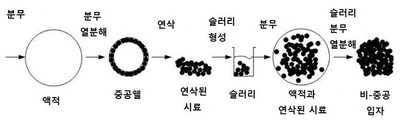
KR20150132375A - 비-중공, 다공성 입자의 제조를 위한 분무 열분해시 슬러리의 사용 방법 - Google Patents
비-중공, 다공성 입자의 제조를 위한 분무 열분해시 슬러리의 사용 방법 Download PDFInfo
- Publication number
- KR20150132375A KR20150132375A KR1020157029101A KR20157029101A KR20150132375A KR 20150132375 A KR20150132375 A KR 20150132375A KR 1020157029101 A KR1020157029101 A KR 1020157029101A KR 20157029101 A KR20157029101 A KR 20157029101A KR 20150132375 A KR20150132375 A KR 20150132375A
- Authority
- KR
- South Korea
- Prior art keywords
- particles
- solid phase
- metal oxide
- average
- product
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000002245 particle Substances 0.000 title claims abstract description 220
- 239000002002 slurry Substances 0.000 title claims abstract description 71
- 238000005118 spray pyrolysis Methods 0.000 title claims abstract description 47
- 238000000034 method Methods 0.000 title claims description 93
- 238000004519 manufacturing process Methods 0.000 title claims description 13
- 239000002243 precursor Substances 0.000 claims abstract description 85
- 239000007790 solid phase Substances 0.000 claims abstract description 69
- 150000004706 metal oxides Chemical class 0.000 claims abstract description 53
- 229910044991 metal oxide Inorganic materials 0.000 claims abstract description 52
- 239000000843 powder Substances 0.000 claims abstract description 51
- 150000001875 compounds Chemical class 0.000 claims abstract description 50
- 239000007788 liquid Substances 0.000 claims abstract description 25
- 229910052751 metal Inorganic materials 0.000 claims abstract description 13
- 238000010304 firing Methods 0.000 claims abstract description 6
- 239000000463 material Substances 0.000 claims description 63
- 239000000203 mixture Substances 0.000 claims description 63
- 230000008569 process Effects 0.000 claims description 40
- 239000011163 secondary particle Substances 0.000 claims description 22
- 239000002904 solvent Substances 0.000 claims description 20
- 239000000126 substance Substances 0.000 claims description 18
- 239000011164 primary particle Substances 0.000 claims description 16
- 229910052759 nickel Inorganic materials 0.000 claims description 14
- 239000000443 aerosol Substances 0.000 claims description 13
- 238000000354 decomposition reaction Methods 0.000 claims description 13
- 229910052748 manganese Inorganic materials 0.000 claims description 13
- 238000000137 annealing Methods 0.000 claims description 10
- 239000013078 crystal Substances 0.000 claims description 10
- 239000002184 metal Substances 0.000 claims description 10
- 230000015572 biosynthetic process Effects 0.000 claims description 9
- 238000001704 evaporation Methods 0.000 claims description 9
- 229910052804 chromium Inorganic materials 0.000 claims description 8
- 229910052802 copper Inorganic materials 0.000 claims description 8
- 230000003647 oxidation Effects 0.000 claims description 8
- 238000007254 oxidation reaction Methods 0.000 claims description 8
- 229910052782 aluminium Inorganic materials 0.000 claims description 7
- 229910052710 silicon Inorganic materials 0.000 claims description 7
- 229910052719 titanium Inorganic materials 0.000 claims description 7
- 230000008020 evaporation Effects 0.000 claims description 6
- 229910052742 iron Inorganic materials 0.000 claims description 6
- 229910052749 magnesium Inorganic materials 0.000 claims description 6
- 229910021645 metal ion Inorganic materials 0.000 claims description 6
- 229910052725 zinc Inorganic materials 0.000 claims description 6
- 229910052794 bromium Inorganic materials 0.000 claims description 5
- 239000002131 composite material Substances 0.000 claims description 5
- 238000009826 distribution Methods 0.000 claims description 5
- 229910052731 fluorine Inorganic materials 0.000 claims description 5
- 229910052740 iodine Inorganic materials 0.000 claims description 5
- 238000010899 nucleation Methods 0.000 claims description 5
- 229910052700 potassium Inorganic materials 0.000 claims description 5
- 229910052708 sodium Inorganic materials 0.000 claims description 5
- 229910013553 LiNO Inorganic materials 0.000 claims description 4
- 238000001354 calcination Methods 0.000 claims description 4
