JP7280547B2 - 多孔質ガラス部材の製造方法 - Google Patents
多孔質ガラス部材の製造方法 Download PDFInfo
- Publication number
- JP7280547B2 JP7280547B2 JP2018095314A JP2018095314A JP7280547B2 JP 7280547 B2 JP7280547 B2 JP 7280547B2 JP 2018095314 A JP2018095314 A JP 2018095314A JP 2018095314 A JP2018095314 A JP 2018095314A JP 7280547 B2 JP7280547 B2 JP 7280547B2
- Authority
- JP
- Japan
- Prior art keywords
- porous glass
- base material
- glass member
- content
- glass
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000005373 porous glass Substances 0.000 title claims description 44
- 238000004519 manufacturing process Methods 0.000 title claims description 16
- 238000000034 method Methods 0.000 title description 9
- 239000011521 glass Substances 0.000 claims description 42
- 239000000463 material Substances 0.000 claims description 33
- 229910004298 SiO 2 Inorganic materials 0.000 claims description 20
- 239000002253 acid Substances 0.000 claims description 13
- 229910018072 Al 2 O 3 Inorganic materials 0.000 claims description 12
- 238000010438 heat treatment Methods 0.000 claims description 9
- 229910052796 boron Inorganic materials 0.000 claims description 5
- 229910052708 sodium Inorganic materials 0.000 claims description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 42
- 239000002585 base Substances 0.000 description 31
- 239000000377 silicon dioxide Substances 0.000 description 20
- 238000007654 immersion Methods 0.000 description 17
- 239000011734 sodium Substances 0.000 description 17
- JKWMSGQKBLHBQQ-UHFFFAOYSA-N diboron trioxide Chemical compound O=BOB=O JKWMSGQKBLHBQQ-UHFFFAOYSA-N 0.000 description 14
- GEIAQOFPUVMAGM-UHFFFAOYSA-N ZrO Inorganic materials [Zr]=O GEIAQOFPUVMAGM-UHFFFAOYSA-N 0.000 description 13
- 239000000084 colloidal system Substances 0.000 description 13
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 12
- 238000005191 phase separation Methods 0.000 description 12
- 229910052810 boron oxide Inorganic materials 0.000 description 10
- 239000000203 mixture Substances 0.000 description 9
- 239000011148 porous material Substances 0.000 description 9
- 229910052681 coesite Inorganic materials 0.000 description 8
- 229910052906 cristobalite Inorganic materials 0.000 description 8
- 235000012239 silicon dioxide Nutrition 0.000 description 8
- 229910052682 stishovite Inorganic materials 0.000 description 8
- 229910052905 tridymite Inorganic materials 0.000 description 8
- 230000000694 effects Effects 0.000 description 7
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 6
- 239000003513 alkali Substances 0.000 description 6
- 238000009826 distribution Methods 0.000 description 6
