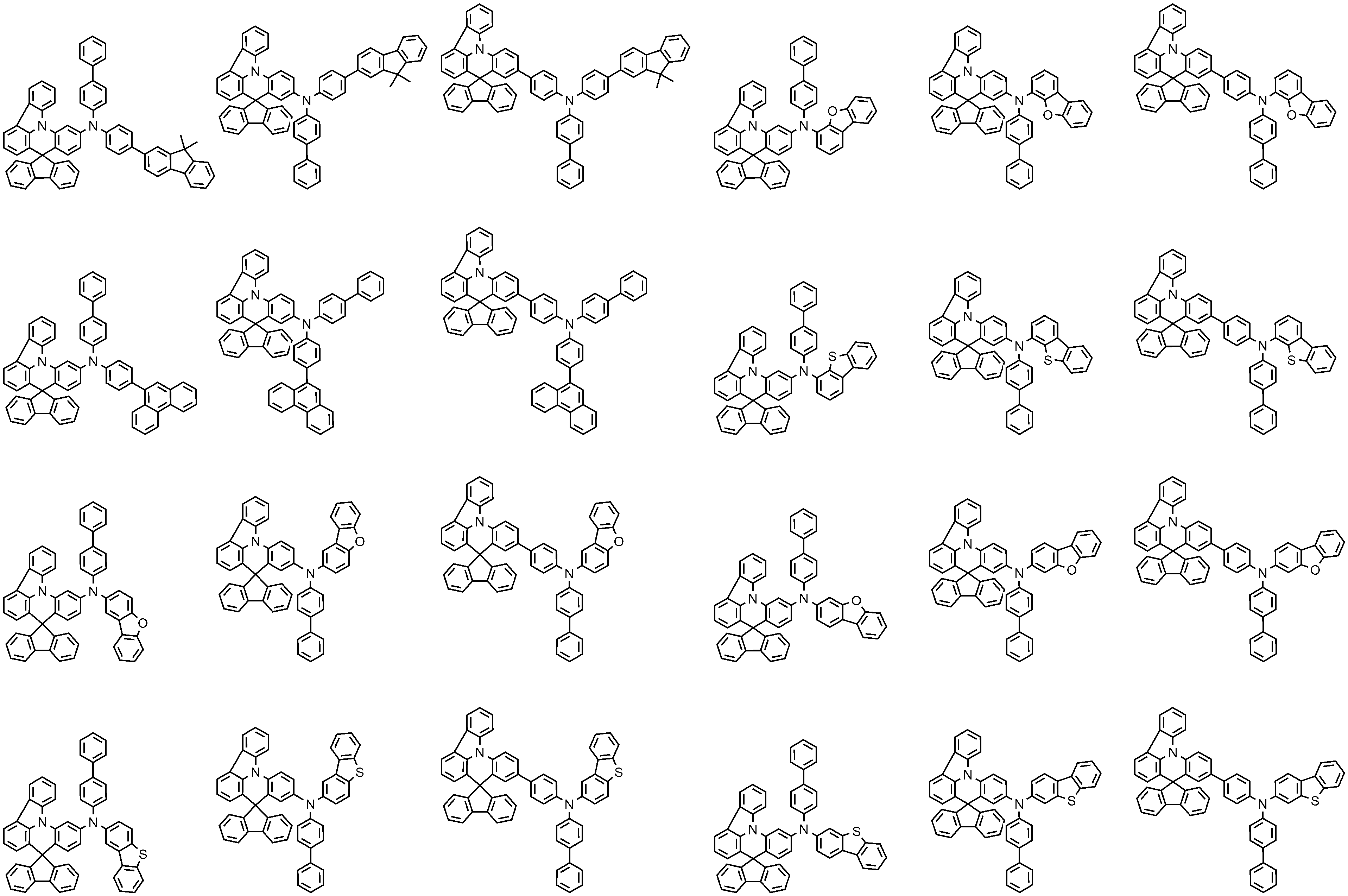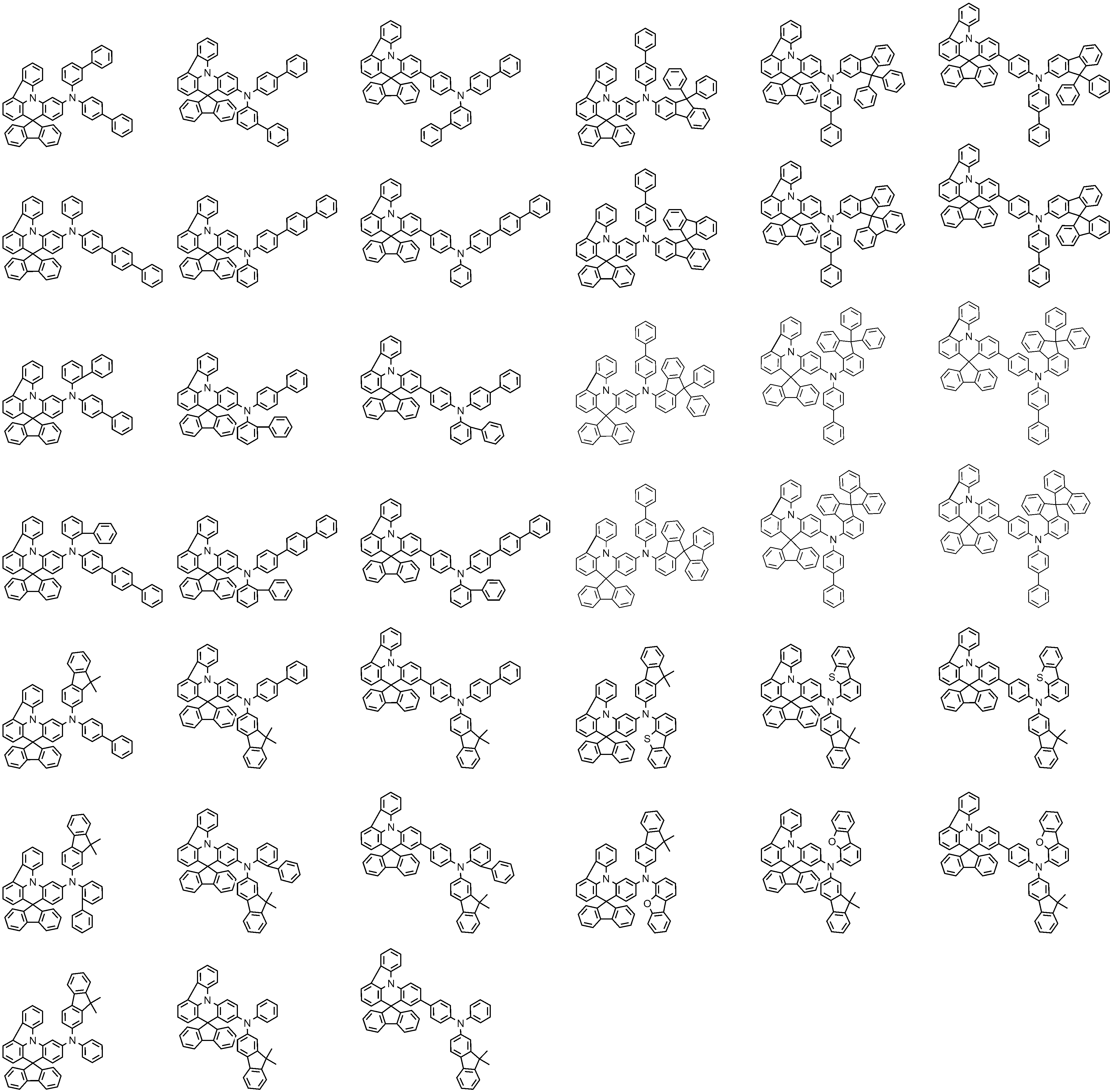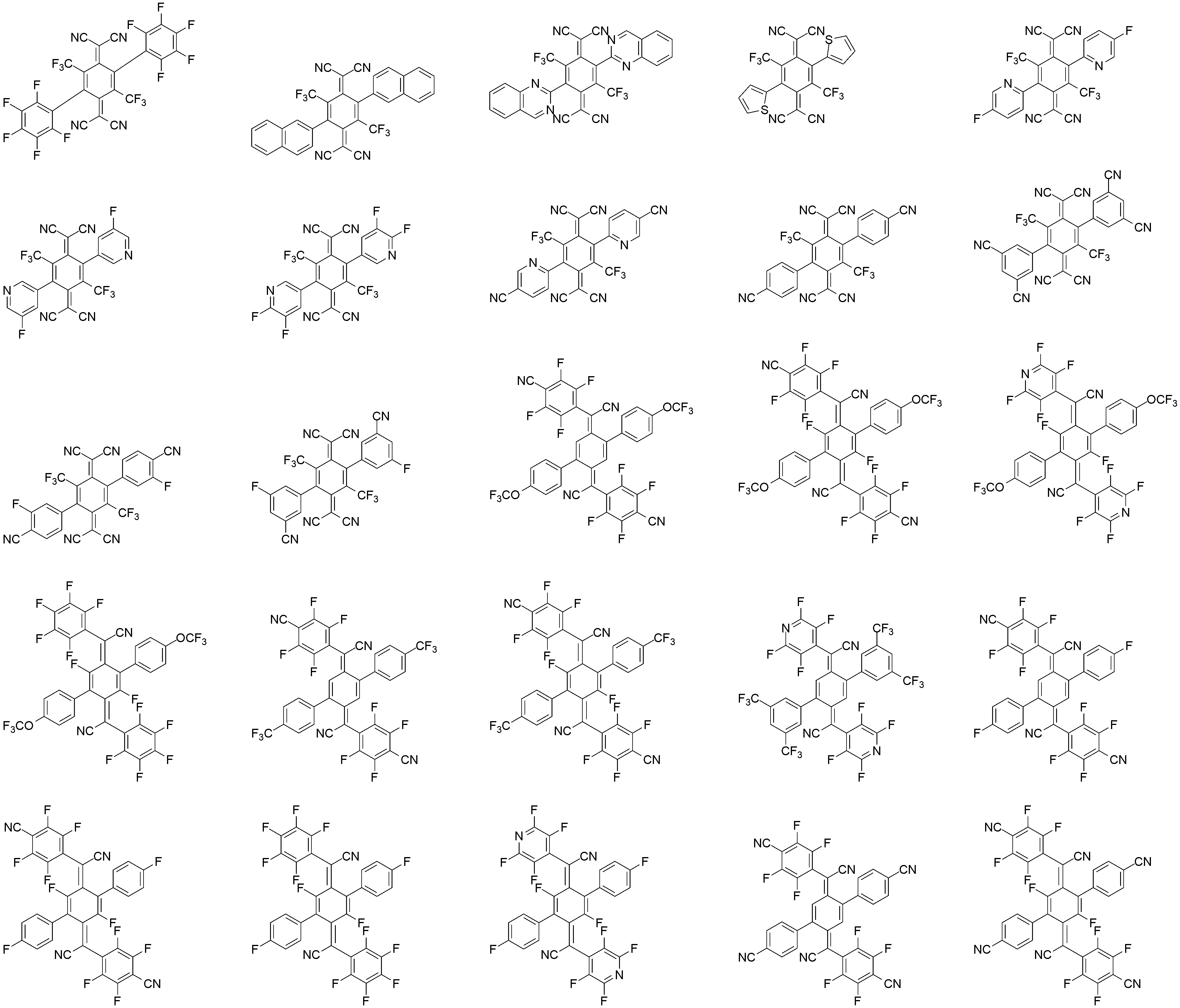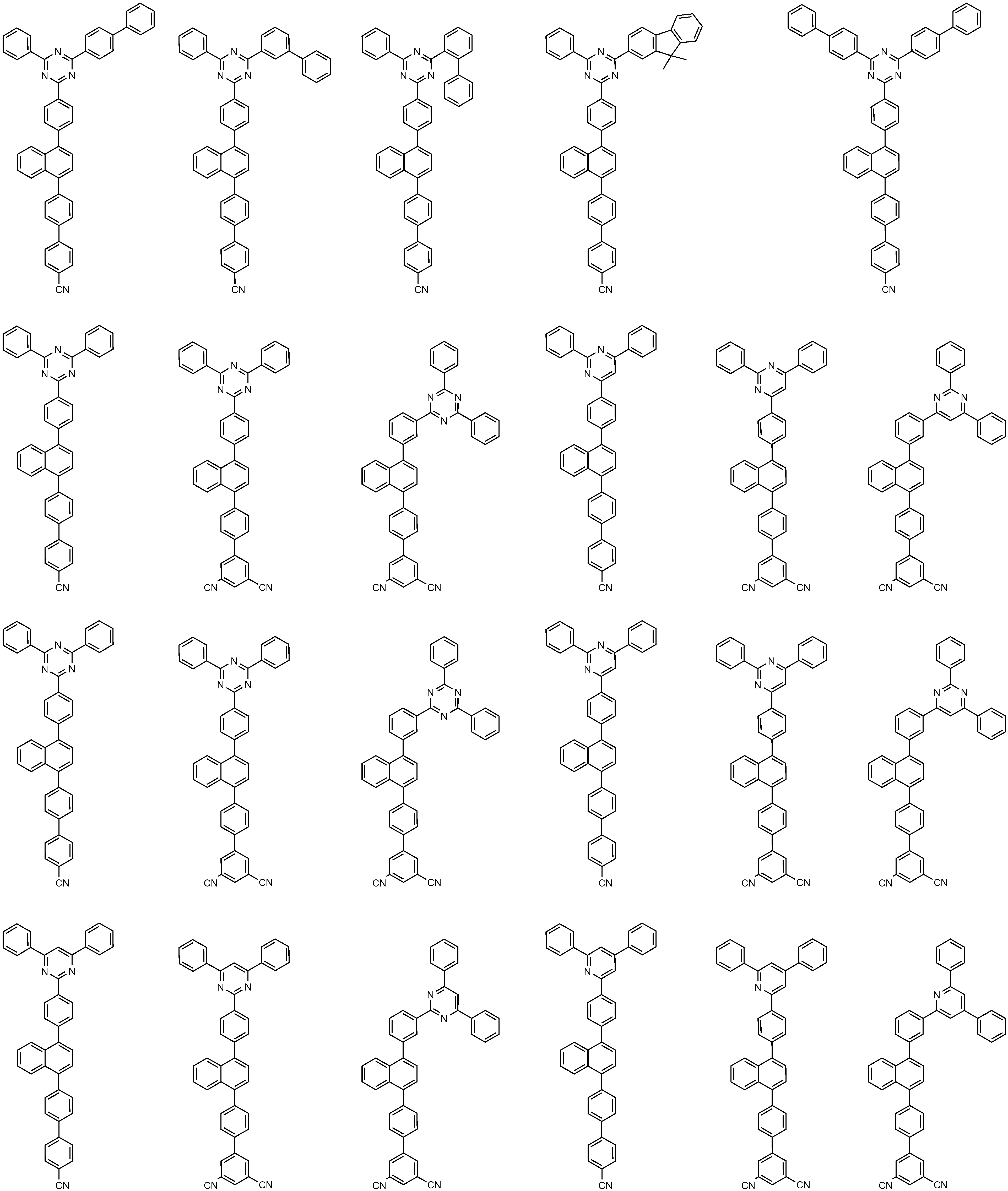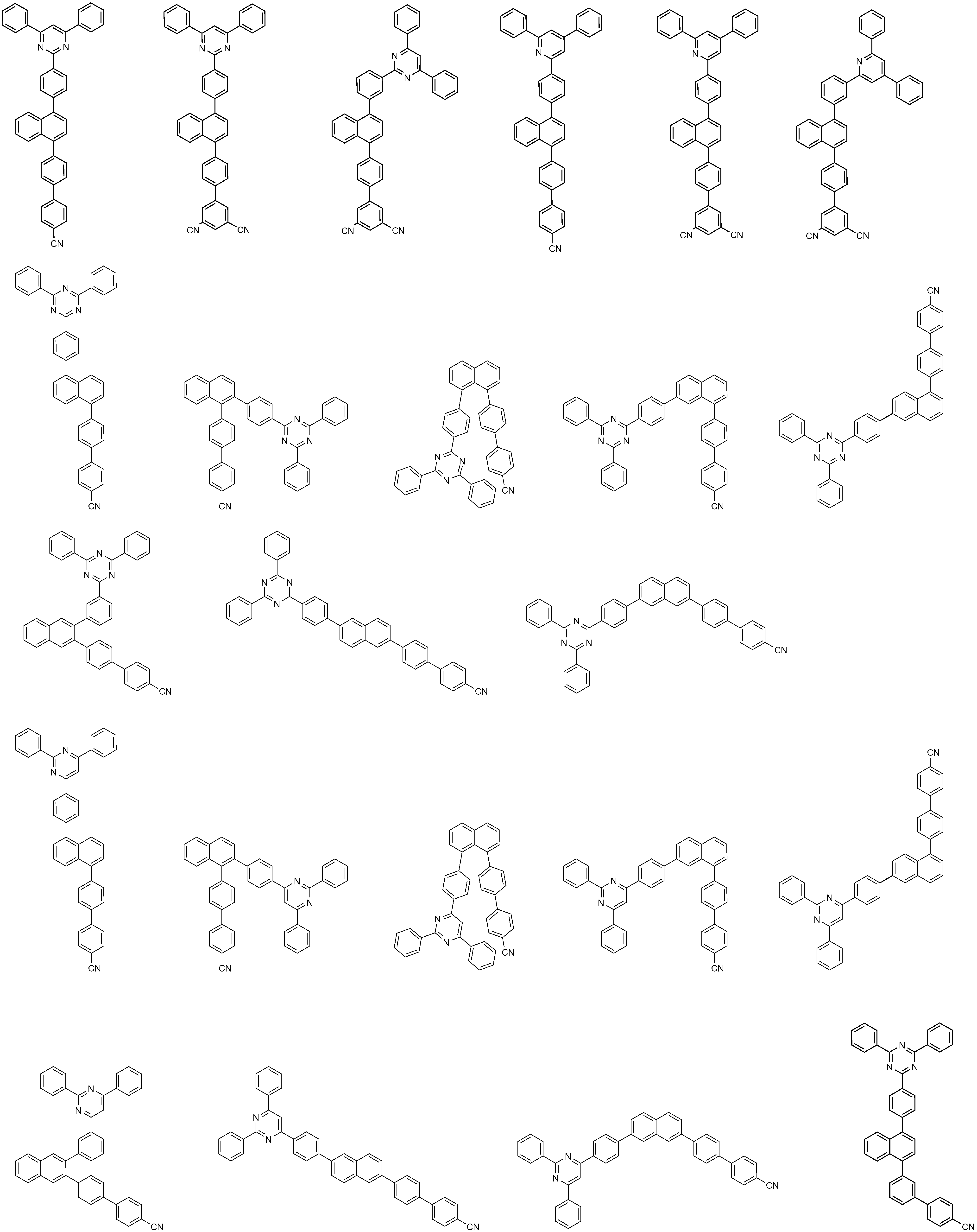KR20180098121A - 유기 발광 소자 - Google Patents
유기 발광 소자 Download PDFInfo
- Publication number
- KR20180098121A KR20180098121A KR1020170177273A KR20170177273A KR20180098121A KR 20180098121 A KR20180098121 A KR 20180098121A KR 1020170177273 A KR1020170177273 A KR 1020170177273A KR 20170177273 A KR20170177273 A KR 20170177273A KR 20180098121 A KR20180098121 A KR 20180098121A
- Authority
- KR
- South Korea
- Prior art keywords
- group
- light emitting
- substituted
- unsubstituted
- organic light
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 0 CC(N(C)*=O)=O Chemical compound CC(N(C)*=O)=O 0.000 description 10
- CKHABRPAYWRBEV-UHFFFAOYSA-N CC(CC=C1c2c3c(C45c6ccccc6C6=CC=CCC46C)ccc2)C=C1N3c(cc1)c5cc1Cl Chemical compound CC(CC=C1c2c3c(C45c6ccccc6C6=CC=CCC46C)ccc2)C=C1N3c(cc1)c5cc1Cl CKHABRPAYWRBEV-UHFFFAOYSA-N 0.000 description 1
- ODPJQTGHDFWLPR-UHFFFAOYSA-N CC(N(C)C(C1CCCC1)=O)=O Chemical compound CC(N(C)C(C1CCCC1)=O)=O ODPJQTGHDFWLPR-UHFFFAOYSA-N 0.000 description 1
- ZSBDPRIWBYHIAF-UHFFFAOYSA-N CC(NC(C)=O)=O Chemical compound CC(NC(C)=O)=O ZSBDPRIWBYHIAF-UHFFFAOYSA-N 0.000 description 1
- GCCHEFAXUVFOKS-UHFFFAOYSA-N CCC(N(CC)C(C)=O)=O Chemical compound CCC(N(CC)C(C)=O)=O GCCHEFAXUVFOKS-UHFFFAOYSA-N 0.000 description 1
- RIDYSAUBERCXSL-UHFFFAOYSA-N CN(C=O)C(c1ccccc1)=O Chemical compound CN(C=O)C(c1ccccc1)=O RIDYSAUBERCXSL-UHFFFAOYSA-N 0.000 description 1
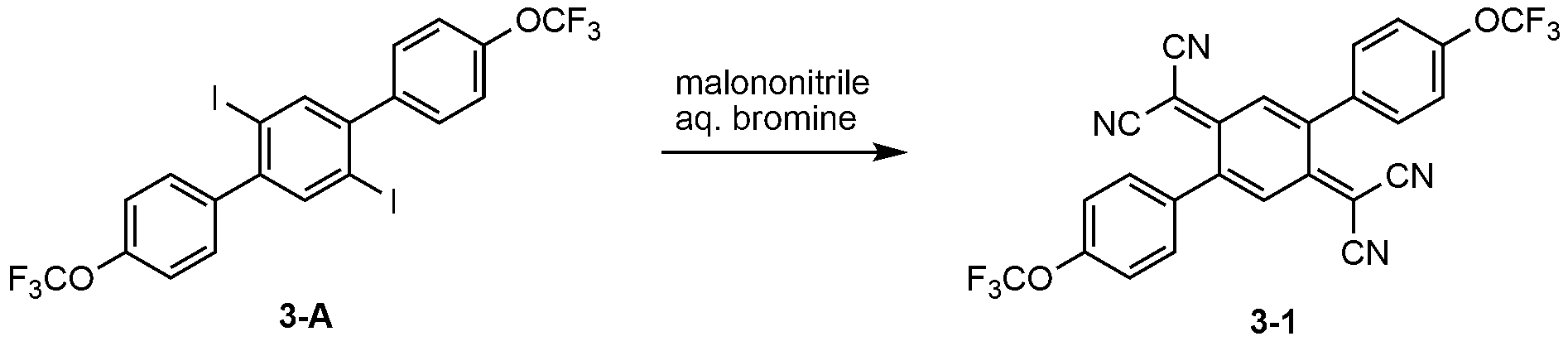
- KYTFDGYTSRGDCJ-UHFFFAOYSA-N Cc1cc(C(C(C(C(F)(F)F)=C2c(cc3)cc(C)c3OC(F)(F)F)=C(C#N)C#N)=C(C(F)(F)F)C2=C(C#N)C#N)ccc1OC(F)(F)F Chemical compound Cc1cc(C(C(C(C(F)(F)F)=C2c(cc3)cc(C)c3OC(F)(F)F)=C(C#N)C#N)=C(C(F)(F)F)C2=C(C#N)C#N)ccc1OC(F)(F)F KYTFDGYTSRGDCJ-UHFFFAOYSA-N 0.000 description 1
- PIRZRRXHAPFZGY-OGGGYYITSA-N N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c(cc2)ccc2C#N)\C(c(cc2)ccc2C#N)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N Chemical compound N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c(cc2)ccc2C#N)\C(c(cc2)ccc2C#N)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N PIRZRRXHAPFZGY-OGGGYYITSA-N 0.000 description 1
- SYRFRQNKOGBNJG-MBALSZOMSA-N N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c(cc2)ccc2F)\C(c(cc2)ccc2F)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N Chemical compound N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c(cc2)ccc2F)\C(c(cc2)ccc2F)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N SYRFRQNKOGBNJG-MBALSZOMSA-N 0.000 description 1
- QIPBCPUBNGWUTI-MBALSZOMSA-N N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c(cc2)ccc2OC(F)(F)F)\C(c(cc2)ccc2OC(F)(F)F)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N Chemical compound N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c(cc2)ccc2OC(F)(F)F)\C(c(cc2)ccc2OC(F)(F)F)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N QIPBCPUBNGWUTI-MBALSZOMSA-N 0.000 description 1
- UDDCCYVMRPDBSP-MBALSZOMSA-N N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c2ccc(C(F)(F)F)cc2)\C(c2ccc(C(F)(F)F)cc2)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N Chemical compound N#C/C(/c(c(F)c(c(C#N)c1F)F)c1F)=C(/C=C1c2ccc(C(F)(F)F)cc2)\C(c2ccc(C(F)(F)F)cc2)=C/C1=C(\c(c(F)c(c(C#N)c1F)F)c1F)/C#N UDDCCYVMRPDBSP-MBALSZOMSA-N 0.000 description 1