- 229910052801 chlorine Inorganic materials 0.000 claims description 4
- 238000001816 cooling Methods 0.000 claims description 4
- 230000006911 nucleation Effects 0.000 claims description 4
- 229910052720 vanadium Inorganic materials 0.000 claims description 4
- 229910052726 zirconium Inorganic materials 0.000 claims description 4
- 229910012851 LiCoO 2 Inorganic materials 0.000 claims description 3
- 229910014089 LiMn1/3Ni1/3Co1/3O2 Inorganic materials 0.000 claims description 3
- 229910002651 NO3 Inorganic materials 0.000 claims description 3
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 claims description 3
- 229910052733 gallium Inorganic materials 0.000 claims description 3
- 229910052739 hydrogen Inorganic materials 0.000 claims description 3
- 229910052738 indium Inorganic materials 0.000 claims description 3
- 238000002844 melting Methods 0.000 claims description 3
- 230000008018 melting Effects 0.000 claims description 3
- 229910001960 metal nitrate Inorganic materials 0.000 claims description 3
- 229910052758 niobium Inorganic materials 0.000 claims description 3
- 150000002823 nitrates Chemical class 0.000 claims description 3
- 229910052757 nitrogen Inorganic materials 0.000 claims description 3
- 229910052717 sulfur Inorganic materials 0.000 claims description 3
- 229910016087 LiMn0.5Ni0.5O2 Inorganic materials 0.000 claims description 2
- 150000003839 salts Chemical class 0.000 claims description 2
- 230000014509 gene expression Effects 0.000 claims 3
- 229910052732 germanium Inorganic materials 0.000 claims 2
- 150000004677 hydrates Chemical class 0.000 claims 2
- 229910052750 molybdenum Inorganic materials 0.000 claims 2
- 230000007547 defect Effects 0.000 claims 1
- 229910052596 spinel Inorganic materials 0.000 claims 1
- 239000011029 spinel Substances 0.000 claims 1
- 239000000047 product Substances 0.000 description 55
- 239000007787 solid Substances 0.000 description 31
- 239000000243 solution Substances 0.000 description 29
- 239000011572 manganese Substances 0.000 description 26
- 239000011148 porous material Substances 0.000 description 17
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 15
- 238000012360 testing method Methods 0.000 description 14
- 239000007789 gas Substances 0.000 description 13
- 238000000197 pyrolysis Methods 0.000 description 13
- 239000012159 carrier gas Substances 0.000 description 11
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 10
- 229910052744 lithium Inorganic materials 0.000 description 10
- 150000002894 organic compounds Chemical class 0.000 description 9
- 239000012467 final product Substances 0.000 description 8
- 239000012071 phase Substances 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 239000012530 fluid Substances 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- IIPYXGDZVMZOAP-UHFFFAOYSA-N lithium nitrate Chemical compound [Li+].[O-][N+]([O-])=O IIPYXGDZVMZOAP-UHFFFAOYSA-N 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 238000003786 synthesis reaction Methods 0.000 description 6
- 239000010936 titanium Substances 0.000 description 6
- 239000011800 void material Substances 0.000 description 6
- 239000000919 ceramic Substances 0.000 description 5
- 238000002485 combustion reaction Methods 0.000 description 5
- 239000010949 copper Substances 0.000 description 5
- 238000001035 drying Methods 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 5
- 238000001878 scanning electron micrograph Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 239000011651 chromium Substances 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 239000012705 liquid precursor Substances 0.000 description 4
- 238000001000 micrograph Methods 0.000 description 4
- 239000002114 nanocomposite Substances 0.000 description 4
- 239000002105 nanoparticle Substances 0.000 description 4
- 239000007921 spray Substances 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 229910015700 LiMn0.33Ni0.33Co0.33O2 Inorganic materials 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- UFMZWBIQTDUYBN-UHFFFAOYSA-N cobalt dinitrate Chemical compound [Co+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O UFMZWBIQTDUYBN-UHFFFAOYSA-N 0.000 description 3
- 229910001981 cobalt nitrate Inorganic materials 0.000 description 3
- 230000001351 cycling effect Effects 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 239000000446 fuel Substances 0.000 description 3
- 150000004820 halides Chemical class 0.000 description 3
- 239000011777 magnesium Substances 0.000 description 3
- MIVBAHRSNUNMPP-UHFFFAOYSA-N manganese(2+);dinitrate Chemical compound [Mn+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O MIVBAHRSNUNMPP-UHFFFAOYSA-N 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 229910003455 mixed metal oxide Inorganic materials 0.000 description 3
- KBJMLQFLOWQJNF-UHFFFAOYSA-N nickel(ii) nitrate Chemical compound [Ni+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O KBJMLQFLOWQJNF-UHFFFAOYSA-N 0.000 description 3
- 235000021317 phosphate Nutrition 0.000 description 3
- 239000001294 propane Substances 0.000 description 3
- 229920005989 resin Polymers 0.000 description 3
- 239000011347 resin Substances 0.000 description 3
- 239000011343 solid material Substances 0.000 description 3
- 239000011701 zinc Substances 0.000 description 3
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 229910015901 Bi-Sr-Ca-Cu-O Inorganic materials 0.000 description 2
- 229910020599 Co 3 O 4 Inorganic materials 0.000 description 2
- 229910021193 La 2 O 3 Inorganic materials 0.000 description 2
- 229910013716 LiNi Inorganic materials 0.000 description 2
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 2
- 229910006404 SnO 2 Inorganic materials 0.000 description 2
- 229910002367 SrTiO Inorganic materials 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 229910010413 TiO 2 Inorganic materials 0.000 description 2
- 229910009203 Y-Ba-Cu-O Inorganic materials 0.000 description 2
- 238000012387 aerosolization Methods 0.000 description 2
- 229910052786 argon Inorganic materials 0.000 description 2
- IWOUKMZUPDVPGQ-UHFFFAOYSA-N barium nitrate Chemical compound [Ba+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O IWOUKMZUPDVPGQ-UHFFFAOYSA-N 0.000 description 2
- VLYDPWNOCPZGEV-UHFFFAOYSA-M benzyl-dimethyl-[2-[2-[2-methyl-4-(2,4,4-trimethylpentan-2-yl)phenoxy]ethoxy]ethyl]azanium;chloride;hydrate Chemical compound O.[Cl-].CC1=CC(C(C)(C)CC(C)(C)C)=CC=C1OCCOCC[N+](C)(C)CC1=CC=CC=C1 VLYDPWNOCPZGEV-UHFFFAOYSA-M 0.000 description 2
- NLSCHDZTHVNDCP-UHFFFAOYSA-N caesium nitrate Chemical compound [Cs+].[O-][N+]([O-])=O NLSCHDZTHVNDCP-UHFFFAOYSA-N 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- ZCCIPPOKBCJFDN-UHFFFAOYSA-N calcium nitrate Chemical compound [Ca+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O ZCCIPPOKBCJFDN-UHFFFAOYSA-N 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 229910052729 chemical element Inorganic materials 0.000 description 2
- PHFQLYPOURZARY-UHFFFAOYSA-N chromium trinitrate Chemical compound [Cr+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O PHFQLYPOURZARY-UHFFFAOYSA-N 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 239000011258 core-shell material Substances 0.000 description 2
- 239000000428 dust Substances 0.000 description 2
- 238000000840 electrochemical analysis Methods 0.000 description 2
- 239000012634 fragment Substances 0.000 description 2
- 230000005484 gravity Effects 0.000 description 2
- 238000000227 grinding Methods 0.000 description 2
- 238000009413 insulation Methods 0.000 description 2
- 229910001416 lithium ion Inorganic materials 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 239000004570 mortar (masonry) Substances 0.000 description 2
- 229910052755 nonmetal Inorganic materials 0.000 description 2
- 230000001590 oxidative effect Effects 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 238000012856 packing Methods 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 2
- FGIUAXJPYTZDNR-UHFFFAOYSA-N potassium nitrate Chemical compound [K+].[O-][N+]([O-])=O FGIUAXJPYTZDNR-UHFFFAOYSA-N 0.000 description 2
- 230000000630 rising effect Effects 0.000 description 2
- 238000012216 screening Methods 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 239000010944 silver (metal) Substances 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- VWDWKYIASSYTQR-UHFFFAOYSA-N sodium nitrate Chemical compound [Na+].[O-][N+]([O-])=O VWDWKYIASSYTQR-UHFFFAOYSA-N 0.000 description 2
- 238000001694 spray drying Methods 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- 230000003068 static effect Effects 0.000 description 2
- 238000011144 upstream manufacturing Methods 0.000 description 2
- ONDPHDOFVYQSGI-UHFFFAOYSA-N zinc nitrate Chemical compound [Zn+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O ONDPHDOFVYQSGI-UHFFFAOYSA-N 0.000 description 2
- BNGXYYYYKUGPPF-UHFFFAOYSA-M (3-methylphenyl)methyl-triphenylphosphanium;chloride Chemical compound [Cl-].CC1=CC=CC(C[P+](C=2C=CC=CC=2)(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 BNGXYYYYKUGPPF-UHFFFAOYSA-M 0.000 description 1
- 239000006245 Carbon black Super-P Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 229910010707 LiFePO 4 Inorganic materials 0.000 description 1
- 229910015643 LiMn 2 O 4 Inorganic materials 0.000 description 1
- 229910014689 LiMnO Inorganic materials 0.000 description 1
- 229910002099 LiNi0.5Mn1.5O4 Inorganic materials 0.000 description 1
- 229910014411 LiNi1/2Mn1/2O2 Inorganic materials 0.000 description 1
- 229910014422 LiNi1/3Mn1/3Co1/3O2 Inorganic materials 0.000 description 1
- 229910013086 LiNiPO Inorganic materials 0.000 description 1
- 229910013870 LiPF 6 Inorganic materials 0.000 description 1
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- 239000002033 PVDF binder Substances 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 229910003077 Ti−O Inorganic materials 0.000 description 1
- MSDRXVQUKGAPDZ-UHFFFAOYSA-N [C].CN1C(CCC1)=O Chemical compound [C].CN1C(CCC1)=O MSDRXVQUKGAPDZ-UHFFFAOYSA-N 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 239000003570 air Substances 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000000712 assembly Effects 0.000 description 1
- 238000000429 assembly Methods 0.000 description 1
- 238000000889 atomisation Methods 0.000 description 1
- JRPBQTZRNDNNOP-UHFFFAOYSA-N barium titanate Chemical compound [Ba+2].[Ba+2].[O-][Ti]([O-])([O-])[O-] JRPBQTZRNDNNOP-UHFFFAOYSA-N 0.000 description 1
- 229910002113 barium titanate Inorganic materials 0.000 description 1
- XIEPJMXMMWZAAV-UHFFFAOYSA-N cadmium nitrate Inorganic materials [Cd+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O XIEPJMXMMWZAAV-UHFFFAOYSA-N 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000010406 cathode material Substances 0.000 description 1
- 239000006257 cathode slurry Substances 0.000 description 1
- 239000012671 ceramic insulating material Substances 0.000 description 1
- 150000004770 chalcogenides Chemical class 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- IVMYJDGYRUAWML-UHFFFAOYSA-N cobalt(ii) oxide Chemical class [Co]=O IVMYJDGYRUAWML-UHFFFAOYSA-N 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 230000003750 conditioning effect Effects 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 239000000498 cooling water Substances 0.000 description 1
- XTVVROIMIGLXTD-UHFFFAOYSA-N copper(II) nitrate Chemical compound [Cu+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O XTVVROIMIGLXTD-UHFFFAOYSA-N 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 239000002019 doping agent Substances 0.000 description 1
- 239000008151 electrolyte solution Substances 0.000 description 1
- 238000004146 energy storage Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000003822 epoxy resin Substances 0.000 description 1
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910021389 graphene Inorganic materials 0.000 description 1
- 239000011796 hollow space material Substances 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910001867 inorganic solvent Inorganic materials 0.000 description 1
- 239000003049 inorganic solvent Substances 0.000 description 1
- 230000002687 intercalation Effects 0.000 description 1
- 238000009830 intercalation Methods 0.000 description 1
- MVFCKEFYUDZOCX-UHFFFAOYSA-N iron(2+);dinitrate Chemical compound [Fe+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O MVFCKEFYUDZOCX-UHFFFAOYSA-N 0.000 description 1
- 238000010902 jet-milling Methods 0.000 description 1
- XIXADJRWDQXREU-UHFFFAOYSA-M lithium acetate Chemical compound [Li+].CC([O-])=O XIXADJRWDQXREU-UHFFFAOYSA-M 0.000 description 1
- FRMOHNDAXZZWQI-UHFFFAOYSA-N lithium manganese(2+) nickel(2+) oxygen(2-) Chemical class [O-2].[Mn+2].[Ni+2].[Li+] FRMOHNDAXZZWQI-UHFFFAOYSA-N 0.000 description 1
- 229910021437 lithium-transition metal oxide Inorganic materials 0.000 description 1
- OTCKOJUMXQWKQG-UHFFFAOYSA-L magnesium bromide Chemical compound [Mg+2].[Br-].[Br-] OTCKOJUMXQWKQG-UHFFFAOYSA-L 0.000 description 1
- 229910001623 magnesium bromide Inorganic materials 0.000 description 1
- 229940071125 manganese acetate Drugs 0.000 description 1
- UOGMEBQRZBEZQT-UHFFFAOYSA-L manganese(2+);diacetate Chemical compound [Mn+2].CC([O-])=O.CC([O-])=O UOGMEBQRZBEZQT-UHFFFAOYSA-L 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 239000011859 microparticle Substances 0.000 description 1
- 239000005543 nano-size silicon particle Substances 0.000 description 1
- 239000002086 nanomaterial Substances 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 238000013021 overheating Methods 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- JTJMJGYZQZDUJJ-UHFFFAOYSA-N phencyclidine Chemical class C1CCCCN1C1(C=2C=CC=CC=2)CCCCC1 JTJMJGYZQZDUJJ-UHFFFAOYSA-N 0.000 description 1
- NMHMNPHRMNGLLB-UHFFFAOYSA-N phloretic acid Chemical compound OC(=O)CCC1=CC=C(O)C=C1 NMHMNPHRMNGLLB-UHFFFAOYSA-N 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 238000005424 photoluminescence Methods 0.000 description 1
- 229920000647 polyepoxide Polymers 0.000 description 1
- 239000007774 positive electrode material Substances 0.000 description 1
- 235000010333 potassium nitrate Nutrition 0.000 description 1
- 239000004323 potassium nitrate Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000009877 rendering Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- RTHYXYOJKHGZJT-UHFFFAOYSA-N rubidium nitrate Inorganic materials [Rb+].[O-][N+]([O-])=O RTHYXYOJKHGZJT-UHFFFAOYSA-N 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000007613 slurry method Methods 0.000 description 1
- 235000010344 sodium nitrate Nutrition 0.000 description 1
- 239000004317 sodium nitrate Substances 0.000 description 1
- 239000012265 solid product Substances 0.000 description 1
- 238000000935 solvent evaporation Methods 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 238000005979 thermal decomposition reaction Methods 0.000 description 1
- HPGGPRDJHPYFRM-UHFFFAOYSA-J tin(iv) chloride Chemical compound Cl[Sn](Cl)(Cl)Cl HPGGPRDJHPYFRM-UHFFFAOYSA-J 0.000 description 1
- XJDNKRIXUMDJCW-UHFFFAOYSA-J titanium tetrachloride Chemical compound Cl[Ti](Cl)(Cl)Cl XJDNKRIXUMDJCW-UHFFFAOYSA-J 0.000 description 1
- KHAUBYTYGDOYRU-IRXASZMISA-N trospectomycin Chemical compound CN[C@H]([C@H]1O2)[C@@H](O)[C@@H](NC)[C@H](O)[C@H]1O[C@H]1[C@]2(O)C(=O)C[C@@H](CCCC)O1 KHAUBYTYGDOYRU-IRXASZMISA-N 0.000 description 1
- 239000007966 viscous suspension Substances 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/485—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of mixed oxides or hydroxides for inserting or intercalating light metals, e.g. LiTi2O4 or LiTi2OxFy
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2/00—Processes or devices for granulating materials, e.g. fertilisers in general; Rendering particulate materials free flowing in general, e.g. making them hydrophobic
- B01J2/02—Processes or devices for granulating materials, e.g. fertilisers in general; Rendering particulate materials free flowing in general, e.g. making them hydrophobic by dividing the liquid material into drops, e.g. by spraying, and solidifying the drops
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29B—PREPARATION OR PRETREATMENT OF THE MATERIAL TO BE SHAPED; MAKING GRANULES OR PREFORMS; RECOVERY OF PLASTICS OR OTHER CONSTITUENTS OF WASTE MATERIAL CONTAINING PLASTICS
- B29B9/00—Making granules
- B29B9/16—Auxiliary treatment of granules
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01D—COMPOUNDS OF ALKALI METALS, i.e. LITHIUM, SODIUM, POTASSIUM, RUBIDIUM, CAESIUM, OR FRANCIUM
- C01D15/00—Lithium compounds
- C01D15/02—Oxides; Hydroxides
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G1/00—Methods of preparing compounds of metals not covered by subclasses C01B, C01C, C01D, or C01F, in general
- C01G1/02—Oxides
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Complex oxides containing nickel and at least one other metal element
- C01G53/42—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2
- C01G53/44—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese
- C01G53/50—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese of the type (MnO2)n-, e.g. Li(NixMn1-x)O2 or Li(MyNixMn1-x-y)O2
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/5825—Oxygenated metallic salts or polyanionic structures, e.g. borates, phosphates, silicates, olivines
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G45/00—Compounds of manganese
- C01G45/12—Complex oxides containing manganese and at least one other metal element
- C01G45/1221—Manganates or manganites with trivalent manganese, tetravalent manganese or mixtures thereof
- C01G45/1228—Manganates or manganites with trivalent manganese, tetravalent manganese or mixtures thereof of the type (MnO2)-, e.g. LiMnO2 or Li(MxMn1-x)O2
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G45/00—Compounds of manganese
- C01G45/12—Complex oxides containing manganese and at least one other metal element
- C01G45/1221—Manganates or manganites with trivalent manganese, tetravalent manganese or mixtures thereof
- C01G45/125—Manganates or manganites with trivalent manganese, tetravalent manganese or mixtures thereof of the type (MnO3)n-, e.g. CaMnO3
- C01G45/1257—Manganates or manganites with trivalent manganese, tetravalent manganese or mixtures thereof of the type (MnO3)n-, e.g. CaMnO3 containing lithium, e.g. Li2MnO3 or Li2(MxMn1-x)O3
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G51/00—Compounds of cobalt
- C01G51/40—Complex oxides containing cobalt and at least one other metal element
- C01G51/42—Complex oxides containing cobalt and at least one other metal element containing alkali metals, e.g. LiCoO2
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G51/00—Compounds of cobalt
- C01G51/40—Complex oxides containing cobalt and at least one other metal element
- C01G51/42—Complex oxides containing cobalt and at least one other metal element containing alkali metals, e.g. LiCoO2
- C01G51/44—Complex oxides containing cobalt and at least one other metal element containing alkali metals, e.g. LiCoO2 containing manganese
- C01G51/50—Complex oxides containing cobalt and at least one other metal element containing alkali metals, e.g. LiCoO2 containing manganese of the type (MnO2)n-, e.g. Li(CoxMn1-x)O2 or Li(MyCoxMn1-x-y)O2
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G51/00—Compounds of cobalt
- C01G51/40—Complex oxides containing cobalt and at least one other metal element
- C01G51/42—Complex oxides containing cobalt and at least one other metal element containing alkali metals, e.g. LiCoO2
- C01G51/44—Complex oxides containing cobalt and at least one other metal element containing alkali metals, e.g. LiCoO2 containing manganese
- C01G51/56—Complex oxides containing cobalt and at least one other metal element containing alkali metals, e.g. LiCoO2 containing manganese of the type (MnO3)2-, e.g. Li2(CoxMn1-x)O3 or Li2(MyCoxMn1-x-y)O3
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Complex oxides containing nickel and at least one other metal element
- C01G53/42—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Complex oxides containing nickel and at least one other metal element
- C01G53/42—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2
- C01G53/44—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese
- C01G53/56—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese of the type (MnO3)2-, e.g. Li2(NixMn1-x)O3 or Li2(MyNixMn1-x-y)O3
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/30—Three-dimensional structures
- C01P2002/32—Three-dimensional structures spinel-type (AB2O4)
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
- C01P2002/52—Solid solutions containing elements as dopants
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/01—Particle morphology depicted by an image
- C01P2004/03—Particle morphology depicted by an image obtained by SEM
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/30—Particle morphology extending in three dimensions
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/30—Particle morphology extending in three dimensions
- C01P2004/45—Aggregated particles or particles with an intergrown morphology
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/61—Micrometer sized, i.e. from 1-100 micrometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/62—Submicrometer sized, i.e. from 0.1-1 micrometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/80—Particles consisting of a mixture of two or more inorganic phases
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/80—Particles consisting of a mixture of two or more inorganic phases
- C01P2004/82—Particles consisting of a mixture of two or more inorganic phases two phases having the same anion, e.g. both oxidic phases
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/11—Powder tap density
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/12—Surface area
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/16—Pore diameter
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/40—Electric properties
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Crystallography & Structural Chemistry (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
- Glanulating (AREA)
- Mechanical Engineering (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361780059P | 2013-03-13 | 2013-03-13 | |
| US61/780,059 | 2013-03-13 | ||
| PCT/US2014/026641 WO2014160445A1 (en) | 2013-03-13 | 2014-03-13 | Method for the use of slurries in spray pyrolysis for the production of non-hollow, porous particles |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20150132375A true KR20150132375A (ko) | 2015-11-25 |
Family
ID=51625427
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020157029101A Withdrawn KR20150132375A (ko) | 2013-03-13 | 2014-03-13 | 비-중공, 다공성 입자의 제조를 위한 분무 열분해시 슬러리의 사용 방법 |
Country Status (5)
| Country | Link |
|---|---|
| US (2) | US9748567B2 (enExample) |
| EP (1) | EP2969170A4 (enExample) |
| JP (1) | JP2016520411A (enExample) |
| KR (1) | KR20150132375A (enExample) |
| WO (1) | WO2014160445A1 (enExample) |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6043321B2 (ja) * | 2014-07-07 | 2016-12-14 | 大陽日酸株式会社 | リチウムイオン二次電池用正極材及びその製造方法 |
| WO2017027439A1 (en) * | 2015-08-07 | 2017-02-16 | Yangchuan Xing | Synthesis of deep eutectic solvent chemical precursors and their use in the production of metal oxides |
| FR3043572B1 (fr) | 2015-11-12 | 2020-05-15 | Pylote | Materiaux thermiquement isolants integrant des particules inorganiques spheriques et creuses |
| JP6871235B2 (ja) * | 2016-04-01 | 2021-05-12 | デンカ株式会社 | チタン酸バリウム質粉末及びその製造方法、用途 |
| KR102242799B1 (ko) | 2017-01-09 | 2021-04-21 | 에보니크 오퍼레이션즈 게엠베하 | 분무 열분해에 의해 금속 산화물을 제조하는 방법 |
| KR20190112809A (ko) * | 2017-02-09 | 2019-10-07 | 와커 헤미 아게 | 리튬 이온 배터리의 애노드 재료를 위한 규소 입자 |
| EP3424883A1 (de) | 2017-07-05 | 2019-01-09 | Evonik Degussa GmbH | Sprühverdampfung eines flüssigen rohstoffes zur herstellung von siliciumdioxid und metalloxiden |
| EP3495321A1 (de) | 2017-12-07 | 2019-06-12 | Evonik Degussa GmbH | Herstellung von pulverförmigen, porösen kristallinen metallsilikaten mittels flammensprühpyrolyse |
| CN110918047B (zh) * | 2018-09-20 | 2021-04-20 | 比亚迪股份有限公司 | 一种锂铝复合材料及其制备方法和从含锂卤水中富集锂离子的方法 |
| KR101993149B1 (ko) * | 2018-10-31 | 2019-09-30 | 주황윤 | 제강분진으로부터 황산아연분말을 제조하는 방법 |
| CN113165905B (zh) * | 2018-12-19 | 2023-12-29 | 托普索公司 | 锂正极活性材料 |
| US20210399298A1 (en) * | 2018-12-19 | 2021-12-23 | Haldor Topsøe A/S | Lithium positive electrode active material |
| GB201901061D0 (en) * | 2019-01-25 | 2019-03-13 | Ceramic Powder Tech As | Process |
| CN110935394B (zh) * | 2019-11-05 | 2021-07-30 | 南京清大迈特新材料有限公司 | 一种微纳粉体精细加工方法及装置 |
| CN112520792A (zh) * | 2021-01-30 | 2021-03-19 | 浙江博瓦德新材料科技有限公司 | 一种镍沉淀物回用制备氧化亚镍的方法 |
| CN116351419B (zh) * | 2023-03-02 | 2024-12-24 | 中科合成油技术股份有限公司 | 一种重质含碳原料浆态床加氢铁基催化剂及其制备方法 |
| WO2025096857A1 (en) * | 2023-11-03 | 2025-05-08 | Stratus Materials Inc. | Systems and methods for preparing electrode materials via aerosol decomposition |
| CN117987921A (zh) * | 2023-12-29 | 2024-05-07 | 华友新能源科技(衢州)有限公司 | 金属氧化物前驱体及其制备方法和应用 |
| CN119215904A (zh) * | 2024-09-05 | 2024-12-31 | 宁波中科远东催化工程技术有限公司 | 火焰喷雾热解制备绿色甲醇合成用催化剂的方法 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5128291A (en) * | 1990-12-11 | 1992-07-07 | Wax Michael J | Porous titania or zirconia spheres |
| US5958361A (en) * | 1993-03-19 | 1999-09-28 | Regents Of The University Of Michigan | Ultrafine metal oxide powders by flame spray pyrolysis |
| US6338809B1 (en) | 1997-02-24 | 2002-01-15 | Superior Micropowders Llc | Aerosol method and apparatus, particulate products, and electronic devices made therefrom |
| US6268014B1 (en) | 1997-10-02 | 2001-07-31 | Chris Eberspacher | Method for forming solar cell materials from particulars |
| JP2000340226A (ja) * | 1999-05-26 | 2000-12-08 | Kawasaki Steel Corp | リチウムマンガン複合酸化物粒子およびその製造方法 |
| US7704483B2 (en) | 2005-04-29 | 2010-04-27 | Cabot Corporation | High surface area tetragonal zirconia and processes for synthesizing same |
| US20120128577A1 (en) | 2010-10-06 | 2012-05-24 | Blue Juice, Inc. | Metal oxide synthesis |
| TW201311545A (zh) * | 2011-05-02 | 2013-03-16 | Univ Washington | 用於高能量鋰離子電池之介孔性正電極材料的噴霧熱製解合成法 |
| WO2012155196A1 (en) * | 2011-05-13 | 2012-11-22 | University Of Wollongong | Spray pyrolysis method for in situ production of graphene oxide based composites |
-
2014
- 2014-03-13 JP JP2016502203A patent/JP2016520411A/ja active Pending
- 2014-03-13 WO PCT/US2014/026641 patent/WO2014160445A1/en not_active Ceased
- 2014-03-13 EP EP14773759.7A patent/EP2969170A4/en not_active Withdrawn
- 2014-03-13 US US14/775,231 patent/US9748567B2/en active Active
- 2014-03-13 KR KR1020157029101A patent/KR20150132375A/ko not_active Withdrawn
-
2017
- 2017-07-24 US US15/658,014 patent/US10490814B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| JP2016520411A (ja) | 2016-07-14 |
| EP2969170A4 (en) | 2016-09-07 |
| US9748567B2 (en) | 2017-08-29 |
| EP2969170A1 (en) | 2016-01-20 |
| US20160049652A1 (en) | 2016-02-18 |
| WO2014160445A1 (en) | 2014-10-02 |
| US20170324088A1 (en) | 2017-11-09 |
| US10490814B2 (en) | 2019-11-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10490814B2 (en) | Method for the use of slurries in spray pyrolysis for the production of non-hollow, porous particles | |
| Oh et al. | Effect of calcination temperature on morphology, crystallinity and electrochemical properties of nano-crystalline metal oxides (Co3O4, CuO, and NiO) prepared via ultrasonic spray pyrolysis | |
| JP4894969B1 (ja) | ニッケルマンガン複合水酸化物粒子とその製造方法、非水系電解質二次電池用正極活物質とその製造方法、ならびに、非水系電解質二次電池 | |
| JP2009302044A (ja) | 無機物粒子の製造方法、及びそれを用いた二次電池正極並びに二次電池 | |
| TW201512079A (zh) | 用於製備電池單元材料的方法和系統 | |
| TW201311545A (zh) | 用於高能量鋰離子電池之介孔性正電極材料的噴霧熱製解合成法 | |
| WO2012131881A1 (ja) | ニッケルマンガン複合水酸化物粒子とその製造方法、非水系電解質二次電池用正極活物質とその製造方法、および非水系電解質二次電池 | |
| JP2020177860A (ja) | ニッケルマンガンコバルト含有複合水酸化物およびその製造方法、リチウムニッケルマンガンコバルト含有複合酸化物およびその製造方法、リチウムイオン二次電池用正極活物質およびその製造方法、並びに、リチウムイオン二次電池 | |
| JPWO2016068263A1 (ja) | ニッケル含有複合水酸化物とその製造方法、非水系電解質二次電池用正極活物質とその製造方法、および非水系電解質二次電池 | |
| JP2009062256A (ja) | 無機物粒子の製造方法 | |
| JP2005347134A (ja) | リチウムイオン二次電池用正極活物質の製造方法 | |
| JP7452569B2 (ja) | 非水系電解質二次電池用正極活物質 | |
| JP7452570B2 (ja) | 非水系電解質二次電池用正極活物質 | |
| US20250210702A1 (en) | Large-scale synthesis of powders of solid-state electrolyte material particles for solid-state batteries, systems and methods thereof | |
| JP2021005475A (ja) | リチウムイオン二次電池用正極活物質およびその製造方法、並びに、リチウムイオン二次電池 | |
| KR101510528B1 (ko) | 분무건조 공정에 의한 응집체 구조의 리튬 이차 전지용 전극활물질 분말의 제조방법 및 이로부터 제조된 전극활물질 분말 | |
| JP2002260656A (ja) | 電池材料の製造方法 | |
| JP2019021426A (ja) | 非水系電解質二次電池用正極活物質前駆体、非水系電解質二次電池用正極活物質、非水系電解質二次電池用正極活物質前駆体の製造方法、非水系電解質二次電池用正極活物質の製造方法 | |
| WO2020261962A1 (ja) | リチウムイオン二次電池用正極活物質およびその製造方法、並びに、リチウムイオン二次電池 | |
| JP7119302B2 (ja) | 非水系電解質二次電池用正極活物質前駆体、非水系電解質二次電池用正極活物質前駆体の製造方法、非水系電解質二次電池用正極活物質の製造方法 | |
| Fauteux et al. | Flexible synthesis of mixed metal oxides illustrated for LiMn2O4 and LiCoO2 | |
| JP4032784B2 (ja) | リチウム遷移金属複合酸化物の製造方法 | |
| US20240317605A1 (en) | Positive electrode active material for lithium ion secondary battery, and lithium ion secondary battery | |
| JPH10114528A (ja) | リチウムニッケル複合酸化物の製造方法 | |
| Liu et al. | Synthesizing high performance LNMO cathode materials with porous structure by manipulating reynolds number in a microreactor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20151013 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
Patent event date: 20170222 Comment text: Notification of Change of Applicant Patent event code: PN23011R01D |
|
| PC1203 | Withdrawal of no request for examination | ||
| WITN | Application deemed withdrawn, e.g. because no request for examination was filed or no examination fee was paid |