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 4
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 230000008602 contraction Effects 0.000 description 3
- 238000002844 melting Methods 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- KKCBUQHMOMHUOY-UHFFFAOYSA-N Na2O Inorganic materials [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 description 2
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 239000006066 glass batch Substances 0.000 description 2
- 230000036571 hydration Effects 0.000 description 2
- 238000006703 hydration reaction Methods 0.000 description 2
- 239000006060 molten glass Substances 0.000 description 2
- ZKATWMILCYLAPD-UHFFFAOYSA-N niobium pentoxide Chemical compound O=[Nb](=O)O[Nb](=O)=O ZKATWMILCYLAPD-UHFFFAOYSA-N 0.000 description 2
- 229910017604 nitric acid Inorganic materials 0.000 description 2
- 229910052697 platinum Inorganic materials 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- DLYUQMMRRRQYAE-UHFFFAOYSA-N tetraphosphorus decaoxide Chemical compound O1P(O2)(=O)OP3(=O)OP1(=O)OP2(=O)O3 DLYUQMMRRRQYAE-UHFFFAOYSA-N 0.000 description 2
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 2
- 229910015902 Bi 2 O 3 Inorganic materials 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 229910003069 TeO2 Inorganic materials 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000012670 alkaline solution Substances 0.000 description 1
- ADCOVFLJGNWWNZ-UHFFFAOYSA-N antimony trioxide Inorganic materials O=[Sb]O[Sb]=O ADCOVFLJGNWWNZ-UHFFFAOYSA-N 0.000 description 1
- 239000005388 borosilicate glass Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 238000010924 continuous production Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 238000004031 devitrification Methods 0.000 description 1
- 239000007772 electrode material Substances 0.000 description 1
- RSEIMSPAXMNYFJ-UHFFFAOYSA-N europium(III) oxide Inorganic materials O=[Eu]O[Eu]=O RSEIMSPAXMNYFJ-UHFFFAOYSA-N 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000445 field-emission scanning electron microscopy Methods 0.000 description 1
- CMIHHWBVHJVIGI-UHFFFAOYSA-N gadolinium(III) oxide Inorganic materials [O-2].[O-2].[O-2].[Gd+3].[Gd+3] CMIHHWBVHJVIGI-UHFFFAOYSA-N 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- MRELNEQAGSRDBK-UHFFFAOYSA-N lanthanum oxide Inorganic materials [O-2].[O-2].[O-2].[La+3].[La+3] MRELNEQAGSRDBK-UHFFFAOYSA-N 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- KTUFCUMIWABKDW-UHFFFAOYSA-N oxo(oxolanthaniooxy)lanthanum Chemical compound O=[La]O[La]=O KTUFCUMIWABKDW-UHFFFAOYSA-N 0.000 description 1
- 238000005498 polishing Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000001330 spinodal decomposition reaction Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- PBCFLUZVCVVTBY-UHFFFAOYSA-N tantalum pentoxide Inorganic materials O=[Ta](=O)O[Ta](=O)=O PBCFLUZVCVVTBY-UHFFFAOYSA-N 0.000 description 1
- LAJZODKXOMJMPK-UHFFFAOYSA-N tellurium dioxide Chemical compound O=[Te]=O LAJZODKXOMJMPK-UHFFFAOYSA-N 0.000 description 1
- YEAUATLBSVJFOY-UHFFFAOYSA-N tetraantimony hexaoxide Chemical compound O1[Sb](O2)O[Sb]3O[Sb]1O[Sb]2O3 YEAUATLBSVJFOY-UHFFFAOYSA-N 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- RUDFQVOCFDJEEF-UHFFFAOYSA-N yttrium(III) oxide Inorganic materials [O-2].[O-2].[O-2].[Y+3].[Y+3] RUDFQVOCFDJEEF-UHFFFAOYSA-N 0.000 description 1
Landscapes
- Surface Treatment Of Glass (AREA)
- Glass Compositions (AREA)
Description
Claims (4)
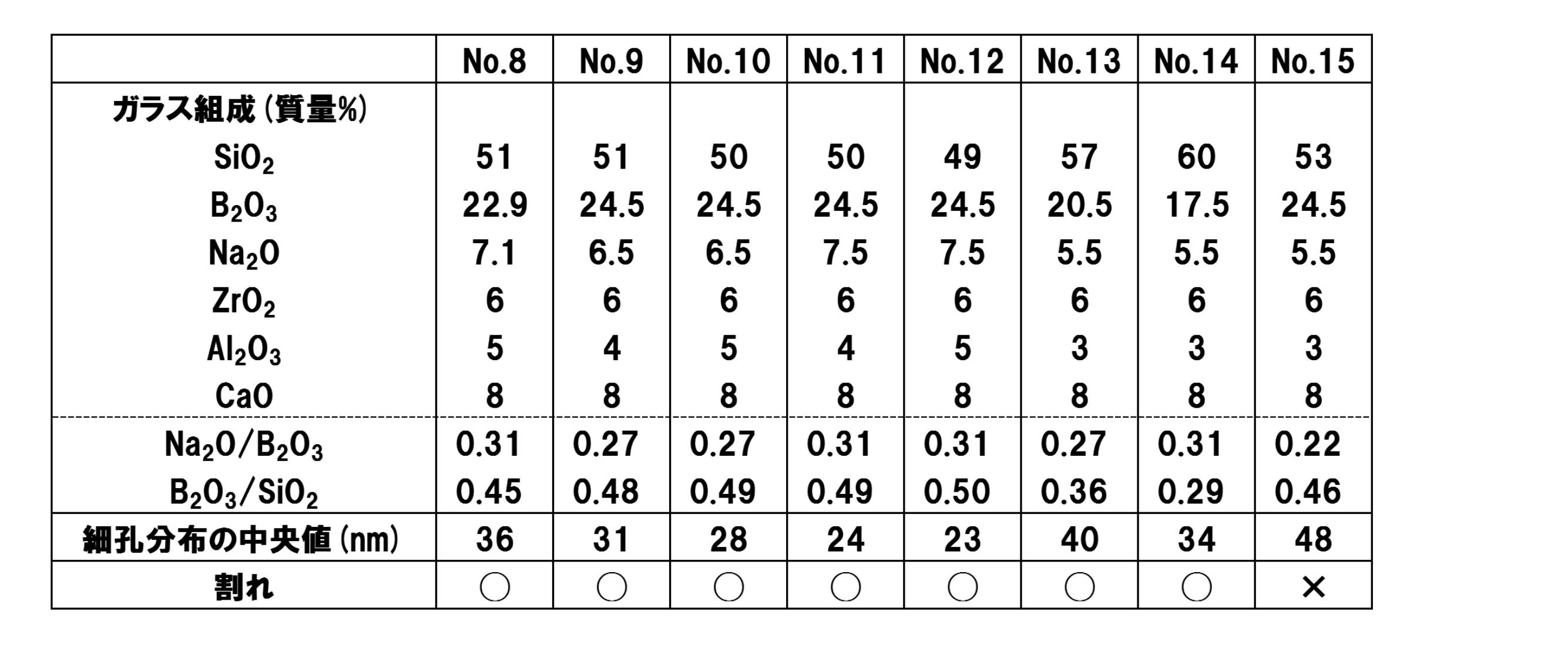
- 質量%で、SiO2 40~70%、B2O3 10~30%、Na2O 3~15%、ZrO2 3超~10%、Al2O3 2~5%、RO(RはMg、Ca、Sr及びBaから選択される少なくとも1種) 8~20%、CaO 8~20%を含有し、質量比でNa2O/B2O3が0.3~0.5であるガラス母材を熱処理し、2相に分相させる工程、及び、一方の相を酸で除去する工程を含むことを特徴とする多孔質ガラス部材の製造方法。
- ガラス母材が、2~1000のアスペクト比を有することを特徴とする請求項1に記載の多孔質ガラス部材の製造方法。
- 熱処理温度が500~800℃であることを特徴とする請求項1又は2に記載の多孔質ガラス部材の製造方法。
- 質量%で、SiO2 40~70%、B2O3 10~30%、Na2O 3~15%、ZrO2 3超~10%、Al2O3 2~5%、RO(RはMg、Ca、Sr及びBaから選択される少なくとも1種) 8~20%、CaO 8~20%を含有し、質量比でNa2O/B2O3が0.3~0.5であることを特徴とする多孔質ガラス部材用ガラス母材。
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017111421 | 2017-06-06 | ||
| JP2017111421 | 2017-06-06 | ||
| JP2017216983 | 2017-11-10 | ||
| JP2017216983 | 2017-11-10 | ||
| JP2018046303 | 2018-03-14 | ||
| JP2018046303 | 2018-03-14 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2019163198A JP2019163198A (ja) | 2019-09-26 |
| JP7280547B2 true JP7280547B2 (ja) | 2023-05-24 |
Family
ID=68065818
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018095314A Active JP7280547B2 (ja) | 2017-06-06 | 2018-05-17 | 多孔質ガラス部材の製造方法 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP7280547B2 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021095544A1 (ja) * | 2019-11-11 | 2021-05-20 | 日本電気硝子株式会社 | 多孔質ガラス部材の製造方法 |
| CN114650973A (zh) * | 2019-11-11 | 2022-06-21 | 日本电气硝子株式会社 | 多孔玻璃材料的制造方法 |
| US20230286853A1 (en) * | 2020-07-13 | 2023-09-14 | Nippon Electric Glass Co., Ltd. | Porous glass member |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61106437A (ja) * | 1984-10-26 | 1986-05-24 | Asahi Glass Co Ltd | 多孔質ガラス用組成物及び多孔質ガラスの製造法 |
| JPS62202839A (ja) * | 1985-10-14 | 1987-09-07 | Agency Of Ind Science & Technol | 耐薬品性多孔質ガラス及びその製造方法 |
| JPH01145349A (ja) * | 1987-11-30 | 1989-06-07 | Agency Of Ind Science & Technol | 多孔質ガラス用組成物 |
| JPH03146440A (ja) * | 1989-10-31 | 1991-06-21 | Central Glass Co Ltd | ポーラスガラス用組成物 |
-
2018
- 2018-05-17 JP JP2018095314A patent/JP7280547B2/ja active Active
Non-Patent Citations (2)
| Title |
|---|
| 森本繁樹,SiO2-B2O3-Al2O3-ZrO2-RO-R2O系ガラスの分相,日本セラミックス協会学術論文誌,日本,1990年,98 [10],pp.1093-1096 |
| 森本繁樹,高化学的耐久性ポーラスガラスの作製,日本セラミックス協会学術論文誌,日本,1990年,98 [12],pp.1291-1295 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2019163198A (ja) | 2019-09-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5318748B2 (ja) | 陽極接合用ガラス | |
| CN112047626B (zh) | 低气泡数、低介电常数的玻璃组成物及由其制得的玻璃纤维 | |
| CN109803937B (zh) | 玻璃陶瓷 | |
| JP5950587B2 (ja) | 多孔質ガラスの製造方法および光学部材の製造方法 | |
| JP2011241130A (ja) | 相分離ガラスおよび多孔質ガラス | |
| TW201130755A (en) | Glass plate for display device, plate glass for display device and production process thereof | |
| JP2016513068A (ja) | 相分離したクラッド層を有する積層化ガラス物品およびその形成方法 | |
| EP2731919B1 (en) | Chemically durable porous glass with enhanced alkaline resistance | |
| JP2021512843A (ja) | ガラス用組成物、低介在物含有量のガラス、その製造方法及び使用 | |
| JP7280547B2 (ja) | 多孔質ガラス部材の製造方法 | |
| JPH0676224B2 (ja) | 強化ガラスの製造法 | |
| US20140349831A1 (en) | Alumina-rich glasses and methods for making the same | |
| JPWO2015111524A1 (ja) | 強化用ガラス組成物、強化ガラス物品およびその製造方法 | |
| JP7648988B2 (ja) | 多孔質ガラス部材の製造方法 | |
| JP7168901B2 (ja) | 多孔質ガラス部材の製造方法 | |
| CN115461314A (zh) | 多孔玻璃材料 | |
| JP2012091996A (ja) | 多孔質ガラスの製造方法 | |
| TW202340110A (zh) | 分相玻璃 | |
| JP2024060430A (ja) | 多孔質ガラス材の製造方法及び多孔質ガラス材 | |
| WO2021095545A1 (ja) | 多孔質ガラス部材の製造方法 | |
| JP7303480B2 (ja) | 多孔質ガラス部材 | |
| JP7425400B2 (ja) | 多孔質ガラス部材の製造方法 | |
| JP2012193067A (ja) | ホウケイ酸塩ガラス、多孔質ガラスおよびその製造方法 | |
| JP2023004533A (ja) | 多孔質ガラス部材の製造方法 | |
| JP7301284B2 (ja) | アルデヒド系ガス検出用材料及びノナナールガス検出用材料 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20210405 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20211206 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20211213 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20220204 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220328 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220620 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20220808 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220914 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20221004 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20221202 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20230105 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20230125 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20230322 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20230412 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20230425 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7280547 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |