-granulysin antibodies and methods of use thereof | |
| US11242381B2 (en) | Anti-ApoC3 antibodies and methods of use thereof | |
| US11248041B2 (en) | Anti-ApoC3 antibodies | |
| US10538583B2 (en) | Anti-APOC3 antibodies and compositions thereof | |
| US20210347865A1 (en) | Antibodies specific for human and cynomolgus apoc3 and methods of use thereof | |
| RU2781074C2 (ru) | АНТИ-ApoC3 АНТИТЕЛА И СПОСОБЫ ИХ ПРИМЕНЕНИЯ | |
| HK40031744A (en) | Anti-apoc3 antibodies and methods of use thereof | |
| HK40018468A (en) | Anti-apoc3 antibodies and methods of use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20190128 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20200706 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20220719 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20230131 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20220719 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |