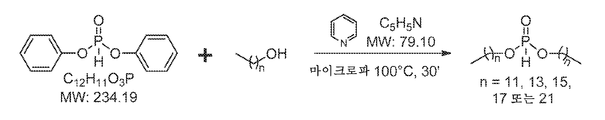
KR20140067070A - H-포스포네이트에 의한 나노입자 peg 개질 - Google Patents
H-포스포네이트에 의한 나노입자 peg 개질 Download PDFInfo
- Publication number
- KR20140067070A KR20140067070A KR1020147007934A KR20147007934A KR20140067070A KR 20140067070 A KR20140067070 A KR 20140067070A KR 1020147007934 A KR1020147007934 A KR 1020147007934A KR 20147007934 A KR20147007934 A KR 20147007934A KR 20140067070 A KR20140067070 A KR 20140067070A
- Authority
- KR
- South Korea
- Prior art keywords
- group
- agent
- compound
- peg
- stealth
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids [RP(=O)(OH)2]; Thiophosphonic acids ; [RP(=X1)(X2H)2(X1, X2 are each independently O, S or Se)]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/548—Phosphates or phosphonates, e.g. bone-seeking
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/554—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound the modifying agent being a steroid plant sterol, glycyrrhetic acid, enoxolone or bile acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/69—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit
- A61K47/6905—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a colloid or an emulsion
- A61K47/6911—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a colloid or an emulsion the form being a liposome
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids [RP(=O)(OH)2]; Thiophosphonic acids ; [RP(=X1)(X2H)2(X1, X2 are each independently O, S or Se)]
- C07F9/40—Esters thereof
- C07F9/4071—Esters thereof the ester moiety containing a substituent or a structure which is considered as characteristic
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids [RP(=O)(OH)2]; Thiophosphonic acids ; [RP(=X1)(X2H)2(X1, X2 are each independently O, S or Se)]
- C07F9/44—Amides thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J41/00—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring
- C07J41/0033—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring not covered by C07J41/0005
- C07J41/0055—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring not covered by C07J41/0005 the 17-beta position being substituted by an uninterrupted chain of at least three carbon atoms which may or may not be branched, e.g. cholane or cholestane derivatives, optionally cyclised, e.g. 17-beta-phenyl or 17-beta-furyl derivatives
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S977/00—Nanotechnology
- Y10S977/70—Nanostructure
- Y10S977/773—Nanoparticle, i.e. structure having three dimensions of 100 nm or less
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S977/00—Nanotechnology
- Y10S977/70—Nanostructure
- Y10S977/832—Nanostructure having specified property, e.g. lattice-constant, thermal expansion coefficient
- Y10S977/835—Chemical or nuclear reactivity/stability of composition or compound forming nanomaterial
- Y10S977/836—Chemical or nuclear reactivity/stability of composition or compound forming nanomaterial having biological reactive capability
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Botany (AREA)
- Dispersion Chemistry (AREA)
- Biophysics (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Polyethers (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161529694P | 2011-08-31 | 2011-08-31 | |
| US61/529,694 | 2011-08-31 | ||
| PCT/US2012/053197 WO2013033438A2 (en) | 2011-08-31 | 2012-08-30 | Nanoparticle peg modification with h-phosphonates |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20140067070A true KR20140067070A (ko) | 2014-06-03 |
Family
ID=46888663
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020147007934A Withdrawn KR20140067070A (ko) | 2011-08-31 | 2012-08-30 | H-포스포네이트에 의한 나노입자 peg 개질 |
Country Status (9)
| Country | Link |
|---|---|
| US (2) | US8889657B2 (enExample) |
| EP (1) | EP2750712A2 (enExample) |
| JP (1) | JP2014527071A (enExample) |
| KR (1) | KR20140067070A (enExample) |
| CN (1) | CN103930135A (enExample) |
| BR (1) | BR112014004544A2 (enExample) |
| CA (1) | CA2845845A1 (enExample) |
| IL (1) | IL231215A0 (enExample) |
| WO (1) | WO2013033438A2 (enExample) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20200078962A (ko) * | 2018-12-24 | 2020-07-02 | 연세대학교 산학협력단 | 야누스 펩타이드 덴드리머 및 이의 용도 |
Families Citing this family (109)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2807552A1 (en) | 2010-08-06 | 2012-02-09 | Moderna Therapeutics, Inc. | Engineered nucleic acids and methods of use thereof |
| EP3431485B2 (en) | 2010-10-01 | 2024-09-04 | ModernaTX, Inc. | Engineered nucleic acids and methods of use thereof |
| WO2012135805A2 (en) | 2011-03-31 | 2012-10-04 | modeRNA Therapeutics | Delivery and formulation of engineered nucleic acids |
| US9464124B2 (en) | 2011-09-12 | 2016-10-11 | Moderna Therapeutics, Inc. | Engineered nucleic acids and methods of use thereof |
| EP2763701B1 (en) | 2011-10-03 | 2018-12-19 | Moderna Therapeutics, Inc. | Modified nucleosides, nucleotides, and nucleic acids, and uses thereof |
| KR20140102759A (ko) | 2011-12-16 | 2014-08-22 | 모더나 세라퓨틱스, 인코포레이티드 | 변형된 뉴클레오사이드, 뉴클레오타이드 및 핵산 조성물 |
| US9878056B2 (en) | 2012-04-02 | 2018-01-30 | Modernatx, Inc. | Modified polynucleotides for the production of cosmetic proteins and peptides |
| US9572897B2 (en) | 2012-04-02 | 2017-02-21 | Modernatx, Inc. | Modified polynucleotides for the production of cytoplasmic and cytoskeletal proteins |
| DE18200782T1 (de) | 2012-04-02 | 2021-10-21 | Modernatx, Inc. | Modifizierte polynukleotide zur herstellung von proteinen im zusammenhang mit erkrankungen beim menschen |
| US9283287B2 (en) | 2012-04-02 | 2016-03-15 | Moderna Therapeutics, Inc. | Modified polynucleotides for the production of nuclear proteins |
| CA2892529C (en) | 2012-11-26 | 2023-04-25 | Moderna Therapeutics, Inc. | Terminally modified rna |
| WO2014152211A1 (en) | 2013-03-14 | 2014-09-25 | Moderna Therapeutics, Inc. | Formulation and delivery of modified nucleoside, nucleotide, and nucleic acid compositions |
| US8980864B2 (en) | 2013-03-15 | 2015-03-17 | Moderna Therapeutics, Inc. | Compositions and methods of altering cholesterol levels |
| EP3019559A4 (en) | 2013-08-22 | 2017-04-05 | Sony Corporation | Water soluble fluorescent or colored dyes and methods for their use |
| US20160194368A1 (en) | 2013-09-03 | 2016-07-07 | Moderna Therapeutics, Inc. | Circular polynucleotides |
| US10023626B2 (en) | 2013-09-30 | 2018-07-17 | Modernatx, Inc. | Polynucleotides encoding immune modulating polypeptides |
| EA201690675A1 (ru) | 2013-10-03 | 2016-08-31 | Модерна Терапьютикс, Инк. | Полинуклеотиды, кодирующие рецептор липопротеинов низкой плотности |
| WO2016011226A1 (en) | 2014-07-16 | 2016-01-21 | Moderna Therapeutics, Inc. | Chimeric polynucleotides |
| US20170210788A1 (en) | 2014-07-23 | 2017-07-27 | Modernatx, Inc. | Modified polynucleotides for the production of intrabodies |
| DK3350157T3 (da) | 2015-09-17 | 2022-02-14 | Modernatx Inc | Forbindelser og sammensætninger til intracellulær afgivelse af terapeutiske midler |
| WO2017067592A1 (en) | 2015-10-21 | 2017-04-27 | Biontech Ag | Cytotoxic immunostimulating particles and uses thereof |
| MA52645B1 (fr) | 2015-10-22 | 2022-06-30 | Modernatx Inc | Vaccins contre le virus respiratoire |
| MA46316A (fr) | 2015-10-22 | 2021-03-24 | Modernatx Inc | Vaccin contre le cytomégalovirus humain |
| KR20180096592A (ko) | 2015-10-22 | 2018-08-29 | 모더나티엑스, 인크. | 호흡기 세포융합 바이러스 백신 |
| MX2018004918A (es) | 2015-10-22 | 2019-04-01 | Modernatx Inc | Vacuna del virus herpes simple. |
| CA3003103A1 (en) | 2015-10-22 | 2017-04-27 | Modernatx, Inc. | Broad spectrum influenza virus vaccine |
| CN117731769A (zh) | 2015-10-22 | 2024-03-22 | 摩登纳特斯有限公司 | 用于水痘带状疱疹病毒(vzv)的核酸疫苗 |
| MD3386484T2 (ro) | 2015-12-10 | 2022-11-30 | Modernatx Inc | Compoziții și metode de livrare a unor agenți terapeutici |
| AU2016377681B2 (en) | 2015-12-22 | 2021-05-13 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of agents |
| EP4039699A1 (en) | 2015-12-23 | 2022-08-10 | ModernaTX, Inc. | Methods of using ox40 ligand encoding polynucleotides |
| US20190241658A1 (en) | 2016-01-10 | 2019-08-08 | Modernatx, Inc. | Therapeutic mRNAs encoding anti CTLA-4 antibodies |
| KR102530707B1 (ko) | 2016-04-01 | 2023-05-11 | 소니그룹주식회사 | 초휘도 이량체성 또는 중합체성 염료 |
| KR20180133890A (ko) | 2016-04-06 | 2018-12-17 | 소니 주식회사 | 스페이싱 링커 그룹을 포함하는 초고명도 이량체성 또는 중합체성 염료 |
| EP3455300A1 (en) | 2016-05-11 | 2019-03-20 | Sony Corporation | Ultra bright dimeric or polymeric dyes |
| US12018159B2 (en) | 2016-07-29 | 2024-06-25 | Sony Group Corporation | Ultra bright dimeric or polymeric dyes and methods for preparation of the same |
| US10072032B2 (en) * | 2016-09-19 | 2018-09-11 | Afton Chemical Corporation | AminoBisPhosphonate antiwear additives |
| US11583504B2 (en) | 2016-11-08 | 2023-02-21 | Modernatx, Inc. | Stabilized formulations of lipid nanoparticles |
| CA3055653A1 (en) | 2017-03-15 | 2018-09-20 | Modernatx, Inc. | Lipid nanoparticle formulation |
| AU2018234692B2 (en) | 2017-03-15 | 2022-06-02 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of therapeutic agents |
| ES2911186T3 (es) | 2017-03-15 | 2022-05-18 | Modernatx Inc | Formas cristalinas de aminolípidos |
| EP3625345B1 (en) | 2017-05-18 | 2023-05-24 | ModernaTX, Inc. | Modified messenger rna comprising functional rna elements |
| WO2018213731A1 (en) | 2017-05-18 | 2018-11-22 | Modernatx, Inc. | Polynucleotides encoding tethered interleukin-12 (il12) polypeptides and uses thereof |
| EP3638292A1 (en) | 2017-06-14 | 2020-04-22 | ModernaTX, Inc. | Polynucleotides encoding coagulation factor viii |
| WO2018232120A1 (en) | 2017-06-14 | 2018-12-20 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of agents |
| US11786607B2 (en) | 2017-06-15 | 2023-10-17 | Modernatx, Inc. | RNA formulations |
| MX2020002348A (es) | 2017-08-31 | 2020-10-08 | Modernatx Inc | Métodos de elaboración de nanopartículas lipídicas. |
| EP3691689A1 (en) | 2017-10-05 | 2020-08-12 | Sony Corporation | Programmable dendritic drugs |
| JP7551056B2 (ja) | 2017-10-05 | 2024-09-17 | ソニーグループ株式会社 | プログラマブルなポリマー薬物 |
| CN111836645A (zh) | 2017-11-16 | 2020-10-27 | 索尼公司 | 可编程的聚合药物 |
| WO2019104152A1 (en) | 2017-11-22 | 2019-05-31 | Modernatx, Inc. | Polynucleotides encoding ornithine transcarbamylase for the treatment of urea cycle disorders |
| EP3714045A1 (en) | 2017-11-22 | 2020-09-30 | Modernatx, Inc. | Polynucleotides encoding propionyl-coa carboxylase alpha and beta subunits for the treatment of propionic acidemia |
| US11939601B2 (en) | 2017-11-22 | 2024-03-26 | Modernatx, Inc. | Polynucleotides encoding phenylalanine hydroxylase for the treatment of phenylketonuria |
| US11802146B2 (en) | 2018-01-05 | 2023-10-31 | Modernatx, Inc. | Polynucleotides encoding anti-chikungunya virus antibodies |
| WO2019140301A1 (en) | 2018-01-12 | 2019-07-18 | Sony Corporation | Polymers with rigid spacing groups comprising biologically active compounds |
| EP4616912A3 (en) | 2018-01-12 | 2025-11-19 | Sony Group Corporation | Phosphoalkyl ribose polymers comprising biologically active compounds |
| US20190262793A1 (en) * | 2018-02-28 | 2019-08-29 | Southwest Research Institute | Polymer-Encapsulated Polyhemoglobin-Based Oxygen Carrier |
| KR102742386B1 (ko) | 2018-03-19 | 2024-12-16 | 소니그룹주식회사 | 형광 신호 향상을 위한 2가 금속의 사용 |
| WO2019182766A1 (en) | 2018-03-21 | 2019-09-26 | Sony Corporation | Polymeric tandem dyes with linker groups |
| WO2019200171A1 (en) | 2018-04-11 | 2019-10-17 | Modernatx, Inc. | Messenger rna comprising functional rna elements |
| EP3796893A1 (en) | 2018-05-23 | 2021-03-31 | Modernatx, Inc. | Delivery of dna |
| KR102854332B1 (ko) | 2018-06-27 | 2025-09-03 | 소니그룹주식회사 | 디옥시리보오스를 포함하는 링커 군을 갖는 중합체성 염료 |
| US20220184185A1 (en) | 2018-07-25 | 2022-06-16 | Modernatx, Inc. | Mrna based enzyme replacement therapy combined with a pharmacological chaperone for the treatment of lysosomal storage disorders |
| US20220110966A1 (en) | 2018-09-02 | 2022-04-14 | Modernatx, Inc. | Polynucleotides encoding very long-chain acyl-coa dehydrogenase for the treatment of very long-chain acyl-coa dehydrogenase deficiency |
| WO2020056161A1 (en) | 2018-09-12 | 2020-03-19 | University Of Florida Research Foundation, Inc. | Slow-cycling cell-rna based nanoparticle vaccine to treat cancer |
| WO2020056147A2 (en) | 2018-09-13 | 2020-03-19 | Modernatx, Inc. | Polynucleotides encoding glucose-6-phosphatase for the treatment of glycogen storage disease |
| MA53608A (fr) | 2018-09-13 | 2021-07-21 | Modernatx Inc | Polynucléotides codant pour les sous-unités e1-alpha, e1-beta et e2 du complexe alpha-cétoacide déshydrogénase à chaîne ramifiée pour le traitement de la leucinose |
| CA3112398A1 (en) | 2018-09-14 | 2020-03-19 | Modernatx, Inc. | Polynucleotides encoding uridine diphosphate glycosyltransferase 1 family, polypeptide a1 for the treatment of crigler-najjar syndrome |
| CA3113025A1 (en) | 2018-09-19 | 2020-03-26 | Modernatx, Inc. | Peg lipids and uses thereof |
| EP4509118A3 (en) | 2018-09-19 | 2025-05-14 | ModernaTX, Inc. | High-purity peg lipids and uses thereof |
| US12263248B2 (en) | 2018-09-19 | 2025-04-01 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of therapeutic agents |
| US12090235B2 (en) | 2018-09-20 | 2024-09-17 | Modernatx, Inc. | Preparation of lipid nanoparticles and methods of administration thereof |
| US20220152225A1 (en) | 2018-09-27 | 2022-05-19 | Modernatx, Inc. | Polynucleotides encoding arginase 1 for the treatment of arginase deficiency |
| WO2020069718A1 (en) * | 2018-10-01 | 2020-04-09 | Johannes Gutenberg-Universität Mainz | Rna particles comprising polysarcosine |
| SG11202101732WA (en) * | 2018-10-01 | 2021-03-30 | Univ Mainz Johannes Gutenberg | Rna particles comprising polysarcosine |
| US20220001026A1 (en) | 2018-11-08 | 2022-01-06 | Modernatx, Inc. | Use of mrna encoding ox40l to treat cancer in human patients |
| EP3965806A1 (en) | 2019-05-08 | 2022-03-16 | Modernatx, Inc. | Polynucleotides encoding methylmalonyl-coa mutase for the treatment of methylmalonic acidemia |
| EP3965797A1 (en) | 2019-05-08 | 2022-03-16 | AstraZeneca AB | Compositions for skin and wounds and methods of use thereof |
| MA56539A (fr) | 2019-06-24 | 2022-04-27 | Modernatx Inc | Arn messager résistant à l'endonucléase et utilisations correspondantes |
| US20220387628A1 (en) | 2019-06-24 | 2022-12-08 | Modernatx, Inc. | Messenger rna comprising functional rna elements and uses thereof |
| CA3154618A1 (en) | 2019-09-19 | 2021-03-25 | Modernatx, Inc. | Branched tail lipid compounds and compositions for intracellular delivery of therapeutic agents |
| KR102486779B1 (ko) | 2019-09-26 | 2023-01-12 | 소니그룹주식회사 | 링커 그룹을 갖는 중합체성 탠덤 염료 |
| US20230235298A1 (en) | 2020-06-01 | 2023-07-27 | Modernatx, Inc. | Phenylalanine hydroxylase variants and uses thereof |
| US20230272378A1 (en) | 2020-06-12 | 2023-08-31 | University Of Rochester | ENCODING AND EXPRESSION OF ACE-tRNAs |
| AU2021377895A1 (en) | 2020-11-13 | 2023-06-15 | Modernatx, Inc. | Polynucleotides encoding cystic fibrosis transmembrane conductance regulator for the treatment of cystic fibrosis |
| EP4256078A1 (en) | 2020-12-07 | 2023-10-11 | Sony Group Corporation | Spacing linker group design for brightness enhancement in dimeric or polymeric dyes |
| US11524023B2 (en) | 2021-02-19 | 2022-12-13 | Modernatx, Inc. | Lipid nanoparticle compositions and methods of formulating the same |
| WO2022204380A1 (en) | 2021-03-24 | 2022-09-29 | Modernatx, Inc. | Lipid nanoparticles containing polynucleotides encoding propionyl-coa carboxylase alpha and beta subunits and uses thereof |
| JP2024512026A (ja) | 2021-03-24 | 2024-03-18 | モデルナティエックス インコーポレイテッド | オルニチントランスカルバミラーゼ欠損症の治療を目的とした脂質ナノ粒子及びオルニチントランスカルバミラーゼをコードするポリヌクレオチド |
| WO2022204369A1 (en) | 2021-03-24 | 2022-09-29 | Modernatx, Inc. | Polynucleotides encoding methylmalonyl-coa mutase for the treatment of methylmalonic acidemia |
| WO2022204390A1 (en) | 2021-03-24 | 2022-09-29 | Modernatx, Inc. | Lipid nanoparticles containing polynucleotides encoding phenylalanine hydroxylase and uses thereof |
| US20240207374A1 (en) | 2021-03-24 | 2024-06-27 | Modernatx, Inc. | Lipid nanoparticles containing polynucleotides encoding glucose-6-phosphatase and uses thereof |
| EP4355882A2 (en) | 2021-06-15 | 2024-04-24 | Modernatx, Inc. | Engineered polynucleotides for cell-type or microenvironment-specific expression |
| US20240376445A1 (en) | 2021-06-22 | 2024-11-14 | Modernatx, Inc. | Polynucleotides encoding uridine diphosphate glycosyltransferase 1 family, polypeptide a1 for the treatment of crigler-najjar syndrome |
| US20250017867A1 (en) | 2021-10-01 | 2025-01-16 | Modernatx, Inc. | Polynucleotides encoding relaxin for the treatment of fibrosis and/or cardiovascular disease |
| AU2023215397A1 (en) | 2022-02-07 | 2024-08-22 | University Of Rochester | Optimized sequences for enhanced trna expression or/and nonsense mutation suppression. |
| US20250327041A1 (en) | 2022-02-24 | 2025-10-23 | Io Biotech Aps | Nucleotide delivery of cancer therapy |
| WO2023177904A1 (en) | 2022-03-18 | 2023-09-21 | Modernatx, Inc. | Sterile filtration of lipid nanoparticles and filtration analysis thereof for biological applications |
| WO2023183909A2 (en) | 2022-03-25 | 2023-09-28 | Modernatx, Inc. | Polynucleotides encoding fanconi anemia, complementation group proteins for the treatment of fanconi anemia |
| WO2023196399A1 (en) | 2022-04-06 | 2023-10-12 | Modernatx, Inc. | Lipid nanoparticles and polynucleotides encoding argininosuccinate lyase for the treatment of argininosuccinic aciduria |
| WO2023215498A2 (en) | 2022-05-05 | 2023-11-09 | Modernatx, Inc. | Compositions and methods for cd28 antagonism |
| TW202412818A (zh) | 2022-07-26 | 2024-04-01 | 美商現代公司 | 用於暫時控制表現之經工程化多核苷酸 |
| EP4577519A1 (en) | 2022-08-23 | 2025-07-02 | ModernaTX, Inc. | Methods for purification of ionizable lipids |
| WO2024097639A1 (en) | 2022-10-31 | 2024-05-10 | Modernatx, Inc. | Hsa-binding antibodies and binding proteins and uses thereof |
| WO2024118866A1 (en) | 2022-12-01 | 2024-06-06 | Modernatx, Inc. | Gpc3-specific antibodies, binding domains, and related proteins and uses thereof |
| WO2024197033A1 (en) | 2023-03-21 | 2024-09-26 | Modernatx, Inc. | Polynucleotides encoding relaxin for the treatment of heart failure |
| WO2024229321A1 (en) | 2023-05-03 | 2024-11-07 | Modernatx, Inc. | Polynucleotides encoding cystic fibrosis transmembrane conductance regulator for the treatment of cystic fibrosis |
| EP4520345A1 (en) | 2023-09-06 | 2025-03-12 | Myneo Nv | Product |
| WO2025072482A1 (en) | 2023-09-27 | 2025-04-03 | Modernatx, Inc. | Immunoglobulin a protease polypeptides, polynucleotides, and uses thereof |
| WO2025101685A1 (en) | 2023-11-09 | 2025-05-15 | University Of Rochester | Suppression of nonsense mutations using anticodon engineered (ace)-trnas |
Family Cites Families (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE514095C (de) * | 1927-07-24 | 1930-12-11 | Knoll Akt Ges Chemische Fabrik | Verfahren zur Herstellung von Estern der Sterine mit Sauerstoffsaeuren des Phosphors |
| CH336065A (de) * | 1954-03-05 | 1959-02-15 | Ciba Geigy | Verfahren zur Herstellung von neuen Stickstoff und Phosphor enthaltenden Verbindungen |
| GB1128086A (en) * | 1966-10-27 | 1968-09-25 | Pure Chem Ltd | Improvements relating to hydroxy-containing phosphonates and polyurethanes |
| US3974209A (en) * | 1971-08-04 | 1976-08-10 | Monsanto Company | Substituted tertiary amines and processes for preparing the same |
| DE3202127A1 (de) * | 1982-01-23 | 1983-07-28 | Agfa-Gevaert Ag, 5090 Leverkusen | Fotografisches material mit einer durch organische farbstoffe anfaerbbaren schicht |
| US4568432A (en) | 1984-12-28 | 1986-02-04 | Monsanto Company | Process for preparing glyphosate and glyphosate derivatives |
| EP0209848A3 (en) * | 1985-07-24 | 1988-07-27 | Merck & Co. Inc. | Enzyme inhibitors |
| US5567588A (en) | 1990-06-11 | 1996-10-22 | University Research Corporation | Systematic evolution of ligands by exponential enrichment: Solution SELEX |
| DE4408729A1 (de) * | 1994-03-15 | 1995-09-21 | Hoechst Ag | Nichtcyclische Chelatbildner auf Basis von Aminodialkylphosphoroxiden zur Herstellung von Technetium- oder Rheniumkomplexen |
| US6054608A (en) | 1997-05-05 | 2000-04-25 | Monsanto Company | Method for preparing formylphosphonic acid |
| ZA989394B (en) * | 1997-10-15 | 1999-04-15 | Monsanto Co | Selective functionalization of sodium glycinate |
| JP3799435B2 (ja) * | 1998-04-13 | 2006-07-19 | 三共有機合成株式会社 | 安定化された抗菌性樹脂組成物 |
| US7833992B2 (en) * | 2001-05-18 | 2010-11-16 | Merck Sharpe & Dohme | Conjugates and compositions for cellular delivery |
| DE10049074A1 (de) | 2000-10-02 | 2002-04-18 | Andreas Kage | Verfahren zur Selektion hochaffin an ein Target bindender Nukleinsäuren |
| WO2002062398A2 (en) * | 2001-01-08 | 2002-08-15 | Neorx Corporation | Radioactively labelled conjugates of phosphonates |
| DE10259935A1 (de) * | 2002-12-20 | 2004-07-01 | Bayer Ag | Herstellung und Verwendung von in-situ-modifizierten Nanopartikeln |
| EP1615639A2 (en) * | 2003-04-03 | 2006-01-18 | Semafore Pharmaceuticals, Inc. | Targeted bone marrow protection agents |
| KR20070087266A (ko) * | 2003-04-03 | 2007-08-28 | 세마포르 파머슈티컬즈, 아이엔씨. | 피아이-3 키나아제 억제제 프로드러그 |
| JP2010535701A (ja) * | 2003-04-25 | 2010-11-25 | ギリアード サイエンシーズ, インコーポレイテッド | 抗癌ホスホネートアナログ |
| DE10320071A1 (de) * | 2003-05-05 | 2004-12-02 | Basf Ag | Alkoxygruppen aufweisende Ester von Phosphor-Sauerstoff-Säuren und ihre Verwendung als Korrosionsinhibitoren |
| US7420000B2 (en) * | 2003-09-10 | 2008-09-02 | University Of Southern California | Amino phosphonate and amino bis-phosphonate derivatives |
| DE102004032781A1 (de) * | 2004-01-23 | 2005-08-11 | Mcs Micro Carrier Systems Gmbh | Bisphosphonsäurederivate |
| WO2006078646A2 (en) | 2005-01-18 | 2006-07-27 | Caltagirone Gaetano T | A class of supramolecular drug molecules and methods of identification and use thereof |
| US20090123507A1 (en) * | 2005-03-08 | 2009-05-14 | Reinhold Ohrlein | Metal Oxide Nanoparticles Coated With Specific N-Acylaminomethylene Phosphonates |
| JP5502490B2 (ja) * | 2007-11-19 | 2014-05-28 | 国立大学法人 東京大学 | 標的核酸を位置選択的に切断する方法 |
| WO2010013086A1 (en) * | 2008-08-01 | 2010-02-04 | Centre National De La Recherche Scientifique | Phosphorylated dendrimers as antiinflammatory drugs |
| CN105152939A (zh) * | 2008-11-10 | 2015-12-16 | 阿尔尼拉姆医药品有限公司 | 用于递送治疗剂的脂质和组合物 |
| JP5381269B2 (ja) * | 2009-04-15 | 2014-01-08 | コニカミノルタ株式会社 | 光学フィルム、光学フィルムの製造方法、偏光板及び液晶表示装置 |
| EP2533764A4 (en) * | 2010-02-08 | 2016-08-31 | Univ Nebraska | BIOMINERAL AND METAL BINDING LIPOSOME, THEIR SYNTHESIS AND METHOD FOR THE PRODUCTION THEREOF |
| WO2011101520A2 (es) * | 2010-02-18 | 2011-08-25 | Universidad De Alcalá De Henares (Uah) | Dendrimeros carbosilanos y su uso como antivirales |
| JO3131B1 (ar) * | 2010-04-27 | 2017-09-20 | Glaxosmithkline Llc | مركبات كيميائية |
| CN102093421B (zh) * | 2011-01-28 | 2014-07-02 | 北京康辰药业有限公司 | 一种含磷取代基的喹啉类化合物及其制备方法、以及含有该化合物的药物组合物及其应用 |
-
2012
- 2012-08-30 BR BR112014004544A patent/BR112014004544A2/pt not_active IP Right Cessation
- 2012-08-30 CA CA2845845A patent/CA2845845A1/en not_active Abandoned
- 2012-08-30 US US13/599,819 patent/US8889657B2/en not_active Expired - Fee Related
- 2012-08-30 CN CN201280053606.0A patent/CN103930135A/zh active Pending
- 2012-08-30 JP JP2014528610A patent/JP2014527071A/ja active Pending
- 2012-08-30 EP EP12762126.6A patent/EP2750712A2/en not_active Withdrawn
- 2012-08-30 KR KR1020147007934A patent/KR20140067070A/ko not_active Withdrawn
- 2012-08-30 WO PCT/US2012/053197 patent/WO2013033438A2/en not_active Ceased
-
2014
- 2014-02-27 IL IL231215A patent/IL231215A0/en unknown
- 2014-10-14 US US14/513,994 patent/US20150030541A1/en not_active Abandoned
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20200078962A (ko) * | 2018-12-24 | 2020-07-02 | 연세대학교 산학협력단 | 야누스 펩타이드 덴드리머 및 이의 용도 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2013033438A2 (en) | 2013-03-07 |
| CA2845845A1 (en) | 2013-03-07 |
| BR112014004544A2 (pt) | 2017-04-04 |
| US20130066086A1 (en) | 2013-03-14 |
| EP2750712A2 (en) | 2014-07-09 |
| CN103930135A (zh) | 2014-07-16 |
| IL231215A0 (en) | 2014-04-30 |
| US20150030541A1 (en) | 2015-01-29 |
| US8889657B2 (en) | 2014-11-18 |
| JP2014527071A (ja) | 2014-10-09 |
| WO2013033438A3 (en) | 2013-04-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20140067070A (ko) | H-포스포네이트에 의한 나노입자 peg 개질 | |
| JP5927194B2 (ja) | 治療ナノキャリアおよび/または診断ナノキャリアのターゲティングのためのアプタマーコンジュゲート | |
| Mitchell et al. | Incorporation of paramagnetic, fluorescent and PET/SPECT contrast agents into liposomes for multimodal imaging | |
| DK2501415T3 (en) | Liposomal nanoparticles of magnetic resonance imaging of tumors | |
| KR20140096080A (ko) | Mmp-표적 치료 및/또는 진단 나노담체 | |
| KR20140069269A (ko) | 상보성 올리고뉴클레오티드 링커를 사용한 표적화된 나노입자들의 원격 조립 | |
| KR20140064917A (ko) | H-포스포네이트-엔/-인 히드로포스포닐화 반응을 이용한 표적화된 나노입자의 원격 어셈블리 | |
| US20150017098A1 (en) | Carrier that targets fucosylated molecule-producing cells | |
| US20170165382A1 (en) | Nanocarriers for cancer treatment | |
| KR20240145481A (ko) | 방사선 민감화 조성물 및 이의 사용 방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20140326 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PC1203 | Withdrawal of no request for examination | ||
| WITN | Application deemed withdrawn, e.g. because no request for examination was filed or no examination fee was paid |