- QHGUUCWHGOQENJ-FLFKKZLDSA-N N#C/C(/c(c(F)c(nc1F)F)c1F)=C(/C=C1c2cc(C(F)(F)F)cc(C(F)(F)F)c2)\C(c2cc(C(F)(F)F)cc(C(F)(F)F)c2)=C/C1=C(\c(c(F)c(nc1F)F)c1F)/C#N Chemical compound N#C/C(/c(c(F)c(nc1F)F)c1F)=C(/C=C1c2cc(C(F)(F)F)cc(C(F)(F)F)c2)\C(c2cc(C(F)(F)F)cc(C(F)(F)F)c2)=C/C1=C(\c(c(F)c(nc1F)F)c1F)/C#N QHGUUCWHGOQENJ-FLFKKZLDSA-N 0.000 description 1
- JXAZNKRAICXWDD-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(c(F)c(c(F)c2F)F)c2F)C(c(c(F)c(c(F)c2F)F)c2F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(c(F)c(c(F)c2F)F)c2F)C(c(c(F)c(c(F)c2F)F)c2F)=C(C(F)(F)F)C1=C(C#N)C#N JXAZNKRAICXWDD-UHFFFAOYSA-N 0.000 description 1
- XVSGHPZPQITZAS-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2C#N)C(c(cc2)ccc2C#N)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2C#N)C(c(cc2)ccc2C#N)=C(C(F)(F)F)C1=C(C#N)C#N XVSGHPZPQITZAS-UHFFFAOYSA-N 0.000 description 1
- CNABCKCUPUSUFZ-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2F)C(c(cc2)ccc2F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2F)C(c(cc2)ccc2F)=C(C(F)(F)F)C1=C(C#N)C#N CNABCKCUPUSUFZ-UHFFFAOYSA-N 0.000 description 1
- NHXNGLVYANTQSQ-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2OC(C(F)(F)F)C(F)(F)F)C(c(cc2)ccc2OC(C(F)(F)F)C(F)(F)F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2OC(C(F)(F)F)C(F)(F)F)C(c(cc2)ccc2OC(C(F)(F)F)C(F)(F)F)=C(C(F)(F)F)C1=C(C#N)C#N NHXNGLVYANTQSQ-UHFFFAOYSA-N 0.000 description 1
- SHHQJGPOBARUOR-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2OC(F)(F)F)C(c(cc2)ccc2OC(F)(F)F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ccc2OC(F)(F)F)C(c(cc2)ccc2OC(F)(F)F)=C(C(F)(F)F)C1=C(C#N)C#N SHHQJGPOBARUOR-UHFFFAOYSA-N 0.000 description 1
- FLAONHUPEXWBOY-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ncc2C#N)C(c(cc2)ncc2C#N)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ncc2C#N)C(c(cc2)ncc2C#N)=C(C(F)(F)F)C1=C(C#N)C#N FLAONHUPEXWBOY-UHFFFAOYSA-N 0.000 description 1
- RDFHUMWXIOGDHH-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ncc2F)C(c(cc2)ncc2F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2)ncc2F)C(c(cc2)ncc2F)=C(C(F)(F)F)C1=C(C#N)C#N RDFHUMWXIOGDHH-UHFFFAOYSA-N 0.000 description 1
- RJMOZEUWALJHLL-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2F)cc(F)c2F)C(c(cc2F)cc(F)c2F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2F)cc(F)c2F)C(c(cc2F)cc(F)c2F)=C(C(F)(F)F)C1=C(C#N)C#N RJMOZEUWALJHLL-UHFFFAOYSA-N 0.000 description 1
- ZAIPPVPRBWTAHW-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2F)ccc2C#N)C(c(cc2)cc(F)c2C#N)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2F)ccc2C#N)C(c(cc2)cc(F)c2C#N)=C(C(F)(F)F)C1=C(C#N)C#N ZAIPPVPRBWTAHW-UHFFFAOYSA-N 0.000 description 1
- DFDMWGFHTCIXGV-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2F)ccc2OC(F)(F)F)C(c(cc2)cc(F)c2OC(F)(F)F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cc2F)ccc2OC(F)(F)F)C(c(cc2)cc(F)c2OC(F)(F)F)=C(C(F)(F)F)C1=C(C#N)C#N DFDMWGFHTCIXGV-UHFFFAOYSA-N 0.000 description 1
- LVIJLSHMURSSFB-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c(cn2)cc(F)c2F)C(c(cn2)cc(F)c2F)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c(cn2)cc(F)c2F)C(c(cn2)cc(F)c2F)=C(C(F)(F)F)C1=C(C#N)C#N LVIJLSHMURSSFB-UHFFFAOYSA-N 0.000 description 1
- CKTWNTHHCOPFEY-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(C#N)cc(C#N)c2)C(c2cc(C#N)cc(C#N)c2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(C#N)cc(C#N)c2)C(c2cc(C#N)cc(C#N)c2)=C(C(F)(F)F)C1=C(C#N)C#N CKTWNTHHCOPFEY-UHFFFAOYSA-N 0.000 description 1
- BLASRMPIHAMYGM-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)c(C(F)(F)F)c(F)c2)C(c2cc(F)c(C(F)(F)F)c(F)c2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)c(C(F)(F)F)c(F)c2)C(c2cc(F)c(C(F)(F)F)c(F)c2)=C(C(F)(F)F)C1=C(C#N)C#N BLASRMPIHAMYGM-UHFFFAOYSA-N 0.000 description 1
- XXCIDDBUAOCKHI-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)c(C(F)(F)F)cc2)C(c2ccc(C(F)(F)F)c(F)c2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)c(C(F)(F)F)cc2)C(c2ccc(C(F)(F)F)c(F)c2)=C(C(F)(F)F)C1=C(C#N)C#N XXCIDDBUAOCKHI-UHFFFAOYSA-N 0.000 description 1
- SKVBGGFAVJRTGW-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)cc(F)c2)C(c2cc(F)cc(F)c2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)cc(F)c2)C(c2cc(F)cc(F)c2)=C(C(F)(F)F)C1=C(C#N)C#N SKVBGGFAVJRTGW-UHFFFAOYSA-N 0.000 description 1
- CWIRZMWGOYJTGJ-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)cnc2)C(c2cc(F)cnc2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc(F)cnc2)C(c2cc(F)cnc2)=C(C(F)(F)F)C1=C(C#N)C#N CWIRZMWGOYJTGJ-UHFFFAOYSA-N 0.000 description 1
- PBDFXTLEURDLMW-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc3ccccc3cc2)C(c2ccc(cccc3)c3c2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2cc3ccccc3cc2)C(c2ccc(cccc3)c3c2)=C(C(F)(F)F)C1=C(C#N)C#N PBDFXTLEURDLMW-UHFFFAOYSA-N 0.000 description 1
- OVLQRNNIJPFQFB-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2ccc(C(F)(F)F)cc2)C(c2ccc(C(F)(F)F)cc2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2ccc(C(F)(F)F)cc2)C(c2ccc(C(F)(F)F)cc2)=C(C(F)(F)F)C1=C(C#N)C#N OVLQRNNIJPFQFB-UHFFFAOYSA-N 0.000 description 1
- UETJEXAREFDQMY-UHFFFAOYSA-N N#CC(C#N)=C(C(C(F)(F)F)=C1c2ccc[s]2)C(c2ccc[s]2)=C(C(F)(F)F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(C(F)(F)F)=C1c2ccc[s]2)C(c2ccc[s]2)=C(C(F)(F)F)C1=C(C#N)C#N UETJEXAREFDQMY-UHFFFAOYSA-N 0.000 description 1
- RLJIXNXYMBHXNS-UHFFFAOYSA-N N#CC(C#N)=C(C(c(c(F)c(c(F)c1F)F)c1F)=C(C1=C(C#N)C#N)F)C(F)=C1c(c(F)c(c(F)c1F)F)c1F Chemical compound N#CC(C#N)=C(C(c(c(F)c(c(F)c1F)F)c1F)=C(C1=C(C#N)C#N)F)C(F)=C1c(c(F)c(c(F)c1F)F)c1F RLJIXNXYMBHXNS-UHFFFAOYSA-N 0.000 description 1
- DOGFOANOLZBVHL-UHFFFAOYSA-N N#CC(C#N)=C(C(c(cc1)cc(C(F)(F)F)c1F)=C1F)C(F)=C(c(cc2C(F)(F)F)ccc2F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(cc1)cc(C(F)(F)F)c1F)=C1F)C(F)=C(c(cc2C(F)(F)F)ccc2F)C1=C(C#N)C#N DOGFOANOLZBVHL-UHFFFAOYSA-N 0.000 description 1
- VWKNGMJRGARQBY-UHFFFAOYSA-N N#CC(C#N)=C(C(c(cc1)cc(F)c1C#N)=C1F)C(F)=C(c(cc2F)ccc2C#N)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(cc1)cc(F)c1C#N)=C1F)C(F)=C(c(cc2F)ccc2C#N)C1=C(C#N)C#N VWKNGMJRGARQBY-UHFFFAOYSA-N 0.000 description 1
- AEMTYLUGBQDQHG-UHFFFAOYSA-N N#CC(C#N)=C(C(c(cc1)ccc1C#N)=C1F)C(F)=C(c(cc2)ccc2C#N)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(cc1)ccc1C#N)=C1F)C(F)=C(c(cc2)ccc2C#N)C1=C(C#N)C#N AEMTYLUGBQDQHG-UHFFFAOYSA-N 0.000 description 1
- JEBZBUSCNNYDIW-UHFFFAOYSA-N N#CC(C#N)=C(C(c(cc1)ccc1F)=C1F)C(F)=C(c(cc2)ccc2F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(cc1)ccc1F)=C1F)C(F)=C(c(cc2)ccc2F)C1=C(C#N)C#N JEBZBUSCNNYDIW-UHFFFAOYSA-N 0.000 description 1
- HGJDWPICZKDJAZ-UHFFFAOYSA-N N#CC(C#N)=C(C(c(cc1)ncc1F)=C1F)C(F)=C(c(nc2)ccc2F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(cc1)ncc1F)=C1F)C(F)=C(c(nc2)ccc2F)C1=C(C#N)C#N HGJDWPICZKDJAZ-UHFFFAOYSA-N 0.000 description 1
- PLJYRHANHJEMTE-UHFFFAOYSA-N N#CC(C#N)=C(C(c(cc1F)cc(F)c1F)=C1F)C(F)=C(c(cc2F)cc(F)c2F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(cc1F)cc(F)c1F)=C1F)C(F)=C(c(cc2F)cc(F)c2F)C1=C(C#N)C#N PLJYRHANHJEMTE-UHFFFAOYSA-N 0.000 description 1
- HOQGBMGDDGQECA-UHFFFAOYSA-N N#CC(C#N)=C(C(c(cn1)cc(F)c1F)=C1F)C(F)=C(c(cc2F)cnc2F)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(cn1)cc(F)c1F)=C1F)C(F)=C(c(cc2F)cnc2F)C1=C(C#N)C#N HOQGBMGDDGQECA-UHFFFAOYSA-N 0.000 description 1
- BRMWBDDNMYVESZ-UHFFFAOYSA-N N#CC(C#N)=C(C(c(nc1)ccc1C#N)=C1F)C(F)=C(c(cc2)ncc2C#N)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c(nc1)ccc1C#N)=C1F)C(F)=C(c(cc2)ncc2C#N)C1=C(C#N)C#N BRMWBDDNMYVESZ-UHFFFAOYSA-N 0.000 description 1
- LLVPUWCNOPJCMX-UHFFFAOYSA-N N#CC(C#N)=C(C(c1cc(C#N)cc(C#N)c1)=C1F)C(F)=C(c2cc(C#N)cc(C#N)c2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1cc(C#N)cc(C#N)c1)=C1F)C(F)=C(c2cc(C#N)cc(C#N)c2)C1=C(C#N)C#N LLVPUWCNOPJCMX-UHFFFAOYSA-N 0.000 description 1
- KLUNVTJMFKIKKF-UHFFFAOYSA-N N#CC(C#N)=C(C(c1cc(C#N)cc(F)c1)=C1F)C(F)=C(c2cc(C#N)cc(F)c2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1cc(C#N)cc(F)c1)=C1F)C(F)=C(c2cc(C#N)cc(F)c2)C1=C(C#N)C#N KLUNVTJMFKIKKF-UHFFFAOYSA-N 0.000 description 1
- ZBIONFFFGATHDU-UHFFFAOYSA-N N#CC(C#N)=C(C(c1cc(C(F)(F)F)cc(F)c1)=C1F)C(F)=C(c2cc(F)cc(C(F)(F)F)c2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1cc(C(F)(F)F)cc(F)c1)=C1F)C(F)=C(c2cc(F)cc(C(F)(F)F)c2)C1=C(C#N)C#N ZBIONFFFGATHDU-UHFFFAOYSA-N 0.000 description 1
- WRSGPOLJMCKQEC-UHFFFAOYSA-N N#CC(C#N)=C(C(c1cc(F)c(C(F)(F)F)c(F)c1)=C1F)C(F)=C(c2cc(F)c(C(F)(F)F)c(F)c2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1cc(F)c(C(F)(F)F)c(F)c1)=C1F)C(F)=C(c2cc(F)c(C(F)(F)F)c(F)c2)C1=C(C#N)C#N WRSGPOLJMCKQEC-UHFFFAOYSA-N 0.000 description 1
- UBWDWUAXUKDBTL-UHFFFAOYSA-N N#CC(C#N)=C(C(c1cc(F)cc(F)c1)=C1F)C(F)=C(c2cc(F)cc(F)c2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1cc(F)cc(F)c1)=C1F)C(F)=C(c2cc(F)cc(F)c2)C1=C(C#N)C#N UBWDWUAXUKDBTL-UHFFFAOYSA-N 0.000 description 1
- AHBPCEUZDLTETH-UHFFFAOYSA-N N#CC(C#N)=C(C(c1cc(F)cnc1)=C1F)C(F)=C(c2cncc(F)c2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1cc(F)cnc1)=C1F)C(F)=C(c2cncc(F)c2)C1=C(C#N)C#N AHBPCEUZDLTETH-UHFFFAOYSA-N 0.000 description 1
- FBAUGQVUQRZSGT-UHFFFAOYSA-N N#CC(C#N)=C(C(c1cc2ccccc2cc1)=C1F)C(F)=C(c2cc3ccccc3cc2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1cc2ccccc2cc1)=C1F)C(F)=C(c2cc3ccccc3cc2)C1=C(C#N)C#N FBAUGQVUQRZSGT-UHFFFAOYSA-N 0.000 description 1
- NRJDIQQPXGNSBF-UHFFFAOYSA-N N#CC(C#N)=C(C(c1ccc(C(F)(F)F)c(F)c1)=C1F)C(F)=C(c2cc(F)c(C(F)(F)F)cc2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1ccc(C(F)(F)F)c(F)c1)=C1F)C(F)=C(c2cc(F)c(C(F)(F)F)cc2)C1=C(C#N)C#N NRJDIQQPXGNSBF-UHFFFAOYSA-N 0.000 description 1
- NSHOMRAUNJLHFB-UHFFFAOYSA-N N#CC(C#N)=C(C(c1ccc[s]1)=C1F)C(F)=C(c2ccc[s]2)C1=C(C#N)C#N Chemical compound N#CC(C#N)=C(C(c1ccc[s]1)=C1F)C(F)=C(c2ccc[s]2)C1=C(C#N)C#N NSHOMRAUNJLHFB-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H01L51/0072—
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent materials, e.g. electroluminescent or chemiluminescent
- C09K11/06—Luminescent materials, e.g. electroluminescent or chemiluminescent containing organic luminescent materials
-
- H01L51/0059—
-
- H01L51/5012—
-
- H01L51/5096—
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/16—Electron transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/18—Carrier blocking layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/622—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing four rings, e.g. pyrene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6576—Polycyclic condensed heteroaromatic hydrocarbons comprising only sulfur in the heteroaromatic polycondensed ring system, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Physics & Mathematics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Organic Chemistry (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
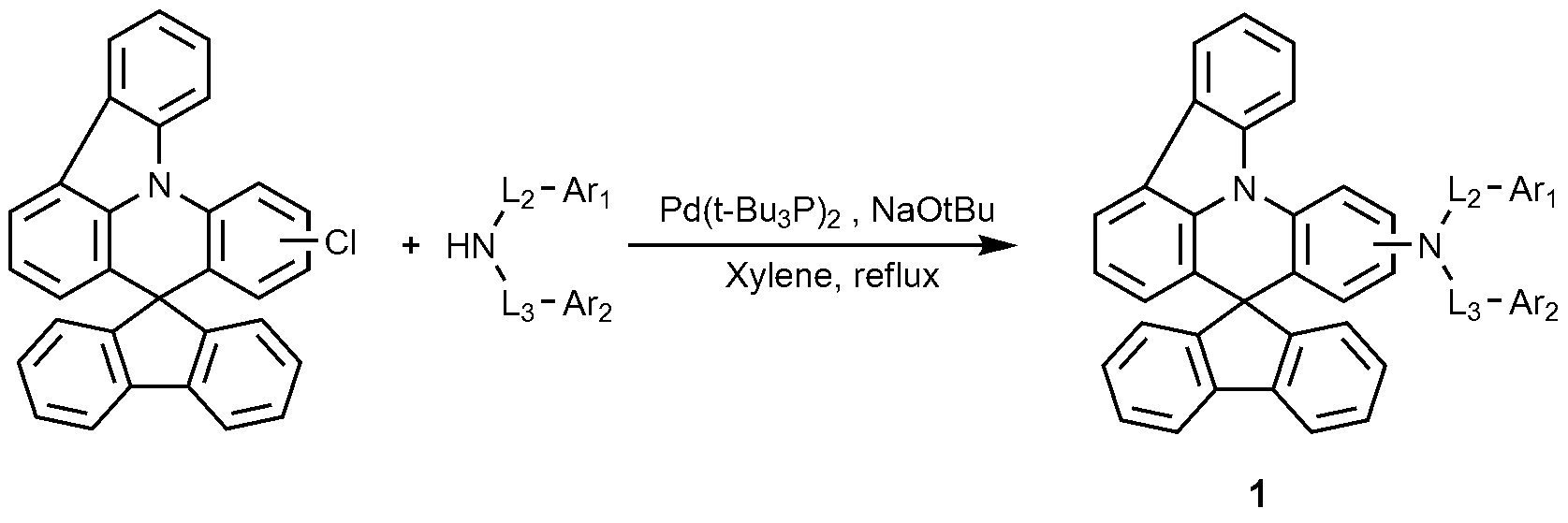
도 2는, 기판(1), 양극(2), 정공주입층(8), 정공수송층(3), 전자억제층(4), 발광층(5), 전자수송층(6), 및 음극(6)으로 이루어진 유기 발광 소자의 예를 도시한 것이다.
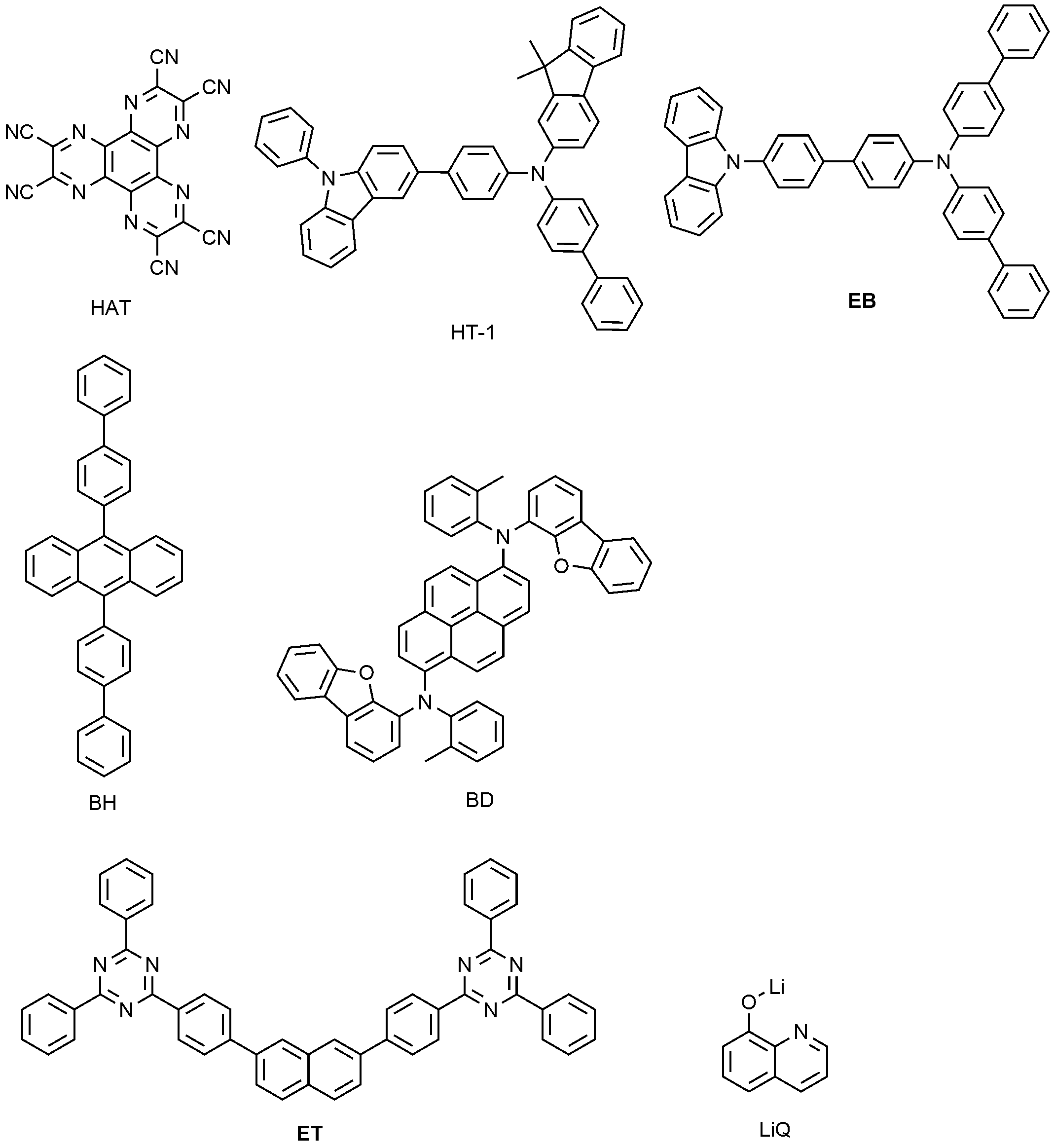
| 발광층의 호스트 |
전자억제층 | 전압 (V) |
휘도 (cd/A) |
T95 (hr) |
|
| 비교 실험예 1-1 | BH | EB | 4.33 | 5.93 | 270 |
| 비교 실험예 1-2 | BH | 1-1 | 4.24 | 6.12 | 280 |
| 비교 실험예 1-3 | BH | 1-3 | 4.15 | 6.16 | 280 |
| 비교 실험예 1-4 | BH | 1-6 | 4.17 | 6.04 | 285 |
| 비교 실험예 1-5 | BH | 1-8 | 4.23 | 6.17 | 290 |
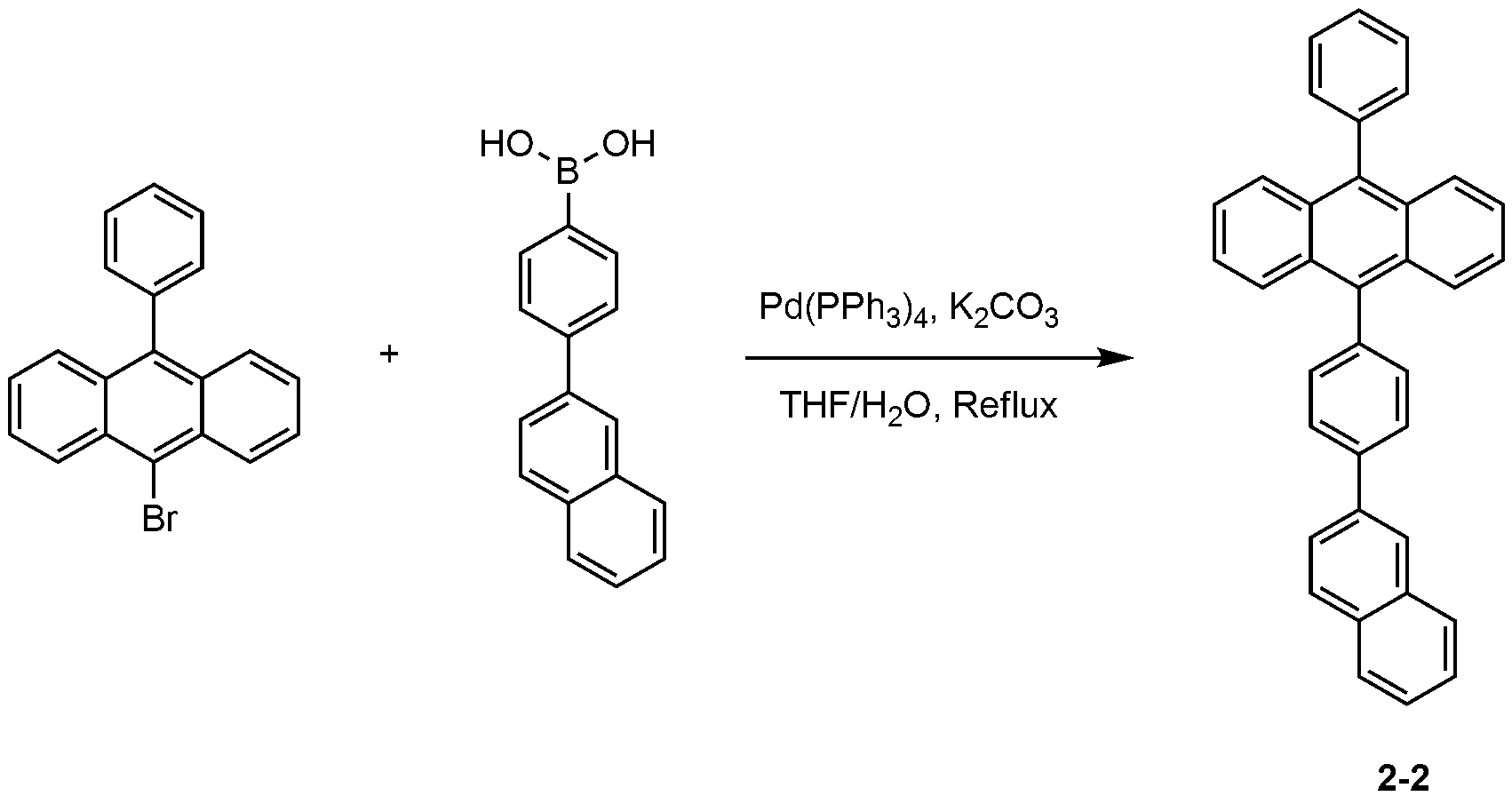
| 비교 실험예 1-6 | 2-2 | EB | 4.22 | 6.05 | 285 |
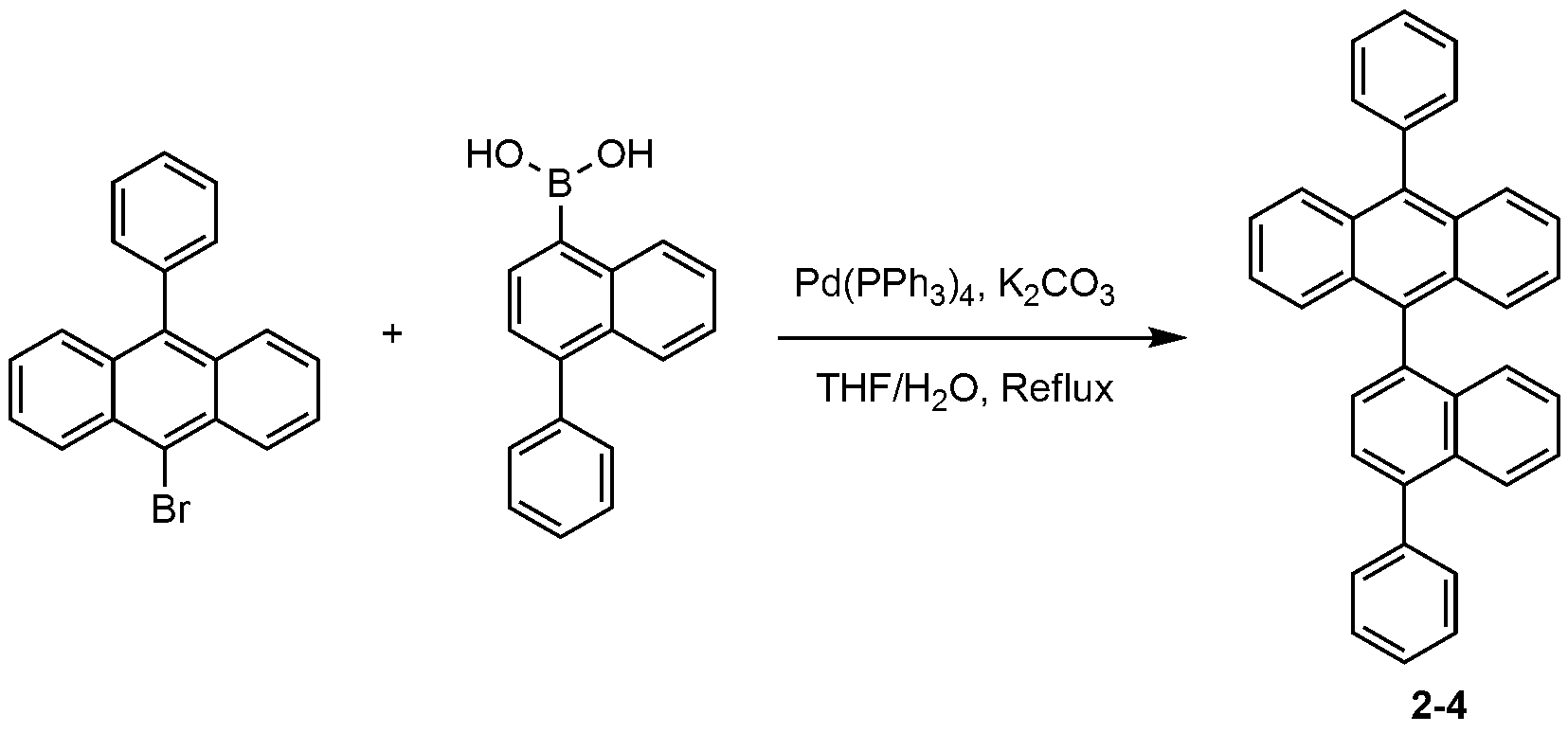
| 비교 실험예 1-7 | 2-4 | EB | 4.23 | 6.18 | 280 |
| 비교 실험예 1-8 | 2-5 | EB | 4.13 | 6.09 | 275 |
| 비교 실험예 1-9 | 2-7 | EB | 4.16 | 6.21 | 280 |
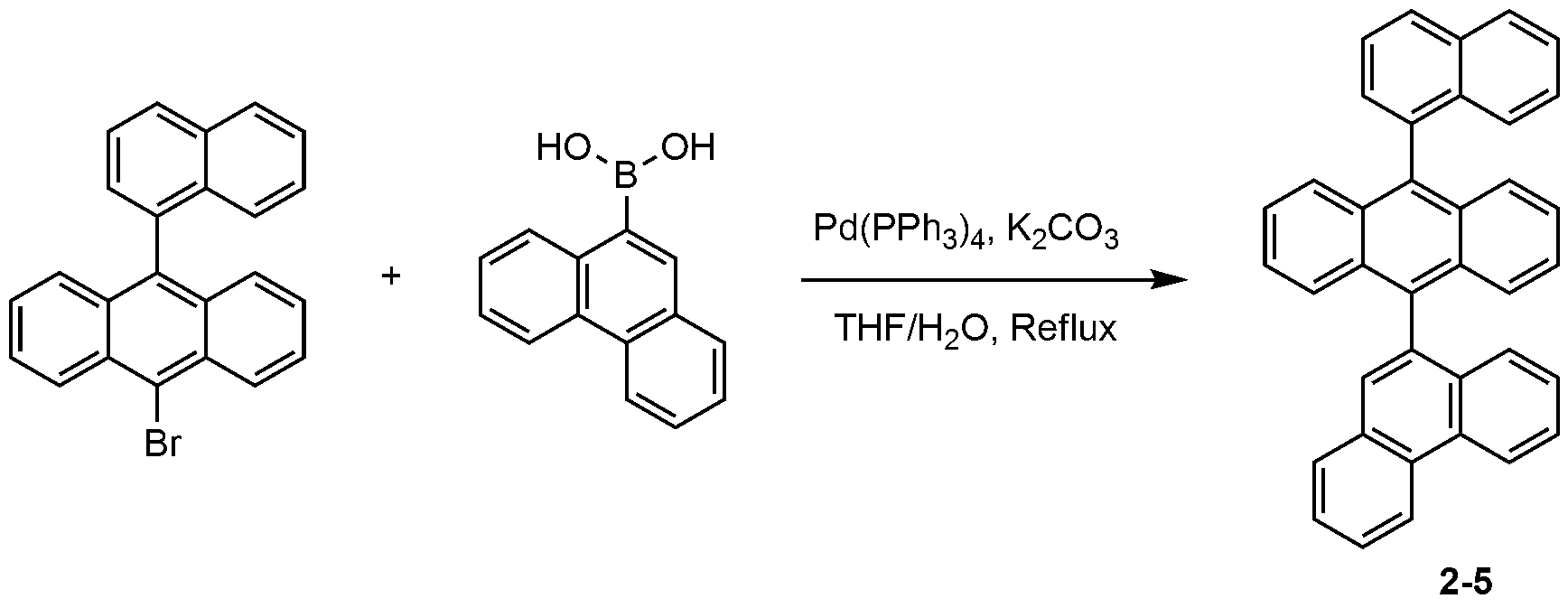
| 실험예 1-1 | 2-2 | 1-1 | 3.64 | 6.55 | 315 |
| 실험예 1-2 | 2-4 | 1-1 | 3.63 | 6.53 | 310 |
| 실험예 1-3 | 2-5 | 1-1 | 3.62 | 6.64 | 315 |
| 실험예 1-4 | 2-7 | 1-1 | 3.68 | 6.75 | 300 |
| 실험예 1-5 | 2-2 | 1-3 | 3.72 | 6.64 | 310 |
| 실험예 1-6 | 2-4 | 1-3 | 3.73 | 6.66 | 305 |
| 실험예 1-7 | 2-5 | 1-3 | 3.75 | 6.69 | 300 |
| 실험예 1-8 | 2-7 | 1-3 | 3.74 | 6.67 | 315 |
| 실험예 1-9 | 2-2 | 1-6 | 3.89 | 6.45 | 305 |
| 실험예 1-10 | 2-4 | 1-6 | 3.86 | 6.41 | 300 |
| 실험예 1-11 | 2-5 | 1-6 | 3.85 | 6.42 | 300 |
| 실험예 1-12 | 2-7 | 1-6 | 3.82 | 6.44 | 305 |
| 실험예 1-13 | 2-2 | 1-8 | 3.74 | 6.56 | 325 |
| 실험예 1-14 | 2-4 | 1-8 | 3.72 | 6.54 | 320 |
| 실험예 1-15 | 2-5 | 1-8 | 3.71 | 6.58 | 325 |
| 실험예 1-16 | 2-7 | 1-8 | 3.79 | 6.55 | 325 |
| 정공주입층 | 발광층의 호스트 |
전자억제층 | 전압 (V) |
휘도 (cd/A) |
T95 (hr) |
|
| 비교 실험예 1-1 | HAT | BH | EB | 4.33 | 5.93 | 270 |
| 비교 실험예 2-1 | HAT | BH | 1-2 | 4.24 | 6.12 | 280 |
| 비교 실험예 2-2 | HAT | BH | 1-4 | 4.15 | 6.16 | 280 |
| 비교 실험예 2-3 | HAT | BH | 1-5 | 4.17 | 6.04 | 285 |
| 비교 실험예 2-4 | HAT | BH | 1-7 | 4.23 | 6.17 | 290 |
| 비교 실험예 2-5 | HAT | 2-1 | EB | 4.22 | 6.05 | 285 |
| 비교 실험예 2-6 | HAT | 2-3 | EB | 4.23 | 6.18 | 280 |
| 비교 실험예 2-7 | HAT | 2-6 | EB | 4.13 | 6.09 | 275 |
| 비교 실험예 2-8 | HAT | 2-8 | EB | 4.16 | 6.21 | 280 |
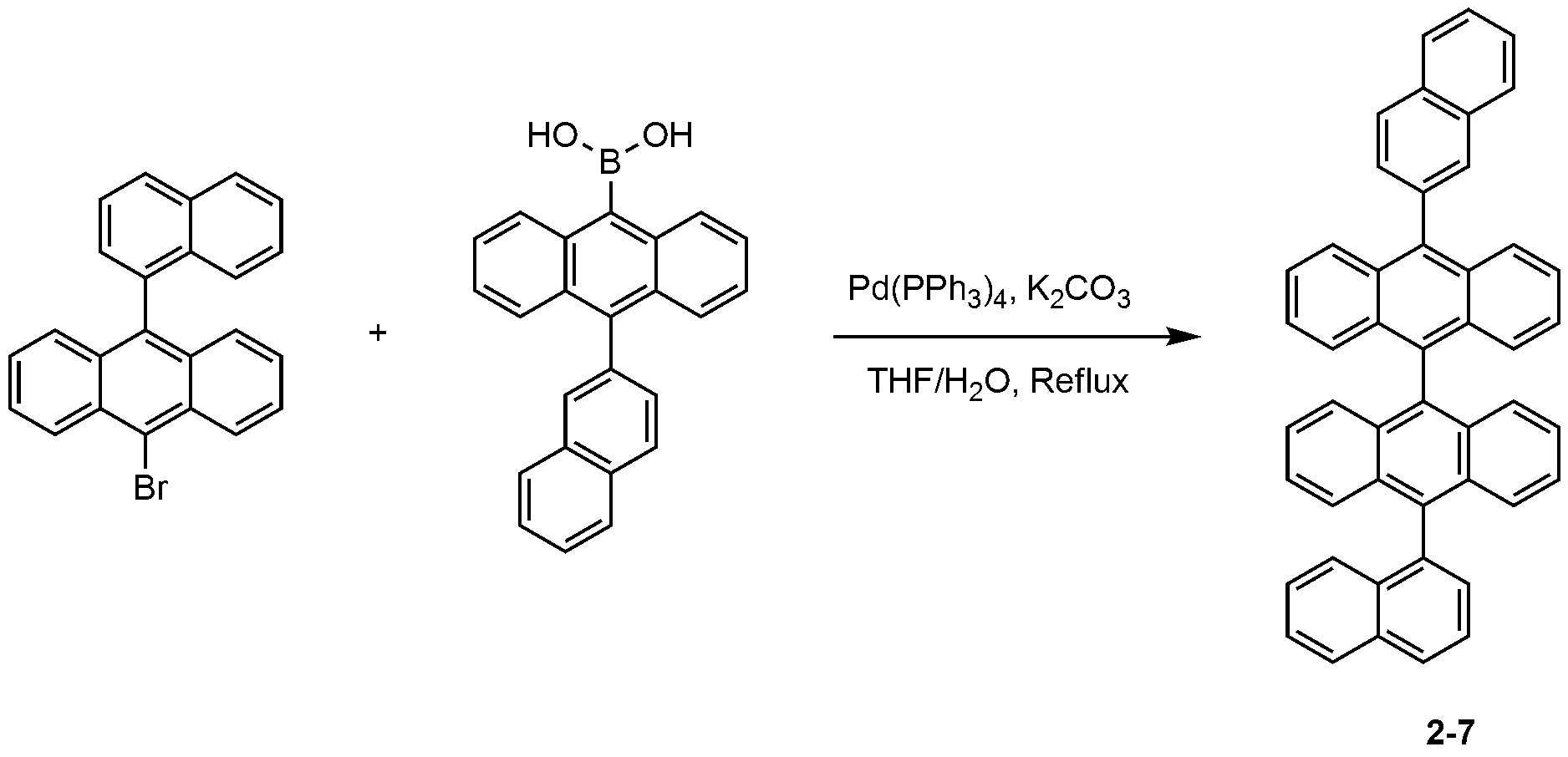
| 실험예 2-1 | HAT | 2-1 | 1-2 | 3.64 | 6.55 | 315 |
| 실험예 2-2 | HAT | 2-3 | 1-2 | 3.63 | 6.53 | 310 |
| 실험예 2-3 | HAT | 2-6 | 1-2 | 3.62 | 6.54 | 315 |
| 실험예 2-4 | HAT | 2-8 | 1-2 | 3.68 | 6.55 | 300 |
| 실험예 2-5 | 3-1 | 2-1 | 1-4 | 3.72 | 6.54 | 310 |
| 실험예 2-6 | 3-3 | 2-3 | 1-4 | 3.73 | 6.56 | 305 |
| 실험예 2-7 | 3-1 | 2-6 | 1-4 | 3.75 | 6.59 | 300 |
| 실험예 2-8 | 3-3 | 2-8 | 1-4 | 3.74 | 6.57 | 315 |
| 실험예 2-9 | 3-3 | 2-1 | 1-5 | 3.59 | 6.69 | 305 |
| 실험예 2-10 | 3-1 | 2-3 | 1-5 | 3.57 | 6.71 | 300 |
| 실험예 2-11 | 3-3 | 2-6 | 1-5 | 3.59 | 6.68 | 300 |
| 실험예 2-12 | 3-1 | 2-8 | 1-5 | 3.58 | 6.67 | 305 |
| 실험예 2-13 | 3-3 | 2-1 | 1-7 | 3.74 | 6.56 | 325 |
| 실험예 2-14 | 3-1 | 2-3 | 1-7 | 3.72 | 6.54 | 320 |
| 실험예 2-15 | 3-1 | 2-6 | 1-7 | 3.71 | 6.58 | 325 |
| 실험예 2-16 | 3-3 | 2-8 | 1-7 | 3.79 | 6.55 | 325 |
| 전자수송층 | 발광층의 호스트 |
전자억제층 | 전압 (V) |
휘도 (cd/A) |
T95 (hr) |
|
| 비교 실험예 1-1 | ET | BH | EB | 4.33 | 5.93 | 270 |
| 비교 실험예 1-2 | ET | BH | 1-1 | 4.24 | 6.12 | 280 |
| 비교 실험예 1-3 | ET | BH | 1-3 | 4.15 | 6.16 | 280 |
| 비교 실험예 1-4 | ET | BH | 1-6 | 4.17 | 6.04 | 285 |
| 비교 실험예 1-5 | ET | BH | 1-8 | 4.23 | 6.17 | 290 |
| 비교 실험예 1-6 | ET | 2-2 | EB | 4.22 | 6.05 | 285 |
| 비교 실험예 1-7 | ET | 2-4 | EB | 4.23 | 6.18 | 280 |
| 비교 실험예 1-8 | ET | 2-5 | EB | 4.13 | 6.09 | 275 |
| 비교 실험예 1-9 | ET | 2-7 | EB | 4.16 | 6.21 | 280 |
| 실험예 1-1 | ET | 2-2 | 1-1 | 3.64 | 6.55 | 315 |
| 실험예 1-2 | ET | 2-4 | 1-1 | 3.63 | 6.53 | 310 |
| 실험예 1-3 | ET | 2-5 | 1-1 | 3.62 | 6.64 | 315 |
| 실험예 3-1 | 4-1 | 2-7 | 1-1 | 3.68 | 6.75 | 345 |
| 실험예 3-2 | 4-4 | 2-2 | 1-3 | 3.72 | 6.64 | 350 |
| 실험예 3-3 | 4-1 | 2-4 | 1-3 | 3.73 | 6.66 | 345 |
| 실험예 3-4 | 4-4 | 2-5 | 1-3 | 3.75 | 6.69 | 345 |
| 실험예 3-5 | 4-4 | 2-7 | 1-3 | 3.74 | 6.67 | 350 |
| 실험예 3-6 | 4-4 | 2-2 | 1-6 | 3.89 | 6.45 | 340 |
| 실험예 3-7 | 4-1 | 2-4 | 1-6 | 3.86 | 6.41 | 345 |
| 실험예 3-8 | 4-1 | 2-5 | 1-6 | 3.85 | 6.42 | 340 |
| 실험예 3-9 | 4-1 | 2-7 | 1-8 | 3.82 | 6.44 | 340 |
| 실험예 3-10 | 4-1 | 2-2 | 1-8 | 3.74 | 6.56 | 360 |
| 실험예 3-11 | 4-4 | 2-4 | 1-8 | 3.72 | 6.54 | 365 |
| 실험예 3-12 | 4-1 | 2-5 | 1-8 | 3.71 | 6.58 | 365 |
| 실험예 3-13 | 4-4 | 2-7 | 1-7 | 3.79 | 6.55 | 360 |
3: 정공수송층 4: 전자억제층
5: 발광층 6: 전자수송층
7: 음극 8: 정공주입층
Claims (13)
- 양극,
음극,
상기 양극과 음극 사이에 발광층, 및
상기 양극과 발광층 사이에 전자억제층을 포함하고,
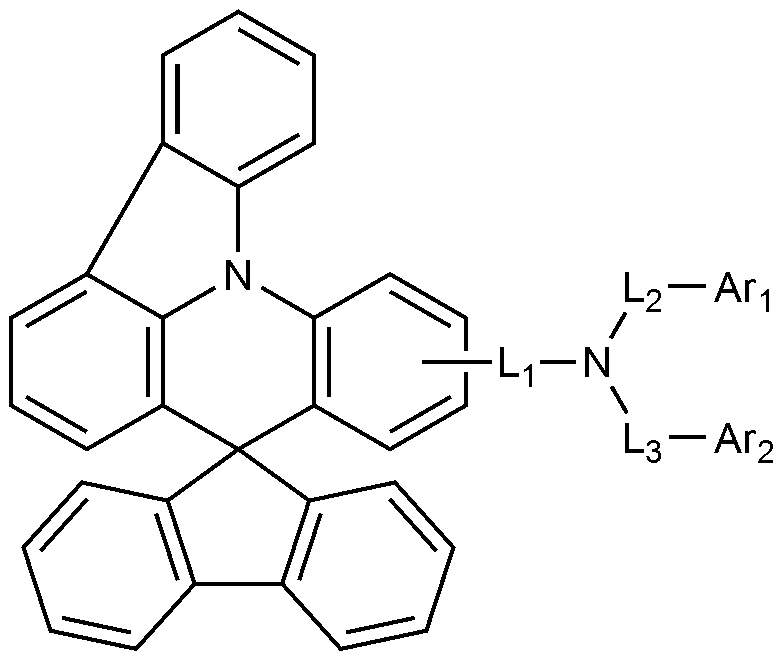
상기 전자억제층은 하기 화학식 1로 표시되는 화합물을 포함하고,
상기 발광층은 하기 화학식 2로 표시되는 화합물을 포함하는,
유기 발광 소자:
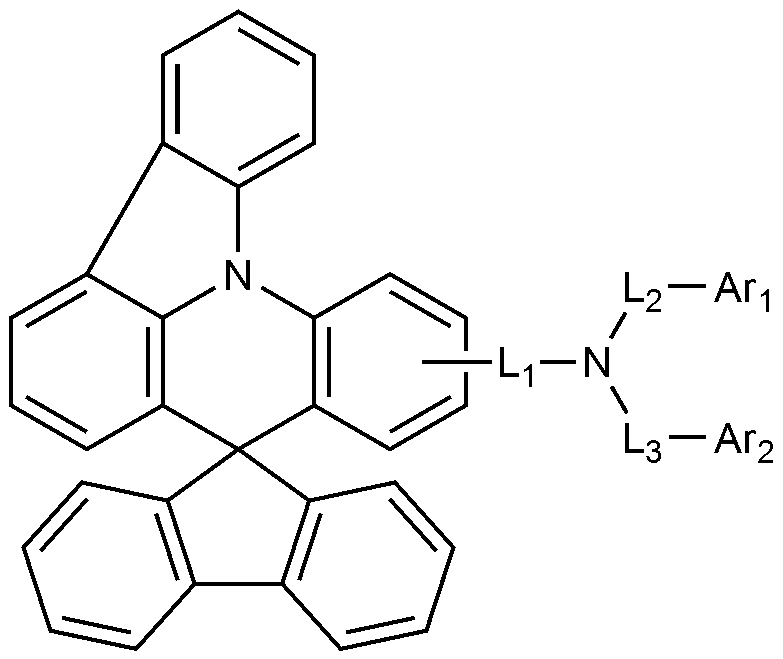
[화학식 1]
상기 화학식 1에서,
Ar1 및 Ar2는 각각 독립적으로 치환 또는 비치환된 C6-18 아릴; 또는 치환 또는 비치환된 O 및 S로 구성되는 군으로부터 선택되는 헤테로원자를 1개 이상을 포함하는 C2-60 헤테로아릴렌이고,
L1, L2, 및 L3는 각각 독립적으로 결합; 치환 또는 비치환된 C6-60 아릴렌; 또는 치환 또는 비치환된 O, N, Si 및 S로 구성되는 군으로부터 선택되는 헤테로원자를 1개 이상을 포함하는 C2-60 헤테로아릴렌이고,
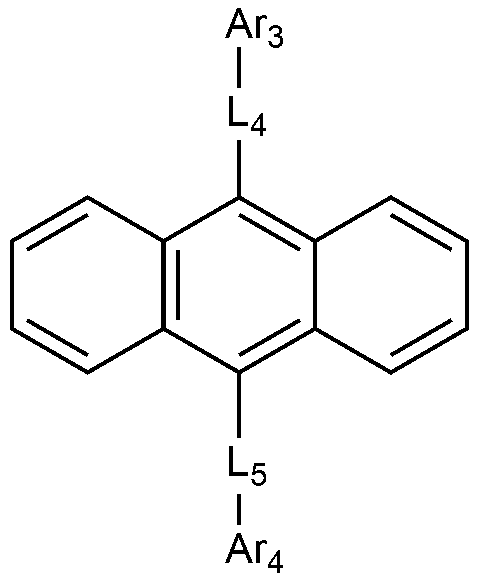
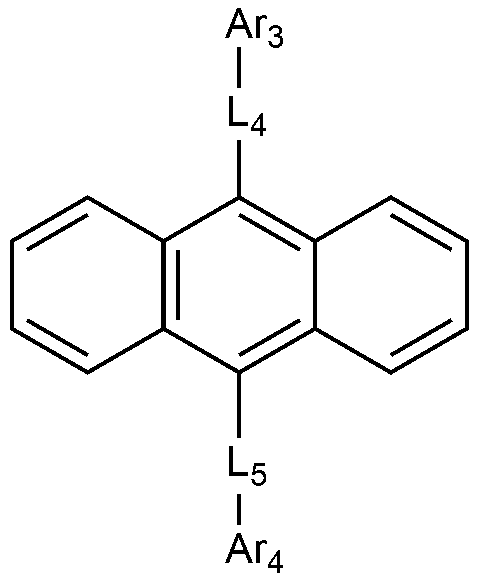
[화학식 2]
상기 화학식 2에서,
Ar3 및 Ar4는 각각 독립적으로 치환 또는 비치환된 C6-60 아릴; 또는 치환 또는 비치환된 O, N, Si 및 S로 구성되는 군으로부터 선택되는 헤테로원자를 1개 이상을 포함하는 C2-60 헤테로아릴렌이고,
L4 및 L5은 각각 독립적으로 결합; 치환 또는 비치환된 C6-60 아릴렌; 또는 치환 또는 비치환된 O, N, Si 및 S로 구성되는 군으로부터 선택되는 헤테로원자를 1개 이상을 포함하는 C2-60 헤테로아릴렌이다.
- 제1항에 있어서,
Ar1 및 Ar2는 각각 독립적으로 페닐, 비페닐린, 터페닐린, 나프틸, 페난쓰레닐, 트리페닐레닐, 디메틸플루오레닐, 디페닐플루오레닐, 스피로비플루오레닐, 디벤조퓨라닐, 또는 디벤조티오페닐이인,
유기 발광 소자.
- 제1항에 있어서,
L1, L2, 및 L3는 각각 독립적으로 결합, 페닐렌, 비페닐릴렌, 나프틸렌, 또는 디메틸플루오레닐렌인,
유기 발광 소자.
- 제1항에 있어서,
Ar3 및 Ar4는 각각 독립적으로 페닐, 비페닐린, 터페닐린, 나프틸, 페난쓰레닐, 트리페닐레닐, 디메틸플루오레닐, 또는 디페닐플루오레닐인,
유기 발광 소자.
- 제1항에 있어서,
L4 및 L5는 각각 독립적으로 결합, 페닐렌, 나프틸렌, 또는 안트라세닐렌인,
유기 발광 소자.
- 제1항 있어서,
상기 양극과 전자억제층 사이에 정공주입층 또는 전하발생층을 포함하고,
상기 정공주입층 또는 전하발생층은 하기 화학식 3으로 표시되는 화합물을 포함하는,
유기 발광 소자:
[화학식 3]
상기 화학식 3에서,
R1은 하기로 구성되는 군으로부터 선택되는 어느 하나이고,
R2는 수소, 중수소, 할로겐, 시아노, 또는 치환 또는 비치환된 C1-60 할로알킬이고,
Ar5는 치환 또는 비치환된 C6-60 아릴; 또는 치환 또는 비치환된 O, N, Si 및 S로 구성되는 군으로부터 선택되는 헤테로원자를 1개 이상을 포함하는 C2-60 헤테로아릴렌이다.
- 제8항에 있어서,
Ar5는 페닐, 나프틸, 피리딘, 퀴나졸리닐, 또는 티오페닐이고, 상기 Ar5는 비치환되거나 또는 할로겐, C1-6 할로알킬, C1-6 할로알콕시, 및 시아노로 구성되는 군으로부터 선택되는 1 내지 5개의 치환기로 치환된,
유기 발광 소자.
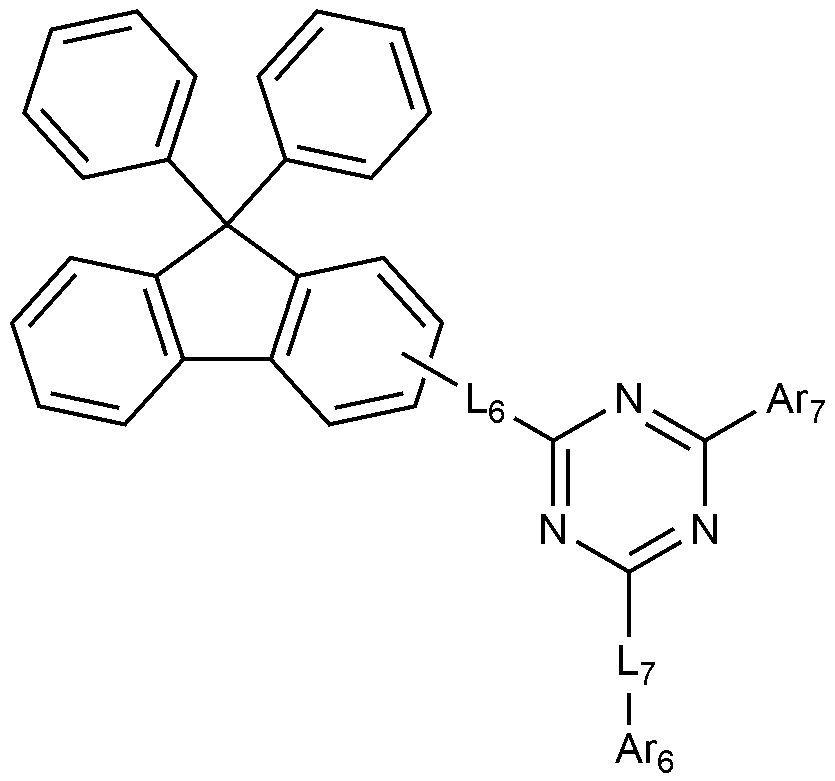
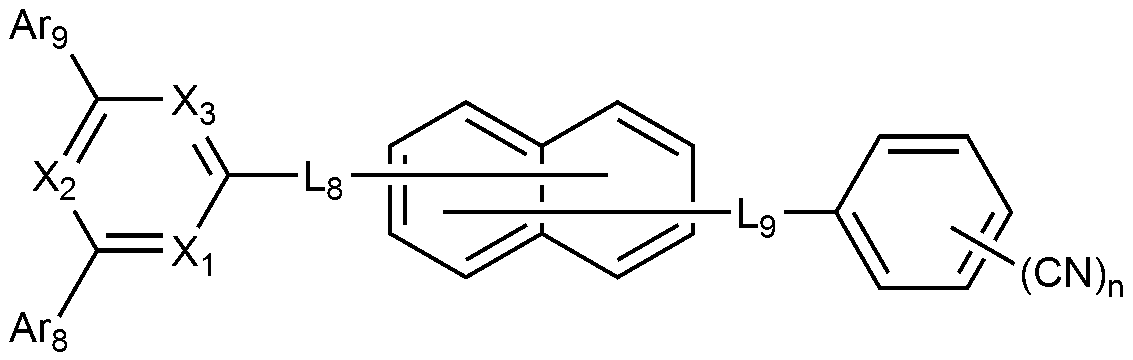
- 제1항에 있어서,
상기 발광층과 음극 사이에 전자수송층 또는 정공억제층을 포함하고,
상기 전자수송층 또는 정공억제층은 하기 화학식 4로 표시되는 화합물, 또는 하기 화학식 5로 표시되는 화합물을 포함하는,
유기 발광 소자:
[화학식 4]
상기 화학식 4에서,
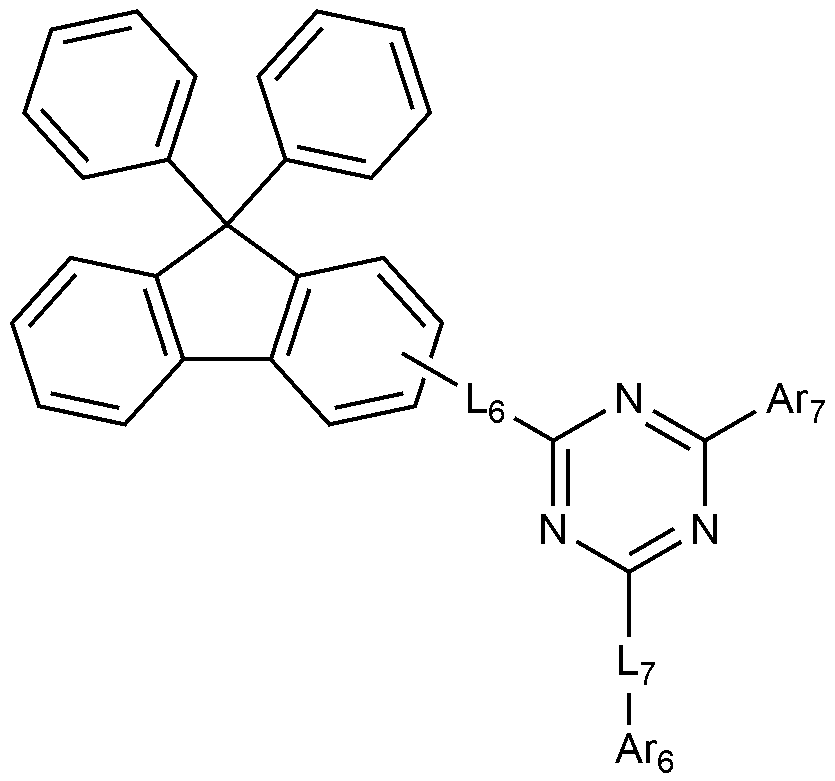
L6 및 L7은 각각 독립적으로 결합; 또는 치환 또는 비치환된 C6-60 아릴렌이고,
Ar6 및 Ar7은 각각 독립적으로 치환 또는 비치환된 C6-60 아릴; 또는 치환 또는 비치환된 O, N, Si 및 S로 구성되는 군으로부터 선택되는 헤테로원자를 1개 이상을 포함하는 C2-60 헤테로아릴이고,
[화학식 5]
상기 화학식 5에서,
X1 내지 X3은 각각 독립적으로 N, 또는 CH이고,
L8 및 L9은 각각 독립적으로, 결합; 또는 치환 또는 비치환된 C6-60 아릴렌이고,
Ar8 및 Ar9은 각각 독립적으로 치환 또는 비치환된 C6-60 아릴; 또는 치환 또는 비치환된 O, N, Si 및 S로 구성되는 군으로부터 선택되는 헤테로원자를 1개 이상을 포함하는 C2-60 헤테로아릴이고,
n은 1 내지 5의 정수이다.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020170024906 | 2017-02-24 | ||
| KR20170024906 | 2017-02-24 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20180098121A true KR20180098121A (ko) | 2018-09-03 |
| KR102109075B1 KR102109075B1 (ko) | 2020-05-11 |
Family
ID=63600830
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020170177273A Active KR102109075B1 (ko) | 2017-02-24 | 2017-12-21 | 유기 발광 소자 |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR102109075B1 (ko) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109273616A (zh) * | 2018-09-11 | 2019-01-25 | 长春海谱润斯科技有限公司 | 一种有机发光器件 |
| WO2020060280A1 (ko) * | 2018-09-20 | 2020-03-26 | 주식회사 엘지화학 | 유기 발광 소자 |
| WO2020096357A1 (ko) * | 2018-11-06 | 2020-05-14 | 주식회사 엘지화학 | 유기 발광 소자 |
| WO2020096358A1 (ko) * | 2018-11-06 | 2020-05-14 | 주식회사 엘지화학 | 유기 발광 소자 |
| KR20200100530A (ko) * | 2019-02-18 | 2020-08-26 | 주식회사 엘지화학 | 유기 발광 소자 |
| KR20210067848A (ko) * | 2019-11-29 | 2021-06-08 | 주식회사 엘지화학 | 유기 발광 소자 |
| KR20210073439A (ko) * | 2019-12-10 | 2021-06-18 | 주식회사 엘지화학 | 유기 발광 소자 |
| CN113307764A (zh) * | 2021-05-08 | 2021-08-27 | 烟台显华化工科技有限公司 | 一种化合物、电子传输材料、有机电致发光器件和显示装置 |
| CN113454803A (zh) * | 2019-05-10 | 2021-09-28 | 株式会社Lg化学 | 有机发光器件 |
| KR20210143552A (ko) * | 2020-05-20 | 2021-11-29 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기발광 소자 |
| JP2022509303A (ja) * | 2018-11-29 | 2022-01-20 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | 電子素子 |
| KR20220126083A (ko) * | 2021-03-08 | 2022-09-15 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물을 이용한 유기전기소자 및 그 전자 장치 |
| US20240237510A1 (en) * | 2021-04-06 | 2024-07-11 | Boe Technology Group Co., Ltd. | Organic light-emitting diode structure and display device |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20000051826A (ko) | 1999-01-27 | 2000-08-16 | 성재갑 | 신규한 착물 및 그의 제조 방법과 이를 이용한 유기 발광 소자 |
| KR20120015883A (ko) * | 2010-08-13 | 2012-02-22 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기 전자 소자 |
| KR20150064795A (ko) * | 2013-12-03 | 2015-06-12 | 엘지디스플레이 주식회사 | 유기 화합물 및 이를 포함하는 유기전계발광소자 |
| KR20160066669A (ko) * | 2014-12-02 | 2016-06-13 | 삼성디스플레이 주식회사 | 화합물 및 이를 포함한 유기 발광 소자 |
| KR20160111780A (ko) * | 2015-03-17 | 2016-09-27 | 주식회사 엘지화학 | 유기 발광 소자 |
| KR20160126862A (ko) * | 2015-04-24 | 2016-11-02 | 주식회사 엘지화학 | 유기 발광 소자 |
-
2017
- 2017-12-21 KR KR1020170177273A patent/KR102109075B1/ko active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20000051826A (ko) | 1999-01-27 | 2000-08-16 | 성재갑 | 신규한 착물 및 그의 제조 방법과 이를 이용한 유기 발광 소자 |
| KR20120015883A (ko) * | 2010-08-13 | 2012-02-22 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기 전자 소자 |
| KR20150064795A (ko) * | 2013-12-03 | 2015-06-12 | 엘지디스플레이 주식회사 | 유기 화합물 및 이를 포함하는 유기전계발광소자 |
| KR20160066669A (ko) * | 2014-12-02 | 2016-06-13 | 삼성디스플레이 주식회사 | 화합물 및 이를 포함한 유기 발광 소자 |
| KR20160111780A (ko) * | 2015-03-17 | 2016-09-27 | 주식회사 엘지화학 | 유기 발광 소자 |
| KR20160126862A (ko) * | 2015-04-24 | 2016-11-02 | 주식회사 엘지화학 | 유기 발광 소자 |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109273616A (zh) * | 2018-09-11 | 2019-01-25 | 长春海谱润斯科技有限公司 | 一种有机发光器件 |
| US11864460B2 (en) | 2018-09-20 | 2024-01-02 | Lg Chem, Ltd. | Organic light emitting device |
| WO2020060280A1 (ko) * | 2018-09-20 | 2020-03-26 | 주식회사 엘지화학 | 유기 발광 소자 |
| WO2020096357A1 (ko) * | 2018-11-06 | 2020-05-14 | 주식회사 엘지화학 | 유기 발광 소자 |
| WO2020096358A1 (ko) * | 2018-11-06 | 2020-05-14 | 주식회사 엘지화학 | 유기 발광 소자 |
| CN112689912A (zh) * | 2018-11-06 | 2021-04-20 | 株式会社Lg化学 | 有机发光器件 |
| US11937497B2 (en) | 2018-11-06 | 2024-03-19 | Lg Chem, Ltd. | Organic light-emitting device |
| JP2022509303A (ja) * | 2018-11-29 | 2022-01-20 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | 電子素子 |
| KR20200100530A (ko) * | 2019-02-18 | 2020-08-26 | 주식회사 엘지화학 | 유기 발광 소자 |
| CN113454803A (zh) * | 2019-05-10 | 2021-09-28 | 株式会社Lg化学 | 有机发光器件 |
| CN113454803B (zh) * | 2019-05-10 | 2024-05-14 | 株式会社Lg化学 | 有机发光器件 |
| KR20210067848A (ko) * | 2019-11-29 | 2021-06-08 | 주식회사 엘지화학 | 유기 발광 소자 |
| US11780856B2 (en) | 2019-11-29 | 2023-10-10 | Lg Chem, Ltd. | Compound and organic light-emitting device comprising same |
| KR20210073439A (ko) * | 2019-12-10 | 2021-06-18 | 주식회사 엘지화학 | 유기 발광 소자 |
| KR20210143552A (ko) * | 2020-05-20 | 2021-11-29 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기발광 소자 |
| KR20220126083A (ko) * | 2021-03-08 | 2022-09-15 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물을 이용한 유기전기소자 및 그 전자 장치 |
| US20240237510A1 (en) * | 2021-04-06 | 2024-07-11 | Boe Technology Group Co., Ltd. | Organic light-emitting diode structure and display device |
| CN113307764B (zh) * | 2021-05-08 | 2023-07-28 | 烟台显华化工科技有限公司 | 一种化合物、电子传输材料、有机电致发光器件和显示装置 |
| CN113307764A (zh) * | 2021-05-08 | 2021-08-27 | 烟台显华化工科技有限公司 | 一种化合物、电子传输材料、有机电致发光器件和显示装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102109075B1 (ko) | 2020-05-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102109075B1 (ko) | 유기 발광 소자 | |
| KR101959047B1 (ko) | 신규한 헤테로 고리 화합물 및 이를 이용한 유기발광 소자 | |
| KR20180111558A (ko) | 유기 발광 소자 | |
| KR102098221B1 (ko) | 유기 발광 소자 | |
| KR20190056336A (ko) | 유기 발광 소자 | |
| KR20180111482A (ko) | 유기발광 소자 | |
| KR101953766B1 (ko) | 신규한 유기 발광 재료 및 이를 포함하는 유기발광 소자 | |
| KR101937996B1 (ko) | 신규한 화합물 및 이를 이용한 유기발광 소자 | |
| JP2020507575A (ja) | 新規なヘテロ環式化合物およびこれを含む有機発光素子 | |
| KR102003351B1 (ko) | 신규한 헤테로 고리 화합물 및 이를 이용한 유기 발광 소자 | |
| KR20190053809A (ko) | 유기 발광 소자 | |
| KR20180127909A (ko) | 신규한 헤테로고리 화합물 및 이를 이용한 유기발광 소자 | |
| KR20190010499A (ko) | 신규한 헤테로 고리 화합물 및 이를 이용한 유기 발광 소자 | |
| KR20190010500A (ko) | 신규한 헤테로 고리 화합물 및 이를 이용한 유기 발광 소자 | |
| KR20180060991A (ko) | 유기 발광 소자 | |
| KR20180131482A (ko) | 신규한 헤테로 고리 화합물 및 이를 이용한 유기발광 소자 | |
| KR20180102937A (ko) | 유기 발광 소자 | |
| JP2020514335A (ja) | 新規な化合物およびこれを用いた有機発光素子 | |
| KR20180120569A (ko) | 신규한 화합물 및 이를 포함하는 유기발광 소자 | |
| KR20180125369A (ko) | 유기 발광 소자 | |
| KR20160143496A (ko) | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 | |
| KR20190053698A (ko) | 신규한 헤테로 고리 화합물 및 이를 이용한 유기발광 소자 | |
| KR20200068568A (ko) | 신규한 화합물 및 이를 포함하는 유기발광 소자 | |
| KR101834433B1 (ko) | 신규한 화합물 및 이를 포함하는 유기발광 소자 | |
| KR20190027708A (ko) | 신규한 페난트렌 화합물 및 이를 이용한 유기발광 소자 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0109 | Patent application |
St.27 status event code: A-0-1-A10-A12-nap-PA0109 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| D13-X000 | Search requested |
St.27 status event code: A-1-2-D10-D13-srh-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| D14-X000 | Search report completed |
St.27 status event code: A-1-2-D10-D14-srh-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U11-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| U11 | Full renewal or maintenance fee paid |
Free format text: ST27 STATUS EVENT CODE: A-4-4-U10-U11-OTH-PR1001 (AS PROVIDED BY THE NATIONAL OFFICE) Year of fee payment: 6 |