JP7237828B2 - エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 - Google Patents
エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 Download PDFInfo
- Publication number
- JP7237828B2 JP7237828B2 JP2019523484A JP2019523484A JP7237828B2 JP 7237828 B2 JP7237828 B2 JP 7237828B2 JP 2019523484 A JP2019523484 A JP 2019523484A JP 2019523484 A JP2019523484 A JP 2019523484A JP 7237828 B2 JP7237828 B2 JP 7237828B2
- Authority
- JP
- Japan
- Prior art keywords
- epoxy resin
- resin composition
- polished surface
- fused silica
- electronic component
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000003822 epoxy resin Substances 0.000 title claims description 131
- 229920000647 polyepoxide Polymers 0.000 title claims description 131
- 239000000203 mixture Substances 0.000 title claims description 90
- 238000004519 manufacturing process Methods 0.000 title description 3
- 239000002245 particle Substances 0.000 claims description 84
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 61
- 239000005350 fused silica glass Substances 0.000 claims description 55
- 239000011148 porous material Substances 0.000 claims description 29
- 238000005498 polishing Methods 0.000 claims description 18
- 239000003795 chemical substances by application Substances 0.000 claims description 15
- 238000000748 compression moulding Methods 0.000 claims description 9
- 238000007789 sealing Methods 0.000 claims description 7
- 239000012778 molding material Substances 0.000 claims description 4
- 238000001723 curing Methods 0.000 description 41
- 239000000463 material Substances 0.000 description 26
- 239000000047 product Substances 0.000 description 26
- 239000000758 substrate Substances 0.000 description 26
- -1 amine compounds Chemical class 0.000 description 24
- 229920005989 resin Polymers 0.000 description 23
- 239000011347 resin Substances 0.000 description 23
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 15
- 239000011248 coating agent Substances 0.000 description 15
- 238000000576 coating method Methods 0.000 description 15
- 239000004065 semiconductor Substances 0.000 description 15
- 229920003986 novolac Polymers 0.000 description 14
- 238000000034 method Methods 0.000 description 13
- 235000012431 wafers Nutrition 0.000 description 13
- 230000000052 comparative effect Effects 0.000 description 12
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 12
- 238000000465 moulding Methods 0.000 description 11
- 238000012360 testing method Methods 0.000 description 11
- 229930185605 Bisphenol Natural products 0.000 description 9
- 238000010438 heat treatment Methods 0.000 description 8
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 7
- 150000001412 amines Chemical class 0.000 description 6
- 239000008393 encapsulating agent Substances 0.000 description 6
- 238000011156 evaluation Methods 0.000 description 6
- 239000000945 filler Substances 0.000 description 6
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 6
- 239000003566 sealing material Substances 0.000 description 6
- KJCVRFUGPWSIIH-UHFFFAOYSA-N 1-naphthol Chemical compound C1=CC=C2C(O)=CC=CC2=C1 KJCVRFUGPWSIIH-UHFFFAOYSA-N 0.000 description 5
- 239000006087 Silane Coupling Agent Substances 0.000 description 5
- 229920001721 polyimide Polymers 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- 150000008064 anhydrides Chemical class 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 238000009826 distribution Methods 0.000 description 4
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 238000011417 postcuring Methods 0.000 description 4
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 150000008065 acid anhydrides Chemical class 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 229910017052 cobalt Inorganic materials 0.000 description 3
- 239000010941 cobalt Substances 0.000 description 3
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 3
- 229930003836 cresol Natural products 0.000 description 3
- 238000000227 grinding Methods 0.000 description 3
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 239000011574 phosphorus Substances 0.000 description 3
- 239000009719 polyimide resin Substances 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 230000003746 surface roughness Effects 0.000 description 3
- HECLRDQVFMWTQS-RGOKHQFPSA-N 1755-01-7 Chemical compound C1[C@H]2[C@@H]3CC=C[C@@H]3[C@@H]1C=C2 HECLRDQVFMWTQS-RGOKHQFPSA-N 0.000 description 2
- GJYCVCVHRSWLNY-UHFFFAOYSA-N 2-butylphenol Chemical compound CCCCC1=CC=CC=C1O GJYCVCVHRSWLNY-UHFFFAOYSA-N 0.000 description 2
- LXBGSDVWAMZHDD-UHFFFAOYSA-N 2-methyl-1h-imidazole Chemical compound CC1=NC=CN1 LXBGSDVWAMZHDD-UHFFFAOYSA-N 0.000 description 2
- JWAZRIHNYRIHIV-UHFFFAOYSA-N 2-naphthol Chemical compound C1=CC=CC2=CC(O)=CC=C21 JWAZRIHNYRIHIV-UHFFFAOYSA-N 0.000 description 2
- ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 2-phenyl-1h-imidazole Chemical compound C1=CNC(C=2C=CC=CC=2)=N1 ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 0.000 description 2
- PLIKAWJENQZMHA-UHFFFAOYSA-N 4-aminophenol Chemical compound NC1=CC=C(O)C=C1 PLIKAWJENQZMHA-UHFFFAOYSA-N 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- 239000004593 Epoxy Substances 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- 241000209094 Oryza Species 0.000 description 2
- 235000007164 Oryza sativa Nutrition 0.000 description 2
- 239000004642 Polyimide Substances 0.000 description 2
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 2
- 239000006227 byproduct Substances 0.000 description 2
- 239000006229 carbon black Substances 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 238000010292 electrical insulation Methods 0.000 description 2
- 150000002460 imidazoles Chemical class 0.000 description 2
- 150000002462 imidazolines Chemical class 0.000 description 2
- 238000007561 laser diffraction method Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 2
- 150000002902 organometallic compounds Chemical class 0.000 description 2
- 125000002524 organometallic group Chemical group 0.000 description 2
- 239000005011 phenolic resin Substances 0.000 description 2
- 238000007639 printing Methods 0.000 description 2
- 230000001681 protective effect Effects 0.000 description 2
- 235000009566 rice Nutrition 0.000 description 2
- 238000004528 spin coating Methods 0.000 description 2
- 230000008719 thickening Effects 0.000 description 2
- 238000001721 transfer moulding Methods 0.000 description 2
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- LTVUCOSIZFEASK-MPXCPUAZSA-N (3ar,4s,7r,7as)-3a-methyl-3a,4,7,7a-tetrahydro-4,7-methano-2-benzofuran-1,3-dione Chemical compound C([C@H]1C=C2)[C@H]2[C@H]2[C@]1(C)C(=O)OC2=O LTVUCOSIZFEASK-MPXCPUAZSA-N 0.000 description 1
- KNDQHSIWLOJIGP-UMRXKNAASA-N (3ar,4s,7r,7as)-rel-3a,4,7,7a-tetrahydro-4,7-methanoisobenzofuran-1,3-dione Chemical compound O=C1OC(=O)[C@@H]2[C@H]1[C@]1([H])C=C[C@@]2([H])C1 KNDQHSIWLOJIGP-UMRXKNAASA-N 0.000 description 1
- MUTGBJKUEZFXGO-OLQVQODUSA-N (3as,7ar)-3a,4,5,6,7,7a-hexahydro-2-benzofuran-1,3-dione Chemical compound C1CCC[C@@H]2C(=O)OC(=O)[C@@H]21 MUTGBJKUEZFXGO-OLQVQODUSA-N 0.000 description 1
- KMOUUZVZFBCRAM-OLQVQODUSA-N (3as,7ar)-3a,4,7,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1C=CC[C@@H]2C(=O)OC(=O)[C@@H]21 KMOUUZVZFBCRAM-OLQVQODUSA-N 0.000 description 1
- HLMLRAFRBPXQHA-UHFFFAOYSA-N (4,5-diethyl-2,3-dimethylphenyl)-phenylmethanediamine Chemical compound CC1=C(CC)C(CC)=CC(C(N)(N)C=2C=CC=CC=2)=C1C HLMLRAFRBPXQHA-UHFFFAOYSA-N 0.000 description 1
- LDCQBHLZLZUAAF-UHFFFAOYSA-N (5-methyl-2-phenyl-1h-imidazol-4-yl)methanediol Chemical compound OC(O)C1=C(C)NC(C=2C=CC=CC=2)=N1 LDCQBHLZLZUAAF-UHFFFAOYSA-N 0.000 description 1
- WBODDOZXDKQEFS-UHFFFAOYSA-N 1,2,3,4-tetramethyl-5-phenylbenzene Chemical group CC1=C(C)C(C)=CC(C=2C=CC=CC=2)=C1C WBODDOZXDKQEFS-UHFFFAOYSA-N 0.000 description 1
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical compound C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 1
- GIWQSPITLQVMSG-UHFFFAOYSA-N 1,2-dimethylimidazole Chemical compound CC1=NC=CN1C GIWQSPITLQVMSG-UHFFFAOYSA-N 0.000 description 1
- FJWZMLSQLCKKGV-UHFFFAOYSA-N 1-(2-ethylphenyl)propane-1,1-diamine Chemical compound CCC1=CC=CC=C1C(N)(N)CC FJWZMLSQLCKKGV-UHFFFAOYSA-N 0.000 description 1
- IJBDNUKXUYJCAV-UHFFFAOYSA-N 1-(2-methylphenyl)ethane-1,1-diamine Chemical compound CC1=CC=CC=C1C(C)(N)N IJBDNUKXUYJCAV-UHFFFAOYSA-N 0.000 description 1
- XQUPVDVFXZDTLT-UHFFFAOYSA-N 1-[4-[[4-(2,5-dioxopyrrol-1-yl)phenyl]methyl]phenyl]pyrrole-2,5-dione Chemical compound O=C1C=CC(=O)N1C(C=C1)=CC=C1CC1=CC=C(N2C(C=CC2=O)=O)C=C1 XQUPVDVFXZDTLT-UHFFFAOYSA-N 0.000 description 1
- FBHPRUXJQNWTEW-UHFFFAOYSA-N 1-benzyl-2-methylimidazole Chemical compound CC1=NC=CN1CC1=CC=CC=C1 FBHPRUXJQNWTEW-UHFFFAOYSA-N 0.000 description 1
- XZKLXPPYISZJCV-UHFFFAOYSA-N 1-benzyl-2-phenylimidazole Chemical compound C1=CN=C(C=2C=CC=CC=2)N1CC1=CC=CC=C1 XZKLXPPYISZJCV-UHFFFAOYSA-N 0.000 description 1
- VILCJCGEZXAXTO-UHFFFAOYSA-N 2,2,2-tetramine Chemical compound NCCNCCNCCN VILCJCGEZXAXTO-UHFFFAOYSA-N 0.000 description 1
- AHDSRXYHVZECER-UHFFFAOYSA-N 2,4,6-tris[(dimethylamino)methyl]phenol Chemical compound CN(C)CC1=CC(CN(C)C)=C(O)C(CN(C)C)=C1 AHDSRXYHVZECER-UHFFFAOYSA-N 0.000 description 1
- YPGATYQQJOPMOG-UHFFFAOYSA-N 2-(4-aminoheptan-4-yl)aniline Chemical compound CCCC(N)(CCC)C1=CC=CC=C1N YPGATYQQJOPMOG-UHFFFAOYSA-N 0.000 description 1
- QVTWJEIWMOOEAG-UHFFFAOYSA-N 2-(5-aminononan-5-yl)aniline Chemical compound CCCCC(N)(CCCC)C1=CC=CC=C1N QVTWJEIWMOOEAG-UHFFFAOYSA-N 0.000 description 1
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 1
- SFRDXVJWXWOTEW-UHFFFAOYSA-N 2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)CO SFRDXVJWXWOTEW-UHFFFAOYSA-N 0.000 description 1
- RESFZFUAOCMIOD-UHFFFAOYSA-N 2-[[4-(oxiran-2-ylmethoxy)-2,5-di(propan-2-yl)phenoxy]methyl]oxirane Chemical compound CC(C)C=1C=C(OCC2OC2)C(C(C)C)=CC=1OCC1CO1 RESFZFUAOCMIOD-UHFFFAOYSA-N 0.000 description 1
- FSYPIGPPWAJCJG-UHFFFAOYSA-N 2-[[4-(oxiran-2-ylmethoxy)phenoxy]methyl]oxirane Chemical compound C1OC1COC(C=C1)=CC=C1OCC1CO1 FSYPIGPPWAJCJG-UHFFFAOYSA-N 0.000 description 1
- MEVBAGCIOOTPLF-UHFFFAOYSA-N 2-[[5-(oxiran-2-ylmethoxy)naphthalen-2-yl]oxymethyl]oxirane Chemical compound C1OC1COC(C=C1C=CC=2)=CC=C1C=2OCC1CO1 MEVBAGCIOOTPLF-UHFFFAOYSA-N 0.000 description 1
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical compound NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 description 1
- PQAMFDRRWURCFQ-UHFFFAOYSA-N 2-ethyl-1h-imidazole Chemical compound CCC1=NC=CN1 PQAMFDRRWURCFQ-UHFFFAOYSA-N 0.000 description 1
- SVNWKKJQEFIURY-UHFFFAOYSA-N 2-methyl-1-(2-methylpropyl)imidazole Chemical compound CC(C)CN1C=CN=C1C SVNWKKJQEFIURY-UHFFFAOYSA-N 0.000 description 1
- OVEUFHOBGCSKSH-UHFFFAOYSA-N 2-methyl-n,n-bis(oxiran-2-ylmethyl)aniline Chemical compound CC1=CC=CC=C1N(CC1OC1)CC1OC1 OVEUFHOBGCSKSH-UHFFFAOYSA-N 0.000 description 1
- UIDDPPKZYZTEGS-UHFFFAOYSA-N 3-(2-ethyl-4-methylimidazol-1-yl)propanenitrile Chemical compound CCC1=NC(C)=CN1CCC#N UIDDPPKZYZTEGS-UHFFFAOYSA-N 0.000 description 1
- SESYNEDUKZDRJL-UHFFFAOYSA-N 3-(2-methylimidazol-1-yl)propanenitrile Chemical compound CC1=NC=CN1CCC#N SESYNEDUKZDRJL-UHFFFAOYSA-N 0.000 description 1
- BVYPJEBKDLFIDL-UHFFFAOYSA-N 3-(2-phenylimidazol-1-yl)propanenitrile Chemical compound N#CCCN1C=CN=C1C1=CC=CC=C1 BVYPJEBKDLFIDL-UHFFFAOYSA-N 0.000 description 1
- SZUPZARBRLCVCB-UHFFFAOYSA-N 3-(2-undecylimidazol-1-yl)propanenitrile Chemical compound CCCCCCCCCCCC1=NC=CN1CCC#N SZUPZARBRLCVCB-UHFFFAOYSA-N 0.000 description 1
- MECNWXGGNCJFQJ-UHFFFAOYSA-N 3-piperidin-1-ylpropane-1,2-diol Chemical compound OCC(O)CN1CCCCC1 MECNWXGGNCJFQJ-UHFFFAOYSA-N 0.000 description 1
- JNRLEMMIVRBKJE-UHFFFAOYSA-N 4,4'-Methylenebis(N,N-dimethylaniline) Chemical compound C1=CC(N(C)C)=CC=C1CC1=CC=C(N(C)C)C=C1 JNRLEMMIVRBKJE-UHFFFAOYSA-N 0.000 description 1
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 1
- CBEVWPCAHIAUOD-UHFFFAOYSA-N 4-[(4-amino-3-ethylphenyl)methyl]-2-ethylaniline Chemical compound C1=C(N)C(CC)=CC(CC=2C=C(CC)C(N)=CC=2)=C1 CBEVWPCAHIAUOD-UHFFFAOYSA-N 0.000 description 1
- NUDSREQIJYWLRA-UHFFFAOYSA-N 4-[9-(4-hydroxy-3-methylphenyl)fluoren-9-yl]-2-methylphenol Chemical compound C1=C(O)C(C)=CC(C2(C3=CC=CC=C3C3=CC=CC=C32)C=2C=C(C)C(O)=CC=2)=C1 NUDSREQIJYWLRA-UHFFFAOYSA-N 0.000 description 1
- CXXSQMDHHYTRKY-UHFFFAOYSA-N 4-amino-2,3,5-tris(oxiran-2-ylmethyl)phenol Chemical compound C1=C(O)C(CC2OC2)=C(CC2OC2)C(N)=C1CC1CO1 CXXSQMDHHYTRKY-UHFFFAOYSA-N 0.000 description 1
- FUGYGGDSWSUORM-UHFFFAOYSA-N 4-hydroxystyrene Chemical compound OC1=CC=C(C=C)C=C1 FUGYGGDSWSUORM-UHFFFAOYSA-N 0.000 description 1
- LWMIDUUVMLBKQF-UHFFFAOYSA-N 4-methyl-3a,4,5,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound CC1CC=CC2C(=O)OC(=O)C12 LWMIDUUVMLBKQF-UHFFFAOYSA-N 0.000 description 1
- BFPKYGRZERDWLA-UHFFFAOYSA-N 4-methylbicyclo[2.2.1]heptane-2,3-dicarboxylic acid Chemical compound C1CC2C(C(O)=O)C(C(O)=O)C1(C)C2 BFPKYGRZERDWLA-UHFFFAOYSA-N 0.000 description 1
- QJNLUNBGDFUULX-UHFFFAOYSA-N 4-n,4-n'-dimethyl-3h-pyridine-4,4-diamine Chemical compound CNC1(NC)CC=NC=C1 QJNLUNBGDFUULX-UHFFFAOYSA-N 0.000 description 1
- TYOXIFXYEIILLY-UHFFFAOYSA-N 5-methyl-2-phenyl-1h-imidazole Chemical compound N1C(C)=CN=C1C1=CC=CC=C1 TYOXIFXYEIILLY-UHFFFAOYSA-N 0.000 description 1
- ULKLGIFJWFIQFF-UHFFFAOYSA-N 5K8XI641G3 Chemical compound CCC1=NC=C(C)N1 ULKLGIFJWFIQFF-UHFFFAOYSA-N 0.000 description 1
- RBHIUNHSNSQJNG-UHFFFAOYSA-N 6-methyl-3-(2-methyloxiran-2-yl)-7-oxabicyclo[4.1.0]heptane Chemical compound C1CC2(C)OC2CC1C1(C)CO1 RBHIUNHSNSQJNG-UHFFFAOYSA-N 0.000 description 1
- MWSKJDNQKGCKPA-UHFFFAOYSA-N 6-methyl-3a,4,5,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1CC(C)=CC2C(=O)OC(=O)C12 MWSKJDNQKGCKPA-UHFFFAOYSA-N 0.000 description 1
- 229910000838 Al alloy Inorganic materials 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 229910000881 Cu alloy Inorganic materials 0.000 description 1
- MQJKPEGWNLWLTK-UHFFFAOYSA-N Dapsone Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=C(N)C=C1 MQJKPEGWNLWLTK-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- AFCARXCZXQIEQB-UHFFFAOYSA-N N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CCNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 AFCARXCZXQIEQB-UHFFFAOYSA-N 0.000 description 1
- LGRFSURHDFAFJT-UHFFFAOYSA-N Phthalic anhydride Natural products C1=CC=C2C(=O)OC(=O)C2=C1 LGRFSURHDFAFJT-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- UUQQGGWZVKUCBD-UHFFFAOYSA-N [4-(hydroxymethyl)-2-phenyl-1h-imidazol-5-yl]methanol Chemical compound N1C(CO)=C(CO)N=C1C1=CC=CC=C1 UUQQGGWZVKUCBD-UHFFFAOYSA-N 0.000 description 1
- DPRMFUAMSRXGDE-UHFFFAOYSA-N ac1o530g Chemical compound NCCN.NCCN DPRMFUAMSRXGDE-UHFFFAOYSA-N 0.000 description 1
- 125000002723 alicyclic group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- JHIWVOJDXOSYLW-UHFFFAOYSA-N butyl 2,2-difluorocyclopropane-1-carboxylate Chemical compound CCCCOC(=O)C1CC1(F)F JHIWVOJDXOSYLW-UHFFFAOYSA-N 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 230000001066 destructive effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000007607 die coating method Methods 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- ZZTCPWRAHWXWCH-UHFFFAOYSA-N diphenylmethanediamine Chemical compound C=1C=CC=CC=1C(N)(N)C1=CC=CC=C1 ZZTCPWRAHWXWCH-UHFFFAOYSA-N 0.000 description 1
- 238000007772 electroless plating Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- FWDBOZPQNFPOLF-UHFFFAOYSA-N ethenyl(triethoxy)silane Chemical compound CCO[Si](OCC)(OCC)C=C FWDBOZPQNFPOLF-UHFFFAOYSA-N 0.000 description 1
- NKSJNEHGWDZZQF-UHFFFAOYSA-N ethenyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)C=C NKSJNEHGWDZZQF-UHFFFAOYSA-N 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 238000007756 gravure coating Methods 0.000 description 1
- 238000013007 heat curing Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 239000012948 isocyanate Substances 0.000 description 1
- 150000002513 isocyanates Chemical class 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000004850 liquid epoxy resins (LERs) Substances 0.000 description 1
- 150000002736 metal compounds Chemical class 0.000 description 1
- VYKXQOYUCMREIS-UHFFFAOYSA-N methylhexahydrophthalic anhydride Chemical compound C1CCCC2C(=O)OC(=O)C21C VYKXQOYUCMREIS-UHFFFAOYSA-N 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- JAYXSROKFZAHRQ-UHFFFAOYSA-N n,n-bis(oxiran-2-ylmethyl)aniline Chemical compound C1OC1CN(C=1C=CC=CC=1)CC1CO1 JAYXSROKFZAHRQ-UHFFFAOYSA-N 0.000 description 1
- GEMHFKXPOCTAIP-UHFFFAOYSA-N n,n-dimethyl-n'-phenylcarbamimidoyl chloride Chemical compound CN(C)C(Cl)=NC1=CC=CC=C1 GEMHFKXPOCTAIP-UHFFFAOYSA-N 0.000 description 1
- 150000004780 naphthols Chemical class 0.000 description 1
- AFEQENGXSMURHA-UHFFFAOYSA-N oxiran-2-ylmethanamine Chemical compound NCC1CO1 AFEQENGXSMURHA-UHFFFAOYSA-N 0.000 description 1
- 229920001568 phenolic resin Polymers 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- ZWGWPHTUOWMLHG-UHFFFAOYSA-N phenyl-(2,3,4,5-tetraethylphenyl)methanediamine Chemical compound CCC1=C(CC)C(CC)=CC(C(N)(N)C=2C=CC=CC=2)=C1CC ZWGWPHTUOWMLHG-UHFFFAOYSA-N 0.000 description 1
- IGALFTFNPPBUDN-UHFFFAOYSA-N phenyl-[2,3,4,5-tetrakis(oxiran-2-ylmethyl)phenyl]methanediamine Chemical compound C=1C(CC2OC2)=C(CC2OC2)C(CC2OC2)=C(CC2OC2)C=1C(N)(N)C1=CC=CC=C1 IGALFTFNPPBUDN-UHFFFAOYSA-N 0.000 description 1
- 150000004714 phosphonium salts Chemical class 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 229920003192 poly(bis maleimide) Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 229920002577 polybenzoxazole Polymers 0.000 description 1
- 238000012643 polycondensation polymerization Methods 0.000 description 1
- 229910052594 sapphire Inorganic materials 0.000 description 1
- 239000010980 sapphire Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- WSFQLUVWDKCYSW-UHFFFAOYSA-M sodium;2-hydroxy-3-morpholin-4-ylpropane-1-sulfonate Chemical compound [Na+].[O-]S(=O)(=O)CC(O)CN1CCOCC1 WSFQLUVWDKCYSW-UHFFFAOYSA-M 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 229940014800 succinic anhydride Drugs 0.000 description 1
- 150000004992 toluidines Chemical class 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 238000011282 treatment Methods 0.000 description 1
- 150000003918 triazines Chemical class 0.000 description 1
- TUQOTMZNTHZOKS-UHFFFAOYSA-N tributylphosphine Chemical compound CCCCP(CCCC)CCCC TUQOTMZNTHZOKS-UHFFFAOYSA-N 0.000 description 1
- UDUKMRHNZZLJRB-UHFFFAOYSA-N triethoxy-[2-(7-oxabicyclo[4.1.0]heptan-4-yl)ethyl]silane Chemical compound C1C(CC[Si](OCC)(OCC)OCC)CCC2OC21 UDUKMRHNZZLJRB-UHFFFAOYSA-N 0.000 description 1
- JXUKBNICSRJFAP-UHFFFAOYSA-N triethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](OCC)(OCC)CCCOCC1CO1 JXUKBNICSRJFAP-UHFFFAOYSA-N 0.000 description 1
- SRPWOOOHEPICQU-UHFFFAOYSA-N trimellitic anhydride Chemical compound OC(=O)C1=CC=C2C(=O)OC(=O)C2=C1 SRPWOOOHEPICQU-UHFFFAOYSA-N 0.000 description 1
- DQZNLOXENNXVAD-UHFFFAOYSA-N trimethoxy-[2-(7-oxabicyclo[4.1.0]heptan-4-yl)ethyl]silane Chemical compound C1C(CC[Si](OC)(OC)OC)CCC2OC21 DQZNLOXENNXVAD-UHFFFAOYSA-N 0.000 description 1
- QXJQHYBHAIHNGG-UHFFFAOYSA-N trimethylolethane Chemical compound OCC(C)(CO)CO QXJQHYBHAIHNGG-UHFFFAOYSA-N 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 238000010333 wet classification Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/20—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the epoxy compounds used
- C08G59/22—Di-epoxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
- C08L63/04—Epoxynovolacs
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C43/00—Compression moulding, i.e. applying external pressure to flow the moulding material; Apparatus therefor
- B29C43/003—Compression moulding, i.e. applying external pressure to flow the moulding material; Apparatus therefor characterised by the choice of material
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/42—Polycarboxylic acids; Anhydrides, halides or low molecular weight esters thereof
- C08G59/4215—Polycarboxylic acids; Anhydrides, halides or low molecular weight esters thereof cycloaliphatic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/68—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the catalysts used
- C08G59/686—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the catalysts used containing nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/0008—Organic ingredients according to more than one of the "one dot" groups of C08K5/01 - C08K5/59
- C08K5/0025—Crosslinking or vulcanising agents; including accelerators
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K7/00—Use of ingredients characterised by shape
- C08K7/22—Expanded, porous or hollow particles
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K7/00—Use of ingredients characterised by shape
- C08K7/22—Expanded, porous or hollow particles
- C08K7/24—Expanded, porous or hollow particles inorganic
- C08K7/26—Silicon- containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J163/00—Adhesives based on epoxy resins; Adhesives based on derivatives of epoxy resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/50—Assembly of semiconductor devices using processes or apparatus not provided for in a single one of the groups H01L21/18 - H01L21/326 or H10D48/04 - H10D48/07 e.g. sealing of a cap to a base of a container
- H01L21/56—Encapsulations, e.g. encapsulation layers, coatings
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/50—Assembly of semiconductor devices using processes or apparatus not provided for in a single one of the groups H01L21/18 - H01L21/326 or H10D48/04 - H10D48/07 e.g. sealing of a cap to a base of a container
- H01L21/56—Encapsulations, e.g. encapsulation layers, coatings
- H01L21/563—Encapsulation of active face of flip-chip device, e.g. underfilling or underencapsulation of flip-chip, encapsulation preform on chip or mounting substrate
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/12—Mountings, e.g. non-detachable insulating substrates
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/29—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the material, e.g. carbon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/29—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the material, e.g. carbon
- H01L23/293—Organic, e.g. plastic
- H01L23/295—Organic, e.g. plastic containing a filler
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/31—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the arrangement or shape
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29K—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES B29B, B29C OR B29D, RELATING TO MOULDING MATERIALS OR TO MATERIALS FOR MOULDS, REINFORCEMENTS, FILLERS OR PREFORMED PARTS, e.g. INSERTS
- B29K2063/00—Use of EP, i.e. epoxy resins or derivatives thereof, as moulding material
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/42—Polycarboxylic acids; Anhydrides, halides or low molecular weight esters thereof
- C08G59/4238—Polycarboxylic acids; Anhydrides, halides or low molecular weight esters thereof heterocyclic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K2201/00—Specific properties of additives
- C08K2201/002—Physical properties
- C08K2201/003—Additives being defined by their diameter
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/17—Amines; Quaternary ammonium compounds
- C08K5/18—Amines; Quaternary ammonium compounds with aromatically bound amino groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2201/00—Properties
- C08L2201/08—Stabilised against heat, light or radiation or oxydation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2203/00—Applications
- C08L2203/20—Applications use in electrical or conductive gadgets
- C08L2203/206—Applications use in electrical or conductive gadgets use in coating or encapsulating of electronic parts
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Power Engineering (AREA)
- General Physics & Mathematics (AREA)
- Computer Hardware Design (AREA)
- Physics & Mathematics (AREA)
- Manufacturing & Machinery (AREA)
- Mechanical Engineering (AREA)
- Structures Or Materials For Encapsulating Or Coating Semiconductor Devices Or Solid State Devices (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Epoxy Resins (AREA)
Description
電子部品としては、例えば、樹脂フィルム上に配列された複数の個片化された半導体チップの集合体が準備される。このような集合体は、例えば、半導体ウエハをダイシングで個片化した後、樹脂フィルムに載置することにより準備される。
エポキシ樹脂組成物による電子部品の封止は、例えば、圧縮成形により行われるが、成形方法は圧縮成形に限るものではなく、印刷成形、トランスファー成形、ディスペンサなどによる塗布成形などであってもよい。圧縮成形では、例えば、金型内に電子部品とエポキシ樹脂組成物とが配置され、所定の圧力下でエポキシ樹脂組成物が加熱される。エポキシ樹脂組成物が硬化することにより、封止体が形成される。
次に、エポキシ樹脂組成物の硬化物の一部を研磨して、再配線層を形成するための研磨面を形成する。研磨は、封止体の片面だけに対して行ってもよく、両面に対して行ってもよい。すなわち、必要に応じて、封止体の両面に再配線層を形成してもよい。このとき、研磨面のRaが0.2μm以下、更には0.15μm以下になるまで硬化物を研磨することが好ましい。研磨はグラインダーなどの研磨機を用いて機械的研磨を実施してもよいし、化学的研磨を実施してもよい。
研磨面には、例えば、コーティング材料もしくは再配線形成用材料の膜が形成される。コーティング材料もしくは再配線形成用材料の膜を形成する方法は、特に限定されないが、例えば、ロール塗工、スクリーン塗工、グラビア塗工、スピンコート法、ダイコート法、スプレーコート法などにより、コーティング材料もしくは再配線形成用材料を塗布する方法などが挙げられる。研磨面は、平坦性が高いため、コーティング材料もしくは再配線形成用材料の膜を均一な厚みで容易に形成することができる。再配線(RDL)の形成、バンプ形成などが行われた後、ダイシングを行えば、個片化されたデバイス(電子部品実装構造体)が得られる。
(エポキシ樹脂)
エポキシ樹脂組成物に含まれるエポキシ樹脂としては、特に限定されないが、ビスフェノールA型エポキシ樹脂、ビスフェノールF型エポキシ樹脂、ビスフェノールAD型エポキシ樹脂、ビスフェノールS型エポキシ樹脂等のビスフェノール型エポキシ樹脂;ビスフェノール型エポキシ樹脂が部分縮合したオリゴマー混合物;ビスフェノールフルオレン型エポキシ樹脂、ビスクレゾールフルオレン型エポキシ樹脂等のフルオレン型エポキシ樹脂;1,6-ビス(2,3-エポキシプロポキシ)ナフタレン等のナフタレン型エポキシ樹脂;ビフェニル型またはテトラメチルビフェニル型エポキシ樹脂;トリグリシジル-p-アミノフェノール(p-アミノフェノール型エポキシ樹脂)などのアミノフェノール型エポキシ樹脂;ジグリシジルアニリンなどのアニリン型エポキシ樹脂;ジグリシジルオルソトルイジンなどのトルイジン型エポキシ樹脂;テトラグリシジルジアミノジフェニルメタンなどのジアミノジフェニルメタン型エポキシ樹脂等のグリシジルアミン型エポキシ樹脂;ジエポキシリモネン等の脂環式エポキシ樹脂;ジシクロペンタジエン型エポキシ樹脂;トリメチロールプロパントリグリシジルエーテル、トリメチロールメタントリグリシジルエーテル、トリメチロールエタントリグリシジルエーテル等のトリメチロールアルカン型エポキシ樹脂;ポリエーテル型エポキシ樹脂;シリコーン変性エポキシ樹脂;ノボラック型エポキシ樹脂;カテコールジグリシジルエーテル、レゾルシンジグリシジルエーテル、フタル酸ジグリシジルエーテル、2,5-ジイソプロピルヒドロキノンジグリシジルエーテル、ヒドロキノンジグリシジルエーテル等のベンゼン環を1個有する一核体芳香族エポキシ樹脂類;これらの核水添型エポキシ樹脂などを用いることができる。これらは単独で用いてもよく、2種以上を組み合わせて使用してもよい。これらの中では、ビスフェノールA型エポキシ樹脂、ビスフェノールF型エポキシ樹脂、ビスフェノールAD型エポキシ樹脂、ナフタレン型エポキシ樹脂、ノボラック型エポキシ樹脂が好ましく、ビスフェノールA型エポキシ樹脂、ビスフェノールF型エポキシ樹脂、ナフタレン型エポキシ樹脂がより好ましく、ナフタレン型エポキシ樹脂が耐湿性の点でさらに好ましい。これらは単独で用いてもよく、2種以上を組み合わせて用いてもよい。
エポキシ樹脂組成物には、エポキシ樹脂組成物の硬化物の熱膨張率を低く抑えるために、溶融シリカが含まれる。溶融シリカとしては、球状溶融シリカ、破砕溶融シリカなどを用いることができる。流動性の点からは球状溶融シリカが好ましい。球状溶融シリカとしては、粉砕したシリカ粉末を高温の火炎中で溶融し、表面張力により球状化させたものが主流である。
エポキシ樹脂組成物に含まれる硬化剤としては、酸無水物、フェノール樹脂、アミン化合物などを用いることができる。
エポキシ樹脂組成物には、エポキシ樹脂、溶融シリカ、硬化剤以外に、硬化促進剤、シランカップリング剤などが含まれてもよい。カーボンブラック、消泡剤、レベリング剤、顔料、応力緩和剤、プレゲル化剤、イオンキャッチャーなどを目的に応じて、適量添加することもできる。
エポキシ樹脂組成物は、各成分を所定の比率で配合し、例えば60~480分間攪拌し、その後、減圧下で脱泡してから用いるとよい。エポキシ樹脂組成物は、液状またはシート状であってもよく、半硬化状態であってもよい。
研磨面に塗布などにより膜化されるコーティング材料としては、特に限定されず、エポキシ樹脂組成物の硬化物の用途に応じて選択することができる。エポキシ樹脂組成物をFOWLPやFOPLPの封止材として用いる場合、再配線形成用材料として、例えば表面保護膜形成用材料が選択される。表面保護膜形成用材料には、例えば、感光性ポリイミド、感光性ポリベンゾオキサゾールなどが用いられる。再配線形成用材料の膜を形成する方法は、特に限定されず、例えば、スピンコート法、ダイコート法、スプレーコート法、デイップ、インクジェット、スクリーン印刷、ジェットディスペンスなどにより、研磨面に再配線形成用材料を塗布する方法などが挙げられる。塗膜形成後、露光、現像、エッチングなどの処理を経て、再配線層が形成される。または研磨面にスパッタリングや無電解メッキでシード層(Cuなど)を形成し、パターン形成してから、再配線層を形成してもよい。再配線層はCu、Cu合金、Al、Al合金等であってよい。
以下、本発明を実施例および比較例に基づいて具体的に説明するが、本発明は以下の実施例に限定されるものではない。
(1)エポキシ樹脂組成物の調製
以下を配合して120分間攪拌し、その後、減圧下で脱泡してエポキシ樹脂組成物を調製した。
<エポキシ樹脂> 100質量部
ビスフェノールA型エポキシ樹脂(日本化薬株式会社製、RE-310、エポキシ当量184g/eq)
<硬化剤> 100質量部
メチルテトラヒドロ無水フタル酸(日立化成株式会社製、酸無水物当量164g/eq)
<硬化促進剤> 6質量部
アミンアダクト系潜在性硬化促進剤(味の素株式会社製、アミキュアPN-23)
<シランカップリング剤> 3質量部
3-グリシドキシプロピルトリメトキシシラン
<カーボンブラック> 1質量部
三菱化学株式会社製、#2600
<溶融シリカA> 1500質量部
樹脂フィルム上に配列された複数の個片化された半導体チップ(厚さ1mm)上に、(1)で調製したエポキシ樹脂組成物を塗布し、圧縮成形により硬化させた。圧縮成形において、加熱は125℃で10分間、後硬化は150℃で1時間とした。
(2)で作製した試験片の硬化物の表面をグラインダーで研磨し、研磨面を形成した。
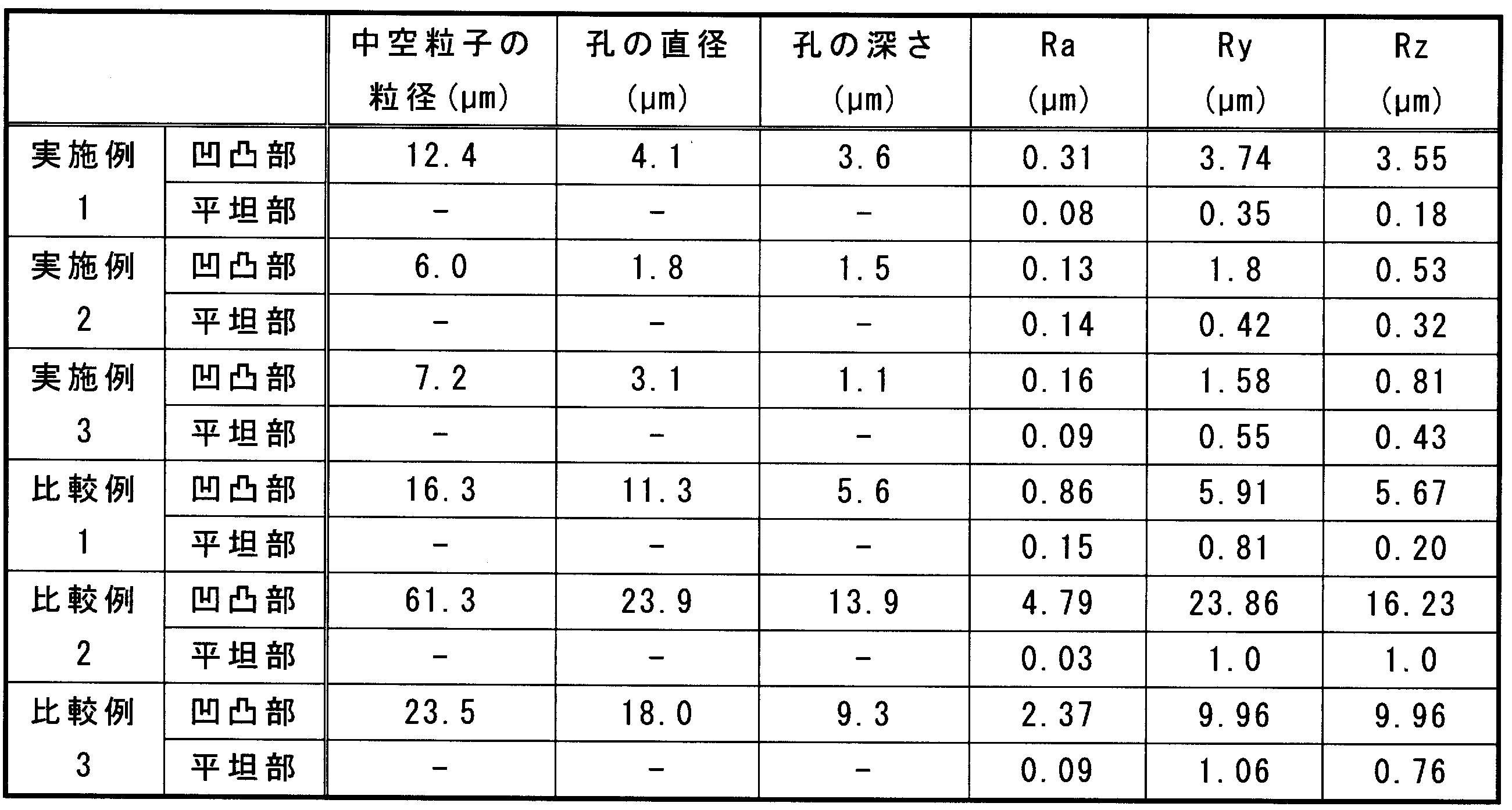
(3)で得られた試験片の硬化物の研磨面を、超深度カラー3D形状測定顕微鏡((株)キーエンス製、VK-9510)を用いて測定した。研磨面の写真を図1に示す。また、研磨面から無作為に選択した25mm2の範囲の10領域において、中空粒子の断面に由来する孔の直径および個数を測定した。直径が1.5μmより大きく5μm以下の孔、直径が5μmより大きく10μm以下の孔、直径が10μmより大きい孔の個数をそれぞれ測定し、平均値を求めた。結果を表1に示す。
溶融シリカとして溶融シリカBを用いたこと以外、実施例1と同様にエポキシ樹脂組成物を調製し、試験片を作製した。溶融シリカBは、精密分級により、できるだけ中空粒子を除去するとともに、最大粒径10μm以下、平均粒径3μmとしたものを用いた。研磨面の写真を図2に、研磨面を評価した結果を表1および表2に示す。
溶融シリカとして溶融シリカCを用いたこと以外、実施例1と同様にエポキシ樹脂組成物を調製し、試験片を作製した。溶融シリカCは、風力による精密分級により、できるだけ中空粒子を除去するとともに、最大粒径10μm以下、平均粒径2μmとしたものを用いた。研磨面の写真を図3に、研磨面を評価した結果を表1および表2に示す。
溶融シリカとして溶融シリカDを用いたこと以外、実施例1と同様にエポキシ樹脂組成物を調製し、試験片を作製した。溶融シリカDは、最大粒径25μm以下、平均粒径7μmのものを用いた。研磨面の写真を図4に、研磨面を評価した結果を表1および表2に示す。
溶融シリカとして溶融シリカEを用いたこと以外、実施例1と同様にエポキシ樹脂組成物を調製し、試験片を作製した。溶融シリカEは、最大粒径が75μm以下、平均粒径は20μmであった。研磨面の写真を図5に、研磨面を評価した結果を表1および表2に示す。
溶融シリカとして溶融シリカFを用いたこと以外、実施例1と同様にエポキシ樹脂組成物を調製し、試験片を作製した。溶融シリカFは、溶融シリカCの精密分級前の溶融シリカである。溶融シリカFの最大粒径は45μm、平均粒径は2μmであった。研磨面の写真を図6に、研磨面を評価した結果を表1および表2に示す。
2:実施例2で観察された中空粒子断面に由来する孔
3:実施例3で観察された中空粒子断面に由来する孔
4:比較例1で観察された中空粒子断面に由来する孔
5:比較例2で観察された中空粒子断面に由来する孔
6:比較例3で観察された中空粒子断面に由来する孔
Claims (8)
- エポキシ樹脂と、中空粒子を含み得る溶融シリカと、硬化剤及び/又は硬化促進剤と、を含むエポキシ樹脂組成物であって、
前記エポキシ樹脂組成物中の前記溶融シリカの含有量が、70~90質量%であり、
前記溶融シリカの最大粒径が5μmより大きく、
前記エポキシ樹脂組成物の硬化物を研磨して得られる研磨面であって、直径1.5μmより大きい孔を有さない領域における算術平均粗さRaが0.2μm以下になるまで研磨された研磨面において、25mm2の範囲に観測される前記中空粒子の断面に由来する直径5μmより大きい孔の数が、1個以下であり、
前記エポキシ樹脂組成物を150℃で1時間熱硬化することにより形成された硬化物に前記研磨面を形成する、エポキシ樹脂組成物。 - 前記25mm2の範囲に観測される前記中空粒子の断面に由来する直径2μmより大きい孔の数が、1個以下である、請求項1に記載のエポキシ樹脂組成物。
- 溶融シリカの最大粒径が25μm以下であり、平均粒径が1μmより大きい、請求項1または2に記載のエポキシ樹脂組成物。
- 前記研磨面は再配線層を形成するための研磨面である、請求項1~3のいずれか1項に記載のエポキシ樹脂組成物。
- ファンアウトパネルレベルパッケージ用またはファンアウトウエハレベルパッケージ用の圧縮成形用材料である、請求項1~4のいずれか1項に記載のエポキシ樹脂組成物。
- 電子部品と、
前記電子部品を封止するエポキシ樹脂組成物の硬化物と、を具備し、
前記エポキシ樹脂組成物は、エポキシ樹脂と、中空粒子を含み得る溶融シリカと、硬化剤及び/又は硬化促進剤と、を含み、
前記エポキシ樹脂組成物中の前記溶融シリカの含有量が、70~90質量%であり、
前記溶融シリカの最大粒径が5μmより大きく、
前記硬化物が、直径1.5μmより大きい孔を有さない領域における算術平均粗さRaが0.2μm以下になるまで研磨された研磨面を有し、
前記研磨面において、25mm2の範囲に観測される前記中空粒子の断面に由来する直径5μmより大きい孔の数が、1個以下である、電子部品実装構造体。 - 前記研磨面に再配線層が形成されている、請求項6に記載の電子部品実装構造体。
- 電子部品と、請求項1~5のいずれか1項に記載のエポキシ樹脂組成物と、を準備する工程と、
前記エポキシ樹脂組成物で前記電子部品を封止するとともに前記エポキシ樹脂組成物を硬化させ、前記電子部品と前記エポキシ樹脂組成物の硬化物とを含む封止体を形成する工程と、
前記硬化物の一部を研磨して研磨面を形成する工程と、
前記研磨面に再配線層を形成する工程と、を含む電子部品実装構造体の製造方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2022198771A JP7420904B2 (ja) | 2017-06-09 | 2022-12-13 | エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017114687 | 2017-06-09 | ||
| JP2017114687 | 2017-06-09 | ||
| PCT/JP2018/020706 WO2018225599A1 (ja) | 2017-06-09 | 2018-05-30 | エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022198771A Division JP7420904B2 (ja) | 2017-06-09 | 2022-12-13 | エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2018225599A1 JPWO2018225599A1 (ja) | 2020-04-09 |
| JP7237828B2 true JP7237828B2 (ja) | 2023-03-13 |
Family
ID=64566175
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2019523484A Active JP7237828B2 (ja) | 2017-06-09 | 2018-05-30 | エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 |
| JP2022198771A Active JP7420904B2 (ja) | 2017-06-09 | 2022-12-13 | エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022198771A Active JP7420904B2 (ja) | 2017-06-09 | 2022-12-13 | エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US11608435B2 (ja) |
| JP (2) | JP7237828B2 (ja) |
| KR (1) | KR102582382B1 (ja) |
| CN (1) | CN110651007A (ja) |
| TW (1) | TWI843701B (ja) |
| WO (1) | WO2018225599A1 (ja) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102581569B1 (ko) * | 2017-10-10 | 2023-10-05 | 아지노모토 가부시키가이샤 | 경화체 및 이의 제조방법, 수지 시트 및 수지 조성물 |
| JP7218575B2 (ja) * | 2018-12-28 | 2023-02-07 | 住友ベークライト株式会社 | 半導体装置の製造方法 |
| US11618835B2 (en) * | 2020-08-18 | 2023-04-04 | National Technology & Engineering Solutions Of Sandia, Llc | Method of controlled conversion of thermosetting resins and additive manufacturing thereof by selective laser sintering |
| JP2022117398A (ja) * | 2021-01-29 | 2022-08-10 | 住友ベークライト株式会社 | 封止用樹脂組成物、半導体装置の製造方法、および中空無機フィラーの検出方法 |
| WO2022220161A1 (ja) * | 2021-04-15 | 2022-10-20 | 株式会社アドマテックス | フェルール用研磨材 |
| CN114598762A (zh) * | 2022-02-25 | 2022-06-07 | 厦门聚视智创科技有限公司 | 一种手机背板去纹理方法 |
| CN117736535A (zh) * | 2022-09-15 | 2024-03-22 | 华为技术有限公司 | 树脂组合物及其应用 |
| JPWO2024100934A1 (ja) * | 2022-11-10 | 2024-05-16 | ||
| JP2025099523A (ja) * | 2023-12-21 | 2025-07-03 | 株式会社レゾナック | 樹脂組成物及び樹脂組成物の製造方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007204510A (ja) | 2006-01-31 | 2007-08-16 | Sumitomo Bakelite Co Ltd | 半導体封止用エポキシ樹脂組成物及び半導体装置 |
| JP2008130796A (ja) | 2006-11-21 | 2008-06-05 | Sumitomo Bakelite Co Ltd | 多層プリント配線板用絶縁樹脂組成物、基材付き絶縁シート、多層プリント配線板及び半導体装置 |
| JP2009009110A (ja) | 2007-05-30 | 2009-01-15 | Sumitomo Bakelite Co Ltd | 感光性接着剤樹脂組成物、接着フィルムおよび受光装置 |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS60210643A (ja) * | 1983-11-30 | 1985-10-23 | Denki Kagaku Kogyo Kk | 充填剤及びその組成物 |
| JP3434029B2 (ja) * | 1994-07-25 | 2003-08-04 | 電気化学工業株式会社 | エポキシ樹脂組成物 |
| JPH09296073A (ja) * | 1996-05-01 | 1997-11-18 | Nitto Denko Corp | 電気絶縁用注型樹脂組成物 |
| EP0933809B1 (en) * | 1998-02-02 | 2006-11-29 | Shin-Etsu Chemical Co., Ltd. | Method for mounting flip-chip semiconductor devices |
| US6303223B1 (en) * | 1998-06-16 | 2001-10-16 | Nippon Chemical Industrial Co., Ltd. | Fused spherical silica for liquid sealant and liquid sealing resin composition |
| JP4714970B2 (ja) * | 2000-08-03 | 2011-07-06 | 住友ベークライト株式会社 | エポキシ樹脂組成物、プリプレグ及びそれを用いた銅張積層板 |
| MY135619A (en) * | 2002-11-12 | 2008-05-30 | Nitto Denko Corp | Epoxy resin composition for semiconductor encapsulation, and semiconductor device using the same |
| JP3888302B2 (ja) | 2002-12-24 | 2007-02-28 | カシオ計算機株式会社 | 半導体装置 |
| JP4306318B2 (ja) * | 2003-04-25 | 2009-07-29 | コニカミノルタオプト株式会社 | 光拡散層の形成方法、光拡散フィルムとその製造方法及び光拡散層形成用のインクジェット装置 |
| US7270845B2 (en) | 2004-03-31 | 2007-09-18 | Endicott Interconnect Technologies, Inc. | Dielectric composition for forming dielectric layer for use in circuitized substrates |
| JP5005603B2 (ja) * | 2008-04-03 | 2012-08-22 | 新光電気工業株式会社 | 半導体装置及びその製造方法 |
| US9196509B2 (en) * | 2010-02-16 | 2015-11-24 | Deca Technologies Inc | Semiconductor device and method of adaptive patterning for panelized packaging |
| US8799845B2 (en) * | 2010-02-16 | 2014-08-05 | Deca Technologies Inc. | Adaptive patterning for panelized packaging |
| WO2011155638A1 (en) | 2010-06-11 | 2011-12-15 | Nec Corporation | Method of redistributing functional element |
| SG191755A1 (en) | 2011-01-28 | 2013-08-30 | Sumitomo Bakelite Co | Epoxy resin composition for sealing, and electronic component device |
| JP5864299B2 (ja) * | 2012-02-24 | 2016-02-17 | 味の素株式会社 | 樹脂組成物 |
| TWI629306B (zh) | 2013-07-19 | 2018-07-11 | Ajinomoto Co., Inc. | Resin composition |
| WO2016129541A1 (ja) | 2015-02-09 | 2016-08-18 | 味の素株式会社 | 熱硬化性樹脂組成物 |
| US9583472B2 (en) * | 2015-03-03 | 2017-02-28 | Apple Inc. | Fan out system in package and method for forming the same |
| KR20180083866A (ko) | 2015-11-18 | 2018-07-23 | 스미토모 세이카 가부시키가이샤 | 에폭시 수지 조성물, 그 제조 방법 및 해당 조성물의 용도 |
| JP6713756B2 (ja) * | 2015-11-19 | 2020-06-24 | 株式会社日本触媒 | シリカ粒子及びその製造方法 |
-
2018
- 2018-05-30 WO PCT/JP2018/020706 patent/WO2018225599A1/ja not_active Ceased
- 2018-05-30 JP JP2019523484A patent/JP7237828B2/ja active Active
- 2018-05-30 US US16/610,992 patent/US11608435B2/en active Active
- 2018-05-30 KR KR1020197032307A patent/KR102582382B1/ko active Active
- 2018-05-30 CN CN201880033290.6A patent/CN110651007A/zh active Pending
- 2018-06-06 TW TW107119454A patent/TWI843701B/zh active
-
2022
- 2022-12-13 JP JP2022198771A patent/JP7420904B2/ja active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007204510A (ja) | 2006-01-31 | 2007-08-16 | Sumitomo Bakelite Co Ltd | 半導体封止用エポキシ樹脂組成物及び半導体装置 |
| JP2008130796A (ja) | 2006-11-21 | 2008-06-05 | Sumitomo Bakelite Co Ltd | 多層プリント配線板用絶縁樹脂組成物、基材付き絶縁シート、多層プリント配線板及び半導体装置 |
| JP2009009110A (ja) | 2007-05-30 | 2009-01-15 | Sumitomo Bakelite Co Ltd | 感光性接着剤樹脂組成物、接着フィルムおよび受光装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20200016834A (ko) | 2020-02-17 |
| US11608435B2 (en) | 2023-03-21 |
| JPWO2018225599A1 (ja) | 2020-04-09 |
| TWI843701B (zh) | 2024-06-01 |
| JP7420904B2 (ja) | 2024-01-23 |
| JP2023036699A (ja) | 2023-03-14 |
| TW201903044A (zh) | 2019-01-16 |
| US20200172724A1 (en) | 2020-06-04 |
| CN110651007A (zh) | 2020-01-03 |
| WO2018225599A1 (ja) | 2018-12-13 |
| KR102582382B1 (ko) | 2023-09-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7420904B2 (ja) | エポキシ樹脂組成物、電子部品実装構造体およびその製造方法 | |
| CN108368326A (zh) | 环氧树脂组合物 | |
| TWI862483B (zh) | 密封用樹脂組成物 | |
| TW201516122A (zh) | 半導體晶片密封用熱固性樹脂片及半導體封裝之製造方法 | |
| JP5911807B2 (ja) | low−k誘電体含有半導体デバイスのためのアンダーフィルシーラントとして有用な硬化性樹脂組成物 | |
| JPWO2017038941A1 (ja) | 樹脂組成物、硬化物、封止用フィルム及び封止構造体 | |
| JP5976073B2 (ja) | 半導体装置の製造方法 | |
| US20110241227A1 (en) | Liquid resin composition and semiconductor device | |
| JP2018039992A (ja) | 樹脂組成物および該樹脂組成物を用いた三次元積層型半導体装置 | |
| JP6906228B2 (ja) | 半導体装置 | |
| JP5557158B2 (ja) | フリップチップ接続用アンダーフィル剤、及びそれを用いる半導体装置の製造方法 | |
| JP2025099748A (ja) | エポキシ樹脂組成物、硬化物、及び半導体装置 | |
| KR20210058107A (ko) | 에폭시 복합 조성물 및 이를 포함하는 반도체 패키지 | |
| KR102403966B1 (ko) | 고방열 igbt 전력 반도체 패키지 및 제조 방법 | |
| JP6388228B2 (ja) | 半導体封止用液状エポキシ樹脂組成物とそれを用いた半導体装置 | |
| JP2012167138A (ja) | プリアプライド用封止樹脂組成物、半導体チップおよび半導体装置 | |
| WO2025204095A1 (ja) | エポキシ樹脂組成物、硬化物、半導体装置、及び半導体装置の製造方法 | |
| TW202544108A (zh) | 環氧樹脂組成物、硬化物、半導體裝置,及半導體裝置之製造方法 | |
| TW201620983A (zh) | 樹脂組成物、及使用其的銅凸塊用液狀密封劑 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20210226 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220315 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220427 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20220913 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20221213 |
|
| C60 | Trial request (containing other claim documents, opposition documents) |
Free format text: JAPANESE INTERMEDIATE CODE: C60 Effective date: 20221213 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20221223 |
|
| C21 | Notice of transfer of a case for reconsideration by examiners before appeal proceedings |
Free format text: JAPANESE INTERMEDIATE CODE: C21 Effective date: 20230110 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20230207 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20230301 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7237828 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |